

Fragile X Syndrome Clinical Presentation
- Author: Jennifer A Jewell, MD, MS; Chief Editor: Maria Descartes, MD more...
- Sections Fragile X Syndrome
- Practice Essentials
- Pathophysiology
- Mortality/Morbidity
- Imaging Studies
- Other Tests
- Medical Care
- Consultations
- Further Outpatient Care
- Inpatient & Outpatient Medications
- Complications
- Patient Education
Significant family, developmental, cognitive, and neuropsychological histories are keys to diagnosis. Unusual musculoskeletal anomalies, feeding difficulties, sleep disturbances (eg, difficulty falling asleep, frequent awakening, loud snoring with/without obstructive sleep apnea), and recurrent nonspecific medical problems are infrequently reported.
Family history
Screening and diagnosis in utero or during infancy is usually the result of a family history that features multiple male relatives with intellectual disability.
Other clues to the diagnosis include a mother with learning disabilities, intellectual disability, or both or family members with ataxia and tremors.
Female infertility secondary to premature ovarian failure and increased rates of dizygotic twinning have been discovered to be more common in fragile X carriers and may provide another clue to the diagnosis.
Developmental history
During infancy (as early as age 6 months in boys), developmental milestones may be delayed, especially language development and gross motor development secondary to hypotonia. After the first year of life, delays in speech and language as well as in gross motor skills predominate. [ 14 ]
A literature review by Wheeler et al indicated that throughout childhood, boys with full mutations increase developmental tasks at about half the rate of unaffected boys. The study also found that girls with full mutations increase developmental tasks at about 75% of the rate of unaffected girls. [ 14 ]
As the patient matures, perseveration and echolalia may dominate speech patterns. Expressive language ability, short-term memory, and attempts at problem solving are significantly impaired.
Cognitive history
Intelligence quotient (IQ) frequently indicates mild-to-severe intellectual disability (20-70). Females and less-affected males may have IQs that approach 80.
IQ may be higher in childhood than in adulthood because of slowing mental development and difficulties with IQ test taking rather than loss of intellect.
IQ in patients with premutations can be normal or slightly decreased.
Neuropsychological history
Patients have many neuropsychological features, including depression, general and separation anxiety, and oppositional defiant disorder.
Autisticlike behavior (especially poor eye contact, social avoidance, and hand biting/hand flapping) is present in 16-30% of patients with fragile X syndrome. However, some patients with autisticlike behavior may have social conversation abilities. Autism is diagnosed in 20% of females and 30% of males with fragile X syndrome; furthermore, an additional 30% of patients with fragile X syndrome are diagnosed with autism spectrum disorder. Molecular investigation for fragile X syndrome is the single laboratory test proven to aid in definitively diagnosing infantile autism .
Near-universal behavioral features of males with fragile X syndrome are similar to those observed in patients with attention deficit hyperactivity disorder (ADHD), including aggressive tendencies and attention deficits.
Approximately 20% of male patients and 5% of female patients have a seizure disorder, with nearly one half of those having persistent seizures that require anticonvulsant therapy. The onset of seizures is typically at age 6-24 months. The seizure type most often diagnosed is complex partial seizure. Additionally, simple febrile partial seizures and generalized tonic-clonic seizures may be present.
Many children have difficulty when routines are altered.
Some people with fragile X syndrome display features of obsessive-compulsive disorder , sensory integration disorder, or both. Others display self-injurious behavior and significant tantrums. Anxiety disorder is common.
In a study of 154 persons with fragile X syndrome, aged 2-50 years, Reisinger et al found that, as measured using the Repetitive Behavior Scale-Revised, restricted repetitive behaviors (RRBs) peaked in severity between the ages of 7 and 12 years. The exceptions were the RRBs in the sensory-motor behavior subscale, which peaked between 2 and 12 years. The investigators also found that among the subscales, only sensory-motor behavior rates significantly differed between males and females (22.2% vs 9.2%, respectively). [ 15 ]
Musculoskeletal features
Features include pes planus, pectus excavatum , joint laxity, scoliosis, and joint dislocation.
Feeding difficulties
Affected individuals may manifest symptoms of reflux, vomiting, or both and, rarely, failure to gain weight during infancy and childhood. A minority of patients with fragile X syndrome demonstrate a Prader-Willi phenotype , which includes obesity due to severe hyperphagia.
Toileting difficulties
Patients typically have delayed toilet training and frequent enuresis after toilet training.
Recurrent nonspecific medical problems
Patients may have recurrent sinusitis , otitis media , and decreased visual acuity.
During the history taking, ask about apnea. [ 16 ]
The phenotype of fragile X syndrome is difficult to diagnose in prepubertal children. Most physical examination findings are notable only after onset of puberty.
Childhood growth is marked by an early growth spurt. However, adult height is often average or slightly below average. Additionally, obesity during adolescence and early adulthood is common.
A study by Lachiewicz et al reported 3 statistically significant phenotypic characteristics of young males with fragile X syndrome compared with young males with other developmental delays. [ 3 ] These characteristics included the presence of a hallucal crease (a single crease between the first and second toes), sensitivity to touch, and the inability to touch the tongue to the lips.
A small subset of male patients were reported with obesity, poor linear growth, small hands and feet, and diffuse hyperpimentation.
Craniofacial
Adolescent and adult patients have a long, thin face with prominent ears, prominent foreheads, facial asymmetry, a head circumference higher than the 50th percentile, and a prominent jaw.
The mouth has dental overcrowding and a high-arched palate.
Ears are typically large and may protrude.
Strabismus is frequently noted. Occasionally, nystagmus, astigmatism, and ptosis are present.
Extremities
Hands and feet manifest nonspecific findings, including hyperextensible finger joints, hand calluses, double-jointed thumbs, a single palmar crease, and pes planus. Clubfeet may be present at birth.
Back and chest
Pectus excavatum and scoliosis are frequent findings.
Macroorchidism is nearly universal in postpubertal males. In unaffected males, average testicular volume is 17 mL; in patients with fragile X syndrome, testicular volume is more than 25 mL and can be as high as 120 mL. During childhood, an increased incidence of inguinal hernias is reported.
A heart murmur or click consistent with mitral valve prolapse is often auscultated and requires consultation with a cardiologist.
The genetic defect is dynamic and lies at the distal end of the long arm of the X chromosome. Careful examination of the karyotype of affected individuals' lymphocytes, cultured in a folate-depleted and thymidine-depleted medium, reveals a constriction followed by a thin strand of genetic material that extends beyond the long arm at the highly conserved band Xq27.3. This constriction and thin strand produce the appearance of a fragile portion of the X chromosome, leading to the term fragile X.
The function of the band Xq27.3, which is also termed the FMR1 gene, is to synthesize FMRP, a regulatory protein that binds messenger RNA (mRNA) in neurons and dendrites. [ 17 ] In patients with a full mutation in the FMR1 gene, FMRP is not manufactured because of hypermethylation of FMR1 , and brain development is impaired primarily because of abnormal synapse connections. Additionally, mutations in the FMR1 gene lead to excessive activity of the metabotropic glutamate receptor 5 (mGluR5), which results in many fragile X syndrome symptoms. FMRP is present in other tissues; however, its role is less understood.
Once identified and sequenced, the gene was discovered to contain a repeating base pair triplet (CGG) expansion, which is responsible for fragile X syndrome.
Unaffected individuals have 5-54 CGG repeats in the first exon at the 5' end of band Xq27.3. Individuals with 45-54 repeats are unaffected, but they risk passing a premutation on to future generations. A span of 55-199 repeats is known as a premutation, whereas 200 or more repeats is a full mutation. Full mutation results in hypermethylation of the cysteine bases and restricts protein binding, leading to gene inactivation and absent FMRP. Mosaic patterns are common. The number of repeats is unstable from generation to generation, making the pattern of inheritance difficult to predict. In addition, the degree of methylation is directly proportional to the signs and symptoms of fragile X syndrome.
Males with a full mutation have fragile X syndrome. Mothers of nearly all males with fragile X syndrome have premutation or fragile X syndrome. Males with fragile X syndrome pass a premutation to their daughters because sperm cells are mosaics. Sons are unaffected because they receive the Y chromosome from their fathers.
Half of females with the full mutation on a single X chromosome are unaffected because of inactivation of the other X chromosome. The other half of females have fragile X syndrome, although with less severe intellectual disability than males with the disorder. These affected females can pass the gene to their children.
Males with a premutation are usually unaffected to mildly affected and transmit the premutation to their daughters. The mutation is stable; thus, the CGG triplets are not increased. Sons of affected males are unaffected because they receive the Y chromosome from their fathers.
Females with a premutation are usually unaffected to mildly affected with fragile X syndrome. However, they have a 20% chance of having fragile X – associated primary ovarian insufficiency. Unlike their male counterparts, the CGG triplets are unstable and increase in size during oogenesis. If the number of repeats exceeds 200 and the oocyte is fertilized, a male child will have fragile X syndrome, and a female child will have a 50% chance of having fragile X syndrome. The number of repeats is directly proportional to the risk of the disorder in an offspring.
Although most patients with fragile X syndrome have a CGG triplet expansion, few patients have a point mutation in the FMR1 gene or a deletion of the gene. [ 18 ]
No spontaneous FMR1 full mutations have been reported. [ 19 ]
A study by Hall et al indicated that in fragile X syndrome, the microstructure of white matter is abnormal in areas of the inferior longitudinal and uncinate fasciculi. The study, included 20 patients with fragile X syndrome and 20 matched controls, found a significant increase in fractional anisotropy in the left and right inferior longitudinal fasciculi, the right uncinate fasciculus, and the left cingulum hippocampus, in the fragile X syndrome group, as well as a significant reduction in mean diffusivity in the right inferior longitudinal fasciculus. The investigators stated that the abnormalities probably result from inefficient synaptic pruning, caused by decreased or absent FMRP. [ 20 ]
Stone WL, Basit H, Los E. Fragile X Syndrome. StatPearls . 2022 Jan. [QxMD MEDLINE Link] . [Full Text] .
Kidd SA, Lachiewicz A, Barbouth D, et al. Fragile X syndrome: a review of associated medical problems. Pediatrics . 2014 Nov. 134 (5):995-1005. [QxMD MEDLINE Link] . [Full Text] .
Lachiewicz AM, Dawson DV, Spiridigliozzi GA. Physical characteristics of young boys with fragile X syndrome: reasons for difficulties in making a diagnosis in young males. Am J Med Genet . 2000 Jun 5. 92(4):229-36. [QxMD MEDLINE Link] .
Yrigollen CM, Durbin-Johnson B, Gane L, et al. AGG interruptions within the maternal FMR1 gene reduce the risk of offspring with fragile X syndrome. Genet Med . 2012 Aug. 14(8):729-36. [QxMD MEDLINE Link] .
Martin JP, Bell J. A pedigree of mental defect showing sex-linkage. J Neurol Psychi . 1943. 6:154-7.
Lubs HA. A marker X chromosome. Am J Hum Genet . 1969 May. 21(3):231-44. [QxMD MEDLINE Link] . [Full Text] .
Myers KA, van 't Hof FNG, Sadleir LG, et al. Fragile Females: Case Series of Epilepsy in Girls With FMR1 Disruption. Pediatrics . 2019 Sep. 144 (3): [QxMD MEDLINE Link] .
Murphy MM. A review of mathematical learning disabilities in children with fragile X syndrome. Dev Disabil Res Rev . 2009. 15(1):21-7. [QxMD MEDLINE Link] .
De Smedt B, Swillen A, Verschaffel L, Ghesquiere P. Mathematical learning disabilities in children with 22q11.2 deletion syndrome: a review. Dev Disabil Res Rev . 2009. 15(1):4-10. [QxMD MEDLINE Link] .
Del Hoyo Soriano L, Thurman AJ, Harvey DJ, Ted Brown W, Abbeduto L. Genetic and maternal predictors of cognitive and behavioral trajectories in females with fragile X syndrome. J Neurodev Disord . 2018 Jun 20. 10 (1):22. [QxMD MEDLINE Link] . [Full Text] .
Spath MA, Feuth TB, Smits AP, Yntema HG, Braat DD, Thomas CM. Predictors and risk model development for menopausal age in fragile X premutation carriers. Genet Med . 2011 Jul. 13(7):643-50. [QxMD MEDLINE Link] .
Abrams L, Cronister A, Brown WT, Tassone F, Sherman SL, Finucane B, et al. Newborn, carrier, and early childhood screening recommendations for fragile x. Pediatrics . 2012 Dec. 130(6):1126-35. [QxMD MEDLINE Link] .
Bailey DB Jr, Raspa M, Bishop E, Holiday D. No change in the age of diagnosis for fragile x syndrome: findings from a national parent survey. Pediatrics . 2009 Aug. 124(2):527-33. [QxMD MEDLINE Link] .
Wheeler AC, Gwaltney A, Raspa M, et al. Emergence of Developmental Delay in Infants and Toddlers With an FMR1 Mutation. Pediatrics . 2021 May. 147 (5): [QxMD MEDLINE Link] . [Full Text] .
Reisinger DL, Shaffer RC, Tartaglia N, Berry-Kravis E, Erickson CA. Delineating Repetitive Behavior Profiles across the Lifespan in Fragile X Syndrome. Brain Sci . 2020 Apr 17. 10 (4): [QxMD MEDLINE Link] . [Full Text] .
de Vries BB, Halley DJ, Oostra BA, Niermeijer MF. The fragile X syndrome. J Med Genet . 1998 Jul. 35(7):579-89. [QxMD MEDLINE Link] .
Hagerman RJ, Berry-Kravis E, Kaufmann WE, Ono MY, Tartaglia N, Lachiewicz A, et al. Advances in the treatment of fragile X syndrome. Pediatrics . 2009 Jan. 123(1):378-90. [QxMD MEDLINE Link] .
Oostra BA, Willemsen R. FMR1: a gene with three faces. Biochim Biophys Acta . 2009 Feb 19. [QxMD MEDLINE Link] .
Smits AP, Dreesen JC, Post JG, Smeets DF, de Die-Smulders C, Spaans-van der Bijl T, et al. The fragile X syndrome: no evidence for any recent mutations. J Med Genet . 1993 Feb. 30(2):94-6. [QxMD MEDLINE Link] . [Full Text] .
Hall SS, Dougherty RF, Reiss AL. Profiles of aberrant white matter microstructure in fragile X syndrome. Neuroimage Clin . 2016. 11:133-8. [QxMD MEDLINE Link] . [Full Text] .
Health supervision for children with fragile x syndrome. Pediatrics . 2011 May. 127(5):994-1006. [QxMD MEDLINE Link] .
Leigh MJS, Nguyen DV, Mu Y, et al. A Randomized Double-Blind, Placebo-Controlled Trial of Minocycline in Children and Adolescents with Fragile X Syndrome. J Dev Behav Pediatr . 2013. 34:147–155. [Full Text] .

Contributor Information and Disclosures
Jennifer A Jewell, MD, MS Assistant Professor of Pediatrics, Tufts University School of Medicine; Pediatric Hospitalist, The Barbara Bush Children's Hospital at Maine Medical Center Jennifer A Jewell, MD, MS is a member of the following medical societies: American Academy of Pediatrics , Massachusetts Medical Society , Sigma Xi, The Scientific Research Honor Society Disclosure: Nothing to disclose.
Mary L Windle, PharmD Adjunct Associate Professor, University of Nebraska Medical Center College of Pharmacy; Editor-in-Chief, Medscape Drug Reference Disclosure: Nothing to disclose.
Eric T Rush, MD, FAAP Professor of Pediatrics, University of Missouri-Kansas City School of Medicine; Clinical Geneticist, Children's Mercy Hospital of Kansas City Eric T Rush, MD, FAAP is a member of the following medical societies: American Academy of Pediatrics , American College of Physicians Disclosure: Serve(d) as a director, officer, partner, employee, advisor, consultant or trustee for: Alexion Pharmaceuticals, Ultragenyx Pharmaceutical, Biomarin Pharmaceuticals, ObsEva, Inozyme Pharma, Ascendis Pharma<br/>Serve(d) as a speaker or a member of a speakers bureau for: Alexion Pharmaceuticals, Ultragenyx Pharmaceutical, and Biomarin Pharmaceuticals<br/>Received research grant from: Alexion Pharmaceuticals, Ultragenyx Pharmaceuticals.
Maria Descartes, MD Medical Geneticist, Miami Cancer Institute, Baptist Health South Florida Maria Descartes, MD is a member of the following medical societies: American Academy of Pediatrics , American College of Medical Genetics and Genomics , American Medical Association , American Society of Human Genetics , International Skeletal Dysplasia Society , Society for Inherited Metabolic Disorders , Southeastern Regional Genetics Group Disclosure: Nothing to disclose.
Michael Fasullo, PhD Senior Scientist, Ordway Research Institute; Associate Professor, State University of New York at Albany; Adjunct Associate Professor, Center for Immunology and Microbial Disease, Albany Medical College Michael Fasullo, PhD is a member of the following medical societies: Radiation Research Society , American Society for Biochemistry and Molecular Biology , Genetics Society of America , Environmental Mutagenesis and Genomics Society Disclosure: Nothing to disclose.
What would you like to print?
- Print this section
- Print the entire contents of
- Print the entire contents of article

- Genetics of Autism Spectrum Disorders
- PET Scanning in Autism Spectrum Disorder
- Autism Spectrum Disorder
- Rapid Review Quiz: Identifying and Managing Autism Spectrum Disorder
- Fast Five Quiz: Autism Spectrum Disorder
- Pervasive Developmental Disorder
- A 20-Year-Old Man Who Stopped Speaking
- Positive Results for Intranasal Oxytocin in Adults With Autism
- Strong Sibling Link With Autism Spectrum Disorder
- Infertility Linked With Slightly Higher Autism Risk in Children

- Drug Interaction Checker
- Pill Identifier
- Calculators

- 2010/viewarticle/1001292 Rapid Review Quiz: Identifying and Managing Autism Spectrum Disorder
A .gov website belongs to an official government organization in the United States.
A lock ( ) or https:// means you've safely connected to the .gov website. Share sensitive information only on official, secure websites.
- Data and Statistics
- Fragile X Syndrome Articles
- Fragile X Associated Disorders
- Free Materials on Fragile X Syndrome
- Fragile X Syndrome Related Concerns
- Healthcare Providers: Top 5 things to know about FXS
- View All Home
About Fragile X Syndrome
- Fragile X syndrome (FXS) is a genetic disorder and one of the most common causes of inherited intellectual disability.
- FXS affects both males and females. However, females often have milder symptoms than males.
- A diagnosis of FXS can be helpful to the family because it can provide a reason for a child’s intellectual disabilities and behavior problems.
- There is no cure for FXS. However, treatment services can help people learn important skills. Taking advantage of all the resources available will help guide success.

What is fragile X syndrome (FXS)?
Fragile X syndrome (FXS) is a genetic disorder caused by changes in a gene called fragile X messenger ribonucleoprotein 1 ( FMR1). FMR1 usually makes a protein called FMRP that is needed for brain development. People who have FXS do not make this protein. Those with fragile X-associated disorders have changes in the FMR1 gene but usually still make some of the protein.
FXS affects both males and females. However, females often have milder symptoms than males. The exact number of people who have FXS is unknown, but a review of research studies estimated that about 1 in 7,000 males and about 1 in 11,000 females have been diagnosed with FXS. 1
Signs and symptoms
Signs that a child might have FXS include
- Developmental delays (not sitting, walking, or talking at the same time as other children the same age);
- Learning disabilities (trouble learning new skills); and
- Social and behavior problems (such as not making eye contact, anxiety, trouble paying attention, hand flapping, acting and speaking without thinking, and being very active).
Males who have FXS usually have some degree of intellectual disability that can range from mild to severe. Females with FXS can have normal intelligence or some degree of intellectual disability. Autism spectrum disorder (ASD) also occurs more frequently in people with FXS.

Testing and diagnosis
FXS can be diagnosed by testing a person's DNA from a blood test. A doctor or genetic counselor can order the test. Testing also can be done to find changes in the FMR1 gene that can lead to fragile X-associated disorders.
A diagnosis of FXS can be helpful to the family because it can provide a reason for a child’s intellectual disabilities and behavior problems. This allows the family and other caregivers to learn more about the disorder and manage care so that the child can reach their full potential. However, the results of DNA tests can affect other family members and raise many issues. So, anyone who is thinking about FXS testing should consider having genetic counseling prior to getting tested.
Uncover the Facts
• FXS Facts for Families
• Providers: Top 5 Things to Know About FXS
There is no cure for FXS. However, treatment services can help people learn important skills. Services can include therapy to learn to talk, walk, and interact with others. In addition, medicine can be used to help control some issues, such as behavior problems.
To develop the best treatment plan, people with FXS, parents, and healthcare providers should work closely with one another, and with everyone involved in treatment and support—which may include teachers, childcare providers, coaches, therapists, and other family members. Taking advantage of all the resources available will help guide success.
Early intervention services
Early intervention services help children from birth to 3 years old (36 months) learn important skills. These services may improve a child’s development. Even if the child has not been diagnosed with FXS, they may be eligible for services. These services are provided through an early intervention system in each state. Through this system, you can ask for an evaluation. In addition, treatment for particular symptoms, such as speech therapy for language delays, often does not need to wait for a formal diagnosis. While early intervention is extremely important, treatment services at any age can be helpful.
What to do if you think your child might have FXS
Local public-school systems can provide services and support for children aged 3 years and older. Children can access some services even if they do not attend public school.
When parents are concerned about a child's development, it can be very challenging for them to figure out the right steps to take. States have created parent centers to help. These centers help families learn how and where to have their children evaluated and how to find services. For information about services in your state, you can access your state's parent center .
Finding support
Having support and community resources can help increase confidence in managing FXS, enhance quality of life, and assist in meeting the needs of all family members. It might be helpful for parents of children with FXS to talk with one another. One parent might have learned how to address some of the same concerns another parent has. Often, parents of children with special needs can give advice about good resources for these children.

Remember that the choices of one family might not be best for another family, so it’s important that parents understand all options and discuss them with their child’s healthcare providers.
- Contact the National Fragile X Foundation at 1-800-688-8765 or [email protected] to get information about treatments, educational strategies, therapies and intervention.
- Connect with a Community Support Network (CSN) at the National Fragile X Foundation. CSNs are organized and run by parent volunteers and provide support to families.
What CDC is doing
CDC is working to learn more about the natural history of fragile X so that better approaches to intervention can be developed.
As part of this effort, CDC
Supported the National Fragile X Foundation to develop the Fragile X Online Registry With Accessible Research Database (FORWARD) . The purpose of FORWARD is to learn more about
- Other conditions that commonly occur along with FXS,
- The impact on the day-to-day lives of individuals living with FXS and their families,
- Short-term and long-term outcomes, and
- What type of interventions and support are most effective for different individuals and their families.
Collaborated with the American Academy of Pediatrics to develop and distribute educational materials to healthcare professionals and families.
- These materials are designed to raise awareness of FXS and encourage early diagnosis so that people with FXS can receive appropriate care and services.
- Developed a free virtual PediaLink training on FXS. This course is designed to increase healthcare provider understanding of the symptoms of FXS to help promote earlier identification and diagnosis.
Moving Fragile X Syndrome Research FORWARD
Fragile X Online Registry With Accessible Research Database (FORWARD) — Funded by CDC, FORWARD is the largest source of data on people with FXS and their families.
FRAXA Research Foundation — FRAXA’s mission is to accelerate progress toward effective treatments and ultimately a cure for fragile X by directly funding the most promising research. FRAXA also supports families affected by FXS.
National Institutes of Health, Office of Rare Diseases Research, Genetic and Rare Diseases Information Center — The Office of Rare Diseases Research (ORDR) answers questions about rare diseases for patients, families, healthcare providers, researchers, students and educators. The ORDR website provides information about National Institutes of Health-sponsored biomedical research, scientific conferences, and rare and genetic diseases.
- Hunter J, Rivero-Arias O, Angelov A, Kim E, Fotheringham I, Leal J. Epidemiology of fragile X syndrome: a systematic review and meta-analysis . Am J Med Genet A. 2014 Jul 164A(7): 1648-58.
Fragile X Syndrome (FXS)
Fragile X syndrome (FXS) is one of the most common causes of inherited intellectual disability.
For Everyone
Health care providers.
Europe PMC requires Javascript to function effectively.
Either your web browser doesn't support Javascript or it is currently turned off. In the latter case, please turn on Javascript support in your web browser and reload this page.
- DOI: 10.24875/gmm.m19000322
- Corpus ID: 211047039
Fragile X syndrome: clinical presentation, pathology and treatment
- M. Salcedo-Arellano , R. Hagerman , V. Martínez‐Cerdeño
- Published in Gaceta de M�xico 6 February 2020
- Gaceta de M�xico
Figures and Tables from this paper

19 Citations
Fragile x syndrome: from molecular aspect to clinical treatment.
- Highly Influenced
Phenotypic variability to medication management: an update on fragile X syndrome
Molecular mechanisms for targeted treatments in fragile x syndrome, fragile x syndrome and fetal alcohol syndrome: occurrence of dual diagnosis in a set of triplets, raising knowledge and awareness of fragile x syndrome in serbia, georgia, and colombia: a model for other developing countries, channelopathies in fragile x syndrome, genetic modifiers of repeat expansion disorders, serum matrix metalloproteinase-9 (mmp9) and amyloid-beta protein precursor (app) as potential biomarkers in children with fragile-x syndrome: a cross sectional study., reduced lynx1 expression in transcriptome of human ipsc-derived neural progenitors modeling fragile x syndrome, circadian rhythm and sleep analyses in a fruit fly model of fragile x syndrome using a video-based automated behavioral research system, 38 references, fragile x syndrome, [fragile x associated tremor/ataxia syndrome: its clinical presentation, pathology, and treatment]., fragile x-associated tremor/ataxia syndrome — features, mechanisms and management, [prevention of fragile x syndrome by prenatal genetic diagnosis: advantages and controversial aspects]., fragile x syndrome and connective tissue dysregulation, the fragile x protein and genome function, comprehensive evaluation of the child with intellectual disability or global developmental delays, genetic cluster of fragile x syndrome in a colombian district, clinical and molecular implications of mosaicism in fmr1 full mutations, fragile x and app: a decade in review, a vision for the future, related papers.
Showing 1 through 3 of 0 Related Papers

DANIEL J. WATTENDORF, MAJ, MC, USAF, AND MAXIMILIAN MUENKE, M.D.
Am Fam Physician. 2005;72(01):111-113
See editorial on page 33.
Author disclosure: Nothing to disclose.
To complement the 2005 Annual Clinical Focus on medical genomics, AFP will be publishing a series of short reviews on genetic syndromes. This series was designed to increase awareness of these diseases so that family physicians can recognize and diagnose children with these disorders and understand the kind of care they might require in the future. The first review in this series discusses fragile X syndrome.
Fragile X syndrome is caused by an expansion mutation in the Fragile X mental retardation 1 (FMR1) gene located on the X chromosome It characteristically leads to some degree of mental retardation. The phenotype is subtle, with minor dysmorphic features and developmental delay during childhood. Characteristic features during adolescence are an elongated face, prominent jaw, large ears, macro-orchidism, and a range of behavioral anomalies and cognitive deficits ( Figure 1 ) . Recently recognized manifestations in premutation carriers include premature ovarian failure and tremor/ataxia. 1 Premature ovarian failure occurs in up to 20 percent of women who are premutation carriers of the FMR1 gene. 1 Fragile X–associated tremor/ataxia syndrome (FXTAS) affects 30 percent of premutation carrier men between the ages of 50 and 60 years, and its prevalence increases with age. 1

Epidemiology
Fragile X syndrome, the phenotype associated with full mutation, occurs in approximately one in 4,000 men and one in 6,000 to 8,000 women. The premutation in the FMR1 gene occurs in approximately one in 800 men and up to one in 100 to 200 women. Premutation male carriers are susceptible to FXTAS after age 50. Premature ovarian failure may be detected in as many as one third of women with a premutation.
Clinical Presentation
Although fragile X syndrome occurs in males and females, females generally present with milder symptoms. The first clinical clue in children often is delayed attainment of one or more developmental milestones. 2 , 3 On average, boys with fragile X syndrome sit without support at 10 months of age and walk and talk at 20 months. 2 With few exceptions, affected males have mental retardation, generally of moderate degree. About one third of affected females have mild to severe mental retardation. 2 There is a specific pattern of deficits in abstract reasoning, sequential processing, and mathematics. Clinical findings during early childhood may include macrocephaly and frontal bossing (unusually prominent forehead). After puberty, macro-orchidism is present in affected men. Additional findings may include strabismus and mild connective tissue dysplasia, such as mitral valve prolapse, hyperextensible joints, and pes planus. Behavior is characterized by attention deficits, hand flapping, hand biting, and gaze aversion. Family physicians are most likely to encounter the undiagnosed child before school age, when formal testing can confirm cognitive deficits. However, the average age of diagnosis currently is eight years, reflecting the subtlety of features in young children. 4
FXTAS is a neurodegenerative disorder with progressive intention tremor and cerebellar ataxia. 5 Affected persons present with parkinsonism, peripheral neuropathies, and dementia after age 50 years.
Premature ovarian failure may occur as an isolated clinical finding in women with premutations. 1 , 6 Follicle-stimulating hormone (FSH) levels are elevated in these women even before the onset of premature ovarian failure. Approximately 1 percent of women in the general population have premature ovarian failure, but the prevalence in women with a premutation is 30 times higher. 6 Women who are infertile and have prematurely elevated FSH levels should be considered for carrier status testing of the FMR1 premutation. A family history of FXTAS or premature ovarian failure in a child with cognitive deficits is another indicator to initiate genetic testing for fragile X syndrome ( Figure 2 ) .
The diagnosis of fragile X syndrome is confirmed by molecular genetic testing of the FMR1 gene. Prenatal testing is available. FMR1 is characterized by a repetitive CGG trinucleotide sequence, which is repeated six to 50 times in unaffected persons ( Figure 2 ) . A full mutation consists of more than 200 CGG repeats in the FMR1 gene, plus hypermethylation, which leads to an inability to produce the FMR1 protein. Almost all males and more than one half of females with full mutations have fragile X syndrome. 2 Premutation carriers, who have between 50 and 200 CGG repeats, are not cognitively affected but may have physical or psychiatric findings. In addition, they are susceptible to developing premature ovarian failure and FXTAS. Rarely, the fragile X phenotype occurs in a premutation carrier if hypermethylation is present. Conversely, the phenotype may be absent in a person with a full mutation without hypermethylation, confirming that fragile X syndrome results from the absence of FMR1 protein.
Genetic Counseling and Inheritance
Fragile X syndrome is an X-linked inherited disorder. It is important to diagnose affected patients as early as possible to provide early intervention and supportive care (i.e., specific developmental therapy and an individualized education plan) and to inform parents for further family planning. One half of families in a 2002 survey 4 reported having an additional child with fragile X syndrome before the older affected child was diagnosed. Family history collection should include questions about other family members, with particular attention to developmental delay, mental retardation, and psychiatric disorders. In addition, a family history of women with premature ovarian failure and men with FXTAS should be ascertained. A positive family history in a proband with developmental delay should prompt consideration of genetic testing of the FMR1 gene. The American College of Medical Genetics 7 recommends testing, regardless of family history, for all males and females with mental retardation of unknown etiology.
Treatment is supportive, requiring a multidisciplinary team and including anxiety-reducing measures, behavior modification, and medications to manage associated psychiatric disorders. Individual education plans are necessary for school-age children.
Additional information about the diagnosis and management of fragile X syndrome is available at the following Web sites: National Fragile X Foundation ( http://www.FragileX.org ); GeneTests ( http://www.genetests.org ); and the American College of Medical Genetics (www.acmg.net/resources/policies/pol-014.asp).
Hagerman PJ, Hagerman RJ. The fragile-X premutation: a maturing perspective [published correction appears in Am J Hum Genet 2004;75:352].. Am J Hum Genet. 2004;74:805-16.
Maes B, Fryns JP, Ghesquiere P, Borghgraef M. Phenotypic checklist to screen for fragile X syndrome in people with mental retardation.. Ment Retard. 2000;38:207-15.
Shevell M, Ashwal S, Donley D, Flint J, Gingold M, Hirtz D, et al. Practice parameter: evaluation of the child with global developmental delay: report of the Quality Standards Subcommittee of the American Academy of Neurology and The Practice Committee of the Child Neurology Society.. Neurology. 2003;60:367-80.
Delayed diagnosis of fragile X syndrome—United States, 1990–1999.. MMWR Morb Mortal Wkly Rep. 2002;51:740-2.
Jacquemont S, Hagerman RJ, Leehey MA, Hall DA, Levine RA, Brunberg JA, et al. Penetrance of the fragile X-associated tremor/ataxia syndrome in a premutation carrier population.. JAMA. 2004;291:460-9.
Machado-Ferreira Mdo C, Costa-Lima MA, Boy RT, Esteves GS, Pimentel MM. Premature ovarian failure and FRAXA premutation: positive correlation in a Brazilian survey.. Am J Med Genet A. 2004;126:237-40.
American College of Medical Genetics. Fragile X syndrome: diagnostic and carrier testing. Accessed online May 31, 2005, at: http://www.acmg.net/resources/policies/pol-014.asp.
Continue Reading
More in afp.
Copyright © 2005 by the American Academy of Family Physicians.
This content is owned by the AAFP. A person viewing it online may make one printout of the material and may use that printout only for his or her personal, non-commercial reference. This material may not otherwise be downloaded, copied, printed, stored, transmitted or reproduced in any medium, whether now known or later invented, except as authorized in writing by the AAFP. See permissions for copyright questions and/or permission requests.
Copyright © 2024 American Academy of Family Physicians. All Rights Reserved.
Information
- Author Services
Initiatives
You are accessing a machine-readable page. In order to be human-readable, please install an RSS reader.
All articles published by MDPI are made immediately available worldwide under an open access license. No special permission is required to reuse all or part of the article published by MDPI, including figures and tables. For articles published under an open access Creative Common CC BY license, any part of the article may be reused without permission provided that the original article is clearly cited. For more information, please refer to https://www.mdpi.com/openaccess .
Feature papers represent the most advanced research with significant potential for high impact in the field. A Feature Paper should be a substantial original Article that involves several techniques or approaches, provides an outlook for future research directions and describes possible research applications.
Feature papers are submitted upon individual invitation or recommendation by the scientific editors and must receive positive feedback from the reviewers.
Editor’s Choice articles are based on recommendations by the scientific editors of MDPI journals from around the world. Editors select a small number of articles recently published in the journal that they believe will be particularly interesting to readers, or important in the respective research area. The aim is to provide a snapshot of some of the most exciting work published in the various research areas of the journal.
Original Submission Date Received: .
- Active Journals
- Find a Journal
- Proceedings Series
- For Authors
- For Reviewers
- For Editors
- For Librarians
- For Publishers
- For Societies
- For Conference Organizers
- Open Access Policy
- Institutional Open Access Program
- Special Issues Guidelines
- Editorial Process
- Research and Publication Ethics
- Article Processing Charges
- Testimonials
- Preprints.org
- SciProfiles
- Encyclopedia

Article Menu

- Subscribe SciFeed
- Recommended Articles
- PubMed/Medline
- Google Scholar
- on Google Scholar
- Table of Contents
Find support for a specific problem in the support section of our website.
Please let us know what you think of our products and services.
Visit our dedicated information section to learn more about MDPI.
JSmol Viewer
Fragile x syndrome: from molecular aspect to clinical treatment.

1. FMR1 Gene and Fragile X Mental Retardation Protein
2. the pathophysiology of fragile x syndrome, brain structure and neuronal morphology, 3. clinical presentation of fragile x syndrome, 4. non-pharmacological therapy of fxs, 5. psychopharmacology in fxs, 5.1. attention deficit hyperactivity disorder (adhd) symptoms, 5.2. anxiety symptoms, 5.3. aggressive behavior, 5.4. sleep problems, 5.5. seizures, 6. targeted treatments, 6.1. phosphodiesterase-4d (pde4d) inhibitor bpn14770, 6.2. cannabidiol (cbd), 6.3. metformin, 6.4. minocycline, 7. conclusions, author contributions, institutional review board statement, informed consent statement, data availability statement, conflicts of interest.
- Verkerk, A.J.; Pieretti, M.; Sutcliffe, J.S.; Fu, Y.-H.; Kuhl, D.P.; Pizzuti, A.; Reiner, O.; Richards, S.; Victoria, M.F.; Zhang, F.; et al. Identification of a gene (FMR-1) containing a CGG repeat coincident with a breakpoint cluster region exhibiting length variation in fragile X syndrome. Cell 1991 , 65 , 905–914. [ Google Scholar ] [ CrossRef ]
- Weiler, I.J.; Irwin, S.A.; Klintsova, A.; Spencer, C.M.; Brazelton, A.D.; Miyashiro, K.; Comery, T.A.; Patel, B.; Eberwine, J.; Greenough, W.T. Fragile X mental retardation protein is translated near synapses in response to neurotransmitter activation. Proc. Natl. Acad. Sci. USA 1997 , 94 , 5395–5400. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Feng, Y.; Gutekunst, C.A.; Eberhart, D.E.; Yi, H.; Warren, S.T.; Hersch, S.M. Fragile X mental retardation protein: Nucleocytoplasmic shuttling and association with somatodendritic ribosomes. J. Neurosci. 1997 , 17 , 1539–1547. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Hagerman, P.J. The fragile X prevalence paradox. J. Med. Genet. 2008 , 45 , 498–499. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Salcedo-Arellano, M.J.; Hagerman, R.J.; Martinez-Cerdeno, V. Fragile X syndrome: Clinical presentation, pathology and treatment. Gac. Med. Mex. 2020 , 156 , 60–66. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Tassone, F.; Iong, K.P.; Tong, T.H.; Lo, J.; Gane, L.W.; Berry-Kravis, E.; Nguyen, D.; Mu, L.Y.; Laffin, J.; Bailey, D.B.; et al. FMR1 CGG allele size and prevalence ascertained through newborn screening in the United States. Genome Med. 2012 , 4 , 100. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Ligsay, A.; Hagerman, R.J. Review of targeted treatments in fragile X syndrome. Intractable Rare Dis. Res. 2016 , 5 , 158–167. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Irwin, S.A.; Galvez, R.; Greenough, W.T. Dendritic spine structural anomalies in fragile-X mental retardation syndrome. Cereb. Cortex 2000 , 10 , 1038–1044. [ Google Scholar ] [ CrossRef ]
- Deng, P.Y.; Klyachko, V.A. Channelopathies in fragile X syndrome. Nat. Rev. Neurosci. 2021 , 22 , 275–289. [ Google Scholar ] [ CrossRef ]
- Bagni, C.; Zukin, R.S. A Synaptic Perspective of Fragile X Syndrome and Autism Spectrum Disorders. Neuron 2019 , 101 , 1070–1088. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Verheij, C.; Bakker, C.E.; de Graaff, E.; Keulemans, J.; Willemsen, R.; Verkerk, A.J.; Galjaard, H.; Reuser, A.J.; Hoogeveen, A.T.; Oostra, B.A. Characterization and localization of the FMR-1 gene product associated with fragile X syndrome. Nature 1993 , 363 , 722–724. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Zafarullah, M.; Tassone, F. Molecular Biomarkers in Fragile X Syndrome. Brain Sci. 2019 , 9 , 96. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Greenough, W.T.; Klintsova, A.Y.; Irwin, S.A.; Galvez, R.; Bates, K.E.; Weiler, I.J. Synaptic regulation of protein synthesis and the fragile X protein. Proc. Natl. Acad. Sci. USA 2001 , 98 , 7101–7106. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Gross, C.; Hoffmann, A.; Bassell, G.J.; Berry-Kravis, E.M. Therapeutic Strategies in Fragile X Syndrome: From Bench to Bedside and Back. Neurotherapeutics 2015 , 12 , 584–608. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Braat, S.; D’Hulst, C.; Heulens, I.; De Rubeis, S.; Mientjes, E.; Nelson, D.L.; Willemsen, R.; Bagni, C.; Van Dam, D.; De Deyn, P.P.; et al. The GABAA receptor is an FMRP target with therapeutic potential in fragile X syndrome. Cell Cycle 2015 , 14 , 2985–2995. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Braat, S.; Kooy, R.F. The GABAA Receptor as a Therapeutic Target for Neurodevelopmental Disorders. Neuron 2015 , 86 , 1119–1130. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Brown, M.R.; Kronengold, J.; Gazula, V.R.; Chen, Y.; Strumbos, J.G.; Sigworth, F.J.; Navaratnam, D.; Kaczmarek, L.K. Fragile X mental retardation protein controls gating of the sodium-activated potassium channel Slack. Nat. Neurosci. 2010 , 13 , 819–821. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Deng, P.Y.; Rotman, Z.; Blundon, J.A.; Cho, Y.; Cui, J.; Cavalli, V.; Zakharenko, S.S.; Klyachko, V.A. FMRP regulates neurotransmitter release and synaptic information transmission by modulating action potential duration via BK channels. Neuron 2013 , 77 , 696–711. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Ferron, L. Fragile X mental retardation protein controls ion channel expression and activity. J. Physiol. 2016 , 594 , 5861–5867. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Darnell, J.C.; Van Driesche, S.J.; Zhang, C.; Hung, K.Y.; Mele, A.; Fraser, C.E.; Stone, E.F.; Chen, C.; Fak, J.J.; Chi, S.W.; et al. FMRP stalls ribosomal translocation on mRNAs linked to synaptic function and autism. Cell 2011 , 146 , 247–261. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Dockendorff, T.C.; Labrador, M. The Fragile X Protein and Genome Function. Mol. Neurobiol. 2019 , 56 , 711–721. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Hagerman, R.J.; Hagerman, P.J. Testing for fragile X gene mutations throughout the life span. JAMA 2008 , 300 , 2419–2421. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Sutcliffe, J.S.; Nelson, D.L.; Zhang, F.; Pieretti, M.; Caskey, C.T.; Saxe, D.; Warren, S.T. DNA methylation represses FMR-1 transcription in fragile X syndrome. Hum. Mol. Genet. 1992 , 1 , 397–400. [ Google Scholar ] [ CrossRef ]
- Saldarriaga, W.; Tassone, F.; González-Teshima, L.Y.; Forero-Forero, J.V.; Ayala-Zapata, S.; Hagerman, R. Fragile X Syndrome. Colomb. Med. 2014 , 45 , 190–198. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Thurman, A.J.; McDuffie, A.; Hagerman, R.; Abbeduto, L. Psychiatric symptoms in boys with fragile X syndrome: A comparison with nonsyndromic autism spectrum disorder. Res. Dev. Disabil. 2014 , 35 , 1072–1086. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Budimirovic, B.; Schlageter, A.; Filipovic-Sadic, S.; Protic, D.D.; Bram, E.; Mahone, E.M.; Nicholson, K.; Culp, K.; Javanmardi, K.; Kemppainen, J.; et al. A Genotype-Phenotype Study of High-Resolution FMR1 Nucleic Acid and Protein Analyses in Fragile X Patients with Neurobehavioral Assessments. Brain Sci. 2020 , 10 , 694. [ Google Scholar ] [ CrossRef ]
- Kim, K.; Hessl, D.; Randol, J.L.; Espinal, G.M.; Schneider, A.; Protic, D.; Aydin, E.Y.; Hagerman, R.J.; Hagerman, P.J. Association between IQ and FMR1 protein (FMRP) across the spectrum of CGG repeat expansions. PLoS ONE 2019 , 14 , e0226811. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Bagni, C.; Tassone, F.; Neri, G.; Hagerman, R. Fragile X syndrome: Causes, diagnosis, mechanisms, and therapeutics. J. Clin. Investig. 2012 , 122 , 4314–4322. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Yrigollen, C.M.; Martorell, L.; Durbin-Johnson, B.; Naudo, M.; Genoves, J.; Murgia, A.; Polli, R.; Zhou, L.; Barbouth, D.; Rupchock, A.; et al. AGG interruptions and maternal age affect FMR1 CGG repeat allele stability during transmission. J. Neurodev. Disord. 2014 , 6 , 24. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Yrigollen, C.M.; Durbin-Johnson, B.; Gane, L.; Nelson, D.L.; Hagerman, R.; Hagerman, P.J.; Tassone, F. AGG interruptions within the maternal FMR1 gene reduce the risk of offspring with fragile X syndrome. Genet. Med. 2012 , 14 , 729–736. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Hagerman, R.J.; Hagerman, P. Fragile X-associated tremor/ataxia syndrome-Features, mechanisms and management. Nat. Rev. Neurol. 2016 , 12 , 403–412. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Chen, L.; Hadd, A.; Sah, S.; Filipovic-Sadic, S.; Krosting, J.; Sekinger, E.; Pan, R.; Hagerman, P.J.; Stenzel, T.T.; Tassone, F.; et al. An information-rich CGG repeat primed PCR that detects the full range of fragile X expanded alleles and minimizes the need for southern blot analysis. J. Mol. Diagn. 2010 , 12 , 589–600. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Hagerman, R.J.; Protic, D.; Rajaratnam, A.; Salcedo-Arellano, M.J.; Aydin, E.Y.; Schneider, A. Fragile X-Associated Neuropsychiatric Disorders (FXAND). Front. Psychiatry 2018 , 9 , 564. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Dahlhaus, R. Of Men and Mice: Modeling the Fragile X Syndrome. Front. Mol. Neurosci. 2018 , 11 , 41. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Drozd, M.; Bardoni, B.; Capovilla, M. Modeling Fragile X Syndrome in Drosophila. Front. Mol. Neurosci. 2018 , 11 , 124. [ Google Scholar ] [ CrossRef ]
- Bostrom, C.A.; Majaess, N.-M.; Morch, K.; White, E.; Eadie, B.D.; Christie, B.R. Rescue of NMDAR-Dependent Synaptic Plasticity in Fmr1 Knock-Out Mice. Cereb. Cortex 2013 , 25 , 271–279. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Gantois, I.; Khoutorsky, A.; Popic, J.; Aguilar-Valles, A.; Freemantle, E.; Cao, R.; Sharma, V.; Pooters, T.; Nagpal, A.; Skalecka, A.; et al. Metformin ameliorates core deficits in a mouse model of fragile X syndrome. Nat. Med. 2017 , 23 , 674–677. [ Google Scholar ] [ CrossRef ]
- Niere, F.; Raab-Graham, K.F. mTORC1 Is a Local, Postsynaptic Voltage Sensor Regulated by Positive and Negative Feedback Pathways. Front. Cell. Neurosci. 2017 , 11 , 152. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Ng, M.-C.; Yang, Y.-L.; Lu, K.-T. Behavioral and synaptic circuit features in a zebrafish model of fragile X syndrome. PLoS ONE 2013 , 8 , e51456. [ Google Scholar ] [ CrossRef ]
- Kim, L.; He, L.; Maaswinkel, H.; Zhu, L.; Sirotkin, H.; Weng, W. Anxiety, hyperactivity and stereotypy in a zebrafish model of fragile X syndrome and autism spectrum disorder. Prog. Neuro-Psychopharmacol. Biol. Psychiatry 2014 , 55 , 40–49. [ Google Scholar ] [ CrossRef ]
- Wu, Y.-J.; Hsu, M.-T.; Ng, M.-C.; Amstislavskaya, T.G.; Tikhonova, M.A.; Yang, Y.-L.; Lu, K.-T. Fragile X Mental Retardation-1 Knockout Zebrafish Shows Precocious Development in Social Behavior. Zebrafish 2017 , 14 , 438–443. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Hu, J.; Chen, L.; Yin, J.; Yin, H.; Huang, Y.; Tian, J. Hyperactivity, memory defects, and craniofacial abnormalities in zebrafish fmr1 mutant larvae. Behav. Genet. 2020 , 50 , 1–9. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Zhang, Y.Q.; Bailey, A.M.; Matthies, H.J.G.; Renden, R.B.; Smith, M.A.; Speese, S.D.; Rubin, G.M.; Broadie, K. Drosophila Fragile X-Related Gene Regulates the MAP1B Homolog Futsch to Control Synaptic Structure and Function. Cell 2001 , 107 , 591–603. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Bolduc, F.V.; Valente, D.; Nguyen, A.T.; Mitra, P.P.; Tully, T. An assay for social interaction in Drosophila fragile X mutants. Fly 2010 , 4 , 216–225. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Choi, C.H.; Schoenfeld, B.P.; Weisz, E.D.; Bell, A.J.; Chambers, D.B.; Hinchey, J.; Choi, R.J.; Hinchey, P.; Kollaros, M.; Gertner, M.J.; et al. PDE-4 inhibition rescues aberrant synaptic plasticity in Drosophila and mouse models of fragile X syndrome. J. Neurosci. 2015 , 35 , 396–408. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Monyak, R.E.; Emerson, D.; Schoenfeld, B.P.; Zheng, X.; Chambers, D.B.; Rosenfelt, C.; Langer, S.; Hinchey, P.; Choi, C.H.; McDonald, T.V.; et al. Insulin signaling misregulation underlies circadian and cognitive deficits in a Drosophila fragile X model. Mol. Psychiatry 2017 , 22 , 1140–1148. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Hagerman, R.J.; Berry-Kravis, E.; Hazlett, H.C.; Bailey, D.B., Jr.; Moine, H.; Kooy, R.F.; Tassone, F.; Gantois, I.; Sonenberg, N.; Mandel, J.L.; et al. Fragile X syndrome. Nat. Rev. Dis. Primers 2017 , 3 , 17065. [ Google Scholar ] [ CrossRef ]
- Irwin, S.A.; Patel, B.; Idupulapati, M.; Harris, J.B.; Crisostomo, R.A.; Larsen, B.P.; Kooy, F.; Willems, P.J.; Cras, P.; Kozlowski, P.B.; et al. Abnormal dendritic spine characteristics in the temporal and visual cortices of patients with fragile-X syndrome: A quantitative examination. Am. J. Med. Genet. 2001 , 98 , 161–167. [ Google Scholar ] [ CrossRef ]
- Rudelli, R.D.; Brown, W.T.; Wisniewski, K.; Jenkins, E.C.; Laure-Kamionowska, M.; Connell, F.; Wisniewski, H.M. Adult fragile X syndrome. Clinico-neuropathologic findings. Acta Neuropathol. 1985 , 67 , 289–295. [ Google Scholar ] [ CrossRef ]
- Wisniewski, K.E.; Segan, S.M.; Miezejeski, C.M.; Sersen, E.A.; Rudelli, R.D. The Fra(X) syndrome: Neurological, electrophysiological, and neuropathological abnormalities. Am. J. Med. Genet. 1991 , 38 , 476–480. [ Google Scholar ] [ CrossRef ]
- Bailey, D.B., Jr.; Raspa, M.; Bishop, E.; Holiday, D. No change in the age of diagnosis for fragile x syndrome: Findings from a national parent survey. Pediatrics 2009 , 124 , 527–533. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Bailey, D.B., Jr.; Raspa, M.; Olmsted, M.; Holiday, D.B. Co-occurring conditions associated with FMR1 gene variations: Findings from a national parent survey. Am. J. Med. Genet. A 2008 , 146A , 2060–2069. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Kaufmann, W.E.; Kidd, S.A.; Andrews, H.F.; Budimirovic, D.B.; Esler, A.; Haas-Givler, B.; Stackhouse, T.; Riley, C.; Peacock, G.; Sherman, S.L.; et al. Autism Spectrum Disorder in Fragile X Syndrome: Cooccurring Conditions and Current Treatment. Pediatrics 2017 , 139 (Suppl. 3), S194–S206. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Budimirovic, D.; Haas-Givler, B.; Blitz, R.; Esler, A.; Kaufmann, W.; Sudhalter, V.; Stackhouse, T.M.; Scharfenaker, S.K.; Berry-Kravis, E. Autism Spectrum Disorder in Fragile X Syndrome. 2014. Available online: https://fragilex.org/wp-content/uploads/2012/08/Autism-Spectrum-Disorder-in-Fragile-X-Syndrome-2014-Nov.pdf (accessed on 18 December 2021).
- Berry-Kravis, E.; Filipink, R.A.; Frye, R.E.; Golla, S.; Morris, S.M.; Andrews, H.; Choo, T.H.; Kaufmann, W.E.; Consortium, F. Seizures in Fragile X Syndrome: Associations and Longitudinal Analysis of a Large Clinic-Based Cohort. Front. Pediatr. 2021 , 9 , 736255. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Hagerman, R.J.; Hagerman, P.J. Fragile X Syndrome and Premutation Disorders ; Mac Keith Press: London, UK, 2020. [ Google Scholar ]
- Hinton, R.; Budimirovic, D.B.; Marschik, P.B.; Talisa, V.B.; Einspieler, C.; Gipson, T.; Johnston, M.V. Parental reports on early language and motor milestones in fragile X syndrome with and without autism spectrum disorders. Dev. Neurorehabil. 2013 , 16 , 58–66. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Budimirovic, D.B.; Cvjetkovic, S.; Bukumiric, Z.; Duy, P.Q.; Protic, D. Fragile X-Associated Disorders in Serbia: Baseline Quantitative and Qualitative Survey of Knowledge, Attitudes and Practices Among Medical Professionals. Front. Neurosci. 2018 , 12 , 652. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Protic, D.; Salcedo-Arellano, M.J.; Stojkovic, M.; Saldarriag, W.; Ávila Vidal, L.A.; Miller, R.M.; Tabatadze, N.; Peric, M.; Hagerman, R.; Budimirovic, D.B. Raising Knowledge and Awareness of Fragile X Syndrome in Serbia, Georgia, and Colombia: A Model for Other Developing Countries? Yale J. Biol. Med. 2021 , 94 , 559–571. [ Google Scholar ]
- Budimirovic, D.B.; Province, H.S. Increasing Interest in Child and Adolescent Psychiatry Through a Structured Tutorial Program. Acad. Psychiatry 2020 . [ Google Scholar ] [ CrossRef ]
- Budimirovic, D.B.; Kaufmann, W.E. What can we learn about autism from studying fragile X syndrome? Dev. Neurosci. 2011 , 33 , 379–394. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Budimirovic, D.B.; Protic, D.; Toma, A.E. Fragile X syndrome: Leading the way as the most common monogenic form of autism spectrum disorder and the most translated among neurodevelopmental disorders in clinical trials. J. Clin. Gen. Genom. 2017 , 1 , 1–2. [ Google Scholar ]
- Budimirovic, D.; Subramanian, M. Neurobiology of Autism and Intellectual Disability: Fragile X Syndrome. In Neurobiology of Disease ; Johnston, M., Adams, H., Fatemi, A., Eds.; Oxford University Press: New York, NY, USA, 2016; pp. 375–384. [ Google Scholar ]
- McDuffie, A.; Thurman, A.J.; Hagerman, R.J.; Abbeduto, L. Symptoms of Autism in Males with Fragile X Syndrome: A Comparison to Nonsyndromic ASD Using Current ADI-R Scores. J. Autism. Dev. Disord. 2015 , 45 , 1925–1937. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Greiss Hess, L.; Fitzpatrick, S.E.; Nguyen, D.V.; Chen, Y.; Gaul, K.N.; Schneider, A.; Lemons Chitwood, K.; Eldeeb, M.A.; Polussa, J.; Hessl, D.; et al. A Randomized, Double-Blind, Placebo-Controlled Trial of Low-Dose Sertraline in Young Children With Fragile X Syndrome. J. Dev. Behav. Pediatr. 2016 , 37 , 619–628. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Berry-Kravis, E.; Sumis, A.; Hervey, C.; Mathur, S. Clinic-based retrospective analysis of psychopharmacology for behavior in fragile x syndrome. Int. J. Pediatr. 2012 , 2012 , 843016. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Salcedo-Arellano, M.J.; Cabal-Herrera, A.M.; Punatar, R.H.; Clark, C.J.; Romney, C.A.; Hagerman, R.J. Overlapping Molecular Pathways Leading to Autism Spectrum Disorders, Fragile X Syndrome, and Targeted Treatments. Neurotherapeutics 2021 , 18 , 265–283. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Filipovic-Sadic, S.; Sah, S.; Chen, L.; Krosting, J.; Sekinger, E.; Zhang, W.; Hagerman, P.J.; Stenzel, T.T.; Hadd, A.G.; Latham, G.J.; et al. A novel FMR1 PCR method for the routine detection of low abundance expanded alleles and full mutations in fragile X syndrome. Clin. Chem. 2010 , 56 , 399–408. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Garber, K.B.; Visootsak, J.; Warren, S.T. Fragile X syndrome. Eur. J. Hum. Genet. 2008 , 16 , 666–672. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Monaghan, K.G.; Lyon, E.; Spector, E.B.; American College of Medical Genetics and Genomics. ACMG Standards and Guidelines for fragile X testing: A revision to the disease-specific supplements to the Standards and Guidelines for Clinical Genetics Laboratories of the American College of Medical Genetics and Genomics. Genet. Med. 2013 , 15 , 575–586. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Biancalana, V.; Glaeser, D.; McQuaid, S.; Steinbach, P. EMQN best practice guidelines for the molecular genetic testing and reporting of fragile X syndrome and other fragile X-associated disorders. Eur. J. Hum. Genet. 2015 , 23 , 417–425. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Volkmar, F.; Siegel, M.; Woodbury-Smith, M.; King, B.; McCracken, J.; State, M.; American Academy of Child and Adolescent Psychiatry (AACAP) Committee on Quality Issues (CQI). Practice parameter for the assessment and treatment of children and adolescents with autism spectrum disorder. J. Am. Acad. Child Adolesc. Psychiatry 2014 , 53 , 237–257. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Martin, G.E.; Ausderau, K.K.; Raspa, M.; Bishop, E.; Mallya, U.; Bailey, D.B., Jr. Therapy service use among individuals with fragile X syndrome: Findings from a US parent survey. J. Intellect. Disabil. Res. 2013 , 57 , 837–849. [ Google Scholar ] [ CrossRef ]
- Hoque, M.; Lane, J.K.; Kaliouby, R.E.; Goodwin, M.; Picard, R.W. Exploring Speech Therapy Games with Children on the Autism Spectrum. In Proceedings of the 10th Annual Conference of the International Speech Communication Association, INTERSPEECH, Providence, RI, USA, 6–10 September 2009; p. 02906. [ Google Scholar ]
- Adams, C.; Lockton, E.; Freed, J.; Gaile, J.; Earl, G.; McBean, K.; Nash, M.; Green, J.; Vail, A.; Law, J. The Social Communication Intervention Project: A randomized controlled trial of the effectiveness of speech and language therapy for school-age children who have pragmatic and social communication problems with or without autism spectrum disorder. Int. J. Lang. Commun. Disord. 2012 , 47 , 233–244. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Case-Smith, J.; Arbesman, M. Evidence-based review of interventions for autism used in or of relevance to occupational therapy. Am. J. Occup. Ther. 2008 , 62 , 416–429. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- McClannahan, L.E.; MacDuff, G.S.; Krantz, P.J. Behavior analysis and intervention for adults with autism. Behav. Modif. 2002 , 26 , 9–26. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Nicholas, D.B.; Attridge, M.; Zwaigenbaum, L.; Clarke, M. Vocational support approaches in autism spectrum disorder: A synthesis review of the literature. Autism 2015 , 19 , 235–245. [ Google Scholar ] [ CrossRef ]
- McDuffie, A.; Banasik, A.; Bullard, L.; Nelson, S.; Feigles, R.T.; Hagerman, R.; Abbeduto, L. Distance delivery of a spoken language intervention for school-aged and adolescent boys with fragile X syndrome. Dev. Neurorehabil. 2018 , 21 , 48–63. [ Google Scholar ] [ CrossRef ]
- McDuffie, A.; Machalicek, W.; Bullard, L.; Nelson, S.; Mello, M.; Tempero-Feigles, R.; Castignetti, N.; Abbeduto, L. A Spoken-Language Intervention for School-Aged Boys with Fragile X Syndrome. Am. J. Intellect. Dev. Disabil. 2016 , 121 , 236–265. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Nelson, S.; McDuffie, A.; Banasik, A.; Tempero Feigles, R.; Thurman, A.J.; Abbeduto, L. Inferential language use by school-aged boys with fragile X syndrome: Effects of a parent-implemented spoken language intervention. J. Commun. Disord. 2018 , 72 , 64–76. [ Google Scholar ] [ CrossRef ]
- Axelrod, S.; McElrath, K.K.; Wine, B. Applied Behavior Analysis: Autism and Beyond. Behav. Interv. 2012 , 27 , 1–15. [ Google Scholar ] [ CrossRef ]
- Baril, E.M.; Humphreys, B.P. An Evaluation of the Research Evidence on the Early Start Denver Model. J. Early Interv. 2017 , 39 , 321–338. [ Google Scholar ] [ CrossRef ]
- Vismara, L.A.; McCormick, C.E.B.; Shields, R.; Hessl, D. Extending the Parent-Delivered Early Start Denver Model to Young Children with Fragile X Syndrome. J. Autism Dev. Disord. 2019 , 49 , 1250–1266. [ Google Scholar ] [ CrossRef ]
- Lieb-Lundell, C.C. Three Faces of Fragile X. Phys. Ther. 2016 , 96 , 1782–1790. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Lee, M.; Won, J.; Lee, S.; Hong, Y.; Kim, J.H.; Hong, Y. Benefits of Physical Exercise for Individuals with Fragile X Syndrome in Humans. J. Lifestyle Med. 2015 , 5 , 35–38. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Gitimoghaddam, M.; Vanderloo, L.M.; Hung, R.; Ryce, A.; McKellin, W.; Miller, A.; Collet, J.P. Impacts of Participation in Community-Based Physical Activity Programs on Cognitive Functions of Children and Youth with Neurodevelopmental Disabilities: A Scoping Review. Brain Sci. 2021 , 11 , 195. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Gomez-Pinilla, F.; Ying, Z.; Roy, R.R.; Molteni, R.; Edgerton, V.R. Voluntary exercise induces a BDNF-mediated mechanism that promotes neuroplasticity. J. Neurophysiol. 2002 , 88 , 2187–2195. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Castren, M.L.; Castren, E. BDNF in fragile X syndrome. Neuropharmacology 2014 , 76 , 729–736. [ Google Scholar ] [ CrossRef ]
- Powers, M.B.; de Kleine, R.A.; Smits, J.A.J. Core Mechanisms of Cognitive Behavioral Therapy for Anxiety and Depression: A Review. Psychiatr. Clin. North Am. 2017 , 40 , 611–623. [ Google Scholar ] [ CrossRef ]
- Weiss, J.A.; Thomson, K.; Burnham Riosa, P.; Albaum, C.; Chan, V.; Maughan, A.; Tablon, P.; Black, K. A randomized waitlist-controlled trial of cognitive behavior therapy to improve emotion regulation in children with autism. J. Child Psychol. Psychiatry 2018 , 59 , 1180–1191. [ Google Scholar ] [ CrossRef ]
- Haessler, F.; Gaese, F.; Huss, M.; Kretschmar, C.; Brinkman, M.; Peters, H.; Elstner, S.; Colla, M.; Pittrow, D. Characterization, treatment patterns, and patient-related outcomes of patients with Fragile X syndrome in Germany: Final results of the observational EXPLAIN-FXS study. BMC Psychiatry 2016 , 16 , 318. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Geretsegger, M.; Elefant, C.; Mossler, K.A.; Gold, C. Music therapy for people with autism spectrum disorder. Cochrane Database Syst. Rev. 2014 , 6 , CD004381. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Hagerman, R.J.; Protic, D.; Berry-Kravis, E. Medical, Psychopharmacological, and Targeted Treatment for FXS. In Fragile X Syndrome and Premutation Disorders ; Hagerman, R.J., Hagerman, P.J., Eds.; Mac Keith Press: London, UK, 2020; pp. 41–59. [ Google Scholar ]
- Salcedo-Arellano, M.J.; Aishworiya, R.; Hagerman, R.J.; Protic, D. Pharmacotherapy of Fragile X Syndrome. In Fragile X Syndrome: From Diagnosis to Treatment ; Stasolla, F., Ed.; Nova Science Publishers: New York, NY, USA, 2022; pp. 61–91. [ Google Scholar ]
- Austerman, J. ADHD and behavioral disorders: Assessment, management, and an update from DSM-5. Clevel. Clin. J. Med. 2015 , 82 (Suppl. 1), S2–S7. [ Google Scholar ] [ CrossRef ]
- Brown, K.A.; Samuel, S.; Patel, D.R. Pharmacologic management of attention deficit hyperactivity disorder in children and adolescents: A review for practitioners. Transl. Pediatr. 2018 , 7 , 36–47. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Arnsten, A.F.T.; Berridge, C.W. Catecholamine Influences on Prefrontal Cortex Circuits and Function. In Attention-Deficit Hyperactivity Disorder in Adults and Children ; Arnsten, A.F.T., Berridge, C.W., Adler, L.A., Spencer, T.J., Wilens, T.E., Eds.; Cambridge University Press: Cambridge, UK, 2015; pp. 161–173. [ Google Scholar ]
- Hagerman, R.J.; Berry-Kravis, E.; Kaufmann, W.E.; Ono, M.Y.; Tartaglia, N.; Lachiewicz, A.; Kronk, R.; Delahunty, C.; Hessl, D.; Visootsak, J.; et al. Advances in the treatment of fragile X syndrome. Pediatrics 2009 , 123 , 378–390. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Hagerman, R.J.; Polussa, J. Treatment of the psychiatric problems associated with fragile X syndrome. Curr. Opin. Psychiatry 2015 , 28 , 107–112. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Markowitz, J.S.; Straughn, A.B.; Patrick, K.S. Advances in the pharmacotherapy of attention-deficit-hyperactivity disorder: Focus on methylphenidate formulations. Pharmacotherapy 2003 , 23 , 1281–1299. [ Google Scholar ] [ CrossRef ]
- Cooper, W.O.; Habel, L.A.; Sox, C.M.; Chan, K.A.; Arbogast, P.G.; Cheetham, T.C.; Murray, K.T.; Quinn, V.P.; Stein, C.M.; Callahan, S.T.; et al. ADHD drugs and serious cardiovascular events in children and young adults. N. Engl. J. Med. 2011 , 365 , 1896–1904. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Olfson, M.; Huang, C.; Gerhard, T.; Winterstein, A.G.; Crystal, S.; Allison, P.D.; Marcus, S.C. Stimulants and cardiovascular events in youth with attention-deficit/hyperactivity disorder. J. Am. Acad. Child Adolesc. Psychiatry 2012 , 51 , 147–156. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Dalsgaard, S.; Kvist, A.P.; Leckman, J.F.; Nielsen, H.S.; Simonsen, M. Cardiovascular safety of stimulants in children with attention-deficit/hyperactivity disorder: A nationwide prospective cohort study. J. Child Adolesc. Psychopharmacol. 2014 , 24 , 302–310. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Berry-Kravis, E.; Potanos, K. Psychopharmacology in fragile X syndrome--Present and future. Ment. Retard. Dev. Disabil. Res. Rev. 2004 , 10 , 42–48. [ Google Scholar ] [ CrossRef ]
- Cinnamon Bidwell, L.; Dew, R.E.; Kollins, S.H. Alpha-2 adrenergic receptors and attention-deficit/hyperactivity disorder. Curr. Psychiatry Rep. 2010 , 12 , 366–373. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Ballinger, E.C.; Cordeiro, L.; Chavez, A.D.; Hagerman, R.J.; Hessl, D. Emotion potentiated startle in fragile X syndrome. J. Autism Dev. Disord. 2014 , 44 , 2536–2546. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Cipriani, A.; La Ferla, T.; Furukawa, T.A.; Signoretti, A.; Nakagawa, A.; Churchill, R.; McGuire, H.; Barbui, C. Sertraline versus other antidepressive agents for depression. Cochrane Database Syst. Rev. 2010 , 4 , Cd006117. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Hanson, A.C.; Hagerman, R.J. Serotonin dysregulation in Fragile X Syndrome: Implications for treatment. Intractable Rare Dis. Res. 2014 , 3 , 110–117. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Winarni, T.I.; Schneider, A.; Borodyanskara, M.; Hagerman, R.J. Early intervention combined with targeted treatment promotes cognitive and behavioral improvements in young children with fragile x syndrome. Case Rep. Genet. 2012 , 2012 , 280813. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Fava, M.; Rush, A.J.; Thase, M.E.; Clayton, A.; Stahl, S.M.; Pradko, J.F.; Johnston, J.A. 15 years of clinical experience with bupropion HCl: From bupropion to bupropion SR to bupropion XL. Prim Care Companion J. Clin. Psychiatry 2005 , 7 , 106–113. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Jain, A.K.; Kaplan, R.A.; Gadde, K.M.; Wadden, T.A.; Allison, D.B.; Brewer, E.R.; Leadbetter, R.A.; Richard, N.; Haight, B.; Jamerson, B.D.; et al. Bupropion SR vs. placebo for weight loss in obese patients with depressive symptoms. Obes. Res. 2002 , 10 , 1049–1056. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Anderson, J.W.; Greenway, F.L.; Fujioka, K.; Gadde, K.M.; McKenney, J.; O’Neil, P.M. Bupropion SR enhances weight loss: A 48-week double-blind, placebo- controlled trial. Obes. Res. 2002 , 10 , 633–641. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Tsiouris, J.A.; Brown, W.T. Neuropsychiatric symptoms of fragile X syndrome: Pathophysiology and pharmacotherapy. CNS Drugs 2004 , 18 , 687–703. [ Google Scholar ] [ CrossRef ]
- Erickson, C.A.; Stigler, K.A.; Wink, L.K.; Mullett, J.E.; Kohn, A.; Posey, D.J.; McDougle, C.J. A prospective open-label study of aripiprazole in fragile X syndrome. Psychopharmacology 2011 , 216 , 85–90. [ Google Scholar ] [ CrossRef ]
- McLennan, Y.; Polussa, J.; Tassone, F.; Hagerman, R. Fragile x syndrome. Curr. Genom. 2011 , 12 , 216–224. [ Google Scholar ] [ CrossRef ]
- Choo, T.H.; Xu, Q.; Budimirovic, D.; Lozano, R.; Esler, A.; Frye, R.E.; Andrews, H.; Martin, V. Height and Body Mass Index in Fragile X Syndrome: A Longitudinal Assessment. Obesity 2022 , in press. [ Google Scholar ]
- Kucka, M.; Tomic, M.; Bjelobaba, I.; Stojilkovic, S.S.; Budimirovic, D.B. Paliperidone and aripiprazole differentially affect the strength of calcium-secretion coupling in female pituitary lactotrophs. Sci. Rep. 2015 , 5 , 8902. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Kronk, R.; Dahl, R.; Noll, R. Caregiver reports of sleep problems on a convenience sample of children with fragile X syndrome. Am. J. Intellect. Dev. Disabil. 2009 , 114 , 383–392. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Budimirovic, D.B.; Protic, D.D.; Delahunty, C.M.; Andrews, H.F.; Choo, T.H.; Xu, Q.; Berry-Kravis, E.; Kaufmann, W.E.; Consortium, F. Sleep problems in fragile X syndrome: Cross-sectional analysis of a large clinic-based cohort. Am. J. Med. Genet. A 2021 . [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Wirojanan, J.; Jacquemont, S.; Diaz, R.; Bacalman, S.; Anders, T.F.; Hagerman, R.J.; Goodlin-Jones, B.L. The efficacy of melatonin for sleep problems in children with autism, fragile X syndrome, or autism and fragile X syndrome. J. Clin. Sleep Med. 2009 , 5 , 145–150. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Reiter, R.J.; Calvo, J.R.; Karbownik, M.; Qi, W.; Tan, D.X. Melatonin and its relation to the immune system and inflammation. Ann. N. Y. Acad. Sci. 2000 , 917 , 376–386. [ Google Scholar ] [ CrossRef ]
- Srinivasan, V.; Pandi-Perumal, S.R.; Trahkt, I.; Spence, D.W.; Poeggeler, B.; Hardeland, R.; Cardinali, D.P. Melatonin and melatonergic drugs on sleep: Possible mechanisms of action. Int. J. Neurosci. 2009 , 119 , 821–846. [ Google Scholar ] [ CrossRef ]
- El-Sherif, Y.; Tesoriero, J.; Hogan, M.V.; Wieraszko, A. Melatonin regulates neuronal plasticity in the hippocampus. J. Neurosci. Res. 2003 , 72 , 454–460. [ Google Scholar ] [ CrossRef ]
- Berry-Kravis, E.M.; Lindemann, L.; Jonch, A.E.; Apostol, G.; Bear, M.F.; Carpenter, R.L.; Crawley, J.N.; Curie, A.; Des Portes, V.; Hossain, F.; et al. Drug development for neurodevelopmental disorders: Lessons learned from fragile X syndrome. Nat. Rev. Drug Discov. 2018 , 17 , 280–299. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Berry-Kravis, E.; Hagerman, R.; Visootsak, J.; Budimirovic, D.; Kaufmann, W.E.; Cherubini, M.; Zarevics, P.; Walton-Bowen, K.; Wang, P.; Bear, M.F.; et al. Arbaclofen in fragile X syndrome: Results of phase 3 trials. J. Neurodev. Disord. 2017 , 9 . [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Budimirovic, D.B.; Berry-Kravis, E.; Erickson, C.A.; Hall, S.S.; Hessl, D.; Reiss, A.L.; King, M.K.; Abbeduto, L.; Kaufmann, W.E. Updated report on tools to measure outcomes of clinical trials in fragile X syndrome. J. Neurodev. Disord. 2017 , 9 , 14. [ Google Scholar ] [ CrossRef ]
- Olmos-Serrano, J.L.; Corbin, J.G.; Burns, M.P. The GABA(A) receptor agonist THIP ameliorates specific behavioral deficits in the mouse model of fragile X syndrome. Dev. Neurosci. 2011 , 33 , 395–403. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Olmos-Serrano, J.L.; Corbin, J.G. Amygdala Regulation of Fear and Emotionality in Fragile X Syndrome. Dev. Neurosci. 2011 , 33 , 365–378. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Budimirovic, D.B.; Dominick, K.C.; Gabis, L.V.; Adams, M.; Adera, M.; Huang, L.; Ventola, P.; Tartaglia, N.R.; Berry-Kravis, E. Gaboxadol in Fragile X Syndrome: A 12-Week Randomized, Double-Blind, Parallel-Group, Phase 2a Study. Front. Pharmacol. 2021 , 12 , 757825. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Berry-Kravis, E.; Des Portes, V.; Hagerman, R.; Jacquemont, S.; Charles, P.; Visootsak, J.; Brinkman, M.; Rerat, K.; Koumaras, B.; Zhu, L.; et al. Mavoglurant in fragile X syndrome: Results of two randomized, double-blind, placebo-controlled trials. Sci. Transl. Med. 2016 , 8 , 321ra5. [ Google Scholar ] [ CrossRef ]
- Youssef, E.A.; Berry-Kravis, E.; Czech, C.; Hagerman, R.J.; Hessl, D.; Wong, C.Y.; Rabbia, M.; Deptula, D.; John, A.; Kinch, R.; et al. Effect of the mGluR5-NAM Basimglurant on Behavior in Adolescents and Adults with Fragile X Syndrome in a Randomized, Double-Blind, Placebo-Controlled Trial: FragXis Phase 2 Results. Neuropsychopharmacology 2018 , 43 , 503–512. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Duy, P.Q.; Budimirovic, D.B. Fragile X Syndrome: Lessons Learned from the Most Translated Neurodevelopmental Disorder in Clinical Trials. Transl. Neurosci. 2017 , 8 , 7–8. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Luu, S.; Province, H.; Berry-Kravis, E.; Hagerman, R.; Hessl, D.; Vaidya, D.; Lozano, R.; Rosselot, H.; Erickson, C.; Kaufmann, W.E.; et al. Response to Placebo in Fragile X Syndrome Clinical Trials: An Initial Analysis. Brain Sci. 2020 , 10 , 629. [ Google Scholar ] [ CrossRef ]
- Brašić, J.R.; Nandi, A.; Russell, D.S.; Jennings, D.; Barret, O.; Mathur, A.; Slifer, K.; Sedlak, T.; Martin, S.D.; Brinson, Z.; et al. Reduced Expression of Cerebral Metabotropic Glutamate Receptor Subtype 5 in Men with Fragile X Syndrome. Brain Sci. 2020 , 10 , 899. [ Google Scholar ] [ CrossRef ]
- Brašić, J.R.; Nandi, A.; Russell, D.S.; Jennings, D.; Barret, O.; Martin, S.D.; Slifer, K.; Sedlak, T.; Seibyl, J.P.; Wong, D.F.; et al. Cerebral Expression of Metabotropic Glutamate Receptor Subtype 5 in Idiopathic Autism Spectrum Disorder and Fragile X Syndrome: A Pilot Study. Int. J. Mol. Sci. 2021 , 22 , 899. [ Google Scholar ] [ CrossRef ]
- Mody, M.; Petibon, Y.; Han, P.; Kuruppu, D.; Ma, C.; Yokell, D.; Neelamegam, R.; Normandin, M.D.; Fakhri, G.E.; Brownell, A.-L. In vivo imaging of mGlu5 receptor expression in humans with Fragile X Syndrome towards development of a potential biomarker. Sci. Rep. 2021 , 11 , 15897. [ Google Scholar ] [ CrossRef ]
- Lee, A.W.; Ventola, P.; Budimirovic, D.; Berry-Kravis, E.; Visootsak, J. Clinical Development of Targeted Fragile X Syndrome Treatments: An Industry Perspective. Brain Sci. 2018 , 8 , 214. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Berry-Kravis, E.M.; Harnett, M.D.; Reines, S.A.; Reese, M.A.; Ethridge, L.E.; Outterson, A.H.; Michalak, C.; Furman, J.; Gurney, M.E. Inhibition of phosphodiesterase-4D in adults with fragile X syndrome: A randomized, placebo-controlled, phase 2 clinical trial. Nat. Med. 2021 , 27 , 862–870. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Tartaglia, N.; Bonn-Miller, M.; Hagerman, R. Treatment of Fragile X Syndrome with Cannabidiol: A Case Series Study and Brief Review of the Literature. Cannabis Cannabinoid Res. 2019 , 4 , 3–9. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Berry-Kravis, E.; Hagerman, R.; Budimirovic, D.; Erickson, C.; Heussler, H.; Tartaglia, N.; Cohen, J.; Dobbins, T.; Merikle, E.; Sebree, T.; et al. A Randomized, Controlled, Trial of ZYN002 Cannabidiol Transdermal Gel in Children and Adolescents with Fragile X Syndrome (CONNECT-FX). Sci. Transl. Med. 2022 . under review. [ Google Scholar ]
- Heussler, H.; Cohen, J.; Silove, N.; Tich, N.; Bonn-Miller, M.O.; Du, W.; O’Neill, C.; Sebree, T. A phase 1/2, open-label assessment of the safety, tolerability, and efficacy of transdermal cannabidiol (ZYN002) for the treatment of pediatric fragile X syndrome. J. Neurodev. Disord. 2019 , 11 , 16. [ Google Scholar ] [ CrossRef ]
- Palumbo, J.M.; Thomas, B.F.; Budimirovic, D.B.; Siegel, S.; Tassone, F.; Hagerman, R.; Faulk, C.; O’Quinn, S.; Sebree, T. Role of the Endocannabinoid System in Fragile X Syndrome: Potential Mechanisms for Benefit From Cannabidiol Treatment. Sci. Transl. Med. 2022 . under review. [ Google Scholar ]
- Romero, R.; Erez, O.; Huttemann, M.; Maymon, E.; Panaitescu, B.; Conde-Agudelo, A.; Pacora, P.; Yoon, B.H.; Grossman, L.I. Metformin, the aspirin of the 21st century: Its role in gestational diabetes mellitus, prevention of preeclampsia and cancer, and the promotion of longevity. Am. J. Obstet. Gynecol. 2017 , 217 , 282–302. [ Google Scholar ] [ CrossRef ]
- Viollet, B.; Guigas, B.; Sanz Garcia, N.; Leclerc, J.; Foretz, M.; Andreelli, F. Cellular and molecular mechanisms of metformin: An overview. Clin. Sci. 2012 , 122 , 253–270. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Salcedo-Arellano, M.J.; Dufour, B.; McLennan, Y.; Martinez-Cerdeno, V.; Hagerman, R. Fragile X syndrome and associated disorders: Clinical aspects and pathology. Neurobiol. Dis. 2020 , 136 , 104740. [ Google Scholar ] [ CrossRef ]
- Wang, Y.; Zhao, J.; Guo, F.L.; Gao, X.; Xie, X.; Liu, S.; Yang, X.; Yang, X.; Zhang, L.; Ye, Y.; et al. Metformin Ameliorates Synaptic Defects in a Mouse Model of AD by Inhibiting Cdk5 Activity. Front Cell Neurosci. 2020 , 14 , 170. [ Google Scholar ] [ CrossRef ]
- Gantois, I.; Popic, J.; Khoutorsky, A.; Sonenberg, N. Metformin for Treatment of Fragile X Syndrome and Other Neurological Disorders. Annu. Rev. Med. 2019 , 70 , 167–181. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Dy, A.B.C.; Tassone, F.; Eldeeb, M.; Salcedo-Arellano, M.J.; Tartaglia, N.; Hagerman, R. Metformin as targeted treatment in fragile X syndrome. Clin. Genet. 2018 , 93 , 216–222. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Biag, H.M.B.; Potter, L.A.; Wilkins, V.; Afzal, S.; Rosvall, A.; Salcedo-Arellano, M.J.; Rajaratnam, A.; Manzano-Nunez, R.; Schneider, A.; Tassone, F.; et al. Metformin treatment in young children with fragile X syndrome. Mol. Genet. Genomic Med. 2019 , 7 , e956. [ Google Scholar ] [ CrossRef ]
- Protic, D.; Aydin, E.Y.; Tassone, F.; Tan, M.M.; Hagerman, R.J.; Schneider, A. Cognitive and behavioral improvement in adults with fragile X syndrome treated with metformin-two cases. Mol. Genet. Genomic Med. 2019 , 7 , e00745. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
- Protic, D.; Kaluzhny, P.; Tassone, F.; Hagerman, R. Prepubertal Metformin Treatment in Fragile X Syndrome Alleviated Macroorchidism: A Case Study. Adv. Clin. Transl. Res. 2019 , 3 , 1–5. [ Google Scholar ]
- Jasoliya, M.; Bowling, H.; Petrasic, I.C.; Durbin-Johnson, B.; Klann, E.; Bhattacharya, A.; Hagerman, R.; Tassone, F. Blood-Based Biomarkers Predictive of Metformin Target Engagement in Fragile X Syndrome. Brain Sci. 2020 , 10 , 361. [ Google Scholar ] [ CrossRef ]
- Proteau-Lemieux, M.; Lacroix, A.; Galarneau, L.; Corbin, F.; Lepage, J.F.; Caku, A. The safety and efficacy of metformin in fragile X syndrome: An open-label study. Prog. Neuropsychopharmacol. Biol. Psychiatry 2021 , 110 , 110307. [ Google Scholar ] [ CrossRef ]
- Kim, H.S.; Suh, Y.H. Minocycline and neurodegenerative diseases. Behav. Brain Res. 2009 , 196 , 168–179. [ Google Scholar ] [ CrossRef ]
- Griffin, M.O.; Fricovsky, E.; Ceballos, G.; Villarreal, F. Tetracyclines: A pleitropic family of compounds with promising therapeutic properties. Review of the literature. Am. J. Physiol. Cell Physiol. 2010 , 299 , C539–C548. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Siller, S.S.; Broadie, K. Neural circuit architecture defects in a Drosophila model of Fragile X syndrome are alleviated by minocycline treatment and genetic removal of matrix metalloproteinase. Dis. Model Mech. 2011 , 4 , 673–685. [ Google Scholar ] [ CrossRef ] [ Green Version ]
- Toledo, M.A.; Wen, T.H.; Binder, D.K.; Ethell, I.M.; Razak, K.A. Reversal of ultrasonic vocalization deficits in a mouse model of Fragile X Syndrome with minocycline treatment or genetic reduction of MMP-9. Behav. Brain Res. 2019 , 372 , 112068. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Leigh, M.J.; Nguyen, D.V.; Mu, Y.; Winarni, T.I.; Schneider, A.; Chechi, T.; Polussa, J.; Doucet, P.; Tassone, F.; Rivera, S.M.; et al. A randomized double-blind, placebo-controlled trial of minocycline in children and adolescents with fragile x syndrome. J. Dev. Behav. Pediatr. 2013 , 34 , 147–155. [ Google Scholar ] [ CrossRef ] [ PubMed ]
- Schneider, A.; Leigh, M.J.; Adams, P.; Nanakul, R.; Chechi, T.; Olichney, J.; Hagerman, R.; Hessl, D. Electrocortical changes associated with minocycline treatment in fragile X syndrome. J. Psychopharmacol. 2013 , 27 , 956–963. [ Google Scholar ] [ CrossRef ] [ PubMed ] [ Green Version ]
Click here to enlarge figure
| Medication | Drug Class | Mechanism of Action | Target Problems | Dose/Day | Common Side Effects |
|---|---|---|---|---|---|
| Sertraline | Selective serotonin reuptake inhibitor (SSRI) | Inhibition of presynaptic neuronal uptake of serotonin (5HT) | Anxiety, aggressive behaviors, social participation, language development in young children | 2.5 to 5.0 mg in young children (2 to 6 years) 10 to 100 mg in older children and adolescents, up to 200 mg per day in adults | Diarrhea, loss of appetite, hyperhidrosis, activation (restlessness, mood changes, disinhibited behaviors), tremor |
| Methylphenidate | Central nervous system (CNS) stimulant | Non-competitively blocks the reuptake of dopamine and noradrenaline | Attention deficit and hyperactivity disorder (ADHD) | 10 to 60 mg per day typically depending on the preparation and dosing frequency | Decreased appetite, nausea, headaches, hypertension, irritability, insomnia |
| Clonidine | Alpha 2-adrenergic receptor agonist | Stimulates presynaptic and postsynaptic alpha 2 adrenergic receptors in the prefrontal cortex | Hyperactivity, overstimulation, attention/concentration problems and aggression, sleep disturbances | Initial dose 0.025 mg/day in children and titrate to maximum dose of 0.4 mg/day in older children and adults | Sedation, postural hypotension, nausea, constipation, bradycardia, xerostomia |
| Guanfacine | Alpha 2-adrenergic receptor agonist | Stimulates presynaptic and postsynaptic alpha 2 adrenergic receptors in the prefrontal cortex | Hyperactivity, frustration intolerance, hyperarousal | Initial dose of 0.5 mg/day and titrate up to a maximum dose of 4 mg/day in adults | Less sedation than clonidine, nausea, constipation, bradycardia, xerostomia |
| Risperidone | Second-generation antipsychotic | Blocks dopamine D2 receptors in the prefrontal cortex and nucleus accumbens. Serotonin and norepinephrine reuptake inhibition | Irritability, aggression, self-injury, social impairment, stereotypic behaviors, psychosis and hyperactivity | 0.5 to 3 mg/day based on weight. Available in long-acting injection | Weight changes, metabolic changes, and sedation. Extrapyramidal symptoms, parkinsonian features, hyperprolactinemia |
| Aripiprazole | Second-generation antipsychotic | Blocks dopamine D2, D3 and 5-HT1A (serotonin) receptors. Antagonist at the 5-HT2A receptor | Irritability, aggression, agitation, and self-injurious behaviors, sleep problems, severe anxiety | 2 to 15 mg/day based on weight and age | Increase in weight, somnolence (dose–response relationship) and extrapyramidal symptoms |
| Melatonin | Biogenic amine/endogenous hormone | Activates melatonin receptors ML1/ML2, leading to inhibition of adenylate cyclase and the cAMP signal transduction pathway; Activation of phospholipase C | Sleep disturbances | Initial low dose of 1 mg in young children and titrating up as needed; maximum dose 10 mg typically | Day-time drowsiness, headache, dizziness, nausea |
| Medication | Drug Class | Mechanism of Action | Target Problems | Dose/Day | Common Side Effects |
|---|---|---|---|---|---|
| Cannabidiol (CBD) | Phytocannabinoid exhibits no effects indicative of any abuse or dependence potential | Multiple: Interact with an FXS-compromised endocannabinoid system; positively affect synaptic plasticity; a positive allosteric modulator of GABAA receptors; Serotonin 1A receptors modulator | Multiple: Reductions in social avoidance and anxiety; improvements in sleep, feeding, motor coordination, language skills, anxiety, seizures and sensory processing | Oral tincture: 25 to 50 mg per dose up to bid Topical ointments: 125 mg to 500 mg bid | Sedation, but rare cases of activation. Higher doses may cause liver function test elevations. Topicals may cause skin rash or irritation |
| BPN14770 | Phosphodiesterase-4D (PDE4D) inhibitor | Selectively binding to and partially suppressing the activity of phosphodiesterase 4D (PDE4D), an enzyme known to regulate a brain cyclic adenosine monophosphate (cAMP). | Cognitive function, language and daily functioning | Adult: 25 mg bid | The most commonly reported side effects were vomiting and upper respiratory infections |
| Metformin | Oral antidiabetic; Class: biguanides | Impacts mTOR and ERK/MAPK pathways through the AMP- activated protein kinase pathway. Decreases the level of protein MMP-9 in FXS | Intellectual abilities, behavioral problems, speech and language deficits, weight management | 500 mg bid < 50 kg 1000 mg bid > 50 kg | Gastrointestinal side effects (nausea, diarrhea and abdominal discomfort), headache, weight loss, dizziness, inadequate vitamin B12 |
| Minocycline | Antibiotic; Class: tetracycline | Anti-inflammatory, MMP inhibition and anti-apoptotic effects. Inhibition of activity of the protein MMP-9 in FXS | Overall clinical improvement; In particular, mood and anxiety-related behaviors | 25 mg < 25 kg 50 mg 25–45 kg 100 mg > 45 mg | Gastrointestinal side effects (nausea, diarrhea, etc.), dizziness, discoloration of skin, nails or gums |
| MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
Share and Cite
Protic, D.D.; Aishworiya, R.; Salcedo-Arellano, M.J.; Tang, S.J.; Milisavljevic, J.; Mitrovic, F.; Hagerman, R.J.; Budimirovic, D.B. Fragile X Syndrome: From Molecular Aspect to Clinical Treatment. Int. J. Mol. Sci. 2022 , 23 , 1935. https://doi.org/10.3390/ijms23041935
Protic DD, Aishworiya R, Salcedo-Arellano MJ, Tang SJ, Milisavljevic J, Mitrovic F, Hagerman RJ, Budimirovic DB. Fragile X Syndrome: From Molecular Aspect to Clinical Treatment. International Journal of Molecular Sciences . 2022; 23(4):1935. https://doi.org/10.3390/ijms23041935
Protic, Dragana D., Ramkumar Aishworiya, Maria Jimena Salcedo-Arellano, Si Jie Tang, Jelena Milisavljevic, Filip Mitrovic, Randi J. Hagerman, and Dejan B. Budimirovic. 2022. "Fragile X Syndrome: From Molecular Aspect to Clinical Treatment" International Journal of Molecular Sciences 23, no. 4: 1935. https://doi.org/10.3390/ijms23041935
Article Metrics
Article access statistics, further information, mdpi initiatives, follow mdpi.

Subscribe to receive issue release notifications and newsletters from MDPI journals
Fragile X syndrome: clinical presentation, pathology and treatment.
- Salcedo-Arellano, María Jimena ;
- Hagerman, Randi J ;
- Martínez-Cerdeño, Verónica
Published Web Location
Fragile X syndrome is the monogenetic condition that produces more cases of autism and intellectual disability. The repetition of CGG triplets (> 200) and their methylation entail the silencing of the FMR1 gene. The FMRP protein (product of the FMR1 gene) interacts with ribosomes by controlling the translation of specific messengers, and its loss causes alterations in synaptic connectivity. Screening for fragile X syndrome is performed by polymerase chain reaction. Current recommendation of the American Academy of Pediatrics is to test individuals with intellectual disability, global developmental retardation or with a family history of presence of the mutation or premutation. Hispanic countries such as Colombia, Chile and Spain report high prevalence of fragile X syndrome and have created fragile X national associations or corporations that seek to bring patients closer to available diagnostic and treatment networks.
Many UC-authored scholarly publications are freely available on this site because of the UC's open access policies . Let us know how this access is important for you.

- Donate --> DONATE

- Fragile X 101
- Signs & Symptoms
- Genetics and Inheritance
- Testing and Diagnosis
- Fragile X Syndrome
- Fragile X & Autism
- Premutation
- New Developments
- Xtraordinary Individuals
- 31 Shareable Fragile X Facts
- Fragile X Info Series
- FRAGILE X MASTERCLASS
- Knowledge Center
- Resources for Families
- Daily Living
- Medications
- Physical & Medical Concerns
- Fragile X and Puberty & Sexuality
- School & Education
- FXS Resources by Age
- The Fragile X Premutation
- FXTAS Resources
- FXPOI Resources
- Reproductive Resources
- Newly Diagnosed
- Research 101: What is Research?
- STX209 Reconsent Project
- International Fragile X Premutation Registry
- Participate in Research
- Original Research Articles
- FORWARD-MARCH
- Research Results Roundup
- Find a Clinic Near You
- Find a Contact Near Your
- Webinars & Videos
- Printable Resources
- Treatment Recommendations
- Resources for Professionals
- NFXF MasterClass™️ for Professionals
- Research Readiness Program
- NFXF Data Repository
- FORWARD-MARCH Registry & Database
- NFXF-Led Patient-Focused Drug Development Meeting
- Marketing Your Research Opportunities
- Randi J. Hagerman Summer Scholars
- Junior Investigator Awards
- Give Monthly
- Other Ways to Give
- Meet Our Donors
- NFXF Merchandise
- Fragile X Awareness
- Fragile X Conference
- Mission & Vision
- Financials & Impact
- Our Advisors
- Our Sponsors
Fragile X Syndrome & Associated Premutation Conditions Webinars
Fragile x syndrome & associated premutation conditions webinars, all webinars, conference sessions.
Here we present all webinars, including our conference panels and presentations, organized from newest to oldest. Please note that these are also all available on our various Fragile X syndrome and premutation topic pages .

Lunch & Learn Series: FMR1 Carriers Report Executive Function Changes Prior to FXTAS: A Longitudinal Study
Anna De Sonia 2024-07-18T07:57:12-04:00 Jul 18, 2024 |
David Hessl, PhD, joined us for a 45-minute webinar where he presented the results of a longitudinal study focusing on executive function changes in people with the FMR1 premutation. The presentation was followed by a moderated Q&A.

Financially Empowering the Fragile X Community Through ABLE Accounts — Webinar
Missy Zolecki 2024-05-21T17:00:11-04:00 May 21, 2024 |
Join us for the Financially Empowering the Fragile X Community Through ABLE Accounts webinar with Mark Raymond Jr. & John Finch.

10 Basic Financial Steps for Special Needs Caregivers — Webinar
Missy Zolecki 2024-05-10T11:49:46-04:00 May 10, 2024 |
Kelly Piacenti joined us for the next topic of the NFXF Webinar Series -10 Basic Financial Steps for Special Needs Caregivers. She shared information to help caregivers get started in preparing for the financial future of their loved one with Fragile X.

Peer-Reviewed Medical Research Program’s FY 24 Funding Opportunities for Researchers — Webinar
Missy Zolecki 2024-04-18T12:33:31-04:00 Apr 18, 2024 |
Dr. Kathryn Argue share tips for applying for PRMRP funding with Fragile X professionals.
I Have the Fragile X Premutation…Now What?— Webinar
Missy Zolecki 2024-03-13T17:15:18-04:00 Mar 13, 2024 |
Drs. Deborah Barbouth, Emily Allen, Reymundo Lozano, and David Hessl joined us for a one-hour Q & A discussing the Fragile X Premutation.

Lunch & Learn Series: Single-Dose Medication Study Results in Fragile X Syndrome
Anna De Sonia 2024-06-11T06:57:53-04:00 Feb 21, 2024 |
Dr. Craig Erickson joined us for a 45-minute webinar where he presented the results of a single-dose medication study in FXS and then answered questions during a moderated Q&A.
2024 NIH Fragile X Centers of Excellence — Webinar
Hilary Rosselot 2024-02-12T17:05:44-05:00 Feb 12, 2024 |
Hear updates about the NIH-funded Fragile X Centers of Excellence, which support research to improve the diagnosis and treatment of Fragile X syndrome and its related conditions.
Introductory Discussion on Estate Planning — Webinar
Missy Zolecki 2024-01-25T15:03:03-05:00 Jan 25, 2024 |
We hosted an introductory discussion on bequests, wills, trusts, life insurance, and IRA transfers. The panel provided insight on navigating the complexities of wealth transfer, ensuring your legacy is preserved and your loved ones are well cared for.
Multidisciplinary Treatment of Fragile X Syndrome (FXS) — Webinar
Missy Zolecki 2023-12-13T13:39:54-05:00 Dec 13, 2023 |
Drs. Craig Erickson, Laura Hess, Kerrie Chitwood, and Rebecca Shaffer joined us for a one-hour Q & A discussing the benefits of a multidisciplinary team.
Fragile X-associated Tremor/Ataxia Syndrome — Webinar
Missy Zolecki 2023-12-05T10:36:12-05:00 Nov 21, 2023 |
Drs. Deborah Hall and Peter Todd joined us for an informative webinar on the topic of Fragile X-associated Tremor/Ataxia syndrome (FXTAS).
Employment Strategies and Successes — Webinar
Missy Zolecki 2023-11-02T12:20:32-04:00 Nov 2, 2023 |
In celebration of National Disability Employment Awareness Month, we were joined by a dynamic panel that shared strategies and their individual employment successes! Meaningful employment is important to parents and self-advocates!

FORWARD-MARCH 2023 — Webinar
Hilary Rosselot 2023-10-25T17:22:52-04:00 Oct 3, 2023 |
Drs. Elizabeth Berry-Kravis and Nicole Tartaglia presented updates about FORWARD-MARCH, followed by a Question and Answer session covering the specifics of the study.

2023 Industry Updates — Webinar
Kristin Bogart 2023-12-14T11:17:44-05:00 Sep 27, 2023 |
The NFXF hosted a Fragile X research updates webinar with three of our industry partners working on treatments for Fragile X syndrome. Each shared the most up-to-date information on their research project(s) in a way that is understood by those who aren’t steeped in drug research day in and day out. A short Q&A followed each presentation.
Reproductive Health — Webinar
Missy Zolecki 2023-10-26T09:43:35-04:00 Sep 6, 2023 |
Drs. Heather Hipp and Victoria Wilkins joined us for an informative webinar on the topic of Reproductive health. This is a broad topic, covering the physical and emotional components of areas like puberty, sexuality, reproductive options, and family planning.
Education Strategies for Success — Webinar
Missy Zolecki 2023-12-14T11:18:44-05:00 Aug 21, 2023 |
Educational strategies are not just helpful for school-aged children! The one-hour Q&A panel discussion, moderated by Hilary Rosselot, includes panelists with expertise and suggestions to support individuals living with Fragile X throughout the lifespan!

Housing Options for Adults With Fragile X Syndrome: Parents Share Their First-Hand Experiences — Webinar
Missy Zolecki 2023-12-14T11:33:38-05:00 Jun 28, 2023 |
Adult housing options and support services may be limited and difficult to access. A group of parents shared their firsthand experiences supporting their loved one with Fragile X determine a suitable living situation.

Aging in Fragile X Syndrome — Webinar
Kristin Bogart 2023-12-14T11:34:43-05:00 May 17, 2023 |
Dr. Berry-Kravis presents a one hour Q&A about what we’ve learned from the FORWARD data on aging for individuals living with Fragile X syndrome.
Females with FXS: Strategies for Developing Executive Functioning and Social Skills — Webinar
Barbara Haas-Givler 2023-10-26T09:41:20-04:00 May 1, 2023 |
Barb Haas-Givler presents a one-hour Q&A session on resources and strategies for the classroom, home, and community.
Peer-Reviewed Medical Research Program’s FY 23 Funding Opportunities for Researchers — Webinar
Hilary Rosselot 2023-12-14T11:41:40-05:00 Mar 23, 2023 |
2023 NIH Fragile X Centers of Excellence — Webinar
Kristin Bogart 2024-02-08T17:04:15-05:00 Jan 18, 2023 |
One Fragile X Experience: Carrier & Mom Answers FAQs — Video
Kristin Bogart 2023-12-20T09:48:18-05:00 Nov 15, 2022 |
Ilana Garber, a Fragile X carrier and mother of a child with Fragile X syndrome, shares her experience with Fragile X and offers her perspective to people interested in testing for themselves or their child.
Housing Options for Adults With Fragile X Syndrome — Webinar
Kristin Bogart 2023-12-14T12:15:37-05:00 Nov 2, 2022 |
Adult services may be limited and difficult to access, so how does a parent deal with that nagging question that keeps them up at night: What will happen when I’m gone?

Using Visual Strategies to Support Daily Function — Presentation
Kristin Bogart 2023-12-14T12:16:50-05:00 Jul 25, 2022 |
This session builds on treatment best practices and makes the concept of visual supports tangible and applicable across ages and stages of life for those with FXS.

Diagnosis and Treatment of Fragile X-Associated Tremor Ataxia Syndrome — Presentation
Kristin Bogart 2023-12-20T17:23:43-05:00 Jul 22, 2022 |
In this conference session, Dr. Deborah Hall provides a clinical overview of the diagnosis and treatment of FXTAS, including a summary of its clinical features, treatment options, and information about in-vitro fertilization.

Sibling Perspective Q&A — Panel
Kristin Bogart 2023-12-14T12:19:31-05:00 Jul 18, 2022 |
Panelists look at and discuss FXS from the perspective of siblings in the household.

Behavior Discussion and Q&A — Panel
Kristin Bogart 2023-12-14T12:21:21-05:00 Jul 17, 2022 |
With our panel of experts Rebecca Shaffer, Lauren Schmitt, Tracy Murnan Stackhouse, Elizabeth M. Berry-Kravis, and Randi J. Hagerman.

Medication for Fragile X: Anxiety, Irritable Behaviors, and Aggression — Panel
Kristin Bogart 2023-12-14T12:22:35-05:00 Jul 17, 2022 |
This session discusses the approved medications available to support behavior conditions associated with fragile X syndrome.

Assistive Technology: Tackling the Trends and the Tried & True — Presentation
Kristin Bogart 2023-12-20T17:31:23-05:00 Jul 17, 2022 |
Learn how Assistive Technology (AT) can be the vehicle affording the opportunity for a person with FXS to be more fully included in play, school, home, work, and the community.

Interpreting Clinical Trial Headlines and Announcements in Fragile X Syndrome — Presentation
Kristin Bogart 2024-01-11T17:50:35-05:00 Jul 17, 2022 |
Explore examples of recent announcements and headlines focused on treatment development in Fragile X syndrome. Presented by Elizabeth Berry-Kravis, MD, PhD, and Craig A. Erickson, MD.

The Progression of Pathology in Longitudinally Followed Patients with FXTAS — Presentation
Kristina Udice 2023-12-19T17:12:39-05:00 Jul 17, 2022 |
Learn in this webinar the pathological progression of several longitudinally followed patients with FXTAS at the UC Davis Fragile X Research and Treatment Center.

The Impact of the COVID-19 Pandemic on School-Aged Children with Fragile X — Presentation
Kristin Bogart 2024-01-11T17:54:02-05:00 Jul 17, 2022 |
Learn about research and findings from a study understanding the impact of COVID-19 and the pandemic on children with FXS.

Fragile X Medications — Panel
Kristin Bogart 2023-12-20T10:48:10-05:00 Jul 17, 2022 |
Hilary Rosselot moderates this discussion on medications for Fragile X with an expert panel that includes Drs. Elizabeth Berry-Kravis, Craig A. Erickson, Randi J. Hagerman, Reymundo Lozano, and Nicole Tartaglia.

Navigating the Challenges of Puberty and Adolescence for Youth with Fragile X Syndrome — Presentation
Contributor 2023-12-20T17:34:33-05:00 Jul 16, 2022 |
This session discusses specific challenges that arise during puberty and adolescence while also providing youth and their caregivers with some strategies and resources to assist in navigating the teenage years. Presented by Holly Harris, MD, FAAP and Dinah Godwin, LCSW.

Improving Sleeping in Children & Youth with Fragile X Syndrome — Presentation
Contributor 2023-12-20T17:43:01-05:00 Jul 16, 2022 |
This conference session discusses common sleep problems and some strategies for treating them in children with FXS, including assessments, behavioral techniques, and medications commonly used to treat sleep problems. Presented by Carol Delahunty, MD.

How Many Times Is He Going to Ask That? Perseverative Questioning and Other Behavioral Inflexibility in FXS — Presentation
Kristin Bogart 2023-12-20T23:06:24-05:00 Jul 16, 2022 |
Dr. Lauren Schmitt, a licensed clinical psychologist with extensive training in cognitive functioning in individuals with neurodevelopmental disorders, discusses how behavioral inflexibility is likened to being stuck — a state in which the individual is unable to move on from a current behavior or thought.

Common Behaviors and Available Treatments Across the Lifespan — Presentation
Contributor 2024-01-11T17:57:48-05:00 Jul 16, 2022 |
How to recognize behavioral challenges in FXS and the most common behavioral difficulties and treatment options at each developmental stage. Specific tips are shared that families can easily implement with their children or adolescents.

Experts Discuss Fragile X-Associated Tremor Ataxia Syndrome (FXTAS) — Panel
Kristin Bogart 2023-12-20T14:14:23-05:00 Jul 16, 2022 |
Explore this premutation webinar to learn more about Fragile X and Associated Tremor/Ataxia Syndrome (FXTAS) with our expert panel: Peter K. Todd, MD, Randi J. Hagerman, MD, David Hessl, PhD, and Dr. Deborah A. Hall, MD.

Experts Discuss Females with Fragile X Syndrome — Panel
Kristin Bogart 2024-04-08T16:34:34-04:00 Jul 16, 2022 |
Experts in the field discuss support and solutions for females with Fragile X in this panel discussion and Q&A. Join Jennifer Barber, Susan Howell, Barbara Haas-Givler, Nicole Tartaglia, Rebecca Shaffer, and Laurie Bridges.

Setting the Stage for Success: Effective Strategies to Promote and Support Social Skills — Presentation
Kristin Bogart 2024-04-08T16:36:28-04:00 Jul 16, 2022 |
This session discusses specific strategies to help promote and support essential social skills for those with Fragile X. Presented by Barbara Haas-Givler.

Experts Discuss Education Strategies for Fragile X Syndrome — Panel
Kristin Bogart 2023-12-20T14:30:11-05:00 Jul 16, 2022 |
Explore this deep dive into education strategies for Fragile X syndrome. Moderated by Linda Sorensen and featuring Tracy Murnan Stackhouse and Anne Hoffmann.

Promoting Inclusive Early Childhood Environments for Young Children with Fragile X Syndrome — Presentation
Kristin Bogart 2024-01-11T17:59:15-05:00 Jul 16, 2022 |
This session explores ways to promote inclusivity at a young age for children with Fragile X syndrome. Presented by Jeanine Coleman, PhD.

Hands-on Sensory & Body-Based (Somatic) Self-Regulation for Hyperarousal & Anxiety in FXS — Presentation
Kristin Bogart 2024-01-11T18:00:34-05:00 Jul 15, 2022 |
Active sensory and body-based strategies are evidence-based to directly reduce hyperarousal, tame anxiety, and limit the impact of sensory reactivity. Presented by Tracy Murnan Stackhouse.

Transitioning to Independence in Adulthood: What Helps? What Hurts? — Panel
Missy Zolecki 2023-12-20T14:39:03-05:00 Jul 15, 2022 |
A panel of experts present research-based information on the factors that promote or hinder the transition to independence of young adults with FXS after completion of high school and formal education. Panelists Leonard Abbeduto, Jessica Klusek, Angie Thurman, and Julie Lounds Taylor also take questions from the audience.

Emotional Regulation in Fragile X Syndrome — Presentation
Rebecca Shaffer 2023-12-20T14:44:20-05:00 Jul 15, 2022 |
Rebecca Shaffer discusses the signs of emotion dysregulation in Fragile X syndrome and ways to respond and practical strategies for families and caregivers.

Encouraging Early Communication: Strategies for Caregivers — Presentation
Dany Petraska 2023-12-20T14:50:12-05:00 Jul 15, 2022 |
This presentation focuses on tools that caregivers can use to encourage communication development for individuals at early stages of development. Presented by Anne Hoffman from Rush University Medical Center.

2022 Industry Updates — Presentation
Kristina Udice 2023-12-20T16:11:06-05:00 Jul 15, 2022 |
It's time for our 2022 FXS industry insights. Moderated by Hilary Rosselot, explore research, insights, clinical trials, discussions, and more with presentations by Stela Filipovic-Sadic from Asurage, Stephen O’Quinn from Zynerba Pharmaceuticals, Chad Coberly from Tetra Therapeutics, and Wayne Chadwick from Healx.

A Discussion and Q&A on the Fragile X Syndrome Research Landscape — Panel
Kristin Bogart 2024-01-11T18:04:09-05:00 Jul 15, 2022 |
How you can get involved in Fragile X research, featuring Elizabeth Berry-Kravis, Craig A. Erickson, Randi J. Hagerman, David Hessl, Tracy King, Angel Wang, Jean Luan McColl, Laurie Bridges, Kara Frech, Emily Mack, Amie Milunovich, and Missy Zolecki, and moderated by Hilary Rosselot.


No Tears for Fears in the School Age Years: IEPs — Presentation
Kristin Bogart 2024-01-11T18:57:14-05:00 Jul 15, 2022 |
This family-friendly presentation focuses on considerations for school-age children with Fragile X syndrome. Presented by Kerrie Lemons Chitwood, Laura Greiss Hess, and Anne Hoffmann.

Keynote: Clinical Trials in Fragile X Syndrome — Presentation
Dany Petraska 2023-12-20T16:57:25-05:00 Jul 15, 2022 |
This keynote session explore completed and active clinical trials relating to Fragile X syndrome. Presented by Elizabeth Berry-Kravis, Craig A. Erickson, and Randi J. Hagerman.

Starting and Monitoring Medications in Fragile X Syndrome — Presentation
Kristin Bogart 2024-01-11T19:00:30-05:00 Jul 15, 2022 |
Learn how to navigate medication strategies and approaches for children with FXS. Presented by Karen Regan, RN, and Nicole Tartaglia, MD.
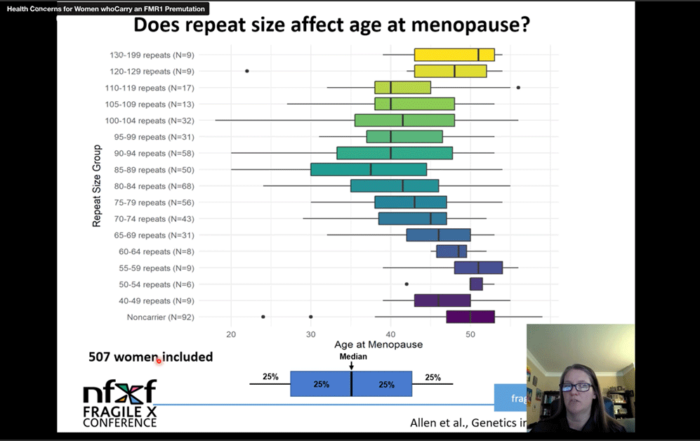
Health Concerns for Women Who Carry an FMR1 Premutation — Presentation
Kristin Bogart 2024-01-11T19:03:21-05:00 Jul 15, 2022 |
This presentation explores health conditions for women who carry an FMR1 premutation for Fragile X syndrome. Presented by Emily Allen, PhD.

Dads Helping Dads With Will Bridges — Presentation
Kristin Bogart 2024-01-11T19:04:42-05:00 Jul 15, 2022 |
Calling all fathers, papas, daddies, pop, and dads—Will Bridges discusses raising a child or children with Fragile X syndrome, followed by questions from the audience.

Fragile X Premutation Discussion — Panel
Hilary Rosselot 2023-12-20T17:20:40-05:00 Jul 14, 2022 |
Panelists Drs. David Hessl, Emily Allen, Deborah Hall and Randi Hagerman share their expertise on the Fragile X premutation at the 18th International Fragile X Conference.

Advancing Our Understanding and Future Treatment of FXTAS — Webinar
Robby Miller 2023-12-20T17:52:55-05:00 May 11, 2022 |
Experts Deborah Hall, Maureen Leehey, Peter Todd, and Nancy Hertzig discuss the latest in FXTAS research and treatment.

2022 Medications Update — Webinar
Jayne Dixon Weber 2024-02-02T17:39:52-05:00 Mar 13, 2022 |
Jayne Dixon Weber and Dr. Craig Erickson discuss new medications and review existing medications used for various issues related to Fragile X syndrome.

Using Mindfulness and Acceptance Techniques to Reduce Stress — Webinar
Kristin Bogart 2023-12-20T23:21:45-05:00 Jun 24, 2021 |
Dr. Anne Wheeler provides an overview of stress vulnerability in women with (and without) a premutation, and how mindfulness-based techniques can be helpful for increasing mindfulness, reducing stress, and improving overall well-being.

Females and Fragile X Syndrome — Webinar
Marcia Braden 2023-12-20T22:49:41-05:00 Jun 9, 2021 |
Dr. Braden helps us understand the difference between males and females with Fragile X syndrome as well as strategies for common challenges for females that include executive functioning and relationships.

The Genetics of Fragile X Disorders
Brenda Finucane 2023-12-20T22:59:42-05:00 Apr 28, 2021 |
Many thanks to Brenda Finucane for this webinar to help each of us understand the genetics of Fragile X. Brenda is a professor and licensed genetic counselor at Geisinger’s Autism and Developmental Medicine Institute in Lewisburg, PA.

What Weʼve Learned from FORWARD About Aging in Fragile X Syndrome — Webinar
Contributor 2023-12-07T08:45:36-05:00 Dec 10, 2020 |
Dr. Elizabeth Berry-Kravis discusses the FORWARD database and its role in understanding problems that face adults living with Fragile X syndrome.
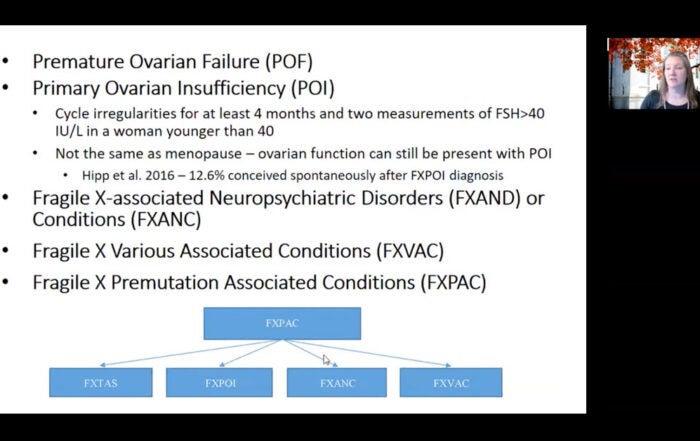
Contributor 2023-12-20T23:43:17-05:00 Nov 20, 2020 |
Emily Allen discusses some of the health concerns of women who are premutation carriers and shares research findings from her group at Emory University.

Fragile X Syndrome Self-Advocates Share Their Challenges and Triumphs — Presentation
Marcia Braden 2023-12-21T07:11:11-05:00 Aug 27, 2020 |
One of the most common questions related to Fragile X is what limitations does a person with Fragile x syndrome have? Meet Allison Cohen, Michael Cohen, Doug Cooper, Aaron Heisel, Samantha Rhodes, Joshua Rocker, Jodi Selinger, Spencer Shelton, and Cassie Stringer.
Caring for Adults with Fragile X Syndrome — Panel
Jayne Dixon Weber 2023-12-20T23:47:55-05:00 Jul 22, 2020 |
Our panel discussion from the 2020 conference on adult issues in Fragile X syndrome. Presented by Elizabeth Berry-Kravis, Marcia Braden, Jayne Dixon Weber, Craig Erickson, and Randi Hagerman.
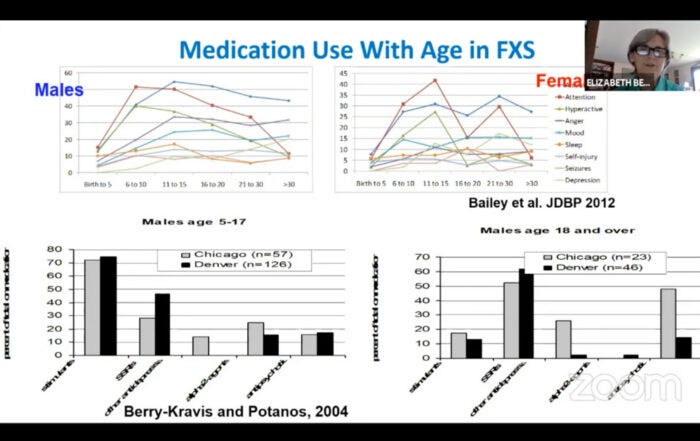
Medications for Fragile X: Anxiety, Irritable Behaviors & Aggression — Presentation
Dr. Craig Erickson 2023-12-20T23:52:38-05:00 Jul 13, 2020 |
Drs. Berry-Kravis and Erickson discuss currently available supportive medications for treating anxiety and irritable, agitated, and aggressive behaviors in Fragile X syndrome.
Premutation Carrier Concerns — Panel
Linda Sorensen 2023-12-20T23:31:11-05:00 Jun 28, 2020 |
Panelists Randi Hagerman, Heather Hipp, Emily Allen, and Marsha Mailick helps answer what it means for your health, your family, and your future at the 17th International Fragile X Conference.
Fragile X-Associated Tremor/Ataxia Syndrome 101 — Presentation
Robby Miller 2023-12-20T23:54:13-05:00 Jun 27, 2020 |
Neurologists Drs. Deborah Hall and Maureen Leehey provide an overview of FXTAS, a summary of the clinical features in the disease, and treatment options, including surgery, for FXTAS patients.
Reproductive and Gynecologic Care for Women with the Fragile X Premutation — Presentation
Jayne Dixon Weber 2023-12-20T23:56:02-05:00 Jun 27, 2020 |
Care for women with FXPOI who struggle with infertility and the health effects of early onset ovarian insufficiency, including hot flushes, night sweats, and risks of osteoporosis. With Heather Hipp, Jennifer Barber, and Keiko Mathewson.
Expert Panel Discussion on Young Children With FXS — Presentation
Linda Sorensen 2023-12-20T23:56:55-05:00 Jun 2, 2020 |
A discussion of the issues around caring for young children with Fragile X syndrome, including speech development, integrating with their typical siblings, occupational therapy, and behavioral issues. With Anne Hoffmann, Jennifer Epstein, Jeanine Coleman, and Tracy Murnan Stackhouse.

Discussion on Medications — Panel
Jayne Dixon Weber 2023-12-20T23:59:21-05:00 May 30, 2020 |
Our expert panel of Drs. Erickson, Berry-Kravis, and Hagerman discuss medications including what age anxiety medications should be considered, medications for aggressive behavior, and side effects.
Transitioning to Independence in Adulthood: What Helps? What Are the Barriers? — Panel
Contributor 2023-12-21T00:01:09-05:00 May 29, 2020 |
Our expert panel, Leonard Abbeduto, Jessica Klusek, Julie Lounds Taylor, and Angela John Thurman, present research-based information on the factors that promote and hinder the transition to independence for adults with Fragile X syndrome.

Female Power to Fight a Crisis — Presentation
Marcia Braden 2023-12-21T00:04:47-05:00 May 8, 2020 |
Females living with Fragile X syndrome were asked to give advice to other females living with FXS on how to cope with this — or any future — crisis. Listen as Marcia talks through their advice and her own advice, built on decades of working with patients with Fragile X syndrome.

Supporting Your Child with Fragile X Syndrome During Special Times — Presentation
Barbara Haas-Givler 2023-12-21T00:11:57-05:00 Apr 22, 2020 |
Barbara Haas-Givler shares her tips and methods for caregivers supporting children with Fragile X syndrome, starting with taking care of yourself first.

The How Toʼs of Sensory Diets in Fragile X Syndrome — Webinar
Tracy Murnan Stackhouse 2023-06-14T16:28:19-04:00 Apr 13, 2020 |
How To's of Sensory Diets in Fragile X Syndrome is the new video for this special time from Tracy Stackhouse from Developmental FX. A sensory diet is an occupational therapy intervention strategy devised to attain and maintain appropriate arousal states throughout each day.

Mindfulness and Me — Webinar
Tracy & Ruth, Developmental FX 2023-12-21T00:13:15-05:00 Apr 10, 2020 |
From our good friends at Developmental FX, Tracy and Ruth on using movement and meditation to deal with the anxiety and challenges of daily life being turned upside down.

My Child is Home… Now What?? Play Skills
Rebecca Shaffer 2024-02-29T14:29:10-05:00 Apr 7, 2020 |
In this webinar, Rebecca Shaffer guides us through some pointers about how to build play skills while your children are at home, and some great resources for thinking through different play opportunities while they’re at home.

My Child is Home… Now What? Keeping a Routine — Webinar
Rebecca Shaffer 2023-06-14T16:29:05-04:00 Mar 25, 2020 |
In this webinar, Rebecca Shaffer guides us through the importance of having a routine, including providing predictability and lowering anxiety, and how to create and build your own for you and your child.

Estate Planning: Special Needs Planning for Individuals and Caregivers — Webinar
Dan Whiting 2023-12-21T00:14:50-05:00 Oct 18, 2019 |
Learn how to leave money and other assets for the benefit of a child with special needs without causing the child to lose important public benefits.

Guardianship for Special Needs Individuals and Caregivers — Webinar
Jayne Dixon Weber 2023-12-21T00:16:09-05:00 Oct 4, 2019 |
Learn about if and when guardianship should be considered and the requirements needed, plus less restrictive alternatives, such as financial and healthcare powers of attorney.

Participating in Research Studies and Trials — Webinar
Dan Whiting 2023-12-21T00:23:21-05:00 Aug 2, 2019 |
Hilary Rosselot gives advice and guidance to participating in research studies and clinical trials. She is also joined by two Fragile X moms who have participated in several studies and trials and share their perspectives and advice.

Moving from Pediatrics to Adult Services — Webinar
Marcia Braden 2023-12-07T09:02:30-05:00 Sep 7, 2018 |
Dr. Marcia Braden on the difficulties associated with patients with Fragile X syndrome transitioning from a pediatric to an adult care provider.

The NFXF Conference, Research Opportunities, and a Fragile X Q&A — Webinar
Dan Whiting 2023-12-21T00:36:20-05:00 Jun 13, 2018 |
A discussion of the upcoming conference including how to get the most out of your time, what to look for, and how to have a great time and connect with the community.

Autism Spectrum Disorder in Fragile X Syndrome: Co-Occurring Conditions and Current Treatment — Webinar
Contributor 2023-03-09T19:07:20-05:00 Oct 2, 2017 |
Individuals with a dual diagnosis of Fragile X syndrome and autism have reported greater cognitive and behavioral impairment. With Dr. Walter E. Kaufmann.

Genetics 101: Fragile X Inheritance — Webinar
NFXF 2023-12-21T00:39:48-05:00 May 22, 2017 |
Whether you’re a newly-diagnosed family in the fragile X community or have been living with the diagnosis for several years, please join us as we speak with Brenda Finucane (Geisinger, ADMI Associate Director & Professor) about the nuts and bolts of Fragile X inheritance.

Adult Living Situations and Fragile X — Webinar
Jayne Dixon Weber 2023-12-07T08:55:42-05:00 Mar 31, 2017 |
As your child gets older, there are many decisions to be made. One of the biggest decisions is determining the best living situation for your young adult.
FXS Autism and Behavior with Mouse and Tracy — Valentineʼs Day Edition — Webinar
Tracy Murnan Stackhouse 2023-12-21T00:41:36-05:00 Feb 22, 2017 |
Love is in the air! Our very good friends from Developmental FX join us to answer questions about Fragile X and behavior.
Fragile X Premutation With Dr. Randi Hagerman — Webinar
NFXF 2023-12-21T00:44:51-05:00 Feb 1, 2017 |
Our very own founder, Dr. Randi Hagerman of the UC Davis MIND Institute, joins us to discuss issues in Fragile X premutation carriers. Needless to say, she is a major force in research for Fragile X and one of the biggest names in our field! Join us for a presentation and some time for Q&A!

NFXF Webinar: Being Prepared for Adulthood
Contributor 2023-12-21T00:49:42-05:00 Dec 15, 2016 |
Dr. Yankowitz discusses the adult transitions you want to start planning well before your child ages out of services. Dr. Yankowitz has more than 30 years experience working with families of individuals with developmental disabilities.
Let's Talk FXS, Autism & Behavior with Tracy & Mouse
NFXF 2015-11-20T14:01:36-05:00 Nov 20, 2015 |
Our favorite duo, Mouse & Tracy of Developmental FX, join us to discuss Fragile X, autism and behavior. As always, they provided insight and wonderful advice to our audience - and their presence is always a delight!
Let's Talk Fragile X Premutation Carriers and Health
NFXF 2015-08-04T14:04:47-04:00 Aug 4, 2015 |
Fragile X premutation carriers have long expressed concern over issues they might be facing because of having the premutation. Only in recent years has research been making headway into the reality of premutation carrier issues.

Let's Talk Behavior and FXS
NFXF 2014-01-22T14:58:07-05:00 Jan 22, 2014 |
Dr. Marcia Braden, licensed psychologist and noted Fragile X expert, leads our latest "Let's Talk" Webinar on Behavior. She begins with a presentation on "How Neurobiology Affects FXS Behavior", followed by a Q&A session from members who were part of the webinar.

Let's Talk Medication For Fragile X Syndrome
NFXF 2013-07-08T15:01:31-04:00 Jul 8, 2013 |
Given the great number of medications in the market that address specific issues with fragile X syndrome, the world of treatment can be tough for parents and other caregivers to navigate. One of the most frequent questions they ask is, "Which medication is best for my child?" The National Fragile X Foundation is pleased to address this topic in the first of its series of "Let's Talk" webinars. "Let's Talk Medication for Fragile X Syndrome" features Dr. Craig Erickson, a noted Fragile X expert and medical director of the Fragile X Clinic in Cincinnati, Ohio.
These are all of our available conference panels and presentations.
NFXF regularly schedules webinars for the Fragile X community. In addition, we share presentations and panel recordings from our biennial NFXF International Fragile X Conferences . If there are topics you’d like to see more resources for, we’d love to hear from you .
Please note that these are also all available on our various Fragile X syndrome and premutation topic pages .
All Webinars: All recordings from our ongoing webinars series’ and NFXF International Fragile X Conference panels and presentations.
Conference Sessions: NFXF International Fragile X Conference panels and presentations only.
Conference Webinars (Only)
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
- My Bibliography
- Collections
- Citation manager
Save citation to file
Email citation, add to collections.
- Create a new collection
- Add to an existing collection
Add to My Bibliography
Your saved search, create a file for external citation management software, your rss feed.
- Search in PubMed
- Search in NLM Catalog
- Add to Search
[Clinical practice guidelines for Fragile X syndrome]
- PMID: 36317200
- DOI: 10.3760/cma.j.cn511374-20220819-00564
Fragile X syndrome (FXS) is the most common monogenic form of inherited intellectual disability and autism spectrum disorder (ASD). More than 99% of individuals with FXS are caused by the unstable expansion of CGG repeats located within the 5'-untranslated region of the FMR1 gene. The clinical features of FXS include various degrees of cognitive deficit, physical, behavioral and psychiatric problems. Early treatment and prevention from having further affected children can be guided by molecular genetic testing of the FMR1 gene. The following guideline has combined the relevant research, guidelines and consensus worldwide, and summarized the genetic knowledge and clinical treatment for FXS in order to achieve a standardized diagnosis, treatment and prevention for patients and families affected by this disease.
PubMed Disclaimer
Similar articles
- Double Genetic Hit: Fragile X Syndrome and Partial Deletion of Protein Patched Homolog 1 Antisense as Cause of Severe Autism Spectrum Disorder. Saldarriaga W, Payán-Gómez C, González-Teshima LY, Rosa L, Tassone F, Hagerman RJ. Saldarriaga W, et al. J Dev Behav Pediatr. 2020 Dec;41(9):724-728. doi: 10.1097/DBP.0000000000000850. J Dev Behav Pediatr. 2020. PMID: 32947579
- Phenotypic variability to medication management: an update on fragile X syndrome. Elhawary NA, AlJahdali IA, Abumansour IS, Azher ZA, Falemban AH, Madani WM, Alosaimi W, Alghamdi G, Sindi IA. Elhawary NA, et al. Hum Genomics. 2023 Jul 7;17(1):60. doi: 10.1186/s40246-023-00507-2. Hum Genomics. 2023. PMID: 37420260 Free PMC article. Review.
- Fragile X Syndrome in children. Acero-Garcés DO, Saldarriaga W, Cabal-Herrera AM, Rojas CA, Hagerman RJ. Acero-Garcés DO, et al. Colomb Med (Cali). 2023 May 20;54(2):e4005089. doi: 10.25100/cm.v54i2.5089. eCollection 2023 Apr-Jun. Colomb Med (Cali). 2023. PMID: 37664646 Free PMC article. Review.
- Intellectual functioning and behavioural features associated with mosaicism in fragile X syndrome. Baker EK, Arpone M, Vera SA, Bretherton L, Ure A, Kraan CM, Bui M, Ling L, Francis D, Hunter MF, Elliott J, Rogers C, Field MJ, Cohen J, Maria LS, Faundes V, Curotto B, Morales P, Trigo C, Salas I, Alliende AM, Amor DJ, Godler DE. Baker EK, et al. J Neurodev Disord. 2019 Dec 26;11(1):41. doi: 10.1186/s11689-019-9288-7. J Neurodev Disord. 2019. PMID: 31878865 Free PMC article.
- Screening for FMR1 CGG Repeat Expansion in Thai Patients with Autism Spectrum Disorder. Hnoonual A, Jankittunpaiboon C, Limprasert P. Hnoonual A, et al. Biomed Res Int. 2021 Dec 8;2021:4359308. doi: 10.1155/2021/4359308. eCollection 2021. Biomed Res Int. 2021. PMID: 34926684 Free PMC article.
- Attitudes of medical professionals toward fragile X carrier screening and genetic counseling in China. Zhu Y, Li J, Pan Y, Huang W, Xi H, Duan R. Zhu Y, et al. J Community Genet. 2024 Apr;15(2):177-185. doi: 10.1007/s12687-024-00696-w. Epub 2024 Jan 26. J Community Genet. 2024. PMID: 38277068 Free PMC article.
Publication types
- Search in MeSH
Related information
Linkout - more resources, full text sources.
- Chinese Medical Association Publishing House Ltd.
- Genetic Alliance
- MedlinePlus Consumer Health Information
- MedlinePlus Health Information

- Citation Manager
NCBI Literature Resources
MeSH PMC Bookshelf Disclaimer
The PubMed wordmark and PubMed logo are registered trademarks of the U.S. Department of Health and Human Services (HHS). Unauthorized use of these marks is strictly prohibited.
- Case report
- Open access
- Published: 31 August 2024
Multiple dermatofibromas in a patient with Ehlers–Danlos syndrome: a case report
- Fatemeh Mohaghegh ORCID: orcid.org/0000-0002-8492-2410 1 ,
- Mahsa Bahraminejad ORCID: orcid.org/0000-0002-3195-4312 2 ,
- Zahra Talebzadeh ORCID: orcid.org/0000-0003-4715-5360 2 &
- Elham Tavousi Tabatabaei ORCID: orcid.org/0000-0003-1864-806X 3
Journal of Medical Case Reports volume 18 , Article number: 417 ( 2024 ) Cite this article
Metrics details
Dermatofibromas, also known as benign fibrous histiocytomas, are among the most common cutaneous soft-tissue lesions. Association of multiple dermatofibromas with some diseases was described and it has not been reported with Ehlers–Danlos syndrome before. We present a case with Ehlers–Danlos syndrome and multiple dermatofibromas.
Case presentation
An 18-year-old Iranian woman presented with multiple purple nodules ranging from 0.5 to 1.5 cm in diameter, which were mobile and located on the proximal part of the lower limb. The dimple sign of these lesions was positive. During the physical examination, several features suggestive of Ehlers–Danlos syndrome were observed, including hyperextensibility and fragility of the skin, wide atrophic scars on the upper limb, hypermobility of joints and fingers, swan neck deformities of the digits, nodules on the knee, and striae alba on the leg. The patient was previously unaware of having Ehlers–Danlos syndrome. She had a history of asthma and atopic dermatitis. Family history was negative. A biopsy of the nodular lesions was performed, and the findings confirmed the diagnosis of dermatofibromas.
We describe a patient with Ehlers–Danlos Syndrome who presented with multiple dermatofibromas. To our knowledge, this combination of findings is a previously unreported occurrence.
Peer Review reports
Ehlers–Danlos syndromes (EDS) comprise a group of relatively rare heritable connective tissue disorders characterized by defects in collagen synthesis or processing. These disorders are associated with a constellation of clinical manifestations, which may include skin hyperextensibility, joint hypermobility, and tissue fragility. The estimated overall prevalence of EDS is ~1 in 5000 individuals and encompass a spectrum of discrete disorders, with 13 recognized subtypes to date, each with its own clinical manifestations and diagnostic criteria. Among these subtypes, the underlying genetic alterations have been elucidated for 12 of these conditions, providing valuable insights into their molecular basis. Hypermobile Ehlers–Danlos syndrome (hEDS) is the most common type, but its genetic basis remains unknown, which makes diagnosis challenging since it relies on clinical evaluation [ 1 , 2 ].
The correct clinical diagnosis of EDS can be established based on family history and clinical criteria, including the degree and nature of involvement of the skin, joints, and blood vessel walls [ 3 ]. The most common cutaneous manifestations are hyperextensibility, fragility, smooth, velvety skin that bruises easily, delayed wound healing, and thin, atrophic scars after wound healing [ 2 ]. The Beighton score is the most common method used for evaluating generalized joint hypermobility in hEDS [ 1 ].
Dermatofibromas, also called benign fibrous histiocytomas, are among the most common cutaneous soft-tissue lesions. They represent a benign dermal proliferation of fibroblasts that can arise as a reactive process following various triggers, such as trauma, injections, arthropod bites, or ruptured folliculitis, but often are idiopathic [ 4 ]. Dermatofibromas typically present as firm, often hyperpigmented, nodules 0.3–1.0 cm in diameter, but giant lesions larger than 3 cm in diameter have been described [ 5 , 6 ]. Diagnosis is usually based on clinical appearance and histopathology. Multiple clustered dermatofibroma (MCDF) is a distinct variant of multiple dermatofibromas and is defined as a well-demarcated plaque composed of individual dermatofibromas [ 7 ].
Previous studies have explored potential links between EDS and various other conditions, including urinary incontinence. While multiple dermatofibromas have been associated with certain systemic diseases, with systemic lupus erythematosus (SLE) being the most prominent, followed by human immunodeficiency virus (HIV) infection [ 8 , 9 ], this presentation has not been previously reported alongside EDS. Our case report describes a patient with a confirmed diagnosis of EDS who also exhibited multiple dermatofibromas.
An 18-year-old Iranian woman presented to the al-Zahra Hospital Dermatology Clinic with the chief complaint of multiple purple nodules that appeared about 2 years ago after trauma and fracture of her left elbow, on the proximal lower limb. The patient did not have a previous diagnosis, and it was her first consultation for these lesions.
On physical examination, purple nodules with diameters ranging from 0.5 cm to 1.5 cm, which were mobile, were observed on the proximal part of the lower limb. The dimple sign of these lesions was positive (Fig. 1 ). Upon inspection, an abnormal shape of the fingers was visible, and during the examination of her whole body to detect any other signs and symptoms, we noted that the skin was hyperextensible and fragile, and wide atrophic scars were evident on the upper limb. Joints displayed hypermobility, especially in the fingers, and Swan-neck deformities of the digits were also apparent (Fig. 2 ). Some nodules were observed on her knee, and striae alba were present on her leg (Fig. 3 ). Skin-colored papules on the sides of the heel (piezogenic papules) were also seen, all of which are consistent with EDS.

Purple nodules with diameter of 0.5–1.5 cm, which are mobile have been seen on the proximal part of the lower limb

A Hypermobile finger. B Swan-neck deformity of the digi

A Atrophic scar on the elbow. B Striae alba on legs
The patient was the first child in her family, and her birth weight was 3.1 kg. She had normal growth and development during the fetal period. EDS symptoms appeared at the time of puberty as skin hyperextensibility, joint hypermobility, brittle skin, and susceptibility to ecchymosis. She mentioned a history of xerosis and atopic dermatitis since her infancy. She also had a history of respiratory disorders and had been diagnosed with asthma about 5 years ago, and inhalation corticosteroids had been prescribed for her for 2 years. She did not mention any history of cardiovascular, ophthalmic, skeletal, or gastrointestinal disorders. Family history was negative.
A biopsy of the nodular lesions was performed and showed fibrohistiocytic proliferation, acanthotic epidermis, and elongated rete ridges, which were suggestive of dermatofibroma (Fig. 4 A, B). The patient was referred to a cardiologist and ophthalmologist. The result of the echocardiography was normal, and no abnormalities in the eyes were observed. Unfortunately, the patient was not cooperative for further diagnostic studies and abstained from genetic testing and any other diagnostic assessments. Clinical diagnosis of classical EDS was established based on physical examination findings that met two major criteria (skin hyperextensibility, atrophic scarring, and generalized joint hypermobility) and two minor criteria (easy bruising and skin fragility). We also ruled out other connective tissue disorders that are included in the differential diagnosis of EDS. Finally, we educated the patient about both the benign nature of the dermatofibroma lesions and the importance of EDS follow-up.

There are papilomatous and acanthotic epidermis with basal layer pigmentation. In dermis, grenz zone and admixture of fibroblasts, histocytes, and blood vessels are seen
Discussion and conclusions
Dermatofibroma, also known as benign fibrous histiocytomas, are common, benign, painless dermal nodules that are usually solitary and measure less than 1 cm. They often appear on the extremities of middle-aged women [ 10 ]. The pathogenesis of dermatofibromas is not fully understood. Histopathologically, dermatofibromas are caused by persistent inflammatory proliferation of either histiocytes, fibroblasts, or dermal dendritic cells.
Some dermatofibromas clearly arise as a reactive process following traumas, injections, arthropod’s bites, or ruptured folliculitis [ 4 ]. Also, in literature, there are reports of multiple dermatofibroma that suggesting association with some diseases including systemic lupus erythematosus as most dominant and then HIV infection. The study that reported multiple dermatofibromas with human immunodeficiency virus (HIV) infection in three men suggests immunosuppression as the basic mechanism for incidence of multiple dermatofibromas [ 9 ]. There are examples of patients who developed dermatofibromas while taking oral prednisone (with SLE) or occurrence of new dermatofibromas related to increase in corticosteroid dosage and a dramatic reduction in the number of dermatofibromas when corticosteroid treatment was discontinued. This indicates that immunosuppressive therapies also may play a role in the pathogenesis of dermatofibroma [ 4 , 10 , 11 ]. In addition, some other conditions in comorbidities with multiple dermatofibromas have been reported including dermatomyositis, Sézary syndrome, hepatitis C, hepatocellular carcinoma, Hashimoto’s thyroiditis, myasthenia gravis, chronic myeloid leukemia, and Sjögren’s syndrome, and recently, a survey was published that showed new onset of multiple dermatofibromas in site of the shunt area after renal transplantation in patients [ 12 , 13 , 14 , 15 , 16 , 17 , 18 , 19 , 20 ].
Our patient was diagnosed with asthma 5 years ago and used an inhaled corticosteroid for 2 years. The lesions developed 1 year after withdrawing the inhaled corticosteroid. Considering the lack of systemic effects and the 1-year gap, we believe the inhaled corticosteroid is unlikely to be a factor in the development of multiple dermatofibromas (MD). Regarding the left elbow trauma and fracture, despite the initial suspicion, the different anatomical locations and the presence of multiple lesions make the association questionable.
Ehlers–Danlos syndromes are a group of inherited connective tissue disorders that can exhibit either an autosomal dominant or autosomal recessive inheritance pattern. These disorders frequently result from genetic alterations in genes responsible for encoding collagen proteins or enzymes involved in the modification of collagen [ 1 ]. In 2017, the International Classification of Ehlers–Danlos syndromes (EDS) underwent a revision, resulting in the delineation of 13 unique subtypes, with EDS hypermobility type being by far the most common and some types being quite rare. This updated classification system was based on a comprehensive analysis of the phenotypic features and the corresponding molecular pathogenesis associated with each subtype. The molecular basis is known in all subtypes except for hypermobile EDS [ 1 , 2 , 21 ]. Nevertheless, genetic testing is not performed for every patient, and the 2017 diagnostic criteria do not provide explicit guidance on the recommended scope of genetic analysis [ 1 ].
There are diagnostic criteria for hypermobile Ehlers–Danlos syndrome (hEDS) provided by International Consortium on Ehlers–Danlos Syndromes and Related Disorders with the Beighton score used for assessing joint hypermobility in patients with EDS. A revised clinical criterion for hypermobile EDS and set of diagnostic clinical criteria for all of EDS subtypes are also proposed by Malfait et al . in 2017. Based on revised diagnostic clinical criteria for EDS, a patient must meet specific criteria for a suggestive diagnosis of classical Ehlers–Danlos Syndrome (cEDS). Firstly, the patient must exhibit the major criterion of skin hyperextensibility and atrophic scarring. In addition to this major criterion, the patient must also have either the second major criterion, generalized joint hypermobility, and/or at least three minor criteria including easy bruising, soft, doughy skin, skin fragility, molluscoid pseudotumors, subcutaneous spheroids, hernia, epicanthal folds, complications of joint hypermobility, and family history of a first-degree relative who meets clinical criteria [ 1 ].
In our case, since the patient was not sufficiently cooperative, we were unable to perform genetic testing for EDS. We established a clinical diagnosis of EDS, with a high suspicion of the classical type, based on physical examination findings that met two major criteria (skin hyperextensibility, atrophic scarring, and generalized joint hypermobility) and two minor criteria (easy bruising and skin fragility). The absence of multiple fractures, short stature, and bluish sclerae in infancy are distinguishing features of osteogenesis imperfecta [ 22 , 23 ]. In patients with Marfan syndrome (MFS), disorders of the cardiovascular system, musculoskeletal system, and eyes, as well as tall stature, are common symptoms. In our case, the lack of lens abnormalities confirmed by slit-lamp examination, a normal echocardiography report, and normal stature helped us exclude MFS [ 24 ]. Cutis laxa is characterized by an absence of skin elasticity, leading to loose and sagging skin. Unlike in EDS, when tension is applied to and then released from the skin of patients with cutis laxa, it does not return to its original position due to the loss of elastin [ 25 , 26 ]. Other disorders that can closely mimic EDS in clinical presentation, such as Loeys–Dietz syndrome, arterial tortuosity syndrome, lateral meningocele syndrome, tenascin-X deficiency, Stickler syndrome, Ullrich congenital muscular dystrophy, and occipital horn syndrome (OHS), did not match the clinical manifestations observed in our case [ 27 ].
A comprehensive search of relevant databases, including Embase, Medline, Web of Science, and Google Scholar, revealed no prior reports of patients with both Ehlers–Danlos Syndrome (EDS) and multiple dermatofibromas. While a causal relationship between multiple dermatofibromas and EDS cannot be definitively established based on our single case observation, this unusual clinical presentation highlights the need for continued observation and reporting of such cases.
Availability of data and materials
All data generated or analyzed during this study are included in this published article (and its additional information files).
Abbreviations
- Ehlers–Danlos syndrome
Multiple clustered dermatofibroma
Human immunodeficiency virus
Hypermobile Ehlers–Danlos syndrome
Marfan syndrome
Occipital horn syndrome
Malfait F, Francomano C, Byers P, Belmont J, Berglund B, Black J, Bloom L, Bowen JM, Brady AF, Burrows NP, Castori M, Cohen H, Colombi M, Demirdas S, De Backer J, De Paepe A, Fournel-Gigleux S, Frank M, Ghali N, Giunta C, Grahame R, Hakim A, Jeunemaitre X, Johnson D, Juul-Kristensen B, Kapferer-Seebacher I, Kazkaz H, Kosho T, Lavallee ME, Levy H, Mendoza-Londono R, Pepin M, Pope FM, Reinstein E, Robert L, Rohrbach M, Sanders L, Sobey GJ, Van Damme T, Vandersteen A, van Mourik C, Voermans N, Wheeldon N, Zschocke J, Tinkle B. The 2017 international classification of the Ehlers-Danlos syndromes. Am J Med Genet C Semin Med Genet. 2017;175(1):8–26.
Article PubMed Google Scholar
Pope FM, Burrows NP. Ehlers-Danlos syndrome has varied molecular mechanisms. J Med Genet. 1997;34(5):400–10.
Article CAS PubMed PubMed Central Google Scholar
Beighton P, De Paepe A, Steinmann B, Tsipouras P, Wenstruo RJ. Ehlers-Danlos syndromes: revised nosology, Villefranche, 1997. Ehlers-Danlos National Foundation (USA) and Ehlers-Danlos Support Group (UK). Am J Med Genet. 1998;77(1):31–7.
Article CAS PubMed Google Scholar
Massone C, Parodi A, Virno G, Rebora A. Multiple eruptive dermatofibromas in patients with systemic lupus erythematosus treated with prednisone. Int J Dermatol. 2002;41(5):279–81.
Pusztaszeri M, Jaquet PY, Williamson C. Giant hemosiderotic dermatofibroma: a case report and review of the literature. Case Rep Dermatol. 2011;31(1):32–6.
Article Google Scholar
Requena L, Fariña MC, Fuente C, Piqué E, Olivares M, Martín L, Sánchez Yus E. Giant dermatofibroma. A little-known clinical variant of dermatofibroma. J Am Acad Dermatol. 1994;30(5 PT 1):714–8.
Bhabha FK, Magee J, Ng SY, Grills CE, Su J, Orchard D. Multiple clustered dermatofibromas presenting in a segmental distribution. Australas J Dermatol. 2016;57(1):e20–2.
McIntosh LJ, Stanitski DF, Mallett VT, Frahm JD, Richardson DA, Evans MI. Ehlers-Danlos syndrome: relationship between joint hypermobility, urinary incontinence, and pelvic floor prolapse. Gynecol Obstet Invest. 1996;41(2):135–9.
Ammirati CT, Mann C, Hornstra IK. Multiple eruptive dermatofibromas in three men with HIV infection. Dermatology. 1997;195(4):344–8.
Kravitz P. Dermatofibromas and systemic lupus erythematosus. Arch Dermatol. 1980;116(12):1347.
Lin RY, Landsman L, Krey PR, Lambert WC. Multiple dermatofibromas and systemic lupus erythematosus. Cutis. 1986;37(1):45–7.
CAS PubMed Google Scholar
Huang PY, Chu CY, Hsiao CH. Multiple eruptive dermatofibromas in a patient with dermatomyositis taking prednisolone and methotrexate. J Am Acad Dermatol. 2007;57(5 Suppl):S81–4.
Zaccaria E, Rebora A, Rongioletti F. Multiple eruptive dermatofibromas and immunosuppression: report of two cases and review of the literature. Int J Dermatol. 2008;47(7):723–7.
Taborda ML, Buffon RB, Bonamigo RR. Dermatofibromas eruptivos múltiplos e infecção pelo HCV tratada com interferon/multiple eruptive dermatofibromas and HCV infection treated by Interferon. Rev AMRIGS. 2006;50(4):334–6.
Google Scholar
García-Millán C, Aldanondo I, Fernández-Lorente M, Carrillo R, Jaén P. Multiple eruptive dermatofibromas in 2 patients infected with the human immunodeficiency virus. Actas Dermosifiliogr. 2007;98(10):702–6.
Bargman HB, Fefferman I. Multiple dermatofibromas in a patient with myasthenia gravis treated with prednisone and cyclophosphamide. J Am Acad Dermatol. 1986;14(2 Pt 2):351–2.
Kimura Y, Kaneko T, Akasaka E, Nakajima K, Aizu T, Nakano H, Sawamura D. Multiple eruptive dermatofibromas associated with Hashimoto’s thyroiditis and myasthenia gravis. Eur J Dermatol. 2010;20(4):538–9.
Alexandrescu DT, Wiernik PH. Multiple eruptive dermatofibromas occurring in a patient with chronic myelogenous leukemia. Arch Dermatol. 2005;141(3):397–8.
Tsunemi Y, Tada Y, Saeki H, Ihn H, Tamaki K. Multiple dermatofibromas in a patient with systemic lupus erythematosus and Sjögren’s syndrome. Clin Exp Dermatol. 2004;29(5):483–5.
Kayıran MA, Aksoy H, Alışır ES, Akdeniz N. Dermatological findings observed after renal transplantation in patients. J Turk Acad Dermatol. 2021;15(3):69–75.
De Paepe A, Malfait F. The Ehlers-Danlos syndrome, a disorder with many faces. Clin Genet. 2012;82(1):1–1.
Gabral WA, Makareeva E, Colige A, Letocha AD, Ty JM, Yeowell HN, Pals G, Leikin S, Marini JC. Mutations near amino end of alpha1(I) collagen cause combined osteogenesis imperfecta/Ehlers-Danlos syndrome by interference with N -propeptide processing. J Biol Chem. 2005;280(19):19259–69.
Forlino A, Cabral WA, Barnes AM, Marini JC. New perspectives on osteogenesis imperfecta. Nat Rev Endocrinol. 2011;7:540–57.
Callewaert B, Malfait F, Loeys B, Paepe AD. Ehlers-Danlos syndromes and Marfan syndromes. Best Pract Res Clin Rheumatol. 2008;22(1):165–89.
Sestak Z. Ehlers-Danlos syndrome and cutis laxa: an account of families in the Oxford area. Ann Hum Genet. 1962;25:313–21.
Banks ND, Redett RJ, Mofid MZ, Manson PN. Cutis laxa: clinical experience and outcomes. Plast Reconstr Surg. 2003;111(7):2434–42.
Murphy-Ryan M, Psychogios A, Lindor NM. Hereditary disorders of connective tissue: a guide to the emerging differential diagnosis. Genet Med. 2010;12(6):344–54.
Download references
Acknowledgements
Not applicable.
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and affiliations.
Department of Dermatology, Skin Diseases and Leishmaniosis Research Center, School of Medicine, Isfahan University of Medical Sciences, Isfahan, 81746-73461, Iran
Fatemeh Mohaghegh
Department of Dermatology, Isfahan University of Medical Science, Isfahan, Iran
Mahsa Bahraminejad & Zahra Talebzadeh
Department of Dermatology, University of Pittsburgh School of Medicine, Pittsburgh, PA, USA
Elham Tavousi Tabatabaei
You can also search for this author in PubMed Google Scholar
Contributions
1. FM: visiting and examining the patients and forming diagnostic and therapeutic plans for the patients, as well as the patient’s representative for reporting in the form of a case report, performing histologic examination on the biopsy specimens, and obtaining and preparing pathology photos. 2. MB: corresponding author, gathering and documenting all medical information related to the patient, taking the photos of patient, making a substantial contributing to the concept and design, and writing the first draft of the case report. 3. ZT: resident in charge of following up with the patients, taking an accurate history, conducting skin biopsy, and revising the manuscript critically for important intellectual content. 4. ETT: resident in charge of following up with the patients, taking an accurate history, conducting skin biopsy, and revising the manuscript critically for important intellectual content. 4. ETT: gathering articles related to the case, contributing to the writing of the article, making a substantial contributing to the concept and design, writing the largest share of the report, and involving in the patient’s care.
Corresponding author
Correspondence to Mahsa Bahraminejad .
Ethics declarations
Ethics approval and consent to participate, consent for publication.
Written informed consent was obtained from the patient for publication of this case report and any accompanying images. A copy of the written consent is available for review by the Editor-in-Chief of this journal.
Competing interests
The authors declare that they have no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/ .
Reprints and permissions
About this article
Cite this article.
Mohaghegh, F., Bahraminejad, M., Talebzadeh, Z. et al. Multiple dermatofibromas in a patient with Ehlers–Danlos syndrome: a case report. J Med Case Reports 18 , 417 (2024). https://doi.org/10.1186/s13256-024-04628-7
Download citation
Received : 28 May 2022
Accepted : 12 June 2024
Published : 31 August 2024
DOI : https://doi.org/10.1186/s13256-024-04628-7
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Multiple dermatofibromas
Journal of Medical Case Reports
ISSN: 1752-1947
- Submission enquiries: Access here and click Contact Us
- General enquiries: [email protected]
- Environment
- Science & Technology
- Business & Industry
- Health & Public Welfare
- Topics (CFR Indexing Terms)
- Public Inspection
- Presidential Documents
- Document Search
- Advanced Document Search
- Public Inspection Search
- Reader Aids Home
- Office of the Federal Register Announcements
- Using FederalRegister.Gov
- Understanding the Federal Register
- Recent Site Updates
- Federal Register & CFR Statistics
- Videos & Tutorials
- Developer Resources
- Government Policy and OFR Procedures
- Congressional Review
- My Clipboard
- My Comments
- My Subscriptions
- Sign In / Sign Up
- Site Feedback
- Search the Federal Register
This site displays a prototype of a “Web 2.0” version of the daily Federal Register. It is not an official legal edition of the Federal Register, and does not replace the official print version or the official electronic version on GPO’s govinfo.gov.
The documents posted on this site are XML renditions of published Federal Register documents. Each document posted on the site includes a link to the corresponding official PDF file on govinfo.gov. This prototype edition of the daily Federal Register on FederalRegister.gov will remain an unofficial informational resource until the Administrative Committee of the Federal Register (ACFR) issues a regulation granting it official legal status. For complete information about, and access to, our official publications and services, go to About the Federal Register on NARA's archives.gov.
The OFR/GPO partnership is committed to presenting accurate and reliable regulatory information on FederalRegister.gov with the objective of establishing the XML-based Federal Register as an ACFR-sanctioned publication in the future. While every effort has been made to ensure that the material on FederalRegister.gov is accurately displayed, consistent with the official SGML-based PDF version on govinfo.gov, those relying on it for legal research should verify their results against an official edition of the Federal Register. Until the ACFR grants it official status, the XML rendition of the daily Federal Register on FederalRegister.gov does not provide legal notice to the public or judicial notice to the courts.
Design Updates: As part of our ongoing effort to make FederalRegister.gov more accessible and easier to use we've enlarged the space available to the document content and moved all document related data into the utility bar on the left of the document. Read more in our feature announcement .
Medicare and Medicaid Programs and the Children's Health Insurance Program; Hospital Inpatient Prospective Payment Systems for Acute Care Hospitals and the Long-Term Care Hospital Prospective Payment System and Policy Changes and Fiscal Year 2025 Rates; Quality Programs Requirements; and Other Policy Changes
A Rule by the Centers for Medicare & Medicaid Services on 08/28/2024
This document has been published in the Federal Register . Use the PDF linked in the document sidebar for the official electronic format.
- Document Details Published Content - Document Details Agencies Department of Health and Human Services Centers for Medicare & Medicaid Services Agency/Docket Number CMS-1808-F CFR 42 CFR 405 42 CFR 412 42 CFR 413 42 CFR 431 42 CFR 482 42 CFR 485 42 CFR 495 42 CFR 512 Document Citation 89 FR 68986 Document Number 2024-17021 Document Type Rule Pages 68986-70046 (1061 pages) Publication Date 08/28/2024 RIN 0938-AV34 Published Content - Document Details
- View printed version (PDF)
- Document Dates Published Content - Document Dates Effective Date 10/01/2024 Dates Text With the exception of instruction 2 (Sec. 405.1845), instruction 29 (Sec. 482.42(e)) and instruction 31 (Sec. 485.640(d)), this final rule is effective October 1, 2024. The regulation at Sec. 405.1845 is effective January 1, 2025. The regulations at Sec. Sec. 482.42(e) and 485.640(d) are effective on November 1, 2024. Published Content - Document Dates
This table of contents is a navigational tool, processed from the headings within the legal text of Federal Register documents. This repetition of headings to form internal navigation links has no substantive legal effect.
FOR FURTHER INFORMATION CONTACT:
Supplementary information:, tables available on the cms website, i. executive summary and background, a. executive summary, 1. purpose and legal authority, 2. summary of the major provisions, a. continuation of the low wage index hospital policy, b. separate ipps payment for establishing and maintaining access to essential medicines, c. dsh payment adjustment, additional payment for uncompensated care, and supplemental payment, d. adoption of the patient safety structural measure in the hospital iqr program and pchqr program, e. updated hospital consumer assessment of healthcare providers and systems (hcahps) survey measure in the hospital iqr program, hospital vbp program, and pchqr program, f. hospital value-based purchasing (vbp) program, g. hospital inpatient quality reporting (iqr) program, h. pps-exempt cancer hospital quality reporting (pchqr) program, i. long-term care hospital quality reporting program (ltch qrp), j. medicare promoting interoperability program, k. proposed distribution of additional residency positions under the provisions of section 4122 of subtitle c of the consolidated appropriations act, 2023 (caa, 2023), l. extension of the medicare-dependent, small rural hospital (mdh) program and the temporary changes to the low-volume hospital payment adjustment, m. transforming episode accountability model (team), n. maternity care request for information (rfi), o. conditions of participation requirements for hospitals and critical access hospitals to report acute respiratory illnesses, p. changes to the severity level designation for z codes describing inadequate housing and housing instability, 3. summary of costs, transfers, savings, and benefits, b. background summary, 1. acute care hospital inpatient prospective payment system (ipps), 2. hospitals and hospital units excluded from the ipps, 3. long-term care hospital prospective payment system (ltch pps), 4. critical access hospitals (cahs), 5. payments for graduate medical education (gme), c. summary of provisions of recent legislation that are implemented in this final rule, 1. the consolidated appropriations act, 2023 (caa 2023; pub. l. 117-328 ), 2. the consolidated appropriations act, 2024 (caa, 2024; pub. l. 118-42 ), d. issuance of a notice of proposed rulemaking and summary of the proposed provisions, 1. proposed changes to ms-drg classifications and recalibrations of relative weights, 2. proposed changes to the hospital wage index for acute care hospitals, 3. payment adjustment for medicare disproportionate share hospitals (dshs) for fy 2025, 4. other decisions and proposed changes to the ipps for operating costs, 5. proposed fy 2025 policy governing the ipps for capital-related costs, 6. proposed changes to the payment rates for certain excluded hospitals: rate-of-increase percentages, 7. proposed changes to the ltch pps, 8. proposed changes relating to quality data reporting for specific providers and suppliers, 9. other proposals and comment solicitations included in the proposed rule, 10. other provisions of the proposed rule, 11. determining prospective payment operating and capital rates and rate-of-increase limits for acute care hospitals, 12. determining prospective payment rates for ltchs, 13. impact analysis, 14. recommendation of update factors for operating cost rates of payment for hospital inpatient services, 15. discussion of medicare payment advisory commission recommendations, e. public comments received in response to the fy 2025 ipps/ltch pps proposed rule, ii. changes to medicare severity diagnosis-related group (ms-drg) classifications and relative weights, a. background, b. adoption of the ms-drgs and ms-drg reclassifications, c. changes to specific ms-drg classifications, 1. discussion of changes to coding system and basis for fy 2025 ms-drg updates, a. conversion of ms-drgs to the international classification of diseases, 10th revision (icd-10), b. basis for fy 2025 ms-drg updates, 2. pre-mdc ms-drg 018 chimeric antigen receptor (car) t-cell and other immunotherapies, 3. mdc 01 (diseases and disorders of the nervous system), a. logic for ms-drgs 023 through 027, b. intraoperative radiation therapy (iort), 4. mdc 05 (diseases and disorders of the circulatory system), a. concomitant left atrial appendage closure and cardiac ablation, b. neuromodulation device implant for heart failure (barostim tm baroreflex activation therapy), c. endovascular cardiac valve procedures, d. ms-drg logic for ms-drg 215, 5. mdc 06 (diseases and disorders of the digestive system): excision of intestinal body parts, 6. mdc 08 (diseases and disorders of the musculoskeletal system and connective tissue), a. ms-drg logic for ms-drgs 456, 457, and 458, b. interbody spinal fusion procedures, 7. mdc 10 (endocrine, nutritional and metabolic diseases and disorders): resection of right large intestine, 8. mdc 15 (newborns and other neonates with conditions originating in perinatal period): ms-drg 795 normal newborn, 9. mdc 17 (myeloproliferative diseases and disorders, poorly differentiated neoplasms): acute leukemia, 10. review of procedure codes in ms-drgs 981 through 983 and 987 through 989, 11. operating room (o.r.) and non-o.r. procedures, a. background, b. non-o.r. procedures to o.r. procedures, (1) laparoscopic biopsy of intestinal body parts, (2) laparoscopic biopsy of gallbladder and pancreas, 12. changes to the ms-drg diagnosis codes for fy 2025, a. background of the cc list and the cc exclusions list, b. overview of comprehensive cc/mcc analysis, c. changes to severity levels, 1. sdoh—inadequate housing/housing instability, 2. causally specified delirium, d. additions and deletions to the diagnosis code severity levels for fy 2025, e. cc exclusions list for fy 2025, part 3: secondary diagnosis cc/mcc severity exclusions in select ms-drgs, 13. changes to the icd-10-cm and icd-10-pcs coding systems, 14. changes to the surgical hierarchies, 15. maintenance of the icd-10-cm and icd-10-pcs coding systems, 16. replaced devices offered without cost or with a credit, b. changes for fy 2025, 17. out of scope public comments received, d. recalibration of the fy 2025 ms-drg relative weights, 1. data sources for developing the relative weights, 2. methodology for calculation of the relative weights, b. relative weight calculation for ms-drg 018, d. cap for relative weight reductions, 3. development of national average cost-to-charge ratios (ccrs), e. add-on payments for new services and technologies for fy 2025, 1. background, a. new technology add-on payment criteria, (1) newness criterion, (2) cost criterion, (3) substantial clinical improvement criterion, b. alternative inpatient new technology add-on payment pathway, (1) alternative pathway for certain transformative new devices, (2) alternative pathway for certain antimicrobial products, c. additional payment for new medical service or technology, d. evaluation of eligibility criteria for new medical service or technology applications, e. new technology liaisons, f. application information for new medical services or technologies, 2. public input before publication of a notice of rulemaking on add-on payments, 3. icd-10-pcs section “x” codes for certain new medical services and technologies, 4. fy 2025 status of technologies receiving new technology add-on payments for fy 2024, 5. fy 2025 applications for new technology add-on payments (traditional pathway), a. casgevy tm (exagamglogene autotemcel) first indication: sickle cell disease (scd), b. casgevy tm (exagamglogene autotemcel) second indication: transfusion-dependent β-thalassemia (tdt), c. duragraft® (vascular conduit solution), d. elrexfio tm (elranatamab-bcmm) and talvey tm (talquetamab-tgvs), e. flopatch fp120, f. hepzato tm kit (melphalan for injection/hepatic delivery system), g. lantidra tm (donislecel-jujn (allogeneic pancreatic islet cellular suspension for hepatic portal vein infusion)), h. amtagvi tm (lifileucel), i. lyfgenia tm (lovotibeglogene autotemcel), j. quicktome software suite (quicktome neurological visualization and planning tool), 6. fy 2025 applications for new technology add-on payments (alternative pathways), a. annalise enterprise computed tomography brain (ctb) triage—obstructive hydrocephalus (oh), b. astar ® system, c. edwards evoque tm tricuspid valve replacement system (transcatheter tricuspid valve replacement system), d. gore® excluder® thoracoabdominal branch endoprosthesis (tambe device), e. limflow tm system, f. paradise tm ultrasound renal denervation system, g. pulseselect tm pulsed field ablation (pfa) loop catheter, h. symplicity spyral tm multi-electrode renal denervation catheter, i. triclip tm g4, j. vader® pedicle system, k. zevtera tm (ceftobiprole medocaril), 7. other comments, 8. change to the method for determining whether a technology would be within its 2- to 3-year newness period when considering eligibility for new technology add-on payments, 9. change to the requirements defining an active fda marketing application for the purpose of new technology add-on payment application eligibility, 10. change to the calculation of the inpatient new technology add-on payment for gene therapies indicated for sickle cell disease, iii. changes to the hospital wage index for acute care hospitals, 1. legislative authority, 2. core-based statistical areas (cbsas) for the fy 2025 hospital wage index, b. implementation of revised labor market area delineations, 1. micropolitan statistical areas, 2. metropolitan divisions, 3. change to county-equivalents in the state of connecticut, 4. urban counties that become rural under the revised omb delineations, 5. rural counties that become urban under the revised omb delineations, 6. urban counties that move to a different urban cbsa under the revised omb delineations, 7. transition, c. worksheet s-3 wage data for the fy 2025 wage index, 1. cost reporting periods beginning in fy 2021 for fy 2025 wage index, 2. use of wage index data by suppliers and providers other than acute care hospitals under the ipps, 3. verification of worksheet s-3 wage data, 4. process for requests for wage index data corrections, a. process for hospitals to request wage index data corrections, b. process for data corrections by cms after the january 31 public use file (puf), d. method for computing the fy 2025 unadjusted wage index, e. occupational mix adjustment to the fy 2025 wage index, 1. use of new 2022 medicare wage index occupational mix survey for the fy 2025 wage index, 2. calculation of the occupational mix adjustment for fy 2025, 3. implementation of the occupational mix adjustment and the fy 2025 occupational mix adjusted wage index, f. hospital redesignations and reclassifications, 1. urban to rural reclassification under section 1886(d)(8)(e) of the act, implemented at § 412.103, a. update to rural criteria at § 412.103(a)(1), b. policy for canceling § 412.103 reclassifications of terminated providers, 2. general policies and effects of mgcrb reclassification and treatment of dual reclassified hospitals, a. revision to allow § 412.103 hospitals to use geographic area or rural area for reclassification, 3. mgcrb reclassification issues for fy 2025, a. fy 2025 reclassification application requirements and approvals, b. effects of implementation of revised omb labor market area delineations on reclassified hospitals, (1) background, (2) assignment policy for hospitals reclassified to a cbsa where one or more counties move to the rural area or one or more rural counties move into the cbsa, (3) assignment policy for hospitals reclassified to a cbsa where the cbsa number changes, or the cbsa is subsumed by another cbsa, (4) assignment policy for hospitals reclassified to cbsas where one or more counties move to a new or different urban cbsa, (5) assignment policy for hospitals reclassified to cbsas reconfigured due to the migration to connecticut planning regions, d. change to timing of withdrawals at 412.273(c), 4. redesignations under section 1886(d)(8)(b) of the act, a. lugar status determinations, b. effects of implementation of revised omb labor market area delineations on redesignations under section 1886(d)(8)(b) of the act, g. wage index adjustments: rural floor, imputed floor, state frontier floor, out-migration adjustment, low wage index, and cap on wage index decrease policies, 1. rural floor, 2. imputed floor, 3. state frontier floor for fy 2025, 4. out-migration adjustment based on commuting patterns of hospital employees, 5. continuation of the low wage index hospital policy and budget neutrality adjustment, 6. cap on wage index decreases and budget neutrality adjustment, h. fy 2025 wage index tables, i. labor-related share for the fy 2025 wage index, iv. payment adjustment for medicare disproportionate share hospitals (dshs) for fy 2025 (§ 412.106), a. general discussion, b. eligibility for empirically justified medicare dsh payments and uncompensated care payments, c. empirically justified medicare dsh payments, d. supplemental payment for indian health service (ihs) and tribal hospitals and puerto rico hospitals, e. uncompensated care payments, 1. calculation of factor 1 for fy 2025, 2. calculation of factor 2 for fy 2025, b. factor 2 for fy 2025, 3. calculation of factor 3 for fy 2025, a. general background, b. background on the methodology used to calculate factor 3 for fy 2023 and subsequent years, (1) scaling factor, (2) new hospital policy for purposes of factor 3, (3) newly merged hospital policy, (4) ccr trim methodology, (5) uncompensated care data trim methodology, c. methodology for calculating factor 3 for fy 2025, • new hospital policy for purposes of factor 3, • newly merged hospital policy for purposes of factor 3, d. per-discharge amount of interim uncompensated care payments for fy 2025, e. process for notifying cms of merger updates and to report upload issues, f. impact on medicare dsh payment adjustment of implementation of new omb labor market delineations, g. withdrawal of 42 cfr 412.106 (fy 2004 and prior fiscal years) to the extent it included only “covered days” in the ssi ratio, v. other decisions and changes to the ipps for operating costs, a. changes to ms-drgs subject to postacute care transfer policy and ms-drg special payments policies (§ 412.4), 2. changes for fy 2025, b. changes in the inpatient hospital update for fy 2025 (§ 412.64(d)), 1. fy 2025 inpatient hospital update, 2. fy 2025 puerto rico hospital update, c. rural referral centers (rrcs) annual updates to case-mix index (cmi) and discharge criteria (§ 412.96), 1. case-mix index (cmi), 2. discharges, 3. qualification under the discharge criterion for osteopathic hospitals, d. payment adjustment for low-volume hospitals (§ 412.101), 2. extension of temporary changes to low-volume hospital payment definition and payment adjustment methodology and conforming changes to regulations, 3. payment adjustment for the portion of fy 2025 beginning on january 1, 2025, and subsequent fiscal years, 4. process for requesting and obtaining the low-volume hospital payment adjustment fy 2025, e. changes in the medicare-dependent, small rural hospital (mdh) program (§ 412.108), 1. background for the mdh program, 2. implementation of legislative extension of mdh program, 3. expiration of the mdh program, f. payment for indirect and direct graduate medical education costs (§§ 412.105 and 413.75 through 413.83), 2. distribution of additional residency positions under the provisions of section 4122 of subtitle c of the consolidated appropriations act, 2023 (caa, 2023), a. overview, b. determinations required for the distribution of residency positions, (1) determination that a hospital has a “demonstrated likelihood” of filling the positions, (2) determination that a hospital is located or treated as being located in a rural area (category one), (3) determination of hospitals for which the reference resident level of the hospital is greater than the otherwise applicable resident limit (category two), (4) determination of hospitals located in states with new medical schools, or additional locations and branch campuses (category three), (5) determination of hospitals that serve areas designated as health professional shortage areas under section 332(a)(1)(a) of the public health service act (category four), (6) determination of a qualifying hospital, c. number of residency positions made available to hospitals and limitation on individual hospitals, (1) number of residency positions made available and distribution for psychiatry or psychiatry subspecialty residencies, (2) pro rata distribution and limitation on individual hospitals, (3) prioritization of applications by hpsa score, (4) requirement for rural hospitals to expand programs, d. distributing at least 10 percent of positions to each of the four categories, e. hospital attestation to national clas standards, f. payment of additional fte residency positions awarded under section 4122 of the caa, 2023, g. aggregation of additional fte residency positions awarded under section 4122 of the caa, 2023, h. conforming regulation amendments for 42 cfr 412.105 and 42 cfr 413.79, i. prohibition on administrative and judicial review, j. application process for receiving increases in fte resident caps, 3. proposed modifications to the criteria for new residency programs and requests for information, a. newness of residents, b. summary of public comments, c. request for information, 4. technical fixes to the dgme regulations, a. correction of cross-references to § 413.79(f)(7), b. removal of obsolete regulations under § 413.79(d)(6), c. correction of typographical errors at § 413.79(k)(2)(i), 5. notice of closure of teaching hospital and opportunity to apply for available slots, b. notice of closure of sacred heart hospital located in eau claire, wi, and the application process—round 23, d. application process for available resident slots, 6. reminder of core-based statistical area (cbsa) changes and application to gme policies, g. reasonable cost payment for nursing and allied health education programs (§ 413.85 and 413.87), 2. medicare advantage nursing and allied health education payments, h. payment adjustment for certain clinical trial and expanded access use immunotherapy cases (§§ 412.85 and 412.312), i. changes to the calculation of the ipps add-on payment for certain end-stage renal disease (esrd) discharges (§ 412.104), j. separate ipps payment for establishing and maintaining access to essential medicines, 1. overview, 2. proposed list of essential medicines, 3. hospital eligibility, 4. size of the buffer stock, 5. separate payment under ipps for establishing and maintaining access to buffer stocks of essential medicines, 6. public comments, 7. policy summary, k. hospital readmissions reduction program, 1. regulatory background, 2. notice of no program proposals or updates, l. hospital value-based purchasing (vbp) program, b. fy 2025 program year payment details, 2. previously adopted quality measures for the hospital vbp program, 3. baseline and performance periods for the fy 2026 through fy 2030 program years, b. summary of baseline and performance periods for the fy 2026 through fy 2030 program years, 4. performance standards for the hospital vbp program, b. previously and newly estimated performance standards for the fy 2027 program year, c. previously established performance standards for certain measures for the fy 2028 program year, d. previously established performance standards for certain measures for the fy 2029 program year, e. newly established performance standards for certain measures for the fy 2030 program year, m. hospital-acquired condition (hac) reduction program, 2. measures for fy 2025 and subsequent years in the hac reduction program, n. rural community hospital demonstration program, 1. introduction, 2. background, 2. budget neutrality, a. statutory budget neutrality requirement, b. general budget neutrality methodology, c. budget neutrality methodology for the extension period authorized by public law 116-260, (1) methodology for estimating demonstration costs for fy 2025, (2) reconciling actual and estimated costs of the demonstration for previous years, (3) total budget neutrality offset amount for fy 2025, vi. changes to the ipps for capital-related costs, a. overview, b. additional provisions, 1. exception payments, 2. new hospitals, 3. payments for hospitals located in puerto rico, c. annual update for fy 2025, vii. changes for hospitals excluded from the ipps, a. rate-of-increase in payments to excluded hospitals for fy 2025, b. report on adjustment (exception) payments, b. critical access hospitals (cahs), 2. frontier community health integration project demonstration, a. introduction, b. background and overview, c. intervention payment and payment waivers, (1) telehealth services intervention payments, (2) ambulance services intervention payments, (3) snf/nf beds expansion intervention payments, d. budget neutrality, (1) budget neutrality requirement, (2) fchip budget neutrality methodology and analytical approach, e. policies for implementing the 5-year extension and provisions authorized by section 129 of the consolidated appropriations act, 2021 ( pub. l. 116-260 ), f. total budget neutrality offset amount for fy 2025, viii. changes to the long-term care hospital prospective payment system (ltch pps) for fy 2025, a. background of the ltch pps, 1. legislative and regulatory authority, 2. criteria for classification as an ltch, a. classification as an ltch, ii. proposed technical clarification, b. hospitals excluded from the ltch pps, 3. limitation on charges to beneficiaries, b. medicare severity long-term care diagnosis-related group (ms-ltc-drg) classifications and relative weights for fy 2025, 2. patient classifications into ms-ltc-drgs, b. changes to the ms-ltc-drgs for fy 2025, a. general overview of the ms-ltc-drg relative weights, b. development of the ms-ltc-drg relative weights for fy 2025, c. changes to the ltch pps payment rates and other changes to the ltch pps for fy 2025, 1. overview of development of the ltch pps standard federal payment rates, 2. fy 2025 ltch pps standard federal payment rate annual market basket update, b. annual update to the ltch pps standard federal payment rate for fy 2025, c. adjustment to the ltch pps standard federal payment rate under the long-term care hospital quality reporting program (ltch qrp), d. annual market basket update under the ltch pps for fy 2025, d. rebasing of the ltch market basket, 2. overview of the 2022-based ltch market basket, 3. development of the 2022-based ltch market basket cost categories and weights, a. use of medicare cost report data, (1) wages and salaries costs, (2) employee benefits costs, (3) contract labor costs, (4) pharmaceuticals costs, (5) professional liability insurance costs, (6) home office/related organization contract labor costs, (7) capital costs, b. final major cost category computation, c. derivation of the detailed operating cost weights, d. derivation of the detailed capital cost weights, e. 2022-based ltch market basket cost categories and weights, 4. selection of price proxies, a. price proxies for the operating portion of the 2022-based ltch market basket, (1) wages and salaries, (2) employee benefits, (3) electricity and other non-fuel utilities, (4) fuel: oil and gas, (5) professional liability insurance, (6) pharmaceuticals, (7) food: direct purchases, (8) food: contract purchases, (9) chemicals, (10) medical instruments, (11) rubber and plastics, (12) paper and printing products, (13) miscellaneous products, (14) professional fees: labor-related, (15) administrative and facilities support services, (16) installation, maintenance, and repair services, (17) all other: labor-related services, (18) professional fees: nonlabor-related, (19) financial services, (20) telephone services, (21) all other: nonlabor-related services, b. price proxies for the capital portion of the 2022-based ltch market basket, (1) capital price proxies prior to vintage weighting, (2) vintage weights for price proxies, c. summary of price proxies of the 2022-based ltch market basket, 5. fy 2025 market basket update for ltchs, 6. fy 2025 labor-related share, ix. quality data reporting requirements for specific providers, b. crosscutting quality program policies and request for comment, 1. adoption of the patient safety structural measure beginning with the cy 2025 reporting period/fy 2027 payment determination for the hospital inpatient quality reporting (iqr) program and the cy 2025 reporting period/fy 2027 program year for the pps-exempt cancer hospital quality reporting (pchqr) program, b. measure alignment to strategy, c. pre-rulemaking process and measure endorsement, (1) recommendation from the pre-rulemaking and measure review process, (2) endorsement and measure review, d. measure overview, e. measure calculation, f. data submission and reporting, 2. modification of the hospital consumer assessment of healthcare providers and systems (hcahps) survey measure beginning with the cy 2025 reporting period/fy 2027 payment determination for the hospital iqr program, the cy 2025 reporting period/fy 2027 program year for the pchqr program, and the fy 2030 program year for the hospital vbp program, b. overview of the modifications to the hcahps survey measure, c. measure alignment to strategy, d. pre-rulemaking process and measure endorsement, (1) recommendation from pre-rulemaking and measure review process, (2) measure endorsement, e. modification of the hcahps survey measure for the hospital iqr program beginning with the cy 2025 reporting period/fy 2027 payment determination and the pchqr program beginning with the cy 2025 reporting period/fy 2027 program year, (1) addition of the care coordination sub-measure in the updated hcahps survey measure, (2) addition of the restfulness of hospital environment sub-measure in the updated hcahps survey measure, (3) addition of the information about symptoms sub-measure in the updated hcahps survey measure, (4) modification of the responsiveness of hospital staff sub-measure in the updated hcahps survey measure, (5) removal of the care transition sub-measure in the updated hcahps survey measure, (6) modification to the “about you” section for the hospital iqr, pchqr, and hospital vbp programs, f. modifications to scoring of the hcahps survey measure for the hospital vbp program for the fy 2027 through fy 2029 program years, (2) scoring modification of the hcahps survey measure for the hospital vbp program for the fy 2027 through fy 2029 program years, g. adoption of the updated hcahps survey measure and associated scoring modifications in the hospital vbp program beginning with the fy 2030 program year, (2) adoption of the updated hcahps survey measure in the hospital vbp program beginning with the fy 2030 program year, (3) modification of the scoring of the hcahps survey measure in the hospital vbp program beginning with the fy 2030 program year, 3. advancing patient safety and outcomes across the hospital quality programs—request for comment, c. requirements for and changes to the hospital inpatient quality reporting (iqr) program, 1. background and history of the hospital iqr program, 2. retention of previously adopted hospital iqr program measures for subsequent payment determinations, 3. removal of and removal factors for hospital iqr program measures, 4. considerations in expanding and updating quality measures, 5. new measures for the hospital iqr program measure set, a. adoption of age friendly hospital measure beginning with the cy 2025 reporting period/fy 2027 payment determination, (2) overview of measure, (3) measure alignment to strategy, (4) pre-rulemaking process and measure endorsement, (a) recommendation from the prmr process, (b) measure endorsement, (5) measure calculation, (6) data submission and reporting, b. adoption of two healthcare-associated infection (hai) measures beginning with the cy 2026 reporting period/fy 2028 payment determination, (1) adoption of cauti-onc measure beginning with the cy 2026 reporting period/fy 2028 payment determination, (a) background, (b) overview of measure, (c) measure alignment to strategy, (d) pre-rulemaking process and measure endorsement, (i) recommendation from the prmr process, (ii) measure endorsement, (e) measure specifications, (f) data submission and reporting, (2) adoption of clabsi-onc measure beginning with the cy 2026 reporting period/fy 2028 payment determination, (i) recommendation from the prmr process, c. adoption of hospital harm—falls with injury ecqm beginning with the cy 2026 reporting period/fy 2028 payment determination, (4) pre-rulemaking process and measure endorsement, (5) measure specifications, d. adoption of hospital harm—postoperative respiratory failure ecqm beginning with the cy 2026 reporting period/fy 2028 payment determination, e. adoption of thirty-day risk-standardized death rate among surgical inpatients with complications (failure-to-rescue) measure beginning with the fy 2027 payment determination, 6. measure removals for the hospital iqr program measure set, a. removal of death among surgical inpatients with serious treatable complications (cms psi 04) measure beginning with the fy 2027 payment determination, b. removal of four clinical episode-based payment measures beginning with the fy 2026 payment determination, 7. refinements to current measures in the hospital iqr program measure set, a. modification of the global malnutrition composite score measure beginning with the cy 2026 reporting period/fy 2028 payment determination, (2) measure alignment to strategy, (3) overview of measure update, 8. summary of previously finalized and new hospital iqr program measures, a. summary of hospital iqr program measures for the fy 2026 payment determination, b. summary of hospital iqr program measures for the fy 2027 payment determination, c. summary of hospital iqr program measures for the fy 2028 payment determination, d. summary of hospital iqr program measures for the fy 2029 payment determination and for subsequent years, 9. form, manner, and timing of quality data submission, b. maintenance of technical specifications for quality measures, c. reporting and submission requirements for ecqms, (2) progressive increase of mandatory ecqm reporting beginning with cy 2026 reporting period/fy2028 payment determination, (a) proposed reporting and submission requirements for ecqms for the cy 2026 reporting period/fy 2028 payment determination, (b) proposed reporting and submission requirements for ecqms for the cy 2027 reporting period/fy 2029 payment determination and for subsequent years, (c) summary of proposed changes to the ecqm reporting and submission requirements, 10. validation of hospital iqr program data, b. modification of ecqm data validation beginning with the cy 2025 reporting period/fy 2028 payment determination, (1) modification of ecqm validation scoring beginning with cy 2025 ecqm data affecting the fy 2028 payment determination, (2) modification of the combined validation scoring process beginning with cy 2025 data affecting the fy 2028 payment determination, 11. data accuracy and completeness acknowledgement (daca) requirements, 12. public display requirements, 13. reconsideration and appeal procedures, 14. hospital iqr program extraordinary circumstances exceptions (ece) policy, d. changes to the pps-exempt cancer hospital quality reporting (pchqr) program, 2. adoption of the patient safety structural measure beginning with the cy 2025 reporting period/fy 2027 program year, 3. modification of the hospital consumer assessment of healthcare providers and systems (hcahps) survey measure beginning with the cy 2025 reporting period/fy 2027 program year, 4. summary of previously adopted and newly finalized pchqr program measures for the cy 2025 reporting period/fy 2027 program year and subsequent years, 5. new start date for public display of the hospital commitment to health equity measure, 6. summary of previously finalized public display policies and newly finalized public display start date change for the pchqr program, e. long-term care hospital quality reporting program (ltch qrp), 1. background and statutory authority, 2. general considerations used for the selection of quality measures for the ltch qrp, 3. quality measures currently adopted for the fy 2025 ltch qrp, 4. collection of four new items as standardized patient assessment data elements and modification of one item collected as a standardized patient assessment data element beginning with the fy 2028 ltch qrp, a. definition of standardized patient assessment data, b. social determinants of health collected as standardized patient assessment data elements, c. collect four new items as standardized patient assessment data elements beginning with the fy 2028 ltch qrp, (1) living situation, (3) utilities, d. stakeholder input, (a) comments on the living situation assessment item, (b) comments on the food assessment items, (c) comments on the utilities assessment item, e. modification of the transportation item beginning with the fy 2028 ltch qrp, 5. ltch qrp quality measure concepts under consideration for future years: request for information (rfi), 1. vaccination composite, 2. pain management, 3. depression, 4. other suggestions for future measure concepts, 1. specific criteria to use in measure selection, 2. presentation of ltch star ratings information, 3. other comments received about an ltch star ratings system, 7. form, manner, and timing of data submission under the ltch qrp, b. reporting schedule for the new items as standardized patient assessment data elements and the modified transportation item beginning with the fy 2028 ltch qrp, c. modification of the lcds admission assessment window to four days beginning with the fy 2028 ltch qrp, 8. policies regarding public display of measure data for the ltch qrp, f. medicare promoting interoperability program, 1. statutory authority for the medicare promoting interoperability program for eligible hospitals and critical access hospitals (cahs), 2. changes to the antimicrobial use and resistance (aur) surveillance measure beginning with the ehr reporting period in calendar year (cy) 2025, a. modification of the aur surveillance measure beginning with the ehr reporting period in cy 2025, b. exclusions for the au surveillance measure and the ar surveillance measure beginning with the ehr reporting period in cy 2025, c. levels of active engagement for the au surveillance measure and ar surveillance measure beginning with the ehr reporting period in cy 2025, d. scoring approach for reporting required measures in the public health and clinical data exchange objective beginning with the ehr reporting period in cy 2025, 3. overview of objectives and measures for the medicare promoting interoperability program for the ehr reporting period in cy 2025, 4. updates to the definition of cehrt in the medicare promoting interoperability program beginning with the ehr reporting period in cy 2024, 5. changes to the scoring methodology beginning with the ehr reporting period in cy 2025, 6. clinical quality measurement for eligible hospitals and cahs participating in the medicare promoting interoperability program, a. updates to clinical quality measures and reporting requirements in alignment with the hospital inpatient quality reporting (iqr) program, (2) adoption of additional ecqms, b. ecqm reporting and submission requirements for the cy 2026 reporting period and subsequent years, 7. potential future update of the safer guides measure, b. status of updates to safer guides, 8. update the definition of meaningful ehr user for healthcare providers that have committed information blocking, 9. future goals of the medicare promoting interoperability program, a. future goals with respect to fast healthcare interoperability resources® (fhir) apis for patient access, b. improving cybersecurity practices, c. improving prior authorization processes, 10. request for information regarding (rfi) public health reporting and data exchange, x. other provisions included in this final rule, a. transforming episode accountability model (team), 1. general provisions, b. basis and scope, c. definitions, d. cooperation with model evaluation and monitoring, e. rights in data and intellectual property, f. remedial action, g. cms innovation center model termination by cms, h. limitations on review, i. miscellaneous provisions on bankruptcy and other notifications, 2. transforming episode accountability model (team)—introduction, b. evidence base for model, c. ace, bpci, bpci advanced, and cjr evaluation results, d. cms innovation center specialty care strategy, 3. provisions of transforming episode accountability model, a. model performance period, team participants, participation tracks, and geographic area selection, (1) model performance period, (2) participants, (b) team participant definition, (i) team participant exclusions and considerations, (c) mandatory participation, (d) financial accountability of a team participant, (i) financial accountability considerations, (3) team participation tracks, (4) approach to select team participants and statistical power, (a) overview and options for geographic area selection, (b) exclusion of certain cbsas, (c) selection strata, (d) random selection of cbsas from strata, b. episodes, (2) overview of episodes, (3) clinical dimensions of episodes, (4) episode category definitions, (a) lower extremity joint replacement episode category, (b) surgical hip femur fracture treatment (excluding lower extremity joint replacement) episode category, (c) coronary artery bypass graft surgery episode category, (d) spinal fusion episode category, (e) major bowel procedure episode category, (5) items and services included in episodes, (a) items and services excluded from episodes, (b) beneficiary inclusion criteria, (c) initiating episodes, (d) episode length, (e) canceling episodes, c. quality measures and reporting, (2) selection of quality measures, (3) quality measures, (a) hybrid hospital-wide all-cause readmission measure with claims and electronic health record data (cmit id #356), (b) cms patient safety and adverse events composite (cms psi 90) (cmit id #135), (c) hospital-level total hip and/or total knee arthroplasty (tha/tka) patient-reported outcome-based performance measure (pro-pm) (cmit id #1618), (d) measures under consideration for future rulemaking, (4) form, manner, and timing of quality measures reporting, (5) display of quality measures and availability of information for public, d. pricing and payment methodology, (i) previous episode-based payment methodologies, (b) bpci advanced, (2) overview of team pricing and payment methodology, (3) target prices, (a) baseline period for benchmarking, (b) regional target prices, (c) services that extend beyond an episode, (d) episodes that begin in one performance year and end in the subsequent performance year, (e) high-cost outlier cap, (f) trending prices, (g) discount factor, (h) special considerations for low volume hospitals, (i) preliminary target prices, (4) risk adjustment and normalization, (5) process for reconciliation, (a) annual reconciliation, (c) team participants that experience a reorganization event, (d) updating preliminary target prices to create reconciliation target prices, (e) composite quality score, (i) overview, (ii) determining composite quality score, (f) calculating the reconciliation payment amount or repayment amount, (g) incorporating the composite quality score into the reconciliation amount, (h) limitations on npra, (i) participant responsibility for increased post-episode payments, (j) reconciliation payments and repayments, (6) appeals process, (a) first level appeal process, (b) reconsideration review process, (c) cms administrator review process, e. model overlap, (2) previous episode-based model overlap policies, (3) beneficiary overlap, (a) considerations for notification process for shared savings or total cost of care model, (b) accounting for beneficiary overlap with new cms models and programs, f. health equity, (2) identification of safety net hospitals, (b) methodological considerations, (i) cms innovation center strategy refresh safety net definition, (ii) medicare safety net index (msni), (iii) area deprivation index, (c) methodology for identifying safety net hospitals, (3) identification of rural hospitals, (b) definition of rural hospital, (4) beneficiary social risk adjustment, (5) health equity plans and reporting, (a) health equity plans, (b) demographic data collection and reporting, (c) health-related social needs data reporting, (6) other considerations, g. financial arrangements, (2) overview of team financial arrangements, (3) team collaborators, (4) sharing arrangements, (a) general, (b) requirements, (c) gainsharing payment and alignment payment conditions and limitations, (d) documentation requirements, (5) distribution arrangements, (6) downstream distribution arrangements, (7) team beneficiary incentives, (a) technology provided to a team beneficiary, (b) clinical goals of team, (c) documentation of beneficiary engagement incentives, (8) enforcement authority, (9) fraud and abuse waiver and oig safe harbor authority, h. waivers of medicare program requirements, (1) overview, (2) post-discharge home visits and homebound requirement, (3) telehealth, (4) snf 3-day rule, (a) additional beneficiary protections under the snf 3-day rule waiver, i. monitoring and beneficiary protection, (2) beneficiary choice and notification, (3) monitoring for access to care, (4) monitoring for quality of care, (5) monitoring for delayed care, j. access to records and record retention, k. data sharing, (2) beneficiary-identifiable claims data, (a) legal authority to share beneficiary-identifiable data, (b) summary and raw beneficiary-identifiable claims data reports, (c) minimum necessary data, (3) regional aggregate data, (4) timing and period of baseline period data, (5) timing and period of performance year data, (6) team data sharing agreement, (a) content of team data sharing agreement, l. referral to primary care services, m. alternative payment model options, (2) team apm options, (3) financial arrangements list and clinician engagement list, n. interoperability, o. evaluation approach, (2) design and evaluation methods, (3) data collection methods, (4) key evaluation research questions, (5) evaluation period and anticipated reports, p. decarbonization and resilience initiative, (a) climate impact on health, (b) greenhouse gas protocol and health, (c) rationale for establishing the decarbonization and resilience initiative, (i) ghg emissions are relevant to monitoring and evaluating quality outcomes, (ii) measuring ghg emissions is a key first step to reducing ghg emissions which could improve quality outcomes for beneficiaries, (iii) measuring ghg emissions could improve hospitals' resilience and beneficiaries' continuity of care, both of which impact quality outcomes, (iv) ghg emissions are relevant to reducing program expenditures, (v) ghg emissions impact on medicare, medicaid, and chip populations, (2) defining the decarbonization and resilience initiative, (3) technical assistance, (4) voluntary reporting, (a) decarbonization and resilience initiative metrics, (i) background on scope and metrics sources, (ii) scope and sources for metrics, (iii) organizational questions, (iv) building energy metrics, (v) anesthetic gas metrics, (vi) transportation metrics, (vii) request for information on scope 3 metrics and mdis, (a) scope 3 metrics, (5) report timing, (6) benefits for team participants who elect to report in the decarbonization and resiliency initiative, (a) individualized feedback reports to team participants, (b) establishment of a publicly reported hospital recognition of decarbonization commitment, (c) indirect benefits, (d) request for information on potential future incentives for participation in the voluntary decarbonization and resilience initiative, q. termination of team, b. provider reimbursement review board (prrb) (§ 405.1845), c. maternity care request for information (rfi), 2. use of medicare data for the calculation of the ipps ms-drg relative weights, 3. request for information, d. changes to the payment error rate measurement (perm), e. cop requirements for hospitals and cahs to report acute respiratory illnesses, 2. hospital respiratory illness data are and will continue to be critical for patient health and safety, 3. provisions of the proposed regulations and analysis and response to public comments, a. proposal to establish ongoing reporting for covid-19, influenza, and rsv, b. proposal to collect additional elements during a phe, c. request for information on health care reporting to the national syndromic surveillance program, xi. medpac recommendations and publicly available files, a. medpac recommendations, b. publicly available files, xii. collection of information requirements, a. statutory requirement for solicitation of comments, b. collection of information requirements, 1. icrs regarding the implementation of section 4122 of the consolidated appropriations act, 2023—distribution of additional residency positions, 2. icrs for payment adjustments for establishing and maintaining access to essential medicines, 3. icrs relating to the hospital readmissions reduction program, 4. icrs for the hospital value-based purchasing (vbp) program, 5. icrs relating to the hospital-acquired condition (hac) reduction program, 6. icrs for the hospital inpatient quality reporting (iqr) program, b. information collection burden estimate for the adoption of the age friendly hospital measure beginning with the cy 2025 reporting period/fy 2027 payment determination, c. information collection burden estimate for adoption of the patient safety structural measure beginning with the cy 2025 reporting period/fy 2027 payment determination, d. information collection burden estimate for adoption of two healthcare-associated infection (hai) measures beginning with the cy 2026 reporting period/fy 2028 payment determination, e. information collection burden for adoption of two ecqms and modification of one ecqm beginning with the cy 2026 reporting period/fy 2028 payment determination, f. information collection burden for the modification of the ecqm reporting requirements beginning with the cy 2026 reporting period/fy 2028 payment determination, g. information collection burden for the adoption of the thirty-day risk-standardized death rate among surgical inpatients with complications (failure-to-rescue) measure beginning with the july 1, 2023-june 30, 2025 reporting period/fy 2027 payment determination, h. information collection burden for the removal of five claims-based measures, i. information collection burden for the modification of the hcahps survey measure beginning with the cy 2025 reporting period/fy 2027 payment determination, j. information collection burden for changes to data validation policies, k. summary of information collection burden estimates for the hospital iqr program, 7. icrs for pps-exempt cancer hospital quality reporting (pchqr) program, a. information collection burden estimate for the adoption of the patient safety structural measure beginning with the cy 2025 reporting period/fy 2027 program year, b. information collection burden for the modification of the hcahps survey beginning with the cy 2025 reporting period/fy 2027 program year, c. information collection burden estimate for the policy to move up the start date of public display of the hospital commitment to health equity measure, d. summary of information collection burden estimates for the pchqr program, 8. icrs for the long-term care hospital quality reporting program (ltch qrp), 9. icrs for the medicare promoting interoperability program, b. information collection burden for the adoption of the two ecqms and modification of one ecqm beginning with the cy 2026 reporting period, c. information collection burden for the modification of the ecqm reporting requirements beginning with the cy 2026 reporting period, d. information collection burden for the separation of the aur surveillance measure into two measures beginning with the ehr reporting period in cy 2025, e. information collection burden for the increase to the minimum scoring threshold beginning with the ehr reporting period in cy 2025, f. summary of estimates used to calculate the collection of information burden, 10. icrs for the transforming episode accountability model, 11. icrs for payment error rate measurement (perm), a. icrs regarding § 431.970 information submission and systems access requirements, b. icrs regarding § 431.992 corrective action plan, c. icrs regarding § 431.998 difference resolution and appeal process, 12. icrs for the cop requirements for hospitals and cahs to report acute respiratory illnesses, a. ongoing reporting, b. phe reporting, list of subjects, 42 cfr part 405, 42 cfr part 412, 42 cfr part 413, 42 cfr part 431, 42 cfr part 482, 42 cfr part 485, 42 cfr part 495, 42 cfr part 512, part 405—federal health insurance for the aged and disabled, part 412—prospective payment systems for inpatient hospital services, part 413—principles of reasonable cost reimbursement; payment for end-stage renal disease services; optional prospectively determined payment rates for skilled nursing facilities, part 431—state organization and general administration, part 482—conditions of participation for hospitals, part 485—conditions of participation: specialized providers, part 495—standards for the electronic health record technology incentive program, part 512—standard provisions for innovation center models and specific provisions for certain models, subpart d [reserved], subpart e—transforming episode accountability model (team), team participation, scope of episodes being tested, pricing methodology, quality measures and composite quality score, reconciliation and review process, data sharing and other requirements, financial arrangements and beneficiary incentives, medicare program waivers, general provisions, the following will not publish in the code of federal regulations addendum—schedule of standardized amounts, update factors, rate-of-increase percentages effective with cost reporting periods beginning on or after october 1, 2024, and payment rates for ltchs effective for discharges occurring on or after october 1, 2024, i. summary and background, ii. changes to prospective payment rates for hospital inpatient operating costs for acute care hospitals for fy 2025, a. calculation of the adjusted standardized amount, 1. standardization of base-year costs or target amounts, 2. computing the national average standardized amount, 3. updating the national average standardized amount, 4. methodology for calculation of the average standardized amount, a. reclassification and recalibration of ms-drg relative weights before cap, b. budget neutrality adjustment for reclassification and recalibration of ms-drg relative weights with cap, c. updated wage index—budget neutrality adjustment, d. reclassified hospitals—budget neutrality adjustment, f. continuation of the low wage index hospital policy—budget neutrality adjustment, g. permanent cap policy for wage index—budget neutrality adjustment, h. rural community hospital demonstration program adjustment, i. outlier payments, (1) methodology to incorporate an estimate of the impact of outlier reconciliation in the fy 2025 outlier fixed-loss cost threshold, (a) incorporating a projection of outlier reconciliations for the fy 2025 outlier threshold calculation, (b) reduction to the fy 2025 capital standard federal rate by an adjustment factor to account for the projected proportion of capital ipps payments paid as outliers, (2) fy 2025 outlier fixed-loss cost threshold, (3) other changes concerning outliers, (4) fy 2023 outlier payments, 5. fy 2025 standardized amount, b. adjustments for area wage levels and cost-of-living, 1. adjustment for area wage levels, 2. adjustment for cost-of-living in alaska and hawaii, c. calculation of the prospective payment rates, 1. general formula for calculation of the prospective payment rates for fy 2025, 2. operating and capital federal payment rate and outlier payment calculation, 3. hospital-specific rate (applicable only to schs and mdhs), a. calculation of hospital-specific rate, b. updating the fy 1982, fy 1987, fy 1996, fy 2002 and fy 2006 hospital-specific rate for fy 2025, iii. changes to payment rates for acute care hospital inpatient capital-related costs for fy 2025, a. determination of the federal hospital inpatient capital-related prospective payment rate update for fy 2025, 1. projected capital standard federal rate update, 2. outlier payment adjustment factor, 3. budget neutrality adjustment factor for changes in drg classifications and weights and the gaf, 4. capital federal rate for fy 2025, b. calculation of the inpatient capital-related prospective payments for fy 2025, c. capital input price index, 2. forecast of the cipi for fy 2025, iv. changes to payment rates for excluded hospitals: rate-of-increase percentages for fy 2025, v. changes to the payment rates for the ltch pps for fy 2025, a. ltch pps standard federal payment rate for fy 2025, 2. development of the fy 2025 ltch pps standard federal payment rate, b. adjustment for area wage levels under the ltch pps for fy 2025, 2. geographic classifications (labor market areas) under the ltch pps, a. urban counties that will become rural under the revised omb delineations, b. rural counties that will become urban under the revised omb delineations, c. urban counties that will move to a different urban cbsa under the revised omb delineations, d. change to county-equivalents in the state of connecticut, 3. labor-related share for the ltch pps standard federal payment rate, 4. wage index for fy 2025 for the ltch pps standard federal payment rate, 5. permanent cap on wage index decreases, a. permanent cap on ltch pps wage index decreases, b. permanent cap on ipps comparable wage index decreases, 6. budget neutrality adjustments for changes to the ltch pps standard federal payment rate area wage level adjustment, c. cost-of-living adjustment (cola) for ltchs located in alaska and hawaii, d. adjustment for ltch pps high cost outlier (hco) cases, 1. hco background, 2. determining ltch ccrs under the ltch pps, b. ltch total ccr ceiling, c. ltch statewide average ccrs, d. reconciliation of hco payments, 3. high-cost outlier payments for ltch pps standard federal payment rate cases, a. high-cost outlier payments for ltch pps standard federal payment rate cases, b. fixed-loss amount for ltch pps standard federal payment rate cases for fy 2025, (1) charge inflation factor for use in determining the fixed-loss amount for ltch pps standard federal payment rate cases for fy 2025, (2) ccrs for use in determining the fixed-loss amount for ltch pps standard federal payment rate cases for fy 2025, (3) proposed fixed-loss amount for ltch pps standard federal payment rate cases for fy 2025, 4. high-cost outlier payments for site neutral payment rate cases, e. update to the ipps comparable amount to reflect the statutory changes to the ipps dsh payment adjustment methodology, f. computing the adjusted ltch pps federal prospective payments for fy 2025, vi. tables referenced in this final rule generally available through the internet on the cms website, appendix a: economic analyses, i. regulatory impact analysis, a. statement of need, a. update to the ipps payment rates, b. changes for the add-on payments for new services and technologies, c. continuation of the low wage index hospital policy, d. implementation of section 4122 of the consolidated appropriations act, 2023 (caa, 2023), e. additional payment for uncompensated care to medicare disproportionate share hospitals (dshs) and supplemental payment, f. rural community hospital demonstration program, 2. frontier community health integration project (fchip) demonstration, 3. update to the ltch pps payment rates, 4. hospital quality programs, 5. other provisions, a. transforming episode accountability model (team), b. provider reimbursement review board (prrb), c. payment error rate measurement (perm), d. hospital cop reporting requirements, b. overall impact, c. objectives of the ipps and the ltch pps, d. limitations of our analysis, e. hospitals included in and excluded from the ipps, f. quantitative effects of the policy changes under the ipps for operating costs, 1. basis and methodology of estimates, 2. analysis of table i, a. effects of the hospital update (column 1), b. effects of the changes to the ms-drg reclassifications and relative cost-based weights with recalibration budget neutrality (column 2), c. effects of the wage index changes (column 3), d. effects of mgcrb reclassifications (column 4), e. effects of the rural floor, including application of national budget neutrality (column 5), f. effects of the application of the imputed floor, frontier state wage index and out-migration adjustment (column 6), g. effects of the expiration of mdh special payment status (column 7), h. effects of all fy 2025 changes (column 8), 3. impact analysis of table ii, 4. impact analysis of table iii: provider deciles by beneficiary characteristics, c. social determinants of health (sdoh), d. behavioral health, e. disability, g. geography, 1. effects of the policy changes relating to new medical service and technology add-on payments, a. fy 2025 status of technologies approved for fy 2024 new technology add-on payments, b. fy 2025 applications for new technology add-on payments, c. total estimated costs for new technology add-on payments in fy 2025, 2. medicare dsh uncompensated care payments and supplemental payment for indian health service hospitals and tribal hospitals and hospitals located in puerto rico, 3. effects of the changes to low-volume hospital payment adjustment policy, 4. effects of the distribution of additional residency positions under the provisions of section 4122 of subtitle c of the consolidated appropriations act, 2023 (caa, 2023), 5. effects of changes to additional payment for hospitals with a high percentage of esrd beneficiary discharges, 6. estimated effects of the ipps payment adjustment for establishing and maintaining access to essential medicines, 7. effects under the hospital readmissions reduction program for fy 2025, 8. effects of changes under the fy 2025 hospital value-based purchasing (vbp) program, 9. effects under the hac reduction program for fy 2025, 10. effects of implementation of the rural community hospital demonstration program in fy 2024, 11. effects of continued implementation of the frontier community health integration project (fchip) demonstration, 12. effects of proposed implementation of the transforming episode accountability model (team), a. effects on the medicare program, (1) assumptions, (2) sensitivity analysis, b. effects on the medicare beneficiaries, c. aggregate effects on the market, 13. effects of changes the provider reimbursement review board (prrb) membership, 14. effects of the removal of the puerto rico exclusion from payment error rate measurement (perm) review, 15. effects of hospital and cah reporting of acute respiratory illnesses, h. effects on hospitals and hospital units excluded from the ipps, i. effects of changes in the capital ipps, 1. general considerations, j. effects of payment rate changes and policy changes under the ltch pps, 1. introduction and general considerations, 2. impact on rural hospitals, 3. anticipated effects of the ltch pps payment rate changes and policy changes, a. budgetary impact, b. impact on providers, c. calculation of ltch pps payments for ltch pps standard federal payment rate cases, (1) location, (2) participation date, (3) ownership control, (4) census region, (5) bed size, 4. effect on the medicare program, 5. effect on medicare beneficiaries, k. effects of requirements for the hospital inpatient quality reporting (iqr) program, l. effects of new requirements for the pps-exempt cancer hospital quality reporting (pchqr) program, m. effects of requirements for the long-term care hospital quality reporting program (ltch qrp), n. effects of requirements regarding the medicare promoting interoperability program, o. alternatives considered, 1. alternatives considered for the distribution of additional residency positions under the provisions of section 4122 of subtitle c of the consolidated appropriations act, 2023 (caa, 2023), 2. alternative considered for the separate ipps payment for establishing and maintaining access to essential medicines, 3. alternatives considered to the ltch qrp reporting requirements, 4. alternatives considered for the transforming episode accountability model, p. overall conclusion, 1. acute care hospitals, q. regulatory review cost estimation, ii. accounting statements and tables, a. acute care hospitals, iii. regulatory flexibility act (rfa) analysis, iv. impact on small rural hospitals, v. unfunded mandates reform act analysis, vi. executive order 13132, vii. executive order 13175, viii. executive order 12866, appendix b: recommendation of update factors for operating cost rates of payment for inpatient hospital services, i. background, ii. inpatient hospital update for fy 2025, a. fy 2025 inpatient hospital update, b. fy 2025 sch and mdh update, c. fy 2025 puerto rico hospital update, d. update for hospitals excluded from the ipps for fy 2025, e. update for ltchs for fy 2025, iii. secretary's recommendations, iv. medpac recommendation for assessing payment adequacy and updating payments in traditional medicare.
Comments are no longer being accepted. See DATES for details.
Additional information is not currently available for this document.
- Sharing Enhanced Content - Sharing Shorter Document URL https://www.federalregister.gov/d/2024-17021 Email Email this document to a friend Enhanced Content - Sharing
- Print this document
Document page views are updated periodically throughout the day and are cumulative counts for this document. Counts are subject to sampling, reprocessing and revision (up or down) throughout the day.
This document is also available in the following formats:
More information and documentation can be found in our developer tools pages .
This PDF is the current document as it appeared on Public Inspection on 08/01/2024 at 4:15 pm.
It was viewed 0 times while on Public Inspection.
If you are using public inspection listings for legal research, you should verify the contents of the documents against a final, official edition of the Federal Register. Only official editions of the Federal Register provide legal notice of publication to the public and judicial notice to the courts under 44 U.S.C. 1503 & 1507 . Learn more here .
Document headings vary by document type but may contain the following:
- the agency or agencies that issued and signed a document
- the number of the CFR title and the number of each part the document amends, proposes to amend, or is directly related to
- the agency docket number / agency internal file number
- the RIN which identifies each regulatory action listed in the Unified Agenda of Federal Regulatory and Deregulatory Actions
See the Document Drafting Handbook for more details.
Department of Health and Human Services
Centers for medicare & medicaid services.
- 42 CFR Parts 405, 412, 413, 431, 482, 485, 495, and 512
- [CMS-1808-F]
- RIN 0938-AV34
Centers for Medicare & Medicaid Services (CMS), Department of Health and Human Services (HHS).
Final rule.
This final rule revises the Medicare hospital inpatient prospective payment systems (IPPS) for operating and capital-related costs of acute care hospitals; makes changes relating to Medicare graduate medical education (GME) for teaching hospitals; updates the payment policies and the annual payment rates for the Medicare prospective payment system (PPS) for inpatient hospital services provided by long-term care hospitals (LTCHs); and makes other policy-related changes.
With the exception of instruction 2 (§ 405.1845), instruction 29 (§ 482.42(e)) and instruction 31 (§ 485.640(d)), this final rule is effective October 1, 2024. The regulation at § 405.1845 is effective January 1, 2025. The regulations at §§ 482.42(e) and 485.640(d) are effective on November 1, 2024.
Donald Thompson, and Michele Hudson, (410) 786-4487 or [email protected] , Operating Prospective Payment, MS-DRG Relative Weights, Wage Index, Hospital Geographic Reclassifications, Graduate Medical Education, Capital Prospective Payment, Excluded Hospitals, Medicare Disproportionate Share Hospital (DSH) Payment Adjustment, Sole Community Hospitals (SCHs), Medicare-Dependent Small Rural Hospital (MDH) Program, Low-Volume Hospital Payment Adjustment, and Inpatient Critical Access Hospital (CAH) Issues.
Emily Lipkin, and Jim Mildenberger, [email protected] , Long-Term Care Hospital Prospective Payment System and MS-LTC-DRG Relative Weights Issues.
Lily Yuan, [email protected] , New Technology Add-On Payments Issues.
Mady Hue, [email protected] , and Andrea Hazeley, [email protected] , MS-DRG Classifications Issues.
Jonathan Rudy, [email protected] , Rural Community Hospital Demonstration Program Issues.
Jeris Smith, [email protected] , Frontier Community Health Integration Project (FCHIP) Demonstration Issues.
Lang Le, [email protected] , Hospital Readmissions Reduction Program—Administration Issues.
Ngozi Uzokwe, [email protected] , Hospital Readmissions Reduction Program—Measures Issues.
Jennifer Tate, [email protected] , Hospital-Acquired Condition Reduction Program—Administration Issues.
Ngozi Uzokwe, [email protected] , Hospital-Acquired Condition Reduction Program—Measures Issues.
Julia Venanzi, [email protected] , Hospital Inpatient Quality Reporting Program and Hospital Value-Based Purchasing Program—Administration Issues.
Melissa Hager, [email protected] , and Ngozi Uzokwe, [email protected] —Hospital Inpatient Quality Reporting Program and Hospital Value-Based Purchasing Program—Measures Issues Except Hospital Consumer Assessment of Healthcare Providers and Systems Issues.
Elizabeth Goldstein, [email protected] , Hospital Inpatient Quality Reporting and Hospital Value-Based Purchasing—Hospital Consumer Assessment of Healthcare Providers and Systems Measures Issues.
Jennifer Tate, [email protected] , PPS-Exempt Cancer Hospital Quality Reporting—Administration Issues.
Kristina Rabarison, [email protected] , PPS-Exempt Cancer Hospital Quality Reporting Program—Measure Issues.
Lorraine Wickiser, [email protected] , Long-Term Care Hospital Quality Reporting Program—Administration Issues.
Jessica Warren, [email protected] and Elizabeth Holland, [email protected] , Medicare Promoting Interoperability Program.
Bridget Dickensheets, [email protected] and Mollie Knight, [email protected] , LTCH Market Basket Rebasing.
Benjamin Cohen, [email protected] , Provider Reimbursement Review Board.
Nicholas Bonomo, [email protected] and Tracy Smith, [email protected] , Payment Error Rate Measurement Program.
[email protected] , Transforming Episode Accountability Model (TEAM).
Lauren Blum, [email protected] , and Kristin Shifflett, [email protected] , Conditions of Participation Requirements for Hospitals and Critical Access Hospitals to Report Acute Respiratory Illnesses.
The IPPS tables for this fiscal year (FY) 2025 final rule are available on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html . Click on the link on the left side of the screen titled “FY 2025 IPPS Final Rule Home Page” or “Acute Inpatient—Files for Download.” The LTCH PPS tables for this FY 2025 final rule are available on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/LongTermCareHospitalPPS/index.html under the list item for Regulation Number CMS-1808-F. For further details on the contents of the tables referenced in this final rule, we refer readers to section VI. of the Addendum to this FY 2025 IPPS/LTCH PPS final rule.
Readers who experience any problems accessing any of the tables that are posted on the CMS websites, as previously identified, should contact Michael Treitel, [email protected] .
This FY 2025 IPPS/LTCH PPS final rule makes payment and policy changes under the Medicare inpatient prospective payment system (IPPS) for operating and capital-related costs of acute care hospitals as well as for certain hospitals and hospital units excluded from the IPPS. In addition, it makes payment and policy changes for inpatient hospital services provided by long-term care hospitals (LTCHs) under the long-term care hospital prospective payment system (LTCH PPS). This final rule also makes policy changes to ( print page 68987) programs associated with Medicare IPPS hospitals, IPPS-excluded hospitals, and LTCHs. In this FY 2025 final rule, we are finalizing our proposal to continue policies to address wage index disparities impacting low wage index hospitals. We are also finalizing our proposed changes relating to Medicare graduate medical education (GME) for teaching hospitals and new technology add-on payments.
We are finalizing our proposal of a separate IPPS payment for establishing and maintaining access to essential medicines.
In the Hospital Value-Based Purchasing (VBP) Program, we are finalizing our proposal to modify scoring of the Person and Community Engagement Domain for the FY 2027 through FY 2029 program years to only score six unchanged dimensions of the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) Survey measure, and we are finalizing our proposal to adopt the updated HCAHPS Survey measure in the Hospital VBP Program beginning with the FY 2030 program year after the updated measure has been publicly reported under the Hospital Inpatient Quality Reporting (IQR) Program for 1 year. We are also finalizing our proposal to modify scoring on the HCAHPS Survey measure beginning with the FY 2030 program year to incorporate the updated HCAHPS Survey measure into nine survey dimensions. Lastly, we provide previously and newly established performance standards for the FY 2027 through FY 2030 program years for the Hospital VBP Program.
In the Hospital IQR Program, we are finalizing our proposals to add seven new measures, with modifications to our proposal to adopt the Patient Safety Structural measure, modify two existing measures including the HCAHPS Survey measure, and remove five measures. We are also finalizing our proposed changes to the validation process for the Hospital IQR Program data. We are finalizing the proposed reporting and submission requirements for electronic clinical quality measures (eCQMs) with modifications.
In the PPS-Exempt Cancer Hospital Quality Reporting Program (PCHQR), we are finalizing the adoption of the Patient Safety Structural measure with modification beginning with the CY 2025 reporting period/FY 2027 program year. We are also finalizing our proposed changes to the HCAHPS Survey measure and our proposal to move up the start date for publicly displaying hospital performance on the Hospital Commitment to Health Equity measure.
In the LTCH Quality Reporting Program (QRP), we are finalizing our proposals to add four assessment items to the LTCH Continuity Assessment Record and Evaluation (CARE) Data Set (LCDS) and modify one assessment item on the LCDS beginning with the FY 2028 LTCH QRP. Additionally, we are finalizing our proposal to extend the admission assessment window for the LCDS beginning with the FY 2028 LTCH QRP. Finally, we summarize the feedback we received on our requests for information on future measure concepts for the LTCH QRP and a future LTCH Star Rating system.
In the Medicare Promoting Interoperability Program, we are finalizing our proposal to separate the Antimicrobial Use and Resistance (AUR) Surveillance measure into two measures, an Antimicrobial Use (AU) Surveillance measure and an Antimicrobial Resistance (AR) Surveillance measure, beginning with the electronic health record (EHR) reporting period in CY 2025. We are also finalizing the following proposals to: increase the performance-based scoring threshold from 60 points to 70 points for the EHR reporting period in CY 2025 and from 70 points to 80 points beginning with the EHR reporting period in CY 2026; adopt two new eCQMs and modify one eCQM, in alignment with the Hospital IQR Program; and change the reporting and submission requirements for eCQMs with modifications, in alignment with the Hospital IQR Program.
We proposed the creation and testing of a new mandatory alternative payment model called the Transforming Episode Accountability Model (TEAM). The intent of TEAM is to improve beneficiary care through financial accountability for episodes categories that begin with one of the following procedures: coronary artery bypass graft surgery (CABG), lower extremity joint replacement (LEJR), major bowel procedure, surgical hip/femur fracture treatment (SHFFT), and spinal fusion. TEAM will test whether financial accountability for these episode categories reduces Medicare expenditures while preserving or enhancing the quality of care for Medicare beneficiaries. We anticipated that TEAM would benefit Medicare beneficiaries through improving the coordination of items and services paid for through Medicare fee-for-service (FFS) payments, encouraging provider investment in health care infrastructure and redesigned care processes, and incentivizing higher value care across the inpatient and post-acute care settings for the episode. We proposed to test TEAM for a 5-year model performance period, beginning January 1, 2026, and ending December 31, 2030. Under the Quality Payment Program (QPP), we anticipated that TEAM would be an Advanced Alternative Payment Model (APM)for Track 2 and Track 3 and a Merit-based Incentive Payment System (MIPS) APM for all participation tracks. We are finalizing some policies as proposed and we are finalizing others with modification. There are also certain proposed policies that we are not finalizing, and we will instead go through future rulemaking to promulgate new policies before the model start date.
We are also finalizing the proposal requiring respiratory illness reporting for hospitals and critical access hospitals as a condition of participation following the expiration of the COVID-19 public health emergency requirements.
Under various statutory authorities, we either discuss continued program implementation or make changes to the Medicare IPPS, the LTCH PPS, other related payment methodologies and programs for FY 2025 and subsequent fiscal years, and other policies and provisions included in this rule. These statutory authorities include, but are not limited to, the following:
- Section 1886(d) of the Social Security Act (the Act), which sets forth a system of payment for the operating costs of acute care hospital inpatient stays under Medicare Part A (Hospital Insurance) based on prospectively set rates. Section 1886(g) of the Act requires that, instead of paying for capital-related costs of inpatient hospital services on a reasonable cost basis, the Secretary use a prospective payment system (PPS).
- Section 1886(d)(1)(B) of the Act, which specifies that certain hospitals and hospital units are excluded from the IPPS. These hospitals and units are: rehabilitation hospitals and units; LTCHs; psychiatric hospitals and units; children's hospitals; cancer hospitals; extended neoplastic disease care hospitals; and hospitals located outside the 50 States, the District of Columbia, and Puerto Rico (that is, hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa). Religious nonmedical health care institutions (RNHCIs) are also excluded from the IPPS.
- Sections 123(a) and (c) of the Balanced Budget Refinement Act of 1999 (BBRA) (Public Law (Pub. L.) 106-113) and section 307(b)(1) of the Benefits Improvement and Protection Act of 2000 (BIPA) ( Pub. L. 106-554 ) (as codified under section 1886(m)(1) of the ( print page 68988) Act), which provide for the development and implementation of a prospective payment system for payment for inpatient hospital services of LTCHs described in section 1886(d)(1)(B)(iv) of the Act.
- Section 1814(l)(4) of the Act requires downward adjustments to the applicable percentage increase, beginning with FY 2015, for CAHs that do not successfully demonstrate meaningful use of certified electronic health record technology (CEHRT) for an EHR reporting period for a payment adjustment year.
- Section 1886(a)(4) of the Act, which specifies that costs of approved educational activities are excluded from the operating costs of inpatient hospital services. Hospitals with approved graduate medical education (GME) programs are paid for the direct costs of GME in accordance with section 1886(h) of the Act. Hospitals paid under the IPPS with approved GME programs are paid for the indirect costs of training residents in accordance with section 1886(d)(5)(B) of the Act.
- Section 1886(d)(5)(F) of the Act provides for additional Medicare IPPS payments to subsection (d) hospitals that serve a significantly disproportionate number of low-income patients. These payments are known as the Medicare disproportionate share hospital (DSH) adjustment. Section 1886(d)(5)(F) of the Act specifies the methods under which a hospital may qualify for the DSH payment adjustment.
- Section 1886(b)(3)(B)(viii) of the Act, which requires the Secretary to reduce the applicable percentage increase that would otherwise apply to the standardized amount applicable to a subsection (d) hospital for discharges occurring in a fiscal year if the hospital does not submit data on measures in a form and manner, and at a time, specified by the Secretary.
- Section 1886(b)(3)(B)(ix) of the Act, which requires downward adjustments to the applicable percentage increase, beginning with FY 2015 (and beginning with FY 2022 for subsection (d) Puerto Rico hospitals), for eligible hospitals that do not successfully demonstrate meaningful use of CEHRT for an EHR reporting period for a payment adjustment year.
- Section 1866(k) of the Act, which provides for the establishment of a quality reporting program for hospitals described in section 1886(d)(1)(B)(v) of the Act, referred to as “PPS-exempt cancer hospitals.”
- Section 1886(n) of the Act, which establishes the requirements for an eligible hospital to be treated as a meaningful EHR user of CEHRT for an EHR reporting period for a payment adjustment year or, for purposes of subsection (b)(3)(B)(ix) of the Act, for a fiscal year.
- Section 1886(o) of the Act, which requires the Secretary to establish a Hospital Value-Based Purchasing (VBP) Program, under which value-based incentive payments are made in a fiscal year to hospitals based on their performance on measures established for a performance period for such fiscal year.
- Section 1886(p) of the Act, which establishes a Hospital-Acquired Condition (HAC) Reduction Program, under which payments to applicable hospitals are adjusted to provide an incentive to reduce hospital-acquired conditions.
- Section 1886(q) of the Act, as amended by section 15002 of the 21st Century Cures Act, which establishes the Hospital Readmissions Reduction Program. Under the program, payments for discharges from an applicable hospital as defined under section 1886(d) of the Act will be reduced to account for certain excess readmissions. Section 15002 of the 21st Century Cures Act directs the Secretary to compare hospitals with respect to the number of their Medicare-Medicaid dual-eligible beneficiaries in determining the extent of excess readmissions.
- Section 1886(r) of the Act, as added by section 3133 of the Affordable Care Act, which provides for a reduction to disproportionate share hospital (DSH) payments under section 1886(d)(5)(F) of the Act and for an additional uncompensated care payment to eligible hospitals. Specifically, section 1886(r) of the Act requires that, for fiscal year 2014 and each subsequent fiscal year, subsection (d) hospitals that would otherwise receive a DSH payment made under section 1886(d)(5)(F) of the Act will receive two separate payments: (1) 25 percent of the amount they previously would have received under the statutory formula for Medicare DSH payments in section 1886(d)(5)(F) of the Act if subsection (r) did not apply (“the empirically justified amount”), and (2) an additional payment for the DSH hospital's proportion of uncompensated care, determined as the product of three factors. These three factors are: (1) 75 percent of the payments that would otherwise be made under section 1886(d)(5)(F) of the Act, in the absence of section 1886(r) of the Act; (2) 1 minus the percent change in the percent of individuals who are uninsured; and (3) the hospital's uncompensated care amount relative to the uncompensated care amount of all DSH hospitals expressed as a percentage.
- Section 1886(m)(5) of the Act, which requires the Secretary to reduce by 2 percentage points the annual update to the standard Federal rate for discharges for a long-term care hospital (LTCH) during the rate year for LTCHs that do not submit data on quality measures in the form, manner, and at a time, specified by the Secretary.
- Section 1886(m)(6) of the Act, as added by section 1206(a)(1) of the Pathway for Sustainable Growth Rate (SGR) Reform Act of 2013 ( Pub. L. 113-67 ) and amended by section 51005(a) of the Bipartisan Budget Act of 2018 ( Pub. L. 115-123 ), which provided for the establishment of site neutral payment rate criteria under the LTCH PPS, with implementation beginning in FY 2016. Section 51005(b) of the Bipartisan Budget Act of 2018 amended section 1886(m)(6)(B) by adding new clause (iv), which specifies that the IPPS comparable amount defined in clause (ii)(I) shall be reduced by 4.6 percent for FYs 2018 through 2026.
- Section 1899B of the Act, which provides for the establishment of standardized data reporting for certain post-acute care providers, including LTCHs.
- Section 1115A of the Act authorizes the testing of innovative payment and service delivery models that preserve or enhance the quality of care furnished to Medicare, Medicaid, and Children's Health Insurance Program (CHIP) beneficiaries while reducing program expenditures.
- Sections 1866 and 1902 of the Act, which requires providers of services seeking to participate in the Medicare or Medicaid program, or both, to enter into an agreement with the Secretary or the state Medicaid agency, as appropriate. Hospitals (all hospitals to which the requirements of 42 CFR part 482 apply, including short-term acute care hospitals, LTC hospitals, rehabilitation hospitals, psychiatric hospitals, cancer hospitals, and children's hospitals) and critical access hospitals (CAHs) seeking to be Medicare and Medicaid providers of services under 42 CFR part 485, subpart F , must be certified as meeting Federal participation requirements (conditions of participation (CoPs) and conditions for coverage (CfCs)). Section 1861(e) of the Act provides the patient health and safety protections established by the Secretary for hospital CoPs. Section 1820(e) of the Act provides similar authority for CAHs.
The following is a summary of the major provisions in this final rule. In ( print page 68989) general, these major provisions are being finalized as part of the annual update to the payment policies and payment rates, consistent with the applicable statutory provisions. A general summary of the changes in this final rule is presented in section I.D. of the preamble of this final rule.
To help mitigate growing wage index disparities between high wage and low wage hospitals, in the FY 2020 IPPS/LTCH PPS rule ( 84 FR 42326 through 42332 ), we adopted a policy to increase the wage index values for certain hospitals with low wage index values (the low wage index hospital policy). This policy was adopted in a budget neutral manner through an adjustment applied to the standardized amounts for all hospitals. We indicated our intention that this policy would be effective for at least 4 years, beginning in FY 2020, in order to allow employee compensation increases implemented by these hospitals sufficient time to be reflected in the wage index calculation. As discussed in section III.G.5. of the preamble of this final rule, while we are using the FY 2021 cost report data for the FY 2025 wage index, we are unable to comprehensively evaluate the effect, if any, the low wage index hospital policy had on hospitals' wage increases during the years the COVID-19 public health emergency (PHE) was in effect. We believe it is necessary to wait until we have useable data from fiscal years after the PHE before reaching any conclusions about the efficacy of the policy. Therefore, after consideration of public comments, we are finalizing our proposal that the low wage index hospital policy and the related budget neutrality adjustment would be effective for at least 3 more years, beginning in FY 2025.
As discussed in section V.J. of the preamble of this final rule, the Biden-Harris administration has made it a priority to strengthen the resilience of medical supply chains and support reliable access to products for public health, including through prevention and mitigation of medical product shortages. As a first step in this initiative, we proposed to establish a separate payment for small, independent hospitals for the IPPS shares of the additional resource costs to voluntarily establish and maintain a 6-month buffer stock of one or more of 86 essential medicines, either directly or through contractual arrangements with a pharmaceutical manufacturer, distributor, or intermediary. For the purposes of this policy, eligibility is limited to small, independent hospitals as hospitals with 100 beds or fewer that are not part of a chain organization. We are finalizing our proposal to make this separate payment in a non-budget neutral manner under section 1886(d)(5)(I) of the Act. We are also finalizing our proposal that the payment adjustments would commence for cost reporting periods beginning on or after October 1, 2024.
Under section 1886(r) of the Act, which was added by section 3133 of the Affordable Care Act, starting in FY 2014, Medicare disproportionate share hospitals (DSHs) receive 25 percent of the amount they previously would have received under the statutory formula for Medicare DSH payments in section 1886(d)(5)(F) of the Act. The remaining amount, equal to 75 percent of the amount that would have been paid as Medicare DSH payments under section 1886(d)(5)(F) of the Act if subsection (r) did not apply, is paid as additional payments after the amount is reduced for changes in the percentage of individuals that are uninsured. Each Medicare DSH that has uncompensated care will receive an additional payment based on its share of the total amount of uncompensated care for all Medicare DSHs for a given time period. This additional payment is known as the uncompensated care payment.
In this final rule, we are finalizing the proposed update to our estimates of the three factors used to determine uncompensated care payments for FY 2025. We also proposed to continue to use uninsured estimates produced by CMS' Office of the Actuary (OACT) as part of the development of the National Health Expenditure Accounts (NHEA) in conjunction with more recently available data in the calculation of Factor 2, and we are finalizing this approach. Consistent with the regulation at § 412.106(g)(1)(iii)(C)( 11 ), which was adopted in the FY 2023 IPPS/LTCH PPS final rule, for FY 2025, we will use the 3 most recent years of audited data on uncompensated care costs from Worksheet S-10 of the FY 2019, FY 2020, and FY 2021 cost reports to calculate Factor 3 in the uncompensated care payment methodology for all eligible hospitals.
Beginning with FY 2023 ( 87 FR 49047 through 49051 ), we also established a supplemental payment for IHS and Tribal hospitals and hospitals located in Puerto Rico. In section IV.D. of the preamble of this final rule, we summarized the ongoing methodology for supplemental payments.
In this final rule, we are finalizing our proposal to calculate the per-discharge amount for interim uncompensated care payments for FY 2025 and subsequent fiscal years with modification. Specifically, for FY 2025, we will calculate the per-discharge amount for interim uncompensated care payments using the average of the most recent 2 years of discharge data. In light of the commenters' concerns regarding a trend of decreasing discharge volume and possible overestimation of discharges in recent years, we believe that, on balance, omitting FY 2021 data from the calculation of interim uncompensated care payments is likely to more accurately estimate FY 2025 discharges. Therefore, we are finalizing our proposal with modification. We are modifying the text of § 412.106(i)(1) to state that for FY 2025, interim uncompensated care payments will be calculated based on an average of the most recent 2 years of available historical discharge data, and, consistent with the proposed rule,, interim uncompensated care payments for FY 2026 and subsequent fiscal years will be calculated based on an average of the most recent 3 years of available historical discharge data.
The Patient Safety Structural measure is an attestation-based measure that assesses whether hospitals have a structure and culture that prioritizes safety as demonstrated by the following five domains: (1) leadership commitment to eliminating preventable harm; (2) strategic planning and organizational policy; (3) culture of safety and learning health system; (4) accountability and transparency; and (5) patient and family engagement. Hospitals will attest to whether they engage in specific evidence-based best practices within each of these domains to achieve a score from zero to five out of five points. We proposed that hospitals would be required to report this measure beginning with the CY 2025 reporting period/FY 2027 program year for the PCHQR Program and for the CY 2025 reporting period/FY 2027 payment determination for the Hospital IQR Program. We are finalizing this proposal, with a modification to one of the domains. ( print page 68990)
The updated version of the HCAHPS Survey measure aligns with the National Quality Strategy goal to bring patient voices to the forefront by incorporating feedback from patients and caregivers. We proposed that the updated HCAHPS Survey measure would be adopted for the Hospital IQR and PCHQR Programs beginning with the CY 2025 reporting period/FY 2027 payment determination and the CY 2025 reporting period/FY 2027 program year, respectively. For the Hospital VBP Program, we proposed to modify scoring on the Person and Community Engagement Domain for the FY 2027 through FY 2029 program years to only score the six dimensions of the HCAHPS Survey measure that would remain unchanged from the current version of the survey. We proposed to adopt the updated HCAHPS Survey measure beginning with the FY 2030 program year, which would result in nine HCAHPS Survey measure dimensions for the Person and Community Engagement Domain. We also proposed to modify scoring of the Person and Community Engagement Domain beginning with the FY 2030 program year to account for the proposed updates to the HCAHPS Survey measure. We are finalizing all of these proposals.
Section 1886(o) of the Act requires the Secretary to establish a Hospital VBP Program under which value-based incentive payments are made in a fiscal year to hospitals based on their performance on measures established for a performance period for such fiscal year. We proposed to modify scoring on the Person and Community Engagement Domain for the FY 2027 through FY 2029 program years while the updated HCAHPS Survey measure would be publicly reported under the Hospital IQR Program. In addition, we proposed to adopt the updated HCAHPS Survey measure beginning with the FY 2030 program year and modify scoring beginning with the FY 2030 program year to account for the updated HCAHPS Survey measure. We are finalizing these proposals.
Under section 1886(b)(3)(B)(viii) of the Act, subsection (d) hospitals are required to report data on measures selected by the Secretary for a fiscal year in order to receive the full annual percentage increase. In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed several changes to the Hospital IQR Program. We proposed the adoption of seven new measures: (1) Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 payment determination; (2) Age Friendly Hospital measure beginning with the CY 2025 reporting period/FY 2027 payment determination; (3) Catheter-Associated Urinary Tract Infection (CAUTI) Standardized Infection Ratio Stratified for Oncology Locations beginning with the CY 2026 reporting period/FY 2028 payment determination; (4) Central Line-Associated Bloodstream Infection (CLABSI) Standardized Infection Ratio Stratified for Oncology Locations beginning with the CY 2026 reporting period/FY 2028 payment determination; (5) Hospital Harm—Falls with Injury eCQM beginning with the CY 2026 reporting period/FY 2028 payment determination; (6) Hospital Harm—Postoperative Respiratory Failure eCQM beginning with the CY 2026 reporting period/FY 2028 payment determination; and (7) Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) measure beginning with the July 1, 2023-June 30, 2025 reporting period/FY 2027 payment determination. We also proposed refinements to two measures currently in the Hospital IQR Program measure set: (1) Global Malnutrition Composite Score (GMCS) eCQM, beginning with the CY 2026 reporting period/FY 2028 payment determination; and (2) the HCAHPS Survey beginning with the CY 2025 reporting period/FY 2027 payment determination. In addition, we proposed the removal of five measures: (1) Death Among Surgical Inpatients with Serious Treatable Complications (CMS PSI 04) measure beginning with the July 1, 2023-June 30, 2025 reporting period/FY 27 payment determination; (2) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Acute Myocardial Infarction (AMI) measure beginning with the July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination; (3) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Heart Failure (HF) measure beginning with the July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination; (4) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Pneumonia (PN) measure beginning with July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination, and (5) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Elective Primary Total Hip Arthroplasty (THA) and/or Total Knee Arthroplasty (TKA) measure beginning with the April 1, 2021-March 31, 2024 reporting period/FY 2026 payment determination. We are finalizing all of these proposals as proposed with the exception of the Patient Safety Structural measure, which we are finalizing with modifications.
Lastly, we proposed to modify eCQM data reporting and submission requirements by proposing a progressive increase in the number of mandatory eCQMs a hospital would be required to report on beginning with the CY 2026 reporting period/FY 2028 payment determination. We also proposed two changes to current policies related to validation of hospital data: (1) to implement eCQM validation scoring based on the accuracy of eCQM data beginning with the validation of CY 2025 eCQM data affecting the FY 2028 payment determination; and (2) modification of the data validation reconsideration request requirements to make medical records submission optional for reconsideration requests beginning with CY 2023 discharges/FY 2026 payment determination. We are finalizing all of these proposals as proposed with the exception of the proposed progressive increase in the number of mandatory eCQMs, which we are finalizing with modifications.
Section 1866(k)(1) of the Act requires, for purposes of FY 2014 and each subsequent fiscal year, that a hospital described in section 1886(d)(1)(B)(v) of the Act (a PPS-exempt cancer hospital, or a PCH) submit data in accordance with section 1866(k)(2) of the Act with respect to such fiscal year. In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed the following:
- To adopt the Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 program year.
- To modify the HCAHPS Survey measure beginning with the CY 2025 reporting period/FY 2027 program year.
- To move up the start date for publicly displaying hospital performance on the Hospital Commitment to Health Equity measure from July 2026 to January 2026 or as soon as feasible thereafter.
We are finalizing all of these proposals as proposed with the ( print page 68991) exception of the Patient Safety Structural measure, which we are finalizing with modifications.
We proposed and are finalizing the following changes to the LTCH QRP: (1) add four assessment items to the LCDS beginning with the FY 2028 LTCH QRP; (2) modify one item on the LCDS beginning with the FY 2028 LTCH QRP; and (3) extend the admission assessment window for the LCDS from 3 days to 4 days beginning with the FY 2028 LTCH QRP. We also summarize the feedback we received on requests for information in the proposed rule on future measure concepts for the LTCH QRP and a future LTCH Star Rating system.
In section X.F. of the preamble of the proposed rule, we proposed several changes to the Medicare Promoting Interoperability Program. Specifically, we proposed: (1) to separate the Antimicrobial Use and Resistance (AUR) Surveillance measure into two measures, an Antimicrobial Use (AU) Surveillance measure and an Antimicrobial Resistance (AR) Surveillance measure, beginning with the EHR reporting period in CY 2025; to add a new exclusion for eligible hospitals or critical access hospitals (CAHs) that do not have a data source containing the minimal discrete data elements that are required for AU or AR Surveillance reporting; to modify the existing exclusions for the AUR Surveillance measure to apply to the proposed AU Surveillance and AR Surveillance measures, respectively; and to treat the AU Surveillance and AR Surveillance measures as new measures with respect to active engagement beginning with the EHR reporting period in CY 2025; (2) to increase the performance-based scoring threshold for eligible hospitals and CAHs reporting under the Medicare Promoting Interoperability Program from 60 points to 80 points beginning with the EHR reporting period in CY 2025; (3) to adopt two new eCQMs that hospitals can select as one of their three self-selected eCQMs beginning with the CY 2026 reporting period: the Hospital Harm—Falls with Injury eCQM and the Hospital Harm—Postoperative Respiratory Failure eCQM; (4) beginning with the CY 2026 reporting period, to modify one eCQM, the Global Malnutrition Composite Score eCQM; and (5) to modify eCQM data reporting and submission requirements by proposing a progressive increase in the number of mandatory eCQMs eligible hospitals and CAHs would be required to report on beginning with the CY 2026 reporting period. We are finalizing all proposals as proposed, with the exception of our proposals to increase the performance-based scoring threshold for eligible hospitals and CAHs, and to progressively increase the number of mandatory eCQMs required for reporting, which we are finalizing with modification. We are finalizing, with modification, an increase to the performance-based scoring threshold for eligible hospitals and CAHs from 60 points to 70 points for the EHR reporting period in CY 2025 and from 70 points to 80 points beginning with the EHR reporting period in CY 2026, and finalizing, with modification, the regulatory text accordingly. We are also finalizing, with modification, our proposal to increase the eCQM reporting requirements in the Medicare Promoting Interoperability Program for the CY 2026, CY 2027, CY 2028, and subsequent years' reporting periods. Specifically, eligible hospitals and CAHs will be required to report a total of eight eCQMs for the CY 2026 reporting period, a total of nine eCQMs for the CY 2027 reporting period, and a total of eleven eCQMs beginning with the CY 2028 reporting period.
In the proposed rule, we included a proposal to implement section 4122 of the CAA, 2023. Section 4122(a) of the CAA, 2023, amended section 1886(h) of the Act by adding a new section 1886(h)(10) of the Act requiring the distribution of additional residency positions (also referred to as slots) to hospitals. After consideration of public comments, we are finalizing this proposal, with minor modifications. We refer readers to section V.F.2. of the preamble of this final rule for a summary of the provisions of section 4122 of the CAA, 2023 that we are implementing in this final rule.
The Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, extended the MDH program and the temporary changes to the low-volume hospital qualifying criteria and payment adjustment under the IPPS for a portion of FY 2025. Specifically, section 306 of the CAA, 2024, further extended the modified definition of low-volume hospital and the methodology for calculating the payment adjustment for low-volume hospitals under section 1886(d)(12) of the Act through December 31, 2024. Section 307 of the CAA, 2024, extended the MDH program under section 1886(d)(5)(G) of the Act through December 31, 2024. Prior to enactment of the CAA, 2024, the low-volume hospital qualifying criteria and payment adjustment were set revert to the statutory requirements that were in effect prior to FY 2011 at the end of FY 2024 and beginning October 1, 2024, the MDH program would have no longer been in effect.
We recognize the importance of these extensions with respect to the goal of advancing health equity by addressing the health disparities that underlie the health system is one of CMS' strategic pillars [ 1 ] and a Biden-Harris Administration priority. [ 2 ] These provisions are projected to increase payments to IPPS hospitals by approximately $137 million in FY 2025.
As discussed in section X.A. of the preamble of this final rule, we are finalizing the Transforming Episode Accountability Model (TEAM). TEAM will be a 5-year mandatory model tested under the authority of section 1115A of the Act, beginning on January 1, 2026, and ending on December 31, 2030. The intent of TEAM is to improve beneficiary care through financial accountability for episode categories that begin with one of the following procedures: coronary artery bypass graft surgery (CABG), lower extremity joint replacement (LEJR), major bowel procedure, surgical hip/femur fracture treatment (SHFFT), and spinal fusion. TEAM will test whether financial accountability for these episode categories reduces Medicare expenditures while preserving or enhancing the quality of care for Medicare beneficiaries.
Under Traditional Medicare, Medicare makes separate payments to providers and suppliers for the items and services furnished to a beneficiary over the course of an episode of care. Because providers and suppliers are paid for each individual item or service delivered, providers may not be incentivized to invest in quality improvement and care coordination ( print page 68992) activities. As a result, care may be fragmented, unnecessary, or duplicative. By holding hospitals accountable for all items and services provided during an episode, providers would be better incentivized to coordinate patient care, avoid duplicative or unnecessary services, and improve the beneficiary care experience during care transitions.
Under TEAM, all acute care hospitals, with limited exceptions, located within the mandatory Core-Based Statistical Areas (CBSAs) that CMS selected for model implementation will be required to participate in TEAM. CMS will allow a one-time opportunity for hospitals that participate until the last day of the last performance period in the BPCI Advanced model or the last day of the last performance year of the CJR model, that are not located in a mandatory CBSA selected for TEAM participation to voluntarily opt into TEAM. [ 3 ] TEAM will have a 1-year glide path opportunity for all TEAM participants and a 3-year glide path opportunity for TEAM participants that are safety net hospitals, which will allow TEAM participants to ease into full financial risk. Episodes will include non-excluded Medicare Parts A and B items and services and would begin with an anchor hospitalization or anchor procedure and will end 30 days after hospital discharge. The following episode categories, when furnished by a TEAM participant, will initiate an episode in TEAM: lower extremity joint replacement, surgical hip femur fracture treatment, spinal fusion, coronary artery bypass graft, and major bowel procedure.
TEAM participants will continue to bill Medicare FFS as usual but will receive target prices for episodes prior to each performance year. Target prices will be based on 3 years of baseline data, prospectively trended forward to the relevant performance year, and calculated at the level of MS-DRG/HCPCS episode type and region. Target prices will also include a discount factor, normalization factor, retrospective trend adjustment factor, and beneficiary and provider level risk-adjustment. Performance in the model will be assessed by comparing TEAM participants' actual Medicare FFS spending during a performance year to their reconciliation target price as well as by performance on three quality measures. TEAM participants will earn a payment from CMS, subject to a quality performance adjustment, if their spending is below the reconciliation target price. TEAM participants will owe CMS a repayment amount, subject to a quality performance adjustment, if their spending is above the reconciliation target price. In section X.A. of the preamble of this final rule some policies as proposed, and we are finalizing others with modification. There are also certain proposed policies that we are not finalizing, and we will instead go through rulemaking in the future to promulgate new policies before the model start date.
In alignment with the Biden-Harris Administration's commitment to addressing the maternal health crisis, this RFI sought to gather information on differences between hospital resources required to provide inpatient pregnancy and childbirth services to Medicare patients as compared to non-Medicare patients. To the extent that the resources required differ between patient populations, we also wanted to gather information on the extent to which non-Medicare payers, or other commercial insurers may be using the IPPS as a basis for determining their payment rates for inpatient pregnancy and childbirth services and the effect, if any, that the use of the IPPS as a basis for determining payment by those payers may have on maternal health outcomes. We summarize the comments received in section X.C. of the preamble of this final rule.
In section X.F. of the preamble of the proposed rule, we proposed to update the hospital and CAH infection prevention and control and antibiotic stewardship programs conditions of participation (CoPs) to extend a limited subset of the current COVID-19 and influenza data reporting requirements. These proposed reporting requirements ensure that hospitals and CAHs have appropriate insight related to evolving infection control needs. Specifically, we proposed to replace the COVID-19 and Seasonal Influenza reporting standards for hospitals and CAHs with a new standard addressing acute respiratory illnesses to require that, beginning on October 1, 2024, hospitals and CAHs would have to electronically report information about COVID-19, influenza, and RSV. We also proposed that outside of a public health emergency (PHE), hospitals and CAHs would have to report these data on a weekly basis. In section X.F. of the preamble of this final rule, we are finalizing these proposals with revisions.
As discussed in section II.C. of the preamble of this final rule, we are finalizing the proposed change to the severity level designation for the social determinants of health (SDOH) diagnosis codes describing inadequate housing and housing instability from non-complication or comorbidity (NonCC) to complication or comorbidity (CC) for FY 2025. Consistent with our annual updates to account for changes in resource consumption, treatment patterns, and the clinical characteristics of patients, we recognize inadequate housing and housing instability as indicators of increased resource utilization in the acute inpatient hospital setting.
Consistent with the Administration's goal of advancing health equity for all, including members of historically underserved and under-resourced communities, as described in the President's January 20, 2021 Executive Order 13985 on “Advancing Racial Equity and Support for Underserved Communities Through the Federal Government,” [ [1] ] we also continue to be interested in receiving feedback on how we might further foster the documentation and reporting of the diagnosis codes describing social and economic circumstances to more accurately reflect each health care encounter and improve the reliability and validity of the coded data including in support of efforts to advance health equity.
The following table provides a summary of the costs, transfers, savings, and benefits associated with the major provisions described in section I.A.2. of the preamble of this final rule.

Section 1886(d) of the Act sets forth a system of payment for the operating costs of acute care hospital inpatient stays under Medicare Part A (Hospital Insurance) based on prospectively set rates. Section 1886(g) of the Act requires the Secretary to use a prospective payment system (PPS) to pay for the capital-related costs of inpatient hospital services for these “subsection (d) hospitals.” Under these PPSs, Medicare payment for hospital inpatient operating and capital-related costs is made at predetermined, specific rates for each hospital discharge. Discharges are classified according to a list of diagnosis-related groups (DRGs).
The base payment rate is comprised of a standardized amount that is divided into a labor-related share and a nonlabor-related share. The labor-related share is adjusted by the wage index applicable to the area where the hospital is located. If the hospital is located in Alaska or Hawaii, the nonlabor-related share is adjusted by a cost-of-living adjustment factor. This base payment rate is multiplied by the DRG relative weight.
If the hospital treats a high percentage of certain low-income patients, it receives a percentage add-on payment applied to the DRG-adjusted base payment rate. This add-on payment, known as the disproportionate share hospital (DSH) adjustment, provides for a percentage increase in Medicare payments to hospitals that qualify under either of two statutory formulas designed to identify hospitals that serve a disproportionate share of low-income patients. For qualifying hospitals, the amount of this adjustment varies based on the outcome of the statutory calculations. The Affordable Care Act revised the Medicare DSH payment methodology and provides for an additional Medicare payment beginning on October 1, 2013, that considers the amount of uncompensated care furnished by the hospital relative to all other qualifying hospitals.
If the hospital is training residents in an approved residency program(s), it receives a percentage add-on payment for each case paid under the IPPS, known as the indirect medical education (IME) adjustment. This percentage varies, depending on the ratio of residents to beds.
Additional payments may be made for cases that involve new technologies or medical services that have been approved for special add-on payments. In general, to qualify, a new technology or medical service must demonstrate that it is a substantial clinical improvement over technologies or services otherwise available, and that, absent an add-on payment, it would be inadequately paid under the regular DRG payment. In addition, certain transformative new devices and certain antimicrobial products may qualify under an alternative inpatient new technology add-on payment pathway by demonstrating that, absent an add-on payment, they would be inadequately paid under the regular DRG payment.
The costs incurred by the hospital for a case are evaluated to determine whether the hospital is eligible for an additional payment as an outlier case. This additional payment is designed to protect the hospital from large financial losses due to unusually expensive cases. Any eligible outlier payment is added to the DRG-adjusted base payment rate, plus any DSH, IME, and new technology or medical service add-on adjustments and, beginning in FY 2023 for IHS and Tribal hospitals and hospitals located in Puerto Rico, the new supplemental payment.
Although payments to most hospitals under the IPPS are made on the basis of the standardized amounts, some categories of hospitals are paid in whole or in part based on their hospital-specific rate, which is determined from their costs in a base year. For example, sole community hospitals (SCHs) receive the higher of a hospital-specific rate based on their costs in a base year (the highest of FY 1982, FY 1987, FY 1996, or FY 2006) or the IPPS Federal rate based on the standardized amount. SCHs are the sole source of care in their areas. Specifically, section 1886(d)(5)(D)(iii) of the Act defines an SCH as a hospital that is located more than 35 road miles from another hospital or that, by reason of factors such as an isolated location, weather conditions, travel conditions, or absence of other like hospitals (as determined by the Secretary), is the sole source of hospital inpatient services reasonably available to Medicare beneficiaries. In addition, certain rural hospitals previously designated by the Secretary as essential access community hospitals are considered SCHs.
With the recent enactment of section 307 of the CAA, 2024, under current law, the Medicare-dependent, small rural hospital (MDH) program is effective through December 31, 2024. For discharges occurring on or after October 1, 2007, but before January 1, 2025, an MDH receives the higher of the Federal rate or the Federal rate plus 75 percent of the amount by which the Federal rate is exceeded by the highest of its FY 1982, FY 1987, or FY 2002 hospital-specific rate. MDHs are a major source of care for Medicare beneficiaries in their areas. Section 1886(d)(5)(G)(iv) of the Act defines an MDH as a hospital that is located in a rural area (or, as amended by the Bipartisan Budget Act of 2018, a hospital located in a State with no rural area that meets certain statutory criteria), has not more than 100 beds, is not an SCH, and has a high percentage of Medicare discharges (not less than 60 percent of its inpatient days or discharges in its cost reporting year beginning in FY 1987 or in two of its three most recently settled Medicare cost reporting years). As section 307 of the CAA, 2024, extended the MDH program through the first quarter of FY 2025 only, beginning on January 1, 2025, the MDH program will no longer be in effect absent a change in law. Because the MDH program is not authorized by statute beyond December 31, 2024, beginning January 1, 2025, all hospitals that previously qualified for MDH status under section 1886(d)(5)(G) of the Act will no longer have MDH status and will be paid based on the IPPS Federal rate.
Section 1886(g) of the Act requires the Secretary to pay for the capital-related costs of inpatient hospital services in accordance with a prospective payment system established by the Secretary. The basic methodology for determining capital prospective payments is set forth in our regulations at 42 CFR 412.308 and 412.312 . Under the capital IPPS, payments are adjusted by the same DRG for the case as they are under the operating IPPS. Capital IPPS payments are also adjusted for IME and DSH, similar to the adjustments made under the operating IPPS. In addition, hospitals may receive outlier payments for those cases that have unusually high costs.
The existing regulations governing payments to hospitals under the IPPS are located in 42 CFR part 412, subparts A through M .
Under section 1886(d)(1)(B) of the Act, as amended, certain hospitals and hospital units are excluded from the IPPS. These hospitals and units are: Inpatient rehabilitation facility (IRF) hospitals and units; long-term care hospitals (LTCHs); psychiatric hospitals and units; children's hospitals; cancer hospitals; extended neoplastic disease care hospitals, and hospitals located outside the 50 States, the District of Columbia, and Puerto Rico (that is, hospitals located in the U.S. Virgin ( print page 68996) Islands, Guam, the Northern Mariana Islands, and American Samoa). Religious nonmedical health care institutions (RNHCIs) are also excluded from the IPPS. Various sections of the Balanced Budget Act of 1997 (BBA) ( Pub. L. 105-33 ), the Medicare, Medicaid and SCHIP [State Children's Health Insurance Program] Balanced Budget Refinement Act of 1999 (BBRA, Pub. L. 106-113 ), and the Medicare, Medicaid, and SCHIP Benefits Improvement and Protection Act of 2000 (BIPA, Pub. L. 106-554 ) provide for the implementation of PPSs for IRF hospitals and units, LTCHs, and psychiatric hospitals and units (referred to as inpatient psychiatric facilities (IPFs)). (We note that the annual updates to the LTCH PPS are included along with the IPPS annual update in this document. Updates to the IRF PPS and IPF PPS are issued as separate documents.) Children's hospitals, cancer hospitals, hospitals located outside the 50 States, the District of Columbia, and Puerto Rico (that is, hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa), and RNHCIs continue to be paid solely under a reasonable cost-based system, subject to a rate-of-increase ceiling on inpatient operating costs. Similarly, extended neoplastic disease care hospitals are paid on a reasonable cost basis, subject to a rate-of-increase ceiling on inpatient operating costs.
The existing regulations governing payments to excluded hospitals and hospital units are located in 42 CFR parts 412 and 413 .
The Medicare prospective payment system (PPS) for LTCHs applies to hospitals described in section 1886(d)(1)(B)(iv) of the Act, effective for cost reporting periods beginning on or after October 1, 2002. The LTCH PPS was established under the authority of sections 123 of the BBRA and section 307(b) of the BIPA (as codified under section 1886(m)(1) of the Act). Section 1206(a) of the Pathway for SGR Reform Act of 2013 ( Pub. L. 113-67 ) established the site neutral payment rate under the LTCH PPS, which made the LTCH PPS a dual rate payment system beginning in FY 2016. Under this statute, effective for LTCH's cost reporting periods beginning in FY 2016 cost reporting period, LTCHs are generally paid for discharges at the site neutral payment rate unless the discharge meets the patient criteria for payment at the LTCH PPS standard Federal payment rate. The existing regulations governing payment under the LTCH PPS are located in 42 CFR part 412, subpart O . Beginning October 1, 2009, we issue the annual updates to the LTCH PPS in the same documents that update the IPPS.
Under sections 1814(l), 1820, and 1834(g) of the Act, payments made to critical access hospitals (CAHs) (that is, rural hospitals or facilities that meet certain statutory requirements) for inpatient and outpatient services are generally based on 101 percent of reasonable cost. Reasonable cost is determined under the provisions of section 1861(v) of the Act and existing regulations under 42 CFR part 413 .
Under section 1886(a)(4) of the Act, costs of approved educational activities are excluded from the operating costs of inpatient hospital services. Hospitals with approved graduate medical education (GME) programs are paid for the direct costs of GME in accordance with section 1886(h) of the Act. The amount of payment for direct GME costs for a cost reporting period is based on the hospital's number of residents in that period and the hospital's costs per resident in a base year. The existing regulations governing payments to the various types of hospitals are located in 42 CFR part 413 . Section 1886(d)(5)(B) of the Act provides that prospective payment hospitals that have residents in an approved GME program receive an additional payment for each Medicare discharge to reflect the higher patient care costs of teaching hospitals relative to non-teaching hospitals. The additional payment is based on the indirect medical education (IME) adjustment factor, which is calculated using a hospital's ratio of residents to beds and a multiplier, which is set by Congress. Section 1886(d)(5)(B)(ii)(XII) of the Act provides that, for discharges occurring during FY 2008 and fiscal years thereafter, the IME formula multiplier is 1.35. The regulations regarding the indirect medical education (IME) adjustment are located at 42 CFR 412.105 .
Section 4122 of the CAA, 2023, amended section 1886(h) of the Act by adding a new section 1886(h)(10) of the Act requiring the distribution of additional residency positions (also referred to as slots) to hospitals. Section 1886(h)(10)(A) of the Act requires that for FY 2026, the Secretary shall initiate an application round to distribute 200 residency positions. At least 100 of the positions made available under section 1886(h)(10)(A) of the Act shall be distributed for psychiatry or psychiatry subspecialty residency training programs. The Secretary is required, subject to certain provisions in the law, to increase the otherwise applicable resident limit for each qualifying hospital that submits a timely application by the number of positions that may be approved by the Secretary for that hospital. The Secretary is required to notify hospitals of the number of positions distributed to them by January 31, 2026, and the increase is effective beginning July 1, 2026.
In determining the qualifying hospitals for which an increase is provided, section 1886(h)(10)(B)(i) of the Act requires the Secretary to take into account the “demonstrated likelihood” of the hospital filling the positions made available within the first 5 training years beginning after the date the increase would be effective, as determined by the Secretary.
Section 1886(h)(10)(B)(ii) of the Act requires a minimum distribution for certain categories of hospitals. Specifically, the Secretary is required to distribute at least 10 percent of the aggregate number of total residency positions available to each of four categories of hospitals. Stated briefly, and discussed in greater detail later in this final rule, the categories are as follows: (1) hospitals located in rural areas or that are treated as being located in a rural area (pursuant to sections 1886(d)(2)(D) and 1886(d)(8)(E) of the Act); (2) hospitals in which the reference resident level of the hospital is greater than the otherwise applicable resident limit; (3) hospitals in States with new medical schools or additional locations and branches of existing medical schools; and (4) hospitals that serve areas designated as Health Professional Shortage Areas (HPSAs). Section 1886(h)(10)(F)(iii) of the Act defines a qualifying hospital as a hospital in one of these four categories.
Section 1886(h)(10)(B)(iii) of the Act further requires that each qualifying hospital that submits a timely application receive at least 1 (or a fraction of 1) of the residency positions made available under section 1886(h)(10) of the Act before any qualifying hospital receives more than 1 residency position.
Section 1886(h)(10)(C) of the Act places certain limitations on the distribution of the residency positions. ( print page 68997) First, a hospital may not receive more than 10 additional full-time equivalent (FTE) residency positions. Second, no increase in the otherwise applicable resident limit of a hospital may be made unless the hospital agrees to increase the total number of FTE residency positions under the approved medical residency training program of the hospital by the number of positions made available to that hospital. Third, if a hospital that receives an increase to its otherwise applicable resident limit under section 1886(h)(10) of the Act is eligible for an increase to its otherwise applicable resident limit under 42 CFR 413.79(e)(3) (or any successor regulation), that hospital must ensure that residency positions received under section 1886(h)(10) of the Act are used to expand an existing residency training program and not for participation in a new residency training program.
Section 306 of the CAA, 2024, extended through the first 3 months of FY 2025 the modified definition of a low-volume hospital and the methodology for calculating the payment adjustment for low-volume hospitals in effect for FYs 2019 through 2024. Specifically, under section 1886(d)(12)(C)(i) of the Act, as amended, for FYs 2019 through 2024 and the portion of FY 2025 occurring before January 1, 2025, a subsection (d) hospital qualifies as a low-volume hospital if it is more than 15 road miles from another subsection (d) hospital and has less than 3,800 total discharges during the fiscal year. Under section 1886(d)(12)(D) of the Act, as amended, for discharges occurring in FYs 2019 through December 31, 2024, the Secretary determines the applicable percentage increase using a continuous, linear sliding scale ranging from an additional 25 percent payment adjustment for low-volume hospitals with 500 or fewer discharges to a zero percent additional payment for low-volume hospitals with more than 3,800 discharges in the fiscal year.
Section 307 of the CAA, 2024, amended sections 1886(d)(5)(G)(i) and 1886(d)(5)(G)(ii)(II) of the Act to provide for an extension of the MDH program through the first 3 months of FY 2025 (that is, through December 31, 2024).
The FY 2025 IPPS/LTCH PPS proposed rule appeared in the May 2, 2024, Federal Register ( 89 FR 35934 ). In this proposed rule, we set forth proposed payment and policy changes to the Medicare IPPS for FY 2025 operating costs and capital-related costs of acute care hospitals and certain hospitals and hospital units that are excluded from IPPS. In addition, we set forth proposed changes to the payment rates, factors, and other payment and policy-related changes to programs associated with payment rate policies under the LTCH PPS for FY 2025.
The following is a general summary of the changes that we proposed to make:
In section II. of the preamble of the proposed rule, we included the following:
- Proposed changes to MS-DRG classifications based on our yearly review for FY 2025.
- Proposed recalibration of the MS-DRG relative weights.
- A discussion of the proposed FY 2025 status of new technologies approved for add-on payments for FY 2024, a presentation of our evaluation and analysis of the FY 2025 applicants for add-on payments for high-cost new medical services and technologies (including public input, as directed by the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) Public Law 108-173 , obtained in a town hall meeting for applications not submitted under an alternative pathway), and a discussion of the proposed status of FY 2025 new technology applicants under the alternative pathways for certain medical devices and certain antimicrobial products.
- A proposed change to the April 1 cutoff to October 1 for determining whether a technology would be within its 2- to 3-year newness period when considering eligibility for new technology add-on payments, beginning in FY 2026, effective for those technologies that are approved for new technology add-on payments starting in FY 2025 or a subsequent year (as discussed in II.E.8. of the preamble of the proposed rule).
- A proposal that, beginning with new technology add-on payment applications for FY 2026, we will no longer consider a hold status to be an inactive status for the purposes of eligibility for the new technology add-on payment (as discussed in section II.E.9. of the preamble of the proposed rule).
- A proposal that, subject to our review of the new technology add-on payment eligibility criteria, for certain gene therapies approved for new technology add-on payments in the FY 2025 IPPS/LTCH final rule that are indicated and used specifically for the treatment of sickle cell disease (SCD), effective with discharges on or after October 1, 2024, and concluding at the end of the 2- to 3-year newness period for such therapy, we would temporarily increase the new technology add-on payment percentage to 75 percent (as discussed in section II.E.10. of the preamble of the proposed rule).
In section III. of the preamble of the proposed rule, we proposed revisions to the wage index for acute care hospitals and the annual update of the wage data. Specific issues addressed include, but are not limited to, the following:
- Proposed changes in core-based statistical areas (CBSAs) as a result of new OMB labor market area delineations and proposed policies related to the proposed changes in CBSAs.
- The proposed FY 2025 wage index update using wage data from cost reporting periods beginning in FY 2019.
- Calculation, analysis, and implementation of the proposed occupational mix adjustment to the wage index for acute care hospitals for FY 2025 based on the 2022 Occupational Mix Survey.
- Proposed application of the rural, imputed and frontier State floors, and continuation of the low wage index hospital policy.
- Proposed revisions to the wage index for acute care hospitals, based on hospital redesignations and reclassifications under sections 1886(d)(8)(B), (d)(8)(E), and (d)(10) of the Act.
- Proposed adjustment to the wage index for acute care hospitals for FY 2025 based on commuting patterns of hospital employees who reside in a county and work in a different area with a higher wage index.
- Proposed labor-related share for the FY 2025 wage index.
In section IV. of the preamble of this proposed rule, we discuss the following:
- Proposed calculation of Factor 1 and Factor 2 of the uncompensated care payment methodology.
- Proposed methodological approach for determining Factor 3 of the uncompensated care payment for FY 2025, which is the same methodology that was used for FY 2024. ( print page 68998)
- Proposed methodological approach for determining the amount of interim uncompensated care payments using the average of the most recent 3 years of discharge data.
In section V. of the preamble of the proposed rule, we discussed proposed changes or clarifications of a number of the provisions of the regulations in 42 CFR parts 412 and 413 , including the following:
- Proposed inpatient hospital update for FY 2025.
- Proposed updated national and regional case-mix values and discharges for purposes of determining RRC status and clarification of the qualification under the discharge criterion for osteopathic hospitals.
- Proposed implementation of the statutory extension of the temporary changes to the low-volume hospital payment adjustment through December 31, 2024, the statutory expiration beginning January 1, 2025, and the proposed payment adjustments for low-volume hospitals for FY 2025.
- Proposed implementation of the statutory extension of the MDH program through December 31, 2024, and the statutory expiration beginning January 1, 2025.
- A proposal to implement a provision of the Consolidated Appropriations Act relating to payments to hospitals for GME and IME costs, proposed direct graduate medical education (DGME) and IME policy modifications to the criteria for new residency programs; technical fixes to the DGME regulations; and a notice of closure of two teaching hospitals and opportunities to apply for available slots and a reminder of CBSA changes and application to GME policies.
- Proposed nursing and allied health education program Medicare Advantage (MA) add-on rates and direct GME MA percent reductions for CY 2023.
- Proposed update to the payment adjustment for certain clinical trial and expanded access use immunotherapy cases.
- Proposed separate IPPS payment for establishing and maintaining access to essential medicines.
- Proposed update to the estimate of the financial impacts for the FY 2025 Hospital Readmissions Reduction Program.
- Proposed modifications to the scoring of the Person and Community Engagement Domain in the Hospital VBP Program.
++ For the FY 2027 through FY 2029 program years to only score on six unchanged dimensions of the HCAHPS Survey.
++ Beginning with the FY 2030 program year to account for the proposed updated HCAHPS Survey.
- Updating the proposed estimate of the financial impacts for the FY 2025 Hospital-Acquired Conditions Reduction Program.
- Discussion of and proposed changes relating to the implementation of the Rural Community Hospital Demonstration Program in FY 2025.
In section VI. of the preamble of the proposed rule, we discussed the proposed payment policy requirements for capital-related costs and capital payments to hospitals for FY 2025.
In section VII. of the preamble of the proposed rule, we discussed the following:
- Proposed changes to payments to certain excluded hospitals for FY 2025.
- Proposed continued implementation of the Frontier Community Health Integration Project (FCHIP) Demonstration.
In section VIII. of the preamble of the proposed rule, we proposed to rebase and revise the LTCH market basket to reflect a 2022 base year, which includes a proposed update to the LTCH PPS labor-related share. In section VIII. of the preamble of the proposed rule, we set forth proposed changes to the LTCH PPS Federal payment rates, factors, and other payment rate policies under the LTCH PPS for FY 2025. We also proposed a technical clarification to the regulations for hospitals seeking to be classified as an LTCH.
In section IX. of the preamble of the proposed rule, we addressed the following:
- Solicitation of comment on adopting measures across the hospital quality reporting and value-based purchasing programs which capture more forms of unplanned post-acute care and encourage hospitals to improve discharge processes.
- Proposed changes to the requirements for the Hospital IQR Program.
- Proposed changes to the requirements for the PCHQR Program.
- Proposed adoption of the Patient Safety Structural measure in the Hospital IQR Program and the PCHQR Program.
- Proposed updated HCAHPS Survey measure in the Hospital IQR Program, PCHQR Program, and Hospital VBP Program.
- Proposed changes to the requirements for the LTCH QRP, and requests for information on future measure concepts for the LTCH QRP and a star rating system for the LTCH QRP.
- Proposed changes to requirements pertaining to eligible hospitals and CAHs participating in the Medicare Promoting Interoperability Program.
Section X. of the preamble of the proposed rule includes the following:
- Proposed implementation of TEAM that would test whether an episode-based pricing methodology linked with accountability for quality measure performance for select acute care hospitals reduces Medicare program expenditures while preserving or improving the quality of care for Medicare beneficiaries.
- Proposed changes to permit a Provider Reimbursement Review Board (PRRB) member to serve up to 3 consecutive terms (9 consecutive years total), and up to 4 consecutive terms (12 consecutive years total) in cases where a PRRB Member who, in their second or third consecutive term, is designated as Chairperson, to continue serving as Chairperson in the fourth consecutive term.
- Solicitation of comments to gather information on differences between hospital resources required to provide inpatient pregnancy and childbirth services to Medicare patients as compared to non-Medicare patients.
- Solicitation of comments to gather information on potential solutions that can be implemented through the hospital CoPs to address well-documented concerns regarding maternal morbidity, mortality, disparities, and maternity care access in the United States. See the calendar year (CY) 2025 Outpatient Prospective Payment System (OPPS) proposed rule (89 FR XXXXX) for more information about this RFI.
- Proposal to remove the exclusion of Puerto Rico from the Payment Error Rate Measurement (PERM) program found at 42 CFR 431.954(b)(3) .
- Proposal for a new hospital CoP to replace the COVID-19 and Seasonal Influenza reporting standards for ( print page 68999) hospitals and CAHs that were created during PHE.
Section XI.A. of the preamble of the proposed rule includes our discussion of the MedPAC Recommendations.
Section XI.B. of the preamble of the proposed rule includes a descriptive listing of the public use files associated with this proposed rule.
Section XII. of the preamble of the proposed rule includes the collection of information requirements for entities based on our proposals.
Section XIII. of the preamble of the proposed rule includes information regarding our responses to public comments.
In sections II. and III. of the Addendum of the proposed rule, we set forth proposed changes to the amounts and factors for determining the proposed FY 2025 prospective payment rates for operating costs and capital-related costs for acute care hospitals. We proposed to establish the threshold amounts for outlier cases. In addition, in section IV. of the Addendum of the proposed rule, we addressed the proposed update factors for determining the rate-of-increase limits for cost reporting periods beginning in FY 2025 for certain hospitals excluded from the IPPS.
In section V. of the Addendum of the proposed rule, we set forth proposed changes to the amounts and factors for determining the proposed FY 2025 LTCH PPS standard Federal payment rate and other factors used to determine LTCH PPS payments under both the LTCH PPS standard Federal payment rate and the site neutral payment rate in FY 2025. We proposed to establish the adjustments for the wage index (including proposed changes to the LTCH PPS labor market area delineations based on the new OMB delineations), labor-related share, the cost-of-living adjustment, and high-cost outliers, including the applicable fixed-loss amounts and the LTCH cost-to-charge ratios (CCRs) for both payment rates.
In Appendix A of the proposed rule, we set forth an analysis of the impact the proposed changes would have on affected acute care hospitals, CAHs, LTCHs and other entities.
In Appendix B of the proposed rule, as required by sections 1886(e)(4) and (e)(5) of the Act, we provided our recommendations of the appropriate percentage changes for FY 2025 for the following:
- A single average standardized amount for all areas for hospital inpatient services paid under the IPPS for operating costs of acute care hospitals (and hospital-specific rates applicable to SCHs and MDHs).
- Target rate-of-increase limits to the allowable operating costs of hospital inpatient services furnished by certain hospitals excluded from the IPPS.
- The LTCH PPS standard Federal payment rate and the site neutral payment rate for hospital inpatient services provided for LTCH PPS discharges.
Under section 1805(b) of the Act, MedPAC is required to submit a report to Congress, no later than March 15 of each year, in which MedPAC reviews and makes recommendations on Medicare payment policies. MedPAC's March 2024 recommendations concerning hospital inpatient payment policies address the update factor for hospital inpatient operating costs and capital-related costs for hospitals under the IPPS. We addressed these recommendations in Appendix B of the proposed rule. For further information relating specifically to the MedPAC March 2024 report or to obtain a copy of the report, contact MedPAC at (202) 220-3700 or visit MedPAC's website at https://www.medpac.gov .
We received approximately 6,180 timely pieces of correspondence containing multiple comments on the proposed rule that appeared in the May 2, 2024 Federal Register ( 89 FR 39534 ) titled “Medicare and Medicaid Programs and the Children's Health Insurance Program; Hospital Inpatient Prospective Payment Systems for Acute Care Hospitals and the Long-Term Care Hospital Prospective Payment System and Policy Changes and Fiscal Year 2025 Rates; Quality Programs Requirements; and Other Policy Changes” (hereinafter referred to as the FY 2025 IPPS/LTCH PPS proposed rule). We note that some of these public comments were outside of the scope of the proposed rule. These out-of-scope public comments are not addressed with policy responses in this final rule. Summaries of the public comments that are within the scope of the proposed rule and our responses to those public comments are set forth in the various sections of this final rule under the appropriate heading.
Section 1886(d) of the Act specifies that the Secretary shall establish a classification system (referred to as diagnosis-related groups (DRGs)) for inpatient discharges and adjust payments under the IPPS based on appropriate weighting factors assigned to each DRG. Therefore, under the IPPS, Medicare pays for inpatient hospital services on a rate per discharge basis that varies according to the DRG to which a beneficiary's stay is assigned. The formula used to calculate payment for a specific case multiplies an individual hospital's payment rate per case by the weight of the DRG to which the case is assigned. Each DRG weight represents the average resources required to care for cases in that particular DRG, relative to the average resources used to treat cases in all DRGs.
Section 1886(d)(4)(C) of the Act requires that the Secretary adjust the DRG classifications and relative weights at least annually to account for changes in resource consumption. These adjustments are made to reflect changes in treatment patterns, technology, and any other factors that may change the relative use of hospital resources.
For information on the adoption of the MS-DRGs in FY 2008, we refer readers to the FY 2008 IPPS final rule with comment period ( 72 FR 47140 through 47189 ).
For general information about the MS-DRG system, including yearly reviews and changes to the MS-DRGs, we refer readers to the previous discussions in the FY 2010 IPPS/rate year (RY) 2010 LTCH PPS final rule ( 74 FR 43764 through 43766 ) and the FYs 2011 through 2023 IPPS/LTCH PPS final rules ( 75 FR 50053 through 50055 ; 76 FR 51485 through 51487 ; 77 FR 53273 ; 78 FR 50512 ; 79 FR 49871 ; 80 FR 49342 ; 81 FR 56787 through 56872 ; 82 FR ( print page 69000) 38010 through 38085; 83 FR 41158 through 41258 ; 84 FR 42058 through 42165 ; 85 FR 58445 through 58596 ; 86 FR 44795 through 44961 ; and 87 FR 48800 through 48891 , respectively).
For discussion regarding our previously finalized policies (including our historical adjustments to the payment rates) relating to the effect of changes in documentation and coding that do not reflect real changes in case mix, we refer readers to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48799 through 48800 ).
Comment: Several commenters requested that CMS make a positive adjustment to the standardized amount to restore the full amount of the documentation and coding recoupment adjustments, which they asserted is required under section (7)(B)(2) and (4) of the TMA [Transitional Medical Assistance], Abstinence Education, and QI [Qualifying Individuals] Programs Extension Act of 2007 ( Pub. L. 110-90 ). Commenters stated that the statute is explicit that CMS may not carry forward any documentation and coding adjustments applied in fiscal years 2010 through 2017 into IPPS rates after FY 2023. Commenters contended that CMS, by its own admission, has restored only 2.9588 percentage points of a total 3.9 percentage point reduction. By not fully restoring the total reductions, commenters believe that CMS is improperly extending payment adjustments beyond the FY 2023 statutory limit.
Response: As of FY 2023, CMS completed the statutory requirements of section 7(b)(1)(B) of Public Law 110-90 as amended by section 631 of the American Taxpayer Relief Act of 2012 (ATRA, Pub. L. 112-240 ), section 404 of the Medicare Access and CHIP Reauthorization Act of 2015 (MACRA), and section 15005 of the 21st Century Cures Act ( Pub. L. 114-255 ). As we discussed in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44794 through 44795 ), the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58444 through 58445 ) and in prior rules, we believe section 414 of the MACRA and section 15005 of the 21st Century Cures Act set forth the levels of positive adjustments for FYs 2018 through 2023. We are not convinced that the adjustments prescribed by MACRA were predicated on a specific adjustment level estimated or implemented by CMS in previous rulemaking. We see no evidence that Congress enacted these adjustments with the intent that CMS would make an additional +0.7 percentage point adjustment in FY 2018 to compensate for the higher than expected final ATRA adjustment made in FY 2017, nor are we persuaded that it would be appropriate to use the Secretary's exceptions and adjustments authority under section 1886(d)(5)(I) of the Act to adjust payments in FY 2025 to restore any additional amount of the original 3.9 percentage point reduction, given Congress' directive regarding prescriptive adjustment levels under section 414 of the MACRA and section 15005 of the 21st Century Cures Act. Accordingly, in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38009 ), we implemented the required +0.4588 percentage point adjustment to the standardized amount for FY 2018. In the FY 2019 IPPS/LTCH PPS final rule (FY 2019 final rule) ( 83 FR 41157 ), the FY 2020 IPPS/LTCH PPS final rule (FY 2020 final rule) ( 84 FR 42057 ), the FY 2021 IPPS/LTCH PPS final rule (FY 2021 final rule) ( 85 FR 58444 and 58445 ), the FY 2022 IPPS/LTCH PPS final rule (FY 2022 final rule) ( 86 FR 44794 and 44795 ), and the FY 2023 IPPS/LTCH PPS final rule (FY 2023 final rule) ( 87 FR 48800 ), consistent with the requirements of section 414 of the MACRA, we implemented 0.5 percentage point positive adjustments to the standardized amount for FY 2019, FY 2020, FY 2021, FY 2022 and FY 2023, respectively. As discussed in the FY 2023 final rule, the finalized 0.5 percentage point positive adjustment for FY 2023 is the final adjustment prescribed by section 414 of the MACRA.
As of October 1, 2015, providers use the International Classification of Diseases, 10th Revision (ICD-10) coding system to report diagnoses and procedures for Medicare hospital inpatient services under the MS-DRG system instead of the ICD-9-CM coding system, which was used through September 30, 2015. The ICD-10 coding system includes the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) for diagnosis coding and the International Classification of Diseases, 10th Revision, Procedure Coding System (ICD-10-PCS) for inpatient hospital procedure coding, as well as the ICD-10-CM and ICD-10-PCS Official Guidelines for Coding and Reporting. For a detailed discussion of the conversion of the MS-DRGs to ICD-10, we refer readers to the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56787 through 56789 ).
As discussed in the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28127 ) and final rule ( 87 FR 48800 through 48801 ), beginning with FY 2024 MS-DRG classification change requests, we changed the deadline to request changes to the MS-DRGs to October 20 of each year to allow for additional time for the review and consideration of any proposed updates. We also described the new process for submitting requested changes to the MS-DRGs via a new electronic application intake system, Medicare Electronic Application Request Information System TM (MEARIS TM ), accessed at https://mearis.cms.gov . We stated that effective with FY 2024 MS-DRG classification change requests, CMS will only accept requests submitted via MEARIS TM and will no longer consider requests sent via email. Additionally, we noted that within MEARIS TM , we have built in several resources to support users, including a “Resources” section available at https://mearis.cms.gov/public/resources with technical support available under “Useful Links” at the bottom of the MEARIS TM site. Questions regarding the MEARIS TM system can be submitted to CMS using the form available under “Contact”, also at the bottom of the MEARIS TM site. Accordingly, interested parties had to submit MS-DRG classification change requests for FY 2025 by October 20, 2023.
We note that the burden associated with this information collection requirement is the time and effort required to collect and submit the data in the request for MS-DRG classification changes to CMS. The aforementioned burden is subject to the Paperwork Reduction Act (PRA) of 1995 and approved under OMB control number 0938-1431, and has an expiration date of 09/30/2025.
As noted previously, interested parties had to submit MS-DRG classification change requests for FY 2025 by October 20, 2023. As we have discussed in prior rulemaking, we may not be able to fully consider all of the requests that we receive for the upcoming fiscal year. We have found that, with the implementation of ICD-10, some types of requested changes to the MS-DRG classifications require more extensive research to identify and analyze all of the data that are relevant to evaluating the potential change. In the proposed rule, we noted those topics for which further research and analysis ( print page 69001) are required, and which we will continue to consider in connection with future rulemaking as summarized in the discussion that follows.
As discussed in the proposed rule, we received four requests to modify the GROUPER logic in a number of cardiac MS-DRGs under Major Diagnostic Category (MDC) 05 (Diseases and Disorders of the Circulatory System). Specifically, we received requests to:
- Modify the GROUPER logic of new MS-DRG 212 (Concomitant Aortic and Mitral Valve Procedures) to be defined by cases reporting procedure codes describing a single open mitral or aortic valve replacement/repair (MVR or AVR) procedure, plus an open coronary artery bypass graft procedure (CABG) or open surgical ablation or cardiac catheterization procedure plus a second concomitant procedure.
- Modify the GROUPER logic of new MS-DRG 212 by redefining the procedure code list that describes the performance of a cardiac catheterization by either removing the ICD-10-PCS codes that describe plain radiography of coronary artery codes from the logic list or adding ICD-10-PCS procedure codes that involve computed tomography (CT) or magnetic resonance imaging (MRI) scanning using contrast to the list. This requestor also suggested that CMS add ICD-10-PCS procedures codes that describe endovascular valve replacement or repair procedures into the GROUPER logic of MS-DRG 212.
- Modify the GROUPER logic of new MS-DRGs 323, 324, and 325 (Coronary Intravascular Lithotripsy with Intraluminal Device with MCC, without MCC, and without Intraluminal Device, respectively). In two separate but related requests, the requestors suggested that we add procedure codes that describe additional percutaneous coronary intervention (PCI) procedures such as percutaneous coronary rotational, laser, and orbital atherectomy to the GROUPER logic of new MS-DRGs 323, 324, and 325.
In the proposed rule, we stated that we appreciated the submissions and related analyses provided by the requestors for our consideration as we reviewed MS-DRG classification change requests for FY 2025; however, we also noted the complexity of the GROUPER logic for these MS-DRGs in connection with these requests requires more extensive analyses to identify and evaluate all of the data relevant to assessing these potential modifications. Specifically, we noted the list of procedure codes that describe the performance of a cardiac catheterization is in the definition of multiple MS-DRGs in MDC 05. Analyzing the impact of revising this list would necessitate evaluating the impact across numerous other MS-DRGs in MDC 05 that also include this list in their definition, in addition to new MS-DRG 212. Secondly, as discussed further in section II.C.4.c. of the preamble of the proposed rule, we stated that our analysis continues to indicate that, when performed, open cardiac valve replacement and supplement procedures are clinically different from endovascular cardiac valve replacement and supplement procedures in terms of technical complexity and hospital resource use. Lastly, as we have stated in prior rule making ( 88 FR 58708 ), atherectomy is distinct from coronary lithotripsy in that each of these procedures are defined by clinically distinct definitions and objectives. Additional analysis to assess for unintended consequences across the classification is needed as we have made a distinction between the root operations used to describe atherectomy (Extirpation) and the root operation used to describe lithotripsy (Fragmentation) in evaluating other requests in rulemaking. We stated we will need to consider the application of these two root operations in other scenarios where we have also specifically stated that Extirpation is not the same as Fragmentation and do not warrant similar MS-DRG assignment ( 85 FR 58572 through 58573 ). Furthermore, as MS-DRG 212 and MS-DRGs 323, 324, and 325 recently became effective on October 1, 2023 (FY 2024), we stated additional time is needed to review and evaluate extensive modifications to the structure of these MS-DRGs.
Comment: Commenters stated that they appreciated CMS' decision to await further data before analyzing the impact of the requested changes to MS-DRG 212 and MS-DRGs 323, 324, and 325, and agreed that any changes to these MS-DRGs should be carefully reviewed, as they stated these changes could have a significant impact on the remaining MS-DRGs in MDC 05. While thanking CMS for the continued consideration of appropriate MS-DRG assignment for concomitant open cardiac procedures, many commenters reiterated the request to modify the GROUPER logic of new MS-DRG 212. Some commenters stated it would be more impactful if cases reporting a single valve procedure, a coronary artery bypass grafting (CABG) procedure, and a procedure code describing surgical ablation were assigned to MS-DRG 212 (Concomitant Aortic and Mitral Valve Procedures). Other commenters stated that they believe that the logic of MS-DRG 212 should be modified to recognize an open aortic valve repair or replacement procedure or a mitral valve repair or replacement procedure when performed with any of the other concomitant procedures currently listed in the logic for MS-DRG 212. Another commenter suggested that MS-DRG 212 be defined by cases reporting either a mitral valve repair or replacement (MVR) procedure or an aortic valve repair or replacement (AVR) procedure, plus two other concomitant cardiac procedures such as surgical ablation, coronary artery bypass graft surgery, pulmonary valve replacement, or tricuspid valve replacement. This commenter stated that they performed their own analysis of recent MedPAR data, and stated they found that cases for beneficiaries who are not treated for their atrial fibrillation (AF) during open MVR or AVR (or CABG) procedures (currently assigned to MS-DRGs 216, 217, 218, 219, 220, and 221 (Cardiac Valve & Other Major Cardiothoracic Procedure with and without Cardiac Catheterization, with MCC, with CC, and without CC/MCC, respectively)) may have as much as $7,000 in incremental hospital index costs and 1.6 extra hospital stay days compared to similar non-AF patients during their open MVR or AVR procedures.
Some commenters were not supportive of the suggestion to assign cases reporting a single AVR or MVR procedure and another concomitant procedure to MS-DRG 212. These commenters stated that assigning cases reporting a single AVR or MVR procedure and another concomitant procedure to MS-DRG 212 would have a significant negative impact on the remaining MS-DRGs, notably MS-DRG 216. Other commenters suggested that CMS consider moving the aortic and mitral valve procedure codes with the root operations of “Creation”, “Release”, “Restriction” and “Supplement,” that are currently listed under the Concomitant Procedures list in the GROUPER logic for MS-DRG 212 in the ICD-10 MS-DRG Definitions Manual Version 41.1 to the appropriate logic list of aortic valve or mitral valve procedures. This commenter stated that procedure codes with these other root operations also represent types of valvular repairs and should be included on the aortic valve procedures and mitral valve procedures logic lists rather than the “Concomitant Procedure” logic list. A few commenters urged CMS to devise a broader, more inclusive, supplemental payment mechanism to facilitate incremental payment when ( print page 69002) two major procedures are performed during the same hospital admission.
In regard to the request to modify the GROUPER logic of new MS-DRGs 323, 324, and 325 (Coronary Intravascular Lithotripsy with Intraluminal Device with MCC, without MCC, and without Intraluminal Device, respectively), some commenters stated they agreed with CMS' assessment that atherectomy and coronary lithotripsy are mechanistically and clinically distinct. A commenter specifically noted that this distinction is supported by scientific literature and applauded CMS for demonstrating consistency on these questions and awareness of their impact across MDC 05. Other commenters stated they were disappointed that CMS did not propose to modify MS-DRGs 323, 324, and 325 to add procedure codes describing complex PCI procedures, including percutaneous coronary atherectomy procedures for FY 2025. A commenter stated that they offer a broad portfolio of products across the percutaneous coronary intervention space and believe they can provide additional input and data for consideration that would be helpful to CMS in evaluating potential modifications to the GROUPER logic to include orbital atherectomy procedures in the newly created MS-DRGs. Another commenter noted that the pipeline for additional technologies in the atherectomy family is expanding and recommended that CMS undertake an analysis of all ICD-10-PCS codes for atherectomy. A commenter questioned if Extirpation was the appropriate root operation to describe rotational and orbital atherectomy, as in their view, the procedures themselves are not removing calcified material. This commenter stated that in prior rulemaking CMS has stated procedures such as rotational and orbital atherectomy are reported with the root operation Extirpation because both techniques cut up the calcified material into small particles that are removed from the blood stream by the normal hemofiltration process and noted that in lithotripsy procedures, which are reported with the root operation Fragmentation, the normal hemofiltration process also removes the fragmented calcified material from the blood stream and suggested that CMS reconsider the root operation of atherectomy procedures as Fragmentation rather than Extirpation.
Response: We thank the commenters for sharing their feedback on these requests. As discussed in the proposed rule, we have found that with the implementation of ICD-10, some types of requested changes to the MS-DRG classifications require more extensive research to identify and analyze the relevant data for evaluating a potential change. The comments received in response to our proposed rule discussion of the requests to modify the GROUPER logic of new MS-DRG 212, specifically, illustrate the complexity of the analysis and evaluation required to address these requests. Notably, many commenters believe that a modification to the logic of MS-DRG 212 may be warranted but differ greatly in the solution they believe would best address the concerns noted. We appreciate the public comments we received on these requests and will take these suggestions under consideration as we continue to monitor for impacts in MDC 05 and across the MS-DRGs to avoid unintended consequences or missed opportunities in most appropriately capturing the resource utilization and clinical coherence for these subsets of procedures. We note that we would address any proposed modifications to the existing logic in future rulemaking.
As discussed in the proposed rule, as we continue the analysis of the claims data with respect to MS-DRGs in MDC 05, we welcome public comments and feedback on other factors that should be considered in the potential restructuring of these MS-DRGs. Feedback and other suggestions may be directed to MEARIS TM at: https://mearis.cms.gov/public/home . Interested parties should submit any MS-DRG classification change requests, including any comments and suggestions for FY 2026 consideration by October 20, 2024 via MEARIS TM at: https://mearis.cms.gov/public/home .
As we did for the FY 2024 IPPS/LTCH PPS proposed rule, for the FY 2025 IPPS/LTCH PPS proposed rule we provided a test version of the ICD-10 MS-DRG GROUPER Software, Version 42, so that the public can better analyze and understand the impact of the proposals included in the proposed rule. We noted that this test software reflected the proposed GROUPER logic for FY 2025. Therefore, it included the new diagnosis and procedure codes that are effective for FY 2025 as reflected in Table 6A.—New Diagnosis Codes—FY 2025 and Table 6B.—New Procedure Codes—FY 2025 that were associated with the proposed rule, and does not include the diagnosis codes that are invalid beginning in FY 2025 as reflected in Table 6C.—Invalid Diagnosis Codes—FY 2025, and Table 6D.—Invalid Procedure Codes—FY 2025 associated with the proposed rule. Those tables were not published in the Addendum to the proposed rule, but are available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html as described in section VI. of the Addendum to the proposed rule. Because the diagnosis codes no longer valid for FY 2025 are not reflected in the test software, we made available a supplemental file in Table 6P.1a and 6P.1b that includes the mapped Version 42 FY 2025 ICD-10-CM and ICD-10-PCS codes and the deleted Version 41 FY 2024 ICD-10-CM codes and V41.1 ICD-10-PCS codes that should be used for testing purposes with users' available claims data. Therefore, users had access to the test software allowing them to build case examples that reflect the proposals that were included in the proposed rule. In addition, users were able to view the draft version of the ICD-10 MS-DRG Definitions Manual, Version 42.
Comment: A commenter expressed its appreciation that we provided a test version of the ICD-10 MS-DRG GROUPER Software, Version 42, however, the commenter stated that this version essentially only allows for a case-by-case analysis and a minimal batch analysis. The commenter stated that it would be more beneficial to have a Batch z/OS version of the test GROUPER so that it could be better utilized for broader and more meaningful analysis purposes. The commenter requested that availability of a Batch z/OS version of the test GROUPER be made publicly available for all future rulemaking.
Response: We appreciate the commenter's feedback and will take the suggestion into consideration.
We noted in the proposed rule that in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58764 ), as discussed in the CY 2024 Outpatient Prospective Payment System and Ambulatory Surgical Center (OPPS/ASC) proposed rule (CY 2024 OPPS/ASC proposed rule) ( 88 FR 49552 , July 31, 2023), we stated that, consistent with the process that is used for updates to the “Integrated” Outpatient Code Editor (I/OCE) and other Medicare claims editing systems, we proposed to address any future revisions to the IPPS Medicare Code Editor (MCE), including any additions or deletions of claims edits, as well as the addition or deletion of ICD-10 diagnosis and procedure codes to the applicable MCE edit code lists, outside of the annual IPPS rulemakings. As discussed in the CY 2024 OPPS/ASC proposed rule, we proposed to remove discussion of the IPPS MCE from the annual IPPS rulemakings, beginning with the FY 2025 rulemaking, and to generally address future changes or updates to the MCE through instruction to the ( print page 69003) Medicare administrative contractors (MACs). We encouraged readers to review the discussion in the CY 2024 OPPS/ASC proposed rule and submit comments in response to the proposal by the applicable deadline by following the instructions provided in that proposed rule.
As also discussed in the proposed rule, in the CY 2024 OPPS/ASC final rule ( 88 FR 82121 through 82124 ), after consideration of the public comments we received, we finalized the proposal to remove discussion of the MCE from the annual IPPS rulemakings, beginning with FY 2025 rulemaking, and to generally address future changes or updates to the MCE through instruction to the MACs. We also stated that, beginning with FY 2025, in association with the annual proposed rule, we are making available a draft version of the Definitions of Medicare Code Edits (MCE) Manual to provide the public with an opportunity to review any changes that will become effective October 1 for the upcoming fiscal year. In addition, as a result of new and modified code updates approved after the annual spring ICD-10 Coordination and Maintenance Committee meeting, any further changes to the MCE will be reflected in the finalized Definitions of Medicare Code Edits (MCE) Manual, made available in association with the annual final rule. As such, we made available the draft FY 2025 ICD-10 MCE Version 42 Manual file on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
We noted in the proposed rule that the MCE manual is comprised of two chapters: Chapter 1: Edit code lists provides a listing of each edit, an explanation of each edit, and as applicable, the diagnosis and/or procedure codes for each edit, and Chapter 2: Code list changes summarizes the changes in the edit code lists (for example, additions and deletions) from the prior release of the MCE software. We also stated that the public may submit any questions, comments, concerns, or recommendations regarding the MCE to the CMS mailbox at [email protected] for our review and consideration.
Comment: Several commenters requested that CMS reconsider including updates to the MCE as part of the IPPS rulemaking process. A commenter stated that it recognized the importance of the MCE and expressed concern with the removal of MCE proposals from IPPS rulemaking. The commenter stated that identifying key considerations and mitigating unintended consequences are a key benefit of public review and consideration of stakeholder comments. The commenter stated that the proposed process is not transparent on key areas such as when the manual will be updated, effective dates, or the ability to provide feedback with timely responses. Other commenters stated that the MCE and related proposals include essential topics that warrant thorough review and consideration specific to inpatient hospital admissions and operational processes. The commenters asserted that these topics are vital to coding, clinical documentation, and revenue cycle professionals to ensure awareness and understanding ahead of implementation and historically allowed the opportunity for comment as applicable. According to the commenters, MCE change updates managed outside the IPPS rulemaking process create a strong potential for missed opportunities for pertinent public review and comment. The commenters stated these missed opportunities will create the potential for unintended consequences and administrative burdens for hospital teams. The commenters also stated that a historical review of IPPS comments in response to MCE proposals includes feedback on unacceptable principal diagnoses, age edits, and especially comments that affected the proposal and final implementation of CMS's unspecified code edit implemented in FY 2022.
The commenters stated that the draft version of the Definitions of Medicare Code Edits (MCE) Manual file made available in association with the proposed rule is a helpful reference, however revisions should be explicitly stated as proposed revisions or additions for consideration. According to the commenters, as currently written, the changes are not listed as proposals within the manual and are implied as changes that have already been decided and will be effective with the upcoming fiscal year. Another commenter expressed appreciation that CMS stated it will make available a draft version of the Definitions of Medicare Code Edits (MCE) Manual file in association with the annual proposed rule to provide the public with an opportunity to review any changes that will become effective October 1 for the upcoming fiscal year. However, the commenter also stated that it is difficult to identify the changes in the draft version of the MCE Manual and recommended that CMS provide a list of the draft MCE changes each year (including any additions or deletions of diagnosis or procedure codes or MCE edits).
Response: We appreciate the commenters' feedback. As stated in the CY 2024 OPPS/ASC final rule ( 88 FR 82121 through 82124 ), in the preamble of the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35949 ), and previously described in the preamble of this final rule, after consideration of the public comments we received, we finalized the proposal to remove discussion of the MCE from the annual IPPS rulemakings, beginning with FY 2025 rulemaking. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35949 ), we stated that beginning with FY 2025, in association with the annual proposed rule, we are making available a draft version of the Definitions of Medicare Code Edits (MCE) Manual to provide the public with an opportunity to review any changes that will become effective October 1 for the upcoming fiscal year.
We noted in the proposed rule, and as previously described in this final rule, that the MCE manual is comprised of two chapters: Chapter 1: Edit code lists provides a listing of each edit, an explanation of each edit, and as applicable, the diagnosis and/or procedure codes for each edit, and Chapter 2: Code list changes summarizes the changes in the edit code lists (for example, additions and deletions) from the prior release of the MCE software. We believe that Chapter 2: Code list changes in the MCE manual is clear as it lists the specific edit, followed by the list of codes that were added or deleted. The draft version of the Definitions of Medicare Code Edits (MCE) Manual will continue to be made publicly available in association with the annual proposed rulemaking, and it is referred to as a “draft version”. However, the Chapter 2: Code list changes are not “draft” MCE changes. Rather, consistent with our established process to assign MS-DRGs to new diagnosis codes and new procedures codes, for which we examine the MS-DRG assignment for the predecessor code to determine the most appropriate MS-DRG assignment, we have historically used, and will continue to use, a similar process in the assignment of new diagnosis codes and new procedure codes to the edit codes lists under the MCE. Specifically, we review the predecessor code to determine if there are edits under the MCE for which the predecessor code is listed to determine which edit lists may be appropriate for the newly created codes.
As discussed in prior rulemaking ( 88 FR 58764 ), as a result of new and modified code updates approved after the annual spring ICD-10 Coordination ( print page 69004) and Maintenance Committee meeting, we routinely make changes to the MCE without discussion in IPPS rulemaking. In the past, in both the IPPS proposed and final rules, we have only provided the list of changes to the MCE that were brought to our attention after the prior year's final rule. We historically have not listed all of the changes we have made to the MCE because of the new and modified codes approved after the annual spring ICD-10 Coordination and Maintenance Committee meeting. We stated that these changes are, and would still be, approved too late in the rulemaking schedule for inclusion in the proposed rule. Furthermore, although in the past our MCE policies have been described in our proposed and final rules, we have not provided the detail of each new or modified diagnosis and procedure code edit in the final rule.
Therefore, although we published, and will continue to publish, the edit code list changes in the “draft version” of the MCE manual, because discussion of the MCE has been removed from IPPS rulemakings, beginning with FY 2025 rulemaking as previously described, the edit code lists that appear in the “draft version” of the MCE manual in association with the proposed rule are considered final at the time of the development of the proposed rule. While the public may continue to submit any questions, comments, concerns, or recommendations regarding the MCE to the CMS mailbox at [email protected] for our review and consideration, we will continue to make available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software the changes to the edit code lists for both the draft version (at the time of the development of the proposed rule) and finalized version of the Definitions of Medicare Code Edits (MCE) file, in association with the annual IPPS proposed and final rules.
Comment: Some commenters encouraged CMS to delay, revisit, and provide details of specific code changes and the deactivation of edits. The commenters also stated that the edits are an additional quality assurance mechanism to ensure appropriate ICD-10-CM/PCS assignment for accurate and timely claims submission. The commenters further stated that the edits help to prevent added administrative burden associated with unnecessary claims rework and resubmission.
Response: We thank the commenters for their feedback. We believe that the FY 2025 MCE updates reflect our established process as previously described in this final rule, as well as address concerns related to claims processing discussed in prior rulemaking ( 88 FR 58768 ). We will continue to monitor these updates and consider issuing additional provider guidance to ensure accurate claims submission and processing.
Comment: Similar to the discussion in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58789 ), a commenter requested that CMS implement an edit for claims that group to MS-DRG 014 (Allogeneic Bone Marrow Transplant), that would reject claims when an inpatient type of bill 11X claim is received without charges mapped to revenue code 0815, which is intended to capture the costs of donor search and cell acquisition activities for allogeneic hematopoietic stem cell transplants. The commenter stated that mandatory reporting of the revenue code on inpatient claims would have several benefits, including increasing the accuracy of claims reporting by transplant centers, ensuring the accuracy of CMS's budget neutrality calculations, and helping to ensure that CMS does not inappropriately generate outlier payment on MS-DRG 014 claims (given that CMS removes costs associated with revenue code 0815 from its outlier calculation). The commenter stated it would also mirror the edit established under the outpatient code editor.
Response: We appreciate the commenter's feedback. As stated in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58789 ), we may consider provider education materials regarding the reporting of Allogeneic Stem Cell Acquisition/Donor Services in the future. We continue to believe that the suggested claims processing edit is not necessary at this time and expect providers to appropriately report charges associated with revenue code 0815.
Comment: A commenter stated it supported the removal of the vascular dementia codes from the Unacceptable Principal Diagnosis edit code list and that doing so will reduce administrative challenges with billing for services, improve the clinical accuracy of medical records and encourage appropriate care for this set of patients.
Response: We appreciate the commenter's support.
In summary, we thank the commenters for their views and feedback. Because we finalized the proposal to remove discussion of the MCE from the annual IPPS rulemakings beginning with FY 2025 rulemaking, the public may submit any future questions, comments, concerns, or recommendations regarding the MCE to the CMS mailbox at [email protected] for our review and consideration.
In association with the proposed rule, we made available the test version of the ICD-10 MS-DRG GROUPER Software, Version 42, the draft version of the ICD-10 MS-DRG Definitions Manual, Version 42, the draft version of the Definitions of Medicare Code Edits Manual, Version 42, and the supplemental mapping files in Table 6P.1a and 6P.1b of the FY 2024 and FY 2025 ICD-10-CM diagnosis and ICD-10-PCS procedure codes which are available at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/MS-DRG-Classifications-and-Software .
Following are the changes that we proposed to the MS-DRGs for FY 2025. We invited public comments on each of the MS-DRG classification proposed changes, as well as our proposals to maintain certain existing MS-DRG classifications discussed in the proposed rule. In some cases, we proposed changes to the MS-DRG classifications based on our analysis of claims data and clinical appropriateness. In other cases, we proposed to maintain the existing MS-DRG classifications based on our analysis of claims data and clinical appropriateness. As discussed in the FY 2025 IPPS/LTCH PPS proposed rule, our MS-DRG analysis was based on ICD-10 claims data from the September 2023 update of the FY 2023 MedPAR file, which contains hospital bills received from October 1, 2022, through September 30, 2023. In our discussion of the proposed MS-DRG reclassification changes, we referred to these claims data as the “September 2023 update of the FY 2023 MedPAR file.”
As explained in previous rulemaking ( 76 FR 51487 ), in deciding whether to propose to make further modifications to the MS-DRGs for particular circumstances brought to our attention, we consider whether the resource consumption and clinical characteristics of the patients with a given set of conditions are significantly different than the remaining patients represented in the MS-DRG. We evaluate patient care costs using average costs and lengths of stay and rely on clinical factors to determine whether patients are clinically distinct or similar to other patients represented in the MS-DRG. In evaluating resource costs, we consider both the absolute and percentage differences in average costs between the cases we select for review and the ( print page 69005) remainder of cases in the MS-DRG. We also consider variation in costs within these groups; that is, whether observed average differences are consistent across patients or attributable to cases that are extreme in terms of costs or length of stay, or both. Further, we consider the number of patients who will have a given set of characteristics and generally prefer not to create a new MS-DRG unless it would include a substantial number of cases.
In the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58448 ), we finalized our proposal to expand our existing criteria to create a new complication or comorbidity (CC) or major complication or comorbidity (MCC) subgroup within a base MS-DRG. Specifically, we finalized the expansion of the criteria to include the NonCC subgroup for a three-way severity level split. We stated we believed that applying these criteria to the NonCC subgroup would better reflect resource stratification as well as promote stability in the relative weights by avoiding low volume counts for the NonCC level MS-DRGs. We noted that in our analysis of MS-DRG classification requests for FY 2021 that were received by November 1, 2019, as well as any additional analyses that were conducted in connection with those requests, we applied these criteria to each of the MCC, CC, and NonCC subgroups. We also noted that the application of the NonCC subgroup criteria going forward may result in modifications to certain MS-DRGs that are currently split into three severity levels and result in MS-DRGs that are split into two severity levels. We stated that any proposed modifications to the MS-DRGs would be addressed in future rulemaking consistent with our annual process and reflected in Table 5—Proposed List of Medicare Severity Diagnosis Related Groups (MS-DRGs), Relative Weighting Factors, and Geometric and Arithmetic Mean Length of Stay for the applicable fiscal year.
In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44798 ), we finalized a delay in applying this technical criterion to existing MS-DRGs until FY 2023 or future rulemaking, in light of the public health emergency (PHE). Interested parties recommended that a complete analysis of the MS-DRG changes to be proposed for future rulemaking in connection with the expanded three-way severity split criteria be conducted and made available to enable the public an opportunity to review and consider the redistribution of cases, the impact to the relative weights, payment rates, and hospital case mix to allow meaningful comment prior to implementation.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48803 ), we also finalized a delay in application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split in light of the ongoing PHE and until such time additional analyses can be performed to assess impacts, as discussed in response to public comments in the FY 2022 and FY 2023 IPPS/LTCH PPS final rules.
In association with our discussion of application of the NonCC subgroup criteria in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26673 through 26676 ), we provided an alternate test version of the ICD-10 MS-DRG GROUPER Software, Version 41.A, reflecting the proposed GROUPER logic for FY 2024 as modified by the application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split, available at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/MS-DRG-Classifications-and-Software . Therefore, users had access to the alternate test software allowing them to build case examples that reflect the proposals included in the proposed rule with application of the NonCC subgroup criteria. We also provided additional files including an alternate Table 5—Alternate List of Medicare Severity Diagnosis Related Groups (MS-DRGs), Relative Weighting Factors, and Geometric and Arithmetic Mean Length of Stay, an alternate Length of Stay (LOS) Statistics file, an alternate Case Mix Index (CMI) file, and an alternate After Outliers Removed and Before Outliers Removed (AOR_BOR) file. The files are available in association with the FY 2024 IPPS/LTCH PPS proposed rule on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps . We stated that the alternate test software and additional files were made available so that the public could better analyze and understand the impact on the proposals included in the proposed rule if the NonCC subgroup criteria were to be applied to existing MS-DRGs with a three-way severity level split. We refer readers to the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26673 through 26676 ) for further discussion of the alternate test software and additional files that were made available.
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58655 through 58661 ), we finalized to delay the application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split for FY 2024. We stated that we would continue to review and consider the feedback we had received in response to the additional information we made available in association with the FY 2024 IPPS/LTCH PPS proposed rule for our development of the FY 2025 proposed rule.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35950 ), we noted that the IPPS Payment Impact File made available in connection with our annual IPPS rulemakings includes information used to categorize hospitals by various geographic and special payment consideration groups, including geographic location (urban or rural), teaching hospital status (that is, whether or not a hospital has GME residency programs and receives an IME adjustment), DSH hospital status (that is, whether or not a hospital receives Medicare DSH payments), special payment groups (that is, SCHs, MDHs, and RRCs) and other categories reflected in the impact analysis generally shown in Appendix A of the annual IPPS rulemakings. The IPPS Payment Impact File associated with the FY 2024 IPPS/LTCH PPS final rule can be found on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/fy-2024-ipps-final-rule-home-page#Data .
We proposed to continue to delay application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split for FY 2025, as we continue to consider the public comments received in response to the FY 2024 rulemaking. In addition, we encouraged interested parties to review the impacts and other information made available with the alternate test software (V41.A) and other additional files provided in connection with the FY 2024 IPPS/LTCH PPS proposed rule, as previously discussed, and stated that we continue to welcome feedback for consideration for future rulemaking.
Comment: Numerous commenters supported the proposal to continue to delay application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split for FY 2025.
Response: We thank the commenters for their support.
Comment: Several commenters expressed appreciation that CMS provided the meaningful data analysis and availability of the version 41.A alternate test GROUPER in association with the FY 2024 proposed rule, however, the commenters stated that the ability to utilize an updated alternate test software and a current batch GROUPER along with additional ( print page 69006) streamlined data by hospital type is needed. According to the commenters, updated test software and an available batch GROUPER would allow hospitals to further analyze the operational and monetary impact of this type of proposed change more thoroughly and over a longer time span.
Response: We appreciate the commenters' feedback. As we noted in the proposed rule, the IPPS Payment Impact File made available in connection with our annual IPPS rulemakings includes information used to categorize hospitals by various geographic and special payment consideration groups and other categories reflected in the impact analysis generally shown in Appendix A of the annual IPPS rulemakings. We will consider the commenters' request to provide updated test software and a batch GROUPER for future rulemaking.
Comment: A commenter who agreed with the proposal to delay application of the NonCC subgroup criteria stated that CMS did not provide any new information from, or analysis of, the FY 2023 MedPAR file as it related to base, deleted, or new MS-DRGs related to the application of the NonCC subgroup criteria. The commenter stated that new data should have been included with the proposed rule to continue efforts to view the impact of the policy.
Response: We appreciate the commenter's support and feedback. In response to the commenter's request that we provide the potential impacts using the FY 2023 claims data, we are making it available in Table 6P.4 on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps in association with this final rule.
We note that we did not propose to apply the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split for FY 2025. Moreover, as noted, we are continuing to consider comments received in response to FY 2024 rulemaking.
Comment: A commenter stated it utilized the files provided by CMS to analyze the impact of application of the NonCC subgroup criteria based on its own hospital volumes. The commenter reported that while it found some positive impacts to the relative weight of the MS-DRGs impacted when applying the NonCC subgroup criteria, they continue to have concerns regarding the variations in claims data from year-to-year that may be used in the proposed MS-DRG restructuring. The commenter stated it agreed with comments in prior years from various professional organizations that have noted the variability in claims data and, thus, case mix variations from year-to-year.
Response: We appreciate the commenter's feedback.
Comment: A few commenters expressed concern that application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split will reduce the impact of CCs. The commenters noted from prior year's analyses findings that there are a number of MS-DRGs that would potentially be consolidated to reflect the two-way severity split for “with MCC” and “without MCC” and there were not any that reflected a “with CC/MCC” and “without CC/MCC” severity level split. The commenters stated that the impact of CCs would decrease as a result of the application of the expanded criteria, meaning that conditions designated as CC would increasingly need to be MCCs in order to impact case complexity and severity.
Response: We thank the commenters for their feedback. We disagree that application of the NonCC subgroup criteria specifically reduces the impact of CCs. Rather, we believe that application of the criteria combines the subset of cases that may or may not report a CC into one MS-DRG grouping that reflects the average costs and length of stay for that subset. Because the IPPS MS-DRGs are a system of averages, the cases reporting a CC continue to impact the average costs and average length of stay within the subgroup. We note that in the majority of the MS-DRGs where we previously assessed the impact of application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split and provided the potential MS-DRG changes, the volume of cases in the CC subgroup was significantly greater than those in the NonCC subgroup, thus contributing more to the overall average costs and average length of stay of the “potential” new MS-DRG structure. We also note that providers have the ability to identify the subset of cases reporting a CC within the existing “with MCC” and “without MCC” MS-DRGs construct within their respective facilities.
After consideration of the public comments we received, and for the reasons discussed, we are finalizing our proposal to delay the application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split for FY 2025 as we continue to consider the public comments received in response to the FY 2024 rulemaking. We also continue to encourage interested parties to review the impacts and other information made available with the alternate test software (V41.A) and other additional files provided in connection with the FY 2024 IPPS/LTCH PPS proposed rule, as previously discussed. We continue to welcome feedback for consideration for future rulemaking that may be directed to MEARIS TM at https://mearis.cms.gov/public/home .
As discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58661 ), we continue to apply the criteria to create subgroups, including application of the NonCC subgroup criteria, in our annual analysis of MS-DRG classification requests, consistent with our approach since FY 2021 when we finalized the expansion of the criteria to include the NonCC subgroup for a three-way severity level split. Accordingly, in our analysis of the MS-DRG classification requests for FY 2025 that we received by October 20, 2023, as well as any additional analyses that were conducted in connection with those requests, we applied these criteria to each of the MCC, CC, and NonCC subgroups, as described in the following table.

In general, once the decision has been made to propose to make further modifications to the MS-DRGs as described previously, such as creating a new base MS-DRG, or in our evaluation of a specific MS-DRG classification request to split (or subdivide) an existing base MS-DRG into severity levels, all five criteria must be met for the base MS-DRG to be split (or subdivided) by a CC subgroup. We note that in our analysis of requests to create a new MS-DRG, we typically evaluate the most recent year of MedPAR claims data available. For example, we stated earlier that for the FY 2025 IPPS/LTCH PPS proposed rule, our MS-DRG analysis was based on ICD-10 claims data from the September 2023 update of the FY 2023 MedPAR file. However, in our evaluation of requests to split an existing base MS-DRG into severity levels, as noted in prior rulemaking ( 80 FR 49368 ), we typically analyze the most recent two years of data. This analysis includes two years of MedPAR claims data to compare the data results from one year to the next to avoid making determinations about whether additional severity levels are warranted based on an isolated year's data fluctuation and also, to validate that the established severity levels within a base MS-DRG are supported. The first step in our process of evaluating if the creation of a new CC subgroup within a base MS-DRG is warranted is to determine if all the criteria is satisfied for a three-way split. In applying the criteria for a three-way split, a base MS-DRG is initially subdivided into the three subgroups: MCC, CC, and NonCC. Each subgroup is then analyzed in relation to the other two subgroups using the volume (Criteria 1 and 2), average cost (Criteria 3 and 4), and reduction in variance (Criteria 5). If the criteria fail, the next step is to determine if the criteria are satisfied for a two-way split. In applying the criteria for a two-way split, a base MS-DRG is initially subdivided into two subgroups: “with MCC” and “without MCC” (1_23) or “with CC/MCC” and “without CC/MCC” (12_3). Each subgroup is then analyzed in relation to the other using the volume (Criteria 1 and 2), average cost (Criteria 3 and 4), and reduction in variance (Criteria 5). If the criteria for both of the two-way splits fail, then a split (or CC subgroup) would generally not be warranted for that base MS-DRG. If the three-way split fails on any one of the five criteria and all five criteria for both two-way splits (1_23 and 12_3) are met, we would apply the two-way split with the highest R2 value. We note that if the request to split (or subdivide) an existing base MS-DRG into severity levels specifies the request is for either one of the two-way splits (1_23 or 12_3), in response to the specific request, we will evaluate the criteria for both of the two-way splits; however, we do not also evaluate the criteria for a three-way split.
Comment: A commenter recommended that CMS consider patient risk adjustment as a criterion for creating CC and MCC subgroups, including the impact of multiple comorbidities. According to the commenter, published literature suggests that as comorbidity status increases, patient risk of clinical events increase, as well as potential resource use. For example, the commenter stated that studies suggest that in patients with one presenting risk factor/comorbidity (either hypertension, congenital heart disease, previous stroke, or diabetes), compared to patients without these risks, that the risk of future stroke was 1.96 greater. [ 4 ] According to the commenter, the authors also found patients with 2 or more of these risk factors to have an increased risk of future stroke at 2.87 greater the risk of patients without risk factors and stated that these results suggest the cumulative effect of multiple CCs can dramatically impact a patient's risk and resource use in the absence of an MCC. The commenter suggested that CMS should consider the impact of multiple CCs (heart failure, AF, etc.) as a criterion when grouping an inpatient procedure to an MCC grouping in the absence of MCC.
Response: We appreciate the commenter's input and will take it under consideration as we continue to consider feedback associated with application of the NonCC subgroup criteria.
We are making the FY 2025 ICD-10 MS-DRG GROUPER and Medicare Code Editor (MCE) Software Version 42, the ICD-10 MS-DRG Definitions Manual files Version 42 and the Definitions of Medicare Code Edits Manual Version 42 available to the public on our CMS ( print page 69008) website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35951 through 35952 ), we discussed a request we received to revise the title of Pre-MDC MS-DRG 018 (Chimeric Antigen Receptor (CAR) T-cell and Other Immunotherapies) in connection with an ICD-10-PCS procedure code request that was submitted via MEARIS TM by the December 1, 2023 deadline for consideration as an agenda topic to be discussed at the March 19-20, 2024 ICD-10 Coordination and Maintenance Committee meeting. The procedure code request involves the application of an autologous genetically engineered cell-based gene therapy, prademagene zamikeracel (PZ), that is indicated in the treatment of recessive dystrophic epidermolysis bullosa (RDEB), an extremely rare genetic disease of the skin that leads to large chronic wounds. The proposal was presented and discussed at the March 19-20, 2024, ICD-10 Coordination and Maintenance Committee meeting. We refer the reader to the CMS website at https://www.cms.gov/medicare/coding-billing/icd-10-codes/icd-10-coordination-maintenance-committee-materials for additional detailed information regarding the request, including a recording of the discussion and the related meeting materials. Public comments in response to the code proposal were due by April 19, 2024. The requestor suggested that if finalized, a new procedure code to identify the application of PZ should be assigned to Pre-MDC MS-DRG 018 and that the title for Pre-MDC MS-DRG 018 be revised to reflect “Chimeric Antigen Receptor (CAR) T and Other Autologous Gene and Cell Therapies”.
Because the diagnosis and procedure code proposals that are presented at the March ICD-10-CM Coordination and Maintenance Committee meeting for an October 1 implementation (upcoming FY) are not finalized in time to include in Table 6A.—New Diagnosis Codes and Table 6B.—New Procedure Codes in association with the proposed rule, as we have noted in prior rulemaking, we use our established process to examine the MS-DRG assignment for the predecessor codes to determine the most appropriate MS-DRG assignment. Specifically, we review the predecessor code and MS-DRG assignment most closely associated with the new procedure code, and in the absence of claims data, we consider other factors that may be relevant to the MS-DRG assignment, including the severity of illness, treatment difficulty, complexity of service and the resources utilized in the diagnosis and/or treatment of the condition. We have noted in prior rulemaking that this process does not automatically result in the new procedure code being assigned to the same MS-DRG or to have the same designation (O.R. versus Non-O.R.) as the predecessor code. Under this established process, the MS-DRG assignment for the upcoming fiscal year for any new diagnosis or procedure codes finalized after the March meeting would be reflected in Table 6A.—New Diagnosis Codes and Table 6B.—New Procedure Codes associated with the final rule for that fiscal year. Accordingly, we stated that the MS-DRG assignment for any new procedure codes describing PZ, if finalized following the March meeting, would be reflected in Table 6B.—New Procedure Codes associated with the final rule for FY 2025. As noted in prior rulemaking ( 87 FR 28135 ), the codes that are finalized after the March meeting are specifically identified with a footnote in Table 6A.—New Diagnosis Codes and Table 6B.—New Procedure Codes that are made publicly available in association with the final rule on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps . The public may provide feedback on these finalized assignments, which is then taken into consideration for the following fiscal year.
We note that the proposal to create new procedure codes that describe the application of PZ as discussed at the March 19-20, 2024, ICD-10 Coordination and Maintenance Committee meeting was approved and finalized as reflected in the FY 2025 ICD-10-PCS Code Update files that were made publicly available on the CMS website on June 5, 2024 at https://www.cms.gov/medicare/coding-billing/icd-10-codes/2025-icd-10-pcs .
We stated in the proposed rule that we did not agree with the request to revise the title for Pre-MDC MS-DRG 018 for FY 2025 as requested because the logic for Pre-MDC MS-DRG 018 is intended to include other immunotherapies and is not restricted to CAR T-cell and autologous gene and cell therapies. As discussed in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44798 through 44806 ), we finalized our proposal to revise the title of Pre-MDC MS-DRG 018 to include “Other Immunotherapies” to better reflect the cases reporting the administration of non-CAR T-cell therapies and other immunotherapies that would also be assigned to this MS-DRG, in addition to CAR T-cell therapies. We noted that the term “Other Immunotherapies” is intended to encompass the group of therapies that are currently available and being utilized today (for which codes have been created for reporting in response to industry requests or are being considered for implementation), and to enable appropriate MS-DRG assignment for any future therapies that may also fit into this category and are not specifically identified as a CAR T-cell product, that may become available (for example receive marketing authorization or a newly established procedure code in the ICD-10-PCS classification).
In the proposed rule we also noted that, as discussed in prior rulemaking, this category of therapies continues to evolve, and we are in the process of carefully considering the feedback we have previously received about ways in which we can continue to appropriately reflect resource utilization while maintaining clinical coherence and stability in the relative weights under the IPPS MS-DRGs. We stated we will continue to examine these complex issues in connection with future rulemaking and acknowledged that there may be distinctions to account for as we continue to gain more experience in the use of these therapies and have additional claims data to analyze.
Therefore, we did not propose to revise the title for Pre-MDC MS-DRG 018 to reflect “Chimeric Antigen Receptor (CAR) T and Other Autologous Gene and Cell Therapies” and proposed to maintain the existing title to Pre-MDC MS-DRG 018, “Chimeric Antigen Receptor (CAR) T-cell and Other Immunotherapies” for FY 2025.
Comment: Commenters expressed support for the proposal to maintain the existing title to Pre-MDC MS-DRG 018, “Chimeric Antigen Receptor (CAR) T-cell and Other Immunotherapies” for FY 2025.
Comment: A commenter stated that application of PZ (prademagene zamikeracel) seems to differ significantly in terms of clinical coherence and resource utilization from other therapies currently mapped to MS-DRG 018, specifically in that it requires an operating room and subsequent post-surgical care. According to the commenter, although CMS did not specifically propose to map cases reporting the application of PZ to Pre-MDC MS-DRG 018 for FY ( print page 69009) 2025, PZ does not appear to be a match for the technologies currently included in Pre-MDC MS-DRG 018 since it is not an immunotherapy and would be the only surgical episode of care in the MS-DRG. The commenter requested that CMS not finalize the mapping for application of PZ to Pre-MDC MS-DRG 018 due to differences in resource use.
Another commenter stated that if CMS were to continue to assign new, higher volume, lower cost therapies to MS-DRG 018, it could potentially distort the relative weight of the MS-DRG, resulting in inadequate payment for CAR T-cell therapies. This commenter also recommended that CMS not map cases reporting application of PZ to Pre-MDC MS-DRG 018 due to clinical resource differences with other therapies currently mapped to Pre-MDC MS-DRG 018. The commenter further stated that given the important role CAR T-cell therapies play, and will continue to play for cancer patients, CMS should clarify its methodology for the inclusion of new procedure codes within Pre-MDC MS-DRG 018 and consider the resource costs and needs of potential new therapies to this MS-DRG so as not to limit access to current therapies. Other commenters recommended that CMS provide transparency in the assignment of therapies to Pre-MDC MS-DRG 018 to ensure accurate, predictable, and appropriate payment, including consideration of comparable resource use to existing therapies currently mapped to Pre-MDC MS-DRG 018.
Another commenter requested that CMS map the new procedure codes describing application of PZ to Pre-MDC MS-DRG 018, given the clinical characteristics and resource intensity of the gene and cellular therapy. According to the commenter, administration of both autologous CAR T-cell therapies and PZ is initiated through the collection of a sample of the patient's own cells. The commenter stated the cells are then modified as part of a complex and resource intensive process requiring the insertion of a new gene into the patient's own cells before administering them back to the patient. Specifically, the commenter stated that the keratinocyte cells (that is, the most prominent cells in the epidermis) of patients diagnosed with RDEB are collected via a “punch” biopsy procedure and transduced with a functional COL7A1 transgene using a retroviral vector, which is intended to result in adequate expression and secretion of the type VII collagen protein critical to anchoring the epidermis and facilitating wound healing. The commenter stated the transduced cells are then expanded, matured, and processed into sheets through an approximate 25-day process before they can be delivered to the hospital site and applied to the patient. The commenter stated that this process mirrors the CAR T-cell therapy development and administration process, where cells are harvested from the patient's blood, the patient's T-cells are isolated through a leukapheresis procedure, and the T-cells then are transduced with a CAR-encoding viral vector and expanded over an approximate month-long period before being returned to the treatment center for administration to the patient. The commenter also stated that the application of PZ shares other similarities with the technologies currently assigned to Pre-MDC MS-DRG 018, including the need for an inter-disciplinary team of health care personnel, and an extended length of stay following treatment. According to the commenter, from a resource perspective, like other therapies currently assigned to Pre-MDC MS-DRG 018, the main driver of resource utilization for an inpatient stay is the administration of the technology.
Response: We appreciate the commenters' feedback. In response to the commenters who requested that CMS not finalize the mapping for application of PZ to Pre-MDC MS-DRG 018 due to the belief that there are differences in resource use when compared to other therapies currently mapped to Pre-MDC MS-DRG 018, we note that the commenters did not indicate whether they believed the differences in resource use for application of PZ are higher or lower in comparison to the other therapies currently mapped to Pre-MDC MS-DRG 018, nor did the commenters offer any alternative MS-DRG suggestions for CMS's consideration. We acknowledge that application of PZ requires use of an operating room and the administration of other therapies currently assigned to Pre-MDC MS-DRG 018 do not. We also note that consistent with our established process for assigning new diagnosis or new procedure codes to MDCs, MS-DRGs, and the associated attributes (severity level and O.R. status), we examined the MDCs, MS-DRG assignment and O.R. status of the predecessor procedure codes to inform our assignments and designations. As discussed in prior rulemaking and previously in the preamble of this final rule, we review the predecessor code and MS-DRG assignment most closely associated with the new diagnosis or procedure code, and in the absence of claims data, we consider other factors that may be relevant to the MS-DRG assignment, including the severity of illness, treatment difficulty, complexity of service and the resources utilized in the diagnosis and/or treatment of the condition. We have previously noted that this process does not automatically result in the new diagnosis or procedure code being assigned to the same MS-DRG or to have the same designation as the predecessor code. In our evaluation of MS-DRG classification requests under the IPPS MS-DRGs, consideration is also given to the similarities and differences in resource utilization among patients in each MS-DRG and we strive to ensure that resource utilization is relatively consistent across patients in each MS-DRG. However, some variation in resource intensity will remain among the patients in each MS-DRG because the definition of the MS-DRG is not so specific that every patient is identical, rather the average pattern of resource intensity of a group of patients in an MS-DRG can be predicted.
We note that historically, in the development of the DRGs, the initial step in the determination of the DRG had been the assignment of the appropriate MDC based on the principal diagnosis, however, beginning with the eighth version of the GROUPER (CMS 8.0), the initial step in DRG assignment was based on the procedure being performed, thus the creation of the Pre-MDC DRGs, where the patient is assigned to these DRGs independent of the MDC of the principal diagnosis. Therefore, while the existing therapies (that is, CAR T-cell and non-CAR T-cell) currently mapped to Pre-MDC MS-DRG 018 may be indicated in the treatment of patients with cancer, the logic for case assignment to Pre-MDC MS-DRG 018 does not preclude the assignment of other therapies indicated in the treatment of patients that do not have a diagnosis of cancer. In our review of the MS-DRG assignment for application of PZ, we recognized that this technology is defined as an investigational genetically engineered autologous cell therapy. We also note that similar to the discussions in prior rulemaking with respect to the difficulty in predicting what the associated costs will be in the future for CAR T-cell and other immunotherapies that remain under development ( 87 FR 48806 ), it is also difficult to predict what the associated costs will be in the future for cell and gene therapies that remain under development or in clinical trials.
We further note that, in response to the President's Executive Order 14087 , “Lowering Prescription Drug Costs for Americans”, a Cell and Gene Therapy ( print page 69010) (CGT) Access Model was developed, which could help inform future inpatient payment policy for cell and gene therapies more generally. For additional information on the CGT Access Model, we refer the reader to the CMS website at https://www.cms.gov/priorities/innovation/innovation-models/cgt .
Until such time additional data becomes available, we believe it is appropriate to map cases reporting the application of PZ to Pre-MDC MS-DRG 018 for FY 2025 based on the information currently available indicating similar utilization of resources for other cases currently mapped to MS-DRG 018 with regard to patients' severity of illness, treatment difficulty, and complexity of service.
In response to concerns that the assignment of new, higher volume, lower cost therapies to MS-DRG 018 could potentially distort the relative weight of the MS-DRG resulting in inadequate payment for CAR T-cell therapies, we note that in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48807 ), we addressed similar comments and also noted that we provided detailed summaries and responses to these same or similar comments in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44798 through 44806 ). We also refer the reader to the discussion in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36018 through 36020 ), and in section II.D.2.b. of this final rule, regarding the proposed and finalized relative weight methodology for cases mapping to Pre-MDC MS-DRG 018 effective October 1, 2024, for FY 2025.
After consideration of the public comments we received, we are finalizing our proposal to maintain the existing title to Pre-MDC MS-DRG 018, “Chimeric Antigen Receptor (CAR) T-cell and Other Immunotherapies” for FY 2025. We are also finalizing the assignment of the eight procedure codes describing the use of PZ to Pre-MDC MS-DRG 018 as reflected in Table 6B.—New Procedure Codes, in association with this final rule and available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps .
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58661 through 58667 ), we discussed a request to reassign cases describing the insertion of a neurostimulator generator into the skull in combination with the insertion of a neurostimulator lead into the brain from MS-DRG 023 (Craniotomy with Major Device Implant or Acute Complex CNS Principal Diagnosis with MCC or Chemotherapy Implant or Epilepsy with Neurostimulator) to MS-DRG 021 (Intracranial Vascular Procedures with Principal Diagnosis Hemorrhage with CC) or reassign all cases currently assigned to MS-DRG 023 that involve a craniectomy or a craniotomy with the insertion of device implant and create a new MS-DRG for these cases.
We stated the requestor acknowledged that the relatively low volume of cases that only involve the insertion of a neurostimulator generator into the skull in combination with the insertion of a neurostimulator lead into the brain in the claims data was likely not sufficient to warrant the creation of a new MS-DRG. The requestor further stated given the limited options within the existing MS-DRG structure that fit from both a cost and clinical cohesiveness perspective, they believed that MS DRG 021 was the most logical fit in terms of average costs and clinical coherence for reassignment even though, according to the requestor, the insertion of a neurostimulator generator into the skull in combination with the insertion of a neurostimulator lead into the brain is technically more complex and involves a higher level of training, extreme precision and sophisticated technology than performing a craniectomy for hemorrhage.
We noted that while our data findings demonstrated the average costs are higher for the cases with a principal diagnosis of epilepsy with a neurostimulator generator inserted into the skull and insertion of a neurostimulator lead into brain when compared to all cases in MS-DRG 023, these cases represented a small percentage of the total number of cases reported in this MS-DRG. We stated that while we appreciated the requestor's concerns regarding the differential in average costs for cases describing the insertion of a neurostimulator generator into the skull in combination with the insertion of a neurostimulator lead into the brain when compared to all cases in their assigned MS-DRG, we believed additional time was needed to evaluate these cases as part of our ongoing examination of the case logic to the MS-DRGs for craniotomy and endovascular procedures, which are MS-DRG 023, MS-DRG 024 (Craniotomy with Major Device Implant or Acute Complex CNS Principal Diagnosis without MCC), and MS-DRGs 025, 026, and 027 (Craniotomy and Endovascular Intracranial Procedures with MCC, with CC, and without CC/MCC, respectively).
As discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48808 through 48820 ), in connection with our analysis of cases reporting laser interstitial thermal therapy (LITT) procedures performed on the brain or brain stem in MDC 01, we stated we have started to examine the logic for case assignment to MS-DRGs 023 through 027 to determine where further refinements could potentially be made to better account for differences in the technical complexity and resource utilization among the procedures that are currently assigned to those MS-DRGs. We stated that specifically, we were in the process of evaluating procedures that are performed using an open craniotomy (where it is necessary to surgically remove a portion of the skull) versus a percutaneous burr hole (where a hole approximately the size of a pencil is drilled) to obtain access to the brain in the performance of a procedure. We stated we were also reviewing the indications for these procedures, for example, malignant neoplasms versus epilepsy to consider if there may be merit in considering restructuring the current MS-DRGs to better recognize the clinical distinctions of these patient populations in the MS-DRGs.
As part of this evaluation, as discussed in the FY 2024 IPPS/LTCH PPS final rule, we have begun to analyze the ICD-10 coded claims data to determine if the patients' diagnoses, the objective of the procedure performed, the specific anatomical site where the procedure is performed or the surgical approach used (for example, open, percutaneous, percutaneous endoscopic, among others) demonstrates a greater severity of illness and/or increased treatment difficulty as we consider restructuring MS-DRGs 023 through 027, including how to better align the clinical indications with the performance of specific intracranial procedures. We referred the reader to Tables 6P.2b through 6P.2f associated with the FY 2024 IPPS/LTCH PPS proposed rule (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps ) for data analysis findings of cases assigned to MS-DRGs 023 through 027 from the September 2022 update of the FY 2022 MedPAR file as we continue to look for patterns of complexity and resource intensity.
In summary, we stated that while we agreed that neurostimulator cases can have average costs that are higher than the average costs of all cases in their respective MS-DRGs, in our analysis of this issue, it was difficult to detect ( print page 69011) patterns of complexity and resource intensity. Therefore, for the reasons discussed, we finalized our proposal to maintain the current assignment of cases describing a neurostimulator generator inserted into the skull with the insertion of a neurostimulator lead into the brain for FY 2024.
In the FY 2024 IPPS/LTCH PPS final rule, we stated we continue to believe that additional time is needed to evaluate these cases as part of our ongoing examination of the case logic for MS-DRGs 023 through 027. As part of our ongoing, comprehensive analysis of the MS-DRGs under ICD-10, we stated we would continue to explore mechanisms to ensure clinical coherence between these cases and the other cases with which they may potentially be grouped. We stated that the data analysis as displayed in Tables 6P.2b through 6P.2f associated with the FY 2024 IPPS/LTCH PPS proposed rule was displayed to provide the public an opportunity to review our examination of the procedures by their approach (open versus percutaneous), clinical indications, and procedures that involve the insertion or implantation of a device and to reflect on what factors should be considered in the potential restructuring of these MS-DRGs. We welcomed further feedback on how CMS should define technical complexity, what factors should be considered in the analysis, and whether there are other data not included in Tables 6P.2b through 6P.2f that CMS should analyze. We also stated we are interested in receiving feedback on where further refinements could potentially be made to better account for differences in the technical complexity and resource utilization among the procedures that are currently assigned to these MS-DRGs.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35952 through 35953 ), we discussed two comments we received by the October 20, 2023 deadline in response to this discussion in the FY 2024 IPPS/LTCH PPS final rule. A commenter recommended that CMS not use surgical approach (for example, open versus percutaneous) as a factor to reclassify MS-DRGs 023 through 027. The commenter stated whether the opening is created via a drill into the skull percutaneously or through a larger incision in the skull for a craniotomy, both approaches involve the risk of intracranial bleeding, infection, and brain swelling. The commenter further stated they do not support a consideration of the reassignment of the ICD-10-PCS procedure codes describing LITT, currently assigned to MS-DRGs 025 through 027, based on the diagnosis being treated. The commenter stated that the LITT procedure requires the same steps, time, and clinical resources when performed for brain cancer or epilepsy. In the commenter's view, differences in the disease causing the tumors or lesions do not affect the resources used for performing the procedure or the post-operative care for the patient. Lastly, the commenter stated they support the current structure of MS-DRGs 023 and 024 based on an acute complicated principal diagnosis, or chemotherapy implant, or epilepsy with neurostimulator. The commenter stated these diagnoses represent severe complex conditions that require immediate and urgent intervention.
Another commenter stated that the current logic for MS-DRGs 023 through 027 is sufficient and supports the clinical and resource similarities of the procedures reflected in these MS-DRGs. The commenter performed its own analysis and stated they found that realignment based on surgical approach or root operation could create significant new inequities. The commenter recommended that CMS maintain the current logic for MS-DRGs 025 through 027, as making changes could be disruptive to hospitals and create challenges for Medicare beneficiary access to life-saving technologies. The commenter stated they strongly believe that maintaining the current structure provides payment stability and integrity of these procedures over time.
In this final rule, we summarize the additional comments we received in response to this discussion in the FY 2025 IPPS/LTCH PPS proposed rule.
Comment: Commenters stated they support CMS' decision to continue to monitor the case logic for MS-DRGs 023 through 027 to determine if future changes are warranted. A commenter specifically stated in their review, they were unable to detect misalignment in patterns of complexity or resource intensity within MS-DRGs 023 through 027 and noted the procedures are well-established. Another commenter stated they appreciate CMS reviewing the craniotomy MS-DRGs and stated that CMS should ensure that MS-DRG assignments fully reflect all costs for very resource-intensive craniotomy procedures. This commenter also recommended that CMS expand its review of the craniotomy MS-DRGs to include MS-DRGs 020, 021, and 022 (Intracranial Vascular Procedures with Principal Diagnosis Hemorrhage with MCC, with CC, and without CC/MCC, respectively) and stated that the payments for these MS-DRGs have been highly variable in recent years, notably being proposed to reduce by more than 7 percent for FY 2025, and may fail to adequately reflect the resources associated with care for patients with diagnoses such as aneurysms. The commenter encouraged CMS to examine these MS-DRGs with the goal of providing more stable payments for hospitals that furnish intensive craniotomy procedures and to mitigate the financial impact of large payment declines. Several other commenters expressed caution, however, and stated that CMS should allow providers more time to identify which diagnoses support this procedure code and as such do not agree with moving it to MS-DRG 021.
Response: We thank the commenters and appreciate the commenters' support and feedback. CMS will continue to monitor and analyze the claims data with respect to MS-DRGs 023 through 027 and we will take the recommendation to also review MS-DRGs 020, 021, and 022 into consideration as we further examine the logic for case assignment to the craniotomy MS-DRGs. We note that we did not propose or finalize a change to the GROUPER logic of MS-DRGs 020, 021, and 022 in FY 2024 IPPS/LTCH PPS rulemaking, nor did we propose a change to the GROUPER logic of these MS-DRGS in the FY 2025 IPPS/LTCH PPS proposed rule, therefore, the difference in the relative weights reflected in Table 5—List of Medicare Severity Diagnosis Related Groups (MS-DRGs), Relative Weighting Factors, and Geometric and Arithmetic Mean Length of Stay associated with FY 2025 proposed rule for MS-DRGs 020, 021, and 022 can be attributed to changes in the underlying data.
In response to the comments suggesting that CMS allow more time, it is unclear which diagnosis code and which procedure code the commenters were referring to as CMS did not propose to move any codes to MS-DRG 021 in the FY 2025 IPPS/LTCH proposed rule, and the commenters did not specifically identify any ICD-10 codes for CMS to consider.
CMS appreciates the comments submitted in response to the request for feedback in the FY 2024 IPPS/LTCH PPS final rule, as well as the comments submitted in response to the discussion in the FY 2025 IPPS/LTCH PPS proposed rule. As we continue analysis of the claims data with respect to MS-DRGs 023 through 027, we continue to seek public comments and feedback on other factors that should be considered in the potential restructuring of these MS-DRGs. As stated in prior ( print page 69012) rulemaking, we recognize the logic for MS-DRGs 023 through 027 has grown more complex over the years and believe there is opportunity for further refinement. We refer the reader to the ICD-10 MS-DRG Definitions Manual, Version 42 (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRGs 023 through 027 for FY 2025. Feedback and other suggestions may continue to be directed to MEARIS TM , discussed in section II.C.1.b. of the preamble of this final rule at: https://mearis.cms.gov/public/home .
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35953 through 35956 ), we received a request to add ICD-10-PCS procedure codes D0Y0CZZ (Intraoperative radiation therapy (IORT) of brain) and D0Y1CZZ (Intraoperative radiation therapy (IORT) of brain stem), to the Chemotherapy Implant logic list in MS-DRG 023 (Craniotomy with Major Device Implant or Acute Complex CNS Principal Diagnosis with MCC or Chemotherapy Implant or Epilepsy with Neurostimulator). According to the requestor, intraoperative radiation therapy (IORT) for the brain is always performed as part of the surgery to remove a brain tumor during the same operative episode. The requestor stated that once maximal safe tumor resection is achieved, the tumor cavity is examined for active egress of cerebrospinal fluid or bleeding. Next, intraoperative measurements are made using neuro-navigation or intraoperative imaging such as magnetic resonance imaging (MRI) or computed tomography (CT) to ensure safe distance to organs or tissues at risk, aid in appropriate dose calculation, and selection of proper applicator size. The applicator is then implanted into the tumor cavity and the radiation dose is delivered. The requestor stated that delivery time can be up to 40 minutes and upon completion of the treatment, the source is removed, and the cavity is re-inspected for active egress of cerebrospinal fluid and bleeding.
As discussed in the proposed rule, the requestor stated that currently the ICD-10-PCS procedure codes for excision of a brain tumor, 00B00ZZ (Excision of brain, open approach) and 00B70ZZ (Excision of cerebral hemisphere, open approach) map to both sets of craniotomy MS-DRGs. Specifically, MS-DRG 023 (Craniotomy with Major Device Implant or Acute Complex CNS Principal Diagnosis with MCC or Chemotherapy Implant or Epilepsy with Neurostimulator) and MS-DRG 024 (Craniotomy with Major Device Implant or Acute Complex CNS Principal Diagnosis without MCC), and MS-DRGs 025, 026, and 027 (Craniotomy and Endovascular Intracranial Procedures with MCC, with CC, and without CC/MCC, respectively). However, the requestor also stated that the procedure codes describing IORT (D0Y0CZZ or D0Y1CZZ) are not listed in the GROUPER logic and do not affect MS-DRG assignment. Therefore, cases reporting a procedure code describing excision of a brain tumor (00B00ZZ or 00B70ZZ) with IORT currently map to MS-DRGs 025, 026, and 027. The requestor suggested that cases reporting a procedure code describing excision of a brain tumor (00B00ZZ or 00B70ZZ) with IORT (D0Y0CZZ or D0Y1CZZ) should map to MS-DRG 023 because of the higher costs associated with the addition of IORT to the excision of brain tumor surgery. According to the requestor, MS-DRG 023 includes complicated craniotomy cases involving the placement of radiological sources and chemotherapy implants. The requestor stated that because IORT involves a full course of radiation therapy delivered directly to the tumor bed via an applicator that is implanted into the tumor cavity during the same surgical session and is clinically similar to two other procedures listed in the Chemotherapy Implant logic list, it should also be included in the Chemotherapy Implant logic list. Specifically, the requestor stated procedure code 00H004Z (Insertion of radioactive element, cesium-131 collagen implant into brain, open approach) and procedure code 3E0Q305 (Introduction of other antineoplastic into cranial cavity and brain, percutaneous approach) also involve the delivery of either radiation or chemotherapy directly after tumor resection. According to the requestor, the resources involved in placing the delivery device are similar for all three procedures and the distinction is that the procedures described by codes 00H004Z and 3E0Q305 involve the insertion of devices that deliver radiation or chemotherapy over a period of time, whereas IORT delivers the entire dose of radiation during the operative session. As such, the requestor asserted that IORT is clinically aligned with the other procedures from a therapeutic and resource utilization perspective.
We noted in the proposed rule that the requestor performed its own analysis using the FY 2022 MedPAR file that was made available in association with the FY 2024 IPPS/LTCH PPS final rule and stated it found fewer than 11 cases reporting IORT in MS-DRGs 025, 026, and 027, with the majority of those cases mapping to MS-DRG 025. According to the requestor, the volume of claims reporting IORT is anticipated to increase as appropriate use of the technology is adopted.
We also noted in the proposed rule that the requestor is correct that currently, the logic for case assignment to MS-DRG 023 includes a Chemotherapy Implant logic list and the procedure codes that identify IORT (D0Y0CZZ and D0Y1CZZ) are not listed in the GROUPER logic and do not affect MS-DRG assignment as the procedures are designated as non-O.R. procedures. The requestor is also correct that cases reporting a procedure code describing excision of a brain tumor (00B00ZZ or 00B70ZZ) with IORT currently map to MS-DRGs 025, 026, and 027. We refer the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1 (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic.
As discussed in the proposed rule, we analyzed claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRGs 023, 024, 025, 026, and 027 and for cases reporting excision of brain tumor and IORT. We identified claims reporting excision of brain tumor with procedure code 00B00ZZ or 00B70ZZ and identified claims reporting IORT with procedure code D0Y0CZZ or D0Y1CZZ. The findings from our analysis are shown in the following table. We note that there were no cases found to report IORT of brain (D0Y0CZZ) or brain stem (D0Y1CZZ) with excision of brain (00B00ZZ) or excision of cerebral hemisphere (00B70ZZ).

As the data show, there were no cases found to report the use of IORT in the performance of a brain tumor excision; therefore, we are unable to evaluate whether the use of IORT directly impacts resource utilization. For this reason, we proposed to maintain the current structure of MS-DRGs 023, 024, ( print page 69014) 025, 026, and 027 for FY 2025. We stated that we would continue to monitor the claims data in consideration of any future modifications to the MS-DRGs for which IORT may be reported.
Comment: Commenters supported the proposal to maintain the current structure of MS-DRGs 023, 024, 025, 026, and 027 for FY 2025.
Response: We appreciate the commenters' support.
After consideration of the public comments we received, we are finalizing our proposal to maintain the current structure of MS-DRGs 023, 024, 025, 026, and 027 for FY 2025.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35956 through 35959 ), we received a request to create a new MS-DRG to better accommodate the costs of concomitant left atrial appendage closure and cardiac ablation for atrial fibrillation in MDC 05 (Diseases and Disorders of the Circulatory System). Atrial fibrillation (AF) is an irregular and often rapid heart rate that occurs when the two upper chambers of the heart experience chaotic electrical signals. AF presents as either paroxysmal (lasting < 7 days), persistent (lasting > 7 day, but less than 1 year), or long standing persistent (chronic)(lasting > 1 year) based on time duration and can increase the risk for stroke, heart failure, and mortality. Management of AF has two primary goals: optimizing cardiac output through rhythm or rate control and decreasing the risk of cerebral and systemic thromboembolism. Among patients with AF, thrombus in the left atrial appendage (LAA) is a primary source for thromboembolism. Left Atrial Appendage Closure (LAAC) is a surgical or minimally invasive procedure to seal off the LAA to reduce the risk of embolic stroke.
According to the requestor, the manufacturer of the WATCHMAN TM Left Atrial Appendage Closure (LAAC) device, patients who are indicated for a LAAC device can also have symptomatic AF. For these patients, performing a cardiac ablation and LAAC procedure at the same time is ideal. Cardiac ablation is a procedure that works by burning or freezing tissue on the inside of the heart to disrupt faulty electrical signals causing the arrhythmia, which can help the heart maintain a normal heart rhythm. In the proposed rule, we noted the requestor highlighted a recent study (Piccini et al. Left atrial appendage occlusion with the WATCHMAN TM FLX and concomitant catheter ablation procedures. Heart Rhythm Society Meeting 2023, May 19, 2023; New Orleans, LA.). According to the requestor, the results of this study indicate that when LAAC is performed concomitantly with cardiac ablation, the outcomes are comparable to patients who have undergone these procedures separately.
As discussed in the proposed rule, the requestor identified the following potential procedure code combination that would comprise a concomitant left atrial appendage closure and cardiac ablation procedure: ICD-10-PCS procedure code 02L73DK (Occlusion of left atrial appendage with intraluminal device, percutaneous approach), that identifies the WATCHMAN TM device, in combination with 02583ZZ (Destruction of conduction mechanism, percutaneous approach). We noted in the proposed rule that the requestor performed its own analysis of this procedure code combination and stated that it found the average costs of cases reporting concomitant left atrial appendage closure and cardiac ablation procedures were consistently higher compared to the average costs of other cases within their respective MS-DRG, which it asserted could limit beneficiary access to these procedures. The requestor asserted that improved Medicare payment for providers who perform these procedures concomitantly would help Medicare patients to gain better access to these lifesaving and quality-improving services and decrease the risk of future readmissions and the need for future procedures.
We reviewed this request and in the proposed rule noted concerns regarding making proposed MS-DRG changes based on a specific, single technology (the WATCHMAN TM Left Atrial Appendage Closure (LAAC) device) identified by only one unique procedure code versus considering proposed changes based on a group of related procedure codes that can be reported to describe the same type or class of technology, which is more consistent with the intent of the MS-DRGs. Therefore, in reviewing this request, in the proposed rule we stated we identified eight additional ICD-10-PCS procedure codes that describe LAAC procedures and included these codes in our analysis. The nine codes we identified are listed in the following table.

Similarly, as noted previously, the requestor identified code 02583ZZ (Destruction of conduction mechanism, percutaneous approach) to describe cardiac ablation. In our review of the ICD-10-PCS classification, in the proposed rule we stated we identified 26 additional ICD-10-PCS codes that describe cardiac ablation that we also examined. The 27 codes we included in our analysis are listed in the following table.

In the ICD-10 MS-DRGs Definitions Manual Version 41.1, for concomitant left atrial appendage closure and cardiac ablation procedures, the GROUPER logic assigns MS-DRGs 273 and 274 (Percutaneous and Other Intracardiac Procedures with and without MCC, respectively) depending on the presence of any additional MCC secondary diagnoses. We stated in the proposed rule that we examined claims data from the September 2023 update of the FY 2023 MedPAR file for all cases in MS-DRGs 273 and 274 and compared the results to cases reporting procedure codes describing concomitant left atrial appendage closure and cardiac ablation. Our findings are shown in the following table.

As shown in the table, in MS-DRG 273, we identified a total of 7,250 cases with an average length of stay of 5.4 days and average costs of $35,197. Of those 7,250 cases, there were 80 cases reporting procedure codes describing concomitant left atrial appendage closure and cardiac ablation with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 273 ($70,447 compared to $35,197) and a slightly longer average length of stay (5.8 days compared to 5.4 days). In MS-DRG 274, we identified a total of 47,801 cases with an average length of stay of 1.4 days and average costs of $29,209. Of those 47,801 cases, there were 781 cases reporting procedure codes describing concomitant left atrial appendage closure and cardiac ablation, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 274 ($66,277 compared to $29,209) and a slightly longer average length of stay (1.5 days compared to 1.4 days).
In the proposed rule we stated we reviewed these data and noted, clinically, the management of AF by performing concomitant left atrial appendage closure and cardiac ablation can improve symptoms, prevent stroke, and reduce the risk of bleeding compared with oral anticoagulants. We stated the data analysis clearly shows that cases reporting concomitant left atrial appendage closure and cardiac ablation procedures have higher average costs and slightly longer lengths of stay compared to all the cases in their assigned MS-DRG. For these reasons, we proposed to create a new MS-DRG for cases reporting a LAAC procedure and a cardiac ablation procedure. ( print page 69016)
As discussed in the proposed rule, to compare and analyze the impact of our suggested modifications, we ran a simulation using the claims data from the September 2023 update of the FY 2023 MedPAR file. The following table illustrates our findings for all 1,723 cases reporting procedure codes describing concomitant left atrial appendage closure and cardiac ablation. We stated we believed the resulting proposed MS-DRG assignment is more clinically homogeneous, coherent and better reflects hospital resource use.

We applied the criteria to create subgroups in a base MS-DRG as discussed in section II.C.1.b. of the FY 2025 IPPS/LTCH PPS proposed rule. As shown in the table that follows, a three-way split of the proposed new MS-DRGs failed the criterion that there be at least 500 cases for each subgroup due to low volume. Specifically, for the “with MCC” split, there were only 268 cases in the subgroup.

We noted that we then applied the criteria for a two-way split for the “with CC/MCC” and “without CC/MCC” subgroups and found that the criterion that there be at least a 20% difference in average cost between subgroups could not be met. The following table illustrates our findings.

We also applied the criteria for a two-way split for the “with MCC” and “without MCC” subgroups and found that the criterion that there be at least 500 or more cases in each subgroup similarly could not be met. The criterion that there be at least a 20% difference in average costs between the subgroups also was not met. The following table illustrates our findings.

Therefore, for FY 2025, we did not propose to subdivide the proposed new MS-DRG for cases reporting procedure codes describing concomitant left atrial appendage closure and cardiac ablation into severity levels.
In summary, for FY 2025, taking into consideration that it clinically requires greater resources to perform concomitant left atrial appendage closure and cardiac ablation procedures, we proposed to create a new base MS-DRG for cases reporting a LAAC procedure and a cardiac ablation procedure in MDC 05. The proposed new MS-DRG is proposed new MS-DRG 317 (Concomitant Left Atrial Appendage Closure and Cardiac Ablation). We also proposed to include the nine ICD-10-PCS procedure codes that describe LAAC procedures and the 27 ICD-10-PCS procedure codes that describe cardiac ablation listed previously in the logic for assignment of cases reporting a LAAC procedure and a cardiac ablation procedure for the proposed new MS-DRG.
Comment: Many commenters expressed support for the proposal to create new base MS-DRG 317 for cases reporting a LAAC procedure and a cardiac ablation procedure in MDC 05. Commenters stated the creation of MS-DRG 317 is timely and will ensure more patients have access to needed care during a single hospital stay, reducing the need for additional admissions. Other commenters agreed that some patients with AF undergoing ablation are also candidates for LAAC procedures and stated combining the procedures is feasible, efficacious, and simple to employ. Several commenters stated that the proposal is a significant step forward to support better disease management for some of the most comorbid patients and likely will reduce downstream healthcare costs. A few commenters specifically stated they appreciate CMS' continued evaluation and acknowledgement of the increased resources required for patients requiring multiple procedures during a single inpatient hospitalization. While supporting the proposal to create MS-DRG 317, some commenters suggested that CMS devise a broader, more inclusive, supplemental payment mechanism to facilitate incremental payment when two major procedures are performed during the same hospital admission.
Response: We appreciate the commenters' support and the feedback regarding payment when two major ( print page 69017) procedures are performed during the same hospital admission.
Comment: Another commenter recommended that CMS delay creation of proposed new MS-DRG 317 for concomitant LAAC and cardiac ablation. While expressing support for proposals that will improve patient outcomes and increase efficiencies in the health care system, the commenter stated they believe it is premature for CMS to develop a new MS-DRG at this time. The commenter expressed concern that the evidence to support the safety, effectiveness, and workflow of these two procedures when performed concomitantly has not been well established and suggested that the results of two ongoing randomized control trials (RCTs) focusing on LAAC and ablation should be considered before CMS moves forward to develop a new MS-DRG.
Response: We thank the commenter for their feedback. In response to the suggestion that CMS delay implementation of proposed new MS-DRG 317 for concomitant LAAC and cardiac ablation, we reviewed the commenters' concern and do not agree that a delay is necessary or appropriate. As stated earlier, the data analysis clearly shows that cases reporting concomitant LAAC and cardiac ablation procedures have higher average costs and slightly longer lengths of stay compared to all the cases in their assigned MS-DRG. For these reasons, we proposed to create a new MS-DRG for cases reporting a LAAC procedure and a cardiac ablation procedure. We will continue to monitor the claims data and perform additional analysis if any evidence is presented to us regarding the clinical efficacy of concomitant left atrial appendage closure and cardiac ablation procedures. We would address any modifications to the logic in future rulemaking.
Comment: Other commenters noted a difference in case volume between the table CMS stated reflected the cases reporting procedure codes describing concomitant LAAC and cardiac ablation in MS-DRGs 273 and 274 and the table which CMS stated illustrated the findings for all cases reporting procedure codes describing concomitant LAAC and cardiac ablation found in the claims data from the September 2023 update of the FY 2023 MedPAR file. Specifically, the commenters noted that 861 cases reporting procedure codes describing concomitant LAAC and cardiac ablation were found in MS-DRGs 273 and 274, while 1,723 cases reporting procedure codes describing concomitant LAAC and cardiac ablation were found in the simulation using the claims data from the September 2023 update of the FY 2023 MedPAR file. The commenters stated it is unclear from the tables and data associated with the proposed rule where the additional 862 cases are currently assigned. These commenters performed their own analysis of the supplemental After Outliers Removed (AOR)/Before Outliers Removed (BOR) file available in association with the FY 2025 IPPS/LTCH PPS proposed rule on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps and recommended that CMS consider that cases reporting procedure codes describing concomitant LAAC and cardiac ablation group to other MS-DRGs, and should be incorporated into the analysis based on volume differences they noted in the AOR/BOR file.
Response: We thank the commenters for their feedback.
In response to suggestion that CMS provide insight regarding the difference in case volume between the table which we stated reflects our examination of the claims data from the September 2023 update of the FY 2023 MedPAR file for all cases in MS-DRGs 273 and 274, compared to the results for cases reporting procedure codes describing concomitant LAAC and cardiac ablation in those MS-DRGs, and the table which we stated illustrated our findings for all 1,723 cases reporting procedure codes describing concomitant LAAC and cardiac ablation, we note that as stated in the proposed rule, for concomitant LAAC and cardiac ablation procedures, the GROUPER logic assigns MS-DRGs 273 or 274 (Percutaneous and Other Intracardiac Procedures with or without MCC, respectively) depending on the presence of any additional MCC secondary diagnoses. Therefore, we focused our examination of claims data from the September 2023 update of the FY 2023 MedPAR file for all cases in MS-DRGs 273 and 274 and compared the results to cases reporting procedure codes describing concomitant LAAC and cardiac ablation.
While not explicitly stated, assignment to MS-DRGs 273 or 274 is also dependent on the absence of other procedure codes that could affect MS-DRG assignment on the claim. If other procedure codes that could affect MS-DRG assignment are also reported on the claim along with procedure codes describing concomitant left atrial appendage closure and ablation, the MS-DRG assignment can vary depending on the procedure codes reported.
As discussed in section II.C.14. of the preamble of the proposed rule and this final rule, in our proposal to revise the surgical hierarchy for the MS-DRGs in MDC 05, we proposed to sequence proposed new MS-DRG 317 (Concomitant Left Atrial Appendage Closure and Cardiac Ablation) above MS-DRG 275 (Cardiac Defibrillator Implant with Cardiac Catheterization and MCC) and below MS-DRGs 231, 232, 233, 234, 235, and 236 (Coronary Bypass with or without PTCA, with or without Cardiac Catheterization or Open Ablation, with and without MCC, respectively). Under this proposal, if procedure codes describing concomitant LAAC and cardiac ablation are reported, the GROUPER logic would assign new MS-DRG 317 in the absence of other procedure codes that could affect MS-DRG assignment to an MS-DRG that would be sequenced higher in the surgical hierarchy than MS-DRG 317 in MDC 05. The table which we stated illustrated our findings for all 1,723 cases reporting procedure codes describing concomitant LAAC and cardiac ablation includes cases that are anticipated to potentially shift or be redistributed as a result of the proposal to 1) create a new base MS-DRG 317 and 2) the proposal to sequence the new MS-DRG above MS-DRG 275 and below MS-DRGs 231, 232, 233, 234, 235, and 236 in MDC 05.
To illustrate these shifts for this final rule, we again analyzed the September 2023 update of the FY 2023 MedPAR file for cases reporting procedure codes describing concomitant LAAC and cardiac ablation. We then examined the redistribution of cases that is anticipated to occur as a result of the proposal to create a new base MS-DRG 317 by processing the claims data from the September 2023 update of the FY 2023 MedPAR file through the ICD-10 MS-DRG GROUPER Version 41 and then processing the same claims data through the ICD-10 MS-DRG GROUPER Version 42 for comparison. The number of cases from this comparison that result in different MS-DRG assignments is the number of the cases that are anticipated to potentially shift or be redistributed. Our findings are shown in the following table.

As stated in the proposed rule and reflected in the previous table, we found 1,723 cases reporting procedure codes describing concomitant LAAC and cardiac ablation that are anticipated to potentially shift or be redistributed into MS-DRG 317. The largest number of cases moving into new MS-DRG 317 are moving out of MS-DRGs 274, 229, 228 and 273. In response to the suggestion that CMS incorporate other MS-DRGs into our analysis, we examined the claims data from the September 2023 update of the FY 2023 MedPAR file to identify the average length of stay and average costs for all cases in MS-DRGs 228, 229, 242, 243, 244, 245, 267, 268, 269, 270, 271, 272, 273, 274, 275, 276, 277, 319, and 320. Our findings are shown in the following table.

In reviewing the data analysis performed, the 1,723 cases anticipated to potentially shift or be redistributed into MS-DRG 317 have higher average costs when compared to all the cases in MS-DRGs 228, 229, 273, and 274 ($54,629 versus $44,565, $28,987, $35,197, and $29,209, respectively). The 1,723 cases anticipated to potentially shift or be redistributed into MS-DRG 317 have an average length of stay that is shorter than the average length of stay for all the cases in MS-DRGs 228, 229, and 273 (3.1 days versus 8.7 days, 3.3 days, and 5.4 days, respectively) and a longer average length of stay when compared to all the cases in MS-DRG 274 (3.1 days versus 1.4 days). We note that the 1,723 cases anticipated to potentially shift or be redistributed into MS-DRG 317 have lower average costs when compared to all the cases in MS-DRGs 275, 268, and 276 ($54,629 versus $63,181, $59,383, and $54, 993, respectively), however only seven cases reported procedure codes describing concomitant left atrial appendage closure and cardiac ablation in these MS-DRGs. We also note that the 1,723 cases anticipated to potentially shift or be redistributed into MS-DRG 317 have a longer average length of stay when compared to all the cases in MS-DRGs 244, 272, 269, and 267 (3.1 days versus 2.5 days, 2.3 days, 2 days, and 1.5 days, respectively), however only 20 cases reported procedure codes describing concomitant left atrial appendage closure and cardiac ablation in these MS-DRGs. We reviewed these data and believed the proposal to create new base MS-DRG 317 for cases reporting procedure codes describing concomitant LAAC and cardiac ablation in MDC 05 and the proposed revision to the surgical hierarchy leads to a grouping that is more coherent and better reflects the clinical severity and resource use involved in these cases.
Comment: In reviewing the list of nine ICD-10-PCS procedure codes that describe LAAC procedures that were proposed to be included in the logic for assignment of cases reporting procedure codes describing concomitant LAAC and cardiac ablation for the proposed new MS-DRG, a commenter noted these nine codes are designated as non-O.R. procedures affecting the MS-DRG. The commenter also noted that codes 02570ZK (Destruction of left atrial appendage, open approach), 02573ZK (Destruction of left atrial appendage, percutaneous approach), and 02574ZK (Destruction of left atrial appendage, percutaneous endoscopic approach) included in the list of 27 ICD-10-PCS procedure codes that describe cardiac ablation proposed to be included in the logic for assignment to the proposed new MS-DRG, are also designated as non-O.R. procedures affecting the MS-DRG. The commenter stated that LAAC procedures and cardiac ablation procedures performed by an open or percutaneous endoscopic approach should be designated as operating room procedures to account for the resource utilization required to perform them as these procedures require the use of specialized equipment or devices.
Response: We thank the commenter for their feedback.
We agree that in the ICD-10 MS-DRGs Definitions Manual Version 41.1, the nine ICD-10-PCS procedure codes that describe LAAC procedures are recognized as non-O.R. procedures affecting the MS-DRGs to which they are assigned. We refer the reader to Section II.C.10 in the proposed rule and this final rule for the complete discussion of the designations each ICD-10-PCS code has under the IPPS MS-DRGs that determine whether and in what way the presence of that procedure code on a claim impacts the MS-DRG assignment. In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44898 through 44899 ) we reviewed these nine ICD-10-PCS procedure codes that ( print page 69020) describe LAAC procedures and stated we believe the current designation of LAAC procedures as non-O.R. procedures that affect the assignment for MS-DRGs 273 and 274 is clinically appropriate to account for the subset of patients undergoing left atrial appendage closure specifically. We further stated that we believed that circumstances in which a patient is admitted for a principal diagnosis outside of MDC 05 and a left atrial appendage closure is performed as the only surgical procedure in the same admission are infrequent, and if they do occur, the LAAC procedure would not be a significant contributing factor in the increased intensity of resources needed for facilities to manage these complex cases.
We continue to believe that circumstances in which a patient is admitted for a principal diagnosis outside of MDC 05 and LAAC is performed as the only surgical procedure in the same admission are infrequent, and that the current designation of LAAC procedures as non-O.R. procedures that affect the assignment for MS-DRGs 273 and 274, and now MS-DRG 317, is clinically appropriate to account for the subset of patients undergoing left atrial appendage closure specifically.
Similarly, we agree that in the ICD-10 MS-DRGs Definitions Manual Version 41.1, procedure codes 02570ZK, 02573ZK, and 02574ZK are recognized as non-O.R. procedures affecting the MS-DRGs as reflected in the following table, specifically.

We believe that circumstances in which a patient is admitted for a principal diagnosis outside of MDC 05 and a cardiac ablation is performed as the only surgical procedure in the same admission are infrequent, and that the current designation of 02570ZK, 02573ZK, and 02574ZK as non-O.R. procedures that affect the assignment for the MS-DRGs reflected in the previous table, and now MS-DRG 317, is clinically appropriate to account for the subset of patients undergoing cardiac ablation specifically.
Comment: A commenter suggested that ICD-10-PCS codes 02590ZZ (Destruction of chordae tendineae, open approach), 02593ZZ (Destruction of chordae tendineae, percutaneous approach), and 02594ZZ (Destruction of chordae tendineae, percutaneous endoscopic approach) describing ablation of the chordae tendineae be removed from the list of cardiac ablation procedures for MS-DRG 317 as the chordae tendineae would not be ablated in relation to cardiac ablation procedures, and instead they would be ablated in relation to cardiac valve repair or replacement procedures.
Response: We appreciate the feedback from the commenter.
As noted previously, atrial fibrillation (AF) is an irregular and often rapid heart rate that occurs when the two upper chambers of the heart experience chaotic electrical signals. Cardiac ablation is a procedure that is performed to correct a disturbance in the conduction system of the heart by damaging small areas of tissue using radiofrequency energy or freezing so that the damaged tissue can no longer generate or conduct electrical impulses. We agree that ablation of the chordae tendineae, which are the strong, fibrous connections between the valve leaflets and the papillary muscles, is not performed to stop abnormal electrical pathways as the cardiac conduction system does not pass through the chordae tendineae.
We examined claims data from the September 2023 update of the FY 2023 MedPAR file to evaluate the frequency with which ablation of the chordae tendineae is reported with left atrial appendage closure, and found one case reporting procedure codes 02590ZZ (Destruction of chordae tendineae, open approach) and 02L70ZK (Occlusion of left atrial appendage, open approach) in MS-DRG 219 (Cardiac Valve and Other Major Cardiothoracic Procedures without Cardiac Catheterization with MCC) with a length of stay of 7 days and costs of $28,989.
We note that assignment of this one case to MS-DRG 219 indicates that other procedure code(s) assigned to the GROUPER logic of MS-DRG 219 were reported in addition to procedure codes 02590ZZ and 02L70ZK to drive assignment to this MS-DRG. We reviewed these data and because our analysis identified only one case reporting ablation of the chordae tendineae and left atrial appendage closure, and recognizing that ablation of the chordae tendineae is not performed to stop abnormal electrical pathways, we agree that procedure codes 02590ZZ, 02593ZZ, and 02594ZZ should be removed from the list of 27 ICD-10-PCS procedure codes that describe cardiac ablation listed previously in the proposed logic for assignment of cases reporting a LAAC procedure and a cardiac ablation procedure for the proposed new MS-DRG.
Comment: Many commenters noted that procedure code 02583ZF (Destruction of conduction mechanism using irreversible electroporation, percutaneous approach) to identify irreversible electroporation for cardiac ablation was finalized effective April 1, 2024 as reflected in the FY 2024 ICD-10-PCS Code Update files that were made publicly available on the CMS website at https://www.cms.gov/Medicare/Coding/ICD10 on December 19, 2023. The new procedure code is also reflected in Table 6B.—New Procedure Codes, in association with the proposed rule and available on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS , including the MS-DRG assignments for the new code for FY 2025.
These commenters noted that cardiac ablation procedures performed with the PulseSelect TM Pulsed Field Ablation (PFA) System for the treatment of paroxysmal (PAF) or persistent (PsAF) atrial fibrillation can also be performed concomitantly with left atrial appendage closure and recommended that procedure code 02583ZF also be assigned to new MS-DRG 317 (Concomitant Left Atrial Appendage Closure and Cardiac Ablation) in MDC 05. However, another commenter noted that there is limited data on combining the new pulse field ablation modality with LAAC and suggested that CMS continue to evaluate evidence on the safety and efficacy of using this new modality in concomitant procedures ( print page 69021) before assigning procedure code 02583ZF to new MS-DRG 317.
We note that irreversible electroporation for cardiac ablation, also referred to as pulsed field ablation, delivers electrical pulses that result in destruction of selected cardiac tissue by irreversibly increasing the porosity of the cell membranes, inducing cell death, and can be used as a treatment for paroxysmal and persistent atrial fibrillation. As a procedure code that also describes the performance of cardiac ablation, we agree that procedure code 02583ZF should be added to the list of ICD-10-PCS procedure codes that describe cardiac ablation listed previously in the proposed logic for assignment of cases reporting a LAAC procedure and a cardiac ablation procedure for the proposed new MS-DRG. In response to the suggestion that CMS continue to evaluate evidence on the safety and efficacy of using this new modality in concomitant procedures before assigning procedure code 02583ZF to new MS-DRG 317, we note that procedure code 02583ZF describes a procedure that is clinically coherent with the other procedure codes proposed for assignment to MS-DRG 317, so it is reasonable that cases reporting procedure code 02583ZF and a procedure code describing LAAC group to the same MS-DRG.
Therefore, after consideration of the public comments we received, and for the reasons discussed, we are finalizing our proposal to create new MS-DRG 317 (Concomitant Left Atrial Appendage Closure and Cardiac Ablation) in MDC 05, with modification, effective October 1, 2024, for FY 2025. Specifically, we are modifying the proposed list of ICD-10-PCS procedure codes that describe cardiac ablation in the Version 42 GROUPER logic of new MS-DRG 317 by removing ICD-10-PCS codes 02590ZZ (Destruction of chordae tendineae, open approach), 02593ZZ (Destruction of chordae tendineae, percutaneous approach), and 02594ZZ (Destruction of chordae tendineae, percutaneous endoscopic approach) and adding ICD-10-PCS procedure code 02583ZF (Destruction of conduction mechanism using irreversible electroporation, percutaneous approach), as discussed previously.
The 25 ICD-10-PCS procedure codes that describe cardiac ablation that we are finalizing in the logic for assignment of cases reporting a LAAC procedure and a cardiac ablation procedure for FY 2025 are listed in the following table. This assignment is reflected in the final Version 42 GROUPER logic.

Table 6B.—New Procedure Codes, associated with this final rule reflects the modification to the MS-DRG assignments for procedure code 02583ZF for FY 2025. We refer the reader to section II.C.13. of the preamble of this final rule for further information regarding the table.
Lastly, we are finalizing the inclusion of the nine ICD-10-PCS procedure codes that describe LAAC procedures listed previously in the logic for assignment of cases reporting a LAAC procedure and a cardiac ablation procedure for new MS-DRG 317, without modification, for FY 2025. We refer the reader to section II.C.15. of the preamble of this final rule for the discussion of the surgical hierarchy and the complete list of our proposed modifications to the surgical hierarchy as well as our finalization of those proposals.
The BAROSTIM TM system is the first neuromodulation device system designed to trigger the body's main cardiovascular reflex to target symptoms of heart failure. The system consists of an implantable pulse generator (IPG) that is implanted subcutaneously in the upper chest below the clavicle, a stimulation lead that is sutured to either ( print page 69022) the right or left carotid sinus to activate the baroreceptors in the wall of the carotid artery, and a wireless programmer system that is used to non-invasively program and adjust BAROSTIM TM therapy via telemetry. The BAROSTIM TM system is indicated for the improvement of symptoms of heart failure in a subset of patients with symptomatic New York Heart Association (NYHA) Class III or Class II (who had a recent history of Class III) heart failure, with a low left ventricular ejection fraction, who also do not benefit from guideline directed pharmacologic therapy or qualify for Cardiac Resynchronization Therapy (CRT). The BAROSTIM TM system was approved for new technology add-on payments for FY 2021 ( 85 FR 58716 through 58717 ) and FY 2022 ( 86 FR 44974 ). The new technology add-on payment was subsequently discontinued effective FY 2023 ( 87 FR 48916 ).
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48837 through 48843 ), we discussed a request we received to reassign the ICD-10-PCS procedure codes that describe the implantation of the BAROSTIM TM system from MS-DRGs 252, 253, and 254 (Other Vascular Procedures with MCC, with CC, and without MCC respectively) to MS-DRGs 222, 223, 224, 225, 226, and 227 (Cardiac Defibrillator Implant with and without Cardiac Catheterization with and without AMI/HF/Shock with and without MCC, respectively). The requestor stated that the subset of patients that have an indication for the implantation of a BAROSTIM TM system also have indications for the implantation of Implantable Cardioverter Defibrillators (ICD), Cardiac Resynchronization Therapy Defibrillators (CRT-D) and/or Cardiac Contractility Modulation (CCM) devices, all of which also require the permanent implantation of a programmable, electrical pulse generator and at least one electrical lead. The requestor further stated that the average resource utilization required to implant the BAROSTIM TM system demonstrates a significant disparity compared to all procedures within MS-DRGs 252, 253, and 254.
In the FY 2023 IPPS/LTCH PPS final rule, we stated that the results of the claims analysis demonstrated we did not have sufficient claims data on which to base and evaluate any proposed changes to the current MS-DRG assignment. We also expressed concern in equating the implantation of a BAROSTIM TM system to the placement of ICD, CRT-D, and CCM devices as these devices all differ in terms of technical complexity and anatomical placement of the electrical lead(s). We noted there is no intravascular component or vascular puncture involved when implanting a BAROSTIM TM system. In contrast, the placement of ICD, CRT-D, and CCM devices generally involve a lead being affixed to the myocardium, being threaded through the coronary sinus or crossing a heart valve and are procedures that involve a greater level of complexity than affixing the stimulator lead to either the right or left carotid sinus when implanting a BAROSTIM TM system. We stated that we believed that as the number of cases reporting procedure codes describing the implantation of neuromodulation devices for heart failure increases, a better view of the associated costs and lengths of stay on average will be reflected in the data for purposes of assessing any reassignment of these cases. Therefore, after consideration of the public comments we received, and for the reasons stated earlier, we finalized our proposal to maintain the assignment of cases reporting procedure codes that describe the implantation of a neuromodulation device in MS-DRGs 252, 253, and 254 for FY 2023.
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58712 through 58720 ), we discussed a request we received to add ICD-10-CM diagnosis code R57.0 (Cardiogenic shock) to the list of “secondary diagnoses” that grouped to MS-DRGs 222 and 223 (Cardiac Defibrillator Implant with Cardiac Catheterization with Acute Myocardial Infarction (AMI), Heart Failure (HF), or Shock with and without MCC, respectively). During our review of the issue, we noted that the results of our claims analysis showed that in procedures involving a cardiac defibrillator implant, the average costs and length of stay were generally similar without regard to the presence of diagnosis codes describing AMI, HF, or shock. We stated we believed that it may no longer be necessary to subdivide MS-DRGs 222, 223, 224, 225, 226, and 227 based on the diagnosis codes reported. After consideration of the public comments we received, and for the reasons stated in the rule, we finalized our proposal to delete MS-DRGs 222, 223, 224, 225, 226, and 227. We also finalized our proposal to create new MS-DRG 275 (Cardiac Defibrillator Implant with Cardiac Catheterization and MCC), new MS-DRG 276 (Cardiac Defibrillator Implant with MCC) and new MS-DRG 277 (Cardiac Defibrillator Implant without MCC) in MDC 05 for FY 2024.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35959 through 35962 ), we received a similar request to again review the MS-DRG assignment of the ICD-10-PCS procedure codes that describe the implantation of the BAROSTIM TM system. Specifically, the requestor recommended that CMS consider reassigning the ICD-10-PCS procedure codes that describe the implantation of the BAROSTIM TM system from MS-DRGs 252, 253, and 254 (Other Vascular Procedures with MCC, with CC, and without MCC respectively) to MS-DRGs 275 (Cardiac Defibrillator Implant with Cardiac Catheterization and MCC), MS-DRG 276, and 277 (Cardiac Defibrillator Implant with MCC and without MCC respectively); or to other more clinically coherent MS-DRGs for implantable device procedures indicated for Class III heart failure patients. The requestor stated in their analysis the number of claims reporting procedure codes that describe the implantation of the BAROSTIM TM system has been consistently growing over the past few years. The requestor acknowledged that the implantation of the BAROSTIM TM system is predominantly performed in the outpatient setting but noted that a significant number of severely sick patients with multiple comorbidities (such as chronic kidney disease, end stage renal disease (ESRD), chronic obstructive pulmonary disease (COPD), and AF) are treated in an inpatient setting. The requestor stated in their experience, hospitals that have performed BAROSTIM TM procedures have stopped allowing patients to receive the device in the inpatient setting due to the high losses for each Medicare claim. The requestor asserted it is critically important to allow very sick and fragile patients access to the BAROSTIM TM procedure in an inpatient setting and stated these patients should not be denied access by hospitals due to the perceived gross underpayment of the current MS-DRG.
In the proposed rule we noted that the requestor stated the BAROSTIM TM procedure is not clinically coherent with other procedures assigned to MS-DRGs 252, 253, and 254 (Other Vascular Procedures) as the majority of the ICD-10-PCS codes assigned to MS-DRGs 252, 253, and 254 describe procedures to identify, diagnose, clear and restructure veins and arteries, excluding those that require implantable devices. Furthermore, the requestor stated the costs of the implantable medical devices used for the BAROSTIM TM system (that is, the electrical pulse generator and electrical lead) alone far exceed the ( print page 69023) average costs of other cases assigned to MS-DRGs 252, 253, and 254.
The following ICD-10-PCS procedure codes uniquely identify the implantation of the BAROSTIM TM system: 0JH60MZ (Insertion of stimulator generator into chest subcutaneous tissue and fascia, open approach) in combination with 03HK3MZ (Insertion of stimulator lead into right internal carotid artery, percutaneous approach) or 03HL3MZ (Insertion of stimulator lead into left internal carotid artery, percutaneous approach).
We stated in the proposed rule that to analyze this request, we first examined claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRGs 252, 253, and 254 to identify cases reporting procedure codes describing the implantation of the BAROSTIM TM system with or without a procedure code describing the performance of a cardiac catheterization as MS-DRG 275 is defined by the performance of cardiac catheterization and a secondary diagnosis of MCC. Our findings are shown in the following table.

As shown in the table, in MS-DRG 252, we identified a total of 18,964 cases with an average length of stay of 8 days and average costs of $30,456. Of those 18,964 cases, there was one case reporting procedure codes describing the implantation of the BAROSTIM TM system with a procedure code describing the performance of a cardiac catheterization with costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 252 ($110,928 compared to $30,456) and a longer length of stay (9 days compared to 8 days). There were 12 cases reporting procedure codes describing the implantation of the BAROSTIM TM system without a procedure code describing the performance of a cardiac catheterization, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 252 ($66,291 compared to $30,456) and a slighter shorter average length of stay (7.8 days compared to 8 days). In MS-DRG 253, we identified a total of 15,551 cases with an average length of stay of 5.2 days and average costs of $22,870. Of those 15,551 cases, there were seven cases reporting procedure codes describing the implantation of the BAROSTIM TM system without a procedure code describing the performance of a cardiac catheterization, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 253 ($52,788 compared to $22,870) and a shorter average length of stay (4 days compared to 5.2 days). We found zero cases in MS-DRG 253 reporting procedure codes describing the implantation of a BAROSTIM TM system with a procedure code describing the performance of a cardiac catheterization. In MS-DRG 254, we identified a total of 5,973 cases with an average length of stay of 2.3 days and average costs of $15,778. Of those 5,973 cases, there were three cases reporting procedure codes describing the implantation of the BAROSTIM TM system without a procedure code describing the performance of a cardiac catheterization, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 254 ($29,740 compared to $15,778) and a shorter average length of stay (1.3 days compared to 2.3 days). We found zero cases in MS-DRG 254 reporting procedure codes describing the implantation of a BAROSTIM TM system with a procedure code describing the performance of a cardiac catheterization.
As stated in the proposed rule, we then examined claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRGs 275, 276, and 277. Our findings are shown in the following table.

As the table shows, for MS-DRG 275, there were a total of 3,358 cases with an average length of stay of 10.3 days and average costs of $63,181. For MS-DRG 276, there were a total of 3,264 cases with an average length of stay of 8.2 days and average costs of $54,993. For MS-DRG 277, there were a total of 3,840 cases with an average length of stay of 4.2 days and average costs of $42,111.
In exploring mechanisms to address this request, in the proposed rule we noted in total, there were only 23 cases reporting procedure codes describing the implantation of a BAROSTIM TM system in MS-DRGs 252, 253, and 254 (13, 7, and 3, respectively). We stated we reviewed these data, and stated while we recognize that the average costs of the 23 cases reporting procedure codes describing the implantation of a BAROSTIM TM are greater when compared to the average costs of all cases in MS-DRGs 252, 253, and 254, the number of cases continued to be too small to warrant the creation of a new MS-DRG for these cases.
In the proposed rule we further noted, that of the 23 cases reporting procedure codes describing the implantation of a BAROSTIM TM system identified in MS-DRGs 252, 253, and 254, only one case reported the performance of cardiac catheterization. As discussed in the FY 2024 IPPS/LTCH PPS final rule, when reviewing the consumption of hospital resources for the cases reporting a cardiac defibrillator implant with cardiac catheterization during a hospital stay, the claims data clearly showed that the cases reporting secondary diagnoses designated as MCCs were more resource intensive as compared to other cases reporting cardiac defibrillator implant. Therefore, we finalized the creation of MS-DRG 275 for cases reporting a cardiac defibrillator implant with cardiac catheterization and a secondary diagnosis designated as an MCC. In the proposed rule we stated that of the 23 cases reporting procedure codes describing the implantation of a BAROSTIM TM system, there was only one case reporting a procedure code describing the performance of cardiac catheterization and a secondary diagnosis designated as an MCC, and we noted that there may have been other factors contributing to the higher costs of this one case. We stated that the results of the claims analysis demonstrated we did not have sufficient claims data on which to base and propose a change to the current MS-DRG assignment of cases reporting procedure codes describing the implantation of a BAROSTIM TM system from MS-DRGs 252, 253, and 254 to MS-DRG 275.
As stated in the proposed rule, further analysis of the claims data demonstrated that the 23 cases reporting procedure codes describing the implantation of a BAROSTIM TM system had an average length of stay of 5.8 days and average costs of $59,355, as compared to the 3,264 cases in MS-DRG 276 that had an average length of stay of 8.2 days and average costs of $54,993. We stated that while the cases reporting procedure codes describing the implantation of a BAROSTIM TM system had average costs that were $4,362 higher than the average costs of all cases in MS-DRG 276, as noted, there were only a total of 23 cases, and there may have been other factors contributing to the higher costs. In the proposed rule we noted, however, if we were to reassign all cases reporting procedure codes describing the implantation of a BAROSTIM TM system to MS-DRG 276, even if there is not a MCC present, the cases would receive higher payment and the reassignment could better account for the differences in resource utilization of these cases than in their respective MS-DRG.
In the proposed rule we stated we reviewed the clinical issues and the claims data, and while we continue to note that there is no intravascular component or vascular puncture involved when implanting a BAROSTIM TM system, and that the implantation of a BAROSTIM TM system is distinguishable from the placement of ICD, CRT-D, and CCM devices, as these devices all differ in terms of technical complexity and anatomical placement of the electrical lead(s), as discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48837 through 48843 ), we agreed that ICD, CRT-D, and CCM devices and the BAROSTIM TM system are clinically coherent in that they share an indication of heart failure, a major cause of morbidity and mortality in the United States, and that these cases demonstrate comparable resource utilization. Based on our review of the clinical issues and the claims data, and to better account for the resources required, we proposed to reassign the cases reporting procedure codes describing the implantation of a BAROSTIM TM system to MS-DRG 276, even if there is no MCC reported, to better reflect the clinical severity and resource use involved in these cases.
Therefore, for FY 2025, we proposed to reassign all cases with one of the following ICD-10-PCS code combinations capturing cases reporting procedure codes describing the implantation of a BAROSTIM TM system, to MS-DRG 276, even if there is no MCC reported:
- 0JH60MZ (Insertion of stimulator generator into chest subcutaneous tissue and fascia, open approach) in combination with 03HK3MZ (Insertion of stimulator lead into right internal carotid artery, percutaneous approach); and
- 0JH60MZ (Insertion of stimulator generator into chest subcutaneous tissue and fascia, open approach) in combination with 03HL3MZ (Insertion of stimulator lead into left internal carotid artery, percutaneous approach).
We also proposed to change the title of MS-DRG 276 from “Cardiac Defibrillator Implant with MCC” to “Cardiac Defibrillator Implant with MCC or Carotid Sinus Neurostimulator” to reflect the proposed modifications to MS-DRG assignments. We refer the reader to section II.C.15. of the preamble of this final rule for the discussion of the surgical hierarchy and the complete list of our proposed modifications to the surgical hierarchy as well as our finalization of those proposals.
Comment: Commenters expressed overwhelming support for the proposal to reassign cases reporting a procedure code combination describing the implantation of a BAROSTIM TM system to MS-DRG 276, even if there is no MCC reported. Commenters also expressed support for the proposal to change the title of MS-DRG 276 from “Cardiac Defibrillator Implant with MCC” to “Cardiac Defibrillator Implant with MCC or Carotid Sinus Neurostimulator” to reflect the proposed modifications to MS-DRG assignments. Many commenters stated this reassignment would ensure continued access of this very important therapy to eligible Medicare patients. A commenter specifically stated that assignment to MS-DRG 276 is appropriate on a clinical basis and would also better account for the differences in resource ( print page 69025) utilization of these cases as compared to their current assignments.
After consideration of the public comments we received, we are finalizing our proposal to reassign cases reporting one of the previously listed ICD-10-PCS code combinations describing the implantation of a BAROSTIM TM system to MS-DRG 276, even if there is no MCC reported, without modification, effective October 1, 2024, for FY 2025. We are also finalizing the change to the title of MS-DRG 276 from “Cardiac Defibrillator Implant with MCC” to “Cardiac Defibrillator Implant with MCC or Carotid Sinus Neurostimulator” to reflect the modifications to MS-DRG assignments.
The human heart contains four major valves—the aortic, mitral, pulmonary, and tricuspid valves. These valves function to keep blood flowing through the heart. When conditions such as stenosis or insufficiency/regurgitation occur in one or more of these valves, valvular heart disease may result. Intervention options, including surgical aortic valve replacement or transcatheter aortic valve replacement can be performed to treat diseased or damaged aortic heart valves. Surgical aortic valve replacement (SAVR) is a traditional, open-chest surgery where an incision is made to access the heart. The damaged valve is replaced, and the chest is surgically closed. Since SAVR is a major surgery that involves an incision, recovery time tends to be longer. Transcatheter aortic valve replacement (TAVR) is a minimally invasive procedure that involves a catheter being inserted into an artery, without an incision for most cases, and then guided to the heart. The catheter delivers the new valve without the need for the chest or heart to be surgically opened. Since TAVR is a non-surgical procedure, it is generally associated with a much shorter recovery time.
In the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 49892 through 49893 ), we discussed a request we received to create a new MS-DRG that would only include the various types of cardiac valve replacements performed by an endovascular or transcatheter technique. We reviewed the claims data and stated the data analysis showed that cardiac valve replacements performed by an endovascular or transcatheter technique had a shorter average length of stay and higher average costs in comparison to all of the cases in their assigned MS-DRGs, which were MS-DRGs 216, 217, 218, 219, 220, and 221 (Cardiac Valve & Other Major Cardiothoracic Procedure with and without Cardiac Catheterization, with MCC, with CC, and without CC/MCC, respectively). In the FY 2015 IPPS/LTCH PPS final rule we stated that patients receiving endovascular cardiac valve replacements were significantly different from those patients who undergo an open chest cardiac valve replacement and noted that patients receiving endovascular cardiac valve replacements are not eligible for open chest cardiac valve procedures because of a variety of health constraints, which we stated highlights the fact that peri-operative complications and post-operative morbidity have significantly different profiles for open chest procedures compared with endovascular interventions. We further noted that separately grouping these endovascular valve replacement procedures provides greater clinical cohesion for this subset of high-risk patients. Therefore, we finalized our proposal to create MS-DRGs 266 and 267 (Endovascular Cardiac Valve Replacement, with MCC and without MCC, respectively) for FY 2015.
In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42080 through 42089 ), we discussed a request we received to modify the MS-DRG assignment for transcatheter mitral valve repair (TMVR) with implant procedures. We reviewed the claims data and stated based on our data analysis, transcatheter cardiac valve repair procedures and transcatheter (endovascular) cardiac valve replacement procedures are more clinically coherent in that they describe endovascular cardiac valve interventions with implants and were similar in terms of average length of stay and average costs to cases in MS-DRGs 266 and 267 when compared to other procedures in their current MS-DRG assignment. For the reasons described in the rule and after consideration of the public comments we received, we finalized our proposal to modify the structure of MS-DRGs 266 and 267 by reassigning the procedure codes that describe transcatheter cardiac valve repair (supplement) procedures, to revise the title of MS-DRG 266 from “Endovascular Cardiac Valve Replacement with MCC” to “Endovascular Cardiac Valve Replacement and Supplement Procedures with MCC” and to revise the title of MS-DRG 267 from “Endovascular Cardiac Valve Replacement without MCC” to “Endovascular Cardiac Valve Replacement and Supplement Procedures without MCC”, to reflect the finalized restructuring.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35962 through 35966 ), we received a request to delete MS-DRGs 266 and 267 and to move the cases reporting transcatheter aortic valve replacement or repair (supplement) procedures currently assigned to those MS-DRGs into MS-DRGs 216, 217, 218, 219, 220, and 221. The requestor asserted that under the current IPPS payment methodology, TAVR procedures are not profitable to hospitals and when patients are clinically eligible for both a TAVR and SAVR procedures, factors beyond clinical appropriateness can drive treatment decisions. According to the requestor (the manufacturer of the SAPIEN TM family of transcatheter heart valves) sharing a single set of MS-DRGs would eliminate the current disincentives hospitals face and create financial neutrality between the two lifesaving treatment options. The requestor stated the current disincentives are increasingly problematic because they contribute to treatment disparities among certain racial, socioeconomic, and geographic groups.
As discussed in the proposed rule, the requestor noted that currently surgical cardiac valve replacement and supplement procedures, such as SAVR, are assigned to MS-DRGs 216, 217, 218, 219, 220, and 221, and endovascular cardiac valve replacement and supplement procedures, such as TAVR, are assigned to MS-DRGs 266 and 267. The requestor stated that both sets of MS-DRGs address valve disease and include valve repair or replacement procedures for any of the four heart valves. According to the requestor, while the sets of MS-DRGs involve clinically similar cases their payment rates differ which may be unintentionally influencing clinical decision-making by incentivizing hospitals to choose more invasive SAVR procedures over less-invasive TAVR procedures.
As mentioned earlier, the requestor recommended that CMS delete MS-DRGs 266 and 267 and move the cases reporting transcatheter aortic valve replacement or repair (supplement) procedures currently assigned to those MS-DRGs into MS-DRGs 216, 217, 218, 219, 220, and 221. We stated in the proposed rule that the requestor performed its own analysis and stated that the models of this suggested solution indicated the change would result in moderate differences in per case payments by case type and would ( print page 69026) not increase overall Medicare spending. The requestor noted that while their requested solution would potentially decrease payment to cases currently assigned to MS-DRGs 216, 217, 218, 219, 220, and 221, while at the same time increasing the payment to cases reporting endovascular cardiac valve replacement and supplement procedures, the results of their claim analysis demonstrated that the net difference in total payments across all cases would increase by approximately $6.5 million. The requestor stated that they anticipate that their proposed solution could increase Medicare patients' access to innovative endovascular cardiac valve procedures by establishing payment neutrality between SAVR and TAVR procedures.
As discussed in the proposed rule, we reviewed this request and noted the requestor was correct that in Version 41.1 cases reporting procedure codes that describe endovascular cardiac valve replacement and supplement procedures, including TAVR, group to MS-DRGs 266 and 267. We stated that the requestor was also correct that cases reporting procedure codes that describe surgical cardiac valve replacement and supplement procedures, including SAVR, group to MS-DRGs 216, 217, 218, 219, 220, and 221. We refer the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1 (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRGs 216, 217, 218, 219, 220, 221, 266, and 267.
As discussed in the proposed rule, to begin our analysis, we identified the ICD-10-PCS procedure codes that describe endovascular (transcatheter) cardiac valve replacement and supplement procedures and the ICD-10-PCS procedure codes that describe surgical cardiac valve replacement and supplement procedures. We also identified the ICD-10-PCS codes that describe cardiac catheterization, as MS-DRGs 216, 217, and 218 (Cardiac Valve and Other Major Cardiothoracic Procedures with Cardiac Catheterization with MCC, with CC, and without CC/MCC, respectively) are defined by the performance of cardiac catheterization. We refer the reader to Table 6P.2a, Table 6P.2b, and Table 6P.2c, respectively, associated with the proposed rule and this final rule (available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps ) for the lists of the ICD-10-PCS procedure codes that we identified that describe endovascular cardiac valve replacement and supplement procedures, surgical cardiac valve replacement and supplement procedures, and cardiac catheterization procedures.
As discussed in the proposed rule, we then examined the claims data from the September 2023 update of the FY 2023 MedPAR file for all cases in MS-DRGs 216, 217, 218, 219, 220, and 221 and compared the results to cases reporting surgical cardiac valve replacement and supplement procedures in MS-DRG 216, 217, 218, 219, 220, and 221. The following table shows our findings:

As shown in the table, in MS-DRG 216, we identified a total of 5,033 cases with an average length of stay of 13.9 days and average costs of $84,176. Of those 5,033 cases, there were 2,973 cases reporting surgical cardiac valve replacement and supplement procedures, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 216 ($87,497 compared to $84,176) and a longer average length of stay (16.8 days compared to 13.9 days). In MS-DRG 217, we identified a total of 1,635 cases with an average length of stay of 7.2 days and average costs of $58,381. Of those 1,635 cases, there were 867 cases reporting surgical cardiac valve replacement and supplement procedures, with average costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 217 ($56,829 compared to $58,381) and a longer average length of stay (9.5 days compared to 7.2 days). In MS-DRG 218, we identified a total of 275 cases with an average length of stay of 3.4 days and average costs of $54,624. Of those 275 cases, there were 60 cases reporting surgical cardiac valve replacement and supplement procedures, with average costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 218 ($45,096 compared to $54,624) and a longer average length of stay (6.7 days compared to 3.4 days). In MS-DRG 219, we identified a total of 12,458 cases with an average length of stay of 10.5 days and average costs of $67,228. Of those 12,458 cases, there were 9,780 cases reporting surgical cardiac valve replacement and supplement procedures, with average costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 219 ($64,954 ( print page 69027) compared to $67,228), and a slightly shorter average length of stay (10.3 days compared to 10.5 days). In MS-DRG 220, we identified a total of 9,829 cases with an average length of stay of 6.3 days and average costs of $47,242. Of those 9,829 cases, there were 7,841 cases reporting surgical cardiac valve replacement and supplement procedures, with average costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 220 ($46,245 compared to $47,242)and a slightly longer average length of stay (6.4 days compared to 6.3 days). In MS-DRG 221, we identified a total of 1,242 cases with an average length of stay of 3.8 days and average costs of $41,539. Of those 1,242 cases, there were 627 cases reporting surgical cardiac valve replacement and supplement procedures, with average costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 221 ($39,081 compared to $41,539) and a longer average length of stay (4.9 days compared to 3.8 days).
Next, as discussed in the proposed rule, we examined claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRGs 266 and 267. Our findings are shown in the following table.

As noted in the proposed rule, because there is a two-way split within MS-DRGs 266 and 267 and there is a three-way split within MS-DRGs 216, 217, and 218, and MS-DRGs 219, 220, and 221 (Cardiac Valve and Other Major Cardiothoracic Procedures without Cardiac Catheterization with MCC, with CC, and without CC/MCC, respectively), we also analyzed the cases reporting a code describing an endovascular cardiac valve replacement and supplement procedure with a procedure code describing the performance of a cardiac catheterization for the presence or absence of a secondary diagnosis designated as a complication or comorbidity (CC) or a major complication or comorbidity (MCC). We also analyzed the cases reporting a code describing an endovascular cardiac valve replacement and supplement procedure without a procedure code describing the performance of a cardiac catheterization for the presence or absence of a secondary diagnosis designated as a CC or an MCC.

As shown in the table, the data analysis performed indicates that the 5,443 cases in MS-DRG 266 reporting endovascular cardiac valve replacement and supplement procedures with a procedure code describing the performance of a cardiac catheterization, and with a secondary diagnosis code designated as an MCC have an average length of stay that is shorter than the average length of stay (7.9 days versus 16.8 days) and lower average costs ($63,128 versus $87,497) when compared to the cases in MS-DRG 216 reporting surgical cardiac valve replacement and supplement procedures with a procedure code describing the performance of a cardiac catheterization, and with a secondary diagnosis code designated as an MCC. The 4,761 cases in MS-DRG 267 reporting endovascular cardiac valve replacement and supplement procedures with a procedure code describing the performance of a cardiac catheterization, and with a secondary diagnosis code designated as a CC have an average length of stay that is shorter than the average length of stay (2 days versus 9.5 days) and lower average costs ($42,163 versus $56,829) when compared to the cases in MS-DRG 217 reporting surgical cardiac valve replacement and supplement procedures with a procedure code describing the performance of a cardiac catheterization, and with a secondary diagnosis code designated as an CC. The 1,386 cases in MS-DRG 267 reporting ( print page 69028) endovascular cardiac valve replacement and supplement procedures with a procedure code describing the performance of a cardiac catheterization, and without a secondary diagnosis code designated as a CC or MCC have an average length of stay that is shorter than the average length of stay (1.3 days versus 6.7 days) and lower average costs ($39,709 versus $45,096) when compared to the cases in MS-DRG 218 reporting surgical cardiac valve replacement and supplement procedures with a procedure code describing the performance of a cardiac catheterization, without a secondary diagnosis code designated as a CC or MCC.
The 14,493 cases in MS-DRG 266 reporting endovascular cardiac valve replacement and supplement procedures without a procedure code describing the performance of a cardiac catheterization, and with a secondary diagnosis code designated as an MCC have an average length of stay that is shorter than the average length of stay (3.5 days versus 10.3 days) and lower average costs ($50,831 versus $64,954) when compared to the cases in MS-DRG 219 reporting surgical cardiac valve replacement and supplement procedures without a procedure code describing the performance of a cardiac catheterization, and with a secondary diagnosis code designated as an MCC. The 22,996 cases in MS-DRG 267 reporting endovascular cardiac valve replacement and supplement procedures without a procedure code describing the performance of a cardiac catheterization, and with a secondary diagnosis code designated as a CC have an average length of stay that is shorter than the average length of stay (1.5 days versus 6.4 days) and lower average costs ($43,637 versus $46,245) when compared to the cases in MS-DRG 220 reporting surgical cardiac valve replacement and supplement procedures without a procedure code describing the performance of a cardiac catheterization, and with a secondary diagnosis code designated as an CC. The 7,522 cases in MS-DRG 267 reporting endovascular cardiac valve replacement and supplement procedures without a procedure code describing the performance of a cardiac catheterization, and without a secondary diagnosis code designated as a CC or MCC have an average length of stay that is shorter than the average length of stay (1.2 days versus 4.9 days) and higher average costs ($42,472 versus $39,081) when compared to the cases in MS-DRG 221 reporting surgical cardiac valve replacement and supplement procedures without a procedure code describing the performance of a cardiac catheterization, without a secondary diagnosis code designated as a CC or MCC.
We stated in the proposed rule that this data analysis shows the cases in MS-DRG 266 and 267 reporting endovascular cardiac valve replacement and supplement procedures with a procedure code describing the performance of a cardiac catheterization when distributed based on the presence or absence of a secondary diagnosis designated as a CC or a MCC have average costs lower than the average costs of cases reporting surgical cardiac valve replacement and supplement procedures with a procedure code describing the performance of a cardiac catheterization in the FY 2023 MedPAR file for MS-DRGs 216, 217, and 218 respectively, and the average lengths of stay are shorter. Similarly, the cases in MS-DRG 266 and 267 reporting endovascular cardiac valve replacement and supplement procedures without a procedure code describing the performance of a cardiac catheterization when distributed based on the presence or absence of a secondary diagnosis designated as a CC or a MCC generally have average costs lower than the average costs of cases reporting surgical cardiac valve replacement and supplement procedures without a procedure code describing the performance of a cardiac catheterization in the FY 2023 MedPAR file for MS-DRGs 219, 220, and 221 respectively, and the average lengths of stay are shorter.
We stated in the proposed rule that for patients with an indication for cardiac valve replacement, clinical and anatomic factors must be considered when decision-making between procedures such as TAVR and SAVR. We noted that SAVR is not a treatment option for patients with extreme surgical risk (that is, high probability of death or serious irreversible complication), severe atheromatous plaques of the ascending aorta such that aortic cross-clamping is not feasible, or with other conditions that would make operation through sternotomy or thoracotomy prohibitively hazardous. We stated that we agreed that the endovascular or transcatheter technique presents a viable option for high-risk patients who are not candidates for the traditional open surgical approach, however we also noted that TAVR is not indicated for every patient. TAVR is contraindicated in patients who cannot tolerate an anticoagulation/antiplatelet regimen, or who have active bacterial endocarditis or other active infections, or who have significant annuloplasty ring dehiscence.
In the proposed rule, we stated we had concern with the assertion that clinicians perform more invasive surgical procedures, such as SAVR procedures, only to increase payment to their facility where minimally invasive TAVR procedures are also viable option. The choice of SAVR versus TAVR should not be based on potential facility payment. Instead, the decision on the procedural approach to be utilized should be based upon an individualized risk-benefit assessment that includes reviewing factors such as the patient's age, surgical risk, frailty, valve morphology, and presence of concomitant valve disease or coronary artery disease. As we have stated in prior rulemaking ( 83 FR 41201 ), it is not appropriate for facilities to deny treatment to beneficiaries needing a specific type of therapy or treatment that involves increased costs. Conversely, it is not appropriate for facilities to recommend a specific type of therapy or treatment strictly because it may involve higher payment to the facility.
Also, we stated we had concern with the requestor's assertion that sharing a single set of MS-DRGs could eliminate any perceived disincentives hospitals may face and create financial neutrality between the two lifesaving treatment options. In the proposed rule, we noted that the data analysis shows that cases reporting surgical cardiac valve replacement and supplement procedures have higher costs and longer lengths of stay. We stated that if clinical decision-making is being driven by financial motivations, as suggested by the requestor, in circumstances where the decision on which approach is best (for example, TAVR or SAVR) is left to the providers' discretion, it is unclear how reducing payment for surgical cardiac valve replacement and supplement procedures would eliminate possible disincentives, or not have the opposite effect, and instead incentivize endovascular cardiac valve replacement and supplement procedures.
As discussed in the proposed rule, the MS-DRGs are a classification system intended to group together diagnoses and procedures with similar clinical characteristics and utilization of resources and are not intended to be utilized as a tool to incentivize the performance of certain procedures. When performed, surgical cardiac valve replacement and supplement procedures are clinically different from endovascular cardiac valve replacement and supplement procedures in terms of technical complexity and hospital ( print page 69029) resource use. In the FY 2015 IPPS/LTCH PPS final rule, we stated that separately grouping endovascular valve replacement procedures provides greater clinical cohesion for this subset of high-risk patients. In the FY 2025 IPPS/LTCH PPS proposed rule, we stated our claims analysis demonstrates that this continues to be substantiated by the difference in average costs and average lengths of stay demonstrated by the two cohorts. We stated we continue to believe that endovascular cardiac valve replacement and supplement procedures are clinically coherent in their currently assigned MS-DRGs. Therefore, we proposed to maintain the structure of MS-DRGs 266 and 267 for FY 2025.
Comment: Many commenters expressed support for the proposal to maintain the structure of MS-DRGs 266 and 267 for FY 2025. A commenter stated it is unclear why the requestor would imply that there is any type of bias in patient selection of surgical cardiac valve replacement and repair procedures over endovascular cardiac valve replacement and supplement procedures, and stated in their experience, the decision to perform endovascular or surgical cardiac valve replacement and supplement procedures is typically made by the heart team based on the patient's individualized risk-benefit and associated factors such as the patient's age, surgical risk, frailty, valve morphology, and presence of concomitant valve disease or coronary artery disease. A commenter specifically stated while they firmly believe that procedures such as TAVR should be paid at a rate that makes them efficacious for hospitals to perform, given the analysis provided by CMS, the requested MS-DRG modification may not be the best path to this end. Another commenter stated they agreed with CMS that although both types of cardiac valve interventions treat the same type of disease, the work and resource utilization associated with the procedures is significantly different and noted that surgical cardiac replacement or repair procedures typically require more resources such as increased operating room time, additional supportive staff for the procedure and longer lengths of stay. Another commenter stated in addition to the important points that CMS made in the proposed rule regarding the lack of cost coherence between TAVR and SAVR procedures, in their own analysis, the impact of moving TAVR cases into MS-DRGs 216, 217, 218, 219, 220, and 221 (Cardiac Valve & Other Major Cardiothoracic Procedure with and without Cardiac Catheterization, with MCC, with CC, and without CC/MCC, respectively) would cause a 12 percent decrease in average costs and a 9 percent decrease in relative weight in MS-DRGs 216, 217, 218, 219, 220, and 221.
Response: We appreciate the commenters' support and feedback.
Comment: A commenter (the requestor) disagreed with the proposal to maintain the structure of MS-DRGs 266 and 267 and stated appropriate payment under the IPPS is critical to improving access to TAVR procedures for all eligible patients and ensuring timely access to valve replacement therapies. This commenter stated they continue to maintain that incentives for valve replacement procedures strongly favor SAVR over TAVR due to the payment differential between the two procedures. While acknowledging that SAVR cases have increased clinical labor and indirect costs, the commenter again asserted that merging the procedures into a single set of MS-DRGs would establish better financial neutrality between the procedure options by creating more similarity between TAVR and SAVR contribution margins as hospitals measure per-case profitability. Lastly, the commenter noted in their own analysis, payment rates for MS-DRGs 266 and 267 have declined approximately 6 percent from 2022 to 2025, while the payments rates for MS-DRGs 216, 217, 218, 219, 220, and 221 have increased by 8 percent in the same time frame and stated that the years of declining TAVR payment rates while SAVR payment rates increased do influence hospital decisions about whether to expand their structural heart programs to include TAVR procedures, particularly in hospitals located in geographic areas with low wage indexes.
Response: We appreciate the commenter's feedback. With respect to changes in payment rates in the referenced MS-DRGs, each year we calculate the relative weights by dividing the average cost for cases within each MS-DRG by the average cost for cases across all MS-DRGs. We believe any weight changes observed by the commenter over time to be appropriately driven by the underlying data in the years since CMS began using the ICD-10 data in calculating the relative weights. We also note that over the past five years, there have been changes to the hierarchy and structure of certain MS-DRGs in MDC 05. It is to be expected that when MS-DRGs are restructured, such as when procedure codes are reassigned or the hierarchy within an MDC is revised, resulting in a different case-mix within the MS-DRGs, the relative weights of the MS-DRGs will change as a result. Therefore, the data appear to reflect that the differences in the relative weights reflected in Table 5-List of Medicare Severity Diagnosis Related Groups (MS-DRGs), Relative Weighting Factors, and Geometric and Arithmetic Mean Length of Stay associated with the final rule for each applicable fiscal year can be attributed to the fact that the finalization of these proposals resulted in a different case-mix within the MS-DRGs, which is then being reflected in the relative weights. We refer readers to section II.D.2. of the preamble of this final rule for a discussion of the relative weight calculations.
As stated in prior rulemaking ( 88 FR 58730 ), the MS-DRGs were developed as a patient classification scheme consisting of patients who are similar clinically and with regard to their consumption of hospital resources. While all patients are unique, groups of patients have diagnostic and therapeutic attributes in common that determine their level of resource intensity. Similar resource intensity means that the resources used are relatively consistent across the patients in each MS-DRG. When performed, surgical cardiac valve replacement and supplement procedures are clinically different from endovascular cardiac valve replacement and supplement procedures in terms of technical complexity and hospital resource use. We continue to believe that endovascular cardiac valve replacement and supplement procedures are clinically coherent in their currently assigned MS-DRGs.
As stated earlier, the MS-DRGs are not intended to be utilized as a tool to incentivize the performance of certain procedures. As we have stated in prior rulemaking, we rely on providers to assess the needs of their patients and provide the most appropriate treatment. It is not appropriate for facilities to deny treatment to beneficiaries needing a specific type of therapy or treatment that potentially involves increased costs ( 86 FR 44847 ). It would also not be appropriate to consider modifications to the MS-DRG assignment of cases reporting the performance of a specific procedure solely as an incentive for providers to perform one procedure over another.
Therefore, after consideration of the public comments we received, and for the reasons stated earlier, we are finalizing our proposal to maintain the structure of MS-DRGs 266 and 267, without modification, for FY 2025. ( print page 69030)
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35966 through 35968 ), we discussed a request we received to review the GROUPER logic for MS-DRG 215 (Other Heart Assist System Implant) in MDC 05 (Diseases and Disorders of the Circulatory System). The requestor stated that when the procedure code describing the revision of malfunctioning devices within the heart via an open approach is assigned, the encounter groups to MS-DRG 215. The requestor stated that, in their observation, ICD-10-PCS code 02WA0JZ (Revision of synthetic substitute in heart, open approach) can only be assigned if a more specific anatomical site is not documented in the operative note. The requestor further stated they interpreted this to mean that an ICD-10-PCS procedure code describing the open revision of a synthetic substitute in the heart can only apply to the ventricular wall or left atrial appendage and excludes the atrial or ventricular septum or any valve to qualify for MS-DRG 215 and recommended that CMS consider the expansion of the open revision of heart structures to include the atrial or ventricular septum and heart valves.
In the proposed rule we stated that to begin our analysis, we reviewed the GROUPER logic. We stated that the requestor is correct that ICD-10-PCS procedure code 02WA0JZ is currently one of the listed procedure codes in the GROUPER logic for MS-DRG 215. While the requestor stated that when procedures codes describing the revisions of malfunctioning devices within the heart via an open approach are assigned, the encounter groups to MS-DRG 215, we stated we wished to clarify that the revision codes listed in the GROUPER logic for MS-DRG 215 specifically describe procedures to correct, to the extent possible, a portion of a malfunctioning heart assist device or the position of a displaced heart assist device. Further, we stated it was unclear what was meant by the requestor's statement that ICD-10-PCS code 02WA0JZ can only be assigned if more specific anatomical site is not documented in the operative note, as ICD-10-PCS code 02WA0JZ is used to describe the open revision of artificial heart systems. We noted that total artificial hearts are pulsating bi-ventricular devices that are implanted into the chest to replace a patient's left and right ventricles and can provide a bridge to heart transplantation for patients who have no other reasonable medical or surgical treatment options. We refer the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1 (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRG 215. We encouraged the requestor and any providers that have cases involving heart assist devices for which they need ICD-10 coding assistance and clarification on the usage of the codes, to submit their questions to the American Hospital Association's Central Office on ICD-10 at https://www.codingclinicadvisor.com/ .
As previously noted, as discussed in the proposed rule, the requestor recommended that we consider expansion of the open revision of heart structures to include the atrial or ventricular septum and heart valves. The requestor did not provide a specific list of procedure codes involving the open revision of heart structures. While not explicitly stated, we stated we understood this request to be for our consideration of the reassignment of the procedure codes describing the open revision of devices in the heart valves, atrial septum, or ventricular septum to MS-DRG 215, therefore, we stated we reviewed the ICD-10-PCS classification and identified the following 18 procedure codes. These 18 codes are all assigned to MS-DRGs 228 and 229 (Other Cardiothoracic Procedures with and without MCC, respectively) in MDC 05 in Version 41.1.

Next, in the proposed rule we stated we examined claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRG 228 and 229 to identify cases reporting one of the 18 codes listed previously that describe the open revision of devices in the heart valves, atrial septum, or ventricular septum. Our findings are shown in the following table:

As shown in the table, in MS-DRG 228, we identified a total of 4,391 cases with an average length of stay of 8.7 days and average costs of $44,565. Of those 4,391 cases, there were 12 cases reporting a procedure code describing the open revision of devices in the heart valves, atrial septum, or ventricular septum, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 228 ($51,549 compared to $44,565) and a longer average length of stay (15.7 days compared to 8.7 days). In MS-DRG 229, we identified a total of 5,712 cases with an average length of stay of 3.3 days and average costs of $28,987. Of those 5,712 cases, there was one case reporting a procedure code describing the open revision of devices in the heart valves, atrial septum, or ventricular septum with costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 229 ($11,322 compared to $28,987) and a shorter length of stay (1 day compared to 3.3 days).
We stated we then examined claims data from the September 2023 update of the FY 2023 MedPAR for MS-DRG 215. Our findings are shown in the following table.

In the proposed rule we stated our analysis indicates that the cases assigned to MS-DRG 215 have much higher average costs than the cases reporting a procedure code describing the open revision of devices in the heart valves, atrial septum, or ventricular septum currently assigned to MS-DRGs 228 and 229. Instead, the average costs and average length of stay for case reporting a procedure code describing the open revision of devices in the heart valves, atrial septum, or ventricular septum appear to be generally more aligned with the average costs and average length of stay for all cases in MS-DRGs 228 and 229, where they are currently assigned.
In addition, based on our review of the clinical considerations, we stated we did not believe the procedure codes describing the open revision of devices in the heart valves, atrial septum, or ventricular septum are clinically coherent with the procedure codes currently assigned to MS-DRG 215. We noted that heart assist devices, such as ventricular assist devices and artificial heart systems, provide circulatory support by taking over most of the workload of the left ventricle. Blood enters the pump through an inflow conduit connected to the left ventricle and is ejected through an outflow conduit into the body's arterial system. Heart assist devices can provide temporary left, right, or biventricular support for patients whose hearts have failed and can also be used as a bridge for patients who are awaiting a heart transplant. In the proposed rule we stated that devices placed in the heart valves, atrial septum, or ventricular septum do not serve the same purpose as heart assist devices and we stated we did not believe the procedure codes describing the revision of these devices should be assigned to MS-DRG 215. Further, we stated that the various indications for devices placed in the heart valves, atrial septum or ventricular septum are not aligned with the indications for heart assist devices. We stated we believe that patients with indications for heart assist devices tend to be more severely ill and these inpatient admissions are associated with greater resource utilization. Therefore, for the reasons stated previously, we proposed to maintain the GROUPER logic for MS-DRG 215 for FY 2025.
Comment: Many commenters agreed with CMS' proposal to maintain the GROUPER logic for MS-DRG 215 for FY 2025. A commenter stated that they agreed with CMS that, in general, most patients with indications for heart assist devices tend to be more severely ill and will require greater resource utilization than patients that are admitted for open revision of devices related to heart valves, atrial septum, or ventricular septum.
Therefore, after consideration of the public comments we received, we are finalizing our proposal to maintain the GROUPER logic for MS-DRG 215 for FY 2025, without modification.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35968 through 35969 ), we identified a replication issue from the ICD-9 based MS-DRGs to the ICD-10 based MS-DRGs regarding the assignment of eight ICD-10-PCS codes that describe the excision of intestinal body parts by open, percutaneous, or percutaneous endoscopic approach. Under the Version 32 ICD-9 based MS-DRGs, ICD-9-CM procedure code 45.33 (Local excision of lesion or tissue of small intestine, except duodenum) was designated as an O.R. procedure and was assigned to MDC 06 (Diseases and Disorders of the Digestive System) in MS-DRGs 347, 348, and 349 (Anal and Stomal Procedures with MCC, with CC, and without CC/MCC, respectively).
In the proposed rule, we noted that there are eight ICD-10-PCS code translations that provide more detailed and specific information for ICD-9-CM code 45.33 that also currently group to MS-DRGs 347, 348, and 349 in the ICD-10 MS-DRGs Version 41.1. These eight procedure codes are shown in the following table:

In the proposed rule we stated we noted during our review of this issue that under ICD-9-CM, procedure code 45.33 did not differentiate the specific type of approach used to perform the procedure. This is in contrast to the eight comparable ICD-10-PCS code translations listed in the previous table that do differentiate among various approaches (open, percutaneous, and percutaneous endoscopic). We also noted that there are four additional ICD-10-PCS code translations that provide more detailed and specific information for ICD-9-CM code 45.33, however these four codes currently group to MS-DRGs 329, 330, and 331 (Major Small and Large Bowel Procedures with MCC, with CC, and without CC/MCC, respectively), and not MS-DRGs 347, 348, and 349, in the ICD-10 MS-DRGs Version 41.1. These four procedure codes are shown in the following table:

We refer the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1 (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRGs 329, 330, 331, 347, 348, and 349.
Next, as discussed in the proposed rule we examined claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRG 347, 348, and 349 to identify cases reporting one of the eight codes listed previously that describe excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach. Our findings are shown in the following table:

As shown in the table, in MS-DRG 347, we identified a total of 752 cases with an average length of stay of 7.6 days and average costs of $21,462. Of those 752 cases, there were 66 cases reporting one of eight procedure codes describing the excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 347 ($27,081 compared to $21,462) and a longer average length of stay (8.5 days compared to 7.6 days). In MS-DRG 348, we identified a total of 1,580 cases with an average length of stay of 4.2 days and average costs of $12,020. Of those 1,580 cases, there were 192 cases reporting one of eight procedure codes describing the excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 348 ($17,063 compared to $12,020) and a longer average length of stay (4.9 days compared to 4.2 days). In MS-DRG 349, we identified a total of 644 cases with an average length of stay of 2.2 days and average costs of $9,095. Of those 644 cases, there were 117 cases reporting one of eight procedure codes describing the excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach, with average ( print page 69033) costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 349 ($14,612 compared to $9,095),and a longer average length of stay (3 days compared to 2.2 days).
We stated we then examined claims data from the September 2023 update of the FY 2023 MedPAR for MS-DRGs 329, 330, and 331. Our findings are shown in the following table.

While the average costs for all cases in MS-DRGs 329, 330, and 331 are higher than the average costs of the cases reporting one of eight procedure codes describing the excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach, we stated the data suggest that overall, cases reporting one of eight procedure codes describing the excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach may be more appropriately aligned with the average costs of the cases in MS-DRGs 329, 330, and 331 in comparison to MS-DRGs 347, 348, and 349, even though the average lengths of stay are shorter.
In the proposed rule we stated we reviewed this grouping issue, and our analysis indicates that the eight procedure codes describing the excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach were initially assigned to the list of procedures in the GROUPER logic for MS-DRGs 347, 348, and 349 as a result of replication in the transition from ICD-9 to ICD-10 based MS-DRGs. We also noted that procedure codes 0DB83ZZ, 0DBA3ZZ, 0DBA4ZZ, 0DBB3ZZ, 0DBB4ZZ, 0DBC0ZZ, 0DBC3ZZ, and 0DBC4ZZ do not describe procedures on a stoma, which is an artificial opening on the abdomen that can be connected to either the digestive or urinary system to allow waste to be diverted out of the body, or the anus. We stated we supported the reassignment of codes 0DB83ZZ, 0DBA3ZZ, 0DBA4ZZ, 0DBB3ZZ, 0DBB4ZZ, 0DBC0ZZ, 0DBC3ZZ, and 0DBC4ZZ for clinical coherence and that we believe these eight procedure codes should be appropriately grouped along with the four other procedure codes that describe excision of intestinal body parts by an open, or percutaneous endoscopic approach currently assigned to MS-DRGs 329, 330, and 331.
Accordingly, because the procedures described by the eight procedure codes that describe excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach are not clinically consistent with procedures on the anus or stoma, and it is clinically appropriate to reassign these procedures to be consistent with the four other procedure codes that describe excision of intestinal body parts by an open, or percutaneous endoscopic approach in MS-DRGs 329, 330, and 331, we proposed the reassignment of procedure codes 0DB83ZZ, 0DBA3ZZ, 0DBA4ZZ, 0DBB3ZZ, 0DBB4ZZ, 0DBC0ZZ, 0DBC3ZZ, and 0DBC4ZZ from MS-DRGs 347, 348, and 349 (Anal and Stomal Procedures with MCC, with CC, and without CC/MCC, respectively) to MS-DRGs 329, 330, and 331 (Major Small and Large Bowel Procedures with MCC, with CC, and without CC/MCC, respectively) in MDC 06, effective FY 2025.
Comment: Commenters supported the proposal to reassign the eight procedure codes that describe the excision of intestinal body parts by an open, percutaneous, or percutaneous endoscopic approach from MS-DRGs 347, 348, and 349 to MS-DRGs 329, 330, and 331. A commenter thanked CMS for this review and stated that they agreed that the proposed reassignment would correct an error that was made during the transition from the ICD-9 based MS-DRGs to the ICD-10 based MS-DRGs. Other commenters stated that these procedure codes do not belong in the MS-DRGs they are currently assigned to, and that reassignment will appropriately group these procedures based on the body part involved.
After consideration of the public comments we received, we are finalizing our proposal to reassign procedure codes 0DB83ZZ, 0DBA3ZZ, 0DBA4ZZ, 0DBB3ZZ, 0DBB4ZZ, 0DBC0ZZ, 0DBC3ZZ, and 0DBC4ZZ from MS-DRGs 347, 348, and 349 (Anal and Stomal Procedures with MCC, with CC, and without CC/MCC, respectively) to MS-DRGs 329, 330, and 331 (Major Small and Large Bowel Procedures with MCC, with CC, and without CC/MCC, respectively) in MDC 06, without modification, effective October 1, 2024, for FY 2025.
In the proposed rule we discussed an inconsistency in the GROUPER logic for MS-DRGs 456, 457, and 458 (Spinal Fusion Except Cervical with Spinal Curvature, Malignancy, Infection or Extensive Fusions with MCC, with CC, and without CC/MCC, respectively) related to ICD-10-CM diagnosis codes describing deforming dorsopathies. The logic for case assignment to MS-DRGs 456, 457, and 458 as displayed in the ICD-10 MS-DRG Definitions Manual Version 41.1 (which is available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/MS-DRG-Classifications-and-Software ) is comprised of four logic lists. The first logic list is entitled “Spinal Fusion Except Cervical” and is defined by a list of procedure codes designated as O.R. procedures that describe spinal fusion procedures of the thoracic, thoracolumbar, lumbar, lumbosacral, sacrococcygeal, coccygeal, and sacroiliac joint. The second logic list is entitled “Spinal Curvature/Malignancy/Infection” and is defined by a list of diagnosis codes describing spinal curvature, spinal malignancy, and spinal infection that are used to define the logic for case assignment when any one of the listed diagnosis codes is reported as the principal diagnosis. The third logic list is entitled “OR Secondary Diagnosis” and is defined by a list of diagnosis codes describing curvature of the spine that are used to define the logic for case assignment when any one of the listed codes is reported as a secondary diagnosis. The fourth logic list is entitled “Extensive Fusions” and is defined by a list of procedure codes designated as O.R. procedures that describe extensive spinal fusion procedures. We refer the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1, (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/ ( print page 69034) acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRGs 456, 457, and 458.
In the second logic list entitled “Spinal Curvature/Malignancy/Infection” there are a subset of six diagnosis codes describing other specified deforming dorsopathies as shown in the following table.

In the third logic list entitled “OR Secondary Diagnosis” there are currently 14 diagnosis codes listed, one of which is diagnosis code M43.8X9 (Other specified deforming dorsopathies, site unspecified) as shown in the following table.

In the proposed rule we stated that we recognized that the five diagnosis codes describing deforming dorsopathies of specific anatomic sites that are listed in the second logic list entitled “Spinal Curvature/Malignancy/Infection” are not listed in the third logic list entitled “OR Secondary Diagnosis”, rather, only diagnosis code M43.8X9 (Other specified deforming dorsopathies, site unspecified) appears in both logic lists. Therefore, we considered if it was clinically appropriate to add the five diagnosis codes describing deforming dorsopathies of specific anatomic sites that are listed in the second logic list entitled “Spinal Curvature/Malignancy/Infection” to the third logic list entitled “OR Secondary Diagnosis”.
A deforming dorsopathy is characterized by abnormal bending or flexion in the vertebral column. All spinal deformities involve problems with curve or rotation of the spine, regardless of site specificity. In the proposed rule we stated our belief that the five diagnosis codes describing deforming dorsopathies of specific anatomic sites are clinically aligned with the diagnosis codes currently included in the “OR Secondary Diagnosis” logic list. Therefore, for clinical consistency we proposed to add diagnosis codes M43.8X4, M43.8X5, M43.8X6, M43.8X7, and M43.8X8 to the “OR Secondary Diagnosis” logic list for MS-DRGs 456, 457, and 458, effective October 1, 2024, for FY 2025.
Comment: Commenters supported the proposal to add diagnosis codes M43.8X4, M43.8X5, M43.8X6, M43.8X7, and M43.8X8 to the “OR Secondary Diagnosis” logic list for MS-DRGs 456, 457, and 458.
After consideration of the public comments we received, we are finalizing our proposal to add diagnosis codes M43.8X4, M43.8X5, M43.8X6, M43.8X7, and M43.8X8 to the “OR Secondary Diagnosis” logic list for MS-DRGs 456, 457, and 458 effective October 1, 2024, for FY 2025.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35971 through 35985 ), we discussed a request we received to reassign cases reporting spinal fusion procedures using an aprevo TM customized interbody fusion device from the lower severity MS-DRG 455 (Combined Anterior and Posterior Spinal Fusion without CC/MCC) to the higher severity MS-DRG 453 (Combined Anterior and Posterior Spinal Fusion with MCC), from the lower severity MS-DRG 458 (Spinal Fusion Except Cervical with Spinal Curvature, Malignancy, Infection or Extensive Fusions without CC/MCC) to the higher severity level MS-DRG 456 (Spinal Fusion Except Cervical with Spinal Curvature, Malignancy, Infection or Extensive Fusions with MCC) when a diagnosis of malalignment is reported, and from MS-DRGs 459 and 460 (Spinal Fusion ( print page 69035) Except Cervical with MCC and without MCC, respectively) to MS-DRG 456. We referred the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1 (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic.
In the proposed rule we noted that this topic has been discussed previously in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26726 through 26729 ) and final rule ( 88 FR 58731 through 58735 , as corrected in the FY 2024 final rule correction notice at 88 FR 77211 ). We also noted that the aprevo TM Intervertebral Body Fusion Device technology was approved for new technology add-on payments for FY 2022 ( 86 FR 45127 through 45133 ). We further noted that, as discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49468 through 49469 ), CMS finalized the continuation of the new technology add-on payments for this technology for FY 2023. In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58802 ), we finalized the continuation of new technology add-on payments for the transforaminal lumbar interbody fusion (TLIF) indication for aprevo TM for FY 2024, and the discontinuation of the new technology add-on payments for the anterior lumbar interbody fusion (ALIF) and lateral lumbar interbody fusion (LLIF) indications for FY 2024. We referred the reader to section II.E. for discussion of the FY 2025 status of technologies receiving new technology add-on payments for FY 2024, including the status for the aprevo TM technology.
Additionally, in the proposed rule we noted that in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26726 through 26729 ) and final rule ( 88 FR 58731 through 58735 ), effective October 1, 2021 (FY 2022), we implemented 12 new ICD-10-PCS procedure codes to identify and describe spinal fusion procedures using the aprevo TM customized interbody fusion device. We noted that the manufacturer expressed concerns that there may be unintentional miscoded claims from providers with whom they do not have an explicit relationship and that following the submission of the request for the FY 2024 MS-DRG classification change for cases reporting the performance of a spinal fusion procedure utilizing an aprevo TM customized interbody spinal fusion device, it submitted a code proposal requesting a revision to the title of the procedure codes that were finalized effective FY 2022. We also noted that, as discussed in the FY 2024 IPPS/LTCH PPS final rule, a proposal to revise the code title for the procedure codes that identify and describe spinal fusion procedures using the aprevo TM customized interbody fusion device was presented and discussed as an Addenda item at the March 7-8, 2023 ICD-10 Coordination and Maintenance Committee meeting and subsequently finalized.
As discussed in the proposed rule, the code title changes for the 12 ICD-10-PCS procedure codes to identify and describe spinal fusion procedures using the aprevo TM customized interbody fusion device were reflected in the FY 2024 ICD-10-PCS Code Update files available via the CMS website at: https://www.cms.gov/medicare/coding-billing/icd-10-codes/2024-icd-10-pcs , as well as in Table 6F.—Revised Procedure Code Titles—FY 2024 associated with the FY 2024 IPPS/LTCH PPS final rule and available via the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps . We noted that only the code titles were revised and the code numbers themselves did not change.
Accordingly, effective with discharges on and after October 1, 2023 (FY 2024), the 12 ICD-10-PCS procedure codes to identify and describe spinal fusion procedures using the aprevo TM customized interbody fusion device with their revised code titles are as follows:

As stated in the proposed rule, as part of our analysis of the manufacturer's request to reassign cases involving the aprevo TM device as discussed in the FY 2024 proposed and final rules, we presented findings from our analysis of claims data from the September 2022 update of the FY 2022 MedPAR file for MS-DRGs 453, 454, 455, 456, 457, 458, 459, and 460 and cases reporting any one of the 12 original procedure codes describing utilization of an aprevo TM customized interbody spinal fusion device. We stated that while we agreed that the findings from our analysis appeared to indicate that cases reporting the performance of a procedure using an aprevo TM customized interbody spinal fusion device reflected a higher consumption of resources, due to the concerns expressed with respect to suspected inaccuracies of the coding and therefore, reliability of the claims data, we would continue to monitor the claims data for resolution of the potential coding issues identified by the requestor (the manufacturer). We also stated that we continued to believe additional review of claims data was warranted and would be informative as we continued to consider cases involving this technology for future rulemaking. Specifically, we stated we believed it would be premature to propose any MS-DRG modifications for spinal fusion procedures using an aprevo TM customized interbody spinal fusion device for FY 2024 and finalized our proposal to maintain the structure of MS-DRGs 453, 454, 455, 456, 457, 458, 459, and 460, without modification, for FY 2024 ( 88 FR 58734 through 58735 ). As discussed further in the FY 2024 final rule correction, in response to the manufacturer's comment expressing concern about the reliability of the Medicare claims data in the MedPAR file used for purposes of CMS's claims data analysis, as compared to the manufacturer's analysis of its own customer claims data, we stated that in order for us to consider using non-MedPAR data, the non-MedPAR data must be independently validated, meaning when an entity submits non-MedPAR data, we must be able to independently review the medical records and verify that a particular procedure was performed for each of the cases that purportedly involved the procedure. We noted that, in this particular circumstance, where external ( print page 69037) data for cases reporting the use of an aprevo TM spinal fusion device was provided, we did not have access to the medical records to conduct an independent review; therefore, we were not able to validate or confirm the non-MedPAR data submitted by the commenter for consideration in FY 2024. However, we also noted that our work in this area was ongoing, and we would continue to examine the data and consider these issues as we develop potential future rulemaking proposals. We referred readers to the FY 2024 IPPS/LTCH PPS correction notice ( 88 FR 77211 ) for further discussion.
In the proposed rule, we noted that the manufacturer provided us with a list of the providers with which it indicated it has an explicit relationship, to assist in our ongoing review of its request for reassignment of cases reporting spinal fusion procedures using an aprevo TM interbody fusion device from the lower severity spinal fusion MS-DRGs to the higher severity level spinal fusion MS-DRGs.
As stated in the proposed rule, to continue our analysis of cases reporting spinal fusion procedures using an aprevo TM customized interbody fusion device, we first analyzed claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRGs 453, 454, 455, 456, 457, 458, 459, and 460, and cases reporting any one of the previously listed procedure codes describing the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device. [ 5 ] Our findings are shown in the following tables.

We identified the majority of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRGs 453, 454, and 455 with a total of 242 cases (26 + 129 + 87 = 242) with an average length of stay of 4.6 days and average costs of $68,526. The 26 cases found in MS-DRG 453 appear to have a comparable average length of stay (9.8 days versus 9.5 days) and higher average costs ($99,162 versus $80,420) compared to all the cases in MS-DRG 453, with a difference in average costs of $18,742 for the cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device. The 129 cases found in MS-DRG 454 appear to have a comparable average length of stay (4.9 days versus 4.3 days) and higher average costs ($71,527 versus $54,983) compared to all the cases in MS-DRG 454, with a difference in average costs of $16,544 for the cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device. The 87 cases found in MS-DRG 455 have an identical average length of stay of 2.6 days in comparison to all the cases in MS-DRG 455, however, the difference in average costs is $13,907 ($54,922−$41,015 = $13,907) for the cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device.
For MS-DRGs 456, 457, and 458, we found a total of 19 cases (2 + 11 + 6 = 19) reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device with an average length of stay of 4.7 days and average costs of $51,384. The 2 cases found in MS-DRG 456 have a shorter average length of stay (8.5 days versus 12.6 days) and lower average costs ($69,009 versus $76,060) compared to all the cases in MS-DRG 456. The 11 cases found in MS-DRG 457 also have a shorter average length of stay (5.0 days versus 6.1 days) and lower average costs ($47,221 versus $52,179). For MS-DRG 458, we found 6 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device with a comparable average length of stay (3.0 days versus 3.1 days) and higher average costs ($53,140 versus $39,260) compared to the average costs of all the cases in MS-DRG 458, with a difference in average costs of $13,880 ($53,140−$39,260 = $13,880) for the cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device.
For MS-DRGs 459 and 460, we found a total of 65 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device with an average length of stay of 2.7 days and average costs of $57,128. The single case found in MS-DRG 459 had a longer length of stay (22 days versus 9.6 days) and higher costs ($288,499 versus $53,192) compared to the average costs of all the cases in MS-DRG 459. For MS-DRG 460, the 64 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device had a shorter average length of stay (2.4 days versus 3.4 days) and higher average cost ($53,513 versus $32,586), compared to ( print page 69039) all the cases in MS-DRG 460, with a difference in average costs of $20,927 ($53,513−$32,586 = $20,927) for the cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device.
As discussed in the FY 2024 final rule, the manufacturer expressed concern that there may be unintentional miscoded claims from providers with whom they do not have an explicit relationship and, as previously discussed, subsequently provided the list of providers with which it indicated it has an explicit relationship to assist in our ongoing review. We noted in the proposed rule that in connection with the list of providers submitted, the manufacturer also resubmitted claims data from the Standard Analytical File (SAF) that included FY 2022 claims and the first two quarters (discharges beginning October 1, 2022 through March 31, 2023) of FY 2023 from these providers. We stated that the list of providers the manufacturer submitted to us was considered applicable for the dates of service in connection with the resubmitted claims data. The manufacturer stated that the list of providers with which it has an explicit relationship is subject to change on a weekly basis as additional providers begin to use the technology. The manufacturer also clarified that the external customer data it had previously referenced in connection with the FY 2024 rulemaking that was received directly from the providers with which it has an explicit relationship is Medicare data. As stated in the proposed rule, we reviewed the September update of the FY 2022 MedPAR file and compared it against the claims data file with the list of providers submitted by the manufacturer for FY 2022. We noted that with this updated analysis of the September update of the FY 2022 MedPAR claims data, we were able to confirm that the majority of the cases for the providers with which the manufacturer indicated it has an explicit relationship matched the claims data in our FY 2022 MedPAR file. However, we also stated that we identified 3 claims that appeared in the manufacturer's file that were not found in our FY 2022 MedPAR file and could not be validated. Next, we reviewed the September update of the FY 2023 MedPAR file and compared it against the claims data file with the list of providers submitted by the manufacturer for the first two quarters of FY 2023. We stated we were able to confirm that the majority of the cases for the providers with which the manufacturer indicated it has an explicit relationship matched the claims data in our FY 2023 MedPAR file. However, we also stated that we identified 2 claims that appeared in the manufacturer's file that were not found in our FY 2023 MedPAR file and could not be validated.
As discussed in the proposed rule, in our analysis of the cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRGs 453, 454, 455, 456, 457, 458, 459, and 460 from the September update of the FY 2023 MedPAR file, we also reviewed the findings for cases identified based on the list of providers with which the manufacturer indicated it has an explicit relationship and cases based on other providers, (that is, those providers not included on the manufacturer's list), and compared those to the findings from all the cases we identified in the September update of the FY 2023 MedPAR file reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRGs 453, 454, 455, 456, 457, 458, 459, and 460. The findings from our analysis are shown in the following table. We noted that there were no cases found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device based on the list of providers submitted by the manufacturer in MS-DRG 456.

For MS-DRG 453, the data show that of the 26 cases found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the FY 2023 MedPAR file, 10 cases were reported based on the manufacturer's provider list, and 16 cases were reported based on other providers. The average length of stay is longer (10.5 days versus 9.4 days), and the average costs are higher ($118,863 versus $86,849) for the 10 cases reported based on the manufacturer's provider list compared to the 16 cases that were reported based on other providers. For MS-DRG 454, ( print page 69042) the data show that of the 129 cases found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the FY 2023 MedPAR file, 48 cases were reported based on the manufacturer's provider list, and 81 cases were reported based on other providers. The average length of stay is longer (6.3 days versus 4.1 days), and the average costs are higher ($81,680 versus $65,510) for the 48 cases reported based on the manufacturer's provider list compared to the 81 cases that were reported based on other providers. For MS-DRG 455, the data show that of the 87 cases found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the FY 2023 MedPAR file, 14 cases were reported based on the manufacturer's provider list, and 73 cases were reported based on other providers. The average length of stay is shorter (2.5 days versus 2.6 days), and the average costs are higher ($61,637 versus $53,634) for the 14 cases reported based on the manufacturer's provider list compared to the 73 cases that were reported based on other providers.
For MS-DRG 456, the data show that of the 2 cases found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the FY 2023 MedPAR file, there were no cases reported based on the manufacturer's provider list and the 2 cases reported were based on other providers. For MS-DRG 457, the data show that of the 11 cases found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the FY 2023 MedPAR file, 2 cases were reported based on the manufacturer's provider list, and 9 cases were reported based on other providers. The average length of stay is shorter (4.5 days versus 5.1 days), and the average costs are higher ($53,113 versus $45,912) for the 2 cases reported based on the manufacturer's provider list compared to the 9 cases that were reported based on other providers. For MS-DRG 458, the data show that of the 6 cases found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the FY 2023 MedPAR file, 3 cases were reported based on the manufacturer's provider list, and 3 cases were reported based on other providers. The average length of stay is longer (3.3 days versus 2.7 days), and the average costs are lower ($52,760 versus $53,520) for the 3 cases reported based on the manufacturer's provider list compared to the 3 cases that were reported for other providers.
For MS-DRG 459, the data show that the single case found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the FY 2023 MedPAR file was based on the manufacturer's provider list. There were no cases reported based on other providers. For MS-DRG 460, the data show that of the 64 cases found to report the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the FY 2023 MedPAR file, 13 cases were reported based on the manufacturer's provider list, and 51 cases were reported based on other providers. The average length of stay is comparable (2.6 days versus 2.3 days), and the average costs are higher ($62,829 versus $51,138) for the 13 cases reported based on the manufacturer's provider list compared to the 51 cases that were reported from other providers.
As discussed in the proposed rule, we considered these data findings with regard to the concerns expressed by the manufacturer that there may be unintentional miscoded claims reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from providers with whom the manufacturer does not have an explicit relationship. Based on our review and analysis of the claims data, we stated that we are unable to confirm that the claims from these providers with whom the manufacturer indicated that it does not have an explicit relationship are miscoded.
In the proposed rule we noted that, while a newly established ICD-10 code may be associated with an application for new technology add-on payment, such codes are not generally established to be product specific. We stated that, if, after consulting the official coding guidelines, a provider determines that an ICD-10 code associated with a new technology add-on payment describes the technology that they are billing, the hospital may report the code and be eligible to receive the associated add-on payment. We noted that providers are responsible for ensuring that they are billing correctly for the services they render. In addition, as we noted in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38012 ), coding advice is issued independently from payment policy. We also noted that, historically, we have not provided coding advice in rulemaking with respect to policy ( 82 FR 38045 ). We stated that as one of the Cooperating Parties for ICD-10, we collaborate with the American Hospital Association (AHA) through the Coding Clinic for ICD-10-CM and ICD-10-PCS to promote proper coding. We recommended that an entity seeking coding guidance submit any questions pertaining to correct coding to the AHA.
Accordingly, after review of the list of providers and associated claims data submitted by the manufacturer, and our analysis of the MedPAR data, we stated we believed these MedPAR data are appropriate for our FY 2025 analysis. Therefore, in assessing the request for reassignment of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the lower severity MS-DRG 455 to the higher severity MS-DRG 453, from the lower severity MS-DRG 458 to the higher severity level MS-DRG 456 when a diagnosis of malalignment is reported, and cases from MS-DRGs 459 and 460 to MS-DRG 456 for FY 2025, we considered all the claims data reporting the performance of a spinal fusion procedure, including those spinal fusion procedures using an aprevo TM custom-made anatomically designed interbody fusion device as identified in the September update of the FY 2023 MedPAR file for these MS-DRGs. Consequently, our analysis also included claims based on the list of providers submitted by the manufacturer as well as other providers.
We stated in the proposed rule that, based on the findings from our analysis and clinical review, we do not believe the requested reassignments are supported. Specifically, we stated it would not be appropriate to propose to reassign the 87 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the lower severity level MS-DRG 455 (without CC/MCC) with an average length of stay of 2.6 days and average costs of $54,922 to the higher severity level MS-DRG 453 (with MCC) with an average length of stay of 9.5 days and average costs of $80,420. We noted that if we were to propose to reassign the 87 cases from the lower severity MS-DRG 455 to the higher severity MS-DRG 453, the MS-DRGs would no longer be clinically coherent with regard to severity of illness of the patients, and the cases would reflect a difference in resource utilization, as demonstrated by the difference in average costs of approximately $25,498 ( print page 69043) ($80,420−$54,922 = $25,498), as well as a difference in average length of stay (2.6 days versus 9.5 days) compared to all the cases in MS-DRG 453. Similarly, we stated it would not be appropriate to propose to reassign the 6 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from the lower severity level MS-DRG 458 (without CC/MCC) with an average length of stay of 3.0 days and average costs of $53,140 to the higher severity level MS-DRG 456 (with MCC) with an average length of stay of 12.6 days and average costs of $76,060. We stated that if we were to propose to reassign the 6 cases from the lower severity MS-DRG 458 to the higher severity MS-DRG 456, the MS-DRGs would no longer be clinically coherent with regard to severity of illness of the patients and the cases would reflect a difference in resource utilization, as demonstrated by the difference in average costs of approximately $22,920 ($76,060−$53,140 = $22,920) as well as a difference in average length of stay (3.0 days versus 12.6 days) compared to all the cases in MS-DRG 456. Finally, we stated it would not be appropriate nor consistent with the definition of the MS-DRGs to propose to reassign the 65 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device from MS-DRGs 459 and 460 with an average length of stay of 2.7 days and average costs of $57,128 to MS-DRG 456. In addition to the cases reflecting a difference in resource utilization as demonstrated by the difference in average costs of approximately $18,932 ($76,060−$57,128 = $18,932) as well as having a shorter average length of stay (2.7 days versus 12.6 days), we noted that the logic for case assignment to MS-DRGs 456, 457, and 458 is specifically defined by principal diagnosis logic. As such, cases grouping to this set of MS-DRGs require a principal diagnosis of spinal curvature, malignancy, or infection, or an extensive fusion procedure. We stated that it would not be clinically appropriate to propose to reassign cases from MS-DRGs 459 and 460 that do not have a principal diagnosis of spinal curvature, malignancy, or infection, or an extensive fusion procedure, and are not consistent with the logic for case assignment to MS-DRG 456.
As discussed in the proposed rule, in light of the higher average costs of the cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRGs 453, 454, 455, 458, and 460, we further reviewed the claims data for cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in these MS-DRGs and identified a wide range in the average length of stay and average costs. For example, in MS-DRG 453, the average length of stay for the 26 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device ranged from 3.0 days to 27 days and the average costs ranged from $28,054 to $177,919. In MS-DRG 454, the average length of stay for the 129 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device ranged from 1.0 day to 16 days and the average costs ranged from $10,242 to $316,780. In MS-DRG 455, the average length of stay for the 87 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device ranged from 1.0 day to 9.0 days and the average costs ranged from $7,961 to $216,200. In MS-DRG 456, the length of stay for the 2 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device were 8.0 days and 9.0 days, respectively, with costs of $107,457 and $30,560, respectively. In MS-DRG 457, the average length of stay for the 11 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device ranged from 1.0 day to 17 days and the average costs ranged from $25,955 to $89,176. In MS-DRG 458, the average length of stay for the 6 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device ranged from 1.0 day to 5.0 days and the average costs ranged from $33,165 to $78,720. In MS-DRG 459, the length of stay for the single case reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device was 22 days with a cost of $288,499, indicating it is an outlier. In MS-DRG 460, the average length of stay for the 64 cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device ranged from 1.0 day to 8.0 days and the average costs ranged from $8,981 to $325,104.
As discussed in the proposed rule, in our analysis of the claims data for MS-DRGs 453, 454, and 455, we also identified a number of cases for which additional spinal fusion procedures were performed, beyond the logic for case assignment to the respective MS-DRG. For example, the logic for case assignment to MS-DRGs 453, 454, and 455 requires at least one anterior column fusion and one posterior column fusion (that is, combined anterior and posterior fusion). We noted that the aprevo TM custom-made anatomically designed interbody fusion device is used in the performance of an anterior column fusion. We stated that findings from our analysis of MS-DRG 453 show that of the 26 cases reporting a combined anterior and posterior fusion (including an aprevo TM custom-made anatomically designed interbody fusion device), 24 cases also reported another spinal fusion procedure. We categorized these cases as “multiple level fusions” where another procedure code describing a spinal fusion procedure was reported in addition to the combined anterior and posterior fusion procedure codes. We stated that findings from our analysis of MS-DRG 454 show that of the 129 cases reporting a combined anterior and posterior fusion (including an aprevo TM custom-made anatomically designed interbody fusion device), 100 cases also reported another spinal fusion procedure. Lastly, we stated that findings from our analysis of MS-DRG 455 show that of the 87 cases reporting a combined anterior and posterior fusion (including an aprevo TM custom-made anatomically designed interbody fusion device), 51 cases also reported another spinal fusion procedure.
We noted in the proposed rule that while the findings from our analysis indicate a wide range in the average length of stay and average costs for cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device, we believed the increase in resource utilization for certain cases may be partially attributable to the performance of multiple level fusion procedures and, specifically for MS-DRGs 453 and 454, the reporting of secondary diagnosis MCC and CC conditions. We noted that our analysis of the data for MS-DRGs 453 and 454 show that the cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device also reported multiple MCC and CC conditions, which we believe may be an additional ( print page 69044) contributing factor to the increase in resource utilization for these cases, combined with the reported performance of multiple level fusions.
As discussed in the proposed rule, in our analysis of the data for MS-DRGs 453, 454, and 455 and cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device, we also identified other procedures that were reported, some of which are designated as operating room (O.R.) procedures, that we believed may be another contributing factor to the increase in resource utilization and complexity for these cases. (We noted that because a discectomy is frequently performed in connection with a spinal fusion procedure, we did not consider these procedures as contributing factors to consumption of resources in these spinal fusion cases). We provided a list of the top 5 MCC and CC conditions, as well as the top 5 O.R. procedures (excluding discectomy) reported in MS-DRGs 453, 454, and 455 that we believed may be contributing factors to the increase in resource utilization and complexity for these cases as shown in the tables that follow. We noted that the logic for case assignment to MS-DRG 453 includes the reporting of at least one secondary diagnosis MCC condition (“with MCC”) and cases that group to this MS-DRG may also report secondary diagnosis CC conditions. We provided the frequency data for both the top 5 secondary diagnosis MCC conditions and the top 5 secondary diagnosis CC conditions, in addition to the top 5 O.R. procedures (excluding discectomy) that were reported for spinal fusion cases with an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRG 453. We noted that because the logic for case assignment to MS-DRG 454 includes the reporting of at least one secondary diagnosis CC condition (“with CC”) we provided the top 5 secondary diagnosis CC conditions and the top 5 O.R. procedures (excluding discectomy) that were reported for spinal fusion cases with an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRG 454. We noted that the logic for case assignment to MS-DRG 455 is “without CC/MCC” and does not include any secondary diagnosis MCC or CC conditions, therefore, we only provided a table with the top 5 O.R. procedures (excluding discectomy) reported for that MS-DRG in addition to a spinal fusion procedure.

As previously summarized, our analysis of the claims data for cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device demonstrated a low volume of cases and higher average costs in comparison to all the cases in their respective MS-DRGs (that is, in MS-DRGs 453, 454, 455, 458, 459, and 460). Therefore, as stated in the proposed rule, we expanded our analysis to include all spinal fusion cases in MS-DRGs 453, 454, 455, 456, 457, 458, 459, and 460 to identify and further examine the cases reporting multiple level fusions versus single level fusions, multiple MCCs or CCs, and other O.R. procedures as we believed that clinically, all of these factors may contribute to increases in resource utilization, severity of illness and technical complexity.
As stated in the proposed rule, we began our expanded analysis with MS-DRGs 453, 454, and 455. Based on the findings for a subset of the cases (that is, the subset of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device) in these MS-DRGs as previously discussed, and our review of the logic for case assignment to these MS-DRGs, we developed three categories of spinal fusion procedures to further examine. The first category was for the single level combined anterior and posterior fusions except cervical, the second category was for the multiple level combined anterior and posterior fusions except cervical and the third category was for the combined anterior and posterior cervical spinal fusions. We refer the reader to Table 6P.2d for the list of procedure codes we identified to categorize the single level combined anterior and posterior fusions except cervical, Table 6P.2e for the list of procedure codes we identified to categorize the multiple level combined anterior and posterior fusions except cervical, and Table 6P.2f for the list of procedure codes we identified to categorize the combined anterior and posterior cervical spinal fusions in association with the proposed rule and available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps .
Findings from our analysis are shown in the following table.

The data show that across MS-DRGs 453, 454, and 455, cases reporting multiple level combined anterior and posterior fusion procedures have a comparable average length of stay (9.6 days versus 9.5 days, 4.8 days versus 4.3 days, and 3.0 days versus 2.6 days, respectively) and higher average costs ($91,358 versus $80,420, $64,065 versus $54,983, and $50,097 versus $41,015) compared to all the cases in MS-DRGs 453, 454, and 455, respectively. The data also show that across MS-DRGs 453, 454, and 455, cases reporting multiple level combined anterior and posterior fusion procedures have a longer average length of stay (9.6 days versus 6.4 days, 4.8 days versus 3.4 days, and 3.0 days versus 2.3 days, respectively) and higher average costs ($91,358 versus $47,031, $64,065 versus $38,107, and $50,097 versus $33,010, respectively) compared to cases reporting a single level combined anterior and posterior fusion. For cases reporting a combined anterior and posterior cervical fusion across MS-DRGs 453 and 454, the data show a longer average length of stay (12.5 days versus 9.5 days, and 5.1 days versus 4.3 days, respectively) compared to all the cases in MS-DRGs 453 and 454 and a comparable average length of stay (2.9 days versus 2.6 days) for cases reporting a combined anterior and posterior cervical fusion in MS-DRG 455. The data also show that across MS-DRGs 453, 454, and 455, cases reporting a combined anterior and posterior cervical fusion have higher average costs ($75,077 versus $47,031, $52,274 versus $38,107, and $37,515 versus $33,010, respectively) compared to the single level combined anterior and posterior fusion cases.
The data also reflect that in applying the logic that was developed for the three categories of spinal fusion in MS-DRGs 453, 454, and 455 (single level combined anterior and posterior fusion except cervical, multiple level combined anterior and posterior fusion except cervical, and combined anterior and posterior cervical fusion), there is a small redistribution of cases from the current MS-DRGs 453, 454, and 455 to other spinal fusion MS-DRGs because the logic for case assignment to MS-DRGs 453, 454, and 455 is currently satisfied with any one procedure code from the anterior spinal fusion logic list and any one procedure code from the posterior spinal fusion logic list, however, the logic lists that were developed for our analysis using the three categories of spinal fusion are comprised of specific procedure code combinations to satisfy the criteria for case assignment to any one of the three categories developed. For example, based on our analysis of MS-DRG 453 using the September update of the FY 2023 MedPAR file, the total number of cases found in MS-DRG 453 is 4,066 and with application of the logic for each of the three categories, the total number of cases in MS-DRG 453 is 4,042 (791 + 2,664 + 587 = 4,042), a difference of 24 cases. Using the September update of the FY 2023 MedPAR file, the total number of cases found in MS-DRG 454 is 20,425 and with application of the logic for each of the three categories, the total number of cases in MS-DRG 454 is 20,370 (6,481 + 12,498 + 1,391 = 20,370), a difference of 55 cases. Lastly, using the September update of the FY 2023 MedPAR file, the total number of cases found in MS-DRG 455 is 17,000 and with application of the logic for each of the three categories, the total number of cases in MS-DRG 455 is 16,987 (9,763 + 6,879 + 345 = 16,987), a difference of 13 cases. Overall, a total of 92 cases are redistributed from MS-DRGs 453, 454, ( print page 69047) and 455 to other spinal fusion MS-DRGs.
We stated in the proposed rule that the findings from our analysis of MS-DRGs 453, 454, and 455 are consistent with the expectation that clinically, the greater the number of spinal fusion procedures performed during a single procedure (for example, intervertebral levels fused), the greater the consumption of resources expended. We also stated we believed the use of interbody fusion cages, other types of spinal instrumentation, operating room time, comorbidities, pharmaceuticals, and length of stay may all be contributing factors to resource utilization for spinal fusion procedures. In addition, it is expected that as a result of potential changes to the logic for case assignment to a MS-DRG, there will be a redistribution of cases among the MS-DRGs.
We stated in the proposed rule that, based on our review and analysis of the spinal fusion cases in MS-DRGs 453, 454, and 455, we believe new MS-DRGs are warranted to differentiate between multiple level combined anterior and posterior spinal fusions except cervical, single level combined anterior and posterior spinal fusions except cervical, and combined anterior and posterior cervical spinal fusions, to more appropriately reflect utilization of resources for these procedures, including those performed with an aprevo TM custom-made anatomically designed interbody fusion device. We noted that the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device as identified by any one of the 12 previously listed procedure codes would not be reported for a cervical spinal fusion procedure as reflected in Table 6P.2f associated with the proposed rule and this final rule and available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps .
To compare and analyze the impact of our suggested modifications, we noted that we ran simulations using claims data from the September 2023 update of the FY 2023 MedPAR file. The following table illustrates our findings for all 23,017 cases reporting procedure codes describing multiple level combined anterior and posterior spinal fusions.

We stated we applied the criteria to create subgroups in a base MS-DRG as discussed in section II.C.1.b. of the preamble of the proposed rule and this final rule. We noted that, as shown in the table that follows, a three-way split of the proposed new base MS-DRG was met. The following table illustrates our findings.

For the proposed new MS-DRGs, there is (1) at least 500 or more cases in the MCC group, the CC subgroup, and in the without CC/MCC subgroup; (2) at least 5 percent of the cases are in the MCC subgroup, the CC subgroup, and in the without CC/MCC subgroup; (3) at least a 20 percent difference in average costs between the MCC subgroup and the CC subgroup and between the CC group and NonCC subgroup; (4) at least a $2,000 difference in average costs between the MCC subgroup and the with CC subgroup and between the CC subgroup and NonCC subgroup; and (5) at least a 3-percent reduction in cost variance, indicating that the proposed severity level splits increase the explanatory power of the base MS-DRG in capturing differences in expected cost between the proposed MS-DRG severity level splits by at least 3 percent and thus improve the overall accuracy of the IPPS payment system.
As a result, for FY 2025, we proposed to create new MS-DRG 426 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with MCC), new MS-DRG 427 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with CC), and new MS-DRG 428 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical without CC/MCC). The following table reflects a simulation of the proposed new MS-DRGs.

The next step in our analysis of the impact of our suggested modifications to MS-DRGs 453, 454, and 455 was to review the cases reporting single combined anterior and posterior cervical fusions. The following table ( print page 69048) illustrates our findings for all 16,059 cases reporting procedure codes describing single level combined anterior and posterior spinal fusions.

We stated we applied the criteria to create subgroups in a base MS-DRG as discussed in section II.C.1.b. of the preamble of the proposed rule and this final rule. We noted that, as shown in the table that follows, a three-way split of this proposed new base MS-DRG failed to meet the criterion that at least 5% or more of the cases are in the MCC subgroup. It also failed to meet the criterion that there be at least a 20% difference in average costs between the CC and NonCC (without CC/MCC) subgroup. The following table illustrates our findings.

As discussed in section II.C.1.b. of the preamble of the proposed rule and this final rule, if the criteria for a three-way split fail, the next step is to determine if the criteria are satisfied for a two-way split. We therefore applied the criteria for a two-way split for the “with MCC and without MCC” subgroups. We noted that, as shown in the table that follows, a two-way split of this base MS-DRG failed to meet the criterion that there be at least 5% or more of the cases in the with MCC subgroup.

We then applied the criteria for a two-way split for the “with CC/MCC and without CC/MCC” subgroups. As shown in the table that follows, a two-way split of this base MS-DRG failed to meet the criterion that there be at least a 20% difference in average costs between the “with CC/MCC and without CC/MCC” subgroup.

We noted that because the criteria for both of the two-way splits failed a split (or CC subgroup) is not warranted for the proposed new base MS-DRG. As a result, for FY 2025, we proposed to create new base MS-DRG 402 (Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical). The following table reflects a simulation of the proposed new base MS-DRG.

For the final step in our analysis of the impact of our suggested modifications to MS-DRGs 453, 454, and 455 we reviewed the cases reporting combined anterior and posterior cervical fusions. The following table illustrates our findings for all 2,323 cases reporting procedure codes describing combined anterior and posterior cervical spinal fusions.

We stated we applied the criteria to create subgroups in a base MS-DRG as discussed in section II.C.1.b. of the preamble of the proposed rule and this final rule. We noted that, as shown in the table that follows, a three-way split of this proposed new base MS-DRG failed to meet the criterion that that there be at least 500 cases in the NonCC subgroup.

As discussed in section II.C.1.b. of the preamble of the proposed rule and this final rule, if the criteria for a three-way split fail, the next step is to determine if the criteria are satisfied for a two-way split. We therefore applied the criteria for a two-way split for the “with MCC and without MCC” subgroups. We note that, as shown in the table that follows, a two-way split of this proposed new base MS-DRG was met. For the proposed MS-DRGs, there is at least (1) 500 or more cases in the MCC group and in the without MCC subgroup; (2) 5 percent or more of the cases in the MCC group and in the without MCC subgroup; (3) a 20 percent difference in average costs between the MCC group and the without MCC group; (4) a $2,000 difference in average costs between the MCC group and the without MCC group; and (5) a 3-percent reduction in cost variance, indicating that the proposed severity level splits increase the explanatory power of the base MS-DRG in capturing differences in expected cost between the proposed MS-DRG severity level splits by at least 3 percent and thus improve the overall accuracy of the IPPS payment system. The following table illustrates our findings for the suggested MS-DRGs with a two-way severity level split.

Accordingly, because the criteria for the two-way split were met, we stated we believed a split (or CC subgroup) is warranted for the proposed new base MS-DRG. As a result, for FY 2025, we proposed to create new MS-DRG 429 (Combined Anterior and Posterior Cervical Spinal Fusion with MCC) and new MS-DRG 430 (Combined Anterior and Posterior Cervical Spinal Fusion without MCC). The following table reflects a simulation of the proposed new MS-DRGs.

We then analyzed the cases reporting spinal fusion procedures in MS-DRGs 456, 457, and 458. As previously described, the logic for case assignment to MS-DRGs 456, 457, and 458 is defined by principal diagnosis logic and extensive fusion procedures. Cases reporting a principal diagnosis of spinal curvature, malignancy, or infection or an extensive fusion procedure will group to these MS-DRGs. We referred the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1 available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software for complete documentation of the GROUPER logic for MS-DRGs 456, 457, and 458.
As also previously described, in our initial analysis of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device, the 13 cases we found in MS-DRGs 456 and 457 (2 + 11 = 13, respectively) appeared to be grouping appropriately, however, the average costs for the 6 cases found in MS-DRG 458 showed a difference of approximately $13,880. Because of the low volume of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in the “without CC/MCC” MS-DRG 458, and the low volume of cases reporting the performance of a ( print page 69050) spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRGs 456, 457, and 458 overall (2 + 11 + 6 = 19), for this expanded review of the claims data, we shared the results of our analysis in association with cases reporting extensive fusion procedures in MS-DRGs 456, 457, and 458. Our findings are shown in the following table.

The data show that the 332 cases reporting an extensive fusion procedure in MS-DRG 456 have a shorter average length of stay (11.5 days versus 12.6 days) and higher average costs ($89,773 versus $76,060) compared to all the cases in MS-DRG 456. For MS-DRG 457, the data show that the 171 cases reporting an extensive fusion have a comparable average length of stay (6.6 days versus 6.1 days) and higher average costs ($75,588 versus $52,179) compared to all the cases in MS-DRG 457. Lastly, for MS-DRG 458, the data show that the 146 cases reporting an extensive fusion procedure have a comparable average length of stay (3.8 days versus 3.1 days) and higher average costs ($48,035 versus $39,260) compared to all the cases in MS-DRG 458.
In the proposed rule we stated we believe that over time, the volume of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRGs 456, 457, and 458 may increase and we could consider further in the context of the cases reporting an extensive fusion procedure. However, due to the logic for case assignment to these MS-DRGs also being defined by diagnosis code logic, additional analysis would be needed prior to considering any modification to the current structure of these MS-DRGs. We stated that as we continue to evaluate how we may refine these spinal fusion MS-DRGs, we are also seeking public comments and feedback on other factors that should be considered in the potential restructuring of MS-DRGs 456, 457, and 458. Thus, for FY 2025, we proposed to maintain the current structure of MS-DRGs 456, 457, and 458, without modification. Feedback and other suggestions for future rulemaking may be submitted by October 20, 2024 and directed to MEARIS TM at https://mearis.cms.gov/public/home .
Next, we performed an expanded analysis for spinal fusion cases reported in MS-DRGs 459 and 460. We noted that cases grouping to MS-DRG 459 have at least one secondary diagnosis MCC condition reported (“with MCC”) and because MS-DRG 460 is “without MCC”, cases grouping to this MS-DRG may include the reporting of at least one secondary diagnosis CC condition (in addition to cases that may not report a CC (for example, NonCC)). Based on the findings for a subset of the cases (that is, the subset of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device) in these MS-DRGs as previously discussed, and our review of the logic for case assignment to these MS-DRGs, we developed two categories of spinal fusion procedures to further examine. The first category was for the single level spinal fusions except cervical, and the second category was for the multiple level spinal fusions except cervical. We refer the reader to Table 6P.2g for the list of procedure codes we identified to categorize the single level spinal fusions except cervical and Table 6P.2h for the list of procedure codes we identified to categorize the multiple level spinal fusions except cervical in association with the proposed rule and available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps . Findings from our analysis are shown in the following table.

The data show that the 2,069 cases reporting a multiple level spinal fusion except cervical in MS-DRG 459 have a longer average length of stay (10.1 days versus 9.6 days) and higher average costs ($57,209 versus $53,192) when compared to all the cases in MS-DRG 459. The data also show that the 2,069 cases reporting a multiple level spinal fusion except cervical in MS-DRG 459 have a longer average length of stay (10.1 days versus 8.9 days) and higher average costs ($57,209 versus $46,031) when compared to the 1,098 cases reporting a single level spinal fusion except cervical in MS-DRG 459. For MS-DRG 460, the data show that the 14,677 cases reporting a multiple level spinal fusion except cervical have a comparable average length of stay (3.9 days versus 3.4 days) and higher average costs ($36,932 versus $32,586) when compared to all the cases in MS-DRG 460. The data also show that the 14,677 cases reporting a multiple level spinal fusion except cervical have a comparable average length of stay (3.9 days versus 3.0 days) and higher average costs ($36,932 versus $28,110) when compared to the 14,058 cases reporting a single level spinal fusion except cervical in MS-DRG 460.
In the proposed rule we stated that based on our review and analysis of the spinal fusion cases in MS-DRGs 459 and 460, we believe new MS-DRGs are warranted to differentiate between multiple level spinal fusions except cervical and single level spinal fusions except cervical to more appropriately reflect utilization of resources for these procedures, including those performed with an aprevo TM custom-made anatomically designed interbody fusion device.
To compare and analyze the impact of our suggested modifications, we ran simulations using claims data from the September 2023 update of the FY 2023 MedPAR file. The following table illustrates our findings for all 16,746 cases reporting procedure codes describing multiple level spinal fusions except cervical.

We stated we applied the criteria to create subgroups in a base MS-DRG as discussed in section II.C.1.b. of the preamble of the proposed rule and this final rule. We noted that, as shown in the table that follows, a three-way split of this proposed new base MS-DRG failed to meet the criterion that there be at least a 20% difference in average costs between the CC and NonCC (without CC/MCC) subgroup. The following table illustrates our findings.

As discussed in section II.C.1.b. of the preamble of the proposed rule and this final rule, if the criteria for a three-way split fail, the next step is to determine if the criteria are satisfied for a two-way split. We therefore applied the criteria for a two-way split for the “with MCC and without MCC” subgroups. We noted that, as shown in the table that follows, a two-way split of this proposed new base MS-DRG was met. For the proposed MS-DRGs, there is at least (1) 500 or more cases in the MCC group and in the without MCC subgroup; (2) 5 percent or more of the cases in the MCC group and in the without MCC subgroup; (3) a 20 percent difference in average costs between the MCC group and the without MCC group; (4) a $2,000 difference in average costs between the MCC group and the without MCC group; and (5) a 3-percent reduction in cost variance, indicating that the proposed severity level splits increase the explanatory power of the base MS-DRG in capturing differences in expected cost between the proposed MS-DRG severity level splits by at least 3 percent and thus improve the overall accuracy of the IPPS payment system. The following table illustrates our findings for the suggested MS-DRGs with a two-way severity level split.

As a result, for FY 2025, we proposed to create new MS-DRGs 447 (Multiple Level Spinal Fusion Except Cervical with MCC) and new MS-DRG 448 (Multiple Level Spinal Fusion Except Cervical without MCC). We also proposed to revise the title for existing MS-DRGs 459 and 460 to “Single Level Spinal Fusion Except Cervical with MCC and without MCC”, respectively. In the proposed rule we stated that this proposal would better differentiate the resource utilization, severity of illness and technical complexity between single level and multiple level spinal fusions that do not include cervical spinal fusions in the logic for case assignment. The following table reflects a simulation of the proposed new MS-DRGs.

In conclusion, we proposed to delete MS-DRGs 453, 454, and 455 and proposed to create 8 new MS-DRGs. We proposed to create new MS-DRG 426 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with MCC), MS-DRG 427 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with CC), MS-DRG 428 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical without CC/MCC), MS-DRG 402 (Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical), MS-DRG 429 (Combined Anterior and Posterior Cervical Spinal Fusion with MCC), MS-DRG 430 (Combined Anterior and Posterior Cervical Spinal Fusion without MCC), MS-DRG 447 (Multiple Level Spinal Fusion Except Cervical with MCC) and MS-DRG 448 (Multiple Level Spinal Fusion Except Cervical without MCC) for FY 2025. We proposed the logic for case assignment to these proposed new MS-DRGs as displayed in Table 6P.2d, Table 6P.2e, Table 6P.2f, Table 6P.2g, and Table 6P.2h in association with the proposed rule and available via the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps . We also proposed to revise the title for MS-DRGs 459 and 460 to “Single Level Spinal Fusion Except Cervical with MCC and without MCC”, respectively. Lastly, as discussed in section II.C.14 of the preamble of the proposed rule, we proposed conforming changes to the surgical hierarchy for MDC 08.
Comment: Commenters supported the proposed restructuring for the spinal fusion MS-DRGs for FY 2025. A commenter stated that the existing MS-DRGs have not kept pace with the rapid advancements in spine fusion technology and techniques, leading to significant financial strain on hospitals. According to the commenter, the cost differences associated with performing a one-level lumbar fusion compared to a multi-level fusion are substantial, not only in terms of the surgical time and complexity but also in postoperative care and rehabilitation. The commenter stated that the proposal represents a much-needed advancement in the payment structure for hospitals supporting these complex surgeries and acknowledges the varied complexity and resources required for these distinct types of surgeries. The commenter also stated that the proposal ensures a comprehensive approach that addresses the full spectrum of spinal fusion procedures. In addition, the commenter stated that this refined categorization will enable hospitals to receive more appropriate payment, reflecting the specific nature of each procedure and the level of care provided to patients with diverse spinal conditions. The commenter also stated that by aligning MS-DRGs more closely with the actual costs incurred, the new structure will allow hospitals to allocate resources more effectively and continue investing in high-quality patient care. Lastly, the commenter stated that the proposed changes recognize the variations in patient populations, including the different needs and recovery trajectories of those undergoing non-cervical versus cervical spine fusion surgeries.
Another commenter stated that currently, MS-DRGs 453 through 455 do not adequately differentiate between the complexity and relative resource use associated with multiple level procedures. The commenter stated that this adjustment will lead to more accurate payment, resource allocation, and further aligns with the clinical accuracy and medical advancements of these procedures.
A commenter stated it supported the proposed changes as it would create further specificity in coding. Another commenter also expressed appreciation for CMS's efforts to update the spinal fusion MS-DRGs to better reflect current clinical practice delineating single versus multiple level procedures with the detailed analysis that outlined the proposed changes. This commenter stated they plan to monitor the impact of the proposed revisions, if finalized, for both its customers and patients.
Comment: A commenter stated it reviewed the proposed spinal fusion MS-DRG changes and while it found that most of the redistribution appears appropriate, they have concerns about the proposed MS-DRG 402 (Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical) because the ability to capture the impact of a CC or MCC is not reflected. The commenter performed its own analysis and stated that the single level combined anterior and posterior fusion cases have longer lengths of stay and higher average costs when a CC or MCC is present. According to the commenter, its analysis showed that the proposed MS-DRG 402 does not adequately reflect the resource consumption for patients with significant comorbid conditions. The commenter recommended that MS-DRG 402 not be finalized as a single MS-DRG and instead suggested it be established as a three-way split MS-DRG (with MCC, with CC and without CC/MCC, respectively).
Response: We appreciate the commenter's analysis. As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35981 ), we applied the criteria to create subgroups for the proposed new base MS-DRG. The criteria for a three-way split and both two-way splits failed, therefore, only a proposed new base MS-DRG was supported.
Comment: Several commenters stated they supported CMS's review of the spinal fusion MS-DRGs to consider potential logic revisions. The commenters expressed appreciation and support for the distinction that new, revised and expanded spinal fusion MS-DRGs can provide for data analysis, notably in instances where multiple and single-level anatomically different spinal level location procedures are performed during the same operative episode. However, the commenters stated that it is essential to address and consider the logic for all the spinal fusion MS-DRGs to maintain the stability of reporting and to ensure capture of the technical complexity and medical severity indications for these procedures. The commenters requested that CMS consider delaying the proposal and provide additional insight and rationale as to why MS-DRGs 456, 457, and 458 (Spinal Fusion Except Cervical with Spinal Curvature, Malignancy, Infection or Extensive Fusions with MCC, with CC, and without CC/MCC, respectively) and MS-DRGs 471, 472, and 473 (Cervical Spinal Fusion with MCC, with CC, and without CC/MCC, respectively), were not incorporated into the analysis for FY 2025.
Response: We appreciate the commenters' support. We note that in the preamble of the FY 2025 IPPS/LTCH ( print page 69053) PPS proposed rule ( 89 FR 35971 through 35985 ) and in this final rule, as part of our ongoing analysis of the manufacturer's request to reassign cases involving the aprevo TM device, we presented findings from our analysis of claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRGs 456, 457, and 458, and cases reporting any one of the procedure codes describing the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device. We stated that based on our findings, the 13 cases we found in MS-DRGs 456 and 457 (2 + 11 = 13, respectively) appeared to be grouping appropriately, however, the average costs for the 6 cases found in MS-DRG 458 showed a difference of approximately $13,880. We also stated that, because of the low volume of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in the “without CC/MCC” MS-DRG 458, and the low volume of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRGs 456, 457, and 458 overall (2 + 11 + 6 = 19), for the expanded review of the claims data, we were sharing the results of our analysis in association with cases reporting extensive fusion procedures in MS-DRGs 456, 457, and 458. We further stated that we believed over time, that the volume of cases reporting the performance of a spinal fusion procedure using an aprevo TM custom-made anatomically designed interbody fusion device in MS-DRGs 456, 457, and 458 may increase and we could consider further in the context of the cases reporting an extensive fusion procedure. However, we also noted that due to the logic for case assignment to these MS-DRGs being defined by diagnosis code logic, additional analysis would be needed prior to considering any modification to the current structure of these MS-DRGs. We stated that as we continue to evaluate how we may refine these spinal fusion MS-DRGs, we are also seeking public comments and feedback on other factors that should be considered in the potential restructuring of MS-DRGs 456, 457, and 458. Thus, for FY 2025, we proposed to maintain the current structure of MS-DRGs 456, 457, and 458, without modification. We noted that feedback and other suggestions for future rulemaking may be submitted by October 20, 2024 and directed to MEARIS TM at https://mearis.cms.gov/public/home .
With respect to the commenters' concerns that we excluded analysis of MS-DRGs 471, 472, and 473 for FY 2025, we note that the MS-DRG request under consideration for ongoing review was related to assignment of cases reporting procedures involving use of the aprevo TM custom-made anatomically designed interbody spinal fusion device technology that is used in the performance of a spinal fusion procedure and specifically indicated for treatment of the anterior column of the thoracolumbar, lumbar, or lumbosacral vertebra. The procedure codes describing a custom-made anatomically designed interbody fusion device are not listed in the logic for case assignment to MS-DRGs 471, 472, and 473 because the logic for those MS-DRGs is specifically indicated for the cervical vertebrae. While not specifically discussed in the proposed rule, the manufacturer of the aprevo TM custom-made interbody spinal fusion device technology received a second Breakthrough Device designation for its technology in September 2023 that is indicated specifically for the treatment of patients with cervical spine disease. We anticipate, similar to the approach utilized for the treatment of patients with lumbar spine disease, that it is possible the manufacturer may request a unique procedure code(s) to describe the use of the technology for the cervical spine with the potential of applying for a new technology add-on payment and subsequent MS-DRG classification changes. For these reasons, we believe additional time is necessary as we consider how we may refine the cervical spinal fusion MS-DRGs. We are also seeking feedback on factors that should be considered in the potential restructuring of MS-DRGs 471, 472, and 473 for future rulemaking. For example, are there other patient-specific spinal fusion technologies currently in development or in use and indicated for cervical spine disease that should also be evaluated and considered. Feedback and other suggestions for future rulemaking may be submitted by October 20, 2024 and directed to MEARIS TM at https://mearis.cms.gov/public/home .
Comment: A few commenters who appreciated CMS's attempts to recognize the differences in complexity between single level and multiple level spinal fusion procedures stated their belief that additional time is needed for hospitals to assess the impact of the proposed changes. According to the commenters, the proposed changes may have a negative impact on community hospitals, which they stated tend to treat less-complex cases.
A couple commenters stated that CMS has previously given two years notice to hospitals about potential changes and provided the example of CMS's request for public comments and feedback on potential restructuring for MS-DRGs 023 through 027, as discussed in FY 2024 and FY 2025 rulemaking. Another commenter stated that the proposed restructuring of the spinal fusion MS-DRGs is a major revision, and without any warning to hospitals. However, this commenter also stated that regardless of the outcome for the proposed reorganization of the spinal fusion MS-DRGs, CMS should address the resource utilization disparity related to the use of the aprevo TM custom-made anatomically designed spine fusion devices. According to the commenter, failure to implement this issue for FY2025 will create a financial disincentive for hospitals to utilize this innovative technology, thus eliminating access for patients. The commenter recommended CMS reassign cases reporting a custom-made anatomically designed interbody fusion device to MS-DRGs that address the higher resource utilization and to ensure continued access to the technology. This same commenter also stated its belief that the proposal should undergo a comprehensive review by a spine group to identify and mitigate any unintended consequences.
Response: We appreciate the commenters' feedback. As discussed in prior rulemaking ( 86 FR 44878 ), the MS-DRG system is a system of averages and it is expected that within the diagnostic related groups, some cases may demonstrate higher than average costs, while other cases may demonstrate lower than average costs. It is generally expected that as a result of the annual MS-DRG reclassifications that are finalized, the experience of different categories of hospitals may differ based on the population of patients they treat and the services offered by the facility.
With respect to the commenter's concern that hospitals had no warning regarding the proposed restructuring for a subset of the spinal fusion MS-DRGs, we note that in addition to proposing these changes in the FY 2025 IPPS/LTCH PPS proposed rule, we discussed this topic in the FY 2024 IPPS/LTCH PPS rulemaking, including noting that our work in this area was ongoing, and that we would continue to examine the data and consider these issues as we develop potential future rulemaking proposals. Providers have had the opportunity to consider how spinal fusion cases (including cases reporting ( print page 69054) the use of a custom-made anatomically designed interbody fusion device) are reported in the claims data for their respective facilities and grouped under the IPPS MS-DRGs, as well as to submit requested changes to the classifications for these MS-DRGs for CMS's consideration. We further note that, as stated in the preamble of the annual IPPS rulemakings, section 1886(d)(4)(C) of the Act requires that the Secretary adjust the DRG classifications and relative weights at least annually to account for changes in resource consumption. These adjustments are made to reflect changes in treatment patterns, technology, and any other factors that may change the relative use of hospital resources. We include these changes as part of our annual IPPS rulemaking, which provides the public, including any particular interested parties, the opportunity to review and comment on these proposals.
Comment: A few commenters who expressed appreciation for CMS's efforts to update the spinal fusion MS-DRGs to better reflect current clinical practice and facility costs more accurately stated they need more information about the potential impact of the proposed designations and the opportunity to study the proposed changes further. These commenters recommended that CMS not conduct this restructuring while also considering the spinal fusion episode accountability model under the Transforming Episode Accountability Model (TEAM).
Response: We appreciate the commenters' feedback. We refer the reader to section X.A.3.b. of the preamble of this final rule for further discussion of how the proposed restructuring of the spinal fusion MS-DRGs may be considered in connection with the spinal fusion episode category under TEAM.
Comment: A few commenters who expressed support for potential changes to the logic for case assignment to the spinal fusion MS-DRGs stated they reviewed data provided by CMS with the AOR/BOR (After Outliers Removed/Before Outliers Removed) version 41 and version 42 files, and Table 5—Proposed List of Medicare Severity Diagnosis Related Groups (MS-DRGs), Relative Weighting Factors, and Geometric and Arithmetic Mean Length of Stay that was made available in association with the proposed rule. The commenters stated it was unclear if the current proposed spinal fusion MS-DRGs better reflect resource consumption based on its findings of a minimal change in the case mix index between version 41 (4.6504) and version 42 (4.6454). The commenters suggested further analysis of all the spinal fusion MS-DRGs should be considered.
A commenter stated that the version 42 AOR table showed more spinal fusion cases in comparison to the total spinal fusion cases included in the rule discussion. The commenter questioned if there was duplication of the same patients being counted based on the logic lists for the proposal and stated it was not clear how duplications may have been handled in the data if there was both a multiple level fusion and single level fusion reported on the same case.
Response: It is not entirely clear how the commenters performed the case-mix index calculations, however, based on the data table provided by the commenters, we believe the commenters used the case counts from the AOR file and relative weights to calculate a case-weighted average relative weight for the spinal fusion MS-DRGs and are referring to that as a case-mix index. We note that under the proposed restructuring, the same population of cases among the spinal fusion MS-DRGs is being redistributed, therefore, we would not expect a significant shift in the case-mix index.
With respect to the differences in case counts between the version 42 AOR table in comparison to the number of cases included in the rule discussion for the proposed spinal fusion MS-DRGs, we note that, as stated in the proposed rule, our MS-DRG analysis was based on ICD-10 claims data from the September 2023 update of the FY 2023 MedPAR file, which contains hospital bills received from October 1, 2022, through September 30, 2023. In comparison, as also stated in the proposed rule, the FY 2023 MedPAR file used in developing the proposed MS-DRG relative weights for FY 2025 included discharges occurring on October 1, 2022, through September 30, 2023, based on bills received by CMS through December 31, 2023.
Comment: A commenter noted that the titles of the ICD-10-PCS procedure codes that were created to report spinal fusion procedures using the aprevo TM customized interbody fusion device were revised, effective October 1, 2023, as a result of the manufacturer's concerns that some claims may have been unintentionally miscoded. The commenter stated that per the materials from the March 2023 ICD-10 Coordination and Maintenance Committee meeting, the manufacturer requested the title revision to help minimize misinterpretation of the term “customizable.” The commenter remarked that CMS was unable to confirm that claims reporting any one of the codes created that describe use of the aprevo TM device had in fact been miscoded, and as discussed in the proposed rule, while a newly established ICD-10 code may be associated with an application for a new technology add-on payment, such codes are not generally established to be product specific. The commenter added that CMS further stated that if, after consulting the official coding guidelines, a provider determines that an ICD-10 code associated with a new technology add-on payment describes the technology that they are billing, the hospital may report the code and be eligible to receive the associated add-on payment. The commenter stated that some ICD-10-PCS codes are intended to be product specific, as the code title(s) often represent a manufacturer's specific technology, particularly in the New Technology section. The commenter added that the Coding Clinic for ICD-10-CM/PCS Editorial Advisory Board has determined that some ICD-10-PCS codes are only intended for a specific product and should not be used for other devices or substances.
The commenter stated that in the case of the aprevo TM device, it is not clear why the titles of the associated procedure codes were revised to more clearly describe this specific device and address the manufacturer's concerns regarding miscoding, if it was appropriate to assign the codes for spinal fusion procedures using devices other than the aprevo TM device. The commenter further stated that absence of clarity regarding device specific codes may have an unintended effect on the use of new technology due to concerns regarding lack of payment, which they stated may have a negative impact on clinical outcomes.
Response: We appreciate the commenter's feedback. With respect to the commenter's remarks about revisions made to the code title for the procedure codes describing spinal fusion procedures with an aprevo TM interbody fusion device, we note that we addressed this issue when we were made aware of it and believe the code title is now appropriate. It was brought to our attention that the term “customizable” as reflected in the original code title was leading to confusion with devices that utilize expandable cages and are “customized” to fit during the procedure. In response to the manufacturer's concerns regarding potential miscoded claims and its request to revise the original code descriptor to help minimize misinterpretation of the term “customizable” by providers' coding personnel, we presented and received ( print page 69055) public support to finalize the proposed revision to the code titles. The intent was not to specifically limit the reporting of the code, since, as stated in the proposed rule, while a newly established ICD-10 code may be associated with an application for a new technology add-on payment, such codes are not generally established to be product specific.
We note that historically, our approach to proposing and finalizing new procedure codes through the ICD-10 Coordination and Maintenance Committee meeting process was largely built on the fact that the procedure classification system was designed to report the procedure performed, not the device or other specific technology used. However, we also note that with the implementation of the new technology add-on payment policy, aspects of that approach to creating new procedure codes have been become more complex. While we have strived to maintain consistency with that historical approach, we also recognize the responsibility to balance and support the requirements of the new technology add-on payment policy, which have continued to evolve since its inception.
The commenter is correct that certain ICD-10-PCS codes located in the New Technology section of the ICD-10-PCS procedure classification, also known as “Section X”, are product specific. For example, a procedure code request for the administration of a therapeutic agent, regardless of it being related to a new technology add-on payment application, is often presented as a proposal through the ICD-10 Coordination and Maintenance Committee meeting process, and subsequently finalized (following review and consideration of the public comments) with the generic name of the agent in the code description (title). Oftentimes, there is a clinical need and several benefits to capture a certain level of specificity for purposes of data collection, such as tracking a particular patient population, or assessing clinical outcomes. We note that following the finalization of a new procedure code that is classified within the new technology section (Section X) of ICD-10-PCS, we discuss the disposition of that code after a 3-year period during a future ICD-10 Coordination and Maintenance Committee meeting, which also generally aligns with the expiration of a product's eligibility for an add-on payment under the new technology add-on payment policy. We also take this opportunity to point out that a procedure, service, or technology is not required to submit a new technology add-on payment application for consideration of a Section X code. As discussed in prior rulemaking ( 80 FR 49434 through 49435 ), when the ICD-10-PCS New Technology section was under development, we established that the purpose of the New Technology section is to also provide a mechanism to capture services that would not normally be coded and reported in the inpatient setting.
We appreciate the commenter's feedback on this topic and will continue to consider how to better address coding proposals in connection with new technologies for future discussion at the ICD-10 Coordination and Maintenance Committee meeting.
Comment: A commenter (the manufacturer of the aprevo TM custom-made anatomically designed interbody fusion device) stated that while CMS partially addressed the request to assign spinal fusion procedures reporting the use of a custom-made anatomically designed interbody fusion device to appropriate MS-DRGs that more closely align with the increase in resource utilization, the analysis under the proposed restructuring did not specifically reflect data related to the resource utilization for custom-made anatomically designed devices under the single level versus multiple level MS-DRG construct.
The commenter provided a comprehensive list detailing the sequence of events related to prior rulemaking discussions involving custom-made anatomically designed interbody fusion devices including its approved eligibility for new technology add-on payments, revisions to the procedure code title to change the description from “customizable” to “custom-made anatomically designed” interbody fusion device, and prior data analysis findings. The commenter also provided extensive clinical background on custom-made anatomically designed interbody fusion devices and reiterated the designation as an FDA Breakthrough technology. Additionally, the commenter stated that published clinical data has shown that custom-made anatomically designed interbody fusion devices improve care by delivering more precise patient specific alignment, [ 6 7 ] which they stated has been proven to reduce the risk of revision surgery.
In response to publication of the FY 2025 IPPS/LTCH PPS proposed rule, the commenter stated its belief that 1.) CMS contradicted its position on the original description of the procedure codes by making the statement in the FY 2025 IPPS/LTCH PPS proposed rule that a newly established ICD-10 code may be associated with an application for new technology add-on payment and such codes are not generally established to be product specific and 2.) CMS acknowledged that the description used in the original ICD-10 code inadvertently described several types of technologies, and this likely contributed to the miscoded claims. According to the commenter, because CMS decided to consider resource utilization disparities for all cases reporting the use of a custom-made anatomically designed interbody spinal fusion device in its analysis for FY 2025 (that is, they stated CMS did not limit its analysis to cases associated only with the list of providers provided by the manufacturer), the commenter's original requested reassignments are no longer supported by data and therefore, the commenter stated revised reassignments are appropriate to request.
The commenter stated that CMS sought to find an alternative explanation for the resource incoherence demonstrated across the cases reporting any one of the 12 procedure codes describing a spinal fusion procedure with a custom-made anatomically designed interbody spinal fusion device and that the expanded analysis was unrelated to the original request because it did not provide data related to the use of custom-made anatomically designed devices under the proposed single level versus multiple level MS-DRG construct. The commenter further stated that the absence of this specific data (single level versus multiple level) in the proposed rule necessitated the submission of a revised request under the proposed new structure and the findings from its analysis for CMS's review and consideration.
The commenter provided prior examples of MS-DRG classification requests comparing length of stay differences and low claims volume to demonstrate instances for which CMS reassigned cases from a lower severity level MS-DRG to a higher severity level MS-DRG, including the proposal regarding the Neuromodulation Device Implant for Heart Failure (Barostim TM Baroreflex Activation Therapy), as discussed in the preamble of the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35959 through 35962 ) and in section II.C.4.b. of the preamble of this final rule.
The commenter also remarked on CMS's discussion of the data analysis presented in the proposed rule regarding the wide range in average costs for claims reporting the use of a ( print page 69056) custom-made anatomically designed interbody fusion device. The commenter stated it engaged a contractor to assess the distribution of costs and length of stay for all spinal fusion cases and cases reporting the use of a custom-made anatomically designed interbody fusion device using FY 2023 Q1-Q4 inpatient standard analytical file (SAF) data. According to the commenter, the findings from its analysis demonstrate that cases reporting the use of a custom-made anatomically designed interbody fusion device consistently show higher average costs in comparison to the average costs of all spinal fusion cases in their respective MS-DRG, which they stated are an indication that the higher costs are not an artifact of a few cases.
The commenter conducted additional analyses using the FY 2023 MedPAR data with the logic lists from the tables provided in association with the proposed rule and stated that its findings demonstrate disparities in resource utilization for cases reporting use of a custom-made anatomically designed interbody fusion device among the proposed multiple level and single level spinal fusion MS-DRGs. Specifically, the commenter stated cases reporting the use of a custom-made anatomically designed interbody fusion device under the proposed MS-DRG structure should be reassigned as shown in the table that follows.

According to the commenter, findings from its analysis under the proposed MS-DRG structure support the reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from the lower severity proposed MS-DRGs to the higher severity level proposed MS-DRGs because the resource utilization of cases reporting the use of a custom-made anatomically designed interbody fusion device align more closely with the resource utilization of cases in the requested MS-DRG. The commenter stated that the requested reassignments are consistent with other MS-DRG classifications CMS has previously finalized and therefore, the precedent exists.
The commenter also provided an alternative recommendation for CMS's consideration based on the current, existing spinal fusion MS-DRGs, with minor modifications from its initial FY 2024 request for the reassignment of cases reporting a custom-made anatomically designed interbody fusion device. Specifically, the commenter provided its analysis under the existing MS-DRGs and indicated that cases reporting the use of a custom-made anatomically designed interbody fusion device under the existing MS-DRG structure should be considered for reassignment as shown in the table that follows, if the proposed structure is not finalized.

Based on the findings from its analyses under the proposed and current MS-DRG structure for spinal fusions, the commenter asserted that reassignment of cases reporting the use of a custom-made anatomically ( print page 69057) designed interbody fusion device is supported by compelling data that demonstrates a resource utilization disparity for cases reporting the technology. The commenter stated that without appropriate payment, Medicare beneficiaries will lose access to the technology, and stated they deserve continued access to the technology because it improves patient care. The commenter urged CMS to finalize the reassignment of these cases for FY 2025.
Some commenters stated that the new technology add-on payment for custom-made anatomically designed interbody spinal fusion devices is ending on September 30, 2024, and if CMS decides to move forward with the proposed MS-DRG changes without the reassignment of the procedure codes describing the custom-made anatomically designed technology to more appropriate MS-DRGs, it would create a financial disincentive for hospitals and eliminate access to the breakthrough technology for patients. The commenters reiterated prior concerns raised in public comments by spine surgeons that were discussed in the FY 2024 rulemaking and stated that without adequate payment, hospitals will not authorize use of the technology.
A few commenters suggested that if CMS is going to finalize the proposed restructuring, consideration be given to deleting MS-DRGs 459 and 460 and creating new MS-DRGs for single level spinal fusion except cervical with MCC and without MCC because they stated the proposed revisions would significantly change the types of cases classified to these MS-DRGs.
Response: We appreciate the commenters' feedback. In response to the commenter's statement that CMS contradicted its position on the original description of the procedure codes, we note that the manufacturer contacted CMS about its concerns. CMS' actions were to provide clarity to all parties in light of concerns that the manufacturer raised. As a general matter, CMS aims to provide clarity when possible, and we recognized there could be impacts to coding, data collection, and payment, and therefore we took the opportunity to revise the code title in this case. Specifically, as stated above, in response to the manufacturer's concerns regarding potential miscoded claims and its request to revise the original code descriptor to help minimize misinterpretation of the term “customizable” by providers' coding personnel, we presented and received public support to finalize the proposed revision to the code titles. We wish to clarify that CMS did not specifically acknowledge that the description used in the original ICD-10 code inadvertently described several types of technologies, and that this likely contributed to the miscoded claims. As discussed in the proposed rule and previously in this final rule, we provided clarification that finalization of the revised code title was not intended to specifically limit the reporting of the code, since a newly established ICD-10 code that may be associated with an application for a new technology add-on payment is generally not established to be product specific.
We disagree with the commenter's assertion that CMS contradicted its prior position on length of stay differences with respect to clinical coherence. We note that in the examples provided by the commenter of MS-DRG classification requests comparing length of stay differences and low claims volume to demonstrate instances for which CMS reassigned cases from a lower severity level MS-DRG to a higher severity level MS-DRG, the topics were discussed and considered in more than one rulemaking cycle prior to finalizing the reassignment of cases from the lower severity level to the higher severity level and length of stay was still a factor under consideration. We also note that because of the lag in claims data used in our analysis of MS-DRG classification requests, depending on the specific procedures and technology under consideration, it is not uncommon to delay a decision and continue to monitor the data until additional analysis can be performed.
In this case, CMS performed additional analyses to examine if other factors could be identified as contributing to the increased resource utilization for cases reporting any one of the 12 procedure codes describing a spinal fusion procedure with a custom-made anatomically designed interbody spinal fusion device. We disagree that the expanded analysis was unrelated to the original request because it did not specifically provide data related to the use of custom-made anatomically designed devices under the proposed single level versus multiple level MS-DRG construct, however, we appreciate the commenter's submission of suggested alternative reassignments under the proposed new structure and optional consideration under the existing structure.
In response to the commenter's request to reassign cases reporting the use of a custom-made anatomically designed interbody fusion device under the proposed restructuring for the spinal fusion MS-DRGs, we analyzed claims data from the September update of the FY 2023 MedPAR file for proposed MS-DRGs 402, 426, 427, 428, 447, 448, 459 and 460 and cases reporting spinal fusion using a custom-made anatomically designed interbody fusion device. Our findings are shown in the following table.

The findings show that the 307 cases reporting a spinal fusion procedure using a custom-made anatomically designed interbody fusion device in MS-DRGs 402, 426, 427, 428, 447, 448, 459 and 460 have higher average costs in comparison to the average costs of all the cases in their respective proposed MS-DRG. We note, as shown in the table, that there were zero cases found to report the use of a custom-made anatomically designed interbody fusion device in proposed revised MS-DRG 459. For proposed MS-DRGs 402 and 428, the findings show that the cases reporting the use of a custom-made anatomically designed interbody fusion device have a comparable average length of stay compared to all the cases in their respective proposed MS-DRG. The findings also show that for proposed MS-DRGs 426 and 427, the cases reporting the use of a custom-made anatomically designed interbody fusion device have a longer average length of stay compared to all the cases in their respective proposed MS-DRG. For proposed MS-DRG 447, we note that the single case reporting the use of a custom-made anatomically designed interbody fusion device is an outlier. For proposed MS-DRG 448 and proposed revised MS-DRG 460, cases reporting the use of a custom-made anatomically designed interbody fusion device have a shorter average length of stay compared to all the cases in their respective proposed MS-DRG.
We reviewed the requested reassignment for the 66 cases from proposed MS-DRG 402 to proposed MS-DRG 428 and note that the logic for case assignment to proposed MS-DRG 428 is comprised of cases reporting a multiple level combined anterior and posterior fusion (except cervical) without a CC/MCC and the logic for case assignment for proposed MS-DRG 402 is comprised of cases reporting a single level combined anterior and posterior fusion (except cervical) that may also ( print page 69059) have an MCC or CC reported since it is a proposed base MS-DRG that is not subdivided by severity. The proposed logic for case assignment to each of these proposed MS-DRGs includes the procedure codes describing the use of a custom-made anatomically designed interbody fusion device in the definition of the respective proposed MS-DRG. Therefore, the reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed MS-DRG 402 to proposed MS-DRG 428 would not be feasible and would not be consistent with the logic of the proposed MS-DRGs which is intended to differentiate a single level combined anterior and posterior fusion from a multiple level combined anterior and posterior spinal fusion.
Next, we reviewed the requested reassignment for the 51 cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed MS-DRG 428 (without CC/MCC) to proposed MS-DRG 427 (with CC) and for the 101 cases from proposed MS-DRG 427 (with CC) to proposed MS-DRG 426 (with MCC). We note that because the proposed MS-DRGs are subdivided with a three-way split, it is not feasible to reassign cases reporting the use of a custom-made anatomically designed interbody fusion device as requested at this time. Generally, with a three-way split, the requested reassignment of cases can only be considered for movement from one severity level to the next highest severity level. For example, consideration could be given to reassign cases from the “without CC/MCC” severity level to the “with CC” severity level or from the “with CC” level to the “with MCC” severity level. Because the proposed logic lists for case assignment to each of these proposed MS-DRGs includes the procedure codes describing the use of a custom-made anatomically designed interbody fusion device in the definition of the respective proposed MS-DRG, the GROUPER software is not able to exclude cases reporting a custom-made anatomically designed interbody fusion device from grouping to proposed MS-DRG 426 (“with MCC”) that would otherwise group to proposed MS-DRG 428 (“without CC/MCC”).
We then reviewed the requested reassignment for the 38 cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed revised MS-DRG 460 to proposed MS-DRG 447. We note that the logic for case assignment to proposed MS-DRG 447 is comprised of cases reporting a multiple level spinal fusion (except cervical) and the logic for case assignment for proposed revised MS-DRG 460 is comprised of cases reporting a single level spinal fusion (except cervical). The proposed logic for case assignment to each of these proposed MS-DRGs includes the procedure codes describing the use of a custom-made anatomically designed interbody fusion device in the definition of the respective proposed MS-DRG. Therefore, the reassignment of the 38 cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed revised MS-DRG 460 to proposed MS-DRG 447 would not be feasible and would not be consistent with the logic of the proposed MS-DRGs which is intended to differentiate a single level spinal fusion from a multiple level spinal fusion.
Lastly, we reviewed the requested reassignment for the 26 cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed MS-DRG 448 to proposed MS-DRG 447. Based on the logic lists for case assignment and because these MS-DRGs are subdivided by a two-way split that both describe multiple level spinal fusion (except cervical), we determined it would be feasible to reassign cases from the “without MCC” severity level (MS-DRG 448) to the “with MCC” severity level (MS-DRG 447).
As previously described, when MS-DRGs are subdivided with a three-way split, the requested reassignment of cases can only be considered from one severity level to the next highest severity level. In our review of the data for proposed MS-DRGs 426, 427, and 428, we considered the average costs of the 24 cases found in proposed MS-DRG 426 reporting the use of a custom-made anatomically designed interbody fusion device compared to the average cost of all the cases in proposed MS-DRG 426 ($103,956 versus $91,358) and the average costs of the 101 cases found in proposed MS-DRG 427 reporting the use of a custom-made anatomically designed interbody fusion device compared to the average cost of all the cases in proposed MS-DRG 427 ($76,827 versus $64,065). Although the average length of stay for cases reporting a custom-made anatomically designed interbody fusion device in proposed MS-DRG 427 is shorter in comparison to the average length of stay of all the cases in proposed MS-DRG 426, we believe the reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed MS-DRG 427 (with CC) to proposed MS-DRG 426 (with MCC) is supported and better reflects the resource utilization and complexity of cases using the custom-made anatomically designed interbody fusion device technology in a multiple level combined anterior and posterior spinal fusion. We recognize that the 51 cases found in proposed MS-DRG 428 reporting the use of a custom-made anatomically designed interbody fusion device have higher average costs compared to the average cost of all the cases in MS-DRG 428 ($64,038 versus $50,097), however, as previously described, we are unable to accommodate two severity level reassignment requests for an MS-DRG subdivided by a three-way split at this time.
We noted earlier in this section of the preamble of this final rule, in our review of the requested reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed MS-DRG 448 to proposed MS-DRG 447 that the request was feasible based on the logic of the proposed MS-DRGs that are subdivided with a two-way split. In our review of the data for proposed MS-DRGs 447 and 448, we considered the average costs of the 26 cases found in proposed MS-DRG 448 reporting the use of a custom-made anatomically designed interbody fusion device compared to the average cost of all the cases in proposed MS-DRG 448 ($62,831 versus $36,932). We also considered the one case found in proposed MS-DRG 447 reporting the use of a custom-made anatomically designed interbody fusion device to be an outlier with costs of $288,499 compared to the average costs of all the cases in proposed MS-DRG 447 ($57,209). We believe the reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed MS-DRG 448 (without MCC) to proposed MS-DRG 447 (with MCC) is supported and better reflects the resource utilization and complexity of cases using the custom-made anatomically designed interbody fusion device technology in a multiple level anterior and posterior spinal fusion.
As previously discussed, we determined that the requested reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed revised MS-DRG 460 to proposed MS-DRG 447 would not be feasible based on the logic for case assignment. However, based on the data findings, we believe it is appropriate to consider the reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed revised MS-DRG ( print page 69060) 460 to proposed revised MS-DRG 459. In our review of the data for proposed revised MS-DRGs 459 and 460, we considered the average costs of the 38 cases found in proposed revised MS-DRG 460 reporting the use of a custom-made anatomically designed interbody fusion device compared to the average cost of all the cases in proposed revised MS-DRG 460 ($47,138 versus $32,586). While there were no cases found to report the use of a custom-made anatomically designed interbody fusion device in proposed revised MS-DRG 459, we considered the average costs of all the cases in proposed revised MS-DRG 459 ($53,192). While the average length of stay of the cases reporting a custom-made anatomically designed interbody fusion device are shorter (2.1 days versus 9.6 days), we believe the reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed revised MS-DRG 460 (without MCC) to proposed revised MS-DRG 459 (with MCC) is supported and better reflects the resource utilization of cases using the custom-made anatomically designed interbody fusion device technology in a single level spinal fusion. As also previously discussed, a few commenters suggested that if the proposed restructuring was to be finalized, consideration be given to deleting proposed revised MS-DRGs 459 and 460 and creating new MS-DRGs for single level spinal fusion except cervical with MCC and without MCC, respectively, because the proposed revisions would significantly change the types of cases classified to these MS-DRGs. We agree with the commenters that the proposed revisions to the MS-DRG logic change the types of cases that would be classified to proposed revised MS-DRGs 459 and 460. Specifically, because the logic for case assignment to existing MS-DRGs 459 and 460 was proposed to be restructured to better differentiate between single level spinal fusions (except cervical) and multiple level spinal fusions (except cervical), it would not be appropriate to retain the existing MS-DRG numbers 459 and 460 with revised titles. We proposed to create new MS-DRGs 447 and 448 to reflect multiple level spinal fusion procedures (except cervical) therefore, maintaining the existing MS-DRG numbers of 459 and 460 for the single level spinal fusions (except cervical) logic only could potentially result in confusion about the logic for case assignment. If users were to reference MS-DRG numbers 459 and 460 only, in the absence of the full MS-DRG titles, others may not be aware that the logic for case assignment to these MS-DRGs had changed effective FY 2025. As such, we agree that existing MS-DRG numbers 459 and 460 should be deleted.
We recognize that with the requested reassignments the average length of stay for cases reporting a custom-made anatomically designed interbody fusion device varies from the average length of stay for all the cases in the requested MS-DRGs, and we continue to believe that length of stay is a factor in assessing clinical coherence, however, we also consider the use of a specific technology in the performance of a procedure as a measure of complexity in connection with resource consumption, particularly when that technology is indicated for a specific population. In the case of custom-made anatomically designed interbody fusion devices, the technology is indicated for patients who have complicated spinal anatomy necessitating individualized treatment plants to precisely address spinal alignment needs and reduce the risk of revision surgery.
After consideration of the public comments we received, we are finalizing our proposal to delete MS-DRGs 453, 454, and 455 and to create new MS-DRGs 426, 427, and 428, with modification, for FY 2025. Specifically, we are finalizing our proposal with modification to assign cases reporting the use of a custom-made anatomically designed interbody fusion device with a CC to new MS-DRG 426. Conforming changes to the GROUPER logic are also are shown in Table 6P.2e associated with this final rule and available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps and also as reflected in the final version of ICD-10 MS-DRG Definitions Manual, version 42, available in association with this final rule and available via the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software . Accordingly, the finalized MS-DRG titles are MS-DRG 426 “Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with MCC or Custom-Made Anatomically Designed Interbody Fusion Device”, MS-DRG 427 “Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with CC” and MS-DRG 428 “Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical without CC/MCC” effective October 1, 2024, for FY 2025.
We are also finalizing our proposal to create new MS-DRGs 447 and 448, with modification, for FY 2025. Specifically, we are finalizing our proposal with modification to assign cases reporting the use of a custom-made anatomically designed interbody fusion device without an MCC to MS-DRG 447. Conforming changes to the GROUPER logic are shown in Table 6P.2h associated with this final rule and available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps and also reflected in the final version of ICD-10 MS-DRG Definitions Manual, version 42, available in association with this final rule and available via the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software . Accordingly, the finalized MS-DRG titles are MS-DRG 447 “Multiple Level Anterior and Posterior Spinal Fusion Except Cervical with MCC or Custom-Made Anatomically Designed Interbody Fusion Device” and MS-DRG 448 “Multiple Level Anterior and Posterior Spinal Fusion Except Cervical without MCC” effective October 1, 2024, for FY 2025.
As previously discussed, we stated we believe the reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device from proposed revised MS-DRG 460 (without MCC) to proposed revised MS-DRG 459 (with MCC) is supported and agree with the commenters that the proposed revisions to the MS-DRG logic change the types of cases that would be classified to MS-DRGs 459 and 460. As previously noted, the logic for case assignment to existing MS-DRGs 459 and 460 was proposed to be restructured to better differentiate between single level and multiple level spinal fusions, therefore it would not be appropriate to retain the existing MS-DRG numbers 459 and 460 with revised titles because the cases that group to these MS-DRGs would change. Therefore, for FY 2025, we are deleting MS-DRGs 459 and 460, and finalizing the creation of MS-DRGs 450 and 451. The logic for case assignment to MS-DRGs 450 and 451 is comprised of the logic lists that were initially proposed for revised MS-DRGs 459 and 460, with modification. We are also finalizing the assignment of cases reporting the use of a custom-made anatomically designed interbody fusion device without an MCC to MS-DRG 450. Conforming changes to the GROUPER logic are shown in Table 6P.2g associated with this final rule and available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute- ( print page 69061) inpatient-pps and also reflected in the final version of ICD-10 MS-DRG Definitions Manual, version 42, available in association with this final rule and available via the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software . Accordingly, the finalized MS-DRG titles are MS-DRG 450 “Single Level Spinal Fusion Except Cervical with MCC or Custom-Made Anatomically Designed Interbody Fusion Device” and MS-DRG 451 “Single Level Spinal Fusion Except Cervical without MCC” effective October 1, 2024, for FY 2025.
We are also finalizing our proposal to create new MS-DRG 402, and new MS-DRGs 429 and 430, without modification, for FY 2025. Accordingly, we are finalizing the proposed GROUPER logic for these MS-DRGs as shown in Table 6P.2d and 6P.2f, respectively, associated with this final rule and available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps and as also reflected in the final version of ICD-10 MS-DRG Definitions Manual, version 42, available in association with this final rule and available via the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software . The finalized MS-DRG titles are MS-DRG 402 “Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical”, MS-DRG 429 “Combined Anterior and Posterior Cervical Spinal Fusion with MCC” and MS-DRG 430 “Combined Anterior and Posterior Cervical Spinal Fusion without MCC” effective October 1, 2024, for FY 2025. We will continue to monitor the data for these finalized MS-DRGs and consider if any future modifications may be warranted.
In the proposed rule, we noted that we identified an inconsistency in the MDC and MS-DRG assignment of procedure codes describing resection of the right large intestine and resection of the left large intestine with an open and percutaneous endoscopic approach. ICD-10-PCS procedure codes 0DTG0ZZ (Resection of left large intestine, open approach) and 0DTG4ZZ (Resection of left large intestine, percutaneous endoscopic approach) are currently assigned to MDC 10 in MS-DRGs 628, 629, and 630 (Other Endocrine, Nutritional and Metabolic O.R. Procedures with MCC, with CC, and without CC/MCC, respectively). However, the procedure codes that describe resection of the right large intestine with an open or percutaneous endoscopic approach, 0DTF0ZZ (Resection of right large intestine, open approach) and 0DTF4ZZ (Resection of right large intestine, percutaneous endoscopic approach) are not assigned to MDC 10 in MS-DRGs 628, 629, and 630. To ensure clinical alignment and consistency, as well as appropriate MS-DRG assignment, we proposed to add procedure codes 0DTF0ZZ and 0DTF4ZZ to MDC 10 in MS-DRGs 628, 629, and 630 effective October 1, 2024, for FY 2025.
Comment: Commenters supported our proposal to add procedure codes 0DTF0ZZ and 0DTF4ZZ to MDC 10 in MS-DRGs 628, 629, and 630. A commenter also suggested that CMS consider providing an index of ICD-10-PCS codes that are assigned to each MDC in the ICD-10 MS-DRG Definitions Manual in a “reverse look up” format that could be utilized to identify other potential omissions or inaccuracies such as the issues discussed in the proposed rule. The commenter urged CMS to make this information publicly available in a user-friendly format to enable interested parties to review the MDC and MS-DRG assignments more easily for ICD-10-PCS procedure codes.
Response: We thank the commenters for their support. With respect to the commenter's suggestion that CMS develop a “reverse look up” index of the ICD-10-PCS procedure codes to enable members of the public to more easily review the MDC and MS-DRG assignments of the procedure codes, we appreciate the feedback and will take the suggestion under advisement.
After consideration of the public comments we received, we are finalizing our proposal to add procedure codes 0DTF0ZZ and 0DTF4ZZ to MDC 10 in MS-DRGs 628, 629, and 630 effective October 1, 2024, for FY 2025.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35985 through 35991 ), we received a request to review the GROUPER logic that would determine the assignment of cases to MS-DRG 794 (Neonate with Other Significant Problems). The requestor stated that it appears that MS-DRG 794 is the default MS-DRG in MDC 15 (Newborns and Other Neonates with Conditions Originating in Perinatal Period), as the GROUPER logic for MS-DRG 794 displayed in the ICD-10 MS-DRG Version 41.1 Definitions Manual is defined by a “principal or secondary diagnosis of newborn or neonate, with other significant problems, not assigned to DRG 789 through 793 or 795”. The requestor expressed concern that defaulting to MS-DRG 794, instead of MS-DRG 795 (Normal Newborn), for assignment of cases in MDC 15 could contribute to overpayments in healthcare by not aligning the payment amount to the appropriate level of care in newborn cases. The requestor recommended that CMS update the GROUPER logic that would determine the assignment of cases to MS-DRGs in MDC 15 to direct all cases that do not have the diagnoses and procedures as specified in the Definitions Manual to instead be grouped to MS-DRG 795.
Specifically, as discussed in the proposed rule, the requestor expressed concern that a newborn encounter coded with a principal diagnosis code from ICD-10-CM category Z38 (Liveborn infants according to place of birth and type of delivery), followed by code P05.19 (Newborn small for gestational age, other), P59.9 (Neonatal jaundice, unspecified), Q38.1 (Ankyloglossia), Q82.5 (Congenital non-neoplastic nevus), or Z23 (Encounter for immunization) is assigned to MS-DRG 794. The requestor stated that they performed a detailed claim level study, and in their clinical assessment, newborn encounters coded with a principal diagnosis code from ICD-10-CM category Z38, followed by diagnosis code P05.19, P59.9, Q38.1, Q82.5, or Z23 in fact clinically describe normal newborn encounters and the case assignment should instead be to MS-DRG 795.
We stated in the proposed rule that our analysis of this grouping issue confirmed that when a principal diagnosis code from MDC 15, such as a diagnosis code from category Z38 (Liveborn infants according to place of birth and type of delivery), is reported followed by ICD-10-CM code P05.19 (Newborn small for gestational age, other), Q38.1 (Ankyloglossia) or Q82.5 (Congenital non-neoplastic nevus), the case is assigned to MS-DRG 794.
However, as we examined the GROUPER logic that would determine an assignment of cases to MS-DRG 795, we noted in the proposed rule that the “only secondary diagnosis” list under MS-DRG 795 already includes ICD-10-CM codes P59.9 (Neonatal jaundice, unspecified) and Z23 (Encounter for immunization). Therefore, when a principal diagnosis code from MDC 15, such as a diagnosis code from category ( print page 69062) Z38 (Liveborn infants according to place of birth and type of delivery) is reported, followed by ICD-10-CM code P59.9 or Z23, the case is currently assigned to MS-DRG 795, not MS-DRG 794, as suggested by the requestor. We refer the reader to the ICD-10 MS-DRG Version 41.1 Definitions Manual (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRGs 794 and 795.
Next, we stated in the proposed rule that we reviewed the claims data from the September 2023 update of the FY 2023 MedPAR file; however, we found zero cases across MS-DRGs 794 and 795. We then examined the clinical factors. The description for ICD-10-CM diagnosis code P05.19 is “Newborn small for gestational age, other” and the inclusion term in the ICD-10-CM Tabular List of Diseases for this diagnosis code is “Newborn small for gestational age, 2,500 grams and over.” We noted in the proposed rule that “small-for-gestational age” is diagnosed by assessing the gestational age and the weight of the baby after birth. There is no specific treatment for small-for-gestational-age newborns. Most newborns who are moderately small for gestational age are healthy babies who just happen to be on the smaller side. Unless the newborn is born with an infection or has a genetic disorder, most small-for-gestational-age newborns have no symptoms and catch up in their growth during the first year of life and have a normal adult height. Next, ICD-10-CM diagnosis code Q38.1 describes ankyloglossia, also known as tongue-tie, which is a condition that impairs tongue movement due to a restrictive lingual frenulum. We noted that in infants, tongue-tie is treated by making a small cut to the lingual frenulum to allow the tongue to move more freely. This procedure, called a frenotomy, can be done in a healthcare provider's office without anesthesia. Newborns generally recover within about a minute of the procedure, and pain relief is usually not indicated. Lastly, ICD-10-CM diagnosis code Q82.5 describes a congenital non-neoplastic nevus. A congenital nevus is a type of pigmented birthmark that appears at birth or during a baby's first year. Most congenital nevi do not cause health problems and may only require future monitoring.
In reviewing these three ICD-10-CM codes and the conditions they describe; we stated in the proposed rule that we believe these diagnoses generally do not prolong the inpatient admission of the newborn and newborns with these diagnoses generally receive standard follow-up care after birth. We stated clinically, we agreed with the requestor that newborn encounters coded with a principal diagnosis code from ICD-10-CM category Z38 (Liveborn infants according to place of birth and type of delivery), followed by code P05.19 (Newborn small for gestational age, other), Q38.1 (Ankyloglossia), or Q82.5 (Congenital non-neoplastic nevus) should not map to MS-DRG 794 (Neonate with Other Significant Problems) and should instead be assigned to MS-DRG 795 (Normal Newborn). Therefore, for the reasons discussed, we proposed to reassign diagnosis code P05.19 from the “principal or secondary diagnosis” list under MS-DRG 794 to the “principal diagnosis” list under MS-DRG 795 (Normal Newborn). We also proposed to add diagnosis codes Q38.1 and Q82.5 to the “only secondary diagnosis” list under MS-DRG 795 (Normal Newborn). Under this proposal, cases with a principal diagnosis described by an ICD-10-CM code from category Z38 (Liveborn infants according to place of birth and type of delivery), followed by codes P05.19, Q38.1, or Q82.5 will be assigned to MS-DRG 795.
In response to the recommendation that CMS update the GROUPER logic that would determine an assignment of cases to MS-DRGs in MDC 15, in the proposed rule we stated we agreed with the requestor that the GROUPER logic for MS-DRG 794 is defined by a “principal or secondary diagnosis of newborn or neonate, with other significant problems, not assigned to DRG 789 through 793 or 795”. We acknowledged that MS-DRG 794 utilizes “fall-through” logic, meaning if a diagnosis code is not assigned to any of the other MS-DRGs, then assignment “falls-through” to MS-DRG 794. As discussed in the proposed rule, we have started to examine the GROUPER logic that would determine the assignment of cases to the MS-DRGs in MDC 15, including MS-DRGs 794 and 795, to determine where further refinements could potentially be made to better account for differences in clinical complexity and resource utilization. However, as we have noted in prior rulemaking ( 72 FR 47152 ), we cannot adopt the same approach to refine the newborn MS-DRGs because of the extremely low volume of Medicare patients there are in these MS-DRGs. Additional time is needed to fully and accurately evaluate cases currently grouping to the MS-DRGs in MDC 15 to consider if restructuring the current MS-DRGs would better recognize the clinical distinctions of these patient populations. Any proposed modifications to these MS-DRGs will be addressed in future rulemaking consistent with our annual process.
Comment: Many commenters expressed support for the proposal to reassign diagnosis code P05.19 from the “principal or secondary diagnosis” list under MS-DRG 794 to the “principal diagnosis” list under MS-DRG 795 (Normal Newborn) and the proposal to add diagnosis codes Q38.1 and Q82.5 to the “only secondary diagnosis” list under MS-DRG 795 (Normal Newborn) for FY 2025. Several commenters stated these updates are needed, are very timely, and will better align cases to the appropriate level of care. Other commenters stated they were committed to helping update the GROUPER logic for MS-DRG 794 and expressed their willingness to work with CMS. A commenter specifically stated they applaud CMS' initiation of an examination of the GROUPER logic that would determine the assignment of cases to the MS-DRGs in MDC 15 to determine where further refinements could potentially be made to better account for differences in clinical complexity and resource utilization.
While indicating their support for the proposal, some commenters provided the following list of diagnoses which they stated also clinically describe normal newborn encounters when reported and therefore case assignment should also be to MS-DRG 795 instead of MS-DRG 794.

Response: We thank the commenters for their support for the proposal as well as for broader efforts to evaluate the assignment of cases to the MS-DRGs in MDC 15. In response to the list of diagnoses which commenters stated also clinically describe normal newborn encounters when reported and therefore assignment should be to MS-DRG 795, we note that the “principal diagnosis” list under MS-DRG 795 already includes ICD-10-CM codes P08.1 (Other heavy for gestational age newborn) and P08.21 (Post-term newborn). Additionally, the “only secondary diagnosis” list under MS-DRG 795 already includes ICD-10-CM codes Q82.8 (Other specified congenital malformations of skin), Z05.1 (Observation and evaluation of newborn for suspected infectious condition ruled out), Z05.42 (Observation and evaluation of newborn for suspected metabolic condition ruled out), and Z28.82 (Immunization not carried out because of caregiver refusal). Therefore, when principal diagnosis code P08.1 or P08.21 is reported, the case is currently assigned to MS-DRG 795, not MS-DRG 794. Similarly, when a principal diagnosis code from MDC 15, such as a diagnosis code from category Z38 (Liveborn infants according to place of birth and type of delivery) is reported, followed by ICD-10-CM code Q82.8, Z05.1, Z05.42, or Z28.82, the case is currently assigned to MS-DRG 795, not MS-DRG 794, as suggested by the commenters. We refer the reader to the ICD-10 MS-DRG Version 42 Definitions Manual (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRGs 794 and 795.
We will review the remaining diagnoses suggested by the commenters as we examine the GROUPER logic that would determine the assignment of cases to the MS-DRGs in MDC 15, including MS-DRGs 794 and 795. We note that we would address any proposed modifications to the existing logic in future rulemaking.
Comment: Other commenters disagreed with the proposal. A commenter noted that patients with ankyloglossia can struggle to breastfeed, are at risk of an early transition to formula, can be small for gestational age, and are at risk for malnutrition. Another commenter noted that contrary to statements in the proposed rule, frenotomy or frenectomy procedures are not as simple as once originally thought and can involve rare complications such as bleeding, airway obstruction, damage to surrounding structures, scarring, and oral aversion secondary to damage to the tongue, nerves, or salivary glands and further noted that when undergoing frenotomy without analgesia, researchers found that 18% of infants cried during and 60% cried after the procedure. This commenter stated that their analysis of claims from their facility indicated that of the approximately 1000 cases reporting a secondary diagnosis of ankyloglossia, frenectomy was performed in approximately 60 cases due to issues with breast feeding and 15% of those cases had a length of stay greater than or equal to 4 days.
A commenter disagreed with the proposal to remove ICD-10-CM diagnosis code P05.19 (newborn small for gestation age, other) from the logic for MS-DRG 794 and stated that newborns that are small for gestational age must undergo hypoglycemia screening, which includes the monitoring of glucose levels at 1, 2, 3, 12, and 24 hours of life and are at increased risk for complications such as neonatal asphyxia, hypothermia, hypoglycemia, hypocalcemia, polycythemia, sepsis, and death. This commenter stated review of the neonatal admissions at their facility supports that these neonates often require longer lengths of stay and utilize increased resources as 5% of approximately 800 cases reporting a secondary diagnosis of P05.19 had a length of stay greater or equal to 4 days. This commenter stated that should diagnosis codes P05.19 and Q38.1 be removed from the logic of MS-DRG 794, a new MS-DRG should be created to capture newborns with minor problems.
Response: We appreciate the commenters' feedback. We considered concerns expressed by the commenters and continue to believe that diagnoses P05.19 and Q38.1 generally do not prolong the inpatient admission of the newborn and newborns with these diagnoses generally receive standard follow-up care after birth. As discussed in the proposed rule, the description for ICD-10-CM diagnosis code P05.19 is “Newborn small for gestational age, other” and the inclusion term in the ICD-10-CM Tabular List of Diseases for this diagnosis code is “Newborn small for gestational age, 2,500 grams and over.” We continue to believe that most newborns who are moderately small for gestational age are healthy babies who just happen to be on the smaller side. We further note that under the proposal to reassign diagnosis code P05.19 from the “principal or secondary diagnosis” list under MS-DRG 794 to the ( print page 69064) “principal diagnosis” list under MS-DRG 795, cases reporting other codes from ICD-10-CM subcategory P05.1- (Newborns small for gestation age) describing newborns small for gestation age, 1999 grams or less, will continue to be assigned to MS-DRG 793 (Full Term Neonate with Major Problems). While we agree that newborns can require serial glucose monitoring after birth, blood glucose can be checked with just a few drops of blood, usually taken from the heel of the newborn and does not involve an invasive procedure.
Similarly, in infants with ankyloglossia indicated for frenotomy, the frenotomy is generally a quick, non-invasive procedure that can be done in a healthcare provider's office without anesthesia. Should the uncommon postprocedural complications noted by the commenter arise when frenotomy is performed in the inpatient setting, those complications should be reported to fully reflect the severity of illness, treatment difficulty, complexity of service and the resources utilized in the diagnosis and/or treatment of the complication. We also note, as discussed in prior rulemaking ( 86 FR 44878 ), the MS-DRG system is a system of averages and it is expected that within the diagnostic related groups, some cases may demonstrate higher than average costs, while other cases may demonstrate lower than average costs. We also provide outlier payments to mitigate extreme loss on individual cases.
We will review the suggestion to create an MS-DRG for newborns with minor problems as we examine the GROUPER logic that would determine the assignment of cases to the MS-DRGs in MDC 15 and would address any proposed modifications to the existing logic in future rulemaking.
Therefore, after consideration of the public comments we received, and for the reasons discussed, we are finalizing our proposal to reassign diagnosis code P05.19 from the “principal or secondary diagnosis” list under MS-DRG 794 to the “principal diagnosis” list under MS-DRG 795 (Normal Newborn), without modification, effective October 1, 2024, for FY 2025. We are also finalizing our proposal to add diagnosis codes Q38.1 and Q82.5 to the “only secondary diagnosis” list under MS-DRG 795 (Normal Newborn), without modification, effective October 1, 2024, for FY 2025. Under these finalizations, cases with a principal diagnosis described by an ICD-10-CM code from category Z38 (Liveborn infants according to place of birth and type of delivery), followed by codes P05.19, Q38.1, or Q82.5 will be assigned to MS-DRG 795.
As noted earlier and discussed in the proposed rule, we have started our examination of the GROUPER logic that would determine an assignment of cases to MS-DRGs in MDC 15. During this review, we stated in the proposed rule we noted the logic for MS-DRG 795 (Normal Newborn) includes five diagnosis codes from ICD-10-CM category Q81 (Epidermolysis bullosa). We refer the reader to the ICD-10 MS-DRG Version 41.1 Definitions Manual (available via on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRG 795. The five diagnosis codes and their current MDC and MS-DRG assignments are listed in the following table.

In the proposed rule we stated we reviewed this grouping issue and noted that epidermolysis bullosa (EB) is a group of genetic (inherited) disorders that causes skin to be fragile, blister, and tear easily in response to minimal friction or trauma. In some cases, blisters form inside the body in places such as the mouth, esophagus, other internal organs, or eyes. When the blisters heal, they can cause painful scarring. In severe cases, the blisters and scars can harm internal organs and tissue enough to be fatal. Patients diagnosed with severe cases of EB have a life expectancy that ranges from infancy to 30 years of age.
We noted in the proposed rule that EB has four primary types: simplex, junctional, dystrophic, and Kindler syndrome, and within each type there are various subtypes, ranging from mild to severe. A skin biopsy can confirm a diagnosis of EB and identify which layers of the skin are affected and determine the type of epidermolysis bullosa. Genetic testing may also be ordered to diagnose the specific type and subtype of the disease. In caring for patients with EB, adaptions may be necessary in the form of handling, feeding, dressing, managing pain, and treating wounds caused by the blisters and tears. If there is a known diagnosis of EB, but the neonate has no physical signs at birth, there will still need to be specialty consultation in the inpatient setting or referral for outpatient follow-up. We stated we believe the five diagnosis codes from ICD-10-CM category Q81 (Epidermolysis bullosa) describe conditions that require advanced care and resources similar to other conditions already assigned to the logic of MS-DRG 794 and MS-DRGs 595 and 596 (Major Skin Disorders with MCC and without MCC, respectively), even in cases where the type of EB is unspecified.
Therefore, for clinical consistency, we proposed to reassign ICD-10-CM diagnosis codes Q81.0, Q81.1, Q81.2, Q81.8, and Q81.9 from MS-DRGs 606 and 607 in MDC 09 (Diseases and Disorders of the Skin, Subcutaneous Tissue and Breast) and MS-DRG 795 (Normal Newborn) in MDC 15 to MS-DRGs 595 and 596 in MDC 09 and MS-DRG 794 in MDC 15, effective October 1, 2024, for FY 2025.
Comment: Commenters expressed support for the proposal to reassign ICD-10-CM diagnosis codes Q81.0, Q81.1, Q81.2, Q81.8, and Q81.9 from MS-DRGs 606 and 607 in MDC 09 (Diseases and Disorders of the Skin, Subcutaneous Tissue and Breast) and MS-DRG 795 (Normal Newborn) in MDC 15 to MS-DRGs 595 and 596 in MDC 09 and MS-DRG 794 in MDC 15 for FY 2025.
Response: We appreciate the commenters support.
After consideration of the public comments we received, we are finalizing our proposal to reassign ICD-10-CM diagnosis codes Q81.0, Q81.1, Q81.2, Q81.8, and Q81.9 from MS-DRGs 606 and 607 in MDC 09 (Diseases and Disorders of the Skin, Subcutaneous Tissue and Breast) and MS-DRG 795 ( print page 69065) (Normal Newborn) in MDC 15 to MS-DRGs 595 and 596 (Major Skin Disorders with MCC and without MCC, respectively) in MDC 09 and MS-DRG 794 (Neonate with Other Significant Problems) in MDC 15, without modification, effective October 1, 2024, for FY 2025.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35986 through 35991 ), we identified a replication issue from the ICD-9 based MS-DRGs to the ICD-10 based MS-DRGs regarding the assignment of six ICD-10-CM diagnosis codes that describe a type of acute leukemia. We noted that under the Version 32 ICD-9-CM based MS-DRGs, the ICD-9-CM diagnosis codes as shown in the following table were assigned to surgical MS-DRGs 820, 821, and 822 (Lymphoma and Leukemia with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively), surgical MS-DRGs 823, 824, and 825 (Lymphoma and Non-Acute Leukemia with Other Procedures with MCC, with CC, and without CC/MCC, respectively), and medical MS-DRGs 840, 841, and 842 (Lymphoma and Non-Acute Leukemia with MCC, with CC, and without CC/MCC, respectively) in MDC 17 (Myeloproliferative Diseases and Disorders, Poorly Differentiated Neoplasms). The six ICD-10-PCS code translations also shown in the following table, that provide more detailed and specific information for the ICD-9-CM codes reflected, also currently group to MS-DRGs 820, 821, 822, 823, 824, 825, 840, 841 and 842 in the ICD-10 MS-DRGs Version 41.1. We refer the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1 (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for complete documentation of the GROUPER logic for MS-DRGs 820, 821, 822, 823, 824, 825, 840, 841, and 842.

In the proposed rule we stated that during our review of this issue, we noted that under ICD-9-CM, the diagnosis codes as reflected in the table did not describe the acuity of the diagnosis (for example, acute versus chronic). This is in contrast to their six comparable ICD-10-CM code translations listed in the previous table that provide more detailed and specific information for the ICD-9-CM diagnosis codes and do specify the acuity of the diagnoses.
We noted in the proposed rule that ICD-10-CM codes C94.20, C94.21, and C94.22 describe acute megakaryoblastic leukemia (AMKL), a rare subtype of acute myeloid leukemia (AML) that affects megakaryocytes, platelet-producing cells that reside in the bone marrow. Similarly, ICD-10-CM codes C94.40, C94.41, and C94.42 describe acute panmyelosis with myelofibrosis (APMF), a rare form of acute myeloid leukemia characterized by acute panmyeloid proliferation with increased blasts and accompanying fibrosis of the bone marrow that does not meet the criteria for AML with myelodysplasia related changes. As previously mentioned, these six diagnosis codes are assigned to MS-DRGs 820, 821, 822, 823, 824, 825, 840, 841, and 842. In the proposed rule, we noted that GROUPER logic lists for MS-DRGs 820, 821, and 822 includes diagnosis codes describing lymphoma and both acute and non-acute leukemias, however the logic lists for MS-DRGs 823, 824, 825, 840, 841, and 842 contain diagnosis codes describing lymphoma and non-acute leukemias. We stated that in our analysis of this grouping issue, we also noted that cases reporting a chemotherapy principal diagnosis with a secondary diagnosis describing acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis are assigned to MS-DRGs 846, 847, and 848 (Chemotherapy without Acute Leukemia as Secondary Diagnosis, with MCC, with CC, and without CC/MCC, respectively) in Version 41.1.
Next, in the proposed rule we stated we examined claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRG 823, 824, 825, 840, 841, and 842 to identify cases reporting one of the six diagnosis codes listed previously that describe acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis. We also examined MS-DRGs 846, 847, and 848 (Chemotherapy without Acute Leukemia as Secondary Diagnosis, with MCC, with CC, and without CC/MCC, respectively). Our findings are shown in the following tables:

As shown in the table, in MS-DRG 823, we identified a total of 2,235 cases with an average length of stay of 14 days and average costs of $40,587. Of those 2,235 cases, there were two cases reporting a diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis, with average costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 823 ($49,600 compared to $40,587) and a longer average length of stay (31.5 days compared to 14 days). We found zero cases in MS-DRG 824 reporting a diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis. In MS-DRG 825, we identified a total of 427 cases with an average length of stay of 2.9 days and average costs of $10,959. Of those 427 cases, there was one case reporting a diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis, with costs higher than the average costs in the FY 2023 MedPAR file for MS-DRG 825 ($17,293 compared to $10,959) and a longer length of stay (6 days compared to 2.9 days).

As shown in the table, in MS-DRG 840, we identified a total of 7,747 cases with an average length of stay of 9.6 days and average costs of $26,215. Of those 7,747 cases, there were 12 cases reporting a diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis, with average costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 840 ($21,357 compared to $26,215) and a shorter average length of stay (8.7 days compared to 9.6 days). In MS-DRG 841, we identified a total of 5,019 cases with an average length of stay of 5.3 days and average costs of $13,502. Of those 5,019 cases, there were six cases reporting a diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis, with average costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 841 ($6,976 compared to $13,502) and a shorter average length of stay (2.8 days compared to 5.3 days). We found zero cases in MS-DRG 842 reporting a diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis.

As shown in the table, in MS-DRG 847, we identified a total of 7,329 cases with an average length of stay of 4.4 days and average costs of $11,250. Of those 7,329 cases, there were two cases reporting a chemotherapy principal diagnosis code with a secondary diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis, with average costs lower than the average costs in the FY 2023 MedPAR file for MS-DRG 840 ($7,569 compared to $11,250) and a longer average length of stay (5 days compared to 4.4 days). We found zero cases in MS-DRGs 846 and 848 reporting a diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis.
As discussed in the proposed rule, next, we examined the MS-DRGs within MDC 17. Given that the six diagnoses codes describe subtypes of acute myeloid leukemia, we stated that we determined that the cases reporting a principal diagnosis of acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis would more suitably group to medical MS-DRGs 834, 835, and 836 (Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively). Similarly, we stated cases reporting a chemotherapy principal diagnosis with a secondary diagnosis describing acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis would more suitably group to medical MS-DRGs 837, 838, and 839 (Chemotherapy with Acute Leukemia as Secondary Diagnosis, or with High Dose Chemotherapy Agent with MCC, with CC or High Dose Chemotherapy Agent, and without CC/MCC, respectively).
We stated we then examined claims data from the September 2023 update of the FY 2023 MedPAR for MS-DRGs 834, 835, 836, 837, 838, and 839. Our findings are shown in the following table.

While the average costs for all cases in MS-DRGs 834, 835, 836, 837, 838, and 839 are higher than the average costs of the small number of cases reporting a diagnosis code that describes acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis, or reporting a chemotherapy principal diagnosis with a secondary diagnosis describing acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis, and the average lengths of stay are longer, we noted that diagnosis codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 describe types of acute leukemia. In the proposed rule we stated that for clinical coherence, we believe these six diagnosis codes would be more appropriately grouped along with other ICD-10-CM diagnosis codes that describe types of acute leukemia.
We reviewed this grouping issue, and stated our analysis indicates that the six diagnosis codes describing the acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis were initially assigned to the list of diagnoses in the GROUPER logic for MS-DRGs 823, 824, 825, 840, 841, and 842 as a result of replication in the transition from ICD-9 to ICD-10 based MS-DRGs. We also noted that diagnosis codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 do not describe non-acute leukemia diagnoses.
Accordingly, because the six diagnosis codes that describe acute megakaryoblastic leukemia or acute panmyelosis with myelofibrosis are not clinically consistent with non-acute leukemia diagnoses, and it is clinically ( print page 69068) appropriate to reassign these diagnosis codes to be consistent with the other diagnosis codes that describe acute leukemias in MS-DRGs 834, 835, 836, 837, 838, and 839, we proposed the reassignment of diagnosis codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 from MS-DRGs 823, 824, and 825 (Lymphoma and Non-Acute Leukemia with Other Procedures with MCC, with CC, and without CC/MCC, respectively), and MS-DRGs 840, 841, and 842 (Lymphoma and Non-Acute Leukemia with MCC, with CC, and without CC/MCC, respectively) to MS-DRGs 834, 835, and 836 (Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) and MS-DRGs 837, 838, and 839 (Chemotherapy with Acute Leukemia as Secondary Diagnosis, or with High Dose Chemotherapy Agent with MCC, with CC or High Dose Chemotherapy Agent, and without CC/MCC, respectively) in MDC 17, effective FY 2025. Under this proposal, diagnosis codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 will continue to be assigned to surgical MS-DRGs 820, 821, and 822 (Lymphoma and Leukemia with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively).
Comment: Commenters supported our proposal to reassign diagnosis codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 from MS-DRGs 823, 824, and 825 and MS-DRGs 840, 841, and 842 to MS-DRGs 834, 835, and 836 and MS-DRGs 837, 838, and 839 in MDC 17.
After consideration of the public comments we received, we are finalizing our proposal to reassign diagnosis codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 from MS-DRGs 823, 824, and 825 (Lymphoma and Non-Acute Leukemia with Other Procedures with MCC, with CC, and without CC/MCC, respectively), and MS-DRGs 840, 841, and 842 (Lymphoma and Non-Acute Leukemia with MCC, with CC, and without CC/MCC, respectively) to MS-DRGs 834, 835, and 836 (Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) and MS-DRGs 837, 838, and 839 (Chemotherapy with Acute Leukemia as Secondary Diagnosis, or with High Dose Chemotherapy Agent with MCC, with CC or High Dose Chemotherapy Agent, and without CC/MCC, respectively) in MDC 17, without modification, effective October 1, 2024, for FY 2025. Under this finalization, diagnosis codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 will continue to be assigned to surgical MS-DRGs 820, 821, and 822 (Lymphoma and Leukemia with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively).
As discussed in the proposed rule, in our review of the MS-DRGs in MDC 17 for further refinement, we next examined the procedures currently assigned to MS-DRGs 820, 821, and 822 (Lymphoma and Leukemia with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) and MS-DRGs 826, 827, and 828 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively). We noted that the logic for case assignment to MS-DRGs 820, 821, 822, 826, 827, and 828 is comprised of a logic list entitled “Operating Room Procedures” which is defined by a list of 4,320 ICD-10-PCS procedure codes, including 90 ICD-10-PCS codes describing bypass procedures from the cerebral ventricle to various body parts. We refer the reader to the ICD-10 MS-DRG Definitions Manual Version 41.1 (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps ) for complete documentation of the GROUPER logic for MS-DRGs 820, 821, 822, 826, 827, and 828.
In the proposed rule we stated in our review of the procedures currently assigned to MS-DRGs 820, 821, 822, 826, 827, and 828, we noted 12 ICD-10-PCS procedure codes that describe bypass procedures from the cerebral ventricle to the subgaleal space or cerebral cisterns, such as subgaleal or cisternal shunt placement, that are not included in the logic for MS-DRGs 820, 821, 822, 826, 827, and 828. The 12 procedure codes are listed in the following table.

We noted in the proposed rule that a subgaleal shunt consists of a shunt tube with one end in the lateral ventricles while the other end is inserted into the subgaleal space of the scalp, while a ventriculo-cisternal shunt diverts the cerebrospinal fluid flow from one of the lateral ventricles, via a ventricular catheter, to the cisterna magna of the posterior fossa. Both procedures allow for the drainage of excess cerebrospinal fluid. Indications for ventriculosubgaleal or ventriculo-cisternal shunting include acute head trauma, subdural hematoma, hydrocephalus, and leptomeningeal disease (LMD) in malignancies such as breast cancer, lung cancer, melanoma, acute lymphocytic leukemia (ALL) and non-hodgkin's lymphoma (NHL).
Recognizing that acute lymphocytic leukemia (ALL) and non-hodgkin's lymphoma (NHL) are indications for ventriculosubgaleal or ventriculo- ( print page 69069) cisternal shunting, in the proposed rule we stated we supported adding the 12 ICD-10-PCS codes identified in the table to MS-DRGs 820, 821, 822, 826, 827, and 828 in MDC 17 for consistency to align with the procedure codes listed in the definition of MS-DRGs 820, 821, 822, 826, 827, and 828 and also to permit proper case assignment when a principal diagnosis from MDC 17 is reported with one of the procedure codes in the table that describes bypass procedures from the cerebral ventricle to the subgaleal space or cerebral cisterns. Therefore, we proposed to add the 12 procedure codes that describe bypass procedures from the cerebral ventricle to the subgaleal space or cerebral cisterns listed previously to MS-DRGs 820, 821, 822, 826, 827, and 828 in MDC 17 for FY 2025.
Comment: Commenters agreed with the proposal to add the 12 procedure codes that describe bypass procedures from the cerebral ventricle to the subgaleal space or cerebral cisterns to MS-DRGs 820, 821, 822, 826, 827, and 828 in MDC 17.
After consideration of the public comments we received, we are finalizing our proposal to add the 12 procedure codes that describe bypass procedures from the cerebral ventricle to the subgaleal space or cerebral cisterns listed previously to MS-DRGs 820, 821, 822, 826, 827, and 828 in MDC 17, without modification, effective October 1, 2024, for FY 2025.
Lastly, as discussed in the proposed rule, in our analysis of the MS-DRGs in MDC 17 for further refinement, we noted that the logic for case assignment to medical MS-DRGs 834, 835, and 836 (Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) as displayed in the ICD-10 MS-DRG Version 41.1 Definitions Manual (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps ) is comprised of a logic list entitled “Principal Diagnosis” and is defined by a list of 27 ICD-10-CM diagnosis codes describing various types of acute leukemias. We noted that when any one of the 27 listed diagnosis codes from the “Principal Diagnosis” logic list is reported as a principal diagnosis, without a procedure code designated as an O.R. procedure or without a procedure code designated as a non-O.R. procedure that affects the MS-DRG, the case results in assignment to MS-DRG 834, 835, or 836 depending on the presence of any additional MCC or CC secondary diagnoses. We noted however, that while not displayed in the ICD-10 MS-DRG Version 41.1 Definitions Manual, when any one of the 27 listed diagnosis codes from the “Principal Diagnosis” logic list is reported as a principal diagnosis, along with a procedure code designated as an O.R. procedure that is not listed in the logic list of MS-DRGs 820, 821, and 822 (Lymphoma and Leukemia with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively), the case also results in assignment to medical MS-DRG 834, 835, or 836 depending on the presence of any additional MCC or CC secondary diagnoses.
As medical MS-DRG 834, 835, and 836 contains GROUPER logic that includes ICD-10-PCS procedure codes designated as O.R. procedures, in the proposed rule we stated we examined claims data from the September 2023 update of the FY 2023 MedPAR file for MS-DRG 834, 835, and 836 to identify cases reporting an O.R. procedure. Our findings are shown in the following table:

As shown by the table, in MS-DRG 834, we identified a total of 4,094 cases, with an average length of stay of 16.3 days and average costs of $49,986. Of those 4,094 cases, there were 277 cases reporting an O.R. procedure, with higher average costs as compared to all cases in MS-DRG 834 ($92,246 compared to $49,986), and a longer average length of stay (28.2 days compared to 16.3 days). In MS-DRG 835, we identified a total of 1,682 cases with an average length of stay of 7.2 days and average costs of $19,023. Of those 1,682 cases, there were 79 cases reporting an O.R. procedure, with higher average costs as compared to all cases in MS-DRG 835 ($30,771 compared to $19,023), and a longer average length of stay (10.4 days compared to 7.2 days). In MS-DRG 836, we identified a total of 230 cases with an average length of stay of 4 days and average costs of $11,225. Of those 230 cases, there were 7 cases reporting an O.R. procedure, with higher average costs as compared to all cases in MS-DRG 836 ($17,950 compared to $11,225), and a longer average length of stay (5.9 days compared to 4 days). We stated that the data analysis shows that the average costs of cases reporting an O.R. procedure are higher than for all cases in their respective MS-DRG.
We stated in the proposed rule that the data analysis clearly shows that cases reporting a principal diagnosis code describing a type of acute leukemia with an ICD-10-PCS procedure code designated as O.R. procedure that is not listed in the logic list of MS-DRGs 820, 821, and 822 have higher average costs and longer lengths of stay compared to all the cases in their assigned MS-DRG. For these reasons, we proposed to create a new surgical MS-DRG for cases reporting a principal diagnosis code describing a type of acute leukemia with an O.R. procedure.
To compare and analyze the impact of our suggested modifications, as discussed in the proposed rule, we ran a simulation using the claims data from the September 2023 update of the FY 2023 MedPAR file. The following table illustrates our findings for all 367 cases reporting a principal diagnosis code describing a type of acute leukemia with an ICD-10-PCS procedure code designated as O.R. procedure that is not listed in the logic list of MS-DRGs 820, 821, and 822. We stated we believe the resulting proposed MS-DRG assignment, reflecting these modifications, is more clinically ( print page 69070) homogeneous, coherent, and better reflects hospital resource use.

In the proposed rule, we stated we applied the criteria to create subgroups in a base MS-DRG as discussed in section II.C.1.b. of this FY 2025 IPPS/LTCH PPS proposed rule. As shown in the table, we identified a total of 367 cases using the claims data from the September 2023 update of the FY 2023 MedPAR file, so the criterion that there are at least 500 or more cases in each subgroup could not be met. Therefore, for FY 2025, we did not propose to subdivide the proposed new MS DRG for acute leukemia with other procedures into severity levels.
In summary, for FY 2025, we proposed to create a new base surgical MS-DRG for cases reporting a principal diagnosis describing a type of acute leukemia with an ICD-10-PCS procedure code designated as an O.R. procedure that is not listed in the logic list of MS-DRGs 820, 821, and 822 in MDC 17. The proposed new MS-DRG is proposed new MS-DRG 850 (Acute Leukemia with Other Procedures). We proposed to add the 27 ICD-10-CM diagnosis codes describing various types of acute leukemias currently listed in the logic list entitled “Principal Diagnosis” in MS-DRGs 834, 835, and 836 as well as ICD-10-CM codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 discussed earlier in this section to the proposed new MS-DRG 850. We also proposed to add the procedure codes from current MS-DRGs 823, 824, and 825 (Lymphoma and Non-Acute Leukemia with Other Procedures with MCC, with CC, and without CC/MCC, respectively) to the proposed new MS-DRG 850. In the proposed rule, we noted that in the current logic list of MS-DRGs 823, 824, and 825 there are 189 procedure codes describing stereotactic radiosurgery of various body parts that are designated as non-O.R. procedures affecting the MS-DRG, therefore, as part of the logic for new MS-DRG 850, we also proposed to designate these 189 codes as non-O.R. procedures affecting the MS-DRG.
In addition, we proposed to revise the titles for MS-DRGs 834, 835, and 836 by deleting the reference to “Major O.R. Procedures” in the title. Specifically, we proposed to revise the titles of medical MS-DRGs 834, 835, and 836 from “Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC”, respectively to “Acute Leukemia with MCC, with CC, and without CC/MCC”, respectively to better reflect the GROUPER logic that will no longer include ICD-10-PCS procedure codes designated as O.R. procedures. We refer the reader to section II.C.15. of the preamble of this final rule for the discussion of the surgical hierarchy and the complete list of our proposed modifications to the surgical hierarchy as well as our finalization of those proposals.
Comment: Commenters supported the proposal to create new surgical MS-DRG 850 for cases reporting a principal diagnosis code describing a type of acute leukemia with an O.R. procedure in MDC 17. Commenters also supported the proposal to revise the titles of medical MS-DRGs 834, 835, and 836 from “Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC”, respectively to “Acute Leukemia with MCC, with CC, and without CC/MCC”. Several commenters stated they appreciate CMS' continued analysis and refinement in this MDC and the recognition of the increased resource intensity involved in acute leukemia cases with certain operating room procedures. Another commenter stated they appreciate the agency's detailed explanation and stated they support the changes as proposed.
Comment: While supporting the creation of a new MS-DRG for cases reporting a principal diagnosis describing a type of acute leukemia with an ICD-10-PCS procedure code designated as O.R. procedure that is not listed in the logic list of MS-DRGs 820, 821, and 822 in MDC 17, a few commenters suggested that CMS reconsider the criteria for determining subgroups with small population MS-DRGs such as proposed new MS-DRG 850. According to these commenters, while the data clearly shows differences in the average costs and average lengths of stay in cases reporting secondary diagnoses designated as MCCs, CCs, and NonCCs, the criterion that there are at least 500 or more cases in each subgroup could not be met as only 367 cases were identified, therefore, CMS did not propose to subdivide the proposed new MS DRG for acute leukemia with other procedures into severity levels.
Response: We thank the commenters for their support and feedback. With regard to the suggestion that CMS reconsider the criteria for determining subgroups with small population MS-DRGs, we note in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58448 ), we finalized our proposal to expand our existing criteria to create a new complication or comorbidity (CC) or major complication or comorbidity (MCC) subgroup within a base MS-DRG. Specifically, we finalized the expansion of the criteria to include the NonCC subgroup for a three-way severity level split. We stated we believed that applying these criteria to the NonCC subgroup would better reflect resource stratification as well as promote stability in the relative weights by avoiding low volume counts for the NonCC level MS-DRGs.
As further discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58659 through 58660 ), the minimum case volume requirements were established to avoid overly fragmenting the MS-DRG classification system. We stated that with smaller volumes, the MS-DRGs will be subject to stochastic (unpredictable) effects. We continue to believe that stability of MS-DRG payment is an important objective and therefore, that a volume criterion is a needed adjunct to cost differentiation. We established a 500-case minimum to support this stability. Additionally, we note that in examining the claims data from the September 2023 update of the FY 2023 MedPAR file to identify cases reporting an O.R. procedure and a principal diagnosis code describing various types of acute leukemias, there were only 7 cases reporting an O.R. procedure with a principal diagnosis code describing various types of acute leukemias, without reporting a secondary diagnosis designated as a CC or an MCC. As stated in the proposed rule ( 89 FR 36021 ), we set a threshold of 10 cases as the minimum number of ( print page 69071) cases required to compute a reasonable weight for an MS-DRG. Fewer than 10 cases does not provide sufficient data to set accurate and stable cost relative weights.
We also note, as discussed in prior rulemaking ( 86 FR 44878 ), the MS-DRG system is a system of averages and it is expected that within the diagnostic related groups, some cases may demonstrate higher than average costs, while other cases may demonstrate lower than average costs. We also provide outlier payments to mitigate extreme loss on individual cases.
We refer the reader to section II.C.1.b. of the preamble of this final rule for related discussion regarding our finalization of the expansion of the criteria to include the NonCC subgroup in the FY 2021 final rule and our finalization of the proposal to continue to delay application of the NonCC subgroup criteria to existing MS-DRGs with a three-way severity level split for FY 2025.
After consideration of the public comments we received, and for the reasons discussed, we are finalizing our proposal to create new base surgical MS-DRG 850 (Acute Leukemia with Other Procedures) for cases reporting a principal diagnosis describing a type of acute leukemia with an ICD-10-PCS procedure code designated as an O.R. procedure that is not listed in the logic list of MS-DRGs 820, 821, and 822 in MDC 17, without modification, effective October 1, 2024, for FY 2025. Accordingly for FY 2025, we are finalizing our proposal to add the 27 ICD-10-CM diagnosis codes describing various types of acute leukemias currently listed in the logic list entitled “Principal Diagnosis” in MS-DRGs 834, 835, and 836 as well as ICD-10-CM codes C94.20, C94.21, C94.22, C94.40, C94.41, and C94.42 discussed earlier in this section to new MS-DRG 850. We are finalizing our proposal to add the procedure codes from current MS-DRGs 823, 824, and 825 (Lymphoma and Non-Acute Leukemia with Other Procedures with MCC, with CC, and without CC/MCC, respectively) to new MS-DRG 850. In addition, we are also finalizing our proposal to designate the 189 codes describing stereotactic radiosurgery of various body parts as non-O.R. procedures affecting the MS-DRG as part of the logic for new MS-DRG 850 for FY 2025.
Lastly, we are finalizing our proposal to revise the titles for medical MS-DRGs 834, 835, and 836 from “Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC”, respectively to “Acute Leukemia with MCC, with CC, and without CC/MCC”, respectively for FY 2025.
We annually conduct a review of procedures producing assignment to MS-DRGs 981 through 983 (Extensive O.R. Procedure Unrelated to Principal Diagnosis with MCC, with CC, and without CC/MCC, respectively) or MS-DRGs 987 through 989 (Non-Extensive O.R. Procedure Unrelated to Principal Diagnosis with MCC, with CC, and without CC/MCC, respectively) on the basis of volume, by procedure, to see if it would be appropriate to move cases reporting these procedure codes out of these MS-DRGs into one of the surgical MS-DRGs for the MDC into which the principal diagnosis falls. The data are arrayed in two ways for comparison purposes. We look at a frequency count of each major operative procedure code. We also compare procedures across MDCs by volume of procedure codes within each MDC. We use this information to determine which procedure codes and diagnosis codes to examine.
We identify those procedures occurring in conjunction with certain principal diagnoses with sufficient frequency to justify adding them to one of the surgical MS-DRGs for the MDC in which the diagnosis falls. We also consider whether it would be more appropriate to move the principal diagnosis codes into the MDC to which the procedure is currently assigned.
Based on the results of our review of the claims data from the September 2023 update of the FY 2023 MedPAR file of cases found to group to MS-DRGs 981 through 983 or MS-DRGs 987 through 989, in the proposed rule ( 89 FR 35991 ) we stated we did not identify any cases for reassignment and did not propose to move any cases from MS-DRGs 981 through 983 or MS-DRGs 987 through 989 into a surgical MS-DRGs for the MDC into which the principal diagnosis or procedure is assigned.
In addition to the internal review of procedures producing assignment to MS-DRGs 981 through 983 or MS-DRGs 987 through 989, we also consider requests that we receive to examine cases found to group to MS-DRGs 981 through 983 or MS-DRGs 987 through 989 to determine if it would be appropriate to add procedure codes to one of the surgical MS-DRGs for the MDC into which the principal diagnosis falls or to move the principal diagnosis to the surgical MS-DRGs to which the procedure codes are assigned. As discussed in the proposed rule, we did not receive any requests suggesting reassignment.
We also review the list of ICD-10-PCS procedures that, when in combination with their principal diagnosis code, result in assignment to MS-DRGs 981 through 983, or 987 through 989, to ascertain whether any of those procedures should be reassigned from one of those two groups of MS-DRGs to the other group of MS-DRGs based on average costs and the length of stay. We look at the data for trends such as shifts in treatment practice or reporting practice that would make the resulting MS-DRG assignment illogical. If we find these shifts, we would propose to move cases to keep the MS-DRGs clinically similar or to provide payment for the cases in a similar manner. Generally, we move only those procedures for which we have an adequate number of discharges to analyze the data.
Additionally, we also consider requests that we receive to examine cases found to group to MS-DRGs 981 through 983 or MS-DRGs 987 through 989 to determine if it would be appropriate for the cases to be reassigned from one of the MS-DRG groups to the other. Based on the results of our review of the claims data from the September 2023 update of the FY 2023 MedPAR file, in the proposed rule we stated we did not identify any cases for reassignment. We also did not receive any requests suggesting reassignment. Therefore, for FY 2025 we did not propose to move any cases reporting procedure codes from MS-DRGs 981 through 983 to MS-DRGs 987 through 989 or vice versa.
Comment: Commenters expressed support for CMS' proposal to not move any cases from MS-DRGs 981 through 983 or MS-DRGs 987 through 989 into a surgical MS-DRGs for the MDC into which the principal diagnosis or procedure is assigned. Commenters also expressed support for CMS' proposal to not move any cases reporting procedure codes from MS-DRGs 981 through 983 to MS-DRGs 987 through 989 or vice versa.
After consideration of the public comments we received, we are finalizing, without modification, our proposal to not move any cases from MS-DRGs 981 through 983 or MS-DRGs 987 through 989 into a surgical MS-DRGs for the MDC into which the principal diagnosis or procedure is assigned. We are also finalizing, without modification, our proposal to not move any cases reporting procedure codes ( print page 69072) from MS-DRGs 981 through 983 to MS-DRGs 987 through 989 or vice versa.
Under the IPPS MS-DRGs (and former CMS MS-DRGs), we have a list of procedure codes that are considered operating room (O.R.) procedures. Historically, we developed this list using physician panels that classified each procedure code based on the procedure and its effect on consumption of hospital resources. For example, generally the presence of a surgical procedure which required the use of the operating room would be expected to have a significant effect on the type of hospital resources (for example, operating room, recovery room, and anesthesia) used by a patient, and therefore, these patients were considered surgical. Because the claims data generally available do not precisely indicate whether a patient was taken to the operating room, surgical patients were identified based on the procedures that were performed.
Generally, if the procedure was not expected to require the use of the operating room, the patient would be considered medical (non-O.R.).
Currently, each ICD-10-PCS procedure code has designations that determine whether and in what way the presence of that procedure on a claim impacts the MS-DRG assignment. First, each ICD-10-PCS procedure code is either designated as an O.R. procedure for purposes of MS-DRG assignment (“O.R. procedures”) or is not designated as an O.R. procedure for purposes of MS-DRG assignment (“non-O.R. procedures”). Second, for each procedure that is designated as an O.R. procedure, that O.R. procedure is further classified as either extensive or non-extensive. Third, for each procedure that is designated as a non-O.R. procedure, that non-O.R. procedure is further classified as either affecting the MS-DRG assignment or not affecting the MS-DRG assignment. We refer to these designations that do affect MS-DRG assignment as “non O.R. affecting the MS-DRG.” For new procedure codes that have been finalized through the ICD-10 Coordination and Maintenance Committee meeting process and are proposed to be classified as O.R. procedures or non-O.R. procedures affecting the MS-DRG, we recommend the MS-DRG assignment which is then made available in association with the proposed rule (Table 6B.—New Procedure Codes) and subject to public comment. These proposed assignments are generally based on the assignment of predecessor codes or the assignment of similar codes. For example, we generally examine the MS-DRG assignment for similar procedures, such as the other approaches for that procedure, to determine the most appropriate MS-DRG assignment for procedures proposed to be newly designated as O.R. procedures. As discussed in section II.C.13 of the preamble of this final rule, we are making Table 6B.—New Procedure Codes—FY 2025 available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps.html . We also refer readers to the ICD-10 MS-DRG Version 41.1 Definitions Manual at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software.html for detailed information regarding the designation of procedures as O.R. or non-O.R. (affecting the MS- DRG) in Appendix E—Operating Room Procedures and Procedure Code/MS-DRG Index.
In the FY 2020 IPPS/LTCH PPS proposed rule, we stated that, given the long period of time that has elapsed since the original O.R. (extensive and non-extensive) and non-O.R. designations were established, the incremental changes that have occurred to these O.R. and non-O.R. procedure code lists, and changes in the way inpatient care is delivered, we plan to conduct a comprehensive, systematic review of the ICD-10-PCS procedure codes. This will be a multiyear project during which we will also review the process for determining when a procedure is considered an operating room procedure. For example, we may restructure the current O.R. and non-O.R. designations for procedures by leveraging the detail that is now available in the ICD-10 claims data. We refer readers to the discussion regarding the designation of procedure codes in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38066 ) where we stated that the determination of when a procedure code should be designated as an O.R. procedure has become a much more complex task. This is, in part, due to the number of various approaches available in the ICD-10-PCS classification, as well as changes in medical practice. While we have typically evaluated procedures on the basis of whether or not they would be performed in an operating room, we believe that there may be other factors to consider with regard to resource utilization, particularly with the implementation of ICD-10.
We discussed in the FY 2020 IPPS/LTCH PPS proposed rule that as a result of this planned review and potential restructuring, procedures that are currently designated as O.R. procedures may no longer warrant that designation, and conversely, procedures that are currently designated as non-O.R. procedures may warrant an O.R. type of designation. We intend to consider the resources used and how a procedure should affect the MS-DRG assignment. We may also consider the effect of specific surgical approaches to evaluate whether to subdivide specific MS-DRGs based on a specific surgical approach. We stated we plan to utilize our available MedPAR claims data as a basis for this review and the input of our clinical advisors. As part of this comprehensive review of the procedure codes, we also intend to evaluate the MS-DRG assignment of the procedures and the current surgical hierarchy because both of these factor into the process of refining the ICD-10 MS-DRGs to better recognize complexity of service and resource utilization.
In the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58540 through 58541 ), we provided a summary of the comments we had received in response to our request for feedback on what factors or criteria to consider in determining whether a procedure is designated as an O.R. procedure in the ICD-10-PCS classification system for future consideration. We also stated that in consideration of the PHE, we believed it may be appropriate to allow additional time for the claims data to stabilize prior to selecting the timeframe to analyze for this review.
We stated in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58749 ) that we continue to believe additional time is necessary as we continue to develop our process and methodology. Therefore, we stated we will provide more detail on this analysis and the methodology for conducting this review in future rulemaking. In response to this discussion in the FY 2024 IPPS/LTCH PPS final rule, we received a comment by the October 20, 2023 deadline. As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35992 ), the commenter acknowledged that there is no easy rule that would allow CMS to designate certain surgeries as “non-O.R.” procedures. The commenter stated that they believed that open procedures should always be designated O.R. procedures and approaches other than open should not be a sole factor in designating a procedure as non-O.R. as some minimally invasive procedures ( print page 69073) using a percutaneous endoscopic approach require more training, specialized equipment, time, and resources than traditional open procedures. In addition, the commenter stated that whether a procedure is frequently or generally performed in the outpatient setting should not be used for determination of O.R. vs non-O.R. designation and noted that a surgery that can be performed in the outpatient setting for a clinically stable patient may not be able to be safely performed on a patient who is clinically unstable. The commenter also asserted that for procedures that can be performed in various locations within the hospital, that is, bedside vs operating room, there should be a mechanism to differentiate the setting of the procedure to determine the MS-DRG assignment, as in the commenter's assessment, the ICD-10 classification does not provide a way to indicate the severity of certain conditions, or the complexity of procedures performed.
As discussed in the proposed rule, CMS appreciates the commenter's feedback and recommendations as to factors to consider in evaluating O.R. designations. We stated we agree with the commenter and believe that there may be other factors to consider with regard to resource utilization. As discussed in the FY 2024 IPPS/LTCH PPS final rule, we have signaled in prior rulemaking that the designation of an O.R. procedure encompasses more than the physical location of the hospital room in which the procedure may be performed; in other words, the performance of a procedure in an operating room is not the sole determining factor we will consider as we examine the designation of a procedure in the ICD-10-PCS classification system. We are exploring alternatives on how we may restructure the current O.R. and non-O.R. designations for procedures by leveraging the detail that is available in the ICD-10 claims data.
Comment: Many commenters supported CMS' plan to continue to conduct the comprehensive, systematic review of the ICD-10-PCS codes and to evaluate their current O.R. and non-O.R. designations. These commenters expressed that they were supportive of CMS' decision to continue to develop our process and methodology. Other commenters stated they agreed that the revolution in medical procedures in recent years may render the performance of a procedure in an O.R. a less critical distinction in driving payment policy. A commenter stated that because of technological advances, sophisticated, resource-intensive procedures are no longer confined to the O.R. setting and noted that in their observation, bi-plane radiology interventional suites and cardiac catheterization labs used for procedures such as mechanical thrombectomy or endovascular coiling for aneurysms can utilize more advanced equipment and supplies than a basic operating room with minimal installed equipment. Several commenters recommended that CMS provide detailed impact files prior to the adoption of changes to the designation of procedure codes in the ICD-10-PCS classification and stated that they look forward to commenting on CMS' data analysis and methodology in the future.
As part of the broader and continuing conversation about the designations of procedures in the ICD-10-PCS classification system, a few commenters recommended that CMS work closely with physician specialty societies and industry stakeholders to identify the most important drivers of complexity and resource use in the hospital setting. A commenter specifically recommended that CMS consider a technical expert panel (TEP) made up of industry stakeholders and experts to review methodologies for determining the designation of procedure codes in the ICD-10-PCS classification system. Another commenter encouraged CMS to consider factors such as:
- whether the procedure involves either the intentional non-transient alteration of structures of the body, or cutting into the body, or both;
- the surgical approach ( e.g., open, percutaneous endoscopic or percutaneous approach);
- the requirement of either a surgeon or non-surgeon provider to be present during procedure;
- the complexity of procedures performed in an operating room, or a hybrid operating room, versus procedures performed in an electrophysiology laboratory;
- resource utilization requirements during the performance of the procedure in terms of the need for anesthesia and monitoring, etc.; and
- inpatient versus outpatient status.
Response: We thank the commenters for their support. We also thank commenters for sharing their views and their willingness to provide feedback and recommendations as to what factors to consider in evaluating O.R. versus non-O.R. designations. We agree with commenters and believe that there may be other factors to consider with regard to resource utilization, particularly with the implementation of ICD-10. While CMS has already convened an internal workgroup comprised of clinicians, consultants, coding specialists and other policy analysts, as well as provided opportunity to provide feedback as to what factors to consider in evaluating O.R. versus non-O.R. designations, we look forward to further input and feedback from interested parties. As discussed in the proposed rule, we are considering the feedback received to date on what factors and/or criteria to consider in determining whether a procedure is designated as an O.R. procedure in the ICD-10-PCS classification system as we continue to develop our process and methodology and will provide more detail on this analysis and the methodology for conducting this comprehensive review in future rulemaking. We encourage the public to continue to submit comments on any other factors to consider in our refinement efforts to recognize and differentiate consumption of resources for the ICD-10 MS-DRGs for consideration.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule, we did not receive any requests regarding changing the designation of specific ICD-10-PCS procedure codes from non-O.R. to O.R. procedures, or to change the designation from O.R. procedures to non-O.R. procedures by the October 20, 2023 deadline. In this section of this final rule, as we did in the proposed rule, we discuss the proposals we made based on our internal review and analysis and we discuss the process that was utilized for evaluating each procedure code. For each procedure, we considered—
- Whether the procedure would typically require the resources of an operating room;
- Whether it is an extensive or a non-extensive procedure; and
- To which MS-DRGs the procedure should be assigned.
We note that many MS-DRGs require the presence of any O.R. procedure. As a result, cases with a principal diagnosis associated with a particular MS-DRG would, by default, be grouped to that MS-DRG. Therefore, we do not list these MS-DRGs in our discussion in this section of this final rule. Instead, we only discuss MS-DRGs that require explicitly adding the relevant procedure codes to the GROUPER logic in order for those procedure codes to affect the MS-DRG assignment as intended.
For procedures that would not typically require the resources of an operating room, we determined if the procedure should affect the MS-DRG assignment. In cases where we proposed to change the designation of procedure codes from non-O.R. procedures to O.R. procedures, we also proposed one or more MS-DRGs with which these ( print page 69074) procedures are clinically aligned and to which the procedure code would be assigned.
In addition, cases that contain O.R. procedures will map to MS-DRGs 981, 982, or 983 (Extensive O.R. Procedure Unrelated to Principal Diagnosis with MCC, with CC, and without CC/MCC, respectively) or MS-DRGs 987, 988, or 989 (Non-Extensive O.R. Procedure Unrelated to Principal Diagnosis with MCC, with CC, and without CC/MCC, respectively) when they do not contain a principal diagnosis that corresponds to one of the MDCs to which that procedure is assigned. These procedures need not be assigned to MS-DRGs 981 through 989 in order for this to occur. Therefore, we did not specifically address that aspect in summarizing the proposals we made based on our internal review and analysis in the proposed rule and in this section of this final rule.
As discussed in the proposed rule ( 89 FR 35993 ), during our review, we noted inconsistencies in how procedures involving laparoscopic excisions of intestinal body parts are designated. Procedure codes describing the laparoscopic excision of intestinal body parts differ by qualifier. ICD-10-PCS procedure codes describing excisions of intestinal body parts with the diagnostic qualifier “X”, are used to report these procedures when performed for diagnostic purposes. We identified the following five related codes:

In the proposed rule, we noted the ICD-10-PCS procedure codes describing the laparoscopic excision of intestinal body parts for diagnostic purposes listed previously have been assigned different attributes in terms of designation as an O.R. or non-O.R. procedure when compared to similar procedures describing the laparoscopic excisions of intestinal body parts for nondiagnostic purposes. We noted in the ICD-10 MS-DRGs Version 41, these ICD-10-PCS codes are currently recognized as non-O.R. procedures for purposes of MS-DRG assignment, while similar excision of intestinal body part procedure codes with the same approach but different qualifiers are recognized as O.R. procedures.
As discussed in the proposed rule, upon further review and consideration, we stated we believe that procedure codes 0DBF4ZX, 0DBG4ZX, 0DBL4ZX, 0DBM4ZX and 0DBN4ZX describing a laparoscopic excision of an intestinal body parts for diagnostic purposes warrant designation as an O.R. procedures consistent with other laparoscopic excision procedures performed on the same intestinal body parts for nondiagnostic purposes. We stated we also believe it is clinically appropriate for these procedures to group to the same MS-DRGs as the procedures describing excision procedures performed on the intestinal body parts for nondiagnostic purposes. Therefore, we proposed to add procedure codes 0DBF4ZX, 0DBG4ZX, 0DBL4ZX, 0DBM4ZX and 0DBN4ZX to the FY 2025 ICD-10 MS-DRG Version 42 Definitions Manual in Appendix E—Operating Room Procedures and Procedure Code/MS-DRG Index as O.R. procedures assigned to MS-DRG 264 (Other Circulatory System O.R. Procedures) in MDC 05 (Diseases and Disorders of the Circulatory System); MS-DRGs 329, 330, and 331 (Major Small and Large Bowel Procedures, with MCC, with CC, and without CC/MCC, respectively) in MDC 06 (Diseases and Disorders of the Digestive System); MS-DRGs 820, 821, and 822 (Lymphoma and Leukemia with Major O.R. Procedures with MCC, CC, without CC/MCC, respectively) and MS-DRGS 826, 827, and 828 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) in MDC 17 (Myeloproliferative Diseases and Disorders, Poorly Differentiated Neoplasms); MS-DRGs 907, 908, and 909 (Other O.R. Procedures for Injuries with MCC, with CC, and without CC/MCC, respectively) in MDC 21 (Injuries, Poisonings and Toxic Effects of Drugs); and MS-DRGs 957, 958, and 959 (Other O.R. Procedures for Multiple Significant Trauma with MCC, with CC, and without CC/MCC, respectively) in MDC 24 (Multiple Significant Trauma).
Comment: Commenters supported the proposal to reclassify ICD-10-PCS procedure codes 0DBF4ZX (Excision of right large intestine, percutaneous endoscopic approach, diagnostic), 0DBG4ZX (Excision of left large intestine, percutaneous endoscopic approach, diagnostic), 0DBL4ZX (Excision of transverse colon, percutaneous endoscopic approach, diagnostic), 0DBM4ZX (Excision of descending colon, percutaneous endoscopic approach, diagnostic), and 0DBN4ZX (Excision of sigmoid colon, percutaneous endoscopic approach, diagnostic) as O.R. procedures for the purposes of MS-DRG assignment for FY 2025. A commenter stated they believed that laparoscopic procedures—whether diagnostic or nondiagnostic—will always be performed in an O.R. The commenter further urged CMS to publish O.R. versus non-O.R. designation data on its website for all ICD-10-PCS codes, not just new codes, so that specialty societies can more easily review and identify possible errors.
Response: We appreciate the commenters' support and thank the commenter for their feedback. We also appreciate the commenter's suggestion, however, as stated in the earlier in this section, and as we have signaled in prior rulemaking, the designation of an O.R. procedure encompasses more than the physical location of the hospital room in which the procedure may be performed. In other words, the performance of a procedure in an operating room is not the sole determining factor we consider as we examine the designation of a procedure in the ICD-10-PCS classification system. Additionally, we refer the commenter, and interested specialty societies, to Appendix E of the ICD-10 MS-DRG Version 42 Definitions Manual (which is available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Feefor-Service-Payment/AcuteInpatientPPS/MS- ( print page 69075) DRGClassifications-and-Software ) for a list of all the ICD-10-PCS procedure codes that affect MS-DRG assignment (that is, procedure codes designated as O.R. procedures or as non-O.R. procedures affecting the MS-DRG), the MDCs and MS-DRGs to which they are assigned, and a description of the surgical categories.
After consideration of the public comments we received, we are finalizing our proposal to change the designation of procedure codes 0DBF4ZX, 0DBG4ZX, 0DBL4ZX, 0DBM4ZX and 0DBN4ZX from non-O.R. procedures to O.R. procedures, without modification, effective October 1, 2024.
As discussed in the proposed rule ( 89 FR 35994 ), during our review, we noted inconsistencies in how procedures involving laparoscopic excisions of gallbladder or pancreas are designated. Procedure codes describing the laparoscopic excision of the gallbladder or pancreas differ by qualifier. The ICD-10-PCS procedure code describing an excision of the gallbladder and the procedure code describing an excision of the pancreas with the diagnostic qualifier “X”, are used to report these procedures when performed for diagnostic purposes. We stated we identified the following two related codes:

In the proposed rule, we noted the ICD-10-PCS procedure codes describing the laparoscopic excision of the gallbladder or the pancreas for diagnostic purposes listed previously have been assigned different attributes in terms of designation as an O.R. or a non-O.R. procedure when compared to similar procedures describing the laparoscopic excisions of the gallbladder or the pancreas for nondiagnostic purposes. In the ICD-10 MS-DRGs Version 41, these ICD-10-PCS codes are currently recognized as non-O.R. procedures for purposes of MS-DRG assignment, while similar excision of the gallbladder or the pancreas procedure codes with the same approach but different qualifiers are recognized as O.R. procedures.
As discussed in the proposed rule, upon further review and consideration, we stated we believe that procedure code 0FB44ZX describing a laparoscopic excision of the gallbladder for diagnostic purposes and procedure code 0FBG4ZX describing a laparoscopic excision of the pancreas for diagnostic purposes both warrant designation as an O.R. procedure consistent with other laparoscopic excision procedures performed on the same body parts for nondiagnostic purposes. We stated we also believe it is clinically appropriate for these procedures to group to the same MS-DRGs as the procedures describing excision procedures performed on the gallbladder or pancreas for nondiagnostic purposes. Therefore, we proposed to add procedure code 0FB44ZX to the FY 2025 ICD-10 MS-DRG Version 42 Definitions Manual in Appendix E—Operating Room Procedures and Procedure Code/MS-DRG Index as an O.R. procedure assigned to MS-DRGs 411, 412, and 413 (Cholecystectomy with C.D.E., with MCC, with CC, and without CC/MCC, respectively) and MS-DRGs 417, 418, and 419 (Laparoscopic Cholecystectomy without C.D.E., with MCC, with CC, and without CC/MCC, respectively) in MDC 07 (Diseases and Disorders of the Hepatobiliary System and Pancreas); MS-DRGs 820, 821, and 822 (Lymphoma and Leukemia with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) and MS-DRGS 826, 827, and 828 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) in MDC 17 (Myeloproliferative Diseases and Disorders, Poorly Differentiated Neoplasms); MS-DRGs 907, 908, and 909 (Other O.R. Procedures for Injuries with MCC, with CC, and without CC/MCC, respectively) in MDC 21 (Injuries, Poisonings and Toxic Effects of Drugs); and MS-DRGs 957, 958, and 959 (Other O.R. Procedures for Multiple Significant Trauma with MCC, with CC, and without CC/MCC, respectively) in MDC 24 (Multiple Significant Trauma).
We also proposed to add procedure code 0FBG4ZX to the FY 2025 ICD-10 MS-DRG Version 42 Definitions Manual in Appendix E—Operating Room Procedures and Procedure Code/MS-DRG Index as an O.R. procedure assigned to MS-DRGs 405, 406, and 407 (Pancreas, Liver and Shunt Procedures, with MCC, with CC, and without CC/MCC, respectively) in MDC 06 (Diseases and Disorders of the Digestive System); MS-DRGs 628, 629 and 630 (Other Endocrine, Nutritional and Metabolic O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) in MDC 10 (Endocrine, Nutritional and Metabolic Diseases and Disorders); MS-DRGs 907, 908, and 909 (Other O.R. Procedures for Injuries with MCC, with CC, and without CC/MCC, respectively) in MDC 21 (Injuries, Poisonings and Toxic Effects of Drugs); and MS-DRGs 957, 958, and 959 (Other O.R. Procedures for Multiple Significant Trauma with MCC, with CC, and without CC/MCC, respectively) in MDC 24 (Multiple Significant Trauma).
Comment: Commenters supported the proposal to reclassify ICD-10-PCS procedure codes 0FB44ZX (Excision of gallbladder, percutaneous endoscopic approach, diagnostic) and 0FBG4ZX (Excision of pancreas, percutaneous endoscopic approach, diagnostic) as O.R. procedures for the purposes of MS-DRG assignment for FY 2025.
After consideration of the public comments we received, we are finalizing our proposal to change the designation of procedure codes 0FB44ZX and 0FBG4ZX from non-O.R. procedures to O.R. procedures, without modification, effective October 1, 2024.
Under the IPPS MS-DRG classification system, we have developed a standard list of diagnoses that are considered CCs. Historically, we developed this list using physician panels that classified each diagnosis code based on whether the diagnosis, when present as a secondary condition, would be considered a substantial complication or comorbidity. A substantial complication or comorbidity was defined as a condition that, because of its presence with a specific principal diagnosis, would cause an increase in the length-of-stay by at least 1 day in at least 75 percent of the patients. However, depending on the principal diagnosis of the patient, some diagnoses on the basic list of complications and comorbidities may be excluded if they are closely related to the principal ( print page 69076) diagnosis. In FY 2008, we evaluated each diagnosis code to determine its impact on resource use and to determine the most appropriate CC subclassification (NonCC, CC, or MCC) assignment. We refer readers to sections II.D.2. and 3. of the preamble of the FY 2008 IPPS final rule with comment period for a discussion of the refinement of CCs in relation to the MS DRGs we adopted for FY 2008 ( 72 FR 47152 through 47171 ).
In the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47159 ), we described our process for establishing three different levels of CC severity into which we would subdivide the diagnosis codes. The categorization of diagnoses as a MCC, a CC, or a NonCC was accomplished using an iterative approach in which each diagnosis was evaluated to determine the extent to which its presence as a secondary diagnosis resulted in increased hospital resource use. We refer readers to the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47159 ) for a complete discussion of our approach. Since the comprehensive analysis was completed for FY 2008, we have evaluated diagnosis codes individually when assigning severity levels to new codes and when receiving requests to change the severity level of specific diagnosis codes.
We noted in the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19235 through 19246 ) that with the transition to ICD-10-CM and the significant changes that have occurred to diagnosis codes since the FY 2008 review, we believed it was necessary to conduct a comprehensive analysis once again. Based on this analysis, we proposed changes to the severity level designations for 1,492 ICD-10-CM diagnosis codes and invited public comments on those proposals. As summarized in the FY 2020 IPPS/LTCH PPS final rule, many commenters expressed concern with the proposed severity level designation changes overall and recommended that CMS conduct further analysis prior to finalizing any proposals. After careful consideration of the public comments we received, as discussed further in the FY 2020 IPPS/LTCH PPS final rule, we generally did not finalize our proposed changes to the severity designations for the ICD-10-CM diagnosis codes, other than the changes to the severity level designations for the diagnosis codes in category Z16 (Resistance to antimicrobial drugs) from a NonCC to a CC. We stated that postponing adoption of the proposed comprehensive changes in the severity level designations would allow further opportunity to provide additional background to the public on the methodology utilized and clinical rationale applied across diagnostic categories to assist the public in its review. We refer readers to the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42150 through 42152 ) for a complete discussion of our response to public comments regarding the proposed severity level designation changes for FY 2020.
As discussed in the FY 2021 IPPS/LTCH PPS proposed rule ( 85 FR 32550 ), to provide the public with more information on the CC/MCC comprehensive analysis discussed in the FY 2020 IPPS/LTCH PPS proposed and final rules, CMS hosted a listening session on October 8, 2019. The listening session included a review of this methodology utilized to mathematically measure the impact on resource use. We refer readers to https://www.cms.gov/Outreach-and-Education/Outreach/OpenDoorForums/Downloads/10082019ListingSessionTrasncriptandQandAsandAudioFile.zip for the transcript and audio file of the listening session. We also refer readers to https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software for the supplementary file containing the mathematical data generated using claims from the FY 2018 MedPAR file describing the impact on resource use of specific ICD-10-CM diagnosis codes when reported as a secondary diagnosis that was made available for the listening session.
In the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58550 through 58554 ), we discussed our plan to continue a comprehensive CC/MCC analysis, using a combination of mathematical analysis of claims data as discussed in the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19235 ) and the application of nine guiding principles and plan to present the findings and proposals in future rulemaking. The nine guiding principles are as follows:
- Represents end of life/near death or has reached an advanced stage associated with systemic physiologic decompensation and debility.
- Denotes organ system instability or failure.
- Involves a chronic illness with susceptibility to exacerbations or abrupt decline.
- Serves as a marker for advanced disease states across multiple different comorbid conditions.
- Reflects systemic impact.
- Post-operative/post-procedure condition/complication impacting recovery.
- Typically requires higher level of care (that is, intensive monitoring, greater number of caregivers, additional testing, intensive care unit care, extended length of stay).
- Impedes patient cooperation or management of care or both.
- Recent (last 10 years) change in best practice, or in practice guidelines and review of the extent to which these changes have led to concomitant changes in expected resource use.
We refer readers to the FY 2021 IPPS/LTCH PPS final rule for a complete summation of the comments we received for each of the nine guiding principles and our responses to those comments. In the proposed rule we noted that since the FY 2021 IPPS/LTCH PPS final rule we have continued to solicit feedback regarding the nine guiding principles, as well as other possible ways we can incorporate meaningful indicators of clinical severity. We have encouraged the public to provide a detailed explanation of how applying a suggested concept or principle would ensure that the severity designation appropriately reflects resource use for any diagnosis code when providing feedback or comments. In the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26748 through 26750 ) we illustrated how the nine guiding principles might be applied in evaluating changes to the severity designations of diagnosis codes in our discussion of our proposed changes to the severity level designation for certain diagnosis codes that describe homelessness. In the proposed rule, we stated that we have not received any additional feedback or comments on the nine guiding principles since the FY 2021 IPPS/LTCH PPS final rule; therefore, in the FY 2025 IPPS/LTCH PPS proposed rule we proposed to finalize the nine guiding principles as listed previously. We stated that under this proposal, our evaluations to determine the extent to which the presence of a diagnosis code as a secondary diagnosis results in increased hospital resource use will include a combination of mathematical analysis of claims data as discussed in the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19235 ) and the application of the nine guiding principles.
Comment: Many commenters supported our proposal to finalize the nine guiding principles. Commenters stated they continued to support CMS' consideration of the nine guiding principles in conjunction with its mathematical analysis of the data in ( print page 69077) evaluating whether changes to the severity level designations of diagnoses are needed and to ensure the severity designations appropriately reflect resource use based on review of the claims data, as well as consideration of relevant clinical factors (for example, the clinical nature of each of the secondary diagnoses and the severity level of clinically similar diagnoses).
Comments: Other commenters expressed concerns with the guiding principles. These commenters stated that the nine guiding principles appeared to be open to interpretation or differences in clinical opinion and noted a lack of detailed definitions and criteria for applying the guiding principles. Other commenters stated that it was not clear how CMS will apply the guiding principles in conjunction with the mathematical analyses of claims data to make decisions about severity levels. These commenters stated in their observation, CMS had not stated how it will handle conditions that might not fit any guiding principles, such as obstetrical diagnoses, congenital conditions, or potentially social determinants of health, but reflect mathematical data for the impact on resource use that could suggest a need for a change to the severity designation of the code. Several commenters stated they were unclear as to the impact finalizing the guiding principles would have on diagnosis codes that are currently designated as MCCs or CCs when reported as secondary diagnoses. These commenters stated that more information is needed to better understand CMS' process for decision making on the designation of diagnosis severity levels.
Response: We thank the commenters for sharing their concerns.
We note the focus of our analysis is on the appropriate severity level designation of individual ICD-10-CM codes as secondary diagnosis codes and how they relate to inpatient prospective payment and the resource utilization required while the patient is in the hospital. We wish to clarify for commenters that the application of the nine guiding principles is not a departure from our historic approach of considering both mathematical analysis and clinical factors as described in the FY 2008 IPPS/LTCH PPS final rule. In the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47153 through 47154 ), we stated the need for a revised CC list prompted a reexamination of the secondary diagnoses that qualify as a CC and stated our intent was to better distinguish cases that are likely to result in increased hospital resource use based on secondary diagnoses. We stated that using a combination of mathematical data and the judgment of our medical advisors, we included the condition on the CC list if it could demonstrate that its presence would lead to substantially increased hospital resource use. We stated diagnoses may require increased hospital resource use because of a need for such services as:
- Intensive monitoring (for example, an intensive care unit (ICU) stay).
- Expensive and technically complex services (for example, heart transplant).
- Extensive care requiring a greater number of caregivers (for example, nursing care for a patient with quadriplegia).
In reviewing the diagnosis codes that describe chronic diseases, we stated in the FY 2008 IPPS/LTCH PPS final rule that we made exceptions for diagnosis codes that indicate a chronic disease in which the underlying illness has reached an advanced stage or is associated with systemic physiologic decompensation and debility. We refer readers to the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47153 through 47154 ) for a complete discussion of our approach.
The nine guiding principles were developed to build on the process we described in the FY 2008 IPPS/LTCH PPS final rule and are not intended to turn the analysis into a quantitative exercise, requiring that every diagnosis code satisfy each principle. Instead, as stated in prior rulemaking, the nine guiding principles are intended to provide a framework for assessing relevant clinical factors to help denote if, and to what degree, additional resources are required above and beyond those that are already being utilized to address the principal diagnosis or other secondary diagnoses that might also be present on the claim. In response to the commenter's concerns regarding a lack of detailed definition of each principle, we refer commenters to the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58550 through 58554 ), for a complete discussion of our response to similar public comments regarding each of the nine guiding principles.
Comment: Some commenters stated that the guiding principles appeared to be more applicable to MCC conditions, were too strict, and could potentially eliminate CC conditions. A commenter stated that the application of the guiding principles would represent a substantial revision to the definition of a CC, noting MS-DRG Definition Manual Version 41.1 provides the following definition: “A substantial complication or comorbidity was defined as a condition that because of its presence with a specific principal diagnosis would cause an increase in length of stay by at least one day in at least 75 percent of the patients.”
Response: We appreciate the commenters' feedback.
We do not believe the nine guiding principles would be mostly applicable, or only applicable, to MCC conditions. In applying the nine guiding principles in our review of the appropriate severity level designation, the intention is not to require that a diagnosis code satisfy each principle, or a specific number of principles in assessing whether to designate a secondary diagnosis code as a NonCC versus a CC versus an MCC. Rather, the severity level determinations would be based on the consideration of the clinical factors captured by these principles as well as the empirical analysis of the additional resources associated with the secondary diagnosis.
We wish to clarify that the definition of a “substantial complication or comorbidity” from the MS-DRG Definition Manual that the commenter referenced, is the definition of a CC that was used in Version 8 of the DRGs. In FY 2008, for Version 25 of the MS-DRGs, the diagnoses comprising the CC list were completely redefined and instead each CC was categorized as a major CC or a CC (that is, non-major CC) based on relative resource use. As stated previously, we refer readers to the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47159 ) for a complete discussion of our approach.
We note that in addition to the FY 2021 IPPS/LTCH PPS rule, in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44915 through 44926 ), the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48865 through 48872 ), the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58753 through 58759 ), and in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35997 through 36001 ), we have illustrated how the guiding principles might be applied in evaluating changes to the severity designations of diagnosis codes. We have also continued to solicit feedback regarding the guiding principles, as well as other possible ways we can incorporate meaningful indicators of clinical severity. We note the commenters did not provide alternative principles for consideration, nor was feedback provided as to other possible ways we can incorporate meaningful indicators of clinical severity.
As discussed in prior rulemaking, our intended approach is for CMS to first use these guiding principles in making an initial clinical assessment of the appropriate severity level designation ( print page 69078) for each ICD-10-CM code as a secondary diagnosis. CMS will then use a mathematical analysis of claims data as discussed in the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47159 ) to determine if the presence of the ICD-10-CM code as a secondary diagnosis appears to, or does not appear to, increase hospital resource consumption. There may be instances in which we would decide that the clinical analysis weighs in favor of proposing to maintain or proposing to change the severity designation of an ICD-10-CM code after application of the nine guiding principles. Any proposed modifications to the severity level designation of ICD-10-CM codes would be addressed in future rulemaking consistent with our annual process.
Therefore, after consideration of the public comments received, and for the reasons discussed, we are finalizing the nine guiding principles as listed previously in this FY 2025 IPPS/LTCH PPS final rule. Accordingly, our evaluations to determine the extent to which the presence of a diagnosis code as a secondary diagnosis results in increased hospital resource use will include a combination of mathematical analysis of claims data as discussed in the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19235 ) and the application of the nine guiding principles. We thank commenters for sharing their views and their willingness to support CMS in our efforts to continue a comprehensive CC/MCC analysis.
In the FY 2022 IPPS/LTCH PPS proposed rule ( 86 FR 25175 through 25180 ), as another interval step in our comprehensive review of the severity designations of ICD-10-CM diagnosis codes, we requested public comments on a potential change to the severity level designations for “unspecified” ICD-10-CM diagnosis codes that we were considering adopting for FY 2022. Specifically, we noted we were considering changing the severity level designation of “unspecified” diagnosis codes to a NonCC where there are other codes available in that code subcategory that further specify the anatomic site. As summarized in the FY 2022 IPPS/LTCH PPS final rule, many commenters expressed concern with the potential severity level designation changes overall and recommended that CMS delay any possible change to the designation of these codes to give hospitals and their physicians time to prepare. After careful consideration of the public comments we received, we maintained the severity level designation of the “unspecified” diagnosis codes currently designated as a CC or MCC where there are other codes available in that code subcategory that further specify the anatomic site for FY 2022. We refer readers to the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44916 through 44926 ) for a complete discussion of our response to public comments regarding the potential severity level designation changes. Instead, for FY 2022, we finalized a new Medicare Code Editor (MCE) code edit for “unspecified” codes, effective with discharges on and after April 1, 2022. We stated we believe finalizing this new edit would provide additional time for providers to be educated while not affecting the payment the provider is eligible to receive. We refer the reader to section II.D.14.e. of the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44940 through 44943 ) for the complete discussion.
As discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48866 ), we stated that as the new unspecified edit became effective beginning with discharges on and after April 1, 2022, we believed it was appropriate to not propose to change the designation of any ICD-10-CM diagnosis codes, including the unspecified codes that are subject to the “Unspecified Code” edit, as we continue our comprehensive CC/MCC analysis to allow interested parties the time needed to become acclimated to the new edit.
Comment: A commenter stated that they were pleased that CMS continues to maintain the severity level designation of the “unspecified” diagnosis codes, currently designated as a CC or MCC where there are other codes available in that code subcategory that further specify the anatomic site, that are subject to the “Unspecified Code” edit. The commenter further stated that they agreed that maintaining this status quo will allow time for providers to be educated and adjust to the edit. Another commenter suggested that CMS provide data from the Medicare Code Editor (MCE) that identifies each provider reporting “unspecified” diagnosis codes with designations as a CC or MCC when there are other codes available in that code subcategory that further specify the anatomic site, which can be used to inform providers on the number of “unspecified” diagnosis codes being reported at their facility compared to their peers.
Response: CMS appreciates the commenters' feedback and recommendations. We will give careful consideration to what additional information may be helpful in assisting to educate providers on the documentation required to report to the highest level of specificity as it relates to the laterality of the conditions treated in the inpatient setting as we continue to formulate future next steps in our comprehensive review of the severity designations of ICD-10-CM diagnosis codes.
In the FY 2023 IPPS/LTCH proposed rule ( 87 FR 28177 through 28181 ), we also requested public comments on how the reporting of diagnosis codes in categories Z55-Z65 might improve our ability to recognize severity of illness, complexity of illness, and/or utilization of resources under the MS-DRGs. Consistent with the Administration's goal of advancing health equity for all, including members of historically underserved and under-resourced communities, as described in the President's January 20, 2021 Executive Order 13985 on “Advancing Racial Equity and Support for Underserved Communities Through the Federal Government,” [ 8 ] we stated we were also interested in receiving feedback on how we might otherwise foster the documentation and reporting of the diagnosis codes describing social and economic circumstances to more accurately reflect each health care encounter and improve the reliability and validity of the coded data including in support of efforts to advance health equity.
We noted that social determinants of health (SDOH) are the conditions in the environments where people are born, live, learn, work, play, worship, and age that affect a wide range of health, functioning, and quality-of-life outcomes and risks. [ 9 ] The subset of Z codes that describe the social determinants of health are found in categories Z55-Z65 (Persons with potential health hazards related to socioeconomic and psychosocial circumstances). These codes describe a range of issues related—but not limited—to education and literacy, employment, housing, ability to obtain adequate amounts of food or safe drinking water, and occupational exposure to toxic agents, dust, or radiation.
We received numerous public comments that expressed a variety of views on our comment solicitation, including many comments that were supportive, and others that offered specific suggestions for our consideration in future rulemaking. Many commenters applauded CMS' efforts to encourage documentation and ( print page 69079) reporting of SDOH diagnosis codes given the impact that social risks can have on health outcomes. These commenters stated that it is critical that physicians, other health care professionals, and facilities recognize the impact SDOH have on the health of their patients. Many commenters also stated that the most immediate and important action CMS could take to increase the use of SDOH Z codes is to finalize the evidence-based “Screening for Social Drivers of Health” and “Screen Positive Rate for Social Drivers of Health” measures proposed to be adopted in the Hospital Inpatient Quality Reporting (IQR) Program. In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49202 through 49220 ), CMS finalized the “Screening for Social Drivers of Health” and “Screen Positive Rate for Social Drivers of Health” measures in the Hospital Inpatient Quality Reporting (IQR) Program. We refer readers to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48867 through 48872 ) for the complete discussion of the public comments received regarding the request for information on SDOH diagnosis codes.
As discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58755 through 58759 ), based on our analysis of the impact on resource use for the ICD-10-CM Z codes that describe homelessness and after consideration of public comments, we finalized changes to the severity levels for diagnosis codes Z59.00 (Homelessness, unspecified), Z59.01 (Sheltered homelessness), and Z59.02 (Unsheltered homelessness), from NonCC to CC. We stated our expectation that finalizing the changes would encourage the increased documentation and reporting of the diagnosis codes describing social and economic circumstances and serve as an example for providers that, when they document and report SDOH codes, CMS can further examine the claims data and consider future changes to the designation of these codes when reported as a secondary diagnosis. We further stated CMS would continue to monitor and evaluate the reporting of the diagnosis codes describing social and economic circumstances.
We refer the reader to the following section of this final rule for discussion of our proposed changes to the severity level designation for the diagnosis codes that describe inadequate housing and housing instability for FY 2025, as well as our finalization of that proposal.
We have updated the Impact on Resource Use Files on the CMS website so that the public can review the mathematical data for the impact on resource use generated using claims from the FY 2019 through the FY 2023 MedPAR files. These files are posted on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software . As discussed in prior rulemaking, we also continue to be interested in receiving feedback on how we might further foster the documentation and reporting of the most specific diagnosis codes supported by the available medical record documentation and clinical knowledge of the patient's health condition to more accurately reflect each health care encounter and improve the reliability and validity of the coded data.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35997 ), for new diagnosis codes approved for FY 2025, consistent with our annual process for designating a severity level (MCC, CC, or NonCC) for new diagnosis codes, we first review the predecessor code designation, followed by review and consideration of other factors that may be relevant to the severity level designation, including the severity of illness, treatment difficulty, complexity of service and the resources utilized in the diagnosis or treatment of the condition. We note that this process does not automatically result in the new diagnosis code having the same designation as the predecessor code. We refer the reader to section II.C.13 of this final rule for the discussion of the finalized changes to the ICD-10-CM and ICD-10-PCS coding systems for FY 2025.
As discussed earlier in this section and in the proposed rule ( 89 FR 35997 through 35999 ), in continuation of our examination of the SDOH Z codes, we reviewed the mathematical data on the impact on resource use for the subset of ICD-10-CM Z codes that describe the social determinants of health found in categories Z55-Z65 (Persons with potential health hazards related to socioeconomic and psychosocial circumstances).
As discussed in the proposed rule, the ICD-10-CM SDOH Z codes that describe inadequate housing and housing instability are currently designated as NonCCs when reported as secondary diagnoses. The following table reflects the impact on resource use data generated using claims from the September 2023 update of the FY 2023 MedPAR file. We refer readers to the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47159 ) for a complete discussion of our historical approach to mathematically evaluate the extent to which the presence of an ICD-10-CM code as a secondary diagnosis resulted in increased hospital resource use, and a more detailed explanation of the columns in the table.

The table shows that the C1 value is 2.63 for ICD-10-CM diagnosis code Z59.10 and 1.85 for ICD-10-CM diagnosis code Z59.19. A value close to 2.0 in column C1 suggests that the secondary diagnosis is more aligned with a CC than a NonCC. Because the C1 values in the table are generally close to 2, the data suggest that when these two SDOH Z codes are reported as a secondary diagnosis, the resources involved in caring for a patient experiencing inadequate housing support increasing the severity level from a NonCC to a CC. In contrast, the C1 value for ICD-10-CM diagnosis code Z59.11 is 0.51 and is 0.99 for ICD-10-CM diagnosis code Z59.12. A C1 value generally closer to 1 suggests the resources involved in caring for patients experiencing inadequate housing in terms of environmental temperature and utilities are more aligned with a NonCC severity level than a CC or an MCC severity level.
As discussed in the proposed rule, the underlying cause of the inconsistency between the C1 values for inadequate housing, unspecified and other inadequate housing and the two more specific codes that describe the necessities unavailable in the housing environment is unclear. We noted that diagnosis codes Z59.10 (Inadequate housing, unspecified), Z59.11 (Inadequate housing environmental temperature), Z59.12 (Inadequate housing utilities), and Z59.19 (Other inadequate housing) became effective on April 1, 2023 (FY 2023). In reviewing the historical C1 values for code Z59.1 (Inadequate housing), the predecessor code before the code was expanded to further describe inadequate housing and the basic necessities unavailable in the housing environment, we noted the mathematical data for the impact on resource use generated using claims from the FY 2019, FY 2020, FY 2021, and FY 2022 MedPAR files reflects C1 values for code Z59.1 of 2.09, 1.73, 2.04, and 2.69, respectively. We refer the reader to the Impact on Resource Use Files generated using claims from the FY 2019 through the FY 2022 MedPAR files posted on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software . We stated we believe the lower C1 values for ICD-10-CM codes Z59.11 (Inadequate housing environmental temperature) and Z59.12 (Inadequate housing utilities) reflected in the mathematical data for the impact on resource use generated using claims from the FY 2023 MedPAR file may be attributed to lack of use or knowledge about the newly expanded codes, such that the data may not yet reflect the full impact on resource use for patients experiencing these circumstances.
Similarly, the table shows that the C1 value is 1.97 for ICD-10-CM diagnosis code Z59.811. A value close to 2.0 in column C1 suggests that the secondary diagnosis is more aligned with a CC than a NonCC. Because the C1 value in the table is generally close to 2, the data suggest that when this SDOH Z code is reported as a secondary diagnosis, the resources involved in caring for a patient experiencing an imminent risk of homelessness support increasing the severity level from a NonCC to a CC. In contrast, the C1 value for ICD-10-CM diagnosis code Z59.812 (Housing instability, housed, homelessness in past 12 months) and (Housing instability, housed unspecified) is 0.76 and is 0.92 for ICD-10-CM diagnosis code Z59.819. A C1 value generally closer to 1 suggests the resources involved in caring for patients experiencing housing instability, with history of homelessness in the past 12 months or housing instability, unspecified are more aligned with a NonCC severity level than a CC or an MCC severity level. We stated in the proposed rule that the underlying cause of the inconsistency between the C1 values for codes describing housing instability is unclear.
In the proposed rule, we noted that diagnosis codes Z59.811, Z59.812, and Z59.819 became effective on October 1, 2021 (FY 2022). In reviewing the historical C1 values for code Z59.8 (Other problems related to housing and economic circumstances), the predecessor code before the code was expanded to further describe the ( print page 69081) problems related to housing and economic circumstances, we noted the mathematical data for the impact on resource use generated using claims from the FY 2019 and FY 2020 MedPAR files reflects C1 values for code Z59.8 of 1.92 and 1.63, respectively. There were no data reflected for this code in the Impact on Resource Use File generated using claims from the FY 2021 MedPAR files. The mathematical data for the impact on resource use generated using claims from the FY 2022 MedPAR file reflects C1 values for codes Z59.811, Z59.812, and Z59.819 of 2.44, 3.12, and 2.09, respectively. We stated we were uncertain if the fluctuations in the C1 values from year to year, or FY 2021, in particular, may reflect fluctuations that may be a result of the COVID-19 public health emergency or even reduced hospitalizations of certain conditions. We stated we were also uncertain if the fluctuations may be attributed to lack of use or knowledge about the expanded codes, such that the data on the reporting of codes Z59.812 and Z59.819 may not yet reflect the full impact on resource use for patients experiencing these circumstances.
As discussed in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58550 through 58554 ), and earlier in this section, following the listening session on October 8, 2019, we reconvened an internal workgroup comprised of clinicians, consultants, coding specialists and other policy analysts to identify guiding principles to apply in evaluating whether changes to the severity level designations of diagnoses are needed and to ensure the severity designations appropriately reflect resource use based on review of the claims data, as well as consideration of relevant clinical factors (for example, the clinical nature of each of the secondary diagnoses and the severity level of clinically similar diagnoses) and improve the overall accuracy of the IPPS payments.
In considering the nine guiding principles identified by the workgroup, as summarized previously, in the proposed rule we noted that, similar to homelessness, inadequate housing and housing instability are circumstances that can impede patient cooperation or management of care, or both. In addition, patients experiencing inadequate housing and housing instability can require a higher level of care by needing an extended length of stay.
Inadequate housing is defined as an occupied housing unit that has moderate or severe physical problems (for example, deficiencies in plumbing, heating, electricity, hallways, and upkeep). [ 10 11 ] Features of substandard housing have long been identified as contributing to the spread of infectious diseases. Patients living in inadequate housing may be exposed to health and safety risks, such as vermin, mold, water leaks, and inadequate heating or cooling systems. [ 12 13 ] An increasing body of evidence has associated poor housing conditions with morbidity from infectious diseases, chronic illnesses, exposure to toxins, injuries, poor nutrition, and mental disorders. [ 14 ]
As discussed in the proposed rule, housing instability encompasses a number of challenges, such as having trouble paying rent, overcrowding, moving frequently, or spending the bulk of household income on housing. [ 15 ] These experiences may negatively affect physical health and make it harder to access health care. Studies have found moderate evidence to suggest that housing instability is associated with higher prevalence of overweight/obesity, hypertension, diabetes, and cardiovascular disease, worse hypertension and diabetes control, and higher acute health care utilization among those with diabetes and cardiovascular disease. [ 16 ]
In reviewing the mathematical data for the impact on resource use generated using claims from the FY 2023 MedPAR file for the seven ICD-10-CM codes describing inadequate housing and housing instability comprehensively and reviewing the potential impact these circumstances could have on patients' clinical course, we noted in the proposed rule that whether the patient is experiencing inadequate housing or housing instability, the patient may have limited or no access to prescription medicines or over-the-counter medicines, including adequate locations to store medications away from the heat or cold, and have difficulties adhering to medication regimens. Experiencing inadequate housing or housing instability may negatively affect a patient's physical health and make it harder to access timely health care. [ 12 ] Delays in medical care may increase morbidity and mortality risk among those with underlying, preventable, and treatable medical conditions. [ 17 ] In addition, we noted that findings also suggest that patients experiencing inadequate housing or housing instability are associated with higher rates of inpatient admissions for mental, behavioral, and neurodevelopmental disorders, longer hospital stays, and substantial health care costs. [ 18 ]
Therefore, after considering the impact on resource use data generated using claims from the September 2023 update of the FY 2023 MedPAR file for the seven ICD-10-CM diagnosis codes that describe inadequate housing and housing instability and consideration of the nine guiding principles, we proposed to change the severity level designation for diagnosis codes Z59.10 (Inadequate housing, unspecified), Z59.11 (Inadequate housing environmental temperature), Z59.12 (Inadequate housing utilities), Z59.19 (Other inadequate housing), Z59.811 (Housing instability, housed, with risk of homelessness), Z59.812 (Housing instability, housed, homelessness in past 12 months) and Z59.819 (Housing instability, housed unspecified) from NonCC to CC for FY 2025.
Comment: Commenters expressed overwhelming support for our proposal to change the severity level designation for diagnosis codes Z59.10 (Inadequate housing, unspecified), Z59.11 (Inadequate housing environmental temperature), Z59.12 (Inadequate housing utilities), Z59.19 (Other inadequate housing), Z59.811 (Housing instability, housed, with risk of homelessness), Z59.812 (Housing ( print page 69082) instability, housed, homelessness in past 12 months) and Z59.819 (Housing instability, housed unspecified) from NonCC to CC for FY 2025. These commenters stated this proposal acknowledges the significant impact these circumstances can have on patient outcomes and the increased resource allocation required to effectively manage the care of these patients in terms of increased severity of illness, readmissions resulting from lack of follow-up and continued medical treatment and delayed discharges. A commenter stated that changing the severity level of the seven ICD-10-CM diagnosis codes that describe inadequate housing and housing instability is not only appropriate, it is crucial in order to help address the complex needs of these patients. Commenters stated that this change is a critical step toward increasing health care access for underserved and under-resourced communities and in recognizing and addressing broader factors that impact patient health. A commenter specifically stated this proposal is another notable effort on CMS' part to recognize the interconnectedness of health and social needs. Another commenter stated this change will encourage providers to ask more detailed questions of patients to better understand their housing status, improving overall data quality.
Comment: While commending CMS' efforts, many commenters noted an operational concern in that currently only 25 diagnoses are captured on the institutional electronic claim form and 19 diagnoses are captured on the paper bill. Many commenters stated this issue is becoming increasingly critical as Medicare and other payers move to implement new quality measures that emphasize the screening and identification of patient-level, health-related social needs, which will dictate the need for reporting additional codes. Commenters stated that documenting and reporting the social and economic circumstances patients may be experiencing can require a substantial number of SDOH Z codes, which could lead to the crowding out of other diagnosis codes that also need to be captured on the institutional claim form for both payment and quality measures. Commenters suggested that a factor that may be negatively impacting more comprehensive reporting of the diagnosis codes describing social and economic circumstances is the limit on the number of diagnoses that may be reported on an inpatient claim. A commenter stated they performed their own analysis of the FY 2021 MedPAR file and found that 17 percent of inpatient claims reached the maximum limit of 25 diagnoses that can be reported on the claim. Another commenter stated at their facility approximately one-third of cases have 26 codes or more, with some cases reporting as many as 45 codes. A few commenters suggested that CMS evaluate the potential to expand the number of diagnosis codes that can be submitted, or alternatively, design a separate way to report the Z codes on the claim form, separate and distinct from the fields for the diagnosis codes.
Response: We thank the commenters for their continued feedback on this issue. We note that any proposed changes to the institutional claim form would need to be submitted to the National Uniform Billing Committee (NUBC) for consideration as the NUBC develops and maintains the Uniform Billing (UB) 04 data set and form, not CMS. The NUBC is a Data Content Committee named in the Health Insurance Portability and Accountability Act of 1996 (HIPAA) and is composed of a diverse group of interested parties representing providers, health plans, designated standards maintenance organizations, public health organizations, and vendors.
Comment: Another commenter expressed concern that CMS continues to delay the comprehensive analysis of the severity designation of all the diagnosis codes in the ICD-10-CM classification in favor of reviewing the subset of ICD-10-CM Z codes that describe the social determinants of health. The commenter stated in their view, ensuring MS-DRG payment is congruent with what it costs to care for patients should be CMS' primary objective. Many other commenters encouraged CMS to examine other SDOH Z codes that describe circumstances such as lack of adequate food and drinking water, extreme poverty, lack of transportation, and problems related to employment, physical environment, social environment, upbringing, primary support group, literacy, economic circumstances, and psychosocial circumstances to determine the hospital resource utilization related to addressing these factors and to analyze whether these SDOH Z codes should be considered for severity designation changes in future rulemaking as well. A commenter noted that these social needs create substantial barriers to healthcare and good health, both before and after receiving care.
Specifically, many commenters stated that research has found a strong association between food insecurity and chronic conditions and encouraged CMS to examine the severity designation of ICD-10-CM SDOH Z code Z59.41 (Food insecurity). These commenters stated that food insecurity can be an indicator of food deprivation, malnutrition, or lack of access to healthy foods and diet, which could have differing impacts on a patient and could be associated with higher healthcare utilization and costs. A commenter stated that in their observation, many hospitals have built robust programs to address the food needs of inpatients and stated that several hospitals have even begun to provide patients with fresh fruit, vegetables, and other essential groceries to take home upon discharge without payment.
Response: We appreciate the feedback.
We note that as described in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58761 ), CMS has undertaken interval steps towards a comprehensive CC/MCC analysis. We stated in the FY 2024 IPPS/LTCH PPS final rule, considering the potential impact of implementing a significant number of severity designation changes, and in light of the public health emergency (PHE) that was occurring concurrently from 2020 until 2023, we believe these interval steps were appropriate as we plan to continue a comprehensive CC/MCC analysis, using a combination of mathematical analysis of claims data and the application of nine guiding principles. We refer the reader to the discussion earlier in this section where we discuss the finalization of the nine guiding principles for FY 2025.
In response to comments that CMS examine the severity designation of ICD-10-CM code Z59.41 (Food insecurity), we note that ICD-10-CM code Z59.41 is currently designated as a NonCC when reported as a secondary diagnosis. The following table reflects the impact on resource use data generated using claims from the September 2023 update of the FY 2023 MedPAR file for code Z59.41. We refer readers to the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47159 ) for a complete discussion of our historical approach to mathematically evaluate the extent to which the presence of an ICD-10-CM code as a secondary diagnosis resulted in increased hospital resource use, and a more detailed explanation of the columns in the table.

The table shows that the C1 value is 0.9273 for ICD-10-CM diagnosis code is Z59.41. A C1 value generally closer to 1 suggests the resources involved in caring for patients experiencing food insecurity are more aligned with a NonCC severity level, as the code is currently designated, rather than a CC or an MCC severity level. This contrasts with the conclusions documented in research that has shown that food insecurity empirically can be associated with higher healthcare use and costs, even when accounting for other socioeconomic factors. [ 19 ] We note that the table also shows that code Z59.41 was only reported in a total of 6,634 claims in the September 2023 update of the FY 2023 MedPAR file.
The impact on resource use data generated using claims from the September 2023 update of the FY 2023 MedPAR file for code Z59.41 again illustrates that if SDOH Z codes are not consistently reported in inpatient claims data, our methodology utilized to mathematically measure the impact on resource use, as described previously, may not adequately reflect what additional resources were expended by hospitals to address these circumstances. If SDOH Z codes are consistently reported in inpatient claims, the impact on resource use data may more adequately reflect what additional resources were expended to address these SDOH circumstances in terms of requiring clinical evaluation, extended length of hospital stay, increased nursing care or monitoring or both, and comprehensive discharge planning and we can re-examine these severity designations in future rulemaking.
In Table 6P.3b associated with this final rule, we have made available the data generated using claims from the September 2023 update of the FY 2023 MedPAR file describing the impact on resource use when reported as a secondary diagnosis for the ICD-10-CM codes describing various diagnoses and circumstances that commenters to the FY 2025 IPPS/LTCH proposed rule suggested CMS review to determine if changes to the severity level designations are warranted in future rulemaking. These data are consistent with data historically used to mathematically measure impact on resource use for secondary diagnoses, and the data which we will use in combination with application of the nine guiding principles as we continue the comprehensive CC/MCC analysis. We will examine these suggestions and determine if there are other diagnoses codes, including diagnosis codes that describe SDOH, that should also be considered further and will provide more detail in future rulemaking.
Comment: Some commenters stated there continue to be many challenges for clinicians in documenting SDOH, such as the lack of knowledge surrounding these codes. Many commenters stated there was a lack of standard, nationally accepted definitions of the SDOH Z codes and that ambiguity between Z codes can lead to confusion among clinical staff. A commenter stated that CMS should engage with key stakeholders, including patients and diverse communities to establish a transparent process and timeline for updating Z code terms and definitions.
Other commenters stated that healthcare providers may gravitate towards certain codes, while other, possibly more specific codes, may exist in the classification that could accurately describe similar situations. These commenters stated that this can lead to inconsistent code usage and can limit the ability to see any correlations between these particular conditions and the resulting patient complexity and additional cost of care. A commenter noted that the difference between the seven different codes describing inadequate housing and housing instability requires a nuanced understanding and stated that appropriately documenting the Z codes for inadequate housing and housing instability will require training of staff to understand the differences. Another commenter suggested that CMS focus on addressing infrastructural, technological, and knowledge gaps to facilitate use of Z codes and stated if CMS does not address these gaps, inequities between well-funded hospitals that can afford to train staff to document and report Z codes as compared to other struggling hospitals who lack the means or know-how will be exacerbated.
A commenter expressed concern and stated that documentation of an SDOH circumstance does not always clearly demonstrate whether or how the SDOH impacts the patient's health. This commenter stated that while SDOH Z codes help with mapping the social factors afflicting a patient, that map does not (and cannot) fully describe the patient's life which can present a challenge for the provider when determining what factors to document, and for the coder, when deciding how to report that documentation using the appropriate SDOH Z code(s).
Many other commenters also expressed concern and stated that while they support the use of Z codes to help identify the complexity of issues impacting patients, information about an individual's social risk and needs has been shown to be sensitive. Commenters stated that expressing certain circumstances, such as housing instability or inadequacy, can be uncomfortable for patients, which could result in underreporting. These commenters also stated they believed the descriptions of ICD-10-CM SDOH Z codes can be stigmatizing, and therefore the descriptions should be changed to be more patient friendly to protect the patient-provider relationship since patients can access their code assignments in after-visit summaries.
Response: We appreciate the feedback. As discussed in section II.C.15 of the preamble of this final rule, the CDC/NCHS has lead responsibility for the diagnosis code classification. In response to the suggestion that transparent process and timeline be established for updating Z code terms ( print page 69084) and definitions, we note there is an established process as the ICD-10 Coordination and Maintenance Committee addresses updates to the ICD-10-CM and ICD-10-PCS coding systems, as also discussed in section II.C.15 of the preamble of this final rule. The ICD-10 Coordination and Maintenance Committee holds its meetings each spring and fall to update the codes and the applicable payment and reporting systems by October 1 or April 1 of each year. Proposals for updates to the diagnosis codes, including diagnosis codes describing social determinants of health should be directed to [email protected] for consideration at a future ICD-10 Coordination and Maintenance Committee meeting.
We also note that the ICD-10-CM Official Guidelines for Coding and Reporting have been regularly revised to provide additional guidance as it relates to diagnosis codes describing social determinants of health. We encourage the commenters to review the Official ICD-10-CM Coding Guidelines, which can be found on the CDC website at: https://www.cdc.gov/nchs/icd/icd-10-cm/files.html . The American Hospital Association (AHA)'s Coding Clinic for ICD-10-CM/PCS publication has provided further clarification on the appropriate documentation and use of Z codes to enable hospitals to incorporate them into their processes. The AHA also offers a range of tools and resources for hospitals, health systems and clinicians to address the social needs of their patients. We believe these updates and resources will help alleviate the concerns expressed by these commenters. As one of the four Cooperating Parties for ICD-10, we will continue to collaborate with the AHA to provide guidance for coding problems or risk factors related to SDOH through the AHA's Coding Clinic for ICD-10-CM/PCS publication and to review the ICD-10-CM Coding Guidelines to determine where further clarifications may be made.
Comment: Some commenters recommended that CMS consider payment incentives for documenting and reporting of SDOH Z codes. Several commenters encouraged CMS to explore additional incentives for Z code utilization that do not rely on a code-by-code approach. While applauding CMS proposing to change the severity designation of the seven ICD-10-CM diagnosis codes that describe inadequate housing and housing instability, a commenter stated they also believe it is imperative that CMS continue to take steps towards more fundamental payment and delivery reforms, such as by directly addressing the social drivers of health under alternative payment models (APM) or the Hospital Value-based Purchasing (HVBP) Health Equity Adjustment (HEA), to hold providers accountable for high value, whole person care.
A few commenters stated that simply changing the severity designation of SDOH Z codes to CCs and marginally increasing payment will be inadequate to meaningfully drive CMS' stated equity mission. These commenters stated CMS' reporting and payment rules should better reflect and compensate hospitals for the multiple health-related social needs that patients experience to truly improve health outcomes and mitigate the current health disparities that exist. A commenter suggested that CMS consider an alternative policy that would provide increased payment for a CC designation only in certain MS-DRGs when certain Z codes are reported. Another commenter stated that they believed that the creation of a new Hierarchical Condition Category (HCC) for SDOH Z codes is needed to foster necessary support for delivering consistent levels of care and could help mitigate the challenges that social risk factors pose to creating effective treatment plans for patients. Some commenters suggested that CMS incentivize the use of patient self-report screening tools that are integrated within electronic health records to support the use of Z codes.
Other commenters noted that currently, if another secondary diagnosis designated as a CC or MCC is documented and reported, there will be no additional payment if the clinician reports a diagnosis code describing inadequate housing and housing instability, potentially minimalizing the practical impact of changing the severity level designation of these codes. These commenters recommended CMS provide increased flexibility in payment to account for multiple CCs or MCCs that may be reported for a given patient who may be experiencing numerous health and social concerns at the same time, stating that it is almost impossible to isolate and address only one need and expect an improved health outcome in their view.
Response: We thank commenters for sharing their views and recommendations. We will take the commenters' feedback into consideration in future policy development.
After consideration of the public comments received, we are finalizing changes to the severity levels for diagnosis codes Z59.10, Z59.11, Z59.12, Z59.19, Z59.811, Z59.812, and Z59.819, from NonCC to CC for FY 2025, without modification. In addition, these diagnosis codes are reflected in Table 6J.1—Additions to the CC List—FY 2025 associated with this final rule and available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps . We refer the reader to section II.C.12.d of the preamble of the proposed rule and this final rule for further information regarding Table 6J.1.
We hope and expect that this finalization will foster the increased documentation and reporting of the diagnosis codes describing social and economic circumstances and continue to serve as an example for providers that when they document and report Z codes, CMS can further examine the claims data and consider future changes to the designation of these codes when reported as a secondary diagnoses. As discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48868 ), if SDOH Z codes are not consistently reported in inpatient claims data, our methodology utilized to mathematically measure the impact on resource use, as described previously, may not adequately reflect what additional resources were expended by the hospital to address these SDOH circumstances in terms of requiring clinical evaluation, extended length of hospital stay, increased nursing care or monitoring or both, and comprehensive discharge planning. We will continue to monitor SDOH Z code reporting, including reporting based on SDOH screening performed as a result of quality measures in the Hospital Inpatient Quality Reporting program.
Furthermore, we may consider proposing changes for other diagnosis codes, including SDOH codes, in the future based on our analysis of the impact on resource use, per our methodology, as previously described, and consideration of the guiding principles. We continue to be interested in receiving feedback on how we might otherwise foster the documentation and reporting of the diagnosis codes to more accurately reflect each health care encounter and improve the reliability and validity of the coded data.
To inform future rulemaking, feedback and other suggestions may be submitted by October 20, 2024, and directed to MEARIS TM at: https://mearis.cms.gov/public/home .
Additionally, as discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35999 through 36001 ), we received a request to change the severity level designations of the ICD-10-CM diagnosis codes that describe causally ( print page 69085) specified delirium from CC to MCC when reported as secondary diagnoses. Causally specified delirium is delirium caused by the physiological effects of a medical condition, by the direct physiological effects of a substance or medication, including withdrawal, or by multiple or unknown etiological factors. The requestor noted that ICD-10-CM diagnosis codes G92.8 (Other toxic encephalopathy), G92.9 (Unspecified toxic encephalopathy) and G93.41 (Metabolic encephalopathy) are currently all designated as MCCs. According to the requestor, a diagnosis of delirium implies an underlying acute encephalopathy, and as such, the severity designation of the diagnosis codes that describe causally specified delirium should be on par with the severity designation of the diagnosis codes that describe toxic encephalopathy and metabolic encephalopathy. The requestor stated that toxic encephalopathy, metabolic encephalopathy, and causally specified delirium all describe core symptoms of impairment of level of consciousness and cognitive change caused by a medical condition or substance.
As noted in the proposed rule, the requestor further stated that there is robust literature detailing the impact delirium can have on cognitive decline, rates of functional decline, subsequent dementia diagnosis, institutionalization, care complexity and costs, readmission rates, and mortality. The requestor considered each of the nine guiding principles discussed earlier in this section and noted how each of the principles could be applied in evaluating changes to the severity designations of the diagnosis codes that describe causally specified delirium in their request. Specifically, the requestor stated that delirium is a textbook example that maps to the nine guiding principles for evaluating a potential change in severity designation in that delirium (1) has a bidirectional link with dementia, (2) indexes physiological vulnerability across populations, (3) impacts healthcare systems across levels of care, (4) complicates postoperative recovery, (5) consigns patients to higher levels of care, and for longer, (6) impedes patient engagement in care, (7) has several recent treatment guidelines, (8) indicates neuronal/brain injury, and (9) represents a common expression of terminal illness.
The requestor identified 37 ICD-10-CM diagnosis codes that describe causally specified delirium. In the proposed rule we stated we agree that these 37 diagnosis codes are all currently designated as CCs. We refer the reader to Appendix G of the ICD-10 MS-DRG Version 41.1 Definitions Manual (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for the complete list of diagnoses designated as CCs when reported as secondary diagnoses, except when used in conjunction with the principal diagnosis in the corresponding CC Exclusion List in Appendix C.
To evaluate this request, as discussed in the proposed rule, we analyzed the claims data in the September 2023 update of the FY 2023 MedPAR file. The following table shows the analysis for each of the diagnosis codes identified by the requestor that describe causally specified delirium.

As discussed in the proposed rule, we analyzed these data as described in FY 2008 IPPS final rule ( 72 FR 47158 through 47161 ). The table shows that the C1 values of the diagnosis codes that describe causally specified delirium range from a low of 0.35 to a high of 4.00. As stated earlier, a C1 value close to 2.0 suggests the condition is more like a CC than a NonCC but not as significant in resource usage as an MCC. On average, the C1 values of the diagnoses that describe causally specified delirium suggest that these codes are more like a NonCC than a CC. In the proposed rule, we noted diagnosis code F11.221 (Opioid dependence with intoxication delirium) had a C1 value of 4.00, however our analysis reflects that this diagnosis code was reported as a secondary diagnosis in only 42 claims, and only one claim reported F11.221 as a secondary diagnosis with no other secondary diagnosis or with all other secondary diagnoses that are NonCCs.
The C2 findings of the diagnosis codes that describe causally specified delirium range from a low of 0.28 to a high of 3.22 and the C3 findings range from a low of 1.25 to a high of 3.85. We stated that the data are clearly mixed between the C2 and C3 findings, and do not consistently support a change in the severity level. On average, the C2 and C3 findings again suggest that these codes that describe causally specified delirium are more similar to a NonCC.
As discussed in the proposed rule, in considering the nine guiding principles, as summarized previously, we note that delirium is a diagnosis that can impede patient cooperation or management of care or both. Delirium is a confusional state that can manifest as agitation, tremulousness, and hallucinations or even somnolence and decreased arousal. In addition, patients diagnosed with delirium can require a higher level of care by needing intensive monitoring, and a greater number of caregivers. Managing disruptive behavior, particularly agitation and combative behavior, is a challenging aspect in caring for patients diagnosed with delirium. Prevention and treatment of delirium can include avoiding factors known to cause or aggravate delirium; identifying and treating the underlying acute illness; and where appropriate using low-dose, short-acting pharmacologic agents.
In the proposed rule we stated that after considering the C1, C2, and C3 values of the 37 ICD-10-CM diagnosis codes that describe causally specified delirium and consideration of the nine guiding principles, we believe these 37 codes should not be designated as MCCs. While there is a lack of consistent claims data to support a severity level change from CCs to MCCs, we stated we recognize patients with delirium can utilize increased hospital resources and can be at a higher severity level. Therefore, we proposed to retain the severity designation of the 37 codes listed previously as CCs for FY 2025.
Comment: Some commenters agreed with CMS' proposal to retain the ( print page 69088) severity designation of the 37 ICD-10-CM diagnosis codes that describe causally specified delirium as CCs for FY 2025.
Response: We appreciate the commenters' support of our proposal.
Comment: Many other commenters disagreed with the proposal and urged CMS to change the designation of the 37 ICD-10-CM diagnosis codes that describe causally specified delirium to MCC for FY 2025. Commenters stated that delirium is a complex condition to manage and stated the diagnosis fully satisfies CMS' nine guiding principles for re-evaluating changes to severity levels. Many commenters noted that the terms “delirium” and “encephalopathy” are often used interchangeably and refer to a shared set of acute neurocognitive conditions that require additional resources to treat. Some commenters stated that all diagnoses of delirium imply an underlying acute encephalopathy, while others stated acute encephalopathy is another name for delirium. A commenter noted that ICD-10-CM diagnosis codes G92.8 (Other toxic encephalopathy), G92.9 (Unspecified toxic encephalopathy) and G93.41 (Metabolic encephalopathy) have a higher severity level designation even though, in their view, the diagnosis codes that describe causally specified delirium provide even greater specificity. The commenter further stated that designating the diagnosis codes that describe causally specified delirium as MCCs is the logical conclusion of understanding the integrated nature of delirium and acute encephalopathy and is justified by a robust body of scientific literature and clinical practice guidelines.
Some commenters stated that practitioners have been inclined to report the ICD-10-CM diagnosis codes that describe toxic or metabolic encephalopathy, that are designated as MCCs, rather than report diagnosis codes that describe delirium, which they state is the Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition, Text Revision (DSM-5-TR) preferred terminology to describe the syndrome of cognitive and behavioral changes that can occur in response to acute physical illness. Commenters stated that parity in the severity level designation of the codes that describe causally specified delirium with the severity level designation of the codes that describe acute encephalopathy is essential to enhancing awareness of the clinical and economic costs associated with managing delirium and will encourage widespread delirium prevention efforts. Another commenter stated that if parity is not achieved between the codes that describe causally specified delirium and codes that describe toxic or metabolic encephalopathy, clinicians will continue to favor reporting the relatively uninformative diagnoses of toxic or metabolic encephalopathy, thereby directing attention away from delirium guidelines and care pathways. Other commenters suggested that retaining the severity designation of delirium as a CC reinforces the stigma of mental health conditions, promotes the use of non-specific diagnoses that require no more than a cursory evaluation of mental status, directs clinicians away from the use of delirium clinical practice guidelines, stands against the broad consensus recommendation to use the term “delirium” across invested major medical specialty organizations, and discourages efforts to detect and manage delirium.
Several commenters suggested that the mathematical analysis of the FY 2023 MedPAR file provided in the proposed rule is confounded given that delirium is being preferentially coded as toxic or metabolic encephalopathy. A commenter noted that there is robust literature detailing the impact of delirium on care complexity and costs, readmissions, rates of functional decline, institutionalization, cognitive decline, subsequent dementia diagnosis, and mortality and stated that the evidence suggests that delirium is underdiagnosed or being classified as encephalopathy and is having an impact on the data available for analysis.
Another commenter stated that they believe the September 2023 update of the FY 2023 MedPAR file generally supports the request to change delirium from a CC to an MCC in their review of the analyses for each of the diagnosis codes identified by the requestor that describe causally specified delirium presented in the proposed rule. Specifically, the commenter stated that based on their review of the C1, C2, and C3 values presented for ICD-10-CM code F05 (Delirium due to known physiological condition), which are 1.68, 2.46, and 3.38, respectively, F05 appears to be performing very similarly to many other conditions that are currently designated as MCCs. The commenter further stated that based on their analysis, the weighted average of the C1, C2, and C3 values of the 37 diagnosis codes that describe causally specified delirium are 1.68, 2.47, 3.38, respectively. This commenter stated they also reviewed the updated impact on resource use files provided on the CMS website so that the public can review the mathematical data for the impact on resource use generated using claims from the FY 2023 MedPAR file and stated that many codes currently designated as MCCs have C values similar to the values for causally specified delirium and stated on this basis alone, the severity designation of codes that describe causally specified delirium deserves to be changed from a CC to an MCC. The commenter specifically referenced the mathematical data for the impact on resource use generated using claims from the FY 2023 MedPAR file for the following codes that are designated as MCCs in Version 41.1:

Response: We appreciate the commenters sharing their concerns regarding the severity level designations of the ICD-10-CM diagnosis codes that describe causally specified delirium and thank the commenters for their feedback. We reviewed the commenters' concerns and while we recognize patients with delirium can utilize increased hospital resources, we continue to believe there is a lack of consistent claims data to support a severity level change of these diagnosis codes from CCs to MCCs for FY 2025.
In response to the analysis of the impact on resource use files performed by the commenter, as stated in prior rulemaking ( 84 FR 42150 ), C1, C2, and C3 values are a measure of the ratio of average costs for patients with these conditions to the expected average cost across all cases. We have stated a value close to 1.0 in the C1 field would suggest that the code produces the same expected value as a NonCC diagnosis. That is, average costs for the case are similar to the expected average costs for that subset and the diagnosis is not expected to increase resource usage. A higher value in the C1 (or C2 and C3) field suggests more resource usage is associated with the diagnosis and an increased likelihood that it is more like a CC or major CC than a NonCC. Thus, a value close to 2.0 suggests the condition is more like a CC than a NonCC but not as significant in resource usage as an MCC. A value close to 3.0 suggests the condition is expected to consume resources more similar to an MCC than a CC or NonCC.
Accordingly, the C1, C2, and C3 values highlighted by the commenter for the diagnosis codes reflected in the previous table currently designated as MCCs generally suggests that the conditions actually are more like CCs rather than NonCCs or MCCs and suggests the severity designation of the diagnoses designated as MCCs should be changed to CCs. We will consider these codes as we continue our comprehensive CC/MCC analysis, using a combination of mathematical analysis of claims data and the application of nine guiding principles to determine the extent to which presence of each code as a secondary diagnosis results in increased hospital resource use and will provide more detail in future rulemaking.
In response to the commenters that suggested that delirium “fully satisfies CMS' nine guiding principles”, as stated earlier, the nine guiding principles are not intended to turn the analysis into a quantitative exercise, requiring that every diagnosis code satisfy each principle. As discussed in prior rulemaking and earlier in this section, our intended approach is first, CMS will use the guiding principles in making an initial clinical assessment of the appropriate severity level designation for each ICD-10-CM code as a secondary diagnosis. CMS will then use a mathematical analysis of claims data as discussed in the FY 2008 IPPS/LTCH PPS final rule ( 72 FR 47159 ) to determine if the presence of the ICD-10-CM code as a secondary diagnosis appears to, or does not appear to, increase hospital resource consumption. There may be instances in which we would decide that the clinical analysis weighs in favor of proposing to maintain or proposing to change the severity designation of an ICD-10-CM code after application of the nine guiding principles. The nine guiding principles are intended to provide a framework for assessing relevant clinical factors to help denote if, and to what degree, additional resources are required above and beyond those that are already being utilized to address the principal diagnosis or other secondary diagnoses that might also be present on the claim.
In response to the suggestion that clinicians favor reporting encephalopathy as opposed to delirium, we note that providers are responsible for ensuring that they are documenting as specifically and accurately as possible for the conditions they are treating and the services they render to correctly reflect the severity of illness and capture how truly sick a patient is when causally specified delirium or encephalopathy are present. In addition, as we noted in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38012 ), coding advice is issued independently from payment policy. We also note that, historically, we have not provided coding advice in rulemaking with respect to policy ( 82 FR 38045 ). As one of the Cooperating Parties for ICD-10, we collaborate with the American Hospital Association (AHA) through the Coding Clinic for ICD-10-CM and ICD-10-PCS to promote proper coding. We recommend that an entity seeking coding guidance on reporting causally specified delirium or encephalopathy submit any questions pertaining to correct coding to the AHA.
We consulted with the staff at the Centers for Disease Control's (CDC's) National Center for Health Statistics (NCHS), because NCHS has the lead responsibility for maintaining the ICD-10-CM diagnosis codes. The NCHS' staff acknowledged the terms delirium and encephalopathy are differentiated in the classification, such that coding would usually depend on the specific terms used in the medical record documentation. NCHS confirmed that they would consider further review of the classification, including review of the Excludes notes, for these two diagnoses. As such, we believe it would be appropriate to maintain the current severity level designations of the ICD-10-CM diagnosis codes that describe causally specified delirium at this time in order to further examine the relevant clinical factors and possible similarities in resource consumption in order to best represent this subset of patients within the MS-DRG classification.
Therefore, after consideration of the public comments we received, and for the reasons discussed, we are finalizing our proposal, without modification, to maintain the current severity level designation of the 37 ICD-10-CM diagnosis codes that describe causally specified delirium listed previously as CCs for FY 2025.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36001 ), we noted the following tables identify the proposed additions and deletions to the diagnosis code MCC severity levels list and the proposed additions and deletions to the diagnosis code CC severity levels list for FY 2025 and are available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html
Table 6I.1—Proposed Additions to the MCC List—FY 2025;
Table 6J.1—Proposed Additions to the CC List—FY 2025; and
Table 6J.2—Proposed Deletions to the CC List—FY 2025
Comment: Commenters agreed with the proposed additions and deletions to the MCC and CC lists as shown in tables 6I.1, 6J.1, and 6J.2 associated with the proposed rule.
The following tables associated with this final rule reflect the finalized severity levels under Version 42 of the ICD-10 MS-DRGs for FY 2025 and are available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS ; Table 6I.—Complete MCC List—FY 2025; Table 6I.1—Additions to the MCC List—FY 2025; Table 6I.2—Deletions to the MCC List—FY 2025; Table 6J.—Complete CC List—FY 2025; Table 6J.1—Additions to the CC List—FY 2025; and Table 6J.2—Deletions to the CC List—FY 2025. ( print page 69090)
In the September 1, 1987 final notice ( 52 FR 33143 ) concerning changes to the DRG classification system, we modified the GROUPER logic so that certain diagnoses included on the standard list of CCs would not be considered valid CCs in combination with a particular principal diagnosis. We created the CC Exclusions List for the following reasons: (1) to preclude coding of CCs for closely related conditions; (2) to preclude duplicative or inconsistent coding from being treated as CCs; and (3) to ensure that cases are appropriately classified between the complicated and uncomplicated DRGs in a pair.
In the May 19, 1987 proposed notice ( 52 FR 18877 ) and the September 1, 1987 final notice ( 52 FR 33154 ), we explained that the excluded secondary diagnoses were established using the following five principles:
- Chronic and acute manifestations of the same condition should not be considered CCs for one another;
- Specific and nonspecific (that is, not otherwise specified (NOS)) diagnosis codes for the same condition should not be considered CCs for one another;
- Codes for the same condition that cannot coexist, such as partial/total, unilateral/bilateral, obstructed/unobstructed, and benign/malignant, should not be considered CCs for one another;
- Codes for the same condition in anatomically proximal sites should not be considered CCs for one another; and
- Closely related conditions should not be considered CCs for one another.
The creation of the CC Exclusions List was a major project involving hundreds of codes. We have continued to review the remaining CCs to identify additional exclusions and to remove diagnoses from the master list that have been shown not to meet the definition of a CC. We refer readers to the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50541 through 50544 ) for detailed information regarding revisions that were made to the CC and CC Exclusion Lists under the ICD-9-CM MS-DRGs.
The ICD-10 MS-DRGs Version 41.1 CC Exclusion List is included as Appendix C in the ICD-10 MS-DRG Definitions Manual (available in two formats; text and HTML). The manuals are available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html ) and includes two lists identified as Part 1 and Part 2. Part 1 is the list of all diagnosis codes that are defined as a CC or MCC when reported as a secondary diagnosis. For all diagnosis codes on the list, a link is provided to a collection of diagnosis codes which, when reported as the principal diagnosis, would cause the CC or MCC diagnosis to be considered as a NonCC. Part 2 is the list of diagnosis codes designated as an MCC only for patients discharged alive; otherwise, they are assigned as a NonCC.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36002 through 36006 ), effective for the April 1, 2024, release of the ICD-10 MS-DRG Definitions Manual, Version 41.1, a new section has been added to Appendix C as follows:
Part 3 lists diagnosis codes that are designated as a complication or comorbidity (CC) or major complication or comorbidity (MCC) and included in the definition of the logic for the listed MS-DRGs. When reported as a secondary diagnosis and grouped to one of the listed MS-DRGs, the diagnosis is excluded from acting as a CC/MCC for severity in DRG assignment.
The purpose of this new section is to include the list of MS-DRGs subject to what is referred to as suppression logic. In addition to the suppression logic excluding secondary diagnosis CC or MCC conditions that may be included in the definition of the logic for a DRG, it is also based on the presence of other secondary diagnosis logic defined within certain base DRGs. Therefore, if a MS-DRG has secondary diagnosis logic, the suppression is activated regardless of the severity of the secondary diagnosis code(s) for appropriate grouping and MS-DRG assignment.
In the proposed rule we noted that each MS-DRG is defined by a particular set of patient attributes including principal diagnosis, specific secondary diagnoses, procedures, sex, and discharge status. The patient attributes which define each MS-DRG are displayed in a series of headings which indicate the patient characteristics used to define the MS-DRG. These headings indicate how the patient's diagnoses and procedures are used in determining MS-DRG assignment. Following each heading is a complete list of all the ICD-10-CM diagnosis or ICD-10-PCS procedure codes included in the MS-DRG. One of these headings is secondary diagnosis.
- Secondary diagnosis. Indicates that a specific set of secondary diagnoses are used in the definition of the MS-DRG. For example, a secondary diagnosis of acute leukemia with chemotherapy is used to define MS-DRG 839.
The full list of MS-DRGs where suppression occurs is shown in the following table.
In the proposed rule we stated we believe this additional information about the suppression logic may further assist users of the ICD-10 MS-DRG GROUPER software and related materials.
As noted in the proposed rule, during our review of the MS-DRGs containing secondary diagnosis logic in association with the suppression logic previously discussed, we identified another set of MS-DRGs containing secondary diagnosis logic in the definition of the MS-DRG. Specifically, we identified MS-DRGs 673, 674, and 675 (Other Kidney and Urinary Tract Procedures with MCC, with CC, and without CC/MCC, respectively) in MDC 11 (Diseases and Disorders of the Kidney and Urinary Tract), as displayed in the ICD-10 MS-DRG Version 41.1 Definitions Manual (which is available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) which contains secondary diagnosis logic.
As stated in the proposed rule, of the seven logic lists included in the definition of MS-DRGs 673, 674, and 675, there are three “Or Principal Diagnosis” logic lists and one “With ( print page 69091) Secondary Diagnosis” logic list. The first “Or Principal Diagnosis” logic list is comprised of 21 diagnosis codes describing conditions such as chronic kidney disease, kidney failure, and complications related to a vascular dialysis catheter or kidney transplant. The second “Or Principal Diagnosis” logic list is comprised of four diagnosis codes describing diabetes with diabetic chronic kidney disease followed by a “With Secondary Diagnosis” logic list that includes diagnosis codes N18.5 (Chronic kidney disease, stage 5) and N18.6 (End stage renal disease). These logic lists are components of the special logic in MS-DRGs 673, 674, and 675 for certain MDC 11 diagnoses reported with procedure codes for the insertion of tunneled or totally implantable vascular access devices. The third “Or Principal Diagnosis” logic list is comprised of three diagnosis codes describing Type 1 diabetes with different kidney complications as part of the special logic in MS-DRGs 673, 674, and 675 for pancreatic islet cell transplantation performed in the absence of any other surgical procedure.
Under the Version 41.1 ICD-10 MS-DRGs, diagnosis code N18.5 (Chronic kidney disease, stage 5) is currently designated as a CC and diagnosis code N18.6 (End stage renal disease) is designated as an MCC. As discussed in the proposed rule, in our review of the MS-DRGs containing secondary diagnosis logic in association with the suppression logic, we noted that currently, when some diagnosis codes from the “Or Principal Diagnosis” logic lists in MS-DRGs 673, 674, and 675 are reported as the principal diagnosis and either diagnosis code N18.5 or N18.6 from the “With Secondary Diagnosis” logic list is reported as a secondary diagnosis, some cases are grouping to MS-DRG 673 (Other Kidney and Urinary Tract Procedures with MCC) or to MS-DRG 674 (Other Kidney and Urinary Tract Procedures with CC) in the absence of any other MCC or CC secondary diagnoses being reported.
In our analysis of this issue as discussed in the proposed rule, we noted diagnosis codes N18.5 and N18.6 are excluded from acting as a CC or MCC, when reported with principal diagnoses from Principal Diagnosis Collection Lists 1379 and 1380, respectively, as reflected in Part 1 of Appendix C in the CC Exclusion List. We refer the reader to Part 1 of Appendix C in the CC Exclusion List as displayed in the ICD-10 MS-DRG Version 41.1 Definitions Manual (which is available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software ) for the complete list of principal diagnoses in Principal Diagnosis Collection Lists 1379 and 1380. Specifically, when codes N18.5 or N18.6 are reported as secondary diagnoses, we noted they are considered as NonCCs when the diagnosis codes from the “Or Principal Diagnosis” logic lists in MS-DRGs 673, 674, and 675 reflected in the following table are reported as the principal diagnosis under the CC Exclusion logic.

In the proposed rule, we also noted that currently, a subset of diagnosis codes from the first “Or Principal Diagnosis” logic list in MS-DRGs 673, 674, and 675 are not listed in Principal Diagnosis Collection Lists 1379 or 1380 for diagnosis codes N18.5 and N18.6, respectively. As a result, when one of the 13 diagnosis codes listed in the following table are reported as the principal diagnosis, and either diagnosis code N18.5 or N18.6 from the “With Secondary Diagnosis” logic list are reported as a secondary diagnosis, the cases are grouping to MS-DRG 673 (Other Kidney and Urinary Tract Procedures with MCC) or to MS-DRG 674 (Other Kidney and Urinary Tract Procedures with CC) when also reported with a procedure code describing the ( print page 69092) insertion of a tunneled or totally implantable vascular access device.

We noted in the proposed rule that consistent with how other similar logic lists function in the ICD-10 GROUPER software for case assignment to the “with MCC” or “with CC” MS-DRGs, the logic for case assignment to MS-DRG 673 is intended to require any other diagnosis designated as an MCC and reported as a secondary diagnosis for appropriate assignment, and not the diagnoses currently listed in the logic for the definition of the MS-DRG. Likewise, the logic for case assignment to MS-DRG 674 is intended to require any other diagnosis designated as a CC and reported as a secondary diagnosis for appropriate assignment.
Therefore, for FY 2025, we proposed to correct the logic for case assignment to MS-DRGs 673, 674, and 675 by adding suppression logic to exclude diagnosis codes N18.5 (Chronic kidney disease, stage 5) and N18.6 (End stage renal disease) from the logic list entitled “With Secondary Diagnosis” from acting as a CC or an MCC, respectively, when reported as a secondary diagnosis with one of the 13 previously listed principal diagnosis codes from the “Or Principal Diagnosis” logic lists in MS-DRGs 673, 674, and 675 for appropriate grouping and MS-DRG assignment. Under this proposal, when diagnosis codes N18.5 or N18.6 are reported as a secondary diagnosis with one of the 13 previously listed principal diagnosis codes, the GROUPER will assign MS-DRG 675 (Other Kidney and Urinary Tract Procedures without CC/MCC) in the absence of any other MCC or CC secondary diagnoses being reported. In the proposed rule we also noted that the current list of MS-DRGs subject to suppression logic as previously discussed and listed under Version 41.1 includes MS-DRGs that are not subdivided by a two-way severity level split (“with MCC and without MCC” or “with CC/MCC and without CC/MCC”) or a three-way severity level split (with MCC, with CC, and without CC/MCC, respectively), or the listed MS-DRG includes diagnoses that are not currently designated as a CC or MCC. To avoid potential confusion, we proposed to refine how the suppression logic is displayed under Appendix C—Part 3 to not display the MS-DRGs where the suppression logic has no impact on the grouping (meaning the logic list for the affected MS-DRG contains diagnoses that are all designated as NonCCs, or the MS-DRG is not subdivided by a severity level split) as reflected in the draft Version 42 ICD-10 MS-DRG Definitions Manual, which is available in association with the proposed rule at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps .
Comment: Commenters stated they did not agree with the proposed application of the suppression logic within MS-DRGs 673, 674, and 675 when diagnosis codes N18.5 and N18.6 are reported as a secondary diagnosis in conjunction with one of the principal diagnosis codes listed in Part 1 of Appendix C in the CC Exclusion List. The commenters stated that ICD-10-CM codes N18.5 and N18.6 are the highest level of severity for kidney failure with end stage renal disease and stage 5, both of which require dialysis and/or kidney transplant. According to the commenters, the only principal diagnoses that could meet one of the five principles would be I12.0 (Hypertensive chronic kidney disease with stage 5 chronic kidney disease or end stage renal disease) or I13.11 (Hypertensive heart and chronic kidney disease without heart failure, with stage 5 chronic kidney disease or end-stage renal disease) as these two codes actually indicate stage 5 chronic kidney disease or end stage renal disease in the narrative description. The commenters stated their belief that the five conditions established for exclusions were not met for the majority of the diagnoses on the principal diagnosis list ( print page 69093) and for that reason should not be subject to suppression logic.
Response: We wish to clarify for the commenters that the suppression logic is not the same as the CC Exclusion List logic under Part 1 of Appendix C—CC Exclusion List in the ICD-10 MS-DRG Definitions Manual. As previously described, Part 1 of Appendix C is the list of all diagnosis codes that are defined as a CC or MCC when reported as a secondary diagnosis. For all diagnosis codes on the list, a link is provided to a collection of diagnosis codes which, when reported as the principal diagnosis, would cause the CC or MCC diagnosis to be considered as a NonCC. Separate from the CC Exclusion List logic, effective for the April 1, 2024, release of the ICD-10 MS-DRG Definitions Manual, Version 41.1, a new section was added to Appendix C for the suppression logic as listed under Part 3 of Appendix C. As previously described, Part 3 lists diagnosis codes that are designated as a CC or MCC and are included in the definition of the logic for the listed MS-DRGs. As such, when reported as a secondary diagnosis, the diagnosis is intended to be excluded from acting as a CC or MCC for severity in DRG assignment. We stated in the proposed rule that, because the logic for case assignment to MS-DRGs 673, 674, and 675 includes diagnosis codes N18.5 and N18.6 in the definition of the “With Secondary Diagnosis” logic list, we were proposing to correct the logic for appropriate grouping, consistent with other secondary diagnosis logic. Therefore, when diagnosis codes N18.5 or N18.6 are reported as a secondary diagnosis with one of the 13 previously listed principal diagnosis codes from the “Or Principal Diagnosis” logic lists in MS-DRGs 673, 674, and 675, for appropriate grouping and consistency they should be excluded from acting as a CC or MCC.
We note that, because the commenters raised concerns regarding the principal diagnoses listed under Part 1 of Appendix C—CC Exclusions List in Principal Diagnosis Collection Lists 1378 and 1379 that currently exclude diagnosis codes N18.5 and N18.6 from acting as a CC or MCC under the CC exclusion logic in accordance with the list of five principles established in 1987, we intend to perform a broad review of the conditions in these lists to determine if any modifications are warranted and to ensure they continue to be clinically appropriate. To inform future rulemaking, feedback and other suggestions may be submitted by October 20, 2024, and directed to MEARIS TM at: https://mearis.cms.gov/public/home .
After consideration of the public comments we received, and for the reasons discussed, we are finalizing our proposal to add suppression logic to exclude diagnosis codes N18.5 (Chronic kidney disease, stage 5) and N18.6 (End stage renal disease) from the logic list entitled “With Secondary Diagnosis” from acting as a CC or an MCC, respectively, when reported as a secondary diagnosis with one of the 13 previously listed principal diagnosis codes from the “Or Principal Diagnosis” logic lists in MS-DRGs 673, 674, and 675, without modification, effective October 1, 2024 for FY 2025.
We also note that during our review of the 37 diagnosis codes that describe causally specified delirium as discussed in section II.C.12.c.2. of the preamble of this final rule, we identified diagnosis code F05 (Delirium due to known physiological condition) as a condition that is listed on a subset of the Principal Diagnosis Collection Lists under Part 1 of Appendix C—CC Exclusions List. Specifically, we found diagnosis code F05 listed on Principal Diagnosis Collection List numbers 642, 643, 645, 646, and 647. Diagnosis code F05 is listed on the Unacceptable Principal Diagnosis Code edit code list in the Medicare Code Editor and is not appropriate to report as a principal diagnosis according to the ICD-10-CM Tabular List of Diseases and Injuries instructional note to “Code first the underlying physiological condition, such as: dementia (F03.9-)”. Consistent with the MCE Unacceptable Principal Diagnosis Code edit code list and the instructional note in the ICD-10-CM Tabular List of Diseases and Injuries, we are removing diagnosis code F05 from the previously listed Principal Diagnosis Collection Lists effective October 1, 2024, for FY 2025.
Lastly, we are finalizing our proposal to refine how the suppression logic is displayed under Appendix C—Part 3, without modification, effective October 1, 2024, for FY 2025. Under this finalization, MS-DRGs where the suppression logic has no impact on the grouping (meaning the logic list for the affected MS-DRG contains diagnoses that are all designated as NonCCs, or the MS-DRG is not subdivided by a severity level split) will not be displayed in Appendix C—Part 3 as reflected in the Version 42 ICD-10 MS-DRG Definitions Manual, which is available in association with this final rule at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps .
In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed additional changes to the ICD-10 MS-DRGs Version 42 CC Exclusion List based on the diagnosis code updates as discussed in section II.C.12. of the proposed rule and set forth in Tables 6G.1, 6G.2, 6H.1, and 6H.2 associated with the proposed rule and available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS .
We did not receive any public comments opposing the proposed CC Exclusions List, however, during our internal review of the proposed CC Exclusions List we identified some inconsistencies with the 77 new Hodgkin lymphoma diagnosis codes that were proposed to be designated as a CC (based on the predecessor code designation and now finalized as reflected in Table 6A.- New Diagnosis Codes—FY 2025 associated with this final rule). We determined that clinically, all 77 Hodgkin lymphoma diagnosis codes should be excluded from acting as a CC when another Hodgkin lymphoma diagnosis code is reported as the principal diagnosis. Therefore, for FY 2025, we are finalizing, with modification, the CC exclusions for the 77 Hodgkin lymphoma codes after internal review as reflected in Tables 6G.1 and 6G.2 in association with this final rule.
The finalized CC Exclusions List as displayed in Tables 6G.1, 6G.2, 6H.1, 6H.2, and 6K, associated with this final rule reflect the severity levels under V42 of the ICD-10 MS-DRGs. We have developed Table 6G.1.—Secondary Diagnosis Order Additions to the CC Exclusions List—FY 2025; Table 6G.2.—Principal Diagnosis Order Additions to the CC Exclusions List—FY 2025; Table 6H.1.—Secondary Diagnosis Order Deletions to the CC Exclusions List—FY 2025; and Table 6H.2.—Principal Diagnosis Order Deletions to the CC Exclusions List—FY 2025; and Table 6K. Complete List of CC Exclusions-FY 2025.
For Table 6G.1, each secondary diagnosis code finalized for addition to the CC Exclusion List is shown with an asterisk and the principal diagnoses finalized to exclude the secondary diagnosis code are provided in the indented column immediately following it. For Table 6G.2, each of the principal diagnosis codes for which there is a CC exclusion is shown with an asterisk and the conditions finalized for addition to the CC Exclusion List that will not count as a CC are provided in an indented column immediately following the affected principal diagnosis. For Table 6H.1, each secondary diagnosis code finalized for deletion from the CC Exclusion List is shown with an asterisk followed by the principal diagnosis ( print page 69094) codes that currently exclude it. For Table 6H.2, each of the principal diagnosis codes is shown with an asterisk and the finalized deletions to the CC Exclusions List are provided in an indented column immediately following the affected principal diagnosis. Tables 6G.1., 6G.2., 6H.1., and 6H.2. associated with this final rule are available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html .
To identify new, revised, and deleted diagnosis and procedure codes, for FY 2025, we have developed Table 6A.—New Diagnosis Codes, Table 6B.—New Procedure Codes, Table 6C.—Invalid Diagnosis Codes, Table 6D.—Invalid Procedure Codes, Table 6E.—Revised Diagnosis Code Titles, and Table 6F.—Revised Procedure Code Titles for this final rule.
These tables are not published in the Addendum to the proposed rule or final rule, but are available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html as described in section VI. of the Addendum to this final rule. As discussed in section II.C.15. of the preamble of the proposed rule and this final rule, the code titles are adopted as part of the ICD-10 (previously ICD-9-CM) Coordination and Maintenance Committee meeting process. Therefore, although we publish the code titles in the IPPS proposed and final rules, they are not subject to comment in the proposed or final rules.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36006 ), we proposed the MDC and MS-DRG assignments for the new diagnosis codes and procedure codes as set forth in Table 6A.—New Diagnosis Codes and Table 6B.—New Procedure Codes. We also stated that the proposed severity level designations for the new diagnosis codes are set forth in Table 6A. and the proposed O.R. status for the new procedure codes are set forth in Table 6B. Consistent with our established process, we examined the MS-DRG assignment and the attributes (severity level and O.R. status) of the predecessor diagnosis or procedure code, as applicable, to inform our proposed assignments and designations.
Specifically, we reviewed the predecessor code and MS-DRG assignment most closely associated with the new diagnosis or procedure code, and in the absence of claims data, we considered other factors that may be relevant to the MS-DRG assignment, including the severity of illness, treatment difficulty, complexity of service and the resources utilized in the diagnosis and/or treatment of the condition. We noted that this process does not automatically result in the new diagnosis or procedure code being proposed for assignment to the same MS-DRG or to have the same designation as the predecessor code.
In this FY 2025 IPPS/LTCH PPS final rule, we present a summation of the comments we received in response to the proposed assignments, our responses to those comments, and our finalized policies.
Comment: Commenters expressed support for the finalization of three new ICD-10-CM diagnosis codes describing presymptomatic Type 1 diabetes mellitus by stage and three new codes describing hypoglycemia by level, as shown in the following table and reflected in Table 6A.—New Diagnosis Codes—FY 2025 in association with the proposed rule and available at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps .

The commenters stated these new diagnosis codes are intended to facilitate standardized diabetes and hypoglycemia reporting and enable consistent quantification, tracking, and outcomes measurement. According to the commenters, the more granular presymptomatic diabetes diagnosis codes will help identify early disease progression and support appropriate intervention, including documentation of an individual's need for a continuous glucose monitor (CGM). The commenters urged CMS to incorporate these finalized diagnosis codes throughout Medicare payment and coverage policies.
Comment: Commenters expressed support for the proposed CC status designation and proposed MS-DRG assignments under MDC 17 and MDC 25 for the diagnosis codes describing lymphoma in remission as reflected in Table 6A.—New Diagnosis Codes—FY 2025 in association with the proposed rule and available at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps . The commenters stated that patients with these diagnoses are generally more complex and resource-intensive, warranting the CC designation. The commenters requested that we finalize the proposed designation and MS-DRG assignments for FY 2025.
Comment: A couple of commenters requested that CMS designate the following 16 new procedure codes that describe introduction of the AGENT TM Paclitaxel-Coated Balloon Catheter that is indicated to treat coronary in-stent restenosis (ISR) in patients with coronary artery disease as operating room (O.R.) procedures, with assignment to surgical MS-DRGs.

Specifically, the commenters requested assignment of the previously listed procedure codes to the following surgical MS-DRGs:
- MS-DRG 250 Percutaneous Cardiovascular Procedures without Intraluminal Device with MCC
- MS-DRG 251 Percutaneous Cardiovascular Procedures without Intraluminal Device without MCC
- MS-DRG 321 Percutaneous Cardiovascular Procedures with Intraluminal Device with MCC or 4+ Arteries/Intraluminal Devices
- MS-DRG 322 Percutaneous Cardiovascular Procedures with Intraluminal Device without MCC
- MS-DRG 323 Coronary Intravascular Lithotripsy with Intraluminal Device with MCC
- MS-DRG 324 Coronary Intravascular Lithotripsy with Intraluminal Device without MCC
- MS-DRG 325 Coronary Intravascular Lithotripsy without Intraluminal Device
The commenters stated that based on the usual surgical hierarchy rules, the reporting of one of the vessel preparation steps (that is, angioplasty, atherectomy, or lithotripsy), or placement of a new stent in connection with the reported use of the AGENT TM Paclitaxel-Coated Balloon Catheter would mean the procedure would map to one of the previously listed surgical MS-DRGs. The commenters also stated their belief that designating the new procedure codes as O.R. procedures with assignment to the previously listed MS-DRGs would reflect the surgical nature and complexity of the procedure and would be appropriate for the time being.
Response: The 16 new procedure codes describing use of the AGENT TM Paclitaxel-Coated Balloon Catheter were finalized following the March 19, 2024, ICD-10 Coordination and Maintenance Committee meeting and made available via the CMS website on June 5, 2025, at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps . The procedure codes are also reflected in Table 6B—New Procedure Codes—FY 2025 associated with this final rule.
Under our established process, we reviewed the predecessor code and MS-DRG assignment most closely associated with the new procedure codes. We note that because the procedure codes describing the use of an AGENT TM Paclitaxel-Coated Balloon Catheter are describing delivery of the paclitaxel to the coronary vessel(s), the predecessor code is 3E073GC (Introduction of other therapeutic substance into coronary artery, percutaneous approach), which is designated as a non-O.R. procedure and does not affect MS-DRG assignment. As discussed at the March 19, 2024, ICD-10 Coordination and Maintenance Committee meeting and in the commenters' feedback, a preparatory step (that is, vessel preparation by either angioplasty, atherectomy, or lithotripsy) is required to be performed first, before ( print page 69096) the AGENT TM Paclitaxel-Coated Balloon Catheter is deployed. We note that each type of vessel preparation procedure is designated as an O.R. procedure and maps to one of the previously listed surgical MS-DRGs. We also note that the commenters are correct that based on the surgical hierarchy, the reporting of one of the vessel preparation steps (that is, angioplasty, atherectomy, or lithotripsy), or placement of a new stent in connection with the use of the AGENT TM Paclitaxel-Coated Balloon Catheter would result in assignment to one of the previously listed surgical MS-DRGs. We note that use of the AGENT TM Paclitaxel-Coated Balloon Catheter to deliver the paclitaxel to the coronary vessel(s) cannot occur in the absence of a surgical vessel preparation and therefore, it is the vessel preparation procedure that will determine the surgical MS-DRG assignment to one of the previously listed surgical MS-DRGs. As such, we do not agree with designating the 16 new procedure codes as O.R. procedure codes since the resulting MS-DRG assignment is dependent on the surgical vessel preparation procedure that would be reported when the AGENT TM Paclitaxel-Coated Balloon Catheter is used to deliver the paclitaxel to the coronary vessel(s) and result in assignment to one of the previously listed surgical MS-DRGs regardless. We refer the reader to the ICD-10 MS-DRG Definitions Manual, Version 42 available in association with this final rule on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps for complete documentation of the GROUPER logic for the previously listed surgical MS-DRGs under MDC 05. Accordingly, consistent with our established process and for the reasons discussed, we are designating the 16 new procedure codes describing use of the AGENT TM Paclitaxel-Coated Balloon Catheter as non-O.R. for FY 2025.
Comment: A commenter expressed its appreciation to the ICD-10 Coordination and Maintenance Committee for creating and implementing new ICD-10-CM Z codes to describe Duffy null status. The commenter stated that the new codes were requested to ensure that people who have lower absolute neutrophil count (ANC) due to Duffy phenotype are accurately documented within the medical record and are not considered to have “abnormal” ANC levels.
The commenter indicated that the new codes will be critical for proper payment, accurate documentation, appropriate clinical care and management, and the ability to conduct research. The commenter also indicated that accurate documentation of the Duffy status will decrease duplicative testing and allow for more precise medication administration, consistent with need.
Response: We thank the commenter for its support.
Comment: S ome commenters suggested that ICD-10-PCS procedure code 02583ZF (Destruction of conduction mechanism using irreversible electroporation, percutaneous approach) also be added to proposed new MS-DRG 317 (Concomitant Left Atrial Appendage Closure and Cardiac Ablation) in MDC 05. A couple commenters stated that pulsed field ablation is becoming the standard of care for atrial fibrillation ablation, and it should be included in the proposed new MS-DRG if patients who have atrial fibrillation are to be effectively, safely, and efficiently managed.
Response: We appreciate the commenters' feedback. As discussed in section II.C.4.a. of the preamble of this final rule, we are finalizing MS-DRG 317 for FY 2025. Upon review, we believe it is appropriate to add procedure code 02583ZF to the logic for case assignment to MS-DRG 317 as the description of the code describes a type of cardiac ablation and is clinically coherent with the other procedure codes describing cardiac ablation that were proposed and finalized for assignment to MS-DRG 317 effective for FY 2025. We are therefore, finalizing, with modification, the MS-DRG assignments for procedure code 02583ZF as reflected in Table 6B.—New Procedure Codes in association with this final rule.
After consideration of the public comments received, we are finalizing the MDC and MS-DRG assignments for the new diagnosis codes and procedure codes as set forth in Table 6A.—New Diagnosis Codes and Table 6B.—New Procedure Codes associated with this final rule. In addition, the finalized severity level designations for the new diagnosis codes are set forth in Table 6A. and the finalized O.R. status for the new procedure codes are set forth in Table 6B associated with this final rule.
In association with this final rule, we are making the following tables available on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html :
- Table 6A.—New Diagnosis Codes—FY 2025;
- Table 6B.—New Procedure Codes—FY 2025;
- Table 6C.—Invalid Diagnosis Codes—FY 2025;
- Table 6D.—Invalid Procedure Codes—FY 2025;
- Table 6E.—Revised Diagnosis Code Titles—FY 2025;
- Table 6F.—Revised Procedure Code Titles—FY 2025;
- Table 6G.1.—Secondary Diagnosis Order Additions to the CC Exclusions List—FY 2025;
- Table 6G.2.—Principal Diagnosis Order Additions to the CC Exclusions List—FY 2025;
- Table 6H.1.—Secondary Diagnosis Order Deletions to the CC Exclusions List—FY 2025;
- Table 6H.2.—Principal Diagnosis Order Deletions to the CC Exclusions List—FY 2025;
- Table 6I.—Complete MCC List—FY 2025;
- Table 6I.1.—Additions to the MCC List—FY 2025;
- Table 6J.1.—Complete CC List—FY 2025;
- Table 6J.1.—Additions to the CC List—FY 2025;
- Table 6J.2.—Deletions to the CC List—FY 2025; and
- Table 6K.—Complete List of CC Exclusions—FY 2025.
Some inpatient stays entail multiple surgical procedures, each one of which, occurring by itself, could result in assignment of the case to a different MS-DRG within the MDC to which the principal diagnosis is assigned. Therefore, it is necessary to have a decision rule within the GROUPER by which these cases are assigned to a single MS-DRG. The surgical hierarchy, an ordering of surgical classes from most resource-intensive to least resource-intensive, performs that function. Application of this hierarchy ensures that cases involving multiple surgical procedures are assigned to the MS-DRG associated with the most resource-intensive surgical class.
A surgical class can be composed of one or more MS-DRGs. For example, in MDC 11, the surgical class “kidney transplant” consists of a single MS-DRG (MS-DRG 652) and the class “major bladder procedures” consists of three MS-DRGs (MS-DRGs 653, 654, and 655).
Consequently, in many cases, the surgical hierarchy has an impact on more than one MS-DRG. The methodology for determining the most resource-intensive surgical class involves weighting the average resources for each MS-DRG by frequency to determine the weighted average resources for each surgical class. ( print page 69097) For example, assume surgical class A includes MS-DRGs 001 and 002 and surgical class B includes MS-DRGs 003, 004, and 005. Assume also that the average costs of MS-DRG 001 are higher than that of MS-DRG 003, but the average costs of MS-DRGs 004 and 005 are higher than the average costs of MS-DRG 002. To determine whether surgical class A should be higher or lower than surgical class B in the surgical hierarchy, we would weigh the average costs of each MS-DRG in the class by frequency (that is, by the number of cases in the MS-DRG) to determine average resource consumption for the surgical class. The surgical classes would then be ordered from the class with the highest average resource utilization to that with the lowest, with the exception of “other O.R. procedures” as discussed in this final rule.
This methodology may occasionally result in assignment of a case involving multiple procedures to the lower-weighted MS-DRG (in the highest, most resource-intensive surgical class) of the available alternatives. However, given that the logic underlying the surgical hierarchy provides that the GROUPER search for the procedure in the most resource-intensive surgical class, in cases involving multiple procedures, this result is sometimes unavoidable.
We note that, notwithstanding the foregoing discussion, there are a few instances when a surgical class with a lower average cost is ordered above a surgical class with a higher average cost. For example, the “other O.R. procedures” surgical class is uniformly ordered last in the surgical hierarchy of each MDC in which it occurs, regardless of the fact that the average costs for the MS-DRG or MS-DRGs in that surgical class may be higher than those for other surgical classes in the MDC. The “other O.R. procedures” class is a group of procedures that are only infrequently related to the diagnoses in the MDC but are still occasionally performed on patients with cases assigned to the MDC with these diagnoses. Therefore, assignment to these surgical classes should only occur if no other surgical class more closely related to the diagnoses in the MDC is appropriate.
A second example occurs when the difference between the average costs for two surgical classes is very small. We have found that small differences generally do not warrant reordering of the hierarchy because, as a result of reassigning cases on the basis of the hierarchy change, the average costs are likely to shift such that the higher-ordered surgical class has lower average costs than the class ordered below it.
Based on the changes that we proposed to make for FY 2025, as discussed in section II.C. of the preamble of the proposed rule and this final rule, we proposed to modify the existing surgical hierarchy for FY 2025 as follows.
As discussed in section II.C.4.a. of the preamble of the proposed rule and this final rule, we proposed to revise the surgical hierarchy for the MDC 05 (Diseases and Disorders of the Circulatory System) MS-DRGs as follows: In the MDC 05 MS-DRGs, we proposed to sequence proposed new MS-DRG 317 (Concomitant Left Atrial Appendage Closure and Cardiac Ablation) above MS-DRG 275 (Cardiac Defibrillator Implant with Cardiac Catheterization and MCC) and below MS-DRGs 231, 232, 233, 234, 235, and 236 (Coronary Bypass with or without PTCA, with or without Cardiac Catheterization or Open Ablation, with and without MCC, respectively). As discussed in section II.C.4.b. of the preamble of the proposed rule and this final rule, we proposed to revise the title for MS-DRG 276 from “Cardiac Defibrillator Implant with MCC” to “Cardiac Defibrillator Implant with MCC or Carotid Sinus Neurostimulator”.
As discussed in section II.C.6.b. of the preamble of the proposed rule and this final rule, we proposed to delete MS-DRGs 453, 454, and 455 (Combined Anterior and Posterior Spinal Fusion with MCC, with CC, and without CC/MCC, respectively). Based on the changes we proposed to make for those MS-DRGs in MDC 08 (Diseases and Disorders of the Musculoskeletal System and Connective Tissue), we proposed to revise the surgical hierarchy for MDC 08 as follows: In MDC 08, we proposed to sequence proposed new MS-DRGs 426, 427, and 428 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with MCC, with CC, and without CC/MCC, respectively) above proposed new MS-DRG 402 (Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical). We proposed to sequence proposed new MS-DRGs 429 and 430 (Combined Anterior and Posterior Cervical Spinal Fusion with MCC and without MCC, respectively) above MS-DRGs 456, 457, and 458 (Spinal Fusion Except Cervical with Spinal Curvature, Malignancy, Infection or Extensive Fusions with MCC, with CC, and without CC/MCC, respectively) and below proposed new MS-DRG 402. We proposed to sequence proposed new MS-DRGs 447 and 448 (Multiple Level Spinal Fusion Except Cervical with MCC and without MCC, respectively) above proposed revised MS-DRGs 459 and 460 (Single Level Spinal Fusion Except Cervical with and without MCC, respectively) and below MS-DRGs 456, 457, and 458.
Lastly, as discussed in section II.C.9. of the preamble of the proposed rule and this final rule, we proposed to revise the surgical hierarchy for the MDC 17 (Myeloproliferative Diseases and Disorders, Poorly Differentiated Neoplasms) MS-DRGs as follows: For the MDC 17 MS-DRGs, we proposed to sequence proposed new MS-DRG 850 (Acute Leukemia with Other Procedures) above MS-DRGs 823, 824, and 825 (Lymphoma and Non-Acute Leukemia with Other Procedures with MCC, with CC, and without CC/MCC, respectively) and below MS-DRGs 820, 821, and 822 (Lymphoma and Leukemia with Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively).
Our proposal for Appendix D MS-DRG Surgical Hierarchy by MDC and MS-DRG of the version of the ICD-10 MS-DRG Definitions Manual Version 42 is illustrated in the following tables.

Comment: A few commenters stated that they acknowledged the proposed conforming changes to the surgical hierarchy in association with the proposed MS-DRG classification changes, and acknowledged that the MS-DRG weight impacts the cost analysis, which in turn affects the hierarchy in the GROUPER. Regarding the proposed changes to MDC 08, the commenters stated that it is important to consider that it is not all multiple level spinal fusion procedures that appear to have the greatest impact on the proposed surgical hierarchy sequencing, rather it appears that it is the combined spinal fusion procedures. The commenters specified that the four highest MS-DRG categories listed in the proposed surgical hierarchy table for MDC 08 reflect combined spinal fusion procedures (MS-DRGs 453, 454, 455, 426, 427, 428, 420, 429, and 430). The commenters also remarked that proposed MS-DRGs 447 and 448, which describe multiple level spinal fusion procedures, are proposed to be sequenced below proposed MS-DRG 402 that describes single level combined anterior and posterior spinal fusion procedures, and below existing MS-DRGs 456, 457, and 458 that include both single level and multiple level spinal fusion procedures. The commenters stated that although they agreed with the proposed surgical hierarchy, the data appear to indicate that it is not only the multiple level spinal fusion procedures that are impacting the length of stay and average costs among the proposed MS-DRGs since the proposed MS-DRGs describing combined spinal fusion procedures appear to warrant the highest hierarchy regardless of single level or multiple levels. According to the commenters, the discussion in the proposed rule suggested that the number of levels impacts resource utilization, however, the data to differentiate cases where both multiple and single level spinal fusion procedures were performed on the same patient or during the same operative episode did not appear to impact resource utilization based on the data analysis provided.
Response: We appreciate the commenters' feedback and support. We note that while the commenters listed MS-DRGs 453, 454, and 455 among one ( print page 69100) of the four categories reflecting combined spinal fusion procedures having the greatest impact in the proposed surgical hierarchy for MDC 08, we believe that since those MS-DRGs were proposed to be deleted, the commenters' intent was for CMS to instead consider the three categories of proposed spinal fusion MS-DRGs (426, 427, and 428; 402; 429 and 430) for which the proposed logic for case assignment was derived from MS-DRGs 453, 454, and 455. Because the proposed logic for case assignment to the three categories of proposed spinal fusion MS-DRGs includes both concepts, (that is, multiple level combined spinal fusion procedures and single level combined spinal fusion procedures), we believe additional review is warranted with respect to the commenters' concerns regarding which aspect may have a greater impact on resource utilization. We intend to consider if the development of evaluation criteria would be useful for future proposed modifications to the surgical hierarchy for MS-DRGs that have meaningful changes to the clinical logic.
In the absence of a specific example, we are unclear why the commenter referenced data to differentiate cases where both multiple and single level spinal fusion procedures were performed on the same patient or during the same operative episode and its impact on resource utilization with respect to the data analysis provided in the proposed rule since, as stated in the proposed rule, the spinal fusion cases (for example, from MS-DRGs 453, 454, and 455) were separated into three categories (single level combined anterior and posterior fusions except cervical, multiple level combined anterior and posterior fusions except cervical, and combined anterior and posterior cervical spinal fusions), according to the proposed logic lists made available in Tables 6P.2d, 6P.2e, and 6P.2f in association with the proposed rule. The data analysis findings presented in the proposed rule show the difference in the number of cases, average length of stay, and average costs between multiple level and single level combined anterior and posterior spinal fusion cases. In consideration of the proposed logic, it would not be possible for a case to be reflected under both proposed MS-DRG categories at the same time.
Therefore, after consideration of the public comments we received, and based on the changes that we are finalizing for FY 2025, as discussed in section II.C. of the preamble of this final rule, we are finalizing our proposals to modify the existing surgical hierarchy, effective with the ICD-10 MS-DRGs Version 42, with modification. As discussed in section II.C.6.b., we are deleting MS-DRGs 459 and 460 and creating new MS-DRGs 450 and 451 (Single Level Spinal Fusion Except Cervical with MCC and without MCC, respectively). The finalized surgical hierarchy for MDC 08 is shown in the following table.

For issues pertaining to the surgical hierarchy, as with other MS-DRG related requests, we encourage interested parties to submit comments no later than October 20, 2024, via the Medicare Electronic Application Request Information System TM (MEARIS TM ) at https://mearis.cms.gov/public/home , so that they can be ( print page 69101) considered for possible inclusion in the annual proposed rule. We will consider these public comments for possible proposals in future rulemaking as part of our annual review process.
In September 1985, the ICD-9-CM Coordination and Maintenance Committee was formed. This is a Federal interdepartmental committee, co-chaired by the Centers for Disease Control and Prevention's (CDC) National Center for Health Statistics (NCHS) and CMS, charged with maintaining and updating the ICD-9-CM system. The final update to ICD-9-CM codes was made on October 1, 2013. Thereafter, the name of the Committee was changed to the ICD-10 Coordination and Maintenance Committee, effective with the March 19-20, 2014 meeting. The ICD-10 Coordination and Maintenance Committee addresses updates to the ICD-10-CM and ICD-10-PCS coding systems. The Committee is jointly responsible for approving coding changes, and developing errata, addenda, and other modifications to the coding systems to reflect newly developed procedures and technologies and newly identified diseases. The Committee is also responsible for promoting the use of Federal and non-Federal educational programs and other communication techniques with a view toward standardizing coding applications and upgrading the quality of the classification system.
The official list of ICD-9-CM diagnosis and procedure codes by fiscal year can be found on the CMS website at: https://www.cms.gov/medicare/coding-billing/icd-10-codes/icd-9-cm-diagnosis-procedure-codes-abbreviated-and-full-code-titles .
The official list of ICD-10-CM and ICD-10-PCS codes can be found on the CMS website at: http://www.cms.gov/Medicare/Coding/ICD10/index.html .
The NCHS has lead responsibility for the ICD-10-CM and ICD-9-CM diagnosis codes included in the Tabular List and Alphabetic Index for Diseases, while CMS has lead responsibility for the ICD-10-PCS and ICD-9-CM procedure codes included in the Tabular List and Alphabetic Index for Procedures.
The Committee encourages participation in the previously mentioned process by health-related organizations. In this regard, the Committee holds public meetings for discussion of educational issues and proposed coding changes. These meetings provide an opportunity for representatives of recognized organizations in the coding field, such as the American Health Information Management Association (AHIMA), the American Hospital Association (AHA), and various physician specialty groups, as well as individual physicians, health information management professionals, and other members of the public, to contribute ideas on coding matters. After considering the opinions expressed during the public meetings and in writing, the Committee formulates recommendations, which then must be approved by the agencies.
The Committee presented proposals for coding changes for implementation in FY 2025 at a public meeting held on September 12-13, 2023, and finalized the coding changes after consideration of comments received at the meetings and in writing by November 15, 2023.
The Committee held its Spring 2024 meeting on March 19-20, 2024. The deadline for submitting comments on these code proposals was April 19, 2024. It was announced at this meeting that any new diagnosis and procedure codes for which there was consensus of public support, and for which complete tabular and indexing changes would be made by June 2024 would be included in the October 1, 2024 update to the ICD-10-CM diagnosis and ICD-10-PCS procedure code sets. As discussed in earlier sections of the preamble of this final rule, there are new, revised, and deleted ICD-10-CM diagnosis codes and ICD-10-PCS procedure codes that are captured in Table 6A.—New Diagnosis Codes, Table 6B.—New Procedure Codes, Table 6C.—Invalid Diagnosis Codes, Table 6D.—Invalid Procedure Codes, Table 6E.—Revised Diagnosis Code Titles, and Table 6F.—Revised Procedure Code Titles for this final rule, which are available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps .
The code titles are adopted as part of the ICD-10 Coordination and Maintenance Committee process. Therefore, although we make the code titles available for the IPPS proposed and final rules, they are not subject to comment in the proposed or final rule. Because of the length of these tables, they are not published in the Addendum to the proposed or final rule. Rather, they are available on the CMS website as discussed in section VI. of the Addendum to the proposed rule and this final rule.
Recordings for the virtual meeting discussions of the procedure codes at the Committee's September 12-13, 2023 meeting and the March 19-20, 2024 meeting can be obtained from the CMS website at: https://www.cms.gov/Medicare/Coding/ICD10/C-and-M-Meeting-Materials . The materials for the discussions relating to diagnosis codes at the September 12-13, 2023 meeting and March 19-20, 2024 meeting can be found at: https://www.cdc.gov/nchs/icd/icd-10-maintenance/meetings.html . These websites also provide detailed information about the Committee, including information on requesting a new code, participating in a Committee meeting, timeline requirements and meeting dates.
We encourage commenters to submit questions and comments on coding issues involving diagnosis codes via Email to: [email protected] .
Questions and comments concerning the procedure codes should be submitted via Email to: [email protected] .
As discussed in the proposed rule, CMS implemented 41 new procedure codes including the insertion of a palladium-103 collagen implant into the brain, the excision or resection of intestinal body parts using a laparoscopic hand-assisted approach, the transfer of omentum for pedicled omentoplasty procedures, and the administration of talquetamab into the ICD-10-PCS classification effective with discharges on and after April 1, 2024. The procedure codes are as follows:

The 41 procedure codes are also reflected in Table 6B—New Procedure Codes in association with the proposed rule and available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS . As with the other new procedure codes and MS-DRG assignments included in Table 6B in association with the proposed rule, we solicited public comments on the most appropriate MDC, MS-DRG, and operating room status assignments for these codes for FY 2025, as well as any other options for the GROUPER logic. We discuss the comments we received on these assignments in section II.C.13. of this final rule as well as our finalized assignments, including to add new procedure code 02583ZF to the logic for case assignment to new MS-DRG 317 for FY 2025, as reflected in Table 6B.—New Procedure Codes in association with this final rule.
In the proposed rule, we also noted that Change Request (CR) 13458, Transmittal 12384, titled “April 2024 Update to the Medicare Severity—Diagnosis Related Group (MS-DRG) Grouper and Medicare Code Editor (MCE) Version 41.1” was issued on November 30, 2023 (available on the CMS website at: https://www.cms.gov/regulations-and-guidance/guidance/transmittals/2023-transmittals/r12384cp ) regarding the release of an updated version of the ICD-10 MS-DRG GROUPER and Medicare Code Editor software, Version 41.1, effective with discharges on and after April 1, 2024, reflecting the new procedure codes. The updated software, along with the updated ICD-10 MS-DRG Version 41.1 Definitions Manual and the Definitions of Medicare Code Edits Version 41.1 manual is available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
In the September 7, 2001 final rule implementing the IPPS new technology add-on payments ( 66 FR 46906 ), we indicated we would attempt to include proposals for procedure codes that would describe new technology discussed and approved at the Spring meeting as part of the code revisions effective the following October.
Section 503(a) of the Medicare Modernization Act ( Pub. L. 108-173 ) included a requirement for updating diagnosis and procedure codes twice a year instead of a single update on October 1 of each year. This requirement was included as part of the amendments to the Act relating to recognition of new technology under the IPPS. Section 503(a) of Public Law 108-173 amended section 1886(d)(5)(K) of the Act by adding a clause (vii) which states that the Secretary shall provide for the addition of new diagnosis and procedure codes on April 1 of each year, but the addition of such codes shall not require the Secretary to adjust the payment (or diagnosis-related group classification) until the fiscal year that begins after such date. This requirement improves the recognition of new technologies under the IPPS by providing information on these new technologies at an earlier date. Data will ( print page 69105) be available 6 months earlier than would be possible with updates occurring only once a year on October 1.
In the FY 2005 IPPS final rule, we implemented section 1886(d)(5)(K)(vii) of the Act, as added by section 503(a) of Public Law 108-173 , by developing a mechanism for approving, in time for the April update, diagnosis and procedure code revisions needed to describe new technologies and medical services for purposes of the new technology add-on payment process. We also established the following process for making these determinations. Topics considered during the Fall ICD-10 (previously ICD-9-CM) Coordination and Maintenance Committee meeting were considered for an April 1 update if a strong and convincing case was made by the requestor during the Committee's public meeting. The request needed to identify the reason why a new code was needed in April for purposes of the new technology process. Meeting participants and those reviewing the Committee meeting materials were provided the opportunity to comment on the expedited request. We refer the reader to the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44950 ) for further discussion of the implementation of this prior April 1 update for purposes of the new technology add-on payment process.
However, as discussed in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44950 through 44956 ), we adopted an April 1 implementation date, in addition to the annual October 1 update, beginning with April 1, 2022. We noted that the intent of this April 1 implementation date is to allow flexibility in the ICD-10 code update process. With this new April 1 update, CMS now uses the same process for consideration of all requests for an April 1 implementation date, including for purposes of the new technology add-on payment process (that is, the prior process for consideration of an April 1 implementation date only if a strong and convincing case was made by the requestor during the meeting no longer applies). We are continuing to use several aspects of our existing established process to implement new codes through the April 1 code update, which includes presenting proposals for April 1 consideration at the September ICD-10 Coordination and Maintenance Committee meeting, requesting public comments, reviewing the public comments, finalizing codes, and announcing the new codes with their assignments consistent with the new GROUPER release information. We note that under our established process, requestors indicate whether they are submitting their code request for consideration for an April 1 implementation date or an October 1 implementation date. The ICD-10 Coordination and Maintenance Committee makes efforts to accommodate the requested implementation date for each request submitted. However, the Committee determines which requests are to be presented for consideration for an April 1 implementation date or an October 1 implementation date. As discussed earlier in this section of the preamble of this final rule, there were code proposals presented for an April 1, 2024 implementation at the September 12-13, 2023 Committee meetings. Following the receipt of public comments, the code proposals were approved and finalized, therefore, there were new codes implemented April 1, 2024.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule, consistent with the process we outlined for the April 1 implementation date, we announced the new codes in November 2023 and provided the updated code files in December 2023 and ICD-10-CM Official Guidelines for Coding and Reporting in January 2024. In the February 05, 2024 Federal Register ( 89 FR 7710 ), notice for the March 19-20, 2024 ICD-10 Coordination and Maintenance Committee Meeting was published that includes the tentative agenda and identifies which topics are related to a new technology add-on payment application. By February 1, 2024, we made available the updated Version 41.1 ICD-10 MS-DRG GROUPER software and related materials on the CMS web page at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
ICD-9-CM addendum and code title information is published on the CMS website at https://www.cms.gov/medicare/coding-billing/icd-10-codes/updates-revisions-icd-9-cm-procedure-codes-addendum . ICD-10-CM and ICD-10-PCS addendum and code title information is published on the CMS website at https://www.cms.gov/medicare/coding-billing/icd-10-codes . CMS also sends electronic files containing all ICD-10-CM and ICD-10-PCS coding changes to its Medicare contractors for use in updating their systems and providing education to providers. Information on ICD-10-CM diagnosis codes, along with the Official ICD-10-CM Coding Guidelines, can be found on the CDC website at https://www.cdc.gov/nchs/icd/icd-10-cm/files.html . Additionally, information on new, revised, and deleted ICD-10-CM diagnosis and ICD-10-PCS procedure codes is provided to the AHA for publication in the Coding Clinic for ICD-10. The AHA also distributes coding update information to publishers and software vendors.
In the proposed rule, we noted that for FY 2024, there are currently 74,044 diagnosis codes and 78,638 procedure codes. We also noted that as displayed in Table 6A.—New Diagnosis Codes and in Table 6B.—New Procedure Codes associated with the proposed rule (and available on the CMS website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps ), there are 252 new diagnosis codes that had been finalized for FY 2025 at the time of the development of the proposed rule and 41 new procedure codes that were effective with discharges on and after April 1, 2024.
As discussed in section II.C.13 of the preamble of this final rule, we are making Table 6A.—New Diagnosis Codes, Table 6B.—New Procedure Codes, Table 6C.—Invalid Diagnosis Codes, Table 6D.—Invalid Procedure Codes, Table 6E.—Revised Diagnosis Code Titles and Table 6F.—Revised Procedure Code Titles available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps in association with this final rule. As shown in Table 6B.—New Procedure Codes, there were procedure codes discussed at the March 19-20, 2024 ICD-10 Coordination and Maintenance Committee meeting that were not finalized in time to include in the proposed rule and are identified with an asterisk. We refer the reader to Table 6B.—New Procedure Codes associated with this final rule and available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps for the detailed list of these 371 new procedure codes finalized for FY 2025.
We also note, as reflected in Table 6C.—Invalid Diagnosis Codes and in Table 6D.—Invalid Procedure Codes, there are a total of 36 diagnosis codes and 61 procedure codes that will become invalid effective October 1, 2024. Based on these code updates, effective October 1, 2024, there are a total of 74,260 ICD-10-CM diagnosis codes and 78,948 ICD-10-PCS procedure codes for FY 2025 as shown in the following table.

As stated previously, the public is provided the opportunity to comment on any requests for new diagnosis or procedure codes discussed at the ICD-10 Coordination and Maintenance Committee meeting. The code titles are adopted as part of the ICD-10 Coordination and Maintenance Committee process. Thus, although we publish the code titles in the IPPS proposed and final rules, they are not subject to comment in the proposed or final rules.
In the FY 2008 IPPS final rule with comment period ( 72 FR 47246 through 47251 ), we discussed the topic of Medicare payment for devices that are replaced without cost or where credit for a replaced device is furnished to the hospital. We implemented a policy to reduce a hospital's IPPS payment for certain MS-DRGs where the implantation of a device that subsequently failed or was recalled determined the base MS-DRG assignment. At that time, we specified that we will reduce a hospital's IPPS payment for those MS-DRGs where the hospital received a credit for a replaced device equal to 50 percent or more of the cost of the device.
In the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51556 through 51557 ), we clarified this policy to state that the policy applies if the hospital received a credit equal to 50 percent or more of the cost of the replacement device and issued instructions to hospitals accordingly.
As discussed in section II.C.5. of the preamble of the proposed rule and this final rule, for FY 2025, we proposed to revise the title of MS-DRG 276 from “Cardiac Defibrillator Implant with MCC” to “Cardiac Defibrillator Implant with MCC or Carotid Sinus Neurostimulator”.
As stated in the FY 2016 IPPS/LTCH PPS proposed rule ( 80 FR 24409 ), we generally map new MS-DRGs onto the list when they are formed from procedures previously assigned to MS-DRGs that are already on the list. Currently, MS-DRG 276 is on the list of MS-DRGs subject to the policy for payment under the IPPS for replaced devices offered without cost or with a credit as shown in the following table. Therefore, we proposed that if the applicable proposed MS-DRG changes are finalized, we would make conforming changes to the title of MS-DRG 276 as reflected in the table that follows. We also proposed to continue to include the existing MS-DRGs currently subject to the policy.
As discussed in section II.C.5. of the preamble of this final rule, we are finalizing our proposal to revise the title of MS-DRG 276 from “Cardiac Defibrillator Implant with MCC” to “Cardiac Defibrillator Implant with MCC or Carotid Sinus Neurostimulator”. We did not receive any public comments opposing our proposal make conforming changes to the title of MS-DRG 276 in the list of MS-DRGs that will be subject to the replaced devices offered without cost or with a credit policy effective October 1, 2024. Additionally, we did not receive any public comments opposing our proposal to continue to include the existing MS-DRGs currently subject to the policy. Therefore, we are finalizing the list of MS-DRGs in the following table that will be subject to the replaced devices offered without cost or with a credit policy effective October 1, 2024.

The final list of MS-DRGs subject to the IPPS policy for replaced devices offered without cost or with a credit will be issued to providers in the form of a Change Request (CR).
We received public comments on MS-DRG related issues that were ( print page 69109) outside the scope of the proposals included in the FY 2025 IPPS/LTCH PPS proposed rule.
Because we consider these public comments to be outside the scope of the proposed rule, we are not addressing them in this final rule. As stated in section II.C.1.b. of the preamble of this final rule, we encourage individuals with comments about MS-DRG classifications to submit these comments no later than October 20, 2024, via the Medicare Electronic Application Request Information System TM (MEARIS TM ) at: https://mearis.cms.gov/public/home , so that they can be considered for possible inclusion in the annual proposed rule. We will consider these public comments for possible proposals in future rulemaking as part of our annual review process.
Consistent with our established policy, in developing the MS-DRG relative weights for FY 2025, we proposed to use two data sources: claims data and cost report data. The claims data source is the MedPAR file, which includes fully coded diagnostic and procedure data for all Medicare inpatient hospital bills. The FY 2023 MedPAR data used in this final rule include discharges occurring on October 1, 2022, through September 30, 2023, based on bills received by CMS through March 31, 2024, from all hospitals subject to the IPPS and short-term, acute care hospitals in Maryland (which at that time were under a waiver from the IPPS).
The FY 2023 MedPAR file used in calculating the relative weights includes data for approximately 6,916,571 Medicare discharges from IPPS providers. Discharges for Medicare beneficiaries enrolled in a Medicare Advantage managed care plan are excluded from this analysis. These discharges are excluded when the MedPAR “GHO Paid” indicator field on the claim record is equal to “1” or when the MedPAR DRG payment field, which represents the total payment for the claim, is equal to the MedPAR “Indirect Medical Education (IME)” payment field, indicating that the claim was an “IME only” claim submitted by a teaching hospital on behalf of a beneficiary enrolled in a Medicare Advantage managed care plan. In addition, the March 2024 update of the FY 2023 MedPAR file complies with version 5010 of the X12 HIPAA Transaction and Code Set Standards, and includes a variable called “claim type.” Claim type “60” indicates that the claim was an inpatient claim paid as fee-for-service. Claim types “61,” “62,” “63,” and “64” relate to encounter claims, Medicare Advantage IME claims, and HMO no-pay claims. Therefore, the calculation of the relative weights for FY 2025 also excludes claims with claim type values not equal to “60.” The data exclude CAHs, including hospitals that subsequently became CAHs after the period from which the data were taken. In addition, the data exclude Rural Emergency Hospitals (REHs), including hospitals that subsequently became REHs after the period from which the data were taken. We note that the FY 2025 relative weights are based on the ICD-10-CM diagnosis codes and ICD-10-PCS procedure codes from the FY 2023 MedPAR claims data, grouped through the ICD-10 version of the FY 2025 GROUPER (Version 42).
The second data source used in the cost-based relative weighting methodology is the Medicare cost report data files from the Healthcare Cost Report Information System (HCRIS). In general, we use the HCRIS dataset that is 3 years prior to the IPPS fiscal year. Specifically, for this final rule, we used the March 2024 update of the FY 2022 HCRIS for calculating the FY 2025 cost-based relative weights. Consistent with our historical practice, for this FY 2025 final rule, we are providing the version of the HCRIS from which we calculated these 19 cost-to charge-ratios (CCRs) on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS . Click on the link on the left side of the screen titled “FY 2025 IPPS Final Rule Home Page” or “Acute Inpatient Files for Download.”
We calculated the FY 2025 relative weights based on 19 CCRs. The methodology we proposed to use to calculate the FY 2025 MS-DRG cost-based relative weights based on claims data in the FY 2023 MedPAR file and data from the FY 2022 Medicare cost reports is as follows:
- To the extent possible, all the claims were regrouped using the FY 2025 MS-DRG classifications discussed in sections II.B. and II.C. of the preamble of this final rule.
- The transplant cases that were used to establish the relative weights for heart and heart-lung, liver and/or intestinal, and lung transplants (MS-DRGs 001, 002, 005, 006, and 007, respectively) were limited to those Medicare-approved transplant centers that have cases in the FY 2023 MedPAR file. (Medicare coverage for heart, heart-lung, liver and/or intestinal, and lung transplants is limited to those facilities that have received approval from CMS as transplant centers.)
- Organ acquisition costs for kidney, heart, heart-lung, liver, lung, pancreas, and intestinal (or multivisceral organs) transplants continue to be paid on a reasonable cost basis. Because these acquisition costs are paid separately from the prospective payment rate, it is necessary to subtract the acquisition charges from the total charges on each transplant bill that showed acquisition charges before computing the average cost for each MS-DRG and before eliminating statistical outliers.
Section 108 of the Further Consolidated Appropriations Act, 2020 provides that, for cost reporting periods beginning on or after October 1, 2020, costs related to hematopoietic stem cell acquisition for the purpose of an allogeneic hematopoietic stem cell transplant shall be paid on a reasonable cost basis. We refer the reader to the FY 2021 IPPS/LTCH PPS final rule for further discussion of the reasonable cost basis payment for cost reporting periods beginning on or after October 1, 2020 ( 85 FR 58835 through 58842 ). For FY 2022 and subsequent years, we subtract the hematopoietic stem cell acquisition charges from the total charges on each transplant bill that showed hematopoietic stem cell acquisition charges before computing the average cost for each MS-DRG and before eliminating statistical outliers.
- Claims with total charges or total lengths of stay less than or equal to zero were deleted. Claims that had an amount in the total charge field that differed by more than $30.00 from the sum of the routine day charges, intensive care charges, pharmacy charges, implantable devices charges, supplies and equipment charges, therapy services charges, operating room charges, cardiology charges, laboratory charges, radiology charges, other service charges, labor and delivery charges, inhalation therapy charges, emergency room charges, blood and blood products charges, anesthesia charges, cardiac catheterization charges, CT scan charges, and MRI charges were also deleted.
- At least 92.6 percent of the providers in the MedPAR file had charges for 14 of the 19 cost centers. All claims of providers that did not have charges greater than zero for at least 14 ( print page 69110) of the 19 cost centers were deleted. In other words, a provider must have no more than five blank cost centers. If a provider did not have charges greater than zero in more than five cost centers, the claims for the provider were deleted.
- Statistical outliers were eliminated by removing all cases that were beyond 3.0 standard deviations from the geometric mean of the log distribution of both the total charges per case and the total charges per day for each MS-DRG.
- Effective October 1, 2008, because hospital inpatient claims include a Present on Admission (POA) field for each diagnosis present on the claim, only for purposes of relative weight-setting, the POA indicator field was reset to “Y” for “Yes” for all claims that otherwise have an “N” (No) or a “U” (documentation insufficient to determine if the condition was present at the time of inpatient admission) in the POA field.
Under current payment policy, the presence of specific HAC codes, as indicated by the POA field values, can generate a lower payment for the claim. Specifically, if the particular condition is present on admission (that is, a “Y” indicator is associated with the diagnosis on the claim), it is not a HAC, and the hospital is paid for the higher severity (and, therefore, the higher weighted MS-DRG). If the particular condition is not present on admission (that is, an “N” indicator is associated with the diagnosis on the claim) and there are no other complicating conditions, the DRG GROUPER assigns the claim to a lower severity (and, therefore, the lower weighted MS-DRG) as a penalty for allowing a Medicare inpatient to contract a HAC. While the POA reporting meets policy goals of encouraging quality care and generates program savings, it presents an issue for the relative weight-setting process. Because cases identified as HACs are likely to be more complex than similar cases that are not identified as HACs, the charges associated with HAC cases are likely to be higher as well. Therefore, if the higher charges of these HAC claims are grouped into lower severity MS-DRGs prior to the relative weight-setting process, the relative weights of these particular MS-DRGs would become artificially inflated, potentially skewing the relative weights. In addition, we want to protect the integrity of the budget neutrality process by ensuring that, in estimating payments, no increase to the standardized amount occurs as a result of lower overall payments in a previous year that stem from using weights and case-mix that are based on lower severity MS-DRG assignments. If this would occur, the anticipated cost savings from the HAC policy would be lost.
To avoid these problems, we reset the POA indicator field to “Y” only for relative weight-setting purposes for all claims that otherwise have an “N” or a “U” in the POA field. This resetting “forced” the more costly HAC claims into the higher severity MS-DRGs as appropriate, and the relative weights calculated for each MS-DRG more closely reflect the true costs of those cases.
In addition, in the FY 2013 IPPS/LTCH PPS final rule, for FY 2013 and subsequent fiscal years, we finalized a policy to treat hospitals that participate in the Bundled Payments for Care Improvement (BPCI) initiative the same as prior fiscal years for the IPPS payment modeling and ratesetting process without regard to hospitals' participation within these bundled payment models ( 77 FR 53341 through 53343 ). Specifically, because acute care hospitals participating in the BPCI Initiative still receive IPPS payments under section 1886(d) of the Act, we include all applicable data from these subsection (d) hospitals in our IPPS payment modeling and ratesetting calculations as if the hospitals were not participating in those models under the BPCI initiative. We refer readers to the FY 2013 IPPS/LTCH PPS final rule for a complete discussion on our final policy for the treatment of hospitals participating in the BPCI initiative in our ratesetting process. For additional information on the BPCI initiative, we refer readers to the CMS' Center for Medicare and Medicaid Innovation's website at https://innovation.cms.gov/initiatives/Bundled-Payments/index.html and to section IV.H.4. of the preamble of the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53341 through 53343 ).
The participation of hospitals in the BPCI initiative concluded on September 30, 2018. The participation of hospitals in the BPCI Advanced model started on October 1, 2018. The BPCI Advanced model, tested under the authority of section 1115A of the Act, is comprised of a single payment and risk track, which bundles payments for multiple services that beneficiaries receive during a Clinical Episode. Acute care hospitals may participate in BPCI Advanced in one of two capacities: as a model Participant or as a downstream Episode Initiator. Regardless of the capacity in which they participate in the BPCI Advanced model, participating acute care hospitals will continue to receive IPPS payments under section 1886(d) of the Act. Acute care hospitals that are Participants also assume financial and quality performance accountability for Clinical Episodes in the form of a reconciliation payment. For additional information on the BPCI Advanced model, we refer readers to the BPCI Advanced web page on the CMS Center for Medicare and Medicaid Innovation's website at https://innovation.cms.gov/initiatives/bpci-advanced . Consistent with our policy for FY 2024, and consistent with how we have treated hospitals that participated in the BPCI Initiative, for FY 2025, we continue to believe it is appropriate to include all applicable data from the subsection (d) hospitals participating in the BPCI Advanced model in our IPPS payment modeling and ratesetting calculations because, as noted previously, these hospitals are still receiving IPPS payments under section 1886(d) of the Act. Consistent with the FY 2024 IPPS/LTCH PPS final rule, we also proposed to include all applicable data from subsection (d) hospitals participating in the Comprehensive Care for Joint Replacement (CJR) Model in our IPPS payment modeling and ratesetting calculations.
The charges for each of the 19 cost groups for each claim were standardized to remove the effects of differences in area wage levels, IME and DSH payments, and for hospitals located in Alaska and Hawaii, the applicable cost-of-living adjustment. Because hospital charges include charges for both operating and capital costs, we standardized total charges to remove the effects of differences in geographic adjustment factors, cost-of-living adjustments, and DSH payments under the capital IPPS as well. Charges were then summed by MS-DRG for each of the 19 cost groups so that each MS-DRG had 19 standardized charge totals. Statistical outliers were then removed. These charges were then adjusted to cost by applying the national average CCRs developed from the FY 2022 cost report data.
The 19 cost centers that we used in the relative weight calculation are shown in a supplemental data file, Cost Center HCRIS Lines Supplemental Data File, posted via the internet on the CMS website for this final rule and available at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS . The supplemental data file shows the lines on the cost report and the corresponding revenue codes that we used to create the 19 national cost center CCRs. We stated in the proposed rule that if we receive comments about the groupings in this ( print page 69111) supplemental data file, we may consider these comments as we finalize our policy. However, we did not receive any comments on the groupings in this table, and therefore, we are finalizing the groupings as proposed.
Consistent with historical practice, we account for rare situations of non-monotonicity in a base MS-DRG and its severity levels, where the mean cost in the higher severity level is less than the mean cost in the lower severity level, in determining the relative weights for the different severity levels. If there are initially non-monotonic relative weights in the same base DRG and its severity levels, then we combine the cases that group to the specific non-monotonic MS-DRGs for purposes of relative weight calculations. For example, if there are two non-monotonic MS-DRGs, combining the cases across those two MS-DRGs results in the same relative weight for both MS-DRGs. The relative weight calculated using the combined cases for those severity levels is monotonic, effectively removing any non-monotonicity with the base DRG and its severity levels. For this FY 2025 final rule, this calculation was applied to address non-monotonicity for cases that grouped to the following: MS-DRG 016 and MS-DRG 017, MS-DRG 095 and MS-DRG 096, MS-DRG 504 and MS-DRG 505, MS-DRG 797 and MS-DRG 798. In the supplemental file titled AOR/BOR File, we include statistics for the affected MS-DRGs both separately and with cases combined.
We invited public comments on our proposals related to recalibration of the proposed FY 2025 relative weights and the changes in relative weights from FY 2024.
We received several comments that we consider to be out of scope. For example, a commenter requested a “device intensive” cost threshold. Because we consider these comments to be out of scope, we are not responding in this final rule.
After consideration of the comments received, we are finalizing our proposals without modifications related to the recalibration of the FY 2025 relative weights. We summarize and respond to comments relating to the methodology for calculating the relative weight for MS-DRG 018 in the next section of this final rule.
In the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58451 through 58453 ), we created MS-DRG 018 for cases that include procedures describing CAR T-cell therapies. We also finalized our proposal to modify our existing relative weight methodology to ensure that the relative weight for MS-DRG 018 appropriately reflects the relative resources required for providing CAR T-cell therapy outside of a clinical trial, while still accounting for the clinical trial cases in the overall average cost for all MS-DRGs ( 85 FR 58599 through 58600 ). Specifically, we stated that clinical trial claims that group to new MS-DRG 018 would not be included when calculating the average cost for MS-DRG 018 that is used to calculate the relative weight for this MS-DRG, so that the relative weight reflects the costs of the CAR T-cell therapy drug. We stated that we identified clinical trial claims as claims that contain ICD-10-CM diagnosis code Z00.6 or contain standardized drug charges of less than $373,000, which was the average sales price of KYMRIAH and YESCARTA, the two CAR T-cell biological products licensed to treat relapsed/refractory large B-cell lymphoma as of the time of the development of the FY 2021 final rule. In addition, we stated that (a) when the CAR T-cell therapy product is purchased in the usual manner, but the case involves a clinical trial of a different product, the claim will be included when calculating the average cost for new MS-DRG 018 to the extent such cases can be identified in the historical data, and (b) when there is expanded access use of immunotherapy, these cases will not be included when calculating the average cost for new MS-DRG 018 to the extent such cases can be identified in the historical data.
We also finalized our proposal to calculate an adjustment to account for the CAR T-cell therapy cases identified as clinical trial cases in calculating the national average standardized cost per case that is used to calculate the relative weights for all MS-DRGs and for purposes of budget neutrality and outlier simulations. We calculate this adjustor by dividing the average cost for cases that we identify as clinical trial cases by the average cost for cases that we identify as non-clinical trial cases, with the additional refinements that (a) when the CAR T-cell therapy product is purchased in the usual manner, but the case involves a clinical trial of a different product, the claim will be included when calculating the average cost for cases not determined to be clinical trial cases to the extent such cases can be identified in the historical data, and (b) when there is expanded access use of immunotherapy, these cases will be included when calculating the average cost for cases determined to be clinical trial cases to the extent such cases can be identified in the historical data. We stated that to the best of our knowledge, there were no claims in the historical data used in the calculation of this adjustment for cases involving a clinical trial of a different product, and to the extent the historical data contain claims for cases involving expanded access use of immunotherapy we believe those claims would have drug charges less than $373,000.
In the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58842 ), we also finalized an adjustment to the payment amount for applicable clinical trial and expanded access use immunotherapy cases that group to MS-DRG 018, and indicated that we would provide instructions for identifying these claims in separate guidance. Following the issuance of the FY 2021 IPPS/LTCH PPS final rule, we issued guidance [ 20 ] stating that providers may enter a Billing Note NTE02 “Expand Acc Use” on the electronic claim 837I or a remark “Expand Acc Use” on a paper claim to notify the MAC of expanded access use of CAR T-cell therapy. In this case, the MAC would add payer-only condition code “ZB” so that Pricer will apply the payment adjustment in calculating payment for the case. In cases when the CAR T-cell therapy product is purchased in the usual manner, but the case involves a clinical trial of a different product, the provider may enter a Billing Note NTE02 “Diff Prod Clin Trial” on the electronic claim 837I or a remark “Diff Prod Clin Trial” on a paper claim. In this case, the MAC would add payer-only condition code “ZC” so that the Pricer will not apply the payment adjustment in calculating payment for the case.
In the FY 2022 IPPS/LTCH PPS final rule, we revised MS-DRG 018 to include cases that report the procedure codes for CAR T-cell and non-CAR T-cell therapies and other immunotherapies ( 86 FR 44798 through 44806 ). We also finalized our proposal to continue to use the proxy of standardized drug charges of less than $373,000 ( 86 FR 44965 ) to identify clinical trial claims. We also finalized use of this same proxy for the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48894 ).
Following the issuance of the FY 2023 IPPS/LTCH PPS final rule, we issued guidance [ 21 ] stating where there is expanded access use of immunotherapy, the provider may submit condition code “90” on the claim so that Pricer will apply the payment adjustment in ( print page 69112) calculating payment for the case. We stated that MACs would no longer append Condition Code `ZB' to inpatient claims reporting Billing Note NTE02 “Expand Acc Use” on the electronic claim 837I or a remark “Expand Acc Use” on a paper claim, effective for claims for discharges that occur on or after October 1, 2022.
In the FY 2024 IPPS/LTCH PPS final rule, we explained that the MedPAR claims data now includes a field that identifies whether or not the claim includes expanded access use of immunotherapy. We stated that for the FY 2022 MedPAR claims data, this field identifies whether or not the claim includes condition code ZB, and for the FY 2023 MedPAR data and subsequent years, this field will identify whether or not the claim includes condition code 90. We further noted that the MedPAR files now also include a variable that indicates whether the claim includes the payer-only condition code “ZC”, which identifies a case involving the clinical trial of a different product where the CAR T-cell, non-CAR T-cell, or other immunotherapy product is purchased in the usual manner.
Accordingly, and as discussed further in the FY 2024 IPPS/LTCH PPS final rule, we finalized two modifications to our methodology for identifying clinical trial claims and expanded access use claims in MS-DRG 018 ( 88 FR 58791 ). First, we finalized to exclude claims with the presence of condition code “90” (or, for FY 2024 ratesetting, which was based on the FY 2022 MedPAR data, the presence of condition code “ZB”) and claims that contain ICD-10-CM diagnosis code Z00.6 without payer-only code “ZC” that group to MS-DRG 018 when calculating the average cost for MS-DRG 018. Second, we finalized to no longer use the proxy of standardized drug charges of less than $373,000 to identify clinical trial claims and expanded access use cases when calculating the average cost for MS-DRG 018. Accordingly, we finalized that in calculating the relative weight for MS-DRG 018 for FY 2024, only those claims that group to MS-DRG 018 that (1) contain ICD-10-CM diagnosis code Z00.6 and do not include payer-only code “ZC” or (2) contain condition code “ZB” (or, for subsequent fiscal years, condition code “90”) would be excluded from the calculation of the average cost for MS-DRG 018. Consistent with this, we also finalized modifications to our calculation of the adjustment to account for the CAR T-cell therapy cases identified as clinical trial cases in calculating the national average standardized cost per case that is used to calculate the relative weights for all MS-DRGs. We refer readers to the FY 2024 IPPS/LTCH PPS final rule for further discussion of these modifications ( 88 FR 58791 ).
In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to continue to use our methodology as modified in the FY 2024 IPPS/LTCH PPS final rule for identifying clinical trial claims and expanded access use claims in MS-DRG 018. First, we exclude claims with the presence of condition code “90” and claims that contain ICD-10-CM diagnosis code Z00.6 without payer-only code “ZC” that group to MS-DRG 018 when calculating the average cost for MS-DRG 018. Second, we no longer use the proxy of standardized drug charges of less than $373,000 to identify clinical trial claims and expanded access use cases when calculating the average cost for MS-DRG 018. Accordingly, we proposed that in calculating the relative weight for MS-DRG 018 for FY 2025, only those claims that group to MS-DRG 018 that (1) contain ICD-10-CM diagnosis code Z00.6 and do not include payer-only code “ZC” or (2) contain condition code “90” would be excluded from the calculation of the average cost for MS-DRG 018.
We also proposed to continue to use the methodology as modified in the FY 2024 IPPS/LTCH PPS final rule to calculate the adjustment to account for the CAR T-cell therapy cases identified as clinical trial cases in calculating the national average standardized cost per case that is used to calculate the relative weights for all MS-DRGs:
- Calculate the average cost for cases assigned to MS-DRG 018 that either (a) contain ICD-10-CM diagnosis code Z00.6 and do not contain condition code “ZC” or (b) contain condition code “90”.
- Calculate the average cost for all other cases assigned to MS-DRG 018.
- Calculate an adjustor by dividing the average cost calculated in step 1 by the average cost calculated in step 2.
- Apply the adjustor calculated in step 3 to the cases identified in step 1 as applicable clinical trial or expanded access use cases, then add this adjusted case count to the non-clinical trial case count prior to calculating the average cost across all MS-DRGs.
Under our proposal to continue to apply this methodology, based on the December 2023 update of the FY 2023 MedPAR file used for the proposed rule, we estimated that the average costs of cases assigned to MS-DRG 018 that are identified as clinical trial cases ($116,831) were 34 percent of the average costs of the cases assigned to MS-DRG 018 that are identified as non-clinical trial cases ($342,684). Accordingly, as we did for FY 2024, we proposed to adjust the transfer-adjusted case count for MS-DRG 018 by applying the proposed adjustor of 0.34 to the applicable clinical trial and expanded access use immunotherapy cases, and to use this adjusted case count for MS-DRG 018 in calculating the national average cost per case, which is used in the calculation of the relative weights. Therefore, in calculating the national average cost per case for purposes of the proposed rule, each case identified as an applicable clinical trial or expanded access use immunotherapy case was adjusted by 0.34. As we did for FY 2024, we applied this same adjustor for the applicable cases that group to MS-DRG 018 for purposes of budget neutrality and outlier simulations. We also proposed to update the value of the adjustor based on more recent data for the final rule.
Comment: A few commenters supported the proposal to continue to exclude CAR T-cell therapy clinical trial cases from the calculation of the relative weight for MS-DRG 018. A commenter stated that the proposal for CAR T-cell therapy payment is largely responsive to previous requests for a permanent reimbursement solution for CAR T-cell therapy in a manner that reflects the cost of care.
Response: We thank commenters for their support and input on the proposed methodology.
Comment: Some commenters expressed concern that CMS no longer uses the $373,000 threshold to identify clinical trial cases. The commenters stated that a small number of claims are still coded incorrectly, and that this has the potential to reduce the relative weight for MS-DRG 018 due to the presence of lower cost cases that should be flagged as clinical trial cases. Another commenter expressed concern that CMS' methodology may not be accurately capturing some cases where the CAR T product is not purchased in the usual manner, such as when the patient receives the product as part of a patient assistance program. This commenter suggested that CMS establish a mechanism for hospitals to report when a product is obtained at no cost for reasons other than participation in a clinical trial or expanded access use. A commenter requested that CMS provide the proportion of cases with drug charges below $373,000 that do not have a clinical trial or expanded access use code.
Response: As we stated in the FY 2024 IPPS/LTCH PPS final rule, while there continues to be a small percentage of claims that report standardized drug ( print page 69113) charges of less than $373,000 and do not report ICD-10-CM code Z00.6, we do not believe it is necessary to continue the use of the proxy until the number of cases reaches zero. With respect to the commenter's suggestion regarding a mechanism for reporting products obtained at no cost for reasons other than participation in a clinical trial or expanded access use, we may consider this in the future. With respect to the commenter who requested that CMS provide the proportion of cases with drug charges below $373,000, that proportion is 4%, which is the same percentage as last year. We note that information on obtaining the MedPAR Limited Data Set is available on the CMS website, at https://www.cms.gov/Research-Statistics-Data-and-Systems/Files-for-Order/LimitedDataSets/MEDPARLDSHospitalNational .
After consideration of the public comments we received, we are finalizing our proposals without modifications regarding the calculation of the relative weight for MS-DRG 018. Applying this finalized methodology, based on the March 2024 update of the FY 2023 MedPAR file used for this final rule, we estimated that the average costs of cases assigned to MS-DRG 018 that are identified as clinical trial cases ($111,211) were 33 percent of the average costs of the cases assigned to MS-DRG 018 that are identified as non-clinical trial cases ($334,119). Accordingly, as we did for FY 2024, we are finalizing our proposal to adjust the transfer-adjusted case count for MS-DRG 018 by applying the adjustor of 0.33 to the applicable clinical trial and expanded access use immunotherapy cases, and to use this adjusted case count for MS-DRG 018 in calculating the national average cost per case, which is used in the calculation of the relative weights. Therefore, in calculating the national average cost per case for purposes of this final rule, each case identified as an applicable clinical trial or expanded access use immunotherapy case was adjusted by 0.33. As we did for FY 2024, we are applying this same adjustor for the applicable cases that group to MS-DRG 018 for purposes of budget neutrality and outlier simulations.
In the FY 2023 IPPS/LTCH PPS final rule, we finalized a permanent 10-percent cap on the reduction in an MS-DRG's relative weight in a given fiscal year, beginning in FY 2023. We also finalized a budget neutrality adjustment to the standardized amount for all hospitals to ensure that application of the permanent 10-percent cap does not result in an increase or decrease of estimated aggregate payments. We refer the reader to the FY 2023 IPPS/LTCH PPS final rule for further discussion of this policy. In the Addendum to this IPPS/LTCH PPS final rule, we present the budget neutrality adjustment for reclassification and recalibration of the FY 2025 MS-DRG relative weights with application of this cap. We are also making available on the CMS website a supplemental file demonstrating the application of the permanent 10 percent cap for FY 2025. For a further discussion of the budget neutrality adjustment for FY 2025, we refer readers to the Addendum of this final rule.
We developed the national average CCRs as follows:
Using the FY 2022 cost report data, we removed CAHs, Indian Health Service hospitals, all-inclusive rate hospitals, and cost reports that represented time periods of less than 1 year (365 days). We included hospitals located in Maryland because we include their charges in our claims database. Then we created CCRs for each provider for each cost center (see the supplemental data file for line items used in the calculations) and removed any CCRs that were greater than 10 or less than 0.01. We normalized the departmental CCRs by dividing the CCR for each department by the total CCR for the hospital for the purpose of trimming the data. Then we took the logs of the normalized cost center CCRs and removed any cost center CCRs where the log of the cost center CCR was greater or less than the mean log plus/minus 3 times the standard deviation for the log of that cost center CCR. Once the cost report data were trimmed, we calculated a Medicare-specific CCR. The Medicare-specific CCR was determined by taking the Medicare charges for each line item from Worksheet D-3 and deriving the Medicare-specific costs by applying the hospital-specific departmental CCRs to the Medicare-specific charges for each line item from Worksheet D-3. Once each hospital's Medicare-specific costs were established, we summed the total Medicare-specific costs and divided by the sum of the total Medicare-specific charges to produce national average, charge-weighted CCRs.
After we multiplied the total charges for each MS-DRG in each of the 19 cost centers by the corresponding national average CCR, we summed the 19 “costs” across each MS-DRG to produce a total standardized cost for the MS-DRG. The average standardized cost for each MS-DRG was then computed as the total standardized cost for the MS-DRG divided by the transfer-adjusted case count for the MS-DRG. The average cost for each MS-DRG was then divided by the national average standardized cost per case to determine the relative weight. The final FY 2025 cost-based relative weights were then normalized by an adjustment factor of 1.92336 so that the average case weight after recalibration was equal to the average case weight before recalibration. The normalization adjustment is intended to ensure that recalibration by itself neither increases nor decreases total payments under the IPPS, as required by section 1886(d)(4)(C)(iii) of the Act. We then applied the permanent 10-percent cap on the reduction in a MS-DRG's relative weight in a given fiscal year; specifically for those MS-DRGs for which the relative weight otherwise would have declined by more than 10 percent from the FY 2024 relative weight, we set the final FY 2025 relative weight equal to 90 percent of the FY 2024 relative weight. The final relative weights for FY 2025 as set forth in Table 5 associated with this final rule and available on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS reflect the application of this cap.
The 19 national average CCRs for FY 2025 are as follows:

Since FY 2009, the relative weights have been based on 100 percent cost weights based on our MS-DRG grouping system.
When we recalibrated the DRG weights for previous years, we set a threshold of 10 cases as the minimum number of cases required to compute a reasonable weight. We proposed to use that same case threshold in recalibrating the proposed MS-DRG relative weights for FY 2025. Using data from the FY 2023 MedPAR file, there were 8 MS-DRGs that contain fewer than 10 cases. For FY 2025, because we do not have sufficient MedPAR data to set accurate and stable cost relative weights for these low-volume MS-DRGs, we proposed to compute relative weights for the low-volume MS-DRGs by adjusting their final FY 2024 relative weights by the percentage change in the average weight of the cases in other MS-DRGs from FY 2024 to FY 2025. The crosswalk table is as follows.

Comment: A commenter requested that CMS consider if there are mechanisms to reform the role of CCRs in the reimbursement methodology to prevent differential hospital charge practices from skewing reimbursement rates for hospitals—such as either eliminating the role of CCRs or creating a bridge or other CCR for gene and cell therapies that it stated could be used for more accurate rate-setting in the future. Another commenter requested that CMS utilize the “other” CCR for CAR T-cell therapy product charges as a strategy to address charge compression starting in FY 2025 and until CMS proposes an alternative payment solution.
Response: We continue to believe it would not be appropriate to utilize the “other” CCR for CAR T-cell therapy product charges associated with revenue code 0891. Under our cost-based weight methodology, many revenue codes are mapped to each of the 19 cost centers. We believe that relative to those 19 cost centers, cellular therapies are most similar to drugs given that hospitals have generally calibrated their CAR T-cell therapy product charges to the “drugs” cost center CCR. To provide additional clarity, we have renamed the “drugs” cost center to the “drugs and cellular therapies” cost center. We may consider changes to the CCRs used for gene and cellular therapies in future rulemaking.
After consideration of the public comments we received, we are finalizing our proposals without modification.
Effective for discharges beginning on or after October 1, 2001, section 1886(d)(5)(K)(i) of the Act requires the Secretary to establish (after notice and opportunity for public comment) a mechanism to recognize the costs of new medical services and technologies (sometimes collectively referred to in this section as “new technologies”) under the IPPS. Section 1886(d)(5)(K)(vi) of the Act specifies that a medical service or technology will be considered new if it meets criteria established by the Secretary after notice and opportunity for public comment. Section 1886(d)(5)(K)(ii)(I) of the Act specifies that a new medical service or technology may be considered for new technology add-on payment if, based on the estimated costs incurred with respect to discharges involving such service or technology, the DRG prospective payment rate otherwise applicable to such discharges under this subsection is inadequate. The regulations at 42 CFR 412.87 implement these provisions and § 412.87(b) specifies three criteria for a new medical service or technology to receive the additional payment: (1) The medical service or technology must be new; (2) the medical service or technology must be costly such that the DRG rate otherwise applicable to discharges involving the medical service or technology is determined to be inadequate; and (3) the service or technology must demonstrate a substantial clinical improvement over existing services or technologies. In addition, certain transformative new devices and antimicrobial products may qualify under an alternative inpatient new technology add-on payment pathway, as set forth in the regulations at § 412.87(c) and (d).
We note that section 1886(d)(5)(K)(i) of the Act requires that the Secretary establish a mechanism to recognize the costs of new medical services and technologies under the payment system established under that subsection, which establishes the system for paying for the operating costs of inpatient hospital services. The system of payment for capital costs is established under section 1886(g) of the Act. Therefore, as discussed in prior rulemaking ( 72 FR 47307 through 47308 ), we do not include capital costs in the add-on payments for a new medical service or technology or make ( print page 69116) new technology add-on payments under the IPPS for capital-related costs.
In this rule, we highlight some of the major statutory and regulatory provisions relevant to the new technology add-on payment criteria, as well as other information. For further discussion on the new technology add-on payment criteria, we refer readers to the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51572 through 51574 ), the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42288 through 42300 ), and the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58736 through 58742 ).
Under the first criterion, as reflected in § 412.87(b)(2), a specific medical service or technology will no longer be considered “new” for purposes of new medical service or technology add-on payments after CMS has recalibrated the MS-DRGs, based on available data, to reflect the cost of the technology. We note that we do not consider a service or technology to be new if it is substantially similar to one or more existing technologies. That is, even if a medical product receives a new FDA approval or clearance, it may not necessarily be considered “new” for purposes of new technology add-on payments if it is “substantially similar” to another medical product that was approved or cleared by FDA and has been on the market for more than 2 to 3 years. In the FY 2010 IPPS/RY 2010 LTCH PPS final rule ( 74 FR 43813 through 43814 ), we established criteria for evaluating whether a new technology is substantially similar to an existing technology, specifically whether: (1) a product uses the same or a similar mechanism of action to achieve a therapeutic outcome; (2) a product is assigned to the same or a different MS-DRG; and (3) the new use of the technology involves the treatment of the same or similar type of disease and the same or similar patient population. If a technology meets all three of these criteria, it would be considered substantially similar to an existing technology and would not be considered “new” for purposes of new technology add-on payments. For a detailed discussion of the criteria for substantial similarity, we refer readers to the FY 2006 IPPS final rule ( 70 FR 47351 through 47352 ) and the FY 2010 IPPS/LTCH PPS final rule ( 74 FR 43813 through 43814 ).
Under the second criterion, § 412.87(b)(3) further provides that, to be eligible for the add-on payment for new medical services or technologies, the MS-DRG prospective payment rate otherwise applicable to discharges involving the new medical service or technology must be assessed for adequacy. Under the cost criterion, consistent with the formula specified in section 1886(d)(5)(K)(ii)(I) of the Act, to assess the adequacy of payment for a new technology paid under the applicable MS-DRG prospective payment rate, we evaluate whether the charges of the cases involving a new medical service or technology will exceed a threshold amount that is the lesser of 75 percent of the standardized amount (increased to reflect the difference between cost and charges) or 75 percent of one standard deviation beyond the geometric mean standardized charge for all cases in the MS-DRG to which the new medical service or technology is assigned (or the case-weighted average of all relevant MS-DRGs if the new medical service or technology occurs in many different MS-DRGs). The MS-DRG threshold amounts generally used in evaluating new technology add-on payment applications for FY 2025 are presented in a data file that is available, along with the other data files associated with the FY 2024 IPPS/LTCH PPS final rule and correction notification, on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index .
We note that, under the policy finalized in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58603 through 58605 ), beginning with FY 2022, we use the proposed threshold values associated with the proposed rule for that fiscal year to evaluate the cost criterion for all applications for new technology add-on payments and previously approved technologies that may continue to receive new technology add-on payments, if those technologies would be assigned to a proposed new MS-DRG for that same fiscal year.
As finalized in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41275 ), beginning with FY 2020, we include the thresholds applicable to the next fiscal year (previously included in Table 10 of the annual IPPS/LTCH PPS proposed and final rules) in the data files associated with the prior fiscal year. Accordingly, the final thresholds for applications for new technology add-on payments for FY 2026 are presented in a data file that is available on the CMS website, along with the other data files associated with this FY 2025 final rule, by clicking on the FY 2025 IPPS Final Rule Home Page at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index .
In the September 7, 2001 final rule that established the new technology add-on payment regulations ( 66 FR 46917 ), we discussed that applicants should submit a significant sample of data to demonstrate that the medical service or technology meets the high-cost threshold. Specifically, applicants should submit a sample of sufficient size to enable us to undertake an initial validation and analysis of the data. We also discussed in the September 7, 2001 final rule ( 66 FR 46917 ) the issue of whether the Health Insurance Portability and Accountability Act (HIPAA) Privacy Rule at 45 CFR parts 160 and 164, subparts A and E, applies to claims information that providers submit with applications for new medical service or technology add-on payments. We refer readers to the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51573 ) for further information on this issue.
Under the third criterion at § 412.87(b)(1), a medical service or technology must represent an advance that substantially improves, relative to technologies previously available, the diagnosis or treatment of Medicare beneficiaries. In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42288 through 42292 ), we prospectively codified in our regulations at § 412.87(b) the following aspects of how we evaluate substantial clinical improvement for purposes of new technology add-on payments under the IPPS:
- The totality of the circumstances is considered when making a determination that a new medical service or technology represents an advance that substantially improves, relative to services or technologies previously available, the diagnosis or treatment of Medicare beneficiaries.
- A determination that a new medical service or technology represents an advance that substantially improves, relative to services or technologies previously available, the diagnosis or treatment of Medicare beneficiaries means—
++ The new medical service or technology offers a treatment option for a patient population unresponsive to, or ineligible for, currently available treatments; ( print page 69117)
++ The new medical service or technology offers the ability to diagnose a medical condition in a patient population where that medical condition is currently undetectable, or offers the ability to diagnose a medical condition earlier in a patient population than allowed by currently available methods, and there must also be evidence that use of the new medical service or technology to make a diagnosis affects the management of the patient.
++ The use of the new medical service or technology significantly improves clinical outcomes relative to services or technologies previously available as demonstrated by one or more of the following: a reduction in at least one clinically significant adverse event, including a reduction in mortality or a clinically significant complication; a decreased rate of at least one subsequent diagnostic or therapeutic intervention; a decreased number of future hospitalizations or physician visits; a more rapid beneficial resolution of the disease process treatment including, but not limited to, a reduced length of stay or recovery time; an improvement in one or more activities of daily living; an improved quality of life; or, a demonstrated greater medication adherence or compliance; or
++ The totality of the circumstances otherwise demonstrates that the new medical service or technology substantially improves, relative to technologies previously available, the diagnosis or treatment of Medicare beneficiaries.
- Evidence from the following published or unpublished information sources from within the United States or elsewhere may be sufficient to establish that a new medical service or technology represents an advance that substantially improves, relative to services or technologies previously available, the diagnosis or treatment of Medicare beneficiaries: clinical trials, peer reviewed journal articles; study results; meta-analyses; consensus statements; white papers; patient surveys; case studies; reports; systematic literature reviews; letters from major healthcare associations; editorials and letters to the editor; and public comments. Other appropriate information sources may be considered.
- The medical condition diagnosed or treated by the new medical service or technology may have a low prevalence among Medicare beneficiaries.
- The new medical service or technology may represent an advance that substantially improves, relative to services or technologies previously available, the diagnosis or treatment of a subpopulation of patients with the medical condition diagnosed or treated by the new medical service or technology.
We refer the reader to the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42288 through 42292 ) for additional discussion of the evaluation of substantial clinical improvement for purposes of new technology add-on payments under the IPPS.
We note, consistent with the discussion in the FY 2003 IPPS final rule ( 67 FR 50015 ), that while FDA has regulatory responsibility for decisions related to marketing authorization (for example, approval, clearance, etc.), we do not rely upon FDA criteria in our evaluation of substantial clinical improvement for purposes of determining what services and technologies qualify for new technology add-on payments under Medicare. This criterion does not depend on the standard of safety and effectiveness on which FDA relies but on a demonstration of substantial clinical improvement in the Medicare population.
Beginning with applications for FY 2021 new technology add-on payments, under the regulations at § 412.87(c), a medical device that is part of FDA's Breakthrough Devices Program may qualify for the new technology add-on payment under an alternative pathway. Additionally, under the regulations at § 412.87(d) for certain antimicrobial products, beginning with FY 2021, a drug that is designated by FDA as a Qualified Infectious Disease Product (QIDP), and, beginning with FY 2022, a drug that is approved by FDA under the Limited Population Pathway for Antibacterial and Antifungal Drugs (LPAD), may also qualify for the new technology add-on payment under an alternative pathway. We refer the reader to the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42292 through 42297 ) and the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58737 through 58739 ) for further discussion on this policy. We note that CMS reviews the application based on the information provided by the applicant only under the alternative pathway specified by the applicant at the time of application submission. To receive approval for the new technology add-on payment under that alternative pathway, the technology must have the applicable FDA designation and meet all other requirements in the regulations in § 412.87(c) and (d), as applicable.
For applications received for new technology add-on payments for FY 2021 and subsequent fiscal years, a medical device designated under FDA's Breakthrough Devices Program that has received FDA marketing authorization will be considered not substantially similar to an existing technology for purposes of the new technology add-on payment under the IPPS, and will not need to meet the requirement under § 412.87(b)(1) that it represent an advance that substantially improves, relative to technologies previously available, the diagnosis or treatment of Medicare beneficiaries. Under this alternative pathway, a medical device that has received FDA marketing authorization (that is, has been approved or cleared by, or had a De Novo classification request granted by, FDA) as a Breakthrough Device, for the indication covered by the Breakthrough Device designation, will need to meet the requirements of § 412.87(c). We note that in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58734 through 58736 ), we clarified our policy that a new medical device under this alternative pathway must receive marketing authorization for the indication covered by the Breakthrough Devices Program designation. We refer the reader to the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58734 through 58736 ) for further discussion regarding this clarification.
For applications received for new technology add-on payments for certain antimicrobial products, beginning with FY 2021, if a technology is designated by FDA as a QIDP and received FDA marketing authorization, and, beginning with FY 2022, if a drug is approved under FDA's LPAD pathway and used for the indication approved under the LPAD pathway, it will be considered not substantially similar to an existing technology for purposes of new technology add-on payments and will not need to meet the requirement that it represent an advance that substantially improves, relative to technologies previously available, the diagnosis or treatment of Medicare beneficiaries. Under this alternative pathway for QIDPs and LPADs, a medical product that has received FDA marketing authorization and is designated by FDA as a QIDP or approved under the LPAD pathway will need to meet the requirements of § 412.87(d). We refer the reader to the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42292 through ( print page 69118) 42297) and FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58737 through 58739 ) for further discussion on this policy.
We note that, in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58737 through 58739 ), we clarified that a new medical product seeking approval for the new technology add-on payment under the alternative pathway for QIDPs must receive FDA marketing authorization for the indication covered by the QIDP designation. We also finalized our policy to expand our alternative new technology add-on payment pathway for certain antimicrobial products to include products approved under the LPAD pathway and used for the indication approved under the LPAD pathway.
The new medical service or technology add-on payment policy under the IPPS provides additional payments for cases with relatively high costs involving eligible new medical services or technologies, while preserving some of the incentives inherent under an average-based prospective payment system. The payment mechanism is based on the cost to hospitals for the new medical service or technology. As noted previously, we do not include capital costs in the add-on payments for a new medical service or technology or make new technology add-on payments under the IPPS for capital-related costs ( 72 FR 47307 through 47308 ).
For discharges occurring before October 1, 2019, under § 412.88, if the costs of the discharge (determined by applying operating cost-to-charge ratios (CCRs) as described in § 412.84(h)) exceed the full DRG payment (including payments for IME and DSH, but excluding outlier payments), CMS made an add-on payment equal to the lesser of: (1) 50 percent of the costs of the new medical service or technology; or (2) 50 percent of the amount by which the costs of the case exceed the standard DRG payment.
Beginning with discharges on or after October 1, 2019, for the reasons discussed in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42297 through 42300 ), we finalized an increase in the new technology add-on payment percentage, as reflected at § 412.88(a)(2)(ii). Specifically, for a new technology other than a medical product designated by FDA as a QIDP, beginning with discharges on or after October 1, 2019, if the costs of a discharge involving a new technology (determined by applying CCRs as described in § 412.84(h)) exceed the full DRG payment (including payments for IME and DSH, but excluding outlier payments), Medicare will make an add-on payment equal to the lesser of: (1) 65 percent of the costs of the new medical service or technology; or (2) 65 percent of the amount by which the costs of the case exceed the standard DRG payment. For a new technology that is a medical product designated by FDA as a QIDP, beginning with discharges on or after October 1, 2019, if the costs of a discharge involving a new technology (determined by applying CCRs as described in § 412.84(h)) exceed the full DRG payment (including payments for IME and DSH, but excluding outlier payments), Medicare will make an add-on payment equal to the lesser of: (1) 75 percent of the costs of the new medical service or technology; or (2) 75 percent of the amount by which the costs of the case exceed the standard DRG payment. For a new technology that is a medical product approved under FDA's LPAD pathway, beginning with discharges on or after October 1, 2020, if the costs of a discharge involving a new technology (determined by applying CCRs as described in § 412.84(h)) exceed the full DRG payment (including payments for IME and DSH, but excluding outlier payments), Medicare will make an add-on payment equal to the lesser of: (1) 75 percent of the costs of the new medical service or technology; or (2) 75 percent of the amount by which the costs of the case exceed the standard DRG payment. As set forth in § 412.88(b)(2), unless the discharge qualifies for an outlier payment, the additional Medicare payment will be limited to the full MS-DRG payment plus 65 percent (or 75 percent for certain antimicrobial products (QIDPs and LPADs)) of the estimated costs of the new technology or medical service. We refer the reader to the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42297 through 42300 ) for further discussion on the increase in the new technology add-on payment beginning with discharges on or after October 1, 2019.
As discussed further in section II.E.10. of this final rule, we are finalizing our proposal that for certain gene therapies approved for new technology add-on payments in the FY 2025 IPPS/LTCH PPS final rule that are indicated and used specifically for the treatment of SCD, effective with discharges on or after October 1, 2024 and concluding at the end of the 2- to 3-year newness period for such therapy, if the costs of a discharge (determined by applying CCRs as described in § 412.84(h)) involving the use of such therapy for the treatment of SCD exceed the full DRG payment (including payments for IME and DSH, but excluding outlier payments), Medicare will make an add-on payment equal to the lesser of: (1) 75 percent of the costs of the new medical service or technology; or (2) 75 percent of the amount by which the costs of the case exceed the standard DRG payment. We note that these payment amounts would only apply to Casgevy TM (exagamglogene autotemcel) and Lyfgenia TM (lovotibeglogene autotemcel), when indicated and used specifically for the treatment of SCD, which CMS has determined in this FY 2025 IPPS/LTCH PPS final rule meet the criteria for approval for new technology add-on payment, as further discussed in section II.E.5. of this final rule.
We note that, consistent with the prospective nature of the IPPS, we finalize the new technology add on payment amount for technologies approved or conditionally approved for new technology add-on payments in the final rule for each fiscal year and do not make mid-year changes to new technology add-on payment amounts. Updated cost information may be submitted and included in rulemaking to be considered for the following fiscal year.
Section 503(d)(2) of the MMA ( Pub. L. 108-173 ) provides that there shall be no reduction or adjustment in aggregate payments under the IPPS due to add-on payments for new medical services and technologies. Therefore, in accordance with section 503(d)(2) of the MMA, add-on payments for new medical services or technologies for FY 2005 and subsequent years have not been subjected to budget neutrality.
In the FY 2009 IPPS final rule ( 73 FR 48561 through 48563 ), we modified our regulation at § 412.87 to codify our longstanding practice of how CMS evaluates the eligibility criteria for new medical service or technology add-on payment applications. That is, we first determine whether a medical service or technology meets the newness criterion, and only if so, do we then make a determination as to whether the technology meets the cost threshold and represents a substantial clinical improvement over existing medical services or technologies. We specified that all applicants for new technology add-on payments must have FDA approval or clearance by July 1 of the year prior to the beginning of the fiscal year for which the application is being considered. In the FY 2021 IPPS/LTCH ( print page 69119) PPS final rule, to more precisely describe the various types of FDA approvals, clearances and classifications that we consider under our new technology add-on payment policy, we finalized a technical clarification to the regulation to indicate that new technologies must receive FDA marketing authorization (such as pre-market approval (PMA); 510(k) clearance; the granting of a De Novo classification request, or approval of a New Drug Application (NDA)) by July 1 of the year prior to the beginning of the fiscal year for which the application is being considered. Consistent with our longstanding policy, we consider FDA marketing authorization as representing that a product has received FDA approval or clearance when considering eligibility for the new technology add-on payment ( 85 FR 58742 ).
Additionally, in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58739 through 58742 ), we finalized our proposal to provide conditional approval for new technology add-on payment for a technology for which an application is submitted under the alternative pathway for certain antimicrobial products at § 412.87(d) that does not receive FDA marketing authorization by July 1 prior to the particular fiscal year for which the applicant applied for new technology add-on payments, provided that the technology otherwise meets the applicable add-on payment criteria. Under this policy, cases involving eligible antimicrobial products would begin receiving the new technology add-on payment sooner, effective for discharges the quarter after the date of FDA marketing authorization, provided that the technology receives FDA marketing authorization before July 1 of the fiscal year for which the applicant applied for new technology add-on payments.
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ), we finalized that, beginning with the new technology add-on payment applications for FY 2025, for technologies that are not already FDA market authorized for the indication that is the subject of the new technology add-on payment application, applicants must have a complete and active FDA market authorization request at the time of new technology add-on payment application submission and must provide documentation of FDA acceptance or filing to CMS at the time of application submission, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. See § 412.87(e) and further discussion in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ). We also finalized that, beginning with FY 2025 applications, in order to be eligible for consideration for the new technology add-on payment for the upcoming fiscal year, an applicant for new technology add-on payments must have received FDA approval or clearance by May 1 (rather than July 1) of the year prior to the beginning of the fiscal year for which the application is being considered (except for an application that is submitted under the alternative pathway for certain antimicrobial products), as reflected at §§ 412.87(f)(2) and (f)(3), as amended and redesignated in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 , 88 FR 59331 ).
Many interested parties (including device/biologic/drug developers or manufacturers, industry consultants, others) engage CMS for coverage, coding, and payment questions or concerns. In order to streamline engagement by centralizing the different innovation pathways within CMS including new technology add-on payments, CMS has established a team of new technology liaisons that can serve as an initial resource for interested parties. This team is available to assist with all of the following:
- Help to point interested parties to or provide information and resources where possible regarding process, requirements, and timelines.
- Coordinate and facilitate opportunities for interested parties to engage with various CMS components.
- Serve as a primary point of contact for interested parties and provide updates on developments where possible or appropriate.
We receive many questions from parties interested in pursuing new technology add-on payments who may not be entirely familiar with working with CMS. While we encourage interested parties to first review our resources available at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/newtech , we know that there may be additional questions about the application process. Interested parties with further questions regarding Medicare's coverage, coding, and payment processes, and how they can navigate these processes, whether for new technology add-on payments or otherwise, should review the updated resource guide available at: https://www.cms.gov/medicare/coding-billing/guide-medical-technology-companies-other-interested-parties . Parties that would like to further discuss questions or concerns with CMS should contact the new technology liaison team at [email protected] .
Applicants for add-on payments for new medical services or technologies for FY 2026 must submit a formal request, including a full description of the clinical applications of the medical service or technology and the results of any clinical evaluations demonstrating that the new medical service or technology represents a substantial clinical improvement (unless the application is under one of the alternative pathways as previously described), along with a significant sample of data to demonstrate that the medical service or technology meets the high-cost threshold. CMS will review the application based on the information provided by the applicant under the pathway specified by the applicant at the time of application submission. Complete application information, along with final deadlines for submitting a full application, will be posted as it becomes available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/newtech.html .
To allow interested parties to identify the new medical services or technologies under review before the publication of the proposed rule for FY 2026, once the application deadline has closed, CMS will post on its website a list of the applications submitted, along with a brief description of each technology as provided by the applicant.
As discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48986 through 48990 ), we finalized our proposal to publicly post online new technology add-on payment.
Applications, including the completed application forms, certain related materials, and any additional updated application information submitted subsequent to the initial application submission (except certain volume, cost and other information identified by the applicant as confidential), beginning with the application cycle for FY 2024, at the time the proposed rule is published. We also finalized that with the exception of information included in a confidential information section of the application, cost and volume information, and materials identified by the applicant as copyrighted and/or not otherwise releasable to the public, the contents of the application and related materials may be posted publicly, and that we ( print page 69120) will not post applications that are withdrawn prior to publication of the proposed rule. We refer the reader to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48986 through 48990 ) for further information regarding this policy.
We note that the burden associated with this information collection requirement is the time and effort required to collect and submit the data in the formal request for add-on payments for new medical services and technologies to CMS. The aforementioned burden is subject to the PRA and approved under OMB control number 0938-1347 and has an expiration date of December 31, 2026.
Section 1886(d)(5)(K)(viii) of the Act, as amended by section 503(b)(2) of the MMA, provides for a mechanism for public input before publication of a notice of proposed rulemaking regarding whether a medical service or technology represents a substantial clinical improvement. The process for evaluating new medical service and technology applications requires the Secretary to do all of the following:
- Provide, before publication of a proposed rule, for public input regarding whether a new service or technology represents an advance in medical technology that substantially improves the diagnosis or treatment of Medicare beneficiaries.
- Make public and periodically update a list of the services and technologies for which applications for add-on payments are pending.
- Accept comments, recommendations, and data from the public regarding whether a service or technology represents a substantial clinical improvement.
- Provide, before publication of a proposed rule, for a meeting at which organizations representing hospitals, physicians, manufacturers, and any other interested party may present comments, recommendations, and data regarding whether a new medical service or technology represents a substantial clinical improvement to the clinical staff of CMS.
In order to provide an opportunity for public input regarding add-on payments for new medical services and technologies for FY 2025 prior to publication of the FY 2025 IPPS/LTCH PPS proposed rule, we published a notice in the Federal Register on September 28, 2023 ( 88 FR 66850 ) and held a virtual town hall meeting on December 13, 2023. In the announcement notice for the meeting, we stated that the opinions and presentations provided during the meeting would assist us in our evaluations of applications by allowing public discussion of the substantial clinical improvement criterion for the FY 2025 new medical service and technology add-on payment applications before the publication of the FY 2025 IPPS/LTCH IPPS proposed rule.
Approximately 130 individuals registered to attend the virtual town hall meeting. We posted the recordings of the virtual town hall on the CMS web page at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/newtech . We considered each applicant's presentation made at the town hall meeting, as well as written comments received by the December 18, 2023 deadline, in our evaluation of the new technology add-on payment applications for FY 2025 in the development of the FY 2025 IPPS/LTCH PPS proposed rule. In response to the published notice and the December 13, 2023 New Technology Town Hall meeting, we received written comments regarding the applications for FY 2025 new technology add on payments. As explained earlier and in the Federal Register notice announcing the New Technology Town Hall meeting ( 88 FR 66850 through 66853 ), the purpose of the meeting was specifically to discuss the substantial clinical improvement criterion with regard to pending new technology add-on payment applications for FY 2025. Therefore, we did not summarize any written comments in the proposed rule that were unrelated to the substantial clinical improvement criterion. In section II.E.5. of the preamble of the proposed rule, we summarized comments regarding individual applications, or, if applicable, indicated that there were no comments received in response to the New Technology Town Hall meeting notice or New Technology Town Hall meeting, at the end of each discussion of the individual applications.
As discussed in the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49434 ), the ICD-10-PCS includes a new section containing the new Section “X” codes, which began being used with discharges occurring on or after October 1, 2015. Decisions regarding changes to ICD-10-PCS Section “X” codes will be handled in the same manner as the decisions for all of the other ICD-10-PCS code changes. That is, proposals to create, delete, or revise Section “X” codes under the ICD-10-PCS structure will be referred to the ICD-10 Coordination and Maintenance Committee. In addition, several of the new medical services and technologies that have been, or may be, approved for new technology add-on payments may now, and in the future, be assigned a Section “X” code within the structure of the ICD-10-PCS. We posted ICD-10-PCS Guidelines on the CMS website at: https://www.cms.gov/Medicare/Coding/ICD10 , including guidelines for ICD-10-PCS Section “X” codes. We encourage providers to view the material provided on ICD-10-PCS Section “X” codes.
In this section of the final rule, we discuss the FY 2025 status of 31 technologies approved for FY 2024 new technology add-on payments, as set forth in the tables that follow. In the proposed rule, we presented our proposals to continue the new technology add-on payments for FY 2025 for those technologies that were approved for the new technology add-on payment for FY 2024, and which would still be considered “new” for purposes of new technology add-on payments for FY 2025. We also presented our proposals to discontinue new technology add-on payments for FY 2025 for those technologies that were approved for the new technology add-on payment for FY 2024, and which would no longer be considered “new” for purposes of new technology add-on payments for FY 2025.
Additionally, we noted that we conditionally approved DefenCath® (taurolidine/heparin) for FY 2024 new technology add-on payments under the alternative pathway for certain antimicrobial products ( 88 FR 58942 through 58944 ), subject to the technology receiving FDA marketing authorization by July 1, 2024. DefenCath® (taurolidine/heparin) received FDA marketing authorization on November 15, 2023, and was eligible to receive new technology add-on payments in FY 2024 beginning with discharges on or after January 1, 2024. As DefenCath® (taurolidine/heparin) received FDA marketing authorization prior to July 1, 2024, and was approved for new technology add-on payments in FY 2024, we proposed and are finalizing to continue making new technology add-on payments for DefenCath® for FY 2025.
Our policy is that a medical service or technology may continue to be ( print page 69121) considered “new” for purposes of new technology add-on payments within 2 or 3 years after the point at which data begin to become available reflecting the inpatient hospital code assigned to the new service or technology. Our practice has been to begin and end new technology add-on payments on the basis of a fiscal year, and we have generally followed a guideline that uses a 6-month window before and after the start of the fiscal year to determine whether to extend the new technology add-on payment for an additional fiscal year. In general, we extend new technology add-on payments for an additional year only if the 3-year anniversary date of the product's entry onto the U.S. market occurs in the latter half of the fiscal year ( 70 FR 47362 ).
In the proposed rule, we provided a table listing the technologies for which we proposed to continue making new technology add-on payments for FY 2025 because they were still considered “new” for purposes of new technology add-on payments. This table also presented the newness start date, new technology add-on payment start date, 3-year anniversary date of the product's entry onto the U.S. market, relevant final rule citations from prior fiscal years, proposed maximum add-on payment amount, and coding assignments for each technology. We referred readers to the cited final rules in the following table for a complete discussion of the new technology add-on payment application, coding, and payment amount for these technologies, including the applicable indications and discussion of the newness start date.
We invited public comments on our proposals to continue new technology add-on payments for FY 2025 for the technologies listed in Table II.E.-01 of the proposed rule.
Comment: We received multiple comments in support of our proposed continuation of new technology add-on payments for FY 2025 for those technologies that were approved for the new technology add-on payment for FY 2024, and which would still be considered “new” for purposes of new technology add-on payments for FY 2025.
Response: We appreciate the support of the commenters.
Comment: A commenter restated a comment it made in response to previous proposed rules that requiring a manufacturer to submit information rebutting a presumption that the date of first availability is the date of FDA marketing authorization adds unnecessary burden and complexity to the new technology add-on payment application and review process. The commenter further stated that CMS did not appear to have applied this policy consistently and that it has defaulted to the FDA approval date despite other reasons being provided by applicants regarding the first date of commercial availability. The commenter believed that a more efficient and appropriate policy would be for the newness period to begin with the date of the first claim, which it stated is consistent with the definition of newness used in determining the period of eligibility for Transitional Pass-through status in the Hospital Outpatient Prospective Payment System (OPPS).
Response: We thank the commenter for its feedback. As we discussed in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45136 ), regarding the commenter's belief that beginning the newness period on the date of first claim would be a more efficient and appropriate policy and is consistent with the definition of newness used in determining the period of eligibility for Transitional Pass-through status in OPPS, we note that “newness” for the purposes of the OPPS pass-through payment is separate and distinct from “newness” for the purposes of the IPPS new technology add-on payment. We note that “newness” for purposes of the OPPS pass-through payment refers to a drug, biological, or device's eligibility for pass-through payment status. In particular, under § 419.64(a), for a drug or biological's eligibility for OPPS pass-through payment (subject to certain exceptions), “newness” means that the drug or biological was first payable as an outpatient hospital service after December 31, 1996. Under § 419.66(b), for a device's eligibility for OPPS pass-through payment, “newness” means that CMS received the applicant's pass-through application within 3 years from the date of the initial FDA marketing authorization, unless there is a documented, verifiable delay in U.S. market availability after FDA marketing authorization is granted, in which case CMS will consider the pass-through payment application if it is submitted within 3 years from the date of market availability for the device. However, it appears the commenter is referring not to “newness” in terms of eligibility for OPPS pass-through status, but rather to the limited two-to-three-year period of pass-through payment. Under §§ 419.64(c)(2) and 419.66(g), this pass-through payment period begins on the date on which CMS makes its first pass-through payment for a drug, biological, or device.
For new technology add-on payments, as we have discussed in prior rulemaking ( 77 FR 53348 ), generally, our policy is to begin the newness period on the date of FDA approval or clearance or, if later, the date of availability of the technology on the U.S. market. Furthermore, as we have stated in prior rulemaking, the newness period does not necessarily start with the approval date for the medical service or technology. Instead, it begins with availability of the technology on the market, which is when data become available. We have consistently applied this standard, and believe that it is most consistent with the purpose of new technology add-on payments ( 69 FR 49003 ), because section 1886(d)(5)(K)(ii)(II) of the Act requires CMS to establish a mechanism to provide for the collection of data with respect to the costs of a new medical service or technology for a period of not less than two years and not more than three years beginning on the date on which an inpatient hospital code is issued for the service or technology. Our regulations at § 412.87(b)(2), 412.87(c)(2), and 412.87(d)(2) further allow new medical services and technologies to be considered new for the first 2 to 3 years after the point at which data begin to become available reflecting the inpatient hospital code assigned to the new service or technology, which is during the period when the costs of the new technology are not yet fully reflected in the DRG weights. The costs of the new medical service or technology, once paid for by Medicare for this 2-year to 3-year period, are accounted for in the MedPAR data that are used to recalibrate the DRG weights on an annual basis. Therefore, it is appropriate to limit the add-on payment window for those technologies to this 2-to 3-year timeframe. For these reasons, we continue to disagree that the appropriate policy would be for the newness period to begin with the date of the first claim.
Comment: The applicant for REZZAYO TM (rezafungin for injection), submitted a comment regarding its newness start date of March 22, 2023, to explain why REZZAYO TM was not available on the US market until July 26, 2023. The applicant explained that the market entry for REZZAYO TM was delayed due to the steps needed to comply with FDA requirements. The applicant stated that REZZAYO TM received FDA approval on March 22, 2023, and that the product was subjected to a post marketing commitment (PMC) protocol. According to the applicant, the PMC stated that the manufacturer would complete necessary qualification and validation studies of the current assay high-performance ( print page 69122) liquid chromatography analytical procedure to be used for the gross content and assay of reconstituted solution tests in the drug product specification, and update the relevant sections of Module 3 accordingly. The applicant stated that FDA required this information be submitted to FDA via a Changes Being Effected in 0 Days Supplement (CBE-0). The applicant stated that to meet the requirements of the PMC and prepare the CBE-0 for submission, the manufacturer was unable to use anything more than a nominal amount of existing batches of product due to vial size differentials, which meant the manufacturer needed to manufacture new product for its analyses pursuant to the PMC, and that the applicant had to ensure that new product that met these requirements was created prior to launch. The applicant explained that due to the PMC requirements, REZZAYO TM needed to undergo an additional manufacturing cycle prior to launch, and the changes in vial size required changes to REZZAYO's labeling and packaging. The applicant stated that label and packaging changes alone can take an additional six weeks. The applicant stated that the manufacturer was able to complete the PMC requirements and submitted the CBE-0 on July 19, 2023, and that REZZAYO TM was made available for sale to hospitals following the first sale and shipment to a wholesaler on July 26, 2023, as reflected in the Medicaid Drug Rebate Program database.
Response: We thank the applicant for its comment. As stated previously, while CMS may consider a documented delay in the technology's market availability in determining when the newness period begins, our policy for determining whether to extend new technology add-on payments for an additional year generally applies regardless of the volume of claims for the technology after the beginning of the newness period ( 83 FR 41280 ). We do not consider the date of first sale of a product as an indicator of the entry of a product onto the U.S. market. Although the applicant states that REZZAYO TM was made available for sale to hospitals following the first sale and shipment to a wholesaler on July 26, 2023, it is unclear from the information provided if the technology may have first became available on the market between the date of completion of the PMC and submission of the CBE-0 on July 19, 2023, and its first sale on July 26, 2023, as an applicant may commence distribution of a drug product manufactured using a change proposed in a CBE-0 supplement after FDA receives that supplement. [ 22 ] Therefore, based on the information provided by the applicant regarding the documented delay in the technology's availability on the U.S. market, and absent additional information from the applicant, we consider the beginning of the newness period to commence on July 19, 2023.
Comment: We received multiple comments in support of our proposed continuation of new technology add-on payments for FY 2025 for the SAINT Neuromodulation System. A couple of commenters described their experiences and timelines for installation, training, and use of the SAINT Neuromodulation System at their hospitals. Commenters also supported modification of the technology's newness date to April 5, 2024, to recognize the delay in commercial availability.
In particular, the applicant for the SAINT Neuromodulation System submitted a comment to provide an update on its launch timeline and the commercial availability of technology in the provider market. The applicant confirmed that the SAINT Neuromodulation System is currently launching in the United States, and requested that CMS assign a newness date of April 5, 2024. The applicant stated that although SAINT received FDA clearance on September 1, 2022, there were significant product development, manufacturing design, and compliance steps that the company needed to complete before the device could become commercially available and be used to treat patients. It stated that initially, it had planned to develop and manufacture its own hardware; however, after much time and effort, it was determined in the second half of 2023 that the best course was to work with third-party manufacturers for the stimulator and neuronavigation hardware. The applicant provided a summary and timeline of all the activities that it completed prior to the device becoming commercially available to treat patients on April 5, 2024. The timeline also included information regarding the installation, training, and use of the SAINT Neuromodulation System at two hospitals after April 5, 2024.
Response: We thank the applicant and commenters for their comments. In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58937 through 58938 ), we noted that the applicant stated that ICD-10-PCS code X0Z0X18 (Computer-assisted transcranial magnetic stimulation of prefrontal cortex, new technology group 8) may be used to uniquely describe procedures involving the use of the SAINT Neuromodulation System, effective October 1, 2022. We note that between October 1, 2022 and April 4, 2024 (inclusive), we identified 5 claims reporting this ICD-10-PCS code that were associated with an acute care hospital under the IPPS. Three of those claims were made in FY 2024, and all 3 received new technology add-on payment. Therefore, based on our review of the data, we cannot determine a newness date based on a documented delay in the technology's availability on the U.S. market. We continue to consider the beginning of the newness period to commence on September 1, 2022, the date of FDA marketing authorization for the indication covered by its Breakthrough Device designation.
Comment: The applicant for DefenCath® (taurolidine/heparin), submitted a comment in support of our proposed continuation of new technology add-on payments for FY 2025 for DefenCath® and to update its Wholesale Acquisition Cost (WAC). The applicant noted that DefenCath® received conditional new technology add-on payment approval for FY 2024. The applicant stated that DefenCath® was approved by the FDA on November 15, 2023, via the Limited Population Pathway for Antibacterial and Antifungal Drugs (LPAD pathway), and that as such, hospitals were eligible to receive new technology add-on payments for DefenCath® as of January 1, 2024, which was the first date of the first quarter post FDA approval. The applicant stated that its original application included a WAC price of $390 per mL to determine reimbursement, which it stated was based upon market conditions at the time of the submission of the application. The applicant explained that after the submission of its application for FY 2024, and following FDA approval of DefenCath®, it performed additional market research and pricing analysis, and decided to launch with a WAC price that is significantly lower than what was originally submitted. The applicant stated that it launched DefenCath® on April 15, 2024, in the inpatient setting with the WAC price of $249.99 per 3mL vial ($83.33 per mL), and urged CMS to finalize its proposal to continue making new technology add-on payments in FY 2025 for DefenCath®.
Another commenter also submitted a comment requesting that CMS reassess the new technology add-on payment ( print page 69123) amount for DefenCath® based on the WAC price of $249.99 per 3mL vial, and expressed its concern about the impact DefenCath® will have on the Medicare program. The commenter stated that DefenCath® was late to the market with a clinical study using a control group which did not represent the current standard of care, and that it does not improve patient care or outcomes beyond the commenter's product, ClearGuard TM HD Antimicrobial Barrier Caps. The commenter also stated that DefenCath® requires additional labor resources to implement. Therefore, the commenter recommended that CMS monitor the value associated with DefenCath®.
Response: We thank the applicant and commenter for their comments and the updated cost information and recommendations. We have updated the new technology add-on payment amount for DefenCath® accordingly.
Although the applicant states that DefenCath® was launched on April 15, 2024, we did not receive information regarding a documented delay in market availability, and absent additional information from the applicant, we cannot determine a newness date based on a documented delay in the technology's availability on the U.S. market. Therefore, we continue to consider the beginning of the newness period to commence on November 15, 2023, the date of FDA marketing authorization for the indication covered by its QIDP designation.
DefenCath®'s current new technology add-on payment amount is $17,111.25, based on a WAC of $1,170 per 3mL vial. As we noted in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58943 ), on average, patients would receive 9.75 HD treatments per inpatient stay based upon the average length of stay of 13.3 days, which would require 19.5 vials. For FY 2025, the maximum new technology add-on payment amount is $3,656.10, based on an updated WAC of $249.99 per 3mL vial, as reflected in Table II.E.-01 in this final rule.
We further note that, as discussed in section II.E.5.d. of this final rule, because ELREXFIO TM and TALVEY TM are substantially similar to TECVAYLI®, we are using a single cost for purposes of determining the new technology add-on payment amount for ELREXFIO TM , TALVEY TM , and TECVAYLI® for FY 2025. As discussed in section II.E.5.d. of this final rule, we determined a weighted average of the cost of ELREXFIO TM , TALVEY TM , and TECVAYLI® based upon the projected numbers of cases involving each technology to determine the maximum new technology add-on payment. To compute the weighted average cost, we summed the total number of projected cases for each technology provided by the applicants, which equaled 4,376 cases (152 cases for ELREXFIO TM plus 2,318 cases for TALVEY TM plus 1,906 cases for TECVAYLI®). We then divided the number of projected cases for each of the technologies by the total number of cases, which resulted in the following case weighted percentages: 3.47 percent for ELREXFIO TM , 52.97 percent for TALVEY TM and 43.56 percent for TECVAYLI®. For each technology, we then multiplied the estimated cost per patient by the case-weighted percentage (0.0347 * $15,112 = $524.39 for ELREXFIO TM , 0.5297 * $25,164.44 = $13,329.60 for TALVEY TM and 0.4356 * $13,754.67 = $5,991.53 for TECVAYLI®). This resulted in a case-weighted average cost of $19,845.52 for the technology.
Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, the maximum new technology add-on payment for a case involving the use of ELREXFIO TM , TALVEY TM , or TECVAYLI® is $12,899.59 for FY 2025, as reflected in Table II.E.-01 of this final rule.
After consideration of the public comments we received, we are finalizing our proposals to continue new technology add-on payments for FY 2025 for the technologies that were approved for new technology add-on payment for FY 2024 and would still be considered “new” for purposes of new technology add-on payments for FY 2025, as listed in the proposed rule and in the following Table II.E.-01 in this section of this final rule.
We note that the following Table II.E.-01 is the same as Table II.E.-01 that was presented in the proposed rule, but Table II.E.-01 in this final rule includes the updated cost information for TECVAYLI® and DefenCath® and the updated newness start date for REZZAYO TM , as discussed previously. Table II.E.-01 in this final rule also presents the newness start date, new technology add-on payment start date, 3-year anniversary date of the product's entry onto the U.S. market, relevant final rule citations from prior fiscal years, maximum add-on payment amount, and coding assignments for each technology. We refer readers to the final rules cited in the following table for a complete discussion of the new technology add-on payment application, coding, and payment amount for these technologies, including the applicable indications and discussion of the newness start date.

In the proposed rule, we provided Table II.E.-02 listing the technologies for which we proposed to discontinue making new technology add-on ( print page 69126) payments for FY 2025 because they were no longer “new” for purposes of new technology add-on payments. This table also presented the newness start date, new technology add-on payment start date, the 3-year anniversary date of the product's entry onto the U.S. market, and relevant final rule citations from prior fiscal years. We referred readers to the cited final rules in the following table for a complete discussion of each new technology add-on payment application and the coding and payment amount for these technologies, including the applicable indications and discussion of the newness start date.
We invited public comments on our proposals to discontinue new technology add-on payments for FY 2025 for the technologies listed in Table II.E.-02 of the proposed rule.
Comment: A commenter urged CMS to prevent new access hurdles from arising with newer treatments by continuing new technology add-on payments that are now in place for low volume inpatient stays until the MS-DRG calculations reflect the cost of the treatment, as the commenter asserted that is what the new technology add-on payment mechanism was intended to do.
Response: As we have stated previously, our policy for determining whether to extend new technology add-on payments for an additional year generally applies regardless of the volume of claims for the technology after the beginning of the newness period. We do not believe that case volume is a relevant consideration for making the determination as to whether a product is considered “new” for purposes of new technology add-on payments. Consistent with the statute and our implementing regulations, a technology is no longer considered “new” once it is more than 2 to 3 years old, and the costs of the procedures are considered to be included in the relative weights irrespective of how frequently the technology has been used in the Medicare population ( 83 FR 41280 ).
Comment: The manufacturer of Intercept® Fibrinogen Complex (IFC), pathogen reduced cryoprecipitated fibrinogen complex (PRCFC), submitted a comment stating that due to manufacturing delays, its new technology add-on payment should be extended an additional year. The commenter explained that the IFC manufacturing process is unusual in that the IFC product must be made at blood centers, and that it has contracted with several blood centers. The commenter stated that each of these contracted blood centers must be licensed through FDA approval of a Biologics License Application (BLA) for manufacturing to ship the IFC product across state lines. The commenter stated that a complaint filed by a manufacturer of a competitive product resulted in FDA placing the BLA reviews of several of its contracted blood centers on hold and that the BLA reviews remain pending. The commenter stated that at the end of the first year of its new technology add-on payment (FY 2022), only three blood centers were authorized to ship IFC across state lines, and that as of June 2024, it was still waiting for FDA clearance of four additional blood center contract manufacturing facilities, which would increase manufacturing capacity by another 100 percent. The commenter stated that therefore, the majority of hospitals in the country did not have access to IFC in FY 2022 and FY 2023. The commenter asserted that its new technology add-on payment should be extended through FY 2025 given the significant delay in manufacturing due to the delay in BLA approvals and the resulting lack of national IFC availability.
Response: We thank the commenter for its comment. Consistent with the statute and our implementing regulations, a technology is no longer considered as “new” once it is more than 2 to 3 years old, irrespective of how frequently the medical service or technology has been used in the Medicare population ( 70 FR 47349 , 85 FR 58610 ). As such, once a technology has been available on the U.S. market for more than 2 to 3 years, we consider the costs to be included in the MS-DRG relative weights regardless of whether the technology's use in the Medicare population has been frequent or infrequent ( 88 FR 58802 ).
After consideration of the public comments we received, we are finalizing our proposal to discontinue new technology add-on payments for the technologies as listed in the proposed rule and in the following Table II.E.-02 of this final rule for FY 2025 because they are no longer “new” for purposes of new technology add-on payments. This table also presents the newness start date, new technology add-on payment start date, the 3-year anniversary date of the product's entry onto the U.S. market, and relevant final rule citations from prior fiscal years. We refer readers to the final rules cited in the following table for a complete discussion of each new technology add-on payment application and the coding and payment amount for these technologies, including the applicable indications and discussion of the newness start date.

As discussed previously, in the FY 2023 IPPS/LTCH PPS final rule, we finalized our policy to publicly post online applications for new technology add-on payment beginning with FY 2024 applications ( 87 FR 48986 through 48990 ). As noted in the FY 2023 IPPS/LTCH PPS final rule, we are continuing to summarize each application in this final rule. However, while we are continuing to provide discussion of the concerns or issues we identified with respect to applications submitted under the traditional pathway, we are providing more succinct information as part of the summaries in the proposed and final rules regarding the applicant's assertions as to how the medical service or technology meets the newness, cost, and substantial clinical improvement criteria. We refer readers to https://mearis.cms.gov/public/publications/ntap for the publicly posted FY 2025 new technology add-on payment applications and supporting information (with the exception of certain cost and volume information, and information or materials identified by the applicant as confidential or copyrighted), including tables listing the ICD-10-CM codes, ICD-10-PCS codes, and/or MS-DRGs related to the analyses of the cost criterion for certain technologies for the FY 2025 new technology add-on payment applications.
We received 16 applications for new technology add-on payments for FY 2025 under the new technology add-on payment traditional pathway. As discussed previously, in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ), we finalized that beginning with the new technology add-on payment applications for FY 2025, for technologies that are not already FDA market authorized for the indication that is the subject of the new technology add-on payment application, applicants must have a complete and active FDA market authorization request at the time of new technology add-on payment application submission and must provide documentation of FDA acceptance or filing to CMS at the time of application submission, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. See § 412.87(e) and further discussion in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ). Of the 16 applications received under the traditional pathway, one applicant was not eligible for consideration for new technology add-on payment because it did not meet these requirements, and three applicants withdrew their application prior to the issuance of the proposed rule. In accordance with the regulations under § 412.87(f), applicants for FY 2025 new technology add-on payments must have received FDA approval or clearance by May 1 of the year prior to the beginning of the fiscal year for which the application is being considered. Subsequently, prior to the issuance of this final rule, two additional applications were withdrawn for odronextamab (R/R DLBCL indication) and odronextamab (R/R FL indication). We are not including in this final rule the description and discussion of applications that were withdrawn or that are ineligible for consideration for FY 2025. We are addressing the remaining 10 applications. We note that the manufacturer for Casgevy TM (exagamglogene autotemcel) submitted a single application, but for two separate indications, each of which is discussed separately in this section. We are not approving new technology add-on payments for 6 technologies: Casgevy TM (exagamglogene autotemcel) for the indication of transfusion-dependent β-thalassemia, DuraGraft®, FloPatch FP120, Lantidra TM (donislecel-jujn (allogeneic pancreatic islet cellular suspension for hepatic portal vein infusion), AMTAGVI TM (lifileucel), and Quicktome Software Suite, for the reasons discussed in the following sections. For the remaining 5 technologies, we are approving new technology add-on payments for FY 2025 for Casgevy TM (examgamglogene autotemcel) for the indication of sickle cell disease, HEPZATO TM KIT (melphalan for injection/hepatic delivery system), and Lyfgenia TM (lovotibeglogene autotemcel). Because the remaining two technologies, ELREXFIO TM (elranatamab-bcmm) and TALVEY TM (talquetamab-tgvs), are considered substantially similar to TECVAYLI TM (teclistamab-cqyv), which was approved for new technology add-on payments for FY 2024 and is still considered “new” for purposes of new technology add-on payments for FY 2025, these technologies are also eligible for the new technology add-on payment for FY 2025. A discussion of these applications is presented in the following sections.
Vertex Pharmaceuticals, Inc. submitted an application for new technology add-on payments for Casgevy TM for FY 2025 for use in sickle cell disease. According to the applicant, Casgevy TM is a one-time, clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) modified autologous cluster of differentiation (CD)34+ hematopoietic stem & progenitor cell (HSPC) cellular therapy approved for the treatment of sickle cell disease (SCD) in patients 12 years and older with recurrent vaso-occlusive crises (VOC). Per the applicant, using a CRISPR/Cas9 gene editing technique, the patient's CD34+ HSPCs are edited ex vivo via Cas9, a nuclease enzyme that uses a highly specific guide ribonucleic acid (gRNA), at the critical transcription factor binding site GATA1 in the erythroid specific enhancer region of the B-cell lymphoma/leukemia 11A (BCL11A) gene. According to the applicant, as a result of the editing, GATA1 binding is irreversibly disrupted, and BCL11A expression is reduced, resulting in an increased production of fetal hemoglobin (HbF), and recapitulating a naturally occurring, clinically benign condition called hereditary persistence of fetal hemoglobin (HPFH) that reduces or eliminates SCD symptoms. As stated by the applicant, Casgevy TM infusion induces increased HbF production in SCD patients to ≥20 percent, which is known to be associated with fewer SCD complications via addressing the underlying cause of SCD by preventing RBC sickling. We note that the applicant is also seeking new technology add-on payments for Casgevy TM for FY 2025 for use in treating transfusion-dependent beta thalassemia (TDT), as discussed separately later in this section.
Please refer to the online application posting for Casgevy TM , available at https://mearis.cms.gov/public/publications/ntap/NTP2310171VPTU , for additional detail describing the technology and the disease treated by the technology.
With respect to the newness criterion, according to the applicant, Casgevy TM was granted Biologics License Application (BLA) approval from FDA on December 8, 2023, for treatment of SCD in patients 12 years of age or older with recurrent VOCs. According to the applicant, Casgevy TM became commercially available immediately after FDA approval. Casgevy TM is available in 20 mL vials containing 4 to 13 × 10 6 CD34+ cells/mL frozen in 1.5 to 20 mL of solution. The minimum dose is 3 × 10 6 CD34+ cells per kg of body weight, which may be contained within multiple vials.
Effective April 1, 2023, the following ICD-10-PCS codes may be used to uniquely describe procedures involving ( print page 69129) the use of Casgevy TM : XW133J8 (Transfusion of exagamglogene autotemcel into peripheral vein, percutaneous approach, new technology group 8) and XW143J8 (Transfusion of exagamglogene autotemcel into central vein, percutaneous approach, new technology group 8). The applicant provided a list of ICD-10-CM diagnosis codes that may be used to identify this indication for Casgevy TM . Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant. We believe the relevant ICD-10-CM codes to identify the indication of SCD would be: D57.1 (Sickle-cell disease without crisis), D57.20 (Sickle-cell/Hb-C disease without crisis), D57.40 (Sickle-cell thalassemia without crisis), D57.42 (Sickle-cell thalassemia beta zero without crisis), D57.44 (Sickle-cell thalassemia beta plus without crisis), or D57.80 (Other sickle-cell disorders without crisis). In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36031 ), we invited public comments on the use of these ICD-10-CM diagnosis codes to identify the indication of SCD for purposes of the new technology add-on payment, if approved. We note that we did not receive any comments on the use of these codes.
As previously discussed, if a technology meets all three of the substantial similarity criteria under the newness criterion, it would be considered substantially similar to an existing technology and would not be considered “new” for the purpose of new technology add-on payments.
With respect to the substantial similarity criteria, the applicant asserted that Casgevy TM is not substantially similar to other currently available technologies, because Casgevy TM is the first approved therapy to use CRISPR gene editing technology and no other approved technology uses the same or a similar mechanism of action; and therefore, the technology meets the newness criterion. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for Casgevy TM for the applicant's complete statements in support of its assertion that Casgevy TM is not substantially similar to other currently available technologies.

As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36032 ), we noted that Casgevy TM may have the same or similar mechanism of action to Lyfgenia TM , for which we also received an FY 2025 new technology add-on payment application. Casgevy TM and Lyfgenia TM are both gene therapies using modified autologous CD34+ HSPC therapies administered via stem cell transplantation for the treatment of SCD. Lyfgenia TM was approved by FDA for this indication on December 8, 2023. We noted that both technologies are autologous, ex-vivo modified hematopoietic stem-cell biological products. For these technologies, patients are required to undergo CD34+ HSPC mobilization followed by apheresis to extract CD34+ HSPCs for manufacturing and then myeloablative conditioning using busulfan to deplete the patient's bone marrow in preparation for the technologies' modified stem cells to engraft to the bone marrow. Once engraftment occurs for both technologies, the patient's cells start to produce a different form of hemoglobin in order to reduce the sickling hemoglobin. We further noted that both technologies appeared to map to the same MS-DRGs, MS-DRGs 016 and 017 (Autologous Bone Marrow Transplant with CC/MCC, and without CC/MCC, respectively), and to treat the same or similar disease (SCD) in the same or similar patient population (patients 12 years of age and older who have a history of VOCs). Accordingly, as it appeared that Casgevy TM and Lyfgenia TM may use the same or similar mechanism of action to achieve a ( print page 69130) therapeutic outcome (that is, to reduce the amount of sickling hemoglobin to reduce and prevent VOEs associated with SCD), were assigned to the same MS-DRGs, and treated the same or similar patient population and disease, we stated our belief that these technologies may be substantially similar to each other such that they should be considered as a single application for purposes of new technology add-on payments. We noted that if we determined that this technology is substantially similar to Lyfgenia TM , we believed the newness period would begin on December 8, 2023, the date both Casgevy TM and Lyfgenia TM received FDA approval for SCD. We stated we were interested in information on how these two technologies may differ from each other with respect to the substantial similarity criteria and newness criterion, to inform our analysis of whether Casgevy TM and Lyfgenia TM are substantially similar to each other, and therefore, should be considered as a single application for purposes of new technology add-on payments.
We invited public comments on whether Casgevy TM meets the newness criterion, including whether Casgevy TM is substantially similar to Lyfgenia TM and whether these technologies should be evaluated as a single technology for purposes of new technology add-on payments.
Comment: The applicant for Casgevy TM submitted a public comment regarding substantial similarity for Lyfgenia TM and Casgevy TM. The applicant asserted Casgevy TM represents the first therapy approved to use CRISPR/Cas9 gene editing technology and stated that no other approved technologies use this mechanism of action, and CRISPR/Cas9 technology has never previously been used in humans outside of clinical trials. The applicant stated that Casgevy TM is a one-time treatment that uses ex vivo non-viral CRISPR/Cas9 to precisely edit the erythroid-specific enhancer region of BCL11A in CD34+ HSPCs. The applicant stated that, while other non-gene therapy-based therapeutic approaches impact production of HbF, no other approved technology has been able to reactivate production of endogenous HbF to levels known to eliminate disease complications (for example, VOC), consistent with individuals with a clinically benign condition called hereditary persistence of fetal hemoglobin (HPFH) who experience no or minimal disease complications from SCD when they co-inherit both HPFH and SCD; therefore, it stated Casgevy TM satisfies the newness criterion. The applicant stated that CMS focused on perceived similarities in treatment journey and categorical product characteristics between Casgevy TM and certain other technologies, but did not acknowledge material differences in the underlying technology which impact the safety and efficacy profile of these products. The applicant further explained that after Casgevy TM infusion, the edited CD34+ cells engraft in the bone marrow and differentiate to erythroid lineage cells with reduced BCL11A expression, and that this reduced BCL11A expression results in an increase in γ-globin expression and HbF protein production in erythroid cells. The applicant stated that in patients with severe SCD, HbF expression reduces intracellular hemoglobin S (HbS) concentration, preventing the red blood cells from sickling and addressing the underlying cause of disease, thereby eliminating VOCs. The applicant stated that, as such, Casgevy TM is not similar to the current standard of care (bone marrow transplant), nor to other technologies used in the treatment of SCD, and that none of these treatments use a mechanism of action that relies on CRISPR gene editing to reduce intracellular HbS concentration in SCD patients. The applicant explained how Lyfgenia TM uses a separate technology, gene replacement therapy, that utilizes a viral-based mechanism to introduce exogenous genetic material into patients' HSPCs, to add functional copies of a modified βA-globin gene into patients' hematopoietic stem cells (HSCs) through transduction of autologous CD34+ cells with B8305 lentiviral vector (LVV). The applicant stated that due to the LVV-based mechanism of action and the semi-random nature of viral integration, there is a potential risk of LVV-mediated insertional oncogenesis after treatment with Lyfgenia TM used in the treatment of SCD, as documented in FDA-approved labeling. The applicant stated that Casgevy TM , with its non-viral mechanism of action using CRISPR/Cas9 gene editing, does not employ a viral vector and does not insert a transgene; therefore, insertional oncogenesis cannot occur as a matter of scientific principle. The applicant further stated that Casgevy TM uses a unique underlying technology and manufacturing process and has distinct product characteristics that differentiate it from other technologies used to treat SCD. The applicant asserted in its comments that if CMS were to consider gene replacement therapy and gene editing technologies to be substantially similar, it could set a precedent based on overgeneralization which could deter further innovation.
Another commenter who is the manufacturer of Lyfgenia TM also submitted a public comment regarding the newness criterion. With respect to mechanism of action, the applicant stated that Lyfgenia TM has a unique mechanism of action that differs from Casgevy TM 's because it is a one-time gene therapy that adds functional copies of the β A-T87Q -globin gene into a patient's own HSCs ex-vivo through the transduction of autologous CD34+ cells with a BB305 LVV to durably produce HbA T87Q . The commenter added that HbA T87Q is a modified adult hemoglobin (HbA) specifically designed to be anti-sickling while maintaining the same structure and function as naturally occurring HbA. According to the commenter, Lyfgenia TM consists of an autologous CD34+ cell-enriched population from patients with SCD that contains HSCs transduced with BB305 LVV encoding the β A-T87Q -globin gene, suspended in a cryopreservation solution. The commenter stated the BB305 LVV encodes a single amino acid variant of β-globin gene, β A-T87Q -globin: a human β-globin with a genetically engineered single amino acid change (threonine [Thr; T] to glutamine [Gin; Q] at position 87 (T87Q)). The commenter asserted HbA T87Q is nearly identical to wildtype (or “innate”) HbA, which is not prone to sickling. The commenter stated the T87Q substitution introduced in β A-T87Q -globin is designed to physically block or sterically inhibit polymerization of hemoglobin, thus rendering further “anti-sickling” properties to β A-T87Q -globin. According to the commenter, this results in a transgenic, non-immunogenic protein that can be measured in blood allowing for monitoring of the therapeutic protein in vivo and quantification relative to other globin species used to treat SCD. The commenter stated that Lyfgenia TM is not substantially similar to the CRISPR-Cas9 gene editing technique of Casgevy TM . The commenter also stated that, as described previously, Lyfgenia TM adds functional copies of a modified β-globin (HBB) gene, β A-T87Q globin gene, into patients' own HSCs to durably produce HbA T87Q , a modified adult HbA specifically designed to be anti-sickling while maintaining the same morphology and function as naturally occurring HbA. According to the commenter, the CRISPR/Cas9 gene editing technique mechanism of action described for Casgevy TM in the proposed rule differs substantially from Lyfgenia TM , as is evident by Casgevy TM 's ( print page 69131) unique editing approach in which GATA1 binding is irreversibly disrupted, and BCL11A expression is reduced, resulting in an increased production of HbF, and recapitulating a naturally occurring, clinically benign condition called HPFH that reduces or eliminates SCD symptoms.
According to the commenter, increasing HbA T87Q versus increasing HbF are fundamentally distinct mechanistic approaches. For individuals without SCD, HbF production is decreased shortly after birth, coinciding with an increase in HbA, and Lyfgenia TM is designed to replicate this natural state by introducing the production of HbA T87Q . The commenter stated HbA T87Q is nearly identical to HbA in several keyways: sequence homology, protein structure, oxygen affinity and oxygen dissociation curves. The commenter stated that HbF has ~50 percent homology to HbA (two β globin chains are replaced with two γ-chains) and has a higher observed oxygen affinity and different oxygen unloading properties than HbA. According to the commenter, from a clinical perspective, current standard of care approaches (for example, the use of hydroxyurea) are available to increase levels of HbF with variable effectiveness, while the mechanism of action Lyfgenia TM affords is unique in increasing a modified HbA. The commenter commented that while both gene therapies are indicated for the treatment of SCD, the mechanistic approach of each is fundamentally and significantly different from the other, and therefore Lyfgenia TM and Casgevy TM are not substantially similar and should not be considered as a single application for the purposes of new technology add-on consideration.
The commenter also described potential risks associated with consideration of the two technologies as a single application. Specifically, the commenter stated that if Lyfgenia TM and Casgevy TM are treated as a single application and paid under a single maximum new technology add-on payment amount, this could potentially undermine CMS's aim to improve timely, meaningful access to SCD gene therapies for Medicare patients. Per the commenter, not only do the two therapies have distinct mechanisms of action but they also differ in the length of follow-up and the features of the population in which they were studied (for example, the commenter stated that the Lyfgenia TM clinical trials did not exclude patients with a history of chronic pain and included some patients with a history of stroke), and patients should have a choice to work with physicians to decide which therapy is most appropriate, based solely on their specific individual clinical circumstances. The commenter further asserted that given these differences, the finalization of a single new technology add-on payment amount for both therapies could hamper patient access to the most appropriate gene therapy, and potentially create a fiscally problematic and financial loss for IPPS hospitals, given the difference in the wholesale acquisition costs of both therapies, and CMS could potentially over-pay for one product, while under-paying for the other through the use of the historical blended weighted average cost utilizing volume estimates. Therefore, the commenter stated that Lyfgenia TM is not substantially similar to Casgevy TM and should not be considered as a single application with Casgevy TM for the purposes of new technology add-on payments.
Response: We thank the applicant and the other commenters for their comments. Based on our review of comments received and information submitted by the applicant as part of its FY 2025 new technology add-on payment application for Casgevy TM , we agree that Casgevy TM and Lyfgenia TM do not have the same mechanism of action because Casgevy TM modifies a patients' own HSPCs to increase HbF expression to subsequently reduce the expression of intracellular sickled hemoglobin concentration, which is a distinct mechanism of action compared to Lyfgenia TM, which modifies a patients' own HSPCs to increase HbA T87Q (modified adult hemoglobin). Therefore, we agree with the applicant that Casgevy TM has a unique mechanism of action and is not substantially similar to existing treatment options for the treatment of SCD in patients 12 years of age or older with recurrent VOCs and meets the newness criterion. We consider the beginning of the newness period for Casgevy TM to commence on December 8, 2023, when Casgevy TM was granted BLA approval from FDA for the treatment of SCD in patients 12 years of age or older with recurrent VOCs.
With respect to the cost criterion, the applicant searched the FY 2022 MedPAR file and provided multiple analyses to demonstrate that Casgevy TM meets the cost criterion. The applicant included two cohorts in the analyses to identify potential cases representing patients who may be eligible for Casgevy TM : the first cohort included all cases in MS-DRG 014 (Allogeneic Bone Marrow Transplant) to account for the low volume of SCD or transfusion-dependent beta thalassemia (TDT) cases, and the second cohort included cases in MS-DRG 014 (Allogeneic Bone Marrow Transplant) with any ICD-10-CM diagnosis code of SCD or TDT. The applicant explained that the cost analyses for SCD and TDT were combined because the volume of cases with a sickle cell disease or beta thalassemia diagnosis code was very low, and because it believed both indications would be approved in time for new technology add-on payment. In addition, the applicant noted that when searching for cases in MS-DRG 014 with SCD or beta thalassemia diagnosis codes, there were no beta thalassemia cases. The applicant noted that cases included in the analysis may not be a completely accurate representation of cases that will be eligible for Casgevy TM but that the analyses were provided in recognition of the low volume of cases.
The applicant performed two analyses for each cohort: one with all prior drug charges maintained, representing a scenario in which there is no change to patient drug regimen with the use of Casgevy TM ; and another with all prior drug charges removed, representing a scenario in which no ancillary drugs are used in the treatment of Casgevy TM patients. Per the applicant, this was done because some patients receiving Casgevy TM could receive fewer ancillary drugs during the inpatient stay, but it was difficult to know with certainty whether this would be the case or to identify the exact differences in drug regimens between patients receiving Casgevy TM and those receiving allogeneic bone marrow transplants. The applicant noted the analyses with drug charges removed were likely an over-estimation of the ancillary drug charges that would be removed in cases involving the use of Casgevy TM , but these were provided as sensitivity analyses.
According to the applicant, eligible cases for Casgevy TM will be mapped to either Pre-MDC MS-DRGs 016 or 017, depending on whether complications or comorbidities (CCs) or major complications or comorbidities (MCCs) are present. For each analysis, the applicant used the FY 2025 new technology add-on payment threshold for Pre-MDC MS-DRG 016 for all identified cases, because it was typically higher than the threshold for Pre-MDC MS-DRG 017. Each analysis followed the order of operations described in the table later in this section.
For the first cohort, the applicant included all cases associated with MS-DRG 014 (Allogeneic Bone Marrow Transplant). The applicant used the inclusion/exclusion criteria described in the following table and identified 996 ( print page 69132) claims mapping to MS-DRG 014. With all prior drug charges maintained (Scenario 1), the applicant calculated a final inflated average case-weighted standardized charge per case of $12,325,062, which exceeded the average case-weighted threshold amount of $182,491. With all prior drug charges removed (Scenario 2), the applicant calculated a final inflated average case-weighted standardized charge per case of $12,181,526, which exceeded the average case-weighted threshold amount of $182,491.
For the second cohort, the applicant searched for cases within MS-DRG 014 with any ICD-10-CM diagnosis codes representing SCD or TDT. The applicant used the inclusion/exclusion criteria described in the following table and identified 11 claims mapping to MS-DRG 014. With all prior drug charges maintained (Scenario 3), the applicant calculated a final inflated average case-weighted standardized charge per case of $12,125,212, which exceeded the average case-weighted threshold amount of $182,491. With all prior drug charges removed (Scenario 4), the applicant calculated a final inflated average case-weighted standardized charge per case of $12,086,551, which exceeded the average case-weighted threshold amount of $182,491.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant maintained that Casgevy [ TM ] meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36034 ), we invited public comments on whether Casgevy TM meets the cost criterion.
Comment: The applicant reiterated that the cost criterion analyses submitted with the application demonstrate that Casgevy TM meets the cost criterion.
Response: We thank the applicant for its comments. We agree that the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios. Therefore, Casgevy TM meets the cost criterion.
With regard to the substantial clinical improvement criterion, the applicant asserted that Casgevy [ TM ] represents a substantial clinical improvement over existing technologies because it is anticipated to expand patient eligibility for potentially curative SCD therapies, have improved clinical outcomes relative to available therapies, and avoid certain serious risks or side effects associated with existing potentially curative treatment options for SCD. The applicant provided one study to support these claims, as well as eight background articles about clinical outcomes and safety risks of other SCD treatments. [ 24 ] The following table summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for Casgevy TM for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36035 through 36036 ), after reviewing the information provided by the applicant, we stated we had the following concerns regarding whether Casgevy [ TM ] meets the substantial clinical improvement criterion. We noted that the only assessment of the technology submitted was from conference presentations that provided data on the ongoing CLIMB-121 trial, a phase 1/2/3 single-arm trial assessing a single dose of Casgevy [ TM ] in patients 12 to 35 years old with SCD and a history of two or more severe VOCs per year over 2 years. The most recent data presented at ASH in December 2023, [ 25 ] which appears to supersede the earlier results from Locatelli, et al. (2023), [ 26 ] indicated 44 participants received Casgevy TM for SCD, of which only 30 participants were evaluable for the primary and key secondary endpoints because they were followed for at least 16 months (up to 45.5 months) post Casgevy TM infusion. The applicant stated 96.7 percent of patients achieved the primary efficacy endpoint (free of severe VOCs for at least 12 consecutive months) and 100 percent of patients achieved the key secondary efficacy endpoint (free from in-patient hospitalization for severe VOCs for at least 12 consecutive months). Additionally, the applicant noted a safety profile consistent with myeloablative busulfan and autologous HSCT and that there were no malignancies nor serious adverse events related to Casgevy TM . However, we noted that the provided evidence did not include peer-reviewed literature that directly assessed the use of Casgevy TM for SCD. We questioned whether the small study population may limit the generalizability of these study outcomes to a Medicare population. In addition, from the evidence submitted, we noted we were unable to determine where the study took place (that is, within the United States (U.S.) or in locations outside the U.S), which may also limit generalizability to the Medicare population. Additionally, we questioned if the short follow-up duration was sufficient to assess improvements in long-term clinical outcomes.
Furthermore, the applicant asserted that Casgevy TM significantly improves ( print page 69134) clinical outcomes relative to services or technologies previously available. Regarding the claim that Casgevy TM is the first gene therapy specifically approved for the treatment of SCD in patients 12 years and older with recurrent VOCs, the applicant claimed it was first to submit and have its BLA accepted for a genetic therapy for treatment of SCD. The applicant stated the PDUFA date for Casgevy TM was December 8, 2023, and the PDUFA data for another gene therapy for SCD was December 20, 2023, however, we note that Casgevy TM and another product were both approved on December 8, 2023, as the first gene therapies for SCD. While this claim was made in support of the assertion that Casgevy TM significantly improves clinical outcomes, we noted that the information submitted regarding PDUFA dates and FDA approvals did not appear to provide data regarding a significantly improved clinical outcome under § 412.87(b)(1)(ii)(C).
With regards to the claim that Casgevy TM is expected to avoid certain serious risks or side effects associated with approved viral-based gene therapies for SCD, the applicant cited the potential risk of insertional oncogenesis after treatment with Lyfgenia TM , listed per the package insert for this other gene therapy for SCD. We noted that because clinical trials are conducted under widely varying conditions, we questioned whether adverse reaction rates observed in the clinical trials of one drug can be directly compared to rates in the clinical trials of another drug. We also questioned if the follow-up duration for patients treated with Casgevy TM was sufficient to assess improvement in the rate of malignancy.
With regard to the claim that Casgevy TM is expected to avoid certain serious risks or side effects associated with existing potentially curative treatment options for SCD, the applicant stated that there are significant risks associated with allo-HSCT, including graft failure (up to 9 percent frequency), acute and chronic graft-versus-host disease (GVHD) (with chronic GVHD up to 18 percent frequency), severe infection, hematologic malignancy, bleeding events, and death. In contrast, the applicant claimed Casgevy TM does not require an allogeneic donor as each patient is their own donor, and therefore, does not have the risks of acute and chronic GVHD or the immunologic risks of secondary graft failure/rejection, in addition to not requiring post-transplant immunosuppressive therapies. However, we stated that we were interested in additional evidence regarding the frequency and clinical relevance of side effects such as severe infection, hematologic malignancy, bleeding events, and death for both therapies.
We invited public comments on whether Casgevy TM meets the substantial clinical improvement criterion.
Comment: A few commenters, including the applicant, stated support for approval of Casgevy TM for new technology add-on payments for the SCD indication and disagreed with CMS's concerns. A commenter stated that for beneficiaries with SCD, the available therapy of HSCT is a potentially curative treatment especially for patients with significant barriers to access such as lack of a matched sibling who could potentially serve as a donor and the potential increased risks from the side effects of stem cell transplant.
Response: We thank the commenters for their input and have taken it into consideration in determining whether Casgevy TM meets the substantial clinical improvement criterion as discussed later in this section.
Comment: In response to our concerns about the lack of any published, peer-reviewed studies that directly assessed the use of Casgevy TM within the U.S., the applicant provided additional information from a published phase 3, single-group, open-label study by Frangoul, et al. (2024) [ 27 ] which assessed Casgevy TM in patients 12 to 35 years of age with SCD who had at least two severe VOCs in each of the 2 years before screening. The applicant stated that the study was conducted in both the U.S. and European Union in which a total of 44 patients received exagamglogene autotemcel, and the median follow-up was 19.3 months (range, 0.8 to 48.1). The applicant stated that of the 30 patients who had sufficient follow-up to be evaluated, 29 (97 percent; 95 percent CI, 83 to 100) were free from VOCs for at least 12 consecutive months, and all 30 (100 percent; 95 percent Cl, 88 to 100) were free from hospitalizations for VOCs for at least 12 consecutive months (P<0.001 for both comparisons against the null hypothesis of a 50 percent response).
In response to our concerns about providing peer-reviewed evidence of the safety profile of Casgevy TM , the applicant stated that the Frangoul, et al. (2024) study showed that the safety profile of Casgevy TM was generally consistent with that of myeloablative busulfan conditioning and autologous HSPC transplantation and that no cancers occurred. The applicant stated that, while patients treated with Casgevy TM experienced adverse effects, the adverse effects are consistent with the conditioning regimen, similar to adverse effects in autologous transplant. The applicant stated that in the CLIMB SCD-121 trial [ 28 ] for SCD, the most common adverse events were stomatitis (55 percent), febrile neutropenia (48 percent), platelet count decrease (48 percent), and appetite decrease (41 percent). The applicant stated that patients treated with Casgevy TM did not have any reported cases of graft-versus-host-disease (GVHD), which is a common and potentially serious side effect that can be seen in allogeneic transplant.
In response to our concern regarding oncogenesis with gene therapy, the applicant stated that the two primary potential mechanisms for oncogenesis post-treatment include a late effect of alkylating chemotherapy or oncogene activation from off-target editing or insertional oncogenesis, as seen in other technologies used in treatment of SCD. In Frangoul, et al. (2024) no off-target editing was found through multiple orthogonal approaches. The applicant clarified, however, that alkylating agents generally require 5 to 7 years before secondary malignancies occur, and although off-target genome editing was not observed in the edited CD34+ cells evaluated from healthy donors and patients, the risk of unintended, off-target editing in an individual's CD34+ cells cannot be ruled out due to genetic variants and therefore, the clinical significance of potential off-target editing is unknown. The applicant further stated that longest follow-up in both the CLIMB SCD-121 and CLIMB THAL-111 trials has surpassed 4 years, and stated that it will continue to follow study patients for up to 15 years. The applicant further asserted that due to Casgevy TM 's mechanism of action, which does not employ a viral vector and does not insert a transgene, insertional oncogenesis by definition cannot occur.
In response to our concerns about sample size, the applicant stated its belief that the study sample sizes are appropriate. The applicant stated that SCD affects an estimated 100,000 Americans and that the sample size of the studies reflects the challenges ( print page 69135) associated with enrolling larger studies for rare conditions, as well as significant challenges in conducting larger studies for autologous gene therapy that must be individualized to each patient.
In response to our concern about the generalizability of the evidence to the Medicare population, the applicant commented that it believed the study population reflects the patient population for these medical conditions, including Medicare-covered patients who, as noted, may be dually eligible for Medicare and Medicaid (and thus often not over the age of 65). The commenter also stated that, as noted in the CMS SCD Action Plan, [ 29 ] 11 percent of patients with SCD are enrolled in Medicare. The applicant stated that the CLIMB-121's study population is generalizable as it included patients aged 12-35, reflective of dual Medicare and Medicaid-eligible populations. The applicant stated that CMS has previously shared SCD prevalence data, indicating that more than 70 percent of Medicare fee-for-service beneficiaries with SCD are dual eligibles and more than 80 percent of these beneficiaries with SCD are covered under Medicare through disability insurance benefits.
Response: We thank the applicant for the additional information and have taken it into consideration in determining whether Casgevy TM meets the substantial clinical improvement criterion, discussed later in this section.
Comment: A commenter, who is the manufacturer of Lyfgenia TM , stated that it was not possible to make direct comparisons between the safety or efficacy of Casgevy TM and Lyfgenia TM . The commenter stated that while Lyfgenia TM 's prescribing information includes a warning as to the potential risk of insertional oncogenesis after treatment, there have been no cases of insertional oncogenesis nor any positive results for replication competent lentivirus observed [ 30 ] across the utilization of the BB305 vector across all clinical studies. The commenter also cited the prescribing information for Casgevy TM stating: “[a]lthough not observed in healthy donors and patients, the risk of unintended, off-target editing in CD34+ cells due to uncommon genetic variants cannot be ruled out.” The commenter further stated that although no cases of insertional oncogenesis have been observed with BB305 across the clinical program, two cases of acute myeloid leukemia were observed in patients treated with an earlier version of Lyfgenia TM using a different manufacturing process and transplant procedure, and that patients treated with Lyfgenia TM may develop hematologic malignancies and should have lifelong monitoring.
Response: We thank the applicant and other commenters for their comments regarding the substantial clinical improvement criterion. Based on the additional information received, we agree with the applicant that Casgevy TM represents a substantial clinical improvement over existing technologies because Casgevy TM offers a treatment option for certain patients with SCD who are not eligible for bone marrow transplant due to a lack of HLA matching and who experience recurrent VOEs and have not been able to achieve adequate control of the condition with existing treatments such as hydroxyurea.
After consideration of the public comments and the information included in the applicant's new technology add-on payment application, we have determined that Casgevy TM for the indication of SCD meets the criteria for approval for new technology add-on payment. Therefore, we are approving new technology add-on payments for this technology for SCD for FY 2025.
Cases involving the use of Casgevy TM for the indication of SCD that are eligible for new technology add-on payments will be identified by ICD-10-PCS codes: XW133J8 (Transfusion of exagamglogene autotemcel into peripheral vein, percutaneous approach, new technology group 8) or XW143J8 (Transfusion of exagamglogene autotemcel into central vein, percutaneous approach, new technology group 8) in combination with one of the following ICD-10-CM codes: D57.1 (Sickle-cell disease without crisis), D57.20 (Sickle-cell/Hb-C disease without crisis), D57.40 (Sickle-cell thalassemia without crisis), D57.42 (Sickle-cell thalassemia beta zero without crisis), D57.44 (Sickle-cell thalassemia beta plus without crisis), or D57.80 (Other sickle-cell disorders without crisis).
In its application, the applicant estimated that the cost of Casgevy TM is $2,200,000 per patient. As discussed in section II.E.10 of the preamble of this final rule, we are revising the maximum new technology add-on payment percentage to 75 percent, for a medical product that is a gene therapy that is indicated and used specifically for the treatment of SCD and approved for new technology add-on payments for the treatment of SCD in the FY 2025 IPPS/LTCH PPS final rule. Accordingly, under § 412.88(a)(2) as revised in this final rule, we limit new technology add-on payments to the lesser of 75 percent of the average cost of the technology, or 75 percent of the costs in excess of the MS-DRG payment for the case. As a result, the maximum new technology add-on payment for a case involving the use of Casgevy TM for the treatment of SCD is $1,650,000 for FY 2025.
Vertex Pharmaceuticals, Inc. submitted an application for new technology add-on payments for Casgevy TM for FY 2025 for TDT. According to the applicant, Casgevy TM is a one-time, clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein 9 (Cas9) modified autologous cluster of differentiation (CD)34+ hematopoietic stem & progenitor cell (HSPC) cellular therapy indicated for the treatment of TDT in patients 12 years of age or older. Per the applicant, using a CRISPR/Cas9 gene editing technique, the patient's CD34+ HSPCs are edited ex vivo via Cas9, a nuclease enzyme that uses a highly-specific guide ribonucleic acid (gRNA), at the critical transcription factor binding site GATA1 in the erythroid specific enhancer region of the B-cell lymphoma/leukemia 11A (BCL11A) gene. According to the applicant, as a result of the editing, GATA1 binding is irreversibly disrupted, and BCL11A expression is reduced, resulting in an increased production of fetal hemoglobin (HbF). As stated by the applicant, this increase in HbF recapitulates a naturally occurring, clinically benign condition called hereditary persistence of fetal hemoglobin (HPFH). The applicant stated that as a result, Casgevy TM infusion induces increased HbF production in TDT patients so that circulating red blood cells (RBC) exhibit nearly 100 percent HbF, eliminating the need for RBC transfusions. As previously discussed earlier in this section, the applicant is also seeking new technology add-on payments for Casgevy TM for FY 2025 for use in treating SCD.
With respect to the newness criterion, according to the applicant, Casgevy TM ( print page 69136) was granted BLA approval from FDA on January 16, 2024, for the treatment of TDT in patients 12 years of age and older. The applicant also explained that the minimum dosage of Casgevy TM is 3 × 10 6 CD34+ cells per kg of patient's weight. A single dose of Casgevy TM is supplied in one or more vials, with each vial containing 4 to 13 × 10 6 cells/mL suspended in 1.5 to 20 mL of cryo-preservative medium.
Effective April 1, 2023, the following ICD-10-PCS codes may be used to uniquely describe procedures involving the use of Casgevy TM : XW133J8 (Transfusion of exagamglogene autotemcel into peripheral vein, percutaneous approach, new technology group 8) and XW143J8 (Transfusion of exagamglogene autotemcel into central vein, percutaneous approach, new technology group 8). The applicant provided a list of diagnosis codes that may be used to currently identify this indication for Casgevy TM under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36036 ), we stated our belief that the relevant ICD-10-CM codes to identify the indication of TDT would be: D56.1 (Beta thalassemia), D56.2 (Delta-beta thalassemia), or D56.5 (Hemoglobin E-beta thalassemia). We invited public comments on the use of these ICD-10-CM diagnosis codes to identify the indication of TDT for purposes of the new technology add-on payment, if approved.
With respect to the substantial similarity criteria, the applicant asserted that Casgevy TM is not substantially similar to other currently available technologies because Casgevy TM is the first approved therapy to use CRISPR gene editing as its mechanism of action, and therefore, the technology meets the newness criterion. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for Casgevy TM for the applicant's complete statements in support of its assertion that Casgevy TM is not substantially similar to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36037 ), we questioned whether Casgevy TM may be the same or similar to other gene ( print page 69137) therapies used to treat TDT, specifically Zynteglo TM , which was approved for treatment of TDT on August 17, 2022. Casgevy TM and Zynteglo TM are both gene therapies using modified autologous CD34+ HSPC therapies administered via stem cell transplantation for the treatment of TDT. Both technologies are autologous, ex-vivo modified hematopoietic stem-cell biological products. For these technologies, patients are required to undergo CD34+ HSPC mobilization followed by apheresis to extract CD34+ HSPCs for manufacturing and then myeloablative conditioning using busulfan to deplete the patient's bone marrow in preparation for the technologies' modified stem cells to engraft to the bone marrow. Once engraftment occurs, the patient's cells start to produce a different form of hemoglobin to increase total hemoglobin and reduce the need for RBC transfusions. Therefore, we noted that it appeared as if Casgevy TM and Zynteglo TM would use a similar mechanism of action to achieve a therapeutic outcome for the treatment of TDT. Further, both technologies appeared to map to the same MS-DRGs, MS-DRG 016 (Autologous Bone Marrow Transplant with CC/MCC) and 017 (Autologous Bone Marrow Transplant without CC/MCC), and to treat the same or similar disease (beta thalassemia) in the same or similar patient population (patients who require regular blood transfusions). Accordingly, we stated our belief that these technologies may be substantially similar to each other. We noted that if Casgevy TM is substantially similar to Zynteglo TM for the treatment of TDT, we believed the newness period for this technology would begin on August 17, 2022, the BLA approval date for Zynteglo TM .
We invited public comments on whether Casgevy TM is substantially similar to existing technologies and whether Casgevy TM meets the newness criterion.
Comment: The applicant objected to the use of the Zynteglo TM market entry date as the start of the newness period. With respect to substantial similarity, the applicant stated that Casgevy TM is a nonviral, autologous cell therapy that is designed to reactivate fetal hemoglobin production by means of ex vivo CRISPR/Cas9 gene editing at the erythroid enhancer region of BCL11A in a patient's own hematopoietic stem and progenitor cells (HSPCs). The applicant stated that after Casgevy TM infusion, the edited CD34+ cells engraft in the bone marrow and differentiate to erythroid lineage cells with reduced BCL11A expression. The applicant stated that reduced BCL11A expression results in an increase in γ-globin expression and HbF protein production in erythroid cells. The applicant stated that in patients with TDT, γ-globin production improves the α-globin to non-α-globin imbalance thereby reducing ineffective erythropoiesis and hemolysis and increasing total hemoglobin levels, addressing the underlying cause of disease, and eliminating the dependence on regular RBC transfusions. The applicant asserted that Casgevy TM is not similar to the current standard of care for TDT (non-curative, lifelong regular blood transfusions), nor to other technologies used in the treatment of TDT, because it relies on a completely different mechanism of action than either of these treatments and therefore, Casgevy TM satisfies the newness criterion.
Another commenter, who is the manufacturer of Zynteglo TM , also stated that these technologies are not substantially similar to one another. The commenter stated the CRISPR/Cas9 gene editing technique of Casgevy TM is not substantially similar to the gene editing approach used for Zynteglo TM , which works by adding functional copies of a modified form of the β A-T87Q -globin gene into a patient's own HSPCs to allow them to make normal to near-normal levels of total hemoglobin without regular red blood cell transfusions.
Response: Based on our review of comments received and information submitted by the applicant as part of its FY 2025 new technology add-on payment application for Casgevy TM , we agree with the applicant that Casgevy TM modifies HSPCs to stimulate production of endogenous HbF, and does not modify HSPCs to increase HbA T87Q (modified adult hemoglobin) as seen with Zynteglo TM, in order to increase total hemoglobin levels. Therefore, we agree with the applicant that Casgevy TM has a unique mechanism of action and is not substantially similar to existing treatment options for the treatment of TDT in patients 12 years of age and older and meets the newness criterion. We therefore consider the beginning of the newness period to commence on January 16, 2024, when Casgevy TM was granted BLA approval from FDA for the treatment of TDT in patients 12 years of age and older.
With respect to the cost criterion, the applicant searched the FY 2022 MedPAR file and provided multiple analyses to demonstrate that Casgevy TM meets the cost criterion. The applicant included two cohorts in the analyses to identify potential cases representing patients who may be eligible for Casgevy TM : the first cohort included all cases in MS-DRG 014 (Allogeneic Bone Marrow Transplant) to account for the low volume of SCD or TDT cases, and the second cohort included cases in MS-DRG 014 (Allogeneic Bone Marrow Transplant) with any ICD-10-CM diagnosis code of SCD or TDT. The applicant explained that the cost analyses for SCD and TDT were combined because the volume of cases with a sickle cell disease or beta thalassemia diagnosis code was very small, and because it believed both indications would be approved in time for new technology add-on payment. In addition, the applicant noted that when searching for cases in MS-DRG 014 with SCD or beta thalassemia diagnosis codes, there were no beta thalassemia cases. The applicant noted that cases included in the analysis may not be a completely accurate representation of cases that will be eligible for Casgevy TM but that the analyses were provided in recognition of the low volume of cases.
The applicant performed two analyses for each cohort: one with all prior drug charges maintained, representing a scenario in which there is no change to patient drug regimen with the use of Casgevy TM ; and the other with all prior drug charges removed, representing a scenario in which no ancillary drugs are used in the treatment of Casgevy TM patients. Per the applicant, this was done because some patients receiving Casgevy TM could receive fewer ancillary drugs during the inpatient stay, but it was difficult to know with certainty whether this would be the case or to identify the exact differences in drug regimens between patients receiving Casgevy TM and those receiving allogeneic bone marrow transplants. The applicant noted the analyses with drug charges removed were likely an over-estimation of the ancillary drug charges that would be removed in cases involving the use of Casgevy TM , but these were provided as sensitivity analyses.
According to the applicant, eligible cases for Casgevy TM will be mapped to either Pre-MDC MS-DRG 016 (Autologous Bone Marrow Transplant with CC/MCC) or Pre-MDC MS-DRG 017 (Autologous Bone Marrow Transplant without CC/MCC), depending on whether complications or comorbidities (CCs) or major complications or comorbidities (MCCs) are present. For each analysis, the applicant used the FY 2025 new technology add-on payment threshold for Pre-MDC MS-DRG 016 for all identified cases, because it was typically higher than the threshold for Pre-MDC MS-DRG 017. Each analysis followed ( print page 69138) the order of operations described in the table later in this section.
For the first cohort, the applicant included all cases associated with MS-DRG 014 (Allogeneic Bone Marrow Transplant). The applicant used the inclusion/exclusion criteria described in the following table and identified 996 claims mapping to MS-DRG 014. With all prior drug charges maintained (Scenario 1), the applicant calculated a final inflated average case-weighted standardized charge per case of $12,325,062, which exceeded the average case-weighted threshold amount of $182,491. With all prior drug charges removed (Scenario 2), the applicant calculated a final inflated average case-weighted standardized charge per case of $12,181,526, which exceeded the average case-weighted threshold amount of $182,491.
For the second cohort, the applicant searched for cases within MS-DRG 014 (Allogeneic Bone Marrow Transplant) with any ICD-10-CM diagnosis codes representing SCD or TDT. The applicant used the inclusion/exclusion criteria described in the following table and identified 11 claims mapping to MS-DRG 014 (Allogeneic Bone Marrow Transplant). With all prior drug charges maintained (Scenario 3), the applicant calculated a final inflated average case-weighted standardized charge per case of $12,125,212, which exceeded the average case-weighted threshold amount of $182,491. With all prior drug charges removed (Scenario 4), the applicant calculated a final inflated average case-weighted standardized charge per case of $12,086,551, which exceeded the average case-weighted threshold amount of $182,491.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant asserted that Casgevy TM meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36039 ), we invited public comments on whether Casgevy TM meets the cost criterion.
With regard to the substantial clinical improvement criterion, the applicant asserted that Casgevy [ TM ] represents a substantial clinical improvement over existing technologies because it is expected to avoid certain serious risks or side effects associated with the existing approved gene therapy for TDT, Zynteglo [ TM ] . The applicant provided one study to support these claims, as well as two package inserts. [ 32 ] The following table summarizes the applicant's assertion regarding the substantial clinical improvement criterion. Please see the online posting for Casgevy TM for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36039 ), after reviewing the information provided by the applicant, we stated we had the following concerns regarding whether Casgevy [ TM ] meets the substantial clinical improvement criterion. We noted that the provided evidence did not include any peer-reviewed literature that directly assessed the use of Casgevy [ TM ] for TDT. We noted that the only assessment of the technology submitted was from a conference presentation [ 33 ] that provided data on the CLIMB-111 trial, an ongoing phase 1/2/3 single-arm trial assessing a single dose of Casgevy TM in patients 12 to 35 years old with TDT. The data submitted by the applicant indicated that 48 participants aged 12 to 35 years received Casgevy TM for TDT, of which only 27 participants were evaluable for the primary and key secondary endpoints because they were followed for at least 16 months (up to 43.7 months) after Casgevy TM infusion. Per the applicant's conference presentation, 88.9 percent of participants achieved both the primary efficacy endpoint (transfusion independence for 12 consecutive months while maintaining a weighted average hemoglobin of at least 9 g/dL) and the key secondary efficacy endpoint (transfusion independence for 6 consecutive months while maintaining a weighted average hemoglobin of at least 9 g/dL). The applicant noted that two patients had serious adverse events related to Casgevy TM . Due to the small study population and the median age of participants in the study, we questioned if these study outcomes would be generalizable to a Medicare population. In addition, from the evidence submitted, we stated we were also unable to determine where the study took place (that is, within the U.S. or in locations outside the U.S.), which may also limit generalizability to the Medicare population. We also questioned if the short follow-up duration was sufficient to assess improvements in long-term clinical outcomes.
Furthermore, we stated that with regard to the claim that Casgevy TM is expected to avoid certain serious risks or side effects associated with approved viral-based gene therapies for TDT, the applicant stated that Zynteglo TM utilizes gene transfer to use a modified, inert lentivirus to add working exogenous copies of the β-globin gene to increase functional hemoglobin A; due to this mechanism of action and the semi-random nature of viral integration, the applicant stated that treatment with Zynteglo TM carries the risk of lentiviral vector (LVV)-mediated insertional oncogenesis after treatment. The applicant explained that Casgevy TM is an autologous ex-vivo modified hematopoietic stem-cell biological product which uses a non-viral mechanism of action (CRISPR/Cas9 gene editing), and therefore, this technology does not carry a risk for insertional oncogenesis. The applicant also noted that gene editing approaches, including CRISPR/Cas9, have the potential to produce off-target edits, but in trials to date, off-target gene editing has not been observed in the edited CD34+ cells from healthy donors or patients. We noted that we were unclear regarding the frequency and related clinical relevance of LVV-mediated oncogenesis, and we questioned if the follow-up duration for patients treated with Casgevy TM was sufficient to assess improvement in the rate of malignancy. We also noted we were interested in more information on the overall safety profile comparison between Casgevy TM and Zynteglo TM , as well as any comparisons of Casgevy TM to another potentially curative treatment, allogeneic hematopoietic stem cell transplant for patients with TDT.
Comment: A few commenters, including the applicant, stated support for approval of Casgevy TM for new technology add-on payments for the TDT indication. A commenter further stated that it disagreed with CMS's concerns because for patients with TDT, available treatments have historically included regular blood transfusions or transplantation of bone marrow, options that present significant risk and complications; in the young population, bone marrow transplant results in a 23% rejection rate, which can ultimately become fatal. The commenter stated that for a large number of patients with TDT, a gene therapy is the only transformative, durable, and potentially curative treatment option and thus, represents a substantial improvement.
The applicant submitted a public comment regarding the substantial clinical improvement criterion. The applicant stated that following its application submission, additional data were published in the peer-reviewed New England Journal of Medicine for the TDT therapy indication which provides further support for why Casgevy TM satisfies the substantial clinical improvement criterion, as well as further evidence of safety and effectiveness and the transformative potential of Casgevy TM to treat TDT. The applicant stated that in Locatelli, et al. (2024), the authors directly assessed the use of Casgevy TM for TDT in a phase 3, single-group, open-label study (CLIMB THAL-111) in patients 12 to 35 years of age with TDT and a β 0 /β 0 , β 0 /β 0 -like, or non-β 0 /β 0 -like genotype. The applicant stated that the study showed a total of 52 patients with TDT received exagamglogene-autotemcel and were included in this prespecified interim analysis; the median follow-up was 20.4 months (range, 2.1 to 48.1) and neutrophils and platelets were engrafted in each patient. Among the 35 patients with sufficient follow-up data for evaluation, transfusion independence occurred in 32 patients (91 percent; 95 percent confidence interval, 77 to 98; P < 0.001 against the null hypothesis of a 50 percent response). During transfusion independence, the mean total hemoglobin level was 13.1 g per deciliter and the mean HbF level was 11.9 g per deciliter, and HbF had a pancellular distribution (≥94 percent of red cells). The authors of the study ( print page 69140) reported that the safety profile of Casgevy TM was generally consistent with that of myeloablative busulfan conditioning and autologous HSPC transplantation and no deaths or cancers occurred.
The applicant also stated that the study population from the CLIMB THAL-111 trial was generalizable to the Medicare population, stating that Casgevy TM was studied in trials conducted in the United States and the European Union. The applicant stated that the sample size for the studies were appropriate because TDT is a rare medical condition, and impacts only an estimated 1,000 to 1,500 Americans. The applicant stated that sample sizes of the studies involving Casgevy TM are reflective of the challenges associated with enrolling larger studies for rare conditions, as well as significant challenges in conducting larger studies for an autologous gene therapy that must be individualized to each patient. The applicant stated its belief that the study populations are reflective of the patient population for these conditions, including Medicare covered populations who will often be dual eligible (and thus often not over age 65).
The applicant stated that peer-reviewed data demonstrates the well-tolerated safety profile of Casgevy TM for TDT. The applicant stated that while patients treated with Casgevy TM experienced adverse effects, the adverse effects are consistent with the conditioning regimen, similar to adverse effects in autologous transplant. The applicant stated that in the CLIMB THAL-111 trial for TDT, the most common adverse events were febrile neutropenia (54 percent), stomatitis (40 percent), anemia (38 percent), and thrombocytopenia (35 percent), and that the patients treated with Casgevy TM did not have any reported cases of graft-versus-host-disease (GVHD), which is a common and potentially serious side effect associated with allogeneic stem cell transplant.
The applicant stated that with respect to CMS's question about the length of the follow-up durations being studied, a long-term follow-up study is also continuing to monitor total and fetal hemoglobin levels and safety, including (but not limited to) the potential for secondary cancers, vaso-occlusive events, and markers of end-organ damage in patients who have completed the current study (CLIMB-131; NCT04208529); other studies are being conducted to assess the risk of secondary cancers and off-target effects after genome editing.
In response to CMS's concern regarding oncogenesis with gene therapy, the applicant noted that the two primary potential mechanisms for oncogenesis post-treatment include a late effect of alkylating chemotherapy or oncogene activation from off-target editing or insertional oncogenesis, as seen in other technologies used in treatment of TDT. The applicant stated in newly published peer-reviewed research in New England Journal of Medicine, [ 34 ] no off-target editing was found through multiple orthogonal approaches, but that alkylating agents, however, generally require five to seven years before secondary malignancies occur. The applicant stated that the longest follow-up in the CLIMB THAL-111 trial had surpassed four years, and that it would continue to follow study patients for up to 15 years.
In response to CMS's concern regarding whether variations in clinical trial conditions allows for adequate comparison of adverse event rates between clinical trials with respect to the applicant's claim that Casgevy TM is expected to avoid potential risk associated with other technologies and allogenic bone marrow transplant procedures used in treatment of TDT, the applicant noted that the adverse event profile for Casgevy TM in TDT is consistent with busulfan myeloablative conditioning and HSPC transplant. The applicant further stated that the Casgevy TM 's mechanism of action does not employ a viral vector and does not insert a transgene, and therefore, insertional oncogenesis, a documented risk found in FDA-approved labeling for other viral-based technologies used in the treatment of TDT, by definition, cannot occur as a matter of scientific principle. The applicant further stated that although off-target genome editing was not observed in the edited CD34+ cells evaluated from healthy donors and patients, or in clinical trials to date, the risk of unintended, off-target editing in an individual's CD34+ cells cannot be ruled out due to genetic variants. In addition, the applicant stated that the clinical significance of potential off-target editing is unknown. The applicant stated that as an autologous therapy, which is manufactured from the patient's own HSPCs, which are modified with CRISPR/Cas9 gene editing technology and administered to the patient, there is no risk of GVHD or graft rejection, nor a need for immunosuppressive drugs, because the drug product is based on the patient's own cells and, according to the applicant, this is supported by clinical data generated to date in the CLIMB SCD-121 and CLIMB THAL-111 study, in which no GVHD or graft rejection/failure were observed. The applicant further stated that there are no clinical studies which exist to compare Casgevy TM to other technologies and therefore, no comparisons or conclusions of comparable safety or efficacy can be made.
Another commenter, the manufacturer of Zynteglo TM , commented on CMS's request for more information on the overall safety profile comparison between Casgevy TM and Zynteglo TM with regard to the applicant's claim that Casgevy TM is expected to avoid certain serious risks or side effects associated with approved viral-based gene therapies for TDT. The commenter stated that while the Warnings and Precautions section of the Zynteglo TM package insert includes a warning as to the potential risk of insertional oncogenesis after treatment with Zynteglo TM , there have been no cases of insertional oncogenesis nor any positive results for replication competent lentivirus observed across the utilization of the BB305 vector across all clinical studies.
Response: We thank the applicant and commenters for the additional information. After further review, we continue to have concerns as to whether Casgevy TM meets the substantial clinical improvement criterion. We continue to question whether there is evidence to demonstrate that Casgevy TM improves clinical outcomes relative to existing technologies because of the lack of comparison to allo-HSCT and Zynteglo TM , both of which are previously existing standard of care and potentially curative treatment options for this indication, and which treat the same condition in the same patient population. Without a comparison of outcomes between these existing therapies for TDT, we are unable to make a determination as to whether the technology significantly improves clinical outcomes relative to services or technologies previously available, as asserted by the applicant. We further note that the applicant's only assertion regarding whether Casgevy TM improves clinical outcomes for TDT was regarding the avoidance of a potential risk of a single side effect, and as a commenter stated, there have been no cases of insertional oncogenesis nor any positive results for replication competent lentivirus observed across the utilization of the BB305 vector across all clinical studies. We remain unclear how the provided evidence supports this claim, given that the applicant did not ( print page 69141) provide any evidence of this potential side effect occurring with use of the comparator technology. We continue to question if the follow-up duration for TDT patients treated with Casgevy TM is sufficient to adequately support the applicant's single claim, given the lack of existing data presented to assess improvement in long-term clinical outcomes and reduction in clinically significant adverse events, such as the rate of malignancy, compared with existing treatments, especially given the risk of potential unintended off-target editing remains unknown.
After review of the information submitted by the applicant as part of its FY 2025 new technology add-on payment application for Casgevy TM for TDT and consideration of the comments received, we are unable to determine that Casgevy TM for TDT meets the substantial clinical improvement criterion to be approved for new technology add-on payments for the reasons discussed in the proposed rule and in this final rule, and therefore, we are not approving new technology add-on payments for Casgevy TM for TDT for FY 2025.
Marizyme, Inc. submitted an application for new technology add-on payments for DuraGraft® for FY 2025. According to the applicant, DuraGraft® is an intraoperative vein-graft preservation solution used during the harvesting and grafting interval during coronary artery bypass graft (CABG) surgery. The applicant stated that the use of DuraGraft® does not change clinical/surgical practice; it replaces solutions currently used for flushing and storage of the saphenous vein grafts (SVG) from harvesting through grafting, including tests for graft leakage. As noted in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26795 ), Somahlution, Inc., acquired by Marizyme in 2020, [ 35 ] submitted and withdrew applications for new technology add-on payments for DuraGraft® for FY 2018 and FY 2019. The applicant also submitted an application for new technology add-on payments for FY 2020, as summarized in the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19305 through 19312 ), that it withdrew prior to the issuance of the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42180 ). We note that the applicant also submitted an application for new technology add-on payments for FY 2024, as summarized in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26795 through 26803 ), that it withdrew prior to the issuance of the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58804 ).
Please refer to the online application posting for DuraGraft®, available at https://mearis.cms.gov/public/publications/ntap/NTP231012EE9NW , for additional detail describing the technology and intraoperative ischemic injury.
With respect to the newness criterion, according to the applicant, DuraGraft® was granted De Novo classification from FDA on October 4, 2023, for adult patients undergoing CABG surgeries and is intended for flushing and storage of SVGs from harvesting through grafting for up to 4 hours. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36040 ), we noted that, per the applicant, DuraGraft® was not yet commercially available due to a delay related to finalizing the label prior to manufacturing.
The applicant stated that, effective October 1, 2017, the following ICD-10-PCS code may be used to uniquely describe procedures involving the use of DuraGraft®: XY0VX83 (Extracorporeal introduction of endothelial damage inhibitor to vein graft, new technology group 3). Please refer to the online application posting for the complete list of ICD-10-CM and -PCS codes provided by the applicant.
With respect to the substantial similarity criteria, the applicant asserted that DuraGraft® is not substantially similar to other currently available technologies because DuraGraft® is a first-in-class product as a storage and flushing solution for vascular grafts used during CABG surgery and the components of DuraGraft® directly interfere with the mechanisms of oxidative damage, and that therefore, the technology meets the newness criterion. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for DuraGraft® for the applicant's complete statements in support of its assertion that DuraGraft® is not substantially similar to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36040 ), we stated that in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26796 ), we expressed concern that the mechanism of action of DuraGraft® may be the same or similar to other vein graft storage solutions. Similarly, we noted that, according to the applicant, DuraGraft® prevents intraoperative ischemic injury to the endothelial layer of free vascular grafts, reducing the risks for post-CABG vein graft disease and graft failure, which are clinical manifestations of graft ischemia reperfusion injury (IRI), and we questioned whether DuraGraft® might have a similar mechanism of action as existing treatments for preventing ischemic injury of vein grafts during CABG surgery and reducing vein graft disease or its complications following CABG surgery. We invited public comments on whether DuraGraft® is substantially similar to existing technologies and whether DuraGraft® meets the newness criterion.
Comment: The applicant stated that the mechanism of action of DuraGraft® is not substantially similar to other products on the market. The applicant asserted that by FDA definition of a de novo request, there are no other legally marketed treatments or products intended for treating or storing vascular grafts and that the technology has a novel intended use. Therefore, the applicant believed that DuraGraft® met the newness criterion because there no other legally marketed graft storage solutions on the market, there are no other products of this type with FDA market authorization, and that it has a novel intended use according to FDA.
With respect to CMS's concern regarding existing treatments for preventing ischemic injury of vein grafts during CABG surgery, the applicant stated that is not clear which treatments CMS is referring to as there are no legally marketed treatments or products intended for treating or storing vascular grafts besides DuraGraft®. The applicant further stated that while vascular grafts are placed in a liquid, usually Ringers Lactate, Plasmalyte or Normosol, to keep them from drying out between harvesting and implantation, in no way should these liquids be compared to or considered similar to DuraGraft®. The applicant also stated that these liquids cannot prevent ischemic or oxidative damage to vascular grafts. The applicant provided a table showing the components of competing wetting solutions to demonstrate that the solutions do not have the same molecules needed to prevent oxidative damage as DuraGraft®. The applicant stated that on the other hand, the mechanism of action of DuraGraft® as stated in the Instructions for Use (IFU) is through reduction of oxidative damage to maintain the structural and functional integrity of vascular conduits. The applicant asserted that Duragraft®'s primary function is to provide a reducing environment for vascular grafts to prevent oxidative damage which occurs during ischemic storage of grafts, using a combination of L-glutathione and L-Ascorbic acid that is manufactured in such a way as to preserve these molecules in their reduced state. The applicant concluded that, based upon composition, the stated mechanism of action, and preclinical evidence showing maintenance of the graft's structural and functional integrity provided on the DuraGraft® label, there are no similar products.
The applicant also stated that DuraGraft® is considered the first product of its type by FDA. Additionally, the applicant stated that in 2018, CMS established a new ICD-10-PCS code, XYOVX83 (Extracorporeal introduction of endothelial damage inhibitor to vein graft, new technology group 3), to report DuraGraft® when used in CABG procedures. The applicant stated that this ICD-10-PCS code would not be used with other procedures, outside of a few isolated instances. The applicant stated that claims submitted with this ICD-10-PCS code would be in error as DuraGraft® was not authorized or commercialized in the United States prior to October 2023.
A few commenters submitted comments stating that the mechanism of action is not substantially similar to existing technologies and that DuraGraft® has a novel mechanism of action in preventing oxidative damage to prevent ischemic injury and subsequent Ischemic Reperfusion Injury (IRI).
One commenter stated that they remained concerned that the information provided by the applicant does not show that DuraGraft® meets the newness criterion.
Response: We thank the applicant and the commenters for their comments. Based on our review of comments received and information submitted by the applicant as part of its FY 2025 new technology add-on payment application for DuraGraft®, we agree with the applicant and some of the commenters ( print page 69143) that DuraGraft® has a unique mechanism of action compared to other vein graft storage solutions because it creates a reducing environment for vascular grafts to prevent oxidative damage which occurs during ischemic storage of grafts. Therefore, we agree with the applicant that DuraGraft® is not substantially similar to existing treatment options and meets the newness criterion.
Comment: In response to our note in the proposed rule that DuraGraft® was not commercially available, the applicant responded that DuraGraft® is not yet commercially available but is expected to be available near the end of the second quarter of 2024.
Response: We thank the applicant for their response. As discussed in prior rulemaking ( 86 FR 45132 ; 77 FR 53348 ), generally, our policy is to begin the newness period on the date of FDA approval or clearance or, if later, the date of availability of the product on the US market. At this time, as there is not sufficient information to determine a newness date based on a documented delay in the technology's availability on the U.S. market, we consider the newness date for this technology to be October 4, 2023, the date it was granted De Novo classification from FDA.
With respect to the cost criterion, to identify potential cases representing patients who may be eligible for DuraGraft®, the applicant searched the FY 2022 MedPAR file for cases reporting a combination of ICD-10-CM/PCS codes that represent patients who underwent CABG procedures. Please see the online posting for DuraGraft® for a complete list of MS-DRGs and ICD-10-CM and -PCS codes provided by the applicant. Using the inclusion/exclusion criteria described in the following table, the applicant identified 33,511 cases mapping to 59 MS-DRGs, including MS-DRG 236 (Coronary Bypass Without Cardiac Catheterization Without MCC) representing 21.9 percent of the identified cases. The applicant followed the order of operations described in the following table and calculated a final inflated average case-weighted standardized charge per case of $321,620, which exceeded the average case-weighted threshold amount of $235,829. Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount, the applicant asserted that DuraGraft® meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36041 ), we noted the following concerns regarding the cost criterion. Although the applicant did not remove direct or indirect charges related to the prior technology, we noted that the applicant indicated that the use of DuraGraft® replaces solutions currently used for flushing and storage of the SVGs harvested through grafting, including tests for graft leakage, in its discussion of the newness criterion. Therefore, we questioned whether the cost criterion analysis should remove charges for related or prior technologies, such as autologous heparinized blood, Plasmalyte/Normosol, Lactated Ringers, and heparinized saline.
We invited public comments on whether DuraGraft® meets the cost criterion.
Comment: The applicant conducted two new cost analyses to address CMS's concerns about not removing direct or indirect charges related to prior technology. Specifically, the applicant removed 25 percent of the medical supply's charges including Plasmalyte/Noromosol, Lactated Ringers and Heparinized saline, and separately removed all blood charges. Under the first analysis, DuraGraft® exceeded the ( print page 69144) average case-weighted cost threshold amount by $75,125, and under the second analysis, Duragraft® exceeded the average case-weighted cost threshold amount by $85,777. The applicant stated that Duragraft® met the cost criterion as it exceeded the cost threshold with inclusion of the charges for related or prior technologies and when charges for the prior technologies are removed.
Response: We thank the applicant for its comments and new cost analyses. We agree that the final inflated average case-weighted standardized charges per case exceeded the average case-weighted threshold amounts. Therefore, DuraGraft® meets the cost criterion.
With regard to the substantial clinical improvement criterion, the applicant asserted that DuraGraft® represents a substantial clinical improvement over existing technologies because there is no other product or technology that reduces the incidence of peri-operative myocardial infarction. The applicant provided four studies to support this assertion, as well as 47 background articles about reducing major adverse cardiac events (MACE). [ 37 ] The following table summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for DuraGraft® for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36042 through 36043 ), after review of the information provided by the applicant, we stated we ( print page 69146) had the following concerns regarding whether DuraGraft® meets the substantial clinical improvement criterion. As discussed in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26800 through 26801 ), we expressed concern regarding the relatively small sample sizes of the Szalkiewicz, et al. (2022) [ 38 ] and Perrault, et al. (2021) [ 39 ] studies, as compared to the number of potentially eligible patients for this technology, and relatively short follow-up periods. We continued to question whether the sample was representative of the number of Medicare beneficiaries potentially eligible for DuraGraft®. We referred readers to the FY 2024 IPPS/LTCH PPS proposed rule for further discussion of these concerns. We also stated that, for its FY 2025 application, the applicant also cited Lopez-Menendez, et al. (2021), [ 40 ] which we noted used a sample size of 180, and therefore we similarly questioned whether the results of this study would be replicated with a larger patient sample.
Similar to the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26800 through 26801 ), we also questioned whether the results from the Haime, et al. (2018) [ 41 ] study could be generalized to other patient groups, including non-Veterans, women, or those from other racial or ethnic groups. We continued to question whether the demographic profiles in the Perrault, Szalkiewicz, and Haime studies that the applicant submitted were comparable with those of the U.S. Medicare patients who underwent CABG surgery. For its FY 2025 application, the applicant also cited the Lopez-Menendez, et al. (2021) [ 42 ] study, which was based on a European patient population that was predominantly male (82 percent to 90 percent). However, as we noted in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26800 through 26801 ), among the Medicare fee-for-service beneficiaries who underwent CABG surgery, male patients accounted for two-thirds (66 percent) of this population. Therefore, we stated that we continued to question whether the findings of these studies would be replicable among the Medicare population.
We invited public comments on whether DuraGraft® meets the substantial clinical improvement criterion.
Comment: The applicant reiterated the studies provided in its application in support of substantial clinical improvement for DuraGraft® and stated that the FDA label for DuraGraft® includes the following data from the Perrault study and the Internal Study Report: DuraGraft-treated SVGs had smaller wall thickness and lesser maximum focal narrowing at 12 months versus saline controls, [ 43 ] and reduced all-cause mortality at 3 years for Duragraft patients compared to patients who received SOC. [ 44 ]
In response to CMS's concerns regarding small sample sizes in the Perrault, Szalkiewicz, and Lopez-Menendez studies, the applicant stated that the sample size for the Perrault study was typical for a Class 2 medical device pivotal study under FDA, with 125 patients enrolled. The applicant stated that the study utilized a within-patient control design in that each patient received a DuraGraft® treated graft and a control graft, eliminating the need for a control group. The applicant also stated that the study was powered to observe changes in saphenous vein graft wall thickening, a surrogate endpoint in the pathophysiology of vein graft stenosis after CABG. Additionally, the applicant stated that the study, with each patient acting as their own control point, was powered to demonstrate a difference in the primary multidetector computed tomography (MDCT)-based endpoints. The applicant asserted that despite smaller sample sizes compared to those for drug studies, the size is typical for that of a medical device. Per the applicant, the study demonstrates important graft pathophysiology associated with the use of DuraGraft. The applicant further stated that the outcomes of each study are consistent with the growing body of data on DuraGraft® and is generalizable to the Medicare population. The applicant stated that the Szalkiewicz study was retrospective and powered to demonstrate the change in a surrogate endpoint of myocardial damage. The study was based on a sample size of 272 patients, with a mean age of 72 years (inter-quartile range (IQR): 62-75 years) in the DuraGraft® arm and 70 years (IQR: 61-76 years) in the control arm. The applicant stated the age is typical of CABG patients and the Medicare population. Regarding the Lopez-Menendez study, the applicant explained that this observational, prospective, single-center study had a follow-up period of 3 years and a sample size of 180 patients, 90 in the DuraGraft® arm and 90 in the control arm. The applicant stated that the study results showed a reduction of MACE in the DuraGraft® arm and, in a subset analysis, the use of DuraGraft® in diabetic patients was associated with better event-free survival. Additionally, the applicant stated that the observational study in the Marizyme internal study report had more than 5,000 patients and demonstrated DuraGraft®'s long-term treatment effects by comparing 3-year mortality from CABG patients in the DuraGraft® registry to a propensity matched cohort from the STS registry. The applicant asserted that the overall sample size of the patients treated with DuraGraft® in these four studies was 2,700.
The applicant also commented on CMS's concerns about the differences in the distribution of demographic characteristics between the patients in the Perrault, Haime, and Szalkewicz studies and the U.S. Medicare population. To address this concern, the applicant highlighted a head-to-head comparison (described in the DuraGraft IFU) comparing CABG patients on the European DuraGraft® registry, who were exposed to DuraGraft®, to those in the U.S. STS CABG registry, who were not. The applicant stated that outcomes from patients from the European DuraGraft® Registry (n=2522) were compared to randomly selected patients from the STS registry database in the U.S. who were operated on during the same period (n=294,725). The applicant stated that prior to propensity score ( print page 69147) matching, women comprised only 23.9 percent of the population undergoing first-time CABG in the U.S. during the time of the study, while men made up 76.1 percent. The applicant stated that the percentages were also similar to those of Medicare beneficiaries with the top 15 DRGs associated with cardiac surgery in the CMS MedPAR file data from 2022. The applicant stated that after propensity score matching, the sex distribution of the U.S. cohort became comparable to that of the European cohort, with 82.5 percent male and 17.5 percent female. The applicant asserted that the sex distribution of the patient population in the study was similar to that of Medicare patients undergoing first time CABG procedures at the time of the study. The applicant stated that the primary endpoint for the study was mortality through 3 years of follow-up. The applicant stated that the STS database contains data through 30-days after CABG surgery, and the STS' data was merged by STS with the national Death Index, a database maintained by the National Center for Health Statistics (NCHS) which captures all death records for the U.S. and U.S. territories, allowing mortality to be assessed for most STS patients beyond 30 days. The applicant asserted that the study results demonstrated a reduced mortality estimate in DuraGraft® patients at 3 years compared to STS patients. The applicant stated that this additional information should address CMS's concerns regarding DuraGraft® studies' applicability to the female Medicare population.
With respect to the Perrault study, the applicant stated it was a medical device study conducted with input from FDA, which involved radiologic assessment, using MDCT angiography, to evaluate graft morphology changes post-CABG comparing DuraGraft® treated grafts to saline controls. The applicant stated that some of the study results, reduction in wall thickening and reduction in maximal focal narrowing of the graft following use of DuraGraft®, have been allowed in the DuraGraft® label. The applicant stated that the study patient population was 91.2 percent male and 8.8 percent female and somewhat under-represented Medicare eligible women 8.9 percent versus 23.9 percent based on data in the STS database and CMS data for the timeframe. The applicant stated that the study still represented a study that included women, and taken together with the European DuraGraft® registry described earlier in this section, it was the applicant's conclusion that together these studies are representative of the Medicare eligible population. Additionally, the applicant stated that the average age of the patients, 66.2, was consistent with the age of a Medicare beneficiary.
With respect to the Haime study, the applicant stated that it was a single site study performed at the Boston Veterans Affairs (VA) hospital. According to the applicant, while the group was 99 percent male, the population of the VA hospitals typically represents one with elevated cardiac risk factors. The applicant stated that the characteristics of the study participants are typical for studies of CABG surgery and align with the Medicare population in terms of age, BMI, and presence of a diabetes diagnosis. The applicant stated that according to both CMS and STS data taken from the same time period in which both the Perrault and Registry comparison studies were performed, the percentage of Medicare eligible women undergoing CABG surgery ranges between 23.9 to 25.3 percent. The applicant stated that while the Perrault study included 8.9 percent women, the Registry comparison study patient population was 17.5 percent women. The applicant also stated that the percentage of women in the study reported by Lopez-Menendez (2023) was 17.8 percent, only a few percentage points lower than the percentage of Medicare eligible women undergoing CABG surgery based on both CMS and STS data.
The applicant commented that its current data does not allow an assessment of whether race changes the effect of DuraGraft® and acknowledged that the race of the patients in all the DuraGraft® studies was predominantly Caucasian. According to the applicant, in the Marizyme internal study report, it tried to isolate population differences and eliminate possible biases by propensity score matching based on 35 variables most strongly associated with surgical risk. The applicant cited the Shahian 2012 ASCERT study, [ 45 ] which identified perioperative as well as longer-term predictors of mortality post-CABG. The applicant also asserted that race and sex have been demonstrated in these multiple studies as lesser determinants of post-CABG mortality compared to other important risk factors such as age, congestive heart failure, chronic obstructive pulmonary disease, renal failure, myocardial infarction, and emergent operative status.
Another commenter cited a study by Gaudino and team (2023), [ 46 ] which examined outcomes in women undergoing CABG in the US from 2011-2020. According to the commenter, the authors of the study acknowledged that women have been chronically underrepresented in cardiology studies, but stated that there is no biological evidence for gender-based differential effect of DuraGraft® solution. The commenter further stated that CAD is becoming more common among women, which makes them amenable to medical and invasive treatment options, like CABG surgery. The commenter also referred to a Goldstein 2022 study, [ 47 ] which examined the effects of an external saphenous vein graft support device on reducing intimal hyperplasia and graft failure in patients undergoing CABG, and stated that 20.5 percent of the patients in that study were women.
One commenter stated that the study design for the Perrault study, in which each patient received a DuraGraft® treated graft and a control graft to control for patient-specific graft effects, obviated the need for a separate control patient cohort, thereby decreasing the sample size required for the study. The commenter asserted that the study was powered appropriately as the MDCT scanning measurements were taken every 10mm along the whole of the grafts at 1, 3, and 12 months for the endpoints of the study.
One commenter expressed concern that the data received to date did not support the substantial clinical improvement criterion for DuraGraft®.
Response: We thank the applicant and the commenters for their additional input. After review of the comments and all data received to date, we continue to have concerns that the evidence submitted does not support that the technology meets the substantial clinical improvement criterion.
We continue to question whether the patient samples in the studies provided are representative of the Medicare population, as this could impact the extent to which the results related to DuraGraft®'s effects on clinical outcomes could be generalized to the ( print page 69148) Medicare population, including those who may potentially need CABG surgery or be eligible for DuraGraft®. According to the applicant, because the matched STS cohort, with 82.5 percent males and 17.5 percent females, was equivalent to the European DuraGraft® Registry, it was very similar to the sex demographics of Medicare patients undergoing first time CABG procedures at the time of the study. However, as we noted in the FY 2024 and FY 2025 IPPS/LTCH PPS proposed rules ( 88 FR 26801 and 89 FR 36043 ), male patients accounted for only 66 percent of all CABG patients on Medicare, while women accounted for the remaining 34 percent, and statistics have shown that women tend to have poorer outcomes after CABG surgery. We are also concerned that study results, based on predominantly male and white samples, may not be replicable among the Medicare population, especially since the proportion of racial and ethnic minorities has continued to grow, from 20.9 percent in 2008 to 27.2 percent in 2022. [ 48 ] While the applicant asserted that race and sex were demonstrated in the studies provided to be lesser determinants of post-CABG mortality compared to other important risk factors, such as age, congestive heart failure, chronic obstructive pulmonary disease, renal failure, myocardial infarction (MI) and emergent operative status, the applicant also commented that these factors were not well represented in the same studies, such that it is unclear how they could be accurately demonstrated as a lower determinant of mortality. We note that the poorer clinical outcomes of female and minority CABG patients are well-documented in the literature. Compared to male CABG patients, female CABG patients had worse post-CABG outcomes, including a higher rate of unplanned readmissions within 30 or 90 days post-discharge, inpatient mortality, major cardiovascular and cerebrovascular adverse events (MACCE), like non-fatal MI, transient ischemic attack, cardiovascular-related mortality, and all-cause mortality. [ 49 50 51 ] Compared to white CABG patients, African American CABG patients have a higher risk for poor post-CABG outcomes, like longer length of inpatient stay, MACCE, and all-cause mortality. [ 52 53 ] Study results based on patient samples in which women or racial and ethnic minorities were under-represented may limit our ability to generalize the findings to a population with different clinical characteristics. [ 54 55 ]
Furthermore, we continue to have concerns about the sample sizes in some of the studies referenced. Based on the information provided, we remain unclear about how the sample sizes were calculated and the parameters used in the calculations, such as the size of the populations that the samples represented, the effect size, [ 56 ] and how much of a difference in outcomes was clinically meaningful and reflected a true effect between those who were exposed to Duragraft® and those who were not. [ 57 ]
As noted, the applicant stated that the Perrault study showed that DuraGraft®-treated SVGs had smaller mean wall thickness and lesser maximum focal narrowing at 12 months versus saline controls, and that these results were included in the FDA label for DuraGraft®. The applicant stated that the Perrault study was powered to observe changes in SVG wall thickening, which it stated is a surrogate endpoint in the pathophysiology of vein graft stenosis. While we acknowledge that the study demonstrated smaller wall thickness and lesser maximum focal narrowing at 12 months for DuraGraft®-treated SVGs versus saline controls, as included in the FDA label, we also agree that these are surrogate endpoints. As the study was not powered to demonstrate an improved clinical outcome as described under the regulations at 412.87(b)(1)(ii), we do not believe the inclusion of these findings from this study support a finding of substantial clinical improvement. We also note that we do not believe that the inclusion of outcomes on the FDA label by itself supports a finding of substantial clinical improvement. In addition, the Perrault study compared DuraGraft® to saline controls. However, as previously noted, there are other vein graft preservation solutions, including but not limited to Plasmalyte, Normosol, lactated ringers, and heparinized autologous blood, to which DuraGraft® was not compared. We further note that studies have shown that these solutions have differing effects on graft endothelium. [ 58 59 ] We are unclear how improvements demonstrated by use of DuraGraft® as compared to saline controls demonstrate substantial clinical improvement over other existing technologies without an assessment of comparative outcomes to the other vein graft preservation solutions.
With regard to the applicant's assertions that results of the Marizyme internal study report showing reduced mortality at three years in DuraGraft® patients were included in the FDA label, we note that the primary endpoint was mortality through 1 year of follow up. While the applicant stated in its comment that the study results demonstrated a reduced mortality estimate at 3 years for Duragraft®-treated patients compared to SOC controls, we note that differences in mortality were not statistically significant at the primary endpoint of 1 year. Similarly, we do not believe the inclusion of outcomes on the FDA label by itself supports a finding of substantial clinical improvement. In addition, although we acknowledge that, as stated by the applicant, it tried to isolate population differences and eliminate possible biases by propensity score matching ( print page 69149) based on 35 variables most strongly associated with surgical risk, we note that propensity scoring can only control for confounding factors that are measured. Unmeasured confounding factors could still impact the association between exposure to DuraGraft® and clinical outcomes. In particular, it does not appear that intra-operative or post-operative factors that could impact vein integrity or post-CABG outcomes were accounted for in this study (or in other studies provided by the applicant, as we had previously noted in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26801 )) including vein distention pressure, time of intra-operative SVG ischemia, or post-operative antiplatelet therapy or lipid-lowering drugs. We further note that the Marizyme internal study report is unpublished, and it is unclear from the report which vein preservation solutions were included in the control arm as the report only states that SOC solution was used. Further, the study only reports all-cause mortality and does not specify how many patients had mortality due to other causes that could not be attributed to use of a vein preservation solution other than DuraGraft®. Therefore, although the applicant stated that this additional information should address CMS's concerns regarding DuraGraft® studies' applicability to the female Medicare population, we continue to question the generalizability of any outcomes associated with use of DuraGraft®.
Therefore, after consideration of the public comments we received and based on the information stated previously, we are unable to determine that DuraGraft® represents a substantial clinical improvement over existing therapies, and we are not approving new technology add-on payments for DuraGraft® for FY 2025.
Two manufacturers, Pfizer, Inc. and Johnson & Johnson Health Care Systems, Inc. submitted separate applications for new technology add-on payments for FY 2025 for ELREXFIO TM and TALVEY TM , respectively. Both of these technologies are bispecific antibodies (bsAb) used for the treatment of adults with relapsed or refractory multiple myeloma (RRMM) who have received at least four prior lines of therapy, including a proteasome inhibitor (PI), an immunomodulatory (IMiD), and an anti-CD38 monoclonal antibody (mAb). ELREXFIO TM is a B-cell maturation antigen (BCMA) directed cluster of differentiation (CD)3 T-cell engager. Per the applicant, ELREXFIO TM is a bispecific, humanized immunoglobulin 2-alanine (IgG2Δa) kappa antibody derived from two mAbs, administered as a fixed-dose, subcutaneous treatment. TALVEY TM is a G protein-coupled receptor, class C, group 5, member D (GPRC5D) targeting bsAb. GPRC5D is an orphan receptor expressed at a significantly higher level on malignant multiple myeloma (MM) cells than on normal plasma cells. We note that Pfizer, Inc. submitted an application for new technology add-on payments for ELREXFIO TM for FY 2024 under the name elranatamab, as summarized in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26803 through 26809 ), but the technology did not meet the July 1, 2023, deadline for FDA approval or clearance of the technology and, therefore, was not eligible for consideration for new technology add-on payments for FY 2024 ( 88 FR 58804 ).
In the FY 2025 IPPS/LTCH PPS proposed rule ( 86 FR 36043 through 36052 and 36087 through 36092 ), we discussed these applications as two separate technologies. However, after further consideration and as discussed later in this section, we believe ELREXFIO TM and TALVEY TM are substantially similar to each other and that it is appropriate to evaluate both technologies as one application for new technology add-on payments under the IPPS.
Please refer to the online application posting for ELREXFIO TM available at https://mearis.cms.gov/public/publications/ntap/NTP2310176PV9B , for additional detail describing the technology and the disease treated by the technology.
Please refer to the online application posting for TALVEY TM , available at https://mearis.cms.gov/public/publications/ntap/NTP2310163HW2V , for additional detail describing the technology and the disease treated by the technology.
With respect to the newness criterion, the applicant for ELREXFIO TM stated that ELREXFIO TM was granted Biologics License Application (BLA) approval from FDA on August 14, 2023, for the treatment of adult patients with RRMM who have received at least four prior lines of therapy, including a PI, an IMiD, and an anti-CD38 mAb. According to the applicant, ELREXFIO TM was commercially available immediately after FDA approval. Per the applicant, the recommended doses of ELREXFIO TM subcutaneous injection are step-up doses of 12 mg on day 1 and 32 mg on day 4, followed by a first treatment dose of 76 mg on day 8 and subsequent treatment doses as indicated on the label. The applicant noted that treatment doses may be administered in an inpatient or outpatient setting. Per the applicant, patients should be hospitalized for 48 hours after administration of the first step-up dose, and for 24 hours after administration of the second step-up dose. The applicant assumed that there would be a single inpatient stay, with one 44 mg vial used per dose, resulting in two doses (each a step-up dose) being administered.
The applicant for TALVEY TM stated that TALVEY TM was granted BLA approval from FDA on August 9, 2023, for the treatment of adult patients with RRMM who have received at least four prior lines of therapy, including a PI, an IMiD, and an anti-CD38 mAb. According to the applicant, TALVEY TM was commercially available immediately after FDA approval. Per the applicant, patients may be dosed on a weekly or bi-weekly dosing schedule. The applicant noted that patients on a weekly dosing schedule receive three weight-based doses—a 0.01 mg/kg loading dose, a 0.06 mg/kg loading dose, and the first 0.40 mg/kg treatment dose—during the hospital stay; patients on a bi-weekly dosing schedule receive an additional 0.80 mg/kg treatment dose during the hospital stay.
The applicant for ELREXFIO TM stated that effective October 1, 2023, the following ICD-10-PCS code may be used to uniquely describe procedures involving the use of ELREXFIO TM : XW013L9 (Introduction of elranatamab antineoplastic into subcutaneous tissue, percutaneous approach, new technology group 9). The applicant stated that C90.00 (Multiple myeloma not having achieved remission), C90.01 (Multiple myeloma in remission), C90.02 (Multiple myeloma in relapse), and Z51.12 (Encounter for antineoplastic immunotherapy) may be used to currently identify the indication for ELREXFIO TM under the ICD-10-CM coding system.
The applicant for TALVEY TM submitted a request for approval for a unique ICD-10-PCS procedure code for TALVEY TM and was granted approval for the following procedure code effective April 1, 2024: XW01329 (Introduction of talquetamab antineoplastic into subcutaneous tissue, percutaneous approach, new technology group 9). The applicant stated that ICD-10-CM codes C90.00 (Multiple myeloma not having achieved remission) and C90.02 (Multiple myeloma in relapse) may be used to currently identify the indication for TALVEY TM .
As stated earlier and for the reasons discussed further later in this section, we believe that ELREXFIO TM and ( print page 69150) TALVEY TM are substantially similar to each other such that it is appropriate to analyze these two applications as one technology for purposes of new technology add-on payments, in accordance with our policy. We also discuss in this section the information provided by the applicants, as summarized in the proposed rule, regarding whether ELREXFIO TM and TALVEY TM are substantially similar to existing technologies. As discussed earlier, if a technology meets all three of the substantial similarity criteria, it would be considered substantially similar to an existing technology and would not be considered “new” for purposes of new technology add-on payments.
With respect to the substantial similarity criteria, in its application for new technology add-on payments for FY 2025, the applicant for ELREXFIO TM asserted that ELREXFIO TM is not substantially similar to other currently available technologies because it is the only therapy approved for the treatment of patients with RRMM who have received four prior lines of therapy including a PI, IMiD, and mAb, that uses a humanized IgG2Δa antibody for the mechanism of action. Per the applicant, it is also the only BCMA-directed bsAb therapy with clinical study data in its prescribing information supporting its use in patients who have received prior BCMA-directed therapy and that, therefore, the technology meets the newness criterion. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for ELREXFIO TM for the applicant's complete statements in support of its assertion that ELREXFIO TM is not substantially similar to other currently available technologies.

With respect to the substantial similarity criteria, in its application for new technology add-on payments for FY 2025, the applicant for TALVEY TM asserted that TALVEY TM is not substantially similar to other currently ( print page 69152) available technologies because it has a unique mechanism of action as a CD3 T-cell engaging bsAb targeting GPRC5D, and therefore, the technology meets the newness criterion. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for TALVEY TM for the applicant's complete statements in support of its assertion that TALVEY TM is not substantially similar to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 86 FR 36046 through 36047 and 36088 through 36089 ), we stated that we believed ELREXFIO TM and TALVEY TM may be substantially similar to each other. We stated that per the applications for ELREXFIO TM AND TALVEY TM , both are bispecific antibodies approved for the treatment of adults with RRMM who have received at least four prior lines of therapy, including a PI, IMiD, and an antiCD38 monoclonal antibody. We noted that per the applicant for ELREXFIO TM , ELREXFIO TM is as a bsAb that uses binding domains that simultaneously bind the BCMA target on tumor cells and the CD3 T-cell receptor. We also noted that the applicant for TALVEY TM stated that TALVEY TM is the only medicine that targets GPRC5D on myeloma cells and that because TALVEY TM binds to different receptors, it has a different mechanism of action from ELREXFIO TM . However, we questioned how binding to a different protein (GPRC5D) on the tumor cell would result in a different mechanism of action compared to BCMA targeting bsAbs. Furthermore, we noted that the applicant for TALVEY TM claimed that GPRC5D, the target of TALVEY TM , has a unique tissue expression profile, which results in an adverse event profile distinct from those of the currently approved bsAb in RRMM ( print page 69153) targeting BCMA. However, as this is related to the risk of adverse events from TALVEY TM administration but was not critical to the way the drug treats the underlying disease, we questioned whether this therefore related to an assessment of substantial clinical improvement rather than of substantial similarity. We stated that we would welcome additional information on how molecular differences, such as the regulation of expression of GPRC5D and BCMA on MM cells during treatment, should be considered in determining whether a technology utilizes a different mechanism of action to achieve a therapeutic outcome. Additionally, we noted that similar to TALVEY TM , the prescribing information for ELREXFIO TM includes a population with prior exposure to BCMA T-cell redirection therapy.
Accordingly, we noted that, as it appears that ELREXFIO TM and TALVEY TM would use the same or similar mechanism of action to achieve a therapeutic outcome, would be assigned to the same MS-DRG, and would treat the same or similar disease in the same or similar patient population, we believed that these technologies may be substantially similar to each other such that they should be considered as a single application for purposes of new technology add-on payments. We noted that if ELREXFIO TM and TALVEY TM were determined to only be substantially similar to each other, we believed the newness period for ELREXFIO TM and TALVEY TM would begin on August 9, 2023, the date TALVEY TM received FDA approval.
We also stated that we believed ELREXFIO TM and TALVEY TM may be substantially similar to TECVAYLI®, for which we approved an application for new technology add-on payments for FY 2024 ( 88 FR 58891 ) for the treatment of adult patients with RRMM after four or more prior lines of therapy, including a PI, an IMiD, and an anti-CD38 mAb. We noted that TECVAYLI®'s mechanism of action is described as a bsAb, with binding domains that simultaneously bind the BCMA target on tumor cells and the CD3 T-cell receptor ( 88 FR 58886 ). We stated that the applicant for ELREXFIO TM asserted that ELREXFIO TM has a unique CDR (the region of antibody that recognizes and binds to target epitopes) that is critical to the mechanism of action because it results in different targeted regions, impacting how the drug works to target the cancer cells. However, we noted it was unclear whether these differences result in a substantially different mechanism of action from TECVAYLI®. We stated that because of the apparent similarity with the bsAb for ELREXFIO TM that uses binding domains that simultaneously bind the BCMA target on tumor cells and the CD3 T-cell receptor, we believed that the mechanism of action for ELREXFIO TM may be the same or similar to that of TECVAYLI®. We noted that the applicant for ELREXFIO TM also asserted that ELREXFIO TM is different from TECVAYLI® because the two are based on different immunoglobulin isotypes, and with the lower effector function of IgG2, ELREXFIO TM should only activate T-cells in the presence of BCMA and thus should only stimulate an immune response in the tumor. Based on our understanding, however, that this may relate to the risk of adverse event from ELREXFIO TM administration but was not critical to the way the drug treats the underlying disease, we questioned whether this would therefore relate to an assessment of substantial clinical improvement, rather than of substantial similarity.
We also noted that per the applicant for TALVEY TM , TALVEY TM has a different mechanism of action from TECVAYLI® or ELREXFIO TM because it binds to different receptors. The applicant for TALVEY TM noted that TALVEY TM is the only medicine that targets GPRC5D on myeloma cells. As we previously noted, TECVAYLI®'s mechanism of action is described as a bsAb, with binding domains that simultaneously bind the BCMA target on tumor cells and the CD3 T-cell receptor ( 88 FR 58886 ). As discussed previously, the applicant asserted that TALVEY TM had a unique mechanism of action as compared to TECVAYLI® and ELREXFIO TM by binding to different receptors. However, we questioned how binding to a different protein (GPRC5D) on the tumor cell would result in a different mechanism of action compared to BCMA targeting bispecific antibodies. Furthermore, as we noted previously, the applicant for TALVEY TM claimed that the target of TALVEY TM , GPRC5D, has a unique tissue expression profile, which results in an adverse event profile distinct from those of the currently approved bispecific antibodies in RRMM targeting BCMA. However, we noted that this was related to the risk of adverse event from TALVEY TM administration but was not critical to the way the drug treats the underlying disease. As a result, we questioned whether this would therefore relate to an assessment of substantial clinical improvement rather than of substantial similarity. We also stated previously that we would welcome additional information on how molecular differences, such as the regulation of expression of GPRC5D and BCMA on MM cells during treatment, should be considered in determining whether a technology utilizes a different mechanism of action to achieve a therapeutic outcome.
We also noted that ELREXFIO TM , TALVEY TM , and TECVAYLI® may treat the same or similar disease (RRMM) in the same or similar patient population (patients who have previously received a PI, IMiD, and an anti-CD38 mAb). The applicant for ELREXFIO TM claimed that ELREXFIO TM is different from TECVAYLI® because the prescribing information includes a new subpopulation: the patient population that had received prior BCMA-directed therapy. However, we believed that the lack of inclusion of this population in the prescribing information for TECVAYLI® does not necessarily exclude the use of TECVAYLI® in this patient population, nor does FDA prescribing information for TECVAYLI® specifically exclude this patient population. As such, we stated it was unclear whether ELREXFIO TM would in fact treat a patient population different from TECVAYLI®. Accordingly, we noted that as it appears that ELREXFIO TM , TALVEY TM , and TECVAYLI® may be using the same or similar mechanism of action to achieve a therapeutic outcome, would be assigned to the same MS-DRG, and treat the same or similar patient population and disease, we believed that these technologies may be substantially similar to each other. We noted that if we determined that these technologies were substantially similar to TECVAYLI®, we believed the newness period for these technologies would begin on November 9, 2022, the date TECVAYLI® became commercially available.
We stated we were interested in receiving information on how these technologies may differ from each other with respect to the substantial similarity and newness criteria to inform our analysis of whether ELREXFIO TM , TALVEY TM , and TECVAYLI ® are substantially similar to each other.
We invited public comments on whether ELREXFIO TM and TALVEY TM are substantially similar to each other and/or existing technologies and whether ELREXFIO TM and TALVEY TM meet the newness criterion.
Comments: The applicant for ELREXFIO TM submitted a comment regarding the newness criterion. The applicant stated that, based on CMS's newness criteria, it would agree ELREXFIO TM , TECVAYLI ®, and TALVEY TM are all substantially similar ( print page 69154) according to new technology add-on guidelines because they all are bsAb therapies using a mechanism of action that simultaneously binds to a protein on the tumor cell and the CD3 T-cell receptor, bridging the two to kill the tumor cell. Additionally, the applicant commented that the indication statements for all the therapies are for the treatment of adult patients with RRMM who have received prior therapy and are all assigned to the same MS-DRG. The applicant commented that since TECVAYLI ® was approved for new technology add-on payment status effective FY 2024 with a newness period beginning November 9, 2022, CMS should continue new technology add-on payment status for FY 2025 and extend that new technology add-on payment status to ELREXFIO TM.
The applicant for TALVEY TM commented that it agreed with CMS that TALVEY TM , TECVAYLI®, and ELREXFIO TM are all approved for the treatment of the same disease (RRMM) and in a similar patient population (patients receiving a PI, IMiD, and an anti-CD38 mAb. However, the applicant did not agree that TALVEY TM has a similar mechanism of action due to the targeting of different antigens on the surface of malignant plasma cells. According to the applicant, TALVEY TM is unique in targeting GPRC5D instead of BCMA and has demonstrated efficacy in patients who are naive to or exposed to prior bsAb and CAR T-cell therapy. According to the applicant, this emerging population of patients who have been exposed to all other major treatment classes (PI, IMiD, anti-CD38 mAb, and BCMA) is increasingly important, and TALVEY TM has demonstrated efficacy and safety in this growing patient population with an unmet medical need. While GPRC5D and BCMA may have similar expression on plasma cells, the applicant for TALVEY TM stated that the pattern of expression of GPRC5D and BCMA are independent of each other, making GPRC5D a distinct clinical target. Additionally, the applicant asserted that GPRC5D is primarily expressed on malignant plasma cells, while BCMA is also widely expressed on the surface of mature B cells and normal plasma cells, which differentiates the mechanism of action and AE profile of TALVEY TM from those of BCMA targeting therapies. The applicant also stated that TALVEY TM demonstrated an overall response rate of 72 percent (with a durable 9-month duration of response rate of 59 percent) in 32 patients who had received prior T-cell redirection therapy, including 30 patients who had received a prior BCMA CAR-T or bispecific therapy. The applicant added that the limited expression of GPRC5D on normal B cells and plasma cells resulted in serious infections and grade 3-4 infections in 16 percent and 17 percent of patients respectively, compared to 30 percent and 34 percent for TECVAYLI TM and 31 percent and 42 percent for ELREXFIO TM .
The applicant for TALVEY TM further commented that a recent publication by Firestone and team (2024) [ 60 ] described antigen escape, or loss of target antigen, as a mechanism of resistance to BCMA-directed therapies in RRMM. The Firestone study stated that one of the patients who was BCMA refractory responded to TALVEY TM . The applicant for TALVEY TM asserted that because expression of GPRC5D is independent of BCMA, and those two bsAbs target distinct, independent antigens, TALVEY TM can be used to treat patients who have progressed on or do not respond to TECVAYLI ®.
Response: We thank the applicants for their comments regarding the newness criterion for ELREXFIO TM and TALVEY TM . While we agree with the applicant for TALVEY TM that TALVEY TM treats a similar population to ELREXFIO TM and TECVAYLI®, we disagree that TALVEY TM is unique in treating patients who are naïve to or exposed to prior bsAb and CAR T-cell therapy. As previously discussed, ELREXFIO TM is also indicated for patients exposed to prior bsAb and CAR T-cell therapy. We also disagree with that TALVEY TM has a unique mechanism of action. While the applicant for TALVEY TM stated that GPRC5D is primarily expressed on malignant plasma cells and that BCMA is also widely expressed on the surface of mature B cells and normal plasma cells, the applicant for TALVEY TM did not demonstrate how this difference affects the mechanism of action by which patients are treated. We note that the applicant for TALVEY TM seemed to assert that a difference in clinical outcomes demonstrates a difference in the mechanism of action from existing technologies. However, we note that a difference in observed outcomes would relate to an assessment of substantial clinical improvement rather than the newness criterion. Therefore, we remain unclear how binding to a different protein (GPRC5D) on the tumor cell affects the downstream molecular process by which TALVEY TM treats the underlying disease compared to ELREXFIO TM and TECVAYLI®.
After consideration of the comments received, and for the reasons discussed, we believe that ELREXFIO TM , TALVEY TM , and TECVAYLI® use the same or a similar mechanism of action to achieve therapeutic outcomes. Furthermore, as discussed previously, ELREXFIO TM and TALVEY TM map to the same MS-DRG and treat the same patient population (adult patients with RRMM after four or more prior lines of therapy, including a PI, an IMiD, and an anti-CD38 mAb) as TECVAYLI®. Accordingly, because ELREXFIO TM and TALVEY TM meet all three of the substantial similarity criteria, we believe that both are substantially similar to TECVAYLI®. In accordance with our policy, because these technologies are substantially similar to each other, we use the earliest market availability date submitted as the beginning of the newness period for these technologies. Therefore, we consider the beginning of the newness period for ELREXFIO TM and TALVEY TM to be November 9, 2022, the date TECVAYLI® became commercially available.
Consistent with our policy statements in the past regarding substantial similarity, we will not be making a determination on cost and substantial clinical improvement for ELREXFIO TM or TALVEY TM . Specifically, we have noted that approval of new technology add-on payments would extend to all technologies that are substantially similar, and if substantially similar technologies are submitted for review in different (and subsequent) years, we evaluate and make a determination on the first application and apply that same determination to the second application ( 85 FR 58679 ). Since TECVAYLI® was approved for new technology add-on payments for FY 2024 and is still within its newness period for FY 2025, and we have determined that ELREXFIO TM and TALVEY TM are substantially similar to TECVAYLI®, we apply that same approval for new technology add-on payments to ELREXFIO TM and TALVEY TM . We note that we received public comments with regard to the cost and substantial clinical improvement criteria for these technologies, but because the determination made in the FY 2024 IPPS/LTCH PPS final rule for TECVAYLI® is applied to ELREXFIO TM and TALVEY TM due to their substantial similarity, we are not summarizing comments received or making a determination on those criteria in this final rule.
Cases involving the use of ELREXFIO TM that are eligible for new ( print page 69155) technology add-on payments will be identified by ICD-10-PCS code XW013L9 (Introduction of elranatamab antineoplastic into subcutaneous tissue, percutaneous approach, new technology group 9). Cases involving the use of TALVEY TM that are eligible for new technology add-on payments will be identified by ICD-10-PCS code XW01329 (Introduction of talquetamab antineoplastic into subcutaneous tissue, percutaneous approach, new technology group 9).
Each of the applicants submitted cost information for its application. The manufacturer of ELREXFIO TM estimated that the cost of ELREXFIO TM is $15,112 per patient. The manufacturer of TALVEY TM estimated that the cost of TALVEY TM is $25,164.44 per patient. Because ELREXFIO TM and TALVEY TM are substantially similar to TECVAYLI®, we believe using a single cost for purposes of determining the new technology add-on payment amount is appropriate for ELREXFIO TM , TALVEY TM , and TECVAYLI®, even though each technology will be identified by its own ICD-10-PCS code ( 87 FR 48925 ). We also believe using a single cost provides predictability regarding the add-on payment when using ELREXFIO TM , TALVEY TM , and TECVAYLI® for the treatment of patients with RRMM. As such, we believe that the use of a weighted average of the cost of ELREXFIO TM , TALVEY TM , and TECVAYLI® based upon the projected numbers of cases involving each technology to determine the maximum new technology add-on payment would be most appropriate. To compute the weighted average cost, we summed the total number of projected cases for each technology provided by the applicants, which equaled 4,376 cases (152 cases for ELREXFIO TM plus 2,318 cases for TALVEY TM plus 1,906 cases for TECVAYLI®). We then divided the number of projected cases for each of the technologies by the total number of cases, which resulted in the following case weighted percentages: 3.47 percent for ELREXFIO TM , 52.97 percent for TALVEY TM and 43.56 percent for TECVAYLI®. For each technology, we then multiplied the estimated cost per patient by the case-weighted percentage (0.0347 * $15,112 = $524.39 for ELREXFIO TM ; 0.5297 * $25,164.44 = $13,329.60 for TALVEY TM ; and 0.4356 * $13,754.67 = $5,991.53 for TECVAYLI®). This resulted in a case-weighted average cost of $19,845.52 for the technology.
Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, the maximum new technology add-on payment for a case involving the use of ELREXFIO TM , TALVEY TM , or TECVAYLI® is $12,899.59 for FY 2025.
Flosonics Medical (R.A. 1929803 Ontario Corp.) submitted an application for new technology add-on payments for FloPatch FP120 for FY 2025. According to the applicant, FloPatch FP120 is a wireless, wearable, continuous wave (4 MHz) Doppler ultrasound device that adheres over peripheral vessels (that is, carotid and jugular) that assesses blood flow in the peripheral vessels, enabling rapid and repeatable dynamic assessments of both arterial and venous flow simultaneously. According to the applicant, FloPatch FP120 cardiovascular blood flowmeter adheres to a patient's neck (or any other major vessel) and transmits Doppler-shifted ultrasonic waves from the transducer to the artery and vein at a fixed angle of insonation that are then reflected by moving blood cells back to the transducer. Per the applicant, the signal processing unit wirelessly outputs data to a secure iOS mobile medical application, which displays metrics from the Doppler signal, such as maximal velocity trace and corrected flow time, in a user-friendly interface. Per the applicant, FloPatch FP120 will optimize clinical workflow, is easy-to-use and hands-free, cloud-connected, and can be deployed in under one minute, providing instantaneous results.
Please refer to the online application posting for FloPatch FP120, available at https://mearis.cms.gov/public/publications/ntap/NTP231017D56F4 , for additional detail describing the technology and the types of conditions that the technology might help diagnose and/or treat.
With respect to the newness criterion, according to the applicant, FloPatch FP120 received 510(k) clearance from FDA on May 3, 2023, for the noninvasive assessment of blood flow in the carotid artery. Per the applicant, in a more recent FDA 510(k) submission, the proposed indication is for use for the noninvasive assessment of blood flow in peripheral vasculature. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36052 ), we stated that based on the application submitted by the applicant, the new technology add-on payment application for FloPatch FP120 is not eligible for consideration for FY 2025 for the proposed indication (for use for the noninvasive assessment of blood flow in peripheral vasculature) because documentation of FDA acceptance or filing of the marketing authorization request that indicates that FDA has determined that the application is sufficiently complete to allow for substantive review by FDA, was not provided to CMS at the time of new technology add-on payment application submission. As such, we stated that the new technology add-on payment application for FloPatch FP120 is only eligible for consideration for FY 2025 for the narrower indication for use for the noninvasive assessment of blood flow in carotid artery.
In the proposed rule ( 89 FR 36052 ), we noted that prior to the May 3, 2023, clearance, there were two FDA 510(k) clearances for FloPatch FP120; one obtained in 2022 and one in 2020. The indications in the 2020, 2022, and 2023 clearances are identical, that is, for use for the noninvasive assessment of blood flow in the carotid artery. [ 61 ] In addition, the 2020 clearance was based on substantial equivalence to the FloPatch FP110 device, [ 62 ] which was an earlier version of FloPatch FP120 and was also FDA-cleared. According to the applicant, FloPatch FP120 was commercially available for this use as of January 1, 2023. However, we stated that, as noted earlier, the provided FDA 510(k) clearance was dated May 3, 2023. Because the market availability date as indicated by the applicant preceded the 2023 clearance date, and because the 2020 and 2022 clearances had the same indication as the 2023 clearance, we questioned when the technology first became commercially available for use for the noninvasive assessment of blood flow in the carotid artery and requested additional information on the market availability date for this indication. Per the applicant, one FloPatch FP120 device would be used per inpatient stay.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for FloPatch FP120 and was granted approval to use the following procedure code effective October 1, 2024: XX25X0A (Monitoring of blood flow, adhesive ultrasound patch technology, new technology group 10). The applicant provided a list of diagnosis codes that may be used to currently identify the indication for FloPatch FP120 under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
As previously discussed, if a technology meets all three of the substantial similarity criteria under the ( print page 69156) newness criterion, it would be considered substantially similar to an existing technology and would not be considered new for the purpose of new technology add-on payments.
With respect to the substantial similarity criteria, the applicant asserted that FloPatch FP120 is not substantially similar to other currently available technologies because FloPatch FP120 offers real-time, non-invasive monitoring of hemodynamic changes of both the arterial and venous blood flow, improving fluid management decisions. Per the applicant, FloPatch FP120 surpasses current methods by providing continuous data, enhancing patient safety, and addressing unmet clinical needs for immediate, precise assessments, and therefore, the technology meets the newness criterion. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for FloPatch FP120, for the applicant's complete statements in support of its assertion that FloPatch FP120 is not substantially similar to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36054 ), we noted the following concerns with regard to the newness criterion. With respect to the first substantial similarity criterion, whether FloPatch FP120 uses the same or similar mechanism of action for a therapeutic outcome when compared to existing technologies, we stated we had not received information from the applicant regarding predicate devices for FloPatch FP120 that were previously FDA-cleared in its discussion of existing technologies. We stated that there are three FDA 510(k) clearances for FloPatch FP120, with the same indication for use for the noninvasive assessment of blood flow in the carotid artery. [ 63 ] In addition, the 2020 clearance was based on substantial equivalence to the FloPatch FP110 device, [ 64 ] an earlier version of FloPatch FP120 that was also FDA-cleared. We noted that all FloPatch FP120 FDA-cleared devices, as well as the FP110 version, had an identical method of attachment of the ultrasound probe to the human body, and the same intended use and indications for use. Accordingly, we stated that since the technology was already approved for use for this same indication outside of the 2- to 3-year newness period, it appeared that it would no longer be considered new for purposes of new technology add-on payments.
In addition, we questioned whether a different placement method or the addition of a wearable functionality for the noninvasive assessment of blood flow would constitute a different mechanism of action, and whether these differences may instead be relevant to the assessment of substantial clinical improvement, rather than of newness. For example, while the applicant described FloPatch FP120 as user-friendly, we questioned whether ease-of-use in itself represented a mechanism of action unique from existing technologies for a therapeutic outcome, as the primary underlying mechanism of action is still Doppler ultrasound technology.
With respect to the second substantial similarity criterion, that is, whether a product is assigned to the same or a different MS-DRG, the applicant asserted that the device is new and has not undergone sufficient review to be recognized as a treatment within the existing MS-DRGs. However, we noted that the applicant also stated that FloPatch FP120 could be relevant to existing MS-DRGs that pertain to septicemia or severe sepsis for the assessment of volume responsiveness. We stated we believed that, based on its indication, cases involving the use of FloPatch FP120 would be assigned to the same MS-DRGs as those involving existing technologies used for invasive and non-invasive measurements of blood flow, such as for patients with septicemia or severe sepsis.
With respect to the third substantial similarity criterion, that is, whether the technology involves treatment of the same or similar type of disease or patient population when compared to an existing technology, the applicant maintained that existing technologies do not provide clinicians with the information they need, and while FloPatch FP120 serves a similar purpose as existing technology, its process has been optimized by providing a safer, more accurate, and instantaneous method of assessment. While this may be relevant to the assessment of substantial clinical improvement, we stated it did not appear to be related to newness, and we remained unclear about how the patient population for which FloPatch FP120 is used differs from other patients for which existing non-invasive (for example, Doppler ultrasound devices) and invasive technologies are used for hemodynamic monitoring in a same or similar type of disease (such as septicemia or severe sepsis).
Accordingly, we stated that as it appears that the May 3, 2023, FDA 510(k) clearance and prior FDA 510(k) clearances for FloPatch FP120 may use the same or similar mechanism of action to achieve a therapeutic outcome, would be assigned to the same MS-DRG, and treat the same or similar patient population and disease, we believed that these technologies may be substantially similar to each other. We noted that if FloPatch FP120 as described in its 2023 FDA 510(k) clearance is substantially similar to prior versions as described in the 2022 and 2020 FDA 510(k) clearances, we believed the newness period for this technology would begin on March 24, 2020, with the earliest FDA 510(k) clearance date for FloPatch FP120 (K200337) and therefore, because the 3-year anniversary date of the technology's entry onto the U.S. market (March 24, 2023) occurred in FY 2023, the technology would no longer be considered new and would not be eligible for new technology add-on payments for FY 2025.
We invited public comments on whether FloPatch FP120 is substantially similar to existing technologies and whether FloPatch FP120 meets the newness criterion.
Comment: A commenter stated that CMS should deny the new technology add-on payment application because the many previous FDA clearances place the technology outside the FY 2025 eligibility period. The commenter stated that the technology is not new because it is the same technology as previously cleared products and noted the previous versions of FloPatch's 510(k) clearances in 2020 and 2022. The commenter also stated that, as CMS noted, Doppler ultrasound technology is the primary technology that FloPatch FP120 uses, and that many existing devices use the same or similar technology. In addition, the commenter stated that the applicant assertion that existing technologies do not provide clinicians with the information they need and that the current standard for assessing a patient's fluid responsiveness involves invasive cardiac output monitoring or clinical judgment without real-time objective data is not true because there are several existing products—such as (among others) point-of-care ultrasound, the Edwards's Hemosphere monitor, the Deltex TrueVue/ODM+, and the Caretaker Medical monitor—that use the same or similar technology to assess a patient's fluid responsiveness. The commenter also agreed with CMS that FloPatch FP120 would be assigned to the same MS-DRGs as those involving existing technologies used for invasive and non-invasive measurements of blood flow, such as for patients with septicemia or severe sepsis, and stated that many other technologies also do not require invasive procedures. The commenter further stated that it believed that FloPatch FP120 fails to meet the newness criterion because it uses the same technology (Doppler ultrasound) to perform the same task (monitor changes in blood flow) to assess the same reaction (response to fluid administration) to inform the same activity (fluid management) for patients with the same disease or condition (sepsis or septicemia) compared to those devices that are already available to Medicare beneficiaries.
Response: We thank the commenter for its input. Based on the information submitted, we believe that the May 2, 2023, FDA-cleared FloPatch FP120 technology uses a similar or same mechanism of action as existing technologies, including the previously FDA-cleared FloPatch FP120 products and Doppler ultrasound. We agree with the commenter that cases involving the use of FloPatch FP120 are assigned to the same MS-DRGs as those involving the use of the previously FDA-cleared ( print page 69158) FloPatch products and other existing technologies for invasive and non-invasive measurement of blood flow for patients with septicemia or severe sepsis. We also agree with the commenter that FloPatch FP120 informs the same clinical activity (fluid management) for patients with the same disease or condition (sepsis and septicemia) as the previously FDA-cleared FloPatch products and other existing technologies.
Because FloPatch FP120 meets all three of the substantial similarity criteria, we believe FloPatch FP120 is substantially similar to the version of FloPatch FP120 that was FDA-cleared on March 24, 2020, an existing noninvasive technology that assesses blood flow in the carotid artery. Therefore, we consider the newness period for FloPatch FP120 to begin on the date it first received FDA 510(k) clearance for the noninvasive assessment of blood flow in the carotid artery. Since FloPatch FP 120 has been on the U.S. market since 2020, the 3-year anniversary date of its entry onto the market occurred prior to FY 2025. Therefore, FloPatch FP120 does not meet the newness criterion, and is not eligible for new technology add-on payments for FY 2025. We note that we received public comments with regard to the cost and substantial clinical improvement criteria for this technology, but because we have determined that the technology does not meet the newness criterion and therefore is not eligible for approval for new technology add-on payments for FY 2025, we are not summarizing comments received or making a determination on those criteria in this final rule.
Delcath System submitted an application for new technology add-on payments for HEPZATO TM KIT for FY 2025. According to the applicant, HEPZATO TM KIT is a drug/device combination product consisting of melphalan and the Hepatic Delivery System (HDS), indicated as a liver-directed treatment for adult patients with uveal melanoma with unresectable hepatic metastases. Per the applicant, the HDS is used to perform percutaneous hepatic perfusion (PHP), an intensive local hepatic chemotherapy procedure, in which the alkylating agent melphalan hydrochloride is delivered intra-arterially to the liver with simultaneous extracorporeal filtration of hepatic venous blood return (hemofiltration).
Please refer to the online application posting for HEPZATO TM KIT, available at https://mearis.cms.gov/public/publications/ntap/NTP2310160RLLX , for additional detail describing the technology and the disease treated by the technology.
With respect to the newness criterion, according to the applicant, HEPZATO TM KIT was granted approval as a New Drug Application (NDA) from FDA on August 14, 2023, for use as a liver-directed treatment for adult patients with uveal melanoma with unresectable hepatic metastases affecting less than 50 percent of the liver and no extrahepatic disease or extrahepatic disease limited to the bone, lymph nodes, subcutaneous tissues, or lung that is amenable to resection or radiation. According to the applicant, the technology became available for sale on January 8, 2024, because manufacturing did not commence until after FDA approval was granted. Melphalan hydrochloride, a component of HEPZATO; TM KIT, is administered by intra-arterial infusion into the hepatic artery at a dose of 3 mg/kg of body weight with a maximum dose of 220 mg during a single treatment. The drug is infused over 30 minutes, followed by a 30-minute washout period. According to the applicant, treatments should be administered every 6 to 8 weeks, but can be delayed until recovery from toxicities, and as per clinical judgement.
The applicant stated that, effective October 1, 2023, the following ICD-10-PCS code may be used to uniquely describe procedures involving the use of HEPZATO TM KIT: XW053T9 (Introduction of melphalan hydrochloride antineoplastic into peripheral artery, percutaneous approach, new technology group 9). We note that we have also identified ICD-10-PCS code 5A1C00Z (Performance of biliary filtration, single), as an additional, relevant code, which may be used in combination with XW053T9 to more uniquely identify procedures involving the use of HEPZATO TM KIT. The applicant provided a list of diagnosis codes that may be used to currently identify the indication for HEPZATO TM KIT under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM and ICD-10-PCS codes provided by the applicant.
With respect to the substantial similarity criteria, the applicant asserted that HEPZATO TM KIT is not substantially similar to other currently available technologies because it offers the first liver-directed treatment option to patients with liver-dominant metastatic ocular melanoma (mOM) who may be poor candidates for liver resection and/or who may have difficulty tolerating systemic chemotherapy. According to the applicant, HEPZATO TM KIT uses a unique PHP procedure to isolate liver circulation and deliver a high concentration of melphalan to liver tumors via infusion followed by filtration of the hepatic venous flow to remove melphalan out of the blood with extracorporeal filters, and that therefore, the technology meets the newness criterion. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for HEPZATO TM KIT for the applicant's complete statements in support of its assertion that HEPZATO TM KIT is not substantially similar to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36060 ), we invited public comments on whether HEPZATO TM KIT is substantially ( print page 69160) similar to existing technologies and whether HEPZATO TM KIT meets the newness criterion. We also invited public comments on drug-device combination technology considerations for new technology add-on payments. Specifically, we stated that we were seeking comment on whether reformatting the delivery mechanism for a drug would represent a new mechanism of action for drug-device combination technologies, and on factors that should be considered when considering new technology add-on payments for technologies that may use a drug or device component that is no longer new in combination with a new drug or device component.
Comment: We received several comments regarding the newness criterion stating general support for HEPZATO TM KIT as a new treatment for uveal melanoma patients.
Response: We thank commenters for their input.
Comment: The applicant submitted a public comment reiterating that HEPZATO TM KIT does not use the same or a similar mechanism of action when compared to existing technologies to treat mOM and uses a different and novel technology as compared to existing technologies. The applicant stated that HEPZATO TM KIT uses a liver-directed PHP procedure to isolate liver circulation and deliver a high concentration of the chemotherapeutic drug melphalan to liver tumors via infusion, followed by filtration of the hepatic venous flow to remove melphalan from the blood with extracorporeal filters before returning the blood to the patient's systemic circulation. The applicant stated that HEPZATO TM KIT is the only FDA-approved product that saturates the entire liver with high-dose chemotherapy, thus allowing complete treatment of the liver metastases that are often the life-limiting component for mOM patients. The applicant further explained that melphalan was not approved by FDA to treat liver metastases from uveal melanoma until the approval of HEPZATO TM KIT. [ 65 ] The applicant further asserted that other liver-directed treatments only treat specific liver tumors and do not treat smaller tumors or micro-metastases, which limits the efficacy and beneficial clinical outcomes of other treatments in many mOM patients. Therefore, by uniquely saturating the whole liver with a high dose of a proven chemotherapy agent, the applicant asserted that HEPZATO TM KIT treats not only specific liver tumors, but also the multiple small tumors and micro-metastases in mOM. The applicant also stated that another liver-directed therapy, transarterial chemoembolization (TACE), does not treat the whole liver, leaving the patient prone to disease progression due to the growth of the untreated, smaller lesions. The applicant further explained that, in contrast, HEPZATO TM KIT's extracorporeal hemofiltration allows for administration of up to 12 times the conventional intravenous melphalan dose, while limiting systemic toxicities to manageable levels. [ 66 ]
The applicant also stated that by isolating the liver throughout the procedure and actively filtering out melphalan during the 30-minute infusion and subsequent 30-minute washout period, HEPZATO TM KIT creates an improved tumor response of 36.3 percent as compared to 12.5 percent of best alternative care (BAC) patients (p=0.013), and 7.7 percent of patients achieved a complete response in this difficult-to-treat disease. [ 67 ] Finally, the applicant stated that because treatment with HEPZATO TM KIT is minimally invasive, patients can receive multiple treatments extending the duration of response (DOR), resulting in patients with an objective response to treatment with HEPZATO TM KIT achieving responses that lasted for a median of 14 months compared to a reported median DOR of 11.1 months for patients treated with KIMMTRAK®.
Response: We thank the applicant for its comments. Based on our review of comments received and information submitted by the applicant as part of its FY 2025 new technology add-on payment application for HEPZATO TM KIT, we agree with the applicant that HEPZATO TM KIT uses a unique mechanism of action because it is the only FDA-approved product that isolates the liver circulation and allows for the delivery of a high concentration of a chemotherapeutic agent to liver tumors while limiting systemic exposure. Therefore, we agree with the applicant that HEPZATO TM KIT is not substantially similar to existing treatment options and meets the newness criterion. We consider the beginning of the newness period to commence on January 8, 2024, when HEPZATO TM KIT became available for sale on the market.
Comment: In response to the request for comments on drug-device combination technology considerations for new technology add-on payments, the applicant stated that CMS should consider whether the combination either offers a treatment option for a patient population unresponsive to, or ineligible for, currently available treatments or significantly improves clinical outcomes relative to technologies previously available. The applicant asserted that approving technologies like HEPZATO TM KIT signals CMS's support of finding new methodologies to repurpose older drugs that, under an existing technology, are not effective in treating specific cohorts of the targeted disease population. Another commenter expressed concern that there could be a scenario where a new drug-device combination product, with the potential to provide meaningful improvements to a specific patient population, is not approved for new technology add-on payment, limiting patient access to the treatment, despite meeting the substantial clinical improvement criterion because the newness criterion was not set up to evaluate the technology appropriately. The commenter further stated that if a substantial clinical improvement is not evaluated if the newness criterion is not satisfied, then there is a risk that treatments providing meaningful improvements could be overlooked. The commenter stated that the mechanism of action should be evaluated for the treatment provided by the drug-device combination, as a whole. Specifically, the commenter stated that if the mechanism of action for treatment provided with the drug-device combination is different from that of treatment with just the drug component that is no longer new, or that of treatment with the device component that is no longer new with a different drug, then it should not be considered substantially similar. The commenter also stated that the specific patient population that benefits from the drug-device combination should also be considered carefully when making a determination of substantial similarity, and that if the drug device combination provides treatment for the same type of disease as a drug or device component that is not considered new, the specific populations for both need to be compared. The commenter stated that if the drug-device combination provides ( print page 69161) meaningful treatment to a broader or more specific patient population, it should not be considered substantially similar. Additionally, the commenter stated that if the drug device combination provides meaningful treatment to the same patients that are treated with the existing drug or device component that is not new, but the drug-device combination provides a new treatment option for patients that do not respond well to the existing treatment options, the drug-device combination should not be considered substantially similar. The commenter stated that these considerations align with the criteria currently used to evaluate if technologies applying for new technology add-on payment would be considered substantially similar to existing treatment options, but that the current language for the third criterion for substantial similarity (that is, the new use of the technology involves the treatment of the same or similar type of disease and patient population when compared to an existing technology) is broad enough to cause drug-device combinations to be overlooked without evaluating the clinical improvement they may provide for specific patients. The commenter stated that if a drug-device combination provides treatment for patients that do not respond well to existing treatment options, then the patient populations could be considered “similar”, however, it is important to evaluate the clinical improvement of the drug-device combination to identify if it would serve as a potential new treatment for patients running out of treatment options with the long-term goal of reducing the number of treatments these patients need to try before finding an effective solution.
Response: We thank the commenters for their feedback. We will continue to consider these issues relating to the assessment of the mechanism of action for a technology involving a drug-device combination. We agree that the specific patient population that benefits from the drug-device combination should be considered carefully, and that if the drug device combination provides treatment for the same type of disease as a drug or device component that is not considered new, the patient population being treated should be assessed. We note that the third criterion for substantial similarity (that is, the new use of the technology involves the treatment of the same or similar type of disease and patient population when compared to an existing technology) involves these considerations. However, where the commenter stated that if a new technology add-on payment applicant's technology is being assessed for substantial similarity with another technology, and the applicant technology treats a narrower population (and the technologies are otherwise the same or substantially similar under our criteria), we disagree that the applicant technology should be determined to not be substantially similar.
Regarding the concern that the third criterion for substantial similarity is broad enough to cause drug-device combinations to be overlooked without evaluating the clinical improvement they may provide for specific patients, and also the applicant's comment that CMS should consider whether the combination either offers a treatment option for a patient population unresponsive to, or ineligible for, currently available treatments or significantly improves clinical outcomes relative to technologies previously available, we do not agree that we should evaluate clinical improvement while assessing the newness criterion. Rather, we follow a logical sequence of determinations, moving from the newness criterion to the cost criterion and finally to the substantial clinical improvement criterion, as discussed in the FY 2006 IPPS final rule. Therefore, we are reluctant to import substantial clinical improvement considerations into the decision about whether technologies are new ( 70 FR 47348 ). Regarding patient populations unresponsive to, or ineligible for, currently available treatments, as noted, the third criterion for substantial similarity considers whether the new use of the technology involves the treatment of the same or similar type of disease and patient population when compared to an existing technology.
With respect to the cost criterion, the applicant provided multiple analyses to demonstrate that it meets the cost criterion. For each analysis, the applicant searched the FY 2022 MedPAR file using a combination of ICD-10-CM and/or PCS codes to identify potential cases representing patients who may be eligible for HEPZATO TM KIT. The applicant explained that it used different codes to demonstrate different cohorts that may be eligible for HEPZATO TM KIT because it is indicated for a rare condition, hepatic-dominant mOM, which does not have a unique ICD-10-CM diagnosis code to identify potential cases with the specific diagnosis of interest, nor a unique ICD-10-PCS procedure code that would identify patients receiving this specific procedure. The applicant believed the cases identified in the analysis are the closest proxies to the cases potentially eligible for the use of HEPZATO TM KIT. Each analysis followed the order of operations described in the table later in this section.
For the first analysis, the applicant searched for cases with ICD-10-PCS code 3E05305 (Introduction of other antineoplastic into peripheral artery, percutaneous approach) for the PHP procedure, and ICD-10-CM code Z51.11 (Encounter for antineoplastic chemotherapy) as the primary diagnosis for the administration of chemotherapy during an inpatient stay. In addition, the applicant narrowed the analysis to cases with liver-dominant mOM using at least one secondary liver metastases diagnosis plus at least one ocular melanoma diagnosis. Please see the online posting for HEPZATO TM KIT for the complete list of codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 11 claims mapping to one MS-DRG: 829 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Other Procedures with CC/MCC). The applicant calculated a final inflated average case-weighted standardized charge per case of $1,068,530, which exceeded the average case-weighted threshold amount of $104,848.
For the second analysis, the applicant searched for the following combination of ICD-10-CM diagnosis codes: Z51.11 (Encounter for antineoplastic chemotherapy) as the primary diagnosis code, in combination with at least one of the following secondary liver metastases codes: C78.7 (Secondary malignant neoplasm of liver and intrahepatic bile duct), or C22.9 (Malignant neoplasm of liver, not specified as primary or secondary). The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 1,134 claims mapping to nine MS-DRGs, with 94 percent of identified cases mapping to three MS-DRGs: 829 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Other Procedures with CC/MCC), as well as 846 and 847 (Chemotherapy without Acute Leukemia as Secondary Diagnosis with MCC, and with CC, respectively). The applicant calculated a final inflated average case-weighted standardized charge per case of $1,066,207, which exceeded the average case-weighted threshold amount of $81,652.
For the third analysis, the applicant searched for cases where the ICD-10-CM code Z51.11 (Encounter for antineoplastic chemotherapy) is the primary diagnosis or the ICD-10 PCS code 3E05305 (Introduction of other ( print page 69162) antineoplastic into peripheral artery, percutaneous approach) is reported. In addition, the case also needed to include at least one of the following secondary liver metastases codes: C78.7 (Secondary malignant neoplasm of liver and intrahepatic bile duct) or C22.9 (Malignant neoplasm of liver, not specified as primary or secondary). The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 1,277 claims mapping to 12 MS-DRGs with 92 percent of identified cases mapping to three MS-DRGs: 829 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Other Procedures with CC/MCC); as well as 846 and 847 (Chemotherapy without Acute Leukemia as Secondary Diagnosis with MCC, and with CC, respectively). The applicant calculated a final inflated average case-weighted standardized charge per case of $1,067,772, which exceeded the average case-weighted threshold amount of $80,245.
For the fourth analysis, the applicant searched for cases reporting the following combination of ICD-10-CM diagnosis codes: C78.7 (Secondary malignant neoplasm of liver and intrahepatic bile duct) or C22.9 (Malignant neoplasm of liver), in combination with at least one ocular melanoma ICD-10-CM code. Please see the online posting for HEPZATO TM KIT for the complete list of codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 1,059 claims mapping to 91 MS-DRGs with none exceeding 4.91 percent. The applicant calculated a final inflated average case-weighted standardized charge per case of $1,062,553, which exceeded the average case-weighted threshold amount of $66,104.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant asserted that HEPZATO TM KIT meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36062 ), we invited public comments on whether HEPZATO TM KIT meets the cost criterion.
Comment: Multiple commenters stated that DRG payment to the hospitals will not be sufficient to account for the cost of the HEPZATO TM KIT treatment. The applicant commented that the MS-DRG rate otherwise applicable to the HEPZATO TM KIT is inadequate, and stated that use of the HEPZATO TM KIT will likely fall within one of the following MS-DRGs where other chemotherapies administered during inpatient stays would also be assigned: 826 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Major O.R. Procedures with MCC); 827 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Major O.R. Procedures with CC); 828 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with Major O.R. Procedures without CC/MCC); 829 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with other Procedures with CC/MCC); 830 (Myeloproliferative Disorders or Poorly Differentiated Neoplasms with other Procedures without CC/MCC); 846 (Chemotherapy without Acute Leukemia as Secondary Diagnosis with MCC); 847 (Chemotherapy without Acute Leukemia as Secondary Diagnosis with CC); or 848 (Chemotherapy without Acute Leukemia as Secondary Diagnosis without CC/MCC). The applicant stated that the payment rate for each of these MS-DRGs is far below the cost of HEPZATO TM KIT, which is reported in REDBOOK as having a wholesale acquisition cost of $182,500 per 250 mg. The applicant stated that, as such, HEPZATO TM KIT clearly qualifies under the cost test for new technology add-on payment designation.
Response: We thank commenters for their comments.
We agree that the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all ( print page 69165) the scenarios. Therefore, HEPZATO TM KIT meets the cost criterion.
With regard to the substantial clinical improvement criterion, the applicant asserted that HEPZATO TM KIT represents a substantial clinical improvement over existing technologies because it offers a minimally invasive, targeted, effective, and safe treatment option to patients with liver-dominant mOM who may be poor candidates for liver resection or who may have difficulty tolerating systemic chemotherapy which results in a substantial clinical improvement in response and survival rates over best available care (BAC) and quality of life compared to pre-treatment. The applicant provided 11 studies to support these claims, as well as one background article about use of chemosaturation with PHP (CS-PHP) as a palliative treatment option for patients with unresectable cholangiocarcinoma. [ 69 ] The following table summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for HEPZATO TM KIT for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36064 through 36066 ), after review of the information provided by the applicant, we stated we had the following concerns regarding whether HEPZATO TM KIT meets the substantial clinical improvement criterion. With respect to the applicant's assertion that HEPZATO TM KIT offers a treatment option for a patient population unresponsive or ineligible for currently available treatments, while the applicant stated that HEPZATO TM KIT offers an additional treatment option to patients with liver-dominant mOM who may be poor candidates for liver resection or who may have difficulty tolerating systemic chemotherapy, we stated the applicant did not provide evidence in support of this assertion. We noted that we would be interested in information regarding whether there are potential Medicare patient populations that may have difficulty tolerating (or be unresponsive to) KIMMTRAK® or other currently available treatments but would be a good candidate for HEPZATO TM KIT.
Regarding the claim that HEPZATO TM KIT improves survival over other treatment options, we stated that the applicant provided seven peer-reviewed cohort studies, summary material from an unpublished study, and one randomized controlled clinical study to support the claim. We noted that the seven peer reviewed cohort studies [ 70 71 72 73 74 75 76 ] provided a range of results of overall survival as reported for patients treated with the HEPZATO TM KIT (median overall survival after first CS-PHP ranged from 9.6 months to 27.4 months depending on the study, and median 1-year overall survival rate raged from 44 percent to 77 percent depending on study). A few of the seven peer-reviewed cohort studies (Karydis et al. (2018); Tong et al. (2022); Meier et al. (2021)) reported statistically significant improvement in overall survival (OS) when compared to non-responders or stable disease groups. Only one of the seven studies, Dewald et al. (2021), compared results to alternative treatments, but statistical significance was not achieved (P = 0.97) with CS-PHP resulting in a median OS of 24.1 months compared with 23.6 months for patients receiving other therapies. We noted that we believed additional evidence supporting that HEPZATO TM KIT offers a significant difference in OS rates compared to currently available treatments would be helpful in our evaluation of the applicant's assertion. We also stated that several of the studies provided as evidence include small, non-randomized studies without the use of comparators or controls, which may ( print page 69168) affect the ability to draw meaningful conclusions about treatment outcomes from the results of the studies. We also noted that a majority of the studies provided (Bruning et al. (2020); Vogl et al. (2017); Dewald et al. (2021); Meijer et al. (2021); and Artzner et al. (2019)) were conducted outside the U.S. We questioned if there may be differences in treatment guidelines between these countries that may have affected clinical outcomes.
We stated that the applicant also submitted summary presentation material evidence to support this claim in the form of a poster and slides for the FOCUS study, [ 77 ] in which 144 patients were enrolled, with 91 patients receiving PHP treatment and 32 patients receiving BAC. According to the applicant, preliminary results from the phase III FOCUS Trial show that progression free survival (PFS) was 9.03 months among PHP patients and just over 3 months among BAC patients. OS among treated PHP patients was 19.25 months and among treated BAC patients was 14.49 months. However, this study had yet to be published and was not yet available for analysis and peer review. We stated that as of the time of the proposed rule, we were unable to verify the methods, results, and conclusions of this study as the applicant only provided evidence in the form of a poster and presentation. For example, one citation provided by the applicant in the form of a non-peer-reviewed conference presentation details preliminary results from the FOCUS Phase III Trial. We noted we would be interested in the statistical analysis (including p value and CI data) surrounding the OS rates. In addition, we stated that the poster notes that due to slow enrollment and patient reluctance to receive BAC treatment, the trial design was amended to a single arm design with all eligible patients receiving PHP after discussion with FDA. We noted we would be interested in detail about these specific eligibility requirements, as well as how the potential for confounding variables resulting from any differences in the resulting populations were identified and mitigated.
We further noted that in the published randomized clinical trial [ 78 ] (RCT) provided by the applicant, the median hepatic progression free survival (hPFS), the primary endpoint of the trial, was 7.0 months for patients using HEPZATO TM KIT compared to 1.6 months for patients receiving BAC. However, the median overall survival (OS) with the treatment of HEPZATO TM KIT was 10.6 months (95 percent CI 6.9-13.6 months) compared to 10.0 months (95 percent CI 6.0-13.1 months) for the group of patients who received BAC. We stated that the study notes that median OS was not significantly different (PHP-Mel 10.6 months vs. BAC 10.0 months), but OS was 13.1 months (95 percent CI 10.0-20.3 months) in BAC patients who crossed over and received treatment with PHP-Mel (n = 28, 57.1 percent). In the study discussion of OS, Hughes et al. concluded that the 57 percent of patients who were allowed to crossover confounded the ability to analyze any survival advantage associated with PHP Mel. We noted we would be interested in additional evidence in our evaluation of the applicant's assertion that HEPZATO TM KIT substantially improves survival over other treatment options.
Regarding the claim that HEPZATO TM KIT increases response rate over BAC, we noted that across the retrospective studies, response rates ranged from an overall response rate of 42.3 percent [Dewald et al (2021)] to a partial response of 89 percent [Vogl et al. (2017)] depending on the study. However, as the applicant cited many of the same retroactive studies that it referenced in support of the claim of improved survival [Bruning et al. (2020); Vogl et al. (2017); Dewald et al. (2021); Meijer et al. (2021); Artzner et al. (2019); Tong et al. (2022); Karydis et al. (2018)], we noted we had the same questions as discussed previously regarding the ability to draw meaningful conclusions from the results of these studies in evaluation of this claim.
Regarding the unpublished FOCUS study (Delcath ASCO 2022 FOCUS Trial Poster), [ 79 ] previously described, the applicant stated that in the preliminary results from the FOCUS Trial, the overall response rate (ORR) among PHP patients was 36.3 percent, nearly three times better than that of the 12.5 percent ORR among BAC patients. However, as previously noted, we stated we would be interested in details about the eligibility requirements, and how the potential for confounding variables resulting from any differences in the resulting populations were identified and mitigated.
Lastly, we stated that with regard to the assertion that HEPZATO TM KIT improves quality of life over pre-treatment, the applicant submitted the Vogl et al. (2017) study as evidentiary support. The study was a retrospective, multi-center study reporting outcome and safety after percutaneous isolated hepatic perfusion (PIHP) with Melphalan for patients with uveal melanoma and metastatic disease limited to the liver. Thirty-five PIHP treatments were performed in 18 patients (8 male, 10 female) at seven hospitals across the U.S and Germany between January 2012 and December 2016. Patients' life quality was assessed using four-point scale questionnaires to rate overall health and life quality after therapy, how much their health and quality of life had changed after therapy, and how pleased they were with PIHP. We noted that the study used a subjective four-point measurement scale to determine quality-of-life used in the study. We stated that we questioned if a more objective assessment tool would be more helpful in evaluating a patient's quality of life. We noted it was unclear if the survey questions were asked verbally, and by whom, or if the survey was answered in writing by the patient alone. As the study was not randomized and the patients' responses were not anonymous, we noted that we questioned if there may have been resulting response bias, or interviewer bias that would impact our ability to draw meaningful conclusions about a subjective measurement of improved quality of life. In addition, we noted that the study utilized the Delcath Hepatic CHEMOSAT® Delivery System for Melphalan components as part of the treatment, and it was unclear if the technologies used in the study were the same as HEPZATO TM KIT, or what differences may exist between the technologies. We noted we would be interested in information about any differences between Delcath's HEPZATO TM KIT and the technologies used in this study for PIHP with Melphalan.
We invited public comments on whether HEPZATO TM KIT meets the substantial clinical improvement criterion.
Comment: The applicant submitted a public comment regarding the substantial clinical improvement criterion and provided responses to CMS's concerns from the proposed rule. The applicant asserted that HEPZATO TM KIT is the only FDA-approved therapy for the approximately 55 percent of patients with mOM who ( print page 69169) are not eligible for KIMMTRAK® or whose disease has progressed despite using other therapies. The applicant further asserted that the mechanism of action in KIMMTRAK® depends on the presence of the HLA-A*02:01 allele and only approximately 45 percent of people in the U.S. are HLA-A*02:01-positive. Therefore, the applicant concluded that more than half of patients with mOM are ineligible for KIMMTRAK® treatment. The applicant also stated that all patients in the FOCUS study had unresectable liver metastases, and that accordingly, the FOCUS study results demonstrate efficacy for this patient population. The applicant stated that at the doses that patients can tolerate, systemic chemotherapy has been found to have low efficacy. The applicant also cited study results asserting that single-agent and combination chemotherapies have mostly been ineffective in patients with metastatic uveal melanoma, with ORRs ranging from zero to eight percent with the alkylating agents dacarbazine or temozolomide and up to 10 percent with fotemustine, and that most of these clinical studies have been nonrandomized single-arm trials, with only eight randomized trials evaluating systemic therapy alone completed since 2000. [ 80 ] Lastly, the applicant asserted the evidence demonstrates that HEPZATO TM KIT is effective in Medicare beneficiaries who are unresponsive to, or have difficulty tolerating, other treatments. The applicant stated that a subgroup analysis of the FOCUS study demonstrates that HEPZATO TM KIT is a treatment option for Medicare-age patients, as there were similar ORR, OS, and PFS results in patients ≥65 years old and <65 years old, and differences were not statistically significant. The applicant stated that a large proportion of Medicare-aged mOM patients first treated with KIMMTRAK® are likely to experience disease progression and leading immunotherapies have relatively short PFS in mOM patients; therefore, both categories of patients would be good candidates for treatment with HEPZATO TM KIT.
The applicant noted that the previously unpublished FOCUS study (Delcath ASCO 2022 FOCUS Trial Poster) has since been published, but did not make the study available for review. [ 81 ] In response to our interest in receiving additional detail about specific eligibility requirements and how potential for confounding variables resulting from any differences in the resulting populations were identified and mitigated, the applicant stated that the study began as a randomized trial with two treatment arms (HEPZATO TM KIT and BAC), but at the midpoint of the trial, many patients randomized to the BAC arm withdrew consent prior to treatment. Per the applicant, subsequent to discussion with FDA and investigators, all parties supported amending the trial to a single-arm design, in which all enrolled patients would receive melphalan/HDS treatment. The applicant stated that potential differences in demographic and baseline characteristics between patients in the two portions of the study were examined and no significant differences were found among potential confounding variables. Per the applicant, patients in the FOCUS study who had received prior therapy and patients who had not been treated for mOM experienced similar ORR (37.5 percent and 35.3 percent; p = 0.83), OS (20.83 months and 20.53 months; p = 0.499), and PFS (9.18 months and 9.00 months; p = 0.86). The applicant also provided a subgroup analysis of patients in the FOCUS study ≥65 years old and <65 years old and stated that there were statistically insignificant differences in ORR (30.0 percent and 39.3 percent; p = 0.488), OS (20.53 months and 20.83 months; p = 0.574), and PFS (9.00 months and 9.07 months; p = 0.494). The applicant provided additional FOCUS study results, noting patients treated with HEPZATO TM KIT, who were treatment-naive (56.0 percent) or previously treated (44.0 percent) and could have limited extrahepatic disease (29.7 percent), had an ORR of 36.3 percent (95 percent CI: 26.44, 47.01) with 7.7 percent complete response and 28.6 percent partial response; tumor responses were durable, with a median DOR of 14.0 months (95 percent CI: 8.31, 17.74); disease control rate was 73.6 percent (95 percent CI: 63.35, 82.31); median OS was 20.5 months (95 percent CI: 16.79, 25.26); and median PFS was 9.0 months (95 percent CI: 6.34, 11.56). Lastly, the applicant expressed its opinion that the retrospective single-center and multicenter investigations of mOM patients in Europe reinforce the conclusion of HEPZATO TM KIT's efficacy noting that in general, these studies have found ORR and PFS results that are similar to or exceed those reported in the FOCUS study and OS results similar to or exceeding the typical OS of mOM patients (approximately 12 months), with five of the seven studies reporting a median OS between 14.9 and 27.4 months.
In response to CMS's request for additional evidence that HEPZATO TM KIT substantially improves survival over other treatment options, the applicant stated that the Dewald et al. (2021) study [ 82 ] supports this claim and reported an ORR of 42.3 percent and disease control rate (DCR) of 80.8 percent, exceeding those reported in the literature for alternative treatments for mOM (DCR range of 64.7 percent to 80.2 percent). The applicant cited another study, Dewald et al. (2022), which reports on a larger group of patients and treatments (66 patients, 145 treatments) that reported an ORR 59 percent, a DCR of 93.4 percent, and a median OS of 18.4 months. [ 83 ] The applicant also provided details on a post-hoc analysis of FOCUS study patients treated with HEPZATO TM KIT, in which Zager and colleagues (2024) reported that patients treated with HEPZATO TM KIT were found to have a statistically significant relationship between OS and best overall response. According to the applicant, the results from a post hoc analysis of the relationship between tumor response and survival demonstrated a statistically significant difference (p < 0.0001) between the patients who had a best overall response of partial response [median OS of 28.2 months (95 percent CI, 23.46-34.46 months)], stable disease [median OS of 19.3 months (95 percent CI, 15.90-23.00 months)], and progressive disease [median OS of 12.0 months (95 percent CI, 8.18-14.03 months)]. [ 84 ] According to the applicant, this correlation between response and survival suggests that the favorable response rate seen with HEPZATO TM KIT leads to beneficial survival outcomes relative to other treatments.
Regarding the claim that HEPZATO TM KIT improves quality of life over pre-treatment, the applicant stated that CMS questioned whether the Vogel study was ( print page 69170) reliable, given that it was unclear whether the study survey questions were asked verbally, and by whom, or if the survey was answered in writing by the patient alone. The applicant acknowledged that the details related to the conduct of the survey were not reported in the 2017 article by Vogel and colleagues, so response bias and interviewer bias cannot be ruled out. The applicant further stated that there is no reason to believe the study authors did not control for bias, and that the Vogel study is only one of many studies that demonstrated efficacy. The applicant cited a single-center, prospective cohort study of patient-reported quality of life assessments collected before and after treatment with CHEMOSAT® in which patients completed the European Organization for Research and Treatment of Cancer (EORTC) QLQ-C30 version 3, a validated self-report questionnaire that assesses quality of life (QoL) in cancer patients and consists of a global health status scale, functional scales, and symptom scales. [ 85 ] The study authors found that global health status 21 days after treatment with CHEMOSAT® was consistent with global health status pre-treatment, despite temporary decreases in global health status scores at day 2/3 and day 7 post-treatment. Specifically, the study authors concluded that CHEMOSAT® treatment has limited impact on QoL of patients with metastasized UM, and that the general GHS returns to baseline within 3 weeks despite moderate decline in fatigue and physical and role functioning scores. Regarding CMS's concern that it was unclear if the technologies used in the study are the same as HEPZATO TM KIT, or what differences may exist between the technologies ( 89 FR 36066 ), the applicant stated that in Europe the marketed product known as CHEMOSAT® uses the same procedural “Instructions for Use” as the HEPZATO TM KIT. The applicant further stated that the only difference between CHEMOSAT® used in treatments outside the US and HEPZATO TM KIT used in the FOCUS study is that CHEMOSAT® is used in conjunction with the hospital's melphalan supply, whereas HEPZATO TM KIT is prepackaged with melphalan.
We received several additional comments in support of the application for HEPZATO TM KIT. Generally, these additional commenters stated that HEPZATO TM KIT provides a clinically meaningful pathway for mOM patients who previously did not have an approved treatment option and asserted that such treatments will continue to improve outcomes and ultimately make mOM a survivable disease. The commenters asserted that approval would increase access to treatments for a patient population with very limited treatment options, and some of these commenters cited aforementioned study results, inclusive of the FOCUS trial response rates and noting a statistically significant relationship between overall survival and best overall response in patients treated with HEPZATO TM KIT.
We also received one comment stating that it did not support approval of HEPZATO TM KIT. The commenter stated its belief that HEPZATO TM KIT is not necessarily safer or easier to tolerate because patients receiving treatment with HEPZATO TM KIT in the Zager et al. (2024) [ 86 ] study experienced a higher rate of treatment discontinuation due to adverse events (17.9 percent) compared to patients in another study who received treatment with KIMMTRAK® (2 percent). [ 87 ] The commenter also asserted that between these two studies, there was a 73 percent 1-year survival rate for KIMMTRAK® and 80 percent 1-year survival rate for HEPZATO TM KIT. However, the commenter stated that patients in the FOCUS trial had better ECOG performance statuses and more stringent exclusion criteria, including limitations on extrahepatic disease. Lastly, the commenter asserted that, other than HLA-A*02:01-negative patients, there does not appear to be a population of mOM patients that would qualify for HEPZATO TM KIT therapy and not qualify for treatment with KIMMTRAK®.
Response: We thank the applicant and other commenters for their comments regarding the substantial clinical improvement criterion. Based on review of the information submitted by the applicant and the additional information received, we agree with the applicant that HEPZATO TM KIT offers a treatment option for adult patients with uveal melanoma with unresectable hepatic metastases who are ineligible for existing therapies because they may be poor candidates for liver resection or who may have difficulty tolerating systemic chemotherapy and are HLA-A*02:01-negative and therefore ineligible for treatment with KIMMTRAK®. Therefore, we agree that HEPZATO TM KIT represents a substantial clinical improvement over existing technologies.
After consideration of the public comments we received and the information included in the applicant's new technology add-on payment application, we have determined that HEPZATO TM KIT meets the criteria for approval for new technology add-on payment. Therefore, we are approving new technology add-on payments for this technology for FY 2025. Cases involving the use of HEPZATO TM KIT that are eligible for new technology add-on payments will be identified by ICD-10-PCS codes: XW053T9 (Introduction of melphalan hydrochloride antineoplastic into peripheral artery, percutaneous approach), in combination with 5A1C00Z (Performance of biliary filtration, single).
In its application, the applicant stated that the cost of HEPZATO TM KIT is $182,500 per treatment and that patients will receive up to six treatments administered at a dose of 3 mg/kg of body weight, with a maximum dose of 220 mg in a single administration. The applicant anticipates an average of four treatments per patient (based on the median of four treatments per person in the FOCUS trial) but those treatments would be administered across four separate inpatient stays as treatment cycles must take place 6-8 weeks apart. The average cost will therefore be $182,500 per inpatient stay. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, the maximum new technology add-on payment for a case involving the use of HEPZATO TM KIT is $118,625 for FY 2025.
CellTrans Inc. submitted an application for new technology add-on payments for Lantidra TM for FY 2025. According to the applicant, Lantidra TM is an allogeneic pancreatic islet cellular therapy indicated for the treatment of adults with Type 1 diabetes (T1D) who are unable to approach target hemoglobin A1c (HbA1c) because of repeated episodes of severe hypoglycemia despite intensive diabetes management and education. Per the applicant, Lantidra TM is used in ( print page 69171) conjunction with concomitant immunosuppression. The applicant asserted that the route of administration for Lantidra TM is infusion into the hepatic portal vein only. The applicant noted that following transplant, the patient is monitored for graft function and safety issues, including potential adverse reactions due to immunosuppression. The applicant stated that the primary mechanism of action for Lantidra TM is the secretion of insulin by the beta cells within the infused allogeneic islet of Langerhans, which are responsible for regulating blood glucose levels in response to glucose stimulation.
Please refer to the online application posting for Lantidra TM , available at https://mearis.cms.gov/public/publications/ntap/NTP231017H5N2T , for additional detail describing the technology and the disease treated by the technology.
With respect to the newness criterion, according to the applicant, Lantidra TM was granted approval for a Biologics License Application (BLA) from FDA on June 28, 2023, for the treatment of adults with T1D who are unable to approach target HbA1c because of current repeated episodes of severe hypoglycemia despite intensive diabetes management and education. According to the applicant, the technology was commercially available on January 8, 2024. The applicant stated that the approved manufacturing site for Lantidra TM is at the University of Illinois (UI) Health, UI in Chicago and time was needed to transfer islet cell transplant clinical protocols to the UI Health transplant division.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36066 ), we noted that under national coverage determination (NCD) 260.3.1 Islet Cell Transplantation in the Context of a Clinical Trial, Medicare will pay for the routine costs, as well as transplantation and appropriate related items and services, for Medicare beneficiaries participating in a National Institutes of Health (NIH)-sponsored clinical trial(s). Specifically, Medicare will cover transplantation of pancreatic islet cells, the insulin producing cells of the pancreas. Coverage may include the costs of acquisition and delivery of the pancreatic islet cells, as well as clinically necessary inpatient and outpatient medical care and immunosuppressants. Because Lantidra TM may be covered by Medicare when it is used in the setting of a clinical trial, we stated we would evaluate whether Lantidra TM is eligible for new technology add-on payments for FY 2025. We noted that any payment made under the Medicare program for services provided to a beneficiary would be contingent on CMS's coverage of the item, and any restrictions on the coverage would apply.
The applicant stated that the recommended minimum dose is 5,000 equivalent islet number (EIN)/kg for the initial infusion, and 4,500 EIN/kg for subsequent infusion(s) in the same recipient. The maximum dose per infusion is dictated by the estimated tissue volume, which should not exceed 10 cc per infusion, and the total EIN present in the infusion bag (up to a maximum of 1 × 10 ‸ 6 EIN per bag). A second infusion may be performed if the patient does not achieve independence from exogenous insulin within 1-year post-infusion or within 1-year after losing independence from exogenous insulin after a previous infusion. A third infusion may be performed using the same criteria as for the second infusion.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for Lantidra TM and was granted approval to use the following procedure code effective October 1, 2024: XW033DA (Introduction of donislecel-jujn allogeneic pancreatic islet cellular suspension into peripheral vein, percutaneous approach, new technology group 10).
As previously discussed, if a technology meets all three of the substantial similarity criteria under the newness criterion, it would be considered substantially similar to an existing technology and would not be considered new for the purpose of new technology add-on payments.
With respect to the substantial similarity criteria, the applicant asserted that Lantidra TM has not been assigned to the same MS-DRG when compared to an existing technology to achieve a therapeutic outcome. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for Lantidra TM for the applicant's complete statements in support of its assertion that Lantidra TM is not substantially similar to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36067 ), we invited public comments on whether Lantidra TM is substantially similar to existing technologies and whether Lantidra TM meets the newness criterion.
Comment: The applicant submitted a public comment regarding the newness criterion. The applicant stated that in its application, it should have responded no to the question, “Does the use of the technology involve the treatment of the same/similar type of disease and the same/similar patient population when compared to an existing technology?” The applicant further clarified that it did state in its application that, due to its minimally invasive administration, Lantidra TM addresses an unmet need for a small distinct subset of patients with hard-to-control T1D complicated by severe hypoglycemia who cannot receive a whole pancreas transplant due to medical or surgical risk.
Response: We thank the applicant for its clarification. Although the applicant states in its public comment that Lantidra TM treats a new patient population because it addresses an unmet need for a small distinct subset of patients with hard-to-control T1D complicated by severe hypoglycemia who cannot receive a whole pancreas transplant due to medical or surgical risk, we note in addition to whole pancreas transplant, there are other existing technologies for managing glucose control and hypoglycemia. Technologies that are currently available for use in patients with hard-to-control T1D include continuous glucose monitors and automated insulin delivery systems, which can be utilized to manage or reduce severe hypoglycemic episodes, such as the Medtronic MiniMed TM 670G system, Medtronic MiniMed TM 780G, and Tandem Diabetes Care Control-IQ® Technology. We therefore question the applicant's assertion that Lantidra TM meets an unmet need for this specific patient population, given there are several other evidence-based treatment options available that may potentially manage the high glucose variability for this subset of patients.
We also agree with the applicant that the underlying mechanism of action of Lantidra TM is similar to that of whole pancreas transplant. However, we note that the mechanism of action for Lantidra TM is different than that of the previously mentioned technologies used for treatment in the subset of patients with hard-to-control T1D complicated by severe hypoglycemia who cannot receive a whole pancreas transplant due to medical or surgical risk, as identified by the applicant.
In addition, we note that although Lantidra TM would map to a different MS-DRG than whole (solid) pancreas transplant, we believe that Lantidra TM would map to the same MS-DRGs as existing insulin delivery therapies and technologies used to treat the subset of patients with hard-to-control T1D complicated by severe hypoglycemia who cannot receive a whole pancreas transplant due to medical or surgical risk.
Therefore, based on our review of the comments received and information submitted by the applicant as part of its FY 2025 new technology add-on payment application for Lantidra TM , we agree with the applicant that Lantidra TM is not substantially similar to existing treatment options because it has a unique mechanism of action compared to existing insulin delivery therapies and technologies when used to treat the subset of patients with hard-to-control T1D complicated by severe hypoglycemia who cannot receive a whole pancreas transplant due to medical or surgical risk. Therefore, Lantidra TM meets the newness criterion. We consider the beginning of the newness period to commence on January 8, 2024, when Lantidra TM became commercially available.
With respect to the cost criterion, the applicant included the two most recent patient cases with charges of Lantidra TM billed by a hospital that administered the technology, based on that hospital's billing data file on the undiscounted costs. The applicant stated that it attempted to identify potential cases representing patients who may be eligible for Lantidra TM by searching the FY 2022 MedPAR file and the 100 percent sample FY 2022 Standard Analytical Files (SAF) for cases reporting ICD-10-CM/PCS codes and MS-DRGs codes that were relevant to FDA-approved indication and administration of Lantidra TM , however, it could not confirm if cost data from the two most recent patient cases were included in the FY 2022 MedPAR file or SAF. As a result, the applicant provided the charges billed by the hospital for these two cases. The applicant stated that the MS-DRG coded for the two cases was MS-DRG 639 (Diabetes without CC/MCC). The applicant followed the order of operations described in the following table and calculated a final inflated average case-weighted standardized charge per case of $374,547, which exceeded the average case-weighted threshold amount of $32,311. Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount, the applicant asserted that Lantidra TM meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36067 through 36068 ), we noted the following concerns regarding the cost criterion. We noted that the applicant did not remove any charges or indirect charges related to prior technology without providing further details. We stated we were interested in additional information regarding whether Lantidra TM would replace any prior technology. We noted we were also interested in how the applicant estimated an inflation factor of 10 percent to apply to the standardized charges. With respect to the cases included in the cost analysis, we noted that the applicant limited the cost analysis to the two most recent patient cases with charges of Lantidra TM billed by the hospital, which the applicant asserted were the best available data for the FY 2022 cost analysis. We noted the MS-DRG coded for these two cases was MS-DRG 639 (Diabetes without CC/MCC). We stated we were interested in information as to whether cases in other MS-DRGs would be potentially eligible for Lantidra TM and if these cases should also be included in the cost analysis by using appropriate inclusion/exclusion criteria based on reporting of ICD-10-CM/PCS codes.
We invited public comments on whether Lantidra TM meets the cost criterion.
Comment: The applicant clarified that they used a close approximation of expected hospital charges for the administration of Lantidra TM in the cost analysis. Specifically, it estimated the average unstandardized charge per case to be $63,211 (hospital room $6,122, drugs $23,200, laboratory tests $11,160, imaging $12,871, procedure $5,035, and physician services $4,823), which the applicant stated represented actual hospital charges for the administration of Lantidra TM paid by the sponsor for two cases in 2022.
With respect to the estimation of an inflation factor of 10 percent that the applicant applied to the standard charges, the applicant stated that, as claims data were not used in the cost analysis of Lantidra TM , rather than applying the most recent final rule inflation factor, it used the 10 percent inflation factor in the cost threshold example tab in the FY2025 NTAP Cost Analysis spreadsheet.
To address the request for additional analysis, the applicant performed four additional cost analyses. The applicant stated that each scenario and corresponding sensitivity analysis utilized the FY 2022 MedPAR file and the results showed Lantidra TM met the cost criterion. The applicant stated that cost-to-charge ratios (CCR) were used to estimate hospital charges in the additional analysis, which may inflate charges by a multiple of approximately five times the cost and may not represent actual estimated hospital charges for the administration of Lantidra TM .
Per the applicant, the first analysis was to evaluate cases identified by ICD-10-PCS code E033U1 (Introduction of nonautologous pancreatic islet cells into peripheral vein, percutaneous approach), which best describes procedures similar to the administration of Lantidra TM . However, the applicant stated that no cases were identified and a cost analysis could not be performed.
In the second analysis, the applicant evaluated cases with an ICD-10-CM code E10649 (Type 1 diabetes mellitus with hypoglycemia without coma), regardless of MS-DRG assignment. The applicant stated this diagnosis code best describes patients that may be suitable to receive Lantidra TM based on the labeled indication. The applicant identified 14,866 cases across 523 MS-DRGs. The applicant removed 100 percent of all drug charges, which it stated was a conservative analysis as it is highly likely that patients would still receive some drugs as part of their hospital admission. The applicant stated since it could not identify which drugs Lantidra TM would necessarily replace, the analysis removed all of the drug charges to provide a conservative cost estimate. The applicant stated the analysis estimated hospital charges associated with Lantidra TM of $1,666,667 by dividing the per-administration wholesale acquisition cost (WAC) of $300,000 by the national average CCR for drugs provided in the CMS template (0.180).Using the CMS new technology add-on payment template for evaluating the cost criterion, the applicant estimated a final average inflated standardized charge per case of $1,759,387, which it stated is greater than the average case-weighted threshold amount of $80,720. The applicant conducted a sensitivity analysis by applying an estimated CAR-T CCR (0.2669) to the cost of Lantidra TM , and stated that the final average case-weighted standardized charge per case exceeded the average case-weighted threshold amount ($1,216,737 and $80,720, respectively).
In the third analysis, the applicant identified 67,277 cases in MS-DRGs 637 (Diabetes with MCC), 638 (Diabetes with CC), and 639 (Diabetes without CC/MCC). Per the applicant, the majority of ( print page 69174) Lantidra TM cases are expected to be assigned to these MS-DRGs. As in the second analysis, the applicant removed 100 percent of all drug charges for the reasons described earlier in this section. The applicant stated that using the CMS new technology add-on payment template for evaluating the cost criterion, this analysis resulted in a final average inflated standardized charge per case of $1,709,127, which is greater than the average case-weighted threshold amount of $49,659. The applicant conducted a sensitivity analysis by applying an estimated CAR-T CCR (0.2669) to the cost of Lantidra TM , and stated that the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount ($1,166,476 and $49,659, respectively).
In the fourth analysis, to further refine the analysis to patients that may be eligible for Lantidra TM , the applicant evaluated cases within MS-DRGs 637-639 with ICD-10-CM code E10649 (Type 1 diabetes mellitus with hypoglycemia without coma), which, according to the applicant, is the most appropriate code for patients that may receive Lantidra TM based on the labeled indication. The applicant identified 2,851 discharges and removed 100 percent of all drug charges for the reasons described previously. The applicant stated that using the CMS new technology add-on payment template for evaluating the cost criterion, this analysis resulted in a final average inflated standardized charge per case of $1,714,333, which is greater than the average case- weighted threshold amount of $51,535. The applicant conducted a sensitivity analysis by applying an estimated CAR-T CCR (0.2669) to the cost of Lantidra TM , and stated that the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount ($1,171,683 and $51,535, respectively).
The applicant stated that the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all additional analyses. The applicant clarified that administration of Lantidra TM is a minimally invasive procedure with moderate sedation and one night hospital stay, and asserted use of the CCR to approximate hospital charges may result in a large overestimation. The applicant stated that in the application, it added actual hospital charges from two cases in 2022 which resulted in an average standardized charge per case of $67,770 (excluding the cost of Lantidra TM ), and applied a 3-year rate of inflation factor of 1.18. Per the applicant, adding the cost of Lantidra TM results in a final average inflated standardized charge per case of $374,547, which exceeds the average case-weighted threshold amounts in all the additional cost analyses provided by the applicant.
Response: We thank the applicant for its comments and appreciate the updated cost analyses. We agree that the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in the scenarios provided. Therefore, Lantidra TM meets the cost criterion.
With regard to the substantial clinical improvement criterion, the applicant asserted that Lantidra TM represents a substantial clinical improvement over existing technologies. The applicant asserted that patients with the indication of T1D characterized by hypoglycemic unawareness are at risk of severe hypoglycemia, complications, and death, if untreated. According to the applicant, when intensive insulin therapy is not sufficient for addressing symptoms of severe hypoglycemia, Lantidra TM infusion into the hepatic portal vein offers a safe and effective minimally invasive alternative with proven clinical outcomes, fewer complications, and similar overall costs to that of whole pancreas transplantation. The applicant also asserted that Lantidra TM provides a treatment option for patients unresponsive to, or ineligible for, currently available treatments because whole pancreas transplant, a currently available treatment, is associated with greater surgical and post-procedural risk than pancreatic islet transplantation. Additionally, the applicant asserted that due to procedural risks, some patients may not be appropriate surgical candidates for whole pancreas transplantation. [ 88 ] The applicant provided two patient testimonials, one study combining results of a Phase 1/2 and a Phase 3 clinical study to support these claims, as well as one background article. [ 89 ] The following table summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for Lantidra TM for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.

As stated in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36069 ), after review of the information provided by the applicant, we had the following concerns regarding whether Lantidra TM meets the substantial clinical improvement criterion. We noted that we were interested in evidence on clinical outcomes based on a comparison of Lantidra TM with currently available treatments, including whole pancreatic transplant or recent advances in glucose monitoring and insulin delivery systems that are FDA-approved. We also noted that according to the summary of the long-term 6-year follow-up of patients from the Lantidra TM clinical trials, [ 90 ] the number of evaluable patients was reduced from 30 at the baseline to 12 at year 6. We questioned whether the small number would impact the reliability of the conclusions about insulin independence and reduction in severe hypoglycemic events. Regarding the applicant's claim that Lantidra TM patients achieved insulin independence, improved HbA1c endpoints, had fewer hypoglycemia episodes, and experienced improved quality of life, the applicant stated that the Phase 1/2 and 3 trials had over 10 years of extended follow-up, but specific results on long-term efficacy appear to be provided only up to six years post- the last transplant. [ 91 ] We noted we were interested in learning about available results from any longer-term follow-up. In addition, we stated that we were interested in data demonstrating that Lantidra TM results in improved clinical outcomes, like reduced mortality, to support an assessment of whether Lantidra TM represents a substantial clinical improvement.
We invited public comments on whether Lantidra TM meets the substantial clinical improvement criterion.
Comment: The applicant submitted a public comment responding to CMS's concerns regarding the substantial clinical improvement criterion. With regard to the request for comparison of Lantidra TM to currently available treatments, the applicant stated that patients with hard-to-control T1D are, by definition, unable to achieve adequate glycemic control despite currently available diabetes management technologies, such as continuous glucose monitors, insulin pumps, or closed-loop systems, also referred to as artificial pancreas (AP) systems. The applicant included references to studies in which patients were not able to control glucose levels using closed-loop systems. For example, according to the applicant, a clinical trial by Anderson et al. (2019) [ 92 ] demonstrated that the subcutaneous infusion of insulin with these AP systems is suboptimal, resulting in inferior insulin action and a hindrance of the ability of AP systems to cope with meals, exercise, and illness. The applicant stated that in subsequent studies by the Anderson team, in patients with T1D and with hypoglycemia unawareness and a ( print page 69176) history of severe hypoglycemia followed over 1 month, the AP reduced the time that blood glucose was below 70 mg/dL by over three-fold, but did not completely normalize glycemic control, and did not restore hypoglycemia awareness or epinephrine response to hypoglycemia induced in a hospital setting. The applicant also cited a case study of sudden death associated with severe hypoglycemia in a patient with an advanced sensor-pump device. [ 93 ] In addition, the applicant discussed a randomized controlled trial with an 18-month follow-up that tested a closed loop system where a subgroup (55 of 168) of patients in this iDCL Trial Protocol 3 [ 94 ] (the largest AP study performed to date) who were at high risk for hypoglycemia at baseline (defined as >4% continuous glucose monitoring time below 70 mg/dL) showed improved overall glycemic control and time-in range (70-180 mg/dL), but still had residual hypoglycemia and their hypoglycemia awareness did not improve. [ 95 ]
The applicant reiterated that the hard-to-control T1D subgroup of patients does not adequately benefit from current diabetes management technologies, including AP systems. The applicant also cited studies that islet transplantation impacted health-related quality of life of patients by reducing fear of hypoglycemia, changing behaviors adopted to avoid hypoglycemia, reducing depression and confusion, or allowing patients to better engage in vacationing and vigorous physical activities such as hiking, sprinting, etc. For example, the applicant cited a study by Häggström et al. (2011), in which 11 patients who had received islet transplants at Uppsala University Hospital were surveyed about their fear of hypoglycemia, health-related quality of life, and social life situation in relation to their fear of hypoglycemia. The applicant asserted that while the results for health-related quality of life were lower than in the normal population, changes in fear of hypoglycemia suggested an improvement for the patients who had undergone islet transplantation. The applicant stated that the patients felt they experienced improved control over their social situations. [ 96 ] The applicant mentioned a study by Radosevich et al. (2013), [ 97 ] in which 27 patients with T1D had undergone an islet transplant alone (ITA) procedure at the University of Minnesota. According to the applicant, ITA was found to be related to reductions in behaviors adopted to avoid hypoglycemia (P < 0.001) and attenuation in concerns about hypoglycemic episodes (P < 0.001). Further, the applicant stated that health status among the patients who had undergone ITA was also found to have improved, according to scores on the Euro Quality of Life scale (P = 0.002) and the Beck Depression Inventory scale (P = 0.003).
With regards to the concern about long term follow up and the small number of evaluable patients in the studies provided as evidence in the original application, the applicant explained that the number of evaluable patients decreases in each subsequent year after the last islet transplant in part because some of the patients had not yet reached yearly milestones at the time of data cutoff [ 98 ] and because the follow up year resets for patients that receive an additional transplant. Per the applicant, this also accounts for the difference in the number of years of extended follow up referenced. The applicant stated that the statement in the application describing more than 10 years of extended follow-up for patients in Phase 1/2 and 3 clinical trials referred to their overall experience. The applicant further stated that although the number of patients in the clinical trials supporting Lantidra TM is necessarily small, given the carefully limited subset of patients treated with this therapy, the efficacy outcomes are representative of larger studies and clinical data.
The applicant stated that long term studies of more than 10 years have provided direct evidence that islet transplantation in patients with T1D can markedly improve metabolic control and suppress severe hypoglycemic events. [ 99 100 ] According to the applicant, a Marfil-Garza et al. (2022) study published 20-year findings of pancreatic islet cell transplantation from the University of Alberta in Edmonton, Canada. The applicant stated that the cohort study included 255 patients and illustrated the long-term safety of islet cell transplantation. The applicant stated that over the median follow-up of 7.4 years, 90 percent of patients survived with a median islet transplant survival time of 5.9 years. The applicant asserted that patients with sustained graft survival demonstrated significantly higher rates of insulin independence as well as better sustained glycemic control compared with patients with non-sustained graft survival. [ 101 ] The applicant stated that these outcomes were consistent with those of Hering et al (2022), [ 102 ] who reported the outcomes of islet transplantation in 398 recipients with T1D complicated by severe hypoglycemic episodes reported to the Collaborative Islet Transplant Registry. Per the applicant, the Hering team identified age, islet dose, and concomitant immunosuppression protocols as factors associated with favorable outcomes, and demonstrated that when these factors were met, 53 percent of the patients were insulin independent at 5 years following transplant, 76% had an HbA1c under seven percent, and 95 percent were free of severe hypoglycemic events.
The applicant concluded in its public comments that the clinical data supports the safety and efficacy of FDA-approved Lantidra TM to address an unmet medical need for a small subset of patients with hard-to-control T1D complicated by severe hypoglycemic episodes.
A commenter also submitted a public comment in support of Lantidra TM . The commenter stated the benefits of Lantidra TM include being available for patients with hard-to-control diabetes. The commenter cited the unpublished results from a NIH-funded Phase 3 safety and efficacy study for islet cell transplantation as evidence to support the use of islet transplantation to improve the clinical outcomes of this ( print page 69177) subgroup of patients. According to the commenter, 87.5 percent of patients in the NIH-funded study achieved glucose control and freedom from severe hypoglycemic events at 1 year and 71 percent at 2 years, while 52 percent of patients achieved insulin independence a year after transplant. The commenter stated that Lantidra TM uniquely fills an unmet need and provides an important therapy option for T1D patients who are at an increased risk for severe morbidity and mortality.
Response: We thank the applicant and the commenter for their comments regarding the substantial clinical improvement criterion. Based on the additional information received, we continue to have concerns as to whether Lantidra [ TM ] meets the substantial clinical improvement criterion to be approved for new technology add-on payments. Regarding the applicant's assertion that patients with hard to control T1D are, by definition, unable to achieve glycemic control despite currently available diabetes management technologies such as continuous glucose monitors, insulin pumps, or closed-loop systems, we disagree that the data presented adequately supports this assertion. Specifically, we note the many technological advancements in glucose level detection and insulin delivery that have evolved since the beginning of islet cell transplantation. Continuous glucose monitoring systems with real time readings and alert systems can identify episodes of hyper- and hypoglycemia. Trend and pattern data can inform insulin dosing, and we note that severe hypoglycemic episodes and high glycemic variability can be prevented or significantly reduced by setting threshold alarms and having pumps that suspend and control insulin infusion with automated insulin delivery systems. [ 103 104 105 106 ] It is not clear from the additional evidence presented by the applicant that patients eligible for treatment with Lantidra TM could not be appropriately managed with the most recent diabetes management systems that are available.
In addition, while the applicant provided studies to demonstrate that current therapies are inadequate, we note that the Anderson et al. (2019) [ 107 ] study cited by the applicant compared sensor-augmented pump (SAP) therapy to hybrid closed-loop control (HCLC), and per the study, HCLC reduced the risk and frequency of hypoglycemia while improving time in target range. We further note that with regard to the studies provided by the applicant to demonstrate that current therapies do not improve hypoglycemia unawareness, we are unclear how these studies demonstrate a patient population unresponsive to current therapies, particularly since the studies concluded that the therapies used improved hypoglycemia. We further note that the applicant did not assert or otherwise demonstrate that Lantidra TM improves hypoglycemia unawareness. Additionally, the applicant provided a case study of a patient with an advanced sensor pump, and stated that the patient had sudden death due to severe hypoglycemia. However, we note that the study stated that it remained unclear whether hypoglycemia caused the sudden death, as the accuracy of glucose levels on these pumps during cardiopulmonary resuscitation was doubtful. [ 108 ]
With regard to the studies cited by the applicant that islet transplantation impacted health-related quality of life, and the study provided by the commenter regarding safety and efficacy of islet cell transplantation, we note that the additional data provided did not assess Lantidra TM , but instead included data from other formulations of islet cells for transplantation. As a result, we are unclear how these formulations may compare to that of Lantidra TM . We further note that the Hering et al. (2023) study [ 109 ] cited by the applicant identified islet dose and concomitant immunosuppression protocols as factors that could affect outcomes; however we are unclear that data using other formulations of islet cells or transplant protocols, which may vary by clinical site, is relevant to the assessment of Lantidra TM specifically.
We also remain concerned with regard to the generalizability of the outcomes given the small number of evaluable patients. We note the applicant stated that the number of evaluable patients decreased in each subsequent year after the last islet transplant in part because some of the patients had not yet reached yearly milestones at the time of data cutoff, and because the follow up year resets for patients that receive an additional transplant, and also the number of patients in the clinical trials supporting Lantidra TM were necessarily small, given the carefully limited subset of patients treated with this therapy and the efficacy outcomes are representative of larger studies and clinical data. Regardless, we question whether the patients included in the UIH-001 and UIH-002 studies met the criteria for this specific subset of patients, defined as hard to control T1D complicated by severe hypoglycemia, given that at baseline, 37 percent of transplant recipients had a HbA1c at target, and 83 percent of patients in the trial did not have a documented severe hypoglycemic event in the year prior to transplant, according to the FDA panel review. According to the FDA BLA Clinical Review Memorandum, [ 110 ] in the discussion of HbA1c: Of the thirty subjects, 11 (37 percent) had an HbA1c of ≤7% prior to transplant, and 6 (20 percent) had an HbA1c ≤6.5%, with 6.5% and 7% being accepted targets for good glycemic control in diabetic patients. Therefore, it is unclear that these patients would represent the narrower subset of patients, as described by the applicant.
In addition, in response to our question about the availability of evidence that Lantidra TM improved clinical outcomes, like reduced mortality, to inform our assessment of whether Lantidra TM represents a substantial clinical improvement, the applicant cited long-term studies of more than 10 years. According to the applicant, these studies provided direct evidence that islet transplantation in patients with T1D can markedly improve metabolic control and suppress severe hypoglycemic events. The applicant further cited a Marfil-Garza et ( print page 69178) al. (2022) [ 111 ] study which published 20-year findings of pancreatic islet cell transplantation from the University of Alberta in Edmonton, Canada. The applicant stated that the cohort study included 255 patients and illustrated the long-term safety of islet cell transplantation. The applicant further stated that over the median follow-up of 7.4 years, 90 percent of patients survived with a median islet transplant survival time of 5.9 years. The applicant asserted that patients with sustained graft survival demonstrated significantly higher rates of insulin independence as well as better sustained glycemic control compared with patients with non-sustained graft survival. The applicant also stated that these outcomes were consistent with those of the Hering et al. (2023) study, who reported the outcomes of islet transplantation in 398 recipients with T1D complicated by severe hypoglycemic episodes reported to the Collaborative Islet Transplant Registry. Per the applicant, the Hering team identified age, islet dose, and concomitant immunosuppression protocols as factors associated with favorable outcomes, and demonstrated that when these factors were met, 53 percent of patients were insulin independent at 5 years following transplant, 76 percent had an HbA1c under seven percent, and 95 percent were free of severe hypoglycemic events. With regards to the studies cited by the applicant, we note that the additional data provided did not assess the use of Lantidra TM , and given the potential differences in the islet cell formulation used at different transfusion facilities and the potential differences in the various transplant protocols, we question whether the reported results are replicable or comparable to Lantidra TM .
After consideration of all the information received from the applicant, as well as the public comments we received, we are unable to determine that Lantidra TM represents a substantial clinical improvement over existing technologies for the reasons discussed in the proposed rule and in this final rule, and therefore, we are not approving new technology add-on payments for Lantidra TM for FY 2025.
Iovance Biotherapeutics, Inc. submitted an application for new technology add-on payments for AMTAGVI TM for FY 2025. According to the applicant, AMTAGVI TM is a one-time, single-dose autologous tumor-infiltrating lymphocyte (TIL) immunotherapy for the treatment of advanced (unresectable or metastatic) melanoma comprised of a suspension of TIL for intravenous infusion. We note that Iovance Biotherapeutics submitted an application for new technology add-on payments for AMTAGVI TM for FY 2022 under the name lifileucel, as summarized in the FY 2022 IPPS/LTCH PPS proposed rule ( 86 FR 25272 through 25282 ) but withdrew the application prior to the issuance of the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44979 ). We also note that the applicant submitted an application for AMTAGVI TM for FY 2023 under the name lifileucel, as summarized in the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28244 through 28257 ), that it withdrew prior to the issuance of the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48920 ).
Please refer to the online application posting for AMTAGVI TM , available at https://mearis.cms.gov/public/publications/ntap/NTP231012V8Y9J , for additional detail describing the technology and the treatment of unresectable or metastatic melanoma.
With respect to the newness criterion, according to the applicant, AMTAGVI TM was granted Biologics License Application (BLA) approval from FDA on February 16, 2024, for treatment of adult patients with unresectable or metastatic melanoma previously treated with a programmed cell death protein 1 (PD-1) blocking antibody, and if B-raf proto-oncogene (BRAF) V600 mutation positive, a BRAF inhibitor with or without a mitogen-activated extracellular signal-regulated kinase (MEK) inhibitor. The applicant stated that AMTAGVI TM has received Regenerative Medicine Advanced Therapy (RMAT), Orphan Drug, and Fast Track designations from FDA for the treatment of advanced melanoma. According to the applicant, AMTAGVI TM was expected to be commercially available within 30-40 days post-FDA approval due to the need for the physician to prescribe AMTAGVI TM , the treatment center to receive approval from the patient's insurer and to schedule and surgically resect the patient's tumor tissue, the 22-day TIL manufacturing process, and shipment/invoicing of AMTAGVI TM to the treatment center for patient administration. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36069 ), we stated we were interested in additional information regarding the delay in the technology's market availability, as it seems that the technology would need to be available for sale before a physician would be able to prescribe AMTAGVI TM .
According to the applicant, AMTAGVI TM is provided as a single dose for infusion containing a suspension of TIL in up to four patient-specific intravenous (IV) infusion bag(s), with each dose containing 7.5 × 107 ‸ 9 to 72 × 10 ‸ 9 viable cells. The applicant further noted that there is a lymphodepleting regimen administered before infusion of AMTAGVI TM , and post-AMTAGVI TM infusion, an interleukin 2 (IL-2) infusion at 600,000 IU/kg is administered every 8 to 12 hours, for up to a maximum of 6 doses, to support cell expansion in vivo.
The applicant stated that effective October 1, 2022, the following ICD-10-PCS codes may be used to uniquely describe procedures involving the use of AMTAGVI TM : XW033L7 (Introduction of lifileucel immunotherapy into peripheral vein, percutaneous approach, new technology group 7), and XW043L7 (Introduction of lifileucel immunotherapy into central vein, percutaneous approach, new technology group 7). The applicant stated that all diagnosis codes under the category C43 (Malignant melanoma of skin) may be used to currently identify the indication for AMTAGVI TM under the ICD-10-CM coding system.
With respect to the substantial similarity criteria, the applicant asserted that AMTAGVI TM is not substantially similar to other currently available technologies because TIL immunotherapy with AMTAGVI TM has a novel and unique mechanism of action that delivers a highly customized, personalized, and targeted, single-infusion treatment for advanced melanoma, and AMTAGVI TM is the first and only TIL immunotherapy approved for the treatment of advanced (unresectable or metastatic) melanoma, and that therefore, the technology meets the newness criterion. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for AMTAGVI TM for the applicant's complete statements in support of its assertion that AMTAGVI TM is not substantially similar ( print page 69179) to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36071 ), we invited public comments on whether AMTAGVI TM is substantially similar to existing technologies and whether AMTAGVI TM meets the newness criterion.
Comment: The applicant submitted a public comment regarding the newness criterion. The applicant reiterated that AMTAGVI TM is the first and only one-time, individualized T-cell therapy to receive FDA approval as a treatment for a solid tumor cancer. The applicant stated that the proposed mechanism for AMTAGVI TM offers a new cell therapy approach that deploys patient-specific T-cells called TIL cells. The applicant stated that when cancer is detected, the immune system creates TIL cells to locate, attack, and destroy cancer and that TIL cells recognize distinctive tumor markers on the cell surface of each person's cancer. The applicant stated that when cancer develops and prevails, the body's natural TIL cells can no longer perform their intended function to fight cancer. The applicant asserted that TIL cell therapy with AMTAGVI TM uses autologous T-cells isolated from the tumor tissue and expanded ex vivo using a centralized manufacturing process, maintaining the heterogeneous repertoire of T-cells without any prior selection or genetic modification. The applicant stated that by isolating autologous TIL from the tumor microenvironment and expanding them ex vivo in the presence of growth factors, the AMTAGVI TM manufacturing process produces large numbers of reinvigorated T-cells. Further, the applicant asserted that following a one-time infusion of the personalized AMTAGVI TM immunotherapy, the TIL migrates back into primary and metastatic tumors, where they amplify and rejuvenate the patient's own immune system, triggering specific tumor cell killing upon recognition of tumor antigens.
The applicant also responded to CMS's request for additional information regarding the delay in the technology's market availability, stating that AMTAGVI TM was granted FDA approval on February 16, 2024, and became immediately available for providers to order for patients. The applicant further stated that as a tumor-derived autologous T-cell immunotherapy that is individualized for each patient, there is a several-week process for each patient to receive AMTAGVI TM . The applicant stated that AMTAGVI TM is manufactured from resected patient tumor tissue prosected from one or more tumor lesions. [ 117 ] The applicant stated that initially, it had targeted a 34-day turnaround time from the receipt of starting material at their centralized GMP facility to return shipment to the treatment center, which includes the AMTAGVI TM manufacturing and testing/release processes. However, the applicant stated it expected this time to decrease as the product becomes more widely available. In addition, the applicant stated there is a 7-day preconditioning regimen prior to AMTAGVI TM infusion, and as a result, the first AMTAGVI TM shipment to a treatment center was on March 28, 2024, and the first patient was infused on April 4, 2024. The applicant requested April 4, 2024, as the start of the AMTAGVI TM newness period for the new technology add-on payment.
Response: Based on our review of comments received and information submitted by the applicant as part of its FY 2025 new technology add-on payment application for AMTAGVI TM , we agree with the applicant that AMTAGVI TM is the first TIL therapy in the treatment of advanced melanoma and, therefore, has a new mechanism of action compared to existing technologies. We also believe that the use of AMTAGVI TM would be assigned to a different MS-DRG than existing technologies to treat patients with advanced melanoma since AMTAGVI TM maps to Pre-MDC MS-DRG 018 (Chimeric Antigen Receptor (CAR) T-cell and Other Immunotherapies). Therefore, we agree with the applicant that AMTAGVI TM is not substantially similar to existing treatment options and meets the newness criterion.
With respect to the applicant's request to start the AMTAGVI TM newness period for the new technology add-on payment on April 4, 2024, we note that the timeframe that a new technology can be eligible to receive new technology add-on payments begins when data become available ( 69 FR 49003 , 85 FR 58610 ). Consistent with the statute, a technology no longer qualifies as “new” once it is more than 2 to 3 years old, irrespective of how frequently it has been used in the Medicare population. ( print page 69181) Therefore, if a product is more than 2 to 3 years old, we consider its costs to be included in the MS-DRG relative weights whether its use in the Medicare population has been frequent or infrequent. While our policy is generally to begin the newness period on the date of FDA approval or clearance, we may consider a documented delay in a technology's market availability in our determination of newness ( 87 FR 48977 ). However, we do not consider the date of first sale of a product, or first shipment of a product, as an indicator of the entry of a product onto the U.S. market; none of these dates indicate when a technology became available for sale ( 88 FR 58802 ). Similarly, the date of first infusion of a product does not indicate when a technology became available for sale. As the applicant noted, AMTAGVI TM was granted FDA approval on February 16, 2024, and became immediately available for providers to order for patients. Therefore, it appears that AMTAGVI TM was available for sale starting February 16, 2024. We consider the beginning of the newness period to commence on February 16, 2024, when AMTAGVI TM was granted BLA approval from FDA for treatment of adult patients with unresectable or metastatic melanoma previously treated with a PD-1 blocking antibody, and if BRAF V600 mutation positive, a BRAF inhibitor with or without a MEK inhibitor.
With respect to the cost criterion, the applicant provided multiple analyses to demonstrate that it meets the cost criterion. For each analysis, the applicant searched the FY 2022 MedPAR file using different combinations of ICD-10-CM codes, ICD-10-PCS codes, and/or inpatient length-of-stay (LOS) of 10 or more days. The applicant explained that it used different combinations to demonstrate four different cohorts that may be eligible for the technology. According to the applicant, eligible cases for AMTAGVI TM will be mapped to Pre-MDC MS-DRG 018 (Chimeric Antigen Receptor (CAR) T-cell and Other Immunotherapies). For each analysis, the applicant used the FY 2025 new technology add-on payments threshold for Pre-MDC MS-DRG 018 for all identified cases. Each analysis followed the order of operations described in the table later in this section.
For the first analysis, the applicant searched for potential cases for the following combination of ICD-10-CM diagnosis/procedure codes: any melanoma and metastasis diagnosis codes and any IL-2 or chemotherapy procedure codes. Please see the online posting for AMTAGVI TM for the complete list of codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 176 claims mapping to 16 MS-DRGs, with each MS-DRG representing 6.3 percent of identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $2,150,682, which exceeded the average case-weighted threshold amount of $1,374,450.
For the second analysis, the applicant searched for potential cases for the following ICD-10-CM diagnosis/procedure codes in combination with an inpatient LOS of 10 or more days: any melanoma and metastasis diagnosis codes and any IL-2 or chemotherapy procedure codes. Please see the online posting for AMTAGVI TM for the complete list of codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 77 claims mapping to seven MS-DRGs, with each MS-DRG representing 14.3 percent of identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $2,207,367, which exceeded the average case-weighted threshold amount of $1,374,450.
For the third analysis, the applicant searched for potential cases for the following combination of ICD-10-CM diagnosis/procedure codes: a code describing primary or admitting diagnosis of melanoma and a metastasis diagnosis code. Please see the online posting for AMTAGVI TM for the complete list of codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 735 claims mapping to 64 MS-DRGs, with each MS-DRG representing 3.4 percent to 1.5 percent of identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $2,017,903, which exceeded the average case-weighted threshold amount of $1,374,450.
For the fourth analysis, the applicant searched for potential cases for the following combination of ICD-10-CM diagnosis/procedure codes: a code describing any diagnosis of melanoma and a metastasis diagnosis code. Please see the online posting for AMTAGVI TM for the complete list of codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 6,648 claims mapping to 358 MS-DRGs, each MS-DRG representing 0.2 percent to 6.7 percent of identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $2,018,905, which exceeded the average case-weighted threshold amount of $1,374,450.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant asserted that AMTAGVI TM meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36074 ), we invited public comments on whether AMTAGVI TM meets the cost criterion.
We did not receive any comments on whether AMTAGVI TM meets cost criterion. Based on the information submitted by the applicant as part of its FY 2025 new technology add-on payment application, as previously summarized, the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount. Therefore, AMTAGVI TM meets the cost criterion.
With regard to the substantial clinical improvement criterion, the applicant asserted that AMTAGVI TM represents a substantial clinical improvement over existing technologies because the efficacy and safety profile of the single infusion of AMTAGVI TM TIL immunotherapy addresses an important unmet need in the advanced (unresectable or metastatic) melanoma population who lack effective or approved treatment options after being previously treated with ICI therapy. The applicant asserted that the clinically meaningful and durable activity of AMTAGVI TM represents a substantial clinical improvement over published outcomes for chemotherapy. The applicant provided four studies to support these claims, as well as 22 background articles about treatments for advanced melanoma. [ 119 ]
The following table summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for AMTAGVI TM for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36075 through 36076 ), after reviewing the information provided by the applicant, we stated we had the following concerns regarding whether AMTAGVI TM meets the substantial clinical improvement criterion.
In support of its application, the applicant provided data from the C-144-01 study, an ongoing phase two multicenter study (NCT02360579) to assess the efficacy and safety of autologous TIL in patients with stage IIIc-IV metastatic melanoma, which consisted of: Cohort 1 (n = 30 generation 1 no-cryopreserved TIL product); Cohort 2 (n = 66 generation 2 cryopreserved TIL product); Cohort 3 (a sub-sample of n = 10 from Cohorts 1, 2, and 4); and Cohort 4 (n = 75 generation 2 cryopreserved TIL product). Regarding the sample studied (Cohorts 2 & 4 combined) by Chesney, et al. (2022), [ 120 ] similar to concerns raised in the FY 2022 IPPS/LTCH PPS proposed rule ( 86 FR 25281 ), we stated that we continued to question the appropriateness of combining Cohorts 2 and 4 together. Furthermore, similar to concerns raised in the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28256 through 28257 ), we noted that in the study of Chesney, et al. (2022), 54 percent of the sample size included males with a median age of 56; data on race, ethnicity, and other demographics are not presented. Given that the average age of Medicare beneficiaries is substantially older, and that Medicare beneficiaries often have multiple comorbidities, we questioned whether the sample evaluated is appropriately representative of the Medicare population and whether this sample has a disease burden similar to that seen in Medicare beneficiaries. [ 121 122 123 ] Thus, ( print page 69185) similar to concerns raised in the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28256 through 28257 ), we stated we were concerned that the findings may not be generalizable to Medicare beneficiaries. Furthermore, as discussed in the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28256 ), we noted that we continued to question whether the patient sample evaluated in the Sarnaik et al. (2021) [ 124 ] study is appropriately representative of the Medicare population and whether this sample has a disease burden similar to that seen in Medicare beneficiaries.
Second, similar to concerns raised in the FY 2022 IPPS/LTCH PPS proposed rule ( 86 FR 25279 through 25282 ) and the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28256 through 28257 ), we stated that we continued to note that while multiple background studies were provided in support of the applicant's claims for substantial clinical improvement, those that evaluate AMTAGVI TM were based solely on the C-144-01 trial. The background studies focus primarily on describing the limitations of other therapies rather than supporting the role of AMTAGVI TM , and no direct comparisons to other existing therapies, such as targeted therapies with combination BRAF plus MEK inhibitors or nivolumab plus ipilimumab, were provided. Therefore, we stated that we would be interested in additional information comparing AMTAGVI TM to existing treatments (for example, evidence comparing AMTAGVI TM phase two studies to the phase two studies of existing or approved treatments by using meta-analysis after systematic review, or evidence based on retrospective cohort studies of the relevant patients to assess whether AMTAGVI TM had significantly different impact on any outcomes compared to existing or approved treatments).
Third, similar to concerns raised in the FY 2022 IPPS/LTCH PPS proposed rule ( 86 FR 25279 through 25282 ), and the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28256 through 28257 ), we noted that the Chesney et al. (2022) [ 125 ] study uses a surrogate endpoint, ORR, which combines the results of complete and partial responders; we questioned whether this correlated to improvement in clinical outcomes such as overall survival (OS).
Finally, similar to concerns raised in the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28256 through 28257 ), we noted that according to the applicant, high-dose IL-2 has been used to treat metastatic melanoma in the past and is given as a post-treatment to AMTAGVI TM . According to the applicant, the occurrence of grade 3 and 4 treatment-emergent adverse events (TEAEs) was early and consistent with the lymphodepletion regimen (NMA-LD) and known profile of IL-2. If AMTAGVI TM is always given in conjunction with the pre- and post-treatments, we questioned how it is possible to determine the cause of the TEAEs which are categorized as severe based on the Common Terminology Criteria for Adverse Events v4.03. We noted that we continued to question whether the effect seen in C-144-01 is due to AMTAGVI TM itself or due to other factors such as the use of IL-2, general changes in medical practice over time, and the specific sample identified for the trial at hand.
We invited public comments on whether AMTAGVI TM meets the substantial clinical improvement criterion.
Comment: The applicant submitted a public comment regarding the substantial clinical improvement criterion and provided responses to CMS's concerns in the proposed rule. With regards to the appropriateness of combining cohorts in the C-144-01 study, the applicant stated that the findings for Cohorts 2 and 4 were presented separately as well as combined (pooled results), and that in both cases AMTAGVI TM demonstrated a substantial clinical improvement in ORR over chemotherapy, which the applicant asserted offers poor ORR (4-10%). Specifically, the applicant stated that Cohorts 2 and 4 provided ORRs of 34.8 percent and 28.7 percent, respectively, when assessed separately, and an ORR of 31.4 percent when combined. [ 126 ] The applicant asserted that pooling efficacy and safety from Cohort 2 and 4 increased the sample size and therefore supported confidence in the point estimates of efficacy (ORR and DOR) and better characterized the AMTAGVI TM safety profile. Additionally, the applicant clarified that both cohorts used the same eligibility criteria and enrolled similar patient populations as demonstrated by the baseline disease characteristics, patient demographics and prior therapies received; and both cohorts had the same primary and secondary objectives. The applicant stated that to minimize investigator bias, both Cohort 2 and 4 evaluated the efficacy of AMTAGVI TM in patients with unresectable or metastatic melanoma using the ORR. Lastly, the applicant stated that the investigational product used in Cohort 2 and 4 was manufactured using the same manufacturing process and was released using the same product specification. The applicant also provided the pooled efficacy results that were assessed by FDA that included 153 patients (from Cohorts 2 and 4). Therefore, the applicant concluded that pooling Cohorts 2 and 4 from Study C-144-01 provided the most comprehensive dataset supporting the safety and efficacy of AMTAGVI TM.
In addition, in response to CMS's concerns regarding whether the C-144-01 study population age, demographics, and disease burden were representative of the Medicare population, the applicant provided information about the inclusion criteria for Cohorts 2 and 4. The applicant noted that Medicare beneficiaries with advanced melanoma qualify for Medicare coverage by age (65 or greater) or disability (any age insured by Social Security Disability Insurance, SSDI) as well as noting that the study C-144-01 population is extrapolatable across the advanced melanoma Medicare-age population, with 24 percent of enrollees in Cohorts 2 and 4 aged 66 years or older. [ 127 128 ] The applicant stated that between Cohorts 2, 4 and the pooled cohorts, all patients had a high disease burden (median target lesion sum of diameters [SOD] was 98 mm) at baseline, and patients had received a median of 3 lines of prior therapies (range, 1 to 9). The applicant stated that all patients had a confirmed progressive disease on their prior therapy before study entry. The applicant stated that as reported by Chesney et al, 2022, response to AMTAGVI TM was observed across all subgroups analyzed including by age group (<65 years and greater than or equal to 65 years), where ORR was similar, and that this data is most representative of the real-life population with advanced melanoma. The applicant stated that moreover, when creating its Category of Evidence 2A recommendation for AMTAGVI TM , the ( print page 69186) National Comprehensive Cancer Network (NCCN) Guidelines did not distinguish its recommendation by age. The applicant asserted that accessibility to AMTAGVI TM treatment is highly important to all Medicare beneficiaries whether eligible by age or disability status; specifically, increasing age is associated with a higher incidence of melanoma death. The applicant also provided the study C-144-01 inclusion requirements for additional context.
Regarding CMS's interest in additional information comparing AMTAGVI TM to existing treatment, the applicant stated that there are no approved therapies for the line of treatment for which AMTAGVI TM was approved, and that chemotherapy is the most commonly used therapy for patients with advanced melanoma post-progression. The applicant stated that most patients with advanced melanoma relapse on, or do not tolerate, treatment with ICls and BRAF-targeted therapies and respond poorly to subsequent rounds of therapy with these agents. The applicant stated that primary resistance to immune checkpoint blockade occurs in approximately 40 to 65 percent of patients with melanoma treated with anti-PD-1 based therapy, depending on whether anti-PD-1 therapy is given upfront or after progression on other therapies, and in >70 percent of those treated with anti-CTLA-4 therapy. The applicant stated that of those with initial disease control, 30 to 40 percent develop acquired resistance. The applicant stated that approximately 15 to 20 percent of BRAF V600 mutation-positive patients fail to respond to targeted therapy initially, and only 22 percent remain progression-free at 3 years. The applicant noted that although primary resistance is lower in patients treated with anti-PD-1 therapy plus anti-CTLA-4 therapy, 38 percent of patients discontinue therapy because of treatmentemergent adverse events (TEAEs), with 88 percent developing immune-related adverse events (irAEs), many of these being persistent. The applicant further stated that before AMTAGVI TM 's approval, there were no FDA-approved treatment options for patients with advanced melanoma whose disease progressed following initial treatment with an immune checkpoint inhibitor and, if appropriate, targeted therapy. The applicant stated that in its FY 2025 new technology add-on payment application, ORR and OS results for C-144-01 were compared to chemotherapy and asserted that only 4 percent to 10 percent of advanced melanoma patients who progress after retreatment have objective responses to chemotherapy, with a median OS of 7 months. The applicant stated that in contrast, a single infusion of AMTAGVI TM provides clinically meaningful and durable responses in patients with advanced melanoma previously treated with ICI therapy. The applicant stated that in a four-year analysis of C-144-01 submitted to CMS in February 2024 as supplemental information for its new technology add-on payment application, the longest duration of independent review committee (IRC)-assessed response was ongoing at 55.8 months. The applicant stated that the median DOR was not reached; median OS was 13.9 months, with 1-, 2-, 3-, and 4-year OS rates of 54.0%, 33.9%, 28.4%, and 21.9%, respectively. The applicant stated that no new late-onset AMTAGVI TM -related serious AE was reported. The applicant stated that based on its estimation of ORR, the planned sample size of Cohort 2 was 66 patients, and the planned sample size for Cohort 4 was 75 patients to demonstrate statistical significance to the historical ORR. The applicant provided additional methodology for its hypothesis testing for the primary endpoint of Cohort 4 as assessed by the IRC. The applicant also stated that AMTAGVI TM recently became the first and the only Category of Evidence 2A designated agent approved on-label for second line therapy in advanced melanoma in an April 2024 update to the NCCN Guidelines, as a preferred high-dose therapy as secondline or subsequent systemic therapy, as a component of TIL therapy unresectable disease, and after progression on anti-PD-1-based therapy and BRAF/MEK inhibitor therapy (if BRAF V600 mutation positive). The applicant asserted that the clinically meaningful and durable responses following the single infusion of AMTAGVI TM address the high unmet medical need in patients with advanced melanoma and high tumor burden, who are heavily pre-treated and difficult-to-treat and have a poor prognosis with no treatment options available after progression on immunotherapy and targeted agents or who are primary refractory to anti-PD-1/PD-Ll therapy.
In response to CMS's concern about using a surrogate endpoint, ORR, and whether it correlated to clinical outcomes such as OS, the applicant cited an analysis of C-144-01 in patients who achieved response at first assessment (6 weeks or approximately 1.5 months) and provided a Kaplan-Meier curve showing a statistically significant difference in overall survival between patients who achieved an early response versus non-responders. [ 129 ] The applicant also referred to a public comment letter to CMS on June 17, 2021 from the principal investigator for the C-144-01 trial that directed CMS to FDA guidance to industry describing the significance of ORR as assessed by its magnitude and duration of effect. In addition, the applicant stated that the guidance states use of ORR can represent direct clinical benefit based on the specific disease, context of use, magnitude of effect, the number of complete responses, the durability of response, and other factors. [ 130 ] The applicant stated that in addition to the association between early response and OS in the C-144-01 landmark analysis, further evidence was presented from a multicenter, randomized Phase 3 trial evaluating treatment with locally-produced TIL therapy versus ipilimumab (n=84 per arm) for advanced melanoma. [ 131 132 ] The applicant indicated that in this study, the overall survival of responders compared to non-responders had a hazard ratio of 0.14 (95% CI: 0.06, 0.33, p < .001), favoring TIL responders, and provided additional results for the TIL study population. The applicant also stated that AMTAGVI TM was approved under FDA's accelerated approval program, which allows for earlier approval of drugs that treat serious conditions and fill an unmet medical need, and can also be based on the effect on a “surrogate endpoint,” such as ORR that is reasonably likely to predict clinical benefit. Finally, the applicant noted that CMS has a well-established history of granting new technology add-on payment status to drugs and biologics that receive FDA approval under the accelerated approval pathway. The applicant stated that based on a review of past IPPS/LTCH PPS final rules, effective in FY 2016 and extending through FY 2024, CMS had 10 approvals for new technology add-on payments for new therapies approved under FDA's accelerated approval pathway for oncology and hematology uses, and the majority of these therapies were granted FDA accelerated approvals based on surrogate efficacy endpoints, including the same ORR and DOR endpoints used for accelerated approval ( print page 69187) of AMTAGVI TM . Another commenter emphasized the importance of the use of surrogate/intermediary endpoints within clinical trials for immunotherapy and that it is a critically important tool that emphasizes both patient access and scientific rigor.
Regarding CMS's concerns related to the cause of grade 3 and 4 TEAEs and questions about being able to distinguish the effect of AMTAGVI TM from other factors that are part of the treatment process, the applicant asserted that the C-144-01 study and other publications have had consistent findings about the safety profile of the TIL regimen. The applicant stated that TEAEs associated with TIL treatment have been found to be largely due to the lymphodepleting preparative regimen or IL-2 components of the TIL regimen. [ 133 ] The applicant stated that in C-144-01, the safety profile of the AMTAGVI TM regimen is consistent with the underlying advanced disease and the known safety profiles of NMA-LD and IL-2, with no new safety signal identified during long-term follow-up, and that the safety profile was similar between cohorts 2 and 4. The applicant stated that most TEAEs were transient, expected, and manageable; the incidence decreased rapidly over the first two weeks after AMTAGVI TM infusion. Furthermore, the applicant stated that the autologous nature of AMTAGVI TM was associated with a low risk for off-target effects and no long-term toxicities related to the AMTAGVI TM regimen have been noted. The applicant asserted that AMTAGVI TM produced durable response and a favorable safety profile across subgroups of heavily pretreated patients with high tumor burden, regardless of age, BRAF mutation status, PD-L1 status, baseline ECOG PS status, and presence of liver and/or brain lesions at baseline. With respect to the role of IL-2 in the AMTAGVI TM regimen, the applicant stated IL-2 is not used for its antineoplastic effect but is intended to support the activation, proliferation, and cytolytic activity of AMTAGVI TM . The applicant cited two post-hoc analyses of study C-144-01 by Hassel, et al. (2022) [ 134 ] and Larkin, et al. (2023) [ 135 ] which concluded that the abbreviated course of high-dose IL-2 (600,000 IU/kg, ≤6 doses) used as part of the AMTAGVI TM regimen to promote T-cell activity does not independently contribute to anti-neoplastic activity. Another commenter asserted that experiments in the commenter's lab were clear in demonstrating that the impact of IL-2 alone depends on the stimulation of endogenous T-cells with antitumor activity and stated that the AMTAGVI TM therapy protocol involves giving cells with antitumor activity and then IL-2, so that IL-2 solely acts on the administered cells to mediate antitumor effects. This commenter further claimed that the efficacy and durability of TIL has been shown in patients who progressed after previous IL-2 monotherapy, and also asserted that the toxicities of TIL therapy have been extensively reported and are largely due to the lymphodepleting preparative regimen or IL-2.
We also received several additional comments in support of the application for AMTAGVI TM that addressed points related to the substantial clinical improvement criterion. Several commenters indicated general support for AMTAGVI TM as a therapy that can provide a new treatment option for individuals with advanced melanoma who have not responded to standard of care treatments. A commenter stated that FDA approval of AMTAGVI TM reflects a significant advancement in TIL cell therapy, while another commenter stated its appreciation of CMS's efforts to improve patient access to novel cell therapies like AMTAGVI TM . One commenter also encouraged CMS to assign new technology add-on payment status for new treatments and technologies supporting personalized medicine that meet the required criteria, including the application for AMTAGVI TM as an example.
Response: We thank the applicant and commenters for their comments regarding the substantial clinical improvement criterion. Based on the additional information received, we continue to have concerns as to whether AMTAGVI TM meets the substantial clinical improvement criterion to be approved for new technology add-on payments. Specifically, it remains unclear if the use of AMTAGVI TM significantly improves clinical outcomes over existing technologies and whether AMTAGVI TM TIL immunotherapy offers a treatment option for a patient population unresponsive to, or ineligible for, currently available treatments for patients with advanced (unresectable or metastatic) melanoma who relapse on or do not tolerate current therapies. Although the applicant asserts that the clinically meaningful and durable responses following treatment with AMTAGVI TM address an unmet medical need in patients with advanced melanoma and high tumor burden, who are heavily pre-treated and difficult-to-treat and have a poor prognosis with no treatment options available after progression on immunotherapy and targeted agents or who are primary refractory to anti-PD-1/PD-Ll therapy, we remain concerned that the evidence provided by the applicant did not sufficiently address our concern regarding the lack of comparison to other standard of care therapies used in the treatment of metastatic melanoma, to allow us to assess whether AMTAGVI TM substantially improved outcomes compared to existing treatments for this heavily pre-treated and difficult-to-treat patient population. In addition, while the applicant stated that there are no treatment options available for this patient population, it appears there are a number of therapies that are FDA approved for unresectable or malignant melanoma that can be used in any line of therapy such as pembrolizumab, [ 136 ] ipilimumab, [ 137 ] and immunotherapy combinations such as nivolumab plus ipilimumab [ 138 ] and nivolumab-relatlimab. [ 139 ] Further, as these other treatments, as well as chemotherapy, are available therapies for patients with advanced melanoma, we remain unclear if there is a patient population that is eligible for AMTAGVI TM that would be ineligible for other currently available treatments. In addition, although the applicant presented results from a multicenter, randomized Phase 3 trial from the Netherlands and Denmark that evaluated treatment with a locally-produced TIL therapy versus ipilimumab for advanced melanoma, we note that the applicant did not address how the conditions of the study differed from that of the C-144-01 trial nor how the locally-produced TIL regimen used differed from AMTAGVI TM.
With respect to the applicant's assertion that CMS had previously approved new technology add-on payments for therapies approved under FDA's accelerated approval pathway for oncology and hematology uses, and the majority of these therapies were granted FDA accelerated approvals based on surrogate efficacy endpoints, including the same ORR and DOR endpoints used ( print page 69188) for accelerated approval of AMTAGVI TM , we note that, as previously stated, consistent with the discussion in the FY 2003 IPPS/LTCH PPS final rule ( 67 FR 50015 ), we do not rely on FDA criteria in our evaluation of substantial clinical improvement for purposes of determining what drugs, devices, or technologies qualify for new technology add-on payments under Medicare. This criterion does not depend on the standard of safety and efficacy on which FDA relies but on a demonstration of substantial clinical improvement in the Medicare population. Therefore, we do not believe that the FDA approvals for these other technologies relate to an assessment of substantial clinical improvement for AMTAGVI TM . We also note that we are unsure which technologies are being referenced by the applicant, and whether those technologies were determined to be a substantial clinical improvement because they improved the ORR, or whether the technologies demonstrated substantial clinical improvement under § 412.87(b)(1)(ii) through other claims that would not correlate with outcomes for AMTAGVI TM .
In addition, as described in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36075 through 36076 ), we continue to have concerns that the patient population evaluated in the C-144-01, Chesney, et al. (2022), and Sarnaik, et al. (2021) studies are not appropriately representative of the Medicare population and the disease burden seen in Medicare beneficiaries. While the applicant provided clarifying information about the inclusion and exclusion criteria for the C-144-01 study, the applicant did not provide additional information about the race, ethnicity, or other demographics of these individuals to demonstrate general applicability to the Medicare population. In addition, while the applicant stated that all patients had a high disease burden, it did not comment on whether the study population had a disease burden inclusive of the comorbidities generally found in the Medicare population. Thus, we remain concerned that the findings may not be generalizable to Medicare beneficiaries and their disease burden.
After consideration of all the information from the applicant, as well as the comments we received, we are unable to determine that AMTAGVI TM represents a substantial clinical improvement over existing technologies for the reasons discussed in the proposed rule and in this final rule, and therefore we are not approving new technology add-on payments for AMTAGVI TM for FY 2025.
Bluebird bio, Inc. submitted an application for new technology add-on payments for Lyfgenia TM (lovotibeglogene autotemcel) for FY 2025. According to the applicant, Lyfgenia TM is an autologous hematopoietic stem cell-based gene therapy indicated for the treatment of patients 12 years of age or older with sickle cell disease (SCD) and a history of vaso-occlusive events (VOE). Lyfgenia TM , administered as a single-dose intravenous infusion, consists of an autologous cluster of differentiation 34+ (CD34+) cell-enriched population from patients with SCD that contains hematopoietic stem cells (HSCs) transduced with BB305 lentiviral vector (LVV) encoding the β-globin gene (β A-T87Q -globin gene), suspended in a cryopreservation solution. The applicant explained that Lyfgenia TM is designed to add functional copies of a modified form of the β A-T87Q -globin gene into a patient's own HSCs, which allows their red blood cells to produce an anti-sickling adult hemoglobin (HbA T87Q ), to reduce or eliminate downstream complications of SCD.
Please refer to the online application posting for Lyfgenia TM , available at https://mearis.cms.gov/public/publications/ntap/NTP231013X3AK8 , for additional detail describing the technology and the disease treated by the technology.
With respect to the newness criterion, according to the applicant, Lyfgenia TM was granted BLA approval from FDA on December 8, 2023, for the treatment of patients 12 years of age or older with SCD and a history of VOEs. The applicant stated that it anticipated that Lyfgenia TM would have become available for sale on April 16, 2024, and that the first commercial claim for Lyfgenia TM would occur within approximately 130 days post-FDA approval to allow for the one-time activity to commercially qualify the contract manufacturer organization (CMO), followed by apheresis of the first patient at the qualified treatment center (QTC), where the personalized starting material will be shipped to the CMO for drug product manufacturing, release testing, and shipment of final product to the QTC for the one-time infusion. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36076 ), we stated that we were interested in additional information regarding the delay in the technology's market availability, as it appears that the technology would need to be available for sale prior to the enrollment of the first patient at the QTC. According to the applicant, Lyfgenia TM is provided in infusion bags containing 1.7 to 20 × 10 6 cells/mL (1.4 to 20 × 10 6 CD34+ cells/mL) in approximately 20 mL of solution and is supplied in one to four infusion bags. Per the applicant, the minimum dose is 3.0 × 10 6 CD34+ cells/kg patient weight.
According to the applicant, as of October 1, 2023, there are currently two ICD-10-PCS procedure codes to distinctly identify the intravenous administration of Lyfgenia TM : XW133H9 (Transfusion of lovotibeglogene autotemcel into central vein, percutaneous approach, new technology group 9) and XW143H9 (Transfusion of lovotibeglogene autotemcel into peripheral vein, percutaneous approach, new technology group 9). The applicant provided a list of diagnosis codes that may be used to currently identify the indication for Lyfgenia TM under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
With respect to the substantial similarity criteria, the applicant asserted that Lyfgenia TM is not substantially similar to other currently available technologies, because Lyfgenia TM has a distinct mechanism of action, which converts SCD at the genetic, cellular, and physiologic level to a non-sickling phenotype through the expression of the gene therapy-derived anti-sickling β A-T87Q -globin gene, and that therefore, the technology meets the newness criterion. Additionally, the applicant stated Lyfgenia TM is not substantially similar to other currently available therapeutic approaches indicated for SCD or to any drug therapy assigned to any MS-DRG in the 2022 MedPAR file data.
The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for Lyfgenia TM for the applicant's complete statements in support of its assertion that Lyfgenia TM is not substantially similar to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36077 through 36078 ), we noted that Lyfgenia TM may have the same or similar mechanism of action to Casgevy TM , for which we also received an application for new technology add-on payments for FY 2025. We stated that Lyfgenia TM and Casgevy TM are both gene therapies using modified autologous CD34+ hematopoietic stem and progenitor cell (HSPC) therapies administered via stem cell transplantation for the treatment of SCD. Both technologies are autologous, ex-vivo modified hematopoietic stem-cell biological products. As previously discussed, Casgevy TM was approved by FDA for this indication on December 8, 2023. For these technologies, patients are required to undergo CD34+ HSPC mobilization followed by apheresis to extract CD34+ HSPCs for manufacturing and then myeloablative conditioning using busulfan to deplete the patient's bone marrow in preparation for the technologies' modified stem cells to engraft to the bone marrow. Once engraftment occurs for both technologies, the patient's cells start to produce a different form of hemoglobin to reduce the amount of sickling hemoglobin. Further, we noted that both technologies appear to map to the same MS-DRGs, MS-DRG 016 (Autologous Bone Marrow Transplant with CC/MCC) ( print page 69190) and 017 (Autologous Bone Marrow Transplant without CC/MCC), and to treat the same or similar disease (SCD) in the same or similar patient population (patients 12 years of age and older who have a history of VOEs). Accordingly, as it appeared that Lyfgenia TM and Casgevy TM may use the same or similar mechanism of action to achieve a therapeutic outcome (that is, to reduce the amount of sickling hemoglobin to reduce and prevent VOEs associated with SCD), would be assigned to the same MS-DRG, and treat the same or similar patient population and disease, we stated we believed that these technologies may be substantially similar to each other such that they should be considered as a single application for purposes of new technology add-on payments. We noted that if we determined that this technology is substantially similar to Casgevy TM , we believed the newness period would begin on December 8, 2023, the date both Lyfgenia TM and Casgevy TM received FDA approval for SCD. We stated we were interested in information on how these two technologies may differ from each other with respect to the substantial similarity criteria and newness criterion, to inform our analysis of whether Lyfgenia TM and Casgevy TM are substantially similar to each other and therefore should be considered as a single application for purposes of new technology add-on payments.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36078 ), we invited public comment on whether Lyfgenia TM meets the newness criterion, including whether Lyfgenia TM is substantially similar to Casgevy TM and whether these technologies should be evaluated as a single technology for purposes of new technology add-on payments.
Comment: The applicant for Casgevy TM submitted a public comment regarding substantial similarity for Lyfgenia TM and Casgevy TM . The commenter asserted Casgevy TM represents the first therapy approved to use CRISPR/Cas9 gene editing technology and stated that no other approved technologies use this mechanism of action, and CRISPR/Cas9 technology has never previously been used in humans outside of clinical trials. The commenter stated that Casgevy TM is a one-time treatment that uses ex vivo non-viral CRISPR/Cas9 to precisely edit the erythroid-specific enhancer region of BCL11A in CD34+ HSPCs. The commenter stated that, while other non-gene therapy-based therapeutic approaches impact production of fetal hemoglobin (HbF), no other approved technology has been able to reactivate production of endogenous HbF to levels known to eliminate disease complications (for example, VOC), consistent with individuals with a clinically benign condition called hereditary persistence of fetal hemoglobin (HPFH) who experience no or minimal disease complications from SCD when they co-inherit both HPFH and SCD. The commenter stated that CMS focused on perceived similarities in treatment journey and categorical product characteristics between Casgevy TM and certain other technologies, but did not acknowledge material differences in the underlying technology which impact the safety and efficacy profile of these products. The commenter further explained that after Casgevy TM infusion, the edited CD34+ cells engraft in the bone marrow and differentiate to erythroid lineage cells with reduced BCL11A expression, and that this reduced BCL11A expression results in an increase in γ-globin expression and HbF protein production in erythroid cells. The commenter stated that in patients with severe SCD, HbF expression reduces intracellular hemoglobin S (HbS) concentration, preventing the red blood cells from sickling and addressing the underlying cause of disease, thereby eliminating VOCs. The commenter stated that, as such, Casgevy TM is not similar to the current standard of care (bone marrow transplant), nor to other technologies used in the treatment of SCD, and that none of these treatments use a mechanism of action that rely on CRISPR gene editing to reduce intracellular HbS concentration in SCD patients. The commenter explained how Lyfgenia TM uses a separate technology, gene replacement therapy, that utilizes a viral-based mechanism to introduce exogenous genetic material into patients' HSPCs, to add functional copies of a modified βA-globin gene into patients' HSCs through transduction of autologous CD34+ cells with B8305 lentiviral vector (LVV). The commenter stated that due to the LVV-based mechanism of action and the semi-random nature of viral integration, there is a potential risk of LVV-mediated insertional oncogenesis after treatment with Lyfgenia TM used in the treatment of SCD, as documented in FDA-approved labeling. The commenter stated that Casgevy TM , with its non-viral mechanism of action using CRISPR/Cas9 gene editing, does not employ a viral vector and does not insert a transgene; therefore, insertional oncogenesis cannot occur as a matter of scientific principle. The commenter further stated that Casgevy TM uses a unique underlying technology and manufacturing process and has distinct product characteristics that differentiate it from other technologies used to treat SCD. The commenter asserted in its comments that if CMS were to consider gene replacement therapy and gene editing technologies to be substantially similar, it could set a precedent based on overgeneralization which could deter further innovation.
The applicant also submitted a public comment regarding the newness criterion. With respect to mechanism of action, the applicant stated that Lyfgenia TM has a unique mechanism of action that differs from Casgevy TM 's because it is a one-time gene therapy that adds functional copies of the β A-T87Q -globin gene into a patient's own HSCs ex-vivo through the transduction of autologous CD34+ cells with a BB305 LVV to durably produce HbA T87Q . The applicant added that HbA T87Q is a modified adult hemoglobin (HbA) specifically designed to be anti-sickling while maintaining the same structure and function as naturally occurring HbA. According to the applicant, Lyfgenia TM consists of an autologous CD34+ cell-enriched population from patients with SCD that contains HSCs transduced with BB305 LVV encoding the β A-T87Q -globin gene, suspended in a cryopreservation solution. The applicant stated the BB305 LVV encodes a single amino acid variant of β-globin gene, β A-T87Q -globin: a human β-globin with a genetically engineered single amino acid change (threonine [Thr; T] to glutamine [Gin; Q] at position 87 (T87Q)). The applicant asserted HbA T87Q is nearly identical to wildtype (or “innate”) HbA, which is not prone to sickling. The applicant stated the T87Q substitution introduced in β A-T87Q -globin is designed to physically block or sterically inhibit polymerization of hemoglobin, thus rendering further “anti-sickling” properties to β A-T87Q -globin. According to the applicant, this results in a transgenic, non-immunogenic protein that can be measured in blood allowing for monitoring of the therapeutic protein in vivo and quantification relative to other globin species used to treat SCD. The applicant stated that Lyfgenia TM is not substantially similar to the CRISPR-Cas9 gene editing technique of Casgevy TM . The applicant also stated that, as described previously, Lyfgenia TM adds functional copies of a modified β-globin (HBB) gene, β A-T87Q globin gene, into patients' own HSCs to durably produce HbA T87Q , a modified adult HbA specifically designed to be ( print page 69191) anti-sickling while maintaining the same morphology and function as naturally occurring HbA. According to the applicant, the CRISPR/Cas9 gene editing technique mechanism of action described for Casgevy TM in the proposed rule differs substantially from Lyfgenia TM , as is evident by Casgevy TM 's unique editing approach in which GATA1 binding is irreversibly disrupted, and BCL11A expression is reduced, resulting in an increased production of HbF, and recapitulating a naturally occurring, clinically benign condition called HPFH that reduces or eliminates SCD symptoms.
According to the applicant, increasing HbA T87Q versus increasing HbF are fundamentally distinct mechanistic approaches. For individuals without SCD, HbF production is decreased shortly after birth, coinciding with an increase in HbA, and Lyfgenia TM is designed to replicate this natural state by introducing the production of HbA T87Q . The applicant stated HbA T87Q is nearly identical to HbA in several ways: sequence homology, protein structure, oxygen affinity and oxygen dissociation curves. The applicant stated that HbF has ~50 percent homology to HbA (two β globin chains are replaced with two γ-chains) and has a higher observed oxygen affinity and different oxygen unloading properties than HbA. According to the applicant, from a clinical perspective, current standard of care approaches (for example, the use of hydroxyurea) are available to increase levels of HbF with variable effectiveness, while the mechanism of action Lyfgenia TM affords is unique in increasing a modified HbA. The applicant commented that while both gene therapies are indicated for the treatment of SCD, the mechanistic approach of each is fundamentally and significantly different from the other, and therefore Lyfgenia TM and Casgevy TM are not substantially similar and should not be considered as a single application for the purposes of new technology add-on consideration.
The applicant also described potential risks associated with consideration of the two technologies as a single application. Specifically, the applicant commented that if Lyfgenia TM and Casgevy TM were treated as a single application and paid under a single maximum new technology add-on payment amount, this could potentially undermine CMS's aim to improve timely, meaningful access to SCD gene therapies for Medicare patients. Per the applicant, not only do the two therapies have distinct mechanisms of action but they also differ in the length of follow-up and the features of the population in which they were studied (for example, the commenter stated that the Lyfgenia TM clinical trials did not exclude patients with a history of chronic pain and included some patients with a history of stroke), and patients should have a choice to work with physicians to decide which therapy is most appropriate for them, based solely on their specific individual clinical circumstances. The applicant further asserted that given these differences, the finalization of a single new technology add-on payment amount for both therapies could hamper patient access to the most appropriate gene therapy for them, and potentially create a fiscally problematic and financial loss for IPPS hospitals, given the difference in the wholesale acquisition costs of both therapies, and CMS could potentially over-reimburse for one product, while under-reimbursing for the other through the use of the historical blended weighted average cost utilizing volume estimates. It is for these reasons, the applicant further stated, that Lyfgenia TM is not substantially similar to Casgevy TM , and therefore should not be considered as a single application with Casgevy TM for the purposes of new technology add-on payments.
The applicant also stated that provided that CMS finalize its policy to change the newness cutoff date from April 1 to October 1 to determine whether a new technology is within its 2- to 3-year newness period (as further described in section II.E.8 of the preamble of this final rule), the applicant would agree that it was reasonable to consider the start of Lyfgenia TM 's newness period to be on the date of FDA approval, December 8, 2023. However, the applicant requested that should CMS not finalize its proposal and the cutoff date remains April 1, that CMS should establish the beginning of the newness period on the date of Lyfgenia TM 's first commercial availability, as described in its new technology add-on payment application.
Response: We thank the applicant and the commenter for their comments. Based on our review of comments received and information submitted by the applicant as part of its FY 2025 new technology add-on payment application for Lyfgenia TM, we agree that Lyfgenia TM , which modifies a patients' own HSCs to increase HbA T87Q (modified adult hemoglobin), has a distinct mechanism of action compared to that of Casgevy TM , which uses a different mechanism of action of modifying a patients' HSPCs to increase expression of HbF to subsequently reduce the expression of intracellular sickled hemoglobin concentration. Therefore, we agree with the applicant that Lyfgenia TM utilizes a unique mechanism of action and is not substantially similar to existing treatment options and meets the newness criterion.
With regards to the market availability of Lyfgenia TM , as we have discussed in prior rulemaking ( 86 FR 45132 ; 77 FR 53348 ), generally, our policy is to begin the newness period on the date of FDA approval or clearance or, if later, the date of availability of the product on the U.S. market. Although the applicant stated in its application that Lyfgenia TM would become available for sale on April 16, 2024, we noted that we were interested in additional information regarding any delay in the technology's market availability, as it appears that the technology would need to be available for sale prior to the enrollment of the first patient at the QTC, and we did not receive additional information regarding the delay. Therefore, at this time, there is not sufficient information to determine a newness date based on a documented delay in the technology's availability on the U.S. market. Absent additional information, we consider the beginning of the newness period to commence on December 8, 2023, when Lyfgenia TM was granted BLA approval from FDA for the treatment of patients 12 years of age or older with SCD and a history of VOEs.
With respect to the cost criterion, the applicant provided multiple analyses to demonstrate that it meets the cost criterion. For each analysis, the applicant searched the FY 2022 MedPAR file using different ICD-10-CM codes to identify potential cases representing patients who may be eligible for Lyfgenia TM . Per the applicant, Lyfgenia TM is intended for patients who have not already undergone allogeneic bone marrow transplant or autologous bone marrow transplant. The applicant explained that it used different ICD-10-CM codes to demonstrate different cohorts of SCD patients that may be eligible for the technology.
According to the applicant, eligible cases for Lyfgenia [ TM ] will be mapped to either Pre-MDC MS-DRG 016 (Autologous Bone Marrow Transplant with CC/MCC) or 017 (Autologous Bone Marrow Transplant without CC/MCC). For each cohort, the applicant performed two sets of analyses using either the FY 2025 new technology add-on payments threshold for Pre-MDC MS-DRG 016 or Pre-MDC MS-DRG 017 for all identified cases. We noted that the FY 2025 new technology add-on payments thresholds for both Pre-MDC ( print page 69192) MS-DRG 016 and Pre-MDC MS-DRG 017 are $182,491. Each analysis followed the order of operations described in the table later in this section.
For the primary cohort, the applicant searched for an appropriate group of patients with any ICD-10-CM diagnosis code for SCD with crisis. Please see the online posting for Lyfgenia TM for the complete list of ICD-10-CM codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 12,357 claims mapping to 167 MS-DRGs, including MS-DRGs 811 and 812 (Red Blood Cell Disorders with MCC and without MCC, respectively) representing 76.0 percent of total identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $11,677,887, which exceeded the average case-weighted threshold amount of $182,491.
For the sensitivity 1 cohort, the applicant searched for a narrower cohort of patients with the admitting or primary ICD-10-CM diagnosis codes of Hemoglobin-SS (Hb-SS) SCD with crisis for the most common genotype of SCD. Please see the online posting for Lyfgenia TM for a complete list of ICD-10-CM codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 10,987 claims mapping to 160 MS-DRGs, including MS-DRGs 811 and 812 (Red Blood Cell Disorders with and without MCC, respectively) representing 75.1 percent of total identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $11,680,025, which exceeded the average case-weighted threshold amount of $182,491.
For the sensitivity 2 cohort, the applicant searched for a broader cohort of patients with the primary or secondary ICD-10-CM diagnosis codes for SCD with or without crisis. Please see the online posting for Lyfgenia TM for a complete list of ICD-10-CM codes provided by the applicant. The applicant used the inclusion/exclusion criteria described in the following table. Under this analysis, the applicant identified 17,120 claims mapping to 453 MS-DRGs, including MS-DRGs 811 and 812 (Red Blood Cell Disorders with and without MCC, respectively) representing 56.3 percent of total identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $11,681,718, which exceeded the average case-weighted threshold amount of $182,491.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant maintained that Lyfgenia TM meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36079 ), we invited public comments on whether Lyfgenia TM meets the cost criterion.
Comment: The applicant commented that the cost criterion for Lyfgenia TM was met for the primary cohort and two sensitivity cohorts of cases.
Response: We thank the applicant for its comments. We agree that the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount under all scenarios. Therefore, Lyfgenia TM meets the cost criterion.
With regard to the substantial clinical improvement criterion, the applicant asserted that Lyfgenia [ TM ] represents a substantial clinical improvement over existing technologies, because Lyfgenia [ TM ] is a one-time administration gene therapy that uniquely impacts the pathophysiology of SCD at the genetic level and offers the potential for stable, durable production of anti-sickling hemoglobin HbA [ T87Q ] , with approximately 85 percent of RBCs producing HbA [ T87Q ] , leading to complete resolution of severe VOEs in patients with SCD through 5.5 years of follow-up. The applicant asserted that for these reasons Lyfgenia [ TM ] is a much-needed treatment option for a patient population ineligible for allo-HSCT or without a matched related donor and significantly improves health-related quality of life. The applicant provided seven studies on Lyfgenia TM to support these claims, as well as 22 background articles about SCD and its current treatments. [ 141 ] The following table summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for Lyfgenia TM for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36081 ), after reviewing the information provided by the applicant, we stated we had the ( print page 69195) following concerns regarding whether Lyfgenia [ TM ] meets the substantial clinical improvement criterion. With respect to the claim that Lyfgenia [ TM ] presents an acceptable risk-benefit profile in terms of efficacy and safety for patients with SCD while allowing clinically meaningful improvements in HRQoL, the applicant stated the safety profile remains generally consistent with risk of autologous stem cell transplant, myeloablative conditioning, and underlying SCD. Additionally, the applicant mentioned that serious treatment-emergent adverse events (TEAEs) of grade 3 or higher TEAEs were reported, but no cases of veno-occlusive liver disease, graft failure, or vector-mediated replication competent lentivirus were reported. Per the applicant, three patients had adverse events attributed to Lyfgenia [ TM ] , including 2 events deemed possibly related and 1 event deemed definitely related, with all 3 resolving within 1 week of onset. We noted that the applicant submitted one published article about Group C results, an interim analysis by Kanter, et al. (2022) [ 142 ] in which Lyfgenia TM 's safety and efficacy were evaluated in a nonrandomized, open-label, single-dose phase 1-2 clinical trial (HGB-206) where 35 Group C patients had received Lyfgenia TM infusion. Group C was established after optimizing the treatment process in the initial cohorts, Groups A (7 patients) and B (2 patients). There was also a more stringent inclusion criterion for severe vaso-occlusive events before enrollment for Group C. The median follow-up was 17.3 months (range, 3.7-37.6) and 25 patients met both the inclusion criteria for vaso-occlusive events before enrollment and a minimum 6-month follow-up required for assessment of vaso-occlusive events. After receiving Lyfgenia TM , 12 patients (34 percent) had at least one serious adverse event; the most frequently reported were abdominal pain, drug withdrawal syndrome (opiate), nausea, and vomiting (6 percent each). The two events that were deemed to be possibly related to Lyfgenia TM were grade 2 leukopenia and grade 1 decreased diastolic blood pressure and the one event that was deemed to be definitely related was grade 2 febrile neutropenia. Although this evidence was provided to assert Lyfgenia TM improves clinical outcomes relative to previously available therapies, we noted that the risk-benefit profile and HRQoL for Lyfgenia TM was not compared to existing therapies. We stated we were interested in additional information regarding the risk-benefit profile of Lyfgenia TM compared to existing therapies, including clarification regarding an acceptable risk-benefit profile for patients with SCD and whether Lyfgenia TM fits this profile. We also questioned if the length of patient follow-up (median: 17.3 months, range: 3.7 to 37.6) would be sufficient to assess long-term safety outcomes.
Finally, with respect to the applicant's assertion that Lyfgenia TM improves clinical outcomes by halting SCD progression, presenting an acceptable risk-benefit profile with clinically meaningful improvement in HRQoL, and results in complete resolution of sVOEs, we noted that the applicant provided multiple sources of evidence that analyze the same phase 1-2 clinical study for Lyfgenia TM , HGB-206. We received an additional unpublished source [ 143 ] that provided some data on the phase 3 HGB-210 trial and combined this with data from HGB-206 with a total of 34 patients being evaluable for efficacy and 47 for safety. The median age of these 47 patients was 23 years. Due to the small study population and the median age of participants in the studies, we questioned if the safety and efficacy data from these studies would be generalizable to the Medicare population.
We invited public comments on whether Lyfgenia TM meets the substantial clinical improvement criterion.
Comment: The applicant submitted a public comment regarding the substantial clinical improvement criterion. In response to our concerns regarding the risk-benefit profile of Lyfgenia TM compared to existing therapies and whether the length of patient follow-up was sufficient to assess long-term safety outcomes, the applicant stated that within the efficacy and safety pools, among the 47 patients who received Lyfgenia TM, the median follow-up time was 35.5 months; overall exposure was 126.2 patient years. The applicant stated the longest patient follow up was 61.0 months (5.1 years). Per the applicant, efficacy was sustained across the duration of follow up (up to 61 months); 30 of 34 evaluable patients (88.2 percent; 95 percent CI, 72.5-96.7) achieved complete resolution of VOEs (VOE-CR), the primary endpoint (evaluated at 6-18 months post infusion). The key secondary endpoint, complete resolution of severe VOE (sVOE-CR), was achieved by 32 of 34 evaluable patients (94.1 percent; 95 percent CI, 80.3-99.3) in the same evaluation time period. The applicant reported that, at 36 months (N=20), clinically meaningful improvements occurred early and were sustained in pain intensity (57 percent), pain interference (64 percent), and fatigue (64 percent). The applicant further stated that the safety profile of Lyfgenia TM was consistent with underlying SCD and known effects of myeloablative conditioning, and there were no reports of graft failure or graft-versus-host disease. The applicant stated that SCD is associated with progressive and significant morbidity and mortality, with the burden of disease increasing with age. Per the applicant, current therapies do not target the underlying cause of disease and significant unmet need persists. The applicant also emphasized that while allo-HSCT is a potentially curative option, only a small percentage of patients are eligible for this treatment option due to lack of a matched donor and other reasons, such as age.
In response to our concern that the safety and efficacy data for Lyfgenia TM may not be generalizable to the Medicare population, the applicant explained that the overwhelming majority of Medicare beneficiaries with SCD were eligible because of disability (97.3 percent), not age. According to the applicant, Wilson-Frederick, et al. (2019) [ 144 ] found that 85.8 percent of the Medicare SCD population were non-elderly (ages 18-64), and 14.2 percent were ages 65-75 years, with ages 31- 45 years (36.4 percent) representing the largest Medicare-covered age category.
A few commenters commented in support of approving Lyfgenia TM . A commenter disagreed with CMS's concerns on substantial clinical improvement and discussed the barriers to access hematopoietic stem cell transplantation.
Response: We thank the applicant and other commenters for their comments regarding the substantial clinical improvement criterion. Based on a review of all the clinical studies and information submitted, we agree with the applicant that Lyfgenia TM represents ( print page 69196) a substantial clinical improvement over existing technologies because the technology offers a treatment option for certain patients with SCD who experience recurrent VOEs and who have not been able to achieve adequate control of the condition with existing treatments such as hydroxyurea and are ineligible for allo-HSCT due to the lack of a matched donor or other reasons (for example, age of the patients).
After consideration of the public comments received, and the information included in the applicant's new technology add-on payment application, we have determined that Lyfgenia TM meets the criteria for approval for new technology add-on payment. Therefore, we are approving new technology add-on payments for this technology for FY 2025. Cases involving the use of Lyfgenia TM that are eligible for new technology add-on payments will be identified by ICD-10-PCS codes: XW133H9 (Transfusion of lovotibeglogene autotemcel into central vein, percutaneous approach, new technology group 9) or XW143H9 (Transfusion of lovotibeglogene autotemcel into peripheral vein, percutaneous approach, new technology group 9).
In its application, the applicant estimated that the cost of Lyfgenia TM is $3,100,000 per patient. As discussed in section II.E.10. of the preamble of this final rule, we are revising the maximum new technology add-on payment percentage to 75 percent, for a medical product that is a gene therapy that is indicated and used specifically for the treatment of SCD and approved for new technology add-on payments for the treatment of SCD in the FY 2025 IPPS/LTCH PPS final rule. Accordingly, under § 412.88(a)(2) as revised in this final rule, we limit new technology add-on payments to the lesser of 75 percent of the average cost of the technology, or 75 percent of the costs in excess of the MS—DRG payment for the case. As a result, the maximum new technology add-on payment for a case involving the use of Lyfgenia TM for the treatment of SCD is $2,325,000 for FY 2025.
Omniscient Neurotechnology submitted an application for new technology add-on payments for Quicktome Software Suite for FY 2025. According to the applicant, Quicktome Software Suite is a cloud-based software that uses artificial intelligence (AI) tools and the scientific field of connectomics to analyze millions of data points derived from a patient's magnetic resonance imaging (MRI). Per the applicant, Quicktome Software Suite's proprietary Structural Connectivity Atlas (SCA) uses machine learning and tractographic techniques to create highly specific and personalized maps of a patient's brain or connectome from a standard MRI scan, regardless of brain shape, size, or physical distortion. The applicant asserted that the SCA is combined with a key refinement algorithm that identifies the location of parcels based on the specific structural characteristics of an individual's brain. The applicant asserted that Quicktome Software Suite uses resting-state functional MRI (rs-fMRI) to unveil the brain's network architecture or functional connectome by mapping blood oxygen level dependent (BOLD) signal correlations across brain parcels. Per the applicant, using data from a structural or a functional MRI (fMRI) scan, Quicktome Software Suite's proprietary AI allows clinicians to quickly and accurately assess the structural layout (that is, the locations and integrity) or the functional connectivity (that is, how different brain regions are working together) of a patient's brain.
Please refer to the online application posting for Quicktome Software Suite, available at https://mearis.cms.gov/public/publications/ntap/NTP23101722NQE , for additional detail describing the technology and the disease for which the technology is used.
With respect to the newness criterion, according to the applicant, Quicktome Software Suite received FDA 510(k) clearance on May 30, 2023. Per the FDA-cleared indication, Quicktome Software Suite is composed of a set of modules intended for the display of medical images and other healthcare data. It includes functions for image review, image manipulation, basic measurements, planning, three-dimensional (3D) visualization (multiplanar reconstructions (MPR) and 3D volume rendering), and the display of BOLD rs-MRI scan studies. The FDA clearance for Quicktome Software Suite was based on substantial equivalence to the legally marketed predicate device, StealthViz Advanced Planning Application with Stealth Diffusion Tensor Imaging (DTI) TM Package (hereafter referred to as StealthViz TM ), as both of these devices allow the import and export of Digital Imaging and Communications in Medicine (DICOM) images to a hospital picture archiving and communication system (PACS); contain a graphical user interface to conduct planning and visualization; display MRI anatomical images, as well as tractography constructed from Diffusion Weighted Images, in two-dimensional (2D) and 3D views; register tractography and an atlas to the underlying anatomical images; allow adding, removing, and editing of objects (including automatically segmented and manually defined regions of interest); and are delivered as software on an off-the-shelf hardware platform. [ 145 ] Prior to the FDA 510(k) clearance of Quicktome Software Suite in 2023, the technology, under the trade name Quicktome, received FDA 510(k) clearance on March 9, 2021, based on substantial equivalence to StealthViz TM . [ 146 ] StealthViz TM received FDA 510(k) clearance on May 16, 2008, for use in 2D and 3D surgical planning and image review and analysis. According to the FDA 510(k) summary for StealthViz TM , it enables digital diagnostic and functional imaging datasets, reviewing and analyzing the data in various 2D and 3D presentation formats, performing image fusion of datasets, segmenting structures in the images with manual and automatic tools and converting them into 3D objects for display, and exporting results to other Medtronic Navigation planning applications, to a PACS or to Medtronic Navigation surgical navigation systems such as StealthStation System. According to the applicant, Quicktome Software Suite was commercially available immediately after FDA clearance.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for Quicktome Software Suite and was granted approval to use the following procedure code effective October 1, 2024: 00K0XZ1 (Map brain using connectomic analysis, external approach). The applicant provided a list of diagnosis codes that it stated may currently be used to identify the indication for Quicktome Software Suite under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
With respect to the substantial similarity criteria, the applicant asserted ( print page 69197) that Quicktome Software Suite is not substantially similar to other currently available technologies because it is the first and only FDA-cleared platform to enable connectomic analysis at an individual level using machine learning and tractographic techniques to create personalized maps of the human brain. In addition, the applicant asserted that Quicktome Software Suite is the first cleared neurological planning tool to offer rs-fMRI capabilities. Per the applicant, Quicktome Software Suite eliminates the need for highly trained personnel, who may not be available at most institutions, and therefore, the technology meets the newness criterion. The applicant further asserted that current technologies that rely on task-based fMRI (tb-fMRI) can be problematic in brain tumor patients who may be cognitively impaired because they may be unable to perform required tasks. The following table summarizes the applicant's assertions regarding the substantial similarity criteria. Please see the online application posting for Quicktome Software Suite for the applicant's complete statements in support of its assertion that Quicktome Software Suite is not substantially similar to other currently available technologies.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36083 ), we noted the following concerns regarding whether Quicktome Software Suite meets the newness criterion. With respect to the applicant's claim that Quicktome Software Suite does not use the same or similar mechanism of action as existing technologies to achieve a therapeutic outcome, we noted that, according to the 510(k) application, it appears that Quicktome Software Suite is equivalent to StealthViz TM , its predicate device. We stated it was unclear how Quicktome Software Suite's mechanism of action, which enables patient-specific connectomic analysis for neurological planning, is different from that of StealthViz TM . We noted that StealthViz TM received FDA 510(k) clearance on May 16, 2008, for use in 2D/3D surgical planning and image review and analysis, and therefore is no longer considered new for purposes of new technology add-on payments. According to the applicant, Quicktome Software Suite is the first and only FDA-cleared platform to enable brain network mapping and analysis at an individual level and provides clinicians with information that was previously only available in a research setting. We noted that we were interested in further information to support that Quicktome Software Suite does not use the same or similar mechanism of action as StealthViz TM to achieve a therapeutic outcome, including information regarding capabilities of Quicktome Software Suite not found in StealthViz TM , and whether and how those capabilities are the result of a new mechanism of action.
In addition, we noted that there are several existing FDA-approved or cleared technologies (for example, StealthViz TM , Brainlab's Elements and iPlan products) that analyze fMRI and other medical imaging data to create 3D maps of a patient's brain, including white matter tracts. Furthermore, while the applicant asserted that Quicktome Software Suite is the only FDA-cleared device that uses a rs-fMRI, we questioned whether other FDA-cleared neurosurgical planning and visualization technologies integrate rs-fMRI, or if the analysis of rs-fMRI for neurosurgical planning is a mechanism of action unique to Quicktome Software Suite. We noted that we were interested in more information on the relevant current standard of care and technologies utilized for neurosurgical planning and how the mechanism of action of Quicktome Software Suite compares to the mechanism of action of existing technologies and connectomics software.
With respect to the third criterion, whether Quicktome Software Suite involves the treatment of the same or similar disease and patient population compared to existing technologies, we noted that according to the applicant, Quicktome Software Suite does not treat a new disease type or patient population but does provide new information for the treatment of existing patient populations. However, the provision of new information for the treatment of existing patient populations does not mean that the technology treats a new disease type or patient population, and ( print page 69198) therefore, we noted that it was unclear what the basis is for the applicant's statement that the third criterion is not met. We stated we were interested in additional information to support whether and how Quicktome Software Suite may involve the treatment of a different type of disease or patient population.
We stated that, as discussed in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44981 ), we also continued to be interested in public comments regarding issues related to determining newness for technologies that use AI, an algorithm, or software. Specifically, we stated that we were interested in public comment on how these technologies may be considered for the purpose of identifying a unique mechanism of action; how updates to AI, an algorithm, or software would affect an already approved technology or a competing technology; whether software changes for an already approved technology could be considered a new mechanism of action; and whether an improved algorithm by competing technologies would represent a unique mechanism of action if the outcome is the same as an already approved AI new technology.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36083 ), we invited public comments on whether Quicktome Software Suite is substantially similar to existing technologies and whether Quicktome Software Suite meets the newness criterion.
Comment: We received a few comments in support of new technology add-on payments for Quicktome Software Suite. The commenters stated that Quicktome Software Suite has a new mechanism of action because it distinguishes itself from existing technologies such as StealthViz TM by harnessing the power of AI, the structural connectivity atlas, and connectomics. The commenters further stated that unlike conventional methods, Quicktome Software Suite leverages AI algorithms to analyze complex structural and functional brain data, enabling the creation of comprehensive brain network maps that go beyond tractography. The commenters also stated that this approach, discussed in studies by Hendricks et al. (unpublished) [ 147 ] and Morell et al. (2022), [ 148 ] offers a more nuanced understanding of brain connectivity which includes higher order brain networks responsible for cognitive functions and emotion to which only Quicktome Software Suite can map. The commenters stated that existing technologies like StealthViz TM only go as far as tractography, which is able to map the white matter connections of the brain but does not delineate a patient's unique brain networks. The commenters stated that another of Quicktome Software Suite's hallmark features is its utilization of rs-fMRI for functional connectomic analysis, which is a mechanism of action that sets it apart from existing technologies. The commenters stated that studies such as the ones by Shimony et al. (2009), [ 149 ] Hacker et al. (2019), [ 150 ] and Lee et al. (2013) [ 151 ] have demonstrated the unique efficacy of rs-fMRI in delineating functional brain networks, enabling surgeons to tailor their approaches to minimize damage to critical neural circuits. The commenters stated that Quicktome Software Suite is also set apart from existing technology because Quicktome Software Suite offers fully automated post-processing of rs-fMRI, eliminating the need for specialized radiology personnel who are typically only available at the most advanced academic centers.
The commenters also stated, with regard to whether Quicktome Software Suite treats the same or similar type of disease and patient population as existing technologies that analyze fMRI and other medical imaging data for neurologic planning, such as StealthViz TM , that the unique processing of rs-fMRI underscores Quicktome Software Suite's potential to revolutionize neurosurgical planning and improve patient outcomes for all Medicare patients, not just the ones at the most elite academic institutions. Per the commenters, this is a critical consideration for Medicare patients who suffer from cognitive or motor impairments and cannot fully cooperate with task-based protocols (which are the only pre-surgical functional imaging paradigms currently available outside of Quicktome Software Suite).
A commenter stated that it is important to note that receiving clearance through a 510(k) should not be a definitive determination that a technology is substantially similar, particularly for one that has received FDA Breakthrough Device designation. The commenter further stated that over the last few years, CMS has approved a number of technologies for new technology add-on payments (thus having demonstrated newness) that received 510(k) clearance by demonstrating substantial equivalence to a previously approved or cleared technology.
Response: We appreciate the additional information from the commenters with respect to whether Quicktome Software Suite is substantially similar to existing technologies. We note that the studies presented by commenters (Shimony et al. (2009), [ 152 ] Hacker et al. (2019), [ 153 ] and Lee et al. (2013) [ 154 ] ) do not appear to discuss the specific mechanism of action of Quicktome Software Suite and how it represents a new mechanism of action compared to existing technologies, but rather more generally describe the potential uses of rs-fMRIs. We note that Quicktome was not mentioned in any of the three articles. We are unclear if the technology discussed in the articles was identical to Quicktome Software Suite, or rather, if it is an existing technology that would have a similar mechanism of action as Quicktome Software Suite. Absent additional information, we are unable to determine if Quicktome Software Suite's mechanism of action, which utilizes AI-based patient-specific analysis for neurological planning, is different from the mechanism(s) of action of existing technologies that analyze medical imaging data to create 3D maps of a patient's brain, including white matter tracts. While the commenters asserted Quicktome Software Suite distinguishes itself from existing technologies such as StealthViz TM by harnessing the power of AI, the structural connectivity atlas, and connectomics, it remains unclear specifically how this use of AI constitutes a unique mechanism of ( print page 69199) action when compared to non-AI technologies used in the same way for neurosurgical planning and visualization. We also disagree with commenters that the fully automated post-processing of rs-MRIs offered by Quicktome Software Suites represents a new mechanism of action, as it appears to describe an ease-of-use feature that may instead relate to an assessment of substantial clinical improvement. As a result, we believe that Quicktome Software Suite uses the same or similar mechanism of action as existing technologies like StealthViz TM.
However, with regard to whether a technology treats the same or similar type of disease and patient population, we agree with the commenters that Medicare patients who suffer from cognitive or motor impairments and cannot cooperate with task-based protocols would represent a patient population that could not utilize existing technologies for patient-specific connectomic analysis for neurological planning. Therefore, based on our review of the comments received, we agree that Quicktome Software Suite is not substantially similar to existing technologies and meets the newness criterion. We consider the beginning of the newness period to commence on May 30, 2023, when Quicktome Software Suite received FDA market authorization.
Comment: A few commenters responded to our request for comments regarding issues related to determining newness for technologies that use AI, an algorithm, or software. A commenter stated that FDA defines mechanism of action (referred to as mode of action) as “the means by which a product achieves its intended therapeutic effect or action.” [ 155 ] Per the commenter, in reviewing Quicktome Software Suite and similar technologies that involved the use of AI, it is important to note that the AI, algorithm, or software do not represent the mechanism of action per se. The commenter stated that AI, algorithm, or software plays an important role, such as analyzing images and creating brain mapping, but that piece alone is not sufficient to achieve the clinical effect. It stated that the AI, algorithm, or software is a component of the technology, not the entirety of the technology itself. The commenter stated that technologies that incorporate AI, an algorithm or software should be evaluated for newness in the same way as CMS evaluates any other medical device applying for a new technology add-on payment. The commenter stated that CMS should not take a broad policy position on the newness of these types of technologies, but should use its existing criteria and existing framework in evaluating these technologies individually on a case-by-case basis.
Another commenter recommended that CMS consider revisions to the regulations for new technology add-on payments under 42 CFR 412.87 to establish an alternative pathway for high-value AI technologies. The commenter suggested that newness should be determined by whether the technology enables a clinically valuable task for the Medicare patient population not previously achievable without the technology. The commenter continued by stating that “uniqueness” should be determined by whether the technology addresses a high-value clinical use case not previously addressed by other available technologies or medical procedures. The commenter also stated CMS's understanding of “value” of the Medicare population should be guided primarily by input from physician-experts and/or specialists in the related fields as well as product performance data.
Response: We thank the commenters for their input. We will continue to consider these comments as we gain more experience in this area and continue to welcome comments on determining newness and assessing mechanism of action for technologies that use AI, an algorithm or software.
With respect to the cost criterion, to identify potential cases representing patients who may be eligible for Quicktome Software Suite, the applicant searched 2020 Medicare Inpatient Hospitals—by Provider and Service data. [ 156 ] The applicant included all cases from the following MS-DRGs: 025 (Craniotomy and Endovascular Intracranial Procedures with MCC), 026 (Craniotomy and Endovascular Intracranial Procedures with CC), and 027 (Craniotomy and Endovascular Intracranial Procedures without CC/MCC). Using the inclusion/exclusion criteria described in the following table, the applicant identified 28,401 cases mapping to these three craniotomy MS-DRGs, with 64 percent of the identified cases mapping to MS-DRG 025. The applicant followed the order of operations described in the following table and calculated a final inflated average case-weighted standardized charge per case of $179,317, which exceeded the average case-weighted threshold amount of $134,802. Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount, the applicant asserted that Quicktome Software Suite meets the cost criterion.

We noted the following concerns regarding the cost criterion. We noted that the applicant limited its cost analysis to MS-DRGs 025, 026, and 027 because those three MS-DRGs represent brain tumor resection procedures, which are the first and most clearly established procedures for which the technology offers clinical utility. We stated that we were interested in information as to whether the technology would map to other MS-DRGs, such as 023 and 024 (Craniotomy with Major Device Implant or Acute Complex CNS PDX with MCC or Chemotherapy, or without MCC, respectively), or 054 and 055 (Nervous System Neoplasms with and without MCC, respectively), and if these MS-DRGs should also be included in the cost analysis. In addition, we questioned whether every case within MS-DRGs 025, 026, and 027 would be eligible for the technology and whether there would be any appropriate inclusion/exclusion criteria by ICD-10-CM/PCS codes within these MS-DRGs to identify potential cases representing patients who may be eligible for Quicktome Software Suite.
We invited public comments on whether Quicktome Software Suite meets the cost criterion.
We did not receive any comments on whether the Quicktome Software Suite cost analysis should include other MS-DRGs, such as 023 and 024 (Craniotomy with Major Device Implant or Acute Complex CNS PDX with MCC or Chemotherapy, or without MCC, respectively), or 054 and 055 (Nervous System Neoplasms with and without MCC, respectively), or if any additional inclusion/exclusion criteria should be applied to the MS-DRGs the applicant included in its cost analysis, specifically MS-DRGs 025, 026, and 027. As previously discussed, based on the information submitted by the applicant as part of its new technology add-on payment application, the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount for MS-DRGs 025, 026, and 027, which represent brain tumor resection procedures. Therefore, Quicktome Software Suite meets the cost criterion for use with brain tumor resection procedures mapping to MS-DRGs 025, 026, and 027.
With regard to the substantial clinical improvement criterion, the applicant asserted that Quicktome Software Suite represents a substantial clinical improvement over existing technologies because Quicktome Software Suite supports the visualization and brain mapping that improve clinical outcomes such as reducing the risk of an extended length of stay (LOS) and unplanned readmissions for craniotomy patients by reducing new postoperative neurological deficits that are caused by damage to brain networks or a patient's connectome. The applicant further asserted that Quicktome Software Suite is the first and only FDA-cleared platform to enable connectomic analysis at an individual level, enabling surgeons to visualize and avoid damaging these brain networks during surgery, thereby significantly improving clinical outcomes relative to services or technologies previously available. The applicant submitted three published studies and one unpublished study evaluating Quicktome Software Suite to support these claims, as well as four background articles about complications leading to unplanned readmissions after cranial surgery, factors associated with extended LOS in patients undergoing craniotomy for tumor resection, the association of incorporating fMRI in presurgical planning with mortality and morbidity in brain tumor patients, and the clinical importance of non-traditional, large-scale brain networks with respect to the potential adverse effects on patients when these networks ( print page 69201) are disrupted during surgery. [ 157 ] We noted in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36085 ) that one of the articles submitted as a study using the technology, the Dadario and Sughrue (2022) [ 158 ] study, should more appropriately be characterized as a background article because it does not directly assess the use of Quicktome Software Suite.
The following table summarizes the applicant's assertions regarding the substantial clinical improvement criterion. Please see the online posting for Quicktome Software Suite for the applicant's complete statements regarding the substantial clinical improvement criterion and the supporting evidence provided.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36085 through 36087 ), after our review of the information provided by the applicant, we stated that we had the following concerns regarding whether Quicktome Software Suite meets the substantial clinical improvement criterion. With respect to the applicant's claim that Quicktome Software Suite supports the visualization of brain networks and surgical planning to avoid damaging them during surgery, we stated we were concerned that the evidence does not appear to demonstrate that the Quicktome Software Suite's visualization and brain mapping techniques improve clinical outcomes relative to services or technologies already available by avoiding or reducing damage to the brain networks during surgery. For example, the Shah et al. (2023) [ 159 ] study describes the use of connectomics in planning and guiding an awake craniotomy for a tumor impinging on the language area in a 31-year-old bilingual woman. The authors stated that Quicktome Software Suite was used to generate preoperative connectome imaging for the patient, which helped in assessing the risk of functional deficits, guiding surgical planning, directing intraoperative mapping stimulation, and providing insights into postoperative function. The authors further described how preoperative imaging demonstrated proximity of the tumor to parcellations of the language area, and how ( print page 69202) intraoperative awake language mapping was performed, revealing speech arrest and paraphasic errors at areas of the tumor boundary correlating to functional regions that explained these findings. However, we noted that we were concerned that the report is based on a single case, and we questioned whether these findings would be generalizable to the broader Medicare population. In addition, we noted that the applicant did not provide evidence based on comparison of the use of Quicktome Software Suite technology with currently available cranial mapping software or tractography tools, and we noted that we would be interested in comparisons that assess the use of Quicktome Software Suite technology to improve these clinical outcomes relative to currently available technologies, such as StealthViz TM or Brainlab's Elements and iPlan products.
In addition, we questioned whether the findings related to Quicktome Software Suite's efficacy were generalizable to the Medicare population. Specifically, the Wu et al. (2023) [ 160 ] study examined the involvement of non-traditional brain networks in insulo-Sylvian gliomas and evaluated the potential of Quicktome Software Suite in optimizing surgical approaches to preserve cognitive function. The study included three parts. The first part involved a retrospective analysis of the location of insulo-Sylvian gliomas in 45 adult patients who underwent glioma surgery centered in the insular lobe. According to the research team, Quicktome Software Suite showed that 98 percent of the tumors involved a non-traditional eloquent brain network, which is associated with cognitive or neurological function. In part two, the research team prospectively collected neuropsychological data on seven patients to assess tumor-network involvement with change in cognition. Using Quicktome Software Suite, the research team found that all seven patients had a tumor involving a non-traditional eloquent brain network. Part three described how the research team used Quicktome Software Suite's network mapping capabilities to inform surgical decision-making and predict the preservation of cognitive function post-surgery for two prospective patients. We noted that while Quicktome Software Suite was used to assist surgical decision-making in two patients, as previously discussed, we questioned whether these limited findings would be generalizable to the broader Medicare population, and we stated that we would be interested in comparisons between Quicktome Software Suite and other currently available technologies to improve these clinical outcomes.
We also questioned whether the use of Quicktome Software Suite had a direct impact on significantly reducing neurological or cognitive deficits post-surgery. The applicant cited Morell et al. (2022), [ 161 ] a retrospective, single-center study of 100 patients who underwent surgery for brain tumor resection. The research team used Quicktome Software Suite to map and evaluate the integrity of nine large-scale brain networks in these patients. According to the research team, Quicktome Software Suite's analysis showed that for more than half of these patients, at least one of their brain networks were either affected during brain surgery or at risk of postsurgical deficits. Among those at risk of postsurgical deficits, their cortical regions or white matter fibers were either displaced by the mass effect of the tumor or damaged during surgery due to proximity to the tumor and/or planned transcortical trajectory. We noted that the primary focus of the study was to retrospectively map large-scale brain networks in brain tumor patients using Quicktome Software Suite platform, and therefore we stated that it did not appear to demonstrate that use of Quicktome Software Suite avoided damaging these networks during surgery.
Similarly, we noted that the applicant cited Hendricks et al. (n.d.), [ 162 ] which retrospectively analyzed the outcomes of 346 adult patients who underwent resection of superficial cerebral cavernous malformations from November 2008 through June 2021. We noted that the focus of the study was the use of Quicktome Software Suite to support the identification of areas of eloquent noneloquence, or cortex injured or transgressed that causes unexpected deficits. Therefore, we stated we remained interested in evidence that incorporating Quicktome Software Suite's analytics into surgical strategies and navigational tools during craniotomy surgery is associated with improved post-surgical outcomes.
With respect to the applicant's claim that damaging brain networks during surgery leads to neurologic complications, which are a leading contributor to increased length of stay (LOS), ICU admission, and readmissions, the applicant asserted that Quicktome Software Suite enables surgeons to visualize these brain networks and change their surgical approach as needed to avoid damages. We noted that the applicant submitted two documents in support of this claim, both of which are background documents rather than studies that evaluate clinical outcomes associated with the use of Quicktome Software Suite. In particular, the Elsamadicy et al. (2018) [ 163 ] study showed that altered mental status and sensory or motor deficits were the primary complications of craniotomies. The Philips et al. (2023) [ 164 ] study demonstrated that post-operative neurological deficits, caused by damage to brain networks or a patient's connectome were responsible for extended LOS. Although these studies supported the applicant's claim that damage to brain networks resulted in neurological complications, increasing LOS and inpatient service use, we noted that the evidence provided for this claim did not assess the use of Quicktome Software Suite to improve these clinical outcomes, nor did the evidence appear to demonstrate that use of the technology substantially improves these clinical outcomes relative to existing technologies, such as StealthViz TM or Brainlab's Elements and iPlan products. We stated that we would be interested in evidence demonstrating that utilization of Quicktome Software Suite improves clinical outcomes related to LOS, ICU admissions, and readmissions relative to existing technologies.
With respect to the applicant's claim that damaging brain networks during surgery has adverse effects for patients, including decreased quality of life and loss of function, the applicant asserted that Quicktome Software Suite enables surgeons to visualize brain networks ( print page 69203) and change their surgical approach as needed to avoid damaging these networks. The applicant further asserted that while other techniques have enabled the visualization of tractography or of parts of eloquent networks, this is not an adequate substitute for the ability to review the entirety of a patient's connectome (networks such as motor, language, and vision). Per the applicant, Quicktome Software Suite is the first of its kind to show the location and function of these networks and that damage to these networks is associated with poor outcomes. The applicant cited Vysotski et al. (2019), [ 165 ] who demonstrated that brain tumor patients who underwent a preoperative fMRI experienced significantly lower risks for mortality than those who did not. The applicant also cited Dadario and Sughrue (2022), [ 166 ] who discussed the clinical importance of preserving non-traditional brain networks for neurosurgical patients. Similar to our previous concern, we noted that the evidence provided for this claim did not assess the use of Quicktome Software Suite to improve quality of life and loss of function, nor did the evidence appear to demonstrate that use of the technology substantially improves these clinical outcomes relative to existing technologies. Therefore, we stated that we continued to question whether there was evidence to assess the effectiveness of Quicktome Software Suite to reduce damage to brain networks during surgery.
We stated in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36087 ) that we were also interested in public comments related to how we should evaluate issues related to determining substantial clinical improvement for technologies that use AI, an algorithm or software, including issues related to algorithm transparency, and how CMS should consider these issues in our assessment of substantial clinical improvement, as we continue to gain experience in this area. We noted that algorithm transparency refers to whether, and the extent to which, clinical users are able to access a consistent, baseline set of information about the algorithms they use to support their decision making and to assess such algorithms for fairness, appropriateness, validity, effectiveness, and safety. [ 167 ]
We invited public comments on whether Quicktome Software Suite meets the substantial clinical improvement criterion.
Comment: We received a few comments in support of new technology add-on payments for Quicktome Software Suite. The commenters stated that they believe Quicktome Software Suite provides a substantial clinical improvement over existing technologies. The commenters stated that out of the box, StealthViz TM does not allow a surgeon to visualize the patient's Default Mode Network (DMN) or the Dorsal Attention Network (DAN). Per the commenters, damage to the DMN can lead to memory loss and psychiatric disorders, and dysfunction of the DAN has been shown to be related to declines in cognitive abilities including attention and executive function. The commenters also stated if these higher order networks are damaged during surgery, deficits occur which can be just as debilitating to the patient as damage to the networks previously deemed eloquent—language, motor, and vision. In addition, the commenters stated that their own experiences support the assertion that Quicktome Software Suite's visualization and brain mapping techniques lead to tangible improvements in clinical outcomes by mitigating the risk of damage to vital brain networks during surgery. The commenters further stated that by providing surgeons with personalized insights into a patient's brain's structural and functional architecture, Quicktome Software Suite empowers them to navigate complex surgeries with greater confidence. A commenter additionally stated that it currently uses Quicktome Software Suite to improve outcomes from its brain tumor practice and would not go back to standard methodology. The commenters stated that they acknowledge the lack of randomized controlled trials evaluating the efficacy of Quicktome Software Suite technology, with a commenter stating that results from studies supporting the importance of preserving brain networks, such as the study by Hendricks, et al., can be conferred to Quicktome Software Suite, as it is the only technology that allows for the visualization of those brain networks. The commenters also wanted to point out the challenges associated with effectively studying a technology such as Quicktome Software Suite in large scale, randomized, long-term studies. The commenters further stated that aside from the difficulty getting patients to agree to be randomized to a control group (that is, not being treated using the latest tools and best information possible), it is challenging to distinguish the specific impact of tools from factors such as patient selection, case complexity, brain shift, and the myriad decisions made by a surgeon throughout a procedure. The commenters stated that in summary, the technology's integration of AI, the structural connectivity atlas, and connectomics, coupled with its unique utilization of rs-fMRI, positions it as a groundbreaking tool for improving patient outcomes and enhancing surgical precision.
Response: We thank the commenters for their input. After further review, we continue to have concerns as to whether Quicktome Software Suite meets the substantial clinical improvement criterion as noted in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36085 through 36087 ). Specifically, we continue to question whether Quicktome Software Suite's visualization and brain mapping techniques improve clinical outcomes by avoiding or reducing damage to the brain networks during surgery, thereby reducing neurological or cognitive deficits post-surgery, as compared to services or technologies already available. We do not have information about the difference in outcomes, such as reduction in neurological complications, LOS, or inpatient service use, and other clinical outcomes, such as quality of life and loss of function, when Quicktome Software Suite versus similar technologies, such as StealthViz TM , earlier versions of Quicktome Software Suite, or other currently available cranial mapping software or tractography tools, are used. We further note that while the commenters have stated they have noted improved clinical outcomes with use of the technology, they did not describe the improved outcomes or provide evidence for CMS to evaluate regarding these improvements. In addition, we continue to question whether the findings related to the efficacy of Quicktome Software Suite are generalizable to the Medicare population due to the very limited number of patients in which Quicktome Software Suite was used to assist in surgical decision-making, as previously stated. Further, while we appreciate the challenges in designing trials that effectively study technologies such as ( print page 69204) Quicktome Software Suite, as noted by the commenters, we note that without evidence to support a demonstration of improved clinical outcomes as compared to existing technologies, we are unable to make a determination regarding substantial clinical improvement.
We did not receive comments relating to how we should evaluate issues related to determining substantial clinical improvement for technologies that use AI, an algorithm or software, including issues related to algorithm transparency, and how CMS should consider these issues in our assessment of substantial clinical improvement. We will continue to consider these questions as we gain more experience, and we continue to welcome comments in this area.
After consideration of all the information submitted by the applicant as well as the comments we received, we are unable to determine that Quicktome Software Suite meets the substantial clinical improvement criterion for the reasons discussed in the proposed rule and in this final rule, and therefore, we are not approving new technology add-on payments for Quicktome Software Suite for FY 2025.
As discussed previously, beginning with applications for FY 2021, a medical device designated under FDA's Breakthrough Devices Program that has received marketing authorization as a Breakthrough Device, for the indication covered by the Breakthrough Device designation, may qualify for the new technology add-on payment under an alternative pathway. Additionally, beginning with FY 2021, a medical product that is designated by the FDA as a Qualified Infectious Disease Product (QIDP) and has received marketing authorization for the indication covered by the QIDP designation, and, beginning with FY 2022, a medical product that is a new medical product approved under FDA's Limited Population Pathway for Antibacterial and Antifungal Drugs (LPAD) and used for the indication approved under the LPAD pathway, may also qualify for the new technology add-on payment under an alternative pathway. Under an alternative pathway, a technology will be considered not substantially similar to an existing technology for purposes of the new technology add-on payment under the IPPS and will not need to meet the requirement that it represents an advance that substantially improves, relative to technologies previously available, the diagnosis or treatment of Medicare beneficiaries. These technologies must still be within the 2-to-3-year newness period to be considered “new,” and must also still meet the cost criterion.
As discussed previously, in the FY 2023 IPPS/LTCH PPS final rule, we finalized our proposal to publicly post online applications for new technology add-on payment beginning with FY 2024 applications ( 87 FR 48986 through 48990 ). As noted in the FY 2023 IPPS/LTCH PPS final rule, we are continuing to summarize each application in this final rule. However, while we are continuing to provide discussion of the concerns or issues, we identified with respect to applications submitted under the alternative pathway, we are providing more succinct information as part of the summaries in the proposed and final rules regarding the applicant's assertions as to how the medical service or technology meets the applicable new technology add-on payment criteria. We refer readers to https://mearis.cms.gov/public/publications/ntap for the publicly posted FY 2025 new technology add-on payment applications and supporting information (with the exception of certain cost and volume information, and information or materials identified by the applicant as confidential or copyrighted), including tables listing the ICD-10-CM codes, ICD-10-PCS codes, and/or MS-DRGs related to the analyses of the cost criterion for certain technologies for the FY 2025 new technology add-on payment applications.
We received 23 applications for new technology add-on payments for FY 2025 under the new technology add-on payment alternative pathway. As discussed previously, in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ), we finalized that beginning with the new technology add-on payment applications for FY 2025, for technologies that are not already FDA market authorized for the indication that is the subject of the new technology add-on payment application, applicants must have a complete and active FDA market authorization request at the time of new technology add-on payment application submission and must provide documentation of FDA acceptance or filing to CMS at the time of application submission, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. See § 412.87(e) and further discussion in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ). Of the 23 applications received under the alternative pathway, seven applications were not eligible for consideration for new technology add-on payment because they did not meet these requirements; and two applicants withdrew their applications prior to the issuance of the proposed rule, including the withdrawal of the application for DefenCath TM (taurolidine/heparin), which received conditional approval for new technology add-on payments for FY 2024, subsequently received FDA approval in November 2023, and therefore was eligible to receive new technology add-on payments beginning with discharges on or after January 1, 2024. As discussed in section II.E.4. of this final rule, we proposed and are finalizing to continue making new technology add-on payments for DefenCath® (taurolidine/heparin) for FY 2025. Subsequently, prior to the issuance of this final rule, three additional applicants withdrew their respective applications for restor3d TIDAL TM Fusion Cage, Transdermal GFR Measurement System utilizing Lumitrace, and cefepime-taniborbactam. For the remaining 11 applications, we are approving 12 new technology add-on payments for FY 2025 (including ZEVTERA TM (ceftobiprole medocaril) for which the applicant submitted a single application for multiple indications, and for which we are approving two separate new technology add-on payments). A discussion of these 11 applications is presented in this final rule, including 10 technologies that have received a Breakthrough Device designation from FDA and 1 that was designated as a QIDP by FDA. We did not receive any applications for technologies approved through the LPAD pathway.
In accordance with the regulations under § 412.87(f)(2), applicants for new technology add-on payments for FY 2025 for Breakthrough Devices must have FDA marketing authorization by May 1 of the year prior to the beginning of the fiscal year for which the application is being considered. Under § 412.87(f)(3), applicants for new technology add-on payments for FY 2025 for QIDPs and technologies approved under the LPAD pathway must have FDA marketing authorization by July 1 of the year prior to the beginning of the fiscal year for which the application is being considered. The policy finalized in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58742 ) provides for conditional approval for a technology for which an application is submitted under the alternative pathway for certain antimicrobial products (QIDPs and LPADs) at § 412.87(d) that does not receive FDA ( print page 69205) marketing authorization by July 1 prior to the particular fiscal year for which the applicant applied for new technology add-on payments, provided that the technology receives FDA marketing authorization before July 1 of the fiscal year for which the applicant applied for new technology add-on payments. We refer the reader to the FY 2021 IPPS/LTCH final rule for a complete discussion of this policy ( 85 FR 58737 through 58742 ).
As we did in the FY 2024 IPPS/LTCH PPS proposed rule, for applications under the alternative new technology add-on payment pathway, in the FY 2025 IPPS/LTCH PPS proposed rule we proposed to approve or disapprove each of these 11 applications for FY 2025 new technology add-on payments. Therefore, in this section of the preamble of this final rule, we provide background information on each of the remaining alternative pathway applications and our determination on whether or not each technology is eligible for the new technology add-on payment for FY 2025. We are not including in this final rule the description and discussion of applications that were withdrawn or that are ineligible for consideration for FY 2025.
We refer readers to section II.H.8. of the preamble of the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42292 through 42297 ) and section II.F.6 of preamble of the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58715 through 58733 ) for further discussion of the alternative new technology add-on payment pathways for these technologies.
Annalise-Ai Pty Ltd submitted an application for new technology add-on payments for the Annalise Enterprise CTB Triage—OH for FY 2025. According to the applicant, the Annalise Enterprise CTB Triage—OH is a medical device software application used to aid in the triage and prioritization of studies with features suggestive of obstructive hydrocephalus (OH). Per the applicant, the device analyzes studies using an artificial intelligence (AI) algorithm to identify suspected OH findings in non-contrast computed tomography (NCCT) brain scans and makes study-level output available to an order and imaging management system for worklist prioritization or triage.
Please refer to the online application posting for the Annalise Enterprise CTB Triage—OH available at https://mearis.cms.gov/public/publications/ntap/NTP231017D5AA7 , for additional detail describing the technology and how it is used.
According to the applicant, the Annalise Enterprise CTB Triage—OH received Breakthrough Device designation from FDA on February 17, 2023, for use in the medical care environment to aid in triage and prioritization of studies with features suggestive of OH. The device analyzes studies using an AI algorithm to identify findings. It makes study-level output available to an order and imaging management system for worklist prioritization or triage. The applicant stated that the technology received 510(k) clearance from FDA on August 15, 2023, for the same indication consistent with the Breakthrough Device designation. Per the applicant, the Annalise Enterprise CTB Triage—OH was not immediately available for sale because there were additional steps to be completed following 510(k) clearance prior to the product becoming commercially available. According to the applicant, these additional steps involved generating a new unique device identifier (UDI) to incorporate the recently cleared finding for OH, integrating this UDI into the device, and releasing it. Per the applicant, the Annalise Enterprise CTB Triage—OH became commercially available on October 10, 2023.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for the Annalise Enterprise CTB Triage—OH beginning in FY 2025 and was granted approval for the following procedure code effective October 1, 2024: XXE0X1A (Measurement of intracranial cerebrospinal fluid flow, computer-aided triage and notification, new technology group 10). The applicant provided a list of diagnosis codes that may be used to currently identify the indication for the Annalise Enterprise CTB Triage—OH under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
With respect to the cost criterion, the applicant provided three analyses to demonstrate that the technology meets the cost criterion. The applicant stated that for all three analyses, it used the 2021 Standard Analytic Files (SAF) Limited Data Set (LDS) to identify the top admitting diagnosis codes for inpatient stays that were admitted from the emergency room (ER) and included a non-contrast CT head scan. Next, it searched the FY 2022 MedPAR data to identify applicable inpatient stays based on different sets of admitting diagnosis codes for each of the three analyses. The applicant explained that it used admitting diagnosis codes from the inpatient stays, rather than discharge diagnosis codes, because the Annalise Enterprise CTB Triage—OH is an AI-based technology used to identify and prioritize patients suspected of OH. As a result, it will commonly be used in the ER before the doctor and/or the hospital has assigned the primary or secondary diagnosis for the inpatient stay. The applicant stated that admitting diagnosis codes may be better predictors for whether the Annalise Enterprise CTB Triage—OH service will be used, rather than primary or secondary diagnosis at discharge, which will likely represent information known after the procedure is performed. Per the applicant, for identifying the top admitting diagnosis codes, the inpatient stays were further narrowed down to only those where the patient had a physician claim during the inpatient stay or one day before for a non-contrast CT head scan (defined as CPT codes 70450, 70480, 70486), or had an outpatient claim for a non-contrast CT head scan the day of admission or one day before. Each analysis followed the order of operations described in the table that follows later in this section.
For the primary analysis, the applicant stated that it searched the FY 2022 MedPAR file for cases with emergency room charges (that is, emergency room charge amount greater than $0) and/or an inpatient admission type code (IP_ADMSN_TYPE_CD) equal to 1 for emergency, and reporting one of the top 25 diagnosis codes associated with 50 percent of all identified inpatient stays in the 2021 SAF. According to the applicant, it identified 2,206,036 claims mapping to 714 MS-DRGs, including MS-DRG 871 (Septicemia or Severe Sepsis without MV >96 Hours with MCC), which represented 16 percent of identified cases. The applicant stated that it calculated a final inflated average case-weighted standardized charge per case of $80,407, which exceeded the average case-weighted threshold amount of $69,892.
For the second analysis, the applicant stated that it conducted a sensitivity analysis using cases with emergency room charges (that is, emergency room charge amount greater than $0) and/or an inpatient admission type code (IP_ADMSN_TYPE_CD) equal to 1 for emergency, and reporting one of the top 186 admitting diagnosis codes associated with 80 percent of all identified inpatient stays in the 2021 SAF LDS. The applicant noted that it identified 3,991,354 claims mapping to 739 MS-DRGs, including MS-DRG 871 ( print page 69206) (Septicemia or Severe Sepsis without MV >96 Hours with MCC), which represented 11 percent of identified cases. The applicant noted that it calculated a final inflated average case-weighted standardized charge per case of $78,356, which exceeded the average case-weighted threshold amount of $68,660.
For the third analysis, the applicant stated that it conducted a sensitivity analysis that identified cases using the same criteria as the primary analysis, and further limited it to cases that also incurred CT charges. Per the applicant, it performed this sensitivity analysis because although doctors are likely to order the Annalise AI technology when a NCCT head scan is performed and the patient is admitted through the emergency room, the MedPAR file variable for CT charges does not differentiate between contrast and NCCTs, or the area of the body where the CT is performed, and does not capture CT charges billed by physicians during the inpatient stay. As a result, it further limited the cases to those with charges for CT to assess if this would impact whether the technology would meet the cost criterion. Per the applicant, it identified 1,546,504 claims mapping to 702 MS-DRGs, including MS-DRG 871 (Septicemia or Severe Sepsis without MV >96 Hours with MCC), which represented 17 percent of identified cases. The applicant stated that it calculated a final inflated average case-weighted standardized charge per case of $89,176, which exceeded the average case-weighted threshold amount of $71,344.
The applicant asserted that because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the Annalise Enterprise CTB Triage—OH meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36108 ), we noted the following concern regarding the cost criterion. According to the applicant, the technology is used to aid in the triage and prioritization of studies with features suggestive of OH. However, the diagnosis codes that the applicant used to identify eligible cases included non-neurologic diagnosis codes (for example, U071, R0602, J189). We questioned whether these diagnosis codes were applicable, and whether using neurologic diagnosis codes for diagnoses that exhibit symptoms similar ( print page 69207) to OH would more accurately identify eligible cases.
Subject to the applicant adequately addressing this concern, we agreed with the applicant that the technology meets the cost criterion and proposed to approve the Annalise Enterprise CTB Triage—OH for new technology add-on payments for FY 2025.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the total cost of the Annalise Enterprise CTB Triage—OH to the hospital to be $371.37 per patient. According to the applicant, hospitals acquire the Annalise Enterprise CTB Triage—OH system on a subscription-based model, with an annual cost of $180,000 per hospital. The applicant stated that the average cost per patient per hospital will vary by the volume of the NCCT cases for which the software is used. To determine the cost per case, the applicant used the following methodology:
First, the applicant conducted market research to estimate the percent of NCCT cases where this software would likely be ordered, which was estimated at 50 percent of NCCT head scans for older patients (>65 years of age) and 30 percent of NCCT head scans for younger patients (<65 years of age).
Second, the applicant used the 2021 SAF LDS to identify total NCCT scans by hospital. To represent the full Medicare fee-for-service population, the applicant multiplied total NCCT head scans at each hospital from the data by 20.
Third, to calculate the total number of NCCT head scans for each hospital, the applicant assumed that 56.5 percent of all NCCT scans are for Medicare beneficiaries, based on literature on trends in the utilization of head CT scans in the United States. [ 169 ]
Fourth, to calculate the cost per case for each hospital, the applicant divided $180,000 by the estimated number of NCCT head scans analyzed by the technology for each hospital. Per the applicant, the average cost per case across all IPPS hospitals was then calculated at $371.37.
The applicant asserted that calculating the cost per case across all IPPS hospitals was reasonable. The applicant noted that given its limited time on the market and low number of subscribers, it used all IPPS hospitals to calculate cost per case rather than limiting the analysis to current subscribers. The applicant mentioned that for technologies that are commercially available for a longer period of time and with more subscribers, it may make sense to limit the cost per case analysis to hospitals that are current subscribers rather than using all IPPS hospitals in the calculation.
As we noted in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58630 ) and in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 44983 ), we understand that there are unique circumstances with respect to determining a cost per case for a technology that utilizes a subscription for its cost and we will continue to consider the issues relating to calculation of the cost per unit of technologies sold on a subscription basis as we gain more experience in this area. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36109 ), we stated that we continued to welcome comments from the public as to the appropriate method to determine a cost per case for such technologies, including comments on whether the cost analysis should be updated based on the most recent subscriber data for each year for which the technology may be eligible for add-on payment.
We noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of the Annalise Enterprise CTB Triage—OH would be $241.39 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether the Annalise Enterprise CTB Triage—OH meets the cost criterion and our proposal to approve new technology add-on payments for the Annalise Enterprise CTB Triage—OH for FY 2025 for use in the medical care environment to aid in triage and prioritization of studies with features suggestive of OH.
Comment: The applicant submitted a public comment in response to our concern regarding the use of non-neurologic diagnosis codes to identify eligible cases of OH. The applicant stated that it intentionally included cases with non-neurological diagnosis codes to reflect patients who may have received the test based on the presenting symptoms in the Emergency Department because only a subset of those patients have an admitting diagnosis of OH or other neurological condition. The applicant explained that removing the inpatient stays with a non-neurological admitting diagnosis would undercount the inpatient stays and underestimate potential volume. However, in response to the request from CMS, the applicant stated that it conducted an additional sensitivity analysis by removing the non-neurological diagnoses (for example, A41.9, R53.1, N39.9, N17.9, U07.1, R06.02, J18.9, E87.1, R07.9, R50.9, I21.4, J96.01, E86.0, I46.9) from the list of top 25 admitting diagnoses and re-ran analyses 1 and 3. The applicant stated that the remaining 11 admitting diagnoses mapped to 651 and 640 MS-DRGs respectively, with the top 10 MS-DRGs representing about 43 percent of the total volume in both analyses. The applicant asserted that using the same methodology for the previously run analyses, it determined the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, and the Annalise Enterprise CTB Triage—OH meets the cost criterion.
Response: We thank the applicant for its comment. We agree that the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount. Therefore, Annalise Enterprise CTB Triage—OH meets the cost criterion.
Comment: The applicant submitted a comment in response to the discussion in the proposed rule on the appropriate method to determine a cost per case for the technologies sold on a subscription basis. The applicant stated that calculating the cost per case across all IPPS hospitals was reasonable since there were not enough subscribers for Annalise Enterprise CTB Triage—OH at the time of the cost analysis. The applicant stated that Annalise Enterprise CTB Triage—OH had only been commercially available for less than 30 days prior to the new technology add-on payment application submission deadline.
Response: We thank the applicant for its comment. We agree with the applicant's rationale in calculating the cost of the technology given the limited time that the technology has been on the market and small number of subscribers. We will continue to consider the issues relating to calculation of the cost per unit of technologies sold on a subscription basis as we gain more experience in this area. We also continue to welcome comments from the public as to the appropriate method to determine a cost per case for such technologies, including comments on ( print page 69208) whether the cost analysis should be updated based on the most recent subscriber data for each year for which the technology may be eligible for add-on payment.
Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the Annalise Enterprise CTB Triage—OH meets the cost criterion. The technology received 510(k) clearance on August 15, 2023 as a Breakthrough Device, with an indication for use in the medical care environment to aid in triage and prioritization of studies with features suggestive of OH, which is covered by its Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for the Annalise Enterprise CTB Triage—OH for FY 2025. We consider the beginning of the newness period to commence on October 10, 2023, the date on which the technology became commercially available for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the Annalise Enterprise CTB Triage—OH is $371.37. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the Annalise Enterprise CTB Triage—OH is $241.39 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of the Annalise Enterprise CTB Triage—OH that are eligible for new technology add-on payments will be identified by ICD-10-PCS procedure code: XXE0X1A (Measurement of intracranial cerebrospinal fluid flow, computer-aided triage and notification, new technology group 10).
Q-linea submitted an application for new technology add-on payments for the ASTar ® System for FY 2025. According to the applicant, the ASTar ® System is a fully automated system for rapid antimicrobial susceptibility testing (AST). The applicant stated that the proprietary AST technology is based on broth microdilution (BMD), optimized for high sensitivity and short time-to-result, delivering phenotypic AST with true minimum inhibitory concentration (MIC) results in approximately six hours.
Please refer to the online application posting for the ASTar ® System, available at https://mearis.cms.gov/public/publications/ntap/NTP231013T7Y5F , for additional detail describing the technology and how it is used.
According to the applicant, the ASTar ® System consists of the ASTar ® Instrument and the ASTar ® BC G-Kit. According to the applicant, the ASTar ® Instrument and ASTar ® BC G-Kit, which includes the ASTar ® BC G-Consumable Kit and the ASTar BC G-Frozen Insert, received Breakthrough Device designation from FDA on April 7, 2022. The ASTar ® BC G-Kit is a multiplexed, in vitro, diagnostic test utilizing AST methods and is intended for use with the ASTar ® Instrument. The ASTar ® BC G-Kit is performed directly on positive blood cultures confirmed positive for Gram-negative bacilli only by Gram stain, and tests antimicrobial agents with nonfastidious and fastidious bacterial species. The technology received FDA 510(k) clearance on April 26, 2024 with the following indication for use: the ASTar ® System, comprised of the ASTar ® Instrument with the ASTar ® BC G-Kit (ASTar ® BC G-Consumable kit, ASTar ® BC G-Frozen insert, and ASTar ® BC G-Kit software), utilizes high-speed, time-lapse microscopy imaging of bacteria for the in vitro, quantitative determination of antimicrobial susceptibility of on-panel gram-negative bacteria. The test is performed directly on positive blood culture samples signaled as positive by a continuous monitoring blood culture system and confirmed to contain gram-negative bacilli by Gram stain. Since the indication for which the technology received FDA 510(k) clearance is included within the scope of the Breakthrough Device designation, we believe that the FDA 510(k) indication is appropriate for consideration for new technology add-on payment under the alternative pathway criteria. The applicant stated that it anticipates the technology will be available on the market immediately after 510(k) clearance from FDA.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for the ASTar® System beginning in FY 2025 and was granted approval for the following procedure code effective October 1, 2024: XXE5X2A (Measurement of infection, phenotypic fully automated rapid susceptibility technology with controlled inoculum, new technology group 10). The applicant provided a list of diagnosis codes that may be used to currently identify the indication for the ASTar® System under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
With respect to the cost criterion, the applicant provided multiple analyses to demonstrate that it meets the cost criterion. Each analysis used different ICD-10-CM codes to identify potential cases in the FY 2022 MedPAR file representing patients who may be eligible for the ASTar® System. According to the applicant, Cohort 1 comprised patients with non-sepsis infections and Cohort 2 consisted of patients with sepsis resulting from bacteria identifiable by the ASTar® System. The applicant explained that these scenarios were separated as the applicant believed that charges and MS-DRG assignments may differ due to the resources required to treat sepsis patients compared to those required for less severe infections. Finally, Cohort 3 included all ICD-10-CM codes from Cohorts 1 and 2 because the applicant stated that the ASTar® System may be used to identify any infection caused by the bacteria listed in Cohorts 1 and 2. The applicant stated that in all three cohorts, the patients mapped to a large number of MS-DRGs based on the listed ICD-10-CM codes. Therefore, in the analyses, the applicant only included the most common MS-DRGs, that is, the MS-DRGs containing at least 1 percent of the potential case volume within each of the three cohorts, as these are the MS-DRGs to which potential ASTar® System cases would most closely map. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section to identify claims for each cohort. Each analysis followed the order of operations described in the table that follows later in this section.
For Cohort 1, the applicant identified 440,838 claims mapping to 14 MS-DRGs, including MS-DRG 871 (Septicemia or Severe Sepsis with MV >96 Hours with MCC) representing 25 percent of identified cases, and calculated a final inflated average case-weighted standardized charge per case of $85,525, which exceeded the average case-weighted threshold amount of $70,398.
For Cohort 2, the applicant identified 224,825 claims mapping to 7 MS-DRGs, including MS-DRG 871 (Septicemia or Severe Sepsis with MV >96 Hours with MCC) representing 54 percent of identified cases, and calculated a final inflated average case-weighted standardized charge per case of $99,508, which exceeded the average case-weighted threshold amount of $82,171. ( print page 69209)
For Cohort 3, the applicant identified 603,877 claims mapping to 13 MS-DRGs, including MS-DRG 871 (Septicemia or Severe Sepsis with MV >96 Hours with MCC) representing 34 percent of identified cases, and calculated a final inflated average case-weighted standardized charge per case of $88,395 which exceeded the average case-weighted threshold amount of $73,727.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all the three cohorts, the applicant asserted that the ASTar® System meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36110 ), we agreed with the applicant that the ASTar® System meets the cost criterion and therefore proposed to approve the ASTar® System for new technology add-on payments for FY 2025, subject to the technology receiving FDA marketing authorization as a Breakthrough Device for the indication corresponding to the Breakthrough Device designation by May 1, 2024.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the operating cost of the ASTar® System to the hospital to be $150 per patient, based on the operating component ASTar® BC G-Kit (composed of the ASTar® BC G-Consumable Kit ($141) and ASTar BC G-Frozen Insert ($9)). The applicant also noted a capital cost of $200,000 for the ASTar® Instrument. Because section 1886(d)(5)(K)(i) of the Act requires that the Secretary establish a mechanism to recognize the costs of new medical services or technologies under the payment system established under that subsection, which establishes the system for payment of the operating costs of inpatient hospital services, we do not include capital costs in the add-on payments for a new medical service or technology or make new technology add-on payments under the IPPS for capital-related costs ( 86 FR 45145 ). As noted, the applicant stated that the cost of the ASTar® Instrument is a capital cost. Therefore, we stated that it appeared that this component was not eligible for new technology add-on payment because, as discussed in prior ( print page 69210) rulemaking and as noted, we only make new technology add-on payments for operating costs ( 72 FR 47307 through 47308 ). We noted that any new technology add-on payment for the ASTar® System would include only the cost of ASTar® BC G-Kit ($150). We also noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of the ASTar® System would be $97.50 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether the ASTar® System meets the cost criterion and our proposal to approve new technology add-on payments for the ASTar® System for FY 2025, subject to the technology receiving FDA marketing authorization as a Breakthrough Device for the indication corresponding to the Breakthrough Device designation by May 1, 2024.
Comment: The applicant submitted a public comment expressing support for our proposal to approve new technology add-on payments for FY 2025 for the ASTar® System. The applicant reiterated that the ASTar® System meets the cost criterion and confirmed the maximum new technology add-on payment for the ASTar® System to cover the ASTar® BC G-Kit.
Response: We thank the applicant for its support to approve the new technology add-on payments for the ASTar® System.
Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the ASTar® System meets the cost criterion. The technology received 510(k) clearance on April 26, 2024, as a Breakthrough Device, with the following indication for use: the ASTar® System, comprised of the ASTar® Instrument with the ASTar® BC G-Kit (ASTar® BC G-Consumable kit, ASTar® BC G-Frozen insert, and ASTar® BC G-Kit software), utilizes high-speed, time-lapse microscopy imaging of bacteria for the in vitro, quantitative determination of antimicrobial susceptibility of on-panel gram-negative bacteria. The test is performed directly on positive blood culture samples signaled as positive by a continuous monitoring blood culture system and confirmed to contain gram-negative bacilli by Gram stain. Since the indication for which the applicant received FDA 510(k) clearance is included within the scope of the Breakthrough Device designation, we are finalizing our proposal to approve new technology add-on payments for the ASTar® System for FY 2025. We consider the beginning of the newness period to commence on April 26, 2024, the date on which technology received FDA marketing authorization for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the ASTar® System is $150, based on the operating component ASTar® BC G-Kit (composed of the ASTar® BC G-Consumable Kit ($141) and ASTar BC G-Frozen Insert ($9). Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the ASTar® System is $97.50 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of the ASTar® System that are eligible for new technology add-on payments will be identified by ICD-10-PCS procedure code XXE5X2A (Measurement of infection, phenotypic fully automated rapid susceptibility technology with controlled inoculum, new technology group 10).
Edwards Lifesciences LLC submitted an application for new technology add-on payments for the Edwards EVOQUE TM Tricuspid Valve Replacement System (“EVOQUE TM System”) for FY 2025. According to the applicant, the EVOQUE TM System is a new, transcatheter treatment option for patients with at least severe tricuspid regurgitation. Per the applicant, the EVOQUE TM System is designed to replace the native tricuspid valve and consists of a transcatheter bioprosthetic valve, a catheter-based delivery system, and supporting accessories.
Please refer to the online application posting for the Edwards EVOQUE TM Tricuspid Valve Replacement System, available at https://mearis.cms.gov/public/publications/ntap/NTP231013MRRBG , for additional detail describing the technology and the condition treated by the technology.
According to the applicant, the EVOQUE TM System received Breakthrough Device designation from FDA on December 18, 2019, for the treatment of patients with symptomatic moderate or above tricuspid regurgitation. The applicant stated that the technology received premarket approval from FDA on February 1, 2024 for a narrower indication for use, for the improvement of health status in patients with symptomatic severe tricuspid regurgitation despite optimal medical therapy, for whom tricuspid valve replacement is deemed appropriate by a heart team. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36113 ), we noted that since the indication for which the applicant received premarket approval is included within the scope of the Breakthrough Device designation, it appears that the PMA indication is appropriate for consideration for new technology add-on payment under the alternative pathway criteria. According to the applicant, the EVOQUE TM System was commercially available immediately after FDA approval.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for the EVOQUE TM System beginning in FY 2025 and was granted approval for the following procedure code effective October 1, 2024: X2RJ3RA (Replacement of tricuspid valve with multi-plane flex technology bioprosthetic valve, percutaneous approach, new technology group 10). The applicant stated that ICD-10-CM diagnosis codes I07.1 (Rheumatic tricuspid insufficiency), I07.2 (Rheumatic tricuspid stenosis and insufficiency), I36.1 (Nonrheumatic tricuspid (valve) insufficiency), and I36.2 (Nonrheumatic tricuspid (valve) stenosis with insufficiency) may be used to currently identify the indication for the EVOQUE TM System under the ICD-10-CM coding system.
With respect to the cost criterion, the applicant provided two analyses to demonstrate that the technology meets the cost criterion. To identify potential cases representing patients who may be eligible for the EVOQUE TM System, each analysis used the same ICD-10-CM diagnosis codes in different positions, with and without selected ICD-10-PCS procedure codes, to identify relevant cases in the FY 2022 MedPAR file. Each analysis followed the order of operations described in the table that follows later in this section.
For the first analysis, the applicant searched for cases assigned to MS-DRGs 266 (Endovascular Cardiac Valve Replacement and Supplement Procedures with MCC) and 267 (Endovascular Cardiac Valve ( print page 69211) Replacement and Supplement Procedures without MCC) that included one of the four ICD-10-CM diagnosis codes in any position, as listed in the table that follows later in this section. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 2,728 claims mapping to the two MS-DRGs and calculated a final inflated average case-weighted standardized charge per case of $267,720, which exceeded the average case-weighted threshold amount of $194,848.
For the second analysis, the applicant searched for the cases that included any of the ICD-10-PCS codes for percutaneous repair or replacement of the tricuspid valve in any position, in combination with one of the four ICD-10-CM codes for tricuspid valve insufficiency as the primary diagnosis, as listed in the table that follows later in this section. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 198 claims mapping to 6 MS-DRGs and calculated a final inflated average case-weighted standardized charge per case of $327,236, which exceeded the average case-weighted threshold amount of $219,225.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant asserted that the EVOQUE TM System meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36114 ), we agreed with the applicant that the EVOQUE TM System meets the cost criterion and therefore proposed to approve the EVOQUE TM System for new technology add-on payments for FY 2025.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the total cost of the EVOQUE TM System to the hospital to be $49,000 per patient, which includes the following components: the EVOQUE TM Tricuspid Delivery System, the EVOQUE TM Dilator Kit, the EVOQUE TM Loading System, the Stabilizer, Base, and Plate, and the EVOQUE TM Valve. The applicant noted that the listed ( print page 69213) components of the EVOQUE TM System are sold together as one unit because they are all needed to perform the procedure, are all single patient use, and are not sold separately. We noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of the EVOQUE TM System would be $31,850 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether the EVOQUE TM System meets the cost criterion and our proposal to approve new technology add-on payments for the EVOQUE TM System for FY 2025 for the improvement of health status in patients with symptomatic severe tricuspid regurgitation despite optimal medical therapy, for whom tricuspid valve replacement is deemed appropriate by a heart team.
Comment: The applicant and other commenters submitted public comments expressing support for the approval of the EVOQUE TM System for new technology add-on payment for FY 2025.
Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the EVOQUE TM System meets the cost criterion. The technology received FDA premarket approval on February 1, 2024 as a Breakthrough Device, with an indication for use for the improvement of health status in patients with symptomatic severe tricuspid regurgitation despite optimal medical therapy, for whom tricuspid valve replacement is deemed appropriate by a heart team, which is covered by its Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for the EVOQUE TM System for FY 2025. We consider the beginning of the newness period to commence on February 1, 2024, the date on which the technology received its FDA marketing authorization for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the EVOQUE TM System is $49,000 per patient. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the EVOQUE TM System is $31,850 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of the EVOQUE TM System that are eligible for new technology add-on payments will be identified by ICD-10-PCS procedure code: X2RJ3RA (Replacement of tricuspid valve with multi-plane flex technology bioprosthetic valve, percutaneous approach, new technology group 10).
W.L. Gore & Associates, Inc. submitted an application for new technology add-on payments for the TAMBE Device for FY 2025. According to the applicant, the TAMBE Device is used for endovascular repair in patients with thoracoabdominal aortic aneurysms (TAAA) and high-surgical risk patients with pararenal abdominal aortic aneurysms (PAAA) who have appropriate anatomy. Per the applicant, the TAMBE Device is comprised of multiple required components, including: (1) an Aortic Component, (2) Branch Components, (3) a Distal Bifurcated Component, and (4) Contralateral Leg Component. According to the applicant, these components together comprise the TAMBE Device.
Please refer to the online application posting for the GORE® EXCLUDER® Thoracoabdominal Branch Endoprosthesis (TAMBE Device), available at https://mearis.cms.gov/public/publications/ntap/NTP231016DYQQX , for additional detail describing the technology and the condition treated by the technology.
According to the applicant, the TAMBE Device received Breakthrough Device designation from FDA on October 1, 2021, for endovascular repair of thoracoabdominal and pararenal aneurysms in the aorta in patients who have appropriate anatomy. According to the applicant, the TAMBE Device received premarket approval (PMA) from FDA on January 12, 2024, for a slightly narrower indication for use, namely, TAAA and high-surgical risk patients with PAAA who have appropriate anatomy. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36115 ), we noted that since the indication for which the applicant received premarket approval is included within the scope of the Breakthrough Device designation, it appears that the PMA indication is appropriate for consideration for new technology add-on payment under the alternative pathway criteria. According to the applicant, the TAMBE Device is not yet available for sale due to the required lead time to train physicians on the TAMBE Device, and the first commercial device will only be implanted May 1, 2024 or later. We stated in the proposed rule that we were interested in additional information regarding the delay in the technology's market availability, as we questioned whether the date the device first became available for sale would be the same as the date the first commercial device is implanted.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for the TAMBE Device beginning in FY 2025 and was granted approval for the following procedure code effective October 1, 2024: X2VE3SA (Restriction of descending thoracic aorta and abdominal aorta using branched intraluminal device, manufactured integrated system, four or more arteries, percutaneous approach, new technology group 10). The applicant provided a list of diagnosis codes that may be used to currently identify the proposed indication for the TAMBE Device under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
With respect to the cost criterion, to identify potential cases representing patients who may be eligible for the TAMBE Device, the applicant searched the FY 2022 MedPAR file for claims that had at least one of the ICD-10-CM codes and at least one of the ICD-10-PCS codes as listed in the following table. Using the inclusion/exclusion criteria described in the following table, the applicant identified 1,005 claims mapping to 19 MS-DRGs, including MS-DRG 269 (Aortic and Heart Assist Procedures except Pulsation Balloon without MCC), which represented 54.5 percent of the identified cases. The applicant followed the order of operations described in the following table and calculated a final inflated average case-weighted standardized charge per case of $448,347, which exceeded the average case-weighted threshold amount of $185,799. Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold ( print page 69214) amount, the applicant asserted that the TAMBE Device meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36116 ), we agreed with the applicant that the TAMBE Device meets the cost criterion and therefore proposed to approve the TAMBE Device for new technology add-on payments for FY 2025.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the total cost of the TAMBE Device to the hospital to be $72,675 per patient. Per the applicant, the TAMBE Device has a number of required components, including the aortic component ($29,000), branch components ($3,355), distal bifurcated component (DBC) ($10,758), DBC extender component ($3,037), contralateral leg endoprosthesis ($4,390), and iliac extender endoprosthesis ($3,037). The applicant stated that the actual type and number of components used varies by patient depending on their anatomy and the extent of the patient's aneurysm. The applicant determined the number and types of components that were used in an average patient based on a multicenter pivotal clinical trial conducted predominantly in the U.S. and calculated the case cost per component. We noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of the TAMBE Device would be $47,238.75 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether the TAMBE Device meets the cost criterion and our proposal to approve new technology add-on payments for the TAMBE Device for FY 2025, for endovascular repair in patients with thoracoabdominal aortic ( print page 69215) aneurysms and high-surgical risk patients with pararenal aortic aneurysms who have appropriate anatomy.
Comment: The applicant submitted a public comment in response to CMS's request for additional information regarding the delay in the technology's market availability. The applicant reiterated that it anticipated the device would become available for sale in early May 2024, and the first implanted case would occur May 1, 2024 or later, as discussed in the proposed rule. The applicant stated that the first implant was conducted by the leading clinical investigator on May 10, 2024, and the TAMBE Device became commercially available on May 10, 2024, to U.S. physicians who have completed the necessary training. The applicant further stated that the FDA-approved Instructions for Use (IFU) requires that the TAMBE device should only be used by physicians who have successfully completed the appropriate physician training program. The applicant stated that to ensure high standards of care with this device, it had instituted a comprehensive clinical training program for physicians prior to implanting the device.
The applicant and another commenter expressed support for our proposal to approve new technology add-on payments for FY 2025 for the TAMBE Device. The applicant also agreed with the proposed maximum new technology add-on payment amount for the TAMBE Device.
Response: We thank the commenters for their comments. As discussed in prior rulemaking, we note that the timeframe that a new technology can be eligible to receive new technology add-on payments begins when data become available ( 69 FR 49003 , 85 FR 58610 ). Specifically, § 412.87(c)(2) states that a new medical device that is part of FDA's Breakthrough Devices Program and has received marketing authorization for the indication covered by the Breakthrough Device designation may be considered new for not less than 2 years and not more than 3 years after the point at which data begin to become available reflecting the inpatient hospital code assigned to the new service or technology (depending on when a new code is assigned and data on the new service or technology become available for DRG recalibration). We do not consider the date of first sale of a product as an indicator of the entry of a product onto the U.S. market ( 87 FR 48911 ). Similarly, although the applicant states that the date of first implantation of the TAMBE device was May 10, 2024, and that the TAMBE Device became commercially available on May 10, 2024 to U.S. physicians who have completed the necessary training, it is unclear from the information provided when the technology first became available for sale. We note that the information provided by the applicant indicating that the FDA-approved IFU requires that the TAMBE device should only be used by physicians who have successfully completed the appropriate physician training and that there was a comprehensive clinical training program for physicians prior to implanting the device, does not appear to address the delay in the technology's market availability, because the information provided identifies when the device was first able to be used by a physician, rather than when the device first became available for sale. Absent additional information from the applicant regarding when the technology first became available for sale, we cannot determine a newness date based on a documented delay in the technology's availability on the U.S. market.
Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the TAMBE Device meets the cost criterion. The technology received FDA premarket approval on January 12, 2024 as a Breakthrough Device, with an indication for endovascular repair in patients with TAAA and high-surgical risk patients with PAAA who have appropriate anatomy, which is covered by its Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for the TAMBE Device for FY 2025. As previously discussed, absent additional information from the applicant, we consider the beginning of the newness period to commence on January 12, 2024, the date on which the technology received FDA marketing authorization for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the TAMBE Device is $72,675, based on the average case cost per component for the: aortic component, branch components, distal bifurcated component (DBC), DBC extender component, contralateral leg endoprosthesis, and iliac extender endoprosthesis. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the TAMBE Device is $47,238.75 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of the TAMBE Device that are eligible for new technology add-on payments will be identified by ICD-10-PCS procedure code X2VE3SA (Restriction of descending thoracic aorta and abdominal aorta using branched intraluminal device, manufactured integrated system, four or more arteries, percutaneous approach, new technology group 10).
LimFlow Inc. submitted an application for new technology add-on payments for the LimFlow TM System for FY 2025. According to the applicant, the LimFlow TM System is a single-use, medical device system designed to treat patients who have chronic limb-threatening ischemia with no suitable endovascular or surgical revascularization options and are at risk of major amputation. Per the applicant, the LimFlow TM System consists of LimFlow's Cylindrical and Conical Stent Grafts that are used in conjunction with a LimFlow TM Arterial Catheter, a LimFlow TM Venous Catheter, and a LimFlow TM Valvulotome. According to the applicant, the LimFlow TM System is used for transcatheter arterialization of the deep veins, a minimally invasive procedure that aims to restore blood flow to the ischemic foot by diverting a stream of oxygenated blood through tibial veins in order to permanently bypass heavily calcified and severely stenotic arteries defined as unreconstructable. We note that LimFlow Inc. submitted an application for new technology add-on payments for the LimFlow TM System for FY 2024 as summarized in the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 26938 through 26940 ), but the technology did not meet the applicable deadline of July 1, 2023 for FDA approval or clearance of the technology and, therefore, was not eligible for consideration for new technology add-on payments for FY 2024 ( 88 FR 58919 ).
Please refer to the online application posting for the LimFlow TM System, available at https://mearis.cms.gov/public/publications/ntap/NTP23101627LXC , for additional detail describing the technology and the condition treated by the technology.
According to the applicant, the LimFlow TM System received Breakthrough Device designation from FDA on October 3, 2017, for the treatment of critical limb ischemia by minimally invasively creating an ( print page 69216) arterio-venous bypass graft to produce the venous arterialization procedure in the below-the-knee vasculature. The applicant stated that the technology was granted premarket approval from FDA on September 11, 2023, for patients who have chronic limb-threatening ischemia with no suitable endovascular or surgical revascularization options and are at risk of major amputation. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36117 ), we noted that since the indication for which the applicant received premarket approval is considered equivalent to the Breakthrough Device designation, it appears that the premarket approval indication is appropriate for consideration for new technology add-on payment under the alternative pathway criteria. Per the applicant, the LimFlow TM System was not immediately available for sale because inventory build and ramp for commercial sales was set to commence following FDA approval to allow time for the conduct of surgeon training and medical education on patient selection, indications, and surgical technique. The applicant stated that the technology became commercially available on November 1, 2023.
The applicant provided a list of ICD-10-PCS codes that, effective October 1, 2018, can be used to uniquely describe procedures involving the use of the LimFlow TM System under the ICD-10-PCS coding system. Please see the online posting for the LimFlow TM System for the complete list of ICD-10-PCS codes provided by the applicant. The applicant provided a list of diagnosis codes that may be used to currently identify the indication for the LimFlow TM System under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
With respect to the cost criterion, the applicant provided three analyses to demonstrate that it meets the cost criterion. Each analysis used the same ICD-10-PCS codes to identify potential cases representing patients who may be eligible for the LimFlow TM System. The applicant stated that the selected claims represent the exact situations in which the LimFlow TM System would be used and represent the cost of care associated with the use of the LimFlow TM System. The applicant utilized a different year of MedPAR data in each analysis. According to the applicant, it used multiple years of data because the case count in each individual year was low. The applicant imputed a value of 11 cases for MS-DRGs with less than 11 cases. Each analysis followed the order of operations described in the table that follows later in this section.
For the first analysis, the applicant searched FY 2022 MedPAR data for claims reporting at least one of the ICD-10-PCS codes listed in the table that follows later in this section to identify cases that may be eligible for the LimFlow TM System. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 88 claims mapping to 8 MS-DRGs, with none exceeding more than 13 percent of the total identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $307,461 which exceeded the average case-weighted threshold amount of $124,971.
For the second analysis, the applicant searched FY 2021 MedPAR data for claims reporting at least one of the ICD-10-PCS codes listed in the table that follows later in this section to identify cases that may be eligible for the LimFlow TM System. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 111 claims mapping to 10 MS-DRGs, with none exceeding more than 11 percent of the total identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $277,454, which exceeded the average case-weighted threshold amount of $116,278.
For the third analysis, the applicant searched FY 2020 MedPAR data for claims reporting at least one of the ICD-10-PCS codes listed in the table that follows later in this section to identify cases that may be eligible for the LimFlow TM System. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 99 claims mapping to 9 MS-DRGs, with none exceeding more than 12 percent of the total identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $273,638 which exceeded the average case-weighted threshold amount of $125,153.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant asserted that the LimFlow TM System meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36118 ), we agreed with the applicant that the LimFlow TM System meets the cost criterion and therefore proposed to approve the LimFlow [ TM ] System for new technology add-on payments for FY 2025.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the total cost of the LimFlow TM System to the hospital to be $25,000 per patient. According to the applicant, the LimFlow TM System is sold as a system, as such, the components of the LimFlow TM System are not priced or sold to hospitals independently. The applicant stated that all components of the LimFlow TM System are single-use and the entire system is an operating cost. We noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of the LimFlow TM System would be $16,250 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether the LimFlow TM System meets the cost criterion and our proposal to approve new technology add-on payments for the LimFlow TM System for FY 2025 for patients who have chronic limb-threatening ischemia with no suitable endovascular or surgical revascularization options and are at risk of major amputation. ( print page 69218)
Comment: Commenters, including the applicant, submitted public comments expressing support for the approval of the LimFlow TM System for new technology add-on payment for FY 2025.
The applicant stated that the LimFlow TM System addresses an unmet need in late-stage chronic limb-threatening ischemia patients, who are no longer candidates for conventional endovascular or open bypass surgery to resolve their artery blockage and face major amputation as their only therapeutic option. The applicant stated that the LimFlow TM System is the first and only FDA approved device for transcatheter arterialization of the deep veins. The applicant restated that the LimFlow TM System received Breakthrough Device designation from FDA on October 3, 2017, and the LimFlow TM System received FDA premarket approval on September 11, 2023. The applicant stated that the LimFlow TM System was not immediately available for sale after FDA approval because it had to modify its commercial manufacturing strategy at the end of the PMA review process. The applicant asserted that this manufacturing delay prevented the first commercial product from being available for sale until November 1, 2023. The applicant also included a letter from the treating physician who performed the first U.S. commercial case on November 2, 2023. The applicant reiterated that the LimFlow TM System meets the cost criterion and confirmed the proposed cost of the LimFlow TM System to the hospital of $25,000 per patient. The applicant agreed that the proposed maximum new technology add-on payment amount for a case involving the use of the LimFlow TM System would be $16,250 for FY 2025 (that is, 65 percent of the average cost of the technology).
Response: We thank the commenters for their support. Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the LimFlow TM System meets the cost criterion. The technology received FDA premarket approval on September 11, 2023, as a Breakthrough Device, with an indication for patients who have chronic limb-threatening ischemia with no suitable endovascular or surgical revascularization options and are at risk of major amputation, which is considered equivalent to the Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for the LimFlow TM System for FY 2025. We consider the beginning of the newness period to commence on November 1, 2023, the date on which the technology became commercially available for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the LimFlow TM System is $25,000. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the LimFlow TM System is $16,250 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of the LimFlow TM System that are eligible for new technology add-on payments will be identified by one of the following ICD-10-PCS procedure codes:

ReCor Medical submitted an application for new technology add-on payments for the Paradise TM Ultrasound Renal Denervation System for FY 2025. According to the applicant, the Paradise TM Ultrasound Renal Denervation System is an endovascular catheter-based system that delivers SonoWave360 TM ultrasound energy circumferentially, thermally ablating and disrupting overactive renal sympathetic nerves to lower blood pressure in adult (≥22 years of age) patients with uncontrolled hypertension who may be inadequately responsive to or who are intolerant to anti-hypertensive medications.
Please refer to the online application posting for the Paradise TM Ultrasound Renal Denervation System, available at https://mearis.cms.gov/public/publications/ntap/NTP23101772HBQ , for additional detail describing the technology and the condition treated by the technology.
According to the applicant, the Paradise TM Ultrasound Renal Denervation System received Breakthrough Device designation from FDA on December 4, 2020, for reducing blood pressure in adult (≥22 years of age) patients with uncontrolled hypertension, who may be inadequately responsive to, or who are intolerant to anti-hypertensive medications. The applicant received FDA premarket approval for the technology on November 7, 2023, for reducing blood pressure as an adjunctive treatment in hypertension patients in whom lifestyle modifications and antihypertensive ( print page 69219) medications do not adequately control blood pressure. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36119 ), we noted that because we consider the indication for which the applicant received premarket approval to be within the scope of the Breakthrough Device designation, and FDA considers this marketing authorization to be for the Breakthrough Device designation, [ 173 ] it appears that the premarket approval indication is appropriate for consideration for new technology add-on payment under the alternative pathway criteria. According to the applicant, the technology was commercially available immediately after FDA approval.
The applicant stated that effective October 1, 2023, the following ICD-10-PCS code may be used to uniquely describe procedures involving the use of the Paradise TM Ultrasound Renal Denervation System: X051329 (Destruction of renal sympathetic nerve(s) using ultrasound ablation, percutaneous approach, new technology group 9). The applicant stated that ICD-10-CM codes I10 (Essential (primary) hypertension), I15.1 (Hypertension secondary to other renal disorders), I15.8 (Other secondary hypertension), I15.9 (Secondary hypertension, unspecified), and I1A.0 (Resistant hypertension) may be used to currently identify the indication for the Paradise TM Ultrasound Renal Denervation System under the ICD-10-CM coding system.
With respect to the cost criterion, the applicant provided multiple analyses to demonstrate that it meets the cost criterion. Each analysis used different MS-DRGs and/or ICD-10-CM codes to identify potential cases representing patients who may be eligible for the Paradise TM Ultrasound Renal Denervation System. The applicant explained that it used different codes to demonstrate different cohorts that may be eligible for the technology. Each analysis followed the order of operations described in the table that follows later in this section.
For the first analysis, the applicant searched the FY 2022 MedPAR file for all cases that map to MS-DRG 264 (Other Circulatory System O.R. Procedures). The applicant stated that medical MS-DRGs 304 and 305 (Hypertension with MCC and without MCC) are specific to hypertension. However, given the nature of the procedure, the applicant's expectation is that the DRG Grouper logic would assign potential cases representing patients who may be eligible for the Paradise TM Ultrasound Renal Denervation System to a surgical MS-DRG. To identify the surgical MS-DRG, the applicant identified ICD-10-PCS code 015M3ZZ (Destruction of abdominal sympathetic nerve, percutaneous approach) as the procedure most similar to the procedure performed using the Paradise TM Ultrasound Renal Denervation System, and determined the specific MS-DRG to which that ICD-10-PCS code maps. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 7,064 claims mapping to MS-DRG 264 (Other Circulatory System O.R. Procedures) and calculated a final inflated average case-weighted standardized charge per case of $357,807, which exceeded the average case-weighted threshold amount of $98,708.
For the second analysis, as a sensitivity analysis the applicant searched the FY 2022 MedPAR file for all cases that map to MS-DRGs 304 or 305 (Hypertension with MCC and without MCC), which are specific to hypertension. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 32,433 claims mapping to MS-DRG 304 (Hypertension with MCC) or 305 (Hypertension without MCC) and calculated a final inflated average case-weighted standardized charge per case of $268,298, which exceeded the average case-weighted threshold amount of $46,986.
For the third analysis, the applicant provided a sensitivity analysis that combined the first and second scenario together for a broader list of MS-DRGs. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 39,497 claims mapping to MS-DRGs 264 (Other Circulatory System O.R. Procedures), 304 (Hypertension with MCC), or 305 (Hypertension without MCC) and calculated a final inflated average case-weighted standardized charge per case of $284,306, which exceeded the average case-weighted threshold amount of $56,237.
For the fourth analysis, the applicant performed a sensitivity analysis to subset the cases assigned to MS-DRG 264 (Other Circulatory System O.R. Procedures) to those reporting the following ICD-10-CM codes: I10 (Essential (primary) hypertension), I15.1 (Hypertension secondary to other renal disorders), I15.8 (Other secondary hypertension), or I15.9 (Secondary hypertension, unspecified) in any position. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 1,477 claims mapping to MS-DRG 264 (Other Circulatory System O.R. Procedures) and calculated a final inflated average case-weighted standardized charge per case of $325,810, which exceeded the average case-weighted threshold amount of $98,708.
For the fifth analysis, the applicant performed a sensitivity analysis to subset the cases assigned to MS-DRGs 264 (Other Circulatory System O.R. Procedures), 304 (Hypertension with MCC), or 305 (Hypertension without MCC) to those reporting the following ICD-10-CM codes: I10 (Essential (primary) hypertension), I15.1 (Hypertension secondary to other renal disorders), I15.8 (Other secondary hypertension), or I15.9 (Secondary hypertension, unspecified) in any position. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 14,415 claims mapping to MS-DRGs 264 (Other Circulatory System O.R. Procedures), 304 (Hypertension with MCC), or 305 (Hypertension without MCC) and calculated a final inflated average case-weighted standardized charge per case of $272,701, which exceeded the average case-weighted threshold amount of $50,817.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all analyses, the applicant asserted that the Paradise TM Ultrasound Renal Denervation System meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36121 ), we agreed with the applicant that the Paradise TM Ultrasound Renal Denervation System meets the cost criterion and therefore proposed to approve the Paradise [ TM ] Ultrasound Renal Denervation System for new technology add-on payments for FY 2025.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the total cost of the Paradise TM Ultrasound Renal Denervation System to the hospital to be $23,000 per patient, based on single-use components including the operating costs of the catheter kit ($22,000), cable ($250), and cartridge ($750). We noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of the Paradise TM Ultrasound Renal Denervation System would be $14,950 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether the Paradise TM Ultrasound ( print page 69221) Renal Denervation System meets the cost criterion and our proposal to approve new technology add-on payments for the Paradise TM Ultrasound Renal Denervation System for FY 2025 for reducing blood pressure as an adjunctive treatment in hypertension patients in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure, which corresponds to the Breakthrough Device designation.
Comment: Multiple commenters including the applicant expressed support for approval of the Paradise TM Ultrasound Renal Denervation System for new technology add-on payments for FY 2025. The applicant restated that the Paradise TM Ultrasound Renal Denervation System received Breakthrough Device designation from FDA on December 4, 2020, and the Paradise TM Ultrasound Renal Denervation System received FDA premarket approval on November 7, 2023, for the same indication as the Breakthrough Device designation. The applicant reaffirmed that the Paradise TM Ultrasound Renal Denervation System is new for FY 2025, and acknowledged our proposed newness date as the date of FDA approval, when the technology became commercially available. The applicant reiterated that the Paradise TM Ultrasound Renal Denervation System meets the cost criterion and agreed with the proposed maximum new technology add-on payment amount for the Paradise TM Ultrasound Renal Denervation System. The applicant requested that CMS finalize as proposed.
Response: We thank the commenters for their support to approve new technology add-on payments for the Paradise TM Ultrasound Renal Denervation System.
Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the Paradise TM Ultrasound Renal Denervation System meets the cost criterion. The technology received FDA premarket approval on November 7, 2023, as a Breakthrough Device, with an indication for reducing blood pressure as an adjunctive treatment in hypertension patients in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure, which is covered by its Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for the Paradise TM Ultrasound Renal Denervation System for FY 2025. We consider the beginning of the newness period to commence on November 7, 2023, the date on which technology received FDA marketing authorization for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the Paradise TM Ultrasound Renal Denervation System is $23,000, based on single-use components including the operating costs of the catheter kit ($22,000), cable ($250), and cartridge ($750). Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the Paradise TM Ultrasound Renal Denervation System is $14,950 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of the Paradise TM Ultrasound Renal Denervation System that are eligible for new technology add-on payments will be identified by ICD-10-PCS procedure code X051329 (Destruction of renal sympathetic nerve(s) using ultrasound ablation, percutaneous approach, new technology group 9).
Medtronic, Inc. submitted an application for new technology add-on payments for the PulseSelect TM PFA Loop Catheter for FY 2025. According to the applicant, the PulseSelect TM PFA Loop Catheter is used to perform pulmonary vein isolation in cardiac catheter ablation to treat atrial fibrillation. Per the applicant, unlike existing methods that rely on thermal energy (either radiofrequency or cryoablation), PulseSelect TM employs non-thermal irreversible electroporation to induce cell death in cardiac tissue at the target site. According to the applicant, PulseSelect TM technology's non-thermal approach can avoid risks associated with existing thermal cardiac catheter ablation technologies.
Please refer to the online application posting for the PulseSelect TM PFA Loop Catheter, available at https://mearis.cms.gov/public/publications/ntap/NTP231017BMQKQ , for additional detail describing the technology and the disease treated by the technology.
According to the applicant, the PulseSelect TM PFA System, which includes a compatible Medtronic multi-electrode cardiac ablation catheter (the PulseSelect TM PFA Loop Catheter), received Breakthrough Device designation from FDA on September 27, 2018, for the treatment of drug refractory recurrent symptomatic atrial fibrillation. The Medtronic multi-electrode cardiac ablation catheter is also intended to be used for cardiac electrophysiological (EP) mapping and measuring of intracardiac electrograms, delivery of diagnostic pacing stimuli and verifying electrical isolation post-treatment. According to the applicant, the PulseSelect TM PFA System received premarket approval on December 13, 2023 for the following indication that reflects a slightly narrower patient population compared to the Breakthrough Device designation: for cardiac electrophysiological mapping (stimulation and recording) and for treatment of drug refractory, recurrent, symptomatic paroxysmal atrial fibrillation or persistent atrial fibrillation (episode duration less than 1 year). The applicant noted that the PulseSelect TM PFA System consists of two primary elements: the PulseSelect TM PFA Loop Catheter and the PulseSelect TM PFA Generator system, but that as capital equipment, the PulseSelect TM PFA Generator system is not the subject of this new technology add-on payment application. According to the applicant, the technology was commercially available immediately after FDA approval.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for the PulseSelect TM PFA System and was granted approval for the following procedure code effective April 1, 2024: 02583ZF (Destruction of conduction mechanism using irreversible electroporation, percutaneous approach). The applicant provided a list of diagnosis codes that may be used to currently identify the indication for the PulseSelect TM PFA Loop Catheter under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
With respect to the cost criterion, the applicant provided multiple analyses to demonstrate that it meets the cost criterion. The applicant stated that there is an expectation the PulseSelect TM PFA Loop Catheter will predominantly be used when both indicated uses are employed in a single patient case. Each analysis used different ICD-10-CM codes to identify potential cases representing patients who may be eligible for the PulseSelect TM PFA Loop Catheter. The applicant explained that it used different codes to demonstrate different cohorts that may be eligible for the technology. Each analysis followed the order of operations described in the table that follows later in this section. ( print page 69222)
For the first analysis, the applicant searched the FY 2022 MedPAR file for claims that had the ICD-10-PCS code 02583ZZ (Destruction of conduction mechanism, percutaneous approach) in any procedure code position on the claim and identified 98 MS-DRGs. The applicant limited the cost analysis to the top six MS-DRGs that had over 2 percent of cases in each MS-DRG (see the table that follows later in this section for a complete list of MS-DRGs provided by the applicant). According to the applicant, these six MS-DRGs represented 86 percent of all cardiac catheter ablation cases. Using the inclusion/exclusion criteria described in the table that follows later in this section, the applicant identified 14,695 claims mapping to these 6 MS-DRGs. The applicant followed the order of operations described in the table that follows later in this section and calculated a final inflated average case-weighted standardized charge per case of $176,942, which exceeded the average case-weighted threshold amount of $136,813.
For the second analysis, the applicant searched the FY 2022 MedPAR file for claims that had the ICD-10-PCS code 02583ZZ (Destruction of conduction mechanism, percutaneous approach) in any procedure code position on the claim, and had one of the ICD-10-CM codes for atrial fibrillation listed in the table that follows later in this section. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 12,088 claims mapping to the top six MS-DRGs (representing 82.3 percent of all cases) and calculated a final inflated average case-weighted standardized charge per case of $179,931, which exceeded the average case-weighted threshold amount of $136,782.
For the third analysis, the applicant searched the FY 2022 MedPAR file for claims that had the ICD-10-PCS code 02583ZZ (Destruction of conduction mechanism, percutaneous approach) in any procedure code position on the claim and had one of the ICD-10-CM codes for paroxysmal or persistent atrial fibrillation listed in the table that follows later in this section. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 9,446 claims mapping to the top six MS-DRGs (representing 64.3 percent of all cases) and calculated a final inflated average case-weighted standardized charge per case of $180,114, which exceeded the average case-weighted threshold amount of $136,193.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant asserted that the PulseSelect TM PFA Loop Catheter meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36123 ), we agreed with the applicant that the PulseSelect TM PFA Loop Catheter meets the cost criterion and therefore proposed to approve the PulseSelect TM PFA Loop Catheter for new technology add-on payments for FY 2025.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the cost of the PulseSelect TM PFA Loop Catheter to the hospital to be $9,750 per patient, and for the PulseSelect TM PFA Catheter Interface Cable to be $800 per patient, totaling $10,550 per inpatient stay. We noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives ( print page 69224) prior to the final rule. We noted that the applicant stated that the PulseSelect TM Pulsed Field Ablation (PFA) Interface Cable is listed as a component of the PulseSelect TM Pulsed Field Ablation (PFA) Generator Reusable Accessories. However, we noted the submitted new technology add-on payment application is for the PulseSelect TM PFA Loop Catheter, and that the applicant had specified in its application that the PulseSelect TM PFA Generator System is not the subject of this new technology add-on payment application. Therefore, we believed the total cost per inpatient stay should be based only on the cost of the PulseSelect TM PFA Loop Catheter, which is $9,750 per the applicant. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of the PulseSelect TM PFA Loop Catheter would be $6,337.50 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether the PulseSelect TM PFA Loop Catheter meets the cost criterion and our proposal to approve new technology add-on payments for the PulseSelect TM PFA Loop Catheter for FY 2025 for cardiac electrophysiological mapping (stimulation and recording) and for treatment of drug refractory, recurrent, symptomatic paroxysmal atrial fibrillation or persistent atrial fibrillation (episode duration less than 1 year).
Comment: The applicant submitted a public comment requesting that the cost of the PulseSelect TM PFA Catheter Interface Cable ($800) be included as an operating cost rather than a capital cost, since it is a sterilized, one-time use connector between the PulseSelect TM PFA Loop Catheter and the PulseSelect TM PFA Generator System.
Response: We thank the commenter for its comments. As we had noted in the proposed rule, the submitted new technology add-on payment application is for the PulseSelect TM PFA Loop Catheter, and not for the PulseSelect TM PFA System. As noted by the applicant, and in the FDA Summary of Safety and Effectiveness Data for the PulseSelect TM PFA System, [ 176 ] the PulseSelect TM PFA Interface Cable is not a component of the PulseSelect TM PFA Loop Catheter. Therefore, we do not consider the cost of the PulseSelect TM PFA Catheter Interface Cable as an operating cost for the PulseSelect TM PFA Loop Catheter, and the PulseSelect TM PFA Catheter Interface Cable is not eligible to be included in new technology add-on payments.
Comment: The applicant submitted a public comment requesting that the PulseSelect TM PFA technology be the only product eligible for the new technology add-on payment designation and requested clarity on how eligibility for the new technology add-on payment would be properly determined on hospital claims. The applicant stated that CMS has established that a technology that is substantially similar to an existing technology approved for new technology add-on payment under the traditional pathway also qualifies for new technology add-on payment within the eligibility period, even if a specific application for that technology was not submitted and considered through rulemaking ( 82 FR 38110 ). The applicant also stated that CMS wrote in the FY 2023 IPPS/LTCH PPS final rule that “. . . applications received for new technology add-on payments for FY 2021 and subsequent fiscal years for medical devices that are part of FDA's Breakthrough Devices Program and received FDA marketing authorization will be considered not substantially similar to an existing technology for purposes of the new technology add-on payment under the IPPS” ( 87 FR 48915 ). The applicant stated this language establishes that Breakthrough Devices approved for new technology add-on payment under the alternative pathway cannot be considered substantially similar to any other technologies, by definition. The applicant further stated that it was not evident how this distinction between new technology add-on payments approved under the traditional pathway and new technology add-on payments for Breakthrough Devices approved under the alternative pathway would be effectuated on a claim-by-claim basis, in instances when the same code may be used to describe procedures involving the new technology add-on payment-approved Breakthrough Device as well as other devices (which may or may not have Breakthrough Device status). The applicant stated it obtained a new ICD-10-PCS code for the PulseSelect TM PFA System, and that the code could be used to describe procedures involving at least one other irreversible electroporation device. The applicant requested that CMS clarify in the final rule that the PulseSelect TM PFA technology, as a Breakthrough device, is not substantially similar to any other technologies for purposes of new technology add-on payments under the alternative pathway, and provide guidance on how this policy will be effectuated in terms of claims processing to ensure the new technology add-on payment is triggered only in cases where the PulseSelect TM PFA System is used.
Response: We thank the commenter for its comments. As we previously noted, under the alternative pathway, in evaluating eligibility for the new technology add-on payment, a medical device designated under FDA's Breakthrough Devices Program that has received FDA marketing authorization will be considered not substantially similar to an existing technology for purposes of the new technology add-on payment under the IPPS, and will not need to meet the requirement under § 412.87(b)(1) that it represent an advance that substantially improves, relative to technologies previously available, the diagnosis or treatment of Medicare beneficiaries.
In addition, we note that procedure codes under the ICD-10-PCS are not manufacturer specific; rather, they are used to describe the hospital service that was performed. If, after consulting current official coding guidelines a hospital determines that an ICD-10-PCS procedure code associated with a new technology add-on payment describes the technology that it used in the performance of a procedure, the hospital may report the code and may be eligible to receive the associated new technology add-on payment. We note that similar procedures using the same device or technology may also be appropriately coded differently under the ICD-10-PCS classification. An entity that is seeking coding guidance may contact the American Hospital Association's Central Office on ICD-10-CM/PCS systems for such advice. [ 177 ] Hospitals are responsible for ensuring that they are correctly billing for the services they render.
Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the PulseSelect TM PFA Loop Catheter meets the cost criterion. The technology received FDA premarket approval on December 13, 2023 as a Breakthrough Device, with an indication for use for cardiac electrophysiological mapping (stimulation and recording) and for treatment of drug refractory, recurrent, symptomatic paroxysmal atrial fibrillation or persistent atrial ( print page 69225) fibrillation (episode duration less than 1 year), which is covered by its Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for the PulseSelect TM PFA Loop Catheter for FY 2025. We consider the beginning of the newness period to commence on December 13, 2023, the date on which the technology received its FDA marketing authorization for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the PulseSelect TM PFA Loop Catheter is $9,750 per inpatient stay. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the PulseSelect TM PFA Loop Catheter is $6,337.50 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of the PulseSelect TM PFA Loop Catheter that are eligible for new technology add-on payments will be identified by ICD-10-PCS procedure code: 02583ZF (Destruction of conduction mechanism using irreversible electroporation, percutaneous approach).
Medtronic submitted an application for new technology add-on payments for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter for FY 2025. According to the applicant, the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter provides a treatment option for patients with uncontrolled hypertension, when used with the Symplicity G3 TM Generator, by delivering targeted radiofrequency energy to the renal nerves, safely disrupting overactive sympathetic signaling between the kidneys and brain, as a treatment for uncontrolled hypertension.
Please refer to the online application posting for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter, available at https://mearis.cms.gov/public/publications/ntap/NTP2310161U617 , for additional detail describing the technology and the condition treated by the technology.
According to the applicant, the Symplicity Spyral TM Multi-Electrode Renal Denervation System received Breakthrough Device designation from FDA on March 27, 2020, for the reduction of blood pressure in patients with uncontrolled hypertension despite the use of anti-hypertensive medications or in patients who may have documented intolerance to anti-hypertensive medications. The applicant received premarket approval for the technology on November 17, 2023, for reducing blood pressure as an adjunctive treatment in patients with hypertension in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36126 ), we noted that because we consider the indication for which the applicant received premarket approval to be within the scope of the Breakthrough Device designation, and FDA considers this marketing authorization to be for the Breakthrough Device, [ 178 ] it appears that the premarket approval indication is appropriate for consideration for new technology add-on payment under the alternative pathway criteria. According to the applicant, the technology was commercially available immediately after FDA approval.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter beginning in FY 2025 and was granted approval for the following procedure code effective October 1, 2024: X05133A (Destruction of renal sympathetic nerve(s) using radiofrequency ablation, percutaneous approach, new technology group 10). The applicant provided a list of diagnosis codes that may be used to currently identify the indication for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter under the ICD-10-CM coding system. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant.
With respect to the cost criterion, the applicant provided two analyses and two sensitivity analyses to demonstrate that it meets the cost criterion. Each analysis used a common set of ICD-10-CM codes but different criteria for the inclusion/exclusion of MS-DRGs and outlier cases to identify potential cases representing patients who may be eligible for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter. The applicant explained that it used different codes to demonstrate different cohorts that may be eligible for the technology. Each analysis followed the order of operations described in the table that follows later in this section.
For the first scenario (Cost Analysis #1), the applicant searched the FY 2022 MedPAR file for cases where essential (primary) hypertension was the reason for the admission, using at least one of the ICD-10-CM diagnosis codes in the table that follows later in this section. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 490,387 claims mapping to 99 MS-DRGs, including MS-DRG 291 (Heart Failure and Shock With MCC) representing 67 percent of identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $136,450, which exceeded the average case-weighted threshold amount of $62,312.
The second scenario (Cost Analysis #1 with Outliers) was a sensitivity analysis that mirrored the first scenario, except that cases with outlier payments were included. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 501,760 claims mapping to 101 MS-DRGs, including MS-DRG 291 (Heart Failure and Shock With MCC) representing 66.7 percent of identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $145,001, which exceeded the average case-weighted threshold amount of $63,789.
For the third scenario (Cost Analysis #2), the applicant searched the FY 2022 MedPAR file for claims reporting any of the ICD-10-CM diagnosis codes listed in the table that follows later in this section but limited the case selection to MS-DRGs where the principal diagnosis was essential hypertension, and no procedures were performed. Per the applicant, this list represents a subset of cases that were most likely to benefit from the new procedural treatment option for primary hypertension. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 390,384 claims mapping to 8 MS-DRGs, including MS-DRG 291 (Heart Failure and Shock With MCC) representing 84.4 percent of identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $124,525, which exceeded the average case-weighted threshold amount of $52,861.
The fourth scenario (Cost Analysis #2 with Outliers) mirrored the third ( print page 69226) scenario, except that cases with outlier payments were included. The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 395,634 claims mapping to 8 MS-DRGs, including MS-DRG 291 (Heart Failure and Shock With MCC) representing 84.5 percent of identified cases. The applicant calculated a final inflated average case-weighted standardized charge per case of $128,356, which exceeded the average case-weighted threshold amount of $52,873.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant asserted that the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36128 ), we agreed with the applicant that the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter meets the cost criterion and therefore proposed to approve the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter for new technology add-on payments for FY 2025.
We noted in the proposed rule that an estimate for the cost of the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter was not available for publication at the time of the proposed rule. We stated that we expected the applicant to release cost information prior to the final rule, and we would provide an update regarding the new technology add-on payment amount for the technology, if approved, in the final rule. The applicant stated that there would be two components for the cost of the technology, including operating costs for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter and capital costs for the Symplicity G3 TM Generator. Because section 1886(d)(5)(K)(i) of the Act requires that the Secretary establish a mechanism to recognize the costs of new medical services or technologies under the payment system established under that subsection, which establishes the system for payment of the operating costs of inpatient hospital services, we do not include capital costs in the add-on payments for a new medical service or technology or make new technology add-on payments under the IPPS for capital-related costs ( 86 FR 45145 ). Based on the information from the applicant, it appeared that the Symplicity G3 TM Generator is a capital cost. Therefore, it appeared that this component is not eligible for new technology add-on payment because, as discussed in prior rulemaking and as noted, we only make new technology add-on payments for operating costs ( 72 FR 47307 through 47308 ). Any new technology add-on payment for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter would be subject to our policy under § 412.88(a)(2) where we limit new technology add-on payment to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case.
We invited public comments on whether the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter meets the cost criterion and our proposal to approve new technology add-on payments for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter for FY 2025 for reducing blood pressure as an adjunctive treatment in patients with hypertension in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure, which corresponds to the Breakthrough Device designation.
Comment: Multiple commenters including the applicant submitted public comments in support of our proposal to approve new technology add-on payments for FY 2025 for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter. The applicant provided the cost of the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter to the hospital of $16,000 per patient and requested that we finalize the proposed maximum new technology add-on payment amount of $10,400 for FY 2025 (that is, 65 percent of the average cost of the technology).
Response: We thank the commenters for their support to approve new technology add-on payments for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter.
Comment: A commenter expressed concerns regarding not disclosing cost information for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter at the time of the proposed rule. The commenter acknowledged that there was precedent to not disclose cost information where the technology has not received FDA marketing authorization at the time of the proposed rule. However, the commenter stated that given the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter received FDA marketing authorization on November 17, 2023 and was immediately available for sale after FDA approval, they believed that the applicant could have estimated the cost of the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter prior to December 18, 2023, which is the deadline for submitting additional information for its application for new technology add-on payment. The commenter stated that not disclosing the technology's cost prevents stakeholders from submitting fully informed comments given the lack of information. The commenter stated that it is critically important that going forward, CMS consistently apply its requirements and processes across all applicants to ensure a level playing field.
Response: We appreciate the commenter sharing its concern regarding not disclosing cost information for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter at the time of the proposed rule. As stated by the commenter, we frequently do not have cost information from some applicants at the time of the proposed rule, and therefore do not include cost information on those applications in the proposed rule. As discussed in previous rulemaking ( 87 FR 48981 ), where cost information is not yet available at the time of the proposed rule, we note (in the proposed rule) our expectation that the applicant ( print page 69228) will submit cost information prior to the final rule, and we indicate that we will provide an update regarding the new technology add-on payment amount for the technology, if approved, in the final rule. We further note that in assessing the cost criterion for new technology add-on payments, consistent with the formula specified in section 1886(d)(5)(K)(ii)(I) of the Act, we assess the adequacy of the MS-DRG prospective payment rate otherwise applicable to discharges involving the new medical service or technology by evaluating whether the charges for cases involving the new technology exceed certain threshold amounts. As discussed in the proposed rule, we agreed that based on the applicant's cost analysis, the final inflated case-weighted average standardized charge per case for the technology exceeded the applicable average case-weighted threshold amount. We also note that we include descriptions of the cost analyses provided for all applications in the proposed rule to allow for public comments on how the applications meet the cost criterion. Nevertheless, we will continue to consider the commenter's concerns with respect to those applications for which information about the technology's cost is not included in the proposed rule.
Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter meets the cost criterion. The technology received FDA premarket approval on November 17, 2023, as a Breakthrough Device, with an indication for reducing blood pressure as an adjunctive treatment in patients with hypertension in whom lifestyle modifications and antihypertensive medications do not adequately control blood pressure, which is covered by its Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter for FY 2025. We consider the beginning of the newness period to commence on November 17, 2023, the date on which technology received FDA marketing authorization for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the single-use Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter is $16,000.00. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter is $10,400.00 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of the Symplicity Spyral TM Multi-Electrode Renal Denervation Catheter that are eligible for new technology add-on payments will be identified by ICD-10-PCS procedure code X05133A (Destruction of renal sympathetic nerve(s) using radiofrequency ablation, percutaneous approach, new technology group 10).
Abbott submitted an application for new technology add-on payments for TriClip TM G4 for FY 2025. According to the applicant, TriClip TM G4 is intended for reconstruction of the insufficient tricuspid valve through tissue approximation via a transcatheter approach. The TriClip TM G4 System consists of the TriClip TM G4 Implant, Clip Delivery System and Steerable Guide. The applicant explained that the TriClip TM G4 Implant is a percutaneously delivered mechanical implant that helps close the tricuspid valve leaflets resulting in fixed tricuspid leaflet approximation throughout the cardiac cycle. According to the applicant, TriClip TM G4 is intended for the treatment of patients with symptomatic, severe tricuspid valve regurgitation, whose symptoms and tricuspid regurgitation (TR) severity persist despite being treated optimally with medical therapy.
Please refer to the online application posting for TriClip TM G4, available at https://mearis.cms.gov/public/publications/ntap/NTP231016N52MH , for additional detail describing the technology and the disease treated by the technology.
According to the applicant, the TriClip TM G4 System received Breakthrough Device designation from FDA on November 19, 2020, for the treatment of patients with symptomatic, severe tricuspid valve regurgitation, whose symptoms and TR severity persist despite being treated optimally with medical therapy. The technology received FDA premarket approval on April 1, 2024 as a Breakthrough Device, with an indication for improving the quality of life and functional status in patients with symptomatic severe tricuspid regurgitation despite optimal medical therapy, who are at intermediate or greater risk for surgery and in whom transcatheter edge-to-edge valve repair is clinically appropriate and is expected to reduce tricuspid regurgitation severity to moderate or less, as determined by a multidisciplinary heart team, which is covered by its Breakthrough Device designation.
According to the applicant, the following ICD-10-PCS code may be used to describe procedures involving the use of TriClip TM G4: 02UJ3JZ (Supplement tricuspid valve with synthetic substitute, percutaneous approach). The applicant noted at the time of its application that there were no FDA-approved technologies using this procedure code. The applicant stated that ICD-10-CM diagnosis codes I07.1 (Rheumatic tricuspid insufficiency) and I36.1 (Nonrheumatic tricuspid (valve) insufficiency) may be used to currently identify the indication for TriClip TM G4 under the ICD-10-CM coding system.
With respect to the cost criterion, to identify potential cases representing patients who may be eligible for TriClip TM G4, the applicant searched the 2022 Medicare Inpatient Hospital Standard Analytical File (100%) for claims that had one of the following ICD-10-CM codes, I07.1 (Rheumatic tricuspid insufficiency) or I36.1 (Nonrheumatic tricuspid (valve) insufficiency) in the primary position, in combination with ICD-10-PCS code 02UJ3JZ (Supplement tricuspid valve with synthetic substitute, percutaneous approach). Using the inclusion/exclusion criteria described in the following table, the applicant identified 235 claims mapping to two MS-DRGs, MS-DRG 266 (Endovascular Cardiac Valve Replacement and Supplement Procedures, with MCC), and 267 (Endovascular Cardiac Valve Replacement and Supplement Procedures, without MCC). The applicant followed the order of operations described in the following table and calculated a final inflated average case-weighted standardized charge per case of $313,389 which exceeded the average case-weighted threshold amount of $192,861.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount, the applicant asserted that TriClip TM G4 meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36132 ), we agreed with the applicant that TriClip TM G4 meets the cost criterion and therefore proposed to approve TriClip TM G4 for new technology add-on payments for FY 2025, subject to the technology receiving FDA marketing authorization as a Breakthrough Device for the indication corresponding to the Breakthrough Device designation by May 1, 2024.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the total cost of TriClip TM G4 to the hospital to be $40,000 per procedure. According to the applicant, the TriClip TM System is composed of multiple components: the TriClip TM G4 Implant, Clip Delivery System, and Steerable Guide Catheter. The applicant stated that all the components typically required for a single procedure are sold together for a single operating cost (for example, it is the same cost per procedure whether the patient requires one or two implants). We noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of TriClip TM G4 would be $26,000 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether TriClip TM G4 meets the cost criterion and our proposal to approve new technology add-on payments for TriClip TM G4 for FY 2025, subject to the technology receiving FDA marketing authorization as a Breakthrough Device for the indication corresponding to the Breakthrough Device designation by May 1, 2024.
Comment: We received multiple public comments in support of our proposal to approve new technology add-on payments for the TriClip TM G4 System. The commenters stated the technology meets FDA marketing authorization and cost criterion requirements for approval and also supported CMS's proposed maximum new technology add-on payment.
Response: We thank the commenters for their comments. Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe TriClip TM G4 meets the cost criterion. The technology received FDA premarket approval on April 1, 2024 as a Breakthrough Device, with an indication for improving the quality of life and functional status in patients with symptomatic severe tricuspid regurgitation despite optimal medical therapy, who are at intermediate or greater risk for surgery and in whom transcatheter edge-to-edge valve repair is clinically appropriate and is expected to reduce tricuspid regurgitation severity to moderate or less, as determined by a multidisciplinary heart team, which is covered by its Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for TriClip TM G4 for FY 2025. We consider the beginning of the newness period to commence on April 1, 2024, the date on which the technology received its market authorization for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of TriClip TM G4 (composed of the ( print page 69230) TriClip TM G4 Implant, Clip Delivery System, and Steerable Guide Catheter) is $40,000 per procedure. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of TriClip TM G4 is $26,000 for FY 2025 (that is, 65 percent of the average cost of the technology). Cases involving the use of TriClip TM G4 that are eligible for new technology add-on payments will be identified by ICD-10-PCS procedure code 02UJ3JZ (Supplement tricuspid valve with synthetic substitute, percutaneous approach).
Icotec Medical, Inc. submitted an application for new technology add-on payments for the VADER® Pedicle System for FY 2025. According to the applicant, the VADER® Pedicle System is a pedicle screw system for standard posterior fixation of the spinal column used to provide stabilization of infected spinal segments after debridement of infectious tissues. According to the applicant, the VADER® Pedicle System is made from high strength carbon fiber reinforced polyether ether ketone, which provides low artifact imaging to allow for post-operative surveillance of the healing of the infected spinal segment.
Please refer to the online application posting for the VADER® Pedicle System, available at https://mearis.cms.gov/public/publications/ntap/NTP231016CMGH3 , for additional detail describing the technology and the condition treated by the technology.
According to the applicant, the VADER® Pedicle System received Breakthrough Device designation from FDA on July 31, 2023 for stabilizing the thoracic and/or lumbar spinal column as an adjunct to fusion in patients diagnosed with an active spinal infection (for example, spondylodiscitis, osteomyelitis) who are at risk of spinal instability, progressive spinal deformity, or neurologic compromise, following surgical debridement. The applicant stated that the technology received 510(k) clearance from FDA on February 26, 2024, for the following indication, which is the subject of the new technology add-on payment application, and is consistent with the Breakthrough Device designation: to stabilize the thoracic and/or lumbar spinal column in patients who are or will be receiving concurrent medical treatment for an active spinal infection (for example, spondylodiscitis, osteomyelitis) that, without stabilization, could lead to deterioration of bony structures and misalignment with neurological compromise. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36132 ), we noted that the VADER® Pedicle System has received FDA 510(k) clearance for multiple indications since 2019. [ 180 ] We also noted that, under the eligibility criteria for approval under the alternative pathway for certain transformative new devices, only the use of the VADER® Pedicle System to stabilize the thoracic and/or lumbar spine as an adjunct to fusion in patients with spinal infection, and the FDA Breakthrough Device designation it received for that use, are relevant for purposes of the new technology add-on payment application for FY 2025. According to the applicant, the technology was commercially available immediately after 510(k) clearance from FDA.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for the VADER® Pedicle System beginning in FY 2025 and was granted approval for the following procedure codes effective October 1, 2024:

The applicant provided a list of diagnosis codes that may be used to currently identify the indication for the VADER® Pedicle System under the ICD-10-CM coding system, describing spinal infections including osteomyelitis, discitis, and spondylopathies of various vertebral spine body parts including the cervical, thoracic, and lumbar regions. Please refer to the online application posting for the complete list of ICD-10-CM codes provided by the applicant. As previously noted, only use of the technology for the indications corresponding to the Breakthrough Device designation would be relevant for new technology add-on payment purposes. Therefore, in the proposed rule ( 89 FR 36132 ) we stated that we believed that the relevant ICD-10-CM codes to identify the Breakthrough Device-designated indication would be the codes included in category M46 (Other inflammatory spondylopathies) under the ICD-10-CM classification in subcategories: M46.2- (Osteomyelitis of vertebra), M46.3- (Infection of intervertebral disc (pyogenic)), M46.4- (Discitis, unspecified), M46.5- (Other infective spondylopathies), M46.8- (Other specified inflammatory spondylopathies), and M46.9- (Unspecified inflammatory spondylopathy). We invited public comment on the use of these ICD-10-CM diagnosis codes to identify the Breakthrough Device-designated indication for purposes of the new technology add-on payment, if approved.
With respect to the cost criterion, to identify potential cases representing patients who may be eligible for the VADER® Pedicle System, the applicant searched the FY 2022 MedPAR file for claims reporting a combination of ICD-10-CM/PCS codes as listed in the online posting for the VADER® Pedicle System. The applicant believes these cases represent patients who have undergone fusion procedures and have been diagnosed with an active spinal infection (such as spondylodiscitis or osteomyelitis), and these patients are at risk of spinal instability, progressive spinal deformity, or neurologic compromise following surgical debridement, making them suitable candidates for the use of the technology. Using the inclusion/exclusion criteria ( print page 69232) described in the following table, the applicant identified 2,116 claims mapping to 22 MS-DRGs, with none exceeding more than 15 percent of the total identified cases. The applicant followed the order of operations described in the following table and calculated a final inflated average case-weighted standardized charge per case of $473,636, which exceeded the average case-weighted threshold amount of $197,922. Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount, the applicant asserted that the VADER® Pedicle System meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36133 ), we agreed with the applicant that the VADER® Pedicle System meets the cost criterion and therefore proposed to approve the VADER® Pedicle System for new technology add-on payments for FY 2025.
Based on preliminary information from the applicant at the time of the proposed rule, the applicant anticipated the total cost of the VADER® Pedicle System to the hospital to be $43,450 per patient. According to the applicant, the unit prices are $6,500 for a pedicle screw, $4,600 for a rod, and $350 for a set screw. The applicant stated that an average of five pedicle screws, two rods, and five set screws would be used for a spinal fusion procedure. The applicant calculated the total cost of the technology by multiplying the unit price of each component by the average number of that component used in the procedure. We noted that the cost information for this technology may be updated in the final rule based on revised or additional information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of the VADER® Pedicle System would be $28,242.50 for FY 2025 (that is, 65 percent of the average cost of the technology).
We invited public comments on whether the VADER® Pedicle System meets the cost criterion and our ( print page 69233) proposal to approve new technology add-on payments for the VADER® Pedicle System for FY 2025, when used to stabilize the thoracic and/or lumbar spinal column in patients who are or will be receiving concurrent medical treatment for an active spinal infection (for example, spondylodiscitis, osteomyelitis) that, without stabilization, could lead to deterioration of bony structures and misalignment with neurological compromise.
Comment: We received comments supporting our proposal to approve new technology add-on payments for FY 2025 for the VADER® Pedicle System.
Response: We thank the commenters for their support to approve new technology add-on payments for the VADER® Pedicle System.
We note that we did not receive any public comments on the ICD-10-CM diagnosis codes we provided in the proposed rule to identify the Breakthrough Device-designated indication for purposes of the new technology add-on payment.
Based on the information provided in the application for new technology add-on payments, and after consideration of the public comments we received, we believe the VADER® Pedicle System meets the cost criterion. The technology received 510(k) clearance from FDA on February 26, 2024 as a Breakthrough Device, with an indication for use to stabilize the thoracic and/or lumbar spinal column in patients who are or will be receiving concurrent medical treatment for an active spinal infection (for example, spondylodiscitis, osteomyelitis) that, without stabilization, could lead to deterioration of bony structures and misalignment with neurological compromise, which is covered by its Breakthrough Device designation. Therefore, we are finalizing our proposal to approve new technology add-on payments for the VADER® Pedicle System for FY 2025. We consider the beginning of the newness period to commence on February 26, 2024, the date on which technology received FDA marketing authorization for the indication covered by its Breakthrough Device designation.
Based on the information available at the time of this final rule, the cost per case of the VADER® Pedicle System is $43,450 per patient. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 65 percent of the average cost of the technology, or 65 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of the VADER® Pedicle System is $28,242.50 for FY 2025 (that is, 65 percent of the average cost of the technology). As noted earlier in this section, the VADER® Pedicle System has received FDA 510(k) clearance for multiple indications since 2019, and only the use of the VADER® Pedicle System to stabilize the thoracic and/or lumbar spine as an adjunct to fusion in patients with spinal infection, and the FDA Breakthrough Device designation it received for that use, are relevant for purposes of the new technology add-on payment application for FY 2025. Therefore, cases involving the use of the VADER® Pedicle System that are eligible for new technology add-on payments will be identified by any of the ICD-10-PCS procedure codes listed in the following table, in combination with any one of the ICD-10-CM diagnosis codes listed in a following table.

Basilea Pharmaceutica International Ltd, Allschwil submitted an application for new technology add-on payments for ZEVTERA TM (ceftobiprole medocaril) for FY 2025. According to the applicant, ZEVTERA TM is an advanced intravenous cephalosporin antibiotic designed to combat infections caused by antibiotic resistant pathogens. The applicant stated that ZEVTERA TM targets a wide range of Gram-positive and Gram-negative bacteria, including methicillin-resistant Staphylococcus aureus (MRSA), Streptococcus pneumoniae, including penicillin-non-susceptible pneumococci (PNSP) and Enterococcus faecalis, as well as non-Extended Spectrum Beta-Lactamase (non-ESBL) producing Enterobacterales. The applicant noted that ZEVTERA TM 's bactericidal activity is achieved by binding to essential penicillin-binding proteins, disrupting the synthesis of the bacterial cell wall's peptidoglycan layer and leading to bacterial cell death, which differentiates it from other beta-lactams by effectively addressing MRSA. Per the applicant, ZEVTERA TM is stable against certain beta-lactamases in both gram-positive and gram-negative bacteria. The applicant stated that Phase 3 studies submitted to the FDA demonstrate its non-inferiority compared to standard treatments in various infections, including Staphylococcus aureus bacteremia (SAB), acute bacterial skin and skin structure infections (ABSSSI), and community-acquired bacterial pneumonia (CABP).
Please refer to the online application posting for ZEVTERA TM , available at https://mearis.cms.gov/public/publications/ntap/NTP2310161DBB8 , for additional detail describing the technology and the disease treated by the technology.
According to the applicant, ZEVTERA TM received QIDP designations for CABP on July 20, 2015, for ABSSI on August 7, 2015, and for SAB on December 8, 2017. According to the applicant, ZEVTERA TM would be commercially available immediately after FDA approval. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36134 ), we noted that, as an application submitted under the alternative pathway for certain antimicrobial products at § 412.87(d), ZEVTERA TM is eligible for conditional approval for new technology add-on payments if it does not receive FDA marketing authorization by July 1, 2024, provided that the technology receives FDA marketing authorization before July 1 of the fiscal year for which the applicant applied for new technology add-on payments (that is, July 1, 2025), as provided in § 412.87(f)(3). The technology was granted NDA approval from FDA on April 3, 2024, with indications for the treatment of: adults with SAB, including those with right-sided infective endocarditis; adults with ABSSSI; and adult and pediatric patients three months to less than 18 years old with CABP. According to the applicant, for CABP and ABSSSI, ZEVTERA TM is dosed at 500mg and administered three times daily (Q8h) as a 2-hour intravenous infusion for 5-14 days. For SAB, it is administered four times daily (Q6h) for the first 8 days, followed by Q8h daily infusion for the subsequent days, up to a total of 42 days.
The applicant submitted a request for approval for a unique ICD-10-PCS procedure code for ZEVTERA TM beginning in FY 2025 and was granted approval for the following procedure codes effective October 1, 2024: XW0335A (Introduction of ceftobiprole medocaril anti-infective into peripheral vein, percutaneous approach) and XW0435A (Introduction of ceftobiprole medocaril anti-infective into central vein, percutaneous approach, new technology group 10). The applicant provided a list of diagnosis codes that may be used to currently identify the indication for ZEVTERA TM under the ICD-10-CM coding system, describing SAB, ABSSSI, and CABP. Please refer to the online application posting for the complete list of ICD-10-CM (and PCS) codes provided by the applicant. In the proposed rule ( 89 FR 36134 ), we stated our belief that the relevant combination of ICD-10-CM codes to identify the indication of SAB would be: R78.81 (Bacteremia) in combination with B95.61 (Methicillin susceptible Staphylococcus aureus infection as the cause of diseases classified elsewhere) or B95.62 (Methicillin resistant Staphylococcus aureus infection as the cause of diseases classified elsewhere). We invited public comments on the use of these ICD-10-CM diagnosis codes to identify the indication of SAB for purposes of the new technology add-on payment, if approved.
With respect to the cost criterion, the applicant provided multiple analyses to demonstrate that it meets the cost criterion. For each analysis, the applicant searched the FY 2022 MedPAR file using different sets of ICD-10-CM codes in the first five diagnosis positions to identify potential cases representing different cohorts of patients who may be eligible for ZEVTERA TM . The applicant performed the same analysis on ABSSSI, CABP, and SAB cases individually and for all indications combined.
For the first analysis, the applicant searched for claims with a diagnosis code for ABSSSI using the ICD-10-CM codes listed in the online posting for ZEVTERA TM . The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 261,397 claims mapping to 663 MS-DRGs and calculated a final inflated average case-weighted standardized charge per case of $114,279, which exceeded the average case-weighted threshold amount of $63,767.
For the second analysis, the applicant searched for claims with a diagnosis code for CABP using the ICD-10-CM codes listed in the online posting for ZEVTERA TM . The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 635,628 claims mapping to 611 MS-DRGs and calculated a final inflated average case-weighted standardized charge per case of $143,456, which exceeded the average case-weighted threshold amount of $78,778.
For the third analysis, the applicant searched for claims with a diagnosis code for SAB using the ICD-10-CM codes listed in the online posting for ZEVTERA TM . The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 105,068 claims mapping to 626 MS-DRGs and calculated a final inflated average case-weighted standardized charge per case of $165,809, which exceeded the average case-weighted threshold amount of $82,238.
For the fourth analysis, the applicant searched for claims with diagnosis codes for ABSSSI, CABP, or SAB in the first five positions on a claim, using the ICD-10-CM codes listed in the online ( print page 69237) posting for ZEVTERA TM . The applicant used the inclusion/exclusion criteria described in the table that follows later in this section. Under this analysis, the applicant identified 958,104 claims mapping to 680 MS-DRGs and calculated a final inflated average case-weighted standardized charge per case of $137,861, which exceeded the average case-weighted threshold amount of $75,097.
Because the final inflated average case-weighted standardized charge per case exceeded the average case-weighted threshold amount in all scenarios, the applicant asserted that ZEVTERA [ TM ] meets the cost criterion.

In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36135 ), we agreed with the applicant that ZEVTERA TM meets the cost criterion and therefore proposed to approve ZEVTERA TM for new technology add-on payments for FY 2025, subject to the technology receiving FDA marketing authorization for the indication corresponding to the QIDP designation by July 1, 2024. We noted as an application submitted under the alternative pathway for certain antimicrobial products at § 412.87(d), ZEVTERA TM is eligible for conditional approval for new technology add-on payments if it does not receive FDA marketing authorization by July 1, 2024, provided that the technology receives FDA marketing authorization before July 1 of the fiscal year for which the applicant applied for new technology add-on payments (that is, July 1, 2025), as provided in § 412.87(f)(3). We stated that if ZEVTERA TM receives FDA marketing authorization before July 1, 2025, the new technology add-on payment for cases involving the use of this technology would be made effective for discharges beginning in the first quarter after FDA marketing authorization is granted. We noted if FDA marketing authorization is received on or after July 1, 2025, no new technology add-on payments would be made for cases involving the use of ZEVTERA TM for FY 2025.
Based on preliminary information from the applicant at the time of the proposed rule, the pricing for this treatment is set at $125 per vial, and the recommended dosage varies depending on the condition being treated. The applicant stated that for ABSSSI and CABP, the suggested daily dose is 3 vials per day for a duration of 5-14 days, resulting in an estimated average cost of $3,750 for a 10-day therapy. The applicant noted that for SAB, the recommended dose is every 6 hours for the first 8 days, followed by every 8 hours for up to 42 days. The applicant made the assumption that patients would be inpatient for 28 days and then continue the therapy as an outpatient for up to 42 days, which resulted in an average inpatient cost of $11,500. We noted that the cost information for this technology may be updated in the final rule based on revised or additional ( print page 69238) information CMS receives prior to the final rule. Under § 412.88(a)(2), we limit new technology add-on payments for technologies designated as QIDPs to the lesser of 75 percent of the average cost of the technology, or 75 percent of the costs in excess of the MS-DRG payment for the case. As a result, we proposed that the maximum new technology add-on payment for a case involving the use of ZEVTERA TM for FY 2025 would be $8,625.00 for the indication of SAB and $2,812.50 for the indications of ABSSSI and CABP (that is, 75 percent of the average cost of the technology).
We invited public comments on whether ZEVTERA TM meets the cost criterion and our proposal to approve new technology add-on payments for ZEVTERA TM for FY 2025 for SAB, ABSSSI, and CABP, subject to the technology receiving FDA marketing authorization consistent with its QIDP designations by July 1, 2024.
We did not receive any comments related to ZEVTERA TM .
Based on the information provided in the application for new technology add-on payments, we believe ZEVTERA TM meets the cost criterion. The technology received NDA approval from FDA on April 3, 2024, with indications for the treatment of: adults with SAB, including those with right-sided infective endocarditis; adults with ABSSSI; and adult and pediatric patients three months to less than 18 years old with CABP. Therefore, we are finalizing our proposal to approve ZEVTERA TM for new technology add-on payments for FY 2025. We consider the beginning of the newness period to commence on April 3, 2024, the date on which the technology received its FDA marketing authorization for the indications covered by its QIDP designations.
Based on the information available at the time of this final rule, the average inpatient cost per case of ZEVTERA TM is $11,500 for the indication of SAB and $3,750 for the indication of ABSSSI and CABP. Under § 412.88(a)(2), we limit new technology add-on payments to the lesser of 75 percent of the average cost of the technology, or 75 percent of the costs in excess of the MS-DRG payment for the case. As a result, we are finalizing that the maximum new technology add-on payment for a case involving the use of ZEVTERA TM is $8,625.00 for the indication of SAB and $2,812.50 for the indications of ABSSSI and CABP for FY 2025 (that is, 75 percent of the average cost of the technology).
Cases involving the use of ZEVTERA TM for the indications of ABSSSI and CABP that are eligible for new technology add-on payments will be identified by the following ICD-10-PCS procedure codes: XW0335A (Introduction of ceftobiprole medocaril anti-infective into peripheral vein, percutaneous approach) or XW0435A (Introduction of ceftobiprole medocaril anti-infective into central vein, percutaneous approach).
Cases involving the use of ZEVTERA TM for the indication of SAB that are eligible for new technology add-on payments will be identified by the following ICD-10-PCS procedure codes: XW0435A (Introduction of ceftobiprole medocaril anti-infective into central vein, percutaneous approach) or XW0435A (Introduction of ceftobiprole medocaril anti-infective into central vein, percutaneous approach), in combination with ICD-10-CM codes R78.81 (Bacteremia), in combination with B95.61 (Methicillin susceptible Staphylococcus aureus infection as the cause of diseases classified elsewhere) or B95.62 (Methicillin resistant Staphylococcus aureus infection as the cause of diseases classified elsewhere).
We received several public comments requesting changes to the new technology add-on payment policies such as creating new alternative pathway categories for different FDA designations or types of treatments, expanding the conditional approval process to additional types of technologies or designations, moving to a biannual process that would set two annual deadlines for manufacturers to apply for new technology add-on payment, and requiring Medicare Advantage (MA) to provide new technology add-on payment. These comments were outside the scope of the proposals included in the FY 2025 IPPS/LTCH PPS proposed rule and we are therefore not addressing them in this final rule.
As discussed previously in this rule, section 1886(d)(5)(K)(i) of the Act requires the Secretary to establish (after notice and opportunity for public comment) a mechanism to recognize the costs of new medical services and technologies under the IPPS. Section 1886(d)(5)(K)(vi) of the Act specifies that a medical service or technology will be considered new if it meets criteria established by the Secretary after notice and opportunity for public comment. The regulations at 42 CFR 412.87 implement these provisions. As further discussed in FY 2005 IPPS final rule ( 69 FR 49002 ), the intent of section 1886(d)(5)(K) of the Act and regulations under § 412.87(b)(2) is to pay for new medical services and technologies for the first 2 to 3 years that a product comes on the market, during the period when the costs of the new technology are not yet fully reflected in the DRG weights. Generally, we use the FDA marketing authorization date as the indicator of the time when a technology begins to become available on the market and data reflecting the costs of the technology begin to become available for recalibration of the DRG weights. In specific circumstances, we have recognized a date later than the FDA marketing authorization date as the appropriate starting point for the 2- to 3-year newness period. For example, we have recognized a later date where an applicant could prove a delay in actual availability of a product after FDA approval or clearance. The costs of the new medical service or technology, once paid for by Medicare for this 2- to 3-year period, are accounted for in the MedPAR data that are used to recalibrate the DRG weights on an annual basis. Therefore, we stated it is appropriate to limit the add-on payment window for technologies that have passed this 2- to 3-year timeframe.
As discussed previously in this rule, our policy is that a medical service or technology may continue to be considered “new” for purposes of new technology add-on payments within 2 or 3 years after the point at which data begin to become available reflecting the inpatient hospital code assigned to the new service or technology. Our practice has been to begin and end new technology add-on payments on the basis of a fiscal year, and we have generally followed a guideline that uses a 6-month window before and after the start of the fiscal year to determine whether to extend the new technology add-on payment for an additional fiscal year. In general, we extend new technology add-on payments for an additional year only if the 3-year anniversary date of the product's entry onto the U.S. market occurs in the latter half of the fiscal year, that is, after April 1 ( 70 FR 47362 ).
We have not implemented a policy to stop new technology add-on payment in the middle of the fiscal year (for example, during the month that a technology reaches its three-year anniversary date of entry onto the U.S. market) because, as we discussed in the FY 2005 IPPS final rule, we believe that predictability is an important aspect of the prospective payment system ( print page 69239) methodology. Accordingly, we believe that it is appropriate to apply a consistent payment methodology for new technologies throughout the fiscal year ( 69 FR 49016 ).
As previously discussed, in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ), we finalized that beginning with the new technology add-on payment applications for FY 2025, for technologies that are not already FDA market authorized for the indication that is the subject of the new technology add-on payment application, applicants must have a complete and active FDA marketing authorization request at the time of new technology add-on payment application submission and must provide documentation of FDA acceptance or filing to CMS at the time of application submission, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. We also finalized that, beginning with FY 2025 applications, in order to be eligible for consideration for new technology add-on payment for the upcoming fiscal year, an applicant for new technology add-on payments must have received FDA approval or clearance by May 1 (rather than July 1) of the year prior to the beginning of the fiscal year for which the application is being considered (except for an application that is submitted under the alternative pathway for certain antimicrobial products).
As we summarized in the FY 2024 IPPS/LTCH PPS final rule, commenters raised concerns that this policy would adversely impact their ability to receive maximum flexibility with respect to when to apply to FDA and when they apply for new technology add-on payment ( 88 FR 58953 ). Many commenters expressed specific concerns regarding moving the FDA marketing authorization deadline to May 1 and the impact it would have on how long technologies may be eligible for new technology add-on payment. Several of the commenters asserted that this policy change would prevent a 3-year new technology add-on payment duration for almost all applicants, as only those technologies that receive FDA marketing authorization in April would be eligible for 3 years of new technology add-on payments, shortening the window from 3 months under the former policy (April 1 until July 1) to just 1 month (April 1 until May 1) ( 88 FR 58954 ). In response, we noted in that even under the former policy, not all applicants receive the full 3 years of new technology add on payments, and that there are many factors (including timing of interactions with the FDA and manufacturing readiness) that can delay a technology's approval by the FDA that would disrupt a technology's ability to receive the full 3 years of payment. However, we also noted the commenters' concerns regarding the shortened time period between April 1 and May 1 under the new policy and stated that we would consider for future rulemaking how we assess new technology add-on payment eligibility in the third year of newness, such as consideration of adjusting the April 1 cutoff to allow for a longer window of eligibility ( 88 FR 58955 ).
In the proposed rule ( 89 FR 36136 through 36137 ), after further consideration of commenters' concerns that the policy we finalized in the FY 2024 IPPS/LTCH PPS final rule may limit the ability of new technology add-on payment applicants to be eligible for a third year of new technology add-on payments due to the shortened timeframe between April 1 and May 1, we stated that we agreed that there may be merit to modifying our current 6-month guideline to provide additional flexibility for applications submitted in accordance with this new policy. While technologies that are FDA approved or cleared in April, and technologies with a documented delay in availability on the U.S. market such that the product's entry onto the U.S. market falls within the second half of the fiscal year, would still be eligible for a third year of new technology add-on payments under current policy, we agreed that the change in the FDA marketing authorization deadline from July 1 to May 1 may limit the ability of new technology add-on payment applicants to be eligible for 3 years of new technology add-on payments. Therefore, we proposed to change the April 1 cutoff for determining whether a technology would be within its 2- to 3-year newness period when considering eligibility for new technology add-on payments. We stated that we believed this proposed change would continue the flexibility applicants had with respect to when they apply to FDA and when they apply for new technology add-on payment, while preserving a predictable and consistent payment methodology for new technologies throughout the fiscal year.
Specifically, we proposed that beginning with new technology add-on payments for FY 2026, in assessing whether to continue the new technology add-on payments for those technologies that are first approved for new technology add-on payments in FY 2025 or a subsequent year, we would extend new technology add-on payments for an additional fiscal year when the 3-year anniversary date of the product's entry onto the U.S. market occurs on or after October 1 of that fiscal year. We proposed that this policy change would become effective beginning with those technologies that are initially approved for new technology add-on payments in FY 2025 or a subsequent year to allow additional flexibility for those applications for new technologies which were first subject to the change in the deadline for FDA marketing authorization from July 1 to May 1. Therefore, for technologies that were first approved for new technology add-on payments prior to FY 2025, including for technologies we determine to be substantially similar to those technologies, we stated we would continue to use the midpoint of the upcoming fiscal year (April 1) when determining whether a technology would still be considered “new” for purposes of new technology add-on payments. Similarly, we also proposed that beginning with applications for new technology add-on payments for FY 2026, we would use the start of the fiscal year (October 1) instead of April 1 to determine whether to approve new technology add-on payment for that fiscal year.
We sought public comment on our proposal to change the April 1 cutoff to October 1 for determining whether a technology would be within its 2- to 3-year newness period when considering eligibility for new technology add-on payments, beginning in FY 2026, effective for those technologies that are approved for new technology add-on payments starting in FY 2025 or a subsequent year.
Comment: Commenters were supportive of our proposal to use the start of the fiscal year, October 1, instead of April 1, to determine whether a product is within its 2-to-3-year newness period for new technology add-on payment, and requested that CMS finalize this proposal. Commenters stated that they appreciated CMS's acknowledgement that the FY 2024 policy change in the FDA marketing authorization deadline may limit the ability of new technology add-on payment applicants to be eligible for 3 years of new technology add-on payments, and stated that the proposal would improve the flexibility for applicants with respect to FDA timing. Commenters stated that this proposal provided a more balanced and appropriate evaluation of whether a technology qualifies for a 2-year or 3-year period of new technology add-on payment. Another commenter agreed that predictability is an important aspect of the prospective payment system methodology and appreciated ( print page 69240) CMS's application of consistent payment methodology for new technologies throughout the fiscal year.
Commenters stated that the policy would allow more innovative technologies to receive new technology add-on payment for a third year. Commenters further stated that this would help to incentivize new treatment options and ensure continued access to breakthrough and life-saving technologies for Medicare beneficiaries and their providers during the first years of product availability and with substantially reduced payment disincentives inherent in how IPPS payment rates are established. Commenters stated this would help more hospitals offer these technologies, which would improve the claims data for MS-DRG assignments and ensure appropriate MS-DRG recalibration following the new technology add-on payment period. Commenters were also supportive of the increased time for cost data collection, stating that it would be particularly helpful in accruing data for low-volume technologies and/or those with a significant delay between their newness date and the timeframe when claims began accumulating in the data.
Response: We thank commenters for their support of our proposal, and agree that this proposal would provide additional flexibility for new technology add-on payment applications submitted in accordance with the change in the FDA marketing authorization deadline.
Comment: Commenters also requested that CMS provide additional flexibility to guarantee a third year of new technology add-on payment for all technologies regardless of when they receive FDA marketing authorization. A commenter further stated that this would maximize patient access to future CAR T-cell therapies and other important technologies advancing personalized medicine. Other commenters requested that CMS guarantee a third year of new technology add-on payment for specific technologies, such as CAR-T cell therapies or cell and gene therapies. A commenter further explained that new cell and gene therapies technologies might take several months to gain market availability, post-FDA approval. The commenter stated that personalized medical technologies like cell and gene therapies are uniquely developed for each patient and, therefore, often have a delay in actual availability of a product after FDA approval or clearance, due to the time needed for development and administration. The commenter stated that ensuring a third year of add-on payment for using cell and gene therapies would accommodate the time required for patient-specific development while advancing patient access to innovative technologies. A commenter also requested that CMS create a five-year add-on payment period for autologous gene and cell therapy products that qualify for new technology add-on payment.
Some commenters stated that this proposed policy would leave CMS with unreliable claims data for rate-setting at the expiration of new technology add-on payment because CMS uses MedPAR data from two years prior to the applicable fiscal year for ratesetting, and for services that use new technology with only 2 years of new technology add-on payment status, CMS is effectively relying on the first year of data to set rates for the first fiscal year following the end of new technology add-on payment status. Commenters stated that the first year of new technology add-on payment is typically when a technology is first coming to market and there are typically fewer claims reflecting use of the technology, especially for technologies that may not have received new technology add-on payment until a year or more after their FDA approval date, resulting in a small number of claims that may not be stable or reliable. Commenters stated that by granting all new technologies 3 years of new technology add-on payment status, CMS can ensure sufficient reliable claims data for ratesetting at the end of new technology add-on payment status, and that the applicable statute and regulations discuss newness in relation to the availability of claims data.
Some commenters believed that this and other proposals did not adequately address what they described as the consistent and severe underfunding of gene therapies and breakthrough drugs. A commenter stated that although this change would enable more products to qualify for add-on payments during the third year, it did not guarantee that these products would benefit from a full three years of new technology add-on payment. The commenter stated that the proposal narrowly addresses the issue of timing but fails to expand the overall eligibility window in a meaningful way that would support a greater number of innovative products.
A few commenters further stated that the proposal did not actually help the technologies impacted by the July 1 to May 1 FDA marketing authorization deadline change, as the only products for which this proposal would allow for an additional third year of new technology add-on payment are those products approved between October 1 and March 31. The commenters stated that products approved April 1 to May 1 were not affected by the change in the FDA approval deadline and would not be impacted by this proposal. The commenters stated that products approved May 2 to July 1 lost a third year of new technology add-on payment status when the July 1 to May 1 rule was finalized and would not gain back the third year of new technology add-on payment status under CMS's proposal because their FDA approval date occurs before October 1 of what would be the third fiscal year. The commenters also stated that products approved July 1 to October 1 would continue to remain ineligible for the third year of new technology add-on payment. Therefore, commenters stated that granting all new technologies three years of payment would rectify the problem for technologies approved between May 1 and July 1.
Some commenters also stated that the statute did not mention the FDA approval date, nor was there a statutory preclusion from granting all products a third year of payment. Another commenter asserted that CMS could statutorily grant three full years of new technology add-on payment status to new technologies based on the effective date of the ICD-10-PCS code that describes the service/technology, which could be set at October 1, the date that new technology add-on payment status begins, and that this approach was in line with how CMS makes passthrough payment under the OPPS. The commenter explained that FDA approvals can arbitrarily occur in the second half of the fiscal year, rather than in the first half of the following fiscal year, and that it is challenging to time FDA application submissions to try to get approval in the first half of the fiscal year, and that new therapeutic products approved between April 2 and September 30 face potentially slow uptake given the up to 17 months before new technology add-on payment adjustments would be effective.
Response: We thank commenters for their feedback. However, we do not agree that we should guarantee a third year of new technology add-on payment for all technologies regardless of when they receive FDA marketing authorization. The intent of our policy was not to ensure that more technologies would receive three years of new technology add-on payments, but rather to address how the change in the FDA marketing authorization deadline, effective beginning with new technology add-on payment applications for FY 2025, may limit the ability of new technology add-on payment applicants to be eligible for a third year of new technology add-on ( print page 69241) payments under our general practice for determining whether to extend the payment for an additional fiscal year, as described previously in this rule. We recognize that there may be a small subset of technologies that would not benefit from this proposal.
As we stated in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58955 ), section 1886(d)(5)(K)(ii) of the Act establishes a period of not less than 2 years and not more than 3 years for the collection of data with respect to the costs of new services or technologies; a full 3 years is not required. As we had stated, consistent with the statute and our implementing regulations, a technology is no longer considered “new” once it is more than 2 to 3 years old, irrespective of how frequently the medical service or technology has been used in the Medicare population ( 70 FR 47349 ). As such, once a technology has been available on the U.S. market for more than 2 to 3 years, we consider the costs to be included in the MS-DRG relative weights regardless of whether the technology's use in the Medicare population has been frequent or infrequent. Therefore, we do not believe that 2 years' worth of data would be insufficient to inform rate-setting for the inpatient setting.
We also disagree that this proposed policy would leave CMS with unreliable claims data for ratesetting for technologies that would be on the market for a year or more before they could begin receiving new technology add-on payment and receive payment for at most two years based on their FDA marketing authorization dates. As described in the FY 2005 IPPS final rule ( 69 FR 49003 ), even if a technology does not receive new technology add-on payments, CMS continues to pay for new technologies through the regular payment mechanism established by the DRG payment methodology. In addition, the costs incurred by the hospital for a case are evaluated to determine whether the hospital is eligible for an additional payment as an outlier case. This additional payment is designed to protect the hospital from large financial losses due to unusually expensive cases. Any eligible outlier payment is added to the DRG-adjusted base payment rate ( 88 FR 58648 ). We further note that whether a technology receives new technology add-on payments or not does not affect coverage of the technology or the ability for hospitals to provide a technology to patients where appropriate. Therefore, data reflecting the costs of a new technology begin to become available for recalibration of the DRG weights starting from when the technology became available on the U.S. market. As we previously stated, the newness period does not necessarily start with the approval date for the medical service or technology and does not necessarily start with the issuance of a distinct code. Instead, it begins with availability of the product on the market, which is when data become available ( 69 FR 49003 ).
Comment: Some commenters also requested that CMS make the proposal effective immediately. Commenters recommended that CMS apply the proposal to other technologies that are currently receiving new technology add-on payment, such as to those for which the new technology add-on payment is set to expire in FY 2024, to those that first received new technology add-on payment for FY 2024, and to new technology add-on payment applications for FY 2025 that are determined to be substantially similar to technologies first approved for new technology add-on payments prior to FY 2025. A commenter stated that applying the proposal to technologies that are currently receiving new technology add-on payment that would qualify for a third year under the change would apply to only three technologies that CMS proposed to discontinue in FY 2025, and stated this would better serve Medicare beneficiaries, improve the quality of data, and capture more mature usage patterns for LIVTENCITY TM and other affected products to ensure more robust claims data for ratesetting.
Another commenter further stated that CMS should finalize this proposal such that technologies that received a second year of new technology add-on payment status in FY 2024, receive a third year of new technology add-on payment status in FY 2025; technologies that received their first year of new technology add-on payment status in FY 2024 would receive new technology add-on payment through FY 2026; and technologies first approved for new technology add-on payment in FY 2025 and future years are automatically eligible for three full years of new technology add-on payment. The commenter suggested that if CMS were to move forward with its current proposal, it should be effective for applicants that first receive new technology add-on payment starting in FY 2024, rather than FY 2025, as otherwise CMS would be relying on the FY 2024 claims data for rate-setting in FY 2026, and there may be a low number of claims with significant variability in reported charges resulting in less reliable ratesetting.
Response: We thank the commenters for their comments. As we previously noted, the intent of our proposal was not to ensure that more technologies would receive three years of new technology add-on payments. We had stated that, after further consideration of commenters' concerns that the policy we finalized in the FY 2024 IPPS/LTCH PPS final rule may limit the ability of new technology add-on payment applicants to be eligible for a third year of new technology add-on payments due to the shortened timeframe between April 1 and May 1, we agreed that there may be merit to modifying our current 6-month guideline to provide additional flexibility for applications submitted in accordance with this new policy ( 89 FR 36136 ). Applications submitted for new technology add-on payment prior to FY 2025 were not subject to the change in the deadline for FDA marketing authorization from July 1 to May 1 as finalized in the FY 2024 IPPS/LTCH PPS final rule. Similarly, for technologies that we determine to be substantially similar to technologies first approved for new technology add-on payments prior to FY 2025, under our longstanding policy, if substantially similar technologies are submitted for review in different (and subsequent) years, we evaluate and make a determination on the first application and apply that same determination to the second application ( 85 FR 58679 ), and we use the earliest market availability date submitted as the beginning of the newness period for both technologies ( 87 FR 48925 ). Therefore, we disagree with commenters that this proposal should be effective immediately or applied to other technologies outside of our proposal.
Comment: Commenters offered additional suggestions as to how CMS could establish the newness period. A commenter stated that the period of “newness” for a technology or medical service to receive add-on payments is based generally on the date of FDA approval/clearance, which is not necessarily the date of commercial availability. Commenters suggested that CMS consider the date of assignment of a new ICD-10 code when determining a technology's date of commercial availability, with a commenter stating that it would be consistent with CMS's stated policy that a technology may continue to be considered “new” for new technology add-on payment purposes “within 2 or 3 years after the point at which data begin to become available reflecting the inpatient hospital code assigned to the new service or technology.” A commenter believed there was merit in considering the newness period to start on the date ( print page 69242) of first sale or upon issuance of an ICD-10-PCS code, as otherwise, a technology may not be commercially available or without an ICD-10-PCS code that allows the collection of accurate cost data as the new technology is not identifiable in any claims data. A commenter also requested that CMS start the newness period at the date of first administration of the technology for autologous gene and cell therapy products that qualify for new technology add-on payment.
Response: As we have stated in prior rulemaking, the newness period does not necessarily start with the approval date for the medical service or technology and does not necessarily start with the issuance of a distinct code. Instead, it begins with availability of the product on the market, which is when data become available. We have consistently applied this standard, and believe that it is most consistent with the purpose of new technology add-on payments ( 69 FR 49003 ).
We have also stated that for technologies that do not have a unique ICD-10 code, while it may be impossible to identify when a particular product was used because there is no unique code to identify it amongst other products in the category, the product is nonetheless used and paid for. In addition, hospital charges reflect the services provided to patients receiving the new service or device whether or not a specific code is assigned. Therefore, data containing payments for these new technologies are already in our MedPAR database and when DRG recalibration occurs these costs are accounted for. Furthermore, assignment of new codes can occur for many reasons other than the introduction of new procedures and technologies. For example, new codes can simply reflect more refined and discriminating descriptions of existing procedures and technologies ( 69 FR 49003 ).
We also disagree that the newness period should start on the date of the first sale or at the first administration of a technology. As we previously noted, while CMS may consider a documented delay in a technology's availability on the U.S. market in determining when the newness period begins, under our historical policy, we do not consider how frequently the medical service or technology has been used in our determination of newness ( 70 FR 47349 ). Consistent with the statute, a technology no longer qualifies as new once it is more than 2 to 3 years old irrespective of how frequently it has been used in the Medicare population.
Therefore, after consideration of the comments received, and for the reasons discussed previously and in the FY 2025 IPPS/LTCH PPS proposed rule, we are finalizing our proposal that, beginning with new technology add-on payments for FY 2026, in assessing whether to continue the new technology add-on payments for those technologies that are first approved for new technology add-on payments in FY 2025 or a subsequent year, we will extend new technology add-on payments for an additional fiscal year when the 3-year anniversary date of the product's entry onto the U.S. market occurs on or after October 1 of that fiscal year. This policy change will become effective beginning with those technologies that are initially approved for new technology add-on payments in FY 2025 or a subsequent year. For technologies that were first approved for new technology add-on payments prior to FY 2025, including for technologies we determine to be substantially similar to those technologies, we will continue to use the midpoint of the upcoming fiscal year (April 1) when determining whether a technology would still be considered “new” for purposes of new technology add-on payments. Similarly, we are also finalizing that beginning with applications for new technology add-on payments for FY 2026, we will use the start of the fiscal year (October 1) instead of April 1 to determine whether to approve new technology add-on payment for that fiscal year.
As previously discussed, in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ), we finalized that beginning with the new technology add-on payment applications for FY 2025, for technologies that are not already FDA market authorized for the indication that is the subject of the new technology add-on payment application, applicants must have a complete and active FDA market authorization request at the time of new technology add-on payment application submission, and must provide documentation of FDA acceptance or filing to CMS at the time of application submission, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. See § 412.87(e) and further discussion in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ).
As we discussed further in the FY 2024 IPPS/LTCH PPS final rule, the documentation of FDA acceptance or filing of a marketing authorization request must be provided at the time of new technology add-on payment application, and be consistent with the type of FDA marketing authorization the applicant has submitted to FDA. We stated that we only accept new technology add-on payment applications once FDA has received all of the information necessary to determine whether it will accept (such as in the case of a 510(k) premarket submission or De Novo Classification request) or file (such as in the case of a PMA, NDA, or BLA) the application as demonstrated by documentation of the acceptance/filing that is provided by FDA. The applicant is required to submit documentation with its new technology add-on payment application to demonstrate that FDA has determined that the application is sufficiently complete to allow for substantive review by the FDA ( 88 FR 58955 ).
We also explained that, for the purposes of new technology add-on payment applications, we consider an FDA marketing authorization application to be in an active status when it has not been withdrawn, is not the subject of a Complete Response Letter or final decision from FDA to refuse to approve the application, and is not on hold ( 88 FR 58955 through 58956 ).
We stated in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36137 ) that, as noted in the FY 2024 IPPS/LTCH PPS final rule, we collaborated with FDA in developing the terminology used for purposes of this policy, and the intent behind using the terms we did was to ensure that the requirement could apply to and be inclusive of the various FDA applications and approval pathways for different types of drugs and devices. We stated in the proposed rule that, as such, we did not use terms defined in statute or existing regulations or terms defined by FDA ( 88 FR 58955 ). We stated that while FDA may consider an application for an FDA marketing authorization to be under active review despite a hold status, under our current policy we do not consider marketing authorization applications in a hold with FDA to be in an active status for the purposes of new technology add-on payment application eligibility. As discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58956 ) our intent with respect to considering applications that are on hold at the time of new technology add-on payment application submission to be inactive was to ensure that applicants are far enough along in the FDA review process that applicants would be able to reasonably provide sufficient information at the time of new technology add-on payment application submission for CMS to identify critical ( print page 69243) questions regarding the technology's eligibility for add-on payments and to allow the public to assess the relevant new technology evaluation criteria in the proposed rule. We stated that, as noted in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58956 ), we have received applications over the years for technologies that are in a hold status with up to 360 days allowed for submission of additional information.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36137 through 36138 ), we stated that we also recognized that applications for FDA marketing authorization may go in and out of a hold status at various stages during the FDA application process and for various reasons. The maximum length of a hold status can vary based on the FDA approval pathway, such that the time remaining for an applicant to resolve the hold may vary from days to several months after the start of the new technology add-on payment application cycle, depending on the FDA pathway, reason(s) for the hold status, and how the timing of the hold coincides with the annual new technology add-on payment application submission date. Additionally, FDA may need to issue secondary letters of request for additional information, often depending on the quality of initial response from the applicant. Accordingly, we stated that while we continued to believe that an application that is in a hold status with FDA pending additional information may lack critical information that is needed to evaluate whether the technology meets the eligibility criteria, we also recognized the variability in the reasons for a hold and the varying lengths of time for which an application can be on hold with FDA, such that some applicants may be farther along in the process to obtain FDA marketing authorization at the time of the hold.
Further, we stated that after further consideration, based on the variability in the timing of and reasons underlying hold statuses with FDA, we believed it was appropriate to propose to update our policy. Specifically, we proposed, beginning with new technology add-on payment applications for FY 2026, to no longer consider a hold status to be an inactive status for the purposes of eligibility for the new technology add-on payment. We stated we would continue to consider an application to be in an inactive status where it is withdrawn, the subject of a Complete Response Letter, or the subject of a final decision from FDA to refuse to approve the application. Because of the variety of circumstances for which a technology may be in a hold, as previously discussed, we noted that we may reassess this policy for future years, if finalized, based on ongoing experience.
We invited public comments on our proposal to no longer consider a hold status to be an inactive status for the purposes of eligibility for new technology add-on payment, beginning with new technology add-on payment applications for FY 2026.
Comment: Commenters overwhelmingly supported CMS's proposal to no longer consider a hold to be an inactive status for the purposes of new technology add-on payment eligibility. Commenters stated that the FDA application review process is dynamic and that applications may go in and out of a hold at various stages during the FDA application process and for various reasons including administrative reasons that might not necessarily be due to lack of critical information in the FDA application, such as user fee holds, incorrect eCopy, outdated submission templates, or omission of an administrative element, and that these holds may be resolved within days or months after the start of the new technology add-on payment application cycle. Commenters further stated that being on hold does not materially affect the ability for the technology to receive FDA authorization by the May 1 new technology add-on payment deadline as some applicants may be farther along in the process to obtain FDA marketing authorization at the time of the hold. Commenters also stated that they believe that this proposed change would enhance the predictability of the new technology add-on payment process and ensure that new technology add-on payment applications are not inadvertently pushed back to a later new technology add-on payment application cycle, even though FDA may have continued reviewing the product's marketing application, despite a hold at the time the product's new technology add-on payment application is submitted to CMS. Commenters concluded that finalizing this proposal would remove a significant barrier for applications that may be placed on a brief hold status and would be rendered ineligible for new technology add-on payments for a full year.
Response: We thank commenters for their support of our proposal to no longer consider a hold with FDA to be an inactive status for the purposes of eligibility for new technology add-on payment, beginning with new technology add-on payment applications for FY 2026.
Comment: Many commenters also requested that CMS reverse other aspects of the policy finalized in the FY 2024 IPPS/LTCH PPS final rule including the requirement for a complete and active FDA marketing authorization application request at the time of new technology add-on payment application, the FDA documentation requirement, and moving the FDA marketing deadline from May 1 back to July 1. The commenters stated that these requirements are already disqualifying applicants and therefore delaying beneficiary access to innovative technologies in contradiction of the new technology add-on payment program goals. The commenters further stated that reversing the policy completely would give new technology add-on payment applicants the most flexibility. A few commenters specified that it is especially critical that CMS reverse the FDA market authorization requirement for particular application types such as Breakthrough Devices or those undergoing rolling review or real-time oncology review (RTOR) to prevent financial barriers to adoption of these new technologies and other access delays, and because these applications could become complete shortly after the application deadline. Some commenters further stated that applications with rolling review or RTOR are reserved for therapies with Breakthrough Therapy or Fast Track designations, or for therapies likely to demonstrate substantial clinical improvement and CMS should therefore reverse its policy for these therapies. A commenter also stated concerns specifically with the exact type of FDA documentation required at the time of new technology add-on payment submission because they said it has limited forecasting of final FDA approval by May 1 for the new technology add-on payment applicant, and recommended CMS rescind the FDA documentation requirement.
Several commenters suggested that CMS should instead provide an alternate deadline to provide the necessary information regarding FDA marketing authorization, such as within 60 days after application submission, the December supplemental information deadline, or no earlier than March 1, and that CMS should make these changes via notice and comment rulemaking.
Several commenters made additional requests from CMS, if CMS were to decide not to reverse the policy. A commenter requested that CMS provide an analysis of the impact of the FDA submission/authorization requirements on new technology add-on payment application volume, CMS workload, and the average time between marketing authorization and new technology add- ( print page 69244) on payment availability for medical devices.
Response: We thank the commenters for sharing their concerns, as well as their suggestions and recommendations. CMS shares the goal of ensuring Medicare beneficiaries and their providers have access to new technologies. However, as described in the FY 2005 IPPS final rule ( 69 FR 49003 and 49009 ), patient access to these technologies should not be adversely affected if a technology does not qualify to receive new technology add-on payments, as CMS continues to pay for new technologies through the regular payment mechanism established by the MS-DRG methodology. In addition, the costs incurred by the hospital for a case are evaluated to determine whether the hospital is eligible for an additional payment as an outlier case. This additional payment is designed to protect the hospital from large financial losses due to unusually expensive cases. Any eligible outlier payment is added to the DRG-adjusted base payment rate ( 88 FR 58648 ). As noted in an earlier section, whether a technology receives new technology add-on payments or not does not affect coverage of the technology or the ability for hospitals to provide a technology to patients where appropriate. As stated in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58953 ), the new technology add-on payment application eligibility requirements related to FDA application status did not eliminate flexibilities built into the new technology add-on payment process, as FDA marketing authorization is not required at the time of application, and eligible applicants can continue to provide some information as it becomes available according to our standard processes (such as the December supplemental deadline and the public comment period). Although we continue to believe in providing maximum flexibility to applicants where feasible, the policy was put in place due to the increasing complexity and volume of applications lacking critical information that is needed to evaluate whether the technology meets the eligibility criteria at § 412.87(b), (c), or (d). As discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58949 ), in prior years, a significant number of applicants had submitted new technology add-on payment applications that resulted in information not being available for the proposed rule and during the comment period. Specifically, many applicants submitted new technology add-on payment applications prior to submitting a request to FDA for the necessary marketing authorization, and applicants have stated that information missing from their applications, which is needed to evaluate the technology for the add-on payment, will not become available until after submission to FDA. With regard to the alternative pathways, such applications may also be missing information that would help inform understanding of the details and interrelationship between the intended indication and FDA Breakthrough Device or QIDP designation, which is the basis for a product's eligibility for the alternative pathway. Ultimately, it is difficult for CMS to review and for interested parties to comment on a product that has not yet been submitted to FDA and for which FDA has not determined that the marketing authorization request is sufficiently complete to allow for substantive review by FDA (regardless of FDA Breakthrough Device designation, Breakthrough Therapy designation, Fast Track designation, or RTOR participation), as multiple sections of the new technology add-on payment applications lack preliminary information that is more likely to be available after an FDA submission. Public input is an important part of our assessment of whether a technology meets the new technology add-on payment criteria, particularly as technology becomes more complex and specialized. Thus, we believe that requiring applicants to have already submitted a marketing authorization request to FDA that FDA has determined is sufficiently complete to allow for substantive review by FDA at the time of submission of the new technology add-on payment application further increases transparency and improves the evaluation process, including the identification of critical questions in the proposed rule, particularly as the number and complexity of the applications have been increasing over time. We will therefore continue to require documentation of FDA acceptance (for a 510(k) premarket submission or De Novo Classification request) or FDA filing (for a PMA, NDA, or BLA) at the time of new technology add-on payment application submission, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. We still believe this approach provides the clearest and most effective means of documenting that the applicant has submitted a complete request to FDA ( 88 FR 58950 ). We continue to believe these policies facilitate a more transparent process that will improve public engagement and help improve and streamline our review processes. Many of these products are novel and complex, and CMS has a responsibility to appropriately and thoroughly review applications for eligibility for new technology add-on payments against our established eligibility criteria. As noted in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58958 ), CMS will require documentation demonstrating that FDA has determined that the marketing authorization request is sufficiently complete to allow for substantive review by FDA ( e.g., documentation of FDA acceptance or FDA filing, depending on the type of FDA marketing authorization application the applicant has submitted to FDA) at the time of submission of the new technology add-on payment application. We have not accepted and will not accept documentation in which the date that FDA made the determination to accept (for a 510(k) premarket submission or De Novo Classification request) or file (for a PMA, NDA, or BLA) the request occurred after new technology add-on payment application submission; such documentation could not have been provided at the time of new technology add-on payment application submission and therefore does not meet the requirement. Further, we note that while documentation of FDA acceptance/filing may also include the date of submission of the FDA marketing authorization request, for new technology add-on payment purposes this is not the date on which FDA determined the request is sufficiently complete for substantive review, and therefore, this does not meet the new technology add-on payment application FDA status requirement at § 412.87(e)(2). For these reasons, we are not reversing other aspects of the policy finalized in the FY 2024 IPPS/LTCH PPS final rule.
Comment: A few commenters expressed concern about applications for technologies that were determined to be ineligible for consideration for new technology add-on payments for FY 2025 at the time of application for new technology add-on payments. The commenters were concerned about the impact that the ineligibility determination would have on Medicare beneficiaries' access to these innovative technologies in the upcoming year, as well as the financial viability of these technologies. Some of the commenters suggested that CMS provide mitigating intervention for technologies that were found ineligible for new technology add-on payment consideration in FY 2025, such as reversing the ineligibility ( print page 69245) and making an interim decision determination subject to public comment regarding overall qualification in this final rule using the “good cause” exception as provided in the APA; [ 183 ] extended eligibility to three years of new technology add-on payments; or extension of an additional year of new technology add-on payments following review in FY 2026.
Response: We thank the commenters for their comments and recommendations. We note that, as described previously, patient access to these technologies should not be adversely affected if a technology does not qualify to receive new technology add-on payments, as CMS continues to pay for new technologies through the regular payment mechanism established by the MS-DRG methodology. In addition, and as previously noted, a hospital may be eligible for additional payment for outlier cases. As also previously noted, whether a technology is approved for new technology add-on payments does not affect coverage of the technology or the ability for hospitals to provide a technology to patients where appropriate. We evaluated all applications for FY 2025 that were submitted by the new technology add-on payment deadline under the applicable eligibility requirements, and we will continue to do so for applications that are submitted or resubmitted for FY 2026. We further note that submission of a new technology add-on payment application does not guarantee that a technology will be approved for a new technology add-on payment.
Comment: A commenter stated that while this flexibility is an improvement, it applies mainly to devices and does not fully address challenges with CMS's new requirements for a “complete and active” FDA market authorization request. The commenter encouraged CMS to further clarify this language to ensure the gamut of personalized medicine treatments and technologies remain eligible for new technology add-on payments and reach patients who need them, without creating further delays in the availability of new technology add-on payment status.
Response: We thank the commenter for their comment. As discussed previously, the intent behind using the terminology we did was to ensure that the requirement could apply to and be inclusive of the various FDA applications and approval pathways for different types of drugs and devices. We disagree with the commenter's assessment that this flexibility applies mainly to devices. We note that our current hold policy applies to all technologies, irrespective of category (drugs, devices) or pathway (alternative, traditional). Regarding the commenter's request for CMS to further clarify the requirements for a “complete and active” FDA market authorization request, we note that, as discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58948 through 58958 ), we consider an FDA marketing authorization application to be “complete” when the full application has been submitted to FDA (including all modules or all information following a rolling review or RTOR, where relevant) and FDA has provided documentation of acceptance (for a 510k application or De Novo Classification request) or filing (for a PMA, NDA, or BLA) to the applicant indicating that FDA has determined that the application is sufficiently complete to allow for substantive review by FDA. Applicants are required to provide this documentation of FDA acceptance (for a 510k application or De Novo Classification request) or filing (for a PMA, NDA, or BLA) of the request to CMS at the time of application submission, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. Additionally, as noted in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58955 through 58956 ), for the purposes of new technology add-on payment applications, we consider an FDA marketing authorization application to be in an “active” status when the application has been determined by FDA to be sufficiently complete to permit substantive review by FDA, and when it is not in an “inactive” status at the time of new technology add-on payment application submission. We further note that “active” FDA status for the purposes of new technology add-on payment application eligibility begins once FDA has determined that the application is sufficiently complete to allow for substantive review by FDA, which as described earlier in this section, applicants must demonstrate at the time of new technology add-on payment application submission by providing FDA's acceptance (for a 510k application or De Novo Classification request) or filing (for a PMA, NDA, or BLA) of the request, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. We continue to consider an application to be in an inactive status where it is withdrawn, the subject of a Complete Response Letter, or the subject of a final decision from FDA to refuse to approve the application.
After consideration of the public comments we received, we are finalizing our proposal that, beginning with new technology add-on payment applications for FY 2026, we will no longer consider an FDA hold to be an inactive status for the purposes of eligibility for the new technology add-on payment for technologies that are not already FDA market authorized for the indication that is the subject of the new technology add-on payment application. As previously noted, because of the variety of circumstances for which a technology may be on hold, we may reassess this policy for future years based on ongoing experience.
As discussed previously in this section, section 1886(d)(5)(K)(ii)(I) of the Act specifies that a new medical service or technology may be considered for a new technology add-on payment if, based on the estimated costs incurred with respect to discharges involving such service or technology, the DRG prospective payment rate otherwise applicable to such discharges under this subsection is inadequate. Under our current policy, as set forth in § 412.88(b)(2), unless the discharge qualifies for an outlier payment, the additional Medicare payment will be limited to the full MS-DRG payment plus 65 percent (or 75 percent for a medical product designated by the FDA as a Qualified Infectious Disease Product [QIDP] or approved under FDA's Limited Population Pathway for Antibacterial and Antifungal Drugs [LPAD]) of the estimated costs of the new technology or medical service.
Since establishing the new technology add-on payment, we have been cautious about increasing the new technology add-on payment percentage. As stated in the May 4, 2001 proposed rule ( 66 FR 22695 ), we believe limiting the new technology add-on payment percentage would provide hospitals an incentive for continued cost-effective behavior in relation to the overall costs of the case. In the FY 2020 IPPS/LTCH PPS final rule, in adopting the general increase in the new technology add-on payment percentage from 50 percent to 65 percent, we stated that we believed that 65 percent would be an incremental increase that would reasonably balance the need to maintain the incentives inherent to the prospective payment system while also encouraging the development and use of new ( print page 69246) technologies. We continue to believe that it is important to balance these incentives in assessing any potential change to the new technology add-on payment calculation.
In the FY 2020 IPPS/LTCH PPS final rule, we also finalized an increase in the new technology add-on payment percentage for QIDPs from 65 percent to 75 percent. We stated that we shared commenters' concerns related to antimicrobial resistance and its serious impact on Medicare beneficiaries and public health overall. We noted that the Centers for Disease Control and Prevention (CDC) described antimicrobial resistance as “one of the biggest public health challenges of our time.” We stated that we believe that Medicare beneficiaries may be disproportionately impacted by antimicrobial resistance due in large part to the unique vulnerability to drug-resistant infections (for example, due to age-related and/or disease-related immunosuppression, greater pathogen exposure via catheter use) among individuals aged 65 or older. We further stated that antimicrobial resistance results in a substantial number of additional hospital days for Medicare beneficiaries, resulting in significant unnecessary health care expenditures.
To address the continued issues related to antimicrobial resistance resulting in a substantial number of increased hospital days and significant unnecessary health care expenditures for Medicare beneficiaries, in the FY 2021 IPPS/LTCH PPS final rule, we finalized a proposal to expand the alternative new technology add-on payment pathway for QIDPs to include products approved under the LPAD pathway and to increase the maximum new technology add-on payment percentage for a product approved under FDA's LPAD pathway, from 65 percent to 75 percent, consistent with the new technology add-on payment percentage for a product that is designated by FDA as a QIDP, beginning with discharges occurring on or after October 1, 2020 ( 85 FR 58739 ).
In the proposed rule ( 89 FR 36138 through 36139 ), we stated that since finalizing our current policy for QIDPs and LPADs, we continued to receive feedback from interested parties regarding the adequacy of new technology add-on payments for certain categories of technologies, including cell and gene therapies to treat sickle cell disease (SCD). We stated that although we still believe it is prudent to proceed cautiously with increasing the new technology add-on payment percentage, we recognize that SCD, the most common inherited blood disorder, has historically had limited treatment options. In addition, hospitalizations and other health episodes related to SCD cost the health system $3 billion per year. [ 184 ] We further noted that the Administration has identified a need to address SCD and has made a commitment to improving outcomes for patients with SCD by facilitating access to cell and gene therapies that treat SCD. [ 185 ]
Accordingly, we stated that we believe that further facilitating access to these gene therapies for Medicare beneficiaries with SCD may have the potential to simultaneously improve the health of impacted Medicare beneficiaries and potentially lead to long-term savings in the Medicare program. We also noted that some gene therapies that treat SCD are among the costliest treatments to date, and we were concerned about a hospital's ability to sustain a potential financial loss to provide access to such treatments. As we discussed when we increased the new technology add-on payment for QIDPs in the FY 2020 IPPS/LTCH PPS final rule and products approved under FDA's LPAD in the FY 2021 IPPS/LTCH PPS final rule from 65 percent to 75 percent, we stated that we believe that it may be appropriate to increase the maximum add-on amount in limited cases where the current new technology add-on payment does not provide a sufficient incentive for the use of a new technology, which we believed may be the case for gene therapies that treat SCD. Accordingly, and consistent with our new technology add-on payment policy for products designated by the FDA as a QIDP or LPAD, we stated that we believe there would be merit in also increasing the new technology add-on payment percentage for gene therapies that are indicated and used for the treatment of SCD to 75 percent.
Therefore, we proposed that, subject to our review of the new technology add-on payment eligibility criteria, for certain gene therapies approved for new technology add-on payments in the FY 2025 IPPS/LTCH PPS final rule for the treatment of SCD, effective with discharges on or after October 1, 2024 and concluding at the end of the 2- to 3-year newness period for such therapy, if the costs of a discharge (determined by applying CCRs as described in § 412.84(h)) involving the use of such therapy for the treatment of SCD exceed the full DRG payment (including payments for IME and DSH, but excluding outlier payments), Medicare would make an add-on payment equal to the lesser of: (1) 75 percent of the costs of the new medical service or technology; or (2) 75 percent of the amount by which the costs of the case exceed the standard DRG payment. We stated that, if finalized, these payment amounts would only apply to any gene therapy indicated and used specifically for the treatment of SCD that CMS determines in the FY 2025 IPPS/LTCH PPS final rule meets the criteria for approval for new technology add-on payment. We also proposed to add new § 412.88(a)(2)(ii)(C) and § 412.88(b)(2)(iv) to reflect this proposed change to the calculation of the new technology add-on payment amount, beginning in FY 2025 and concluding at the end of the 2- to 3-year newness period for each such therapy. With this incremental increase, we stated that we believe hospitals would continue to have an incentive to balance the desirability of using the new technology for patients as medically appropriate while also maintaining an incentive for continued cost-effective behavior in relation to the overall costs of the case.
We invited public comments on this proposal to temporarily increase the new technology add-on payment percentage to 75 percent for a gene therapy that is indicated and used for the treatment of SCD as described previously. We also sought comment on whether we should make this proposed 75 percent add-on payment percentage available only to applicants that meet certain additional criteria, such as attesting to offering and/or participating in outcome-based pricing arrangements with purchasers (without regard to whether the specific purchaser availed itself of the outcome-based arrangements), or otherwise engaging in behaviors that promote access to these therapies at lower cost.
Comment: Some commenters were supportive of the proposal. Commenters were pleased that CMS is making efforts to improve access to this rapidly advancing area of medicine, and stated that increasing the add-on percentage to 75 percent for gene therapies for sickle cell disease reflects a targeted approach aligned with the Cell and Gene Therapy Access Model. Commenters stated that they appreciate the Agency's commitment to the SCD patient population which they stated has historically been marginalized, as ( print page 69247) outlined in the SCD Action Plan. The commenters stated that the proposed payment policy represents a meaningful change for Medicare beneficiaries, as the increased add-on payment will incentivize hospital adoption and expand patient access to these critical technologies. A commenter stated that in addition to improving the lives of patients, investing in therapies that reduce the need for chronic care and, especially, costly hospitalizations for SCD patients has the potential for significant long-term savings for the Medicare program. Another commenter stated that incentivizing use of SCD gene therapies will reduce associated care costs for patients, providers, and payers by preventing the need for future medical services.
Most of the commenters supporting the policy stated that they believed CMS should finalize as proposed, and also requested that CMS extend the policy further in various ways, while some stated they would support the proposal with varied modifications. Many of the commenters requested that CMS expand or modify the proposal to increase the add-on percentage to other therapies in addition to gene therapies treating SCD, stating that increasing the percentage allows for hospital adoption of groundbreaking therapies and advances the new technology add-on payment program's objective for expanding patient access to innovative new technologies. A commenter stated that while the focus on SCD is commendable, the narrow application of the proposal to specific therapies, and potentially only those engaged in value-based purchasing agreements, indicates a limited scope of financial support.
A few of the commenters recommended that all technologies that meet the new technology add-on payment eligibility criteria should receive 75 percent. A commenter stated that hospitals find the current 65 percent add-on payment insufficient to cover the costs of using new technologies, and that 75 percent would mitigate losses and encourage adoption of new technologies. The commenter further stated that an analysis demonstrated that hospitals receive millions in outlier payments on the same cases that receive new technology add-on payment payments, highlighting how inadequate the new technology add-on payment is. Another commenter stated that a consistent payment percentage for all therapies would eliminate inequity for manufacturers, improve transparency, and reduce payment confusion for hospitals. One of these commenters stated that this piecemeal approach (that is, highlighting one group of technologies for a higher payment percentage) fails to recognize the financial difficulties that hospitals face in adopting other innovative technologies not yet reflected in Medicare rates. Other commenters believed that by having a higher payment percentage for select groups of technologies (such as SCD therapies or QIDP/LPADs), CMS is making a value judgement that these therapies are more valuable than other qualifying technologies or medical conditions, and that this is beyond the purview of CMS and not the intent of the new technology add-on payment program. The commenters stated that while each technology has varying levels of impact on the Medicare population, once CMS has established that a technology meets the new technology add-on payment criteria, all drugs and devices should be treated equally. A few commenters also requested that CMS provide details regarding any criteria that CMS uses to determine which categories of technologies warrant increased payment levels, as well as the appropriate payment level for each class of technologies via rulemaking to allow for stakeholder input. A commenter further requested that as an alternative to raising the payment percentage to 75 percent for all technologies, CMS should, at a minimum, establish a process and criteria by which manufacturers can request an enhanced new technology add-on payment percentage. The commenter stated that it is difficult to discern a clear and consistent set of criteria that were used to determine which technologies should receive enhanced payment from discussions of these decisions in the Federal Register , and whether the decisions resulted from manufacturer/stakeholder requests or from internal CMS requests. The commenter further stated that it believes that the lack of clear process and criteria for these decisions creates risk that the decisions will be viewed as arbitrary and capricious.
A few commenters requested that CMS extend the 75 percent to therapies with regenerative medicine advanced therapy (RMAT) or Breakthrough Therapy designations; to those targeting rare diseases, unmet needs, or vulnerable groups; or to other transformative therapies that Medicare beneficiaries may have difficulty accessing. Some commenters requested that CMS extend an increased new technology add-on payment percentage to align with other Administration priorities, such as hospital preterm deliveries, very low birth weight babies, other critically ill pediatric patients, and maternal health technologies. A commenter requested that CMS extend the increased maximum percentage to transformative therapies as opportunities arise, and that CMS monitor when additional increases higher than 75 percent are warranted.
Some of the commenters stated that all cell and gene therapies should receive the increase to 75 percent, stating that CMS's stated reasons for the proposal apply to these therapies as well, and that cell and gene therapies may pose similar beneficiary access challenges based on inadequate payment. Commenters cited as their rationales that these therapies are generally treating small patient populations, rare disease, certain cancers, underserved populations, and/or orphan indications with significant unmet medical need. A commenter explained that cell and gene therapies often require complex manufacturing processes, specialized infrastructure, and intensive monitoring, and that these costs are embedded in the cost of these products, making them more costly. The commenter added that these therapies often have no historical claims data to characterize resource use associated with the inpatient admissions since patients may not even have been admitted previously due to a lack of treatment options (as compared to other types of new technology add-on payment technologies that represent improvements on or alternatives to existing treatments), and that therefore new technology add-on payment is needed to compensate for the absence of any costs from the rate setting methodology. Another commenter added that cell and gene therapies cause a significant strain on hospital financial resources; even with a new technology add-on payment, these therapies are more likely than other inpatient stays to qualify for outlier payments. A commenter stated that there is a need to incentivize newly approved high-cost, high-reward cellular and gene therapies through new technology add-on payment as there continues to be insufficient inpatient reimbursement for autologous cellular therapies, like CAR T-cell therapies. Commenters stated that inpatient stays with cell and gene therapies are inadequately paid, even with new technology add-on payments, which could dissuade hospitals from providing these therapies. A commenter specified further that particularly cell and gene therapies that treat other inherited, debilitating, and under-treated conditions like hemophilia and Duchenne muscular dystrophy (DMD) ( print page 69248) should receive this increase, stating that the significant costs and limited therapies to treat these patients justify an increase above other new technology add-on payment applicants. Commenters also requested that therapies that share characteristics with gene therapies for SCD should be included in the proposal, including the significant up-front costs to hospitals and significant reduction in chronic care needs and costs to the Medicare program on an ongoing basis. A commenter stated that reductions in chronic care costs accrue to Medicare rather than providers, and new technology add-on payment is a pathway to bridge the gap by providing support for hospitals that incur the up-front cost of purchasing these therapies. Another commenter also stated that increasing the new technology add-on payment percentage for cell and gene therapies would, in addition to supporting Medicare beneficiary access to these therapies, be beneficial to Medicaid patients as many are dually eligible.
Several commenters requested that CMS expand its proposal to include transfusion-dependent beta thalassemia (TDT). Commenters questioned why this proposal from CMS only applied to gene therapies for SCD and did not include FDA-approved gene therapies for TDT, which have the same public policy, pricing, and access concerns as SCD, and also have no curative alternatives. A commenter further stated that like SCD, historical treatment options for TDT also carry numerous limitations resulting in significantly under-served patient populations. The commenter also stated that extending enhanced new technology add-on payment to gene therapies used for TDT would be likely to have a minimal impact to the IPPS from a budget neutrality perspective because there was only an estimated 1,000 to 1,500 individuals in the U.S. living with TDT, with a far smaller proportion of Medicare-eligible individuals.
Response: We appreciate the commenters' feedback. We thank commenters for their support of the proposal. We continue to believe that the policy aligns with the Administration-identified commitment to improving outcomes for patients with SCD by facilitating access to gene therapies that treat SCD, [ 186 ] and also balances the need to maintain the incentives inherent to the prospective payment system.
With regard to commenters requesting that the proposal include different groups of therapies or those with particular designations, or all therapies approved for new technology add-on payment, we recognize that the goal of facilitating access to new technologies for Medicare beneficiaries could also apply to other types of therapies. However, as discussed in the proposed rule ( 89 FR 36138 ), we focused our proposal on gene therapies for Medicare beneficiaries with SCD, as the most common inherited blood disorder, with historically limited treatment options and a significant clinical and financial impact on the healthcare system, and consistent with the Administration's commitment to improving outcomes for patients with SCD by facilitating access to gene therapies that treat SCD. We appreciate commenters' interest in improving access to these and other technologies through the new technology add-on payment program, and will continue to consider the interest areas raised by commenters.
With respect to comments that stated hospitals receive millions in outlier payments on the same cases that receive new technology add-on payment payments, highlighting how inadequate the new technology add-on payment is, and that even with a new technology add-on payment, cell and gene therapies are more likely than other inpatient stays to qualify for outlier payments, we disagree that the existence of outlier payments for some new technology cases is evidence that those payments are necessarily inadequate, as there may be unrelated reasons why a hospital would receive outlier payments. There may also be circumstances where new technology payments and outlier payments work in a complementary manner for related reasons, that do not necessarily mean the appropriate policy is to increase new technology payments.
Comment: Some of the commenters requested that CMS modify its proposal and finalize a maximum payment higher than 75 percent, stating that an increase of 10 percent would not adequately address the underlying problem of insufficient reimbursement. Many of these commenters stated that, considering the transformational potential of these therapies and the fact that these are among the costliest treatments to date, CMS should increase the percentage to 100 percent to provide a better incentive for hospitals to provide these therapies and not impede access for Medicare beneficiaries. Commenters stated that this is important since hospitals already incur losses on treatments that trigger new technology add-on payments, and these SCD therapies are even more costly. A commenter stated that in the absence of any other evaluation or discussion of reimbursement solutions, hospitals will be left to bear enormous losses for an essential therapy where there are no alternatives with similar outcomes, which would directly obstruct Medicare patients' access to gene therapies based on prices that are beyond the control of the provider and hinder future treatment options for this patient population. In addition, a commenter stated that Medicare payment policy sets the standard for other payers, so there would be a downstream effect of limited access if the policy is finalized as proposed at 75 percent. The commenter further stated that if these SCD therapies are not provided due to inadequate new technology add-on payment, there will be no data available to set appropriate rates after the new technology add-on payment period expires that include the costs of the therapies and associated inpatient costs. Another commenter stated that anything less than 100 percent would be inadequate due to significant financial losses that would need to be absorbed on every case, particularly for high DSH hospitals, which many hospitals that treat SCD are likely to be. The commenters stated that a payment rate of 100 percent would allow CMS to most effectively incentivize the development of important new technologies like gene therapies, help ensure patient access, reduce health disparities, positively impact other payer coverage decisions, and appropriately recognize the durable and transformative value that gene therapies offer to patients, their families, and society. A commenter stated that a 100 percent payment rate would demonstrate the same commitment to equity in the Medicare FFS population that the Cell and Gene Therapy (CGT) Access Model demonstrates for the Medicaid population. The commenter stated that 100 percent is reasonable given that the costs may be lower than anticipated due to the limited number of patients who may be candidates for SCD gene therapy and the limited manufacturing capacity, which is estimated to be less than 200 treatments per year.
A commenter shared data modeling simulating potential payment scenarios to demonstrate the impact of the current methodology to hospitals and the impact of potential new technology add-on payment percentage amounts at 65 percent, 75 percent, 85 percent and 100 ( print page 69249) percent using claims in MS-DRG 016 from the FY 2023 MedPAR file. The commenter stated that new technology add-on payment amounts at or below 75 percent would still leave hospitals severely under-reimbursed for the product and patient care costs, with a loss of over $700,000 with each case, which it stated would create vast barriers to utilization, no matter the clinical benefit. The commenter further asserted that even at 100 percent, some hospitals would lose over $250,000 or much more. The commenter explained that the analysis assumed that hospitals set charges for the gene therapy in line with the national average drug CCR of 0.182, which would be more than five times their cost. However, the commenter stated that in reality, hospitals do not markup higher cost drugs by that ratio, especially for gene therapies. The commenter stated that if SCD therapies had a 50 percent markup (for example, charging $3.3 million for a $2.2 million drug, reflecting a CCR of 0.666), but CMS applied a much lower CCR of 0.182 to the $3.3 million charge, CMS would drastically underestimate the cost of the drug at $600,600, only 27 percent of the actual cost. The commenter explained that this calculation, combined with new technology add-on payment as the lesser of the 75 percent of cost of the drug or 75 percent of the amount by which costs of the case exceed the standard DRG payment, would mean that hospitals would receive much smaller new technology add-on payments than under its analysis, and urged CMS to consider these dynamics as it implements new technology add-on payment for SCD gene therapy. The commenter suggested that rather than using the “lesser of” methodology, that CMS instead use the actual costs of such therapies, such as a percentage of wholesale acquisition costs (WAC) or the hospital's actual acquisition cost, as reported on the claims using value code 90.
A commenter stated that CMS had the statutory authority to provide for additional payment beyond the proposed 75 percent. The commenter stated that for SCD gene therapy, CMS's new technology add-on payment mechanism fails to “adequately reflect[ ] the estimated average cost of such service or technology” as required by the applicable statute, and that payment based on a portion of charges reduced to costs under section 412.88 would result in significant financial losses for providers. Therefore, the commenter recommended that CMS temporarily adopt a 100 percent cost-based reimbursement methodology for SCD gene therapy and/or take other measures to ensure that the payment methodology fully recognizes the estimated average cost of the care.
Another commenter stated that anything short of 100 percent reimbursement of acquisition costs would be inadequate for cell and gene therapies while eligible for new technology add-on payment. The commenter stated that increasing the payment to the full cost amount would ensure health equity and access. Another commenter suggested that CMS fully cover the costs of SCD gene therapy either by increasing the payment rate or through another innovative approach such as developing a new DRG with a higher base payment. A commenter also suggested that as an alternative to 100 percent payment, CMS should negotiate drug prices directly with drug manufacturers, or alternative pathways to support coverage and access. Another commenter advocated for a policy solution that would ensure providers recoup at least the invoice cost of high-cost therapies such as Casgevy TM and Lyfgenia TM , as the invoice cost of drugs is a factor over which providers have no control.
A few commenters requested that CMS instead increase the marginal payment rate (which we understand to refer to the maximum new technology add-on payment percentage) to at least 80 percent to better account for the high costs of these therapies and to address the lack of significant payment proposals related to these therapies. A commenter who requested a marginal payment rate of 100 percent stated that a marginal cost factor of less than 100 percent encourages efficient selection of alternative existing treatments for a condition, but for this particular set of patients there is no alternative treatment that is equal to an effective cure. The commenter further stated that an argument for the efficient selection of alternative treatments for these patients is an argument for early adoption of advanced curative services.
A few commenters who requested expansion of the proposal to additional therapies or a further increase in the payment percentage also commented on the short-term nature of the proposal, noting that there is no opportunity for other future new technology add-on payment-approved therapies to receive the increased percentage. The commenters requested that CMS make any changes permanent rather than limiting it to therapies approved for new technology add-on payment for FY 2025.
With regard to the comments requesting an increase to the new technology add-on payment percentage above the proposed rate of 75 percent, we acknowledge that SCD gene therapies are among the costliest therapies to date and there may be significant related costs associated with inpatient stays during which the therapies are provided. We also recognize that new technology add-on payment would not fully cover a hospital's costs, even with a 100 percent payment rate, due to the inherent design of the IPPS. At the same time, we note that we remain concerned about the extremely high cost of these products, and want to ensure we do not create incentives to increase prices. We continue to believe that limiting the new technology add-on payment percentage provides hospitals an incentive for continued cost-effective behavior in relation to the overall costs of the case. In response to commenters requesting a new technology add-on payment percentage of 100 percent, we believe that this would result in very little of the incentive for cost-effective behavior inherent to the prospective payment system. While we continue to believe that our standard add-on payment percentage is generally appropriate, due to the particular concerns related to SCD gene therapies previously discussed and confirmed by comments and consistent with the Administration's commitment to improving outcomes for patients with SCD by facilitating access to gene therapies that treat SCD, at this time we believe it is appropriate to apply a higher new technology add-on payment of 75 percent for SCD gene therapies approved for new technology add-on payment for FY 2025 during their new technology add-on payment period. We believe that the proposed 75 percent payment rate would reasonably address these concerns while also maintaining the incentives inherent to the prospective payment system, and it is consistent with our new technology add-on payment policy for QIDPs and LPADs. For these reasons, we are finalizing the increase in the new technology add-on payment percentage for cell and gene therapies that treat SCD as proposed.
With respect to commenters' other requested changes to our current payment mechanisms, due to the relative newness of these gene therapies for SCD and our continued consideration of approaches and authorities to encourage value-based care and lower prices of costly ( print page 69250) therapies, we believe it would be premature to adopt further structural changes to our existing payment mechanism specifically for these therapies. For these reasons, we disagree with the commenters' requested changes to our current payment mechanisms for FY 2025. For these same reasons, we also believe it would be premature to adopt a permanent increase in the new technology add-on payment percentage at this time. We will consider these comments should we develop additional policies and consider longer-term solutions related to SCD gene therapies in the future as we gain more experience with the unique considerations of these therapies. We also note that while Medicare payment policy may set the standard for other payers, payers consider many factors in designing and operating their programs.
Comment: Commenters opposed limiting the increase in the new technology add-on payment percentage to applicants that met certain additional criteria, such as attesting to offering and/or participating in outcomes-based pricing arrangements. A few of the commenters stated that CMS should not require additional criteria beyond the existing criteria of newness, cost, and substantial clinical improvement. Other commenters stated that CMS did not provide sufficient information regarding the feedback it is requesting related to outcomes-based arrangements, details on how it would operationalize such a requirement, or discuss the potential impact on claims data. They further stated that CMS must describe what arrangements or behaviors it is considering, in addition to the rationale and legal basis for any related proposal, so that stakeholders can appropriately comment on a proposal that has sufficient detail for effective evaluation via notice and comment rulemaking. A commenter stated that CMS should also consider the variability in such arrangements, which could lead to substantial inequities in which therapy patients would be able to access if this was a requirement to receive the new technology add-on payment amount, as well as the competitive disadvantage that may occur. A commenter stated that any such restrictions as described in CMS's proposal would impact patient access to transformative therapies by placing undue burden on providers and payers. The commenter further stated that a variety of factors may inhibit a manufacturer's ability to offer or participate in such arrangements, including lack of clarity in best price reporting, limited resources available within states to establish such agreements, and time needed to measure outcomes for new products. A commenter explained that IPPS hospitals are operating within a “buy-and-bill” environment without access to alternative contracting mechanisms, outcomes-based pricing arrangements, or other opportunities to control these therapies' prices, and that unless CMS links the Center for Medicare & Medicaid Innovation's (CMS Innovation Center) CGT Access Model efforts to Medicare FFS beneficiaries, these considerations would not apply to its member providers and hospitals. Another commenter stated that the arrangements CMS describes are encouraged to take place under the CMS Innovation Center's CGT Access Model and new technology add-on payment should not be tied to participation in the model, which is still under development. A commenter also stated that mandates related to outcomes-based pricing arrangements are not provided in the new technology add-on payment statute, and there is currently no mechanism by which FFS Medicare can engage in value-based payment arrangements. A commenter stated that CMS should work closely with impacted stakeholders before considering developing an alternative pricing requirement in the future to ensure any proposal would align with the new technology add-on payment program goals. Some commenters further stated that it is not clear how such additional criteria relate to or advance the purpose of the new technology add-on payment program.
Response: We appreciate the feedback from commenters. We note that we were seeking comments regarding other criteria that could demonstrate that applicants were engaging in behaviors that promote access to these therapies at lower cost, in alignment with the Administration's broader effort to further drive down prescription drug costs. [ 187 ] Consistent with our concerns about incentives for manufacturers to increase prices, we continue to welcome comments on this topic for future consideration. At this time, we are not making this 75 percent add-on payment percentage available only to applicants that meet certain additional criteria, but we will continue to evaluate this topic and may consider changes in the future.
Comment: A few commenters disagreed with CMS's proposal, stating that a new technology add-on payment of 75 percent will not create access to gene therapies. The commenters stated that a new technology add-on payment rate of 75 percent for these costly therapies would still leave a significant burden of unreimbursed costs on hospitals, while keeping drug manufacturers financially whole. The commenters stated that this would represent an unsustainable model for reimbursement and may disincentivize hospitals from providing these therapies, potentially leading to access issues for patients.
A commenter stated that CMS did not discuss its evaluation of any other solutions for improving the overall MS-DRG payment system, nor propose any other solutions for gene therapies, despite stakeholders having provided many ideas in the past. The commenter stated that CMS risked creating a two-tier system by fostering innovation for Medicaid patients via the CMS Innovation Center's new CGT Access Model, while offering no solutions for traditional Medicare FFS or Medicaid-Medicare dual-eligible patients with SCD or TDT, and did not view the proposal to be in harmony with the attention and effort being put into the CMS Innovation Center model. The commenter also asserted that the new technology add-on payment increase that CMS proposed does not address the series of compounding losses for hospitals that wish to provide these therapies: a low base MS-DRG payment rate, an inadequate new technology add-on payment percentage, the highest-ever fixed-loss threshold, and recovery of only 80 percent of remaining calculated costs through the outlier formula, which it stated directly obstruct Medicare patients' access to gene therapies. The commenter requested that CMS reimburse hospitals for 100 percent of their product acquisition costs related to gene therapies for SCD and TDT, potentially using CMS's adjustment authority under section 1886(d)(5)(I) of the Act. The commenter stated that this request could be operationalized by requiring hospitals to use value code 90 to report the product acquisition cost, providing payment at 100 percent of the reported product cost, and remove the charges reported in revenue code 0892 when calculating total case payment in determining whether an outlier payment is warranted. The commenter explained that hospitals would still be incentivized to provide cost-effective care, as the MS-DRG payment and outlier calculations would still be applicable to the clinical care portion of the claim. The commenter also expressed concern that charge ( print page 69251) compression, price transparency, and new technology add-on payment `lesser of' language combined to create a challenge that is impossible for hospitals to successfully navigate, as it stated that this required hospitals to mark-up multimillion dollar products, and was ineffective at achieving adequate reimbursement. The commenter asserted that if a hospital set charges for these therapies in accordance with its own CCR, it was entirely justifiable that a hospital would list the charges between $10 to 12 million, but was likely to be perceived as ethically problematic and predatory. In further support of its assertions, the commenter modeled the impact to hospitals using a simplified model of reimbursement for two hospitals, with one using a 10 percent policy and one using a CCR of 0.25 to mark-up the gene therapy product costs, to demonstrate that even hospitals that charged appropriately for these therapies and received the maximum 75 percent new technology add-on payment amount would face a significant financial loss. The commenter stated that the results showed that the hospitals had very different product charges, with different total claim charges—despite the fact that patient care charges are identical, leading CMS to compute a very different case cost estimate for each hospital. The commenter stated that the `lesser of' language used for new technology add-on payment meant that even when hospitals set their charges appropriately, they would be underpaid even the product acquisition cost, resulting in prohibitive financial choices, and where costs would largely be paid through outlier dollars. The commenter asserted that its proposal would have a limited total fiscal impact to CMS because of the limited number of treatments that will happen in the next few years, and the small percentage of applicable Medicare beneficiaries. The commenter referenced a prior letter from the American Hospital Association to CMS, [ 188 ] asserting that CMS has not typically fully spent the pool of new technology add-on payment dollars it allocates. The commenter further stated that adopting its proposal would allow for claims data with information on case volume, clinical care costs, and transparent product acquisition costs that could be used at the new technology add-on payment timeframe to create a new MS-DRG and/or an alternate payment mechanism to reflect the resources utilized to administer these therapies. Finally, the commenter noted a variety of suggestions it had previously provided, including Town Hall sessions, evaluating the creation of separate MS-DRGs for clinical care and product acquisition costs, creating a new MS-DRG, proposing new payment mechanism for acquisition of HSC gene therapy products, adding Medicare and dual-eligible beneficiaries in the CMS Innovation Center's CGT Access Model, and using a temporary CCR, and stated it was not clear as to why the agency chose to propose an increase to the new technology add-on payment percentage instead.
Some commenters stated that, while they were supportive of the proposed increase in payment for SCD gene therapies, they were concerned that the change would not adequately address gaps in payment or access issues for these therapies. A commenter stated that the SCD gene therapies map to DRGs that have base rates far below the costs of these products, and that reimbursement only covers a minimal portion of the drug cost and no provider and facility costs for the 30-days of inpatient care.
Multiple commenters also discussed similar concerns generally with new technology add-on payment methodology and in particular for costly therapies. They referenced the practice of “charge compression” due to CCRs and the way that the add-on payment amount is calculated as the “lesser of” two different values, which they stated results in hospitals incurring at least 35 percent of the new technology costs even with the new technology add-on payment (based on a 65 percent maximum add-on payment). Another commenter also suggested that CMS should eliminate the “lesser of” new technology add-on payment methodology for gene therapies targeting SCD and other technologies, which it stated required hospitals to artificially inflate their charges to obtain appropriate reimbursement.
Response: We appreciate the commenters' feedback. We note that the prospective payment system is an average-based system and it is expected that some cases may demonstrate higher than average costs, while other cases may demonstrate lower than average costs. In deciding which treatment is most appropriate for any particular patient, physicians are expected to balance the clinical needs of patients with the efficacy and costliness of particular treatments.
We continue to believe that changing the “lesser of” methodology, using the acquisition costs, or otherwise further increasing the add-on payment percentage would remove consideration of the costs of new technology from treatment decisions, and that it is important to maintain some incentive to weigh the costs of new technology in making clinical decisions. Similar to our discussion in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42299 ), we believe that paying hospitals for 100 percent of their product acquisition costs related to gene therapies would result in very little of the incentives inherent to the prospective payment system.
We also disagree with the commenter that this proposal, or other suggestions offered by other commenters, would have a limited total fiscal impact to CMS because of the limited number of treatments that will happen in the next few years and the small percentage of applicable Medicare beneficiaries. With regard to the commenter's statement regarding a pool of new technology add-on payment dollars that are allocated, we note that CMS does not allocate dollars to new technology add-on payments. We note that the citation provided by the commenter indicated that when implementing the new technology add-on payment in the September 7, 2001 final rule ( 66 FR 46902 ), CMS set a target limit for these payments at 1 percent of total operating prospective payments. However, the new mechanism was initially required to be implemented in a budget neutral manner, and as we had noted at that time, this limit was set to address CMS's concern that new technology add-on payments should not result in inappropriately large redistributions of payments from hospitals that do not employ new technology to those that do ( 66 FR 46920 ). In the FY 2005 IPPS final rule, we provided an update, that as a result of the enactment of section 503(d) of Public Law 108-173 , we will no longer include the impact of additional payments for new medical services and technologies in the budget neutrality factor ( 69 FR 49084 ). Due to the high cost of these gene therapy technologies, and because the total number of patients that will receive these treatments and the amount of new technology add-on payments associated with care of these patients in the future is unknown, it is unclear to us that the fiscal impact to CMS would be limited. We also note that because new technology add-on payments are not administered in a budget neutral manner, by default, they have the potential to result in increases to Medicare spending that are unpredictable and beyond our control, which is why we have remained ( print page 69252) cautious when assessing potential changes to the new technology add-on payment program to maintain the incentives inherent to the prospective payment system.
Comment: Many commenters stated that the Agency should work with stakeholders to identify adequate and sustainable reimbursement mechanisms for covering payment of outlier drug acquisition costs for both SCD and for other life-saving cell and gene therapies. Commenters stated that the current payment system was not designed to address market developments including rapid introduction of therapies with high costs, and was not sufficient to appropriately reimburse hospitals. Some commenters were particularly concerned about Medicare payment for these therapies after the new technology add-on payment expires, stating that the current MS-DRGs assigned have reimbursement rates inadequate to reimburse these high-cost therapies. The commenters urged CMS to consider alternative methods of reimbursement to support appropriate patient access in accordance with the goals of this proposal such as a continued pass-through payment for the gene therapies or some other mechanism, stating that the MS-DRG system was not structured to support therapies as costly as these SCD gene therapies. A commenter further stated the need for CMS to develop longer-term solutions to ensure reimbursement sustainability, and that a CMS-convened Town Hall session may be beneficial to facilitate innovative solutions. Commenters also suggested other potential pathways such as the creation of new MS-DRGs for high-cost treatments, and changes to the role of cost-to-charge ratios (CCRs) in the reimbursement methodology, such as eliminating the role of CCRs or creating a new CCR for more accurate rate-setting. A commenter further stated that these options are already within CMS's statutory authority and implementable through notice and comment rulemaking. The commenter further believed Congress must permanently resolve how to pay for these therapies, preferably through broad-scale reform of national drug development, production, and distribution policies. The commenter recommended that in the meantime, CMS work with Congress on changes specific to coverage and payment, such as by carving payment for these products out of the DRG system, as currently done for solid organ and stem cell transplants, or other policies, including split-DRGs, that would enable hospitals to recoup all their costs for these therapies.
A commenter voiced concerns over the rise of high-cost therapies generally and CMS's ability to appropriately account for their costs when determining payments to hospitals and health systems, urging CMS to examine the adequacy of its payments to hospitals. The commenter noted that many of these therapies' prices are beyond what would have been predicted when the inpatient PPS system was designed, and they are therefore adding to the existing and rising challenge of paying for a massive increase in high-cost therapies and technologies in health care.
Response: We thank commenters for their feedback and suggestions. As noted by commenters, longer-term solutions are outside of the scope of the new technology add-on payment program and this rulemaking. We will continue to consider these issues.
Therefore, after consideration of the public comments received, for the reasons discussed previously and in the FY 2025 IPPS/LTCH PPS proposed rule, we are finalizing our policy as proposed. We are finalizing that for certain gene therapies approved for new technology add-on payments in the FY 2025 IPPS/LTCH PPS final rule that are indicated and used specifically for the treatment of SCD, effective with discharges on or after October 1, 2024 and concluding at the end of the 2- to 3-year newness period for such therapy, if the costs of a discharge (determined by applying CCRs as described in § 412.84(h)) involving the use of such therapy for the treatment of SCD exceed the full DRG payment (including payments for IME and DSH, but excluding outlier payments), Medicare will make an add-on payment equal to the lesser of: (1) 75 percent of the costs of the new medical service or technology; or (2) 75 percent of the amount by which the costs of the case exceed the standard DRG payment. We note that these payment amounts would only apply to Casgevy TM (exagamglogene autotemcel) and Lyfgenia TM (lovotibeglogene autotemcel), when indicated and used specifically for the treatment of SCD, which CMS has determined in this FY 2025 IPPS/LTCH PPS final rule meet the criteria for approval for new technology add-on payment. We are also adding new § 412.88(a)(2)(ii)(C) and (b)(2)(iv) to reflect this change to the calculation of the new technology add-on payment amount, beginning in FY 2025 and concluding at the end of the 2- to 3-year newness period for each such therapy. As noted earlier, we will continue to assess this policy and may propose changes in the future.
Section 1886(d)(3)(E) of the Act requires that, as part of the methodology for determining prospective payments to hospitals, the Secretary adjust the standardized amounts for area differences in hospital wage levels by a factor (established by the Secretary) reflecting the relative hospital wage level in the geographic area of the hospital compared to the national average hospital wage level. We currently define hospital labor market areas based on the delineations of statistical areas established by the Office of Management and Budget (OMB). A discussion of the FY 2025 hospital wage index based on the statistical areas appears under section III.B. of the preamble of this final rule.
Section 1886(d)(3)(E) of the Act requires the Secretary to update the wage index annually and to base the update on a survey of wages and wage-related costs of short-term, acute care hospitals. CMS collects these data on the Medicare cost report, CMS Form 2552-10, Worksheet S-3, Parts II, III, IV. The OMB control number for this information collection request is 0938-0050, which expires on September 30, 2025. Section 1886(d)(3)(E) of the Act also requires that any updates or adjustments to the wage index be made in a manner that ensures that aggregate payments to hospitals are not affected by the change in the wage index. The adjustment for FY 2025 is discussed in section II.B. of the Addendum to this final rule.
As discussed in section III.I. of the preamble of this final rule, we also take into account the geographic reclassification of hospitals in accordance with sections 1886(d)(8)(B) and 1886(d)(10) of the Act when calculating IPPS payment amounts. Under section 1886(d)(8)(D) of the Act, the Secretary is required to adjust the standardized amounts so as to ensure that aggregate payments under the IPPS after implementation of the provisions of sections 1886(d)(8)(B), 1886(d)(8)(C), and 1886(d)(10) of the Act are equal to the aggregate prospective payments that would have been made absent these provisions. The budget neutrality adjustment for FY 2025 is discussed in section II.A.4.b. of the Addendum to this final rule.
Section 1886(d)(3)(E) of the Act also provides for the collection of data every 3 years on the occupational mix of employees for short-term, acute care ( print page 69253) hospitals participating in the Medicare program to construct an occupational mix adjustment to the wage index. The OMB control number for approved collection of this information is 0938-0907, which expires on January 31, 2026. A discussion of the occupational mix adjustment that we are applying to the FY 2025 wage index appears under section III.E. of the preamble of this final rule.
The wage index is calculated and assigned to hospitals on the basis of the labor market area in which the hospital is located. Under section 1886(d)(3)(E) of the Act, beginning with FY 2005 ( 69 FR 49026 through 49032 ), we delineate hospital labor market areas based on OMB-established Core-Based Statistical Areas (CBSAs). The current statistical areas (which were implemented beginning with FY 2021) are based on revised OMB delineations issued on Sept 14, 2018, in OMB Bulletin No. 18-04. [ 189 ] OMB Bulletin No. 18-04 established revised delineations for Metropolitan Statistical Areas, Micropolitan Statistical Areas, and Combined Statistical Areas in the United States and Puerto Rico based on the 2010 Census and the American Community Survey (ACS) and Census Bureau population estimates for 2015.
Historically, OMB issued major revisions to statistical areas every 10 years, based on the results of the decennial census, and occasionally issues minor updates and revisions to statistical areas in the years between the decennial censuses through OMB Bulletins. On February 28, 2013, OMB issued Bulletin No. 13-01. CMS adopted these delineations, based on the results of the 2010 census, effective beginning with the FY 2015 IPPS wage index ( 79 FR 49951 through 49957 ). OMB subsequently issued Bulletin No. 15-01 on July 15, 2015, followed by OMB Bulletin No. 17-01 on August 15, 2017, which provided updates to and superseded OMB Bulletin No. 15-01. The attachments to OMB Bulletin No. 17-01 provided detailed information on the update to statistical areas since July 15, 2015, and were based on the application of the 2010 Standards for Delineating Metropolitan and Micropolitan Statistical Areas to Census Bureau population estimates for July 1, 2014, and July 1, 2015. In the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41362 through 41363 ), we adopted the updates set forth in OMB Bulletin No. 17-01 effective October 1, 2018, beginning with the FY 2019 wage index. OMB Bulletin No. 17-01 was superseded by the April 10, 2018, OMB Bulletin No. 18-03, and then by the September 14, 2018, OMB Bulletin No. 18-04. These bulletins established revised delineations for Metropolitan Statistical Areas, Micropolitan Statistical Areas, and Combined Statistical Areas, and provided guidance on the use of the delineations of these statistical areas. In FY 2021, we adopted the updates set forth in OMB Bulletin No. 18-04 ( 85 FR 58743 through 58753 ). Thus, most recently in the FY 2024 IPPS/LTCH PPS final rule, we continued to use the OMB delineations that were adopted beginning with FY 2015 (based on the revised delineations issued in OMB Bulletin No. 13-01) to calculate the area wage indexes, with updates as reflected in OMB Bulletin Nos. 15-01, 17-01, and 18-04.
In the July 16, 2021, Federal Register ( 86 FR 37777 ), OMB finalized a schedule for future updates based on results of the decennial Census updates to commuting patterns from the ACS. In accordance with that schedule, on July 21, 2023, OMB released Bulletin No. 23-01. A copy of OMB Bulletin No. 23-01 may be obtained at https://www.whitehouse.gov/wp-content/uploads/2023/07/OMB-Bulletin-23-01.pdf . According to OMB, the delineations reflect the 2020 Standards for Delineating Core Based Statistical Areas (“the 2020 Standards”), which appeared in the Federal Register on July 16, 2021 ( 86 FR 37770 through 37778 ), and the application of those standards to Census Bureau population and journey-to-work data (that is, 2020 Decennial Census, American Community Survey, and Census Population Estimates Program data).
We believe that using the revised delineations based on OMB Bulletin No. 23-01 will increase the integrity of the IPPS wage index by creating a more accurate representation of current geographic variations in wage levels. Therefore, we proposed to implement the revised OMB delineations as described in the July 21, 2023, OMB Bulletin No. 23-01, beginning with the FY 2025 IPPS wage index. We proposed to use these revised delineations to calculate area wage indexes in a manner that is generally consistent with the CMS' implementation of CBSA-based wage index methodologies.
CMS has recognized that hospitals in certain areas may experience a negative impact on their IPPS payment due to the proposed adoption of the revised OMB delineations, and has finalized transition policies to mitigate negative financial impacts and provide stability to year-to-year wage index variations. We refer readers to the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 49956 through 49962 ) for discussion of the transition period finalized the last time CMS adopted revised OMB delineations after a decennial census, and to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49018 ) for discussion of wage index transition policies that we finalized for FYs 2020, 2021, and 2022 to apply a 5 percent cap on any decrease in a hospital's final wage index from the prior fiscal year. Beginning with FY 2023, we finalized and codified at 42 CFR 412.64(h)(7) a permanent policy to apply a 5 percent cap on any decrease to a hospital's wage index from its wage index in the prior FY, regardless of the circumstances causing the decline ( 87 FR 49018-49020 ). This 5 percent cap policy is discussed in further detail in section III.G.6 of the preamble of this final rule. We believe it is important for the IPPS to use the updated labor market area delineations to maintain a more accurate and up-to-date payment system that reflects the reality of current labor market conditions. We believe the 5 percent cap policy will sufficiently mitigate any potential significant disruptive financial impacts on hospitals that are negatively affected by the proposed adoption of the revised OMB delineations and thus, we did not propose a transition period for these hospitals.
For the reasons described in this section, we are finalizing the use of the revised labor market area delineations based on OMB Bulletin No. 23-01 beginning with the FY 2025 IPPS hospital wage index as proposed.
The OMB “2020 Standards” define a “Micropolitan Statistical Area” as being associated with at least one urban area that has a population of at least 10,000, but less than 50,000. A Micropolitan Statistical Area comprises the central county or counties containing the core, plus adjacent outlying counties having a high degree of social and economic integration with the central county or counties as measured through commuting ( 86 FR 37778 ). We refer to these areas as Micropolitan Areas. Since FY 2005, we have treated Micropolitan Areas as rural and included hospitals located in Micropolitan Areas in each State's rural wage index. We refer ( print page 69254) readers to the FY 2005 IPPS final rule ( 69 FR 49029 through 49032 ) and the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 49952 ) for a complete discussion regarding this policy and our rationale for treating Micropolitan Areas as rural. Based upon the new 2020 Decennial Census data, a number of urban counties have switched status and have joined or became Micropolitan Areas, and some counties that once were part of a Micropolitan Area, under current OMB delineations, have become urban. Overall, there are a similar number of Micropolitan Areas (542) under the new OMB delineations based on the 2020 Census as existed under the latest data from the 2010 Census (541). We stated in the proposed rule that we believe that the best course of action would be to continue the policy established in the FY 2005 IPPS final rule and include hospitals located in Micropolitan Areas in each State's rural wage index. These areas continue to be defined as having relatively small urban cores (populations of 10,000-49,999). We do not believe it would be appropriate to calculate a separate wage index for areas that typically may include only a few hospitals for the reasons set forth in the FY 2005 IPPS/LTCH PPS final rule ( 69 FR 49029 through 49032 ) and the FY 2015 IPPS final rule ( 79 FR 49952 ). Therefore, in conjunction with our proposal to implement the new OMB statistical area delineations beginning in FY 2025, we proposed to continue to treat Micropolitan Areas as “rural” and to include Micropolitan Areas in the calculation of each state's rural wage index.
According to OMB's “2020 Standards” ( 86 FR 37776 ), a metropolitan division is a county or group of counties within a metropolitan statistical area (MSA) with a population of at least 2.5 million. Thus, MSAs may be subdivided into metropolitan divisions. A county qualifies as a “main county” of a metropolitan division if 65 percent or more of workers living in the county also work within the county and the ratio of the number of workers working in the county to the number of workers living in the county is at least 0.75. A county qualifies as a “secondary county” if 50 percent or more, but less than 65 percent, of workers living in the county also work within the county and the ratio of the number of workers working in the county to the number of workers living in the county is at least 0.75. After all the main and secondary counties are identified and grouped, each additional county that already has qualified for inclusion in the MSA falls within the metropolitan division associated with the main/secondary county or counties with which the county at issue has the highest employment interchange measure. Counties in a metropolitan division must be contiguous. In the FY 2005 IPPS final rule ( 69 FR 49029 ), CMS finalized our policy to use the metropolitan divisions where applicable under the CBSA definitions. CMS concluded that including the metropolitan divisions in the CBSA definitions most closely approximated the labor market delineation from the “Primary Metropolitan Statistical Areas” delineations in place prior to FY 2005.
Under the current delineations, 11 MSAs are subdivided into a total of 31 metropolitan divisions. The revised OMB delineations have subdivided two additional existing MSAs into metropolitan divisions relative to the previous delineations, resulting in 13 MSAs (the 11 currently subdivided MSAs plus two additional MSAs) that are subdivided into 37 metropolitan divisions. Since the configurations of most subdivided MSAs remain substantially similar in the revised delineations compared to those used for the wage index in FY 2024, to maintain continuity and predictability in labor market delineations, we proposed to continue our policy to include metropolitan divisions as separate CBSAs for wage index purposes.
In a June 6, 2022, Notice ( 87 FR 34235 through 34240 ), the Census Bureau announced that it was implementing the State of Connecticut's request to replace the 8 counties in the State with 9 new “Planning Regions.” Planning regions now serve as county-equivalents within the CBSA system. OMB Bulletin No. 23-01 is the first set of revised delineations that referenced the new county-equivalents for Connecticut. We have evaluated the change in hospital assignments for Connecticut hospitals and proposed to adopt the planning regions as county equivalents for wage index purposes. As all forthcoming county-based delineation data will utilize these new county-equivalent definitions for Connecticut, we believe it is necessary to adopt this migration from counties to planning region county-equivalents to maintain consistency with OMB Bulletin No. 23-01 and future OMB updates. We are providing the following crosswalk for each hospital in Connecticut with the current and proposed Federal Information Processing Standard (FIPS) county and county-equivalent codes and CBSA assignments.

We note that we proposed that a remote location of a multicampus hospital currently indicated with 07B033 would be located in the same ( print page 69256) CBSA as the main provider (070033). Therefore, consistent with the policy for remote locations of multicampus hospitals discussed in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41369 through 41374 ), it would no longer be necessary to identify this remote location separately from the main provider for wage index purposes.
We also note, as discussed in Section III.B.3 of the preamble of this final rule, we proposed to add both of the newly proposed rural planning regions in Connecticut to the list of “Lugar” counties.
As previously discussed, we proposed to implement the revised OMB statistical area delineations (based upon OMB Bulletin No. 23-01) beginning in FY 2025. Our analysis shows that a total of 53 counties (and county equivalents) and 33 hospitals that were once considered part of an urban CBSA would be considered to be located in a rural area, beginning in FY 2025, under these revised OMB delineations. The following chart lists the 53 urban counties that will become rural under the revised OMB delineations. We note that there are four cases (CBSA 14100 [Bloomsburg-Berwick, PA], CBSA 19180 [Danville, IL], CBSA 20700 [East Stroudsburg, PA], and CBSA 35100 [New Bern, NC]) where all constituent counties in an urban CBSA become rural under the revised OMB delineations.

We proposed that the wage data for all hospitals located in the counties listed in the chart above would now be considered when calculating their ( print page 69258) respective State's rural wage index. We refer readers to section III.G.6 of the preamble of this final rule for a discussion of the 5 percent cap policy. We believe that this policy, which caps any reduction in a hospital's wage index value at 5 percent of the prior year wage index value, provides an adequate transition to mitigate any potential sudden negative financial impacts due to the adoption of wage index policies, including the adoption of revised OMB labor market delineations.
We also proposed revisions to the list of counties deemed urban under section 1886(d)(8)(B) of the Act, which would affect a number of the hospitals located in these proposed rural counties. We note that we proposed to add 17 of the 53 counties listed above to the list of “Lugar” counties whose hospitals, pursuant to section 1886(d)(8)(B), are deemed to be in an urban area. We refer readers to section III.F.4.b for further discussion.
In addition, we note the provisions of § 412.102 of our regulations continue to apply with respect to determining DSH payments. Specifically, in the first year after a hospital loses urban status, the hospital will receive an adjustment to its DSH payment that equals two-thirds of the difference between the urban DSH payments applicable to the hospital before its redesignation from urban to rural and the rural DSH payments applicable to the hospital subsequent to its redesignation from urban to rural. In the second year after a hospital loses urban status, the hospital will receive an adjustment to its DSH payment that equals one third of the difference between the urban DSH payments applicable to the hospital before its redesignation from urban to rural and the rural DSH payments applicable to the hospital subsequent to its redesignation from urban to rural.
As previously discussed, we proposed to implement the revised OMB statistical area delineations (based upon OMB Bulletin No. 23-01) beginning in FY 2025. Analysis of these OMB statistical area delineations shows that a total of 54 counties (and county equivalents) and 24 hospitals that were located in rural areas would be located in urban areas under the revised OMB delineations. The following chart lists the 54 rural counties that will be urban under the revised OMB delineations.

We proposed that when calculating the area wage index, the wage data for hospitals located in these counties would be included in their new respective urban CBSAs. We also note that due to the proposed adoption of the revised OMB delineations, some CAHs that were previously located in rural areas may be located in urban areas. The regulations at §§ 412.103(a)(6) and 485.610(b)(5) provide affected CAHs with a two-year transition period that begins from the date the redesignation becomes effective. The affected CAHs must reclassify as rural during this transition period to retain their CAH status after the two-year transition period ends. We refer readers to the FY 2015 IPPS/LTCH final rule ( 79 FR 50162 through 50163 ) for further discussion of the two-year transition period for CAHs. We also note that special statuses limited to hospitals located in rural areas (such as MDH or SCH status) may be terminated if hospitals are located in proposed urban counties. In these cases, affected hospitals should apply for rural reclassification status under § 412.103 prior to October 1, 2024, to ensure no disruption in status.
In addition to rural counties becoming urban and urban counties becoming rural, some urban counties shift from one urban CBSA to a new or existing urban CBSA under the new OMB delineations.
In some cases, the change in CBSA extends only to a change in name. Revised CBSA names can be found in Table 3 of the addendum of the final rule. In other cases, the CBSA number also changes. For these CBSAs, the list of constituent urban counties in FY 2024 and FY 2025 is the same (except in instances where an urban county became rural, or a rural county became urban, as discussed in the previous section). The following table lists the CBSAs where, under the new delineations, the CBSA name and number change but the constituent counties do not change (not including instances where an urban county became rural, or a rural county became urban).

In some cases, all of the urban counties from a FY 2024 CBSA have moved and been subsumed by another CBSA in FY 2025. The following table lists the CBSAs that, under the new delineations, are subsumed by an another CBSA.

In other cases, some counties shift between existing and new CBSAs, changing the constituent makeup of the CBSAs. For example, Calvert County, MD moved from the current CBSA 12580 (Washington-Arlington-Alexandria, DC-VA-MD-WV) into proposed CBSA 30500 (Lexington Park, MD). The other constituent counties of CBSA 12580 are split into urban CBSAs 47664 (Washington, DC-MD) and 11694 (Arlington-Alexandria-Reston, VA-WV). The following chart lists the urban counties that split off from one urban CBSA and move to a newly proposed or modified urban CBSA under the revised OMB delineations.

For hospitals located in these counties that move from one CBSA to another under the revised OMB delineations, there may be impacts, both negative and positive, upon their specific wage index values. We refer readers to section III.F.3.b.. of the preamble of this final rule for discussion of our proposals to address the assignment of MGCRB wage index reclassifications for hospitals currently reclassified to these modified CBSAs.
Comment: Multiple commenters were broadly supportive of CMS's proposed ( print page 69264) update to the IPPS wage index with the revised OMB delineations and the continuation of the policy to cap wage index decreases that a hospital can experience in a given year. MedPAC reiterated its concern with flaws in the wage index methodology, including continued concern with the rise in the number of MGCRB reclassifications. MedPAC urged CMS to improve the accuracy and equity of Medicare's wage index methodologies for IPPS hospitals and other providers by ensuring that wage indexes are less manipulable, more accurately and precisely reflect geographic differences in market-wide labor costs, and limit how much wage index values can differ among providers that are competing for the same pool of labor. MedPAC cited its June 2023 report to Congress, which recommended that Congress repeal the existing Medicare wage index statutes, including current exceptions, and require the Secretary to phase in new wage index methodologies for hospitals and other types of providers that:
- use all-employer, occupation-level wage data with different occupation weights for the wage index of each provider type;
- reflect local area level differences in wages between and within metropolitan statistical areas and statewide rural areas; and
- smooth wage index differences across adjacent local areas.
Another commenter requested that CMS solicit input from the hospital community on reforms to the wage index and efforts to improve the sustainability of workforce, especially in rural and underserved communities.
Response: We appreciate the comments supporting adoption of the revised OMB delineations and refer commenters to section III.G.2 of this final rule for additional discussion of the continuation of the 5 percent annual cap on hospital wage index reductions. We appreciate commenters' continued concern and MedPAC's recommendations for Congressional action on wage index reform. In the 2012 Report to Congress: Plan to Reform the Medicare Wage Index, CMS addressed several of MedPAC's recommendations and found significant benefits to an alternative wage index methodology. However, CMS concluded that any potential changes must be considered in conjunction with the statutorily required reclassifications and adjustments that are applicable to the current wage index determinations. There are several statutory provisions that enable a hospital to receive a wage index other than that which is computed for its geographic area. We believe that these provisions, which may have been designed to ameliorate or correct perceived inequities that hospitals may experience, would complicate the implementation of the significant modifications to the current wage index framework described in MedPAC's June 2023 report to Congress.
Comment: A commenter did not agree with CMS' adoption of OMB's CBSA delineation revisions. The commenter stated that OMB cautions that CBSAs are not intended for any non-statistical uses and should only be used with full consideration of the effects of using these delineations for such purposes. Further, the commenter stated that the Metropolitan Areas Protection and Standardization Act (MAPS Act) bars the automatic propagation of OMB revisions in CBSA delineations to geographic area determinations in non-statistical federal programs, and shall propagate for any non-statistical use only if the relevant agency determines that such a propagation supports the purposes of the program, is in the public interest, and adopts the change through notice-and-comment rulemaking. The commenter contends that if CMS chooses to adopt new OMB delineations, CMS must fully explain why reliance on the updated CBSAs as set forth by OMB is appropriate for purposes of the FY 2025 hospital wage index adjustments. The commenter stated that CMS has not provided an appropriate rationale for relying on the updated CBSAs and proposed to adopt the revised CBSAs by default. The commenter contends that CMS must make a fact-specific determination of those CBSAs' suitability for Medicare reimbursement purposes, including whether it would be appropriate to use additional data to modify OMB's delineations to ensure that such changes are appropriate for purposes of defining regional labor markets for hospital workers.
Response: CMS acknowledges that the CBSA definitions and delineations were not specifically created for the purpose of determining a hospital wage index. However, based on the reasons stated in prior rulemaking, we continue to believe that these definitions and delineations, which are regularly reviewed and updated by OMB, are the best proxy for CMS to use to adjust hospital payment rates based on geographic variations in labor costs in accordance with the statute. Section 1886(d)(3)(E) of the Act requires that, as part of the methodology for determining prospective payments to hospitals, the Secretary must adjust the standardized amounts “for area differences in hospital wage levels by a factor (established by the Secretary) reflecting the relative hospital wage level in the geographic area of the hospital compared to the national average hospital wage level.” We refer readers to the FY 1985 IPPS final rule ( 50 FR 24375 through 24377 ) and the FY 1995 IPPS final rule ( 60 FR 29218 through 29220 ) for a history of the outreach, consultation, and discussion of the challenges faced in defining appropriate labor market areas for purposes of the wage index methodology. As with any classification system in which boundaries must be established, it is impossible to designate boundaries that will be completely satisfactory to all concerned. There was no consensus among the interested parties on a choice for new labor market areas, and CMS concluded the adoption and continuation of an MSA-based framework was the most prudent course of action. We also refer readers to the FY 2005 rule ( 69 FR 49027 through 49028 ) for further discussion regarding the process and outreach CMS undertook before initially adopting OMB CBSAs as the basis for the wage index methodology. We found that the CBSA framework offered a useful proxy for labor market area delineations and that none of the alternative labor market areas that were studied provided a distinct improvement over the use of MSAs.
As stated previously, CMS continued to evaluate other potential methods to calculate variations in geographic labor markets in a manner that maintains or improves consistency and equity in hospital payments in response to recommendations from MedPAC. However, as stated in the 2012 Report to Congress: Plan to Reform the Medicare Wage Index (on the web at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/Wage-Index-Reform ), CMS has concluded that implementing any of the recommended revisions to wage index methodologies would require Congressional action. The commenter did not suggest any alternative method for defining geographic labor market areas and, given our decades long history of using OMB CBSA (and the prior Primary MSA) definitions and delineations for wage index purposes, we continue to believe adopting OMB revisions in a timely manner is essential to the IPPS wage index by creating a more accurate representation of geographic variations in wage levels. CMS is aware of the MAPS Act requirements for the adoption of CBSA definitions for non-statistical use and believes that we have ( print page 69265) provided an adequate rationale to support our proposed adoption through notice and comment rulemaking. As we stated in the proposed rule ( 89 FR 36140 ), we believe that using the revised delineations based on OMB Bulletin No. 23-01 will increase the integrity of the IPPS wage index by creating a more accurate representation of current geographic variations in wage levels. While the adoption of the revised delineations will have both positive and negative impacts on specific hospitals and labor markets, we believe that periodically updating the labor market delineations using objective criteria and based on the most recently available commuting data will serve the public's interest in ensuring accurate Medicare payments to hospitals under the IPPS by more accurately reflecting current geographic variations in labor costs in the hospital payment methodology. While some CBSAs would be modified in significant ways, the criteria for MSA, Micropolitan Statistical Area, and Metropolitan Division definitions finalized by OMB are generally consistent with past updates, and we do not find that the adoption of these delineations will create extreme variations in payments to hospitals, especially when considering the impact of the policy to cap annual wage index reductions at 5 percent. On this basis, and for the reasons we stated in prior rulemaking as described above, we have determined that their use supports the purpose of adjusting hospital payment rates based on geographic variations in labor costs in accordance with the statute. We have reviewed our findings and impacts relating to the new OMB delineations and find no compelling reason to delay implementation. Therefore, we are finalizing the proposed policies implementing the revised OMB delineations, including the policy for the treatment of Micropolitan Statistical areas, Metropolitan divisions, and the change to county-equivalent definitions for the State of Connecticut.
Overall, we believe implementing the new OMB labor market area delineations would result in wage index values being more representative of the actual current costs of labor in a given area. However, we recognize that some hospitals would experience decreases in wage index values as a result of our proposed implementation of the new labor market area delineations. We also realize that some hospitals would have higher wage index values due to our proposed implementation of the new labor market area delineations.
In the past, we have provided for transition periods when adopting changes that have significant payment implications, particularly large negative impacts. When adopting new OMB delineations based on the decennial census for the 2005 and 2015 wage indexes, we applied a 3-year transition for urban hospitals that became rural under the new delineations and a 50/50 blended wage index adjustment for all hospitals that would experience any decrease in their actual payment wage index ( 69 FR 49032 through 49034 and 79 FR 28060 through 28062 ).
In connection with our adoption in FY 2021 of the updates in OMB Bulletin 18-04, which included more modifications to the CBSAs than are typical for OMB bulletins issued between decennial censuses, we adopted a policy to place a 5 percent cap on any decrease in a hospital's wage index from the hospital's final wage index in FY 2020 so that a hospital's final wage index for FY 2021 would not be less than 95 percent of its final wage index for FY 2020 ( 85 FR 58753 through 58755 ). Given the unprecedented nature of the COVID-19 public health emergency (PHE), we adopted a policy in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45164 through 45165 ) to apply an extended transition to the FY 2022 wage index for hospitals affected by the transition in FY 2021 to mitigate significant negative impacts of, and provide additional time for hospitals to adapt to, the CMS decision to adopt the revised OMB delineations. In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49018 through 49021 ), under the authority at sections 1886(d)(3)(E) and 1886(d)(5)(I)(i) of the Act, we finalized a policy for FY 2023 and subsequent years to apply a 5 percent cap on any decrease to a hospital's wage index from its wage index in the prior FY, regardless of the circumstances causing the decline.
We believe that this permanent cap policy, reflected at 42 CFR 412.64(h)(7) and discussed in section in III.G.6. of the preamble of this final rule, sufficiently mitigates any large negative impacts of adopting the new delineations. As we stated when finalizing the permanent 5 percent cap policy in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49018 through 49021 ), we further considered the comments we received during the FY 2022 rulemaking recommending a permanent 5 percent cap policy to prevent large year-to-year variations in wage index values as a means to reduce overall volatility for hospitals. We do not believe any additional transition period is necessary considering that the current cap on wage index decreases, which was not in place when we implemented the decennial census updates in FY 2005 and FY 2015, ensures that a hospital's wage index would not be less than 95 percent of its final wage index for the prior year.
Comment: A commenter requested that in addition to the permanent cap policy, CMS implement a 3-year wage index transition period consistent with prior updates to the CBSA categorizations made due to OMB updates.
Response: We note that when we previously adopted revised OMB delineations, the majority of negatively impacted hospitals received a wage index adjustment for only one fiscal year via a 50/50 blend of wage index values using the then-current and newly adopted delineations ( 79 FR 49960 ). Hospitals that were reassigned from an urban to rural area as a result of our adoption of the revised OMB delineations received a 3-year transition from their previous urban area, as long as they did not obtain a new MGCRB reclassification during that time period. As discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49018 through 49021 ), the 5 percent cap on annual wage index reductions was intended to make unnecessary any future transitions in connection with wage index policy implementations, including the adoption of revised labor market area definitions. Based on our analysis of wage index differences between FY 2024 and FY 2025, we estimate that only 117 hospitals (less that 4 percent) will receive a wage index cap that did not receive the cap in FY 2024. This indicates any impact on overall wage index values that could be caused by the adoption of the revised delineations would be relatively small. Furthermore, given the iterative and interactive effects of different reclassification and wage index hold-harmless policies, it is difficult to isolate the effects on wage index values (both positive and negative) that are due solely to the adoption of the revised delineations. That is, hospitals may make different reclassification decisions based on the transition policy, rather than the actual impacts of the revised delineations. We believe that any attempt to tailor a transition policy specifically to the impacts of adopting revised labor market delineations is not likely to yield results that more accurately reflect current differences in area wages than the 5 percent cap policy. We believe the 5 percent cap policy ensures that hospitals will not experience large payment reductions as a result of annual changes in their wage index value, ( print page 69266) allows adequate time for hospitals to evaluate reclassification options, and provides consistency and predictability in wage index values. Largely due to the modification of the rural wage index calculation finalized in FY 2024 IPPS/LTCH final rule ( 88 FR 58971 through 58977 ), a much larger number of urban and rural hospitals within the same state (nearly 60 percent) receive identical wage index values (prior to the application of other policies, such as the outmigration adjustment, 5 percent cap on annual wage index decreases, and low-wage index hospital policy). This fact suggests that there is even less need for separate transition policies for urban and rural hospitals in response to changes in geographic delineations than there was previously. Furthermore, we did not receive a comment from any hospital (urban or rural) citing specific negative impacts due solely or primarily to the proposed adoption of the revised OMB delineations. For these reasons, we do not believe it is necessary to implement any additional or alternative transition policy to the 5 percent cap discussed in section III.G.6 of this final rule.
The FY 2025 wage index values are based on the data collected from the Medicare cost reports submitted by hospitals for cost reporting periods beginning in FY 2021 (the FY 2024 wage indexes were based on data from cost reporting periods beginning during FY 2020).
The FY 2025 wage index includes all of the following categories of data associated with costs paid under the IPPS (as well as outpatient costs):
- Salaries and hours from short-term, acute care hospitals (including paid lunch hours and hours associated with military leave and jury duty).
- Home office costs and hours.
- Certain contract labor costs and hours, which include direct patient care, certain top management, pharmacy, laboratory, and nonteaching physician Part A services, and certain contract indirect patient care services (as discussed in the FY 2008 final rule with comment period ( 72 FR 47315 through 47317 )).
- Wage-related costs, including pension costs (based on policies adopted in the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51586 through 51590 ) and modified in the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49505 through 49508 )) and other deferred compensation costs.
Consistent with the wage index methodology for FY 2024, the wage index for FY 2025 excludes the direct and overhead salaries and hours for services not subject to IPPS payment, such as skilled nursing facility (SNF) services, home health services, costs related to GME (teaching physicians and residents) and certified registered nurse anesthetists (CRNAs), and other subprovider components that are not paid under the IPPS. The FY 2025 wage index also excludes the salaries, hours, and wage-related costs of hospital-based rural health clinics (RHCs), and Federally Qualified Health Centers (FQHCs), because Medicare pays for these costs outside of the IPPS ( 68 FR 45395 ). In addition, salaries, hours, and wage-related costs of CAHs are excluded from the wage index for the reasons explained in the FY 2004 IPPS final rule ( 68 FR 45397 through 45398 ). Similar to our treatment of CAHs, as discussed later in this section, we proposed to exclude Rural Emergency Hospitals (REHs) from the wage index.
For FY 2020 and subsequent years, other wage-related costs are also excluded from the calculation of the wage index. As discussed in the FY 2019 IPPS/LTCH final rule ( 83 FR 41365 through 41369 ), other wage-related costs reported on Worksheet S-3, Part II, Line 18 and Worksheet S-3, Part IV, Line 25 and subscripts, as well as all other wage-related costs, such as contract labor costs, are excluded from the calculation of the wage index.
Comment: One commenter encouraged CMS needs to give consideration to policy options that can incentivize safe staffing practices, for the sake of Medicare patients, without simultaneously encouraging hospitals to pay excessively high wages for temporary staff. The commenter also asked that CMS include sick leave in the wage index with the expectation that hospitals and other facilities will allow payment for the entire uncapped time that staff are sick.
Response: We include the categories of data listed above that are associated with costs paid under the IPPS, which includes temporary staff. We also include sick leave in the wage index. For complete detail on what is allowed to be included in the wage data, we refer the reader to the Provider Reimbursement Manual (PRM), Part 2 (Pub. 15-2), Chapter 40, sections 4005.2-4005.4. Also, we appreciate the commenters concern with regard to safe staffing practices. We note, that since the time the end of the COVID-19 PHE, hospitals have begun to reduce their reliance on temporary staff such as traveling nurses. Also, some states have begun to explore capping the wages charged by travel nursing agencies. We thank the commenter for their input on this matter.
Data collected for the IPPS wage index also are currently used to calculate wage indexes applicable to suppliers and other providers, such as SNFs, home health agencies (HHAs), ambulatory surgical centers (ASCs), and hospices. In addition, they are used for prospective payments to IRFs, IPFs, and LTCHs, and for hospital outpatient services. We note that, in the IPPS rules, we do not address comments pertaining to the wage indexes of any supplier or provider except IPPS providers and LTCHs. Such comments should be made in response to separate proposed rules for those suppliers and providers.
The wage data for the FY 2025 wage index were obtained from Worksheet S-3, Parts II, III and IV of the Medicare cost report, CMS Form 2552-10 (OMB Control Number 0938-0050 with an expiration date September 30, 2025) for cost reporting periods beginning on or after October 1, 2020, and before October 1, 2021. For wage index purposes, we refer to cost reports beginning on or after October 1, 2020, and before October 1, 2021, as the “FY 2021 cost report,” the “FY 2021 wage data,” or the “FY 2021 data.” Instructions for completing the wage index sections of Worksheet S-3 are included in the Provider Reimbursement Manual (PRM), Part 2 (Pub. 15-2), Chapter 40, Sections 4005.2 through 4005.4. The data file used to construct the FY 2025 wage index includes FY 2021 data submitted to us as of June 2024. As in past years, we performed an extensive review of the wage data, mostly through the use of edits designed to identify aberrant data.
Consistent with the IPPS and LTCH PPS ratesettings, our policy principles with regard to the wage index include generally using the most current data and information available, which is usually data on a 4-year lag (for example, for the FY 2023 wage index we used cost report data from FY 2019). We stated in the FY 2023 IPPS/LTCH final rule ( 87 FR 48994 ) that we will be looking at the differential effects of the COVID-19 PHE on the audited wage data in future fiscal years. We also stated we plan to review the audited ( print page 69267) wage data, and the impacts of the COVID-19 PHE on such data and evaluate these data for future rulemaking. For the FY 2025 wage index, the best available data typically would be from the FY 2021 wage data.
In the proposed rule we stated that in considering the impacts of the COVID-19 PHE on the FY 2021 wage data, we compared that data with recent historical data. Based on pre reclassified wage data, the changes in the wage data from FY 2020 to FY 2021 show the following compared to the annual changes for the most recent 3 fiscal year periods (that is, FY 2017 to FY 2018, FY 2018 to FY 2019 and FY 2019 to FY 2020):
- Approximately 91 percent of hospitals have an increase in their average hourly wage (AHW) from FY 2020 to FY 2021 compared to a range of 76-86 percent of hospitals for the most recent 3 fiscal year periods.
- Approximately 97 percent of all CBSA AHWs are increasing from FY 2020 to FY 2021 compared to a range of 84-91 percent of all CBSA AHWs for the most recent 3 fiscal year periods.
- Approximately 51 percent of all urban areas have an increase in their area wage index from FY 2020 to FY 2021 compared to a range of 36-43 percent of all urban areas for the most recent 3 fiscal year periods.
- Approximately 55 percent of all rural areas have an increase in their area wage index from FY 2020 to FY 2021 compared to a range of 31-46 percent of all rural areas for the most recent 3 fiscal year periods.
- The unadjusted national average hourly wage increased by a range of 2.4-5.4 percent per year from FY 2017-FY 2020. For FY 2021, the unadjusted national average hourly increased by 8.7 percent from FY 2020.
Similar to the FY 2024 wage index, we stated it is not readily apparent even if the comparison with the historical trends had indicated greater differences at a national level in this context, how any changes due to the COVID-19 PHE differentially impacted the wages paid by individual hospitals. Furthermore, even if changes due to the COVID-19 PHE did differentially impact the wages paid by individual hospitals over time, it is not clear how those changes could be isolated from changes due to other reasons and what an appropriate potential methodology might be to adjust the data to account for the effects of the COVID-19 PHE.
Lastly, we also noted that we have not identified any significant issues with the FY 2021 wage data itself in terms of our audits of this data. As usual, the data was audited by the Medicare Administrative Contractors (MACs), and there were no significant issues reported across the data for all hospitals.
Taking all of these factors into account, we stated that we believe the FY 2021 wage data is the best available wage data to use for FY 2025 and proposed to use the FY 2021 wage data for FY 2025.
We welcomed comments from the public with regard to the FY 2021 wage data. We also noted, AHW data by provider and CBSA, including the data upon which the comparisons provided previously are based, is available in our Public Use Files released with each proposed and final rule each fiscal year. The Public Use Files for the respective FY Wage Index Home Page can be found on the Wage Index Files web page at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/wage-index-files .
Comment: A commenter noted that for FY 2025, CMS proposed to use data from the FY 2021 cost reports to determine the area wage index modifications. The commenter stated that CMS is already using the FY 2022 cost reports for rate setting and therefore CMS should use the FY 2022 cost reports for area wage index modifications.
Response: As discussed previously, the latest available audited wage data is from FY 2021. We do not possess audited wage data from a more recent period. We are uncertain to what the commenter meant to refer with respect to the use of FY 2022 cost reports for rate setting and are unable to respond further to the commenter's suggestion.
Comment: One commenter stated that although CMS provides some information about this analysis, they recommend CMS provide additional information, such as specific tables or files for the public to review, to confirm the agency's conclusion. The commenter stated that they are skeptical of the agency's conclusion, as workforce costs continue to account for a substantial portion of hospital expenses, driven in part by use of contract labor and shortages that were accelerated by many of the impacts of the pandemic.
Response: As stated above, AHW data by provider and CBSA, including the data upon which the comparisons as previously described are based, is available in our Public Use Files released with each proposed and final rule each fiscal year. The Public Use Files for the respective FY Wage Index Home Page can be found on the Wage Index Files web page at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/Wage-Index-Files . Also, as usual, the data was audited by the MACs, and there were no significant issues reported across the data for all hospitals including contract labor.
We did not receive any other comments regarding the use of the FY 2021 wage data for FY 2025. We are finalizing as proposed to use the FY 2021 wage data for the FY 2025 wage index.
We requested that our MACs revise or verify data elements that resulted in specific edit failures. For the proposed FY 2025 wage index, we identified and excluded 69 providers with aberrant data that should not be included in the wage index. However, we stated that if data elements for some of these providers are corrected, we intend to include data from those providers in the final FY 2025 wage index. We also adjusted certain aberrant data and included these data in the wage index. For example, in situations where a hospital did not have documentable salaries, wages, and hours for housekeeping and dietary services, we imputed estimates, in accordance with policies established in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 49965 through 49967 ). We instructed MACs to complete their verification of questionable data elements and to transmit any changes to the wage data no later than March 20, 2024. After we issued the proposed rule, for the final FY 2025 wage index, we restored the data of 8 hospitals to the wage index, because their data was either verified or improved and removed the data of 3 hospitals with aberrant data. Thus, 64 hospitals with aberrant data remain excluded from the FY 2025 wage index (69−8 + 3 = 64).
Comment: One commenter stated that certain Medicare Administrative Contractors (MACs) may be taking different stances on whether to allow or how to calculate the allowable portion of contract labor when determining a hospital's wage index. The commenter indicated that although it seems some MACs have taken steps to correct this after hospitals have appealed such actions, CMS should ensure a uniform process is followed.
Response: All hospitals and MACs are provided with the same instructions for reviewing the wage data, including instructions for determining the allowable portion of contract labor. Also, complete instructions with regard to what hospitals can and cannot include in the wage data and contract labor are in PRM, Part 2 (Pub. 15-2), Chapter 40, section 4005.2. Further, as the commenter mentions, if there is an issue during the review process, ( print page 69268) hospitals can follow the appeal process described below.
In constructing the proposed FY 2025 wage index, we included the wage data for facilities that were IPPS hospitals in FY 2021, inclusive of those facilities that have since terminated their participation in the program as hospitals, as long as those data did not fail any of our edits for reasonableness. We stated in the proposed rule ( 89 FR 36151-36152 ) that we believe that including the wage data for these hospitals is, in general, appropriate to reflect the economic conditions in the various labor market areas during the relevant past period and to ensure that the current wage index represents the labor market area's current wages as compared to the national average of wages. However, we excluded the wage data for CAHs as discussed in the FY 2004 IPPS final rule ( 68 FR 45397 through 45398 ); that is, any hospital that is designated as a CAH by 7 days prior to the publication of the preliminary wage index public use file (PUF) is excluded from the calculation of the wage index. For the proposed FY 2025 wage index, we removed 8 hospitals that converted to CAH status on or after January 23, 2023, the cut-off date for CAH exclusion from the FY 2024 wage index, and through and including January 24, 2024, the cut-off date for CAH exclusion from the FY 2025 wage index. We noted that we also removed 2 hospitals that converted to CAH status prior to January 23, 2023. We did not receive any comments with regard to this proposal, and we are finalizing as proposed to exclude hospitals that have subsequently converted to CAH from the wage index calculation. Since the proposed rule, we learned of 1 more hospital that converted to CAH status on or after January 22, 2023, and through and including January 23, 2024. We removed this additional hospital from the FY 2025 wage index due to its conversion to CAH status.
The Consolidated Appropriations Act (CAA), 2021, was signed into law on December 27, 2020. Section 125 of Division CC (section 125) established a new rural Medicare provider type: Rural Emergency Hospitals (REHs). (We refer the reader to the CMS website at https://www.cms.gov/medicare/health-safety-standards/guidance-for-laws-regulations/hospitals/rural-emergency-hospitals for additional information on REHs.) In doing so, section 125 amended section 1861(e) of the Act, which provides the definition of a hospital and states that the term “hospital” does not include, unless the context otherwise requires, a critical access hospital (as defined in subsection (mm)(1)) or a rural emergency hospital (as defined in subsection (kkk)(2)). Section 125 also added section 1861(kkk) to the Act, which sets forth the requirements for REHs. Per section 1861(kkk)(2) of the Act, one of the requirements for an REH is that it does not provide any acute care inpatient services (other than post-hospital extended care services furnished in a distinct part unit licensed as a skilled nursing facility (SNF)). In the proposed rule we stated that, similar to CAHs, we believe hospitals that have subsequently converted to REH status should be removed from the wage index calculation, because they are a separately certified Medicare provider type and are not comparable to other short-term, acute care hospitals as they do not provide inpatient hospital services. For FY 2025, we proposed to treat REHs the same as CAHs and to exclude 15 REHs from the wage index. Accordingly, we proposed that, similar to our policy on CAHs, any hospital that is designated as a REH by 7 days prior to the publication of the preliminary wage index public use file (PUF) is excluded from the calculation of the wage index. We did not receive any comments with regard to this proposal, and we are finalizing as proposed to exclude hospitals that have subsequently converted to REH from the wage index calculation. Since the proposed rule, we learned of 4 more hospitals that converted to REH status on or after January 22, 2023, and through and including January 23, 2024, the cut-off date for REH exclusion from the FY 2025 wage index, for a total of 19 hospitals that were removed from the FY 2025 wage index due to conversion to REH status. In summary, we calculated the FY 2025 wage index using the Worksheet S-3, Parts II and III wage data of 3,074 hospitals.
For the FY 2025 wage index, we allotted the wages and hours data for a multicampus hospital among the different labor market areas where its campuses are located using campus full-time equivalent (FTE) percentages as originally finalized in the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51591 ). Table 2, which contains the FY 2025 wage index associated with this final rule (available via the internet on the CMS website), includes separate wage data for the campuses of 26 multicampus hospitals. The following chart lists the multicampus hospitals by CMS certification number (CCN) and the FTE percentages on which the wages and hours of each campus were allotted to their respective labor market areas:

We note that, in past years, in Table 2, we have placed a “B” to designate the subordinate campus in the fourth position of the hospital CCN. However, for the FY 2019 IPPS/LTCH PPS proposed and final rules and subsequent rules, we have moved the “B” to the third position of the CCN. Because all IPPS hospitals have a “0” in the third position of the CCN, we believe that placement of the “B” in this third position, instead of the “0” for the subordinate campus, is the most efficient method of identification and interferes the least with the other variable digits in the CCN.
The preliminary, unaudited Worksheet S-3 wage data files for the proposed FY 2025 wage index were made available on May 23, 2023, through the internet on the CMS website at https://www.cms.gov/medicare/medicare-fee-service-payment/acuteinpatientpps/wage-index-files/fy-2025-wage-index-home-page . We subsequently identified some providers that were inadvertently omitted from the FY 2025 preliminary Worksheet S-3 wage data file originally posted on May 23, 2023. Therefore, on July 12, 2023, we posted an updated FY 2025 preliminary Worksheet S-3 wage data file to include these missing providers. In addition, the Calendar Year (CY) 2022 occupational mix survey data was made available on July 12, 2023, through the internet on the CMS website at https://www.cms.gov/medicare/medicare-fee-service-payment/acuteinpatientpps/wage-index-files/fy-2025-wage-index-home-page . On August 14, 2023, we posted an updated CY 2022 Occupational Mix survey data file that includes survey data for providers that were inadvertently omitted from the file posted on July 12, 2023.
On January 31, 2024, we posted a public use file (PUF) at https://www.cms.gov/medicare/medicare-fee-service-payment/acuteinpatientpps/wage-index-files/fy-2025-wage-index-home-page containing FY 2025 wage index data available as of January 31, 2024. This PUF contains a tab with the Worksheet S-3 wage data (which includes Worksheet S-3, Parts II and III wage data from cost reporting periods beginning on or after October 1, 2020, through September 30, 2021; that is, FY 2021 wage data), a tab with the occupational mix data (which includes data from the CY 2022 occupational mix survey, Form CMS-10079), a tab containing the Worksheet S-3 wage data of hospitals deleted from the January 31, 2024 wage data PUF, and a tab containing the CY 2022 occupational mix data of the hospitals deleted from the January 31, 2024 occupational mix PUF. In a memorandum dated January 31, 2024, we instructed all MACs to inform the IPPS hospitals that they service of the availability of the January 31, 2024, wage index data PUFs, and the process and timeframe for requesting revisions in accordance with the FY 2025 Hospital Wage Index Development Time Table available at https://www.cms.gov/files/document/fy2025-hospital-wage-index-development-timetable.pdf .
In the interest of meeting the data needs of the public, beginning with the proposed FY 2009 wage index, we post an additional PUF on the CMS website that reflects the actual data that are used in computing the proposed wage index. The release of this file does not alter the current wage index process or schedule. We notify the hospital community of the availability of these data as we do with the current public use wage data files through our Hospital Open Door Forum. We encourage hospitals to sign up for automatic notifications of information ( print page 69270) about hospital issues and about the dates of the Hospital Open Door Forums at the CMS website at https://www.cms.gov/Outreach-and-Education/Outreach/OpenDoorForums .
In a memorandum dated May 4, 2023, we instructed all MACs to inform the IPPS hospitals that they service of the availability of the preliminary wage index data files and the CY 2022 occupational mix survey data files posted on May 23, 2023, and the process and timeframe for requesting revisions.
If a hospital wished to request a change to its data as shown in the May 23, 2023, preliminary wage data files and occupational mix data files, the hospital had to submit corrections along with complete, detailed supporting documentation to its MAC so that the MAC received them by September 1, 2023. Hospitals were notified of these deadlines and of all other deadlines and requirements, including the requirement to review and verify their data as posted in the preliminary wage index data files on the internet, through the letters sent to them by their MACs.
November 3, 2023, was the date by when MACs notified State hospital associations regarding hospitals that failed to respond to issues raised during the desk reviews. Additional revisions made by the MACs were transmitted to CMS throughout January 2024. CMS published the wage index PUFs that included hospitals' revised wage index data on January 31, 2024. Hospitals had until February 16, 2024, to submit requests to the MACs to correct errors in the January 31, 2024, PUF due to CMS or MAC mishandling of the wage index data, or to revise desk review adjustments to their wage index data as included in the January 31, 2024, PUF. Hospitals also were required to submit sufficient documentation to support their requests. Hospitals' requests and supporting documentation must have been received by the MAC by the February deadline (that is, by February 16, 2024, for the FY 2025 wage index).
After reviewing requested changes submitted by hospitals, MACs were required to transmit to CMS any additional revisions resulting from the hospitals' reconsideration requests by March 20, 2024. Under our current policy as adopted in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38153 ), the deadline for a hospital to request CMS intervention in cases where a hospital disagreed with a MAC's handling of wage data on any basis (including a policy, factual, or other dispute) was April 3, 2024. Data that were incorrect in the preliminary or January 31, 2024, wage index data PUFs, but for which no correction request was received by the February 16, 2024, deadline, are not considered for correction at this stage. In addition, April 3, 2024, was the deadline for hospitals to dispute data corrections made by CMS of which the hospital was notified after the January 31, 2024, PUF and at least 14 calendar days prior to April 3, 2024 (that is, March 20, 2024), that do not arise from a hospital's request for revisions. The hospital's request and supporting documentation must be received by CMS (and a copy received by the MAC) by the April deadline (that is, by April 3, 2024, for the FY 2025 wage index). We refer readers to the FY 2025 Hospital Wage Index Development Time Table for complete details.
Hospitals were given the opportunity to examine Table 2 associated with the proposed rule, which is listed in section VI. of the Addendum to the proposed rule and available via the internet on the CMS website at https://www.cms.gov/medicare/medicare-fee-service-payment/acuteinpatientpps/wage-index-files/fy-2025-wage-index-home-page . Table 2 associated with the proposed rule contained each hospital's proposed adjusted average hourly wage used to construct the wage index values for the past 3 years, including the proposed FY 2025 wage index, which was constructed from FY 2021 data. We noted in the proposed rule that the proposed hospital average hourly wages shown in Table 2 only reflected changes made to a hospital's data that were transmitted to CMS by early February 2024.
We posted the final wage index data PUFs on April 29, 2024, on the CMS website at https://www.cms.gov/medicare/medicare-fee-service-payment/acuteinpatientpps/wage-index-files/fy-2025-wage-index-home-page . The April 2024 PUFs are made available solely for the limited purpose of identifying any potential errors made by CMS or the MAC in the entry of the final wage index data that resulted from the correction process (the process for disputing revisions submitted to CMS by the MACs by March 20, 2024, and the process for disputing data corrections made by CMS that did not arise from a hospital's request for wage data revisions as discussed earlier), as previously described.
After the release of the April 2024 wage index data PUFs, changes to the wage and occupational mix data can only be made in those very limited situations involving an error by the MAC or CMS that the hospital could not have known about before its review of the final wage index data files. Specifically, neither the MAC nor CMS will approve the following types of requests:
- Requests for wage index data corrections that were submitted too late to be included in the data transmitted to CMS by the MACs on or before March 20, 2024.
- Requests for correction of errors that were not, but could have been, identified during the hospital's review of the January 31, 2024, wage index PUFs.
- Requests to revisit factual determinations or policy interpretations made by the MAC or CMS during the wage index data correction process.
If, after reviewing the April 2024 final wage index data PUFs, a hospital believes that its wage or occupational mix data are incorrect due to a MAC or CMS error in the entry or tabulation of the final data, the hospital is given the opportunity to notify both its MAC and CMS regarding why the hospital believes an error exists and provide all supporting information, including relevant dates (for example, when it first became aware of the error). The hospital was required to send its request to CMS and to the MAC so that it was received no later than May 29, 2024. May 29, 2024, was also the deadline for hospitals to dispute data corrections made by CMS of which the hospital was notified on or after 13 calendar days prior to April 3, 2024 (that is, March 21, 2024), and at least 14 calendar days prior to May 29, 2024 (that is, May 15, 2024), that did not arise from a hospital's request for revisions. (Data corrections made by CMS of which a hospital is notified on or after 13 calendar days prior to May 29, 2024 (that is, May 16, 2024), may be appealed to the Provider Reimbursement Review Board (PRRB)). In accordance with the FY 2025 Hospital Wage Index Development Time Table posted on the CMS website at https://www.cms.gov/files/document/fy2025-hospital-wage-index-development-timetable.pdf , the May appeals were required to be submitted to CMS through an online submission process or through email. We refer readers to the FY 2025 Hospital Wage Index Development Time Table for complete details.
Verified corrections to the wage index data received timely (that is, by May 29, 2024) by CMS and the MACs were incorporated into the final FY 2025 wage index, which will be effective October 1, 2024.
We created the processes previously described to resolve all substantive wage index data correction disputes before we finalize the wage and occupational mix data for the FY 2025 payment rates. Accordingly, hospitals ( print page 69271) that do not meet the procedural deadlines set forth earlier will not be afforded a later opportunity to submit wage index data corrections or to dispute the MAC's decision with respect to requested changes. Specifically, our policy is that hospitals that do not meet the procedural deadlines as previously set forth (requiring requests to MACs by the specified date in February and, where such requests are unsuccessful, requests for intervention by CMS by the specified date in April) will not be permitted to challenge later, before the PRRB, the failure of CMS to make a requested data revision. We refer readers also to the FY 2000 IPPS final rule ( 64 FR 41513 ) for a discussion of the parameters for appeals to the PRRB for wage index data corrections. As finalized in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38154 through 38156 ), this policy also applies to a hospital disputing corrections made by CMS that do not arise from a hospital's request for a wage index data revision. That is, a hospital disputing an adjustment made by CMS that did not arise from a hospital's request for a wage index data revision is required to request a correction by the first applicable deadline. Hospitals that do not meet the procedural deadlines set forth earlier will not be afforded a later opportunity to submit wage index data corrections or to dispute CMS' decision with respect to changes.
Again, we believe the wage index data correction process described earlier provides hospitals with sufficient opportunity to bring errors in their wage and occupational mix data to the MAC's attention. Moreover, because hospitals had access to the final wage index data PUFs by late April 2024, they have an opportunity to detect any data entry or tabulation errors made by the MAC or CMS before the development and publication of the final FY 2025 wage index by August 2024, and the implementation of the FY 2025 wage index on October 1, 2024. Given these processes, the wage index implemented on October 1 should be accurate. Nevertheless, in the event that errors are identified by hospitals and brought to our attention after May 29, 2024, we retain the right to make midyear changes to the wage index under very limited circumstances.
Specifically, in accordance with § 412.64(k)(1) of our regulations, we make midyear corrections to the wage index for an area only if a hospital can show that: (1) The MAC or CMS made an error in tabulating its data; and (2) the requesting hospital could not have known about the error or did not have an opportunity to correct the error, before the beginning of the fiscal year. For purposes of this provision, “before the beginning of the fiscal year” means by the May deadline for making corrections to the wage data for the following fiscal year's wage index (for example, May 29, 2024, for the FY 2025 wage index). This provision is not available to a hospital seeking to revise another hospital's data that may be affecting the requesting hospital's wage index for the labor market area. As indicated earlier, because CMS makes the wage index data available to hospitals on the CMS website prior to publishing both the proposed and final IPPS rules, and the MACs notify hospitals directly of any wage index data changes after completing their desk reviews, we do not expect that midyear corrections will be necessary. However, under our current policy, if the correction of a data error changes the wage index value for an area, the revised wage index value will be effective prospectively from the date the correction is made.
In the FY 2006 IPPS final rule ( 70 FR 47385 through 47387 and 47485 ), we revised § 412.64(k)(2) to specify that, effective on October 1, 2005, that is, beginning with the FY 2006 wage index, a change to the wage index can be made retroactive to the beginning of the Federal fiscal year only when CMS determines all of the following: (1) The MAC or CMS made an error in tabulating data used for the wage index calculation; (2) the hospital knew about the error and requested that the MAC and CMS correct the error using the established process and within the established schedule for requesting corrections to the wage index data, before the beginning of the fiscal year for the applicable IPPS update (that is, by the May 29, 2024, deadline for the FY 2025 wage index); and (3) CMS agreed before October 1 that the MAC or CMS made an error in tabulating the hospital's wage index data and the wage index should be corrected.
In those circumstances where a hospital requested a correction to its wage index data before CMS calculated the final wage index (that is, by the May 29, 2024 deadline for the FY 2025 wage index), and CMS acknowledges that the error in the hospital's wage index data was caused by CMS' or the MAC's mishandling of the data, we believe that the hospital should not be penalized by our delay in publishing or implementing the correction. As with our current policy, we indicated that the provision is not available to a hospital seeking to revise another hospital's data. In addition, the provision cannot be used to correct prior years' wage index data; it can only be used for the current Federal fiscal year. In situations where our policies would allow midyear corrections other than those specified in § 412.64(k)(2)(ii), we continue to believe that it is appropriate to make prospective-only corrections to the wage index.
We note that, as with prospective changes to the wage index, the final retroactive correction will be made irrespective of whether the change increases or decreases a hospital's payment rate. In addition, we note that the policy of retroactive adjustment will still apply in those instances where a final judicial decision reverses a CMS denial of a hospital's wage index data revision request.
The process set forth with the wage index timetable discussed in section III.C.4. of the preamble of this final rule allows hospitals to request corrections to their wage index data within prescribed timeframes. In addition to hospitals' opportunity to request corrections of wage index data errors or MACs' mishandling of data, CMS has the authority under section 1886(d)(3)(E) of the Act to make corrections to hospital wage index and occupational mix data to ensure the accuracy of the wage index. As we explained in the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49490 through 49491 ) and the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56914 ), section 1886(d)(3)(E) of the Act requires the Secretary to adjust the proportion of hospitals' costs attributable to wages and wage-related costs for area differences reflecting the relative hospital wage level in the geographic areas of the hospital compared to the national average hospital wage level. We believe that, under section 1886(d)(3)(E) of the Act, we have discretion to make corrections to hospitals' data to help ensure that the costs attributable to wages and wage-related costs in fact accurately reflect the relative hospital wage level in the hospitals' geographic areas.
We have an established multistep, 15-month process for the review and correction of the hospital wage data that is used to create the IPPS wage index for the upcoming fiscal year. Since the origin of the IPPS, the wage index has been subject to its own annual review process, first by the MACs, and then by CMS. As a standard practice, after each annual desk review, CMS reviews the results of the MACs' desk reviews and focuses on items flagged during the desk ( print page 69272) review, requiring that, if necessary, hospitals provide additional documentation, adjustments, or corrections to the data. This ongoing communication with hospitals about their wage data may result in the discovery by CMS of additional items that were reported incorrectly or other data errors, even after the posting of the January 31 PUF, and throughout the remainder of the wage index development process. In addition, the fact that CMS analyzes the data from a regional and even national level, unlike the review performed by the MACs that review a limited subset of hospitals, can facilitate additional editing of the data the need for which may not be readily apparent to the MACs. In these occasional instances, an error may be of sufficient magnitude that the wage index of an entire CBSA is affected. Accordingly, CMS uses its authority to ensure that the wage index accurately reflects the relative hospital wage level in the geographic area of the hospital compared to the national average hospital wage level, by continuing to make corrections to hospital wage data upon discovering incorrect wage data, distinct from instances in which hospitals request data revisions.
We note that CMS corrects errors to hospital wage data as appropriate, regardless of whether that correction will raise or lower a hospital's average hourly wage. For example, as discussed in section III.C. of the preamble of the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41364 ), in situations where a hospital did not have documentable salaries, wages, and hours for housekeeping and dietary services, we imputed estimates, in accordance with policies established in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 49965 through 49967 ). Furthermore, if CMS discovers after conclusion of the desk review, for example, that a MAC inadvertently failed to incorporate positive adjustments resulting from a prior year's wage index appeal of a hospital's wage-related costs such as pension, CMS would correct that data error, and the hospital's average hourly wage would likely increase as a result.
While we maintain CMS' authority to conduct additional review and make resulting corrections at any time during the wage index development process, in accordance with the policy finalized in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38154 through 38156 ) and as first implemented with the FY 2019 wage index ( 83 FR 41389 ), hospitals are able to request further review of a correction made by CMS that did not arise from a hospital's request for a wage index data correction. Instances where CMS makes a correction to a hospital's data after the January 31 PUF based on a different understanding than the hospital about certain reported costs, for example, could potentially be resolved using this process before the final wage index is calculated. We believe this process and the timeline for requesting review of such corrections (as described earlier and in the FY 2018 IPPS/LTCH PPS final rule) promote additional transparency in instances where CMS makes data corrections after the January 31 PUF and provide opportunities for hospitals to request further review of CMS changes in time for the most accurate data to be reflected in the final wage index calculations. These additional appeals opportunities are described earlier and in the FY 2025 Hospital Wage Index Development Time Table, as well as in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38154 through 38156 ).
The method used to compute the proposed FY 2025 wage index without an occupational mix adjustment follows the same methodology that we used to compute the wage indexes without an occupational mix adjustment in the FY 2021 IPPS/LTCH PPS final rule (see 85 FR 58758-58761 ), and we did not propose any changes to this methodology. We have restated our methodology in this section of this rule.
Step 1.—We gathered data from each of the non-Federal, short-term, acute care hospitals for which data were reported on the Worksheet S-3, Parts II and III of the Medicare cost report for the hospital's cost reporting period relevant to the wage index (in this case, for FY 2025, these were data from cost reports for cost reporting periods beginning on or after October 1, 2020, and before October 1, 2021). In addition, we included data from hospitals that had cost reporting periods beginning prior to the October 1, 2020 begin date and extending into FY 2021 but that did not have any cost report with a begin date on or after October 1, 2020 and before October 1, 2021. We include this data because no other data from these hospitals would be available for the cost reporting period as previously described, and because particular labor market areas might be affected due to the omission of these hospitals. However, we generally describe these wage data as data applicable to the fiscal year wage data being used to compute the wage index for those hospitals. We note that, if a hospital had more than one cost reporting period beginning during FY 2021 (for example, a hospital had two short cost reporting periods beginning on or after October 1, 2020, and before October 1, 2021), we include wage data from only one of the cost reporting periods, the longer, in the wage index calculation. If there was more than one cost reporting period and the periods were equal in length, we included the wage data from the later period in the wage index calculation.
Step 2.—Salaries.—The method used to compute a hospital's average hourly wage excludes certain costs that are not paid under the IPPS. (We note that, beginning with FY 2008 ( 72 FR 47315 ), we included what were then Lines 22.01, 26.01, and 27.01 of Worksheet S-3, Part II of CMS Form 2552-96 for overhead services in the wage index. Currently, these lines are lines 28, 33, and 35 on CMS Form 2552-10. However, we note that the wages and hours on these lines are not incorporated into Line 101, Column 1 of Worksheet A, which, through the electronic cost reporting software, flows directly to Line 1 of Worksheet S-3, Part II. Therefore, the first step in the wage index calculation is to compute a “revised” Line 1, by adding to the Line 1 on Worksheet S-3, Part II (for wages and hours respectively) the amounts on Lines 28, 33, and 35.) In calculating a hospital's Net Salaries (we note that we previously used the term “average” salaries in the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51592 ), but we now use the term “net” salaries) plus wage-related costs, we first compute the following: Subtract from Line 1 (total salaries) the GME and CRNA costs reported on CMS Form 2552-10, Lines 2, 4.01, 7, and 7.01, the Part B salaries reported on Lines 3, 5 and 6, home office salaries reported on Line 8, and exclude salaries reported on Lines 9 and 10 (that is, direct salaries attributable to SNF services, home health services, and other subprovider components not subject to the IPPS). We also subtract from Line 1 the salaries for which no hours were reported. Therefore, the formula for Net Salaries (from Worksheet S-3, Part II) is the following:
((Line 1 + Line 28 + Line 33 + Line 35)−(Line 2 + Line 3 + Line 4.01 + Line 5 + Line 6 + Line 7 + Line 7.01 + Line 8 + Line 9 + Line 10)).
To determine Total Salaries plus Wage-Related Costs, we add to the Net Salaries the costs of contract labor for direct patient care, certain top management, pharmacy, laboratory, and nonteaching physician Part A services (Lines 11, 12 and 13), home office salaries and wage-related costs reported by the hospital on Lines 14.01, 14.02, and 15, and nonexcluded area wage- ( print page 69273) related costs (Lines 17, 22, 25.50, 25.51, and 25.52). We note that contract labor and home office salaries for which no corresponding hours are reported are not included. In addition, wage-related costs for nonteaching physician Part A employees (Line 22) are excluded if no corresponding salaries are reported for those employees on Line 4. The formula for Total Salaries plus Wage-Related Costs (from Worksheet S-3, Part II) is the following:
((Line 1 + Line 28 + Line 33 + Line 35)−(Line 2 + Line 3 + Line 4.01 + Line 5 + Line 6 + Line 7 + Line 7.01 + Line 8 + Line 9 + Line 10)) + (Line 11 + Line 12 + Line 13 + Line 14.01 + 14.02 + Line 15) + (Line 17 + Line 22 + 25.50 + 25.51 + 25.52).
Step 3.—Hours.—With the exception of wage-related costs, for which there are no associated hours, we compute total hours using the same methods as described for salaries in Step 2. The formula for Total Hours (from Worksheet S-3, Part II) is the following:
((Line 1 + Line 28 + Line 33 + Line 35)−(Line 2 + Line 3 + Line 4.01 + Line 5 + Line 6 + Line 7 + Line 7.01 + Line 8 + Line 9 + Line 10)) + (Line 11 + Line 12 + Line 13 + Line 14.01 + 14.02 + Line 15).
Step 4.—For each hospital reporting both total overhead salaries and total overhead hours greater than zero, we then allocate overhead costs to areas of the hospital excluded from the wage index calculation. First, we determine the “excluded rate”, which is the ratio of excluded area hours to Revised Total Hours (from Worksheet S-3, Part II) with the following formula:
(Line 9 + Line 10)/(Line 1 + Line 28 + Line 33 + Line 35)−(Lines 2, 3, 4.01, 5, 6, 7, 7.01, and 8 and Lines 26 through 43).
We then compute the amounts of overhead salaries and hours to be allocated to the excluded areas by multiplying the previously discussed ratio by the total overhead salaries and hours reported on Lines 26 through 43 of Worksheet S-3, Part II. Next, we compute the amounts of overhead wage-related costs to be allocated to the excluded areas using three steps:
- We determine the “overhead rate” (from Worksheet S-3, Part II), which is the ratio of overhead hours (Lines 26 through 43 minus the sum of Lines 28, 33, and 35) to revised hours excluding the sum of lines 28, 33, and 35 (Line 1 minus the sum of Lines 2, 3, 4.01, 5, 6, 7, 7.01, 8, 9, 10, 28, 33, and 35). We note that, for the FY 2008 and subsequent wage index calculations, we have been excluding the overhead contract labor (Lines 28, 33, and 35) from the determination of the ratio of overhead hours to revised hours because hospitals typically do not provide fringe benefits (wage-related costs) to contract personnel. Therefore, it is not necessary for the wage index calculation to exclude overhead wage-related costs for contract personnel. Further, if a hospital does contribute to wage-related costs for contracted personnel, the instructions for Lines 28, 33, and 35 require that associated wage-related costs be combined with wages on the respective contract labor lines. The formula for the Overhead Rate (from Worksheet S-3, Part II) is the following:
(Lines 26 through 43−Lines 28, 33 and 35)/((((Line 1 + Lines 28, 33, 35)−(Lines 2, 3, 4.01, 5, 6, 7, 7.01, 8, and 26 through 43))−(Lines 9 and 10)) + (Lines 26 through 43−Lines 28, 33, and 35)).
- We compute overhead wage-related costs by multiplying the overhead hours ratio by wage-related costs reported on Part II, Lines 17, 22, 25.50, 25.51, and 25.52.
- We multiply the computed overhead wage-related costs by the previously described excluded area hours ratio.
Finally, we subtract the computed overhead salaries, wage-related costs, and hours associated with excluded areas from the total salaries (plus wage-related costs) and hours derived in Steps 2 and 3.
Step 5.—For each hospital, we adjust the total salaries plus wage-related costs to a common period to determine total adjusted salaries plus wage-related costs. To make the wage adjustment, we estimate the percentage change in the employment cost index (ECI) for compensation for each 30-day increment from October 14, 2020, through April 15, 2022, for private industry hospital workers from data obtained from the Bureau of Labor Statistics' (BLS') Office of Compensation and Working Conditions. We use the ECI because it reflects the price increase associated with total compensation (salaries plus fringes) rather than just the increase in salaries. In addition, the ECI includes managers as well as other hospital workers. This methodology to compute the monthly update factors uses actual quarterly ECI data and assures that the update factors match the actual quarterly and annual percent changes. We also note that, since April 2006 with the publication of March 2006 data, the BLS' ECI uses a different classification system, the North American Industrial Classification System (NAICS), instead of the Standard Industrial Codes (SICs), which no longer exist. We have consistently used the ECI as the data source for our wages and salaries and other price proxies in the IPPS market basket, and we did not propose to make any changes to the usage of the ECI for FY 2025. The factors used to adjust the hospital's data are based on the midpoint of the cost reporting period, as indicated in this rule.
Step 6.—Each hospital is assigned to its appropriate urban or rural labor market area before any reclassifications under section 1886(d)(8)(B), 1886(d)(8)(E), or 1886(d)(10) of the Act. Within each urban or rural labor market area, we add the total adjusted salaries plus wage-related costs obtained in Step 5 for all hospitals in that area to determine the total adjusted salaries plus wage-related costs for the labor market area.
Step 7.—We divide the total adjusted salaries plus wage-related costs obtained under Step 6 by the sum of the corresponding total hours (from Step 4) for all hospitals in each labor market area to determine an average hourly wage for the area.
Step 8.—We add the total adjusted salaries plus wage-related costs obtained in Step 5 for all hospitals in the Nation and then divide the sum by the national sum of total hours from Step 4 to arrive at a national average hourly wage.
Step 9.—For each urban or rural labor market area, we calculate the hospital wage index value, unadjusted for occupational mix, by dividing the area average hourly wage obtained in Step 7 by the national average hourly wage computed in Step 8.
Step 10.—For each urban labor market area for which we do not have any hospital wage data (either because there are no IPPS hospitals in that labor market area, or there are IPPS hospitals in that area but their data are either too new to be reflected in the current year's wage index calculation, or their data are aberrant and are deleted from the wage index), we finalized in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42305 ) that, for FY 2020 and subsequent years' wage index calculations, such CBSAs' wage index would be equal to total urban salaries plus wage-related costs (from Step 5) in the State, divided by the total urban hours (from Step 4) in the State, divided by the national average hourly wage from Step 8 (see 84 FR 42305 and 42306 ,). We stated that we believe that, in the absence of wage data for an urban labor market area, it is reasonable to use a statewide urban average, which is based on actual, acceptable wage data of hospitals in that State, rather than impute some other ( print page 69274) type of value using a different methodology. For calculation of the FY 2025 wage index, we note there is one urban CBSA for which we do not have IPPS hospital wage data. In Table 3 (which is available via the internet on the CMS website), which contains the area wage indexes, we include a footnote to indicate to which CBSA this policy applies. This CBSA's wage index is calculated as described, based on the FY 2020 IPPS/LTCH PPS final rule methodology ( 84 FR 42305 ). Under this step, we also apply our policy with regard to how dollar amounts, hours, and other numerical values in the wage index calculations are rounded, as discussed in this section of this final rule.
We refer readers to section II. of the Appendix of the final rule for the policy regarding rural areas that do not have IPPS hospitals.
Step 11.—Section 4410 of Public Law 105-33 provides that, for discharges on or after October 1, 1997, the area wage index applicable to any hospital that is located in an urban area of a State may not be less than the area wage index applicable to hospitals located in rural areas in that State. The areas affected by this provision are identified in Table 2 listed in section VI. of the Addendum to the final rule and available via the internet on the CMS website.
The following is our policy with regard to rounding of the wage data (dollar amounts, hours, and other numerical values) in the calculation of the unadjusted and adjusted wage index, as finalized in the FY 2020 IPPS/LTCH final rule ( 84 FR 42306 ). For data that we consider to be “raw data,” such as the cost report data on Worksheets S-3, Parts II and III, and the occupational mix survey data, we use such data “as is,” and do not round any of the individual line items or fields. However, for any dollar amounts within the wage index calculations, including any type of summed wage amount, average hourly wages, and the national average hourly wage (both the unadjusted and adjusted for occupational mix), we round the dollar amounts to 2 decimals. For any hour amounts within the wage index calculations, we round such hour amounts to the nearest whole number. For any numbers not expressed as dollars or hours within the wage index calculations, which could include ratios, percentages, or inflation factors, we round such numbers to 5 decimals. However, we continue rounding the actual unadjusted and adjusted wage indexes to 4 decimals, as we have done historically.
As discussed in the FY 2012 IPPS/LTCH PPS final rule, in “Step 5,” for each hospital, we adjust the total salaries plus wage-related costs to a common period to determine total adjusted salaries plus wage-related costs. To make the wage adjustment, we estimate the percentage change in the ECI for compensation for each 30-day increment from October 14, 2020, through April 15, 2022, for private industry hospital workers from the BLS' Office of Compensation and Working Conditions data. We have consistently used the ECI as the data source for our wages and salaries and other price proxies in the IPPS market basket, and we did not propose any changes to the usage of the ECI for FY 2025. The factors used to adjust the hospital's data were based on the midpoint of the cost reporting period, as indicated in the following table.

For example, the midpoint of a cost reporting period beginning January 1, 2021, and ending December 31, 2021, is June 30, 2021. An adjustment factor of 1.03606 was applied to the wages of a hospital with such a cost reporting period.
Previously, we also would provide a Puerto Rico overall average hourly wage. As discussed in the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56915 ), prior to January 1, 2016, Puerto Rico hospitals were paid based on 75 percent of the national standardized amount and 25 percent of the Puerto Rico-specific standardized amount. As a result, we calculated a Puerto Rico specific wage index that was applied to the labor-related share of the Puerto Rico-specific standardized amount. Section 601 of Division O, Title VI (section 601) of the Consolidated Appropriations Act, 2016 ( Pub. L. 114-113 ) amended section 1886(d)(9)(E) of the Act to specify that the payment calculation with respect to operating costs of inpatient hospital services of a subsection (d) Puerto Rico hospital for inpatient hospital discharges on or after January 1, 2016, shall use 100 percent of the national standardized amount. As we stated in the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56915 through ( print page 69275) 56916), because Puerto Rico hospitals are no longer paid with a Puerto Rico specific standardized amount as of January 1, 2016, under section 1886(d)(9)(E) of the Act, as amended by section 601 of the Consolidated Appropriations Act, 2016, there is no longer a need to calculate a Puerto Rico specific average hourly wage and wage index. Hospitals in Puerto Rico are now paid 100 percent of the national standardized amount and, therefore, are subject to the national average hourly wage (unadjusted for occupational mix) and the national wage index, which is applied to the national labor-related share of the national standardized amount. Therefore, for FY 2025, there is no Puerto Rico-specific overall average hourly wage or wage index.
Based on the previously discussed methodology, we stated in the proposed rule ( 89 FR 36159 ) that the proposed FY 2025 unadjusted national average hourly wage was $54.80.
Based on the previously described methodology, the final FY 2025 unadjusted national average hourly wage is the following:
As stated earlier, section 1886(d)(3)(E) of the Act provides for the collection of data every 3 years on the occupational mix of employees for each short-term, acute care hospital participating in the Medicare program, to construct an occupational mix adjustment to the wage index, for application beginning October 1, 2004 (the FY 2005 wage index). The purpose of the occupational mix adjustment is to control for the effect of hospitals' employment choices on the wage index. For example, hospitals may choose to employ different combinations of registered nurses, licensed practical nurses, nursing aides, and medical assistants for the purpose of providing nursing care to their patients. The varying labor costs associated with these choices reflect hospital management decisions rather than geographic differences in the costs of labor.
Section 304(c) of Appendix F, Title III of the Consolidated Appropriations Act, 2001 ( Pub. L. 106-554 ) amended section 1886(d)(3)(E) of the Act to require CMS to collect data every 3 years on the occupational mix of employees for each short-term, acute care hospital participating in the Medicare program and to measure the earnings and paid hours of employment for such hospitals by occupational category. As discussed in the FY 2022 IPPS/LTCH PPS proposed rule ( 86 FR 25402 through 25403 ) and final rule ( 86 FR 45173 ), we collected data in 2019 to compute the occupational mix adjustment for the FY 2022, FY 2023, and FY 2024 wage indexes. A new measurement of occupational mix is required for FY 2025.
The FY 2025 occupational mix adjustment is based on a new calendar year (CY) 2022 survey. Hospitals were required to submit their completed 2022 surveys (Form CMS-10079, OMB Number 0938-0907, expiration date January 31, 2026) to their MACs by July 1, 2023. The preliminary, unaudited CY 2022 survey data were posted on the CMS website on July 12, 2023. As with the Worksheet S-3, Parts II and III cost report wage data, as part of the FY 2025 desk review process, the MACs revised or verified data elements in hospitals' occupational mix surveys that resulted in certain edit failures.
Consistent with the IPPS and LTCH PPS ratesettings, our policy principles with regard to the occupational mix adjustment include generally using the most current data and information available, which is usually occupational mix data on a 3-year lag in the first year of the use of the occupational mix survey (for example, for the FY 2022 wage index we used occupational mix data from 2019; we also used this data for the FY 2023 and FY 2024 wage indexes). In the FY 2024 IPPS/LTCH final rule ( 88 FR 58969-58970 ), a commenter had concerns that the occupational mix data [that would be used for FY 2025?] may be skewed due to the COVID-19 PHE, and we stated that we planned to assess the CY 2022 Occupational Mix Survey data in the FY 2025 IPPS final rule.
In the proposed rule, we explained that based on pre-reclassified wage data, we computed the unadjusted and adjusted wage indexes for FY 2025 using the 2022 occupational mix survey data. We then measured the increases and decreases by CBSA as a result of the 2022 occupational mix survey data. We compared this table to the same table for the FY 2024 wage indexes, which used the 2019 occupational mix data, as well as the FY 2021 wage indexes, which used the 2016 occupational mix data. We stated that this table demonstrates the impact of the occupational mix adjusted wage data compared to unadjusted wage data for the most recent three occupational mix surveys using the 2022 survey data compared to the 2019 survey data and the 2016 survey data. That is, it shows whether hospitals' wage indexes will increase or decrease under the 2022 survey data as compared to the most recent years using the prior 2019 survey data and 2016 survey data respectively.

Based on the table, we stated that increases and decreases by CBSA are alike across each year of occupational mix data. For example, 60.19 percent of urban areas' wage indexes are increasing in FY 2025 due to the CY 2022 occupational mix data compared to 56.07 percent in FY 2024 using CY 2019 occupational mix data. Similarly, 59.57 percent of rural areas' wage indexes are increasing in FY 2025 due to the CY 2022 occupational mix data compared to 57.45 percent in FY 2024 using CY 2019 occupational mix data. We also noted that similar to the wage data, it is not readily apparent, even if the comparison with the historical trends had indicated greater differences by CBSA in this context, how any changes due to the COVID-19 PHE differentially impacted the occupational mix adjusted wages paid in each CBSA. Furthermore, even if hypothetically changes due to the COVID-19 PHE did differentially impact the occupational mix adjusted wage index over time, it is not clear how those changes could be isolated from changes due to other reasons and what an appropriate potential methodology might be to adjust the data accordingly.
Lastly, we also noted that we have not identified any significant issues with the 2022 occupational mix data itself in terms of our audits of this data. As usual, the data was audited by the MACs, and there were no significant issues reported across the data for all hospitals.
Taking all these factors into account, we stated that we believe the CY 2022 occupational mix data is the best available data to use for FY 2025 and proposed to use the CY 2022 occupational mix data for FY 2025.
We did not receive any comments with regard to the use of the CY 2022 occupational mix data for FY 2025.We are finalizing as proposed to use the CY 2022 occupational mix data for the FY 2025 wage index.
For FY 2025, we proposed to calculate the occupational mix adjustment factor using the same methodology that we have used since the FY 2012 wage index ( 76 FR 51582 through 51586 ) and to apply the occupational mix adjustment to 100 percent of the FY 2025 wage index. In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42308 ), we modified our methodology with regard to how dollar amounts, hours, and other ( print page 69277) numerical values in the unadjusted and adjusted wage index calculation are rounded, to ensure consistency in the calculation. According to the policy finalized in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42308 and 42309 ), for data that we consider to be “raw data,” such as the cost report data on Worksheets S-3, Parts II and III, and the occupational mix survey data, we continue to use these data “as is”, and not round any of the individual line items or fields. However, for any dollar amounts within the wage index calculations, including any type of summed wage amount, average hourly wages, and the national average hourly wage (both the unadjusted and adjusted for occupational mix), we round such dollar amounts to 2 decimals. We round any hour amounts within the wage index calculations to the nearest whole number. We round any numbers not expressed as dollars or hours in the wage index calculations, which could include ratios, percentages, or inflation factors, to 5 decimals. However, we continue rounding the actual unadjusted and adjusted wage indexes to 4 decimals, as we have done historically.
Similar to the method we use for the calculation of the wage index without occupational mix, salaries and hours for a multicampus hospital are allotted among the different labor market areas where its campuses are located. Table 2 associated with this final rule (which is available via the internet on the CMS website), which contains the final FY 2025 occupational mix adjusted wage index, includes separate wage data for the campuses of multicampus hospitals. We refer readers to section III.C. of the preamble of this final rule for a chart listing the multicampus hospitals and the FTE percentages used to allot their occupational mix data.
Because the statute requires that the Secretary measure the earnings and paid hours of employment by occupational category not less than once every 3 years, all hospitals that are subject to payments under the IPPS, or any hospital that would be subject to the IPPS if not granted a waiver, must complete the occupational mix survey, unless the hospital has no associated cost report wage data that are included in the proposed FY 2025 wage index. For the proposed FY 2025 wage index, we used the Worksheet S-3, Parts II and III wage data of 3,075 hospitals, and we used the occupational mix surveys of 2,950 hospitals for which we also had Worksheet S-3 wage data, which represented a “response” rate of 96 percent (2,950/3,075). For the proposed FY 2025 wage index, we applied proxy data for noncompliant hospitals, new hospitals, or hospitals that submitted erroneous or aberrant data in the same manner that we applied proxy data for such hospitals in the FY 2012 wage index occupational mix adjustment ( 76 FR 51586 ). As a result of applying this methodology, the proposed FY 2025 occupational mix adjusted national average hourly wage was $54.73.
We did not receive any comments on our proposed calculation of the occupational mix adjustment to the FY 2025 wage index. Thus, for the reasons discussed in this final rule and in the FY 2025 IPPS/LTCH PPS proposed rule, we are finalizing our proposal without modification to calculate the occupational mix adjustment factor using the same methodology that we have used since the FY 2012 wage index and to apply the occupational mix adjustment to 100 percent of the FY 2025 wage index.
Comment: A commenter asked CMS to consider not accepting occupational mix surveys that may show data that is detrimental to patient care. The commenter cited an example such as downgrading registered nurse (RN) positions to licensed practical nurse positions in a way that forces the ratio of RNs to patients to an unsafe level.
Response: We thank the commenter for their comments. We understand the commenter has concerns with regard to hospital patient care. However, as stated previously, the purpose of the occupational mix adjustment is to control for the effect of hospitals' employment choices on the wage index; not to control for hospital patient care.
Comment: We received a comment with regard to the methodology to compute the FY 2025 wage index advocating that CMS do so without an occupational mix adjustment.
Response: Section 1886(d)(3)(E) of the Act requires CMS to collect data every 3 years on the occupational mix of employees for each short-term, acute care hospital participating in the Medicare program, in order to construct an occupational mix adjustment to the wage index. Therefore, per current law, we must apply an occupational mix adjustment to the wage index. For the final FY 2025 wage index, we are using the Worksheet S-3, Parts II and III wage data of 3,074 hospitals, and we are using the occupational mix surveys of 2,956 hospitals for which we also had Worksheet S-3 wage data, which represented a “response” rate of 96 percent (2,956/3,074). For the final FY 2025 wage index, we are applying proxy data for noncompliant hospitals, new hospitals, or hospitals that submitted erroneous or aberrant data in the same manner that we applied proxy data for such hospitals in the FY 2012 wage index occupational mix adjustment ( 76 FR 51586 ). As a result of applying this methodology, the final FY 2025 occupational mix adjusted national average hourly wage is the following:
As discussed in section III.E. of the preamble of this final rule, for FY 2025, we are applying the occupational mix adjustment to 100 percent of the FY 2025 wage index. We calculated the occupational mix adjustment using data from the 2022 occupational mix survey, using the methodology described in the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51582—51586 ).
Based on the 2022 occupational mix survey data, the FY 2025 national average hourly wages for each occupational mix nursing subcategory as calculated in Step 2 of the occupational mix calculation are as follows:

The national average hourly wage for the entire nurse category is computed in Step 5 of the occupational mix calculation. Hospitals with a nurse category average hourly wage (as calculated in Step 4) of greater than the national nurse category average hourly wage receive an occupational mix adjustment factor (as calculated in Step 6) of less than 1.0. Hospitals with a nurse category average hourly wage (as calculated in Step 4) of less than the national nurse category average hourly wage receive an occupational mix adjustment factor (as calculated in Step 6) of greater than 1.0.
Based on the 2022 occupational mix survey data, we determined (in Step 7 of the occupational mix calculation) the following:
The following sections III.F.1 through III.F.4 discuss revisions to the wage index based on hospital redesignations and reclassifications. Specifically, hospitals may have their geographic area changed for wage index payment by applying for urban to rural reclassification under section 1886(d)(8)(E) of the Act (implemented at § 412.103), reclassification by the Medicare Geographic Classification Review Board (MGCRB) under section 1886(d)(10) of the Act, Lugar status redesignations under section 1886(d)(8)(B) of the Act, or a combination of the foregoing.
Under section 1886(d)(8)(E) of the Act, a qualifying prospective payment hospital located in an urban area may apply for rural status for payment purposes separate from reclassification through the MGCRB. Specifically, section 1886(d)(8)(E) of the Act provides that, not later than 60 days after the receipt of an application (in a form and manner determined by the Secretary) from a subsection (d) hospital that satisfies certain criteria, the Secretary shall treat the hospital as being located in the rural area (as defined in paragraph (2)(D)) of the State in which the hospital is located. We refer readers to the regulations at § 412.103 for the general criteria and application requirements for a subsection (d) hospital to reclassify from urban to rural status in accordance with section 1886(d)(8)(E) of the Act (such hospitals are referred to herein as “§ 412.103 hospitals”). The FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51595 through 51596 ) includes our policies regarding the effect of wage data from reclassified or redesignated hospitals. We refer readers to the FY 2024 IPPS/LTCH final rule ( 88 FR 58971 through 58977 ) for a review of our policy finalized in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49004 ) to calculate the rural floor with the wage data of urban hospitals reclassifying to rural areas under § 412.103, and discussion of our modification to the calculation of the rural wage index and its implications for the rural floor.
In the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41369 through 41374 ), we codified certain policies regarding multicampus hospitals in the regulations at §§ 412.92, 412.96, 412.103, and 412.108. We stated that reclassifications from urban to rural under § 412.103 apply to the entire hospital (that is, the main campus and its remote location(s)). We also stated that a main campus of a hospital cannot obtain Sole Community Hospital (SCH), Rural Referral Center (RRC), or Medicare Dependent Hospital (MDH) status, or rural reclassification under § 412.103, independently or separately from its remote location(s), and vice versa. In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49012 and 49013 ), we added § 412.103(a)(8) to clarify that for a multicampus hospital, approved rural reclassification status applies to the main campus and any remote location located in an urban area, including a main campus or any remote location deemed urban under section 1886(d)(8)(B) of the Act. If a remote location of a hospital is located in a different CBSA than the main campus of the hospital, it is CMS' longstanding policy to assign that remote location a wage index based on its own geographic area to comply with the statutory requirement to adjust for geographic differences in hospital wage levels (section 1886(d)(3)(E) of the Act). Hospitals are required to identify and allocate wages and hours based on FTEs for remote locations located in different CBSAs on Worksheet S-2, Part I, Lines 165 and 166 of form CMS-2552-10. In calculating wage index values, CMS identifies the allocated wage data for these remote locations in Table 2 with a “B” in the 3rd position of the CCN. These remote locations of hospitals with § 412.103 rural reclassification status in a different CBSA are identified in Table 2, and hospitals should evaluate potential wage index outcomes for their remote location(s) when withdrawing or terminating MGCRB reclassification, or canceling § 412.103 rural reclassification status.
We also note that in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59038 through 59039 ), we changed the effective date of rural reclassification for a hospital qualifying for rural reclassification under § 412.103(a)(3) by meeting the criteria for SCH status (other than being located in a rural area), and also applying to obtain SCH status under § 412.92, where eligibility for SCH classification depends on a hospital merger. Specifically, we finalized that in these circumstances, and subject to the hospital meeting the requirements set forth at § 412.92(b)(2)(vi), the effective date for rural reclassification will be the effective date set forth in § 412.92(b)(2)(vi).
Finally, we remind hospitals currently located in rural areas becoming urban under the adoption of the revised OMB delineations that if they have SCH, MDH, or RRC status, they may choose to apply for a § 412.103 urban to rural reclassification if qualifying criteria are met to maintain ( print page 69279) the SCH, MDH, or RRC status. We advise hospitals to evaluate their options and if desired, apply for § 412.103 urban to rural reclassification before the beginning of FY 2025, to avoid a lapse in SCH, MDH, or RRC status at the beginning of FY 2025 should we finalize our proposal to adopt the revised OMB delineations.
Comment: A commenter suggested that CMS remove the 1 year waiting period required by § 412.103(g)(4) for hospitals to cancel rural reclassification. The commenter stated that CMS' concern in promulgating the policy is no longer applicable due to its choice to link the rural floor and rural wage index as one calculation. The commenter asserted that this rule, which was finalized in FY 2022 IPPS rulemaking, was intended to disincentivize hospitals from cancelling their rural reclassification before the lock-in date at 412.103(b)(6), and then obtaining a new rural reclassification after the lock-in date, so the hospital could receive the rural wage index without having its wage data included in the rural wage index calculation (effectively receiving a higher rural wage index than if its wage data was included).
Response: We appreciate the commenter's input. We did not propose any changes to § 412.103(g)(4), because we still believe that the 1 year waiting period to cancel rural reclassifications is relevant. While the incentive to game the rural wage index may be less now that the rural wage index is the same as the rural floor, as the commenter described, hospitals can still attempt to maximize payment by strategically timing cancellation of a § 412.103 rural reclassification. Hospitals may choose to hold a § 412.103 rural reclassification for a variety of reasons, such as the 340B drug pricing program administered by HRSA, SCH or RRC eligibility, or to use rural criteria for reclassifying through the MGCRB under § 412.230. Without the minimum waiting period at § 412.103(g)(4), such a hospital could cancel its § 412.103 rural reclassification each year in time to not be included in the rural floor calculation (for example, if the hospital expects the inclusion of its wage data would lower the calculation), and then obtain a new § 412.103 rural reclassification after the lock-in date. We continue to believe that including § 412.103 rural reclassifications in the rural wage index calculation for at least one fiscal year before they may be canceled will help to ensure consistency and predictability of wage index values.
Section 1886(d)(8)(E) of the Act describes criteria for hospitals located in urban areas to be treated as being located in a rural area of their state. The criterion at section 1886(d)(8)(E)(ii)(I) of the Act requires that the hospital be located in a rural census tract of a metropolitan statistical area (as determined under the most recent modification of the Goldsmith Modification, originally published in the Federal Register on February 27, 1992 ( 57 FR 6725 )).
This condition is implemented in the regulation at § 412.103(a)(1), which currently states: “the hospital is located in a rural census tract of a Metropolitan Statistical Area (MSA) as determined under the most recent version of the Goldsmith Modification, the Rural-Urban Commuting Area codes, as determined by the Office of Rural Health Policy (ORHP) of the Health Resources and Services Administration (HRSA), which is available via the ORHP website at: http://www.ruralhealth.hrsa.gov or from the U.S. Department of Health and Human Services, Health Resources and Services Administration, Office of Rural Health Policy, 5600 Fishers Lane, Room 9A-55, Rockville, MD 20857.”
The Goldsmith Modification [ 190 ] was originally designed to identify rural census tracts located in Metropolitan counties for purposes of grant eligibility unrelated to the hospital IPPS but were incorporated by section 1886(d)(8)(E)(ii)(I) of the Act for purposes related to the hospital wage index.
The Federal Office of Rural Health Policy (FORHP) (known as ORHP in § 412.103) later funded development of Rural-Urban Commuting Area (RUCA) codes via the U.S. Department of Agriculture's (USDA) Economic Research Service as the latest version of the Goldsmith Modification, described in a May 3, 2007 Federal Register notice ( 72 FR 24589 ), to address limitations of the original Goldsmith Modification. RUCAs, like the Goldsmith Modification, are based on a sub-county unit, the census tract, permitting a finer delineation of what constitutes rural areas inside Metropolitan areas ( 72 FR 24590 ). In that notice, HRSA stated it believes that the use of RUCAs allows more accurate targeting of resources intended for the rural population to determine programmatic eligibility for rural areas inside of Metropolitan counties. Using data from the Census Bureau, every census tract in the United States is assigned a RUCA code. In the May 3, 2007 Federal Register , HRSA stated that FORHP considers all census tracts with RUCA codes 4-10 to be rural, plus an additional 132 large area census tracts with RUCA codes 2 or 3 ( 72 FR 24591 ). They also stated that FORHP will continue to seek refinements in the use of RUCAs.
FORHP has since published a revised definition of eligibility for rural health grants for FY 2022 in a January, 12, 2021 Federal Register Notice ( 86 FR 2418 through 2420 ). Specifically, FORHP added Metropolitan Statistical Area (MSA) counties that contain no Urbanized Area (UA) [ 191 ] to the areas eligible for the rural health grant programs. FORHP did not remove any areas from the rural definition in the FY 2022 Federal Register Notice.
It has come to our attention that our current regulation text at § 412.103(a)(1) does not describe FORHP's expanded definition of a “rural area” from the FY 2022 Federal Register Notice. In addition, § 412.103(a)(1) contains a web link that is no longer active and requires updating. We believe the current rural definition used by FORHP for purposes of the rural health grant program constitutes “the most recent modification of the Goldsmith Modification” referred to in the statute, since the expanded definition of rural constitutes a refinement to the use of RUCA codes, which were developed as the latest version of the Goldsmith Modification. As stated in the FY 2022 Federal Register Notice ( 86 FR 2420 ), the expanded criteria reflect FORHP's desire to accurately identify areas that are rural in character using a data-driven methodology that relies on existing geographic identifiers and utilizes standard, national level data sources. Therefore, we proposed to amend our regulation text at § 412.103(a)(1) to provide a reference to the most recent Federal Register notice issued by HRSA defining “rural areas.” In this way, there will be no need to update the Medicare regulations if FORHP develops a further modification of the Goldsmith Modification or if the weblink changes. FORHP has published the current link in the Federal Register notice ( 86 FR 2418-2420 ) along with the most recent revisions to the current complete rural definition, and it is ( print page 69280) available via the Rural Health Grants Eligibility Analyzer at https://data.hrsa.gov/tools/rural-health .
We proposed to amend the regulation text at 412.103(a)(1) to read: the hospital is located in a rural census tract of a Metropolitan Statistical Area (MSA) as determined under the most recent version of the Goldsmith Modification, using the Rural-Urban Commuting Area codes and additional criteria, as determined by the Federal Office of Rural Health Policy (FORHP) of the Health Resources and Services Administration (HRSA), which is available at the web link provided in the most recent Federal Register notice issued by HRSA defining rural areas.
We did not receive any comments on this proposal and are finalizing as proposed to amend the regulation text at § 412.103(a)(1).
In the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49499 through 49500 ), CMS discussed its longstanding policy to terminate MGCRB wage index reclassification status under section 1886(d)(10) of the Act for hospitals with terminated CMS certification numbers (CCN). We determined that it would be appropriate to terminate the MGCRB reclassification status for these hospitals (with a limited exception for certain locations acquired by another hospital in a different CBSA), as the hospital may no longer be able to make timely and informed decisions regarding reclassification statuses.
At the time, we did not articulate a similar policy for hospitals reclassified as rural under § 412.103. While policies regarding MGCRB reclassification were adopted for purposes related to the hospital wage index, § 412.103 reclassifications may have broader implications. At the time the policy to terminate MGCRB reclassifications for hospitals with terminated CCNs was implemented, § 412.103 reclassifications were less common, and generally had negligible effects on State rural wage index values. Prior to FY 2024, as a result of various wage index value hold-harmless policies, discussed in detail in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58973-58974 ), § 412.103 hospital data rarely affected a state's final rural wage index value. Under the current policy first implemented in FY 2024, however, § 412.103 hospital data is only excluded from the rural wage index when indicated by the hold harmless provision at section 1886(d)(8)(C)(ii) of the Act. Hospitals reclassified under § 412.103 now impact the rural wage index value of most states. We refer readers to the FY 2024 IPPS/LTCH final rule ( 88 FR 58973 through 58977 ) for discussion on how CMS finalized the current policy to include the wage index data for § 412.103 hospitals in more iterations of the rural wage index calculation. Furthermore, following the policy implemented in the April 21, 2016, interim final rule with comment period (IFC) ( 81 FR 23428 through 23438 ), which allowed hospitals to maintain dual § 412.103 and MGCRB reclassification status, the number of rural reclassifications has grown significantly. We now believe it is appropriate to propose a policy regarding terminated or “tied-out” hospitals, effective for FY 2025, to address our concerns regarding the impacts these hospitals would have on rural wage index values. Therefore, we proposed that § 412.103 reclassifications will be considered cancelled for the purposes of calculating the area wage index for any hospital with a CCN listed as terminated or “tied-out” as of the date that the hospital ceased to operate with an active CCN. We propose to obtain and review the best available CCN termination status lists as of the § 412.103(b)(6) “lock-in” date (60 days after the proposed rule for the FY is displayed in the Federal Register ). The lock-in date is used to determine whether a hospital has been approved for § 412.103 reclassification in time for that status to be included in the upcoming year's wage index development. We believe using this date for evaluating CCN terminations would be consistent with the wage index development timeline.
As stated previously, § 412.103 reclassification may have other implications for hospital status and payment. Hospitals may obtain rural reclassification for several reasons, such as to convert to a Critical Access Hospital (CAH), or to obtain SCH status. Eligibility requirements for Rural Emergency Hospital (REH) qualification under section 1861(kkk)(3) of the Act included a reference to reclassification under section 1886(d)(8)(E) (implemented by § 412.103). We note that our proposal to consider § 412.103 reclassifications cancelled for the purposes of calculating area wage index for any hospital with a CCN listed as terminated or “tied-out” is not intended to alter or affect the qualification for such statuses or to have other effects unrelated to hospital wage index calculations. The rural reclassification status would remain in effect for any period that the original PPS hospital remains in operation with an active CCN. For REH qualification requirement purposes, this would include the date of enactment of the Consolidated Appropriations Act, 2021 ( Pub. L. 116-260 ), which was December 27, 2020. We believe this policy provides consistency and predictability in wage index values.
Comment: Commenters were supportive of our proposed policy to cancel the rural reclassification status for hospitals with terminated (“tied-out”) CCNs. Commenters reiterated CMS' concern that these hospitals may no longer be able to make timely and informed decisions regarding their reclassification status.
Response: We thank commenters for their support and are finalizing the proposed policy to consider rural reclassifications to be cancelled for the purposes of calculating the area wage index for any hospital with a CCN listed as terminated or “tied-out” as of the date that the hospital ceased to operate with an active CCN. CMS will obtain and review the best available CCN termination status lists as of the § 412.103(b)(6) “lock-in” date (60 days after the proposed rule for the FY is displayed in the Federal Register ).
Under section 1886(d)(10) of the Act, the MGCRB considers applications by hospitals for geographic reclassification for purposes of payment under the IPPS. Hospitals must apply to the MGCRB to reclassify not later than 13 months prior to the start of the fiscal year for which reclassification is sought (usually by September 1). Generally, hospitals must be proximate to the labor market area to which they are seeking reclassification and must demonstrate characteristics similar to hospitals located in that area. The MGCRB issues its decisions by the end of February for reclassifications that become effective for the following fiscal year (beginning October 1). The regulations applicable to reclassifications by the MGCRB are located in §§ 412.230 through 412.280. (We refer readers to a discussion in the FY 2002 IPPS final rule ( 66 FR 39874 and 39875 ) regarding how the MGCRB defines mileage for purposes of the proximity requirements.) The general policies for reclassifications and redesignations and the policies for the effects of hospitals' reclassifications and redesignations on the wage index are discussed in the FY 2012 IPPS/LTCH PPS final rule for the FY 2012 final wage index ( 76 FR 51595 and 51596 ).
In addition, in the FY 2012 IPPS/LTCH PPS final rule, we discussed the effects on the wage index of urban ( print page 69281) hospitals reclassifying to rural areas under § 412.103. In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42332 through 42336 ), we finalized a policy to exclude the wage data of urban hospitals reclassifying to rural areas under § 412.103 from the calculation of the rural floor, but we reverted to the pre-FY 2020 policy in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49002 through 49004 ). Hospitals that are geographically located in States without any rural areas are ineligible to apply for rural reclassification in accordance with the provisions of § 412.103.
On April 21, 2016, we published an interim final rule with comment period (IFC) in the Federal Register ( 81 FR 23428 through 23438 ) that included provisions amending our regulations to allow hospitals nationwide to have simultaneous § 412.103 and MGCRB reclassifications. For reclassifications effective beginning FY 2018, a hospital may acquire rural status under § 412.103 and subsequently apply for a reclassification under the MGCRB using distance and average hourly wage criteria designated for rural hospitals. In addition, we provided that a hospital that has an active MGCRB reclassification and is then approved for redesignation under § 412.103 will not lose its MGCRB reclassification; such a hospital receives a reclassified urban wage index during the years of its active MGCRB reclassification and is still considered rural under section 1886(d) of the Act for other purposes.
We discussed that when there is both a § 412.103 redesignation and an MGCRB reclassification, the MGCRB reclassification controls for wage index calculation and payment purposes. Prior to FY 2024, we excluded hospitals with § 412.103 redesignations from the calculation of the reclassified rural wage index if they also have an active MGCRB reclassification to another area. That is, if an application for urban reclassification through the MGCRB is approved and is not withdrawn or terminated by the hospital within the established timelines, we consider the hospital's geographic CBSA and the urban CBSA to which the hospital is reclassified under the MGCRB for the wage index calculation. We refer readers to the April 21, 2016 IFC ( 81 FR 23428 through 23438 ) and the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56922 through 56930 ), in which we finalized the April 21, 2016 IFC, for a full discussion of the effect of simultaneous reclassifications under both the § 412.103 and the MGCRB processes on wage index calculations. For FY 2024 and subsequent years, we refer readers to section III.G.1 of the preamble of the FY 2024 IPPS/LTCH PPS final rule for discussion of our policy to include hospitals with a § 412.103 redesignation that also have an active MGCRB reclassification to another area in the calculation of the reclassified rural wage index ( 88 FR 58971 through 58977 ).
Comment: A commenter explained that due to the lag in IPPS rulemaking where the cost report data used to set rates can be from up to four years prior, there is a period of up to 4 years in which there is no AHW data associated with a newly created “B” campus in the case of a new multicampus hospital. The commenter encouraged CMS to close the lag of up to four years during which a newly merged provider is ineligible to receive a new MGCRB reclassification because there is no AHW data associated with the “B” provider number. The commenter suggested that CMS amend the regulations at § 412.230(d)(2) to provide that when a new owner accepts assignment of the existing hospital's Medicare provider agreement, or in the case of a common ownership provider consolidation in which a new subcampus provider number is created, the wage data associated with the previous hospital's provider number can be used in calculating the new hospital's 3-year average hourly wage until such time as at least 1 year of wage data is accumulated under the new subprovider.
Response: We did not propose any modifications to the regulations at § 412.230(d)(2) and consider this comment out of scope of the proposed rule. We may consider revisiting our policies in future rulemaking to address the scenario of newly merged providers. We note that, as described in section III.G.6, remote locations with “B” provider number are eligible to receive a 5 percent cap on annual wage index decreases relative to the wage index assigned in the prior fiscal year.
Comment: A couple of commenters asked CMS to revise the regulations for appropriate proximity data at § 412.230(c)(1) to include waterways travelled by ferry boat as travel over an improved road. These commenters stated that each year, the MGCRB denies reclassification requests based on use of a ferry route to meet the proximity criteria, and these decisions are overturned via administrative appeal. The commenters urged CMS to eliminate unnecessary appeals by clarifying in the regulations that distance traveled by ferry boats is included when calculating proximity for reclassification requests.
Response: We agree with the commenter that a modification to § 412.230(c)(1) to address waterways travelled by ferry boat could reduce administrative appeals. However, we did not propose any modifications to the regulations at § 412.230(c)(1) and are not finalizing any changes in this final rule. We note that a potential future proposal to modify § 412.230(c)(1) could contemplate whether the MGCRB should include or exclude the distances traveled via ferry boats for purposes of determining proximity during its review of reclassification requests.
On May 10, 2021, we published an interim final rule with comment period (IFC) in the Federal Register ( 86 FR 24735 through 24739 ) that included provisions amending our regulations to allow hospitals with a rural redesignation to reclassify through the MGCRB using the rural reclassified area as the geographic area in which the hospital is located. We revised our regulation so that the redesignated rural area, and not the hospital's geographic urban area, is considered the area a § 412.103 hospital is located in for purposes of meeting MGCRB reclassification criteria, including the average hourly wage comparisons required by § 412.230(a)(5)(i) and (d)(1)(iii)(C). Similarly, we revised the regulations to consider the redesignated rural area, and not the geographic urban area, as the area a § 412.103 hospital is located in for purposes of applying the prohibition at § 412.230(a)(5)(i) on reclassifying to an area with a pre-reclassified average hourly wage lower than the pre-reclassified average hourly wage for the area in which the hospital is located. Effective for reclassification applications due to the MGCRB for reclassification beginning in FY 2023, a § 412.103 hospital could apply for a reclassification under the MGCRB using the State's rural area as the area in which the hospital is located. We refer readers to the May 10, 2021 IFC ( 86 FR 24735 through 24739 ) and the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45187 through 45190 ), in which we finalized the May 10, 2021 IFC, for a full discussion of these policies.
In a comment on the May 10, 2021 IFC ( 86 FR 24735 through 24739 ), a commenter noted that the IFC states that a hospital reclassified under § 412.103 could potentially reclassify to any area with a pre-reclassified average hourly wage that is higher than the pre-reclassified average hourly wage for the rural area of the state for purposes of the regulation at § 412.230(a)(5)(i). The commenter asserted that CMS' use of ( print page 69282) the word “could” in this context seems to suggest that CMS would allow the hospital to use either its home average hourly wage or the rural average hourly wage for purposes of the regulation at § 412.230(a)(5)(i). The commenter suggested that CMS allow both comparison options, because the rural average hourly wage may occasionally be higher than the hospital's home urban area's average hourly wage.
In response, we clarified that the commenter's interpretation of our policy is correct. We stated that while the court's decision in Bates County Memorial Hospital v. Azar requires CMS to permit hospitals to reclassify to any area with a pre-reclassified average hourly wage that is higher than the pre-reclassified average hourly wage for the rural area of the state, we do not believe that we are required to limit hospitals from using their geographic home area for purposes of the regulation at § 412.230(a)(5)(i). Therefore, we clarified that we would allow hospitals to reclassify to an area with an average hourly wage that is higher than the average hourly wage of either the hospital's geographic home area or the rural area ( 86 FR 45189 ).
While we clarified our policy in response to the aforementioned comment, the regulation text inadvertently was not similarly clarified to reflect this policy. Therefore, we proposed to revise the regulation text at § 412.230(a)(5)(i) to reflect our policy clarified in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45189 ). While it has been CMS' policy to allow a § 412.103 hospital to use either its geographic area or the rural area of the state for purposes of § 412.230(a)(5)(i), we believe that synchronizing the regulation text with our policy clarified in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45189 ) is necessary for consistency and to reduce unnecessary administrative appeals.
Specifically, we proposed to replace the phrase in the regulation at § 412.230(a)(5)(i) that reads “in the rural area of the state” with the phrase “either in its geographic area or in the rural area of the state.” Section 412.230(a)(5)(i) with this proposed revision would read: An individual hospital may not be redesignated to another area for purposes of the wage index if the pre-reclassified average hourly wage for that area is lower than the pre-reclassified average hourly wage for the area in which the hospital is located. An urban hospital that has been granted redesignation as rural under § 412.103 is considered to be located either in its geographic area or in the rural area of the state for the purposes of this paragraph (a)(5)(i).
Comment: A commenter supported this proposal to revise the regulations at § 412.230(a)(5)(i), stating that it would promote consistency between CMS policy and MGCRB practice by eliminating unnecessary administrative appeals.
Response: We appreciate the commenter's support. In consideration of the public comment received, we are finalizing our proposal to revise the regulations at § 412.230(a)(5)(i) as proposed without modification.
As previously stated, under section 1886(d)(10) of the Act, the MGCRB considers applications by hospitals for geographic reclassification for purposes of payment under the IPPS. The specific procedures and rules that apply to the geographic reclassification process are outlined in regulations under 42 CFR 412.230 through 412.280 . There are 470 hospitals approved for wage index reclassifications by the MGCRB starting in FY 2025. Because MGCRB wage index reclassifications are effective for 3 years, for FY 2025, hospitals reclassified beginning in FY 2023 or FY 2024 are eligible to continue to be reclassified to a particular labor market area based on such prior reclassifications for the remainder of their 3-year period. There were 256 hospitals approved for wage index reclassifications in FY 2023 that will continue for FY 2025, and 352 hospitals approved for wage index reclassifications in FY 2024 that will continue for FY 2025. Of all the hospitals approved for reclassification for FY 2023, FY 2024, and FY 2025, 1,078 hospitals (approximately 32.5 percent of IPPS hospitals) are in a MGCRB reclassification status for FY 2025 (with 237 of these hospitals reclassified back to their urban geographic location). We refer readers to Section III.F.3.b of this final rule for information on the effects of implementation of new OMB labor market area delineations on reclassified hospitals.
Under the existing regulations at § 412.273, hospitals that have been reclassified by the MGCRB are permitted to withdraw their applications if the request for withdrawal is received by the MGCRB any time before the MGCRB issues a decision on the application, or after the MGCRB issues a decision, provided the request for withdrawal is received by the MGCRB within 45 days of the date that CMS' annual notice of proposed rulemaking is issued in the Federal Register concerning changes to the inpatient hospital prospective payment system and proposed payment rates for the fiscal year for which the application has been filed. Please note that Section III.F.3.c. of this final rule finalizes our proposal to change the deadline for the withdrawal requests to 45 days from the date of filing for public inspection of the proposed rule at the website of the Office of the Federal Register.
For information about the current process for withdrawing, terminating, or canceling a previous withdrawal or termination of a 3-year reclassification for wage index purposes, we refer readers to § 412.273, as well as the FY 2002 IPPS final rule ( 66 FR 39887 through 39888 ) and the FY 2003 IPPS final rule ( 67 FR 50065 through 50066 ). Additional discussion on withdrawals and terminations, and clarifications regarding reinstating reclassifications and “fallback” reclassifications were included in the FY 2008 IPPS final rule ( 72 FR 47333 ) and the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38148 through 38150 ).
Applications for FY 2026 reclassifications are due to the MGCRB by September 1, 2024. This is also the current deadline for canceling a previous wage index reclassification withdrawal or termination under § 412.273(d) for the FY 2025 cycle.
Applications and other information about MGCRB reclassifications may be obtained beginning in mid-July 2024 via the internet on the CMS website at https://www.cms.gov/medicare/regulations-guidance/geographic-classification-review-board . This collection of information was previously approved under OMB Control Number 0938-0573, which expired on January 31, 2021. A reinstatement of this PRA package is currently being developed. The public will have an opportunity to review and submit comments regarding the reinstatement of this PRA package through a public notice and comment period separate from this rulemaking.
Comment: A commenter asked that CMS issue additional guidance to provide clarity for the process and timeline of MGCRB decisions, noting that there is no limit in how early the MGCRB can issue its decisions. The commenter requested that CMS prohibit the MGCRB from issuing decisions prior to the first week of February to allow hospitals ample time to submit documentation of rural reclassification, SCH and RRC status in support of their reclassification applications, or to submit withdrawals based on the ( print page 69283) January PUF. The commenter also suggested that to alleviate the burden of hospitals appealing MGCRB decisions, CMS could modify § 412.256(c) to provide for the MGCRB to also issue requests for additional information rather than deny applications due to incomplete information or if the MGCRB maps distance for proximity differently than the hospital's submission.
Response: We disagree with the commenter that CMS should limit how early the MGCRB can issue its decisions to provide time for hospitals to submit additional documentation. According to § 412.256(a)(2), a complete application must be received not later than the first day of the 13-month period preceding the Federal fiscal year for which reclassification is requested. Hospitals could avoid a denial due to incomplete information or avoid an administrative appeal by submitting a complete application at the time of filing, rather than relying on the MGCRB's current practice of accepting supporting documentation up until the date of review. Hospitals wishing to withdraw based on the January PUF can still withdraw after the MGCRB's decision in accordance with the regulations at § 412.273.
With regard to the commenter's suggested revision to the regulation at § 412.256(c), we did not propose any modifications to the regulations at § 412.256(c) and believe that the current regulation at § 412.256(c) already provides for a robust and transparent process. Specifically, the regulation at 412.256(c)(1) states: “The MGCRB will review an application within 15 days of receipt to determine if the application is complete. If the MGCRB determines that an application is incomplete, the MGCRB will notify the hospital, with a copy to CMS, within the 15 day period, that it has determined that the application is incomplete and may dismiss the application if a complete application is not filed by September 1.” We reiterate that a hospital can avoid the administrative burden of an appeal by submitting a complete application at the time of filing.
Reclassifications granted under section 1886(d)(10) of the Act are effective for 3 fiscal years, so that a hospital or county group of hospitals would be assigned a wage index based upon the wage data of hospitals in the labor market area to which it reclassified for a 3-year period. Because hospitals that have been reclassified beginning in FY 2023, 2024, or 2025 were reclassified based on the current labor market delineations, under the revised OMB delineations based on the OMB Bulletin No. 23-01 beginning in FY 2025 the CBSAs to which they have been reclassified, or the CBSAs where they are located, may change. In the proposed rule, we encouraged hospitals with current reclassifications to verify area wage indexes in Table 2 in the appendix, and to confirm that the CBSAs to which they have been reclassified for FY 2025 would continue to provide a higher wage index than their geographic area wage index. Hospitals were able to withdraw or terminate their FY 2025 reclassifications by contacting the MGCRB within 45 days from the date the proposed rule was issued in the Federal Register (§ 412.273(c)).
We proposed that in the case where a CBSA adds a current rural county, or loses a current constituent rural county, a hospital's current reclassification to the resulting CBSA would be maintained. In some cases, a hospital may be located in a rural county that would join the CBSA to which the hospital is reclassified. We note that in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 49977 ), CMS terminated reclassifications when, as a result of adopting the revised OMB delineations, a hospital's geographic county was located in the CBSA for which it was approved for MGCRB reclassification. At that time, there was no means for a hospital to obtain an MGCRB reclassification to its own geographic area (which we refer to as “home area” reclassifications). However, as discussed in the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56925 ), “home area” reclassifications have since become possible as a result of the change in policy in the 2016 IFC ( 81 FR 23428 through 23438 ) discussed earlier allowing for dual reclassifications. We therefore do not believe it is necessary to terminate these reclassifications as we did in FY 2015. In general, once the MGCRB has approved a reclassification in accordance with subpart L of 42 CFR part 412 , that reclassification remains in place for 3 years (see § 412.274(b)(2)) unless terminated by the hospital pursuant to § 412.273, and CMS does not reevaluate whether the hospital continues to meet the criteria for reclassification during the three-year period. As such, we proposed to maintain these as “home area” reclassifications instead of terminating them.
If a county is removed from a CBSA and becomes rural, a hospital in that county with a current “home area” reclassification would no longer be geographically located in the CBSA to which they are reclassified. We proposed that these reclassifications would no longer be considered “home area” reclassifications, and the hospital would be assigned the wage index applicable to other hospitals that reclassify into the CBSA (which may be lower than the wage index calculated for hospitals geographically located in the CBSA due to the hold harmless provision at section 1886(d)(8)(C)(i) of the Act). [ 192 ]
Finally, as discussed in section III.B.4, all the constituent counties of CBSA 14100 (Bloomsberg-Berwick, PA), CBSA 19180 (Danville, IL), CBSA 20700 (East Stroudsburg, PA) and CBSA 35100 (New Bern, NC) become rural under the revised OMB delineations. There are 6 hospitals with reclassifications to these previously urban CBSAs.

As there is no sufficiently similar urban CBSA in the revised delineations, we proposed that hospitals' MGCRB reclassifications to these CBSAs would be terminated for FY 2025. The effect of such terminations would be that these hospitals would receive the wage index for the CBSA in which they are geographically located, or in the case of hospitals with § 412.103 reclassification, the rural wage index. While we would prefer to maintain the remaining years of a MGCRB reclassification and transition these reclassified hospitals to the most appropriate CBSA under the revised delineations, because there are no urban counties remaining in the CBSAs listed above to which they are currently reclassified, there is no urban area to which they can be assigned that includes at least one county from the CBSA to which the MGCRB approved reclassification. We received no comments regarding our proposed policy to maintain MGCRB reclassification to a CBSA that either gains or loses one or more counties to or from a rural area, nor did we receive comments regarding our proposed policy for addressing home area reclassifications in these areas. We are finalizing these policies as proposed.
Comment: A commenter described the treatment of the hospitals that had active MGCRB reclassifications through FY 2025 to CBSAs where all constituent counties become rural under the revised OMB delineations as unfair. The commenter stated the proposal to terminate these reclassifications without reassignment to another urban area disadvantages certain hospitals. The commenter contended that as many as four hospitals will be assigned a lower wage index based on their state's rural wage index or rural floor value. The commenter noted, as discussed above, that CMS does not generally reevaluate whether the hospital continues to meet the criteria for reclassification during the three-year period approved by the MGCRB. The commenter also cited impacts on the state rural wage index due to the requirement under section 1886(d)(8)(C)(ii) of the Act to exclude wage data for urban hospitals with dual § 412.103 and MGCRB reclassifications in calculating the rural wage index unless doing reduces the rural wage index. The commenter stated that by terminating reclassifications in this manner, CMS has disadvantaged these hospitals by limiting their actions when it comes to their preferred wage index area. The commenter provided several alternative methods to assign the reclassification for these hospitals, including assigning the reclassification to their “home” geographic area, the next closest CBSA, or another CBSA to which the hospital can demonstrate it would meet reclassification criteria, or would have a high level of commuting interchange.
Response: We considered the commenter's concern and alternative suggestions to avoid terminating the MGCRB reclassifications. As we discussed previously, once approved by the MGCRB, a reclassification to the approved area is valid for a period of three fiscal years and generally is not subject to review. However, as discussed later in this section, we believe that when the CBSA to which reclassification was approved is substantially changed due to the adoption of revised labor market delineations, in order to continue to give effect to the approved reclassification, CMS should identify which area best represents the urban labor market to which a hospital's reclassification was approved. That is, when the labor market area delineations are updated, the new delineations may or may not contain a CBSA resembling that to which a hospital was previously reclassified. Where possible, CMS assigns a hospital's reclassification to a CBSA that contains the nearest urban county that was previously located in the CBSA to which the MGCRB approved reclassification or to another nearby CBSA that contains at least one urban county from the approved CBSA. In the case of these hospitals, which had reclassified to urban CBSAs, this is not possible, as no part of their approved CBSA would remain urban under the revised delineations. Furthermore, section 1886(d)(10)(C) of the Act indicates that the Board is responsible for reviewing and approving MGCRB applications, and CMS's policy aims to give effect only to reclassifications approved by the Board. By assigning a reclassified hospital to a CBSA that contains at least one urban county from its previously approved CBSA, we believe that we are substantively maintaining an existing approved reclassification. We do not believe it would be possible to assign a hospital temporarily to another CBSA (as suggested by the commenter) in an equitable manner. Any number of hospitals might hypothetically be eligible for MGCRB reclassification to different labor markets due to changes to labor market delineations and would potentially request immediate reclassification by CMS, rather than waiting at least one fiscal year to apply to the MGCRB. As stated in the proposed rule, we believe that the 5 percent cap on annual decreases in wage index values provides for an adequate transition for any hospitals that are negatively affected by the adoption of the revised OMB labor market delineations. CMS evaluated the impacts on the hospitals that the commenter asserted would be negatively impacted by our proposal to terminate their MGCRB reclassifications (listed in the table above). We find minimal impact on their wage index values for FY 2025. The wage index values for the six hospitals for which we proposed to terminate the reclassification are all increasing in FY 2025 compared to FY 2024 (in amounts ranging from 1.7 to 9.7 percent). While some of these hospitals may have been able to obtain higher wage index values by having an MGCRB reclassification to another urban area, the overall benefits would be nominal.
Furthermore, while we acknowledge that dual § 412.103 and MGCRB reclassification status has an impact on the rural wage index, as described in detail in the FY 2024 final rule ( 88 FR 58971 through 58977 ), we are not convinced that this impact warrants any special exception or treatment by CMS. ( print page 69285) Section 1886(d)(8)(C)(ii) of the Act ensures that the effects of MGCRB and “Lugar” reclassification policies do not reduce the rural wage index. In the case of a dual reclass hospital losing its MGCRB reclassification, each hospital had adequate time to cancel its § 412.103 reclassification by the June 9, 2024 deadline, if preferred. We believe this option allowed hospitals to evaluate whether the benefits of rural reclassification outweighed any negative impact its wage data would have on the rural wage index calculation. We do not believe our approach of terminating the reclassifications of hospitals that had reclassified to CBSAs that have no comparator under the revised OMB delineations negatively impacts the overall accuracy of the IPPS wage index.
For these reasons, CMS will not adopt any of the alternative reclassification assignment approaches suggested by the commenter. Each recommendation requires CMS to effectively initiate and approve a new MGCRB reclassification. In each recommended option, no part of any CBSA that could be assigned was included in the original application approved by the MGCRB. We are finalizing the policy to terminate MGCRB reclassifications in cases where the CBSA to which a hospital's reclassification was approved became rural under the revised OMB delineations adopted in this final rule.
We proposed that in the case of a CBSA that experiences a change in CBSA number, or where all urban counties in the CBSA are subsumed by another CBSA, MGCRB reclassifications approved to the FY 2024 CBSA would be assigned the revised FY 2025 CBSA (as described in the section III.B.6). In some cases, this reconfiguration of CBSAs would result in an MGCRB reclassification approved to a different area becoming a “home area” reclassification, if a hospital's current geographic urban CBSA is subsumed by its reclassified CBSA. Otherwise, the current reclassification would continue to the proposed revised CBSA number.
We did not receive any comments specific to this proposal and are finalizing this policy to assign the revised CBSA number to hospitals reclassified to a CBSA where the CBSA number changes, or the CBSA is subsumed by another CBSA.
In some cases, adopting the revised OMB delineations would result in one or more counties splitting apart from their current CBSAs to form new CBSAs, or counties shifting from one CBSA designation to another CBSA. If CBSAs are split apart, or if counties shift from one CBSA to another under the revised OMB delineations, for hospitals that have reclassified to these CBSAs we must determine which reclassified area to assign to the hospital for the remainder of a hospital's 3-year reclassification period.
Consistent with the policy implemented in FY 2021 ( 85 FR 58743 through 58753 ), we proposed to assign current “home area” reclassifications to these CBSAs to the hospital's geographic CBSA. That is, hospitals that were approved for MGCRB reclassification to the geographic area they are located in effective for FYs 2023, 2024, or 2025 would continue to be assigned a reclassification to their geographic “home area.” The assigned “home area” reclassification CBSA may be different from previous years if the hospital is located in a county that was relocated to a new or different urban CBSA.
The following is a table of hospitals with current “home area” reclassification to CBSAs where one or more counties move to a new or different urban CBSA under the revised OMB delineations. The reclassification noted by an asterisk on the “MGCRB Case Number” was withdrawn for FY 2025, but may be reinstated for FY 2026.

Consistent with the policy CMS implemented in the FY 2005 IPPS final rule ( 69 FR 49054 through 49056 ), the FY 2015 IPPS final rule ( 79 FR 49973 through 49977 ), and in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58743 through 58753 ), for FY 2025, if a CBSA would be reconfigured due to adoption of the revised OMB delineations and it would not be possible for the reclassification to continue seamlessly to the reconfigured CBSA (not including ( print page 69286) “home area” reclassifications, which were discussed previously), we believe it would be appropriate for us to determine the best alternative location to assign current reclassifications for the remaining 3 years. Therefore, to maintain the integrity of a hospital's 3-year reclassification period, we proposed that current geographic reclassifications (applications approved effective for FY 2023, FY 2024, or FY 2025) that would be affected by CBSAs that are split apart or counties that shift to another CBSA under the revised OMB delineations, would ultimately be assigned to a CBSA under the revised OMB delineations that contains at least one county (or county equivalent) from the reclassified CBSA under the current FY 2024 delineations, and that would be generally consistent with rules that govern geographic reclassification. That is, consistent with the policy finalized in FY 2015 ( 79 FR 49973 ) we proposed a policy that other affected reclassified hospitals be assigned to a CBSA that would contain the most proximate county that (1) is located outside of the hospital's proposed FY 2025 geographic labor market area, and (2) is part of the original FY 2024 CBSA to which the hospital is reclassified. We believe that assigning reclassifications to the CBSA that contains the nearest county that meets the aforementioned criteria satisfies the statutory requirement at section 1886(d)(10)(D)(v) of the Act by maintaining reclassification status for a period of 3 fiscal years, while generally respecting the longstanding principle of geographic proximity in the labor market reclassification process. For county group reclassifications, we proposed that we would follow the same policy, except that we would reassign hospitals in a county group reclassification to the CBSA under the revised OMB delineations that contains the county to which the majority of hospitals in the group reclassification are geographically closest. We also proposed to allow such hospitals, or county groups of hospitals, to submit a request to the [email protected] mailbox for reassignment to another proposed CBSA that would contain a county that is part of the current CBSA to which it was approved to be reclassified (based on FY 2024 delineations) if the hospital or county group of hospitals can demonstrate compliance with applicable reclassification proximity rules, as described later in this section.
The following Table X provides a list of current FY 2024 CBSAs (column 1) where one or more counties would be relocated to a new or different urban CBSA under the proposed policy. Hospitals with active MGCRB reclassifications into the current FY 2024 CBSAs in column 1 would be subject to the reclassification assignment policy described in this subsection. The third column of “eligible” CBSAs lists all proposed revised CBSAs that contain at least one county that is part of the current FY 2024 CBSA (in column 1).

We did not receive any comments regarding the MGCRB reclassification assignment and reassignment policy. We are finalizing the policy as proposed.
We received five requests to reassign the assigned CBSA to a different eligible CBSA (as described in Table X). One request was related to the comment summarized above regarding the termination of reclassification for hospitals reclassified to areas where all counties in the CBSA would become rural. This hospital (CCN 140113) requested to have its MGCRB reclassification reassigned to its geographic “home” CBSA 16580. We did not propose this CBSA as eligible for reassignment and are denying this request for the reasons discussed earlier. We note that this hospital, by cancelling its current § 412.103 rural reclassification, will receive the wage index for its geographic CBSA in FY 2025.The remaining requests are as follows:

We note that MGCRB Case No. 25C0250 (CCN 490113) was a “home area” reclassification and was assigned its new geographic “home area” in the proposed rule. We did not explicitly address in the proposed rule whether hospitals with “home area” reclassifications to a CBSA that had one or more counties move to a new or different CBSA would be eligible to request reassignment to that new or different CBSA. However, we find that the case meets the proposed requirements for reassignment applicable generally to hospitals whose reclassifications are reassigned on the basis of changes to the CBSA under the revised OMB delineations, as the hospital is requesting to be reclassified to an area that is a) not its geographic CBSA and b) contains at least one county from its approved CBSA.
After reviewing the submitted materials, we have determined these four requests meet the appropriate distance requirements for reassignment and have approved the requests as described.
Table Y lists all hospitals subject to our reclassification assignment and reassignment policy and the CBSA assigned or reassigned for FY 2025 under this policy. Cases marked with an asterisk were withdrawn or terminated for FY 2025 but may be reinstated in FY 2026.

We note that the Office of Hearings Case and Document Management System (OH CDMS) may not be updated to reflect different CBSA numbers for reclassification assignments and reassignments finalized in this rule. When making withdrawal, termination, or reinstatement requests for these cases, the original CBSA number may be displayed in the OH CDMS. If hospitals require further assistance in this matter, please contact [email protected] .
As discussed in section III.B., CMS is adopting the revised OMB Bulletin No. 23-01 delineations, which use planning regions instead of counties as the basis for CBSA construction in the State of Connecticut. There are five current urban CBSAs that include at least one county in Connecticut. These are 14860 (Bridgeport-Stamford-Norwalk, CT), 25540 (Hartford-East Hartford-Middletown, CT), 35300 (New Have-Milford, CT), 35980 (Norwich-New London, CT), and 49340 (Worcester, MA-CT). In the FY 2025 CBSAs, based on the OMB Bulletin No. 23-01 delineations, there are five CBSAs that will contain at least one county-equivalent “planning region.” The five CBSAs are 14860 (Bridgeport-Stamford-Danbury, CT), 25540 (Hartford-West Hartford-East Hartford, CT), 35300 (New Haven, CT), 35980 (Norwich-New London-Willimantic, CT), and 47930 (Waterbury-Shelton, CT).
As there was significant reconfiguration of the CBSAs due to the transition from counties to planning regions, we proposed to adopt a similar assignment policy for hospitals reclassified to CBSAs that currently include Connecticut counties as we did for hospitals reclassified to CBSAs where one or more counties move to a new or different urban CBSA (described in the previous subsection).
The following table lists all current “home area” reclassifications to one of the CBSAs that currently contain at least one county in Connecticut.

The following table provides a list of current FY 2024 CBSAs (column 1) that contain at least one county in Connecticut. Under the proposal, hospitals with active MGCRB reclassifications into the CBSAs in column 1 would be subject to the reclassification assignment policy. The third column of “eligible” CBSAs lists all revised CBSAs that contain at least one planning region that is part of the current FY 2025 CBSA (in column 1). Consistent with the policy discussed in the previous section, we proposed a policy that affected reclassified hospitals be assigned to a CBSA that would contain the most proximate planning region that (1) is located outside of the hospital's proposed FY 2025 geographic labor market area, and (2) contains a portion of a county included in the original FY 2024 CBSA to which the hospital is reclassified.

We believe that assigning reclassifications to the CBSA that contains the nearest county-equivalent planning region that meets the aforementioned criteria satisfies the statutory requirement at section 1886(d)(10)(v) of the Act by maintaining reclassification status for a period of 3 fiscal years, while generally respecting the longstanding principle of geographic proximity in the labor market reclassification process. For county group reclassifications, we would follow our proposed policy, as previously discussed, except that we proposed to reassign hospitals in a county group reclassification to the CBSA under the revised OMB delineations that contains the county-equivalent to which the majority of hospitals in the group reclassification are geographically closest. We also proposed to allow such hospitals, or county groups of hospitals, to submit a request to the [email protected] mailbox for reassignment to another proposed CBSA that would contain a county that is part of the current CBSA to which it was approved to be reclassified (based on FY 2024 delineations) if the hospital or county group of hospitals can demonstrate compliance with applicable reclassification proximity rules.
We did not receive any comments regarding the MGCRB reclassification assignment and reassignment policy due to the adoption of the revised Connecticut county-equivalents. We are finalizing the policy as proposed.
We received two requests from hospitals affected by this policy to reassign the assigned CBSA to a different eligible CBSA (as described in Table X).

After reviewing the submitted materials, we have determined both requests meet the appropriate distance requirements for reassignment and have approved these requests.
Table Y lists all hospitals subject to our reclassification assignment and reassignment policy for CBSAs reconfigured due to the migration to Connecticut planning regions and the CBSA assigned or reassigned for FY 2025 under this policy. Cases marked with an asterisk were withdrawn for FY 2025 but may be reinstated in FY 2026.

We note that the OH CDMS may not be updated to reflect different CBSA numbers for reclassification assignments and reassignments finalized in this rule. When making withdrawal, termination, or reinstatement requests for these cases, the original CBSA number may be displayed in the OH CDMS. If hospitals require further assistance in this matter, please contact [email protected] .
As mentioned in section III.F.3.a of this final rule, under the regulations at § 412.273, hospitals that have been reclassified by the MGCRB are permitted to withdraw or terminate an approved reclassification. The current regulations at § 412.273(c)(1)(ii) and (c)(2) for withdrawals and terminations require the request to be received by the MGCRB within 45 days of the date that CMS' annual notice of proposed rulemaking is issued in the Federal Register concerning changes to the IPPS and proposed payment rates.
In the 2018 IPPS/LTCH PPS Final Rule ( 82 FR 38148 through 38150 ), we finalized changes to the 45-day notification rules so that hospitals have 45 days from the public display of the annual proposed rule for the IPPS instead of 45 days from publication to inform CMS of certain requested changes relating to the development of the hospital wage index. We stated that we believe that the public has access to the necessary information from the date of public display of the proposed rule at the Office of the Federal Register and on its website to make the decisions at issue. While we finalized changes to the 45-day notification rules for decisions about the outmigration adjustment and waiving Lugar status, we did not finalize a change to the timing for withdrawing or terminating MGCRB decisions.
Instead, in response to comments expressing concern that some hospitals may be disadvantaged if the Administrator's decision on a hospital's request for review of an MGCRB decision has not been issued prior to the proposed deadline for submitting withdrawal or termination requests to the MGCRB, we maintained our existing policy of requiring hospitals to request from the MGCRB withdrawal or termination of an MGCRB reclassification within 45 days of issuance in the Federal Register . We stated in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38149 ) that considering the usual dates of the MGCRB's decisions (generally early February) and of the public display of the IPPS proposed rule, the maximum amount of time for an Administrator's decision to be issued may potentially extend beyond the proposed deadline of 45 days from the date of public display.
However, the MGCRB currently issues decisions earlier, in January, which mitigates this concern. For example, the MGCRB has sent decision letters to hospitals via email on January 23, 2024, for the FY 2025 cycle and on January 31, 2023, for the FY 2024 cycle. We believe that the MGCRB will continue to issue its decisions in January, due to their upgrade to an electronic system that expedites processing applications and issuing decision letters efficiently. The regulations at §§ 412.278(a) and (b)(1) provide that a hospital may request the Administrator to review the MGCRB decision within 15 days after the date the MGCRB issues its decision. Under § 412.278(f)(2)(i), the Administrator issues a decision not later than 90 days following receipt of the party's request for review. Consequently, MGCRB decisions could be issued as late as the end of January, and the 15 days the hospital has to request the Administrator's review, plus the 90 days the Administrator has to issue a decision, would result in hospitals receiving the results of the review prior to 45 days after display (which would be May 16th if the proposed rule is displayed on the target date of April 1, but later if there is a delay).
While the current timing of MGCRB decisions in January allows for hospitals to receive the results of any review prior to 45 days after display of the proposed rule for the relevant FY, and we expect this timing to continue, we acknowledge that section 1886(d)(10)(C)(iii)(I) of the Act grants the MGCRB 180 days after the application deadline to render a decision. If the MGCRB were to delay issuing decisions until the last day possible according to the Statute, which is February 28th, a hospital requesting the Administrator's review may not receive the results of the review prior to 45 days after display.
Therefore, we proposed to change the deadline for hospitals to withdraw or terminate MGCRB classifications from within 45 days of the date that the annual notice of proposed rulemaking is issued in the Federal Register to within 45 days of the public display of the annual notice of proposed rulemaking on the website of the Office of the Federal Register, or within 7 calendar days of receiving a decision of the Administrator in accordance with § 412.278 of this part, whichever is later. This change will synchronize this deadline with other wage index deadlines, such as the deadlines for accepting the outmigration adjustment ( print page 69292) and waiving or reinstating Lugar status. As hospitals typically know the results of the Administrator's decisions on reviews within 45 days of the public display of the proposed rule for the upcoming fiscal year, we believe hospitals have access to the information they need to make reclassification decisions. In the rare circumstance that a hospital would not receive the results of the review prior to 45 days of the public display date, or receives the results of the review less than 7 days before the deadline, the hospital would have 7 calendar days after receiving the Administrator's decision to request to withdraw or terminate MGCRB classification. While we do not anticipate frequent use of this extension, we believe this fully addresses the concern that some hospitals may be disadvantaged if the Administrator's decision on a hospital's request for review of an MGCRB decision has not been issued prior to the deadline for submitting withdrawal or termination requests to the MGCRB. We believe that 7 days after receiving the Administrator's decision affords hospitals adequate time to make calculated reclassification decisions.
Specifically, we proposed to change the words “within 45 days of the date that CMS' annual notice of proposed rulemaking is issued in the Federal Register ” in the regulation text at 412.273(c)(1)(ii) and 412.273(c)(2) for withdrawals and terminations to “within 45 days of the date of filing for public inspection of the proposed rule at the website of the Office of the Federal Register, or within 7 calendar days of receiving a decision of the Administrator in accordance with § 412.278 of this part, whichever is later”.
Comment: We received several comments opposing our proposal. Commenters expressed that the proposed change would give providers less time to analyze their reclassification options and to make appropriate elections. Some of the commenters pointed out that under CMS' proposal, hospitals would have less time to make decisions based on the final wage data PUF, which was issued this year on April 29. A commenter asked that if CMS finalizes this proposal, it should make available all relevant information for a hospital to make an informed decision by the same public display date, including: the final wage data PUF, an updated list of Administrator appeal decisions, and the MGCRB's listing of its FY 2025 group & individual decisions. Another commenter noted that the timeframe could be even tighter in future years if the target date of April 1st for issuing the IPPS proposed rule is met, which would give a hospital only 14 business days from the April 29th PUF until 45 days from display (May 16th) to make reclassification decisions.
Response: We understand the commenters' concern that the proposal shortens the timeframe for hospitals to make reclassification decisions. However, we note that none of the commenters maintained that hospitals would not have access to the information necessary to make an informed decision, just that the timeframe would be shortened, which our proposal discusses is necessary to synchronize this deadline with other wage index deadlines. We also note that none of the commenters requested that we modify the proposed extended deadline of within 7 calendar days of receiving a decision of the Administrator. Therefore, we continue to believe that the revised timeframe provides hospitals adequate time to access the information they need to make informed reclassification decisions. Furthermore, the other information that commenters requested be made available by the start of the 45-day timeframe, such as the final wage data PUF and administrative appeal decisions, is not necessarily available at the current start of the 45 day timeframe. Hospitals currently expect to begin evaluating their reclassification options based on the best available information and may choose to finalize their decisions as more updated information becomes available during the timeframe for withdrawals. Other than adjusting to a shortened timeframe, we believe that this proposal does not create a new disadvantage for hospitals, nor does it prevent hospitals from making informed reclassification decisions since more updated information does become available during the timeframe for withdrawals. For the reasons enumerated in our proposal and in this response, we continue to believe that the revised timeline provides hospitals adequate time to make informed decisions about their reclassification options.
Therefore, we are finalizing our proposed changes without modification to the regulations for withdrawals and terminations at § 412.273(c)(1)(ii) & (c)(2).
In the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51599 through 51600 ), we adopted the policy that, beginning with FY 2012, an eligible hospital that waives its Lugar status to receive the out-migration adjustment has effectively waived its deemed urban status and, thus, is rural for all purposes under the IPPS effective for the fiscal year in which the hospital receives the outmigration adjustment. In addition, in that rule, we adopted a minor procedural change that would allow a Lugar hospital that qualifies for and accepts the out-migration adjustment (through written notification to CMS within 45 days from the issuance of the proposed rule in the Federal Register ) to waive its urban status for the full 3-year period for which its out-migration adjustment is effective. By doing so, such a Lugar hospital would no longer be required during the second and third years of eligibility for the out-migration adjustment to advise us annually that it prefers to continue being treated as rural and receive the out-migration adjustment. In the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56930 ), we further clarified that if a hospital wishes to reinstate its urban status for any fiscal year within this 3-year period, it must send a request to CMS within 45 days of the issuance of the proposed rule in the Federal Register for that particular fiscal year. We indicated that such reinstatement requests may be sent electronically to [email protected] . In the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38147 through 38148 ), we finalized a policy revision to require a Lugar hospital that qualifies for and accepts the out-migration adjustment, or that no longer wishes to accept the out-migration adjustment and instead elects to return to its deemed urban status, to notify CMS within 45 days from the date of public display of the proposed rule at the Office of the Federal Register. These revised notification timeframes were effective beginning October 1, 2017. In addition, in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38148 ), we clarified that both requests to waive and to reinstate “Lugar” status may be sent to [email protected] . To ensure proper accounting, we request hospitals to include their CCN, and either “waive Lugar” or “reinstate Lugar”, in the subject line of these requests. We received five timely requests for hospitals to accept the county out-migration adjustment in lieu of its “Lugar” reclassification. The requests were from CCNs 150030, 320033, 340126, 390183, and 390330. When applicable, the hospitals were informed that this election would result in a cancelation of their rural reclassification status under § 412.103, effective Oct 1, 2024. We also informed hospital that for ( print page 69293) the request to be approved, the hospital must withdraw or terminate any active MGCRB reclassification. All requests have been approved and will remain in effect for the remainder of the 3-year out-migration adjustment period.
In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42314 and 42315 ), we clarified that in circumstances where an eligible hospital elects to receive the outmigration adjustment within 45 days of the public display date of the proposed rule at the Office of the Federal Register in lieu of its Lugar wage index reclassification, and the county in which the hospital is located would no longer qualify for an outmigration adjustment when the final rule (or a subsequent correction notice) wage index calculations are completed, the hospital's request to accept the outmigration adjustment would be denied, and the hospital would be automatically assigned to its deemed urban status under section 1886(d)(8)(B) of the Act. We stated that final rule wage index values would be recalculated to reflect this reclassification, and in some instances, after taking into account this reclassification, the out-migration adjustment for the county in question could be restored in the final rule. However, as the hospital is assigned a Lugar reclassification under section 1886(d)(8)(B) of the Act, it would be ineligible to receive the county outmigration adjustment under section 1886(d)(13)(G) of the Act.
As discussed in section III.A.2. of the preamble of this final rule, CMS is updating the CBSA labor market delineations to reflect the changes made in the July 15, 2023, OMB Bulletin 23-01. In that section, we noted that 54 currently rural counties will be added to new or existing urban CBSAs. Of those 54 counties, 22 are currently deemed urban under section 1886(d)(8)(B) of the Act. We proposed that hospitals located in such a “Lugar” county, barring another form of wage index reclassification, are assigned the reclassified wage index of a designated urban CBSA. Section 1886(d)(8)(B) of the Act defines a deemed urban county as a “rural county adjacent to one or more urban areas” that meets certain commuting thresholds. Since we proposed to modify the status of these 22 counties from rural to urban, they would no longer qualify as “Lugar” counties. Hospitals located within these counties would be considered geographically urban under the revised OMB delineations. The table in this section of this rule lists the counties that are no longer deemed urban under section 1886(d)(8)(B) of the Act under the revised OMB delineations. We note that in almost all instances, the “Lugar” county is joining the same (or a substantially similar) urban CBSA as it was deemed to in FY 2024.

We note that in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 49973 through 49977 ), when we adopted large scale changes to the CBSA labor market delineations based on the new 2010 decennial census, we also re-evaluated the commuting data thresholds for all eligible rural counties in accordance with the requirement set forth in section 1886(d)(8)(B)(ii)(II) of the Act to base the list of qualifying hospitals on the most recently available decennial population data. Therefore, we proposed to reevaluate the “Lugar” status for all counties in FY 2025 using the same commuting data table used to develop the OMB Bulletin No. 23-01 revised delineations. The data table is the “2016-2020 5-Year American Community Survey Commuting Flows” (available on OMB's website: https://www.census.gov/data/tables/2020/demo/metro-micro/commuting-flows-2020.html ). We also proposed to use the same methodology discussed in the FY 2020 IPPS/LTCH final rule ( 84 FR 42315 through 42318 ) to assign the appropriate reclassified CBSA for hospitals in “Lugar” counties. That is, when assessing which CBSA to assign, we will sum the total number of workers that commute from the “Lugar” county to both “central” and “outlying” urban counties (rather than just “central” county commuters). ( print page 69294)
By applying the 2020 American Community Survey (ACS) commuting data to the updated OMB labor market delineations, we proposed the following changes to the current “Lugar” county list: 17 of the 53 urban counties that were proposed to become rural under the revised OMB delineations, and both newly created rural Connecticut planning region county-equivalents would qualify as “Lugar” counties. We also determined that, as proposed, 33 rural counties (an approximately 11 hospitals) would lose “Lugar” status, as the county no longer meets the commuting thresholds or adjacency criteria specified in section 1886(d)(8)(B) of the Act.

The following table lists all proposed “Lugar” counties for FY 2025. We indicated additions to the list for FY 2025 with “New” in column 5.

We noted that Litchfield County, CT is no longer listed as a “Lugar” county as it is not included in the revised CBSA delineations. The majority of Litchfield County is now within the Northwest Hills Planning Region county-equivalent, with some of the county's current constituent townships assigned to other urban county-equivalents. We ( print page 69298) also noted that in prior fiscal years, Merrimack County, NH was included as a “Lugar” redesignated county pursuant to the provision at § 412.62(f)(1)(ii)(B), which deems certain rural counties in the New England region to be part of urban areas. Merrimack County now meets the commuting standards to be considered deemed urban under the “Lugar” statute at section 1886(d)(8)(B) of the Act.
We recognize that the changes to the “Lugar” list may have negative financial impacts for hospitals that lose deemed urban status. We believe that the 5 percent cap on negative wage index changes discussed in section III.G.6, would mitigate significant negative payment impacts for FY 2025, and would afford hospitals adequate time to fully assess any additional reclassification options available to them. We also note that special statuses limited to hospitals located in rural areas (such as MDH or SCH status) may be terminated if hospitals are deemed urban under section 1886(d)(8)(B) of the Act. In these cases, hospitals should apply for rural reclassification status under § 413.103 prior to October 1, 2024, if they wish to ensure no disruption in status.
We did not receive any comments regarding the implementation of revised OMB labor market area delineations for redesignations under section 1886(d)(8)(B) of the Act. We are finalizing without modification the list of proposed qualifying counties listed in the prior table.
The following adjustments to the wage index are listed in the order that they are generally applied. First, the rural floor, imputed floor, and state frontier floor provide a minimum wage index. The rural floor at section 4410(a) of the Balanced Budget Act of 1997 ( Pub. L. 105-33 ) provides that the wage index for hospitals in urban areas of a State may not be less than the wage index applicable to hospitals located in rural areas in that State. The imputed floor at section 1886(d)(3)(E)(iv) of the Act provides a wage index minimum for all-urban states. The state frontier floor at section 1886(d)(3)(E)(iii) of the Act requires that hospitals in frontier states cannot be assigned a wage index of less than 1.0000. Next, the out-migration adjustment at section 1886(d)(13)(A) of the Act is applied, potentially increasing the wage index for hospitals located in certain counties that have a relatively high percentage of hospital employees who reside in the county but work in a different county or counties with a higher wage index. The low-wage index hospital adjustment finalized in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42325 through 42339 ) is then applied, which increases the wage index values for hospitals with wage indexes at or below the 25th percentile. Finally, all hospital wage index decreases are capped at 95 percent of the hospital's final wage index in the prior fiscal year, according to the policy finalized in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49018 through 49021 ).
Section 4410(a) of the Balanced Budget Act of 1997 ( Pub. L. 105-33 ) provides that, for discharges on or after October 1, 1997, the area wage index applicable to any hospital that is located in an urban area of a State may not be less than the area wage index applicable to hospitals located in rural areas in that State. This provision is referred to as the rural floor. Section 3141 of the Patient Protection and Affordable Care Act ( Pub. L. 111-148 ) also requires that a national budget neutrality adjustment be applied in implementing the rural floor. Based on the FY 2025 wage index associated with this final rule (which is available via the internet on the CMS website), and based on the calculation of the rural floor including the wage data of hospitals that have reclassified as rural under § 412.103, we estimate that 771 hospitals would receive the rural floor in FY 2025. The budget neutrality impact of the proposed application of the rural floor is discussed in section II.A.4.e. of Addendum A of this final rule.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48784 ), CMS finalized a policy change to calculate the rural floor in the same manner as we did prior to the FY 2020 IPPS/LTCH PPS final rule, in which the rural wage index sets the rural floor. We stated that for FY 2023 and subsequent years, we would include the wage data of § 412.103 hospitals that have no MGCRB reclassification in the calculation of the rural floor, and include the wage data of such hospitals in the calculation of “the wage index for rural areas in the State in which the county is located” as referred to in section 1886(d)(8)(C)(iii) of the Act.
In the FY 2024 IPPS/LTCH final rule ( 88 FR 58971-77 ), we finalized a policy change beginning that year to include the data of all § 412.103 hospitals, even those that have an MGCRB reclassification, in the calculation of the rural floor and the calculation of “the wage index for rural areas in the State in which the county is located” as referred to in section 1886(d)(8)(C)(iii) of the Act. We explained that after revisiting the case law, prior public comments, and the relevant statutory language, we agreed that the best reading of section 1886(d)(8)(E)'s text that CMS “shall treat the [§ 412.103] hospital as being located in the rural area” is that it instructs CMS to treat § 412.103 hospitals the same as geographically rural hospitals for the wage index calculation.
Accordingly, in the FY 2024 IPPS/LTCH PPS final rule, we finalized a policy to include hospitals with § 412.103 reclassification along with geographically rural hospitals in all rural wage index calculations, and to exclude “dual reclass” hospitals (hospitals with simultaneous § 412.103 and MGCRB reclassifications) that are implicated by the hold harmless provision at section 1886(d)(8)(C)(ii) of the Act. (For additional information on these changes, we refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58971 through 58977 ).)
Comment: Commenters supported CMS' continued treatment of hospitals reclassified as rural under § 412.103 in the same manner as geographically rural hospitals for the rural wage index and rural floor calculations. A commenter specifically agreed with CMS' interpretation of case law as discussed in the proposed rule and stated that restoring equality between a state's rural floor and its rural wage index is an appropriate and fair implementation of the statute. One commenter requested that CMS confirm whether the pre-reclassified wage index for each hospital reflects if the hospital has reclassified under § 412.103.
Multiple commenters expressed concern over the rural floor budget neutrality factor. A commenter disagreed with CMS' decision to budget neutralize the policy to include hospitals with simultaneous § 412.103 and MGCRB reclassifications in the rural wage index calculation. The commenter stated that some hospitals are paying a substantial cost for an artificial increase in the wage index of other hospitals, and that this cost escalates as hospitals around the country make reclassification decisions to take advantage of this policy change. Another commenter pointed out that the rural floor budget neutrality factor has more than doubled over the past decade, with the most notable increase occurring in FY 2024 due to CMS' decision to include § 412.103 reclassifications along with geographically rural hospitals in the ( print page 69299) rural wage index calculations. The commenter stated that the rural floor budget neutrality factor decreased IPPS payments by 2.87% that year, compared to 1.56% the year before. Similarly, a commenter requested that CMS provide an impact table with the FY 2025 final rule and with subsequent rulemakings showing the number of hospitals and total payments impacted by the policy, with results aggregated at the state level.
Other commenters acknowledged CMS' statutory budget neutrality requirement but challenged CMS' application of the rural floor. These commenters argued that section 4410(b) of the Balanced Budget Act of 1997 (BBA) exempts urban and reclassified rural hospitals that receive the rural floor from having their wage indexes reduced. According to these commenters, the rural floor budget neutrality adjustment should be applied only to the wage indexes of hospitals not receiving the rural floor ( i.e.: non-reclassified rural hospitals, and urban hospitals with wage indexes above the rural floor).
Response: While we did not propose any changes to the rural floor policy in the FY 2025 IPPS/LTCH PPS proposed rule, we appreciate the commenters' continued support.
In response to the commenter asking for clarification about how § 412.103 hospitals are reflected in the pre-reclassified wage index, we are clarifying that the pre-reclassified wage index reflects hospitals' locations prior to any form of reclassification for budget neutrality purposes.
We understand the commenter's concerns regarding the effect that the rural floor budget neutrality factor has on some hospitals as other hospitals make reclassification decisions to take advantage of the rural floor policy. The commenter noting the increase in the rural floor budget neutrality factor in FY 2024 is correct that the budget neutrality factor increased by 2.87% that year, compared to 1.56% the year before. As we noted in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58975 ), we expect that the number of IPPS hospitals assigned their State's rural wage index will increase in future years as hospitals adjust to the policy and as the relative value of States' rural wage index values increase due to the inclusion of hospitals that strategically obtain § 412.103 reclassification. As a result, the majority of hospitals (if not all) will be assigned identical wage index values within their states. For example, for FY 2025, 58% of geographically urban hospitals are receiving a wage index equal to their State's rural floor, imputed floor, or frontier floor prior to any outmigration, low wage index hospital, or 5 percent decrease cap adjustments. As we stated in last year's IPPS/LTCH PPS final rule ( 88 FR 58975 ), as substantially more hospitals receive the rural floor, there will be a consequently greater budget neutrality impact. However, we believe this result would be unavoidable given the requirement of section 1886(d)(8)(E) of the Act to treat § 412.103 hospitals `as being located in the rural area' of the state, as well as the requirement at sections 4410(b) of the BBA 1997 and 3141 of the Patient Protection and Affordable Care Act ( Pub. L. 111-148 ) that a uniform, national budget neutrality adjustment be applied in implementing the rural floor. With regard to the commenter requesting evaluation of the impacts of the policy at the hospital and state-specific levels, we refer the commenter to the IPPS Payment Impact File associated with this final rule (available on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/fy-2025-ipps-final-rule-home-page ) and to section II.A.4.d. of the Addendum to this final rule for a discussion of the rural floor budget neutrality factor. The area wage index prior to the application of the rural floor is available in Table 3.
With regard to the commenters' assertion that urban and reclassified rural hospitals that receive the rural floor should be excluded from the application of the rural floor budget neutrality factor, we considered this approach in the FY 2008 IPPS proposed and final rules ( 72 FR 24787 and 72 FR 47325 ) and believe we have applied the rural floor budget neutrality adjustment in a manner consistent with the statute. Specifically, in the FY 2008 IPPS proposed rule, we rejected a reading of section 4410(b) of the BBA requiring that the budget neutrality adjustment would be applied only to those hospitals that do not receive the rural floor, because urban hospitals receiving the rural floor would receive a higher wage index than the rural hospitals within the same State (because hospitals receiving the rural floor would not be subject to budget neutrality, whereas rural hospitals would be) ( 72 FR 24787 ). We continue to believe that such a reading would not be consistent with the best reading of the statute. The statute sets a floor for urban hospitals. The statute does not instruct CMS to pay urban hospitals a wage index higher than the wage index applicable to rural hospitals, and contains no suggestion that the general budget neutrality provisions of section 1886(d)(8)(D)—which expressly apply to the adjustments made in section 1886(d)(C)—should not apply . In the FY 2008 IPPS final rule, we adopted the current approach to implement rural floor budget neutrality by applying a uniform, national adjustment to the wage index ( 72 FR 47325 ). Since then, Congress specifically endorsed our approach in section 3141 of the Patient Protection and Affordable Care Act ( Pub. L. 111-148 ), which requires that the rural floor budget neutrality adjustment be applied “in the same manner as the Secretary administered such [adjustment] for discharges occurring during fiscal year 2008 (through a uniform, national adjustment to the area wage index).”
In addition, we note that section 4410 of the BBA to which the commenter refers provides that the rural floor is equal to “the area wage index applicable under [section 1886(d)(3)(E) of the Social Security Act] to hospitals located in rural areas in the State.” Under our existing policy, the rural floor and the rural wage index for the state are the same after application of the rural floor budget neutrality adjustment factor, and nothing in section 4410 of the BBA requires otherwise. Put differently, CMS' methodology amounts to merely calculating the amount of the rural floor such that it is the same as the final rural wage index for the state, rather than reducing the wage indices of low wage urban hospitals or reclassified rural hospitals that receive the rural floor relative to what they would be otherwise—in that way it appropriately implements both section 4410 of the BBA and section 3141 of the ACA. Thus, consistent with our longstanding methodology for implementing the rural floor, we believe it is appropriate to continue to apply a budget neutrality adjustment to all hospitals' wage indexes.
In the FY 2005 IPPS final rule ( 69 FR 49109 through 49111 ), we adopted the imputed floor policy as a temporary 3-year regulatory measure to address concerns from hospitals in all-urban States that have stated that they are disadvantaged by the absence of rural hospitals to set a wage index floor for those States. We extended the imputed floor policy eight times since its initial implementation, the last of which was adopted in the FY 2018 IPPS/LTCH PPS final rule and expired on September 30, 2018. We refer readers to further discussions of the imputed floor in the IPPS/LTCH PPS final rules from FYs 2014 through 2019 ( 78 FR 50589 through 50590 , 79 FR 49969 through ( print page 69300) 49971, 80 FR 49497 through 49498 , 81 FR 56921 through 56922 , 82 FR 38138 through 38142 , and 83 FR 41376 through 41380 , respectively) and to the regulations at § 412.64(h)(4). For FYs 2019, 2020, and 2021, hospitals in all-urban states received a wage index that was calculated without applying an imputed floor, and we no longer included the imputed floor as a factor in the national budget neutrality adjustment.
Section 9831 of the American Rescue Plan Act of 2021 ( Pub. L. 117-2 ), enacted on March 11, 2021, amended section 1886(d)(3)(E)(i) of the Act and added section 1886(d)(3)(E)(iv) of the Act to establish a minimum area wage index for hospitals in all-urban States for discharges occurring on or after October 1, 2021. Specifically, section 1886(d)(3)(E)(iv)(I) and (II) of the Act provides that for discharges occurring on or after October 1, 2021, the area wage index applicable to any hospital in an all-urban State may not be less than the minimum area wage index for the fiscal year for hospitals in that State established using the methodology described in § 412.64(h)(4)(vi) as in effect for FY 2018. Unlike the imputed floor that was in effect from FYs 2005 through 2018, section 1886(d)(3)(E)(iv)(III) of the Act provides that the imputed floor wage index shall not be applied in a budget neutral manner. Section 1886(d)(3)(E)(iv)(IV) of the Act provides that, for purposes of the imputed floor wage index under clause (iv), the term all-urban State means a State in which there are no rural areas (as defined in section 1886(d)(2)(D) of the Act) or a State in which there are no hospitals classified as rural under section 1886 of the Act. Under this definition, given that it applies for purposes of the imputed floor wage index, we consider a hospital to be classified as rural under section 1886 of the Act if it is assigned the State's rural area wage index value.
Effective beginning October 1, 2021 (FY 2022), section 1886(d)(3)(E)(iv) of the Act reinstates the imputed floor wage index policy for all-urban States, with no expiration date, using the methodology described in § 412.64(h)(4)(vi) as in effect for FY 2018. We refer readers to the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45176 through 45178 ) for further discussion of the original imputed floor calculation methodology implemented in FY 2005 and the alternative methodology implemented in FY 2013.
Based on data available for this final rule, States that will be all-urban States as defined in section 1886(d)(3)(E)(iv)(IV) of the Act, and thus hospitals in such States that will be eligible to receive an increase in their wage index due to application of the imputed floor for FY 2025, are identified in Table 3 associated with this final rule. States with a value in the column titled “State Imputed Floor” are eligible for the imputed floor.
The regulations at § 412.64(e)(1) and (4) and (h)(4) and (5) implement the imputed floor required by section 1886(d)(3)(E)(iv) of the Act for discharges occurring on or after October 1, 2021. The imputed floor will continue to be applied for FY 2025 in accordance with the policies adopted in the FY 2022 IPPS/LTCH PPS final rule. For more information regarding our implementation of the imputed floor required by section 1886(d)(3)(E)(iv) of the Act, we refer readers to the discussion in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45176 through 45178 ).
Comment: We received comments supporting the application of the imputed floor. A commenter opposed the imputed floor stating that the imputed floor continues to unfairly manipulate the wage index to benefit a handful of only-urban states and territories.
Response: We thank the commenters for their input. As discussed earlier, the imputed floor is a statutory requirement under section 9831 of the American Rescue Plan Act of 2021 ( Pub. L. 117-2 ) which requires the Secretary to establish a minimum area wage index for hospitals in all-urban States for discharges occurring on or after October 1, 2021. We did not propose any changes to the methodology for calculating the imputed floor as set forth in § 412.64(e)(1) and (4) and (h)(4) and (5). Therefore, in accordance with the statute and existing regulations, we are applying the imputed floor for hospitals in all-urban States for FY 2025.
Section 10324 of Public Law 111-148 requires that hospitals in frontier States cannot be assigned a wage index of less than 1.0000. (We refer readers to the regulations at § 412.64(m) and to a discussion of the implementation of this provision in the FY 2011 IPPS/LTCH PPS final rule ( 75 FR 50160 through 50161 ).) In the FY 2025 IPPS/LTCH PPS proposed rule, we did not propose any changes to the frontier floor policy for FY 2025. In the proposed rule we stated 41 hospitals would receive the frontier floor value of 1.0000 for their FY 2025 proposed wage index. These hospitals are located in Montana, North Dakota, South Dakota, and Wyoming.
We did not receive any public comments on the application of the State frontier floor for FY 2025. In this final rule, 41 hospitals will receive the frontier floor value of 1.0000 for their FY 2025 wage index. These hospitals are located in Montana, North Dakota, South Dakota, and Wyoming. We note that while Nevada meets the criteria of a frontier State, all hospitals within the State currently receive a wage index value greater than 1.0000.
The areas affected by the rural and frontier floor policies for the final FY 2025 wage index are identified in Table 3 associated with this final rule, which is available via the internet on the CMS website.
In accordance with section 1886(d)(13) of the Act, as added by section 505 of Public Law 108-173 , beginning with FY 2005, we established a process to make adjustments to the hospital wage index based on commuting patterns of hospital employees (the “out-migration” adjustment). The process, outlined in the FY 2005 IPPS final rule ( 69 FR 49061 ), provides for an increase in the wage index for hospitals located in certain counties that have a relatively high percentage of hospital employees who reside in the county but work in a different county (or counties) with a higher wage index.
Section 1886(d)(13)(B) of the Act requires the Secretary to use data the Secretary determines to be appropriate to establish the qualifying counties. When the provision of section 1886(d)(13) of the Act was implemented for the FY 2005 wage index, we analyzed commuting data compiled by the U.S. Census Bureau that were derived from a special tabulation of the 2000 Census journey-to-work data for all industries (CMS extracted data applicable to hospitals). These data were compiled from responses to the “long-form” survey, which the Census Bureau used at that time, and which contained questions on where residents in each county worked ( 69 FR 49062 ). However, the 2010 Census was “short form” only; information on where residents in each county worked was not collected as part of the 2010 Census. The Census Bureau worked with CMS to provide an alternative dataset based on the latest available data on where residents in each county worked in 2010, for use in developing a new out-migration adjustment based on new commuting patterns developed from the ( print page 69301) 2010 Census data beginning with FY 2016.
To determine the out-migration adjustments and applicable counties for FY 2016, we analyzed commuting data compiled by the Census Bureau that were derived from a custom tabulation of the American Community Survey (ACS), an official Census Bureau survey, utilizing 2008 through 2012 (5-year) Microdata. The data were compiled from responses to the ACS questions regarding the county where workers reside and the county to which workers commute. As we discussed in prior IPPS/LTCH PPS final rules, most recently in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58983 ), we have applied the same policies, procedures, and computations since FY 2012. We refer readers to the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49500 through 49502 ) for a full explanation of the revised data source. We also stated that we would consider determining out-migration adjustments based on data from the next Census or other available data, as appropriate.
As discussed earlier in section III.B., CMS proposed to adopt revised delineations from the OMB Bulletin 23-01, published July 21, 2023. The revised delineations incorporate population estimates based on the 2020 decennial census, as well as updated journey-to-work commuting data. The Census Bureau once again worked with CMS to provide an alternative dataset based on the latest available data on where residents in each county worked, for use in developing a new out-migration adjustment based on new commuting patterns. We analyzed commuting data compiled by the Census Bureau that were derived from a custom tabulation of the ACS, utilizing 2016 through 2020 data. The Census Bureau produces county level commuting flow tables every 5 years using non-overlapping 5-year ACS estimates. The data include demographic characteristics, home and work locations, and journey-to-work travel flows. The custom tabulation requested by CMS was specific to general medical and surgical hospital and specialty (except psychiatric and substance use disorder treatment) hospital employees (hospital sector Census code 8191/NAICS code 6221 and 6223) who worked in the 50 States, Washington, DC, and Puerto Rico and, therefore, provided information about commuting patterns of workers at the county level for residents of the 50 States, Washington, DC, and Puerto Rico.
For the ACS, the Census Bureau selects a random sample of addresses where workers reside to be included in the survey, and the sample is designed to ensure good geographic coverage. The ACS samples approximately 3.5 million resident addresses per year. [ 193 ] The results of the ACS are used to formulate descriptive population estimates, and, as such, the sample on which the dataset is based represents the actual figures that would be obtained from a complete count.
For FY 2025, and subsequent years, we proposed that the out-migration adjustment will be based on the data derived from the previously discussed custom tabulation of the ACS utilizing 2016 through 2020 (5-year) Microdata. As discussed earlier, we believe that these data are the most appropriate to establish qualifying counties, because they are the most accurate and up-to-date data that are available to us. Furthermore, we stated in the proposed rule ( 89 FR 36183 ) that with the proposed transition of several counties in Connecticut to “planning region” county equivalents (discussed in section III.B.3. of the preamble the proposed rule), the continued use of a commuting dataset developed with expiring county definitions would be less accurate in approximating commuting flows. We proposed that the FY 2025 out-migration adjustments continue to be based on the same policies, procedures, and computation that were used for the FY 2012 out-migration adjustment. We have applied these same policies, procedures, and computations since FY 2012, and we believe they continue to be appropriate for FY 2025. (We refer readers to a full discussion of the out-migration adjustment, including rules on deeming hospitals reclassified under section 1886(d)(8) or section 1886(d)(10) of the Act to have waived the out-migration adjustment, in the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51601 through 51602 ).) Table 2 of this final rule (which is available via the internet on the CMS website) lists the out-migration adjustments for the FY 2025 wage index.
We did not receive any comments regarding the proposed policy to use the custom tabulations of the ACS 2016 through 2020 (5-year) Microdata as the basis for the out-migration adjustment. We are finalizing the policy as proposed. We also note that we published the raw data file received from the US Census Bureau on the FY 2025 IPPS Proposed Rule Home Page. The file is item 15 in the supplementary file section at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/fy-2025-ipps-proposed-rule-home-page .
In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42325 through 42339 ), we finalized a policy to address the artificial magnification of wage index disparities, based in part on comments we received in response to our request for information included in our FY 2019 IPPS/LTCH PPS proposed rule ( 83 FR 20372 through 20377 ). In the FY 2020 IPPS/LTCH final rule, based on those public comments and the growing disparities between wage index values for high- and low-wage-index hospitals, we explained that those growing disparities are likely caused, at least in part, by the use of historical wage data to prospectively set hospitals' wage indexes. That lag creates barriers to hospitals with low wage index values being able to increase employee compensation, because those hospitals will not receive corresponding increases in their Medicare payment for several years ( 84 FR 42327 ). Accordingly, we finalized a policy that provided certain low wage index hospitals with an opportunity to increase employee compensation without the usual lag in those increases being reflected in the calculation of the wage index (as they would expect to do if not for the lag). [ 194 ] We accomplished this by temporarily increasing the wage index values for certain hospitals with low wage index values and doing so in a budget neutral manner through an adjustment applied to the standardized amounts for all hospitals, as well as by changing the calculation of the rural floor. As explained in the FY 2020 IPPS/LTCH proposed rule ( 84 FR 19396 ) and final rule ( 84 FR 42329 ), we indicated that the Secretary has authority to implement the low wage index hospital policy proposal under both section 1886(d)(3)(E) of the Act and under his ( print page 69302) exceptions and adjustments authority under section 1886(d)(5)(I) of the Act.
We increased the wage index for hospitals with a wage index value below the 25th percentile wage index value for a fiscal year by half the difference between the otherwise applicable final wage index value for a year for that hospital and the 25th percentile wage index value for that year across all hospitals (the low wage index hospital policy). We stated in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42326 through 42328 ) our intention that this policy would be effective for at least 4 years, beginning in FY 2020, to allow employee compensation increases implemented by these hospitals sufficient time to be reflected in the wage index calculation.
We note that the FY 2020 low wage index hospital policy and the related budget neutrality adjustment are the subject of pending litigation in multiple courts. On July 23, 2024, the Court of Appeals for the D.C. Circuit held that the Secretary lacked authority under 1886(d)(3)(E) or 1886(d)(5)(I)(i) of the Act to adopt the low wage index hospital policy for FY 2020, and that the policy and related budget neutrality adjustment must be vacated. Bridgeport Hosp. v. Becerra, Nos. 22-5249, 22-5269, 2024 WL 3504407, at *7-*8 & n.6 (D.C. Cir. July 23, 2024). As of the date of this Rule's publication, the time to seek further review of the D.C. Circuit's decision in Bridgeport Hospital has not expired. See Fed. R. App. P. 40(a)(1). The government is evaluating the decision and considering options for next steps.
As noted earlier, we finalized this policy in the FY 2020 IPPS/LTCH final rule to provide low wage index hospitals with an opportunity to increase employee compensation without the usual lag in those increases being reflected in the calculation of the wage index (as they would expect to do if not for the lag). This continues to be the purpose of the policy. We stated in the FY 2020 IPPS/LTCH PPS final rule our intention that it would be in effect for at least 4 years beginning October 1, 2019 ( 84 FR 42326 ). We also stated we intended to revisit the issue of the duration of this policy in future rulemaking as we gained experience under the policy. What could not have been anticipated at the time the policy was promulgated was that implementation of the policy would occur during the COVID-19 PHE, which was declared starting in January of 2020 and continued until May of 2023. The effects of the COVID-19 PHE complicate our ability to evaluate the low wage index hospital policy and our ability to determine whether low-wage hospitals have been provided a sufficient opportunity to increase employee compensation under the policy without the usual lag.
In the proposed rule, to help gauge the impact of the COVID-19 PHE relative to the impact of the low wage index hospital policy, we examined the aggregate revenue each hospital reported on their FY 2020 cost reports from the COVID-19 PHE Provider Relief Fund, the Small Business Administration Loan Forgiveness program, and other sources of COVID-19 related funding such as payroll retention credits and state emergency relief funds. Specifically, we examined Worksheet G-3, lines 24.50 through 24.60 for each IPPS hospital's 2020 cost report. We found that hospitals in the aggregate reported $31.1 billion in COVID-19 related funding, and of that amount low-wage hospitals reported $3.6 billion. These amounts are much larger than, and likely had a much greater impact on hospital operations, the approximately $230 million impact of the low wage index hospital policy. [ 195 ] For example, COVID-19 related funding impacted the ability of hospitals, both low-wage hospitals and non-low wage hospitals, to change employee compensation in ways that overshadowed any differential impact of the low wage index hospital policy between the two groups that may have occurred in the absence of the COVID-19 PHE.
In addition to examining the COVID-19 related funding data, we also examined the wage index data itself. For the FY 2025 wage index the best available data typically would be from the FY 2021 wage data from hospital cost reports. As discussed earlier in more detail in section III.C, in considering the impacts of the COVID-19 PHE on the FY 2021 hospital wage data, we compared that data with recent historical data. While there are some differences, in the proposed rule we stated that it is not readily apparent how any changes due to the COVID-19 PHE differentially impacted the wages paid by individual hospitals. Furthermore, even if changes due to the COVID-19 PHE did differentially impact the wages paid by individual hospitals over time, it is not clear how those changes could be isolated from changes due to other reasons and what an appropriate potential methodology might be to adjust the data to account for the effects of the COVID-19 PHE. Our inability to isolate the wage data changes due to the COVID-19 PHE and disentangle them from changes due to the low wage index hospital policy makes isolating and evaluating the impact of the low wage index hospital policy challenging. We reached similar conclusions with respect to the FY 2020 hospital wage data.
To help further inform our FY 2025 rulemaking with respect to the low wage index hospital policy, in the proposed rule we stated that we also conducted an analysis of hospitals that received an increase to their wage index due to the policy in FY 2020 (referred to as the low wage index hospitals for brevity in the following discussion). Specifically, for each low wage index hospital we calculated the percent increase in its average hourly wages (AHWs) from FY 2019 to FY 2021 based on dividing its FY 2021 average hourly wage (using the wage data one year after the low wage index hospital policy was implemented in FY 2020, available on the FY 2025 IPPS Proposed Rule web page) by its average hourly wage from the FY 2019 wage data (the wage data one year before the low wage index hospital policy was implemented in FY 2020, available on the FY 2023 IPPS final rule web page). We performed the same calculation for the hospitals that were not low wage index hospitals. We then compared the distributions of the average hourly wage increases between the two groups. The results are shown in the following chart (Chart 1).

In general, the chart shows that the distribution of the changes in the average hourly wages of the low wage index hospitals (mean=15.1%, standard deviation=11.0%) is similar to the distribution of the changes in the average hourly wages of the non-low wage index hospitals (mean=14.7%, standard deviation=8.9%). Although some low-wage hospitals have indicated to us that they did use the increased payments they received under the low wage index hospital policy to increase wages more than they otherwise would have, the similarity in the two distributions indicates that, based on the audited wage data available to us, the policy has generally not yet had the effect of substantially reducing the wage index disparities that existed at the time the policy was promulgated. Also, to the extent that wage index disparities for a subset of low wage index hospitals has diminished, it is unclear to what extent that is attributable to the low wage index hospital policy given the effects of the COVID-19 PHE (as discussed later in this section).
The COVID-19 PHE ended in May of 2023. With regard to the wage index, 4 years is the minimum time before increases in employee compensation included in the Medicare cost report could be reflected in the wage index data. The first full fiscal year of wage data after the COVID-19 PHE is the FY 2024 wage data, which would be available for the FY 2028 IPPS/LTCH PPS rulemaking. As we explained earlier in this section, at the time the low wage index hospital policy was finalized, our intention was that it would be in effect for at least 4 fiscal years beginning October 1, 2019, and to revisit the issue of the duration of this policy as we gained experience under the policy. Because the effects of the COVID-19 PHE complicate our ability to evaluate the low wage index hospital policy and our ability to determine whether low-wage hospitals have been provided a sufficient opportunity to increase employee compensation under the policy without the usual lag, we proposed that the low wage index hospital policy and the related budget neutrality adjustment would be effective for at least three more years, beginning in FY 2025. We noted that this would result in the policy being in effect for at least 4 full fiscal years in total after the end of the COVID-19 PHE in May of 2023. We also stated that this will allow us to gain experience under the policy for the same duration and in an environment more similar to the one we expected at the time the policy was first promulgated.
To offset the estimated increase in IPPS payments to hospitals with wage index values below the 25th percentile wage index value, for FY 2025 and for subsequent fiscal years during which the low wage index hospital policy is in effect, we proposed to apply a budget neutrality adjustment in the same manner as we have applied it since FY 2020, as a uniform budget neutrality factor applied to the standardized amount. We refer readers to section II.A.4.f. of the Addendum to the proposed rule and this final rule for further discussion of the budget neutrality adjustment for FY 2025.
Comment: Several commenters supported the proposed low wage index hospital policy and asked CMS to continue the policy. Commenters benefiting from the low wage policy indicated that the policy has been helpful in addressing wage disparities and supporting hospitals in economically challenged areas. Commenters also stated that the policy prioritizes patients, promoting health equity and benefitting millions of patients across the country. Commenters conveyed that the policy allows for the ability to further sustain and build the health care system our changing population deserves, and to rebalance the disparity of care that exists between economically diverse areas of our nation.
Commenters indicated that the low wage index policy has helped to slightly level the playing field in Medicare reimbursement for rural hospitals and cautioned CMS that any efficacy analysis regarding the policy should recognize that the policy does not operate in a vacuum. According to commenters, low-wage hospitals face numerous challenges beyond just the wage index that make it difficult for them to significantly increase wages, particularly in relation to high-wage hospitals. Commenters stated that having a lower wage index over many years makes it difficult to ever catch up to high-wage hospitals. According to commenters, even though the wage index for many low-wage hospitals has increased since the policy began, their wage index remains significantly below the wage index for most high-wage hospitals.
Commenters also expressed that beyond staffing issues, low-wage hospitals generally have higher Medicare utilization in relation to total patient volume, and as a result Medicare losses are a higher proportion of their operations. The commenters indicated these hospitals face difficulty in making up for these losses as they receive less utilization from patients with traditional third-party insurance payment. ( print page 69304) Commenters explained that because of this, additional reimbursement provided by the low wage index hospital policy minimizes operating losses and allows operations to continue, thereby creating the potential for low-wage hospitals to be more competitive in recruiting staff than they would be absent the adjustment.
Response: We thank the commenters for their support of the low wage index hospital policy and for the proposal to extend the policy for at least three more years, beginning in FY 2025. We also appreciate learning from the commenters that the policy has been meaningful and effective in reducing wage index disparities. We also thank commenters for their thoughts on the unique circumstances faced by low-wage hospitals compared to high-wage hospitals with respect to the wage index.
Comment: Several commenters expressed their support for the continued implementation of the low wage index hospital policy but urged CMS to not include the budget neutral aspect, stating that CMS is not bound by statute to implement this policy in a budget neutral manner. A commenter stated that if low-wage hospitals were using increased payments associated with the low wage index hospital policy to increase wages at a rate faster than the national average (according to the commenter this would indicate the wage gap is closing), the budget neutrality adjustment associated with the policy should decrease, as it would cost less for CMS to maintain the policy over time as wages in bottom quartile markets converged with other CBSAs, compressing the wage index. According to this commenter, this is not happening as there has been a significant increase in the budget neutrality adjustment required to implement this policy in FY 2024 and FY 2025 suggesting that the bottom quartile policy has been ineffective. The commenter stated that they chose FY 2024 and FY 2025 for their analysis because these FYs are the first to consist of wage data impacted by the low wage index hospital policy, as FY 2024 used FY 2020 wage data, the FY the low wage index hospital policy was first effective, and FY 2025 uses FY 2021 wage data, the most current available FY wage data reflecting low wage index hospital policy data. Commenters also stated that the lever CMS has to pull to make the policy budget neutral, reducing the national standardized amount for all PPS hospitals nationally, intensifies historical Medicare underpayment and harms some of the very hospitals the policy is intended to help. Some commenters suggested that new funding replace the need for the policy to be budget neutral. These commenters stated that Medicare consistently reimburses inpatient PPS hospitals less than the cost of care and referenced that MedPAC estimates that hospitals' aggregate Medicare margins will be negative 13% in 2024 and that aggregate Medicare margins in 2022 were a negative 12.7% excluding federal relief funds. These commenters stated that these figures represent a continuance of a longstanding trend of substantially negative Medicare margins, [ 196 ] suggesting a strong need to add funds into the system by implementing the low wage hospital policy in a non-budget neutral manner. Commenters also stated that those hospitals that fall between approximately the 22nd and 25th percentile are receiving a reduction to the wage-adjusted standardized rate because the amount of benefit received is less than the cost to fund the benefit (the low wage index hospital policy budget neutrality factor applied is allegedly larger than the increase the hospital would receive due to the policy). These commenters suggested holding hospitals under the 25th percentile harmless.
Commenters also provided other suggestions for data and alternative methodologies to include: reducing the wage index for hospitals with values above the 75th percentile; working with Congress on a more permanent fix to address the disparities in the wage index by establishing a national floor for all hospitals; and seeking input from the hospital community on best overall reform options that will improve the sustainability of the workforce, especially in rural and underserved communities.
Response: As discussed in previous rulemaking regarding the low wage index hospital policy in response to comments, we disagree with the commenters that the low wage index hospital policy should be implemented in a non-budget neutral manner. Specifically, as we stated in response to similar comments in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42331 and 42332 ), the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45180 ), the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49007 ), and the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58979 ), under section 1886(d)(3)(E) of the Act, “[a]ny adjustments or updates” to the wage index are required to be implemented in a budget neutral manner. However, even if the wage index were not required to be budget neutral under section 1886(d)(3)(E) of the Act, we would consider it inappropriate to use the wage index to increase or decrease overall IPPS spending. As we stated in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42331 ), the wage index is designed to be a relative measure of the wages and wage-related costs of subsection (d) hospitals. As a result, as we explained in the FY 2020 IPPS/LTCH PPS final rule, if it were determined that section 1886(d)(3)(E) of the Act does not require the wage index to be budget neutral, we invoke our authority at section 1886(d)(5)(I) of the Act in support of such a budget neutrality adjustment.
Regarding the comment stating that an increase in the budget neutrality adjustment required to implement the low wage index hospital policy in FY 2024 and FY 2025 suggests that the policy has been ineffective, we disagree. As discussed earlier in this section, the effects of the COVID-19 PHE complicate our ability to evaluate the low wage index hospital policy and our ability to determine whether low-wage hospitals have been provided a sufficient opportunity to increase employee compensation under the policy without the usual lag. Because of this, we proposed that the low wage index hospital policy and the related budget neutrality adjustment would be effective for at least three more years, beginning in FY 2025. We noted that this would result in the policy being in effect for at least 4 full fiscal years in total after the end of the COVID-19 PHE in May of 2023. We also stated that this will allow us to gain experience under the policy for the same duration and in an environment more similar to the one we expected at the time the policy was first promulgated. For FY 2025 and for subsequent fiscal years during which the low wage index hospital policy is in effect, we also proposed to apply the associated budget neutrality adjustment in the same manner as we have applied it since FY 2020, as a uniform budget neutrality factor applied to the standardized amount.
With regard to the commenter's concern that application of the low wage index hospital policy may result in a reduction to overall payment if the amount of benefit received from the policy is less than the reduction to the standardized amount, as explained in response to comments in previous rulemaking, we believe we have applied both the quartile policy and the budget neutrality policy appropriately. ( print page 69305) Specifically, as we explained most recently in response to comments in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58979 ), the quartile adjustment is applied to the wage index, which results in an increase to the wage index for hospitals below the 25th percentile. The budget neutrality adjustment is applied to the standardized amount to ensure that the low wage index hospital policy is implemented in a budget neutral manner. Thus, consistent with our current methodology for implementing wage index budget neutrality under section 1886(d)(3)(E) of the Act and with how we implemented budget neutrality for the low wage index hospital policy in FY 2020, we believe it is appropriate to continue to apply a budget neutrality adjustment to the national standardized amount for all hospitals so that the low wage index hospital policy is implemented in a budget neutral manner for FY 2025.
Regarding the comment suggesting funds be added to the wage index system through implementation of the low wage index hospital policy in a non-budget neutral manner to account for alleged Medicare reimbursement underpayments, we disagree. We believe it would be inappropriate to use the wage index to increase or decrease overall IPPS spending. As we discuss elsewhere in this section, the intent of the low wage index hospital policy is to increase the accuracy of the wage index as a technical adjustment, and not to use the wage index as a policy tool to address non-wage issues related to hospitals, or the laudable goals of the overall financial health of hospitals in low-wage areas or broader wage index reform.
Regarding the comment about reducing the wage index for hospitals with values above the 75th percentile, as we discussed in our response to comments in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58979 ), in the FY 2020 IPPS/LTCH final rule ( 84 FR 42329 ), we noted that we originally proposed to reduce the wage index values for high wage index hospitals using a methodology analogous to the methodology used to increase the wage index values for low wage index hospitals, as described in section III.N.3.a. of the preamble of the proposed rule; that is, we proposed to decrease the wage index values for high wage index hospitals by a uniform factor of the distance between the hospital's otherwise applicable wage index and the 75th percentile wage index value for a fiscal year across all hospitals. In response to comments we received ( 84 FR 42329 and 42330 ), we acknowledged that some commenters presented reasonable policy arguments that we should consider further regarding the relationship between our proposed budget neutrality adjustment targeting high-wage hospitals and the design of the wage index to be a relative measure of the wages and wage-related costs of subsection (d) hospitals in the United States. Therefore, in the FY 2020 IPPS/LTCH final rule, we did not finalize our proposal to target that budget neutrality adjustment on high-wage hospitals ( 84 FR 42331 ). Regarding the comment about the establishment of a national floor for all hospitals, as we pointed out in response to comments in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58979 through 58980 ), we noted in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42338 through 42339 ) that we do not have evidence a national rural labor market exists or would be created if we were to adopt this alternative, and this alternative would not increase the accuracy of the wage index. Also, we believe we have applied both the quartile policy and the budget neutrality policy appropriately, as we explained in response to comments in the FYs 2021, 2022 and 2023 IPPS/LTCH PPS final rules and most recently FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58979 through 58980 ). The quartile adjustment is applied to the wage index, which resulted in an increase to the wage index for hospitals below the 25th percentile. The budget neutrality adjustment is applied to the standardized amount to ensure that the low wage index hospital policy is implemented in a budget neutral manner.
Comment: Several commenters opposed the low wage index hospital policy, stating that it is outside the agency's statutory authority under section 1886(d)(3)(E) of the Act. Specifically, some commenters expressed their belief that although the policy is intended to help rural hospitals, some rural hospitals in certain states do not benefit from this policy. Furthermore, commenters stated that the policy undermines the intent of the wage index by not recognizing real differences in labor costs.
Similar to comments made on the low wage index hospital policy in the FY 2022 IPPS/LTCH PPS rulemaking ( 86 FR 45179 ), a commenter argued that the low wage index hospital policy is ineffective, pointing to an Office of Inspector General (OIG) report [ 197 ] that suggests a complicated set of issues in local labor markets determines hospital wages in addition to Medicare payment rates. The commenter encouraged CMS to replicate the OIG's analysis using the most recently available audited wage data (FYs 2020 and 2021) and share the results in the final rule. According to the commenter, if the findings are the same as in the OIG's analysis, it will further demonstrate that the low wage index hospital policy has not had the intended effect and should not be continued.
Response: As we stated in response to similar comments in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58980 ), we believe we addressed the stated concerns in our responses to comments when we first finalized the low wage index hospital policy and the related budget neutrality adjustment in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42325 through 42332 ). Regarding the policy's effect on rural hospitals, as we stated in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42328 ), the wage index is a technical payment adjustment. The intent of the low wage index hospital policy is to increase the accuracy of the wage index as a technical adjustment, and not to use the wage index as a policy tool to address non-wage issues related to rural hospitals, or the laudable goals regarding the overall financial health of hospitals in low-wage areas or broader wage index reform. The low wage index hospital policy aims to increase the accuracy of the wage index as a relative measure, because it addresses barriers that low wage index hospitals face in increasing their employee compensation in ways that we would expect if there were no lag between the time a hospital increases employee compensation and the time these increases are reflected in the wage index, and allows those increases to be more timely reflected in the wage index. While one effect of the policy may be to improve the overall financial well-being of low-wage hospitals, and we would welcome that effect, it is not the rationale for our policy. In response to comments stating the policy exceeds CMS' statutory authority, we refer the commenters to our prior discussion of the authority for the policy in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42326 through 42332 ).
In response to the assertion that the low wage index hospital policy does not recognize real differences in labor costs, we continue to believe, for the reasons stated in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42327 and 42328 ), that by preserving the rank order in wage index values, our policy continues to reflect meaningful distinctions between the employee compensation costs faced by hospitals in different geographic ( print page 69306) areas. We note, as we have discussed earlier in this section, generally the wage index for the upcoming fiscal year is predictive in nature as wage index data that will be used for the upcoming fiscal year is the most current data and information available, which is usually historical data on a 4-year lag (for example, for the FY 2024 wage index we used cost report data from FY 2020). Thus, under the low wage index hospital policy, we believe the wage index for low wage index hospitals appropriately reflects the relative hospital wage level in those areas compared to the national average hospital wage level.
Some commenters stated that our policy is ineffective, referencing the OIG report cited above. As we explained in our response to comments in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45179 ), we believe that the numerous comments we continue to receive in support of this policy indicate that many low-wage hospitals are indeed helped by this policy. Specifically, comments stating that the policy has been helpful in addressing wage disparities and allowing low wage hospitals to be more competitive in recruiting staff, indicate that the policy helped low wage hospitals to raise wages. In response to the commenter's suggestion to refine our criteria to target a subset of low-wage hospitals, such as low-wage hospitals that are rural or that have negative profit margins, we believe that this would not maintain the rank order in wage index values. As we stated earlier, we believe that maintaining the rank order of wage index values is important to reflect meaningful distinctions between the employee compensation costs faced by hospitals in different geographic areas. Even several commenters that disagreed with our policy stressed the need for the wage index to be an accurate measure of the relative level of wages in different areas. A highly targeted approach that selected individual hospitals for relief would not maintain the rank order of wage index values and thus would be inconsistent with the construction of a relative measure of area wage levels. While it might be possible to refine our criteria for a more targeted approach, we believe it is reasonable to conclude that our current policy will have the intended effect of providing the opportunity for low-wage hospitals to increase compensation. As we stated earlier in this section, the policy being in effect for at least 4 full fiscal years in total after the end of the COVID-19 PHE will allow us to gain experience under the policy for the same duration and in an environment more similar to the one we expected at the time the policy was first promulgated. The availability of wage data from low-wage hospitals applicable to this time period will help us assess our reasonable expectation that hospitals will increase their employee compensation as a result of wage index increases under this policy. Once the increased employee compensation is reflected in the wage data, there may be no need for the continuation of the policy, given that we would expect the resulting increases in the wage index to continue after the temporary policy is discontinued. Again, we refer readers to the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45179 through 45180 ) for more information regarding our summary of and response to public comments about the aforementioned OIG report.
Comment: Many commenters noted that the low wage index hospital policy is currently the subject of pending litigation in Bridgeport. A few commenters urged CMS not to finalize the policy for FY 2025, or to wait until a final court decision is reached. Commenters suggested CMS should eliminate the budget neutrality adjustments for FYs 2020, 2021, 2022, 2023 and 2024 in light of Bridgeport.
Response: We appreciate the commenters' input. As noted previously, the FY 2020 low wage index hospital policy and the related budget neutrality adjustment are the subject of pending litigation in multiple courts. On July 23, 2024, the Court of Appeals for the D.C. Circuit held that the Secretary lacked authority under 1886(d)(3)(E) or 1886(d)(5)(I)(i) of the Act to adopt the low wage index hospital policy for FY 2020, and that the policy and related budget neutrality adjustment must be vacated. Bridgeport Hosp. v. Becerra, Nos. 22-5249, 22-5269, 2024 WL 3504407, at *7-*8 & n.6 (D.C. Cir. July 23, 2024). As of the date of this Rule's publication, the time to seek further review of the D.C. Circuit's decision in Bridgeport Hospital has not expired. See Fed. R. App. P. 40(a)(1). The government is evaluating the decision and considering options for next steps.
Commenters: Many commenters agreed with CMS that there is currently insufficient data to support modifying or discontinuing the low wage index hospital policy because of the COVID-19 pandemic impacts on wage data. According to commenters, hospitals are still recognizing lasting impacts of the COVID-19 PHE and appreciate the agency identifying this as a reality. Commenters indicated that the continuation of this critical policy in FY 2025 and beyond will provide stability and allow hospitals with an ability to recruit and retain desperately needed health care staff. Commenters recommended an extension of the low wage index hospital policy through FY 2030, at minimum, to ensure there is adequate post-pandemic wage data to support keeping or ending the policy. Commenters supporting an extension of the policy through FY 2030 referred to and supported CMS' acknowledgement that the first full FY of wage data after the COVID-19 PHE ended would not be available until the FY 2028 IPPS/LTCH PPS rulemaking given that the policy began in FY 2020. Commenters also noted that the policy should have a specific expiration date or definitive end date.
Regarding CMS' comparison of the distribution of the percentage change in AHWs from FY 2019 to FY 2021 for low wage index hospitals and non-low wage index hospitals, commenters agreed with CMS that the analysis did not show a substantial effect on reducing wage disparities. However, commenters asked CMS to evaluate whether this is due to other factors, such as inflation and other market forces impacted by the effects of the COVID-19 PHE, which are not clearly accounted for or represented in the current low wage index hospital policy, or if this is due to the ineffectiveness of the low wage index hospital policy. Commenters submitted the results of a separate analysis to emphasize that wages across the board have increased in recent years. Specifically, commenters submitted an analysis from the Kaiser Family Foundation (KFF) and Peterson Center, which evaluated changes in hospital employment data, including wage data, from February 2020 at the start of the COVID-19 pandemic through early 2024. Commenters stated that this analysis found that the average weekly earnings for healthcare employees had gone up 20.8% from $1,038 to $1,254 weekly in January 2024. Commenters further stated that even more specific to the IPPS, the report found that hospital workers wages saw a 20.3% increase between February 2020 to January 2024, going from $1,269 to $1,527 per week. [ 198 ] Commenters pointed out that CMS also observed this shift in wages, as outlined in CMS' analysis of audited wage data for FY 2020 to 2021 in the ( print page 69307) proposed rule, which saw larger increases in average hourly wages and wage indexes than compared to years prior and noted that CMS acknowledged that there are several challenges related to determining the cause of these changes, including uncertainty around the impact of the COVID-19 PHE. According to commenters, for a variety of reasons, including the COVID-19 PHE and other factors impacting wages, it is likely that changes observed in employee compensation may not be directly related to the low wage index hospital policy. These commenters urged CMS to tackle these issues in a more thoughtful and comprehensive manner that improves the standing of low wage index hospitals without impairing the standing of high wage index hospitals.
According to some commenters, CMS misunderstands the various programs that provided financial support to hospitals during the COVID-19 PHE. These commenters explained that all of the programs, including the Provider Relief Fund, state emergency relief funds and Small Business Administration Loan Forgiveness program, were intended in some fashion to replace some or all revenue hospitals lost due to decreases in demand associated with the COVID-19 PHE and cover the extraordinary costs hospitals incurred responding to the PHE that are not reimbursed through payments from Medicare, Medicaid, and commercial payers. The commenters stated that the funds from these programs were distributed using consistent criteria that applied to all eligible hospitals and therefore were not intended to advantage certain hospitals over others by increasing revenue for some hospitals and not others in a given category, as the low wage index hospital policy does. According to the commenters, the COVID-19 relief programs were intended to replace revenue that hospitals lost as a result of circumstances beyond their control and cover the extraordinary costs of saving patients' lives, mitigating the spread of a deadly pathogen, and protecting communities during a global pandemic. These commenters stated that if CMS was concerned data from the COVID-19 period were so flawed it could not determine the impact of the bottom quartile policy on impacted hospitals, it might stand to reason that the data would also be so flawed that they could not be used for payment updates. According to the commenters, CMS had enough confidence in the “normalcy” of data from years (federal and calendar) impacted by COVID-19 to use it to set MS-DRG weights, fixed-loss outlier thresholds, wage index values, and other key components of the IPPS in FY 2024 and it further proposes to use data from COVID-19 impacted years for the same functions in FY 2025. Finally, the commenters explained that CMS even acknowledges this in the proposed rule by stating that while there are some differences, it is not readily apparent how any changes due to the COVID-19 PHE differentially impacted the wages paid by individual hospitals. According to the commenters, the proposed rule attempts to justify this continuation by discussing the challenges of normalizing hospital wage data to understand the impact of this policy if changes due to the COVID-19 PHE did differentially impact wages paid by hospitals over time. The commenters stated that if CMS is confident enough in the data to use them for rate setting, then it should be confident enough to assume there was no differential impact that would spoil an impact analysis of the bottom quartile policy.
Response: We thank commenters for their input and concurrence regarding insufficient data to support modifying or discontinuing the policy because of the COVID-19 PHE impacts on wage data. We also thank the commenters for their support for this policy continuing for FY 2025 and beyond. We continue to believe that the comments in support of the policy, specifically comments from relatively low-wage hospitals stating that the increased payments under the policy have allowed them stability and an ability to recruit and retain desperately needed health care staff, have reduced hiring and employment barriers for these hospitals. Regarding the requests by commenters to extend the policy until FY 2030 or to establish a definitive end or expiration date for the policy, as we mentioned in the proposed rule, the COVID-19 PHE ended in May of 2023. Four years is the minimum time before increases in employee compensation included in the Medicare cost report could be reflected in the wage index data. The first full fiscal year of wage data after the COVID-19 PHE is the FY 2024 wage data, which would be available for the FY 2028 IPPS/LTCH PPS rulemaking. As we explained earlier in this section, at the time the low wage index hospital policy was finalized, our intention was that it would be in effect for at least 4 fiscal years beginning October 1, 2019, and to revisit the issue of the duration of this policy as we gained experience under the policy. Because the effects of the COVID-19 PHE complicate our ability to evaluate the low wage index hospital policy and our ability to determine whether low-wage hospitals have been provided a sufficient opportunity to increase employee compensation under the policy without the usual lag, we proposed that the low wage index hospital policy and the related budget neutrality adjustment would be effective for at least three more years, beginning in FY 2025. This would result in the policy being in effect for at least 4 full fiscal years in total after the end of the COVID-19 PHE in May of 2023. This will allow us to gain experience under the policy for the same duration and in an environment more similar to the one we expected at the time the policy was first promulgated. Until we are able to evaluate hospital wage data from the period after the end of the COVID-19 PHE and gain experience under the low wage index hospital policy to determine the policy's effectiveness, we are not able to determine or project a definitive end date of the policy. Therefore, in this final rule, we are not extending the policy until FY 2030, nor establishing a definitive end or expiration date.
Regarding the comments about CMS' attempt to gauge the impact of the COVID-19 PHE relative to the impact of the low wage index hospital policy by examining the aggregate revenue each hospital reported on their FY 2020 cost reports from the COVID-19 PHE Provider Relief Fund, the Small Business Administration Loan Forgiveness program, and other sources of COVID-19 related funding such as payroll retention credits and state emergency relief funds, we disagree with the commenters. Our intention was not to convey that the purpose of various COVID-19 related funding opportunities was the same as the purpose of the low wage index hospital policy, but instead, to provide a statistical comparison examining the overall amount hospitals reported in COVID-19 related funding to the portion of that amount the amount low-wage hospitals received. Our explanation in the proposed rule and earlier in this section also aimed to explain our thinking that COVID-19 related funding played a role in increasing hospital employee compensation, and since that was and remains the goal of the low wage index hospital policy, it was not possible to quantify which sources of funding, COVID-19 related or low wage index hospital policy, actually contributed to hospitals increasing employee compensation. In addition, as explained earlier in this section we compared FY 2021 wage data from hospital cost ( print page 69308) reports and historical cost report data to consider the impacts of the COVID-19 PHE on the FY 2021 hospital wage data. In both comparisons, the COVID-19 related funding data comparison between all hospitals and low-wage hospitals and the comparison between FY 2021 wage data from hospital cost reports and historical cost report data, while there were differences noted, as we stated in the proposed rule and again earlier in this section, it is not readily apparent how any changes due to the COVID-19 PHE differentially impacted the wages paid by individual hospitals. Again, as we have stated earlier in this section, the effects of the COVID-19 PHE complicate our ability to evaluate the low wage index hospital policy and our ability to determine whether low-wage hospitals have been provided a sufficient opportunity to increase employee compensation under the policy without the usual lag.
We appreciate the comment concerning CMS' use of data from the COVID-19 PHE period for payment update purposes. However, the purpose of and data used for general payment updates is different than that of the low wage index hospital policy. We primarily use two data sources in the IPPS and LTCH PPS ratesetting: claims data and cost report data. The claims data source is the Medicare Provider Analysis and Review (MedPAR) file, which includes fully coded diagnostic and procedure data for all Medicare inpatient hospital bills for discharges in a fiscal year. The cost report data source is the Medicare hospital cost report data files from the most recent quarterly Healthcare Cost Report Information System (HCRIS) release. Our goal is always to use the best available data overall for ratesetting. However, due to the impact of the COVID-19 PHE on our ordinary payment update data, we finalized modifications to our usual payment update procedures in order to satisfy the purpose of updating payments, approximating the inpatient experience at IPPS hospitals and LTCHs in FY 2024 ( 88 FR 58651 through 58553 ). As we discuss throughout this section, the purpose of the low wage index hospital policy is to provide low wage index hospitals with an opportunity to increase employee compensation without the usual lag in those increases being reflected in the calculation of the wage index (as they would expect to do if not for the lag). We also discussed earlier in this section that to the extent that wage index disparities for a subset of low wage index hospitals has diminished, it is unclear to what extent that is attributable to the low wage index hospital policy given the effects of the COVID-19 PHE (as discussed below). Again, as we stated earlier in this section, even if changes due to the COVID-19 PHE did differentially impact the wages paid by individual hospitals over time, it is not clear how those changes could be isolated from changes due to other reasons and what an appropriate potential methodology might be to adjust the data accordingly. Therefore, the concerns we identified about the use of data from the time period during the COVID-19 PHE are specific to the purpose of the low wage index hospital policy. Maintaining the policy for at least 4 full fiscal years in total after the end of the COVID-19 PHE in May of 2023 will allow us to gain experience under the policy for the same duration and in an environment more similar to the one we expected at the time the policy was first promulgated and will provide us with the best opportunity to evaluate the effectiveness of the policy.
Regarding the commenters' thoughts on CMS' comparison of the distribution of the percentage change in AHWs from FY 2019 to FY 2021 for low wage index hospitals and non-low wage index hospitals, we appreciate the separate analysis referenced by commenters that the commenters indicate confirms CMS' analysis that based on the data currently available, the low wage index hospital policy has not yet had the effect of substantially reducing the wage index disparities that existed at the time the policy was promulgated. We also appreciate the commenters' suggestions for further evaluation of data as it becomes available and acknowledgement of whether other factors may play a role in assessing the effectiveness of the low wage index hospital policy. As we explained earlier in this section, the uncertainty around the impact of the COVID-19 PHE and its effects on CMS' ability to assess and compare wage data make it difficult to sufficiently assess the effectiveness of the policy at this time. We also appreciate the input from commenters charging CMS to be as thoughtful and comprehensive as possible to address the effectiveness and implementation of this policy going forward. As we indicated in the proposed rule and previous rulemaking, we finalized this policy in the FY 2020 IPPS/LTCH final rule to provide low wage index hospitals with an opportunity to increase employee compensation without the usual lag in those increases being reflected in the calculation of the wage index (as they would expect to do if not for the lag). This continues to be the purpose of the policy. As we explained earlier in this section, at the time the low wage index hospital policy was finalized, our intention was that it would be in effect for at least 4 fiscal years beginning October 1, 2019, and to revisit the issue of the duration of this policy as we gained experience under the policy. The effects of the COVID-19 PHE complicate our ability to evaluate the low wage index hospital policy and our ability to determine whether low wage hospitals have been provided a sufficient opportunity to increase employee compensation under the policy without the usual lag. As we discussed in the proposed rule, if the policy were to be in effect for at least 4 full fiscal years in total after the end of the COVID-19 PHE in May of 2023, it would allow us to gain experience under the policy for the same duration and in an environment more similar to the one we expected at the time the policy was first promulgated.
Therefore, after consideration of the comments received, and for the reasons stated previously in the proposed rule, we are finalizing as proposed that the low wage index hospital policy and the related budget neutrality adjustment be effective for at least three more years, beginning in FY 2025. For purposes of the low wage index hospital policy, based on the data for this final rule, the table displays the 25th percentile wage index value across all hospitals for FY 2025.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49018 through 49021 ), we finalized a wage index cap policy and associated budget neutrality adjustment for FY 2023 and subsequent fiscal years. Under this policy, we apply a 5-percent cap on any decrease to a hospital's wage index from its wage index in the prior FY, regardless of the circumstances causing the decline. A hospital's wage index will not be less than 95 percent of its final wage index for the prior FY. If a hospital's prior FY wage index is calculated with the application of the 5-percent cap, the following year's wage index will not be less than 95 percent of the hospital's capped wage index in the prior FY. Except for newly opened hospitals, we apply the cap for a FY using the final wage index applicable to the hospital on the last day of the prior FY. A newly opened hospital will be paid the wage index for the area in ( print page 69309) which it is geographically located for its first full or partial fiscal year, and it will not receive a cap for that first year, because it will not have been assigned a wage index in the prior year. The wage index cap policy is reflected at § 412.64(h)(7). We apply the cap in a budget neutral manner through a national adjustment to the standardized amount each fiscal year. For more information about the wage index cap policy and associated budget neutrality adjustment, we refer readers to the discussion in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49018 through 49021 ).
We explained in the proposed rule that for FY 2025, we would apply the wage index cap and associated budget neutrality adjustment in accordance with the policies adopted in the FY 2023 IPPS/LTCH PPS final rule. We noted that the budget neutrality adjustment will be updated, as appropriate, based on the final rule data. We refer readers to the Addendum of this final rule for further information regarding the budget neutrality calculations.
Comment: Commenters thanked CMS for the 5% cap on all wage index decreases regardless of the circumstances causing the decline, including the adoption of revised CBSA delineations. Many commenters specifically stated that they appreciate CMS' recognition that significant year-over-year changes in the wage index can occur due to external factors beyond a hospital's control and that this policy increases predictability of IPPS payments. Many commenters supported the cap but urged CMS to apply this policy in a non-budget neutral manner. MedPAC supported the policy to cap wage index decreases, but urged CMS to apply a cap to wage index increases as well. A commenter stated that even a 5% decrease could be impactful to the financial stability of certain hospitals, and asked CMS to consider a smaller percentage point cap for safety net hospitals.
Response: We thank the commenters for their support. We note that we did not propose any changes to this policy in the FY 2025 IPPS/LTCH PPS proposed rule. With regard to the commenters requesting that CMS apply this policy in a non-budget neutral manner, we refer readers to our response to similar comments in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58981 ). We appreciate MedPAC's suggestion that the cap on wage index changes should also be applied to increases in the wage index. However, as we stated in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49021 ), one purpose of the proposed policy is to help mitigate the significant negative impacts of certain wage index changes. That is, we cap decreases because we believe that a hospital would be able to more effectively budget and plan when there is predictability about its expected minimum level of IPPS payments in the upcoming fiscal year. We do not have a policy to limit wage index increases because we do not believe such a policy is needed to enable hospitals to more effectively budget and plan their operations. Therefore, we believe it is appropriate for hospitals that experience an increase in their wage index value to receive that wage index value. With regard to the commenter's request for CMS to consider a smaller percentage point cap for safety net hospitals, we do not believe it is appropriate to bifurcate the policy to provide a greater benefit to specific hospitals, nor is it clear how CMS would define “safety net hospitals” for the specialized cap the commenter requested.
In this FY 2025 IPPS/LTCH PPS final rule, we have included the following wage index tables: Table 2 titled “Case-Mix Index and Wage Index Table by CCN”; Table 3 titled “Wage Index Table by CBSA”; Table 4A titled “List of Counties Eligible for the Out-Migration Adjustment under Section 1886(d)(13) of the Act”; and Table 4B titled “Counties redesignated under section 1886(d)(8)(B) of the Act (Lugar Counties).” We refer readers to section VI. of the Addendum to this final rule for a discussion of the wage index tables for FY 2025.
Section 1886(d)(3)(E) of the Act directs the Secretary to adjust the proportion of the national prospective payment system base payment rates that are attributable to wages and wage-related costs by a factor that reflects the relative differences in labor costs among geographic areas. It also directs the Secretary to estimate from time to time the proportion of hospital costs that are labor-related and to adjust the proportion (as estimated by the Secretary from time to time) of hospitals' costs that are attributable to wages and wage-related costs of the DRG prospective payment rates. We refer to the portion of hospital costs attributable to wages and wage-related costs as the labor-related share. The labor-related share of the prospective payment rate is adjusted by an index of relative labor costs, which is referred to as the wage index.
Section 403 of Public Law 108-173 amended section 1886(d)(3)(E) of the Act to provide that the Secretary must employ 62 percent as the labor-related share unless this would result in lower payments to a hospital than would otherwise be made. However, this provision of Public Law 108-173 did not change the legal requirement that the Secretary estimate from time to time the proportion of hospitals' costs that are attributable to wages and wage-related costs. Thus, hospitals receive payment based on either a 62-percent labor-related share, or the labor-related share estimated from time to time by the Secretary, depending on which labor-related share results in a higher payment.
In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45194 through 45208 ), we rebased and revised the hospital market basket to a 2018-based IPPS hospital market basket, which replaced the 2014-based IPPS hospital market basket, effective beginning October 1, 2021. Using the 2018-based IPPS market basket, we finalized a labor-related share of 67.6 percent for discharges occurring on or after October 1, 2021. In addition, in FY 2022, we implemented this revised and rebased labor-related share in a budget neutral manner ( 86 FR 45193 , 86 FR 45529 through 45530 ). However, consistent with section 1886(d)(3)(E) of the Act, we did not take into account the additional payments that would be made as a result of hospitals with a wage index less than or equal to 1.0000 being paid using a labor-related share lower than the labor-related share of hospitals with a wage index greater than 1.0000.
The labor-related share is used to determine the proportion of the national IPPS base payment rate to which the area wage index is applied. We include a cost category in the labor-related share if the costs are labor intensive and vary with the local labor market. In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45204 through 45207 ), we included in the labor-related share the national average proportion of operating costs that are attributable to the following cost categories in the 2018-based IPPS market basket: Wages and Salaries; Employee Benefits; Professional Fees: Labor-Related; Administrative and Facilities Support Services; Installation, Maintenance, and Repair Services; and All Other: Labor-Related Services. In the proposed rule, for FY 2025, we did not propose to make any further changes to the labor-related share. For FY 2025, we are finalizing the policy to continue to use a labor-related share of 67.6 percent for discharges occurring on or after October 1, 2024. We note that, ( print page 69310) consistent with our established frequency of rebasing the IPPS market basket every 4 years, we anticipate proposing to rebase and revise the IPPS market basket in the FY 2026 IPPS/LTCH PPS proposed rule. Our preliminary evaluation of more recent Medicare cost report data for IPPS hospitals for 2022 indicates that the major IPPS market basket cost weights (particularly the compensation and drug cost weights) are similar to those finalized in the 2018-based IPPS market basket.
As discussed in section V.B. of the preamble of this final rule, prior to January 1, 2016, Puerto Rico hospitals were paid based on 75 percent of the national standardized amount and 25 percent of the Puerto Rico-specific standardized amount. As a result, we applied the Puerto Rico-specific labor-related share percentage and nonlabor-related share percentage to the Puerto Rico-specific standardized amount. Section 601 of the Consolidated Appropriations Act, 2016 ( Pub. L. 114-113 ) amended section 1886(d)(9)(E) of the Act to specify that the payment calculation with respect to operating costs of inpatient hospital services of a subsection (d) Puerto Rico hospital for inpatient hospital discharges on or after January 1, 2016, shall use 100 percent of the national standardized amount. Because Puerto Rico hospitals are no longer paid with a Puerto Rico-specific standardized amount as of January 1, 2016, under section 1886(d)(9)(E) of the Act as amended by section 601 of the Consolidated Appropriations Act, 2016, there is no longer a need for us to calculate a Puerto Rico-specific labor-related share percentage and nonlabor-related share percentage for application to the Puerto Rico-specific standardized amount. Hospitals in Puerto Rico are now paid 100 percent of the national standardized amount and, therefore, are subject to the national labor-related share and nonlabor-related share percentages that are applied to the national standardized amount. Accordingly, for FY 2025, we did not propose a Puerto Rico-specific labor-related share percentage or a nonlabor-related share percentage.
Tables 1A and 1B, which are published in section VI. of the Addendum to this FY 2025 IPPS/LTCH PPS final rule and available via the internet on the CMS website, reflect the national labor-related share. Table 1C, in section VI. of the Addendum to this FY 2025 IPPS/LTCH PPS final rule and available via the internet on the CMS website, reflects the national labor-related share for hospitals located in Puerto Rico. For FY 2025, for all IPPS hospitals (including Puerto Rico hospitals) whose wage indexes are less than or equal to 1.0000, we are applying the wage index to a labor-related share of 62 percent of the national standardized amount. For all IPPS hospitals (including Puerto Rico hospitals) whose wage indexes are greater than 1.000, for FY 2025, we are applying the wage index to a labor-related share of 67.6 percent of the national standardized amount.
Comment: Several commenters recommended CMS raise the labor-related share from the current 67.6 percent to at least 72.8 percent, which is the figure CMS calculated for the proposed updated labor-related share for LTCHs for FY 2025. A commenter supported CMS not proposing to increase the labor-related share.
Response: We did not propose to make any further changes to the labor related share for FY 2025. Also, we do not believe it would be appropriate to use the labor-related share for LTCHs, which was calculated specifically for the LTCH PPS instead of the labor related share computed for the hospitals paid under the IPPS. As discussed earlier, for FY 2025, we are continuing to use a labor-related share of 67.6 percent for discharges occurring on or after October 1, 2024.
Section 1886(d)(5)(F) of the Act provides for additional Medicare payments to subsection (d) hospitals that serve a significantly disproportionate number of low-income patients. The Act specifies two methods by which a hospital may qualify for the Medicare disproportionate share hospital (DSH) adjustment. Under the first method, hospitals that are located in an urban area and have 100 or more beds may receive a Medicare DSH payment adjustment if the hospital can demonstrate that, during its cost reporting period, more than 30 percent of its net inpatient care revenues are derived from State and local government payments for care furnished to patients with low incomes. This method is commonly referred to as the “Pickle method.” The second method for qualifying for the DSH payment adjustment, which is the more commonly used method, is based on a complex statutory formula under which the DSH payment adjustment is based on the hospital's geographic designation, the number of beds in the hospital, and the level of the hospital's disproportionate patient percentage (DPP).
A hospital's DPP is the sum of two fractions: the “Medicare fraction” and the “Medicaid fraction.” The Medicare fraction (also known as the “SSI fraction” or “SSI ratio”) is computed by dividing the number of the hospital's inpatient days that are furnished to patients who were entitled to both Medicare Part A and Supplemental Security Income (SSI) benefits by the hospital's total number of patient days furnished to patients entitled to benefits under Medicare Part A. The Medicaid fraction is computed by dividing the hospital's number of inpatient days furnished to patients who, for such days, were eligible for Medicaid, but were not entitled to benefits under Medicare Part A, by the hospital's total number of inpatient days in the same period.

Because the DSH payment adjustment is part of the IPPS, the statutory references to “days” in section 1886(d)(5)(F) of the Act have been interpreted to apply only to hospital acute care inpatient days. Regulations located at 42 CFR 412.106 govern the Medicare DSH payment adjustment and specify how the DPP is calculated as well as how beds and patient days are counted in determining the Medicare DSH payment adjustment. Under § 412.106(a)(1)(i), the number of beds for the Medicare DSH payment adjustment is determined in accordance with bed counting rules for the IME adjustment under § 412.105(b).
Section 3133 of the Patient Protection and Affordable Care Act ( Pub. L. 111-148 ), as amended by section 10316 of the same Act and section 1104 of the Health Care and Education Reconciliation Act ( Pub. L. 111-152 ), added a section 1886(r) to the Act that modifies the methodology for computing the Medicare DSH payment adjustment. We refer to these provisions collectively as section 3133 of the Affordable Care Act. Beginning with discharges in FY 2014, hospitals that qualify for Medicare DSH payments under section 1886(d)(5)(F) of the Act receive 25 percent of the amount they previously would have received under the statutory formula for Medicare DSH payments. This provision applies equally to hospitals that qualify for DSH payments under section 1886(d)(5)(F)(i)(I) of the Act and those hospitals that qualify under the Pickle method under section 1886(d)(5)(F)(i)(II) of the Act.
The remaining amount, equal to an estimate of 75 percent of what otherwise would have been paid as Medicare DSH payments, reduced to reflect changes in the percentage of individuals who are uninsured, is available to make additional payments to each hospital that qualifies for Medicare DSH payments and that has uncompensated care. The payments to each hospital for a fiscal year are based on the hospital's amount of uncompensated care for a given time period relative to the total amount of uncompensated care for that same time period reported by all hospitals that receive Medicare DSH payments for that fiscal year.
Since FY 2014, section 1886(r) of the Act has required that hospitals that are eligible for DSH payments under section 1886(d)(5)(F) of the Act receive 2 separately calculated payments:
| Medicare DSH Payment | An empirically justified DSH payment equal to 25% of the amount determined under the statutory formula in section 1886(d)(5)(F) of the Act. |
| Medicare DSH Uncompensated Care Payment | An uncompensated care payment determined as the product of 3 factors, as discussed in this section. |
Specifically, section 1886(r)(1) of the Act provides that the Secretary shall pay to such subsection (d) hospital 25 percent of the amount the hospital would have received under section 1886(d)(5)(F) of the Act for DSH payments, which represents the empirically justified amount for such payment, as determined by the MedPAC in its March 2007 Report to Congress. [ 199 ] We refer to this payment as the “empirically justified Medicare DSH payment.”
In addition to this empirically justified Medicare DSH payment, section 1886(r)(2) of the Act provides that, for FY 2014 and each subsequent fiscal year, the Secretary shall pay to such subsection (d) hospital an additional amount equal to the product of three factors. The first factor is the difference between the aggregate amount of payments that would be made to subsection (d) hospitals under section 1886(d)(5)(F) of the Act if subsection (r) did not apply and the aggregate amount of payments that are made to subsection (d) hospitals under section 1886(r)(1) of the Act for such fiscal year. Therefore, this factor amounts to 75 percent of the payments that would otherwise be made under section 1886(d)(5)(F) of the Act.
The second factor is, for FY 2018 and subsequent fiscal years, 1 minus the percent change in the percent of individuals who are uninsured, as determined by comparing the percent of individuals who were uninsured in 2013 (as estimated by the Secretary, based on data from the Census Bureau or other sources the Secretary determines appropriate, and certified by the Chief Actuary of CMS) and the percent of individuals who were uninsured in the most recent period for which data are available (as so estimated and certified).
The third factor is a percent that, for each subsection (d) hospital, represents the quotient of the amount of uncompensated care for such hospital for a period selected by the Secretary (as estimated by the Secretary, based on appropriate data), including the use of alternative data where the Secretary determines that alternative data are available which are a better proxy for the costs of subsection (d) hospitals for treating the uninsured, and the aggregate amount of uncompensated care for all subsection (d) hospitals that receive a payment under section 1886(r) of the Act. Therefore, this third factor represents a hospital's uncompensated care amount for a given time period relative to the uncompensated care amount for that same time period for all hospitals that receive Medicare DSH payments in the applicable fiscal year, expressed as a percent.
For each hospital, the product of these three factors represents its additional payment for uncompensated care for the applicable fiscal year. We refer to the additional payment determined by these factors as the “uncompensated care payment.” In brief, the uncompensated care payment for an individual hospital is determined as the product of the following 3 factors:
| Factor 1 | 75% of the total amount of DSH payments that would otherwise be made under section 1886(d)(5)(F) of the Act. |
| Factor 2 | 1 minus the percent change in the percent of individuals who are uninsured. |
| Factor 3 | The hospital's uncompensated care amount relative to the uncompensated care amount for all hospitals that receive DSH payments, expressed as a percentage. |
Section 1886(r) of the Act applies to FY 2014 and each subsequent fiscal year. In the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50620 through 50647 ) and the FY 2014 IPPS interim final rule with comment period ( 78 FR 61191 through 61197 ), we set forth our policies for implementing the required changes to the Medicare DSH payment methodology made by section 3133 of the Affordable Care Act for FY 2014. In those rules, we noted that, because section 1886(r) of the Act modifies the payment required under section 1886(d)(5)(F) of the Act, it affects only the DSH payment under the operating IPPS. It does not revise or replace the capital IPPS DSH payment provided under the regulations at 42 CFR part 412, subpart M , which was established through the exercise of the Secretary's discretion in implementing the capital IPPS under section 1886(g)(1)(A) of the Act.
Finally, section 1886(r)(3) of the Act provides that there shall be no administrative or judicial review under section 1869, section 1878, or otherwise of any estimate of the Secretary for purposes of determining the factors described in section 1886(r)(2) of the Act or of any period selected by the Secretary for the purpose of determining those factors. Therefore, there is no administrative or judicial review of the estimates developed for purposes of applying the three factors used to determine uncompensated care payments, or of the periods selected to develop such estimates.
The payment methodology under section 3133 of the Affordable Care Act applies to “subsection (d) hospitals” that would otherwise receive a DSH payment made under section 1886(d)(5)(F) of the Act. Therefore, hospitals must receive empirically justified Medicare DSH payments in a fiscal year to receive an additional Medicare uncompensated care payment for that year. Specifically, section 1886(r)(2) of the Act states that, in addition to the empirically justified Medicare DSH payment made to a subsection (d) hospital under section 1886(r)(1) of the Act, the Secretary shall pay to “such subsection (d) hospitals” the uncompensated care payment. Section 1886(r)(2)'s reference to “such subsection (d) hospitals” refers to hospitals that receive empirically justified Medicare DSH payments under section 1886(r)(1) for the applicable fiscal year.
In the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50622 ) and the FY 2014 IPPS interim final rule with comment period ( 78 FR 61193 ), we explained that hospitals that are not eligible to receive empirically justified Medicare DSH payments in a fiscal year will not receive uncompensated care payments for that year. We also specified that we would make a determination concerning eligibility for interim uncompensated care payments based on each hospital's estimated DSH status (that is, eligibility to receive empirically justified Medicare DSH payments) for the applicable fiscal year (using the most recent data that are available). For the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36188 through 36189 ), we estimated DSH status for all hospitals using the most recent available SSI ratios and information from the most recent available Provider Specific File. We noted that FY 2020 SSI ratios available on the CMS website were the most recent available SSI ratios at the time of developing the proposed rule. [ 200 ] We stated that if more recent data on DSH eligibility became available before the final rule, we would use such data in the final rule. The FY 2021 SSI ratios are the most recent data available at the time of developing this FY 2025 IPPS/LTCH PPS final rule.
Our final determinations of a hospital's eligibility for uncompensated care and empirically justified Medicare DSH payments will be based on the hospital's actual DSH status at cost report settlement for FY 2025.
In the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50622 ) and in the rulemakings for subsequent fiscal years, we have specified our policies for several specific classes of hospitals within the scope of section 1886(r) of the Act. Eligible hospitals include the following:
- Subsection (d) Puerto Rico hospitals that are eligible for DSH payments also are eligible to receive empirically justified Medicare DSH payments and uncompensated care payments under section 1886(r) of the Act ( 78 FR 50623 and 79 FR 50006 ).
- Sole community hospitals (SCHs) that are paid under the IPPS Federal rate receive interim payments based on what we estimate and project their DSH status to be prior to the beginning of the fiscal year (based on the best available data at that time) subject to settlement through the cost report. If they receive interim empirically justified Medicare DSH payments in a fiscal year, they will also be eligible to receive interim uncompensated care payments for that fiscal year on a per discharge basis. Final eligibility determinations will be made at the end of the cost reporting period at settlement, and both interim empirically justified Medicare DSH payments and uncompensated care payments will be adjusted accordingly ( 78 FR 50624 and 79 FR 50007 ).
- Medicare-dependent, small rural hospitals (MDHs) are paid based on the IPPS Federal rate or, if higher, the IPPS Federal rate plus 75 percent of the amount by which the Federal rate is exceeded by the updated hospital-specific rate from certain specified base years ( 76 FR 51684 ). The IPPS Federal rate that is used in the MDH payment methodology is the same IPPS Federal rate that is used in the SCH payment methodology. Because MDHs are paid based on the IPPS Federal rate, they continue to be eligible to receive empirically justified Medicare DSH payments and uncompensated care payments if their DPP is at least 15 percent, and we apply the same process to determine MDHs' eligibility for interim empirically justified Medicare DSH and interim uncompensated care payments as we do for all other IPPS hospitals. Recently enacted legislation has extended the MDH program through December 31, 2024. We refer readers to section V.F. of the preamble of this final rule for further discussion of the MDH program.
Section 307 of the Consolidated Appropriations Act, 2024 extended the MDH program through December 31, 2024. We will continue to make a determination concerning an MDH's eligibility for interim empirically justified Medicare DSH and uncompensated care payments based on the hospital's estimated DSH status for the applicable fiscal year.
- IPPS hospitals that elect to participate in the Bundled Payments for Care Improvement Advanced (BPCI Advanced) model, will continue to be paid under the IPPS and, therefore, are eligible to receive empirically justified Medicare DSH payments and uncompensated care payments until the Model's final performance year, which ends on December 31, 2025. For further information regarding the BPCI Advanced model, we refer readers to the CMS website at https://innovation.cms.gov/innovation-models/bpci-advanced .
- IPPS hospitals that participate in the Comprehensive Care for Joint Replacement (CJR) Model's ( 80 FR 73300 ) continue to be paid under the IPPS and, therefore, are eligible to receive empirically justified Medicare DSH payments and uncompensated care ( print page 69313) payments We refer the reader to the final rule that appeared in the May 3, 2021, Federal Register ( 86 FR 23496 ), which extended the CJR Model for an additional three performance years. The Model's final performance year ends on December 31, 2024. For additional information on the CJR Model, we refer readers to the CMS website at https://www.cms.gov/priorities/innovation/innovation-models/CJR .
- Transforming Episode Accountability Model (TEAM) is a new episode-based payment model, which is discussed in section X.A. of the preamble of this final rule. Hospitals participating in TEAM would continue to be paid under the IPPS and, therefore, are eligible to receive empirically justified Medicare DSH payments and uncompensated care payments. The model's start date is January 1, 2026.
Ineligible hospitals include the following:
- Maryland hospitals are not eligible to receive empirically justified Medicare DSH payments and uncompensated care payments under the payment methodology of section 1866(r) of the Act because they are not paid under the IPPS. As discussed in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41402 through 41403 ), CMS and the State have entered into an agreement to govern payments to Maryland hospitals under a new payment model, the Maryland Total Cost of Care (TCOC) Model, which began on January 1, 2019. Under the Maryland TCOC Model, which concludes on December 31, 2026, Maryland hospitals are not paid under the IPPS and are ineligible to receive empirically justified Medicare DSH payments and uncompensated care payments under section 1886(r) of the Act.
- SCHs that are paid under their hospital-specific rate are not eligible for Medicare DSH and uncompensated care payments ( 78 FR 50623 and 50624 ).
- Hospitals participating in the Rural Community Hospital Demonstration Program are not eligible to receive empirically justified Medicare DSH payments and uncompensated care payments under section 1886(r) of the Act because they are not paid under the IPPS ( 78 FR 50625 and 79 FR 50008 ). The Rural Community Hospital Demonstration Program was originally authorized for a 5-year period by section 410A of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) ( Pub. L. 108-173 ). [ 201 ] The period of participation for the last hospital in the demonstration under this most recent legislative authorization will end on June 30, 2028. Under the payment methodology that applies during this most recent extension of the demonstration program, participating hospitals do not receive empirically justified Medicare DSH payments, and they are excluded from receiving interim and final uncompensated care payments. At the time of development of this final rule, we believe 23 hospitals may participate in the demonstration program at the start of FY 2025.
In response to our comment solicitation on these policies in the proposed rule, we received comments related to the eligibility of SCHs paid under hospital-specific rates and MDHs to receive empirically justified DSH and uncompensated care payments. Because we consider these public comments to be outside the scope of the proposed rule, we are not addressing them in this final rule.
As we have discussed earlier, section 1886(r)(1) of the Act requires the Secretary to pay 25 percent of the amount of the Medicare DSH payment that would otherwise be made under section 1886(d)(5)(F) of the Act to a subsection (d) hospital. Because section 1886(r)(1) of the Act merely requires the Secretary to pay a designated percentage of these payments, without revising the criteria governing eligibility for DSH payments or the underlying payment methodology, we stated in the FY 2014 IPPS/LTCH PPS final rule that we did not believe that it was necessary to develop any new operational mechanisms for making such payments.
Therefore, in the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50626 ), we implemented this provision by advising Medicare Administrative Contractors (MACs) to simply adjust subsection (d) hospitals' interim claim payments to an amount equal to 25 percent of what would have been paid if section 1886(r) of the Act did not apply. We also made corresponding changes to the hospital cost report so that these empirically justified Medicare DSH payments could be settled at the appropriate level at the time of cost report settlement. We provided more detailed operational instructions and cost report instructions following issuance of the FY 2014 IPPS/LTCH PPS final rule that are available on the CMS website at https://www.cms.gov/Regulations-and-Guidance/Guidance/Transmittals/2014-Transmittals-Items/R5P240.html .
In response to our comment solicitation on these policies in the proposed rule, a commenter stated that some subsection (d) hospitals' ability to meet the eligibility requirements for empirically justified DSH payments is at risk due to changes to the Medicaid fraction of their DPPs. The commenter explained that many hospitals will no longer be eligible for empirically justified payments as a result of the unwinding of the Medicaid continuous enrollment condition. The commenter also stated that the unexpectedly high rate of Medicaid beneficiaries losing coverage because of redeterminations is placing many hospitals at risk of falling below the 15 percent minimum DPP. The commenter requested that CMS allow hospitals whose eligibility for empirically justified payments has been impacted by unwinding to receive empirically justified payments, retroactively and in the future. Because we consider this public comment to be outside the scope of the proposed rule, we are not addressing this comment in this final rule.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49047 through 49051 ), we established a new supplemental payment for IHS/Tribal hospitals and hospitals located in Puerto Rico for FY 2023 and subsequent fiscal years. This payment was established to help to mitigate the impact of the decision to discontinue the use of low-income insured days as a proxy for uncompensated care costs for these hospitals and to prevent undue long-term financial disruption for these providers. The regulations located at 42 CFR 412.106(h) govern the supplemental payment. In brief, the supplemental payment for a fiscal year is determined as the difference between the hospital's base year amount and its uncompensated care payment for the applicable fiscal year as determined under § 412.106(g)(1). The base year ( print page 69314) amount is the hospital's FY 2022 uncompensated care payment adjusted by one plus the percent change in the total uncompensated care amount between the applicable fiscal year (that is, FY 2025 for purposes of this rulemaking) and FY 2022, where the total uncompensated care amount for a fiscal year is determined as the product of Factor 1 and Factor 2 for that year. If the base year amount is equal to or lower than the hospital's uncompensated care payment for the current fiscal year, then the hospital would not receive a supplemental payment because the hospital would not be experiencing financial disruption in that year as a result of the use of uncompensated care data from the Worksheet S-10 in determining Factor 3 of the uncompensated care payment methodology.
In the FY 2025 IPPS/LTCH PPS proposed rule, we did not propose any changes to the methodology for determining supplemental payments. For FY 2025, we will calculate the supplemental payments to eligible IHS/Tribal and Puerto Rico hospitals consistent with the methodology described in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49047 through 49051 ) and § 412.106(h).
As discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49048 and 49049 ), the eligibility and payment processes for the supplemental payment are consistent with the processes for determining eligibility to receive interim and final uncompensated care payments adopted in FY 2014 IPPS/LTCH PPS final rule. We note that the MAC will make a final determination with respect to a hospital's eligibility to receive the supplemental payment for a fiscal year, in conjunction with its final determination of the hospital's eligibility for DSH payments and uncompensated care payments for that fiscal year.
Comment: One commenter reiterated their recommendations that were submitted in response to the proposal to establish these supplemental payments in the FY 2023 IPPS/LTCH PPS proposed rule. The commenter recommended that CMS calculate the supplemental payment for Puerto Rico hospitals using a base year amount determined using a Medicare SSI days proxy of at least 42 percent of the hospital's Medicaid days, to reflect the local poverty level, instead of the current base year amount, which incorporates the proxy that was applied from FY 2017 through FY 2022 of 14 percent of the hospital's Medicaid days and that was based on national data on the relationship between Medicare SSI days and Medicaid days. The commenter also requested that CMS extend eligibility for uncompensated care payments to all acute care hospitals in Puerto Rico, including those that do not qualify for empirically justified DSH payments, stating that it is consistent with the plain language and intent of Section 3133 of the Affordable Care Act. The commenter also stated that there are eight Puerto Rico hospitals that are projected to not receive empirically justified DSH payments for FY 2025 and these hospitals may miss the qualifying threshold because of the lack of SSI coverage for residents of the U.S. territories. As an alternative to the recommended policy of extending eligibility for uncompensated care payments to all acute care hospitals in Puerto Rico, the same commenter proposed that CMS could determine a hospital's eligibility to receive uncompensated care payments and supplemental payments using the suggested proxy data for the hospitals' Medicare SSI days of 42 percent.
Another commenter thanked CMS for continuing to provide supplemental payments but requested that CMS evaluate alternatives that would better support hospitals in Puerto Rico if uninsured days increased. This commenter asserted that the current supplemental payment policy only protects against the reduction of uncompensated care payments below FY 2022 levels. The commenter stated that the current policy is not helpful if uninsured patient volumes rise above FY 2022 levels. The same commenter further expressed that they would support a return to the prior method of using a proxy to determine uninsured days for hospitals in Puerto Rico given the challenges related to Worksheet S-10 data collection for hospitals in Puerto Rico.
Response: We appreciate the concerns and input raised by commenters regarding the calculation of Factor 3 for hospitals in Puerto Rico and IHS and Tribal hospitals. We continue to recognize the unique financial circumstances and challenges faced by Puerto Rico hospitals related to uncompensated care cost reporting on Worksheet S-10.
Regarding the commenter's request that all acute care hospitals in Puerto Rico receive uncompensated care payments regardless of DSH eligibility, we refer readers to the policy initially adopted in the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50622 and 50623 ), which explains that hospitals, including Puerto Rico hospitals, must be eligible to receive empirically justified Medicare DSH payments to receive an uncompensated care payment for that fiscal year. As discussed earlier in this section of this final rule and in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49048 and 49049 ), the processes for determining eligibility for supplemental payments and making interim and final payments are consistent with the processes for determining eligibility to receive interim and final uncompensated care payments adopted in the FY 2014 IPPS/LTCH PPS final rule and the approach used to make interim uncompensated care payments on a per discharge basis.
With respect to the commenters who recommended that CMS determine eligibility for uncompensated care payments and supplemental payments using the suggested Medicare SSI days proxy of 42 percent and calculate the supplemental payment for Puerto Rico hospitals using a base year amount determined from that same Medicare SSI days proxy data, we note that in the FY 2025 IPPS/LTCH PPS proposed rule, we did not propose to adopt any changes to our policies for determining eligibility for uncompensated care payments or supplemental payments, nor did we propose changes to our methodology for calculating supplemental payments. We also note that we did not propose to adopt a proxy for Puerto Rico hospitals' Medicare SSI days for purposes of determining eligibility for empirically justified DSH payments. Therefore, we consider these comments to be outside the scope of the proposed rule. However, we refer readers to our responses to similar comments in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58992 and 58993 ) and the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49049 and 49050 ) for further discussion on these issues.
Concerning the comment encouraging CMS to evaluate alternatives to supplemental payments to better support hospitals in the case of increasing uninsured days, including using a proxy to determine uninsured days for hospitals in Puerto Rico, we refer readers to our responses to similar comments in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48780 ) and the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58640 ). As we explained in those rulemakings, prior to FY 2023, we used low-income insured days as a proxy for uncompensated care costs. Fluctuations in uninsured days were never a direct consideration in the calculation of uncompensated care payments. Therefore, we continue to believe that supplemental payments, which are based on the FY 2022 uncompensated care payments calculated for Puerto ( print page 69315) Rico hospitals and IHS and Tribal hospitals using low income insured days proxy data, are the appropriate approach for hospitals located in Puerto Rico and IHS and Tribal hospitals.
As discussed earlier in this section, for FY 2025, we will calculate the supplemental payments to eligible IHS/Tribal and Puerto Rico hospitals consistent with the methodology described in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49047 through 49051 ) and § 412.106(h).
As we discussed earlier, section 1886(r)(2) of the Act provides that, for each eligible hospital in FY 2014 and subsequent years, the uncompensated care payment is the product of three factors, which are discussed in the next sections.
Section 1886(r)(2)(A) of the Act establishes Factor 1 in the calculation of the uncompensated care payment. The regulations located at 42 CFR 412.106(g)(1)(i) govern the Factor 1 calculation. Under a prospective payment system, we would not know the precise aggregate Medicare DSH payment amounts that would be paid for a fiscal year until cost report settlement for all IPPS hospitals is completed, which occurs several years after the end of the fiscal year. Therefore, section 1886(r)(2)(A)(i) of the Act provides authority to estimate this amount by specifying that, for each fiscal year to which the provision applies, such amount is to be estimated by the Secretary. Similarly, we would not know the precise aggregate empirically justified Medicare DSH payment amounts that would be paid for a fiscal year until cost report settlement for all IPPS hospitals is completed. Thus, section 1886(r)(2)(A)(ii) of the Act provides authority to estimate this amount. In brief, Factor 1 is the difference between the Secretary's estimates of: (1) the amount that would have been paid in Medicare DSH payments for the fiscal year, in the absence of section 1886(r) of the Act; and (2) the amount of empirically justified Medicare DSH payments that are made for the fiscal year, which takes into account the requirement to pay 25 percent of what would have otherwise been paid under section 1886(d)(5)(F) of the Act.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36190 ), we proposed to continue the policy that has applied since the FY 2014 final rule ( 78 FR 50627 through 50631 ): to determine Factor 1 from the most recently available estimates of the aggregate amount of Medicare DSH payments that would be made for FY 2025 in the absence of section 1886(r)(1) of the Act and the aggregate amount of empirically justified Medicare DSH payments that would be made for FY 2025, both as calculated by CMS' Office of the Actuary (OACT). Consistent with the policy that has applied in previous years, these estimates will not be revised or updated subsequent to publication of our final projections in this FY 2025 IPPS/LTCH PPS final rule.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36190 through 36192 ), to calculate both estimates, we used the most recently available projections of Medicare DSH payments for the fiscal year, as calculated by OACT using the most recently filed Medicare hospital cost reports with Medicare DSH payment information and the most recent DPPs and Medicare DSH payment adjustments provided in the IPPS Impact File. The projection of Medicare DSH payments for the fiscal year is also partially based on OACT's Part A benefits projection model, which projects, among other things, inpatient hospital spending. Projections of DSH payments additionally require projections of expected increases in utilization and case-mix. The assumptions that were used in making these inpatient hospital spending, utilization, and case-mix projections and the resulting estimates of DSH payments for FY 2022 through FY 2025 are discussed later in this section and in the table titled “Factors Applied for FY 2022 through FY 2025 to Estimate Medicare DSH Expenditures Using FY 2021 Baseline.”
For purposes of calculating Factor 1 and modeling the impact of the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36190 through 36192 ), we used OACT's January 2024 Medicare DSH estimates, which were based on data from the December 2023 update of the Medicare Hospital Cost Report Information System (HCRIS) and the FY 2024 IPPS/LTCH PPS final rule IPPS Impact File, published in conjunction with the publication of the FY 2024 IPPS/LTCH PPS final rule. Because SCHs that are projected to be paid under their hospital-specific rate are ineligible for empirically justified Medicare DSH payments and uncompensated care payments, they were excluded from the January 2024 Medicare DSH estimates. Because Maryland hospitals are not paid under the IPPS, they are also ineligible for empirically justified Medicare DSH payments and uncompensated care payments and were also excluded from OACT's January 2024 Medicare DSH estimates.
The 23 hospitals that CMS expects will participate in the Rural Community Hospital Demonstration Program in FY 2025 were also excluded from OACT's January 2024 Medicare DSH estimates because under the payment methodology that applies during the demonstration, these hospitals are not eligible to receive empirically justified Medicare DSH payments or uncompensated care payments.
For the proposed rule, using the data sources previously discussed, OACT's January 2024 estimates of Medicare DSH payments for FY 2025 without regard to the application of section 1886(r)(1) of the Act was approximately $13.943 billion. Therefore, also based on OACT's January 2024 Medicare DSH estimates, the estimate of empirically justified Medicare DSH payments for FY 2025, with the application of section 1886(r)(1) of the Act, was approximately $3.486 billion (or 25 percent of the total amount of estimated Medicare DSH payments for FY 2025). Under § 412.106(g)(1)(i), Factor 1 is the difference between these two OACT estimates. Therefore, in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35934 ), we proposed that Factor 1 for FY 2025 would be $10,457,250,000, which is equal to 75 percent of the total amount of estimated Medicare DSH payments for FY 2025 ($13.943 billion minus $3.486 billion). We noted that, consistent with our approach in previous rulemakings, OACT intended to use more recent data that may become available for purposes of projecting the final Factor 1 estimates for the FY 2025 IPPS/LTCH PPS final rule ( 89 FR 36191 ).
In the FY 2025 IPPS/LTCH PPS proposed rule, we noted that the Factor 1 estimates for IPPS/LTCH PPS proposed rules are generally consistent with the economic assumptions and actuarial analysis used to develop the President's Budget estimates under current law, and Factor 1 estimates for IPPS/LTCH PPS final rules are generally consistent with those used for the Midsession Review of the President's Budget. [ 202 ] Consistent with historical practice, we stated in the proposed rule that we expected the Midsession Review would have updated economic assumptions and actuarial analysis, which we would use for the ( print page 69316) development of Factor 1 estimates in the FY 2025 IPPS/LTCH PPS final rule.
For a general overview of the principal steps involved in projecting future inpatient costs and utilization, we referred readers to the “2024 Annual Report of the Boards of Trustees of the Federal Hospital Insurance and Federal Supplementary Medical Insurance Trust Funds,” available on the CMS website at https://www.cms.gov/oact/tr/2024 under “Downloads.” [ 203 ] The actuarial projections contained in these reports are based on numerous assumptions regarding future trends in program enrollment, utilization and costs of health care services covered by Medicare, as well as other factors affecting program expenditures. In addition, although the methods used to estimate future costs based on these assumptions are complex, they are subject to periodic review by independent experts to ensure their validity and reasonableness. We also referred readers to the 2018 Actuarial Report on the Financial Outlook for Medicaid for a discussion of general issues regarding Medicaid projections (available at https://www.cms.gov/data-research/research/actuarial-studies/actuarial-report-financial-outlook-medicaid ).
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36190 through 36192 ), we included information regarding the data sources, methods, and assumptions employed by OACT's actuaries in determining our estimate of Factor 1. We indicated the historical HCRIS data update OACT used to estimate Medicare DSH payments; we explained that the most recent Medicare DSH payment adjustments provided in the IPPS Impact File were used, and we provided the components of all the update factors that were applied to the historical data to estimate the Medicare DSH payments for the upcoming fiscal year, along with the associated rationale and assumptions. The discussion also included descriptions of the “Other” and “Discharges” assumptions and provided additional information regarding how we address Medicaid expansion.
We invited public comments on our proposed Factor 1 for FY 2025.
Comment: As in previous years, some commenters expressed concerns and requested greater transparency in the methodology used by CMS and OACT to calculate Factor 1. A few commenters emphasized their inability to accurately replicate CMS' calculations without clarity on how inputs, such as the effects of the COVID-19 PHE on Medicare discharges, case mix, Medicaid enrollment, and subsequent disenrollment through redeterminations, impact Factor 1 estimates. Some of these commenters requested that CMS provide details of its Factor 1 calculation in advance of the publication of the IPPS/LTCH PPS final rule and in the IPPS/LTCH PPS proposed rule each year going forward, so that sufficient data is available to replicate CMS' DSH payment calculations and enable commenters to provide more informed comments in future years. Another commenter requested that CMS provide detailed explanations for how the agency calculates Factor 1 to ensure safety net providers are not being disproportionately impacted.
A few commenters asserted that the lack of opportunity afforded to hospitals to review the data used in rulemaking is in violation of the Administrative Procedure Act. These commenters expressed concerns about the lack of transparency in how Factor 1 is calculated, arguing that hospitals cannot meaningfully comment on the Factor 1 calculation methodology given the lack of details provided by CMS in each IPPS/LTCH PPS proposed rule. In particular, these commenters stated that the FY 2025 IPPS/LTCH PPS proposed rule provided neither sufficient details nor a complete explanation of the treatment of Medicaid expansions in the calculation for Factor 1.
Additionally, while some commenters thanked CMS for increasing the “Other” factor from the amount finalized in the FY 2024 final rule, several commenters stated that CMS failed to provide sufficient details on how the “Other” factor is calculated, including both the overall calculation and individual inputs used to determine the estimate. Some of these commenters requested that CMS publish a detailed methodology of its “Other” calculation, specifying how all components contribute to changes in its estimate from year to year. Other commenters expressed concern about the lack of clarity regarding the ending of COVID-19 PHE flexibilities, such as payment add-ons and the unwinding of the Medicaid continuous enrollment condition, and their impact on the “Other” factor. These commenters suggested that CMS address this issue by disaggregating the variables that contribute to the “Other” factor and then demonstrating the separate impacts of each of those variables on the final value. A couple of commenters requested that CMS clarify why the “Other” factor frequently varies in successive rulemaking cycles.
Response: We thank the commenters for their input. We disagree with commenters' assertion regarding the lack of transparency with respect to the methodology and assumptions used in the calculation of Factor 1. As explained in the FY 2025 IPPS/LTCH PPS proposed rule and in this section of this final rule, we have been and continue to be transparent about the methodology and data used to estimate Factor 1. Regarding the commenters who reference the Administrative Procedure Act, we note that under the Administrative Procedure Act, a proposed rule is required to include either the terms or substance of the proposed rule or a description of the subjects and issues involved. In this case, the FY 2025 IPPS/LTCH PPS proposed rule ( 86 FR 36190-36192 ) included a detailed discussion of our proposed Factor 1 methodology and the data sources that would be used in making our final estimate. Accordingly, we believe commenters were able to meaningfully comment on our proposed estimate of Factor 1.
To provide additional context, and as we have explained in prior rulemakings ( see example, 88 FR 58995 ), we note that Factor 1 is not estimated in isolation from other projections made by OACT. The Factor 1 estimates for the proposed rules are generally consistent with the economic assumptions and actuarial analyses used to develop the President's Budget estimates under current law, and the Factor 1 estimates for the final rule are generally consistent with those used for the Midsession Review of the President's Budget. As we have in the past, we refer readers to the “Midsession Review of the President's FY 2025 Budget” for additional information on the development of the President's Budget and the specific economic assumptions used in the Midsession Review of the President's FY 2025 Budget, available on the Office of Management and Budget website at: https://www.whitehouse.gov/omb/budget . Consistent with our prior rulemakings, in the FY 2025 IPPS/LTCH proposed rule, we indicated that we expected that the Midsession Review would have updated economic assumptions and actuarial analysis, which would be used in the development of Factor 1 estimates for this final rule. We recognize that our reliance on the economic assumptions and actuarial analyses used to develop the President's Budget and the Midsession Review of the President's Budget in estimating Factor 1 has an ( print page 69317) impact on hospitals, health systems, and other impacted parties who wish to replicate the Factor 1 calculation by, for example, modeling the relevant Medicare Part A portion of the President's Budget. Yet, we believe commenters are able to meaningfully comment on our proposed estimate of Factor 1 without replicating the budget.
For a general overview of the principal steps involved in projecting future inpatient costs and utilization, we refer readers to the “2024 Annual Report of the Boards of Trustees of the Federal Hospital Insurance and Federal Supplementary Medical Insurance Trust Funds,” available under “Downloads” on the CMS website at: https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/ReportsTrustFunds/index.html . We note that the annual reports of the Medicare Boards of Trustees to Congress represent the Federal Government's official evaluation of the financial status of the Medicare Program. The actuarial projections contained in these reports are based on numerous assumptions regarding future trends in program enrollment, utilization, and costs of health care services covered by Medicare, as well as other factors affecting program expenditures. In addition, given that the methods used to estimate future costs based on these assumptions are complex, they are subject to periodic review by independent experts to ensure their validity and reasonableness.
Additionally, as described in more detail later in this section, in the FY 2025 IPPS/LTCH PPS proposed rule, we included information regarding the data sources, methods, and assumptions employed by the actuaries to determine the OACT's estimate of Factor 1. We explained that the most recent Medicare DSH payment adjustments provided in the IPPS Impact File were used, and we provided the components of all update factors that were applied to the historical data to estimate the Medicare DSH payments for the upcoming fiscal year, along with the associated rationale and assumptions. This discussion also included a description of the “Other,” “Case-Mix,” and “Discharges” assumptions, as well as additional information regarding the estimated impact of the COVID-19 PHE on our calculation of Factor 1. For additional context, our calculation of the “Other” factor for FY 2025 reflects the expectation that DSH payments will grow faster than IPPS payments in 2025.
Regarding the commenter who expressed concern that our proposed calculation of Factor 1 would disproportionately impact safety net providers, we continue to believe that estimating Factor 1 based on the economic data and assumptions detailed in this final rule and the FY 2025 IPPS/LTCH PPS proposed rule is appropriate and consistent with the requirements of section 1886(r)(2)(A) of the Act.
Comment: Many commenters requested that CMS provide additional detail on the calculations and assumptions related to the “Discharges” component used in the Factor 1 formula. A couple commenters asked that CMS provide an explanation as to why the “Discharges” component for FY 2023 and FY 2024 finalized in the FY 2024 IPPS/LTCH PPS final rule decreased in the FY 2025 IPPS proposed rule. Several commenters questioned the actuarial assumption of “recent trends recovering back to the long-term trend and assumption related to how many beneficiaries will be enrolled in Medicare Advantage (MA) plans.” A commenter requested that CMS ensure the “Discharges” component of Factor 1 accurately reflects trends in Medicare Fee-for-Service (FFS) utilization in FY 2025, given concerns about the adequacy of the CY 2025 MA rate update and the recent trend of providers terminating contracts with MA plans due to excessive prior authorization denial rates and slow payments. The same commenter further detailed that these considerations would steer beneficiaries with greater health needs away from MA and into Medicare FFS. To address changing FFS utilization, the commenter recommended that CMS use more recent data to accurately reflect discharge volumes.
Finally, a commenter commended CMS for increasing the Factor 1 estimate for FY 2025, while another commenter requested that CMS increase the FY 2025 Factor 1 “Update” component consistent with the MedPAC recommended increases to the IPPS market basket used to estimate DSH payments for FY 2022, FY 2024, and FY 2025. This commenter cited MedPAC's March 2023 and March 2024 Reports to Congress, where the Commission recommended a 1.0 percent increase to the FY 2024 market basket percentage and a 1.5 percent increase to the FY 2025 market basket percentage increase.
Response: We thank the commenters for their input. Regarding commenters' request for additional detail on the calculations and assumptions underlying the “Discharges” factor, we refer the commenters to the discussion elsewhere in this section of this final rule and the relevant discussion in the FY 2025 IPPS/LTCH PPS proposed rule ( 86 FR 36190-36192 ), which detail the calculations and assumptions we used to calculate the FY 2025 “Discharges” factor. We also note that in updating our estimate of Factor 1 for this final rule, we considered, as appropriate, the same set of factors that we used in the FY 2024 IPPS/LTCH PPS proposed rule and in prior rulemakings ( see example, ( 88 FR 58993 through 58998 )). As we stated we would do in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36191 ), we then updated our estimates for the FY 2025 “Discharges” component, and other Factor 1 components, to incorporate the latest available data based on more recent economic assumptions and actuarial analyses.
In response to commenters' request that CMS explain why the projection of the “Discharges” component in the FY 2025 IPPS/LTCH PPS proposed rule was lower than the projections for FY 2023 and FY 2024, we point commenters to discussion elsewhere in this section of this final rule and relevant discussion in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36192 ), which detail the calculations and assumptions we used to calculate the FY 2025 “Discharges” factor. We also note that consistent with the policy that we have applied since FY 2014 ( see example, ( 78 FR 50628 through 50630 and 78 FR 61194 )), our estimates for the “Discharges” component in our proposed and final rules are updated using the most recently available data and economic assumptions and actuarial analyses at the time of rulemaking.
Regarding the comments on the impacts of MA enrollment on Medicare FFS discharge volume, we refer commenters to the actuarial projections and assumptions regarding future trends in Medicare FFS and MA program enrollment, utilization, and costs of health care services covered by Medicare, as well as other factors affecting Medicare FFS and MA program expenditures, contained in the “2024 Annual Report of the Boards of Trustees of the Federal Hospital Insurance and Federal Supplementary Medical Insurance Trust Funds,” available under “Downloads” on the CMS website at: https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/ReportsTrustFunds/index.html , which we considered in developing our estimate of the “Discharges” factor for FY 2025. We also note that, consistent with prior years ( see example, ( 88 FR 58997 )) our estimate of the “Discharges” component for FY 2025 in this final rule incorporates only claims from the Medicare FFS program rather than claims from the MA program. Accordingly, we believe that the FY ( print page 69318) 2025 “Discharges” factor in this final rule accurately reflects trends in Medicare FFS discharges.
Regarding the commenter who requested that CMS increase the FY 2025 Factor 1 “Update” component consistent with the MedPAC recommended increases to the IPPS market basket used to estimate DSH payments for FY 2022, FY 2024, and FY 2025, we refer readers to the discussion in section V.B. of the preamble of this final rule. Consistent with the inpatient hospital update discussion in section V.B. of the preamble of this final rule, OACT is using the final inpatient hospital market basket update and productivity adjustment for FY 2025 based on the more recent data available for this final rule for the final FY 2025 “Update” component in the Factor 1 calculation.
After consideration of the public comments we received, we are finalizing, as proposed, the methodology for calculating Factor 1 for FY 2025. We discuss the resulting Factor 1 amount for FY 2025 in this final rule. Consistent with prior rulemakings, for this final rule, OACT used the most recently submitted Medicare cost report data from the March 31, 2024, update of HCRIS to identify Medicare DSH payments and the most recent Medicare DSH payment adjustments provided in the Impact File and applied update factors and assumptions for projected changes in utilization and case-mix to estimate Medicare DSH payments for the upcoming fiscal year.
The June 2024 OACT estimate for Medicare DSH payments for FY 2025, without regard to the application of section 1886(r)(1) of the Act, was approximately $14.013 billion. This estimate excluded Maryland hospitals, which participate in the Maryland Total Cost of Care Model and are not paid under the IPPS, hospitals participating in the Rural Community Hospital Demonstration, and SCHs paid under their hospital-specific payment rate. Therefore, based on this June 2024 estimate, the estimate of empirically justified Medicare DSH payments for FY 2025, with the application of section 1886(r)(1) of the Act, was approximately $3.503 billion (or 25 percent of the total amount of estimated Medicare DSH payments for FY 2025). Under § 412.106(g)(1)(i), Factor 1 is the difference between these two OACT estimates. Therefore, the final Factor 1 for FY 2025 is $10,509,750,000, which is equal to 75 percent of the total amount of estimated Medicare DSH payments for FY 2025 ($14,013,000,000 minus $3,503,250,000).
OACT's estimates for FY 2025 for this final rule began with a baseline of $13.401 billion in Medicare DSH expenditures for FY 2021. The following table shows the factors applied to update this baseline through the current estimate for FY 2025:

In this table, the discharges column shows the changes in the number of Medicare FFS inpatient hospital discharges. The discharge figures for FY 2022 and FY 2023 are based on Medicare claims data that have been adjusted by a completion factor to account for incomplete claims data. We note that these claims data reflect the impact of the COVID-19 pandemic. The discharge figure for FY 2024 is based on preliminary data. The discharge figure for FY 2025 is an assumption based on recent historical experience, an assumed partial return to pre-COVID 19 trends, and assumptions related to how many beneficiaries will be enrolled in MA plans. Accordingly, the discharge figures for FY 2022 to FY 2025 incorporate the actual impact and estimated future impact of the COVID-19 pandemic.
The case-mix column shows the estimated change in case-mix for IPPS hospitals. The case-mix figures for FY 2022 and FY 2023 are based on actual claims data adjusted by a completion factor to account for incomplete claims data. We note that these claims data reflect the impact of the COVID-19 pandemic. The case-mix figures for FY 2024 and for FY 2025 are assumptions based on the 2012 “Review of Assumptions and Methods of the Medicare Trustees' Financial Projections” report by the 2010-2011 Medicare Technical Review Panel. [ 204 ]
The “Other” column reflects the change in other factors that contribute to the Medicare DSH estimates. These factors include the difference between the total inpatient hospital discharges and IPPS discharges and various adjustments to the payment rates that have been included over the years but are not reflected in the other columns (such as the 20 percent add-on for COVID-19 discharges). In addition, the “Other” column includes a factor for the estimated changes in Medicaid enrollment through FY 2023. Based on the most recent available data, Medicaid enrollment is estimated to change as follows: +8.3 percent in FY 2022, +5.2 percent in FY 2023, −11.9 percent in FY 2024, and −5.3 percent in FY 2025. In future IPPS rulemakings, our assumptions regarding Medicaid enrollment may change based on actual enrollment in the States.
We note that, in developing their estimates of the effect of Medicaid expansion on Medicare DSH expenditures, our actuaries have assumed that the new Medicaid enrollees are healthier than the average Medicaid enrollee and, therefore, receive fewer hospital services. [ 205 ] Specifically, based on the most recent ( print page 69319) available data at the time of developing this final rule, OACT assumed per capita spending for Medicaid beneficiaries who enrolled due to the expansion to be approximately 80 percent of the average per capita expenditures for a pre-expansion Medicaid beneficiary, due to the better health of these beneficiaries. The same assumption was used for the new Medicaid beneficiaries who enrolled in 2020 and thereafter due to the COVID-19 pandemic. This assumption is consistent with recent internal estimates of Medicaid per capita spending pre-expansion and post-expansion. In future IPPS rulemakings, the assumption about the average per-capita expenditures of Medicaid beneficiaries who enrolled due to the COVID-19 pandemic may change.
The following table shows the factors that are included in the “Update” column of the previous table:

Section 1886(r)(2)(B) of the Act establishes Factor 2 in the calculation of the uncompensated care payment. Section 1886(r)(2)(B)(ii) of the Act provides that, for FY 2018 and subsequent fiscal years, the second factor is 1 minus the percent change in the percent of individuals who are uninsured, as determined by comparing the percent of individuals who were uninsured in 2013 (as estimated by the Secretary, based on data from the Census Bureau or other sources the Secretary determines appropriate, and certified by the Chief Actuary of CMS) and the percent of individuals who were uninsured in the most recent period for which data are available (as so estimated and certified).
We are continuing to use the methodology that was used in FY 2018 through FY 2024 to determine Factor 2 for FY 2025—to use the National Health Expenditure Accounts (NHEA) data to determine the percent change in the percent of individuals who are uninsured. We refer readers to the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38197 and 38198 ) for a complete discussion of the NHEA and why we determined, and continue to believe, that it is the data source for the rate of uninsurance that, on balance, best meets all our considerations and is consistent with the statutory requirement that the estimate of the rate of uninsurance be based on data from the Census Bureau or other sources the Secretary determines appropriate.
In brief, the NHEA represents the government's official estimates of economic activity (spending) within the health sector. The NHEA includes comprehensive enrollment estimates for total private health insurance (PHI) (including direct and employer-sponsored plans), Medicare, Medicaid, the Children's Health Insurance Program (CHIP), and other public programs, and estimates of the number of individuals who are uninsured. The NHEA data are publicly available on the CMS website at https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/NationalHealthExpendData/index.html .
To compute Factor 2 for FY 2025, the first metric that is needed is the proportion of the total U.S. population that was uninsured in 2013. For a complete discussion of the approach OACT used to prepare the NHEA's estimate of the rate of uninsurance in 2013, including the data sources used, we refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58998 and 58999 ).
The next metrics needed to compute Factor 2 for FY 2025 are projections of the rate of uninsurance in both CY 2024 and CY 2025. On an annual basis, OACT projects enrollment and spending trends for the coming 10-year period. The most recent projections are for 2023 through 2032 and were published on June 12, 2024. Those projections used the latest NHEA historical data that were available at the time of their construction (that is, historical data through 2022). The NHEA projection methodology accounts for expected changes in enrollment across all of the categories of insurance coverage previously listed. For a complete discussion of how the NHEA data account for expected changes in enrollment across all the categories of insurance coverage previously listed, we refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58999 ).
Using these data sources and the previously described methodologies, at the time of developing the proposed rule and using the NHEA data for 2022 through 2031 that were published on June 14, 2023, OACT estimated that the uninsured rate for the historical, baseline year of 2013 was 14 percent, and that the uninsured rates for CYs 2024 and 2025 were 8.5 percent and 8.8 percent, respectively ( 89 FR 36193 ). As required by section 1886(r)(2)(B)(ii) of the Act, the Chief Actuary of CMS certified these estimates. We refer readers to OACT's Memorandum on Certification of Rates of Uninsured prepared for the FY 2025 IPPS/LTCH PPS proposed rule for further details on the methodology and assumptions that were used in the projection of these rates of uninsurance. [ 206 ]
As with the CBO estimates on which we based Factor 2 for fiscal years before FY 2018, the NHEA estimates are for a calendar year. Under the approach originally adopted in the FY 2014 IPPS/LTCH PPS final rule, we have used a weighted average approach to project ( print page 69320) the rate of uninsurance for each fiscal year. We continue to believe that, in order to estimate the rate of uninsurance during a fiscal year accurately, Factor 2 should reflect the estimated rate of uninsurance that hospitals will experience during the fiscal year, rather than the rate of uninsurance during only one of the calendar years that the fiscal year spans. Accordingly, in the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to continue to apply the weighted average approach used in past fiscal years to estimate this final rule's rate of uninsurance for FY 2025.
OACT certified the estimate of the rate of uninsurance for FY 2025 determined using this weighted average approach to be reasonable and appropriate for purposes of section 1886(r)(2)(B)(ii) of the Act. In the proposed rule ( 89 FR 36193 ), we noted that we may also consider the use of more recent data that may become available for purposes of estimating the rates of uninsurance used in the calculation of the final Factor 2 for FY 2025.
In the proposed rule, we outlined the calculation of the proposed Factor 2 for FY 2025 as follows:
- Percent of individuals without insurance for CY 2013: 14 percent.
- Percent of individuals without insurance for CY 2024: 8.5 percent.
- Percent of individuals without insurance for CY 2025: 8.8 percent.
- Percent of individuals without insurance for FY 2025: (0.25 times 0.085) + (0.75 times 0.088) = 8.7 percent.
- Factor 2: 1 − |((0.14−0.087)/0.14)| = 1−0.3786 = 0.6214 (62.14 percent).
We proposed that Factor 2 for FY 2025 would be 62.14 percent.
The proposed FY 2025 uncompensated care amount was equivalent to proposed Factor 1 multiplied by proposed Factor 2, which was $6,498,135,150.00.
We invited public comments on our proposed Factor 2 for FY 2025.
Comment: Most commenters discussed Factor 2 in the context of the impact of the temporary COVID-19 PHE provisions on the uninsured rate, such as the Families First Coronavirus Response Act's Medicaid continuous coverage provision and the American Rescue Plan's Marketplace enhanced premium tax credits. Many large and small healthcare organizations and associations disagreed with CMS' estimates for the FY 2025 uninsured rate and urged OACT to update its estimate of Factor 2 to account for the projected increases in the number of uninsured individuals as Medicaid unwinding continues and Medicaid redeterminations continue to be processed.
A few commenters expressed their concern that the NHEA data source that CMS proposed to use for Factor 2 does not reflect current trends in the uninsured rate as the Medicaid continuous enrollment provisions unwind. Many commenters also indicated that they expect increases in the uninsured rates in their communities. Citing CMS' statement in the proposed rule that the agency could consider more recent data that may become available for the calculation of final Factor 2 for FY 2025, these commenters urged CMS to use more recent and accurate data sources to account for the anticipated increase in the uninsured rate. Considering the expiration of the COVID-19 PHE and the unwinding of the Medicaid continuous enrollment provisions, some of these commenters urged CMS to consider utilizing alternative data sources and calculations to ensure that the Factor 2 estimate accurately reflects the current coverage landscape, including uninsurance rates.
Several commenters referenced data sources and analyses, such as analyses by the Kaiser Family Foundation (KFF) and the Urban Institute, that project that at least 22 million individuals will lose their Medicaid coverage in FY 2024, with the number expected to grow in FY 2025. These commenters stated that they expect at least an additional 5.0 million uninsured individuals for processed redeterminations and an additional 1.7 million for those yet to be processed. Another commenter cited an analysis by the Alliance of Safety-Net Hospitals that indicated that there will be 32.5 million uninsured individuals in FY 2024, yielding an uninsurance rate of 9.6 percent for FY 2024. Accordingly, these commenters requested that CMS increase Factor 2 to reflect the anticipated increase in the uninsured population. A commenter recommended that CMS consider implementing a one-time increase in the percentage used in Factor 2 to account for the lag in data and anticipated rise in the uninsured rate as Medicaid unwinding continues in FY 2025.
Several commenters indicated their support for CMS' proposed increases in FY 2025's Factor 2 and Medicare DSH uncompensated care payments, compared to the FY 2024 Factor 2 and Medicare DSH uncompensated care payments. Some commenters raised concerns regarding the proposed increase in uncompensated care payments for FY 2025, stating that an increase in uncompensated care payments in one year does not make up for underpayments in prior years. In addition, a few commenters asked CMS to increase the uncompensated care amount beyond the amount proposed in the FY 2025 IPPS/LTCH PPS proposed rule, while others urged CMS to increase the uncompensated care amount for community safety-net hospitals in particular given that these hospitals are already financially strained.
A commenter requested that CMS ensure that the assumptions used for the FY 2025 IPPS/LTCH PPS proposed rule's Factor 1 are internally consistent with the assumptions used in the FY 2025 IPPS/LTCH PPS proposed rule's Factor 2. This commenter noted that CMS estimated an 18.2 percentage point decline in Medicaid enrollment between FY 2023 and FY 2025 when calculating Factor 1 but did not account for the same decline in the number of Medicaid beneficiaries when estimating the uninsured rate in Factor 2.
Response: We thank the commenters for their input and diligence regarding the estimate of Factor 2 included in the proposed rule. In response to the comments concerning the NHEA data source used for calculating Factor 2 for FY 2025, we refer readers to the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38197 and 38198 ) for a complete discussion of the NHEA and why we determined, and continue to believe, that it is the data source for the rate of uninsurance that, on balance, best meet all our considerations for ensuring that the data source meets the statutory requirement that the estimate be based on data from the Census Bureau, or other sources the Secretary determines appropriate. We continue to believe that the NHEA will provide reasonable estimates for the rate of uninsurance that are available in conjunction with the IPPS rulemaking cycle.
In the FY 2025 IPPS/LTCH PPS proposed rule, we explained that we used the most recent available estimates from the NHEA at that time, and we refer readers to the relevant discussion in the proposed rule and OACT's Memorandum on Certification of Rates of Uninsured prepared for the proposed rule for further details on the methodology and assumptions used in the proposed rule's calculation of the projected uninsured rate. In brief, we indicated that our projection of the rates of uninsurance for CY 2024 and CY 2025 were from the latest NHEA historical data available and accounted for expected changes in enrollment across all categories of insurance coverage. Using estimates from the NHEA that were publicly available at the time of the proposed rule, OACT ( print page 69321) estimated the legislative impacts and effects of the COVID-19 PHE on insurance coverage when it developed the estimate of rates of uninsurance included in the proposed rule. We note, in particular, that OACT's estimates in the proposed rule considered the COVID-19 PHE provisions and the latest available Medicaid projections publicly available at that time.
In response to commenters who requested that we update the Factor 2 estimates and account for any anticipated changes in the uninsured rate using more recent or alternative data sources, in the proposed rule ( 89 FR 36193 ), we stated we may consider the use of more recent data that may become available for purposes of estimating the rates of uninsurance used in the calculation of the final Factor 2 for FY 2025. In this final rule, we are using the most recent NHEA estimates for the rate of uninsurance, which became available on June 12, 2024, and account for the legislative impacts of the expiration of the Families First Coronavirus Response Act's Medicaid continuous coverage provision, the extension of the American Rescue Plan's Marketplace enhanced premium tax credits via the Inflation Reduction Act, and the effects of the COVID-19 PHE on insurance coverage. Consistent with prior final IPPS/LTCH PPS rulemakings ( see, e.g., the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59000 )), we are using the updated NHEA data for the final Factor 2 calculation because we believe that it is the most appropriate measure of changes in the rate of uninsurance.
Based on these latest projections, we note that the insured share of the population is expected to have been 93.1 percent in CY 2023. In CY 2024, a decrease in Medicaid enrollment on an average monthly basis of 10.2 million enrollees is expected, with an additional decline of 1.6 million enrollees projected in CY 2025. 207 Notably, many individuals who are being disenrolled as a result of Medicaid unwinding are expected to already have comprehensive coverage from another source (such as through an employer). Over 2023-2025, enrollment in direct-purchased insurance, a category of insurance that includes Marketplace qualified health plans, is projected to increase by a total of 8.3 million enrollees largely as a result of the Inflation Reduction Act's temporary extension of enhanced Marketplace subsidies and a temporary Special Enrollment Period for consumers losing Medicaid or Children's Health Insurance coverage due to Medicaid unwinding.
Regarding the commenter who expressed concerns that there may be a discrepancy between assumptions regarding Medicaid enrollment used in FY 2025 IPPS/LTCH PPS proposed rule's Factor 1 and Factor 2, we note that the Medicaid enrollment data used for purposes of uninsured rate projections use the most recent available calendar year data and are generally consistent with the Federal fiscal year data used for purposes of the Factor 1 estimates. [ 208 ]
These changes in enrollment, along with projected trends in other forms of coverage ( e.g., employer-sponsored or direct purchase insurance), are expected to result in an insured share of the population of 92.7 percent in CY 2024 (a decrease from 93.1 percent in CY 2023) and 92.3 percent in CY 2025. We note that the most recent NHEA projections anticipate that the uninsured population will increase from 22.8 million in CY 2023 and 24.4 million in CY 2024 to 26.1 million in CY 2025 and 29.6 million in CY 2026. The projected increase of the uninsured population in CY 2026 is related to the expiration of the enhanced Marketplace subsidies that year. For more detailed projections of health insurance enrollment that underlie the estimation of final Factor 2, we refer readers to NHEA's Table 17 Health Insurance Enrollment and Enrollment Growth Rates. (Available on the CMS website at: https://www.cms.gov/data-research/statistics-trends-and-reports/national-health-expenditure-data/projected )
Regarding the comments requesting that CMS increase the uncompensated care amount for FY 2025, generally or for community safety-net hospitals in particular, we continue to believe that estimating Factor 2 based on the best available data is appropriate and consistent with the requirements of section 1886(r)(2)(B)(ii) of the Act.
Comment: Several commenters urged CMS to be transparent in the calculation of Factor 2 and how it accounts for the expiration of the Medicaid continuous enrollment provisions, while others urged CMS to be transparent regarding the data sources used for calculating Factor 2 and the assumptions behind the uninsured rate. Other commenters requested that CMS publish a detailed methodology on the calculation of the FY2025 proposed rule's Factor 2 and the NHEA projections.
Response: In response to the comments concerning transparency, we note that the accompanying OACT memo contains additional background describing the methods used to derive the FY 2025 rate of uninsured for this final rule. [ 209 ] We also note that section 1886(r)(2)(B)(ii) of the Act permits us to use a data source other than CBO estimates to determine the percent change in the rate of uninsurance beginning in FY 2018. As explained elsewhere in this section of this final rule, the NHEA data and methodology that were used to estimate Factor 2 for this final rule are transparent and best meet all of our considerations for ensuring reasonable estimates for the rate of uninsurance that are available in conjunction with the IPPS/LTCH PPS rulemaking cycle, and we have concluded it is appropriate to update the projection of the FY 2025 rate of uninsurance using the most recent NHEA data. For additional information on the projection of the uninsured, see page 28 of the projection's methodology documentation. (Available on the CMS website at: https://www.cms.gov/research-statistics-data-and-systems/statistics-trends-and-reports/nationalhealthexpenddata/downloads/projectionsmethodology.pdf ).
After consideration of the public comments we received, we are updating the calculation of Factor 2 for FY 2025 to incorporate more recent data from NHEA. The final estimates of the percent of uninsured individuals have been certified by the Chief Actuary of CMS. We note that the CY 2024 and CY 2025 uninsurance rates are projected to be higher than CY 2023's partly because of the expiration of the Medicaid continuous enrollment provisions and the projected declines in Medicaid enrollment in CY 2024 and CY 2025, which are also larger in the final rule than in the proposed rule. However, the lower projected rates of uninsurance in CY 2024 and CY 2025 in the final rule relative to the proposed rule largely reflect higher expected enrollment in direct-purchase insurance in those years. This higher expected enrollment is associated with enrollment in Marketplace plans and is related to (i) the Inflation Reduction Act's extension of the American Rescue Plan Act's enhanced Marketplace premium subsidies through 2025 and (ii) a ( print page 69322) Special Enrollment Period open to those who are no longer eligible for Medicaid coverage due to state-based redeterminations.
The calculation of the final Factor 2 for FY 2025 using a weighted average of OACT's updated projections for CY 2024 and CY 2025 is as follows:
- Percent of individuals without insurance for CY 2013: 14.0 percent.
- Percent of individuals without insurance for CY 2024: 7.3 percent.
- Percent of individuals without insurance for CY 2025: 7.7 percent,
- Percent of individuals without insurance for FY 2025: (0.25 times 0.073) + (0.75 times 0.077) = 7.6 percent.
- Factor 2: 1−|((0.076−0.14)/0.14)| = 1−0.457 = 0.5429 (54.29 percent).
Therefore, the final Factor 2 for FY 2025 is 54.29 percent. The final FY 2025 uncompensated care amount is $10,509,750, 000 * 0.5429 = $5,705,743,275.
| Final FY 2025 Uncompensated Care Amount | $5,705,743,275 |
Section 1886(r)(2)(C) of the Act defines Factor 3 in the calculation of the uncompensated care payment. As we have discussed earlier, section 1886(r)(2)(C) of the Act states that Factor 3 is equal to the percent, for each subsection (d) hospital, that represents the quotient of: (1) the amount of uncompensated care for such hospital for a period selected by the Secretary (as estimated by the Secretary, based on appropriate data (including, in the case where the Secretary determines alternative data are available that are a better proxy for the costs of subsection (d) hospitals for treating the uninsured, the use of such alternative data)); and (2) the aggregate amount of uncompensated care for all subsection (d) hospitals that receive a payment under section 1886(r) of the Act for such period (as so estimated, based on such data).
Therefore, Factor 3 is a hospital-specific value that expresses the proportion of the estimated uncompensated care amount for each subsection (d) hospital and each subsection (d) Puerto Rico hospital with the potential to receive Medicare DSH payments relative to the estimated uncompensated care amount for all hospitals estimated to receive Medicare DSH payments in the fiscal year for which the uncompensated care payment is to be made. Factor 3 is applied to the product of Factor 1 and Factor 2 to determine the amount of the uncompensated care payment that each eligible hospital will receive for FY 2014 and subsequent fiscal years. In order to implement the statutory requirements for this factor of the uncompensated care payment formula, it was necessary for us to determine: (1) the definition of uncompensated care or, in other words, the specific items that are to be included in the numerator (that is, the estimated uncompensated care amount for an individual hospital) and the denominator (that is, the estimated uncompensated care amount for all hospitals estimated to receive Medicare DSH payments in the applicable fiscal year); (2) the data source(s) for the estimated uncompensated care amount; and (3) the timing and manner of computing the quotient for each hospital estimated to receive Medicare DSH payments. The statute instructs the Secretary to estimate the amounts of uncompensated care for a period based on appropriate data. In addition, we note that the statute permits the Secretary to use alternative data in the case where the Secretary determines that such alternative data are available that are a better proxy for the costs of subsection (d) hospitals for treating individuals who are uninsured. For a discussion of the methodology, we used to calculate Factor 3 for fiscal years 2014 through 2022, we refer readers to the FY 2024 IPPS/LTCH final rule ( 88 FR 59001 and 59002 ).
Section 1886(r)(2)(C) of the Act governs the selection of the data to be used in calculating Factor 3 and allows the Secretary the discretion to determine the time periods from which we will derive the data to estimate the numerator and the denominator of the Factor 3 quotient. Specifically, section 1886(r)(2)(C)(i) of the Act defines the numerator of the quotient as the amount of uncompensated care for a subsection (d) hospital for a period selected by the Secretary. Section 1886(r)(2)(C)(ii) of the Act defines the denominator as the aggregate amount of uncompensated care for all subsection (d) hospitals that receive a payment under section 1886(r) of the Act for such period. In the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50634 through 50647 ), we adopted a process of making interim payments with final cost report settlement for both the empirically justified Medicare DSH payments and the uncompensated care payments required by section 3133 of the Affordable Care Act. Consistent with that process, we also determined the time period from which to calculate the numerator and denominator of the Factor 3 quotient in a way that would be consistent with making interim and final payments. Specifically, we must have Factor 3 values available for hospitals that we estimate will qualify for Medicare DSH payments for a fiscal year and for those hospitals that we do not estimate will qualify for Medicare DSH payments for that fiscal year but that may ultimately qualify for Medicare DSH payments for that fiscal year at the time of cost report settlement.
As described in the FY 2022 IPPS/LTCH PPS final rule, commenters expressed concerns that the use of only 1 year of data to determine Factor 3 would lead to significant variations in year-to-year uncompensated care payments. Some stakeholders recommended the use of 2 years of historical data from Worksheet S-10 data of the Medicare cost report ( 86 FR 45237 ). In the FY 2022 IPPS/LTCH PPS final rule, we stated that we would consider using multiple years of data when the vast majority of providers had been audited for more than 1 fiscal year under the revised reporting instructions. Audited FY 2019 cost reports were available for the development of the FY 2023 IPPS/LTCH PPS proposed and final rules. Feedback from previous audits and lessons learned were incorporated into the audit process for the FY 2019 reports.
In consideration of the comments discussed in the FY 2022 IPPS/LTCH PPS final rule, in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49036 through 49047 ), we finalized a policy of using a multi-year average of audited Worksheet S-10 data to determine Factor 3 for FY 2023 and subsequent fiscal years. We explained our belief that this approach would be generally consistent with our past practice of using the most recent single year of audited data from the Worksheet S-10, while also addressing commenters' concerns regarding year-to-year fluctuations in uncompensated care payments. Under this policy, we used a 2-year average of audited FY 2018 and FY 2019 Worksheet S-10 data to ( print page 69323) calculate Factor 3 for FY 2023. We also indicated that we expected FY 2024 would be the first year that 3 years of audited data would be available at the time of rulemaking. For FY 2024 and subsequent fiscal years, we finalized a policy of using a 3-year average of the uncompensated care data from the 3 most recent fiscal years for which audited data are available to determine Factor 3. Consistent with the approach that we followed when multiple years of data were previously used in the Factor 3 methodology, if a hospital does not have data for all 3 years used in the Factor 3 calculation, we will determine Factor 3 based on an average of the hospital's available data. For IHS and Tribal hospitals and Puerto Rico hospitals, we use the same multi-year average of Worksheet S-10 data to determine Factor 3 for FY 2024 and subsequent fiscal years as is used to determine Factor 3 for all other DSH-eligible hospitals (in other words, hospitals eligible to receive empirically justified Medicare DSH payments for a fiscal year) to determine Factor 3.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49033 through 49047 ), we also modified our policy regarding cost reports that start in one fiscal year and span the entirety of the following fiscal year. Specifically, in the rare cases when we use a cost report that starts in one fiscal year and spans the entirety of the subsequent fiscal year to determine uncompensated care costs for the subsequent fiscal year, we would not use the same cost report to determine the hospital's uncompensated care costs for the earlier fiscal year. We explained that using the same cost report to determine uncompensated care costs for both fiscal years would not be consistent with our intent to smooth year-to-year variation in uncompensated care costs. As an alternative, we finalized our proposal to use the hospital's most recent prior cost report, if that cost report spans the applicable period. [ 210 ]
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59003 ), we continued the policy finalized in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49042 ) to address the effects of calculating Factor 3 using data from multiple fiscal years, in which we apply a scaling factor to the Factor 3 values calculated for all DSH-eligible hospitals so that total uncompensated care payments to hospitals that are projected to be DSH-eligible for a fiscal year will be consistent with the estimated amount available to make uncompensated care payments for that fiscal year. Pursuant to that policy, we divide 1 (the expected sum of all DSH-eligible hospitals' Factor 3 values) by the actual sum of all DSH-eligible hospitals' Factor 3 values and then multiply the quotient by the uncompensated care payment determined for each DSH-eligible hospital to obtain a scaled uncompensated care payment amount for each hospital. This process is designed to ensure that the sum of the scaled uncompensated care payments for all hospitals that are projected to be DSH-eligible is consistent with the estimate of the total amount available to make uncompensated care payments for the applicable fiscal year.
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59003 ), we continued our new hospital policy that was modified in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49042 ) and initially adopted in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42370 through 42371 ) to determine Factor 3 for new hospitals. Consistent with our policy of using multiple years of cost reports to determine Factor 3, we defined new hospitals as hospitals that do not have cost report data for the most recent year of data being used in the Factor 3 calculation. Under this definition, the cut-off date for the new hospital policy is the beginning of the fiscal year after the most recent year for which audits of the Worksheet S-10 data have been conducted. For FY 2024, the FY 2020 cost reports were the most recent year of cost reports for which audits of Worksheet S-10 data had been conducted. Thus, hospitals with CMS Certification Numbers (CCNs) established on or after October 1, 2020, were subject to the new hospital policy for FY 2024.
Under our modified new hospital policy, if a new hospital has a preliminary projection of being DSH-eligible based on its most recent available disproportionate patient percentage, it may receive interim empirically justified DSH payments. However, new hospitals will not receive interim uncompensated care payments because we would have no uncompensated care data on which to determine what those interim payments should be. The MAC will make a final determination concerning whether the hospital is eligible to receive Medicare DSH payments at cost report settlement. In FY 2024, while we continued to determine the numerator of the Factor 3 calculation using the new hospital's uncompensated care costs reported on Worksheet S-10 of the hospital's cost report for the current fiscal year, we determined Factor 3 for new hospitals using a denominator based solely on uncompensated care costs from cost reports for the most recent fiscal year for which audits have been conducted. In addition, we applied a scaling factor to the Factor 3 calculation for a new hospital. [ 211 ]
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59004 ), we continued our policy of treating hospitals that merge after the development of the final rule for the applicable fiscal year similar to new hospitals. As explained in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50021 ), for these newly merged hospitals, we do not have data currently available to calculate a Factor 3 amount that accounts for the merged hospital's uncompensated care burden. In the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50021 and 50022 ), we finalized a policy under which Factor 3 for hospitals that we do not identify as undergoing a merger until after the public comment period and additional review period following the publication of the final rule or that undergo a merger during the fiscal year will be recalculated similar to new hospitals.
Consistent with the policy adopted in the FY 2015 IPPS/LTCH PPS final rule, in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59004 ), we stated that we would continue to treat newly merged hospitals in a similar manner to new hospitals, such that the newly merged hospital's final uncompensated care payment will be determined at cost report settlement where the numerator of the newly merged hospital's Factor 3 will be based on the cost report of only the surviving hospital (that is, the newly merged hospital's cost report) for the current fiscal year. However, if the hospital's cost reporting period includes less than 12 months of data, the data from the newly merged hospital's cost report will be annualized for purposes of the Factor 3 calculation. Consistent ( print page 69324) with the methodology used to determine Factor 3 for new hospitals described in section IV.E.3. of the preamble of this final rule, we continued our policy for determining Factor 3 for newly merged hospitals using a denominator that is the sum of the uncompensated care costs for all DSH-eligible hospitals, as reported on Worksheet S-10 of their cost reports for the most recent fiscal year for which audits have been conducted. In addition, we apply a scaling factor, as discussed in section IV.E.3. of the preamble of this final rule, to the Factor 3 calculation for a newly merged hospital. In the FY 2024 IPPS/LTCH PPS final rule, we explained that consistent with past policy, interim uncompensated care payments for the newly merged hospital would be based only on the data for the surviving hospital's CCN available at the time of the development of the final rule.
The calculation of a hospital's total uncompensated care costs on Worksheet S-10 requires the use of the hospital's cost to charge ratio (CCR). In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59004 through 59005 ), we continued the policy of trimming CCRs, which we adopted in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49043 ), for FY 2024. Under this policy, we apply the following steps to determine the applicable CCR separately for each fiscal year that is included as part of the multi-year average used to determine Factor 3:
Step 1: Remove Maryland hospitals. In addition, we will remove all-inclusive rate providers because their CCRs are not comparable to the CCRs calculated for other IPPS hospitals.
Step 2: Calculate a CCR “ceiling” for the applicable fiscal year with the following data: for each IPPS hospital that was not removed in Step 1 (including hospitals that are not DSH-eligible), we use cost report data to calculate a CCR by dividing the total costs on Worksheet C, Part I, Line 202, Column 3 by the charges reported on Worksheet C, Part I, Line 202, Column 8. (Combining data from multiple cost reports from the same fiscal year is not necessary, as the longer cost report will be selected.) The ceiling is calculated as 3 standard deviations above the national geometric mean CCR for the applicable fiscal year. This approach is consistent with the methodology for calculating the CCR ceiling used for high-cost outliers. Remove all hospitals that exceed the ceiling so that these aberrant CCRs do not skew the calculation of the statewide average CCR.
Step 3: Using the CCRs for the remaining hospitals in Step 2, determine the urban and rural statewide average CCRs for the applicable fiscal year for hospitals within each State (including hospitals that are not DSH-eligible), weighted by the sum of total hospital discharges from Worksheet S-3, Part I, Line 14, Column 15.
Step 4: Assign the appropriate statewide average CCR (urban or rural) calculated in Step 3 to all hospitals, excluding all-inclusive rate providers, with a CCR for the applicable fiscal year greater than 3 standard deviations above the national geometric mean for that fiscal year (that is, the CCR “ceiling”).
Step 5: For hospitals that did not report a CCR on Worksheet S-10, Line 1, we assign them the statewide average CCR for the applicable fiscal year as determined in step 3.
After completing these steps, we re-calculate the hospital's uncompensated care costs (Line 30) for the applicable fiscal year using the trimmed CCR (the statewide average CCR (urban or rural, as applicable)).
After applying the CCR trim methodology, there are rare situations where a hospital has potentially aberrant uncompensated care data for a fiscal year that are unrelated to its CCR. Therefore, under the trim methodology for potentially aberrant uncompensated care costs (UCC) that was included as part of the methodology for purposes of determining Factor 3 in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58832 ), if the hospital's uncompensated care costs for any fiscal year that is included as a part of the multi-year average are an extremely high ratio (greater than 50 percent) of its total operating costs in the applicable fiscal year, we will determine the ratio of uncompensated care costs to the hospital's total operating costs from another available cost report, and apply that ratio to the total operating expenses for the potentially aberrant fiscal year to determine an adjusted amount of uncompensated care costs for the applicable fiscal year. [ 212 ]
However, we note that we have audited the Worksheet S-10 data that will be used in the Factor 3 calculation for a number of hospitals. Because the UCC data for these hospitals have been subject to audit, we believe that there is increased confidence that if high uncompensated care costs are reported by these audited hospitals, the information is accurate. Therefore, as we explained in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58832 ), we determined it is unnecessary to apply the UCC trim methodology for a fiscal year for which a hospital's UCC data have been audited.
In rare cases, hospitals that are not currently projected to be DSH-eligible and that do not have audited Worksheet S-10 data may have a potentially aberrant amount of insured patients' charity care costs (line 23 column 2). In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59004 ), we stated that in addition to the UCC trim methodology, we will continue to apply an alternative trim specific to certain hospitals that do not have audited Worksheet S-10 data for one or more of the fiscal years that are used in the Factor 3 calculation. For FY 2023 and subsequent fiscal years, in the rare case that a hospital's insured patients' charity care costs for a fiscal year are greater than $7 million and the ratio of the hospital's cost of insured patient charity care (line 23 column 2) to total uncompensated care costs (line 30) is greater than 60 percent, we will not calculate a Factor 3 for the hospital at the time of proposed or final rulemaking. This trim will only impact hospitals that are not currently projected to be DSH-eligible; and therefore, are not part of the calculation of the denominator of Factor 3, which includes only uncompensated care costs for hospitals projected to be DSH-eligible. Consistent with the approach adopted in the FY 2022 IPPS/LTCH PPS final rule, if a hospital would be trimmed under both the UCC trim methodology and this alternative trim, we will apply this trim in place of the existing UCC trim methodology. We continue to believe this alternative trim more appropriately addresses potentially aberrant insured patient charity care costs compared to the UCC trim methodology, because the UCC trim is based solely on the ratio of total uncompensated care costs to total operating costs and does not consider the level of insured patients' charity care costs.
Similar to the approach initially adopted in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45245 and 45246 ), in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59005 ), we also stated that we would continue to use a threshold of 3 standard deviations from the mean ratio of insured patients' charity care costs to total uncompensated care costs (line 23 column 2 divided by line 30) and a dollar threshold that is the median total uncompensated care cost reported on ( print page 69325) most recent audited cost reports for hospitals that are projected to be DSH-eligible. We stated that we continued to believe these thresholds are appropriate to address potentially aberrant data. We also continued to include Worksheet S-10 data from IHS/Tribal hospitals and Puerto Rico hospitals consistent with our policy finalized in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49047 through 49051 ). In addition, we continued our policy adopted in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49044 ) of applying the same threshold amounts originally calculated for the FY 2018 reports to identify potentially aberrant data for FY 2024 and subsequent fiscal years to facilitate transparency and predictability. If a hospital subject to this trim is determined to be DSH-eligible at cost report settlement, the MAC will calculate the hospital's Factor 3 using the same methodology used to calculate Factor 3 for new hospitals.
For FY 2025, consistent with § 412.106(g)(1)(iii)(C)( 11 ), we are following the same methodology as applied in FY 2024 and described in the previous section of this final rule to determine Factor 3 using the most recent 3 years of audited cost reports, from FY 2019, FY 2020, and FY 2021. Consistent with our approach for FY 2024, for FY 2025, we are also applying the scaling factor, new hospital, newly merged hospital, CCR trim methodology, UCC trim, and alternative trim methodology policies discussed in the previous section of this final rule. For purposes of the FY 2025 IPPS/LTCH PPS proposed rule, we used reports from the December 2023 HCRIS extract to calculate Factor 3. In the proposed rule, we noted that we intended to use the March 2024 update of HCRIS to calculate the final Factor 3 for the FY 2025 IPPS/LTCH PPS final rule.
Thus, for FY 2025, we will use 3 years of audited Worksheet S-10 data to calculate Factor 3 for all eligible hospitals, including IHS and Tribal hospitals and Puerto Rico hospitals that have a cost report for 2013, following these steps:
Step 1: Select the hospital's longest cost report for each of the most recent 3 years of fiscal year (FY) audited cost reports (FY 2019, FY 2020, and FY 2021). Alternatively, in the rare case when the hospital has no cost report for a particular year because the cost report for the previous fiscal year spanned the more recent fiscal year, the previous fiscal year cost report will be used in this step. In the rare case that using a previous fiscal year cost report results in a period without a report, we would use the prior year report, if that cost report spanned the applicable period. [ 213 ] In general, we note that, for purposes of the Factor 3 methodology, references to a fiscal year cost report are to the cost report that spans the relevant fiscal year.
Step 2: Annualize the UCC from Worksheet S-10 Line 30, if a cost report is more than or less than 12 months. (If applicable, use the statewide average CCR (urban or rural) to calculate uncompensated care costs.)
Step 3: Combine adjusted and/or annualized uncompensated care costs for hospitals that merged using the merger policy.
Step 4: Calculate Factor 3 for all DSH-eligible hospitals using annualized uncompensated care costs (Worksheet S-10 Line 30) based on cost report data from the most recent 3 years of audited cost reports (from Step 1, 2 or 3). New hospitals and other hospitals that are treated as if they are new hospitals for purposes of Factor 3 are excluded from this calculation.
Step 5: Average the Factor 3 values from Step 4; that is, add the Factor 3 values, and divide that amount by the number of cost reporting periods with data to compute an average Factor 3 for the hospital. Multiply by a scaling factor, as discussed in the previous section of this final rule.
We received comments regarding the definition of uncompensated care costs for purposes of the Factor 3 calculation, Worksheet S-10 cost report audits, the newly merged hospitals policy, and our Factor 3 calculation instructions.
Comment: Several commenters expressed their support for CMS' proposal to calculate Factor 3 for FY 2025 based on a three-year average of audited FY 2019, FY 2020, and FY 2021 Worksheet S-10 data and to use a three-year average of uncompensated care data from the 3 most recent fiscal years for which audited data are available to determine Factor 3 in subsequent fiscal years. Commenters specified that the use of a multi-year average of Worksheet S-10 data minimizes year-to-year volatility in uncompensated care payments. For example, commenters mentioned that use of a three-year average will smooth out significant fluctuations in the data across the COVID-19 PHE years. A commenter noted their long-standing support for using audited Worksheet S-10 data to calculate Factor 3, which they stated promotes an accurate and consistent calculation of uncompensated care costs.
Response: We are grateful to those commenters who expressed their support for our methodology of using a three-year average of audited FY 2019, FY 2020, and FY 2021 Worksheet S-10 data to calculate Factor 3 for FY 2025. As explained in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36194 , we believe that using a multi-year average of Worksheet S-10 data will help provide assurance that hospitals' uncompensated care payments remain stable and are not subject to unpredictable swings and anomalies in a hospital's uncompensated care costs.
Comment: A commenter suggested alternative approaches to the uncompensated care payment calculation outside of the scope of methodological concepts concerning the blending of historical Worksheet S-10 data. The commenter recommended that CMS monitor changes in uncompensated care reported during the COVID-19 PHE to ensure Worksheet S-10 data accuracy and avoid large redistributions of Medicare DSH funding away from essential hospitals.
Response: With regard to the commenter's suggestions unrelated to the previously discussed methodological concepts for the blending of historical Worksheet S-10 data, we consider these public comments to be outside the scope of the proposed rule, and we are not addressing them in this final rule. However, we appreciate the commenter's input and note that we may address it and other considerations in future rulemaking.
Comment: A few commenters suggested approaches to mitigate the impact of the COVID-19 PHE on the three-year average of Worksheet S-10 data. A few commenters recommended that CMS exclude FY 2020 data entirely from FY 2025 DSH calculations and instead use FY 2019, FY 2021, and FY 2022 data, as FY 2020 data is flawed due to the impacts of the COVID-19 PHE. The same commenters stated that FY 2020 data should be excluded from FY 2025 DSH calculations because it was excluded from most quality reporting metrics. A commenter encouraged CMS to regularly assess and identify any unusual trends in the Worksheet S-10 data. Another commenter expressed concern about the use of FY 2021 and FY 2022 data to ( print page 69326) calculate Factor 3 and requested that CMS lessen the effect of any large reductions in uncompensated care costs due to the COVID-19 PHE. The same commenter suggested that CMS ensure that its use of FY 2020 and FY 2021 Worksheet S-10 data for purposes of determining Factor 3 for FY 2025 does not reduce Factor 3 amounts for essential health systems. One commenter requested that CMS refine its methodology to calculate Factor 3 to account for changes in uncompensated care costs and recommended that CMS mitigate the effect of anomalies in the cost report data for the COVID-19 PHE period.
Response: Regarding requests for CMS to mitigate the impact of the COVID-19 PHE on the three-year average of Worksheet S-10 cost report data, we note that we will continue to use the three-year average of the most recently audited cost report data to determine Factor 3 for FY 2025 and subsequent years, consistent with the policy finalized in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49038 ) and § 412.106(g)(1)(iii)(C)( 11 ). In response to the comments requesting that we exclude FY 2020 data, we continue to believe that using the three-year average will smooth the variation in year-to-year uncompensated care payments and lessen the impacts of the COVID-19 PHE and future unforeseen events. We also note that the calculations for Factor 1 and Factor 2 for FY 2025 reflect the estimated impact of the COVID-19 PHE on DSH payments. Further, we anticipate that there will be less fluctuation in cost report data as the PHE disruptions on healthcare utilization stabilize. In response to the commenters who encouraged CMS to regularly assess and identify any unusual trends in the Worksheet S-10 data and recommended that CMS mitigate the effect of anomalies in the cost report data for the COVID-19 PHE period, we note that the audit process for Worksheet S-10 cost reports will continue to be an important part of identifying potential irregularities in the data. We will continue to monitor the impacts of the PHE and will consider this issue further in future rulemaking, as appropriate.
Comment: A commenter recommended changes to the definition of uncompensated care costs and requested that CMS ensure its Factor 3 calculation methodology accurately captures the full range of uncompensated care costs that hospitals incur while providing care for disadvantaged patients. This commenter urged CMS to include all patient care costs in the cost-to-charge ratio (CCR), including teaching costs and costs for providing physician and other professional services, to ensure an accurate distribution of uncompensated care payments to hospitals with the highest levels of uncompensated care. This commenter stated that excluding Graduate Medical Education (GME) costs when calculating the CCR disproportionately impacts teaching hospitals. This commenter further suggested that CMS treat the unreimbursed portion of state or local indigent care as charity care. Finally, the commenter suggested that CMS revise the Worksheet S-10 data collected on Medicaid shortfalls to better capture actual shortfalls incurred by hospitals by allowing hospitals to deduct intergovernmental transfers (IGTs), certified public expenditures (CPEs), and provider taxes from their Medicaid revenue.
Response: We appreciate the commenter's suggestions for revisions and/or modifications to Worksheet S-10. We will consider modifications as necessary to further improve and refine the information that is reported on Worksheet S-10 to support collection of the information regarding uncompensated care costs.
Regarding the request to include costs for teaching and providing physician and other professional services, including GME costs, when calculating the CCR, we note that because the CCR on Line 1 of Worksheet S-10 is obtained from Worksheet C, Part I, and is also used in other IPPS rate setting contexts (such as high-cost outliers and the calculation of the MS-DRG relative weights) from which it is appropriate to exclude the costs associated with physician and professional services and GME costs, we remain reluctant to adjust CCRs in the narrower context of calculating uncompensated care costs. Therefore, as stated in past final rules, including the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45241 and 45242 ) and the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49039 ), we continue to believe that it is not appropriate to modify the calculation of the CCR on Line 1 of Worksheet S-10 to include any additional costs in the numerator of the CCR calculation.
With regard to the comments requesting that payment shortfalls from Medicaid and state and local indigent care programs be included in uncompensated care cost calculations, we have consistently explained in past final rules ( see, e.g., the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58826 ), the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45238 ), and the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49039 )), in response to similar comments that we believe there are compelling arguments for excluding such shortfalls from the definition of uncompensated care. We refer readers to those prior rules for further discussion.
Comment: A commenter expressed concern that insufficient Medicare DSH uncompensated care payments threaten to hamper CMS' focus on health equity efforts across certain programs, stating their belief that failing to keep pace with the need for uncompensated care resources affects safety-net hospitals that serve a disproportionate share of patients who experience inequitable health outcomes.
Response: We thank the commenter for their concern regarding the impact of the distribution of uncompensated care payments on health equity efforts generally, and on safety-net hospitals, in particular. We may consider this issue in future rulemaking, as appropriate.
Comment: Several commenters reiterated support for using audited Worksheet S-10 data to promote accuracy and consistency. They stated that use of audited Worksheet S-10 data results in uncompensated care data that is most appropriate for use in calculating uncompensated care payments. A commenter encouraged CMS to continue auditing Worksheet S-10 data to ensure the most accurate information is used to calculate Factor 3. Another commenter commended CMS' revisions to the Worksheet S-10 audit protocols, stating that recent audits have been less resource intensive for hospitals compared to prior audit cycles, and that the adjustments after review were largely as expected or as requested.
Other commenters proposed changes to the Worksheet S-10 audit process. For example, a commenter requested that CMS disseminate a comprehensive audit policy and protocols that must be employed by all auditors and MACs and disclose these through notice and comment rulemaking. The same commenter reiterated a previous request made in prior comments that CMS implement a workable appeal or review process to correct errors and inconsistent audit disallowances in a timely manner. A commenter encouraged CMS to work with auditors to streamline the audit process and improve consistency. Another commenter requested that CMS make audit protocols publicly available and ensure that Worksheet S-10 audits impose minimal burden and are equitable and uniform across hospitals.
Response: We thank commenters for their feedback on the audits of the FY 2021 Worksheet S-10 data and their ( print page 69327) recommendations for future audits, as well as their support for the changes CMS has made to the Worksheet S-10 audit protocols. As we have stated in previous rulemakings in response to comments regarding audit protocols ( see, e.g., the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59008 )), the audit protocols are provided to the MACs in advance of the audit to ensure consistency and timeliness in the audit process. CMS began auditing the FY 2021 Worksheet S-10 data for selected hospitals last year so that the audited uncompensated care data for these hospitals would be available in time for use in the FY 2025 IPPS/LTCH PPS proposed rule. We chose to focus the audit on the FY 2021 cost reports in order to maximize the available audit resources. We also note that FY 2021 data are the most recent year of audited data under the revised cost report instructions that became effective on October 1, 2018.
We appreciate all commenters' input and recommendations on how to improve our audit process and reiterate our commitment to continue working with MACs and providers on audit improvements, which include making changes to increase the efficiency of the audit process, building on the lessons learned in previous audit years. We will take commenters' recommendations into consideration for future rulemaking.
Regarding the request to make public the audit policies and protocols, as we previously explained in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59008 ), the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58822 ), the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42368 ), and the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56964 ) we do not make our protocols public as CMS desk review and audit protocols are confidential and are for CMS and MAC use only. In addition, there is no requirement under either the Administrative Procedure Act or the Medicare statute that CMS adopt audit policies or protocols through notice and comment rulemaking. As previously discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59008 ) and the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58822 ), to most efficiently and appropriately utilize our limited audit resources, we do not plan on introducing an audit appeal process at this time.
Comment: A commenter requested that CMS clarify the instructions for line 29 of Worksheet S-10 so that non-Medicare bad debt is not multiplied by the CCR. This commenter stated that while CMS' revised cost report instructions indicate that non-reimbursed Medicare bad debt is not multiplied by the CCR, CMS' September 2017 transmittal [ 214 ] states that non-Medicare bad debt should be multiplied by the CCR.
Response: We appreciate the commenter's concerns regarding the need for clarification of the Worksheet S-10 instructions. We reiterate our commitment to continuing to work with impacted parties to address their concerns regarding the Worksheet S-10 instructions through provider education and further refinement of the instructions, as appropriate. We also encourage providers to share with their respective MAC any questions they have regarding Worksheet S-10 instructions, reporting, and submission deadlines.
We continue to believe our efforts to refine the Worksheet S-10 instructions and related guidance have improved provider understanding of Worksheet S-10 and made the instructions clearer. We also recognize that there are continuing opportunities to further improve the accuracy and consistency of the information that is reported on the Worksheet S-10, and to the extent that commenters have raised new questions and concerns regarding the reporting requirements, we will attempt to address them through future rulemaking and/or sub-regulatory guidance and subsequent outreach [to MACs and providers]. However, as stated in previous IPPS/LTCH PPS rulemakings ( see, e.g., the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59008 and 59009 )), we continue to believe that the Worksheet S-10 instructions are sufficiently clear and allow hospitals to accurately complete Worksheet S-10.
Regarding the commenter's request that CMS clarify whether non-Medicare bad debt is multiplied by CCR, we believe that the Worksheet S-10 instructions are clear and indicate that the CCR will not be applied to the deductible and coinsurance amounts for insured patients approved for charity care and non-reimbursed Medicare bad debt.
For purposes of identifying new hospitals, for FY 2025, the FY 2021 cost reports are the most recent year of cost reports for which audits of Worksheet S-10 data have been conducted. Thus, hospitals with CCNs established on or after October 1, 2021, will be subject to the new hospital policy in FY 2025. If a new hospital is ultimately determined to be eligible for Medicare DSH payments for FY 2025, the hospital will receive an uncompensated care payment calculated using a Factor 3 where the numerator is the uncompensated care costs reported on Worksheet S-10 of the hospital's FY 2025 cost report, and the denominator is the sum of the uncompensated care costs reported on Worksheet S-10 of the FY 2021 cost reports for all DSH-eligible hospitals. In addition, we will apply a scaling factor, as discussed previously, to the Factor 3 calculation for a new hospital. As we explained in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59004 ), we believe applying the scaling factor is appropriate for purposes of calculating Factor 3 for all hospitals, including new hospitals and hospitals that are treated as new hospitals, to improve consistency and predictability across all hospitals.
For FY 2025, the eligibility of a newly merged hospital to receive interim uncompensated care payments will be based on whether the surviving CCN has a preliminary projection of being DSH-eligible, and the amount of any interim uncompensated care payments will be based on the uncompensated care costs from the FY 2019, FY 2020, and FY 2021 cost reports available for the surviving CCN at the time the final rule is developed. However, at cost report settlement, we will determine the newly merged hospital's final uncompensated care payment based on the uncompensated care costs reported on its FY 2025 cost report. That is, we will revise the numerator of Factor 3 for the newly merged hospital to reflect the uncompensated care costs reported on the newly merged hospital's FY 2025 cost report. The denominator will be the sum of the uncompensated care costs reported on Worksheet S-10 of the FY 2021 cost reports for all DSH-eligible hospitals, which is the most recent fiscal year for which audits have been conducted. We will also apply a scaling factor, as described previously.
Comment: A few commenters expressed support for the uncompensated care payment policies currently in place for newly merged hospitals—specifically, the policy stating that final uncompensated care payments for these hospitals will be determined during cost report settlement based on the surviving hospital's cost report for the applicable fiscal year. These commenters also indicated support for our policy whereby MACs make the final determination concerning whether new ( print page 69328) hospitals are eligible to receive DSH payments at cost report settlement based on the new hospital's cost report for the respective fiscal year.
Response: We appreciate the continued support for our policies for new and newly merged hospitals.
For a hospital that is subject to either of the trims for potentially aberrant data (the UCC trim and alternative trim methodology explained in the previous section of this final rule) and is ultimately determined to be DSH-eligible at cost report settlement, its uncompensated care payment will be calculated only after the hospital's reporting of insured charity care costs on its FY 2025 Worksheet S-10 has been reviewed. Accordingly, the MAC will calculate a Factor 3 for the hospital only after reviewing the uncompensated care information reported on Worksheet S-10 of the hospital's FY 2025 cost report. Then we will calculate Factor 3 for the hospital using the same methodology used to determine Factor 3 for new hospitals. Specifically, the numerator will reflect the uncompensated care costs reported on the hospital's FY 2025 cost report, while the denominator will reflect the sum of the uncompensated care costs reported on Worksheet S-10 of the FY 2021 cost reports of all DSH-eligible hospitals. In addition, we will apply a scaling factor, as discussed previously, to the Factor 3 calculation for the hospital.
We did not receive any comments on the discussion of the CCR trim methodology, the UCC trim methodology, or the alternative trim methodology.
Under the CCR trim methodology, for purposes of this final rule, the statewide average CCR was applied to 10 hospitals' FY 2019 reports, of which 4 hospitals had FY 2019 Worksheet S-10 data. The statewide average CCR was applied to 8 hospitals' FY 2020 reports, of which 3 hospitals had FY 2020 Worksheet S-10 data. The statewide average CCR was applied to 9 hospitals' FY 2021 reports, of which 4 hospitals had FY 2021 Worksheet S-10 data.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36197 ), we stated that for purposes of this FY 2025 IPPS/LTCH PPS final rule, consistent with our Factor 3 methodology since the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50642 ), we intended to use data from the March 2024 HCRIS extract for this calculation. We explained that the March 2024 HCRIS extract would be the latest quarterly HCRIS extract that would be publicly available at the time of the development of the FY 2025 IPPS/LTCH PPS final rule.
Regarding requests from providers to amend and/or reopen previously audited Worksheet S-10 data for the most recent 3 cost reporting years that are used in the methodology for calculating Factor 3, we noted that MACs follow normal timelines and procedures. We explained that for purposes of the Factor 3 calculation for the FY 2025 IPPS/LTCH PPS final rule, any amended reports and/or reopened reports would need to have completed the amended report and/or reopened report submission processes by the end of March 2024. In other words, if the amended report and/or reopened report was not available for the March HCRIS extract, then that amended and/or reopened report data would not be part of the FY 2025 IPPS/LTCH PPS final rule's Factor 3 calculation. We noted that the March HCRIS data extract would be available during the comment period for the proposed rule if providers wanted to verify that their amended and/or reopened data is reflected in the March HCRIS extract.
Since FY 2014, we have made interim uncompensated care payments during the fiscal year on a per-discharge basis. Typically, we use a 3-year average of the number of discharges for a hospital to produce an estimate of the amount of the hospital's uncompensated care payment per discharge. Specifically, the hospital's total uncompensated care payment amount for the applicable fiscal year is divided by the hospital's historical 3-year average of discharges computed using the most recent available data to determine the uncompensated care payment per discharge for that fiscal year.
In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45247 and 45248 ), we modified this calculation for FY 2022 to be based on an average of FY 2018 and FY 2019 historical discharge data, rather than a 3-year average using the most recent 3 years of discharge data, which would have included data from FY 2018, FY 2019, and FY 2020. We explained our belief that computing a 3-year average with FY 2020 discharge data would underestimate discharges, due to the decrease in discharges during the COVID-19 pandemic. For the same reason, in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49045 ), we calculated interim uncompensated care payments based on the 3-year average of discharges from FY 2018, FY 2019, and FY 2021 rather than a 3-year average using the most recent 3 years of discharge data.
We explained in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59010 ) that we believed that computing a 3-year average using the most recent 3 years of discharge data would potentially underestimate the number of discharges for FY 2024 due to the effects of the COVID-19 pandemic during FY 2020, which was the first year of the COVID-19 pandemic. We considered using an average of FY 2019, FY 2021, and FY 2022 discharge data to calculate the per-discharge amount for interim uncompensated care payments for FY 2024. However, we agreed with commenters that using FY 2019 data may overestimate discharge volume because updated claims data used to estimate the FY 2024 discharges in the Factor 1 calculation indicated that discharge volumes were not expected to return to pre-pandemic levels during FY 2024. Therefore, for FY 2024, we finalized a policy of calculating the per-discharge amount for interim uncompensated care payments using an average of FY 2021 and FY 2022 discharge data.
For FY 2025 and subsequent fiscal years, we proposed to calculate the per-discharge amount for interim uncompensated care payments using the average of the most recent 3 years of discharge data. Accordingly, for FY 2025, we proposed to use an average of discharge data from FY 2021, FY 2022, and FY 2023. We stated that we believed that our proposed approach would likely result in a better estimate of the number of discharges during FY 2025 and subsequent years for purposes of the interim uncompensated care payment calculation.
As we explained in the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50645 ), we generally believe that it is appropriate to use a 3-year average of discharge data to reduce the degree to which we would over- or under-pay the uncompensated care payment on an interim basis. In any given year, a hospital could have low or high Medicare utilization that differs from other years. For example, if a hospital had two Medicare discharges in its most recent year of claims data but experienced four discharges in FY 2025, during the fiscal year, we would pay two times the amount the hospital should receive and need to adjust for that at cost report settlement. Similarly, if a hospital had four Medicare discharges in its most recent year of claims data, but experienced two discharges in FY 2025, during the fiscal year, we would only pay half the amount the hospital should receive and need to adjust for that at cost report settlement. ( print page 69329)
In the FY 2025 IPPS/LTCH PPS proposed rule, we stated that we believed that, generally, use of the most recent 3 years of discharge data, rather than older data, is more likely to reflect current trends in discharge volume and provide an approximate estimate of the number of discharges in the applicable fiscal year. In addition, we noted that including discharge data from FY 2023 to compute this 3-year average would be consistent with the proposed use of FY 2023 Medicare claims in the IPPS ratesetting, as discussed in section I.E. of the preamble of this FY 2025 IPPS/LTCH PPS final rule.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to use the resulting 3-year average of the most recent years of available historical discharge data to calculate a per-discharge payment amount that would be used to make interim uncompensated care payments to each projected DSH-eligible hospital during FY 2025 and subsequent fiscal years. Interim uncompensated care payments made to a hospital during the fiscal year are reconciled following the end of the year to ensure that the final payment amount is consistent with the hospital's prospectively determined uncompensated care payment for the fiscal year.
We proposed to make conforming changes to the regulations under 42 CFR 412.106 . Specifically, we proposed to modify paragraph (1) of § 412.106(i) to state that for FY 2025 and subsequent fiscal years, interim uncompensated care payments will be calculated based on an average of the most recent 3 years of available historical discharge data. We requested comments on this proposal.
Comment: Several commenters requested that CMS use a two-year average of discharge data to estimate the per-discharge amount of interim uncompensated care payments for FY 2025 and/or for future fiscal years. One commenter suggested that CMS use an average of the two most recent years of discharge data. These commenters stated that a two-year average would better reflect anticipated FY 2025 discharges. Some commenters stated CMS overestimated discharge volume in its rulemaking in recent years. Some commenters stated that this overestimation depresses interim uncompensated care payments. A commenter urged CMS to modify its interim uncompensated care payment methodology to improve the effectiveness of DSH payments and reduce overreliance on the reconciliation process for uncompensated care payments. This commenter also stated that it is inconsistent for CMS to project a decline in discharges for the Factor 1 calculation while not assuming the same decline when projecting the discharges used to calculate the per-discharge amount. This same commenter stated there may be year-to-year variations in discharge volume, but there are also larger trends that reflect changing treatment patters, technology, Medicare Advantage penetration, and other factors. This same commenter supported a methodology that incorporates more than one year of data to appropriately temper volatility in year-to-year changes in discharge volume, but the commenter recommended to appropriately use more current data. This same commenter recommended using a two-year average of discharges to estimate the per-discharge amount of interim uncompensated care payments, in addition to incorporating a national adjustment factor so that the historical discharges can be trended forward to FY2025 estimate of discharges.
Response: We thank commenters for their feedback on calculation of the per-discharge amount of interim uncompensated care payments for FY 2025. In light of the commenters' concerns regarding a trend of decreasing discharge volume and possible overestimation of discharges in recent years, we believe that, on balance, omitting FY 2021 data from the calculation of interim uncompensated care payments is likely to more accurately estimate FY 2025 discharges. Therefore, we are finalizing our proposal with modification. Specifically, we will calculate the per-discharge amount of uncompensated care payments for FY 2025 using an average of the most recent 2 years of available historical discharge data: FY 2022 and FY 2023 discharge data. We are modifying the text of § 412.106(i)(1) to state that for FY 2025, interim uncompensated care payments will be calculated based on an average of the most recent 2 years of available historical discharge data.
Additionally, we believe using an average of the most recent 3 years of available historical discharge data will appropriately reflect year-to-year variations in discharge volumes in FY 2026 and subsequent fiscal years. As explained earlier in this section of this final rule and in the proposed rule, the effect of the COVID-19 pandemic on discharges was the rationale for modifying the interim uncompensated care payment methodology in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45247 and 45248 ). We believe the effect on discharge volume of the COVID-19 pandemic will likely be diminished beginning in FY 2026. Therefore, consistent with the proposed rule, we are modifying the text of § 412.106(i)(1) to state that for FY 2026 and subsequent fiscal years, interim uncompensated care payments will be calculated based on an average of the most recent 3 years of available historical discharge data.
At this time, we are not adopting a national adjustment approach because, as we explain more fully earlier in this section and in the proposed rule, we believe that in FY 2026 and subsequent years, using an average of the most recent 3 years of available discharge data will likely result in a reasonable estimate at the provider level, for purposes of interim uncompensated care payments. We will consider commenters' other suggested modifications to our interim uncompensated care payment policies, such as using a national adjustment factor, in future rulemaking.
Further, as we explained in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36198-36199 ), we finalized a voluntary process in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58833 and 58834 ), through which a hospital may submit a request to its MAC for a lower per-discharge interim uncompensated care payment amount, including a reduction to zero, once before the beginning of the fiscal year and/or once during the fiscal year. In conjunction with this request, the hospital must provide supporting documentation demonstrating that there would likely be a significant recoupment at cost report settlement if the per-discharge amount is not lowered (for example, recoupment of 10 percent or more of the hospital's total uncompensated care payment, or at least $100,000). For example, a hospital might submit documentation showing a large projected increase in discharges during the fiscal year to support reduction of its per-discharge uncompensated care payment amount. As another example, a hospital might request that its per-discharge uncompensated care payment amount be reduced to zero midyear if the hospital's interim uncompensated care payments during the year have already surpassed the total uncompensated care payment calculated for the hospital.
Under the policy we finalized in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58833 through 58834 ), the hospital's MAC will evaluate these requests and the supporting documentation before the beginning of the fiscal year and/or with midyear requests when the historical average number of discharges ( print page 69330) is lower than the hospital's projected discharges for the current fiscal year. If, following review of the request and the supporting documentation, the MAC agrees that there likely would be significant recoupment of the hospital's interim Medicare uncompensated care payments at cost report settlement, the only change that will be made is to lower the per-discharge amount either to the amount requested by the hospital or another amount determined by the MAC to be appropriate to reduce the likelihood of a substantial recoupment at cost report settlement. If the MAC determines it would be appropriate to reduce the interim Medicare uncompensated care payment per-discharge amount, that updated amount will be used for purposes of the outlier payment calculation for the remainder of the fiscal year. We are continuing to apply this policy for FY 2025. We refer readers to the Addendum in the FY 2023 IPPS/LTCH final rule for a more detailed discussion of the steps for determining the operating and capital Federal payment rate and the outlier payment calculation ( 87 FR 49431 through 49432 ). No change would be made to the total uncompensated care payment amount determined for the hospital on the basis of its Factor 3. In other words, any change to the per-discharge uncompensated care payment amount will not change how the total uncompensated care payment amount will be reconciled at cost report settlement.
We received comments related to the uncompensated care payment reconciliation process.
Comment: A commenter recommended that CMS use the traditional payment reconciliation process to calculate final payments for uncompensated care costs pursuant to section 1886(r)(2) of the Act. This commenter did not object to CMS using prospective estimates, derived from the best data available, to calculate interim payments for uncompensated care costs. However, the commenter stated that interim payments should be subject to later reconciliation based on estimates derived from actual data from the fiscal year. This commenter also stated that CMS' current IPPS/LTCH PPS rulemaking process is flawed because CMS may use data and calculations in final rules that were not included in the relevant proposed rules without providing advance notice to hospitals, limiting their ability to provide informed comments. Several commenters stated that CMS fails to provide meaningful explanations of its uncompensated care payment calculations and is in violation of the Administrative Procedure Act. These commenters recommended that CMS satisfy its legal obligation by providing hospitals the opportunity to review and comment on the more recent data used to calculate Factors 1, 2, and 3 in each final rulemaking before the agency publishes the final rule.
Response: Consistent with the position that we have taken in past rulemakings, we continue to believe that applying our best estimates of the three factors used in the calculation of uncompensated care payments to determine payments prospectively is most conducive to administrative efficiency, finality, and predictability in payments ( e.g., the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59011 ). We continue to believe that, in affording the Secretary the discretion to estimate the three factors used to determine uncompensated care payments and by including a prohibition against administrative and judicial review of those estimates in section 1886(r)(3) of the Act, Congress recognized the importance of finality and predictability under a prospective payment system. As a result, we do not agree with the commenter's suggestion that we should establish a process for reconciling our estimates of uncompensated care payments, which would be contrary to the notion of prospectivity in a payment system. Furthermore, we note that this rulemaking has been conducted consistent with the requirements of the Administrative Procedure Act and Title XVIII of the Act. Under the Administrative Procedure Act, a proposed rule is required to include either the terms or substance of the proposed rule, or a description of the subjects and issues involved. In this case, the FY 2025 IPPS/LTCH PPS proposed rule ( 86 FR 369193-36199 ) included a detailed discussion of our proposed methodology for calculating Factor 3 and the data that would be used. We made public the best data available at the time of the proposed rule to allow hospitals to understand the anticipated impact of the proposed methodology and submit comments, and we have considered those comments in determining our final policies for FY 2025.
As we have done for every proposed and final rule beginning in FY 2014, in conjunction with this final rule, we will publish on the CMS website a table listing Factor 3 for hospitals that we estimate will receive empirically justified Medicare DSH payments in FY 2025 (that is, those hospitals that will receive interim uncompensated care payments during the fiscal year), and for the remaining subsection (d) hospitals and subsection (d) Puerto Rico hospitals that have the potential of receiving an uncompensated care payment in the event that they receive an empirically justified Medicare DSH payment for the fiscal year as determined at cost report settlement. However, we note that a Factor 3 will not be published for new hospitals and hospitals that are subject to the alternative trim for hospitals with potentially aberrant data that are not projected to be DSH-eligible.
We also will publish a supplemental data file containing a list of the mergers that we are aware of and the computed uncompensated care payment for each merged hospital. In the DSH uncompensated care supplemental data file, we list new hospitals and the 8 hospitals that would be subject to the alternative trim for hospitals with potentially aberrant data that are not projected to be DSH-eligible, with aN/A in the Factor 3 column.
Hospitals had 60 days from the date of public display of the FY 2025 IPPS/LTCH PPS proposed rule in the Federal Register to review the table and supplemental data file published on the CMS website in conjunction with the proposed rule and to notify CMS in writing of issues related to mergers and/or to report potential upload discrepancies due to MAC mishandling of Worksheet S-10 data during the report submission process. [ 215 ] In the proposed rule, we stated that comments raising issues or concerns that are specific to the information included in the table and supplemental data file should be submitted by email to the CMS inbox at [email protected] . We indicated that we would address comments related to mergers and/or reporting upload discrepancies submitted to the CMS DSH inbox as appropriate in the table and the supplemental data file that we publish on the CMS website in conjunction with the publication of the FY 2025 IPPS/LTCH PPS final rule. We also stated that all other comments submitted in response to our proposals for FY 2025 must be submitted in one of the three ways found in the ADDRESSES section of the proposed rule before the close of the comment period in order to be assured consideration. In addition, we noted that the CMS DSH inbox is not intended for Worksheet S- ( print page 69331) 10 audit process related emails, which should be directed to the MACs.
As discussed in section III.B. of the preamble of this final rule, in the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to implement the new OMB labor market area delineations (which are based on 2020 Decennial Census data) for the FY 2025 wage index. We stated that this proposal also would have an impact on the calculation of Medicare DSH payment adjustments to certain hospitals. Hospitals that are designated as rural with less than 500 beds and are not rural referral centers (RRCs) or Medicare-dependent, small rural hospitals (MDHs) are subject to a maximum DSH payment adjustment of 12 percent. Accordingly, hospitals with less than 500 beds that are currently in urban counties that would become rural if we finalize our proposal to adopt the new OMB delineations, and that do not become RRCs or MDHs, would be subject to a maximum DSH payment adjustment of 12 percent. (We note, as discussed in section V.F.2. of the preamble of this final rule, under current law the MDH program will expire on December 31, 2024). We also note that urban hospitals are only subject to a maximum DSH payment adjustment of 12 percent if they have less than 100 beds.
In that same proposed rule, we explained that our existing regulations at 42 CFR 412.102 will apply in FY 2025 with respect to the calculation of the DSH payments to hospitals that are currently located in urban counties that would become rural if we finalize our proposal to adopt the new OMB delineations. The provisions of 42 CFR 412.102 specify that an urban hospital that was part of an MSA, but was redesignated as rural (as defined in the regulations), as a result of the most recent OMB standards for delineating statistical areas adopted by CMS, may receive an adjustment to its rural Federal payment amount for operating costs for two successive fiscal years. Specifically, the regulations state that, in the first year after a hospital loses urban status, the hospital will receive an additional payment that equals two thirds of the difference between the disproportionate share payments as applicable to the hospital before its redesignation from urban to rural and disproportionate share payments otherwise, applicable to the hospital subsequent to its redesignation from urban to rural. In the second year after a hospital loses urban status, the hospital will receive an additional payment that equals one-third of the difference between the disproportionate share payments applicable to the hospital before its redesignation from urban to rural and disproportionate share payments otherwise applicable to the hospital subsequent to its redesignation from urban to rural.
Comment: Commenters generally supported the application of 42 CFR 412.102 for urban hospitals located in an area that is redesignated as rural as a result of the most recent OMB standards for delineating statistical areas adopted by CMS. A few commenters expressed concern about the impact on DSH payments when an urban hospital becomes rural with the adoption of the updates to the CBSA designations. According to these commenters, rural hospitals are disadvantaged in the DSH statutory formula.
Response: We appreciate the support of the commenters as well as the concerns raised by the commenters. As discussed in section III.B. of this preamble, after consideration of public comments, we are finalizing our proposal to implement the new OMB labor market area delineations for FY 2025. Therefore, 42 CFR 412.102 will apply to those urban hospitals currently located in an area that will be redesignated as rural beginning October 1, 2024. We believe the special treatment for these hospitals under the regulations at 42 CFR 412.102 helps mitigate the commenters' concerns as urban hospitals in areas that will be redesignated as rural due to the new OMB labor market area delineations may receive an additional payment for two years as described previously in this section.
In Becerra v. Empire Health Foundation, for Valley Hospital Medical Center, 597 U.S. 424 (2022) ( Empire Health ), the Supreme Court addressed the question of whether Medicare patients remain “entitled to benefits under part A” when Medicare does not pay for their care, such as when they have exhausted their Medicare benefits for a spell of illness. Prior to fiscal year (FY) 2005, when we calculated a hospital's DSH adjustment we included in the Medicare fraction (also referred to as the Medicare-SSI fraction, SSI fraction, or SSI ratio) only “covered” Medicare patient days, that is, days paid by Medicare (see 42 CFR 412.106(b)(2)(i) (2003)). The “covered” days rule originated in the FY 1986 IPPS interim final rule ( 51 FR 16772 and 16788 ) and originally appeared in § 412.106(a)(1)(i) but was later re-numbered. The approach of excluding from the Medicare fraction patient days for which Medicare did not pay was based on an interpretation of the statute's parenthetical phrase “(for such days).” Section 1886(d)(5)(F)(vi)(I) of the Act. Following a series of judicial decisions rejecting a parallel interpretation of the same language in the numerator of the Medicaid fraction as counting only patient days actually paid by the Medicaid program, the Secretary revisited that approach in a 2004 rulemaking. Thus, the “covered days” rule was the relevant Medicare payment policy until it was revised and replaced by the FY 2005 IPPS final rule ( 69 FR 48916 , 49099 , and 49246 ).
The FY 2005 regulation at issue in Empire Health —codified in the FY 2005 IPPS final rule—interpreted the statute to mean that the Medicare fraction includes non-covered days in the SSI ratio. (For more information see 69 FR 48916 , 49099 , and 49246 (amending 42 CFR 412.106(b)(2)(i) to include in the Medicare fraction all days associated with patients who were entitled to Medicare Part A during their hospital stays, regardless of whether Medicare paid for those days).) In Empire Health, the Supreme Court upheld the FY 2005 regulation and held that the statute “disclose[s] a surprisingly clear meaning,” 597 U.S. at 434, namely that beneficiaries remain “entitled to benefits under part A” on days for which Medicare does not pay and thus the Medicare fraction includes total days, not only covered days. The Supreme Court also definitively resolved the meaning of the parenthetical phrase “(for such days)” in the Medicare fraction, rejecting the provider's contention that the phrase changed the consistent meaning of “entitled to benefits under Part A” from “meeting Medicare's statutory (age or disability) criteria on the days in question,” to “actually receiving Medicare payments.” Id. at 440. The Court determined that the “for such days” parenthetical “instead works as HHS says: hand in hand with the ordinary statutory meaning of `entitled to [Part A] benefits.' ” Id.
The Supreme Court has concluded that the interpretation set forth in the FY 2005 IPPS final rule “correctly construes the statutory language at issue.” Empire Health, 597 U.S. at 434. Because the pre-FY 2005 rule conflicts with the plain meaning of the statute, as confirmed by the Supreme Court, it cannot govern the calculation of DSH ( print page 69332) payments for hospitals with properly pending claims in DSH appeals or open cost reports that include discharges that need to be determined pursuant to the statute, regardless of whether such discharges would otherwise pre-date the change in the regulation finalized by the FY 2005 IPPS final rule. For that reason, we proposed to formally withdraw 42 CFR 412.106 as it existed prior to the effective date of the FY 2005 IPPS final rule to the extent it included only covered days in the SSI ratio. We will apply the statute as understood by the Supreme Court in Empire Health, instead of the pre-FY 2005 regulation, to any properly pending claim in a DSH appeal or open cost report to which that regulation would otherwise have applied. We do not believe this change constitutes an exercise of our “retroactive” rulemaking authority under section 1871(e)(1)(A) of the Act. Rather, we will apply the plain meaning of the statute (as it has existed unchanged, in relevant part, since its enactment on April 7, 1986). Moreover, because we are applying the substantive legal standard established by the statute itself, and not filling any gap therein, notice-and-comment rulemaking is not required by section 1871(e)(1)(A) of the Act, as construed in Azar v. Allina Health Services, 139 S. Ct. 1804 (June 3, 2019).
The withdrawal of this regulation will not serve as a basis to reopen a CMS or contractor determination, a contractor hearing decision, a CMS reviewing official decision, or a decision by the Provider Reimbursement Review Board or the Administrator. We recognize that hospitals may have anticipated receiving greater Medicare reimbursement for still-open pre-FY 2005 cost reporting periods in circumstances where the “covered” days limitation would have resulted in a larger DSH adjustment. However, we are obliged to apply the statute as the Supreme Court determined Congress wrote it.
Comment: A commenter opposed our proposal to withdraw 42 CFR 412.106 for several reasons. The commenter stated that our proposal is based on a misreading of Empire Health. According to the commenter, the Supreme Court held that our interpretation of section 1886(d)(5)(F)(vi)(I) of the Act to include unpaid patient days in the Medicare fraction is merely supported by the statute, not required by the statute. The commenter argued that the proposal rests on an interpretation not required by statute, citing Northeast Hospital Corp. v. Sebelius, 657 F.3d 1 (D.C. Cir. 2011), and is against the public interest, thus constituting improper retroactive rulemaking. The commenter further argued that our proposal would unfairly penalize affected hospitals and deprive them of fair notice and due process. In addition, the commenter argued our proposal would be unfair to hospitals that are still waiting to receive DSH payments calculated in accordance with the pre-FY 2005 version of 42 CFR 412.106 because other hospitals already received the benefit of that rule before the Supreme Court issued its decision in Empire Health. The commenter also asserted that we did not finalize the 2005 revision until 2007 with a technical correction to the regulation text.
Response: We disagree with the commenter's reading of the Supreme Court's Empire Health decision. Empire Health addressed the question of whether, for purposes of calculating a hospital's DSH adjustment and Medicare fraction, Medicare patients remain “entitled to benefits under part A” when Medicare does not pay for their care, such as when they have exhausted their Medicare benefits for a spell of illness. As we explained in the proposed rule, prior to FY 2005, our approach to calculating a hospital's DSH adjustment, as provided in our regulations starting with the FY 1986 IPPS interim final rule ( 51 FR 16772 and 16788 ), was to include in the Medicare fraction only “covered” Medicare patient days, that is, days paid by Medicare. The “covered days” approach was based on an interpretation of the statute's parenthetical phrase “(for such days).” Following a series of judicial decisions rejecting a parallel interpretation of the “(for such days)” language in the numerator of the Medicaid fraction as counting only patient days actually paid by the Medicaid program, we revised and replaced this rule in the FY 2005 IPPS final rule ( 69 FR 48916 , 49099 , and 49246 ). We note that we further disagree with the commenter's assertion that we did not finalize this revision until 2007 with a technical correction to the regulation text. Under the policy finalized in the FY 2005 IPPS final rule, we interpreted the statute to mean that the Medicare fraction includes covered and non-covered Medicare patient days (that is, “total days”) because Medicare patients remain entitled to Part A benefits even on patient days not covered by Medicare. In upholding this reading of the statute, the Supreme Court in Empire Health did not conclude merely, as the commenter states, that the statute “supported” our interpretation. Rather, the Court concluded that “being `entitled' to Medicare benefits . . . means—in the [DSH] fraction descriptions, as throughout the statute—meeting the basic statutory criteria, not actually receiving payment for a given day's treatment.” 597 U.S. at 435. The Court reaffirmed that this was its own conclusion when it said elsewhere, “The structure of the relevant statutory provisions reinforces our conclusion that `entitled to [Part A] benefits' means qualifying for those benefits, and nothing more.” Id. at 442 (alteration in original) (emphasis added). And the Court rejected the notion that the “(for such days)” parenthetical required a “covered days” approach, concluding instead that it “works as HHS says: hand in hand with the ordinary statutory meaning of `entitled to [Part A] benefits.' ” Id. at 440 (alteration in original). We note that the Supreme Court's holding in Empire Health displaced Northeast Hospital Corp. v. Sebelius, 657 F.3d 1, 6-13 (D.C. Cir. 2011), to the extent that it supports a conclusion that the statutory language was ambiguous on the issue of “covered days” versus “total days.” Thus, in the wake of Empire Health, the commenter's reliance on Northeast in support of its opposition to our proposal is unfounded.
We also disagree with the commenter's suggestion that Empire Health left a gap in the statute for the agency to fill that would require notice-and-comment rulemaking under the Supreme Court's earlier decision in Azar v. Allina Health Services, 587 U.S. 566 (2019). To the contrary, Empire Health made clear that the statute established the substantive legal standard that we articulated in the FY 2005 IPPS final rule ( i.e., for purposes of the DSH adjustment calculation, Medicare patients are “entitled to [Part A] benefits” if they are qualified for those benefits, regardless of whether Medicare pays for their hospital stay, and all patient days associated with these patients are counted in the Medicare fraction). It follows from this, as we stated in the FY 2025 proposed rule, that the pre-FY 2005 rule that counted only covered days in the Medicare fraction conflicts with the plain meaning of the statute, and it should thus be withdrawn.
Contrary to the commenter's contention and consistent with what we said in the proposed rule, we do not believe that withdrawing a regulation that conflicts with the governing statute constitutes an exercise of our “retroactive” rulemaking authority under section 1871(e)(1)(A) of the Act. Rather, we are simply giving effect to the language of the statute as it has ( print page 69333) existed throughout the relevant time period. Nonetheless, even if the withdrawal could be seen as an exercise of retroactive rulemaking, the Secretary has determined that the withdrawal is necessary to comply with statutory requirements; namely, the statutory requirement that we include in the Medicare fraction all patient days attributable to Medicare patients, regardless of whether Medicare paid for services on those days. As we stated in the proposed rule, we must follow the statute as the Supreme Court determined Congress wrote it. This would be a sufficient basis for us to engage in retroactive rulemaking, per section 1871(e)(1)(A)(i) of the Act, if this withdrawal could be seen as retroactive rulemaking, which it is not. As such, it is unnecessary for us to address the commenter's additional assertion that the Secretary is not authorized here, under section 1871(e)(1)(A)(ii) of the Act, to apply a change in regulations retroactively to further the public interest. We do not have authority to make DSH adjustments that do not comply with the statute as written by Congress and interpreted by the Supreme Court, and it would therefore be contrary to the public interest for us to maintain the rule in its pre-FY 2005 form. We also disagree with the commenter's claim that our proposed withdrawal of the pre-FY 2005 rule is contrary to the public interest because some hospitals' DSH adjustments will be reduced. Following the statute as written and in accordance with Supreme Court precedent is in the public interest, not contrary to it, even if it results in smaller DSH payment adjustments than some hospitals may have hoped for. Moreover, while we agree with the commenter that advance notice-and-comment rulemaking is generally both necessary and in the public interest, we have taken that interest into account here by finalizing the proposed withdrawal only after a notice-and-comment process. In any event, the commenter does not point to any authority for the proposition that the interest in advance notice-and-comment rulemaking, as they envision it, could permit us to keep on the books and follow a regulation that is contrary to the statute.
We also disagree with the commenter's assertion that our proposal would penalize affected hospitals and deprive them of fair notice and due process. Our proposal applies the meaning of the statute as it was written in 1986, and the operative language has not changed in any material way since then. As we said in the proposed rule, we recognize that hospitals may have anticipated receiving greater Medicare reimbursement for their still-open pre-FY 2005 cost reporting periods in circumstances where the “covered days” limitation would have resulted in a larger DSH payment. Nonetheless, it would not be reasonable for those hospitals to expect us to ignore the statute and pay them more than Congress allowed or, to the extent their still-open DSH adjustments were already paid based on the pre-FY 2005 rule, allow them to retain overpayments not authorized by Congress, after the Supreme Court settled the plain meaning of the statute. Our proposal was not meant to penalize affected hospitals in any way, and it is not an enforcement action. Rather, our proposal was intended to ensure, going forward, that providers' DSH adjustments are paid in accordance with the statute.
Finally, we disagree with the commenter's assertion that our proposal would be unfair to affected hospitals because other hospitals whose cost reporting periods were settled before the Supreme Court issued Empire Health received the benefit of the “covered days” limitation. That other hospitals were paid on the basis of “covered days” in the past cannot justify continuing to do so going forward now that the Supreme Court has settled the meaning of the statute. It is neither unfair nor unusual for cost reports to be finalized differently from one another with respect to a legal issue depending on the outcome of litigation raising that issue and the status of a hospital's cost report at the time of a final non-appealable decision. And while Empire Health did not specifically address the legality of the pre-FY 2005 rule, that rule directly conflicts with the meaning of the statute as settled by the Supreme Court in that case. Further, to the extent the commenter argues that it is unfair that affected hospitals had to wait for years to receive their DSH adjustment payments and, in the process, lost the benefit of the “covered days” limitation that other hospitals already benefited from, we note that the wait was caused by protracted litigation over numerous aspects of the DSH calculation and the scope of the Medicare statute's rulemaking requirements, which significantly slowed (and, at times, ground to a halt) our ability to perform such calculations and enable our contractors to settle providers' open cost reports that were involved in, or affected by, such litigation.
After considering the comment received, we are finalizing our proposal to formally withdraw 42 CFR 412.106 as it existed prior to the effective date of the FY 2005 IPPS final rule to the extent it included only covered days in the SSI ratio when calculating a hospital's DSH adjustment. The withdrawal of this regulation will not serve as a basis to reopen a CMS or contractor determination, a contractor hearing decision, a CMS reviewing official decision, or a decision by the Provider Reimbursement Review Board or the Administrator.
We received several comments outside the scope of the proposed rule. These comments related to the exclusion of days associated with uncompensated care pools under section 1115 waivers from the numerator of the Medicaid fraction, requests for CMS to modernize DSH and work more closely with other agencies, Medicaid eligibility and redetermination, and concern over 340B eligibility. Because we consider these public comments to be outside the scope of the proposed rule, we are not addressing these comments in this final rule.
Existing regulations at 42 CFR 412.4(a) define discharges under the IPPS as situations in which a patient is formally released from an acute care hospital or dies in the hospital. Section 412.4(b) defines acute care transfers, and § 412.4(c) defines postacute care transfers. Our policy set forth in § 412.4(f) provides that when a patient is transferred and his or her length of stay is less than the geometric mean length of stay for the MS-DRG to which the case is assigned, the transferring hospital is generally paid based on a graduated per diem rate for each day of stay, not to exceed the full MS-DRG payment that would have been made if the patient had been discharged without being transferred.
The per diem rate paid to a transferring hospital is calculated by dividing the full MS-DRG payment by the geometric mean length of stay for the MS-DRG. Based on an analysis that showed that the first day of hospitalization is the most expensive ( 60 FR 45804 ), our policy generally provides for payment that is twice the per diem amount for the first day, with each subsequent day paid at the per diem amount up to the full MS-DRG payment (§ 412.4(f)(1)). Transfer cases also are eligible for outlier payments. In ( print page 69334) general, the outlier threshold for transfer cases, as described in § 412.80(b), is equal to (Fixed-Loss Outlier threshold for Nontransfer Cases adjusted for geographic variations in costs/Geometric Mean Length of Stay for the MS-DRG) * (Length of Stay for the Case plus 1 day).
We established the criteria set forth in § 412.4(d) for determining which DRGs qualify for postacute care transfer payments in the FY 2006 IPPS final rule ( 70 FR 47419 through 47420 ). The determination of whether a DRG is subject to the postacute care transfer policy was initially based on the Medicare Version 23.0 GROUPER (FY 2006) and data from the FY 2004 MedPAR file. However, if a DRG did not exist in Version 23.0 or a DRG included in Version 23.0 is revised, we use the current version of the Medicare GROUPER and the most recent complete year of MedPAR data to determine if the DRG is subject to the postacute care transfer policy. Specifically, if the MS-DRG's total number of discharges to postacute care equals or exceeds the 55th percentile for all MS-DRGs and the proportion of short-stay discharges to postacute care to total discharges in the MS-DRG exceeds the 55th percentile for all MS-DRGs, CMS will apply the postacute care transfer policy to that MS-DRG and to any other MS-DRG that shares the same base MS-DRG. The statute at subparagraph 1886(d)(5)(J) of the Act directs CMS to identify MS-DRGs based on a high volume of discharges to postacute care facilities and a disproportionate use of postacute care services. As discussed in the FY 2006 IPPS final rule ( 70 FR 47416 ), we determined that the 55th percentile is an appropriate level at which to establish these thresholds. In that same final rule ( 70 FR 47419 ), we stated that we will not revise the list of DRGs subject to the postacute care transfer policy annually unless we are making a change to a specific MS-DRG.
To account for MS-DRGs subject to the postacute care policy that exhibit exceptionally higher shares of costs very early in the hospital stay, § 412.4(f) also includes a special payment methodology. For these MS-DRGs, hospitals receive 50 percent of the full MS-DRG payment, plus the single per diem payment, for the first day of the stay, as well as a per diem payment for subsequent days (up to the full MS-DRG payment (§ 412.4(f)(6))). For an MS-DRG to qualify for the special payment methodology, the geometric mean length of stay must be greater than 4 days, and the average charges of 1-day discharge cases in the MS-DRG must be at least 50 percent of the average charges for all cases within the MS-DRG. MS-DRGs that are part of an MS-DRG severity level group will qualify under the MS-DRG special payment methodology policy if any one of the MS-DRGs that share that same base MS-DRG qualifies (§ 412.4(f)(6)).
Prior to the enactment of the Bipartisan Budget Act of 2018 ( Pub. L. 115-123 ), under section 1886(d)(5)(J) of the Act, a discharge was deemed a “qualified discharge” if the individual was discharged to one of the following postacute care settings:
- A hospital or hospital unit that is not a subsection (d) hospital.
- A skilled nursing facility.
- Related home health services provided by a home health agency provided within a timeframe established by the Secretary (beginning within 3 days after the date of discharge).
Section 53109 of the Bipartisan Budget Act of 2018 amended section 1886(d)(5)(J)(ii) of the Act to also include discharges to hospice care provided by a hospice program as a qualified discharge, effective for discharges occurring on or after October 1, 2018. In the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41394 ), we made conforming amendments to § 412.4(c) of the regulation to include discharges to hospice care occurring on or after October 1, 2018, as qualified discharges. We specified that hospital bills with a Patient Discharge Status code of 50 (Discharged/Transferred to Hospice—Routine or Continuous Home Care) or 51 (Discharged/Transferred to Hospice, General Inpatient Care or Inpatient Respite) are subject to the postacute care transfer policy in accordance with this statutory amendment.
As discussed in the proposed rule and section II.C. of the preamble this final rule, based on our analysis of FY 2023 MedPAR claims data, CMS proposed to make changes to a number of MS-DRGs, effective for FY 2025. Specifically, we proposed the following changes:
- Adding ICD-10-PCS codes describing left atrial appendage closure (LAAC) procedures and cardiac ablation procedures to proposed new MS-DRG 317 (Concomitant Left Atrial Appendage Closure and Cardiac Ablation).
- Deleting existing MS-DRGs 453, 454, and 455 (Combined Anterior and Posterior Spinal Fusion with MCC, with CC, and without CC/MCC, respectively) and to reassign procedures from the existing MS-DRGs, 453, 454, and 455 and MS-DRGs 459 and 460 (Spinal Fusion Except Cervical with MCC and without MCC, respectively) to proposed new MS-DRG 402 (Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical), proposed new MS-DRGs 426, 427, and 428 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with MCC, with CC, without MCC/CC, respectively), proposed new MS-DRGs 429 and 430 (Combined Anterior and Posterior Cervical Spinal Fusion with MCC and without MCC, respectively), and proposed new MS-DRGs 447 and 448 (Multiple Level Spinal Fusion Except Cervical with MCC and without MCC, respectively). We also proposed to revise the title of MS-DRGs 459 and 460 to “Single Level Spinal Fusion Except Cervical with MCC and without MCC, respectively”. As discussed in section II.C. of the preamble of this final rule and later in this section, we are finalizing our proposals, with modification, to reflect the reassignment of cases reporting the use of a custom-made anatomically designed interbody fusion device and to delete MS-DRGs 459 and 460 and renumber as MS-DRGs 450 and 451.
- Reassigning cases that report a principal diagnosis of acute leukemia with an “other” O.R. procedure from MS-DRGs 834, 835, and 836 (Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC, respectively) to new MS-DRG 850 (Acute Leukemia with Other O.R. Procedures). We note that we also proposed to revise the title of MS-DRGs 834, 835, and 836 from “Acute Leukemia without Major O.R. Procedures with MCC, with CC, and without CC/MCC”, respectively to “Acute Leukemia with MCC, with CC, and without CC/MCC”.
We noted in the proposed rule that proposed revised MS-DRGs 459 and 460 are currently subject to the postacute care transfer policy. We stated that we believe it is appropriate to reevaluate the postacute care transfer policy status for MS-DRGs 459 and 460. When proposing changes to MS-DRGs that involve adding, deleting, and reassigning procedures between proposed new and revised MS-DRGs, we continue to believe it is necessary to evaluate all of the affected MS-DRGs to determine whether they should be subject to the postacute care transfer policy.
We stated that MS-DRGs 834, 835, and 836 are currently not subject to the postacute care transfer policy. We noted that while we are proposing to reassign certain cases from these MS-DRGs to newly proposed MS-DRGs, we estimated that less than 5 percent of the current cases would shift from the current assigned MS-DRGs to the proposed new MS-DRGs. We stated that ( print page 69335) we do not consider these proposed revisions to constitute a material change that would warrant reevaluation of the postacute care status of MS-DRGs 834, 835, and 836. CMS may further evaluate what degree of shifts in cases for existing MS-DRGs warrant consideration for the review of postacute care transfer and special payment policy status in future rulemaking.
In light of the proposed changes to the MS-DRGs for FY 2025, according to the regulations under § 412.4(d), we evaluated the MS-DRGs using the general postacute care transfer policy criteria and data from the FY 2023 MedPAR file. If an MS-DRG qualified for the postacute care transfer policy, we also evaluated that MS-DRG under the special payment methodology criteria according to regulations at § 412.4(f)(6). We continue to believe it is appropriate to assess new MS-DRGs and reassess revised MS-DRGs when proposing reassignment of procedure codes or diagnosis codes that would result in material changes to an MS-DRG.
We stated that proposed new MS-DRGs 426, 427, 447, and 448 would qualify to be included on the list of MS-DRGs that are subject to the postacute care transfer policy. As described in the regulations at § 412.4(d)(3)(ii)(D), MS-DRGs that share the same base MS DRG will all qualify under the postacute care transfer policy if any one of the MS-DRGs that share that same base MS-DRG qualifies. We therefore proposed to add proposed new MS-DRGs 426, 427, 428, 447, and 448 to the list of MS-DRGs that are subject to the postacute care transfer policy.
We noted that MS-DRGs 459 and 460 are currently subject to the postacute care transfer policy. As a result of our review, these MS-DRGs, as proposed to be revised, would not qualify to be included on the list of MS-DRGs that are subject to the postacute care transfer policy. We therefore proposed to remove revised MS-DRGs 459 and 460 from the list of MS-DRGs that are subject to the postacute care transfer policy.
As discussed in section II.C. of the preamble of this final rule, we are finalizing these proposed changes to the MS-DRGs, with modification, to delete MS-DRGs 459 and 460 and renumber these MS-DRGs as MS-DRGs 450 and 451. We therefore have evaluated the renumbered MS-DRGs 450 and 451 in the updated analysis that follows.
Using the March 2024 update of the FY 2023 MedPAR file, we have developed the following chart which sets forth the most recent analysis of the postacute care transfer policy criteria completed for this final rule with respect to each of these new or revised MS-DRGs.

During our annual review of proposed new or revised MS-DRGs and analysis of the December 2023 update of the FY 2023 MedPAR file, we reviewed the list of proposed revised or new MS-DRGs that qualify to be included on the list of MS-DRGs subject to the postacute care transfer policy for FY 2025 to determine ( print page 69338) if any of these MS-DRGs would also be subject to the special payment methodology policy for FY 2025.
Based on our analysis of the proposed changes to MS-DRGs included in the proposed rule, we determined that proposed new MS-DRGs 426, 427, and 447 meet the criteria for the MS-DRG special payment methodology. As described in the regulations at § 412.4(f)(6)(iv), MS-DRGs that share the same base MS-DRG will all qualify under the MS-DRG special payment policy if any one of the MS-DRGs that share that same base MS-DRG qualifies. We proposed that MS-DRGs 426, 427, 428, 447, 448, would be subject to the MS-DRG special payment methodology, effective for FY 2025. For this final rule, we updated this analysis using data from the March 2024 update of the FY 2023 MedPAR file.

Comment: We received a comment stating that the new MS-DRGs proposed as eligible for the postacute care policy are all related to spinal fusions. The commenter stated that these MS-DRGs have extremely high upfront costs. The commenter stated that CMS should not adopt this proposal due to the negative impact on hospitals that provide these services.
Response: The spinal fusion MS-DRGs that were proposed to be added to the list of MS-DRGs subject to the postacute care transfer policy were also proposed to be added to the special payment policy. Under this policy, the transferring hospital would receive 50 percent of the full MS-DRG payment, plus a single per diem payment, for the first day of the stay, as well as a per diem payment for subsequent days (up to the full MS-DRG payment). The intent of the special payment policy is specifically to address MS-DRGs with high initial costs. We believe the proposed addition of MS-DRGs 426, 427, 428, 447, and 448 to the special payment policy adequately addresses the specific concerns expressed by the commenter.
After consideration of the comment we received, we are finalizing our proposal to add MS-DRGs 426, 427, 428, 447, and 448 to the list of MS-DRGs subject to the postacute care and special payment policies. As noted, we proposed to remove MS-DRGs 459 and 460 from the list of MS-DRGS subject to the postacute care policy. These MS-DRGs are being deleted and renumbered to MS-DRGs 450 and 451, which will not be added to the postacute care policy list.
The postacute care transfer and special payment policy status of these MS-DRGs is reflected in Table 5 associated with this final rule, which is listed in section VI. of the Addendum to this final rule and available on the CMS website.
In accordance with section 1886(b)(3)(B)(i) of the Act, each year we update the national standardized amount for inpatient hospital operating costs by a factor called the “applicable percentage increase.” For FY 2025, we stated in the proposed rule that we are setting the applicable percentage increase by applying the adjustments listed in this section in the same sequence as we did for FY 2024. (We note that section 1886(b)(3)(B)(xii) of the Act required an additional reduction each year only for FYs 2010 through 2019.) Specifically, consistent with section 1886(b)(3)(B) of the Act, as amended by sections 3401(a) and 10319(a) of the Affordable Care Act, we stated that we are setting the applicable percentage increase by applying the following adjustments in the following sequence. The applicable percentage increase under the IPPS for FY 2025 is equal to the rate-of-increase in the hospital market basket for IPPS hospitals in all areas, subject to all of the following:
- A reduction of one-quarter of the applicable percentage increase (prior to the application of other statutory adjustments; also referred to as the market basket update or rate-of-increase (with no adjustments)) for hospitals that fail to submit quality information under rules established by the Secretary in accordance with section 1886(b)(3)(B)(viii) of the Act.
- A reduction of three-quarters of the applicable percentage increase (prior to the application of other statutory adjustments; also referred to as the market basket update or rate-of-increase (with no adjustments)) for hospitals not considered to be meaningful EHR users in accordance with section 1886(b)(3)(B)(ix) of the Act.
- An adjustment based on changes in economy-wide multifactor productivity (MFP) (the productivity adjustment).
Section 1886(b)(3)(B)(xi) of the Act, as added by section 3401(a) of the Affordable Care Act, states that application of the productivity adjustment may result in the applicable percentage increase being less than zero.
As published in the FY 2006 IPPS final rule ( 70 FR 47403 ), in accordance with section 404 of Public Law 108-173 , CMS determined a new frequency for rebasing the hospital market basket of every 4 years. In compliance with section 404 of the of Public Law 108-173 , in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45194 through 45204 ), we replaced the 2014-based IPPS operating and capital market baskets ( print page 69339) with the rebased and revised 2018-based IPPS operating and capital market baskets beginning in FY 2022. Consistent with our established frequency of rebasing the IPPS market basket every 4 years, we plan on proposing to rebase and revise the IPPS market basket in the FY 2026 IPPS/LTCH PPS proposed rule. We note that our preliminary evaluation of more recent Medicare cost report data for IPPS hospitals for 2022 indicates that the major IPPS market basket cost weights (particularly the compensation and drug cost weights) are similar to those finalized in the 2018-based IPPS market basket.
We proposed to base the FY 2025 market basket update used to determine the applicable percentage increase for the IPPS on IHS Global Inc.'s (IGI's) fourth quarter 2023 forecast of the 2018-based IPPS market basket rate-of-increase with historical data through third quarter 2023, which was estimated to be 3.0 percent. We also proposed that if more recent data subsequently became available (for example, a more recent estimate of the market basket update), we would use such data, if appropriate, to determine the FY 2025 market basket update in the final rule.
Comment: Several commenters expressed concerns that the proposed FY 2025 market basket update does not adequately reflect the rising inflation and costs that hospitals have faced over the last few years. Commenters stated that economy-wide inflation grew by 12.4 percent from 2021 through 2023 (as measured by the Consumer Price Index (CPI)), more than two times faster than Medicare reimbursement for hospital inpatient care, which increased by 5.2 percent during the same time. Several commenters noted that the most recent CPI for March 2024 reported nationwide inflation at 3.5 percent and inpatient hospital services inflation of 6.9 percent, outpacing Medicare's reimbursement.
Many commenters stated that rapid and sustained growth in labor costs have put persistent cost pressure on hospitals. They also noted increases in drug prices, citing a recent study and a report by the Health and Human Services (HHS) Assistant Secretary for Planning and Evaluation which found that in 2022 and 2023, prices for nearly 2,000 drugs increased faster than the rate of general inflation, with an average price increase of 15.2 percent. Several commenters also stated that hospitals have seen significant growth in administrative costs due to what they described as inappropriate practices by large commercial health insurers, including Medicare Advantage and Medicaid managed care plans, such as automatic claim denials and onerous prior authorization requirements. Several commenters also discussed the continued costs of addressing past and preventing future cyberattacks and a commenter stated they have seen significant increases in capital costs, particularly since the pandemic. A commenter stated that recently increased tariffs on imported supplies from China will result in substantial price increase for gloves, masks, needles, and other supplies. A commenter stated private equity firms continue to achieve greater penetration across healthcare markets and the costs of contracting with specialties such as physician practices has skyrocketed, which the commenter stated is not factored into CMS' payments. Commenters urged CMS to consider the changing health care environment which they state is putting enormous financial strain on hospitals and health systems and is expected to continue through 2025. A commenter stated that the net market basket update is too low, and that the budget neutrality impact of the low wage policy will exacerbate the insufficient market basket update for high wage areas.
Several commenters proposed CMS apply a payment increase of at least 4.1 percent which is aligned with MedPAC's March 2024 Report to Congress, which recommended a 1.5 percentage points increase over the FY 2025 payment update. These commenters noted that this was the second year that MedPAC made a recommendation of increasing the market basket update. A commenter stated that, while they fully understand the need to protect the Medicare Hospital Insurance Trust Fund, they requested CMS review data beyond normal data and consider increasing the market basket amount to at least 3.5 percent to more realistically reflect inflation. Several commenters suggested various higher market basket increases, which they believe better reflects hospitals' input prices and the contract labor staffing challenge. A commenter encouraged CMS to consider, at a minimum, matching the 3.7 percent increase that the commenter stated Medicare Advantage will receive. A commenter supported an annual inflation-based payment update based on the full Medicare Economic Index.
Several commenters recommended CMS look to alternative data sources that they asserted better reflect true labor and input cost increases in a timely manner. The commenters stated that the proposed payment update does not recognize these challenges, nor does it factor in the realities of inflation impacting operating costs. Commenters also stated CMS must use data that better reflects the input price inflation that hospitals have experienced and are projected to experience in FY 2025. A commenter stated that they did not understand why the FY 2025 market basket increase is lower than FY 2024. A commenter recommended CMS use more recent data to update adjustments to 2025 IPPS rates.
Several commenters requested that CMS use its “special exceptions and adjustments” authority to implement a market basket adjustment that is more consistent with the significant cost increases that are being experienced by hospitals. They urged CMS to revisit its assumptions and focus on appropriately accounting for recent and future trends in inflationary pressure and cost increases in the hospital payment update, which they stated is essential to ensure that Medicare payments for acute care services more accurately reflect the cost of providing hospital care. A commenter urged CMS to adjust its methodology for calculating the annual payment update for FY 2025 to ensure it provides a robust payment update that adequately incorporates the effects of inflation and rising workforce costs on hospitals. A commenter asked that CMS, at a minimum, reconsider the proposed labor expense calculations to provide a more appropriate update based on growing and unsustainable costs. Several commenters recommended a comprehensive evaluation of the current rate-setting methodology to accurately capture the true costs of care delivery and provide a fair and sustainable reimbursement framework. A commenter stated it was unacceptable that CMS' payment update does not factor in changes in hospital admissions, case-mix intensity, or the mandatory 2 percent sequestration adjustment reductions.
Many commenters noted their financial pressures due to the PHE, aging, more complex patients, negative Medicare margins of −12.7 percent as estimated by MedPAC, and reliance on public payers. Several commenters noted that historically, hospitals mitigated losses incurred from serving underinsured patients by negotiating higher payment rates from commercial payors; however, due to high inflation and an increasing deficit generated by serving governmental payor patients, they stated hospitals can no longer rely on commercial payors to offset those losses. Several commenters urged CMS to consider and assess the financial position of hospitals, particularly those with low margins. A commenter ( print page 69340) advocated for a comprehensive review of the Medicare margins and asked CMS increase rates to cover the cost of care for Medicare Advantage and Medicaid patients. Some commenters stated that hospitals will continue to face increased costs due to the Change Healthcare cyberattack, such as interest costs on loan payments for loans acquired during the cyberattack, expected denials that will require additional administrative costs, and manual processing of claims.
Response: Section 1886(b)(3)(B)(iii) of the Act states the Secretary shall update IPPS payments based on a market basket percentage increase, which is defined as the percentage, estimated by the Secretary before the beginning of the period or fiscal year, by which the cost of the mix of goods and services (including personnel costs but excluding nonoperating costs) comprising routine, ancillary, and special care unit inpatient hospital services, based on an index of appropriately weighted indicators of changes in wages and prices which are representative of the mix of goods and services included in such inpatient hospital services, for the period or fiscal year will exceed the cost of such mix of goods and services for the preceding 12-month cost reporting period or fiscal year. We believe that the 2018-based IPPS market basket is consistent with the statute as it is a fixed-weight, Laspeyres-type price index that measures the change in price, over time, while maintaining a mix of goods and services purchased by hospitals consistent with a base period. Therefore, the market basket is designed to measure price inflation for IPPS hospitals and would not reflect increases in costs associated with changes in the volume or intensity of input goods and services (such as the quantity of labor used). Regarding the commenter who stated that the budget neutrality adjustment from the low wage policy would exacerbate the inadequate market basket update, we note that the market basket update does not consider the impact of budget neutrality adjustments. We refer the reader to section IV.D. of the preamble of this final rule where we respond to comments about the low wage policy.
CMS understands that the market basket updates may differ from other overall inflation indexes such as the topline CPI; however, we would reiterate that these topline indexes are not comparable since they measure different mixes of products, services, or wages than the legislatively defined CMS IPPS hospital market basket. In addition, the CPI for hospital inpatient services does not reflect the input price inflation facing hospitals, and in some instances can reflect hospital charges or list prices.
We would highlight that the market basket percentage increase is a forecast of the price pressures that hospitals are expected to face in FY 2025. We also note that when developing its forecast for the ECI for hospital workers, IGI considers overall labor market conditions (including rise in contract labor employment due to tight labor market conditions) as well as trends in contract labor wages, which both have an impact on wage pressures for workers employed directly by the hospital. As projected by IGI and other independent forecasters, compensation growth and upward price pressures are expected to slow in 2025 relative to 2023 and 2024.
As is our general practice, we proposed that if more recent data became available, we would use such data, if appropriate, to derive the final FY 2025 IPPS market basket update for the final rule. We appreciate the commenters' concern regarding inflationary pressure and other rising costs and the request to use more recent data to determine the FY 2025 IPPS market basket update. For this final rule, we are using an updated forecast of the price proxies underlying the market basket that incorporates more recent historical data and reflects a revised outlook regarding the U.S. economy, including compensation and inflationary pressures.
Based on the more recent IGI second quarter 2024 forecast with historical data through the first quarter of 2024, the projected 2018-based IPPS market basket increase factor for FY 2025 is 3.4 percent, which is 0.4 percentage point higher than the projected FY 2025 market basket increase factor in the proposed rule and reflects an increase in compensation prices of 3.9 percent. We would note that the 10-year historical average (2014-2023) growth rate of the 2018-based IPPS market basket is 2.8 percent with compensation prices increasing 2.8 percent.
For these reasons, we believe that the 2018-based IPPS market basket continues to appropriately reflect IPPS cost structures, and we believe the price proxies used (such as those from BLS that reflect wage and benefit price growth) are an appropriate representation of price changes for the inputs used by hospitals in providing services. Given that we believe the 2018-based IPPS market basket reflects an index of appropriately weighted indicators of changes in wages and prices that are representative of the mix of goods and services included in such inpatient hospital services and the percentage change of the 2018-based IPPS market basket is based on IGI's more recent forecast reflecting the prospective price pressures for FY 2025, we do not believe it would be appropriate to use our exceptions and adjustment authority to create a separate payment that would have the effect of modifying the current law update.
Comment: Many commenters expressed concerns with the Employment Cost Index (ECI) used to measure changes in labor compensation in the market basket, which they state may no longer accurately capture the changing composition and cost structure of the hospital labor market given the large increases in short-term contract labor use and its growing costs. The commenters stated labor costs have increased by more than 18 percent from CY 2020 to CY 2023. They attributed this increase to expensive contract labor costs (as a result of higher utilization rates and higher costs per hour) and faster growth in salaries for employed workers (reflecting sign-on and retention bonuses). They further stated that while salaries for contract nurses have decreased some from a peak in certain geographical areas, they still remained nearly 60 percent higher at the end of FY 2023 compared to the start of FY 2020. They further stated that CMS recognizes that the ECI does not capture shifts in composition of labor, and the commenters stated that by design, the ECI is not capturing the shifts that have occurred as hospitals have had to turn to contract labor to meet patient demand. Several commenters recommended that CMS use its exceptions and adjustments authority to adopt new or supplemental data sources, to ensure labor costs are adequately reflected in the payment update in the final rule. They further requested CMS utilize supplemental data sources to evaluate the accuracy of the ECI proxy and to modify methodologies, including adopting new or supplemental data, to calculate the payment update if its analysis determines that the ECI is not adequately capturing labor costs.
Response: We believe that the ECI for wages and salaries for hospital workers is accurately reflecting the price change associated with the labor used to provide hospital care. The ECI appropriately does not reflect other factors that might affect the rate of price changes associated with labor costs, such as a shift in the occupations that may occur due to increases in case-mix or shifts in hospital purchasing decisions (for instance, to hire or to use contract labor). We believe that the ( print page 69341) prices of employed staff and contract labor are influenced by the same factors and should generally grow at similar rates. In most periods when there are not significant occupational shifts or significant shifts between employed and contract labor, the data has shown that the growth in the ECI for wages and salaries for hospital workers has generally been consistent with overall hospital wage trends. For example, our analysis of the Medicare cost report data shows from 2011 to 2019 the compound annual growth rate of both IPPS Medicare allowable salaries per hour and contract labor costs per hour was 2.5 percent, near the 2.0 percent growth rate of the ECI for wages and salaries for hospital workers over the same period (note the ECI would not reflect skill mix change whereas the salaries data would reflect these changes).
From 2019 to 2022, however, as noted by the commenters, contract labor utilization increased and IPPS Medicare allowable salaries and contract labor costs per hour increased faster than prior historical periods. We note there has likely been a shift to higher-skilled occupations for the 2019 to 2022 period associated with a 6.5-percent increase in case mix for inpatient hospital services, with notable increases of 3.8 percent in 2020 and 2.9 percent in 2021 (see table IV.A.1. of the 2024 Medicare Trustees Report [ 216 ] ); by comparison, case mix for inpatient hospital services increased 3.2 percent cumulatively from 2016 to 2019. The likely shift to more skilled occupations associated with the faster case mix increase over the last several years would also account for a portion of the difference between the growth in the ECI for wages and salaries for hospital workers and the growth in combined hospital salaries and contract labor costs per hour over this period.
For this final rule, based on the more recent IGI second quarter 2024 forecast with historical data through the first quarter of 2024, the projected 2018-based IPPS market basket increase factor for FY 2025 reflects a projected increase in compensation prices of 3.9 percent, which is 1.1 percentage points faster than the 10-year historical average (2014-2023) growth rate of compensation prices.
Comment: A commenter recommended that CMS reevaluate the data sources it uses for rebasing its market basket and calculating the annual market basket update, including labor costs. They strongly encouraged CMS to adopt new or supplemental data sources in future rulemaking that more accurately reflect the costs to hospitals, such as through use of more real time data from the hospital community. They stated that they believe that the current market basket does not account for the higher costs of contract labor, which has become more common in hospitals in an era of clinical labor shortages. A commenter requested that CMS rebase the market baskets more frequently and at least every 3 years to ensure the market basket reflects the appropriate mix of services provided to Medicare beneficiaries.
Response: We appreciate the commenter's request to rebase more frequently. Section 404 of Public Law 108-173 states the Secretary shall establish a frequency for revising the cost weights of the IPPS market basket more frequently than once every 5 years. We established a rebasing frequency of every 4 years, in part because the cost weights obtained from the Medicare cost reports typically do not indicate much of a change in the weights from year to year. The most recent rebasing of the IPPS market basket was for the FY 2022 payment update and reflected a base year of 2018 costs. We also regularly monitor the Medicare cost report data to assess whether a rebasing is technically appropriate, and we will continue to do so in the future. Based on preliminary analysis of the Medicare cost report data for IPPS hospitals for 2022 that became available for this final rule, there are small observed differences in the cost weights for 2022, as the IPPS compensation cost weight is estimated to be within roughly 1 percentage point of the 2018-based IPPS market basket compensation cost weight of 53.0 percent (and reflects a combined decrease in the salary and benefit cost weights that is larger than the increase in the contract labor cost weight). In addition, there is an estimated increase in the cost weight for home office contract labor compensation cost weight of roughly 0.5 percentage point. As stated in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36186 ), consistent with our established frequency of rebasing the IPPS market basket every 4 years, we anticipate proposing to rebase and revise the IPPS market basket in the FY 2026 IPPS/LTCH PPS proposed rule.
We believe the Medicare cost report data is the most complete, timely and relevant data source for the development of the cost weights. We also welcome information on alternative publicly available data sources.
Comment: Commenters stated that since the COVID-19 PHE, IGI has shown a consistent 3-year trend of under-forecasting the market basket growth and expressed concern this may indicate a more systematic issue with IGI's forecasting. They stated that these missed forecasts are permanently established in the standard payment rate for IPPS and will continue to compound, which they estimate to be $4 billion.
Several commenters, including many associations, urged CMS to use its special exceptions and adjustments authority under section 1886(d)(5)(I)(i) of the Act to implement a retrospective one-time adjustment for FY 2025 to account for the underestimation of the market basket updates over the last several years. Commenters recommended that CMS implement various one-time adjustments to account for underpayments in 1 or more years between FY 2021 and FY 2023 as well as for forecasted underpayments for FY 2024. The commenters stated the underestimation is, in large part, because the market basket is a time-lagged estimate that cannot fully account for unexpected changes that occur, such as historic inflation and increased labor and supply costs. They stated this is exactly what occurred at the end of the CY 2021 into CY 2022, which resulted in a large forecast error in the FY 2022 market basket update.
Several commenters noted that CMS currently implements a capital IPPS market basket forecast error adjustment as well as SNF PPS market basket forecast error adjustment policy which resulted in FY 2024 and FY 2025 SNF forecast error adjustments of 3.6 percentage points and 1.7 percentage points, respectively. They stated while CMS has not developed an analogous policy for the IPPS operating update, they believe such a forecast error adjustment to the FY 2025 IPPS operating update could be adopted under CMS' existing authority. They noted the forecast errors for FY 2021 through FY 2023 for IPPS exceeded the 0.5 percentage point threshold that is used for the SNF forecast error adjustment policy. A commenter recommended CMS establish a forecast error threshold of 1.5 percentage points, and retroactively adjust payments for that year.
Many commenters noted financial hardships, particularly in 2022 with high inflation and workforce shortages. They noted that MedPAC found that all-payment operating and overall Medicare margins both fell to record lows, estimating Medicare hospital margins for FY 2022 of negative 12.7 percent. MedPAC's FY 2024 recommendation was to increase the market basket update by one percentage point and for FY 2025 recommended that Congress increase the acute hospital market basket by 1.5 percentage points over ( print page 69342) current law. A commenter stated that, understanding the caveat that MedPAC was created specifically to advise Congress on issues impacting the Medicare program, it is disappointing that, following MedPAC's recommendation that Congress increase the IPPS market basket by an additional 1.5 percent, CMS proposed a smaller payment update than last year's 2.8 percent. Commenters further stated that margins at this level are simply unsustainable, and that hospitals in rural and underserved communities continue to close, with nine closing in FY 2023 despite a new Medicare provider type that allows them to convert to a rural emergency hospital. Commenters also stated that the missed forecasts have a significant and permanent impact on hospitals as they are permanently established in the standard payment rate for IPPS and absent action from CMS will continue to compound.
Response: While the projected IPPS hospital market basket updates have been under forecast (actual increases less forecasted increases were positive) for this most recent period, over longer periods the forecasts have generally averaged close to the historical measures (for instance, from FY 2014 through FY 2023 the cumulative forecast error was 0.0 percentage point). CMS will continue to monitor the methods associated with the market basket forecasts to ensure there are not underlying systematic issues in the forecasting approach.
We note that the under forecast of the IPPS market basket increase in the recent time period was largely due to unanticipated inflationary and labor market pressures as the economy emerged from the COVID-19 PHE. However, an analysis of the forecast error of the IPPS market basket over a longer period of time shows the forecast error has been both positive and negative. Only considering the forecast error for years when the final hospital market basket update was lower than the actual market basket update does not consider the full experience and impact of forecast error, in particular the numerous years that providers benefited from the forecast error. Relatedly, as we discussed in the FY 2024 IPPS/LTCH PPS final rule in response to similar comments ( 88 FR 59034 ), the capital IPPS and SNF PPS forecast error adjustments were adopted very early in both payment systems and, unlike what commenters are requesting here for the IPPS, forecast errors over many years have been consistently addressed within each of the Capital IPPS and SNF PPS.
For these reasons, we continue to believe it is not appropriate to include adjustments to the market basket update for future years based on the difference between the actual and forecasted market basket increase in prior years. We thank the commenters for their comments. After consideration of the comments received and consistent with our proposal, we are finalizing to use more recent data to determine the FY 2025 market basket update for the final rule. Specifically, based on more recent data available, we determined final applicable percentage increases to the standardized amount for FY 2025, as specified in the table that appears later in this section.
In the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51689 through 51692 ), we finalized our methodology for calculating and applying the productivity adjustment. As we explained in that rule, section 1886(b)(3)(B)(xi)(II) of the Act, as added by section 3401(a) of the Affordable Care Act, defines this productivity adjustment as equal to the 10-year moving average of changes in annual economy-wide, private nonfarm business MFP (as projected by the Secretary for the 10-year period ending with the applicable fiscal year, calendar year, cost reporting period, or other annual period). The U.S. Department of Labor's Bureau of Labor Statistics (BLS) publishes the official measures of private nonfarm business productivity for the U.S. economy. We note that previously the productivity measure referenced in section 1886(b)(3)(B)(xi)(II) of the Act was published by BLS as private nonfarm business multifactor productivity. Beginning with the November 18, 2021, release of productivity data, BLS replaced the term multifactor productivity (MFP) with total factor productivity (TFP). BLS noted that this is a change in terminology only and will not affect the data or methodology. As a result of the BLS name change, the productivity measure referenced in section 1886(b)(3)(B)(xi)(II) of the Act is now published by BLS as private nonfarm business total factor productivity. However, as mentioned, the data and methods are unchanged. Please see www.bls.gov for the BLS historical published TFP data. A complete description of IGI's TFP projection methodology is available on the CMS website at https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-research-and-information . In addition, we note that beginning with the FY 2022 IPPS/LTCH PPS final rule, we refer to this adjustment as the productivity adjustment rather than the MFP adjustment, to more closely track the statutory language in section 1886(b)(3)(B)(xi)(II) of the Act. We note that the adjustment continues to rely on the same underlying data and methodology.
For FY 2025, we proposed a productivity adjustment of 0.4 percent. Similar to the proposed market basket rate-of-increase, for the proposed rule, the estimate of the proposed FY 2025 productivity adjustment was based on IGI's fourth quarter 2023 forecast. As noted previously, we proposed that if more recent data subsequently became available, we would use such data, if appropriate, to determine the FY 2025 productivity adjustment for the final rule.
Comment: Several commenters expressed concerns about the application of the productivity adjustment particularly given the extreme pressures in which hospital and health systems operate. They stated the use of the private nonfarm business TFP is meant to capture gains from new technologies, economies of scale, business acumen, managerial skills and changes in productions. Thus, they stated this measure effectively assumes the hospital sector can mirror productivity gains from the private nonfarm business sector. They stated, however, in an economy marked by great uncertainty due to the PHE and labor and other productivity shocks, this assumption is significantly flawed. They further stated these assumed gains do not consider the impact of additional regulation and requirements on productivity. A commenter recommended CMS revisit the methodology for calculating the productivity adjustment or remove the measure entirely. Commenters requested CMS use its “special exceptions and adjustments” authority to eliminate the productivity adjustment for FY 2025. A commenter stated they do not understand why the productivity adjustment is higher than for FY 2024, and recommended CMS implement a productivity adjustment of no more than the 0.2 percentage point adjustment in FY 2024.
Response: Section 1886(b)(3)(B)(xi) of the Act requires the application of the productivity adjustment. As required by statute, the FY 2025 productivity adjustment is derived based on the 10-year moving average growth in economy-wide productivity for the period ending FY 2025.
As stated in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36204 ) and ( print page 69343) described previously, BLS publishes the official measures of annual economy-wide, private nonfarm business total factor productivity. IGI forecasts total factor productivity (TFP) consistent with BLS methodology by forecasting the detailed components of TFP. (As noted previously, a complete description of IGI's TFP projection methodology is available on the CMS website at https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-research-and-information .) We believe our methodology for the productivity adjustment is consistent with the statute which states the productivity adjustment is equal to the 10-year moving average of changes in annual economy-wide private nonfarm business multi-factor productivity (as projected by the Secretary for the 10-year period ending with the applicable fiscal year, year, cost reporting period, or other annual period).
The proposed FY 2025 productivity adjustment of 0.4 percent was based on IGI's forecast of the 10-year moving average of annual economy-wide private nonfarm business TFP, reflecting historical data through 2022 as published by BLS and forecasted TFP growth for 2023 through 2025. Based on more recent data available, the final FY 2025 productivity adjustment of 0.5 percent is based on IGI's forecast of the 10-year moving average of annual economy-wide private nonfarm business TFP, reflecting historical data through 2023 as published by BLS and forecasted TFP growth for 2024 through 2025. The higher productivity adjustment for FY 2025 (0.5 percent for the final rule) compared to FY 2024 (0.2 percent) is primarily a result of incorporating BLS's revised historical data through 2022 and a preliminary historical growth rate in TFP for 2023, as well as an updated forecast for TFP growth for 2024 reflecting higher expected growth in economic output.
We thank the commenters for their comments. After consideration of the comments received and consistent with our proposal, we are finalizing as proposed to use more recent data to determine the FY 2025 productivity adjustment for the final rule.
In summary, based on more recent data available for this FY 2025 IPPS/LTCH PPS final rule (that is, IGI's second quarter 2024 forecast of the 2018-based IPPS market basket rate-of-increase with historical data through the first quarter of 2024), we estimate that the FY 2025 market basket update used to determine the applicable percentage increase for the IPPS is 3.4 percent. Based on more recent data available for this FY 2025 IPPS/LTCH PPS final rule (that is, IGI's second quarter 2024 forecast of the productivity adjustment), the current estimate of the productivity adjustment for FY 2025 is 0.5 percentage point. Based on these data, we have determined four applicable percentage increases to the standardized amount for FY 2025, as specified in the following table:

In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42344 ), we revised our regulations at 42 CFR 412.64(d) to reflect the current law for the update for FY 2020 and subsequent fiscal years. Specifically, in accordance with section 1886(b)(3)(B) of the Act, we added paragraph (d)(1)(viii) to § 412.64 to set forth the applicable percentage increase to the operating standardized amount for FY 2020 and subsequent fiscal years as the percentage increase in the market basket index, subject to the reductions specified under § 412.64(d)(2) for a hospital that does not submit quality data and § 412.64(d)(3) for a hospital that is not a meaningful EHR user, less a productivity adjustment.
As discussed in section V.F. of the preamble of this final rule, section 4102 of the Consolidated Appropriations Act (CAA), 2023 ( Pub. L. 117-328 ) extended the MDH program through FY 2024 (that is, for discharges occurring on or before September 30, 2024). Subsequently, section 307 of the Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, further extended the MDH program for FY 2025 discharges occurring before January 1, 2025. Prior to enactment of the CAA, 2024, the MDH program was only to be in effect through the end of FY 2024. Under current law, the MDH program will expire for discharges on or after January 1, 2025. We refer readers to section V.F. of the preamble of this final rule for further discussion of the MDH program.
Section 1886(b)(3)(B)(iv) of the Act provides that the applicable percentage increase to the hospital-specific rates for SCHs and MDHs equals the applicable percentage increase set forth in section 1886(b)(3)(B)(i) of the Act (that is, the ( print page 69344) same update factor as for all other hospitals subject to the IPPS). Therefore, the update to the hospital-specific rates for SCHs and MDHs also is subject to section 1886(b)(3)(B)(i) of the Act, as amended by sections 3401(a) and 10319(a) of the Affordable Care Act.
For FY 2025, we proposed the following updates to the hospital-specific rates applicable to SCHs and MDHs: A proposed update of 2.6 percent for a hospital that submits quality data and is a meaningful EHR user; a proposed update of 0.35 percent for a hospital that submits quality data and is not a meaningful EHR user; a proposed update of 1.85 percent for a hospital that fails to submit quality data and is a meaningful EHR user; and a proposed update of −0.4 percent for a hospital that fails to submit quality data and is not an meaningful EHR user. As previously discussed, we proposed that if more recent data subsequently became available (for example, a more recent estimate of the market basket update and the productivity adjustment), we would use such data, if appropriate, to determine the market basket update and the productivity adjustment in the final rule.
We did not receive any public comments on our proposed updates to hospital-specific rates applicable to SCHs and MDHs. The general comments we received on the proposed FY 2025 update (including the proposed market basket update and productivity adjustment) are discussed earlier in this section. For FY 2025, we are finalizing the proposal to determine the update to the hospital specific rates for SCHs and MDHs in this final rule using the more recent available data, as previously discussed.
For this final rule, based on more recent available data, we are finalizing the following updates to the hospital specific rates applicable to SCHs and MDHs: An update of 2.9 percent for a hospital that submits quality data and is a meaningful EHR user; an update of 2.05 percent for a hospital that fails to submit quality data and is a meaningful EHR user; an update of 0.35 percent for a hospital that submits quality data and is not a meaningful EHR user; and an update of −0.5 percent for a hospital that fails to submit quality data and is not a meaningful EHR user.
Section 602 of Public Law 114-113 amended section 1886(n)(6)(B) of the Act to specify that subsection (d) Puerto Rico hospitals are eligible for incentive payments for the meaningful use of certified EHR technology, effective beginning FY 2016. In addition, section 1886(n)(6)(B) of the Act was amended to specify that the adjustments to the applicable percentage increase under section 1886(b)(3)(B)(ix) of the Act apply to subsection (d) Puerto Rico hospitals that are not meaningful EHR users, effective beginning FY 2022. Accordingly, for FY 2022, section 1886(b)(3)(B)(ix) of the Act in conjunction with section 602(d) of Public Law 114-113 requires that any subsection (d) Puerto Rico hospital that is not a meaningful EHR user as defined in section 1886(n)(3) of the Act and not subject to an exception under section 1886(b)(3)(B)(ix) of the Act will have “three-quarters” of the applicable percentage increase (prior to the application of other statutory adjustments), or three-quarters of the applicable market basket rate-of-increase, reduced by 33 1/3 percent. The reduction to three-quarters of the applicable percentage increase for subsection (d) Puerto Rico hospitals that are not meaningful EHR users increases to 66 2/3 percent for FY 2023, and, for FY 2024 and subsequent fiscal years, to 100 percent. (We note that section 1886(b)(3)(B)(viii) of the Act, which specifies the adjustment to the applicable percentage increase for “subsection (d)” hospitals that do not submit quality data under the rules established by the Secretary, is not applicable to hospitals located in Puerto Rico.) The regulations at 42 CFR 412.64(d)(3)(ii) reflect the current law for the update for subsection (d) Puerto Rico hospitals for FY 2022 and subsequent fiscal years. In the FY 2019 IPPS/LTCH PPS final rule, we finalized the payment reductions ( 83 FR 41674 ).
For FY 2025, consistent with section 1886(b)(3)(B) of the Act, as amended by section 602 of Public Law 114-113 , we are setting the applicable percentage increase for Puerto Rico hospitals by applying the following adjustments in the following sequence. Specifically, the applicable percentage increase under the IPPS for Puerto Rico hospitals will be equal to the rate of-increase in the hospital market basket for IPPS hospitals in all areas, subject to a reduction of three-quarters of the applicable percentage increase (prior to the application of other statutory adjustments; also referred to as the market basket update or rate-of-increase (with no adjustments)) for Puerto Rico hospitals not considered to be meaningful EHR users in accordance with section 1886(b)(3)(B)(ix) of the Act, and then subject to the productivity adjustment at section 1886(b)(3)(B)(xi) of the Act. As noted previously, section 1886(b)(3)(B)(xi) of the Act states that application of the productivity adjustment may result in the applicable percentage increase being less than zero.
In the FY 2025 IPPS/LTCH PPS proposed rule, based on IGI's fourth quarter 2023 forecast of the 2018-based IPPS market basket update with historical data through third quarter 2023, in accordance with section 1886(b)(3)(B) of the Act, as discussed previously, for Puerto Rico hospitals we proposed a market basket update of 3.0 percent less a productivity adjustment of 0.4 percentage point. For FY 2025, depending on whether a Puerto Rico hospital is a meaningful EHR user, there are two possible applicable percentage increases that could be applied to the standardized amount. Based on these data, we determined the following proposed applicable percentage increases to the standardized amount for FY 2025 for Puerto Rico hospitals:
- For a Puerto Rico hospital that is a meaningful EHR user, we proposed a FY 2025 applicable percentage increase to the operating standardized amount of 2.6 percent (that is, the FY 2025 estimate of the proposed market basket rate-of-increase of 3.0 percent less 0.4 percentage point for the proposed productivity adjustment).
- For a Puerto Rico hospital that is not a meaningful EHR user, we proposed a FY 2025 applicable percentage increase to the operating standardized amount of 0.35 percent (that is, the FY 2025 estimate of the proposed market basket rate-of-increase of 3.0 percent, less an adjustment of 2.25 percentage points (the proposed market basket rate-of-increase of 3.0 percent × 0.75 for failure to be a meaningful EHR user), and less 0.4 percentage point for the proposed productivity adjustment).
As noted previously, we proposed that if more recent data subsequently became available, we would use such data, if appropriate, to determine the FY 2025 market basket update and the productivity adjustment for the FY 2025 IPPS/LTCH PPS final rule. We did not receive any public comments on our proposed updates to the standardized amount for FY 2025 for Puerto Rico hospitals. The general comments we received on the proposed FY 2025 update (including the proposed market basket update and productivity adjustment) are discussed in greater detail earlier in this section. For FY 2025, we are finalizing the proposal to determine the update to the standardized amount for FY 2025 for Puerto Rico hospitals in this final rule using the more recent available data, as previously discussed. ( print page 69345)
As previously discussed in section V.A.1. of the preamble of this final rule, based on more recent data available for this final rule (that is, IGI's second quarter 2024 forecast of the 2018-based IPPS market basket rate-of-increase with historical data through the first quarter of 2024), we estimate that the FY 2025 market basket update used to determine the applicable percentage increase for the IPPS is 3.4 percent and a productivity adjustment of 0.5 percent. For FY 2025, depending on whether a Puerto Rico hospital is a meaningful EHR user, there are two possible applicable percentage increases that can be applied to the standardized amount. Based on these data, in accordance with section 1886(b)(3)(B) of the Act, we determined the following applicable percentage increases to the standardized amount for FY 2025 for Puerto Rico hospitals:
- For a Puerto Rico hospital that is a meaningful EHR user, an applicable percentage increase to the operating standardized amount of 2.9 percent (that is, the FY 2025 estimate of the market basket rate-of-increase of 3.4 percent less an adjustment of 0.5 percentage point for the productivity adjustment).
- For a Puerto Rico hospital that is not a meaningful EHR user, an applicable percentage increase to the operating standardized amount of 0.35 percent (that is, the FY 2025 estimate of the market basket rate-of-increase of 3.4 percent, less an adjustment of 2.55 percentage point (the market basket rate-of-increase of 3.4 percent × 0.75 for failure to be a meaningful EHR user), and less an adjustment of 0.5 percentage point for the productivity adjustment).

Under the authority of section 1886(d)(5)(C)(i) of the Act, the regulations at § 412.96 set forth the criteria that a hospital must meet in order to qualify under the IPPS as a rural referral center (RRC). RRCs receive special treatment under both the DSH payment adjustment and the criteria for geographic reclassification.
Section 402 of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 ( Pub. L. 108-173 ) raised the DSH payment adjustment for RRCs such that they are not subject to the 12-percent cap on DSH payments that is applicable to other rural hospitals. RRCs also are not subject to the proximity criteria when applying for geographic reclassification. In addition, they do not have to meet the requirement that a hospital's average hourly wage must exceed, by a certain percentage, the average hourly wage of the labor market area in which the hospital is located.
Section 4202(b) of the Balanced Budget Act of 1997 ( Pub. L. 105-33 ) states, in part, that any hospital classified as an RRC by the Secretary for FY 1991 shall be classified as such an RRC for FY 1998 and each subsequent fiscal year. In the August 29, 1997, IPPS final rule with comment period ( 62 FR 45999 through 46000 ), we reinstated RRC status for all hospitals that lost that status due to triennial review or MGCRB reclassification. However, we did not reinstate the status of hospitals that lost RRC status because they were now urban for all purposes because of the OMB designation of their geographic area as urban. Subsequently, in the August 1, 2000 IPPS final rule ( 65 FR 47087 ), we indicated that we were revisiting that decision. Specifically, we stated that we would permit hospitals that previously qualified as an RRC and lost their status due to OMB redesignation of the county in which they are located from rural to urban, to be reinstated as an RRC. Otherwise, a hospital seeking RRC status must satisfy all of the other applicable criteria. We use the definitions of “urban” and “rural” specified in subpart D of 42 CFR part 412 . One of the criteria under which a hospital may qualify as an RRC is to have 275 or more beds available for use (§ 412.96(b)(1)(ii)). A rural hospital that does not meet the bed size requirement can qualify as an RRC if the hospital meets two mandatory prerequisites (a minimum case-mix index (CMI) and a minimum number of discharges), and at least one of three optional criteria (relating to specialty composition of medical staff, source of inpatients, or referral volume). (We refer readers to § 412.96(c)(1) through (5) and the September 30, 1988, Federal Register ( 53 FR 38513 ) for additional discussion.) With respect to the two mandatory prerequisites, a hospital may be classified as an RRC if the hospital's—
- CMI is at least equal to the lower of the median CMI for urban hospitals in its census region, excluding hospitals with approved teaching programs, or the median CMI for all urban hospitals nationally; and
- Number of discharges is at least 5,000 per year, or, if fewer, the median number of discharges for urban hospitals in the census region in which the hospital is located. The number of discharges criterion for an osteopathic hospital is at least 3,000 discharges per year, as specified in section 1886(d)(5)(C)(i) of the Act.
In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45217 ), in light of the COVID-19 PHE, we amended the regulations at § 412.96(h)(1) to provide for the use of the best available data rather than the latest available data in calculating the national and regional CMI criteria. We also amended the regulations at § 412.96(c)(1) to indicate that the individual hospital's CMI value for discharges during the same Federal fiscal year used to compute the national and regional CMI values is used for purposes of determining whether a hospital qualifies for RRC classification. ( print page 69346) We also amended the regulations § 412.96(i)(1) and (2), which describe the methodology for calculating the number of discharges criteria, to provide for the use of the best available data rather than the latest available or most recent data when calculating the regional discharges for RRC classification.
Section 412.96(c)(1) provides that CMS establish updated national and regional CMI values in each year's annual notice of prospective payment rates for purposes of determining RRC status. The methodology we used to determine the national and regional CMI values is set forth in the regulations at § 412.96(c)(1)(ii). The national median CMI value for FY 2025 is based on the CMI values of all urban hospitals nationwide, and the regional median CMI values for FY 2025 are based on the CMI values of all urban hospitals within each census region, excluding those hospitals with approved teaching programs (that is, those hospitals that train residents in an approved GME program as provided in § 413.75). These values are based on discharges occurring during FY 2023 (October 1, 2022 through September 30, 2023), and include bills posted to CMS' records through March 2024. We believe that this is the best available data for use in calculating the national and regional median CMI values and is consistent with our use of the FY 2023 MedPAR claims data for FY 2025 ratesetting.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36206 ), we proposed that, in addition to meeting other criteria, if rural hospitals with fewer than 275 beds are to qualify for initial RRC status for cost reporting periods beginning on or after October 1, 2024, they must have a CMI value for FY 2023 that is at least—
- 1.7764 (national—all urban); or
- The median CMI value (not transfer-adjusted) for urban hospitals (excluding hospitals with approved teaching programs as identified in § 413.75) calculated by CMS for the census region in which the hospital is located. (We refer readers to the table set forth in the FY 2025 IPPS/LTCH PPS proposed rule at 89 FR 36207 ). In the proposed rule we stated that we intended to update the proposed CMI values in the FY 2025 IPPS/LTCH PPS final rule to reflect the updated FY 2023 MedPAR file, which contains data from additional bills received through March 2024.
Comment: Commenters supported our proposal to use FY 2023 data to calculate the national and regional median CMI values for FY 2025.
Therefore, based on the best available data (FY 2023 bills received through March 2024), in addition to meeting other criteria, if rural hospitals with fewer than 275 beds are to qualify for initial RRC status for cost reporting periods beginning on or after October 1, 2024, they must have a CMI value for FY 2023 that is at least:
- 1.7789 (national—all urban); or
- The median CMI value (not transfer-adjusted) for urban hospitals (excluding hospitals with approved teaching programs as identified in § 413.75) calculated by CMS for the census region in which the hospital is located.
The final CMI values by region are set forth in the following table.

A hospital seeking to qualify as an RRC should obtain its hospital-specific CMI value (not transfer-adjusted) from its MAC. Data are available on the Provider Statistical and Reimbursement (PS&R) System. In keeping with our policy on discharges, the CMI values are computed based on all Medicare patient discharges subject to the IPPS MS-DRG-based payment.
Section 412.96(c)(2)(i) provides that CMS set forth the national and regional numbers of discharges criteria in each year's annual notice of prospective payment rates for purposes of determining RRC status. As specified in section 1886(d)(5)(C)(ii) of the Act, the national standard is set at 5,000 discharges. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36207 ), we proposed to update the regional standards based on discharges for urban hospitals' cost reporting periods that began during FY 2022 (that is, October 1, 2021 through September 30, 2022). Because this is the latest available cost reporting data, we believe that this is the best available data for use in calculating the median number of discharges by region and is consistent with our finalized data proposal to use cost report data from cost reporting periods beginning during FY 2022 for FY 2025 ratesetting. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36207 ), we proposed that, in addition to meeting other criteria, a hospital, if it is to qualify for initial RRC status for cost reporting periods beginning on or after October 1, 2024, must have, as the number of discharges for its cost reporting period that began during FY 2022, at least—
- 5,000 (3,000 for an osteopathic hospital); or
- If less, the median number of discharges for urban hospitals in the census region in which the hospital is located. (We refer readers to the table set forth in the FY 2025 IPPS/LTCH PPS proposed rule at 89 FR 36207 ). In the proposed rule, we stated that we ( print page 69347) intended to update these numbers in the FY 2025 final rule based on the latest available cost report data.
Comment: Commenters supported our proposal to use FY 2022 data to calculate median number of discharges by region for FY 2025.
Therefore, based on the best available discharge data at this time, that is, for cost reporting periods that began during FY 2022, the final median number of discharges for urban hospitals by census region are set forth in the following table.

We note that because the median number of discharges for hospitals in each census region is greater than the national standard of 5,000 discharges, under this final rule, 5,000 discharges is the minimum criterion for all hospitals, except for osteopathic hospitals for which the minimum criterion is 3,000 discharges.
Section 1886(d)(5)(C) of the Act sets forth certain criteria that must be met for a hospital to be classified as a rural referral center, including a discharge criterion specifying the hospital has at least 5,000 discharges a year or, if less, the median number of discharges in urban hospitals in the region in which the hospital is located. Section 9106 of the Consolidated Omnibus Budget Reconciliation Act of 1985 (Pub. L. 99-272) amended section 1886(d)(5)(C) of the Act to provide for a separate discharge criterion for an osteopathic hospital to qualify for classification as a rural referral center, effective for cost reporting periods beginning on or after January 1, 1986. To implement this statutory provision, in the FY 1987 IPPS final rule, we revised 42 CFR 412.96(c)(2) to specify that for cost reporting periods beginning on or after January 1, 1986 an osteopathic hospital, recognized by the American Osteopathic Hospital Association, that is located in a rural area must have at least 3,000 discharges during its most recently completed cost reporting period to meet the number of discharges criterion ( 51 FR 31471 ). In the FY 1996 IPPS final rule, in light of a name change of the American Osteopathic Hospital Association to the American Osteopathic Healthcare Association, we subsequently revised 42 CFR 412.96(c)(2) to specify that the osteopathic hospital must be recognized by the American Osteopathic Healthcare Association “(or any successor organization)” ( 60 FR 45810 ).
As we discussed in implementing the number of discharges criterion for osteopathic hospitals in the FY 1987 IPPS final rule, “[b]ecause section 1886(d)(5)(C)(i) of the Act specifically limits this qualification to osteopathic hospitals, we do not believe that this standard should apply to all hospitals” ( 51 FR 31473 ). Accordingly, to qualify under this lower number of discharges criterion, a hospital must be an osteopathic hospital. It has come to the attention of CMS that the successor organization to the American Osteopathic Healthcare Association, namely the Accreditation Commission for Health Care, accredits acute care hospitals, including hospitals that are not osteopathic. Thus, a hospital receiving an accreditation letter or certificate from the successor organization is not necessarily an osteopathic hospital. Therefore, we proposed to revise the regulations at 42 CFR 412.96(c)(2) to clarify that, to qualify for RRC classification based on the lower discharge criterion for osteopathic hospitals, a hospital must be an osteopathic hospital and by itself recognition (such as an accreditation letter) by a successor organization to the American Osteopathic Healthcare Association is not necessarily sufficient to demonstrate that a hospital is an osteopathic hospital.
We proposed to amend our regulations at 42 CFR 412.96 by revising paragraph (c)(2)(ii) as follows: “(ii) For cost reporting periods beginning on or after January 1, 1986, an osteopathic hospital, recognized by the American Osteopathic Healthcare Association (or any successor organization), that is located in a rural area must have at least 3,000 discharges during its cost reporting period that began during the same fiscal year as the cost reporting periods used to compute the regional median discharges under paragraph (i) of this section to meet the number of discharges criterion. A hospital applying for rural referral center status under the number of discharges criterion in this paragraph must demonstrate its status as an osteopathic hospital.” Consistent with section 1886(d)(5)(C)(i) of the Act, evidence of osteopathic status may include, but is not limited to, the hospital's scope of services and its mix of medical specialties. CMS will consider the totality of the information demonstrating whether an applicant hospital is an osteopathic hospital. We sought comment on additional types of evidence we should consider in the determination of a hospital's osteopathic status.
Comment: We received one comment on our proposed revisions to the regulations at 42 CFR 412.96(c)(2) . The commenter requested that CMS consider more definitive measures of determining osteopathic status but cautioned that determination of a hospital's osteopathic status on the basis of offering osteopathic services or having osteopathic doctors on staff presents threshold related challenges. CMS did not receive any specific recommendations regarding the appropriate scope of services, mix of medical specialties, or any other ( print page 69348) criterion for determining osteopathic status of a hospital applying for rural referral status under the reduced discharge criterion.
Response: We thank the commenter for their feedback on our proposed revisions to the regulation text. CMS may consider further refinements in future rulemaking, such as more definitive measures, as we gain further experience with the types of evidence used by applicant hospitals to demonstrate their osteopathic status.
After consideration of the comment received, we are finalizing our updates to the regulation text as proposed. CMS will determine osteopathic status of a hospital applying for rural referral status according to the totality of the information submitted.
Section 1886(d)(12) of the Act provides for an additional payment to each qualifying low-volume hospital under the IPPS beginning in FY 2005. The low-volume hospital payment adjustment is implemented in the regulations at 42 CFR 412.101 . The additional payment adjustment to a low-volume hospital provided for under section 1886(d)(12) of the Act is in addition to any payment calculated under section 1886 of the Act and is based on the per discharge amount paid to the qualifying hospital. In other words, the low-volume hospital payment adjustment is based on total per discharge payments made under section 1886 of the Act, including capital, DSH, IME, and outlier payments. For SCHs and MDHs, the low-volume hospital payment adjustment is based in part on either the Federal rate or the hospital-specific rate, whichever results in a greater operating IPPS payment. The payment adjustment for low-volume hospitals is not budget neutral.
As discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59041 through 59045 ), section 4101 of the CAA, 2023 ( Pub. L. 117-328 ) extended through FY 2024 the modified definition of a low-volume hospital and the methodology for calculating the payment adjustment for low-volume hospitals in effect for FYs 2019 through 2022. The Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, extended the temporary changes to the low-volume hospital qualifying criteria and payment adjustment under the IPPS for a portion of FY 2025. Specifically, section 306 of the CAA, 2024 further extended the modified definition of low-volume hospital and the methodology for calculating the payment adjustment for low-volume hospitals under section 1886(d)(12) through December 31, 2024. Beginning January 1, 2025, the low-volume hospital qualifying criteria and payment adjustment will revert to the statutory requirements that were in effect prior to FY 2011, and the preexisting low-volume hospital payment adjustment methodology and qualifying criteria, as implemented in FY 2005 and discussed later in this section, will resume. We discuss our proposals for the payment policies for FY 2025, which we are finalizing as proposed after consideration of public comments, in section V.E.2. of the preamble of this final rule.

As discussed previously, section 4101 of the CAA, 2023 modified the definition of low-volume hospital and the methodology for calculating the payment adjustment for low-volume hospitals under section 1886(d)(12) of the Act through September 30, 2024. Prior to the enactment of the CAA, 2024 ( Pub. L. 118-42 ), the temporary changes to the low-volume hospital qualifying criteria and payment adjustment provided by section 4101 of CAA, 2023 were set to expire on October 1, 2024. Section 306 of the CAA, 2024 extends the temporary changes to the low-volume hospital qualifying criteria and payment adjustment under the IPPS for the portion of FY 2025 beginning on October 1, 2024, and ending on December 31, 2024 (that is, for discharges occurring before January 1, 2025).
Under section 1886(d)(12)(C)(i) of the Act, as amended by Public Law 118-42 , for FYs 2019 through 2024 and the portion of FY 2025 occurring before January 1, 2025, a subsection (d) hospital qualifies as a low-volume hospital if it is more than 15 road miles from another subsection (d) hospital and has less than 3,800 total discharges during the fiscal year. In accordance with the existing regulations at § 412.101(a), we define the term “road miles” to mean “miles” as defined at § 412.92(c)(1). Under section 1886(d)(12)(D) of the Act, as amended, for discharges occurring in FY 2019 through December 31, 2024, the Secretary determines the applicable percentage increase using a continuous, linear sliding scale ranging from an additional 25 percent payment adjustment for low-volume hospitals with 500 or fewer discharges to a zero percent additional payment for low volume hospitals with more than 3,800 discharges in the fiscal year. Consistent with the requirements of section 1886(d)(12)(C)(ii) of the Act, the term “discharge” for purposes of these provisions refers to total discharges, regardless of payer (that is, Medicare and non-Medicare discharges).
In the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41399 ), we specified a continuous, linear sliding scale formula to determine the low volume payment adjustment, as reflected in the regulations at § 412.101(c)(3)(ii). Consistent with the statute, we provided ( print page 69349) that qualifying hospitals with 500 or fewer total discharges will receive a low-volume hospital payment adjustment of 25. For qualifying hospitals with fewer than 3,800 discharges but more than 500 discharges, the low-volume payment adjustment is calculated by subtracting from 25 percent the proportion of payments associated with the discharges in excess of 500. For qualifying hospitals with fewer than 3,800 total discharges but more than 500 total discharges, the low-volume hospital payment adjustment is calculated using the formula at § 412.101(c)(3)(ii) (which is shown in the Table V.E.-01). For this purpose, the term “discharge” refers to total discharges, regardless of payer (that is, Medicare and non-Medicare discharges). The hospital's most recently submitted cost report is used to determine if the hospital meets the discharge criterion to receive the low volume payment adjustment in the current year (§ 412.101(b)(2)(iii)). The low-volume hospital payment adjustment for FYs 2019 through 2024 is set forth in the regulations at § 412.101(c)(3).
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36209 ), consistent with the extension of the methodology for calculating the payment adjustment for low-volume hospitals through FY 2024, we proposed to continue using the previously specified continuous, linear sliding scale formula to determine the low-volume hospital payment adjustment for the portion of FY 2025 occurring before January 1, 2025. We also proposed to make conforming changes to the regulation text in § 412.101 to reflect the extensions of the changes to the qualifying criteria and the payment adjustment methodology for low-volume hospitals in accordance with provisions of the CAA, 2024. Specifically, we proposed to make conforming changes to paragraphs (b)(2)(iii) and (c)(3) introductory text of § 412.101 to reflect that the low-volume hospital payment adjustment policy in effect for the portion of FY 2025 through December 31, 2024, is the same low-volume hospital payment adjustment policy in effect for FYs 2019 through 2024 (as described in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41398 through 41399 ) and in the FY 2024 IPPS/LTCH final rule ( 88 FR 59041 through 59045 )). In addition, in accordance with the provisions of the CAA, 2024, we proposed to make conforming changes to paragraphs (b)(2)(i) and (c)(1) of § 412.101 to reflect that for the portion of FY 2025 beginning on January 1, 2025 and for subsequent fiscal years, the low-volume hospital payment adjustment policy will revert back to the low-volume hospital payment adjustment policy in effect for FYs 2005 through 2010, as described in section V.E.3. of the preamble of this final rule. We further proposed that if the temporary changes to the low-volume payment adjustment were extended through legislation beyond December 31, 2024, we would make the conforming changes to the regulations at § 412.101(b)(2)(i), (b)(2)(iii), (c)(1), and (c)(3) to reflect any further extension.
Comment: Commenters supported the legislative extension of the temporary changes to the definition and payment adjustment for low-volume hospitals through December 31, 2024, and expressed support for additional legislative extensions. Many commenters requested that CMS collaborate with Congress to extend or make permanent the temporary modifications to the low-volume hospital payment policy. A commenter asked CMS to clarify how it would handle any legislation that that would provide a continuation of the modified low-volume hospital payment policy beyond the end of the year. Another commenter urged CMS to expeditiously process claims and provide instructions to MACs for any subsequent extensions, especially in instances when extensions are made retroactively.
Response: We appreciate the commenters sharing their support for legislative extension. As we have said in the past, we make every effort to implement any extension of the low-volume hospital payment policy as expeditiously as possible, however we believe it would be premature to opine on exactly how any subsequent extension would be implemented. As with past extensions, we would continue work to implement any subsequent extensions as quickly and seamlessly as possible based on the s specific legislative requirements of the particular extension.
After consideration of the public comments we received regarding the temporary changes to the qualifying criteria and the payment adjustment methodology for low-volume hospitals through December 31, 2024, we are finalizing our proposals on the extension of these changes without modification, including our proposal to codify these extensions in the regulation text at § 412.101 without modification.
In accordance with section 1886(d)(12) of the Act, as amended by section 306 of the CAA, 2024, beginning with FY 2025 discharges occurring on or after January 1, 2025, the low-volume hospital definition and payment adjustment methodology will revert to the statutory requirements that were in effect prior to the amendments made by the Affordable Care Act and subsequent legislation. Specifically, section 1886(d)(12)(B) of the Act requires, for discharges occurring in FYs 2005 through 2010, FY 2025 discharges occurring on or after January 1, 2025 and subsequent years, that the Secretary determine an applicable percentage increase for these low-volume hospitals based on the “empirical relationship” between the standardized cost-per-case for such hospitals and the total number of discharges of such hospitals and the amount of the additional incremental costs (if any) that are associated with such number of discharges. The statute thus mandates that the Secretary develop an empirically justifiable adjustment based on the relationship between costs and discharges for these low-volume hospitals.
Therefore, effective for the portion of FY 2025 beginning on January 1, 2025 and subsequent years, under current policy at § 412.101(b), to qualify as a low-volume hospital, a subsection (d) hospital must be more than 25 road miles from another subsection (d) hospital and have less than 200 discharges (that is, less than 200 discharges total, including both Medicare and non-Medicare discharges) during the fiscal year. For the portion of FY 2025 beginning on January 1, 2025, and subsequent years, the statute specifies that a low-volume hospital must have less than 800 discharges during the fiscal year. However, as required by section 1886(d)(12)(B)(i) of the Act, the Secretary has developed an empirically justifiable payment adjustment based on the relationship, for IPPS hospitals with less than 800 discharges, between the additional incremental costs (if any) that are associated with a particular number of discharges. Based on an analysis we conducted for the FY 2005 IPPS final rule ( 69 FR 49099 through 49102 ), a 25-percent low-volume adjustment to all qualifying hospitals with less than 200 discharges was found to be most consistent with the statutory requirement to provide relief for low-volume hospitals where there is empirical evidence that higher incremental costs are associated with low numbers of total discharges. (Under the policy we established in that same final rule, hospitals with between 200 ( print page 69350) and 799 discharges do not receive a low-volume hospital adjustment.)
As discussed previously, for FYs 2005 through 2010 and FY 2019 and subsequent years, the discharge determination is made based on the hospital's number of total discharges, that is, Medicare and non-Medicare discharges. The hospital's most recently submitted cost report is used to determine if the hospital meets the discharge criterion to receive the low-volume payment adjustment in the current year (§ 412.101(b)(2)(i)). We use cost report data to determine if a hospital meets the discharge criterion because this is the best available data source that includes information on both Medicare and non-Medicare discharges. We note that, for FYs 2011 through 2018, we used the most recently available MedPAR data to determine the hospital's Medicare discharges because only Medicare discharges were used to determine if a hospital met the discharge criterion for those years.
In addition to the discharge criterion, a hospital must also meet the mileage criterion to qualify for the low-volume payment adjustment. As specified by section 1886(d)(12)(C)(i) of the Act, a low-volume hospital must be more than 25 road miles (or 15 road miles for FYs 2011 through 2024) from another subsection (d) hospital. Accordingly, for FY 2025 and subsequent fiscal years, in addition to the discharge criterion, the eligibility for the low-volume payment adjustment is also dependent upon the hospital meeting the mileage criterion at § 412.101(b)(2)(i), which specifies that a hospital must be located more than 25 road miles from the nearest subsection (d) hospital, consistent with section 1886(d)(12)(C)(i) of the Act. We define, at § 412.101(a), the term “road miles” to mean “miles” as defined at § 412.92(c)(1) ( 75 FR 50238 through 50275 and 50414 ). As previously noted, we proposed to make conforming changes to paragraphs (b)(2)(i) and (c)(1) of § 412.101 to reflect that for the portion of FY 2025 beginning on January 1, 2025, and subsequent fiscal years, the low-volume hospital payment adjustment policy is the same as that in effect for FYs 2005 through 2010.
On average, approximately 600 hospitals per year were eligible for the low-volume hospital payment adjustment for FYs 2019 through 2024 under the temporary changes in the low-volume hospital payment policy as amended by section 50204 of the Bipartisan Budget Act of 2018 ( Pub. L. 115-123 ), and section 4101 of the Consolidated Appropriations Act, 2023 (CAA, 2023) ( Pub. L. 117-328 ). As discussed previously, the CAA, 2024 further extended the modified definition of low-volume hospital and the methodology for calculating the payment adjustment for low-volume hospitals under section 1886(d)(12) through December 31, 2024. Therefore, for the portion of FY 2025 beginning on January 1, 2025 and for subsequent years the low-volume hospital qualifying criteria and payment adjustment will revert to the statutory requirements that were in effect prior to FY 2011. Based on historical data for hospitals that qualified during FYs 2005-2010, we estimate that fewer than 10 hospitals would qualify for the low-volume hospital payment adjustment for the portion of FY 2025 beginning on January 1, 2025 under current law.
Comment: Many commenters urged CMS to collaborate with Congress to make permanent the modifications to the low-volume hospital payment policy. Some commenters requested CMS continue the temporary changes to the definition and the methodology for calculating the payment adjustment for low-volume hospitals for the portion of FY 2025 beginning on January 1, 2025 and subsequent years. Commenters stated that not continuing these temporary changes would result in significant reductions in payment that could impede the services hospitals, including those in rural communities, provide in the communities they serve.
Response: We appreciate the feedback from commenters on continuation of the enhanced low-volume hospital payment policy for the portion of FY 2025 beginning on January 1, 2025 and subsequent years. As previously discussed, the statute only extends those temporary changes to the low-volume hospital policy through December 31, 2024. Therefore, in absence of subsequent legislation, beginning on January 1, 2025, the low-volume hospital qualifying criteria and the amount of the payment adjustment to such hospitals will revert back to those policies that were in effect prior to the amendments made by recent legislation.
Comment: For the portion of FY 2025 beginning on January 1, 2025 and subsequent years, several commenters requested expanding low-volume hospital payment adjustment eligibility criteria to include hospitals with 200-799 discharges as provided by the statute. A commenter stated that under the originally established low-volume hospital adjustment policy only a small number of hospitals would qualify to receive the adjustment under the low-volume hospital payment policy beginning January 1, 2025. The impact, the commenter argued, would make nearly all rural hospitals ineligible to receive the low-volume hospital payment adjustment incurring a loss of several million dollars annually. The commenter stated that even if the low-volume hospital discharge criteria were expanded to less than 800 total discharges, more rural hospitals would qualify for low-volume payment adjustment which will help those communities maintain access to care.
Response: As previously discussed, as required by section 1886(d)(12)(B)(i) of the Act, we developed an empirically justifiable payment adjustment based on the relationship, for IPPS hospitals with less than 800 discharges, between the additional incremental costs (if any) that are associated with a particular number of discharges. Based on our analysis, a 25-percent low-volume adjustment to all qualifying hospitals with less than 200 discharges was found to be most consistent with the statutory requirement to provide relief for low-volume hospitals where there is empirical evidence that higher incremental costs are associated with low numbers of total discharges ( 69 FR 49099 through 49102 ). In the future, we may reevaluate the low-volume hospital adjustment policy; that is, the definition of a low-volume hospital and the payment adjustment. However, we are not aware of any analysis or empirical evidence that would support expanding the originally established low-volume hospital adjustment policy. We further note that we did not make any proposals regarding the low-volume hospital payment adjustment for the portion of FY 2025 beginning on January 1, 2025 and subsequent years.
After consideration of the public comments we received, we are finalizing our proposals, without modification. Consistent with current law, effective beginning with the portion of FY 2025 beginning on January 1, 2025, the low-volume hospital definition and payment adjustment methodology will revert to the policy established under statutory requirements that were in effect prior to the amendments made by the Affordable Care Act and extended through subsequent legislation.
In the FY 2011 IPPS/LTCH PPS final rule ( 75 FR 50238 through 50275 and 50414 ) and subsequent rulemaking, most recently in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59044 through 59045 ), we discussed the process for requesting and obtaining the low-volume hospital payment ( print page 69351) adjustment. Under this previously established process, a hospital makes a written request for the low-volume payment adjustment under § 412.101 to its MAC. This request must contain sufficient documentation to establish that the hospital meets the applicable mileage and discharge criteria. The MAC will determine if the hospital qualifies as a low-volume hospital by reviewing the data the hospital submits with its request for low-volume hospital status in addition to other available data. Under this approach, a hospital will know in advance whether or not it will receive a payment adjustment under the low-volume hospital policy. The MAC and CMS may review available data such as the number of discharges, in addition to the data the hospital submits with its request for low-volume hospital status, to determine whether or not the hospital meets the qualifying criteria. (For additional information on our existing process for requesting the low-volume hospital payment adjustment, we refer readers to the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41399 through 41401 ).)
As explained earlier, for FY 2019 and subsequent fiscal years, the discharge determination is made based on the hospital's number of total discharges, that is, Medicare and non-Medicare discharges, as was the case for FYs 2005 through 2010. Under § 412.101(b)(2)(i) and (iii), a hospital's most recently submitted cost report is used to determine if the hospital meets the discharge criterion to receive the low-volume payment adjustment in the current year. As discussed in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41399 and 41400 ), we use cost report data to determine if a hospital meets the discharge criterion because this is the best available data source that includes information on both Medicare and non-Medicare discharges. (For FYs 2011 through 2018, the most recently available MedPAR data were used to determine the hospital's Medicare discharges because non-Medicare discharges were not used to determine if a hospital met the discharge criterion for those years.) Therefore, a hospital must refer to its most recently submitted cost report for total discharges (Medicare and non-Medicare) to decide whether or not to apply for low-volume hospital status for a particular fiscal year.
In addition to the discharge criterion, eligibility for the low-volume hospital payment adjustment is also dependent upon the hospital meeting the applicable mileage criterion specified in section 1886(d)(12)(C)(i) of the Act, which is codified at § 412.101(b)(2), for the fiscal year. Specifically, to meet the mileage criterion to qualify for the low-volume hospital payment adjustment for the portion of FY 2025 beginning October 1, 2024 through December 31, 2024, a hospital must be located more than 15 road miles from the nearest subsection (d) hospital, as reflected in proposed revised § 412.101(b)(2). Additionally, to meet the mileage criterion to qualify for the low-volume hospital payment adjustment for the portion of FY 2025 beginning January 1, 2025 through September 30, 2025, a hospital must be located more than 25 road miles from the nearest subsection (d) hospital. (We define in § 412.101(a) the term “road miles” to mean “miles” as defined in § 412.92(c)(1) ( 75 FR 50238 through 50275 and 50414 ).) For establishing that the hospital meets the mileage criterion, the use of a web-based mapping tool as part of the documentation is acceptable. The MAC will determine if the information submitted by the hospital, such as the name and street address of the nearest hospital(s), location on a map, and distance from the hospital requesting low-volume hospital status, is sufficient to document that it meets the mileage criterion. If not, the MAC will follow up with the hospital to obtain additional necessary information to determine whether or not the hospital meets the applicable mileage criterion.
In accordance with our previously established process, a hospital must make a written request for low-volume hospital status that is received by its MAC by September 1 immediately preceding the start of the Federal fiscal year for which the hospital is applying for low-volume hospital status in order for the applicable low-volume hospital payment adjustment to be applied to payments for its discharges for the fiscal year beginning on or after October 1 immediately following the request (that is, the start of the Federal fiscal year). For a hospital whose request for low-volume hospital status is received after September 1, if the MAC determines the hospital meets the criteria to qualify as a low-volume hospital, the MAC will apply the applicable low-volume hospital payment adjustment to determine payment for the hospital's discharges for the fiscal year, effective prospectively within 30 days of the date of the MAC's low-volume status determination.
Consistent with this previously established process, for FY 2025, we proposed that a hospital must submit a written request for low-volume hospital status to its MAC that includes sufficient documentation to establish that the hospital meets the applicable mileage and discharge criteria (as described earlier). Specifically, for the portion of FY 2025 beginning October 1, 2024 through December 31, 2024, a hospital must make a written request for low-volume hospital status that is received by its MAC no later than September 1, 2024, in order for the low-volume, add-on payment adjustment to be applied to payments for its discharges beginning on or after October 1, 2024. If a hospital's written request for low-volume hospital status for the portion of FY 2025 beginning October 1, 2024 through December 31, 2024 is received after September 1, 2024, and if the MAC determines the hospital meets the criteria to qualify as a low-volume hospital, the MAC would apply the low-volume hospital payment adjustment to determine the payment for the hospital's FY 2025 discharges prior to January 1, 2025, effective prospectively within 30 days of the date of the MAC's low-volume hospital status determination.
Additionally, we proposed that a hospital must also submit a written request for low-volume hospital status to its MAC that includes sufficient documentation to establish that the hospital continues to meet the applicable mileage and discharge criteria for the portion of FY 2025 beginning on January 1, 2025 through September 30, 2025 (as described earlier). Specifically, for the portion of FY 2025 beginning on January 1, 2025, a hospital must make a written request for low-volume hospital status that is received by its MAC no later than December 1, 2024, in order for the 25-percent, low-volume, add-on payment adjustment to be applied to payments for its discharges beginning on or after January 1, 2025. If a hospital's written request for low-volume hospital status for the portion of FY 2025 beginning on January 1, 2025 is received after December 1, 2024, and if the MAC determines the hospital meets the criteria to qualify as a low-volume hospital, the MAC would apply the low-volume hospital payment adjustment to determine the payment for the hospital's FY 2025 discharges on or after January 1, 2025, effective prospectively within 30 days of the date of the MAC's low-volume hospital status determination.
A hospital may choose to make a single written request for low-volume hospital status to its MAC for both the portion of FY 2025 beginning on October 1, 2024, and ending December 31, 2024, and the portion of FY 2025 beginning on January 1, 2025, through September 30, 2025, by the September 1, 2024, deadline discussed previously. Alternatively, a hospital may choose to submit separate written requests, one for ( print page 69352) the portion of FY 2025 beginning on October 1, 2024, and ending on December 31, 2024 (by the September 1, 2024, deadline discussed previously), and another for the portion of FY 2025 beginning on January 1, 2025, through September 30, 2025 (by the December 1, 2024 deadline discussed previously).
Under this process, a hospital that qualified for the low-volume hospital payment adjustment for FY 2024 may continue to receive a low-volume hospital payment adjustment for FY 2025 without reapplying if it meets both the discharge criterion and the mileage criterion applicable for FY 2025 (that is, the discharge criterion and mileage criterion for the period beginning October 1, 2024 through December 31, 2024, as well as the discharge criterion and mileage criterion for the period beginning on January 1, 2025 through September 30, 2025, respectively). As discussed previously, for the portion of FY 2025 beginning on January 1, 2025, the discharge and the mileage criteria are reverting to the statutory requirements that were in effect prior to FY 2011, and to the preexisting low-volume hospital qualifying criteria, as implemented in FY 2005 and specified in the existing regulations at § 412.101(b)(2)(i). As in previous years, we proposed that such a hospital must send written verification that is received by its MAC no later than September 1, 2024 or December 1, 2024, respectively, stating that it meets the mileage criterion for the applicable portion(s) of FY 2025, as described previously. For example, for the portion of FY 2025 beginning October 1, 2024 through December 31, 2024, the hospital must state it is located more than 15 road miles from the nearest “subsection (d)” hospital. Similarly, for the portion of FY 2025 beginning on January 1, 2025, the hospital must state it is located more than 25 road miles from the nearest “subsection (d)” hospital. For FY 2025, we further proposed that this written verification must also state, based upon the most recently submitted cost report, that the hospital meets the discharge criterion for the applicable portion(s) of FY 2025, as described previously. For example, for the portion of FY 2025 beginning October 1, 2024 through December 31, 2024, the hospital must have less than 3,800 discharges total, including both Medicare and non-Medicare discharges. Similarly, for the portion of FY 2025 beginning on January 1, 2025, the hospital must have less than 200 discharges total, including both Medicare and non-Medicare discharges. If a hospital's request for low-volume hospital status for FY 2025 is received after September 1, 2024, (or after December 1, 2024 for the portion of FY 2025 beginning on January 1, 2025) and if the MAC determines the hospital meets the criteria to qualify as a low-volume hospital, the MAC will apply the applicable low-volume add-on payment adjustment to determine the payment for the hospital's discharges for the applicable portion(s) of FY 2025, effective prospectively within 30 days of the date of the MAC's low-volume hospital status determination.
We did not receive any comments on our process for requesting and obtaining the low-volume payment adjustment for the portion of FY 2025 beginning October 1, 2024 through December 31, 2024 or the portion of FY 2025 beginning on January 1, 2025. Therefore, we are finalizing our proposals, without modification.
Section 1886(d)(5)(G) of the Act provides special payment protections, under the IPPS, to a Medicare-dependent, small rural hospital (MDH). (For additional information on the MDH program and the payment methodology, we refer readers to the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51683 through 51684 ).) As discussed in section V.B. of the preamble of this final rule, section 307 of the Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, extended the MDH program for FY 2025 discharges occurring before January 1, 2025. Prior to enactment of the CAA, 2024, the MDH program was only to be in effect through the end of FY 2024. Under current law, the MDH program provisions at section 1886(d)(5)(G) of the Act will expire for discharges on or after January 1, 2025. Beginning with discharges occurring on or after January 1, 2025, all hospitals that previously qualified for MDH status will be paid based on the Federal rate.
Since the extension of the MDH program through FY 2012 provided by section 3124 of the Affordable Care Act, the MDH program had been extended by subsequent legislation as follows: section 606 of the American Taxpayer Relief Act ( Pub. L. 112-240 ) extended the MDH program through FY 2013 (that is, for discharges occurring before October 1, 2013). Section 1106 of the Pathway for SGR Reform Act of 2013 ( Pub. L. 113-67 ) extended the MDH program through the first half of FY 2014 (that is, for discharges occurring before April 1, 2014). Section 106 of the Protecting Access to Medicare Act ( Pub. L. 113-93 ) extended the MDH program through the first half of FY 2015 (that is, for discharges occurring before April 1, 2015). Section 205 of the MACRA ( Pub. L. 114-10 ) extended the MDH program through FY 2017 (that is, for discharges occurring before October 1, 2017). Section 50205 of the Bipartisan Budget Act ( Pub. L. 115-123 ) extended the MDH program through FY 2022 (that is for discharges occurring before October 1, 2022). Section 102 of the Continuing Appropriations and Ukraine Supplemental Appropriations Act, 2023 ( Pub. L. 117-180 ) extended the MDH program through December 16, 2022. Section 102 of the Further Continuing Appropriations and Extensions Act, 2023 ( Pub. L. 117-229 ) extended the MDH program through December 23, 2022. Section 4102 of the Consolidated Appropriations Act, 2023 ( Pub. L. 117-328 ) extended the MDH program through FY 2024 (that is for discharges occurring before October 1, 2024). Lastly, under current law, section 307 of the CAA, 2024 ( Pub. L. 118-42 ) extended the MDH program through December 31, 2024 (that is, for discharges occurring before January 1, 2025).
For additional information on the extensions of the MDH program after FY 2012, we refer readers to the following Federal Register documents: The FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53404 through 53405 and 53413 through 53414 ); the FY 2013 IPPS notice ( 78 FR 14689 ); the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50647 through 50649 ); the FY 2014 interim final rule with comment period ( 79 FR 15025 through 15027 ); the FY 2014 notice ( 79 FR 34446 through 34449 ); the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50022 through 50024 ); the August 2015 interim final rule with comment period ( 80 FR 49596 ); the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57054 through 57057 ); the FY 2018 notice ( 83 FR 18303 through 18305 ); the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41429 ); and the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59045 ).
Prior to the enactment of Public Law 118-42 , under section 4102 of Public Law 117-328 , the MDH program authorized by section 1886(d)(5)(G) of the Act was set to expire at the end of FY 2024. Section 307 of Public Law 118-42 amended sections 1886(d)(5)(G)(i) and 1886(d)(5)(G)(ii)(II) of the Act by striking “October 1, 2024” and inserting “January 1, 2025”. Section 307 of Public Law 118-42 also made ( print page 69353) conforming amendments to sections 1886(b)(3)(D)(i) and 1886(b)(3)(D)(iv) of the Act.
Therefore, in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36212 ), we proposed to make conforming changes to the regulations governing the MDH program at § 412.108(a)(1) and (c)(2)(iii) and the general payment rules at § 412.90(j) to reflect the extension of the MDH program through December 31, 2024.
As a result of the extension of the MDH program through December 31, 2024 as provided by section 307 of Public Law 118-42 , a provider that is classified as an MDH as of September 30, 2024, will continue to be classified as an MDH as of October 1, 2024, with no need to reapply for MDH classification.
Because section 307 of the CAA, 2024 extended the MDH program through December 31, 2024 only, beginning January 1, 2025, the MDH program will no longer be in effect. Since the MDH program is not authorized by statute beyond December 31, 2024, beginning January 1, 2025, all hospitals that previously qualified for MDH status under section 1886(d)(5)(G) of the Act will no longer have MDH status and will be paid based on the IPPS Federal rate. There are currently 173 MDHs, of which we estimate 117 would have been paid under the blended payment of the Federal rate and hospital-specific rate while the remaining 56 would have been paid based on the IPPS Federal rate. With the expiration of the MDH program, all these providers will all be paid based on the IPPS Federal rate beginning with discharges occurring on or after January 1, 2025.
When the MDH program was set to expire at the end of FY 2012, in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53404 through 53405 ), we revised our sole community hospital (SCH) policies to allow MDHs to apply for SCH status in advance of the expiration of the MDH program and be paid as such under certain conditions. We codified these changes in the regulations at § 412.92(b)(2)(i) and (b)(2)(v). Specifically, the existing regulations at § 412.92(b)(2)(i) and (b)(2)(v) allow for an effective date of an approval of SCH status that is the day following the expiration date of the MDH program. We note that these same conditions apply to MDHs that intend to apply for SCH status with the expiration of the MDH program on December 31, 2024. Therefore, in order for an MDH to receive SCH status effective January 1, 2025, the MDH must apply for SCH status at least 30 days before the expiration of the MDH program; that is, the MDH must apply for SCH status by December 2, 2024. The MDH also must request that, if approved as an SCH, the SCH status be effective with the expiration of the MDH program; that is, the MDH must request that the SCH status, if approved, be effective January 1, 2025, immediately after its MDH status expires with the expiration of the MDH program on December 31, 2024. We emphasize that an MDH that applies for SCH status in anticipation of the expiration of the MDH program would not qualify for the January 1, 2025 effective date for SCH status if it does not apply by the December 2, 2024 deadline. If the MDH does not apply by the December 2, 2024 deadline, the hospital would instead be subject to the usual effective date for SCH classification as specified at § 412.92(b)(2)(i); that is, as of the date the MAC receives the complete application from the provider.
In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to make conforming changes to the regulations governing the MDH program at § 412.108(a)(1) and (c)(2)(iii) and the general payment rules at § 412.90(j) to reflect the extension of the MDH program through December 31, 2024. We further proposed that if the MDH program were to be extended by law beyond December 31, 2024, similar to how it was extended by prior legislation as described previously, we would, depending on timing of such legislation in relation to the final rule, modify our proposed conforming changes to the regulations governing the MDH program at § 412.108(a)(1) and (c)(2)(iii) and the general payment rules at § 412.90(j) to reflect any such further extension of the MDH program. We also noted that these modifications to our proposed conforming changes would only be made if the MDH program were to be extended by statute beyond December 31, 2024.
Comment: Many commenters expressed support for extending the MDH program or making the MDH program permanent and noted that they would continue supporting congressional efforts to protect the MDH program. Some commenters also expressed support for increasing the base year for these hospitals. Others supported an additional base year for calculating MDH payments. Several state hospital associations expressed their concern that hospitals in their states would experience significant payment decreases as a result of the expiration of the MDH program. A few commenters urged CMS for action to be taken to ensure that the MDH program is extended.
Response: While we appreciate the commenters' concerns about the expiration of the MDH program and the financial impact to affected providers if the MDH program is not extended beyond CY 2024, CMS does not have the authority under current law to extend the MDH program beyond the December 31, 2024 statutory expiration date. Similarly, Section 1886(b)(3)(D) of the Act specifies the applicable base years or “target amounts” for hospitals classified as MDHs. These comments are similar to comments we received previously, prior to the statutory extension of the MDH program for FYs 2023 and 2024 provided by subsequent legislation, and discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49064 ).
Comment: Several commenters expressed support for CMS' policy that allows MDHs to apply for SCH status in advance of the expiration of the MDH program and be paid as such under certain conditions. A commenter requested that CMS clearly communicate this option to rural hospitals in the event the designation lapses. Some commenters also requested that CMS automatically reinstate MDH status to all previously qualifying hospitals, including hospitals that became SCHs and hospitals that cancelled rural status in anticipation of the MDH program expiration, if a retroactive extension to the MDH program is made.
Response: We appreciate the commenters' support of our policy allowing MDHs to apply for SCH status in advance of the expiration of the MDH program and to be paid as such under certain conditions and allow for a seamless transition from MDH classification to SCH classification. As we have done with prior legislative expirations of the MDH program, CMS will communicate this information to the provider community. In response to the suggestion that CMS provide former MDHs with ability to rescind their newly acquired SCH status and reinstate their MDH status in a seamless manner if a retroactive extension to the MDH program is made, we understand the desire on the part of hospitals for certainty in the face of MDH program expiration and will consider for future rulemaking any potential mechanisms to further streamline such transitions in connection with legislative extensions of the MDH program. We note that under the current regulations at § 412.108(b)(4), the effective date for MDH classification is as of the date the MAC receives the complete application. ( print page 69354) A MDH that applied for and was classified as an SCH in advance of the MDH expiration per the regulations at § 412.92(b)(2)(v) could request a cancellation of its SCH status and simultaneously re-apply for MDH status if the MDH program were to be extended, and the MDH classification would be effective as of the date that the MAC receives the complete application. In response to the suggestion that CMS automatically reinstate MDH status to providers that cancelled their rural status in anticipation of the MDH program expiration, we note that per the regulations at § 412.103(g)(4), a hospital's cancellation of its rural classification is effective beginning with the next Federal fiscal year.
Comment: Commenters urged CMS to expedite restoration of MDH status, should Congress act to extend these programs. They requested that CMS move expeditiously to restore payments and act quickly to retroactively address a program lapse in the event that the program is extended after December 31, 2024. A commenter requested that CMS clarify how it might handle the continuation of the program, should Congress enact legislation to extend it. A few commenters expressed appreciation for CMS' most recent implementation of the extension of the MDH program. A commenter expressed support for the decision to not require MDHs to reapply for classification for the period of October 1, 2024 through December 31, 2024.
Response: We appreciate the commenters' support for CMS' implementation of the most recent MDH extensions. We also appreciate the commenters' sharing their concerns relating to a retroactive restoration of the MDH program. As with past extensions, CMS will evaluate enacted legislation to determine the most appropriate approach to implement changes to the law, including instructions to the MACs to reinstate MDH status to eligible hospitals. As in the past, and as acknowledged by some of the commenters, we will make every effort to implement any extension of the MDH program as expeditiously as possible.
In summary, under current law, beginning January 1, 2025, all hospitals that previously qualified for MDH status will no longer have MDH status.
After consideration of the public comments we received, we are adopting as final the proposed conforming changes to the regulations text at §§ 412.90 and 412.108 to reflect the extension of the MDH program through December 31, 2024 in accordance with section 307 of the CAA, 2024 ( Pub. L. 118-42 ). We are finalizing the proposed changes in paragraphs (a)(1) and (c)(2)(iii) of § 412.108 and paragraph (j) of § 412.90 without modification.
Section 1886(h) of the Act, as added by section 9202 of the Consolidated Omnibus Budget Reconciliation Act (COBRA) of 1985 (Pub. L. 99-272) and as currently implemented in the regulations at 42 CFR 413.75 through 413.83 , establishes a methodology for determining payments to hospitals for the direct costs of approved graduate medical education (GME) programs. Section 1886(h)(2) of the Act sets forth a methodology for the determination of a hospital-specific base-period per resident amount (PRA) that is calculated by dividing a hospital's allowable direct costs of GME in a base period by its number of full-time equivalent (FTE) residents in the base period. The base period is, for most hospitals, the hospital's cost reporting period beginning in FY 1984 (that is, October 1, 1983 through September 30, 1984). The base year PRA is updated annually for inflation. In general, Medicare direct GME payments are calculated by multiplying the hospital's updated PRA by the weighted number of FTE residents working in all areas of the hospital complex (and at nonprovider sites, when applicable), and the hospital's Medicare share of total inpatient days.
Section 1886(d)(5)(B) of the Act provides for a payment adjustment known as the indirect medical education (IME) adjustment under the IPPS for hospitals that have residents in an approved GME program, in order to account for the higher indirect patient care costs of teaching hospitals relative to nonteaching hospitals. The regulations regarding the calculation of this additional payment are located at 42 CFR 412.105 . The hospital's IME adjustment applied to the DRG payments is calculated based on the ratio of the hospital's number of FTE residents training in either the inpatient or outpatient departments of the IPPS hospital (and, for discharges occurring on or after October 1, 1997, at non-provider sites, when applicable) to the number of inpatient hospital beds.
The calculation of both direct GME payments and the IME payment adjustment is affected by the number of FTE residents that a hospital is allowed to count. Generally, the greater the number of FTE residents a hospital counts, the greater the amount of Medicare direct GME and IME payments the hospital will receive. In an attempt to end the implicit incentive for hospitals to increase the number of FTE residents, Congress established a limit on the number of allopathic and osteopathic residents that a hospital could include in its FTE resident count for direct GME and IME payment purposes in the Balanced Budget Act of 1997 ( Pub. L. 105-33 ). Under section 1886(h)(4)(F) of the Act, for cost reporting periods beginning on or after October 1, 1997, a hospital's unweighted FTE count of residents for purposes of direct GME cannot exceed the hospital's unweighted FTE count for direct GME in its most recent cost reporting period ending on or before December 31, 1996. Under section 1886(d)(5)(B)(v) of the Act, a similar limit based on the FTE count for IME during that cost reporting period is applied, effective for discharges occurring on or after October 1, 1997. Dental and podiatric residents are not included in this statutorily mandated cap.
We received some IME and direct GME (DGME) related comments that were outside the scope of the proposed rule, including a comment related to the eligibility of SCHs paid under the hospital-specific rate and MDHs to receive IME payments. Because we consider these public comments to be outside the scope of the proposed rule, we are not addressing these comments in this final rule.
CMS has increased the overall number of slots available to teaching hospitals on several previous occasions. Notably, Congress authorized Medicare payment for one thousand additional FTE GME resident slots in section 126(a) of the Consolidated Appropriations Act, 2021, adding paragraph 1886(h)(9) to the Act.
Most recently, section 4122(a) of the CAA, 2023 amended section 1886(h) of the Act by adding a new section 1886(h)(10) of the Act requiring the distribution of additional residency positions (also referred to as slots) to hospitals. Section 1886(h)(10)(A) of the Act requires that for FY 2026, the Secretary shall initiate an application round to distribute 200 residency positions. At least 100 of the positions made available under section ( print page 69355) 1886(h)(10)(A) shall be distributed for psychiatry or psychiatry subspecialty residency training programs. The Secretary is required, subject to certain provisions in the law, to increase the otherwise applicable resident limit for each qualifying hospital that submits a timely application by the number of positions that may be approved by the Secretary for that hospital. The Secretary is required to notify hospitals of the number of positions distributed to them by January 31, 2026, and the increase is effective beginning July 1, 2026.
Section 1886(h)(10)(B)(ii) of the Act requires a minimum distribution for certain categories of hospitals. Specifically, the Secretary is required to distribute at least 10 percent of the aggregate number of total residency positions available to each of four categories of hospitals. Stated briefly, and discussed in greater detail later in this final rule, the categories are as follows: (1) hospitals located in rural areas or that are treated as being located in a rural area (pursuant to sections 1886(d)(2)(D) and 1886(d)(8)(E) of the Act); (2) hospitals in which the reference resident level of the hospital is greater than the otherwise applicable resident limit; (3) hospitals in states with new medical schools or additional locations and branches of existing medical schools; and (4) hospitals that serve areas designated as Health Professional Shortage Areas (HPSAs). Section 1886(h)(10)(F)(iii) of the Act defines a qualifying hospital as a hospital in one of these four categories.
Section 1886(h)(10)(C) of the Act places certain limitations on the distribution of the residency positions. First, a hospital may not receive more than 10 additional full-time equivalent (FTE) residency positions. Second, no increase in the otherwise applicable resident limit of a hospital may be made unless the hospital agrees to increase the total number of FTE residency positions under the approved medical residency training program of the hospital by the number of positions made available to that hospital. Third, if a hospital that receives an increase to its otherwise applicable resident limit under section 1886(h)(10) of the Act is eligible for an increase to its otherwise applicable resident limit under 42 CFR 413.79(e)(3) (or any successor regulation), that hospital must ensure that residency positions received under section 1886(h)(10) of the Act are used to expand an existing residency training program and not for participation in a new residency training program.
Section 1886(h)(10)(B)(i) of the Act directs the Secretary to take into account the “demonstrated likelihood” of the hospital filling the positions made available within the first 5 training years beginning after the date the increase would be effective, as determined by the Secretary. In accordance with section 1886(h)(10)(A)(iv) of the Act, the increase would be effective beginning July 1 of the fiscal year of the increase; therefore, additional residency positions under section 1886(h)(10) of the Act would be effective July 1, 2026.
Consistent with the application cycle established for section 126 of the CAA, 2021 ( 86 FR 73419 through 73445 ) we proposed that the application deadline for the additional positions made available for a fiscal year be March 31 of the prior fiscal year; that is, for FY 2026, the application deadline would be March 31, 2025. Accordingly, all references in this section to the application deadline are references to the application deadline of March 31, 2025.
We proposed that a hospital show a “demonstrated likelihood” of filling the additional positions (sometimes equivalently referred to as slots) for which it applies by demonstrating that it does not have sufficient room under its current FTE resident cap(s) to accommodate a planned new program or expansion of an existing program. In order to be eligible for additional positions, the new program or expansion of an existing program could not begin prior to July 1, 2026, the effective date of the section 4122 residency positions.
In order to demonstrate that a hospital does not have sufficient room under its current FTE resident cap(s) for purposes of the prioritization discussed at section c.3. of this preamble, if applicable, we proposed that a hospital would be required to submit copies of its most recently submitted Worksheet E, Part A and Worksheet E-4 from the Medicare cost report (CMS-Form- 2552-10) as part of its application for an increase to its FTE resident cap(s). The hospital would demonstrate and attest to a planned new program or expansion of an existing program by meeting at least one of the following two “Demonstrated Likelihood” criteria:
- “Demonstrated Likelihood” Criterion 1 (New Residency Program). The hospital does not have sufficient room under its FTE resident cap, is not a rural hospital eligible for an increase to its cap under 42 CFR 413.79(e)(3) (or any successor regulation), and intends to use the additional FTEs as part of a new residency program that it intends to establish on or after the date the increase would be effective (that is, a new program that begins training residents at any point within the hospital's first 5 training years beginning on or after the effective date of the increase). Under “Demonstrated Likelihood” Criterion 1, the hospital will be required to meet at least one of the following conditions as part of its application:
++ Application for accreditation of the new residency program has been submitted to the Accreditation Council for Graduate Medical Education (ACGME) (or application for approval of the new residency program has been submitted to the American Board of Medical Specialties (ABMS)) by the application deadline.
++ The hospital has received written correspondence from the ACGME (or ABMS) acknowledging receipt of the application for the new residency program, or other types of communication concerning the new program accreditation or approval process (such as notification of site visit) by the application deadline.
- “Demonstrated Likelihood” Criterion 2 (Expansion of an Existing Residency Program). The hospital does not have sufficient room under its FTE resident cap, and the hospital intends to use the additional FTEs to expand an existing residency training program within the hospital's first 5 training years beginning on or after the date the increase would be effective. Under “Demonstrated Likelihood” Criterion 2, the hospital will be required to meet at least one of the following conditions as part of its application:
++ The hospital has received approval by the application deadline from an appropriate accrediting body (the ( print page 69356) ACGME or ABMS) to expand the number of FTE residents in the program.
++ The hospital has submitted a request by the application deadline for a permanent complement increase of the existing residency program.
++ The hospital currently has unfilled positions in its residency program that have previously been approved by the ACGME and is now seeking to fill those positions.
Under “Demonstrated Likelihood” Criterion 2, the hospital is applying for an increase in its FTE resident cap because it is expanding an existing residency program. We proposed that as of the application deadline the hospital is either already training residents in this program, or, if the program exists at another hospital as of that date, the residents will begin to rotate to the applying hospital on or after the effective date of the increase. In addition, we note that section 1886(h)(10)(C)(ii) of the Act requires that if a hospital is awarded positions, that hospital must increase the number of its residency positions by the amount the hospital's FTE resident cap increases, based on the newly awarded positions under section 4122 of CAA, 2023. Therefore, we proposed that a hospital must, as part of its application, attest to increasing the number of its residency positions by the amount of the hospital's FTE resident cap increase based on any newly awarded positions, in accordance with the provisions of section 1886(h)(10)(C)(ii) of the Act.
In this section we present a summary of the public comments and our responses related to the proposal determining whether a hospital has a “demonstrated likelihood” of filling the positions awarded under section 4122 of the CAA, 2023.
Comment: A few commenters expressed concern that requiring a hospital to demonstrate that it does not have sufficient room under its current FTE cap to accommodate a program expansion or a new program would not benefit rural programs. The commenters stated that large academic medical centers generally have more resources and are better funded, thus they are able to take on additional residents above their Medicare FTE cap. The commenters stated that rural hospitals are unlikely to be able to take on residents that are not funded through Medicare GME. As a result, rural hospitals would be disadvantaged because they would not be seen as “likely to fill” additional slots since most rural hospitals are not training above their cap due to limited resources.
A commenter asked that CMS reconsider the policy related to “demonstrated likelihood” and allow for exceptions for certain unique situations where a hospital may be training under its cap at the time of the application but would be training at or over its cap by the time the additional slots under section 4122 would be effective. The commenter provided the example of a hospital training residents in a new residency program. In this example the hospital is operating below its cap because it is currently building the program, but the hospital expects to be operating above its cap when the additional section 4122 slots would be effective. The commenter stated that such a hospital should be eligible for its full FTE request under section 4122.
Response: We appreciate the commenters' concerns related to rural hospitals and hospitals with new programs potentially training below their FTE caps, and therefore being unable to demonstrate the need for an increase to their FTE caps. The comparison between a hospital's FTE count and its adjusted FTE cap will be made where we distribute any slots remaining by HPSA score after we distribute the “up to 1.00 FTE” to each qualifying hospital. We did not propose to compare a hospital's FTE count to its adjusted FTE cap when awarding up to 1.00 FTE to each qualifying hospital.
Specifically, in the proposed rule we stated that “in order to demonstrate that a hospital does not have sufficient room under its current FTE resident cap(s) for purposes of the prioritization discussed at section c.3. of this preamble, if applicable . . .” ( 89 FR 36214 ). Section c.3. referred to the section “Prioritization of Applications by HPSA Score”, this is a separate section from the discussion of awarding each qualifying hospital up to 1.00 FTE, which was included at section c.2., “Pro Rata Distribution and Limitation on Individual Hospitals”. We note that if we prioritize the distribution of any remaining slots by HPSA score, we would only consider the FTE cap and count information included on the cost report submitted with the application; we would not consider a future cost report as the commenter suggests. In addition to providing a level of efficiency with respect to the section 4122 application reviews, we attempt to limit the need to have decision criteria based on future expectations versus cost report data, as the latter can be audited under existing processes.
Comment: One commenter requested clarification on the requirements to receive additional slots so that hospitals can accurately complete the application process. The commenter stated that guidance would be appreciated as to all program requirements, including specifically how hospitals can show a “demonstrated likelihood” that they will fill additional positions and that their current FTE caps leave insufficient room for new or expanded programs.
Response: We refer the commenters to section j. of this preamble which discusses the “Application Process for Receiving Increases in FTE Resident Caps”. This section lists the options for attesting to meeting Demonstrated Likelihood Criterion One or Two as part of the attestation that will be included with the section 4122 application module. Prior to the start of the application period, additional resources related to the section 4122 application process will be included on CMS' Direct Graduate Medical Education (DGME) website at https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/direct-graduate-medical-education-dgme .
After consideration of the comments received, we are finalizing our proposed policies related to the determination that a hospital has demonstrated a likelihood of filling the positions for “Demonstrated Likelihood” Criterion 1 (New Residency Program) or for “Demonstrated Likelihood” Criterion 2 (Expansion of an Existing Residency Program), without modification.
Section 1886(h)(10)(B)(ii) of the Act requires the Secretary to distribute not less than 10 percent of resident positions available for distribution to each of four categories of hospitals. Under section 1886(h)(10)(B)(ii)(I) of the Act, the first of these categories consists of hospitals that are located in a rural area (as defined in section 1886(d)(2)(D) of the Act) or are treated as being located in a rural area (pursuant to section 1886(d)(8)(E) of the Act). We refer to this category as Category One. We note that the definition of Category One for purposes of section 4122 of the CAA, 2023 mirrors the definition of Category One included under section 1886(h)(9)(B)(ii)(I) for purposes of section 126 of the CAA, 2021. Therefore, we proposed to determine Category One eligibility as discussed in the final rule implementing section 126 of the CAA, 2021 ( 86 FR 73422 through 73424 ).
For purposes of determining whether a hospital is considered rural, we proposed to use the County to CBSA Crosswalk and Urban CBSAs and Constituent Counties for Acute Care Hospitals File, or successor files containing similar information, from the ( print page 69357) most recent FY IPPS final rule (or correction notice if applicable). This file will be available on the CMS website in approximately August 2024, the year prior to the year of the application deadline, March 31, 2025. Under the file's current format, blank cells in Columns D and E indicate an area outside of a CBSA.
Under section 1886(d)(8)(E) of the Act, a subsection (d) hospital (that is, generally, an IPPS hospital) that is physically located in an urban area is treated as being located in a rural area for purposes of payment under the IPPS if it meets criteria specified in section 1886(d)(8)(E)(ii) of the Act, as implemented in the regulations at § 412.103. Under these regulations, a hospital may apply to CMS to be treated as located in a rural area for purposes of payment under the IPPS. Given the fixed number of available residency positions, it is necessary to establish a deadline by which a hospital must be treated as being located in a rural area for purposes of Category One. We proposed to use Table 2, or a successor table containing similar information, posted with the most recent IPPS final rule, available on the CMS website in approximately August 2024, (or correction notice if applicable), to determine whether a hospital is reclassified to rural under § 412.103. If a hospital is not listed as reclassified to rural on Table 2, but has been subsequently approved by the CMS Regional Office to be treated as being located in a rural area for purposes of payment under the IPPS as of the March 31, 2025 application deadline, the hospital would submit its approval letter with its application in order to be treated as being located in a rural area for purposes of Category One.
In this section we present a summary of the public comments and our responses to the proposed determination of which hospitals are located in a rural area or are treated as being located in a rural area (Category One).
Comment: Several commenters stated that although not referenced in these proposed rules, a change in rural categorization to eliminate hospitals “treated as rural” that are not in fact geographically rural is essential to increasing the number of geographically rural hospitals gaining new positions, and hopefully that change can be made in legislation if not in rules.
A few commenters encouraged CMS to consider how to incentivize rural hospitals to apply for the section 4122 opportunity and award slots that will increase rural training.
Response: We agree with the commenters that increasing the number geographically rural hospitals that receive additional slots is an essential goal. We note that the law requires that both hospitals that are located in a rural area (as defined in section 1886(d)(2)(D) of the Act) and hospitals that are treated as being located in a rural area (pursuant to section 1886(d)(8)(E) of the Act), qualify as Category One hospitals.
In order to support geographically rural hospitals in the application process we anticipate continuing the outreach efforts that we have in place for the section 126 distribution, and adding outreach regarding the section 4122 distribution in these efforts. CMS has worked in conjunction with the Health Resources and Services Administration's (HRSA) Federal Office of Rural Health Policy to educate potential applicants about the section 126 application process. On February 13, 2023 and January 17, 2024, CMS participated with HRSA and Rural Residency Planning and Development—Technical Assistance Center ( www.ruralgme.org ) in webinars aimed at educating potential rural applicants about the section 126 application process. CMS has also participated in the rural health and hospital open door forums and is accessible to anyone who submits a question through our section 126 email inbox at [email protected] . In addition, background information regarding the section 126 application process and frequently asked questions are posted on CMS' DGME website, https://www.cms.gov/Medicare/payment/prospective-payment-systems/acute-inpatient-pps/direct-graduate-medical-education-dgme . The DGME website also provides instructions on how to submit a question directly to CMS using the Medicare Electronic Application Request Information System TM (MEARIS TM ), the application module that will be used for both the section 126 and section 4122 application processes. We will be updating the CMS DGME website to include similar resources and communication tools for the section 4122 application process After consideration of the comments received, we are finalizing our policy with respect to Category One as proposed, without modification.
Under section 1886(h)(10)(B)(ii)(II) of the Act, the second category consists of hospitals in which the reference resident level of the hospital (as specified in section 1886(h)(10)(F)(iv) of the Act) is greater than the otherwise applicable resident limit. We refer to this category as Category Two. We note the definition of Category Two under section 1886(h)(10)(B)(ii)(II) of the Act mirrors the definition of Category Two under section 1886(h)(9)(B)(ii)(II), section 126 of the CAA, 2021. Therefore, we proposed to determine Category Two eligibility as discussed in the final rule implementing section 126 of the CAA, 2021 ( 86 FR 73424 through 73425 ) with adjustments to consider the provisions of sections 126, 127, and 131 of the CAA, 2021, as discussed later.
Under section 1886(h)(10)(F)(iv) of the Act, the term “reference resident level” means, with respect to a hospital, the resident level for the most recent cost reporting period of the hospital ending on or before the date of enactment of section 1886(h)(10) of the Act, December 29, 2022, for which a cost report has been settled (or, if not, submitted (subject to audit)).
Under section 1886(h)(10)(F)(v) of the Act, the term “resident level” has the meaning given such term in paragraph (7)(C)(i). That section defines “resident level” as with respect to a hospital, the total number of full-time equivalent residents, before the application of weighting factors (as determined under paragraph (4)), in the fields of allopathic and osteopathic medicine for the hospital.
Under section 1886(h)(10)(F)(i) of the Act, the term “otherwise applicable resident limit” means, “with respect to a hospital, the limit otherwise applicable under subparagraphs (F)(i) and (H) of paragraph (4) on the resident level for the hospital determined without regard to the changes made by this provision of the CAA, 2023, but taking into account section 1886(h)(7)(A), (7)(B), (8)(A), (8)(B), and (9)(A)” of the Act. These cross-referenced sub-paragraphs all address the distribution of positions and redistribution of unused positions.
As finalized for purposes of section 126 of the CAA, 2023, the “reference resident level” refers to a hospital's allopathic and osteopathic FTE resident count for a specific period. The definition can vary based on what calculation is being performed to determine the correct allopathic and osteopathic FTE resident count (see, for example, 42 CFR 413.79(c)(1)(ii) ) ( 86 FR 73424 )). As noted previously, section 4122 of the CAA, 2023, under new section 1886(h)(10)(F)(iv) of the Act defines the “reference resident level” as coming from the most recent cost reporting period of the hospital ending ( print page 69358) on or before the date of enactment of the CAA, 2023 (that is, December 29, 2022).
Under new section 1886(h)(10)(F)(i) of the Act, the term “otherwise applicable resident limit” is defined as “the limit otherwise applicable under subparagraphs (F)(i) and (H) of paragraph (4) on the resident level for the hospital determined without regard to this paragraph [that is, section 1886(h)(10) of the Act], but taking into account paragraphs (7)(A), (7)(B), (8)(A), (8)(B), and (9)(A).” In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 25505 ), we finalized for purposes of section 126 of the CAA, 2021, the definition of “otherwise applicable resident limit” as the hospital's 1996 cap during its reference year, adjusted for the following: “new medical residency training programs” as defined at § 413.79(l); participation in a Medicare GME affiliation agreement as defined at §§ 413.75(b) and referenced at 413.79(f); participation in an Emergency Medicare GME affiliation agreement as defined at § 413.79(f); participation in a hospital merger; whether an urban hospital has a separately accredited rural training track program as defined at § 413.79(k); applicable decreases or increases under section 422 of the MMA, applicable decreases or increases under section 5503 of the Affordable Care Act, and applicable increases under section 5506 of the Affordable Care Act. For purposes of section 4122 of the CAA, 2023, we proposed to use this same definition of “otherwise applicable resident limit” and adding to this definition the following: applicable increases or adjustments under sections 126, 127, and 131 of the CAA, 2021.
Regarding the term “resident level”, in the CY 2011 OPPS final rule ( 75 FR 46391 ) we indicated that we generally refer to a hospital's number of unweighted allopathic and osteopathic FTE residents in a particular period as the hospital's resident level, which we proposed to define consistently with the definition in section 4122 of the CAA, 2023; that is, the “resident level” under section 1886(h)(7)(c)(i) of the Act, which is defined as the total number of full-time equivalent residents, before the application of weighting factors (as determined under paragraph 1886(h)(4) of the Act), in the fields of allopathic and osteopathic medicine for the hospital.
For the purposes of section 4122 of the CAA, 2023 we proposed that the definitions of the terms “otherwise applicable resident limit,” “reference resident level,” and “resident level” should be as similar as possible to the definitions those terms have in the regulations at § 413.79(c), as initially set out in the CY 2011 OPPS rulemaking, as revised for purposes of section 126 of the CAA, 2021 ( 86 FR 73424 ) with adjustments made to the definition of “otherwise applicable resident limit” for sections 126, 127, and 131 of the CAA, 2021.
We did not receive any public comments on our proposal for determining whether a hospital's refence resident level is greater than its otherwise applicable resident limit (Category Two). We are finalizing this policy as proposed.
The third category specified in section 1886(h)(10)(B)(ii)(III) of the Act, as added by section 4122 of CAA, 2023, consists of hospitals located in States with new medical schools that received “Candidate School” status from the Liaison Committee on Medical Education (LCME) or that received “Pre-Accreditation” status from the American Osteopathic Association (AOA) Commission on Osteopathic College Accreditation (the COCA) on or after January 1, 2000, and that have achieved or continue to progress toward “Full Accreditation” status (as such term is defined by the LCME) or toward “Accreditation” status (as such term is defined by the COCA); or additional locations and branch campuses established on or after January 1, 2000, by medical schools with “Full Accreditation” status (as such term is defined by LCME) or “Accreditation” status (as such term is defined by the COCA). We note that the statutory language is specific with respect to these definitions. We refer to this category as Category Three. We note that the definition of Category Three for purposes of section 4122 of the CAA, 2023, mirrors the definition of Category Three included under section 1886(h)(9)(B)(ii)(III) of the Act for purposes of section 126 of the CAA, 2021. Therefore, we proposed to determine Category Three eligibility as discussed in the final rule implementing section 126 of the CAA, 2021 ( 86 FR 73425 through 73426 ).
We proposed that the hospitals located in the following 35 States and one territory, referred to as Category Three States, would be considered Category Three hospitals: Alabama, Arizona, Arkansas, California, Colorado, Connecticut, Delaware, Florida, Georgia, Idaho, Illinois, Indiana, Kansas, Kentucky, Louisiana, Massachusetts, Michigan, Mississippi, Missouri, Nevada, New Jersey, New Mexico, New York, North Carolina, Ohio, Oklahoma, Pennsylvania, Puerto Rico, South Carolina, Tennessee, Texas, Utah, Virginia, Washington, West Virginia, and Wisconsin. If a hospital is located in a state not listed here, but it believes the state in which it is located should be on this list, the hospital may contact CMS through the MEARIS TM application module to make a change to this list, or must provide documentation with submission of its application to CMS that the state in which it is located has a medical school or additional location or branch campus of a medical school established on or after January 1, 2000. Pursuant to the statutory language, all hospitals in such states are eligible for consideration; the hospitals, themselves, do not need to meet the conditions of section 1886(h)(10)(B)(ii)(III)(aa) or (bb) of the Act in order to be considered.
In this section we present a summary of the public comments and our responses related to the proposal determining which hospitals are located in states with new medical schools or additional locations and branch campuses (Category Three).
Comment: We received several requests to add states to the list of Category Three states. A commenter stated Minnesota has an additional branch campus of a medical school established after January 1, 2000. The commenter stated that beginning in the 2025-2026 academic year and as notified November 27, 2023, the University of Minnesota Medical School CentraCare Regional Campus St. Cloud formally expanded as a new regional campus of the University of Minnesota Medical School. Another commenter stated that Minnesota should be added to the list of Category Three states due to expansion of the University of Minnesota Medical School, which accepted its first medical school applications at a branch campus in May 2024.
A commenter requested CMS amend the list of states where hospitals may qualify under Category Three to include Montana and Oregon. The commenter stated that colleges of osteopathic medicine locations in Montana and Oregon meet the definition of “New Medical Schools, or Additional Locations and Branch Campuses”. The commenter noted that Western University of Health Sciences/College of Osteopathic Medicine of the Pacific- Northwest in Lebanon, Oregon, began operation in 2011, Touro College of Osteopathic Medicine Montana opened in 2023, and Rocky Vista University Montana College of Osteopathic Medicine opened in 2023. ( print page 69359)
Response: We thank the commenters for notifying us that these states should be added to the list of Category Three states. We are adding Minnesota, Montana, and Oregon to the list of Category Three states for purpose of section 4122. In addition, since the list of Category Three states for section 4122 mirrors the list of Category Three states for section 126, these states will be added to the list of Category Three states for round 4 of section 126 (FY 2026) and future rounds. Therefore, for both section 4122 and round 4 of section 126 and future rounds, hospitals in the following 38 States and one territory, referred to as Category Three States, would be considered Category Three hospitals: Alabama, Arizona, Arkansas, California, Colorado, Connecticut, Delaware, Florida, Georgia, Idaho, Illinois, Indiana, Kansas, Kentucky, Louisiana, Massachusetts, Michigan, Minnesota, Mississippi, Missouri, Montana, Nevada, New Jersey, New Mexico, New York, North Carolina, Ohio, Oklahoma, Oregon, Pennsylvania, Puerto Rico, South Carolina, Tennessee, Texas, Utah, Virginia, Washington, West Virginia, and Wisconsin.
The fourth category specified in the law consists of hospitals that serve areas designated as HPSAs under section 332(a)(1)(A) of the Public Health Service Act (PHSA), as determined by the Secretary. Category Four for section 4122 of the CAA, 2023 mirrors the definition of Category Four included under section 1886(h)(9)(B)(ii)(IV) for purposes of implementing section 126 of the CAA, 2021. Therefore, we proposed to determine Category Four eligibility as discussed in the final rule implementing section 126 of the CAA, 2021 ( 86 FR 73426 through 73430 ).
We proposed that an applicant hospital qualifies under Category Four if it participates in training residents in a program in which the residents rotate for at least 50 percent of their training time to a training site(s) physically located in a primary care or mental-health-only geographic HPSA. Specific to mental-health-only geographic HPSAs, we proposed that the program must be a psychiatry program or a subspecialty of psychiatry. In addition, a Category Four hospital must submit an attestation, signed and dated by an officer or administrator of the hospital who signs the hospital's Medicare cost report, that it meets the requirement that residents rotate for at least 50 percent of their training time to a training site(s) physically located in a primary care or mental-health-only geographic HPSA.
In this section we present a summary of the public comments and our responses related to determining which hospitals serve areas designated as HPSAs under section 332(a)(1)(A) of the PHSA (Category Four).
Comment: A commenter stated that CMS should adjust its definition of Category Four in light of the small number of programs that apply and meet this definition. The commenter stated that CMS should revise its requirement that at least 50 percent of the resident's training time must occur at facilities located in a HPSA. The commenter stated this change will provide programs with greater flexibility, particularly if some rotations are not located in a designated HPSA site.
Response: We appreciate the commenter's suggestion to add flexibility to the qualifying criterion for Category Four. However, the language at section 1886(h)(10)(B)(ii)(IV) of the Act states “[h]ospitals that serve areas designated as health professional shortage areas under section 332(a)(1)(A) of the Public Health Service Act” (emphasis added). We continue to believe that the inclusion of eligibility Category Four was meant to support residency training programs that aim to provide a considerable amount of training in primary care or mental-health-only geographic HPSAs and that any amount less than 50 percent is not sufficiently indicative of a program that adequately serves the needs of residents of these HPSAs. After consideration of the comments received, we are finalizing our policy with respect to Category Four as proposed, without modification.
Section 1886(h)(10)(F)(iii) of the Act defines a “qualifying hospital” as “a hospital described in any of the subclauses (I) through (IV) of subparagraph (B)(ii).” As such, and consistent with the definition of “qualifying hospital” used for purposes of section 126 of the CAA, 2021 ( 86 FR 73430 through 73431 ), we proposed to define a qualifying hospital as a Category One, Category Two, Category Three, or Category Four hospital, or one that meets the definitions of more than one of these categories.
In this section we present a summary of the public comments and our responses related to determining whether a hospital is considered a qualifying hospital.
Comment: A few commenters expressed support for hospitals that are training over their caps being able to qualify for additional residency slots under section 4122. One commenter stated that they support the eligibility categories, particularly Category Two. The commenter stated that this category would be crucial for allotting slots to hospitals that truly need them, particularly since these hospitals bear an additional financial burden for investing in the healthcare workforce. The commenter stated that when these hospitals are in areas with new medical schools, the need for additional training positions would be even more critical in order to accommodate growing the healthcare workforce.
After consideration of the comments received, we are finalizing our policy with respect to the definition of a qualifying hospital as proposed, without modification.
Section 1886(h)(10)(A)(ii) of the Act limits the aggregate number of total new residency positions made available in FY 2026 across all hospitals to no more than 200. Section 1886(h)(10)(A)(iii) of the Act further specifies that at least 100 of the positions made available under section 1886(h)(10) must be distributed for a psychiatry or psychiatry subspecialty residency. The phrase “psychiatry or psychiatry subspecialty residency” is defined at section 1886(h)(10)(F)(ii) of the Act to mean “a residency in psychiatry as accredited by the Accreditation Council for Graduate Medical Education (ACGME) for the purpose of preventing, diagnosing, and treating mental health disorders.”
We proposed that of the total residency slots distributed under section 4122 of the CAA, 2023, at least 100 but not more than 200 slots would be distributed to hospitals applying for residency programs in psychiatry and psychiatry subspecialties. For purposes of determining which programs are considered psychiatry subspecialties, we proposed to refer to the list included on ACGME website at https://www.acgme.org/ under the “Specialties” tab, currently: Addiction Medicine, Addiction Psychiatry, Brain Injury Medicine, Child and Adolescent Psychiatry, Consultation-Liaison ( print page 69360) Psychiatry, Forensic Psychiatry, Geriatric Psychiatry, Hospice and Palliative Medicine, and Sleep Medicine. We note that the ACGME list of psychiatry subspecialties may change, and we proposed that the list of psychiatry subspecialties included on the ACGME website at the time of application submission would guide determination of which programs CMS would consider psychiatry subspecialties. In accordance with statute, the subspecialty would have to be accredited with psychiatry as a core specialty. We also proposed that the remaining non-psychiatric slots would be awarded to other approved medical residency programs under 42 CFR 413.75(b) .
In this section we present a summary of public the comments and our responses related to the requirement that at least 100 but not more than 200 of the positions made available under section 4122 must be distributed for a psychiatry or psychiatry subspecialty residency.
Comment: One commenter stated that they applaud the proposals that focus on areas of known need in their rural and underserved communities, particularly the needs surrounding psychiatric health disorders. The commenter stated that they stand ready to meet the needs of their communities under any slot expansions, including providing substance use disorder care.
Response: We appreciate the commenter's support and their efforts in providing the necessary psychiatric health services to members of their community.
Comment: Commenters requested that CMS clarify how they would address the situation if fewer than 100 positions are awarded to psychiatry or psychiatry subspecialty residences, requested that other specialties be prioritized, and requested an equivalent increase to hospitals' IPF teaching adjustments.
A few commenters stated that CMS should consider scenarios under which the agency receives applications for fewer than 100 psychiatry FTEs for FY 2026. The commenters requested that in the final rule, CMS address two scenarios: (1) Where fewer than 100 FTEs are awarded to psychiatry or psychiatry subspecialty programs; and (2) Where fewer than 200 positions are awarded in total. The commenters stated that they interpret the statutory language in section 4122 to mean that slots will become effective as of “July 1 of the fiscal year of the increase,” which should allow CMS to award positions through another application cycle if fewer than 100 positions are awarded to psychiatry programs or fewer than 200 positions are awarded in total.
Another commenter stated that it is not clear how CMS would proceed if it does not receive enough requests to allocate 100 slots to psychiatry or psychiatry subspecialty programs. The commenter stated that they recognize the need to train, recruit, and retain behavioral health providers, but believe that CMS should not reserve unfilled slots from this application round for any specialty for future rounds of distribution. As an example, if CMS receives applications for only 90 psychiatry slots, those 10 remaining slots should be allocated to other programs that have submitted applications and qualify under the proposed eligibility criteria. The commenter stated that withholding slots for certain specialties would ignore the growing urgency of physician shortages across all specialties and therefore asked CMS to clarify what it intends to do if the psychiatry slots are not filled in a single round.
Another commenter recommended that if CMS does not receive enough applications to distribute the 100 slots designated for psychiatry programs (or the full 200 slots more generally) in a single round, that CMS hold another application cycle to distribute the remaining slots.
Response: The language that the commenter is referring to “July 1 of the fiscal year of the increase,” refers to July 1, 2026, which is the effective date of the slots awarded under section 4122. However, we believe that while the statute only contemplates a single round for section 4122 occurring in FY 2026, the requirement that 200 slots be distributed and that 100 of the slots go to psychiatry residencies or subspecialties of psychiatry takes precedence. Therefore, in the situation where we are unable to distribute 200 slots and/or fewer than 100 slots are going to psychiatry programs or subspecialties of psychiatry in FY 2026, we would initiate another round of section 4122 distributions in order to meet these statutory criteria.
Comment: A few commenters stated that the application for section 4122 mirrors the application for section 126 in that psychiatry programs are required to subtract the time residents rotate to inpatient psychiatric facilities (IPFs) from their IME FTE requests for awards. Resident time at IPFs is removed from the IME application because IPF facilities and units file a separate cost report under the IPF PPS and receive a facility-level payment adjustment for teaching status. The commenters stated that the amount of required training time for psychiatry residents in inpatient or outpatient settings is significant and noted that the Accreditation Council on Graduate Medical Education (ACGME) requires psychiatry residents to receive between 6 months and 16 months of inpatient psychiatry training and at least 12 months of outpatient psychiatry experience. The commenters stated that while IME FTEs are capped by the Balanced Budget Act of 1997, there is no statutory limitation on the number of FTE residents that CMS may reimburse IPFs for under the IPF PPS. The commenters stated that CMS has limited the number of residents that an IPF can count towards the teaching ratio, as a matter of policy, since the implementation of the IPF PPS in FY 2005.
The commenters stated that awards made under section 4122 would likely represent the largest increase in Medicare-funded psychiatry or psychiatry subspecialty training since Congress capped hospitals' FTE counts in 1997. The commenters requested that because psychiatry residents often spend a significant amount of time training at IPF hospitals and units, CMS should use its authority to increase the number of FTEs at IPFs excluded from requests for increases for IPPS purposes, for slot distributions under section 4122 and section 126.
Response: We understand the commenters' request to receive additional payment under the IPF PPS for residency training time spent in psychiatry distinct party units or psychiatric hospitals since this time is not countable for IME payment purposes. However, we did not propose any increases to the IPF teaching adjustment for purposes of section 4122 and therefore consider these comments to be out of scope and are not responding to them in this final rule. We will consider the issue of increases to the IPF teaching adjustment for future rulemaking.
Comment: A commenter stated that they deeply appreciate CMS' focus on prioritizing the ongoing behavioral health crisis and on reducing disparities through its planned distribution of residency slots. The commenter stated that the COVID-19 pandemic emphasized the importance of mental health and having an adequate mental health care workforce. The commenter stated that addressing behavioral health workforce issues is critically important for those experiencing access issues, such as people living in rural areas, people of color, and people who identify as LGBTQ+. The commenter stated that the proposed rule helps ( print page 69361) address that shortage by increasing the number of GME slots dedicated to psychiatry and related specialties, with a particular emphasis on improving access in areas with provider shortages. The commenter stated that while they understand that the proposed rule is limited to the additional GME slots allocated through section 4122 of the CAA, 2023, they urge CMS to adopt additional training requirements for GME slots to ensure that all trained physicians are able to provide culturally responsive care.
Response: We appreciate the commenter's support related to the distribution of additional slots for psychiatry and subspecialties of psychiatry, with an emphasis on access to care in areas with provider shortages. We agree with the importance of requiring that all physicians are trained in providing culturally responsive care which is why we are requiring for both section 126 and section 4122 (see section e. below) that all applicant hospitals for slots allocated under these provisions are required to attest that they meet the National CLAS Standards to ensure that the these slot distributions broaden the availability of quality care and services to all individuals, regardless of preferred language, cultures, and health beliefs. The website https://thinkculturalhealth.hhs.gov , which provides guidance related to the National CLAS Standards, includes educational material designed to help providers provide culturally and linguistically appropriate services. Educational tools are provided for behavioral health services, which address all aspects of a provider's and client's cultural identity including geography, gender identity, race, and sexual orientation, see https://thinkculturalhealth.hhs.gov/education/behavioral-health .
Comment: A commenter stated that while they understand that it is a statutory requirement that 50 percent of the additional residency positions are dedicated to psychiatry and psychiatry subspecialties, they remind CMS that specialties such as pathology are experiencing significant workforce shortages that need to be addressed in future rules, particularly for rural areas. The commenter stated that physician shortages in specialty care are significant and often overlooked by policy makers, for example, in recent years, annual demand for pathologists in the US has far outstripped the number of new pathologists entering the workforce. The commenter stated that in 2023, only 30% of pathology practice leaders who were seeking to hire at least one or more pathologists reported that they expected to fill all open positions. The commenter stated they believe that the CMS has not done enough to address the issue of physician shortages in the proposed rule. The commenter provided many examples of the influence of pathologists' services on clinical decision-making and stated these services are pervasive and constitute the critical foundation for appropriate patient care. The commenter urged CMS to create opportunities and incentives for the pathologist workforce to expand as needed to meet population growth and ageing.
Another commenter stated that they recognize that CMS is required to prioritize distribution to psychiatry specialties and subspecialties to improve access to critical mental health services, however, they urged CMS to ensure that an adequate number of slots go towards primary care and other specialties with well documented shortages, like internal medicine, family medicine, and pediatrics. The commenter stated that it is important to note that primary care physicians play a significant role in providing mental health care services. The commenter referred to a cross-sectional study using Medical Expenditure Panel Survey data, which found that during the COVID-19 pandemic, primary care physicians provided a significant proportion of care for people with mental health disorders—nearly 40 percent of visits for depression, anxiety, and any mental illness were performed by primary care physicians. The commenter stated that primary care physicians also provided over one-third of the care and wrote a quarter of the prescribed medications for patients with severe mental illness. Another commenter stated that the forecasted insufficiency of primary care physicians in the future health care workforce makes this a pressing concern for public health agencies to take immediate action to prioritize educating and training the next generation of primary care physicians and providing sufficient resources to training centers to competently supervise and instruct these scarce professionals. The commenter recommended that primary care be prioritized in the distribution of the remaining 100 GME slots.
A commenter stated that they support CMS' proposal to include Hospice and Palliative Medicine as a psychiatry subspecialty that may qualify for the reserved psychiatry GME positions under section 4122. The commenter stated that Hospice and Palliative Medicine is an important component of psychiatric care, and the prioritization of this subspecialty will help to build a workforce capable of addressing the needs of patients with serious illness through a psychiatric lens. The commenter requested that as CMS contemplates final policies for allocating the remaining, non-psychiatry GME positions, CMS add a method for prioritizing specialties that offer high value and/or demonstrate significant shortage, such as Hospice and Palliative Medicine. The commenter stated that programs that maintain partnerships with Hospice and Palliative Medicine fellowship programs, for example, surgery residencies that include a paired Hospice and Palliative fellowship track, should also be prioritized. The commenter stated that these changes would help build a physician workforce closely aligned with the nation's evolving healthcare needs and improve care and quality of life for millions of Americans facing serious illness, along with their families and caregivers.
A commenter stated that CMS should enable applicants to tailor programs to support positions needed most in rural and underserved communities. The commenter stated that they commend the emphasis on behavioral health but that the dedication of at least one-half of the total number of positions to psychiatry or psychiatry subspecialty residencies may result in some slots going unused. The commenter stated that in Iowa and nationally, there are additional and significant specialty needs in family medicine, particularly in rural areas (but urban as well); OBGYN, and geriatrics, among others. The commenter stated that they discourage CMS from establishing a set-aside percentage for behavioral health and recommend that CMS defer to local needs.
A commenter stated that while they understand the requirement to distribute at least 100 slots to psychiatry is statutory, the commenter's psychiatry programs are not full, and psychiatrists are permitted to start their practices without completing their last year of residency training. The commenter stated that they have not experienced the need for more slots to train psychiatry residents and requiring 100 slots to be dedicated to psychiatry means those slots cannot be allocated to other programs that are pushing hospitals over their caps.
A commenter stated that they support the provision that directs half of the resident slots towards psychiatry or psychiatry subspecialities, but there is no assurance that these slots will go to the areas that need them most. The commenter stated that CMS should create guardrails to ensure the lack of ( print page 69362) psychiatrists and related specialists in underserved areas throughout the country gets addressed. The commenter stated that if CMS is considering the allocation of GME FTE slots by specialty, it should implement this policy as a pilot project, gather validated data by specialty across the nation, then prioritize primary care physician and psychiatry shortages, and if successful, widely implement such a policy across all of GME.
Response: We understand the commenters' concerns related to physician shortages in specialties in addition to psychiatry and we appreciate the commenters' efforts to address these shortages. We note that the requirement under section 4122 to distribute at least 100 slots to psychiatry or psychiatry subspecialties is statutory and there is no statutory requirement for other specialties.
After consideration of the comments received, we are finalizing the policy to distribute at least 100 slots to psychiatry or subspecialties of psychiatry as proposed, without modification.
As noted earlier in this preamble, section 1886(h)(10)(B)(iii) of the Act requires that each qualifying hospital that submits a timely application under subparagraph 1886(h)(10)(A) of the Act would receive at least 1 (or a fraction of 1) of the positions made available under section 1886(h)(10) of the Act before any qualifying hospital receives more than 1 of such positions. Section 1886(h)(10)(C)(i) of the Act limits a qualifying hospital to receiving no more than 10 additional FTEs from those authorized under section 1886(h)(10) of the Act. As stated earlier in this preamble, we proposed that a qualifying hospital is a Category One, Category Two, Category Three, or Category Four hospital, or one that meets the definitions of more than one of these categories. For purposes of distributing residency slots under section 4122 of the CAA, 2023, we proposed to first distribute slots by prorating the available 200 positions among all qualifying hospitals such that each qualifying hospital receives up to 1.00 FTE, that is, 1.00 FTE or a fraction of 1.00 FTE. We proposed that if residency positions are awarded based on a fraction of 1.00 FTE, each qualifying hospital would receive the same FTE amount. Consistent with the number of decimal places used for the FTE slots awards in other distributions such as section 126 of the CAA, 2021, we proposed to prorate the slot awards under section 4122 of the CAA, 2023, rounded to two decimal places. The table later in this section provides examples of how the 200 slots would be prorated based on the number of qualifying applicants. Given the limited number of residency positions available and the number of hospitals we expect to apply, we proposed that a hospital may not submit more than one application under section 4122 of the CAA, 2023.
We refer readers to further below in this section where we discuss an alternative we considered for the distribution of slots under section 4122 of the CAA, 2023 and present a summary of the public comments we received and our responses. We also refer readers to section I.O.6. of Appendix A of this final rule where we discuss the same alternative considered.
In this section we present a summary of the public comments and our responses related to the requirement to distribute at least 1 (or a fraction of 1) of the positions made available under section 4122 of the CAA, 2023, before any qualifying hospital receives more than 1 position.
Comment: A few commenters supported the proposal to award each qualifying hospital up to 1.00 FTE. A commenter stated that unlike the formula for distribution of the 1,000 GME slots made available through the CAA, 2021, CMS did not propose a “super-prioritization” of HPSA-designated hospitals for the CAA, 2023. The commenter stated that they support the equitable distribution methodology proposed for the 200 slots created by the CAA, 2023, and encouraged CMS to take a similar approach with the slots created by the CAA, 2021. Another commenter stated that they believe the proposed methodology will allow for more participation from qualified providers versus a strictly HSPA-based approach.
Response: We appreciate the commenters' support. We will not be applying this prorating methodology to section 126 of CAA, 2021 because the explicit instruction to award each qualifying hospital 1.00 FTE or a fraction of 1.00 is only included for purposes of the slot distribution under section 4122 of CAA, 2023.
Comment: Several commenters expressed concerns regarding the proposal to award each qualifying hospital up to 1.00 FTE. A commenter stated they continue to support awards being aligned with program lengths, so that for example a hospital applying to train residents in a three-year program can request up to three FTE residents per fiscal year, as is the case for the policy finalized for purposes of section 126 of the CAA, 2021. The commenter stated that they understand that for section 4122 of the CAA, 2023, subsection (B)(iii), “Pro Rata Application”, may prevent CMS from being able to align hospital GME awards with program lengths and that if this is the case, they recommend CMS award a minimum of 1.00 FTE to qualifying hospitals and not award fractional positions. The commenter stated that they believe anything less than 1.00 FTE would harm family medicine residencies—particularly small programs—as it would deter many programs from being able to expand. The commenter stated that while fractional FTE awards may be workable in large academic institutions where there are multiple funding options available, these FTE awards would be a barrier for small residencies that do not have similarly deep resources. The commenter urged CMS to support the sustainability of small programs by distributing a minimum of 1.00 FTE to qualifying residency programs.
A few commenters expressed concern that awarding up to 1.00 FTE per hospital would dissuade rural programs from applying. The commenters noted that these programs are already deterred from applying for section 126 slots because of the HPSA score prioritization and that a disadvantageous pro rata distribution under section 4122 would add yet another barrier to applying. The commenters stated that some rural hospitals may not apply because they may not receive a full slot, or a full FTE. The commenters stated that one slot, or one FTE, covers the cost of training one resident for one year whereas a fraction of an FTE is not incentive enough for rural residency programs to apply because most of the resident's training would not be funded by Medicare. The commenter stated that rural residency programs are less able to shoulder unfunded training compared to large urban academic medical centers and that this situation makes CMS' decision on how to administer the pro rata distribution paramount.
Several commenters expressed concern related to hospitals having to self-fund additional FTEs under the scenario where each qualifying hospital would receive up to 1.00 FTE. Commenters stated that as part of the proposal for section 126, CMS attempted to award slots in a similar ( print page 69363) manner, limiting the award to each qualifying hospital to 1.00 FTE. The commenters stated that in this instance, there was also consensus from the GME community that the 1.00 FTE limitation on awards would not be a meaningful increase for institutions. The commenters stated that because of the longitudinal requirement to train residents over the course of several years, the limitation of 1.00 FTE would limit the development of a full complement in subsequent postgraduate years. The commenters stated that this policy would require hospitals awarded a pro-rata distribution of 1.00 FTE to self-fund full complement increases beyond the 1.00 FTE awarded.
A commenter stated they have significant concerns that CMS' proposed methodology could result in many hospitals receiving only a 1.00 FTE (or less) cap slot, which does not support expanding or starting a new multi-year residency program. The commenter stated they recognize that the language within section 4122 mandating awarding every applicant hospital that applies with up to 1.00 FTE presents certain implementation challenges, however, the commenter requested that CMS consider the implications of not tying the initial pro rata distribution for hospitals to the distribution of remaining slots to those same hospitals. The commenter stated that residency programs typically expand by the length of their program. For example, if a hospital with a four-year psychiatry program currently training 16 residents applied to expand, it would normally do so for four positions (to become a 20-resident training program) or some multiple of four positions. The commenter stated that under CMS' proposal, if a hospital applied to expand a four-year psychiatry program and received only 1.00 FTE under section 4122, the hospital would not receive any reimbursement for the three FTEs required to expand the program by one resident each year. The commenter stated that the only specialty training programs that could reasonably be expected to expand by one resident are transitional year programs and one-year fellowship programs. The commenter stated that while these are important specialties to expand, they do not believe it was Congress's intent to incentivize training in just these programs. The commenter stated that the CMS proposal leaves little incentive for hospitals to apply for these slots for psychiatry, primary care, general surgery, geriatrics, or other shortage specialties if hospitals are likely to be responsible for most of the cost of expanding or starting these programs. The commenter stated that they note that in a separate section of the proposed rule, which discusses how to evaluate new residency programs for rural-based or small programs, CMS states, “[W]e solicit comment on defining a small residency program as a program accredited for 16 or fewer resident positions, because 16 positions would encompass the minimum number of resident positions required for accredited programs in certain specialties, such as primary care and general surgery, that have historically experienced physician shortages, and therefore have been prioritized by Congress and CMS for receipt of slots under sections 5503 and 5506 of the Affordable Care Act [emphasis added].” The commenter stated they agree with CMS that Congress has repeatedly prioritized these specialty programs, and they encourage CMS to use the implementation of section 4122 to continue to prioritize these and other shortage specialty programs.
Another commenter stated that if more than 200 applicants apply, the resulting award would be pro-rated FTEs and if the number of applicants exceed 400, the award would be virtually unworkable for many programs. The commenter stated that from a sustainability standpoint, it is operationally preferrable to have CMS guarantee an award of at least 1.00 FTE, and ideally to fund entirely in 1.00 FTE increments. Another commenter requested that CMS provide a minimum of 1.0 FTE to each qualifying hospital.
Response: We understand the commenters' concerns that a fraction of an FTE does not provide for the resources necessary to allow for a significant expansion or for the establishment of a new program without additional funding sources. However, we note that we are bound by the language of section 1886(h)(10)(B)(iii), which states “[t]he Secretary shall ensure that each qualifying hospital that submits a timely application under subparagraph (A) receives at least 1 (or a fraction of 1) of the positions made available under this paragraph before any qualifying hospital receives more than 1 of such positions.” Given that there are over 1,000 teaching hospitals and the likelihood that many of these hospitals qualify for additional slots under at least one eligibility category, committing to a prorated distribution that exceeds 1.00 FTE may conflict with the statutory requirement to distribute at least a fraction of an FTE to each qualifying hospital. In addition, while we acknowledge the challenges associated with finding alternative funding streams, we note that the Medicare GME program, as currently structured in the statute, is not intended to function as the only financing source for residency training.
After consideration of the comments received, we are finalizing our policies as proposed with respect to the pro rata distribution of slots under section 4122, without modification. Specifically, we will first distribute slots by prorating the available 200 positions among all qualifying hospitals such that each qualifying hospital receives up to 1.00 FTE, that is, 1.00 FTE or a fraction of 1.00 FTE up to two decimal places. If residency positions are awarded based on a fraction of 1.00 FTE, each qualifying hospital would receive the same FTE amount.
The following section includes a summary of the comments and our responses related to the alternative considered for the prioritization of slots under section 4122 of the CAA, 2023. We considered an alternative approach to distributing the 200 residency slots under section 4122 of the CAA, 2023, which would place greater emphasis on the distribution of additional residency positions to hospitals that are training residents in geographic and population HPSAs. Under this approach, the statutory requirement that each qualifying hospital receive 1 slot or a fraction of 1 slot would be met by awarding each qualifying hospital 0.01 FTE. The remaining residency slots would be prioritized for distribution based on the HPSA score associated with the program for which each hospital is applying using the HPSA prioritization methodology we finalized for purposes of implementing section 126 of the CAA, 2021 ( 86 FR 73434 through 73440 ). To illustrate, if 1,000 qualifying hospitals were to apply under section 4122 of the CAA, 2023, we would first award each qualifying hospital 0.01 FTEs, resulting in the distribution of 10.00 FTEs (1,000 × 0.01). We would then distribute the remaining 190 slots (200−10) based on the HPSA prioritization method we finalized for implementation of section 126 of the CAA, 2021, such that applications associated with higher HPSA scores would receive priority.
We believed that under this alternative distribution methodology we would further the work achieved by section 126 of the CAA, 2021, by distributing residency slots to underserved areas in greatest need of additional physicians. Using this alternative distribution methodology, we would limit a qualifying hospital's total award under section 4122 of the CAA, 2023, to 10.00 additional FTEs ( print page 69364) consistent with section 1886(h)(10)(C)(i) of the Act. Consistent with the methodology we use for implementation of section 126 of the CAA, 2021, as part of determining eligibility for additional slots, we would compare the hospital's FTE resident count to its adjusted FTE resident cap on the cost report worksheets submitted with its application. If the hospital's FTE count is below its adjusted FTE cap, the hospital would be ineligible for its full FTE request. We note that in calculating the adjusted FTE cap we do not consider adjustments for Medicare GME Affiliation Agreements, since these adjustments are temporary. We sought comment on this alternative proposal, including awarding each qualifying hospital 0.01 FTEs and use of HPSA scores to determine priority for remaining slots.
Comment: Several commenters did not support CMS' alternative distribution proposal. According to commenters the alternative distribution proposal would award FTEs to qualifying hospitals in an amount that would be too low to meaningfully increase residency training in qualifying hospitals, particularly during a period of time with significant projected workforce shortages and would result in an overreliance on the HPSA prioritization methodology. Commenters also noted that the alternative distribution proposal, if implemented, would create an administrative burden on hospitals as they would have to potentially account for an increase of 0.01 FTE on their cost reports. Additionally, commenters referenced the statutory language under 1886(h)(10)(C)(ii) which states that hospitals awarded slots under section 4122 agree to increase the total number of full-time equivalent residency positions under the approved medical residency training program of such hospital by the number of such positions made available by such increase under this paragraph. According to commenters, hospitals could be obligated to demonstrate an increase in the program's FTE resident count consistent with an award, even if the award was too minimal to represent a full FTE.
A few commenters stated that although they believe that the alternative distribution proposal would not benefit the expansion of rural residency programs, it would provide rural hospitals with a better chance of receiving new positions. The commenters explained that if a high number of hospitals apply for residency positions under section 4122, under the alternative distribution methodology, there would be more slots leftover to be distributed to each of the four categories, prioritized by HPSA score compared to the other proposed distribution method. According to a commenter, the alternative distribution method creates more potential for rural hospitals to receive multiple slots whereas the other proposed distribution method would make it less likely that rural hospitals would receive more than 1.0 FTE if 200 or more hospitals apply. Commenters referenced round 1 of the distribution of residency positions under section 126, where 291 hospitals applied for residency positions, to support their projection that 200 or more hospitals were likely to apply for the distribution of section 4122 residency positions. The commenters stated that rural hospitals likely need to receive 3-5 residency positions to fully fund a resident for an entire residency and that the alternative distribution methodology gives these hospitals the best chance for that outcome, whereas the other distribution methodology would provide each qualifying hospital with about 0.68 FTE with no remaining residency positions available for distribution.
Response: We thank the commenters for their feedback on the prioritization method described in the “Alternatives Considered” portion of the proposed rule. For the commenters who stated that under the alternative considered rural hospitals may be able to receive more slots, we share the commenters' goal of ensuring hospitals with residency programs that are serving HPSAs are able to experience opportunities to grow and better meet the healthcare needs of the communities they serve. We encourage rural applicants to reach out to CMS directly with any questions or concerns related to the section 4122 application process and as noted above, we will continue to engage with and provide outreach to potential rural applicants.
For the several commenters opposed to the alternative considered, we agree that an increase of only 0.01 FTE may not make a significant enough impact to allow a program to begin or expand. We also agree that there is significant burden associated with having to account for 0.01 FTE on a cost report and to attest that the hospital was able to meet the statutory requirement to increase the total number of FTE residency positions in their residency program by 0.01 FTE. Under the proposed methodology, applicants also have the potential to be awarded a fraction of an FTE, but that amount would likely be higher than 0.01 (based on the number of qualifying hospitals that apply), and therefore provide a relatively larger FTE cap increase. We refer readers to the table earlier in this section which provides examples of how the 200 slots would be prorated based on the number of qualifying applicants.
After consideration of the comments received and the limited support for the alternative considered, we are not finalizing the alternative methodology.
We proposed that if any residency slots remain after distributing up to 1.00 FTE to each qualifying hospital, we would prioritize the distribution of the remaining slots based on the HPSA score associated with the program for which each hospital is applying. Taking an example from the table in the previous section, if 180 qualifying hospitals apply under section 4122 of the CAA, 2023, each qualifying hospital would receive 1.00 FTE and the 20 remaining residency positions would be prioritized for distribution based on the HPSA score associated with the program for which each hospital is applying. We proposed the HPSA prioritization methodology would be the methodology we finalized for purposes of section 126 of the CAA, 2021 ( 86 FR 73434 through 73440 ). We believe including such a prioritization would further support the training of residents in underserved and rural areas thereby helping to address physician shortages and the larger issue of health inequities in these areas. Using this HPSA prioritization method, we proposed to limit a qualifying hospital's total award under section 4122 of the CAA, 2023, to 10.00 additional FTEs, consistent with section 1886(h)(10)(C)(i) of the Act. Consistent with the methodology we use for implementing section 126 of the CAA, 2021, as part of determining eligibility for additional slots, we would compare the hospital's FTE resident count to its adjusted FTE resident cap on the cost report worksheets submitted with its application. If the hospital's FTE count is below its adjusted FTE cap, the hospital would be ineligible for its full FTE request, because the facility had not yet fully utilized the already-allotted slots. We note that in calculating the adjusted FTE cap we would not consider adjustments for Medicare GME Affiliation Agreements since these adjustments are temporary.
We proposed that as finalized under section 126 of the CAA, 2021 ( 86 FR 73435 ), for purposes of prioritization under section 4122 of the CAA, 2023, primary care and mental-health-only population and geographic HPSAs ( print page 69365) would apply. As discussed in the final rule implementing section 126 of the CAA, 2021, each year in November, prior to the beginning of the application period, CMS would request HPSA ID and score information from HRSA so that recent HPSA information is available for use for the application period. CMS would only use this HPSA information, HPSA ID's and their corresponding HPSA scores, in order to review and prioritize applications. To assist hospitals in preparing for their applications, the HPSA information received from HRSA will also be posted when the online application system becomes available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/DGME . The information would also be posted on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/IPPS-Regulations-and-Notices . Click on the link on the left side of the screen associated with the appropriate final rule home page or “Acute Inpatient—Files for Download” ( 86 FR 73445 ).
Given that residency slots under section 4122 of the CAA, 2023 are to be distributed in FY 2026, we proposed that the HPSA IDs and scores used for the prioritization of slots, if applicable, would be the same HPSA IDs and scores used for the prioritization of slots under round 4 of section 126 of the CAA, 2021. This group would include HPSAs that are in designated or proposed for withdrawal status at the time the HPSA information is received from HRSA. As noted in section j. of this preamble, CMS would request HPSA data from HRSA in November 2024 to be used for purposes of section 4122 of the CAA, 2023.
In this section we present a summary of the public comments and our responses related to prioritizing the distribution of slots by HPSA score for purposes of the section 4122, if any slots remain after awarding each qualifying hospital up to 1.00 FTE.
Comment: A few commenters supported the proposal to prioritize the distribution of slots by HPSA score if any slots remain after awarding each qualifying hospital up to 1.00 FTE.
A commenter stated they advocated for and are deeply supportive of CMS' proposal to apply the same methodology for distributing the new slots that was finalized for the slots enacted by section 126 of the 2021 CAA, including the proposal to require hospitals that serve areas designated as HPSAs to have at least 50 percent of residents' training time occur at training locations within a primary care or mental health-only geographic HPSA in order to be able to apply for new GME slots. The commenter stated that they strongly believe continuing this equity-focused methodology would help mitigate health access disparities and more effectively address physician shortages.
Another commenter stated that they encourage CMS to prioritize the distribution of slots by awarding to primary care programs and that they support the proposal to prioritize the distribution of remaining slots by HPSA score. The commenter stated that they believe such a prioritization would ensure that an appropriate number of the new residency positions would go to the hospitals where they would have the greatest impact on access to care—where there were well-documented shortages in primary care and other internal medicine subspecialties. The commenter stated that this HPSA-based approach would not only address the current maldistribution of the physician workforce and mitigate workforce shortages in primary care, including general internal medicine, but also address health inequities and reach underserved populations.
Response: We appreciate the commenters' support. We note that while the law requires that at least half of the 200 slots be distributed to psychiatry programs or subspecialties of psychiatry, it does not limit the specialties or subspecialties that are eligible to apply for the remaining positions.
Comment: A commenter stated that they agree the HPSA designation would be useful for identifying underserved geographies and some patient populations that are disproportionately impacted by the addiction crisis, such as people experiencing homelessness and those who are eligible for Medicaid. The commenter noted the exclusion of clinicians who specialize in treating substance use disorder (SUD) from the list of core health professionals used to define the current mental health HPSA designation. The commenter stated that this definition does not include addiction medicine physicians nor certified addiction registered nurses—advance practice (CARN-AP), despite areas with “a high degree of substance abuse” being included in the determination of “unusually high needs for mental health services” criterion. The commenter urged Federal agencies, including CMS, to work with HRSA to revise the mental health HPSA definition and related criterion to include clinicians that specialize in treating SUD, particularly addiction medicine physicians and CARN-APs. The commenter stated that including these clinicians would more accurately measure the SUD treatment workforce and allow residency positions and other funding opportunities to be better targeted to underserved areas with high SUD and overdose burdens but limited treatment access.
Response: We appreciate the commenter sharing their concerns related to the exclusion of clinicians who specialize in treating substance use disorder from the list of core health professionals used to define the current mental health HPSA designation. The list of core health professionals used to define the current mental health HPSA designation is outside the scope of this rulemaking, but we will share the commenter's concerns with HRSA.
Comment: Numerous commenters expressed concern with the proposal to prioritize the distribution of slots by HPSA score if any slots remain after awarding each qualifying hospital up to 1.00 FTE. Commenters stated that the proposed HPSA prioritization is not consistent with legislative or statutory intent. A commenter stated that prioritizing hospitals located in HPSAs deviates from the statute, which states that slots are to be distributed to hospitals that serve HPSAs. The commenter stated that limiting distribution priority to hospitals located in HPSAs may inadvertently disqualify hospitals that disproportionately serve large numbers of low income and underserved individuals, particularly because HPSAs presumably do not have many access points for healthcare services.
A commenter stated that they do not believe that Congress intended for CMS to revert to the methodology used under section 126. The commenter stated that Congress newly added the pro rata distribution in section 4122 as a directive to CMS to not simply use the same methodology as was used in section 126. The commenter stated that the fact CMS is using that same methodology after implementing the pro rata distribution seems to fly in the face of how Congress deliberately modified the distribution methodology for section 4122 from what was included in section 126. The commenter stated that had Congress wished to create a “super prioritization” and focus on hospitals that train residents in HPSAs, it would have done so. The commenter stated that prioritization using HPSAs favors rural areas over urban areas and that according to their analysis, 75.4 percent of HPSAs are rural or partially rural, and rural and partially rural HPSAs are disproportionately represented among those HPSAs with the highest scores. The commenter stated they do not ( print page 69366) believe that it was the intention of section 4122 to prioritize rural applicants in this manner. Rather, Congress set up the prioritization among hospitals in creating the four categories of qualifying hospitals and specifying that a minimum of 10 percent of the slots must be distributed to hospitals in each of those four categories. The commenter stated that the only further prioritization needed for section 4122 was to determine how to prioritize applicant specialty programs as was done in section 5503 of the Affordable Care Act.
Several commenters conveyed their opposition to what they believe is CMS' overreliance on HPSA scores in the distribution of slots and stated HPSAs should be used sparingly. The commenters stated the HPSA prioritization has no foundation in the enabling legislation and that it is inherently unfair to deserving hospitals that may qualify for new residency slots in the other three (nonHPSA) categories. The commenters stated that CMS noted in the past that its methodology does not intend to exclude hospitals that do not serve HPSAs from receiving new residency slots, but regardless of this intention, commenters argued this could ultimately be a result of continuing to rely so heavily on HPSAs. A few commenters referred to an analysis from the Alliance of Safety Net Hospitals, which found that giving exclusive priority to applications from hospitals with high HPSA scores would have this effect and that time has proven this analysis to be accurate; they suggested that this has made vast parts of the country virtually ineligible for new residency slots. The commenters further stated that this outcome does not reflect Congress's intention when it authorized the new residency slots.
A commenter stated that for Pennsylvania, where HPSAs exist in all corners of the commonwealth but the individual HPSA scores are much lower than they are in other states, giving exclusive priority to applications from hospitals with high HPSA scores has the effect of excluding almost all Pennsylvania teaching hospitals from this program even when they meet the statutory criteria. The commenter stated that this outcome does not reflect Congress's intention when it authorized the new residency slots.
A commenter stated that Wisconsin has currently seen no new slots awarded, despite having multiple entities that fit at least one, if not more, of the four criteria in statute. Rather, they state that the majority of slots CMS awarded so far were not distributed to geographically rural hospitals, but rather, urban and suburban hospitals that serve rural patients, and notes that this is not following Congressional intent.
A commenter urged the agency to also consider the population and communities that a hospital system serves when awarding residency slots. The commenter stated that while it is headquartered in two small Wisconsin cities, its hospital system serves rural communities. The commenter stated that because of its headquarters, its hospital frequently does not score high enough to receive additional slots and requests CMS consider the hospital system's service area, not just the headquarters, when distributing the new residency positions.
Response: We thank the commenters for their comments. We remind readers that the HPSA prioritization does not require that the applicant hospital be located in a HPSA. Rather, at least 50 percent of the training time associated with the program for which the hospital is applying must occur at training sites located within the primary care or mental-health-only population or geographic HPSA. Given the number of applications we have received under the first three rounds of section 126, which request a HPSA to be used for purposes of prioritization, we do not believe that there is a shortage of access points that can be used as training sites for purposes of meeting the 50 percent HPSA prioritization criterion.
In addition, we do not agree that the proposed methodology for the distribution of slots under section 4122 exhibits an overreliance on HPSA scores or is inconsistent with the law. The prioritization of HPSA scores is only part of the distribution process under section 4122 and applies after each qualifying hospital receives up to 1.00 FTE as required by statute, which means qualifying hospitals in states with limited HPSAs will still receive FTE cap increases under section 4122. As noted earlier, section 4122 added this requirement not found in section 126. There is no added provision of section 4122, which was enacted after our implementation of section 126, that precludes the use of HPSA scores for purposes of prioritization. If there are any remaining slots to be distributed after each qualifying hospital receives up to 1.00 FTE as required by statute, there needs to be some prioritization if the number of slots requested across all hospitals exceeds the number of slots authorized under section 4122. Allocation by the severity of the health professional shortage in a HPSA is a reasonable and transparent prioritization approach. If a program serving a particular HPSA, or programs serving HPSAs in a particular state, do not receive additional slots under section 4122 that does not mean that those areas have sufficient health professionals. Rather, it is a reflection that the number of slots authorized by section 4122 is less than the requested number of slots from applicant hospitals with teaching programs that serve HPSAs.
Comment: Several commenters expressed concern that prioritization of applications by HPSA score may negatively impact rural hospitals. A few commenters stated that such a prioritization removes from the distribution some rural hospitals that are ready and able to grow their residency programs.
A few commenters stated that during the first two rounds of the section 126 slot distributions, only 7 geographically rural hospitals received slots. The commenters stated that they believe high HPSA scores serve as a barrier to entry for rural hospitals seeking slots because HPSA scores often do not prioritize or accurately reflect the needs of areas with small populations. The commenters noted that three primary factors are used in scoring criteria across primary care, mental health, and dental HPSAs: population-to-provider ratio; poverty rates; and travel distance or time to the nearest accessible source of care. They further noted that there is no measure to account for rurality or unique access problems associated with rural areas. Another commenter questioned whether all states comprehensively and accurately survey and present data to have HSPAs be the best measure for rural health care access.
Commenters opined that the health needs measured by HPSAs are not reflective of the needs of older populations. A commenter stated that the higher utilization of services by older adult populations in rural areas and their related risk factors are not accounted for in the current HPSA scoring methodology. Another commenter stated that the existing components that factor into a HPSA score are not reflective of access problems that many rural areas face. The commenter stated that the older adult populations of rural areas result in higher utilization of health services, and their respective risk factors are not accounted for in the existing HPSA formula. The commenter stated that unless the HPSA methodology is updated to reflect these concerns, they do not believe that basing distribution of the additional residency slots on the HPSA score alone will provide for GME ( print page 69367) funding to go to areas that could most use the additional resources from CMS.
A few commenters stated that if plans for section 4122 mirror section 126 there would be continued limits on allocations to geographically rural hospitals. Commenters stated that some geographically rural hospitals may have lower HPSA scores or be discouraged from applying when they are not located in a HPSA. Commenters stated that updating rurality to CMS defined criteria (that is, non-metropolitan training sites) and allocating at least 10 percent of those slots to rural areas regardless of HPSA score may better align with legislative intent. Commenters stated that equally important is eliminating the application of HPSA prioritization from within the rural category. Commenters stated that many rural hospitals are saddled with low HPSA scores as an artifact of the methodology for calculating those scores and are thus eliminated from consideration even though they are serving communities most in need of new physicians. Commenters asked CMS to consider changing the definition of which hospitals qualify for “rural” categorization to eliminate hospitals “treated as rural” that are not in fact geographically rural and include in the “rural” categorization all hospitals located in rural geographic locations regardless of HPSA score. The commenters requested that if eliminating urban hospitals “treated as rural” is not possible, CMS continue the HPSA score prioritization for those urban hospitals.
Another commenter stated they are concerned that the proposal to prioritize slots based on HPSA score will unnecessarily end up excluding hospitals that no longer reside in HPSAs due to HRSA's misguided shortage designation modernization project that, while well-intended, is exacerbating challenges for rural hospitals. The commenter stated that, for example, around 25 Wisconsin hospitals lost their HPSA designations at the start of 2024 due to the way the HRSA updated its HPSA renewal process. The commenter stated that some applicants have reported their concerns that relying too much on HPSA scores has unfairly led to the exclusion of their GME slot applications from consideration and has further discouraged other interested applicants from expending resources on an application that is unlikely to result in an award.
Another commenter stated that due to their smaller populations, rural communities that add new physicians as faculty and retain residents, can significantly shift their HPSA scores or lose their HPSA designation, which can prevent a hospital in a rural area from receiving GME slots based on current CMS policy.
Response: We appreciate the detailed analysis submitted by the commenters regarding their concerns that prioritizing the distribution of slots by HPSA score may not benefit rural hospitals. We acknowledge that few geographically rural hospitals have submitted applications under rounds 1 and 2 of section 126. We believe, based on our experience to date under section 126, that the reasons for this may be more complex than the existence of the HPSA prioritization. For example, rural hospitals may be utilizing other opportunities to increase their FTE caps through section 127 of the CAA, 2021, which provides FTE cap increases for participation in rural training programs and the regulatory provision at section 413.79(e), which allows rural hospitals to receive an increase in their cap each time they participate in training residents in a new program. We acknowledge that rural hospitals may find these alternatives more worthwhile as they may allow for permanent FTE cap increases that exceed those available under section 126 and section 4122. We believe that continuing education and outreach regarding the opportunities available under both sections 126 and 4122, rather than abandoning the HPSA prioritization method which has successfully allocated slots to programs serving underserved communities and populations, is the appropriate course of action at this point. We will continue to monitor this issue. As stated previously, we encourage rural hospitals to reach out to CMS directly with questions they have about the section 4122 application process.
Comment: Several commenters stated that HPSAs are not necessarily the best measure of where residents should train. A commenter stated that HPSA scores were developed to determine priorities for the assignment of clinicians in a state, not to determine the ability of the hospitals in those states to train more residents or to provide care for patients who live in HPSAs. Another commenter stated that the HPSA designations are a measure of a shortage of providers but do not consider whether a hospital can train residents through academic medical programs within the HPSA area. The commenter stated that CMS should recognize that there is an overall shortage of physicians, particularly in psychiatry. The commenter stated that it should only matter that additional physicians are available to meet demand, not where the physicians are trained and that incentives directed towards newly trained physicians to practice in a HPSA is a more effective method to address the particularly high portion of the physician shortage experienced by HPSAs. Another commenter stated that while HPSA scores may adequately indicate places in the country where there is a need for more providers, they may not be the best representation of where hospitals are prepared to provide the best and most complete training environment. The commenter stated that they applaud CMS for focusing on underserved areas and strongly encouraged the agency not to rely too heavily on a single metric and ensure residents are given the best opportunity for a well-rounded training experience. Another commenter stated that over the last two distribution cycles, it has heard from many frustrated institutions that are adjacent to a HPSA, but resident training time does not take place in a HPSA. These institutions need additional slots to expand training and treat HPSA populations but are not eligible to receive prioritization in the distribution of section 126 awards.
Response: We understand that training sites may be located adjacent to HPSAs and provide essential care for individuals living within those HPSAs. However, due to the limited number of FTE slots available under section 4122 that could be prioritized by HPSA score (after completion of the pro rata distribution requirement), we are choosing to prioritize training time in HPSAs in order to further support the likelihood of residents choosing to practice in these areas. While we disagree that hospitals located in HPSAs may not provide the best and most complete training environment, we note again that the applicant hospital itself is not required to be physically located in the HPSA in order for the program to meet the 50 percent criterion for HPSA prioritization. Furthermore, we believe that increasing residency training in non-provider sites outside of hospitals, such as community health clinics located in HPSAs, is an important tool in addressing the shortage of primary care providers in underserved areas. We continue to welcome ideas for a clear and accessible prioritization methodology, which would include providers located adjacent to a HPSA that provide significant patient care to individuals living within the HPSA.
Comment: Commenters suggested that CMS consider alternatives to prioritizing slots based on HPSA score and advocated for relying more heavily on the other eligibility categories. A commenter stated that the HPSA construct is antiquated and that gaining HPSA status starts with a costly undertaking by state governments, and increasingly, state governments are proving reluctant to make this investment—and even when they do, the process is burdensome. The commenter stated that HPSA status depends in part on an area's level of poverty, but this is an uneven playing field because it costs more to live in high-cost areas. The commenter stated that using HPSAs as a major part of the criteria consequently favors—unfairly—some areas over others and therefore should be used sparingly, if at all, and it significantly undermines the other three criteria for additional residency slots. The commenter urged CMS to withdraw this proposal and develop an alternative methodology for distributing residency slots that does not rely so heavily on HPSAs and gives greater weight to the other three criteria for new slots.
Several commenters stated that CMS should evaluate the application pool and, if able to meet the statutory distribution requirements, award all slots on a pro-rata basis. The commenters stated that if CMS is unable to meet the statutory requirements using this methodology, CMS should prioritize the remaining slots or pro rata slots to hospitals that meet all four qualifying categories listed above first; then hospitals that meet three criteria and so forth, until all slots are distributed.
A commenter noted that they do not believe that training residents in HPSAs is an appropriate measure of reducing health inequities, which was CMS's stated goal in implementing this distribution methodology for section 126. The commenter stated that while they agree that HPSA designation is a good starting point for identifying an area that needs more physician services, the designation system for HPSAs is not without controversy. The commenter suggested that a more holistic approach to addressing the physician shortage would be to recognize medical education hubs such as those located in densely populated and diverse urban areas because training residents in densely populated urban areas with a diverse set of patients is the single best means of exposing physicians in training to the cultural complexities that CMS should want all physicians exposed to during their training to promote health equity. The commenter stated that CMS should review data indicating which areas of the country and which organizations are producing physicians for HPSAs. The commenter stated that New York's teaching hospitals for example are a major “feeder” for the rest of the nation's physician workforce and included data supporting their statement. The commenter stated that after the initial pro rata distribution is complete, CMS should prioritize making those applications more “whole” by awarding the applicant as many of the number of slots that is commensurate with their planned expansion of existing residency programs or establishment of new programs. The commenter stated that CMS should accomplish this process by prioritizing those hospitals that meet all four qualifying criteria first, and then hospitals that meet three criteria and so on until all slots are distributed. The commenter stated that if CMS determines that not enough slots remain to make all hospitals that received a pro rata distribution whole, it should prioritize doing so for psychiatry and psychiatry subspecialty programs. The commenter stated they believe that this approach would more closely align with the intent of the legislation to prioritize residency slots for psychiatry programs while also operating within the requirements that each applicant receive at least one (or a fraction of one) of the residency positions made available. The commenter stated that if there are not enough slots to make all hospitals that received a pro rata distribution whole, CMS should allow these hospitals to apply for the number of slots that would make the program whole in round five of the section 126 distributions. These slots would be effective July 1, 2027. Using both distributions to make an application whole would allow hospitals to expand and start new programs more easily.
A commenter stated that Congress has voiced concerns about a shortage of physicians serving rural areas and referred to data from U.S. House Committee on Ways & Means. The commenter stated they agree with the findings of the Committee on Ways & Means and believe that physicians who participate in rural residency programs are more likely to practice in underserved rural areas. The commenter encouraged CMS to implement a process by which the first round of slots are granted to hospitals located in areas that truly are rural. Once those slots have been awarded, they recommend CMS distribute remaining slots as proposed.
A commenter recommended CMS consider distributing slots in areas where there are high rates of maternal mortality. The commenter stated that when considering residency and fellowship positions, they believe it would be beneficial to take this data into account, coupled with geographic areas with high rates of sickle cell diseases and other hemoglobinopathies. The commenter stated that this approach might not always align with traditional HPSA delineations, but they believe it is worth exploring given the serious hematologic needs of these patients.
A commenter stated that they do not believe CMS should finalize a distribution for new residency positions that incorporates a HPSA prioritization and, consistent with their prior comments, they encourage CMS to work more closely with the GME community regarding distribution.
A commenter urged CMS to consider historical state-by-state distribution of GME slots. The commenter stated that the caps established through the Balanced Budget Act of 1997 created an inequity in states that did not have robust residency programs at the time but have had significant population growth since the 1997 caps were implemented. The commenter stated that it is critical to develop a workforce that can meet the needs of a state's population and that around two-thirds of doctors live in the state they train in. The commenter stated that Florida, which will have an expected shortage of more than 18,000 physicians by 2035, is one of the nation's fastest growing states, and has the second largest number of Medicare beneficiaries in the country. The commenter stated that it is in the interest of the Medicare program to ensure that Florida has enough physicians, and this requirement could be met by increasing the number of physicians trained in the state. The commenter stated that CMS should give preferential consideration to states that are historically underserved by the Medicare GME program and states that have a large Medicare population.
A commenter stated that CMS should prioritize the distribution of new resident slots to essential hospitals. The commenter stated that essential hospitals are committed to training the next generation of health professionals and equipping them with the necessary skills to provide culturally and linguistically competent care to all populations, including underrepresented and marginalized people. The commenter stated that because essential hospitals play such a unique and critical role in preparing ( print page 69369) health care professionals to care for underserved populations, prioritizing the distribution of residency slots to essential hospitals would help advance CMS' equity goals. Another commenter stated that it is not located in a primary care or mental health HPSA. The commenter stated that assuming that this situation is likely the case for many other urban safety-net hospitals, these hospitals are categorically disadvantaged under the proposed distribution methodology. The commenter recommended that CMS adopt a distribution methodology that prioritizes hospitals that serve a high percentage of Medicare, Medicaid, and uninsured patients, or some other measure that accurately targets hospitals that serve low-income patients.
Response: We appreciate the commenters' suggestions of additional ways to prioritize the distribution of slots under section 4122. However, as stated in the final rule implementing section 126, we continue to believe that HPSA scores, while not a perfect measure, provide the best prioritization approach available at this time. They are transparent, widely used, publicly available, regularly updated, and have verifiable inputs for measuring the severity of a service area's need for additional providers ( 86 FR 73438 ). We believe the continued use of HPSA scores for prioritization is consistent with the Administration's policy objective to increase residency training and thereby increase the number of physicians practicing in underserved areas.
With respect to prioritizing by eligibility category such that the more eligibility categories the hospital meets the higher its prioritization, we do not believe that this methodology would provide a sufficient level of prioritization since our experience with section 126 to date indicates that many applicants would meet two or three out of the four eligibility categories.
While we agree with the comment suggesting that training residents in medical education hubs, located in densely populated and diverse urban areas, allows residents to gain experience caring for a diverse set of patients and promotes an understanding of cultural complexities necessary for well-rounded patient care, we believe that such a methodology would be limited in that it does not fully consider the advantages of training residents in rural areas.
With respect to making a program whole in round 5 of section 126 if it did not receive all of the slots it was eligible for under section 4122, we do not believe there is any statutory language precluding a hospital from applying for unfilled slots under round 5 of section 126 if it applied for that same program under section 4122.
With respect to prioritizing geographically rural hospitals for the distribution of slots under section 4122, while our goal is to support rural hospitals in applying for additional slots under section 4122, we do not believe we have the authority to distinguish between geographically rural hospitals and hospitals that have reclassified as rural when awarding slots since the statute considers both types of hospitals to be Category One hospitals.
In response to the recommendation that CMS account for areas of high maternal mortality and areas with high rates of sickle cell diseases and other hemoglobinopathies in its prioritization, we agree that these geographic and population groups would benefit from an increased supply of physicians. We are currently unaware of a nationally defined measure that we could incorporate into the HPSA methodology to distribute any slots remaining after the pro-rata distribution of slots, and we welcome feedback on any available measures.
Lastly, we support the general goal of increasing residency training at essential hospitals and safety-net hospitals since they are often the primary means of accessing healthcare for underserved members of the population. However, a lack of a specific, generally accepted, and existing definition of an “essential hospital” or “safety-net hospital” for purposes of GME makes it challenging to concretely incorporate these concepts currently into slot distributions under section 4122.
After the consideration of the comments received, and for the reasons discussed above, we are finalizing our proposal without modification to prioritize the distribution of any remaining slots under section 4122 by HPSA score. Specifically, if any slots remain after awarding up to 1.00 FTE to each qualifying hospital, we will prioritize the distribution of the remaining slots by the HPSA score associated with the program for which the hospital is applying. Primary care and mental-health-only geographic HPSAs are applicable for this prioritization. If a hospital is applying using a mental-health-only HPSA, it must apply for slots for a psychiatry program or a subspecialty of psychiatry. We continue to welcome comments from the GME community related to an alternative means for distributing slots under section 126 and for potential future slot distributions.
Section 1886(h)(10)(C)(iii) of the Act requires that if a hospital that receives an increase in the otherwise applicable resident limit under section 1886(h)(10) of the Act would be eligible for an adjustment to the otherwise applicable resident limit for participation in a new medical residency training program under 42 CFR 413.79(e)(3) (or any successor regulation), the hospital shall ensure that any positions made available under this paragraph are used to expand an existing program of the hospital, and not be utilized for new medical residency training programs. Under the regulations at 42 CFR 413.79(e)(3) , a rural hospital may receive an increase to its cap for participating in training residents in a new program, which is effective after a 5-year cap-building period for that new program. We note that if a rural hospital were to receive a cap increase for a new program under the 5-year cap-building period as well as a cap increase for the new program under section 4122 of the CAA, 2023, there may be duplicative awarding of cap slots for the same program. Therefore, we proposed to implement section 1886(h)(10)(C)(iii) of the Act by allowing rural hospitals to apply for slots to expand an existing program, but not for slots to begin a new program. We proposed that this policy apply to both geographically rural hospitals and hospitals that have reclassified as rural under 42 CFR 412.103 , since both groups of hospitals are considered rural under section 1886(h)(10)(B)(ii)(I), which we refer to as Category One hospitals. Only geographically urban hospitals that have not reclassified as rural under 42 CFR 412.103 would be permitted to apply for slots to begin a new program.
In this section we present a summary of the public comments and our responses related to the requirement that if a hospital is eligible for a cap increase under 42 CFR 413.79(e)(3) (or any successor regulation), the hospital may only apply for section 4122 slots to expand an existing program.
Comment: A few commenters disagreed with CMS' proposal to allow hospitals that have reclassified as rural to receive slots to expand an existing program, but not for a new program.
A commenter stated that they support CMS' proposal that if a hospital is eligible for a cap adjustment for participation a new program (as is the case for rural hospitals and hospitals that have reclassified as rural), the hospital can only use awarded slots to expand an existing program and not for ( print page 69370) a new program. However, the commenter stated that they believe that this limitation should only apply to IME cap adjustments for urban hospitals that have reclassified as rural. The commenter referred to the language in the proposed rule that states, “We note that if a rural hospital were to receive a cap increase for a new program under the 5-year cap-building period as well as a cap increase for the new program under section 4122 of the CAA, 2023, there may be duplicative awarding of cap slots for the same program.” The commenter stated that while they agree with this rationale, urban hospitals that have reclassified as rural only receive adjustments to their IME caps, not their DGME caps. The commenter recommended that CMS allow hospitals that have reclassified as rural to apply for new program slots, but to limit their application to only DGME slots. The commenter further stated that CMS' policy analysis also applies in the case of section 126 of the CAA, 2021. The commenter stated that while Congress did not explicitly state within section 126 that newly awarded slots cannot be used to establish new programs in rural hospitals, they also did not state that newly awarded slots can be used for these purposes. The commenter urged CMS to use its discretion to apply this policy equally to the section 126 slot distribution.
Another commenter stated that they disagree with CMS' proposed limitation on rural reclassified hospitals to apply only for slots to expand an existing program, but not for slots to begin a new program. The commenter stated that while they concur with the potential overlap of cap adjustments for geographically rural hospitals, rural reclassified hospitals cannot generate cap slots for DGME under the regulations at 42 CFR 413.79(e)(3) . The commenter encouraged CMS to allow rural reclassified hospitals to apply for new program slots, but to limit their application to only DGME slots, similar to the current methodology employed for section 126.
Response: We thank the commenters for their comments. The commenters are correct that hospitals that have reclassified as rural can receive IME but not DGME cap adjustments for new programs. The statutory provisions for both IME and rural reclassification are found at section 1886(d), whereas the statutory provision for DGME is included at section 1886(h). Therefore, hospitals that have reclassified as rural are considered rural for IME, but urban for DGME. However, we continue to believe that in including both geographically rural hospitals and hospitals that have reclassified as rural under Category One, the Congressional intent was to treat these two groups of hospitals in the same manner for purposes of cap increases under section 4122.
After consideration of the comments received, we are finalizing our policy as proposed without modification; that is, both geographically rural hospitals and hospitals that have reclassified as rural may apply for section 4122 slots for a program expansion, however, they may not apply for slots for a new program. We are not extending this policy to section 126 because the statutory language that is explicit in section 4122 is not included in section 126. We note that under both provisions, any hospital that is applying for slots for a new program must make sure that the slots for which they are applying are not duplicative of slots they will receive under the normal 5-year cap-building process.
Section 1886(h)(10)(B)(ii) of the Act requires the Secretary to distribute at least 10 percent of the aggregate number of total residency positions available to each of the following categories of hospitals discussed earlier. Given our experience with distributing slots under section 126 of the CAA, 2021, we expect many hospitals will meet the qualifications of more than one category. We proposed to collect information regarding qualification for all four categories in the distribution of slots under section 4122 of the CAA, 2023, to allow us to confirm that we have met this statutory requirement. Like the CAA, 2023 provision, section 1886(h)(9)(B)(ii) of the Act from 2021 also requires the Secretary to distribute at least 10 percent of the aggregate number of total residency positions available to the same four categories of hospitals. Section 126 of the CAA, 2021, makes available 1,000 residency positions and therefore, at least 100 residency positions must be distributed to hospitals qualifying in each of the four categories. In the final rule implementing section 126 of the CAA, 2021, we stated we would track progress in meeting all statutory requirements and evaluate the need to modify the distribution methodology in future rulemaking ( 86 FR 73441 ).
To date, we have completed the distribution of residency slots under rounds 1 and 2 of the section 126 distributions (refer to CMS' DGME web page for links to the round 1 and 2 awards: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/direct-graduate-medical-education-dgme ). In tracking the statutory requirement that at least 10 percent of the aggregate number of total residency positions (100 out 1,000 slots) be distributed to hospitals qualifying in each of the four categories, we have determined that in rounds 1 and 2, only 12.76 DGME slots and 18.06 IME slots were distributed to hospitals qualifying under Category Four. For each of the other 3 categories based on the slots awarded in rounds 1 and 2, we anticipate meeting the 10 percent requirement. For example, we have determined that in rounds 1 and 2, 374.59 DGME and 375.11 IME slots were distributed to hospitals qualifying under Category Three.
As discussed in the final rule implementing section 126 of the CAA, 2021, an applicant hospital qualifies under Category Four if it participates in training residents in a program in which the residents rotate for at least 50 percent of their training time to a training site(s) physically located in a primary care or mental-health-only geographic HPSA. Specific to mental-health-only geographic HPSAs, the program must be a psychiatric or a psychiatric subspecialty program ( 86 FR 73430 ). Given that only 12.76 DGME slots and 18.06 IME slots have been distributed to hospitals qualifying under Category Four, we proposed an amendment to our prioritization methodology for rounds 4 and 5 of section 126 of the CAA, 2021, to ensure that at least 100 residency slots are distributed to these hospitals. We did not propose an amendment to our prioritization methodology for round 3 because the application period for round 3 runs from January 9, 2024 to March 31, 2024, prior to the date any proposals in this rule might be finalized.
Our current methodology for distributing residency slots under section 126 prioritizes slot awards based on the HPSA score associated with the program for which the hospital is applying, with higher scores receiving priority ( 86 FR 73434 through 73440 ). We proposed that in rounds 4 and 5 of section 126 of the CAA, 2021, we would prioritize the distribution of slots to hospitals that qualify under Category Four, regardless of HPSA score. The remaining slots awarded under rounds 4 and 5 would be distributed using the existing methodology based on HPSA score ( 86 FR 73434 through 73440 ). That is, the remaining slots would be distributed to hospitals qualifying under Category One, Category Two, or Category Three, or hospitals that meet the definitions of more than one of these categories, based on the HPSA score ( print page 69371) associated with the program for which each hospital is applying.
In this section we present a summary of the public comments and our responses related to (1) distributing at least 10 percent of the aggregate number of total residency positions available to each of the four eligibility categories under section 4122 of the CAA, 2023; and (2) prioritizing the distribution of slots to hospitals that qualify under Category Four, regardless of HPSA score, for rounds 4 and 5 of section 126 of the CAA, 2021.
Comment: A few commenters had concerns related to meeting the requirement that at least 10 percent of the total number of slots be distributed to each of the four eligibility categories under section 4122 of the CAA, 2023. Commenters stated that CMS has not addressed the structural shortcoming of the HPSA prioritization distribution methodology and has not established how the agency would meet the 10 percent statutory distribution requirement for section 4122 slots. Commenters stated that it is crucial for CMS to ensure that the distribution process is in full compliance with the law, as any deviation could have serious implications for the fairness and effectiveness of the program. A commenter emphasized the need to find another prioritization metric to avoid the maldistribution between categories of hospitals that occurred under section 126 when distributing section 4122 slots. Commenters stated that CMS' proposal to prioritize Category Four hospitals for rounds 4 and 5 of section 126 could have been avoided if CMS had considered factors beyond HPSA scores as part of the section 126 distribution prioritization. Commenters stated that CMS would likely face the same issue with the section 4122 slot distribution and that CMS should explain to stakeholders how the agency would ensure that 10 percent of slots are distributed to the four categories of qualifying hospitals.
Commenters stated that they were similarly concerned with CMS' proposal to prioritize hospitals serving HPSAs for rounds 4 and 5 of section 126. The commenters urged CMS to prioritize slot distribution based solely on the four categories included in the law because they believe such an approach was consistent with the statute, which would be less burdensome, and offer a much clearer metric for qualifying hospitals.
Response: We thank the commenters for raising their concerns related to whether CMS can meet the statutory requirement to distribute at least 10 percent of section 4122 slots to each of the four categories of qualifying hospitals. We do not necessarily agree that we will be unable to meet this statutory requirement; the methodology for distributing section 4122 slots, as finalized in this rule, includes two parts—distributing up to 1.00 FTE to each qualifying hospital and then prioritizing the distribution of the remaining slots based on the HPSA score of the program for which the hospital is applying. However, in the event that we are unable to meet the statutory requirement in a single round, we would take a similar approach to the approach we are taking with section 126. We also note that we are not amending the prioritization methodology for rounds 4 and 5 of section 126 to consider the number of eligibility categories that a hospital meets. As stated above, we do not believe that this methodology would provide a sufficient level of prioritization since our experience with section 126 to date indicates that many applicants would meet two or three out of the four eligibility categories.
Comment: Several commenters generally supported the proposal to prioritize Category Four applicants in rounds 4 and 5 of section 126. A commenter stated that hospitals qualifying in the HPSA category (Category Four) have been left behind compared to hospitals that have qualified in the other categories. The commenter stated they appreciate CMS recognizing this discrepancy and prioritizing hospitals that qualify in this category regardless of their HSPA score for future distribution of residency slots. However, the commenter stated that they disagree with smaller hospitals being prioritized over larger hospitals in case of a tie when prioritizing applications with equal HPSA scores. The commenter stated they believe prioritizing smaller hospitals is doing a disfavor to future residents because larger hospitals are usually the primary teaching sites for programs, are better able to add residency positions, and provide more diverse and comprehensive training environments, and thus more training opportunities for residents.
A few commenters suggested CMS prioritize applications from geographically rural hospitals regardless of HPSA score. One commenter stated that they appreciate CMS' careful tracking of the round 1 and 2 slot distributions related to section 126 of the CAA, 2021. The commenter stated that while it is unfortunate that Category Four hospitals did not have their slots filled during rounds 1 or 2, the commenter is broadly supportive of CMS' proposed amendment to their prioritization methodology for rounds 4 and 5. However, the commenter stated that although they support the proposal to prioritize Category Four hospitals, the current HPSA methodology limits the ability of many geographically rural hospitals to receive slots, as their HPSA scores are not high enough or they are not located in a HPSA. The commenter stated that to better align with legislative intent going forward, CMS should consider updating its definition of rural to align with other CMS-defined criteria (all people and territory in an area with less than 50,000 people) and using that parameter to allocate at least 10 percent of slots to rural areas, regardless of HPSA score. The commenter stated that they applaud the work CMS has undertaken in recent years to promote health and health equity in rural and underserved communities and believe this change would support goals of delivering better care where patients most need it. The commenter stated that evidence indicates that physicians typically practice within 100 miles of their residency program and therefore, the distribution of trainees in large academic hospitals leads to physician shortages in medically underserved and rural areas. The commenter stated that family medicine is also facing a particularly critical workforce shortage and directing Medicare GME resources to underserved areas is an essential strategy for advancing health equity.
A commenter stated that the requirement that 10 percent of slots go to each of the four categories of qualifying hospitals helps to ensure appropriate distribution of training slots to the communities that need them, however, this goal should not be undermined by a policy design that results in positions being unallocated if there are insufficient applicants in a given category. The commenter stated they appreciate CMS modifying its methodology from the CAA 2021 to address this issue and they urge CMS to ensure that the policy finalized allows all funded slots to be distributed to programs. The commenter stated that they encourage CMS to perform a meaningful estimate of the future distribution of available slots to help ensure that another “catch-up” change in priorities is not needed and hospitals have a consistent and clear metric for applying for new slots.
Response: We thank the commenters for their support. In the FY 2022 IPPS final rule with comment, we finalized the policy of prioritizing hospitals with less than 250 beds in the event a tiebreaker is needed to distribute slots ( print page 69372) among hospitals with the same HPSA score ( 86 FR 73439 ). We included this provision in response to a commenter's recommendation that we prioritize the distribution of slots among hospitals with less than 250 beds and hospitals with a single residency training program. Under this policy, we first distribute FTE slots to applications from hospitals with less than 250 beds. If there are insufficient FTE slots to distribute to applications from hospitals with less than 250 beds, we prorate among those applications. If there are sufficient slots to distribute to applications from hospitals with less than 250 beds, we prorate the remaining slots among the applications from hospitals with 250 beds or more. We are not considering a change in this methodology at this time because we believe it may provide a benefit to small hospitals in rural and underserved areas that are seeking to expand their residency training.
Similarly, we are not considering updating our definition of “rural” for purposes of distributing slots under future rounds of section 126, as suggested by a commenter. The definition of rural that we use to implement section 126 is consistent with how that term is defined in the context of the Medicare statute, specifically section 1886(h)(9)(B)(ii)(I) of the Act, as added by section 126, which refers to the definition of a rural area at section 1886(d)(2)(D) of the Act. Furthermore, as we stated in the December 27, 2021 Federal Register , this definition of “rural” is consistent with our policy concerning designation of rural areas for other purposes, including the wage index ( 86 FR 73423 ).
In response to the comment recommending that CMS ensure the policy finalized allows all funded slots to be distributed and that CMS perform a meaningful estimate of the future distribution of available slots to help ensure that another change in priorities is not needed, we note that the requirement to distribute at least 10 percent of slots of hospitals in each eligibility category is statutory and we must therefore consider amending our distribution process if we anticipate that this requirement will not be met. However, as stated earlier, the section 4122 distribution methodology as finalized in this rule includes two processes for distributing slots and we do not believe we need to consider any further adjustments to the finalized methodologies at this time.
Comment: Several commenters referenced their comments on the FY 2022 IPPS proposed rule regarding the use of prioritizing by HPSA score for slot distributions under section 126 of the CAA. The commenters noted that they had urged the agency to prioritize slot distribution based solely on the four categories included in the law and give priority to hospitals that qualify in more than one, with the highest priority given to hospitals qualifying in all four categories. The commenters stated that they had warned CMS in prior comments that if the agency prioritized distributions based on HPSA score, it may result in qualifying hospitals not meeting the 10 percent statutory requirement by category. The commenters stated they continue to urge their original approach and believe that it would be less burdensome and offer a much clearer metric for qualifying hospitals. The commenters stated that such a policy is consistent with the statutory criteria, which do not place any additional emphasis on HPSA service or scores, and still supports teaching hospitals serving underrepresented and historically marginalized populations. A commenter urged CMS to examine whether previous awardees fall into more than one category and how many awardees may already fall into Category Four for which the agency has not accounted. Another commenter stated that they understand CMS' proposed change related to prioritizing eligibility Category Four applicants in the context of meeting the requirements of the law and asked CMS to comment in the final rule on how this change might disadvantage hospitals that may qualify under the other three categories.
Response: We thank the commenters for continuing to note their concerns regarding prioritizing the distribution of section 126 awards by HPSA score. As noted previously, in most cases section 126 round 1 and round 2 awardees qualified for more than one eligibility category. We believe we have accounted for the section 126 round 1 and round 2 awardees that met eligibility Category Four as we verified the HPSAs each awardee selected to use for prioritization of their application with the HPSA Public ID and Score Information included on CMS' DGME website and the HPSA Find tool at https://data.hrsa.gov/tools/shortage-area/hpsa-find . We do not believe that prioritizing Category Four applicants regardless of HPSA score in rounds 4 and 5 of section 126 will disadvantage applicants who fall into other categories as it is unlikely that an applicant would only qualify under eligibility Category Four.
Comment: A commenter stated that it recognizes CMS' argument that (a) the statute mandates it shall distribute at least ten percent of new positions to each of the four categories, that (b) prior rounds did not achieve this requirement for Category Four, and therefore (c) the agency may deviate from the statutory criteria which assigns equal ranking to all categories by elevating Category Four for rounds 4 and 5. The commenter stated that, however, they do not believe the conclusion follows from the premises, as Congress (a) did not contemplate this scenario in the statute, and therefore (b) did not permit the agency to deviate from the prescribed methodology of four equal eligibility categories ranked by HPSA score. The commenter stated they would consider deviation from the prescribed methodology if CMS demonstrated that a failure to distribute slots to Category Four applicants was undermining the success of section 126 in achieving Congressional goals, that is, failing to increase GME residencies in underserved areas, but CMS does not make that case in the proposed rule. The commenter stated that absent other compelling arguments justifying the elevation of Category Four applicants above all others, they strongly recommend the agency withdraw the proposal and proceed with rounds 4 and 5 of section 126 following the same protocols deployed in rounds 1 through 3.
Response: We appreciate the commenter's analysis of the statutory requirements relative to section 126. While we do not believe that a failure to distribute slots to Category Four applicants is undermining the success of section 126 in achieving Congressional goals, we must adhere to the statutory requirement to distribute at least 10 percent of the total number of slots, or at least 100 slots, to hospitals qualifying in each eligibility category. While this requirement will most certainly be met with respect to the remaining three eligibility categories, under both rounds 1 and 2 of section 126, only 12.76 DGME slots and 18.06 IME slots have been distributed to hospitals qualifying under Category Four ( 89 FR 36218 ). As a result of the small number of FTEs being distributed to Category Four hospitals to date, we believe it is necessary to take action now to ensure we meet the statutory 10 percent distribution requirement for Category Four upon completion of all rounds of section 126.
Comment: A commenter stated that they are concerned that CMS may have determined the number of slots that have been distributed to hospitals qualifying under Category Four based on incomplete data. The commenter stated that because hospitals are limited ( print page 69373) to selecting only one eligibility category (even if they qualify under multiple) when applying for section 126, CMS may not be considering the full population of hospitals that qualify for Category Four when calculating the number of slots that these hospitals have received. The commenter stated that CMS should provide additional detail regarding how it conducted the analysis to determine how many hospitals received slots under Category Four. The commenter stated that they suspect that some hospitals would have qualified under Category Four but self-identified their qualifying hospital status using other categories. The commenter stated that CMS itself acknowledges that it needs more information to accurately identify how many slots are distributed to each eligibility category. The commenter stated that in the proposed rule, CMS proposes to collect information regarding qualification for all four categories in the distribution of slots under section 4122 of the CAA, 2023, based on its experience with many hospitals qualifying under more than one category for section 126 slots. The commenter encouraged CMS to also collect this information in future rounds of section 126 slot distributions and provide data in the final rules detailing its progress in meeting the 10 percent threshold in each eligibility category. The commenter also encouraged CMS to analyze awardee information from section 126, rounds 1 to 3 to get a more accurate picture of how many hospitals that received slots based on qualifying in other categories also qualified under Category Four.
Response: We believe there may be a misunderstanding related to the section 126 application process. Hospitals are not limited to selecting a single eligibility category. In the MEARIS TM application module, the screen that includes eligibility category selections is titled “[w]hich eligibility category or categories does your hospital meet?” The following instruction is provided on the screen “[s]elect all eligibility categories that apply to your hospital.” We will further refine this instruction for future rounds of section 126 so that applicants understand that they are to select each eligibility category that applies to their application.
As noted above, in order to check Category Four eligibility, we verified the HPSAs each awardee selected to use for prioritization of their application with the HPSA Public ID and Score Information included on CMS' DGME website and the HPSA Find tool at https://data.hrsa.gov/tools/shortage-area/hpsa-find . We are unsure what language the commenter is referring to when they state that CMS itself acknowledges that it needs more information to accurately identify how many slots are distributed to each eligibility category. In addition to verifying Category Four eligibility, we are able to verify that an applicant meets eligibility categories one through three by using Table 2 posted with the most recent IPPS final rule associated with the application period for the specific section 126 round, using the cost report worksheets submitted with the application, and referring to the list of states included in the December 27, 2021, Federal Register (86 73426). For rounds 1 and 2 of section 126, twelve awardee hospitals qualified under eligibility Category Four using the methodology noted above. Each of these hospitals qualified for at least one other eligibility category. For each section 126 awardee hospital we will continue to verify which eligibility categories the applicants qualify for instead of simply deferring to the selection made on the hospital's application. We will also verify this information for section 4122 awardee hospitals.
Information regarding progress towards meeting the 10 percent requirement for each category will be available on the CMS DGME website.
Comment: Several commenters expressed concerns about the section 126 distribution process in general. Commenters stated that CMS created an overall prioritization that has significantly disadvantaged many New Jersey teaching hospitals that were otherwise positioned to receive GME slots based on the statutory eligibility criteria. The commenters urged CMS to prioritize slot distribution solely based on the four categories in the law. The commenters stated that the reliance on HPSAs minimizes Congress' other priorities to expand training slots for hospitals in rural areas, hospitals training above their cap, and states with new medical schools.
The commenters stated that the statute requires that 10 percent of the aggregate number of residency slots must go to each of the four eligible hospital categories, however, CMS' prioritization disproportionately impacts states like New Jersey in which the HPSA designation is not an accurate reflection of patient access to care. The commenters stated that as of March 2023, New Jersey has only one geographic HPSA and no population HPSAs while by comparison, it has 32 medically underserved areas and 5 medically underserved populations. The commenters urged CMS to prioritize slot distribution based solely on the four categories included in the law but if the agency chooses to continue the practice of super-prioritization for round 3, the commenter requested that CMS either: (a) make exceptions for all-urban states for which a HPSA score is not an accurate measure of patient access; or (b) use a substitute measure that considers the unique population characteristics of those states. A commenter stated that they believe CMS' contention that it is satisfying Congressional intent is misplaced and instead CMS achieved a minimum 10 percent in three categories by coincidence, rather than the intent to prioritize hospitals across each of the four enumerated categories. The commenter stated that they urge CMS to reassess its HPSA prioritization and rebalance its methodology for assessing resident cap slot applications prior to the awarding of round 3 slots.
A commenter stated that CMS not meeting the 10 percent statutory requirement for Category Four is likely due to CMS prioritizing applications based on both population and geographic HPSA scores. The commenter stated that in many cases it is easier to achieve a higher population HPSA score compared to a geographic HPSA score, therefore hospitals with a high HPSA score that have received slots are not serving a geographic HPSA because of how CMS is prioritizing applications. A few commenters stated that 7 geographically rural hospitals have received slots in the first two rounds of distribution of section 126 and that in the second round, only 3 programs that received slots trained for more than 50 percent of the time in a CMS or Federal Office of Rural Health Policy designated rural area. The commenters stated that these two programs include 2 geographically rural hospitals and 1 urban hospital serving as an urban partner in a Rural Track Program. The commenters stated that this analysis implies that reclassified hospitals are making up the bulk of those that receive slots set aside for rural hospitals. The commenters stated that limiting the rural set aside to geographically rural hospitals would align with the legislative intent of section 126 and the commenter stated they are committed to working with Congress and CMS on ensuring that rural hospitals receive future slots.
Response: We appreciate the commenters sharing their concerns about the section 126 prioritization process. Although we proposed to prioritize Category Four hospitals regardless of HPSA score when awarding slots under rounds 4 and 5 of section 126, we did not propose any ( print page 69374) additional changes to the section 126 prioritization process and therefore we consider these comments to be out of scope with respect to section 126 and we will consider them for future rulemaking. We note that the definition of Category One hospitals is statutory, and we do not have the authority to remove hospitals that have reclassified as rural from this eligibility category.
After consideration of the comments received, we are finalizing our policy as proposed with respect to prioritizing hospitals that qualify under Category Four regardless of HPSA score for rounds 4 and 5 of section 126, without modification. The remaining slots awarded under rounds 4 and 5 will be distributed using the existing methodology based on HPSA score ( 86 FR 73434 through 73440 ). That is, the remaining slots will be distributed to hospitals qualifying under Category One, Category Two, or Category Three, or hospitals that meet the definitions of more than one of these categories, based on the HPSA score associated with the program for which each hospital is applying.
For section 126 of the CAA, 2021, we previously finalized a policy that all applicant hospitals be required to attest that they meet the National Standards for Culturally and Linguistically Appropriate Services in Health and Health Care (the National CLAS Standards) ( 86 FR 73441 ). This was to ensure that the section 126 distribution broadened the availability of quality care and services to all individuals, regardless of preferred language, cultures, and health beliefs. We stated in the final rule that the National CLAS standards are aligned with the Administration's commitment to addressing healthcare barriers, which include that residents are educated and trained in culturally and linguistically appropriate policies and practices. This continues to be the case today. Therefore, we proposed the same requirement for section 4122 of the CAA, 2023, that we adopted for section 126 of the CAA, 2021, for the same reason. Specifically, we proposed that in order to ensure that residents are educated and trained in culturally and linguistically appropriate policies and practices, all applicant hospitals for slots allocated under section 4122 of the CAA, 2023, would be required to attest that they meet the National CLAS Standards to ensure that the section 4122 distribution broadens the availability of quality care and services to all individuals, regardless of preferred language, cultures, and health beliefs. (For more information on the CLAS standards, please refer to https://thinkculturalhealth.hhs.gov/ )
We did not receive any public comments related to our proposal that all applicant hospitals be required to attest that they meet the National Standards for Culturally and Linguistically Appropriate Services in Health and Health Care (the National CLAS Standards). We are finalizing this policy as proposed.
Section 1886(h)(10)(D) requires that CMS pay a hospital for additional positions awarded under this paragraph using the hospital's existing direct GME nonprimary care PRAs consistent with the regulations at § 413.77. We note that as specified in section 1886(h)(2)(D)(ii) of the Act, for cost reporting periods beginning on or after October 1, 1993, through September 30, 1995, each hospital's PRA for the previous cost reporting period was not updated for inflation for any FTE residents who were not either a primary care or an obstetrics and gynecology resident. As a result, hospitals with both primary care and obstetrics and gynecology residents and nonprimary care residents in FY 1994 or FY 1995 have two separate PRAs: one for primary care and obstetrics and gynecology and one for nonprimary care. Those hospitals that only trained primary care and/or obstetrics and gynecology residents and those that did not become teaching hospitals until after this 2-year period, have a single PRA for direct GME payment purposes. Therefore, we proposed that for purposes of direct GME payments for section 4122 of the CAA, 2023, if a hospital has both a primary care and obstetrics and gynecology PRA and a nonprimary care PRA, the nonprimary care PRA will be used, and if a hospital has a single PRA, that PRA will be used. Furthermore, similar to the policy finalized for purposes of direct GME payments under section 126 of the CAA, 2021 ( 86 FR 73441 ), we proposed that a hospital that receives additional positions under section 4122 of the CAA, 2023, would be paid for the FTE residents counted under those positions using the PRAs for which payment is made for FTE residents subject to the 1996 FTE cap. We expect to revise Worksheet E-4 to add a line on which hospitals will report the number of FTEs by which the hospital's FTE caps were increased for direct GME positions received under section 4122 of the CAA, 2023.
We did not receive any public comments related to our proposal for payment of additional FTE residency positions awarded under section 4122 of the CAA, 2023. We are finalizing this policy as proposed.
Section 1886(h)(10)(E) of the Act states that the Secretary shall permit hospitals receiving additional residency positions attributable to the increase provided under 1886(h)(10) to, beginning in the fifth year after the effective date of such increase, apply such positions to the limitation amount under paragraph (4)(F) that may be aggregated pursuant to paragraph (4)(H) among members of the same affiliated group. Therefore, we proposed that FTE resident cap positions added under section 4122 of the CAA, 2023, may be used in a Medicare GME affiliation agreement beginning in the 5th year after the effective date of the FTE resident cap positions consistent with the regulations at 42 CFR 413.75(b) and 413.79(f) . We proposed to amend paragraph (8) at 42 CFR 413.79(f) to state that FTE resident cap slots added under section 4122 of Public Law 117-328 may be used in a Medicare GME affiliation agreement beginning in the fifth year after the effective date of those FTE resident cap slots.
We did not receive any public comments related to our proposal for the aggregation of additional FTE residency positions awarded under section 4122 of the CAA, 2023. We are finalizing this policy as proposed.
Section 4122 of the CAA, 2023, under subsection (b), amends section 1886(d)(5)(B) of the Act to provide for increases in FTE resident positions for IME payment purposes. Specifically, subsection (b) adds a new section 1886(d)(5)(B)(xiii) of the Act, which states that for discharges occurring on or after July 1, 2026, if additional payment is made for FTE resident positions distributed to a hospital for direct GME purposes under section 1886(h)(10) of the Act, the hospital will receive IME payments based on the additional residency positions awarded using the same IME adjustment factor used for the hospital's other FTE residents. We proposed conforming amendments to the IME regulations at 42 CFR 412.105(f)(1)(iv)(C)(4) to specify that effective for portions of cost reporting periods beginning on or after July 1, 2026, a hospital may qualify to receive ( print page 69375) an increase in its otherwise applicable FTE resident cap if the criteria specified in 42 CFR 413.79(q) are met. We expect to revise Worksheet E Part A to add a line on which hospitals will report the number of FTEs by which the hospital's FTE caps were increased for IME positions received under section 4122 of the CAA, 2023.
We also proposed to amend our regulations at 42 CFR 413.79 by adding a paragraph (q) to specify that for portions of cost reporting periods beginning on or after July 1, 2026, a hospital may receive an increase in its otherwise applicable FTE resident cap (as determined by CMS) if the hospital meets the requirements and qualifying criteria under section 1886(h)(10) of the Act and if the hospital submits an application to CMS within the timeframe specified by CMS.
We did not receive any public comments on our proposal related to the conforming amendments for 42 CFR 412.105 and 42 CFR 413.79 . We are finalizing this policy as proposed.
Section 4122 of the CAA, 2023, under subsection (c), prohibits administrative and judicial review of actions taken under section 1886(h)(10) of the Act. Specifically, subsection (c) amends section 1886(h)(7)(E) of the Act by inserting “paragraph (10),” after “paragraph (8),” adding to the that paragraph to the list of residency distributions not subject to review. Therefore, we proposed that the determinations and distribution of residency positions under sections 1886(d)(5)(B)(xiii) and 1886(h)(10) of the Act would be final and could not be subject to administrative or judicial review.
We did not receive any comments related to our proposal on the prohibition on administrative or judicial review. We are finalizing this policy as proposed.
All qualifying hospitals seeking increases in their FTE resident caps must submit timely applications for this distribution by March 31, 2025. The completed application must be submitted to CMS using an online application system, the Medicare Electronic Application Request Information System TM (MEARIS TM ). The burden associated with this information collection requirement is the time and effort necessary to review instructions and register for MEARIS TM as well as the time and effort to gather, develop and submit various documents associated with a formal request of resident position increases from teaching hospitals to CMS. The aforementioned burden is subject to the Paperwork Reduction Act (PRA); and as discussed in section XII.B. of this final rule, the burden associated with these requests will be captured under OMB control number 0938-1417 (expiration date March 31, 2025). We will submit a revised information collection estimate to OMB for approval under OMB control number 0938-1417 (expiration date March 31, 2025).
We proposed that the following information be submitted as part of an application for the application to be considered complete:
- The name and Medicare provider number (CCN) of the hospital.
- The name of the Medicare Administrative Contractor to which the hospital submits its Medicare cost report.
- The residency program for which the hospital is applying to receive an additional position(s).
- FTE resident counts for direct GME and IME and FTE resident caps for direct GME and IME reported by the hospital in the most recent as-filed cost report. (Including copies of Worksheet E, Part A, and Worksheet E-4).
- If the hospital qualifies under “Demonstrated Likelihood” Criterion 1 (New Residency Program), which of the following applies:
++ Application for accreditation of the new residency program has been submitted to the Accreditation Council for Graduate Medical Education (ACGME) (or application for approval of the new residency program has been submitted to the American Board of Medical Specialties (ABMS)) by March 31, 2025.
++ The hospital has received written correspondence from the ACGME (or ABMS) acknowledging receipt of the application for the new residency program, or other types of communication concerning the new program accreditation or approval process (such as notification of a site visit) by March 31, 2025.
- If the hospital qualifies under “Demonstrated Likelihood” Criterion 2 (Expansion of an Existing Residency Program), which of the following applies:
++ The hospital has received approval by March 31, 2025 from an appropriate accrediting body (the ACGME or ABMS) to expand the number of FTE residents in the program.
++ The hospital has submitted a request by March 31, 2025 for a permanent complement increase of the existing residency training program.
- Indication of the categories under section 1886(h)(10)(F)(iii) of the Act under which the hospital believes itself to qualify:
++ (I) The hospital is located in a rural area (as defined in section 1886(d)(2)(D) of the Act) or is treated as being located in a rural area (pursuant to section 1886(d)(8)(E) of the Act).
++ (II) The reference resident level of the hospital (as specified in section 1886(h)(10)(F)(iv) of the Act) is greater than the otherwise applicable resident limit.
++ (III) The hospital is located in a State with a new medical school (as specified in section 1886(h)(10)(B)(ii)(III)(aa) of the Act), or with additional locations and branch campuses established by medical schools (as specified in section 1886(h)(10)(B)(ii)(III)(bb) of the Act) on or after January 1, 2000.
++ (IV) The hospital serves an area designated as a HPSA under section 332(a)(1)(A) of the Public Health Service Act, as determined by the Secretary.
- The HPSA (if any) served by the residency program for which the hospital is applying and the HPSA ID for that HPSA.
- An attestation, signed and dated by an officer or administrator of the hospital who signs the hospital's Medicare cost report, stating the following:
“I hereby certify that the hospital is a Qualifying Hospital under section 1886(h)(10)(F)(iii) of the Social Security Act, and that there is a “demonstrated likelihood” that the hospital will fill the position(s) made available under section 1886(h)(10) of the Act within the first 5 training years beginning after the date the increase would be effective.”
“I hereby certify that (choose if applicable):
__If my application is for a currently accredited residency program, the number of full-time equivalent (FTE) positions requested by the hospital does not exceed the number of positions for which the program is accredited.
__If my hospital currently has unfilled positions in its residency program that have previously been approved by the ACGME, the number of FTE positions requested by the hospital does not exceed the number of previously approved unfilled residency positions. ( print page 69376)
__If my application is for a residency training program with more than one participating site, I am only requesting the FTE amount that corresponds with the training occurring at my hospital, and any FTE training occurring at nonprovider settings consistent with 42 CFR 412.105(f)(1)(ii)(E) and 413.78(g) .”
“I hereby certify that the hospital agrees to increase the number of its residency positions by the amount the hospital's FTE resident caps are increased under section 4122 of Subtitle C of the Consolidated Appropriations Act, 2023, if awarded positions under section 1886(h)(10)(C)(ii) of the Act.”
“I hereby certify that (choose one):
__In the geographic HPSA the hospital is requesting that CMS use for prioritization of its application, at least 50 percent of the program's training time based on resident rotation schedules (or similar documentation) occurs at training sites that treat the population of the HPSA and are physically located in the HPSA.
__In the population HPSA the hospital is requesting that CMS use for prioritization of its application, at least 50 percent of the program's training time based on resident rotation schedules (or similar documentation) occurs at training sites that treat the designated underserved population of the HPSA and are physically located in the HPSA.
__In the geographic HPSA the hospital is requesting that CMS use for prioritization of its application, at least 5 percent of the program's training time based on resident rotation schedules (or similar documentation) occurs at training sites that treat the population of the HPSA and are physically located in the HPSA, and the program's training time at those sites plus the program's training time at Indian or Tribal facilities located outside of the HPSA is at least 50 percent of the program's training time.
__In the population HPSA the hospital is requesting that CMS use for prioritization of its application, at least 5 percent of the program's training time based on resident rotation schedules (or similar documentation) occurs at training sites that treat the designated underserved population of the HPSA and are physically located in the HPSA, and the program's training time at those sites plus the program's training time at Indian or Tribal facilities located outside of that HPSA is at least 50 percent of the program's training time.
__None of the above apply.”
“I hereby certify that the hospital meets the National Standards for Culturally and Linguistically Appropriate Services in Health and Health Care (the National CLAS Standards).”
“I hereby certify that I understand that misrepresentation or falsification of any information contained in this application may be punishable by criminal, civil, and administrative action, fine and/or imprisonment under Federal law. Furthermore, I understand that if services identified in this application were provided or procured through payment directly or indirectly of a kickback or where otherwise illegal, criminal, civil, and administrative action, fines and/or imprisonment may result. I also certify that, to the best of my knowledge and belief, it is a true, correct, and complete application prepared from the books and records of the hospital in accordance with applicable instructions, except as noted. I further certify that I am familiar with the laws and regulations regarding Medicare payment to hospitals for the training of interns and residents.”
The completed application must be submitted to CMS using the online application system MEARIS TM . A link to the online application system as well as instructions for accessing the system and completing the online application process will be made available on the CMS Direct GME website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/DGME .
We note that if the hospital is applying using a HPSA ID, the HPSA score associated with that ID will automatically populate in the application module. In preparing their applications for additional residency positions, hospitals should refer to HRSA's Find Shortage Areas by Address ( https://data.hrsa.gov/tools/shortage-area/by-address ) to obtain the HPSA ID of the HPSA served by the program and include this ID in its application. Using this HPSA Find Shortage Areas by Address, applicants may enter the address of a training location (included on the hospital's rotation schedule or similar documentation), provided the location chosen participates in training residents in a program where at least 50 percent (5 percent if an Indian and Tribal facility is included) of the training time occurs in the HPSA. In November 2024, prior to the beginning of the application period, CMS will request HPSA ID and score information from HRSA so that recent HPSA information is available for use for the application period. CMS will only use this HPSA information, HPSA IDs and their corresponding HPSA scores, in order to review and prioritize applications. To assist hospitals in preparing for their applications, the HPSA information received from HRSA will also be posted when the MEARIS TM application module becomes available on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/DGME .
The information will also be posted on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/IPPS-Regulations-and-Notices . Click on the link on the left side of the screen associated with the appropriate final rule home page or “Acute Inpatient—Files for Download.”
Comment: We did not receive any public comments with respect to the proposed application process for section 4122 of the CAA, 2023, and therefore we are finalizing the application process as proposed.
However, we did receive comments asking CMS to provide guidance regarding the interaction between round 4 of section 126 of the CAA, 2021 and section 4122 of the CAA, 2023, since slots for both of these provisions will be effective July 1, 2026. Specifically, commenters asked whether a hospital may: (1) apply for an increase through section 126 round 4 and section 4122; and (2) apply for increases in the same residency program for both section 126 round 4 and section 4122. Another commenter asked whether the same provider site could apply for pediatrics FTE(s) under section 4122 and internal medicine FTE(s) under round 4 of section 126. The commenter asked, if such an application is allowed, whether there would be any impact in prioritization in dual applications.
Response: We do not believe there is any language in the statute that precludes a hospital from applying for slots under both round 4 of section 126 and section 4122. However, the statute doesn't require us to, and we will not, award duplicative FTE cap slots for the same program under these provisions. We strongly recommend that if an applicant is applying for the same program (same ACGME accreditation number) under both round 4 of section 126 and section 4122, it submit with its application a note indicating as such. Lastly, if an applicant submits an application under both round 4 of section 126 and section 4122, there is no impact on prioritization as the prioritization for awards is performed separately for these two provisions. ( print page 69377)
Section 1886(h)(4)(H)(i) of the Act requires CMS to establish rules for applying the direct GME cap in the case of medical residency training programs established on or after January 1, 1995. Under section 1886(d)(5)(B)(viii) of the Act, this provision also applies for purposes of the IME adjustment. Accordingly, we issued regulations at §§ 413.79(e)(1) through (3) discussing the direct GME cap calculation for a hospital that begins training residents in a new medical residency training program(s) on or after January 1, 1995. The same regulations apply for purposes of the IME cap calculation at § 412.105(f)(1)(vii). CMS implemented these statutory requirements in the August 29, 1997 Federal Register ( 62 FR 46005 ) and in the May 12, 1998 Federal Register ( 63 FR 26333 ). The calculation of both the DGME cap and IME cap for new programs is discussed in the August 31, 2012 Federal Register ( 77 FR 53416 ).
Section 413.79(l) defines a new medical residency training program as “a medical residency that receives initial accreditation by the appropriate accrediting body or begins training residents on or after January 1, 1995.” In the August 27, 2009 Federal Register ( 74 FR 43908 through 43917 ), CMS clarified the definition of a “new” residency program and adopted supporting criteria regarding whether or not a residency program can be considered “new” for the purpose of determining if a hospital can receive additional direct GME and/or IME cap slots for that program. CMS adopted these criteria in part to prevent situations where a program at an existing teaching hospital would be transferred to a new teaching hospital, resulting in cap slots created for the same program at two different hospitals. To be considered a “new” program for which new cap slots would be created, a previously non-teaching hospital would have to ensure that the program meets three primary criteria ( 74 FR 43912 ):
- The residents are new, and
- The program director is new, and
- The teaching staff are new.
Over the years, we have received questions regarding the application of these criteria, such as whether CMS would still consider a program to be new for cap adjustment purposes if the three criteria were partially, but not fully, met. We have answered such questions by stating that, generally, a residency program's newness would not be compromised as long as the “overwhelming majority” of the residents or staff are not coming from previously existing programs in that same specialty.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36221 ), the question of what constitutes a “new” program for purposes of receiving additional Medicare-funded GME slots has taken on increasing significance in light of the ability of urban hospitals to reclassify as rural under 42 CFR 412.103 for IME purposes, and thus receive additional IME cap slots for any new program started. In the proposed rule, we stated that, to continue to ensure that newly funded cap slots are created appropriately, we ultimately would like to establish in rulemaking additional criteria for determining program newness. However, we also indicated that we were not yet certain about some of the criteria that should be proposed. Therefore, we proposed a policy for determining whether the residents in a program are genuinely new, while we solicited comments through a Request for Information (RFI) to gain additional clarity on best practices in other areas.
We received many comments in response to our proposed criterion for ensuring newness of residents, and to our RFIs regarding criteria for program directors, teaching staff, and other issues such as commingling of residents. With regard to the RFIs, we will carefully consider comments received and will take them into account for future rulemaking. We acknowledge that the vast majority of commenters opposed any restrictions on the program director, faculty, comingling of residents, and one hospital sponsoring two programs in the same specialty.
Regarding our proposed criterion for ensuring newness of residents, we received many wide-ranging comments and commenters did not arrive at a consensus on the best approach to this issue. Accordingly, at this time, we are not finalizing our proposal that at least 90 percent of the individual resident trainees (not FTEs) must not have previous training in the same specialty as the new program. Instead, in an effort to achieve greater consensus on this issue, we are initiating another RFI particularly focused on the criterion regarding newness of residents. Commenters should review and consider all of the comments summarized below when formulating responses to this RFI. We look forward to receiving additional feedback from commenters after they have had the opportunity to review the comments and suggestions of others.
Generally, when a hospital is creating a new residency program, it recruits individuals that have recently graduated from medical school, have no previous residency training experience, and would be entering the program as first year (PGY1) residents. However, new programs sometimes receive inquiries from applicants that have training experience already, but for a variety of reasons need to transfer to another program. If the program that such a resident wishes to join is still within the 5-year cap building period, then, consistent with the criteria adopted in the August 27, 2009 final rule, the program director of this “new” program should be judicious with regard to accepting residents who have received previous training in the same specialty. In order to maintain the classification as a “new” residency program, the “overwhelming majority” of residents in the program must be new. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36222 ), we stated that we believe it would be useful for the provider community to have a concrete standard to refer to in determining whether the “overwhelming majority” of residents in a program are in fact new. Therefore, we proposed that, in order for a residency program to be considered new, at least 90 percent of the individual resident trainees (not FTEs) must not have previous training in the same specialty as the new program. For example, if there were 50 trainees (not FTEs) entering the program over the course of the 5-year cap building period, then at least 45 of the trainees (90 percent of 50) must enter the program as brand new first year residents in that particular specialty. If more than 10 percent of the trainees (not FTEs) transferred from another program at a different hospital/sponsor in the same specialty, even during their first year of training, we proposed that this would render the program ineligible for new cap slots. (We noted that we would apply standard rounding when 90 percent of a number does not equal a whole number, rounding down to the nearest whole number when the remainder is less than 0.5, and rounding up to the nearest whole number when the remainder is 0.5 or above. For example, if there were 48 trainees (not FTEs) entering the program over the course of the 5-year cap building period, then at least 43 of the trainees (90 percent of 48 = 43.2, which rounds down to 43) must enter the program as brand new first year residents in that particular ( print page 69378) specialty. If there were 45 trainees (not FTEs) entering the program, then at least 41 of the trainees (90 percent of 45 = 40.5, which rounds up to 41) must enter the program as brand new first year residents in that particular specialty.)
For example, if a new program is in internal medicine, then, under our proposal, at least 90 percent of the entering residents must not have previously enrolled and trained in an internal medicine program. If a resident was formally enrolled in an internal medicine program (either preliminary or categorical), even if that resident switched programs during their first year of training, then we would consider that resident to have had previous training in that same specialty. Conversely, if an individual was a resident in a specialty other than internal medicine, and that resident switched into the new internal medicine program and began training in the new internal medicine program as a PGY1, then that resident would not be considered to have had previous training in the same specialty, and would be counted as a brand new resident. (Note, we are distinguishing between a resident that is not enrolled in an internal medicine program but may have done a rotation in internal medicine as part of the requirements for a different specialty, from a resident that actually was enrolled and participated in an internal medicine program, consistent with the definition of “resident” at 42 CFR 413.75(b) . In this example, we are generally focusing on individuals who were accepted, enrolled, and participated in internal medicine; we are generally not concerned with an individual that was enrolled, accepted, and participated in a program other than internal medicine but did a rotation in internal medicine.) We proposed that the proportion of brand new residents in a residency program would be determined by the MAC based on all the individuals (not FTEs) that enter the program as a whole at any point during the 5-year cap building period, after the end of the 5 years.
We proposed a threshold of 90 percent for new residents as that is generally consistent with the concept of an “overwhelming majority,” and because we have precedent for such a threshold in the regulations for section 5506 of the Affordable Care Act, which state that a hospital is considered to have taken over an “entire” program from a closed hospital if it can demonstrate that it took in 90 percent or more of the FTE residents in that program. Accordingly, for a program to be considered “new” for the purpose of determining if a hospital can receive additional direct GME and/or IME cap slots for that program, we proposed that at least 90 percent of the individual resident trainees (not FTEs) in the program as a whole must not have had previous training in the same specialty as the new program. If more than 10 percent of the trainees (not FTEs) transferred from another program at a different hospital/sponsor in the same specialty, even during their first year of training, we proposed that this would render the program as a whole (but not the entire hospital or its other new programs, if applicable) ineligible for new cap slots.
In addition, we stated in the proposed rule that we understand that there may be certain challenges that are unique to small or rural-based programs in developing new residencies, and that meeting a proposed threshold of 90 percent of resident trainees with no previous training experience in the specialty may be more difficult for those programs. Accordingly, we solicited comments on what should be considered a “small” program and what percentage threshold or other approach regarding new resident trainees should be applied to these programs. We also solicited comment on defining a small residency program as a program accredited for 16 or fewer resident positions, because 16 positions would encompass the minimum number of resident positions required for accredited programs in certain specialties, such as primary care and general surgery, that have historically experienced physician shortages, and therefore have been prioritized by Congress and CMS for receipt of slots under sections 5503 and 5506 of the Affordable Care Act.
Several commenters expressed general opposition to our proposal, and to the suggestions presented in our Requests for Information, stating that these policies would be administratively burdensome, ineffective at preventing the transfer of existing programs or the duplication of FTE cap slots, and detrimental to graduate medical education in general and in particular to small and rural residency programs. Other commenters, while supportive in principle of the need for implementing more concrete standards, nevertheless expressed concern that any new guidelines should not adversely affect the educational quality of new programs or impede the establishment of new programs, which are critical to addressing workforce shortages. Furthermore, a number of commenters generally opposed the consideration of individuals' prior training or employment history in the determination of program newness, stating that these factors are extraneous to CMS's central concerns about whether a program has been transferred and whether FTE cap slots may have been duplicated. Commenters also argued that many of the issues addressed in the proposed rule and suggested policies, including the transfer of residents and recruitment of faculty and program directors, are already regulated by entities such as the ACGME, the ABMS, and the National Resident Matching Program (NRMP), and urged CMS to defer to the judgment and expertise of those organizations. For example, several commenters recommended that CMS generally defer to the assessment of the accrediting body and that the determining question in establishing newness should be whether the program has received initial accreditation for the first time from the ACGME. In addition to receiving initial accreditation for the first time from the ACGME, some commenters suggested that CMS could address its concerns if it undertook a “cursory overview” of the program and/or required an attestation from the hospital that the program has not been transferred and that it does not duplicate FTE cap slots associated with an existing program.
A few commenters supported our proposal that at least 90 percent of FTE residents must not have previous training in the same specialty as the new program, stating that this policy would ensure that cap adjustments are granted to genuinely new programs while still providing for the limited inclusion of experienced residents. In addition, several commenters expressed support for our proposed 90 percent threshold, but recommended that we make exceptions for small or mid-sized programs and for circumstances outside of a hospital's control (for example, situations in which departing residents are replaced by transfers from an existing program).
A few commenters recommended that newness of residents should be established if the program fills most resident positions at the PGY 1 level via a separate and distinct recruiting process (as evidenced, for example, by separate NRMP match numbers, or for fellowships, an applicable distinct process). However, commenters stressed that CMS should not penalize hospitals that attempt to fill PGY 1 positions via the National Resident Matching Program (NRMP), but that may need to fill positions in a different manner due to the results of the Match. Commenters ( print page 69379) recommended that in such cases the hospital should be allowed to submit documentation demonstrating a program's original intent to satisfy the 90 percent criterion.
A few commenters supported the approach of defining a minimum threshold for new residents, but recommended adopting a more lenient standard, such as 75 percent or 70 percent, whereas other commenters recommended that our proposed 90 percent threshold should apply only to residents in their first year of training (that is, PGY 1). Alternatively, some commenters suggested that, in order to address the concern about the transfer of existing programs (or “cannibalism”), CMS should focus on limiting the number of residents who all come from the same existing residency program. In addition, some commenters argued that the presence of experienced residents should not disqualify a program from being deemed new, but suggested that those residents could be excluded from the program's FTE cap calculation.
Several commenters also requested that CMS clarify the methodology for determining the proportions of new and experienced residents, with some commenters specifically recommending that CMS make this calculation based on a count of all residents training over the course of the entire five-year cap-building period. Another commenter recommended that if CMS adopts a new resident threshold, it should count residents on the basis of FTEs rather than individual trainees as proposed, since a program's count could be skewed by enrolling a high proportion of partial FTEs. A few commenters also requested confirmation that fellows in subspecialty programs, residents who have completed transitional or preliminary year programs, and residents from closed programs would not be considered to have prior experience training in the same specialty.
A number of commenters suggested alternative methods for assessing program newness that do not depend on the proportion of residents without previous experience training in the same specialty. Some commenters suggested that CMS consider the relationship between the “new” program and the program that appears to have been transferred or duplicated. For example, if the original program remains open for a minimum of one full academic year, then the second program should be considered genuinely “new.”
A commenter recommended that CMS adopt a “totality of circumstances” approach in which we would assess various aspects of the program, such as its age and whether it has existed previously at another site, rather than focusing on rigid individual metrics. Another commenter stated that CMS should apply a “reasonable person” standard in determining whether a program is genuinely new. In addition, a few commenters stated that if CMS were to implement the proposed 90 percent threshold, then a program that fails to meet the threshold should be given the opportunity to demonstrate newness by other means, and that CMS should also consider mitigating factors such the size of the program or matched residents who did not disclose prior training experiences.
In addition to the specific recommendations discussed above, commenters generally expressed concern that any criteria adopted should be objective, transparent and administratively feasible, especially given the significant costs and high financial stakes associated with developing a new residency program. A commenter also recommended that CMS should carry out periodic evaluations of newness during a program's five-year cap-building period to ensure that a hospital has time to make any changes necessary to bring the program into compliance.
Commenters generally agreed that CMS should create exceptions to the newness criteria for small and rural programs; several commenters also argued that we should give similar consideration to programs in urban underserved areas. In particular, commenters noted that many small programs would fail to meet our proposed 90 percent threshold if they admitted even one experienced resident. Several commenters also reported that it is common for new rural programs, including Rural Track Programs, to accept a higher proportion of non-PGY 1 residents as a means of “jump starting” the program and promoting stability. A few commenters indicated that small and rural or urban underserved programs should be exempted from any restrictions on the recruitment of experienced residents (as well as on the recruitment of faculty and program directors). Although commenters agreed that our proposed 90 percent new resident threshold would be too strict for such programs, there was no consensus as to a specific standard that would be appropriate: a few commenters recommended a much lower threshold of 25 percent, while others suggested that 50 percent of PGY 1 residents should be new, with no restrictions on non-PGY 1 residents. Several commenters agreed with our suggestion that a small program should be defined as one accredited for 16 or fewer resident positions; however, a few commenters stated that for purposes of determining exceptions to the newness criteria a small program should also be required to train residents for greater than half the time in a rural area or an urban underserved area. Other commenters recommended lower thresholds for defining small programs, with specific suggestions including 12, 10, 6 or 4 positions. A few commenters recommended that the newness of small programs be evaluated on a case-by-case basis, taking into account the totality of a hospital's financial, geographic and other circumstances.
Several commenters stressed that any new policies should only apply to programs that begin training residents after the effective date of the final rule (that is, on or after October 1, 2024), so as not to adversely impact programs currently in their cap-building period.
Some commenters also objected to CMS's administration and interpretation of the newness criteria promulgated in the August 27, 2009 Federal Register ( 74 FR 43908 through 43917 ), describing CMS's approach as “unnecessarily cynical” and stating that the criteria for assessing program newness are not reflected in the statutes or regulations. Commenters also alleged that CMS has been interpreting the existing newness criteria in ways that differ substantially from how members of the provider community have been interpreting these policies. For example, a few commenters stated that it was not clear from August 27, 2009 Federal Register that teaching staff and program directors must have no prior experience in these roles for a program to be considered genuinely new.
Section 1886(h)(4)(H)(i) of the Act states that the Secretary shall, consistent with the principles of subparagraphs (F) {Initial Residency Period} and (G) {Period of Board Eligibility} and subject to paragraphs (7) {Redistribution of Unused Residency Positions} and (8) {Distribution of Additional Residency Positions}, prescribe rules for the application of such subparagraphs in the case of medical residency training programs established on or after January 1, 1995 (that is, new programs). In promulgating such rules for purposes of subparagraph (F), the Secretary shall give special consideration to facilities that meet the needs of underserved rural areas.
Thus, the Secretary has broad statutory authority to prescribe rules for counting residents in new programs. ( print page 69380)
As we stated at the beginning of this section, we received many wide-ranging comments and commenters did not arrive at a consensus on the best approach to the issue of the newness of residents. Accordingly, at this time, we are not finalizing our proposal that at least 90 percent of the individual resident trainees (not FTEs) must not have previous training in the same specialty as the new program. Instead, in an effort to achieve greater consensus on this issue, we are initiating another RFI seeking comment on the appropriate criterion regarding newness of residents. Commenters should review and consider the broad statutory authority provided to the Secretary in this area, our prior rulemaking on this issue, and all of the public comments on our proposal as summarized in Section F.3.b of this final rule when formulating responses to this RFI. We look forward to receiving additional feedback from commenters after they have had the opportunity to review the comments and suggestions of others. We also, in the interest of facilitating consensus, encourage commenters to provide feedback on what alternative to their primary recommended approach they would consider to be most acceptable among the different approaches suggested by commenters.
In the course of our ongoing implementation of policies concerning payment for graduate medical education, we have become aware of the existence of several technical errors in the direct GME regulations at 42 CFR 413.75 through 413.83 . In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36224 through 36225 ), we proposed to correct the following technical errors:
In the FY 2010 IPPS final rule ( 74 FR 43918 and 44001 , August 27, 2009), we amended 42 CFR 413.79(f) by adding a new paragraph (f)(6) and redesignating existing paragraph (f)(6) as paragraph (f)(7). The new § 413.79(f)(6) sets forth requirements for participation in a Medicare GME affiliated group by a hospital that is new after July 1 and begins training residents for the first time after the July 1 start date of an academic year, while the redesignated § 413.79(f)(7) contains the regulations pertaining to emergency Medicare GME affiliated groups.
We have discovered that, after redesignating the former § 413.79(f)(6) as § 413.79(f)(7), we inadvertently did not update the cross-references to this paragraph at §§ 413.75(b) and 413.78. Accordingly, in the proposed rule, we proposed to revise the language of the definition of “Emergency Medicare GME affiliated group” under § 413.75(b), as well as the language at §§ 413.78(e)(3)(iii) and (f)(3)(iii), by correcting the cross-references to read “§ 413.79(f)(7).”
Under 42 CFR 413.79(h) , a hospital may receive a temporary adjustment to its FTE cap to reflect displaced residents added as a result of the closure of another hospital or residency training program. Furthermore, under § 413.79(d)(6)(i) (previously § 413.79(d)(6)), displaced residents counted under a temporary cap adjustment are added to the receiving hospital's FTE count after application of the three-year rolling average for the duration of the time that the displaced residents are training at the receiving hospital.
In the November 24, 2010 final rule ( 75 FR 72212 through 72238 ), we implemented the provisions of section 5506 of the Affordable Care Act, which directs the Secretary to redistribute Medicare GME residency slots from teaching hospitals that close after March 23, 2008. A hospital that had previously accepted residents displaced by a teaching hospital closure and received a temporary cap adjustment for training those residents under § 413.79(h) may subsequently apply for a permanent cap increase under section 5506.
As part of the implementation of section 5506, we finalized several ranking criteria to prioritize applications, and specified the dates on which awards would become effective for hospitals that apply under each of those criteria. In particular, we finalized Ranking Criteria One and Three, which describe applicant hospitals that take over, respectively, an entire residency program(s) or part of a residency program(s) from the closed hospital. Consistent with the policy finalized in the November 24, 2010 final rule, a permanent cap increase awarded under Ranking Criterion One or Three would generally override any temporary cap adjustment that the applying hospital may have received under § 413.79(h), with the result that those resident slots would immediately become subject to the three-year rolling average calculation ( 75 FR 72224 ).
We also stated, however, that we believed it would still be appropriate to allow a hospital that ultimately would qualify to receive slots permanently under any of the ranking criteria and that took in displaced residents to receive temporary cap adjustments and, in a limited manner, an exemption from the three-year rolling average. Therefore, we finalized a policy that, in the first cost reporting period in which the applying hospital takes in displaced residents and the hospital closure occurs, the applying hospital could receive a temporary cap adjustment and an exemption from the rolling average for the displaced residents. Then, effective beginning with the cost reporting period following the one in which the hospital closure occurred, the applying hospital's permanent cap increase would take effect, and there would be no exemption from the rolling average ( 75 FR 72225 and 72263 ).
Therefore, we amended § 413.79(d) by redesignating the existing paragraph (d)(6) as (d)(6)(i) and by adding new (d)(6)(ii), which states stated that if a hospital received a permanent increase in its FTE resident cap under § 413.79(o)(1) due to redistribution of slots from a closed hospital, the displaced FTE residents that the hospital received would be added to the FTE count after applying the averaging rules only in the first cost reporting period in which the receiving hospital trained the displaced FTE residents. In subsequent cost reporting periods, the displaced FTE residents would be included in the receiving hospital's rolling average calculation.
Subsequently, in the FY 2013 IPPS final rule ( 77 FR 53437 through 53443 , August 31, 2012), we finalized revisions to our policy concerning the effective dates of section 5506 cap increases awarded under the various ranking criteria. In particular, we finalized a policy that slots awarded under Ranking Criteria One and Three become effective seamlessly with the expiration of temporary cap adjustments under § 413.79(h) (that is, on the day after the graduation date(s) of the displaced residents). As stated in that final rule, under this revised policy, permanent cap increases under section 5506 would no longer “replace” temporary cap adjustments under § 413.79(h), and exemptions from the three-year rolling average would no longer be suspended as a consequence of the receipt of permanent slots ( 77 FR 53441 ).
Under the policy finalized in the FY 2013 IPPS final rule, there is no longer any need for the regulation at § 413.79(d)(6)(ii), which would apply in the situation where a permanent cap increase under section 5506 would otherwise have overridden a temporary cap adjustment for displaced residents under § 413.79(h). Instead, our policy is that displaced residents are excluded ( print page 69381) from the receiving hospital's rolling average calculation for the duration of the time that they are training at the receiving hospital, as specified at § 413.79(d)(6)(i). However, we have discovered that we neglected to make the appropriate revisions to the regulations text to reflect our current policy.
Accordingly, we proposed to amend § 413.79(d)(6) by removing the no longer applicable paragraph (d)(6)(ii), and by redesignating existing (d)(6)(i) as (d)(6).
In the final rule published on December 27, 2021, as part of the implementation of section 127 of the CAA, 2021 ( Pub. L. 116-260 ), we finalized various changes throughout the regulations text at 42 CFR 413.79(k) , “Residents training in rural track programs” ( 86 FR 73445 through 73457 and 73514 through 73515 ). We have discovered that the final sentence of § 413.79(k)(2)(i), as amended in that rule, incorrectly states, “For Rural Track Programs prior to the start of the urban or rural hospital's cost reporting period that coincides with or follows the start of the sixth program year of the rural track's existence . . .”
The beginning of the quoted sentence should instead refer to “cost reporting periods beginning on or after October 1, 2022,” and should otherwise be analogous to the similar text that appears at § 413.79(k)(1)(i). Accordingly, we proposed to revise § 413.79(k)(2)(i) to read as follows: “For cost reporting periods beginning on or after October 1, 2022, before the start of the urban or rural hospital's cost reporting period that coincides with or follows the start of the sixth program year of the Rural Track Program's existence, the rural track FTE limitation for each hospital will be the actual number of FTE residents training in the Rural Track Program at the urban or rural hospital and, subject to the requirements under § 413.78(g), at the rural nonprovider site(s).”
We did not receive any comments on our proposed technical revisions to the direct GME regulations. Therefore, we are finalizing the changes as proposed without modification.
Section 5506 of the Patient Protection and Affordable Care Act ( Pub. L. 111-148 ), as amended by the Health Care and Education Reconciliation Act of 2010 ( Pub. L. 111-152 ) (collectively, “Affordable Care Act”), authorizes the Secretary to redistribute residency slots after a hospital that trained residents in an approved medical residency program closes. Specifically, section 5506 of the Affordable Care Act amended the Act by adding subsection (vi) to section 1886(h)(4)(H) of the Act and modifying language at section 1886(d)(5)(B)(v) of the Act, to instruct the Secretary to establish a process to increase the FTE resident caps for other hospitals based upon the full-time equivalent (FTE) resident caps in teaching hospitals that closed on or after a date that is 2 years before the date of enactment (that is, March 23, 2008). In the CY 2011 Outpatient Prospective Payment System (OPPS) final rule with comment period ( 75 FR 72264 ), we established regulations at 42 CFR 413.79(o) and an application process for qualifying hospitals to apply to CMS to receive direct GME and IME FTE resident cap slots from the hospital that closed. We made certain additional modifications to § 413.79 in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53434 ), and we made changes to the section 5506 application process in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50122 through 50134 ). The procedures we established apply both to teaching hospitals that closed on or after March 23, 2008, and on or before August 3, 2010, and to teaching hospitals that close after August 3, 2010 ( 75 FR 72215 ).
CMS has learned of the closure of Sacred Heart Hospital, located in Eau Claire, WI (CCN 520013). Accordingly, this notice serves to notify the public of the closure of this teaching hospital and initiate another round (“Round 23”) of the application and selection process. This round will be the 23rd round (“Round 23”) of the application and selection process. The table in this section of this rule contains the identifying information and IME and direct GME FTE resident caps for the closed teaching hospital, which are part of the Round 23 application process under section 5506 of the Affordable Care Act.

The application period for hospitals to apply for slots under section 5506 of the Affordable Care Act is 90 days following notice to the public of a hospital closure ( 77 FR 53436 ). Therefore, hospitals that wish to apply for and receive slots from the previously noted hospitals' FTE resident caps must submit applications using the electronic application intake system, Medicare Electronic Application Request Information System TM (MEARIS TM ), with application submissions for Round 23 due no later than October 30, 2024. The Section 5506 application can be accessed at: https://mearis.cms.gov/public/home .
CMS will only accept Round 23 applications submitted via MEARIS TM . Applications submitted through any other method will not be considered. Within MEARIS TM , we have built in several resources to support applicants:
- Please refer to the “Resources” section for guidance regarding the application submission process at: https://mearis.cms.gov/public/resources .
- Technical support is available under “Useful Links” at the bottom of the MEARIS TM web page.
- Application related questions can be submitted to CMS using the form available under “Contact” at: https://mearis.cms.gov/public/resources .
Application submission through MEARIS TM will not only help CMS track applications and streamline the review process, but it will also create efficiencies for applicants when compared to a paper submission process.
We have not established a deadline by when CMS will issue the final determinations to hospitals that receive slots under section 5506 of the Affordable Care Act. However, we review all applications received by the application deadline and notify applicants of our determinations as soon as possible.
We refer readers to the CMS Direct Graduate Medical Education (DGME) website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/direct-graduate-medical-education-dgme . Hospitals should access this website for a list of additional section 5506 guidelines for the policy and procedures for applying for slots, and the redistribution of the slots under sections 1886(h)(4)(H)(vi) and 1886(d)(5)(B)(v) of the Act.
In section III.B. of the preamble of this final rule, we discuss the proposed changes to the most recent OMB standards for delineating statistical areas announced in the July 21, 2023 OMB Bulletin No. 23-01. We refer to these statistical areas as Core-Based Statistical Areas (CBSAs). As a result of the new OMB delineations, some teaching hospitals may be redesignated from location in a rural CBSA to an urban CBSA, or from location in an urban CBSA to a rural CBSA. In the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50111 , August 22, 2014), we last discussed the effects of the CBSA changes on IME and DGME payment policy, as at that time, we implemented the changes to the statistical areas resulting from the February 28, 2013, OMB Bulletin No. 13-01. We refer readers to the FY 2015 IPPS/LTCH PPS final rule to learn more about CMS' policies regarding changes to the CBSAs and how IME and DGME payments are impacted. We emphasize that we did not propose any additional policies as a result of the latest CBSA changes; we are merely providing a reference for readers that may have questions about our existing policies. As a general overview, the FY 2015 IPPS/LTCH PPS final rule discusses the effect on the FTE caps of a hospital that was located in a rural CBSA, either at the time that it started training residents in a new residency program, or was located in a rural area when it received accreditation for a new program, but either prior to actually starting the program or during the 5-year cap building period, the CBSA in which the hospital was located became an urban CBSA ( 79 FR 50111 through 50113 ). We also discussed what happens to a rural training track when a rural hospital that is participating as the rural site is redesignated as urban, either during the period when the rural track is being established, or after it has been established ( 79 FR 50113 ). (Note that under 42 CFR 413.75(b) and 413.79(k) , we now refer to rural training tracks as Rural Training Programs (RTPs)). We provided for a transition period, wherein either the redesignated urban hospital must reclassify as rural under § 412.103 for purposes of IME payment only (in addition, this reclassification option only applies to IPPS hospitals (or CAHs under 42 CFR 412.103(a)(6) ), not other nonprovider sites), or the “original” urban hospital must have found a new site in a geographically rural area that will serve as the rural site for purposes of the rural track in order for the “original” urban hospital to receive payment under § 413.79(k)(1) or (k)(2). Also see DGME regulations at 42 CFR 413.79(c)(6) , 42 CFR 413.79(k)(7) , and for IME, at 42 CFR 412.105(f)(1)(iv)(D) .
Comment: We received one question related to DGME PRA determination of a hospital whose CBSA designation changes as a result of CBSA redesignations. The commenter noted that under 42 CFR 413.77 , a new teaching hospital's PRA is determined based on the lower of the hospital's cost per FTE, or the weighted average PRA of other existing teaching hospitals in the same CBSA as the new teaching hospital. For a new teaching hospital whose CBSA changed status as a result of OMB changes, is the weighted average PRA based on the hospitals in the CBSA at the time the new teaching hospital first began training residents, or the CBSA in effect at the time the MAC audits and calculates the PRA?
Response: The relevant CBSA for the purpose of the weighted average PRA calculation is the CBSA in which the new teaching hospital was located during its PRA base period, under 42 CFR 413.77(e)(1)(i) .
Under section 1861(v) of the Act, Medicare has historically paid providers for Medicare's share of the costs that providers incur in connection with approved educational activities. Approved nursing and allied health (NAH) education programs are those that are, in part, operated by a provider, and meet State licensure requirements, or are recognized by a national accrediting body. The costs of these programs are excluded from the definition of “inpatient hospital operating costs” and are not included in the calculation of payment rates for hospitals or hospital units paid under the IPPS, IRF PPS, or IPF PPS, and are excluded from the rate-of-increase ceiling for certain facilities not paid on a PPS. These costs are separately identified and “passed through” (that is, paid separately on a reasonable cost basis). Existing regulations on NAH education program costs are located at 42 CFR 413.85 . The most recent substantive rulemakings on these regulations were in the January 12, 2001 final rule ( 66 FR 3358 through 3374 ), and in the August 1, 2003, final rule ( 68 FR 45423 and 45434 ). ( print page 69383)
Section 541 of the Balanced Budget Refinement Act (BBRA) of 1999 provides for additional payments to hospitals for costs of nursing and allied health education associated with services to Medicare+Choice (now called Medicare Advantage (MA [ 221 ] )) enrollees. Hospitals that operate approved nursing or allied health education programs and receive Medicare reasonable cost reimbursement for these programs may receive additional payments to account for MA enrollees. Section 541 of the BBRA limits total spending under the provision to no more than $60 million in any calendar year (CY). (In this document, we refer to the total amount of $60 million or less as the payment “pool”.) Section 541 of the BBRA also provides that direct graduate medical education (GME) payments for Medicare+Choice utilization are reduced to the extent that these additional payments are made for nursing and allied health education programs. This provision was effective for portions of cost reporting periods occurring in a CY, on or after January 1, 2000.
Section 512 of the Benefits Improvement and Protection Act (BIPA) of 2000 changed the formula for determining the additional amounts to be paid to hospitals for Medicare+Choice nursing and allied health costs. Under section 541 of the BBRA, the additional payment amount was determined based on the proportion of each individual hospital's nursing and allied health education payment to total nursing and allied health education payments made to all hospitals. However, this formula did not account for a hospital's specific Medicare+Choice utilization. Section 512 of the BIPA revised this payment formula to specifically account for each hospital's Medicare+Choice utilization. This provision was effective for portions of cost reporting periods occurring in a calendar year, beginning with CY 2001.
The regulations at 42 CFR 413.87 codified both statutory provisions. We first implemented the BBRA NAH Medicare+Choice provision in the August 1, 2000 IPPS interim final rule with comment period (IFC) ( 65 FR 47036 through 47039 ), and subsequently implemented the BIPA provision in the August 1, 2001 IPPS final rule ( 66 FR 39909 and 39910 ). In those rules, we outlined the qualifying conditions for a hospital to receive the NAH Medicare+Choice payment, how we would calculate the NAH Medicare+Choice payment pool, and how a qualifying hospital would calculate its “share” of payment from that pool. Determining a hospital's NAH Medicare+Choice payment essentially involves applying a ratio of the hospital-specific NAH Part A payments, total inpatient days, and Medicare+Choice inpatient days, to national totals of those same variables, from cost reporting periods ending in the fiscal year that is 2 years prior to the current calendar year. The formula is as follows:
(((Hospital NAH pass-through payment/Hospital Part A Inpatient Days) * Hospital MA [ 222 ] Inpatient Days)/((National NAH pass-through payment/National Part A Inpatient Days) * National MA Inpatient Days)) * Current Year Payment Pool.
With regard to determining the total national amounts for NAH pass-through payment, Part A inpatient days, and Medicare+Choice inpatient days, we note that section 1886(l) of the Act, as added by section 541 of the BBRA, gives the Secretary the discretion to “estimate” the national components of the formula noted previously. For example, section 1886(l)(2)(A) of the Act states that the Secretary would estimate the ratio of payments for all hospitals for portions of cost reporting periods occurring in the year under subsection 1886(h)(3)(D) of the Act to total direct GME payments estimated for the same portions of periods under section 1886(h)(3) of the Act.
Accordingly, we stated in the August 1, 2000 IFC ( 65 FR 47038 ) that each year, we would determine and publish in a final rule the total amount of nursing and allied health education payments made across all hospitals during the fiscal year 2 years prior to the current calendar year. We would use the best available cost reporting data for the applicable hospitals from the Hospital Cost Report Information System (HCRIS) for cost reporting periods in the fiscal year that is 2 years prior to the current calendar year ( 65 FR 47038 ).
To calculate the pool, in accordance with section 1886(l) of the Act, we stated that we would “estimate” a total amount for each calendar year, not to exceed $60 million ( 65 FR 47038 ). To calculate the proportional reduction to Medicare+Choice (now MA) direct GME payments, we stated that the percentage is estimated by calculating the ratio of the Medicare+Choice nursing and allied health payment “pool” for the current calendar year to the projected total Medicare+Choice direct GME payments made across all hospitals for the current calendar year. We stated that the projections of Medicare+Choice direct GME and Part A direct GME payments are based on the best available cost report data from the HCRIS (for example, for calendar year 2000, the projections are based on the best available cost report data from HCRIS 1998), and these payment amounts are increased using the increases allowed by section 1886(h) of the Act for these services (using the percentage applicable for the current calendar year for Medicare+Choice direct GME and the Consumer Price Index (CPI-U) increases for Part A direct GME). We also stated that we would publish the applicable percentage reduction each year in the IPPS proposed and final rules ( 65 FR 47038 ).
Thus, in the August 1, 2000 IFC, we described our policy regarding the timing and source of the national data components for the NAH Medicare+Choice add-on payment and the percent reduction to the direct GME Medicare+Choice payments, and we stated that we would publish the rates for each calendar year in the IPPS proposed and final rules. While the rates for CY 2000 were published in the August 1, 2000 IFC (see 65 FR 47038 and 47039 ), the rates for subsequent CYs were only issued through Change Requests (CRs) (CR 2692, CR 11642, CR 12407). After recent issuance of the CY 2019 rates in CR 12407 on August 19, 2021, we reviewed our update procedures, and were reminded that the August 1, 2000 IFC states that we would publish the NAH Medicare+Choice rates and direct GME percent reduction every year in the IPPS rules. Accordingly, for CY 2020 and CY 2021, we proposed and finalized the NAH MA add-on rates in the FY 2023 IPPS/LTCH PPS proposed and final rules. We stated that for CYs 2022 and after, we would similarly propose and finalize their respective NAH MA rates and direct GME percent reductions in subsequent IPPS/LTCH PPS rulemakings (see 87 FR 49073 , August 10, 2022).
In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed the rates for CY 2023. Consistent with the use of HCRIS data for past calendar years, we proposed to use data from cost reports ending in FY 2021 HCRIS (the fiscal year that is 2 years prior to CY 2023) to compile these national amounts: NAH pass-through payment, Part A Inpatient Days, MA Inpatient Days. ( print page 69384)
For the proposed rule ( 89 FR 36227 through 36228 ), we accessed the FY 2021 HCRIS data from the fourth quarterly HCRIS update of 2023. However, to calculate the “pool” and the direct GME MA percent reduction, we “project” Part A direct GME payments and MA direct GME payments for the current calendar year, which in the proposed rule and in this final rule is CY 2023, based on the “best available cost report data from the HCRIS” ( 65 FR 47038 ). Next, consistent with the method we described previously from the August 1, 2000 IFC, we increased these payment amounts from midpoint to midpoint of the appropriate calendar year using the increases allowed by section 1886(h) of the Act for these services (using the percentage applicable for the current calendar year for MA direct GME, and the Consumer Price Index-Urban (CPI-U) increases for Part A direct GME). For CY 2023, the direct GME projections are based on the fourth quarterly update of CY 2021 HCRIS, adjusted for the CPI-U and for increasing MA enrollment.
For CY 2023, the proposed national rates and percentages, and their data sources, are set forth in this table. We stated in the proposed rule that we intend to update these numbers in the FY 2025 final rule based on the latest available cost report data.

For this final rule, consistent with the use of HCRIS data for past calendar years, for CY 2023, we use data from cost reports ending in FY 2021 HCRIS (the fiscal year that is 2 years prior to CY 2023) to compile these national amounts: NAH pass-through payment, Part A Inpatient Days, MA Inpatient Days. For this final rule, we accessed the HCRIS data from the first quarterly HCRIS update of 2024. However, to calculate the “pool” and the direct GME MA percent reduction, we project Part A direct GME payments and MA direct GME payments for the current calendar year, which in this final rule is CY 2023, based on the best available cost report data. Next, consistent with the method we described previously from the August 1, 2000 IFC, we increased these payment amounts from midpoint to midpoint of the appropriate calendar year using the increases allowed by section 1886(h) of the Act for these services (using the percentage applicable for the current calendar year for MA direct GME, and the Consumer Price Index-Urban (CPI-U) increases for Part A direct GME). For CY 2023, the direct GME projections are based on FY 2021 HCRIS, and the final national rates and percentages, and their data sources, are set forth in this table.

We only received comments on this section that were out of the scope of the proposal. In summary, we are finalizing our proposal to use NAH MA add-on rates as well as the direct GME MA percent reductions for CY 2023, based on sufficient HCRIS data to develop the rates for these years. We expect to propose to issue the rates for CY 2024 in the FY 2026 IPPS/LTCH PPS proposed rule, when sufficient HCRIS data is available to develop the rates for CY 2024.
Effective for FY 2021, we created MS-DRG 018 for cases that include procedures describing CAR T-cell therapies, which were reported using ICD-10-PCS procedure codes XW033C3 or XW043C3 ( 85 FR 58599 through 58600 ). Effective for FY 2022, we revised MS-DRG 018 to include cases that report the procedure codes for CAR T-cell and non-CAR T-cell therapies and other immunotherapies ( 86 FR 44798 through 448106 ).
Effective for FY 2021, we modified our relative weight methodology for MS-DRG 018 in order to develop a relative weight that is reflective of the typical costs of providing CAR T-cell therapies relative to other IPPS services. Specifically, under our finalized policy we do not include claims determined to be clinical trial claims that group to MS-DRG 018 when calculating the average cost for MS-DRG 018 that is used to calculate the relative weight for this MS-DRG, with the additional refinements that: (a) when the CAR T-cell therapy product is purchased in the usual manner, but the case involves a clinical trial of a different product, the claim will be included when calculating the average cost for MS DRG 018 to the extent such claims can be identified in the historical data; and (b) when there is expanded access use of immunotherapy, these cases will not be included when calculating the average cost for MS-DRG 018 to the extent such claims can be identified in the historical data ( 85 FR 58600 ). The term “expanded access” (sometimes called “compassionate use”) is a potential pathway for a patient with a serious or ( print page 69385) immediately life-threatening disease or condition to gain access to an investigational medical product (drug, biologic, or medical device) for treatment outside of clinical trials when, among other criteria, there is no comparable or satisfactory alternative therapy to diagnose, monitor, or treat the disease or condition ( 21 CFR 312.305 ). [ 223 ]
Effective FY 2021, we also finalized an adjustment to the payment amount for applicable clinical trial and expanded access immunotherapy cases that group to MS-DRG 018 using the same methodology that we used to adjust the case count for purposes of the relative weight calculations ( 85 FR 58842 through 58844 ). (As previously noted, effective beginning FY 2022, we revised MS-DRG 018 to include cases that report the procedure codes for CAR T-cell and non-CAR T-cell therapies and other immunotherapies ( 86 FR 44798 through 448106 ).) Specifically, under our finalized policy we apply a payment adjustment to claims that group to MS-DRG 018 and include ICD-10-CM diagnosis code Z00.6, with the modification that when the CAR T-cell, non-CAR T-cell, or other immunotherapy product is purchased in the usual manner, but the case involves a clinical trial of a different product, the payment adjustment will not be applied in calculating the payment for the case. We also finalized that when there is expanded access use of immunotherapy, the payment adjustment will be applied in calculating the payment for the case. This payment adjustment is codified at 42 CFR 412.85 (for operating IPPS payments) and 42 CFR 412.312 (for capital IPPS payments), for claims appropriately containing Z00.6, as described previously, and reflects that the adjustment is also applied for cases involving expanded access use immunotherapy, and that the payment adjustment only applies to applicable clinical trial cases; that is, the adjustment is not applicable to cases where the CAR T-cell, non-CAR T-cell, or other immunotherapy product is purchased in the usual manner, but the case involves a clinical trial of a different product. The regulations at 42 CFR 412.85(c) also specify that the adjustment factor will reflect the average cost for cases to be assigned to MS-DRG 018 that involve expanded access use of immunotherapy or are part of an applicable clinical trial to the average cost for cases to be assigned to MS-DRG 018 that do not involve expanded access use of immunotherapy and are not part of a clinical trial ( 85 FR 58844 ).
For FY 2025, we proposed to continue to apply an adjustment to the payment amount for expanded access use of immunotherapy and applicable clinical trial cases that would group to MS-DRG 018, as calculated using the same methodology, as modified in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59062 ), that we proposed to use to adjust the case count for purposes of the relative weight calculations, as described in section II.D. of the preamble of this final rule.
As discussed in the FY 2024 IPPS/LTCH PPS final rule, the MedPAR claims data now includes a field that identifies whether or not the claim includes expanded access use of immunotherapy. For the FY 2023 MedPAR data and for subsequent years, this field identifies whether or not the claim includes condition code 90. The MedPAR files now also include information for claims with the payer-only condition code “ZC”, which is used by the IPPS Pricer to identify a case where the CAR T-cell, non-CAR T-cell, or other immunotherapy product is purchased in the usual manner, but the case involves a clinical trial of a different product so that the payment adjustment is not applied in calculating the payment for the case (for example, see Change Request 11879, available at https://www.cms.gov/files/document/r10571cp.pdf ). We refer the readers to section II.D. of the preamble of this final rule for further discussion of our methodology for identifying clinical trial claims and expanded access use claims in MS-DRG 018 and our methodology used to adjust the case count for purposes of the relative weight calculations, as modified in the FY 2024 IPPS/LTCH PPS final rule.
Using the same methodology that we proposed to use to adjust the case count for purposes of the relative weight calculations, we proposed to calculate the adjustment to the payment amount for expanded access use of immunotherapy and applicable clinical trial cases as follows:
- Apply this adjustor when calculating payments for expanded access use of immunotherapy and applicable clinical trial cases that group to MS-DRG 018 by multiplying the relative weight for MS-DRG 018 by the adjustor.
We refer the readers to section II.D. of the preamble of this final rule for further discussion of our methodology.
Consistent with our calculation of the proposed adjustor for the relative weight calculations, for the proposed rule we proposed to calculate this adjustor based on the December 2023 update of the FY 2023 MedPAR file for purposes of establishing the FY 2025 payment amount. Specifically, in accordance with 42 CFR 412.85 (for operating IPPS payments) and 42 CFR 412.312 (for capital IPPS payments), for the proposed rule, we proposed to multiply the FY 2025 relative weight for MS-DRG 018 by a proposed adjustor of 0.34 as part of the calculation of the payment for claims determined to be applicable clinical trial or expanded use access immunotherapy claims that group to MS-DRG 018, which includes CAR T-cell and non-CAR T-cell therapies and other immunotherapies. We also proposed to update the value of the adjustor based on more recent data for the final rule.
We did not receive any comments specifically relating to the proposed payment adjustment for applicable clinical trial and expanded access use immunotherapy cases and are therefore finalizing our proposal without modification. We are also finalizing our proposal to update the value of this adjustor based on more recent data for this final rule. Therefore, using the March 2024 update of the FY 2023 MedPAR data, we are finalizing an adjustor of 0.33 for FY 2025, which will be multiplied by the final FY 2025 relative weight for MS-DRG 018 as part of the calculation of the payment for claims determined to be applicable clinical trial or expanded use access immunotherapy claims that group to MS-DRG 018.
Under existing regulations at § 412.104, we provide an additional payment to a hospital for inpatient services provided to certain Medicare beneficiaries with ESRD who receive a dialysis treatment during a hospital stay, if the hospital's ESRD Medicare beneficiary discharges, excluding discharges classified into the MS-DRGs listed at § 412.104(a), where the beneficiary received dialysis services during the inpatient stay, are 10 percent ( print page 69386) or more of its total Medicare discharges. The additional payment (referred to as the ESRD add-on payment) is intended to lessen the impact of the added costs for hospitals that deliver inpatient dialysis services to a high concentration of ESRD Medicare beneficiaries ( 76 FR 51692 ). The additional payment is based on the average length of stay for ESRD beneficiaries in the facility times a factor based on the average direct cost of furnishing dialysis services during a usual beneficiary stay ( 49 FR 34747 ). The payment to a hospital equals the average length of stay of ESRD beneficiaries in the hospital, expressed as a ratio to 1 week, times the estimated weekly cost of dialysis multiplied by the number of ESRD beneficiary discharges not excluded under § 412.104(a). The average direct cost of dialysis was determined from data obtained in connection with establishing the composite rate reimbursement for outpatient maintenance dialysis ( 49 FR 34747 ).
On January 1, 2011, we implemented the ESRD PPS, a case-mix adjusted, bundled PPS for renal dialysis services furnished by ESRD facilities as required by section 1881(b)(14) of the Act, as added by section 153(b) of the Medicare Improvements for Patients and Providers Act of 2008 (MIPPA) ( Pub. L. 110-275 ). Section 1881(b)(14)(F) of the Act, as added by section 153(b) of MIPPA, and amended by section 3401(h) of the Patient Protection and Affordable Care Act (the Affordable Care Act) ( Pub. L. 111-148 ), established that beginning CY 2012, and each subsequent year, the Secretary of the Department of Health and Human Services (the Secretary) shall annually increase payment amounts by an ESRD market basket percentage increase, reduced by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act ( 74 FR 49927 ). The ESRD PPS replaced the basic case-mix adjusted composite rate payment system and the payment methodologies for separately billable outpatient renal dialysis items and services. Payment under Medicare Part B for outpatient renal dialysis services has been based entirely on the ESRD PPS since January 1, 2014 ( 78 FR 72160 ). The ESRD PPS pays ESRD facilities a case-mix-adjusted, bundled payment, which includes former composite rate services and ESRD-related drugs, laboratory services, and medical equipment and supplies ( 80 FR 68973 ). The ESRD PPS base rate is designed to reflect the average cost per-treatment of providing renal dialysis services. [ 224 ] The per treatment payment amount (that is, the ESRD PPS base rate, subject to applicable adjustments) [ 225 ] is typically applied to a regimen of three hemodialysis treatments per week. CMS updates the ESRD PPS base rate annually. We refer readers to the August 12, 2010, ESRD PPS final rule ( 75 FR 49030 through 49214 ) for additional details on the establishment of the ESRD PPS, including a discussion of the transition from the basic case-mix adjusted composite rate payment system to the ESRD PPS.
As described previously, under current regulations the ESRD add-on payment is based on the average direct cost of furnishing dialysis services determined from data obtained in connection with establishing the composite rate. Under the current regulations, the average cost of dialysis is reviewed and adjusted, if appropriate, at the time the composite rate reimbursement for outpatient dialysis is reviewed. The last time CMS updated the composite rate was in the CY 2013 ESRD PPS final rule ( 77 FR 67454 ), as this was the final year in which payments to ESRD facilities were based on a blend of the composite rate and the ESRD PPS. In light of the time that has passed since the last update to the composite rate, we proposed to change the methodology used to calculate the ESRD add-on payment under current regulations to the ESRD PPS base rate used under the ESRD PPS. In addition, since the renal dialysis services reflected in the ESRD PPS base rate do not include those services that are not essential for the delivery of maintenance dialysis (see § 413.171), using the ESRD PPS base rate to calculate the ESRD add-on payment would maintain consistency with the current calculation, which is based on the average costs determined to be directly related to the renal dialysis service, as determined from the composite rate.
As described previously, under § 412.104(b)(1), the ESRD add-on payment is based on the estimated weekly cost of dialysis and the average length of stay of ESRD beneficiaries for the hospital. In the FY2025 IPPS/LTCH PPS proposed rule ( 89 FR 35934 ), we proposed that effective for cost reporting periods beginning on or after October 1, 2024, the estimated weekly cost of dialysis would be calculated as the applicable ESRD PPS base rate (as defined in 42 CFR 413.171 ) multiplied by three, which represents the typical number of dialysis sessions per week. The ESRD PPS base rate is applicable for renal dialysis services furnished during the calendar year (CY) (that is, effective January 1 through December 31 each year) and updated annually (see § 413.196). In the FY 2025 IPPS/LTCH PPS proposed rule we proposed that the annual CY ESRD PPS base rate (as published in the applicable CY ESRD PPS final rule or subsequent corrections, as applicable) multiplied by three would be used to calculate the ESRD add-on payment for hospital cost reporting periods that begin during the Federal FY for the same year. For example, the CY 2025 ESRD PPS base rate would be used for all cost reports beginning during Federal FY 2025 (that is, for cost reporting periods starting on or after October 1, 2024, through September 30, 2025). The table that follows illustrates the applicable CY ESRD PPS base rate that would be used to determine the add-on amount for eligible discharges during the hospital's cost reporting periods beginning on or after October 1, 2024 (FY 2025) and on or after October 1, 2025 (FY 2026) under this methodology.
In the FY 2025 IPPS/LTCH PPS proposed rule, we noted that use of the applicable CY ESRD PPS base rate to determine the add-on payment amount for the hospital's discharges occurring during the entire cost reporting period based on the cost report's begin date would be consistent with the determination of eligibility for the ESRD add-on payment, which occurs at cost report settlement and is based on the discharges that occur during that cost reporting period.

In the FY 2025 IPPS/LTCH PPS proposed rule, we stated that the payment to a hospital would continue to be calculated as the average length of stay of ESRD beneficiaries in the hospital, expressed as a ratio to 1 week, multiplied by the estimated weekly cost of dialysis multiplied by the number of applicable ESRD beneficiary discharges. Specifically, for cost reporting periods beginning on or after October 1, 2024, the payment to a hospital would equal the average length of stay of ESRD beneficiaries in the hospital, expressed as a ratio to 1 week, multiplied by the estimated weekly cost of dialysis (calculated as the applicable ESRD PPS base rate (as defined in 42 CFR 413.171 ), multiplied by 3) multiplied by the number of ESRD beneficiary discharges except for those excluded under § 412.104(a).
In the FY2025 IPPS/LTCH PPS proposed rule, we proposed to revise the regulations under 42 CFR 412.104(b) to reflect this proposed change to the calculation of the payment amount for cost reporting periods beginning on or after October 1, 2024. We proposed to revise § 412.104(b)(2) to specify that, effective for cost reporting periods beginning on or after October 1, 2024, the estimated weekly cost of dialysis is calculated as 3 dialysis sessions per week multiplied by the applicable ESRD PPS base rate (as defined in 42 CFR 413.171 ) that corresponds with the fiscal year in which the cost reporting period begins. For example, the CY 2025 ESRD PPS base rate (multiplied by 3 to determine the estimated weekly cost of dialysis, as described previously) would apply for all hospital cost reporting periods beginning during FY 2025 (that is, for cost reporting periods beginning on or after October 1, 2024, through September 30, 2025). We proposed to make conforming changes to § 412.104(b)(3) and § 412.104(b)(4) to reflect the proposed change in methodology for calculating the ESRD add-on payment amount for cost reporting periods beginning on or after October 1, 2024.
Comment: Commenters supported our proposal to update the ESRD add-on payment amount for cost reporting periods beginning on or after October 1, 2024 by using the applicable CY ESRD PPS base rate.
After consideration of the public comments we received, we are finalizing our proposal, without modification, to update the ESRD add-on payment methodology effective for cost reporting periods beginning on or after October 1, 2024 to use the annual CY ESRD PPS base rate (as published in the applicable CY ESRD PPS final rule or subsequent corrections, as applicable) multiplied by three to calculate the ESRD add-on payment for hospital cost reporting periods that begin during the Federal FY for the same year. We are also revising §§ 412.104(b)(2), (b)(3), and § 412.104(b)(4), as proposed, to reflect the new methodology for calculating the ESRD add-on payment amount for cost reporting periods beginning on or after October 1, 2024.
As discussed in the CY 2024 OPPS/ASC proposed rule ( 88 FR 49867 ), on January 26, 2021, President Biden issued Executive Order 14001 , “A Sustainable Public Health Supply Chain” ( 86 FR 7219 ), which launched a whole-of-government effort to strengthen the resilience of medical supply chains, especially for pharmaceuticals and simple medical devices. This effort was bolstered subsequently by Executive Orders 14005, 14017, and 14081 ( 86 FR 7475 , 11849 , and 25711 , respectively). In June 2021, as tasked in Executive Order 14017 on “America's Supply Chains,” the Department of Health and Human Services released a review of pharmaceuticals and active pharmaceutical ingredients, analyzing risks in these supply chains and recommending solutions to increase their reliability. [ 226 ] In July 2021, as tasked in Executive Order 14001 , the Biden-Harris Administration also released the National Strategy for a Resilient Public Health Supply Chain, which laid out a roadmap to support reliable access to products for public health in the future, including through prevention and mitigation of medical product shortages. [ 227 ]
Over the last several years, shortages for critical medical products have persisted, with the average drug shortage lasting about 1.5 years. [ 228 ] For pharmaceuticals, even before the COVID-19 pandemic, nearly two-thirds of hospitals reported more than 20 drug shortages at any one time—from antibiotics used to treat severe bacterial infections to crash cart drugs necessary to stabilize and resuscitate critically ill adults. [ 229 ] The frequency and severity of these supply disruptions has only been exacerbated over the last few years. [ 230 ]
Recent data suggests that hospitals are estimated to spend more than 8.6 million personnel hours and $360 million per year to address drug shortages, [ 231 ] which will likely further ( print page 69388) result in treatment delays and denials, changes in treatment regimens, medication errors, [ 232 233 234 ] as well as higher rates of hospital-acquired infections and in-hospital mortality. [ 235 236 ] The additional time, labor, and resources required to navigate drug shortages and supply chain disruptions also increase health care costs. [ 237 238. ]
Hospitals' procurement preferences can be leveraged to help foster a more resilient supply of lifesaving drugs and biologicals. With respect to shortages, supply chain resiliency includes having sufficient inventory that can be leveraged in the event of a supply disruption or demand increase—as opposed to relying on “just-in-time” inventory-management efficiency at the manufacturer level that can leave supply chains vulnerable to shortage. [ 239 240 ] This concept is especially true for essential medicines, which generally comprise products that are medically necessary to have available at all times in an amount adequate to serve patient needs and in the appropriate dosage forms. A hospital's resilient supply can also include essential medicines from multiple manufacturers, including the availability of domestic pharmaceutical manufacturing capacity, to diversify the sourcing of essential medicines. We stated that we believe it is necessary to support practices that can mitigate the impact of pharmaceutical shortages of essential medicines and promote resiliency to safeguard and improve the care hospitals are able to provide to beneficiaries. Additionally, sustaining sources of domestically sourced medical supplies can help support continued availability in the event of public health emergencies and other disruptions. This concept is consistent with our current policy for domestic National Institute for Occupational Safety and Health (NIOSH) approved surgical N95 respirators ( 87 FR 72037 ). Hospitals, as major purchasers and users in the U.S. of essential medicines, can support the existence of domestic sources by sourcing domestically made essential medicines.
When hospitals have insufficient supply of essential medicines, such as during a shortage, care for Medicare beneficiaries can be negatively impacted. To mitigate negative care outcomes in the event of insufficient supply, hospitals can adopt procurement strategies that foster a consistent, safe, stable, and resilient supply of these essential medicines. Such procurement strategies can include provisions to maintain or otherwise provide for extra stock of product (for example, either to maintain or to hold directly at the hospital, arrange contractually for a distributor to hold off-site, or arrange contractually with a wholesaler for a manufacturer to hold product) which can act as a buffer in the event of an unexpected increase in product use or disruption to supply. In the event an essential medicine goes into shortage without existing procurement or substitution strategies for affected drugs, negative patient care outcomes can result in reduced quality of care and, in some instances, increased costs by the Medicare program to provide payment for unnecessary services that could have been avoided had the drug been available to the hospital.
In the CY 2024 OPPS/ASC proposed rule ( 88 FR 49867 ), CMS requested public comments on a potential Medicare payment policy that would provide separate payment to hospitals under the IPPS for Medicare's share of the inpatient costs of establishing and maintaining access to a 3-month buffer stock of one or more of 86 essential medicines (referred to herein as the “CY 2024 Request for Comment”). Under this potential policy, the allowable costs would have included the hospital's reasonable costs of establishing and maintaining buffer stock(s) of the essential medicines but not the cost of the medicines themselves. We stated that we expected that the resources required to establish and maintain access to a buffer stock of essential medicines would generally be greater than the resources required to establish and maintain access to these medicines without such a buffer stock. While CMS did not finalize any policy regarding payment under the IPPS and OPPS for establishing and maintaining access to essential medicines, we stated we intended to propose new Conditions of Participation in forthcoming notice and comment rulemaking addressing hospital processes for pharmaceutical supply and that we would continue to consider policies related to buffer stock.
As discussed in the CY 2024 OPPS/ASC final rule, many commenters on the CY 2024 Request for Comment supported CMS's efforts to promote resiliency but expressed concerns regarding the potential for such a payment policy to induce or exacerbate drug shortages through demand shocks to the supply chain. Some commenters stated that a 3-month buffer stock may be inadequate to insulate hospitals from drug shortages, and that the policy may encourage hoarding behaviors and further fragment the existing supply of essential medicines, which would primarily disadvantage smaller, less resourced hospitals ( 88 FR 82129 through 82130 ). While commenters stated that a 3-month buffer stock may be inadequate to insulate hospitals from shortages given the duration of many drug shortages, some commenters further stated that even a 6-month buffer stock may not fully protect hospitals in the event of a shortage. Commenters cautioned that drug shortages are difficult to predict and often due to problems at the manufacturer level, which can be compounded by panic buying and hoarding behaviors. Some commenters stated that any buffer stock would need to be sufficiently large to account for the ramp up time that manufacturers need to reestablish supply of a given drug in shortage.
As a first step in this initiative, and based on consideration of the comments we received on the CY 2024 Request for Comment, for cost reporting periods beginning on or after October 1, 2024, we proposed to establish a separate payment under the IPPS to small (100 beds or fewer), independent hospitals for the estimated additional resource ( print page 69389) costs of voluntarily establishing and maintaining access to 6 month buffer stocks of essential medicines to foster a more reliable, resilient supply of these medicines for these hospitals. This proposed separate payment could be provided biweekly or as a lump sum at cost report settlement. As discussed further in section V.J.3. of the preamble of this final rule, we focused this proposal on small, independent hospitals, many of which are rural, that may lack the resources available to larger hospitals and hospital chains to establish and maintain buffer stocks of essential medicines for use in the event of drug shortages. We stated that we believe we can also mitigate concerns raised by commenters regarding large demand driven shocks to the supply chain by limiting separate payment to smaller, independent hospitals.
As stated in the proposed rule, the appropriate time to establish a buffer stock for a drug is before it goes into shortage or after a shortage period has ended. To further mitigate any potential for the proposed policy to exacerbate existing shortages or contribute to commenters' concerns of hoarding, if an essential medicine is listed as “Currently in Shortage” on the FDA Drug Shortages Database, [ 241 ] we proposed that a hospital that newly establishes a buffer stock of that medicine while it is in shortage would not be eligible for separate buffer stock payment for that medicine for the duration of the shortage. However, if a hospital had already established and was maintaining a buffer stock of that medicine prior to the shortage, we proposed that the hospital would continue to be eligible for separate buffer stock payment for that medicine for the duration of the shortage. We proposed that hospitals would continue to be eligible even if the number of months of supply of that medicine in the buffer stock were to drop to less than 6 months as the hospital draws down that buffer stock. We stated that once an essential medicine is no longer listed as “Currently in Shortage” in the FDA Drug Shortages Database, our proposed policy does not differentiate that essential medicine from other essential medicines and hospitals would be eligible to establish and maintain buffer stocks for the medicine as they would have before the shortage. We further stated that CMS will conduct provider education regarding additions and deletions to the publicly available FDA Drug Shortages Database to assist hospitals with this proposed policy.
As described in sections V.J.2. and .4. of the preamble of this final rule, we proposed that if the number of months of supply of medicine in the buffer stock were to drop to less than 6 months for a reason other than the essential medicine(s) actively being listed as “Currently in Shortage,” any separate payment to a hospital under this policy would be adjusted based on the proportion of the cost reporting period for which the hospital did maintain the 6-month buffer stock of that essential medicine.
We proposed to make this separate payment under the IPPS for the additional resource costs of establishing and maintaining access to buffer stocks of essential medicines under section 1886(d)(5)(I) of the Act, which authorizes the Secretary to provide by regulation for such other exceptions and adjustments to the payment amounts under section 1886(d) of the Act as the Secretary deems appropriate. We did not propose to make this payment adjustment budget neutral under the IPPS.
The report Essential Medicines Supply Chain and Manufacturing Resilience Assessment, as developed by the U.S. Department of Health and Human Services (HHS) Office of the Assistant Secretary for Preparedness and Response (ASPR) with the Advanced Regenerative Manufacturing Institute's (ARMI's) Next Foundry for American Biotechnology, prioritized 86 essential medicines (hereinafter referred to as the “ARMI List” or “ARMI's List”) from the Executive Order 13944 List of Essential Medicines, Medical Countermeasures, and Critical Inputs (hereinafter referred to as the “ E.O. 13944 List”), as developed under the E.O. by the U.S. Food and Drug Administration (FDA). [ 242 ]
The ARMI List is a prioritized list of 86 medicines that are either critical for minimum patient care in acute settings or important for acute care with no comparable alternatives available. The medicines included in the ARMI List were considered, by consensus, to be most critically needed for typical acute patient care. In this context, acute patient care was defined as: rescue use or lifesaving use or both (that is, Intensive Care Units, Cardiac/Coronary Care Units, and Emergency Departments), stabilizing patients in hospital continued care to enable discharge, and urgent or emergency surgery.
Development of the ARMI List focused on assessing the clinical criticality and supply chains of small molecules and therapeutic biologics. The development of the ARMI List was informed by meetings with multiple key pharmaceutical supply chain stakeholders (for example, manufacturers, group purchasing organizations, wholesale distributors, providers, pharmacies), surveys and workshops with groups of clinicians and industry stakeholders, public feedback on the E.O. 13944 List (provided during a public comment period starting in October 2020), and other research.
We proposed that for purposes of the separate payment under the IPPS, the costs of buffer stocks that would be eligible for separate payment are the additional resource costs of establishing and maintaining access to a 6-month buffer stock for any eligible medicines on ARMI's List of 86 essential medicines, including any subsequent revisions to that list of medicines. As previously discussed, the ARMI List represents a prioritized list of 86 medicines that were considered, by consensus, to be most critically needed for typical acute patient care. We stated that we believe that the ARMI List constitutes an appropriate set of medicines to initially prioritize under this proposed payment policy to help insulate small, independent hospitals, and the inpatient care they provide, from the negative effects of drug shortages.
As noted earlier, the appropriate time to establish a buffer stock for a drug is before it goes into shortage or after a shortage period has ended. If an essential medicine is listed as “Currently in Shortage” on the FDA Drug Shortages Database, we proposed that a hospital that newly establishes a buffer stock of that medicine while it is in shortage would not be eligible for separate buffer stock payment for that medicine for the duration of the shortage. However, if a hospital had already established and was maintaining a buffer stock of that medicine prior to the shortage, we proposed that the hospital would continue to be eligible for separate buffer stock payment for that medicine for the duration of the shortage as the hospital draws down that buffer stock even if the number of months of supply of that medicine in the buffer stock were to drop to less than 6 months. By proposing to limit eligibility in this way, we stated that we believed that we can ( print page 69390) both insulate smaller hospitals from short-term drug shortages and mitigate the potential for the proposed policy to exacerbate existing shortages or contribute to concerns of hoarding.
As an illustrative example, suppose a hospital established and maintained 6-month buffer stocks for five essential medicines. However, one of those essential medicines was subsequently listed as “Currently in Shortage” on the FDA Drug Shortages Database. The hospital would no longer be required to maintain a 6-month buffer stock of the essential medicine that is in shortage to receive separate payment for maintaining the buffer stock of that essential medicine during the period of shortage. The hospital would continue to be eligible for the separate payment from CMS for the buffer stock for that medicine during the period of shortage as it draws down its established buffer stock of the medicine in shortage as needed. However, the hospital would be required to maintain buffer stocks of no less than 6 months for the other four essential medicines that are not in shortage to be eligible to receive separate payment for those four medicines.
Because medicine can remain on the FDA Drug Shortage Database for years, we requested comments on the duration that CMS should continue to pay hospitals for the maintenance of a less than 6-month buffer stock of the essential medicine if it is “Currently in Shortage.” We also requested comments on if there is a quantity or dosage minimum floor where CMS should no longer pay to maintain a 6-month buffer stock of the essential medicine if it is “Currently in Shortage.”
We proposed that if the ARMI List is updated to add or remove any essential medicines, all medicines on the updated list would be eligible for separate payment under this policy for the IPPS shares of the costs of establishing and maintaining access to 6-month buffer stocks as of the date the updated ARMI List is published. To the extent that in the future other medicines or lists are identified for eligibility in future iterations of this policy, we sought comment on the potential mechanism and timing for incorporating those updates. We stated that comments could consider, among other factors, medicines that were excluded from the ARMI List, the E.O. 13944 List, or both. For example, some categories from the E.O. 13944 List—including Blood and Blood Products, Fractionated Plasma Products, Vaccines, and Volume Expanders—were excluded from the ARMI List due to differences in their supply chains. Additionally, other categories were identified as not needed for routine/typical acute patient care (that is, Biological Threat Medical Countermeasures, Burn and Blast Injuries, Chemical Threat Medical Countermeasures, Pandemic Influenza Medical Countermeasures, Radiologic-Nuclear Threat Medical Countermeasures). The ARMI List does not include certain medicines that have recently been in shortage and that may be considered essential and are more prevalent in specific care settings other than an inpatient hospital, such as drugs used in oncology care on an outpatient basis. Further, there are medicines that are not included on the ARMI List nor the E.O. 13944 List, such as buprenorphine-based medications for treatment of substance use disorder. We sought comment on whether eligibility for separate payment for the IPPS share of the costs of establishing and maintaining access to 6-month buffer stocks of essential medicines should include oncology drugs or other types of drugs not currently on the ARMI List.
We stated in the proposed rule that CMS would conduct provider education regarding additions and deletions to the publicly available FDA Drug Shortages Database to assist hospitals with this proposed policy.
Commenters on the CY 2024 Request for Comment ( 88 FR 82129 through 82130 ) raised a number of concerns relating to access to essential medicines for small hospitals and potential hoarding behaviors among better resourced hospitals. Commenters also cautioned against the potential for the policy to cause demand-driven shocks to the pharmaceutical supply chain, exacerbating pharmaceutical access issues for hospitals, which they claimed would disproportionately impact smaller hospitals due to their smaller purchasing power. As hospitals and hospital systems increase in size through expansion of bed count or consolidation or both and vertical integration with other hospitals and health systems, they accrue bargaining leverage for payment negotiations and thereby increase their purchasing power. [ 243 ] Those smaller (and often rural) hospitals that lack this increased purchasing power are faced with potentially lower payments from payers and less operating capital. [ 244 ] To address this concern, and attempt to better insulate these smaller, independent hospitals against future supply disruptions of essential medicines, we proposed to limit eligibility for separate payment for the resource costs of establishing and maintaining access to buffer stocks of essential medicines to small, independent hospitals that are paid under the IPPS, as defined later in this section. As many of these small, independent hospitals are located in rural areas, we stated that we also expect this policy to support rural hospitals, in line with the rural health strategy of the Biden-Harris Administration. [ 245 246 ]
We stated that we believe that by focusing eligibility on small, independent hospitals, we can both support these types of hospitals in their efforts to provide patient care during drug shortages and lessen any potential demand shocks to the pharmaceutical supply chain because the buffer stocks these hospitals would require are likely smaller compared to larger hospitals and hospital chains. As discussed further in the regulatory impact analysis associated with this proposed policy in section I.G.6. of Appendix A of the proposed rule, we initially identified 493 potentially eligible hospitals based on FY 2021 hospital cost report data. Of these hospitals, 249 were identified as geographically rural, 6 were identified as geographically urban but reclassified as rural (under our reclassification regulations at § 412.103), and 238 were identified as geographically urban without a reclassification as rural. These hospitals had 216,557 Medicare discharges in total and an average of 442 Medicare discharges per hospital for the FY 2021 cost reporting year.
Small Hospital: For the purposes of this policy, we proposed to define a small hospital as one with not more than 100 beds. This definition is consistent with the definition of a small ( print page 69391) hospital used for Medicare-dependent, small rural hospitals (MDH) in section 1886(d)(5)(G)(iv)(II) of the Act. Consistent with the MDH regulations at § 412.108(a)(1)(ii), we proposed that a hospital would need to have 100 or fewer beds as defined in § 412.105(b) during the cost reporting period for which it is seeking the payment adjustment to be considered a small hospital for purposes of this payment adjustment. We requested comment on using criteria other than the MDH bed size criterion to identify small hospitals for the purposes of this proposed payment policy.
Independent Hospital: For the purposes of this policy, we proposed to define an independent hospital as one that is not part of a chain organization, as defined for purposes of hospital cost reporting. A chain organization is defined as a group of two or more health care facilities which are owned, leased, or through any other device, controlled by one organization. This proposed definition is the definition of chain organization in CMS Pub 15-1, Provider Reimbursement Manual, Chapter 21, Cost Related to Patient Care § 2150: “Home Office Costs—Chain Operations” and used by a hospital when completing its cost report.
Because this proposed definition is the definition of chain organization used by a hospital when filling out its cost report, to operationalize our proposed separate payment policy, we proposed that any hospital that appropriately answers “yes” (denoted “Y”) to line 140 column 1 or fills out any part of lines 141 through line 143 on Worksheet S-2, Part I, on Form CMS-2552-10 would be considered to be part of a chain organization and not independent, and therefore not eligible for separate payment under this proposal. Please see Table V.J.-01 for a partial example of this section of Form CMS-2552-10.

Thus, we proposed that to be eligible for this separate payment, under this policy, a hospital would need to be a small hospital with 100 or fewer beds and meet the definition of independent described previously. We sought comment on our proposed eligibility criteria and proposed definition of a small, independent hospital.
We note that critical access hospitals (CAHs) are paid for inpatient and outpatient services at 101 percent of Medicare's share of reasonable costs, including Medicare's share of the reasonable costs of establishing and maintaining access to buffer stocks of medicines. We sought comment on the use of buffer stocks by CAHs, including the medicines in the buffer stocks, the costs of establishing and maintaining the buffer stocks, whether CAHs tend to contract out this activity, and any barriers that CAHs may face in establishing and maintaining access to buffer stocks.
As summarized in the CY 2024 OPPS/ASC final rule and section V.J.1. of the preamble of this final rule, some commenters on the CY 2024 Request for Comment expressed concerns that a 3-month supply of essential medicines may not be sufficient to adequately insulate hospitals from the detrimental effects of future drug shortages. Commenters stated that drug shortages often persist for durations of time in excess of 3 months, such that a 3-month buffer stock may be inadequate to insulate hospitals from the longer-term effects of drug shortages. As noted in section V.J.1. of the preamble of this final rule, drug shortages generally persist for many months, and some research suggests that these shortages last for an average of 1.5 years. Accordingly, we stated in the proposed rule that we believe a buffer stock of at least 6 months would better support small, independent hospitals in contending with future shortages. To better address commenters' concerns and hospital needs during drug shortages, we proposed separate payment for the costs of establishing and maintaining access to a buffer stock that is sufficient for no less than a 6-month period of time for each of one or more essential medicines. As discussed in section V.J.5 of the preamble of this final rule, we also sought comments on whether a phase-in approach that, for example, would provide separate payment for establishing and maintaining access to a 3-month supply for the first year in which the policy is implemented and a 6-month supply for all subsequent years would be appropriate.
We stated in the proposed rule that in estimating the amount of a buffer stock needed for each essential medicine, the hospital should consider that the amount needed to maintain a buffer stock could vary month to month and ( print page 69392) throughout the applicable months of the cost reporting period; that is, a hospital's historical use of a medicine may indicate that it is typically needed more often in January than June, for example. Accordingly, we stated the size of the buffer stock should reflect this anticipated variation and be based on a reasonable estimate of the hospital's need for that essential medicine in the upcoming 6-month period. We stated this estimate would be determined by the hospital and could be based on the historical usage of the essential medicine by the hospital for that 6-month period in a prior year, or another reasonable method to estimate its need for that upcoming period. We stated that if a hospital did not maintain a 6-month buffer stock of an essential medicine for an entire cost reporting period, any separate payment to the hospital under this policy would be adjusted based on the proportion of the cost reporting period for which the hospital did maintain the 6-month buffer stock of that essential medicine. As described in section V.J.2 of the preamble of this final rule, we stated in the proposed rule that in the event that a hospital is not able to maintain a buffer stock of at least 6 months due to one or more of their chosen medicine(s) being listed as “Currently in Shortage” on the FDA's Drug Shortage Database after establishment of the buffer stock under this policy, the hospital would continue to be eligible for the buffer stock payment for the medicine(s) in shortage as the hospital draws down the buffer stock even if the number of months of supply of that medicine in the buffer stock were to drop to less than 6 months. We stated that hospitals would be permitted to use multiple contracts to establish and maintain at least a 6-month buffer stock for any given essential medicine.
As discussed in the CY 2024 Request for Comment, CMS requested public comments on a potential separate payment under the IPPS for the additional, reasonable costs of establishing and maintaining a 3-month buffer stock of one or more essential medicine(s). We stated that participating hospitals could establish and maintain their buffer stocks directly, or through contractual arrangements with pharmaceutical distributors, intermediaries, or manufacturers.
We received comments in response to the CY 2024 Request for Comment stating that hospitals that maintain buffer stocks of essential medicines typically do so through upstream entities, such as pharmaceutical group purchasing organizations and manufacturers. Furthermore, these commenters stated that hospitals typically lack the capacity to stockpile large quantities of essential medicines directly. Some of these commenters stated that any buffer stocks established under the potential policy should be maintained by upstream intermediaries or a neutral third party instead of directly maintained by hospitals, as they stated that these upstream intermediaries are generally better positioned and equipped to maintain these buffer stocks. While other commenters were receptive to directly maintaining their buffer stock(s) or indicated that they already maintained substantial buffer stocks of medicines, these commenters were generally larger, better resourced hospitals or hospital systems.
In this year's proposed rule, we stated that we agreed with commenters that pharmaceutical intermediaries and manufacturers are generally better positioned to establish and maintain larger (for example, 6-month or greater) buffer stocks of essential medicines, as small, independent hospitals may generally lack the space, staff, and specific equipment (like large-scale refrigeration and large, onsite storage) to directly maintain 6-month buffer stock(s) of essential medicine(s). While we stated that we anticipate that most hospitals that elect to establish and maintain buffer stocks under this policy will do so through contractual arrangements with pharmaceutical intermediaries, manufacturers, and distributors, we proposed that the additional resource costs associated with directly maintaining 6-month buffer stock(s) of essential medicine(s) would also be eligible for separate payment under this policy. Accordingly, we proposed that for purposes of the proposed separate payment under the IPPS to small, independent hospitals for the estimated additional resource costs of voluntarily establishing and maintaining access to 6-month buffer stocks of essential medicines, those costs associated with establishing and maintaining access to 6-month buffer stocks either directly or through contractual arrangements with pharmaceutical manufacturers, intermediaries, or distributors would be eligible for additional payment under this policy. These costs do not include the cost of the medicines themselves which would continue to be paid in the current manner. We also noted that the proposed payment is only for the IPPS share of the costs of establishing and maintaining access to buffer stock(s) of one or more essential medicine(s).
We noted the costs associated with directly establishing and maintaining a buffer stock may include utilities like cold chain storage and heating, ventilation, and air conditioning, warehouse space, refrigeration, management of stock including stock rotation, managing expiration dates, and managing recalls, administrative costs related to contracting and record-keeping, and dedicated staff for maintaining the buffer stock(s). We requested comments on other types of costs intrinsic to directly establishing buffer stocks of essential medicines that should be considered eligible for purposes of separate payment under this policy. We also requested comment regarding whether staff costs would increase with the number of essential medicines in buffer stock, and whether there would be efficiencies if multiple hospitals elect to establish buffer stocks of essential medicines with the same pharmaceutical manufacturer, intermediary, or distributor.
We also requested comment on whether this proposed policy should be phased in by the size of the buffer stock to address concerns about infrastructure investments that may be needed to store and maintain the supply. We also referred readers to the Collection of Information Requirements in section XII.B.2. of the preamble of the proposed rule regarding the estimated burden associated with this policy proposal and sought comment on whether there are any other potential methods for hospitals to report costs included under this policy besides the forthcoming supplemental cost reporting worksheet.
Currently, payment for the resources required to establish and maintain access to medically reasonable and necessary drugs and biologicals is generally part of the IPPS payment. As noted in section V.J.2. of the preamble of this final rule, we expect that the resources required to establish and maintain access to buffer stocks of essential medicines will generally be greater than the resources required to establish and maintain access to these medicines without such buffer stocks. Given these additional resource costs and our concern that small, independent hospitals may lack the resources available to larger hospitals and hospital chains to establish buffer stocks of essential medicines, we stated that we believe it is appropriate to propose to pay these hospitals separately for the additional resource costs associated with voluntarily establishing and maintaining access, ( print page 69393) either directly or through contractual arrangements, to buffer stocks of essential medicines. As also noted in section V.J.2 of the preamble of this final rule, we proposed that if the ARMI List is updated to add or remove any essential medicines, all medicines on the updated list would be eligible for separate payment under this policy for the IPPS shares of the costs of establishing and maintaining access to 6-month buffer stocks as of the date the updated ARMI List is published. Any medicine(s) that are removed from the ARMI List in any future updates to the list would no longer be eligible for separate payment under this policy for the IPPS shares of the costs of establishing and maintaining access to 6-month buffer stocks as of the date the updated ARMI List is published.
CMS proposed to base the IPPS payment under this policy on the IPPS shares of the additional reasonable costs of a hospital to establish and maintain access to its buffer stock. The use of IPPS shares in this payment adjustment would be consistent with the use of these shares for the payment adjustment for domestic NIOSH approved surgical N95 respirators, which is based on the IPPS and OPPS shares of the difference in cost between domestic and non-domestic NIOSH approved surgical N95 respirators for the cost reporting period in which costs are claimed ( 87 FR 72037 ). We stated that the hospital would report these costs to CMS on the forthcoming supplemental cost reporting worksheet associated with this proposed policy. The hospital's costs may include costs associated with contractual arrangements between the hospital and a manufacturer, distributor, or intermediary or costs associated with directly establishing and maintaining buffer stock(s). We further stated that these costs would not include the costs of the essential medicine itself, which would continue to be paid in the current manner.
If a hospital establishes and maintains access to buffer stock(s) of essential medicine(s) through contractual arrangements with pharmaceutical manufacturers, intermediaries, or distributors, we stated that the hospital would be required to disaggregate the costs specific to establishing and maintaining the buffer stock(s) from the remainder of the costs present on the contract for purposes of reporting these disaggregated costs under this proposed policy. This disaggregated information, reported by the hospital on the new supplemental cost reporting worksheet, along with existing information already collected on the cost report, would be used to calculate a Medicare payment for the IPPS share of the hospital's costs of establishing and maintaining access to the buffer stock(s) of essential medicine(s).
If a hospital contracts with one or more manufacturers or wholesalers or other intermediaries to establish and maintain 6-month buffer stocks of one or more essential medicines, we stated that the hospital must clearly identify those costs separately from the costs of other provisions of the contract(s). As a simplified example for purposes of illustration, suppose a hospital has a $500,000 contract with a pharmaceutical wholesaler. The contract is for pharmaceutical products, 50 of which are qualifying essential medicines. Additionally, the contract contains a provision for the wholesaler to establish and maintain 6-month buffer stocks of those 50 essential medicines on the hospital's behalf. The contract further specifies that $10,000 of the $500,000 is for the provision of the contract that establishes and maintains the 6-month buffer stocks of those 50 essential medicines. This $10,000 amount does not include any costs to the hospital for the drugs themselves which, as previously noted, would continue to be paid in the current manner. We explained that under this proposal, the hospital would report the $10,000 cost for establishing and maintaining the 6-month buffer stocks of the 50 essential medicines on the supplemental cost reporting worksheet. That $10,000 cost, in addition to other information already existing on the cost report, would be used to calculate the additional payment under this policy including the hospital-specific Medicare IPPS share percentage of this cost, expressed as the percentage of inpatient Medicare costs to total hospital costs. We stated that on average for the small, independent hospitals that are eligible for this policy, the Medicare IPPS share percentage is approximately 11 percent.
If a hospital chooses to directly establish and maintain buffer stock(s) of one or more essential medicines under this policy, we stated that the hospital would be required to report the additional costs associated with establishing and maintaining its buffer stock(s) on the supplemental cost reporting form. We stated that the hospital should clearly specify the total additional resource costs to establish and maintain its 6-month buffer stock(s) of essential medicine(s). As in the previous example, this amount should not include the cost of the essential medicine(s) themselves and would be used, along with other information already existing on the cost report, to calculate the additional payment under this policy.
Additionally, we stated that we would anticipate that when a hospital contracts with one or more manufacturers or wholesalers or other intermediaries to establish and maintain 6-month buffer stocks of one or more essential medicines, it would ensure that a discrete buffer stock is maintained for that hospital. For example, if two hospitals held contracts with a manufacturer arranging for 6-month buffer stocks of certain essential medicines, the hospitals would verify that the manufacturer is maintaining sufficient total buffer stock to account for the 6-month demand of both hospitals in aggregate.
We stated that we seek to support the establishment of buffer stocks when drugs are not currently in shortage to promote the overall resiliency of drug supply chains. As previously discussed, we proposed that buffer stocks for any of the essential medicines on the ARMI List that are listed as “Currently in Shortage” on the FDA Drug Shortages Database would not be eligible for additional payment under this policy for a hospital's cost reporting period unless the hospital had already established and was maintaining a buffer stock of that medicine prior to the shortage. Additionally, we proposed that any essential medicine(s) for which a hospital has successfully established and maintained a buffer stock(s) of at least 6 months that is subsequently listed as “Currently in Shortage” on the FDA Drug Shortages Database would be exempt from the requirement to maintain a 6-month supply of such essential medicine(s) for the duration of the period in which the medicine is in shortage. We stated that we are interested in public comments on the burden associated with hospitals' monitoring of the FDA Drug Shortage Database, and excluding from the cost report any resource costs associated with maintaining a buffer stock of an essential medicine that was listed as “Currently in Shortage,” except where the hospital had already established and was maintaining a 6-month buffer stock of that medicine prior to the shortage. We stated that as of the date that medicine is no longer listed as “Currently in Shortage,” eligibility for separate payment to the hospital for the drug in shortage would be prospectively adjusted based on the proportion of the cost reporting period for which the hospital does maintain the 6-month buffer stock of that essential medicine. Once an essential medicine is no longer listed as “Currently in Shortage” in the FDA Drug Shortages Database, our ( print page 69394) proposed policy does not differentiate that essential medicine from other essential medicines. However, we also sought comment on whether some minimum period, such as 6 months, should elapse after a shortage of a given essential medicine is resolved before that medicine can become eligible for separate payment under this proposed policy.
We proposed to make separate payments for the IPPS shares of these additional resource costs of establishing and maintaining access to buffer stocks of essential medicines. Payment could be provided as a lump sum at cost report settlement or biweekly as interim lump-sum payments to the hospital, which would be reconciled at cost report settlement. In accordance with the principles of reasonable cost as set forth in section 1861(v)(1)(A) of the Act and in 42 CFR 413.1 and 413.9 , Medicare could make a lump-sum payment for Medicare's share of these additional inpatient costs at cost report settlement. Alternatively, a provider may make a request for biweekly interim lump sum payments for an applicable cost reporting period, as provided under 42 CFR 413.64 (Payments to providers: Specific rules) and 42 CFR 412.116(c) (Special interim payments for certain costs). These payment amounts would be determined by the Medicare Administrative Contractor (MAC) consistent with existing policies and procedures. In general, interim payments are determined by estimating the reimbursable amount for the year using Medicare principles of cost reimbursement and dividing it into 26 equal biweekly payments. The estimated amount would be based on the most current cost data available, which will be reviewed and, if necessary, adjusted at least twice during the reporting period. (See CMS Pub 15-1 § 2405.2 for additional information). The MACs would determine the interim lump-sum payments based on the data the hospital may provide that reflects the information that would be included on the new supplemental cost reporting form. CMS is separately seeking comment through the Paperwork Reduction Act (PRA) process on a supplemental cost reporting form that would be used for this purpose. We stated that in future years, the MACs could determine the interim biweekly lump-sum payments utilizing information from the prior year's cost report, which may be adjusted based on the most current data available. This is consistent with the current policies for medical education costs, and bad debts for uncollectible deductibles and coinsurance paid on interim biweekly basis as noted in CMS Pub 15-1 § 2405.2. It is also consistent with the payment adjustment for domestically sourced NIOSH approved surgical N95 respirators ( 87 FR 72037 ).
We proposed to codify this payment adjustment in the regulations by adding new paragraph (g) to 42 CFR 412.113 to state the following:
- Essential medicines are the 86 medicines prioritized in the report Essential Medicines Supply Chain and Manufacturing Resilience Assessment developed by the U.S. Department of Health and Human Services Office of the Assistant Secretary for Preparedness and Response and published in May of 2022, and any subsequent revisions to that list of medicines. A buffer stock of essential medicines for a hospital is a supply, for no less than a 6-month period, of one or more essential medicines.
- The additional resource costs of establishing and maintaining access to a buffer stock of essential medicines for a hospital are the additional resource costs incurred by the hospital to directly hold a buffer stock of essential medicines for its patients or arrange contractually for such a buffer stock to be held by another entity for use by the hospital for its patients. The additional resource costs of establishing and maintaining access to a buffer stock of essential medicines does not include the resource costs of the essential medicines themselves.
- For cost reporting periods beginning on or after October 1, 2024, a payment adjustment to a small, independent hospital for the additional resource costs of establishing and maintaining access to buffer stocks of essential medicines is made as described in paragraph (g)(4) of this section. For purposes of this section, a small, independent hospital is a hospital with 100 or fewer beds as defined in § 412.105(b) during the cost reporting period that is not part of a chain organization, defined as a group of two or more health care facilities which are owned, leased, or through any other device, controlled by one organization.
- The payment adjustment is based on the estimated reasonable cost incurred by the hospital for establishing and maintaining access to buffer stocks of essential medicines during the cost reporting period.
We also proposed to make conforming changes to 42 CFR 412.1(a) and 412.2(f) to reflect this proposed payment adjustment for small, independent hospitals for the additional resource costs of establishing and maintaining access to buffer stocks of essential medicines.
In summary, for cost reporting periods beginning on or after October 1, 2024, we proposed to establish a separate payment under the IPPS to small, independent hospitals for the additional resource costs involved in voluntarily establishing and maintaining access to 6-month buffer stocks of essential medicines, either directly or through contractual arrangements with a manufacturer, distributor, or intermediary. We proposed that the costs of buffer stocks that are eligible for separate payment are the costs of buffer stocks for one or more of the medicines on ARMI's List of 86 essential medicines. The separate payment would be for the IPPS share of the additional costs and could be issued in a lump sum, or as biweekly payments to be reconciled at cost report settlement. We proposed that the separate payment would not apply to buffer stocks of any of the essential medicines on the ARMI List that are currently listed as “Currently in Shortage” on the FDA Drug Shortages Database unless a hospital had already established and was maintaining a 6-month buffer stock of that medicine prior to the shortage. Once an essential medicine is no longer listed as “Currently in Shortage” in the FDA Drug Shortages Database, we stated that our proposed policy does not differentiate that essential medicine from other essential medicines and hospitals would be eligible to establish and maintain buffer stocks for the medicine as they would have before the shortage. CMS is separately seeking comment through the PRA process on a supplemental cost reporting form for this proposed payment.
After consideration of the comments received on our proposal, which we summarize and respond to in the section that follows, we are finalizing the proposed separate payment under the IPPS to small, independent hospitals for the additional resource costs involved in voluntarily establishing and maintaining access to 6-month buffer stocks of essential medicines. In future years as we gain experience under this policy, we plan to assess the program's impact and consider revisions, where appropriate, to help ensure availability of essential medicines for patients.
Comment: Overall, the majority of commenters were generally supportive of our proposed separate payment for a hospital's cost to maintain buffer stock. Those that were opposed to the policy generally expressed concerns regarding ( print page 69395) potential “unintended consequences” that may arise from establishing separate stockpiles of essential medicines throughout the country. Commenters that were opposed to the policy generally echoed concerns that they previously expressed in their comments on our prior Request for Comment in the CY 2024 OPPS/ASC proposed rule, including that the proposed policy could contribute to fragmentation of the pharmaceutical supply chain and had the potential to induce new drug shortages or exacerbate existing shortages.
Many commenters requested that we expand or otherwise modify our eligibility requirements of small, independent hospitals of 100 beds or fewer that are not part of a chain organization. Some commenters had specific recommendations for provider types that should be made eligible for the proposed policy, regardless of bed size or independent status, with particular emphasis on CAHs, MDHs, SCHs, children's hospitals, and various outpatient facilities. Several commenters requested that we remove entirely the independent status eligibility requirement, stating that hospitals that are part of chain organizations also face substantial financial obstacles. Other commenters requested that we expand the policy to make all Medicare providers eligible. A commenter requested that we further restrict the pool of eligible providers to test the effects of the proposed policy on the pharmaceutical supply chain.
Some commenters, including MedPAC, indicated that Medicare payment policy is neither a sufficient, nor the best suited, mechanism to support adequate supplies of essential medicines. These commenters generally expressed support for broader policy initiatives beyond the Medicare program to address drug shortages.
Response: We appreciate all the comments received on our proposed policy. We also recognize the general concerns of some commenters that the current lack of resiliency in the pharmaceutical supply chain makes it potentially sensitive to fragmentation or significant demand shocks from additional pharmaceutical purchasing. However, we continue to believe that our pool of eligible hospitals is sufficiently small and has significantly less purchasing power than larger hospitals and hospital chains, such that the policy would not create such demand shocks or result in fragmentation that would cause or exacerbate shortages. HHS will continue to monitor drug shortages, [ 247 ] and will propose as needed appropriate modifications to the policy, if any, in future rulemaking.
For similar reasons, we disagree with commenters who requested that we expand the pool of eligible hospitals now in this initial implementation of the new policy. Without the benefit of actual experience under the policy, expansion of the policy at this time to include hospitals with greater purchasing power than small, independent hospitals could risk inducing or exacerbating drug shortages. Similarly, we disagree that we should restrict the policy to exclude some hospitals with lesser purchasing power, as this policy is meant to assist these hospitals in responding to future drug shortages, and at the same time, we continue to believe that their purchasing power is such that it would not significantly increase the risk of inducing or exacerbating drug shortages, as compared to those hospitals with greater purchasing power. Accordingly, we believe the current scope of identified eligible hospitals is appropriate for purposes of the first year of this policy. As noted, we may consider any future modifications to the scope of eligible hospitals, including potential expansions to hospitals with larger bed counts or certain provider types, as we gain experience under this policy.
Furthermore, as stated in the proposed rule, we note that CAHs are already paid for inpatient and outpatient services at 101 percent of Medicare's share of reasonable costs, including Medicare's share of the reasonable costs of establishing and maintaining access to buffer stocks of medicines. We also note that MDHs and SCHs are not excluded from eligibility under this policy, provided they meet the bed size and independent status requirements. Children's hospitals are exempt from the IPPS and paid according to a hospital-specific target amount updated for inflation with the option to apply for a temporary or permanent adjustment to their target amount for the reasonable costs they incur in furnishing inpatient care to Medicare beneficiaries, including those costs attributable to buffer stocks of essential medicines.
After consideration of the comments received, we are finalizing our criteria for eligible hospitals without modification.
Comment: Some commenters asked that we shift the policy to a domestic add-on payment, similar to the domestic add-on payment for NIOSH-approved surgical N95 respirators. Commenters requested clarification on whether there is a domestic manufacturing requirement under this policy.
Response: We note that HHS has taken significant actions to enable investment in domestic manufacturing of essential medicines, medical countermeasures, and other critical inputs, and will continue to do so. [ 248 ] We note that while we continue to be supportive of domestic manufacturing of essential medicines, we are not requiring at this time that hospitals exclusively establish and maintain buffer stocks of domestically manufactured essential medicines to be eligible for separate payment under this policy. As we gain experience under our policy and as the domestic manufacturing capacity of essential medicines increases, we may consider the comments regarding domestic manufacturing requirements for future rulemaking.
Comment: Many commenters suggested phasing in the size of the buffer stock, with a 3-month minimum buffer stock size in the first year of implementation and a 6-month minimum buffer stock size in all subsequent years. These commenters stated that phasing in the policy may ease the upfront costs of establishing buffer stocks, as the costs of establishing a smaller initial buffer stock ( e.g., a 3-month or similarly sized buffer stock) would pose lesser costs than a 6- month buffer stock. These commenters also stated that phasing in the policy would lessen any potential impacts to the pharmaceutical supply chain and better allow manufacturers to ramp up production of essential medicines. Some commenters requested that we reduce the minimum buffer stock size to 3 months, stating that the small, independent hospitals that we targeted for eligibility would have higher upfront costs than most larger hospitals and hospital chains and those upfront costs would be lower with a 3-month buffer stock. These commenters stated that small, independent hospitals would struggle to establish 6-month buffer stocks due to the associated costs. Several commenters suggested that we permit a range of buffer stock sizes, from 2- to 6-month buffer stocks for example, or that we permit hospitals to determine the appropriate size of buffer stock for their chosen essential medicines. Commenters also suggested that we ( print page 69396) consider implementing a cap on the total volume of an applicable generic that any one hospital may obtain under our proposed policy.
Response: We agree with commenters that there are multiple factors to consider in determining the appropriate size of the buffer stock for purposes of this policy. As stated in the preamble of the proposed rule and as emphasized by several commenters, a 6-month buffer stock is generally more effective at mitigating shortages than a 3-month buffer stock. A commenter also stated, and we agree, that buffer stocks are not necessarily meant to supply a hospital for the duration of a shortage, but are needed to give other manufacturers time to produce and deliver more of the affected medicine. As such, 6 months provides manufacturers more time to respond as compared to 3 months or some other, shorter period.
However, we also recognize the concerns raised by commenters who believe a smaller buffer stock size would be more appropriate because the costs of establishing and maintaining buffer stocks of 6 months are greater than the costs for 3 months or other shorter periods. In weighing these competing concerns, at the present time we believe that providing separate payment for the longer 6-month buffer stock is the most appropriate policy because a longer period of buffer stock would better serve to bridge a drug shortage and provide manufacturers with more time to increase production of an affected medicine. However, as we gain experience under this policy, including the extent to which the size of the buffer stock may affect hospital participation, we may revisit this issue in future rulemaking.
In response to commenters who suggested that we consider implementing a cap on the total volume of an applicable generic that any one hospital may obtain under our proposed policy, given that the eligible hospitals are those with lesser purchasing power we do not think these hospitals would use their comparatively limited financial resources to establish buffer stocks of excessive size that would make the establishment of such a cap on the size of the applicable buffer stock for purposes of separate payment under this policy necessary at this time. However, although we do not believe this to be a likely outcome of our policy, we may further consider this issue for future rulemaking as we gain experience under this policy. We reiterate that establishment of one or more buffer stock(s) is purely voluntary on the part of eligible hospitals.
After consideration of the comments received, we are finalizing our proposal on the size of the buffer stock without modification.
Comment: Many commenters expressed concern about the administrative burden associated with the policy as proposed. Commenters stated that small, independent hospitals would likely have the highest relative costs associated with establishing and maintaining buffer stock(s) of essential medicines, including the administrative and staffing costs of separately tracking and maintaining buffer stock established under the proposed policy, as well as tracking the shortage status of eligible essential medicines. Commenters were generally opposed to the use of a supplemental cost reporting form to report the costs associated with establishing and maintaining a buffer stock, stating that this would increase administrative and recordkeeping costs for participating hospitals. Some commenters requested that we instead permit contracted manufacturers, distributors, and intermediaries to directly report the costs associated with establishing and maintaining a buffer stock for a hospital to CMS in lieu of the supplemental cost reporting form, or to base payment to hospitals on an attestation from contracted manufacturers, distributors, or intermediaries.
Response: We appreciate commenters' concerns regarding the administrative costs associated with separately reporting the costs of establishing and maintaining buffer stocks of essential medicines. However, as is the case for other Medicare payment policies based on reasonable cost, we continue to believe that the Medicare cost report is presently the most feasible and least burdensome method of collecting and being able to audit this cost information. While some commenters suggested CMS require contracted manufacturers, distributors, and intermediaries to report these costs directly to CMS, they did not suggest a mechanism for doing so.
We also appreciate commenters sharing their concerns regarding the administrative burden of tracking shortage status of eligible essential medicines. To help mitigate concerns of added administrative burden associated with tracking the shortage status of a given essential medicine, in connection with this final policy, the MACs will inform hospitals of all eligible medicines and their associated shortage status on a calendar quarter basis on or about the start of each quarter. The shortage status information that the MACs will provide to the hospital will be based on the shortage status of each essential medicine(s) as reported on the FDA's Drug Shortage Database. For example, hospitals will be informed by the MACs on or about January 1st each year which essential medicines are considered in shortage for purposes of this policy for the calendar year quarter starting January 1st. The MACs will similarly provide this information for the calendar year quarters beginning April 1st, July 1st, and October 1st.
After consideration of the comments received, we are finalizing without modification our approach of using a supplemental cost reporting form to report the costs associated with establishing and maintaining a buffer stock, subject to the Paperwork Reduction Act Review of the supplemental cost reporting form itself.
Comment: Commenters were divided on CMS's proposed approach to payment under this policy for buffer stocks of essential medicines in shortage. As stated in the preamble of the proposed rule, CMS would not pay for newly established buffer stocks of essential medicines that are listed as “Currently in Shortage” on the FDA's Drug Shortage Database. However, CMS would continue to pay for buffer stocks of essential medicines that had already been established under this policy prior to the medicine being listed as “Currently in Shortage,” even if those buffer stocks were drawn down to less than 6 months in size. While some commenters were supportive of these provisions in the proposed rule, some commenters stated that continuing to pay for any amount of a buffer stock after a drug is listed as “Currently in Shortage” incentivizes unnecessary retention of stock and potential for hoarding. Commenters stated that this incentive may adversely affect patient care, as hospitals may withhold medicines from patient care to maintain their 6-month stockpile of a given essential medicine.
Regarding our request for comments on the duration of time that CMS should continue to pay for a buffer stock of an essential medicine after that medicine is listed as “Currently in Shortage,” several commenters stated that we should not limit the amount of time that CMS will continue to pay for the buffer stock. Several of these commenters stated that not applying a limit would be consistent with pharmaceutical purchasing and may promote resiliency in the pharmaceutical supply chain. Other commenters stated that CMS should consider paying for essential medicines that enter shortage on a pro-rated basis. A commenter recommended that we limit payments to 6 months after ( print page 69397) the drug has entered shortage. A commenter requested that we clarify if a hospital will continue to be eligible for the separate payment for a drug that has entered shortage even if the hospital does not draw its buffer stock down below 6 months in size.
Response: We thank the commenters for their suggestions. We acknowledge the concerns of commenters regarding incentives for hospitals to stockpile a medicine during a shortage and thus potentially exacerbate that shortage. To reiterate, the intent of this buffer stock policy is to encourage hospitals to preventatively establish a buffer stock prior to a shortage occurring and then, in the event of a shortage, to draw down the buffer stock by administering needed drugs to patients. Further, similar to our earlier response to concerns raised more regarding the potential to induce or exacerbate shortages of essential medicines under this policy, we continue to believe that the pool of hospitals eligible for separate payment under this policy is sufficiently small, and has sufficiently less purchasing power than larger hospitals and hospital chains, that the ability of these hospitals to stockpile during a shortage is limited, and even if it were possible for them to stockpile, their ability to significantly exacerbate a shortage is limited. Finally, we believe it is unlikely that a small, independent hospital would withhold essential medicines from patient care during a shortage to maintain a 6-month buffer stock. As a practical matter, we expect that small, independent hospitals may be more likely to be in a position where they would need to draw down their buffer stock below a 6-month supply during a shortage because these hospitals may lack sufficient purchasing power to readily obtain these drugs, as compared to larger hospitals and hospitals that are part of chains. Taking these factors into account, we agree with commenters who supported continuing to separately pay for the reasonable costs of maintaining an already established buffer stock after a drug enters shortage as an appropriate approach, even if the number of months of supply of that medicine in the buffer stock drops to less than 6 months during the shortage. For the same reasons, we also agree with commenters who indicated that CMS should not limit the amount of time that it will continue to pay for the reasonable costs of maintaining the buffer stock after an essential medicine is listed as “Currently in Shortage.” We believe that hospitals that voluntary establish and maintain a buffer stock of essential medicines may continue to incur additional, reasonable costs for the maintenance of that buffer stock during a shortage, even if the size of the buffer stocks drops below 6 months. Accordingly, we believe that these hospitals should continue to be able to receive separate payment for the IPPS shares of these additional costs for the duration of the shortage. However, as we gain additional experience under the policy, we may consider adjusting the amount of time that hospitals may continue to receive separate payment for maintaining buffer stocks of essential medicines that are subsequently listed as “Currently in Shortage.” After consideration of the comments received, we are finalizing as proposed to continue to separately pay for maintaining an already established buffer stock after a drug enters shortage, even if the number of months of supply of that medicine in the buffer stock drops to less than 6 months during the shortage. We note that larger hospitals and hospitals that are part of chains may have a greater ability to avoid drawing down their buffer stocks during a shortage, though they may also face some challenges. If we were to expand hospital eligibility in the future, we may revisit this aspect of the policy for these hospitals.
Comment: While most commenters were supportive of not permitting hospitals to newly establish buffer stocks for medicines in shortage, some commenters stated that permitting hospitals to establish buffer stocks of drugs regardless of shortage status may contribute to stability in pharmaceutical purchasing in a manner similar to continuing to pay for buffer stocks after medicines enter shortage. Commenters also noted that regulatory flexibility exists for other entities, such as compounding pharmacies, to produce drugs that are listed as “Currently in Shortage” on the FDA's Drug Shortage Database. These commenters stated that, given the small pool of eligible hospitals, permitting hospitals to continue to establish buffer stocks of essential medicines in shortage would be unlikely to markedly exacerbate shortages.
Response: We disagree with commenters who suggested that a hospital that failed to establish a buffer stock before a drug entered shortage be allowed to receive separate payment for subsequently establishing such a buffer stock during the shortage. As we stated in the proposed rule and continue to believe, the appropriate time to establish a buffer stock for a drug is before it goes into shortage or after a shortage period has ended, but not during a shortage. As a general matter, this policy was developed to support hospitals in establishing a buffer stock before a drug enters shortage, so that medicines remain available to patients while the shortage is in effect. Given that small, independent hospitals are less likely to be able to establish a buffer stock after a drug enters shortage, or as robust of a buffer stock even taking into account the potentially limited ability of a small, independent hospital to avail itself of compounding, [ 249 ] not establishing a buffer stock of these medicines in advance of the shortage would generally mean that those drugs are not as available to their patients. Therefore, we continue to believe that the separate payment should be available only where the buffer stock is established prior to an essential medicine entering shortage.
After consideration of the comments received, we are finalizing as proposed to not separately pay for a buffer stock newly established after a drug goes into shortage.
Comment: Many commenters supported the use of ASPR's “ARMI” list of essential medicines developed in 2022. Commenters stated that regular (for example, annual) review of this eligible medicines list, in consultation with stakeholders or under an established public-private partnership, would be crucial to identifying other essential medicines and providing updates. A few commenters suggested expanding participation requirements, while narrowing payment-eligible medicines to better ensure the most needed buffer inventories are developed and maintained by the most appropriate type of facility. Other commenters proposed other lists, such as the Executive Order (E.O.) 13944 List of Essential Medicines, Medical Countermeasures and Critical Inputs List developed in 2020 under the E.O. by the U.S. Food and Drug Administration (hereafter referred to as the E.O. 13944 List), the World Health Organization's Essential Medicines List, American Society of Health-System Pharmacists Drug Shortages List, U.S. Pharmacopeia's Medicine Supply Map, and Vizient's Essential Medications For High-Quality Patient Care. Some commenters stated the need to include certain products that are not included in the ARMI list (for example, oncology drugs; blood and blood products) and ( print page 69398) thus stated the E.O. 13944 List might better serve this proposal's interests and, according to such commenters, is the most recognized among healthcare providers. Others asserted that ASPR's ARMI List was limited, left out critical medicines, should be harmonized with the E.O. 13944 List, and included many medicines for which it is impractical for eligible hospitals to establish a buffer stock. Some commenters recommended the creation of a separate list for the outpatient setting, including outpatient cancer care, physicians' offices, and infusion centers.
Several commenters proposed the gradual expansion of eligible medicines that could be considered essential and provide care to unique patient populations that were otherwise not included. A few commenters also recommended that essential medical devices be included. Others suggested expanded medicines including chemotherapy and other cancer treatment drugs. A commenter suggested excluding immune globulin because these products share the unique supply chain of the excluded fractionated plasma products.
Response: We appreciate the commenters' feedback and diverse clinical perspectives on defining an appropriate and effective list of essential medicines. As we discussed in the proposed rule, the ARMI List is a prioritized subset of 86 essential medicines from the E.O. 13944 List that are either critical for minimum patient care in acute settings or important for acute care with no comparable alternatives available. The medicines included in the ARMI List were considered, by consensus, to be most critically needed for typical acute patient care. In this context, acute patient care was defined as: rescue use or lifesaving use or both (that is, Intensive Care Units, Cardiac/Coronary Care Units, and Emergency Departments), stabilizing patients in hospital continued care to enable discharge, and urgent or emergency surgery. Development of the ARMI List focused on assessing the clinical criticality and supply chains of small molecules and therapeutic biologics. The development of the ARMI List was informed by expert input and perspectives from multiple key pharmaceutical supply chain stakeholders (material suppliers, pharmaceutical manufacturers, group purchasing organizations, wholesale distributors) and clinical stakeholders (doctors, nurses, pharmacists, and public health experts representing major hospital systems, professional societies, and government agencies serving underrepresented populations). This involved conducting and analyzing data from more than 80 surveys, conducting more than 40 interviews, holding 4 workshops that combined clinical and industry expertise, and consulting more than 100 sources to clarify inputs from interview, surveys, and workshops. The ARMI List was also informed by public feedback on the E.O. 13944 List provided during a public comment period starting in October 2020
Further, while the E.O. 13944 List includes blood and blood products, this policy is not intended to include medicines that would be used for longer-term chronic management, including those needed to cure a condition through weeks or months of outpatient treatment in the outpatient setting or chronic care (for example, oncology).
Based on the comprehensive assessment and process followed to develop the ARMI List, as well as the inclusion of a variety of inputs and perspectives across the pharmaceutical supply chains—from industry to clinical community and the public at large—we believe that use of the ARMI List to identify essential medicines for purposes of this policy is appropriate to promote supply chain resilience. at this juncture. After consideration of the comments received, we are finalizing as proposed our use of the ARMI List.
Comment: Commenters expressed concerns regarding the use of the FDA's Drug Shortage Database as a means of establishing the shortage status of a given essential medicine. Specifically, commenters stated that the list is not sensitive to regional shortages, such that it is possible that hospitals may have to draw down their buffer stock(s) below 6 months in size for a regional shortage, despite the medicine not being listed as “Currently in Shortage” on the FDA's Drug Shortage Database. Commenters also stated that the FDA's Drug Shortage Database tends to only capture the most extreme of shortages and may not be sensitive to other supply challenges that hospitals face. Commenters further stated that the FDA's Drug Shortage List tends to lag alternative sources of drug shortage status, such as the American Society of Health-System Pharmacists' (ASHP's) Drug Shortages List. For these reasons, commenters recommended that CMS consider modifying its proposal to adopt the ASHP Drug Shortages List as its source for determining shortage status of a given essential medicine.
Commenters requested clarification on whether all formulations of a drug will be removed from eligibility if a common Active Pharmaceutical Ingredient (API) enters shortage.
Response: We thank commenters for their feedback regarding our use of the FDA's Drug Shortage Database. We recognize that the purpose, audience, scope, source of information, methodology and timing for determining shortage status differs between the FDA's Drug Shortage Database and the ASHP's Drug Shortages List. These differences are also documented by researchers, ASHP, and others, and were reviewed by CMS in developing this policy. [ 250 251 252 253 ]
As described on the FDA's Drug Shortage Database website, [ 254 ] the FDA Drug Shortage Database is maintained by a dedicated team within the agency and manufacturers are required to report drug shortages to the FDA. FDA defines a shortage as a period of time when the demand for a drug in the United States exceeds supply. FDA's definition considers the entire United States market supply from all manufacturers combined based on manufacturer reporting of their inventory and production for the potentially medically necessary use(s) at the patient level. FDA receives information from manufacturers about their ability to supply the market and uses this information to track shortages at the national level. Manufacturers provide FDA most drug shortage information, and FDA works closely with them to prevent or reduce the impact of shortages. When a shortage is listed as current on the FDA Drug Shortage Database, FDA is aware of the supply situation and works on efforts to mitigate the supply disruption. FDA also works with manufacturers on shortage prevention efforts for drugs not yet listed on the Drug Shortage Database. [ 255 ]
By contrast, CMS understands the American Society of Health-System Pharmacists (ASHP) list defines a shortage as a supply issue that affects how a pharmacy prepares or dispenses a drug product, and would post a shortage if one manufacturer was out of stock even if the other manufacturers are able to cover the supply gap. This often leads to more drugs being declared in `shortage' by ASHP when compared ( print page 69399) to FDA's definition of a shortage. For these reasons, we believe that the FDA's Drug Shortage Database is the most appropriate source for determining the shortage status of our eligible essential medicines for purposes of this policy.
As discussed previously, in conjunction with this final policy, CMS will conduct provider outreach on a quarterly basis regarding essential medicines that are in shortage. We intend to make it clear to hospitals on or about the start of each calendar year quarter which drugs are or are not in shortage for the purposes of eligibility for separate payment for the costs of establishing or maintaining their respective buffer stocks. As discussed, we believe this provider outreach will help to mitigate concerns regarding the administrative burden on hospitals of tracking when a drug is considered in shortage under our policy.
After consideration of the public comments received, we are finalizing as proposed our use of the FDA Drug Shortages Database as the basis for determining when an essential medicine is in shortage.
Comment: Some commenters noted that some of the 86 essential medicines eligible under our policy are controlled substances. These commenters asked that CMS work with the Drug Enforcement Agency (DEA) to ensure that hospitals are able to adequately establish and maintain buffer stocks of these medicines under the policy.
Response: All applicable DEA requirements with respect to any controlled substances that are essential medicines are unaltered by our policy and continue to apply. Changes to any DEA requirements are outside of the scope of this rulemaking. As we gain additional experience under the policy we may consider unique aspects, if any, of its applicability to controlled substances in future rulemaking.
Comment: Some commenters also requested that CMS delay the effective date beyond October 1, 2024, to allow manufacturers to ramp up production.
Response: As noted in our earlier responses, we continue to believe that our pool of eligible hospitals is sufficiently small, and that these hospitals have sufficiently less purchasing power than larger hospitals and hospital chains, such that the policy would not create demand shocks that would cause or exacerbate shortages. As such, we do not believe there is a need to delay the policy to permit manufacturers to increase production. We also note that the policy is entirely voluntary on the part of eligible hospitals and does not permit separate payment for newly establishing buffer stocks for drugs that are already in shortage.
After consideration of the public comments received, we are finalizing as proposed the effective date of October 1, 2024.
Comment: Some commenters requested we clarify if Medicare Advantage costs will be included as eligible costs for establishing the Medicare share of hospital costs.
Response: The separate payment for establishing and maintaining access to essential medicines under this policy is for the costs that are currently bundled into the IPPS payments. Those IPPS payments do not include Medicare Advantage payments. Therefore, the Medicare inpatient share of costs under this policy appropriately does not include Medicare Advantage.
Comment: Commenters requested that we clarify if eligible hospitals will be permitted to establish a shared buffer stock, or if each hospital will have to separately establish and maintain their respective buffer stock(s).
Response: Eligible hospitals that elect to maintain a shared buffer stock of essential medicines with other eligible hospitals may receive separate payment for establishing and maintaining the shared buffer stock only if all of the requirements for payment under this policy are met independently by each hospital (for example, there is sufficient buffer stock that each hospital has access to a 6-month supply for itself if all the hospitals begin to access the buffer stock at the same time in the event of a shortage), and the costs associated with establishing and maintaining the shared buffer stock are reasonably allocated to each hospital without duplication of those costs (for example, the total costs reported to Medicare—in accordance with the principles of reasonable cost as set forth in section 1861(v)(1)(A) of the Act and in 42 CFR 413.9 —across the hospitals for establishing and maintaining that shared buffer stock must equal the total costs of establishing and maintaining that buffer stock). If one or more of the buffer stock(s) of essential medicines that comprise the established shared buffer stock are subsequently listed as “Currently in Shortage” on the FDA's Drug Shortage Database, the buffer stock(s) of those essential medicines in shortage may remain eligible for separate payment under this policy for the duration of the shortage. Eligibility for separate payment for essential medicines that are “Currently in Shortage” will be maintained consistent with the manner in which individual buffer stocks of essential medicines remain eligible for payment after being listed as “Currently in Shortage,” as described previously.
Comment: A commenter asked if internal compounding of an essential medicine in shortage will be permitted to build up a buffer stock of an essential medicine.
Response: The appropriate clinical use of compounding and all applicable regulations and requirements associated with compounding are beyond the scope of this rulemaking. We remind hospitals, however, that the costs of establishing and maintaining a buffer stock of an essential medicine do not include the cost of the essential medicine itself, meaning that the cost of compounding would not be included in the cost for establishing and maintaining a buffer stock of an essential medicine.
Comment: A commenter requested that we use Average Daily Census (ADC) data for the beginning of the cost reporting period to determine a given hospital's bed count.
Response: We proposed to use the hospital bed count as established in accordance with § 412.105(b), which is consistent with how bed count is established for other IPPS payment purposes. We do not see the need to establish an alternative methodology for determining hospital bed count specific to this policy. We are finalizing this aspect of the policy as proposed.
Comment: A commenter requested that we clarify if CMS will provide an Explanation of Benefits with specific codes relevant to the payment adjustment.
Response: There is no additional payment required of a beneficiary who, during their IPPS inpatient stay, receives an essential medicine from a hospital that receives separate payment for establishing and maintaining a buffer stock of that essential medicine under the IPPS. Information on which hospitals receive separate payment under the policy will be publicly available as part of the cost report information reported by hospitals.
Comment: We received a number of comments requesting broader policy actions. Many commenters stated that addressing drug shortages will require actions beyond the Medicare program, including actions directed at pharmaceutical suppliers. Commenters asked that we seek legislative authority to make additional payments, including any potential expansion to the outpatient setting, in a non-budget neutral manner. Several commenters requested that we convene a technical workgroup to consult on the policy, with both federal and private-sector members. A commenter requested that ( print page 69400) we require drug manufacturers to equitably disburse medicines to smaller hospitals, as these smaller hospitals often face difficulties in purchasing medicines. Commenters requested that we require drug manufacturers to produce more stock above and beyond their purchase demand, or that we directly pay distributors, manufacturers, or wholesalers to hold a national buffer supply for disbursement to hospitals. Some commenters requested that we establish measures to prevent hospitals participating in this policy from contracting with manufacturers that have outstanding pharmaceutical quality issues at their facilities. A commenter requested that we shift to stockpiling Active Pharmaceutical Ingredients (API) instead of Finished Drug Form (FDF) pharmaceuticals. Commenters requested that we direct Medicare Advantage, Medicaid Managed Care Organizations, and Children's Health Insurance Program Plans to release guidance waiving prior authorization for suitable alternatives to drugs in shortage.
Response: We thank commenters for their feedback regarding broader policy actions to address drug shortages and supply chain resiliency, which we note are beyond the scope of this rulemaking.
After consideration of the public comments we received, we are finalizing our policy as proposed. In summary, for cost reporting periods beginning on or after October 1, 2024, we are establishing a separate payment under the IPPS to small, independent hospitals for the additional resource costs involved in voluntarily establishing and maintaining access to 6-month buffer stocks of essential medicines, either directly or through contractual arrangements with a manufacturer, distributor, or intermediary. The costs of buffer stocks that are eligible for separate payment are the costs of buffer stocks for one or more of the medicines on ARMI's List of 86 essential medicines. The separate payment will be for the IPPS share of the additional costs and could be issued in a lump sum, or as biweekly payments to be reconciled at cost report settlement. The separate payment will not apply to buffer stocks of any of the essential medicines on the ARMI List that are listed as “Currently in Shortage” on the FDA Drug Shortages Database, as communicated to hospitals by the MACs on a quarterly basis, unless a hospital had already established and was maintaining a 6-month buffer stock of that medicine prior to the shortage. Once an essential medicine is no longer in shortage, as communicated by the MACs for the calendar quarter, our policy does not differentiate that essential medicine from other essential medicines, and hospitals would be eligible to establish and maintain buffer stocks for the medicine as they would have before the shortage. We are also finalizing our proposal to codify this payment adjustment in the regulations by adding new paragraph (g) to 42 CFR 412.113 , as well as our proposed conforming changes to 42 CFR 412.1(a) and 412.2(f) , without modification. In future years as we gain additional experience under this policy, we plan to assess the program's impact and consider revisions.
Section 3025 of the Patient Protection and Affordable Care Act, as amended by section 10309 of the Patient Protection and Affordable Care Act, added section 1886(q) to the Act, which establishes the Hospital Readmissions Reduction Program effective for discharges from applicable hospitals beginning on or after October 1, 2012. Under the Hospital Readmissions Reduction Program, payments to applicable hospitals may be reduced to account for certain excess readmissions. We refer readers to the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49530 through 49543 ) and the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38221 through 38240 ) for a general overview of the Hospital Readmissions Reduction Program. We also refer readers to 42 CFR 412.152 through 412.154 for codified Hospital Readmissions Reduction Program requirements. Additionally, we refer readers to the CY 2025 OPPS/ASC proposed rule where we are soliciting input on potential future methodological modifications regarding the Safety of Care measure group within the Overall Hospital Quality Star Rating ( 89 FR 59509 through 59515 ).
There were no proposals or updates in the FY 2025 IPPS/LTCH PPS proposed rule for the Hospital Readmissions Reduction Program ( 89 FR 36238 ). We refer readers to section I.G.7. of Appendix A of the final rule for an updated estimate of the financial impact of using the proportion of dually eligible beneficiaries, ERRs, and aggregate payments for each condition/procedure and all discharges for applicable hospitals from the FY 2025 Hospital Readmissions Reduction Program applicable period (that is, July 1, 2020, through June 30, 2023).
For background on the Hospital VBP Program, we refer readers to the CMS website at: https://www.cms.gov/medicare/quality/initiatives/hospital-quality-initiative/hospital-value-based-purchasing . We also refer readers to our codified requirements for the Hospital VBP Program at 42 CFR 412.160 through 412.168 . Additionally, we refer readers to the CY 2025 OPPS/ASC proposed rule where we are soliciting input on potential future methodological modifications regarding the Safety of Care measure group within the Overall Hospital Quality Star Rating ( 89 FR59509 through 59515 ).
Under section 1886(o)(7)(C)(v) of the Act, the applicable percent for the FY 2025 program year is 2.00 percent. Using the methodology we adopted in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53571 through 53573 ), we estimate that the total amount available for value-based incentive payments for FY 2025 is approximately $1.67 billion, based on the March 2024 update of the FY 2023 MedPAR file.
As finalized in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53573 through 53576 ), we will utilize a linear exchange function to translate this estimated amount available into a value-based incentive payment percentage for each hospital, based on its Total Performance Score (TPS). We published proxy value-based incentive payment adjustment factors in Table 16 associated with the FY 2025 IPPS/LTCH PPS proposed rule (which is available via the internet on the CMS website). We are publishing updated proxy value-based incentive payment adjustment factors in Table 16A associated with this final rule (which is available via the CMS website) to reflect changes based on the March 2024 update to the FY 2023 MedPAR file. We note that these updated proxy adjustment factors will not be used to adjust hospital payments. These updated proxy adjustment factors were calculated using the historical baseline and performance periods for the FY 2024 Hospital VBP Program. These updated proxy adjustment factors were calculated using the March 2024 update to the FY 2023 MedPAR file. The slope of the linear exchange function used to calculate these proxy factors ( print page 69401) was 4.7264532378, and the estimated amount available for value-based incentive payments to hospitals for FY 2025 is approximately $1.67 billion. We will add a new table, Table 16B to display the actual value-based incentive payment adjustment factors, exchange function slope, and estimated amount available for the FY 2025 Hospital VBP Program. We expect that Table 16B will be posted on the CMS website in the fall of 2024.
We refer readers to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49110 through 49111 ) for summaries of previously adopted measures for the FY 2025 and FY 2026 program years and to the FY 2024 IPPS/LTCH PPS final rule for summaries of previously adopted measures beginning with the FY 2026 program year ( 88 FR 59081 through 59083 ). We did not propose any new measure adoptions or removals to the measure set in the FY 2025 IPPS/LTCH PPS proposed rule. Table V.L.-01 summarizes the previously adopted Hospital VBP Program measure set for the FY-2025 program year.

As discussed in section IX.B.2.g(2) of this final rule, we are finalizing the adoption of the updates to the HCAHPS Survey measure beginning with the FY 2030 program year. We are also finalizing the adoption of the updates to the HCAHPS Survey measure in the Hospital Inpatient Quality Reporting (IQR) Program, beginning with the FY 2027 program year, as described in section IX.B.2.e. of this final rule. Additionally, we are finalizing the modification to the Hospital VBP Program's scoring of the HCAHPS Survey measure for the FY 2027 through FY 2029 program years to score hospitals on only those dimensions of the survey that will remain unchanged from the current version, as described in section IX.B.2.f. of this final rule. Lastly, ( print page 69402) we are also finalizing the modification to the Hospital VBP Program's scoring of the HCAHPS Survey measure beginning in FY 2030 to account for the adoption of the modifications to the survey, which will result in a total of nine survey dimensions for the updated HCAHPS Survey measure in the Hospital VBP Program, as described in section IX.B.2.g(3) of this final rule. Table V.L.-02 summarizes the previously adopted Hospital VBP Program measures for the FY 2026 through FY 2030 program years.

We refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59084 through 59087 ) for previously adopted baseline and performance periods for the FY 2025 through FY 2029 program years. We also refer readers to the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56998 ) in which we finalized a schedule for all future baseline and performance periods for all measures.
Tables V.L.-03, V.L.-04, V.L.-05, V.L.-06, and V.L.-07 summarize the baseline and performance periods that we have previously adopted.

We refer readers to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49115 through 49118 ) for previously established performance standards for the FY 2025 program year. We also refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59089 through 59090 ) for the previously established performance standards for the FY 2026 program year. We refer readers to the FY 2021 IPPS/LTCH PPS final rule for further discussion on performance standards for which the measures are calculated with lower values representing better performance ( 85 FR 58855 ). ( print page 69406)
We have adopted certain measures for the Safety domain, Clinical Outcomes domain, and the Efficiency and Cost Reduction domain for future program years to ensure that we can adopt baseline and performance periods of sufficient length for performance scoring purposes. In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45294 through 45295 ), we established performance standards for the FY 2027 program year for the Clinical Outcomes domain measures (MORT-30-AMI, MORT-30-HF, MORT-30-PN (updated cohort), MORT-30-COPD, MORT-30- CABG, and COMP-HIP-KNEE) and the Efficiency and Cost Reduction domain measure (MSPB). We note that the performance standards for the MSPB Hospital measure are based on performance period data. Therefore, we are unable to provide numerical equivalents for the standards at this time. The previously established and newly estimated performance standards for the FY 2027 program year are set out in Tables V.L.-08 and V.L.-09.

As discussed in section IX.B.2.f. of this final rule, we are finalizing a modification to the scoring of the HCAHPS Survey for the FY 2027 through FY 2029 program years while the updates to the survey are adopted and publicly reported under the Hospital IQR Program. Scoring will be modified to only score hospitals on the six Hospital VBP Program dimensions of the HCAHPS Survey that will remain unchanged from the current version. These six dimensions of the HCAHPS Survey for the Hospital VBP Program will be:
- “Communication with Nurses,”
- “Communication with Doctors,”
- “Communication about Medicines,”
- “Discharge Information,”
- “Cleanliness and Quietness,” and
- “Overall Rating.” ( print page 69407)
We are finalizing the proposal to exclude the “Responsiveness of Hospital Staff” and “Care Transition” dimensions from scoring in the Hospital VBP Program's HCAHPS Survey measure in the Person and Community Engagement domain for the FY 2027 through FY 2029 program years. This will allow hospitals to be scored on only those dimensions of the survey in the Hospital VBP Program that will remain unchanged from the current version of the survey while the updated HCAHPS Survey is publicly reported on under the Hospital IQR Program for one year as required by statute. We are also finalizing the proposal to adopt the updated version of the HCAHPS Survey measure for use in the Hospital VBP Program beginning in FY 2030 as outlined in section IX.B.2.g. of this final rule.
Scoring will be modified such that for each of the six dimensions listed previously, Achievement Points (0-10 points) and Improvement Points (0-9 points) will be calculated, the larger of which will be summed across these six dimensions to create a pre-normalized HCAHPS Base Score of 0-60 points (as compared to 0-80 points with the current eight dimensions). The pre-normalized HCAHPS Base Score will then be multiplied by 8/6 (1.3333333) and rounded according to standard rules (values of 0.5 and higher are rounded up, values below 0.5 are rounded down) to create the normalized HCAHPS Base Score. Each of the six dimensions will be of equal weight, so that, as currently scored, the normalized HCAHPS Base Score will range from 0 to 80 points. HCAHPS Consistency Points will be calculated in the same manner as the current method and will continue to range from 0 to 20 points. Like the Base Score, the Consistency Points Score will consider scores across the six unchanged dimensions of the Person and Community Engagement domain. The final element of the scoring formula, which will remain unchanged from the current formula, will be the sum of the HCAHPS Base Score and the HCAHPS Consistency Points Score for a total score that ranges from 0 to 100 points. The method for calculating the performance standards for the six dimensions will remain unchanged. We refer readers to the Hospital Inpatient VBP Program final rule ( 76 FR 26511 through 26513 ) for our methodology for calculating performance standards. The estimated performance standards for the six dimensions that are finalized to be scored on for the FY 2027 program year are set out in Table V.L.-09.

We invited public comment on this proposal in the FY 2025 IPPS/LTCH PPS proposed rule and have summarized all comments and responses in section IX.B.2. of this final rule.
We have adopted certain measures for the Safety domain, Clinical Outcomes domain, and the Efficiency and Cost Reduction domain for future program years to ensure that we can adopt baseline and performance periods of sufficient length for performance scoring purposes. In the FY 2023 IPPS/LTCH PPS final rule ( 86 FR 49118 ), we established performance standards for the FY 2028 program year for the Clinical Outcomes domain measures (MORT-30-AMI, MORT-30-HF, MORT-30-PN (updated cohort), MORT-30-COPD, MORT-30-CABG, and COMP-HIP-KNEE) and the Efficiency and Cost Reduction domain measure (MSPB Hospital). We note that the performance standards for the MSPB Hospital measure are based on performance period data. Therefore, we are unable to provide numerical equivalents for the standards at this time. The previously established performance standards for these measures are set out in Table V.L.-10.

We have adopted certain measures for the Safety domain, Clinical Outcomes domain, and the Efficiency and Cost Reduction domain for future program years to ensure that we can adopt baseline and performance periods of sufficient length for performance scoring purposes. In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59091 through 59092 ), we established performance standards for the FY 2029 program year for the Clinical Outcomes domain measures (MORT-30-AMI, MORT-30-HF, MORT-30-PN (updated cohort), MORT-30-COPD, MORT-30- CABG, and COMP-HIP-KNEE) and the Efficiency and Cost Reduction domain measure (MSPB Hospital). We note that the performance standards for the MSPB Hospital measure are based on performance period data. Therefore, we are unable to provide numerical equivalents for the standards at this time. The previously established performance standards for these measures are set out in Table V.L.-11.

As discussed previously, we have adopted certain measures for the Clinical Outcomes domain (MORT-30- AMI, MORT-30-HF, MORT-30-PN (updated cohort), MORT-30-COPD, MORT-30-CABG, and COMP-HIP- KNEE) and the Efficiency and Cost Reduction domain (MSPB Hospital) for future program years to ensure that we can adopt baseline and performance periods of sufficient length for performance scoring purposes. In accordance with our methodology for calculating performance standards discussed more fully in the Hospital Inpatient VBP Program final rule ( 76 FR 26511 through 26513 ), which is codified at 42 CFR 412.160 , we are establishing the following performance standards for the FY 2030 program year for the Clinical Outcomes domain and the Efficiency and Cost Reduction domain. We note that the performance standards for the MSPB Hospital measure are based on performance period data. Therefore, we are unable to provide numerical equivalents for the standards at this time. The newly established performance standards for these measures are set out in Table V.L.-12.

We refer readers to the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50707 through 50709 ) for a general overview of the HAC Reduction Program and a detailed discussion of the statutory basis for the Program. We also refer readers to 42 CFR 412.170 through 412.172 for codified HAC Reduction Program requirements. Additionally, we refer readers to the CY 2025 OPPS/ASC proposed rule where we are soliciting input on potential future methodological modifications regarding the Safety of Care measure group within the Overall Hospital Quality Star Rating ( 89 FR 59509 through 59515 ).
The previously finalized measures for the HAC Reduction Program are shown in table V.M.-01. Technical specifications for the CMS PSI 90 measure can be found on the QualityNet website available at: https://qualitynet.cms.gov/inpatient/measures/psi/resources . Technical specifications for the CDC National Healthcare Safety Network (NHSN) healthcare-associated infection (HAI) measures can be found at the CDC's NHSN website at http://www.cdc.gov/nhsn/acute-care-hospital/index.html and on the QualityNet website available at: https://qualitynet.cms.gov/inpatient/measures/hai/resources . These web pages provide measure updates and other information necessary to guide hospitals participating in the collection of HAC Reduction Program data.

We did not make any proposals or policy updates for the HAC Reduction Program in the FY 2025 IPPS/LTCH PPS proposed rule. We refer readers to section I.G.9. of Appendix A of this final rule for an updated estimate of the impact of the Program policies on the proportion of hospitals in the worst performing quartile of the Total HAC Scores for the FY 2025 HAC Reduction Program.
While we did not make any proposals or policy updates to the HAC Reduction Program, we did receive comments from interested parties.
Comment: Many commenters provided recommendations for program improvements and potential future measures, including the Hospital Harm—Falls with Injury electronic clinical quality measure (eCQM), a hospital-onset COVID-19 measure, the NHSN Hospital-Onset Bacteremia and Fungemia Outcome Measure, and endoscope-associated infection eCQMs. Many commenters recommended including a hospital-acquired COVID-19 measure within the HAC Reduction Program to incentivize facilities to adopt mitigation approaches and prevent the transmission of COVID-19 in healthcare settings. Many commenters recommended that hospital-onset COVID should be defined as infections diagnosed after 5+ days of admission. Many commenters recommended providing financial support to hospitals for hospital-onset COVID-19 reporting efforts. Many commenters also recommended timely and accurate public reporting of hospital-onset COVID-19 data, aggregated by state and facility name, to aid patients in making informed decisions on where to receive care. Many commenters recommended incentivizing healthcare settings to implement preventative measures to reduce COVID-19 transmission, including requiring universal mask wearing, universal screening testing, and improved air quality. Many commenters expressed concern about COVID-19 as a health equity issue disproportionately impacting populations at higher risk, including people with disabilities, populations that have been historically marginalized, and communities that are under-resourced; and recommended aggregating the data by demographics, socio-economic status, and disability status.
Response: We thank the commenters for these recommendations on potential future measures to include in the HAC Reduction Program and will consider them for future program years.
The Rural Community Hospital Demonstration was originally authorized by section 410A of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) ( Pub. L. 108-173 ). The demonstration has been extended three times since the original 5-year period mandated by the MMA, each time for an additional 5 years. These extensions were authorized by sections 3123 and 10313 of the Affordable Care Act ( Pub. L. 111-148 ), section 15003 of the 21st Century Cures Act ( Pub. L. 114-255 ) (Cures Act) enacted in 2016, and most recently, by section 128 of the Consolidated Appropriations Act of 2021 ( Pub. L. 116-260 ). In this final rule, we summarize the status of the demonstration program, and the current methodologies for implementation and calculating budget neutrality.
We are also finalizing the amount to be applied to the national IPPS payment rates to account for the costs of the demonstration in FY 2025, incorporating the reconciled amount of demonstration costs for FY 2019 into the total offset the national IPPS payment rates for FY 2025.
Section 410A(a) of Public Law 108-173 required the Secretary to establish a demonstration program to test the feasibility and advisability of establishing rural community hospitals to furnish covered inpatient hospital services to Medicare beneficiaries. The demonstration pays rural community hospitals under a reasonable cost-based methodology for Medicare payment purposes for covered inpatient hospital services furnished to Medicare beneficiaries. A rural community hospital, as defined in section 410A(f)(1) of Public Law 108-173 , is a hospital that—
- Is located in a rural area (as defined in section 1886(d)(2)(D) of the Act) or is treated as being located in a rural area under section 1886(d)(8)(E) of the Act;
- Has fewer than 51 beds (excluding beds in a distinct part psychiatric or ( print page 69412) rehabilitation unit) as reported in its most recent cost report;
- Provides 24-hour emergency care services; and
- Is not designated or eligible for designation as a CAH under section 1820 of the Act.
Our policy for implementing the 5-year extension period authorized by Public Law 116-260 (the Consolidated Appropriations Act of 2021) follows upon the previous extensions under the ACA ( Pub. L. 111-148 ) and the Cures Act (Pub. L.114-255). Section 410A of Public Law 108-173 (MMA) initially required a 5-year period of performance. Subsequently, sections 3123 and 10313 of Public Law 111-148 required the Secretary to conduct the demonstration program for an additional 5-year period, to begin on the date immediately following the last day of the initial 5-year period. In addition, Public Law 111-148 limited the number of hospitals participating to no more than 30. Section 15003 of the Cures Act required a 10-year extension period in place of the 5-year extension period under the ACA, thereby extending the demonstration for another 5 years. Section 128 of Public Law 116-260 , in turn, revised the statute to indicate a 15-year extension period, instead of the 10-year extension period mandated by the Public Law 114-159 (Cures Act). Please refer to the FY 2023 IPPS proposed and final rules ( 87 FR 28454 through 28458 and 87 FR 49138 through 49142 , respectively) for an account of hospitals entering and withdrawing from the demonstration with these re-authorizations. There are currently 22 hospitals participating in the demonstration.
Section 410A(c)(2) of Public Law 108-173 requires that, in conducting the demonstration program under this section, the Secretary shall ensure that the aggregate payments made by the Secretary do not exceed the amount that the Secretary would have paid if the demonstration program under this section was not implemented. This requirement is commonly referred to as “budget neutrality.” Generally, when we implement a demonstration program on a budget neutral basis, the demonstration program is budget neutral on its own terms; in other words, the aggregate payments to the participating hospitals do not exceed the amount that would be paid to those same hospitals in the absence of the demonstration program. We note that the payment methodology for this demonstration, that is, cost-based payments to participating small rural hospitals, makes it unlikely that increased Medicare outlays will produce an offsetting reduction to Medicare expenditures elsewhere. Therefore, in the IPPS final rules spanning the period from FY 2005 through FY 2016, we adjusted the national inpatient PPS rates by an amount sufficient to account for the added costs of this demonstration program, thus applying budget neutrality across the payment system as a whole rather than merely across the participants in the demonstration program. (We applied a different methodology for FY 2017, with the demonstration expected to end prior to the Cures Act extension). As we discussed in the FYs 2005 through 2017 IPPS/LTCH PPS final rules ( 69 FR 49183 ; 70 FR 47462 ; 71 FR 48100 ; 72 FR 47392 ; 73 FR 48670 ; 74 FR 43922 , 75 FR 50343 , 76 FR 51698 , 77 FR 53449 , 78 FR 50740 , 77 FR 50145 ; 80 FR 49585 ; and 81 FR 57034 , respectively), we believe that the statutory language of the budget neutrality requirements permits the agency to implement the budget neutrality provision in this manner.
We resumed this methodology of offsetting demonstration costs against the national payment rates in the IPPS final rules from FY 2018 through FY 2024. Please see the FY 2024 IPPS final rule for an account of how we applied the budget neutrality requirement for these fiscal years ( 88 FR 59114 through 59116 ).
We have generally incorporated two components into the budget neutrality offset amounts identified in the final IPPS rules in previous years. First, we have estimated the costs of the demonstration for the upcoming fiscal year, generally determined from historical, “as submitted” cost reports for the hospitals participating in that year. Update factors representing nationwide trends in cost and volume increases have been incorporated into these estimates, as specified in the methodology described in the final rule for each fiscal year. Second, as finalized cost reports became available, we determined the amount by which the actual costs of the demonstration for an earlier, given year differed from the estimated costs for the demonstration set forth in the final IPPS rule for the corresponding fiscal year, and incorporated that amount into the budget neutrality offset amount for the upcoming fiscal year. If the actual costs for the demonstration for the earlier fiscal year exceeded the estimated costs of the demonstration identified in the final rule for that year, this difference was added to the estimated costs of the demonstration for the upcoming fiscal year when determining the budget neutrality adjustment for the upcoming fiscal year. Conversely, if the estimated costs of the demonstration set forth in the final rule for a prior fiscal year exceeded the actual costs of the demonstration for that year, this difference was subtracted from the estimated cost of the demonstration for the upcoming fiscal year when determining the budget neutrality adjustment for the upcoming fiscal year.
We note that we have calculated this difference for FYs 2005 through 2018 between the actual costs of the demonstration as determined from finalized cost reports once available, and estimated costs of the demonstration as identified in the applicable IPPS final rules for these years.
For the most-recently enacted extension period, under the Consolidated Appropriations Act of 2021, we have continued upon the general budget neutrality methodology used in previous years, as described above in the citations to earlier IPPS final rules. Based on the methodology outlined in the FY 2025 proposed rule, we are finalizing the calculation of the offset amount to be applied to the national IPPS payment rates for FY 2025.
Consistent with the general methodology from previous years, we are estimating the costs of the demonstration for the upcoming fiscal year and incorporating this estimate into the budget neutrality offset amount to be applied to the national IPPS rates for the upcoming fiscal year, that is, FY 2025. We are conducting this estimate for FY 2025 based on the 22 currently participating hospitals. The methodology for calculating this amount for FY 2025 proceeds according to the following steps:
Step 1: For each of these 22 hospitals, we identify the reasonable cost amount calculated under the reasonable cost-based methodology for covered inpatient hospital services, including swing beds, as indicated on the “as submitted” cost report for the most recent cost reporting period available. ( print page 69413) For each of these hospitals, the “as submitted” cost report is that with cost report period end date in CY 2022. We sum these hospital-specific amounts to arrive at a total general amount representing the costs for covered inpatient hospital services, including swing beds, across the total 22 hospitals eligible to participate during FY 2025.
Then, we multiply this amount by the FYs 2023, 2024, and 2025 IPPS market basket percentage increases, which are calculated by the CMS Office of the Actuary. (We are using the market basket percentage increase for FY 2025, which can be found at section II.B. of the preamble of this final rule). The result for the 22 hospitals is the general estimated reasonable cost amount for covered inpatient hospital services for FY 2025.
Consistent with our methods in previous years for formulating this estimate, we are applying the IPPS market basket percentage increases for FYs 2023 through 2025 to the applicable estimated reasonable cost amount (previously described) in order to model the estimated FY 2025 reasonable cost amount under the demonstration. We believe that the IPPS market basket percentage increases appropriately indicate the trend of increase in inpatient hospital operating costs under the reasonable cost methodology for the years involved.
Step 2: For each of the participating hospitals, we identify the estimated amount that would otherwise be paid in FY 2025 under applicable Medicare payment methodologies for covered inpatient hospital services, including swing beds (as indicated on the same set of “as submitted” cost reports as in Step 1), if the demonstration were not implemented. We sum these hospital-specific amounts, and, in turn, multiply this sum by the FYs 2023, 2024, and 2025 IPPS applicable percentage increases. (For FY 2025, we are using the applicable percentage increase, per section V.B. of the preamble of this final rule). This methodology differs from Step 1, in which we apply the market basket percentage increases to the hospitals' applicable estimated reasonable cost amount for covered inpatient hospital services. We believe that the IPPS applicable percentage increases are appropriate factors to update the estimated amounts that generally would otherwise be paid without the demonstration. This is because IPPS payments constitute the largest part of the payments that would otherwise be made without the demonstration and the applicable percentage increase is the factor used under the IPPS to update the inpatient hospital payment rates.
Step 3: We subtract the amount derived in Step 2 from the amount derived in Step 1. According to our methodology, the resulting amount indicates the total difference for the 22 hospitals (for covered inpatient hospital services, including swing beds), which will be the general estimated amount of the costs of the demonstration for FY 2025.
For this final rule, the resulting amount is $49,914,526, to be incorporated into the budget neutrality offset adjustment for FY 2025. This estimated amount is based on the specific assumptions regarding the data sources used, that is, recently available “as submitted” cost reports and revised historical update factors for cost and payment for the FY 2025 IPPS final rule.
As described earlier, we have calculated the difference for FYs 2005 through 2018 between the actual costs of the demonstration, as determined from finalized cost reports once available, and estimated costs of the demonstration as identified in the applicable IPPS final rules for these years.
At this time, for the FY 2025 final rule, all of the finalized cost reports are available for the 27 hospitals that completed cost report periods beginning in FY 2019 under the demonstration payment methodology. We have determined the actual costs of the demonstration for FY 2019 based on these cost reports to be $40,429,606. (We note that the Medicare Administrative Contractors (MACs) have corrected the calculation of cost amounts under the demonstration for several of the participating hospitals, and that, although the MACs have not issued the final revised cost reports, we have included these revised cost amounts for these specific hospitals in our determination of the total cost amount for FY 2019).
The estimated amount for the demonstration costs for FY 2019 that was incorporated into the finalized budget neutrality offset amount in the 2019 IPPS final rule was $70,929,313. ( 83 FR 41504 ). Therefore, the actual costs of the demonstration for FY 2019 as determined from finalized cost reports fell short of this estimated amount by $30,499,707. In keeping with previous policy, we are subtracting the amount of this difference for the prior year (that is, $30,499,707) for FY 2019) from the estimated amount of the costs of the demonstration for the upcoming fiscal year, (that is, $49,914,526 for FY 2025) in determining the total budget neutrality offset amount for FY 2025
Therefore, for this FY 2025 IPPS/LTCH PPS final rule, the final budget neutrality offset amount for FY 2025 is the difference between: (1) $49,914,526, which is the amount determined under section II.A.4.h. of the Addendum of this final rule, representing the difference applicable to FY 2025 between the sum of the estimated reasonable cost amounts that would be paid under the demonstration for covered inpatient services to the 22 hospitals eligible to participate in the fiscal year and the sum of the estimated amounts that would generally be paid if the demonstration had not been implemented; and (2) $30,499,707, which is the difference between the estimated costs for the demonstration for FY 2019, which was incorporated into the finalized budget neutrality offset amount for 2019, and the actual costs of the demonstration for FY 2019, determined from finalized cost reports for the 27 hospitals that participated in FY 2019. This difference between (1) and (2) is $19,414,819, representing the budget neutrality offset amount to be applied to the national IPPS payment rates for FY 2025.
Comment: The parent company for two of the participating hospitals expressed support for the continuation of the of the Rural Community Hospital Demonstration program, while noting that it does not offer long-term financial stability needed to maintain health care access in rural areas. The commenter requests that the demonstration be made a permanent program, and, in addition, that CMS institute an application process to ensure the demonstration meets program capacity. Furthermore, the commenter requests several technical adjustments to the administration of the demonstration that may enhance stability in the payment to the participating hospitals.
Response: We appreciate the comments. We have conducted the demonstration program in accordance with Congressional mandates. Title XVIII does not extend authority to make the demonstration a permanent program. With regard to any further actions, we intend to work with the commenter and other rural stakeholders to examine the issues involved. ( print page 69414)
Section 1886(g) of the Act requires the Secretary to pay for the capital-related costs of inpatient acute hospital services in accordance with a prospective payment system established by the Secretary. Under the statute, the Secretary has broad authority in establishing and implementing the IPPS for acute care hospital inpatient capital-related costs. We initially implemented the IPPS for capital-related costs in the FY 1992 IPPS final rule ( 56 FR 43358 ). In that final rule, we established a 10-year transition period to change the payment methodology for Medicare hospital inpatient capital-related costs from a reasonable cost-based payment methodology to a prospective payment methodology (based fully on the Federal rate).
FY 2001 was the last year of the 10-year transition period that was established to phase in the IPPS for hospital inpatient capital-related costs. For cost reporting periods beginning in FY 2002, capital IPPS payments are based solely on the Federal rate for almost all acute care hospitals (other than hospitals receiving certain exception payments and certain new hospitals). (We refer readers to the FY 2002 IPPS final rule ( 66 FR 39910 through 39914 ) for additional information on the methodology used to determine capital IPPS payments to hospitals both during and after the transition period.)
The basic methodology for determining capital prospective payments using the Federal rate is set forth in the regulations at 42 CFR 412.312 . For the purpose of calculating capital payments for each discharge, the standard Federal rate is adjusted as follows:
(Standard Federal Rate) × (DRG Weight) × (Geographic Adjustment Factor (GAF) × (COLA for hospitals located in Alaska and Hawaii) × (1 + Capital DSH Adjustment Factor + Capital IME Adjustment Factor, if applicable).
In addition, under § 412.312(c), hospitals also may receive outlier payments under the capital IPPS for extraordinarily high-cost cases that qualify under the thresholds established for each fiscal year.
The regulations at 42 CFR 412.348 provide for certain exception payments under the capital IPPS. The regular exception payments provided under § 412.348(b) through (e) were available only during the 10-year transition period. For a certain period after the transition period, eligible hospitals may have received additional payments under the special exceptions provisions at § 412.348(g). However, FY 2012 was the final year hospitals could receive special exceptions payments. For additional details regarding these exceptions policies, we refer readers to the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51725 ).
Under § 412.348(f), a hospital may request an additional payment if the hospital incurs unanticipated capital expenditures in excess of $5 million due to extraordinary circumstances beyond the hospital's control. Additional information on the exception payment for extraordinary circumstances in § 412.348(f) can be found in the FY 2005 IPPS final rule ( 69 FR 49185 and 49186 ).
Under the capital IPPS, the regulations at 42 CFR 412.300(b) define a new hospital as a hospital that has operated (under previous or current ownership) for less than 2 years and lists examples of hospitals that are not considered new hospitals. In accordance with § 412.304(c)(2), under the capital IPPS, a new hospital is paid 85 percent of its allowable Medicare inpatient hospital capital related costs through its first 2 years of operation, unless the new hospital elects to receive full prospective payment based on 100 percent of the Federal rate. We refer readers to the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51725 ) for additional information on payments to new hospitals under the capital IPPS.
In the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57061 ), we revised the regulations at 42 CFR 412.374 relating to the calculation of capital IPPS payments to hospitals located in Puerto Rico beginning in FY 2017 to parallel the change in the statutory calculation of operating IPPS payments to hospitals located in Puerto Rico, for discharges occurring on or after January 1, 2016, made by section 601 of the Consolidated Appropriations Act, 2016 ( Pub. L. 114-113 ). Section 601 of Public Law 114-113 increased the applicable Federal percentage of the operating IPPS payment for hospitals located in Puerto Rico from 75 percent to 100 percent and decreased the applicable Puerto Rico percentage of the operating IPPS payments for hospitals located in Puerto Rico from 25 percent to zero percent, applicable to discharges occurring on or after January 1, 2016. As such, under revised § 412.374, for discharges occurring on or after October 1, 2016, capital IPPS payments to hospitals located in Puerto Rico are based on 100 percent of the capital Federal rate.
The annual update to the national capital Federal rate, as provided for in 42 CFR 412.308(c) , for FY 2025 is discussed in section III. of the Addendum to this FY 2025 IPPS/LTCH PPS final rule.
Comment: A commenter encouraged CMS to expand capital DSH eligibility to geographically rural hospitals. The commenter believes this would bolster the rural health care safety net. The commenter cited negative capital margins at geographically rural hospitals, low occupancy rates in geographically rural hospitals, as well as recent closure of geographically rural hospitals as reasons why expanding capital DSH eligibility to geographically rural hospitals would be justified.
Response: We believe this comment is out of scope of this rulemaking. We thank the commenter for this suggestion and may consider it in future rulemaking. We note that the capital DSH payment adjustments were finalized in the FY 1992 IPPS final rule ( 56 FR 43377 through 43379 ) based on a cost regression analysis.
Comment: A commenter stated that the cost of capital improvements required to reduce the spread of airborne infections should be included under the capital IPPS.
Response: We appreciate the commenter's interest in capital project investments related to airborne infections. The regulations on capital-related costs can be found in subpart G of part 413 of Title 42 of the CFR. In general, we believe these regulations do not preclude such costs as being considered allowable capital-related costs. As described previously, the statute requires capital-related costs of inpatient acute hospital services be paid under a prospective payment system established by the Secretary. The basic methodology for determining prospective payments under the capital IPPS is set forth in the regulations at 42 CFR 412.312 .
Certain hospitals excluded from a prospective payment system, including children's hospitals, 11 cancer ( print page 69415) hospitals, and hospitals located outside the 50 States, the District of Columbia, and Puerto Rico (that is, hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa) receive payment for inpatient hospital services they furnish on the basis of reasonable costs, subject to a rate-of-increase ceiling. A per discharge limit (the target amount, as defined in § 413.40(a) of the regulations) is set for each hospital based on the hospital's own cost experience in its base year, and updated annually by a rate-of-increase percentage. For each cost reporting period, the updated target amount is multiplied by total Medicare discharges during that period and applied as an aggregate upper limit (the ceiling as defined in § 413.40(a)) of Medicare reimbursement for total inpatient operating costs for a hospital's cost reporting period. In accordance with § 403.752(a) of the regulations, religious nonmedical health care institutions (RNHCIs) also are subject to the rate-of-increase limits established under § 413.40 of the regulations discussed previously. Furthermore, in accordance with § 412.526(c)(3) of the regulations, extended neoplastic disease care hospitals also are subject to the rate-of-increase limits established under § 413.40 of the regulations discussed previously.
As explained in the FY 2006 IPPS final rule ( 70 FR 47396 through 47398 ), beginning with FY 2006, we have used the percentage increase in the IPPS operating market basket to update the target amounts for children's hospitals, the 11 cancer hospitals, and RNHCIs.
Consistent with the regulations at §§ 412.23(g) and 413.40(a)(2)(ii)(A) and (c)(3)(viii), we also have used the percentage increase in the IPPS operating market basket to update target amounts for short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa. In the FY 2018 IPPS/LTCH PPS final rule, we rebased and revised the IPPS operating market basket to a 2014 base year, effective for FY 2018 and subsequent fiscal years ( 82 FR 38158 through 38175 ), and finalized the use of the percentage increase in the 2014-based IPPS operating market basket to update the target amounts for children's hospitals, the 11 cancer hospitals, RNHCIs, and short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa for FY 2018 and subsequent fiscal years. As discussed in section IV. of the preamble of the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45194 through 45207 ), we rebased and revised the IPPS operating market basket to a 2018 base year. Therefore, we used the percentage increase in the 2018-based IPPS operating market basket to update the target amounts for children's hospitals, the 11 cancer hospitals, RNHCIs, and short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa for FY 2022 and subsequent fiscal years.
For the FY 2025 IPPS/LTCH PPS proposed rule, based on IGI's 2023 fourth quarter forecast, we estimated that the 2018-based IPPS operating market basket percentage increase for FY 2025 would be 3.0 percent (that is, the estimate of the market basket rate-of-increase). Based on this estimate, the FY 2025 rate-of-increase percentage that we proposed to apply to the FY 2024 target amounts in order to calculate the FY 2025 target amounts for children's hospitals, the 11 cancer hospitals, RNCHIs, and short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa was 3.0 percent, in accordance with the applicable regulations at 42 CFR 413.40 . However, we proposed that if more recent data became available for the FY 2025 IPPS/LTCH PPS final rule, we would use such data, if appropriate, to calculate the final IPPS operating market basket update for FY 2025. Based on more recent data available (IGI's second quarter 2024 forecast), we estimate that the 2018-based IPPS operating market basket percentage increase for FY 2025 is 3.4 percent (that is, the estimate of the market basket rate-of-increase). Based on this estimate, the FY 2025 rate-of-increase percentage that we will apply to the FY 2024 target amounts in order to calculate the FY 2025 target amounts for children's hospitals, the 11 cancer hospitals, RNCHIs, and short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa is 3.4 percent, in accordance with the applicable regulations at 42 CFR 413.40 .
In addition, payment for inpatient operating costs for hospitals classified under section 1886(d)(1)(B)(vi) of the Act (which we refer to as “extended neoplastic disease care hospitals”) for cost reporting periods beginning on or after January 1, 2015, is to be made as described in 42 CFR 412.526(c)(3) , and payment for capital costs for these hospitals is to be made as described in 42 CFR 412.526(c)(4) . (For additional information on these payment regulations, we refer readers to the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38321 through 38322 ).) Section 412.526(c)(3) provides that the hospital's Medicare allowable net inpatient operating costs for that period are paid on a reasonable cost basis, subject to that hospital's ceiling, as determined under § 412.526(c)(1), for that period. Under § 412.526(c)(1), for each cost reporting period, the ceiling was determined by multiplying the updated target amount, as defined in § 412.526(c)(2), for that period by the number of Medicare discharges paid during that period. Section 412.526(c)(2)(i) describes the method for determining the target amount for cost reporting periods beginning during FY 2015. Section 412.526(c)(2)(ii) specifies that, for cost reporting periods beginning during fiscal years after FY 2015, the target amount will equal the hospital's target amount for the previous cost reporting period updated by the applicable annual rate-of-increase percentage specified in § 413.40(c)(3) for the subject cost reporting period ( 79 FR 50197 ).
For FY 2025, in accordance with §§ 412.22(i) and 412.526(c)(2)(ii) of the regulations, for cost reporting periods beginning during FY 2025, the proposed update to the target amount for extended neoplastic disease care hospitals (that is, hospitals described under § 412.22(i)) is the applicable annual rate-of-increase percentage specified in § 413.40(c)(3), which was estimated to be the percentage increase in the 2018-based IPPS operating market basket (that is, the estimate of the market basket rate-of-increase). Accordingly, the proposed update to an extended neoplastic disease care hospital's target amount for FY 2025 was 3.0 percent, which was based on IGI's fourth quarter 2023 forecast. Furthermore, we proposed that if more recent data became available for the FY 2025 IPPS/LTCH PPS final rule, we would use such data, if appropriate, to calculate the IPPS operating market basket rate of increase for FY 2025. Based on more recent data available (IGI's second quarter 2024 forecast), we estimate that the 2018-based IPPS operating market basket percentage increase for FY 2025 is 3.4 percent (that is, the estimate of the market basket rate-of-increase). Accordingly, the FY 2025 rate-of-increase percentage that we will apply to the FY 2024 target amounts in order to calculate the FY 2025 target amounts to an extended neoplastic disease care hospital is 3.4 percent, which is based on IGI's second quarter 2024 forecast.
We received no comments on this proposal and therefore are finalizing ( print page 69416) this provision without modification. Incorporating more recent data available for this final rule, as we proposed, we are adopting a 3.4 percent update for FY 2025.
Section 4419(b) of Public Law 105-33 requires the Secretary to publish annually in the Federal Register a report describing the total amount of adjustment payments made to excluded hospitals and hospital units by reason of section 1886(b)(4) of the Act during the previous fiscal year.
The process of requesting, reviewing, and awarding an adjustment payment is likely to occur over a 2-year period or longer. First, generally, an excluded hospital must file its cost report for the fiscal year in accordance with § 413.24(f)(2) of the regulations. The MAC reviews the cost report and issues a notice of provider reimbursement (NPR). Once the hospital receives the NPR, if its operating costs are in excess of the ceiling, the hospital may file a request for an adjustment payment. After the MAC receives the hospital's request in accordance with applicable regulations, the MAC or CMS, depending on the type of adjustment requested, reviews the request and determines if an adjustment payment is warranted. This determination is sometimes not made until more than 180 days after the date the request is filed because there are times when the request applications are incomplete and additional information must be requested in order to have a completed request application. However, in an attempt to provide interested parties with data on the most recent adjustment payments for which we have data, we are publishing data on adjustment payments that were processed by the MAC or CMS during FY 2023.
The table that follows includes the most recent data available from the MACs and CMS on adjustment payments that were adjudicated during FY 2023. As indicated previously, the adjustments made during FY 2023 only pertain to cost reporting periods ending in years prior to FY 2023. Total adjustment payments made to IPPS-excluded hospitals during FY 2023 are $98,720,259.00. The table depicts for each class of hospitals, in the aggregate, the number of adjustment requests adjudicated, the excess operating costs over the ceiling, and the amount of the adjustment payments.

Section 1820 of the Act provides for the establishment of Medicare Rural Hospital Flexibility Programs (MRHFPs), under which individual States may designate certain facilities as critical access hospitals (CAHs). Facilities that are so designated and meet the CAH conditions of participation under 42 CFR part 485, subpart F , will be certified as CAHs by CMS. Regulations governing payments to CAHs for services to Medicare beneficiaries are located in 42 CFR part 413 .
The Frontier Community Health Integration Project Demonstration was originally authorized by section 123 of the Medicare Improvements for Patients and Providers Act of 2008 ( Pub. L. 110-275 ). The demonstration has been extended by section 129 of the Consolidated Appropriations Act, 2021 ( Pub. L. 116-260 ) for an additional 5 years. In this final rule, we are summarizing the status of the demonstration program, and the ongoing methodologies for implementation and budget neutrality for the demonstration extension period.
As discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59119 through 59122 ), section 123 of the Medicare Improvements for Patients and Providers Act of 2008, as amended by section 3126 of the Affordable Care Act, authorized a demonstration project to allow eligible entities to develop and test new models for the delivery of health care services in eligible counties to improve access to and better integrate the delivery of acute care, extended care and other health care services to Medicare beneficiaries. The demonstration was titled “Demonstration Project on Community Health Integration Models in Certain Rural Counties,” and commonly known as the Frontier Community Health Integration Project (FCHIP) Demonstration.
The authorizing statute stated the eligibility criteria for entities to be able to participate in the demonstration. An eligible entity, as defined in section 123(d)(1)(B) of Public Law 110-275 , as amended, is a Medicare Rural Hospital Flexibility Program (MRHFP) grantee under section 1820(g) of the Act (that is, a CAH); and is located in a state in which at least 65 percent of the counties in the state are counties that have 6 or less residents per square mile.
The authorizing statute stipulated several other requirements for the demonstration. In addition, section 123(g)(1)(B) of Public Law 110-275 required that the demonstration be budget neutral. Specifically, this provision stated that, in conducting the demonstration project, the Secretary shall ensure that the aggregate payments made by the Secretary do not exceed the amount which the Secretary estimates would have been paid if the demonstration project under the section were not implemented. Furthermore, section 123(i) of Public Law 110-275 stated that the Secretary may waive such requirements of titles XVIII and XIX of the Act as may be necessary and appropriate for the purpose of carrying out the demonstration project, thus allowing the waiver of Medicare payment rules encompassed in the demonstration. CMS selected CAHs to participate in four interventions, under which specific waivers of Medicare payment rules would allow for enhanced payment for telehealth, skilled nursing facility/nursing facility beds, ambulance services, and home health services. These waivers were formulated with the goal of increasing access to care with no net increase in costs. ( print page 69417)
Section 123 of Public Law 110-275 initially required a 3-year period of performance. The FCHIP Demonstration began on August 1, 2016, and concluded on July 31, 2019 (referred to in this section of the final rule as the “initial period”). Subsequently, section 129 of the Consolidated Appropriations Act, 2021 ( Pub. L. 116-260 ) extended the demonstration by 5 years (referred to in this section of the final rule as the “extension period”). The Secretary is required to conduct the demonstration for an additional 5-year period. CAHs participating in the demonstration project during the extension period began such participation in their cost reporting year that began on or after January 1, 2022.
As described in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59119 through 59122 ), 10 CAHs were selected for participation in the demonstration initial period. The selected CAHs were located in three states—Montana, Nevada, and North Dakota—and participated in three of the four interventions identified in the FY 2024 IPPS/LTCH PPS final rule. Each CAH was allowed to participate in more than one of the interventions. None of the selected CAHs were participants in the home health intervention, which was the fourth intervention.
In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45323 through 45328 ), CMS concluded that the initial period of the FCHIP Demonstration (covering the performance period of August 1, 2016, to July 31, 2019) had satisfied the budget neutrality requirement described in section 123(g)(1)(B) of Pub L. 110-275 . Therefore, CMS did not apply a budget neutrality payment offset policy for the initial period of the demonstration.
Section 129 of Public Law 116-260 , stipulates that only the 10 CAHs that participated in the initial period of the FCHIP Demonstration are eligible to participate during the extension period. Among the eligible CAHs, five have elected to participate in the extension period. The selected CAHs are located in two states—Montana and North Dakota—and are implementing three of the four interventions. The eligible CAH participants elected to change the number of interventions and payment waivers they would participate in during the extension period. CMS accepted and approved the CAHs intervention and payment waiver updates. For the extension period, five CAHs are participants in the telehealth intervention, three CAHs are participants in the skilled nursing facility/nursing facility bed intervention, and three CAHs are participants in the ambulance services intervention. As with the initial period, each CAH was allowed to participate in more than one of the interventions during the extension period. None of the selected CAHs are participants in the home health intervention, which was the fourth intervention.
As described in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59119 through 59122 ), CMS waived certain Medicare rules for CAHs participating in the demonstration initial period to allow for alternative reasonable cost-based payment methods in the three distinct intervention service areas: telehealth services, ambulance services, and skilled nursing facility/nursing facility (SNF/NF) beds expansion. The payments and payment waiver provisions only apply if the CAH is a participant in the associated intervention. CMS Intervention Payment and Payment Waivers for the demonstration extension period consist of the following:
CMS waives section 1834(m)(2)(B) of the Act, which specifies the facility fee to the originating site for Medicare telehealth services. CMS modifies the facility fee payment specified under section 1834(m)(2)(B) of the Act to make reasonable cost-based reimbursement to the participating CAH where the participating CAH serves as the originating site for a telehealth service furnished to an eligible telehealth individual, as defined in section 1834(m)(4)(B) of the Act. CMS reimburses the participating CAH serving as the originating site at 101 percent of its reasonable costs for overhead, salaries and fringe benefits associated with telehealth services at the participating CAH. CMS does not fund or provide reimbursement to the participating CAH for the purchase of new telehealth equipment.
CMS waives section 1834(m)(2)(A) of the Act, which specifies that the payment for a telehealth service furnished by a distant site practitioner is the same as it would be if the service had been furnished in-person. CMS modifies the payment amount specified for telehealth services under section 1834(m)(2)(A) of the Act to make reasonable cost-based reimbursement to the participating CAH for telehealth services furnished by a physician or practitioner located at distant site that is a participating CAH that is billing for the physician or practitioner professional services. Whether the participating CAH has or has not elected Optional Payment Method II for outpatient services, CMS would pay the participating CAH 101 percent of reasonable costs for telehealth services when a physician or practitioner has reassigned their billing rights to the participating CAH and furnishes telehealth services from the participating CAH as a distant site practitioner. This means that participating CAHs that are billing under the Standard Method on behalf of employees who are physicians or practitioners (as defined in section 1834(m)(4)(D) and (E) of the Act, respectively) would be eligible to bill for distant site telehealth services furnished by these physicians and practitioners. Additionally, CAHs billing under the Optional Method would be reimbursed based on 101 percent of reasonable costs, rather than paid based on the Medicare physician fee schedule, for the distant site telehealth services furnished by physicians and practitioners who have reassigned their billing rights to the CAH. For distant site telehealth services furnished by physicians or practitioners who have not reassigned billing rights to a participating CAH, payment to the distant site physician or practitioner would continue to be made as usual under the Medicare physician fee schedule. Except as described herein, CMS does not waive any other provisions of section 1834(m) of the Act for purposes of the telehealth services intervention payments, including the scope of Medicare telehealth services as established under section 1834(m)(4)(F) of the Act.
CMS waives 42 CFR 413.70(b)(5)(i)(D) and section 1834(l)(8) of the Act, which provides that payment for ambulance services furnished by a CAH, or an entity owned and operated by a CAH, is 101 percent of the reasonable costs of the CAH or the entity in furnishing the ambulance services, but only if the CAH or the entity is the only provider or supplier of ambulance services located within a 35-mile drive of the CAH, excluding ambulance providers or suppliers that are not legally authorized to furnish ambulance services to transport individuals to or from the CAH. The participating CAH would be paid 101 percent of reasonable costs for its ambulance services regardless of whether there is any provider or supplier of ambulance services located within a 35-mile drive of the participating CAH or participating CAH-owned and operated entity. CMS would ( print page 69418) not make cost-based payment to the participating CAH for any new capital (for example, vehicles) associated with ambulance services. This waiver does not modify any other Medicare rules regarding or affecting the provision of ambulance services.
CMS waives 42 CFR 485.620(a) , 42 CFR 485.645(a)(2) , and section 1820(c)(2)(B)(iii) of the Act which limit CAHs to maintaining no more than 25 inpatient beds, including beds available for acute inpatient or swing bed services. CMS waives 1820(f) of the Act permitting designating or certifying a facility as a critical access hospital for which the facility at any time is furnishing inpatient beds which exceed more than 25 beds. Under this waiver, if the participating CAH has received swing bed approval from CMS, the participating CAH may maintain up to ten additional beds (for a total of 35 beds) available for acute inpatient or swing bed services; however, the participating CAH may only use these 10 additional beds for nursing facility or skilled nursing facility level of care. CMS would pay the participating CAH 101 percent of reasonable costs for its SNF/NF services furnished in the 10 additional beds.
In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45323 through 45328 ), we finalized a policy to address the budget neutrality requirement for the demonstration initial period. As explained in the FY 2022 IPPS/LTCH PPS final rule, we based our selection of CAHs for participation in the demonstration with the goal of maintaining the budget neutrality of the demonstration on its own terms meaning that the demonstration would produce savings from reduced transfers and admissions to other health care providers, offsetting any increase in Medicare payments as a result of the demonstration. However, because of the small size of the demonstration and uncertainty associated with the projected Medicare utilization and costs, the policy we finalized for the demonstration initial period of performance in the FY 2022 IPPS/LTCH PPS final rule provides a contingency plan to ensure that the budget neutrality requirement in section 123 of Public Law 110-275 is met.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49144 through 49147 ), we adopted the same budget neutrality policy contingency plan used during the demonstration initial period to ensure that the budget neutrality requirement in section 123 of Public Law 110-275 is met during the demonstration extension period. If analysis of claims data for Medicare beneficiaries receiving services at each of the participating CAHs, as well as from other data sources, including cost reports for the participating CAHs, shows that increases in Medicare payments under the demonstration during the 5-year extension period are not sufficiently offset by reductions elsewhere, we would recoup the additional expenditures attributable to the demonstration through a reduction in payments to all CAHs nationwide.
As explained in the FY 2023 IPPS/LTCH PPS final rule, because of the small scale of the demonstration, we indicated that we did not believe it would be feasible to implement budget neutrality for the demonstration extension period by reducing payments to only the participating CAHs. Therefore, in the event that this demonstration extension period is found to result in aggregate payments in excess of the amount that would have been paid if this demonstration extension period were not implemented, CMS policy is to comply with the budget neutrality requirement finalized in the FY 2023 IPPS/LTCH PPS final rule, by reducing payments to all CAHs, not just those participating in the demonstration extension period.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49144 through 49147 ), we stated that we believe it is appropriate to make any payment reductions across all CAHs because the FCHIP Demonstration was specifically designed to test innovations that affect delivery of services by the CAH provider category. We explained our belief that the language of the statutory budget neutrality requirement at section 123(g)(1)(B) of Public Law 110-275 permits the agency to implement the budget neutrality provision in this manner. The statutory language merely refers to ensuring that aggregate payments made by the Secretary do not exceed the amount which the Secretary estimates would have been paid if the demonstration project was not implemented and does not identify the range across which aggregate payments must be held equal.
In the FY 2023 IPPS/LTCH PPS final rule, we finalized a policy that in the event the demonstration extension period is found not to have been budget neutral, any excess costs would be recouped within one fiscal year. We explained our belief that this policy is a more efficient timeframe for the government to conclude the demonstration operational requirements (such as analyzing claims data, cost report data or other data sources) to adjudicate the budget neutrality payment recoupment process due to any excess cost that occurred as result of the demonstration extension period.
As explained in the FY 2022 IPPS/LTCH PPS final rule, we finalized a policy to address the demonstration budget neutrality methodology and analytical approach for the initial period of the demonstration. In the FY 2023 IPPS/LTCH PPS final rule, we finalized a policy to adopt the budget neutrality methodology and analytical approach used during the demonstration initial period to ensure budget neutrality for the extension period. The analysis of budget neutrality during the initial period of the demonstration identified both the costs related to providing the intervention services under the FCHIP Demonstration and any potential downstream effects of the intervention-related services, including any savings that may have accrued.
The budget neutrality analytical approach for the demonstration initial period incorporated two major data components: (1) Medicare cost reports; and (2) Medicare administrative claims. As described in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45323 through 45328 ), CMS computed the cost of the demonstration for each fiscal year of the demonstration initial period using Medicare cost reports for the participating CAHs, and Medicare administrative claims and enrollment data for beneficiaries who received demonstration intervention services.
In addition, to capture the full impact of the interventions, CMS developed a statistical modeling, Difference-in-Difference (DiD) regression analysis to estimate demonstration expenditures and compute the impact of expenditures on the intervention services by comparing cost data for the demonstration and non-demonstration groups using Medicare administrative claims across the demonstration period of performance under the initial period of the demonstration. The DiD regression analysis would compare the direct cost and potential downstream effects of intervention services, including any savings that may have accrued, during the baseline and performance period for both the demonstration and comparison groups.
Second, the Medicare administrative claims analysis would be reconciled ( print page 69419) using data obtained from auditing the participating CAHs' Medicare cost reports. We would estimate the costs of the demonstration using “as submitted” cost reports for each hospital's financial fiscal year participation within each of the demonstration extension period performance years. Each CAH has its own Medicare cost report end date applicable to the 5-year period of performance for the demonstration extension period. The cost report is structured to gather costs, revenues and statistical data on the provider's financial fiscal period. As a result, we finalized a policy in the FY 2023 IPPS/LTCH PPS final rule that we would determine the final budget neutrality results for the demonstration extension once complete data is available for each CAH for the demonstration extension period.
As stated in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59119 through 59122 ), our policy for implementing the 5-year extension period for section 129 of Public Law 116-260 follows same budget neutrality methodology and analytical approach as the demonstration initial period methodology. While we expect to use the same methodology that was used to assess the budget neutrality of the FCHIP Demonstration during initial period of the demonstration to assess the financial impact of the demonstration during this extension period, upon receiving data for the extension period, we may update and/or modify the FCHIP budget neutrality methodology and analytical approach to ensure that the full impact of the demonstration is appropriately captured.
At this time, for the FY 2025 IPPS/LTCH PPS final rule, while this discussion represents our anticipated approach to assessing the financial impact of the demonstration extension period based on upon receiving data for the full demonstration extension period, we may update and/or modify the FCHIP Demonstration budget neutrality methodology and analytical approach to ensure that the full impact of the demonstration is appropriately captured.
Therefore, we did not propose to apply a budget neutrality payment offset to payments to CAHs in FY 2025. This policy would have no impact for any national payment system for FY 2025. We received no comments on this provision and therefore are finalizing this provision without modification.
Section 123 of the Medicare, Medicaid, and SCHIP (State Children's Health Insurance Program) Balanced Budget Refinement Act of 1999 (BBRA) ( Pub. L. 106-113 ), as amended by section 307(b) of the Medicare, Medicaid, and SCHIP Benefits Improvement and Protection Act of 2000 (BIPA) ( Pub. L. 106-554 ), provides for payment for both the operating and capital- related costs of hospital inpatient stays in long-term care hospitals (LTCHs) under Medicare Part A based on prospectively set rates. The Medicare prospective payment system (PPS) for LTCHs applies to hospitals that are described in section 1886(d)(1)(B)(iv) of the Act, effective for cost reporting periods beginning on or after October 1, 2002.
Section 1886(d)(1)(B)(iv)(I) of the Act originally defined an LTCH as a hospital that has an average inpatient length of stay (as determined by the Secretary) of greater than 25 days.
Section 1886(d)(1)(B)(iv)(II) of the Act also provided an alternative definition of LTCHs (“subclause II” LTCHs). However, section 15008 of the 21st Century Cures Act ( Pub. L. 114-255 ) amended section 1886 of the Act to exclude former “subclause II” LTCHs from being paid under the LTCH PPS and created a new category of IPPS-excluded hospitals, which we refer to as “extended neoplastic disease care hospitals,” to be paid as hospitals that were formally classified as “subclause (II)” LTCHs ( 82 FR 38298 ).
Section 123 of the BBRA requires the PPS for LTCHs to be a “per discharge” system with a diagnosis-related group (DRG) based patient classification system that reflects the differences in patient resource use and costs in LTCHs.
Section 307(b)(1) of the BIPA, among other things, mandates that the Secretary shall examine, and may provide for, adjustments to payments under the LTCH PPS, including adjustments to DRG weights, area wage adjustments, geographic reclassification, outliers, updates, and a disproportionate share adjustment.
In the August 30, 2002, Federal Register ( 67 FR 55954 ), we issued a final rule that implemented the LTCH PPS authorized under the BBRA and BIPA. For the initial implementation of the LTCH PPS (FYs 2003 through 2007), the system used information from LTCH patient records to classify patients into distinct long-term care-diagnosis-related groups (LTCDRGs) based on clinical characteristics and expected resource needs. Beginning in FY 2008, we adopted the Medicare severity-long-term care-diagnosis related groups (MS-LTC-DRGs) as the patient classification system used under the LTCH PPS. Payments are calculated for each MS-LTC-DRG and provisions are made for appropriate payment adjustments. Payment rates under the LTCH PPS are updated annually and published in the Federal Register .
The LTCH PPS replaced the reasonable cost-based payment system under the Tax Equity and Fiscal Responsibility Act of 1982 (TEFRA) (Pub. L. 97248) for payments for inpatient services provided by an LTCH with a cost reporting period beginning on or after October 1, 2002. (The regulations implementing the TEFRA reasonable-cost-based payment provisions are located at 42 CFR part 413 .) With the implementation of the PPS for acute care hospitals authorized by the Social Security Amendments of 1983 (Pub. L. 98-21), which added section 1886(d) to the Act, certain hospitals, including LTCHs, were excluded from the PPS for acute care hospitals and paid their reasonable costs for inpatient services subject to a per discharge limitation or target amount under the TEFRA system. For each cost reporting period, a hospital specific ceiling on payments was determined by multiplying the hospital's updated target amount by the number of total current year Medicare discharges. (Generally, in this section of the preamble of this final rule, when we refer to discharges, we describe Medicare discharges.) The August 30, 2002, final rule further details the payment policy under the TEFRA system ( 67 FR 55954 ).
In the August 30, 2002, final rule, we provided for a 5-year transition period from payments under the TEFRA system to payments under the LTCH PPS. During this 5-year transition period, an LTCH's total payment under the PPS was based on an increasing percentage of the Federal rate with a corresponding decrease in the percentage of the LTCH PPS payment that is based on reasonable cost concepts, unless an LTCH made a one-time election to be paid based on 100 percent of the Federal rate. Beginning with LTCHs' cost reporting periods beginning on or after ( print page 69420) October 1, 2006, total LTCH PPS payments are based on 100 percent of the Federal rate.
In addition, in the August 30, 2002, final rule, we presented an in-depth discussion of the LTCH PPS, including the patient classification system, relative weights, payment rates, additional payments, and the budget neutrality requirements mandated by section 123 of the BBRA. The same final rule that established regulations for the LTCH PPS under 42 CFR part 412, subpart O , also contained LTCH provisions related to covered inpatient services, limitation on charges to beneficiaries, medical review requirements, furnishing of inpatient hospital services directly or under arrangement, and reporting and recordkeeping requirements. We refer readers to the August 30, 2002, final rule for a comprehensive discussion of the research and data that supported the establishment of the LTCH PPS ( 67 FR 55954 ).
In the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49601 through 49623 ), we implemented the provisions of the Pathway for Sustainable Growth Rate (SGR) Reform Act of 2013 ( Pub. L. 113-67 ), which mandated the application of the “site neutral” payment rate under the LTCH PPS for discharges that do not meet the statutory criteria for exclusion beginning in FY 2016. For cost reporting periods beginning on or after October 1, 2015, discharges that do not meet certain statutory criteria for exclusion are paid based on the site neutral payment rate. Discharges that do meet the statutory criteria continue to receive payment based on the LTCH PPS standard Federal payment rate. For more information on the statutory requirements of the Pathway for SGR Reform Act of 2013, we refer readers to the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49601 through 49623 ) and the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57068 through 57075 ).
In the FY 2018 IPPS/LTCH PPS final rule, we implemented several provisions of the 21st Century Cures Act (“the Cures Act”) ( Pub. L. 114-255 ) that affected the LTCH PPS. (For more information on these provisions, we refer readers to ( 82 FR 38299 ).)
In the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41529 ), we made conforming changes to our regulations to implement the provisions of section 51005 of the Bipartisan Budget Act of 2018 ( Pub. L. 115-123 ), which extends the transitional blended payment rate for site neutral payment rate cases for an additional 2 years. We refer readers to section VII.C. of the preamble of the FY 2019 IPPS/LTCH PPS final rule for a discussion of our final policy. In addition, in the FY 2019 IPPS/LTCH PPS final rule, we removed the 25-percent threshold policy under 42 CFR 412.538 , which was a payment adjustment that was applied to payments for Medicare patient LTCH discharges when the number of such patients originating from any single referring hospital was in excess of the applicable threshold for given cost reporting period.
In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42439 ), we further revised our regulations to implement the provisions of the Pathway for SGR Reform Act of 2013 ( Pub. L. 113-67 ) that relate to the payment adjustment for discharges from LTCHs that do not maintain the requisite discharge payment percentage and the process by which such LTCHs may have the payment adjustment discontinued.
Under the regulations at § 412.23(e)(1), to qualify to be paid under the LTCH PPS, a hospital must have a provider agreement with Medicare. Furthermore, § 412.23(e)(2)(i), which implements section 1886(d)(1)(B)(iv) of the Act, requires that a hospital have an average Medicare inpatient length of stay of greater than 25 days to be paid under the LTCH PPS. In accordance with section 1206(a)(3) of the Pathway for SGR Reform Act of 2013 ( Pub. L. 113-67 ), as amended by section 15007 of Public Law 114-255 , we amended our regulations to specify that Medicare Advantage plans' and site neutral payment rate discharges are excluded from the calculation of the average length of stay for all LTCHs, for discharges occurring in cost reporting period beginning on or after October 1, 2015.
As explained more fully previously, LTCHs are required to have an average length of stay (ALOS) of greater than 25 days. Prior to a hospital being classified as an LTCH, the hospital must first participate in Medicare as a hospital (typically a hospital paid under the IPPS) during which time ALOS data is gathered. This data is used to determine whether the hospital has an ALOS of greater than 25 days, which is required to be classified as an LTCH. We generally refer to the period during which a hospital seeks to establish the required ALOS as a “qualifying period.” The qualifying period is the 6-month period immediately preceding the hospital's conversion to an LTCH, and it has been our policy that the requisite ALOS must be demonstrated based on patient data from at least 5 consecutive months of this period. For example, for a hospital seeking to become an LTCH effective January 1, 2025, the qualifying period would be July 1, 2024 through December 31, 2024 (that is, the 6 months immediately preceding the conversion to an LTCH). In order for the hospital to convert to an LTCH, the ALOS must be demonstrated for a period of at least 5 consecutive months (for example, July 1, 2024 through November 30, 2024 or July 15, 2024 to December 14, 2024) of the 6 month qualifying period.
It has been our general policy to allow a hospital to be classified as an LTCH after only the 6-month qualifying period (as opposed to requiring the completion of the more typical 12-month cost reporting period). We have also referred to the ability of a hospital to be classified as an LTCH after a 6-month qualifying period in preamble previously ( 73 FR 29705 ), and the Provider Reimbursement Manual at 3001.4 refers to using data from a 6-month period for hospitals which have not yet filed a cost report. However, our regulations have never explicitly articulated how the qualifying period policy applies to a hospital seeking classification as an LTCH. Therefore, we proposed to revise our regulations at 42 CFR 412.23(e)(4) to explicitly state that a hospital that seeks to be classified as an LTCH may do so after completion of a 6-month qualifying period, provided that the hospital demonstrates an average length of stay (calculated under our existing regulations) of greater than 25 days during at least five consecutive months of the 6-month qualifying period (which is the same timeframe as the “cure period” for existing LTCHs). Specifically, we proposed to add new paragraph § 412.23(e)(4)(iv) to explain the qualifying period for hospitals seeking LTCH classification.
Further, we proposed to revise certain paragraphs and reorder certain paragraphs in § 412.23(e) to improve the clarity of the regulation by clarifying how provisions apply to existing LTCHs and which provisions apply to hospitals seeking classification as an LTCH. First, we proposed to revise paragraph § 412.23(e)(3)(i) to cross-reference new subparagraphs § 412.23(e)(4)(iv) and (e)(4)(v). Second, we proposed to revise paragraph § 412.23(e)(3)(iii) to clarify that it applies in cases of hospitals that have already obtained LTCH classification when the LTCH would not otherwise maintain an average Medicare inpatient length of stay of greater than ( print page 69421) 25 days. Third, we proposed to reserve § 412.23(e)(3)(iv) and move that text to new (e)(4)(v) to clarify that this regulation applies to hospitals seeking new LTCH classification. Fourth, we proposed to revise § 412.23(e)(4) to clarify that the provisions of paragraph (e)(3), with the exception of subparagraphs (e)(3)(iii) and (v), apply to hospitals seeking new LTCH classification. Fifth, we proposed to revise paragraph § 412.23(e)(4)(i) to reflect the addition of new § 412.23(e)(4)(iv) and (e)(4)(v) and clarify existing regulatory language.
We noted that none of these proposed revisions reflect a change to our existing policy; instead, we stated that we believe these revisions will improve the clarity of the regulatory text and better reflect our existing policy.
Comment: Several commenters objected to our use of the word “consecutive” in the proposed regulatory text revisions to codify our existing policy regarding the qualifying period for hospitals seeking to become LTCHs. These commenters believed that the use of the word “consecutive” was both not in accordance with our historical policy and unnecessarily strict. Rather than finalizing the proposed revision, these commenters argued that we should finalize a policy under which, for the qualifying period, hospitals should be able to demonstrate compliance with the ALOS requirement using the average lengths of stay calculated for non-consecutive months.
Response: While we acknowledge the concerns raised by the commenters, we believe that they have misunderstood our proposal. Our reference to “at least five consecutive months” is a reference to one, single, continuous period for which the ALOS would be calculated, just like the ALOS for an existing LTCH is calculated based on the entire cost reporting period, not each individual month therein, and the cure period for an LTCH which falls below the ALOS threshold for a cost reporting period is based on a single, continuous period of at least consecutive five months and not each individual month within that period. Further, we note that our proposed revision to the regulations refers specifically to “ an average length of stay” (emphasis added) and not “average lengths of stay,” which would be how the regulation text for the calculation such as that opposed by commenters would be phrased. Our proposed revisions to the regulation text were not meant to reflect a policy under which the ALOS would be calculated during 5 separate months and the ALOS for each month must be greater than 25 days, as described by some commenters. Our proposed regulatory language was meant to reflect our current policy, under which the ALOS for the entire qualifying period, which must be at least 5 consecutive months, must be greater than 25 days.
However, in considering this comment, we noticed that our existing regulation text at § 412.23(e)(3)(iii) (which describes the “cure period” policy for when an existing LTCH's ALOS does not meet the greater than 25 days threshold for a cost reporting period), § 412.23(e)(4)(iii) (which describes the rules for provider based satellite facilities or remote locations of LTCHs becoming separately participating hospitals), § 412.23(e)(4)(v) (which describes the rules for hospitals seeking to participate in Medicare as LTCHs which experience a change of ownership), as well as our proposed clarification to § 412.23(e)(3)(iii), § 412.23(e)(4)(iii), and § 412.23(e)(4)(v) inadvertently omit the word “consecutive” when describing the time period for the calculation. We believe that this may be the source of commenters' confusion on our proposed regulatory language describing how the calculation is performed for the qualifying period; i.e., that by using the word “consecutive” in the proposed clarification for the qualifying period but not for the cure period, our policy for calculating the ALOS in these situations would not be the same and that the calculation for qualifying periods would be more stringent than our policy for cure periods, provider based facilities becoming separately participating hospitals, and hospitals seeking to participate in Medicare as LTCHs which experience a change of ownership. This was not our intention in making our proposed clarification; rather, our intention was to amend our regulations such that the policy for calculating the ALOS for the qualifying period for a hospital seeking LTCH classification would be consistent with our policy for calculating an existing LTCH's ALOS in other contexts. Therefore, in the interest of making our ALOS regulations as clear as possible, we believe it would be appropriate to make a conforming change to our proposed revisions to § 412.23(e)(3)(iii), § 412.23(e)(4)(iii), and § 412.23(e)(4)(v) to add the word “consecutive.” Additionally, in the case of § 412.23(e)(4)(iii), we are adding “the period of at least” to the regulation, consistent with our language at § 412.23(e)(3)(iii) and § 412.23(e)(4)(iv) and (v). With these additions, the regulation text will be consistent and, we believe, more fully and accurately reflect our current policy. We wish to reassure commenters that, just like the calculation of the ALOS for the qualifying period, this will not change our existing policy for calculating the ALOS for an LTCH's cure period. The cure period calculation at § 412.23(e)(3)(iii) will continue to be based on one, single, continuous period that is at least 5 consecutive months long and there is no requirement for the ALOS in each individual month to be greater than 25 days.
We believe that, consistent with the way the ALOS is calculated for existing LTCHs, whether for cost reporting periods or cure periods, the ALOS for a hospital's qualifying period should be calculated based on one, single, continuous period. For this reason, we believe that the inclusion of the word “consecutive” is both appropriate and necessary in the regulation text and are finalizing our proposed addition of new paragraph § 412.23(e)(4)(iv). Moreover, for consistency with language used in § 412.23(e)(3)(iii) and § 412.23(e)(4)(iii), we are also adding “the period of” to the text of the finalized regulation. Additionally, in the interest of making our regulations consistent with each other and current policy, as discussed previously, we are adding the word “consecutive” to § 412.23(e)(3)(iii) and § 412.23(e)(4)(iii), and § 412.23(e)(4)(v) as well as the words “the period of at least” to § 412.23(e)(4)(iii). We note, as stated previously, these changes do not represent a change from existing policy, and are instead merely a clarification of our existing policy. We will also clarify in our guidance subsequent to this rule that the requirement is that the ALOS for the qualifying period or cure period, as applicable, in its entirety needs to be greater than 25 days, however it is not necessary for the ALOS in each individual month of that period be greater than 25 days.
We received no other comments with respect to our proposed reordering of and revisions to other paragraphs in 412.23 and as such are finalizing those proposals without modification.
Comment: Some commenters requested that we change the method by which we calculate the ALOS by excluding certain discharges, such as deaths and discharges associated with model demonstrations, from the calculation.
Response: We consider these comments outside the scope of the proposed rule as we did not make any policy proposals related to the method by which the ALOS would be calculated; however, we will keep these comments in mind for future rulemaking. ( print page 69422)
The following hospitals are paid under special payment provisions, as described in § 412.22(c) and, therefore, are not subject to the LTCH PPS rules:
- Veterans Administration hospitals.
- Hospitals that are reimbursed under State cost control systems approved under 42 CFR part 403 .
- Hospitals that are reimbursed in accordance with demonstration projects authorized under section 402(a) of the Social Security Amendments of 1967 (Pub. L. 90-248) ( 42 U.S.C. 1395b- 1 ), section 222(a) of the Social Security Amendments of 1972 (Pub. L. 92-603) ( 42 U.S.C. 1395 b1 (note)) (Statewide-all payer systems, subject to the rate-of increase test at section 1814(b) of the Act), or section 3021 of the Patient Protection and Affordable Care Act ( Pub. L. 111-148 ) ( 42 U.S.C. 1315a ).
- Nonparticipating hospitals furnishing emergency services to Medicare beneficiaries.
In the August 30, 2002 final rule, we presented an in-depth discussion of beneficiary liability under the LTCH PPS ( 67 FR 55974 through 55975 ). This discussion was further clarified in the RY 2005 LTCH PPS final rule ( 69 FR 25676 ). In keeping with those discussions, if the Medicare payment to the LTCH is the full LTC-DRG payment amount, consistent with other established hospital prospective payment systems, § 412.507 currently provides that an LTCH may not bill a Medicare beneficiary for more than the deductible and coinsurance amounts as specified under §§ 409.82, 409.83, and 409.87, and for items and services specified under § 489.30(a). However, under the LTCH PPS, Medicare will only pay for services furnished during the days for which the beneficiary has coverage until the short-stay outlier (SSO) threshold is exceeded. If the Medicare payment was for a SSO case (in accordance with § 412.529), and that payment was less than the full LTC-DRG payment amount because the beneficiary had insufficient coverage as a result of the remaining Medicare days, the LTCH also is currently permitted to charge the beneficiary for services delivered on those uncovered days (in accordance with § 412.507). In the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49623 ), we amended our regulations to expressly limit the charges that may be imposed upon beneficiaries whose LTCHs' discharges are paid at the site neutral payment rate under the LTCH PPS. In the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57102 ), we amended the regulations under § 412.507 to clarify our existing policy that blended payments made to an LTCH during its transitional period (that is, an LTCH's payment for discharges occurring in cost reporting periods beginning in FYs 2016 through 2019) are considered to be site neutral payment rate payments.
Comment: A commenter requested that we provide additional payments for ESRD patients in LTCHs, similar to the ESRD add-on payment for IPPS hospitals.
Response: We consider this comment outside the scope of the proposed rule. We did not make any proposals related to additional payments for ESRD patients in LTCHs; however, we will keep the commenter's request in mind for future rulemaking.
Section 123 of the BBRA required that the Secretary implement a PPS for LTCHs to replace the cost-based payment system under TEFRA. Section 307(b)(1) of the BIPA modified the requirements of section 123 of the BBRA by requiring that the Secretary examine the feasibility and the impact of basing payment under the LTCH PPS on the use of existing (or refined) hospital DRGs that have been modified to account for different resource use of LTCH patients.
Under both the IPPS and the LTCH PPS, the DRG-based classification system uses information on the claims for inpatient discharges to classify patients into distinct groups (for example, DRGs) based on clinical characteristics and expected resource needs. When the LTCH PPS was implemented for cost reporting periods beginning on or after October 1, 2002, we adopted the same DRG patient classification system utilized at that time under the IPPS. We referred to this patient classification system as the “long-term care diagnosis-related groups (LTC-DRGs).” As part of our efforts to better recognize severity of illness among patients, in the FY 2008 IPPS final rule with comment period ( 72 FR 47130 ), we adopted the MS-DRGs and the Medicare severity long-term care diagnosis-related groups (MS-LTC-DRGs) under the IPPS and the LTCH PPS, respectively, effective beginning October 1, 2007 (FY 2008). For a full description of the development, implementation, and rationale for the use of the MS-DRGs and MS-LTC-DRGs, we refer readers to the FY 2008 IPPS final rule with comment period ( 72 FR 47141 through 47175 and 47277 through 47299 ). (We note that, in that same final rule, we revised the regulations at § 412.503 to specify that for LTCH discharges occurring on or after October 1, 2007, when applying the provisions of 42 CFR part 412, subpart O , applicable to LTCHs for policy descriptions and payment calculations, all references to LTC-DRGs would be considered a reference to MS-LTC-DRGs. For the remainder of this section, we present the discussion in terms of the current MS-LTC-DRG patient classification system unless specifically referring to the previous LTC-DRG patient classification system that was in effect before October 1, 2007.)
Consistent with section 123 of the BBRA, as amended by section 307(b)(1) of the BIPA, and § 412.515 of the regulations, we use information derived from LTCH PPS patient records to classify LTCH discharges into distinct MS-LTC-DRGs based on clinical characteristics and estimated resource needs. As noted previously, we adopted the same DRG patient classification system utilized at that time under the IPPS. The MS-DRG classifications are updated annually, which has resulted in the number of MS-DRGs changing over time. For FY 2025, there will be 773 MS-DRG, and by extension, MS-LTC-DRG, groupings based on the changes, as discussed in section II.E. of the preamble of this final rule.
Although the patient classification system used under both the LTCH PPS and the IPPS are the same, the relative weights are different. The established relative weight methodology and data used under the LTCH PPS result in relative weights under the LTCH PPS that reflect the differences in patient resource use of LTCH patients, consistent with section 123(a)(1) of the BBRA. That is, we assign an appropriate weight to the MS-LTC-DRGs to account for the differences in resource use by patients exhibiting the case complexity and multiple medical problems characteristic of LTCH patients.
The MS-DRGs (used under the IPPS) and the MS-LTC-DRGs (used under the LTCH PPS) are based on the CMS DRG structure. As noted previously in this section, we refer to the DRGs under the LTCH PPS as MS-LTC-DRGs although they are structurally identical to the MS-DRGs used under the IPPS. ( print page 69423)
The MS-DRGs are organized into 25 major diagnostic categories (MDCs), most of which are based on a particular organ system of the body; the remainder involve multiple organ systems (such as MDC 22, Burns). Within most MDCs, cases are then divided into surgical DRGs and medical DRGs. Surgical DRGs are assigned based on a surgical hierarchy that orders operating room (O.R.) procedures or groups of O.R. procedures by resource intensity. The GROUPER software program does not recognize all ICD-10-PCS procedure codes as procedures affecting DRG assignment. That is, procedures that are not surgical (for example, EKGs) or are minor surgical procedures (for example, a biopsy of skin and subcutaneous tissue (procedure code 0JBH3ZX)) do not affect the MS-LTC-DRG assignment based on their presence on the claim.
Generally, under the LTCH PPS, a Medicare payment is made at a predetermined specific rate for each discharge that varies based on the MS-LTC-DRG to which a beneficiary's discharge is assigned. Cases are classified into MS-LTC-DRGs for payment based on the following six data elements:
- Principal diagnosis.
- Additional or secondary diagnoses.
- Surgical procedures.
- Discharge status of the patient.
Currently, for claims submitted using the version ASC X12 5010 standard, up to 25 diagnosis codes and 25 procedure codes are considered for an MS-DRG assignment. This includes one principal diagnosis and up to 24 secondary diagnoses for severity of illness determinations. (For additional information on the processing of up to 25 diagnosis codes and 25 procedure codes on hospital inpatient claims, we refer readers to section II.G.11.c. of the preamble of the FY 2011 IPPS/LTCH PPS final rule ( 75 FR 50127 ).)
Under the HIPAA transactions and code sets regulations at 45 CFR parts 160 and 162 , covered entities must comply with the adopted transaction standards and operating rules specified in subparts I through S of part 162. Among other requirements, on or after January 1, 2012, covered entities are required to use the ASC X12 Standards for Electronic Data Interchange Technical Report Type 3—Health Care Claim: Institutional (837), May 2006, ASC X12N/005010X223, and Type 1 Errata to Health Care Claim: Institutional (837) ASC X12 Standards for Electronic Data Interchange Technical Report Type 3, October 2007, ASC X12N/005010X233A1 for the health care claims or equivalent encounter information transaction ( 45 CFR 162.1102(c) ).
HIPAA requires covered entities to use the applicable medical data code sets when conducting HIPAA transactions ( 45 CFR 162.1000 ). Currently, upon the discharge of the patient, the LTCH must assign appropriate diagnosis and procedure codes from the International Classification of Diseases, 10th Revision, Clinical Modification (ICD-10-CM) for diagnosis coding and the International Classification of Diseases, 10th Revision, Procedure Coding System (ICD-10-PCS) for inpatient hospital procedure coding, both of which were required to be implemented October 1, 2015 ( 45 CFR 162.1002(c)(2) and (3) ). For additional information on the implementation of the ICD-10 coding system, we refer readers to section II.F.1. of the preamble of the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 56787 through 56790 ) and section II.E.1. of the preamble of this final rule. Additional coding instructions and examples are published in the AHA's Coding Clinic for ICD-10-CM/PCS.
To create the MS-DRGs (and by extension, the MS-LTC-DRGs), base DRGs were subdivided according to the presence of specific secondary diagnoses designated as complications or comorbidities (CCs) into one, two, or three levels of severity, depending on the impact of the CCs on resources used for those cases. Specifically, there are sets of MS-DRGs that are split into 2 or 3 subgroups based on the presence or absence of a CC or a major complication or comorbidity (MCC). We refer readers to section II.D. of the preamble of the FY 2008 IPPS final rule with comment period for a detailed discussion about the creation of MS-DRGs based on severity of illness levels ( 72 FR 47141 through 47175 ).
Medicare Administrative Contractors (MACs) enter the clinical and demographic information submitted by LTCHs into their claims processing systems and subject this information to a series of automated screening processes called the Medicare Code Editor (MCE). These screens are designed to identify cases that require further review before assignment into a MS-LTC-DRG can be made. During this process, certain types of cases are selected for further explanation ( 74 FR 43949 ).
After screening through the MCE, each claim is classified into the appropriate MS-LTC-DRG by the Medicare LTCH GROUPER software on the basis of diagnosis and procedure codes and other demographic information (age, sex, and discharge status). The GROUPER software used under the LTCH PPS is the same GROUPER software program used under the IPPS. Following the MS-LTC-DRG assignment, the MAC determines the prospective payment amount by using the Medicare PRICER program, which accounts for hospital-specific adjustments. Under the LTCH PPS, we provide an opportunity for LTCHs to review the MS-LTC-DRG assignments made by the MAC and to submit additional information within a specified timeframe as provided in § 412.513(c).
The GROUPER software is used both to classify past cases to measure relative hospital resource consumption to establish the MS-LTC-DRG relative weights and to classify current cases for purposes of determining payment. The records for all Medicare hospital inpatient discharges are maintained in the MedPAR file. The data in this file are used to evaluate possible MS-DRG and MS-LTC-DRG classification changes and to recalibrate the MS-DRG and MS-LTC-DRG relative weights during our annual update under both the IPPS (§ 412.60(e)) and the LTCH PPS (§ 412.517), respectively.
As specified by our regulations at § 412.517(a), which require that the MS-LTC-DRG classifications and relative weights be updated annually, and consistent with our historical practice of using the same patient classification system under the LTCH PPS as is used under the IPPS, in this final rule, as we proposed, we updated the MS-LTC-DRG classifications effective October 1, 2024 through September 30, 2025 (FY 2025) consistent with the changes to specific MS-DRG classifications presented in section II.F. of the preamble of this final rule. Accordingly, the MS-LTC-DRGs for FY 2025 are the same as the MS-DRGs being used under the IPPS for FY 2025. In addition, because the MS-LTC-DRGs for FY 2025 are the same as the MS-DRGs for FY 2025, the other changes that affect MS-DRG (and by extension MS-LTC-DRG) assignments under GROUPER Version 42, as discussed in section II.E. of the preamble of this final rule, including the changes to the MCE software and the ICD-10-CM/PCS coding system, are also applicable under the LTCH PPS for FY 2025.3. Development of the FY 2025 MS-LTC-DRG Relative Weights. ( print page 69424)
One of the primary goals for the implementation of the LTCH PPS is to pay each LTCH an appropriate amount for the efficient delivery of medical care to Medicare patients. The system must be able to account adequately for each LTCH's case-mix to ensure both fair distribution of Medicare payments and access to adequate care for those Medicare patients whose care is costlier ( 67 FR 55984 ). To accomplish these goals, we have annually adjusted the LTCH PPS standard Federal prospective payment rate by the applicable relative weight in determining payment to LTCHs for each case. Under the LTCH PPS, relative weights for each MS-LTC-DRG are a primary element used to account for the variations in cost per discharge and resource utilization among the payment groups (§ 412.515). To ensure that Medicare patients classified to each MS-LTC-DRG have access to an appropriate level of services and to encourage efficiency, we calculate a relative weight for each MS-LTC-DRG that represents the resources needed by an average inpatient LTCH case in that MS-LTC-DRG. For example, cases in an MS-LTC-DRG with a relative weight of 2 would, on average, cost twice as much to treat as cases in an MS-LTC-DRG with a relative weight of 1.
The established methodology to develop the MS-LTC-DRG relative weights is generally consistent with the methodology established when the LTCH PPS was implemented in the August 30, 2002 LTCH PPS final rule ( 67 FR 55989 through 55991 ). However, there have been some modifications of our historical procedures for assigning relative weights in cases of zero volume or nonmonotonicity or both resulting from the adoption of the MS-LTC-DRGs. We also made a modification in conjunction with the implementation of the dual rate LTCH PPS payment structure beginning in FY 2016 to use LTCH claims data from only LTCH PPS standard Federal payment rate cases (or LTCH PPS cases that would have qualified for payment under the LTCH PPS standard Federal payment rate if the dual rate LTCH PPS payment structure had been in effect at the time of the discharge). We also adopted, beginning in FY 2023, a 10-percent cap policy on the reduction in a MS-LTC-DRG's relative weight in a given year. (For details on the modifications to our historical procedures for assigning relative weights in cases of zero volume and nonmonotonicity or both, we refer readers to the FY 2008 IPPS final rule with comment period ( 72 FR 47289 through 47295 ) and the FY 2009 IPPS final rule ( 73 FR 48542 through 48550 ). For details on the change in our historical methodology to use LTCH claims data only from LTCH PPS standard Federal payment rate cases (or cases that would have qualified for such payment had the LTCH PPS dual payment rate structure been in effect at the time) to determine the MS-LTC-DRG relative weights, we refer readers to the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49614 through 49617 ). For details on our adoption of the 10-percent cap policy, we refer readers to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49152 through 49154 ).)
For purposes of determining the MS-LTC-DRG relative weights, under our historical methodology, there are three different categories of MS-LTC-DRGs based on volume of cases within specific MS-LTC-DRGs: (1) MS-LTC-DRGs with at least 25 applicable LTCH cases in the data used to calculate the relative weight, which are each assigned a unique relative weight; (2) low-volume MS-LTC-DRGs (that is, MS-LTC-DRGs that contain between 1 and 24 applicable LTCH cases that are grouped into quintiles (as described later in this section in Step 3 of our methodology) and assigned the relative weight of the quintile); and (3) no-volume MS-LTC-DRGs that are cross-walked to other MS-LTC-DRGs based on the clinical similarities and assigned the relative weight of the cross-walked MS-LTC-DRG (as described later in this section in Step 8 of our methodology). For FY 2025, we are continuing to use applicable LTCH cases to establish the same volume-based categories to calculate the FY 2025 MS-LTC-DRG relative weights.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36259 through 36266 ), we presented our proposed methodology for determining the MS-LTC-DRG relative weights for FY 2025.
We received several comments requesting that CMS modify certain high-volume MS-LTC-DRGs to better account for the variation in patient severity and costs among the cases grouped to these MS-LTC-DRGs. Since these comments were primarily focused on the impact these high-volume MS-LTC-DRGs have on the FY 2025 outlier fixed-loss amount, we have summarized and responded to these comments in section V.D.3. of the Addendum to this final rule. We also received comments requesting CMS to modify our ratesetting methodologies to account for the impact of COVID-19 on the ratesetting data. Since these comments were primarily focused on the specific use of FY 2023 data when determining the FY 2025 outlier fixed-loss amount, we also have summarized and responded to these comments in section V.D.3. of the Addendum to this final rule.
After consideration of the comments we received, we are finalizing, without modification, our proposed methodology for determining the MS-LTC-DRG relative weights for FY 2025. In the remainder of this section, we present our finalized methodology. We first list and provide a brief description of our steps for determining the FY 2025 MS-LTC-DRG relative weights. We then, later in this section, discuss in greater detail each step. We note that, as we did in FY 2024, we used our historical relative weight methodology as described in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58898 through 58907 ), subject to a ten percent cap as described in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49162 ).
- Step 1—Prepare data for MS-LTC-DRG relative weight calculation. In this step, we select and group the applicable claims data used in the development of the MS-LTC-DRG relative weights.
- Step 2—Remove cases with a length of stay of 7 days or less. In this step, we trim the applicable claims data to remove cases with a length of stay of 7 days or less.
Step 3—Establish low-volume MS-LTC-DRG quintiles. In this step, we employ our established quintile methodology for low-volume MS-LTC-DRGs (that is, MS-LTC-DRGs with fewer than 25 cases).
- Step 4—Remove statistical outliers. In this step, we trim the applicable claims data to remove statistical outlier cases.
- Step 5—Adjust charges for the effects of Short Stay Outliers (SSOs). In this step, we adjust the number of applicable cases in each MS-LTC-DRG (or low-volume quintile) for the effect of SSO cases.
- Step 6—Calculate the relative weights on an iterative basis using the hospital-specific relative weights methodology. In this step, we use our established hospital-specific relative value (HSRV) methodology, which is an iterative process, to calculate the relative weights.
- Step 7—Adjust the relative weights to account for nonmonotonically increasing relative weights. In this step, we make adjustments that ensure that within each base MS-LTC-DRG, the relative weights increase by MS-LTC-DRG severity. ( print page 69425)
- Step 8—Determine a relative weight for MS-LTC-DRGs with no applicable LTCH cases. In this step, we cross-walk each no-volume MS-LTC-DRG to another MS-LTC-DRG for which we calculated a relative weight.
- Step 9—Budget neutralize the uncapped relative weights. In this step, to ensure budget neutrality in the annual update to the MS-LTC-DRG classifications and relative weights, we adjust the relative weights by a normalization factor and a budget neutrality factor that ensures estimated aggregate LTCH PPS payments will be unaffected by the updates to the MS-LTC-DRG classifications and relative weights.
- Step 10—Apply the 10-percent cap to decreases in MS-LTC-DRG relative weights. In this step we limit the reduction of the relative weight for a MS-LTC-DRG to 10 percent of its prior year value. This 10-percent cap does not apply to zero-volume MS-LTC-DRGs or low-volume MS-LTC-DRGs.
- Step 11—Budget neutralize the application of the 10-percent cap policy. In this step, to ensure budget neutrality in the application of the MS-LTC-DRG cap policy, we adjust the relative weights by a budget neutrality factor that ensures estimated aggregate LTCH PPS payments will be unaffected by our application of the cap to the MS-LTC-DRG relative weights.
We next describe each of the 11 steps for calculating the FY 2025 MS-LTC-DRG relative weights in greater detail.
Step 1—Prepare data for MS-LTC-DRG relative weight calculation.
For the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36260 ), we obtained total charges from FY 2023 Medicare LTCH claims data from the December 2023 update of the FY 2023 MedPAR file and used proposed Version 42 of the GROUPER to classify LTCH cases. Consistent with our historical practice, we proposed that if better data become available, we would use those data and the finalized Version 42 of the GROUPER in establishing the FY 2025 MS-LTC-DRG relative weights in the final rule. Accordingly, for this final rule, we are establishing the FY 2025 MS-LTC-DRG relative weights based on updated FY 2023 Medicare LTCH claims data from the March 2024 update of the FY 2023 MedPAR file, which is the best available data at the time of development of this final rule, and the finalized Version 42 of the GROUPER to classify LTCH cases.
To calculate the FY 2025 MS-LTC-DRG relative weights under the dual rate LTCH PPS payment structure, as we proposed, we continue to use applicable LTCH data, which includes our policy of only using cases that meet the criteria for exclusion from the site neutral payment rate (or would have met the criteria had they been in effect at the time of the discharge) ( 80 FR 49624 ). Specifically, we began by first evaluating the LTCH claims data in the March 2024 update of the FY 2023 MedPAR file to determine which LTCH cases would meet the criteria for exclusion from the site neutral payment rate under § 412.522(b) or had the dual rate LTCH PPS payment structure applied to those cases at the time of discharge. We identified the FY 2023 LTCH cases that were not assigned to MS-LTC-DRGs 876, 880, 881, 882, 883, 884, 885, 886, 887, 894, 895, 896, 897, 945, and 946, which identify LTCH cases that do not have a principal diagnosis relating to a psychiatric diagnosis or to rehabilitation; and that either—
- The admission to the LTCH was “immediately preceded” by discharge from a subsection (d) hospital and the immediately preceding stay in that subsection (d) hospital included at least 3 days in an ICU, as we define under the ICU criterion; or
- The admission to the LTCH was “immediately preceded” by discharge from a subsection (d) hospital and the claim for the LTCH discharge includes the applicable procedure code that indicates at least 96 hours of ventilator services were provided during the LTCH stay, as we define under the ventilator criterion. Claims data from the FY 2023 MedPAR file that reported ICD-10-PCS procedure code 5A1955Z were used to identify cases involving at least 96 hours of ventilator services in accordance with the ventilator criterion. (We note that section 3711(b)(2) of the CARES Act provided a waiver of the application of the site neutral payment rate for LTCH cases admitted during the COVID-19 PHE period. The COVID-19 PHE expired on May 11, 2023. Therefore, all LTCH PPS cases in FY 2023 with admission dates on or before the PHE expiration date were paid the LTCH PPS standard Federal rate regardless of whether the discharge met the statutory patient criteria. However, for purposes of setting rates for LTCH PPS standard Federal rate cases for FY 2025 (including MS-LTC-DRG relative weights), we used FY 2023 cases that meet the statutory patient criteria without consideration to how those cases were paid in FY 2023.)
Furthermore, consistent with our historical methodology, we excluded any claims in the resulting data set that were submitted by LTCHs that were all-inclusive rate providers and LTCHs that are paid in accordance with demonstration projects authorized under section 402(a) of Public Law 90-248 or section 222(a) of Public Law 92-603. In addition, consistent with our historical practice and our policies, we excluded any Medicare Advantage (Part C) claims in the resulting data. Such claims were identified based on the presence of a GHO Paid indicator value of “1” in the MedPAR files.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49448 ), we discussed the abnormal charging practices of an LTCH (CCN 312024) in FY 2021 that led to the LTCH receiving an excessive amount of high cost outlier payments. In that rule, we stated our belief, based on information we received from the provider, that these abnormal charging practices would not persist into FY 2023. Therefore, we did not include their cases in our model for determining the FY 2023 outlier fixed-loss amount. In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59127 through 59128 ), we stated that the FY 2022 MedPAR claims also reflect the abnormal charging practices of this LTCH. Therefore, we removed claims from CCN 312024 when determining the FY 2024 MS-LTC-DRG relative weights and from all other FY 2024 ratesetting calculations, including the calculation of the area wage level adjustment budget neutrality factor and the fixed-loss amount for LTCH PPS standard Federal payment rate cases. Given recent actions by the Department of Justice regarding CCN 312024 (see https://www.justice.gov/opa/pr/new-jersey-hospital-and-investors-pay-united-states-306-million-alleged-false-claims-related ), as we proposed, we again removed claims from CCN 312024 when determining the FY 2025 MS-LTC-DRG relative weights and all other FY 2025 ratesetting calculations, including the calculation of the area wage level adjustment budget neutrality factor and the fixed-loss amount for LTCH PPS standard Federal payment rate cases.
In summary, in general, we identified the claims data used in the development of the FY 2025 MS-LTC-DRG relative weights in this final rule by trimming claims data that were paid the site neutral payment rate or would have been paid the site neutral payment rate had the provisions of the CARES Act not been in effect. We trimmed the claims data of all-inclusive rate providers reported in the March 2024 update of the FY 2023 MedPAR file and any Medicare Advantage claims data. There were no data from any LTCHs that are paid in accordance with a demonstration project reported in the March 2024 update of the FY 2023 MedPAR file, but had there been any, ( print page 69426) we would have trimmed the claims data from those LTCHs as well, in accordance with our established policy. We also removed all claims from CCN 312024.
We used the remaining data (that is, the applicable LTCH data) in the subsequent steps to calculate the MS-LTC-DRG relative weights for FY 2025.
Step 2—Remove cases with a length of stay of 7 days or less.
The next step in our calculation of the FY 2025 MS-LTC-DRG relative weights is to remove cases with a length of stay of 7 days or less. The MS-LTC-DRG relative weights reflect the average of resources used on representative cases of a specific type. Generally, cases with a length of stay of 7 days or less do not belong in an LTCH because these stays do not fully receive or benefit from treatment that is typical in an LTCH stay, and full resources are often not used in the earlier stages of admission to an LTCH. If we were to include stays of 7 days or less in the computation of the FY 2025 MS-LTC-DRG relative weights, the value of many relative weights would decrease and, therefore, payments would decrease to a level that may no longer be appropriate. We do not believe that it would be appropriate to compromise the integrity of the payment determination for those LTCH cases that actually benefit from and receive a full course of treatment at an LTCH by including data from these very short stays. Therefore, as we proposed, consistent with our existing relative weight methodology, in determining the FY 2025 MS-LTC-DRG relative weights, we removed LTCH cases with a length of stay of 7 days or less from applicable LTCH cases. (For additional information on what is removed in this step of the relative weight methodology, we refer readers to 67 FR 55989 and 74 FR 43959 .)
Step 3—Establish low-volume MS-LTC-DRG quintiles.
To account for MS-LTC-DRGs with low-volume (that is, with fewer than 25 applicable LTCH cases), consistent with our existing methodology, as we proposed, we are continuing to employ the quintile methodology for low-volume MS-LTC-DRGs, such that we grouped the “low-volume MS-LTC-DRGs” (that is, MS-LTC-DRGs that contain between 1 and 24 applicable LTCH cases into one of five categories (quintiles) based on average charges ( 67 FR 55984 through 55995 ; 72 FR 47283 through 47288 ; and 81 FR 25148 )).
In this final rule, based on the best available data (that is, the March 2024 update of the FY 2023 MedPAR file), we identified 235 MS-LTC-DRGs that contained between 1 and 24 applicable LTCH cases. This list of MS-LTC-DRGs was then divided into 1 of the 5 low-volume quintiles. We assigned the low-volume MS-LTC-DRGs to specific low-volume quintiles by sorting the low-volume MS-LTC-DRGs in ascending order by average charge in accordance with our established methodology. Based on the data available for this final rule, the number of MS-LTC-DRGs with less than 25 applicable LTCH cases was evenly divisible by 5. Therefore, the quintiles each contained 47 MS-LTC-DRGs (235/5 = 47). Since the number of MS LTC DRGs with less than 25 applicable LTCH cases was evenly divisible by 5, it was unnecessary to employ our historical methodology of assigning each remainder low-volume MS-LTC-DRG to the low-volume quintile that contains an MS-LTC-DRG with an average charge closest to that of the remainder low-volume MS-LTC-DRG. In cases where these initial assignments of low-volume MS-LTC-DRGs to quintiles results in nonmonotonicity within a base-DRG, as we proposed, we adjusted the resulting low-volume MS-LTC-DRGs to preserve monotonicity, as discussed in Step 7 of our methodology.
To determine the FY 2025 relative weights for the low-volume MS-LTC-DRGs, consistent with our historical practice, we used the five low-volume quintiles described previously. We determined a relative weight and (geometric) average length of stay for each of the five low-volume quintiles using the methodology described in Step 6 of our methodology. We assigned the same relative weight and average length of stay to each of the low-volume MS-LTC-DRGs that make up an individual low-volume quintile. We note that, as this system is dynamic, it is possible that the number and specific type of MS-LTC-DRGs with a low-volume of applicable LTCH cases would vary in the future. Furthermore, we note that we continue to monitor the volume (that is, the number of applicable LTCH cases) in the low-volume quintiles to ensure that our quintile assignments used in determining the MS-LTC-DRG relative weights result in appropriate payment for LTCH cases grouped to low-volume MS-LTC-DRGs and do not result in an unintended financial incentive for LTCHs to inappropriately admit these types of cases.
For this final rule, we are providing the list of the composition of the low-volume quintiles for low-volume MS-LTC-DRGs in a supplemental data file for public use posted via the internet on the CMS website for this final rule at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html to streamline the information made available to the public that is used in the annual development of Table 11.
Step 4—Remove statistical outliers.
The next step in our calculation of the FY 2025 MS-LTC-DRG relative weights is to remove statistical outlier cases from the LTCH cases with a length of stay of at least 8 days. Consistent with our existing relative weight methodology, as we proposed, we are continuing to define statistical outliers as cases that are outside of 3.0 standard deviations from the mean of the log distribution of both charges per case and the charges per day for each MS-LTC-DRG. These statistical outliers are removed prior to calculating the relative weights because we believe that they may represent aberrations in the data that distort the measure of average resource use. Including those LTCH cases in the calculation of the relative weights could result in an inaccurate relative weight that does not truly reflect relative resource use among those MS-LTC-DRGs. (For additional information on what is removed in this step of the relative weight methodology, we refer readers to 67 FR 55989 and 74 FR 43959 .) After removing cases with a length of stay of 7 days or less and statistical outliers, in each set of claims, we were left with applicable LTCH cases that have a length of stay greater than or equal to 8 days. In this final rule, we refer to these cases as “trimmed applicable LTCH cases.”
Step 5—Adjust charges for the effects of Short Stay Outliers (SSOs).
As the next step in the calculation of the FY 2025 MS-LTC-DRG relative weights, consistent with our historical approach, as we proposed, we adjusted each LTCH's charges per discharge for those remaining cases (that is, trimmed applicable LTCH cases) for the effects of SSOs (as defined in § 412.529(a) in conjunction with § 412.503). Specifically, as we proposed, we made this adjustment by counting an SSO case as a fraction of a discharge based on the ratio of the length of stay of the case to the average length of stay of all cases grouped to the MS-LTC-DRG. This has the effect of proportionately reducing the impact of the lower charges for the SSO cases in calculating the average charge for the MS-LTC-DRG. This process produces the same result as if the actual charges per discharge of an SSO case were adjusted to what they would have been had the patient's length of stay been equal to the average length of stay of the MS-LTC-DRG.
Counting SSO cases as full LTCH cases with no adjustment in ( print page 69427) determining the FY 2025 MS-LTC-DRG relative weights would lower the relative weight for affected MS-LTC-DRGs because the relatively lower charges of the SSO cases would bring down the average charge for all cases within a MS-LTC-DRG. This would result in an “underpayment” for non-SSO cases and an “overpayment” for SSO cases. Therefore, we are continuing to adjust for SSO cases under § 412.529 in this manner because it would result in more appropriate payments for all LTCH PPS standard Federal payment rate cases. (For additional information on this step of the relative weight methodology, we refer readers to 67 FR 55989 and 74 FR 43959 .)
Step 6—Calculate the relative weights on an iterative basis using the hospital-specific relative value methodology.
By nature, LTCHs often specialize in certain areas, such as ventilator-dependent patients. Some case types (MS-LTC-DRGs) may be treated, to a large extent, in hospitals that have, from a perspective of charges, relatively high (or low) charges. This nonrandom distribution of cases with relatively high (or low) charges in specific MS-LTC-DRGs has the potential to inappropriately distort the measure of average charges. To account for the fact that cases may not be randomly distributed across LTCHs, consistent with the methodology we have used since the implementation of the LTCH PPS, in this FY 2025 IPPS/LTCH PPS final rule, as we proposed, we are continuing to use a hospital-specific relative value (HSRV) methodology to calculate the MS-LTC-DRG relative weights for FY 2025. We believe that this method removes this hospital-specific source of bias in measuring LTCH average charges ( 67 FR 55985 ). Specifically, under this methodology, we reduced the impact of the variation in charges across providers on any particular MS-LTC-DRG relative weight by converting each LTCH's charge for an applicable LTCH case to a relative value based on that LTCH's average charge for such cases.
Under the HSRV methodology, we standardize charges for each LTCH by converting its charges for each applicable LTCH case to hospital-specific relative charge values and then adjusting those values for the LTCH's case-mix. The adjustment for case-mix is needed to rescale the hospital-specific relative charge values (which, by definition, average 1.0 for each LTCH). The average relative weight for an LTCH is its case-mix; therefore, it is reasonable to scale each LTCH's average relative charge value by its case-mix. In this way, each LTCH's relative charge value is adjusted by its case-mix to an average that reflects the complexity of the applicable LTCH cases it treats relative to the complexity of the applicable LTCH cases treated by all other LTCHs (the average LTCH PPS case-mix of all applicable LTCH cases across all LTCHs). In other words, by multiplying an LTCH's relative charge values by the LTCH's case-mix index, we account for the fact that the same relative charges are given greater weight at an LTCH with higher average costs than they would at an LTCH with low average costs, which is needed to adjust each LTCH's relative charge value to reflect its case-mix relative to the average case-mix for all LTCHs. By standardizing charges in this manner, we count charges for a Medicare patient at an LTCH with high average charges as less resource-intensive than they would be at an LTCH with low average charges. For example, a $10,000 charge for a case at an LTCH with an average adjusted charge of $17,500 reflects a higher level of relative resource use than a $10,000 charge for a case at an LTCH with the same case-mix, but an average adjusted charge of $35,000. We believe that the adjusted charge of an individual case more accurately reflects actual resource use for an individual LTCH because the variation in charges due to systematic differences in the markup of charges among LTCHs is taken into account.
Consistent with our historical relative weight methodology, as we proposed, we calculated the FY 2025 MS-LTC-DRG relative weights using the HSRV methodology, which is an iterative process. Therefore, in accordance with our established methodology, for FY 2025, we continued to standardize charges for each applicable LTCH case by first dividing the adjusted charge for the case (adjusted for SSOs under § 412.529 as described in Step 5 of our methodology) by the average adjusted charge for all applicable LTCH cases at the LTCH in which the case was treated. The average adjusted charge reflects the average intensity of the health care services delivered by a particular LTCH and the average cost level of that LTCH. The average adjusted charge is then multiplied by the LTCH's case-mix index to produce an adjusted hospital-specific relative charge value for the case. We used an initial case-mix index value of 1.0 for each LTCH.
For each MS-LTC-DRG, we calculated the FY 2025 relative weight by dividing the SSO-adjusted average of the hospital-specific relative charge values for applicable LTCH cases for the MS-LTC-DRG (that is, the sum of the hospital-specific relative charge value, as previously stated, divided by the sum of equivalent cases from Step 5 for each MS-LTC-DRG) by the overall SSO-adjusted average hospital-specific relative charge value across all applicable LTCH cases for all LTCHs (that is, the sum of the hospital-specific relative charge value, as previously stated, divided by the sum of equivalent applicable LTCH cases from Step 5 for each MS-LTC-DRG). Using these recalculated MS-LTC-DRG relative weights, each LTCH's average relative weight for all of its SSO-adjusted trimmed applicable LTCH cases (that is, it's case-mix) was calculated by dividing the sum of all the LTCH's MS-LTC-DRG relative weights by its total number of SSO-adjusted trimmed applicable LTCH cases. The LTCHs' hospital-specific relative charge values (from previous) are then multiplied by the hospital-specific case-mix indexes. The hospital-specific case-mix adjusted relative charge values are then used to calculate a new set of MS-LTC-DRG relative weights across all LTCHs. This iterative process continued until there was convergence between the relative weights produced at adjacent steps, for example, when the maximum difference was less than 0.0001.
Step 7—Adjust the relative weights to account for nonmonotonically increasing relative weights.
The MS-DRGs contain base DRGs that have been subdivided into one, two, or three severity of illness levels. Where there are three severity levels, the most severe level has at least one secondary diagnosis code that is referred to as an MCC (that is, major complication or comorbidity). The next lower severity level contains cases with at least one secondary diagnosis code that is a CC (that is, complication or comorbidity). Those cases without an MCC or a CC are referred to as “without CC/MCC.” When data do not support the creation of three severity levels, the base MS-DRG is subdivided into either two levels or the base MS-DRG is not subdivided. The two-level subdivisions may consist of the MS-DRG with CC/MCC and the MS-DRG without CC/MCC. Alternatively, the other type of two-level subdivision may consist of the MS-DRG with MCC and the MS-DRG without MCC.
In those base MS-LTC-DRGs that are split into either two or three severity levels, cases classified into the “without CC/MCC” MS-LTC-DRG are expected to have a lower resource use (and lower costs) than the “with CC/MCC” MS-LTC-DRG (in the case of a two-level split) or both the “with CC” and the “with MCC” MS-LTC-DRGs (in the case of a three-level split). That is, theoretically, cases that are more severe ( print page 69428) typically require greater expenditure of medical care resources and would result in higher average charges. Therefore, in the three severity levels, relative weights should increase by severity, from lowest to highest. If the relative weights decrease as severity increases (that is, if within a base MS-LTC-DRG, an MS-LTC-DRG with CC has a higher relative weight than one with MCC, or the MS-LTC-DRG “without CC/MCC” has a higher relative weight than either of the others), they are nonmonotonic. We continue to believe that utilizing nonmonotonic relative weights to adjust Medicare payments would result in inappropriate payments because the payment for the cases in the higher severity level in a base MS-LTC-DRG (which are generally expected to have higher resource use and costs) would be lower than the payment for cases in a lower severity level within the same base MS-LTC-DRG (which are generally expected to have lower resource use and costs). Therefore, in determining the FY 2025 MS-LTC-DRG relative weights, consistent with our historical methodology, as we proposed, we continued to combine MS-LTC-DRG severity levels within a base MS-LTC-DRG for the purpose of computing a relative weight when necessary to ensure that monotonicity is maintained. For a comprehensive description of our existing methodology to adjust for nonmonotonicity, we refer readers to the FY 2010 IPPS/RY 2010 LTCH PPS final rule ( 74 FR 43964 through 43966 ). Any adjustments for nonmonotonicity that were made in determining the FY 2025 MS-LTC-DRG relative weights by applying this methodology are denoted in Table 11, which is listed in section VI. of the Addendum to this final rule and is available via the internet on the CMS website.
Step 8—Determine a relative weight for MS-LTC-DRGs with no applicable LTCH cases.
Using the trimmed applicable LTCH cases, consistent with our historical methodology, we identified the MS-LTC-DRGs for which there were no claims in the March 2024 update of the FY 2023 MedPAR file and, therefore, for which no charge data was available for these MS-LTC-DRGs. Because patients with a number of the diagnoses under these MS-LTC-DRGs may be treated at LTCHs, consistent with our historical methodology, we generally assign a relative weight to each of the no-volume MS-LTC-DRGs based on clinical similarity and relative costliness (with the exception of “transplant” MS-LTC-DRGs, “error” MS-LTC-DRGs, and MS-LTC-DRGs that indicate a principal diagnosis related to a psychiatric diagnosis or rehabilitation (referred to as the “psychiatric or rehabilitation” MS-LTC-DRGs), as discussed later in this section of this final rule). (For additional information on this step of the relative weight methodology, we refer readers to 67 FR 55991 and 74 FR 43959 through 43960 .)
Consistent with our existing methodology, as we proposed, we cross-walked each no-volume MS-LTC-DRG to another MS-LTC-DRG for which we calculated a relative weight (determined in accordance with the methodology as previously described). Then, the “no-volume” MS-LTC-DRG is assigned the same relative weight (and average length of stay) of the MS-LTC-DRG to which it was cross-walked (as described in greater detail in this section of this final rule).
Of the 773 MS-LTC-DRGs for FY 2025, we identified 426 MS-LTC-DRGs for which there were no trimmed applicable LTCH cases. The 426 MS-LTC-DRGs for which there were no trimmed applicable LTCH cases includes the 11 “transplant” MS-LTC-DRGs, the 2 “error” MS-LTC-DRGs, and the 15 “psychiatric or rehabilitation” MS-LTC-DRGs, which are discussed in this section of this rule, such that we identified 398 MS-LTC-DRGs that for which, we assigned a relative weight using our existing “no-volume” MS-LTC-DRG methodology (that is, 426−11−2−15 = 398). As we proposed, we assigned relative weights to each of the 398 no-volume MS-LTC-DRGs based on clinical similarity and relative costliness to 1 of the remaining 347 (773−426 = 347) MS-LTC-DRGs for which we calculated relative weights based on the trimmed applicable LTCH cases in the FY 2023 MedPAR file data using the steps described previously. (For the remainder of this discussion, we refer to the “cross-walked” MS-LTC-DRGs as one of the 347 MS-LTC-DRGs to which we cross-walked each of the 398 “no-volume” MS-LTC-DRGs.) Then, in general, we assigned the 398 no-volume MS-LTC-DRGs the relative weight of the cross-walked MS-LTC-DRG (when necessary, we made adjustments to account for nonmonotonicity).
We cross-walked the no-volume MS-LTC-DRG to a MS-LTC-DRG for which we calculated relative weights based on the March 2024 update of the FY 2023 MedPAR file, and to which it is similar clinically in intensity of use of resources and relative costliness as determined by criteria such as care provided during the period of time surrounding surgery, surgical approach (if applicable), length of time of surgical procedure, postoperative care, and length of stay. (For more details on our process for evaluating relative costliness, we refer readers to the FY 2010 IPPS/RY 2010 LTCH PPS final rule ( 73 FR 48543 ).) We believe in the rare event that there would be a few LTCH cases grouped to one of the no-volume MS-LTC-DRGs in FY 2025, the relative weights assigned based on the cross-walked MS-LTC-DRGs would result in an appropriate LTCH PPS payment because the crosswalks, which are based on clinical similarity and relative costliness, would be expected to generally require equivalent relative resource use.
Then we assigned the relative weight of the cross-walked MS-LTC-DRG as the relative weight for the no-volume MS-LTC-DRG such that both of these MS-LTC-DRGs (that is, the no-volume MS-LTC-DRG and the cross-walked MS-LTC-DRG) have the same relative weight (and average length of stay) for FY 2025. We note that, if the cross-walked MS-LTC-DRG had 25 applicable LTCH cases or more, its relative weight (calculated using the methodology as previously described in Steps 1 through 4) is assigned to the no-volume MS-LTC-DRG as well. Similarly, if the MS-LTC-DRG to which the no-volume MS-LTC-DRG was cross-walked had 24 or less cases and, therefore, was designated to 1 of the low-volume quintiles for purposes of determining the relative weights, we assigned the relative weight of the applicable low-volume quintile to the no-volume MS-LTC-DRG such that both of these MS-LTC-DRGs (that is, the no-volume MS-LTC-DRG and the cross-walked MS-LTC-DRG) have the same relative weight for FY 2025. (As we noted previously, in the infrequent case where nonmonotonicity involving a no-volume MS-LTC-DRG resulted, additional adjustments are required to maintain monotonically increasing relative weights.)
For this final rule, we are providing the list of the no-volume MS-LTC-DRGs and the MS-LTC-DRGs to which each was cross-walked (that is, the cross-walked MS-LTC-DRGs) for FY 2025 in a supplemental data file for public use posted via the internet on the CMS website for this final rule at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html to streamline the information made available to the public that is used in the annual development of Table 11.
To illustrate this methodology for determining the relative weights for the FY 2025 MS-LTC-DRGs with no applicable LTCH cases, we are providing the following example. ( print page 69429)
Example: There were no trimmed applicable LTCH cases in the FY 2023 MedPAR file that we are using for this final rule for MS-LTC-DRG 061 (Ischemic stroke, precerebral occlusion or transient ischemia with thrombolytic agent with MCC). We determined that MS-LTC-DRG 064 (Intracranial hemorrhage or cerebral infarction with MCC) is similar clinically and based on resource use to MS-LTC-DRG 061. Therefore, we assigned the same relative weight (and average length of stay) of MS-LTC-DRG 064 of 1.3008 for FY 2025 to MS-LTC-DRG 061 (we refer readers to Table 11, which is listed in section VI. of the Addendum to this final rule and is available via the internet on the CMS website).
Again, we note that, as this system is dynamic, it is entirely possible that the number of MS-LTC-DRGs with no volume would vary in the future. Consistent with our historical practice, as we proposed, we used the best available claims data to identify the trimmed applicable LTCH cases from which we determined the relative weights in the final rule.
For FY 2025, consistent with our historical relative weight methodology, as we proposed, we are establishing a relative weight of 0.0000 for the following transplant MS-LTC-DRGs: Heart Transplant or Implant of Heart Assist System with MCC (MS-LTC-DRG 001); Heart Transplant or Implant of Heart Assist System without MCC (MS-LTC-DRG 002); Liver Transplant with MCC or Intestinal Transplant (MS-LTC-DRG 005); Liver Transplant without MCC (MS-LTC-DRG 006); Lung Transplant (MS-LTC-DRG 007); Simultaneous Pancreas and Kidney Transplant (MS-LTC-DRG 008); Simultaneous Pancreas and Kidney Transplant with Hemodialysis (MS-LTC-DRG 019); Pancreas Transplant (MS-LTC-DRG 010); Kidney Transplant (MS-LTC-DRG 652); Kidney Transplant with Hemodialysis with MCC (MS-LTC-DRG 650), and Kidney Transplant with Hemodialysis without MCC (MS LTC DRG 651). This is because Medicare only covers these procedures if they are performed at a hospital that has been certified for the specific procedures by Medicare and presently no LTCH has been so certified. At the present time, we include these 11 transplant MS-LTC-DRGs in the GROUPER program for administrative purposes only. Because we use the same GROUPER program for LTCHs as is used under the IPPS, removing these MS-LTC-DRGs would be administratively burdensome. (For additional information regarding our treatment of transplant MS-LTC-DRGs, we refer readers to the RY 2010 LTCH PPS final rule ( 74 FR 43964 ).) In addition, consistent with our historical policy, we are establishing a relative weight of 0.0000 for the 2 “error” MS-LTC-DRGs (that is, MS-LTC-DRG 998 (Principal Diagnosis Invalid as Discharge Diagnosis) and MS-LTC-DRG 999 (Ungroupable)) because applicable LTCH cases grouped to these MS-LTC-DRGs cannot be properly assigned to an MS-LTC-DRG according to the grouping logic.
Additionally, we are establishing a relative weight of 0.0000 for the following “psychiatric or rehabilitation” MS-LTC-DRGs: MS-LTC-DRG 876 (O.R. Procedures with Principal Diagnosis of Mental Illness); MS-LTC-DRG 880 (Acute Adjustment Reaction & Psychosocial Dysfunction); MS-LTC-DRG 881 (Depressive Neuroses); MS-LTC-DRG 882 (Neuroses Except Depressive); MS-LTC-DRG 883 (Disorders of Personality & Impulse Control); MS-LTC-DRG 884 (Organic Disturbances & Intellectual Disability); MS-LTC-DRG 885 (Psychoses); MS-LTC-DRG 886 (Behavioral & Developmental Disorders); MS-LTC-DRG 887 (Other Mental Disorder Diagnoses); MS-LTC-DRG 894 (Alcohol, Drug Abuse or Dependence, Left AMA); MS-LTC-DRG 895 (Alcohol, Drug Abuse or Dependence with Rehabilitation Therapy); MS-LTC-DRG 896 (Alcohol, Drug Abuse or Dependence without Rehabilitation Therapy with MCC); MS-LTC-DRG 897 (Alcohol, Drug Abuse or Dependence without Rehabilitation Therapy without MCC); MS-LTC-DRG 945 (Rehabilitation with CC/MCC); and MS-LTC-DRG 946 (Rehabilitation without CC/MCC). We are establishing a relative weight of 0.0000 for these 15 “psychiatric or rehabilitation” MS-LTC-DRGs because the blended payment rate and temporary exceptions to the site neutral payment rate would not be applicable for any LTCH discharges occurring in FY 2025, and as such payment under the LTCH PPS would be no longer be made in part based on the LTCH PPS standard Federal payment rate for any discharges assigned to those MS-LTC-DRGs.
Step 9—Budget neutralize the uncapped relative weights.
In accordance with the regulations at § 412.517(b) (in conjunction with § 412.503), the annual update to the MS-LTC-DRG classifications and relative weights is done in a budget neutral manner such that estimated aggregate LTCH PPS payments would be unaffected, that is, would be neither greater than nor less than the estimated aggregate LTCH PPS payments that would have been made without the MS-LTC-DRG classification and relative weight changes. (For a detailed discussion on the establishment of the budget neutrality requirement for the annual update of the MS-LTC-DRG classifications and relative weights, we refer readers to the RY 2008 LTCH PPS final rule ( 72 FR 26881 and 26882 ).
To achieve budget neutrality under the requirement at § 412.517(b), under our established methodology, for each annual update the MS-LTC-DRG relative weights are uniformly adjusted to ensure that estimated aggregate payments under the LTCH PPS would not be affected (that is, decreased or increased). Consistent with that provision, as we proposed, we continued to apply budget neutrality adjustments in determining the FY 2025 MS-LTC-DRG relative weights so that our update of the MS-LTC-DRG classifications and relative weights for FY 2025 are made in a budget neutral manner. For FY 2025, as we proposed, we applied two budget neutrality factors to determine the MS-LTC-DRG relative weights. In this step, we describe the determination of the budget neutrality adjustment that accounts for the update of the MS-LTC-DRG classifications and relative weights prior to the application of the ten-percent cap. In steps 10 and 11, we describe the application of the 10-percent cap policy (step 10) and the determination of the budget neutrality factor that accounts for the application of the 10-percent cap policy (step 11).
In this final rule, to ensure budget neutrality for the update to the MS-LTC-DRG classifications and relative weights prior to the application of the 10-percent cap (that is, uncapped relative weights), under § 412.517(b), we continued to use our established two-step budget neutrality methodology. Therefore, in the first step of our MS-LTC-DRG update budget neutrality methodology, for FY 2025, we calculated and applied a normalization factor to the recalibrated relative weights (the result of Steps 1 through 8 discussed previously) to ensure that estimated payments are not affected by changes in the composition of case types or the changes to the classification system. That is, the normalization adjustment is intended to ensure that the recalibration of the MS-LTC-DRG relative weights (that is, the process itself) neither increases nor decreases the average case-mix index.
To calculate the normalization factor for FY 2025, we used the following three steps: (1.a.) use the applicable LTCH cases from the best available data (that is, LTCH discharges from the FY 2023 MedPAR file) and group them ( print page 69430) using the FY 2025 GROUPER (that is, Version 42 for FY 2025) and the recalibrated FY 2025 MS-LTC-DRG uncapped relative weights (determined in Steps 1 through 8 discussed previously) to calculate the average case-mix index; (1.b.) group the same applicable LTCH cases (as are used in Step 1.a.) using the FY 2024 GROUPER (Version 41) and FY 2024 MS-LTC-DRG relative weights in Table 11 of the FY 2024 IPPS/LTCH PPS final rule and calculate the average case-mix index; and (1.c.) compute the ratio of these average case-mix indexes by dividing the average case-mix index for FY 2024 (determined in Step 1.b.) by the average case-mix index for FY 2025 (determined in Step 1.a.). As a result, in determining the MS-LTC-DRG relative weights for FY 2025, each recalibrated MS-LTC-DRG uncapped relative weight is multiplied by the normalization factor of 1.27408 (determined in Step 1.c.) in the first step of the budget neutrality methodology, which produces “normalized relative weights.”
In the second step of our MS-LTC-DRG update budget neutrality methodology, we calculated a budget neutrality adjustment factor consisting of the ratio of estimated aggregate FY 2025 LTCH PPS standard Federal payment rate payments for applicable LTCH cases before reclassification and recalibration to estimated aggregate payments for FY 2025 LTCH PPS standard Federal payment rate payments for applicable LTCH cases after reclassification and recalibration. That is, for this final rule, for FY 2025, we determined the budget neutrality adjustment factor using the following three steps: (2.a.) simulate estimated total FY 2025 LTCH PPS standard Federal payment rate payments for applicable LTCH cases using the uncapped normalized relative weights for FY 2025 and GROUPER Version 42; (2.b.) simulate estimated total FY 2025 LTCH PPS standard Federal payment rate payments for applicable LTCH cases using the FY 2024 GROUPER (Version 41) and the FY 2024 MS-LTC-DRG relative weights in Table 11 of the FY 2024 IPPS/LTCH PPS final rule; and (2.c.) calculate the ratio of these estimated total payments by dividing the value determined in Step 2.b. by the value determined in Step 2.a. In determining the FY 2025 MS-LTC-DRG relative weights, each uncapped normalized relative weight is then multiplied by a budget neutrality factor of 0.9885836 (the value determined in Step 2.c.) in the second step of the budget neutrality methodology.
Step 10—Apply the 10-percent cap to decreases in MS-LTC-DRG relative weights.
To mitigate the financial impacts of significant year-to-year reductions in MS-LTC-DRGs relative weights, beginning in FY 2023, we adopted a policy that applies, in a budget neutral manner, a 10-percent cap on annual relative weight decreases for MS-LTC-DRGs with at least 25 applicable LTCH cases (§ 412.515(b)). Under this policy, in cases where CMS creates new MS-LTC-DRGs or modifies the MS-LTC-DRGs as part of its annual reclassifications resulting in renumbering of one or more MS-LTC-DRGs, the 10-percent cap does not apply to the relative weight for any new or renumbered MS-LTC-DRGs for the fiscal year. We refer readers to section VIII.B.3.b. of the preamble of the FY 2023 IPPS/LTCH PPS final rule with comment period for a detailed discussion on the adoption of the 10-percent cap policy ( 87 FR 49152 through 49154 ).
Applying the 10-percent cap to MS-LTC-DRGs with 25 or more cases results in more predictable and stable MS-LTC-DRG relative weights from year to year, especially for high-volume MS-LTC-DRGs that generally have the largest financial impact on an LTCH's operations. For this final rule, in cases where the relative weight for a MS-LTC-DRG with 25 or more applicable LTCH cases would decrease by more than 10-percent in FY 2025 relative to FY 2024, as we proposed, we limited the reduction to 10-percent. Under this policy, we do not apply the 10 percent cap to the low-volume MS-LTC-DRGs identified in Step 3 or the no-volume MS-LTC-DRGs identified in Step 8.
Therefore, in this step, for each FY 2025 MS-LTC-DRG with 25 or more applicable LTCH cases (excludes low-volume and zero-volume MS-LTC-DRGs) we compared its FY 2025 relative weight (after application of the normalization and budget neutrality factors determined in Step 9), to its FY 2024 MS-LTC-DRG relative weight. For any MS-LTC-DRG where the FY 2025 relative weight would otherwise have declined more than 10 percent, we established a capped FY 2025 MS-LTC-DRG relative weight that is equal to 90 percent of that MS-LTC-DRG's FY 2024 relative weight (that is, we set the FY 2025 relative weight equal to the FY 2024 weight × 0.90).
In section II.E. of the preamble of this final rule, we discuss our changes to the MS-DRGs, and by extension the MS-LTC-DRGs, for FY 2025. As discussed previously, under our current policy, the 10-percent cap does not apply to the relative weight for any new or renumbered MS-LTC-DRGs. We did not propose any changes to this policy for FY 2025, and as such any new or renumbered MS-LTC-DRGs for FY 2025 were not eligible for the 10-percent cap.
Step 11—Budget neutralize application of the 10-percent cap policy.
Under the requirement at existing § 412.517(b) that aggregate LTCH PPS payments will be unaffected by annual changes to the MS-LTC-DRG classifications and relative weights, consistent with our established methodology, we continued to apply a budget neutrality adjustment to the MS-LTC-DRG relative weights so that the 10-percent cap on relative weight reductions (step 10) is implemented in a budget neutral manner. Therefore, we determined the budget neutrality adjustment factor for the 10-percent cap on relative weight reductions using the following three steps: (a) simulate estimated total FY 2025 LTCH PPS standard Federal payment rate payments for applicable LTCH cases using the capped relative weights for FY 2025 (determined in Step 10) and GROUPER Version 42; (b) simulate estimated total FY 2025 LTCH PPS standard Federal payment rate payments for applicable LTCH cases using the uncapped relative weights for FY 2025 (determined in Step 9) and GROUPER Version 42; and (c) calculate the ratio of these estimated total payments by dividing the value determined in step (b) by the value determined in step (a). In determining the FY 2025 MS-LTC-DRG relative weights, each capped relative weight is then multiplied by a budget neutrality factor of 0.9945741 (the value determined in step (c)) to achieve the budget neutrality requirement.
Table 11, which is listed in section VI. of the Addendum to this final rule and is available via the internet on the CMS website, lists the MS-LTC-DRGs and their respective relative weights, geometric mean length of stay, and five-sixths of the geometric mean length of stay (used to identify SSO cases under § 412.529(a)) for FY 2025. We also are making available on the website the MS-LTC-DRG relative weights prior to the application of the 10 percent cap on MS-LTC-DRG relative weight reductions and corresponding cap budget neutrality factor. ( print page 69431)
The basic methodology for determining LTCH PPS standard Federal payment rates is currently set forth at 42 CFR 412.515 through 412.533 and 412.535 . In this section, we discuss the factors that we use to update the LTCH PPS standard Federal payment rate for FY 2025, that is, effective for LTCH discharges occurring on or after October 1, 2024, through September 30, 2025. Under the dual rate LTCH PPS payment structure required by statute, beginning with discharges in cost reporting periods beginning in FY 2016, only LTCH discharges that meet the criteria for exclusion from the site neutral payment rate are paid based on the LTCH PPS standard Federal payment rate specified at 42 CFR 412.523 . (For additional details on our finalized policies related to the dual rate LTCH PPS payment structure required by statute, we refer readers to the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49601 through 49623 ).)
Prior to the implementation of the dual payment rate system in FY 2016, all LTCH discharges were paid similarly to those now exempt from the site neutral payment rate. That legacy payment rate was called the standard Federal rate. For details on the development of the initial standard Federal rate for FY 2003, we refer readers to the August 30, 2002 LTCH PPS final rule ( 67 FR 56027 through 56037 ). For subsequent updates to the standard Federal rate from FYs 2003 through 2015, and LTCH PPS standard Federal payment rate from FY 2016 through present, as implemented under 42 CFR 412.523(c)(3) , we refer readers to the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42445 through 42446 ).
In this FY 2025 IPPS/LTCH PPS final rule, we present our policies related to the annual update to the LTCH PPS standard Federal payment rate for FY 2025.
The update to the LTCH PPS standard Federal payment rate for FY 2025 is presented in section V.A. of the Addendum to this final rule. The components of the annual update to the LTCH PPS standard Federal payment rate for FY 2025 are discussed in this section, including the statutory reduction to the annual update for LTCHs that fail to submit quality reporting data for FY 2025 as required by the statute (as discussed in section VIII.C.2.c. of the preamble of this final rule). As we proposed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36267 ), we also made an adjustment to the LTCH PPS standard Federal payment rate to account for the estimated effect of the changes to the area wage level for FY 2025 on estimated aggregate LTCH PPS payments, in accordance with 42 CFR 412.523(d)(4) (as discussed in section V.B. of the Addendum to this final rule).
Historically, the Medicare program has used a market basket to account for input price increases in the services furnished by providers. The market basket used for the LTCH PPS includes both operating and capital-related costs of LTCHs because the LTCH PPS uses a single payment rate for both operating and capital-related costs. We adopted the 2017-based LTCH market basket for use under the LTCH PPS beginning in FY 2021 ( 85 FR 58907 through 58909 ). As discussed in section VIII.D. of the preamble of this final rule, we are finalizing our proposal to rebase and revise the 2017-based LTCH market basket to reflect a 2022 base year. For additional details on the historical development of the market basket used under the LTCH PPS, we refer readers to the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53467 through 53476 ), and for a complete discussion of the LTCH market basket and a description of the methodologies used to determine the operating and capital-related portions of the 2017-based LTCH market basket, we refer readers to the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58909 through 58926 ).
Section 3401(c) of the Affordable Care Act provides for certain adjustments to any annual update to the LTCH PPS standard Federal payment rate and refers to the timeframes associated with such adjustments as a “rate year.” We note that, because the annual update to the LTCH PPS policies, rates, and factors now occurs on October 1, we adopted the term “fiscal year” (FY) rather than “rate year” (RY) under the LTCH PPS beginning October 1, 2010, to conform with the standard definition of the Federal fiscal year (October 1 through September 30) used by other PPSs, such as the IPPS ( 75 FR 50396 through 50397 ). Although the language of sections 3004(a), 3401(c), 10319, and 1105(b) of the Affordable Care Act refers to years 2010 and thereafter under the LTCH PPS as “rate year,” consistent with our change in the terminology used under the LTCH PPS from “rate year” to “fiscal year,” for purposes of clarity, when discussing the annual update for the LTCH PPS standard Federal payment rate, including the provisions of the Affordable Care Act, we use “fiscal year” rather than “rate year” for 2011 and subsequent years.
As previously noted, for FY 2025, we are finalizing our proposal to rebase and revise the 2017-based LTCH market basket to reflect a 2022 base year. The 2022-based LTCH market basket is primarily based on the Medicare cost report data submitted by LTCHs and, therefore, specifically reflects the cost structures of LTCHs. As described in more detail in section VIII.D.1 of the preamble of this final rule, we used data from cost reporting periods beginning on and after April 1, 2021, and prior to April 1, 2022 because these data reflect the most recent information that are most representative of FY 2022. We believe that the 2022-based LTCH market basket appropriately reflects the cost structure of LTCHs, as discussed in greater detail in section VIII.D. of the preamble of this final rule. Therefore, in this final rule, as we proposed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36267 ), we use the 2022-based LTCH market basket to update the LTCH PPS standard Federal payment rate for FY 2025.
Section 1886(m)(3)(A) of the Act provides that, beginning in FY 2010, any annual update to the LTCH PPS standard Federal payment rate is reduced by the adjustments specified in clauses (i) and (ii) of subparagraph (A), as applicable. Clause (i) of section 1886(m)(3)(A) of the Act provides for a reduction, for FY 2012 and each subsequent rate year, by “the productivity adjustment” described in section 1886(b)(3)(B)(xi)(II) of the Act. Section 1886(b)(3)(B)(xi)(II) of the Act, as added by section 3401(a) of the Affordable Care Act, defines this productivity adjustment as equal to the 10-year moving average of changes in annual economy-wide, private nonfarm business multifactor productivity (as projected by the Secretary for the 10-year period ending with the applicable fiscal year, year, cost reporting period, or other annual period). The U.S. Department of Labor's Bureau of Labor Statistics (BLS) publishes the official measures of private nonfarm business productivity for the U.S. economy. We note that previously the productivity measure referenced in section 1886(b)(3)(B)(xi)(II) was published by ( print page 69432) BLS as private nonfarm business multifactor productivity. Beginning with the November 18, 2021 release of productivity data, BLS replaced the term multifactor productivity with total factor productivity (TFP). BLS noted that this is a change in terminology only and will not affect the data or methodology. As a result of the BLS name change, the productivity measure referenced in section 1886(b)(3)(B)(xi)(II) is now published by BLS as private nonfarm business total factor productivity. However, as mentioned, the data and methods are unchanged. Please see www.bls.gov for the BLS historical published TFP data. A complete description of IGI's TFP projection methodology is available on the CMS website at https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-research-and-information . Clause (ii) of section 1886(m)(3)(A) of the Act provided for a reduction, for each of FYs 2010 through 2019, by the “other adjustment” described in section 1886(m)(4)(F) of the Act; therefore, it is not applicable for FY 2025.
Section 1886(m)(3)(B) of the Act provides that the application of paragraph (3) of section 1886(m) of the Act may result in the annual update being less than zero for a rate year, and may result in payment rates for a rate year being less than such payment rates for the preceding rate year.
In accordance with section 1886(m)(5) of the Act, the Secretary established the Long-Term Care Hospital Quality Reporting Program (LTCH QRP). The reduction in the annual update to the LTCH PPS standard Federal payment rate for failure to report quality data under the LTCH QRP for FY 2014 and subsequent fiscal years is codified under 42 CFR 412.523(c)(4) . The LTCH QRP, as required for FY 2014 and subsequent fiscal years by section 1886(m)(5)(A)(i) of the Act, requires that a 2.0 percentage points reduction be applied to any update under 42 CFR 412.523(c)(3) for an LTCH that does not submit quality reporting data to the Secretary in accordance with section 1886(m)(5)(C) of the Act with respect to such a year (that is, in the form and manner and at the time specified by the Secretary under the LTCH QRP) ( 42 CFR 412.523(c)(4)(i) ). Section 1886(m)(5)(A)(ii) of the Act provides that the application of the 2.0 percentage points reduction may result in an annual update that is less than 0.0 for a year, and may result in LTCH PPS payment rates for a year being less than such LTCH PPS payment rates for the preceding year. Furthermore, section 1886(m)(5)(B) of the Act specifies that the 2.0 percentage points reduction is applied in a noncumulative manner, such that any reduction made under section 1886(m)(5)(A) of the Act shall apply only with respect to the year involved and shall not be taken into account in computing the LTCH PPS payment amount for a subsequent year. These requirements are codified in the regulations at 42 CFR 412.523(c)(4) . (For additional information on the history of the LTCH QRP, including the statutory authority and the selected measures, we refer readers to section IX. of the preamble of this final rule.)
Consistent with our historical practice, we estimate the market basket percentage increase and the productivity adjustment based on IHS Global Inc.'s (IGI's) forecast using the most recent available data. Based on IGI's fourth quarter 2023 forecast, the proposed FY 2025 market basket percentage increase for the LTCH PPS using the proposed 2022-based LTCH market basket was 3.2 percent. The proposed productivity adjustment for FY 2025 based on IGI's fourth quarter 2023 forecast was 0.4 percentage point.
For FY 2025, section 1886(m)(3)(A)(i) of the Act requires that any annual update to the LTCH PPS standard Federal payment rate be reduced by the productivity adjustment, described in section 1886(b)(3)(B)(xi)(II) of the Act. Consistent with the statute, we proposed to reduce the FY 2025 market basket percentage increase by the FY 2025 productivity adjustment. To determine the proposed market basket update for LTCHs for FY 2025 we subtracted the proposed FY 2025 productivity adjustment from the proposed FY 2025 market basket percentage increase. (For additional details on our established methodology for adjusting the market basket percentage increase by the productivity adjustment, we refer readers to the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51771 ).) In addition, for FY 2025, section 1886(m)(5) of the Act requires that, for LTCHs that do not submit quality reporting data as required under the LTCH QRP, any annual update to an LTCH PPS standard Federal payment rate, after application of the adjustments required by section 1886(m)(3) of the Act, shall be further reduced by 2.0 percentage points.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36268 ), in accordance with the statute, we proposed to reduce the proposed FY 2025 market basket percentage increase of 3.2 percent (based on IGI's fourth quarter 2023 forecast of the proposed 2022-based LTCH market basket) by the proposed FY 2025 productivity adjustment of 0.4 percentage point (based on IGI's fourth quarter 2023 forecast). Therefore, under the authority of section 123 of the BBRA as amended by section 307(b) of the BIPA, consistent with 42 CFR 412.523(c)(3)(xvii) , we proposed to establish an annual market basket update to the LTCH PPS standard Federal payment rate for FY 2025 of 2.8 percent (that is, the proposed LTCH PPS market basket percentage increase of 3.2 percent less the proposed productivity adjustment of 0.4 percentage point). For LTCHs that fail to submit quality reporting data under the LTCH QRP, under 42 CFR 412.523(c)(3)(xvii) in conjunction with 42 CFR 412.523(c)(4) , we proposed to further reduce the annual update to the LTCH PPS standard Federal payment rate by 2.0 percentage points, in accordance with section 1886(m)(5) of the Act. Accordingly, we proposed to establish an annual update to the LTCH PPS standard Federal payment rate of 0.8 percent (that is, the proposed 2.8 percent LTCH market basket update minus 2.0 percentage points) for FY 2025 for LTCHs that fail to submit quality reporting data as required under the LTCH QRP. Consistent with our historical practice, we proposed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36268 ) to use a more recent estimate of the market basket percentage increase and the productivity adjustment, if appropriate, to establish an annual update to the LTCH PPS standard Federal payment rate for FY 2025 in the final rule. We note that, consistent with historical practice, we also proposed to adjust the FY 2025 LTCH PPS standard Federal payment rate by an area wage level budget neutrality factor in accordance with 42 CFR 412.523(d)(4) (as discussed in section V.B.5. of the Addendum to the proposed rule).
Comment: Several commenters stated that the proposed LTCH PPS payment update is inadequate given that inflationary pressures persist and LTCHs are experiencing higher costs for items such as labor, medical supplies, drugs, cybersecurity, administrative burdens, and other operational costs. Commenters stated that the proposed payment increase falls below economywide inflation over the past ( print page 69433) year (3.5 percent) and below what Medicare Advantage plans will receive for 2025 (3.7 percent). A few commenters stated concerns regarding access to care for Medicare beneficiaries treated in LTCHs given the inadequate proposed update for FY 2025. Other challenges cited by commenters included the impact of the implementation of the LTCH dual-rate payment system and other market dynamics, including declines in patient volume, concentration of LTCH cases in fewer payment groupings, the growth in Medicare Advantage, and the resulting worsening financial situation.
A commenter urged CMS to consider the effects of changing health care system dynamics and the unlikelihood of these dynamics returning to “normal” trends, which they stated are straining and will continue to strain hospitals and health systems. For example, commenters cited the disruption to the health care system from the cyberattack on Change Healthcare. The commenter urged CMS to focus on appropriately accounting for recent and future trends in inflationary pressures and cost increases in the hospital payment update, stating it is essential to ensure that Medicare payments for acute care services more accurately reflect the cost of providing hospital care.
Commenters stated that labor costs, especially for clinicians, are continuing to increase at rates that are faster than what CMS factored into the proposed market basket update. A commenter claimed that CMS did not fully account for the increased labor costs that LTCHs are bearing and stated it is important that CMS modify its customary LTCH PPS rate setting methodology to account for the effects of these unprecedented labor costs on the cost to care for Medicare beneficiaries in LTCHs. The commenter stated that its labor costs (for both employed and contract labor) have been increasing at high rates. The commenter stated that increases in its compensation and total operating expenses have been significantly higher than the market basket increases from FY 2020 through FY 2022. The commenter stated that these data as well as other studies and reports highlight the need for additional increases in payments to cover the significant increases in costs that LTCHs have been experiencing. A commenter requested that CMS provide for a “special” increase to the proposed market basket update to account for significantly higher labor and supply costs incurred by LTCHs in recent years and in FY 2025.
A commenter stated that the authorizing statutes for the LTCH PPS do not require or mention the use of an index for an annual update, therefore, CMS is not restrained by the use of the IGI price proxy forecasts or any index with similar data for an annual LTCH PPS rate update. In addition, the commenter noted the broad LTCH PPS authority in the statute to account for circumstances like higher labor and supply costs, stating that Congress would not have included this language in the statute if it did not expect CMS to make such adjustments when appropriate. According to the commenter it is entirely appropriate for CMS to use this broad authority to increase the market basket update to account for high labor and supply costs after the end of the COVID-19 pandemic that LTCHs continue to experience.
Commenters stated that the cumulative impact of inflationary pressure coupled with the proposed low Medicare payment increases for FY 2025 will continue to have negative effects on LTCH PPS operating margins. The commenters urged CMS to use more current data that includes the recent inflationary increases in cost. In the absence of such data, they requested that CMS consider an alternative approach to better align the market basket increases with the rising cost of treating patients.
Response: CMS has historically used a market basket to account for input price increases in the services furnished by fee-for-service providers. Since the inception of the LTCH PPS, the LTCH PPS standard Federal payment rates (with the exception of statutorily mandated updates) have been updated based on a projection of a market basket percentage increase. The LTCH market basket (as well as other CMS market baskets) is a fixed-weight, Laspeyres type index that measures price changes over time and does not reflect increases in costs associated with changes in the volume or intensity of input goods and services until the index is rebased. As such, the LTCH market basket update reflects the prospective price pressures described by the commenters as increasing during a high inflation period (such as faster wage growth or higher energy prices), but does not inherently reflect other factors that might increase the level of costs, such as the quantity of labor used. However, the impact of changes in quantity or use of services on the market basket cost weights are captured when the market basket is rebased.
As discussed in section VIII. D. of this final rule, after consideration of public comments, we are finalizing our proposal to rebase and revise the LTCH market basket to reflect a 2022 base year. We appreciate the commenters' concern regarding inflationary pressure, including labor and supply costs, encountered by LTCHs. The compensation cost weight in the 2022-based LTCH market basket is 8.6 percentage points higher than the compensation cost weight in the 2017-based LTCH market basket, reflecting the faster labor cost growth relative to other input costs as noted by the commenters. We note that the market basket percentage increase is a forecast of the price pressures that LTCHs are expected to face in FY 2025, and the final FY 2025 LTCH market basket percentage increase reflects IGI's (a nationally recognized economic and financial forecasting firm with which CMS contracts to forecast the price proxies of the market baskets) projected inflation and overall economic outlook. We also note that when developing its forecast for the ECI for hospital workers, IGI considers overall labor market conditions (including rise in contract labor employment due to tight labor market conditions) as well as trends in contract labor wages, both of which could potentially impact wages for workers employed directly by the hospital. As projected by IGI and other independent forecasters, compensation growth and upward price pressures are expected to slow in FY 2025 relative to FY 2023 and FY 2024.
As is our general practice, we proposed that if more recent data became available, we would use such data, if appropriate, to derive the final FY 2025 LTCH market basket update for the final rule. For this final rule, we are using an updated forecast of the price proxies underlying the market basket that incorporates more recent historical data and reflects a revised outlook regarding the U.S. economy, including compensation and inflationary pressures. Based on IGI's second quarter 2024 forecast with historical data through the first quarter of 2024, the projected 2022-based LTCH market basket percentage increase factor for FY 2025 is 3.5 percent, which is 0.3 percentage point higher than the projected FY 2025 LTCH market basket percentage increase factor in the proposed rule, and reflects a projected increase in compensation prices of 4.0 percent. As discussed earlier, we believe the LTCH market basket percentage increase appropriately reflects the input price growth (including compensation price growth) that LTCHs incur in providing medical services. We would note that the 10-year historical average (2014-2023) growth rate of the 2022-based LTCH market basket is 2.8 percent ( print page 69434) with compensation prices increasing 2.9 percent. For these reasons, as discussed previously, we believe the LTCH market basket is methodologically sound and uses the best available data for FY 2025. Therefore, we disagree with the commenters that CMS should increase the market basket update or apply a “special” payment adjustment to the LTCH PPS rates to account for or offset higher labor and supply costs or unprecedented inflation.
Comment: Some commenters stated that since the COVID-19 PHE, IGI's forecasted growth for the LTCH market basket has shown a consistent trend of under-forecasting actual market basket growth. They stated they were cognizant of the fact that forecasts will always be imperfect, but the commenters claimed that in the past, they have been more balanced. However, with four straight years of under-forecasts, the commenters were concerned that there is a more systemic issue with IGI's forecasting. Many commenters stated that the missed forecasts have resulted in significant and permanent underpayments to LTCHs, through direct Medicare payments and through influence on other payers, and have improperly allowed Medicare to underpay LTCHs for the costs to care for Medicare beneficiaries for FY 2021 through FY 2024.
Many commenters urged CMS to use the broad LTCH PPS statutory authority to implement forecasting error adjustments in FY 2025 to account for the differences in the market basket forecasts and actual increases since FY 2021, based on the most recent data. Some commenters stated that adopting a one-time forecast error adjustment is necessary to address unprecedented circumstances surrounding the COVID-19 PHE. One commenter urged CMS to increase the market basket whenever CMS determines that the actual market basket percentage increase exceeds the forecasted market basket percentage increase. A few commenters noted that CMS has made forecasting error adjustments for payments to other types of health care providers in the past, stating it would be appropriate to do so for LTCHs as well. Other commenters claimed that CMS wrongly dismissed the option of applying a special payment adjustment to the LTCH PPS rates that accounts for forecast errors in the FY 2023 IPPS/LTCH PPS final rule and FY 2024 IPPS/LTCH PPS final rule leading to underpayments for LTCHs. These commenters stated that a correction to the FY 2025 payment rate is required to prevent underpayments to LTCHs going forward.
Commenters specifically requested that a forecast error adjustment of 4.3 percentage points be added to the FY 2025 annual update to account for the combined understatement of the FY 2021 through FY 2023 LTCH market baskets. A few commenters also requested that, in addition to that estimated 4.3 percentage points forecast error adjustment, CMS also add an unspecified additional amount to compensate LTCHs for four years of underpayments. Other commenters requested that CMS add 3.0 percentage points to the FY 2025 annual update to account for the difference between the market basket update that was implemented for FY 2022 and the actual market basket for FY 2022. One commenter requested that this FY 2022 forecast error adjustment be applied retroactively to the FY 2024 update.
Response: In responding to similar comments in the FY 2023 and FY 2024 IPPS/LTCH PPS final rules ( 87 FR 49165 , 88 FR 59136 ), we explained that under the law, the LTCH PPS is a per-discharge prospective payment system that uses a market basket percentage increase to set the annual update prospectively. This means that the update relies on a mix of both historical data for part of the period for which the update is calculated and forecasted data for the remainder. (For instance, the 2022-based LTCH market basket growth rate for FY 2025 in this final rule is based on IGI's second quarter 2024 forecast with historical data through the first quarter of 2024.) While there is currently no mechanism to adjust for market basket forecast error in the LTCH PPS payment update, the forecast error for a market basket update is equal to the actual market basket percentage increase for a given year less the forecasted market basket percentage increase. Due to the uncertainty regarding future price trends, forecast errors can be both positive and negative.
While the projected LTCH basket updates for FY 2021 through FY 2023 were under forecast (actual increases less forecasted increases were positive), this was largely due to unanticipated inflation and labor market pressures as the economy emerged from the COVID-19 PHE. However, an analysis of the forecast error of the LTCH market basket over a longer period of time shows the forecast error has been both positive and negative. The 10-year cumulative forecast error for FY 2014 to FY 2023 (excluding 2018 as the update was statutorily mandated) is +0.7 percent. In addition, for each fiscal year from 2012 through 2020, the forecasted LTCH market basket update implemented in the final rule was shown to be higher than the actual LTCH market basket update once historical data were available. Only considering the forecast error for years when the final LTCH market basket update is lower than the actual LTCH market basket update would not adequately reflect the full impact of forecast error over the past 10 years. For these reasons, we are not adopting the commenters' requests to implement an adjustment for FY 2025 to account for the difference between the actual and forecasted LTCH market basket updates for FYs 2021 through 2023, and, for the reasons stated previously, we disagree that we wrongly dismissed commenters' requests to apply an adjustment that accounts for forecast errors in the FY 2023 and FY 2024 IPPS/LTCH PPS final rules.
Comment: A commenter stated it appreciated CMS' proposal to increase the market basket update by 3.2 percent for FY 2025. However, the commenter believed that LTCHs should be provided the full 3.2 percent amount without a productivity adjustment. Several commenters stated that CMS should at least temporarily suspend the productivity adjustment due to declines in hospital productivity. A commenter requested that CMS provide more transparency about how the productivity adjustment is calculated. The commenter stated that besides CMS stating that it estimates the productivity adjustment based on IGI's forecast using the most recent available data, CMS does not provide any more information about the data or how it used the data to calculate a 0.4 percentage point reduction to the market basket update. Some commenters urged CMS to eliminate the productivity adjustment for FY 2025.
A commenter stated that the private nonfarm business TFP is intended to allow for productivity gains resulting from new technologies, economies of scale and changes in production, but because the factor is a 10-year moving average, the status of the current workforce is not appropriately reflected. The commenter further stated that hospitals continue to encounter staffing difficulties, such as obtaining nurses and nursing assistants to care for patients and actions to regulate staffing, which will lead to less efficiency, increased costs and recruitment difficulties and should be accounted for when determining a productivity factor. This commenter and other commenters urged CMS to use its broad LTCH PPS statutory authority to eliminate the productivity adjustment for FY 2025, particularly in light of inadequate market basket increases. ( print page 69435)
Response: As set forth in section 1886(b)(3)(B)(xi) of the Act, the FY 2025 productivity adjustment is derived based on the 10-year moving average growth in economy-wide productivity for the period ending in FY 2025. We recognize the concerns of the commenters regarding the appropriateness of the productivity adjustment; however, as we explained in response to similar comments in the FY 2023 and FY 2024 IPPS/LTCH PPS final rules, section 1886(m)(3)(A)(i) of the Act requires the application of the specific productivity adjustment described in section 1886(b)(3)(B)(xi) of the Act.
As stated in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36267 ), BLS publishes the official measures of annual economy-wide, private nonfarm business total factor productivity (TFP) (previously referred to as annual economy-wide, private nonfarm business multifactor productivity). IGI forecasts TFP consistent with BLS methodology by forecasting the detailed components of TFP. A complete description of IGI's TFP projection methodology is available on the CMS website at https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-research-and-information . We believe our methodology for the productivity adjustment is consistent with section 1886(b)(3)(B)(xi) of the Act which states that the productivity adjustment is equal to the 10-year moving average of changes in annual economy-wide private nonfarm business multi-factor productivity (as projected by the Secretary for the 10-year period ending with the applicable fiscal year, year, cost reporting period, or other annual period).
The FY 2025 proposed productivity adjustment of 0.4 percent was based on IGI's forecast of the 10-year moving average of annual economy-wide private nonfarm business TFP, reflecting historical data through 2022 as published by BLS and forecasted TFP growth for 2023 through 2025. The FY 2025 final productivity adjustment of 0.5 percent is based on IGI's forecast of the 10-year moving average of annual economy-wide private nonfarm business TFP, reflecting historical data through 2023 as published by BLS, and forecasted TFP growth for 2024 through 2025.
In response to commenters' request for more transparency regarding the productivity adjustment calculation, we have provided the following information on the CMS website and in the Federal Register regarding the general method for calculating the productivity adjustment. As stated in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36267 ), the most recent BLS historical TFP data can be downloaded from the BLS website at https://www.bls.gov/productivity . This allows interested parties to obtain TFP annual index levels for 1987 through 2023. The IGI projection model as described on the CMS website ( https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-research-and-information ) is then used to derive annual TFP growth rates for 2024 and 2025, which are then applied to the historical BLS levels to obtain a projection of index levels for 2024 and 2025. As further described in the documentation on the CMS website at https://www.cms.gov/research-statistics-data-and-systems/statistics-trends-and-reports/medicareprogramratesstats/downloads/tfp_methodology.pdf , these annual index levels are then interpolated to quarterly levels. The FY 2025 productivity adjustment is equal to the percent change in the 40-quarter moving average projected level for the period ending September 30, 2025 relative to the 40-quarter moving average projected level for the period ending September 30, 2024. If there are specific questions regarding the methodology for deriving the productivity adjustment, the public may email CMS at the email address provided on the CMS website ( [email protected] ) to request clarification or more information on the market baskets and productivity adjustment calculations.
After consideration of public comments, we are finalizing the LTCH PPS payment rate update using the most recent forecast of the 2022-based LTCH market basket percentage increase and productivity adjustment. As such, based on IGI's second quarter 2024 forecast, the FY 2025 market basket percentage increase for the LTCH PPS using the 2022-based LTCH market basket is 3.5 percent. The current estimate of the productivity adjustment for FY 2025 based on IGI's second quarter 2024 forecast is 0.5 percentage point. Therefore, under the authority of section 123 of the BBRA as amended by section 307(b) of the BIPA, consistent with 42 CFR 412.523(c)(3)(xvii) , we are establishing an annual market basket update to the LTCH PPS standard Federal payment rate for FY 2025 of 3.0 percent (that is, the most recent estimate of the LTCH PPS market basket percentage increase of 3.5 percent less the productivity adjustment of 0.5 percentage point). For LTCHs that fail to submit quality reporting data under the LTCH QRP, under 42 CFR 412.523(c)(3)(xvii) in conjunction with 42 CFR 412.523(c)(4) , as we proposed, we are further reducing the annual update to the LTCH PPS standard Federal payment rate by 2.0 percentage points, in accordance with section 1886(m)(5) of the Act. Accordingly, we are establishing an annual update to the LTCH PPS standard Federal payment rate of 1.0 percent (that is, the 3.0 percent LTCH market basket update minus 2.0 percentage points) for FY 2025 for LTCHs that fail to submit quality reporting data as required under the LTCH QRP.
The input price index (that is, the market basket) that was used to develop the LTCH PPS for FY 2003 was the “excluded hospital with capital” market basket. That market basket was based on 1997 Medicare cost report data and included data for Medicare-participating IRFs, IPFs, LTCHs, cancer hospitals, and children's hospitals. Although the term “market basket” technically describes the mix of goods and services used in providing hospital care, this term is also commonly used to denote the input price index (that is, cost category weights and price proxies combined) derived from that mix. Accordingly, the term “market basket,” as used in this section, refers to an input price index.
Since the LTCH PPS inception, the market basket used to update LTCH PPS payments has been rebased and revised to reflect more recent data. We last rebased and revised the market basket applicable to the LTCH PPS in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58909 through 58926 ), where we adopted a 2017-based LTCH market basket. References to the historical market baskets used to update LTCH PPS payments are listed in the FY 2021 LTCH PPS final rule ( 85 FR 58909 through 58910 ).
For the FY 2025 IPPS/LTCH proposed rule, we proposed to rebase and revise the 2017-based LTCH market basket to reflect a 2022 base year, which would maintain our historical frequency of rebasing the market basket every 4 years. The proposed 2022-based LTCH market basket is primarily based on Medicare cost report data for LTCHs for FY 2022, specifically for cost reporting periods beginning on and after April 1, 2021, and prior to April 1, 2022. For the 2017-based LTCH market, we used Medicare cost report data for LTCHs from cost reporting periods beginning ( print page 69436) on and after October 1, 2016, and before October 1, 2017, or reports that began in FY 2017. The majority of LTCHs have a cost report begin date of September 1 and so those LTCHs with a cost report begin date of September 1, 2021 have the majority of their expenses occurring in the FY 2022 time period. We proposed to use data from cost reporting periods beginning on and after April 1, 2021, and prior to April 1, 2022 because these data reflected the most recent Medicare cost report data for LTCHs at the time of rulemaking where the majority of their costs are occurring in FY 2022 while still maintaining our historical frequency of rebasing the market basket every 4 years.
At the time of proposed rulemaking, we were unable to use data from the FY 2022 HCRIS file, which reflects cost reporting periods beginning on and after October 1, 2021 and prior to September 30, 2022, as most reporters have a begin date of September 1, so the dataset in the file was not yet complete. In the interest of utilizing the most recent, complete data available, we proposed to combine data from multiple HCRIS files to obtain a 2022 base year. We proposed to use a composite timeframe of cost reporting periods beginning on and after April 1, 2021 and prior to April 1, 2022, because April 1 reflects the middle of the fiscal year and this timeframe would allow data from 2022 to be included in this rebasing. Using this proposed method, the weighted average of costs occurring in FY 2022 (accounting for the distribution of providers by Medicare cost report begin date) is 82 percent. Therefore, we believe our proposed methodology of using Medicare cost report data based on cost reporting periods beginning on or after April 1, 2021 and prior to April 1, 2022 reflects the most recent information that is most representative of FY 2022.
As described in the FY 2023 IPPS/LTCH final rule ( 87 FR 49164 through 49165 ), we received comments on the FY 2023 IPPS/LTCH PPS proposed rule where stakeholders expressed concern that the proposed market basket update was inadequate relative to input price inflation experienced by LTCHs, particularly as a result of the COVID-19 PHE. These commenters stated that the PHE, along with inflation, has significantly driven up operating costs. Specifically, some commenters noted changes to the labor markets that led to the use of more contract labor. As described in more detail later in this section, we verified this trend when analyzing the Medicare cost reports submitted by LTCHs through 2022. Therefore, we believe it is appropriate to incorporate more recent data to reflect updated cost structures for LTCHs, and so we proposed to use 2022 as the base year because we believe that the Medicare cost reports for this year represent the most recent, complete set of Medicare cost report data available for developing the proposed LTCH market basket at the time of this rulemaking. Given the recent trends in the major cost weights derived from the Medicare cost report data as discussed later in this section, we will continue to monitor these data going forward and any additional changes to the LTCH market basket will be proposed in future rulemaking.
In the following discussion, we provide an overview of the proposed LTCH market basket, describe the proposed methodologies for developing the operating and capital portions of the proposed 2022-based LTCH market basket, and provide information on the proposed price proxies. In each section, we describe any comments received, responses to these comments, and our final policies for this final rule. We received the following comments on our proposal to rebase the LTCH market basket to a 2022 base year.
Comment: A few commenters were supportive of CMS rebasing the market basket to reflect a 2022 base year. A commenter cited support for CMS' proposed approach of using Medicare cost report data for LTCHs collected between April 1, 2021 and April 1, 2022 stating that under this proposed approach, a significant portion of FY 2022 data would be included in this rebasing and CMS would be including the most recent and representative data in its rebasing process.
However, a commenter stated that for FY 2025 payment rates, CMS proposed to return to its standard pre-pandemic rate setting methodologies (FY 2023 MedPAR file and FY 2022 HCRIS file) without any discussion of what are the “best available” data for the FY 2025 rate setting. The commenter stated that CMS is making an exception for the revised and rebased LTCH market basket where it proposed to use cost reports for periods beginning on or after April 1, 2021, and prior to April 1, 2022 to obtain a cost report dataset that is representative of FY 2022. The commenter claimed that based on its own experiences and analysis of its LTCH data, it is clear that LTCH utilization during the pandemic was not representative of typical LTCH utilization patterns. The commenter stated that the highest surge of COVID-19 hospitalizations occurred during FY 2022; therefore, CMS erred by not considering this when deciding which data to use for rate setting in FY 2025.
Response: As stated in the FY 2025 IPPS/LTCH proposed rule ( 89 FR 36268 ) and discussed previously in this final rule, the market basket used to update LTCH PPS payments has been periodically rebased and revised over the history of the LTCH PPS to reflect more recent data on LTCH cost structures. It has been our longstanding practice to rebase the market baskets using the most recent data that are available at the time of proposed rulemaking. For the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to rebase and revise the LTCH market basket using 2022 Medicare cost reports, defined as cost reports for periods beginning on or after April 1, 2021, and prior to April 1, 2022. Using this proposed method, the weighted average of costs occurring in FY 2022 (accounting for the distribution of providers by Medicare cost report begin date) is 82 percent. This allowed us to obtain a cost report dataset that was complete and representative of FY 2022, which is the most recent year of complete data available at the time of rulemaking. Data for 2023 are incomplete at this time. The Medicare cost report data showed an increase in the Compensation cost weight from 2017 to 2022, which is consistent with comments received on the FY 2024 IPPS/LTCH proposed rule ( 88 FR 59134 ) that stated the 2017-based LTCH market basket did not sufficiently account for the dramatic increases in labor costs that LTCHs were incurring. As we stated in that rule in response to public comments, we are continually monitoring the trends in the LTCH cost data to ensure the market basket reflects the costs faced by LTCHs in providing care. Thus, we believe it is more appropriate to update the base year cost weights to 2022 to reflect changes over this period rather than to delay the rebasing. It has been our longstanding practice to rebase the market basket on a regular basis to ensure it reflects the input cost structure of LTCHs. We will continue to monitor the Medicare cost report data as they become available and, if appropriate, propose any changes to the LTCH market basket in future rulemaking.
Similar to the 2017-based LTCH market basket, the proposed 2022-based LTCH market basket is a fixed-weight, Laspeyres-type price index. A Laspeyres price index measures the change in price, over time, of the same mix of goods and services purchased in the base period. Any changes in the quantity or mix (that is, intensity) of ( print page 69437) goods and services purchased over time relative to the base period are not measured. The index itself is constructed using three steps. First, a base period is selected (in the proposed rule, we proposed to use 2022 as the base period) and total base period costs are estimated for a set of mutually exclusive and exhaustive spending categories, with the proportion of total costs that each category represents being calculated. These proportions are called cost weights. Second, each cost category is matched to an appropriate price or wage variable, referred to as a “price proxy.” In almost every instance, these price proxies are derived from publicly available statistical series that are published on a consistent schedule (preferably at least on a quarterly basis). Finally, the cost weight for each cost category is multiplied by the level of its respective price proxy. The sum of these products (that is, the cost weights multiplied by their price index levels) for all cost categories yields the composite index level of the market basket in a given period. Repeating this step for other periods produces a series of market basket levels over time. Dividing an index level for a given period by an index level for an earlier period produces a rate of growth in the input price index over that timeframe. As previously noted, the market basket is described as a fixed-weight index because it represents the change in price over time of a constant mix (quantity and intensity) of goods and services needed to furnish hospital services. The effects on total costs resulting from changes in the mix of goods and services purchased subsequent to the base period are not measured. For example, a hospital hiring more nurses to accommodate the needs of patients would increase the volume of goods and services purchased by the hospital but would not be factored into the price change measured by a fixed-weight hospital market basket. Only when the index is rebased would changes in the quantity and intensity be captured, with those changes being reflected in the cost weights. Therefore, we rebase the market basket periodically so that the cost weights reflect recent changes in the mix of goods and services that hospitals purchase to furnish inpatient care between base periods.
We invited public comments on our proposed methodology, discussed in this section of this rule, for deriving the proposed 2022-based LTCH market basket.
The major types of costs underlying the proposed 2022-based LTCH market basket are derived from the Medicare cost reports (CMS Form 2552-10, OMB Control Number 0938-0050) for LTCHs. Specifically, we use the Medicare cost reports for seven specific costs: Wages and Salaries, Employee Benefits, Contract Labor, Pharmaceuticals, Professional Liability Insurance (PLI), Home Office/Related Organization Contract Labor, and Capital. A residual category is then estimated and reflects all remaining costs not captured in the seven types of costs identified previously. The 2017-based LTCH market basket similarly used the Medicare cost reports.
Medicare cost report data include costs for all patients (including but not limited to those covered by Medicare, Medicaid, and private insurance). Because our goal is to measure cost shares for facilities that serve Medicare beneficiaries and are reflective of case mix and practice patterns associated with providing services to Medicare beneficiaries in LTCHs, we proposed to limit our selection of Medicare cost reports to those from LTCHs that have a Medicare average length of stay (LOS) that is within a comparable range of their total facility average LOS. We define the Medicare average LOS based on data reported on the Medicare cost report (CMS Form 2552-10, OMB Control Number 0938-0050) Worksheet S-3, Part I, line 14. We believe that applying the LOS edit results in a more accurate reflection of the structure of costs associated with Medicare covered days as our proposed edit excludes those LTCHs that had an average total facility LOS that were notably different than the average Medicare LOS. For the 2017-based LTCH market basket, we used the cost reports submitted by LTCHs with Medicare average LOS within 25 percent (that is, 25 percent higher or lower) of the total facility average LOS for the hospital. Based on our analysis of the 2022 Medicare cost reports, for the proposed 2022-based LTCH market basket, we proposed to again use the cost reports submitted by LTCHs with Medicare average LOS within 25 percent (that is, 25 percent higher or lower) of the total facility average LOS for the hospital. The universe of LTCHs had an average Medicare LOS of 26 days, an average total facility LOS of 35 days, and aggregate Medicare utilization (as measured by Medicare inpatient LTCH days as a percentage of total facility inpatient LTCH days) of 34 percent in 2022. Applying the proposed trim excludes 11 percent of LTCH providers and results in a subset of LTCH Medicare cost reports with an average Medicare LOS of 26 days, average facility LOS of 30 days, and aggregate Medicare utilization (based on days) of 40 percent. The 11 percent of providers that are excluded had an average Medicare LOS of 29 days, average facility LOS of 71 days, and aggregate Medicare utilization of 14 percent.
We proposed to use the cost reports for LTCHs that meet this requirement to calculate the costs for the seven major cost categories (Wages and Salaries, Employee Benefits, Contract Labor, Professional Liability Insurance, Pharmaceuticals, Home Office/Related Organization Contract Labor, and Capital) for the market basket. Also, as described in section VIII.D.3.d. of the preamble of this final rule, and as done for the 2017-based LTCH market basket, we also proposed to use the Medicare cost report data to calculate the detailed capital cost weights for the Depreciation, Interest, Lease, and Other Capital-Related cost categories.
We proposed to derive Wages and Salaries costs as the sum of routine inpatient salaries, ancillary salaries, and a proportion of overhead (or general service cost center) salaries as reported on Worksheet A, column 1. Because overhead salary costs are attributable to the entire LTCH, we proposed to only include the proportion attributable to the Medicare allowable cost centers. For the 2022-based LTCH market basket, we proposed that routine and ancillary Wages and Salaries costs would be equal to salary costs as reported on Worksheet A, column 1, lines 30 through 35, 50 through 76 (excluding 52 and 75), 90 through 91, and 93. Then, we proposed to estimate the proportion of overhead salaries that are attributed to Medicare allowable costs centers. We proposed to first calculate overhead salaries as the sum of Worksheet A, column 1, lines 4 through 18. We then calculate the “Medicare allowable ratio” equal to routine and ancillary Wages and Salaries divided by total non-overhead salaries (Worksheet A, column 1, line 200 less overhead salaries). We proposed to multiply this Medicare allowable ratio by overhead salaries to determine the overhead salaries attributed to Medicare allowable cost centers. The sum of routine salaries, ancillary salaries, and the estimated Medicare allowable portion of overhead salaries represent Wages and Salaries costs. A similar methodology was used to derive Wages and Salaries costs in the 2017-based LTCH market basket. ( print page 69438)
Similar to the 2017-based LTCH market basket, we proposed to calculate Employee Benefits costs using data from Worksheet S-3, part II, column 4, lines 17, 18, 20, and 22. The completion of Worksheet S-3, part II is only required for IPPS hospitals. For 2022, we found that approximately 42 percent of LTCHs voluntarily reported the Employee Benefits data, which has increased from the approximately 20 percent of LTCHs that reported these data that were used for the 2017-based LTCH market basket. Our analysis of the Worksheet S-3, part II data submitted by these LTCHs indicates that we continue to have a large enough sample to enable us to produce a reasonable Employee Benefits cost weight. Specifically, we found that when we recalculated the cost weight after weighting to reflect the characteristics of the universe of LTCHs (such as by type of ownership—nonprofit, for-profit, and government—and by region), the recalculation did not have a material effect on the resulting cost weight. Therefore, we proposed to use Worksheet S-3, part II data (as was done for the 2017-based LTCH market basket) to calculate the Employee Benefits cost weight in the proposed 2022-based LTCH market basket.
We note that, effective with the implementation of CMS Form 2552-10, OMB Control Number 0938-0050, we began collecting Employee Benefits and Contract Labor data on Worksheet S-3, part V, which is applicable to LTCHs. However, approximately 12 percent of LTCHs reported data on Worksheet S-3, part V for 2022, which has fallen since 2017 when roughly 17 percent of LTCHs reported these data. Because a greater percentage of LTCHs continue to report data on Worksheet S-3, part II than Worksheet S-3, part V, we did not propose to use the Employee Benefits and Contract Labor data reported on Worksheet S-3, part V to calculate the Employee Benefits and Contract Labor cost weights in the proposed 2022-based LTCH market basket. We continue to encourage all providers to report Employee Benefits and Contract Labor data on Worksheet S-3, part V.
Contract Labor costs reported on the Medicare cost reports are primarily associated with direct patient care services. Contract Labor costs for services such as accounting, billing, and legal are estimated using other government data sources as described in this section of this final rule. Approximately 40 percent of LTCHs voluntarily reported Contract Labor costs on Worksheet S-3, part II, which was similar to the percentage obtained from 2017 Medicare cost reports.
As was done for the 2017-based LTCH market basket, we proposed to derive the Contract Labor costs for the proposed 2022-based LTCH market basket using voluntarily reported data from Worksheet S-3, part II. Our analysis of these data indicates that we have a large enough sample to enable us to produce a representative Contract Labor cost weight. Specifically, we found that when we recalculated the cost weight after weighting to reflect the characteristics of the universe of LTCHs by region, the recalculation did not have a material effect on the resulting cost weight. Therefore, we proposed to use data from Worksheet S-3, part II, column 4, lines 11 and 13 to calculate the Contract Labor cost weight in the proposed 2022-based LTCH market basket.
We proposed to calculate Pharmaceuticals costs using non-salary costs reported for the pharmacy cost center (line 15) and drugs charged to patients cost center (line 73). We proposed to calculate these costs as Worksheet A, column 7, less Worksheet A, column 1 for each of these lines. A similar methodology was used for the 2017-based LTCH market basket.
We proposed that Professional Liability Insurance (PLI) costs (often referred to as malpractice costs) be equal to premiums, paid losses and self-insurance costs reported on Worksheet S-2, part I, columns 1 through 3, line 118. A similar methodology was used for the 2017-based LTCH market basket.
We proposed to calculate the Home Office/Related Organization Contract Labor costs using data reported on Worksheet S-3, part II, column 4, lines 1401, 1402, 2550, and 2551 for those LTCH providers reporting total salaries on Worksheet S-3, part II, line 1. A similar methodology was used for the 2017-based LTCH market basket.
We proposed that Capital costs be equal to Medicare allowable capital costs as reported on Worksheet B, part II, column 26, lines 30 through 35, 50 through 76 (excluding 52 and 75), 90 through 91 and 93. A similar methodology was used for the 2017-based LTCH market basket.
After we derive costs for the major cost categories for each provider using the Medicare cost report data as previously described, we proposed to trim the data for outliers. For each of the seven major cost categories, we first proposed to divide the calculated costs for the category by total Medicare allowable costs calculated for the provider to obtain cost weights for the universe of LTCH providers. For the 2022-based LTCH market basket (similar to the approach used for the 2017-based LTCH market basket), we proposed that total Medicare allowable costs would be equal to the total costs as reported on Worksheet B, part I, column 26, lines 30 through 35, 50 through 76 (excluding 52 and 75), 90 through 91, and 93.
For the Wages and Salaries, Employee Benefits, Contract Labor, Pharmaceuticals, Professional Liability Insurance, and Capital cost weights, after excluding cost weights that are less than or equal to zero, we proposed to then remove those providers whose derived cost weights fall in the top and bottom 5 percent of provider specific derived cost weights to ensure the exclusion of outliers. We note that missing values are assumed to be zero consistent with the methodology for how missing values were treated in the 2017-based LTCH market basket. After the outliers have been excluded, we sum the costs for each category across all remaining providers. We proposed to divide this by the sum of total Medicare allowable costs across all remaining providers to obtain a cost weight for the 2022-based LTCH market basket for the given category. This trimming process is done for each cost weight separately.
For the Home Office/Related Organization Contract Labor cost weight, we proposed to apply a 1-percent top only trimming methodology. We believe, as the Medicare cost report data (Worksheet S-2, part I, line 140) indicate, that not all LTCHs have a home office. LTCHs without a home office can incur these expenses directly by having their own staff, for which the costs would be included in the Wages and Salaries and Employee Benefits cost weights. Alternatively, LTCHs without a home office could also purchase related services from external contractors for which these expenses would be captured in the residual “All Other” cost weight. We believe this 1-percent top-only trimming methodology is appropriate as it addresses outliers while allowing providers with zero Home Office/Related Organization Contract Labor costs to be included in ( print page 69439) the Home Office/Related Organization Contract Labor cost weight calculation. If we applied both the top and bottom 5 percent trimming methodology, we would exclude providers who have zero Home Office/Related Organization Contract Labor costs.
Finally, we proposed to calculate the residual “All Other” cost weight that reflects all remaining costs that are not captured in the seven cost categories listed.
We did not receive any specific comments on the proposed methodology to derive the major cost weights using the Medicare cost reports and therefore are finalizing this methodology without modification. We note that comments we received on the overall market basket method, transparency of the method, and resulting market basket updates and labor-related share are discussed later in section VIII.D.5 and VIII.D.6 of the preamble of this final rule. We refer readers to Table EEEE 1 for the resulting proposed and final cost weights for these major cost categories.

The Wages and Salaries and Employee Benefits cost weights calculated from the Medicare cost reports for the 2022-based LTCH market basket are similar to the Wages and Salaries and Employee Benefits cost weights for the 2017-based LTCH market basket. The Contract Labor cost weight, however, is approximately 8 percentage points higher than the Contract Labor cost weight in the 2017-based LTCH market basket. The 2022-based Pharmaceuticals and Capital cost weights are lower than the 2017-based LTCH market basket by 1.7 percentage points and 1.4 percentage points, respectively. The 2022-based Home Office/Related Organization Contract Labor cost weight has increased by 1.8 percentage points compared to the 2017-based LTCH market basket.
As we did for the 2017-based LTCH market basket, we proposed to allocate the Contract Labor cost weight to the Wages and Salaries and Employee Benefits cost weights based on their relative proportions under the assumption that Contract Labor costs are comprised of both Wages and Salaries and Employee Benefits. The Contract Labor allocation proportion for Wages and Salaries is equal to the Wages and Salaries cost weight as a percent of the sum of the Wages and Salaries cost weight and the Employee Benefits cost weight. This rounded percentage is 87 percent. Therefore, we proposed to allocate 87 percent of the Contract Labor cost weight to the Wages and Salaries cost weight and 13 percent to the Employee Benefits cost weight. We received no comments on the proposed methodology to allocate the Contract Labor cost weight to the Wages and Salaries cost weight and Employee Benefits cost weight and therefore, are finalizing this methodology without modification. We refer readers to Table EEEE 2 that shows the proposed and final Wages and Salaries and Employee Benefits cost weights after Contract Labor cost weight allocation for both the 2022-based LTCH market basket and the 2017-based LTCH market basket.

After the allocation of the Contract Labor cost weight, the 2022-based Wages and Salaries cost weight is 7.2 percentage points higher and the Employee Benefits cost weight is 1.4 percentage points higher, relative to the respective cost weights for the 2017-based LTCH market basket. As a result, in the 2022-based LTCH market basket, the compensation cost weight is 8.6 percentage points higher than the Compensation cost weight for the 2017-based LTCH market basket. ( print page 69440)
To further divide the residual “All Other” cost weight estimated from the 2022 Medicare cost report data into more detailed cost categories, we proposed to use the 2017 Benchmark I-O “The Use Table (Supply-Use Framework)” data for NAICS 622000, Hospitals, published by the Bureau of Economic Analysis (BEA). These data are publicly available at the following website: https://www.bea.gov/industry/input-output-accounts-data . For the 2017-based LTCH market basket, we used the 2012 Benchmark I-O data, the most recent data available at the time ( 85 FR 58913 ).
The BEA Benchmark I-O data are scheduled for publication every 5 years with the most recent data available for 2017. The 2017 Benchmark I-O data are derived from the 2017 Economic Census and are the building blocks for BEA's economic accounts. Therefore, they represent the most comprehensive and complete set of data on the economic processes or mechanisms by which output is produced and distributed. [ 256 ] BEA also produces Annual I-O estimates. However, while based on a similar methodology, these estimates reflect less comprehensive and less detailed data sources and are subject to revision when benchmark data becomes available. Instead of using the less detailed Annual I-O data, we proposed to inflate the 2017 Benchmark I-O data forward to 2022 by applying the annual price changes from the respective price proxies to the appropriate market basket cost categories that are obtained from the 2017 Benchmark I-O data, and calculated the cost shares that each cost category represents using the inflated data. These resulting 2022 cost shares were applied to the residual “All Other” cost weight to obtain the detailed cost weights for the proposed 2022-based LTCH market basket. For example, the cost for Food: Direct Purchases represents 4.3 percent of the sum of the residual “All Other” 2017 Benchmark I-O Hospital Expenditures inflated to 2022. Therefore, the Food: Direct Purchases cost weight represents 4.3 percent of the proposed 2022-based LTCH market basket's residual “All Other” cost category (20.8 percent), yielding a “final” Food: Direct Purchases proposed cost weight of 0.9 percent in the proposed 2022-based LTCH market basket (0.043 × 20.8 percent = 0.9 percent).
Using this methodology, we proposed to derive seventeen detailed LTCH market basket cost category weights within the proposed 2022-based LTCH market basket residual “All Other” cost weight (20.8 percent). These categories are: (1) Electricity and Other Non-Fuel Utilities; (2) Fuel: Oil and Gas; (3) Food: Direct Purchases; (4) Food: Contract Services; (5) Chemicals; (6) Medical Instruments; (7) Rubber and Plastics; (8) Paper and Printing Products; (9) Miscellaneous Products; (10) Professional Fees: Labor-Related; (11) Administrative and Facilities Support Services; (12) Installation, Maintenance, and Repair Services; (13) All Other Labor-Related Services; (14) Professional Fees: Nonlabor-Related; (15) Financial Services; (16) Telephone Services; and (17) All Other Nonlabor-Related Services. We note that these are the same categories as were used in the 2017-based LTCH market basket (with several cost categories being renamed for clarification purposes).
We did not receive any specific comments on the proposed methodology to derive the detailed operating cost weights and therefore are finalizing this methodology without modification. We note that general comments we received on the resulting market basket cost weights are discussed later in section VIII.D.5 and VIII.D.6 of the preamble of this final rule.
As described in section VIII.D.3.b. of the preamble of this final rule, we proposed a Capital-Related cost weight of 8.5 percent in the proposed 2022-based LTCH market basket as calculated from the 2022 Medicare cost reports for LTCHs after applying the proposed trims as previously described. We proposed to then separate this total Capital-Related cost weight into more detailed cost categories. Using Worksheet A-7 in the 2022 Medicare cost reports, we are able to group capital-related costs into the following categories: Depreciation, Interest, Lease, and Other Capital-Related costs, as shown in Table EEEE 3, which is the same methodology used for the 2017-based LTCH market basket.
We also proposed to allocate lease costs, which are 65 percent of total capital costs in the proposed 2022-based LTCH market basket, across each of the remaining detailed capital-related cost categories as was done in the 2017-based LTCH market basket. This would result in three primary capital-related cost categories in the proposed 2022 based LTCH market basket: Depreciation, Interest, and Other Capital-Related costs. Lease costs are unique in that they are not broken out as a separate cost category in the proposed 2022-based LTCH market basket. Rather, we proposed to proportionally distribute these costs among the cost categories of Depreciation, Interest, and Other Capital-Related, reflecting the assumption that the underlying cost structure of leases is similar to that of capital-related costs in general. As was done for the 2017-based LTCH market basket, we proposed to assume that 10 percent of the lease costs represents overhead and to assign those costs to the Other Capital-Related cost category accordingly. Therefore, we are assuming that approximately 6.5 percent (65.0 percent × 0.1) of total capital-related costs represent lease costs attributable to overhead, and we proposed to add this 6.5 percentage points to the 7.3 percent Other Capital-Related cost category weight. We also proposed to distribute the remaining lease costs (58.5 percent, or 65.0 percent less 6.5 percentage points) proportionally across the three cost categories (Depreciation, Interest, and Other Capital-Related) based on the proportion that these categories comprise of the sum of the Depreciation, Interest, and Other Capital-Related cost categories (excluding lease expenses). For example, the Other Capital-Related cost category represented 21.0 percent of all three cost categories (Depreciation, Interest, and Other Capital-Related) prior to any lease expenses being allocated. This 21.0 percent is applied to the 58.5 percent of remaining lease expenses so that another 12.3 percentage points of lease expenses as a percent of total capital-related costs is allocated to the Other Capital-Related cost category. Therefore, the resulting proposed Other Capital-Related cost weight is 26.1 percent (7.3 percent + 6.5 percent + 12.3 percent). This is the same methodology used for the 2017-based LTCH market basket. The proposed allocation of these lease expenses are shown in Table EEEE 3.
Finally, we proposed to further divide the Depreciation and Interest cost categories. We proposed to separate Depreciation cost category into the following two categories: (1) Building and Fixed Equipment and (2) Movable Equipment. We also proposed to separate the Interest cost category into the following two categories: (1) Government/Nonprofit; and (2) For profit.
To disaggregate the Depreciation cost weight, we needed to determine the percent of total depreciation costs for LTCHs (after the allocation of lease costs) that are attributable to Building and Fixed equipment, which we ( print page 69441) hereafter refer to as the “fixed percentage.” We proposed to use depreciation and lease data from Worksheet A-7 of the 2022 Medicare cost reports, which is the same methodology used for the 2017-based LTCH market basket. Based on the 2022 LTCH Medicare cost report data, we have determined that depreciation costs for building and fixed equipment account for 39 percent of total depreciation costs, while depreciation costs for movable equipment account for 61 percent of total depreciation costs. As previously mentioned, we proposed to allocate lease expenses among the Depreciation, Interest, and Other Capital-Related cost categories. We determined that leasing building and fixed equipment expenses account for 94 percent of total leasing expenses, while leasing movable equipment expenses account for 6 percent of total leasing expenses. We proposed to sum the depreciation and leasing expenses for building and fixed equipment, as well as sum the depreciation and leasing expenses for movable equipment. This results in the proposed Building and Fixed Equipment Depreciation cost weight (after leasing costs are included) representing 78 percent of total depreciation costs and the Movable Equipment Depreciation cost weight (after leasing costs are included) representing 22 percent of total depreciation costs.
To disaggregate the Interest cost weight, we determine the percent of total interest costs for LTCHs that are attributable to government and nonprofit facilities, which we hereafter refer to as the “nonprofit percentage,” because price pressures associated with these types of interest costs tend to differ from those for for-profit facilities. We proposed to use interest costs data from Worksheet A-7 of the 2022 Medicare cost reports for LTCHs, which is the same methodology used for the 2017-based LTCH market basket. The nonprofit percentage determined using this method is 48 percent.
We received no specific comments on the proposed methodology to derive the detailed capital cost weights and therefore are finalizing this methodology without modification. Table EEEE 3 provides the proposed and final detailed capital cost shares obtained from the Medicare cost reports. Ultimately, these detailed capital cost shares are applied to the total Capital-Related cost weight determined in section VIII.D.3.b. of the preamble of this final rule to separate the total Capital-Related cost weight of 8.5 percent into more detailed cost categories and weights.

Table EEEE 4 shows the cost categories and weights for the proposed and final 2022-based LTCH market basket compared to the 2017-based LTCH market basket.

After developing the proposed cost weights for the 2022-based LTCH market basket, we selected the most appropriate wage and price proxies currently available to represent the rate of price change for each cost category. For the majority of the cost weights, we base the price proxies on U.S. Bureau of Labor Statistics (BLS) data and group them into one of the following BLS categories:
- Employment Cost Indexes. Employment Cost Indexes (ECIs) measure the rate of change in employment wage rates and employer costs for employee benefits per hour worked. These indexes are fixed-weight indexes and strictly measure the change in wage rates and employee benefits per hour. ECIs are superior to Average Hourly Earnings (AHE) as price proxies for input price indexes because they are not affected by shifts in occupation or industry mix, and because they measure pure price change and are available by both occupational group and by industry. The industry ECIs are based on the NAICS and the occupational ECIs are based on the Standard Occupational Classification System (SOC).
- Producer Price Indexes. Producer Price Indexes (PPIs) measure the average change over time in the selling prices received by domestic producers for their output. The prices included in the PPI are from the first commercial transaction for many products and some services ( https://www.bls.gov/ppi/ ).
- Consumer Price Indexes. Consumer Price Indexes (CPIs) measure the average change over time in the prices paid by urban consumers for a market basket of consumer goods and services ( https://www.bls.gov/cpi/ ). CPIs are only used when the purchases are similar to those of retail consumers rather than purchases at the producer level, or if no appropriate PPIs are available.
We evaluate the price proxies using the criteria of reliability, timeliness, availability, and relevance:
- Reliability. Reliability indicates that the index is based on valid statistical methods and has low sampling variability. Widely accepted statistical methods ensure that the data were ( print page 69443) collected and aggregated in a way that can be replicated. Low sampling variability is desirable because it indicates that the sample reflects the typical members of the population. (Sampling variability is variation that occurs by chance because only a sample was surveyed rather than the entire population.)
- Timeliness. Timeliness implies that the proxy is published regularly, preferably at least once a quarter. The market baskets are updated quarterly, and therefore, it is important for the underlying price proxies to be up-to-date, reflecting the most recent data available. We believe that using proxies that are published regularly (at least quarterly, whenever possible) helps to ensure that we are using the most recent data available to update the market basket. We strive to use publications that are disseminated frequently, because we believe that this is an optimal way to stay abreast of the most current data available.
- Availability. Availability means that the proxy is publicly available. We prefer that our proxies are publicly available because this will help ensure that our market basket updates are as transparent to the public as possible. In addition, this enables the public to be able to obtain the price proxy data on a regular basis.
- Relevance. Relevance means that the proxy is applicable and representative of the cost category weight to which it is applied.
We believe that the CPIs, PPIs, and ECIs that we have selected meet these criteria. Therefore, we believe that they continue to be the best measure of price changes for the cost categories to which they would be applied.
Table EEEE 7 lists all price proxies that we proposed to use for the 2022-based LTCH market basket. The next section of the rule contains a detailed explanation of the price proxies we proposed for each cost category weight.
We proposed to continue to use the ECI for Wages and Salaries for All Civilian workers in Hospitals (BLS series code CIU1026220000000I) to measure the wage rate growth of this cost category. This is the same price proxy used in the 2017-based LTCH market basket ( 85 FR 58917 ).
We proposed to continue to use the ECI for Total Benefits for All Civilian workers in Hospitals to measure price growth of this category. This ECI is calculated using the ECI for Total Compensation for All Civilian workers in Hospitals (BLS series code CIU1016220000000I) and the relative importance of wages and salaries within total compensation. This is the same price proxy used in the 2017-based LTCH market basket ( 85 FR 58917 ).
We proposed to continue to use the PPI Commodity Index for Commercial Electric Power (BLS series code WPU0542) to measure the price growth of this cost category. This is the same price proxy used in the 2017-based LTCH market basket ( 85 FR 58917 ).
For the 2022-based LTCH market basket, we proposed to use a blend of the PPI Industry for Petroleum Refineries (NAICS 3241), PPI for Other Petroleum and Coal Products (NAICS 32419) and the PPI Commodity for Natural Gas. Our analysis of the Bureau of Economic Analysis' 2017 Benchmark I-O data for NAICS 622000 Hospitals shows that Petroleum Refineries expenses account for approximately 86 percent, Other Petroleum and Coal Products expenses account for about 7 percent and Natural Gas expenses account for approximately 7 percent of Hospitals' (NAICS 622000) total Fuel: Oil and Gas expenses. Therefore, we proposed to use a blend of 86 percent of the PPI Industry for Petroleum Refineries (BLS series code PCU324110324110), 7 percent of the PPI for Other Petroleum and Coal Products (BLS series code PCU32419) and 7 percent of the PPI Commodity Index for Natural Gas (BLS series code WPU0531) as the price proxy for this cost category. The 2017-based LTCH market basket used a 90/10 blend of the PPI Industry for Petroleum Refineries and PPI Commodity for Natural Gas, reflecting the 2012 I-O data ( 85 FR 58917 ). We believe that the three proposed price proxies are the most technically appropriate indices available to measure the price growth of the Fuel: Oil and Gas cost category in the 2022-based LTCH market basket.
We proposed to continue to use the CMS Hospital Professional Liability Index as the price proxy for PLI costs in the 2022-based LTCH market basket. To generate this index, we collect commercial insurance medical liability premiums for a fixed level of coverage while holding non-price factors constant (such as a change in the level of coverage). This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58917 ).
We proposed to continue to use the PPI Commodity for Pharmaceuticals for Human Use, Prescription (BLS series code WPUSI07003) to measure the price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58917 ).
We proposed to continue to use the PPI Commodity for Processed Foods and Feeds (BLS series code WPU02) to measure the price growth of this cost category. This is the same price proxy used in the 2017-based LTCH market basket ( 85 FR 58917 ).
We proposed to continue to use the CPI for Food Away From Home (BLS series code CUUR0000SEFV) to measure the price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58917 ).
Similar to the 2017-based LTCH market basket, we proposed to use a four-part blended PPI as the proxy for the chemical cost category in the 2022-based LTCH market basket. The proposed blend is composed of the PPI Industry for Industrial Gas Manufacturing, Primary Products (BLS series code PCU325120325120P), the PPI Industry for Other Basic Inorganic Chemical Manufacturing (BLS series code PCU32518-32518), the PPI Industry for Other Basic Organic Chemical Manufacturing (BLS series code PCU32519-32519), and the PPI Industry for Other Miscellaneous Chemical Product Manufacturing (BLS series code PCU325998325998). For the 2022-based LTCH market basket, we proposed to derive the weights for the PPIs using the 2017 Benchmark I-O data. The 2017-based LTCH market basket used the 2012 Benchmark I-O data to derive the weights for the four PPIs ( 85 FR 58917 through 58918 ). We did not receive comments on the proposed methodology to derive the blended Chemicals price proxy using the 2017 Benchmark I-O and therefore are finalizing this methodology without modification. Table EEEE 5 shows the weights for each of the four PPIs used to create the proposed and final blended Chemicals proxy for the 2022-based LTCH market basket compared to the 2017-based blended Chemicals proxy.

We proposed to use a blended price proxy for the Medical Instruments category. The 2017 Benchmark I-O data shows the majority of medical instruments and supply costs are for NAICS 339112—Surgical and medical instrument manufacturing costs (approximately 64 percent) and NAICS 339113—Surgical appliance and supplies manufacturing costs (approximately 36 percent). To proxy the price changes associated with NAICS 339112, we proposed to use the PPI for Surgical and medical instruments (BLS series code WPU1562). This is the same price proxy we used in the 2017-based LTCH market basket. To proxy the price changes associated with NAICS 339113, we proposed to use a 50/50 blend of the PPI for Medical and surgical appliances and supplies (BLS series code WPU1563) and the PPI for Miscellaneous products, Personal safety equipment and clothing (BLS series code WPU1571). We proposed to include the latter price proxy as it would reflect personal protective equipment including but not limited to face shields and protective clothing. The 2017 Benchmark I-O data does not provide specific expenses for these products; however, we recognize that this category reflects costs faced by LTCHs. For the 2017-based LTCH market basket, we used a blend composed of 57 percent of the commodity-based PPI Commodity for Surgical and Medical Instruments (BLS series code WPU1562) and 43 percent of the PPI Commodity for Medical and Surgical Appliances and Supplies (BLS series code WPU1563) reflecting the 2012 Benchmark I-O data ( 85 FR 58918 ).
We proposed to continue to use the PPI Commodity for Rubber and Plastic Products (BLS series code WPU07) to measure price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58918 ).
We proposed to use a 61/39 blend of the PPI Commodity for Publications Printed Matter and Printing Material (BLS Series Code WPU094) and the PPI Commodity for Converted Paper and Paperboard Products (BLS series code WPU0915) to measure the price growth of this cost category. The 2017 Benchmark I-O data shows that 61 percent of paper and printing expenses are for Printing (NAICS 323110) and the remaining expenses are for Paper manufacturing (NAICS 322). The 2017-based LTCH market basket ( 85 FR 58918 ) used the PPI Commodity for Converted Paper and Paperboard Products (BLS series code WPU0915) as this comprised the majority of expenses as reported in the 2012 Benchmark I-O data.
We proposed to continue to use the PPI Commodity for Finished Goods Less Food and Energy (BLS series code WPUFD4131) to measure the price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58918 ).
We proposed to continue to use the ECI for Total Compensation for Private Industry workers in Professional and Related (BLS series code CIU2010000120000I) to measure the price growth of this category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58918 ).
We proposed to continue to use the ECI for Total Compensation for Private Industry workers in Office and Administrative Support (BLS series code CIU2010000220000I) to measure the price growth of this category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58918 ).
We proposed to continue to use the ECI for Total Compensation for All Civilian workers in Installation, Maintenance, and Repair (BLS series code CIU1010000430000I) to measure the price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58918 ).
We proposed to continue to use the ECI for Total Compensation for Private Industry workers in Service Occupations (BLS series code CIU2010000300000I) to measure the price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58918 ).
We proposed to continue to use the ECI for Total Compensation for Private Industry workers in Professional and Related (BLS series code CIU2010000120000I) to measure the price growth of this category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58919 ).
We proposed to continue to use the ECI for Total Compensation for Private Industry workers in Financial Activities (BLS series code CIU201520A000000I) to measure the price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58919 ).
We proposed to continue to use the CPI for Telephone Services (BLS series code CUUR0000SEED) to measure the price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58919 ).
We proposed to continue to use the CPI for All Items Less Food and Energy ( print page 69445) (BLS series code CUUR0000SA0L1E) to measure the price growth of this cost category. This is the same proxy used in the 2017-based LTCH market basket ( 85 FR 58919 ).
We received the following comments on our proposed price proxies for the 2022-based LTCH market basket.
Comment: A few commenters stated that the Bureau of Labor Statistics' Employment Cost Index (ECI) used by CMS to calculate the labor portion of hospital costs only considers the salary costs of hospitals' employed staff; it does not reflect the portion of labor costs associated with contract labor that have risen in recent years. The commenters stated that this has proven to be a significant problem—and a major shortcoming of the current approach to calculating rate increases.
The commenters further stated that while the public health emergency has ended, LTCHs' reliance on contract staffing, and in particular contract nurses, has not ended, nor will it anytime soon. The commenters stated that while the need for such supplemental staffing has declined to a degree, it is not going away. The commenters stated their belief that CMS's calculation of Medicare LTCH rates should reflect this. For this reason, the commenters asked CMS to find new or additional data sources that capture this aspect of hospitals' labor costs when calculating future LTCH rate increases, including the final rate increase for FY 2025.
Response: We believe that the ECI for wages and salaries for hospital workers is accurately reflecting the price change associated with the labor used to provide hospital care. We believe that the price of employed staff and contract labor are influenced by the same factors and should generally grow at similar rates. The ECI appropriately does not reflect other factors that might affect the rate of price changes associated with labor costs such as a shift in the occupations that may occur due to increases in case-mix or shifts in hospital purchasing decisions (for instance, to hire or to use contract labor). In most periods when there are not significant occupational shifts or significant shifts between employed and contract labor, the data has shown that the growth in the ECI for wages and salaries for hospital workers has generally been consistent with overall hospital wage trends. For example, our analysis of the Medicare cost report data shows from 2011 to 2019 the compound annual growth rate of both IPPS Medicare allowable salaries per hour and contract labor costs per hour was 2.5 percent, near the 2.0-percent growth rate of the ECI for wages and salaries for hospital workers over the same period (note the ECI would not reflect skill mix change whereas the salaries data would reflect these changes).
From 2019 to 2022, however, as noted by the commenters, contract labor utilization increased and employed labor utilization decreased, the combination of which is reflected in the LTCH compensation cost weight for the proposed LTCH market basket. Over this same period, the ECI for hospital workers grew 3.6 percent, which is about 1.6 percentage points faster than the 2011 to 2019 historical average growth rate, reflecting the recent wage price inflation cited by the commenters.
For this final rule, based on the more recent IGI second quarter 2024 forecast with historical data through the first quarter of 2024, the projected 2022-based LTCH market basket increase factor for FY 2025 reflects an increase in compensation prices of 4.0 percent.
After consideration of public comments, we are finalizing the price proxies for the operating portion of the 2022-based LTCH market basket as proposed without modification.
We proposed to continue to use the same price proxies for the capital-related cost categories as were applied in the 2017-based LTCH market basket, which are provided in Table EEEE 7 and described in this section of this rule. Specifically, we proposed to proxy:
- Depreciation: Building and Fixed Equipment cost category by BEA's Chained Price Index for Nonresidential Construction for Hospitals and Special Care Facilities (BEA Table 5.4.4. Price Indexes for Private Fixed Investment in Structures by Type).
- Depreciation: Movable Equipment cost category by the PPI Commodity for Machinery and Equipment (BLS series code WPU11).
- Nonprofit Interest cost category by the average yield on domestic municipal bonds (Bond Buyer 20-bond index).
- For-profit Interest cost category by the average yield of the iBoxx AAA Corporate Bond Yield index.
- Other Capital-Related cost category by the CPI-U for Rent of Primary Residence (BLS series code CUUS0000SEHA).
We believe these are the most appropriate proxies for LTCH capital-related costs that meet our selection criteria of relevance, timeliness, availability, and reliability. We also proposed to continue to vintage weight the capital price proxies for Depreciation and Interest in order to capture the long-term consumption of capital. This vintage weighting method is similar to the method used for the 2017-based LTCH market basket and is described in section VIII.D.4.b.(2). of the preamble of this final rule.
We received no comments on the proposed price proxies for the capital portion of the 2022-based LTCH market basket and therefore are finalizing the use of these price proxies without modification.
Because capital is acquired and paid for over time, capital-related expenses in any given year are determined by both past and present purchases of physical and financial capital. The vintage-weighted capital-related portion of the proposed 2022-based LTCH market basket is intended to capture the long-term consumption of capital, using vintage weights for depreciation (physical capital) and interest (financial capital). These vintage weights reflect the proportion of capital-related purchases attributable to each year of the expected life of building and fixed equipment, movable equipment, and interest. We proposed to use vintage weights to compute vintage-weighted price changes associated with depreciation and interest expenses.
Capital-related costs are inherently complicated and are determined by complex capital-related purchasing decisions, over time, based on such factors as interest rates and debt financing. In addition, capital is depreciated over time instead of being consumed in the same period it is purchased. By accounting for the vintage nature of capital, we are able to provide an accurate and stable annual measure of price changes. Annual nonvintage price changes for capital are unstable due to the volatility of interest rate changes and, therefore, do not reflect the actual annual price changes for LTCH capital-related costs. The capital-related component of the proposed 2022-based LTCH market basket reflects the underlying stability of the capital-related acquisition process.
The methodology used to calculate the vintage weights for the proposed 2022-based LTCH market basket is the same as that used for the 2017-based LTCH market basket with the only difference being the inclusion of more recent data. To calculate the vintage weights for depreciation and interest expenses, we first need a time series of capital-related purchases for building ( print page 69446) and fixed equipment and movable equipment. We found no single source that provides an appropriate time series of capital-related purchases by hospitals for all of the previously mentioned components of capital purchases. The early Medicare cost reports did not have sufficient capital-related data to meet this need. Data we obtained from the American Hospital Association (AHA) do not include annual capital-related purchases. However, the AHA does provide a consistent database of total expenses from 1963 to 2020—the latest available data. Consequently, we proposed to use data from the AHA Panel Survey and the AHA Annual Survey to obtain a time series of total expenses for hospitals. We also proposed to use data from the AHA Panel Survey supplemented with the ratio of depreciation to total hospital expenses obtained from the Medicare cost reports to derive a trend of annual depreciation expenses for 1963 through 2020. We proposed to separate these depreciation expenses into annual amounts of building and fixed equipment depreciation and movable equipment depreciation as previously determined. From these annual depreciation amounts we derive annual end-of-year book values for building and fixed equipment and movable equipment using the expected life for each type of asset category. While data are not available that are specific to LTCHs, we believe this information for all hospitals serves as a reasonable proxy for the pattern of depreciation for LTCHs.
To continue to calculate the vintage weights for depreciation and interest expenses, we also needed to account for the expected lives for building and fixed equipment, movable equipment, and interest for the proposed 2022-based LTCH market basket. We proposed to calculate the expected lives using Medicare cost report data for LTCHs. The expected life of any asset can be determined by dividing the value of the asset (excluding fully depreciated assets) by its current year depreciation amount. This calculation yields the estimated expected life of an asset if the rates of depreciation were to continue at current year levels, assuming straight-line depreciation. Using this proposed method, we determined the average expected life of building and fixed equipment to be equal to 16 years, and the average expected life of movable equipment to be equal to 9 years. For the expected life of interest, we believe that vintage weights for interest should represent the average expected life of building and fixed equipment because, based on previous research described in the FY 1997 IPPS final rule ( 61 FR 46198 ), the expected life of hospital debt instruments and the expected life of buildings and fixed equipment are similar. We note that for the 2017-based LTCH market basket, we derived an expected average life of building and fixed equipment of 18 years and an expected average life of movable equipment of 9 years ( 85 FR 58920 ).
Multiplying these expected lives by the annual depreciation amounts results in annual year-end asset costs for building and fixed equipment and movable equipment. Then we calculated a time series, beginning in 1964, of annual capital purchases by subtracting the previous year's asset costs from the current year's asset costs.
For the building and fixed equipment and movable equipment vintage weights, we proposed to use the real annual capital-related purchase amounts for each asset type to capture the actual amount of the physical acquisition, net of the effect of price inflation. These real annual capital-related purchase amounts are produced by deflating the nominal annual purchase amount by the associated price proxy as previously provided. For the interest vintage weights, we proposed to use the total nominal annual capital-related purchase amounts to capture the value of the debt instrument (including, but not limited to, mortgages and bonds). Using these capital-related purchase time series specific to each asset type, we proposed to calculate the vintage weights for building and fixed equipment, for movable equipment, and for interest.
The vintage weights for each asset type are deemed to represent the average purchase pattern of the asset over its expected life (in the case of building and fixed equipment and interest, 16 years, and in the case of movable equipment, 9 years). For each asset type, we used the time series of annual capital-related purchase amounts available from 2020 back to 1964. These data allow us to derive forty-two 16-year periods of capital-related purchases for building and fixed equipment and interest, and forty-nine 9-year periods of capital-related purchases for movable equipment. For each 16-year period for building and fixed equipment and interest, or 9-year period for movable equipment, we proposed to calculate annual vintage weights by dividing the capital-related purchase amount in any given year by the total amount of purchases over the entire 16-year or 9-year period. This calculation is done for each year in the 16-year or 9-year period and for each of the periods for which we have data. Then we proposed to calculate the average vintage weight for a given year of the expected life by taking the average of these vintage weights across the multiple periods of data.
We received no comments on the proposed methodology to derive the vintage weights for the 2022-based LTCH market basket and therefore are finalizing these vintage weights without modification.
The vintage weights for the capital-related portion of the proposed and final 2022-based LTCH market basket and the 2017-based LTCH market basket are presented in Table EEEE 6.

The process of creating vintage-weighted price proxies requires applying the vintage weights to the price proxy index where the last applied vintage weight in Table EEEE6 is applied to the most recent data point. We have provided on the CMS website an example of how the vintage weighting price proxies are calculated, using example vintage weights and example price indices. The example can be found at the following link: http://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/MedicareProgramRatesStats/MarketBasketResearch.html in the zip file titled “Weight Calculations as described in the IPPS FY 2010 Proposed Rule.”
Table EEEE 7 shows both the operating and capital price proxies for the proposed and final 2022-based LTCH market basket.

For FY 2025 (that is, October 1, 2024 through September 30, 2025), we proposed to use an estimate of the proposed 2022-based LTCH market basket to update payments to LTCHs based on the best available data. Consistent with historical practice, we estimate the LTCH market basket update for the LTCH PPS based on IHS Global, Inc.'s (IGI) forecast using the most recent available data. IGI is a nationally recognized economic and financial forecasting firm with which CMS contracts to forecast the components of the market baskets and total factor productivity (TFP).
Based on IGI's fourth quarter 2023 forecast with history through the third quarter of 2023, the projected market basket update for FY 2025 was 3.2 percent. This projected 2022-based LTCH market basket update reflected an increase in compensation prices (proxied by the ECIs for All Civilian workers in Hospitals) of 3.7 percent. IGI's forecast of the ECIs considers overall labor market conditions (including rise in contract labor employment due to tight labor market conditions) as well as trends in contract labor wages, which both have an impact on wage pressures for workers employed directly by the hospital.
Consistent with our historical practice of estimating market basket increases ( print page 69449) based on the best available data, we proposed a market basket update of 3.2 percent for FY 2025. Furthermore, because the proposed FY 2025 annual update is based on the most recent market basket estimate for the 12-month period (currently 3.2 percent), we also proposed that if more recent data became subsequently available (for example, a more recent estimate of the market basket), we would use such data, if appropriate, to determine the FY 2025 annual update in the final rule. (The proposed annual update to the LTCH PPS standard payment rate for FY 2025 is discussed in greater detail in section V.A.2. of the Addendum to the proposed rule.)
Based on the more recent data available for this FY 2025 IPPS/LTCH final rule (that is, IGI's second quarter 2024 forecast of the 2022-based LTCH market basket with historical data through the first quarter of 2024), we estimate that the FY 2025 market basket update is 3.5 percent.
Using the current 2017-based LTCH market basket and IGI's second quarter 2024 forecast for the market basket components, the FY 2025 market basket update would be 3.4 percent (before taking into account any statutory adjustment). Therefore, the update based on the 2022-based LTCH market basket is currently projected to be 0.1 percentage point higher for FY 2025 compared to the current 2017-based LTCH market basket. This higher update is primarily due to the higher Compensation cost weight in the 2022-based market basket (61.8 percent) compared to the 2017-based LTCH market basket (53.2 percent). This is partially offset by the lower cost weight associated with All Other Services (such as Professional Fees and Installation, Maintenance, and Repair Services) for the 2022-based LTCH market basket relative to the 2017-based LTCH market basket. Table EEEE 8 compares the 2022-based LTCH market basket and the 2017-based LTCH market basket percent changes.

Over the historical time period covering FY 2020 through FY 2023, the average growth rate of the 2022-based LTCH market basket is the same as the average growth rate of the 2017-based LTCH market basket. Over the forecasted time period covering FY 2024 through FY 2027, the average growth rate of the 2022-based LTCH market basket is 0.1 percentage point higher than the average growth rate of the 2017-based LTCH market basket. This is driven by higher projected growth for FY 2024 and FY 2025 for the 2022-based LTCH market basket, which is primarily a result of the higher Compensation cost weight combined with faster projected growth in Compensation prices for FY 2024 and FY 2025 relative to projected prices for All Other Services. In FY 2026 and FY 2027 prices for these two aggregate cost categories are projected to grow at similar rates.
We summarize the public comments we received on the adequacy of the proposed LTCH market basket increase and our responses in section VIII.C.2.d. of the preamble of this final rule. Below are comments we received regarding the proposed methods for deriving the LTCH market basket.
Comment: Some commenters expressed concern that the proposed 3.2 percent market basket update and the 4.3 percentage point increase in the labor-related share do not sufficiently account for the dramatic increase in labor costs that LTCHs are incurring. Several commenters stated that there were proposed increases in the cost category weights for Contract Labor (12.6 percent versus 4.4 percent currently) and Home Office/Related Organization Contract Labor (3.7 percent versus 1.9 percent currently), but that CMS forecasted that the overall update will only be 0.1 percentage point higher for FY 2025 through FY 2027 using this 2022-based market basket. The commenters stated that according to CMS, this is primarily due to the offset from the lower All Other cost category weight (20.8 percent versus 28.3 percent currently), which includes things such as Professional Fees and Installation, Maintenance, and Repair Services. The commenters claimed that CMS is saying that most of the increases in labor costs reflected in the updated market basket are effectively removed by the All Other cost category. A commenter stated that this would only be true if All Other costs decreased substantially in FY 2022 compared to FY 2017 and the commenter stated that it is not clear from the proposed rule that this is the case. A commenter stated that the assigned cost weights for labor costs and All Other costs only reflect the relative proportion of such costs in the market basket, not how much overall costs in those categories have grown since FY 2017. Commenters stated that a 0.1 percentage point increase to the market basket update, using the rebased and revised LTCH market basket, does not reflect an overall increase in the cost of ( print page 69450) LTCH goods and services, compared to the 2017-based market basket.
Commenters stated that CMS needs to either modify the methodology it used to rebase and revise the market basket or apply a separate adjustment to the market basket rate to account for significantly higher labor and supply costs incurred by providers. Another commenter requested CMS adjust the market basket to reflect staffing levels and ratios as well as stated that it should also account for facilities using highly priced temporary staff during surges or staff shortages.
Response: As stated previously, to derive the LTCH market basket for a specific base year, total base period costs are estimated for a set of mutually exclusive and exhaustive spending categories. Of those total costs, we estimate the proportion of total costs that each category represents, with these proportions called cost weights. Therefore, any changes in the cost weight from a prior base period will reflect the growth in the costs for that specific category relative to the growth in the costs for other categories. As a result, while costs for a particular category may have increased from 2017 to 2022, the cost weight (such as contract labor) would only increase if these specific costs increased faster than the increase in total costs from 2017 to 2022. Therefore, we disagree with the commenter that a decrease in the All Other cost category would only occur if All Other costs decreased substantially in 2022 compared to 2017.
As indicated by the commenters, the cost weights of the LTCH market basket are intended to reflect the relative proportion that specific costs represent of total costs, and not how much overall costs in those categories have increased since the prior base year. The LTCH market basket is described as a fixed-weight index because it represents the change in price over time of a constant mix (quantity and intensity) of goods and services needed to provide LTCH services. We believe that the proposed methodology to derive the cost categories and cost weights of the proposed market basket is detailed and robust and that this proposed method produces valid relative cost weights that are representative of LTCH cost structures. To allow for interested parties to evaluate this methodology, we have provided the detailed calculations including the data sources (such as the specific Medicare cost report fields) and trimming methodology so that commenters are able to replicate the methodology and provide specific comments on the derivation of these cost weights. We will continue to monitor the Medicare cost reports as new data becomes available, and any changes to the LTCH market basket will be proposed in future rulemaking.
Comment: Some commenters requested that CMS publish additional information and underlying data regarding its market basket methodology, as they have been unable to replicate some of CMS' figures. A commenter was concerned about the lack of transparency from CMS regarding the rebased and revised market basket. The commenter stated that they conducted an independent analysis of the rebased market basket CMS proposed for FY 2025. The commenter stated that it was unclear how CMS arrived at the 3.2 percent market basket update from the revised market basket cost categories. The commenter stated that their analyst discovered that there were many uncertainties in the data that CMS used to rebase and revise the market basket, including whether the data accurately represents LTCH costs, how the data were trimmed, and the exact subcomponents of each cost category. Accordingly, the commenter requested that CMS provide more transparency in the final rule regarding the data used to rebase the market basket and how that data resulted in the proposed 3.2 percent market basket update for the FY 2025 LTCH PPS payment update.
Response: In the FY 2025 IPPS/LTCH proposed rule ( 89 FR 36268 through 36271 ), for each of the major cost categories of the market basket, we provided detailed descriptions of the Worksheet, column number, and line number on the Medicare cost report that we proposed to use to derive the costs for each category as well as for total Medicare allowable costs. For categories such as benefits and contract labor where data reporting is more limited, we performed detailed analysis of the data by reweighting the cost weights by ownership type using the distribution of the universe of LTCHs and compared these results to the proposed cost weights to help ensure that the data were representative of LTCHs, which we noted in the FY 2025 IPPS/LTCH proposed rule ( 89 FR 36270 ). In addition, in the proposed rule, we provided descriptions of the trimming methods applied for each cost weight, which is again described in section VIII.D.3.b of the preamble of this final rule. We believe this information is sufficient to allow stakeholders to replicate the market basket cost weights that we proposed and are finalizing for the 2022-based LTCH market basket.
Information on the CMS market baskets can be found at the CMS website: https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-research-and-information . This website provides information including but not limited to how a top-line market basket level is derived from the detailed cost categories, how a four-quarter percent change moving average is calculated, and a link to a spreadsheet containing an example of how the detailed market basket cost weights are calculated for the 2006-based IPPS market basket, which is similar to the approach followed for the LTCH market basket as well as most of the other CMS market baskets. In addition, the latest, publicly available CMS market baskets are available at the CMS website: https://www.cms.gov/data-research/statistics-trends-and-reports/medicare-program-rates-statistics/market-basket-data . We note that publicly available market baskets on the CMS website would reflect an updated forecast only after a proposed or final rule is published. Using these spreadsheets, stakeholders are able to replicate the top-line market basket index levels in the historical time period by multiplying the detailed index level for each cost category by the associated cost weight. These products (weight multiplied by index level) can then be summed up to derive the aggregate market basket index level. In response to the commenter's request for more transparency, in this final rule, we are also providing the projected increase for FY 2025 for some of the aggregated cost categories that underlie the most recent forecast of the FY 2025 LTCH market basket increase (3.5 percent). This detail is consistent with the level of information that we publish on the CMS website on a quarterly basis as described above. We note that prices for the compensation cost weight, which accounts for about 62 percent of the market basket are projected to increase 4.0 percent in FY 2025; prices for All Other Products and Services, which accounts for about 28 percent of the market basket are projected to increase 2.8 percent; and prices for Capital-Related costs, which accounts for about 8.5 percent of the LTCH market basket are projected to increase 3.2 percent. Weighting the projected price increases for these aggregated categories (reflecting 98.5 percent of the LTCH market basket cost weights with the remaining 1.5 percent reflecting Utilities and PLI), we obtain a weighted average projected increase of 3.5 percent. While the projected market basket increase is calculated using the aggregation of the detailed price forecasts multiplied by ( print page 69451) their respective cost weights for each of the 26 individual cost categories, we want to provide an estimate of how the broader cost categories are contributing to the overall increase. We strive for transparency regarding our methods and regularly respond to questions from stakeholders regarding the market baskets via email at [email protected] .
Comment: A few commenters requested CMS closely evaluate its current forecasting and market basket practices for further refinement. Commenters stated that during this period of high cost growth, Medicare payment updates for LTCHs have now shown a consistent pattern of failing to not only forecast, but also eventually capture this growth. The commenters stated that despite the high rates of medical inflation, LTCH payments have not kept up with general inflation. Commenters claimed that since fee-for-service Medicare patients make up more than half of all LTCH discharges, and other insurers adjust payment rates relative to Medicare reimbursement, these missed forecasts compound the obstacles facing LTCHs.
Commenters also stated that it is confounding how hospitals, and especially labor-intensive LTCHs, could have a market basket that is significantly below general inflation. The commenters stated that there has been very large growth in LTCH costs in the last several years which has exceeded general inflation. However, they stated, even the actual market basket growth (not forecasts) was below general inflation during this time. The commenters stated that the market basket itself may have shortcomings that fail to properly capture growth.
The commenters stated that there may be many overlapping, contributing factors to the market basket failing to capture inflationary factors. One such factor is the increased utilization in contract labor, which the Employment Cost Index does not capture. They encouraged CMS to thoroughly reexamine the market basket and its recent shortcomings to identify other potential areas for refinement and stated support for working with CMS to assist with such an endeavor.
Response: Since the inception of the LTCH PPS, the LTCH payment rates (with the exception of statutorily-mandated updates) have been updated by a projection of a market basket percentage increase—consistent with other CMS PPS updates (including the IPPS, SNF PPS, and Home Health PPS). The LTCH market basket (as well as other CMS market baskets) is a fixed-weight, Laspeyres-type index that measures price changes over time and would not reflect increases in costs associated with changes in the volume or intensity of input goods and services. As such, the LTCH market basket update would reflect the prospective price pressures described by the commenters as increasing during a high inflation period (such as faster wage growth or higher energy prices), but would not reflect other factors that might increase the level of costs, such as the quantity of labor used. The impact of changes in quantity or use of services on the market basket cost weights would be captured when the market basket is rebased.
We note that the market basket percentage increase is a forecast of the price pressures that hospitals are expected to face in 2025. We also note that when developing its forecast for the ECI for hospital workers, IGI (a nationally recognized economic and financial forecasting firm with which CMS contracts to forecast the price proxies of the market baskets) considers overall labor market conditions (including rise in contract labor employment due to tight labor market conditions) as well as trends in contract labor wages, both of which could potentially impact wages for workers employed directly by the hospital. As projected by IGI and other independent forecasters, compensation growth and upward price pressures are expected to slow in 2025 relative to 2022 and 2023. Therefore, we believe the projected increase in the LTCH market basket for FY 2025 is reflective of expectations for input price growth in FY 2025.
As is our general practice, we proposed that if more recent data became available, we would use such data, if appropriate, to derive the final FY 2025 LTCH market basket update for the final rule. For this final rule, based on the more recent IGI second quarter 2024 forecast with historical data through the first quarter of 2024, the projected 2022-based LTCH market basket increase factor for FY 2025 is 3.5 percent, which is 0.3 percentage point higher than the projected FY 2025 LTCH market basket increase factor in the proposed rule, and reflects an increase in compensation prices of 4.0 percent. We would note that the 10-year historical average (2014-2023) growth rate of the 2022-based LTCH market basket is 2.8 percent with compensation prices increasing 2.9 percent. The final FY 2025 LTCH market basket increase reflects IGI's projected inflation and overall economic outlook.
As discussed in section V.B. of the Addendum to this final rule, under the authority of section 123 of the BBRA as amended by section 307(b) of the BIPA, we established an adjustment to the LTCH PPS payments to account for differences in LTCH area wage levels (§ 412.525(c)). The labor-related portion of the LTCH PPS standard Federal payment rate, hereafter referred to as the labor-related share, is adjusted to account for geographic differences in area wage levels by applying the applicable LTCH PPS wage index. The labor-related share is determined by identifying the national average proportion of total costs that are related to, influenced by, or vary with the local labor market. As discussed in more detail in this section of this rule and similar to the 2017-based LTCH market basket, we classify a cost category as labor-related and include it in the labor-related share if the cost category is defined as being labor-intensive and its cost varies with the local labor market. As stated in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58988 ), the labor-related share for FY 2024 was defined as the sum of the FY 2024 relative importance of Wages and Salaries; Employee Benefits; Professional Fees: Labor-Related Services; Administrative and Facilities Support Services; Installation, Maintenance, and Repair Services; All Other: Labor-related Services; and a portion of the Capital-Related Costs from the 2017-based LTCH market basket.
We proposed to continue to classify a cost category as labor-related if the costs are labor-intensive and vary with the local labor market. Given this, based on our definition of the labor-related share and the cost categories in the 2022-based LTCH market basket, we proposed to include in the labor-related share for FY 2025 the sum of the FY 2025 relative importance of Wages and Salaries; Employee Benefits; Professional Fees: Labor-Related; Administrative and Facilities Support Services; Installation, Maintenance, and Repair Services; All Other: Labor-Related Services; and a portion of the Capital-Related cost weight from the 2022-based LTCH market basket.
Similar to the 2017-based LTCH market basket, the 2022-based LTCH market basket includes two cost categories for nonmedical Professional fees (including but not limited to, expenses for legal, accounting, and engineering services). These are Professional Fees: Labor-Related and Professional Fees: Nonlabor-Related. For the 2022-based LTCH market basket, we proposed to estimate the labor-related percentage of non-medical professional fees (and assign these expenses to the Professional Fees: Labor-Related ( print page 69452) services cost category) based on the same method that was used to determine the labor-related percentage of professional fees in the 2017-based LTCH market basket.
As was done for the 2017-based LTCH market basket, we proposed to determine the proportion of legal, accounting and auditing, engineering, and management consulting services that meet our definition of labor-related services based on a survey of hospitals conducted by CMS in 2008. We notified the public of our intent to conduct this survey on December 9, 2005 ( 70 FR 73250 ) and did not receive any public comments in response to the notice ( 71 FR 8588 ). A discussion of the composition of the survey and post-stratification can be found in the FY 2010 IPPS/LTCH PPS final rule ( 74 FR 43850 through 43856 ). Based on the weighted results of the survey, we determined that hospitals purchase, on average, the following portions of contracted professional services outside of their local labor market:
- 34 percent of accounting and auditing services.
- 30 percent of engineering services.
- 33 percent of legal services.
- 42 percent of management consulting services.
For the 2022-based LTCH market basket, we proposed to apply each of these percentages to the respective 2017 Benchmark I-O cost category underlying the professional fees cost category to determine the Professional Fees: Nonlabor-Related costs. The Professional Fees: Labor-Related costs were determined to be the difference between the total costs for each Benchmark I-O category and the Professional Fees: Nonlabor-Related costs. This is the same methodology that we used to separate the 2017-based LTCH market basket professional fees category into Professional Fees: Labor-Related and Professional Fees: Nonlabor-Related cost categories.
Effective for transmittal 18 ( https://www.cms.gov/Regulations-and-Guidance/Guidance/Transmittals/Transmittals/r18p240i ), the hospital Medicare Cost Report (CMS Form 2552-10, OMB No. 0938-0050) is collecting information on whether a hospital purchased professional services (for example, legal, accounting, tax preparation, bookkeeping, payroll, advertising, and management or consulting services or both) from an unrelated organization and if the majority of these expenses were purchased from unrelated organizations located outside of the main hospital's local area labor market. We encourage all providers to provide this information so we can potentially use these more recent data in future rulemaking to determine the labor-related share.
In the 2022-based LTCH market basket, we proposed that nonmedical professional fees that were subject to allocation based on these survey results represent approximately 3.6 percent of total costs (and are limited to those fees related to Accounting and Auditing, Legal, Engineering, and Management Consulting services). Based on our survey results, we proposed to apportion approximately 2.3 percentage points of the 3.6 percentage point figure into the Professional Fees: Labor-Related cost category and designate the remaining approximately 1.3 percentage points into the Professional Fees: Nonlabor-Related cost category.
In addition to the professional services as previously listed, for the 2022-based LTCH market basket, we proposed to allocate a proportion of the Home Office/Related Organization Contract Labor cost weight, calculated using the Medicare cost reports as previously stated, into the labor-related and nonlabor-related cost categories. We proposed to classify these expenses as labor-related and nonlabor-related as many facilities are not located in the same geographic area as their home office and, therefore, do not meet our definition for the labor-related share that requires the services to be purchased in the local labor market.
Similar to the 2017-based LTCH market basket, we proposed for the 2022-based LTCH market basket to use the Medicare cost reports for LTCHs to determine the home office labor-related percentages. The Medicare cost report requires a hospital to report information regarding their home office provider. Using information on the Medicare cost report, we compare the location of the LTCH with the location of the LTCH's home office. We proposed to classify a LTCH with a home office located in their respective labor market if the LTCH and its home office are located in the same Metropolitan Statistical Area (MSA). Then we determine the proportion of the Home Office/Related Organization Contract Labor cost weight that should be allocated to the labor-related share based on the percent of total Home Office/Related Organization Contract Labor costs for those LTCHs that had home offices located in their respective MSA of total Home Office/Related Organization Contract Labor costs for LTCHs with a home office. We determined a LTCH's and its home office's MSA using their zip code information from the Medicare cost report. Using this methodology with the 2022 Medicare cost reports, we determined that 4 percent of LTCHs' Home Office/Related Organization Contract Labor costs were for home offices located in their respective MSA, or local labor markets. Therefore, we are allocating 4 percent of the Home Office/Related Organization Contract Labor cost weight (0.1 percentage point = 3.7 percent × 4 percent) to the Professional Fees: Labor-Related cost weight and 96 percent of the Home Office/Related Organization Contract Labor cost weight to the Professional Fees: Nonlabor-Related cost weight (3.6 percentage points = 3.7 percent × 96 percent). For comparison, for the 2017-based LTCH market basket we also allocated 4 percent of the Home Office/Related Organization Contract Labor cost weight to the Professional Fees: Labor-Related cost weight ( 85 FR 58924 ).
In summary, based on the two allocations mentioned earlier, we proposed to apportion 2.4 percentage points (2.3 percentage points + 0.1 percentage point) of the Professional Fees and Home Office/Related Organization Contract Labor cost weights into the Professional Fees: Labor-Related cost category. This amount was added to the portion of professional fees that we already identified as labor-related using the I-O data such as contracted advertising and marketing costs (approximately 0.6 percentage point of total costs) resulting in a total Professional Fees: Labor-Related cost weight of 3.0 percent.
We summarize the public comments we received on our proposed methodology for deriving the proposed labor-related share for FY 2025 and our responses here.
Comment: A commenter appreciated the proposal to increase the labor-related share based on data that better reflect increased labor costs as a percentage of LTCH's overall cost structure. However, the commenter disagreed with CMS' assertion that some portion of professional contract labor costs is not subject to geographic variation in labor costs. The commenter requested that CMS allocate all 3.6 percentage points for professional services costs to the Professional Services: Labor-Related Category for the final rule.
The commenter claimed that CMS' assumption that fees for services provided by firms located outside of a hospital's core-based statistical area (CBSA) do not vary based on geography is invalid. The commenter stated that the implied underpinning of this assumption is that national and regional professional services firms do not compete with local professional services firms based in a hospital's CBSA. ( print page 69453) However, the commenter stated that this is an erroneous assumption as hospitals seeking professional services solicit proposals for these services from local, regional, and national firms and therefore, regional and national firms have the incentive to adjust their pricing in response to local labor market conditions. The commenter stated that if the local labor market has lower wages than the national average—which will influence the pricing of a local firm's response to a request for proposal from a hospital—regional and national firms must reduce the offered price of their services to be competitive with local firms that offer the same services. Conversely, the commenter stated, if the local labor market has higher wages than the national average, regional and national firms have every incentive to price accordingly to increase their profit margins on a given contract. Therefore, the commenter claimed that pricing for services offered by regional and national firms to hospitals in differing CBSAs will vary significantly based on local rates due to these firms competing with local firms that provide the same service.
Therefore, the commenter asked CMS to provide evidence that pricing for professional services delivered by regional and national firms to hospitals is offered in a market that is not subject to geographic cost variation. The commenter stated that unless the agency can produce strong evidence that prices for professional services provided by firms outside of a hospital's local labor market are homogenous—that an LTCH in San Antonio, Texas, is charged the same hourly rates for audit services by the same national accounting firm as a hospital in Sacramento, Calif.—it asks CMS to restore the 1.3 percentage points it proposes to reclassify to Professional Services: Nonlabor-Related to the Professional Services: Labor-Related category. In the absence of data that show standardized pricing by regional and national professional services firms, the commenter stated that the Professional Services: Labor-Related category cost weight should be 3.6 percentage points.
Response: We disagree with the commenter and believe it is appropriate that a proportion of Accounting & Auditing, Legal, Engineering, and Management Consulting services costs purchased by hospitals should be excluded from the labor-related share. Under the authority of section 123 of the BBRA, as amended by section 307(b) of the BIPA, we established an adjustment to the LTCH PPS standard Federal payment rate to account for differences in LTCH area wage levels under § 412.525(c). The labor-related share of the LTCH PPS standard Federal payment rate is adjusted to account for geographic differences in area wage levels by applying the applicable LTCH PPS wage index.
The purpose of the labor-related share is to reflect the proportion of the national PPS base payment rate that is adjusted by the hospital's wage index (representing the relative costs of their local labor market to the national average). Therefore, we include a cost category in the labor-related share if the costs are labor intensive and vary with the local labor market.
As acknowledged by the commenter and confirmed by the survey of hospitals conducted by CMS in 2008 (as stated previously in this final rule), professional services can be purchased from local firms as well as national and regional professional services firms. It is not necessarily the case, as asserted by the commenter, that these national and regional firms have fees that match those in the local labor market even though providers have the option to utilize those firms. That is, fees for services purchased from firms outside the local labor market may differ from those that would be purchased in the local labor market for any number of reasons (including but not limited to, the skill level of the contracted personnel, higher capital costs, etc.). As noted earlier in this section of this final rule, the definition for the labor-related share requires the services to be purchased in the local labor market; therefore, CMS' allocation of approximately 64 percent (2.3 percentage points of 3.6 percentage points) of the Professional Fees cost weight to Professional Fees: Labor-Related costs based on the 2008 survey results [ 257 ] is consistent with the commenter's assertion that not all Professional Fees services are purchased in the local labor market. We believe it is reasonable to conclude that the costs of those Professional Fees services purchased directly within the local labor market are directly related to local labor market conditions and, thus, should be included in the labor-related share. The remaining approximately 36 percent of Professional Fees costs, which are purchased outside the local labor market, reflect different and additional factors outside the local labor market and, thus, should be excluded from the labor-related share. In addition, we note the compensation costs of professional services provided by hospital employees (which would reflect the local labor market) are included in the labor-related share as they are included in the Wages and Salaries and Employee Benefits cost weights.
Therefore, for the reasons discussed, we believe our proposed methodology of continuing to allocate only a portion of Professional Fees to the Professional Fees: Labor-Related cost category is appropriate. As stated previously, effective for transmittal 18 ( https://www.cms.gov/Regulations-andGuidance/Guidance/Transmittals/Transmittals/r18p240i ), the hospital Medicare Cost Report (CMS Form 2552- 10, OMB No. 0938-0050) is collecting information on whether a hospital purchased professional services (for example, legal, accounting, tax preparation, bookkeeping, payroll, advertising, and management or consulting services or both) from an unrelated organization and if the majority of these expenses were purchased from unrelated organizations located outside of the main hospital's local area labor market. We encourage all providers to provide this information so we can potentially use it in future rulemaking to determine the labor-related share.
As previously stated, we proposed to include in the labor-related share the sum of the relative importance of Wages and Salaries; Employee Benefits; Professional Fees: Labor- Related; Administrative and Facilities Support Services; Installation, Maintenance, and Repair Services; All Other: Labor-Related Services; and a portion of the Capital-Related cost weight from the 2022-based LTCH market basket. The relative importance reflects the different rates of price change for these cost categories between the base year (2022) and FY 2025. Based on IGI's fourth quarter 2023 forecast of the proposed 2022-based LTCH market basket, the sum of the FY 2025 relative importance for operating costs (Wages and Salaries, Employee Benefits, Professional Fees: Labor-Related, Administrative and Facilities Support Services, Installation Maintenance and Repair Services, and All Other: Labor-Related Services) was 68.9 percent. The portion of Capital costs that is estimated to be influenced by the local labor market is 46 percent, which is the same percentage applied to the 2017-based LTCH market basket. Since the relative importance for Capital is 8.4 percent of the proposed 2022-based LTCH market basket in FY 2025, we took 46 percent of 8.4 percent to determine the proposed labor-related ( print page 69454) share of Capital for FY 2025 of 3.9 percent. Therefore, we proposed a total labor-related share for FY 2025 of 72.8 percent (the sum of 68.9 percent for the operating cost and 3.9 percent for the labor-related share of Capital).
Based on IGI's second quarter 2024 forecast of the 2022-based LTCH market basket, the sum of the FY 2025 relative importance for Wages and Salaries, Employee Benefits, Professional Fees: Labor-Related, Administrative and Facilities Support Services, Installation Maintenance & Repair Services, and All Other: Labor-Related Services is 68.9 percent. The portion of Capital costs that is influenced by the local labor market is estimated to be 46 percent, which is the same percentage applied to the 2017-based LTCH market basket. Since the relative importance for Capital is 8.4 percent of the 2022-based LTCH market basket in FY 2025, we take 46 percent of 8.4 percent to determine the labor-related share of Capital for FY 2025 of 3.9 percent. Therefore, using more recent data, the total labor-related share for FY 2025 is 72.8 percent (the sum of 68.9 percent for the operating cost and 3.9 percent for the labor-related share of Capital).
We summarize the comments we received on the proposed FY 2025 labor-related share and our responses here.
Comment: A commenter does not support the proposed increase in the labor-related share, as any increase to the labor-related share percentage penalizes any facility that has a wage index less than 1.0. The commenter stated that across the country, there is a growing disparity between high-wage and low-wage states that harms hospitals in many rural and underserved communities. The commenter claimed that limiting the increase in the labor-related share would help mitigate that growing disparity. The commenter stated that they do not support any increases in the labor-related share percentages.
Response: The total difference between the FY 2025 labor-related share using the proposed 2022-based LTCH market basket (72.8 percent) and the FY 2024 labor-related share using the 2017-based LTCH market basket (68.5 percent) is 4.3 percentage points and this difference is primarily attributable to the revision to the base year cost weights for those categories included in the labor-related share. We periodically rebase the LTCH market basket in order to reflect more recent data on LTCH cost structures. From 2017 to 2022, the Medicare cost report data showed a notable increase in the Compensation cost weight for LTCHs, which is consistent with comments that we received in prior rulemaking, specifically the FY 2024 IPPS/LTCH proposed rule comments ( 88 FR 59134 ) that stated the 2017-based LTCH market basket did not sufficiently account for the dramatic increases in labor costs that LTCHs were incurring. We believe incorporating these more recent data in the LTCH market basket is appropriate, and is in response to public comments, resulting in a corresponding increase in the labor-related share. In addition, we proposed to use the FY 2025 relative importance values for the labor-related cost categories from the 2022-based LTCH market basket because it accounts for more recent data regarding price pressures and cost structure of LTCHs. This methodology is consistent with the determination of the labor-related share since the implementation of the LTCH PPS. As stated in the FY 2025 IPPS/LTCH proposed rule, we also proposed that if more recent data became available, we would use such data, if appropriate, to determine the FY 2025 labor-related share for the final rule. Based on IHS Global Inc.'s second quarter 2024 forecast with historical data through the first quarter of 2024, the FY 2025 labor-related share for the final rule is 72.8 percent.
After consideration of public comments, we are finalizing a FY 2025 labor-related share of 72.8 percent.
Table EEEE 9 shows the FY 2025 labor-related share using the 2022-based LTCH market basket relative importance and the FY 2024 labor-related share using the 2017-based LTCH market basket.

The total difference between the FY 2025 labor-related share using the 2022-based LTCH market basket (72.8 percent) and the FY 2024 labor-related share using the 2017-based LTCH market basket (68.5 percent) is 4.3 percentage points and this difference is primarily attributable to the revision to the base year cost weights for those categories included in the labor-related share. The 4.3 percentage points revision to the base year cost weights is a result of: (1) an 8.6 percentage points upward revision to the base year Compensation cost weight, which is derived using the LTCH Medicare cost report data; (2) a 3.6 percentage points downward revision in the base year labor-related categories associated with incorporating the 2017 Benchmark I-O data; and (3) a 0.7 percentage point ( print page 69455) downward revision in the base year labor-related portion of capital costs, which is derived using the LTCH Medicare cost report data.
In section IX. of the proposed rule, we sought comment on and proposed changes to a number of Medicare quality reporting programs. Specifically,
- In section IX.B. of the proposed rule ( 89 FR 36284 through 36306 ), we made the following crosscutting quality program proposals or request for comment:
++ Adoption of the Patient Safety Structural Measure in the Hospital IQR Program and PCHQR Program.
++ Modification to the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) Survey Measure in the Hospital IQR Program, Hospital VBP Program, and PCHQR Program.
++ Advancing Patient Safety and Outcomes Across the Hospital Quality Programs—Request for Comment.
- In section IX.C. of the proposed rule ( 89 FR 36306 through 36341 ), the Hospital IQR Program.
- In section IX.D. of the proposed rule ( 89 FR 36341 through 36343 ), the PCHQR Program.
- In section IX.E. of the proposed rule ( 89 FR 36343 through 36352 ), the LTCH QRP.
- In section IX.F. of the proposed rule ( 89 FR 36352 through 36381 ), the Medicare Promoting Interoperability Program for eligible hospitals and critical access hospitals (CAHs) (previously known as the Medicare EHR Incentive Program).
We respond to public comments on each of these sections below.
A foundational commitment of providing healthcare services is to ensure safety, as embedded in the centuries-old Hippocratic Oath, “First, do no harm.” Yet, the landmark reports To Err is Human [ 258 ] and Crossing the Quality Chasm [ 259 ] surfaced major deficits in healthcare quality and safety. These reports resulted in widespread awareness of the alarming prevalence of patient harm and, over the past two decades, healthcare facilities implemented various interventions and strategies to improve patient safety, with some documented successes. [ 260 ] However, progress has been slow, and preventable harm to patients in the clinical setting resulting in significant morbidity and mortality remains common. A recent systematic analysis of literature concluded that preventable mortality among inpatients results in approximately 22,165 preventable deaths annually. [ 261 ] In another recent study, researchers identified adverse events in almost one-quarter of admissions and showed that more than one-fifth were deemed preventable and almost one-third were considered serious (that is, caused harm that required intervention or prolonged recovery). [ 262 ]
Despite established patient safety protocols and quality measures, the COVID-19 public health emergency (PHE) strained the healthcare system substantially, introducing new safety risks and negatively impacting patient safety in the normal delivery of care. Since the onset of the COVID-19 PHE, the U.S. has seen marked declines in patient safety metrics, as evidenced by considerable increases in healthcare-associated infections (HAIs). [ 263 264 ] Studies found that central line-associated blood stream infections (CLABSIs) in hospitals were 60 percent higher than predicted in the absence of COVID-19, catheter-associated urinary tract infections (CAUTIs) were 43 percent higher, and methicillin-resistant Staphylococcus aureus (MRSA) bacteremia infections were 44 percent higher. Studies have shown that these results were likely due at least in part to disrupted routine infection control practices during the COVID-19 PHE. [ 265 266 ] Notably, recent reports demonstrate that some HAI rates have begun to decrease towards pre-PHE levels as the U.S. saw a 9 percent overall decrease in CLABSI, a 12 percent overall decrease in CAUTI and a 16 percent overall decrease in hospital onset MRSA bacteremia between 2021 and 2022 in acute care hospital settings. [ 267 ]
As healthcare facilities struggled to address the challenges posed by the COVID-19 PHE, safety gaps and risks in healthcare delivery were illuminated, [ 268 ] revealing a lack of resiliency in the healthcare system. [ 269 270 ] Beyond HAIs, other preventable types of patient harm that were brought to the forefront by the ( print page 69456) COVID-19 PHE include occurrences of pressure injuries [ 271 ] and patient falls [ 272 ] among hospitalized patients.
In addition to safety issues illuminated during the COVID-19 PHE, two other key patient safety indicators that are worth noting for their prevalence are postoperative respiratory failure [ 273 274 275 ] and acute kidney injuries (AKI). [ 276 277 ]
While the COVID-19 PHE may have disrupted routine infection control practices, these key patient safety indicators nevertheless show the importance of addressing gaps in safety to save lives, provide equitable medical care, and ensure that the U.S. healthcare system is resilient enough to withstand future challenges. Now is the time to recommit to better safety practices for both patients and healthcare workers, establish new protocols, and implement early interventions that would save many lives from preventable harms.
To accomplish these goals, the federal government is taking a multi-pronged approach to improve safety and reduce preventable harm to patients. The Agency for Healthcare Research and Quality (AHRQ), on behalf of HHS, has established the National Action Alliance for Patient and Workforce Safety (the National Action Alliance) as a public-private collaboration to improve both patient and workforce safety. [ 278 ] As described by AHRQ, the National Action Alliance is a partnership between HHS and its federal agencies and private stakeholders, including healthcare systems, clinicians, allied health professionals, patients, families, caregivers, professional societies, patient and workforce safety advocates, the digital healthcare sector, health services researchers, employers, and payors interested in recommitting the U.S. to advancing patient and workforce safety to move toward zero harm in healthcare. [ 279 ]
In September 2023, the President's Council of Advisors on Science and Technology (PCAST) published the “Report to the President: A Transformational Effort on Patient Safety,” with a call to action to renew “our nation's commitment to improving patient safety.” [ 280 ] The PCAST report put forth the following recommendations as a part of the call to action: (1) Establish and maintain federal leadership for the improvement of patient safety as a national priority; (2) Ensure that patients receive evidence-based practices for preventing harm and addressing risks; (3) Partner with patients and reduce disparities in medical errors and adverse outcomes; and (4) Accelerate research and deployment of practices, technologies, and exemplar systems of safe care. [ 281 ]
As part of this national recommitment to safety in healthcare, we are promoting the use of safety measures throughout our quality programs to identify and measure quality gaps and processes, and to make that information transparent and available to the public. Effective measurement is paramount to monitoring harm events, identifying key gaps, and tracking progress toward safer, more reliable care. Within CMS' hospital quality measurement programs, there are several outcome and process measures in use that capture specific conditions or procedures such as the Severe Sepsis and Septic Shock: Management Bundle measure, Patient Safety and Adverse Events Composite measure, Severe Obstetric Complications electronic clinical quality measure (eCQM), and the Safe Use of Opioids—Concurrent Prescribing eCQM. While these metrics are important, they are not sufficient by themselves to measure and incentivize investment in a resilient safety culture or the infrastructure necessary for sustainable high performance within the broad and complex domain of patient safety. The systems-level approach to patient safety maintains that errors and accidents in medical care are a reflection of system-level failures, rather than failings on the part of individuals. [ 282 ] There is a strong alignment among patient safety experts to shift to a more holistic, proactive, systems-based approach to patient safety. [ 283 284 285 286 287 288 ] While each of our existing measures address processes and outcomes that encourage providers to improve patient safety for specific conditions or related to specific treatments, these measures do not address the overall culture in which the care is provided. Including a systems-level measure would contribute to a culture that improves performance on these individual metrics as well as improves safety for all care provided within the hospital.
To drive action and improvements in safety and address this gap in systems-level measurement for safety within the Hospital IQR and PCHQR Programs, we proposed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36284 through 36293 ) the adoption of the Patient Safety Structural measure, a new ( print page 69457) attestation-based measure that assesses whether hospitals demonstrate a structure, culture, and leadership commitment that prioritize safety. The Patient Safety Structural measure includes five complementary domains, each containing a related set of statements that aim to capture the most salient, evidenced-based, structural, and cultural elements of safety. This measure is intended to be a foundational measure and designed to assess hospital implementation of a systems-based approach to safety best practices, as demonstrated by: leaders who prioritize and champion safety; organizational policies, protocols, goals, and metrics reflecting safety as a core value; a diverse group of patients and families meaningfully engaged with healthcare providers as partners in safety; practices indicative of a culture of safety; accountability and transparency in addressing adverse events; and continuous learning and improvement. This Patient Safety Structural measure is informed by Safer Together: The National Action Plan to Advance Patient Safety, [ 289 ] developed by the National Steering Committee for Patient Safety convened by the Institute for Healthcare Improvement (IHI), as well as scientific evidence from existing patient safety literature, and detailed input from patient safety experts, advocates, and patients. Combining this systems-level structural measure with other high priority safety outcome measures would result in a robust and complementary patient safety measure set.
We note that other safety measures discussed in this FY 2025 IPPS/LTCH PPS final rule complement the goals we have outlined for the Patient Safety Structural measure. Interested parties are encouraged to review our discussion of measures for Hospital Harm—Falls with Injury (section IX.C.5.c), Hospital Harm—Postoperative Respiratory Failure (section IX.C.5.d), and the adoption of two healthcare-associated infection measures (section IX.C.5.b).
In addition to the other federal safety initiatives noted previously, this measure also aligns with the CMS National Quality Strategy. Specifically, the CMS National Quality Strategy identifies four priority areas and eight goals, each with an identified objective, success target, and initial action steps for advancing a “high-quality, safe, equitable, and resilient health care system for all individuals.” [ 290 ] The Patient Safety Structural measure addresses the priority area Safety and Resiliency, and aligns with the goals to enable a responsive and resilient healthcare system to improve quality and to achieve zero preventable harm. For example, attestation statements within the measure require hospitals to confirm if their strategic plan includes publicly sharing their commitment to patient safety as a core value and outlines specific safety goals and associated metrics, including the goal of “zero preventable harm.”
This measure aligns with our efforts under the CMS National Quality Strategy's goal of advancing equity and whole-person care. [ 291 ] As stated in the measure attestation under Domain 2: Strategic Planning & Organizational Policy (see Table IX.B.1-01 of this final rule), “Patient safety and equity in care are inextricable, and therefore equity, with the goal of safety for all individuals, must be embedded in safety planning, goal-setting, policy and processes.” This measure furthers a patient-centered approach by promoting conversations on equity among hospital staff, leadership, and patients and caregivers that consider the diverse communities served by participants in CMS programs and the particular needs of each hospital's own community.
The measure also aligns with our Meaningful Measures Framework, which identifies high-priority areas for quality measurement and improvement to assess core issues most critical to high-quality healthcare and improving patient outcomes. [ 292 ] In 2021, we launched Meaningful Measures 2.0 to promote innovation and modernization of all aspects of quality, and to address a wide variety of settings, interested parties, and measure requirements. [ 293 ] The Patient Safety Structural measure supports these efforts and is aligned with the Meaningful Measures Area of “Safety” and the Meaningful Measures 2.0 goal to “Ensure Safe and Resilient Health Care Systems.” This measure also supports the Meaningful Measures 2.0 priority to “promote a safety culture within a health care organization.” This attestation measure focused on patient safety policies, processes, and activities aims to help hospitals better understand priorities for improving safety and serve as a prompt for action to invest in the infrastructure and safety culture necessary to reduce preventable harm to patients. When measure results are made public, patients and families would be able to make informed decisions on what facilities are best for them.
As required under section 1890A of the Act, the Consensus-Based Entity (CBE), currently Battelle, established the Partnership for Quality Measurement (PQM) to convene members comprised of clinicians, patients, measure experts, and health information technology specialists, to participate in the pre-rulemaking process and the measure endorsement process. The pre-rulemaking process, which we refer to as the Pre-Rulemaking Measure Review (PRMR), includes a review of measures published on the publicly available list of Measures Under Consideration (MUC List), [ 294 295 ] by one of several committees convened by the PQM, for the purpose of providing multi-stakeholder input to the Secretary on the selection of quality and efficiency measures under consideration for use in certain Medicare quality programs, including the PCHQR and Hospital IQR Programs. The PRMR process includes opportunities for public comment through a 21-day public comment period, as well as public listening sessions. The PQM posts the compiled comments and listening session inputs received during the public comment period and the listening sessions within 5 days of the close of the public comment period. More details regarding the PRMR process may be found in the PQM Guidebook of Policies and Procedures for Pre-Rulemaking Measure Review and Measure Set Review, available at: https://p4qm.org/PRMR , ( print page 69458) including details of the measure review processes in Chapter 3.
The CBE-established PQM also conducts the measure endorsement and maintenance (E&M) process to ensure a measure submitted for endorsement is evidence-based, reliable, valid, verifiable, relevant to enhanced health outcomes, actionable at the caregiver level, feasible to collect and report, and responsive to variations in patient characteristics—such as health status, language capabilities, race or ethnicity, and income level—and is consistent across types of health care providers, including hospitals and physicians (see section 1890(b)(2) of the Act). The PQM convenes several E&M project groups twice yearly, formally called the E&M Committees, each comprised of an E&M Advisory Group and an E&M Recommendations Group, to vote on whether a measure meets certain quality measure criteria. More details regarding the E&M process may be found in the PQM Endorsement and Maintenance (E&M) Guidebook available at: https://p4qm.org/EM , including details of the measure endorsement process in the section titled, “Endorsement and Review Process.”
For the voting procedures of the PRMR and E&M processes, the PQM utilizes the Novel Hybrid Delphi and Nominal Group (NHDNG) multi-step process, which is an iterative consensus-building approach aimed at a minimum of 75 percent agreement among voting members, rather than a simple majority vote, and supports maximizing the time spent to build consensus by focusing discussion on measures where there is disagreement. For example, the PRMR Hospital Recommendation Group can reach consensus and have the following voting results: (A) Recommend, (B) Recommend with conditions (with 75 percent of the votes casted as recommend with conditions or 75 percent between recommend and recommend with conditions), and (C) Do not recommend. If no voting category reaches 75 percent or greater (including the combined [A] recommend and [B] recommend with conditions), the PRMR Hospital Recommendation Group did not come to consensus and the voting result is `Consensus not reached.' Consensus not reached signals continued disagreement amongst the committee despite being presented with perspectives from public comment, committee member feedback and discussion, and highlights the multi-faceted assessments of quality measures. More details regarding the PRMR voting procedures may be found in Chapter 4 of the PQM Guidebook of Policies and Procedures for Pre-Rulemaking Measure Review and Measure Set Review. More details regarding the E&M voting procedures may be found in the PQM Endorsement and Maintenance (E&M) Guidebook.
As part of the PRMR process, the PRMR Hospital Recommendation Group reviewed the Patient Safety Structural measure (MUC2023-188) during a meeting on January 18 and 19, 2024. The Patient Safety Structural measure was included for consideration in the Hospital IQR and PCHQR Programs on the publicly available “2023 Measures Under Consideration List” (MUC List). [ 296 ]
The voting results of the PRMR Hospital Recommendation Group for the Patient Safety Structural measure for the Hospital IQR Program were: eight members of the group recommended adopting the measure into the Hospital IQR Program without conditions; five members recommended adoption with conditions; three committee members voted not to recommend the measure for adoption. Additionally, nine members of the group recommended adopting the measure into the PCHQR Program without conditions; four members recommended adoption with conditions; three committee members voted not to recommend the measure for adoption. Taken together, 81.3 percent of the votes were recommended with conditions for each program. Thus, the committee reached consensus and recommended the Patient Safety Structural measure for the Hospital IQR Program and the PCHQR Program with conditions.
The conditions recommended by the voting committee were: the publication of an implementation guide that clearly documents how safety is to be measured; and using data to narrow the scope before approving the measure for programs. An Attestation Guide was made available at the time of the publication of the proposed rule on the respective Hospital IQR Program and PCHQR Program pages on QualityNet. [ 297 ] Data obtained from the measure's national use would allow us to evaluate the effectiveness of, and the potential to narrow the future scope of, the proposed attestations. Therefore, we have adequately addressed the conditions raised by the PRMR Hospital Recommendations Group and proposed this measure for adoption.
In addition to the formal voting results on the adoption of the Patient Safety Structural measure, we note that the majority of public comments received on this measure during the PRMR process were supportive, with 91 out of 97 public comments (94%) either supporting (81) adoption or supporting adoption with conditions (10). Comments in support of the proposal included the need for a zero preventable harm goal, robust hospital leadership, developing trust through transparency, and the involvement of patients and their families in safety work. We thank the large number of patients, family members, and other interested parties who publicly participated in the PRMR process.
We proposed to adopt this measure into the Hospital IQR Program and the PCHQR Program despite the measure not being endorsed by the CBE. Section 1886(b)(3)(B)(viii)(IX)(aa) of the Act requires that each measure specified by the Secretary for use in the Hospital IQR Program be endorsed by the entity with a contract under section 1890(a) of the Act, and section 1866(k)(3)(A) of the Act imposes the same requirement for measures specified for use in the PCHQR Program. Sections 1886(b)(3)(B)(viii)(IX)(bb) and 1866(k)(3)(B) of the Act state, however, that in the case of a specified area or medical topic determined appropriate by the Secretary for which a feasible and practical measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not so endorsed as long as due consideration is given to a measure that has been endorsed or adopted by a consensus organization identified by the Secretary.
We reviewed measures endorsed by both the CBE which currently holds the contract under section 1890(a) of the Act and measures endorsed by the entity which formerly held that contract and were unable to identify any other CBE-endorsed measures on strategies and practices to strengthen hospitals' systems and culture for safety. Considering the lack of endorsed ( print page 69459) measures on this specified area or medical topic, we have determined that it would be appropriate to use a measure that is not endorsed by the CBE. This measure is relevant to enhanced health outcomes. As described in the background section for this measure (section IX.B.1.a. of the preamble of this final rule), medical errors and adverse events occur frequently and lead to adverse patient outcomes. This measure is designed to identify hospitals that practice a system-based approach to safety and embrace the importance of a safety culture. Demonstrating a structure, culture, and leadership commitment that prioritizes safety can improve care and outcomes for all patients. [ 298 ] The validity, feasibility and relevance of the measure have been thoroughly vetted by a Technical Expert Panel (TEP) convened by a CMS contractor and comprised of thought leaders in the field. [ 299 ] In response to the question of whether the domains capture the most important elements for advancing patient safety, most TEP members agreed that they do. [ 300 ] Furthermore, the measure developers engaged the members of the TEP for their operational and clinical expertise to assure that each domain was actionable and measurable. [ 301 ] As noted, the PRMR Hospital Committee received a total of 91 public comments expressing support for the Patient Safety Structural measure. [ 302 ] Most commenters were patients and family members who described their individual experiences with the medical system and preventable harms to which they were exposed. These commenters then emphasized the importance of the Patient Safety Structural measure's intent and domains for improving patient safety related to these experiences. [ 303 ] Due to the rigorous alignment with patient safety guidelines and literature as noted within section IX.B.1.a. of the preamble of this final rule, as well as strong support from expert stakeholders, patients, and caregivers as noted previously, we are confident that the foundational principles are sound, and the specifications are attainable, measurable, and actionable. We intend to submit the measure for future CBE endorsement.
The Patient Safety Structural measure is a structural measure developed to assess how well hospitals have implemented strategies and practices to strengthen their systems and culture for safety. The Patient Safety Structural measure comprises a set of complementary statements (or, attestations) that aim to capture the most salient, systems-oriented actions to advance safety. These statements should exemplify a culture of safety and leadership commitment to transparency, accountability, patient and family engagement, and continuous learning and improvement. Table IX.B.1-01 includes the five attestation domains and the corresponding attestation statements.

The Patient Safety Structural measure consists of five domains, each representing a complementary but separate safety commitment. Each of the five domains include five related attestation statements. Hospitals would need to evaluate and determine whether they can affirmatively attest to each domain. For a hospital to affirmatively attest to a domain, and receive a point for that domain, a hospital would evaluate and determine whether it engaged in each of the statements that comprise the domain (see Table IX.B.1-01), for a total of five possible points (one point per domain). A hospital would not be able to receive partial points for a domain.
For example, for Domain 2 (“Strategic Planning & Organizational Policy”), a hospital would evaluate and determine whether it meets the statements related to its strategic plan (Statement A), its safety goals (Statement B), policies and protocols for a “just culture” (Statement C), a patient safety curriculum and competencies for all hospital staff (Statement D), and an action plan for workforce safety (Statement E) (see Table IX.B.1-01). If its plan meets all five of these statements, the hospital would attest “yes” to each of the five attestation statements and would receive one point for Domain 2. If, for example, its plan only meets Statement A and Statement B, but does not meet Statement C, Statement D, and Statement E, the hospital would attest “yes” to Statement A and Statement B, attest “no” to Statement C, Statement D, and Statement E, and receive zero points for Domain 2. The hospital's overall score for the Patient Safety Structural measure can range from a total of zero to five points. If a hospital is comprised of more than one acute care hospital facility under one CCN, all such facilities reporting under the same CCN would need to satisfy these criteria for the hospital to affirmatively attest and receive points.
For more details on the measure specifications and the Attestation Guide for the Hospital IQR Program, we refer readers to the Web-Based Data Collection tab under the IQR Measures page and PCHQR measures page on QualityNet at both: https://qualitynet.cms.gov/inpatient/iqr/measures#tab2 and https://qualitynet.cms.gov/pch/measures , respectively. For more details on the measure specifications for the PCHQR Program, we refer readers to the Measures tab under the PCHQR page on QualityNet at: https://qualitynet.cms.gov/pch/measures .
Hospitals would be required to submit information for the Patient Safety Structural measure once annually using the data submission and reporting standard procedures set forth by the CDC for the National Healthcare Safety Network (NHSN). Presently, hospitals report measure data to the CDC NHSN on a monthly or quarterly basis, depending on the measure. Under the data submission and reporting process for the Patient Safety Structural measure, hospitals would be required to submit data once annually. We refer readers to the CDC's NHSN website ( https://www.cdc.gov/nhsn/index.html ) for data submission and reporting ( print page 69464) procedures; information more specific to the Patient Safety Structural measure would be available through NHSN before the first data submission period opens. We refer readers to sections IX.C.9. and IX.D.4 of the preamble of this final rule for more details on our previously finalized data submission and deadline requirements for structural measures in the Hospital IQR Program and PCHQR Program, respectively. We further refer readers to sections IX.C.9. and IX.D.4 of the preamble of this final rule for more details on our previously finalized data submission requirements for measures submitted via the CDC NHSN in the Hospital IQR Program and PCHQR Program, respectively. We proposed to adopt the Patient Safety Structural measure in the Hospital IQR Program beginning with the CY 2025 reporting period/FY 2027 payment determination and the PCHQR Program beginning with the CY 2025 reporting period/FY 2027 program year. Hospitals participating in the Hospital IQR Program and the PCHQR Program would satisfy their reporting requirement for the measure if they attest “yes” or “no” to each attestation statement in all five domains.
We proposed to publicly report the hospital's measure performance score, which would range from 0 to 5 points, on an annual basis on Care Compare beginning in Fall 2026 and on the Provider Data Catalog available at data.cms.gov for the PCHQR Program beginning in Fall 2026.
We invited public comment on this proposal.
Comment: Many commenters expressed support for the adoption of the Patient Safety Structural measure. Many commenters stated that this measure includes activities known to reduce harm, improve patient-centered care, encourage patient involvement, and encourage transparency. Some commenters stated that the measure emphasizes the importance of a systems-level and non-punitive approach to patient safety. A commenter stated that the Patient Safety Structural measure is data-driven, actionable, and workable. A commenter stated that the measure received a positive response during the MUC review process and that the participants shared personal experiences related to the benefits of this measure in improving patient safety, indicating its importance.
Response: We thank the commenters for their support for the adoption of the Patient Safety Structural measure and for their engagement in the PRMR process. We agree that the measure will provide greater transparency and encourage hospitals to implement activities to improve patient-centered care and reduce preventable harm to patients. We also agree that the measure will drive system-level changes that are actionable for hospitals. We also recognize and restate our appreciation for the public involvement in the PRMR meeting, including the personal experiences shared by patients and patient advocates that emphasized the importance of this measure.
Comment: Several commenters stated that patient safety is an urgent topic and supported adoption of the Patient Safety Structural measure to address it. Some of these commenters stated that while many of the items in the Patient Safety Structural measure are known best practices, there has been a delay in implementing them. Some commenters stated that the Patient Safety Structural measure would address this by ensuring prioritization of these activities through sending a signal to hospital governance bodies and executive leadership. Other commenters stated that patient safety has been declining and that the COVID-19 PHE accelerated this decline. These commenters expressed the belief that the Patient Safety Structural measure would provide guidance towards delivering safer care and would create a way to recognize hospitals that are exemplars in patient safety. Some commenters stated that the domains identified in the measure are critical areas for hospitals to focus on for patient safety.
Response: We thank the commenters for their support. We agree that it is important for hospitals to not delay adopting patient safety best practices and acknowledge that the COVID-19 PHE was disruptive for hospitals and illuminated gaps in patient safety. We agree that the domains of the Patient Safety Structural measure are critical areas of focus, and that the measure will offer guidance to hospitals on prioritizing patient safety in their organizational structure, culture, strategy, and overall care delivery.
Comment: Several commenters expressed support for the Patient Safety Structural measure because it is an attestation-based structural measure. Several of these commenters stated that being an attestation measure, this measure would have relatively low reporting burden, and that the benefits of adopting this measure, including providing strategies for improved patient safety, would outweigh the additional burden of reporting the measure. A commenter stated that an attestation-based, structural measure is preferential to outcomes-based measures for driving patient safety improvements because outcomes-based measures are lagging indicators. Another commenter stated that the Patient Safety Structural measure complements patient safety indicators (PSIs) but emphasizes building a culture that would drive improvements in safety. Some commenters stated that structural measures can set new expectations for the development of evidence-based programs and processes that would support improvements in high impact areas. A few commenters stated that by adopting an attestation measure CMS would provide motivation for implementing these activities without impacting hospital payments. A commenter stated that the process of understanding and attesting to these statements would increase the focus on patient safety (regardless of whether hospitals can attest positively). A commenter expressed support for the Attestation Guide.
Response: We thank the commenters for their support of the Patient Safety Structural measure as an attestation-based structural measure and for their support of the Attestation Guide. We agree with the importance of using attestation measures and encouraging hospitals to facilitate a culture to improve safety. We also agree that the attestation measure encourages hospitals to focus on safety regardless of how they score. We also agree that, while attestation measures do entail some burden, they are less demanding than requiring reporting of the data or details underlying each of the attestation statements. By adopting an attestation measure in combination with measures that specifically assess processes and outcomes, we seek to achieve a holistic, systematic approach to advancing patient safety which balances measure types and reporting burden. We further agree that adoption of this attestation measure will complement outcome and process measures currently in CMS' hospital quality measurement programs. We note that within CMS' hospital quality measurement programs, there are several outcome and process measures in use that capture specific conditions or procedures such as the Severe Sepsis and Septic Shock: Management Bundle measure, Patient Safety and Adverse Events Composite measure, Severe Obstetric Complications electronic clinical quality measure (eCQM), and the Safe Use of Opioids—Concurrent Prescribing eCQM. Furthermore, we discuss Hospital Harm—Falls with Injury (section IX.C.5.c), Hospital Harm—Postoperative Respiratory Failure (section IX.C.5.d), and the adoption of two healthcare-associated ( print page 69465) infection measures (section IX.C.5.b) in this final rule.
Comment: A few commenters supported adoption of the Patient Safety Structural measure because of its alignment with other guidance. A commenter specifically expressed support for the Patient Safety Structural measure's alignment with the IHI's National Action Plan for Advancing Patient Safety. This commenter emphasized that both the National Action Plan for Advancing Patient Safety and the Patient Safety Structural measure include interdependence among the categories.
Response: We thank the commenters for their support and acknowledge that in developing the Patient Safety Structural measure, we strove to align it with other national efforts to advance patient safety.
Comment: A commenter specifically supported public reporting of this measure to ensure transparency to patients, families, and community members.
Response: We thank the commenter for their support of publicly reporting the Patient Safety Structural measure. We agree that providing publicly reported measure results on the Care Compare website for the Hospital IQR Program promotes transparency to patients, families, communities, and other interested parties, and will allow patients to make more informed decisions on their care. We note that the PCHQR Program publicly reports measure data on data.cms.gov .
Comment: A few commenters expressed support for measures that address patient safety and specifically recommended measures to improve the accuracy of blood and blood culture tests. Some commenters specifically stated that quickly identifying and appropriately treating blood stream infections can reduce inappropriate treatment.
Response: We thank the commenters for their support of safety measures. While the Patient Safety Structural measure does not directly assess the speed and accuracy with which hospitals identify and treat blood stream infections, establishing a structural, cultural, and leadership commitment to prioritizing safety can improve all elements of patient safety, including appropriate treatment of blood stream infections. We continually seek to develop and adopt quality measures that address important quality and patient safety aspects of care and may consider measures directly related to the speed and accuracy with which hospitals identify and treat blood stream infections as we review the Hospital IQR Program and the PCHQR Program in the future.
Comment: A commenter stated that the Patient Safety Structural measure provides an opportunity to update the physician credentialing process to include a focus on safety behaviors as well as clinical competence.
Response: We thank this commenter for their support of the Patient Safety Structural measure; however, we note that the physician credentialing process is outside the scope of the Hospital IQR Program and the PCHQR Program.
Comment: A few commenters recommended adding domains to the Patient Safety Structural measure. Some of these commenters specifically recommended adding a domain related to workforce well-being and engagement which includes attestations related to soliciting ideas for improved care processes from the workforce and using closed-loop, transparent communications regarding improvement efforts. Other commenters recommended including diagnostic excellence as a domain.
Response: We agree that workforce well-being and engagement is linked with a learning culture that prioritizes safety. We also agree that diagnostic excellence underlies safe and appropriate healthcare. However, the Patient Safety Structural measure was developed by identifying and focusing on the highest priority domains. This allows us to balance the total number of attestations and associated burden on hospitals. Most TEP members agreed that the domains capture the most important elements for advancing patient safety. [ 309 ] As this measure was developed to capture the most important elements, it is appropriate to adopt the measure without additional domains or attestations. We will continue to evaluate the measure's performance and consider updating it if appropriate in the future.
Comment: A few commenters stated that some attestations are already covered by actions required of hospitals under the Conditions of Participation (CoPs) and recommended that CMS streamline the measure to eliminate duplication. Specifically, commenters stated that Domains 1 and 2 are covered by the hospital CoPs related to quality assessment and performance improvement at 42 CFR 482.2
1(a) through (e). A few commenters recommended aligning with existing requirements, such as those of The Joint Commission or counties or states.
Response: We acknowledge that the hospital CoPs related to quality assessment and performance improvement (QAPI) programs at 42 CFR 482.21(a) through (e) and requirements set forth by entities such as The Joint Commission and other regulatory entities (such as counties and states) address similar topics to the attestations required to report the Patient Safety Structural measure and helped inform its development. However, we disagree that the Patient Safety Structural measure is redundant to these CoPs and other requirements and maintain that it is complementary to them. While existing requirements may outline the minimum activities related to developing, implementing, and maintaining an effective, ongoing, hospital-wide, data-driven quality assessment and performance improvement program, the Patient Safety Structural measure requires hospitals to attest to whether they have built upon these minimum activities to exemplify a culture of safety and leadership with transparency, accountability, patient and family engagement, and continuous learning and improvement. In addition, the public display requirements of the Hospital IQR and PCHQR Programs provide for information on this measure to be available to patients, consumers, family and caregivers, and other interested parties. This transparency can further incentivize quality improvement.
Comment: A few commenters recommended that CMS update the CoPs with the items from this measure instead of adopting a structural measure. Some of these commenters stated that this would allow hospitals to be assessed and receive feedback on their performance during the survey and certification process.
Response: We agree that hospitals benefit from receiving feedback on their patient safety structures and other CoP requirements during the survey and certification process. However, measures are intended to evaluate, and this measure evaluates the current state of patient safety structures within hospitals. Through standardized measurement and transparency, the Patient Safety Structural measure can also encourage hospitals to build upon the activities already required under the CoPs to establish a culture of safety and leadership commitment to transparency, accountability, patient and family engagement, and continuous learning ( print page 69466) and improvement. We also note that hospitals are surveyed for CoPs, on average, every three to five years and this quality measure provides more frequent updates to the public.
Comment: Many commenters recommended updating the Attestation Guide. A few of these commenters suggested including examples provided through public comments to improve the Attestation Guide's ability to support reporting. A few commenters recommended providing detailed guidance on data collection, including how hospitals should document that they are satisfying each domain. Some of these commenters stated that additional guidance would improve the Patient Safety Structural measure's ability to support cross-hospital comparisons.
Response: While we agree with commenters that examples can be meaningful and provide hospitals with information to help adopt these evidence-based practices, we also intend the Patient Safety Structural measure to maintain flexibility and allow each hospital to adopt practices that are most effective for its individual circumstances. Because these practices will not be identical across hospitals, the documentation supporting the practice may also vary. We will provide education and outreach materials to support hospitals in identifying additional evidence-based practices they could adopt and in documenting that they have adopted those practices. While we are not updating the Attestation Guide to add detailed documentation guidance, we have updated the Attestation Guide to provide some additional clarification on other topics based on the public comments we received. Version 2.0 of the Attestation Guide is available at both: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures .
Comment: A commenter stated that these domains align with those in the Office of the National Coordinator for Health Information Technology's (ONC's) Safety Assurance Factors for EHR Resilience (SAFER) Guides and recommended adopting a staged approach, like the approach used for the SAFER Guides. Specifically, the commenter recommended a staged Yes/No attestation without any financial impacts the first year with expanded requirements in future years.
Response: We agree with the commenter that the SAFER Guides complement the Patient Safety Structural measure. We note that the SAFER Guides are focused on optimizing the safety and safe use of EHRs [ 310 ] while the Patient Safety Structural measure solicits information about whether hospitals have built upon these minimum activities to exemplify a culture of safety and leadership commitment to transparency, accountability, patient and family engagement, and continuous learning and improvement. In the FY 2024 IPPS/LTCH PPS final rule, we modified requirements for the SAFER Guides measure in the Medicare Promoting Interoperability Program to require eligible hospitals and critical access hospitals (CAHs) to attest “yes” to having conducted an annual self-assessment of all nine SAFER Guides at any point during the calendar year in which the EHR reporting period occurs, beginning with the EHR reporting period in CY 2024 ( 88 FR 59262 through 59265 ). We note that, unlike the Medicare Promoting Interoperability Program, the Hospital IQR Program is a pay-for-reporting program, which means that hospitals that report the required measure data in accordance with the form, manner, and timing policies specified by the Secretary are not subject to a financial penalty under this program and the PCHQR Program is a quality reporting program that does not have a financial penalty associated with it. Therefore, there will be no financial penalties for hospitals that attest either “yes” or “no” to each of the domains of the Patient Safety Structural measure.
Comment: A commenter stated that because this is a patient safety measure it would be appropriate to remove references to workforce safety and create a new measure to address those safety challenges, which the commenter stated are different than those faced by patients.
Response: We agree that there are different safety challenges faced by healthcare workers than those faced by patients. However, not only is workforce safety an important component to identifying hospitals that exemplify a culture of safety and leadership commitment to transparency, accountability, patient and family engagement, and continuous learning and improvement, but it is also a precondition to advancing patient safety with a unified, total systems-based approach to eliminate harm to both patients and the workforce. [ 311 ] Because workplace safety is a precondition to advancing patient safety, this measure necessarily includes attestations related to workforce safety. We thank the commenter for their suggestion for a new measure in CMS programs, as we recognize this as an important and ongoing concern for the healthcare workforce.
Comment: A few commenters recommended renaming the measure “Patient and Workforce Safety Structural measure” because items such as “just culture” apply to the workforce as well as patients.
Response: We agree that many of the domains and attestations in the Patient Safety Structural measure apply to the workforce. While workforce safety is an important element of this measure, these elements are included because workforce safety is a precondition to advancing patient safety. Because the measure is focused on establishing structure, culture, and leadership commitment to prioritizing patient safety, it is appropriate for the measure title to focus on advancing patient safety.
Comment: A commenter recommended refining the attestations to include efforts made by health systems instead of at the individual hospital level.
Response: We agree that for hospitals that are part of health systems, there are many best practices and resources that can be shared among hospitals across the system. However, patient safety is ultimately the responsibility of the institution providing the care, in this case the individual hospital. Therefore, we encourage hospitals to use resources available through their health systems to meet these attestations, but our intention is that the attestation should represent the structure, culture, and leadership commitment to prioritizing safety at the individual hospital.
Comment: A commenter recommended incentivizing voluntary safety initiatives as opposed to implementing an attestation measure.
Response: The Hospital IQR Program is a pay-for-reporting program, which means that hospitals that report the required measure data in accordance with the form, manner, and timing policies specified by the Secretary are not subject to a financial penalty under this program. The PCHQR Program is a quality reporting program that does not have a financial penalty associated with it. A hospital's performance on the measure, which for the Patient Safety Structural measure is a score from 0 to 5 points, has no impact on a hospital's Medicare reimbursement. Therefore, the activities in the Patient Safety Structural measure are voluntary safety initiatives. ( print page 69467) While we recognize that a hospital's performance on the measure may impact the hospital's reputation through public reporting, this reputational impact is a means of encouraging the voluntary adoption of safety related best practices.
Comment: A commenter recommended CMS engage with leaders to identify barriers to improving safety.
Response: We agree that hospital leaders are a critical source of information regarding barriers to improving safety. The Patient Safety Structural measure is informed by scientific evidence from existing patient safety research and literature, guidance from established healthcare quality and safety organizations, and detailed input from patient safety experts, advocates and patients. The TEP that provided detailed input on the Patient Safety Structural measure included clinicians and representatives of hospitals and healthcare systems. Because achieving zero preventable harm is part of our National Quality Strategy, [ 312 ] we welcome additional ideas or input in how we can build on our current activities to increase our progress towards this goal.
Comment: A few commenters stated that hospitals are already engaged in efforts using meaningful, measurable data and that this measure would take away from that.
Response: We disagree that the Patient Safety Structural measure will take away from efforts to use meaningful, measurable data to improve patient safety. Several of the attestations in this measure are related to the use of measurable data to inform patient safety improvement. For example, Domain 1 Statement B requires a hospital to attest to whether C-suite leaders oversee the development of specific improvement plans with metrics, Domain 3 Statement C requires a hospital to attest to whether it has a patient safety metrics dashboard; and Domain 3 Statement D includes the high reliability practice of a data infrastructure to measure safety. Therefore, efforts using meaningful, measurable data will likely be applicable to one or more of the attestation statements, such that hospitals already engaged in these efforts will be able to attest positively to one or more statements based on these efforts.
Comment: Some commenters specifically expressed concern regarding what they characterize as a lack of standard definitions for terms within the attestation statements. Specifically, commenters recommended standard definitions for the following terms: (1) senior governing board, (2) regular board agenda, (3) annual leadership performance reviews and compensation, (4) serious safety event, (5) core institutional value, (6) action plan, and (7) free flow of information.
Response: We thank commenters for these recommendations. We have intentionally left these terms undefined within this measure to maintain flexibility to allow each hospital to adopt practices that are most effective for its individual circumstances. However, common definitions currently used by safety experts in the field, which may guide hospitals attesting to this measure, are described in the Attestation Guide, available on the Web-Based Data Collection tab under the IQR Measures page and PCHQR Measures page on QualityNet at both: https://qualitynet.cms.gov/inpatient/iqr/measures#tab2 and https://qualitynet.cms.gov/pch/measures , respectively.
The Attestation Guide provides a description of “serious safety event” (that is, an event judged by the clinical team OR the patient to be “temporary major” or greater). The Attestation Guide also provides more detail and examples of what will be characterized as an event that is greater than “temporary major.” Additionally, the Attestation Guide provides a description of the senior governing board (that is, “the senior governing board is intended to be the body with fiduciary responsibility for the hospital, in charge of resource management, with ultimate authority. The senior governing board may or may not oversee other, subordinate hospital boards and committees”). We monitor measure performance for all of the measures in our quality reporting programs, and if we identify that there is a need for additional guidance, we provide it through our regular education and outreach efforts.
Comment: Some commenters stated that there is a lack of empirical evidence due to insufficient hospital-specific field-testing. Some commenters specifically stated that entity level reliability testing was not performed, performance scores were not reported, and workflow analysis was not conducted.
Response: We acknowledge the commenters' concern about hospital-specific field-testing. Although entity level testing of this measure has not been conducted, we are confident that the foundational principles are sound, and the included specifications are attainable, measurable, and actionable. We refer readers to section IX B(1)(a) and the Background section of this finalized proposal for more discussion of the basis on which we determined this; specifically, we discuss the details of the patient safety guidelines and literature that informed this measure, the TEP input provided, and significant public comment support expressed from expert stakeholders, patients and caregivers. As data are obtained on this measure, we will continue to monitor and evaluate the measure.
Comment: A few commenters recommended deferring public reporting for at least the first year. A commenter stated that the Hospital IQR Program does not usually publicly report scores of new measures and recommended aligning with that approach by delaying public reporting of the Patient Safety Structural measure.
Response: While we do not expect all hospitals to achieve a score of five on the measure, the information collected under the Patient Safety Structural measure will provide valuable information for patients, families, and caregivers, as well as for healthcare researchers, providers and other members of the public. We note that we may delay public reporting for measures with an initial voluntary reporting period, or measures that may require more complicated implementation of data collection processes as is often the case with EHRs (for an example of a measure for which we have delayed public reporting, we refer readers to our adoption of the Hybrid Hospital-Wide Readmission measure ( 84 FR 42465 through 42479 )), but an attestation-based measure does not entail this level of complexity and hospitals have sufficient time to review and prepare an annual attestation that does not entail a large amount of detailed data.
Comment: A few commenters stated that attestation to a structural measure does not provide actionable data because the improvement has already been achieved to be able to positively attest to the measure.
Response: We agree that one value of an attestation measure is to encourage hospitals to update and improve structures so that they can positively attest to the measure. This measure may also serve to differentiate those hospitals that have already fully implemented the best practices identified. We note that an additional benefit of attestation measures is to provide valuable information for patients, families, and caregivers, as well as for healthcare researchers, providers, and other members of the ( print page 69468) public on the distribution of these institutional practices.
Comment: Many commenters expressed concern regarding reporting the measure through NHSN instead of the Hospital Quality Reporting (HQR) system. Some commenters stated that NHSN has operational challenges which could impact hospital reporting. Other commenters stated that this measure would be reported by different personnel than the data currently reported to NHSN and requiring these staff to get access to NHSN would increase the administrative burden.
Response: We recognize that most current quality measures are reported through the HQR System and that because data currently reported through the NHSN are generally related to healthcare-associated infections or vaccinations for healthcare personnel, these data may be collected and reported by different personnel within the hospital (for example, infection control personnel) than those personnel likely to be responsible for reporting the Patient Safety Structural measure. However, because the Patient Safety Structural measure is related to healthcare safety, and the NHSN collects information related to patient safety, reporting of the Patient Safety Structural measure through the NHSN will be most appropriate. We understand some hospitals may want additional staff to obtain access to the NHSN to report the Patient Safety Structural measure. We note that the CDC has streamlined the registration process for new NHSN users in recent years, and the process can often be completed in less than a week. Interested parties may wish to review the NHSN website for details of the latest registration process at: https://www.cdc.gov/nhsn/index.html . The CDC is consistently working to further modernize the NHSN application in a constant effort to improve speed and functionality, as well as the user experience.
In recognition that new users have faced challenges in the past, the CDC plans to open this measure for test access in NHSN several weeks before the submission period opens on April 1, 2026, to ensure new users have ample time to obtain and test their access to the system before the first reporting deadline of May 15, 2026. More details about test access outside of the submission period will be provided by NHSN through guidance at a later date.
Comment: Several commenters recommended developing a monitoring, evaluation, and improvement plan to identify necessary clarifications or modifications for the Patient Safety Structural measure after implementation. A few commenters recommended monitoring for topped-out performance. These commenters also recommended removing the measure if it does not correlate with improved patient outcomes or if patients and families have difficulty interpreting the measure. A few commenters recommended ensuring there are no unintended consequences, such as limiting access or increasing health care costs.
Response: We have a monitoring and evaluation process for both the Hospital IQR Program and the PCHQR Program. Therefore, we intend to monitor and evaluate the performance of the Patient Safety Structural measure in achieving our programmatic goals including encouraging hospital improvement on the measure and providing meaningful information to patients and their families. As part of our monitoring and evaluation efforts we continually evaluate measures for topped-out status, correlation with other measures, and unintended consequences. As we indicated in the proposed rule summary of the PRMR process ( 89 FR 36287 ), as data is obtained, we intend to evaluate the effectiveness of, and the potential to narrow, the future scope of the attestations.
Comment: Some commenters expressed concern that this measure adopts overly prescriptive policies. These commenters stated that it would be preferable to encourage “safety via guided adaptability” instead of “safety via control” activities and stated that encouraging “safety via guided adaptability” would improve innovation in safety. These commenters further stated that public reporting, organizational accountability, and transparency have not worked and therefore adopting a measure based on public reporting would likely be ineffective.
Response: The attestation statements within the five domains of the Patient Safety Structural measure were developed in collaboration with a TEP convened by a CMS contractor and comprised of thought leaders in the field. [ 313 ] Most TEP members agreed that the domains capture the most important elements for advancing patient safety. Furthermore, the measure developers engaged the members of the TEP for their operational and clinical expertise to assure that each domain was actionable and measurable. [ 314 ] Therefore, these domains and attestations are appropriate for encouraging establishment of a structure, culture, and leadership commitment to prioritizing safety. We note that the domains within Patient Safety Structural measure have been designed to be non-prescriptive in how hospitals implement these policies and procedures and therefore provide flexibility for hospitals to establish “safety via guided adaptability” protocols within these domains.
Comment: A commenter recommended that instead of attestation measures, CMS advance the use of Artificial Intelligence (AI) in its electronic measure reporting strategy to increase the availability of data and reduce provider burden and burnout.
Response: While we recognize the future potential of advancing health quality, increasing data availability, and reducing provider burden using AI, this technology has not been sufficiently tested and validated to use for measures in our quality reporting programs.
Comment: A commenter recommended combining multiple structural measures into one multi-dimensional, streamlined measure.
Response: We appreciate the commenter's recommendation to combine multiple structural measures into one multi-dimensional, streamlined measure; however, we are concerned that such a measure may be administratively complex to report and challenging for interested parties to interpret. As we continue to evolve the Hospital IQR Program and the PCHQR Program we will seek to identify ways to streamline our measures, including potentially combining measures.
Comment: Many commenters did not support adoption of the Patient Safety Structural measure because of the belief that the number of attestations is excessive. Some of these commenters stated that this appears to be a survey, not a quality measure because of the number of statements and stated that because it is a survey it is not meaningful in hospital measurement programs.
Response: We disagree with commenters that the number of statements to which hospitals will need to attest is indicative that this measure is a survey because a survey is not ( print page 69469) defined by the number of questions contained within. [ 315 ] Survey research seeks to collect information from a sample of the population through responses to questions to understand the characteristics of a group. [ 316 ] The Patient Safety Structural measure evaluates and publicly reports information about the quality of care in each hospital that participates in the Hospital IQR Program and the PCHQR Program. The Patient Safety Structural measure is not focused on the characteristics of hospitals as a group, but on the patient safety structures of each individual hospital and how that information can lead to patient safety improvement efforts and inform patient choice. We have adopted other structural measures which require hospitals to attest to specific statements to collect information about specific structures (for example, the Hospital Commitment to Health Equity measure finalized in the FY 2024 IPPS/LTCH PPS final rule ( 87 FR 49191 through 49201 )) to provide meaningful information to consumers regarding individual hospital characteristics.
Comment: Many commenters did not support adoption of the Patient Safety Structural measure because it is an attestation measure which does not measure patient outcomes or patient care. Some of these commenters recommended that CMS identify measure gaps related to patient safety in the current quality reporting programs and develop measures to assess these gaps that would provide actionable data. Some commenters stated that CMS has not shown a link between the attestations and improved patient outcomes.
Response: While this measure does not measure patient outcomes or specific activities of patient care, it does assess hospital implementation of a systems-based approach to safety best practices, which is applicable to all patient care activities and patient outcomes. Safety is a foundational aspect of high-quality care for all patients, regardless of their health condition or if they are at risk of experiencing a specific type of potential harm. Our measure inventory currently lacks measures that emphasize the importance of structure, culture, and leadership commitment to prioritizing safety. Therefore, we have identified that there is a patient safety measure gap in our current quality reporting programs. The Patient Safety Structural measure is informed by scientific evidence from existing patient safety research and literature, guidance from established healthcare quality and safety organizations, and detailed input from patient safety experts, advocates and patients. Statement-level review and input of each measure attestation was provided by a national TEP. We reiterate that research shows a link between these hospital characteristics and improved care and outcomes for patients. [ 317 318 ]
Comment: Many commenters did not support adoption of the Patient Safety Structural measure because of the concern that attestation measures are subjective. These commenters stated that because of this subjectivity the Patient Safety Structural measure would not meaningfully distinguish between hospitals.
Response: The Patient Safety Structural measure provides hospitals flexibility in meeting each of the attestations. We recognize that there is significant variation between hospitals and the local communities they serve, which means that policies and procedures that are effective in some hospitals may not be effective in other hospitals. Therefore, the attestations in the Patient Safety Structural measure have been developed to encourage hospitals to adopt policies and procedures consistent with a structure, culture, and leadership commitment to prioritizing safety; without being prescriptive in how hospitals implement these policies and procedures. We acknowledge commenters' concerns regarding the potential for there to be subjectivity in how hospitals interpret each attestation statement within the Patient Safety Structural measure. We have developed and provided the Attestation Guide to limit this subjectivity and to help hospitals accurately attest to this measure while still providing flexibility to hospitals. We further note the Patient Safety Structural measure is one measure within a larger portfolio of measures which balances more narrowly specified measures with broader measures. Some of these more narrowly specified measures specifically address patient safety (such as the measures for Hospital Harm—Falls with Injury (section IX.C.5.c) and Hospital Harm—Postoperative Respiratory Failure (section IX.C.5.d)).
Comment: Many commenters did not support the Patient Safety Structural measure because of concerns that the attestations are difficult to implement and therefore hospitals would not be able to prepare for this measure prior to its adoption. Some commenters recommended delaying implementation of the measure to allow hospitals more time to prepare for reporting. A few commenters recommended revising the measure to focus on two to three items per domain. Some of these commenters recommended gradually increasing the number of attestations in future years.
Response: We understand commenters' concerns that many hospitals will not be able to positively attest to all the statements for each domain prior to the Patient Safety Structural measure's implementation for the CY 2025 reporting period. We do not expect all hospitals to achieve a score of five on the measure, especially not in the first reporting year. This measure is intended to further the current state of patient safety structures within hospitals. [ 319 ] By adopting the measure for the CY 2025 reporting period, we can establish a baseline of the current state of patient safety structures within hospitals, which we can use to understand change as hospitals seek to incorporate more of these practices because of the adoption of the Patient Safety Structural measure. Requiring attestation to just two or three items per domain would not be as effective at encouraging hospitals which have already adopted those patient safety structures to advance the state of patient safety as quickly or effectively as adopting all attestations in the first year.
Comment: Some commenters did not support the Patient Safety Structural measure due to concerns that hospitals already engage in the listed activities and that CMS has not shown that there are gaps in these practices. These commenters specifically stated that Patient and Family Advisory Councils (PFACs), PSO participation, participation in large-scale learning networks, and tracking progress against benchmarks are already common practice. ( print page 69470)
Response: We acknowledge the commenters' concerns; however, the purpose of this measure is in part to uniformly assess whether hospitals perform these activities. Based on the information available to us, there are demonstrated gaps in hospital participation in meaningful data collection, reporting, and learning system activities that would make the uniform evaluation of hospital performance on patient safety improvement activities helpful. For example, hospital adoption of PFACs has waned in recent years, with the 2021 American Hospital Association (AHA) annual survey of over 6,200 U.S. hospitals finding that the number of hospitals with PFACs is 51 percent. [ 320 ] A 2019 OIG report found that only 59 percent of general acute care hospitals participating in Medicare work with a PSO. [ 321 ]
Comment: Several commenters did not support the Patient Safety Structural measure because of concerns regarding the reporting burden. These commenters stated that the benefits of the measure are not sufficient to offset the cost associated with determining and documenting whether a hospital's practices meet each attestation. Some of these commenters expressed concern that the time spent attesting to this measure would take away from patient care and could lead to patient harm.
Response: We understand that there will be administrative burden with understanding each of the attestation statements and determining whether a hospital's patient safety structures are in alignment with the attestation statements. We further recognize that this administrative burden may be greater during the first reporting year as hospitals familiarize themselves with the attestation statements. However, we have concluded that the benefits of this measure justify its costs. Safety is a foundational aspect of high-quality care for all patients, regardless of their health condition or if they are at risk of experiencing a specific type of potential harm. By adopting the Patient Safety Structural measure, we not only assess hospital implementation of a systems-based approach to safety best practices but also promote such implementation. Therefore, the Patient Safety Structural measure has considerable benefit for all patients. While we understand that hospital staff will have to spend time reviewing the attestations and assessing their hospital's safety practices in light of the attestation statements, this activity will further encourage hospitals to understand and implement a systems-based approach to safety best practices, which will in turn improve patient care. We refer the readers to section XII.B.6. of this rule for our estimate of the expected cost for a hospital to report this measure.
Comment: A commenter expressed concern that under-resourced hospitals (such as community and rural hospitals) would face greater challenges documenting and reporting than large hospital systems.
Response: We understand that hospitals with fewer resources for identifying and implementing patient safety best practices may face additional challenges in documenting and reporting their practices. However, safety is a foundational aspect of high-quality care for all patients, regardless of the hospital in which they seek care. The reporting of this measure by all hospitals will provide valuable information and a considerable benefit to patients and encourages hospitals to establish a structural, cultural, and leadership commitment to prioritizing safety. Furthermore, we note that HHS provides resources to assist hospitals in their focus on patient safety including, for example, CMS's Quality Improvement Organization Program [ 322 ] and AHRQ's patient safety resources. [ 323 ]
Comment: Many commenters did not support this measure because of the scoring approach in which hospitals would not receive a point for a domain unless they could positively attest to all statements within the domain. A few commenters recommended allowing a hospital to receive credit for the domain by affirmatively attesting to a smaller number of practices within the domain (three or four, instead of all five). Some commenters recommended providing partial credit and stated that this would improve tracking over time.
Response: We understand commenters' concerns that many hospitals will not be able to positively attest to all the statements for each of the domains, which will affect the hospital's score for the entire domain. Nonetheless, we intentionally chose to score the measure at the domain level, for a score of 0-5, instead of allowing partial credit. The Patient Safety Structural measure assesses hospitals in terms of their systemic approach to safety in five domains. Each action within a domain is an important best practice necessary to achieving a high level of performance through the domain on preventable harm reduction for patients. For this reason, a hospital must attest to all the actions within a domain to receive credit for the domain. We reiterate that we do not expect all hospitals to achieve a score of five on the measure, especially not in the first reporting year. The measure is intended to further the current state of patient safety structures within hospitals. Furthermore, requiring attestation to fewer items per domain would be less effective at furthering the current state of patient safety structures within hospitals that currently implement many important elements for advancing patient safety. The decision to use full point scoring is also intended to keep the level of complexity to a minimum and therefore ease the general public's ability to understand the measure.
Comment: Several commenters stated that the measure results would be difficult for patients, the public, and staff to understand and recommended partnering with patients and families on what data regarding safety culture would be meaningful to them. A commenter recommended education and outreach for the public and healthcare community on the nature and purpose of structural measures. A few commenters recommended publicly reporting the results of this measure with additional granularity to identify quality improvement opportunities.
Response: We note that the measure developer convened a TEP to inform development of the Patient Safety Structural measure. In addition to members of healthcare systems and patient safety experts, more than 50 percent of the TEP consisted of individuals that identified as patients or caregivers, some of whom were also representatives of patient and caregiver advocacy organizations. These TEP members provided input on what would be meaningful to patients and their families. The measure scoring structure was developed with their input. Furthermore, as part of the pre-rulemaking process the Patient Safety Structural measure received a total of 91 comments expressing support. [ 324 ] Most ( print page 69471) commenters were patients and family members who described their individual experiences with the medical system and preventable harms to which they were exposed. These commenters then emphasized the importance of the Patient Safety Structural measure's intent and domains for improving patient safety related to these experiences. [ 325 ] We may consider potential reporting of more granular data that would allow identification of improvement opportunities. We note that each hospital will know how it attested to each statement and therefore would be able to determine which areas would be most appropriate for improvement opportunities.
Comment: A few commenters recommended developing a strategy to publicly report the results of this measure with results from safety outcome measures for comparison.
Response: Section 1886(b)(3)(B)(viii)(VII) of the Act and section 1866(k)(4) of the Act require the Secretary to report quality measures used in the Hospital IQR Program and the PCHQR Program, respectively, on a CMS website. Section 1886(b)(3)(B)(viii)(VII) of the Act and section 1866(k)(4) of the Act for the Hospital IQR Program and the PCHQR Program, respectively, also require that the Secretary establish procedures for making information regarding measures available to the public after ensuring that a hospital has the opportunity to review its data before they are made public. Our current policy is to report data from the Hospital IQR Program and PCHQR Program as soon as it is feasible on CMS websites such as the Compare tool hosted by HHS, currently available at: https://www.medicare.gov/care-compare , or its successor website, after a 30-day preview period ( 78 FR 50776 through 50778 ). We refer readers to section IX.C.12 for more details on our public display requirement policies for the Hospital IQR Program. Consistent with this requirement, we will publicly report the results of the Patient Safety Structural measure on a CMS website on which we also publicly report the results of other measures in the relevant quality reporting programs (either the Hospital IQR Program or the PCHQR Program) including safety outcome measure. We thank commenters for their suggestion and encourage interested parties to access these data for comparison and analysis when they become publicly available.
Comment: Some commenters stated that the existing patient safety measures, supplemented by other patient safety measures proposed in the FY 2025 IPPS/LTCH PPS proposed rule, are sufficient for measuring patient safety. A commenter stated that the CMS Hospital Star Ratings and condition-specific quality measures are already available to help patients make informed care decisions.
Response: We agree that the existing patient safety measures, the Overall Hospital Quality Star Rating, and the condition-specific quality measures are valuable resources to help patients make informed care decisions. This measure is an important complement to these resources. Each of the existing patient safety measures serves an important purpose in assessing a specific element of patient safety, for example, a specific condition, procedure, or harm event (such as falls), but none of the existing patient safety measures provides a holistic view of a hospital's structural, cultural, and leadership commitment to prioritizing safety. These elements are critical aspects of ensuring safety for all patients regardless of their health condition or if they are at risk of experiencing a specific type of potential harm. We refer readers to the CY 2025 OPPS/ASC proposed rule where we are soliciting input on potential future methodological modifications regarding the Safety of Care measure group within the Overall Hospital Quality Star Rating ( 89 FR 59509 through 59515 ).
Comment: Several commenters did not support the Patient Safety Structural measure because it has not been endorsed by a CBE.
Response: While we recognize the value of measures undergoing CBE endorsement review, we need not adopt solely measures endorsed by a CBE (see section IX.B.1.c). Given the urgency of improving patient safety and the current lack of CBE-endorsed measures that address hospital structures for creating a culture of safety, we determined that it is appropriate to adopt this measure. This measure is designed to identify hospitals that practice a system-based approach to safety and embrace the importance of a safety culture. Demonstrating a structural, cultural, and leadership commitment that prioritizes safety can improve care and outcomes for all patients. [ 326 ] Because of the measure's potential to improve care for all patients, we have determined that this is an appropriate topic for a measure for the Hospital IQR Program and the PCHQR Program. We reviewed measures endorsed by both the CBE which currently holds the contract under section 1890(a) of the Act and measures endorsed by the entity which formerly held that contract and did not identify any other CBE-endorsed measures on strategies and practices to strengthen hospitals' systems and culture for safety. In light of the lack of endorsed measures on this specified area or medical topic, we have determined that it is appropriate to use a measure that is not endorsed by the CBE. We intend to submit the measure for future CBE endorsement after endorsement criteria for structural measures have been made available.
Comment: A commenter stated that they do not support the Patient Safety Structural measure and expressed the belief that it is a punitive measure tied to reimbursement.
Response: The Hospital IQR Program is a pay-for-reporting program, which means that hospitals that report the required measure data in accordance with the form, manner, and timing policies specified by the Secretary are not subject to a financial penalty under this program. A hospital's performance on the measure, which for the Patient Safety Structural measure is a score from 0 to 5 points, has no impact on a hospital's Medicare reimbursement. We note that the PCHQR Program is a quality reporting program that does not have a financial penalty associated with it. The measure determines the level at which hospitals are performing these identified best practices and identifies opportunities for improvement in structural safety practices. We do not expect all hospitals to achieve a maximum score of five points on the measure, especially during the initial years of using the measure in these programs.
Comment: Several commenters supported Domain 1 because of the goal of eliminating preventable harm. A commenter expressed support for Domain 1 Statement B, and specifically supported requiring hospitals to attest that their “specific plans and metrics are widely shared”. A few commenters expressed support for Domain 1 Statement C. These commenters stated that leadership engagement would empower leadership to respond expeditiously. A few commenters expressed support for Domain 1 Statement D, stating that it is important to encourage hospitals to use board meetings to discuss patient safety topics ( print page 69472) because of the commenters' belief that this is not currently a widespread practice. A few commenters expressed support for Domain 1 Statement E; some of these commenters expressed the belief that it is important for senior leaders to learn of patient safety events through internal channels as close to real-time as possible, even though the event review may not be complete until after the notification.
Response: We thank the commenters for their support of Domain 1 and specifically Domain 1 Statements B, C, D, and E. We appreciate commenters' support for leveraging board meetings, developing targeted patient safety plans and metrics, and notifying senior leaders early during serious safety events. Domain 1 includes core activities that place patient safety at the forefront of governing boards and executive leadership's priorities for greater leadership accountability and prioritization in operational, financial, and strategic plans.
Comment: A few commenters recommended linking the language describing Domain 1 Statements A and B with the statement in the Attestation Guide that “no preventable harm” is a long-term goal (which is currently associated with Domain 2 Statement A).
Response: We thank commenters for this suggestion. While Domain 1 is titled, “Leadership Commitment to Eliminating Preventable Harm,” Domain 1 Statements A and B do not include the phrases “no preventable harm” or “zero preventable harm.” Domain 2 Statement A does include this phrase, and thus Domain 2 Statement A remains an appropriate place for this language. We intend to monitor performance on the Patient Safety Structural measure and, if we identify that there is a need for additional guidance, provide it through our regular education and outreach efforts.
Comment: A few commenters recommended ensuring accurate attestation to this measure by developing an audit plan.
Response: We understand commenters' concerns regarding the accuracy of provider self-reported data and are continuously evaluating new ways to ensure accurate information is submitted to CMS and shared with the public. We do require all hospitals participating in the Hospital IQR Program and PCHs participating in the PCHQR Program to complete the Data Accuracy and Completeness Acknowledgement (DACA) each year, which requires an annual attestation that all the information reported to CMS for these respective programs is accurate and complete to the best of the submitters' knowledge because CMS expects all hospitals to submit complete and accurate data with respect to quality measures. For more information on the Hospital IQR Program's DACA requirements, we refer readers to section IX.C.11. of this final rule. For more information on the PCHQR Program's DACA requirements, we refer readers to 42 CFR 412.24(c) .
Comment: A few commenters recommended updates to Domain 1. A few commenters recommended ensuring that hospitals include physicians and medical staff as part of operational and strategic planning. A commenter recommended changing the name to “Leadership Commitment to Eliminating Preventable Harm and Creating an Environment of Ongoing Safety” because the absence of measurable preventable harm does not always indicate a safe environment. A commenter recommended revising the language of Domain 1 to remove references to “eliminate” and replacing these references with “engineer out” to focus on the multi-disciplinary approach to improving safety.
Response: We thank commenters for their recommended updates to Domain 1. We note that statement-level review and input of each measure attestation was provided by a national TEP. Most TEP members agreed that the domains capture the most important elements for advancing patient safety. [ 327 ] Furthermore, the Patient Safety Structural measure is intended to provide hospitals flexibility in meeting each of the attestations. We recognize that there is significant variation between hospitals, which means that policies and procedures that are effective in some hospitals may not be effective in other hospitals. Therefore, the attestations in the Patient Safety Structural measure have been developed to encourage hospitals to adopt policies and procedures consistent with a structure, culture, and leadership commitment to prioritizing safety, without being prescriptive in how hospitals implement these policies and procedures including specifying the staff responsible for engaging in operational and strategic planning.
With respect to the specific recommendations raised by commenters, we agree that the absence of measurable preventable harm does not always indicate a safe environment but note that the domain refers to the elimination of preventable harm, regardless of whether and how that harm is measured. We disagree with the commenter that the term “engineer out” more effectively conveys a multidisciplinary approach to safety than the term “eliminate.” We note that the term “engineer out” could be interpreted to compartmentalize responsibility for reducing preventable harm in a way that implies that the staff responsible for developing and implementing processes have responsibility for safety to the exclusion of staff responsible for other functions (such as staff responsible for operations).
Comment: A few commenters recommended updates to specific attestation statements within Domain 1. These recommendations were:
- Statement A: Remove language related to annual performance reviews and compensation because of concerns that annual performance reviews are too infrequent to motivate change and that there may be unintended consequences (such as funding going to executive bonuses instead of safety initiatives).
- Statement B: Require each unit or department to set and publicly share a goal which supports the plans and metrics.
- Statement C: Require hospitals to attest that they embed patient safety into everyday clinical operations instead of that they ensure adequate resources to support patient safety.
- Statement D: Increase flexibility with respect to the percentage of regular board meetings focused on safety to allow quality and patient safety subcommittees to meet the requirement, to reduce the 20 percent threshold dedicated to these topics during board meetings, and to not disadvantage hospitals that are required to have open board meetings (that is, public hospitals).
- Statement E: Shorten the timeframe for reporting serious safety events to the board. Require reporting of employee injuries to C-suite executives within 24 hours.
Response: We thank commenters for these recommended updates. We reiterate that the Patient Safety Structural measure was developed with input from national experts to allow hospitals flexibility in how they meet each individual statement. With respect to Domain 1 Statement A, we understand commenters' concern that annual activities may be too infrequent for active learning, review, reprioritization, and problem solving. Often annual performance reviews include regular status checks ( print page 69473) throughout the year to ensure progress and address barriers to achieving goals to allow for improved active learning, review, reprioritization, and problem solving. While we recognize it is possible that some hospitals or health systems may allocate funding that had previously been used for patient safety to increase executive pay due to the attestation in Domain 1 Statement A, we note that this will be reflected in other attestations in the Patient Safety Structural measure, such as Domain 1 Statement C, which requires attestation to whether the hospital governing board, in collaboration with leadership, ensures adequate resources to support patient safety (such as equipment, training, systems, personnel, and technology).
We agree with the commenter that setting and publicly sharing a goal which supports the plans and metrics would be a way of ensuring that these plans and metrics are widely shared across the hospital and note that hospitals will have flexibility to implement this policy under Domain 1 Statement B.
We note that Domain 1 refers to the Leadership Commitment to Eliminating Preventable Harm and that Statement C requires hospitals to attest to whether their hospital governing board, in partnership with leadership, ensures adequate resources to support patient safety. While ensuring adequate resources is a function of the governing board, in partnership with leadership, embedding patient safety into everyday clinical operations is a shared responsibility across the entire hospital workforce. Therefore, the recommended statement exceeds the scope of Domain 1.
With respect to Domain 1 Statement D, we note that there is support for a 20 percent threshold for hospital leadership and board meetings in the Self-Assessment Tool created to complement the recommendations in Safer Together: A National Action Plan to Advance Patient Safety. In support of the report's recommendation to ensure safety is a “demonstrated core value,” the National Steering Committee for Patient Safety provided the suggestion that hospitals could “[a]llocate and evaluate the effectiveness of time spent in leadership meetings and all board meetings to address quality and safety and share patient and family experiences with staff, leaders, and board members” as a means of achieving this recommendation. In the Self-Assessment Tool, this committee of patient safety stakeholders and experts targeted 20 percent as the allocation of time that should be dedicated to these topics during leadership and board meetings, under the heading of “Culture, Leadership, and Governance,” awarding a score of 3 or 4 (of 4) for hospitals at which “At least 20 percent of all leadership and board meeting agendas are dedicated to review and discussion of safety.” [ 328 ] We understand commenters' concerns that public or government owned hospitals may be required to hold open board meetings. We note that there are benefits to open meetings including transparency, maintaining a close relationship with interested parties, generating trust, and fostering openness and accountability. [ 329 ] We understand that some specific patient safety discussions such as those that include identifiable or protected information may be more appropriate for closed sessions. However, the patient safety benefits of allocating at least 20 percent of leadership and board meetings to patient safety topics extend to both public and private hospitals and warrant the inclusion of Domain 1 Statement D. Furthermore, while we understand that many hospitals have quality and patient safety subcommittees, because of the importance of safety, the awareness, discussion, and responsibility for understanding safety issues in the organization ultimately rests with the organization's leaders with fiduciary responsibility for the hospital. We encourage hospitals to address this attestation by integrating patient safety into other topics during board meetings where appropriate. We reiterate that there are no financial penalties for hospitals that attest either “yes” or “no” to each of the attestation statements to calculate the 0-5-point score for the Patient Safety Structural measure.
With respect to Domain 1 Statement E, we agree with the commenter that it may be appropriate in some situations to notify C-suite executives and individuals on the governing board within 24 hours of employee injuries. However, we wanted to provide hospitals with some flexibility and did not identify a 24 hour deadline as a top priority for safety during our extensive literature review and interaction with patient safety experts, advocates, and patients. We support earlier reporting to the board for serious safety events and recognize that some state and local laws may require more immediate reporting. We reiterate that the Patient Safety Structural measure is intended to provide hospitals flexibility in meeting each of the attestations.
Comment: A commenter requested clarification on Domain 1 Statement A, specifically regarding whether the performance review needs to cite specific metrics or only requires implementation of initiatives to improve quality and safety.
Response: With respect to the elements of annual leadership performance reviews we have intentionally maintained flexibility to allow each hospital to adopt practices that are most effective for its individual circumstances; that is, we have not included a requirement that the performance review cite specific metrics. We monitor performance on all of the measures in our quality reporting programs and, if we identify that there is a need for additional guidance we provide it through our regular education and outreach efforts.
Comment: A commenter recommended that leaders should be coached to improve performance on patient safety and patient safety structures, not penalized for poor performance.
Response: We agree with the commenter that coaching is an effective means to improve performance. The Hospital IQR Program is a pay-for-reporting program, which means that hospitals that report the required measure data in accordance with the form, manner, and timing policies specified by the Secretary are not subject to a financial penalty under this program, and the PCHQR Program is a quality reporting program that does not have a financial penalty associated with it. Therefore, there are no financial penalties for hospitals that attest either “yes” or “no” to each of the attestation statements to calculate the 0-5-point score for the Patient Safety Structural measure. Therefore, there are no financial penalties associated with hospital performance on the Patient Safety Structural measure. However, the commenter may be referring to the Domain 1 Statement A attestation, which requires hospitals to attest whether “Our hospital senior governing board prioritizes safety as a core value, holds hospital leadership accountable for patient safety, and includes patient safety metrics to inform annual leadership performance reviews and compensation.” This attestation is not prescriptive in how the governing board should hold hospital leadership accountable for patient safety. Hospitals ( print page 69474) retain the flexibility to use positive reinforcement strategies such as coaching and incentives.
Comment: Many commenters did not support Domain 1 Statement E and expressed concern that the three-day timeline for notifying senior leaders of patient safety events would not provide adequate time for such notice. Many commenters stated that the three-day timeline was insufficient for serious events because of the complexity of analyzing the event and providing recommendations. Some of these commenters stated that reporting within three days would lead to superficial reviews which could lead to missed opportunities for meaningful impact and long-term improvement. A few commenters stated that the C-suite's role does not include operational oversight or root cause analysis and stated that presenting information to the board without an actionable plan may not be meaningful. A few commenters stated that this attestation contradicts the CoPs, specifically, 42 CFR 482.21(e)(3) , which provides executives with the authority to set clear expectations for safety. These commenters stated that these clear expectations include the authority to set appropriate timelines and processes for reporting and therefore setting a specific timeline is contradictory. A commenter stated that the three-day deadline is not supported by safety science. Another commenter stated that their state requires reporting to the state within 15 days and recommended deferring to state timeframes when available.
Response: We acknowledge the commenters' concern about a three-day timeline for notifying senior leaders of patient safety events; however, there is support for timely notification of hospital senior leadership in the Self-Assessment Tool created to complement the recommendations in Safer Together: A National Action Plan to Advance Patient Safety. In the Self-Assessment Tool, the National Steering Committee for Patient Safety, under the heading of “Culture, Leadership, and Governance,” determined it was appropriate to award a score of 3 or 4 (of 4) for hospitals at which “The CEO and Board Chair are notified within 24 hours of a serious adverse event.” [ 330 ] To allow flexibility, consistent with the CoPs, and provide hospitals the ability to set the practices that make the most sense for their individual circumstances, the timeframe identified in Domain 1 Statement E extends the timeframe from 24 hours to three business days. This will provide hospitals more time to report confirmed serious safety events while retaining a timely approach for informing hospital and board leadership. Because hospitals still retain flexibility within the three-day period and for setting other expectations with respect to safety, this is not contradictory to the CoP. Furthermore, we reiterate that the Patient Safety Structural measure builds upon the activities already required under the CoPs, [ 331 ] therefore hospitals that do not choose to require executives be notified within 3 days of safety events can attest no to Domain 1 Statement E.
Comment: A few commenters expressed support for Domain 2 Statement A because the commenters stated that setting a goal of zero harm is important to establish a culture that prioritizes safety. A few commenters expressed support for Domain 2 Statement C. These commenters stated that a “just culture” is foundational to patient safety and that, although training and collaboratives to produce a “just culture” exist, hospitals have been slow to implement these activities. A commenter expressed support for Domain 2 Statement D because of the importance of safety skills and competency assessments. A commenter expressed support for Domain 2 Statement E because of the commenter's concern about the risk of workplace violence.
Response: We appreciate the commenters' support for Domain 2 Statements A, C, D, and E. We agree that setting a goal of zero preventable harm, establishing a “just culture”, requiring safety competence assessments, and mitigating workplace violence are all critical activities to hospitals establishing a culture of safety. These activities ensure that patient safety is a core value throughout an organization's strategy and policies.
Comment: A few commenters recommended removing the phrase “zero preventable harm” from the description of Domain 2. These commenters stated that eliminating preventable harm should be a continuous aspirational aim instead of a part of a strategic plan that is regularly updated. Some commenters stated that use of the phrase “zero preventable harm” can lead to under reporting due to concern about “breaking the streak” or overly complex efforts to determine whether an event was preventable. A few commenters recommended setting the goal of year-over-year improvement in rates of preventable harm to avoid gaming of the strict definition of zero preventable harm (that is, defining events as non-preventable or not reporting them).
Response: We thank the commenters for their recommendation to remove the phrase “zero preventable harm.” The inclusion in the Patient Safety Structural measure of a goal of zero preventable harm was informed by extensive input from a TEP, and directly reflects the CMS National Quality Strategy Goal for Safety to “Achieve zero preventable harm.” [ 332 ] While we agree that a year-over-year measurement approach may be attractive, it would not be appropriate to track improvement rates under the structure of this measure. We note that existing measures in the Hospital IQR Program and the PCHQR Program require reporting outcome data in the care environment.
Comment: A few commenters recommended updates to specific attestation statements within Domain 2. These recommendations were:
- Statement A: Remove the term “core value” because core values are often differentiating factors, especially for faith-based organizations, and safety should be a universal aspiration integrated into all operations instead of creating value statements.
- Statement B: Add the phrase “either alone or in partnership with community organizations” after the phrase “identify and address” to reflect that many hospitals cannot address disparities in their communities due to resource limitations.
- Statement C: Expand to include a focus on psychological safety as part of a “just culture.” Discuss the proactive use of “just culture” frameworks as part of intentional system design. Reword to read, “Our hospital has written policies and protocols to cultivate a just culture that emphasizes learning, where frontline staff feel safe to speak up about the challenges they face in delivering care. In turn, hospital leadership demonstrates a commitment to those staff who take the time to report concerns by supporting those that experience errors, coaches well-intentioned but misaligned actions, and seeks corrective action for conscious, reckless choices that do not align with our organizational value” to align with a systems view of safety. ( print page 69475)
- Statement D: Include a patient safety curriculum and competency assessment for all employees and all medical staff regardless of their role or where they work in the system to show that all employees are valued as part of achieving patient safety. Include examples of curricula and competencies to improve workforce safety and provide guidance on measuring these competencies. Rephrase to focus on advancing human factors and systems design improvements instead of specifically advancing skills and behaviors. Exclude C-suite executives and governing board members from the patient safety curriculum and competencies due to concerns that by including executive leaders in the training the training would be too high level to focus on operational issues affecting care providers.
- Statement E: Include “safety for all” instead of more narrowly focusing on workforce safety. Also address fatigue, mental health, and emotional wellbeing.
Response: We thank commenters for these recommended updates. We reiterate that the Patient Safety Structural measure was developed with input from national experts to allow hospitals flexibility in how they meet each individual statement. We recognize that some hospitals, including faith-based organizations, may have other contexts for using the term “core value.” Therefore, we have intentionally not provided guidance or definitions for how a hospital would incorporate patient safety as a “core institutional value.”
We agree with the commenter that hospitals cannot address disparities in their communities without partnership with outside organizations. We note that Domain 2 Statement B specifically asks hospitals to attest whether their “hospital safety goals include the use of metrics to identify and address disparities in safety outcomes based on the patient characteristics determined by the hospital to be most important to health care outcomes for the populations served.” We note that in the Attestation Guide for Domain 2 Statement B we recommend potential harm indicators that hospitals can use to identify safety outcomes. [ 333 ] These harm indicators include the AHRQ Patient Safety Indicators (PSI), which include items such as “Death Rate in Low-Mortality Diagnosis Related Groups,” “Pressure Ulcer Rate,” and “Death Rate Among Surgical Inpatients with Serious Treatable Conditions,” among others, [ 334 ] which are more significantly affected by hospital policies, procedures, and operations than patient risk factors or disparities within the community. Nevertheless, we recognize that there may be community factors which can increase some patients' risks for certain adverse safety outcomes and that it is appropriate for hospitals to work with community organizations to address those factors. We note that hospitals will be able to attest “yes” to this statement regardless of whether they partner with community organizations to identify and address disparities.
We agree with commenters that the elements of a “just culture” would increase the likelihood of establishing an environment conducive to psychological safety. Specifically, because a “just culture” recognizes that individual practitioners should not be held accountable for system failings over which they have no control and recognizes that many individual or “active” errors represent predictable interactions between human operators and the systems in which they work, [ 335 ] a “just culture” can create an atmosphere in which there is a shared belief that it is permissible to express ideas, ask questions, and admit mistakes without a fear of negative consequences. [ 336 ] Domain 2 Statement C's reference to a “just culture” therefore necessarily incorporates considerations of psychological safety.
We agree with the commenter that the use of “just culture” frameworks is part of intentional system design. We address the role of “just culture” frameworks as part of intentional system design in the Attestation Guide by stating that, “a just culture recognizes that individual practitioners should not be held accountable for system failings over which they have no control. A just culture also recognizes many individual or `active' errors represent predictable interactions between human operators and the systems in which they work.”
The current language for Domain 2 Statement C is, “Our hospital has implemented written policies and protocols to cultivate a “just culture” that balances no blame and appropriate accountability and reflects the distinction between human error, at-risk behavior, and reckless behavior.” AHRQ defines a “just culture” as a system that holds itself accountable, holds staff members accountable, and has staff members that hold themselves accountable. [ 337 ] Furthermore, the Attestation Guide states that a “just culture” “recognizes that individual practitioners should not be held accountable for system failings over which they have no control. A just culture also recognizes many individual or `active' errors represent predictable interactions between human operators and the systems in which they work. However, in contrast to a culture that touts `no blame' as its governing principle, a `just culture' does not tolerate conscious disregard of clear risks to patients or gross misconduct (for example, falsifying a record, performing professional duties while intoxicated).” This guidance on “just culture” provides an explanation of the practical application of a “just culture” in balancing accountability with encouraging staff to feel safe in addressing challenges they face.
With respect to Domain 2 Statement D, we agree with commenters that including all employees, even those who do not work in the hospital, in training with respect to a patient safety curriculum or competency assessment has the potential to show that they are valued members of the patient safety team. We note that this statement does include all clinical and non-clinical staff at the hospital to cover all hospital-based employees. Hospitals and health systems that choose to include additional employees in the patient safety training and competency assessments will be able to do so.
We agree that human factors and system design improvements are a vital part of advancing patient safety. As stated in the Attestation Guide, the development of the Patient Safety Structural measure is anchored in best practices and evidence for improving patient safety and reducing harm using a total systems framework that views patient safety events as a result of system failure rather than individual error and encourages a systems approach which takes the view that most errors reflect predictable human failings in the context of poorly designed systems. [ 338 ] However, even within a well-designed system, individuals responsible for processes ( print page 69476) need to have sufficient competencies to fulfill their responsibilities. Therefore, we designed Domain 2 Statement D to complement the other systems-based attestations within the Patient Safety Structural measure.
We agree with commenters that patient safety curriculum and competencies for clinical and non-clinical staff would vary based on role. To avoid hospitals using trainings that are so high level that they do not address operational issues affecting care providers we refer readers to the following examples of validated, industry-standard trainings and competencies, which are also included in the Attestation Guide:
- ComprehensiveUnitbased Safety Program (CUSP); [ 339 ]
- AHRQ's Communication and Optimal Resolution (CANDOR) toolkit; [ 340 ]
- The CDC's Infection Control Assessment and Response program and tool; [ 341 ]
- TeamSTEPPS communication framework; [ 342 ]
- Institute for Healthcare Improvement's Root Cause Analyses and Action (RCA [ 2 ] ) resources; [ 343 ]
- Shared Decision Making; [ 344 ]
- Tools to reduce central line infections; [ 345 ]
- Utilization of data analytics;
- Performance improvement methodologies such as PlanDoStudyAct; [ 346 ] and
- Ethical standards.
To allow hospitals flexibility with respect to the curricula they have selected and the competencies that they have determined are most critical to develop within their hospital we have intentionally maintained flexibility to allow each hospital to develop their own competency assessments. We intend to monitor measure performance on the Patient Safety Structural measure and, if we identify that there is a need for additional guidance, provide it through our regular education and outreach efforts.
The statement to which hospitals will attest for Domain 2 Statement E is: “Our hospital has an action plan for workforce safety with improvement activities, metrics and trends that address issues such as slips/trips/falls prevention, safe patient handling, exposures, sharps injuries, violence prevention, fire/electrical safety, and psychological safety.” We note that the list of potential workforce safety elements is intended to be a set of examples, and not a comprehensive list of items affecting workforce safety.
The intent of the Patient Safety Structural measure is to identify hospitals that exemplify a culture of safety and leadership commitment to transparency, accountability, patient and family engagement, and continuous learning and improvement to advance our goal of achieving zero preventable harm. We have focused Domain 2 Statement E on workforce safety, instead of the broader “safety for all” recommended by the commenter because workforce safety is a precondition to advancing patient safety with a unified, total systems-based approach to eliminate harm to both patients and the workforce. [ 347 ] We note that hospitals maintain the flexibility to address “safety for all” under this attestation, as long as they include a focus on workforce safety within their broader strategy for “safety for all.”
Comment: Some commenters stated that introducing competency assessments for Domain 2 Statement D would be costly and administratively burdensome.
Response: We understand that hospitals that do not currently have a structure in place for assessing staff competency with respect to patient safety would need to develop and implement a plan for these assessments to attest “yes” to this statement. While we acknowledge that adopting this practice may entail costs for such hospitals, the value of implementing these assessments justifies the investment required. We reiterate that we do not expect all hospitals to achieve a score of five on the measure, especially in the first year. Hospitals that are not able to adopt these assessments can attest to the other domains as appropriate and receive a score of 0-4 without any financial penalties.
Comment: A commenter expressed support for Domain 3 because of the importance of creating a culture of patient safety in improving patient safety. A few commenters supported Domain 3 Statement A and stated that safety culture surveys are another established tool to provide feedback to leaders. A commenter expressed support for Domain 3 Statement C because of the use of external benchmarks. A few commenters supported Domain 3 Statement E because learning collaboratives are a proven way to spread innovation and improve outcomes.
Response: We thank the commenters for their support of Domain 3, specifically Domain 3 Statements A, C, and E. We appreciate the commenters' support for activities that would foster a culture of safety and continuous learning systems, including external benchmarking, using tools to provide feedback to leaders, and participation in learning collaboratives. We note that culture of safety surveys have been shown to provide feedback to leaders and, with a structured debrief, to have a positive effect on working patterns and safety culture. [ 348 ] These activities support evidence-based practices that encourage hospital-wide approaches to addressing safety.
Comment: A few commenters recommended updates to specific attestation statements within Domain 3. These recommendations were:
- Statement A: Emphasize that the culture of safety survey should include psychological safety.
- Statement B: Clarify whether an analysis needs to be completed for every event or cluster, and encourage hospitals to perform analyses for events that do not reach the threshold of serious safety events. A commenter also recommended taking an approach of ( print page 69477) prospective evaluation when things go correctly.
- Statement C: Transition from benchmarking on external metrics to benchmarking on zero harm to encourage high performing hospitals to continue to improve. Reframe safety as the capacity to make things go right instead of a count of the number of things that go wrong.
- Statement D: Require all the practices listed because they are interconnected and rely on one another. Rephrase as “characteristics of a learning and improving organization” because high reliability is a framework but some of the elements included in the list are improvement methods. Refine the high reliability practices as follows:
++ Require safety and process improvement huddles to start at the unit level, cover all shifts, and roll up on Monday—Friday with additional safety huddles on weekends;
++ Require hospital leaders to participate in weekly safety related rounding to provide sufficient visibility;
++ Encourage coaching leaders between the care delivery personnel and executives on leading for safety and process improvement;
++ Expand from training on team communication and collaboration to include other training elements (specifically, personal habits and skills to reduce lapse, cognitive bias training, and tools to reduce bias);
++ Reframe training on team communication and collaboration to incorporate patient safety principles into leadership training to avoid over reliance on online modules;
++ Add a statement regarding a standard daily process improvement method for every work;
++ Add a statement regarding leadership skills training in leading a team to 100 percent performance on safety practices; and
++ Refine the description of defined improvement methods to ensure that the method is appropriate to the problem(s) being solved.
- Statement E: Remove this statement because many large-scale learning networks are not PSOs and the commenters state that sharing data with these networks would be a violation of the Patient Safety and Quality Improvement Act of 2005 (PSQIA) ( Pub. L. 109-41 ) for hospitals that report to PSOs.
Response: We note that Statement A in Domain 3 requires hospitals to attest to whether they “conduct a hospital-wide culture of safety survey using a validated instrument annually.” In the Attestation Guide for this measure, we provide two examples of validated instruments which hospitals can use to conduct this survey. [ 349 ] The AHRQ Survey on Patient Safety Culture includes 10 composite measures, including “Response to Error,” “Communication About Error,” and “Communication Openness.” [ 350 ] The Safety Attitude Questionnaire includes statements such as, “It is easy for personnel here to ask questions when there is something that they do not understand,” “In this clinical area, it is difficult to discuss errors,” and “Morale in this clinical area is high.” [ 351 ] Therefore, while neither of these survey instruments specifically have a domain related to psychological safety, key elements of psychological safety are embedded in the existing domains and questions. While hospitals may choose to use other surveys, we intend the Patient Safety Structural measure to maintain flexibility to allow each hospital to adopt practices that are most effective for its individual circumstances.
Domain 3 Statement B requires hospitals to attest whether they have a “dedicated team that conducts event analysis of serious safety events using an evidence-based approach, such as the National Patient Safety Foundation's Root Cause Analysis and Action.” This statement does not introduce limitations or requirements on the activities of the dedicated team; instead, it requires hospitals to attest “yes” or “no” as to whether the hospital has such a team. We agree with commenters that it would be worthwhile for these teams to also evaluate events that do not reach the threshold of serious safety events and to seek best practices from situations in which everything went correctly. However, we did not identify this as a top priority for safety during our extensive literature review and interaction with patient safety experts, advocates, and patients and therefore we are not currently requiring hospitals to attest to whether their dedicated teams evaluate these situations. We will monitor performance on the Patient Safety Structural measure, and if we identify opportunities for additional improvements in patient safety structures, we will consider these in future refinements.
As more hospitals begin to benchmark based on external metrics and use those metrics to identify best practices, we anticipate that national performance on patient safety will improve. This improved national performance would, in turn, lead to a higher standard of patient safety in the external metrics that hospitals are using for benchmarking. High performing hospitals could benchmark on zero harm in addition to analyzing their performance on external metrics to further drive quality improvement.
Consistent with AHRQ guidance, we consider patient safety to refer to freedom from accidental or preventable injuries produced by medical care. [ 352 ] This includes both proactively following practices that ensure positive outcomes as well as implementing practices or interventions that reduce the occurrence of preventable adverse events. [ 353 ]
The high reliability practices listed under Domain 3 Statement D reflect practices of high reliability organizations. These are organizations or systems that operate in hazardous conditions but have fewer adverse events than comparable organizations. With respect to patient safety, high reliability organizations are considered to operate with nearly failure-free performance records, not simply better than average ones. [ 354 ] Therefore, these practices are not inherently interrelated; they are practices that are associated with consistently high performance. While we agree that implementing all of the listed practices would advance a hospital's commitment to patient safety, for the purposes of the Patient Safety Structural measure, a hospital may attest “yes” to Domain 3 Statement D as long as it has implemented at least four of the listed practices. Because the items listed under Domain 3 Statement D are practices associated with high reliability organizations it is appropriate and accurate to refer to them as high reliability. High reliability hospitals achieve safety, quality, and efficiency goals by applying five key principles: (1) sensitivity to operations (that is, heightened awareness of the state of relevant systems and processes); (2) ( print page 69478) reluctance to simplify (that is, acceptance that work is complex, with the potential to fail in new and unexpected ways); (3) preoccupation with failure (that is, to view near misses as opportunities to improve, rather than proof of success); (4) deference to expertise (that is, to value insights from staff with the most pertinent safety knowledge over those with greater seniority); and (5) practicing resilience (that is, to prioritize emergency training for many unlikely, but possible, system failures). [ 355 ] The listed items are consistent with the practices of these organizations and thus reflect appropriate and effective high reliability practices. We agree with commenters that a leadership process improvement method for every work unit and leadership skills training in leading a team to 100 percent performance are both practices with the potential to further improve a hospital's culture of safety; however, we did not identify them as consistent practices across organizations identified as high reliability organizations.
With respect to the concern that by submitting data to a PSO, hospitals would no longer be able to work with another large-scale learning network, we disagree. Some information becomes patient safety work product (PSWP) protected under the PSQIA when that information is submitted to a PSO. We refer readers to 42 CFR 3.20 for information regarding what constitutes PSWP. Depending on the individual circumstances of a hospital that chooses to work with both a PSO and a large-scale learning network that is not a PSO, the hospital could disclose other, non-PSWP information to the learning network or disclose non-identifiable PSWP consistent with its obligations under the PSQIA. Therefore, we are not modifying Domain 3 Statement E because there is sufficient flexibility for hospitals to work with large-scale learning networks of their choosing.
Due to commenters' concerns regarding Domain 4 Statement B, discussed in detail below, we are modifying the attestation in Domain 4 Statement B, to the following, “Our hospital voluntarily works with a Patient Safety Organization listed by the Agency for Healthcare Research and Quality (AHRQ) [ 356 ] to carry out patient safety activities as described in 42 CFR 3.20 , such as, but not limited to, the collection and analysis of patient safety work product, dissemination of information such as best practices, encouraging a culture of safety, or activities related to the operation of a patient safety evaluation system.” A hospital could positively attest to this revised statement even if it chooses to work with a large-scale learning network that is not a PSO to analyze and understand patient safety events and with a PSO for other patient safety activities. We note that if a hospital chooses to work with a large-scale learning network other than a PSO, information disclosed to that network does not become PSWP and is not subject to the confidentiality and privilege protections established by the PSQIA.
Comment: A commenter recommended creating a sense of urgency to improve workforce wellbeing because many staff do not feel heard and an annual survey, as described in Domain 3 Statement A, would not improve that.
Response: While we agree that an annual survey, on its own, may not be sufficient to lead to a culture that encourages staff to express their concerns, this is not the only attestation in the Patient Safety Structural measure that addresses workforce safety and workforce wellbeing. Specifically, Domain 2 Attestation E focuses on the development of an action plan for workforce safety, and includes psychological safety as a potential area for improvement. Additionally, a positive attestation on Domain 2 Attestation C would require policies and protocols to cultivate a “just culture,” the cultivation of which can create an atmosphere in which there is a shared belief that it is permissible to express ideas, ask questions, and admit mistakes without a fear of negative consequences. [ 357 ] An atmosphere in which it is permissible to express ideas and ask questions would also be an atmosphere which would improve staff feelings of being heard.
Comment: A few commenters recommended establishing a method to ensure that use of dashboards and benchmarking in Domain 3 Statement C does not allow staff to overlook harm, including potential harm, because the specific harm was not included on the dashboard.
Response: We agree with commenters that it is important that staff does not overlook harm or potential harm that does not meet the threshold or criteria for specific dashboards. We note that the Patient Safety Structural measure is comprised of interrelated domains and attestations. Therefore, while harm or potential harm may not be identified because of its presence on a specific dashboard, activities to support the other attestations (including having a dedicated team that conducts event analysis, adopting high reliability practices, participating in large-scale learning networks, and engaging patients and their families as co-producers of safety and health) could minimize the likelihood that harm or potential harm is overlooked.
Comment: A few commenters recommended providing information on the purpose and activities of a huddle, including clarifying that it is not just to encourage situational awareness but also to identify ongoing risk to patients and to engage leadership support, as needed, to improve consistent attestation for Domain 3 Statement D.
Response: The first high reliability practice identified in Domain 3 Statement D describes “[t]iered and escalating ( e.g., unit, department, facility, system) safety huddles at least 5 days a week, with 1 day being a weekend, that include key clinical and non-clinical ( e.g., lab, housekeeping, security) units and leaders, with a method in place for follow-up on issues identified.” In the Attestation Guide, we provide further information on the purpose and structure of a “tiered and escalating huddle” system including clarifying that the purpose is to identify potential risk and engage leadership support. We specifically state that “`tiered and escalating huddle' system involves a series of brief, focused conversations, typically daily, that rapidly identify and escalate safety, quality, and operational issues from a broad array of frontline staff to a focused group of senior leaders.” [ 358 ]
Comment: A few commenters recommended providing additional guidance on how to use electronic medical records (EMRs) to identify and track safety events for the data infrastructure-related high reliability practice within Domain 3 Statement D.
Response: While we agree with commenters that examples can be meaningful and provide hospitals with information to help adopt these ( print page 69479) evidence-based practices, we also intend the Patient Safety Structural measure to maintain flexibility to allow each hospital to adopt practices that are most effective for its individual circumstances. Specifically, with respect to the use of EMRs to identify and track safety events, flexibility is appropriate so each hospital can work with its EMR vendor to establish the best methods for using the hospital's system to identify and track safety events. We note as an example that EMR-based tracking of healthcare-associated infections and other harms that hospitals are already collecting for reporting to the CDC's National Healthcare Safety Network or electronic clinical quality measures (eCQMs) used in the Hospital IQR and Promoting Interoperability Programs, respectively, could be part of an electronic data infrastructure to measure safety. Hospitals could similarly identify and track additional safety events that reflect the hospital's prioritized areas of focus and improvement.
Comment: A commenter recommended clarifying that hospital staff are not responsible for device and equipment selection and therefore the high reliability practice of incorporating human factors engineering in device, equipment, and process design listed within Domain 3 Statement D would be more appropriate at the board level.
Response: We note that the referenced high reliability practice in Domain 3 Statement D describes “[t]he use of human factors engineering principles in selection and design of devices, equipment, and processes.” We agree with commenters that hospital staff are usually not responsible for selecting or designing devices and equipment at the hospital. We did not specify who would be responsible for each of these activities because it may vary from hospital to hospital and the same employees may not be responsible for all these activities. For example, a senior leader may influence device and equipment purchases, while a frontline supervisor may be responsible for designing operational processes.
Comment: A commenter stated that for Domain 3 Statement D it is not feasible for safety council leaders to round monthly on all units. This commenter also expressed the concern that while conducting rounds, if leaders specifically focus on safety, these leaders may overlook other potential concerns.
Response: With regard to the high reliability practice in Domain 3 Statement D on whether “[h]ospital leaders participate in monthly rounding for safety on all units, with C-suite executives rounding at least quarterly, with a method in place for follow-up on issues identified,” we understand that having each hospital leader round in every unit monthly would not be feasible for most hospitals. We have retained flexibility for hospitals to identify which leaders round in each unit, as long as leaders round in every unit and C-suite executives round at least quarterly. We also note that attesting to this high reliability practice would not require that these rounds be exclusive to safety and would recommend including other practices or concerns as appropriate.
Comment: A commenter recommended removing practices that are already established as common practices from Domain 3 Statement D to further advance safety, so that hospitals are not rewarded for standard practices.
Response: We reiterate that the Patient Safety Structural measure was developed with input from national experts and through this input we determined that these practices are highly indicative of a culture of safety and are therefore important to include, regardless of their current prevalence. We further note that to positively attest to Domain 3 Statement D hospitals must implement a minimum of four of these practices, which will further encourage hospitals to adopt practices which are less well established.
Comment: Several commenters expressed support for Domain 4 Statement B. These commenters stated that this would incentivize hospitals and PSOs to engage in an activity that supports analysis of harm events. A commenter stated that this attestation would support the legislative purpose of the PSO program to become a national learning system. A commenter stated that use of the Network of Patient Safety Databases (NPSD) is crucial and that the potential increase in data submissions would improve inter-hospital collaboration on patient safety.
Response: We thank the commenters for their support of Domain 4 Statement B. We agree that hospitals voluntarily working with PSOs to track and report patient harm and safety events can be an important and effective activity to help analyze and learn from prior events. We also agree that increased use of the NPSD may further facilitate collaboration across hospitals and support the full potential of the important role that PSOs can play. While we continue to believe that working with a PSO that shares information with the NPSD is an important goal for hospitals to strive towards, we recognize that there are feasibility limitations that would make it difficult for most hospitals to positively attest yes to this domain at this time. Therefore, as discussed below, we are modifying Domain 4 Statement B to read, “Our hospital voluntarily works with a Patient Safety Organization listed by the Agency for Healthcare Research and Quality (AHRQ) [ 359 ] to carry out patient safety activities as described in 42 CFR 3.20 , such as, but not limited to, the collection and analysis of patient safety work product, dissemination of information such as best practices, encouraging a culture of safety, or activities related to the operation of a patient safety evaluation system.”
Comment: A few commenters expressed support for Domain 4 Statement D. Some of these commenters stated that it would be particularly important for patients experiencing harm to know there is a defined communication and resolution program in place. Another commenter noted that communication and resolution programs interrelate to the elements of the other domains, underscoring the importance of establishing a defined communication and resolution program. Other commenters stated a communication and resolution program would benefit healthcare personnel who are currently discouraged from discussing patient harm with patients. A commenter stated that there is a growing body of evidence to support Domain 4 Statement D and, as such, there are support materials available for hospitals seeking to implement communication and resolution programs.
Response: We thank the commenters for their support of Domain 4 Statement D. We agree that well-defined communications and resolution programs are critical for continued engagement and support of patients who experienced harm and would encourage healthcare staff to have transparent conversations with patients. Evidence-based resources, like AHRQ's Communication and Optimal Resolution (CANDOR) toolkit, are helpful tools for hospitals seeking to improve and implement communication and resolution programs.
Comment: A few commenters recommended updates to specific attestation statements within Domain 4. These recommendations were as follows:
- Statement B: Remove the requirement that the PSO reports to NPSD and instead work with AHRQ to ( print page 69480) increase the number of PSOs that participate in the NPSD prior to including this attestation. A commenter stated that their state requires reporting to a state-designated PSO, but not all state-designated PSOs report to the NPSD so some hospitals in their state would be unable to attest positively to this statement. Replace the phrase “voluntary reporting” with “voluntary aggregate reporting” to demonstrate that data reported to the NPSD would be non-identifiable.
- Statement C: Require hospitals to have patient safety metrics available upon request instead of publishing them to reduce operational complexity.
Response: We understand that currently there are only a few PSOs that participate in voluntary reporting to the NPSD and agree with commenters that it would be appropriate for HHS to seek to increase that number. Because of concerns that very few hospitals would be able to positively attest to this statement, and that hospitals may be unable to improve performance by engaging with PSOs that voluntarily report to the NPSD because of insufficient numbers of such PSOs, we are modifying the attestation in Domain 4 Statement B to remove the portion of the attestation related to voluntary reporting to the NPSD. The attestation will thus focus instead on the beneficial activities possible through engagement with a PSO. Through this revision, a hospital that works with a PSO, including a state-designated PSO, from AHRQ's published list of PSOs [ 360 ] to carry out patient safety activities as described in 42 CFR 3.20 will be able to positively attest to this statement.
Commenters' concerns that data submitted by PSOs to the NPSD would be identifiable are misplaced. Before any PSWP is shared with the NPSD, a PSO submits the data to the PSO Privacy Protection Center [ 361 ] to ensure all identifying information has been removed and the data are aggregated before transferring it to the NPSD. There is no cost to PSOs for these privacy protection services. Nonetheless, the revisions to Domain 4 Statement B mean that a hospital need not attest that its PSO voluntarily submits data to the NPSD. Although we are modifying Domain 4 Statement B, we continue to encourage PSOs to voluntarily report serious safety events, near misses, and precursor events to the NPSD to increase the amount of nationally representative safety data for research and analyses (analogous to the National Transportation Safety Board's safety research of US civil aviation accident and injury data [ 362 ] ).
With respect to the commenters' recommendation that we update Statement C to allow hospitals to make patient safety metrics available upon request instead of publishing them, we understand that hospitals seek to balance transparency and operational complexity. By establishing workflows to report and present safety data, hospitals establish an additional pathway to ensure continuing focus on patient safety. Furthermore, by reporting metrics to all clinical and non-clinical staff and making these data public in hospital units, hospitals provide the opportunity for patients to have access to these important data without the potential for fear of reprisal that may be associated with having to request these data from hospital personnel.
Comment: A commenter stated that under current regulations there are three options for patient safety improvement: (1) join a PSO; (2) create a PSO; or (3) use other non-PSO channels for similar functions. The commenter expressed concern that Domain 4 Statement B allows less flexibility without demonstrating that working with a PSO is preferable to other non-PSO channels.
Response: We believe that the commenter is referring to the Medicare CoP for hospitals at 42 CFR 482.21 that requires the development, implementation, and maintenance of an effective, ongoing, hospital-wide, data-driven quality assessment and performance improvement program. The Patient Safety Structural measure does not make any changes to the requirements at 42 CFR 482.21 nor does it affect the voluntary nature of a hospital working with a PSO, creating a PSO, or choosing to not work with a PSO. While Domain 4 Statement B is intended to encourage hospitals to work with a PSO because evidence shows that most hospitals working with PSOs say that this work has helped them understand the causes of patient safety events and prevent future patient safety events, [ 363 ] it does not mandate that all hospitals must work with a PSO, only to attest “yes” or “no” as to whether or not they work with a PSO. We note there is no financial penalty for attesting “no”.
Comment: Some commenters stated that the attestation statement in Domain 4 Statement B on whether a hospital reports serious safety events, near misses, and precursor events to a PSO that participates in voluntary reporting to the NPSD mandates hospitals to report to PSOs in violation of the PSQIA.
Response: To avoid the risk of hospitals unintentionally disclosing PSWP, as discussed previously, we are modifying the Domain 4 Statement B attestation to more broadly describe working with a PSO. The modified statement, “Our hospital voluntarily works with a Patient Safety Organization listed by the Agency for Healthcare Research and Quality (AHRQ) [ 364 ] to carry out patient safety activities as described in 42 CFR 3.20 , such as, but not limited to, the collection and analysis of patient safety work product, dissemination of information such as best practices, encouraging a culture of safety, or activities related to the operation of a patient safety evaluation system,” continues to encourage hospitals to work with PSOs without specifying any reporting of serious safety events, near misses, or precursor events to a PSO. Working with a PSO is a practice consistent with a culture of safety because evidence shows that most hospitals working with PSOs say that this work has helped them understand the causes of patient safety events and prevent future patient safety events. [ 365 ] However, the modified statement does not require hospitals to attest whether they report patient safety event information to PSOs.
Comment: Some of the commenters who were concerned about the attestation in Domain 4 Statement B expressed concern that adopting a measure with this attestation into CMS quality reporting programs would make reporting to a PSO mandatory by penalizing them and impacting hospital payment. Some of these commenters also expressed concern that hospitals would be penalized for not achieving a perfect score.
Response: Domain 4 Statement B encourages hospitals to report to a PSO as this is an indicator of a hospital's ( print page 69481) efforts to improve the quality of its care, as described elsewhere in this rule. Nonetheless, the revisions we have made to the statement provide hospitals with additional flexibility regarding how they work with PSOs and does not require that a hospital report PSWP to a PSO to be completed. No hospital is required by the Hospital IQR and PCHQR Programs to report to a PSO, nor is a hospital's Medicare payment affected by whether a hospital responds to Domain 4 Statement B in the affirmative or the negative. The Hospital IQR Program is a pay-for-reporting program, which means that hospitals that report the required measure data in accordance with the form, manner, and timing policies specified by the Secretary are not subject to a financial penalty under this program. A hospital's performance on the measure, which for the Patient Safety Structural measure is a score from 0 to 5 points, has no impact on a hospital's Medicare reimbursement. Therefore, hospitals that choose not to work with a PSO can attest “no” to Domain 4 and can earn up to 4 points on the measure; there is no financial penalty for attesting “no”. We note that the PCHQR Program is a quality reporting program that does not have a financial penalty associated with it.
As we noted, this measure is intended to determine the level at which hospitals are performing these identified best practices and to identify opportunities for improvement in structural safety practices. Like other quality measures, we would not expect all hospitals to achieve a perfect or maximum score on the measure, especially during the initial years of using the measure in these programs. We proposed this measure for the Hospital IQR and PCHQR Programs because of the identified gaps and variation in the adoption of these safety practices across hospitals ( 89 FR 36285 ). We do want to encourage hospitals to improve their scores on measures over time as a fundamental goal to improve the quality of care. For the Patient Safety Structural measure, improvement on the measure could include hospitals working with PSOs if not already doing so. Furthermore, we note that the TEP included hospital representatives and clinicians who did not express concern that this statement created an implicit mandate for hospitals to report patient safety event information to PSOs. [ 366 ]
Comment: Some commenters expressed concern that the attestation in Domain 4 Statement B would make reporting to a PSO mandatory through impacting hospital reputations and that this would violate the PSQIA.
Response: As modified, a hospital could respond to Domain 4 Statement B in the affirmative without reporting serious safety events, near misses, or precursor events to a PSO so long as it performs other patient safety activities with a PSO. While we understand commenters' concerns about the potential negative impact on a hospital's reputation associated with public display of measure performance information of a score less than five out of five points, or a score that is less than other hospitals, we note the fundamental goal of both the Hospital IQR Program and the PCHQR Program is to drive quality improvement through measurement and transparency in the public display of quality information. Section 1886(b)(3)(B)(viii)(VII) of the Act and section 1866(k)(4) of the Act require the Secretary to report quality measures used in the Hospital IQR Program and the PCHQR Program, respectively, on a CMS website (currently, the Compare tool on Medicare.gov). This public reporting of quality measures gives patients, consumers, and other interested parties access to important information to make informed healthcare decisions.
For the Patient Safety Structural measure, we would publicly report each hospital's score for this measure, which would range from 0 to 5 points, on the Compare tool. We would also publicly report the national average score and the average score in the hospital's state. Therefore, while public reporting of this score could encourage hospitals to work with a PSO if they are not already working with one (as well as encourage improvement upon the other safety practices asked about in the measure), the Patient Safety Structural measure does not require hospitals to achieve any of the listed attestations.
Comment: A commenter stated their belief that including this attestation in the domain titled “Accountability and Transparency” indicates reporting to the PSO is being used for regulatory accountability in violation of the PSQIA which blocks federal oversight agencies from using PSOs as a federal reporting program.
Response: We disagree with the commenters' characterization that the title of Domain 4 or the attestation in Statement B adds a regulatory accountability component beyond the statutory authorities of the Hospital IQR Program and the PCHQR Program or the PSQIA, or that a measure asking hospitals about reporting patient safety data to a PSO creates a federal PSO reporting program. The title of this domain was intended to emphasize the importance of accountability and transparency related to a broad range of a hospital's internal and external safety practices. We have no intention to establish a federal PSO reporting program through this or any other quality measure. Furthermore, as modified, a hospital could respond to Domain 4 Statement B in the affirmative without reporting serious safety events, near misses, or precursor events to a PSO so long as it performs other patient safety activities with a PSO.
Comment: Some commenters stated their concern that Domain 4 Statement B would compel PSOs that do not currently report to the NPSD to begin doing so, and that this amounts to governmental interference with private sector business operations. These commenters further stated that the government does not have the right to mandate government collection of the proprietary data collected by PSOs.
Response: This measure does not affect the voluntary nature of a hospital working with a PSO, nor does it require a hospital to work with a specific PSO. The Patient Safety Structural measure seeks to evaluate and report information about practices that hospitals have in place which demonstrate a structure, culture, and leadership commitment that prioritizes safety and to encourage adoption of these practices. Nonetheless, we have modified Domain 4 Statement B to remove references to whether the hospital reports PSWP to a PSO and whether the PSO voluntarily reports information to the NPSD.
Comment: Some commenters stated that CMS did not provide scientific evidence that this measure would improve the quality of hospital care. Some commenters specifically stated that CMS did not provide evidence that reporting to a PSO which reports to the NPSD is correlated with improved quality of hospital care. Some commenters expressed concern that CMS did not explain why participation in a PSO which reports to NPSD is preferential to participation in any other PSO.
Response: The systems-level approach to patient safety maintains that errors and accidents in medical care are a reflection of system-level failures, rather than failings on the part of ( print page 69482) individuals. [ 367 ] There is a strong alignment among patient safety experts to shift to a more holistic, proactive, systems-based approach to patient safety. [ 368 369 370 371 372 373 ] This Patient Safety Structural measure supports and encourages this shift to a holistic, proactive, systems-based approach to patient safety by highlighting evidence-based practices that hospitals can implement to establish such an approach. We refer readers to the Background section of this discussion for the details of the patient safety guidelines and literature that informed this measure. Furthermore, evidence shows that most hospitals working with PSOs say that this work has helped them understand the causes of patient safety events and prevent future patient safety events. [ 374 ] There are many ways in which PSOs engage their member hospitals to support improvement in patient safety. As an example, PSOs can offer root-cause analyses of specific events, or analysis of data aggregated across hospitals and other non-hospital providers. Through these analyses PSOs can show members how their safety compares to that of their peers. Among hospitals that receive that service, 96 percent of hospitals find the service helpful for improving quality and safety. [ 375 ] By encouraging hospitals to work with PSOs if they do not already, we can expand the use of this valuable patient safety resource and improve quality at hospitals which do not currently use these services. As discussed, we are modifying the Domain 4 Statement B attestation to no longer include specific references to the reporting of PSWP to a PSO or references to PSO reporting to the NPSD. Still, we continue to encourage PSOs to voluntarily submit data to the NPSD. Nonidentifiable PSWP data reported to the NPSD can inform research that further improves the quality of clinical care while ensuring patient and provider confidentiality. [ 376 ] Nationally representative safety data is vital to identifying and improving patient safety practices and driving innovation in quality improvement. It would also allow for the development of best practices which could then be implemented at hospitals to further improve patient safety and quality.
Comment: Some commenters expressed their concern that the measure would penalize hospitals for using PSOs that choose not to report to the NPSD. Some commenters also stated that this measure would limit membership to only certain PSOs which is not consistent with Congressional intent in establishing PSOs through the PSQIA.
Response: As discussed previously, we are modifying the Domain 4 Statement B attestation to no longer include references to PSO reporting to the NPSD, which will more broadly encourage hospitals to work with PSOs if they are not already doing so.
Comment: A commenter stated that AHRQ is only authorized to collect nonidentifiable PSWP for the NPSD.
Response: We agree that AHRQ is only authorized to share nonidentifiable PSWP through the NPSD. Specifically, before any PSWP is shared with the NPSD, a PSO submits the data to the PSO Privacy Protection Center [ 377 ] to ensure all identifying information has been removed and the data are aggregated before transferring it to the NPSD. There is no cost to PSOs for these privacy protection services. We note that while we continue to encourage PSOs to voluntarily report PSWP to the NPSD, we have removed the reference to the NPSD from Domain 4 Statement B.
Comment: A commenter stated that this measure would make hospital reimbursement dependent on the actions of organizations outside the control of the hospital, specifically PSOs.
Response: We disagree that this measure would make hospital reimbursement dependent on the actions of PSOs. The only effect this measure would have on hospital reimbursement from CMS would be if a hospital participating in the Hospital IQR Program chose not to report on the measure at all or did not submit the measure consistent with the form, manner, and timing specified.
Comment: A few commenters expressed concern that mandatory reporting to PSOs for accountability may stifle voluntary exchange of patient safety information which could have a detrimental effect on innovation and learning systems. These commenters also expressed concern that if the measure requires reporting of patient safety data to the federal government, it may discourage hospitals from working with PSOs because of concerns that sensitive hospital information could be used in quality or accountability programs. These commenters stated that discouraging hospitals from working with PSOs would, in turn, limit the ability of PSOs and hospitals to improve healthcare quality and safety.
Response: As discussed previously, we have modified the measure in a way that increases flexibility regarding hospital relationships with PSOs. We do agree with the commenter that an important element of the PSQIA is the voluntary exchange of patient safety information to support innovation and contribute to a learning health system aimed at reducing preventable harms. In light of PSQIA data protections, including the treatment of PSWP as privileged and confidential, we disagree that healthcare providers would stop working with PSOs if more PSOs voluntarily submit patient safety events to the NPSD. Furthermore, increased aggregation and analysis of more comprehensive, nationally representative patient safety data in the NPSD, which is non-identifiable, could potentially help accelerate innovation and the identification of potential solutions to improve safety. The use of quality measures focused on safety, including the Patient Safety Structural measure, complements the important collaboration among hospitals and PSOs and helps set national priorities for safety in hospitals.
Comment: Some commenters expressed concern regarding which entities voluntarily report to the NPSD. ( print page 69483) Some of these commenters stated that many PSOs do not currently report patient safety events to the NPSD, and that many PSOs do not collect patient safety event data. These commenters were concerned that adding this attestation may compel PSOs to change their business models to avoid going out of business if hospitals select PSOs that do report to NPSD. Some commenters stated that some hospitals work with other entities that are not PSOs, such as insurers or Accountable Care Organizations, for analyzing data and developing best practices to ensure patient safety. Because insurers, among others, are not allowed to form PSOs, hospitals that work with insurers for risk management would not be able to have their data reported to the NPSD by their risk management organization. Other commenters stated that organizations other than PSOs, including healthcare providers, can report to the NPSD and adoption of this measure may limit hospital choices for how to report data to the NPSD if they choose to.
Response: We recognize and applaud hospitals that have developed a multi-pronged approach for improving patient safety, including working with PSOs whether or not such PSOs voluntarily report to the NPSD, as well as working with other organizations and entities that are not PSOs, or also large-scale learning networks as described in Domain 3 Statement E. We also understand that while some PSOs may focus on patient safety activities such as development and dissemination of best practices, all PSOs are required to collect and analyze PSWP. [ 378 ] Because evidence shows that most hospitals working with PSOs say that this work has helped them understand the causes of patient safety events and prevent future patient safety events, [ 379 ] we proposed to include in the measure an attestation statement specifically regarding the reporting of patient safety events to PSOs. Due to concerns about the availability of PSOs which report to the NPSD and to avoid the risk of hospitals unintentionally disclosing PSWP, we have modified the attestation in Domain 4 Statement B to allow a more flexible approach to PSO engagement.
Comment: Some commenters also expressed concern that there is no standardized definition for the terms used in this measure (specifically, “serious patient safety event”, “precursor event”, and “near miss”), which the commenters stated would affect how hospitals and PSOs interpret these terms and therefore make the measure less effective.
Response: We intentionally left these terms undefined within this measure to maintain flexibility to allow each hospital to adopt practices that are most effective for its individual circumstances. However, Domain 4 Statement B has been modified and no longer includes these terms. Domain 4 Statement B now instead refers to patient safety activities as described in 42 CFR 3.20 , such as, but not limited to, the collection and analysis of patient safety work product, dissemination of information such as best practices, encouraging a culture of safety, or activities related to the operation of a patient safety evaluation system. We made this change in recognition of the range of activities PSOs may engage in and to improve the clarity of the measure.
Comment: Some commenters expressed concern that the measure would require hospitals and PSOs to use AHRQ's Common Formats to report patient safety data to the NPSD, and that this would disrupt existing data systems, including the risk of no longer being able to conduct long-term trend analyses, as well as add substantial time and expense for PSOs.
Response: The modified Domain 4 Statement B no longer references submission of data to the NPSD, which will provide additional flexibility for hospitals and PSOs to establish reporting formats that are most effective for their relationships. Use of the AHRQ Common Formats is voluntary and available as a potential tool for hospitals (whether they work with a PSO or not). Common Formats for Event Reporting are presently required for submitting data to the PSO Privacy Protection Center for inclusion in the NPSD. [ 380 ] The use of these formats improves the comparability of data across PSOs and ensures privacy protection.
Comment: A commenter expressed concern that the measure's TEP stated that this measure is part of a long-term strategy to generate data and convey priorities to Medicare providers, which the commenters interpreted to mean that CMS would include more requirements for hospital reporting through PSOs in the future.
Response: The statement the commenters are referring to was made by several participants in the TEP that informed development of the Patient Safety Structural measure during an advocacy event for Patients for Patient Safety. [ 381 ] This event was not an official part of the measure development process, and neither the event nor the statement was endorsed by CMS. The statements made at that event do not reflect CMS policy. For information about our strategy with respect to measure development and improving patient safety, we refer readers to the CMS National Quality Strategy [ 382 ] and the specific action steps in the CMS National Quality Strategy: Quality in Motion document. [ 383 ]
Comment: Some commenters were concerned that Domain 4 Statement B would be difficult to achieve because the technology to efficiently transmit serious safety events, precursor events, and near miss events to a PSO is not available so this would be administratively burdensome.
Response: We have modified Domain 4 Statement B in a way that increases flexibility for hospitals to engage with PSOs. As modified, a hospital could respond to Domain 4 Statement B in the affirmative without reporting serious safety events, near misses, or precursor events to a PSO so long as it performs other patient safety activities with a PSO. Furthermore, we understand that there is considerable variation in how hospitals engage in patient safety related learning networks, including whether and how they work with a PSO. We also recognize that not all hospitals have established processes for transmitting serious safety events, precursor events, and near-miss events to their chosen PSO. While this practice is no longer specified in Domain 4 Statement B, and we acknowledge that adopting this practice may entail costs such as investment in a system to support ( print page 69484) patient safety event reporting if a hospital does not already have such a system in place, the value of collecting and analyzing adverse events data to improve patient safety justifies any investment required as evidenced by the improvements in safety associated with the use of a PSO. [ 384 ] We encourage hospitals that are seeking to establish a system for reporting these data to a PSO to use the tools available including the Common Formats [ 385 ] and AHRQ's guidance on choosing a PSO [ 386 ] to reduce the time and costs involved in establishing these systems.
Comment: A commenter supported Domain 5 Statement A and stated that adopting a PFAC is a good way to achieve diversity and equity goals. A few commenters expressed support for Domain 5 Statement C. These commenters stated that patient access to medical records is an important means of engaging patients in their care that further improves quality and safety. A commenter also stated that because of the 21st Century Cures Act there are readily available tools and resources to support patient access to their medical records. A few commenters expressed support for Domain 5 Statement D. These commenters specifically supported the use of patient complaints to prevent similar issues in the future. A few commenters expressed support for Domain 5 Statement E. These commenters stated that patient engagement at the bedside is a longstanding best practice.
Response: We thank the commenters for their support of Domain 5 and specific comments supporting Domain 5's Statements A, C, D and E. We reiterate that Domain 5 activities are aimed to improve equitable and effective engagement of patients, families, and caregivers to promote safer care. We agree a PFAC provides a pathway for patients, families, and caregivers to provide input on critical safety related activities. We also agree that there should be continuous improvement and learning from patient inputs on patient safety events or challenges and that patients should have comprehensive access to their own medical records to further facilitate their engagement in care. We encourage hospitals to implement evidence-based tools and best practices to provide safe, high-quality care to patients.
Comment: A commenter stated the belief that there are situations in which patient, family, and caregiver involvement is inappropriate and recommended modifying the Domain 5 description to reflect that hospitals “should” involve patients, families, and caregivers instead of “must” involve patients, families, and caregivers.
Response: While we acknowledge the possibility that there may be some specific situations in which involving patients, families, or caregivers is infeasible or inappropriate, we note that the Domain 5 description represents the hospital's overall culture. Embedding patients, families, and caregivers through meaningful involvement in safety activities, quality improvement, and oversight is a critical practice to achieve safer, better care. Therefore, the possible existence of some situations in which this is impractical or inappropriate does not preclude establishing a culture that prioritizes such engagement. We note that the specific attestations for Domain 5 do not require patients, families, or caregivers to be involved in every element of the hospital's operations in order to attest “yes,” and that in situations where limitations may be necessary flexibility is provided. Specifically, we note that Domain 5 Statement E requires the hospital to attest to whether it “supports the presence of family and other designated persons (as defined by the patient) as essential members of a safe care team and encourages engagement in activities such as bedside rounding and shift reporting, discharge planning, and visitation 24 hours a day, as feasible. ” (emphasis added).
Comment: A few commenters recommended updates to specific attestation statements within Domain 5. These recommendations were:
- Statement A: Allow different structures for PFACs to address safety (such as at the system level or as a subset of another PFAC).
- Statement C: Include “family caregivers or other parties” because hospitalized patients may not be able to use the portal.
Response: Domain 5 Statement A requires hospitals to attest whether “Our hospital has a Patient and Family Advisory Council that ensures patient, family, caregiver and community input to safety-related activities, including representation at board meetings, consultation on safety-goal setting and metrics, and participation in safety improvement initiatives.” This attestation is not prescriptive of the form or structure for this PFAC and provides flexibility for hospitals and health systems to develop and adopt a PFAC structure that works for their individual circumstances.
We agree with commenters that some hospitalized patients may not be able to use patient portals without assistance. In addition to providing and encouraging access to medical records and clinician notes via patient portals, to affirmatively attest to Domain 5 Statement C hospitals must both provide patients with other options for accessing these data and provide support to help patients interpret the information. Therefore, while expanding access to patient approved family caregivers or other parties is a means of engaging these parties, it is not necessary for a hospital to affirmatively attest to Domain 5 Statement C because patient access to medical information is otherwise addressed by the attestation's reference to additional options and support.
Comment: A commenter expressed concern regarding Domain 5 Statement A that CMS had not provided evidence that a PFAC is associated with improved safety or quality.
Response: Patients, caregivers, and family members offer critical vantage points that can help hospitals identify gaps and priorities for improving patient care. In particular, working with patients and families as advisors at the organizational level can help reduce medical errors and improve safety and quality of health care. 387 388 389 Many leading healthcare quality and safety organizations advocate for the importance of patient and family engagement, and there is support for a safety focused PFAC in the Self-Assessment Tool created and implemented to complement the recommendations in Safer Together: A National Action Plan to Advance Patient Safety. In the Self-Assessment Tool, the National Steering Committee for Patient Safety determined it was appropriate, under the heading of “Patient and Family Engagement,” to award a score of 2 (of 4) for a hospital that has a PFAC and a score of 3 or 4 (of 4) for a hospital at which “The organization has an actively engaged PFAC. Senior leaders ensure the PFAC informs an organization- or system-wide ( print page 69485) strategy and measurement plan for patient engagement.” [ 390 ] For these reasons, the TEP informing development of this measure strongly supported attestation statements addressing PFAC.
Comment: A few commenters expressed concern because of potential complexities related to establishing a PFAC. A commenter expressed concern that maintaining a PFAC could disproportionately burden small hospitals.
Response: We recognize the concern that hospitals with fewer resources may face challenges instituting PFAC. However, engagement of patients, families, and caregivers is essential for improving safety and hospitals such that the long-term benefits would outweigh the costs. The TEP informing development of this measure strongly supported attestation statements addressing PFAC because patients, caregivers, and family members offer critical vantage points that can help hospitals identify gaps and priorities for improving patient care. In particular, working with patients and families as advisors at the organizational level can help reduce medical errors and improve safety and quality of health care. [ 391 392 393 ]
Comment: A few commenters stated that providing access to patient information in Domain 5 Statement C is covered by the requirements of the Promoting Interoperability Program and required by the 21st Century Cures Act.
Response: We believe that the commenters are referring to the Medicare Promoting Interoperability Program reporting requirement for the Provider to Patient Exchange objective, which requires that for at least one unique patient discharged from the eligible hospital or CAH inpatient or emergency department (Place of Service (POS) 21 or 23) the following apply: (1) the patient (or patient-authorized representative) is provided timely access to view online, download, and transmit their health information; and (2) the eligible hospital or CAH ensures the patient's health information is available for the patient (or patient-authorized representative) to access using any application of their choice that is configured to meet the technical specifications of the application programming interface (API) in the eligible hospital's or CAH's certified electronic health record technology (CEHRT) ( 88 FR 59273 ). While a large majority of hospitals are meeting this Medicare Promoting Interoperability Program requirement as part of the meaningful use of certified EHR technology, the attestation in Domain 5 Statement C describes a broader approach to engaging patients as partners in their care. Under this attestation, hospitals would not only provide the patient access but would encourage them to review their data and support them in interpreting the information in a way that is culturally and linguistically appropriate. Hospitals would also help patients submit comments for potential corrections, which could be a vital element of patient safety if the patient's record were incomplete or inaccurate.
After consideration of the public comments we received, we are finalizing adoption of the Patient Safety Structural measure in the Hospital IQR and PCHQR Programs with a modification to the attestation statement in Domain 4 Statement B. The attestation statements we are finalizing are set forth in Table IX.B.1-02.

We refer readers to the FY 2024 IPPS/LTCH PPS final rule for our most recent updates to HCAHPS survey administration requirements and additional background information for the Hospital VBP Program, the Hospital IQR Program, and the PCHQR Program ( 88 FR 59083 through 59089 , 88 FR 59196 through 59201 , and 88 FR 59229 through 59232 , respectively). For more details including information about patient eligibility for the HCAHPS Survey, please refer to the current HCAHPS Quality Assurance Guidelines, which can be found on the official HCAHPS website at: https://hcahpsonline.org/en/quality-assurance/ .
The HCAHPS Survey measure (CBE #0166) asks recently discharged patients questions about aspects of their hospital inpatient experience that they are uniquely suited to respond to. The HCAHPS Survey as a whole is termed as a single “measure” for purposes of the Hospital IQR, PCHQR, and Hospital VBP Programs. We refer to the elements of the HCAHPS Survey that are publicly reported as “sub-measures” and to the questions within each sub-measure as survey “questions,” for the Hospital IQR and PCHQR Programs. Sub-measures are comprised of one, two, or three survey questions. For example, the sub-measure, “Overall Hospital Rating,” consists of one survey question and the sub-measure “Communication with Nurses” consists of three survey questions. In the Hospital VBP Program, the sub-measures of the HCAHPS Survey are referred to as “dimensions.” We refer readers to the HCAHPS On-Line website, www.HCAHPSonline.org , for a map of each question on the HCAHPS Survey and its sub-measures.
The current HCAHPS Survey measure consists of 29 survey questions that are organized into ten sub-measures in the Hospital IQR and PCHQR Programs, including 19 questions that ask “how often” or whether patients experienced a critical aspect of hospital care, rather than whether they were “satisfied” with their care. The current survey measure also includes three screener questions that direct patients to relevant questions, five questions to adjust for the mix of patients across hospitals, and two questions (race and ethnicity) that support Congressionally mandated reports outlined in the Healthcare Research and Quality Act of 1999 ( Pub. L. 106-129 ). [ 399 ] These components of the survey measure are used to construct the ten publicly reported HCAHPS Survey sub-measures in the Hospital IQR and PCHQR Programs. The survey questions are organized into eight dimensions in the Person and Community Engagement Domain for the Hospital VBP Program. We note that the Hospital VBP Program uses eight dimensions while the Hospital IQR and PCHQR Programs use 10 sub-measures because “Cleanliness” and “Quietness” have been combined as a single dimension in the Hospital VBP Program for scoring purposes and the “Recommend Hospital” sub-measure is not included in the Hospital VBP Program. The rationale for combining these elements of the survey is described further in section IX.B.2.g(3) of this final rule and can be found in the Hospital Inpatient VBP Program final rule ( 76 FR 26497 through 26526 ). The current HCAHPS Survey can be found at https://hcahpsonline.org/en/survey-instruments/ .
We proposed to adopt the updated HCAHPS Survey measure for the Hospital IQR, PCHQR, and Hospital VBP Programs in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36298 through 36304 ). We proposed the updated HCAHPS Survey measure would result in a survey with 32 questions that make up a total of 11 sub-measures, with seven of those sub-measures being multi-question sub-measures and the other four sub-measures being single-question sub-measures. Four of the multi-question sub-measures and three of the single-question sub-measures in the updated version of the HCAHPS Survey measure would remain unchanged from those that are in the current version of the HCAHPS Survey measure. For the Hospital VBP Program, the 32-question survey in the updated HCAHPS Survey measure would be organized into nine dimensions. We outline the specific updates later in this section.
We identified the need for the updates to the HCAHPS Survey measure through focus groups and cognitive interviews with patients and caregivers, discussions with technical experts, and literature reviews that were conducted by a CMS contractor who made recommendations to CMS. A literature scan was used to compile and review items from existing surveys, focusing on topics not covered in the current HCAHPS Survey measure. CMS, patients, and providers reviewed the questions identified through the scan. Four patient focus groups were conducted to assign importance to and inform the further development of potential new questions, while also refining existing questions. This replicates the approach taken during the original development of the HCAHPS Survey measure. The focus groups included people with both planned and unplanned hospital stays, a variety of racial and ethnic groups, and both older and younger adults. The focus groups used both an exploratory and confirmatory approach to explore new topics and confirm the topics we had identified through the survey literature. The group discussion explored what it means to have a quality patient experience and what participants thought of their hospital stay—what went well and what went poorly. Group discussions were conducted in English and Spanish.
The findings from the focus group informed the development of the updates to the HCAHPS Survey questions, including the newly developed questions that were tested in cognitive interviews. Cognitive interviews were also conducted in English and in Spanish. Lastly, a CMS contractor convened a technical expert panel that provided feedback on the ( print page 69490) current survey content and the new content areas.
We have determined that adopting the updated version of the HCAHPS Survey measure would amount to a minimal change in burden because the combination of removals and additions of survey questions would result in only an additional 45 seconds to complete the survey. The time required to complete the 32-question survey is estimated to average eight minutes. Additionally, prior to the removal of the “Communication About Pain” questions in the CY 2019 OPPS/ASC final rule ( 83 FR 59140 through 59149 ), the HCAHPS Survey measure previously included 32 questions. We refer readers to sections XII.B.4., XII.B.6., and XII.B.7. of this final rule for more information on our estimated changes to the information collection burden.
The adoption of the updated version of the HCAHPS Survey measure would not result in any changes to the survey administration, the data submission and reporting requirements, or the data collection protocols. The updated version of the HCAHPS Survey measure includes three new sub-measures: the multi-item “Care Coordination” sub-measure, the multi-item “Restfulness of Hospital Environment” sub-measure, and the “Information About Symptoms” single-item sub-measure. The updated HCAHPS Survey measure also removes the existing “Care Transition” sub-measure and modifies the existing “Responsiveness of Hospital Staff” sub-measure. The seven new questions are as follows:
- During this hospital stay, how often were doctors, nurses and other hospital staff informed and up-to-date about your care?
- During this hospital stay, how often did doctors, nurses and other hospital staff work well together to care for you?
- Did doctors, nurses or other hospital staff work with you and your family or caregiver in making plans for your care after you left the hospital?
- During this hospital stay, how often were you able to get the rest you needed?
- During this hospital stay, did doctors, nurses and other hospital staff help you to rest and recover?
- During this hospital stay, when you asked for help right away, how often did you get help as soon as you needed?
- Did doctors, nurses or other hospital staff give your family or caregiver enough information about what symptoms or health problems to watch for after you left the hospital?
As discussed more fully later in this section, these new questions address aspects of hospital care identified by patients and then tested in the 2021 HCAHPS Survey large-scale mode experiment described in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59196 through 59197 ) as important to measuring the quality of hospital care.
The updated HCAHPS Survey measure would no longer include the following four questions:
- During this hospital stay, after you pressed the call button, how often did you get help as soon as you wanted it?
- During this hospital stay, staff took my preferences and those of my family or caregiver into account in deciding what my health care needs would be when I left.
- When I left the hospital, I had a good understanding of the things I was responsible for in managing my health.
- When I left the hospital, I clearly understood the purpose for taking each of my medications.
In the updated HCAHPS Survey measure, the question on the use of the call button is removed in response to hospital input indicating that call buttons have been replaced by other mechanisms (such as a direct phone line). The other questions are removed because they do not follow standard Consumer Assessment of Healthcare Providers & Systems (CAHPS) question wording and were perceived as duplicative of existing and new survey questions by the patients who participated in our content testing.

We refer hospitals and HCAHPS Survey vendors to the official HCAHPS website at https://www.hcahpsonline.org for information regarding the HCAHPS Survey measure, its administration, oversight, and data adjustments. Detailed information on current HCAHPS Survey data collection protocols can be found in the HCAHPS Quality Assurance Guidelines, located at: https://www.hcahpsonline.org/en/quality-assurance/ . The Draft Quality Assurance Guidelines for the proposed updated HCAHPS Survey measure were made available in May 2024 at the official HCAHPS website at: https://www.hcahpsonline.org/en/quality-assurance/ .
The HCAHPS Survey measure produces systematic, standardized, and comparable information about patients' experience of hospital care and promotes person-centered care. We have identified that patient experience measures, including the HCAHPS Survey measure, are foundational metrics, known as the Universal Foundation of quality measures. The Universal Foundation is intended to focus provider attention, reduce burden, identify disparities in care, prioritize development of interoperable, digital quality measures, allow for cross-comparisons across programs, and help identify measurement gaps. [ 400 ] One of the goals of the National Quality Strategy [ 401 ] is to foster engagement and to bring the voices of patients to the forefront. As part of fostering engagement, it is critical to hear the ( print page 69493) voices of individuals by obtaining feedback directly from patients on hospital performance and to incorporate their feedback as part of our comprehensive approach to quality.
We refer readers to section IX.B.1.c. of this final rule for details on the Pre-Rulemaking Measure Review (PRMR) process including the voting procedures the PRMR process uses to reach consensus on measure recommendations. The PRMR Hospital Committee, comprised of the PRMR Hospital Advisory Group and PRMR Hospital Recommendation Group, reviewed the proposed updated version of the HCAHPS Survey measure. The PRMR Hospital Recommendation Group reviewed the proposed updated HCAHPS Survey measure (MUC2023-146, 147, 148, 149) during a meeting on January 18-19, 2024, to vote on a recommendation with regard to use of this measure for the PCHQR, Hospital IQR, and Hospital VBP Programs.
The PRMR Hospital Recommendation Group reached consensus for each of the three programs. For each program, they recommended the updates to the HCAHPS Survey measure with conditions. [ 402 ]
The voting results of the PRMR Hospital Recommendation Group for the proposed updates to the HCAHPS Survey measure within the Hospital IQR Program were: nine members of the group recommended adopting the updates without conditions; eight members recommended adoption with conditions; and two committee members voted not to recommend the updates for adoption. Taken together, 89.5 percent of the votes were between “recommend” and “recommend with conditions.” Thus, the committee reached consensus and recommended the updates to the HCAHPS Survey measure within the Hospital IQR Program with conditions.
The voting results of the PRMR Hospital Recommendation Group for the proposed updates to the HCAHPS Survey measure within the Hospital VBP Program were: ten members of the group recommended adopting the updates without conditions; seven members recommended adoption with conditions; and two committee members voted not to recommend the updates for adoption. Taken together, 89.5 percent of the votes were between “recommend” and “recommend with conditions.” Thus, the committee reached consensus and recommended the updates to the HCAHPS Survey measure within the Hospital VBP Program with conditions.
The voting results of the PRMR Hospital Recommendation Group for the proposed updates to the HCAHPS Survey measure within the PCHQR Program were: eleven members of the group recommended adopting the updates without conditions; six members recommended adoption with conditions; and two committee members voted not to recommend the updates for adoption. Taken together, 89.5 percent of the votes were between “recommend” and “recommend with conditions.” Thus, the committee reached consensus and recommended the updates to the HCAHPS Survey measure within the PCHQR Program with conditions.
The conditions that the committee recommended for all three programs were: CBE endorsement; consideration should be given to not extending the survey length and removal of overlapping items; use of adaptive questions in computerized administration to minimize items; and use of a mechanism to monitor trends in performance data over time.
After taking these conditions into account, we proposed to adopt the updated HCAHPS Survey measure in all three programs. As noted in section IX.B.2.b. of this final rule and in response to the committee's condition that consideration be given to not extending the survey length, the updated HCAHPS Survey measure would result in only an additional 45 seconds to complete the survey. We have estimated that the total time required to complete the 32-question survey is, on average, eight minutes. Additionally, in response to the committee's condition that consideration be given to removing overlapping items, we note that similar or overlapping questions were identified and considered for removal during the development and testing of the updated HCAHPS Survey measure, as described further in section IX.B.2.b. of this final rule. By developing items with patients' and caregivers' input and then empirically testing the new questions, we have ensured that the questions in the updated HCAHPS Survey add unique, non-redundant information about key aspects of patient experience of care. [ 403 ] The committee also raised the condition that the survey use adaptive questions in computerized administration to minimize items. However, adaptive questions in computerized administration would be infeasible in the mail mode of the HCAHPS Survey measure. Since all modes of survey administration that are available for the updated HCAHPS Survey (Mail Only, Phone Only, Mail-Phone, Web-Mail, Web-Phone, and Web-Mail-Phone) must be parallel, adaptive questions in computerized modes would not be appropriate for this measure at this time. We will take this feedback into consideration for any future potential changes to survey administration. In response to the committee's condition that a mechanism to monitor trends in performance data over time be used, we note that as part of administering each of these quality programs, we regularly monitor and evaluate hospitals' performance data trends. We will continually monitor these trends in performance with the updated HCAHPS Survey measure. We address the committee's condition of CBE endorsement in the following section.
We refer readers to section IX.B.1.c. of this final rule for details on the endorsement and maintenance (E&M) process including the measure evaluation procedures the CBE's E&M Committees use to evaluate measures and whether they meet endorsement criteria. The HCAHPS Survey measure was first endorsed in 2005 by the former CBE, the National Quality Forum. The former CBE renewed its endorsement of the current HCAHPS Survey measure in 2009, 2015, and 2019. The current HCAHPS Survey measure was most recently submitted to the CBE for maintenance endorsement review in the Spring 2019 cycle (CBE #0166) and was endorsed on October 25, 2019. [ 404 ] We note that the HCAHPS Survey measure remains an endorsed measure, and we intend to submit the updated HCAHPS Survey measure to the current CBE for endorsement in Fall 2025. Section 1886(b)(3)(B)(viii)(IX)(bb) of the Act states that in the case of a specified area or medical topic determined appropriate ( print page 69494) by the Secretary for which a feasible and practical measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We have determined that the updates to the HCAHPS Survey measure are appropriately specified. The HCAHPS Survey measure remains endorsed, and the updated survey only modifies some of the questions and sub-measures within the survey. The HCAHPS Survey measure is designed to produce standardized information about patients' perspectives of care that allow objective and meaningful comparisons of hospitals on topics that are important to consumers, and these updates would improve the feedback we receive directly from patients on hospital performance. Therefore, we proposed these updates to the measure before the updates received CBE endorsement.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36298 through 36300 ), we proposed to update the current HCAHPS Survey measure in the Hospital IQR and PCHQR Programs by adding three new sub-measures:
- “Care Coordination” sub-measure
- “Restfulness of Hospital Environment” sub-measure
- “Information About Symptoms” sub-measure
The updates also remove the existing “Care Transition” sub-measure and modify the existing “Responsiveness of Hospital Staff” sub-measure. The new “Care Coordination” sub-measure encompasses and broadens the current “Care Transition” sub-measure and the new questions in the “Care Coordination” sub-measure are more congruent with the other survey questions. The updated measure replaces one of the two survey questions in the current “Responsiveness of Hospital Staff” sub-measure with a new survey question that strengthens this sub-measure. The updates to the HCAHPS Survey measure are detailed in section IX.B.2.b. of this final rule and we refer readers to the HCAHPS website at https://www.hcahpsonline.org for further details.
In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed that the updated HCAHPS Survey measure would be implemented in the Hospital IQR and PCHQR Programs beginning with patients discharged on January 1, 2025 ( 89 FR 36298 ). Reporting of responses from the updated HCAHPS Survey measure for patients discharged between January 1, 2025, and December 31, 2025, would be used for the CY 2025 reporting period/FY 2027 payment determination for the Hospital IQR Program and for the CY 2025 reporting period/FY 2027 program year for the PCHQR Program. HCAHPS Survey sub-measures are publicly reported on a CMS website quarterly on a rolling basis, with the oldest quarter of data rolled off, and the most recent quarter rolled on with each refresh. As such, there would be a period during which some quarters of reporting data come from the current version of the HCAHPS Survey measure, and others come from the updated HCAHPS Survey measure. Through this time period, publicly reported HCAHPS Survey data for the Hospital IQR and PCHQR Programs would consist only of data from the eight unchanged sub-measures in the current HCAHPS Survey measure. When four quarters of the updated HCAHPS Survey data have been submitted, public reporting would reflect all of the modifications in the updated HCAHPS Survey measure. The public reporting timeline of the updates to the HCAHPS Survey measure for the Hospital IQR and PCHQR Programs can be found in Table IX.B.2-02.

The “Care Coordination” sub-measure is a newly developed multi-question sub-measure and is composed of three new survey questions that ask patients how often hospital staff were informed and up-to-date about the patient's care, how often hospital staff worked well together to care for the patient, and whether hospital staff worked with the patient and family or caregiver in making plans for the patient's care post-hospitalization. The new questions address aspects of hospital care identified by patients participating in focus groups as important to measuring the quality of hospital care. Cognitive testing demonstrated the new questions were accurately and consistently interpreted. The “Care Coordination” sub-measure was shown to have good measurement properties (hospital-level reliability is 0.792 and Cronbach's alpha is 0.765) and construct validity in the 2021 mode experiment. [ 405 ] This sub-measure would fill a gap of furthering coordination efforts within the hospital setting and support our goals of including measures related to seamless care coordination and person-centered care. Across multiple focus groups, patients indicated that how well doctors, nurses, and other staff work together or as a team in caring for a patient was the most important information to have to understand what their care would be like in one hospital versus another.
The Restfulness of Hospital Environment—Hospital Patient sub-measure would fill a gap related to providing a restful and healing environment within the hospital setting and support our goal of including measures related to person-centered care. The “Restfulness” sub-measure is a newly developed multi-question sub-measure comprised of three survey questions: two new questions that ask how often patients were able to get the rest they needed, and whether hospital staff helped the patient to rest and recover, and one current survey question that asks how often the area around the patient's room was quiet at night (“Quietness”). Cognitive testing demonstrated the new questions were accurately and consistently interpreted. The 2021 mode experiment established that the “Restfulness” sub-measure has good measurement properties (hospital-level reliability is 0.870 and Cronbach's alpha is 0.735) and construct validity. [ 406 ] The existing “Quietness” sub-measure ( print page 69496) is currently a stand-alone question in the HCAHPS Survey measure. The updates to the HCAHPS Survey measure would move the stand-alone “Quietness” sub-measure into the new Restfulness of Hospital Environment sub-measure. In the updated version of the HCAHPS Survey measure, the “Quietness” question itself would not change and would continue to be publicly reported.
The “Information About Symptoms” sub-measure is a newly developed single-question sub-measure that would fill a gap of providing instructions and information for family and caregivers to take care of patients after discharge and supports our goal of including measures related to person-centered care. The new question captures an aspect of hospital care identified by patients participating in focus groups as important, and cognitive testing demonstrated the question was accurately and consistently interpreted. The sub-measure is a stand-alone question that asks the patient whether doctors, nurses, or other hospital staff gave the patient's family or caregiver enough information about symptoms or health problems to watch out for after the patient left the hospital. The sub-measure has good hospital level-reliability (0.729) at the expected average number of completed surveys per hospital. [ 407 ]
The revisions to the “Responsiveness of Hospital Staff” sub-measure would entail adding one new survey question to this sub-measure and removing one current survey question from this sub-measure. The current survey question that would be removed from the “Responsiveness of Hospital Staff” sub-measure is the “Call Button” question. Input from hospitals indicated that call buttons have largely been replaced by other mechanisms (such as a direct phone line), and qualitative testing demonstrated that the new question captures all modes of requesting help. The 2021 mode experiment established that the modified “Responsiveness of Hospital Staff” sub-measure has good measurement properties (hospital-level reliability is 0.786 and Cronbach's alpha is 0.749) and construct validity. [ 408 ] Having patients report their experience of the responsiveness of hospital staff highlights an important aspect of hospital care from the patient's perspective about getting help for one's needs during a hospital stay, which is a component of person-centered care. These modifications to the “Responsiveness of Hospital Staff” sub-measure would fill a gap related to the care by nursing and other staff within the hospital setting and support our goals of including measures assessing person-centered care and the quality of hospital staff. The revised “Responsiveness of Hospital Staff” sub-measure would be comprised of two survey questions: one current survey question that asks how often patients received help in getting to the bathroom or in using a bedpan as soon as they wanted, and one new survey question that asks how often patients got help as soon as they needed it when they asked for help right away.
In the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53513 through 53516 ), we added the three-question “Care Transition” sub-measure (CTM-3) to the HCAHPS Survey measure in the Hospital IQR Program. We finalized the addition of the HCAHPS Survey measure, including the CTM-3 sub-measure, for the PCHQR Program in the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50844 through 50845 ). The updates to the HCAHPS Survey measure would remove this three-question sub-measure from the HCAHPS Survey measure and replace it with a new “Care Coordination” sub-measure, which would encompass and broaden the current “Care Transition” sub-measure and is more congruent with the other questions in the HCAHPS Survey measure in terms of question form and response options. For these reasons, the updated version of the HCAHPS Survey measure removes the “Care Transition” sub-measure.
The “About You” questions are used either for patient-mix adjustment or for Congressionally-mandated reports.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36300 ), we proposed that the changes to the “About You” section of the updated HCAHPS Survey measure would be:
- replacing the existing “Emergency Room Admission” question with a new, “Hospital Stay Planned in Advance” question;
- reducing the number of response options for the existing “Language Spoken at Home” question;
- alphabetizing the response options for the existing ethnicity question; and
- alphabetizing the response options for the existing race question.
We note that to achieve the goal of fair comparisons across all hospitals that participate in HCAHPS Survey measure, it is necessary to adjust for factors that are not directly related to hospital performance but do affect how patients answer HCAHPS Survey questions. To ensure that differences in HCAHPS Survey measure results reflect differences in hospital quality only, HCAHPS Survey measure results are adjusted for patient-mix and mode of survey administration. Only the adjusted results are publicly reported and considered the official results. Information about the HCAHPS Survey patient-mix adjustment can be found at: https://hcahpsonline.org/en/mode--patient-mix-adj . We do not collect or adjust for patients' socioeconomic status, however, the HCAHPS Survey patient-mix adjustment does include patients' highest level of education, which can be related to socioeconomic status. Several questions on the HCAHPS Survey, as well as information drawn from hospital administrative data, are used for the patient-mix adjustment. The questions in the “About You” section of the survey that are used in patient-mix adjustment are:
- In general, how would you rate your overall health?
- In general, how would you rate your overall mental or emotional health?
- What is the highest grade or level of school that you have completed?
- What language do you mainly speak at home?
Administrative data provided by hospitals are also used in patient-mix adjustment, including patient's age, sex, and service line. Lag time, which is the number of days between a patient's discharge from the hospital and the return of the mail survey or the final disposition of the telephone or interactive voice recognition (IVR) ( print page 69497) survey, is also used in patient-mix adjustment. [ 409 ]
Neither patient race nor ethnicity is used to adjust HCAHPS Survey results; these questions are included on the survey to support Congressionally-mandated reports. The adjustment model also addresses the effects of non-response bias. More information about the patient-mix adjustment coefficients for publicly reported HCAHPS Survey measure results can be found under “Mode and Patient-Mix Adjustment” at: https://www.hcahpsonline.org .
The current “About You” survey question that asks whether the patient was admitted to the hospital through the emergency room would be replaced with a new question that asks whether this hospital stay was planned in advance. This “Hospital Stay Planned in Advance” question is being adopted for use as a patient-mix adjuster to distinguish between planned and unplanned stays. Cognitive testing indicated that “Hospital Stay Planned in Advance” is better understood as intended than the current admission through the emergency room question. Unplanned stays are not within the hospital's control but can result in worse patient experiences than hospital stays that had been planned. Accounting for these differences in this preadmission characteristic allows for fairer comparisons of hospital performance.
To make survey administration more efficient and reduce respondent burden, especially in the telephone mode of survey administration, we proposed that the response options for the “Language Spoken at Home” question would be changed to: “English,” “Spanish,” “Chinese,” or “Another language” ( 89 FR 36300 ). English, Spanish, and Chinese account for 98.2 percent of all HCAHPS Survey measure responses. The response options for the two race/ethnicity questions would be alphabetized to correspond to current best survey practices.
These modifications would not be included in public reporting of the HCAHPS Survey measure and would not affect scoring under the Hospital VBP Program, but the “Hospital Stay Planned in Advance” question would be employed in the patient-mix adjustment of survey responses.
In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to implement these changes along with the proposed updated version of the HCAHPS Survey measure for the Hospital IQR, PCHQR, and Hospital VBP Programs described in earlier sections ( 89 FR 36300 ).
We received public comment on the overall updates to the HCAHPS Survey measure.
Comment: Many commenters broadly supported adopting the updates to the HCAHPS Survey measure across the Hospital IQR, Hospital VBP, and PCHQR Programs as proposed because they stated that the updates modernize the survey, promote person-centered care, reflect new technology and the best practices for patient care, better align with CMS's quality strategies, and make the questions more relevant to patients and families while also being useful to hospitals. Several commenters commended CMS for the approach to align the updates across three programs, for considering stakeholder feedback in identifying opportunities for improvement, and for continuing to improve capturing patient experiences and the voice of the patient. Several commenters supported the removal of questions they deemed redundant and efforts to reduce survey length by limiting the number of supplemental items to manage survey burden. A few commenters supported the inclusion of family caregivers in the updated survey questions, and a few commenters specifically supported the staggered implementation of the updates in public reporting and in the Hospital VBP Program. A commenter supported CMS' efforts to refine measuring patient experience, while another commenter stated that the additional information from patients and caregivers would help hospital administrators ensure they are delivering high quality care.
Response: We thank the commenters for their support. We agree with commenters that the updates align with our national quality strategies. As noted in the FY 2025 IPPS/LTCH PPS proposed rule, one of our key goals is to foster engagement and bring the voices of patients to the forefront ( 89 FR 36296 ). The updates to the HCAHPS Survey measure enable us to obtain feedback directly from patients on hospital performance and to incorporate their feedback as part of our comprehensive approach to quality.
Comment: Several commenters requested clarifications on the updates to the HCAHPS Survey measure, with a few commenters requesting clarification on the overall testing of the updates, questioning whether the new components would be representative of a hospital making improvements. A few commenters supported the updates to the HCAHPS Survey measure but requested additional information on how the items were tested to help understand whether they measure hospitals accurately. A commenter recommended shortening the survey, removing redundant questions, and authorizing real-time survey alternatives because they gather broader patient feedback.
Response: We thank the commenters for their support and refer them to the PRMR report that outlines the testing we conducted, [ 410 ] which included a literature review, technical expert panels, focus groups and cognitive interviews with patients and caregivers, and a mode experiment in 2021 among 46 hospitals. We refer readers to the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36293 through 36297 ) which outlines the content testing, hospital input, and patient focus groups that informed our updates to the survey. As described in the proposed rule, the patient focus groups identified the aspects of hospital care addressed in the new questions as important to measuring the quality of hospital care, and the updated sub-measures were tested for hospital-level reliability and validity at the expected average number of completed surveys per hospital. We also refer readers to the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36299 ) where we outline the reliability and validity testing of each sub-measure. Along with empirically testing these updates, the updates to this survey were developed with patients' and caregivers' input, and we have ensured that the questions in the updated HCAHPS Survey measure add unique, non-redundant information about key aspects of patient experience of care. We thank the commenter for the recommendation to continue reducing the question set, and we will continue to evaluate and test ways to improve the survey in future program years. We do not anticipate authorizing real-time survey alternatives at this time, but we will re-evaluate alternatives in future program years.
Comment: A few commenters expressed support for the new web-first survey modalities and the extended 49-day window for survey responses because they stated they have been shown to increase survey response rates, especially among historically underrepresented populations.
Response: We thank the commenters for their support. We wish to note that ( print page 69498) the web-first survey modalities and 49-day window for survey responses were finalized in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59197 through 59199 ).
Comment: A few commenters generally did not support the updates to the HCAHPS Survey measure because they stated the addition of more questions would create resistance among patients who are already discouraged by the current number of questions.
Response: We thank the commenters for their feedback. As discussed in the FY 2025 IPPS/LTCH PPS proposed rule, four patient focus groups were conducted to inform the development of the updates to the HCAHPS Survey ( 89 FR 36293 ). These patients identified the aspects of hospital care that were important to them when measuring the quality of care they received. As a result, we do not agree that the updates to the survey would create resistance among patients. Additionally, we did not receive negative feedback about the length of the survey that was tested in the 2021 HCAHPS mode experiment, which was 43 items compared to the updated HCAHPS Survey measure, which includes 32 items. [ 411 ] The survey did not require multiple calls to complete, and respondents in the 2021 mode experiment did not have complaints about the time on the phone or the length of the interview. Moreover, interviewers did not report any challenges keeping respondents engaged through the end of the survey. Additionally, the updates to the HCAHPS Survey measure would create minimal change in burden because they result in only an additional 45 seconds to complete the survey, even without considering the reduction in total survey length that would result from the new limit on supplemental items. The 12-item limit on supplemental items, which was finalized in the FY 2024 IPPS/LTCH PPS final rule, effectively reduces the average length of the HCAHPS Survey measure ( 88 FR 59199 ). Currently, the median number of supplemental items added to the HCAHPS Survey is 14, while 25 percent of hospitals add 30 or more extra items. The 12-item limit of supplemental items will help to reduce the length of the updated HCAHPS Survey measure.
Comment: A few commenters requested clarifications regarding the impact of the updates to the HCAHPS Survey measure on the Star Ratings, including how the HCAHPS Summary Star Rating would be calculated both during the transition period and after the new survey is publicly reported, how the abbreviated set of HCAHPS Survey measure scores released during the transition would be used to calculate the Overall Hospital Quality Star Rating, and how CMS would incorporate the new HCAHPS Survey measure scores into the Overall Hospital Quality Star Rating after the new survey is publicly reported. They also requested clarification on how the modified public reporting schedule might impact the inclusion of HCAHPS in Hospital Overall Stars.
Response: In response to the requests for clarifications regarding the Star Ratings, the HCAHPS Summary Star Rating would continue to be the average of the publicly reported HCAHPS Survey measure as described in the Technical Notes for HCAHPS Star Ratings. [ 412 ] During the transition period when the number of HCAHPS Survey sub-measures are reduced from 10 to 8 sub-measures, the HCAHPS Summary Star Rating would be constructed by averaging the Star Ratings from “Communication with Nurses,” “Communication with Doctors,” “Communication about Medicines,” “Discharge Information,” the average of the Star Ratings assigned to “Cleanliness of Hospital Environment” and “Quietness of Hospital Environment,” and the average of the Star Ratings assigned to “Hospital Rating” and “Recommend the Hospital.” We will update the HCAHPS Star Rating Technical Notes on the official HCAHPS On-Line website ( https://hcahpsonline.org/en/hcahps-star-ratings/ ) prior to the January 2026 public reporting on the Compare tool to describe how the 8 sub-measures are used to calculate the HCAHPS Summary Star Rating.
The HCAHPS Star Rating Technical Notes will be updated again prior to the October 2026 public reporting on the Compare tool to describe the calculation of the HCAHPS Star Ratings when the number of publicly reported HCAHPS Survey sub-measures increases from 8 to 11. For the October 2026 public reporting and forward, the HCAHPS Summary Star Rating will be constructed by averaging the HCAHPS Star Ratings from “Communication with Nurses,” “Communication with Doctors,” “Restfulness of Hospital Environment,” “Care Coordination,” “Responsiveness of Hospital Staff,” “Communication about Medicines,” “Discharge Information,” the average of the Star Ratings assigned to “Cleanliness of Hospital Environment” and “Information About Symptoms,” and the average of the Star Ratings assigned to “Hospital Rating” and “Recommend the Hospital.” The weight of the Patient Experience measure group in the Overall Hospital Quality Star Rating, which includes the HCAHPS Survey measure, would not change without notice-and-comment rulemaking.
Comment: Many commenters expressed concerns over the updates to the HCAHPS Survey measure. Several commenters stated the new length of the survey can have negative effects on patient completion such as increased burden, survey fatigue, and reduced response rates at a time when response rates are already trending downward. A few commenters requested clarification on whether the updated 32 question survey had been tested with patients to determine if the added length had any negative effects on the patient's likelihood of completing the survey. A commenter recommended limiting the Restfulness sub-measure to one question because they stated that including repetitive questions may decrease survey completion. A commenter expressed concern that the additional 45 seconds equates to a 10 percent extension. A commenter stated that the additions to the survey were outpacing the removal of items and recommended working with AHRQ to research longer term solutions to reduce length and improve response rates.
Response: We have developed the new items with patients' and caregivers' input and empirically tested the new questions and sub-measures. We refer readers to the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36293 through 36297 ) and this final rule which outlines the content testing, hospital input, and patient focus groups that informed our updates to the survey. The new items address those aspects of hospital care that patient focus groups identified as important to measuring the quality of hospital care, and the new sub-measures were tested for hospital-level reliability and validity at the expected average number of completed surveys per hospital. 413 414 We refer ( print page 69499) readers to the PRMR report for additional information on the testing we conducted. [ 415 ] Therefore, we have ensured that the questions proposed in the updated HCAHPS Survey measure add unique, non-redundant information about key aspects of patient experience of care. Additionally, if we limited the Restfulness sub-measure to one question, the reliability of this sub-measure would be reduced. We have determined that the modified version of the HCAHPS Survey measure creates minimal change in burden because the updates result in only an additional 45 seconds to complete the survey. Limiting the supplemental items to no more than 12 will also effectively reduce the length of the survey. We also remind commenters that the HCAHPS Survey measure has previously included 32 questions—the same number of questions in the updated version. The previous 32 question version of the HCAHPS Survey measure did not negatively affect response rates. Prior CAHPS studies suggest that the effect of a three-item change in survey length on the response rate is less than one percentage point. [ 416 417 ]
Comment: A commenter questioned whether the data from the new sub-measures could be used to improve performance because the measures are not based upon clinical practice guidelines.
Response: The HCAHPS Survey measure is not intended to measure clinical outcomes. Rather, it is intended to measure patients' experiences of hospital care, a key metric in assisting healthcare organizations to move toward patient-centered care.
Comment: A few commenters expressed concerns about the validity of the survey, believing that the survey may not measure quality of staff but rather capture wider system-level issues such as staffing shortages, and that there was limited evidence to demonstrate a relationship between patient satisfaction, care quality, and clinical outcomes.
Response: With regard to concerns that the HCAHPS Survey measure results may be a reflection of system-wide issues, we agree that these issues, such as staffing shortages, can adversely affect patient experience; when that happens, HCAHPS accurately captures the impact of the system-wide issue on patient experience. [ 418 ] Patient experience of care surveys, including the HCAHPS Survey measure, capture an independently important dimension of quality of care and are associated with better care and outcomes in other areas, such as lower hospital readmissions. [ 419 420 ]
Comment: A commenter expressed concern that the changes to the survey may disrupt years of data and comparisons that have been used to judge improvements in patient satisfaction.
Response: With regard to possible disruptions to survey continuity and hospitals' ability to compare results from the updated HCAHPS Survey measure with the current version, 8 of 10 current HCAHPS sub-measures would be unchanged on the updated HCAHPS Survey measure (see Table IX.B.2-02); there would be no discontinuity in historical comparisons for these sub-measures. The “Quietness” question would be unchanged in the updated HCAHPS Survey measure but would be made part of the new “Restfulness of Hospital Environment” sub-measure. However, the “Quietness” question would be reported as a single question in both the preview reports hospitals receive before each public reporting and in the Provider Data Catalog, thus permitting continuous comparisons with historical data. Only one sub-measure, “Care Transition,” and one substantive question, “Call Button,” would be removed from the survey.
Comment: A commenter expressed that patient care needs were in direct conflict with the HCAHPS Survey's priorities, such as when a patient may require more intensive monitoring and regular interventions, and therefore interruptions, overnight. The commenter acknowledged that creating a restful environment is an important dimension of helping patients get better, but that restfulness cannot be prioritized to the detriment of patient needs and outcomes and recommended that CMS engage interested parties to determine how to better balance competing priorities.
Response: In response to the request to engage interested parties, we note that we engaged multiple interested parties. Patients were engaged during initial work to identify concepts they deemed important in assessing quality of care during a hospital stay as well as concepts they deemed less important. Multiple audiences were included as technical experts in the technical expert panel convened by a CMS contractor, which included discussions of new content and priorities and trade-offs between new and existing content. In addition, multiple rounds of qualitative testing and discussions were conducted with patients and caregivers. We do not agree that the survey's priorities are in conflict with patient care because patient focus groups identified these aspects of care as important to measuring the quality of hospital care, and survey development and recent refinement took patient care needs, patient information needs, and input from interested parties into account when developing and refining the HCAHPS Survey measure. [ 421 422 ]
Comment: Many commenters provided additional recommendations for the HCAHPS Survey measure including that CMS go further in expanding use of the survey to address challenges with under-reporting patient safety events and speeding up the process for integrating and reporting on the HCAHPS Survey measure changes because they are discouraged that the implementation for these updates is extended over several years.
Response: We appreciate the commenters' recommendations and will consider additional ways to expand use of the HCAHPS Survey measure in future program years. While the HCAHPS Survey measure does not ask patients directly about patient safety events, the survey information could complement other patient safety data ( print page 69500) collection. In regard to speeding up the process to integrate and report on the HCAHPS Survey measure, we note that changing the HCAHPS Survey entails thorough development and testing, followed by thorough vetting of the proposed changes through a number of internal, external, and rulemaking processes. Then, to publicly report HCAHPS sub-measures, four quarters of survey data must be collected. Lastly, the adoption of new or revised HCAHPS sub-measures into the Hospital VBP Program entails meeting the statutory requirements as outlined in section 1886(o)(2)(C)(i) of the Act which precludes us from adopting a measure into the Hospital VBP Program until we have specified the updates under the Hospital IQR Program and included them on Care Compare for at least one year prior to the beginning of the performance period for such fiscal year. Therefore, we cannot speed the timeline up for implementation.
Comment: Several commenters recommended additional testing and analyses to ensure that the updates reflect a streamlined approach to the survey before they are adopted in the CMS programs. Their recommendations included analyzing the reading levels of all the proposed new questions and modifying the wording as necessary, having a third party fully vet and endorse the updates before implementing them, conducting further validity and reliability testing, ensuring the questions are worded in a way that allows patients to assess an aspect of quality, and providing more information on the survey design process and the criteria used to determine when questions are considered to overlap.
Response: Regarding the recommendation for additional testing and analyses when adopting the updates, we have conducted substantial testing through the 2021 mode experiment, patient focus groups, literature reviews, technical expert panels, and reliability and validity testing, as described in both the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36293 through 36299 ) and the PRMR report. [ 423 ] Changing the HCAHPS Survey entails thorough development and testing, followed by thorough vetting of the proposed changes through a number of internal, external, and rulemaking processes. As such, we do not agree that additional testing is needed before adopting these updates.
Comment: A few commenters offered recommendations for additional survey questions including a medication reconciliation question because medication errors are estimated to be the most common error made in hospitals, a patient consent question that would eliminate the need for organizations to add supplemental questions, and a question similar to one in the Medicare Advantage CAHPS Survey that addresses patients' perceptions of unfair or insensitive treatment during their hospital stay. A few commenters made recommendations around the languages offered for the HCAHPS Survey measure including expanding the approved HCAHPS languages, offering the survey in all approved languages for all survey modes similar to the Outpatient and Ambulatory Surgery CAHPS, reconsidering limiting the HCAHPS Survey measure to only support English and Spanish languages, and requiring hospitals to offer the survey in the language preferred by the patient or family member.
Response: We thank commenters for their suggestions of additional questions to add to the HCAHPS Survey measure and we will consider testing and potentially adding these questions in future program years. We also thank the commenters for their recommendations regarding offering the HCAHPS Survey measure in additional languages, and we will take these recommendations into consideration for future program years.
We note that the HCAHPS Survey measure is available in 8 official non-English translations, and that the official Spanish translation must be administered to all Spanish-preferring patients beginning in January 2025. [ 424 ] We welcome suggestions for new translations in future program years.
Comment: A few commenters recommended additional changes to the survey including changing the “Likelihood to Recommend” responses to “Net Promoter Score” responses and adding “Caregiver Status” to the list of standardized patient assessment data elements for reporting.
Response: We thank the commenters for their recommendations. In response to changing the “Likelihood to Recommend” (“Recommend Hospital”) responses to “Net Promoter Score” responses, the response options for the “Recommend Hospital,” which have been cognitively tested, empirically validated, and used in the HCAHPS Survey measure since its inception as well as in other CAHPS surveys, are appropriate for achieving the goals of the HCAHPS Survey measure. We understand that the Net Promoter Score is a popular surveying method to capture customer loyalty, however we disagree with using the Net Promoter Score because there is a lack of research to support the use of the Net Promoter Score as a primary metric of patient experience at this time, including information about validity and reliability in the hospital setting. [ 425 ]
Comment: A few commenters stated that some questions are redundant or subjective, with a commenter believing that questions 20, “Information about Symptoms,” and 23, “Discharge Information in Writing” both provide information about symptoms post-discharge. A few commenters also recommended combining or clarifying questions 20, “Information about Symptoms,” and 23, “Discharge Information in Writing”, and 19, “Care Coordination Post-Hospital” and 22, “Discharge Information Help” to avoid redundancies, replacing “doctors, nurses, and other hospital staff” to “healthcare team” throughout the survey because they stated that “healthcare team” encompasses all individuals who may care for a patient, and reviewing the “Discharge Information” questions, the “Information About Symptoms” question, and the new “Care Coordination” questions to determine how to incorporate the concept of language preferences. The commenters also requested clarification on whether the information being provided in response to the “Discharge Information” questions, the “Information About Symptoms” question, and the new “Care Coordination” questions is actually understood by the patient and family or caregiver and whether inclusion of both the “Information About Symptoms” and “Discharge Information” questions will provide enough differentiated information to warrant adding to the length of the survey. A few commenters requested clarification on what “other hospital staff” refers to in the “Care Coordination” sub-measure because not every individual in a hospital environment would have reason to be included in a patient's plan of care. ( print page 69501)
Response: We note that we do not collect standardized patient assessment data or protected health information from patients or from hospitals and the HCAHPS Survey measure does not include patient assessment data, and therefore, we cannot add “Caregiver Status” to the list of standardized patient assessment data elements. We may consider developing and testing items about caregiver status for future use; however we also note that we have added questions about communicating with family and caregivers in both the new “Care Coordination” sub-measure and in the new “Information about Symptoms” single-item sub-measure. We also remind the commenter that a patient's proxy is permitted to respond to the HCAHPS Survey beginning with January 2025 discharges, as finalized in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59198 ).
Survey questions 20 and 23 differ in significant ways. Question 20, “Information About Symptoms,” asks about the engagement of family members or caregivers, specifically whether the patient's family or caregiver received enough information about what symptoms or health problems to help watch for after the patient leaves the hospital. Patients identified information communicated to a patient's family members or caregivers as an aspect of care that is critical to measuring quality. In contrast, Question 23, “Discharge Information in Writing,” is specific to the patient's experience. It asks whether the patient received written information about symptoms or health problems to look out for after leaving the hospital, which patients also identified as an aspect of care that is important to measuring quality and is only asked of patients who go directly home after leaving the hospital. These are different topics and are measured via separate items to ensure that the data collected are actionable. Questions 20 and 23 are also not empirically redundant. The empirical testing of the new questions for the updated HCAHPS Survey measure, both from their content and from statistical evidence, demonstrated that these questions address different aspects of patient care and that each question independently predicts the overall rating of the hospital. We refer the commenter to the PRMR report for additional information on the testing we conducted. [ 426 ] Given these important differences, it is appropriate to maintain questions 20 and 23 as separate questions. Similarly, we have determined that the “Information About Symptoms” and “Discharge Information” questions use terms that are well understood by the patient and family or caregiver based on focus groups and cognitive interviews and therefore need not be clarified.
Additionally, we do not agree with commenters' recommendations to combine questions 19 and 22 in the HCAHPS Survey measure because we have determined via qualitative and quantitative testing that these questions address different key aspects of care as identified by patients and caregivers. Question 19, the “Care Coordination Post-Hospital” question, collects information on whether the patient's family or caregiver was involved in discussion of the patient's post-discharge care needs, while question 22, the “Discharge Information Help” question, is specific to the patient's experience and asks patients who were discharged to their own home or someone else's home whether doctors, nurses or other hospital staff talked with them about whether they would have the needed level of help or support after leaving the hospital. As explained above, questions 20 and 23 similarly address different aspects of patient care, focusing on either the experience of the patient's family or caregiver (question 20) or the experience of the patient (question 23) in receiving information about symptoms or health problems to watch for after the patient leaves the hospital.
Comment: A few commenters expressed concerns with the verbiage in the new “Care Coordination” sub-measure, with a commenter noting that the repetition of the language, “doctors, nurses, and other hospital staff” may confuse patients and instead recommended collapsing the list into “hospital team” to be more inclusive and aligned with health literacy standards. Another commenter expressed concern about the use of “other hospital staff” because they stated that other hospital staff should not be informed about a patient's care. The commenter recommended removing the term or better defining it in the question. A commenter also recommended modifying the question in the “Discharge Information” sub-measure about whether patients have the help they need after they leave the hospital to address needed support for family caregivers. A commenter recommended limiting the addition of new questions to only those that provide meaningful and actionable data because they stated that repetitive questions can limit response rates.
Response: The phrase “doctors, nurses, and other hospital staff” has been used since the inception of HCAHPS and was subject to multiple rounds of testing during HCAHPS development and the current refinement of HCAHPS content. These efforts confirmed that the phrase is clearly understood by patients. Cognitive testing indicated that patients understood that other hospital staff included staff such as individuals providing therapy who should be aware of the patient's condition.
Patients indicated that how well “doctors, nurses, and other staff work together or as a team” in caring for a patient was the most important information to have in determining what their care would be like at a particular hospital. The term “other hospital staff” refers to anyone else involved in the patient's care during their hospital stay, including but not limited to those who take patients for X-rays or medical tests, individuals providing treatment or therapy during the in-patient stay, and those who participate in discharge planning. Cognitive testing indicates that repeated use of this phrase ensures that patients understand who is included; terms such as “Care team” and “Hospital team” are less familiar to patients. Based on these efforts, we determined that the survey language is clear and intelligible to patients.
We agree with the commenter that the addition of new questions should be limited to only those that provide meaningful and actionable data and have identified that the updates to the HCAHPS Survey measure provide such data. A CMS contractor convened a technical expert panel that engaged physicians, nurses, academics, and representatives of hospitals, insurers, and patient advocacy groups to assess the actionability of all new items proposed for testing to ensure we focused the new content on actionable events, and we note that every proposed question had statistical evidence that it improved measurement of the sub-measure to which it belonged. [ 427 ] We will consider how to incorporate the concept of language preferences into the sub-measures in future program years.
Comment: A commenter did not support the “Information About Symptoms” sub-measure because they ( print page 69502) believed it is substantially similar to the “Discharge Information in Writing” question. A commenter questioned the sub-measure's intent as they stated the question seems to be more about whether the hospital gave the patient information rather than whether the patient was able to understand the information. A few commenters made recommendations about the new “Information About Symptoms” question including modifying the question to focus more on the patient's understanding than on the task of handing over education, and explicitly mentioning the patient in the question to reinforce a patient-and-family centered care model. A commenter recommended modifying the “Discharge Information in Writing” question to incorporate information about systems and to say, “in your preferred language in writing.”
Response: We appreciate the commenter's concern; however, through our 2021 mode experiment, focus groups, technical expert panel, and literature review, we have ensured that the questions proposed, including the “Information About Symptoms” question, add unique, non-redundant information about key aspects of patient experience of care. The “Information About Symptoms” sub-measure focuses on information communicated to a patient's family or caregiver, an aspect of care that patients identified as critical to measuring quality. In contrast, the “Discharge Information in Writing” question asks about written information provided to the patient, which is also important to measuring quality. These are different topics and are measured via separate items to ensure that the data collected are actionable. We refer the commenter to the PRMR report for additional information on the testing we conducted. [ 428 ]
We also note that the “Information About Symptoms” question captures an important aspect of hospital care identified by patients and caregivers participating in focus groups. Cognitive testing demonstrated that the “Information About Symptoms” question was accurately and consistently interpreted, as described in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36299 ). We agree that ensuring patient comprehension is important and will take this feedback into consideration, along with the suggestion to include “in your preferred language in writing,” in the “Discharge Information in Writing” question for future program years.
Comment: A few commenters offered additional recommendations including that CMS report survey results by race and ethnicity to aid in reducing disparities. Another commenter recommended that CMS should talk to employers and other purchasers to utilize HCAHPS for their maternity populations.
Response: We appreciate the commenters' recommendations. We will consider reporting survey results by race and ethnicity in future program years. Maternity patients have been eligible for the HCAHPS Survey measure since its inception. Our research indicates that maternity patients are particularly affected by mode of survey administration; we recommend that hospitals carefully choose the mode of HCAHPS administration that will fully capture their entire patient population. [ 429 430 ]
Comment: A commenter recommended using Short Message Service (SMS) or other forms of text messages as an additional survey mode because they stated it would help increase response rates.
Response: While the current web administration mode does not include a text message option, we will take these recommendations into consideration for future program years while also taking into consideration the Telephone Consumer Protection Act (TCPA) requirements. We evaluated the possibility of using text message as a mode for survey implementation but determined that varying standards across states, possible charges for text messages, as well as the requirements of TCPA, make a text survey infeasible for the national, standardized HCAHPS Survey measure at this time. However, we will continue to explore this as an option for the future.
Comment: A commenter recommended additional financial support for under-resourced hospitals to help them move beyond process improvements.
Response: We cannot provide additional financial support for under-resourced hospitals as part of the HCAHPS Survey measure at this time.
We also received public comments on the specific addition of the “Care Coordination” sub-measure to the HCAHPS Survey measure.
Comment: Many commenters specifically supported the adoption of the “Care Coordination” sub-measure because they stated that it is broader and clearer than the “Care Transition” sub-measure, addresses important dimensions of patient experience not previously addressed, and provides information about how well a patient felt their care team worked together. Several commenters noted that the new “Care Coordination” sub-measure reflects CMS's commitment to the role of patient reported experiences and another commenter stated it would serve to reduce overlap between care transition and discharge information. A few commenters stated the new sub-measure would enhance the HCAHPS Survey measure, better capture patient experience of hospital care, and improve understanding of the challenges faced in coordinating care across the care continuum. A commenter expressed support for the “Care Coordination” measure because they stated it is broader than the “Care Transition” sub-measure but noted that care coordination is important at care transition.
Response: We thank commenters for their support of the new “Care Coordination” sub-measure. We agree that care coordination is important at care transition, and have determined, through the four patient focus groups that were conducted before proposing these updates, that the updated question set captures the key aspects of patient experience of care including at the point of care transition. We reiterate that the new “Care Coordination” sub-measure focuses on how well hospital staff worked together and whether doctors and staff worked to make care transition plans for the patient post-hospitalization.
Comment: A commenter did not support removing the question, “During this hospital stay, staff took my preferences and those of my family or caregiver into account in deciding what my health care needs would be when I left,” because the commenter stated the new “Care Coordination” questions do not inherently take personal preferences into account.” The commenter recommended maintaining this question and the new “Care Coordination” questions.
Response: We appreciate the commenter's concern, however, the question asking about preferences was removed because it was perceived by patients in the focus groups as ( print page 69503) duplicative of existing and new survey questions, as described in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36294 ).
Comment: A commenter recommended broadening the “Care Coordination” sub-measure to include whether family caregivers received any needed support to capture additional data on caregiver support.
Response: We thank the commenter for the recommendation and will consider further broadening the “Care Coordination” sub-measure in future program years. We note patients and caregivers identified these questions, as written, as very important to addressing key aspects of patient care.
We also received public comments on the specific addition of the “Restfulness of Hospital Environment” sub-measure to the HCAHPS Survey measure.
Comment: Several commenters supported the addition of the new “Restfulness” sub-measure, believing that the questions would enhance the HCAHPS Survey measure and are significant contributions that reflect CMS's commitment to expanding the role of patient reported experiences. A commenter supported the “Restfulness” sub-measure because they stated that rest and sleep are foundational occupations that affect daily patient function and quality of life, and another supported the addition of the “Restfulness” sub-measure, but expressed concern that combining all hospital staff into a single question complicates hospitals' work. Another commenter supported the wording of “doctors, nurses, and other hospital staff” because they stated a patient may not always know what type of staff a specific person is, and thus the wording lessens the possibility of inaccurate survey responses. A commenter specifically supported the “Quietness” question because it enhances the HCAHPS Survey measure and better captures patient experience of hospital care.
Response: We thank commenters for their support of the new “Restfulness” sub-measure.
Comment: A few commenters did not support the “Restfulness” sub-measure because they stated the questions are too subjective and may create confusion. A few commenters also stated that hospitals by nature are not restful environments and proper care and safety should take precedence over rest. A few commenters expressed concern that the questions may divert attention from more critical elements of care. A few commenters requested additional testing information and data about how patients interpret the “Restfulness” sub-measure in light of their concerns that the questions are subjective and stated that there may be important reasons to interrupt a patient's rest. A commenter recommended that the sub-measure be sent to a workgroup to make changes to the questions to ensure there are no unintended consequences. A commenter recommended removing the “Quietness” question altogether.
Response: Cognitive testing demonstrated that the new questions were accurately and consistently interpreted by patients. Additionally, we have identified the need for this sub-measure through focus groups and cognitive interviews with patients and caregivers, discussions with technical experts, and literature reviews that were conducted by a CMS contractor who made recommendations to CMS. “Restfulness of Hospital Environment” was deemed an important new topic to add to the HCAHPS Survey measure based on stakeholder feedback, including that from hospital staff and patient groups. Clinicians on our technical expert panel, patients, and patient advocates supported these questions. This sub-measure can be satisfied by avoiding needless disruptions and explaining to patients the importance of necessary ones. We have also conducted reliability and validity testing at the expected average number of completed surveys per hospital and therefore do not agree that additional review by a workgroup is necessary at this time. We refer the commenter to the PRMR report for additional information on the testing conducted on these updates, which did not identify any unintended consequences. [ 431 ]
In response to the request to remove the “Quietness” question, we note that this question is already included in the current version of the HCAHPS Survey measure as a single-question sub-measure, and the question itself is not changing in the updated version of the survey.
Comment: Several commenters expressed concerns about the “Restfulness” sub-measure including concerns about the validity and reliability.
Response: We appreciate the commenters' concerns. We reiterate that we identified the need for these updates through focus groups and cognitive interviews with patients and caregivers, discussions with technical experts, and literature reviews. The new questions within the “Restfulness” sub-measure fill a gap related to providing a restful and healing environment within the hospital setting and support our goal of including measures related to person-centered care. We also reiterate that we have conducted reliability and validity testing at the expected average number of completed surveys per hospital. We refer the commenter to the PRMR report for additional information on the testing conducted on these updates, including testing specifically on the “Restfulness” sub-measure. [ 432 ]
Comment: A few commenters also requested clarification on the extent of the risk adjustment approach that may account for differences in the score of the “Restfulness” sub-measure.
Response: We thank commenters for their request. The HCAHPS patient-mix adjustment (risk adjustment) approach accounts for factors not under a hospital's control that affect how patients answer survey items, such as, patients' service line by sex, age, education, language spoken at home, and self-rated overall and mental health. The same patient-mix model used for all other HCAHPS sub-measures was proposed for the “Restfulness of Hospital Environment” sub-measure. The current HCAHPS patient-mix adjustments can be found at https://hcahpsonline.org/en/modepatient-mix-adj/#jan2023publiclyreported . An example of patient-mix adjustment for the updated HCAHPS Survey measure can be found at https://hcahpsonline.org/globalassets/hcahps/training-materials/2024_training-materials_slides.pdf .
Comment: A few commenters expressed concerns that the sub-measure may result in providers prioritizing a quiet environment over providing necessary medical rounds, and that if certain services need to be provided overnight, the disruption may affect performance on the survey. A few commenters also expressed concern that the sub-measure may unfairly penalize or disadvantage certain hospitals such as quaternary hospitals with high acuity patients, hospitals in densely urban neighborhoods, and hospitals with dual occupancy rooms. A commenter supported the inclusion of the new “Restfulness” questions, but expressed concern that rooms with one or more other patients are hard to control and that prioritizing restfulness could lead hospitals to limit family visitations. Another commenter recommended monitoring implementation to ensure no unintended consequences. A ( print page 69504) commenter recommended reconsidering implementation of the “Restfulness” sub-measure because they stated that the practice of checking on patients regularly throughout the night could diminish with this sub-measure, which could lead to more falls. Another commenter recommended removing the “Rest and Recover” question, which asks, “During this hospital stay, did doctors, nurses, and other hospital staff help you rest and recover?”, because hospitals provide 24/7 care, and providers often need to interrupt patients throughout the night for treatment or to take vitals.
Response: The HCAHPS Survey measure is designed to produce standardized information about patients' perspectives of care that allows comparison of hospitals on topics that are important to consumers. While we acknowledge that commenters' concerns about prioritization of safe patient care practices is valid and that hospitals should be conducting necessary medical rounds, the survey is designed to measure patients' experience of care and the care should be provided to promote as restful an experience as possible while delivering the necessary clinical care. The goal of the “Restfulness of the Hospital Environment” items is not merely comfort, but to promote recovery. Restfulness can be accomplished with no reduction in rounding. We remind commenters that the HCAHPS Survey measure has always included the “Quietness” question, which would be retained in the updated HCAHPS Survey measure in the new “Restfulness of Hospital Environment” sub-measure. There is evidence that both quiet and rest are important for recovery. [ 433 ] [ 434 ] We are not aware of any unintended consequences from the “Quietness” question. “Restfulness of the Hospital Environment,” which is based on one current HCAHPS item and two new HCAHPS items, was deemed an important new topic to add to HCAHPS based on stakeholder feedback from hospital staff and patient groups. The concept of “rest and recovery” was important to the technical expert panel convened by a CMS contractor to provide feedback on updating the HCAHPS Survey measure. In particular, the panel encouraged CMS to add items that asked about rest and/or recovery, noting the concept is distinct from sleep, which is also important to recovery and is independently important in care. The items were designed and worded to acknowledge that activities such as rounding, tending to other patients, or managing emergencies are necessary. Cognitive testing suggests that if patients are told why they are being woken they do not rate this item negatively. The goal of this measure is to discourage needless disruptions and to encourage communication between providers and patients. Dual occupancy rooms, like hospitals with lower staffing, may result in poorer patient experience; the HCAHPS Survey measure seeks to measure and report actual performance.
Comment: Several commenters offered recommendations to modify the “Restfulness” questions. Their recommendations included removing the “Rest and Recover” question (Question 18) because they stated it is unclear what “rest and recover” means and feedback around “rest” is captured by other questions in the “Restfulness of the Hospital Environment” sub-measure; combining questions 8, “During this hospital stay, how often are you able to get the rest you needed?” and 18, “During this hospital stay, did the doctors, nurses, and other hospital staff help you rest and recover?” into one question asking if the hospital staff helped the patient rest and recover because they stated it speaks more to the care provided by the staff; modifying the language to say “During this hospital stay, did your hospital team help you rest?” because they stated a team approach prevents the possibility of incorrect survey responses; and asking patients to report the ability of the environment to support “rest” versus “rest you need” because they stated rest may come secondary to treatment needs and a patient may have difficulty getting a good night's sleep anywhere except their own bed. A few commenters expressed concerns with the term “recover” with one believing that rest and recovery are two different dimensions and another noting that not all inpatients are anticipated to recover from their condition. The commenters recommended removing the words, “and recover” to better focus on “restfulness.”
Response: We appreciate the commenters' recommendations. However, we wish to note that the questions as currently written were reviewed by patient focus groups and tested for reliability and validity, and we therefore do not agree with modifying the wording or combining the questions as the commenters have suggested earlier. Cognitive testing suggests that if patients are told why they are being woken they do not rate this item negatively. The goal of this measure is to discourage needless disruptions and to encourage communication between providers and patients. Empirical testing of the “Restfulness of Hospital Environment” sub-measure in the 2021 mode experiment provides strong support for each question in the sub-measure and the sub-measure as a whole. We note that the “Quietness” question has always been included in the HCAHPS Survey measure and will provide continuity in both public reporting and in the Hospital VBP program.
We received public comments on the specific addition of the “Information about Symptoms” sub-measure to the HCAHPS Survey measure.
Comment: Several commenters expressed support for the adoption of the new “Information About Symptoms” single-item sub-measure because they stated the addition of the “Information About Symptoms” question is a significant contribution to committing to the role of patient reported experiences that would enhance the HCAHPS Survey measure. A few commenters supported the sub-measure with a commenter stating that it can provide important information about how well a patient felt that his or her care team assisted with post-discharge planning, and another commenter noting that the sub-measure has been shown to improve patient outcomes. A commenter recommended strengthening the question to also include information on how to address symptoms, such that the question would read, “During this hospital stay, did doctors, nurses, or other hospital staff give your family enough information about what symptoms or health problems to watch for after you left the hospital and how to address them?” because they stated that this revision would provide useful information for hospitals, consumers, and caregivers.
Response: We thank the commenters for their support of the new “Information About Symptoms” sub-measure, and we will continue to consider additional ways to strengthen the survey, including revising the question to include information on how to address a patient's symptoms in future program years. Any changes to a question's wording would need to be tested before we can consider incorporating them in future years.
We also received public comments on the specific modification of the “Responsiveness of Hospital Staff” sub- ( print page 69505) measure to the HCAHPS Survey measure.
Comment: Several commenters supported adopting the modifications to the “Responsiveness of Hospital Staff” sub-measure because they enhance the person-centeredness of care, represent current workflows within hospitals, are more inclusive of different hospital strategies, and accurately measure the patient experience. A few commenters specifically supported the removal of the “Call Button” question because they stated that the technology is evolving, and the new questions better reflect current practices.
Response: We thank the commenters for their support of the modifications to the Responsiveness of Hospital Staff sub-measure. We agree that the modifications are more inclusive and accurately measure the patient experience. We also appreciate the support of the removal of the “Call Button” question.
Comment: A few commenters did not support the verbiage of “right away” and “as soon as you needed” in the new “Responsiveness” sub-measure and recommended rewording or removing the questions because they stated the language is too subjective. Some commenters suggested that CMS should develop alternative phrasing to enhance clarity and accessibility such as replacing “right away” with “quickly,” while another commenter recommended removing the word “right away” entirely from the question because they stated that it makes the question too wordy and indicates that the question pertains only to those who need urgent help.
Response: We thank the commenters for their suggestions regarding the wording of the “Responsiveness of Hospital Staff” sub-measure, however, we do not agree that the wording of the question should be changed. The terms “right away” and “as soon as you needed” are commonly used in CAHPS surveys to identify care needs that are time-sensitive. We discussed the item wording and terms with patients in multiple focus groups and cognitive interviews and found that the uses of “right away” and “as soon as you needed” promote common, consistent interpretation of the survey item across patients. As part of the cognitive interviews, we discussed their understanding of the terms included in these questions and found no issues.
Comment: A commenter also questioned whether patients tend to respond more positively to questions framed around immediate responsiveness versus those related to the call bell and recommended that CMS grant access to the research demonstrating the impact of the modified questions.
Response: We refer the commenter requesting access to our research to the PRMR report for additional information on the testing we conducted on these updates. [ 435 ] Because the “Call Button” question and the new “Help Right Away” question, which asks, “During this hospital stay, when you asked for help right away, how often did you get help as soon as you needed?” were not tested in the same study, it is not possible to confidently compare their mean scores. We do, however, have strong evidence of the reliability and validity of the new “Help Right Away” question, which was developed in response to stakeholder concerns about the “Call Button” question.
We also received public comments on the specific removal of the “Care Transition” sub-measure from the HCAHPS Survey measure.
Comment: Several commenters supported the removal of the “Care Transition” sub-measure because they stated that the “Care Coordination” sub-measure encompasses a broader range of questions than “Care Transition” did and removing “Care Transition” would reduce repetitiveness and overlap. A commenter stated the changes would enhance the HCAHPS Survey measure and another commenter supported that the removal of the “Care Transition” sub-measure in conjunction with the adoption of the “Care Coordination” sub-measure would ensure there is not an increase to the survey's length.
Response: We thank the commenters for their support of the removal of the “Care Transition” sub-measure and agree that the “Care Coordination” sub-measure broadens the current “Care Transition” sub-measure.
Comment: A commenter recommended that the updates to the survey be delayed until the conclusion of the Magnet application period because the Magnet teams look closely at measures within the “Care Transition” dimension so the updates could impact entities undergoing Magnet submission.
Response: We understand that outside credentialing programs, such as Magnet, may employ the HCAHPS Survey measure in their own eligibility, assessment, or credentialing processes. There are numerous credentials that hospitals can choose to pursue, and while we respect the commitment to excellence, we do not control or oversee such secondary uses and cannot base our implementation timelines on outside credentialing. We invite these organizations to familiarize themselves with the updated HCAHPS Survey measure to assess its suitability for their needs.
We also received public comments on the specific modifications to the “About You” section of the HCAHPS Survey measure.
Comment: A few commenters specifically supported the updates to the “About You” section, with a few supporting the alphabetization of the response options for the race and ethnicity questions and one supporting the modified language spoken at home question because they stated it makes survey completion less burdensome. A commenter supported incorporating the “Hospital Stay Planned in Advance” question because they stated it more appropriately captures the reason for admission. Another commenter supported the updates but recommended aligning the HCAHPS Survey measure questions with the updated OMB standards for maintaining, collecting, and presenting federal data on race and ethnicity to reduce disparities and harmonize data collection across agencies.
Response: We thank the commenters for their support of the updates to the “About You” section, and we agree that the updates make the survey less burdensome to complete. We appreciate the recommendation to align with the OMB standards and harmonize data collection across agencies. We will take this into consideration in future program years.
Comment: A few commenters did not support the new “Hospital Stay Planned in Advance” question, with a commenter believing the new verbiage is ambiguous and may result in a lack of meaningful data, and another commenter expressed concern that the new question could have unintended consequences for how patient mix is adjusted and recommended not finalizing.
Response: As described in the FY 2025 IPPS/LTCH PPS proposed rule, the new “Hospital Stay Planned in Advance” question would account for differences in this preadmission characteristic, as an unplanned hospital stay can result in worse patient experiences than if the hospital stay had been planned ( 89 FR 36300 ). The cognitive testing that we conducted indicated that the new question is better understood than the current ( print page 69506) “Admission through the Emergency Room” question.
Comment: Several commenters expressed concerns about the updates to the “About You” section, including a few commenters who expressed concerns about limiting the number of language response options in the “About You” section because they stated restricting the language options could undermine and underrepresent patient experiences, particularly when the release of the web-first modalities has been found to increase response rates for many of the language options that CMS is proposing to remove. A few commenters recommended maintaining or even expanding the range of languages identified in the survey because they stated it would provide a more accurate picture of enrollee demographics and inform decisions regarding survey translation. A commenter recommended monitoring the percent of responses in the “Other” category and broadening the options again in the future.
Response: We thank commenters for their feedback about limiting the number of language response options in the “About You” section. We note that reciting a long list of languages in question 29 increases survey burden for patients, especially in the telephone mode. Additionally, the response option, “Another language,” which remains available as a response option for anyone who does not speak English, Spanish, or Chinese, would more efficiently gather important information for use in patient-mix adjustment. Patient-mix adjustment of “Language Spoken at Home” will employ four categories: “English,” “Spanish,” “Chinese,” and “Another language.” Because patient-mix adjustment combines all other languages into one category, the response option “Another language” will improve survey efficiency and reduce burden, especially on telephone surveys. [ 436 ] Official translations of the HCAHPS Survey in Spanish, Chinese, Russian, Vietnamese, Portuguese, German, Tagalog, and Arabic will continue to be available for use, though only English and Spanish versions will be required. We also appreciate the recommendations to maintain or increase the range of language options, but we remind commenters that we identified these changes as an effort to make survey administration more efficient and reduce respondent burden, especially in the telephone mode of survey administration. Additionally, we will continue to monitor trends in patient language and will consider broadening the options again in the future.
Comment: A few commenters expressed concerns about the new “Hospital Stay Planned in Advance” question because they stated the question may require more clarity and further guidance to ensure that patients understand the question as intended. A commenter expressed concern that the “Hospital Stay Planned in Advance” question has the possibility to be doubly adjusted with a service line adjustment for maternity and another potential adjustment for the admission being unplanned. Another commenter expressed concern that in cases related to hospital stays for childbirths, the hospital admission date for a vaginal delivery could be viewed by a patient as either unplanned or planned in advance. The commenter therefore recommended further guidance to survey respondents about how to answer the question, including covering the most common situations where there may be ambiguity.
Response: We appreciate commenters' feedback about the new “Hospital Stay Planned in Advance” question. Cognitive testing indicated that the question is well understood and is better understood than the current “Admission Through the Emergency Room” question. Our results from the 2021 mode experiment also indicate that adjusting for unplanned stays improves the accuracy of HCAHPS scores. Patient-mix adjustment is implemented via multiple regression in such a way that ensures that double adjustment does not occur. When an adjustor is added, any “overlapping” adjustment with other adjustors is automatically removed. Data collected in the 2021 mode experiment also indicated that maternity care is not often reported by patients as being unplanned. We note that the response options for the new “Hospital Stay Planned in Advance” question offer patients the choice of selecting among “Yes, definitely,” “Yes, somewhat,” and “No.” The response option, “Yes, somewhat,” is available for hospital stays that were neither “yes, definitely” planned in advance, nor “no,” unplanned.
Comment: A few commenters made recommendations about the “About You” section including a few commenters that made recommendations about race and ethnicity data. Their recommendations included implementing OMB's revised standards for the collection of race and ethnicity data, combining the two race and ethnicity questions into one question on both race and ethnicity, and adding a Middle Eastern or North African category. A commenter recommended that race and ethnicity be separated instead of grouped together.
Response: We appreciate the commenters' recommendations for additional updates to the race and ethnicity questions and will take these into consideration in future program years.
We also invited public comment on the proposed adoption of the updated HCAHPS Survey measure for the Hospital IQR Program beginning with the CY 2025 reporting period/FY 2027 payment determination and the PCHQR Program beginning with the CY 2025 reporting period/FY 2027 program year.
Comment: A commenter specifically supported the adoption of the updates to the HCAHPS Survey measure in the PCHQR Program but recommended examining whether “Restfulness” disproportionately penalizes urban hospitals.
Response: We thank the commenter for their support of the adoption of the updates in the PCHQR Program. The “Restfulness of Hospital Environment” sub-measure was identified to have good hospital-level reliability (0.729) at the expected average number of completed surveys per hospital. Testing found no evidence that the measure is less accurate for urban than rural hospitals.
Comment: A commenter specifically supported the modified public reporting schedule for the Hospital IQR Program.
Response: We thank the commenter for their support of the public reporting schedule.
Comment: A commenter recommended that CMS complete validity testing and receive CBE endorsement to understand the strength of the correlations of the multi-item and single-item measures with the overall measures before implementing the changes into the Hospital IQR Program.
Response: We refer the commenter to the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36299 ) and this final rule where we outline the reliability and validity testing we conducted on all of the updates we proposed to the HCAHPS Survey measure. We will submit the updated HCAHPS Survey measure to the current CBE for endorsement in Fall 2025. We note that section 1886(b)(3)(B)(viii)(IX)(bb) of the Act states that in the case of a specified area or medical topic determined appropriate by the Secretary for which a feasible and practical measure has not been endorsed by the entity with a ( print page 69507) contract under section 1890(a) of the Act, the Secretary may specify a measure that is not endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We have determined that the updates to the HCAHPS Survey measure are appropriately specified. The HCAHPS Survey measure remains endorsed, and the updated survey only modifies some of the questions and sub-measures within the survey. The HCAHPS Survey measure is designed to produce standardized information about patients' perspectives of care that allow objective and meaningful comparisons of hospitals on topics that are important to consumers, and these updates would improve the feedback we receive directly from patients on hospital performance. Therefore, we are adopting these updates to the measure before the updates receive CBE endorsement.
Comment: A commenter recommended rolling out the new questions beginning with the CY 2026 discharges because health systems and vendors will only have about three months to transition to the new requirements once the final rule is released in August.
Response: Since these updates to the HCAHPS Survey measure are limited to changes to some of the survey questions, we have identified that there would be sufficient time from public display of this final rule on or about August 1, 2024, and when the updated survey would begin to be administered to patients who are discharged in January 2025. The updated HCAHPS Survey has been available in all survey modes on the official HCAHPS website since May 2024, including the official Spanish translation, and the Quality Assurance Guidelines for the updated HCAHPS Survey measure have been available since May 2024. As noted in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36294 ), the updated version of the HCAHPS Survey measure would not result in any changes to the survey administration or other reporting requirements. We note that changes to the administration of the survey were finalized in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59196 through 59201 ) and that approved HCAHPS Survey vendors were trained on the updated HCAHPS Survey measure in May 2024. We, therefore, have determined that hospitals would not need additional time before the updated survey is implemented.
After consideration of the public comments received, we are finalizing adoption of the updated HCAHPS Survey measure in the Hospital IQR and PCHQR Programs as proposed.
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to adopt an updated version of the HCAHPS Survey measure so that IPPS hospitals and PCHs can report patient responses to the updated survey for purposes of the Hospital IQR Program and PCHQR Program, respectively, beginning with January 1, 2025, discharges ( 89 FR 36298 through 36300 ). We also proposed to adopt the updated version of the HCAHPS Survey measure for purposes of the Hospital VBP Program in section IX.B.2.g. of this final rule; however, section 1886(o)(2)(C)(i) precludes us from doing so until we have specified the updates under the Hospital IQR Program and included them on Care Compare for at least one year prior to the beginning of the performance period for such fiscal year. For this reason, we proposed to adopt the updated version of the HCAHPS Survey measure beginning with the FY 2030 program year in the Hospital VBP Program. However, to relieve hospitals of the burden of having to use two different versions of the survey between FY 2027 and FY 2029, we proposed that hospitals would be able to administer the updated version of the survey starting with January 1, 2025 discharges, and for the purposes of the Hospital VBP Program, we would only score hospitals on the six dimensions of the HCAHPS Survey measure that would remain unchanged from the current version of the survey.
In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to modify scoring to not include the “Responsiveness of Hospital Staff” and “Care Transition” dimensions from scoring in the Hospital VBP Program's HCAHPS Survey measure in the Person and Community Engagement domain for the FY 2027 through FY 2029 program years ( 89 FR 36300 through 36301 ). As noted earlier, we must collect and publicly report four quarters of data on the updated HCAHPS Survey measure before the updates can be adopted into the Hospital VBP Program. As described in section IX.B.2.g(2) of this final rule, the updates to the “Responsiveness of Hospital Staff” dimension would be adopted in the Hospital VBP Program beginning with the FY 2030 program year along with the rest of the updates to the survey after the statutory requirements of section 1886(o)(2)(C)(i) of the Act have been met. As described in section IX.B.2.g(3) of this final rule, scoring on the updated “Responsiveness of Hospital Staff” dimension would begin with the FY 2030 program year. In addition, the “Care Transition” dimension in the current version of the survey would be removed permanently in the proposed updated HCAHPS Survey measure beginning with the FY 2030 program year. Until these updates can be adopted in the Hospital VBP Program beginning in FY 2030, we are excluding these dimensions from scoring for the FY 2027 through FY 2029 program years.
With the adoption of the proposal to not score the “Care Transition” and “Responsiveness of Hospital Staff” dimensions in the Person and Community Engagement domain for the FY 2027 through FY 2029 program years, only six dimensions would continue to be used in the Hospital VBP Program for FY 2027, FY 2028, and FY 2029. By excluding these two dimensions from scoring within the Hospital VBP Program for the FY 2027 through FY 2029 program years, hospitals can continue to be scored on the remaining unchanged dimensions of the current HCAHPS Survey measure until the proposed updated HCAHPS Survey measure could be adopted for use in the Hospital VBP Program beginning in FY 2030.
We proposed to score hospitals only on these six dimensions because we cannot score hospitals on any of the new or updated dimensions associated with the updated HCAHPS Survey measure until they have been adopted and reported in the Hospital IQR Program for one year prior to the beginning of the first performance period of their use in the Hospital VBP Program ( 89 FR 36300 through 36301 ). These six unchanged dimensions of the HCAHPS Survey measure would be:
- “Overall Rating.”
We proposed to modify the scoring such that for each of these six dimensions, Achievement Points (0-10 points) and Improvement Points (0-9 points) would be calculated, the larger of which would be summed across these six dimensions to create a pre- ( print page 69508) normalized HCAHPS Base Score of 0-60 points (as compared to 0-80 points with the current eight dimensions). The pre-normalized HCAHPS Base Score would then be multiplied by 8/6 (1.3333333) and then rounded according to standard rules (values of 0.5 and higher are rounded up, values below 0.5 are rounded down) to create the normalized HCAHPS Base Score. Each of the six unchanged dimensions would be of equal weight, so that, as currently scored, the normalized HCAHPS Base Score would range from 0 to 80 points. HCAHPS Consistency Points would be calculated using our current methodology and would continue to range from 0 to 20 points. Like the Base Score, the Consistency Points Score would only consider scores across the remaining six unchanged dimensions of the Person and Community Engagement domain. The final element of the scoring formula, which would remain unchanged from the current formula, would be the sum of the HCAHPS Base Score and the HCAHPS Consistency Points Score for a total score that ranges from 0 to 100 points. In the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50065 ) and the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49565 ), we adopted a similar modified scoring methodology when the Care Transition sub-measure was added to the current HCAHPS Survey measure in the Hospital VBP Program.
This scoring modification would ensure that hospitals can continue to receive scores on the dimensions of the HCAHPS Survey measure that would remain unchanged in the current survey and would provide a period of transition until the Hospital VBP Program can adopt the updates to the survey. The updated version of the HCAHPS Survey measure would be adopted in the Hospital IQR and PCHQR Programs beginning with January 1, 2025 discharges, however, those updated sub-measures would not be scored as dimensions for the Hospital VBP Program until the FY 2030 program year. We reiterate that hospitals would only have to circulate one version of the HCAHPS Survey measure at a time.
We invited public comment on this proposal to modify scoring on the HCAHPS Survey measure in the Hospital VBP Program for the FY 2027 through FY 2029 program years to only score on the six dimensions discussed earlier.
Comment: Many commenters expressed support for the proposal to modify scoring in the Hospital VBP Program for the FY 2027 through FY 2029 program years because they stated it prevents duplicate, simultaneous survey reporting, minimizes burden and inconvenience, and allows hospitals to consistently administer a single survey under both the Hospital IQR and Hospital VBP Programs. A few commenters commended CMS for respecting statutory requirements, considering stakeholder feedback, and allowing hospitals time to implement the new survey. A commenter recognized that the modifications necessitate a commensurate adjustment to the Hospital VBP Program scoring methodology, and a commenter conditionally supported the scoring modifications with the recommendation that CMS ensure that the resulting scores of the modified HCAHPS Survey measure would still be reliable and valid.
Response: We appreciate commenters' support of the scoring modifications for the FY 2027 through FY 2029 program years and agree that these modifications ensure that we meet statutory requirements and allow hospitals to consistently administer a single survey under both the Hospital IQR and Hospital VBP Programs. In response to the recommendation, we have determined that the resulting scores of the modified HCAHPS Survey measure would still be reliable and valid given that we are utilizing the normalization methodology, where, as described in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36300 through 36301 ), for each of these six dimensions, Achievement Points (0-10 points) and Improvement Points (0-9 points) would be calculated, the larger of which would be summed across these six dimensions to create a pre-normalized HCAHPS Base Score of 0-60 points (as compared to 0-80 points with the current eight dimensions). Then, that pre-normalized HCAHPS Base Score would be multiplied by 8/6 (1.3333333) and rounded according to standard rules (values of 0.5 and higher are rounded up, values below 0.5 are rounded down) to create the normalized HCAHPS Base Score. Each of the six unchanged dimensions would thus be of equal weight, so that just as hospitals are currently scored, the normalized HCAHPS Base Score would range from 0 to 80 points. This normalization methodology ensures that the updated survey measure is scored on the same scale as it is on the current HCAHPS Survey measure and therefore reliable and viable.
Comment: A commenter expressed concern that only using six HCAHPS Survey sub-measures places greater pressure on the scores in the existing dimensions which they stated creates performance stress for hospitals and payment implications. The commenter recommended delaying operationalizing the scoring modifications for a minimum of one additional year.
Response: We appreciate the commenter's concern, but we disagree that the scoring modification would create performance stress for hospitals and payment implications. Normalizing the scores accounts for some of the differences in the number of dimensions. During the FY 2027 through FY 2029 program years, the six unchanged dimensions will each equally account for 16.7 percent of the Person and Community Engagement domain, up from 12.5 percent under the current survey. However, we determined this was a smaller impact on hospitals than pausing scoring of the entire survey for the three transitional program years. We refer the commenter to the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36300 through 36301 ) for additional information on how the scoring is modified and normalized. We adopted a similar normalization methodology in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50065 ) and the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49565 ) when the Care Transition sub-measure was added to the current HCAHPS Survey measure in the Hospital VBP Program.
Comment: A commenter requested clarification on whether the domain weights for the domains in the Hospital VBP Program would remain the same through the FY 2027 through FY 2029 program years when the scoring of the HCAHPS Survey measure is modified.
Response: We thank the commenter for their question and affirm that the domain weights for the Hospital VBP Program would remain unchanged, with each of the four domains in the program being weighted at 25 percent as before.
After consideration of the public comments received, we are finalizing adoption of the modifications to scoring in the Hospital VBP Program for the FY 2027 through FY 2029 program years as proposed.
As described in the FY 2025 IPPS/LTCH PPS proposed rule and section IX.B.2.e. of this final rule, the modifications to the proposed updated version of the HCAHPS Survey measure include adding three new sub-measures, “Care Coordination,” “Restfulness of Hospital Environment,” and “Information About Symptoms” to the survey ( 89 FR 36298 through 36300 ). ( print page 69509) The updates also include removing the existing “Care Transition” sub-measure and modifying the existing “Responsiveness of Hospital Staff” sub-measure. Additionally, we proposed to adopt the updated HCAHPS Survey measure in the Hospital VBP Program beginning with the FY 2030 program year and additional scoring modifications beginning with FY 2030 ( 89 FR 36301 through 36304 ). This timeline would allow for the updated HCAHPS Survey measure to be adopted and publicly reported under the Hospital IQR Program for one year, as statutorily mandated. We describe the adoption of these updates and scoring modifications in the following sections.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36301 through 36304 ), we proposed to adopt the updated HCAHPS Survey measure in the Hospital VBP Program beginning with the FY 2030 program year to align with the adoption of the updated HCAHPS Survey measure that we proposed to adopt in the Hospital IQR Program, as described in section IX.B.2.e. of this final rule. Under this proposal, the updated HCAHPS Survey measure would have been publicly reported for one year in the Hospital IQR Program prior to the beginning of the performance period for the HCAHPS Survey measure in the Hospital VBP Program for the FY 2030 program year, which consists of a performance period of CY 2028 and a baseline period of CY 2026.
We note that the number and content of dimensions from the proposed updated HCAHPS Survey measure in the Person and Community Engagement Domain in the Hospital VBP Program in FY 2030 differs slightly from the number and content of the sub-measures in the Hospital IQR and PCHQR Programs. Namely, the “Cleanliness” and “Information about Symptoms” sub-measures are single-item sub-measures in the updated HCAHPS Survey measure in the Hospital IQR and PCHQR Programs, but they would be combined into one dimension in the updated HCAHPS Survey measure for the Hospital VBP Program beginning with the FY 2030 program year.
The dimensions in the Person and Community Engagement Domain in the Hospital VBP Program beginning with the FY 2030 program year are:
- “Responsiveness of Hospital Staff,”
- “Cleanliness and Information About Symptoms,”
- “Overall Rating of Hospital,”
- “Care Coordination,” and
- “Restfulness of Hospital Environment.”
We refer readers to Table IX.B.2-03 for the timelines for the current and newly adopted HCAHPS Survey dimensions for the Hospital VBP Program.
In the updated HCAHPS Survey measure, the “Care Transition” dimension is removed. The new “Care Coordination” dimension and the new “Information about Symptoms” question, which is included in the new “Cleanliness and Information about Symptoms” dimension, encompass a broader depiction of person-centered care than does the “Care Transition” dimension. The updated HCAHPS Survey measure includes the new “Care Coordination” dimension, the new “Restfulness of the Hospital Environment” dimension, and the new “Cleanliness and Information about Symptoms” dimension. In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to begin using these three new dimensions in the Hospital VBP Program beginning with the FY 2030 program year ( 89 FR 36301 through 36304 ). As noted in section IX.B.2.e(1) of this final rule, the “Care Coordination” dimension would further coordination efforts within the hospital setting and support our goals of including measures related to seamless care coordination and person-centered care. Additionally, the new “Restfulness of the Hospital Environment” dimension is comprised of three survey questions: two new questions that ask how often patients were able to get the rest they needed, and whether hospital staff helped the patient to rest and recover, and one current survey question that asks how often the area around the patient's room was quiet at night (“Quietness”).
The updated version of the HCAHPS Survey measure further modifies the current “Cleanliness and Quietness” dimension in two ways. Beginning with the FY 2030 program year, the “Quietness” question would be removed from the “Cleanliness and Quietness” dimension and would instead be included in the new “Restfulness of Hospital Environment” dimension; however, the “Quietness” question itself would remain unchanged on the updated HCAHPS Survey measure. Additionally, beginning with the FY 2030 program year, we proposed to modify the “Cleanliness and Quietness” dimension to be called the “Cleanliness and Information About Symptoms” dimension, which would include the existing “Cleanliness” question and the new “Information About Symptoms” question from the updated HCAHPS Survey measure ( 89 FR 36301 through 36304 ). The newly developed “Information About Symptoms” question asks the patient whether doctors, nurses, or other hospital staff gave the patient's family or caregiver enough information about symptoms or health problems to watch out for after the patient left the hospital.
We refer readers to section IX.B.2.b. of this final rule where we further describe the updates included in the updated HCAHPS Survey measure and to Table IX.B.2-03 for the timelines for the current and newly adopted HCAHPS Survey dimensions for the Hospital VBP Program.

We invited public comment on the proposal to adopt the updated HCAHPS Survey measure in the Hospital VBP ( print page 69511) Program beginning with the FY 2030 program year.
Comment: Several commenters supported the Hospital VBP Program linking patient satisfaction to payment incentives. The commenters also commended CMS for considering stakeholder feedback and respecting statutory requirements.
Response: We appreciate the commenters' support of the adoption of the updated HCAHPS Survey measure and linking the results to payment incentives through the Hospital VBP Program.
Comment: A commenter did not support combining “Information about Symptoms” and “Cleanliness” as a single domain because they stated that the two sub-measures are not related.
Response: While we understand the concern about combining the “Information About Symptoms” and “Cleanliness” questions into a single dimension in the Hospital VBP Program, we proposed that these two questions be combined into one dimension because separating them into two single-question dimensions would give them each more weight than the rest of the HCAHPS Survey measure questions, which are organized into multi-question dimensions (with the exception of Overall Rating). If these questions were each separate dimensions, “Cleanliness,” for example, as a single-question dimension, would receive as much weight as the “Communication with Nurses” dimension, which includes three questions. Therefore, we have determined that the combined “Cleanliness and Information about Symptoms” dimension as a two-question dimension is more comparable to the other HCAHPS Survey measure dimensions.
Comment: A few commenters recommended, with respect to the Hospital VBP Program's adoption of the updates to the HCAHPS Survey measure, that CMS complete validity testing and receive CBE endorsement before implementing the changes in the Hospital VBP Program and that CMS send the questions to a workgroup to make appropriate changes and ensure that there are not unintended consequences for the Hospital VBP Program.
Response: We appreciate the commenters' concerns, however, as described in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36299 ), we conducted validity testing on all of the updates. In addition, we intend to submit the updated HCAHPS Survey measure to the current CBE for endorsement in Fall 2025, so we anticipate the re-endorsement process would be completed well before the FY 2030 program year when the updates to the HCAHPS Survey measure are fully implemented in the Hospital VBP Program. We note that section 1886(b)(3)(B)(viii)(IX)(bb) of the Act states that in the case of a specified area or medical topic determined appropriate by the Secretary for which a feasible and practical measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. We have determined that the updates to the HCAHPS Survey measure are appropriately specified. The HCAHPS Survey measure remains endorsed, and the updated survey only modifies some of the questions and sub-measures within the survey. The HCAHPS Survey is designed to produce standardized information about patients' perspectives of care that allow objective and meaningful comparisons of hospitals on topics that are important to consumers, and these updates would improve the feedback we receive directly from patients on hospital performance. Therefore, we have determined it is appropriate to adopt these updates to the measure before the updates receive CBE endorsement.
Comment: A commenter expressed concern with the dimensions of the HCAHPS Survey measure that should and should not be included in the Hospital VBP Program methodology for future program years.
Response: We appreciate the commenter's concern; however, the dimensions of the HCAHPS Survey measure are made up of the same survey questions used in the Hospital IQR Program to ensure that hospitals only have to implement one version of the survey. We do not agree that only certain dimensions should be included in the Hospital VBP Program in future program years. We refer readers to the Hospital Inpatient VBP Program final rule in which we explain the inclusion of dimensions in the Hospital VBP Program ( 76 FR 26517 through 26520 ).
Comment: A few commenters recommended, with respect to the Hospital VBP Program's adoption of the HCAHPS Survey measure updates, ensuring that the questions are worded in a way that allows patients to accurately assess an aspect of quality and testing the impact of the “Restfulness” sub-measure before implementing these updates in the Hospital VBP Program.
Response: We thank the commenters for their recommendations, but as we discussed above, we identified the need for these updates through focus groups and cognitive interviews with patients and caregivers, discussions with technical experts, and literature reviews. Therefore, the phrasing of the questions, including that of the “Restfulness” questions, has been tested with patients and the validity of the questions has been tested at the expected average number of completed surveys per hospital.
After consideration of the public comments received, we are finalizing adoption of the updated HCAHPS Survey measure in the Hospital VBP Program beginning with the FY 2030 program year as proposed.
In the FY 2025 IPPS/LTCH PPS proposed rule, we also proposed to adopt a new scoring methodology beginning with the FY 2030 program year ( 89 FR 36304 ). For each of the nine dimensions, Achievement Points (0-10 points) and Improvement Points (0-9 points) would be calculated, the larger of which would be summed across the nine dimensions to create a pre-normalized HCAHPS Base Score of 0-90 points (as compared to 0-80 points with the current eight dimensions). The pre-normalized HCAHPS Base Score would then be multiplied by 8/9 (0.88888889) and rounded according to standard rules (values of 0.5 and higher are rounded up, values below 0.5 are rounded down) to create the normalized HCAHPS Base Score. Each of the nine dimensions would be of equal weight, so that, as currently scored, the normalized HCAHPS Base Score would range from 0 to 80 points. HCAHPS Consistency Points would then be calculated in the same manner as with the original HCAHPS Survey measure in the Hospital VBP Program and would continue to range from 0 to 20 points. Like the Base Score, the Consistency Points Score would consider scores across all nine of the Person and Community Engagement domain dimensions. The final element of the scoring formula, which would remain unchanged from the current formula in the Hospital VBP Program, would be the sum of the HCAHPS Base Score and the HCAHPS Consistency Points Score for a total score that ranges from 0 to 100 points, as before. In the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50065 ) and the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49565 ), we adopted a similar ( print page 69512) scoring methodology when the Care Transition dimension was added to the Person and Community Engagement domain in the Hospital VBP Program.
Additionally, we note that in the scoring of the current HCAHPS Survey measure in the Hospital VBP Program, the “Cleanliness and Quietness” dimension is the average of the publicly reported stand-alone “Cleanliness” and “Quietness” questions. As previously noted, the adoption of the updated HCAHPS Survey measure would result in “Quietness” being removed from this dimension and included as a question in the new “Restfulness of the Hospital Environment” dimension, and “Cleanliness” would be combined with the new “Information about Symptoms.” Therefore, “Quietness” would be scored as part of the “Restfulness of the Hospital Environment” dimension in conjunction with the other questions under that dimension. For the “Cleanliness and Information about Symptoms” dimension, we would take the average of the stand-alone “Cleanliness” and “Information about Symptoms” questions to obtain a score for the “Cleanliness and Information about Symptoms” dimension. For the purposes of the Hospital VBP Program, we proposed these two questions be combined so as not to put more weight on these questions compared to the rest of the HCAHPS Survey questions, which are included in multi-question dimensions (with the exception of Overall Rating) ( 89 FR 36304 ). If “Cleanliness,” and “Information About Symptoms,” were treated as single-question dimensions, “Cleanliness,” for example, would receive as much weight as the “Communication with Nurses” dimension, which includes three questions. Therefore, the combined “Cleanliness and Information about Symptoms” dimension is more comparable to the other HCAHPS Survey dimensions.
We invited public comment on this proposal to modify scoring of the HCAHPS Survey measure in the Hospital VBP Program beginning with the FY 2030 program year to account for the adoption of the updated HCAHPS Survey measure.
Comment: A few commenters supported the scoring changes beginning in the FY 2030 program year because they stated that the updates to the HCAHPS Survey measure would necessitate a commensurate adjustment to the scoring.
Response: We appreciate the support of the scoring modifications beginning with the FY 2030 program year in the Hospital VBP Program.
Comment: A commenter requested clarification on whether domain weights for the domains in the Hospital VBP Program would remain the same after implementation of the updated HCAHPS Survey measure.
After consideration of the public comments received, we are finalizing adoption of the modification of the scoring of the HCAHPS Survey measure in the Hospital VBP Program beginning with the FY 2030 program year as proposed.
The Hospital Readmissions Reduction Program was implemented to reduce excess readmissions effective for discharges from applicable hospitals beginning on or after October 1, 2012. The program uses six claims-based measures to track unplanned inpatient admissions within 30 days following discharge. Using the data collected from these measures, we have observed that since the inception of the program, inpatient readmission rates for the conditions and procedures included in the program have gone down. [ 437 ]
However, studies have found a concurrent increase in patients who, after being discharged from an inpatient stay, visit the emergency department (ED) or receive observation services as an outpatient. [ 438 439 440 441 442 ] As a result, we are concerned that our hospital quality reporting and value-based purchasing programs may not be adequately incentivizing hospitals to improve quality of care by accounting for more types of post-discharge events, such as a return to the ED or the receipt of observation services.
From a patient perspective, unexpectedly returning to any acute care setting, including the ED, or receiving observation services after being discharged from an inpatient hospital stay, [ 443 ] is an undesirable outcome of care. Patients who are discharged from an inpatient stay but then make an unplanned return to the hospital may incur higher healthcare costs than those who do not return to the hospital setting due to potential out-of-pocket charges for the unplanned follow-up care. Research has found that the median out-of-pocket cost of observation services received by Medicare beneficiaries as outpatients was $448.94, with low-income beneficiaries being more likely to report being concerned about costs of follow-up care, as compared to higher income beneficiaries, and limiting health care utilization that could otherwise be deemed essential in response to higher out-of-pocket costs. [ 444 ]
While these unplanned returns to the hospital impose significant burden on patients, such visits can often be avoided with greater attention to care coordination. [ 445 ] This coordination can include addressing barriers such as poor health literacy or social determinants of health that complicate a patient's ability to follow post-discharge instructions, fill prescriptions, or alert hospital staff to new symptoms. [ 446 ] For example, in ( print page 69513) one study, nurses implemented evidence-based practices for transition care, including engaging in patient education, providing clear post-discharge instructions, and following up with patients via phone calls. The study found that 9.4 percent of patients who received such interventions were readmitted 30 days after discharge, compared to an 18.8 percent readmission rate among patients not receiving such interventions. Similarly, 19.8 percent of patients receiving evidence-based transitional care were readmitted within 90 days after discharge, compared to 31.5 percent among patients in the usual care group. [ 447 ] These findings indicate that supporting patients' discharges by proactively addressing potential barriers is effective in reducing unplanned readmissions.
Therefore, we are continually seeking ways to build on current measures in several quality reporting programs that account for unplanned patient hospital visits to encourage hospitals to improve discharge processes. Current measures include three Excess Days in Acute Care (EDAC) measures currently in the Hospital Inpatient Quality Reporting (IQR) Program, which estimate days spent in acute care within 30 days post-discharge from an inpatient hospitalization for a principal diagnosis of the measure's specified condition. The acute care outcomes include ED visits, receipt of observation services, and unplanned readmissions. [ 448 ] The measures are:
- Excess Days in Acute Care (EDAC) after Hospitalization for Acute Myocardial Infarction (AMI), adopted in the FY 2016 IPPS/LTCH PPS final rule beginning with the FY 2018 payment determination ( 80 FR 49680 through 49682 );
- Excess Days in Acute Care (EDAC) after Hospitalization for Heart Failure (HF), adopted in the FY 2016 IPPS/LTCH PPS final rule beginning with the FY 2018 payment determination ( 80 FR 49682 through 49690 ); and
- Excess Days in Acute Care (EDAC) after Hospitalization for Pneumonia, adopted in the FY 2017 IPPS/LTCH PPS final rule beginning with the FY 2019 payment determination ( 81 FR 57142 through 57148 ).
Another existing measure that we use to assess unplanned hospital returns is the Hospital Visits After Hospital Outpatient Surgery measure. We adopted this measure into the Hospital Outpatient Quality Reporting (OQR) Program in the CY 2017 OPPS/ASC final rule beginning with the CY 2020 reporting period ( 81 FR 79764 through 79771 ) and the Rural Emergency Hospital Quality Reporting (REHQR) Program in the CY 2024 OPPS/ASC final rule beginning with the CY 2024 reporting period ( 88 FR 82064 through 82066 ). This measure's outcome includes any unplanned hospital visits (ED visits, receipt of observation services, or unplanned inpatient admissions) within seven days of outpatient surgery. The measure calculates facility-level measure scores based on the ratio of predicted to expected number of post-surgical hospital visits. By publicly reporting these scores, the measure encourages providers to engage in quality improvement activities to reduce unplanned follow-up visits ( 81 FR 79765 ).
While our hospital quality reporting and value-based purchasing programs currently encourage hospitals to address concerns about unplanned returns through several existing measures, we recognize that these measures, taken together, do not comprehensively capture unplanned patient returns to inpatient or outpatient care after discharge. The EDAC measures currently in the Hospital IQR Program only cover patients with a primary discharge of AMI, HF, or Pneumonia. Meanwhile, the Hospital Visits After Hospital Outpatient Surgery measure only covers patients following outpatient surgeries. Furthermore, since both the Hospital IQR and Hospital OQR Programs are quality reporting programs, a hospital's performance on these measures is not tied to payment incentives.
Therefore, we invited public comment on how these programs could further encourage hospitals to improve discharge processes, such as by introducing measures currently in quality reporting programs into value-based purchasing to link outcomes to payment incentives. We noted we were specifically interested in input on adopting measures which better represent the range of outcomes of interest to patients, including unplanned returns to the ED and receipt of observation services within 30 days of a patient's discharge from an inpatient stay.
We invited public comment on this topic. The following provides a summary of the responses we received.
Comment: Many commenters supported measuring a wider range of post-discharge patient outcomes, including ED visits and observation services. Several commenters believed that these outcomes are more relevant to patients and would encourage hospitals to enhance discharge processes to improve patient outcomes. A few commenters also stated that including data on ED visits and observation services would allow for better analysis of post-discharge care, such as determining which visits and services were preventable.
A few commenters supported having the Hospital Readmissions Reduction Program track ED visits and observation services in addition to inpatient readmissions, while others recommended only tracking post-discharge observation services but not ED visits. A commenter recommended that CMS seek to modify the statutory language of the Hospital Readmissions Reduction Program if its current statute does not allow for the measurement of ED visits and observation services. A commenter noted that if the program's statute does not allow for reporting of observation services, CMS could minimize penalties under the program and instead focus on other quality measures and programs. A few commenters recommended that CMS adopt measures that focus on post-discharge outcomes in value-based purchasing programs, to provide greater incentive for hospitals to reduce excess healthcare utilization.
Several commenters supported the EDAC measures as a better measure of preventable hospital returns than CMS' readmission measures, but requested refinements to the EDAC measures first. For example, several commenters recommended that CMS introduce more comprehensive readmissions measures that would encourage involving patients and their caregivers in discharge planning. Another commenter suggested that CMS create a broader EDAC measure that can be segmented by condition and setting, or create an inpatient and outpatient version of this EDAC measure.
Response: We appreciate the commenters' support and recommendations, including the recommendations to include measures that represent a wider range of outcomes of interest to patients, such as unplanned returns to the ED and receipt of observation services, and to adopt ( print page 69514) measures that focus on post-discharge outcomes in the value-based purchasing programs. We will take this input into account for future notice-and-comment rulemaking.
Comment: Many commenters expressed concerns with the potential unintended consequences of readmission measures for both hospitals and patients. Many commenters believed that readmission measures place the burden of patient outcomes on hospitals without appropriately accounting for factors outside of their control, such as the patient's condition severity, social determinants of health, and admissions for conditions unrelated to the initial admission. A few commenters urged CMS to ensure that measures capturing readmissions would not unfairly penalize hospitals that disproportionately serve populations with health-related social needs. Another concern from a few commenters was that including ED visits and observation services as unplanned readmissions could lead to physicians deferring timely evaluation of post-operative concerns or choosing healthier patients.
Several commenters did not support adding the EDAC measures to the Hospital Readmissions Reduction Program. Several commenters stated that the definition of readmissions in the program's statute does not include observation services or ED visits, while one comment requested that CMS clarify its authority to introduce the EDAC measures into the program. A few other commenters had concerns about adopting the EDAC measures to the Hospital Value-Based Purchasing (VBP) Program, noting that it could lead hospitals to be penalized for some of the same readmissions as in the Hospital Readmissions Reduction Program. A commenter stated that CMS does not have the statutory authority to add EDAC measures to the Hospital VBP Program. A few of the commenters suggested that CMS instead introduce readmission measures into quality reporting programs because they do not have performance-based penalties. As an alternative to quality reporting, a commenter recommended addressing unplanned hospital visits through value-based care models such as the Comprehensive Care for Joint Replacement Model or the Bundled Payments for Care Improvement Advanced Model.
Response: We thank the commenters for their feedback and share their concern for avoiding negative effects on patient care arising from measuring unplanned hospital returns. We currently use claims-based readmission, mortality, complication, and EDAC measures (there are currently three EDAC measures in the Hospital IQR Program) to track important clinical conditions. As claims-based measures, they use claims and enrollment data that are already available and do not require any additional burden for hospitals to report. We continuously reevaluate the risk models to ensure they are as valid and reliable as possible, including considering additional data to augment existing definitions of frailty in these measures. We acknowledge that hospitals do not have control over all factors influencing patients' health outcomes. However, hospitals are usually one of the most resourced healthcare entities in their communities and, as such, have important influence over the health of their patients and the communities they serve. [ 449 450 451 ] We continue to prioritize addressing health disparities through carefully considered approaches that aim to illuminate these disparities. Additionally, we acknowledge that the Hospital VBP Program cannot adopt readmission measures per section 1886(o)(2)(A) of the Act. We will continue to take public feedback into account and evaluate ways to measure patient outcomes within the statutory limits of quality reporting and value-based purchasing programs.
We are also working to harmonize measurement across settings, as noted in the Universal Foundation approach that we published in 2023. [ 452 ] This will help align priorities and accountability across healthcare settings. We also note that any future proposal to implement a new measure or program modification would be announced through notice-and-comment rulemaking.
Comment: A few commenters suggested that the Hospital Readmissions Reduction Program shorten the 30-day timeframe of its current readmission measures to around seven days after discharge. The commenters stated that 30 days is too long to accurately assess care quality because it leaves too much time for extraneous factors to impact hospital readmissions.
Several commenters offered ways for CMS to ensure better discharge care. A few commenters suggested that CMS reduce readmissions by measuring and promoting access to at home care such as the Acute Hospital Care at Home program. They cited that hospital at home results in fewer complications and improved discharge planning. To ensure more equitable outcomes, a few commenters urged CMS to promote more focus on social determinants of health at discharge, such as by updating Z code reporting. Another commenter recommended that CMS allow hospitals to report transitional care management codes for patients being discharged from the ED, so that patients can receive appropriate discharge care. A commenter emphasized the importance of patient, family, and caregiver engagement, as well as ensuring patient, family, and caregiver health literacy. To that end, the commenter requested that CMS issue guidance to hospitals on how to empower and engage these groups in the patient's care. A commenter suggested that CMS identify opportunities to provide additional financial support for hospitals to improve discharge planning, as financial constraints can pose a barrier to improvement efforts.
A few commenters suggested that CMS consider other measures to reduce readmissions. A commenter suggested that CMS keep the 30-day episode-based cost measures in the Hospital IQR Program while adopting them in the Hospital VBP Program. The commenter believed that this would reduce readmissions because a significant portion of the variation in these measures is driven by readmissions and other returns to acute care. In another suggestion, a commenter recommended that CMS adopt the National Healthcare Safety Network (NHSN) Hospital-Onset Bacteremia Fungemia Outcome measure into CMS quality reporting and value-based purchasing programs to enhance infection prevention efforts and thus reduce readmissions. Another commenter promoted the Medicare Spending Per Beneficiary measure in the Hospital VBP Program as a comprehensive evaluation of care transition efforts.
A few commenters stated that the word “readmissions” should not be used in the rule to refer interchangeably ( print page 69515) to inpatient stays, ED visits, and observation services. To support this, they cited other CMS regulations and resources which make a distinction between inpatient stays and outpatient stays, with emergency department visits and observation services being categorized as outpatient stays.
A commenter asked that CMS quality measurement include public reporting on ED boarding, that is, when patients are held in the ED while there are no inpatient beds available. The commenter believed that public reporting would hold facilities accountable for long boarding times and make consumers aware of an important aspect of hospital quality of care. Another commenter stated that if CMS chooses to measure observation services in readmission measures, CMS should also include observation services under Medicare Part A payments, or eliminate observation status to create a single hospitalization status. They also suggested counting observation stays towards the skilled nursing facility 3-day rule.
Response: We thank the commenters for their feedback regarding introducing quality measures that encourage hospitals to improve discharge processes. We also thank commenters for other suggestions on ways to reduce unplanned readmissions and improve patient post-discharge outcomes. We will take this feedback into consideration as part of future notice-and-comment rulemaking. We also refer readers to section II.C.12., where the Center for Medicare (CM) is updating their Z code payment policy regarding homelessness and housing instability.
Through the Hospital IQR Program, we strive to ensure that patients, along with their clinicians, can use information from meaningful quality measures to make better decisions about their healthcare. We support technology that reduces burden and allows clinicians to focus on providing high-quality healthcare for their patients. We also support innovative approaches to improve quality, accessibility, affordability, and equity of care while paying particular attention to improving clinicians' and beneficiaries' experiences when interacting with CMS programs. In combination with other efforts across the Department of Health and Human Services (HHS), the Hospital IQR Program incentivizes hospitals to improve healthcare quality and value, while giving patients the tools and information needed to make the best decisions for themselves.
We seek to promote higher quality, equitable, and more efficient healthcare for Medicare beneficiaries. The adoption of widely agreed upon quality and cost measures supports this effort. We work with relevant interested parties to define measures in almost every care setting and currently measure some aspects of care for almost all Medicare beneficiaries. These measures assess clinical processes and outcomes, patient safety and adverse events, patient experiences with care, care coordination, and cost of care. We have implemented quality measure reporting programs for multiple settings of care. To measure the quality of hospital inpatient services, we implemented the Hospital IQR Program. We refer readers to the following final rules for detailed discussions of the history of the Hospital IQR Program, including statutory history, and for the measures we have previously adopted for the Hospital IQR Program measure set:
- The FY 2010 IPPS/LTCH PPS final rule ( 74 FR 43860 through 43861 );
- The FY 2011 IPPS/LTCH PPS final rule ( 75 FR 50180 through 50181 );
- The FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51605 through 61653 );
- The FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53503 through 53555 );
- The FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50775 through 50837 );
- The FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50217 through 50249 );
- The FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49660 through 49692 );
- The FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57148 through 57150 );
- The FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38326 through 38328 and 82 FR 38348 );
- The FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41538 through 41609 );
- The FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42448 through 42509 );
- The FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58926 through 58959 );
- The FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45360 through 45426 );
- The FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49190 through 49310 ); and
- The FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59144 through 59203 ).
We also refer readers to 42 CFR 412.140 for Hospital IQR Program regulations.
We refer readers to 42 CFR 412.140(g)(1) for our finalized measure retention policy. We first adopted these policies in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53512 through 53513 ) and codified them in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59174 through 59175 ). Pursuant to this policy, when we adopt measures for the Hospital IQR Program beginning with a particular payment determination, we automatically readopt these measures for all subsequent payment determinations unless a different or more limited period is proposed and finalized. Measures are also retained unless we propose to remove, suspend, or replace the measures.
We did not propose any changes to these policies in the proposed rule.
We refer readers to 42 CFR 412.140(g)(2) and (3) for the Hospital IQR Program's policy regarding the factors CMS considers when removing measures from the program. We first adopted these factors in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41540 through 41544 ) and codified them in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59174 through 59175 ).
We refer readers to the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53510 through 53512 ) for a discussion of the previous considerations we have used to expand and update quality measures under the Hospital IQR Program. We also refer readers to the CMS National Quality Strategy that we launched in 2022, with the aims of promoting the highest quality outcomes and safest care for all individuals. [ 453 ]
To comply with statutory requirements that the Secretary of HHS make publicly available certain quality and efficiency measures that the Secretary is considering for adoption through rulemaking under Medicare, [ 454 ] the Consensus-Based Entity (CBE), currently Battelle, convenes the Partnership for Quality Measurement (PQM), which is comprised of clinicians, patients, measure experts, and health information technology specialists, to participate in the pre-rulemaking process and the measure endorsement process. We refer readers ( print page 69516) to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for more details on the updated pre-rulemaking measure reviews (PRMR) process, including the measure endorsement and maintenance (EM) process, for the purpose of providing multi-interested party input to the Secretary on the selection of quality and efficiency measures under consideration for use in certain Medicare quality programs, including the Hospital IQR Program.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36284 through 36293 ), we proposed to adopt seven new measures: (1) Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 payment determination; (2) Age Friendly Hospital measure beginning with the CY 2025 reporting period/FY 2027 payment; (3) Catheter-Associated Urinary Tract Infection (CAUTI) Standardized Infection Ratio Stratified for Oncology Locations measure beginning with the CY 2026 reporting period/FY 2028 payment determination; (4) Central Line-Associated Bloodstream Infection (CLABSI) Standardized Infection Ratio Stratified for Oncology Locations measure beginning with the CY 2026 reporting period/FY 2028 payment determination; (5) Hospital Harm—Falls with Injury electronic clinical quality measure (eCQM) beginning with the CY 2026 reporting period/FY 2028 payment determination; (6) Hospital Harm—Postoperative Respiratory Failure eCQM beginning with the CY 2026 reporting period/FY 2028 payment determination; and (7) Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) measure beginning with the July 1, 2023-June 30, 2025 reporting period/FY 2027 payment determination. We provide more details on these proposals in the subsequent sections of the preamble, and details on the proposed Patient Safety Structural measure are in section IX.B.1.
The U.S. population is aging rapidly, with nearly one in seven Americans at age 65 years or older in 2019. [ 455 ] In the next 10 years, one in five Americans is estimated to be over 65 years old, reaching 80.8 million by 2040. [ 456 ] As the population ages, care can become more complex, [ 457 ] with patients often developing multiple chronic conditions such as dementia, heart disease, arthritis, type 2 diabetes, and cancer. [ 458 ] These chronic conditions are among the nation's leading drivers of illness, disability, and healthcare costs. [ 459 ]
Hospitals are increasingly faced with treating older patients who have complex medical, behavioral, and psychosocial needs that are often inadequately addressed by the current healthcare infrastructure. [ 460 ] The Centers for Disease Control and Prevention (CDC), and other interested parties, have estimated that over 60 percent of Medicare beneficiaries have two or more chronic conditions. [ 461 462 ] To address the challenges of delivering care to older adults with multiple chronic conditions from a hospital and health system perspective, multiple organizations, including American College of Surgeons (ACS), the Institute for Healthcare Improvement (IHI), and the American College of Emergency Physicians, collaborated to identify and establish age-friendly initiatives based on evidence-based best practices that provide goal centered, clinically effective care for older patients. [ 463 464 ] These organizations define age-friendly care as: (1) following an essential set of evidence-based practices; (2) causing no harm; and (3) aligning with “What Matters” [ 465 ] to the older adult and their family or other caregivers. [ 466 ] Based on these age-friendly initiatives and definition, these organizations have developed a framework comprised of a set of four evidence-based elements of high-quality care to older adults, called the “4 Ms”: What Matters, Medication, Mentation, and Mobility. [ 467 ] The elements of the “4 Ms” help organize care for older adults wellness regardless of the number of chronic conditions, a person's culture, or their racial, ethnic, or religious background. [ 468 ]
The collective evidence from these age-friendly efforts demonstrates that hospitals should prioritize patient-centered care for aging patient populations with multiple chronic conditions. With CMS being the largest provider of healthcare coverage for the 65 years and older population, proposing a quality measure aimed at optimizing care for older patients, using a holistic approach to better serve the needs of this unique population, is timely. Although existing quality metrics have improved both the rate and reporting of clinical outcomes that are important to older individuals, these measures can be narrow in scope and may have limited long term effectiveness due to ceiling effects. We therefore proposed this attestation-based structural measure, the Age Friendly Hospital measure, for the Hospital IQR Program, beginning with the CY 2025 reporting period/FY 2027 payment determination in the FY 2025 IPPS/ ( print page 69517) LTCH PPS proposed rule ( 89 FR 36307 through 36314 ). This structural measure seeks to ensure that hospitals are reliably implementing the “4 M's”, and thus providing evidence-based elements of high-quality care for all older adults. [ 469 ] The elements in the Age Friendly Hospital measure align with IHI's and Hartford Foundation national initiative for Age Friendly Systems in which many hospitals already participate. [ 470 ]
In the FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 27103 through 27109 ), we solicited public comments about the potential inclusion of two geriatric care measures in the Hospital IQR Program measure set. These two potential geriatric care measures focused on ensuring hospitals were committed to implementing surgical, and general hospital best practices, for geriatric populations. Public commenters were largely in support of both geriatric care measures ( 88 FR 59185 through 59193 ) and stated that measures focused on geriatric care would help a rapidly aging population with unique characteristics find the care they need. The two potential measures, Geriatric Hospital (MUC2022-112) and Geriatric Surgical (MUC2022-032), were included in the “2022 Measures Under Consideration List” (MUC List) [ 471 ] and received significant support from the CBE, and it was recommended that the two measures be combined into one. [ 472 ] In response to CBE and public feedback, we proposed this streamlined and combined version of the former two measures ( 88 FR 59185 through 59193 ). This structural measure applies a broad scope of evidence-based best practices, focused on goal centered, clinically effective care for older patients in the hospital inpatient setting.
We note that past comments have reflected concerns regarding structural measures because they do not explicitly link to improved outcomes. This is because there is no existing validation process confirming the accuracy of hospitals' responses to these types of measures. Despite this, structural measures, over time and in select circumstances, have certain advantages over other types of measures. Structural measures provide a way to address a new topic for which no outcome measure exists, such as the Age Friendly Hospital measure, the Hospital Commitment to Health Equity measure ( 87 FR 49191 through 49201 ), and the Maternal Morbidity structural measure ( 86 FR 45361 through 45365 ). In these examples, structural measures set a new expectation for the development of evidence-based programs and processes that would support improvements in these high impact areas. In the future, these structural measures can also be linked to new outcome measures or included in the Hospital Star Ratings Program.
The Age Friendly Hospital measure assesses hospital commitment to improving care for patients 65 years or older receiving services in the hospital, operating room, or emergency department. This measure consists of five domains that address essential aspects of clinical care for older patients. Table IX.C.1 includes the five attestation domains and corresponding attestation statements.

This measure aligns with our efforts under the CMS National Quality Strategy priority area of “Equity and Engagement” that seeks to advance equity and whole-person care as well as to engage individuals and communities to become partners in their care. [ 473 ] This measure additionally aligns with the CMS National Quality Strategy priority area of “Outcomes and Alignment” that aims to improve quality and health outcomes across the care journey including the objective to improve quality in high-priority clinical areas and supportive services. [ 474 ]
The domains and attestation statements in this measure span the breadth of the clinical care pathway and, together, provide a framework for optimal care of the older adult patient. More specifically, the domains focus on patient goals, medication management, frailty, social vulnerability, and leadership/governance commitment. This structural measure identifies the best evidence-based practices for hospital leadership, operations, and high reliability across each domain, particularly with the unavailability of more direct metrics related to each of the domains. In addition, this measure complements current patient safety reporting, supports hospitals in improving the quality of care for a complex patient population, and furthers our commitment to advancing health equity among the diverse older communities served by participants in CMS programs.
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of the preamble of this final rule for details on the PRMR process including the voting procedures used to reach consensus on measure recommendations. The PRMR Hospital Committee met on January 18-19, 2024, to review measures included by the Secretary on a publicly available “2023 Measures Under Consideration List” (MUC List), [ 475 476 ] including the Age Friendly Hospital measure (MUC2023-219), and to vote on a recommendation regarding use of this measure.
The PRMR Hospital Recommendation Group for the Age Friendly Hospital measure did not reach consensus and did not recommend including this measure in the Hospital IQR Program either with or without conditions. Eleven of the sixteen members of the group recommended adopting the ( print page 69519) measure into the Hospital IQR Program without conditions; zero members recommended adoption with conditions; five committee members voted not to recommend the measure for adoption. No voting category reached 75 percent or greater, including the combination of the recommend and the recommend with conditions categories. Thus, the committee did not reach consensus and did not recommend including this measure in the Hospital IQR Program either with or without conditions.
Several PRMR Hospital Committee members applauded the intent of this measure and the push toward transparency and consistency in reporting, noting these types of measures signal to hospital leadership and governance the importance of prioritizing initiatives and implementing frameworks outlined in the measure, highlighting how important this specific measure is for prioritizing improving care for older patients. [ 477 ] PRMR Hospital Committee members also commented on the measure's flexibility regarding screening tools noting it was not overly prescriptive. [ 478 ] Several PRMR Hospital Committee members noted concerns about structural measures in general and whether they drive action. [ 479 ] Specifically, PRMR Hospital Committee members expressed concerns that the measure domains were not tightly scoped enough to drive discrete action. We acknowledge the concerns identified by the PRMR Hospital Committee members. Nevertheless, we have concluded that this measure does support reliable practices that drive change, transparent reporting, and prioritization of resources to implement these best practices. The measure was developed from a large collaborative that has evaluated the elements incorporated into these domains across many different geographic locations, hospital sizes, and patient demographics. We also refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59186 ) where we discussed previous CBE review of the Geriatric Hospital and Geriatric Surgical measures, which were combined by the measure developer based on previous CBE recommendations to create the Age Friendly Hospital measure. As previously discussed, this structural measure plays a role in establishing the foundation for health outcome quality measures, and this measure would support improvements in quality of care in hospitals participating in the Hospital IQR Program by filling gaps in care management for older adults.
The measure has not been submitted for CBE endorsement at this time. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36307 through 36314 ), we proposed adoption of this measure into the Hospital IQR Program despite the measure not yet being endorsed by the CBE. Although section 1886(b)(3)(B)(viii)(IX)(aa) of the Act requires that measures specified by the Secretary for use in the Hospital IQR Program be endorsed by the entity with a contract under section 1890(a) of the Act, section 1886(b)(3)(B)(viii)(IX)(bb) of the Act states that in the case of a specified area or medical topic determined appropriate by the Secretary for which a feasible and practical measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not so endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. During measure endorsement, the CBE considers whether a measure “is evidence-based, reliable, valid, verifiable, relevant to enhanced health outcomes, actionable at the caregiver level, feasible to collect and report, and responsive to variations in patient characteristics, such as health status, language capabilities, race or ethnicity, and income level; and is consistent across types of health care providers, including hospitals and physicians (section 1890(b)(2)(A) and (B) of the Act).”
We reviewed CBE-endorsed measures and were unable to identify any other CBE-endorsed measures on this topic. We are adopting this measure pursuant to section 1886(b)(3)(B)(viii)(IX)(bb) of the Act. As previously discussed, we have determined this an appropriate topic for a measure to be adopted absent endorsement because this measure is important for establishing a foundation for future health outcome measures and that this measure provides a framework of best practices for delivering care to older adults with multiple chronic conditions from a hospital and health system perspective.
The Age Friendly Hospital measure consists of five domains, each representing a separate domain commitment. Hospitals or health systems would need to evaluate and determine whether they can affirmatively attest to each domain, some of which have multiple attestation statements, for each hospital reported under their CMS certification number (CCN). For a hospital or a health system to affirmatively attest to a domain, and receive a point for that domain, a hospital or health systems would evaluate and determine whether it engaged in each of the elements that comprise the domain (see Table IX.C.1), for a total of five possible points (one point per domain).
A hospital or health system would not be able to receive partial points for a domain. For example, for Domain 3 (“Frailty Screening and Intervention”), a hospital or health system would evaluate and determine whether their hospital or health system's processes meet each of the corresponding attestation statements described in (A), (B), (C), and (D) (see Table IX.C.1). If the hospital or health system's processes meet all four attestation statements in Domain 3, the hospital or health system would receive a point for that domain. However, if the hospital could only affirmatively attest to (B) and (C), for example, then no points could be earned for Domain 3. We note that because the Hospital IQR Program is a pay-for-reporting program, hospitals would receive credit for the reporting of their measure results regardless of their responses to the attestation questions.
For more details on the measure specifications for the Hospital IQR Program, we refer readers to the Web-Based Data Collection tab under the Hospital IQR Program measures page on QualityNet at: https://qualitynet.cms.gov/inpatient/iqr/measures#tab1 (or other successor CMS designated websites).
Hospitals and/or health systems are required to submit information for structural measures once annually using a CMS-approved web-based data collection tool available within the Hospital Quality Reporting (HQR) System. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36307 through 36314 ) we proposed the mandatory reporting of this measure beginning with the CY 2025 reporting period/FY 2027 payment determination. We refer readers to section IX.C.9. of the preamble of this final rule for more ( print page 69520) details on our data submission and deadline requirements for structural measures. Specifications for the measure would also be posted on the QualityNet web page at: https://qualitynet.cms.gov/inpatient/iqr/measures#tab1 (or other successor CMS designated websites)
We refer readers to section IX.C.9. of this final rule for our previously finalized structural measure reporting and submission requirements.
We invited public comment on our proposal to adopt the Age Friendly Hospital measure beginning with CY 2025 reporting period/FY 2027 payment determination.
Comment: Many commenters supported our proposal to adopt the Age Friendly Hospital measure in the Hospital IQR Program. Many commenters noted this measure would address the care needed to improve outcomes for older and vulnerable patients. Many commenters expressed support on the basis that the measure is evidence-based and builds on guidelines established by several medical specialty societies focused on health care provisions for older adults. Many commenters specifically referred to the “4 M's” because the measure organizes care around what matters most to the patient, encourages review and reconciliation of medications, requires the assessment of mobility and mentation, and that these matters would benefit older adults and would result in improved outcomes and quality of care for older adults. Several commenters supported this measure and noted that it is a first step towards focusing on and measuring many aspects of geriatric care. A few commenters cited the improvements from this measure that would create a safer environment for older adults in the hospital.
Response: We thank the commenters for their support. We agree that the measure's focus on patient goals, medication management, frailty, social vulnerability, and leadership/governance commitment would encourage improvements in care provided to older adults in the inpatient hospital setting. We also agree that this measure builds on evidence-based guidelines established from multiple organizations specializing in care delivery for older adults. Lastly, we agree with commenters that this measure would be an initial effort toward quality measures related to geriatric care in the hospital inpatient setting.
Comment: A commenter supported this measure and the expected benefits of having emergency departments that focus on the care and needs of the geriatric population and recommend continuing to advance geriatric emergency care.
Response: We thank the commenter for their support. We note that any future proposal to implement measures focused on geriatric care in the emergency department would be announced through notice-and-comment rulemaking.
Comment: A commenter supported this measure and emphasized that Veterans are at a higher risk for developing dementia and suggested that this measure would encourage hospitals to treat the Veteran as a whole person.
Response: We thank the commenter for their support. We agree that this measure would encourage hospitals to treat those at risk for cognitive changes as a whole person, including Veterans.
Comment: A commenter supported the Age Friendly Hospital measure and stated that public reporting of this measure would help consumers select high-quality care.
Response: We thank the commenter for their support of this measure. We agree that this measure would support consumers' identification of high-quality care.
Comment: A few commenters recommended removing malnutrition screening under Domain 3 because it is not a standard requirement, and hospitals would need to reconsider and integrate this new element into the workflow.
Response: We disagree with the recommendation to remove malnutrition screening discussed in Domain 3. Measure developers included nutrition screening as a part of delirium prevention because studies show that the implementation of cognitive screening and delirium prevention strategies such as education, sleep hygiene, reorientation, and attention to nutrition significantly decreases the number and duration of episodes of delirium in hospitalized patients. [ 480 481 482 ] Thus, nutrition screening has significant value to providing high quality inpatient care for older populations and we will not be removing this language from the measure.
We also refer readers to sections IX.C.5.a.(1)., IX.C.5.a.(2)., and IX.C.5.a.(4). of the preamble of this final rule for more details on the guidelines and literature which informed this measure, the CBE input provided, and the significant public comment support expressed from experts, patients, and caregivers.
Comment: A few commenters did not support the Age Friendly Hospital measure because they stated that some of the practices captured by the measure are duplicative of other reporting requirements, including aspects of accreditation by The Joint Commission or parts of Medicare's Conditions of Participation (CoPs).
Response: We believe the commenters are referring to the Medicare CoP for hospitals related to quality assessment and improvement programs at 42 CFR 482.21(a) through (e) . We disagree that the Age Friendly Hospital measure is redundant to the CoPs and maintain that it is complementary to them. While the CoPs set forth minimum activities related to developing, implementing, and maintaining an effective, ongoing, hospital-wide, data-driven quality assessment and performance improvement program, the elements of the Age Friendly Hospital measure requires hospitals to attest to whether hospitals have built upon these minimum activities to exemplify optimizing care for older patients with a multifaceted vulnerability profile through implementation of geriatric vulnerability screens, evaluation of patient social determinants of health, preventing the prescription of inappropriate medications, providing goal concordant care, and implementation of a quality framework for oversight of the care pathway for older patients.
Additionally, we continually look for methods to minimize provider reporting burden and do not find that this measure is duplicative of other efforts or currently available measures in the Hospital IQR Program.
Comment: A commenter had concerns about Domain 5, Age-Friendly Care Leadership, specifically the statement requiring a designated point person or interprofessional committee. The commenter suggested that this requirement is unnecessary and redundant as all phases of hospital care have the appropriate hierarchy and structures in place to ensure high quality standards without requiring them to be specifically stated for this measure. ( print page 69521)
Response: We acknowledge the commenter's concerns regarding potentially duplicative processes and procedures that already exist in hospitals. We do not agree that the Age Friendly Hospital measure, Domain 5, is redundant and that all phases of hospital care in all hospitals have the appropriate structures in place to ensure high quality standards for older patients. Existing geriatric models of care have been shown to have multiple benefits for older hospitalized patients such as decreased rates of delirium, shorter lengths of stay, and improved function. [ 483 484 485 486 487 ] This measure has been developed to ensure that implementation of quality improvement frameworks is championed by geriatric leadership and that this wide-ranging initiative is lead in a unified manner and continually re-evaluated for opportunities for clinical improvement for older patients. Clinical programs generally cannot succeed without dedicated leadership and ongoing quality monitoring and improvement. The pillars of quality improvement have been well established in the literature, including consistent data collection with continuous feedback allowing for the identification of areas for improvement in process, structure, and outcomes. [ 488 ]
Comment: A few commenters recommended including patients 45 years or older instead of patients 65 years and older.
Response: We thank the commenters for their recommendation and will share this feedback with the measure developers. While the measure does not include patients 45 years and older at this time, we note that hospitals are not prevented from implementing the activities described in the domains of this measure and the ensuing care improvements for patients younger than 65 years.
Comment: A few commenters had concerns about the perceived lack of oversight for structural measures, including the Age Friendly Hospital measure. A commenter recommended a stronger audit function to strengthen the measure and ensure accurate reflection of what is occurring in the hospital. A commenter suggested that the measure may lack direct oversight and may not be easily audited, meaning that hospitals may attest to current practices and refuse to address domains that require significant time, effort, and resources.
Response: We understand commenters' concerns regarding the accuracy of provider self-reported data and are continuously evaluating new ways, including but not limited to an audit plan, to ensure accurate information is submitted to CMS and shared with the public. We acknowledge that past comments have reflected concerns regarding structural measures because they do not explicitly link to improved outcomes. This is because there is no existing validation process confirming the accuracy of hospitals' responses to these types of measures. Despite this, structural measures, over time and in select circumstances, have certain advantages over other types of measures. Structural measures provide a way to address a new topic for which no outcome measure exists, such as the Age Friendly Hospital measure, the Hospital Commitment to Health Equity measure ( 87 FR 49191 through 49201 ), and the Maternal Morbidity structural measure ( 86 FR 45361 through 45365 ). In these examples, structural measures set a new expectation for the development of evidence-based programs and processes that would support improvements in these high impact areas. We note that we require all hospitals participating in the Hospital IQR Program to complete the Data Accuracy and Completeness Acknowledgement (DACA) each year, which requires an annual attestation that all the information reported to CMS for this program is accurate and complete to the best of the submitters' knowledge. CMS expects all hospitals to submit complete and accurate data with respect to quality measures. We refer readers to section IX.C.11. of this final rule for more details on the DACA policies for the Hospital IQR Program.
Comment: Several commenters questioned the evidence that implementation of the measure would lead to better outcomes. A few commenters did not support this measure and recommended instead developing a geriatric-care surgical outcome-based measure that helps hospitals identify gaps in care for older adults. A commenter recommended evaluating gaps in quality and working with relevant parties to develop meaningful outcome measures for older populations. A few commenters called for an immediate adoption of this measure and encouraged CMS to move with urgency to further develop this into an outcome measure, given the vulnerability of older populations.
Response: While this measure does not measure patient outcomes or specific activities of patient care, it assesses hospitals' implementation of a systems-based approach to age friendly care best practices, which is applicable to all older patient care activities and outcomes. Age friendly care is a top priority for the rapidly aging populations across the U.S. and although existing quality metrics have improved both the rate and reporting of clinical outcomes that are important to older adults, these measures may be narrow in scope. Optimizing care for older patients with multifaceted vulnerability profiles requires a holistic approach with the goal of improving the entire care pathway to better serve the needs of this unique population. Therefore, we have identified that there is a measure gap in our current quality reporting programs. That is, we have identified a gap in measures that emphasize the importance of complex medical, physiological, and psychosocial needs that are often inadequately addressed in the hospital inpatient infrastructure. Regarding commenter recommendations to develop geriatric-care surgical outcome-based measures, the Age Friendly Hospital measure plays a role in establishing the foundation for health outcome quality measures on this topic and this measure will support improvements in quality of care in hospitals participating in the Hospital IQR Program by filling gaps in care management for older adults. We also refer readers to sections IX.C.5.a.(1)., IX.C.5.a.(2)., and IX.C.5.a.(4). of the preamble of this final rule for more details on the guidelines and literature which informed this measure, the CBE input provided, and significant public ( print page 69522) comment support expressed from experts, patients and caregivers.
We thank commenters for their support regarding the inclusion of the Age Friendly Hospital measure and who recommended implementing it sooner than proposed. While we are not finalizing adoption of this measure before the CY 2025 reporting period/FY 2027 payment determination, this does not preclude hospitals from implementing the practices described in the measure sooner if not doing so already.
Comment: A few commenters had concerns that attestations without quantitative metrics would not lead to quantifiable improvements in quality of care and patient outcomes and recommended continuing to work with interested parties on development of quantifiable metrics to inform data driven quality measures. Commenters noted that attestation measures do not have the same level of significance as outcome measures and do not provide meaningful, actionable data to be used for performance improvement, and are not specific enough to provide comparable information.
Response: We acknowledge commenter concerns regarding whether structural measures explicitly link to quantifiable improvement in quality of care and patient outcomes, however, we disagree that structural measures do not hold the same level of significance as outcome measures. We note that structural measures, in selected circumstances, over time and with experience, offer certain advantages over other types of measures. Structural measures provide a way to address a new topic for which no outcome measure exists, such as the Age Friendly Hospital measure, the Hospital Commitment to Health Equity measure ( 87 FR 49191 through 49201 ), and the Maternal Morbidity Structural measure ( 86 FR 45361 through 45365 ). In these examples, structural measures set a new expectation for the development of evidence-based programs and processes that would support improvements in these high impact areas. Specifically, regarding the Age Friendly Hospital measure, this measure assesses whether the appropriate structures are in place to ensure high quality care for older patients. In the future, these can also be linked to new outcome measures or included in the Hospital Star Ratings Program. We will continue to engage interested parties as we continue to build on our efforts to address quality of care and patient outcomes for older patients.
Comment: A few commenters had concerns or recommendations regarding the burden of implementing this measure in the timeline proposed. A few commenters had concerns about the financial and resource burden of implementing two new structural measures for CY 2025 reporting period. Another commenter recommended a transitional approach to implementing this measure by increasing the number of questions year over year. A commenter argued that the Age Friendly Hospital measure would adversely affect care delivery teams and cause additional stress through the expanded requirements for patient screenings in complex patient encounters. The commenter recommended postponing the measure's adoption until at least CY 2026. A commenter recommended a voluntary reporting period.
Response: We understand commenters' concerns. However, we do not expect all hospitals to achieve a score of five out of five on the measure in the first reporting year. This measure is intended to advance the current state of age friendly care structures within hospitals. By adopting the measure for the CY 2025 reporting period, we establish a baseline measure performance, which we can use to understand changes in care practices as hospitals seek to incorporate more of the practices outlined in the measure's requirements. Requiring attestation to two or three items per domain would not allow establishment of a baseline, nor would it as effectively encourage increased adoption of age friendly initiatives within inpatient hospitals. Regarding commenters' concerns about additional burden and the stress this may place on their hospitals, we acknowledge that reporting this measure may increase administrative burden, and we refer readers to section XII.B.6.b. for our discussion of information collection burden. We also acknowledge that hospitals may have to invest in changes to their systems in order to be able to attest “yes” to some domains. We reiterate that we do not expect all hospitals to achieve a score of five on the measure in the first year. Hospitals that are not able to attest positively to all the domains can still meet the measure reporting requirements; therefore, we are not including a voluntary reporting period for this measure. They may receive a score lower than five but would not be subject to a payment reduction so long as they attest to each domain (positively or negatively).
Comment: A few commenters made recommendations to strengthen the measure's elements regarding cognitive changes. A few commenters suggested a requirement to establish a communication plan when cognitive changes have been identified during screening for dementia or cognitive dysfunction. A commenter recommended better cognitive tests for dementia. A few commenters recommended that this measure include education and training about cognitive changes. A commenter recommended hospitals should establish programs and protocols to identify health conditions that affect older populations, such as dementia and Alzheimer's.
Response: We thank the commenters for their input and appreciate the many meaningful practices that could be utilized in hospitals across our nation and the commitment to care for those who are at risk of cognitive dysfunction, including dementia and Alzheimer's disease. While we will not be making the suggested recommendations at this time, we will share all feedback with the measure steward for the future. For this first version of the Age Friendly Hospital measure, the measure steward developed the measure to allow some flexibility in meeting the domains and not be overly prescriptive. Any future proposals to implement such a measure, or to substantively modify the measure to include these recommendations, would be announced through notice-and-comment rulemaking.
Comment: A commenter recommended that CMS consider adding language to the measure to account for the functions of health systems rather than only individual hospitals.
Response: As described in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36307 through 36311 ), we developed this measure to address the challenges of delivering care to older adults with multiple chronic conditions from both a hospital and health system perspective. Multiple organizations, including American College of Surgeons (ACS), the Institute for Healthcare Improvement (IHI), and the American College of Emergency Physicians (ACEP), collaborated to identify and establish age-friendly initiatives based on evidence-based best practices that provide goal centered, clinically effective care for older patients. 489 490 ( print page 69523) The elements in the Age Friendly Hospital measure align with IHI's and Hartford Foundation's national initiative for Age Friendly Systems which has been implemented in hospitals of various sizes, including health systems. [ 491 ] For the Hospital IQR Program, hospitals should report the measure to CMS' Hospital Quality Reporting (HQR) System based on their CMS Certification Number (CCN).
Comment: A few commenters expressed concern about Domain 4, Social Vulnerability. A commenter had concerns about hospitals that serve disadvantaged, rural, or low socioeconomic status communities may be unfairly penalized in element (b) of this Domain. The commenters noted the importance of ensuring that patients are connected to resources to address social vulnerabilities; however, hospitals treating disproportionate numbers of these patients may not have adequate access to such resources to attest “yes” to this portion of the measure, or there could be a lack of available resources in a particular community that is beyond the control of the hospital. A commenter appreciated that element (a) of Domain 4 considers screening for caregiver stress and recommended including “appropriate referrals and resources for patients' family caregivers, as applicable” to element (b) of Domain 4, to be inclusive of caregivers for all elements of Domain 4.
Response: We acknowledge commenters' concerns regarding hospitals that serve historically underserved, rural, or low socioeconomic status communities being unfairly penalized in element (b) of this Domain. We acknowledge that facilitating quality improvement for rural hospitals and small hospitals can present unique challenges and is a high priority under the CMS National Quality Strategy. [ 492 ] We continue to consider ways to support small and rural hospital efforts toward achieving health equity. We note that during development of this measure, we gave this topic significant consideration, and the intent of Domain 4 is to promote adoption of social vulnerability screening of older adults and to emphasize the importance of having systems in place to ensure that social issues are identified and addressed as a part of the care plan, not to impose undue strain on hospitals treating disproportionate numbers of patients that may not have adequate access to such resources.
We acknowledge commenter concerns about public reporting of their score on this measure through Care Compare if a hospital is unable to attest “yes” to a domain. While we recognize that a hospital's performance on the measure may impact the hospital's reputation through public reporting, this reputational impact is a means of encouraging the adoption of frameworks centered around providing high quality care to vulnerable elderly patients with multiple medical, psychological, and social needs at highest risk for adverse events after major health events requiring hospitalization. Further, we believe public reporting of healthcare quality data promotes transparency in the delivery of care by increasing the involvement of leadership in healthcare quality improvement, creating a sense of accountability, helping to focus organizational priorities, and providing a means of delivering important healthcare information to consumers. [ 493 ] Lastly, this measure is intended to provide information to hospitals on high yield points of intervention for older adults who are admitted to a hospital or an emergency department and provide a framework for the optimal care of the older adult patient ( 89 FR 36307 through 36311 ).
We note that the Hospital IQR Program is a pay-for-reporting program, and hospitals' payments are not based on their performance on quality measures. Hospitals would receive credit for successful reporting of their measure results regardless of whether they positively or negatively attest to each statement within a domain. We refer readers to section IX.C.5.a.(6). of this final rule for information on the submission and reporting requirements for this measure. We thank the commenter for their recommendation to include providing referrals to caregivers in element (b) in Domain 4 and will share all feedback with the measure steward.
Comment: A few commenters expressed concern regarding the “all or nothing” scoring methodology regarding the five domain attestations and recommended considering partial credit, noting this would enable hospitals to better understand barriers and where to prioritize efforts for quality improvement.
Response: We understand commenters' concerns that many hospitals will not be able to positively attest to all the statements for each of the domains, which will affect the hospital's score for the entire domain. However, the measure steward intentionally chose to score on a zero to five basis at the domain level instead of allowing partial credit (allowing a hospital to receive credit for each positively attested statement) to emphasize the interdependencies of the statements within each domain. Because the Age Friendly Hospital measure assesses hospitals in terms of their systemic approach to age friendly care, it is important to achieve all the actions within each domain, rather than to only meet several items. We reiterate that we do not expect all hospitals to achieve a score of five on the measure in the first reporting year. Furthermore, requiring attestation to fewer items per domain would be less effective at furthering the current state of age friendly structures within hospitals. In addition, full point scoring is also intended to keep the level of complexity to a minimum and therefore ease the general public's ability to understand the measure. We reiterate that the Hospital IQR Program is a pay-for-reporting program, and hospitals would receive credit for the reporting of their measure results regardless of their responses to the attestation questions.
Comment: A few commenters did not support this measure and had concerns regarding elements included in Domain 3. Specifically, a few commenters had concerns regarding the transfer of older patients out of the emergency room within eight hours of arrival or three hours of decision to admit as factors that are influenced beyond the control of the hospital. A few commenters questioned the wait time element used in this domain because of workforce shortages, unforeseen hastened disposition of patients, and lack of acceptable boarding time parameters recommendations.
Response: We acknowledge commenters' concerns regarding elements included in Domain 3 that would require attestation to the transfer of older patients out of the emergency room within eight hours of arrival or three hours of the decision to admit. We understand that there may be many factors, including work shortages, that are outside of the control of the hospital that can delay transfer from the emergency room, and that this may be especially true for rural hospitals. However, we remain concerned about ( print page 69524) patients being subjected to lengthy waiting periods for hospital care, and this element of Domain 3 would encourage hospitals to review their practices, especially for older patients, to ensure that patients are seen as quickly as is reasonable.
Comment: Several commenters had concerns about the limited performance gap for this measure during its testing and stated that reporting on this measure would waste a hospital's limited resources because of the limited potential for performance improvement. A commenter had concerns that this measure would quickly “top out,” leaving no room for improvement across hospitals.
Response: We disagree with the concern that this measure has limited potential for improvement across hospitals. The measure developer's initial pilot study showed significant variation in hospital performance. The majority of institutions reported completing some or all of the domains, but there was a wide range between partially and fully attesting to each element across the five domains of this measure. Rates of full compliance on domains ranged from 12.5 percent to 87.5 percent, while rates of partial compliance were higher than the rates of full compliance, accounting for the relatively low noncompliance rates. As with all Hospital IQR Program measures, we will monitor performance on the measure carefully and will consider removing the measure in the future if it becomes topped-out.
Comment: Several commenters had concerns that this measure may be prone to inconsistent interpretations and therefore be implemented and reported differently across hospitals. A few commenters were concerned that this measure may be inconsistently reported across hospitals that patients and their families may misinterpret the results of this measure when they are publicly reported. A commenter conveyed that the attestations in this measure are not specific enough to ensure that hospitals are reporting comparable information and that the measure therefore is unlikely to lead to improvement in care for this population.
Response: We acknowledge commenters' concern about public reporting of this measure and interpretation by the public. We refer readers to sections IX.C.5.a.(5).and IX.C.5.a.(6). (Measure Calculation and Data Submission and Reporting, respectively) of this final rule for detailed descriptions of how we calculate and publicly report this measure on the Compare tool hosted by HHS, currently available at: https://www.medicare.gov/care-compare . This measure includes five attestation-based questions, each representing a separate domain of commitment. Hospitals receive one point for each domain to which they attest “yes” stating they are meeting the required competencies. For each domain there are between one and four associated yes or no sub-questions for related structures or activities within the hospital. Hospitals will only receive a point for each domain if they attest “yes” to all related elements in a domain. A hospital's score can range from a total of zero to five points. For more details on the measure specifications for this measure, we refer readers to the Web-Based Data Collection tab under the Hospital IQR Program measures page on QualityNet at: https://qualitynet.cms.gov/inpatient/iqr/measures#tab1 (or other successor CMS designated websites). We intend to provide educational materials as part of our outreach and public reporting of this measure to ensure understanding and interpretation of publicly reported data. For measures that are submitted annually, we strive to have educational materials related to publicly reported data available to hospitals the following fall. For example, public reporting related educational materials for measures with a CY 2025 performance period will be available in the fall of CY 2026.
Regarding commenters concerns about the specificity of this measure and concerns about meaningful improvements, this measure has been developed to provide insightful information to healthcare providers and the public on the number of hospitals currently participating in age friendly care including social vulnerability screening for older adults, ensuring consistent quality care of older patients by ensuring hospitals have an age friendly champion or interprofessional committee, frailty screening for geriatric issues, eliciting patient health related care goals and treatment preferences, and responsible medication management. We also refer readers to sections IX.C.5.a.(1)., IX.C.5.a.(2)., and IX.C.5.a.(4). of the preamble of this final rule for more details on the guidelines and literature which informed this measure, the CBE input provided, and significant public comment support expressed from experts, patients and caregivers.
Comment: A few commenters recommended providing detailed and clear guidance on data collection and data entry to help facilitate meaningful reporting, including how hospitals should document whether they are satisfying each domain, and how it would be published for the public if finalized. A commenter requested guidance on the scope and depth of interventions required to get a point for Domain 3. A commenter requested data specifications or required elements for frailty screenings, medication management, malnutrition, and cognitive impairment, as well as clarity of scope for those requirements. A commenter expressed concern over the lack of definition of “evidence” in Domain 1.
Response: The Age Friendly Hospital measure is intended to provide hospitals flexibility in meeting each of the attestations and allow each hospital to adopt practices that are most effective for its individual circumstances. We recognize that there is significant variation among hospitals, which means that policies and procedures that are effective in some hospitals may not be effective in other hospitals. Because these practices would not be identical across hospitals the documentation supporting the practice may also vary. Therefore, the attestations in the Age Friendly Hospital measure have been developed to encourage hospitals to adopt policies and procedures consistent with a structure, culture, and leadership commitment to age friendly structures without being overly prescriptive in how hospitals implement these policies and procedures.
Regarding the request for data specifications and clarification on required elements for frailty screenings, medication management, malnutrition, and cognitive impairment, and clarity of scope for those requirements, we remind readers that the Hospital IQR Program is a pay-for-reporting program, and therefore, there are no set performance targets. We refer readers to the measure specifications at https://qualitynet.cms.gov/inpatient/iqr/ “resources” tab for more details.
We wish to clarify that we will provide educational and training materials to help with consistent implementation which will be conveyed through routine communication channels to hospitals, vendors, and QIOs, including, but not limited to, issuing memos, emails, and notices on the QualityNet website. Additionally, we will provide education and outreach materials to support hospitals in identifying additional evidence-based practices they could adopt and in documenting that they have adopted those practices. For more details on measure specifications, we also refer readers to the Web-Based Data Collection tab under the IQR Measures ( print page 69525) page on QualityNet at: https://qualitynet.cms.gov/inpatient/iqr/measures#tab2 .
Comment: A few commenters did not support this measure, noting that structural measures are burdensome to implement. A commenter stated that the benefits of adopting the measure do not sufficiently outweigh the burden of reporting it and suggested that we consider additions to the Conditions of Participation instead.
Response: We understand that there would be administrative burden with understanding each of the attestation statements and determining whether a hospital's age friendly structures are in alignment with the attestation statements. We acknowledge that reporting this measure may increase administrative burden, and we refer readers to section XII.B.6.b. of this final rule for our discussion of information collection burden. We further recognize that this administrative burden may be more significant during the first reporting year as hospitals familiarize themselves with the attestation statements. However, implementing evidence-based practices for high-quality care for older adult patients with multiple complex medical conditions is essential to avoiding unnecessary harm such as mortality and loss of function. By adopting the Age Friendly Hospital measure, we are not only assessing hospital implementation of a systems-based approach that spans the breadth of the care pathway, we are also promoting such implementation. Therefore, we expect the Age Friendly Hospital measure to have significant benefits for a large portion of the U.S. patient population. While we understand that hospital staff and leaders will have to spend time reviewing the attestations and assessing their hospital's practices in relation to the attestation statements, this measure assessment will further encourage hospitals to understand and consider implementing systems-based approaches to older patient best practices if they are not already doing so, which will in turn, improve patient care and outcomes.
Comment: Several commenters did not support this measure and recommended that this measure receive endorsement from the CBE prior to adoption into the Hospital IQR Program. A few commenters had concerns that the PRMR Hospital Committee did not reach consensus on this measure and suggested that the measure should be reviewed in more detail before its adoption.
Response: We thank commenters for their recommendation to submit this measure to the CBE for endorsement. While we recognize the value of measures undergoing CBE endorsement review, given the importance of this health topic and, as there are currently no CBE-endorsed measures that address age friendly hospital care from a hospital and healthcare system level, it is important to implement this measure as soon as possible. Although section 1886(b)(3)(B)(viii)(IX)(aa) of the Act requires that measures specified by the Secretary for use in the Hospital IQR Program be endorsed by the entity with a contract under section 1890(a) of the Act, section 1886(b)(3)(B)(viii)(IX)(bb) of the Act states that in the case of a specified area or medical topic determined appropriate by the Secretary for which a feasible and practical measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not so endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. During measure endorsement, the CBE considers whether a measure is evidence-based, reliable, valid, verifiable, relevant to enhanced health outcomes, actionable at the caregiver level, feasible to collect and report, and responsive to variations in patient characteristics, such as health status, language capabilities, race or ethnicity, and income level; and is consistent across types of health care providers, including hospitals and physicians (section 1890(b)(2)(A) and (B) of the Act).”
We acknowledge commenters concerns regarding the PRMR Hospital Committee not reaching consensus. We note that this measure has been developed to provide insightful information to healthcare providers and the public on the number of hospitals currently participating in age friendly care including social vulnerability screening for older adults, ensuring consistent quality care of older patients through ensuring hospitals have an age friendly champion or interprofessional committee, frailty screening for geriatric issues, eliciting patient health related care goals and treatment preferences, and responsible medication management. We agree that the potential for unintended consequences exists and note that we consistently monitor all the measures in the Hospital IQR Program for unintended consequences. Furthermore, we note that under our previously finalized measure removal policy, codified at 42 CFR 412.140(g)(2) and (3) ( 88 FR 59144 ), if we were to identify unintended consequences related to this measure we would consider it for removal. We also refer readers to sections IX.C.5.a.(1)., IX.C.5.a.(2)., and IX.C.5.a.(4). of the preamble of this final rule for more details on the guidelines and literature which informed this measure, the CBE input provided, and significant public comment support expressed from experts, patients and caregivers.
After consideration of the public comments we received, we are finalizing the Age Friendly Hospital measure as proposed beginning with the CY 2025 reporting period/FY 2027 payment determination.
Healthcare-associated infections (HAIs) are a major cause of illness and death in hospitals, posing a significant threat to patient safety. One in 31 hospital patients in the U.S. have an HAI at any given time, totaling about 687,000 cases per year. [ 494 ] The CDC estimated that about 72,000 patients die from HAIs per year. [ 495 ] HAIs not only put patients at risk, but also increase the hospitalization days required for patients and add considerably to healthcare costs. The CDC estimates that HAIs cost the U.S. healthcare system $28.4 billion per year. [ 496 ] Statistics on preventability vary but suggest that 55-70 percent of HAIs could be prevented through evidence-based practices including hand hygiene, cleaning ( print page 69526) surfaces with an appropriate antiseptic, and wearing gowns and gloves. [ 497 ]
Given the high risk to patient safety, we previously adopted the National Healthcare Safety Network (NHSN) Catheter-Associated Urinary Tract Infection (CAUTI) and NHSN Central Line-Associated Bloodstream Infection (CLABSI) measures in various quality reporting programs that measure the annual risk-adjusted standardized infection ratio (SIR) among adult inpatients. The measures were originally introduced in the Hospital IQR Program in the FY 2011 IPPS/LTCH PPS final rule ( 75 FR 50200 through 50202 ) and the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51617 through 51618 ). In the FY 2014 IPPS/LTCH PPS final rule, the CAUTI and CLABSI measures were then adopted in the Hospital-Acquired Condition (HAC) Reduction Program ( 78 FR 50717 ) and the Hospital Value-Based Purchasing (VBP) Program ( 78 FR 50681 through 50687 ).
Patients with cancer are especially vulnerable to developing HAIs. Chemotherapy, a common treatment for patients with cancer, can weaken patients' immune systems and leave them vulnerable to opportunistic infections. [ 498 ] Cancer treatment may also require major surgeries or invasive devices, which can act as another vector for infections. [ 499 ] It is estimated that 10.5 percent of patients undergoing major cancer surgery contract a HAI, compared to only three percent of patients undergoing elective surgeries. [ 500 ] Researchers from the same study also found that patients undergoing major cancer surgery who contracted a HAI were significantly more likely to die in the hospital than patients who did not contract a HAI. [ 501 ] In another study, researchers found that developing a HAI was linked to higher costs of care and longer lengths of stay for patients with cancers of the lip, oral cavity, and pharynx. [ 502 ] Therefore, in the FY 2013 IPPS/LTCH PPS final rule, beginning with the FY 2014 program year, we adopted the CAUTI and CLABSI measures in the PPS-Exempt Cancer Hospital Quality Reporting (PCHQR) Program ( 77 FR 53557 through 53559 ).
While many oncology services have transitioned to outpatient settings, acute care hospitals continue to specialize in the treatment of certain types of patients with cancer, for example, patients who have received a hematopoietic stem cell transplant and patients who have febrile neutropenia. [ 503 ] Based on an internal CMS analysis, in 2019 there were 321,961 Medicare beneficiaries with a primary diagnosis of cancer who received some portion of their care in an inpatient hospital setting. Within these inpatient settings, most Medicare beneficiaries with a primary diagnosis of cancer received their care at National Cancer Institute (NCI)-designated hospitals or other acute care hospitals, while only about four percent of Medicare beneficiaries received care at PPS-exempt cancer hospitals (PCHs). Additionally, based on internal CMS analysis, a portion of these Medicare beneficiaries who received care at a PCH also received at least some of their inpatient care at non-PCHs (NCI-affiliated or other hospitals).
The Biden-Harris administration's Cancer Moonshot Program has put a renewed focus on improving outcomes for patients with cancer. [ 504 ] Under this initiative, we seek to ensure that patients with cancer treated at hospitals reporting to the Hospital IQR Program are able to benefit from public reporting of hospital safety data and choose the best provider for their needs. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36311 through 36317 ), we proposed to adopt the Catheter-Associated Urinary Tract Infection (CAUTI) Standardized Infection Ratio Stratified for Oncology Locations and the Central Line-Associated Bloodstream Infection (CLABSI) Standardized Infection Ratio Stratified for Oncology Locations (hereinafter referred to as the CAUTI-Onc measure and CLABSI-Onc measure, respectively), beginning with the CY 2026 reporting period/FY 2028 payment determination. These measures would supplement, not duplicate, the existing hospital CAUTI and CLABSI measures, as the original hospital CAUTI and CLABSI measures look at hospital inpatients except for those in oncology wards, and the CAUTI-Onc and CLABSI-Onc measures look only at patients in oncology wards. Our proposals to adopt the CAUTI-Onc and CLABSI-Onc measures are part of our renewed effort to improve patient safety. We refer readers to the proposed Patient Safety Structural measure in section IX.B.1. for more information.
Urinary tract infections (UTIs) are a common type of HAI and come with many risks to patients. About 12-16 percent of adult patients in inpatient hospitals will have a urinary catheter at some point during their hospital stay, and almost all healthcare associated UTIs are introduced through instrumentation in the urinary tract. [ 505 ] Furthermore, each day the indwelling urinary catheter remains, a patient has between a three and seven percent increased risk of acquiring a catheter-associated urinary tract infection. [ 506 ] Based on data from the NHSN, the CDC reported that among the 3,780 general acute care hospitals that reported data in 2022, there were 20,237 CAUTIs in that year. [ 507 ]
CAUTIs can lead to many negative consequences for patients including cystitis, pyelonephritis, gram-negative bacteremia, endocarditis, vertebral osteomyelitis, septic arthritis, endophthalmitis, and meningitis. [ 508 ] Other consequences of CAUTIs include prolonged hospital stays, higher healthcare costs, and an increased likelihood of mortality. [ 509 ]
However, CAUTIs can often be prevented by following guidelines for ( print page 69527) urinary catheter use, insertion, and maintenance. At a large academic hospital system, a study investigated the effects of implementing a CAUTI prevention bundle in the intensive care unit (ICU). Prevention practices in this bundle included reducing unnecessary catheter use, following proper catheter maintenance, and ordering a urine culture only when warranted by a clear indication. The research team also updated the electronic health record (EHR) system to support compliance with these prevention guidelines. Researchers found that the CAUTI rates in the ICU decreased from 6.0 CAUTIs per 1,000 urinary catheter days to 0.0. The rest of the hospital then implemented the CAUTI prevention bundle, leading to a decrease in CAUTI rates from 2.0 cases per 1,000 catheter days to 0.6 cases per 1,000 catheter days. [ 510 ]
In another study, nurses at a large urban teaching hospital implemented CAUTI prevention protocols, including removing catheters from patients no longer needing them and finding alternatives to indwelling urinary catheters. As a result of this initiative, catheter days decreased by 11.8 percent and CAUTI rates declined by 38 percent. [ 511 ] More information on the prevention of CAUTIs is available at the CDC's Guideline for Prevention of Catheter-associated Urinary Tract Infections, including recommendations regarding who should receive a catheter, catheter insertion, proper insertion techniques, maintenance, quality improvement, and surveillance. [ 512 ]
To encourage the use of best practices for urinary catheters and reduce the incidence of CAUTIs, we previously adopted the CAUTI measure (CBE #0138) in several quality reporting and value-based payment programs, including the Hospital IQR, Hospital VBP, and HAC Reduction Programs ( 76 FR 51617 through 51618 , 78 FR 50681 through 50687 , and 78 FR 50717 , respectively), as discussed earlier. We adopted the measure as part of the HHS Action Plan to Prevent HAIs, as this measure was included among the prevention metrics established in the plan which is available at: https://www.hhs.gov/oidp/topics/health-care-associated-infections/hai-action-plan/index.html . Eventually, in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41547 through 41553 ), we removed the CAUTI measure from the Hospital IQR Program beginning with the CY 2019 reporting period/FY 2021 payment determination to streamline reporting through the HAC Reduction Program.
As noted earlier, the CAUTI measure used in the HAC Reduction and Hospital VBP Programs does not include inpatients in oncology wards. Because patients with cancer are especially vulnerable to developing HAIs like CAUTIs, [ 513 ] it is important to implement quality reporting for patients with cancer, as we have done in adopting the CAUTI measure in the PCHQR Program. Significant associations have been found between UTIs and post-surgery complications, longer hospitalizations, and higher hospital costs among patients with cancer, [ 514 ] and post-surgery CAUTI incidence has been found to be as high as 12.5 percent in specific cancer populations. [ 515 ] Therefore, it is important to address the needs of this high-risk population and adopt the CAUTI-Onc measure to the Hospital IQR Program. The adoption of this measure would also provide more data to compare CAUTI rates between PCHs and non-PCHs.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36312 through 36314 ), we proposed to adopt the CAUTI-Onc measure for the Hospital IQR Program beginning with the CY 2026 reporting period/FY 2028 payment determination. The purpose of this measure is to encourage the use of best practices for urinary catheters as set by the CDC and to reduce the incidence of CAUTIs for patients with cancer. To report this measure, hospitals would need to verify that all locations, including those housing oncology patients, are correctly mapped in NHSN.
Reducing CAUTI incidence through the adoption of this measure could lead to improved cancer patient outcomes, including reduced morbidity and mortality, less need for antimicrobials, and reduced patient length of stays and medical costs. [ 516 ]
The proposal to adopt the CAUTI-Onc measure supports the CMS National Quality Strategy priority area of “Safety and Resiliency.” [ 517 ] Specifically, this supports our safety goal to “achieve zero preventable harm,” and to expand the collection and use of safety indicator data across programs for key areas to improve tracking and show progress toward reducing harm. The adoption of this measure additionally supports the “Outcomes and Alignment” priority area in the CMS National Quality Strategy by collaborating with other federal agencies, namely the CDC, to promote alignment in quality measurement and close the existing reporting gap among vulnerable patients with cancer in inpatient settings. [ 518 ] This proposal to adopt the CAUTI-Onc measure not only supports two of the CMS National Quality Strategy priority areas, it also supports the Biden-Harris Administration's Cancer Moonshot program that aims to improve outcomes for patients with cancer.
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of the preamble of this final rule for details on the PRMR process, including the voting procedures used to reach consensus on measure recommendations. The PRMR Hospital Committee met on January 18-19, 2024, to review measures included by the Secretary on a publicly available “2023 Measures Under Consideration List” (MUC List), including the CAUTI-Onc measure (MUC2023-220), [ 519 520 ] and ( print page 69528) to vote on a recommendation regarding use of this measure. [ 521 ]
The PRMR Hospital Committee reached consensus and recommended including this measure in the Hospital IQR Program with conditions. Fourteen members of the group recommended adopting the measure into the Hospital IQR Program without conditions; four members recommended adoption with conditions; and one committee member voted not to recommend the measure for adoption. Taken together, 94.7 percent of the votes recommended this measure in the Hospital IQR Program with conditions. [ 522 ]
Four members of the voting committee recommended the adoption of this measure into the Hospital IQR Program with the first condition being that CMS consider expanding the reporting period. This would increase the patient volume included in the denominator and increase precision. We have reviewed this recommendation and concluded that expanding the reporting period would result in a critical loss in the ability to observe changes in the SIR over time. Obscuring any observable changes in the SIR would degrade the measure's ability to assess prevention efforts and further drive quality improvement. Therefore, we proposed this measure for adoption without the modification suggested by four committee members to preserve the measure's ability to observe changes in the SIR more quickly.
The second condition the PRMR Hospital Committee recommended for the Hospital IQR Program was that the measure should evaluate data by oncology unit type, such as hematology-oncology versus solid organ. [ 523 ] We acknowledge this condition and may consider it for future rulemaking. We proposed to adopt the CAUTI-Onc measure in the Hospital IQR Program having taken into consideration the conditions raised by the PRMR Hospital Committee.
The measure received strong support from the committee as it addresses an important patient safety concern. During the PRMR Hospital Committee's discussion, some expressed concern about the burden of manual abstraction. Others asked about the measure's validity, and whether the measure should include risk adjustments when HAIs are an issue across the board.
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for details on the EM process including the measure evaluation procedures the EM Committees use to evaluate measures and whether they meet endorsement criteria. The CAUTI measure was most recently submitted to the CBE for endorsement review in the Spring 2019 cycle (CBE #0138) and was endorsed on October 23, 2019. [ 524 ] In the submission of the CAUTI-Onc measures to the 2023 MUC list, the CDC provided additional oncology-only reliability testing based on existing data submitted to the CDC's NHSN. Because the CAUTI-Onc measure has the same specifications as the CAUTI measure, with the only difference being that it is stratified for oncology locations, additional endorsement of the oncology specific locations is not necessary. The calculations pertinent to those locations are inherently part of the endorsement performed for the CAUTI measure, and the measure (that is, numerator/denominator) is endorsed across all inpatient hospital settings, including oncology locations. The calculation of the SIR includes and accounts for the location of the patient within the facility. The CDC would incorporate information on the stratification by oncology patients during the regularly scheduled measure maintenance re-endorsement process.
For this measure, the NHSN calculates the quarterly risk-adjusted SIR of CAUTIs among inpatients at acute care hospitals who are in oncology wards. [ 525 ] The CDC then calculates the SIR using all four quarters of data from the reporting period year, which CMS uses for performance calculation and public reporting purposes. The CDC defines an oncology ward as an area for the evaluation and treatment of patients with cancer. For more details, we refer readers to the CDC Locations and Descriptions and Instructions for Mapping Patient Care Locations document. [ 526 ]
The numerator is the number of annually observed CAUTIs among acute care hospital inpatients in oncology wards. The denominator is the number of annually predicted CAUTIs among acute care hospital inpatients in oncology wards. By dividing the number of observed CAUTIs by the number of predicted CAUTIs, the SIR compares the actual number of cases to the expected number of cases. However, this does not preclude SIRs from being ranked. The SIR is calculated when there is at least one predicted CAUTI, to achieve a minimum level of precision. [ 527 ]
The measure requires a facility to have at least one predicted CAUTI before calculating the SIR because the precision of a facility's CAUTI rate can vary, especially in low volume hospitals. For this reason, the NHSN calculates the SIR instead of reporting the CAUTI rate directly. A facility's SIR is not meant to be compared directly to that of another facility. Rather, the primary role of the SIR is to compare a facility's CAUTI rate to the national rate after adjusting for facility- and patient-level risk factors. [ 528 ]
The numerator and denominator exclude the following because they are not considered indwelling catheters by NHSN definitions: suprapubic catheters, condom catheters, “in and out” catheters, and nephrostomy tubes. If a patient has either a nephrostomy tube or a suprapubic catheter and has an indwelling urinary catheter, the indwelling urinary catheter would be included in the CAUTI surveillance. [ 529 ]
The SIR also adjusts for various facility and patient-level factors that ( print page 69529) contribute to HAI risk within each facility. For more information on the risk adjustment methodology please reference the CDC website at: https://www.cdc.gov/nhsn/2022rebaseline/index.html .
We proposed to collect data for the CAUTI-Onc measure via the NHSN, consistent with the current approach for HAI reporting for the HAC Reduction and Hospital VBP Programs. The NHSN is a secure, internet-based surveillance system maintained and managed by the CDC and provided free of charge to providers. To report to the NHSN, hospitals must first agree to the NHSN Agreement to Participate and Consent form, which specifies how NHSN data would be used, including fulfilling CMS's quality measurement reporting requirements for NHSN data. [ 530 ]
Beginning in 2012, hospitals participating in the Hospital IQR Program began reporting CAUTIs in all adult, pediatric, and neonatal intensive care locations followed by reporting all adult and pediatric medical, surgical, and medical/surgical wards in 2015 using NHSN. According to a 2022 CDC report, 3,780 hospitals are reporting CAUTI data to NHSN; of these, 478 hospitals reported CAUTI data from at least one oncology location. [ 531 ] We anticipate that because most of the hospitals which would begin to report the CAUTI-Onc measure for the Hospital IQR Program are already reporting via NHSN for CAUTI in other locations as well as other measures, they have already set up an account. Hospitals currently reporting CAUTI must verify that locations housing oncology patients are correctly mapped as an oncology location based on NHSN's location mapping guidance for accurate event location attribution.
Hospitals would report their data for the CAUTI-Onc measure on a quarterly basis for the purposes of Hospital IQR Program requirements. Presently, hospitals report CAUTI data to the NHSN monthly and the SIR is calculated on a quarterly basis. Under the data submission and reporting process, hospitals would collect the numerator and denominator for the CAUTI-Onc measure each month and submit the data to the NHSN. The data from all 12 months would be calculated into quarterly reporting periods which would then be used to determine the SIR for CMS performance calculation and public reporting purposes. We refer readers to the NHSN website for further information about NHSN reporting requirements. We refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59141 ) for information on data submission and reporting requirements for our most recent updates to data submission and reporting requirements for measures submitted via the CDC NHSN.
We invited public comment on our proposal to adopt the CAUTI-Onc measure beginning with the CY 2026 reporting period/FY 2028 payment determination. We received one comment specifically on the CAUTI-Onc measure.
Comment: A commenter stated that requiring culturing to test for CAUTI in febrile oncology patients would be of questionable value to patients and lead to inappropriate culturing stewardship.
Response: We thank the commenter for sharing their feedback. However, we respectfully disagree that culturing is unnecessary for determining if oncology patients have CAUTIs or that adopting the CAUTI-Onc measure would lead to inappropriate culturing. Identifying an eligible positive urine culture is important as the first step for determining if a UTI event occurred. If a patient is febrile but the urine culture does not meet the criteria, then a UTI event did not occur according to the NHSN definition. Furthermore, facilities determine culture ordering policies, and inappropriate culturing practices should be addressed by individual facilities. [ 532 ]
We respond to comments that address both the CAUTI-Onc and CLABSI-Onc measures in the following subsection that discusses adoption of the CLABSI-Onc measure.
Central venous catheters (CVCs) are a crucial aspect of hospital care for administering medications, fluids, and nutrients to patients, as well as running medical tests. [ 533 ] However, they also carry the risk of introducing infections, referred to as central line-associated bloodstream infections (CLABSIs). [ 534 ] CLABSIs are a leading cause of HAIs and are associated with increased morbidity and mortality, prolonged hospitalization, and increased costs. [ 535 ]
According to one study, the development of bloodstream infections (BSIs) after CVC insertion was associated with longer hospital stays of on average seven additional days and a three times higher risk of death during the patient's hospital stay. [ 536 ] Additionally, a single CLABSI episode costs hospitals an estimated $48,108 on average. [ 537 ] While the CLABSI SIR has declined by 16 percent since 2015, CLABSIs still remain prevalent. [ 538 ] Based on data from the NHSN, the CDC reported that among the 3,728 general acute care hospitals that reported data in 2022, there were 23,389 CLABSIs in that year. [ 539 ]
In one study conducted on a group of academic medical centers across a three-year period, the overall CLABSI rate was 1.73 cases per 1,000 central-line days. [ 540 ] Another study, retrospectively conducted on patients with a CVC in four U.S. hospitals within the same health system, found that patients with a CVC who developed a CLABSI had a 36.6 percent higher likelihood of mortality, and 37 percent higher chance of being readmitted compared to patients who did not develop a CLABSI. The study also found that the average hospital length of stay in patients who developed a CLABSI increased by two ( print page 69530) days when compared to patients without a CLABSI. [ 541 ]
Following evidence-based guidelines when inserting and maintaining central lines can help prevent the occurrence of CLABSIs. [ 542 ] Proper central line insertion practices include applying skin antiseptic, ensuring proper hand hygiene, using sterile barrier precautions, and ensuring the skin preparation agent has dried completely before insertion. [ 543 ] One study of 30 long-term acute care hospitals found that adoption of a catheter maintenance bundle led to the CLABSI rate decreasing by 29 percent. [ 544 ] In another study, researchers implemented the standard CDC bundle along with additional measures in a large acute care hospital. As a result, the CLABSI rate decreased by 68 percent from 2013 to 2017. [ 545 ] Despite a large body of evidence indicating that adopting a central line bundle decreases CLABSI rates, adoption of these best practices remains inconsistent. A systematic review of the available literature on hospital adherence to the CDC's central line bundle checklist found that none of the medical facilities in the studies followed all elements of the bundle, and compliance rates remained low in follow-up studies. [ 546 ] For more information on the standard CDC bundle, we refer readers to the Guidelines for the Prevention of Intravascular Catheter-Related Infections. [ 547 ]
To encourage adherence to best practices for central line use and to reduce the incidence of CLABSIs, we previously adopted the CLABSI measure (CBE #0139) in several quality reporting and value-based payment programs, including the Hospital IQR, Hospital VBP, and HAC Reduction Programs ( 75 FR 50200 through 50202 , 78 FR 50681 through 50687 , and 78 FR 50717 , respectively), as discussed earlier. We adopted the measure as part of the HHS Action Plan to Prevent HAIs, as this measure was included among the prevention metrics established in the plan which is available at: https://www.hhs.gov/oidp/topics/health-care-associated-infections/hai-action-plan/index.html . In the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41547 through 41553 ), we removed the CLABSI measure from the Hospital IQR Program beginning with the CY 2019 reporting period/FY 2021 payment determination to streamline reporting through the HAC Reduction Program.
Currently, the CLABSI measure used in the HAC Reduction and Hospital VBP Programs does not include inpatients in oncology wards. Because patients with cancer are especially vulnerable to developing HAIs like CLABSIs, [ 548 ] it is important to implement quality reporting for patients with cancer, as we have done in adopting the CLABSI measure in the PCHQR Program. While central lines are a crucial component of cancer treatment, they are also associated with at least 400,000 bloodstream infections in oncology patients every year in the U.S. [ 549 ] CLABSIs in patients with cancer may lead to sepsis, require interruptions in chemotherapy, and increase the hospital length of stay. [ 550 ] CLABSIs among patients with cancer also incur a high economic burden, costing the U.S. healthcare system over $18 billion annually. [ 551 ] Therefore, it is important to address the needs of this high-risk population and adopt the CLABSI-Onc measure to the Hospital IQR Program. The adoption of this measure would also provide more data to compare CLABSI rates between PCHs and non-PCHs.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36314 through 36317 ), we proposed to adopt the CLABSI-Onc measure to the Hospital IQR Program beginning with the CY 2026 reporting period/FY 2028 payment determination. The purpose of this measure is to promote CLABSI prevention activities and reduce the incidence of CLABSIs for patients with cancer. Unlike the version of the measure previously in the Hospital IQR Program and that is currently in the HAC Reduction and Hospital VBP Programs, this version we proposed to adopt is limited to inpatients at acute care hospitals in oncology wards. To report this measure, hospitals would need to verify that all locations, including those housing oncology patients, are correctly in NHSN.
Reducing the CLABSI incidence through the adoption of this measure could lead to improved cancer patient outcomes, including reduced morbidity and mortality, less need for antimicrobials, and reduced patient length of stays and medical costs. [ 552 ]
The proposal to adopt the CLABSI-Onc measure supports the CMS National Quality Strategy priority area of “Safety and Resiliency.” Specifically, this supports our safety goal to “achieve zero preventable harm,” and to expand the collection and use of safety indicator data across programs for key areas to improve tracking and show progress toward reducing harm. The adoption of this measure additionally supports the “Outcomes and Alignment” priority area in the CMS National Quality Strategy by collaborating with other federal agencies, namely the CDC, to promote alignment in quality measurement and close the existing reporting gap among vulnerable patients ( print page 69531) with cancer in inpatient settings. [ 553 ] This proposal to adopt CLABSI-Onc not only supports two of the CMS National Quality Strategy priority areas, it also supports the Biden-Harris Administration's Cancer Moonshot program that aims to improve outcomes for patients with cancer.
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for details on the PRMR process including the voting procedures the PRMR process uses to reach consensus on measure recommendations. The PRMR Hospital Committee met on January 18-19, 2024, to review measures included by the Secretary on a publicly available “2023 Measures Under Consideration List” (MUC List), including the CLABSI-Onc measure (MUC2023-219), [ 554 555 ] and to vote on a recommendation regarding use of this measure. [ 556 ]
The committee reached consensus and recommended including this measure in the Hospital IQR Program with conditions. Fourteen members of the group recommended adopting the measure into the Hospital IQR Program without conditions; four members recommended adoption with conditions; and one committee member voted not to recommend the measure for adoption. Taken together, 94.7 percent of the votes recommended the measure. [ 557 ]
Four members of the voting committee recommended the adoption of this measure into the Hospital IQR Program, with the first condition being that CMS consider expanding the reporting period. This would increase the patient volume included in the denominator and increase precision. We have reviewed this recommendation and concluded that expanding the reporting period would result in a critical loss in the ability to observe changes in the SIR over time. Obscuring any observable changes in the SIR would degrade the measure's ability to assess prevention efforts and further drive quality improvement. Therefore, we proposed this measure for adoption without the modification suggested by four committee members to preserve the measure's ability to observe changes in the SIR more quickly.
The second condition the committee recommended for the Hospital IQR Program was that the measure should evaluate data by oncology unit type, such as hematology-oncology versus solid organ. [ 558 ] We acknowledge this condition and may consider it for future rulemaking. We proposed to adopt the CLABSI-Onc measure in the Hospital IQR Program having taken into consideration the conditions raised by the PRMR Hospital Recommendation Committee.
The measure received strong support from the committee as it addresses an important patient safety concern. During the committee's discussion, some expressed concern about the burden of manual abstraction. Others asked about the measure's validity, and whether the measure should include risk adjustments when HAIs are an issue across the board.
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for details on the EM process including the measure evaluation procedures the EM Committees use to evaluate measures and whether they meet endorsement criteria. The CLABSI measure was most recently submitted to the CBE for endorsement review in the Spring 2019 cycle (CBE #0139) and was endorsed on October 23, 2019. [ 559 ] In the submission of the CLABSI-Onc measure to the 2023 MUC list, the CDC provided additional oncology-only reliability testing based on existing data submitted to the CDC's NHSN. Because the CLABSI-Onc measure has the same specifications as the CLABSI measure, with the only difference being that it is stratified for oncology locations, additional endorsement of CLABSI-Onc is not necessary. The calculations pertinent to those locations are inherently part of the endorsement performed for the CLABSI measure, and the measure (that is, numerator/denominator) is endorsed across all inpatient hospital settings, including oncology locations. The calculation of the SIR includes and accounts for the location of the patient within the facility. The CDC would incorporate information on the stratification by oncology patients during the regularly scheduled measure maintenance re-endorsement process.
For this measure, the NHSN calculates the quarterly risk-adjusted SIR of CLABSIs among inpatients at acute care hospitals who are in oncology wards. [ 560 ] The CDC then calculates the SIR using all four quarters of data from the reporting period year, which CMS uses for performance calculation and public reporting purposes. The CDC defines an oncology ward as an area for the evaluation and treatment of patients with cancer. For more details, we refer readers to the CDC Locations and Descriptions and Instructions for Mapping Patient Care Locations document. [ 561 ]
The numerator is the number of annually observed CLABSIs among acute care hospital inpatients in oncology wards. The denominator is the number of annually predicted CLABSIs among acute care hospital inpatients in oncology wards. By dividing the number of observed CLABSIs by the number of predicted CLABSIs, the SIR compares the actual number of cases to the expected number of cases. However, this does not preclude SIRs from being ranked. The SIR is calculated when there is at least one predicted CLABSI, to achieve a minimum level of precision. [ 562 ]
The measure requires a facility to have at least one predicted CLABSI before calculating the SIR because the precision of a facility's CLABSI rate can ( print page 69532) vary, especially in low volume hospitals. For this reason, the NHSN calculates the SIR instead of reporting the CLABSI rate directly. A facility's SIR is not meant to be compared directly to that of another facility. Rather, the primary role of the SIR is to compare a facility's CLABSI rate to the national rate after adjusting for facility- and patient-level risk factors. [ 563 ]
The numerator and denominator exclude the following devices because they are not considered central lines: arterial catheters (unless in the pulmonary artery, aorta or umbilical artery), arteriovenous fistula, arteriovenous graft, atrial catheters (also known as transthoracic intra-cardiac catheters, those catheters inserted directly into the right or left atrium via the heart wall), extracorporeal membrane oxygenation (ECMO), hemodialysis reliable outflow (HERO) dialysis catheter, intra-aortic balloon pump (IABP) devices, peripheral IV or midlines, or ventricular assist devices (VAD). Additionally, CLABSI events reported to the NHSN as mucosal barrier injury laboratory-confirmed bloodstream infections (MBI-LCBIs) are excluded from the SIR. [ 564 ]
The SIR also adjusts for various facility and patient-level factors that contribute to HAI risk within each facility. For more information on the risk adjustment methodology please reference the CDC website at: https://www.cdc.gov/nhsn/2022rebaseline/index.html .
We proposed to collect data for the CLABSI-Onc measure via the NHSN, consistent with the current approach for HAI reporting for the HAC Reduction and Hospital VBP Programs. The NHSN is a secure, internet-based surveillance system maintained and managed by the CDC and provided free of charge to providers. To report to the NHSN, hospitals must first agree to the NHSN Agreement to Participate and Consent form, which specifies how NHSN data would be used, including fulfilling CMS's quality measurement reporting requirements for NHSN data. [ 565 ]
Starting in 2011, facilities operating under the Hospital IQR Program began reporting CLABSIs in all adult, pediatric, and neonatal intensive care locations followed by reporting all adult and pediatric medical, surgical, and medical/surgical wards in 2015 using NHSN. According to a 2022 CDC report, 3,728 hospitals are reporting CLABSI data to NHSN; of these, 488 hospitals reported data from at least one oncology location. [ 566 ] We anticipate that because most of the hospitals which would begin to report the CLABSI-Onc measure for the Hospital IQR Program are already reporting via NHSN for other measures, they have already set up an account. Hospitals currently reporting CLABSI must verify that locations housing oncology patients are correctly mapped as an oncology location based on NHSN's location mapping guidance for accurate event location attribution.
Hospitals would report their data for the CLABSI-Onc measure on a quarterly basis for the purposes of Hospital IQR Program requirements. Presently, hospitals report CLABSI data to the NHSN monthly and the SIR is calculated on a quarterly basis. Under the data submission and reporting process, hospitals would collect the numerator and denominator for the CLABSI-Onc measure each month and submit the data to the NHSN. The data from all 12 months would be calculated into quarterly reporting periods which would then be used to determine the SIR for CMS performance calculation and public reporting purposes. We refer readers to the NHSN website for further information about NHSN reporting requirements. We refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59141 ) for information on data submission and reporting requirements for our most recent updates to data submission and reporting requirements for measures submitted via the CDC NHSN.
We invited public comment on our proposal to adopt the CLABSI-Onc measure beginning with the CY 2026 reporting period/FY 2028 payment determination. We received two comments specifically on the CLABSI-Onc measure. Because many commenters discussed the two proposed oncology-specific measures (CAUTI-Onc and CLABSI-Onc) together, we summarize their comments and provide our responses after our discussion of CLABSI-specific public comments.
Comment: A commenter supported CMS's proposal to adopt the CLABSI-Onc measure, noting that many patients suffer from CLABSIs each year, that the mortality rate is high, and that CLABSIs extract a high cost on the healthcare system. The commenter stated that the measure would reduce CLABSI incidence and lead to improved outcomes for patients with cancer. The commenter also recommended that CMS adopt other measures designed to improve quality of care for patients requiring CVCs.
Response: We thank the commenters for their feedback. We agree that the CLABSI-Onc measure can help prevent CLABSIs and improve outcomes for patients with cancer. We will also take the feedback to adopt additional measures into account for future rulemaking.
Comment: A commenter recommended that CMS exclude Mucosal Barrier Injury (MBI) confirmed bloodstream infections from the measure definition.
Response: We thank the commenter for sharing this recommendation. We wish to clarify that the CLABSI-Onc measure does exclude events that meet the definition of an MBI. This exclusion is applied regardless of patient care location or setting. [ 567 ]
In addition to a few commenters who specifically discussed either the CAUTI-Onc or CLABSI-Onc measure, many commenters discussed these two proposed oncology-specific measures together. We summarize their comments below and provide our responses.
Comment: Many commenters supported adoption of the CAUTI-Onc and CLABSI-Onc measures because of their potential to improve care quality by promoting prevention practices and reducing CAUTI and CLABSI incidence. Commenters also stated that these measures would fill a gap in quality reporting for patients with cancer, who are particularly vulnerable to developing HAIs.
Response: We thank the commenters for their feedback. We agree that the CAUTI-Onc and CLABSI-Onc measures can improve care quality for patients with cancer and would allow more of them to be covered under quality reporting.
Comment: Several commenters supported the measure but had feedback or questions regarding the methodology of the measures. A few commenters asked CMS to conduct an analysis prior to public reporting to ensure equitable comparisons across hospitals. A commenter requested more information ( print page 69533) on how SIRs would be calculated at the unit level.
Response: We thank the commenters for their feedback and provide additional information in response to their concerns. The CDC developed a risk adjustment methodology for these measures to ensure equitable comparisons across hospitals. To calculate risk adjustments, the NHSN relies on facility survey data and designation of patient care locations to serve as high-level surrogate markers for patient acuity. While using patient-level data may better characterize the patient population, the NHSN uses uniformly reported data on patient care locations to determine an appropriate risk adjustment without significantly increasing hospitals' data collection burden.
Regarding the commenter's question about how SIRs would be calculated, the SIR is a scalable, risk-adjusted metric. In CAUTI and CLABSI SIRs, risk adjustment is applied at the individual location level, resulting in a count of infection events (SIR numerator) and predicted number of infections (SIR denominator). In CAUTI and CLABSI SIRs, risk adjustment is applied at the individual location level, resulting in a count of infection events (SIR numerator) and predicted number of infections (SIR denominator). The NHSN then aggregates location-specific results for all of a facility's locations prior to calculating the SIR.
Comment: A commenter requested clarification on the quarterly submission process and the impact of a “not applicable” response for hospitals.
Response: We thank the commenter for their questions. Since the NHSN does not have an option to submit a “not applicable” response when reporting these measures, hospitals that do not have oncology wards do not have a place to specify that within NHSN. Therefore, these location types are left blank within the system. In this case, the NHSN is not able to calculate a SIR and the hospital's data would not be publicly reported. Hospitals may indicate that they do not have any locations mapped as an oncology ward on the Measure Exception form, as applicable. If a hospital indicates this on the form, the hospital need not report zero cases to NHSN, but completion of the Measure Exception form is required to avoid a penalty through a reduction in a hospital's annual payment update (APU) for failing to report the measure. For more information about the submission process, we refer readers to the CDC's operational guidance for reporting CAUTI and CLABSI data, available at: https://www.cdc.gov/nhsn/cms/ach.html .
Comment: A few commenters expressed concern that the smaller denominators for these measures could lead to low volume bias and asked CMS to ensure statistical reliability and validity.
Response: We wish to clarify that the calculated SIR for the CAUTI-Onc and CLABSI-Onc measures are adjusted for volume. The SIR compares the actual number of cases to the predicted number of cases. The SIR is calculated when there is at least one predicted infection event, to achieve a minimum level of precision. The measures require a facility to have at least one predicted event before calculating the SIR because the precision of a facility's infection rate can vary, especially in low volume hospitals. For this reason, the NHSN calculates the SIR instead of reporting the rate directly. Using the SIRs, we determine and publicly report each hospital's national percentile ranking. While a direct comparison between hospitals' infection rates would be subject to low volume bias, the SIRs account for this bias by comparing the observed to the expected rate.
Comment: A few commenters did not support the measures because they stated that the risk adjustment does not adequately reflect the immunosuppression of patients with cancer. Another commenter objected to the risk adjustment methodology for these measures, stating that the difficulty in preventing HAIs for certain patients does not justify adjustments that hide these difficult cases.
Response: We thank the commenters for sharing their concerns. However, we disagree that the measures do not provide adequate risk adjustment to account for the immunosuppression of patients with cancer. To calculate risk adjustments, the NHSN relies on facility survey data and designation of patient care locations to serve as high-level surrogate markers for patient acuity. Risk adjustment relies on patient location to account for risk factors such as immunosuppression in patients with cancer. It is important for the measures to stratify by location to account for patient risk factors which are not within a hospital's control and ensure that measure results represent each hospital's performance. We will continue to collaborate with the CDC to review and refine this measure, including the risk adjustment model, as part of our measure maintenance and evaluation.
Comment: Several commenters offered suggestions for ensuring the effectiveness of the measures in promoting patient safety and transparent reporting. A few commenters requested that CMS continue to verify the reliability and validity of the measures. A few commenters urged CMS to monitor for unintended consequences, including using publicly reported SIR information to make inappropriate comparisons between facilities. A commenter suggested that CMS provide more background information about airborne infections and preventive practices when discussing these measures.
Response: We appreciate this input and will take it into account. We also note that the measures have been tested and were recommended for inclusion in the Hospital IQR Program by the PRMR Hospital Committee. [ 568 ] Additionally, we will continue to monitor the CAUTI-Onc and CLABSI-Onc measures in the Hospital IQR Program to ensure that the publicly reported data are helpful to consumers when choosing the best hospital for their needs.
Comment: A few commenters expressed concerns about the burden of measure reporting, stating that these measures would impose workloads on hospital staff that outweigh the reporting benefits. One commenter recommended that CMS provide facilities with sufficient time to prepare for collecting data on the CAUTI-Onc and CLABSI-Onc measures.
A commenter stated that the CAUTI-Onc measure should be combined with the CAUTI measure, and the CLABSI-Onc measure should be combined with the CLABSI measure. According to the commenter, the oncology-specific measures are duplicative and dividing reporting between oncology wards and other locations creates additional burden for hospitals.
Response: We appreciate commenters sharing their concerns about the measure reporting burden. We carefully consider the benefit of adopting new measures with attention to the burden on hospitals. We do not agree that the burden of measure reporting for these measures outweighs the reporting benefits. While the measures would require staff time to report, we are adopting the measures because of their ability to encourage the use of best practices and thus reduce the incidence of HAIs for patients with cancer. With measure reporting beginning with the CY 2026 reporting period/FY 2028 payment determination, hospitals should have sufficient time to prepare ( print page 69534) for reporting. We note that hospitals are already reporting the CAUTI and CLABSI measures, which have the same specifications as the CAUTI-Onc and CLABSI-Onc measures, the only difference being the locations at which the measures stratify. In 2022, 3,780 hospitals reported CAUTI events [ 569 ] and 3,728 reported CLABSI events. [ 570 ] In addition, 488 hospitals have already reported data from at least one oncology location. [ 571 ]
We further note that since the NHSN system currently collects CAUTI-Onc and CLABSI-Onc data, hospitals that have not been reporting such data could begin to do so now in preparation for when the data would be reported to CMS for the Hospital IQR Program beginning with the CY 2026 reporting period.
With regard to the suggestion to combine the oncology-specific measures with the CAUTI and CLABSI measures, we considered this in the development of the proposed rule; however, in collaboration with the CDC, we proposed the CAUTI-Onc and CLABSI-Onc measures as separate measures from the CAUTI and CLABSI measures in the Hospital VBP Program and HAC Reduction Program because patients with cancer are especially vulnerable to HAIs and this can greatly affect measure results. We therefore proposed oncology-specific measures so that they can provide a stratified risk-adjusted measure and report data in the context of the patient population. This would also allow more direct comparison with the CAUTI-Onc and CLABSI-Onc measures used in the PCHQR Program.
Comment: Several commenters expressed concern about the CAUTI-Onc and CLABSI-Onc measures on the basis that many hospitals do not have defined oncology wards and thus would not be able to report the measures. A few commenters requested further clarification on how oncology wards would be defined by NHSN. Another commenter stated that defining oncology locations according to the 80 percent rule as listed by the CDC Locations and Descriptions and Instructions for Mapping Patient Care Locations [ 572 ] would exclude some oncology patients from public reporting. A few commenters asked if the measures would distinguish between oncology ward characteristics, such as whether they serve pediatric or adult patients.
Response: We thank the commenters for their input and questions about identifying oncology wards. The CAUTI-Onc and CLABSI-Onc measures allow for the reporting of HAIs for patients with cancer in hospitals that are not PCHs, and the NHSN has developed a location mapping methodology for these measures that allows the model to consider the hospital locations that have higher incidences of HAIs. As currently established by the CDC for reporting to the NHSN, the CLABSI-Onc and CAUTI-Onc measures are collected under a location-based surveillance method, using the CDC's location definitions. Patients with cancer receiving care in other non-oncology inpatient locations would not be captured by the oncology-specific measures. CDC location codes are determined by the type of patient that makes up 80 percent or more of the location's population. This 80 percent rule is standard across all healthcare settings to define hospital locations and does not single out any specific location type. Per the CDC's location definitions, hospitals can report CLABSI and CAUTI data for pediatric oncology locations separately from adult oncology locations. [ 573 ]
After consideration of the public comments we received, we are finalizing the CAUTI-Onc and CLABSI-Onc measures as proposed beginning with the CY 2026 reporting period/FY 2028 payment determination.
Patient falls are among the most common hospital harms reported and can increase length of stay and patient costs. [ 574 575 576 ] It has been estimated that there are 700,000-1,000,000 inpatient falls in the U.S. annually, with more than one-third resulting in injury and up to 11,000 resulting in patient death. [ 577 578 ] Protocols and prevention measures to reduce patient falls with injury include using fall risk assessment tools to gauge individual patient risk, implementing fall prevention protocols directed at individual patient risk factors, and implementing environmental rounds to assess and correct environmental fall hazards. [ 579 ] There is wide variation in fall rates between hospitals which suggests that this is an area where quality measurement and further improvement is still needed. [ 580 581 582 583 ]
Currently there are no electronic clinical quality measures (eCQMs) that focus specifically on acute care inpatient falls with major or moderate injury in any of the hospital quality reporting or value-based purchasing programs. The Patient Safety Indicator (PSI) 90 composite measure, [ 584 ] which is currently included in the HAC ( print page 69535) Reduction Program, does include a fall related component, (PSI 08): In Hospital Fall-Associated Fracture Rate; however, it is a claims-based measure that uses a two-year performance period, it is focused on the Medicare fee-for-service (FFS) population, and the numerator is limited to fractures and does not include other fall-associated major and moderate injuries. In the FY 2022 IPPS/LTCH PPS final rule, we highlighted our commitment to developing new digital quality measures that assess various aspects of patient safety in the inpatient setting ( 87 FR 49181 through 49190 ). As discussed later in this section of the preamble, the Hospital Harm—Falls with Injury eCQM provides the opportunity to assess the rate of falls that result in a wider range of injuries, in a much larger patient population, and using more timely information from patients' electronic medical records instead of administrative claims data.
The Hospital Harm—Falls with Injury measure is a risk-adjusted outcome eCQM. The denominator is inpatient hospitalizations for patients aged 18 and older with a length of stay less than or equal to 120 days that ends during the measurement period. The numerator is inpatient hospitalizations where the patient has a fall that results in moderate injury (such as lacerations, open wounds, dislocations, sprains, and strains) or major injury (such as fractures, closed head injuries, internal bleeding). The diagnosis of a fall and of a moderate or major injury that was present on admission would be excluded from the measure.
The baseline risk-adjustment model accounts for age and several risk factors present on admission (weight loss or malnutrition, delirium, dementia, and other neurological disorders). [ 585 ] The risk-adjustment model has been developed to ensure that hospitals that care for sicker and more complex patients are evaluated fairly. [ 586 ] We refer readers to the eCQI Resource Center ( https://ecqi.healthit.gov/eh-cah ) for more details on the measure specifications and risk methodology.
This measure aligns with several goals under the CMS National Quality Strategy in addition to supporting our re-commitment to better patient and healthcare worker safety. [ 587 ] The COVID-19 public health emergency (PHE) put significant strain on hospitals and health systems which negatively impacted patient safety in routine care delivery, highlighting the need to address gaps in safety. Adopting the Hospital Harm—Falls with Injury measure is one of several initial actions we are taking in response to the President's Council of Advisors on Science and Technology (PCAST) call to action to renew “our nation's commitment to improving patient safety.” [ 588 ] By establishing additional safety indicators, such as this measure, we are building a stronger, more resilient U.S. healthcare system. We refer readers to section IX.B.1. for more details on other efforts toward better patient and healthcare workers safety practices and the proposed Patient Safety Structural measure into the Hospital IQR Program and the PCHQR Program.
This measure aligns with the “Safety and Resiliency” goal of our CMS National Quality Strategy to achieve zero preventable harm, the “Equity and Engagement” goal to ensure that all individuals have the information needed to make the best choices and complements the HHS National Action Alliance to Advance Patient Safety. By providing hospitals with the opportunity to assess the rate of falls with injury in a much larger patient population (all-payer) compared to current measures such as PSI 08 (limited to Medicare FFS), this measure expands the available safety indicator data within CMS programs and promotes equitable care for all. This measure additionally supports the “Outcomes and Alignment” goals to improve quality and health outcomes by providing hospitals a mechanism to track falls with injury event rates and improve falls intervention efforts over time, a key patient safety metric across the care journey. Third, this measure supports CMS' Interoperability goal to improve quality measure efficiency by transitioning to digital measures in CMS quality reporting programs. As an eCQM, this measure increases the digital measure footprint and can also serve as a potential replacement for the claims-based PSI 08 measure (reported within the PSI 90 composite) in the future.
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of the preamble of this final rule for details on the PRMR process including the voting procedures used to reach consensus on measure recommendations. The PRMR Hospital Committee met on January 18-19, 2024, to review measures included by the Secretary on a publicly available “2023 Measures Under Consideration List” (MUC List), including the Hospital Harm—Falls with Injury measure (MUC2023-048), and to vote on a recommendation regarding use of this measure. [ 589 590 ]
The committee reached consensus and recommended including this measure in the Hospital IQR Program with conditions. Twelve members of the group voted to adopt the measure into the Hospital IQR Program without conditions; six members voted to adopt with conditions; one committee member voted not to recommend the measure for adoption. Taken together, 94.7 percent of the votes were recommended or recommended with conditions. The six members who voted to adopt with conditions specified the condition as monitoring unintended consequences, such as use of patient restraints. We agree that the potential for unintended consequences exists and note that we consistently monitor all the measures in the Hospital IQR Program for unintended consequences. Furthermore, we note that under our previously finalized measure removal policy, codified at 42 CFR 412.140(g)(2) and (3) ( 88 FR 59144 ), if we were to identify unintended consequences related to this measure we would consider it for removal. Furthermore, we note that various programs have been instituted that reduce hospital falls without decreasing mobility (such as the Hospital Elder Life Program) [ 591 ] and that ( print page 69536) the benefits of promoting mobility outweigh any increase in fall risk. [ 592 ]
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for details on the EM process including the measure evaluation procedures the EM Committees uses to evaluate measures and whether they meet endorsement criteria. The EM Management of Acute Events, Chronic Disease, Surgery, and Behavioral Health Committee [ 593 ] convened in the Fall 2023 cycle to review the Hospital Harm—Falls with Injury measure (CBE #4120e) submitted to the CBE for endorsement. The EM Management of Acute Events, Chronic Disease, Surgery, and Behavioral Health Committee ultimately voted to endorse the measure on January 29, 2024. [ 594 ]
This ratio measure is reported as the number of inpatient hospitalizations with falls with moderate or major injury per 1,000 patient days. The measure is calculated using the following: (Total number of encounters with falls with moderate or major injury/total number of eligible hospital days) × 1,000. To calculate the numerator (that is, the total number of encounters with falls with moderate or major injury): (1) identify the initial population (inpatient hospitalizations for patients aged 18 and older with a length of stay less than or equal to 120 days that ends during the measurement period), (2) remove exclusions (patients who had a fall diagnosis present at the time the order for inpatient admission occurs), and (3) determine if the patient meets numerator criteria (patient has both a fall diagnosis and major or moderate injury diagnosis not present on admission). Hospital days are measured in 24-hour periods starting from the time of arrival at the hospital (including time in the emergency department and or observation). The number of hospital days is rounded down to whole numbers; any fractional periods are dropped. All data elements necessary to calculate the numerator and denominator are defined within value sets available in the Value Set Authority Center (VSAC). [ 595 ]
The measure was tested in 12 hospital test sites with two different EHR vendors (Epic and Allscripts) with varying bed size, geographic location, and teaching status. Risk-adjusted rates showed substantial variation in performance scores across the 12 test hospitals indicating ample room for quality improvement. [ 596 ] Test results using one year of data indicated strong measure reliability and validity (including agreement between data exported from the EHR and data in the patient chart). [ 597 ] As PSI 08 uses a two-year performance period, this eCQM would allow hospitals to receive more timely information about measure performance.
We recognize there may be concern regarding measure duplication with PSI 08 (a component of PSI 90 that is currently measured and publicly reported in the HAC Reduction Program). However, as described earlier, the Hospital Harm—Falls with Injury eCQM assesses the rate of falls with a wider range of injuries in a larger population compared to PSI 08. We envision the potential future use of patient safety eCQMs not only in the Hospital IQR Program, but also pay-for-performance programs such as the HAC Reduction Program, including as a potential replacement for the claims-based PSI 90 measure. However, until that time we are retaining PSI 08 (within the PSI 90 composite) in the HAC Reduction Program as well as include the Hospital Harm—Falls with Injury eCQM in the Hospital IQR Program.
This eCQM uses data collected through hospitals' EHRs. The measure is designed to be calculated by the hospitals' certified electronic health record technology (CEHRT) using patient-level data and then submitted by hospitals to CMS. As with all quality measures we develop, testing was performed to confirm the feasibility of the measure, data elements, and validity of the numerator, using clinical adjudicators who validated the EHR data compared with medical chart-abstracted data. Testing demonstrated that all critical data elements were reliably and consistently captured in hospital EHRs, and measure implementation is feasible.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36306 through 36341 ), we proposed the adoption of the Hospital Harm—Falls with Injury eCQM as part of the eCQM measure set beginning with the CY 2026 reporting period/FY 2028 payment determination. The eCQM measure set is the measure set from which hospitals can self-select measures to report to meet the eCQM reporting requirement. We refer readers to section IX.C.9.c. of this final rule for a discussion of our previously finalized eCQM reporting and submission requirements, as well as proposed modifications to these requirements. Additionally, we refer readers to section IX.F.6.a.(2). of the preamble of this final rule for a discussion of a similar measure adoption in the Medicare Promoting Interoperability Program.
We invited public comment on our proposal to adopt the Hospital Harm—Falls with Injury eCQM beginning with the CY 2026 reporting period/FY 2028 payment determination.
Comment: Many commenters supported adopting the measure. Specifically, commenters stated that the measure represents an important measurement area that targets one of the most common hospital harms, promotes patient safety, captures a broader population than PSI 08, and would allow a timelier delivery of information than claims data. A commenter stated that the measure can be used to identify gaps in care, optimize care delivery, and improve patient outcomes. Another commenter stated that the measure would raise awareness of fall rates and lower healthcare costs. A commenter supported our consideration of malnutrition in the risk adjustment. Another commenter stated that the implementation timeline is consistent with the amount of time the industry needs to implement the measure.
Comment: A commenter supported our proposal to adopt the Hospital Harm—Falls with Injury eCQM beginning with the CY 2026 reporting period. The commenter stated that malnutrition is a risk factor for severe clinical events and suggested that some of its effects, such as loss of lean body mass, can contribute to frailty and possible falls. ( print page 69537)
Response: We thank the commenter for their support for including the measure's risk-adjustment model.
Comment: A commenter supported the adoption of the Hospital Harm—Falls with Injury eCQM but encouraged CMS to consider the workflow and education impact of the proposed volume of new measures and to align adoption and attestation timelines accordingly. Another commenter had concerns with the proposals to adopt new eCQMs and recommended adopting only one new eCQM per reporting period due to the burdensome process for building, tracking, and implementing new eCQMs.
Response: We carefully consider the benefit of adopting new measures and transitioning to eCQMs with attention to the burden on hospitals. The program's progressive shift toward digital measures would ultimately decrease the burden for hospitals because eCQMs use electronic standards, which help reduce the burden of manual abstraction and reporting for measured entities. We additionally note that hospitals would initially have the option to self-select whether to report this measure, which provides sufficient flexibility for those hospitals that may need more time to implement this measure before being able to report it.
Comment: A commenter supported the addition of the Hospital Harm—Falls with Injury eCQM in the Hospital IQR Program but cautioned CMS against moving this measure into pay-for-performance programs such as the Hospital VBP Program. The commenter stated that there is a limited evidence base for best practices on fall prevention within the hospital.
Response: We note that we proposed adopting the Hospital Harm—Falls with Injury eCQM in the Hospital IQR and Medicare Promoting Interoperability Programs. We have not proposed to adopt this measure in a pay-for-performance program. If we decide to use this measure for additional CMS programs, such as the Hospital VBP Program, we would do so through notice-and-comment rulemaking.
Comment: A commenter supported adoption of the Hospital Harm—Falls with Injury eCQM into the Hospital IQR Program as an eCQM that hospitals can select to meet the eCQM reporting requirements but encouraged CMS to work with technical experts to improve risk standardization under the measure. The commenter stated that fall risk varies considerably based on the patient's diagnoses, procedures and other aspects of care (for example, medications) and stated that the measure could be enhanced using indirect standardization (as the method of risk adjustment), whereby the expected value is conditioned on MS-DRG and potentially other patient- and visit-level factors.
Response: We note that the measure, as proposed, is risk-adjusted for several factors including medications active on admission, medications administered during the hospitalization, diagnoses present on admission which may increase the risk for a fall with injury, and physical traits, such as body mass index. Additional information regarding the measure specifications and risk methodology is available at the eCQI Resource Center ( https://ecqi.healthit.gov/eh-cah ). We will monitor measure performance and consider any future adjustments to the measure, including adjustments to the measure's risk-adjustment methodology.
Comment: A commenter supported the implementation of the Hospital Harm—Falls with Injury eCQM, stating that falls are an important patient safety issue, that the measure is risk-adjusted so that outcome rates can be compared across hospitals, and that pilot testing revealed that hospitals were able to map key elements for reporting without changes to current workflow. However, the commenter recommended that the 120-day length of stay exclusion be removed in alignment with most eCQMs where this exclusion has already been removed.
Response: We thank the commenter for their support. We note that several eCQMs such as Venous Thromboembolism Prophylaxis, Safe Use of Opioids—Concurrent Prescribing, and Discharged on Antithrombotic Therapy eCQMs specify a length of stay less than or equal to 120 days; therefore, this measure is in alignment with those eCQMs.
Comment: A few commenters supported the Hospital Harm—Falls with Injury eCQM, as an outcome measure of patient falls while hospitalized, which is an important measure area, but did not support the fact that the measure excludes patients admitted due to a fall diagnosis from the measure calculation. Those commenters were concerned that the measure excluded the patients most vulnerable to falls and stated that a hospital should be able to document and separate when a fall happened prior to admission and while hospitalized. The commenters also stated that these falls are preventable. Another commenter stated that a more reasonable approach to categorizing these events would be to stratify the measure by unit, allowing comparison within ICU, medical surgical, or other specific units where patients are of similar risks.
Response: We thank the commenters for this feedback. As currently specified, the Hospital Harm—Falls with Injury eCQM excludes patients from the numerator and the denominator who have a fall diagnosis which is present on admission. Therefore, a patient who had a fall present on admission and then had a subsequent fall during the inpatient hospitalization would be excluded from both the numerator and denominator. Patients who have a fall present on admission are excluded from the denominator population because the measure focuses on falls with injury that occurred during hospitalization. This measure exclusion is necessary to reduce the measure's false positive rate and to prevent hospitals from being penalized by including falls that occurred prior to the encounter, when injuries resulting from these falls may be diagnosed later in a hospital stay.
For a patient safety measure that may be used to compare hospital performance, it is important to aim for some level of case mix uniformity across hospitals and therefore stratifying by unit may not be practical, but we will consider whether such stratification is appropriate in future measure updates. Although the measure already uses risk adjustment, including a highly heterogenous group of patients already admitted for a fall may exceed the ability of the risk adjustment model to provide a level playing field and complicate the use of the measure for comparing hospital performance. However, we recognize the importance of assessing fall risk for all hospitalized patients, including those patients with a fall present on admission. We also refer readers to the discussion of the Domain 3: Frailty Screening and Intervention domain of the finalized Age Friendly Hospital measure where hospitals will be required to attest to whether they screen patients for risks regarding mobility and whether data is collected on the rate of falls. We will consider removing the exclusion of patients admitted due to a fall diagnosis from the Hospital Harm—Falls with Injury eCQM in future updates to the measure.
Comment: Several commenters raised concerns regarding unintended consequences of the measure. A few commenters supported the measure but encouraged CMS to monitor results carefully to ensure the measure does not result in unintended consequences associated with immobilization, such as pressure injuries or patient restraints. A commenter did not support the measure due to concerns that the measure could inadvertently discourage early patient ( print page 69538) mobilization, which the commenter stated is vital for recovery.
Response: We agree that the potential for unintended consequences exists and note that we consistently monitor all the measures in the Hospital IQR Program for unintended consequences. We note that various programs have been instituted that reduce hospital falls without decreasing mobility (such as the Hospital Elder Life Program). [ 598 ] We also note that the Hospital IQR Program adopted a Hospital Harm—Pressure Injury eCQM in the FY 2024 IPPS/LTCH PPS final rule as one of the eCQMs hospitals have the option to self-select for reporting beginning with the CY 2025 reporting period/FY 2027 payment determination ( 88 FR 59149 ), which will also encourage early patient mobilization.
Comment: A few commenters supported adding the measure to the list of available eCQMs that hospitals can choose to self-select to report but urged CMS not to require its reporting until questions about variations in the capture of data by EHR vendors can be answered.
Response: As finalized in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49299 through 49302 ), hospitals must report on six total eCQMs beginning with the CY 2024 reporting period and subsequent years. Hospitals must report on the following three eCQMs: (1) Hospital Harm—Severe Hypoglycemia eCQM; (2) Hospital Harm—Severe Hyperglycemia eCQM; and (3) Hospital Harm—Opioid-Related Adverse Events eCQM. Hospitals must also report three additional eCQMs that are self-selected from the list of remaining eCQMs. We proposed this measure would be included as one of the eCQMs hospitals have the option to self-select for reporting beginning with the CY 2026 reporting period/FY 2028 payment determination. We note that in section IX.C.9.c., we proposed to increase progressively the number of eCQMs a hospital must report beginning with the CY 2026 reporting period/FY 2028 payment determination, however the Hospital Harm—Falls with Injury eCQM would remain one of the eCQMs hospitals can self-select to report. Future changes to the eCQM reporting requirements, including any additional eCQMs for mandatory reporting, would go through notice-and-comment rulemaking.
Comment: A few commenters discussed differences in how falls are documented by hospitals and expressed concerns that the documentation does not lend itself to eCQMs. Some commenters stated that falls are not necessarily captured as discrete data points in the hospital EHR, but may be documented in narrative notes, and to capture falls they would need a discrete field in the EHR. The commenters stated that clinicians may be using structured fields differently to input data and documentation may not be captured in a standardized manner. The commenter stated this could lead to measure performance being more dependent on the sensitivity of the screening technologies and approaches used than on underlying performance. Another commenter stated that details about patient falls, and resultant injuries are often captured more reliably in a hospital's safety event reporting database than in a patient's medical record. Further, commenters noted that sometimes when a fall happens, it is not immediately known if there is an injury, or the extent of the injury and this information would need to be manually entered into the record after getting test or imaging results. A commenter raised a concern that hospitals with better, more complete, data would be punished because their results would compare unfavorably against hospitals that failed to accurately capture falls with injury.
A commenter stated that information related to a fall prior to arrival could be recorded in the EHR and could be pulled into the numerator falsely by key words if the process is not set up correctly and staff are not educated to enter the information correctly. Another commenter encouraged CMS to assess the feasibility of collecting the required data elements from EHRs and determine if the measure is reliable and valid across a broader set of EHR vendors and hospitals. The commenter stated that assessing measure performance using only two vendor systems and 12 hospitals is insufficient.
Response: Data element feasibility was assessed during testing, where all 13 hospital sites that participated in the evaluation of feasibility confirmed that the data elements used in the proposed measure can be captured within the electronic health record in a structured and codified manner either using nationally accepted terminology standards or local system codes that could be mapped. [ 599 ] While one hospital did not always use its structured fields to capture a fall that occurred during hospitalization, the three other sites using an EHR from the same vendor did not encounter the same workflow challenges. The remaining 12 hospitals proceeded with validity testing that evaluated electronic clinical data compared to manually extracted data. The results indicate that the positive predictive value for the numerator was 98.77 percent, meaning that in 98.77 percent of the cases where the patient was identified as experiencing the harm using EHR data, the result was confirmed using the manually extracted data. [ 600 ] These results generally indicate that most hospitals would have the data available to calculate the measure accurately in structured fields in their EHR and to provide complete data for measurement. Resources such as the QRDA implementation guide that can assist with eCQM implementation, including ensuring that data is captured in a consistent format, can be found on the eCQI Resource Center. [ 601 ]
Comment: A commenter raised concerns about the measure definitions. The commenter acknowledged that the published measure definitions include examples of injuries categorized as moderate or major but questioned whether the definitions would be applied universally by all reporting hospitals. Another commenter stated that standardization of national operational definition of a fall, moderate injury, and severe injury must occur before adopting the measure into the Hospital IQR Program.
Response: The measure specifications include definitions for the terms fall, moderate injury, and major injury. A fall is defined as: A sudden, unintentional descent, with or without injury to the patient, that results in the patient coming to rest on the floor, on or against some other surface (for example, a counter), on another person, or on an object (for example, a trash can). A fall with moderate or major injury is defined as: A fall and a diagnosis of moderate or major injury during the inpatient hospitalization. Examples of moderate injuries include lacerations, open wounds, dislocations, sprains, and muscle strains. Examples of major injuries include fractures, closed head injuries, and internal bleeding. These definitions are available on the eCQI Resource Center at: https:// ( print page 69539) ecqi.healthit.gov/ecqm/eh/2026/cms1017v1?qt-tabs_measure=measure-information .
Comment: Several commenters raised concerns that the Hospital Harm—Falls with Injury eCQM overlaps with the PSI 08 component of the PSI 90 measure in the HAC Reduction Program. These commenters asked CMS to consider the potential burden of overlapping eCQMs and claims-based measures that could give differing results and require duplicative reporting. A commenter stated that we should consider removing PSI 90 in the future when replacement eCQMs have been implemented or that we could allow hospitals to choose whether to report a claims-based or electronic measure. A commenter added that if the Hospital Harm—Falls with Injury eCQM is included, CMS should outline a clear timeline for elimination of duplicative PSI measures. A commenter recommended CMS not finalize the Hospital Harm—Falls with Injury eCQM and instead retain PSI 08 within the HAC Reduction Program.
Response: As we discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36319 ) and in this final rule, the Hospital Harm—Falls with Injury eCQM would assess the rate of falls with a wider range of injuries in a larger population compared to PSI 08 and uses more timely information from patients' electronic medical records instead of administrative claims data. In addition to these two measurement improvements, the development and implementation of the Hospital Harm—Falls with Injury eCQM aligns with CMS's commitment to moving to digital quality measurements as highlighted in the FY 2022 IPPS/LTCH PPS final rule ( 87 FR 49181 through 49190 ). We envision the potential future use of patient safety eCQMs not only in the Hospital IQR Program, but also pay-for-performance programs such as the HAC Reduction Program, including as a potential replacement for the claims-based PSI 90 measure. However, until that time, we are retaining PSI 08 (within the PSI 90 composite) in the HAC Reduction Program while hospitals gain experience with the Hospital Harm—Falls with Injury eCQM in the Hospital IQR Program.
Comment: A few commenters stated that the performance scores ranged from 0.0 to 0.258 across 12 hospitals and questioned whether this measure demonstrates a sufficient performance gap to support its use in the Hospital IQR Program. The commenters recommended that CMS continue to test this measure across a broad range of hospitals and vendor systems to determine the extent to which there is sufficient variation in performance scores to warrant the measure's use in the Hospital IQR Program.
Response: The data from 12 hospitals demonstrates wide performance variation, suggesting room for improvement. Specifically, the median risk-adjusted measure rate was 0.053 falls per 1,000 encounter days, ranging from 0 falls to 0.257 falls per 1000 encounter days. The sample data show that the average number of encounter days per year per hospital is about 100,000. Using that assumption, the mean risk-adjusted number of falls per year across the 12 hospitals was approximately 8, with a range of 0 to 26, underscoring the measure's underlying performance gap and variation. In other words, the worst performing hospital has a performance rate that is more than three times higher than the sample average. Additionally, there may be disparities in the rate of in-hospital falls based on certain factors. For example, according to a report from the Leapfrog Group, the rate of in-hospital falls with hip fracture is significantly higher for patients insured by Medicare and Medicaid than for privately insured patients. [ 602 ] This analysis also found the rate of in-hospital falls with hip fracture is also significantly lower for Non-Hispanic Black and Hispanic patients than for White patients. [ 603 ] During the measure testing, patient and caregiver representatives agreed that the rate of hospital-acquired falls resulting in major or moderate injury is important to measure and can help improve care for patients. [ 604 ] During an additional TEP meeting, one member additionally stressed that the measure has importance from a patient safety standpoint. [ 605 ]
The measure was tested in 12 hospitals, which represented a diversity of hospital characteristics, including teaching status, size, EHR vendor, and geographic location. In terms of teaching status, three hospitals were major teaching hospitals and nine were community teaching hospitals. In terms of size, three hospitals had between 100-199 beds, seven hospitals had between 200-499 beds, and two hospitals had 499 beds. In terms of EHR systems, two different EHR vendors were used in the hospitals. Finally, in terms of geographic locations, hospitals were headquartered in the Southeast, Northeast, and Western parts of the United States.
Comment: A commenter requested that CMS share more details about the Hospital Harm—Falls with Injury eCQM, noting that previous eCQM implementations have had issues with their logic and code sets. The commenter was concerned that the measure's codes may not be robust enough to capture falls and recommended that CMS and the measure steward incorporate more flexibility for rapid cycle improvements into the measure.
Response: We thank the commenter for their feedback. We refer readers to the eCQI Resource Center ( https://ecqi.healthit.gov/ ) as well as the PQM's site ( https://p4qm.org/measures/4120e ) for more details on the Hospital Harm—Falls with Injury eCQM specifications, including the logic and value sets used in the specifications. We also reiterate that this eCQM underwent testing which demonstrated that all critical data elements were reliably and consistently captured in patient EHRs, and measure implementation is feasible. We also note that CMS has a process to receive feedback on issues with measure implementation, including a ticketing process, and we will consider how we can incorporate more flexibility for faster measure updates. [ 606 ]
Comment: A commenter did not support the adoption of the measure and suggested that CMS consider a measure that rewards an increase in patient mobility as opposed to a measure that captures falls because encouraging improved mobility is a better approach than tracking higher rates of patient falls.
Response: We thank the commenter for their suggestion. The measure would promote patient safety and encourage hospitals to reduce patient falls through promoting patient mobility. We also note that the Hospital IQR Program adopted a Hospital Harm—Pressure Injury eCQM in the FY 2024 IPPS/LTCH PPS final rule as one of the eCQMs hospitals have the option to self-select for reporting beginning with the CY ( print page 69540) 2025 reporting period/FY 2027 payment determination ( 88 FR 59149 ) which will promote increased patient mobility. We will monitor the measure and will take into consideration whether a measure focusing specifically on increasing patient mobility should be considered in future rulemaking.
Comment: A commenter expressed concerns regarding the expansion of eCQMs in hospital and ambulatory quality reporting programs. The commenter stated that the introduction of new eCQMs is shortsighted, burdensome, and fails to recognize the effort to shift toward digital quality measures (dQMs). The commenter stated that new eCQMs should not be required as part of quality reporting or pay-for-performance programs but that rather, CMS should invest efforts towards the future development of dQMs.
Response: We agree with the commenter in prioritizing investment in dQMs for quality measurement in order to improve accuracy, improve the timeliness of the information, and to reduce reporting burden. In general, CMS considers eCQMs to be a subset of dQMs. [ 607 ] The definition of dQMs is: “Quality measures that use standardized, digital data from one or more sources of health information that are captured and exchanged via interoperable systems; apply quality measure specifications that are standards-based and use code packages; and are computable in an integrated environment without additional effort.” [ 608 ] CMS has developed a dQM Strategic Roadmap to outline the activities required to transition to digital quality measurement. [ 609 ]
After consideration of the public comments we received, we are finalizing the measure as proposed beginning with the CY 2026 reporting period/FY 2028 payment determination. We refer readers to section IX.F.6.a.(2). of the preamble of this final rule for a discussion of the adoption of this measure in the Medicare Promoting Interoperability Program. We also refer readers to section XXXX of the preamble of this final rule where we discuss the use of this measure in the Transforming Episode Accountability Model (TEAM).
Postoperative respiratory failure is defined as unplanned intubation or prolonged mechanical ventilation (MV) after an operation. [ 610 ] It is considered to be the most serious of the postoperative respiratory complications because it represents the “end stage” of several types of pulmonary complications (for example, pneumonia, aspiration, pulmonary edema, and acute respiratory distress syndrome) and non-pulmonary problems (for example, sepsis, oversedation, seizures, stroke, heart failure, pulmonary embolism, and fluid overload), and it often results in negative outcomes, including prolonged morbidity, longer hospital stays, increased readmissions, higher costs, or death. [ 611 612 613 ] Postoperative respiratory failure is potentially preventable with optimal care, such as carefully managing intraoperative ventilator use and fluids, reducing surgical duration, using regional anesthesia, and preventing wound infection and pain. [ 614 615 616 ] Published data suggest room for improvement; a Nationwide Inpatient Sample (NIS) database study of over 500,000 hospitalizations involving a brain tumor between 2002 and 2010 found the incidence of postoperative respiratory failure varied by hospital characteristics, with higher reported rates of postoperative respiratory failure in nonteaching hospitals than teaching hospitals, and incidence increased with hospital bed size. [ 617 ]
Currently there are no eCQMs that focus specifically on postoperative respiratory failure in the inpatient setting in any of the hospital quality reporting or value-based purchasing programs. The PSI 90 composite measure, [ 618 ] which is currently included in the HAC Reduction Program, does include a postoperative respiratory failure related component, PSI 11: Postoperative Respiratory Failure Rate; however, it is a claims-based measure that uses a two-year performance period, it is focused on the Medicare FFS population, and is dependent upon ICD-10-CM codes. In the FY 2022 IPPS/LTCH PPS final rule, we highlighted our commitment to developing new digital quality measures that assess various aspects of patient safety in the inpatient setting ( 87 FR 49181 through 49190 ). The Hospital Harm—Postoperative Respiratory Failure eCQM provides the opportunity to assess the rate of postoperative respiratory failure in a much larger patient population and use more timely information from patients' electronic medical records instead of administrative claims data.
The Hospital Harm—Postoperative Respiratory Failure measure is a risk-adjusted outcome eCQM. The denominator is elective inpatient hospitalizations that end during the measurement period for patients 18 years old and older without an obstetrical condition and at least one surgical procedure was performed within the first three days of the encounter. The numerator is elective inpatient hospitalizations for patients with postoperative respiratory failure: For more detail on how postoperative respiratory failure is determined we refer readers to the measure specifications at the eCQI Resource ( print page 69541) Center ( https://ecqi.healthit.gov/eh-cah ).
The baseline risk-adjustment model accounts for ten comorbidities present on admission (weight loss, deficiency anemias, heart failure, diabetes with chronic complications, moderate to severe liver disease, peripheral vascular disease, pulmonary circulation disease, valvular disease, American Society of Anesthesiologists categories 3 through 5) and lab values for oxygen (partial pressure), leukocytes, albumin, blood urea nitrogen, bilirubin, and pH of arterial blood. [ 619 ] The risk-adjustment ensures that hospitals that care for sicker and more complex patients are evaluated fairly. [ 620 ] We refer readers to the eCQI Resource Center ( https://ecqi.healthit.gov/eh-cah ) for more details on the measure specifications and risk-adjustment methodology.
This measure aligns with several goals under the CMS National Quality Strategy in addition to supporting our re-commitment to better patient and healthcare worker safety. [ 621 ] The COVID-19 public health emergency (PHE) highlighted the need to address gaps in safety by putting significant strain on hospitals and health systems which, in turn, negatively impacted patient safety. Adopting the Hospital Harm—Postoperative Respiratory Failure measure is one of several initial actions we are taking in response to the President's Council of Advisors on Science and Technology (PCAST), call to action to renew “our nation's commitment to improving patient safety.” [ 622 ] By establishing additional safety indicators, such as this measure, we are building a stronger, more resilient U.S. healthcare system. We refer readers to section IX.B.1. for more details on other efforts toward better patient and healthcare workers safety practices and the proposed Patient Safety Structural measure into the Hospital IQR Program and the PCHQR Program.
In alignment with the CMS National Quality Strategy [ 623 ] this measure supports the “Safety and Resiliency” goal to achieve zero preventable harm, the “Equity and Engagement” goal to ensure that all individuals have the information needed to make the best choices and complements the HHS National Action Alliance to Advance Patient Safety. By providing hospitals the opportunity to assess postoperative respiratory failure rates in a much larger patient population (all-payer) compared to current measures such as PSI 11 (limited to Medicare FFS), this measure expands the available safety indicator data within CMS programs and promotes equitable care for all. Second, this measure supports the “Outcomes and Alignment” goals to improve quality and health outcomes by providing hospitals a mechanism to track their postoperative respiratory failure incidents and improve harm reduction efforts over time, a key patient safety metric across the care journey. Third, this measure supports CMS' Interoperability goal to improve quality measure efficiency by transitioning to digital measures in CMS quality reporting programs. As an eCQM, this measure increases the digital measure footprint and can also serve as a potential replacement for the claims-based PSI 11 measure (reported within the PSI-90 composite) in the future.
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for details on the PRMR process including the voting used to reach consensus on measure recommendations. The PRMR Hospital Committee met on January 18-19, 2024, to review measures included by the Secretary on a publicly available “2023 Measures Under Consideration List” (MUC List), [ 624 625 ] including the Hospital Harm—Postoperative Respiratory Failure measure (MUC2023-050), and to vote on a recommendation for rulemaking for the Hospital IQR Program.
The committee reached consensus and recommended including this measure in the Hospital IQR Program with conditions. Twelve members of the group voted to adopt the measure into the Hospital IQR Program without conditions; five members voted to adopt with conditions; two committee members voted not to recommend the measure for adoption. Taken together, 89.5 percent of the votes were between recommend and recommend with conditions. The five members who voted to adopt with conditions specified the condition as monitoring unintended consequences, such as avoidance of life-saving procedures with higher risk for respiratory failure. We agree that the potential for unintended consequences exists and note that we consistently monitor all the measures in the Hospital IQR Program for unintended consequences. Furthermore, we note that under our previously finalized measure removal policy, codified at 42 CFR 412.140(g)(2) and (3) ( 88 FR 59144 ), if we were to identify unintended consequences related to this measure, we would consider it for removal. Furthermore, the measure logic allows for the use of mechanical ventilation or intubation or extubation documentation outside of a procedural area to trigger a postoperative respiratory event, thus expanding opportunities for electronic capture of information and accommodating varying clinical documentation workflows.
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for details on the EM process including the measure evaluation procedures the EM Committees uses to evaluate measures and whether they meet endorsement criteria. The EM Management of Acute and Chronic Events Committee convened in the Fall 2023 cycle to review the Hospital Harm—Postoperative Respiratory Failure measure (CBE #4130e) submitted to the CBE for endorsement. [ 626 ] The EM Management of Acute and Chronic Events Committee ultimately voted to endorse the measure on January 29, 2024. [ 627 ]
Postoperative respiratory failure is evaluated using MV documentation, intubation or extubation documentation to determine if an unplanned initiation of MV occurred or if MV was continued without interruption after a procedure.
The following calculation is applied to report the overall performance rate: [Number of encounters in numerator/(Number of encounters in denominator—Number of encounters in denominator exclusions)] × 1,000. All data elements necessary to calculate the numerator and denominator are defined within value sets available in the VSAC. [ 628 ]
The measure was tested in 12 hospitals (test sites) with two different EHR vendors (Epic and Cerner) with varying bed size, geographic location, and teaching status. Risk-adjusted rates showed substantial variation in performance scores across the 12 test hospitals. [ 629 ] Test results indicated high measure reliability and validity (including agreement between data exported from the EHR and data in the patient chart). [ 630 ]
This eCQM uses data collected through hospitals' EHRs. The measure is designed to be calculated by the hospitals' CEHRT using patient-level data and then submitted by hospitals to CMS. As with all quality measures we develop, testing was performed to confirm the feasibility of the measure, data elements, and validity of the numerator, using clinical adjudicators who validated the EHR data compared with medical chart-abstracted data. Testing demonstrated that all critical data elements were reliably and consistently captured in patient EHRs, and measure implementation is feasible.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36306 through 36341 ), we proposed the adoption of the Hospital Harm—Postoperative Respiratory Failure eCQM as part of the eCQM measure set beginning with the CY 2026 reporting period/FY 2028 payment determination. The eCQM measure set is the measure set from which hospitals can self-select measures to report to meet the eCQM reporting requirement. We refer readers to section IX.C.9.c. of this final rule for a discussion of our previously finalized eCQM reporting and submission policies, as well as modifications for these requirements. Additionally, we refer readers to section IX.F.6.a.(2). of the preamble of this final rule for a discussion of a similar measure adoption in the Medicare Promoting Interoperability Program.
We invited public comment on our proposal to adopt the Hospital Harm—Postoperative Respiratory Failure eCQM beginning with the CY 2026 reporting period/FY 2028 payment determination.
Comment: Many commenters supported our proposal to adopt the Hospital Harm—Postoperative Respiratory Failure eCQM. Specifically, commenters noted that the measure represents an important measurement area, would promote patient safety, provide more timely information than claims data, and advance the use of eCQMs. A commenter also noted that the timeline was consistent with the amount of time that the industry needs to implement the measure.
Comment: A commenter supported the adoption of the Hospital Harm—Postoperative Respiratory Failure eCQM to encourage hospital prevention efforts while minimizing hospital reporting burdens and agreed that postoperative respiratory failure is “the most serious of the postoperative respiratory complications,” representing the end-stage of certain pulmonary complications (for example, pneumonia) and non-pulmonary problems (for example, sepsis). The commenter encouraged CMS to increase focus on preventing non-ventilator hospital-acquired pneumonia, which the commenter stated is a common, costly, and largely avoidable problem that is often unrecognized in hospitals.
Response: We thank the commenter for their support and will take their recommendation into consideration for future rulemaking as appropriate.
Comment: A commenter supported the measure but recommended that CMS consider expanding the exclusion criteria to include patients who are placed on mechanical ventilation for airway protection rather than respiratory failure. The commenter stated that examples of this population would include patients who experience a seizure or a cardiopulmonary event and require resuscitative measures and mechanical ventilatory support. Another commenter was also concerned that it may be inappropriate to consider any reintubation or mechanical ventilation after a procedure to be a postoperative respiratory failure, especially if the patient is recovering well or if the event occurs several days after the procedure.
Response: We thank the commenters for their feedback regarding the expansion of the denominator exclusion criteria. Regarding the appropriateness of considering any reintubation or mechanical ventilation after a procedure to be a postoperative respiratory failure, we note that high-quality, routine post-operative medical care and monitoring, including among patients who are stable or doing well several days after surgical procedures, should reduce the incidence of postoperative respiratory failure requiring reintubation and/or mechanical ventilation. [ 631 ] While we recognize that there will always be rare, unavoidable emergencies that require reintubation or mechanical ventilation, high quality post-operative nursing and medical care can typically catch the preventable problems that could lead to these situations. We will consider the recommendation to include patients placed on mechanical ventilation for airway protection and investigate the possibility of eliciting the nuances of intubation for airway protection versus intubation for respiratory failure from EHR data and existing codes for future measure updates.
Comment: A commenter supported adoption of the Hospital Harm—Postoperative Respiratory Failure eCQM in the Hospital IQR Program and encouraged the measure developer to consider including non-elective hospitalizations with appropriate risk stratifications and denominator exclusions to further improve postoperative respiratory failure monitoring.
Response: We thank the commenters for their feedback regarding the expansion of the denominator criteria to include non-elective hospitalizations. We will consider this expansion for future updates to the eCQM. The measure currently focuses on elective hospitalizations as postoperative respiratory failure may be more avoidable after elective procedures, which tend to be more clinically homogenous. Postoperative respiratory failure is generally considered a significant marker for complications after elective surgeries.
Comment: A commenter supported the adoption of the measures but encouraged CMS to consider the workflow and education impact of new measures and to align adoption and ( print page 69543) attestation timelines accordingly. Another commenter had concerns with the proposals to adopt new eCQMs and recommended adopting only one new eCQM per reporting period due to the burdensome process for building, tracking, and implementing new eCQMs.
Response: We carefully consider the benefit of adopting new measures in relation to any burden on hospitals. The program's shift toward digital measures would ultimately decrease the burden for hospitals because eCQMs use electronic standards, which help reduce the burden of manual abstraction and reporting for measured entities. We additionally note that the hospitals would initially have the option to self-select whether to report this eCQM to meet the eCQM reporting requirement for the Hospital IQR Program, providing flexibility for those hospitals that may need more time to implement this measure before being able to report it.
Comment: A few hospitals raised concerns about how hospitals capture the data used in the measure and how that data would be mapped to an eCQM. A commenter stated that hospital staff may write post-operative respiratory failure when the condition is due to an underlying condition and not related to the surgery. Another commenter stated that the relevant terminology is not well understood by physicians and is frequently documented incorrectly. Another commenter stated that the measure relies on procedure timing and stated that timestamps on measure components can complicate measure accuracy. The commenter stated that timestamps are metadata about an event and are not as easily mapped to eCQM logic as the event itself. A few commenters stated that the pre-rulemaking review of this measure raised concerns about variations in the capture of data by EHR vendors and that, as a result, clinicians may be using structured fields differently to input data, and documentation may not be captured in a standardized manner. The commenters stated that this could lead to measure performance being more dependent on the sensitivity of the screening technologies and approaches used than on underlying performance. A commenter said that it was important that CMS first align the data capture across a variety of vendors to allow ample time for hospitals to evaluate their EHR capabilities. A commenter stated that hospitals may require additional guidance to capture anesthesia data elements, stating that operating room documentation frequently uses notes or scanned paper records. That commenter stated that, as a result, anesthesia data are not always documented in a structured field or system that is interoperable with the certified EHR used for measure reporting.
Response: Feasibility testing was performed at 13 hospitals across three different EHR systems, and reliability and validity testing was performed at 12 hospitals across two different EHR systems. [ 632 ] This testing sample exceeds minimum testing requirements for an eCQM. [ 633 ] Feasibility testing results indicated that all the hospital sites confirmed that the data elements used in the measure are captured within the EHR in a structured and codified manner using nationally accepted terminology standards or local system codes that could be easily mapped. [ 634 ] Testing results found that while mechanical ventilation was captured in structured fields at all sites, documentation was not standardized. To account for differences in documentation workflows related to mechanical ventilation, the measure also accommodates the use of intubation and extubation outside of a procedural area to trigger a postoperative respiratory event. Additionally, during the 2022 call for public comment on this measure, feedback from interested parties noted that required data elements for the measure are routinely captured in structured data.
Validity testing results evaluated electronic clinical data compared to manually extracted data. The results indicate that in 89.6 percent of the cases where the patient was identified as experiencing the harm using EHR data, the result was confirmed using the manually extracted data. These results are an acceptable level of validity for measure testing and indicate that most hospitals would have the data available to calculate the measure in structured fields in their EHR. Further, we are proposing to implement the measure beginning with the CY 2026 reporting period, which would provide time for hospitals to review their workflows and make documentation updates if needed.
We acknowledge that certain underlying conditions place some patients at a higher risk of respiratory failure following a surgical procedure. To account for this, the measure excludes inpatient hospitalizations for patients with select underlying conditions and diagnoses that may increase their risk for postoperative respiratory failure. These exclusions, the full list of which can be found in the measure specification published on the eCQI Resource Center, were informed by clinical input received by the measure's technical expert panel and the 2022 public comment period for the measure.
To account for any ambiguity in defining postoperative respiratory failure, the measure's numerator includes explicit conditions that must be met for an event to be considered postoperative respiratory failure. These conditions were also informed by clinical input received by the measure's technical expert panel and the 2022 public comment period for the measure. Specifically, to meet the measure's numerator criteria, patients must have experienced (1) the initiation of mechanical ventilation within 30 days after the first operating room procedure, or (2) mechanical ventilation with a duration of more than 48 hours after the first operating room procedure. To account for differences in documentation workflows related to mechanical ventilation, the measure accommodates the use of intubation and extubation outside of a procedural area to trigger a postoperative respiratory event.
Regarding the commenter's concern related to reliance on procedure timing, eCQMs are updated during the eCQM annual update cycle to modify or improve the measure's logic expressions, value sets, and code systems, as necessary. In future updates to the eCQM, we will consider whether consolidation of this measure's timing elements into definitions, where appropriate, would improve the measure's accuracy by simplifying the measure's logic and making the relationships of these timing elements more evident. Feasibility testing results for the “Procedure, Performed”: “General and Neuraxial Anesthesia” data element showed 100% scores in data availability, accuracy, standards and in workflow. These results suggest that anesthesia data is typically documented in a structured field and can be accurately captured for the purpose of measure reporting.
Comment: Several commenters suggested that CMS carefully examine the potential for unintended consequences, such as inappropriate use ( print page 69544) of noninvasive positive pressure ventilation in lieu of mechanical respiration, excessive use of preventive tracheostomy, or avoidance of offering necessary procedures for high-risk patients. A commenter stated that CMS should address these issues before adopting this measure to ensure that the measure effectively enhances patient safety without unintended drawbacks.
Response: We agree that the potential for unintended consequences exists, although none were identified during the measure development and testing process. The intent of this measure is not to dictate clinical practice. Rather, it is to assist healthcare providers in highlighting, tracking, and responding to clinical quality improvement opportunities and to focus on prevention of patient harm. CMS expects hospitals to continue providing appropriate life-saving interventions based on sound clinical judgments. However, we note that we consistently monitor all the measures in the Hospital IQR Program for unintended consequences. Under our previously finalized measure removal policies, codified at 42 CFR 412.140(g)(2) and (3) ( 88 FR 59144 ), we could consider this measure for removal if we were to identify unintended consequences related to implementation of this measure.
Comment: Several commenters raised concerns that the Hospital Harm—Postoperative Respiratory Failure eCQM was duplicative of the PSI 11 component of the PSI 90 measure. A few commenters stated that CMS should consider the potential burden of overlapping measures and consider retiring PSI 90 in the future or allowing hospitals a choice of reporting method. A few commenters stated that if the Hospital Harm—Postoperative Respiratory Failure eCQM is adopted, CMS should outline a clear timeline for elimination of duplicative PSI measures. A commenter stated that the measure was similar to CMS PSI 11, and that they would not want two measures that are too similar but that CMS should not adopt the eCQM in order to replace CMS PSI 90, stating that CMS should not replace a claims-based measure with an eCQM since that shifts the burden to the hospital to build, map, and report the eCQM rather than it being based on claims.
Response: As we discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36306 through 36341 ) and in this final rule, while the CMS PSI 90 measure in the HAC Reduction Program includes a postoperative respiratory failure related component (PSI 11), it is a claims-based measure that uses a two-year performance period, it is focused on the Medicare FFS population, and is dependent upon ICD-10-CM codes. In the FY 2022 IPPS/LTCH PPS final rule, we highlighted our commitment to developing new digital quality measures that assess various aspects of patient safety in the inpatient setting ( 87 FR 49181 through 49190 ). The Hospital Harm—Postoperative Respiratory Failure eCQM provides the opportunity to assess the rate of postoperative respiratory failure in a much larger patient population and use more timely information from patients' electronic medical records instead of administrative claims data. The eCQM logic also allows for the use of mechanical ventilation, intubation, or extubation documentation outside of a procedural area to trigger a postoperative respiratory event, thus expanding opportunities for the electronic capture of information and accommodating varying clinical documentation workflows. In addition to these three measurement improvements, the development and implementation of the Hospital Harm—Postoperative Respiratory Failure eCQM aligns with CMS's commitment to moving to digital quality measures as highlighted in the FY 2022 IPPS/LTCH PPS final rule ( 87 FR 49181 through 49190 ). We note that we did not propose to remove the CMS PSI 90 measure from the HAC Reduction Program, while hospitals gain experience with the Hospital Harm—Postoperative Respiratory Failure eCQM.
Comment: A few commenters raised concerns about the testing of the measure. A commenter encouraged CMS to assess the feasibility of collecting the required data elements from EHRs and determine if the measure is reliable and valid across a broader set of EHRs vendors and hospitals. The commenter stated that assessment of how the measure performs using only three vendor systems and 13 hospitals is insufficient to generalize the measure's suitability to a broader population of facilities. A few commenters expressed concerns that the measure was tested only in teaching hospitals and thought that raised questions about the feasibility of implementation for all hospital types.
Response: We thank the commenters for their feedback and note that eCQM development must rely on a sample of hospitals. To guide and evaluate measure development and testing, CMS has developed the Measures Management System (MMS) Hub, and we consult the CBE's EM criteria. The MMS Blueprint and the CBE suggest that the minimum requirement for eCQM testing to test for feasibility of collecting the required data points and to determine if the measure is reliable and valid is to perform testing in at least three testing sites and within two EHR systems. [ 635 636 ] The testing for the Hospital Harm—Postoperative Respiratory Failure eCQM exceeds these requirements. Specifically, feasibility testing was performed at 13 hospitals across three different EHR systems, and reliability and validity testing was performed at 12 hospitals across two different EHR systems. [ 637 ] These hospital sites represent a diversity of hospital characteristics, including teaching status, size, electronic health record (EHR) vendor, and geographic location. In terms of teaching status, two hospitals were non-teaching hospitals, five were major teaching hospitals, and six were community teaching hospitals. In terms of size, four hospitals had between 100-199 beds, five hospitals had between 200-499 beds, and four hospitals had 499 beds. In terms of EHR systems, the hospitals used three different EHR vendors, combined these three vendors are used in more than two-thirds of acute care hospitals in the United States. [ 638 ] Finally, in terms of geographic locations, hospitals were headquartered in the Southeast, Northeast, and Western parts of the United States.
Comment: A commenter asked for a delay in required reporting on the Hospital Harm—Postoperative Respiratory Failure eCQM.
Response: Currently, as finalized in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49299 through 49302 ), hospitals must report on six total eCQMs beginning with the CY 2024 reporting period and subsequent years. Hospitals must report the following three eCQMs: (1) Hospital Harm—Severe Hypoglycemia eCQM; (2) Hospital Harm—Severe Hyperglycemia eCQM; and (3) Hospital Harm—Opioid-Related Adverse Events eCQM. Hospitals must also report three additional eCQMs that are self-selected from the list of ( print page 69545) remaining eCQMs. We reiterate this measure would be included as one of the eCQMs hospitals have the option to self-select for reporting beginning with the CY 2026 reporting period/FY 2028 payment determination. In section IX.C.9.c., we are finalizing our proposal to progressively increase the number of eCQMs a hospital must report beginning with the CY 2026 reporting period/FY 2028 payment determination, however, the Hospital Harm—Postoperative Respiratory Failure would remain a measure that hospitals can self-select to report. Future changes to the eCQM reporting requirements, including any additional eCQMs for mandatory reporting, would go through notice-and-comment rulemaking.
Comment: A commenter stated that the measure specification includes imprecise and repetitive data elements and timing expressions that would result in unnecessary administrative burden for EHR vendors, hospitals, and CMS' data receipt system, including the IntubationTime, MVTime, FirstProcedureTime, and GATime definitions. Another commenter requested that CMS share more details about the proposed Hospital Harm—Postoperative Respiratory Failure eCQM, noting that previous eCQM implementations have had issues with their logic and code sets.
Response: We thank the commenters for their feedback regarding the measure's data elements. We refer readers to the eCQI Resource Center ( https://ecqi.healthit.gov ) for more information about the measure's logic and code sets and definitions. The guidance section of the measure specification notes that post respiratory failure is evaluated using mechanical ventilation (MV) documentation or intubation and extubation documentation to allow for hospital documentation variances. Therefore, if MV documentation is not available, intubation and extubation can serve as a proxy for determining if MV occurred and its duration. During future measure development cycles, we will review the data elements included in this measure for specific sets of data (MVTime, AnesthesiaTime, FirstProcedureTime, etc.) and continue to identify opportunities to establish functions or definitions that generalize a number of the common patterns.
Additionally, eCQMs are updated during the eCQM annual update cycle to modify or improve the measure's logic expressions, value sets, and code systems, as necessary. Vendors and implementers have several opportunities throughout the year to provide input on potential measure changes. At any time during the year, implementers can submit specific measure questions through the eCQM Issue Tracker. [ 639 ] Common issues or questions received through the eCQM Issue Tracker are taken into consideration during the eCQM annual update process. Vendors and implementers are also able to preview and provide feedback on specific measure changes that are being considered each year during the annual update cycle's vendor and implementer review period, via the eCQM Issue Tracker. To receive updates about the vendor and implementer review period, users may create an account with the eCQI Resource Center.
Comment: A commenter expressed concerns regarding the expansion of eCQMs in hospital and ambulatory quality reporting programs. The commenter stated that the introduction of new eCQMs is shortsighted, burdensome, and fails to recognize the impending effort to shift toward digital quality measures (dQMs). The commenter stated that new eCQMs should not be required as part of quality reporting or pay-for-performance programs but that rather, CMS should invest efforts towards the future development of dQMs.
Response: We agree with the commenter in prioritizing investment in dQMs for quality measurement in order to improve accuracy, improve the timeliness of the information, and to reduce reporting burden. We note the definition of dQM that we have published as part of strategic materials on the eCQI Resource Center states that in general, eCQMs are a subset of dQMs. This definition states that dQMs are “quality measures that use standardized, digital data from one or more sources of health information that are captured and exchanged via interoperable systems; apply quality measure specifications that are standards-based and use code packages; and are computable in an integrated environment without additional effort.” This definition of a dQM is available on the eCQI Resource Center at: https://ecqi.healthit.gov/dqm?qt-tabs_dqm=about-dqms .
After consideration of the public comments we received, we are finalizing our proposal of the Hospital Harm—Postoperative Respiratory Failure eCQM as proposed beginning with the CY 2026 reporting period/FY 2028 payment determination. We refer readers to section IX.F.6.a.(2). of the preamble of this final rule for a discussion of the adoption of this measure in the Medicare Promoting Interoperability Program. We also refer readers to section XXXX of the preamble of this final rule where we discuss the use of this measure in the Transforming Episode Accountability Model (TEAM).
Failure-to-rescue is defined as the probability of death given a postoperative complication. [ 640 641 642 ] Hospitals can implement evidence-supported interventions to improve timely identification of clinical deterioration and treatment of potentially preventable complications, including improved nurse staffing, simulation training, standardized communication tools, electronic monitoring and/or warning systems, and rapid response systems. [ 643 644 645 646 647 648 ] Studies also ( print page 69546) show that other processes of care can influence failure-to-rescue rates, including a hospital's aggressiveness of care (defined as the level of resources or inpatient spending), with hospitals that treat patients more aggressively (such as providing more inpatient days or ICU days in the last 2 years of life) having lower surgical mortality and failure-to-rescue rates than otherwise similar hospitals that treat patients less aggressively. [ 649 650 ] Hospitals and healthcare providers benefit from knowing not only their institution's mortality rate, but also their institution's ability to rescue patients after an adverse occurrence. Using a failure-to-rescue measure is especially important if hospital resources needed for preventing complications are different from those needed for rescue.
This Failure-to-Rescue measure was designed to improve upon the CMS Patient Safety Indicator 04 Death Rate Among Surgical Inpatients with Serious Treatable Complications (CMS PSI 04) measure in the Hospital IQR Program. We refer readers to section IX.C.6.a. for our proposal to remove the CMS PSI 04 measure contingent upon the adoption of the Failure-to-Rescue measure. Both the Failure-to-Rescue measure and the CMS PSI 04 measure focus on hospitals' ability to rescue patients who experience clinically significant complications after inpatient operations, so that these complications do not result in death. Both measures are sensitive to factors such as appropriate nurse staffing and nursing skill-mix, which enable hospitals to identify complications earlier and intervene effectively to prevent death.
The Failure-to-Rescue measure directly addresses concerns about the CMS PSI 04 measure, including:
- Complications sometimes develop before the index operation in CMS PSI 04, even before transferring to the index hospital. For example, the operation is part of an effort to “rescue” the patient.
- The heterogeneous cohort includes patients with very high-risk surgery (for example, trauma surgery, burn surgery, organ transplants, intracranial hemorrhage) and very low-risk surgery (for example, eye, ear, urolithiasis).
- Mean length of stay and prevalence of early discharge to post-acute facilities vary across hospitals, causing bias in comparing performance.
- CMS PSI 04 may slightly disadvantage teaching hospitals, even after risk-adjustment, due to residual confounding from unmeasured case-mix differences.
The Failure-to-Rescue measure has four major differences compared to CMS PSI 04:
1. Captures all deaths of denominator-eligible patients within 30 days of the first qualifying operating room procedure, regardless of site.
2. Limits the denominator to patients in general surgical, vascular, and orthopedic Medicare Severity Diagnosis Related Groups (MS-DRGs).
3. Excludes patients whose relevant complications preceded (rather than followed) their first inpatient operating room procedure, while broadening the definition of denominator-triggering complications to include other complications that may predispose to death (for example, pyelonephritis, osteomyelitis, acute myocardial infarction, stroke, acute renal failure, heart failure/volume overload).
4. Measure cohort includes Medicare Advantage patients.
We proposed to adopt the Failure-to-Rescue measure beginning with the performance period of July 1, 2023—June 30, 2025, affecting the FY 2027 payment determination.
The Failure-to-Rescue measure is a risk-standardized measure of death after hospital-acquired complication. The measure denominator includes patients 18 years old and older admitted for certain procedures in the General Surgery, Orthopedic, or Cardiovascular Medicare Severity Diagnosis Related Groups (MS-DRGs) who were enrolled in the Medicare program and had a documented complication that was not present on admission. The measure numerator includes patients who died within 30 days from the date of their first “operating room” procedure, regardless of site of death.
We refer readers to CMS' QualityNet website: https://qualitynet.cms.gov/inpatient/iqr/proposedmeasures#tab2 (or other successor CMS designated websites) for more details on the measure specifications.
The Failure-to-Rescue measure aligns with several goals under the CMS National Quality Strategy. [ 651 ] In alignment with the goal to “Promote Alignment” and “Improved Health Outcomes,” this outcome-based measure would allow hospitals to track their institution's ability to rescue patients after an adverse occurrence and encourage hospitals to focus on early identification and rapid treatment of complications, thereby improving the overall quality of care and health outcomes of patients in the inpatient setting. In alignment with the goal to “Ensure Safe and Resilient Health Care Systems,” the Failure-to-Rescue measure includes a larger patient population than the CMS PSI 04 measure. The Failure-to-Rescue measure includes Medicare Advantage data, and the denominator includes a much broader range of hospital-acquired complications (for example, kidney dysfunction, seizures, stroke, heart failure, and wound infection) than the CMS PSI 04 measure.
We refer readers to section IX.B.1.c. of the preamble of this final rule for details on the PRMR process including the voting procedures the PRMR process uses to reach consensus on measure recommendations. The PRMR Hospital Committee, comprised of the PRMR Hospital Advisory Group and PRMR Hospital Recommendation Group, reviewed measures included by the Secretary on a publicly available “2023 Measures Under Consideration List” (MUC List), [ 652 653 ] including the Failure-to-Rescue measure (MUC2023-049). The PRMR Hospital Recommendation Group reviewed the proposed updates to the Failure-to-Rescue measure (MUC2023-049) during a meeting on January 18-19, 2024. [ 654 655 ]
The committee reached consensus and recommended including this measure in the Hospital IQR Program with conditions. Twelve members of the ( print page 69547) group voted to adopt the measure into the Hospital IQR Program without conditions; five members voted to adopt with conditions; two committee members voted not to recommend the measure for adoption. Taken together, 89.5 percent of the votes were recommend or recommended with conditions. The five members of the voting committee who voted to adopt with conditions specified the condition as collecting data to evaluate possible unintended consequences, such as hospitals encouraging patients to sign a DNR order or enter hospice. We agree with the potential for unintended consequences and note that we consistently monitor all the measures in the Hospital IQR Program for unintended consequences. Furthermore, we note that under our previously finalized measure removal Factor 6, codified at 42 CFR 412.140(g)(3)(i)(F) and (3) ( 88 FR 59144 ), collection or reporting of a measure leads to negative unintended consequences other than patient harm, if we were to identify unintended consequences related to this measure we would consider it for removal.
Feedback was generally positive with some discussion around whether the measure was enough of an improvement on CMS PSI 04. The measure developer highlighted several areas of improvement compared to CMS PSI 04, including increased reliability and validity largely due to the application of this measure to both Medicare Advantage and fee-for-service enrollees, as well as the inclusion of deaths after hospital discharge but within 30 days of the index operative procedure. [ 656 ]
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for details on the EM process including the measure evaluation procedures the EM Committees uses to evaluate measures and whether they meet endorsement criteria. The EM Management of Acute Events, Chronic Disease, Surgery, and Behavioral Health Committee convened in the Fall Cycle 2023 to review the Failure-to-Rescue measure (CBE #4125) which was submitted to the CBE for endorsement. The EM Management of Acute Events, Chronic Disease, Surgery, and Behavioral Health Committee ultimately voted to endorse with conditions on January 29th, 2024. [ 657 ] The condition was: perform additional reliability testing for endorsement review, namely conducting additional simulation analyses of minimum case volume adjustments. [ 658 ] We will monitor the data as part of the standard measure maintenance.
The measure is calculated using Medicare fee-for-service (FFS) Part A inpatient claims data and Medicare Inpatient Encounter data for Medicare Advantage enrollees, in combination with validated death data from the Medicare Beneficiary Summary File or equivalent resources. CMS receives death information from several sources: Medicare claims data from the Medicare Common Working File (CWF); online date of death edits submitted by family members; and benefit information used to administer the Medicare program collected from the Railroad Retirement Board (RRB) and the Social Security Administration (SSA). Like the CMS 30-day mortality measures, the “Valid Date of Death Switch” is used to confirm that the exact day of death has been validated.
This measure was tested using Medicare inpatient hospital discharge data from 2,055 IPPS hospitals with at least 25 eligible discharges from January 1, 2021, through June 30, 2022. Hospital-level performance rates are depicted in Table IX.C-2. [ 659 ] Because lower scores are better the lower performance percentiles are better performing hospitals than those in the higher percentiles (for example, the hospitals in the fifth percentile are the best performing hospitals).

If hospitals currently in the worst quartile (that is, those at the 75th percentile) were to improve performance to the performance of hospitals in the best quartile (that is, those at the 25th percentile) it would represent a 50 percent decrease in the frequency of deaths after postoperative complications at those hospitals. [ 660 ]
Test results indicated moderate measure reliability and strong validity. [ 661 ]
This measure uses readily available administrative claims data routinely generated and submitted to CMS for all Medicare beneficiaries, which includes Medicare Advantage and Medicare fee-for-service patients. Hospitals would not ( print page 69548) be required to report any additional data. We have used a similarly designed claims-based measure (CMS PSI 04) for over a decade. The Failure-to-Rescue measure would be calculated and publicly reported on annual basis using a rolling 24 months of prior data for the measurement period, consistent with the approach currently used for CMS PSI 04 and PSI 90, the Patient Safety and Adverse Events Composite.
We invited public comment on our proposal to adopt the Thirty-day Risk-Standardized Death Rate Among Surgical Inpatients with Complications measure beginning with the CY 2025 reporting period/FY 2027 payment determination.
Comment: Many commenters supported the proposal to add the Failure-to-Rescue measure as a replacement for CMS PSI 04 in the Hospital IQR Program. Several commenters noted the adoption of this measure would incentivize hospitals to provide patients and family members with clear mechanisms to voice their concerns and to empower staff (such as less senior staff) to do so as well. A few commenters noted that the measure can serve as a valuable metric to help hospitals identify areas where they can improve their quality of care, enhance patient outcomes, and prevent death. Another commenter noted that the adoption of this measure would capture additional deaths while employing more precise exclusions than CMS PSI 04. A commenter stated that the adoption of the measure would help to recognize hospitals that are exemplars in patient safety, as well as refine the data collected so that it is meaningfully used to prioritize safety improvement for the most vulnerable populations. A commenter applauded CMS for applying this measure to both Medicare FFS and Medicare Advantage patients. A commenter expressed their support for the removal of CMS PSI 04 contingent on the adoption of the Failure-to-Rescue measure.
Response: We thank commenters for their support.
Comment: A few commenters noted the adoption of the Failure-to-Rescue measure improves upon CMS PSI 04 because it includes a denominator limitation to a more appropriate set of index DRGs for inclusion, adds Medicare Advantage patients to the measure population, and excludes complications that occur prior to surgery rather than focusing on complications that occur post-procedure.
Response: We thank commenters for their support and agree with their feedback. Using the Failure-to-Rescue measure instead of the CMS PSI 04 would allow us to assess an expanded population, encourage safe practices for the widest range of surgical inpatients, and allow hospitals to identify opportunities to improve their quality of care.
Comment: Many commenters expressed concerns about using patient safety measures derived from claims data, because in their view, such data would not accurately reflect hospital performance and would eliminate the clinical components of care from quality calculations. To close this gap, commenters requested that CMS explore alternative data sources other than claims data to ensure an accurate and fair hospital performance assessment. A commenter also suggested the Failure-to-Rescue measure become an eCQM.
Response: We disagree that patient safety measures derived from claims data eliminate clinical components of care from quality measurement, or that claims-based measures are inaccurate. Studies have shown that using claims data is a robust approach to capturing variation in mortality outcomes across hospital systems. [ 662 ] Additionally, claims data contain all the necessary data elements to accurately calculate the Failure-to-Rescue measure. Testing results for the Failure-to-Rescue measure were shown to have high face-validity; and 90 percent of the TEP members supported the measure's relevance in assessing quality of care and agreed that the measure could improve quality of care by reducing the frequency of failure to rescue. [ 663 ] Furthermore, the measure exhibited adequate signal-to-noise reliability, indicating its ability to distinguish between the quality of care across facilities. Notably, hospitals with higher nurse staffing levels and skill mix tend to exhibit lower mortality rates following serious postoperative complications. Conversely, hospitals that delay identification or fail to aggressively treat complications are associated with higher rates of 30-day readmissions and mortality post complications. [ 664 ]
We note that during the measure development process we are responsible for determining whether to account for variation in factors intrinsic to the patient before comparing outcomes and to determine how to best apply these factors in the quality measure specifications. Additionally, in our view, utilizing claims data where practical helps to minimize the reporting burden on hospitals and provides sufficient and high-quality data and therefore the Failure-to-Rescue measure is appropriate as a claims-based measure rather than an eCQM. However, we will consider exploring alternative data sources for other quality measurement topics in the future.
Comment: A few commenters expressed concerns about the risk adjustment methodologies for the Failure-to-Rescue measure. A commenter strongly opposed the risk adjustment for patients who enter the hospital with COVID-19 because the commenter noted that the policy excludes a wide range of patients. A commenter stated that it is unclear whether the revised risk adjustment methodology for the Failure-to-Rescue measure would appropriately account for differences between hospitals. A commenter recommended that CMS risk-adjust the Failure-to-Rescue measure to account for differences in patient populations and case mixes across hospitals. The commenter suggested that such risk adjustment factors should include severity of illness, comorbidity burden, socioeconomic factors, and care setting characteristics. The commenter argued that by incorporating risk adjustment methodologies in the Failure-to-Rescue measure, CMS will ensure a comprehensive and equitable evaluation of hospitals because patient outcomes can be influenced by factors outside the control of healthcare providers.
Response: We thank commenters for their feedback on risk adjustment methodologies. However, as we discussed in the proposed rule ( 89 FR 36324 ), measure testing results indicated moderate measure reliability and strong validity. Further, we have used a similarly designed claims-based measure (CMS PSI 04) for over a decade. For additional details on the measure's risk adjustment model, please refer to the measure details available on the Partnership for Quality Measurement website. [ 665 ] The Failure-to-Rescue measure, like all quality measures, would undergo a rigorous maintenance review process, in which we would evaluate the potential inclusion of additional factors into the measure's risk adjustment model. We note that including COVID-19 as a risk factor in the risk adjustment model does not exclude patients with COVID-19. Rather, by including COVID-19 status in the model, we recognize COVID-19 as an important risk factor within the model. This approach ensures that hospitals are not penalized for treating ( print page 69549) a higher number of COVID-19 patients, but we agree with the commenter that all patients deserve quality care, including patients with COVID-19. Additionally, we have ensured the Failure-to-Rescue measure appropriately accounts for differences between hospitals, as we recognize this is an important risk factor within the model.
Comment: A few commenters requested CMS consider the workflow and education impact of the high volume of measures proposed for FY2025 and ensure adoption alignment.
Response: We thank commenters for their input. We are mindful of the reporting burden for participants in the Hospital IQR Program. Adopting a measure can result in changes in workflow as well as staff training. However, those changes are necessary where quality measures ensure that hospitals meet the applicable standard of care. We remind commenters that the Failure-to-Rescue measure is a claims-based hospital measure, calculated using Medicare Advantage data and Medicare FFS claims that are already reported to the Medicare program for payment purposes. Adopting this measure would not result in a change in hospitals' reporting burden.
Comment: Several commenters requested that CMS release the full measure specifications as soon as possible to ensure that interested parties have sufficient clinical documentation of coding. Commenters suggested that providing documentation regarding justification for each surgical complication used in the Failure-to-Rescue measure along with their respective ICD-10 based definitions would be helpful to program participants.
Response: We thank the commenters for their feedback regarding measure specifications. As described in the proposed rule, we refer readers to CMS' QualityNet website: https://qualitynet.cms.gov/inpatient/iqr/measures (or other successor CMS designated websites) for the measure specifications. The measure specifications detail the surgical complications included in the Failure-to-Rescue measure along with their respective ICD-10 based definitions and their relevant exclusions. Any future updates to the technical measure specification will be announced through the QualityNet website and listserv announcements.
Comment: A few commenters expressed concern that the abbreviated name of the measure, Failure-to-Rescue, may not appropriately describe the underlying measure's focus. Commenters suggested that the name of the measure is misleading to consumers and may evoke an image of disregard for human suffering and elicit feelings of distrust or avoidance of any hospital with a non-zero performance rate. A commenter suggested that the abbreviated name uses blaming language that implies hospitals are directly at fault for patients who may die within the 30 days of surgical complication. To close this gap, a commenter requested that CMS work with patients and communities to determine whether the measure's name appropriately meets patients' understanding of the measure and suggested CMS rename the measure to something that adequately correlates to patients' understanding of the measure. The commenter further requested CMS identify how the Failure-to-Rescue measure would be used among patients to make determinations about where to seek inpatient surgical care.
Response: We thank commenters for their feedback on the abbreviated name of the measure, Failure-to-Rescue, and will take this suggestion into consideration during future measure updates. The measure's full name is Thirty-Day Risk-Standardized Death Rate Among Surgical Inpatients With Complications, and we will consider the most appropriate way to display measure performance on this clinical topic for purposes of public reporting. Additionally, we acknowledge the suggestion to identify how the measure would be used to make determinations about where to seek inpatient surgical care. We reiterate that this measure assesses the percentage of surgical inpatients who experienced a complication and then died within 30-days from the date of their first “operating room” procedure. Hospitals can use the measure to identify opportunities to improve their quality of care and patient safety. With strategies that focus on improving patient centered care, the measure would ensure that the decisions from these hospitals respect patients' needs and preferences to make the appropriate determinations for their care.
Comment: A commenter requested that CMS provide clarity in defining the Failure-to-Rescue measure. The commenter suggested the Failure-to-Rescue measure's definition should provide a comprehensive understanding of what constitutes as a “failure to rescue” event, including specific criteria for identifying such events.
Response: We thank commenters for their feedback on clarifying the definition of a “failure to rescue” event. The Failure-to-Rescue measure is a risk-standardized measure of death after a hospital-acquired complication. However, we note that, as we discussed in the proposed rule ( 89 FR 36322 ), the measure's denominator includes patients 18 years old and older admitted for certain procedures in the General Surgery, Orthopedic, or Cardiovascular Medicare Severity Diagnosis Related Groups (MS-DRGs) who were enrolled in the Medicare program and had a documented complication that was not present on admission. The measure's numerator includes patients who died within 30 days from the date of their first “operating room” procedure, regardless of site of death. We will work with interested parties to ensure that the measure's documentation is as clear as possible to ensure that hospitals can provide the best possible care to their patients. Additional details about the Failure-to-Rescue measure can be found on the QualityNet website: https://qualitynet.cms.gov/inpatient/iqr/proposedmeasures#tab2 and the Partnership for Quality Measurement website: https://p4qm.org/measures/4125 . We consistently monitor all the measures in the Hospital IQR Program for unintended consequences, and we will monitor performance on this measure to ensure that those unintended consequences do not result from its adoption.
Comment: Several commenters expressed concerns that the proposed performance period for the Failure-to-Rescue measure would begin on July 1, 2023, more than a year prior to the measure's adoption in the Hospital IQR Program. Commenters questioned if it was appropriate for CMS to utilize claims data from July 2023 for quality measurement purposes. A commenter elaborated and requested that CMS reconsider the timeline of the Failure-to-Rescue measure proposal to avoid incorporating claims data for years in which the measure had not been adopted in the Hospital IQR Program. The commenters further requested that CMS delay the measure's adoption for at least one year to allow hospitals time to familiarize and educate staff around the Failure-to-Rescue measure requirements and changes compared to CMS PSI 04 and to align with the other Hospital IQR Program proposals this year.
Response: We thank the commenters for their feedback on the proposed measurement period. As we noted in the proposed rule ( 89 FR 36324 ), the measure is calculated and publicly reported on an annual basis using a rolling 24 months of prior data for the measurement period. This performance period is consistent with the CMS PSI 04 measure and the CMS PSI 90 composite measure. We acknowledge ( print page 69550) that the Failure-to-Rescue measure's performance period would begin on July 1, 2023, as with many other quality measures whose performance period occurs in the past and whose calculations are incorporated into payment determinations for future years. However, because this measure is calculated and publicly reported on an annual basis using a rolling 24 months of prior data, this policy is necessary to ensure that we calculate the measure with sufficient reliability and validity using a sufficiently large claims data set. Additionally, by adopting the measure with this performance period, we can ensure that we continue assessing hospitals on this important clinical subject rather than delaying the measure's adoption until the FY 2028 payment determination, as would be necessary if we adopted the measure with a performance period beginning on July 1, 2024. Since this measure will replace CMS PSI 04, it would be appropriate to delay measurement of the clinical topic when we have an improved measure available. We remain confident that hospitals and hospital staff will be able to familiarize themselves with the measure's requirements in comparison to CMS PSI 04, particularly because the measure represents an improvement on CMS PSI 04 rather than a fundamental change to the clinical topic's measurement.
Comment: A commenter requested that CMS provide hospitals with a dry run of their Failure-to-Rescue measure results prior to adoption and mandatory reporting in the Hospital IQR Program.
Response: We thank the commenter for their feedback. We intend to provide hospitals with information on their performance on the measure as part of the Hospital IQR Program's preview period process prior to public reporting. As finalized in the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50776 ), quality data displayed for each quarter on Care Compare are made available to providers for a 30-day preview period approximately two months in advance of display.
Comment: A few commenters did not support the measure's adoption, citing concerns with its reliability. Commenters stated that testing for this measure demonstrated that the reliability was 0.231 using the measure's case minimum of 25 patients and that it required roughly 600 patients to achieve a high level of reliability (0.7 at minimum). Commenters further noted that the low reliability results were questioned during the recent CBE endorsement review. Commenters explained that the committee placed conditions to perform additional reliability testing, and to conduct additional simulation on the measure's endorsement. The commenters requested CMS conduct the recommended additional testing and that the conditions be removed from endorsement before the Failure-to-Rescue measure is used in the Hospital IQR Program.
Response: We appreciate commenters' feedback regarding the reliability of the Failure-to-Rescue measure. The committee placed conditions on the measure's endorsement to perform additional reliability testing for endorsement review, namely, to conduct additional simulation analyses of minimum case volume adjustments. During the EM committee review, the measure developer responded to concerns regarding low reliability by emphasizing that the Failure-to-Rescue measure is an improvement compared to the CMS PSI 04 measure due to increased reliability and validity largely due to the application of this measure to both Medicare Advantage and fee-for-service enrollees, as well as the inclusion of deaths after hospital discharge but within 30 days of the index operative procedure. [ 666 ] Additional details related to the Failure-to-Rescue measure's reliability results can be found on the Partnership for Quality Measurement website. [ 667 ] As discussed earlier, we agree with the potential for unintended consequences and note that we consistently monitor all the measures in the Hospital IQR Program for unintended consequences. Furthermore, we note that under our previously finalized measure removal Factor 6, codified at 42 CFR 412.140(g)(3)(i)(F) and (3) , collection or reporting of a measure leads to negative unintended consequences other than patient harm, if we were to identify unintended consequences related to this measure we would consider it for removal.
Comment: A few commenters expressed concerns with the exclusion of various patient populations in the Failure-to-Rescue measure. A commenter noted the Failure-to-Rescue measure excludes the most vulnerable patients, who it stated are often at the highest risk of death. To close this gap, the commenter recommended that CMS add a measure to the Hospital IQR Program that gauges hospital mortality performance, observing the most vulnerable cases that are at a heightened risk of death. The commenter expressed they do not support the adoption of the measure unless CMS provides an alternative method to account for the most vulnerable patients. A commenter noted concerns with the exclusion of patients whose complications preceded a surgical event. A commenter also requested that CMS consider expanding the Failure-to-Rescue measure to include other populations and procedures, such as recipients of inpatient cellular therapy services that experience complications. The commenter further suggested that CMS consider adding stratifications for interested parties to better understand performance by procedure.
Response: We thank the commenters for their feedback on the exclusion of various patient populations. The Hybrid Hospital-Wide All-Cause Risk-Standardized Mortality (Hybrid HWM) measure in the Hospital IQR Program complements the Failure-to-Rescue measure and meets the need the commenter suggested for a measure of mortality that can account for the most vulnerable cases that are at a heightened risk of death. Currently, we do not plan to expand the Failure-to-Rescue measure to include other populations or stratify the measure. The Failure-to-Rescue measure, like all quality measures, would undergo a rigorous maintenance review process, in which the measure steward would evaluate the need for changes to the measure.
Comment: A commenter expressed concerns about the data challenges to reliably measure the quality of care for Medicare Advantage patients. The commenter elaborated that the Medicare Payment Advisory Commission has consistently noted challenges with the completeness and accuracy of MA encounter data. To close this gap, the commenter recommended CMS consider policies to ensure that Medicare Advantage plans would be able to provide complete encounter data for quality measurement.
Response: We thank the commenter for their feedback. However, as we stated in the proposed rule ( 89 FR 36323 ), the measure developer highlighted the measure's increased reliability and validity in comparison to CMS PSI 04 largely due to the application of this measure to both Medicare Advantage and fee-for-service enrollees as well as the inclusion of deaths after hospital discharge but within 30 days of the index operative procedure. As required under ( print page 69551) § 422.504(l), Medicare Advantage organizations certify the accuracy, completeness, and truthfulness of their encounter data (based on best knowledge, information, and belief). [ 668 ] Medicare Advantage plans conduct self-assessments regarding the accuracy and completeness of their encounter data submissions for each contract they have with CMS, and each year Medicare Advantage plans apply the findings from their self-assessments to improve the accuracy and completeness of their submissions. [ 669 ] There is an established compliance framework, including identification of initial metrics for assessing completeness and accuracy, [ 670 ] to ensure that the encounter data provided is as reliable and as complete as possible for the Failure-to-Rescue measure.
Comment: A commenter suggested that CMS maintain transparency and actively engage with interested parties throughout the development and implementation process of the Failure-to-Rescue measure to ensure the success of measures aimed at enhancing high-quality, patient-centered care. The commenter further recommended that CMS seek input from a diverse range of interested parties, including clinicians, researchers, and patient advocacy groups, to ensure the measure aligns with its objectives and reflects the need to prioritize all those involved.
Response: We thank the commenter for their feedback. To ensure transparency throughout the measure development process, a Technical Evaluation Panel (TEP) provided direction and input from interested parties to the measure developer in every phase of the measure development process. Measure developers incorporated feedback from TEPs upon their review of the measure testing results. We also submitted this measure through the PRMR process for input from a multistakeholder group of clinicians, patients, and other interested parties. We refer readers to section IX.B.1.c. of the preamble of this final rule for details on the PRMR process including the voting procedures the PRMR process uses to reach consensus on measure recommendations.
Comment: A commenter expressed concerns with the Failure-to-Rescue measure having exclusions regarding documentation errors and missing demographic information, which in the commenter's opinion allows too much leeway for hospitals to remove such cases from the measure's calculation.
Response: We thank the commenter for their input on excessive exclusions. However, we note that because the measure is claims-based, hospitals do not have the discretion to withhold cases from its calculation so long as they have submitted those claims for payment. We will monitor the data that underpins this measure carefully to ensure that the measure's exclusions are implemented appropriately.
Comment: A commenter suggested it would be helpful for CMS to provide hospitals with additional guidance and resources for support in implementing evidence-based practices that aim to reduce Failure-to-Rescue rates and improve patient outcomes.
Response: We thank the commenter for their feedback and the recommendation to provide additional guidance and resources to support hospitals in implementing evidence-based practices that aim to reduce Failure-to-Rescue measure rates and improve patient outcomes. As noted in the Partnership for Quality Measurement website ( https://p4qm.org/measures/4125 ), there are evidence-supported interventions that hospitals can implement to improve timely identification of clinical deterioration and treatment of preventable complications, including improved nurse staffing, simulation training, standardized communication tools, electronic monitoring or warning systems, and rapid response systems.
Comment: A commenter recommended that CMS exclude Medicare Advantage patients from the Failure-to-Rescue measure's calculations because, in the commenter's view, this population is subject to quality initiatives under specific managed care payer contracts.
Response: While the commenter is correct that we implement other quality initiatives for Medicare Advantage patients, the measure's incorporation of Medicare Advantage patients improves its reliability and validity and appropriately provides Medicare beneficiaries with additional information about the quality of care that they receive from hospitals.
Comment: Several commenters expressed concerns that the Failure-to-Rescue measure may hold hospitals accountable for factors outside of their control. Commenters elaborated on how a person may die within 30 days of a hospital procedure for various reasons unrelated to the hospital's quality of care, including self-harm or trauma, which may increase the risk of skewing the data. To address this issue, commenters requested CMS to consider adding additional exclusions to the Failure-to-Rescue measure to ensure the measure is reflective of the hospital's performance. A commenter also noted concerns with deaths outside the hospital system may have missing information, ultimately creating challenges for hospitals for potential improvement.
Response: The measure's denominator exclusions control for factors affecting patients' clinical care that are beyond the hospital's control, such as a patient age of over 90 years, a “do not resuscitate (DNR)” status present on admission, or a departure against medical advice. We refer readers to the Failure-to-Rescue Measure Specifications on the QualityNet website at: https://qualitynet.cms.gov/inpatient/iqr/measures for more details and a complete list of the denominator exclusions. We note additionally that the complications in the Failure-to-Rescue measure are all serious adverse health events. We will monitor the measure's effects on the provision of clinical care to avoid any unintended consequences of its adoption.
Comment: A commenter urged CMS to ensure that healthcare providers have access to sufficient resources and support to enable accurate data collection and reporting. To encourage participation and compliance, the commenter stated that CMS must minimize any administrative burdens and streamline data submission mechanisms wherever possible.
Response: We are mindful of the administrative burden for participants in the Hospital IQR Program. We reiterate the Failure-to-Rescue measure would be included as one of the claims-based hospital measures, calculated using Medicare Advantage data and Medicare FFS claims that are already reported to the Medicare program for payment purposes. Hospitals would not be required to report any additional data, so adoption of this measure would not result in a change in burden.
Comment: A commenter expressed concerns with the unintended consequences that may arise based on ( print page 69552) the well-known limitations in measurement science. The commenter further elaborated that measures should be assessed regularly for their effectiveness, impact, and overall performance, which are all an essential part of measurement science. To address this issue, the commenter recommended that CMS utilize an unbiased party to conduct such assessments, and to track and report any issues that arise in a timely manner.
Response: We agree that the potential for unintended consequences exists and note that we consistently monitor and evaluate all the measures in the Hospital IQR Program for unintended consequences. With respect to the concern regarding the use of unbiased parties to assess the measure for potential unintended consequences and enhancements, the current CBE (which consists of a variety of experts including clinicians, measure experts, and health IT specialists) conducts annual reviews to provide recommendations regarding the quality and efficiency measures in CMS programs. [ 671 ] Common issues or questions received are taken into consideration during the annual update process. Additionally, the CBE's Measure Set Review process provides additional recommendations for us to consider as we refine our programs.
Comment: A commenter did not support the adoption of the Failure-to-Rescue measure because they stated that the measure lacks insight into post discharge care or related complications. The commenter argued that CMS PSI 04 and the current 30-day mortality measures provide hospitals with opportunities to improve their care and post-discharge planning and therefore adoption of the Failure-to-Rescue measure is not necessary.
Response: As we stated in the proposed rule ( 89 FR 36323 ), the measure aligns with several goals under the CMS National Quality Strategy and is outcome-based. We consider the Failure-to-Rescue measure as an improvement on CMS PSI 04 and therefore, in connection with the proposal to adopt this measure in the Hospital IQR Program, we also proposed to remove the CMS PSI 04 measure from the Hospital IQR Program. We discuss removal of the CMS PSI 04 measure in detail in the next section.
After consideration of the public comments we received, we are finalizing adoption of the Failure-to-Rescue measure as proposed beginning with the July 1, 2023-June 30, 2025, reporting period/FY 2027 payment determination. We also refer readers to section XXXX of the preamble of this final rule where we discuss the use of this measure in the Transforming Episode Accountability Model (TEAM).
We proposed to remove five measures: (1) Death Among Surgical Inpatients with Serious Treatable Complications (CMS PSI 04) measure beginning with the July 1, 2023-June 30, 2025 reporting period/FY 2027 payment determination; (2) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Acute Myocardial Infarction (AMI) measure beginning with the July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination; (3) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Heart Failure (HF) measure beginning with the July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination; (4) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Pneumonia (PN) measure beginning with the July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination; and (5) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Elective Primary Total Hip Arthroplasty (THA) and/or Total Knee Arthroplasty (TKA) measure beginning with the April 1, 2021-March 31, 2024 reporting period/FY 2026 payment determination. We provide more details on each of these proposals in the subsequent sections.
We proposed to remove the Death Among Surgical Inpatients with Serious Treatable Complications (CMS PSI 04) measure, beginning with the FY 2027 payment determination associated with the performance period of July 1, 2023-June 30, 2025, based on removal Factor 3, [ 672 ] the availability of a more broadly applicable measure (across settings, populations), or the availability of a measure that is more proximal in time to desired patient outcomes for the particular topic. The CMS PSI 04 measure was adopted into the Hospital IQR Program in the FY 2009 IPPS/LTCH PPS final rule ( 73 FR 48607 ). The CMS PSI 04 measure records in-hospital deaths per 1,000 elective surgical discharges, among patients ages 18 through 89 years old or obstetric patients with serious treatable complications (shock/cardiac arrest, sepsis, pneumonia, deep vein thrombosis/pulmonary embolism, or gastrointestinal hemorrhage/acute ulcer). [ 673 ] It is a claims-based measure which uses claims and administrative data to calculate the measure without any additional data collection from hospitals. The measure was previously endorsed (CBE #0351), but given the measurement's limitations, endorsement was not maintained by the measure steward, and the measure has not been updated since 2017. [ 674 ]
In the FY 2022 IPPS/LTCH PPS proposed rule ( 86 FR 25579 through 25580 ), we proposed to remove this measure under removal Factor 3, codified at 42 CFR 412.140(g)(3)(i)(C) , noting at that time that the Hybrid Hospital-Wide Mortality measure (Hybrid HWM) (CBE #3502) was more broadly applicable. Some public commenters, however, expressed concerns about replacing CMS PSI 04 with the Hybrid HWM measure since the Hybrid HWM measure would report on the mortality rate of the entire hospital, instead of specifically measuring the deaths of surgical inpatients in an effort to assess postoperative mortality distinct from hospital-wide mortality ( 86 FR 45391 ). Other commenters elaborated on this concern stating that by removing a postoperative-specific mortality measure, hospitals may lose the ability to account for what resources they need to better care for surgical inpatients since that population's needs often differs from the needs of non-surgical IPPS hospital patients ( 86 FR 45390 through 45391 ). [ 675 ] Some commenters suggested modifications to the existing CMS PSI 04 measure such as changing its methodology to refine the types of surgical patients and complications included in the measure and to expand the measure beyond surgical inpatients ( print page 69553) ( 86 FR 45390 through 45391 ). Other commenters suggested keeping CMS PSI 04 unchanged because of the importance of evaluating patient deaths when assessing patient safety and suggested adding more patient safety measures to the Hospital IQR Program measure set, expressing that there were too few patient safety measures in the program ( 86 FR 45391 ). After consideration of the public comments on our proposal to remove CMS PSI 04 in the FY 2022 IPPS/LTCH PPS proposed rule ( 86 FR 25579 through 25580 ) we decided not to finalize removal of the measure at that time.
Since then, we have developed the Thirty-Day Risk-Standardized Death Rate Among Surgical Inpatients with Complications (Failure-to-Rescue) (CBE #4125) measure, as proposed for adoption in section IX.C.5.e. of this final rule beginning with the FY 2027 payment determination. The Failure-to-Rescue measure is a more broadly applicable measure that would be more appropriate for inclusion in the Hospital IQR Program. Recent studies have indicated that the CMS PSI 04 measure does not consistently recognize preventable in-hospital deaths (failure to rescue cases). A 2023 study indicated that CMS PSI 04 is being used to an unknown extent outside of postoperative cases, and there is often erroneous categorization of patients as having a CMS PSI 04 complication. [ 676 ] This same study found significant variation in the identification of CMS PSI 04 complications at different procedure locations (For example: bedside versus operating room procedures). [ 677 ] Therefore, both the temporal and causal relationship attributing a CMS PSI 04 complication to patient mortality has been found to be poorly understood, particularly because CMS PSI 04 relates to a complication being deemed treatable. [ 678 ]
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36322 through 36324 ), we proposed to adopt the Failure-to-Rescue measure to replace CMS PSI 04 as a more broadly applicable patient safety indicator and one which can better address concerns previously raised by interested parties. The Failure-to-Rescue measure assesses the percentage of surgical inpatients who experienced a complication and then died within 30-days from the date of their first “operating room” procedure. We refer readers to section IX.C.5.e. of this final rule for more detail on the Failure-to-Rescue measure including the timeline for its initial performance, reporting, and payment determination periods.
While CMS PSI 04 only measures the rate of in-hospital deaths among surgical inpatients within a set of serious treatable conditions, the Failure-to-Rescue measure assesses the probability of death given a postoperative complication and is inclusive of a broader range of conditions commonly experienced by surgical inpatients. To best address the needs of a broader scope of surgical inpatients and conditions, it allows for more context-specific approaches to measure preventable deaths due to the highly variable nature of surgical procedures between specialties. This highly variable and context-specific nature of postoperative cases has been considered a challenge of using CMS PSI 04 as an effective universal patient safety metric. [ 679 ] There would be minimal burden for hospitals associated with replacing CMS PSI 04 with the Failure-to-Rescue measure due to the Failure-to Rescue measure's data sources, including its use of Medicare Advantage encounter data. Thus, the Failure-to-Rescue measure would include a wider range of patients and better reflect the true nature of postoperative patient safety at institutions. In addition, multiple failure-to-rescue measures have been repeatedly validated by their consistent association with nurse staffing, nursing skill mix, technological resources, rapid response systems, and other activities that improve early identification and prompt intervention when complications arise after surgery. [ 680 681 682 ]
By using the Failure-to-Rescue measure, hospitals can identify opportunities to improve their quality of care and patient safety. Hospitals and healthcare providers can benefit from knowing not only their institution's mortality rate, but also their institution's ability to provide each patient with the appropriate and necessary standard of care after an adverse occurrence. [ 683 ] Using the Failure-to-Rescue measure as opposed to the current CMS PSI 04 measure is especially important if the hospital resources needed for preventing and treating 30-day postoperative complications among surgical inpatients are different from those needed for targeted care after an adverse event, such as more skilled care personnel or equipment specific to postoperative care. From a quality improvement perspective, the Failure-to-Rescue measure rate would complement the mortality rate to improve our understanding of mortality statistics and identify opportunities for improvement. [ 684 ] Therefore, the quality-of-care measurement may be improved if both mortality and Failure-to-Rescue measure rates are reported instead of relying on the Hybrid HWM measure alone. Using the Failure-to-Rescue measure instead of the CMS PSI 04 measure would allow us to assess an expanded population and encourage safe practices for the widest range of surgical inpatients.
We proposed to remove the CMS PSI 04 measure from the Hospital IQR Program beginning with the FY 2027 payment determination associated with the performance period of July 1, 2023-June 30, 2025, contingent upon finalizing our proposal to adopt the Failure-to-Rescue measure beginning with the FY 2027 payment determination so that there is no gap in measuring this important topic area.
We invited public comment on our proposal to remove the CMS PSI 04 measure from the Hospital IQR Program beginning with the FY 2027 payment determination associated with the performance period of July 1, 2023-June 30, 2025, contingent upon finalizing our proposal to adopt the Failure-to-Rescue ( print page 69554) measure beginning with the FY 2027 payment determination.
Comment: Many commenters broadly supported the removal of the CMS PSI 04 measure. Several commenters supported the removal of the CMS PSI 04 measure contingent upon its replacement with the Failure-to-Rescue measure. A few commenters supported the measure removal, regardless of its replacement with a new measure. A few commenters supported the removal of CMS PSI 04 because they had concerns about the measure's validity and reliability. A commenter stated that the Failure-to-Rescue measure would be more reliable and valid because it includes Medicare Advantage and Medicare fee-for-service participants, as well as deaths after hospital discharge but within 30 days of the index operative procedure.
Response: We thank the commenters for their support. We agree with this feedback.
Comment: A few commenters supported removal of the CMS PSI 04 measure because they stated it is redundant with existing or proposed Hospital IQR Program measures.
Response: We thank commenters for their support. The Failure-to-Rescue measure would be complementary, rather than redundant, to the mortality measures used in the Hospital IQR Program and Hospital VBP Program. We refer readers to sections IX.C.8. and V.L.2. for more details on the previously adopted mortality measures in the Hospital IQR Program and Hospital VBP Program, respectively. Quality of care measurement would be improved through reporting both the Failure-to-Rescue measure and the mortality measures.
Comment: A commenter voiced support for the removal of CMS PSI 04, as well as the four payment-based measures. However, the commenter did not support the addition of eight measures for a net increase of three quality reporting measures.
Response: We thank the commenter for their support. While we understand that there are times when quality reporting poses challenges for hospitals, we view quality-of-care measurement as an essential aspect of improving clinical outcomes and encouraging safe practices for a wide range of surgical inpatients.
Comment: A few commenters raised concerns about removing CMS PSI 04 and replacing it with the Failure-to-Rescue measure. A commenter mentioned that the change from in-hospital deaths to 30-day mortality increases the risk of skewing the data with mortalities that are unrelated to hospital complications. Another commenter stated that an alternate measure must account for the most vulnerable patients since the proposed replacement measure, the Failure-to-Rescue measure, excludes these patients from consideration.
Response: We thank the commenters for their input. Recent studies have indicated that the CMS PSI 04 measure does not consistently recognize preventable in-hospital deaths. A 2023 study indicated that the CMS PSI 04 measure is used outside of postoperative cases, and patients are often erroneously categorized as having a PSI 04 complication. [ 685 ] We understand the Failure-to-Rescue measure is a more broadly applicable patient safety indicator than the CMS PSI 04 measure in that it assesses the probability of death given a postoperative complication. It is not necessarily intended to attribute a causal relationship between the complication and death; rather, it would allow patients to compare hospitals and determine the nature of postoperative patient safety at institutions on a global level. In contrast to the CMS PSI 04 measure, the Failure-to-Rescue measure excludes patients whose relevant complications preceded (rather than followed) their first inpatient operating room procedure. It also limits the patients assessed to the general surgical, vascular, and orthopedic Medicare Severity Diagnosis Related Groups (MS-DRGs). These limitations were implemented in response to specific concerns with CMS PSI 04—namely, that the cohort of patients assessed was too heterogenous and included those who had undergone very high-risk and very low-risk surgeries. In contrast, the exclusion criteria of the Failure-to-Rescue measure would allow prospective patients to make a truer comparison when evaluating hospitals for postoperative safety. The measure's risk-standardization process appropriately controls for factors impacting patients' clinical care that are beyond the hospital's control. We note that the complications in the Failure-to-Rescue measure are all serious adverse health events. We will monitor the measure's effects on the provision of clinical care to avoid any unintended consequences of its adoption.
Comment: A commenter only supported the removal of the CMS PSI 04 measure if there was no gap in publicly reporting performance between this measure and the implementation of the Failure-to-Rescue measure. The commenter noted that more than 500 people die every day due to hospital errors; therefore, it is critical that no day goes by without reporting on hospital mortality.
Response: We appreciate and agree with the commenter's input. We would like to clarify that there would be no gap in public reporting between the removal of the CMS PSI 04 measure and the implementation of the Failure-to-Rescue measure. The CMS PSI 04 measure would be removed beginning with the July 1, 2023-June 30, 2025, reporting period/FY 2027 payment determination, and the Failure-to-Rescue measure would be adopted beginning with the July 1, 2023-June 30, 2025, reporting period/FY 2027 payment determination. Therefore, no gap in reporting would exist.
After consideration of the public comments received, we are finalizing the removal of CMS PSI 04 as proposed beginning with the July 1, 2023-June 30, 2025, reporting period/FY 2027 payment determination.
We proposed to remove four clinical episode-based payment measures from the Hospital IQR Program beginning with the FY 2026 payment determination ( 89 FR 36326 through 36392 ):
- Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode of Care for Acute Myocardial Infarction (AMI) (CBE #2431) (AMI Payment) (adopted at 78 FR 50802 through 50805 ). This measure assesses hospital risk-standardized payment associated with a 30-day episode-of-care for acute myocardial infarction for Medicare FFS patients aged 65 or older for any hospital participating in the Hospital IQR Program;
- Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode of Care for Heart Failure (HF) (CBE #2436) (HF Payment) (adopted at 79 FR 50231 through 50235 ). This measure assesses hospital risk-standardized payment associated with a 30-day episode-of-care for heart failure for Medicare FFS patients aged 65 or older for any hospital participating in the Hospital IQR Program;
- Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode of Care for Pneumonia (PN) (CBE #2579) (PN Payment) (adopted at 79 FR 50227 through 50231 ). This measure assesses hospital risk- ( print page 69555) standardized payment associated with a 30-day episode-of-care for pneumonia for any hospital participating in the Hospital IQR Program and includes Medicare FFS patients aged 65 or older; and
- Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode of Care for Elective Primary Total Hip Arthroplasty (THA) and/or Total Knee Arthroplasty (TKA) (CBE #3474) (THA/TKA Payment) (adopted at 80 FR 49674 through 49680 ; revised at 87 FR 49267 through 49269 ). This measure assesses hospital risk-standardized payment (including payments made by CMS, patients, and other insurers) associated with a 90-day episode-of-care for elective primary THA/TKA for any hospital participating in the Hospital IQR Program and includes Medicare FFS patients aged 65 or older.
The final performance periods for these four payment measures are indicated in the following table:

We proposed to remove the AMI Payment, HF Payment, PN Payment, and THA/TKA Payment measures under measure removal Factor 3, codified at 42 CFR 412.140(g)(3)(i)(C) , the availability of a more broadly applicable measure (across settings, populations, or the availability of a measure that is more proximal in time to desired patient outcomes for the particular topic)—specifically, the Medicare Spending Per Beneficiary Hospital measure (CBE #2158) (MSPB Hospital measure) in the Hospital VBP Program ( 89 FR 36326 ). [ 686 ] The MSPB Hospital measure has been intermittently included in the Hospital IQR Program's measure set, most recently to update the measure specifications in the Hospital VBP Program. The Hospital VBP Program's statute requires that measures be publicly reported for one year in the Hospital IQR Program prior to the beginning of the performance period in the Hospital VBP Program (section 1886(o)(2)(B)(ii) of the Act and 42 CFR 412.164(b) ). [ 687 ] In the FY 2023 IPPS/LTCH PPS final rule, we re-adopted the previously removed MSPB Hospital measure into the Hospital IQR Program with refinements ( 87 FR 28529 through 28532 ) to update the measure specifications for purposes of the Hospital VBP Program. We subsequently removed it again from the Hospital IQR Program and concurrently adopted the refined version into the Hospital VBP Program ( 88 FR 59064 through 59067 , 59170 through 59171 , respectively). We refer readers to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49257 through 49263 ) for more details on this measure's history in the Hospital IQR and Hospital VBP Programs.
The MSPB Hospital measure evaluates hospitals' efficiency and resource use relative to the efficiency of the national median hospital. The MSPB Hospital measure is a more broadly applicable measure because it captures the same data as the four clinical episode-based payment measures proposed for removal but incorporates a much larger set of conditions and procedures. We note that we recently adopted refinements to the MSPB Hospital measure to ensure a more comprehensive and consistent assessment of hospital performance ( 87 FR 49257 through 49263 , 88 FR 59064 through 59067 ). Those refinements allow the measure to capture more episodes and adjusted the measure calculation. [ 688 ]
The four clinical episode-based payment measures proposed for removal are condition-specific whereas the MSPB Hospital measure is not. Although the MSPB Hospital measure does not provide the same level of granularity as the four condition-specific measures, the important data elements would be captured more broadly under the Hospital VBP Program by evaluating and publicly reporting the hospitals' efficiency relative to the efficiency of the median national hospital. Specifically, the MSPB Hospital measure assesses the cost to Medicare for services performed by hospitals and other healthcare providers during an episode of care, which includes the three days prior to, during, and 30 days following an inpatient's hospital stay. [ 689 ] Additionally, providers would continue to receive confidential feedback reports containing details on the MSPB Hospital measure.
We note that performance on these four clinical episode-based payment measures has either remained stable or decreased since FY 2019. Based on an internal CMS analysis, the mean performance for the PN Payment, HF Payment, and AMI Payment measures ( print page 69556) has decreased, while the mean performance for the THA/TKA Payment measure has remained stable. Considering these performance trends, we highlight that these four clinical episode-based payment measures have not been as beneficial in recent years to the Hospital IQR Program.
We invited public comment on our proposal to remove these four clinical episode-based payment measures from the Hospital IQR Program beginning with the FY 2026 payment determination.
Comment: Several commenters expressed general support for the proposal to remove the four clinical episode-based payment measures. Many commenters agreed that the existing and proposed new Hospital IQR Program measures are more broadly applicable and that retaining these four measures would result in program measure redundancies. Several commenters expressed support contingent upon replacement measures as outlined in the proposed rule. A few commenters supported CMS's proposal and noted that removal of the four payment measures aligns with the National Quality Strategy and Meaningful Measures Framework, enabling hospitals to prioritize patient care and quality improvement initiatives more effectively. A few commenters appreciated that removing these four measures would reduce the administrative impact of quality measure reporting and allow hospitals to meet reporting requirements more efficiently. A commenter suggested CMS consider the administrative impact of quality measure reporting and remove measures that are redundant or no longer needed. A commenter noted that removing these measures would allow for other measures to be added in the future.
Response: We thank the commenters for their support. We agree that the MSPB Hospital measure is a more broadly applicable measure such that removing these four clinical episode-based payment measures would enable hospitals to more efficiently report on, and work toward, improved quality of care. We note that we continually assess the Hospital IQR Program's measure set and appreciate feedback regarding removal of measures. We will continue engaging with interested parties through education and outreach opportunities for any feedback about suggested additional measure removals in the future.
Comment: A commenter supported CMS's proposal to remove the four payment measures and stated that the data produced by these measures were difficult to interpret or act upon for quality improvement purposes. A commenter specifically supported CMS's proposal to remove the AMI Payment and HF Payment measures, expressing concern about whether they had been appropriately risk adjusted.
Response: We thank the commenters for their support and appreciate their feedback regarding their perceived utility as well as the lack of risk adjustment for these measures.
Comment: A few commenters did not support removal of the four clinical episode-based payment measures and expressed concern about adequacy of the MSPB Hospital measure as a replacement. Specifically, commenters expressed concern about whether the MSPB Hospital measure considers patient risk factors, that it does not include non-Medicare costs, and that it lacks granular detail which the four procedure- or condition-based payment measures provide.
Response: We thank the commenters for their feedback, but we respectfully disagree. The MSPB Hospital measure is an adequate replacement for the four clinical episode-based payment measures because it captures the same data as the four clinical episode-based payment measures proposed for removal and incorporates a much larger set of conditions and procedures. The MSPB Hospital measure's risk adjustment methodology also adjusts for several factors, including patient age and severity of illness. We note that we recently adopted refinements to the MSPB Hospital measure to ensure a more comprehensive and consistent assessment of hospital performance ( 87 FR 49257 through 49263 , 88 FR 59064 through 59067 ). Those refinements allow the measure to capture more episodes and adjust the measure calculation. Although the MSPB Hospital measure does not provide the same level of granularity as the four condition-specific measures, the important data elements would be captured more broadly under the Hospital VBP Program by evaluating and publicly reporting the hospitals' efficiency relative to the efficiency of the median national hospital. Removing the four clinical episode-based payment measures helps ensure that we are moving the Hospital IQR Program forward in the least burdensome manner possible while continuing to encourage improvement in the quality of care provided to patients. We note that hospitals have access to patient-level information through confidential hospital-specific reports for the MSPB Hospital measure.
Comment: A few commenters recommended that CMS retain all four clinical episode-based payment measures and stated that they provide valuable information for consumers assessing where to pursue care. A commenter specifically did not support removal of the AMI Payment and HF Payment measures, noting that these payment measures, when directly linked with clinical quality measures, help interested parties assess whether reducing costs leads to improved patient outcomes.
Response: We thank the commenters for their input. While we agree that these measures have provided valuable information in the past, we note that performance trends on these four clinical episode-based payment measures indicate that they had not been as beneficial in recent years to the Hospital IQR Program. In alignment with our ongoing effort to implement a more parsimonious measure set while continuing to incentivize improvement in the quality of care provided to patients, we concluded it is appropriate to remove these four clinical episode-based payment measures at this time, resulting in a streamlined set of the most meaningful measures.
Comment: A commenter specifically did not support removal of the PN Payment measure, stating that pneumonia often results from a preventable airborne infection and the measure should remain in the Hospital IQR Program.
Response: While we appreciate the commenter's concern, the PN Payment measure is not currently incentivizing prevention of pneumonia. We do agree that pneumonia is an important topic for quality measurement and note that the Hospital Readmissions Reduction Program includes an unplanned readmission measure for Pneumonia (Hospital 30-day, All-Cause, Risk-Standardized Readmission Rate (RSRR) Following Pneumonia Hospitalization (NQF #0506), 76 FR 51666 and 51667 ) and that the Hospital VBP Program includes the Hospital 30-Day, All-Cause, Risk-Standardized Mortality Rate Following Pneumonia Hospitalization measure.
After consideration of the public comments we received, we are finalizing our proposal to remove the four clinical episode-based payment measures beginning with the FY 2026 payment determination.
We proposed refinements to two measures currently in the Hospital IQR Program measure set: (1) Global ( print page 69557) Malnutrition Composite Score (GMCS) eCQM, beginning with the CY 2026 reporting period/FY 2028 payment determination and for subsequent years, and (2) the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) Survey measure beginning with the CY 2025 reporting period/FY 2027 payment determination. We provide more details on modifications to the GMCS eCQM in the subsequent sections and details on the modification to HCAHPS Survey measure are in section IX.B.2.e. of this final rule.
The previously finalized GMCS eCQM (CBE #3592e) assesses the percentage of hospitalizations for adults 65 years old and older prior to the start of the measurement period with a length of stay equal to or greater than 24 hours who received optimal malnutrition care during the current inpatient hospitalizations where care performed was appropriate to the patient's level of malnutrition risk and severity. We adopted the GMCS eCQM in the FY 2023 IPPS/LTCH PPS final rule beginning with the CY 2024 reporting period/FY 2026 payment determination ( 87 FR 49239 through 49246 ). We refer readers to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49241 through 49242 ) for more detailed discussion of the CBE review and endorsement of the current GMCS eCQM, which received CBE endorsement in July 2021 (CBE #3592e). [ 690 ] [ 691 ]
While we understand the unique challenges malnutrition creates for older adults, we also recognize that hospital and disease-related malnutrition is not limited to that population ( 87 FR 49239 ). Data from the Agency for Healthcare Research and Quality (AHRQ) indicate that approximately eight percent of all hospitalized adults have a diagnosis of malnutrition, [ 692 ] and additional research finds that malnutrition and malnutrition risk can be found in 20 to 50 percent of hospitalized adults 18 years old and older. [ 693 ] Failure to diagnose and insufficient treatment of malnutrition in hospitals is also associated with poor institutional coordination between nurses, physicians, and other hospital staff regarding screening, diagnosis, and treatment, further emphasizing the need to address malnutrition in all hospitalized adults. [ 694 ] Because malnutrition impacts adults of all ages, preventive screening and intervention among all hospitalized adults 18 years old and older would greatly reduce the risk and improve the treatment of malnutrition. [ 695 ] A 2020 study estimated that every dollar spent on nutrition interventions in a hospital setting can result in up to $99 in savings on subsequent medical care. [ 696 ] Screening all patients over age 18 for malnutrition instead of only those over age 65 could result in both improved clinical outcomes for patients and substantial financial savings for the healthcare system.
We, therefore, proposed modifications to the GMCS eCQM to expand the applicable population from hospitalized adults 65 or older to hospitalized adults 18 or older in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36327 through 36329 ). The modified GMCS eCQM would broaden the measure to assess hospitalized adults 18 years old and older who received care appropriate to their level of malnutrition risk and malnutrition diagnosis, if properly identified.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49239 ), we noted that the adoption of a malnutrition measure may help address several priority areas identified in the CMS Framework for Health Equity [ 697 ] ( 87 FR 49240 through 49241 ) and expanding the current measure's population to include all adults over 18 years old would further address these priorities. Malnutrition in the U.S., whether caused by challenges from disease and functional limitations, food insecurity, other factors, or a combination of causes, is more frequently experienced by underserved populations and can thus be a contributing factor to health inequities. [ 698 ] Adopting the updated measure as proposed would lead to a more diverse population being assessed for malnutrition, and by identifying instances of malnutrition among younger populations, the benefits of proper nutrition could be felt over a lifetime. As part of the CMS National Quality Strategy, the modified GMCS eCQM would also address the priority area of “Promote Aligned and Improved Health Outcomes.” [ 699 ] Under the CMS Meaningful Measures 2.0 Initiative, which is a key component of the CMS National Quality Strategy, the modified GMCS eCQM addresses the quality priorities of “Seamless Care Coordination,” “Person-Centered Care,” and “Equity.” It would address these priorities by connecting providers at different levels of care to ensure the largest possible population of adult patients with in-hospital malnutrition are identified and treated using a patient-centered approach.
The modified GMCS eCQM still includes the four component measures corresponding to documented best practices as described in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49241 ) and in the first column of Table IX.C.4. The only change we proposed is to expand the applicable population for this measure. The measure specifications for the modified GMCS eCQM can be found on the eCQI Resource Center website, available at: https://ecqi.healthit.gov/ecqm/eh/2024/cms0986v2 . ( print page 69558)
We refer readers to the proposed Patient Safety Structural measure in section IX.B.1.c. of this final rule for details on the PRMR process including the voting procedures used to reach consensus on measure recommendations. The PRMR Hospital Committee met on January 18-19, 2024, to review measures included by the Secretary on a publicly available “2023 Measures Under Consideration List” (MUC List), [ 700 701 ] including the modified GMCS eCQM (MUC2023-114), to vote on a recommendation regarding use of this measure. [ 702 703 ]
The PRMR Hospital Committee reached consensus and recommended including this measure (MUC2023-114) in the Hospital IQR Program with conditions. Fourteen members of the group recommended adopting the measure into the Hospital IQR Program without conditions; three members recommended adoption with conditions; two committee members voted not to recommend the measure for adoption. Taken together, 84.2 percent of the votes were recommended with conditions. [ 704 ] The three members who voted to adopt with conditions specified the condition as screening and assessment includes hospital-acquired malnutrition and high-risk nutritional practices in hospitals, such as prolonged fasting for rescheduled procedures, and to obtain more feedback from patient groups. We agree that the potential for unintended consequences exists and note that we consistently monitor all the measures in the Hospital IQR Program for unintended consequences. Furthermore, we note that under our previously finalized measure removal Factor 6, codified at 42 CFR 412.140(g)(3)(i)(F) , collection or public reporting of a measure leads to negative unintended consequences other than patient harm, if we were to identify unintended consequences related to this measure, we would consider it for removal.
We refer readers to section IX.B.1.c. of the preamble of this final rule for details on the EM process including the measure evaluation procedures the EM Committees, comprised of the EM Advisory Group and EM Recommendation Group, uses to evaluate measures and whether they meet endorsement criteria. The GMCS eCQM was initially endorsed in the Fall 2020 cycle by the CBE (CBE #3592e) and is scheduled for endorsement review with the proposed modification in 2024. [ 705 ] Section 1886(b)(3)(B)(viii)(IX)(aa) of the Act requires that measures specified by the Secretary for use in the Hospital IQR Program be endorsed by the entity with a contract under section 1890(a) of the Act. Section 1886(b)(3)(B)(viii)(IX)(bb) of the Act states that in the case of a specified area or medical topic determined appropriate by the Secretary for which a feasible and practical measure has not been endorsed by the entity with a contract under section 1890(a) of the Act, the Secretary may specify a measure that is not so endorsed as long as due consideration is given to measures that have been endorsed or adopted by a consensus organization identified by the Secretary. After reviewing the modified measure, we found no measures, other than the proposed GMCS measure, on this topic. We determined this was an appropriate medical topic for us to propose the adoption of an unendorsed measure because of its general consistency with the current, endorsed measure, and the usefulness of the measure would be substantially improved by the modification.
The modified GMCS eCQM would still use data collected through hospitals' EHRs. The measure is designed to be calculated by the hospitals' CEHRT using the patient-level data and then submitted by hospitals to CMS.
The modified GMCS eCQM continues to consist of four component measures, which are first scored separately. [ 706 707 ] The overall composite score is derived from averaging the individual performance scores of the four component measures. The malnutrition component measures are all fully specified for use in EHRs. Table IX.C.4 describes each of the four measure components with the expanded population.

The modified GMCS eCQM numerator is comprised of the four component measures, that are individually scored for patients 18 years old and older who are admitted to an acute inpatient hospital. The measure denominator is the composite, or total, of the four component measures for patients 18 years old and older who are admitted to an acute inpatient hospital. The only exclusion for this measure population remains as patients whose length of stay is less than 24 hours, the same as previously adopted in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49244 ).
Each measure component is a proportion with a possible performance score of 0 to 100 percent (higher percent reflects better performance). After each component score is calculated individually, an unweighted average of all four scores is computed to determine the final composite score for the individual with a total score ranging from 0 to 100 percent (higher percent reflects better performance). [ 708 ]
We proposed the adoption of the modified GMCS eCQM as part of the Hospital IQR Program measure set from which hospitals can self-select beginning with the CY 2026 reporting period/FY 2028 payment determination. Since this modification uses the same data sources and collection methods as the current version of the GMCS eCQM, there is not expected to be any major impact to workflows or other aspects of data collection. The only anticipated change to data collection processes is that the data would be collected from a larger patient population. We refer readers to section IX.C.9.c. of this final rule for our previously finalized eCQM reporting and submission requirements, as well as proposed modifications for these requirements.
We also refer readers to section IX.F.6.a.(2). of the preamble of this final rule for discussion of a similar adoption of this measure in the Medicare Promoting Interoperability Program.
We invited public comment on our proposal to modify the GMCS eCQM to expand the applicable population from hospitalized adults 65 years old or older to hospitalized adults 18 years old or older beginning with the CY 2026 reporting period/FY 2028 payment determination.
Comment: Many commenters supported the proposal to expand the patient population for the GMCS eCQM. Many commenters supported it because they stated that malnutrition has a significant impact on patient outcomes and that this expansion would lead to valuable data, improved patient outcomes for a broader range of patients, and a more comprehensive understanding of malnutrition in the adult population.
Response: We thank the commenters for their support and agree with their feedback.
Comment: A few commenters supported the proposal to expand the patient population for the GMCS eCQM and emphasized that this modification, in addition to enhancing the quality of care for patients with malnutrition, has the potential to advance CMS's goal of reducing health disparities.
Response: We appreciate and agree with the commenters' perspective. As we noted in the FY 2025 IPPS/LTCH PPS proposed rule, malnutrition in the U.S., whether caused by challenges from disease and functional limitations, food insecurity, other factors, or a combination of causes, is more frequently experienced by underserved populations and can thus be a contributing factor to health disparities ( 89 FR 36328 ). Expanding the population for the GMCS eCQM would ensure the largest possible population of adult patients with in-hospital malnutrition are identified and treated.
Comment: A commenter appreciated that in this proposal, CMS acknowledged the valuable work of registered dieticians in addressing malnutrition and improving patient care outcomes.
Response: We thank the commenter for their input. Nutrition screening is an important aspect of a patient's health, and it is the responsibility of all clinicians to support appropriate nutrition, particularly in inpatient settings ( 87 FR 49244 through 49245 ).
Comment: Many commenters supported the proposal and recommended that CMS accelerate implementation of the expansion from CY 2026 to CY 2025 to encourage hospitals to provide high-quality malnutrition care to all adults. Many commenters who supported accelerating the proposal's implementation noted that the modification to the measure would not impose a burden increase on providers.
Response: We thank the commenters for their feedback; however, in determining the proposed implementation timeline for the GMCS eCQM we considered how this timeline allows hospitals time to review the results of the first full year of reporting on the GMCS eCQM before implementation of the modification. This does not preclude organizations from engaging in preventive screening and intervention for malnutrition in patients 18 years of age and older. We encourage commenters to engage in this work, which could result in both improved clinical outcomes and substantial financial savings on subsequent medical care for hospital systems. [ 709 ]
Comment: A commenter recommended that CMS align the GMCS eCQM logic with that of other eCQMs, such that the record is included only if the encounter ends during the measurement period. A few commenters suggested that CMS should further refine GMCS to better capture changes in nutritional status during an inpatient stay because they state that malnutrition may arise during a hospitalization. Another commenter recommended that the measure steward be more flexible for rapid cycle improvements of the measure logic that allow for the measure to function as intended.
Response: We appreciate the input from the commenters. We will continue to evaluate the appropriateness of refinements to the GMCS eCQM, including possible adjustments to the inclusion criteria. In addition, we continually assess the Hospital IQR Program's measure set and will take this feedback into consideration.
Comment: A commenter recommended that CMS perform additional feasibility assessments on this measure across a broader set of EHR vendors and hospitals and suggested that the proposed refinement be reviewed and approved by the CBE.
Response: We appreciate the commenter's input regarding feasibility assessments and CBE endorsement. We emphasize that eCQMs, like all other types of quality measures in the Hospital IQR Program, undergo rigorous testing during the measure development process for feasibility, validity, and reliability. As we discussed in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49241 through 49242 ), the measure developer conducted additional testing after the measure was initially reviewed by the CBE, and the testing results demonstrated that the four component measures were usable for identifying key improvement areas in malnutrition care. A subsequent test with additional hospitals showed that the component measures could be implemented in a cohort of diverse hospitals and lead to meaningful improvements in measure performance as all four components were significantly associated with improved outcomes for 30-day readmissions. Notably, the GMCS eCQM is endorsed by the CBE and the modified GMCS eCQM is scheduled for endorsement review in the fall of 2024.
Comment: A commenter did not support the proposed modification to the GMCS eCQM because they stated it would create an undue burden on the hospital and would not adequately address the root cause of malnutrition. The commenter suggested that CMS does not compensate hospitals sufficiently to help address root causes of malnutrition. A commenter expressed concern that hospitals lack the resources to screen and refer the expanded population appropriately.
Response: We appreciate this feedback, and we remind the commenter that reporting on the GMCS eCQM is not required under the current eCQM reporting requirements, as hospitals may self-select to report on this eCQM. Furthermore, the modification to the GMCS eCQM would use the same data sources and collection methods as the current version of the GMCS eCQM. Therefore, no major impact on workflows or data collection is expected. The only change is that the data would be collected from a larger patient population. A 2020 study estimated that every dollar spent on nutrition interventions in a hospital setting can result in up to $99 in savings on subsequent medical care. [ 710 ] Therefore, we believe that the measure can help address the root cause of malnutrition. While we understand the commenter's concern about hospitals' resources to screen and refer the expanded population, we reiterate that the GMCS eCQM is not required to be reported. We encourage hospitals to consider reporting this eCQM, as it addresses an important clinical topic.
Comment: A commenter did not support modifying the GMCS eCQM because they state it would overlap with the Screening for Social Drivers of Health measure and should instead focus on implementation of that measure to create interoperable data.
Response: The GMCS eCQM and the Screening for Social Drivers of Health measure, while topically related, are not duplicative. The Screening for Social Drivers of Health measure and the GMCS eCQM both address nutrition as a driver of health because it is an important contributor to a healthy population, but they address different goals ( 87 FR 49245 ). While the Screening for Social Drivers of Health measure incentivizes the screening and identifying of patients for food insecurity, the GMCS eCQM focuses on screening for malnutrition risk (of which food insecurity may be a contributing factor), but also the performance of a nutrition assessment and development of a care plan for identified malnourished patients ( 87 FR 49245 ). The GMCS eCQM and the Screening for Social Drivers of Health measure are complementary to one another but are not duplicative as they measure different aspects of the quality care processes ( 87 FR 49245 ). We thank the commenter for their recommendation regarding interoperable data for the Screening for Social Drivers of Health measure and will take it into consideration as we assess the Hospital IQR Program's measure set.
After consideration of the public comments received, we are finalizing the GMCS eCQM measure modification as proposed beginning with the CY 2026 reporting period/FY 2028 payment determination. We also refer readers to section IX.F.6.a.(2). of the preamble of this final rule for discussion of adoption of this measure in the Medicare Promoting Interoperability Program.
This table summarizes the previously finalized Hospital IQR Program measure set for the FY 2026 payment determination updated to reflect the removals of four claims-based payment measures:

We refer readers to the CY 2025 OPPS/ASC proposed rule where we are proposing to continue voluntary reporting of the core clinical data elements (CCDEs) and linking variables for both the Hybrid Hospital-Wide Readmission (HWR) and Hybrid Hospital-Wide Standardized Mortality (HWM) measures, for the performance ( print page 69562) period of July 1, 2023 through June 30, 2024, impacting the FY 2026 payment determination for the Hospital IQR Program ( 89 FR 59500 through 59502 ).
This table summarizes the previously finalized and newly finalized Hospital IQR Program measure set for the FY 2027 payment determination including the adoption of two new structural measures, one new claims-based patient safety measure, and the removal of the CMS PSI 04 measure:

This table summarizes the previously finalized and newly finalized Hospital IQR Program measure set for the FY 2028 payment determination including the adoption of two new Hospital Harm eCQMs, two new NHSN measures, modification of the GMCS eCQM, and the modification of the HCAHPS Survey measure:

This table summarizes the previously finalized and newly finalized Hospital IQR Program measure set for the FY 2029 payment determination and for subsequent years:

We proposed changes to our reporting and submission requirements for eCQMs. There are no proposed changes to the following requirements, and thus have been omitted from the Form, Manner, and Timing of Quality Data Submission section: procedural requirements; data submission requirements for chart-abstracted measures; data submission and reporting requirements for hybrid measures; sampling and case thresholds for chart-abstracted measures; HCAHPS Survey administration and submission requirements; data submission requirements for structural measures; data submission and reporting requirements for CDC NHSN measures; and data submission and reporting requirements for Patient-Reported Outcome-Based Performance Measures (PRO-PMs). We refer readers to the QualityNet website at: https://qualitynet.cms.gov/inpatient/iqr (or other successor CMS designated websites) for more details on the Hospital IQR Program data submission and procedural requirements.
Section 1886(b)(3)(B)(viii)(I) and (b)(3)(B)(viii)(II) of the Act state that the applicable percentage increase for FY 2015 and each subsequent year shall be reduced by one-quarter of such applicable percentage increase (determined without regard to sections 1886(b)(3)(B)(ix), (xi), or (xii) of the Act) for any subsection (d) hospital that does not submit data required to be submitted on measures specified by the Secretary in a form and manner and at a time specified by the Secretary. To successfully participate in the Hospital IQR Program, hospitals must meet specific procedural, data collection, submission, and validation requirements.
Section 412.140(c)(1) of title 42 of the Code of Federal Regulations generally requires that a subsection (d) hospital participating in the Hospital IQR Program must submit to CMS data on measures selected under section 1886(b)(3)(B)(viii) of the Act in a form and manner, and at a time, specified by CMS. The data submission requirements, specifications manual, measure methodology reports, and submission deadlines are posted on the QualityNet website at: https://qualitynet.cms.gov (or other successor CMS designated websites). The CMS Annual Update for the Hospital Quality Reporting Programs (Annual Update) contains the technical specifications for eCQMs. The Annual Update contains updated measure specifications for the year prior to the reporting period. For example, for the CY 2024 reporting period/FY 2026 payment determination, hospitals are collecting and would submit eCQM data using the May 2023 Annual Update and any applicable addenda. The Annual Update and implementation guidance documents are available on the Electronic Clinical Quality Improvement (eCQI) Resource Center website at: https://ecqi.healthit.gov/ .
Hospitals must register and submit quality data through the HQR System (previously referred to as the QualityNet Secure Portal) ( 42 CFR 412.140(a) ). The HQR System is safeguarded in accordance with the HIPAA Privacy and Security Rules to protect submitted patient information. See 45 CFR parts 160 and 164, subparts A, C, and E.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36336 through 36339 ), we proposed a progressive increase in the number of mandatory eCQMs a hospital must report beginning with the CY 2026 reporting period/FY 2028 payment determination. We did not propose any changes to the current eCQM reporting or submission requirements for the CY 2024 reporting period/FY 2026 payment determination or the CY 2025 reporting period/FY 2027 payment determination. We provide additional detail in our proposal later in this section of the preamble.
We began requiring hospitals to report on eCQMs in the CY 2016 reporting period, with a goal of progressively increasing the number of eCQMs ( print page 69569) hospitals are required to report in the Hospital IQR Program while also being responsive to hospitals' concerns about timing, readiness, and burden associated with the increased number of measures ( 80 FR 49693 through 49698 , and 81 FR 57150 through 57157 ). To allow hospitals and their vendors time to gain experience with reporting eCQMs we gradually increased the number of eCQMs on which hospitals were required to report over the course of several years. We required hospitals to report on certain specific eCQMs that we prioritized while retaining an element of choice by allowing hospitals to self-select some eCQMs. We also gradually increased the number of reporting quarters to improve measure reliability for public reporting of performance information ( 84 FR 42503 through 42505 , 85 FR 58932 through 58939 , 86 FR 45418 , and 87 FR 49299 through 49302 ).
Under previously adopted eCQM reporting policies, hospitals must report four calendar quarters of data for each required eCQM: (1) the Safe Use of Opioids—Concurrent Prescribing eCQM; (2) the Cesarean Birth eCQM; (3) the Severe Obstetric Complications eCQM; and (4) three self-selected eCQMs; for a total of six eCQMs for the CY 2024 reporting period/FY 2026 payment determination and subsequent years ( 85 FR 58932 through 58939 , 86 FR 45418 , and 87 FR 49298 through 49302 ). We refer readers to the QualityNet website for additional information on previous reporting and submission requirements policies for eCQMs at: https://qualitynet.cms.gov/inpatient/measures/ecqm (or other successor CMS designated websites).
In the CY 2024 Medicare Physician Fee Schedule (PFS) final rule ( 88 FR 79307 through 79312 ), we finalized the revisions to the definition of CEHRT for the Medicare Promoting Interoperability Program at 42 CFR 495.4 . Specifically, we finalized the addition of a reference to the revised name of “Base Electronic Health Record (EHR) definition,” proposed in the Health Data, Technology, and Interoperability: Certification Program Updates, Algorithm Transparency, and Information Sharing (HTI-1) proposed rule ( 88 FR 23759 , 23905 ), to ensure, if the HTI-1 proposals were finalized, the revised name of “Base EHR definition” would be applicable for the CEHRT definitions going forward ( 88 FR 79309 through 79312 ). We also finalized the replacement of our references to the “2015 Edition health IT certification criteria” with “ONC health IT certification criteria,” and the addition of the regulatory citation for ONC health IT certification criteria in 45 CFR 170.315 . We finalized the proposal to specify that technology meeting the CEHRT definition must meet ONC's health IT certification criteria “as adopted and updated in 45 CFR 170.315 ” ( 88 FR 79553 ). This approach is consistent with the definitions and approach subsequently finalized in ONC's HTI-1 final rule, which appeared in the Federal Register on January 9, 2024 ( 89 FR 1205 through 1210 ). For additional background and information on this update, we refer readers to the discussion in the CY 2024 PFS final rule on this topic ( 88 FR 79307 through 79312 ).
Increasing the number of mandatory eCQMs, specifically to include the five previously adopted Hospital Harm eCQMs, would support our re-commitment to better safety practices for both patients and healthcare workers to save lives from preventable harms. [ 711 ] Proposing mandatory reporting of these Hospital Harms eCQMs are a part of our initial actions in responding and joining the President's Council of Advisors on Science and Technology (PCAST) call to action to renew “our nation's commitment to improving patient safety.” [ 712 ] We refer readers to section IX.B.1. for more details on other efforts toward better patient and healthcare worker safety practices and the Patient Safety Structural measure for the Hospital IQR and PCHQR Programs.
We developed our proposal to also align with CMS' National Quality Strategy priority area of “Patient Safety and Resiliency,” that seeks to “improve performance on key patient safety metrics through the applications of CMS levers such as quality measurement, payment, health and safety standards, and quality improvement support.” [ 713 ] It is important to more comprehensively collect data on these measures from all hospitals participating in the Hospital IQR and Medicare Promoting Interoperability Programs instead of limiting data collection to just those hospitals that chose to report it. Capturing this important quality information is crucial to improve surveillance on safety metrics in hospitals and support the CMS National Quality Strategy target success goal of reducing preventable harm. [ 714 ] Additionally, the proposal was developed in alignment with the “Interoperability” goal outlined in the National Quality Strategy that eCQMs use standard and interoperable data requirements that are less burdensome than other types of measures. By increasing the number of required eCQMs, and prioritizing the measures focused on preventable hospital harms, we are progressing towards our goal of using all digital measures. Thus, we proposed to increase the number of mandatory eCQMs over a two-year period to ultimately require reporting on five additional eCQMs ( 89 FR 36336 through 36339 ).
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36336 through 36339 ), beginning with the CY 2026 reporting period/FY 2028 payment determination, we proposed to modify the eCQM reporting and submission requirements to require hospitals to report on the following three eCQMs in addition to the existing eCQMs: (1) Hospital Harm—Severe Hypoglycemia eCQM; (2) Hospital Harm—Severe Hyperglycemia eCQM; and (3) Hospital Harm—Opioid-Related Adverse Events eCQM. This proposal would require hospitals to report four calendar quarters of data for a total of nine eCQMs (six specified eCQMs and three self-selected eCQMs).
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36336 through 36339 ), beginning with the CY 2027 reporting period/FY 2029 payment determination, we proposed to modify the eCQM reporting and submission requirements to require hospitals to report on the following two eCQMs in addition to the eCQMs proposed for the CY 2026 reporting period/FY 2028 ( print page 69570) payment determination: (1) Hospital Harm—Pressure Injury eCQM; and (2) Hospital Harm—Acute Kidney Injury eCQM. This proposal would require hospitals to report four calendar quarters of data for a total of eleven eCQMs (eight specified eCQMs and three self-selected eCQMs).
We proposed this stepwise approach to increasing the number of required eCQMs in response to public comments noting the burden and resources necessary to implement new eCQMs ( 88 FR 59145 through 59149 , and 88 FR 59149 through 59154 ), while also balancing the need to prioritize more comprehensive reporting on important safety and preventable harm metrics. Waiting until the CY 2027 reporting period/FY 2029 payment determination to require that hospitals report on these two Hospital Harm eCQMs would allow hospitals to experience 2 years of self-selecting to report on these relatively new eCQMs and build the infrastructure necessary to report these measures ( 88 FR 59145 through 59149 , and 88 FR 59149 through 59154 ). Therefore, we proposed to require these two measures in the CY 2027 reporting period instead of the CY 2026 reporting period to provide hospitals with additional time to gain experience with these newer measures ( 89 FR 36336 through 36339 ).
We refer readers to section IX.C.8. for the full list of eCQMs by payment determination in the Hospital IQR Program. If a hospital does not have patients that meet the denominator criteria for any of the eCQMs included in this proposal, the hospital would submit a zero-denominator declaration for the measure that allows a hospital to meet the reporting requirements for a particular eCQM. We refer readers to the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50258 ), the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49705 through 49708 ), and the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57170 ) for our previously adopted eCQM file format requirements. A QRDA Category I file with patients meeting the initial patient population of the applicable measures, a zero-denominator declaration, and/or a case threshold exemption all count toward a successful submission for eCQMs for the Hospital IQR Program ( 82 FR 38387 ). The following Table IX.C.9 summarizes the proposed policies:

We invited public comment on our proposal to increase the number of mandatory eCQMs over a two-year period to ultimately require reporting on five additional eCQMs beginning with CY 2026 Reporting Period/FY 2028 Payment Determination. We refer readers to section IX.F.6.b. of this final rule, in which we outline similar reporting and submission requirements under the Medicare Promoting Interoperability Program.
Comment: Many commenters supported our proposal to modify eCQM reporting requirements. A few commenters supported modifying eCQM requirements because it is a reasonable step in moving towards the goal to transition all quality measure reporting to digital quality measures (dQMs). Another commenter supported the proposed requirements because they would support transition to dQMs, which would in turn provide more real-time, actionable data, and reduce the burden of chart-abstracted measures.
Many commenters specifically supported making the Hospital Harm—Severe Hypoglycemia and the Hospital Harm—Severe Hyperglycemia eCQMs mandatory, noting that both conditions are serious adverse events that can be avoided with proper glycemic management through tracking blood glucose levels. A few commenters support modifying eCQM requirements specifically because the proposed mandatory eCQMs are patient safety outcome eCQMs and mandatory reporting ensures data collected are useful to beneficiaries and the public.
A commenter supported the proposed addition of the Hospital Harm—Opioid-Related Adverse Events eCQMs for mandatory reporting because opioids have dangerous adverse effects in the inpatient hospital setting, are among the most frequently implicated medications in adverse drug events among hospitalized patients, and most opioid-related adverse events are preventable with better monitoring and response.
A few commenters strongly supported incorporation of the Hospital Harm— ( print page 69571) Pressure Injury eCQM into the expanded mandatory eCQM measure set and recommended accelerating the timeline for mandatory reporting to CY 2026 reporting period/FY 2028 payment determination. Commenters noted that accelerating the timeline would enhance prevention efforts and improve care for Medicare patients. A few commenters recommended making all patient safety outcome eCQMs mandatory.
Response: We thank commenters for their support. We agree that modifications to eCQM reporting requirements to include patient safety outcome eCQMs would increase public reporting on quality and safety, thus empowering individuals to make decisions about where to go for care, which is one of our key actions to drive improvements in safety as outlined in the CMS National Quality Strategy. [ 715 ] While we did not propose to make all patient safety outcome eCQMs mandatory at this time, we will continue to prioritize improving safety and consider additional eCQMs that focus on safety in future program years. In addition, we encourage hospitals to voluntarily report on as many patient safety eCQMs as feasible to support efforts toward improving patient safety in the hospital inpatient setting. As we note in section IX.C.5.c. and IX.C.5.d. of this final rule, we are adopting two new hospital harm eCQMs, Hospital Harm—Falls with Injury eCQM and Hospital Harm—Postoperative Respiratory Failure eCQM, beginning with the CY 2026 reporting period/FY 2028 payment determination to provide additional reporting options for patient safety.
Comment: Many commenters did not support this proposal and recommended building out the digital quality strategy further and ensuring that new requirements align with its future data collection approach. A few commenters expressed concerns about modifying eCQM reporting requirements and recommended providing greater clarity on the vision for digital quality and how eCQM reporting fits within that vision. A commenter recommended pausing new requirements to focus on efforts towards the future development of dQMs.
Response: We acknowledge commenters' concerns and requests for clarification regarding our digital quality strategy and how modifying eCQM reporting aligns with this strategy. We wish to highlight that in the FY 2022 IPPS/LTCH PPS final rule, we discussed our goal of moving to digital quality measurement for all CMS quality reporting and value-based purchasing programs ( 86 FR 45342 ). In the FY 2023 IPPS/LTCH PPS final rule, we further described our goals to transition to dQMs, which include: reducing burden of reporting; provision of multi-dimensional data in a timely fashion, rapid feedback, and transparent reporting of quality measures; leveraging digital measures for advanced analytics to define, measure, and predict key quality issues; and employing quality measures that support development of a learning health system, which uses key data that are also used for care, quality improvement, public health, and research ( 87 FR 49181 through 49188 ). We also wish to highlight that our previously described vision for future dQMs would leverage interoperability standards to decrease mapping burden and align standards for quality measurement with interoperability standards used in other healthcare exchange methods ( 87 FR 49181 through 49188 ).
The definition of dQM that we have published as part of strategic materials on the eCQI Resource Center states that in general, eCQMs are a subset of dQMs. As defined, dQMs are quality measures that use standardized, digital data from one or more sources of health information that are captured and exchanged via interoperable systems; apply quality measure specifications that are standards-based and use code packages; and are computable in an integrated environment without additional effort. [ 716 ] As we previously described, increasing eCQM reporting requirements are a part of CMS' National Quality Strategy to “accelerate and support the transition to a digital and data-driven health care system” by taking action to annually increasing the percentage of digital quality measures used in quality programs. [ 717 ] Regarding the recommendation to pause new eCQM reporting requirements to focus efforts on the future development of dQMs, we wish to note that the addition of these eCQMs to our reporting requirements further advances CMS' goal of transition toward a fully digital quality measurement landscape promoting interoperability that would help decrease burden. [ 718 719 ]
Comment: A commenter did not support this proposal because feedback to hospitals about their performances on eCQMs is infrequent and seldom helpful as a basis for performance improvement. The commenter recommended that CMS provide more frequent and actionable eCQM performance feedback.
Response: We disagree with the commenter that eCQM performance feedback is infrequent and not useful for performance improvement. We note that eCQMs provide real or near-real-time performance information because they are calculated using data in hospitals' EHRs. By using eCQMs, hospitals are not reliant on calculations provided by CMS and do not need to wait until they receive performance reports from CMS the way that they do with claims-based measures. Based on this immediate, or near-immediate, feedback embodied in eCQMs, hospitals should receive frequent and actionable feedback on their measured performance.
Comment: A commenter requested clarification regarding whether the number of voluntary eCQMs is being reduced.
Response: We interpret the commenter's question as asking for clarification about the number of self-selected eCQMs hospitals would have to report as part of the eCQM reporting requirements. In response, we did not propose any changes; hospitals would continue to report three self-selected eCQMs, as described in the proposal to modify eCQM reporting requirements in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36336 through 36339 ).
Comment: Several commenters did not support the proposed modifications to eCQM reporting, stating that the required reporting of the previously adopted Hospital Harm eCQMs is premature, noting the proposed mandatory eCQMs are very new to the Hospital IQR Program. These commenters recommended maintaining the current requirements until important issues with the existing Hospital Harm eCQMs have been addressed. Several commenters similarly expressed concern about the feasibility of implementing the two Hospital Harm glycemic control eCQMs ( print page 69572) and recommended additional testing for these measures. A commenter noted that three of the proposed mandatory eCQMs (Hospital Harm—Opioid-Related Adverse Events, Hospital Harm—Pressure Injury, and Hospital Harm—Acute Kidney Injury) are measures that hospitals do not yet have in use, noting hospitals have not had an adequate opportunity to receive feedback on these measures. A commenter expressed concern that a one-year voluntary reporting period for new eCQMs may not be sufficient to implement and validate.
Response: Regarding the recommendation to delay mandatory reporting requirements for both the Hospital Harm—Severe Hyperglycemia eCQM and Hospital Harm—Severe Hypoglycemia eCQM, we wish to note that hospitals will have had three years to self-select to report these eCQMs as we adopted these eCQMs beginning with the CY 2023 reporting period/FY 2025 payment determination ( 86 FR 45382 through 45390 ). We have thus provided three years for hospitals to report and incorporate feedback, and we continue to encourage hospitals to self-select to report on these eCQMs as feasible to support efforts toward patient safety in the hospital inpatient setting. We acknowledge the Hospital Harm—Opioid-Related Adverse Events, Hospital Harm—Pressure Injury, and Hospital Harm—Acute Kidney Injury eCQMs are relatively newer to the Hospital IQR Program eCQM measure set.
Comment: Many commenters did not support eCQM reporting modifications and expressed concern with the burden associated with implementing new eCQMs per the proposed timeline. Many commenters, stressing the importance of flexibility and incremental change to quality reporting, recommended adopting a more phased approach to implement the new requirements. Commenters maintained that participants in the Hospital IQR Program required additional time to implement workflow changes and required updates to keep pace with this impactful increase in eCQM reporting. Several commenters recommended additional time specifically for staff training, education, and rollout.
Many commenters expressed concerns about the burden associated with meeting these new requirements with limited health IT resources available. Many commenters noted that hospitals have experienced a significant increase in requirements over a short period of time and that they must invest significant time and staff resources, noting this places a significant burden on hospitals. Commenters expressed concern that the volume of changes being implemented introduces a significant administrative burden for small and rural hospitals, including critical access hospitals (CAHs). A few commenters described the intensive process to meet new eCQM reporting requirements, particularly for small teams, noting IT staff in hospitals is often very limited and that EHR vendor lead-times to implement changes can often take several years to go- live. Commenters noted that EHR vendors need considerable advance notice to complete upgrades and programming to meet new eCQM reporting requirements and that CMS should incrementally ramp up eCQM reporting requirements in order to advance digital quality measurement. A commenter recommended allowing more time for hospitals and EHR vendors to focus on eCQM optimization as they currently exist before adding new eCQMs and further increasing administrative burdens. Another commenter emphasized that the hospital workforce is under tremendous strain, noting quality and health IT resources are stretched thin, and that adding more reporting mandates to hospitals may prove unsustainable. Another commenter recommended considering providing direct funding for increased efforts by hospitals to implement eCQMs.
Several commenters had specific recommendations for a further phased approach. A commenter recommended that CMS require only one additional eCQM in CY 2026 and only one additional eCQM in CY 2027 to reduce the burden and resources necessary to comply with the proposal. A commenter requested delay of the Hospital Harm—Pressure Injury eCQM Requirement and the Hospital Harm—Acute Kidney Injury eCQM for an additional year, noting the additional year would provide hospitals time to implement these measures and respond to feedback. This commenter specifically suggested adopting the Hospital Harm—Opioid-Related Adverse Events measure next year and Hospital Harm—Pressure Injury as well as Hospital Harm—Acute Kidney Injury the following year. Another commenter recommended transitioning fewer eCQMs to mandatory reporting. A commenter noted that hospitals should be able to self-select the majority of their reported eCQMs.
Response: We acknowledge the concerns of commenters related to the burden that the proposed timeline imposes on hospitals to implement a set of new eCQMs, particularly on small and rural hospitals, including CAHs. We further acknowledge comments regarding needing time to map EHR data and that there is often novel data collection involved in implementing new eCQMs that can be challenging and burdensome for providers. We also recognize that there are many new requirements placing burden on hospital IT staff, including working through the challenges associated with the implementation of the Hybrid Hospital-Wide Readmission and Hybrid Hospital-Wide Mortality measures.
After considering these comments, and in response to commenters' feedback recommending additional time for implementing new eCQMs, we are finalizing a modification of our proposal on eCQM reporting requirements.
Specifically, we are finalizing a modification of our proposal such that for the CY 2026 reporting period/FY 2028 payment determination, hospitals would be required to submit data for eight total eCQMs: three self-selected, Safe Use of Opioids, Severe Obstetric Complications, Cesarean Birth, Hospital Harm—Severe Hypoglycemia, and Hospital Harm—Severe Hyperglycemia. For the CY 2027 reporting period/FY 2029 payment determination, hospitals would be required to submit data for these eight eCQMs in addition to the Hospital Harm—Opioid-Related Adverse Events eCQM, for a total of nine eCQMs. Lastly, beginning with the CY 2028 reporting period/FY 2030 payment determination, hospitals would be required to submit data for these nine eCQMs in addition to the Hospital Harm—Pressure Injury and Hospital Harm—Acute Kidney Injury eCQMs, for a total of eleven eCQMs. We refer readers to Table IX.C.XXXX for a summary of the newly finalized eCQM reporting and submission requirement policies. We reiterate that we are fully committed to our National Quality Strategy priority area of “Patient Safety and Resiliency” and, likewise, taking action to expand the collection and use of safety indicator data across programs, including data on key areas such as adverse events. [ 720 ] In finalizing eCQM reporting requirements with revisions, we sought to balance the need for hospitals and their vendors to prepare for reporting the new eCQMs with the urgency of measuring at a national scale and addressing important patient safety events in hospital inpatient settings in the U.S.
The following Table IX.C.XXXX summarizes the newly finalized policies:

We proposed changes to our policies for eCQM validation scoring processes beginning with validation of eCQMs affecting the FY 2028 payment determinations.
In the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53539 through 53553 ), we finalized the processes and procedures for validation of chart-abstracted measures in the Hospital IQR Program for the FY 2015 payment determination and subsequent years. In the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38398 through 38403 ), we finalized several requirements for the validation of eCQM ( print page 69574) data, including a policy requiring submission of at least 75 percent of sampled eCQM medical records in a timely and complete manner for validation ( 81 FR 57181 ). In the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58950 through 58952 ), we finalized the existing Hospital IQR Program validation scoring processes such that a combined score is calculated based on a weighted combination of a hospital's validation performance for chart-abstracted measures and eCQMs. Under the aligned validation policies, each hospital selected for validation is expected to submit medical record data for both chart-abstracted measures and eCQMs ( 85 FR 58942 through 58953 ). Beginning with validation procedures affecting the FY 2024 payment determination, we finalized a policy to annually identify one pool of up to 200 hospitals selected through random selection and one pool of up to 200 hospitals selected using targeting criteria to participate in both chart-abstracted measure and eCQM validation ( 85 FR 58942 through 58953 ).
We refer readers to 42 CFR 412.140(d) for our codification of validation policies and to the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49308 through 49310 ) for a discussion of the most recent changes to chart-abstracted and eCQM data validation requirements for the Hospital IQR Program wherein we finalized the requirement that hospitals selected for validation must submit timely and complete data for 100 percent of requested records for eCQM validation. We refer readers to the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57178 through 57180 ) for details on the Hospital IQR Program data submission requirements for chart-abstracted measures.
Under the existing eCQM data validation policy, as described in the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57180 through 57181 ), the accuracy of eCQM data (the extent to which data abstracted for validation matches the data submitted in the QRDA I file) has not affected a hospital's validation score. Instead, hospitals have been scored on the completeness of eCQM medical record data that were submitted for the validation process. In the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38401 ), we noted our intention for the accuracy of eCQM data validation to affect validation scores in the future.
We have assessed agreement rates, or the rates by which hospitals' reported eCQM data agree with the data resulting from the review process that we conduct as part of validation. The agreement rates for validation accuracy, which have been confidentially reported to hospitals selected for eCQM validation in recent years, are consistently robust overall. For example, around 90 percent (national average agreement rate) for current eCQMs that would be validated in FY 2028 (ranging from a low average of about 84 percent for the Anticoagulation Therapy for Atrial Fibrillation/Flutter eCQM to a high of average of about 94 percent for the Antithrombotic Therapy by the End of Hospital Day Two eCQM), based on FY 2024 validation results. With the low end of the average accuracy range being well above a passing threshold of 75 percent, it is now appropriate to move forward with scoring hospitals' eCQM data based on the accuracy of the data submitted for purposes of determining whether a hospital has met the validation requirements under the Hospital IQR Program. Therefore, in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36339 ), we proposed to implement eCQM validation scoring based on the accuracy of eCQM data beginning with CY 2025 eCQM data affecting the FY 2028 payment determination. By the time our eCQM validation scoring methodology would go into effect, we would have been validating eCQM data for completeness for 8 years, which is ample time for hospitals to have prepared for data to be validated based on its accuracy. We also noted that because hospitals are already required to submit 100 percent of requested eCQM medical records to pass the eCQM validation requirement, there is no additional burden to hospitals associated with this policy to begin scoring the submitted records.
In addition, in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36339 through 36340 ), we proposed to remove the requirement at § 412.140(d)(2)(ii) that hospitals submit 100 percent of the requested eCQM medical records to pass the eCQM validation requirement and proposed that missing eCQM medical records would be treated as mismatches, beginning with the validation of CY 2025 eCQM data affecting the FY 2028 payment determination. This is the same methodology that is applied for missing medical records in chart-abstracted measure validation to incentivize the timely submission of requested medical records. Because mismatches count against the agreement rate, by treating missing eCQM medical records as mismatches, we can ensure our validation scoring methodology clearly requires that hospitals submit all necessary eCQM data for our review without also requiring medical records submissions.
In the proposed rule ( 89 FR 39339 ), we proposed that eCQM validation scores be determined using the same methodology that is currently used to score chart-abstracted measure validation. Hospitals' eCQM data would be used to compute an agreement rate and its associated confidence interval. The upper bound of the two-tailed 90 percent confidence interval would be used as the final eCQM validation score for the selected hospital. A minimum score of 75 percent accuracy would be required for the hospital to pass the eCQM validation requirement. Based on the FY 2024 results, most measures had national agreement rates well above the proposed 75 percent threshold, however these FY 2024 results are based on only two quarters of data and included data only from eCQMs that have been in the Hospital IQR Program for several years. We anticipate that the average agreement rates may decrease with a full year of data and the introduction of newer eCQMs that hospitals may have less experience reporting. As such, while we may consider raising the minimum passing threshold from 75 percent in future years, at this time we have determined that the 75 percent threshold is appropriate for initial scoring of eCQMs in Hospital IQR Program validation.
We invited public comment on our proposal to Modify eCQM Validation Scoring beginning with CY 2025 eCQM data affecting the FY 2028 payment determination. We summarize the public comments that we received, along with our responses, in the next subsection.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36339 ), we proposed to remove the existing combined validation score based on a weighted combination of a hospital's validation performance for chart-abstracted measures and eCQMs and replace it with two separate validation scores, one for chart-abstracted measures, and one for eCQMs. Based on our current policies, the eCQM portion of the combined agreement rate is multiplied by zero percent, and the chart-abstracted measure agreement rate ( print page 69575) is weighted at 100 percent. A minimum passing score for this combined score is set at 75 percent.
Reporting requirements and procedures for eCQMs are different than those for chart-abstracted measures. For instance, hospitals implement electronic algorithms to query eCQM data and submit eCQM measure results using a custom file layout for quality data reporting to CMS. In contrast, validation of chart-abstracted measures is conducted using measure specifications written to support manual abstraction processes. As such, separate validation scores are consistent with the distinct requirements and procedures for the reporting of quality measure data. Moreover, CMS intends to retain an emphasis on data accuracy through the validation efforts across both measure types (that is, chart-abstracted measures and eCQMs). It is important to ensure necessary analysis and resources are placed on chart-abstracted measures that are still currently being validated, especially because of their use within the Hospital Value-Based Purchasing (VBP) Program. Therefore, in the proposed rule ( 89 FR 36339 through 36340 ), we proposed to implement two separate scoring processes, one for chart-abstracted measures and one for eCQMs, for the FY 2028 payment determination and subsequent years. Hospitals would be required to receive passing validation scores for both chart-abstracted measure data and eCQM data to pass validation.
Under our proposal, beginning with the validation of CY 2025 data affecting the FY 2028 payment determination, hospitals would receive separate validation scores for both chart-abstracted measure data and eCQM data, which would be used to determine a hospital's overall annual payment update. As established in the FY 2006 IPPS final rule ( 70 FR 47420 through 47428 ), a hospital that fails to meet validation requirements may not receive the full annual payment update. Under our proposal, if a hospital fails either chart-abstracted validation requirements or eCQM validation requirements, it may not receive the full annual payment update. To be eligible for a full annual payment update, provided all other Hospital IQR Program requirements are met, a hospital would have to attain at least a 75 percent validation score for chart-abstracted measure validation and at least a 75 percent validation score for eCQM data validation.
Our existing and newly proposed validation scoring changes are summarized in Table IX.C.10.

We invited public comment on our eCQM validation proposals.
Comment: Some commenters supported our eCQM validation proposals in alignment with other validation scoring standards, including changing the weighting of the eCQM validation score from 0 percent to 50 percent.
Comment: Some commenters appreciated that the proposed eCQM validation requirements are meant to align with other validation criteria in the Hospital IQR Program. However, commenters had some concerns about eCQM validation scoring, noting that hospitals have little detail about validation decisions and questioned the value of the feedback reports provided to hospitals. Commenters requested that CMS publish a resource guide and requested that CMS not penalize hospitals for issues outside their control, like measure definition issues. Some commenters requested that CMS allow a grace period for new validation of new eCQMs since many new eCQMs have been introduced into the Hospital IQR Program in recent years.
Response: We thank the commenters for this feedback. However, our experience in validating eCQMs in previous years, coupled with consistently high agreement rates, confirms that these measures are ready for validation. We have a number of resources related to validation located on our QualityNet website at: https://qualitynet.cms.gov/inpatient/data-management/data-validation/resources , and will consider publishing additional resources to support hospitals' efforts to report eCQM data accurately in the future.
Comment: Some commenters requested that CMS consider changing the timeline for validation of eCQM data. A commenter requested additional time for review and validation of measure data to avoid incorrect validation decisions. The commenter argued that hospitals and vendors often need six or more weeks to complete coding, leaving little time for validation prior to data submission deadlines under the current practice. The commenter suggested that CMS align the eCQM reporting deadline with MIPS' deadline for March 31 instead of the current February 28th. Another commenter requested that the increased number of eCQMs should be delayed to avoid coinciding with increased validation requirements and called for an extended timeline for eCQM data submissions. The commenter also suggested analyzing how charts submitted for eCQM validation are being assessed to ensure that correct validation methods are used. ( print page 69576)
Response: We thank the commenters for this feedback. However, as stated earlier, our experience with validating eCQMs in previous years, coupled with consistently high agreement rates, confirms that the measures are ready for validation, and correspondingly, that hospitals are sufficiently prepared to submit their eCQM data and for those data to be sufficiently accurate for validation. We do not agree that we should delay implementation of additional validation requirements for eCQMs because we continue to believe that accurate and complete eCQM data are crucial to informing members of the public about the care quality that Medicare beneficiaries receive in hospitals. We understand that hospitals would need to adjust processes and workflows to account for additional eCQM reporting requirements, but delaying validation would not impact hospitals' efforts to deliver the highest quality care to their patients. We will continue to work with hospitals to ensure that they are fully informed about the validation program and that they continue to report their eCQM data accurately.
Comment: Some commenters opposed CMS's eCQM validation proposals, arguing that validation imposes a significant administrative burden on hospitals and that validating eCQMs for accuracy would increase that burden. Commenters noted that targeted reviews during eCQM validation would require additional cases per quarter to be submitted during the 30-day window to retrieve and review records and suggested that CMS allow up to 45 days for those processes.
Response: We thank the commenters for this feedback. However, while we understand that validation imposes some reporting burden on hospitals, we have determined, based on the power analysis for the confidence interval calculation, that a final sample size of eight eCQM cases per quarter is essential to achieve sufficient and accurate validation results. We do not intend to increase the hospital sample size for eCQM validation because we have attempted to ensure that our eCQM validation proposals achieve both the objectives of minimizing reporting burden on participating hospitals and ensuring that hospitals report fully accurate data. We will consider whether to extend the timeframe for hospitals' retrieval of records for validation purposes in the future. However, we note that we are required by section 1886(b)(3)(B)(viii)(XI) to establish a process to validate measure data submitted by hospitals under the Program, with the process to include random audits. Our validation program continues to ensure that the data reported by hospitals, and that we report publicly, are as accurate as feasible and to the extent that the commenters oppose validation requirements in their entirety do not understand that such a position would adhere to our statutory direction.
Comment: Some commenters requested that CMS consider requiring validation of fewer measures for selected hospitals and start with a required validation score lower than 75 percent. A commenter suggested that CMS delay eCQM validation scoring until after some chart-abstracted measures are removed from the program. Another commenter argued that the 75 percent threshold is more appropriate for chart-abstracted measures with which hospitals have more experience than eCQMs.
Response: We proposed the 75 percent agreement rate for eCQM validation scoring in alignment with common standards that we have adopted in other validation programs, including chart-abstracted measure validation previously adopted in the Hospital IQR Program ( 81 FR 57179 through 57180 ), to ensure sufficient accuracy of the measures. Given the high overall agreement rates that we have observed for eCQMs, we anticipate that this threshold would uphold the integrity of the validation process consistently and that its alignment with other validation programs would help hospitals meet validation requirements consistently across measure types. We will continue observing hospitals' experience with eCQM validation and will consider whether we should propose further refinements in future years.
Comment: Some commenters opposed eCQM validation scoring changes for measures that have been adopted for their first or second year of reporting, arguing that validation is more reasonable for measures that hospitals have reported for at least two years. Commenters suggested that CMS consider a phased-in approach to eCQM validation starting in CY 2026 or later. Commenters noted that hospitals often receive their eCQM data at the very end of the calendar year and thus do not have time for their staff to review their performance and work to implement improvements.
Response: We thank commenters for this feedback. However, as we stated earlier, our experience in validating eCQMs in previous years, coupled with consistently high agreement rates, confirms that these measures are ready for validation and that hospitals can successfully report eCQM data accurately. We understand that hospitals must adjust their processes to account for new eCQMs, but ensuring that hospitals report accurately on their eCQMs necessitates the validation policies that we have proposed. We will continue to observe hospitals' experience with eCQM validation and will consider whether to revisit these policies in future years.
Comment: A commenter opposed CMS's eCQM validation proposals, arguing that hospitals have never received any feedback on eCQM validation from the agency.
Response: We would like to clarify that, as we discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36339 ), we have confidentially reported agreement rates for validation accuracy to hospitals selected for eCQM validation in recent years. Those agreement rates range between about 84 percent to about 94 percent based on FY 2024 validation results.
Comment: A commenter was concerned about the time it takes CMS to make changes to quality measure specifications and the speed with which eCQMs can be adopted thereafter. The commenter noted that, in one case, it took CMS five years to make a change to the perinatal care eCQM specifications, but the measure became a requirement the following year. The commenter argued that this timeframe is too short for hospitals to adjust and ensure data integrity, particularly for eCQMs where the commenter stated that hospitals and CMS must partner to identify and correct issues with measure specifications.
Response: We thank the commenter for this feedback. We note that quality measure specifications changes are the purview of the measure steward, which may be CMS in some cases or may be a third party. Minor or technical changes to quality measure specifications are not significant enough to impair hospitals' delivery of high-quality care to their patients while those changes are implemented in EHR systems. Based on the high agreement rates we have observed to date, hospitals have been successful at implementing the necessary updates to their systems and care practices. We agree with the commenter that we must partner with hospitals to ensure that issues identified in measure specifications are appropriately captured in eCQMs so that hospitals report data successfully and accurately to CMS and we will continue to engage collaboratively with hospitals to that end. ( print page 69577)
Comment: A commenter expressed concerns about eCQM validation when, in the commenter's view, hospitals are at the mercy of measure developers and EHR vendors. The commenter argued that, unless code sets are fully updated, hospitals are unable to capture patient chart data accurately. The commenter requested that CMS provide examples of inaccurately reported data to inform hospitals and requested that CMS consider a confidential feedback period before penalizing hospitals over eCQM data accuracy.
Response: As stated earlier, minor or technical changes to quality measure specifications are not significant enough to alter hospitals' ability to provide care in accordance with the applicable clinical standard. We understand that hospitals must rely on measure developers and EHR vendors for their eCQMs reporting, and we work closely with both groups to ensure that the measures that we propose to adopt and that we propose to validate are fully ready for hospitals' implementation. We will consider whether we should provide additional confidential feedback to hospitals in the future, potentially including examples of inaccurately reported data, as we continue working to keep hospitals fully informed about our validation programs.
After consideration of the comments that we have received, we are finalizing our eCQM validation policies as proposed.
We refer readers to the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53554 ) for previously adopted details on DACA requirements. We did not propose any changes to this policy in this final rule. We refer readers to the QualityNet website at: https://qualitynet.cms.gov (or other successor CMS designated websites) for more details on DACA requirements.
Section 1886(b)(3)(B)(viii)(VII) of the Act requires the Secretary to report quality measures of process, structure, outcome, patients' perspectives on care, efficiency, and costs of care that relate to services furnished in inpatient settings in hospitals on the internet website of CMS. Section 1886(b)(3)(B)(viii)(VII) of the Act also requires that the Secretary establish procedures for making information regarding measures available to the public after ensuring that a hospital can review its data before they are made public. Our current policy is to report data from the Hospital IQR Program as soon as it is feasible on CMS websites such as the Compare tool hosted by HHS, currently available at: https://www.medicare.gov/care-compare , or its successor website, after a 30-day preview period ( 78 FR 50776 through 50778 ).
We did not propose any changes to these policies or the public reporting of eCQM data or overall hospital star ratings in this final rule. We also refer readers to the QualityNet website at: https://qualitynet.cms.gov/inpatient/public-reporting (or other successor CMS designated websites) for details on public display requirements.
We refer readers to the CY 2025 OPPS/ASC proposed rule where we are soliciting input on potential future methodological modifications regarding the Safety of Care measure group within the Overall Hospital Quality Star Rating ( 89 FR 59509 through 59515 ).
In the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51650 through 51651 ), the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50836 ), and 42 CFR 412.140(e) , we established an approach for reconsideration and appeal procedures for the Hospital IQR Program. As part of this reconsideration process, hospitals can request reconsideration if CMS determines that the hospital did not meet the Hospital IQR Program's validation requirements. Under these requirements as established in the FY 2011 IPPS/LTCH PPS final rule ( 75 FR 50225 through 50229 ), for purposes of validation, hospitals are required to resubmit copies of all medical records that were originally submitted to the Clinical Data Abstraction Center (CDAC) each relevant quarter. With the transition to all electronic submission of copies of medical records for Hospital IQR Program validation as established in they FY 2021 IPPS/LTCH final rule ( 85 FR 58949 through 58950 ), both through eCQMs and digitized charts, the current reconsideration requirement to resubmit records used for validation results is no longer necessary and creates duplicative files and work.
Therefore, we proposed to revise § 412.140(e)(2)(vii)(A) to no longer require hospitals to resubmit medical records as part of their request for reconsideration of validation, beginning with CY 2023 discharges affecting the FY 2026 payment determination.
Under our proposal, hospitals that need to submit a revised medical record may still do so, but those hospitals that would otherwise be resubmitting copies of the previously submitted records would no longer be required to submit them. Removing record submission as a requirement for validation reconsideration would reduce hospital administrative burden for most hospitals that do not have revised records to submit. Making this step optional would also reduce the burden for CMS to collect and track medical records that are already available.
We invited public comment on our proposal to remove the requirement for hospitals to resubmit medical records as part of their request for reconsideration of validation, beginning with CY 2023 discharges affecting the FY 2026 payment determination.
Comment: A commenter supported our proposal to remove the requirement to resubmit records for validation, agreeing with us that this change would reduce burden on participating hospitals.
Response: We thank the commenter for their support.
After consideration of the comment that we received, we are finalizing this policy as proposed beginning with the CY 2023 discharges affecting the FY 2026 payment determination.
We did not propose any changes to this policy in this final rule. We refer readers to § 412.140(c)(2) and the QualityNet website at: https://qualitynet.cms.gov (or other successor CMS designated websites) for our current requirements for submission of a request for an exception.
The PPS-Exempt Cancer Hospital Quality Reporting (PCHQR) Program, authorized by section 1866(k) of the Act, applies to hospitals described in section 1886(d)(1)(B)(v) of the Act (referred to as “PPS-Exempt Cancer Hospitals” or “PCHs”). In the FY 2025 IPPS/LTCH PPS proposed rule ( 86 FR 36341 ), we proposed to adopt the Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 program year as described in section IX.B.1. of this final rule. We also proposed to modify the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) Survey measure as described in section IX.B.2. of this final rule and proposed to move up the start date for publicly displaying hospital performance on the ( print page 69578) Hospital Commitment to Health Equity measure ( 86 FR 36341 ). [ 721 ]
We refer readers to section IX.B.1. of this final rule where we finalize with modification the adoption of the Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 program year for the PCHQR Program. We are also finalizing with modification the adoption of this measure for the Hospital Inpatient Quality Reporting (IQR) Program, as discussed in that section.
We refer readers to section IX.B.2. of this final rule where we finalize the modification of the HCAHPS Survey measure (CBE #0166) beginning with the CY 2025 reporting period/FY 2027 program year for the PCHQR Program. We are also finalizing the adoption of the same modifications to this measure for purposes of the Hospital IQR Program and the Hospital VBP Program, as discussed in the same section.
Table IX.D.-01 summarizes the previously adopted and the newly finalized measures for the PCHQR Program measure set beginning with the CY 2025 reporting period/FY 2027 program year.

In the FY 2024 IPPS/LTCH PPS final rule, we adopted the Hospital Commitment to Health Equity measure for the PCHQR measure set beginning with the CY 2024 reporting period/FY 2026 program year ( 88 FR 59204 through 59210 ). We also finalized that we would publicly report PCH performance on this measure beginning with CY 2024 data beginning July 2026 or as soon as feasible thereafter ( 88 FR 59209 ; 59228).
In the FY 2025 IPPS/LTCH PPS proposed rule, we proposed to accelerate the timeline for beginning to publicly report PCH performance on this measure. Specifically, we proposed to start public reporting of PCH performance on this measure using CY 2024 data beginning January 2026 or as soon as feasible thereafter. We stated that the public could benefit from having access to the information sooner because the data provide an opportunity to recognize PCHs that have attested to their commitment to health equity at an earlier date. We also stated that the modification of the date for public reporting would promote efficiencies through alignment of the performance periods, data submission periods, and the anticipated public reporting release with the Inpatient Psychiatric Facility Quality Reporting (IPFQR) Program that adopted the Facility Commitment to Health Equity measure (which requires the same attestations as the Hospital Commitment to Health Equity measure) beginning with reporting of CY 2024 data for the FY 2026 payment determination and would provide this information for providers participating in the PCHQR Program and the IPFQR Program simultaneously. We invited public comment on this proposal.
Comment: A few commenters supported moving up the public reporting timeline for the Hospital Commitment to Health Equity measure ( print page 69580) because they believe that patients will benefit from being able to access the information sooner, and that the earlier publication timeframe will promote greater efficiencies through alignment with other CMS quality reporting programs without changing the submission timeline for PCH data.
After consideration of the public comments we received, we are finalizing the start of public reporting of the Hospital Commitment to Health Equity measure to January 2026 or as soon as feasible thereafter.
Our previously finalized public display policies and newly finalized public display start date change for the Hospital Commitment to Health Equity measure for the PCHQR Program are described in Table IX.D.-02:

The Long-Term Care Hospital Quality Reporting Program (LTCH QRP) is authorized by section 1886(m)(5) of the Act, and it applies to all hospitals certified by Medicare as Long-Term Care Hospitals (LTCHs). Section 1886(m)(5)(C) of the Act requires LTCHs to submit to the Secretary quality measure data specified under section 1886(m)(5)(D) in a form and manner, and at a time, specified by the Secretary. In addition, section 1886(m)(5)(F) of the Act requires LTCHs to submit data on quality measures under section 1899B(c)(1) of the Act, resource use or other measures under section 1899B(d)(1) of the Act, and standardized patient assessment data required under section 1899B(b)(1) of the Act. LTCHs must submit the data required under section 1886(m)(5)(F) of the Act in the form and manner, and at the time, specified by the Secretary. Under the LTCH QRP, the Secretary must reduce by two percentage points the annual update to the LTCH PPS standard federal rate for discharges for an LTCH during a fiscal year (FY) if the LTCH has not complied with the LTCH QRP requirements specified for that FY. Section 1890A of the Act requires that the Secretary establish and follow a pre-rulemaking process, in coordination with the consensus-based entity (CBE) with a contract under section 1890(a) of the Act, to solicit input from certain groups regarding the selection of quality and efficiency measures for the LTCH QRP. We have codified our program requirements in our regulations at 42 CFR 412.560 .
We proposed to require LTCHs to report four new items to the LTCH Continuity Assessment and Record of Evaluation (CARE) Data Set (LCDS) and modify one item on the LCDS as described in section IX.E.4. of the preamble of this final rule. Second, we proposed to extend the Admission assessment window for the LCDS as described in section IX.E.7.c. Third, we sought information on future measure concepts for the LTCH QRP, and finally, we sought information on a future LTCH Star Rating system.
For a detailed discussion of the considerations, we historically use for the selection of LTCH QRP quality, resource use, and other measures, we refer readers to the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49728 ).
The LTCH QRP currently has 18 adopted measures, which are set out in Table IX.E.-01. For a discussion of the factors used to evaluate whether a measure should be removed from the LTCH QRP, we refer readers to the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41624 through 41634 ) and to the regulations at 42 CFR 412.560(b)(3) .

We did not propose to adopt any new measures for the LTCH QRP.
In the proposed rule, we proposed to add four new items [ 722 ] to be collected as standardized patient assessment data elements under the social determinants of health (SDOH) category under the LTCH QRP: Living Situation (one item); Food (two items); and Utilities (one item). We also proposed to modify one of the current items collected as a standardized patient assessment data element under the SDOH category (the Transportation item). [ 723 ]
Section 1886(m)(5)(F)(ii) of the Act requires LTCHs to submit standardized patient assessment data required under section 1899B(b)(1) of the Act. Section 1899B(b)(1)(A) of the Act requires post-acute care (PAC) providers to submit standardized patient assessment data under applicable reporting provisions (which, for LTCHs, is the LTCH QRP) with respect to the admission and discharge of an individual (and more frequently as the Secretary deems appropriate). Section 1899B(a)(1)(C) of the Act requires, in part, the Secretary to modify the PAC assessment instruments in order for PAC providers, including LTCHs, to submit standardized patient assessment data under the Medicare program. LTCHs are currently required to report patient assessment data through the LCDS. Section 1899B(b)(1)(B) of the Act describes standardized patient assessment data as data required for at least the quality measures described in section 1899B(c)(1) of the Act and that is with respect to the following categories: (1) functional status, such as mobility and self-care at admission to a PAC provider and before discharge from a PAC provider; (2) cognitive function, such as ability to express ideas and to understand, and mental status, such as depression and dementia; (3) special services, treatments, and interventions, such as need for ventilator use, dialysis, chemotherapy, central line placement, and total parenteral nutrition; (4) medical conditions and comorbidities, such as diabetes, congestive heart failure, and pressure ulcers; (5) impairments, such as incontinence and an impaired ability to hear, see, or swallow; and (6) other categories deemed necessary and appropriate by the Secretary. ( print page 69583)
Section 1899B(b)(1)(B)(vi) of the Act authorizes the Secretary to collect standardized patient assessment data elements with respect to other categories deemed necessary and appropriate. Accordingly, we finalized the creation of the SDOH category of standardized patient assessment data elements in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42577 through 42581 ), and defined SDOH as the socioeconomic, cultural, and environmental circumstances in which individuals live that impact their health. [ 724 ] According to the World Health Organization, research shows that the SDOH can be more important than health care or lifestyle choices in influencing health, accounting for between 30-55% of health outcomes. [ 725 ] This is a part of a growing body of research that highlights the importance of SDOH on health outcomes. Subsequent to the FY 2020 IPPS/LTCH PPS final rule, we expanded our definition of SDOH: SDOH are the conditions in the environments where people are born, live, learn, work, play, worship, and age that affect a wide range of health, functioning, and quality-of-life outcomes and risks. [ 726 727 728 ] This expanded definition aligns our definition of SDOH with the definition used by HHS agencies, including OASH, the Centers for Disease Control and Prevention (CDC) and the White House Office of Science and Technology Policy. [ 729 730 ] We currently collect seven items in this SDOH category of standardized patient assessment data elements: ethnicity, race, preferred language, interpreter services, health literacy, transportation, and social isolation ( 84 FR 42577 through 42581 ). [ 731 ]
In accordance with our authority under section 1899B(b)(1)(B)(vi) of the Act, we similarly finalized the creation of the SDOH category of standardized patient assessment data elements for Skilled Nursing Facilities (SNFs) in the FY 2020 SNF PPS final rule ( 84 FR 38805 through 38817 ), for Inpatient Rehabilitation Facilities (IRFs) in the FY 2020 IRF PPS final rule ( 84 FR 39149 through 39161 ), and for Home Health Agencies (HHAs) in the Calendar Year (CY) 2020 HH PPS final rule (84 60597 through 60608). We also collect the same seven SDOH items in these PAC providers' respective patient/resident assessment instruments ( 84 FR 38817 , 39161 , and 60610 , respectively).
Access to standardized data relating to SDOH on a national level permits us to conduct periodic analyses, and to assess their appropriateness as risk adjustors or in future quality measures. Our ability to perform these analyses and to make adjustments relies on existing data collection of SDOH items from PAC settings. We adopted these SDOH items using common standards and definitions across the four PAC providers to promote interoperable exchange of longitudinal information among these PAC providers, including LTCHs, and other providers. We believe this information may facilitate coordinated care, improve patient focused care planning, and allow for continuity of the discharge planning process from PAC settings.
We noted in our FY 2020 IPPS/LTCH PPS final rule that each of the items was identified in the 2016 National Academies of Sciences, Engineering, and Medicine (NASEM) report as impacting care use, cost, and outcomes for Medicare beneficiaries ( 84 FR 39150 ). At that time, we acknowledged that other items may also be useful to understand. The SDOH items we proposed to collect as standardized patient assessment data elements under the SDOH category in the proposed rule were also identified in the 2016 NASEM report [ 732 ] or the 2020 NASEM report [ 733 ] as impacting care use, cost, and outcomes for Medicare beneficiaries. These items have the potential to affect treatment preferences and goals of patients and their caregivers. Identification of these SDOH items may also help LTCHs be in a position to offer assistance, by connecting patients and their caregivers with these associated needs to social support programs, as well as inform our understanding of patient complexity.
Health-related social needs (HRSNs) are the resulting effects of SDOH, which are individual-level, adverse social conditions that negatively impact a person's health or health care. [ 734 ] Examples of HRSNs include lack of access to food, housing, or transportation, and have been associated with poorer health outcomes, greater use of emergency departments and hospitals, and higher health care costs. Certain HRSNs can lead to unmet social needs that directly influence an individual's physical, psychosocial, and functional status. [ 735 ] This is particularly true for food security, housing stability, utilities security, and access to transportation. [ 736 ]
We proposed to require LTCHs collect and submit four new items in the LCDS as standardized patient assessment data elements under the SDOH category because these items would collect information not already captured by the current SDOH items. Specifically, we believe the ongoing identification of SDOH would have three significant benefits. First, promoting screening for SDOH could serve as evidence-based ( print page 69584) building blocks for supporting healthcare providers in actualizing their commitment to address disparities that disproportionately impact underserved communities. Second, screening for SDOH improves health equity through identifying potential social needs so the LTCH may address those with the patient, their caregivers, and community partners during the discharge planning process, if indicated. [ 737 ] Third, these SDOH items could support our ongoing LTCH QRP initiatives by providing data with which to stratify LTCHs' performance on measures or in future quality measures.
Collection of additional SDOH items would permit us to continue developing the statistical tools necessary to maximize the value of Medicare data and improve the quality of care for all beneficiaries. For example, we recently developed and released the Health Equity Confidential Feedback Reports, which provided data to LTCHs on whether differences in quality measure outcomes are present for their patients by dual-enrollment status and race and ethnicity. [ 738 ] We note that advancing health equity by addressing the health disparities that underlie the country's health system is one of our strategic pillars [ 739 ] and a Biden-Harris Administration priority. [ 740 ]
We proposed to require LTCHs to collect four new items as standardized patient assessment data elements under the SDOH category using the LCDS: one item for Living Situation, as described in section IX.E.4.c.(1) of the proposed rule; two items for Food, as described in section IX.E.4.c.(2) of the proposed rule; and one item for Utilities, as described in section IX.E.4.c.(3) of the proposed rule.
We selected the proposed SDOH items from the Accountable Health Communities (AHC) Health Related Social Needs (HRSN) Screening Tool developed for the AHC Model. [ 741 ] The AHC HRSN Screening Tool is a universal, comprehensive screening for HRSNs that addresses five core domains as follows: (i) housing instability (for example, homelessness, poor housing quality), (ii) food insecurity, (iii) transportation difficulties, (iv) utility assistance needs, and (v) interpersonal safety concerns (for example, intimate-partner violence, elder abuse, child maltreatment). [ 742 ]
We believe that requiring LTCHs to report new items that are included in the AHC HRSN Screening Tool would further standardize the screening of SDOH across quality programs. For example, our proposal would align, in part, with the requirements of the Hospital Inpatient Quality Reporting (IQR) Program and the Inpatient Psychiatric Facility Quality Reporting (IPFQR) Program. As of January 2024, hospitals are required to report whether they have screened patients for the standardized SDOH categories of housing stability, food security, utility difficulties, transportation needs, and interpersonal safety to meet the Hospital IQR Program requirements. [ 743 ] Beginning January 2025, IPFs will also be required to report whether they have screened patients for the same set of SDOH categories. [ 744 ] As we continue to standardize data collection across PAC settings, we believe using common standards and definitions for new items is important to promote interoperable exchange of longitudinal information between LTCHs and other providers to facilitate coordinated care, continuity in care planning, and the discharge planning process.
Below we describe each of the four proposed items in more detail.
Healthy People 2030 prioritizes economic stability as a key SDOH, of which housing stability is a component. [ 745 746 ] Lack of housing stability encompasses several challenges, such as having trouble paying rent, overcrowding, moving frequently, or spending the bulk of household income on housing. [ 747 ] These experiences may negatively affect one's physical health and access to health care. Housing instability can also lead to homelessness, which is housing deprivation in its most severe form. [ 748 ] On a single night in 2023, roughly 653,100 people, or 20 out of every 10,000 people in the United States, were experiencing homelessness. [ 749 ] Studies also found that people who are homeless have an increased risk of premature death and experience chronic disease more often than among the general population. [ 750 ]
We believe that LTCHs can use information obtained from the Living Situation item during a patient's discharge planning. For example, LTCHs could work in partnership with community care hubs and community-based organizations to establish new care transition workflows, including referral pathways, contracting mechanisms, data sharing strategies, and implementation training that can track HRSNs to ensure unmet needs, such as housing, are successfully ( print page 69585) addressed through closed loop referrals and follow-up. [ 751 ] LTCHs could also take action to help alleviate a patient's other related costs of living, like food, by referring the patient to community-based organizations that would allow the patient's additional resources to be allocated towards housing without sacrificing other needs. [ 752 ] Finally, LTCHs could use the information obtained from the Living Situation item to better coordinate with other healthcare providers, facilities, and agencies during transitions of care, so that referrals to address a patient's housing stability are not lost during vulnerable transition periods.
Due to the potential negative impacts housing instability can have on a patient's health, we proposed to adopt the Living Situation item as a new standardized patient assessment data element under the SDOH category. This proposed Living Situation item is based on the Living Situation item collected in the AHC HRSN Screening Tool, [ 753 754 ] and was adapted from the Protocol for Responding to and Assessing Patients' Assets, Risks, and Experiences (PRAPARE) tool. [ 755 ] The proposed Living Situation item asks, “What is your living situation today?” The proposed response options are: (1) I have a steady place to live; (2) I have a place to live today, but I am worried about losing it in the future; (3) I do not have a steady place to live; (7) Patient declines to respond; and (8) Patient unable to respond. A draft of the proposed Living Situation item to be adopted as a standardized patient assessment data element under the SDOH category can be found in the Downloads section of the LCDS and LTCH Manual web page at https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-care-data-set-ltch-qrp-manual .
The U.S. Department of Agriculture, Economic Research Service defines a lack of food security as a household-level economic and social condition of limited or uncertain access to adequate food. [ 756 ] Adults who are food insecure may be at an increased risk for a variety of negative health outcomes and health disparities. For example, a study found that food insecure adults may be at an increased risk for obesity. [ 757 ] Another study found that food insecure adults have a significantly higher probability of death from any cause or cardiovascular disease in long-term follow-up care, in comparison to adults that are food secure. [ 758 ]
While having enough food is one of many predictors for health outcomes, a diet low in nutritious foods is also a factor. [ 759 ] The United States Department of Agriculture (USDA) defines nutrition security as “consistent and equitable access to healthy, safe, affordable foods essential to optimal health and well-being.” [ 760 ] Nutrition security builds on and complements long standing efforts to advance food security. [ 761 ] Studies have shown that older adults struggling with food security consume fewer calories and nutrients and have lower overall dietary quality than those who are food secure, which can put them at nutritional risk. [ 762 ] Older adults are also at a higher risk of developing malnutrition, which is considered a state of deficit, excess, or imbalance in protein, energy, or other nutrients that adversely impacts an individual's own body form, function, and clinical outcomes. [ 763 ] About 50% of older adults are affected by malnutrition, which is further aggravated by a lack of food security and poverty. [ 764 ] These facts highlight why the Biden-Harris Administration launched the White House Challenge to End Hunger and Build Health Communities. [ 765 ]
We believe that adopting items to collect and analyze information about a patient's food security at home could provide additional insight to their health complexity and help facilitate coordination with other healthcare providers, facilities, and agencies during transitions of care, so that referrals to address a patient's food security are not lost during vulnerable transition periods. For example, an LTCH's dietitian or other clinically qualified nutrition professional could work with the patient and their caregiver to plan healthy, affordable food choices prior to discharge. [ 766 ] LTCHs could also refer a patient that indicates lack of food security to government initiatives such as the Supplemental Nutrition Assistance Program (SNAP) and food pharmacies (programs to increase access to healthful foods by making them affordable), two initiatives that have been associated with lower health care costs and reduced hospitalization and emergency department visits. [ 767 ]
We proposed to adopt two Food items as new standardized patient assessment data elements under the SDOH category. These proposed items are based on the Food items collected in the AHC HRSN Screening Tool, and were adapted from the USDA 18-item Household Food Security Survey (HFSS). [ 768 ] The first proposed Food item states, “Within the past 12 months, you worried that your food would run out before you got money to buy more.” [ 769 ] The second proposed Food item states, “Within the past 12 months, the food you bought just didn't last and you didn't have money to get more.” We proposed the same response options for both items: (1) Often true; (2) Sometimes true; (3) Never True; (7) Patient declines to respond; and (8) Patient unable to respond. A draft of the proposed Food items to be adopted as a standardized patient assessment data element under the SDOH category can be found in the Downloads section of the LCDS and LTCH Manual web page at https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-care-data-set-ltch-qrp-manual .
A lack of energy (utility) security can be defined as an inability to adequately meet basic household energy needs. [ 770 ] According to the Department of Energy, one in three households in the U.S. are unable to adequately meet basic household energy needs. [ 771 ] The consequences associated with a lack of utility security are represented by three primary dimensions: economic, physical, and behavioral. Patients with low incomes are disproportionately affected by high energy costs, and they may be forced to prioritize paying for housing and food over utilities. [ 772 ] Some patients may face limited housing options and therefore are at increased risk of living in lower-quality physical conditions with malfunctioning heating and cooling systems, poor lighting, and outdated plumbing and electrical systems. [ 773 ] Patients with a lack of utility security may use negative behavioral approaches to cope, such as using stoves and space heaters for heat. [ 774 ] In addition, data from the Department of Energy's U.S. Energy Information Administration confirm that a lack of energy security disproportionately affects certain populations, such as low-income and African American households. [ 775 ] The effects of a lack of utility security include vulnerability to environmental exposures such as dampness, mold, and thermal discomfort in the home, which have a direct impact on a person's health. [ 776 777 ] For example, research has shown associations between a lack of energy security and respiratory conditions as well as mental health-related disparities and poor sleep quality in vulnerable populations such as the elderly, children, the socioeconomically disadvantaged, and the medically vulnerable. [ 778 ]
We believe adopting an item to collect information about a patient's utility security upon admission to an LTCH would facilitate the identification of patients who may not have utility security and who may benefit from engagement efforts. For example, LTCHs may be able to use the information on utility security to help connect identified patients in need, such as older adults, to programs that can help pay for home energy (heating/cooling) costs, like the Low-Income Home Energy Assistance Program (LIHEAP). LTCHs may also be able to partner with community care hubs and community-based organizations to assist the patient in applying for these and other local utility assistance programs, as well as helping them navigate the enrollment process. [ 779 ]
We proposed to adopt a new item, Utilities, as a new standardized patient assessment data element under the SDOH category. This proposed item is based on the Utilities item collected in the AHC HRSN Screening Tool and was adapted from the Children's Sentinel Nutrition Assessment Program (C-SNAP) survey. [ 780 ] The proposed Utilities item asks, “In the past 12 months, has the electric, gas, oil, or water company threatened to shut off services in your home?” The proposed response options are: (1) Yes; (2) No; (3) Already shut off; (7) Patient declines to respond; and (8) Patient unable to respond. A draft of the proposed Utilities item to be adopted as a standardized patient assessment data element under the SDOH category can be found in the Downloads section of the LCDS and LTCH Manual web page at https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-care-data-set-ltch-qrp-manual .
We developed our proposal to add these items after considering feedback we received in response to our request for information (RFI) on Closing the Health Equity Gap in CMS Hospital Quality Programs in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45349 through 45362 ). This RFI sought to update providers on CMS initiatives to make reporting of health disparities more comprehensive and actionable for LTCHs, providers, and patients. The RFI also invited public comment on future potential stratification of quality measures and improving demographic data collection. In response to the solicitation of public comment on future potential stratification and improving demographic data collection, commenters generally supported and recommended that CMS collect additional social and demographic data, like gender expression, disability status, language including English proficiency, ( print page 69587) housing security, food security, and forms of economic or financial insecurity to help provides address health equity in LTCHs. In addition, some commenters suggested CMS use standardized data collection across agencies when incorporating health equity initiatives, while also expressing concern about the burden additional data collection efforts would place on providers ( 86 FR 45358 ).
Furthermore, we considered feedback we received when we proposed the creation of the SDOH category of standardized patient assessment data elements in the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19545 ). Many commenters were generally in favor of the concept of collecting SDOH items and noted the inclusion of additional SDOH would provide greater breadth and depth of data when developing policies to address social factors related to health. Many commenters also recommended including additional factors, such as food insecurity, housing insecurity, and independent living status, to ensure the full spectrum of social needs is examined. The FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42578 through 42581 ) includes a summary of the public comments that we received and our responses to those comments. We incorporated this input into the development of this proposal.
We solicited comment on the proposal to adopt four new items as standardized patient assessment data elements under the SDOH category beginning with the FY 2028 LTCH QRP: one Living Situation item; two Food items; and one Utilities item.
We received public comments on these proposals. The following is a summary of the comments we received and our responses.
Comment: Some commenters expressed support for the proposed new SDOH assessment items, viewing this proposal as an important step towards improving patient outcomes, advancing health equity, advancing patient-centered care, improving health outcomes, and early identification of important areas that affect downstream health costs, health outcomes, and quality of life. One of these commenters also emphasized the importance of understanding the patient's environmental and personal factors to guide the selection of interventions provided through the plan of care.
Several commenters also noted the importance of the proposed new SDOH assessment items in facilitating discharge planning strategies that can account for an individual's housing, food, utilities, and transportation needs. Three of these commenters noted that the information obtained from these proposed new SDOH assessment items will help LTCHs identify future needs in collaboration with the patient and their caregivers during the discharge planning process. Two of these commenters also highlighted how SDOH influence a patient's ability to execute post-discharge recommendations and could impact patient recovery and readmission rates. These commenters said that by addressing these non-medical factors during the LTCH stay, they can help connect patients to the resources they may need, resulting in successful discharges to the community or improved health outcomes.
Response: We appreciate the support from commenters. We agree that the collection of the proposed SDOH assessment items will support LTCHs that want to understand and address health disparities that affect their patient population, facilitate coordinated care, foster continuity in care planning, and assist with the discharge planning process from the LTCH setting.
Comment: Several commenters appreciated CMS' efforts at standardizing collection of patient assessment data elements related to SDOH by proposing to adopt the four new assessment items, Living Situation, Food, and Utilities, in the LCDS. Three of these commenters recognized that the standardized collection of the proposed SDOH assessment items will support interoperability and comparability across LTCHs and within facilities for different patient populations. A couple of these commenters noted that the standardized SDOH assessment items are essential to reducing practice variance, which can lead to inconsistencies in data collection and reporting. Further, a commenter highlighted that a unified approach to SDOH can ensure that those critical social and economic factors are accurately captured and utilized to inform care decisions, ultimately leading to a more effective and equitable healthcare system.
Response: We thank the commenters for recognizing the importance of standardized SDOH assessment items in the LTCH QRP. As we continue to standardize data collection across settings, we believe using common standards and definitions for new assessment items is important to promote interoperable exchange of longitudinal information between LTCHs and other providers, including hospitals. We also believe collecting this information may facilitate coordinated care, continuity in care planning, and the discharge planning process from PAC settings, including LTCHs.
Comment: A commenter suggested that feedback from patients on their experiences of SDOH-related screenings should be used to inform updates to quality measures, while another commenter suggested that CMS could use the SDOH-related screening data in quality programs to stratify patient data.
Response: We thank the commenters for their recommendations and agree that the standardized SDOH assessment items will be valuable sources of information that would permit us to continue developing the statistical tools necessary to maximize the value of Medicare data and improve the quality of care for all beneficiaries. We will take the commenters' recommendations into consideration for future rulemaking.
Comment: Two commenters acknowledged there would be an increase in burden in collecting these four new assessment items. However, one of these commenters said that they still support the proposal because the adoption of consistent, standardized questions will reduce the burden of implementation and have a positive impact on discharge planning. The other commenter noted that the additional burden on their LTCHs will be relatively low because they are already collecting most of this information through their electronic medical record system.
Two commenters did not support the proposal to collect the new SDOH assessment items and noted significant concerns about the cumulative collection burden for critically ill patients, the cost of updating the data collection systems, and training staff members. One of these commenters noted that asking the proposed SDOH assessment items will increase burden on their only discharge planner and reduce the time they can spend on actual discharge planning. Another one of these commenters noted that their facility already has concerns with the high administrative burden of LCDS data collection and its impact on patient care, particularly considering ongoing workforce challenges.
Response: We acknowledge the addition of four new SDOH assessment items will increase the burden associated with completing the LCDS, and we carefully weighed the burden of collecting new assessment items against the benefits of adopting those assessment items for the LCDS. We prioritized balancing the reporting burden for LTCHs with our policy objective to collect additional SDOH standardized patient assessment data elements that will inform care planning and coordination and quality ( print page 69588) improvement across care settings. We interpret the commenters who acknowledged the increase in burden associated with collecting these four new assessment items while still supporting the proposal to recognize this important balance. We interpret the comment regarding the reduction of burden associated with the adoption of consistent, standardized questions to be supportive of the proposal to adopt four new SDOH assessment items from the AHC HRSN Screening Tool since implementing standardized questions across all LCDS will be easier on staff administering the assessment.
As we noted in section IX.E.4.b., the proposed new SDOH assessment items were identified in either the 2016 NASEM report [ 781 ] or the 2020 NASEM report [ 782 ] as impacting care use, cost, and outcomes for Medicare beneficiaries. In addition, Healthy People 2030 [ 783 ] and related work across HHS [ 784 ] underscores that social risk factors and unmet social needs contribute to wide health and health care disparities and inequities. Stakeholders across the health care spectrum have a role to play in addressing SDOH. We believe by integrating the proposed new SDOH assessment items into routine practice and, when indicated, facilitating referrals to downstream interventions informed by patient data, then the risk for negative outcomes, such as hospital readmissions, can be reduced. Collection of these new SDOH items will provide key information to LTCHs to support effective discharge planning. Therefore, we hope that the proposed new SDOH assessment items, when collected at admission, can inform the discharge planning process for LTCHs, reducing discharge planning burden overall, rather than negatively impacting the time LTCHs spend on discharge planning.
In response to the commenters with concerns about the cumulative collection burden on critically ill patients, we interpret the comments to be referring to LTCH patients on admitted on ventilators or other critically ill LTCH patients who may be unable to respond to interview questions at the time of admission. We note that we did propose these new and modified SDOH items to include additional response options for patients that decline to respond or are unable to respond ( 89 FR 36347 through 36350 ). We encourage LTCHs to assess all patients and select the appropriate response options for the SDOH items on the LCDS.
In response to the commenters with concerns about the cost of updating the data collection systems, CMS continually looks for opportunities to minimize the cost to LTCHs associated with collection and submission of the LCDS through strategies that simplify collection and submission requirements. This includes standardizing instructions, providing a help desk, hosting a dedicated web page, communication strategies, free data specifications, and free on-demand reports.
Finally, in response to the commenters with concerns about training their staff on collecting the proposed new SDOH assessment items, we plan to provide training resources in advance of the initial collection of the assessment items to ensure that LTCHs have the tools necessary to administer the new SDOH assessment items in a respectful way and reduce the burden to LTCHs in creating their own training resources. These training resources may include online learning modules, tip sheets, questions and answers documents, and recorded webinars and videos, and would be available to providers as soon as technically feasible, allowing LTCHs several months to ensure their staff take advantage of the learning opportunities.
Comment: Three commenters who did not support the proposal suggested that the proposed SDOH assessment items need further testing and refinement to ensure they work as intended in the LTCH setting, and referred to the CMS second evaluation of the AHC Model from 2018 through 2021 as evidence of this suggestion. [ 785 ] These commenters interpret the Findings at a Glance to conclude that the AHC HRSN Screening Tool “did not appear to increase beneficiaries' connection to community services or HRSN resolution.”
Response: The HRSN domains in the AHC HRSN Screening Tool were chosen based upon literature review and expert consensus utilizing the following criteria: (1) availability of high-quality scientific evidence linking a given HRSN to adverse health outcomes and increased healthcare utilization, including hospitalizations and associated costs; (2) ability for a given HRSN to be screened and identified in the inpatient setting prior to discharge, addressed by community-based services, and potentially improve healthcare outcomes, including reduced readmissions; and (3) evidence that a given HRSN is not systematically addressed by healthcare providers. [ 786 ] In addition to established evidence of their association with health status, risk, and outcomes, these domains were selected because they can be assessed across the broadest spectrum of individuals in a variety of settings. [ 787 788 ]
The commenters also referred to the two-page summary of the AHC Model 2018-2021 [ 789 ] which describes the results of testing whether systematically identifying and connecting beneficiaries to community resources for their HRSNs improved health care utilization outcomes and reduced costs. To ensure consistency in the screening offered to beneficiaries across both an individual community's clinical delivery sites and across all the communities in the model, CMS developed a standardized HRSN screening tool. This AHC HRSN Screening Tool was used to screen Medicare and Medicaid beneficiaries for core HRSNs to determine their eligibility for inclusion in the AHC Model. If a Medicare or Medicaid beneficiary was eligible for the AHC Model, they were randomly assigned to one of two tracks: (1) Assistance; or (2) Alignment. The Assistance Track tested whether navigation assistance that connects navigation-eligible beneficiaries with community services results in increased HRSN resolution, reduced health care expenditures, and unnecessary utilization. The Alignment Track tested whether navigation ( print page 69589) assistance, combined with engaging key stakeholders in continuous quality improvement (CQI) to align community service capacity beneficiaries' HRSNs, results in greater increases in HRSN resolution and greater reductions in health expenditures and utilization than navigation assistance alone. Regardless of assigned track, all beneficiaries received HRSN screening, community referrals, and navigation to community services. [ 790 ]
We believe the commenter inadvertently misinterpreted the findings, believing these findings were with respect to the effectiveness and scientific validity of the AHC HRSN Screening Tool itself. The findings section of this two-page summary described six key findings from the AHC Model, which examined whether the Assistance Track or the Alignment Track resulted in greater increases in HRSN resolution and greater reductions in health expenditures and utilization. Particularly, the AHC Model reduced emergency department visits among Medicaid and FFS Medicare beneficiaries in the Assistance Track, which was suggestive that navigation may help patients use the health care system more effectively. We acknowledge that navigation alone did not increase beneficiaries' connection to community services or HRSN resolution, and this was attributed to gaps between community resource availability and beneficiary needs. The AHC HRSN Screening Tool use in the AHC Model was limited to identifying Medicare and Medicaid beneficiaries with at least one core HRSN who could be eligible to participate in the AHC Model. Our review of the AHC Model did not identify any issues with the validity and scientific reliability of the AHC HRSN Screening Tool.
Finally, as part of our routine item and measure monitoring work, we continually assess the implementation of new assessment items, and we will include the four new proposed SDOH assessment items in our monitoring work.
Comment: Three commenters requested we articulate our vision for how we plan to use the data collected from the SDOH standardized patient assessment data elements in quality and payment programs. These commenters noted concern that CMS may use the SDOH assessment data to develop an LTCH QRP measure that would hold LTCHs solely accountable to address the social drivers of health that require resources and engagement across an entire community.
Response: We proposed the four new SDOH assessment items because collection of additional SDOH items would permit us to continue developing the statistical tools necessary to maximize the value of Medicare data and improve the quality of care for all beneficiaries. For example, we recently developed and released the Health Equity Confidential Feedback Reports, which provided data to LTCHs on whether differences in quality measure outcomes are present for their patients by dual-enrollment status and race and ethnicity. [ 791 ] We note that advancing health equity by addressing the health disparities that underlie the country's health system is one of our strategic pillars [ 792 ] and a Biden-Harris Administration priority. [ 793 ] Furthermore, any updates to the LTCH QRP measure set or payment system would be addressed through future notice-and-comment rulemaking, as necessary.
Comment: Five commenters were concerned that the proposed SDOH assessment items are not applicable to LTCH patients because many LTCH patients are generally unable to respond to questioning due to mechanical ventilation or sedation and are more severely ill than the average Medicare beneficiary for which the AHC HRSN Screening Tool was developed. Two of these commenters do not think LTCHs are the appropriate reporting and accountability entity for the SDOH assessment items unless the items are used in patient reporting or case-mix adjustment of measures or the healthcare entity can redress the disadvantage because LTCHs generally see patients at the end of the care continuum and have very little control over the SDOH of patients. These commenters were also concerned with the stigma patients may feel when they are asked about their living situation, and food and utilities availability, and the potential risk of violence against their staff due to having to ask sensitive and repetitive questions.
Response: We believe the proposed SDOH assessment items are important to collect on all LTCH patients. We acknowledge that many patients are admitted to LTCHs on mechanical ventilation. However, based on our internal analysis of LTCH data reported from October 1, 2021 through September 30, 2023 (Quarter 4 of CY 2021 through Quarter 3 CY 2023), over 70% of patients were not on invasive mechanical ventilation support when they were admitted to an LTCH. While we acknowledge the medical complexity of LTCH patients, we believe LTCHs are accustomed to working with patients with very complex medical conditions, including those who are on mechanical ventilation, sedated, or severely ill, and we are confident in their ability to collect this data in a consistent manner. There are currently several patient interview assessment items on the LCDS, and LTCHs are accustomed to administering these questions to impaired patients. In addition, the new and modified assessment items we proposed include additional response options for patients that decline to respond or are unable to respond ( 89 FR 36347 through 36350 ) We encourage LTCHs to assess all patients and select the appropriate response options for the SDOH.
In response to the commenters that stated LTCHs are not the appropriate entity for these SDOH assessment items because LTCHs generally see patients at the end of the care continuum and have very little control over the SDOH for that reason, we refer the commenters to section IX.E.4.b of this final rule. In section IX.E.4.b of this final rule, we noted that the assessment items we proposed to collect as standardized patient assessment data elements under the SDOH category were identified in the 2016 NASEM report [ 794 ] or the 2020 NASEM report [ 795 ] as impacting care use, ( print page 69590) cost, and outcomes for Medicare beneficiaries. These items have the potential to affect treatment preferences and goals of patients and their caregivers, and therefore will support LTCHs in implementing an effect discharge planning process. The discharge planning process requires that the LTCH must identify, at an early stage of hospitalization, those patients who are likely to suffer adverse health consequences upon discharge in the absence of adequate discharge planning and must provide a discharge planning evaluation for those patients so identified.
Finally, we respectfully disagree that the proposed SDOH assessment items are inherently stigmatizing. We understand the potentially sensitive nature of these questions, and we want patients to feel comfortable answering the questions. We plan to provide training resources in advance of the initial collection of the assessment items to ensure that LTCHs have the tools necessary to administer the new SDOH assessment items in a respectful way and reduce the burden to LTCHs in creating their own training resources. These training resources may include online learning modules, tip sheets, questions and answers documents, and recorded webinars and videos, and would be available to providers as soon as technically feasible, allowing LTCHs several months to ensure their staff take advantage of the learning opportunities. Finally, as previously noted, we proposed that these new and modified SDOH items include response options for patients that decline to respond or are unable to respond ( 89 FR 36347 through 36350 ).
Comment: Two commenters noted the opportunities to advance interoperability through the adoption of the items. One of these commenters supported the proposal to adopt four SDOH assessment items as standardized patient assessment data elements, but encouraged CMS to ensure that the data generated by the LCDS is interoperable with existing screening standards, like the Gravity Project, to ensure that consumers are not asked multiple times for the same information. The other commenter encouraged CMS to consider supporting data portability and screening interoperability across acute hospitals, LTCHs, and other PAC settings to avoid unnecessary duplication of screenings and assessments. This commenter recognized that repeating screenings and assessments at appropriate intervals can support the identification of emerging or changing needs, but also noted that duplication may lead to patient mistrust.
Response: We appreciate the statements from commenters encouraging CMS to support data portability and screening interoperability. As we have noted in the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28122 and 28123 ), to further interoperability in post-acute care settings, CMS and the Office of the National Coordinator for Health Information Technology (ONC) participate in the Post-Acute Care Interoperability Workgroup (PACIO) to facilitate collaboration with interested parties to develop Health Level Seven International® (HL7) Fast Healthcare Interoperability Resource® (FHIR) standards. These standards could support the exchange and reuse of patient assessment data derived from the post-acute care (PAC) setting assessment tools, such as the Minimum Data Set (MDS), Inpatient Rehabilitation Facility—Patient Assessment Instrument (IRF-PAI), LCDS, Outcome and Assessment Information Set (OASIS), and other sources. The CMS Data Element Library (DEL) [ 796 ] continues to be updated and serves as a resource for PAC assessment data elements, as well as furthers CMS' goal of data standardization and interoperability. We acknowledge that there are still opportunities to advance these goals, and we will take these comments into consideration.
We find the commenter's statement unclear that repeating screenings and assessments may lead to patient mistrust.We interpret the commenter to mean that patients may become concerned that the LTCH does not have the necessary information to appropriately care for the patient if the LTCH asks similar questions as the previous healthcare setting. We disagree and believe that patient trust can be strengthened when interview questions are introduced appropriately by the LTCH clinical staff, including explain the reason for asking the question again. We note that LTCH patients may experience changing needs over the course of their hospital stay. For example, some patients may have been housing secure prior to their condition, but their prior living situation may no longer be suitable for their current needs, which may include specific requirements such as mobility equipment.
Comment: Three commenters did not agree with CMS that the proposed SDOH assessment items would produce interoperable data within Medicare's quality reporting programs because the proposed requirements for LTCH are not aligned with the SDOH screening requirements in the Hospital IQR Program and IPFQR Program. Specifically, these commenters noted that the Screening for SDOH measures in the Hospital IQR and IPFQR Programs do not specify when a patient is screened (for example, at admission) nor how the screening questions are asked (that is, specific wording and responses). Instead, these providers are only asked to document that a patient was screened for the following domains: housing instability, food insecurity, transportation difficulties, utility assistance needs, and interpersonal safety concerns. These commenters contrast requirements for reporting the Screening for SDOH measures with our proposal to add assessment items to the LCDS with standardized questions and responses and would require screening at admission.
Response: We disagree that the proposed collection of four new SDOH assessment items and one modified SDOH assessment item for the LTCH QRP and the requirements for the Hospital IQR and IPFQR Programs do not promote standardization. Although hospitals and IPFs participating in these programs can use a self-selected SDOH screening tool, the Screening for SDOH and Screen Positive Rate for SDOH measures we have adopted for the Hospital IQR and IPFQR Programs address the same SDOH domains that we proposed to collect as standardized patient assessment data under the LTCH QRP: housing instability, food insecurity, utility difficulties, transportation needs. We believe that this partial alignment will facilitate longitudinal data collection on the same topics across healthcare settings. As we continue to standardize data collection, we believe using common standards and definitions for new assessment items is important to promote interoperable exchange of longitudinal information between LTCHs and other providers to facilitate coordinated care, continuity in care planning, and the discharge planning process. This is evidenced by our recent proposals to add these four SDOH assessment items and one modified SDOH assessment item in the SNF QRP ( 89 FR 23462 through 23468 ), IRF QRP ( 89 FR 22275 through 22280 ), ( print page 69591) and Home Health QRP ( 89 FR 55383 through 55388 ).
Comment: Two commenters specifically supported the proposal to adopt the Living Situation assessment item as a standardized patient assessment data element in the LCDS. These commenters emphasized that having information on living situation is critical for understanding social and environmental factors that affect their patient' health outcomes. One of these commenters suggested that having information related to a patient's living situation would enable LTCHs to better understand the social and environmental factors that impact their patient's outcomes. They also agree with CMS that it will support LTCHs in their efforts to partner with community care hubs and community-based organizations (CBOs) to tailor transitions of care plans and ensure that patients are able to access resources from community providers. The other commenter also highlighted how understanding the patient's living situation can ensure patients' adaptive equipment needs are addressed.
Response: We thank the commenters and agree that the collection of the Living Situation item will support LTCHs in collecting information to integrate into their admission and discharge processes in order to facilitate partnerships with community care hubs and community-based organizations, continuity in care planning, and their discharge planning process.
Comment: A commenter noted that the cognitive function of patients might not allow them to accurately recall their living situation prior to being in the LTCH. They also noted the possibility that patients would be confused with the item which asks the person to identify their living situation “today.” This commenter suggested that, depending on the person's condition or cognitive status, they may not be able to recall or determine this, nor might they be able to say whether they will be able to return to the living situation they had before their illness or injury.
Response: We thank the commenters for their input. We acknowledge the complex medical conditions of most LTCH patients. However, there are other patient interview assessment items that LTCHs are already collecting, and we believe LTCHs have experience in managing these complex scenarios successfully in order to obtain the information required. We encourage LTCHs to assess all patients and select the appropriate response options for the SDOH, and remind commenters that we specifically proposed additional response options for patients that are unable to respond or decline to respond to the Living Situation item ( 89 FR 36347 ).
Comment: Another commenter recommended that CMS simplify the responses for the Living Situation assessment item because they are likely to lead to confusion. This commenter suggested that CMS align the responses for the Living Situation assessment item with the proposed Food assessment item that has a “Often true,” “Sometimes true,” and “Never true” response option or the modified Transportation assessment item that has a “Yes” or “No” response. They believe this would be simpler for patients to answer and easier on the LTCH staff to collect the information.
Response: We agree that standardized patient assessment data elements should be easy to understand and have clear response options. However, we believe that including the specific distinction in the Living Situation item's response options is needed. Specifically, we believe that additional response options to indicate whether a patient is worried about their living situation in the future helps reduce ambiguity for patients who may only have temporary housing. For example, having a “Yes” and “No” response and eliminating an option for “I have a place to live today, but I am worried about losing it in the future” would not capture those patients that may be at risk of losing their place to live due to lost income because of the traumatic injury or event precipitating their admission to the LTCH. Identifying these patients who are worried about losing their housing in the future would help LTCHs facilitate discharge planning and make the appropriate community referrals.
Comment: Three commenters supported the collection of the two proposed Food assessment items because of the importance of nutrition and food access to LTCH patients' health outcomes, and the usefulness of this information for treatment and discharge planning. One of these commenters commended CMS for acknowledging within the proposed rule how older adults grappling with food insecurity experience lower dietary quality, placing them at nutritional risk. The commenter acknowledged that inadequate access to nutritious food elevates the risk of health issues such as malnutrition, diabetes, and cardiovascular diseases, and when issues with access to food can be addressed, patients may experience better health outcomes and enhanced quality of life. Another one of these commenters noted that information from the two proposed Food assessment items can give healthcare professionals a greater understanding of a patient's complex needs and improve coordination with other healthcare providers, such as dieticians, or referring patients to SNAP and food pharmacies to increase access to healthy foods. Finally, the other commenter noted that the responses to the proposed Food assessment items would help providers incorporate treatment strategies that may be necessary to address a patient's ability to physically access food sources.
Response: We thank the commenters for their responses and we agree that an individual's access to food affects their health outcomes and risk for adverse events. Understanding the potential needs of patients admitted to LTCH through the collection of the two proposed Food assessment items can help LTCHs facilitate resources for LTCH patients, if indicated, when discharged.
Comment: Several commenters expressed concerns that the proposed Food assessment items ask patients to rate the frequency of their food shortage using a three-point scale, which is inconsistent with other questions on the LCDS such as the patient mood, behavioral symptoms, and daily preference assessment items, which use a four-point scale to determine frequency. This commenter suggested this inconsistency may lead to confusion for staff and patients.
Response: We clarify that the proposed Food assessment items include three frequency responses in addition to response options in the event the patient declines to respond or is unable to respond: (0) Often true; (1) Sometimes true; (2) Never True; (7) Patient to declines to respond; and (8) Patient unable to respond. We acknowledge there are several patient interview assessment items on the LCDS that use a four-point scale, but there are also assessment items on the LCDS that do not use a four-point scale. For example, the Health Literacy (B1300), Social Isolation (D0700), and the Pain Interference with Therapy Activities (J0520) assessment items currently use a five-point scale. We chose the proposed Food assessment items from the AHC HRSN Screening Tool, and they were tested and validated using a three-point response scale.
Since the LCDS currently includes assessment items that use varying ( print page 69592) response scales, we do not believe staff and patients will be confused. We will develop coding guidance for these new assessment items and develop training resources for LTCH staff in advance of the initial collection of the assessment items to ensure LTCHs have the tools necessary to administer the new SDOH assessment items. Additionally, we plan to develop resources LTCH staff can use to ensure patients understand the proposed assessment item questions and response options. For example, CMS developed cue cards to assist LTCHs in conducting the Brief Interview for Mental Status (BIMS) in Writing, the Patient Mood Interview (PHQ-2 to 9), the Pain Assessment Interview, and the Interview for Daily and Activity Preference. [ 797 ]
Comment: A commenter was concerned with the 12-month look back period of the proposed Food assessment items, noting that this broad look back period may capture needs that occurred in the past that have already been resolved. This commenter recommended a three-month look back period instead, to capture true concerns that should inform LTCHs' care and discharge planning.
Response: We disagree that the 12-month look back period for the proposed Food assessment items is too long and that it will not result in reliable responses. We believe a 12-month look back is more appropriate than a shorter, three-month look back period, since it is common for a person's Food situation to fluctuate over time and especially throughout the treatment journey. One study of Medicare Advantage beneficiaries found that approximately half of U.S. adults report one or more HRSNs over four quarters. [ 798 ] However, at the individual level, participants had substantial fluctuations: 47.4 percent of the participants fluctuated between 0 and 1 or more over the four quarters, and 21.7 percent of participants fluctuated between one, two, three, or four or more over the four quarters. The researchers noted that the dynamic nature of individual-level HRSNs requires consideration by healthcare providers screening for HRSNs.
To account for potentially changing Food needs over time, we believe it is important to use a longer lookback window to comprehensively capture any Food needs an LTCH patient may have had, so that LTCHs may consider them in their care and discharge planning.
Comment: Two commenters who support the proposal to add a new Utility assessment item to the LCDS stated that understanding patient's access to utilities is crucial for maintaining good health. Specifically, a commenter pointed out that understanding patients' living situation can ensure appropriate provision of adaptive equipment and engagement with community partners. The other commenter highlighted that access to utilities like electricity, heating, and water are necessary to maintain a safe and healthy living environment. This commenter noted that by collecting information about a patient's utility security at admission, an LTCH may be able to assist their patients in addressing their basic needs by referring them to agencies and programs like the Low-Income Home Energy Assistance Program (LIHEAP) or organizations like community care hubs that are well-positioned to support patients in applying for related assistance programs.
Response: We thank the commenters for their support and agree that patients' utilities needs can affect LTCH patients' health outcomes, and the collection of the proposed Utilities assessment item can equip providers with information to inform care plans and discharge planning.
Comment: A commenter was concerned that the 12-month look back period of the proposed Utility assessment item is too broad to result in reliable or valid responses. Specifically, patients may have difficulty remembering if a relevant event, such as a utility shut off threat, occurred within such a long period or the issue may no longer be valid for the person at time of discharge. In addition, if the patient is experiencing cognitive deficits, they may be unable to provide reliable responses to the Utilities assessment item. This commenter recommended that CMS consider a shorter look back period for the Utilities assessment item and reconsider the inclusion of all utilities, including electric, gas, oil, or water, in the assessment item to truly capture concerns that need to be part of the coordination of an appropriate discharge.
Response: We disagree that the 12-month look back period for the proposed Utility assessment item is too long and that it will not result in reliable responses. We believe a 12-month look back is more appropriate than a shorter, three-month look back period, because an individual's Utilities situation may fluctuate over time and especially throughout the treatment journey. One study of Medicare Advantage beneficiaries found that approximately half of U.S. adults report one or more HRSNs over four quarters. [ 799 ] However, at the individual level, participants had substantial fluctuations: 47.4 percent of the participants fluctuated between 0 and 1 or more over the four quarters, and 21.7 percent of participants fluctuated between one, two, three, or four or more over the four quarters. The researchers noted that the dynamic nature of individual-level HRSNs requires consideration by healthcare providers screening for HRSNs. To account for potentially changing Utilities needs over time, we believe it is important to use a longer look back period to comprehensively capture any Utilities needs an LTCH patient may have had, so that LTCHs may consider them in their care and discharge planning.
We note that LTCHs are accustomed to working with patients with very complex medical conditions, including those with cognitive deficits, and we are confident in their ability to collect this data in a consistent manner. There are currently several patient interview assessment items on the LCDS, and LTCHs are accustomed to administering these questions to impaired patients.
We also believe it is important to capture utility needs across electric, gas, oil, and water services, in order to comprehensively understand patients' access to necessary utility services, especially since patients' needs for utilities may vary depending on their equipment needs at discharge. We note that although we proposed to require the collection of the Utilities item for the LTCH QRP, nothing would preclude LTCHs from choosing to screen their patients for additional SDOH they believe are relevant to their patient population and the community they serve. For example, if it is useful to understand patients' access to a specific type of utility service (for example, water or electricity capacity), LTCHs ( print page 69593) may consider follow-up questions to collect granular information.
After careful consideration of the public comments we received, we are finalizing our proposal to adopt four new items as standardized patient assessment data elements under the SDOH category beginning with the FY 2028 LTCH QRP: one Living Situation item; two Food items; and one Utilities item.
Beginning October 1, 2022, LTCHs began collecting seven standardized patient assessment data elements under the SDOH category on the LCDS. [ 800 ] One of these items, A1250. Transportation, collects data on whether a lack of transportation has kept a patient from getting to and from medical appointments, meetings, work, or from getting things they need for daily living. This item was adopted as a standardized patient assessment data element under the SDOH category in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42587 ). As we discussed in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42586 ), we continue to believe that access to transportation for ongoing health care and medication access needs, particularly for those with chronic diseases, is essential to successful chronic disease management and the collection of a Transportation item would facilitate the connection to programs that can address identified needs.
As part of our routine item and measure monitoring work, we continually assess the implementation of the new SDOH items. We have identified an opportunity to improve the data collection for A1250. Transportation by aligning it with the Transportation category collected in our other programs. [ 801 802 ] Specifically, we proposed to modify the current Transportation item so that it aligns with a Transportation item collected on the AHC HRSN Screening Tool available to the IPFQR and Hospital IQR Programs.
A1250. Transportation currently collected in the LCDS asks: “Has lack of transportation kept you from medical appointments, meetings, work, or from getting things needed for daily living?” The response options are: (A) Yes, it has kept me from medical appointments or from getting my medications; (B) Yes, it has kept me from non-medical meetings, appointments, work, or from getting things that I need; (C) No; (X) Patient unable to respond; and (Y) Patient declines to respond. The Transportation item collected in the AHC HRSN Screening Tool asks, “In the past 12 months, has lack of reliable transportation kept you from medical appointments, meetings, work or from getting things needed for daily living?” The two response options are: (1) Yes; and (2) No. Consistent with the AHC HRSN Screening Tool, we proposed to modify the A1250. Transportation item currently collected in the LCDS in two ways: (1) revise the look back period for when the patient experienced lack of reliable transportation; and (2) simplify the response options.
First, the proposed modification of the Transportation item would use a defined 12-month look back period, while the current Transportation item uses a look back period of six to 12 months. We believe the distinction of a 12-month look back period would reduce ambiguity for both patients and clinicians, and therefore improve the validity of the data collected. Second, we proposed to simplify the response options. Currently, LTCHs separately collect information on whether a lack of transportation has kept the patient from medical appointments or from getting medications, and whether a lack of transportation has kept the patient from non-medical meetings, appointments, work, or from getting things they need. Although transportation barriers can directly affect a person's ability to attend medical appointments and obtain medications, a lack of transportation can also affect a person's health in other ways, including accessing goods and services, obtaining adequate food and clothing, and social activities. [ 803 ] The proposed modified Transportation item would collect information on whether a lack of reliable transportation has kept the patient from medical appointments, meetings, work, or from getting things needed for daily living, rather than collecting the information separately. As discussed previously, we believe reliable transportation services are fundamental to a person's overall health, and as a result, the burden of collecting this information separately outweighs its potential benefit.
For the reasons stated, we proposed to modify A1250. Transportation based on the Transportation item adopted for use in the AHC HRSN Screening Tool and adapted from the PRAPARE tool. The proposed Transportation item asks, “In the past 12 months, has a lack of reliable transportation kept you from medical appointments, meetings, work or from getting things needed for daily living?” The proposed response options are: (0) Yes; (1) No; (7) Patient declines to respond; and (8) Patient unable to respond. A draft of the proposed modified Transportation item [ 804 ] to be adopted as a standardized patient assessment data element under the SDOH category can be found in the Downloads section of the LCDS and LTCH Manual web page at https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-care-data-set-ltch-qrp-manual .
We solicited comment on the proposal to modify the current Transportation item previously adopted as a standardized patient assessment data element under the SDOH category beginning with the FY 2028 LTCH QRP.
We received public comments on this proposal. The following is a summary of the comments we received and our responses.
Comment: Several commenters supported the proposal to modify the Transportation assessment item. Three of these commenters noted various reasons for their support for the new 12-month lookback period, including that it would help clarify the intent of the question, reduce provider burden associated with collecting the information, and identify transportation needs that might fluctuate throughout the year. Two of these commenters supported the simplified response options, noting that it would make it easier for patients to answer the question.
Three commenters also agreed with CMS' proposal to simplify the response options and agreed it would reduce data collection burden. One of these commenters acknowledged the important relationship between transportation and an individual's ability to access food. This commenter noted that having transportation to access nutritious food is important and can improve patient outcomes related to ( print page 69594) chronic conditions, such as diabetes and hypertension.
Finally, several commenters supported the modification to the Transportation item because it would align better with other CMS quality reporting programs which would permit comparability of data between providers and settings, and across patients.
Response: We thank the commenters for their support of the proposed modification of the Transportation assessment item. We agree that simplifying the response options would help streamline the data collection process for both patients and staff collecting the data. We also believe specifying a 12-month look back period would reduce ambiguity for both patients and staff and improve the validity of the data collected.
Comment: A commenter did not support the proposal to modify the Transportation assessment item due to concerns with the 12-month look back period and the simplified response options, “Yes” and “No.” The commenter noted that the responses do not collect information about the frequency of patients' concerns and the reasons why they do not have reliable transportation, which does not allow for nuanced understanding of the patient's transportation needs. They also noted the lack of consideration for patients with a disability that requires special accommodations for transportation. Therefore, this commenter recommended that CMS shorten the look back period to three months and reconsider the reliability and validity of the proposed modifications.
Response: We believe a 12-month look back is more appropriate than a shorter, three-month look back period, since a person's Transportation needs may fluctuate over time and especially throughout the treatment journey. As we have previously noted in an earlier response, a study of Medicare Advantage beneficiaries found that approximately half of U.S. adults report one or more HRSNs over four quarters. [ 805 ] However, at the individual level, participants had substantial fluctuations: 47.4 percent of the participants fluctuated between 0 and 1 or more HRSNs over the four quarters, and 21.7 percent of participants fluctuated between one, two, three, or four or more HRSNs over the four quarters. The researchers noted that the dynamic nature of individual-level HRSNs requires consideration by healthcare providers screening for HRSNs. To account for potentially changing Transportation needs over time, we believe it is important to use a longer lookback window to comprehensively capture any Transportation needs an LTCH patient may have had, so that LTCHs may consider them in their care and discharge planning.
Regarding the comment on the response options and the commenter's concern there is not enough information in the responses, we remind LTCHs that although the proposal would require the collection of the Transportation assessment item at admission, we hope that the interview would cultivate future conversations between LTCHs and their patients about the patient's specific transportation needs, rather than being the only time the LTCH discusses the patient's specific transportation needs. Additionally, LTCHs may collect any additional information they believe relevant for their patient population and to inform their care and discharge planning process.
Finally, in response to the comment that we reconsider the reliability and validity of the proposed modified Transportation item, the AHC HRSN Screening Tool was tested across many care delivery sites in diverse geographic locations across the United States. More than one million Medicare and Medicaid beneficiaries have been screened using the AHC HRSN Screening Tool, which was evaluated psychometrically and demonstrated evidence of both reliability and validity, including inter-rater reliability and concurrent and predictive validity.
After careful consideration of the public comments we received, we are finalizing our proposal to modify the current Transportation item previously adopted as a standardized patient assessment data element under the SDOH category beginning with the FY 2028 LTCH QRP.
In the proposed rule, we solicited input on the importance, relevance, appropriateness, and applicability of each of the concepts under consideration listed in Table IX.E.-02 for future years in the LTCH QRP. The FY 2024 IPPS/LTCH PPS proposed rule ( 88 FR 27150 through 27153 ) included a request for information (RFI) on a set of principles for selecting and prioritizing LTCH QRP measures, identifying measurement gaps, and suitable measures for filling these gaps. Within this proposed rule, we also sought input on data available to develop measures, approaches for data collection, perceived challenges or barriers, and approaches for addressing identified challenges. We refer readers to the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59250 and 59251 ) for a summary of the public comments we received in response to the RFI.
Subsequently, our measure development contractor convened a Technical Expert Panel (TEP) on December 15, 2023 to obtain expert input on future measure concepts that could fill the measurement gaps identified in the FY 2024 RFI. [ 806 ] The TEP also discussed the alignment of PAC and Hospice measures with CMS's “Universal Foundation” of quality measures. [ 807 ]
In consideration of the feedback we received through these activities, we solicited input on three measure concepts for the LTCH QRP (see Table IX.E.-02). One is a composite of vaccinations, [ 808 ] which could represent overall immunization status of patients such as the Adult Immunization Status measure [ 809 ] in the Universal Foundation. A second concept on which we sought feedback is the concept of depression for the LTCH QRP, which may be similar to the Clinical Screening for Depression and Follow-up measure [ 810 ] in the Universal Foundation. Finally, we sought feedback on the concept of pain management.

We received several public comments with feedback on these measure concepts. The following is a summary of the comments we received.
Comments: We received many comments on the vaccination composite measure, and several commenters supported the concept of a vaccination composite measure. One commenter noted that it could improve vaccination rates for those vaccines recommended by the Advisory Committee on Immunization Practices (ACIP), reduce administrative burden through alignment with the Universal Foundation, [ 811 ] and potentially improve immunization rates in PAC settings, including LTCHs. Another commenter noted how the Adult Immunization Status measure is a well-tested and reliable means of assessing routine adult vaccinations recommended by the ACIP, and would emphasize the importance of vaccination as a core prevention intervention, streamline existing adult immunization measures, and provide meaningful data to better assess gaps in vaccine coverage.
Several commenters, however, did not support the idea of adding a composite vaccination measure to the LTCH QRP and noted a number of reasons why it was not a good fit for the LTCH setting. Most of these commenters do not believe the LTCH is the appropriate setting for collecting vaccination rates, and other commenters noted that adding such a measure potentially could increase LTCHs' administrative burden in collecting and reporting the data. Finally, two of these commenters questioned how the information provided by patients would be verified.
Several commenters noted that reporting vaccination rates and documenting patients' vaccine status is more appropriate as a measure in the primary care setting, and suggested the information could be shared with other health care providers. Commenters also suggested that a vaccination composite measure would be of fairly low value and not indicative of the quality of LTCH patient care. Finally, a few commenters noted there are numerous reasons patients may decide to decline vaccinations, including cultural preferences and norms, and these reasons are largely dependent on factors outside of an LTCH's control.
Comments: We received several comments that supported the pain management measure concept, favoring the idea of further probes to identify a patient's depression status in the LTCH QRP. A commenter encouraged aligning a pain management measure with the CDC Clinical Practice Guideline for Prescribing Opioids for Pain since LTCH patients may appropriately need pain medications. Another commenter underscored the importance of understanding the impact of pain on therapy and other daily activities in order to improve the quality of services provided. A commenter agreed with the importance of measuring pain management, but recommended CMS consider appropriate exclusions for patient-reported measures. This commenter also recommended exploring ways to promote pain management for patients who may have challenges communicating.
Several commenters opposed the concept of a pain management measure in the LTCH QRP and provided several reasons why they believe it would not be an appropriate measure concept for the LTCH QRP. Four of these commenters noted that pain is often an unavoidable part of a patient's recovery and is not an indicator for whether the patient is improving. These commenters also explained that any kind of pain management measure would need to consider implications for the use of opioids or other pain medications, to avoid unintentionally incentivizing the use of pain medication. One of these commenters recommended that a pain management measure should not include an expectation of an improvement of pain. Another commenter suggested that a pain management measure may not be necessary since LTCHs currently collect data on the frequency that pain affects a patient's sleep and the frequency of pain interference with a patient's ability to participate with therapy activities and with day-to-day activities in Section J of the LCDS.
Comments : We received several comments on the concept of depression for a future LTCH QRP measure, and over half of these commenters supported the concept and favored the idea of further probes in identifying depression measures in the LTCH QRP. One of these commenters noted that their organization had recently revised its policy priorities to advocate for physical and mental health parity, and therefore supported the work to develop a depression measure for the LTCH QRP. Two of these commenters noted that depression can strongly affect health and quality of life and an LTCH stay can specifically impact a patient both mentally and physically. However, one of these commenters recommended that, in developing a depression measure, CMS carefully consider exclusion criteria and timing of a screen for depression since patients are often admitted to an LTCH on a ventilator.
Several commenters, however, opposed the measure concept of depression for reasons related to potential redundancy in data collection, concern about the lack of resources to treat depression, and the administrative burden of collecting the information. Five of these commenters noted that LTCHs already screen for depression through the Patient Health Questionnaire (PHQ-2 to-9) [ 812 ] on the LCDS and use information in the patient's chart to identify mental health conditions or other behavioral health issues. Several commenters also noted that, since LTCHs already screen for depression, a depression quality measure is not necessary. Three commenters also noted that LTCHs do not generally have psychiatrists or psychologists on staff or available to ( print page 69596) provide the comprehensive services needed to treat behavioral health problems, and one of these commenters expressed concern that adding a measure for depression screening would result in consumers expecting that they should have these resources available. Finally, one of these commenters encouraged CMS to consider all aspects of data collection and reporting when prioritizing the appropriateness of the measures selected for the LTCH QRP, suggesting that collecting the same data as other entities when it is not suited to the LTCH patient population is alignment for alignment's sake without benefit to patients or CMS.
Comments: In addition to comments received on the three measure concepts of pain, depression, and composite vaccinations, we also received several comments recommending other measures for future inclusion. A commenter recommended CMS develop and utilize metrics associated with ventilator, dialysis, wound, and nutritional issues. Two commenters recommended the addition of a Patient Experience of Care/Patient Satisfaction measure, highlighting that patient self-report is the gold standard to assess care quality. Commenters also recommended other measure concepts for development and inclusion in the LTCH QRP, including: a “Needs Navigation” measure for the new Principal Illness Navigation codes [ 813 ] in the 2024 Physician Fee Schedule ( 88 FR 78937 through 78950 ), an Advance Care Planning measure, [ 814 ] LTCH-acquired COVID-19 infection morbidity and deaths, a patient safety structural measure, palliative care access and utilization, and timely and appropriate referral to hospice.
Response: We thank all commenters for responding to this RFI. We will take this feedback into consideration regarding our future measure development efforts for the LTCH QRP.
6. Future LTCH Star Rating System: Request for Information (RFI)
In the proposed rule, we sought feedback on the development of a five-star methodology for LTCHs that can meaningfully distinguish between quality of care offered by LTCHs. We refer readers to the RFI in the proposed rule ( 89 FR 36351 ). Specifically, we invited public comment on the following questions:
1. Are there specific criteria CMS should use to select measures for a star rating system?
2. How should CMS present star ratings information in a way that it is most useful to consumers?
We received several comments in response to this RFI, which are summarized below.
Comments: We received a few comments that offered a wide range of recommendations on the criteria we should use for selecting measures to include in a future LTCH Star Ratings system. Several commenters suggested selecting measures that focus on patient and diagnostic safety outcomes. Three commenters recommended CMS align selected measures with the Universal Foundation measure set and stated they believe a consistent approach will make the rating more understandable for all interested parties. One of these three commenters specifically recommended CMS consider the Universal Foundation PAC Add-On Set, [ 815 ] noting that it would allow CMS to compare quality of care more easily and consistently to other settings. Several commenters recommended utilizing existing measures in the LTCH QRP. However, four commenters disagreed, suggesting the measures available in the LTCH QRP were never selected to create a holistic picture of LTCH care. They stated the measures in the LTCH QRP have been added to the program to achieve disparate goals, ranging from agency priorities to statutory requirements, such as the Improving Medicare Post-Acute Care Transformation Act of 2014 (IMPACT Act). [ 816 ] Therefore, they are concerned that an overall rating may reflect performance on measures that are irrelevant to the reasons a patient is seeking care. Several commenters recommended an LTCH Star Rating system include measures of patient experience. However, other commenters were not in favor of including measures of patient experience, suggesting the reliability would be low since many LTCH patients have undergone traumatic brain injuries and may not be able to respond to patient experience questions in the same way as in general acute care. A commenter suggested an LTCH Star Rating system should align with the CMS Meaningful Measures framework focused on person-centered care, equity, safety, affordability, efficiency, chronic conditions, wellness and prevention, seamless care coordination, and behavioral health.
Other commenters provided more general recommendations, such as selecting measures that matter most to patients and families, and utilizing LTCH-specific measures, such as the Ventilator Liberation Rate measure. Two commenters emphasized the need to minimize the burden of data collection when selecting measures for a star ratings system. Other commenters recommended including measures that focus on nutrition, function, staff turnover, data reported to NHSN, patient reported experiences of discrimination or bias and missed or delayed diagnoses, vaccination, cardiovascular disease, and diabetes.
Comments: We received several comments strongly recommending we engage with patients, caregivers, providers, and specialty societies to inform the development and display of the LTCH Star Rating system. Most of these commenters suggested holding a TEP, while one recommended a listening session. Additionally, three commenters urged CMS to ensure full transparency for consumers, including how scores are converted into ratings and reporting periods for each of the reported measures.
We also received comments about the timeliness of data reported and LTCHs' need for additional reports to support their efforts at improving patient outcomes. Several commenters ( print page 69597) suggested that the age of the LTCH quality measures currently displayed does not represent the current performance of the providers, which patients need to make care decisions. Three of these commenters specifically suggested that LTCHs should receive patient-level results for claims-based measures on a quarterly basis and, without these reports they are limited in their ability to implement tailored improvements to the care they provide. They also note that CMS currently provides this level of information to hospitals.
Several commenters also raised concerns about the limited number of LTCH quality measures and lower patient volumes as compared to other settings with a Star Rating System. These commenters were concerned about CMS' ability to develop an overall star rating that is reliable and valid for consumers. Two of these commenters also highlighted their concern that there would be even more instability in an LTCH five-star rating system, which would undermine confidence in the rating system.
Comments: Commenters also provided feedback for CMS to consider when developing a potential methodology and shared their insights and experience with other CMS Star Ratings Systems. Several commenters recommended accounting for factors that differentiate LTCHs, such as patient characteristics and complexities; whether an LTCH is located within another hospital or is freestanding; and the number of patients admitted requiring dialysis. These commenters were concerned that a future star rating methodology that did not incorporate appropriate “risk adjustment” or weighting methodologies would be ineffective in appropriately differentiating between LTCH providers. Although a commenter recommended aligning the methodology used in an LTCH Star Ratings system with other existing star ratings to help consumers navigate the ratings, a number of commenters shared their concerns about developing an LTCH Star Rating system given what they described as issues with CMS' other star rating systems. Two of these commenters suggested CMS apply lessons learned from the development and maintenance of the existing star ratings programs and urged CMS to allow for a sufficient timeline for development and implementation.
Response: We thank all the commenters for responding to our RFI on this important CMS priority. We will take these recommendations into consideration in our future star rating development efforts.
We refer readers to the regulatory text at 42 CFR 412.560(b) for information regarding the current policies for reporting specified data for the LTCH QRP.
As discussed in section IX.E.4. of this final rule, we proposed to adopt four new items as standardized patient assessment data elements under the SDOH category (one Living Situation item, two Food items, and one Utilities item), and to modify the Transportation standardized patient assessment data elements previously adopted under the SDOH category beginning with the FY 2028 LTCH QRP.
We proposed that LTCHs would be required to report these new items and the modified Transportation item using the LCDS beginning with patients admitted on October 1, 2026 for purposes of the FY 2028 LTCH QRP. Starting in CY 2027, LTCHs would be required to submit data for the entire calendar year for purposes of the FY 2029 LTCH QRP.
We also proposed that LTCHs who submit the Living Situation, Food, and Utilities items proposed for adoption as standardized patient assessment data elements under the SDOH category with respect to admission only would be deemed to have submitted those items with respect to both admission and discharge. We proposed that LTCHs would be required to submit these items at admission only (and not at discharge), because it is unlikely that the assessment of those items at admission will differ from the assessment of the same item at discharge. This would align the data collection for these proposed items with other SDOH items (that is, Race, Ethnicity, Preferred Language, and Interpreter Services) which are only collected at admission. [ 817 ] A draft of the proposed items is available in the Downloads section of the LCDS and LTCH Manual web page at https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-care-data-set-ltch-qrp-manual .
As we noted in Section IX.E.4.e. of this proposed rule, we continually assess the implementation of the new SDOH items, including A1250. Transportation, as part of our routine item and measure monitoring work. We received feedback from stakeholders in response to the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19551 ) noting their concern with the burden of collecting the Transportation item at admission and discharge. Specifically, commenters stated that a patient's access to transportation is unlikely to change between admission and discharge. We analyzed the data LTCHs reported from October 1, 2022 to June 30, 2023 (Q4 CY 2022 through Q2 CY 2023) and found that patient responses did not significantly change from admission to discharge. [ 818 ] Specifically, the proportion of patients [ 819 ] who responded “Yes” to the Transportation item at admission versus at discharge differed by only 1.65 percentage points during this period. We find these results convincing, and therefore we proposed to require LTCHs to collect and submit the proposed modified standardized patient assessment data element, Transportation, at admission only.
We solicited public comment on our proposal to collect data on the following items proposed as standardized patient assessment data elements under the SDOH category at admission beginning October 1, 2026 with the FY 2028 LTCH QRP: (1) Living Situation as described in section IX.E.4.c.(1) of the proposed rule and this final rule; (2) Food as described in section IX.E.4.c.(2) of the proposed rule and this final rule; and (3) Utilities as described in section IX.E.4.c.(3) of the proposed rule and this final rule. We also invited comment on our proposal to submit the proposed modified standardized patient assessment data element, Transportation, at admission only beginning October 1, 2026 with the FY 2028 LTCH QRP as described in section IX.E.4.e. of the proposed rule and this final rule.
We received a number of comments related to our proposals for the collection of the proposed SDOH assessment items. The following is a summary of the comments we received and our responses.
Comment: Two commenters supported the proposed collection of the ( print page 69598) four new SDOH assessment items once, upon admission, noting that it would mitigate the administrative burden of data collection and reduce redundancy.
Response: We appreciate the commenters' support of our proposal to collect the four new SDOH items at admission only. We are mindful of provider burden and appreciate the support from several commenters who agreed that collection upon admission would mitigate the administrative burden of data collection for this item.
Comment: A commenter suggested that CMS offer flexibility for LTCHs on how to collect the proposed SDOH assessment items, rather than requiring LTCHs to use assessment items from the AHC HRSN Screening Tool. This commenter stated they believed CMS' focus should be on whether the information is collected and less on the specific vendor or tool used for collection. Similarly, another commenter encouraged CMS to have flexibility in collection and reporting, such as obtaining data from existing items in electronic medical record systems and case management systems, and allowing caregivers to provide responses to the SDOH assessment items. Another commenter noted that these items are also collected by referring hospitals, and stated it therefore would be duplicative to collect the same information in the LTCH.
Response: We interpret these commenters to be suggesting that CMS should not require LTCHs to use a specific tool to collect the information if it is collected elsewhere, such as in previous healthcare settings, rather than CMS requiring LTCHs to question patients and collect their verbal responses on the LCDS upon the patient's admission to the LTCH.
In response to the comments suggesting CMS should not require LTCHs to use a specific tool to collect the information as long as it is collected, we disagree and believe it is important to collect standardized information. As we continue to standardize data collection across settings, we believe using common standards and definitions for new assessment items is important to promote interoperable exchange of patient information between and within LTCHs and other providers. This will facilitate standardized patient data to enhance coordinated care, continuity in care planning, and the discharge planning process. Section 1899B(b) of the Act already requires LTCHs to collect standardized patient assessment data via the LCDS or another assessment instrument. The proposed and modified SDOH assessment items will be added to a future version of the LCDS, in the same way other standardized patient assessment data elements are collected, which will contribute to further standardized data collection across LTCHs. This is important to supporting our ongoing LTCH QRP initiatives by providing standardized data with which to stratify LTCH's performance on current measures and or in future quality measures.
In response to the comments suggesting LTCHs should be able to utilize information collected in previous healthcare settings, we are intentional in our efforts to increase the patient's voice in the assessment process and the LTCH QRP. Obtaining information about the Living Situation, Food, Utilities, and Transportation assessment items directly from the patient, sometimes called “hearing the patient's voice,” is more reliable and accurate than obtaining it from a health care provider that previously cared for the patient for several reasons: the LTCH would not know whether it was collected from the patient or from a family member or other source; the LTCH would not know how the SDOH domain was defined—for example, whether utilities included electricity, gas, oil, or water or only asked about electricity; and the LTCH would not be able to determine whether the potential problem had been resolved since then. Most importantly, we believe that by asking the patient these questions at admission, it may prompt further discussion with the patient about their needs and help formulate an appropriate discharge care plan. We also clarify that LTCHs may use different methods to collect the information from the patient, if they are consistent with the requirements for these new and modified SDOH items set forth in sections IX.E.4 and IX.E.7.b of this final rule.
Comment: A commenter noted that it would be helpful if SDOH item collection requirements were focused on patients that are being discharged home from the LTCH, because patients who go from LTCHs to IRFs or SNFs could have their SDOH information collected in their next setting of care.
Response: While we understand that some LTCH patients may be transferred to IRFs or SNFs, we believe that it is important to collect the proposed SDOH items at admission to the LTCH. This information may support LTCHs in effective discharge planning. Patients receiving services in an LTCH may have a longer length of stay than in other PAC settings, and therefore, LTCHs may not know whether a patient will be discharged home or transferred to another PAC setting at the time they are admitted to the LTCH. It is also possible that a patient's living situation, food, utilities, and transportation needs could change over the course of their treatment or the patient's discharge plans may change due to lost income as a result of the traumatic injury or event precipitating their admission to the LTCH. Collecting this information at admission equips the LTCH to adjust their discharge plans as needed.
Comment: A commenter encouraged CMS to ensure this requirement includes that these data elements be standardized and documented in patients' medical records.
Response: We thank the commenter for their support and input. We proposed these SDOH assessment items as standardized patient assessment data elements to ensure they are standardized. The Living Situation, Food, and Utilities assessment items will be collected on the LCDS and electronically submitted to CMS' data submission system, in the same way other standardized patient assessment data elements are collected. Additionally, we recommend that an LTCH maintain the original LCDS as part of the patient's medical record.
Comment: Four commenters offered suggestions or recommendations for guidance related to collecting the proposed SDOH assessment items. Three of these commenters recommended that CMS include coding logic to allow skipping the Utilities assessment item if a patient indicated that they do not have a steady place to live, since it would be inappropriate to ask about utilities if a patient has no place to live. One of the commenters asked CMS to work with LTCHs to ensure the data are collected in a respectful and person-centered way, and encouraged CMS to educate and build trust with beneficiaries on why LTCHs are collecting this data. This commenter also encouraged CMS to work with the National Committee for Quality Assurance and other organizations to develop frameworks, workflows, and guidance to collect this data.
Response: We acknowledge that the proposed SDOH assessment items require the patient to be asked potentially sensitive questions. We will provide training materials and guidance for LTCH staff to collect the information from LTCH patients on these new and modified SDOH items. However, we decline the recommendation to add a skip pattern if a patient responds to the Living Situation item that they either have a steady place to live today, but are worried about losing it in the future (Response 2) or they do not have a steady place to live (Response 3). We are ( print page 69599) concerned that patients that provide such responses may live somewhere, temporarily, that may or may not have adequate utilities. For example, a patient may be living in temporary housing that does or does not have adequate utilities, or their situation may be that they have electricity but no running water. Therefore, we believe that asking both questions of every patient (that is, the Living Situation and Utilities items) provides a more complete assessment for LTCHs to use in their discharge planning. We also note that we proposed a response option for patients that decline to respond for each of the new and modified SDOH items ( 89 FR 36347 through 36350 ).
Comment: Several commenters suggested that CMS consider adopting additional items from the AHC HRSN Screening Tool in the LTCH QRP, especially those addressing disability and financial strain. These commenters noted that both factors can affect patient safety outcomes or be HRSNs that contribute to bias. Additionally, another commenter suggested assessing family caregiver burden and whether referrals resulted in actual service delivery, both of which can be a factor in both the patient's health and the use of emergency department visits or hospitalization.
Response: We appreciate the comments and suggestions provided by the commenters, and we agree that it is important to understand the needs of patients with disabilities. While disability is not currently assessed through the LCDS, it is comprehensively assessed as part of existing protocols around care plans and health goals. However, as we continue to evaluate SDOH standardized patient assessment data elements, we will consider this feedback. We note that, although we proposed to require the collection of these new and modified SDOH items for the LTCH QRP, nothing would preclude LTCHs from choosing to screen their patients for additional SDOH they believe are relevant to their patient population and the community they serve, including screening for lack of financial strain and caregiver burden. For example, the AHC HRSN Screening Tool includes questions for eight supplemental domains, including financial strain.
After careful consideration of the public comments we received, we are finalizing our proposal to collect data on the following items adopted as standardized patient assessment data elements under the SDOH category at admission only beginning with October 1, 2026 LTCH admissions: (1) Living Situation as described in section IX.E.4(c)(1) of this final rule; (2) Food as described in section IX.E.4(c)(2) of this final rule; and (3) Utilities as described in section IX.E.4(c)(3) of this final rule. We are also finalizing our proposal to collect the modified standardized patient assessment data element, Transportation, at admission only beginning with October 1, 2026 LTCH admissions as described in section IX.E.4(e) of this final rule.
Since the FY 2012 IPPS/LTCH PPS final rule, LTCHs have collected information for the LTCH QRP utilizing the LCDS. [ 820 ] Since 2012, the LTCH QRP has evolved in response to both quality initiatives and statutory requirements, and as a result, the LCDS has evolved to support data collection for evaluation of health outcomes in the LTCH. The LCDS Version 5.0 was implemented on October 1, 2022, and is currently in use. [ 821 ]
As specified in the LCDS Manual, the LCDS Admission assessment has a maximum three-day assessment period, beginning with the date of admission, in which the patient's assessment must be conducted to obtain information for the LCDS Admission assessment items. All LTCHs are required to record the Assessment Reference Date (ARD) (A0210) on each LCDS, which is defined as the end point of the assessment period for the LCDS assessment record. LTCHs can set their own ARD, as long as it is no later than the third calendar day (date of admission plus two calendar days) of the patient's stay.
We continually look for opportunities to minimize LTCHs' burden associated with collection of the LCDS through strategies that include improving communication and conducting outreach with users, as well as simplifying collection and submission requirements. In recent years, we have received feedback regarding the difficulty of collecting the required LCDS data elements within the three-day assessment window when medically complex patients are admitted prior to and on weekends. On October 17th, 2023, our measure development contractor hosted an LTCH Listening Session on the Administrative Burden of the LTCH QRP, and invited providers to comment on several LTCH QRP topics, including a potential expansion of the assessment period to four days. [ 822 ] During the listening session, we received support for revising the Admission assessment window, with participants suggesting that extending the assessment window would ease the difficulties noted above.
We proposed to extend the Admission assessment period from three days to four days, beginning with LTCH admissions on October 1, 2026. For example, if a patient was admitted on Friday, October 19, the ARD for the LCDS Admission assessment could be no later than Monday, October 22. This change to the assessment period would only apply to the LCDS Admission assessment and have no impact on burden.
We solicited public comment on our proposal to extend the LCDS Admission assessment window from three to four days beginning with the FY 2028 LTCH QRP.
Comment: We received support from all interested parties who commented on our proposal for modifying the LTCH Admission assessment window from three days to four days. Several commenters noted that patient assessments are extremely time consuming, and support the extension, especially when medically complex patients are transferred to an LTCH during an evening or weekend. Several commenters stated that an additional day to conduct a patient assessment will ease the administrative burden associated with completing assessments on their workforce, particularly for patients admitted on a Friday or Saturday. Two of these commenters noted their appreciation that CMS considered the comments from interested parties and acted to ease some of the administrative burden of completing the LCDS.
Response: We thank commenters for their support to modify the assessment window from three days to four days and for recognizing that our proposal is in response to LTCHs' requests to reconsider the admission assessment ( print page 69600) window. We are also pleased to hear that many LTCHs will find this modification supports their admission process, especially when a patient is admitted on a Friday or Saturday. As part of our routine item and measure monitoring work, we continually assess the implementation of the LCDS to look for opportunities to improve and streamline the data collection process.
Comment: We received several comments requesting that the proposed modification to the assessment window be implemented earlier than the proposed date of October 1, 2026, and three commenters requested this change be implemented on October 1, 2024. Two other commenters requested it be implemented as soon as possible, especially since CMS has acknowledged that completing the assessment within three days of admission is a burdensome requirement, and believes CMS has confirmed through its proposal that a four-day assessment window for completing the LCDS is feasible for the agency.
Response: We acknowledge the commenters' requests that we implement the modification earlier than October 1, 2026 and understand that completing the LCDS within three days of admission imposes some burden on LTCHs. However, our proposal to modify the Admission assessment window does not decrease the overall burden of collecting the data in the LCDS. With this proposed modification, LTCHs would have more time to collect the same data in a response to LTCHs' concerns. However, it is not feasible for us to implement this change earlier than the proposed date of October 1, 2026 for the FY 2028 LTCH QRP. Any modification to the LCDS has downstream logistical implications. For example, CMS has already finalized and published the LCDS 5.1 item set that will be effective October 1, 2024, approximately 12 months early, to allow providers adequate time for preparation. The LCDS Manual Version 5.1 and LTCH data specifications V4.00.1 were published over 7 months early on February 1 and February 14, 2024, respectively. Additionally, we typically follow a 2-year cycle of updates/modifications to the item sets. We proposed that this modification be effective beginning with patients admitted on October 1, 2026 for FY 2028 LTCH QRP because it is the earliest feasible date to implement this modification.
After careful consideration of the public comments we received, we are finalizing our proposal to modify the LCDS Admission assessment from three to four days beginning with the FY 2028 LTCH QRP.
As described in the proposed rule, we did not propose any new policies regarding the public display of measure data at this time. For a more detailed discussion about our policies regarding public display of LTCH QRP measure data and procedures for the opportunity to review and correct data and information, we refer readers to the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57231 through 57236 ).
Sections 1886(b)(3)(B)(ix) and 1814(l)(4) of the Social Security Act (as amended by the Health Information Technology for Economic and Clinical Health Act, Title XIII of Division A and Title IV of Division B of the American Recovery and Reinvestment Act of 2009, Pub. L. 111-5 ) authorize downward payment adjustments under Medicare, beginning with fiscal year (FY) 2015 for eligible hospitals and CAHs that do not successfully demonstrate meaningful use of certified electronic health record technology (CEHRT) for the applicable electronic health record (EHR) reporting periods. Section 602 of Title VI, Division O of the Consolidated Appropriations Act, 2016 ( Pub. L. 114-113 ) added subsection (d) hospitals in Puerto Rico as eligible hospitals under the Medicare EHR Incentive Program and extended the participation timeline for these hospitals such that downward payment adjustments were authorized beginning in FY 2022 for section (d) Puerto Rico hospitals that do not successfully demonstrate meaningful use of CEHRT for the applicable EHR reporting periods.
The Medicare Promoting Interoperability Program encourages healthcare data exchange for public health purposes through the Public Health and Clinical Data Exchange objective. In the FY 2023 IPPS/LTCH PPS final rule, we finalized the requirement for eligible hospitals and CAHs to report the AUR Surveillance measure with a modification to begin reporting with the EHR reporting period in CY 2024 ( 87 FR 49337 ). Under the AUR Surveillance measure, eligible hospitals and CAHs are required to report two kinds of data to the Centers for Disease Control and Prevention (CDC) National Healthcare Safety Network (NHSN): Antimicrobial Use (AU) data and Antimicrobial Resistance (AR) data ( 87 FR 49335 ). Separate data elements and technical capabilities are required for reporting the AU data and AR data, and we refer readers to the CDC NHSN AUR protocols for technical details regarding implementation. [ 823 ] Eligible hospitals and CAHs that report a “yes” response indicate that they have submitted data for both AU and AR, and will receive credit for reporting the measure, unless they claim an exclusion for which they are eligible. Eligible hospitals and CAHs must also use technology certified to the criterion at 45 CFR 170.315(f)(6) , “Transmission to public health agencies—antimicrobial use and resistance reporting” for data submission ( 87 FR 49337 ).
After finalizing the AUR Surveillance measure, we received feedback from some eligible hospitals and CAHs seeking clarity regarding reporting requirements and exclusion eligibility for eligible hospitals and CAHs. Comments and questions included whether eligible hospitals or CAHs with an applicable exclusion preventing their participation in reporting either AU data or AR data were required or able to report any available data to receive credit under the AUR Surveillance measure. Under this policy, if an eligible hospital or CAH meets the exclusion criteria with respect to reporting either AU data or AR data, the hospital is excluded from the entire AUR Surveillance measure ( 87 FR 49337 ).
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36352 through 36353 ), in collaboration with the CDC, we identified the need to separate the AUR Surveillance measure into two measures, to clarify reporting requirements and to incentivize greater data reporting from eligible hospitals and CAHs. In addition, because AU and AR reporting rely on different data sources, such as an electronic medication administration record (eMAR) or bar-coded medication administration (BCMA) for AU, and lab information systems (LISs) for AR, we discussed how separating the measure into two measures will more ( print page 69601) appropriately target the availability of exclusions for participants who have difficulty with data transmission using a single data source.
Specifically, we proposed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36352 through 36353 ) to separate the AUR Surveillance measure into two measures, beginning with the EHR reporting period in CY 2025:
- AU Surveillance measure: The eligible hospital or CAH is in active engagement with CDC's NHSN to submit AU data for the selected EHR reporting period and receives a report from NHSN indicating its successful submission of AU data for the selected EHR reporting period.
- AR Surveillance measure: The eligible hospital or CAH is in active engagement with CDC's NHSN to submit AR data for the selected EHR reporting period and receives a report from NHSN indicating its successful submission of AR data for the selected EHR reporting period.
Under the AU Surveillance measure, eligible hospitals and CAHs would be required to report AU data to CDC's NHSN. Under the AR Surveillance measure, eligible hospitals and CAHs would also be required to report AR data to CDC's NHSN. Eligible hospitals and CAHs would be required to report a “yes” response or claim an applicable exclusion, separately, to receive credit for reporting on the AU Surveillance measure and the AR Surveillance measure. For both measures, eligible hospitals and CAHs would be required to use technology certified to the Office of the National Coordinator for Health Information Technology (ONC) Certification Program for Health Information Technology (health IT) certification criterion at 45 CFR 170.315(f)(6) , “Transmission to public health agencies—antimicrobial use and resistance reporting,” as they are for the AUR Surveillance measure. We believe that separating the AUR Surveillance measure into two measures would encourage participation from eligible hospitals and CAHs that could report data for only the AU Surveillance measure or for only the AR Surveillance measure that might previously have been excluded because of their inability to report both AU data and AR data as required by the AUR Surveillance measure.
Under the requirements for the AUR Surveillance measure, eligible hospitals and CAHs that meet one of the exclusion criteria with respect to reporting data of one kind (for example, AR), are excluded from all AUR Surveillance measure reporting requirements, even if they could report data of the other kind (for example, AU). Offering an exclusion based on an eligible hospital's or CAH's inability to report only one kind of data results in eligible hospitals and CAHs being unable to report on the entire AUR Surveillance measure, even when they could report either AU or AR data. This result is contrary to the goals of the Public Health and Clinical Data Exchange objective because it discourages the sending of partial data as available. Separating the single AUR Surveillance measure into two measures better reflects the reality that AU data reporting and AR data reporting rely on different data sources that require different types of exclusions to reflect the separate clinical and data domains of prescribing and microbiological testing. Separation of AU data reporting and AR data reporting into two measures also supports the Medicare Promoting Interoperability Program's administrative requirements with respect to scoring, because the scoring approach for the Public Health and Clinical Data Exchange objective does not grant partial credit for reporting on individual measures. We note that separating the AUR Surveillance measure into two measures does not expand on the previously finalized requirements of the measure. Separating one measure into two measures allows eligible hospitals and CAHs the opportunity to submit data for either AU or AR if the eligible hospital or CAH can only submit data for one of the two, versus an all or nothing approach.
We invited public comment on our proposal to separate the AUR Surveillance measure into two measures, AU Surveillance and AR Surveillance, beginning with the EHR reporting period in CY 2025.
Comment: Many commenters supported our proposal to split the AUR Surveillance measure into two measures for a variety of reasons. Several commenters supported the change because they stated it would increase the number of eligible hospitals and CAHs that could report on one of the measures, or conversely, it could reduce the number of facilities that are excluded from reporting on the existing singular measure. Several commenters appreciated separating the single measure into two measures because it allows eligible hospitals and CAHs to submit data for either measure versus an all or nothing approach given the different technical requirements and data sources for each measure. A few commenters stated the separation would allow more time for health organizations and EHR vendors to develop additional technologies necessary for reporting on both measures. In addition, a few commenters supported separating the AUR Surveillance measure because they stated CAHs, smaller hospitals, and hospitals serving socioeconomically vulnerable populations often can report AU data but cannot report AR as discrete data due to the investments required in EHR and laboratory information systems, and the lack of discrete electronic access to required data elements for complete AR reporting. One of these commenters stated this change could allow hospitals, particularly those serving socioeconomically vulnerable populations, to more meaningfully participate in reporting AU and AR data to support national public health goals. A few commenters supported the separation because it aligns with other NHSN reporting requirements or furthers the goals of public health and development of robust interoperability programs. A commenter agreed that this change is clinically appropriate and allows for more comprehensive reporting by eligible hospitals and CAHs. Another commenter supported the change because they stated it could provide the flexibility necessary for continued data exchange. Another commenter agreed with separating the measure as the additional reporting burden associated with the proposed change is less than a minute per year for each eligible hospital and CAH. A commenter supported separating the measure as they stated it supports the Medicare Promoting Interoperability Program's administrative requirements by providing a clear, achievable path for eligible hospitals and CAHs to earn credit for their reporting efforts, even if they can only report one type of data. A commenter stated the change will support the ability to separately assess compliance and rates of exclusions for each component separately.
Response: We thank the commenters for their support of the proposal to split the AUR Surveillance measure into two measures. We agree that this approach allows eligible hospitals and CAHs the opportunity to report on data that is available to them and offers additional time without penalty to address technological updates required for reporting data that are not currently available.
Comment: A commenter thanked CMS for recognizing the challenges associated with the consolidated AUR Surveillance measure and expressed interest in learning how an exclusion for one component of the consolidated measure (that is, either AU or AR) currently affords exclusion to both AU and AR reporting. ( print page 69602)
Response: We thank the commenter for their feedback. In order to report AU data, an eligible hospital or CAH must have an eMAR or a BCMA, and an electronic admission discharge transfer (ADT). To report AR data, an eligible hospital or CAH must have an LIS and an ADT. When we finalized the AUR Surveillance measure, we established an exclusion for eligible hospitals and CAHs that lack an eMAR, BCMA, or ADT. We also established an exclusion for eligible hospitals and CAHs that lack an LIS or ADT. As a result, and for example, even if an eligible hospital or CAH had an LIS and ADT and could report AR data, it could receive an exclusion from the entire AUR Surveillance measure if it did not have an eMAR or BCMA.
Comment: Several commenters who supported the proposed change to the AUR Surveillance measure provided recommendations for the Medicare Promoting Interoperability Program. A few commenters suggested CMS consider making the AU and AR Surveillance measures bonus measures to reward early adopters and to allow more time for the remaining eligible hospitals and CAHs to report. A commenter recommended evaluating the need for partial credit if one of the two new measures is disproportionately reported. A commenter suggested adding complementary measures such as sepsis-associated antibiotic use and nephrotoxic acute kidney injury, to better understand antibiotic prescribing patterns in healthcare.
Response: We thank the commenters for their support and feedback and may consider some of these recommendations in the future. We disagree, however, with the recommendation to establish a bonus for the AU Surveillance and AR Surveillance measures because the AUR Surveillance measure is currently required for reporting for the EHR reporting period in CY 2024. Because the separated measures would be treated as new measures with respect to level of active engagement, eligible hospitals and CAHs would have an additional year of Pre-production and Validation (Option 1) before progressing to Validated Data Production (Option 2). We believe this offers eligible hospitals and CAHs the additional time requested to gain more familiarity with the new measures. We continue to work closely with the CDC regarding how best to support eligible hospitals and CAHs in AU Surveillance and AR Surveillance reporting. For implementation questions regarding reporting issues, we recommend contacting CDC's NHSN ( [email protected] ), or contacting CMS through the “Help” page at the CMS QualityNet website at https://cmsqualitysupport.servicenowservices.com/qnet_qa .
Comment: A few commenters recommended adopting the measure change beginning with CY 2024 instead of CY 2025 as they stated adopting the change in CY 2024 would benefit more eligible hospitals and CAHs.
Response: We thank commenters for their support and feedback; however, we believe that by adopting this change in CY 2025, eligible hospitals and CAHs will have had an additional year of experience in the Pre-production and Validation stage (Option 1). In the Medicare and Medicaid Programs; Electronic Health Record Incentive Program-Stage 3 and Modifications to Meaningful Use in 2015 Through 2017 final rule ( 80 FR 62862 through 62864 ), beginning with the EHR reporting period in CY 2016, we defined active engagement under the Public Health and Clinical Data Registry Reporting objective as when an eligible hospital or CAH is in the process of moving towards sending “production data” to a public health agency or clinical data registry, or is sending production data to a public health agency or clinical data registry. In the FY 2023 IPPS/LTCH PPS final rule, we required that eligible hospitals and CAHs report their level of active engagement as either Option 1: Pre-production and Validation, or Option 2: Validated Data Production for each required or optional Public Health and Clinical Data Exchange objective measure they report, beginning with the EHR reporting period in CY 2023 ( 87 FR 49338 through 49340 ). We also adopted the requirement that eligible hospitals and CAHs may spend only one EHR reporting period at the Option 1: Pre-production and Validation level of active engagement per measure, and that they must progress to the Option 2: Validated Data Production level for the next EHR reporting period for which they report a particular measure, beginning with the EHR reporting period in CY 2024 ( 87 FR 49342 ).
Our proposal to treat the AU Surveillance measure and AR Surveillance measure as new measures, as finalized in section IX.F.2.(c) of this final rule, will provide eligible hospitals and CAHs an additional year in Pre-production and Validation (Option 1) before progressing to Validated Data Production (Option 2). This means that eligible hospitals and CAHs could spend two years in Option 1 before moving to Validated Data Production (Option 2), while reporting on the same data.
For example, an eligible hospital or CAH submitting data on the AUR Surveillance measure in CY 2024 could be in Option 1. In CY 2025, that eligible hospital or CAH could remain in Option 1 when reporting the separated AU Surveillance and AR Surveillance measures, and in CY 2026, the eligible hospital or CAH would be required to be in Option 2 for the AU Surveillance and AR Surveillance measures.
Comment: A few commenters recommended that CMS continue to work with rural and small hospitals to ensure they have the resources and technical assistance to fulfill the intent of the measure. A commenter requested that CMS provide transparency regarding data submission requirements and implementation guidance in the final rule like what CMS and CDC provided regarding CY 2024 data submission. A commenter described a significant burden in reporting AU and AR data to NHSN. The commenter stated their belief that the measure does not meet the intent of “interoperable” because of the resources involved to extract reports and file sets to upload to NHSN, difficulties with NHSN reporting software, and the cost to automate the process.
Response: We will continue to work with small and rural hospitals to provide technical assistance and other resources to successfully meet Medicare Promoting Interoperability Program requirements, as commenters recommended. In collaboration with the CDC, we will strive to provide open, transparent communication about how eligible hospitals and CAHs can fulfill the AU Surveillance and AR Surveillance measures. We disagree that a measure to promote the standards-based transmission of AU and AR data does not meet the intent of interoperability. Standards-based interoperability does require investment and configuration of health IT modules and the work of staff skilled in that domain to execute it. Eligible hospitals and CAHs are important contributors to public health efforts to address antibiotic use and resistance. Therefore, we believe that the public health value of antibiotic use and resistance reporting outweighs the burden incurred by reporting eligible hospitals and CAHs. Nevertheless, we agree that the best model of interoperable public health data exchange is one that delivers necessary data in the least burdensome fashion. We will continue to work with CDC and ONC to identify opportunities to reduce the reporting burden for eligible hospitals and CAHs.
After consideration of the public comments we received, we are ( print page 69603) finalizing our proposal to separate the AUR Surveillance measure into two measures, AU Surveillance and AR Surveillance, beginning with the EHR reporting period in CY 2025.
We previously finalized in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49337 ) the availability of three exclusions for an eligible hospital or CAH reporting on the AUR Surveillance measure that: (1) Does not have any patients in any patient care location for which data are collected by NHSN during the EHR reporting period; (2) Does not have an eMAR/BCMA records or an ADT system during the EHR reporting period; or (3) Does not have an electronic LIS or electronic ADT system during the EHR reporting period.
We received feedback from eligible hospitals and CAHs requesting clarity on whether an AUR Surveillance exclusion applies when they possess all necessary health IT systems but lack discrete electronic access to data elements necessary for NHSN AUR reporting. For example, an eligible hospital or CAH may possess an LIS, but it may refer AR testing to an outside reference laboratory that does not provide data elements necessary for NHSN AUR reporting results to the referring laboratory. As the eligible hospital or CAH has an LIS system and therefore could not claim the third exclusion, assuming it could not claim another exclusion, the eligible hospital or CAH would be required to manually extract the data elements to successfully report the AUR Surveillance measure.
This policy inadvertently caused difficulties for eligible hospitals and CAHs, such as the one in the example, because manual reporting of NHSN AUR data is both infeasible and against NHSN AUR recommendations. [ 824 ] In addition, we require that eligible hospitals and CAHs must use technology certified to the criterion at 45 CFR 170.315(f)(6) , “Transmission to public health agencies—antimicrobial use and resistance reporting” for data submission ( 87 FR 49337 ). We believe an exclusion that applies to eligible hospitals and CAHs that lack discrete electronic access to required data elements, including interface or configuration issues beyond their control, will address the difficulties for eligible hospitals and CAHs engaging in manual data collection to conduct AU or AR reporting. Therefore, in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36353 through 36354 ), we proposed to add a new exclusion to account for scenarios where eligible hospitals or CAHs lack a data source containing discrete electronic data elements that are required for reporting the AU Surveillance or AR Surveillance measures, meaning an eligible hospital or CAH cannot query, extract, or download the data elements in a discrete, structured manner from the systems to which it has access. Specifically, under this new exclusion, an eligible hospital or CAH will be excluded from reporting the AU Surveillance measure when it does not have a data source containing the minimal discrete data elements that are required for reporting the AU Surveillance measure. Similarly, an eligible hospital or CAH will be excluded from reporting the AR Surveillance measure when it does not have a data source containing the minimal discrete data elements that are required for reporting the AR Surveillance measure.
Specifically, we proposed to modify the existing exclusions under the AUR Surveillance measure, to maintain applicability to the AU Surveillance and AR Surveillance measures ( 89 FR 36353 through 36354 ). For example, we would assign exclusion 2 to the AU Surveillance measure because it relies on eMAR or BCMA data, and exclusion 3 to the AR Surveillance measure because it relies on LIS data. For the AU Surveillance measure, we proposed to adopt three eligible exclusions, as follows: Any eligible hospital or CAH may be excluded from the AU Surveillance measure if the eligible hospital or CAH: (1) Does not have any patients in any patient care location for which data are collected by NHSN during the EHR reporting period; (2) Does not have an eMAR/BCMA electronic records or an electronic ADT system during the EHR reporting period; or (3) Does not have a data source containing the minimal discrete data elements that are required for reporting. For the AR Surveillance measure, we proposed to adopt three eligible exclusions, as follows: Any eligible hospital or CAH may be excluded from the AR Surveillance measure if the eligible hospital or CAH: (1) Does not have any patients in any patient care location for which data are collected by NHSN during the EHR reporting period; (2) Does not have an electronic LIS or electronic ADT system during the EHR reporting period; or (3) Does not have a data source containing the minimal discrete data elements that are required for reporting.
We invited public comment on our proposals to adopt three applicable exclusions for the AU Surveillance measure and for the AR Surveillance measure, of which the third exclusion for each measure is a new exclusion for eligible hospitals and CAHs that lack discrete electronic access to data elements that are required for reporting.
Comment: Many commenters supported adopting the exclusions for the AU Surveillance and AR Surveillance measures. A few stated the proposal to separate the AUR Surveillance measure into two measures provided clarification for the associated exclusions. A few commenters stated the exclusion criteria would ensure smaller acute care hospitals that lack the infrastructure to report the level of data are not unduly penalized. A few commenters supported the change because the AU Surveillance and AR Surveillance measures rely on different data sources, and there are certain data fields that require a discrete, structured format. A few commenters stated including measure-specific exclusions could provide the flexibility necessary for continued participation and success. A commenter appreciated that eligible hospitals or CAHs could qualify for an exclusion for one or both measures, without penalty. Another commenter stated that the new exclusion for scenarios where eligible hospitals or CAHs lack a data source containing discrete electronic data elements that are required for reporting would reduce the administrative burden by removing the need for eligible hospitals or CAHs to manually extract the data elements needed to successfully report on the measures.
Response: We thank the commenters for their support and feedback. We agree that the new exclusions allow eligible hospitals and CAHs to avoid penalties in situations where reporting on AU Surveillance measure data, AR Surveillance measure data, or both, is infeasible. In proposing to separate the AUR Surveillance exclusions, we tailored the exclusions to the specific measure.
After consideration of the public comments we received, we are finalizing our proposal to adopt three exclusions each, for the AU Surveillance and AR Surveillance measures as follows. For the AU Surveillance measure, we are finalizing our proposal to adopt three exclusions, as follows, beginning with the EHR reporting period in CY 2025: Any eligible hospital or CAH may be excluded from the AU Surveillance ( print page 69604) measure if the eligible hospital or CAH: (1) Does not have any patients in any patient care location for which data are collected by NHSN during the EHR reporting period; (2) Does not have an eMAR/BCMA electronic records or an electronic ADT system during the EHR reporting period; or (3) Does not have a data source containing the minimal discrete data elements that are required for reporting. For the AR Surveillance measure, we are finalizing our proposal to adopt three exclusions, as follows, beginning with the EHR Reporting Period in CY 2025: Any eligible hospital or CAH may be excluded from the AR Surveillance measure if the eligible hospital or CAH: (1) Does not have any patients in any patient care location for which data are collected by NHSN during the EHR reporting period; (2) Does not have an electronic LIS or electronic ADT system during the EHR reporting period; or (3) Does not have a data source containing the minimal discrete data elements that are required for reporting.
In the FY 2023 IPPS/LTCH PPS final rule, we finalized a policy to limit the amount of time an eligible hospital or CAH may spend in the Option 1: Pre-production and Validation level of active engagement to one EHR reporting period ( 87 FR 49340 through 49342 ). As finalized, this limitation applies beginning with the EHR reporting period in CY 2024. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36354 ), we proposed to consider the AU Surveillance and AR Surveillance measures as new measures with respect to level of active engagement beginning with the EHR reporting period in CY 2025, independent of the eligible hospital's or CAH's prior level of active engagement for the AUR Surveillance measure in the EHR reporting period in CY 2024. We proposed that, should we finalize the AU Surveillance and AR Surveillance measures, for each measure, eligible hospitals and CAHs may spend only one EHR reporting period at the Option 1: Pre-production and Validation level of active engagement before they must progress to the Option 2: Validated Data Production level for the next EHR reporting period for which they report the measure. We discussed in our proposal that this will offer eligible hospitals and CAHs an additional year to gain familiarity with reporting to the NHSN before they are required to move to Option 2: Validated Data Production.
We invited public comment on our proposal to consider the AU Surveillance and AR Surveillance measures as new measures with respect to level of active engagement beginning with the EHR reporting period in CY 2025, independent of the eligible hospital's or CAH's prior level of active engagement for the AUR Surveillance measure.
Comment: Many commenters supported our proposal to treat the AU Surveillance and AR Surveillance measures as new measures with respect to level of active engagement. Several commenters agreed the proposal would allow eligible hospitals and CAHs additional time to gain familiarity with reporting to the NHSN. A commenter stated this proposal would smooth the transition and allow eligible hospitals and CAHs additional time for testing and validation prior to submitting production data for the two new measures. Another commenter stated that treating the AU Surveillance and AR Surveillance measures as new measures with respect to level of active engagement is necessary given the difficulties hospitals have experienced with AR data reporting and the current inability to claim an exclusion specific to AR data.
Response: We thank commenters for their support. We agree that treating the AU Surveillance and AR Surveillance measures as new measures with respect to level of active engagement will be helpful for eligible hospitals and CAHs and will allow them additional time to gain familiarity with reporting to the NHSN.
Comment: A commenter supported the proposal to allow eligible hospitals and CAHs to spend only one EHR reporting period at the “Pre-production and Validation” level of active engagement but recommended that the new measures and their proposed requirements begin in CY 2026 rather than CY 2025 because of expected workflow changes.
Response: We thank the commenter for this feedback. We expect that internal workflow considerations regarding the new AU Surveillance and AR Surveillance measures compared to the prior AUR Surveillance measure will be relatively small because the content of the measures is unchanged. We believe a delay in the separation of the AUR Surveillance measure by an additional year is unnecessary because eligible hospitals and CAHs already have experience reporting the AUR Surveillance measure beginning with the EHR reporting period in CY 2024. .
After consideration of the public comments we received, we are finalizing our proposal to treat the AU Surveillance and AR Surveillance measures as new measures with respect to level of active engagement, beginning with the EHR reporting period in CY 2025 and subsequent years.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36354 through 36355 ), we stated that we do not believe separating the AUR Surveillance measure into two measures, AU Surveillance and AR Surveillance, should affect scoring or the exclusion redistributions for the Public Health and Clinical Data Exchange objective previously adopted in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59266 ). We noted that the separation of the AUR Surveillance measure does not expand on the previously finalized requirements of the measure. In other words, eligible hospitals and CAHs are required to report AU and AR data, whether combined under the AUR Surveillance measure, or separated into AU Surveillance and AR Surveillance measures.
Therefore, in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36354 through 36355 ), we proposed maintaining a scoring value of 25 points for reporting on all required measures in the Public Health and Clinical Data Exchange objective, which would increase from five measures to six measures, including the four previously finalized measures and the two proposed required measures (AU Surveillance and AR Surveillance). We also proposed to maintain the exclusion redistribution policy we adopted in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59267 ) but modify it to indicate there are six measures as opposed to five measures. If an eligible hospital or CAH claims an exclusion for each of the six required measures, the 25 points of the Public Health and Clinical Data Exchange objective would continue to be redistributed to the Provide Patients Electronic Access to their Health Information measure.
We invited public comment on our proposal to maintain the approach to scoring and point redistribution for the Public Health and Clinical Data Exchange objective.
Comment: A few commenters supported our proposal to maintain the scoring approach for the Public Health and Clinical Data Exchange objective with the new AU Surveillance and AR Surveillance measures. One of the ( print page 69605) commenters expressed concern that the scoring approach does not account for challenges in reporting among resource-constrained hospitals. The commenter recommended a weighted scoring system considering each measure's complexity.
Response: We thank the commenters for their feedback and may consider a weighted scoring approach that takes each measure's complexity into account in the future.
After consideration of the public comments we received, we are finalizing our proposal to maintain the approach to scoring for the Public Health and Clinical Data Exchange objective. We are also finalizing our proposal to maintain the existing exclusion redistribution policy for the Public Health and Clinical Data Exchange objective but modify it to indicate there are six measures rather than five measures.
For ease of reference, Table IX.F.-01 lists the objectives and measures for the Medicare Promoting Interoperability Program for the EHR reporting period in CY 2025, as revised, to reflect the previously finalized and newly finalized measures and objectives in this final rule.

In the CY 2024 Medicare Physician Fee Schedule (PFS) final rule ( 88 FR 79307 through 79312 ), we finalized revisions to the definition of CEHRT for the Medicare Promoting Interoperability Program at 42 CFR 495.4 . Specifically, we finalized the addition of a reference to the revised name of “Base EHR definition,” proposed in the Health Data, Technology, and Interoperability: Certification Program Updates, Algorithm Transparency, and Information Sharing (HTI-1) proposed rule ( 88 FR 23759 , 23905 ), to ensure, if the HTI-1 proposals were finalized, the revised name of “Base EHR definition” will be applicable for the CEHRT definitions going forward ( 88 FR 79309 through 79312 ). We also finalized the replacement of our references to the “2015 Edition health IT certification criteria” with “ONC health IT certification criteria,” and the addition of the regulatory citation for ONC health IT certification criteria in 45 CFR 170.315 . We finalized the proposal to specify that technology meeting the CEHRT definition must meet ONC's health IT certification criteria “as adopted and updated in 45 CFR 170.315 ” ( 88 FR 79553 ). This approach is consistent with the definitions and approach subsequently finalized in ONC's HTI-1 final rule, which appeared in the Federal Register on January 9, 2024 ( 89 FR 1205 through 1210 ). For additional background and information on this update, we refer readers to the discussion in the CY 2024 PFS final rule on this topic ( 88 FR 79307 through 79312 ).
In consideration of the updates finalized in the CY 2024 PFS final rule and the HTI-1 final rule, we refer to “ONC health IT certification criteria” throughout this final rule where we previously would have referred to “2015 Edition health IT certification criteria.” We believe that these revisions to the definition of CEHRT in 42 CFR 495.4 will ensure that updates to the definition of Base EHR in 45 CFR 170.102 , and updates to applicable ONC health IT certification criteria in 45 CFR 170.315 , will be incorporated into the CEHRT definition without additional regulatory action by CMS. We also believe these updates align with the transition from designating health IT certification criteria as part of year themed “editions,” to the “edition-less” approach finalized in the ONC HTI-1 final rule. For ease of reference, Table IX.F.-02. lists the ONC health IT certification criteria required to meet the Medicare Promoting Interoperability Program objectives and measures.
We also wish to highlight certain updates to ONC health IT certification criteria finalized in the ONC HTI-1 final rule that impact certification criteria referenced under the CEHRT definition. ONC adopted the certification criterion, “decision support interventions (DSI)” in 45 CFR 170.315(b)(11) to replace the “clinical decision support (CDS)” certification criterion in 170.315(a)(9) included in the Base EHR definition ( 89 FR 1231 ). The finalized DSI criterion ensures that Health IT Modules certified to 45 CFR 170.315(b)(11) must, among other functions, enable a limited set of identified users to select (activate) ( print page 69614) evidence-based and Predictive DSIs (as defined in 45 CFR 170.102 ) and support “source attributes”—categories of technical performance and quality information—for both evidence-based and Predictive DSIs. ONC further finalized that a Health IT Module may meet the Base EHR definition by either being certified to the existing CDS version of the certification criterion in 45 CFR 170.315(a)(9) or being certified to the revised DSI criterion in 45 CFR 170.315(b)(11) , for the period up to, and including, December 31, 2024. On and after January 1, 2025, ONC finalized that only the DSI criterion in 45 CFR 170.315(b)(11) will be included in the Base EHR definition, and the adoption of the criterion in 45 CFR 170.315(a)(9) will expire on January 1, 2025 ( 89 FR 1281 ).
In addition to the DSI criterion, which is required to meet the Base EHR definition after January 1, 2025, in the ONC HTI-1 final rule, ONC finalized other updates related to health IT certification criteria referenced in the CEHRT definition. For these updates, health IT developers must update and provide certified Health IT Modules to their customers by January 1, 2026, including updates resulting from the following finalized policies:
- ONC updated the “Transmission to public health agencies—electronic case reporting” criterion in 45 CFR 170.315(f)(5) specifying consensus-based, industry-developed electronic standards and implementation guides (IGs) to replace functional, descriptive requirements in the existing criterion ( 89 FR 1226 ).
- ONC adopted the United States Core Data for Interoperability (USCDI) version 3 in 45 CFR 170.213(b) and finalized that USCDI version 1 in 45 CFR 170.213(a) will expire on January 1, 2026. This change impacts ONC health IT certification criteria that reference the USCDI, including the “transitions of care” certification criteria in 45 CFR 170.315(b)(1)(iii)(A) ( 1 )-( 2 ), “Clinical information reconciliation and incorporation—Reconciliation” ( 45 CFR 170.315(b)(2)(iii)(D) ( 1 ) through ( 3 )); and “View, download, and transmit to 3rd party” ( 45 CFR 170.315(e)(1)(i)(A) ( 1 )) ( 89 FR 1210 ).
- ONC updated the “demographics” certification criterion ( 45 CFR 170.315(a)(5) ), including renaming the criterion to “patient demographics and observations” ( 89 FR 1295 ).
- ONC updated the “standardized API for patient and population services” certification criterion in 45 CFR 170.315(g)(10) to include newer versions of certain standards and updated functionality to support the criterion ( 89 FR 1283 ).
For complete information about the updates to ONC health IT certification criteria finalized in the HTI-1 final rule, we refer readers to the text of the final rule ( 89 FR 1192 ) as well as resources available on ONC's website. [ 825 ]
We did not propose and are not finalizing any changes to these policies.

In the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41636 through 41645 ), we adopted a performance-based scoring methodology for eligible hospitals and CAHs reporting under the Medicare Promoting Interoperability Program beginning with the EHR reporting period in CY 2019, which included a minimum scoring threshold of a total score of 50 points or more, that eligible hospitals and CAHs must meet to satisfy the requirement to report on the objectives and measures of meaningful use under 42 CFR 495.24 . In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45491 through 45492 ), we increased the minimum scoring threshold from 50 points to 60 points beginning with the EHR reporting period in CY 2022 and adopted corresponding changes to the regulatory text at 42 CFR 495.24(e)(1)(i)(C) for the EHR reporting period in CY 2022. In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49410 through 49411 ), we extended the 60-point threshold for the EHR reporting period in CY 2023 and subsequent years in the regulatory text at 42 CFR 495.24(f)(1)(i)(B) .
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36369 through 36371 ), we proposed to increase the minimum scoring threshold from 60 points to 80 points and proposed corresponding changes to the regulation text at 42 CFR 495.24(f)(1)(i) for the EHR reporting period in CY 2025 and subsequent years. Our review of the CY 2022 Medicare Promoting Interoperability Program's performance results found 98.5 percent of eligible hospitals and CAHs (that is 97 percent of CAHs and 99 percent of eligible hospitals) that reported to the Medicare Promoting Interoperability Program successfully met the minimum scoring threshold of 60 points, and 81.5 percent of eligible hospitals and CAHs (that is 78 percent of CAHs and 83 percent of eligible hospitals) that reported to the Medicare Promoting Interoperability Program exceeded the score of 80 points. Given the widespread success of eligible hospitals and CAHs participating in the Medicare Promoting Interoperability Program in CY 2022, we stated that adopting a higher scoring threshold would incentivize more eligible hospitals and CAHs to align their health information systems with evolving industry standards and will encourage increased data exchange. We noted that eligible hospitals and CAHs will have gained 3 years of experience in the Medicare Promoting Interoperability Program (CYs 2022, 2023, and 2024) at the 60-point minimum score threshold to improve performance. We stated that an increase from 60 points to 80 points would encourage higher levels of performance through the advanced use of CEHRT to further incentivize eligible hospitals and CAHs to improve interoperability and health information exchange. We also proposed to make corresponding changes to the regulatory text at 42 CFR 495.24(f)(1)(i) to reflect the scoring threshold change. Specifically, in the proposed rule, we proposed to adopt new regulatory text at 42 CFR 495.24(f)(1)(i)(C) , to state “In 2025 and subsequent years, earn a total score of at least 80 points.” We proposed that this change would take effect for the EHR reporting period in CY 2025 and subsequent years.
We invited public comment on our proposals to increase the minimum scoring threshold from 60 points to 80 points for the EHR reporting period in CY 2025 and subsequent years, and to make corresponding changes to the regulatory text at 42 CFR 495.24(f)(1)(i) .
Comment: A few commenters expressed support for the proposal to increase the minimum scoring threshold ( print page 69617) from 60 points to 80 points. These commenters agreed that a higher scoring threshold will incentivize more eligible hospitals and CAHs to align their health information systems with evolving industry standards, including advanced use of CEHRT, improved interoperability and health information exchange, and increased data exchange.
Comment: Several commenters supported the proposal to increase the scoring threshold from 60 points to 80 points and offered additional recommendations for CMS's consideration. A commenter suggested that CMS conduct further research and release de-identified data on which categories of eligible hospitals and CAHs are performing well to better understand the success rates of participation in the Medicare Promoting Interoperability Program. Another commenter recommended CMS ensure the increased scoring threshold is scaled appropriately, considering the evolving nature of health IT and varying capabilities of organizations. A commenter supported the proposal to raise the scoring threshold to 80 points but recommended raising it to 100 points because they stated that would have the most effect on quality and safety. A few commenters supported the increase in the minimum scoring threshold but noted that beginning with the EHR reporting period in CY 2025 was too soon. A few commenters recommended an incremental increase over two years, from 60 points to 70 points in CY 2025 and from 70 points to 80 points in CY 2026. A commenter recommended CMS consider a three-year phased approach, remaining at 60 points in year one, increasing to 70 points in year two, and finally increasing to 80 points in year three. A few commenters supported raising the scoring threshold but recommended increasing it to 75 points rather than 80 points because of the difficulty smaller hospitals may have with performing on the HIE objective.
Response: We thank the commenters for their support and recommendations. We continue to believe that adopting a higher scoring threshold will incentivize more eligible hospitals and CAHs to align their health information systems with evolving industry standards and will encourage increased data exchange. With regard to the comment requesting release of de-identified data, we remind readers of our previously adopted policy to publicly report total scores for each eligible hospital and CAH, beginning with data from the EHR reporting period in CY 2023 ( 87 FR 49347 ). When these data become publicly available, which we anticipate will be in January 2025, researchers, consumers, and other interested parties will have access to hospitals' scoring information. As we discuss hereafter, we agree with commenters who recommended an incremental increase to the minimum scoring threshold over two years, from 60 points to 70 points for the EHR reporting period in CY 2025 and from 70 points to 80 points for the EHR reporting period in CY 2026. In response to the commenter recommending that we consider increasing the minimum scoring threshold to 100 points, we may consider this for future rulemaking.
Comment: A few commenters did not support the proposal to increase the scoring threshold from 60 points to 80 points. A commenter stated that according to the CY 2022 Medicare Promoting Interoperability Program's performance results CMS cited, over 1000 hospitals would not meet the new scoring threshold and would be adversely impacted by this change. Another commenter stated the additional reporting burden for CY 2025 from past finalized rules, proposed rule changes, as well as changes to requirements of CEHRT increase the program requirements and negate the need to increase the performance threshold. Another commenter stated they did not believe changing the scoring threshold would produce a more comprehensive score of reliable data as EHR developers and vendors are responsible for providing certified functionality to obtain such a score.
Many commenters did not support the proposal to increase the scoring threshold from 60 points to 80 points and offered alternative recommendations for CMS' consideration. Several commenters recommended that CMS consider a delayed implementation of the change in scoring over several years. A few commenters were concerned that hospitals and EHR developers needed more time to adjust to the reporting requirements. A few commenters stated CMS should give hospitals time to independently analyze the Medicare Promoting Interoperability Program performance data CMS referenced. A commenter noted that in past years, CMS has provided fair warning and time for adjustment, and therefore this proposal should be delayed avoiding increased failure rates and decreased compliance. A commenter stated the most likely path to increased points would be HIE or TEFCA participation, which would require more time and money. Several commenters opposed raising the minimum scoring threshold to 80 points at this time, recommending a smaller increase. A few commenters urged CMS to maintain the 60-point threshold because they stated the proposed increase is too drastic.
Response: We thank the commenters for their feedback and concerns. We disagree with the commenter who stated that over 1,000 hospitals would not meet an 80-point scoring threshold. According to the CY 2022 Medicare Promoting Interoperability Program's performance results we cited in the FY 2025 LTCH/IPPS proposed rule ( 89 FR 36369 through 36371 ), 18.5 percent of hospitals did not meet a scoring threshold of at least 80 points. That is 739 hospitals, or 502 eligible hospitals and 236 CAHs. We reiterate that these statistics refer to the CY 2022 performance period and eligible hospitals and CAHs will have gained 3 additional years (CYs 2022, 2023, and 2024) of experience in the Medicare Promoting Interoperability Program with the threshold of 60 points.
We also disagree with the commenter who stated the additional reporting burden for CY 2025 from past finalized rules, proposed rule changes, as well as changes to requirements of CEHRT increase the program requirements and negate the need to increase the minimum performance threshold. We believe that there has been sufficient time since CY 2022 for programmatic stability in the Medicare Promoting Interoperability Program's available objectives and measures to warrant an increase to the minimum scoring threshold. According to the Medicare Promoting Interoperability Program's performance results, the average scores for eligible hospitals and CAHs have steadily increased since 2020; the average final score was 72.5 in 2020 (72.4 for eligible hospitals and 73.8 for CAHs), 74.9 in 2021 (74.5 for eligible hospitals and 76.6 for CAHs), and 94.6 in 2022 (95.5 for eligible hospitals and 91.7 for CAHs). An increase to the minimum scoring threshold represents our goals of encouraging higher levels of program performance and further advancement toward interoperability, promoting greater health information exchange, and raising overall patient care quality.
While participation in TEFCA or HIE bidirectional exchange are highly scored, we disagree that choosing one of these options under the HIE objective is the most likely path to increasing overall points. We note that there have been several finalized changes in the Medicare Promoting Interoperability Program that allow increased scoring on ( print page 69618) new measures (for example, the HIE Bi-Directional Exchange, Enabling Exchange under TEFCA, AU Surveillance and AR Surveillance measures) as well as opportunities to earn bonus points that could allow eligible hospitals and CAHs to achieve the 70-point scoring threshold for CY 2025 and 80-point scoring threshold for CY 2026 that we are finalizing as a modification of our proposal. We have balanced this scoring threshold increase against the full scope of the Medicare Promoting Interoperability Program's changes, which consider the role of bonus points in meeting or surpassing the minimum threshold. We believe that these efforts offer more than sufficient opportunity for eligible hospitals and CAHs to earn more points with an increase to the Medicare Promoting Interoperability Program's minimum scoring threshold.
We also disagree with commenters who stated that an increase to the minimum scoring threshold beginning with the EHR reporting period in CY 2025 would be too drastic. As the 60-point threshold has been in place since CY 2022, we maintain that the Program is prepared to adapt and evolve toward an increased standard of participation for eligible hospitals and CAHs to be considered meaningful EHR users. We remind readers that according to CY 2022 Medicare Promoting Interoperability Program performance results, 98.5 percent of eligible hospitals and CAHs (that is 97 percent of CAHs and 99 percent of eligible hospitals) that reported to the Medicare Promoting Interoperability Program successfully met the minimum threshold score of 60 points, and 81.5 percent of eligible hospitals and CAHs (that is 78 percent of CAHs and 83 percent of eligible hospitals) that reported to the Medicare Promoting Interoperability Program exceeded the score of 80 points. According to the same data, 92.8 percent of eligible hospitals and CAHs (that is 93.8 percent of eligible hospitals and 90.2 percent of CAHs) achieved a scoring threshold of 70 points in CY 2022. We reiterate that the average scores for eligible hospitals and CAHs have remained above 70 since 2020 and have shown upward trends year after year. Such successful Program results signify the need for raising the minimum score. Given the widespread success of eligible hospitals and CAHs participating in the Medicare Promoting Interoperability Program in CY 2022, the expanded opportunities to earn points on new measures as well as additional bonus points that have become available since CY 2022, as well as the fact that eligible hospitals and CAHs have gained three years of experience in the Medicare Promoting Interoperability Program at the 60-point minimum score threshold to improve performance (CYs 2022, 2023, and 2024), we believe that an increase to the minimum scoring threshold is more than feasible. Increasing the minimum scoring threshold will encourage higher levels of performance through the advanced use of CEHRT to further incentivize eligible hospitals and CAHs to improve interoperability and health information exchange.
While increasing the minimum scoring threshold is important for incentivizing eligible hospitals and CAHs to improve interoperability and health information exchange, after considering public comments, we concluded that an incremental approach would provide additional time for eligible hospitals, CAHs, and EHR developers to meet updated reporting requirements. Specifically, we agree with commenters who recommended an incremental increase to the minimum scoring threshold over two years, from 60 points to 70 points for the EHR reporting period in CY 2025 and from 70 points to 80 points for the EHR reporting period beginning in CY 2026. We believe this will give eligible hospitals, CAHs, and EHR developers additional time to adjust to an eventual 80-point minimum scoring threshold, while continuing to incentivize more eligible hospitals and CAHs to align their health information systems with evolving industry standards. Increasing the minimum scoring threshold to 70 points for the EHR reporting period in CY 2025 will also be less likely to disproportionately impact eligible hospitals and CAHs that have struggled to achieve a score of 80 points, especially smaller and under resourced hospitals. This incremental increase will reduce the burden of increased reporting requirements by providing a phased approach. Finally, a 10-point increase for the EHR reporting period in CY 2025, and a subsequent 10-point increase for the EHR reporting period beginning in CY 2026, would align with previous gradual increases to the minimum scoring threshold in the Medicare Promoting Interoperability Program, such as the previous increase from 50 points to 60 points in CY 2022 ( 86 FR 45492 ).
After consideration of the public comments we received, we are finalizing, with modification, our proposal to increase the minimum performance-based scoring threshold from 60 points to 80 points, beginning with the EHR reporting period in CY 2025. We are finalizing an increase to the minimum performance-based scoring threshold from 60 points to 70 points for the EHR reporting period in CY 2025, and from 70 points to 80 points beginning with the EHR reporting period in CY 2026 and continuing in subsequent years. We maintain our intent to heighten the required standards for the Medicare Promoting Interoperability Program's performance levels and encourage higher levels of performance through the advanced usage of CEHRT in order to further incentivize eligible hospitals and CAHs to improve interoperability and health information exchange. This gradual increase will be more feasible for eligible hospitals and CAHs, while providing an opportunity to show continued growth in the Program and reflect the success of its participants.
We are therefore also finalizing, with modification, our proposal to adopt regulatory text at 42 CFR 495.24(f)(1)(i)(C) , which stated “In 2025 and subsequent years, earn a total score of at least 80 points,”. Instead, we are modifying the regulatory text to align with the finalized policy to increase the performance-based scoring threshold from 60 points to 70 points for the EHR reporting period in CY 2025, and from 70 points to 80 points beginning with the EHR reporting period in CY 2026 and continuing in subsequent years. We are finalizing changes to the regulatory text at 42 CFR 495.24(f)(1)(i)(B) to state “In 2023 and 2024, earn a total score of at least 60 points”, and modifying our proposal by adding regulatory text at 42 CFR 495.24(f)(1)(i)(C) to state “In 2025, earn a total score of at least 70 points.” and by adding regulatory text at 42 CFR 495.24(f)(1)(i)(D) to state “In 2026 and subsequent years, earn a total score of at least 80 points.”
As shown in Table IX.F.-03., the points associated with the required measures sum to 100 points and reporting one of the optional measures under the Public Health and Clinical Data Exchange objective adds an additional 5 bonus points. The scores for each of the measures are added together to calculate a total score of up to 100 possible points for each eligible hospital or CAH. We refer readers to Table IX.F.-03. in this final rule, which summarizes the objectives, measures, maximum points available, and whether a measure is required or optional for the EHR reporting period in CY 2025 based on our previously adopted policies, and the finalized measure changes included in this final rule.

The maximum points available, by measure, in this final rule, as shown in Table IX.F.-03, do not include the points that will be redistributed in the event an exclusion is claimed for a given measure. We did not propose any changes to our policy for point redistribution in the event an exclusion is claimed. We did propose and have finalized a revision to the redistribution for the Public Health and Clinical Data Exchange objective to reflect the six measures under that objective (rather than five) after the division of the AUR Surveillance measure into AU Surveillance and AR Surveillance measures, as discussed in section IX.F.2.a. We refer readers to Table IX.F.- ( print page 69620) 04 in this final rule, which shows how points will be redistributed among the objectives and measures for the EHR reporting period in CY 2025, in the event an eligible hospital or CAH claims an exclusion.

Under sections 1814(l)(3)(A) and 1886(n)(3)(A) of the Social Security Act, and the definition of “meaningful EHR user” under 42 CFR 495.4 , eligible hospitals and CAHs must report on clinical quality measures selected by CMS using CEHRT (also referred to as eCQMs), as part of being a meaningful EHR user under the Medicare Promoting Interoperability Program.
Tables IX.F.-05. and IX.F.-06 in this final rule summarize the previously finalized eCQMs available for eligible hospitals and CAHs to report under the Medicare Promoting Interoperability Program for the CY 2024 and CY 2025 reporting periods, as finalized in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59280 through 59281 ). To maintain alignment with the Hospital IQR Program (sections IX.C.5.c and IX.C.5.d of the preamble of this final rule), the order of the eCQMs displayed in Tables IX.F.-05 and IX.F.-06 mirrors that of the Hospital IQR program. In addition, the short names, and the consensus-based entity (CBE) numbers of the measures in the tables match the measures on the Electronic Clinical Quality Improvement Resource Center website at: https://ecqi.healthit.gov/ .

As we stated in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38479 ), we intend to continue to align the eCQM reporting requirements and eCQM measure set for the Medicare Promoting Interoperability Program with similar requirements under the Hospital IQR Program, to the extent feasible. Section 1886(n)(3)(B)(i)(I) of the Act sets forth a preference for the selection of eCQMs that are also used in the Hospital IQR Program or endorsed by the CBE.
In the FY 2025 LTCH/IPPS PPS proposed rule ( 89 FR 36373 through 36373 ), we proposed to adopt two new eCQMs for the Medicare Promoting Interoperability Program and to modify one eCQM, beginning with the CY 2026 reporting period, in alignment with the Hospital IQR Program. Specifically, we proposed to add the following two eCQMs to the Medicare Promoting Interoperability Program eCQM measure set from which eligible hospitals and CAHs can self-select to report, beginning with the CY 2026 reporting period: (1) the Hospital Harm—Falls with Injury eCQM (CBE #4120e) and (2) the Hospital Harm—Postoperative Respiratory Failure eCQM (CBE #4130e). We also proposed to modify ( print page 69622) the Global Malnutrition Composite Score eCQM (CBE #3592e) beginning with the CY 2026 reporting period, by adding patients ages 18 to 64 to the current cohort of patients 65 years or older.
We invited public comment on these proposals for the Medicare Promoting Interoperability Program. We also refer readers to sections IX.C.5 and IX.C.7 where we discuss comments received on these eCQM proposals for both the Medicare Promoting Interoperability and Hospital IQR Programs or only the Hospital IQR Program.
Comment: Several commenters supported CMS's proposals to adopt the Hospital Harm—Falls eCQM and the Hospital Harm—Postoperative Respiratory Failure eCQM, as well as to modify the Global Malnutrition Composite Score eCQM. A few commenters commended CMS's continued efforts to align clinical quality measures across its public reporting programs. A commenter appreciated the measured scope changes of CMS's proposals. Another commenter emphasized the need for quality measures to monitor populations with cognitive and age-related conditions.
Response: We thank the commenters for their support of these measures.
Comment: A commenter recommended that CMS not add more than one new eCQM per reporting period, stating it is a burdensome process to build, track, and implement new eCQMs to maintain alignment between the Hospital IQR Program and the Medicare Promoting Interoperability Program.
Response: We acknowledge the commenter's concerns about the costs associated with adding new eCQMs to their hospital's EHR. While the initial implementation cost for an eCQM may be higher, we anticipate maintenance costs to be much less costly than chart-abstracted quality measures. We believe that aligning eCQM reporting requirements between the Medicare Promoting Interoperability Program and the Hospital IQR Program allows for improved coordination, burden reduction, and promotes quality care. Eligible hospitals that participate in both the Medicare Promoting Interoperability Program and the Hospital IQR Program only need to report eCQM data once for credit in both programs. In addition, the Hospital Harm—Falls eCQM, Hospital Harm—Postoperative Respiratory Failure eCQM, and the Global Malnutrition Composite Score eCQM are among the eCQMs in the eCQM measure set for which eligible hospitals and CAHs may self-select and choose whether to report on.
Comment: A few commenters did not support the Medicare Promoting Interoperability Program's proposal to add two new eCQMs to the measure set in alignment with the Hospital IQR Program. A commenter stated that allowing hospitals to perform their own data extracts and submit data directly to CMS, instead of mandating that eligible hospitals and CAHs utilize CEHRT, would provide benefits such as decreased vendor reliance, increase agility to adapt to changes, and consume fewer resources.
Response: We thank the commenters for their feedback. By aligning the Hospital IQR Program and Medicare Promoting Interoperability Program measure sets, eligible hospitals and CAHs must utilize CEHRT for the electronic transmission of eCQM data to fulfill reporting requirements. Furthermore, we believe that aligning eCQM reporting requirements between the Medicare Promoting Interoperability Program and the Hospital IQR Program allows for improved coordination, burden reduction, and promotes quality care. While we recognize the perceived independence, flexibility, and potential cost savings associated with allowing eligible hospitals and CAHs to extract and submit their own data to CMS, requiring the use of CEHRT for the transmission of this data helps to ensure standardization, interoperability, data accuracy, and integrity.
After consideration of the public comments we received, we are finalizing our proposal to adopt into the measure set from which eligible hospitals and CAHs could self-select to report (1) the Hospital Harm—Falls with Injury eCQM (CBE #4120e) eCQM beginning with the CY 2026 reporting period, (2) the Hospital Harm—Postoperative Respiratory Failure eCQM (CBE #4130e) beginning with the CY 2026 reporting period, and (3) to modify the Global Malnutrition Composite Score eCQM (CBE #3592e) beginning with the CY 2026 reporting period. Table IX.F.-07 summarizes the newly adopted and previously adopted eCQMs for the Medicare Promoting Interoperability Program for the CY 206 reporting period and for subsequent years.

Consistent with our goal to align the eCQM reporting periods and criteria in the Medicare Promoting Interoperability Program with the Hospital IQR Program, eligible hospitals and CAHs have been required to report four calendar quarters of data for each required eCQM: (1) the Safe Use of Opioids—Concurrent Prescribing eCQM; (2) the Severe Obstetric Complications eCQM; (3) the Cesarean Birth eCQM; and (4) three self-selected eCQMs, for the CY 2024 reporting period and subsequent years ( 87 FR 49365 through 49367 ).
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36375 through 36376 ), we proposed that eligible hospitals and CAHs under the Medicare Promoting Interoperability Program would be required to report four calendar quarters of data for each of the following: (1) Three self-selected eCQMs; (2) the Safe Use of Opioids—Concurrent Prescribing eCQM; (3) the Severe Obstetric Complications eCQM; (4) the Cesarean Birth eCQM; (5) the Hospital Harm—Severe Hypoglycemia eCQM; (6) the Hospital Harm—Severe Hyperglycemia eCQM; and (7) the Hospital Harm—Opioid-Related Adverse Events eCQM, beginning with the CY 2026 reporting period. This proposal would require eligible hospitals and CAHs to report a total of nine eCQMs for the CY 2026 reporting period.
We also proposed that eligible hospitals and CAHs under the Medicare Promoting Interoperability Program would be required to report four calendar quarters of data for each of the following: (1) Three self-selected eCQMs; (2) the Safe Use of Opioids—Concurrent Prescribing eCQM; (3) the Severe Obstetric Complications eCQM; (4) the Cesarean Birth eCQM; (5) the Hospital Harm—Severe Hypoglycemia eCQM; (6) the Hospital Harm—Severe Hyperglycemia eCQM; (7) the Hospital Harm—Opioid-Related Adverse Events eCQM; (8) the Hospital Harm—Pressure Injury eCQM; and (9) the Hospital Harm—Acute Kidney Injury eCQM, for a total of eleven eCQMs, beginning with the CY 2027 reporting period and subsequent years.
We invited public comment on our proposals to increase the number of mandatory eCQM measures to a total of nine beginning with the CY 2026 reporting period, and to increase the number of mandatory eCQM measures to a total of eleven beginning with the CY 2027 reporting period and subsequent years. We also refer readers ( print page 69624) to sections IX.C.5.c. and IX.C.5.d. where we discuss comments we received on these eCQM reporting and submission proposals for both the Medicare Promoting Interoperability and Hospital IQR Programs or only the Hospital IQR Program.
Comment: A few commenters supported CMS's proposals to revise the eCQM reporting and submission requirements for the CY 2026 reporting period and subsequent years.
Response: We thank the commenters for their support for these proposals.
Comment: Several commenters did not support increasing the number of required eCQMs, believing the increase may create additional burden to implement, monitor and maintain. A few commenters recommended CMS delay increased reporting requirements or take a less aggressive and phased approach.
Response: We acknowledge commenters' concerns regarding the burden to implement, monitor and maintain eCQMs. However, while the initial implementation cost for an eCQM may be higher, we anticipate maintenance costs to be much less and overall less costly than chart-abstracted quality measures. We acknowledge commenters' recommendations to provide more time to comply with an increase in reporting requirements and a delayed or phased approach. We are committed to supporting eligible hospitals and CAHs through the eCQM implementation process by providing sufficient time to update their systems to comply with new reporting requirements, while balancing the need of including important new patient safety metrics. We also note that we are finalizing with modification our proposal to increase the required number of eCQMs from eight to eleven eCQMs over two years and we are instead finalizing an increase in the number of required eCQMs from eight to eleven over three years
Comment: A few commenters supported CMS's efforts to align eCQM reporting requirements for the Hospital IQR Program and the Medicare Promoting Interoperability Program, believing alignment partially mitigates administrative and cost burdens.
Response: We thank the commenters for their comments and support. We remain committed to implementing quality measures to improve healthcare outcomes while reducing the reporting burden by aligning reporting requirements across multiple quality reporting programs.
Comment: A commenter was concerned that eligible hospitals and CAHs are not required to report on all of the eCQMs in the measure set and suggested that CMS consider this in the future.
Response: We thank the commenter for their feedback. CMS is committed to a balanced approach in implementing quality measures, and a considered approach to increasing the number of measures required for reporting over time. Our goal is to ensure that the measures we use are meaningful, actionable, and not overly burdensome for providers. While we understand the desire for comprehensive reporting and faster implementation, we also must consider factors such as the readiness of providers to report new measures, the feasibility of data collection, and the potential impact on patient care. This is particularly important for rural and small hospitals with limited resources, which is why we proposed a stepwise increase in the number of required eCQMs over a two-year period. After considering comments expressing concerns about burden and requests for more lead time to increase the number of required eCQMs, we are modifying and finalizing our proposal by increasing the number of required eCQMs over a three-year period instead of a two-year period as further described below.
Comment: A few commenters suggested that CMS consider implementation timeframes when introducing a new measure, taking into account voluntary versus mandatory reporting, software development requirements, and organizational readiness to operationalize workflows and tracking. These commenters recommended CMS wait at least three years post-introduction of a new measure before requiring it.
Response: We agree on the importance of considering implementation timeframes and providing sufficient time for hospitals to implement new eCQMs into their EHRs, to take into account offering the opportunity for hospitals to self-select measures so they can gain experience with the measures before they become mandatory, and the potential benefits of a phased approach. We disagree with the recommendation to wait at least three years post-introduction of a new measure before requiring it because hospitals can self-select measures to gain experience with the measures before they become mandatory. CMS is committed to implementing measures in a manner that supports quality improvement while minimizing the burden on providers.
After consideration of the public comments we received, we are finalizing, with modification, our proposal to increase eCQM reporting requirements in the Medicare Promoting Interoperability Program for the CY 2026 reporting period. Specifically, for the CY 2026 reporting period, eligible hospitals and CAHs will be required to report a total of eight eCQMs: three self-selected, and the Safe Use of Opioids, Severe Obstetric Complications, Cesarean Birth, Hospital Harm—Severe Hypoglycemia, and Hospital Harm—Severe Hyperglycemia eCQMs.
We are also finalizing, with modification, our proposal to increase eCQM reporting requirements in the Medicare Promoting Interoperability Program for the CY 2027 reporting period. Specifically, for the CY 2027 reporting period, eligible hospitals and CAHs will be required to submit data for the eight eCQMs finalized for the CY 2026 reporting period as well as the Hospital Harm—Opioid-Related Adverse Events eCQM, for a total of nine eCQMs.
Lastly, we are finalizing, with modification, our proposal to increase eCQM reporting requirements in the Medicare Promoting Interoperability Program beginning with the CY 2028 reporting period. Specifically, beginning with the CY 2028 reporting period and for subsequent years, eligible hospitals and CAHs will be required to submit data for the nine eCQMs required for the CY 2027 reporting period as well as the Hospital Harm—Pressure Injury and Hospital Harm—Acute Kidney Injury eCQMs, for a total of eleven eCQMs.
In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45479 through 45481 ), we adopted the SAFER Guides measure under the Protect Patient Health Information objective beginning with the EHR reporting period in CY 2022. Eligible hospitals and CAHs are required to attest to whether they have conducted an annual self-assessment using all nine SAFER Guides, [ 826 ] at any point during the calendar year in which the EHR reporting period occurs, with one “yes/no” attestation statement. Beginning in CY 2022, the reporting of this measure was required, but eligible hospitals and CAHs were not scored, and an attestation of “yes” or “no” were both acceptable answers without penalty. For additional information, please refer to the discussion of the SAFER Guides measure in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45479 through 45481 ). In the FY 2024 IPPS/ ( print page 69625) LTCH PPS final rule, we finalized a proposal to modify our requirement for the SAFER Guides measure beginning with the EHR reporting period in CY 2024 and continuing in subsequent years, to require eligible hospitals and CAHs to attest “yes” to having conducted an annual self-assessment using all nine SAFER Guides, at any point during the calendar year in which the EHR reporting period occurs to be considered a meaningful user ( 88 FR 59262 ).
We received comments on the FY 2024 IPPS/LTCH PPS proposed rule recommending that we work with ONC to update the SAFER Guides, citing that the SAFER Guides were last updated in 2016 ( 88 FR 59264 ). In response to these comments, we noted that, while the current SAFER Guides reflect relevant and valuable guidelines for safe practices with respect to current EHR systems, we would consider exploring updates in collaboration with ONC. We reminded readers to visit the CMS resource library website at https://www.cms.gov/regulations-guidance/promoting-interoperability/resource-library and the ONC website at https://www.healthit.gov/topic/safety/safer-guides for resources on the content and appropriate use of the SAFER Guides ( 88 FR 59262 ). We also noted that future updates to the SAFER Guides would be provided with accompanying educational and promotional materials to notify participants, in collaboration with ONC, when available ( 88 FR 59265 ). In this final rule, we seek to make readers aware that efforts to update the SAFER Guides are currently underway. We anticipate that updated versions of the SAFER Guides may become available as early as CY 2025, and we would consider proposing a change to the SAFER Guides measure for the EHR reporting period beginning in CY 2026 to permit use of an updated version of the SAFER Guides at that time. We encourage eligible hospitals and CAHs to become familiar with the updated versions of the SAFER Guides when they become available and consider them as they implement appropriate EHR safety practices.
The Department of Health and Human Services (HHS) rule, 21st Century Cures Act: Establishment of Disincentives for Health Care Providers That Have Committed Information Blocking final rule (hereafter referred to as the Disincentives final rule) ( 89 FR 54662 ), appeared in the Federal Register on July 1, 2024. The Disincentives final rule implements the provision of the 21st Century Cures Act specifying that a healthcare provider, determined by the HHS Office of Inspector General (OIG) to have committed information blocking, shall be referred to the appropriate agency to be subject to appropriate disincentives set forth through notice and comment rulemaking. Through policies finalized in the Disincentives final rule, an eligible hospital or CAH will not be considered a meaningful EHR user in an EHR reporting period if the OIG refers, during the calendar year of the EHR reporting period, a determination that the eligible hospital or CAH committed information blocking as defined at 45 CFR 171.103 ( 89 FR 54663 ). Accordingly, we revised the definition of “Meaningful EHR User” in 42 CFR 495.4 to state that an eligible hospital or CAH is not a meaningful EHR user in a payment adjustment year if the OIG refers a determination that the eligible hospital or CAH committed information blocking, as defined at 45 CFR 171.103 , during the calendar year of the EHR reporting period ( 89 FR 54687 through 54691 ). The downward payment adjustment will apply 2 years after the year the referral was made by the OIG, and the EHR reporting period in which the eligible hospital was not a meaningful EHR user. For CAHs, the downward payment adjustment will apply to the payment adjustment year in which the OIG referral was made ( 89 FR 54691 ).
An eligible hospital subject to this disincentive will be subject to a three quarters reduction of the annual market basket increase, while a CAH subject to this disincentive will have its payment reduced to 100 percent of reasonable costs, from the 101 percent of reasonable costs it might have otherwise earned, for failing to qualify as a meaningful EHR user in an applicable year. Additional regulatory provisions have been finalized at 45 CFR 171 Subpart J , related to the application of disincentives ( 89 FR 74953 ).
We note the revised definition of Meaningful EHR User in 42 CFR 495.4 became effective on July 31, 2024, when the Disincentives final rule became effective. For additional background and information on this update, we refer readers to the discussion in the Disincentives final rule ( 89 FR 54687 through 54691 ).
We did not propose and are not finalizing any changes to these policies in this final rule.
In partnership with ONC, we envision a future where patients have timely, secure, and easy access to their health information through the health application of their choice. We are working with ONC to enable this type of access to health information by requiring the use of APIs that utilize the Health Level Seven International® (HL7) FHIR. We work with ONC and other federal partners to improve timely and accurate data exchange, partner with industry to enhance digital capabilities, advance adoption of FHIR, support enterprise transformation efforts that increase our technological capabilities, and promote interoperability. In the FY 2021 IPPS/LTCH PPS proposed rule ( 85 FR 32858 ), we described our future vision for the Medicare Promoting Interoperability Program and stated that we will continue to consider changes that support a variety of HHS goals, including supporting alignment with the 21st Century Cures Act, advancing interoperability and the exchange of health information, and promoting innovative uses of health IT. We also solicited public comment on issues relevant to the Medicare Promoting Interoperability Program that related to policies finalized in the 21st Century Cures Act: Interoperability, Information Blocking, and the ONC Health IT Certification Program final rule, including finalization of a new certification criterion for a standards-based API using FHIR, among other health IT topics ( 85 FR 32858 ).
ONC finalized the HTI-1 final rule ( 89 FR 1192 ), effective March 11, 2024, to further implement the 21st Century Cures Act, among other policy goals. ONC finalized revisions to the “standardized API for patient and population services” certification criterion at 45 CFR 170.315(g)(10) . It also adopted the HL7 FHIR US Core Implementation Guide (IG) Standard for Trial Use version 6.1.0 at 45 CFR 170.215(b)(1)(ii) , which provides the latest consensus-based capabilities aligned with the USCDI version 3 [ 827 ] data elements for FHIR APIs. The HTI-1 final rule also created the Insights Condition and Maintenance of Certification requirements (Insights ( print page 69626) Condition) within the ONC Health IT Certification Program to provide transparent reporting on certified health IT ( 89 FR 1199 ). This Insights Condition requires developers of certified health IT subject to the requirements to report on measures that provide information about the use of specific certified health IT functionalities by end users. One such measure calculates the number of unique individuals who access their electronic health information overall and by different methods such as through a standardized API for patient and population services.
By adopting these new and updated standards, implementation specifications, certification criteria, and conditions of certification, provisions in the HTI-1 final rule advance interoperability, improve transparency, and support the access, exchange, and use of electronic health information. CMS aims to further advance the use of FHIR APIs through policies in the Medicare Promoting Interoperability Program to advance interoperability, encourage the exchange of health information, and promote innovative uses of health IT. We also hope to gain insights into the adoption and use of FHIR APIs by eligible hospitals and CAHs due to the ONC Health IT Certification Program Insights Condition. We believe maintaining our focus on promoting interoperability, alignment, and simplification will reduce healthcare provider burden while allowing flexibility to pursue innovative applications that improve care delivery. For additional background and information, we refer readers to the discussion in the ONC HTI-1 final rule on this topic ( 89 FR 1192 ).
The Medicare Promoting Interoperability Program encourages the advancement of patient safety by promoting appropriate cybersecurity practices through the Security Risk Analysis and the SAFER Guides measures. On February 14, 2023, the National Institute of Standards and Technology (NIST) published updated guidance for health care entities implementing requirements of the Health Insurance Portability and Accountability (HIPAA) Security Rule (45 CFR part 160 and subparts A and C of part 164; see also, most recently, 75 FR 40868 and 78 FR 5566 ). The guidance, NIST SP 800-66r2, provides information and resources to HIPAA-covered entities to improve their cybersecurity risk practices. [ 828 ] We also wish to alert readers of additional HHS resources and activities regarding cybersecurity best practices as recently summarized in an HHS strategy document that provides an overview of HHS recommendations to help the health care sector address cyber threats. [ 829 ] HHS has also recently published a website detailing recommended cybersecurity performance goals. [ 830 ] We intend to consider how the Medicare Promoting Interoperability Program can promote cybersecurity best practices for eligible hospitals and CAHs to inform potential future rulemaking proposals.
We recently released the CMS Interoperability and Prior Authorization final rule (CMS-0057-F), which appeared in the Federal Register on February 8, 2024 ( 89 FR 8758 ). This final rule aims to enhance health information exchange and access to health records for patients, healthcare providers, and payers, and improve prior authorization processes. In the final rule, we finalized the “Electronic Prior Authorization” measure under the HIE objective of the Merit-based Incentive Payment System (MIPS) Promoting Interoperability performance category and under the HIE objective of the Medicare Promoting Interoperability Program, beginning, for the Medicare Promoting Interoperability Program, in the EHR reporting period in CY 2027 ( 89 FR 8909 through 8927 ).
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36377 through 36381 ), we sought feedback in response to efforts across HHS to advance the public health information infrastructure, aimed to offer opportunities to further evolve the Medicare Promoting Interoperability Program, in collaboration with the CDC and ONC. We outlined a series of goals, followed by questions for commenters to consider and provide feedback for consideration in future rulemaking.
We received many comments on the RFI regarding public health reporting and data exchange, and we thank the commenters for responding to our request for information. While we are not responding to specific comments submitted in response to this RFI, we believe that this input is valuable in our efforts to continue to promote public health reporting and data exchange. We may consider some of this feedback to inform potential future rulemaking proposals.
In the May 2, 2024 Federal Register ( 89 FR 35934 ), we published the proposed rule titled “Medicare and Medicaid Programs and the Children's Health Insurance Program; Hospital Inpatient Prospective Payment Systems for Acute Care Hospitals and the Long-Term Care Hospital Prospective Payment System and Policy Changes and Fiscal Year 2025 Rates; Quality Programs Requirements; and Other Policy Changes” that would implement a new mandatory Medicare payment model under section 1115A of the Act—the Transforming Episode Accountability Model (TEAM).
As we stated in the proposed rule, we believe that this model will test ways to further our goals of reducing Medicare expenditures while preserving or enhancing the quality of care furnished to beneficiaries. We are finalizing several of the provisions from the proposed rule but not all of them, and we intend to address and finalize some provisions of the proposed rule in future rulemaking. We also note that some of the public comments were outside of the scope of the proposed rule. These out-of-scope public comments are not addressed in this final rule. We have summarized the public comments that are within the scope of the proposed rule and our responses to those public comments. However, we note that in this final rule we are not addressing most comments received with respect to the provisions of the proposed rule that we are not finalizing at this time. Rather, we will address them at a later time, in subsequent rulemaking, as appropriate.
The CMS Innovation Center has designed and tested numerous alternative payment models that each include specific payment, quality, and other policies. However, there are some ( print page 69627) general provisions that are very similar across models. The general provisions address beneficiary protections, model evaluation and monitoring, audits and record retention, rights in data and intellectual property, monitoring and compliance, remedial action, model termination by CMS, limitations on review, and miscellaneous provisions on bankruptcy and other notifications.
We proposed to implement the general provisions, described later in this section and in subpart E of part 512, based on similar requirements that have been previously finalized in existing model tests. In addition to the general provisions discussed here, TEAM-specific provisions that are uniquely tailored to this model are described elsewhere in this rule ( 89 FR 36381 ).
In § 512.500 of the proposed rule, we proposed that the general provisions would only be applicable to TEAM. We stated that the proposed general provisions would not, except as specifically noted in proposed part 512, subpart E, affect the applicability of other provisions affecting providers and suppliers under Medicare FFS, including the applicability of provisions regarding payment, coverage, and program integrity (such as those in parts 413, 414, 419, 420, and 489 of chapter IV of 42 CFR and those in parts 1001 through 1003 of chapter V of 42 CFR) ( 89 FR 36381 ).
We invited public comment on the general provisions proposed for TEAM.
Summaries of the public comments received, and our responses are set forth in this section of the final rule under the appropriate headings.
We proposed at § 512.505 of the proposed rule to define certain terms. We proposed to define the term “TEAM participant” to mean an acute care hospital that is identified under the terms of and defined in proposed § 512.505. We proposed to define “downstream participant” to mean an individual or entity that has entered into a written arrangement with a TEAM participant pursuant to which the downstream participant engages in one or more TEAM activities. We further proposed that a downstream participant may include, but would not be limited to, an individual practitioner, as defined for purposes of TEAM. We proposed to define “TEAM activities” to mean any activities impacting the care of model beneficiaries related to the test of TEAM performed under the terms of proposed 512 subpart E ( 89 FR 36381 ).
We describe additional proposed definitions in context throughout this section X.A.1. of the preamble of this final rule.
Section 1115A(b)(4) of the Act requires the Secretary to evaluate each model tested under the authority of section 1115A of the Act and to publicly report the evaluation results in a timely manner. The evaluation must include an analysis of the quality of care furnished under the model and the changes in program spending that occurred due to the model. Models tested by the CMS Innovation Center are rigorously evaluated. For example, when evaluating models tested under section 1115A of the Act, we require the production of information that is representative of a wide and diverse group of model participants and includes data regarding potential unintended or undesirable effects, such as cost-shifting. The Secretary must take the evaluation into account if making any determinations regarding the expansion of a model under section 1115A(c) of the Act.
In addition to model evaluations, the CMS Innovation Center regularly monitors model participants for compliance with model requirements. For the reasons described in section X.A.1. of the preamble of this final rule, these compliance monitoring activities are an important and necessary part of the model test.
Therefore, we proposed to codify at § 512.584 that TEAM participants and their downstream participants must comply with the requirements of 42 CFR 403.1110(b) (regarding the obligation of entities participating in the testing of a model under section 1115A of the Act to report information necessary to monitor and evaluate the model) and must otherwise cooperate with CMS' model evaluation and monitoring activities as may be necessary to enable CMS to evaluate TEAM in accordance with section 1115A(b)(4) of the Act. Participation in the evaluation may include, but is not limited to, responding to surveys and participating in focus groups. Additional details on the specific research questions that we proposed that the TEAM evaluation would consider can be found in section X.A.3.o.(4) of the preamble of this final rule. Further, we proposed to conduct monitoring activities according to proposed § 512.590(b), described in section X.A.3.i. of the preamble of this final rule, including obtaining such data as may be required by CMS to evaluate or monitor TEAM, which may include protected health information as defined in 45 CFR 160.103 and other individually identifiable data. ( 89 FR 36381 )
We received no comments on the proposals to require cooperation with model evaluation and monitoring and are finalizing the proposals without modification.
In the proposed rule, we proposed to allow CMS to use any data obtained in accordance with proposed § 512.588 to evaluate and monitor the proposed TEAM, as required by section 1115A(b)(4) of the Act and pursuant to § 512.590, described at section X.A.3.i. of the preamble of this final rule. We further proposed that, consistent with section 1115A(b)(4)(B) of the Act, that CMS would be allowed to disseminate quantitative and qualitative results and successful care management techniques, including factors associated with performance, to other providers and suppliers and to the public. We proposed that the data to be disseminated would include, but would not be limited to, patient de-identified results of patient experience of care and quality of life surveys, as well as patient de-identified measure results calculated based upon claims, medical records, and other data sources ( 89 FR 36381 ).
In the proposed rule we stated that in order to protect the intellectual property rights of TEAM participants and downstream participants, we proposed in § 512.588(c) to require TEAM participants and their downstream participants to label data they believe is proprietary and should be protected from disclosure under the Trade Secrets Act. We noted that this approach is already in use in other models currently being tested by the CMS Innovation Center, including End Stage Renal Disease Treatment Choices models. Any such assertions would be subject to review and confirmation prior to CMS acting upon such assertion.
We further proposed to protect such information from disclosure to the full extent permitted under applicable laws, including the Freedom of Information Act. Specifically, in proposed § 512.588(b), we proposed to not release data that has been confirmed by CMS to be proprietary trade secret information and technology of the TEAM participant or its downstream participants without the express written consent of the TEAM participant or its downstream participants, unless such release is required by law ( 89 FR 36382 ).
We received no comments on the proposed rights in data and intellectual ( print page 69628) property provisions and are finalizing the proposals without modification.
As stated in the proposed rule, as part of the CMS Innovation Center's monitoring and assessment of the impact of models tested under the authority of section 1115A of the Act, we have a special interest in ensuring that these model tests do not interfere with the program integrity interests of the Medicare program. For this reason, we monitor for compliance with model terms as well as other Medicare program rules. When we become aware of noncompliance with these requirements, it is necessary for CMS to have the ability to impose certain administrative remedial actions on a noncompliant model participant ( 89 FR 36382 ).
In the proposed rule, we stated that the terms of many models currently being tested by the CMS Innovation Center permit CMS to impose one or more administrative remedial actions to address noncompliance by a model participant. We proposed that CMS may impose any of the remedial actions set forth in proposed § 512.592 if we determine that the TEAM participant or a downstream participant—
- Has failed to comply with any or all of the terms of TEAM;
- Has failed to comply with any applicable Medicare program requirement, rule, or regulation;
- Has taken any action that threatens the health or safety of a beneficiary or other patient;
- Has submitted false data or made false representations, warranties, or certifications in connection with any aspect of TEAM;
- Has undergone a change in control (as defined in proposed § 512.505) that presents a program integrity risk;
- Is subject to any sanctions of an accrediting organization or a Federal, state, or local government agency;
- Is subject to investigation or action by HHS (including the HHS-OIG and CMS) or the Department of Justice due to an allegation of fraud, a pattern of improper billing, or significant misconduct, including being subject to the filing of a complaint or filing of a criminal charge, being subject to an indictment, being named as a defendant in a False Claims Act qui tam matter in which the Federal Government has intervened, or similar action; or
- Has failed to demonstrate improved performance following any remedial action imposed by CMS.
- Has misused or disclosed beneficiary-identifiable data in a manner that violates any applicable statutory or regulatory requirements or that is otherwise non-compliant with the provisions of the TEAM data sharing agreement.
At proposed § 512.592(b), we proposed to codify that CMS may take one or more of the following remedial actions if CMS determined that one or more of the grounds for remedial action described in proposed § 512.592(a) had taken place—
- Notify the TEAM participant and, if appropriate, require the TEAM participant to notify its downstream participants of the violation;
- Require the TEAM participant to provide additional information to CMS or its designees;
- Subject the TEAM participant to additional monitoring, auditing, or both;
- Prohibit the TEAM participant from distributing TEAM payments;
- Require the TEAM participant to terminate, immediately or by a deadline specified by CMS, its agreement with a downstream participant with respect to TEAM;
- Terminate the TEAM participant from the model test;
- Require the TEAM participant to submit a corrective action plan in a form and manner and by a date specified by CMS;
- Discontinue the provision of data sharing and reports to the TEAM participant;
- Recoup TEAM payments;
- Reduce or eliminate a TEAM payment otherwise owed to the TEAM participant, as applicable; or
- Such other action as may be permitted under the terms of TEAM.
In the proposed rule, we noted that because TEAM is a mandatory model, we would not expect to use the proposed provision that would allow CMS to terminate a TEAM participant's participation in the model, except in circumstances in which the TEAM participant has engaged, or is engaged in, egregious actions.
We invited public comment on these proposed provisions regarding the proposed grounds for remedial actions, remedial actions generally, and whether additional types of remedial action would be appropriate ( 89 FR 36382 ).
We received no comments on the proposed remedial actions and are finalizing the proposals without modification.
In the proposed rule, we proposed certain provisions that would allow CMS to terminate TEAM under certain circumstances. Section 1115A(b)(3)(B) of the Act requires the CMS Innovation Center to terminate or modify the design and implementation of a model, after testing has begun and before completion of the testing, unless the Secretary determines, and the Chief Actuary certifies with respect to program spending, that the model is expected to: improve the quality of care without increasing program spending; reduce program spending without reducing the quality of care; or improve the quality of care and reduce spending ( 89 FR 36382 ).
We proposed at § 512.596 that CMS could terminate TEAM for reasons including, but not limited to, one of the following circumstances:
- CMS determines that it no longer has the funds to support TEAM.
- CMS terminates TEAM in accordance with section 1115A(b)(3)(B) of the Act.
As stated in the proposed rule, section 1115A(d)(2)(E) of the Act and proposed § 512.596 provide that termination of TEAM in accordance with section 1115A(b)(3)(B) of the Act would not be subject to administrative or judicial review.
To ensure model participants had appropriate notice in the case of the termination of TEAM by CMS, we also proposed to codify at § 512.596 that we would provide TEAM participants with written notice of the model termination, which would specify the grounds for termination as well as the effective date of the termination.
We received no comments on the model termination by CMS proposals and are finalizing the proposals without modification.
In proposed § 512.594, we proposed to codify the preclusion of administrative and judicial review under section 1115A(d)(2) of the Act ( 89 FR 36382 ). Section 1115A(d)(2) of the Act states that there is no administrative or judicial review under section 1869 or 1878 of the Act or otherwise for any of the following:
- The selection of models for testing or expansion under section 1115A of the Act.
- The selection of organizations, sites, or participants to test models selected.
- The elements, parameters, scope, and duration of such models for testing or dissemination.
- Determinations regarding budget neutrality under section 1115A(b)(3) of the Act.
- The termination or modification of the design and implementation of a model under section 1115A(b)(3)(B) of the Act. ( print page 69629)
- Determinations about expansion of the duration and scope of a model under section 1115A(c) of the Act, including the determination that a model is not expected to meet criteria described in paragraph (1) or (2) of such section.
In the proposed rule, we proposed to interpret the preclusion from administrative and judicial review regarding the CMS Innovation Center's selection of organizations, sites, or participants to test TEAM to preclude from administrative and judicial review our selection of a TEAM participant, as well as our decision to terminate a TEAM participant, as these determinations are part of our selection of participants for TEAM. ( 89 FR 36383 )
We invited public comment on the proposed codification of these statutory preclusions of administrative and judicial review for TEAM, as well as our proposed interpretations regarding their scope.
The following is a summary of the comments we received on the limitations on review proposals and our responses:
Comment: A commenter believed the use of administrative and judicial preclusion language is unlawful, beyond the agency's authority, and unenforceable.
Response: We thank the commenter for their feedback. However, we disagree that preclusion of administrative and judicial review is beyond the agency's authority. We note that the language in section 1115A(d)(2) of the Act precludes administrative or judicial review for a range of policies for Innovation Center models, including selection of sites or participants. We also believe that the language in 1115A(d)(2) clearly indicates that the intent of the statute was to ensure that CMS has authority to test models, and precluding administrative or judicial review of the specific policies listed above is necessary to ensure our ability to implement models. Allowing administrative or judicial review could hinder our ability to test models, if entities are able to appeal CMS' decisions such as our selection of sites or participants and certain aspects of model design and implementation.
Comment: A commenter stated that administrative and judicial review may be the only effective option available to assure that a TEAM participant's network is adequate. The commenter stated that a beneficiary or downstream participant should have the opportunity to challenge an exclusion of a downstream participant from participating in the model to protect beneficiary choice rights. They are concerned that in the case of a geographically established mandatory bundle, beneficiary choice or provider protection could be seriously eroded in areas where there are a limited number of hospitals, and adversely affect Medicare beneficiaries' access to appropriate downstream care following an acute care hospital stay.
Response: We share the commenter's interest in ensuring that beneficiaries are not negatively affected by model requirements and preserving and protecting beneficiary access but disagree that allowing for administrative or judicial review of certain TEAM policies would address the potential issues raised by the commenter. TEAM does not affect a beneficiary's ability to choose how or where they receive care, and we are finalizing several policies in section X.A.3.i. of this final rule in order to protect beneficiary choice and access for those receiving services from TEAM participants. At § 512.582(a)(1), we are finalizing that TEAM participants and their collaborators and other partners may not restrict beneficiaries' freedom to choose to receive care from any provider or supplier. We are also finalizing (at § 512.582(a)(3)) a requirement that TEAM participants provide, at discharge from the hospital, a list of all local post-acute care providers participating in the Medicare program. In addition, while TEAM participants may recommend certain providers or suppliers, they may not restrict access or limit beneficiary choice to those providers or suppliers with whom they have a relationship.
Finally, we note that the TEAM participants will be acute care hospitals, not downstream care partners such as post-acute care facilities or other providers. In addition, we note that the model does not require a provider network. We believe our policies referenced above with regard to beneficiary protections, choice, and access, will address the commenter's concern.
After consideration of the public comments we received on the proposed limitations on review, we are finalizing § 512.594 as proposed without modification.
We noted in the proposed rule that the proposed TEAM would have a defined period of performance, but final payment under the model might occur long after the end of the performance period. In some cases, a TEAM participant could owe money to CMS. We recognize that the legal entity that is the TEAM participant could experience significant organizational or financial changes during or after the period of performance for TEAM. To protect the integrity of the proposed TEAM and Medicare funds, we proposed a number of provisions to ensure that CMS is made aware of events that could affect a TEAM participant's ability to perform its obligations under TEAM, including the payment of any monies owed to CMS ( 89 FR 36383 ).
First, in proposed § 512.595(a), we proposed that a TEAM participant must promptly notify CMS and the local U.S. Attorney Office if it files a bankruptcy petition, whether voluntary or involuntary. Because final payment may not take place until after the TEAM participant ceases active participation in TEAM, we further proposed that this requirement would apply until final payment has been made by either CMS or the TEAM participant under the terms of the model and all administrative or judicial review proceedings relating to any payments under TEAM have been fully and finally resolved.
In the proposed rule, we, specifically, proposed that notice of the bankruptcy must be sent by certified mail within 5 days after the bankruptcy petition has been filed and that the notice must contain a copy of the filed bankruptcy petition (including its docket number), unless final payment has been made under the terms of TEAM and all administrative or judicial review proceedings regarding TEAM payments between the TEAM participant and CMS have been fully and finally resolved. The notice to CMS must be addressed to the CMS Office of Financial Management, Mailstop C3-01-24, 7500 Security Boulevard, Baltimore, Maryland 21244 or to such other address as may be specified for purposes of receiving such notices on the CMS website.
In the proposed rule, we stated that by requiring the submission of the filed bankruptcy petition, CMS would obtain information necessary to protect its interests, including the date on which the bankruptcy petition was filed and the identity of the court in which the bankruptcy petition was filed. We recognized that such notices may already be required by existing law, but CMS often does not receive them in a timely fashion, and they may not specifically identify TEAM. The failure to receive such notices on a timely basis can prevent CMS from asserting a claim in the bankruptcy case. We were particularly concerned that a TEAM participant may not furnish notice of bankruptcy after it has completed its performance in TEAM, but before final ( print page 69630) payment has been made or administrative or judicial proceedings have been resolved. We believe our proposal was necessary to protect the financial integrity of the proposed TEAM and the Medicare Trust Funds.
Second, in proposed § 512.595(b), we proposed that the TEAM participant would have to provide written notice to CMS within 30 days of any change in the TEAM participant's legal name becoming effective. The notice of legal name change would have to be in a form and manner specified by CMS and include a copy of the legal document effecting the name change, which would have to be authenticated by the appropriate state official. The purpose of this final notice requirement is to ensure the accuracy of our records regarding the identity of TEAM participants and the entities to whom TEAM payments should be made or against whom payments should be demanded or recouped. We solicited comment on requiring notice to be furnished promptly, that is, within 30 days after a change in legal name has become effective.
Third, in proposed § 512.595(c), we proposed that the TEAM participant would have to provide written notice to CMS at least 90 days before the effective date of any change in control. We proposed that the written notification must be furnished in a form and manner specified by CMS. For purposes of this notice obligation, we proposed that a “change in control” would mean any of the following: (1) The acquisition by any “person” (as such term is used in sections 13(d) and 14(d) of the Securities Exchange Act of 1934) of beneficial ownership (within the meaning of Rule 13d-3 promulgated under the Securities Exchange Act of 1934), of beneficial ownership (within the meaning of Rule 13d-3 promulgated under the Securities Exchange Act of 1934), directly or indirectly, of voting securities of the TEAM participant representing more than 50 percent of the TEAM participant's outstanding voting securities or rights to acquire such securities; (2) the acquisition of the TEAM participant by any individual or entity; (3) the sale, lease, exchange or other transfer (in one transaction or a series of transactions) of all or substantially all of the assets of the TEAM participant; or (4) the approval and completion of a plan of liquidation of the TEAM participant, or an agreement for the sale or liquidation of the TEAM participant. The proposed requirement and definition of change in control are the same requirements and definition used in certain models that are currently being tested under section 1115A authority. We believe this final notice requirement is necessary to ensure the accuracy of our records regarding the identity of model participants and to ensure that we pay and seek payment from the correct entity. For this reason, we proposed that if CMS determined in accordance with proposed § 512.592(a)(5) that a TEAM participant's change in control would present a program integrity risk, CMS could take remedial action against the TEAM participant under proposed § 512.592(b). In addition, to ensure payment of amounts owed to CMS, we proposed that CMS may require immediate reconciliation and payment of all monies owed to CMS by a model participant that is subject to a change in control.
We received one timely public comment on the proposed bankruptcy and other notifications requirements.
Comment: A commenter requested CMS change the timelines for the bankruptcy and other notifications provision to be consistent with other Medicare reporting timelines, as provider burden is lessened when timelines are consistent. Specifically, the commenter asked that TEAM provision conform to the timelines set forth in the “Disclosures of Ownership and Additional Disclosable Parties Information for Skilled Nursing Facilities and Nursing Facilities; Definitions of Private Equity Companies and Real Estate Investment Trusts for Medicare Providers and Suppliers” Final Rule published November 17, 2023, which implemented portions of the Affordable Care Act, requiring the disclosure of certain ownership, managerial, and other information and the required timelines associated with each.
Response: While we agree with the commenter that in general, aligning reporting timelines across the various Medicare program requirements is desirable, we note that the specific timelines and parameters of TEAM (and other Innovation Center models) necessitate that we collect many types of information, such as ownership and financial information, on a more frequent basis, as applicable, than those finalized in the regulation referenced by the commenter ( 88 FR 80141 ). In the “Disclosures of Ownership and Additional Disclosable Parties Information for Skilled Nursing Facilities and Nursing Facilities; Definitions of Private Equity Companies and Real Estate Investment Trusts for Medicare Providers and Suppliers” Final Rule, CMS finalized certain timelines for reporting of specific information for Medicare-enrolled providers and other entities; such reporting would occur upon initial enrollment into Medicare or Medicaid, upon change of ownership, or during revalidation, which occurs every five years. We do not believe this frequency of reporting would be sufficient for TEAM. We proposed to require reporting on the timelines specified in the proposed rule in order to effectively protect CMS against potential program integrity issues that may arise due to changes in control during model participation, expeditiously make payments (or send demand letters) to TEAM participants, and generally ensure we have accurate information on the entities participating in our models in order to monitor and enforce model requirements. As noted above, our proposed timelines and requirements mirror those utilized by other models tested under section 1115A authority. Given this, we are finalizing at § 512.595 our proposals to require a TEAM participant to (1) promptly notify CMS and the local U.S. Attorney Office if it files a bankruptcy petition, whether voluntary or involuntary, within 5 days of the bankruptcy petition; (2) provide written notice to CMS within 30 days of any change in the TEAM participant's legal name becoming effective; and (3) provide written notice to CMS at least 90 days before the effective date of any change in control.
As discussed in the proposed rule ( 89 FR 35934 ), we proposed the implementation and testing of the Transforming Episode Accountability Model (TEAM), a new mandatory alternative payment model under the authority of section 1115A of the Act, beginning on January 1, 2026, and ending on December 31, 2030. TEAM would test whether an episode-based pricing methodology linked with quality measure performance for select acute care hospitals reduces Medicare program expenditures while preserving or improving the quality of care for Medicare beneficiaries who initiate certain episode categories. Specifically, TEAM proposals included the testing of five surgical episode categories: Coronary Artery Bypass Graft Surgery (CABG), Lower Extremity Joint Replacement (LEJR), Major Bowel Procedure, Surgical Hip/Femur Fracture Treatment (SHFFT), and Spinal Fusion.
As stated in the proposed rule, this model falls within a larger framework of activities initiated by the CMS Innovation Center during the past several years, including the release of the CMS Innovation Center strategic ( print page 69631) refresh and the comprehensive specialty strategy. [ 831 832 ] The strategic refresh includes a goal of having 100 percent of Medicare FFS beneficiaries and the vast majority of Medicaid beneficiaries in an accountable care relationship by 2030. Episode-based payment models, such as TEAM, can be a tool to support this goal by increasing provider participation in value-based care initiatives with accountability for quality and cost outcomes. To further the goals of the strategic refresh, the CMS Innovation Center released the comprehensive specialty care strategy in 2022, which includes an element to maintain momentum established by episode-based payment models and supports development of TEAM. [ 833 ] In addition, in July 2023, we published a Request for Information (RFI) to gain public input on design elements for a new mandatory bundled payment model. [ 834 ] Given TEAM's alignment with many strategic facets of the CMS Innovation Center, our proposal to test a new episode-based payment model for acute care hospitals is based on: (1) lessons learned from testing the Bundled Payments for Care Improvement (BPCI) Initiative, the BPCI Advanced Model, and the Comprehensive Care for Joint Replacement (CJR) Model; and (2) comments received from the Episode-based Payment Model RFI ( 88 FR 45872 ) published in the Federal Register .
As stated in the proposed rule, and finalized in this final rule, TEAM participants continue to bill Medicare under the traditional FFS system for services furnished to Medicare FFS beneficiaries. However, the TEAM participant may also receive a reconciliation payment amount from CMS depending on their Composite Quality Score (CQS) and if their performance year spending is less than their reconciliation target price. As TEAM is a two-sided risk model, meaning the model requires TEAM participants to be accountable for performance year spending that is above or below their reconciliation target price, TEAM participants may also owe CMS a repayment amount depending on their CQS and if their performance year spending is more than their reconciliation target price.
As stated in the proposed rule, and finalized in this final rule, the model performance period for TEAM will consist of five performance years, beginning January 1, 2026, and ending December 31, 2030, with final data submission of clinical data elements and quality measures in CY 2031 to account for episodes ending in CY 2030, and final reconciliation reports and TEAM reconciliation payment amounts and repayment amounts in CY 2031. Further information about all the proposals in TEAM may be found at ( 89 FR 35934 ).
CMS believes an episode-based payment structure may improve beneficiary care by aligning incentives in pursuit of improved quality and reduced spending. A FFS payment system pays health care providers and suppliers for discrete services over a single episode, potentially resulting in fragmented care and duplicative use of resources. Paying for discrete services may also not provide sufficient financial incentive for health care providers and suppliers to invest in quality improvement and care coordination that could help avoid adverse outcomes. Further, providers and suppliers may be paid under different FFS payment systems which may create challenges managing beneficiaries in an episode. Therefore, providers and suppliers have less of an incentive to collaborate to improve the quality of care and decrease the cost and unnecessary utilization of services.
An episode-based payment methodology creates an incentive for participating providers and suppliers to coordinate across care settings as the participating entity takes responsibility for the quality and cost outcomes across the entire episode. All of the projected payments to the physician, hospital, and other health care provider and supplier services are combined into a target price. This target price represents the expected cost of all items and services furnished to a beneficiary during an episode. Health care providers included in such initiatives may either realize a financial gain or loss, based on how successfully they perform on quality measure assessment and manage resources and total costs throughout each episode. Payment models that hold entities accountable for spending and quality performance metrics for an entire episode can motivate health care providers to furnish services more efficiently, to better coordinate care, and to improve the quality of care.
The CMS Innovation Center has tested episode-based payment models for over a decade, including the BPCI initiative, the BPCI Advanced Model, and the CJR Model. The CJR Model and the BPCI Advanced Models are current CMS Innovation Center model tests that are set to end on December 31, 2024, and December 31, 2025, respectively. When considering the future of episode-based payment models, we reviewed results of the CJR Model and the BPCI Advanced Model given promising evaluation findings that support these models reducing episode payments, before accounting for incentive payments, and maintaining quality of care, as described further in section X.A.2.c. of the preamble of this final rule. However, both models experienced significant model changes, including changes in participation volume, in the later years of their model test and assessing the results of these models based on their current methodologies requires additional evaluation data, which would not be available until after each model has concluded. We also note additional challenges that arise from voluntary models, including the BPCI Advanced model, where selection bias, stemming from self-selection into and out of the model and selection of clinical episode categories or clinical episode service line groups, can make it more difficult to evaluate and produce generalizable results. We believe TEAM will allow the CMS Innovation Center to test a new episode-based payment model that builds upon lessons learned in previous episode-based payment models by incorporating the most promising model features, while also continuing care transformation efforts that we have promoted through the CJR or BPCI Advanced models.
As stated in the proposed rule, if TEAM is successful, we hope this model would establish the framework for managing episodes as a standard practice in Traditional Medicare. TEAM includes features that are attentive to operational feasibility for both participants and CMS, such as how often reconciliation would be conducted to minimize administrative burden, a pricing methodology that would be responsive to providers with varying levels of experience and different patient populations, and the selection of episodes with sufficient volume that would warrant standard care pathways during the acute and post-acute care periods of an episode. In the proposed rule, we stated that any future policy changes to this proposed model test, such as the addition of episode categories, would be implemented ( print page 69632) through future notice and comment rulemaking.
Increasing quality, patient-centeredness, and cost-effective care requires collaboration among hospitals, physicians, and post-acute care (PAC) providers. To encourage this collaboration, TEAM proposed to further align incentives between hospitals and physicians by specifying certain types of financial arrangements that participants may elect to pursue to share reconciliation payment amounts received from CMS under the model. By doing so, TEAM participants would be able to share incentives with downstream providers and suppliers when they achieve higher quality and more cost-effective care through collaboration.
Medicare beneficiaries can experience fragmented and costly care, distinguished by frequent diagnostics, imaging, tests and other treatment approaches delivered by different providers across different sites of care. [ 835 ] A 2022 study examining fragmentation of ambulatory care for Medicare FFS beneficiaries found that four in ten beneficiaries experience highly fragmented care, with a mean of 13 ambulatory visits across seven practitioners in one year. [ 836 ] Fragmented care is further evident when focusing on the clinical management of Medicare beneficiaries for acute procedural care since these beneficiaries may be receiving care from different physicians in different settings before, during, and after their procedure. [ 837 ] In the absence of effective communication between patients, families, physicians, hospitals, and other care settings, beneficiaries receiving acute procedural care may not receive comprehensive care management and coordination. TEAM is based on the premise—supported by evidence from the CJR and BPCI Advanced model evaluations—that appropriately aligned financial incentives would improve or maintain quality of care for beneficiaries who are in an episode, while also achieving reductions in episode spending. [ 838 ]
Care fragmentation in acute surgical procedures in the United States is well documented, leading to care variation and inefficiencies producing unfavorable patient outcomes and increased health spending. [ 839 840 841 ] Given the variation in acute surgical care and costs, including post-acute care costs immediately following a procedure, significant literature has been devoted to evaluating opportunities to improve the quality and efficiency of care. [ 842 843 ] This includes the design and implementation of standardized care processes that emphasize high-value care that can support episode-based care initiatives. For example one study found that, “Enhanced Recovery After Surgery protocols have resulted in shorter length of hospital stay by 30 percent to 50 percent and similar reductions in complications, while readmissions and costs are reduced”. [ 844 ] Moreover, other findings focus on perioperative care delivery and indicate, “that through elements that emphasize care coordination, standardization, and patient-centeredness, perioperative surgical home programs can improve patient postoperative recovery outcomes and decrease hospital utilization”. [ 845 ]
CMS, commercial payers, and other stakeholders are continuously testing a variety of approaches to constructing episodes of care, including through different patient populations, clinical episode categories, and pricing methodologies. [ 846 847 848 ] Though the results of alternative payment models focused on episodes of care have been mixed, evidence related to models' ability to realize savings and improve quality is promising, especially given the 10 years of experience yielded from participants and the CMS Innovation Center model tests. The BPCI Advanced and CJR models are still being tested, and the effects of the models' care redesign changes aimed to achieve Medicare savings and maintain or improve quality of care are still being evaluated, see section X.A.2.c. of the preamble of this final rule, but have generated evidence from multiple evaluation reports to support the design of TEAM. Beyond quantitative data, qualitative data collected from model participants and data from site visits indicate care transformation is happening, and quality of care is improving across the spectrum. Qualitative data range from reported improved relationships between inpatient providers and post-acute care (PAC) providers, to reshaping patient and provider expectations about appropriate discharge destinations, to process changes, such as standardized care pathways, identification and mitigation of medical and social risk factors, monitoring patients in the post-discharge period, and connecting patients to primary care providers. As noted in section X.A.2.c. of the preamble of this final rule, evaluation results from the previous and current episode-based payment models consistently indicate that these models can reduce episode payments, before considering incentive payments, and ( print page 69633) generally without compromising quality of care.
The CMS Innovation Center previously tested episode-based payment approaches among acute episodes, including the Medicare Acute Care Episode (ACE) demonstration and the BPCI Initiative, and currently is testing additional approaches under the BPCI Advanced model and the CJR model. [ 849 ] The ACE demonstration tested a bundled payment approach for cardiac and orthopedic inpatient surgical services and procedures. All Medicare Part A and Part B services pertaining to the inpatient stay were included in the ACE demonstration episodes of care. Evaluations results found that Medicare saved an average of $585 per episode from the combined Medicare Part A and B expected payments or a total of $7.3 million across all episodes (12,501 episodes), all ACE MS-DRGs, and four ACE Sites. However, increases in post-acute care spending reduced these savings by approximately 45 percent, resulting in per episode savings of $319 and total net savings of approximately $4 million. With respect to quality of care, findings suggest that the ACE sites maintained their quality-of-care levels without any systematic or consistent changes in clinical outcomes or in the type of patients they admitted in response to the demonstration. Despite the lack of strong quantitative evidence for realized improvements in quality, there was qualitative evidence that hospitals worked to improve processes and outcomes. [ 850 ]
The BPCI initiative tested whether linking payments for providers that furnish Medicare-covered items and services during an episode related to an inpatient hospitalization could reduce Medicare expenditures while maintaining or improving quality of care.
- Model 1 episodes were limited to the acute inpatient hospitalizations for all MS-DRGs.
- Model 2 episodes began with a hospital admission and extended for 30, 60, or 90 days after discharge.
- Model 3 episodes began with the initiation of post-acute care following a hospital admission and extended for 30, 60, or 90 days.
- Model 4 episodes began with a hospital admission and included readmissions within 30 days after discharge.
Model 1 was unique, as compared to Models 2-4, in that target prices weren't generated but awardees received a predetermined discount percentage to their Medicare Inpatient Prospective Payment System (IPPS) operating payment rates for episodes at their hospital. Model 1 had a small volume of participants, however, evaluation results found that there were no consistent negative or positive statistically significant impacts to Medicare payments or quality of care effects on Medicare beneficiaries. [ 851 ] Similarly, Model 4 had a small volume of participants, and by the end of the model there was no change in allowed payments nor were there any changes in the quality of care as measured by claims-based quality measures. [ 852 ]
Evaluation results for BPCI Models 2 and 3 were more robust given the greater volume of participants in each model. Similar to Model 1 and Model 4, quality of care generally remained unchanged in BPCI Models 2 and 3. With respect to the financial performance of the models, findings demonstrated reductions in FFS payments of $1,193 million for Model 2 and $232 million for Model 3. However, Medicare experienced net losses of $418 million (p 0.05) for Model 2, or $332 per episode, and $110 million (p 0.05) for Model 3, or $714 per episode, after accounting for reconciliation payments to participants. These net losses to Medicare represented 1.3 percent of what payments would have been absent BPCI under Model 2 and 3.1 percent under Model 3. The largest contributing factor to these losses was the elimination of participants' repayment responsibility during the initial part of the model for all participants, and then in later years for certain participants due to episode attribution errors. If CMS had not eliminated repayment responsibility, and assuming model participation remained the same, Model 2 would have resulted in no net losses or savings, and net losses under Model 3 would have been reduced to $66 million (p 0.05), or 1.9 percent of what payments would have been absent BPCI. [ 853 ]
We currently are testing the BPCI Advanced model, which is a voluntary episode-based model based on the BPCI Initiative's Model 2, that tests whether linking payments for an episode will incentivize health care providers to invest in innovation and care redesign to improve care coordination and reduce expenditures, while maintaining or improving the quality of care for Medicare FFS beneficiaries. We are still evaluating the effects of the BPCI Advanced model on patient experience of care, quality outcomes, and cost of care for Medicare FFS beneficiaries. However, evaluation results to date demonstrated reductions in episode payments and maintenance of quality of care, but the model has thus far been unable to generate Medicare savings. As of Model Year 3 (2020), BPCI Advanced participants reduced average episode payments by 3.8 percent or $1,028 per episode, and more specifically 3.1 percent ($796 per episode) for medical episodes and 5.8 percent ($1,800 per episode) for surgical episodes. Despite the reductions in FFS payments, after accounting for reconciliation payments to participants, Medicare had a net loss of $114 million in 2020, or 0.8 percent of what Medicare payments would have been in absence of the model. When looking at Medicare savings by episode type, surgical episodes resulted in an estimated net savings of $71.3 million, or 2.3 percent, but those savings were offset by medical episodes which resulted in an estimated net loss of $200.5 million, or 1.9 percent. [ 854 ] The BPCI Advanced model implemented changes, most notably in 2021-23, and most recently made further changes to extend the model through 2025 and support provider engagement in value-based care.
We are also currently testing the CJR model, which is a mandatory episode-based payment model in 34 metropolitan statistical areas (MSAs) for lower extremity joint replacement episodes that encourages hospitals, physicians, and PAC providers to work together to improve the quality and coordination of care from the initial hospitalization or outpatient procedure through recovery. Evaluation results to date have indicated that in the first four performance years, mandatory hospitals generated $72 million dollars in savings to Medicare, although not statistically significant. But in Performance Year 5, reconciliation payments substantially increased generating $95.4M in statistically significant Medicare losses, due to adjustments made to the model made during the COVID-19 Public Health Emergency (PHE). CMS enacted these temporary adjustments, which effectively waived downside risk for all CJR episodes, in order to minimize any financial burden associated with model participation given the financial challenges and uncertainties hospitals faced early in the COVID-19 PHE. These adjustments resulted in reconciliation payments being triple what they were in previous years, which reversed the savings trajectory and resulted in statistically significant losses to Medicare for mandatory hospitals. The losses in Performance Year 5 were large enough to offset total estimated savings prior to the public health emergency. [ 855 ] Like the BPCI Advanced model, the CJR model was revised and extended until December 31, 2024.
As stated in the proposed rule, we believe that providers', suppliers', and CMS' experiences with the BPCI Advanced and CJR models support the design of TEAM. Stakeholders both directly and indirectly involved in testing the BPCI Advanced and CJR models have conveyed that they perceive episode-based payments to be an effective mechanism for advancing better, more accountable care through care coordination and opportunities to improve care efficiency. CMS has also heard similar sentiment through other efforts including the CMS Innovation Center's Specialty Care Strategy Listening Session and recent Episode-based Payment Model Request for Information (RFI) ( 88 FR 45872 ). [ 856 ]
Further information of why specific elements of the models and initiatives were incorporated into TEAM's designs is discussed later in this final rule.
In 2021, the CMS Innovation Center announced a strategic refresh with a vision of having a health care system that achieves equitable outcomes through high quality, affordable, person-centered care. [ 857 ] To guide this updated vision, the CMS Innovation Center intends to design, implement, and evaluate future models with a focus on five strategic objectives: (i) driving accountable care; (ii) advancing health equity; (iii) supporting innovation; (iv) addressing affordability; and (v) partnering to achieve system transformation. One of the goals established by the strategic refresh was having 100 percent of traditional Medicare beneficiaries and the vast majority of Medicaid beneficiaries in accountable care relationships by 2030. This means that beneficiaries should experience longitudinal, accountable care with providers that are responsible for the quality and total cost of their care. Beneficiaries will experience accountable care relationships mostly through advanced primary care or accountable care organizations (ACOs), and these entities are expected to coordinate with or fully integrate specialty care to deliver whole-person care.
To support specialty care integration, the CMS Innovation Center released a comprehensive specialty strategy to test models and innovations supporting access to high-quality, integrated specialty care across the patient journey—both longitudinally and for procedural or acute services. [ 858 ] Specialty integration cannot be achieved with a single approach given a beneficiary's health needs may change influencing the types of providers and settings where they receive care. Therefore, the specialty care strategy consists of four elements: (i) enhancing specialty care performance data transparency; (ii) maintaining momentum on acute episode payment models and condition-based models; (iii) creating financial incentives within primary care for specialist engagement; and (iv) creating financial incentives for specialists to affiliate with population-based models and move to value-based care. As stated in the proposed rule, TEAM falls within the second element of the specialty care strategy and utilizes lessons learned from our experience with the BPCI Advanced model and the CJR model to design TEAM as a new episode-based payment model that would focus on accountability for quality and cost, health equity, and specialty integration. TEAM is further informed by the Episode-Based Payment Model RFI ( 88 FR 45872 ) published in July 2023, which gathered public comment on potential model design elements.
As stated in the proposed rule, TEAM represents one aspect of the specialty care strategy, and does not capture all beneficiaries, providers, and care settings to achieve complete person-centered value-based care on its own. Improving the health care system for Medicare beneficiaries requires a comprehensive approach that cannot be addressed by a single model or initiative since beneficiary health care needs are dynamic across the patient care continuum. This means TEAM would center accountability on beneficiary health care needs during narrow, focused periods of acute and post-acute care while health care needs outside of this scope would be addressed with other elements of the specialty care strategy. Therefore, we believe TEAM would complement other elements of the specialty care strategy (for example, another element of the strategy is to share TEAM-style episode data with ACOs) and would promote care transformation that generates standard care pathways and new best practices across broad patient populations (not just Medicare FFS).
The following is a summary of comments we received on the overall goals and evidence supporting the implementation of TEAM, as well as general comments about TEAM, and our responses to these comments:
Comment: Many commenters supported certain model aspects of TEAM while others supported the overarching goals of TEAM and its advancement towards greater value-based care. Some commenters were excited TEAM was built from previous bundled payment models, with a commenter noting successful participation in the CJR model and another commenter providing specific examples how episode-based payment models support medication reconciliation strategies and create incentives to address social needs. A ( print page 69635) commenter noted that TEAM will bring more hospitals into patient-first care initiatives. Another commenter applauded CMS efforts to financially incentivize performance on safety and quality across the continuum of care through payment models such as TEAM.
Response: We thank these commenters for their support of our efforts to move forward with TEAM. We are finalizing the majority of TEAM's proposals in this final rule and finalizing others with modifications. We are also not finalizing certain proposals and we may undergo notice and comment rulemaking to propose new policies in the future.
Comment: Several commenters requested CMS extend the proposed rule comment period to review TEAM proposals due to the scope and breadth of the model. Of these commenters, a commenter indicated CMS has not heeded calls for heightened engagement with providers as the model is developed and implemented and provided an example of CMS denying a comment period extension for the Episode-Based Payment Model RFI ( 88 FR 45872 ). Another commenter indicated that additional time beyond 60 days is necessary to fully evaluate and analyze these proposed policies and their full impact across the health care spectrum, especially in light of CMS proposing another hospital-based mandatory payment model just four weeks after it proposed TEAM.
Response: We acknowledge the commenters request for a comment period extension and considered the request prior to the proposed rule ( 89 FR 35934 ) comment period end date. However, we received robust comment on the proposed rule, so we believe the 60-day comment period window provided sufficient time to review TEAM proposals. This time period is consistent with comment period windows for other rules where a CMS Innovation Center model has been proposed. We had publicly signaled during the publication of the Episode-Based Payment Model RFI ( 88 FR 45872 ), that a model would be implemented via notice and comment rulemaking, and that we anticipated the model to be implemented no earlier than 2026. Given the desire to start this model in 2026, we wanted to propose TEAM in notice and comment rulemaking well in advance of model implementation to give ample time for participants to prepare for participation. We recognize that stakeholders had to review both the Medicare Program; Alternative Payment Model Updates and the Increasing Organ Transplant Access (IOTA) Model ( 89 FR 43518 ) proposed rule, and this proposed rule ( 89 FR 35934 ) including TEAM, but feel that staggered, rather than simultaneous, publication of these two proposed rules provided the public more time to review each model's policies.
We will continue to engage the public and stakeholders throughout the implementation of TEAM.
Comment: Some commenters acknowledged the goal of advancing toward more accountable and coordinated care but have overarching concerns for the implementation of TEAM. Many commenters indicated that CMS is placing too much financial risk on providers and that there is not enough opportunity for reward, especially considering the significant upfront investments required. Some commenters suggested that TEAM's focus was on reducing Medicare spending and not enough emphasis on improving patient care. Some commenters suggested TEAM not be finalized unless significant changes are implemented.
Response: We appreciate these commenters concerns, but we disagree that TEAM does not have enough emphasis on improving beneficiary care. As discussed in section X.A.3.c of the preamble of this final rule, we purposefully selected specific quality measures for TEAM to assess patient safety, care coordination, and patient outcomes. Further, we are finalizing a referral to primary care services policy, in section X.A.3.l of the preamble of this final rule, because we believe taking steps to link beneficiaries to primary care can lead to positive longer term health outcomes. With respect to commenters' concerns about too much financial risk, we have addressed these comments throughout the applicable sections of this final rule, including in, but not limited to, the discussions about participation tracks in section X.A.3.a.(3), and the pricing methodology in section X.A.3.d of the preamble of this final rule. In response to the commenters who suggested that TEAM not be finalized unless significant changes are implemented, we note that we are finalizing TEAM; we are finalizing some policies as proposed and we are finalizing others with modification. There are also certain proposed policies that we are not finalizing, and we will instead go through rulemaking in the future to promulgate new policies that could be finalized before the model start date.
Comment: A commenter supported the surgical nature of TEAM and looks forward to other APM options for non-surgical conditions and suggested MIPS Value Pathways or total cost of care models tailored to sepsis and Congestive Heart Failure, as these two conditions account for nearly half of all readmissions and take an extensive amount of time to cure or cover. Another commenter is supportive of CMS' efforts to increase opportunities for specialists to engage in APMs and believes episode-based payment models present an opportunity to move specialists off the FFS chassis and increase integration and coordination with broader delivery system reform efforts.
Response: We appreciate the support and agree that TEAM helps to provide opportunities for providers and suppliers to collaborate to improve quality and reduce Medicare spending for beneficiaries receiving specialty-specific care. We may consider including non-surgical (medical) episodes in TEAM in the future. We recognize that TEAM is just one aspect of the CMS Innovation Center's specialty care strategy that aims to test models and innovations that support access to high-quality, integrated specialty care across the patient journey. We refer readers to a recently released RFI in the CY 2025 Physician Fee Schedule Notice of Proposed Rulemaking to gather public feedback on the design of a potential future model to increase the engagement of specialists in value-based care that supports the specialty care strategy. [ 859 ]
Comment: A couple of commenters requested that CMS separate TEAM and future models from the IPPS and other larger payment rules. Commenters specifically requested TEAM be finalized in a separate rule and future models be standalone rules with their own public notice and rulemaking.
Response: We appreciate commenters providing their request for models to be in standalone rules and may consider this request in the future. We note that there may be benefits to proposing models in larger CMS payment rules versus proposing models in standalone rules. For example, because TEAM requires select hospital participation and accountability, we assumed that by proposing and finalizing TEAM in this larger CMS payment rule that is focused on hospital policies, TEAM would capture greater public attention and feedback but also allow for efficient review since we expect most hospitals to already be reading the proposed and final rule. ( print page 69636)
Comment: A commenter suggested that CMS must provide a final rule with comment period or other avenue for public input because CMS has not specified which geographic areas will be subject to the model and it's critical that the affected hospitals have the opportunity to offer geographic and hospital-specific feedback on TEAM prior to the model's start date.
Response: We disagree that a final rule with a comment period, such as an interim final rule with comment period (“IFC”), would be appropriate for purposes of finalizing TEAM, because the proposed rule for TEAM was published May 2, 2024 ( 89 FR 35934 ) and it provided an opportunity to comment on TEAM's policies. We note that we intend to go through rulemaking in the future to address certain policies, which would allow another opportunity for the public, including hospitals that are required to participate in TEAM, to share feedback.
We acknowledge that the list of mandatory CBSA's selected for participation was not included in the proposed rule but is included in section X.A.3.a(4) of the preamble of this final rule. However, we did provide the full list of eligible CBSAs, effectively putting all hospitals located in one of the eligible CBSA's on notice for potential participation, as indicated in section X.A.3.a(4) of the preamble of the proposed rule and of this final rule. Given this notice, hospitals located within an eligible CBSA could have provided their comments, including geographic comments, during the proposed rule comment period. Further, we believe TEAM participants have sufficient time to prepare for the model start date, which is January 1, 2026. We are publishing this final rule, which includes a list of the selected mandatory CBSAs in section X.A.3.a.(4) of the preamble of this final rule, approximately 17 months prior to the beginning of the model start date.
Comment: A commenter had questioned how the outcome of other proposed changes to the IPPS contained in the Proposed Rule could affect stakeholder views on the potential impact of TEAM.
Response: We recognize the breadth of proposals included in the proposed rule ( 89 FR 35934 ) required time to review and to assess the potential impact of TEAM, especially in light of the uncertainty of whether a proposal would be finalized. However, we believe proposing TEAM in the larger IPPS payment rule provides the public with the most comprehensive set of information to gauge how an episode-based payment model could impact them in conjunction with other potential future changes to Medicare payment policy.
Comment: Some commenters suggested we did not provide sufficient information for them to assess either the impact of our proposals or the potential opportunity for improved performance for their facility. Of those commenters, a commenter indicated the scope of TEAM is too broad and implementing TEAM would put their hospitals at greater risk of increased financial and operational challenges.
Response: We strived to notify the public of the model's proposed policies in the most comprehensive manner, while balancing the burden that can be associated with regulatory review. We believe we provided sufficient detail in the proposed rule ( 89 FR 35934 ) to assess the impact of TEAM. Specifically, our proposed rule defined the type of participant; identified the types of episode categories and eligible beneficiaries, corresponding billing codes, and included items and services; established the quality measures and process for assessment; detailed the methodology used to construct target prices and determine reconciliation payment amounts and repayment amounts; identified health equity reporting; described in detail financial arrangements and Medicare payment policy waivers; specified data sharing requirements, outlined beneficiary monitoring; and numerous other policies to help the public, and potential TEAM participants understand and assess the policies being proposed. We have also published evaluations from prior CMS episode-based payment models that informed the development of this model. While we did not provide a proposed list of hospitals that would be required to participate in the model, we did provide a list of potential CBSAs eligible for selection into TEAM, along with table that identified selection strata probabilities.
We disagree that the scope of TEAM is too broad. TEAM was designed from lessons learned from other CMS episode-based payment models that captures a similar scope and balances policies tested in either voluntary models, mandatory models, or both. For example, TEAM is testing far fewer episode categories than the BPCI Advanced model but only four more than the CJR model.
With respect to commenters concerns about participants' financial risk in TEAM, we have addressed those comments in sections X.A.3.a.(3) and X.A.3.d of the preamble of this final rule.
Comment: A couple of commenters had concerns about what CMS would learn from testing TEAM. A commenter indicated that each of the five TEAM episode categories have been previously tested and analyzed with nearly identical parameters in either in BPCI or CJR, with the salient lesson from evaluations showing the predominant way to achieve Medicare savings is to use a discounted spending benchmark (target price) to force hospitals to send patients to less intensive and less costly post-acute care settings. Another commenter requested CMS indicate what insights and lessons will be garnered from TEAM that have not already emerged through the BPCI and CJR models, particularly when these five clinical episodes have already been tested.
Response: We appreciate the commenters concerns and clarifying questions. We believe the evaluation findings from the BPCI, BPCI Advanced, and CJR models support the continued testing of episode-based payment models. Evaluation findings demonstrated that participants in these episode-based payment models generally maintained quality of care and generated savings by reducing post-acute care spending through mechanisms such as reducing institutional post-acute care length of stay. [ 860 ] While TEAM builds upon lessons learned from episode-based payment models by incorporating the most promising model features, we disagree that TEAM has nearly identical parameters as previous or current episode-based payment models that have been tested in a single model. For example, the five episode categories tested in TEAM have been tested in the BPCI Advanced model, but BPCI Advanced was not a mandatory model and participants self-selected into the model and self-selected into certain clinical episode categories or clinical episode service line groups, making evaluation results much less generalizable. Likewise, the CJR model was a mandatory model for hospitals in 34 MSAs, but it only tested a single episode category. Model 2 of the BPCI initiative tested 30-day post-discharge lengths but very few awardees signed up for that post-discharge length because it was associated with a higher discount factor. We believe TEAM represents the right combination of features tested in other episode-based payment models, and that if successful, TEAM could potentially be used to establish the framework for managing episodes as a ( print page 69637) standard practice in Traditional Medicare and could meet criteria to be expanded, as permitted under section 1115A(c) of the Act. Further, TEAM also includes policies and features that have not been tested in other episode-based payment models, including the requirement to refer beneficiaries to primary care services and the novel Decarbonization and Resilience Initiative.
Comment: A commenter indicated the importance of physical therapists in functional care management to promote the connection of Medicare beneficiaries to prescribed community programs that support evidence-based physical activity. A commenter also indicated that for identifying higher risk patient and for whom physical therapist-facilitated early mobilization and ongoing PT care is necessary, a basic frailty screen prior to one of the targeted procedures is recommended.
Response: We agree physical therapists can play a key role in the care management of Medicare beneficiaries. Some TEAM beneficiaries may require physical therapy services after the anchor hospitalization and anchor procedure to improve their physical abilities and functional status. To support collaboration with TEAM participants and drive improved patient outcomes, TEAM allows providers and suppliers of outpatient therapy services, therapists in a private practice, and therapy group practices to be TEAM collaborators in the model. We may also take into consideration the recommendation of including a frailty screen in the future, though we note that in other episode-based models, CMS has generally avoided imposing requirements on participants before the episode is initiated, and ultimately before they are accountable for the cost and quality of the episode. However, TEAM participants are encouraged to implement early screening or intervention for their patients.
Comment: Some commenters requested greater transparency and support mechanisms, such as guidance on data reporting, care coordination strategies, and best practices for managing complex patient populations, in order to help hospitals appropriately participate in TEAM. Another commenter suggested a learning system or other mechanism for CMS to provide technical assistance to model participants and to facilitate peer-to-peer sharing of best practices.
Response: We intend to do as much as we can to provide learning resources and support for TEAM participants. In general, most CMS Innovation Center models include a learning system that helps to disseminate, share, and integrate lessons learned, quality improvement concepts, tactics, and resources so that participants can benefit from their participation experience in the model. We anticipate TEAM will have a learning system that would mimic these same functions, with a goal of engaging TEAM participants prior to the model start date to help them prepare for model implementation. In addition to the learning system, we will continue to make publicly available updated model resources, including the model-specific web page, Frequently Asked Questions (FAQs), and fact sheet, among other resources. We are always looking for and welcome feedback for better ways to educate and assist participants and their partners in care redesign and knowledge sharing.
Comment: Some commenters recommended CMS to continue to proactively engage with potentially-impacted people on the front end so their needs are heard and incorporated before a model is fully developed, specifically incorporating beneficiary and caregiver and provider perspectives into model design. A commenter noted that patient and caregiver engagement must guide the development, implementation, and evaluation of all care models, including TEAM, to ensure that patient perspectives and lived experiences are incorporated into every step of the process. Another commenter indicated that it is critical that CMS directly engage relevant practicing physicians in model development and implementation, including defining appropriate participation parameters, episode triggers, quality measures, and risk adjustments, as well as methods for assessing model success over time.
Response: We agree engagement with interested parties, including beneficiaries, providers, and suppliers, as well as the public at large, is an important aspect of model development. During the design of TEAM, we believed it was important to seek input in a variety of mechanisms in order to capture feedback from a broad scope of individuals, groups, and entities. That is why we released a request for information in the Federal Register ( 88 FR 45872 ), and most importantly proposed TEAM in rulemaking to ensure robust opportunity for public notice and comment on the model and its design ( 89 FR 35934 ). We believe engagement is essential to the success of the models we design and test and will continue to do so throughout TEAM implementation. We look forward to continued feedback from all members of the public about the model.
Comment: A few commenters had concerns about the amount of burden placed on providers to implement the model. A commenter noted TEAM produces complex, burdensome administrative requirements where providers must expend a substantial outlay of time, money, and attention to comply. Another commenter indicated that certain hospitals will experience an increase in costs which will then be passed on to private payors or patients and they do not believe CMS should willingly contribute to the volume of administrative costs that already exist within the healthcare system. Another commenter indicated they would need to dedicate staff to TEAM, taking them away from other tasks when they are already struggling to maintain sufficient staffing for both patient care and back-office functions.
Response: We thank these commenters for sharing their concerns, but we do not believe TEAM will create significant provider burden. TEAM will not alter the way TEAM participants bill Medicare. We believe that there will be no additional burden for TEAM participants related to billing practices, even in cases where CMS waives certain policies for purposes of TEAM (for example, the telehealth waivers discussed in section X.A.3.h of this final rule). We do recognize the time and effort to establish financial arrangements, which may vary based on a TEAM participant's experience and capabilities partnering with entities and setting up the terms and conditions. of such partnerships. However, TEAM participants are not required to engage financial arrangements for the model.
The model evaluation for TEAM will include surveys, site visits, and other modalities to obtain of obtaining evaluation data. The burden for these evaluation efforts will depend on their length, complexity, and frequency, but we note that we will try to minimize the length, complexity, and frequency of model evaluation related tasks.
Lastly, we believe TEAM will not be adding to quality measure reporting or health equity reporting burden because we are using quality measures that TEAM participants will already be reporting for other CMS quality reporting programs and health equity reporting is voluntary.
Comment: A commenter suggested CMS only implement payment models that are designed by physicians or designed in close collaboration with physicians.
Response: We appreciate the commenter's viewpoint and agree that it's important to include physician input. However, we do not agree that ( print page 69638) only models designed by physicians should be tested because it's important to capture input from all parties potentially impacted by a model, including but not limited to, beneficiaries, caregivers, providers, other non-physician clinicians, and the public.
Comment: A commenter expressed concern that TEAM may result in disparities among providers, especially in areas that are economically distressed where social determinants of health and socioeconomic barriers pose significant challenges to implementation. The commenter notes that health systems with more vertically integrated structures, including SNFs, will likely have an advantage in adopting this model.
Response: We thank the commenter for their concerns, however, we do not believe TEAM will result in or increase disparities among hospitals participating in the model. TEAM includes features that acknowledges that certain types of TEAM participants, such as safety net hospitals, as defined in section X.A.3.f of the preamble of this final rule, may need additional support given their financial constraints and the care they provide to underserved populations. One such feature, the different participation tracks, as described in section X.A.3.a.(3) in the preamble of this final rule, allows safety net hospitals to participate in TEAM with reduced financial risk and reward, including the opportunity to participate with no downside risk for a limited time. We also believe that other model features, such as financial arrangements, as discussed in section X.A.3.g of the preamble of this final rule, will help TEAM participants to engage other Medicare providers and suppliers, such as SNFs, to promote improved quality of care and reductions in Medicare spending. Lastly, we note in section X.A.3.a.(1) of the preamble of this final rule that TEAM participants will have approximately 17 months before the model start date to prepare for model participation, allowing them time to plan and structure their care redesign processes for successful participation.
Comment: Some commenters had concerns about TEAM participants controlling discharge decisions for post-acute care. A commenter noted they believe there is an inherent bias against institutional rehabilitation facilities and there are insufficient safeguards for TEAM beneficiaries from discharge decisions that are motivated purely by the financial parameters of TEAM. Another commenter noted that based on their experience with other payment models, they are concerned that the TEAM participants (that is, IPPS hospitals) will choose to collaborate with only some post-acute care providers and will exclude other post-acute care providers from this new model.
Response: We acknowledge the commenters concerns. TEAM participants may not limit access to medically necessary items and services, nor limit the TEAM beneficiary's choice of Medicare providers and suppliers, including post-acute care providers such as long-term care hospitals and inpatient rehabilitation facilities. This means that TEAM beneficiaries are not precluded from seeking care from providers or suppliers who do not participate in TEAM and a TEAM participant is prohibited from limiting beneficiaries to a preferred or recommended providers list that is not compliant with restrictions existing under current statutes and regulations., as discussed in section X.A.3.i of the preamble of this final rule. However, we do expect the model to encourage TEAM participants to better coordinate post-acute care, which may include referring to certain facilities that better meet the needs of the patient and goals of improving patient care while reducing cost. We will monitor beneficiary care, as discussed in section X.A.3.i of the preamble of this final rule, to ensure beneficiary freedom of choice is not compromised. Through monitoring of the model, CMS will aim to ensure steering or other efforts to limit beneficiary access or move beneficiaries out of the model are not occurring. We also note the breadth of monitoring activities, which includes audits, CMS monitoring of utilization and outcomes within the model, and the availability of Quality Improvement Organization (QIOs) and 1-800-MEDICARE for reporting beneficiary concerns, that can help us identify any beneficiary access or freedom of choice concerns in TEAM.
Comment: Several commenters stated that hospitals may not be successful in TEAM due to challenges discharging beneficiaries to post-acute care, citing financial and staffing challenges and the lack of vacancies in the post-acute-care settings. A commenter indicated that they believed TEAM would lead nursing homes to reduce capacity or close outright, including those that are otherwise high performers on quality and safety metrics. Another commenter indicated that across the country, acute-care hospitals are retaining patients long after they can safely be discharged into post-acute care for the simple and unfortunate reason that there are no vacancies in the post-acute-care settings those patients need when they need them. A commenter stated that the success under the model depends largely on reducing post-acute care costs, and the post-acute care sector is in the midst of a well-documented staffing crisis. A commenter requested CMS to allow flexibility based on the availability of post-acute resources because the availability of home health, skilled nursing, and swing bed services can vary widely among communities, regions and states.
Response: We do not expect the TEAM will result in adverse results such as post-acute care closures, decrease in availability of services, or disruption of patient care. In contrast, CMS believes that TEAM may have the opposite effects. The financial incentives in the model are designed to incentivize innovative care delivery methods that focus on improving care and reducing Medicare spending. We believe TEAM will spur partnerships between TEAM participants and post-acute care providers, such as skilled nursing facilities and home health agencies, to share financial risk and collaborate on care redesign strategies. We recognize that partnerships with post-acute care providers could be a crucial driver of episode spending and quality, given that many beneficiaries in TEAM may receive post-acute care services after discharge from the hospital. We believe the opportunities to find savings in post-acute care could be a motivator for these partnerships to help address some of the challenges with vacancies and capacities. Evaluation evidence from testing other episode-based payment models indicates participants tend to find efficiencies in the post-acute care space such as reducing the length of stay in institutional post-acute care. [ 861 862 ] Reductions in the length of stay may free up institutional post-acute care beds, thereby allowing beneficiaries to not remain in the acute care setting unnecessarily. We also believe that model incentives could be a catalyst to financially support additional staffing needs through the sharing of reconciliation payment amounts established by financial arrangements between the TEAM participant and post-acute care provider. We emphasize the importance of beneficiary quality and access to care in TEAM and we will monitor the impact of the model closely, as described in section X.A.3.i of this final rule. In the event that adverse ( print page 69639) outcomes such as these arise, CMS may modify or terminate the model accordingly.
We also acknowledge that post-acute care can vary across different communities, regions, and states and will take into consideration policies, waivers, or pricing methodology adjustments that may address these variances. Any changes resulting from this consideration would be proposed in future notice and comment rulemaking.
Comment: A commenter indicated CMS should review research surrounding previous episode-based payment models that when nursing home care goes down, home health use goes up. They cite this as a positive result, but findings also show that at the end of the patient's home health episode that patients needed more help from their caregivers than they did before the bundled payment was used which may increase patient and provider burden or stress health care resources in communities.
Response: We appreciate the commenter highlighting this research. When designing CMS Innovation Center models, including the design of TEAM, extensive investigation is performed using data from our independent evaluations as well as reviewing external research data. Our model evaluations generally assess the impact of the model on the beneficiary, including obtaining data directly from beneficiaries through beneficiary surveys. We intend for the evaluation of TEAM to continue looking into beneficiary impacts, such as beneficiary care experience and functional status, and may also capture caregiver experience and burden as well. We will also monitor beneficiary quality of care, as discussed in section X.A.3.i in the preamble of this final rule and may modify or terminate the model if we identify adverse outcomes.
Comment: A commenter suggested TEAM consider including a longitudinal feature that extends the episode well before surgery and accounts for savings due to avoided procedures which would better address the physical, psychological, and economic burden of back pain.
Response: We appreciate the commenters suggestion and agree that beneficiaries can benefit from care management before surgical intervention. We believe it's important for episode-based payment models to have clear episode time periods and triggers and extending the episode to start before the anchor hospitalization or anchor procedure can make defining the episode challenging. Further, starting the episode before the anchor hospitalization or anchor procedure can make it difficult to avoid including unrelated items and is more likely to encompass costs that vary widely among beneficiaries, which would make the episode more difficult to price appropriately. However, we believe TEAM is complementary to existing longitudinal, population-based models, such as ACO models and initiatives, that can manage beneficiaries before and after an episode and potentially reduce avoidable procedures that would lead to an episode of care.
Comment: A commenter observed across other episode-based payment programs, improvements and savings take time and investment to realize and recommended that CMS grant hospitals a fair opportunity to achieve enough savings to garner a reconciliation payment.
Response: We appreciate the comment observing that improvements and savings take time in episode-based payment programs. CMS believes that offering multiple participation tracks, as discussed in section X.A.3.a.(3) of the preamble of the final rule, with varying levels of risk helps to alleviate the time and investment associated with participation.
Comment: A commenter requested clarification on whether hospitals participating in TEAM will also still participate in the Medicare Promoting Interoperability Program.
Response: TEAM participants eligible for the Medicare Promoting Interoperability Program must comply with all requirements of the program to avoid being potentially subject to a downward payment adjustment. We did not include any proposals nor are we finalizing any policies that would exclude a TEAM participant from participating in the Medicare Interoperability Program.
Comment: A commenter is concerned with the incentives that episode-based payment models may create when focused on procedures, rather than better managing patients' underlying conditions.
Response: We thank the commenter for their feedback. While TEAM will focus on testing five surgical episode categories, we believe model incentives and goals will help TEAM participants to manage beneficiaries underlying conditions and comorbidities. Specifically, we are requiring TEAM participants to refer TEAM beneficiaries to primary care services, as described in section X.A.3.l in the preamble of this final rule, prior to discharge from the anchor hospitalization or anchor procedure. We believe this requirement will promote collaboration between the TEAM participant and primary care providers, so that the TEAM beneficiary is being managed by a care team that includes clinicians who can manage the surgical procedure and clinicians who can manage underlying, chronic conditions. It is important to note that eligible Medicare beneficiaries may be attributed to both TEAM and population-based models, such as ACOs, which we hope will promote collaboration between providers who generally manage acute, specialty care, such as TEAM participants, and providers who generally manage primary care, such as those participating in an ACO, to work together to ensure of the TEAM beneficiary's needs are met.
Comment: A commenter voiced their belief that episode-based models with complex quality and outcomes requirements are an exercise in diminishing return and their experience resulted in operational and financial challenges created by the payment and regulatory policies of these programs year after year.
Response: While we purposely tried to minimize complexity when designing TEAM, we recognize that some hospitals participating in TEAM will be new to episode-based payment models, and other hospitals participating may have experience, but that experience does not guarantee successful participation or understanding of the pricing or quality aspects of the model. Since understanding, operationalizing, and gaining experience in an episode-based payment model takes time, we are not starting the model until January 1, 2026. We are also finalizing a participation track that provides a glide path to full financial risk, as indicated in section X.A.3.a.(3) of this preamble of the final rule, that allows all TEAM participants to participate in TEAM without downside financial risk, and TEAM participants that are safety net hospitals, as defined in section X.A.3.f of the preamble of this final rule, additional time without downside financial risk. This will allow TEAM participants the time and experience to operationalize their care redesign interventions without the financial pressures of owing a repayment amount to CMS for the first performance year, and safety net hospitals up to the third performance year. Also, prior to model implementation and during model implementation, CMS will be providing TEAM participants with support, by sharing learning resources and holding webinars to help TEAM participants understand complex topics, such as the target price methodology. ( print page 69640)
We proposed a 5-year “model performance period”, defined as the 60-month period from January 1, 2026, to December 31, 2030, during which TEAM is being tested and the TEAM participants are held accountable for Medicare spending and quality of care. We proposed that the model would have 5 “performance years” (PYs). We proposed to define a PY as a 12-month period beginning on January 1 and ending on December 31 of each year during the model performance period in which TEAM is being tested and TEAM participants are held accountable for spending and quality. We proposed to define the start of the model performance period as the “model start date”.
We proposed a 5-year model performance period to allow for a sufficient time period for TEAM Participants to invest in care delivery transformation and observe return on investments. We stated that a five-year period would also allow for an adequate evaluation period to determine model results, given that many of the episode categories we proposed to test under TEAM have thus far only been tested among voluntary model participants ( 89 FR 36387 ).
We alternatively considered a 3- or 10-year model performance period. However, we believe a 3-year period to be too short to allow adequate time to invest in transformations and achieve considerable model savings to the Medicare trust fund. We also considered a 10-year model performance period, similar to several recently announced CMS Innovation Center models; however, given this would be a mandatory model, we believe 5 years would be sufficient to gather the necessary data to evaluate whether the model is successful for the included episode categories.
We also considered beginning TEAM on April 1, 2026, July 1, 2026, or October 1, 2026, to allow selected TEAM participants more time to prepare for model implementation. However, based on our experience with prior and current episode-based payment models, we believe that potential participants would have sufficient time to prepare to participate in a model that begins January 1, 2026, which is why we proposed TEAM at least 18 months before the proposed model start date. In addition, given that the current BPCI Advanced model concludes on December 31, 2025, beginning TEAM on January 1, 2026, would ensure continuity between models for those hospitals in BPCI Advanced that are in the mandatory CBSAs selected to participate in TEAM. We also recognize the potential misalignment between the performance measurement period based on the calendar year and an alternative model start date, so if we were to adjust the model start date based on public input, we proposed that we would also alter the model performance period. For example, if TEAM were to begin April 1, 2026, the PY would still be defined as a 12-month period from the start date, meaning April 1, 2026, to March 31, 2027. As a result, the model performance period end date would also shift to reflect a 60-month period from the model start date of the first PY—for example, April 1, 2026, to March 31, 2031.
We sought comment on the proposed model performance period of 5 years and proposed model start date of January 1, 2026, for PY 1, and on the alternatively considered start dates (April 1, 2026, July 1, 2026, and October 1, 2026), and the subsequent adjustment to dates of the model performance period if we were to change the model start date.
The following is a summary of comments we received on the proposed model performance period and model start date and our responses to these comments:
Comment: A commenter supported a calendar year start date because of the alignment in timing with other Medicare programmatic requirements. A commenter also supported a model length of five years, indicating that as an appropriate length of time to be able to evaluate a model to determine success.
Response: We thank the commenter for the support and agree that a calendar year model start date would align with other Medicare requirements or initiatives. We also agree a five-year model test should provide sufficient evidence to determine if TEAM is achieving its goals of improving quality of care and reducing Medicare expenditures.
Comment: Many commenters requested CMS delay the model start date for TEAM. Some commenters indicated that the volume of relationships, processes, and contracts that TEAM participants would need to establish would be substantial given the large scope of the proposed model. Some commenters had concerns about the impact that they thought TEAM may have on the care that hospitals provide, and, ultimately, the quality of care provided to Medicare beneficiaries. A commenter noted their belief that January 2026 was ambitious to start the model and recommended that CMS undertake a phased implementation of the model. A few commenters urged CMS to delay the model until such time that more episode specific quality measures can be established and to engage subject matter experts in this effort before making this model mandatory. Another commenter indicated that they believed it would be difficult for hospitals to participate without subjecting themselves and their communities to significant financial risk and recommended that CMS delay implementation until these issues can be resolved. A commenter recommended delaying the model and including a dedicated payment bump for administrative costs that would allow hospitals to prepare for additional costs.
Response: We appreciate comments expressing concerns around the timing of this model. We believe that it is important to initiate TEAM after the conclusion of the CJR and BPCI Advanced models to continue testing the care transformation effects that episode-based payment models have on the health care system. Testing TEAM well after the BPCI Advanced ends may slow the speed and adoption of broad care transformation practices and may lead to a loss of the efficiencies that were achieved during the testing of the BPCI Advanced and CJR models. We believe it's important for TEAM to have a model start date of January 1, 2026, as this start date will provide essential information to CMS and others about the potential for a new episode-based payment model to improve care and lower spending and to continue the momentum of testing episodes and associated care transformation.
We are sensitive to commenters' concerns about the level of preparation needed to implement care redesign activities, develop relationships and processes, especially for hospitals new to episode-based payment models. A key reason we began soliciting information for the design of TEAM last year through the Episode-Based Payment Model Request for Information ( 88 FR 45872 ) was to signal our desire to test a mandatory episode-based payment model and to put the public on early notice that a model might be developed in the near future. We purposely proposed TEAM with at least 18 months before the proposed model start date to give potential TEAM participants time to consider preparations if their CBSA was selected ( print page 69641) as one of the required areas in this model.
We disagree that TEAM's scope is too large for future TEAM participants. We note that TEAM will only include a limited number of episode categories, and those episodes include higher volume procedures where hospitals may already have established care protocols. We believe it is reasonable for TEAM participants to begin to analyze data and identify care patterns and opportunities for care redesign for these episode categories prior to assuming accountability for quality and spending outcomes in order to prepare for model implementation. Prior to each performance year, and thus prior to the model start date, we intend to offer TEAM participants the opportunity to request baseline period data, as indicated in section X.A.3.k of the preamble of this final rule. CMS would share such data with TEAM participants in accordance with the TEAM data sharing agreement. This data will assist TEAM participants to prepare for model implementation by helping to evaluate their potential performance, conduct quality assessment and improvement activities, conduct population-based activities relating to improving health or reducing health care costs, or conduct other health care operations.
We also disagree that TEAM will create significant financial risk for TEAM participants, and that the financial risk in the model would warrant either delaying the model or providing TEAM participants with an additional payment to account for administrative costs. As discussed in section X.A.3.a.(3) of the preamble of this final rule, we believe that allowing all TEAM participants the opportunity to participate in Track 1 for the first performance year will provide additional preparation time before being subject to downside financial risk. We are also finalizing changes to other TEAM policies in an effort to minimize financial risk for TEAM participants, including reducing the discount factor, reducing the stop-gain and stop-loss limits for Track 2, and allowing safety net hospitals the opportunity to remain in Track 1 for the first three performance years, as discussed in sections X.A.3.d.(3)(g), X.A.3.d.(5)(h), and X.A.3.a.(3) of the preamble of this final rule. However, it's important to note that TEAM participants who feel prepared and want to assume two-sided financial risk, both upside and downside risk, from the model start date may do so by participating Track 3, as described in section X.A.3.a.(3) of the preamble of this final rule.
Likewise, we do not agree that the models should be implemented after episode-specific quality measures are established. We recognize the value in having episode-specific quality measures but for purposes of TEAM, we chose measures that hospitals are already reporting under existing CMS quality reporting programs in an effort to minimize TEAM participant burden. Previous episode-based payment models, including the BPCI Advanced model, have used some similar hospital level quality measures to assess participant quality performance and therefore we believe this approach is consistent with other models. We have engaged interested parties on quality measure selections, and we took the public comments received during the Episode-Based Payment Model Request for Information ( 88 FR 45872 ) and the proposed rule ( 89 FR 35934 ) into consideration, and we will continue engagement throughout the implementation of TEAM. However, as noted in section X.A.3.c of the preamble of this final rule, we are interested in considering episode-specific quality measures and if we identify appropriate measures, we may propose them in future notice and comment rulemaking.
Lastly, we do not believe the model start date of January 1, 2026, will compromise beneficiary access or reduce quality of care. As discussed in section X.A.3.c of the preamble of this final rule, we are including quality measures for purposes of evaluating participating hospitals' performance both individually and in aggregate across the model. Also, as discussed in section X.A.3.i of the preamble of this final rule, we are finalizing policies and actions to monitor both care access and quality. We believe these features will help ensure that beneficiary access to high quality care is not compromised under the model.
Comment: Some commenters suggested that CMS provide at least 18 months from when the list of selected mandatory CBSAs is finalized to when the model starts to provide ample implementation time, especially for potential participants with a lack of experience in episodic models or accountable care. A commenter suggested CMS should accommodate participants' annual budgeting cycles by providing at least an 18 months' lead time. A commenter noted that migrating the volume of procedures to mandatory bundles across multiple service lines in such a rapid timeframe would be untenable. Another commenter suggested that 2026 be an optional year because the short implementation timeline would impose a great burden on hospitals to set up the appropriate infrastructure to support a complex model such as TEAM by the proposed 2026 model start date.
Response: We appreciate comments expressing concerns around providing sufficient notice between when the final listed of mandatory CBSAs are publicly shared and when the model starts. We identified the selected mandatory CBSAs for participation in TEAM, as noted in section X.A.3.a.(4) of the preamble of this final rule, and keeping a January 1, 2026, model start date provides approximately 17 months of time to prepare. We believe 17 months is sufficient time for participants to implement the kinds of changes needed to successfully participate in the model.
Further, we don't believe making 2026 an optional year is necessary because we are providing the opportunity for all TEAM participants to participate in Track 1, as discussed in section X.A.3.a.(3) of the preamble of this final rule, which allows them to participate with no downside financial risk for PY 1, effectively increasing the preparation time and experience to operationalize their care redesign without the financial pressure of downside risk for one year before owing a repayment amount. Additionally, for TEAM participants who meet our definition of safety net hospital, as defined in section X.A.3.f.(2) of the preamble of this final rule, we are extending their ability to remain in Track 1 for the first three performance years, allowing a longer on-ramp to downside risk.
We disagree with the commenter that the timeframe to implement TEAM is untenable given the volume of procedures spanning multiple service lines. The episode categories that will be tested in TEAM are higher volume procedures that we anticipate most TEAM participants can leverage their existing standard care pathways to find efficiencies. Additionally, TEAM does not require a TEAM participant to change how they perform these procedures, thus there is no bearing to the timeframe before these procedures are mandatorily tested in TEAM.
As noted previously, we expect that hospitals will spend the first performance year of the model analyzing data, identifying care pathways, forming clinical and financial relationships with other providers and suppliers, and assessing opportunities for savings under the model. Therefore, we do not believe that CMS needs to change the model start date or make other changes related to the timing of the model. ( print page 69642)
Comment: A commenter requested that CMS delay the model start date because there are many unknown implications of minimum staffing standards for long-term care (LTC) facilities.
Response: We do not agree that the model should be delayed because of the minimum staffing standards for long-term care facilities ( 89 FR 40876 ). Staffing in LTC facilities has remained a persistent concern and the minimum staffing standards for LTC facilities final rule represents a critical step in addressing adequate staffing and reducing the risk of residents receiving unsafe and low-quality care in LTC facilities. As such, we believe that the ongoing efforts of LTC facilities to comply with the minimum staffing standards may create improvements in the quality of care for residents that may support the goals of TEAM. We also do not believe that the minimum staffing standards for long-term care facilities, in particular, will impede TEAM participants' abilities to participate in TEAM successfully.
After consideration of the public comments we received, we are finalizing our proposed definitions for model performance period, performance year, and model start date at § 512.505 without modifications.
We indicated in the proposed rule that TEAM builds upon previous CMS Innovation Center episode-based payment models, including the BPCI Advanced and CJR models. While these models have similarities, they have some notable differences with regard to participant structure and the entity who can initiate episodes. The BPCI Advanced model is a voluntary model that includes convener and non-convener participants. A non-convener participant initiates episodes, is either an acute care hospital or physician group practice (PGP) and bears financial risk for itself. A convener participant is an entity willing to bear financial risk for downstream episode initiators, either acute care hospitals or PGPs, and generally provides supportive services such as data analytics or clinical care navigators. In contrast, the CJR model is a mandatory model in 34 MSAs that does not include convener participants or allow PGPs to initiate episodes but does parallel BPCI Advanced by including participant hospitals (non-convener) that initiate episodes. While the CJR Model does not have a formal convener role, some CJR participant hospitals contract with (non-model participant) convener-organizations to provide administrative, operational, analytical, and clinical services.
In the proposed rule we stated that we were interested in testing and evaluating the impact of a mandatory episode-based payment model in selected geographic areas, see section X.A.3.a.(4) of the preamble of this final rule, for acute care hospitals that initiate certain episode categories, including among those hospitals that have not chosen to voluntarily participate in the BPCI Advanced model or those that were selected to participate in the CJR model. We stated that testing the model among acute care hospitals in select geographic areas would allow CMS and participants to gain experience testing and evaluating an episode-based payment approach for certain episodes furnished by hospitals with a variety of historic utilization patterns; roles within their local markets, including with regard to accountable care organization participation or affiliation; volume of services provided; access to financial, community, or other resources; and population and health care provider density. Further, Medicare beneficiaries and providers in rural and underserved areas can be underrepresented in voluntary models, whereas under a mandatory model we may include these entities, with safeguards as appropriate, for participation so that beneficiaries have equitable access to care redesign approaches intended to improve the quality care, and such providers gain experience in value-based care. Lastly, we noted that participation of hospitals in selected geographic areas would allow CMS to test episode-based payments without introducing participant attrition or selection bias such as the selection bias inherent in the BPCI Advanced model due to self-selected participation in the model and self-selection of episode categories ( 89 FR36388 ).
We noted in the proposed rule that the CJR model has participant hospitals who are acute care hospitals that initiate episodes whereas the BPCI Advanced model allows either acute care hospitals or PGPs to initiate episodes, who may either be a participant or a downstream episode initiator in the model. Since two different types of entities are permitted to initiate episodes in BPCI Advanced and they may be co-located, meaning the PGP may initiate episodes and practices at a hospital that also initiates episodes, the BPCI Advanced model includes precedence rules. Precedence rules dictate which entity will be attributed the episode and will be held accountable for quality and cost performance, but they also contribute to operational complexity. For example, in BPCI Advanced a single episode could be attributed to one of three potential provider or suppliers: the attending PGP, the operating PGP, or the hospital. Data feeds can help inform entities of episode attribution when multiple provider or suppliers have interacted with the beneficiary, but BPCI Advanced participants have expressed challenges with identifying their potential episodes due to lack of real-time data.
Given the challenges of having multiple providers or suppliers in a single model initiate an episode, we stated in the proposed rule that we believed it would benefit TEAM to only allow a single entity to initiate episodes and be the participant in TEAM ( 89 FR 36388 ). We stated in the proposed rule that this is because it would simplify episode attribution, meaning it would avoid precedence rules, and make it easier for the single entity to identify beneficiaries that may be included in the model. Therefore, similar to the CJR model, we proposed that acute care hospitals would be the TEAM participant and the only entity able to initiate an episode in TEAM. Specifically, we proposed defining a TEAM participant as an acute care hospital that initiates episodes and is paid under the IPPS with a CMS Certification Number (CCN) primary address located in one of the geographic areas selected for participation in TEAM, as described in section X.A.3.a.(4) of the preamble of this final rule. We are also proposing that the term “hospital” has the same meaning as hospital as defined in section 1886(d)(1)(B) of the Act. This statutory definition of hospital includes only acute care hospitals paid under the IPPS.
We believe that hospitals are more likely than other providers or suppliers to have an adequate volume of episodes to justify an investment in episode management. We also believe that hospitals, compared to other providers or suppliers, are most likely to have access to resources that would allow them to appropriately manage and coordinate care throughout these episodes. Further, the hospital staff is already involved in discharge planning and placement recommendations for Medicare beneficiaries, and more efficient PAC service delivery provides substantial opportunities for improving quality and reducing costs in TEAM.
In the proposed rule, we also noted that we believed hospitals being TEAM participants aligns with how episodes ( print page 69643) are initiated in TEAM, as described in section X.A.3.b.(5)(c) of the preamble of this final rule, since it relies on a beneficiary's inpatient admission to a hospital or a beneficiary receiving a procedure in a hospital outpatient department. Additionally, we believe that utilizing the hospital as the TEAM participant is a straightforward approach for this model because the hospital furnishes the acute surgical procedure and plans for and manages post-discharge (or post-procedure) care. We also want to test a broad model in a variety of hospitals, including safety net hospitals specified in section X.A.3.f.(2) and rural hospitals specified in section X.A.3.f.(3) of the preamble of this final rule, under TEAM to examine results from a more generalized payment model. Finally, as described in the following sections that present our finalized approach to geographic area selection, our geographic area selection approach relies upon our definition of hospitals as the TEAM participant and the entity that initiates episodes.
We sought comment on our proposal at § 512.505 to define TEAM participants as an acute care hospital that initiates episodes and paid under the IPPS with a CMS CCN primary address located in one of the geographic areas selected for participation in TEAM. We also sought comment on our proposal at § 512.505 to define hospital as defined in section 1886(d)(1)(B) of the Act.
We stated in the proposed rule that all acute care hospitals in Maryland would be excluded from being TEAM participants because Maryland hospitals are not currently paid under the IPPS and OPPS. Therefore, any acute care hospital located in Maryland would not be able to satisfy the definition of TEAM participant. Currently, CMS and the state of Maryland are testing the Maryland Total Cost of Care (TCOC) Model, which sets a per capita limit on Medicare total cost of care in Maryland. The TCOC Model holds the state fully at risk for the total cost of care for Medicare beneficiaries. Maryland acute care hospitals are not paid under the IPPS or OPPS, but rather are paid using a global budget methodology that establishes pricing of medical services provided by hospitals, primary care doctors, and specialists across all payers. Therefore, we proposed that payments to Maryland acute care hospitals would be excluded in the pricing calculations as described in section X.A.3.d. of the preamble of this final rule. We sought comment on this proposal and whether there were potential approaches for including Maryland acute care hospitals as TEAM participants. In addition, we sought comment on whether Maryland hospitals should be TEAM participants in the future ( 89 FR 36389 ).
In the proposed rule we also stated we recognize that the Maryland TCOC Model may not be the only CMS model or initiative that may use hospital global budgets as part of their alternative payment models. The States Advancing All-Payer Health Equity Approaches and Development (AHEAD) Model is a state-based voluntary TCOC model that will incorporate hospital global budgets. We indicated there are several cohorts in which states may participate, and we expect that the AHEAD Model implementation period would overlap with the performance years of TEAM. Given that CMS envisions that up to eight states would participate in the AHEAD Model, unlike the Maryland TCOC Model, we said in the proposed rule that we were hesitant to propose excluding hospitals that participate in the AHEAD Model from being TEAM participants because it could reduce the volume of beneficiaries that may benefit from episodic, acute coordinated care. We said that we were aware that allowing overlap may introduce model complexities with respect to constructing TEAM prices or the AHEAD global budgets and statewide total cost of care calculations. However, there may be other opportunities, such as sharing of TEAM-style summary episode data (not beneficiary-identifiable) with AHEAD hospitals, to support episodes without allowing hospitals participating in the AHEAD Model to participate in TEAM as TEAM participants. Thus, we stated that we were unsure if we should allow AHEAD hospitals located in areas selected for TEAM participation to participate in TEAM as TEAM participants. We sought comment on whether there may be potential approaches for including hospitals participating in the AHEAD Model in TEAM as TEAM participants, or other approaches that may not result in participation in both models but support the integration of episodes and hospital global budgets. We indicated in the proposed rule, that the AHEAD Model would be voluntary for participating states and hospitals within those states, and as such, we also sought comment on whether hospitals located in AHEAD states should be required to participate in TEAM as TEAM participants if they either do not participate in in the AHEAD Model or if they terminate their participation in the AHEAD Model (or CMS terminates them) before the AHEAD Model ends.
We stated in the proposed rule that since TEAM is built from lessons learned from previous episode-based payment models, including the BPCI Advanced model, we considered including PGPs in the definition of TEAM participant in the future. We recognized that PGPs demonstrated some successes in the BPCI Advanced model, most specifically that BPCI Advanced PGPs reduced average episode payments by $2,157 for surgical episodes in Model Year 3 (2020) and reduced unplanned hospital readmissions for surgical episodes in Model Years 12 (October 2018-December 2019). [ 863 864 ] We indicated in the proposed rule that despite these favorable findings, we have concerns about requiring PGPs, who are generally smaller entities and care for a lower volume of Medicare beneficiaries, to participate in an Advanced APM such as TEAM given the more than nominal financial risk standard required of Advanced APMs set forth in regulation in the Quality Payment Program ( 42 CFR 414.1415 ). We noted that while BPCI Advanced is an Advanced APM, participation is voluntary, and PGPs have the autonomy to determine if they have the infrastructure and resources to take on the level of financial risk to participate in the model and determine if they have sufficient episode volume to create systematic care redesign efficiencies. Further, we are aware from internal reports that most eligible clinicians in the BPCI Advanced model do not meet Qualifying APM Participant determinations in the model due to not meeting the required thresholds for Medicare Part B payments or Medicare beneficiaries, suggesting that acute care-based episodes may not sufficiently capture the full panel of patients a PGP manages. We stated in the proposed rule that we believe there are other meaningful opportunities for PGPs to engage in TEAM, specifically through financial arrangements with TEAM participants, or through other CMS value-based care initiatives, including future PGP-specific opportunities under development through the CMS Innovation Center specialty care ( print page 69644) strategy. For these reasons, we did not propose PGPs to be included in the definition of TEAM participant in TEAM. However, we sought comment on whether we should include PGPs in the definition of TEAM participant through future rulemaking, or if there are other ways, beyond financial arrangements, that we could incorporate PGPs to promote collaboration between TEAM participants and other providers who may care for a TEAM beneficiary over the course of the episode.
We sought comment on our proposal to exclude hospitals located in Maryland from TEAM participation, and how to address hospitals that would participate in the AHEAD model. We also sought comment on including PGPs in the definition of TEAM participant.
The following is a summary of comments we received on the proposed TEAM participant definition, Maryland hospital exclusion, and AHEAD hospital overlap and our responses to these comments:
Comment: We had a few commenters support the TEAM participant definition to only include hospitals. A commenter agreed that the definition is relatively unambiguous and is consistent with a larger goal of making the “initiating hospital” of a surgical episode responsible for a defined set of downstream costs and patient outcomes.
Comment: A couple of commenters requested that CMS include critical access hospitals (CAHs) in the TEAM participant definition. A commenter had concerns with the proposed TEAM participant definition, which only included IPPS hospitals, and stated that this definition would have unintended consequences that will detrimentally impact CAHs. Specifically, the commenter thought that TEAM may result in more surgeries being referred to urban partner facilities instead of CAHs in order to “meet the target price,” and more importantly that TEAM would result in a patient having to travel a much further distance to obtain care.
Response: We believe it would be challenging to include CAHs in the TEAM participant definition since CAHs are not paid under the IPPS or OPPS, but rather they are paid for most inpatient and outpatient services to patients at 101 percent of reasonable costs. [ 865 ] Given these and other differences between CAHs and IPPS hospitals, it would be difficult to construct a reasonable target price for CAHs using TEAM's current pricing methodology.
With respect to impacting patient care, we will monitor beneficiary care, as discussed in section X.A.3.i of the preamble of this final rule, to ensure beneficiary freedom of choice is not compromised. Medicare beneficiaries are not precluded from seeking care from providers or suppliers who do not participate in TEAM and a TEAM participant is prohibited from limiting beneficiaries to a preferred or recommended providers list. CMS's monitoring efforts will aim to ensure steering or other efforts to limit beneficiary access or move beneficiaries out of the model are not occurring.
Comment: Many commenters requested that CMS include physicians or PGPs as TEAM participants, so that physicians or PGPs could also initiate episodes and assume financial responsibility for episodes. Some commenters recognized that PGPs may be unprepared for mandatory participation but indicated more must be done to recognize and favor the physician's role in the model as the individual responsible for clinical care. A commenter noted that allowing hospitals to form partnerships or joint ventures with PGPs would facilitate shared responsibilities and rewards, promoting a holistic and patient-centered care experience. Another commenter noted that by not directly including PGPs as participants, CMS places an additional burden on hospitals to organize financial and legal arrangements. Another commenter believed that excluding PGPs from being a TEAM participant gives more power to health care facilities and could further drive consolidation in health care providers. A commenter wanted to know what considerations CMS had for risk bearing entities other than the hospital, such as hospital-PGPs or clinically integrated networks because these types of providers are amenable to serving as risk-bearing entities and are highly-focused, team-based, and able to construct an episode-directed quality program and track episode costs-regardless of site of care. A commenter thought that allowing the clinical team to participate in TEAM as a risk bearing entity could better align incentives around the patient because the team has responsibility for the care journey, including time in the hospital and time to discharge.
Response: We believe it is most appropriate to identify a single type of provider or supplier to bear financial responsibility for making repayment to CMS under TEAM as one entity needs to be ultimately responsible for ensuring that care for TEAM beneficiaries is appropriately furnished and coordinated to avoid fragmented approaches that are often less effective and more costly. Hospitals play a central role in coordinating episode-related care and ensuring smooth transitions for beneficiaries from the hospital inpatient and hospital outpatient department. Most hospitals already have some infrastructure in place related to patient and family education and health information technology to coordinate care across different providers and settings. In addition, hospitals are required by the hospital Conditions of Participation (CoPs) to have in effect a standard discharge planning process (§ 482.43 (a)) that includes requirements related to post-acute care services (PAC) (§ 482.43(c)), which includes coordinating with PAC providers, a function usually performed by hospital discharge planners or case managers. Thus, hospitals can build upon already established infrastructure, practices, and procedures to achieve efficiencies under this episode-based payment model.
Many hospitals also have recently heightened their focus on aligning their efforts with those of community providers to provide an improved continuum of care due to the incentives under other CMS models and programs, including ACO initiatives such as the Medicare Shared Savings Program, and the Hospital Readmissions Reduction Program (HRRP), establishing a base for augmenting these efforts under TEAM. Hospitals are also more likely than other providers and suppliers to have an adequate number of episode cases to justify an investment in episode management for this model, have access to resources that would allow them to appropriately manage and coordinate care throughout the episode, and hospital staff is already involved in discharge planning and placement recommendations for Medicare beneficiaries, and more efficient PAC service delivery provides substantial opportunities for improving quality and reducing costs under TEAM.
We considered whether to make physicians or their associated PGPs, if applicable, financially responsible for the episode under TEAM ( 89 FR 36391 ). However, standardizing episodes and determining the appropriate level of financial risk if physicians or PGPs initiated episodes and were responsible for them would be challenging because the services of providers and suppliers other than the hospital where the hospitalization or hospital outpatient procedure occurs would not necessarily be furnished in every episode in TEAM. ( print page 69645) For example, physicians of different specialties play varying roles in managing patients during an acute care hospitalization for a surgical procedure and during the recovery period, depending on the hospital and community practice patterns and the clinical condition of the beneficiary and therefore, we do not think that every episode in TEAM could account for all specialties and all roles. This variability would make requiring a particular physician or PGP to be financially responsible for a given episode in TEAM very challenging. If we were to assign financial responsibility to the operating physician, it is likely that there would be significant variation in the number of relevant episodes that could be assigned to an individual person, potentially resulting in significant financial risk for a given physician Assigning financial responsibility to a PGP may help to mitigate individual physician risk, but even at the PGP level there still may be significant risk depending on the volume of physicians within a PGP and the volume of episodes the PGP may initiate. We acknowledge that providers and suppliers with low volumes of cases may not find it in their financial interests to make systematic care redesigns or engage in an active way with TEAM. We expect that physicians typically do not have the case volume to justify an investment in the infrastructure needed to adequately provide the care coordination services required under TEAM (such as dedicated support staff for case management), which leads us to believe that as a result, the physician, PGP, and model would have more challenges if physicians and PGPs were TEAM participants.
Although the BPCI Advanced model allows a PGP to have financial accountability for clinical episodes, the PGPs electing to participate in BPCI Advanced have done so because their business structure supports care redesign and other infrastructure necessary to bear financial responsibility for episodes and is not necessarily representative of the typical group practice. The incentive to invest in the infrastructure necessary to accept financial responsibility for the entire episode, starting at the anchor hospitalization or anchor procedure and ending 30 days after the date of discharge from the hospital, would not be present across all PGPs. Thus, we do not believe it would be appropriate to designate physicians or PGPs to bear the financial responsibility for making repayment amounts to CMS under TEAM. Further, with respect to commenters who were interested in CMS providing more opportunities for physicians and PGPs to assume risk, we note that those comments seemed to focus on voluntary participation in models rather than including and mandating physicians or PGPs to be TEAM participants.
We would emphasize that physicians, PGPs, and other providers or suppliers are encouraged to collaborate with TEAM participants and take advantage of establishing financial arrangements that would align financial incentives to improve quality of care and reduce Medicare spending through improved beneficiary care transitions and reduced fragmentation. While such PGPs would not be accountable directly to CMS, their arrangements with TEAM participants could include financial risk and accountability to the TEAM participant. We disagree with the comment that there would be additional burden on hospitals to organize financial and legal arrangements since CMS is not requiring TEAM participants to have financial arrangements nor is CMS requiring TEAM participants to change other types of arrangements as a result of participation in the model. We also disagree that excluding PGPs from TEAM could further drive consolidation in the future because while PGPs may not be TEAM participants, they are still given the opportunity to engage in the model, such as being TEAM collaborators, that allows them to be in financial arrangements with TEAM participants to share reconciliation payment amounts and repayment amounts.
We recognize the important role of physicians and non-physician practitioners in caring for Medicare beneficiaries and are committed to testing models that may be more appropriate for these types of Medicare suppliers, or the practices they work with, to assume risk. We are actively developing other opportunities for physicians and non-physician practitioners to be included in value-based care and alternative payment models and refer to the recently published RFI “Building Upon the Merit-based Incentive Payment System (MIPS) Value Pathways (MVPs) Framework to Improve Ambulatory Specialty Care,” in the CY 2025 Physician Fee Schedule Notice of Proposed Rulemaking that seeks feedback on the design of a potential model to increase the engagement of specialists in value-based care. [ 866 ]
Comment: A commenter requested that physicians and non-physician organizations, with requisite qualifications, should be permitted to participate in any CMS Innovation Center model as conveners.
Response: As described in section X.A.3.a.(2) of the preamble of this final rule, TEAM participants may enter into administrative or risk sharing arrangements related to TEAM with entities that may provide similar support as a convener, except to the extent that such arrangements are restricted or prohibited by existing law.
Comment: A commenter recommended that CMS require that hospitals include anesthesiologists and their contracted anesthesia services in their participation lists.
Response: CMS will collect from TEAM participants a financial arrangements list and a clinician engagement list, as applicable, and as finalized in section X.A.3.m of the preamble of this final rule. The financial arrangements list will identify eligible clinicians or MIPS eligible clinicians that have a financial arrangement, as discussed in section X.A.3.g of the preamble of this final rule, with the TEAM participant, TEAM collaborator, collaboration agent, and downstream collaboration agent. The clinician engagement list will identify eligible clinicians or MIPS eligible clinicians that participate in TEAM activities and have a contractual relationship with the TEAM participant, and who are not listed on the financial arrangements list. We do not believe a requirement that certain providers or suppliers be included on these lists is necessary to meet the goals of the model. We believe it is important for the TEAM participant, and not CMS, to dictate who should be included on these lists.
Comment: A commenter suggested CMS should be sensitive to the prevalence and movement of surgeons between hospitals and should include PGPs as direct participants in the model to address practice patterns shifting.
Response: We recognize that individual Medicare suppliers that were present during the baseline period may be different during the performance year. However, the shifting of providers is inevitable and will continually occur during the baseline period and performance year and that alone does not support the commenter's assertion that PGPs should be direct TEAM participants and assume financial accountability in the model. We are hopeful that TEAM participants will ( print page 69646) take steps to partner with PGPs during the performance year to engage physicians in value-based care.
Comment: A commenter suggested the development of specific quality measures and performance standards that reflect the unique contributions of PGPs to ensure the inclusion of PGPs positively impacts TEAM.
Response: We thank the commenter for the feedback. While we are finalizing the TEAM participant definition as proposed with slight modification to account for hospitals eligible to voluntarily opt into TEAM, as discussed in section X.A.3.a.(2)(c) of the preamble of this final rule. We may consider including PGPs and/or different quality measures in the model in the future.
Comment: Some commenters suggested that CMS should allow Ambulatory Surgical Centers (ASCs) to participate as participants in the model or allow ASCs to be a care setting where episodes may initiate. A commenter noted potential concerns with TEAM participants shifting volume to ASCs to diminish exposure to TEAM or individual providers shifting volume to ASCs to avoid downside risk. Another commenter noted that there is a huge shift of LEJR out of the hospitals into ASCs, leaving only the higher risk patients remaining in the hospital. A commenter noted that procedures done in an ASC would not accrue shared savings under TEAM and questioned how CMS will track these activities for impact on patients or unintended consequences in the model. Another commenter indicated that some hospitals could feel incentivized to direct high-cost, complex LEJR patients to ASCs to improve their own facility's quality scores.
Response: We appreciate the interest of the commenters in providing certain episode categories, such as LEJR, under TEAM to Medicare beneficiaries in ASCs as a further opportunity to test strategies to provide high quality, efficient care for beneficiaries. We recognize that testing episodes that initiate in an ASC setting would require us to expand the TEAM participant definition to include ASCs or PGPs. We have previously noted our concerns with requiring PGPs to participate in TEAM and are also hesitant to expand the definition of TEAM participant to include ASCs as TEAM participants without having data to support an assertion that they can assume accountability in a two-sided risk model. We have not tested or allowed ASCs to be participants or awardees in the previous episode-based models that served as examples during our development of TEAM. However, we will take commenters feedback into consideration should we want to expand the definition of TEAM participant in future notice and comment rulemaking.
We acknowledge that testing episodes in ASCs is an area where the CMS Innovation Center can expand its understanding of site neutrality in episode-based payment models. We have experience from BPCI Advanced and CJR constructing site neutral target prices from IPPS and OPPS data but have not constructed target prices using data from the ASC Payment System. We are unsure if including episodes that initiate in the ASC setting in the model would require a more ASC-specific target price or if a more site neutral approach would be appropriate when considering episodes initiated in the hospital inpatient, hospital outpatient department, and ASC settings. Further, the current quality measures that we are finalizing for TEAM, as discussed in section X.A.3.c of the preamble of this final rule, are hospital-level measures and may not be appropriate for episodes initiated at ASCs and thus would need further consideration if TEAM included episodes initiated in ASCs.
With respect to shifting volume or unintended consequences for not including episodes that initiate in ASCs in TEAM, we note that in a review of Medicare payments that compared hospitals in the CJR model and a comparison group, CMS found that for total hip arthroplasty (THA) and total knee arthroplasty (TKA), two procedures captured in the LEJR episode, the rates of ASC utilization have slowly been increasing over the years, but overall have remained fairly low. While ASC utilization may still be relatively low, we disagree that utilization may increase because of TEAM participants directing high-cost, complex beneficiaries to ASCs in order to avoid inclusion of these beneficiaries in the model. Generally, high-cost, medically complex beneficiaries have procedures performed in the hospital inpatient setting and we believe TEAM participants will work with beneficiaries to make medically appropriate decisions. We intend to monitor beneficiary care, as discussed in section X.A.3.i of the preamble of this final rule. We will monitor for any patterns of inappropriate care, which includes monitoring the proportion of patients who are treated in different care settings by TEAM participants in comparison to non-TEAM participant hospitals. If we see that certain hospitals are treating patients in the various care settings at a rate that is different from their peers and cannot be explained by aspects of the hospital's patient population such as age or area-level socioeconomic factors, then we have multiple options for remedial action, as described in section X.A.1.f of the preamble of this final rule. This may include requiring the TEAM participant to develop a corrective action plan and reducing or eliminating a TEAM participant's reconciliation payment amount, as described in section X.A.3.d.(5)(j) of the preamble of this final rule. We will also continue to share changes in practice patterns and trends we identify through evaluation reports and other means.
Comment: A commenter wanted to know how anesthesia and hospital-based anesthesia providers fit into TEAM.
Response: The episode categories tested in TEAM are all surgical procedures that may require the use of anesthesia during the anchor hospitalization or anchor procedure, or during some other provider encounter, such as a hospital readmission. The cost of anesthesia items and services are included in the episode and factored into target prices. Hospital-based anesthesia providers may furnish services to the TEAM beneficiary, be part of the care team, and could potentially be a TEAM collaborator, as described in section X.A.3.g.(3) of the preamble of this final rule, that has a financial arrangement with a TEAM participant that would allow sharing reconciliation payment amounts or repayment amounts.
Comment: Some commenters recommended that CMS not allow overlap between TEAM and the AHEAD model. A few commenters suggested allowing hospitals participating in AHEAD to opt-out of TEAM, especially since hospitals in AHEAD will be voluntarily assuming global budgets for all hospital services, including the hospital-based portion of the episodes in TEAM. A commenter recommended that CMS exclude the states or the CBSAs that are participating in the AHEAD model from TEAM as they will have accountability for total cost of care, including hospital global budgets, and should have the ability to implement their specific state-based approach. A commenter noted that such overlap could lead to unintended consequences and exacerbate hospitals' financial challenges, especially given the unknown impact of each model. A commenter acknowledged that the discharge planning processes required under TEAM could end up aligning well with a hospital's AHEAD model strategy and believed it would be premature to decide now about whether to exempt ( print page 69647) AHEAD hospitals from TEAM participation. A commenter supported sharing TEAM-style summary episode data with AHEAD hospitals to encourage the integration of episodes of care into hospital global budgets and that hospitals in AHEAD states that decline to join AHEAD should be part of TEAM. A commenter supported excluding hospitals in Maryland from TEAM.
Response: We thank the commenters for their feedback. As noted in the proposed rule ( 89 FR 36389 ) and preamble of this final rule, we were uncertain on how to handle overlap between hospitals selected to participate in TEAM and those who may also participate in the AHEAD model. We acknowledge commenters opinions, but we are not convinced that the two models should be mutually exclusive. Specifically, we think that different service and payment delivery models could co-exist and work synergistically to improve health care cost and quality outcomes. We disagree that a hospital being in a total-cost-of care (TCOC) model or assuming hospital global budgets is reason enough to be excluded or to allow an opt-out policy. Similar to Medicare ACO initiatives, where organizations are accountable for total cost of care, we believe there are complementary incentives between TEAM and the AHEAD model that could result in hospitals achieving maximum success in improving beneficiary quality of care, reducing acute care costs, and reducing post-acute care costs through participation in both initiatives. For example, hospital global budgets focus on controlling hospital, or acute care volume and spending while episodes generally elicit reductions in post-acute care spending. We believe a hospital participating in both a hospital global budget initiative, like the AHEAD model, and in an episode-based payment model, like TEAM, could benefit from both models' unique cost savings opportunities. Further, hospital global budgets can encourage improvements in population health, while episodes help providers to focus on making improvements for a narrower pool of patients associated with higher cost clinical conditions or procedures. We believe combining both approaches could help hospitals to achieve the best outcomes in patient care and cost reductions broadly and for specific beneficiaries.
We agree that the overall impact of allowing model overlap is unknown, however, we believe there could be an opportunity for us to evaluate and learn from the interaction of both models. The BPCI Advanced and CJR models, which TEAM is predicated on, did not allow overlap with hospital global budget models operating at that time (which were more limited in scope). Thus, we do not have insightful data on how these types of payment models can coexist. We believe that excluding AHEAD participants, except Maryland, from TEAM would prevent us from evaluating their combined effects. Further, permitting voluntary opt-out from TEAM for AHEAD participants, meaning a hospital selected for TEAM participation could opt-out of TEAM if they participated in AHEAD, would introduce selection bias and potentially yield less generalizable results for TEAM.
Therefore, we are finalizing the definition of TEAM participant as proposed with slight modification to account for hospitals eligible to voluntarily opt into TEAM, as discussed in section X.A.3.a.(2)(c) of the preamble of this final rule. We are also finalizing allowing overlap between hospitals selected to participate in TEAM and those that may also participate in the AHEAD Model, except for hospitals in Maryland. We are not finalizing a policy that would allow future AHEAD participants to voluntarily opt-out of TEAM, nor are we finalizing any payment adjustments to account for the same beneficiaries attributed to both models, which is consistent with our approach to TEAM and ACO overlap, as discussed in section X.A.3.e of the preamble of this final rule. We recognize that as of the date of publication of this final rule the hospitals that may choose to voluntarily participate in the AHEAD model are unknown. However, we believe it's important to be transparent about our policy desire to allow overlap—for purposes of testing the interaction of both model designs, as well as responsibly planning for potential model scalability. We will be considering more detailed overlap policies in future notice and comment rulemaking to ensure that hospitals considering joining the AHEAD model do not view participation in TEAM as a deterrent. We will consider more detailed overlap policy with the AHEAD model as plans surrounding the participating states and hospitals in those states develop.
We note that, as proposed, we are finalizing our policy to exclude Maryland acute care hospitals from TEAM.
Comment: A commenter asked CMS to maintain as a guiding principle that hospitals do not propose and perform surgical procedures; surgeons do.
Response: We recognize that there are multiple providers and suppliers involved in a beneficiary's care during an episode of care in TEAM. We acknowledge that the physician and non-physician practitioners are the individuals ordering and performing the surgery and providing the hands-on care to the beneficiary, while the TEAM participant furnishes hospital services and is the financially accountable entity facilitating care coordination and responsible for quality and cost outcomes.
Comment: A commenter suggested CMS allow health systems, rather than individual hospitals, to participate in TEAM because it would support health systems' development of centers of excellence without penalizing the sites where the most complex and least elective care is provided.
Response: We appreciate the commenter's suggestion. We believe it would be challenging to require health systems to participate in TEAM, given the lack of a standard definition for a “health system.” We anticipate that hospitals in health systems that participate in this model would leverage their participation to share learnings and standardize their care practices across all the hospitals in the health system. We also note the commenter's concern for potential disincentives for hospitals that care for medically complex beneficiaries, and we would like to highlight that target prices in TEAM include beneficiary level risk adjusters that account for patient acuity, as discussed in section X.A.3.d.(4) of the preamble of this final rule.
After consideration of the public comments we received, we are finalizing our proposed TEAM participant definition at § 512.505 with slight modification to include hospitals that make a voluntary opt-in participation election to participate in TEAM in accordance with § 512.510 and are accepted to participate in TEAM by CMS, as described in section X.A.3.a.(2)(c) of the preamble of this final rule. We are also finalizing as proposed our proposal to exclude Maryland hospitals from TEAM. Lastly, we are finalizing a policy to allow TEAM participants to also participate in the AHEAD model.
We proposed to require hospitals located in selected CBSAs, as described in section X.A.3.a.(4) of the preamble of this final rule, that meet the proposed TEAM participant definition to participate in TEAM. Such hospitals would be required to participate in the Model even if they have not had previous episode-based payment model or value-based care experience. Shifting ( print page 69648) hospitals away from the traditional Medicare FFS payment system to value-based care may require significant time, effort, and resources to build infrastructure and establish care redesign processes. [ 867 ] We stated in the proposed rule that we intend to provide sufficient time for potential TEAM participants to prepare for model implementation, which is why we proposed TEAM at least 18 months before the proposed model start date. However, we acknowledged that time alone may not be adequate to prepare TEAM participants for model participation, especially those with limited or no value-based care experience. We sought comment on whether one year would be a sufficient amount of time for hospitals required to participate in TEAM to prepare for TEAM participation or whether a longer timeframe (for example, 18 months) or shorter timeframe (for example, 6 months) would be sufficient time for hospitals to prepare to become TEAM participants, effective on the model start date ( 89 FR 36390 ).
We alternatively considered making participation in TEAM voluntary. However, we noted in the proposed rule that we would be concerned that a fully voluntary model would not lead to meaningful evaluation findings especially since the CMS Innovation Center has tested voluntary episode-based payment models for over a decade ( 89 FR 36390 ). We note that voluntary models have been impacted by selection bias through self-selection in and out of the model and selections of episodes or clinical episode service line groups. We recognize that a mandatory model test limits the selection of participants to only those captured within the selected geographic areas. We also recognize there may be participants of previous or current models that wish to continue their care redesign efforts, further care transformation, and maintain efficiencies to avoid reliance on the volume-based FFS payment system. We considered allowing hospitals that have previously participated (or are currently participating) in a Medicare episode-based payment model to voluntarily opt-in to TEAM to increase the footprint of the model and allow those entities to maintain their momentum in value-based care. We noted in the proposed rule that we recognize several challenges with including a voluntary opt-in for a model such as TEAM. We noted in the proposed rule that allowing an opt-in may limit the ability of the model to achieve Medicare savings, given that opt-in participants may self-select into the model based on their belief that they would benefit financially. Second, we also noted in the proposed rule that a voluntary opt-in may compromise the rigor of our evaluation of TEAM, because it could limit the number of hospitals available for our comparison group and our ability to detect generalizable evaluation results, due to participant self-selection into the model. Finally, we noted in the proposed rule that we have been testing the five episode categories that we proposed to include in TEAM, as described in section X.A.3.b. of the preamble of this final rule, on a voluntary basis via BPCI Advanced and the BPCI Initiative, so we have a significant amount of data on the performance of those episode categories in a voluntary structure already.
For these reasons, we did not propose a voluntary opt-in participation arm to TEAM. However, for the reasons discussed below, we sought comment regarding a voluntary opt-in participation arm for TEAM. Specifically, we considered limiting voluntary opt-in participation in TEAM to hospitals that currently participate in the BPCI Advanced or the CJR model, that are not located in an area mandated for TEAM participation, and that continue to participate until completion in the model in which they are currently participating. [ 868 ] For those hospitals that meet this criteria and that would want to voluntarily opt-in to TEAM participation, we stated in the proposed rule that we would require those hospitals to participate in all TEAM episode categories for the full five-year model performance period and they would not be permitted to voluntarily terminate model participation. The TEAM voluntary opt-in would be a one-time opportunity to join TEAM and those hospitals would need to submit a completed application to CMS in a form and manner and by a date specified by CMS, prior to the first performance year of TEAM. Further, we stated that, at a minimum, hospitals that submit an application would need to undergo and pass multiple levels of program integrity and law enforcement screening. Hospitals that pass this screening would be offered a participation agreement from CMS to participate in TEAM, which would, at a minimum, subject them to all the same terms, conditions and requirements of those hospitals mandated to participate in TEAM. Lastly, hospitals offered a participation agreement to voluntarily opt-in to TEAM would be required to submit and execute a participation agreement with CMS in a manner and form, and by a date specified by CMS prior to the model start date.
We stated in the proposed rule that we believe that offering this potential voluntary opt-in consideration would allow those hospitals that have made significant investments in care redesign and episode management to further their efforts to improve beneficiary quality of care and reduce Medicare spending. We recognize the pool of hospitals that could potentially apply for voluntary opt-in participation may be narrow. However, we believe extending the voluntary opt-in opportunity to hospitals not mandared to participate in TEAM that terminated BPCI Advanced or CJR model participation or to hospitals not mandated to participate in TEAM that did not participate in BPCI Advanced or CJR at all would result in the voluntary opt-in policy applying to too many hospitals and could jeopardize the model's ability to have a robust evaluation. This is because we would want to ensure we have a sufficient comparison group of hospitals not participating in TEAM to produce generalizable findings. As previously indicated, we did not propose a voluntary opt-in participation arm to TEAM; however, we considered and sought comment regarding a voluntary opt-in participation arm in the proposed TEAM. Lastly, we sought comment on our proposal for hospitals located in selected geographic areas that meet the proposed TEAM participant definition to participate in TEAM.
The following is a summary of comments we received on the proposed mandatory participation in TEAM and the voluntary opt-in considerations and our responses to these comments:
Comment: A few commenters supported the mandatory participation in TEAM. A commenter noted that such comprehensive testing is crucial for understanding how these models can save Medicare funds and enhance care efficiencies. Another commenter indicated mandatory advanced APMs are more likely to generate net savings for Medicare than voluntary advanced APMs because mandatory models do not experience the selection problems that have undermined voluntary models. A commenter stated that requiring hospital participation will ( print page 69649) allow CMS to understand its impact more fully on different patient groups, especially underserved populations, ahead of any further model expansion.
Response: We thank the commenters for their support and agree with their comments. As explained in the preamble of this final rule, mandatory participation eliminates selection bias and participant attrition issues, helps to capture a representative sample of different types of hospitals, and facilitates a comparable evaluation comparison group. We maintain that the mandatory design for TEAM is necessary to enable CMS to detect change reliably in a generalizable sample of hospitals to support a potential model expansion.
Comment: Numerous commenters requested that CMS make TEAM a voluntary model and allow hospitals to select individual episode categories. Many commenters identified the increased financial risk and the upfront costs required for implementation of the model. Some commenters had concerns with the scope of the model being too large for a mandatory model. A commenter urged CMS to revise the mandatory nature of the proposal and instead create incentives for interested participants that would reward innovation and high-quality patient care. Another commenter suggested that the model first be tested among those hospitals with the requisite experience, competencies, and strong post-acute care referral partners to ensure patients receive high-quality and appropriate levels of care. A commenter suggested waiving mandatory participation of hospitals with recent participation in the BPCI Advanced and CJR models because these hospitals have made substantial strides in improving efficiencies and optimizing episode performance.
Response: We thank the commenters for their feedback but disagree with the suggestion to finalize TEAM as a voluntary model. Testing TEAM as a mandatory model will give CMS the ability to test how an episode payment model might function among participants that would otherwise not participate in such a model and is also responsive to federal partners feedback supporting mandatory model tests. [ 869 870 ] As such, we expect the results from TEAM will produce data that are more broadly representative than what might be achieved under a voluntary model. We do not agree with allowing TEAM participants to select individual episode categories as that will introduce selection bias and make evaluating TEAM more difficult and produce less generalizable findings. Requiring TEAM participants to be accountable for all episode categories tested helps to broaden care transformation efforts, include more beneficiaries in value-based care, and apply efficiencies across multiple different service lines. We also disagree with excluding hospitals that have previously participated in the BPCI Advanced and CJR models because previous participation in these models does not guarantee these hospitals were able to find efficiencies, improve patient outcomes, and achieve overall success. Further, including hospitals from the BPCI Advanced and CJR models will incentivize them to continue improved care and efficiencies they started under the previous models. We also disagree that TEAM should be tested only where hospitals meet certain requirements as this would limit evaluation findings and not capture hospitals new to value-based care, where we believe it's important that they have the same opportunity for participation in a mandatory model to gain experience and work towards improving beneficiary quality of care and reducing Medicare spending.
We believe the relatively narrow scope of the model of testing five episode categories and the availability of Track 1, which allows the phasing in of full financial risk, along with our plan to engage with hospitals through the learning system and provide data to help them succeed under this model will aid hospitals in succeeding under TEAM. As discussed in section X.A.3.a.(1) of the preamble of this final rule, we are finalizing that the model start date is January 1, 2026, which provides TEAM participants with approximately 17 months' notice before implementation and what we believe is sufficient time to prepare for participation by identifying care redesign opportunities, beginning to form financial and clinical partnerships with other providers and suppliers, and using data to assess opportunities for success under the model.
As previously mentioned, we disagree that TEAM will create significant financial risk or necessitate a payment to account for administrative costs. We believe that by allowing all TEAM participants the opportunity to participate in Track 1 for the first performance year, this will provide additional preparation time before being subject to downside financial risk. We are also finalizing TEAM policies that we believe will minimize financial risk for TEAM participants, including reducing the discount factor, reducing the stop-gain and stop-loss limits for Track 2, and allowing safety net hospitals the opportunity to remain in Track 1 for the first three performance years, as discussed in sections X.A.3.d.(3)(g), X.A.3.d.(5)(h), and X.A.3.a.(3) of the preamble of this final rule.
We believe that by holding hospitals accountable for episodes of care, TEAM will incentivize care coordination and care redesign activities that may reduce readmissions, complications, and unnecessary health care spending. We believe TEAM will improve beneficiary care by improving care transitions and the overall care experience during the anchor hospitalization or anchor procedure and post-discharge period. Hospitals stand to benefit from TEAM, in the form of the opportunity to earn reconciliation payment amounts if successful under the model.
Comment: Some commenters have concerns with CMS' authority to test TEAM. A commenter believes TEAM is an overreach of CMS's authority that contradicts the statutory mandate of section 1115A and raises concerns about impermissible delegation of lawmaking authority to the executive branch and unjust compensation for services provided to Medicare beneficiaries. The commenter also notes that they believe requiring Medicare providers to be held financially accountable if spending exceeds the model's reconciliation target price means that Medicare providers will be required to furnish medically necessary services to Medicare beneficiaries without payment. They believe mandatory demonstrations with two-sided risk does not justly compensate Medicare providers for the use of their services by Medicare beneficiaries and is in violation of the Fifth Amendment of the United States Constitution and the Medicare statute.
Another commenter objected to the way the CMS Innovation Center is testing TEAM and indicated that new payment and delivery models should not impede patient access, undermine physician practices, or discourage medical progress through top-down governmental price-setting, and that they comply with the statute (section 1115A of the Act) and U.S. Constitution. Further, the commenter indicated that CMS is essentially proposing TEAM as a Phase II mandatory model before proper testing and evaluation has been performed on a limited, voluntary basis under Phase I, especially since prior models to date have not been evaluated ( print page 69650) and found to meet the criteria for Phase II expansion. A few commenters urged CMS to test TEAM on a voluntary basis first before mandating participation.
Response: CMS' testing of payment and service delivery models, including TEAM, complies with section 1115A of the Act and other governing laws and regulations, including the U.S. Constitution. We believe that we have the legal authority to test TEAM and to require the participation of all hospitals, as defined and finalized in section X.A.3.a.(2)(b) of the preamble of this final rule, located in the mandatory CBSAs selected for participation, as described and finalized in section X.A.3.a.(4) of the preamble of this final rule. We believe this model test is not an impermissible delegation of lawmaking authority that is inconsistent with section 1115A of the Act. First, we note that TEAM will not be the first CMS Innovation Center model that requires participation under the authority of section 1115A of the Act; we refer readers to the Comprehensive Care for Joint Replacement (CJR) Payment Model for Acute Care Hospitals Furnishing Lower Extremity Joint Replacement Services Final Rule ( 80 FR 73274 ), and the Home Health Prospective Payment System (HHPPS) Final Rule ( 80 FR 68624 ) implementing the Home Health Value-Based Purchasing (HHVBP) Model. Hospitals in selected Metropolitan Statistical Area (MSAs) were required to participate in the CJR Model beginning in April 2016, and home health agencies in selected states were required to participate in the HHVBP Model beginning in January 2016.
We believe that both section 1115A of the Act and the Secretary's existing authority to operate the Medicare program authorize us to finalize mandatory participation in TEAM for selected mandatory CBSAs, and we note that Medicare participation remains voluntary regardless of TEAM mandatory participation. Section 1115A of the Act authorizes the Secretary to test payment and service delivery models intended to reduce Medicare costs while preserving quality of care. The statute does not require that models be voluntary or be tested first as a voluntary model, but rather gives the Secretary broad discretion to design and test models that meet certain requirements as to spending and quality. Although section 1115A(b) of the Act describes a number of payment and service delivery models that the Secretary may choose to test, the Secretary is not limited to those models. Rather, as specified in section 1115A(b)(1) of the Act, models to be tested under section 1115A of the Act must address a defined population for which there are either deficits in care leading to poor clinical outcomes or potentially avoidable expenditures. Here, TEAM addresses a defined population (FFS Medicare beneficiaries who initiate an anchor hospitalization or anchor procedure for specific episode categories) for which there are potentially avoidable expenditures (arising from incentives that may encourage volume of services over the value of services). We designed TEAM to require participation for hospitals to avoid the selection bias inherent to any model in which providers and suppliers may choose whether or not to participate. Such a design will ensure sufficient participation of hospitals, including different types of hospitals such as safety net hospitals, which is necessary to obtain a diverse, representative sample of hospitals that will allow a statistically robust test of the model. We believe this is the most prudent approach for the following reasons. Under the mandatory TEAM, we will test and evaluate a model across a wide range of hospitals, representing varying degrees of experience with episode-based payment models. We note that TEAM is not a nationwide test and mandatory CBSAs are selected through randomization in order to have adequate comparison groups to evaluate the model. The information gained from testing the mandatory TEAM will allow CMS to comprehensively assess whether TEAM would be appropriate for a potential expansion in duration or scope, including on a nationwide basis. Thus, we disagree that TEAM is not a Phase I model test and believe that TEAM meets the criteria required for Phase I model tests.
Moreover, the Secretary has the authority to establish regulations to carry out the administration of Medicare. Specifically, the Secretary has authority under sections 1102 and 1871 of the Act to implement regulations as necessary to administer Medicare, including testing this Medicare payment and service delivery model. We note that TEAM is not a permanent feature of the Medicare program; TEAM will test different methods for delivering and paying for services covered under the Medicare program, which the Secretary has clear legal authority to regulate. The proposed rule went into detail about the provisions of the proposed TEAM, enabling the public to understand how TEAM was designed and could apply to affected hospitals. As permitted by section 1115A of the Act, we are testing TEAM within specified limited geographic areas. The fact that TEAM will require the participation of certain hospitals does not mean it is not a Phase I Model test. If the TEAM test meets the statutory requirements for expansion, and the Secretary determines that expansion is appropriate, we would undertake rulemaking to implement the expansion of the scope or duration of TEAM to additional geographic areas or for additional time periods, as required by section 1115AI of the Act.
We do not believe TEAM will impede patient access. We rely on Medicare providers and suppliers to furnish appropriate care to Medicare beneficiaries. TEAM upholds a Medicare beneficiary's freedom of choice and access to care, as discussed in section X.A.3.i of the preamble of this final rule, and we will monitor for unintended consequences of TEAM including but not limited to beneficiary access to care. If our monitoring reveals that TEAM reduces patient access, we would investigate and consider making changes to the model via future rulemaking.
We also do not believe TEAM will undermine physician practices or discourage medical progress by price-setting. TEAM will continue to drive greater value-based care participation, whereby providers and suppliers can focus on the value of care provided compared to the volume of items and services delivered. As an episode-based payment model, care coordination plays a significant role in TEAM participants achieving improved beneficiary quality of care and reduced Medicare spending. Hospitals selected to participate in TEAM will need to coordinate and communicate with many providers and suppliers, including physician practices, to ensure TEAM beneficiaries receive optimal care and outcomes. This provides an opportunity for increased collaboration between TEAM participants and providers and suppliers to improve care pathways and offer access to model financial incentives, through financial arrangements.
With respect to the constitutional claim raised by one commenter, we also disagree that two-sided risk models, such as TEAM, require Medicare providers and suppliers to furnish medically necessary services to Medicare beneficiaries without payment. TEAM participants will continue to bill Medicare FFS and be compensated for the services they provide to TEAM beneficiaries throughout the course of the model. TEAM participants may be eligible to receive a reconciliation payment amount from CMS or may be required ( print page 69651) to pay CMS a repayment amount depending on their quality performance and spending compared to the reconciliation target price. This incentive structure is consistent with other Medicare programs in which if a Medicare provider or supplier does not meet certain performance metrics, they may experience positive or negative financial impacts. For example, the Hospital Value-Based Purchasing Program rewards acute care hospitals, through positive or negative payment adjustments under the IPPS, based on their quality of care provided in the inpatient hospital setting. [ 871 ] We also remind commenters that participation in the Medicare program is voluntary, and that TEAM is a time-limited model test.
Another commenter urged us to ensure that this program is implemented in a manner consistent with the statute and the U.S. Constitution, noting that the same commenter had submitted previous comments regarding constitutional constraints (but not providing any citation to that previous comment). For the reasons described elsewhere in this preamble, we disagree with the commenter's vague suggestion that this model runs afoul of statutory or constitutional constraints.
Lastly, we acknowledge that the BPCI Advanced and CJR models are still being evaluated, but we believe it would be premature to assume these models do not or would not meet the criteria for Phase II expansion. Both the BPCI Advanced and CJR models underwent significant changes based on interested parties' feedback and from findings that suggested the models may incur significant Medicare losses. While we have not published CJR model evaluation results that include findings from these changes yet, we recently released BPCI Advanced evaluation results that encompass the changes to the model which demonstrates significant Medicare net savings of approximately $465 million (or 3.4 percent of what Medicare payments would have been had the model not existed), offsetting losses in earlier model years. [ 872 ] As we gain further evaluation results from the BPCI Advanced and CJR models, we will take these findings into account, in conjunction with the findings from TEAM's evaluation, when determining which model or model features we may want to consider for Phase II expansion.
Comment: Some commenters expressed concerns for testing TEAM when evidence from CMS Innovation Center models have not been fully evaluated, specifically the ongoing evaluations of the BPCI Advanced and CJR models, for which both models were used to inform TEAM design. While other commenters suggested the existing evidence from CMS Innovation Center models does not support testing TEAM because the BPCI Advanced and CJR models have not successfully generated Medicare savings. A commenter recommends that CMS delay finalizing TEAM until the publication of the final CJR and BPCI Advanced evaluations. Another commenter indicated that adverse selection in voluntary models has not been an issue and the commenter believed that this is not a cause of the failures of past CMS Innovation Center demonstrations in generating savings and improving outcomes. Another commenter believed findings that the physician-led ACO's yielded savings for Medicare and not mandatory, hospital-controlled APMs supported the need to test voluntary models and not mandatory. [ 873 ]
Response: We do not agree with commenters that implementation of TEAM is premature or that it should not be implemented until results for the final BPCI Advanced or CJR model evaluations are available. These models have been tested for many years and we believe evidence already produced from these models supports the continued testing of episode-based payment models. We also anticipate that these future model evaluations may offer valuable information to assist CMS in potentially refining TEAM policies, while TEAM will offer additional insights that are not available under the BPCI Advanced and CJR models; in particular, insights with respect to episode-based payment models on a distinct set of episode categories for participants that would not otherwise participate under a model such as BPCI Advanced. Also, this model tests a different target pricing methodology and has shorter episode lengths as compared to the BPCI Advanced and CJR models. Testing this model will provide additional information for CMS and providers on successful payment structures and care redesign strategies.
We acknowledge that the BPCI Advanced and CJR model evaluation are still ongoing. At the time the proposed rule was published, publicly available evaluation data demonstrated that surgical episodes in the BPCI Advanced model consistently resulted in an estimated net savings to Medicare for the first three model years: approximately $204 million in savings for Model Years 12 and $71 million in savings for Model Year 3. More recent BPCI Advanced evaluation findings that were published after the publication of the proposed rule, demonstrated a net Medicare savings of $465 million in Model Year 4 for all episodes, not just surgical episodes. [ 874 ] We will continue to take into consideration these models future evaluation results, and if warranted, may propose policies in future notice and comment rulemaking to support our goals of improving beneficiary quality of care and reducing Medicare expenditures.
We disagree with the notion that adverse selection is not an in issue in CMS Innovation Center models. For example, the BPCI Advanced model is a voluntary model and the first evaluation report found that hospitals that have opted to participate in the model were more likely to be larger, urban facilities that were part of a health system and located in more competitive markets than all eligible hospitals. [ 875 ] Findings like this suggest that it may be more difficult to generalize evaluation results from a voluntary model and expect the model to have similar outcomes for participants that do not have these same characteristics.
Lastly, we acknowledge the commenter's statements about the contribution of voluntary, physician-led ACOs however, the voluntary Shared Savings Program is a different model design than TEAM's episode-based payment model construction. Further, models tested by the CMS Innovation Center generally go through a rigorous evaluation and these evaluations may differ in the breadth and scope as compared to evaluations done for other CMS programs and initiatives.
Comment: Many commenters urged CMS to exclude safety net hospitals, rural hospitals, Sole Community Hospitals (SCHs) and Medicare-Dependent Hospitals (MDHs) from mandatory participation in TEAM. Many commenters indicated that these types of providers are unable to absorb the additional costs and potential payment reductions that may arise from compulsory payment models. Some commenters indicated these hospitals do not have the experience or the infrastructure to be successful in risk-based models. Other commenters ( print page 69652) indicated that these hospitals will be disproportionately burdened and penalized under this model if required to participate in TEAM.
Response: We understand the commenters' concerns but a key reason for testing a model with required participation is, in fact, to examine and better understand the impact of a model on a broader range of hospital types, beneficiaries, and communities that are not usually included in a voluntary model. We believe that excluding these hospitals from the model test diminishes the generalizability of evaluation findings and could limit TEAM's ability to capture beneficiaries in the markets where these hospitals are located and thus prevent these beneficiaries from receiving the benefits of value-based care. This means a mandatory model that includes these types of hospitals could increase beneficiary access to improved care transitions, coordination and communication across acute and post-acute care, and screening/referral for health-related social needs for better recovery. Further, a mandatory model helps to continue value-based care for beneficiaries and providers since providers are required to participate and cannot leave at will. Further, we believe TEAM will encourage these hospitals, who may be new to episode-based payment models, to adopt and employ innovative approaches to caring for beneficiaries in an episode of care.
However, we recognize commenters' concerns with these hospitals having less experience with fewer financial resources, and potentially caring for a greater proportion of underserved beneficiaries, that may make participating in TEAM more challenging. As such, we address these concerns in sections X.A.3.a.(3) and X.A.3.d.(5)(h) of the preamble of this final rules, which includes allowing safety net hospitals to participate in Track 1 for the first three performance years with no downside risk and reducing the stop-gain and stop-loss limits for Track 2, the participation track that is open to safety net hospitals, rural hospitals, Medicare Dependent Hospitals, Sole Community Hospitals, and Essential Access Community Hospitals.
Comment: Many commenters indicated that CMS is not giving enough consideration to the potential harm that such a mandatory model could have on selected hospitals and the beneficiaries they serve. Some commenters stated that TEAM would create significant access and patient choice-related issues for beneficiaries following their underlying procedure. Other commenters recommended CMS to use its authority to implement compulsory pilot programs sparingly, as unintended ramifications could harm patients. A commenter indicated that hospital administrators with no clinical experience could be empowered by this model to alter hospital operations to optimize their facility's short-term performance metrics at the expense of quality and cost. Another commenter was concerned about hospice services included in the costs that TEAM participants would be accountable for in the shorter episodes which could lead to a risk of hospitals delaying appropriate hospice care. Another commenter said that TEAM will create unfortunate financial incentives for hospitals to: (1) reduce the number of services for higher-need patients below the level they require to achieve good outcomes; and (2) to simply avoid performing these surgeries on higher-need patients altogether.
Response: We do not see how participation in TEAM, in and of itself, would lead to beneficiary harm and that if beneficiary harm were to occur, that CMS would be responsible. First, and most importantly, we note that under the model, providers and suppliers are still required to provide all medically necessary services to beneficiaries, and that this model does not change beneficiary access to services, providers, or suppliers. Second, we note that there are already payment policies under Medicare FFS systems and payment models, such as BPCI Advanced, CJR and ACOs, that include similar incentives to promote efficiency, and we have not determined that beneficiaries have been harmed by those systems and models. Third, and as previously mentioned, we will monitor beneficiary care, as discussed in section X.A.3.i of the preamble of this final rule, to ensure beneficiary freedom of choice is not compromised. Through monitoring of the model, CMS will aim to ensure steering or other efforts to limit beneficiary access or move beneficiaries out of the model are not occurring. We also note the breadth of monitoring activities, which includes audits, CMS monitoring of utilization and outcomes within the model, and the availability of Quality Improvement Organization (QIOs) and 1-800-MEDICARE for reporting beneficiary concerns that can help us identify any beneficiary access or freedom of choice concerns in TEAM.
The model pricing methodology, discussed in section X.A.3.d of the preamble of this final rule, also includes features to protect against such potential harm, such as responsibility for post-episode spending increases that may capture if a TEAM participant is withholding or delaying medically necessary care, stop-gain policies that set a maximum threshold a hospital can earn a reconciliation payment amount, and other policies as detailed in that section. In summary, we note that TEAM does not constrain the practice of medicine and we do not expect clinical decisions to be made on the basis of TEAM participation and we do not expect TEAM to harm Medicare beneficiaries. As noted in section X.A.3.c of the preamble of this final rule, CMS will hold TEAM participants accountable for quality of care.
Comment: A commenter noted that if CMS finalizes the requirements for mandatory participation in TEAM, they must closely monitor for unintended consequences.
Response: We will conduct ongoing monitoring and evaluation analyses to watch for any unintended consequences of the model, as finalized in section X.A.3.o of the preamble of this final rule. We will also be monitoring beneficiary care for unintended consequences and refer to section X.A.3.i of the preamble of this final rule, for more discussion about how we will monitor for unintended consequences under TEAM.
Comment: A commenter indicated that when the primary goal of the mandatory feature is evaluation, it would seem that hospitals, staff, and patients are being asked to be involved in research without informed consent—a practice that would never be allowed if the organizing entity were not a government body.
Response: We recognize informed consent is a process used in research and in general health care decisions between providers and patient about health care procedures or interventions. However, we note that while CMS is required to evaluate its models in accordance with section 1115A of the Act, the primary goal of mandatory participation in TEAM is not evaluation, it is to test an innovative payment and service delivery model using an episode-based pricing methodology for five surgical episode categories that aims to preserve or enhance the quality of care and reduce Medicare costs, in a large, nationally representative group of providers. We also refer readers to our earlier response about CMS' authority to test TEAM. Further, a Medicare beneficiary's freedom of choice is not changed or affected by TEAM, as discussed in section X.A.3.i of the preamble of this final rule, and beneficiaries have the ability to seek care from hospitals participating in the model and hospitals that are not ( print page 69653) participating in the model. Further, the beneficiary notification informs TEAM beneficiaries about the model, specifically how it will impact their care, their freedom of choice, their ability to report concerns, and other requirements as discussed in section X.A.3.i.(2).
Comment: A commenter indicated that hospitals selected for participation in TEAM that find themselves participating in multiple initiatives at one time could struggle to keep up with all the various quality and financial incentives, which could impact their overall operations and actually lead to higher overall administrative and regulatory compliance costs.
Response: We recognize that a hospital could be selected for participation in TEAM and be participating in other CMS initiatives at the same time. Hospitals are adept to handle rapid changes in the health care ecosystem, including policy and practice changes, along with multiple payer initiatives. It is not uncommon for CMS to test multiple models concurrently rather than sequentially. For example, the BPCI Advanced and CJR models are currently being tested and hospitals required to participate in CJR may also participate in the BPCI Advanced model for all episode categories except LEJR. In addition, CMS has a permanent ACO program (the Medicare Shared Savings Program), as well as multiple other ACO models in the testing phase, such as the ACO Realizing Equity, Access, and Community Health (ACO REACH) Model. We believe our decision to test TEAM at this time is consistent with the approach taken for other models and programs to test payment models that may share similar design features or target similar providers or beneficiaries. Such an approach provides CMS with additional information on the potential success of various model and program aspects and design features.
We also note that hospitals are already participating in various CMS quality reporting programs, and TEAM is not making changes to these existing initiatives. Further, a reason for using the quality measures selected in TEAM, as discussed in section X.A.3.c of the preamble of this final rule, was to minimize TEAM participant burden and use measures that hospitals were already reporting to the Hospital Inpatient Quality Reporting Program and the Hospital-Acquired Condition Reduction Program.
Comment: Many commenters supported a voluntary opt-in approach for TEAM to allow providers the means to continue care redesign efforts. Some commenters requested allowing PGPs to voluntarily opt-in to those geographic regions not selected for mandatory participation. Some commenters suggested expanding voluntary opt-in to previous BPCI Advanced and CJR participants, or to all hospitals regardless of geography or participation status in other episode-based models. A commenter indicated that mandatory participation in TEAM would undermine progress made to date by individual providers outside of the model and may exclude those who have historically participated in the BPCI Advanced and CJR model and done well, leaving them with no option once the two models end.
Response: We thank the commenters for their support of a voluntary opt-in opportunity in TEAM. As discussed in the proposed rule ( 89 FR 35934 ), we sought comment on the approach given it introduces self-selection into the model and could compromise the rigor of the evaluation of TEAM. However, since many commenters supported voluntary opt-in, demonstrating sufficient interest from the public, and for the additional reasons stated below, we have decided to finalize a voluntary opt-in policy for hospitals that participate in the BPCI Advanced and CJR models.
We recognize the value of allowing voluntary opt-in because it: (i) captures more beneficiaries in value-based care and gives them access to the benefits of the model (for example, improved care transitions); (ii) increases the volume of providers in APMs; (iii) supports continued investment in care transformation; (iv) maintains efficiencies and moves more providers away from the volume-based FFS payment system; and (v) furthers a CMS Innovation Center specialty care strategy goal to maintain momentum on acute episode payment models. [ 876 ] We believe these reasons, coupled with the public's interest in the approach, support our decision to allow voluntary opt-in for TEAM. Therefore, we are finalizing the policy to allow a one-time opportunity for BPCI Advanced and CJR participants to voluntarily opt-in to TEAM. This opt-in opportunity is only available to for hospitals that currently participate in the BPCI Advanced or the CJR model, that are not located in a mandatory CBSA selected for TEAM participation and continue to participate in BPCI Advanced or CJR until the last day of the last performance period or last performance year of the respective model. For the BPCI Advanced model, the last day of the last performance period, performance period 14, is December 31, 2025. For the CJR model, the last day of the last performance year, performance year 8, is December 31, 2024. To overcome selection bias concerns for selection of episode categories they will be accountable for, we will require these hospitals to participate in all episode categories tested in TEAM. We will also require the hospitals that voluntarily opt-in to TEAM to remain in the model for the full model performance period and they will not be permitted to voluntarily terminate model participation, which avoids attrition concerns. To mitigate evaluation concerns, we are finalizing our CBSA selection strata with modifications to accommodate the voluntary opt-in policy, as discussed in section X.A.3.a.(4) of the preamble of this final rule and are purposely keeping the pool of hospitals eligible to voluntarily opt-in narrow to ensure we can construct a sufficient, comparable comparison group.
We are also finalizing that prior to PY 1, any eligible hospitals, meaning hospitals that currently participate in the BPCI Advanced or the CJR model, that are not located in a mandatory CBSA selected for TEAM participation, and continue to participate in BPCI Advanced or CJR until the last day of the last performance period or last performance year of the respective model, that wish to pursue voluntarily opt-in to TEAM, must submit a written participation election letter to CMS in a form and manner specified by CMS during the voluntary election period of January 1, 2025-January 31, 2025. [ 877 ] The participation election letter will serve as the participation agreement which would bind and subject the hospitals to the same terms, conditions, and requirements in TEAM's regulations at § 512.500. However, CMS may choose to not accept a hospitals participation election letter, for reasons including, but not limited to, program integrity concerns or ineligibility. In instances where CMS does not accept a hospital's participation letter, CMS will notify the hospital within 30 days of the determination. For example, we recognize that the participation election letter will need to be submitted prior to December 31, 2025, the last day of the last performance period in the BPCI Advanced model. Hospitals eligible for ( print page 69654) voluntary opt-in based on BPCI Advanced participation that submit a participation election letter and then terminate their BPCI Advanced participation agreement will not be permitted to participate in TEAM. [ 878 ] Further, we are finalizing that the participation election letter must contain, at minimum, the following elements:
- Hospital Name
- Hospital Address
- Hospital CCN
- Hospital contact name, telephone number, and email address
- Model name (TEAM)
- Certification that—
++ The hospital will comply with all requirements of TEAM (that is, 42 CFR part 512.500 ) and all other laws and regulations that are applicable to its participation in TEAM; and
++ Any data or information submitted to CMS will be accurate, complete and truthful, including, but not limited to, the participation election letter and any other data or information that CMS uses for purposes of TEAM.
- Signed by the hospital administrator, chief financial officer, or chief executive officer with authority to bind the hospital.
Lastly, we recognize that the TEAM participant definition, as proposed, did not account for potential hospitals that might voluntarily opt into TEAM. Therefore, we are finalizing a slight modification to the definition of TEAM participant to include hospitals that make a voluntary opt-in participation election in TEAM, in accordance with § 512.510 and are accepted to participate in TEAM by CMS.
After consideration of the public comments we received, we are finalizing our proposal without modification for the mandatory participation of TEAM participants in mandatory CBSAs selected for participation. We are also finalizing a policy to allow a one-time opportunity to voluntarily opt-in to TEAM for hospitals that currently participate in the BPCI Advanced or the CJR model, that are not located in a mandatory CBSA selected for TEAM participation and continue to participate until the last day of the last performance period or last day of the last performance year, of the model in which they are currently participating. For the BPCI Advanced model, the last day of the last performance period, performance period 14, is December 31, 2025. For the CJR model, the last day of the last performance year, performance year 8, is December 31, 2024. Further, we are finalizing that hospitals eligible for voluntary opt-in must submit a written participation election letter during the voluntary election period of January 1, 2025-January 31, 2025, in the regulations at § 512.510. Lastly, we are finalizing our proposed TEAM participant definition at § 512.505 with slight modification to include hospitals that make a voluntary opt-in participation election to participate in TEAM in accordance with § 512.510 and are accepted to participate in TEAM by CMS.
We stated in the proposed rule that as we did with the CJR model, we continue to believe it is most appropriate to identify a single entity to bear financial accountability for making repayment to CMS if quality and spending performance metrics are not met under the model after CMS performs reconciliation. Consistent with the CJR model, we proposed to make TEAM participants financially accountable for the episode for the following reasons:
- We believe hospitals would play a central role in coordinating episode-related care and ensuring smooth transitions for beneficiaries undergoing services related to episodes. A large portion of a beneficiary's recovery trajectory from an episode would begin during the hospital inpatient stay or procedure performed in the hospital outpatient department.
- Most hospitals already have some infrastructure related to health information technology, patient and family education, and care management and discharge planning. This infrastructure includes post-acute care coordination infrastructure and resources such as case managers, which hospitals can build upon to achieve efficiencies under TEAM.
- We proposed that episodes in TEAM begin with an acute care hospital stay or hospital outpatient department procedure visit. Some episodes may be preceded by an emergency room visit and possible transfer from another hospital's emergency room, or followed by PAC. However, we do not believe it would be appropriate to hold a PAC provider or a hospital other than the TEAM participant where the inpatient stay or initial hospital outpatient procedure that initiated the episode happened fully financially accountable for an episode under this model.
Episodes in TEAM may be associated with multiple hospitalizations through readmissions or transfers. When more than one hospitalization occurs during a single episode, we proposed to hold the TEAM participant that initiated the episode, as described in section X.A.3.b.(5)(c) of the preamble of this final rule, financially accountable for the episode, nonetheless. We recognize that, particularly where the hospital admission may be preceded by an emergency room visit and subsequent transfer to a tertiary or other regional hospital facility, patients often wish to return home to their local area for post-acute care. Many hospitals have recently heightened their focus on aligning their efforts with those of community providers, both those in the immediate area as well as more outlying areas from which they receive transfers and referrals, to provide an improved continuum of care. In many cases, this heightened focus on alignment is due to the incentives under other CMS models and programs, including ACO initiatives such as the Shared Savings Program or the Hospital Readmissions Reduction Program (HRRP). In the proposed rule, we noted that by focusing on the TEAM participant as the accountable or financially responsible entity, we hope to continue to encourage this coordination across providers and sought comment on ways we can best encourage these relationships within the scope of TEAM ( 89 FR 36391 ).
We sought comment on our proposal to require TEAM participants to be financially accountable for episodes in TEAM.
In the proposed rule, we recognized for purposes of TEAM that a beneficiary in an episode may receive care from multiple providers and suppliers, and not just from the TEAM participant where the episode was initiated. We considered allowing providers or suppliers, other than the TEAM participant, to bear financial accountability for episodes given their involvement in a TEAM beneficiary's care. Specifically, we considered splitting financial accountability between the TEAM participant and other providers and suppliers that provide items and services to the TEAM beneficiary. For example, we considered the TEAM participant being financially accountable for a majority of the episode spending, such as all Medicare Part A spending, and other suppliers, such as PGPs, being accountable for a portion of episode spending related to Medicare Part B spending. However, we noted in ( print page 69655) the proposed rule that we have concerns about how to accurately determine a reasonable sharing methodology that reflects the portion of spending either the TEAM participant or the PGP should be financially accountable for. Further, we have concerns about requiring PGPs to be financially accountable given practices can vary by size and resources. As previously noted, the BPCI Advanced model includes PGPs, and the physician groups electing to participate in BPCI Advanced have done so because their practice structure supports care redesign and other infrastructure necessary to bear financial accountability for episodes. However, these physician groups are not necessarily representative of the typical group practice. The infrastructure necessary to accept financial accountability for episodes is not present across all PGPs, and thus we do not believe it would be appropriate to designate PGPs to bear a portion of the financial accountability for episodes under the proposed TEAM. Further, shared financial accountability would require more than hospitals being TEAM participants and introduces model complexity. We sought comment on approaches to splitting financial accountability when multiple providers care for a single beneficiary in an episode ( 89 FR 36391 ).
While we proposed that the TEAM participant would be financially responsible for the episode, we also believe that effective care redesign requires meaningful collaboration among acute care hospitals, PAC providers, physicians, and other providers and suppliers within communities to achieve the highest value care for Medicare beneficiaries. We believe it may be essential for key providers and suppliers to be aligned and engaged, financially and otherwise, with the TEAM participants, with the potential to share financial accountability for an episode with those TEAM participants. We noted in the proposed rule that all relationships between and among TEAM participants and other providers and suppliers would still need to comply with all relevant laws and regulations, including the fraud and abuse laws and all Medicare payment and coverage requirements unless otherwise specified further in this section and in section X.A.3.g of the preamble of this final rule. Depending on a TEAM participant's current degree of clinical integration, new and different contractual relationships among hospitals and other health care providers may be important, although not necessarily required, for TEAM success in a community. We acknowledged in the proposed rule that there may need to be incentives for other providers and suppliers to partner with TEAM participants and develop strategies to improve episode efficiency ( 89 FR 36392 ).
We acknowledged in the proposed rule the important role that conveners play in the BPCI Advanced model with regard to providing financial responsibility and infrastructure support to hospital and PGP participation in BPCI Advanced. The convener relationship (where another entity assumes financial responsibility) may take numerous forms, including contractual (such as a separate for-profit company that agrees to take on a hospital or PGP's financial risk in the hopes of achieving financial gain through better management of the episodes) and through ownership (such as when risk is borne at a corporate level within a hospital chain). We considered allowing convener entities, like those recognized in the BPCI Advanced model, to have formal roles in TEAM. At peak BPCI Advanced participation, over 70 percent, or 1,439, of the hospitals and PGPs in Model Year 3 (2020) participated as downstream episode initiators under one of the 92 convener participants. [ 879 ] While the majority of BPCI Advanced hospitals and PGPs participated under a convener participant, some hospitals and PGPs found the participation relationship with a convener challenging. Specifically, some hospitals and PGPs felt removed from participation decisions since they were not party to the participation agreement between CMS and the convener participant. Additionally, we noted in the proposed rule that convener participants that are not Medicare providers or suppliers may need financial guarantees that can impose significant upfront financial investment for participation and be administratively burdensome for CMS and the participant. We did not propose to require convener entities in this model, and we do not intend to identify or require any Medicare-enrolled providers or suppliers (or providers and suppliers that are not enrolled in Medicare) to be convener entities in TEAM, in light of the experiences and resources that would be needed to “convene” over one or more TEAM participants. As with the CJR model, we do not intend to restrict the ability of TEAM participants to enter into administrative or risk sharing arrangements related to TEAM with entities that may provide similar support as a convener, except to the extent that such arrangements are restricted or prohibited by existing law. We did not propose to require TEAM participants to partner with convener entities and we did not propose to require any entities, providers, or suppliers to serve as conveners for purposes of TEAM. We refer readers to section X.A.3.g. of the preamble of this final rule for further discussion of model design elements that may outline financial arrangements between TEAM participants and other providers and suppliers ( 89 FR 36392 ).
We sought comment on approaches to splitting financial accountability when multiple providers or suppliers care for a single beneficiary in an episode.
The following is a summary of comments we received on the proposed financial accountability of TEAM participants and other financial accountability considerations and our responses to these comments:
Comment: A commenter supported holding acute care hospitals accountable for all items and services during an episode.
Response: We thank the commenter for the support on the financial accountability for hospitals.
Comment: A commenter recommended that CMS make TEAM a shared savings model, where the participant and CMS have shared accountability for earning savings.
Response: We thank the commenter for the suggestion. If CMS were to structure TEAM as a shared savings initiative, then it may prevent TEAM from overlapping with other shared savings initiatives, including the Shared Savings Program, as described in 42 CFR 425.114 . CMS proposed, and is finalizing, a policy that permits overlap between TEAM and shared savings initiatives, as described in section X.A.3.e of the preamble of this final rule. We believe that overlaps between TEAM and shared savings initiatives are important, and we aim to encourage TEAM participants to collaborate with ACOs and ensure TEAM beneficiaries are connected back to the longitudinal providers to support continuity of care positive long-term health outcomes. We also note that TEAM participants may use financial arrangements to set-up their own sharing of accountability through sharing reconciliation payment amounts, or repayment amounts with TEAM collaborators and other entities, ( print page 69656) as discussed in section X.A.3.g of the preamble of this final rule.
Comment: Some commenters suggested that CMS require TEAM participants to set up financial arrangements with other providers and suppliers. A commenter believed that organizations outside of the hospital have no incentive or requirement to implement efficiencies when the hospital bears all financial responsibility. A commenter recommended allowing the clinical team to participate as the risk-bearing entity to better align incentives around the patient. Another commenter recommended that CMS adopt a mechanism to ensure that clinically relevant physicians have the option to be integrated into leadership and governance roles within this proposed model and to share in the savings generated by the model and direct participants to allot a meaningful portion of the shared savings payment within the model to physicians to account for their leadership and management of care redesign activities. Another commenter requested that CMS require equitable distribution of shared savings among physicians and clinical staff participating in the surgical episodes; establish performance parameters at the specialty-level; and embrace clinical integration that redistribute incentives in an upside/downside approach.
Response: We agree that financial arrangements provide an opportunity for TEAM participants to share their reconciliation payment amount or repayment amount resulting from participation in TEAM with certain providers and suppliers participating in TEAM activities. This means that providers and suppliers other than the TEAM participant could be subject to upside and downside financial risk depending on the terms of their financial arrangement. We also support TEAM participants including other providers and suppliers in the governance of their processes to implement TEAM as a mechanism to collaborate and encourage the use of financial arrangements to incentivize high value care. We believe that financial arrangements, as described in section X.A.3.g of the preamble of this final rule, can strengthen the relationship between TEAM participants and other providers and suppliers and encourage redesigned care processes for higher quality and more efficient service delivery. However, TEAM is not a shared savings initiative, and we want to give flexibility to TEAM participants to enter into financial arrangements or require providers and suppliers to be integrated in governance structures as they desire, consistent with law and regulation. We believe including such requirements in TEAM would increase burden on the TEAM participants and reduce their flexibility. The TEAM participant, not CMS, is best positioned to partner with providers and suppliers to determine the terms of the arrangements, which may vary by the TEAM participant's specific circumstances and capabilities, that ensure alignment to financial incentives to improve quality of care, drive equitable outcomes, and reduce Medicare spending.
Comment: A commenter encouraged CMS to meet with clinical stakeholders and prospective TEAM participants to discuss appropriate sharing methodologies and funds flow strategies suited for modern integrated delivery networks and physician-owned surgical practices.
Response: We thank the commenter for the recommendation and are always looking for opportunities to engage with interested parties on how to improve collaboration between all Medicare providers and suppliers that may care for a beneficiary in an episode of care.
Comment: A few commenters requested that CMS give other providers and suppliers, other than the hospital including post-acute care providers, the opportunity to be episode initiators and have financial accountability in the model. A couple of commenters suggested that CMS require the hospital to include these providers and suppliers in gainsharing incentives or require hospitals to pass on a proportional portion of the shared savings generated under this model.
Response: While we acknowledge the critical importance of other providers and suppliers, other than the hospital, including providing post-acute care, in helping to manage episodes which extend 30 days beyond discharge from the anchor hospitalization or anchor procedure, we continue to believe the hospital should be the financially accountable, episode initiator for TEAM. For hospitals to be successful in TEAM, we believe that they will need support from physicians, post-acute care providers, and other clinical care providers to provide the best quality of care in a cost-effective manner. This may involve establishing financial arrangements with such providers and suppliers. We support other types of providers and suppliers assuming risk where they are financially able to do so, and we agree that providers and suppliers that have a share in the risk, both positive and negative, may be more motivated to participate in value-based care. However, we do not believe that in a mandatory model like TEAM, any other provider or supplier is consistently as financially positioned to assume risk as the hospital. We also do not want to require financial arrangements or mandate a specific division of risk between TEAM participants and other providers and suppliers given we believe that the hospital is best positioned to determine the terms of these types of arrangements and not CMS.
Comment: A few of commenters requested that clinicians receive greater decision-making authority with respect to TEAM in light of CMS's proposal to hold hospitals financially accountable in the model. A commenter stated that given the hospital's financial control of the episode, it is imperative that all involved clinicians (including those involved post-discharge) have the authority to make the right decisions, which will impact patients' next level of care, as well as outcomes. Another commenter recommended that CMS consider providing hospital systems in the model with flexibility to have more decision-making capacity regarding referrals and appropriateness of post-acute facility utilization. Another commenter requested CMS consider greater flexibility and coverage to promote what is the appropriate care at the right place at the right time.
Response: TEAM participants will be financially accountable for episodes initiated in their hospital or hospital outpatient department and should work with all providers and suppliers, in addition to the beneficiary and their caregivers, to determine the proper plan of care for each TEAM beneficiary. We do not believe that providers and suppliers will compromise their patients' safety or deviate from the standard practice of care in an attempt to avoid negative financial implications of the model, and TEAM does not affect or change a participant's ability or authority to make the “right” decisions with respect to a beneficiary's care. Moreover, the TEAM beneficiary's role in decision making is imperative as well. TEAM beneficiaries will still be able to seek care from their choice of Medicare providers and suppliers. We will monitor beneficiary access and quality of care, as described in section X.A.3.i of the preamble in this final rule, to ensure steering or other efforts to limit beneficiary access or move beneficiaries out of the model are not occurring.
After consideration of the public comments we received, we are finalizing our proposals to hold the TEAM participant financially ( print page 69657) accountable for episodes in TEAM without modification.
In the proposed rule we stated that one way to help providers and suppliers gain experience in alternative payment models is through model participation tracks where the levels of risk and reward are reduced while the participants establish and hone their care redesign processes. Stakeholders have urged CMS to offer a glide path in its models, most recently in the Episode-based Payment Model RFI ( 88 FR 45872 ), to smooth the transition to risk. Such a glide path could provide more time for participants to gain experience with two-sided financial risk by phasing-in risk rather than requiring full-risk participation at the start of the model. Previous and current CMS models and programs have implemented this approach, including the recently announced Making Care Primary Model, which offers a progressive three-track approach that increases participants' accountability, and the Medicare Shared Savings Program, which offers an incremental glide path for ACOs to transition to higher levels of potential risk and reward. We note that these models and programs have longer durations than the model duration that we proposed in TEAM, which makes it easier to offer a gradual transition to two-sided financial risk or higher levels of risk and reward. However, in light of our proposal to make TEAM a five-year model test, we believe that TEAM participants would still benefit from the opportunity to ease into two-sided financial risk participation as they develop efficiencies ( 89 FR 36392 ).
We proposed that there will be three tracks in TEAM, each with differing financial risk and quality performance adjustments. Track 1 would be available only in PY 1 for all TEAM participants and would have only upside financial risk with the quality adjustment applied to positive reconciliation amounts. Track 2 would be available in PYs 2 through 5 to a limited set of TEAM participants, including safety net hospitals, and would have two-sided financial risk with the quality adjustment applied to reconciliation amounts. Lastly, Track 3 would be available in PYs 1 through 5 for all TEAM Participants and would have two-sided financial risk with the quality adjustment applied to reconciliation amounts ( 89 FR 36392 ).
We proposed a one-year glide path to two-sided risk for TEAM participants in an effort to ensure that TEAM participants have time to prepare for two-sided financial risk. We proposed to allow all TEAM participants to select between one of two tracks for the first performance year of TEAM. For PY 1, a TEAM participant may elect to participate in either Track 1 or Track 3. For PY 1, Track 1 would have upside-only financial risk provided through reconciliation payments, subject to a 10 percent stop-gain limit and a Composite Quality Score (CQS) adjustment percentage of up to 10 percent, as described in sections X.A.3.d.(5)(h) and X.A.3.d.(5)(g) of the preamble of this final rule, that would allow TEAM participants to be rewarded for their work to improve quality and cost outcomes for their episodes, but not be held financially accountable if spending exceeds the reconciliation target price. We believe the 10 percent stop-gain limit and a CQS adjustment percentage of up to 10 percent for Track 1 are appropriate and would allow TEAM participants to be rewarded for spending and quality performance while easing into financial risk. We proposed that Track 3 would have two-sided financial risk in the form of reconciliation payments or repayment amounts, subject to 20 percent stop-gain and stop-loss limits and a CQS adjustment percentage of up to 10 percent, as described in sections X.A.3.d.(5)(h) and X.A.3.d.(5)(g) of the preamble of this final rule, that would allow TEAM participants to have higher levels of reward and risk based on their quality and cost performance for their episodes. We proposed to only allow TEAM participants to participate in Track 1 for one performance year, specifically PY 1. We proposed a five-year model test, and we do not believe that making Track 1 available for more than one performance year would motivate TEAM participants to improve quality or spending performance since there would be no financial accountability when spending reductions are not achieved ( 89 FR 36392 ).
As indicated in the proposed rule, we believe a one-year glide path is an appropriate length of time for a five-year model test that aims to improve patient quality of care and reduce Medicare spending. We considered limiting eligibility for Track 1 during PY 1 to TEAM participants that have not previously participated in a Medicare episode-based payment model, but given that TEAM would be a mandatory model, we believe prior experience does not guarantee successful participation, and that it is important for TEAM participants to consider their own unique organizational position and characteristics when determining their desired track selection for PY 1. We sought comment on this proposal and whether there are alternative potential approaches for constructing a glide path in TEAM ( 89 FR 36393 ).
We proposed that TEAM participants would be required to notify CMS of their track selection prior to the start of PY 1, in a form and manner and by a date specified by CMS. TEAM participants who fail to timely notify CMS would be automatically assigned to Track 1 for PY 1. We sought comment on the proposal to require TEAM participants to notify CMS of their track selection and to automatically assign TEAM participants to Track 1 if they fail to timely notify CMS of their desired track selection.
We stated in the proposed rule that the proposed glide path opportunity was limited to one year. We proposed that TEAM participants who elected to participate in Track 1 for PY 1 would automatically be assigned to Track 3 for PY 2 and would remain in Track 3 for the remainder of the model (PYs 2 through 5). We recognize that offering different participation tracks in TEAM presents an opportunity to provide flexibilities to TEAM participants that may care for a greater proportion of underserved beneficiaries and TEAM participants that lack the financial reserves to invest in value-based care, including safety net, rural, and other hospital providers. Research has identified APM participation challenges for these types of providers, such as a lack of capital to finance the upfront costs of transitioning to an APM, including purchasing electronic health record technology, and challenges acquiring or conducting data analysis necessary for participation. [ 880 ] CMS has taken significant steps to address and improve health equity in value-based care models and programs, including health equity adjustments to the Hospital Value-Based Purchasing Program ( 88 FR 58640 ) and the Medicare Shared Savings Program ( 87 FR 69404 ).
We proposed to require different types of hospitals to participate in TEAM, and we believe that certain TEAM participants may benefit from a participation option that has limited two-sided financial risk so that their beneficiaries may receive high quality, coordinated care without imposing significant financial pressure. Therefore, ( print page 69658) we proposed that rather than automatically being assigned to Track 3 beginning in PY 2, certain TEAM participants could elect to participate in Track 2 beginning in PY 2 and stay in Track 2 for the remainder of the model (PYs 2 through 5). As further described in sections X.A.3.d.(5)(h) and X.A.3.d.(5)(g) of the preamble of this final rule, we proposed that Track 2 would have two-sided financial risk in the form of reconciliation payments and repayment amounts, subject to 10 percent stop-gain and stop-loss limits, a CQS adjustment percentage of up to 10 percent for positive reconciliation amounts, and a CQS adjustment percentage of up to 15 percent for negative reconciliation amounts. We believe the CQS adjustment percentage of up to 15 percent for negative reconciliation amounts, is appropriate for Track 2 because it further limits a TEAM participant's financial risk given that a higher CQS adjustment percentage for negative reconciliation amounts results in a lower repayment amount. In the proposed rule, we stated that the proposed payments and payment adjustments would allow TEAM participants to receive reconciliation payment amounts or owe repayment amounts based on their quality and cost performance for their episodes.
We proposed that only the following types of TEAM participants would be eligible to participate in Track 2 for PYs 2 through 5:
- Hospitals that are safety net hospitals, as further described in section X.A.3.f.(2) of the preamble of this final rule. For purposes of TEAM, we proposed that a TEAM participant must meet at least one of the following criteria in order to be considered a safety net hospital:
++ Exceeds the 75th percentile of the proportion of Medicare beneficiaries considered dually eligible for Medicare and Medicaid across all PPS acute care hospitals in the baseline period (as described in section X.A.3.d.(3)(a)).
++ Exceeds the 75th percentile of the proportion of Medicare beneficiaries partially or fully eligible to receive Part D low-income subsidies across all PPS acute care hospitals in the baseline period.
- Hospitals that are rural hospitals, as further described in section X.A.3.f.(3) of the preamble of this final rule. For purposes of TEAM, we proposed that a TEAM participant must meet at least one of the following criteria in order to be considered a rural hospital:
++ Is located in a rural area as defined under § 412.64.
++ Is located in a rural census tract defined under § 412.103(a)(1).
++ Has reclassified as a rural hospital under § 412.103.
++ Is a rural referral center (RRC), which has the same meaning given this term under § 412.96.
- Hospitals that are Medicare dependent hospitals (MDH) as defined under 42 CFR 412.108 .
- Hospitals that are sole community hospitals (SCHs) as defined under 42 CFR 412.92 .
- Hospitals that are essential access community hospitals as defined under 42 CFR 412.109 .
As noted in the proposed rule, we believe that allowing TEAM participants that meet the safety net hospital or rural hospital criteria, as well as those that are Medicare dependent hospitals, sole community hospitals, or essential access community hospitals to participate in Track 2 during PYs 2 through 5 would provide an opportunity for these hospitals to develop capabilities to deliver value-based care and would avoid the financial pressures of a two-sided financial risk model that could make their participation in TEAM untenable.
We proposed that TEAM participants that meet the Track 2 hospital criteria described above would be required to notify CMS on an annual basis prior to the start of every performance year, beginning for PY 2, of their desire to participate in Track 2. We proposed that TEAM participants that meet the Track 2 hospital criteria could switch between Track 2 and Track 3 on an annual basis. Such TEAM participants would need to notify CMS of their preference, in a form and manner and by the date specified by CMS. We proposed that TEAM participants would need to meet the hospital criteria for Track 2 participation by the date CMS requires notification of their preference. TEAM participants who fail to timely notify CMS or do not meet the Track 2 hospital criteria would not be approved by CMS to participate in Track 2 and would be automatically assigned to Track 3 for the given performance year. We recognize that allowing these specific TEAM participants to self-select into Track 2 for PYs 2 through 5 could create challenges when evaluating the model, such as the generalizability of evaluation findings. We also recognize that requiring these specific TEAM participants to notify CMS every year will permit them to switch tracks if they no longer desire to participate in Track 2 or no longer meet the Track 2 hospital criteria. Therefore, we sought comment on whether we should prohibit TEAM participants from switching tracks after PY 2 or if there are other options, we should consider to mitigate evaluation challenges ( 89 FR 36394 ).
We considered but did not propose allowing TEAM participants that meet the safety net hospital criteria to remain in Track 1 for all performance years so that they would not be subject to downside financial risk during their participation in the model. Further, we considered not allowing these TEAM participants that meet the safety net hospital criteria to switch between tracks, meaning that they would have to participate in Track 1 for all performance years. However, as stated in the proposed rule, we did not want to limit a TEAM participant that meets the safety net hospital criteria from making its own decision about whether to participate in a track with downside financial risk. Further, we believe that having downside risk by PY 2 for all TEAM participants would help to drive care improvements and establish care efficiencies that could lead to better outcomes on cost and quality of care. We sought comment on whether we should consider allowing TEAM participants who meet the safety net hospital criteria to participate in Track 1 for all performance years.
Table X.A.-01 summarizes the proposed TEAM tracks.

We sought comment on the proposals for the TEAM Participation Tracks at § 512.520. We also sought comment on the proposal to allow eligible TEAM participants to choose to participate in Track 2 and change their track selection from Track 2 to Track 3 annually.
The following is a summary of comments we received on the proposed participation tracks and our responses to these comments:
Comment: We had several commenters support CMS' effort to include a glide path to downside financial risk, through the construction of different participation tracks. A commenter noted that allowing all participants to join Track 1 for the first performance year creates opportunities to invest in quality improvement and infrastructure investments. Another commenter appreciated CMS offering flexibility to hospitals that care for a higher proportion of underserved individuals, such as safety net hospitals, by allowing several tracks for participation in the model with varying levels of financial risks and rewards.
Response: We thank commenters for their support of the participation tracks in TEAM.
Comment: Numerous commenters requested that CMS provide a longer glide path to downside financial risk for all hospitals. Many commenters suggested that all hospitals should have the opportunity to remain in Track 1 for at least the first two years of the model. Many commenters pointed to other APMs, including the Medicare Shared Savings Program, that have longer glide paths before participants take on downside financial risk. Some commenters recommended that all hospitals be eligible to participate in Track 2 due to the more demanding requirements set forth in Track 3. Many commenters believed that the level of financial risk for all participants was too significant on participants, especially in light of a 3 percent discount factor. Many commenters also requested that CMS consider allowing low volume hospitals to remain in Track 1 throughout the entirety of the model. Some commenters believed that a one-year glide path is insufficient since CMS has underestimated the resources that would be required to establish the care teams needed to lead and participate in TEAM. A commenter indicated that advancing participants who do not meet eligibility for Track 2 to Track 3 in the second performance year will not allow enough time for hospitals to review PY 1 data and make appropriate adjustments. A commenter encouraged CMS to consider extending glide path to two performance years, particularly for TEAM participants that had no prior experience in the CJR or BPCI models. Another commenter stated their belief that an extended upside-only period would grant TEAM participants the necessary time to explore other and possibly less obvious, or more innovative, cost management practices.
Response: We appreciate the comments requesting that CMS extend Track 1 beyond PY 1, and we believe it may be necessary to extend the length of Track 1 for TEAM participants that are safety net hospitals, as discussed in the subsequent comment response, but we are not persuaded that this extension is necessary for TEAM participants that are not safety net hospitals. As discussed in section X.A.3.a.(1) of the preamble of this final rule, we are finalizing a model start date of January 1, 2026. This gives TEAM participants with at least 17 months to prepare for model implementation and another 12 months after that before the TEAM participants that participate in Track 1 during PY 1 would potentially be financially accountable for repayment amounts to Medicare. As indicated in section X.A.3.k of the preamble of this final rule, we intend to make TEAM participants' baseline period data available, pursuant to a request and a TEAM data sharing agreement, in advance of the January 1, 2026, model start date. Access to data before the model starts will allow TEAM participants the opportunity to assess their historical performance as they consider changes to their care practices in advance of the model's start date. In addition, TEAM participants may request and receive data from CMS throughout the performance year, pursuant to a TEAM data sharing agreement, that will help them to gauge their performance in the model and identify areas for care improvement that will inform their care redesign practices in all performance years of the model. We also disagree with commenters that CMS underestimated the resources needed to establish the care teams to lead and participate in TEAM. We intentionally chose and limited the episode categories tested in TEAM, as described in section X.A.3.b of the preamble of this final rule, that were common, higher volume procedures. We believe many hospitals already have established standard care pathways and care teams with experience managing beneficiaries who receive these procedures, so we do not expect TEAM to require an overhaul to care practices but to rather encourage TEAM participants to introduce refinements to existing process that will create the efficiencies to improve quality and reduce spending. Therefore, we believe ( print page 69660) the time period from the publication of this final rule until the end of PY 1 will provide sufficient time to explore and implement care redesign approaches before TEAM participants that participate in Track 1 during PY 1 are required to assume downside financial risk.
We acknowledge that other CMS APMs have longer glide paths, such as the Making Care Primary Model, but note that these APMs generally have longer performance periods. As a five-year model, we believe that a one-year glide path is sufficient for most TEAM participants. We do not believe a longer glide path is necessary for TEAM participants that have not participated in the BPCI Advanced and CJR models, because we do not believe their lack of participation in these models is indicative of needing a greater amount of time before assuming downside financial risk. However, we are finalizing some slight changes to our proposal with respect to certain TEAM participants, as discussed in the following comment and response, which may affect hospitals that have not previously participated in these models.
Also, as discussed in section X.A.3.d.(3)(g) of the preamble of this final rule, we have considered commenter's concerns regarding TEAM participant's exposure to significant financial risk, and we are finalizing changes to our proposed discount factor policy—we are reducing that potential financial risk by lowering the discount factor. In addition, as discussed in section X.A.3.d.(3)(h) of the preamble of this final rule, we are not finalizing our policy for low volume hospitals and will propose an updated policy in future notice and comment rulemaking that will address concerns with respect to the level of risk these TEAM participants may have. We believe that these changes will both facilitate participants' abilities to be successful under this model and allow for a more gradual transition to full financial responsibility under the model.
Comment: Numerous commenters indicated that CMS did not provide adequate safeguards to protect safety net and rural hospitals due to their lack of resources caring for a more complex population with greater health-related social needs and requested that CMS allow these hospitals to remain in Track 1 for the entirety of the model. Numerous commenters cited safety net hospitals operating on negative margins with insufficient resources to participate in a model like TEAM, creating an even greater risk to access to care for populations that have historically suffered inequitable outcomes. Many commenters had concerns with CMS' oversampling CBSAs with safety net hospitals indicating the added administrative demands and financial risk of TEAM may have unintended consequences of placing further strain on systems serving a high proportion of patients with significant social need. Other commenters requested that CMS allow Sole Community Hospitals and Medicare Dependent Hospitals to participate in Track 1 for the entirety of the model. A couple of commenters recommended CMS develop meaningful readiness metrics that would allow these safety net hospitals to better understand their performance and give them adequate time for internal process improvement projects before being held accountable for these episodes of care.
Response: We appreciate and are persuaded by comments expressing concerns that our proposed policies for certain types of hospitals need to be modified. We recognize the importance of including safety net hospitals in TEAM, providing them and the beneficiaries they care for, access to the benefits of value-based care though improving care coordination, avoiding duplicative or unnecessary services, and improving the beneficiary care experience during care transitions. While CMS has tested episode-based payment models for over a decade, we are aware that safety net hospitals have been underrepresented in our previous and current episode-based payment models and believe that oversampling them in TEAM will allow them to gain experience and help them become less dependent on traditional FFS payments. Even though many hospitals may have some experience with episode-based payments, with Medicare or with beneficiaries covered by commercial insurance, we acknowledge that safety net hospitals may have significant financial barriers that have limited their exposure to episode-based payments or have different care priorities given the beneficiary population they care for. Accordingly, we are finalizing our proposed Track 1 policies with slight changes. We are lengthening Track 1 for safety net hospitals, as defined and finalized in section X.A.3.f of the preamble of this final rule, so they will be eligible to remain in Track 1 for PYs 1 through 3 of the model, if they so choose. The election and notification process remains unchanged, meaning TEAM participants that are safety net hospitals that wish to participate in Track 1 for PYs 2 and 3 must notify CMS of their Track selection prior to each performance year in a form and manner, and by a date specified by CMS. The TEAM participant may switch between tracks, however, if the TEAM participant fails to timely notify CMS of their election to participate in Track 1 or Track 2, the TEAM participant will be assigned to Track 3 for the performance year they were requesting Track1 or Track 2 participation. We believe that allowing safety net hospitals to have the option of an additional two years of participating in Track 1 beyond PY 1, if they so choose, will provide them with a more meaningful opportunity to gain experience in the model and make investments into care redesign processes. We note the definitions of safety net hospital and other hospital types, such as rural hospital, are not mutually exclusive. Therefore, if any TEAM participant meets the definition of safety net hospital, then they may participate in Track 1 for PYs 1 through 3. We still believe that all TEAM participants should assume double-sided risk during the performance period of the model, and thus we are not allowing safety net providers to remain in Track 1 for the entirety of the model. We will monitor safety net hospital performance in the model and, if warranted, will propose updated policies in future notice and comment rulemaking.
We also recognize that TEAM participants in Track 2, which includes rural hospitals, as defined and finalized in section X.A.3.f of the preamble of this final rule, and other types of hospitals may need additional safeguards to limit their financial risk in TEAM. We refer readers to section X.A.3.d.(5)(h) of the preamble of this final rule, where we discuss modifications to the stop-gain and stop-loss limits for TEAM participants in Track 2.
In addition, as discussed in section X.A.3.d.(3)(h) of the preamble of this final rule, we are not finalizing our policy for low volume hospitals and will propose an updated policy in future notice and comment rulemaking that will address the level of risk these TEAM participants may have. We believe that a future low volume hospital policy, paired with the changes for Track 1 for safety net hospitals and the stop-gain and stop-loss limit changes for Track 2, will help facilitate TEAM participants' abilities to be successful under this model and allow for a more gradual transition to full financial responsibility under the model.
Lastly, we appreciate the commenters recommendation to develop readiness metrics to help hospitals understand their performance in the model. We may take this recommendation into ( print page 69661) consideration as we develop learning resources and supports that may help TEAM participants achieve success in the model.
Table X.A.-02 summarizes the final TEAM participation tracks based on the modifications made to Track 1 and Track 2.

Comment: A commenter suggested allowing voluntary participation in Track 3 to encourage more hospitals to continue investing in value-based care programs and episodic payment models.
Response: We thank the commenter for the suggestion but do not believe making Track 3 voluntary would yield sufficient evaluation data or offer a sustainable policy.
Comment: A commenter was concerned that the terminology of “participation tracks” may be misleading since Track 1 is available only in PY 1.
Response: We disagree that the term participation track is misleading as it represents a unique pathway with different participation parameters in TEAM. We believe it would be more confusing to not differentiate the time-limited glide path of Track 1, especially given the different eligibility requirements and time lengths for each track. We note that we are finalizing a longer time period for Track 1, specifically allowing TEAM participants who meet the definition of safety net hospitals to participate in Track 1 for PYs 1 through 3.
Comment: A commenter recommended providing advance investment payments to participants in rural or underserved areas and not recoup these payments and allow them to remain in upside-only track for the duration of TEAM.
Response: We thank the commenter for the suggestion. We may consider infrastructure payments for TEAM participants in future notice and comment rulemaking. As previously noted, we are modifying financial risk thresholds, specifically we are modifying the stop-gain and stop-loss limits from 10 percent to 5 percent for TEAM participants eligible to participate in Track 2, as discussed in section X.A.3.d.(5)(h) of the preamble of the final rule. Further, we are also modifying Track 1 policies for participants who satisfy the safety net hospital definition, as defined in section X.A.3.f of the preamble of this final rule, which will allow them to remain eligible to participate in Track 1 for PYs 1 through 3 of the model.
Comment: A commenter recommended we establish different participation tracks for inpatient and outpatient episodes because procedures performed in the inpatient setting are becoming more complex and costly, with greater use of skilled nursing facility services following surgery.
Response: We thank the commenter for this suggestion, but we do not believe creating different participation tracks based on hospital setting is needed and may cause unnecessary confusion given the three participation tracks proposed. Target prices, as described in section X.A.3.d of the preamble of this final rule, will account for episodes that are initiated in these different settings and will include beneficiary-level risk adjustment to adjust for patient acuity.
Comment: A commenter noted that hospitals will have meaningful downside risk in any of the proposed tracks including Track 1 due to operating and reporting expenses that may exceed reconciliation payment amounts.
Response: We indicated in the proposed rule ( 89 FR 36392 ) that Track 1 would have upside-only financial risk provided through reconciliation payments, thus TEAM participants would not be subject to downside risk through a repayment amount in Track 1. We acknowledge that some TEAM participants may make significant investments into their care redesign processes that include financial resources to implement. However, we believe TEAM participants may be able to achieve success by making refinements to their existing care processes and we aim to minimize reporting burden by making health equity plans voluntary in the first performance year and using quality measures hospitals are already required to report on for other CMS quality reporting programs as the required quality measures in TEAM.
Comment: A commenter indicated there are likely many hospitals that would be able to take on downside risk in the first participation year but will not be allowed to do so and disagreed with limiting the option for downside risk in the TEAM program.
Response: CMS recognizes the importance of allowing a TEAM ( print page 69662) participant the autonomy and flexibility to decide what participation track they would like to participate in for the first performance year of the model. As indicated in the proposed rule ( 89 FR 36392 ), we proposed allowing all TEAM participants to select between one of two tracks for the first performance year of TEAM. Therefore, TEAM participants who wish to take on downside risk in the first performance year may do so, by notifying CMS of their election, in a form and manner and by a date specified by CMS. As proposed, TEAM participants may choose to participate in either Track 1 or Track 3 during PY 1.
After consideration of the public comments we received, we are finalizing the participation track proposals with slight modifications. First, safety net hospitals will be eligible to participate in Track 1 for PYs 1 through 3 of the model. Accordingly, we are modifying the regulatory text definition of Track 1 at § 512.505 to specify that TEAM participants who satisfy the definition of safety net hospitals may participate in this track for PYs 1 through 3 of the model. We are also modifying the regulatory text at § 512.520 to specify TEAM participants who satisfy the definition of safety net hospital have the ability to request participation in Track 1 for PYs 1 through 3. We are modifying the regulatory text at § 512.550 (3)(3)(i) to eliminate the reference to PY 1 so that TEAM participants in Track 1 will not owe a repayment amount.
The participant selection methodology that we proposed for TEAM was designed to provide adequate statistical power for evaluating and detecting changes in cost and quality.
We proposed that TEAM would be an episode-based payment model implemented at the hospital level that captures all items and services furnished to a beneficiary over a defined period of time. We proposed to test five episode categories in TEAM, as described in section X.A.3.b. of the preamble of this final rule, focusing on acute clinical procedures initiated in the hospital inpatient and outpatient settings. Specifically, we proposed to test episodes that begin with CABG, LEJR, major bowel procedure, SHFFT, and spinal fusion. We considered whether the model should be limited to hospitals where a high volume of the proposed five episode categories are performed, which would result in a more narrow test on the effects of an episode-based payment approach, or whether to include all hospitals in particular geographic areas, which would result in testing the effects of an episode-based payment approach more broadly across an accountable care community seeking to coordinate care longitudinally across settings. We noted in the proposed rule that selecting only those hospitals where a high volume of the proposed episode categories is performed may result in fewer hospitals being selected as TEAM participants but could still result in a sufficient number of episodes to evaluate the success of the model. We noted in the proposed rule that there would be more potential for behaviors that could impact the model test, such as patient shifting and steering between hospitals in a given geographic area ( 89 FR 36394 ).
We proposed to select geographic areas and require all hospitals, as defined in section X.A.3.a.(2).(b). of the preamble of this final rule, in those selected areas to participate in TEAM to help minimize the risk of TEAM participants shifting higher cost cases to hospitals not participating in TEAM. We proposed that, instead of taking a simple random sampling where all geographic areas have the same chance for selection, we would group these geographic areas according to certain characteristics and then randomly select geographic areas from within those groups, also known as strata, for model implementation. Such a stratified random sampling method based on geographic area would provide several benefits. We stated in the proposed rule that we expected that this method would allow us to observe the experiences of hospitals in geographic areas with various characteristics, such as variations in the number of hospitals, average episode spending, number of hospitals that serve a higher proportion of historically underserved beneficiaries, and differing experience with previous CMS bundled payment models. We noted that we could then examine whether these characteristics impact the effect of the model on patient outcomes and Medicare expenditures within episodes of care. Using a stratified random sampling based on geographic area would also substantially reduce the extent to which the selected hospitals would differ from other hospitals on the characteristics used for stratification, compared to a simple random sample. Simple randomization may ensure similarity between the selected hospitals and hospitals that are not selected, but simple randomization can also lead to differences if enough units are drawn in a group-randomized design where the number of available groups is relatively small. Finally, we stated in the proposed rule that using a stratified random sampling of geographic areas would improve the statistical power of the subsequent model evaluation and improve our ability to reach conclusions about the model's effects on episode spending and the quality of patient care. Section 1115A(a)(5) of the Act allows the Secretary to limit the testing of a model to certain geographic areas, and we proposed for the reasons stated above to use a stratified random sampling method to select geographic areas and require all hospitals within those selected geographic areas to participate in TEAM.
We considered using a stratified random sampling methodology to select the following geographic areas: (1) certain counties based on their CBSAs; (2) certain ZIP codes based on their Hospital Referral Regions (HRR); or (3) certain states. We address each geographic unit in turn.
We considered selecting certain counties based on their CBSA. CBSA includes a core area with a substantial portion of the population in adjacent communities having a high degree of economic and social integration with that core. A county is designated as part of a CBSA when the county is associated with at least one core (urbanized area or urban cluster) with a population of at least 10,000, with the adjacent counties having a high degree of social and economic integration with the core as measured through commuting ties with the other counties associated with the core.
OMB Bulletin 23-01, issued on July 21, 2023, states that there are 935 CBSAs in the United States and Puerto Rico. The 935 CBSAs include 393 Metropolitan Statistical Areas (MSAs), which have an urban core population of at least 50,000, and 542 Micropolitan Statistical Areas (mSAs), which have an urban core population of at least 10,000 but less than 50,000. CBSAs may be further combined into a Combined Statistical Area (CSA) which consists of two or more adjacent CBSAs (including MSAs, mSAs, or both) with substantial employment interchange. Counties not classified as a CBSA are typically categorized and examined at a state level.
The choices for a geographical unit based on CBSA include a CBSA, an MSA, or a CSA. We proposed to select CBSAs in this model, which we will discuss later in this section. We note ( print page 69663) that CJR, a previous mandatory episode-based payment model, utilized MSAs as the geographic unit. Under TEAM, we proposed to expand upon the CJR model's representation of geographic units by also including smaller geographic units, mSAs, in addition to MSAs. We proposed that counties and other areas not located in a CBSA would not be included in the TEAM selection method.
We considered, but ultimately decided against, using CSAs instead of CBSAs as the geographic unit of selection. Under this scenario, we would look at how OMB classifies counties. We would first assess whether a county has been identified as belonging to a CSA, a unit which consists of adjacent CBSAs. If the county was not in a CSA, we would determine if it was in a CBSA that is not part of a larger CSA. Counties not located in a CBSA would be excluded from selection.
We considered a number of factors to decide whether to select geographic areas on the basis of CSAs and CBSAs or just on CBSAs alone, including an assessment of the anticipated degree to which patients who have one of the proposed episode categories would be willing to travel for their initial hospitalization, the extent to which surgeons are expected to have admitting privileges in multiple hospitals located in different CBSAs, and statistical power considerations related to the number of independent geographic units available for selection (there are only 184 CSAs vs. 935 CBSAs). We also considered the risk for patient shifting and steering between CBSAs within a CSA, and we believe that the anticipated risk is not severe enough to warrant selecting CSAs.
We next considered selecting hospital referral regions (HRRs). HRRs represent regional health care markets for tertiary medical care. HRRs are defined by determining where the majority of patients were referred for major cardiovascular surgical procedures and for neurosurgery. There are 306 HRRs with at least one city where both major cardiovascular surgical procedures and neurosurgery are performed. HRRs may not sufficiently reflect referral patterns for the five episode categories we proposed to test in TEAM, as only one of the five proposed episode categories is cardiovascular (coronary artery bypass graft surgery), and this episode category has the smallest procedure volume. Therefore, as stated in the proposed rule, we believe that CBSAs as a geographic unit are preferable over HRRs for this model.
We also considered selecting states as the geographic areas for TEAM. However, we concluded that CBSAs as a geographic unit are preferable over states. Choosing states as the geographic unit would require us to automatically include hospitals in all rural areas within the selected states. Using a unit of selection smaller than a state would allow for a more deliberate choice about the extent of inclusion of rural or small population areas. Selecting states rather than CBSAs would also greatly reduce the number of independent geographic areas subject to selection under the model, which would decrease the statistical power of the model evaluation. Finally, CBSAs straddle state lines where providers and Medicare beneficiaries can easily cross these boundaries for health care. Choosing states as the geographic unit would potentially divide a hospital market and set up a greater potential for patient shifting and steering to different hospitals under the model. CMS decided that the CBSA-level analysis was more analytically appropriate based on the specifics of this model.
For the reasons previously discussed, we proposed to require all hospitals, as defined in section X.A.3.a.(2)(b). of the preamble of this final rule and in proposed § 512.505, within a CBSA that CMS selects through the stratified random sampling methodology, described in section X.A.3.a.(4)(d). of the preamble of this final rule, to participate in TEAM. Although CBSAs are revised periodically, with additional counties added to or removed from certain CBSAs, we proposed to use the CBSA designations in OMB Bulletin 23-01 issued on July 21, 2023, as the CBSA designations for purposes of selecting participants for this model, regardless of whether such CBSA designations have changed since July 21, 2023, or will change at some point during the model performance period. We believe that this approach would best maintain the consistency of the TEAM participants in the model, which is crucial for our ability to evaluate the effects of the model test on quality of care and changes in Medicare spending.
We proposed to exclude from the stratified random sampling of geographic areas any CBSAs that are located entirely in the state of Maryland, and certain CBSAs that straddle Maryland and another state. If a CBSA: (1) includes a portion of Maryland; and (2) more than 50 percent of the episodes that initiated at hospitals within that CBSA between January 1, 2022, and June 30, 2023, for any of the five episode categories proposed for testing in TEAM did so at hospitals in Maryland, that CBSA will also be excluded from TEAM. We proposed to exclude these CBSAs from selection because the state of Maryland is currently participating in another Innovation Center Model—the Maryland Total Cost Ofo Care Model, as further described in section X.A.3.a.(2).(b).(i). of the preamble of this final rule.
We also proposed to exclude CBSAs in which no episodes were initiated at hospitals for any of the five episode categories proposed for testing in TEAM between January 1, 2022, and June 30, 2023. We stated in the proposed rule that we believed it would be highly unlikely for these CBSAs to have data available for evaluation after the model starts. After applying these criteria, 803 CBSAs remain available for selection in TEAM. We proposed to use a stratified random sampling method as described below to select approximately 25 percent of eligible CBSAs in TEAM following the process we describe in the next two sections. We are providing the proposed list of CBSAs eligible for selection in TEAM in Table X.A.-03. [ 881 ]

We proposed to stratify CBSAs into groups based on average historical episode spending, the number of hospitals, the number of safety net hospitals, and the CBSA's exposure to prior CMS bundled payment models.
Stratification enables certain groups of interest to be represented at a higher level, or oversampled, in the model test. One of CMS' policy objectives is to extend the reach of value-based care to more beneficiaries, including beneficiaries from underserved communities. Consistent with that objective, CMS proposed to oversample CBSAs that have limited previous exposure to CMS' bundled payment models and CBSAs with a higher number of safety net hospitals.
We considered stratifying eligible CBSAs into mutually exclusive groups corresponding to the 16 unique combinations of “high” and “low” values for the following four CBSA-level characteristics (based on the median values across all CBSAs):
- Average spend for a broad set of episode categories in the CBSA. There are significant healthcare cost differences across geographic regions. One of the main objectives of TEAM is to reduce episode spending, and the proposed pricing methodology for episodes is regional. Thus, it will be important for the TEAM design to account for the significant variation in average episode spending across geographic regions. We proposed to use the episode categories included in the predecessor bundled payment model, BPCI Advanced, initiated between January 1, 2022, and June 30, 2023, to determine the average spend for a broad set of episode categories for each CBSA. The episode categories are: Acute myocardial infarction; Cardiac arrhythmia; Congestive heart failure; Cardiac defibrillator; Cardiac valve; Coronary artery bypass graft; Endovascular cardiac valve replacement; Pacemaker; Percutaneous coronary intervention; Cardiac defibrillator; Percutaneous coronary intervention; Disorders of liver except malignancy; cirrhosis or alcoholic hepatitis; Gastrointestinal hemorrhage; Gastrointestinal obstruction; Inflammatory bowel disease; Bariatric surgery; Major bowel procedure; Cellulitis; Chronic obstructive pulmonary disease; bronchitis, asthma, Renal failure; Sepsis; Simple pneumonia and respiratory infections; Urinary tract infection; Seizures; Stroke; Double joint replacement of the lower extremity; Fractures of the femur and hip or pelvis; Hip femur procedures except major joint; Lower extremity and humerus procedure except hip, foot, femur; Major joint replacement of the lower extremity; Major joint replacement of the upper extremity; Back neck except spinal fusion; Spinal fusion. [ 882 ]
- Number of hospitals within the CBSA. We proposed to select CBSAs for purposes of model implementation, which include mSA areas in addition to MSAs, meaning that TEAM would be highly representative of the United States and would include many areas with only a single hospital as well as areas with a high number of hospitals. We stated in the proposed rule that we expected significant differences in the healthcare environment and beneficiary characteristics across CBSAs with low and high numbers of hospitals. Consequently, we believe it is important to select areas above and below the median to have broad representation of CBSAs included in the model.
- CBSA's past exposure to CMS' bundled payment models (BPCI Models 2, 3, and 4, CJR, or BPCI Advanced) during the period from October 1, 2013, to December 31, 2022. The extent of previous participation in bundled payment models in a CBSA may be a factor in how successful TEAM participants will be at reducing costs and improving quality of care under the model. We stated in the proposed rule that this stratification will allow CMS to assess how TEAM's impacts vary by past regional exposure to bundled payment models.
- Number of safety net hospitals in the CBSA. Safety net providers have historically not participated in voluntary episode-based payment models as frequently as other providers. Through TEAM, we see an opportunity to improve care for beneficiaries served by safety net providers and want to ensure focus on care redesign and improving quality of care for beneficiaries in underserved communities, consistent with CMS' objectives to improve health equity. Stratifying CBSAs by the number of safety net hospitals will allow CMS to gather robust data to assess TEAM's effects across a range of provider types.
We ultimately decided to create an additional stratum from one of these 16 strata for a total of 17 strata to select CBSAs into TEAM. Below, we identify the stratum we proposed to split into two strata and how we would do that; and describe the reasons for this decision.
We noted in the proposed rule that there are only a handful of outlier CBSAs with a very high number of safety net hospitals. Inclusion of these outlier CBSAs results in an extremely lopsided or asymmetrical distribution when stratifying CBSAs by this characteristic. Depending on the circumstances, these handful of CBSAs may potentially lead to significant ( print page 69683) differences in the total number of safety net hospitals between the mandatory CBSAs that are selected for TEAM and those that are not selected. We therefore proposed to move these CBSAs into a new 17th stratum. Therefore, the proposed stratification process results in 17 mutually exclusive strata of CBSAs.
We proposed to randomly select CBSAs for TEAM from the 17 stratified groups using a method that reflects CMS' policy objectives described above, including expanding the reach of value-based care. We proposed to oversample CBSAs with low past exposure to CMS' bundled payment models and CBSAs with a high number of safety net hospitals. The selection probability for a given CBSA would differ across strata, but all CBSAs within a particular stratum, will have the same chance of being selected. The hospitals located in the selected mandatory CBSAs will be required to participate. We stated in the proposed rule that CMS' proposed method of randomly selecting CBSAs while oversampling CBSAs with certain characteristics would result in the following selection probabilities:
- 33.3 percent of (one out of three) CBSAs will be selected in strata with high number of safety net hospitals and low past exposure to CMS' bundled payment models. Four strata have this selection probability.
- 25 percent of (one out of four) CBSAs will be selected in strata with either high number of safety net hospitals or low past exposure to CMS' bundled payment models (but not both). Eight strata have this selection probability.
- 20 percent of (one out of five) CBSAs will be selected in strata with neither high number of safety net hospitals nor low past exposure to CMS' bundled payment models. Four strata have this selection probability.
- 50 percent of (one out of two) CBSAs will be selected with the highest number of safety net hospitals (One strata has this selection probability: the 17th stratum).
The 17 selection strata and their relationship to the dimensions discussed above are represented in Table X.A.-04.

Through this selection scheme, CMS would select approximately a quarter of eligible CBSAs listed in Table X.A.-04 as the mandatory CBSAs in which TEAM would be implemented. A hospital's probability of being required to participate in TEAM would depend on the stratum their CBSA is in and would range from 20 percent to 50 percent.
We conducted power analyses to identify detectable changes in episode spending between a potential group of mandatory CBSAs selected for the model and a potential control group of CBSAs using a Type I error of 0.05 and Type 2 error of 0.2 (implying a power of 0.8). The analysis shows that, if a quarter of eligible CBSAs are selected for TEAM, we will be able to detect 1.5 percent changes in episode spending, all else being equal. This change in episode spending is within the savings range that CMS might expect to achieve given estimates for surgical episodes from previous episode-based payment models, including BPCI Model 2, CJR, and BPCI Advanced. This is critical to ensuring that CMS can assess the model's impact on Medicare spending.
We sought comment on our proposed approach to selecting TEAM participants at § 512.515.
The following is a summary of comments we received on the proposed approach to selecting TEAM participants and our responses to these comments:
Comment: Numerous commenters noted concerns regarding to oversampling safety-net hospitals. Commenters noted that this would put undue burden on these providers who work in hospitals that care for a high proportion of vulnerable patients. A commenter stated that over-sampling of safety net hospitals will financially harm hospitals who can least afford to take on risk. Another commenter stated that these already-stretched-thin providers may not have the capacity to succeed in this model as proposed. Safety net hospitals may have diminished readiness to bear risk and have fewer resources than non-safety-net hospitals. A couple commenters noted the same factors that increase the cost to deliver care to safety net populations also reduce the likelihood of success in episodic payment models and that these hospitals have not demonstrated significant spending reductions in previous models. A ( print page 69684) commenter suggested eliminating the 17th outlier stratum.
Response: While we recognize the commenters' concerns with requiring participation from safety net hospitals, as defined in section X.A.3.f of the preamble of this final rule, by including all types of hospitals within TEAM CMS will have a better, more accurate picture of the effects of the model for consideration of any potential expansion on a national scale. The stratification approach will allow CMS to assess TEAM across healthcare delivery settings, from high-resource environments to those with limited capabilities. One of CMS' policy objectives is to extend the reach of value-based care to more beneficiaries, including beneficiaries from underserved communities. Determining the impact across diverse markets, hospitals and patient populations is critical for ensuring the design of the payment model is fair and equitable across all types of hospitals. To balance the need for a broad test of TEAM and the concerns about the negative financial impacts on safety-net hospitals, TEAM will provide additional flexibilities to these hospitals as discussed in section X.A.3.a.(3) of the preamble of this final rule.
Comment: A few commenters expressed concerns with the selection approach using CBSAs as the unit of selection. One commenter was worried that this could inadvertently harm academic medical centers because these tertiary care centers often serve patients from outside their CBSA, and patients from outside their region are typically more complex. The commenter suggested that that the difference in where patients reside between academic/referral center/tertiary care centers and community hospitals should be taken into consideration in the model, including benchmarking.
Response: We appreciate the views and expressed concerns of the commenters on our proposal for required participation in the TEAM based on CBSA selection. By using CBSAs as the unit of selection, CMS is ensuring that the model represents a wide range of markets. Additionally, we believe that by requiring the participation of a large number of hospitals with diverse characteristics, TEAM will result in a robust data set for evaluation of this payment approach and will stimulate the rapid development of new evidence-based knowledge. Regarding the comment about differences in patient complexity, the risk adjustment methodology discussed in X.A.3.d.(4) of the final rule explains how patient-level factors are taken into account.
Comment: A couple of commenters outlined concerns for hospitals located in rural areas. These commenters suggested that CMS exclude rural areas and providers located in rural areas from the model or allow these hospitals to voluntarily opt-in because these commenters said that these hospitals may have difficulty in achieving the model aims due to low volume of procedures and a low supply of associated providers. Commenters stated particularly in particular that geographic areas with primary care shortages might encounter difficulties in referring post-surgical patients to appropriate primary care follow-up within the 30-day episode. Commenters said that hospitals that handle very low volumes of the selected episodes do not have the experience or volume to adjust behaviors as envisioned by the proposed model.
Response: We appreciate TEAM and acknowledge commenters' concerns related to the ability of small and rural providers to effectively participate and succeed in the model. The inclusion of hospitals possessing a variety of characteristics is critical to allow CMS to evaluate the impact of the model on a wide range of hospitals in diverse markets. Including hospitals with various volumes of services; different levels of access to financial, community, or other resources; and various levels of population and health provider density, will provide a broad test of the model and generate evidence for consideration of any potential expansion on a national scale.
We acknowledge that providers with low volumes of cases may not find it in their financial interests to make systematic care redesigns or engage in an active way with TEAM. We expect that low volume providers may decide that their resources are better targeted to other efforts because they do not find the financial incentive present in the TEAM sufficiently strong to cause them to shift their practice patterns. We acknowledge that low volume hospitals may achieve less savings because they did not or could not make the necessary changes to the treatment of their qualifying beneficiary population. We believe this choice is similar in nature to that made as hospitals decide their overall business strategies and where to focus their efforts.
Comment: A couple of commenters discussed how prior experience with participation in value-based care could hinder a hospital's ability to be successful in TEAM. Specifically, in CBSAs that include hospitals with a long history of hospital participation in value-based care models, hospitals will have already found efficiencies and they may face undue difficulty finding more efficiencies thus leading to diminishing returns under TEAM. A commenter expressed fears that including these areas will penalize early adopters.
Response: We acknowledge the concerns of the commenters, but we disagree that requiring hospitals located in mandatory CBSAs with a long history of participation in value-based care to participate in TEAM would penalize early adopters. It would be highly unlikely that these historically low-cost hospitals would find regional target prices unachievable. Rather, if all hospitals in a region did not change their behavior, hospitals that are historically low-cost relative to other hospitals in their region and conditional on their episode risk adjusters, would generally be rewarded under the model. We expect that hospitals can build upon already established infrastructure, practices, and procedures to achieve efficiencies under this episode payment model.
Comment: One commenter stated that the selection approach may inadvertently exclude hospitals with particularly high volumes of the surgical procedures proposed for the model and suggested an opportunity for hospitals with high volumes of the surgical procedures to opt-in.
Response: While we acknowledge that some high-volume hospitals not located in a selected mandatory CBSAs may desire to participate in TEAM, maintaining the mandatory, randomized design will allow for a more accurate test of the effects of the model to inform potential expansion on a national scale. CMS designed TEAM based on prior experience with several types of large voluntary episode payment models. TEAM is intended to be a broader test of episode payment models by including hospitals who may not otherwise volunteer for such a model. The participant selection methodology for TEAM was designed to provide adequate statistical power for evaluating and detecting changes in cost and quality and allows us to observe the experiences of hospitals in geographic areas with a broad range of characteristics. The selection approach ensures that important characteristics are balanced across TEAM participants and the control group, ( i.e., eligible hospitals not selected for TEAM). Section X.A.3.a.(2)(c) of the preamble of this final rule discusses the narrow opportunity for select hospitals to opt-in to TEAM if they are not located in a mandatory CBSA. This option has been ( print page 69685) limited to a select group of hospitals currently participating in episode-based payment models to continue care transformation efforts.
Comment: A commenter stated that the proposed sampling methodology will create unnecessary payment complexity and hinder CMS' ability to accurately isolate and evaluate the impacts of the proposed model. They expressed concern that several markets with a high probability of being sampled based on the established methodology also have the highest level of participation in ACOs. The commenter's concern was two-fold (1) they thought this could create an unnecessarily complex payment environment in sampled areas across the country; and (2) they thought it would be particularly difficult for CMS to parse out whether savings are generated by TEAM or other models.
Response: We note that the simultaneous testing of multiple value-based care initiatives is an appropriate strategy in many situations, depending on the care targeted under each model. Section X.A.3.e of the preamble of this final rule lays out our policies for accounting for overlap between models and contains discussion of the potential synergies and improved care coordination we expect will ensue through allowing for hospitals and beneficiaries to be engaged in more than one initiative simultaneously. We see no compelling reason why hospitals participating in ACO initiatives and other efforts cannot be TEAM participants.
Comment: A few commenters had concerns with the model requiring participation for all IPPS hospitals within the mandatory CBSA. A commenter asserted that many hospitals are neither of an adequate size nor in a financial position to support the investments necessary to transition to mandatory bundled payment models. Commenters suggested a variety of alternative options; make TEAM fully voluntary, begin with a period of voluntary participation, allow hospitals the opportunity to opt-in or out based on internal assessment of readiness. A commenter stated that voluntary participation grants entities who have the existing capabilities and resources a window to put in place practices that reduce the risk of unsuccessful financial and quality outcomes. Another commenter suggested adding in a selection variable that would capture CBSA readiness.
Response: We appreciate the concerns of the commenters. The CMS Innovation Center has multiple years of experience with several types of large voluntary episode payment models where we have successfully collaborated with participants on implementation of episode-based payment models in a variety of settings for multiple clinical conditions. Because we believe that it is important to provide an option for hospitals currently participating in episode-based payment models to continue care transformation efforts, Section X.A.3.a.(2)(c) of the preamble discusses an opportunity for select hospitals to opt-in to TEAM if they are not located in a mandatory CBSA. The mandatory, randomized design allows us to observe the experiences of hospitals in geographic areas with various characteristics, such as variation in the number of hospitals, average episode spending, number of hospitals that serve a higher proportion of historically underserved beneficiaries, and differing experience with previous CMS episode-based payment models. The TEAM evaluation could then examine whether these characteristics impact the effect of the model on patient outcomes and Medicare expenditures within episodes of care.
We acknowledge that hospitals will have varying levels of readiness to implement the care redesign activities to be successful in TEAM. As discussed in section X.A.3.a.(3) of the preamble of this final rule, we believe the phasing in of full financial risk by offering participation tracks with differing levels of risk and reward, and our planned efforts to engage with hospitals to help them succeed under this model through the provision of beneficiary-identifiable claims data, pursuant to a request and TEAM data sharing agreement, as discussed in section X.A.3.k of the preamble of this final rule, will aid hospitals to succeed under TEAM.
Comment: A couple commenters shared their concerns regarding of the size of the model and suggested that CMS reduce the percentage of mandatory CBSAs selected from 25 percent to 10-15 percent.
Response: The size of the model was determined based on the ability to have the statistical power to detect significant impacts on payment. We conducted power analyses under TEAM as proposed, and under the TEAM participant definition, discussed in Section X.A.3.a.(2)(b) of the preamble of this final rule, to identify detectable changes in episode spending between a potential group of mandatory CBSAs selected for the model and a potential control group of CBSAs using a Type I error of 0.05 and Type 2 error of 0.2 (implying a power of 0.8). The analysis showed that selecting a quarter of eligible CBSAs for TEAM will enable the detection of 1.5 percent changes in episode spending, all else being equal.
Comment: A commenter suggested TEAM should emphasize the low-risk Track options and choose CBSAs newer to value-based initiatives, then study factors that affect intra-CBSA coordination, such as equity, rurality, and interoperability.
Response: We appreciate comments expressing that it is important to include CBSAs newer to value-based initiates and to evaluate a broad range of factors that may affect the models' impacts. One of CMS' policy objectives is to extend the reach of value-based care to more beneficiaries.
Comment: Many commenters urged significant transparency from CMS by providing the list of selected mandatory CBSAs. They stated that without any information on those mandatory CBSAs selected for participation in TEAM, the public's ability to understand and ultimately articulate potential impacts is compromised. The commenters thought that this transparency is critical not only for fairness and trust but also for enabling systematic evaluation and feedback, which are essential for continuous improvement in public health policy.
Response: We appreciate comments expressing concerns around transparency, fairness, and systematic evaluation. We agree transparency is critical and have provided the list of selected mandatory CBSAs in Table X.A.-07 of the preamble of this final rule to provide sufficient notice between when the final listed of selected mandatory CBSAs is published and when the model starts.
After consideration of the public comments we received, we are finalizing our proposal for the approach to selecting mandatory CBSAs and TEAM Participants with modifications.
As finalized in section X.A.3.a.(2)(c) of the preamble of this final rule, we are finalizing a voluntary opt-in option for BPCI Advanced and CJR model participants that participate in those models until the last day of the last performance period or last performance year of their respective model. [ 883 ] However, we recognize that allowing voluntary opt-in may cause self-selection effects and limit the rigor of randomization, and we are unsure of extent of those effects at this time.
To mitigate such adverse effects of self-selection on evaluation rigor, we are finalizing a slightly modified selection ( print page 69686) approach to isolate potential bias introduced by the potential BPCI Advanced and CJR participants that voluntarily opt to participate in TEAM. In the first 16 strata, we will move the CBSAs that contain at least one hospital participating in BPCI Advanced or CJR model as of January 1, 2024, and CBSAs that are located in a state that is participating in AHEAD states except Maryland, finalized in this preamble section X.A.3.a.(2), to a new stratum—stratum 18. As a result, CMS is finalizing a stratification policy that moves 93 CBSAs from strata 1-16 to the 18th stratum. We are finalizing stratum 17 as proposed, without any changes, as this stratum already includes five CBSAs, in addition to the 93 CBSAs described above, that contain a hospital participating in BPCI Advanced or CJR as of January 1, 2024. Other than the creation of the 18th stratum, we are generally finalizing the proposed eligibility criteria for CBSAs in TEAM as proposed. Thus, there are still 803 CBSAs in the country which include 2,718 IPPS hospitals eligible for selection.
As shown in Table X.A.-06, we are finalizing a policy to assign CBSAs to one of 18 strata. As proposed, CMS is assigning CBSAs to one of 16 strata on the basis of high or low values (based on median values) of four CBSA characteristics: past exposure to CMS' bundled payment models (BPCI Models 2, 3, and 4, CJR, or BPCI Advanced) in the CBSA, the number of safety net hospitals, the number of hospitals, and the average cost of care for surgical and medical episodes in the BPCI Advanced model. As proposed, CMS is assigning six CBSAs with exceptionally large numbers of hospitals and safety net hospitals to stratum 17. Designating a stratum for these unique CBSAs helps selecting control CBSAs that are comparable to TEAM CBSAs, which is critical for unbiased evaluation results. Finally, CMS is moving the 93 CBSAs in strata 1-16 CBSAs that contain at least one hospital that meet the criteria related to BPCI Advanced, CJR model participation, or AHEAD participation, as finalized in section X.A.3.a.(2) of the preamble of this final rule, to the newly created stratum 18.
Among strata 1-16, strata with high past exposure to BPCI Advanced and low counts of safety net hospitals were assigned to TEAM with a probability of 20 percent. Strata with low past exposure to CMS' bundled payment models and high counts of safety net hospitals were assigned to TEAM with a probability of 33 percent. CBSAs with either low past exposure to CMS' bundled payment models or high counts of safety net hospitals (but not both) were assigned to TEAM with a probability of 25 percent. CBSAs in stratum 17 were assigned to TEAM with a probability of 50 percent. Finally, CBSAs in stratum 18 were assigned to TEAM with a probability of 20 percent, the lowest probability of selection from strata 1-16. This ensures that despite the addition of stratum 18, no hospital would have a higher probability of selection than they would have had CMS selected mandatory CBSAs exactly as described in the proposed rule. See Table X.A.-05 for list of CBSAs and their final assigned strata.

CMS randomized the CBSAs as described in this rule and selected 188 CBSAs for TEAM (23.4 percent of 803 CBSAs). CMS has conducted an analysis and found that CBSAs that were randomly selected into TEAM are comparable to those that were not selected for TEAM (that is, the control group), which is essential for a rigorous evaluation of TEAM. See Table X.A.-06 for finalized selection strata and selection probabilities. See Table X.A.-07 for list of mandatory CBSAs selected for TEAM.

In the proposed rule, we stated that a key model design feature for episode-based payment models is the definition of the episodes included in the model. We stated that the episode definition has two significant dimensions—(1) a clinical dimension that describes which clinical conditions and associated services are included in the episode; and (2) a time dimension that describes the beginning and end of the episode, its length, and when the episode may be cancelled prior to the end of the episode ( 89 FR 36412 ).
In the proposed rule, we stated that in selecting episodes to test in TEAM, we considered a variety of factors, including the number and type of episodes best suited to meet the goals of the model ( 89 FR 36413 ). We chose to limit the selection of episode categories for TEAM to those that were included in BPCI Advanced through a robust selection process similar to that used for the CJR model ( 80 FR 73277 ). These episode categories represent high-expenditure, high-volume care delivered to Medicare beneficiaries and are evaluable in an episode-based payment model. BPCI Advanced clinical episodes include both surgical episodes, which are triggered by a surgical procedure, and medical episodes that are primarily non-surgical in nature.
In the proposed rule, we stated that while we continue to strive for our models to reduce Medicare expenditures and improve quality of care, we also want to ensure that there is a potential for participating hospitals to succeed. We want the conditions captured by episode categories in TEAM to be clinically similar enough that participants could drive care improvements by streamlining care pathways and transitions between clinical settings. In general, elective surgical procedures are associated with greater clinical homogeneity than unplanned hospitalizations or medical conditions. In addition, when episodes are clinically similar, episode spending is more predictable. Unsurprisingly, medical episodes are associated with greater spending variability. Medical episodes may also be more difficult to manage for hospitals without previous experience implementing value-based care and care redesign activities.
In the proposed rule, we noted that evaluations of CJR and BPCI Advanced suggest that surgical episode categories do not capture underserved populations to the same degree as medical episodes and that medical episodes may offer relatively greater opportunity to address health equity. Specifically, medical episodes generally have a higher proportion of dual-eligible beneficiaries when compared to surgical episodes. TEAM will test novel ways to improve representation of underserved populations in surgical episodes through targeted flexibilities for safety net hospitals (discussed in section X.A.3.a.(3) of this final rule) and more broadly defined beneficiary-level social risk adjustment (described in section X.A.3.f. of this final rule).
In the proposed rule, we stated that we also selected episodes for this proposed model with a greater proportion of spending in the post-acute period relative to the anchor hospitalization or procedure, as such episodes may reflect a greater opportunity to improve care transitions for beneficiaries and reduce unnecessary hospitalizations and emergency care.
Finally, we acknowledged that testing all 34 BPCI Advanced episodes in a novel mandatory model could overwhelm participants.
For the reasons discussed previously in the proposed rule, we proposed testing five surgical episodes in the model—Coronary Artery Bypass Graft Surgery (CABG), Lower Extremity Joint Replacement (LEJR), Surgical Hip and Femur Fracture Treatment (SHFFT), Spinal Fusion, and Major Bowel Procedure. We stated in the proposed rule that based on our experience with the BPCI Advanced and CJR models and the stakeholder feedback received in response to the July 2023 Episode-Based Payment Model Request for Information ( 88 FR 45872 ), we believe that beginning the model with these five episode categories is the most reasonable course for TEAM. Specifically, we proposed to test surgical episodes because they are time-limited with well-defined triggers, have clinically similar patient populations with common care pathways, and have sufficient spending or quality variability, particularly in the post-acute period, to offer participants the opportunity for improvement ( 89 FR 36413 ).
We stated in the proposed rule that the proposed episodes have been previously tested in BPCI Advanced voluntarily, allowing CMS to assess engagement and gather data. The proposed episodes represent the highest volume and highest cost surgical episodes performed in the inpatient setting. Although CABG and SHFFT episodes were finalized in the Advancing Care Coordination through Episode Payment Models (Cardiac and Orthopedic Bundled Payment Models) Final Rule (CMS-5519-F) on December 20, 2016, that mandatory test was not implemented. The proposed TEAM is the next logical step for applying lessons learned from BPCI Advanced in a mandatory model. TEAM would enable CMS to capture a more diverse population of providers, and potentially beneficiaries.
Regarding our proposed episodes, we direct readers to certain changes that were included in the correction notice for the proposed rule (CMS-1808-CN) published May 31, 2024, which addressed inadvertent errors in this Proposed Episodes section of the proposed rule.
First, we added language to reflect the proposed changes to the spinal fusion Medicare Severity Diagnosis Related Groups (MS-DRGs) outlined in the proposed rule ( 89 FR 35971 ). We stated that TEAM would conform to the new spinal fusion MS-DRGs, if finalized, for the purposes of identifying and defining spinal fusion episodes that would be included in the model. The spinal fusion MS-DRG changes that would directly impact the TEAM Spinal Fusion episode category are also discussed in Section X.A.3.b.(4)(d) and X.A.3.b.(5)(c) of this final rule. ( print page 69711)
Second, in the correction notice, we clarified that the Medicare FFS claims data referenced in the discussion of BPCI Advanced episodes in the proposed rule was limited to BPCI Advanced episodes, not all Medicare FFS claims ( 89 FR 36413 ). While we did not explicitly restate that we were discussing historical BPCI Advanced episodes, we expected this would address any confusion between the estimated volumes in this section of the final rule and those included in section X.A.3.b.(4) for TEAM episodes. Specifically, the estimated BPCI Advanced episode volumes are based on final episode counts, which reflect BPCI Advanced episode-level exclusions and overlap policies. That is, episodes that were excluded at any point during the 0-90 days following discharge from an anchor hospitalization or anchor procedure would not have been counted as a BPCI Advanced episode and would lower episode counts.
Finally, we updated out of date hyperlinks to the ICD-10-CM/PCS MS-DRG Definitions Manual in the episode category footnotes throughout the Overview of Proposed Episodes section of the proposed rule ( 89 FR 36413 ). They have been updated again since the release of the correction notice to account for the release of Version 42 of the ICD-10 MS-DRG Definitions Manual that will be published with this final rule.
In the proposed rule, we stated that the proposed Lower Extremity Joint Replacement (LEJR) episode category would include hip, knee, and ankle replacements performed in either the hospital inpatient or outpatient setting. This episode category was selected because, using 2021 BPCI Advanced episode data, it was the highest volume, highest cost BPCI Advanced surgical episode category. There were 204,160 episodes with a total cost of $5.01 billion, with more than 40 percent of spending occurring in the post-acute period.
In the proposed rule, we stated that the proposed SHFFT episode category, referred to as Hip and Femur Procedures except Major Joint in BPCI Advanced, would include beneficiaries who receive a hip fixation procedure in the presence of a hip fracture. It would not include fractures treated with a joint replacement. This episode was selected because it was the second highest volume, and second-highest cost BPCI Advanced surgical episode performed in the inpatient setting, using 2021 BPCI Advanced episode data. There were 69,076 episodes with a total cost of $3.22 billion, with more than 63 percent of spending occurring in the post-acute period.
In the proposed rule, we stated that the proposed CABG episode category would include beneficiaries undergoing coronary revascularization by CABG surgery. [ 884 ] This episode was selected to maintain the engagement of cardiac surgeons who have participated in prior episode-based models. Among cardiac procedures it was the second highest cost and second highest volume BPCI Advanced surgical episode performed in the inpatient setting using 2021 BPCI Advanced episode data. There were 26,259 episodes with a total cost of $1.39 billion; approximately 22 percent of spending occurred in the post-acute period. In the proposed rule we stated we also considered percutaneous coronary intervention (PCI) for TEAM because it was the highest volume and highest cost surgical cardiac episode. However, we did not propose a PCI episode because PCI has been described as a low-value service by the Medicare Payment Advisory Commission when performed for stable coronary artery disease, [ 885 ] and the majority of PCIs are performed in the outpatient setting and are not associated with an acute event.
In the proposed rule, we stated that the proposed Spinal Fusion episode category would include beneficiaries who undergo certain spinal fusion procedures in either a hospital inpatient or outpatient setting. This episode was selected because it was the third-highest cost BPCI Advanced surgical episode performed in the inpatient setting using 2021 BPCI Advanced episode data. There were 62,345 episodes with a total cost of $3.2 billion; more than 27 percent of spending occurred in the post-acute period.
In the proposed rule, we stated that the proposed Major Bowel Procedure episode category would include beneficiaries who undergo a major small or large bowel surgery. [ 886 ] This episode was selected because it was the fifth-highest volume and fourth-highest cost BPCI Advanced surgical episode performed in the inpatient setting using 2021 BPCI Advanced episode data. There were 54,848 episodes with a total cost of $1.95 billion; 37 percent of spending occurred in the post-acute period ( 89 FR 36414 ).
In the proposed rule, we stated that each of the episodes provides different opportunities for TEAM to improve the coordination and quality of care, as well as efficiency of care during the episode, based on varying current patterns of utilization and Medicare spending. While these episode categories have been tested previously, we believe TEAM will provide additional information that can be used for potential expansion through its greater focus on care transitions back to primary care, health equity, and refined payment methodology, as described in section X.A.3.d. of the preamble of this final rule.
In the proposed rule, we stated that the mandatory nature of TEAM would address selection bias, where high performing hospitals have elected to voluntarily participate in a model but then withdrew from the model in the face of financial losses or uncertainty of receiving financial rewards. In BPCI Advanced, participants were able to select clinical episode categories and, later, service lines, which further ensured selection bias.
In the proposed rule, we stated we performed an analysis of BPCI Advanced episode claims data, beginning in CY 2021, to estimate the average annual number of historical episodes that extended 30 days post-hospital discharge, and, therefore, would have been included in TEAM. Based on that analysis, after applying all BPCI Advanced episode-level exclusion and overlap policies, we anticipate the number of BPCI Advanced episodes that TEAM would capture to be approximately 28,088 for CABG; 75,254 for SHFFT; 59,983 for Major Bowel Procedure; 215,957 for LEJR; and 65,968 for Spinal Fusion. The average episode cost for these historical episodes was approximately $48,905 for CABG, $35,501 for SHFFT, $29,184 for Major Bowel Procedure, $21,063 for LEJR, and $46,326 for Spinal Fusion.
As previously stated in the proposed rule, we proposed five episode categories for TEAM to ease TEAM participants into episode accountability. We stated in the proposed rule that we intend to add additional episode categories in future performance years of the model, offering a gradual transition to greater episode accountability, and ultimately to capture a larger proportion of FFS spending in value-based care. We also solicited input on which medical episodes we should consider for future years of the model. Finally, we stated that any additional episodes would be ( print page 69712) added to TEAM pursuant to notice and comment rulemaking ( 89 FR 36413 ).
We sought comment on the five proposed episode categories, described at § 512.525(d). We also solicited input on additional episode categories, including medical episode categories, we should consider for the model.
The following is a summary of the public comments received on the proposed episode categories, potential additional episode categories, and our responses to those comments. Here, we have included comments related to the number and type of proposed episodes. We have separately summarized and responded to comments regarding the five specific proposed episode categories below in Section X.A.3.b.(4) of this final rule, which covers Episode Category Definitions.
Comment: Several commenters supported the decision to include five episodes in TEAM. One commenter thanked CMS for selecting a smaller panel of clinical episodes while representing multiple service lines to maximize clinical value and physician engagement. Many commenters supported including acute, surgical episodes, finding them appropriate for episode-based models, in part, because procedures are straightforward clinical triggers with defined and well-established care practices and protocols. A commenter believed participants will have an easier time drawing clinical pathways, monitoring cost, finding efficiency, and scoring performance for surgical episodes. Another commenter stated that the selected episodes are good choices given the overall goals of the demonstration.
Response: We thank commenters for supporting the inclusion of five surgical clinical episode categories in TEAM.
Comment: Many commenters thought five episodes was too many for a mandatory model because many health care systems and facilities will be new to the concept of bundled payments. Some commenters suggested CMS commence the demonstration with fewer covered conditions and gradually add episodes in future model years, to allow for the education and process changes that will be necessary for each clinical episode category. Several commenters stated that, given the limited resources available, five episodes will overwhelm many hospitals, create unnecessary burden, and be too disruptive.
Response: We do not believe that five episodes is too many for a mandatory model as we see TEAM as a model designed using lessons learned and experiences gained in prior models, including CJR. We see TEAM has a logical step forward in expanding our testing of mandatory episodes of care.
However, we acknowledge that there will likely be TEAM participants with limited experience with the concept of bundled payments or managing five clinical episodes at one time. For this reason, we proposed a glide path to two-sided risk for TEAM participants to ensure that TEAM participants have time to prepare for two-sided financial risk. This is intended to support a TEAM participant's transition into the model and to allow participants time for education and process changes necessary for each category. Please refer to section X.A.2.d of this rule for additional details on TEAM participant risk tracks.
Comment: Many commenters believed participants should have the option to voluntarily select individual clinical episodes, for which there are opportunities to improve patient outcomes for the specific populations served by each hospital, as opposed to requiring participants to take on risk for large, clinically diverse bundles of episodes. A commenter suggested that CMS adopt episodes that are more closely related, which would enable hospitals to engage common medical specialties and care teams. Several commenters stated that because the episodes selected are dissimilar, they will require different workflows, processes, and specialist engagement, which would equate to not one mandated program but at least five individual programs. A couple of commenters stated that voluntary selection would also allow hospitals with limited resources, such as safety net hospitals, to participate without over-extending staff or expending resources on episodes for which it has marginal volume.
Response: We appreciate that commenters would like to voluntarily select clinical episodes in TEAM. However, we believe that testing all the proposed episode categories in TEAM will enable CMS to test how participants perform in clinical areas that they would otherwise not select. Allowing participants to voluntarily choose episode categories would introduce selection bias, make evaluating TEAM more difficult, and produce less generalizable findings. We expect that TEAM will produce data that are more broadly representative of spending, quality, and outcomes than what might be collected under a voluntary model.
Comment: Some commenters suggested that CMS share additional information with respect to the criteria, clinical or administrative, that CMS used so stakeholders may better understand how CMS chose the clinical episodes. A commenter recommended that CMS develop a process for adding any new clinical episodes to TEAM, detailing exactly what criteria are utilized in considering new episodes and including a standardized and lengthy notice and comment period. Another recommended CMS work with stakeholders, including clinicians, to model and design future episodes.
Response: We appreciate these comments and the desire from commenters to have a better understanding of the criteria used and analyses undertaken to identify and select clinical episodes. We respect that stakeholders want to understand the details used to determine additional clinical episodes to add to TEAM in future years, especially based on the mandatory nature of the model. For a discussion on the criteria and analyses used to select the episode categories for TEAM, we direct readers to section X.A.3.b.(2) of this final rule and page 89 FR 36413 of the proposed rule. With respect to developing a process for adding episode categories and working with stakeholders, we stated above in section X.A.3.b.(2) of this final rule and, previously in the proposed rule ( 89 FR 36413 ), that any additional episode categories would be added pursuant to notice and comment rulemaking. We encourage the public, including clinicians, to submit comments in response to any future TEAM rulemaking. CMS will take into consideration any feedback received should additional clinical episode categories be considered for TEAM in future years. Additional episode categories would be proposed through future notice and comment rulemaking.
Comment: A few commenters raised the need for the model to be inclusive of services and supports that address conditions, such as obesity and osteoporosis, which may precipitate or exacerbate clinical episodes in TEAM. A commenter stated that a broad team of health professionals is essential for coordination purposes and to deliver complete and comprehensive care. Another commenter highlighted the need for comprehensive healthcare as a means to improve long-term costs and overall health outcomes. Another commenter believed TEAM would penalize facilities for the added cost of performing even a cursory inquiry into the underlying causes of bone fragility.
Response: We appreciate these comments and acknowledge the importance of including services for conditions that may precipitate or exacerbate clinical episodes in TEAM. ( print page 69713) Because of this, we proposed to only exclude from episodes certain Part A and B items and services that are clinically unrelated to the anchor hospitalization or anchor procedure, thus ensuring the episode captures all relevant items and services rendered ( 89 FR 36416 ).
Although we recognize that some underlying conditions may require services that would not be captured in the TEAM episode because they occur outside of the episode window, we encourage TEAM participants to ensure they are accurately documenting underlying conditions upon admission for anchor hospitalization or anchor procedure. This information could prove vital in determining the best course of care for the patient during their anchor stay or anchor procedure and after discharge.
Comment: Many commenters provided feedback with respect to the potential inclusion of additional surgical and medical episodes CMS might consider for later years of the model. A commenter stated that CMS should focus on episodes that already have a proven track record in voluntary models. Another commenter stated CMS should select episodes that are high volume and have variability in costs, so participants have meaningful opportunities to participate and achieve success under the model. Many commenters supported additional acute episodes. Many commenters urged CMS not to add new episodes during the model performance period so as not to introduce additional burden onto hospitals. A few commenters stated that, once a model begins, it can be challenging to change workflows and processes if the requirements suddenly change or expand, particularly when health systems are already struggling to meet growing Inpatient Quality Reporting (IQR) requirements. A commenter felt that if CMS ultimately ends up adding additional services during the model, the episodes should be optional for participants.
Response: We appreciate these comments on considerations for future clinical episodes CMS may consider for later years of the model. Many factors would play into the potential introduction of new and additional episodes in TEAM and all episodes would be vetted internally within CMS with analysis to ensure there is sufficient episode volume and opportunity to make the episode worth incorporating. CMS may consider bringing in additional expertise to help guide analysis and decision-making regarding potential new episodes, as well. Additionally, any new episodes would be introduced to TEAM through notice and comment rulemaking so the public would be able to share their opinions and thoughts during the comment period.
Comment: Many commenters urged CMS to consider stratifying emergent and other non-elective episodes because of the substantial cost difference in those procedures that are done on a scheduled basis, compared to those that are done on an emergent basis. A commenter believed only elective surgeries should be included in the model. A commenter noted that elective episodes provide patients and caregivers ample time to prepare for post-hospitalization care, whereas emergent cases do not. Several commenters believe that stratifying by elective status creates targets that are more equitable across hospital types. A commenter stated that hospitals with trauma centers will be disadvantaged whereas community hospitals will be advantaged by a target price that blends in the higher cost of emergent episodes that they do not perform and recommended that CMS resolve this design vulnerability by segmenting all episode types by the presence of a trauma diagnosis code, fracture diagnosis code, or an inpatient charge with an ER related revenue code.
Response: We acknowledge that emergent procedures can be relatively more expensive and complex than elective procedures. In light of the comments received, we are modifying the proposed risk adjustment model to include additional clinically relevant risk adjusters. We refer the readers to section X.A.3.d.(4) of this final rule for the comprehensive list of risk adjustment variables, including individual HCCs, that will be included in TEAM.
We appreciate the commenters' suggestions on the target price methodology for episodes initiated on an emergent basis. We believe that grouping emergent and elective procedures together, rather than stratifying them by an indicator or a separate target price, reduces the incentive for increasing coding intensity. We believe that the expansion of the proposed risk adjustment model, which will include additional clinical risk adjusters, should be sufficient in accounting for pricing differences and clinical complexities among emergent procedures. Risk adjustment will likely result in higher target prices for emergent procedures, so that a hospital with a trauma center would have a higher target price at reconciliation for emergent episodes as compared to a community hospital that only performed elective procedures. Thus, we are not finalizing any policy specific to stratifying emergent procedures. However, in light of comments received, we may consider additional adjustments for emergent procedures in future rulemaking.
Comment: Some commenters support including both inpatient and outpatient episodes in TEAM. A few of commenters suggested that CMS create separate episode categories and target prices for inpatient and outpatient episodes within the same clinical episode category because the clinical characteristics of such patients can vary significantly in terms of complexity, care pathways, and recommended post-discharge treatment. A couple of commenters recommended that CMS set target prices for the inpatient cases without major comorbidity or complication separately from the targets for the outpatient cases. A couple of commenters stated that patients who need to remain in the facility overnight are clinically not able to have the procedure on a purely outpatient basis. A few commenters stated that the national trend towards outpatient surgery has not necessarily left high-waste procedures to the inpatient setting, only high-risk.
Response: We thank the commenters who expressed support for including both inpatient and outpatient episodes in TEAM. We also acknowledge some commenters' concerns regarding the inclusion of outpatient procedures and inpatient hospitalizations in the same episode category. We continue to believe the combined inpatient and outpatient pricing methodology discussed in section X.A.3.d.(2) of this final rule is more appropriate since it reduces any risks for beneficiaries to be inappropriately shifted from the inpatient to the outpatient setting. We agree that patient case-mix can vary between the inpatient and outpatient procedures. We also recognize a blended pricing structure could create pressure for clinicians to recommend the lower cost outpatient setting to minimize total episode costs. However, we believe that our risk adjustment methodology will incentivize clinicians to continue performing LEJR and Spinal Fusion procedures in the appropriate clinical setting based on their assessment of each patients' complexity, particularly since performing these procedures on sicker patients in the outpatient setting could increase the risk of post-acute complications and lead to higher overall episode spending. We understand the commenters' concerns related to the proposed risk adjustment methodology, and as ( print page 69714) discussed in section X.A.3.d.(4) of this rule, we are finalizing the risk adjustment methodology to include additional beneficiary-level covariates as well as some provider-level characteristics from what was proposed ( 89 FR 36433 ). CMS believes these modifications will further address differences in patient characteristics as well as variation in spending between outpatient and inpatient cases with MCCs.
While we acknowledge commenters' concerns with respect to excluding or stratifying certain episodes within the selected categories as either emergent or non-emergent and inpatient or outpatient, we don't believe excluding any of these episodes outright would serve Medicare beneficiaries or be in line with the goals of the model. We do believe the commenters' concerns will be addressed by the final risk adjustment methodology, which differs from the proposed risk adjustment methodology ( 89 FR 36433 ). We direct readers to section X.A.3.d.(4) of this final rule for a discussion on the risk adjustment methodology we are finalizing for TEAM. We have also indicated that we may consider additional updates to the risk adjustment methodology and any further updates would be made pursuant to future notice and comment rulemaking.
We thank commenters for their feedback on potential episode categories and the introduction of medical episodes in future years of the model. We will take commenters feedback into consideration should we expand the number of TEAM episodes in future years. Any additional episodes would be added to TEAM pursuant to notice and comment rulemaking.
In the proposed rule, we stated we believe that a straightforward approach for hospitals and other providers to identify Medicare beneficiaries in this payment model is important for the care redesign that is required for model success. Some of the inpatient procedures that group to the included MS-DRGs are also performed in the outpatient setting. To identify outpatient episodes for TEAM, we proposed to use methods similar to BPCI Advanced and CJR. Specifically, we proposed to match a hospital's institutional claim for TEAM procedure codes billed through the Outpatient Prospective Payment System (OPPS) ( 89 FR 36414 ).
Therefore, we stated in the proposed rule that as in the BPCI Advanced and CJR models, hospitals participating in the proposed TEAM would be able to identify beneficiaries in included episodes through their MS-DRG during the anchor hospitalization or, for hospital outpatient procedures, by their Healthcare Common Procedure Coding System (HCPCS) codes, allowing active coordination of beneficiary care during and after the procedure.
In the proposed rule, we stated that the MS-DRG for inpatient procedures would determine the ultimate MS-DRG assignment for the hospitalization, unless additional surgeries higher in the MS-DRG hierarchy also are reported. [ 887 ] This approach offers operational simplicity for providers and CMS and is consistent with the approach taken by the BPCI Advanced and CJR models to identify beneficiaries whose care is included in those episodes.
We sought comment on our proposal to identify episodes for inclusion in TEAM by MS-DRGs and HCPCS codes.
Comment: A commenter appreciated CMS incorporating previous feedback and experience with past episode-based models and recognizing that surgical MS-DRGs are easiest to predict and identify for purposes of managing patients.
Response: We thank the commenter for their support of the proposed use of MS-DRGs to identify episodes for TEAM.
We are finalizing the proposal to identify episodes with MS-DRGs and HCPCS codes for inclusion in TEAM without modification.
In the proposed rule, we stated that episode definitions have two significant dimensions—(1) a clinical dimension that describes which clinical conditions and associated services are included in the episode category; and (2) a time dimension that describes the beginning and end of the episode, its length, and when the episode may be cancelled prior to the end of the episode ( 89 FR 36414 ).
For the purposes of TEAM, we proposed to define episodes as including all Medicare Part A and Part B items and services described at proposed § 512.525(e), with some exceptions described at proposed § 512.525(f), beginning with an admission to an acute care hospital stay (hereinafter “the anchor hospitalization”) or an outpatient procedure at a hospital outpatient department (HOPD) (hereinafter “anchor procedure”), and ending 30 days following hospital discharge or anchor procedure ( 89 FR 36414 ).
As previously discussed in section X.A.3.b.(2) of the preamble of this final rule, the proposed episode categories were previously tested in BPCI Advanced and were voluntarily selected by BPCI Advanced participants. They represent the highest volume and highest cost surgical episode categories performed in the inpatient setting. However, we believe, based on current patterns of utilization and Medicare spending, there are still efficiencies to be gained by streamlining care pathways and transitions between clinical settings.
We stated in the proposed rule that we selected these episode categories because elective surgical procedures are more clinically similar and have greater spending predictability. In addition, these episode categories have a significant proportion of spending in the post-acute period, reflecting greater opportunity to improve care transitions for beneficiaries and reduce unnecessary hospitalizations and emergency care.
In section X.A.3.b.(2) of this final rule, we highlighted clarifying language that was issued in the correction notice for the proposed rule (CMS-1808-CN) pertaining to the data used in the BPCI Advanced episode analyses and why those estimates are different than those presented below. The volume estimates for the proposed TEAM episode categories described in the proposed rule ( 89 FR 36414 ) were higher than the BPCI Advanced episodes because (1) they reflect the proposed shorter, 30-day episodes which would be expected to have fewer exclusions and (2) the proposed episodes for TEAM were defined more broadly, including additional episode types (for example, outpatient spinal fusion, emergent CABG). Inclusion, exclusion, and overlap policies are discussed in sections X.A.3.b.(5) and X.A.3.e. of this final rule.
As mentioned in proposed rule, we identified the LEJR episode category for inclusion in this model. We noted in the proposed rule that the proposed LEJR episode category would include hip, knee, and ankle replacements, but exclude arthroplasty of the small joints in the foot, and both inpatient and outpatient procedures reimbursed through the IPPS under select MS-DRG and HOPD procedures billed under ( print page 69715) select HCPCS codes through the OPPS ( 89 FR 36414 ). [ 888 ]
While we recognized in the proposed rule that LEJR has been tested in other episode-based payment models. Given the promising findings for this episode category in those model tests, we believe there is value in continuing to test this episode category under an alternate payment methodology, particularly given the high volume of such procedures among the Medicare population. In addition, as mentioned in the proposed rule, TEAM would potentially capture underserved populations who were disproportionately underrepresented in CJR. We proposed to define the LEJR episode category as a hip, knee, or ankle replacement that is paid through the IPPS under MS-DRG 469, 470, 521, or 522 or through the OPPS under HCPCS code 27447, 27130, or 27702. We stated that this approach offers operational simplicity for providers and CMS and is consistent with the approach taken by previous models to identify beneficiaries whose care is included in the LEJR episode category ( 89 FR 36415 ).
We noted in the proposed rule that Medicare-covered outpatient total ankle arthroplasty (TAA) was excluded from both the BPCI Advanced and CJR models. However, since its removal from the Inpatient-Only List in 2021, the majority of TAA procedures have shifted to the outpatient setting. For example, in 2022, there were approximately 2,600 outpatient TAAs and only 600 TAAs performed in the inpatient setting. For this reason, and to be consistent with other episodes in the LEJR episode category, we proposed that both inpatient and outpatient TAAs would trigger an episode in TEAM ( 89 FR 36415 ).
Based on an analysis of 2021 historical LEJR episodes and an estimated number of additional outpatient TAAs, the annual number of potentially eligible beneficiary discharges for this mandatory model nationally would be approximately 226,000.
We sought public comment on our proposed definition of LEJR episodes for TEAM at § 512.525(d)(1). We also sought comment on the proposed MS-DRG and HCPCS codes and our proposal to include outpatient TAA in the LEJR episode category.
The following is a summary of the comments we received on the LEJR episode category and our responses:
Comment: A couple of commenters strongly support including LEJR as one of the five surgical episode categories. Another commenter asked that CMS add revision total joint replacement procedures (MS-DRGs 466, 467 468) to the LEJR episode, as these procedures are performed exclusively on an inpatient basis and the improvements CMS seeks to encourage would also apply to them. Another commenter supported continuing to exclude revision procedures. A few commenters suggested including the not insignificant volume of LEJR procedures that are performed in ASC settings and believe excluding them is a missed opportunity for this model.
Response: We thank the commenters for their support for including the LEJR episode category in the model.
We also acknowledge the commenter's suggestion to include revision total joint replacements in TEAM. While we agree that our goal with TEAM is to improve care for all beneficiaries receiving joint replacements, CMS elected not to include surgical procedures for TEAM that had not previously been tested in the voluntary the BPCI Advanced or mandatory CJR models.
We acknowledge that ASCs are popular settings for lower risk LEJR procedures and appreciate that commenters support testing ASC episodes in TEAM. However, as noted in section X.A.3.a.(2)(b)(i) of this final rule, we have not previously tested ASC episodes in the voluntary models that TEAM is built upon. Additionally, quality measures for ASC procedures would require additional consideration, as those being finalized for TEAM are hospital-level measures and may not be appropriate for episodes initiated at ASCs. Should we decide to expand the definition of TEAM episodes pursuant to future notice and comment rulemaking, we will take this feedback into consideration.
Comment: Several commenters recommended that CMS not include the LEJR episode category in TEAM. A couple of commenters stated that broad participation by hospitals in BPCI, BPCI-A, and CJR has removed much of the variation for this procedure, and it will be difficult for participants to find additional efficiencies in LEJR. Commenters also believe target prices have likely converged to the cost to provide an efficient LEJR episode, particularly for inpatient LEJRs, which have become higher risk with the movement of lower-acuity procedures to the outpatient and ASC settings. A commenter specifically recommended that CMS exclude TAA procedures from TEAM, as these medically-complex, high-cost, low-frequency cases could cause hospitals to face potential economic penalties.
Response: We appreciate that many hospitals have introduced greater efficiency to care pathways for LEJR procedures. We acknowledge the concern for some clinical episode categories like LEJR, which has been tested in both the CJR and BPCI Advanced models with a high participation rate. However, we disagree that the participating providers have a disadvantage in TEAM. CMS analyzed the post-discharge and post-acute care spending among providers participating and not participating in CJR and BPCI Advanced models and observed that both groups had similar spending trends, suggesting that there were additional opportunities for savings for LEJR in the post-discharge period for all providers. For TEAM, we proposed regional target prices where the participant hospitals' performance will be measured relative to their peers and not based on improvement relative to their own historical performance ( 89 FR 36428 ). This will mitigate concerns associated with the individual ratcheting effect.
We also believe there is value in continuing to test this episode category under an alternate payment methodology, particularly given the high volume of such procedures among the Medicare population. In addition, as previously mentioned, TEAM would potentially capture underserved populations who were disproportionately underrepresented in CJR.
We thank the commenter for their input on TAA procedures. We disagree that TAA procedures are high cost and penalize providers. First, we want to note that the blended inpatient and outpatient LEJR pricing approach being finalized in section X.A.3.d.(3) of this final rule, and additional risk adjusters based on patient characteristics being finalized in section X.A.3.d.(4), will allow providers to conduct TAA procedures in the outpatient or inpatient setting based on their assessment of each patient's complexity without creating any disincentives. Second, based on our preliminary analyses, TAA procedures for any clinical setting have lower average anchor and post-discharge costs than all procedures under MS-DRG 469, which will be assigned preliminary benchmark prices. However, we agree that they can be rare and medically complex ( print page 69716) procedures, so we are finalizing an additional risk adjuster for ankle procedures or reattachments in the LEJR episode category to account for any other differences associated with TAA procedures.
CMS also investigated what the equivalent to a 3 percent discount in a 90-day episode would be in a 30-day episode, assuming that anchor costs were not modifiable. As a result of this investigation, and considering savings opportunities, CMS is finalizing a reduced TEAM discount factor of 2 percent for the LEJR episode category, as compared to the discount factor specified in the proposed rule. We direct commenters to section X.A.3.d.(3)(g) for further discussion on the discount factor.
After consideration of the public comments we received, we are finalizing our proposal to include the LEJR episode category in TEAM without modification.
In the proposed rule, we proposed to define the SHFFT episode as a hip fixation procedure, with or without fracture reduction, but excluding joint replacement, that is paid through the IPPS under MS-DRG 480-482. We proposed that the SHFFT episode would include beneficiaries treated surgically for hip and femur fractures, other than hip arthroplasty and would include open and closed surgical hip fixation, with or without reduction of the fracture ( 89 FR 36415 ).
We stated in the proposed rule that the SHFFT episode was selected because it was the second highest volume, and second-highest cost BPCI Advanced surgical episode performed in the inpatient setting, based on an analysis of 2021 episode data. There were 69,076 episodes with a total cost of $3.22 billion. In addition, we stated that more than 63 percent of spending occurred in the post-acute period, signifying potential opportunity for care improvement. Using that same data for historical SHFFT episodes, the annual number of potentially eligible beneficiary discharges for this episode category nationally would be approximately 85,000 ( 89 FR 36415 ).
Together, the LEJR and SHFFT episode categories cover all surgical treatment options for Medicare beneficiaries with hip fracture (that is, hip arthroplasty and fixation). Although a small number of SHFFT procedures are furnished in the outpatient hospital setting, TEAM would only include inpatient procedures, which conforms with hip and femur procedure except major joint episodes under BPCI Advanced.
Thus, we proposed to include episodes for beneficiaries admitted and discharged from an anchor hospitalization paid under a SHFFT MS-DRG (480-482) under the IPPS in TEAM.
We sought comment on our proposed definition of SHFFT and our proposal to include the SHFFT episode category at § 512.525(d)(2).
The following is a summary of the comments we received on the SHFFT episode category and our responses:
Comment: A commenter supported the choice of SHFFT as an episode. Another commenter recommended removing the SHFFT episode category, as these procedures are less likely to be scheduled in advance, removing the opportunity for the hospital to intervene prior to hospitalization. Another commenter expressed concerns about including hip fracture cases in a mandatory model given the variability in costs and outcomes, especially for non-elective, trauma-related cases. Another commenter stated that the large number of procedure codes in these MS-DRGs, represent disparate treatments (for example, repositioning procedure versus insertion procedure) and anatomical locations (for example, upper femur versus lower femur) with different costs, lengths of stay, and readmission rates that are not accounted for in the bundle. Another commenter suggested excluding the episode because those sustaining hip fractures are often elderly, osteoporotic patients with complex co-morbidities.
Response: We thank the commenters for their input on the appropriateness of including the SHFFT episode category in TEAM. We are aware of concerns regarding cost and clinical variability associated with hip fractures and non-elective procedures in general. However, based on our experience with hip fracture bundles in the BPCI Advanced model, we continue to believe there are additional efficiencies and care improvement to be achieved for patients undergoing these procedures. We also point out that the BPCI Advanced Hip and Femur Procedures Except Major Joint episode category was the third highest volume episode category, behind major joint replacement and outpatient percutaneous coronary intervention episodes and was voluntarily selected by participants of the BPCI Advanced model, who tended to choose episodes under which they expected to succeed. We also refer commenters to section X.A.3.d.(4) for a discussion on updates to the risk adjustment methodology and a more robust set of risk adjusters that CMS is finalizing to capture episode spending accurately. Finally, we direct commenters to section X.A.3.d.(3)(g) for a discussion on the discount factor that CMS is finalizing for the SHFFT episode category. CMS is finalizing a 2 percent discount factor for the SHFFT episode category, which is a reduction from the discount factor proposed in the proposed rule ( 89 FR 36431 ).
After consideration of the public comments we received, we are finalizing our proposal to include the SHFFT episode category in TEAM. However, we may consider additional analysis on how to best address emergent and trauma-related episodes to ensure we do not unduly disadvantage participant hospitals selected for the model. If analysis results warrant a new or updated policy, we would address it pursuant to future notice and comment rulemaking.
In the proposed rule, we stated that the proposed CABG episode category would include beneficiaries undergoing coronary revascularization by CABG. [ 889 ] This episode category was selected in order to maintain the engagement of cardiac surgeons who have participated in prior episode-based models. Among cardiac procedures, it was the second highest cost and second highest volume BPCI Advanced surgical episode performed in the inpatient setting using 2021 data. There were 26,259 episodes with a total cost of $1.39 billion ( 89 FR 36415 ).
We stated in the proposed rule that we also considered the percutaneous coronary intervention (PCI) episode category for TEAM because it was the highest volume and highest cost surgical cardiac episode. However, we did not select this episode because PCI has been described as a low-value service by the Medicare Payment Advisory Commission when performed for stable coronary artery disease, [ 890 ] and the majority of PCIs are not associated with an acute care hospitalization.
In the proposed rule, we proposed to define the CABG episode category as any coronary revascularization procedure that is paid through the IPPS under MS-DRG 231-236, including both elective CABG and CABG ( print page 69717) procedures performed during initial acute myocardial infarction treatment (AMI). Based on an analysis of 2021 historical CABG episodes, the annual number of potentially eligible beneficiary discharges for CABG episodes in TEAM would be approximately 30,000.
We sought comment on our proposed definition of the CABG episode category and our proposal to include emergent CABG in episodes at § 512.525(d)(3).
The following is a summary of the comments we received on the CABG episode category and our responses:
Comment: A couple of commenters supported the inclusion of the CABG episode category in TEAM. One stated that the CABG procedure has a more consistent care pathway compared to other cardiovascular conditions, such as atrial fibrillation or congestive heart failure, and has also been included in several commercial and state value-based projects. We also received several comments in support of CMS' decision to not include the PCI episode category. A commenter stated a PCI episode category would be more difficult to implement based on its variation in care including acute and non-acute settings, as well as the number of performed outside of the hospital settings. Another commenter believed including such episodes introduces additional specialists to the episode, complicating attribution, decision-making, and follow up.
Response: We thank commenters for their support of the inclusion of CABG in TEAM and the decision to not include PCI.
Comment: Several commenters strongly encouraged CMS to reconsider the mandatory inclusion of CABG in TEAM and allow for voluntary selection. A commenter said few hospitals perform enough of these procedures to be able to dedicate additional resources to them. Another cautioned the high underlying procedure costs will make it difficult for hospitals to meet target prices and provide less opportunity for hospitals to optimize costs and increase value. Another commenter recommended CMS exclude CABG episodes with acute myocardial infarction to ensure that the remaining episodes are homogenous and more readily analyzed.
Response: We thank the commenters for sharing their concerns about including CABG as a mandatory episode category in TEAM. However, we believe that testing all of the proposed episode categories in TEAM will more effectively test how participants perform in clinical areas that they would otherwise not select voluntarily. Allowing participants to voluntarily choose episode categories would introduce selection bias and make evaluating TEAM more difficult and produce less generalizable findings. We expect that TEAM will produce data that are more broadly representative of spending, quality, and outcomes than what might be collected under a voluntary model.
We disagree that the frequency of CABG procedures and the high costs associated with the anchor period warrant voluntary selection. While the volume of CABG episodes is lower than the other four proposed episode categories, a sufficiently high proportion of hospitals perform CABG procedures. Based on our analyses, 30 to 40 percent of acute care hospitals eligible for TEAM across all regions had a CABG episode using 2023 Quarter 1 and Quarter 2 data. Additionally, as discussed in section X.A.3.d.(3)(h) of the preamble of this final rule, CMS may consider protections in reconciliation for low volume hospitals pursuant to future notice and comment rulemaking.
We disagree that CABG provides less opportunity for savings. As stated in the proposed rule, CABG was the second highest cost and second highest volume BPCI Advanced surgical episode performed in the inpatient setting using 2021 BPCI Advanced data ( 89 FR 36413 ). CMS continues to believe that the high-expenditure services involved in CABG procedures make it an ideal episode category to support the goals of the model and maintain engagement of cardiac surgeons who have participated in prior episode-based models. We also direct commenters to section X.A.3.d.(3)(g) for a discussion on our decision to finalize a 1.5 percent discount factor for the CABG episode category in TEAM, which is a significant reduction from the discount factor that was proposed.
We recognize that the proportion of anchor costs are relatively higher than the post discharge costs in CABG episodes; however, hospitals on average have higher observed than expected spending that accounts for patient acuity and regional trends. Additionally, a patient's surgical course can be affected by perioperative management including surgical technique, anesthesia requirement, extubation times, ambulation, management of bleeding, and prevention of infection. This suggests that there are opportunities for savings in the CABG episode category.
We disagree that hospitals will have difficulty meeting target prices in the CABG episode category. Target prices are based on historical data and expected to capture the underlying mix of anchor and post-acute care services. Regional level target prices will also account for any variation in costs due to patient acuity or regional trends, which are not under the provider's control.
After consideration of the public comments received, we are finalizing our proposal to include the CABG episode category in the model without modification.
We proposed to include in TEAM the Spinal Fusion episode category for beneficiaries undergoing inpatient and outpatient spinal fusion. The spinal fusion episode category was selected because it was the third-highest cost BPCI Advanced surgical episode performed in the inpatient setting using 2021 data. There were 62,345 episodes with a total cost of $3.2 billion. Based on the high number of episodes and its voluntary selection by participants in BPCI Advanced, we believe there are additional opportunities to improve care for beneficiaries undergoing these procedures ( 89 FR 36415 ).
We proposed to define the Spinal Fusion episode category as any cervical, thoracic, or lumbar spinal fusion procedure paid through the IPPS under MS-DRG 453-455, 459-460, or 471-473, or through the OPPS under HCPCS codes 22551, 22554, 22612, 22630, or 22633.
In the correction notice for the proposed rule (CMS-1808-CN) published May 31, 2024, we noted that the proposed changes to several of the spinal fusion MS-DRGs discussed at 89 FR 35971 would, if finalized, directly affect the proposed definition for the Spinal Fusion episode category for TEAM. We also directed the reader to the draft version of the ICD-10 MS-DRG Definitions Manual, Version 42 on the CMS website at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software to view the changes that were proposed to the spinal fusion MS-DRGs for FY 2025. We stated that, if finalized for FY 2025 as proposed ( 89 FR 35971 ), rather than including MS-DRGs 453, 454, and 455 in the definition for the TEAM Spinal Fusion episode category, we would include the eight proposed MS-DRGs: MS-DRG 426 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with MCC), MS-DRG 427 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with CC), MS-DRG 428 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical without CC/MCC), MS-DRG 402 (Single ( print page 69718) Level Combined Anterior and Posterior Spinal Fusion Except Cervical), MS-DRG 429 (Combined Anterior and Posterior Cervical Spinal Fusion with MCC), MS-DRG 430 (Combined Anterior and Posterior Cervical Spinal Fusion without MCC), MS-DRG 447 (Multiple Level Spinal Fusion Except Cervical with MCC) and MS-DRG 448 (Multiple Level Spinal Fusion Except Cervical without MCC). In addition, we stated in the proposed rule that, if finalized as proposed at 89 FR 35984 , we would use the revised titles for existing MS-DRGs 459 and 460, “Single Level Spinal Fusion Except Cervical with MCC and without MCC”, respectively, for the TEAM Spinal Fusion episode definition.
In the proposed rule, we stated that based on an analysis of 2021 BPCI Advanced episodes and an estimated number of additional outpatient episodes, the annual number of potentially eligible TEAM Spinal Fusion episodes would be approximately 94,000.
We sought comment on our definition and inclusion of the Spinal Fusion episode category at § 512.525(d)(4).
The following is a summary of the comments we received on the Spinal Fusion episode category and our responses:
Comment: A couple of commenters supported inclusion of the Spinal Fusion episode category in TEAM.
Response: We thank the commenters for their support of our proposal to include the Spinal Fusion episode category in TEAM.
Comment: Several commenters recommended CMS first test a more limited bundle, focusing on single-level lumbar fusion, as it typically the most common lumbar fusion, to ensure homogeneity of diagnosis, treatment, and patient experiences before expanding to a broader episode category. A commenter noted that including cervical fusion (CPT code 22551) is unnecessary, since CMS initiated a relatively unprecedented prior authorization control over this code in 2021. A commenter recommended both cervical fusion codes (22551 and 22554) be removed from this list in order to focus on the primarily inpatient lumbar fusion procedures. The commenter believed that this would provide consistency in expectations for surgeons and focus on the more resource-intensive inpatient cases.
Response: We thank commenters for the suggestion to only include single-level lumbar fusions and to remove cervical fusion codes from the list of included procedures. We believe that limiting the spinal fusion episode category to single-level lumbar fusions would create unintended consequences by incentivizing multi-level procedures. Including all levels of fusions in the episode category will remove potentially distorted financial incentives to ensure that participants base decisions solely on patient need. We acknowledge that both cervical fusion procedures mentioned by the commenters (HCPCS codes 22551 and 22554) require prior authorization to be performed by a provider. However, CMS believes that prior authorization is applicable to triggering procedures but not the post-acute care services provided after the procedure. Including them in TEAM will allow for improvement in quality of care in the post discharge period while reducing overall Medicare spending.
CMS acknowledges that post-acute care for cervical and lumbar fusion patients may differ, and the pricing methodology takes this into account. As discussed in section X.A.3.d.(3), we are finalizing our proposal to calculate TEAM target prices by episode types and regions, reflecting the potentially different historical episode spending for both cervical and lumbar procedures. Risk adjustment will also be done at the MS-DRG-level to account for the effect of each risk adjuster on episode spending. We direct readers to section X.A.3.d.(4) of this final rule for a discussion of our finalized risk adjustment policy.
Comment: Many commenters strongly recommended that the Spinal Fusion episode category be excluded from TEAM. Several commenters stated that, particularly in light of the proposed discount factor ( 89 FR 36431 ), the target prices for these procedures are more likely to be eroded by the underlying procedure costs and therefore limit the patients' ability to receive medically necessary post-discharge items and services. Some commenters believe CMS must first conduct a thorough and complete evaluation of the current BPCI Advanced model and, until CMS can provide publicly available data ensuring that spinal fusion episodes did not have a negative impact on patient quality, outcomes, and experience of care, these episodes should not be included in a mandatory model.
Response: We thank commenters for these suggestions and acknowledge concerns that episodes with higher procedure costs may reduce the magnitude of savings that can be achieved. To better understand the effect of non-modifiable anchor costs on the ability to meet target prices, CMS investigated what the equivalent to a 3 percent discount in a 90-day episode would be for a 30-day episode. As a result of this investigation, and potential savings opportunities, CMS is finalizing a 2 percent discount factor for the Spinal Fusion episode category in TEAM. This is a reduction from what was originally proposed for TEAM ( 89 FR 36431 ). We direct commenters to section X.A.3.d.(3)(g) of this final rule for further discussion on the discount factor.
We also direct commenters to the most recent BPCI Advanced evaluation for information on the patient quality, outcomes, and experience of care impacts of spinal fusion episodes, available at: https://www.cms.gov/priorities/innovation/data-and-reports/2024/bpci-adv-ar5 .
Comment: Some commenters expressed concern with respect to the proposed restructuring of spinal fusion DRGs discussed in the proposed rule ( 89 FR 35984 ) and in Section II.C.6.b of this final rule. A commenter stated MS-DRG changes often lead to volatility and potential future refinements. For this reason, they stated that selecting spinal fusion for inclusion in the TEAM demonstration creates added complexity that could lead to difficulty for hospitals trying to manage care under the proposed model. A commenter stated that the current MS-DRGs don't directly map to the proposed new spinal fusion MS-DRGs. Some commenters urged CMS to exclude the Spinal Fusion episode category or at least delay implementation until the agency is able to monitor the impact of the proposed MS-DRGs, if finalized, and has three years of data based on the new groupings. The commenters believed this would allow for the proposed MS-DRGs to be fully reflected in both performance year and baseline assessments so TEAM participants may better understand the applicable target prices and needed efforts to manage 30 days of post-discharge care. A couple of commenters suggested CMS also include the newly proposed MS-DRG 402 in the spinal fusion definition if the spinal fusion MS-DRGs are finalized.
Response: As discussed in section II.C.6.b. in the preamble of this final rule, CMS is finalizing the proposed restructuring of the spinal fusion MS-DRGs for FY 2025, with modifications, effective October 1, 2024. We appreciate that there are concerns regarding the restructuring of the spinal fusion MS-DRGs, as these MS-DRGs were also proposed for inclusion in TEAM ( 89 FR 36415 ). However, because the final spinal fusion MS-DRGs affect all spinal fusion MS-DRG payments, TEAM must conform to the changes. We believe that ( print page 69719) the redistribution of the spinal fusion procedures to ten new MS-DRGs will decrease some of the concerns reflected in comments we received about the spinal fusion MS-DRGs currently in use not being granular enough and including too great a spectrum of procedures. We believe that the final spinal fusion MS-DRGs, which separate single and multi-level fusions, respond, in part, to commenters who believe these procedures should be considered differently and will reduce the variance of procedures within each of the finalized MS-DRGs. We also believe the final spinal fusion MS-DRGs will enable us to better analyze the appropriateness of including both single and multi-level fusions in TEAM.
We disagree that the currently used MS-DRGs don't directly map to the final FY 2025 MS-DRGs. Although the final MS-DRGs separate single- and multi-level spinal fusions into different MS-DRGs, as a group, they will capture the same pool of episodes that were previously assigned to the deleted and restructured MS-DRGs.
We intend to conduct a thorough review and analysis of how the composition of episodes under the current spinal fusion MS-DRGs may change with the implementation of the final MS-DRGs for FY 2025. We intend to propose and finalize the pricing methodology for the TEAM Spinal Fusion episode category pursuant to FY2026 IPPS rulemaking. CMS also plans to develop, through rulemaking, a method to address in any future year of the model the potential addition or removal of procedures to or from MS-DRGs that are included in the definitions of TEAM episode categories.
Comment: Several commenters fear that the migration of a group of spine surgeons from a participant hospital could significantly affect their case-mix and episode costs.
Response: We thank the commenters for raising their concerns about the migration of spine surgeons from a participant hospital. Participants in TEAM will be selected based on a stratified random sampling procedure done at the CBSA level. Given the relatively large geographic area that CBSAs cover, CMS does not believe that groups of surgeons are likely to relocate across CBSAs to avoid participation in TEAM. Furthermore, TEAM incentivizes hospitals and surgeons to work together to provide the highest quality, most efficient care, and hospitals are incentivized to retain their best surgeons. While we agree with the commenters that the loss or gain of a large number of surgeons could alter a hospital's case mix and resultant costs, it will likely not affect the participant negatively. The risk adjustment methodology finalized in section X.A.3.d.(3) accounts for patient case-mix nationally using data from the baseline period. For a given participant, the risk adjustment multipliers from the baseline are going to be applied to their performance year episodes, which means that final target prices will take the realized patient case-mix into account.
Comment: Several commenters stated that spinal fusion MS-DRGs are not sufficient to delineate the extreme variance in spinal procedures and that a single target price at the MS-DRG level is inadequate for more complex fusion cases. Some commenters believe the spinal fusion episode category runs the risk of penalizing trauma and other high acuity centers. A commenter asked CMS to consider that there are separate MS-DRGs for hip fractures, while spinal fusion MS-DRGs do not distinguish between the presence or absence of a fracture. Several commenters requested that CMS stratify episodes for a variety of factors, such as non-elective, complexity (for example, spondylolisthesis, scoliosis, kyphosis), trauma, cancer or spinal tumors, spinal infections, and admission through the emergency department. The commenters believe that hospitals that treat these conditions would be at a significant disadvantage within the proposed pricing methodology. A commenter stated that trauma centers caring for spinal fractures will have the same episode target price as community hospitals caring for degenerative spines; despite having the same MS-DRG, the post-hospital clinical experience is vastly different. A commenter stated that the 30-day readmission rate for emergent spine fusions at their hospital is typically in the range of 15 to 25 percent, compared to five percent for elective spine fusions. A commenter recommended segmenting the spine fusion episode types by the presence of a relevant trauma or fracture ICD-10 diagnosis code in the claims data of the anchor hospital. A commenter suggested that CMS exclude revision spine surgery and episodes in which a patient undergoes both anterior and posterior approach in the same admission.
Response: We thank the commenters for sharing their concerns regarding the clinical variation captured in the spinal fusion MS-DRGs and the need for TEAM to adequately account for risk factors that may affect episode spending and quality. We acknowledge the commenters' concerns that more complex spinal fusions, such as those which include cancer, would be more expensive. We agree with the commenters that a more robust risk adjustment methodology is necessary for TEAM. We refer readers to section X.A.3.d.(4), which details the final risk adjustment model that differs from what was proposed ( 89 FR 36433 ). The final risk adjustment policy includes the addition of several beneficiary-level risk adjusters that will adjust the target prices to reflect the complexity of patients demonstrated in the 90-day lookback period. The finalized risk adjustment methodology also incorporates HCC variables, such as an HCC indicator for metastatic cancer and acute leukemia (HCC8), in addition to the HCC count variable, in order to create more accurate episode spending predictions than what was proposed, in that they are based on the clinical complexity of the patient case mix and additional resource use. The finalized risk adjustment methodology also includes a prior post-acute care variable, which was not included in the proposed methodology, to account for patients who have visited a post-acute care facility during the lookback period for LEJR, CABG, and Spinal Fusion. These facilities include long-term care hospitals (LTCH), skilled nursing facilities (SNF), home health (HH), and inpatient rehabilitation facility (IRF). We believe these final policies will address the disadvantages noted by the commenters.
We acknowledge the commenter's suggestion to exclude revision spine surgery. However, CMS is concerned with the ability to identify revision procedures in the coding, since the coding does not specify revisions and the prior surgery may have occurred years before and is not captured in Medicare FFS data. Additionally, we acknowledge that patients which undergo anterior and posterior approach in the same admission may be more expensive. We will monitor spinal fusion episodes and consider whether additional adjustments are necessary in future rulemaking.
After reviewing the public comments, we are finalizing the proposed Spinal Fusion episode category with modification. To conform to the final spinal fusion MS-DRGs discussed in section II.6.b, the Spinal Fusion episode category is defined as any cervical, thoracic, or lumbar spinal fusion procedure paid through the IPPS under the following MS-DRGs or through the OPPS under the following HCPCS codes:
- 402 (Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical). ( print page 69720)
- 426 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with MCC or Custom-Made Anatomically Designed Interbody Fusion Device).
- 427 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with CC).
- 428 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical without CC/MCC).
- 429 (Combined Anterior and Posterior Cervical Spinal Fusion with MCC).
- 430 (Combined Anterior and Posterior Cervical Spinal Fusion without MCC).
- 447 (Multiple Level Spinal Fusion Except Cervical with MCC or Custom-Made Anatomically Designed Interbody Fusion Device).
- 448 (Multiple Level Spinal Fusion Except Cervical without MCC).
- 450 (Single Level Spinal Fusion Except Cervical with MCC or Custom-Made Anatomically Designed Interbody Fusion Device).
- 451 Single Level Spinal Fusion Except Cervical without MCC
- 471 (Cervical Spinal Fusion with MCC).
- 472 (Cervical Spinal Fusion with CC).
- 473 (Cervical Spinal Fusion without CC/MCC).
- 22551 (Anterior Cervical Spinal Fusion with Decompression Below C2).
- 22554 (Anterior Cervical Spinal Fusion without Decompression).
- 22612 (Posterior or Posterolateral Lumbar Spinal Fusion).
- 22630 (Posterior Lumbar Interbody Lumbar Spinal Fusion).
- 22633 (Combined Posterior or Posterolateral Lumbar and Posterior Lumbar Interbody Spinal Fusion).
In the proposed rule, we proposed to include in TEAM the Major Bowel Procedure episode category for beneficiaries undergoing inpatient major small bowel and large bowel procedures. This episode category was selected because it was the fifth-highest volume and fourth-highest cost BPCI Advanced surgical episode performed in the inpatient setting using 2021 data. There were 54,848 episodes with a total cost of $1.95 billion. We believe there are still opportunities to streamline care pathways and improve care transitions for beneficiaries receiving this care ( 89 FR 36415 ).
We proposed to define the Major Bowel Procedure episode category as any small or large bowel procedure paid through the IPPS under MS-DRG 329-331. Based on an analysis of 2021 data for historical Major Bowel Procedure episodes, the annual number of potentially eligible beneficiary discharges for episodes in TEAM would be approximately 64,000.
We sought comment on our proposed definition and inclusion of the Major Bowel Procedure episode at § 512.525(d)(5).
The following is a summary of the comments we received on the Major Bowel Procedure episode category and our responses:
Comment: A commenter was in support of including the Major Bowel Procedure episode category, but encouraged CMS to ensure that target prices and peer adjustment factors reflect the fact that gastrointestinal disorders in the elderly often coincide with major chronic conditions, such as renal failure and congestive heart failure. Several commenters recommended CMS remove the Major Bowel Procedure episode category, as the category is too broad and includes procedures that are less likely to be scheduled in advance, giving hospitals limited opportunity to optimize costs and increase value. A couple of commenters suggested that CMS exclude small bowel procedures from the Major Bowel Procedure episode category because of the complexity of the service line. They further recommended that, if CMS did include small bowel procedures, CMS should create separate small and large bowel procedure episode categories because they have distinctly different diagnoses, surgical treatment, clinical outcomes, and attendant risks.
Response: We thank the commenter for the support of the Major Bowel Procedure episode category in TEAM and disagree with commenters that it should be removed from TEAM. We also thank the commenters for their suggestions to exclude small bowel procedures from the Major Bowel Procedure episode category; however, CMS believes this can significantly affect the episode volume and reach of TEAM for this episode category. We also acknowledge recommendations to stratify small and large bowel procedures and for the additional suggestions regarding the risk adjustment methodology for these episodes.
CMS believes that calculating TEAM target prices at the MS-DRG level accounts for complexity through the presence of diagnosis codes on the Major Complication or Comorbidity (MCC) or CC list. Additionally, as explained in section X.A.3.d.(4) of this final rule, we are finalizing a list of 19 risk adjusters for Major Bowel Procedure, including HCC 21 (Protein-Calorie Malnutrition) and HCC 33 (Intestinal Obstruction/Perforation), which will further address differences in episode spending due to high complexity conditions and negate the need for stratification. This list of risk adjusters is an update from what was proposed ( 89 FR 36433 ). We also direct commenters to section X.A.3.d.(3)(g) for a discussion on our finalized TEAM discount factor of 1.5 percent for the Major Bowel Procedure episode categories., which is a significant reduction from what was proposed ( 89 FR 36431 ).
Comment: A couple of commenters suggested including only elective procedures in the episode, as this would better align with clinical care, determining the value of an episode, assigning quality metrics, informing patients, providing information to referring PCPs, and aiding health plans seeking to contract for episodic-specific services. A couple of commenters also strongly recommended that CMS episode exclude urgent/emergent procedures. A commenter stated that there is major variation in cost per episode in elective versus emergent cases for both small and large bowel services; in their analysis, urgent/emergent episodes cost roughly $15,000-$20,000 more than elective episodes.
Response: We acknowledge the commenters' input on elective and emergent procedures in the Major Bowel Procedure episode category. In BPCI Advanced, 54 percent of Major Bowel Procedure episodes were performed electively, with the remainder performed emergently. CMS is concerned that only including elective procedures and excluding urgent/emergent procedures may drop episode volume substantially and negatively impact the reach of the model. However, we acknowledge that these emergent procedures may be more expensive than elective procedures. We believe we can account for the pricing differences between emergent and elective procedures by including additional HCC risk adjusters in the final list of risk adjusters for this episode category. We refer the readers to section X.A.3.d.(4) of this final rule for the comprehensive list of risk adjustment variables, including individual HCCs, that are being finalized for TEAM. Additionally, we may consider whether additional adjustments for emergent procedures are needed for the Major Bowel Procedure episode category in future years of the model. Any changes will be made pursuant to notice and comment rulemaking. ( print page 69721)
Comment: A commenter requested that CMS delay the inclusion of the Major Bowel Procedure episode category in TEAM, given CMS' proposed shift in procedures from MS-DRGs 347, 348, and 349 to MS-DRGs 329, 330, and 331 ( 89 FR 35968 ). The commenter urged CMS to reconcile the different composition of these MS-DRGs for purposes of setting TEAM episode prices, and to delay inclusion if unable to do so timely.
Response: We acknowledge the commenters' concerns with the proposed reassignment of eight procedure codes that describe excision of intestinal body parts from MS-DRGs 347, 348 and 349 to MS-DRGs 329, 330 and 331 for the TEAM Major Bowel Procedure episode category ( 89 FR 35968 ). We direct readers to section II.C.5. for a full discussion of the reassignments. CMS recognizes that the proposed baseline period for initial TEAM performance years will not account for the patient case-mix and post-discharge resource utilization represented by these procedure codes, impacting the preliminary target prices. In future rulemaking, CMS may consider analyzing and implementing a methodology similar to the BPCI-Advanced methodology to remap the baseline MS-DRGs to performance year MS-DRGs and allow for episodes to be triggered based on the remapped MS-DRGs. This would account for any differences in patient case-mix, post-discharge resource utilization, and other spending patterns between the baseline and performance year.
After consideration of the public comments we received, we are finalizing the Major Bowel Procedure episode category as proposed, without modification.
The following Table X.A.-08 summarizes the five final episode categories and corresponding billing codes that CMS is finalizing for purposes of identifying episodes in TEAM.

Like previous episode-based payment models, TEAM would incentivize comprehensive, coordinated, patient-centered care through inclusive episodes. We proposed to include in the episode all items and services paid under Medicare Part A and Part B during the performance period, unless such items and services fell under one of the proposed exclusions, described in the preamble of the proposed rule ( 89 FR 36416 ).
We proposed to include all Part A services furnished during the proposed 30-day post-discharge period of the episode, other than certain excluded hospital readmissions, as post-hospital discharge Part A services are typically intended to be comprehensive in nature. In particular, we believe that claims for services with diagnosis codes that are directly related to the proposed episode categories or the quality and safety of care furnished during the episode, based on clinical judgment (for example, surgical wound infection) and taking into consideration coding guidelines, should be included in an episode. Thus, we proposed that items and services for episodes would include the following items and services paid under Medicare Part A and Part B, subject to the proposed exclusions in the preamble of the proposed rule ( 89 FR 36416 ) and section X.A.3.b.(5)(a) of this final rule:
- Physicians' services.
- Inpatient hospital services, including services paid through IPPS operating and capital payments.
- Inpatient psychiatric facility (IPF) services.
- Long-Term Care Hospital (LTCH) services.
- Inpatient Rehabilitation Facility (IRF) services.
- Skilled Nursing Facility (SNF) services.
- Home Health Agency (HHA) services.
- Hospital outpatient services.
- Outpatient therapy services.
- Clinical laboratory services.
- Durable medical equipment (DME).
- Part B drugs and biologicals except for those excluded under § 512.525 (f) as proposed.
- Hospice services.
- Part B professional claims dated in the 3 days prior to an anchor hospitalization if a claim for the surgical procedure for the same episode category is not detected as part of the hospitalization because the procedure was performed by the TEAM participant on an outpatient basis, but the patient was subsequently admitted as an inpatient.
We sought comment on the proposed items and services we proposed to include in TEAM episodes in § 512.525(e).
The following is a summary of comments we received on the items and services we proposed to include in TEAM episodes and our responses:
Comment: A commenter believed the model addresses the undervaluation of spinal implants by including all costs attributable to the case, including the physician services, rather than just the hospital costs related to the procedure. They also stated that this will lead to better clinical choices for the patient and help participants to succeed under the model.
Response: We thank the commenter for their support. We are finalizing the items and services included in TEAM episodes as proposed without modification.
In the proposed rule, we proposed to exclude from episodes certain Part A and B items and services that are ( print page 69722) clinically unrelated to the anchor hospitalization or anchor procedure. The proposed exclusions would be applicable to episodes included during the baseline period, the three-year historical period used to construct target prices, as described in section X.A.3.d.(3) of the preamble of this final rule, and episodes initiated during a performance year ( 89 FR 36416 ).
As explained in the proposed rule, the proposed exclusions are similar to those excluded from BPCI Advanced, as discussed in detail later in this section. [ 891 ] We have used similar exclusions in CMS Innovation Center models, with minor adjustments since BPCI, and intend to continue to apply them to TEAM. The exclusions list was developed through a collaborative effort between CMS and external stakeholders and has been vetted broadly in the health care community. We proposed to use the BPCI Advanced exclusions list in TEAM based on several years of experience with these exclusions and their suitability for episodes. As stated in the proposed rule, the rationale for these exclusions described below is consistent with the rationale for exclusions in the CJR model ( 80 FR 73304 ) and in BPCI Advanced.
In the proposed rule, we proposed to exclude from episodes all Part A and B items and services, for both the baseline period and performance years, for hospital admissions and readmissions for specific categories of diagnoses, such as oncology, trauma medical admissions, organ transplant, and ventricular shunts determined by MS-DRGs, as well as all the following excluded Major Diagnostic Categories (MDC): [ 892 ]
- MDC 02 (Diseases and Disorders of the Eye).
- MDC 14 (Pregnancy, Childbirth, and Puerperium).
- MDC 15 (Newborns and other neonates with conditions originating in perinatal period).
- MDC 25 (Human immunodeficiency virus infections).
In the proposed rule, we proposed to exclude from episodes IPPS new technology add-on payments for drugs, technologies, and services identified by value code 77 on IPPS hospital claims for episodes in the baseline period and performance years. [ 893 ] New technology add-on payments are made separately and in addition to the MS-DRG payment under the IPPS for specific new drugs, technologies, and services that substantially improve the diagnosis or treatment of Medicare beneficiaries and would be inadequately paid under the MS-DRG system. We believe it would not be appropriate for TEAM to potentially diminish beneficiaries' access to new technologies or to burden hospitals who choose to use these new drugs, technologies, or services with concern about these payments counting toward TEAM participants' actual episode spending. Additionally, new drugs, technologies, or services approved for the add-on payments vary unpredictably over time in their application to specific clinical conditions. Exclusion of new technology add-on payments for drugs, technologies, or services approved for add-on payments from episodes in TEAM is similar to episode exclusions in the CJR model ( 80 FR 73303 and 73304 and 73315 ) ( 89 FR 36417 ).
In the proposed rule, we also proposed to exclude from episodes OPPS transitional pass-through payments for medical devices as identified through OPPS status indicator H for episodes in the baseline period and performance years. Through the established OPPS review process, we have determined that these technologies have a substantial cost but also lead to substantial clinical improvement for Medicare beneficiaries. This proposal is also consistent with the BPCI Advanced and CJR model final exclusions policies ( 80 FR 73308 and 73315 ).
In the proposed rule, we proposed to exclude from episodes drugs or biologicals that are paid outside of the MS-DRG, specifically hemophilia clotting factors (§ 412.115), identified through HCPCS code, diagnosis code, and revenue center on IPPS claims for episodes in the baseline period and performance years. Hemophilia clotting factors, in contrast to other drugs and biologicals that are administered during an inpatient hospitalization and paid through the MS-DRG, are paid separately by Medicare in recognition that clotting factors are costly and essential to appropriate care for certain beneficiaries. Because we do not believe that there are any spending efficiencies to be gained by including hemophilia clotting factors, we proposed to exclude these high-cost drugs from episodes initiated during the baseline period and performance year.
In the proposed rule, we proposed to exclude from episodes certain Part B payments for high-cost drugs and biologicals, low-volume drugs, [ 894 ] and blood clotting factors for hemophilia patients billed on outpatient, carrier, and DME claims for episodes in the baseline period and initiated in the performance years. These high-cost items are essential to appropriate care of certain beneficiaries, and we do not believe including them in the episode would improve any spending or quality of care efficiencies. We stated in the proposed rule that this proposed list would include:
- For episodes included during the baseline period:
++ Drug/biological HCPCS codes that are billed in fewer than 31 episodes in total across all episodes in TEAM during the baseline period.
++ Drug/biological HCPCS codes that are billed in at least 31 episodes in the baseline period, and have a mean allowed cost of greater than $25,000 per episode in the baseline period; and
++ HCPCS codes corresponding to clotting factors for hemophilia patients, identified in the quarterly average sales price file [ 895 ] for certain Medicare Part B drugs and biologicals as HCPCS codes with clotting factor = 1, HCPCS codes for new hemophilia clotting factors not in the baseline period, and other HCPCS codes identified as hemophilia.
- For episodes initiated during a performance year, in addition to those listed in the previous bullet, Part B payments for high-cost drugs and biologicals, low-volume drugs, and blood clotting factors for hemophilia billed on outpatient, carrier, and DME claims, including, but not limited to:
++ Drug/biological HCPCS codes that were not included in the baseline period and appear in 10 or fewer episodes in the performance year.
++ Drug/biological HCPCS codes that were not included in the baseline period, appear in more than 10 episodes in the performance year, have a mean cost of greater than $25,000 per episode in the performance year; and
++ Drug/biological HCPCS codes that were not included in the baseline period, appear in more than 10 episodes in the performance year, have a mean cost of $25,000 or less per episode in the performance year, and correspond to a drug/biological that appears in the baseline period list but was assigned a ( print page 69723) new HCPCS code between the baseline period and performance year.
++ HCPCS codes for new hemophilia clotting factors not in the baseline period.
We stated in the proposed rule that the complete list of excluded MS-DRGs for readmissions and excluded HCPCS codes for Part B services furnished during TEAM episodes after TEAM beneficiary discharge from an anchor hospitalization would be posted on the CMS TEAM website at https://innovation.cms.gov/initiatives/TEAM ( 89 FR 36417 ). We stated in the proposed rule that the list would apply to all performance years of the model until and unless the list is updated. We proposed that revisions to the TEAM exclusions list would be initiated through rulemaking to allow for public input. Potential updates to the list could include additions to or deletions from the list, reflect changes to ICD-10-CM coding and the MS-DRGs under the IPPS, or address any other issues that are brought to our attention throughout the course of the TEAM performance period.
We sought comment on the proposed excluded services, the TEAM exclusions list, and the process for updating the TEAM exclusions list in § 512.525(f), § 512.525(g), and § 512.525(h).
The following is a summary of the public comments received on the proposed excluded services, the TEAM exclusions list, and the process for updating the list and our responses:
Comment: Several commenters supported the proposal to exclude specific MS-DRGs and high-cost hemophilia drugs, Part B drugs that cost more than $25,000, and products that appear in less than 31 episodes in total.
Response: We appreciate the support received from commenters on our proposed exclusions and are pleased commenters identified these exclusions as the appropriate exclusions to ensure TEAM episodes are comprised of Part A and B items and services that are clinically related to the anchor hospitalization or anchor procedure.
Comment: Several commenters commended CMS for excluding new technology add-on and transitional pass-through payments from the model. Several commenters agree that innovation should be rewarded and certainly not disincentivized. Another commenter acknowledged the role that technology can play to advance clinical goals of TEAM and recommends that CMS ensure TEAM will protect patient access to appropriate and innovative medical devices.
Response: We agree that access to new medical technologies and services should not be withheld from TEAM beneficiaries. We recognize the importance of high value technologies and services that will improve healthcare quality and the lives of Medicare beneficiaries. As discussed in the proposed rule, we proposed to exclude from TEAM new technology add-on payments for drugs, technologies, and services identified by value code 77 on IPPS hospital claims for episodes in the baseline period and performance years ( 89 FR 36417 ). This would mean new technology add-on payments for drugs, technologies, and services would not be included in episode spending or factored into target prices. We believe this exclusion removes any disincentive that a TEAM participant may have to recommend such items and services and may help contribute to the adoption of such items and services.
Also, beneficiary access to medically necessary services and their quality of care are critically important in TEAM. As discussed in section X.A.3.c. of the preamble of this final rule, we are finalizing quality measures for the purpose of evaluating hospitals' performance both individually and in aggregate across the model. Also, as discussed in section X.A.3.i. of the preamble of this final rule, we are finalizing policies and actions to monitor both care access and quality. We believe these features will help ensure that beneficiary access to high quality care is not compromised under the model.
Comment: Several commenters emphasized the importance of utilizing a sufficiently comprehensive list of admissions, readmissions, and services, defined by MS-DRG and HCPCS codes, that would generally be considered unrelated to the trigger episodes and thus excluded. Several commenters further stated that inadequately defining the exclusions within an episode-based model creates frustration and financial burdens. Another commenter stated the exclusions will be especially important in the presence of a 30-day episode. A commenter encouraged CMS to closely review hospital stakeholders' comments on the adequacy of exclusion criteria but acknowledged that TEAM hold participants accountable for the longer-term trajectory of patients' recovery. A few commenters claimed that incorporating a stronger outlier methodology may better exclude the high costs accrued from urgent, emergent, and trauma patients, as well as those patients with unrelated comorbidity complications and high-cost medications, without the need for a specified list of exclusions.
Response: We believe the proposed exclusions list is sufficient to avoid frustration and financial burdens for TEAM participants and that the exclusions list comprehensively captures items and services unrelated to a TEAM episode, even with a 30-day episode. We have several years of experience with these exclusions and their suitability for episodes. As explained in the proposed rule, the exclusions list was developed with significant input from external stakeholders and has been vetted broadly in the health care community ( 89 FR 36416 ).
We also believe that the TEAM participant should only be held accountable for the items and services associated to their attributed TEAM episodes. As such, we disagree with the commenter's recommendation for TEAM participants to be held accountable for care beyond the course of the episode.
Regarding the TEAM outlier methodology, we believe our high-cost outlier cap methodology, as currently designed, does capture unusually high costs from a variety of scenarios, including urgent, emergent and trauma cases, as well as unrelated comorbidity complications that generate catastrophic expenditures during a TEAM episode.
As written, the TEAM exclusions list is designed to capture certain high-cost drugs. CMS recognizes that some drugs, such as hemophiliac blood clotting drugs, incur significant costs which are out of the participant's control. We believe these exclusions adequately support a TEAM participant from being financially impacted by high-cost drugs in their TEAM performance.
Comment: Many commenters recommended that CMS revisit the development of its exclusions list for episodes to ensure participants are only held accountable for care that is truly relevant and clinically appropriate to the episode of care. A commenter stated the current exclusions list is limited in scope and often holds model participants accountable for items and services unrelated to the initial episode of care. A couple of commenters suggested that CMS exclude discharges where patients leave against medical advice or are discharged to hospice. A couple of commenters recommended excluding episodes if a patient is admitted from a congregate care setting (that is, a nursing home), as there is no opportunity to discharge them to an alternate site of care, which is where most savings opportunity will be found in 30-day episodes. A few commenters requested that DME be excluded from episodes due to these services extending ( print page 69724) beyond the 30-days and the ongoing challenge of fraud and abuse around catheters. A commenter also requested that physical therapy be excluded because it also extends past the 30-days.
Response: We acknowledge the importance of holding TEAM participants accountable for services and care related to the beneficiary's TEAM episode. Our proposed exclusions consider many categories of high-cost spending, such as hemophilia blood clotting drugs and certain readmissions, that would be considered unrelated to the beneficiary's episode. We recognize that there are some unique scenarios, such as a patient who leaves against medical advice; however, those situations are not common and thus do not make up enough of a potential risk for participants to warrant exclusion from a TEAM episode.
We also recognize that there will be patients who trigger a TEAM episode that will be admitted and discharged back to a form of congregate care, such as a nursing home. Allowing these services to be included in a TEAM episode aligns with how episode expenditures were calculated in prior models, such as BPCI and BPCI Advanced. From CMS's prior experience in these models, we found model participants were still able to identify and realize savings opportunities through identifying inefficiencies elsewhere in the patient's episode. Therefore, CMS will not be excluding services for beneficiaries when they are admitted and/or discharge back to a congregate care setting.
Finally, we recognize that the Medicare fee-for-service structure sometimes makes claim payment for services spanning a time period that could extend beyond the 30-day episode length. In these situations, CMS will prorate these payments so that only the portion attributable to care during the fixed duration of the episode is attributed to the episode spending. This ensures the TEAM participant is only responsible for the services and associated expenditures for the TEAM episode and not for services beyond the 30-day episode. Please refer to section X.A.3.(b)(5) for more details.
Comment: Several commenters recommended that CMS exclude treatments that are unrelated to the episode such as treatment for substance use disorder or inpatient psychiatric facility services; dialysis, chemotherapy, or other long-term maintenance therapy; patients with a cancer diagnosis, in addition to cancer readmissions; unrelated trauma; auto-immune disorders; previously existing wounds or pressure ulcers requiring ongoing care; and critical care transport (that is, fixed wing helicopter ambulance). A commenter requested that CMS exclude any post-acute care following an excluded readmission, as holding a participant accountable for all patient pathways is unreasonable given how little is known about the causal relationship between the hospital readmission and subsequent post-acute care services.
Response: We recognize that every TEAM episode could incur costs from a variety of items or services rendered, including some the commenters mention above such as cancer treatment, substance use disorder, or post-acute care following an excluded readmission. Although we understand commenters desire to exclude certain services unrelated to a TEAM episode, TEAM was designed to incentivize comprehensive, coordinated, patient-centered care through inclusive episodes. We have provided a thoughtful list of exclusions that adequately capture a variety of high-cost scenarios and we do not believe the TEAM exclusions list should be expanded to include additional items or services. Furthermore, TEAM is designed to encourage participants to make primary care referrals and engage with a patient's aligned total cost of care or shared savings model or program, if applicable. We encourage TEAM participants to work together to engage and collaborate with the patient's primary care provider or aligned total cost of care or shared savings model or program to help support effective management and care coordination of a patient.
Comment: Several commenters urged CMS to ensure that the model does not impede access to drugs that are vitally important but not associated with the surgical episodes included in the model, as the medication exclusions in BPCI Advanced were not adequate. A commenter suggested that CMS exclude any drugs that have a mean cost of more than $25,000 per episode, including drugs for oncology and other conditions. The commenter stated exclusion is necessary due to the high cost of these drugs, combined with the large annual price increases that many high-cost drugs experience. Some commenters recommended that CMS exclude a variety of high-cost drugs, including biologicals, and biosimilars; rare disease drugs; drugs qualifying for pass-through status, including orphan drugs; drugs and biological agents used to treat cancer; and chronic disease medications, such as bone mineral density modifying agents and diabetic medications including GLP-1s.
Response: CMS does not believe the proposed lists of exclusions will impede a TEAM participant's access to vital drugs for their beneficiaries. It is our expectation that TEAM participants will make decisions with optimal patient care in mind and not make decisions based on which drugs may or may not be included in the patient's TEAM episode. We also expect to evaluate the exclusions list and make adjustments, when necessary, through rulemaking to allow for public comment. Based on our experience with the BPCI Advanced model, these exclusions cover a variety of high-cost and rare drugs/biologicals including those used to treat cancer, chronic conditions, etc., and ensure that providers are not discouraged from using drugs that are vitally important to the care of the beneficiary due to fear of financial penalties. Using standard criteria each performance year will allow for the model to maintain consistency in the way low-volume and high-cost drugs are identified year-to-year as providers adopt the use of new drugs/biologicals and previously rare drugs/biologicals get adopted more broadly in later years of TEAM.
Comment: A commenter recommended CMS establish a separate payment for non-opioid pain medications furnished in the inpatient setting to ensure that the shift to the episode-based payment model does not disincentivize clinically appropriate use of novel, non-opioid pain treatments in favor of lower cost generic opioids. The commenter also indicated CMS should exclude any separate payment for non-opioid pain management from the model's episode expenditures to avoid inadvertently discouraging use of these products.
Response: We thank the commenter for their recommendation and will take this into consideration. We acknowledge the benefits of non-opioid pain treatments, which may be associated with less side effects while still providing effective pain management. As an episode-based payment model, TEAM strives to abide to total-cost-of-care principles and include most Medicare Parts A and B items and services into an episode, including drugs. Therefore, we try to limit the items and services excluded from an episode, so that the episode captures most Medicare spending. If we determine a separate payment should be established and excluded from episode costs for non-opioid drugs, we would do so through future notice and comment rulemaking. ( print page 69725)
Comment: Several commenters recommended that CMS engage with stakeholders regarding making updates to the exclusions list, including clinical expert review of potential product exclusions, to ensure TEAM does not disincentivize recommended and necessary care or cause a delay to such services. Several commenters stated that, under TEAM, hospitals will have a tight window in which they are able to control costs and find efficiencies.
Response: We appreciate these comments and the desire to engage with stakeholders to ensure exclusions from TEAM have been vetted by a variety of different experts. As we proposed, the TEAM exclusions list, uses similar exclusions to other CMS Innovation Center Models, with minor adjustments. The exclusions list was developed through a collaborative effort between CMS and external stakeholders and has been vetted broadly in the health care community. We proposed to use the BPCI Advanced exclusions list in TEAM based on several years of experience with these exclusions and their suitability for episodes. As such, we feel confident that our proposed exclusions list has been viewed and discussed by experts who had the opportunity to create a comprehensive, thorough list of exclusions. Additionally, as we mentioned previously, we will consider future modifications to the TEAM exclusions list, as needed, and will use rulemaking to allow for public input.
Comment: Several commenters suggested timeframes for updating the list of excluded items and services. Several commenters stated that the initial period of care and the services provided during anchor stay will be a significant percentage of expenses accrued for a 30-day episode, when compared to other CMS Innovation Center models. For these reasons, they suggested that CMS develop and revisit its cost exclusion criteria at least annually, as is done in BPCI Advanced. A couple of commenters encouraged CMS to consider a semiannual or quarterly update to the list of proposed excluded MS-DRGs for readmissions and proposed excluded HCPCS codes for Part B services for the first performance year.
Response: We appreciate that these commenters want to ensure exclusions from TEAM are as current as possible and evaluated on a regular basis. However, CMS considers quarterly or semiannual updates to be too frequent. Increasing frequency of review to multiple times a year would require an immense increase in resources and CMS does not believe there would be enough gained by frequent reviews, especially in light of CMS Medicare payment rules' schedules which generally only receive updates once annually. We do recognize there may be situations in the future that require changes to the TEAM exclusions list. Should we determine that future modifications to the TEAM exclusions list are needed, we would use notice and comment rulemaking to allow for public comment and review.
After consideration of the public comments we received, we are finalizing our proposed TEAM exclusions list without any modifications. The exclusions list will be posted on the CMS TEAM website at https://innovation.cms.gov/initiatives/TEAM prior to the start of the model.
In the proposed rule, wee proposed to begin an episode with an anchor hospitalization or anchor procedure because of the challenges related to clinical variability leading up to the episodes and identifying unrelated services, given the multiple chronic conditions experienced by many TEAM beneficiaries ( 89 FR 36417 ). We proposed that all services that are included in the IPPS (for example, 3-day payment window payment policies) would be included in the episodes. We further proposed that the population of Medicare beneficiaries whose care would be included in TEAM would be those beneficiaries who meet all of the following criteria at the time of admission to the anchor hospitalization or anchor procedure:
- Enrolled in Medicare Part A and Part B.
- Not eligible for Medicare on the basis of end-stage renal disease.
- Not enrolled in any managed care plan (for example, Medicare Advantage, Health Care Prepayment Plans, cost-based health maintenance organizations).
- Not covered under a United Mine Workers of America health plan, which provides health care benefits for retired mine workers.
- Have Medicare as their primary payer.
We sought comment on the proposed beneficiary inclusion criteria included in § 512.535.
We did not receive any comments on the beneficiary inclusion criteria and are finalizing the proposals without modification.
In the proposed rule, we proposed that, if the beneficiary meets the beneficiary inclusion criteria, an episode would begin when a beneficiary is admitted for an anchor hospitalization or anchor procedure for one of the following MS-DRGs, or by the presence of one of the following HCPCS codes on an outpatient claim (specifically, a hospital's institutional claim for an included outpatient procedure billed through the OPPS)( 89 FR 36418 ):
LEJR MS-DRGs and HCPCS codes—
- 469 (Major Hip and Knee Joint Replacement or Reattachment of Lower Extremity with MCC or Total Ankle Replacement). [ 896 ]
- 470 (Major Hip and Knee Joint Replacement or Reattachment of Lower Extremity without MCC).
- 521 (Hip Replacement with Principal Diagnosis of Hip Fracture with MCC).
- 522 (Hip Replacement with Principal Diagnosis of Hip Fracture without MCC).
- 27447 (Total Knee Arthroplasty).
- 27130 (Total Hip Arthroplasty).
- 27702 (Total Ankle Arthroplasty).
SHFFT MS-DRGs—
- 480 (Hip and Femur Procedures Except Major Joint with MCC).
- 481 (Hip and Femur Procedures Except Major Joint with CC). [ 897 ]
- 482 (Hip and Femur Procedures Except Major Joint without CC/MCC).
CABG MS-DRGs—
- 231 (Coronary Bypass with PTCA with MCC).
- 232 (Coronary Bypass with PTCA without MCC).
- 233 (Coronary Bypass with Cardiac Catheterization or Open Ablation with MCC).
- 234 (Coronary Bypass with Cardiac Catheterization or Open Ablation without MCC).
- 235 (Coronary Bypass without Cardiac Catheterization with MCC).
- 236 (Coronary bypass without Cardiac Catheterization without MCC).
Spinal Fusion MS-DRGs and HCPCS codes—
- 453 (Combined Anterior and Posterior Spinal Fusion with MCC).
- 454 (Combined Anterior and Posterior Spinal Fusion with CC).
- 455 (Combined Anterior and Posterior Spinal Fusion without CC/MCC).
- 459 (Spinal Fusion Except Cervical with MCC).
- 460 (Spinal Fusion Except Cervical without MCC).
- 473 (Cervical Spinal Fusion without CC/MCC). ( print page 69726)
In a correction notice (CMS-1808-CN), we noted that the proposed rule included proposed changes to several of the spinal fusion MS-DRGs that were also included in the proposed definition for the TEAM Spinal Fusion clinical episode category ( 89 FR 35971 ). We stated that if the proposed changes to the spinal fusion MS-DRGs were finalized for FY 2025, we would use the eight new MS-DRGs: MS-DRG 426 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with MCC), MS-DRG 427 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical with CC), MS-DRG 428 (Multiple Level Combined Anterior and Posterior Spinal Fusion Except Cervical without CC/MCC), MS-DRG 402 (Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical), MS-DRG 429 (Combined Anterior and Posterior Cervical Spinal Fusion with MCC), MS-DRG 430 (Combined Anterior and Posterior Cervical Spinal Fusion without MCC), MS-DRG 447 (Multiple Level Spinal Fusion Except Cervical with MCC) and MS-DRG 448 (Multiple Level Spinal Fusion Except Cervical without MCC). In addition, we stated that, if finalized as proposed at 89 FR 35971 , we would use the revised titles for the existing MS-DRGs 459 and 460, “Single Level Spinal Fusion Except Cervical with MCC and without MCC”, respectively.
Major Small and Large Bowel Procedure MS-DRGs—
- 329 (Major Small and Large Bowel Procedures with MCC).
- 330 (Major Small and Large Bowel Procedures with CC).
- 331 (Major Small and Large Bowel Procedures without CC/MCC).
In the proposed rule, we proposed that the episode start date would be the day of the anchor procedure for outpatient procedures or the date of admission on the IPPS claim associated with the anchor hospitalization that triggered the episode ( 89 FR 36418 ). However, as stated in the proposed rule, if an anchor hospitalization is initiated on the same day as or in the 3 days following an outpatient procedure that could initiate an anchor procedure for the same episode category, we proposed to begin the episode on the date of the outpatient procedure rather than the date of the inpatient admission. That is, the outpatient procedure would not initiate an anchor procedure. For example, if a beneficiary undergoes an outpatient TKA and is sent home but is admitted the next day through the emergency department, the episode would be respecified as an anchor hospitalization rather than an anchor procedure with readmission. We proposed this in the proposed rule to ensure we would be able to accurately capture outpatient procedures that may result in admission after a period of observation or shortly after discharge. Moreover, we believe that an inpatient episode should take precedence over an outpatient procedure performed on the same day, given the likelihood of higher spend associated with the inpatient episode and potential for higher clinical acuity.
In the proposed rule, we stated that, although we were not proposing a transfer policy for TEAM, we recognized there could potentially be episodes in TEAM that are initiated as a result of a beneficiary being transferred from one hospital to another, where at least one or both hospitals are TEAM participants and where at least one of the hospital admissions is for an MS-DRG that would initiate an anchor hospitalization in TEAM ( 89 FR 36418 ). In the BPCI Advanced model, this is viewed as one continuous hospitalization, whereas in the CJR model and in the proposed TEAM, it is viewed as two separate hospitalizations that may result in an episode initiating depending on the hospital participation in the model and the MS-DRGs involved in the hospital admissions. Specifically, we stated in the proposed rule if the initial inpatient admission is at a TEAM participant for a proposed MS-DRG in TEAM, then it would initiate an anchor hospitalization and the resulting transfer to the second hospital would not initiate a new anchor hospitalization, rather it would be included in the episode initiated from the first hospitalization. However, if the initial inpatient admission is for an MS-DRG not proposed in TEAM, then an anchor hospitalization is not initiated and the resulting transfer to the second hospital could initiate an episode depending on the second hospital's participation status and the MS-DRG for the inpatient admission.
In the proposed rule, we stated that we considered mimicking the BPCI Advanced model and proposing a transfer policy where a TEAM beneficiary that is transferred from one hospital to another would be considered one continuous hospitalization. Specifically, we considered defining an acute-to-acute hospital transfer as consecutive inpatient stays for a TEAM beneficiary if the admission date of the latter inpatient hospital stay is the same as the discharge date of the initial hospital inpatient stay for different acute care hospitals. In the proposed rule, we stated this would mean that acute-to-acute hospital transfers are treated as one continuous hospitalization and would be assigned the admission date and the hospital from the first leg of the transfer and the MS-DRG and discharge date from the last leg of the transfer. For example, hospital A is a TEAM participant and hospital B is not a TEAM participant. A beneficiary is admitted to hospital A on January 1st for an MS-DRG 637 (which is not a TEAM episode) and discharged on January 5th with a transfer to hospital B on the same day. The beneficiary is admitted to hospital B for MS-DRG 470 (LEJR) and is discharged on January 10th. In this example, the episode is attributed to hospital A and is considered an LEJR episode with an anchor hospitalization start date of January 1st and an anchor hospitalization end date of January 10th. All of the spending between both hospitalizations would be captured in the episode. On the other hand, if hospital A was not a TEAM participant and hospital B was a TEAM participant, then neither hospital would be attributed the episode since hospital A is not a participant and the transfer policy prevents the episode from being attributed to hospital B. We recognized this policy would help keep the initial hospital accountable and may mitigate perverse incentives to transfer a beneficiary; however, it increases complexity for determining when an episode is initiated, and which hospital is accountable for the episode. We also noted that the BPCI Advanced model included additional requirements in their transfer policy, where if one of the hospitals was a critical access hospital or a PPS-exempt cancer hospital or if one of the inpatient admissions was for a MS-DRG on the exclusions list, the episode was cancelled ( 89 FR 36419 ).
We sought comment on our proposal for initiating TEAM episodes based on MS-DRGs or HCPCS codes included in § 512.510 and whether we should consider a transfer policy similar to BPCI Advanced for TEAM.
The following is a summary of the comments received and our responses:
Comment: Several commenters recommended that CMS explore modifying the point at which an episode is triggered and broaden episodes to include pre-operative care or office ( print page 69727) visits related to the procedure, as some episodes of care are planned and start prior to a hospital admission or outpatient procedure.
Response: We appreciate the interest expressed by the commenters in starting comprehensive care coordination prior to the hospital admission, and we recognize that the beneficiary's care which ultimately leads to the procedure that begins a TEAM episode often begins long before the surgical procedure. However, beginning the episode too far in advance of the procedure that initiates the episode would make it difficult to avoid bundling unrelated items, and starting the episode prior to the hospital admission (or outpatient procedure) is more likely to encompass spending that varies widely among beneficiaries, which would make the episode more difficult to price appropriately. In addition, identifying a specific set of related presurgical services to include in the episode, would be of little value in the model because many of the services that are typically necessary or the standard of care prior to a surgical procedure are often included in the IPPS payment (for inpatient episodes) under the three-day payment window payment policies and are therefore already included in the TEAM episodes. We believe that using the date of admission, or date of the outpatient procedure for outpatient episodes, as the start of the TEAM episode is appropriate as hospitals are unlikely to shift related services earlier than when is clinically indicated.
Comment: A commenter stated that it is critical that CMS directly engage relevant practicing physicians in defining episode triggers.
Response: We agree that engaging with providers and other stakeholders is necessary for developing new models and prioritized public outreach throughout model development, including releasing an RFI in July 2023 to gather input and inform the model well before the NPRM was drafted ( 88 FR 45872 ). Throughout the development of this model prior to the drafting of the NPRM, CMS also met with multiple physician associations and additional stakeholders to ensure ample opportunity for the public to contribute to the development of TEAM. We appreciate the time and effort these public groups engaged in to ensure the model team was in receipt of their invaluable input and insight.
We are finalizing our proposal to initiate TEAM episodes with the MS-DRGs and HCPCS codes included in § 512.510. We note that the proposed restructuring of the spinal fusion MS-DRGs is final, with modifications, effective October 1, 2024, for FY 2025. We direct readers to section II.C.6.b. of the preamble of this final rule for a full discussion of the changes. Therefore, if a beneficiary meets the beneficiary inclusion criteria, an episode in the Spinal Fusion episode category would begin when a beneficiary is admitted for an anchor hospitalization or anchor procedure for one of the following MS-DRGs, or by the presence of one of the following HCPCS codes on an outpatient claim (specifically, a hospital's institutional claim for an included outpatient procedure billed through the OPPS):
- 402 (Single Level Combined Anterior and Posterior Spinal Fusion Except Cervical).
We are not finalizing a transfer policy at this time but thank the commenters for their input regarding a potential transfer policy for TEAM. We will take the comments into consideration should we propose a transfer policy through future notice and comment rulemaking.
In the proposed rule, we stated that the proposed episodes would cover time periods marked by significant PAC needs, potential complications of surgery, and short-term, intense management of chronic conditions that may be destabilized by surgery. We believe that hospitals have substantial ability to influence the quality and efficiency of care that TEAM beneficiaries receive over the weeks and months following a procedure. In the proposed rule, we also stated that for this reason, both CJR and BPCI Advanced utilize a 90-day post-discharge episode duration ( 89 FR 36419 ).
However, we stated in the proposed rule that an episode duration longer than 30 days poses greater risk for the hospital because of variability due to medical events outside the intended scope of the model. Our analysis of BPCI Advanced episodes found that the need for care for chronic conditions and other non-anchor MS-DRG-related conditions become much more prevalent during the 31 to 90 days following hospital discharge. Longer episodes also increase the potential for ACO overlap (where a beneficiary aligned or assigned to an ACO has an episode included in TEAM), are associated with a greater number of episode-level exclusions in the post-discharge period, and are more likely to include potential readmissions for an unrelated condition. We also stated that shorter episode lengths are used in other models that employ total cost-of-care approaches. In the Medicare Spending Per Beneficiary (MSPB) measure of the Hospital Value-Based Program (HVBP), episodes include Part A and Part B payments for services furnished three days prior to a patient's inpatient stay and extend for 30 days after discharge.
In the proposed rule, we stated that reducing episode duration to 30 days could both sustain the spending reductions demonstrated in BPCI Advanced and CJR and mitigate some of the current challenges experienced between ACOs, hospitals, and other providers. A 30-day episode would position the specialist as the principal provider near the anchor event with a hand-off back to the primary care provider for longitudinal care management and we believe that ACOs are better equipped to address the population health needs of Medicare beneficiaries. ( print page 69728)
Additionally, we stated that the majority of episode spending occurs in the first 30 days following discharge or the anchor procedure. Based on an internal analysis of BPCI Advanced episodes between 2020 and 2022, seventy-five percent of episode spending occurred in the first 30 days of the episode and 90 percent occurred in the first 60 days. We stated that we expect TEAM to continue to provide hospitals with opportunities to improve care and incentivize coordinated, quality care among acute care hospitals, HOPDs, physicians, and PAC providers throughout care transitions, given that the majority of episode spending during 90-day episodes occurred in the first 30 days.
Based on the rationale noted, we proposed that episodes end 30 days after discharge from the anchor hospitalization or anchor procedure and that day 1 of the 30-day post-acute portion of the episode is the date of the anchor procedure or the date of discharge from an anchor hospitalization. To the extent that a Medicare payment for services included in an episode spans a period of care that extends beyond the episode duration, we proposed that these payments would be prorated so that only the portion attributable to care during the fixed duration of the episode is attributed to the episode spending. The proposal for a 30-day post-discharge episode length is included in § 512.537(a)(1).
We sought comment on our proposal to implement a 30-day post-discharge episode length. We also sought comment on alternative episode durations, such as a 60-day or 90-day post-discharge episode length.
The following is a summary of the public comments received on our proposal to implement a 30-day post-discharge episode length and our responses.
Comment: Many commenters support a 30-day post-discharge episode length for TEAM because they believe it is within the scope of what hospitals can influence, meets the objectives of improving efficiency and reducing variation in cost and outcomes, and captures the majority of post-procedure spend. Several commenters believed that a 30-day episode provides sufficient time for the beneficiary to complete the acute phase of the episode and return to their primary care provider or medical home. Many commenters stated that a shorter episode will help mitigate the risk of chronic or unrelated conditions impacting readmissions, which is more likely to occur with longer episodes. Several commenters agreed that reducing episode duration will mitigate some of the challenges with integrating longitudinal and episodic care between ACOs, hospitals, and other providers and will be an incentive for hospitals to communicate and collaborate with local ACOs, who may welcome the chance to partner with the hospital and manage their patients when discharged to post-acute settings. Another commenter agreed with CMS that a 30-day episode could sustain the spending reductions demonstrated in BPCI Advanced and CJR. Another commenter wrote that it is reasonable and necessary for hospitals to do everything they can while a patient is in their facility to ensure the best possible long-term outcomes. One commenter stated that a 30-day length is consistent with other CMS programs.
Response: We thank the commenters for their support of our proposed 30-day post-discharge episode length. We agree that a 30-day post-discharge episode length provides sufficient time for post-procedure management and care redesign, while limiting the risk of including care for other, unrelated conditions in the episode. We also agree that a 30-day post-discharge episode length promotes our goal of ensuring that episode-based models and ACO initiatives coexist and allow for sufficient financial opportunities and incentives to improve care both during episodes and afterwards.
Comment: A number of commenters believed that a 30-day episode would limit financial opportunity and participants' ability to improve both the quality and efficiency of care furnished during the episode, as a higher proportion of episode costs would be incurred during the hospital stay or procedure. They further noted that this would be most problematic for episodes with the highest-cost index admissions relative to their post-discharge spending, such as CABG and Spinal Fusion. Several commenters stated that given hospitals are paid a MS-DRG payment, any internal cost savings realized during the index admission would not be reflected in their performance and even greater savings would be required during the post-discharge period. Several commenters stated that, although the greatest opportunity is through fewer post-surgical complications and longer-term PAC management, a 30-day episode would deprive participants of vital cost reducing opportunities, as a single post-acute stay may consume the entire episode window. Another commenter expressed concern that the short timeframe will negatively impact patient discharges to the most appropriate PAC setting, in particular to IRFs, which tend to be higher in cost than other PAC providers, whereas a longer episode would reduce the financial pressure to discharge to the lowest cost setting.
Response: We thank commenters for raising their concerns with the 30-day episode window limiting financial opportunity and participants' ability to improve care. In response to the comments, we analyzed the share of anchor and post-discharge spending of total episode spending in 30-day and 90-day episodes using Medicare FFS claims from 2021. CMS acknowledges that the anchor procedure makes up a larger proportion of the total spending in shorter episodes. Mean anchor spending as a percentage of total episode spending is 37 percent for SHFFT and 78 percent for CABG in 90-day episodes, and is 49 percent and 85 percent, respectively, in 30-day episodes. Furthermore, CMS acknowledges the commenters' observation that cost savings during the anchor procedure will not result in lower reimbursement rates for MS-DRGs and HCPCS codes. There are other costs grouped to the anchor period of episodes. However, based on the payment structure and prior evaluation studies for BPCI Advanced and CJR, CMS expects that participants have savings opportunities in the post-discharge period. There are large differences among hospitals with respect to post-discharge spending as a proportion of total spending, in the range of 10-13 percentage points between the 25th and the 75th percentile of the distribution for each episode type. These relatively large differences between hospitals indicate that there are still financial opportunities and a potential for care improvement.
CMS also investigated what the equivalent to a 3 percent discount in a 90-day episode would be in a 30-day episode, assuming that anchor costs were not modifiable. As a result of this investigation, and considering savings opportunities, CMS is finalizing lower discount factors for TEAM episodes than what was proposed ( 89 FR 36433 ). Specifically, we are finalizing a 2 percent discount factor for the LEJR, SHFFT, and Spinal Fusion episode categories and a 1.5 percent discount factor for the CABG and Major Bowel Procedure episode categories. We direct commenters to section X.A.3.d.(3)(g) for further discussion on the discount factor.
CMS also acknowledges the commenter's concern with the trade-off between discharging patients to the most appropriate or cheapest post-acute care settings. CMS plans to monitor ( print page 69729) costs of episodes with IRF utilization. If we determine additional risk adjusters are necessary to improve pricing accuracy, they would be added pursuant to notice and comment rulemaking.
Comment: Several commenters stated that, due to data lag in claims-based models, a shorter episode does not provide enough time for participants to identify that their beneficiaries are included in a bundled payment model. They stated that the episode would end before participants receive data on post-acute care because hospitals typically do not receive claims data within a 30-day window.
Response: Because the clinical episodes included in TEAM will be procedure-based, we believe that TEAM participants should generally be able to identify beneficiaries that will be included in TEAM episodes when the beneficiaries are admitted to the hospital. In fact, this is referenced in section X.A.3.i.(2) where we finalize provisions requiring TEAM participants to notify beneficiaries about their inclusion in a TEAM episode. In addition, we know from prior and current model tests that one of the main mechanisms in which hospitals engage in care redesign in an episode-based payment model has to do with planning for care post-discharge and the selection of and planning for post-acute care (whether in a facility or at home). This care redesign and planning occurs prior to the hospital receiving any relevant data on post-acute care for the episodes in question; generally, the participant does not receive data from the episode until it has concluded, even in models with a longer episode duration. We also note that, as discussed in section X.A.3.k. of this final rule, CMS will be providing comprehensive episode and claims data to TEAM participants that request such data on a monthly basis. TEAM participants will be able to use this data to identify historical spending patterns (using the baseline data) and patterns of care during their performance in the model more generally. We know from operating other bundled payment models that model participants can, and often do, use this data to inform strategies for care redesign, regardless of exact episode duration.
Comment: Several commenters believed that a 30-day episode is not sufficient for capturing clinical outcomes and assessing quality. A commenter pointed out that quality measures already used in CMS programs demonstrate, with clinical evidence, that a longer window is necessary. Another commenter pointed to the 90-day CABG mortality measure as evidence for the appropriateness of a longer episode. Several commenters stated it is impossible to meaningfully analyze the quality of a spine fusion operation so soon after surgery, particularly if the aim of measuring quality is to determine if the operation achieved the surgeon's or patient's stated goals for undergoing the operation; the ultimate outcome of a spinal surgery such as fusion may not be measurable for one to two years in terms of the adequacy of the fusion and the impact it may have on the adjacent levels of the spine. A commenter claimed that a 30-day episode length is insufficient to capture the relevant costs needed to bring patients back to functional independence. Another commenter noted that recovery time for the procedures included in the model can vary widely and a 30-day episode duration will make it difficult for providers to address social risk factors and effectively identify high-risk patients. Several commenters stated that providing quality care for conditions that are directly linked to TEAM episodes, such as osteoporosis and the development of opioid use disorder resulting from a discharge prescription, would likely occur well beyond the acute episode and could not be determined within a 30-day episode. Several commenters stated that the narrowing of episodic scope weights the focus of TEAM almost entirely on the cost of surgical procedures and acute surgical complications and removes patient outcomes, as they will not be known for months after the episode. Another commenter stated that that the 30-day episode length is too short to account for the true timeline of when patients are seen for post-acute care visits from the treating surgical team. Another said it would be difficult to capture meaningful information on the success of a knee replacement in a 30-day episode, as it is the avoidance of complication, reoperations, and long-term functional outcomes that will truly show success.
Response: In designing TEAM, we have attempted to create financial and quality accountability, across the range of potential outcomes and clinical scenarios that occur during and after the procedures included in the model, while ensuring that we do not hold providers accountable for financial and quality outcomes that are not directly related to the anchor procedure that initiated an episode.
We agree that in many cases, the patient's outcome and recovery from surgery (or return to functional independence) may not be fully realized during the timeframe of the episode. However, our analysis of TEAM episode types in the BPCI Advanced model found that the plurality of IRF, SNF, and LTCH stays start and end within the first 30-days of the post-discharge period. So, we believe 30 days is sufficient to capture the most relevant post-acute care visits directed by the surgical team. As proposed, providers would be held accountable for the cost of revisions or reoperations of the same procedure if it occurs within 30 days of the initial procedure through the inclusion of Part A and B related costs in the initial episode or through the triggering of a second episode if it occurs after 30 days.
We have attempted to balance our desire to encourage care redesign of the acute episode and time period immediately after the hospitalization, during which we know the majority of spending historically occurs, and the most intensive post-acute care and follow-up post-procedure occurs, and our desire to simultaneously encourage longer-term, longitudinal management of beneficiaries through other initiatives, such as ACOs. For this reason, we are finalizing several policies with respect to other care management and care redesign efforts that may occur outside of the scope of TEAM episodes. We refer readers to sections X.A.3.l. and X.A.3.e.(3) of this final rule, where we discuss our final policies to require TEAM participants to refer beneficiaries to a primary care provider at the conclusion of a TEAM episode (or at hospital discharge, as applicable), and to allow for beneficiaries aligned to an ACO to initiate TEAM episodes. We believe this addresses the concerns of the commenters who stressed the importance of longer-term patient management and care beyond the scope and duration of the TEAM clinical episodes.
With respect to the timeframe for the quality measures included in TEAM, we believe that it is reasonable to include quality measures in TEAM that have timeframes that differ from the exact time period for financial accountability under the model (that is, 30 days post-discharge). We are committed to aligning our selected measures with those already in use in other required quality reporting programs where possible, and, as such, have limited measure options from which to choose. In addition, while the TEAM episode timeframe may not fully align with a particular quality measure, we believe the underlying goal of the financial and quality accountability under the model is the same: to improve the quality of care provided to beneficiaries and ( print page 69730) choose the most efficient care that is reasonable and safe for the patient.
We disagree that a 30-day episode duration makes it difficult for providers to address social risk factors and effectively identify high-risk patients, and encourage TEAM participants to screen beneficiaries for health-related social needs, or HRSNs, as discussed in section X.A.3.f.(5)(c).
Finally, with respect to patient outcomes, we note that we are finalizing at X.A.3.c.(3)(c) the inclusion of a patient-reported outcomes measure for the LEJR episode and have indicated our interest in the potential inclusion of additional PROMs for other TEAM clinical episodes in the future. We agree with the commenters who emphasized the importance of measuring longer-term functional status and patient outcomes for beneficiaries included in TEAM episodes; we believe the best avenue in which to do that is through quality accountability over a longer time period, not an extension of the 30-day post-discharge period.
Comment: We received many comments in support of longer episode lengths. A commenter expressed concern that CMS' proposal of a 30-day episode represents a significant departure from previous alternative payment models, as both the BPCI Advanced and CJR models utilize 90-day episodes. Another commenter in support of 90-day episodes believed CMS should keep the 90-day episode structure that has proven effective in the CJR and BPCI Advanced models, which would enable robust evaluation and continuity with prior models. Other commenters supported a 90-day episode for spinal fusion and CABG because of the higher proportion of spend for the index procedure.
Response: We thank the commenters for their feedback and suggestions. While commenters have pointed out the success we have had with other episode-based payment models with 90-day episodes, such as CJR and BPCI Advanced, we know from those models that the majority of episode spending occurs in the earlier part of the episode, that is, the hospitalization and first 30 days, not in the ensuing 30 or 60 days (for 60 or 90-day episodes, respectively). In addition, we have stated in section X.A.3.e.(3) of this final rule our desire to complement and encourage the coexistence of episode-based payment models like TEAM alongside more longitudinal, population-based initiatives, such as ACOs. We believe that the best way to do that, without encroaching on the accountable care entity's interest in managing the care for beneficiaries with chronic conditions or care needs that continue beyond the length of the TEAM episode, is to limit TEAM episode financial accountability to a 30-day post-discharge timeframe. By doing so, we can simultaneously encourage TEAM participants to actively manage and plan for post-acute care, prevent readmissions, and otherwise manage follow-up care immediately post-hospitalization, while also allowing for the accountable care entity to “own” the financial accountability for aligned beneficiaries after the acute episode and immediate post-discharge period ends.
Comment: We received many comments in support of variable episode lengths for each clinical episode category. Many commenters encouraged the establishment of episode lengths specific to each clinical episode, so the duration is more reflective of actual opportunities for savings for participants, the clinical needs of patients, and distinct patterns of post-operative care. A couple of commenters recommended tailoring the episode duration to the specific set of MS-DRGs covered in the model; specifically, thirty days for LEJR but something longer for SHFFT, to reflect when the hospital and its surgical team are primarily responsible for the patient's care management. Another commenter noted that total cost of care extends well beyond the episode and that costs in the two years after joint replacement surgery are twice that of the surgical procedure.
Response: We believe a singular episode length for all TEAM episodes will reduce confusion among TEAM participants with regard to recognizing the beginning and end of an episode, analyzing claims data provided to them under the terms of the model, and implementing care redesign strategies focusing on discharge planning, post-acute care planning, and follow-up care, including referral to primary care providers as applicable.
While we appreciate the commenters' arguments in favor of variable episode lengths, we refer readers to our discussion in section X.A.3.e.(3) about our desire to implement TEAM in a way that complements other CMS initiatives focusing on longer-term population-based care, like ACOs.
We recognize that the episodes we are including in TEAM are clinically distinct, as are the beneficiaries that will be included in such episodes. We considered variable lengths for the clinical episodes but ultimately did not propose such a policy because we wanted to limit confusion for providers, and recognized that even within a single clinical episode, there will be meaningful differences between beneficiaries with regard to functional status, post-surgical complications, and other clinical considerations. We believe that a 30-day post-discharge episode length strikes the balance of financial accountability for the majority of spending during and immediately after the procedures included in the model, while not extending the episode so long as to encroach upon potential activities by other entities providing care for beneficiaries included in TEAM, such as ACOs or primary care providers.
Comment: Many commenters objected to a shorter episode combined with the proposed 3 percent discount factor, and the commenters believed that together they would create the most aggressive financial target that CMS has ever adopted in either a mandatory or voluntary episode-based model. A commenter asserted that the bands between benchmark spending and quality and the savings and loss thresholds will fall too narrowly to provide adequate opportunity for success by model participants. Many commenters requested that CMS either adopt a 90-day episode or reduce the discount.
Response: We refer commenters to section X.A.3.d.(3)(g) of this final rule, where we discuss the final discount factors for the episodes included in TEAM.
After consideration of the comments, we are finalizing our policy for a 30-day post-discharge episode length at § 512.537 without modification.
In the proposed rule, we proposed that, similar to the CJR model, once an episode begins, the episode would continue until the end of the episode, unless the episode is canceled because the beneficiary ceases to meet any of the general beneficiary inclusion criteria described in section X.A.3.b.(5)(b) of the preamble of this final rule ( 89 FR 36419 ).
In the proposed rule, we stated that we believe it would be appropriate to cancel the episode when a beneficiary's status changes during the episode, such that they no longer meet the criteria for inclusion, because the episode target price reflects full payment for the episode, yet we would not have full Medicare episode payment data for the beneficiary to reconcile against the target price.
In the proposed rule, we proposed to cancel the episode if a beneficiary dies during the anchor hospitalization or anchor procedure, rather than at any point during the post-discharge period ( print page 69731) of the episode, as is done in BPCI Advanced. As discussed in the CJR Final Rule, we believe there would be limited incentive for efficiency that could be expected when death occurs during the anchor hospitalization itself ( 80 FR 73318 ).
As discussed in the Episode Payment Model proposed rule, we consider mortality to be a harmful beneficiary outcome that should be targeted for improvement through care redesign for these clinical conditions. We do not believe that it would be appropriate to exclude beneficiaries from episodes who die any time during the episode ( 81 FR 50841 ).
Instead, in the proposed rule, we proposed to maintain beneficiary episodes in TEAM unless death occurs during the anchor hospitalization or anchor procedure. We proposed that when a beneficiary dies following discharge from the anchor hospitalization or anchor procedure, but within the 30-day post-hospital discharge episode period, we would calculate actual episode spending and reconcile it against the target price. We believe this would encourage TEAM participants to actively manage beneficiaries to reduce their risk of death, especially as death would often be preceded by expensive care for emergencies and complications. Therefore, we proposed to cancel episodes for death only during the anchor hospitalization or anchor procedure.
In the proposed rule, we stated that, if a beneficiary is admitted to the hospital on the same day as or within 3 days of an outpatient procedure that could have initiated an anchor procedure for the same episode category, the procedure would not initiate an anchor procedure. Rather, the admission would initiate an anchor hospitalization with a start date corresponding to the outpatient procedure. We proposed this policy because we believe that an inpatient episode should take precedence over an outpatient procedure performed on the same day, given the likelihood of higher spend associated with the inpatient episode and potential for higher clinical acuity.
Finally, we proposed that episodes subject to extreme and uncontrollable circumstances (EUC) would be canceled, meaning that the services associated with the episode would continue to be paid through Medicare FFS, but the episode would not be reconciled against a target price. We proposed to base the TEAM EUC definition on the definition finalized in the CJR 2018 Final Rule ( 83 FR 26604 ), which was designed to address the extreme and uncontrollable costs associated with natural disasters such as hurricanes, flooding, and wildfires. Specifically, we proposed that the EUC policy would apply to TEAM participants located in a county where both: (1) a major disaster has been declared under the Stafford Act; and (2) section 1135 waivers have been issued. In the proposed rule, we stated that we believe that it is appropriate for our EUC policy to apply only in the narrow circumstance of a major disaster, which is catastrophic in nature and tends to have significant impacts on infrastructure, rather than the broader grounds for which an emergency could be declared. In regard to determining the start date of episodes to which the EUC would apply, we stated our belief that episodes initiated during an emergency period or in the 30 days before the start date of an emergency period (as defined in section 1135(g) of the Act) should reasonably capture those beneficiaries whose high episode costs could be attributed to extreme and uncontrollable circumstances ( 89 FR 36420 ).
In summary, we proposed that the following circumstances would cancel an episode:
- The beneficiary no longer meets the criteria for inclusion.
- The beneficiary dies during the anchor hospitalization or anchor procedure.
- The participating hospital is subject to the EUC policy.
In the proposed rule, we proposed that when an episode is canceled, the services furnished to beneficiaries prior to and following the episode cancelation would continue to be paid by Medicare as usual but there would be no episode spending calculation that would be reconciled against the TEAM target price (see section X.A.3.d.(5)(f) of the preamble of this final rule). As discussed in section X.A.3.h. of the preamble of this final rule, waivers of program rules applicable to beneficiaries in episodes would apply to the care of beneficiaries who are in episodes at the time the waiver is used to bill for a service that is furnished, even if the episode is later canceled.
We sought comment on our proposals to cancel episodes once they have begun but prior to the end of the 30-day post-discharge period included in § 512.537(b).
The following is a summary of the public comments received on our proposals to cancel episodes and our responses:
Comment: We received a comment regarding the need to ensure planned staged procedures are not counted as a readmission for those patients whose clinical case calls for this type of surgery. The commenter stated that surgeons may stage surgeries as part of the care plan, when appropriate and in the best interest of patient safety. That is, they would perform one surgery on one date and follow up with a second surgery at a future date. The commenter stated that if the first surgery initiates an episode and the second surgery is counted as a readmission, the target prices would not accurately reflect the episode and would penalize the participant hospital for caring for the patient in the safest manner possible.
Response: We appreciate the comment related to staged procedures and the need to account for planned subsequent admission for TEAM episodes from the same clinical episode category during the 30-day discharge period. We did not propose to cancel one of the episodes when two episodes overlap one another, such as in planned staged procedures ( 89 FR 36419 ). Both CJR ( 42 CFR 510.210(b) ) and BPCI Advanced have policies for such an occurrence, where the first episode is canceled, and a new episode begins. However, both CJR and BPCI Advanced have a 90-day episode length.
We recognize that there may be instances where a beneficiary has a subsequent admission for a TEAM episode from the same clinical episode category during the 30-day discharge period, such as a knee replacement on the contralateral knee. However, assuming the need for beneficiaries to be medically optimized before undertaking a second procedure, our belief is that such occurrences will be infrequent within TEAM's shorter 30-day episode. We will further analyze the frequency and circumstances of such occurrences (for example, how often one episode of the same clinical category supersedes another, and the circumstance in which this occurs) to determine if this policy needs to be changed in future rule making.
Comment: We received a couple of comments asking CMS to exclude from TEAM any episode during which a patient expires during the 30-day post-discharge period. Commenters stated that this would be appropriate to account for patients that expire post-surgery because of conditions that are unrelated to the surgery itself. These commenters also believed that CMS should align its episode cancelation policy with the policy under the CJR model, which cancels an episode if a death occurs at any time during the episode, rather than just during the anchor admission or procedure. A ( print page 69732) commenter also stated that this cancelation policy is particularly important, as the model looks to include more safety net providers and address health inequity and may include patients with more complex conditions unrelated to the surgery.
Response: We appreciate comments pertaining to the policy of how to handle episodes in the event of a beneficiary death. We disagree that an episode should be canceled and excluded from TEAM reconciliation, if a patient dies at any point during the 30-day post discharge period. We consider mortality to be a harmful beneficiary outcome that should be targeted for improvement and encourage TEAM participants to actively manage beneficiaries to reduce their risk of death, especially as death would often be preceded by expensive care for emergencies and complications. We believe beneficiaries with heightened needs and acuity would benefit greatly from the care coordination and improved care transitions incentivized under the model.
As discussed in the Episode Payment Model proposed rule ( 81 FR 50841 ), death within the 30 days following hospital discharge would not be rare for certain clinical conditions in TEAM, such as CABG and SHFFT, and could appropriately be targeted for improvement through care redesign. We note that in the case of a 90-day episode, such as in CJR, death occurring later in the episode is less likely to be attributed to the anchor procedure or hospitalization procedure than a death occurring within 30 days. For this reason, we do not believe aligning the TEAM policy with CJR is necessary.
Therefore, we believe that the proposed policy of only canceling those episodes when a beneficiary dies during the anchor procedure, and not canceling those episodes where a beneficiary dies during the 30-day post discharge period, best aligns with the model's greater priorities and incentives, and encourages providers to offer high quality and coordinated care to beneficiaries, including those beneficiaries who may have multiple complex conditions with a high risk of mortality.
Comment: A commenter supported the cancelation of episodes subject to extreme and uncontrollable circumstances (EUC), as it helps health care organizations during climate-related disasters, which are out of their control.
After consideration of the public comments we received, we are finalizing without modification our proposals to cancel certain TEAM episodes once they have begun but prior to the end of the 30-day post-discharge period.
As discussed in the CJR model final rule ( 80 FR 73358 ), Medicare payment policy has moved away from FFS payments unlinked to quality of care. Through the Medicare Modernization Act and the Affordable Care Act, we have implemented specific IPPS programs like the Hospital Inpatient Quality Reporting (IQR) Program (section 1886(b)(3)(B)(viii) of the Act), the Hospital Value-Based Purchasing (VBP) Program (subsection (o) of section 1886), the Hospital-Acquired Condition (HAC) Reduction Program (subsection (q) of section 1886), and the Hospital Readmissions Reduction Program (subsection (p) of section 1886), where payment reflects the quality of care delivered to Medicare beneficiaries. The CJR model similarly incorporates pay-for-performance, offering TEAM participants the potential for financial reward based on quality performance or, in some cases, quality improvement. Through the use of quality measures, CMS is also able to pursue objectives beyond resource alignment, such as the development of new quality measures and performance indicators. [ 898 ] Additionally, CMS may incorporate new quality measures, re-evaluate, or improve existing quality measures, or adjust a quality measure set to take effect at the start of each Model Year, or at other times specified by CMS.
We believe that episode payment models such as the proposed TEAM should include pay-for performance methodologies that incentivize improvements in patient outcomes while simultaneously lowering health care spending. We also believe that improved quality of care, specifically achieved through coordination and communication among providers, patients, and their caregivers, can favorably influence patient outcomes. We proposed that TEAM would incorporate quality measures that focus on care coordination, patient safety, and patient reported outcomes (PROs) which we believe represents areas of quality that are particularly important to patients undergoing acute procedures. Finally, wherever possible, we would align TEAM quality measures with those used in ongoing models and programs to minimize participant burden. Our goal is to focus on improving beneficiary quality of care and capture meaningful quality data for use in the TEAM pay-for-performance methodologies.
We are starting with a parsimonious set of quality measures that are being tied to payment and plan to incorporate more PRO-PMs in the future of the model. We recognize that there are some gaps in the proposed measures with respect to post-acute care settings and limited measures for episode-specific PROs. We considered including generic PRO data to support the collection and reporting of PROs, similar to the CJR model requiring voluntary submission of the Veterans RAND 12 Item Health Survey (VR-12) or Patient-Reported Outcomes Measurement Information System (PROMIS) Global-10 generic PRO survey. However, we recognize PRO collection and reporting may increase participant and patient burden and we do not want to impose this on TEAM participants for generic PRO data since it may be less clinically meaningful to the episodes that would be tested in TEAM. We will continue to assess the evolving inventory of measures and refine measures based on public comments, changes to payment methodologies, recommendations from TEAM participants and their collaborators, and new CMS episode measure development activities.
In the proposed rule, we stated that the proposed TEAM's quality measures would be scored according to the methodology described in section X.A.3.d.(5)(e) of the preamble of this final rule to calculate the CQS. The CQS would be combined with the TEAM participants' reconciliation amount, as specified in section X.A.3.d.(5)(g) of the preamble of this final rule, during the reconciliation process to tie quality performance to payment.
While we believe the proposed measure set would provide CMS with sufficient measures to monitor quality, and to calculate scoring on quality performance, we may adjust the measure set in future performance years by adding new measures or removing measures, if we determine those adjustments to be appropriate at the time. We note that a selection of these measures may be used for evaluation purposes as well. Prior to adding or removing measures for monitoring quality and calculating scores for quality performance, we would use notice and comment rulemaking. ( print page 69733)
The following is a summary of the public comments received on the proposed TEAM quality measures and its impact on other various quality requirements and our responses to these comments:
Comment: Some commenters are in support of the proposed quality measures in TEAM. Commenters mention they greatly appreciate that CMS is utilizing three proposed quality measures which are currently collected and evaluated as part of the Hospital Inpatient Quality Reporting Program, in part because of the limited increase in administrative burden. Commenters note it is important to not increase the administrative burden on hospitals, clinicians, and staffs when possible. A few commenters showed strong support of PROMs being incorporated in TEAM. However, commenters warn CMS might need to provide some additional technical assistance on a case-by-case basis for PRO reporting. Additionally, a commenter suggested that TEAM incorporate the Patient Activation Measure (PAM) to help identify readmission risks, supporting discharge planning, surgical episodes of care, transitions of care, and improving outcomes while reducing costs. Lastly, a commenter agreed that enhancing patient safety should be a top priority in TEAM. This commenter suggested that TEAM adopt the Patient Safety Structural measure (MUC2023-188) that was proposed in the 2023 MUC list within the IQR.
Response: We would like to thank each commenter for their support of the proposed quality measures in TEAM. CMS placed a strong emphasis on wanting to propose measures we believe do not increase the administrative burden on hospitals, clinicians, and staff. CMS will work to ensure all hospitals within TEAM are fully prepared prior to the model's launch date. Lastly, we'd like to thank the commenters who suggested the Patient Activation Measure and the Patient Safety Structural Measure (MUC2023-188). We believe the CMS Patient Safety and Adverse Events Composite (CMS PSI 90) is more appropriate for the episodes we've proposed in TEAM because it includes a broad array of safety events, many of which are relevant to patients in the episodes, with the added benefit of being familiar to hospitals and not introducing additional administrative burden. We will take this commenter's support into account for the inclusion of these two measures and corresponding attestation requirements in future rulemaking.
Comment: A few commenters mentioned their appreciation of CMS using measures that would be reported following existing Hospital Inpatient Quality Reporting (IQR) program processes to reduce reporting burdens. However, commenters mention concerns over the proposed measures being used in other pay-for-performance programs, which could result in duplicative penalties for TEAM participants. A commenter cautions CMS from adopting the three measures into TEAM until hospitals have had time to report the measures for several years under the Hospital IQR Program. Lastly, a few commenters mention concerns since the proposed measures within TEAM will be in their first year of reporting.
Response: We would like to thank the commenters for their appreciation of the proposed quality measures in TEAM aligning with the Hospital IQR program. CMS is aware of the concerns and reporting challenges that commenters have shared. However, we want to clarify that Hospital IQR is a pay-for-reporting program not pay-for-performance. Where possible, we aimed to align with existing quality improvement efforts and selected measures that were not already included in performance-based payment programs, with the exception of PSI 90 which will only be included in TEAM's first Performance Year. CMS recognizes the importance of all-cause readmissions and patient safety in hospitals and the increased emphasis on these broadly applicable, valid, and consensus-entity endorsed measures through inclusion in the Hospital IQR program and TEAM may further incentivize focus on these important areas of care, while placing minimal burden on TEAM participants. We will continue to assess the evolving inventory of measures and refine measures based on public comments, changes to payment methodologies, recommendations from TEAM participants and their collaborators, and new CMS episode measure development activities.
Comment: Some commenters voiced their concerns about the administrative burden to report on certain measures and for CMS to consider the resource allocation and administrative burden that providers, especially hospitals, would have to take on under the TEAM. TEAM participants, as well as the individual health care professions involved in the model, will have to increase their efficiency of care and accuracy of discharge recommendations while collaborating as a team to ensure that the patient received the right services in the right order at the right time. A commenter mentions that TEAM will require that significant hospital procedural modifications are put into practice for all providers who are involved. Another commenter suggested that CMS to refrain from implementing any mandatory documentation, record keeping, or reporting burden that exceeds a minimal level. If CMS would choose to implement such requirements, we would encourage CMS to align closely any required reporting with administrative work already being done. A commenter suggested that CMS ensure that there is an easy way for TEAM participants to port all of their ENERGY STAR TM data to CMS Innovation Center, rather than having to do it independently. This will drastically lessen the administrative burden of the program. This commenter strongly recommended using as much of the data tracking in portfolio manager as we can, and NOT require any additional reporting to CMS Innovation Center. Lastly, a commenter suggested that CMS should create educational resources that help providers make the case to patients for why these data are being requested, and for what purposes they will be used. The process of improving patient-reported data requires a foundation of trust. We encourage CMS to consider its role in addressing this need.
Response: We are appreciative of commenters sharing their concerns around various reporting burden challenges. TEAM proposed the said quality measures within TEAM to align with measures being used in other models and pay-for-performance programs to minimize participant burden. Our goal is to focus on improving beneficiary quality of care and capture meaningful quality data for use in the TEAM pay-for-performance methodologies. Additionally, the proposed quality measures in TEAM will already be mandatorily reported within the Hospital IQR and Hospital-Acquired Condition Reduction Programs. CMS believes participants will have familiarity with reporting on these measures prior to the start of TEAM. Also, we recognize that hospitals will be newly adapting to the Hospital IQR Program requirement for the THA/TKA PRO-PM but that infrastructure and process development should make the incorporation of future PRO-PMs less burdensome. CMS is aware of the educational resources request and will take into account commenters feedback and ensure that participants in TEAM will be well informed prior to the beginning of the model.
Comment: A few commenters shared their concerns over the reporting ( print page 69734) barriers and administrative burden for hospitals required to participate in TEAM. A commenter asked CMS to ensure that the value of certain policies is on the initiation of timely care, and that these policies are adequately enforced to maximize these outcomes. For instance, the proliferation of Medicare Advantage (MA) plans has been widely considered in annual MA rulemaking, specifically concerning the impact of internal coverage criteria that MA plans use to deny care in post-acute settings. Commenters stated that other activities may already be ongoing in some or most hospitals, but a formal demo-like TEAM with strong financial incentive features, will require more of this activity than is typically taking place leading to additional administrative burden. Lastly, a commenter stated that TEAM as proposed would add significant risk and burden to some of the most distressed hospitals at time when they—and especially non-profit community hospitals who are mostly serving Medicare and Medicaid beneficiaries—have no capacity to take on new risks and burden. CMS Innovation Center must make this model, if mandatory, easier to implement, less risky to manage, and more aligned with other work than voluntary models.
Response: We appreciate commenters voicing their concerns over the proposed TEAM quality measures. These quality measures that were proposed in TEAM align with measures being used in other models and pay-for-performance programs to minimize participant burden. Our goal is to focus on improving beneficiary quality of care and capture meaningful quality data for use in the TEAM pay-for-performance methodologies. CMS is aware of the educational resources request and will take into account commenters feedback and ensure that participants in TEAM will be well informed and ready to implement the various TEAM requirements prior to the model's start date.
Comment: Many commenters suggested that TEAM mimic BPCI Advanced which used an administrative (claims-based) measure set and allowed for hospitals to utilize instead an alternative measure set, which frequently made use of registry data, for quality measures. Commenters specifically mention that registry-based metrics are especially useful for engaging specialists and aligning value-based care programs across payers and provide more episode-specific data than the proposed measures. A few commenters suggested episode specific measures such as, Patient-Centered Surgical Risk Assessment and Communication (QPP #358) and the CABG Composite Score (CBE #0696) for specific TEAM episodes categories. Additionally, a commenter mentioned that CMS should include the Advanced Care Plan measure in TEAM to better support person centered care.
Response: We acknowledge there is rich clinical quality data available in clinical data registries, which have an important place in the history of quality measure development. Our aim was to use quality measures that all hospitals would have access and experience with hence why we recommended Hospital IQR measures for the majority of the performance years in TEAM. Introducing new reporting functions and requirements in a mandatory model would create for additional burden especially on hospitals who are not a member of these specialty societies and those who are not reporting on these alternative quality measures. CMS will take into consideration the suggested episode specific measures for TEAM and will propose any new measures in rulemaking.
Comment: A commenter voiced their concerns with the proposed measures within TEAM not aligning with other pay for performance programs. Measures most often included in the core set are primary care centric and may not be appropriate for episodic or specialty care models. As a result, a commenter cautions CMS in its evaluation of measures for inclusion under an episodic model to ensure whatever measures are selected are appropriate and relevant to the model.
Response: We appreciate commenters voicing their concerns with the proposed measures in TEAM. CMS recognizes the difficulties associated with patient reported outcome and hospital wide measures and the underlying data collection tools used in a clinical domain; however, we decline the requests to remove the proposed measures from TEAM because hospitals will have familiarity reporting on the measures and we believe that providing Team participants with measures that are already incorporated within the Hospital IQR and HAC Reduction Program will lead to less burden. Additionally, the measures align with CMS quality goals and support. We will consider incorporating episode-specific measures where applicable in future rulemaking.
Comment: A commenter recommended that evaluations should include quality measures that reflect patients' health and functioning period beyond the 30-day post discharge period. For many individuals, the recovery period extends well beyond 30 days. We recommend aligning the quality measurement period with the Part A deductible period of 60 days post discharge.
Response: We appreciate commenters recommendation to include measures that reflect a patients' health beyond the 30-day post discharge period. CMS will take these recommendations into consideration for future rulemaking.
Comment: A commenter mentioned that we should study whether surgical-related prehabilitation services and care should be included in future TEAM policy. The commenter said that many studies have demonstrated, and hospital perioperative services know, the value anesthesiologists provide in preoperative assessment clinics and that their actions are key to setting patient expectations, reducing patient length of stay, and encouraging patients to take better ownership of their health. Additionally, this commenter mentions that they recognize many hospitals are unable to provide such preoperative services, but perhaps collecting information on such services would acknowledge the importance of those services in reducing costs, improving care, and allocating resources based upon patient needs.
Response: We appreciate this commenter's input regarding pre-hospitalization. We will take this commenter's suggestion in future consideration. Any addition of future measures that focus on surgical-related prehabilitation services would be done through future rulemaking.
After consideration of the public comments we received, we are finalizing our proposals for the proposed measures that are being used within the Hospital IQR and HAC Reduction programs without modification in our regulation at § 512.547.
As proposed, TEAM is designed to provide financial incentives for improving coordination of care for beneficiaries. We expect care redesign activities to reduce post-surgical complications and hospital readmissions and enhance patient experience and outcome. Furthermore, we acknowledge that achieving savings while continuing to ensure high-quality care for Medicare FFS beneficiaries will require close collaboration among hospitals, physicians, PAC providers, and other providers. In order to encourage greater care collaboration among the providers of TEAM beneficiaries, we proposed three measures as described in section ( print page 69735) X.A.3.c.(3) of the preamble of this final rule. These measures would be used to determine hospital quality of care and eligibility for a TEAM reconciliation payment.
The measures we proposed are—
- For all TEAM episodes: Hybrid Hospital-Wide All-Cause Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356).
- For all TEAM episodes: CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135); and
- For LEJR episodes: Hospital-Level Total Hip and/or Total Knee Arthroplasty (THA/TKA) Patient-Reported Outcome-Based Performance Measure (PRO-PM) (CMIT ID #1618).
Beginning in PY 1 and continuing for the duration of the model, we proposed to adjust reconciliation amounts by the TEAM participants' CQS based on their performance of quality measures previously listed.
We initially proposed these three quality measures due to their: (1) Alignment with the goals of TEAM; (2) hospitals' familiarity with the measures due to their use in other CMS hospital quality programs, including the Hospital IQR and HAC Reduction Programs; and (3) alignment to CMS priorities, including the CMS National Quality Strategy which has goals that support safety, outcomes, and engagement. We believe the three quality measures we proposed to link to payment reflect these goals and accurately measure hospitals' level of achievement on such goals.
We note that shared-decision making (SDM) is an important aspect of care around elective procedures, including elective procedures captured in episodes such as the LEJR episode and Spinal Fusion episode. Use of SDM prior to episode initiation can serve as an important tool to ensure appropriate care. SDM allows the clinician and patient to have informed discussion about treatment options, balancing the risks and expected outcomes with a patient's preferences and values, and can help contribute to ensuring appropriate use of procedures and minimization of low value care. CMS has taken steps to incorporate SDM in care pathways, such as requiring SDM interaction prior to ICD implantation for certain patients for national coverage determinations. [ 899 ] However, implementing SDM in episode-based payment models such as TEAM poses challenges with respect to the timing of the patient/provider interaction and when an episode is initiated. While there are upstream opportunities for SDM in the case of elective surgical episodes, unplanned or non-elective episodes may be less conducive to SDM. Although we did not propose a measure initially, we sought feedback on the opportunity for TEAM to capture quality data related to SDM between patients and providers, and avoidance of low value care and procedures. We invited public comment on whether such a measure concept or any existing measures would be appropriate for TEAM.
Lastly, we also recognize that there are certain measures on the 2023 Measures Under Consideration (MUC) List [ 900 901 ] that may be more clinically meaningful and specific to the episodes in TEAM. These measures are as follows:
- Hospital Harm—Falls with Injury (MUC2023-048).
- Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (MUC2023-049).
- Hospital Harm—Postoperative Respiratory Failure (MUC2023-050).
These three outcome measures focus on improving quality and health outcomes across a beneficiary's care journey and allow for hospitals to better align and coordinate care across various programs and care settings. TEAM sought further comment on these three MUC measures, and potentially replacing the CMS PSI 90 measure beginning in 2027, TEAM's second performance year. This timeline will allow TEAM participants to have one year to gain experience with reporting the measures in the Hospital IQR program before their performance is tied to payment beginning in TEAM's second performance year. Further details on these MUC measures can be found in section X.A.3.c.(3)(d) of the preamble of this final rule. The following is a summary of the public comments received on the proposed TEAM quality measures and its impact on other various quality requirements and our responses to these comments:
Comment: A commenter recommended that TEAM incorporate the Preventive Care and Screening: Body Mass Index (BMI) Screening and Follow-Up Plan Measure and quality measures for dementia care to support alignment between TEAM and the GUIDE model. This commenter encourages CMS to also consider the absence of obesity care specific quality metrics in data sets such as HEDIS. Accelerating the development, testing, and emplacement of quality metrics focused on diagnosis, care plan development and implementation, and outcomes, as examples, would support improved use of evidence-based care. Lastly, this commenter believes that any CMS Innovation Center demonstration activity in dementia care should focus on testing ways to improve and support overall care delivery quality, appropriate and timely diagnosis and treatment initiation, follow-up care coordination, and health equity.
Response: We appreciate commenters suggestion to incorporate dementia and obesity related quality measures in TEAM. However, CMS declines to incorporate these measures due to the potential for increasing reporting burden on hospitals who may not be reporting on these said measures. CMS will continue to move forward with our proposed measures in TEAM and will add or remove any new quality measures in TEAM in a future year's rulemaking, where appropriate.
Comment: A couple of commenters were in full support of the TEAM proposed quality measures. Commenters are appreciative of the proposed quality measure set and its alignment with quality reporting programs that TEAM participants will already be supporting. Although a commenter believes that adjustments can be made to align the associated measurement timeframes more appropriately for post-operative episodes with industry standards, as it relates to complications and mortalities.
Response: We appreciate commenters feedback and support of the TEAM proposed quality measures. As of now, we plan to make no adjustments to the proposed quality measure specifications since they align with what is proposed and used in the Hospital IQR and HAC Reduction programs. CMS will continue to move forward with the proposed measures as is. Any future updates or changes will be incorporated into a future year's rulemaking process.
Comment: A couple of commenters suggested that TEAM incorporate the hospital consumer assessment of healthcare providers and systems (HCAHPS) survey data, or the Patient-Reported Outcomes Measurement Information System (PROMIS) Global-10 survey (PROMIS-10) in the quality ( print page 69736) accountability framework for TEAM participants. Commenters believe that the HCAHPS and PROMIS survey may be a valuable addition to include well-established patient reported data and metrics.
Response: We appreciate commenters suggestion to include the HCAHPS and PROMIS survey data in TEAM and will consider the suggestion for future rulemaking, where appropriate.
Comment: Many commenters expressed concern that the proposed measures do not directly measure performance under the model and stated that for measures to be meaningful, those selected must be focused on what the model is trying to accomplish and limited to the model's patient population. This will ensure commenters have meaningful opportunities to improve quality and are held accountable under the model for care that is relevant to their care improvement efforts. The proposed measures utilize hospital-wide metrics that collect data on the entire hospital, rather than being specific to the patient population participating in TEAM. Another commenter mentioned the challenge that proposed measures are a measure of inpatient performance, whereas the model includes both inpatient and outpatient episodes. For procedures that can be furnished in either the inpatient or outpatient setting, typically the patients who continue to receive care in the inpatient setting tend to be higher risk and with higher prevalence's of complications, which can negatively impact performance on measures. A similar challenge has occurred in the CJR model. A couple of commenters recommended that CMS adopt a more diverse and meaningful set of quality measures for use under this model. In selecting more focused measures, it is also critical that CMS better align the manner it evaluates quality and cost under TEAM, which has been a challenge for CMS on other value-based programs. Lastly, a commenter suggested that CMS align TEAM quality measures with MIPS Value Pathways.
Response: We would like to thank all commenters that closely reviewed and shared their suggestions for with the TEAM proposed quality measures. While we recognize that the Hybrid HWR measure and CMS PSI 90 are not specific to surgical episodes, we believe they are critical measures for assessing hospital quality and performance. The Hybrid HWR measure is well suited for an episode accountability model because it reports a single Comment performance score derived from the volume-weighted results of five different models. The measure also indicates the hospital-level standardized risk ratios (SRR) for each of these five specialty cohorts. The Hybrid HWR measure helps to capture an overall view of hospital-level performance while encompassing all the TEAM participants, has the potential to incentivize improved transitions in care, and can serve as an indicator of poor quality of care within a hospital overall. The CMS PSI 90 measure summarizes patient safety across multiple indicators, monitors performance over time, and facilitates comparative reporting and quality improvement at the hospital level. The CMS PSI 90 composite measure intends to reflect the safety climate of a hospital by providing a marker of patient safety during the delivery of care. The CMS Innovation Center is using this measure for TEAM because it may inform how patients select care options, providers allocate resources, and payers evaluate performance. Additionally, CMS PSI 90 is a subset of the AHRQ Patient Safety Indicators and is a more relevant measure for the Medicare population because it utilizes ICD-10 data. CMS will consider incorporating episode-specific measures and aligning with other quality-based payment programs where applicable in future rulemaking.
Comment: A commenter suggested that as CMS considers additional quality measures to include in TEAM, CMS should provide all PAC providers with patient-level feedback data for claims-based measures. The lack of patient-level data for claims-based measures and relative infrequency of claims-based measure reports hampers Inpatient Rehabilitation Facilities' (IRF) ability to adjust or fully optimize any potential modifications to patient care practices and procedures in order to improve quality measure performance, which is the express purpose of the IRF Quality Reporting Program (QRP). Transparency of data and information in Medicare has become a critically important ingredient within the multiple dashboards, frameworks, and portals that have been designed to enable consumers and patients to evaluate quality of care and make better informed decisions about where to receive their care. This transparency should include furnishing IRFs with patient-specific data and information for IRF QRP claims-based quality measures, as it would enable IRFs to take steps toward improving and refining our processes and quality of care initiatives.
Response: We appreciate commenters suggestion to provide additional patient-level feedback data for the proposed claims-based measures and will take this feedback into consideration in future rulemaking where appropriate.
Comment: Many commenters suggested that TEAM include more episode specific measures or align with programmatic measures that focus on team-based care of patients including patient goals, drive quality improvement cycles with clinical data, help guide patients seeking safe and good care, and reduce measurement burden since they are tied to optimal care delivery and improvement. The concept behind the programmatic measure is based on several decades of history implementing programs that demonstrably improve patient care provided by both the clinical team and the facility. Specifically, commenters suggested TEAM include the Advanced Care Plan measure for all episodes proposed in TEAM. Additionally, a few commenters suggested using existing Medicare risk-standardized quality measures tracking TJA readmissions and complications as specific measures for the LEJR episode category. Also, a commenter suggested CMS consider use of existing or new CAHPS surveys to enhance the proposed quality measures and provide a standardized method to assess patient experience. Lastly, a commenter suggested CMS collaborate with surgical providers AND post-acute providers to identify appropriate post-surgical functional outcomes measures, particularly those related to mobility, selfcare, and cognition that are aligned with PAC measures.
Response: We acknowledge commenters suggestions for CMS to consider more episode specific and programmatic measures within TEAM and CMS will consider these suggestions for future notice and comment rulemaking, where appropriate.
Comment: A couple of commenters shared that they are interested in future shared decision-making measures to be incorporated into TEAM in a future year. The inclusion of culturally congruent shared decision making as a quality measure in TEAM would promote health equity by incentivizing providers to ensure all patients and their caregivers, including patients of color and patients with limited English proficiency, receive comprehensive information and support in their health care decisions. Shared decision-making measures will help both policymakers and providers better define and monitor what “value” is to patients and families. Specifically, a commenter suggested that TEAM consider the SDM-Q-9, CollaboRATE and SDM Process 4 survey measures for the TEAM LEJR episode. Additionally, another commenter ( print page 69737) acknowledges that measures around shared decision-making prior to a surgical episode are not being included now due to challenges with “respect to the timing of patient/provider interaction and when an episode is initiated.
Response: We appreciate commenters suggestion of the inclusion of shared decision-making measures into TEAM. CMS will take these considerations into account and any future incorporation of shared decision-making within TEAM will be shared through future rulemaking.
After consideration of the public comments we received, we are finalizing our proposals for the proposed quality measures that are being used within the Hospital IQR and HAC Reduction programs without modification in our regulation at § 512.547. TEAM will consider SDM and share further updates in a future notice and comment rulemaking.
Hospital readmission, for any reason, is disruptive to patients and caregivers, costly to the healthcare system, and puts patients at additional risk of hospital-acquired infections and complications. Readmissions are also a major source of patient and family stress and may contribute substantially to loss of functional ability, particularly in older patients. Some readmissions are unavoidable and result from inevitable progression of disease or worsening of chronic conditions. However, readmissions may also result from poor quality of care or inadequate transitional care. Transitional care includes effective discharge planning, transfer of information at the time of discharge, patient assessment and education, and coordination of care and monitoring in the post-discharge period. Numerous studies have found an association between quality of inpatient or transitional care and early (typically 30-day) readmission rates for a wide range of conditions. [ 902 ] In 2013, CMS contracted with Yale New Haven Services Corporation, Center for Outcomes Research and Evaluation (CORE) to demonstrate whether clinical data derived from electronic health records (EHRs) could be used to reengineer and enhance the Hospital-Wide All-Cause Unplanned Readmission (HWR) measure. [ 903 ] Under the contract with CMS, Yale CORE identified a set of core clinical data elements (CCDE) that are feasibly extracted from hospital EHRs and are related to patients' clinical status at the start of an inpatient encounter.
In the proposed rule, we proposed including the Hybrid Hospital-Wide Readmission (HWR) Measure with Claims and Electronic Health Record Data (CMIT ID #356) measure in TEAM, for all episode categories. Previously, within the CJR rule, CMS proposed using the Hospital-Level 30-day, All-Cause Risk-Standardized Readmission Rate (RSRR) Following Elective Primary Total Hip Arthroplasty (THA) and/or Total Knee Arthroplasty (TKA) (NQF #1551) measure because we believed that this measure aligned with CMS priorities to improve the rate of LEJR complications and readmissions, while improving the overall patient experience. As a result of stakeholder feedback voicing concerns over the requirements already set in place by the Hospital Readmissions Reduction Program for this measure, the Hospital-level 30-day, all-cause RSRR following elective primary THA and/or TKA (NQF #1551) was not included in the CJR Model. Our rationale for including the Hybrid HWR measure within TEAM is because the increased use of EHRs by hospitals creates an opportunity to incorporate clinical data into outcome measures without the laborious process of extracting them from paper medical records. Although claims-based risk adjustment has been shown to be comparable to risk adjustment using clinical data when observing hospital-level performance, clinical providers continue to express preference for using patient-level clinical data. [ 904 905 ] Additionally, we believe this version of HWR provides an opportunity to align the measure with clinical decision support systems that many providers utilize to alert care teams about patients at increased risk of poor outcomes, such as readmission, in real time during the inpatient stay. Further, utilizing the same variables to calculate hospital performance that are used to support clinical decision, we believe, would be clinically sensible and cost effective, as it may reduce the burden of EHR data mapping and extraction required for quality reporting.
In addition, clinical data captured in electronic health records are recorded by clinicians who are interacting with the patient and who value the accuracy of the data to guide the care they provide. Therefore, many clinical data elements that are captured in real-time to support patient care are less susceptible to gaming, coding drift, and variations in billing practices compared with administrative data used for billing purposes. These reporting processes allow for more stable measurements over time. Finally, the measures that are included within HRRP do not capture some of the episodes that we proposed for TEAM. The Hybrid HWR measure is one of the only existing readmission measures that captures readmission data for patients following procedures such as spine surgery. By using the Hybrid HWR measure, we are inclusive of the specified episodes and encourage broader efforts to reduce unnecessary returns to the hospital at participating hospitals within TEAM.
For TEAM, we proposed to use the measure specifications detailed here: https://ecqi.healthit.gov/sites/default/files/ecqm/measures/CMS529v4.html and https://qualitynet.cms.gov/inpatient/measures/hybrid/methodology . If we were to remove the measure, we would use notice and comment rulemaking. This measure would be a pay-for-performance measure beginning in PY 1 and scored in accordance with our proposed methodology in section X.A.3.d.(5)(e) of the preamble of this final rule.
We sought public comment on our proposal to include the Hybrid Hospital-Wide All-Cause Readmission Measure with Claims and Electronic Health Record Data measure in TEAM at § 512.547(a)(1).
The following is a summary of the public comments received on the proposed Hybrid Hospital-Wide Readmission (HWR) measure and our responses to these comments.
Comment: Several commenters raised concern that the Hybrid Hospital-Wide Readmission (HWR) measure provides little meaningful insight into the quality of care for TEAM specific episodes. Many commenters stated that the Hybrid HWR measure cannot assess quality or improvement for patients treated under TEAM and providers should not be held accountable for ( print page 69738) hospital-wide measures, especially if TEAM episodes make-up a small proportion of the hospital's total population.
Response: We would like to thank all commenters for their close review and comments regarding with the Hybrid HWR measure. While we recognize that the Hybrid HWR measure is not specific to surgical episodes, we believe this is a critical measure for assessing hospital quality and performance. This measure is well suited for an episode accountability model because it reports a single Comment performance score derived from the volume-weighted results of five different models. These models are specialty cohorts based on groups of discharge condition categories or procedure categories: surgery/gynecology; general medicine; cardiorespiratory; cardiovascular; and neurology. The measure also indicates the hospital-level standardized risk ratios (SRR) for each of these five specialty cohorts. The Hybrid HWR measure helps to capture an overall view of hospital-level performance while encompassing all the TEAM participants, has the potential to incentivize improved transitions in care, and can serve as an indicator of poor quality of care within a hospital overall. Overall, CMS acknowledges participants preference for episode specific measures. We will consider incorporating episode-specific measures where applicable in future rulemaking.
Comment: Some commenters noted that the novelty of hybrid reporting for the Hybrid HWR measure has created challenges for hospitals. For example, hospitals have been unable to extract the required data from their EHRs for the first year of voluntary reporting in IQR or reported issues with the measure specifications.
Response: We appreciate commenters for voicing their concerns and experience with reporting the HWR measure. CMS is aware of the obstacles hospitals faced reporting this measure in IQR through the hybrid methodology and are working with the developer to address issues commenters have noted. CMS is aware of the EHR reporting requirement concerns and commenters feeling that they won't be ready in time to report this measure for TEAM. We anticipate that hospitals will be better prepared to report the Hybrid HWR measure by the first performance year, but TEAM participants will still have the option to report the claims-based version of the measure throughout the model if they prefer.
Comment: Some commenters voiced their support for the Hybrid HWR measure. Commenters noted that they appreciate CMS aligning with other CMS Hospital Quality Reporting programs, agreed that the inclusion of clinical data likely improves risk adjustment, and agreed that the measure aligns well with the use of clinical decision support systems.
Response: We thank commenters for expressing support for inclusion of the Hybrid HWR measure.
Comment: A commenter asked CMS if TEAM considered any outcome measures that are associated with positive long term health outcomes.
Response: We appreciate this commenters question. CMS considered a number of measures before deciding on its proposed quality measures for TEAM. CMS will has taken commenter feedback into account and any updates or additions to the proposed quality measures in TEAM would be in future rulemaking.
After consideration of the public comments we received, we are finalizing our proposals for the proposed Hybrid HWR measures that is being used in the Hospital IQR program without modification in our regulation at § 512.547.
The Agency for Healthcare Research and Quality (AHRQ) developed patient safety indicators for health providers to identify potential in hospital patient safety problems for targeted institution-level quality improvement efforts. These Patient Safety Indicators (PSIs) are comprised of 26 measures (including 18 provider-level indicators) that highlight safety-related adverse events occurring in hospitals following operations, procedures, and childbirth. AHRQ developed the PSIs after a comprehensive literature review, analysis of available ICD codes, review by clinical panels, implementation of risk adjustment, and empirical analyses. The CMS Patient Safety and Adverse Events Composite (CMS PSI 90) is used in the HAC Reduction Program to support CMS public reporting and pay-for-performance. The CMS PSI 90 measure is calibrated using the Medicare fee-for-service population and based on the AHRQ Patient Safety Indicators. The CMS PSI 90 measure summarizes patient safety across multiple indicators, monitors performance over time, and facilitates comparative reporting and quality improvement at the hospital level. The CMS PSI 90 composite measure intends to reflect the safety climate of a hospital by providing a marker of patient safety during the delivery of care. However, we are aware of the common stakeholder concerns surrounding the CMS PSI 90 measure, including the following: [ 906 ]
- PSI 90 may be associated with adverse prioritization for preventing some conditions over others. Not all conditions are equal with respect to prevention guidelines.
++ Sepsis prevention may include use of prophylactic antibiotics.
++ Fall prevention requires assessment of fall risk and appropriately applied remediation methods.
- Pressure injury prevention consists of a time-consuming, complex series of unrelated tasks for nurses, consisting of daily skin checks and risk assessments, repositioning every 3 to 4 hours, and managing moisture and incontinence among other tasks.
- Simple clinical decision points can expose patients to many risks reflected in PSI 90; however, PSI 90 weighting system may influence risk because HACs are weighted in PSI 90 based on volume and harm.
- The PSI 90 composite score could create incentives to prioritize low hanging fruit (for example, procedures and treatments that are directly remunerated) over pressure injury prevention.
We proposed including the CMS PSI 90 measure in TEAM, for all episode categories, because it includes a broad array of safety events, many of which are relevant to patients in the episodes, are familiar to hospitals and have no additional burden. CMS would use the CMS PSI 90 software to produce the CMS PSI 90 results. Since CMS is currently using the CMS PSI 90 measure in certain quality programs, including the Hospital-Acquired Condition Reduction Program, we do not anticipate additional administrative burden for TEAM participants.
For TEAM, we proposed to use the measure specifications detailed here: https://qualitynet.cms.gov/inpatient/measures/psi/resources . If we were to remove the measure, we would use notice and comment rulemaking. This measure would be a pay-for-performance measure beginning in PY 1 and scored in accordance with our proposed methodology in section X.A.3.d.(5)(e) of the preamble of this final rule.
We sought public comment on our proposal to include the CMS PSI 90 measure in TEAM at proposed § 512.547(a)(2) and sought comment on other hospital level safety measures ( print page 69739) appropriate for these episodes that are not already tied to payment in CMS programs. We also invited public comment on the ones that were on the 2023 MUC list and the possible approach to transition from CMS PSI 90 to the three measures beginning in TEAM's second performance year.
The following is a summary of the public comments received on the proposed CMS PSI 90 measure and our responses to these comments.
Comment: Many commenters did not agree with the inclusion of the CMS PSI 90 measure in TEAM and highlight several reasons. For example, a few commenters mention that PSI 90 is a weighted average of all patient safety indicators (PSIs), despite the varying PSIs do not pose equal risk to a patient. Essentially, the measure is driven by the frequency of different events without accounting for relative severity of harm. Additionally, many commenters state this measure is not episode specific and too broad of a measure to be meaningful enough for the surgical episodes that are being proposed in TEAM. A commenter mentioned while PSI data may assist hospitals in identifying specific cases to investigate for quality improvement purposes, it is not well suited to meaningfully assessing hospital performance on safety issues in comparison to other hospitals. A commenter raised some concerns about the disproportionate impact PSI 90 may have on safety net hospitals. The inclusion of the PSI 90 measure could disadvantage these hospitals, further exacerbating the challenging financial situation of these vital facilities. Commenters shared concerns with this measure being scored on performance in multiple payment programs. The CMS PSI-90 measure currently included in the Hospital-Acquired Condition Reduction Program. Therefore, hospitals are already held financially accountable for their performance on this measure. A commenter does not believe that providers should be held financially accountable for performance on the same measure based on two different methodologies in two separate programs. Doing so risks creating conflicting signals based on performance for the same measure. Many commenters did not agree with the proposed CMS PSI 90 measure and CMS should consider incorporating other CMS patient safety outcome measures in TEAM.
Response: We are aware of commenters concern over the CMS PSI 90 measure; however, the CMS PSI 90 measure includes a broad array of safety events, many of which are relevant to patients in the episodes, are familiar to hospitals and have no additional burden on hospitals. Additionally, TEAM incorporating the CMS PSI 90 measure for only PY 1 and then replacing this measure with our proposed list of 2023 Measures Under Consideration measures along with considering other safety measures that were provided by commenters. CMS will use future rulemaking to incorporate any newly proposed safety measures within TEAM.
Comment: A couple of commenters suggested that CMS revisit the Alternate Quality Measure set that was used in BPCI Advanced and consider applying those measures for applicable TEAM episodes in lieu of the PSI 90 measure. The proposed measures in TEAM utilize mainly hospital wide metrics. Some commenters believe participants will find it difficult to draw direct correlates between the PSI 90 and surgical care redesign. A commenter states that the Alternate Quality Measures are trackable in the EHR, clinically relevant, and process oriented.
Response: We appreciate commenters suggestions on incorporating measures from the Alternate Quality Measure set that was used in BPCI Advanced in lieu of CMS PSI 90. However, CMS has proposed the CMS PSI 90 measure in TEAM because we believe this measure summarizes patient safety across multiple indicators, monitors performance over time, and facilitates comparative reporting and quality improvement at the hospital level. The CMS PSI 90 composite measure intends to reflect the safety climate of a hospital by providing a marker of patient safety during the delivery of care. Additionally, CMS wanted to propose measures that are not administratively burdensome. CMS PSI 90 has been mandatorily reported within HAC Reduction Program for years and hospitals will be familiar with reporting this measure before the start of TEAM.
Comment: A few commenters are in support of the inclusion of the PSI 90 measure with TEAM. A commenter states that the selection of the PSI 90 and all-cause readmission measures aligns heavily with quality improvement efforts already underway in hospitals and reduces the need for duplicate measures. Another commenter states that PSI 90 is fundamental to patient safety measurement because it addresses patient safety across a broad range of events and indicators, monitors performance over time, facilitates comparative reporting and quality improvement, and is already embedded in other quality reporting and pay-for-performance programs, for example, the HAC (Hospital Acquired Condition) Reduction Program.
Response: We appreciate commenters support of the PSI 90 measure being collected in TEAM for PY 1.
After consideration of the public comments we received, we are finalizing our proposals for the proposed CMS PSI 90 measure that is being used within the HAC Reduction program without modification in our regulation at § 512.547.
As part of the CMS Innovation Center's Strategy Refresh, TEAM is working to align with the Center's Patient-Reported Outcome Measure Strategy. This strategy supports the CMS Innovation Center's Advancing Quality Initiative, which aims to support a more person-centered quality strategy in accountable care and specialty care models and demonstrations. The Patient-Reported Outcome Measure Strategy aims to increase the use of patient-reported measures in CMS Innovation Center models and demonstrations. PROs are reported by the patient and capture a person's perception of their own health through surveys and questionnaires. Broadly, patient-reported data includes PROs and ePROs, which is the electronic capture of this data; patient-reported outcome measures (PROMs), which reflect how the PRO data is reported (for example, a survey instrument); and patient-reported outcome-based performance measures (PRO-PMs), which are reliable and valid quality measures of aggregated PRO data reported through a PROM and potentially used for performance assessment.
The CJR model includes voluntary reporting of PRO data. In order to meet the requirements for successful submission of PRO data, hospitals must submit the Veterans RAND 12 Item Health Survey (VR-12) or Patient-Reported Outcomes Measurement Information System (PROMIS) Global-10 generic PRO survey; and the (HOOS Jr.)/(KOOS Jr.) or HOOS/KOOS subscales PRO survey for patients undergoing eligible elective primary THA/TKA procedures. CMS was able to use the CJR THA/TKA PRO data collection to develop the THA/TKA PRO-PM as a part of the Hospital IQR Program, included in the FY 2023 IPPS/LTCH PPS Final rule ( 87 FR 48780 ).
Elective THA/TKAs are most commonly performed for degenerative joint disease, or osteoarthritis, which is the most common joint disorder in the US, affecting more than 32.5 million, or ( print page 69740) 1 in every 7, US adults. [ 907 908 ] This condition is one of the leading causes of disability among non-institutionalized adults; roughly 80 percent of patients with osteoarthritis have some limitation in mobility. [ 909 910 ] Osteoarthritis also significantly burdens the health care system—in 2017, it was the second most expensive treated condition across all payers in US hospitals, and in 2018, it accounted for approximately 1,128,000 hospitalizations. [ 911 912 913 ] THAs and TKAs offer significant improvement in quality of life by decreasing pain and improving function in a majority of patients, without conferring a high risk of complications or death. [ 914 915 ] Over 1 million hip and knee replacements are performed annually in the US, 60 percent of which are paid for by Medicare. This number is expected to double by 2030 with an estimated annual cost of $50 billion to Medicare. [ 916 ]
In order to encourage greater use of patient-reported outcome data, we proposed to require submission of THA/TKA PRO-PM. However, we recognize that this PRO-PM is only applicable to the LEJR episode category and sought comment on other PROs or PROMs that would be applicable to other episode categories tested and could be incorporated in future performance years of TEAM. Please note, that the addition of the use of generic PROs may be applicable across numerous episodes versus PROs that are more episode specific to given procedures. Also, we recognize that hospitals will be newly adapting to the Hospital IQR Program requirement for the THA/TKA PRO-PM, but that infrastructure and process development should make the incorporation of future PRO-PMs less burdensome.
For TEAM, we proposed to use the measure specifications detailed here: https://qualitynet.cms.gov/files/631b6163642a6000163edbf0?filename=THA_TKA-PRO-PM_MeasMthdlgy.pdf , https://www.cms.gov/files/document/draft-technical-report.pdf . If we were to remove the measure, we would use notice and comment rulemaking. This measure would be a pay-for-performance measure beginning in PY 1 and scored in accordance with our proposed methodology in section X.A.3.d.(5)(e) Of the preamble of this final rule.
We sought public comment on our proposal to include the Hospital-Level, Risk-Standardized Patient-Reported Outcomes Following Elective Primary THA/TKA measure in TEAM at § 512.547(a)(3).
The following is a summary of the public comments received on the proposed THA/TKA PRO-PM and our responses to these comments.
Comment: Several commenters stated their support for inclusion of the THA/TKA PRO-PM in the LEJR episode of TEAM. A commenter noted that PRO measures (PROMs) are critical in determining whether additional follow-up is warranted and another acknowledged that PROMs can be the impetus for initiating conversations between patients and providers and improving shared decision making.
Response: We thank commenters for their support of the THA/TKA PRO-PM in the LEJR episode. Requiring the submission of the PRO-PM aligns with the CMS Innovation Center's Advancing Quality Initiative that aims to drive person-centered care by elevating the voice of the patients. We will consider adding other episode-specific PROMs in future rulemaking where appropriate.
Comment: A few commenters proposed that CMS include other LEJR related measures in place of the THA/TKA PRO-PM. A commenter noted that the hospital-level 30-day risk-standardized readmissions quality measure following total hip and knee replacement (NQF #1551) and the hospital-level risk-standardized complication rate following primary total hip arthroplasty (THA) and/or total knee arthroplasty (TKA) (NQF #1550) measure would be more suitable to track short-term care quality following total joint arthroplasty. Another commenter suggested using the complications measure that has been in the IQR program for several years: COMP-HIP-KNEE Hospital-Level Risk-Standardized Complication Rate Following Primary Elective Total Hip Arthroplasty and/or Total Knee Arthroplasty.
Response: We appreciate commenters that suggested alternative measures for the LEJR episode in place of the THA/TKA PRO-PM. However, none of the quality measures suggested are PROMs or PRO-PMs. Patient reported outcomes can be extremely insightful for medical procedures and the THA/TKA PRO-PM is intended to quantify pain and functional improvements with validated PROMs to improve the lives of orthopedic patients.
Comment: Several commenters voiced their concern with required reporting of the THA/TKA PRO-PM, noting challenges related to data collection, administrative burden, and unfamiliarity with the measure. For example, a commenter raised that providers may not yet have the necessary experience submitting the THA/TKA PRO-PM to warrant its inclusion in a pay-for-performance program. Other commenters highlighted difficulties in collecting and reporting due to the degree of data collection burden and potential survey fatigue, especially since the measure involves fielding several pre-op and post-op surveys. A couple of commenters requested that the measure should only be included for voluntarily reporting.
Response: We thank commenters for expressing their concerns and challenges with reporting the THA/TKA PRO-PM. CMS recognizes the difficulties associated with patient reported outcome measures and the underlying data collection tools used in a clinical domain; however, we decline the requests to remove the THA/TKA PRO-PM from the model and requests to make the measure voluntary to report. THAs and TKAs are most commonly performed for degenerative joint disease, or osteoarthritis, and offer significant improvement in quality of life by decreasing pain and improving function in a majority of patients, without conferring a high risk of complications or death. Additionally, PRO-PMs hold providers accountable for the quality of care provided to its patients and support the CMS Innovation Center's goal of advancing person-centered measurement. Similar to the Hybrid HWR measure, we ( print page 69741) anticipate that hospitals will be better prepared to report this measure by the start of TEAM's first performance year.
Comment: A commenter is in support of TEAM's decision to incorporate future PROMs in later performance years of the model. Similar in nature to CJR voluntary submission of RAND 12 (VR-12) or PROMIS Global-10 surveys, a commenter urges CMS to push EHR vendors to support automated collection of these standard measure sets. The manual burden to operationalize collection of these measures within a bundled payment model can be extremely resource-intensive for hospitals that likely already have resource constraints when considering other requirements of TEAM.
Response: We appreciate commenters support for the decision to include general PROMs within TEAM in future performance years. CMS will consider EHR reporting challenges when selecting PROMs to account for future performance. CMS will update and add new measures to TEAM through future notice and comment rulemaking.
After consideration of the public comments we received, we are finalizing our proposals for the proposed THA/TKA PRO-PM that is being used within the Hospital IQR program without modification in our regulation at § 512.547.
We recognize there are other measures that may be more clinically relevant to the proposed TEAM clinical episode categories but are not yet being used in the Hospital IQR Program. Therefore, we sought comment on requiring submission of the Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (MUC2023-049) measure for use in all of our episode categories. This measure assesses the percentage of surgical inpatients who experienced a complication and then died within 30-days from the date of their first “operating room” procedure. Failure-to-rescue (FTR) is defined as the probability of death given a postoperative complication.
We believe inclusion of the potential FTR measure in TEAM would allow hospitals to identify opportunities to improve their quality of care. Hospitals and health care providers benefit from knowing not only their institution's mortality rate, but also their institution's ability to rescue patients after an adverse occurrence. Using a failure-to-rescue measure is especially important if hospital resources needed for preventing complications are different from those needed for rescue. From a research and policy perspective, knowing the failure-to-rescue rate in addition to the mortality rate would improve our understanding of mortality statistics. Since the death rate appears to be composed of two distinct rates, quality of care measurement may be improved if both mortality and FTR rates are reported instead of relying on the adjusted mortality rate alone. Failure to rescue measures have been repeatedly validated by their consistent association with nurse staffing, nursing skill mix, technological resources, rapid response systems, and other activities that improve early identification and prompt intervention when complications arise after surgery.
We also sought comment on requiring submission of two hospital harm measures for potential use in TEAM; the Hospital Harm—Falls with Injury (MUC2023-048) and the Hospital Harm—Postoperative Respiratory Failure (MUC2023-050).
We believe including the Hospital Harm—Falls with Injury (MUC2023-048) would address the importance of patient safety in the acute care setting. We recognize that inpatient falls are among the most common incidents reported in hospitals and can increase length of stay and patient costs. Due to the potential for serious harm associated with patient falls, “patient death or serious injury associated with a fall while being cared for in a health care setting” is considered a Serious Reportable Event by the National Quality Forum (NQF).
Falls (including unplanned or unintended descents to the floor) can result in patient injury ranging from minor abrasion or bruising to death as a result of injuries sustained from a fall. While major injuries (for example, fractures, closed head injuries, internal bleeding) (Mintz, 2022) have the biggest impact on patient outcomes, 2008-2021 data findings from the 2022 Network of Patient Safety Databases (NPSD) demonstrated that 18.8 percent of falls resulted in an injury, and of these falls where there was an injury, 41.8 percent resulted in moderate injuries such as skin tear, avulsion, hematoma, significant bruising, dislocations and lacerations requiring suturing. Moderate injury is, as defined by NDNQI, that resulted in suturing, application of steric-strips or skin glue, splinting, or muscle/joint strain (Press Ganey, 2020). NPSD findings of residual harm also demonstrated that mild to moderate level of harm represent 24.2. percent, 0.4 percent—severe harm, and 0.1 percent—death (levels of harm definitions developed by WHO, 2009).
By focusing on falls with major and moderate injuries, the goal of this hospital harm eCQM is to raise awareness of fall rates and, ultimately, to improve patient safety by preventing falls with injury in all hospital patients. The purpose of measuring the rate of falls with major and moderate injury events is to improve hospitals' practices for monitoring patients at high risk for falls with injury and, in so doing, to reduce the frequency of patient falls with injury. [ 917 918 919 920 ]
Additionally, we considered including the Hospital Harm—Postoperative Respiratory Failure (MUC2023-050). This eCQM assesses the proportion of elective inpatient hospitalizations for patients aged 18 years and older without an obstetrical condition who have a procedure resulting in postoperative respiratory failure (PRF). PRF is defined as unplanned endotracheal reintubation, prolonged inability to wean from mechanical ventilation, or inadequate oxygenation and/or ventilation, and is the most common serious postoperative pulmonary complication, with an incidence of up to 7.5 percent (the incidence of any postoperative pulmonary complication ranges from 10-40 percent). [ 921 ] This measure addresses the prevalence of PRF and the incidence variance between hospitals. PRF is a serious complication that can increase the risk of morbidity and mortality, with in-hospital mortality resulting from PRF estimated at 25 percent to 40 percent. [ 922 ] Surgical ( print page 69742) procedures complicated by PRF have 3.74 times higher adjusted odds of death than those not complicated by respiratory failure, 1.47 times higher odds of 90-day readmission, and 1.86 times higher odds of an outpatient visit with one of 44 postoperative conditions (for example, bacterial infection, fluid and electrolyte disorder, abdominal hernia) within 90 days of hospital discharge. [ 923 ] PRF is additionally associated with prolonged mechanical ventilation and the need for rehabilitation or skilled nursing facility placement upon discharge. [ 924 ]
The incidence of PRF varies by hospital, with higher reported rates of PRF in nonteaching hospitals than teaching hospitals (Rahman, et al., 2013). Additionally, one study found that the odds of developing PRF increased by 6 percent for each level increase in hospital size from small to large. [ 925 ] This finding suggests that there remains room for improvement in hospitals reporting higher rates of PRF.
The most widely used current measures of PRF are based on either claims data (CMS Patient Safety Indicator PSI 11) or proprietary registry data (National Surgical Quality Improvement Program (NSQIP) of the American College of Surgeons). The proposed eCQM is closely modeled after the NSQIP measure of PRF, which has been widely adopted across American hospitals, and is intended to complement and eventually supplant CMS PSI 11. As mentioned of section X.A.3.c.(3)(b) of the preamble of this final rule, these three MUC measures would potentially take the place of the CMS PSI 90 measure beginning in TEAM's second performance year. These three MUC measures will be available for optional reporting in the Hospital IQR Program beginning in 2026.
The following is a summary of the public comments received on the proposed MUC Measures and our responses to these comments on these measures that will be incorporated into TEAM in performance year 2:
Comment: Some commenters have shared concerns that hospitals are newly reporting on these proposed MUC measures and suggested they not be included in TEAM until hospitals have had ample time to familiarize themselves with the reporting requirements. A couple of commenters mention the new Hospital IQR measures that CMS is proposing as alternatives to Hospital IQR are untested, meaning there may be reporting challenges and benchmark data is not available to support quality improvement efforts. CMS acknowledges this uncertainty by proposing the new IQR measures be voluntary. A commenter suggested that for the measures referenced as being under consideration they would encourage CMS to allow the measures to be more widely adopted without financial recourse, highlighting previous implementation issues for eCQMs, before being implemented in TEAM, and suggest fuller implementation for troubleshooting. Lastly, a commenter mentions the proposed measures measure inpatient performance, whereas the model includes both inpatient and outpatient episodes. For procedures that can be furnished in either the inpatient or outpatient setting, typically the patients who continue to receive care in the inpatient setting tend to be higher risk and with higher prevalences of complications, which can negatively impact performance on measures.
Response: We would like to thank commenters for sharing their concerns with the newly proposed MUC measures that are being proposed for TEAM's second performance year. CMS is aware the measures are new to the Hospital IQR program. CMS is planning to align with the proposed reporting period of the Hospital IQR program that is referenced in section IX.C of the proposed rule. Details on the specific reporting periods for these MUC measures can be found within the proposed rule ( 89 FR 35938 ). CMS will continue to develop and make measure adjustments to better align with feedback that has been shared through various testing periods. CMS acknowledges concerns that these measures focused on inpatient performance for episodes that include both inpatient and outpatient procedures and may consider other quality measure options in future years. Any additional measures incorporated into TEAM will be shared in future rulemaking.
Comment: A commenter suggested that under the current proposal, two of the outcomes' measures only measure negative events—all-cause hospital readmissions and a composite adverse events measure, while three other negative events measures are being considered for future years. Only one positive outcomes measure related to a persons' functional abilities and the effectiveness of functional recover interventions (the THA/TKA PRO-PM) is proposed—and only for one of the proposed post-surgical bundles. This commenter believes the measures are unbalanced as the incentive program is focusing most heavily on cost savings and the prevention of medical complications with little or no focus on the primary purpose of the post-acute provider care—to help assure the beneficiary can live safely in their home environment with the optimal function.
Response: We acknowledge this commenter sharing their concerns with the proposed outcome measures. However, we believe our proposed measures focus on care coordination, patient safety, and patient reported outcomes which we believe represents areas of quality that are particularly important to patients undergoing acute procedures. CMS wishes to highlight the important protections `negative' measures allow for monitoring for decrements in care. CMS is finalizing the proposed quality measures for PY 1 in TEAM. CMS acknowledges the importance of the goals of surgical episodes to restore function however disagrees that a focus on reducing readmissions and adverse events is imbalanced, given our central mission to ensure high-quality, person-centered and safe care. Wherever possible, we would align TEAM quality measures with those used in ongoing models and programs to minimize participant burden. CMS will incorporate any changes or updates to our quality measures in a future notice and comment rulemaking where applicable.
Comment: A few commenters showed strong support for the Hospital Harm—Falls with Injury (CMIT ID #1518) measure being included in TEAM. Although some of these commenters mention some concern with how these data will be captured. Commenters agree monitoring to prevent falls is important for hospitals to measure but notes challenges to capturing this accurately with an eCQM. Lastly, a commenter mentions they are concerned with the measure since it lacks specificity and may lead to unequal assignment. This commenter stated that PSI 08—In Hospital Fall with Hip Fracture Rate is clear on which patients qualify, CMIT ID #1518 lacks specificity as to what constitutes a “moderate or major” injury without a clear and distinct definition of everything that is included within this broad category.
Response: We would like to thank the commenters for sharing their support ( print page 69743) and concerns regarding the Hospital Harm—Falls with Injury (CMIT ID #1518) measure being included in TEAM starting in its second performance year. By focusing on falls with major and moderate injuries, the goal of this hospital harm eCQM is to raise awareness of fall rates and, ultimately, to improve patient safety by preventing falls with injury in all hospital patients. The purpose of measuring the rate of falls with major and moderate injury events is to improve hospitals' practices for monitoring patients at high risk for falls with injury and, in so doing, to reduce the frequency of patient falls with injury. Should there be further specificity established in the definition of minor or major injury, CMS may update this measure in future years, and CMS will incorporate any changes or updates to our quality measures in a future notice and comment rulemaking where applicable.
Comment: Multiple commenters are in full support of the inclusion of the Hospital Harm—Postoperative Respiratory Failure (CMIT ID #1788) measure being included in TEAM.
Response: We would like to thank commenters for their support of this proposed MUC Measure being incorporated in TEAM within the model's second performance year.
Comment: Some commenters showed strong support for the Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) being included within TEAM. A commenter stated that hospital data on death could lead to significant learning about death and improved prevention of death if both mortality and failure to rescue data were captured and analyzed.
Response: We would like to thank commenters for their support of the proposed Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) measure being incorporated in TEAM within the model's second performance year.
Comment: A few commenters had some concerns with the proposed Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) measure being included within TEAM. Specifically, a commenter mentioned that the measure fails to account for circumstances outside of the control of the hospital reporting the measure. Additionally, a commenter noted that this measure should contain a 90-day post episode window to account for complications that occur in the CABG, THA/TKA and spinal fusion episodes.
Response: We appreciate commenters voicing their concerns about the Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) measure. We believe inclusion of the potential Failure-To-Rescue measure in TEAM would allow hospitals to identify opportunities to improve their quality of care. From a research and policy perspective, knowing the failure-to-rescue rate in addition to the mortality rate would improve our understanding of mortality statistics. CMS believes that the proposed 30-day window is appropriate to assesses the percentage of surgical inpatients who experienced a complication and then died. CMS will continue to monitor the specifications of this measure and incorporate any changes to this measure in future rulemaking.
After consideration of the public comments we received, we are finalizing the proposed Hospital Harm and Failure-to-Rescue measures to be used within the Hospital IQR program without modification in our regulation at § 512.547. These measures will be incorporated into TEAM during its second performance year.
We believe it is important to be transparent and to outline the form, manner, and timing of quality measure data submission so that accurate measure results are provided to hospitals, and that timely and accurate calculation of measure results are consistently produced to determine reconciliation payment amounts and repayment amounts. We proposed that data submission for the Hybrid Hospital-Wide Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356), CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135), Hospital-Level, and Risk-Standardized Patient-Reported Outcomes Following Elective Primary Total Hip and/or Total Knee Arthroplasty (THA/TKA) (CMIT ID #1618) be accomplished through existing Hospital IQR Program processes. Since these measures are or will soon be reported to the Hospital IQR and HAC Reduction Programs, hospitals would not need to submit additional data for TEAM.
For the Measures Under Consideration (MUC) measures, Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134), Hospital Harm—Postoperative Respiratory Failure (CMIT ID #1788) and Hospital Harm—Falls with Injury (CMIT ID #1518) measures, we would propose that data submission for these measures align with the Hospital IQR Program if they are finalized for that program as proposed. Similar to the proposed required measures noted previously, hospitals would not need to submit any additional data on these proposed measures if they are finalized and implemented for the Hospital IQR Program. We invited public comment on the proposal to collect quality measure data through the existing mechanisms of the Hospital IQR and HAC Reduction Program.
The following is a summary of the public comments received on the proposed performance periods and our responses to these comments.
Comment: A commenter mentioned concern over the proposed performance periods for the TEAM quality measures being significantly lagged. CMS should delay incorporating quality measures until the performance period for those measures is actionable (that is, begins after the model start date). Commenters recognize that any adjustments could be lagged by several years due to the data validation process, but this is certainly more appropriate than holding hospitals accountable for measure performance in a period before the model starts. Furthermore, commenters cannot evaluate hospital-level performance on these measures to meaningfully comment on because data is not yet publicly available for two of the three measures. Therefore, it is premature to propose these measures for inclusion in TEAM.
Response: We appreciate commenters for voicing their concerns over the proposed performance periods for the TEAM quality measures. However, to align with the model timeline, CMS will move forward with the proposed performance periods. We understand the concern that the first performance period is prior to the model start date, but hospitals will already have mandatorily reported data on these measures within the Hospital IQR and HAC Reduction Programs. Any adjustments to the TEAM proposed measures will be shared in a future notice and within comment rulemaking.
After consideration of the public comments we received, we are finalizing our proposals for the proposed measures performance periods that align with those that are being used within the Hospital IQR and HAC Reduction programs without ( print page 69744) modification in our regulation at § 512.547.
We believe that the display of measure results is an important way to educate the public on hospital performance and increase the transparency of the model. We proposed to display quality measure results on the publicly available CMS website in a form and manner consistent with other publicly reported measures. CMS would share each TEAM participants' quality metrics with the hospital prior to display on the CMS website. The timeframe for when TEAM participants would receive data on our proposed measures align with the Care Compare schedule that can be found here: https://data.cms.gov/provider-data/topics/hospitals/measures-and-current-data-collection-periods . The Hybrid HWR and CMS PSI 90 measure results are posted annually in July. The THA/TKA PRO-PM is still in the voluntary reporting stage and the public reporting schedule for this measure will be reported on an annual basis. All measures under the statutory hospital quality programs have a 30-day preview period prior to results being posted on the Care Compare web page. TEAM participant measure scores will be delivered to TEAM participants confidentially. We proposed to publicly report PY 1 measure scores in 2027 and we would continue to publicly report scores every performance year with a one-year lag. TEAM has proposed 2027 as the first performance year for when scores will be publicly available due to the amount of lag time it takes for a few of our measures to fully process. For example, the Hybrid HWR measure which uses claims data and core clinical data elements from the EHR has about a year between from when the data is submitted and when that data is publicly posted. The applicable time periods for the measures during TEAM are summarized in the Table X.A.-09.

The proposed time periods for the Hybrid Hospital-Wide Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356), CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135) and Hospital-Level, Risk-Standardized Patient-Reported Outcomes Following Elective Primary Total Hip and/or Total Knee Arthroplasty (THA/TKA) (CMIT ID #1618) are consistent with the Hospital IQR Program performance periods for the Hybrid HWR measure and THA/TKA PRO-PM and consistent with the HAC Reduction Program performance period for the CMS PSI 90 measure. We believe the public is familiar with the proposed measures, which have mostly been publicly reported in past releases of Care Compare as part of the Hospital IQR and HAC Reduction Programs. We are aware that the Hospital-Level, Risk-Standardized Patient-Reported Outcomes Following Elective Primary Total Hip and/or Total Knee Arthroplasty (THA/TKA) PRO-PM is new to the Hospital IQR Program, although it has been used in the CJR model for several years and sought comment on the use of this measure for TEAM. To minimize confusion and facilitate access to the data on the measures included in TEAM, we proposed to post the data on each TEAM participant's performance on each of the three proposed quality measures in a downloadable format in a section of the website specific to TEAM, similar to what is done for the Hospital Readmissions Reduction Program and the HAC Reduction Program. We invited public comments on these proposals to post data for the required measures on the TEAM specific website.
The following is a summary of the public comments received on the proposed display of public quality measurement data and our responses to these comments.
Comment: A couple of commenters were in support of the public display of TEAM quality measure data.
Response: We appreciate commenters support and will provide quality measure data publicly as appropriate.
Comment: A couple of commenters are against the proposal to publicly report quality measure data within TEAM. Specifically, a commenter mentions that they are not supportive of publicly reporting TEAM quality scores because not all hospitals in a market/region will have TEAM scores which may unfairly advantage/disadvantage participating hospitals when patients seek services in a region and not all hospitals have reporting data. Having only some hospitals in a region participate creates inequality in patients' ability to evaluate the competing hospitals because there is this additional data provided only for some of the hospitals. Although commenters favor publicly reporting of data when all hospitals have an equal opportunity to have their data risk stratified and presented in the same manner. Additionally, a commenter mentioned concern that publicly reporting PRO-PMs for hospitals and surgeons may disadvantage those physicians and hospitals who accept higher risk and socially disadvantaged patients. The difficulty of obtaining PRO-PM metrics in underserved and socially disadvantaged populations is ( print page 69745) further exacerbated by a greater incidence of medical and social risk factors for readmissions, ER visits and adverse outcomes in certain vulnerable populations and geographic regions.
Response: We thank commenters for raising their concerns with publicly reporting TEAM quality scores and we recognize the challenges associated with collecting PRO-PM data. However, we believe that the public display of measure results is an important vehicle for communicating hospital performance to the public and increasing transparency of TEAM. Publicly reported data helps patients make informed decisions about where to seek care that is not just based on cost. We will move forward with the proposal to publicly display TEAM measure results which is consistent with other CMS Innovation Center models and CMS quality reporting programs.
After consideration of the public comments we received, we are finalizing our proposals for the proposed public display of quality measurement data that aligns with how other pay-for-reporting programs display their quality measurement data.
In the proposed rule, we stated that in determining the best methodology for setting target prices for episodes, we drew from lessons learned from multiple iterations of both the CJR and BPCI Advanced target price methodologies. As we developed the methodologies for CJR and BPCI Advanced and refined them over time in response to observed changes in nationwide spending trends and payment system changes (such as the removal of total knee arthroplasty (TKA) and total hip arthroplasty (THA) from the inpatient only (IPO) list, and the reclassification of certain MS-DRGs), each new iteration drew from lessons learned in the previous iteration. For purposes of TEAM, we aimed to find the balance between simplicity and predictive accuracy. CMS wants to adopt a payment methodology that will be as transparent and understandable as possible for participants of varying levels of statistical background and knowledge. On the other hand, the more elements we consider and more sophisticated statistical modeling we use, the better able we are to accurately predict performance period spending. Accurate performance period spending predictions increase the likelihood of achieving our model goals of setting target prices that provide a reasonable opportunity to achieve savings for Medicare but are not too onerous for participants.
When designing the CJR payment methodology, one goal was to be as simple and straightforward as possible, given that it was a mandatory model covering only one episode category. The initial CJR payment methodology included a 3-year baseline period that rolled forward every 2 years. Target prices used a blend of participant-specific and regional spending, which shifted towards 100 percent regional spending for PYs 4-5. Downside risk was waived for the first performance year of the model to allow participants time to enact practice changes that would help them succeed in the model. Beginning in PY 2, participants were subject to both upside and downside risk, within stop-loss and stop-gain limits that increased to a maximum of 20 percent by PY 3 for most hospitals. The stop-loss and stop-gain limits were designed to ensure that participants would neither be subject to an unmanageable level of risk, nor be incentivized to stint on care to achieve savings. The initial CJR payment methodology is described in detail in the final rule titled “Medicare Program; Comprehensive Care for Joint Replacement Payment Model for Acute Care Hospitals Furnishing Lower Extremity Joint Replacement Service” that appeared in the November 24, 2015, Federal Register ( 80 FR 73274 ) (referred to in this proposed rule as the “2015 CJR Final Rule”), starting at 80 FR 73324 .
The initial CJR payment methodology was modified in the final rule titled “Medicare Program: Comprehensive Care for Joint Replacement Model Three-Year Extension and Changes to Episode Definition and Pricing; Medicare and Medicaid Programs; Policies and Regulatory Revisions in Response to the COVID-19 Public Health Emergency” that appeared in the May 3, 2021 Federal Register ( 86 FR 23496 ) (referred to in this proposed rule as the “2021 CJR 3-Year Extension Final Rule”). The CJR model's 3-year extension and modification were due to a number of factors, as described in detail starting at 86 FR 23508 . A principal reason for the modifications to the payment methodology was the fact that the initial CJR target price methodology did not account for changing downward trends in spending on lower extremity joint replacement (LEJR) episodes, both among CJR participant hospitals and non-participant hospitals. The resulting reconciliation payments under the initial methodology rewarded participants for spending reductions that likely would have happened regardless of the model, which led to concerns that target prices could be too high for Medicare to achieve savings in the model over time.
The changes to the model increased the complexity in some ways (for example, the addition of risk adjustment multipliers) while simplifying it in other ways (for example, the removal of update factors) in order to calculate target prices that would more accurately reflect performance period spending. A retrospective Market Trend Factor was applied to target prices at reconciliation to capture changes in spending patterns that occurred nationally during the performance period. This market trend factor, in combination with the change from a 3-year baseline to a 1-year baseline, negated the need for setting-specific update factors that we had used previously to set purely prospective target prices. At the same time, our added risk adjustment increased target prices for episodes with more complex patients, to better reflect the higher costs associated with those patients. The changes to the CJR payment methodology are described in detail in the 2021 CJR 3-Year Extension Final Rule starting at 86 FR 23508 .
By contrast, the BPCI Advanced methodology is more complex. The target price calculation method was designed to support participation from a broad range of providers by accounting for variation in episode payments and factors that contribute to differences that are beyond providers' control. In Model Years 1-3, BPCI Advanced target prices were constructed using a 4-year rolling baseline period and were based on hospital historical payments, patient risk adjustment, a prospective peer group trend factor, and 3 percent CMS discount. Physician group practice (PGP) target prices adjusted hospital target prices for PGP-specific patient case mix and differences between PGP and hospital historical payments. Risk adjustment is performed using a two-stage model, with Stage 1 consisting of a compound log-normal model with episode cost as the dependent variable, and Stage 2 consisting of an Ordinary Least Squares regression with case mix adjusted spending as the dependent variable.
The use of a prospective trend in Model Years 1-3 resulted in prices not accurately predicting spending that ( print page 69746) arose from unanticipated, systematic factors. For example, changes in coding guidelines can lead to cost changes. In fiscal year 2017, there were changes to the guidelines for coding the congestive heart failure (CHF) and simple pneumonia episodes, two of the highest-volume episodes in the BPCI Advanced model. The change resulted in an increase in the share of patients classified as having more serious CHF and simple pneumonia diagnoses in the performance period than in the baseline period. Because target prices are based on the seriousness of a patient's diagnosis, target prices increased leading to larger reconciliation payments to participants and losses to Medicare.
The losses to Medicare spurred changes to the BPCI Advanced pricing methodology. Similar to CJR, the prospective trend factor used in Model Years 1-3 was replaced in Model Year 4 with a retrospective trend factor adjustment at reconciliation, although this retrospective trend adjustment was subject to guardrails. Specifically, the trend at reconciliation could not exceed ±10 percent of the prospective trend for Model Years 4 and 5, and in response to participant feedback, the trend adjustment was limited to ±5 percent beginning in Model Year 6. The CMS discount was also reduced in Model Year 6 from 3 percent to 2 percent for medical episodes. Pricing methodology changes since Model Year 4 were intended to improve pricing accuracy and reflect actual spending trends during the performance period. Future evaluation reports will assess the effectiveness of these changes. Additional information on the BPCI Advanced pricing methodology may be found on the BPCI Advanced participant resources page. [ 926 ]
In TEAM, our goal is a target price methodology that blends the most successful elements of each of these model iterations, striking a balance of predictability and accuracy.
While we describe each element of the pricing and payment methodology in detail in the following sections, here we present an overview of the proposed TEAM pricing and payment methodology. At proposed § 512.540, we proposed to use 3 years of baseline data, trended forward to the performance year, to calculate target prices at the level of MS-DRG/HCPCS episode type and region. In the proposed rule, we proposed to group episodes from the baseline period by applicable MS-DRG for episode types that include only inpatient hospitalizations, and by applicable MS-DRG or HCPCS code for episode types that include both inpatient hospitalizations and outpatient procedures. For episode types that include both inpatient hospitalizations (identified by MS-DRGs) and outpatient procedures (identified by HCPCS codes), HCPCS codes are combined for purposes of target pricing with the applicable MS-DRG representing an inpatient hospitalization without Major Complications and Comorbidities, as we expected those beneficiaries to have similar clinical characteristics and costs. After capping high-cost outlier episodes at the 99th percentile for each of the 24 proposed MS-DRG/HCPCS episode types and 9 regions (which we proposed at proposed § 512.505 to define as the 9 U.S. census divisions, as defined by the U.S. Census Bureau), we stated in the proposed rule we would use average standardized spending for each MS-DRG/HCPCS episode type in each region as the benchmark price for that MS-DRG/HCPCS episode type for that specific region, resulting in 216 MS-DRG/HCPCS episode type/region-level benchmark prices. In the proposed rule, we proposed to apply a prospective trend factor and a discount factor to benchmark prices (as well as a prospective normalization factor, described later in this section) to calculate preliminary target prices. The prospective trend factor would represent expected changes in overall spending patterns between the most recent calendar year of the baseline period and the performance year, based on observed changes in overall spending patterns between the earliest calendar year of the baseline period and the most recent year of the baseline period. The discount factor would represent Medicare's portion of potential savings from the episode.
At proposed § 512.545, we proposed to risk adjust episode-level target prices at reconciliation by the following beneficiary-level variables: age group, Hierarchical Condition Category (HCC) count (a measure of clinical complexity), and social risk (the components of which are described in more detail in sections X.A.3.d.(4) and X.A.3.f of the preamble of this final rule). In the proposed rule, we proposed to calculate risk adjustment multipliers prospectively at the MS-DRG/HCPCS episode type level based on baseline data and hold those multipliers fixed for the performance year. To ensure that risk adjustment does not inflate target prices overall, we further proposed to calculate a prospective normalization factor based on the data used to calculate the risk adjustment multipliers. We proposed to apply the prospective normalization factor, in addition to the prospective trend factor and discount factor described previously, to the benchmark price to calculate the preliminary target price for each MS-DRG/HCPCS episode type and region. We proposed that the prospective normalization factor would be subject to a limited adjustment at reconciliation based on TEAM participants' observed performance period case mix, such that the final normalization factor would not exceed ±5 percent of the prospective normalization factor.
We sought comments on the general design of TEAM pricing and payment methodology.
In response to comments, we are finalizing many elements of the proposed pricing and payment methodology from the proposed rule, as we describe below. However, in certain cases we are finalizing policies that have been modified in order to be responsive to commenters' concerns. Specifically, in § 512.540(c) we are finalizing a 1.5 percent discount for Coronary Artery Bypass Graft Surgery (CABG) and Major Bowel Procedure episodes, and a 2 percent discount for Lower Extremity Joint Replacement (LEJR), (Surgical Hip/Femur Fracture Treatment (SHFFT), and Spinal Fusion episodes.
In § 512.545(f), we are finalizing the application of a 3 percent capped retrospective trend factor at reconciliation. In § 512.545(a), we are finalizing a policy to include additional beneficiary and hospital level risk factors in our risk adjustment model. Although we are finalizing at § 512.545(a)(1) our proposal to use a lookback period to determine which HCC flags the beneficiary is assigned, we are not yet finalizing the length of the lookback period due to concerns raised by commenters. Similarly, after consideration of the public comments we received, we will not be finalizing a policy on low volume hospitals. Accordingly, we are modifying regulatory text in sections § 512.545 (a)(1) to remove references to a 90-day lookback period, and § 512.550 (e)(3) to remove references to a low volume threshold. We intend to propose and finalize a specific length for the lookback period and an alternative low volume hospital policy through notice and comment rulemaking within the ( print page 69747) next year, so that participants will be aware of the final policies prior to the start of the model.
The following is a summary of comments we received regarding the general design of TEAM pricing and payment methodology and our responses to these comments:
Comment: A few commenters expressed concern that the target price methodology is opaque and, when combined with the final target price data lag, will make it onerous for TEAM participants to succeed in the model.
Response: We thank the commenters for their concern regarding the uncertainty of our target price methodology. In order to clearly describe the target price methodology to stakeholders, we have detailed each component used in target price construction throughout the proposed rule (beginning at 89 FR 36426 ) and this final rule preamble. Alongside those details, we also provided comparisons to the BPCI Advanced and CJR models to help readers further understand differences or similarities given these models and methodologies have been around longer and are more familiar to the public. We are also taking multiple steps to assist participants with understanding both the TEAM pricing methodology, and how they can use the data we will provide to help them succeed in the model. We will be sharing preliminary target prices and baseline data, pursuant to a request and TEAM data sharing agreement, as discussed in section X.A.3.k of the preamble of this final rule, which will allow TEAM participants to understand their preliminary target price in advance of the performance year as well as increase transparency on historical performance. We acknowledge that monthly data will have a lag, but we believe that by choosing surgical episode categories, it minimizes the difficulty in determining whether a beneficiary is included in the model. Lastly, TEAM participants will have approximately 17 months to prepare before the model start date, as discussed in section X.A.3.a. of the preamble of this final rule. During this time, we anticipate engaging with TEAM participants and providing learning resources and opportunities to help them further understand TEAM policies, including the construction of target prices. We believe that each of these steps will help to minimize any unnecessary burden of TEAM participation and optimize participants' opportunities for success in the model.
Comment: A couple of commenters were concerned that risk-adjustment was inadequate to ensure accurate target prices. Some procedures, such as hip fractures, are performed as an emergency. Others may be technically elective, but complex.
Response: We thank the commenters for their concern regarding target price accuracy. We have made some modifications to our risk adjustment methodology based on commenters feedback, as discussed in section X.A.3.d.(4) of the preamble of this final rule. We will continue to analyze our risk-adjustment model and will consider changes for future notice and comment rulemaking.
Comment: A commenter expressed concern that bundled payments work best for relatively healthy patients and safety net hospitals may be penalized by TEAM without adequate risk adjustment. They stated their belief that improving target price accuracy for TEAM should be a goal of CMS.
Response: We thank the commenter for their concern regarding safety net hospital participation in TEAM and accurate target prices. We agree that accurate target prices are important for participant success in TEAM, and we will risk adjust for dual eligibility, age, HCC count, and more, as discussed in section X.A.3.d.(4) of the preamble of this final rule. Furthermore, TEAM will include a social risk adjustment factor that should account for hospitals that provide care for underserved beneficiaries such as safety net hospitals. We disagree TEAM will penalize safety net hospitals as we have purposely included provisions to minimize financial risk. Specifically, safety net hospitals, as defined in section X.A.3.f.(2) of the preamble of this final rule, have the option to participate in Track 1 for the first three performance years with no downside risk, as discussed in section X.A.3.a.(3) of the preamble of this final rule. However, we will monitor TEAM participant performance, including safety net hospitals and we will consider further changes in future notice and comment rulemaking.
Comment: A commenter expressed concern that cost reductions in TEAM must come from post-discharge spending relative to the benchmark in a 30-day period.
Response: We thank the commenter for their concern regarding cost reductions via post-discharge spending. We acknowledge that much of the cost reductions achieved in past models such as the CJR and BPCI Advanced models were achieved via reductions in post-acute care while avoiding a reduction of quality of care. We believe TEAM can achieve a similar result with a 30-day episode window rather than a 90-day episode window.
Comment: A commenter is concerned that site neutral target prices for TEAM episodes could incentivize patients to go to lower acuity settings regardless of appropriate care.
Response: We thank the commenter for their concern regarding quality of care and site neutral payments. We disagree that site neutral payments will lead to a decrease in quality of care. TEAM episodes will be risk-adjusted and reconciliation payments made in the aggregate. Furthermore, a CQS adjustment will adjust a TEAM participant's reconciliation amount based on patient quality scores. This will incentivize practices to focus on cost reductions and care redesign that will both reduce costs and maintain or improve quality of care.
Comment: A commenter stated that CMS should monitor target prices to ensure participants are adequately compensated.
Response: We thank the commenter for their concern regarding target prices that ensure that participants are adequately compensated. TEAM's target prices will be risk-adjusted for many factors such as regional pricing over three baseline years, dual eligibility, age, HCC counts, a social risk adjustment, and a discount factor. We will monitor target prices and participant performance throughout TEAM.
Comment: A couple commenters recommended that CMS make considerations for the utilization of critical access hospital (CAH) swing beds. A commenter noted that CAH swing beds are sometimes the only option with access, sometimes they are clinically the best option based on the beneficiary's hometown or primary care provider (PCP), and under freedom of choice, they are often preferred by beneficiaries. Commenters cited that CAH swing bed daily allowed claims can be ten-folder greater than traditional skilled nursing facility (SNF) daily allowed claims. They stated their belief that if CAH swing bed claims are not adjusted for in some way, either in Target Prices or Reconciliation calculations, participants would not be able to meet Target Prices.
Response: We thank the commenters for sharing their concerns regarding CAH swing bed claims. In response to the comments, we analyzed how frequently CAH swing bed claims are included in 30-day episode spending and what proportion of spending in the post-discharge period CAH swing bed claims make up. The 30-day episodes were constructed using Medicare FFS ( print page 69748) claims from 2022 and the first two quarters of 2023.
Our analysis showed that CAH swing bed stays are a low frequency event in 30-day episodes. In 4 of the 5 episode categories proposed for TEAM, among episodes with at least one SNF claim (traditional or CAH swing bed), approximately 4 percent had at least one CAH swing bed claim. In the episode category CABG, approximately 7 percent of episodes had a CAH swing bed claim. Since CAHs swing beds are exempt from the SNF PPS, they are reimbursed at a higher rate. However, these claims will not make up a large proportion of post-discharge spending, or episode spending as a whole in TEAM episodes. Our analysis showed that in 4 of the 5 episode categories, post-discharge spending in the SNF setting among episodes with at least one CAH swing bed claim only accounted for 11 percent to 12 percent of total post-discharge SNF spending among all episodes. In CABG episodes, the proportion was 19 percent of total post-discharge SNF spending.
We are concerned that applying adjustments for CAH swing bed claims will create unintended consequences for utilization of these CAH swing bed services. We believe that the high reimbursement rates for these CAH swing bed claims could encourage providers to seek out relationships with and to increase utilization of traditional SNFs in order to obtain savings for the model. Although it is always good for hospitals to have relationships with both traditional SNFs and CAHs, model participants that have historically utilized CAH swing beds will be in a position to earn significant savings by establishing relationships with traditional SNFs and discharging patients they would otherwise move to CAH swing beds to traditional SNFs. Finally, we note that CAH swing bed allowable charges will be payment standardized as will other Part A and Part B allowable charges in TEAM. CMS has been working closely with the Federal Office of Rural Health Policy to adjust the payment standardization formula for CAH swing beds to reflect resource use that is comparable to that of SNFs. CMS plans to implement this adjusted payment standardization formula for CAH swing beds in the future.
At proposed § 512.540(b)(2) we proposed to use 3 years of baseline episode spending to calculate benchmark prices, which we would further adjust as described in section X.A.3.d.(3)(i) of the preamble of this final rule to create preliminary target prices. In the proposed rule, we proposed to roll this 3-year baseline period forward every year. Specifically, we proposed in the proposed rule that—
- To determine baseline episode spending for performance year (PY) 1, CMS would use baseline episode spending for episodes that started between January 1, 2022, and December 31, 2024;
- To determine baseline episode spending for PY 2, CMS would use baseline episode spending for episodes that started between January 1, 2023, and December 31, 2025;
- To determine baseline episode spending for PY 3, CMS would use baseline episode spending for episodes that started between January 1, 2024, and December 31, 2026;
- To determine baseline episode spending for PY 4, CMS would use baseline episode spending for episodes that started between January 1, 2025, and December 31, 2027;
- To determine baseline episode spending for PY 5, CMS would use baseline episode spending for episodes that started between January 1, 2026, and December 31, 2028.
The use of 3 years of baseline episode spending is consistent with our initial CJR methodology, as described in the 2015 CJR Final Rule at 80 FR 73340 . In that case, the 3-year baseline period moved forward every 2 years. However, in combination with the lack of a retrospective trend factor, the use of a 3-year baseline period that only moved forward every 2 years meant that our methodology was not able to capture the degree to which spending on lower extremity joint replacement (LEJR) episodes was decreasing nationwide, both among CJR and non-CJR hospitals. As a result, we believed our target prices partially reflected spending decreases that were not due specifically to participation in CJR.
Subsequently, in the 2021 CJR 3-Year Extension Final Rule, we finalized a policy to use a 1-year baseline period that would move forward every year (with the exception of skipping data from 2020 due to COVID-19 irregularities) ( 86 FR 23514 ). In combination with a retrospective market trend factor, using 1 year of baseline episode spending updated every year meant that our target prices would not be inflated as they had been under the initial CJR methodology. BPCI Advanced employs a strategy that blends elements of both CJR approaches, with a longer baseline period (4 years) similar to the initial CJR methodology, but shifting forward every year, as we do in the CJR extension.
Participants in episode-based payment models have expressed concerns about a concept known as the ratchet effect when choosing the baseline period from which to calculate target prices. That is, participants do not want to be penalized for achieving lower spending by having lower target prices in subsequent years. The use of fewer years of the most recent baseline episode spending, as well as more frequent rebasing, will generally decrease target prices more quickly year over year if overall episode spending is decreasing, as opposed to a longer, fixed baseline. However, we need to balance this concern against the likelihood of having inaccurate target prices if we use older baseline episode spending or rebase less frequently.
In the proposed rule, one way that we proposed to mitigate the ratchet effect is to use a 3-year baseline period and rebase annually. We believed this approach would achieve a balance between having target prices based on sufficiently up-to-date spending patterns but not requiring participants to compete against only the most recent spending patterns.
In the proposed rule, we proposed to adjust baseline episode spending to trend all episode spending to the most recent year of the baseline period. The adjustment would reflect the impact of inflation and any changes in episode spending due to evolving patterns of care, Medicare payment policies, payment system updates, and other factors during the baseline period. In the proposed rule, we proposed to define a baseline year as any of the three calendar years (CYs) during a given baseline period. For example, baseline year 1 for PY 1 will be CY 2022, baseline year 2 will be CY 2023, and baseline year 3 will be CY 2024. In the proposed rule, we proposed to calculate the adjustment factors for baseline years 1 and 2 by dividing average episode spending for baseline year 3 episodes by average episode spending for episodes from baseline years 1 and 2, respectively. We would then apply the applicable adjustment factors to the episode spending of each episode in baseline years 1 and 2. This adjustment would bring all baseline episode spending forward to the most recent baseline year, so that baseline year 1 and 2 spending would be expressed in baseline year 3 dollars. This method would be consistent with how we calculated the baseline trend factor for CJR in the performance years that used the 3-year baseline period, as described ( print page 69749) in the 2015 CJR Final Rule ( 80 FR 73342 ). In the proposed rule, we proposed to calculate these baseline trend factor adjustments at the MS-DRG/HCPCS episode type and region level.
In recognition of the fact that baseline episode spending from more recent years are likely to be a better predictor of performance year spending, we proposed in the proposed rule to weight recent baseline episode spending more heavily than episode spending from earlier baseline years. Specifically, we stated in the proposed rule we would weight episode spending from baseline year 1 at 17 percent, baseline year 2 at 33 percent, and baseline year 3 at 50 percent. This method of weighting would mean that the most recent episode spending patterns, expected to be the most accurate predictor of performance year spending, would contribute most strongly to the benchmark price at 50 percent. The remaining 50 percent would be divided into thirds, with baseline year 2 contributing approximately 2/3 , while baseline year 1, which is likely to be the least accurate predictor of performance year spending, would contribute 1/3 .
We sought comment on our proposal at proposed § 512.540(b)(2-3) to use 3 years of baseline episode spending, rolled forward for each performance year, with more recent baseline years weighted more heavily, to calculate TEAM target prices.
The following is a summary of public comments received in response to the proposals to construct the baseline period for benchmarking:
Comment: Many commenters were concerned that a three-year, rolling baseline will make it hard for hospitals to continue making efficiency improvements in later years of the model. Most of these commenters specifically drew attention to the ratchet effect, which means that lower spending in the first few years of the model will be incorporated to target prices resulting in lower target prices in later years of the model. A couple commenters also expressed appreciation for CMS's steps to address the ratcheting effect. A few commenters raised the issue that some episode types, like lower extremity joint replacement, have been tested in bundled payment models for several years, making spending in these episode types already low, which would result in low target prices. A few commenters were concerned with the differential weights applied to each of the baseline years making the ratchet effect more severe, while a commenter supported the differential weighing.
Another commenter was concerned that the benchmarking methodology will be too aggressive for providers who are rural and small, and/or have little experience with value-based care, and it will be challenging for them to meet their targets. Another commenter asked CMS to pay careful attention to regions where most participants are efficient at the start of the model as they will require a higher prior savings adjustment to effectively combat ratcheting while rewarding improvements in care coordination. A third commenter pointed out that the regions are broad and in the proposed rule there are no proposed adjustments for pricing in regions that have higher reimbursed facilities in the mix.
Some commenters proposed alternative approaches to calculating target prices other than using and updating historical data. A commenter supported the use of a three-year baseline but recommended against annual rebasing. Another commenter suggested to set a one-time baseline score that is in place for the entirety of the model. A commenter suggested to conduct a study to understand the optimal utilization of services and use that information to set target prices. A few other commenters suggested to incorporate administratively set benchmarks to mitigate the ratchet effect. Another commenter suggested using a retrospective peer group level trend factor adjustment similar to the BPCI Advanced model with an asymmetrical cap so that target prices are not lowered too much due to improvements in care delivery at the time of the performance period reconciliation. A commenter suggested to exclude a TEAM participant's own beneficiaries from regional benchmark calculations referring to similar requests made in the Medicare Shared Savings Program.
Response: We thank commenters for sharing their support for our attempt to address the ratchet effect as well as concerns regarding the ratcheting effect and hospitals' sustained opportunities for savings for the duration of the model. Target prices in TEAM were proposed to be based on regional average spending making TEAM an achievement-based model. In this framework, participant hospitals will not compete against their historical selves but rather strive to outperform their regional peers. Individual improvements will not affect future target prices in a substantive way as the future benchmark is being calculated based on the performance of several hospitals, including those that are not mandated to participate in TEAM. High-performing hospitals will likely continue to receive rewards and avoid being penalized on cost performance given that they can expect to continue having lower spending than their peers.
We acknowledge that some hospitals (small, rural, those serving socially disadvantaged beneficiaries, etc.) may find it harder to meet target prices and compete against other hospitals in their region. We expect the finalized risk adjustment methodology, which will adjust target prices to account for additional beneficiary-level and provider-level characteristics, as discussed in section X.A.3.d.(4) of the preamble of this final rule, to mitigate some of these concerns. Additionally, we acknowledge the challenges faced by safety-net hospitals, as defined in section X.A.3.f of the preamble of this final rule, and will provide flexibilities, as discussed in section X.A.3.a.3 of the preamble of this final rule, to avoid downside risks and allow them safety net hospitals to remain in Track 1 for the first three performance years and eligible to participate in Track 2 in Performance Years 4 and 5 with a lower stop-loss/stop-gain threshold than other hospitals.
Weighting the baseline years equally or differentially results in the same unadjusted regional target prices in the currently proposed framework, thus it does not mitigate or exacerbate the ratchet effect. Prior to calculating the weighted average, baseline episode spending for the first two years of the baseline is trended to the third and most recent year of the baseline period using a ratio of average episode spending in the third baseline year to average episode spending in the specified baseline year, so all components of the weighted average are going to be equal to average spending in the third baseline year. Weighting only plays a meaningful role in the proposed risk adjustment methodology.
We appreciate commenters' suggestions on using alternative approaches to setting target prices. We disagree that a static baseline or a one-time baseline score are appropriate for TEAM. We are finalizing the inclusion of a capped 3 percent retrospective trend factor adjustment applied to reconciliation target prices, as discussed in section X.A.3.d.(3)(f) of the preamble of this final rule. Because of the capping, using a static baseline will lead to more inaccurate trends in subsequent model years, and hence will adversely affect the accuracy of the benchmarks in later years if the baseline remains static. Further, with a static baseline, the risk adjustment coefficients would get less accurate with each passing year. ( print page 69750)
We also disagree with excluding a TEAM participant's own beneficiaries from regional benchmark calculations. This would result in a more complicated target pricing methodology, since each provider in the same region would receive a slightly different unadjusted target price. Additionally, the proposed target price methodology in TEAM relies on average episode spending and average trends across all hospitals in a particular region so the fraction of the episodes belonging to an individual participant and hence their individual influence on the unadjusted regional target price should be low.
Commenters suggested we use administratively set benchmarks. We disagree that administratively set benchmarks are appropriate for TEAM. Administratively set benchmarks appear to be more appropriate for population-based models, like ACOs. Administrative trends used for ACOs are based on a mix of services that are different from the mix of services received after TEAM procedures. Clinical episode-specific trends can capture changes, like Medicare payment update rates or behavioral changes, more appropriately than administrative trends. However, we will continue to consider alternative approaches to trending episode spending, and if we believe adjustments to our trending approach are necessary, then we will propose such adjustments in future notice and comment rulemaking.
Comment: A commenter asked why CMS used a benchmarking weight of 50 percent on the most recent year, 33 percent on baseline year 2, and 17 percent on baseline year 1 rather than the standard typically used by ACOs of 60 percent for baseline year 3, 30 percent for baseline year 2, and 10 percent for baseline year 1.
Response: We thank the commenter for their concern regarding baseline year weights. We were concerned that creating a benchmark with only one baseline year could risk a ratchet effect to the detriment of TEAM participants and thus proposed using a three-year baseline period as the basis for benchmarking in TEAM. In recognition of the fact that baseline episode spending from more recent years are likely to be a better predictor of performance year spending, we proposed to weight recent baseline episode spending more heavily than episode spending from earlier baseline years. Weighting the baseline years equally or differentially results in the same unadjusted regional target prices in the currently proposed framework, thus it does not mitigate or exacerbate the ratcheting effect. Prior to calculating the weighted average, baseline episode spending for the first two years of the baseline is trended to the third and most recent year of the baseline period using a ratio of average episode spending in the third baseline year to average episode spending in the specified baseline year, so all components of the weighted average are going to be equal to average spending in the third baseline year.
However, weighting too heavily in a given year could have unintended consequences if a certain year is an outlier, for example during a pandemic. Because the purpose of using multiple baseline years is to smooth out potential volatility, we prefer to use a baseline that does not too heavily weight any given year. Because TEAM is an episode-based model rather than a population-based model, accounting for volatility from episode-level variation, and time periods that could be correlated with episodes, is a high priority. Based on previous experience in the Oncology Care Model, we proposed using the 17 percent weight for baseline year 1, 33 percent weight for baseline year 2, and 50 percent weight for baseline year 3. For any factors not accounted for by this benchmarking model, as discussed in section X.A.3.d.(3)(f) of the preamble of this final rule, our proposed trend factor should help to account for any unforeseen or unanticipated changes.
Comment: A commenter recommended that the episode spending should be the actual fee-for-service (FFS) claims submitted for TEAM episodes, not the TEAM episode target prices.
Response: We thank the commenter for their input. All items and services paid under Medicare Part A and Part B FFS will be included in the Clinical Episode, unless those items and services meet certain exclusion criteria. We refer readers to section X.A.3.b.(5)(a) of the preamble of this final rule for the TEAM exclusions list. Medicare spending for non-excluded items and services will be used to calculate episode spending. Episode spending will be capped at the 99th percentile for each DRG and region, as discussed in section X.A.3.d.(3)(e) of the preamble of this final rule.
Additionally, preliminary benchmark prices will be calculated as the average episode spending (after capping) for the DRG, and region trended to baseline year 3 dollar values. The preliminary benchmark price will be adjusted to include adjustments for patient risk score from the performance year, final capped normalization factor, and the discount factor. Episode spending will be compared to the final target price to assess the cost performance of TEAM participants.
Comment: A couple commenters expressed concerns that the proposed rule and correction notice did not address the pricing methodology for spinal fusion MS-DRGs given the deletion of MS-DRGs 453-455 and the addition of eight new MS-DRGs. The new MS-DRGs may not appear in the baseline data and may have significantly different payment rates from the deleted and existing MS-DRGs, thus impacting the Target Price calculations.
Response: We acknowledge the concerns brought up by the commenters regarding the pricing for spinal fusion MS-DRGs. We are finalizing the testing of the spinal fusion episode category, with the updated MS-DRGs, as discussed in section X.A.3.b. of the preamble of this final rule. However, we intend to conduct a thorough review of how the composition of episodes under the current spinal fusion MS-DRGs may change with the introduction of the new MS-DRGs and deletion of three MS-DRGs. We intend to propose a policy in future rulemaking for how to construct target prices when there are MS-DRG or HCPCS modification or other payment system changes that may arise over the course of the model. This policy proposal would address how target prices for spinal fusion MS-DRGs in TEAM would be constructed given the new MS-DRGs did not exist in the baseline period.
After consideration of the comments received, we are finalizing at § 512.540(b)(2-3) the proposal to use 3 years of baseline episode spending, rolled forward for each performance year, with more recent baseline years weighted more heavily, to calculate target prices in TEAM.
In the proposed rule, we proposed to provide to TEAM participants target prices for each proposed MS-DRG/HCPCS episode type and region based on 100 percent regional data for all TEAM participants prior to each PY. That approach would be consistent with PYs 4-8 of CJR. While CJR target prices used a blend of 2/3 hospital-specific data and 1/3 regional data for PYs 1-2, and 1/3 hospital-specific data and 2/3 regional data for PY 3, we stated our reasons in the 2015 CJR Final Rule for moving towards fully regional target pricing as participants gained more experience in the model ( 80 FR73347 ). Target prices based on hospital-specific data would require a TEAM participant to compete against its own previous performance, such that improvement over previous ( print page 69751) performance would result in a reconciliation payment. Conversely, target prices based on regional data would require a TEAM participant to compete against its peers in that region, such that only a specific level of achievement, as opposed to improvement alone, would result in a reconciliation payment. For TEAM participants that are historically inefficient compared to their peers, hospital-specific target prices would be higher than regional target prices because hospital-specific baseline episode spending would be greater than average baseline episode spending for the region. For TEAM participants that are historically efficient compared to their peers, hospital-specific target prices would be lower than regional target prices because hospital-specific baseline episode spending would be lower than average baseline episode spending for the region. We noted in the 2015 CJR Final Rule that if we used 100 percent hospital-specific pricing in CJR, historically efficient hospitals could have fewer opportunities for achieving additional efficiencies under the model and would not be rewarded for maintaining high quality and efficiency, whereas less efficient hospitals would be rewarded for improvement even if they did not reach the same level of high quality and efficiency as the more historically efficient hospitals.
However, as described in section X.A.3.f of the preamble of this final rule, health equity has been a priority in the proposed design of TEAM. We are concerned by literature stating that safety net hospitals in CJR were disproportionately likely to owe a repayment once we moved to 100 percent regional pricing. [ 927 928 ] We note that these findings reflect the original CJR payment methodology, which did not include risk adjustment at the beneficiary level. For PYs 6-8, the modified CJR payment methodology incorporates beneficiary level risk adjustment, including an adjustment for dual income eligibility. Additionally, although we provided lower stop-loss limits for rural and low volume hospitals, we did not identify or provide protective stop-loss limits for safety net hospitals.
Therefore, in addition to lower stop-loss limits for Track 1 and Track 2 TEAM participants as compared to Track 3 TEAM participants, and the incorporation of additional measures of social need in our beneficiary-level risk adjustment, we considered in the proposed rule an alternative target price proposal to provide Track 1 and Track 2 TEAM participants with 100 percent hospital-specific, rather than regional, target prices. However, given our proposal in the proposed rule to calculate target prices at the MS-DRG/HCPCS episode type level, we are concerned that many Track 1 or Track 2 TEAM participants would not meet the low volume threshold of baseline episodes to calculate reliable target prices for many of the MS-DRG/HCPCS episode types included in TEAM. Additionally, there may be some hospitals that serve a high proportion of underserved populations yet have already achieved high levels of quality and efficiency, such that a 100 percent hospital-specific target price would be disadvantageous.
In the proposed rule, we also considered blending hospital-specific pricing with regional pricing as we did in the first 3 years of CJR. For instance, we considered using a blend of 50 percent hospital-specific data and 50 percent regional data to calculate target prices for Track 1 and Track 2 participants. We further considered using a different blend for Track 1 and Track 2 participants vs. Track 3 participants. For example, we considered using a blend of 2/3 hospital-specific data and 1/3 regional data for Track 1 and Track 2 participants, and a blend of 1/3 hospital-specific data and 2/3 regional data for Track 3 hospitals. However, blending hospital-specific pricing with regional pricing could be subject to the same concerns regarding low volume or disadvantaging efficient hospitals as 100 percent hospital-specific pricing, though to a lesser degree.
We also considered, but did not propose and are not finalizing, calculating target prices at the region/episode category level as compared to our proposed region/MS-DRG/HCPCS level. Calculating target prices at the region/episode category would help to mitigate some concerns with certain MS-DRG/HCPCS episode types having a low volume of episodes in a given region. However, to ensure target prices are sufficiently risk-adjusted to capture spending differences between the different MS-DRG/HCPCS within a given episode category, we considered including MS-DRG/HCPCS risk adjusters in TEAM's risk adjustment methodology if we calculated target prices at the region/episode category level. We sought comment on calculating target prices at the region/episode category level.
We sought comment on our proposal at proposed § 512.540(b)(1) to provide regional target prices to all TEAM participants for each PY during the model performance period. We also sought comment on other potential ways to set target prices for Track 1 or Track 2 TEAM participants, including adjustments to regional target prices for Track 1 or Track 2 TEAM participants, that would decrease the likelihood of safety net hospitals being disproportionately penalized by regional target prices.
The following is a summary of the public comments received on the proposed methodology to construct regional target prices and our responses to these comments:
Comment: A couple of commenters requested additional information about the TEAM pricing methodology. Specifically, a commenter required more details on regional target pricing beyond the use of average episode cost in the nine regions.
Response: We proposed to calculate TEAM target prices using 3 years of baseline data, as discussed in section X.A.3.d.(3)(a) of the preamble of this final rule, trended forward to the performance year, at the level of MS-DRG/HCPCS episode type and region, with updates to be made using the performance year data, as discussed in section X.A.3.d.(3)(f) of the preamble of this final rule. The regions are defined as the 9 U.S. census divisions. Hospitals in the five U.S. territories (American Samoa, Guam, the Northern Mariana Islands, Puerto Rico, and the U.S. Virgin Islands) will be grouped alongside Census Division 9 (that is, the Pacific region).
Episode spending will be capped at the 99th percentile, as discussed in section X.A.3.d.(3)(e) of the preamble of this final rule, for each of the 29 MS-DRG/HCPCS episode types and 9 regions, and the benchmark price will be calculated as the average capped and standardized spending in baseline year 3 dollars for each MS-DRG/HCPCS episode type in each region, resulting in 261 benchmark prices. Benchmark prices will be calculated using all hospitals in a region, regardless of TEAM participation status, except the hospitals specified in § 512.540(b)(1)(ii). CMS will apply a prospective trend factor and a discount factor, as discussed in section X.A.3.d.(3)(g) of the preamble of this final rule, to benchmark prices, as well as a ( print page 69752) prospective normalization factor, as discussed in section X.A.3.d.(4) of the preamble of this final rule, to calculate preliminary target prices. Each TEAM participant within a region will receive the same preliminary target price for an MS-DRG/HCPCS episode type. During reconciliation, these preliminary target prices will be updated by updating the trend (subject to caps) and normalization factor (subject to caps) and by factoring in each participant's realized risk adjustment multipliers.
Risk adjustment multipliers (that is, coefficients) will be calculated and made available to TEAM participants prior to the start of the performance year, so participants would be able to use them to estimate their episode-level target prices. We proposed to use age group, Hierarchical Condition Category (HCC) count, and beneficiary social risk as risk adjusters, and, as referenced in section X.A.3.d.(4) of the preamble of this final rule, are adding an expanded set of risk adjusters including specific HCC indicators for each episode type and provider-level covariates like safety-net status of the participant. The risk adjustment multipliers will be calculated at the MS-DRG/HCPCS level on baseline episodes, using a weighted linear regression where episodes are weighted differentially based on whether they belong to year 1, 2, or 3 of the baseline periods. Episodes from baseline year 1 will be weighted at 17 percent, baseline year 2 at 33 percent, and baseline year 3 at 50 percent. The risk adjustment multipliers will be held fixed and applied to performance year episodes at reconciliation based on the realized case mix of the TEAM Participant in the performance year.
After risk adjusting for the performance year case-mix, CMS will normalize the target prices to ensure that the average of the total risk-adjusted preliminary target price does not exceed the average of the total non-risk adjusted preliminary target price. The final normalization factor will be calculated as the national mean of the benchmark price for each MS-DRG/HCPCS episode type divided by the national mean of the risk-adjusted benchmark price for the same MS-DRG/HCPCS episode type. However, it will be capped should this ratio exceed ±5 percent of the prospective normalization factor. The final target prices will include a retrospective trend (instead of the prospective trend), as discussed in section X.A.3.d.(3)(f) of the preamble of this final rule, which will be capped at being within 3 percent of the prospective trend. The retrospective trend will be calculated as the average capped performance year episode spending at the MS-DRG/HCPCS episode type and region level divided by the capped mean baseline episode spending in baseline year 3 dollars at the MS-DRG/HCPCS episode type and region level. In summary, the final target price will be calculated as the product of the capped mean baseline episode spending in baseline year 3 dollars, the capped retrospective trend, the risk adjustment multiplier using the performance year case-mix, and the capped final normalization factor.
Comment: We received many comments regarding pricing episodes based on region. A couple commenters explicitly supported regional target prices, and a commenter proposed an alternative regional pricing methodology based more closely on BPCI Advanced. Other commenters expressed concerns about the proposed model and some of these proposed modifications to the model methodology. The most commonly expressed concern about regional target prices was that they may not be achievable for particular types of hospitals including safety net hospitals, rural hospitals, “underserved” hospitals, historically high-cost hospitals, and historically low-cost hospitals. A few commenters also expressed concerns that hospitals in regions with high CJR or BPCI Advanced penetration or a lot of historically efficient providers would be at a disadvantage, or that hospitals inexperienced with episode-based models would be at a disadvantage, one of which was particularly concerned about hospitals inexperienced with episode-based models in regions with high historical penetration of episode-based models. A few commenters expressed the concern that census divisions are not sufficiently granular regions. To address their concerns that the regional pricing approach disadvantages particular kinds of hospitals, a few commenters suggested benchmarking hospitals against their own history rather than a regional average, either just for safety net hospitals, or for all hospitals. One of these commenters suggested this as a solution to the social risk adjuster being allegedly inadequate. A commenter also suggested benchmarking hospitals against a blend of regional and hospital-specific history. Another commenter suggested adjusting target prices for safety net status or rural status.
Response: We thank all the commenters for sharing their support as well as concerns. We agree with the comments in support of regional target pricing and are finalizing this type of pricing approach for TEAM, but we have taken into account concerns about particular hospital types. In particular, we will be including additional risk adjusters for patient and hospital characteristics, including hospital safety net status, as discussed in this section and in section X.A.3.d.(4) of the preamble of the final rule.
Regarding the comment proposing an alternative method of regional pricing based more closely on the BPCI Advanced model, we will be maintaining the regional target price approach that more closely follows the CJR model, as outlined in the proposed rule. The regional pricing approach allows CMS to implement a payment methodology that is as transparent and understandable as possible for participants of varying levels of statistical background and knowledge.
Regarding the concerns about the achievability of regional target prices for safety net, rural, and “underserved” hospitals, to improve the fit of the risk adjustment model while remaining conscious of the risk of overfitting (that is, the risk that including too many covariates in the model can make it worse at predicting spending in the performance year), we used a combination of clinical input and Lasso analyses to develop a more extensive list of risk adjusters (refer to section X.A.3.d.(4) for additional details on the analyses) based on risk adjusters used in the BPCI Advanced model. We tested a number of hospital characteristics including a provider's status as a major teaching hospital, rural hospital, rural referral center, safety net hospital; and its bed size (coded as small, medium, large, or extra-large). Based on our findings, we will be adding a few provider-level risk adjusters to the model including flags for safety net status, and bed size. None of the other provider characteristics tested were selected by the Lasso analysis for any of the episode types. Based on further testing with historical data, we expect this will result in higher target prices for safety net hospitals than they would otherwise receive.
Regarding the concern that historically high-cost hospitals would find regional target prices unachievable, we believe that hospitals that are historically high-cost after conditioning out the effect of patient characteristics and important hospital characteristics (such as safety net) are those that should have the greatest opportunities for savings.
Regarding the concern that historically low-cost hospitals would find regional target prices unachievable, this is highly unlikely. Rather, if all hospitals in a region did not change ( print page 69753) their behavior, hospitals that are historically low-cost relative to other hospitals in their region and conditional on their episode risk adjusters, would generally be rewarded under the model.
We recognize that some hospitals with past experience in episode-based models may be better positioned to participate in an episode-based model than hospitals without past experience in episode-based models, since they may already have the infrastructure in place to transform care. We also acknowledge that past performance in an episode-based payment model does not guarantee successful participation in new models. However, with TEAM not slated to take effect until 2026, and with all participants having the option to participate in a no-downside-risk track in PY 1 (and safety net hospitals having the option to participate in a no-downside-risk track in PYs 1-3), as discussed in section X.A.3.a.(3) of the preamble of this final rule, CMS expects hospitals should be able to get the infrastructure in place prior to bearing any downside risk due to TEAM. Further, TEAM participants will be able to leverage learning resources developed for TEAM and those based on lessons learned in the BPCI Advanced and CJR models.
We, however, disagree that hospitals with past experience in episode-based payment models presently have a cost advantage or that hospitals in regions with high BPCI Advanced or CJR penetration have a disadvantage in TEAM. In designing TEAM, we examined distributions of spending for the proposed TEAM episodes “triggered” between January 2019 to June 2023 stratified by past participation in episode-based models (CJR participation in 2022 or 2023 for LEJR, or BPCI Advanced participation in 2022 or 2023 for all TEAM episode types). We found that episode spending distributions were similar for participants in episode-based models and for non-participants. For Coronary Artery Bypass Graft Surgery (CABG), Surgical Hip/Femur Fracture Treatment (SHFFT), Spinal Fusion, and Major Bowel Procedure, we think this result may be driven by participants with higher BPCI Advanced target prices being more likely to remain in the model in the later years of BPCI Advanced. For LEJR, the explanation is less clear.
In regions with a large proportion of historically efficient providers, it is true that the baseline average of episode spending would tend to be lower (conditional on patient mix) than in regions with a large proportion of historically inefficient providers. In advance, it is problematic to distinguish between regions that have large proportions of historically efficient (or inefficient) providers and regions that have structural factors resulting in lower (or higher) resource utilization beyond what is captured in the risk adjustment. However, the trends in regions with a large proportion of historically efficient providers would be expected to be more positive or less negative than in regions with a large proportion of historically inefficient providers as the model progresses, which would mitigate the difference in target prices over time. The use of retrospective trends (albeit subject to a cap), as discussed in section X.A.3.d.(3)(f) of the preamble of this final rule, helps ensure that trends in regions where efficiency is improving over time do not overshoot what is feasible. Over the life of TEAM, we will continue to assess whether there remain opportunities for savings for each clinical episode type and if warranted, will propose adjustments in future notice and comment rulemaking.
Regarding the comments that census division is not a sufficiently granular unit at which to set target prices, we would like to clarify that the target prices will be based on geographically standardized allowed amounts. This geographic standardization removes any geographic adjustments like wage index adjustments and other hospital-specific adjustments and ensures that hospitals are compared based on resource utilization rather than price levels in their local area. Furthermore, setting target prices at more granular levels will result in less stable prices, particularly for smaller MS-DRGs and regions.
CMS did consider using an improvement framework (that is, benchmarking hospitals against their own history) and a blended framework (that is, benchmarking hospitals against a blend of their own history and their region's history) for certain types of hospitals as suggested by some commenters. We do acknowledge that factors that differ systematically between hospitals within a region which may not be captured in the risk adjustment would be accounted for in a hospital-specific target price but are not accounted for in a regional target price. However, hospital-specific and blended target prices disadvantage historically efficient hospitals and create pricing instability for low volume hospitals. Further, CMS expects that risk adjusting for safety net status and the additional flexibilities provided to safety net hospitals will address the concerns about the achievability of regional target prices for these hospitals.
After consideration of the public comments we received, we are finalizing at § 512.540(b) our proposal to provide regional target prices to all TEAM participants for each performance year during the model performance period where region is defined by the U.S. Census Divisions.
As we proposed in the proposed rule a fixed 30-day post discharge episode length as discussed in section X.A.3.b.(5)(d) of the preamble of this final rule, we recognize that there may be some instances where a service included in the episode begins during the episode but concludes after the end of the episode and for which Medicare makes a single payment under an existing payment system. An example would be a beneficiary in an episode who is admitted to a skilled nursing facility (SNF) for 15 days, beginning on Day 26 post-discharge from the TEAM anchor hospitalization or anchor procedure. The first 5 days of the SNF admission would fall within the episode, while the subsequent 10 days would fall outside of the episode.
We proposed in the proposed rule that, to the extent that a Medicare payment for included episode services spans a period of care that extends beyond the episode, these payments would be prorated so that only the portion attributable to care during the episode is attributed to the episode payment when calculating actual Medicare payment for the episode. For non-Inpatient Prospective Payment System (IPPS) inpatient hospital services (for example, CAH) and inpatient post-acute care (PAC) such as a SNF, inpatient rehabilitation facility (IRF), long-term care hospital (LTCH), and inpatient psychiatric facility (IPF) services; we proposed to prorate payments based on the percentage of actual length of stay (in days) that falls within the episode window. For home health agency (HHA) services that extend beyond the episode, we proposed that the payment proration be based on the percentage of days, starting with the first billable service date (“start of care date”) and through and including the last billable service date, that fall within the episode. The proposed policy would ensure that TEAM participants are not held responsible for the cost of services that did not overlap with the episode period.
For IPPS services that extend beyond the episode (for example, readmissions included in the episode definition), we proposed in the proposed rule to separately prorate the IPPS claim ( print page 69754) amount from episode target price and actual episode payment calculations, called the normal MS-DRG payment amount for purposes of this final rule. The normal MS-DRG payment amount would be pro-rated based on the geometric mean length of stay, comparable to the calculation under the IPPS PAC transfer policy at § 412.4(f) and as published on an annual basis in Table 5 of the IPPS/LTCH PPS final rules. Consistent with the IPPS PAC transfer policy, the first day for a subset of MS-DRGs (indicated in Table 5 of the IPPS/LTCH PPS final rules) would be doubly weighted to count as 2 days to account for likely higher hospital costs incurred at the beginning of an admission. If the actual length of stay that occurred during the episode is equal to or greater than the MS-DRG geometric mean, the normal MS-DRG payment would be fully allocated to the episode. If the actual length of stay that occurred during the episode is less than the geometric mean, the normal MS-DRG payment amount would be allocated to the episode based on the number of inpatient days that fall within the episode. If the full amount is not allocated to the episode, any remainder amount would be allocated to the 30-day post-episode payment calculation discussed in section X.A.3(d)(5) of the preamble of this final rule. The proposed approach for prorating the normal MS-DRG payment amount was consistent with the IPPS transfer per diem methodology.
This methodology would be consistent with CJR and is described as applied to CJR in the 2015 CJR Final Rule ( 80 FR 73333 ). We sought comment on our proposed methodology at proposed § 512.555 for prorating services that extend beyond the episode.
We received no comments on this proposal and therefore are finalizing this provision without modification at § 512.555.
Given that we proposed episodes with a 30-day post discharge period, we recognize that some episodes will begin during one performance year and end during the following performance year. In the proposed rule, we proposed that all episodes would receive the target price associated with the date of discharge from the anchor hospitalization or the anchor procedure, as applicable, regardless of the episode end date, which determines the performance year in which the episode would be reconciled. We note that the assignment of target prices based on the date of discharge from the anchor hospitalization, or the anchor procedure is different from CJR, where the target price was assigned based on the episode start date rather than the discharge date, but it is consistent with BPCI Advanced. As noted in section X.A.3.d.(5)(a) of the preamble of this final rule, annual reconciliation is based on episodes that end during a PY, so if an episode extends past the end of a PY, that episode would factor into the next PY's reconciliation, when the episode ends, which is consistent with both CJR and BPCI Advanced. Accordingly, if an episode were to end after the final performance year of the model, we proposed that it would not be reconciled. We sought comment on our proposal at proposed § 512.540(a)(3) for applying target prices to an episode that begins in one performance year and ends in the subsequent performance year.
The following is a summary of comments we received regarding our proposal for on how to address an episode that begins in one performance year and ends in the subsequent performance year and our responses to these comments:
Comment: A commenter supported the proposal to treat episodes that begin in one performance year and end in the subsequent performance year similar to CJR and BPCI Advanced models, but requested clarification on which target year will be affected by the performance.
Response: We thank the commenters for expressing their support for the assignment of episodes that overlap with two performance years. We note that, in the TEAM methodology in the proposed rule, performance years are not separated into two sub-periods based on calendar and fiscal year, as was done in the BPCI Advanced model. The preliminary target price applied to the episode is based on the performance year that the episode is assigned to, in accordance with § 512.540(a)(3) of the proposed rule.
For example, if an episode has an anchor end date in December 2026 but an episode end date in January 2027, the episode is assigned to PY 1 and will have the PY 1 target price applied to it. However, if the episode starts in 2026 but both the anchor and episode end dates are in 2027, the episode is assigned to Performance Year 2 and will have the Performance Year 2 target price applied to it. Both episodes would be reconciled at the same time, along with the other PY 1 and 2 episodes with episode end dates in 2027.
After consideration of the public comments we received, we are finalizing at § 512.540(a)(3) the proposed methodology to assign episodes to performance years and target prices.
Given the broad episode definition and 30-day proposed post-discharge period in the proposed rule, we want to ensure that hospitals have some protection from the downside risk associated with especially high payment episodes, where the clinical scenarios for these cases each year may differ significantly and unpredictably. As we stated in the 2015 CJR Final Rule ( 80 FR 73335 ), we do not believe that the opportunity for a hospital's systematic care redesign of particular surgical episodes has the significant potential to impact the clinical course of these extremely disparate high payment cases. In the 2015 CJR Final Rule ( 80 FR 73335 ) we finalized a policy to limit the hospital's responsibility for high episode payment cases by utilizing a high price payment ceiling at two standard deviations above the mean episode payment amount in calculating the target price and in comparing actual episode payments during the performance year to the target prices. This policy was designed to prevent participant hospitals from being held responsible for catastrophic episode spending amounts that they could not reasonably have been expected to prevent. The policy, and the reasoning behind it, is described in detail at ( 80 FR 73335 ).
However, as we described in 86 FR 23518 , based on data from the first few years of the CJR model, we observed that the original 2 standard deviation methodology was insufficient to identify and cap high episode spending, as more episodes than expected exceeded the spending cap. We describe in detail our reasoning for finalizing a change to the high episode spending cap in the 2021 CJR 3-Year Extension Final Rule ( 86 FR 23518 ). We finalized a change to the calculation of the high episode spending cap to derive the amount by setting the high episode spending cap at the 99th percentile of historical costs for each MS-DRG for each region. The resulting methodology was similar to the BPCI Advanced methodology for capping high-cost episode spending at the 99th percentile for each MS-DRG.
We proposed a similar high-cost outlier policy for TEAM in the proposed rule, to cap both baseline episode spending and performance year episode spending at the 99th percentile of spending at the MS-DRG/HCPCS episode type and region level, referred to as the high-cost outlier cap. We stated ( print page 69755) in the proposed rule we would determine the 99th percentile of spending at the MS-DRG/HCPCS episode type and region level during the applicable time period, and then set spending amounts that exceed the high-cost outlier cap to the amount of the high-cost outlier cap. For instance, if the high-cost outlier cap was set at $30,000, an episode that had actual episode spending of $45,000 would have its spending amount, for purposes of the model, reduced by $15,000 when the cap was applied and therefore, the spending for that episode would be held at $30,000. In the proposed rule, we proposed to use capped episode spending when calculating benchmark prices in order to ensure that high-cost outlier episodes do not artificially inflate the benchmark. When calculating performance year episode spending at reconciliation, we stated in the proposed rule we would use capped episode spending so that a TEAM participant would not be held responsible for catastrophic episode spending amounts that they could not reasonably have been expected to prevent. We sought comment on our proposal at proposed § 512.540(b)(4) for calculating and applying the high-cost outlier cap.
The following is a summary of public comments we received regarding our proposal for calculating and applying the high-cost outlier cap:
Comment: A few commenters raised concerns that setting the high-cost outlier cap at the 99th percentile of the episode spending distribution is too high. A couple of commenters suggested setting it at the 95th percentile and a commenter suggested setting it at the 90th percentile to reduce variability with outlier cases. A couple commenters were especially concerned about the volatility in episode spending of low volume providers. A commenter noted that this may inadvertently punish providers who take the risk of treating more vulnerable and complex patients and may particularly be of concern for rural and/or small providers with limited experience in value-based care.
Response: We thank commenters for sharing their concerns regarding the high-cost outlier cap. We expect that the proposed method of capping at the 99th percentile of the spending distribution will result in high episode spending caps that accurately represent the cost of infrequent and potentially nonpreventable complications within each MS-DRG and region, which the participant could not have reasonably controlled and for which we do not want to penalize the participant. By setting the cap at this level, we are holding hospitals accountable for patients, including complex cases, whose care hospitals can be expected to have reasonable control over. In response to the comments, we analyzed the increase in episode spending between each percentile from the 95th to the 99th in 30-day episodes using Medicare FFS claims from 2021. These increases were between 5 percent to 10 percent for the majority of MS-DRG and region combinations between the 95th and the 98th percentiles, but above 10 percent for the majority of MS-DRG and region combinations between the 98th and 99th percentiles. Setting the cap below the 99th percentile could lead to a too low high-cost outlier cap, which is contrary to the intention of only capping extreme outliers beyond providers' control and allowing reduction in spending for other high-cost episodes.
Additionally, exclusions for low volume or high-cost drugs are going to be applied in both the baseline and performance year, to further prevent hospitals from being penalized for incurring high costs by using necessary, but rare or very expensive treatment options. This approach is consistent with how we applied the high-cost outlier cap in the 2021 CJR 3-Year Extension Final Rule ( 86 FR 23518 ), and is similar to the BPCI Advanced methodology for capping high-cost episode spending at the 99th percentile for each MS-DRG.
Lastly, we acknowledge commenters concerns about low volume providers. Given concerns and our desire to protect low volume hospitals from greater financial risks, we are not finalizing our low volume hospital policy, as discussed in section X.A.3.d.(3)(h) of the preamble of this final rule. We intend to propose a new policy in future notice and comment rulemaking prior to TEAM being implemented.
After consideration of the public comments we received, we are finalizing at § 512.540(b)(4) the proposal for calculating and applying the high-cost outlier cap.
Target prices are derived from a prediction based on previous Medicare spending patterns, but it is not possible to perfectly predict how Medicare spending patterns may change over the course of the performance year. In the original BPCI model, prospective target prices were not provided to participants, so the trend factor was calculated retrospectively based on the observed spending during the performance period. Quarterly reconciliations in BPCI meant that participants could gain a sense of how their target prices tended to change over time and get relatively frequent feedback on their performance in the model. However, BPCI participants did not like the uncertainty of not knowing their target prices in advance.
In the initial CJR methodology and Model Years 1-3 of BPCI Advanced, CMS provided fully prospective target prices to participants. Participants appreciated the certainty of prospective target prices, where we predict in advance how spending patterns might shift and hold those target prices firm even if we underpredicted or overpredicted spending. This methodology included applying update factors to account for setting-specific payment system updates, allowing us to estimate how a given set of services performed during the baseline would be priced had those same services been subject to the fee schedules in effect during the performance period.
In CJR, we originally overpredicted performance period spending, not accounting for the overall decline in spending on LEJR episodes nationwide that occurred outside of the model during its first few performance years. In BPCI Advanced, we similarly overpredicted performance period spending for certain episodes because our methodology was unable to account for medical coding changes that occurred between the baseline and performance period, or during the performance period itself. For instance, in FY 2016, changes to medical coding guidance were made for Inpatient Congestive Heart Failure, such that certain patients who during the baseline would have been coded as the less expensive MS-DRG 292, were instead coded as the more expensive MS-DRG 291, despite having the same clinical characteristics. This meant that many beneficiaries who received a target price associated with the more expensive MS-DRG 291, actually had the lower performance period costs previously associated with the less expensive MS-DRG 292. The use of a fully prospective trend factor was unable to capture these changes in both practice patterns and coding guidelines.
Subsequently, we modified both models' methodologies to include a retrospective trend adjustment. Starting in Model Year 4, we continued to provide BPCI Advanced participants with a prospective target price using an estimated trend factor, but we adjusted the target price at reconciliation based on the retrospective calculation of the trend factor using performance period data. Initially, this policy included ( print page 69756) guardrails around the magnitude of the retrospective trend factor adjustment of ±10 percent. In response to participant feedback, we lowered the maximum level of the retrospective trend factor adjustment to ±5 percent starting in Model Year 6.
In the CJR extension, the retrospective trend is known as the market trend factor adjustment. It is fully retrospective and calculated at reconciliation, meaning that the unadjusted target price we post on the CJR website prior to the performance year does not include a prospective trend factor. In response to participant requests, we provided estimates of the market trend factor on the CJR website based on the most recently available data to help participants estimate their potential target prices. The market trend factor is calculated separately for each MS-DRG/region combination. For the PY 6 reconciliation (corresponding to episodes that ended between October 1, 2021, and December 31, 2022), the highest market trend factor was 1.294 for MS-DRG 469 episodes in the West South Central region, while the lowest market trend factor was 0.972 for MS-DRG 521 episodes in the New England region.
For TEAM, we proposed in the proposed rule to provide preliminary target prices that incorporate a prospective trend factor to TEAM participants. We stated at proposed § 512.540(b)(7) to calculate this prospective trend factor as the percent difference between the average regional MS-DRG/HCPCS episode type expenditures computed using the most recent year of the applicable baseline period, and the comparison average regional MS-DRG/HCPCS episode type expenditures during the first year of the baseline. By comparing baseline year 3 to baseline year 1, the prospective trend would capture changes across a two-year period, which we believed would be appropriate given that we would be projecting spending patterns in the performance year, which would be two years after baseline year 3. The trend factor calculation as proposed in the proposed rule would be similar to how the market trend factor is currently calculated in the CJR extension, but instead of retrospectively comparing average regional MS-DRG/HCPCS episode type spending during the performance year to spending during the baseline year, the calculation would be performed prospectively, so that performance year expenditures would not be considered. A fully prospective trend factor would give participants more certainty about what their reconciliation target prices would be, although reconciliation target prices as proposed in the proposed rule would incorporate both beneficiary-level risk adjustment and an adjustment to the prospective normalization factor, as applicable (as described in section X.A.3.d.(4) of the preamble of this final rule).
Given our proposal in the proposed rule to use a prospective trend factor to predict future spending for the purposes of pricing stability, we considered but did not propose to include update factors that take into account Medicare payment systems updates for each fiscal year (FY) or CY and could improve pricing accuracy. Specifically, we considered a methodology similar to BPCI Advanced and Performance Years 1-5 of CJR, where preliminary target prices are updated to reflect the most current FY and CY payment system rates using setting-specific update factors for payment system, including the IPPS, the Outpatient Prospective Payment System (OPPS), the Physician Fee Schedule (PFS), the Home Health Prospective Payment System (HH PPS), the Medicare Economic Index (MEI), the IRF Prospective Payment System (PPS), and the SNF PPS. However, updating target prices using setting-specific update factors would result in TEAM participants receiving more than one target price for a MS-DRG/HCPCS episode type in a performance year which can increase complexity. Further, while including update factors would generally increase target prices, it also decreases pricing stability since the preliminary target price would change due to the application of update factors. We sought comment on whether we should include setting-specific update factors in preliminary target prices to improve pricing accuracy, or if there are other ways, we should consider updating target prices that would reflect Medicare payment system updates.
We considered, but did not propose, an alternative proposal to adjust the preliminary target price at reconciliation based on the observed trend during the performance year. We considered proposing to limit the magnitude of this retrospective trend adjustment by applying guardrails, similar to what we currently do in BPCI Advanced. Specifically, if the trend factor calculated at reconciliation based on performance year expenditures differed from the prospective trend factor by up to ±5 percent, we considered, but did not propose, to adjust the preliminary target price at reconciliation by applying the final trend factor to the baseline target price. We considered, but did not propose, only adjusting the preliminary target price by ±5 percent if the final trend factor differed from the prospective trend factor by more than ±5 percent. In other words, that the maximum upward trend adjustment we would make to the preliminary target price at reconciliation would be 5 percent, and the maximum downward trend adjustment we would make to the preliminary target price at reconciliation would be −5 percent. We also considered lower percentages for the guardrails, including 3 percent and 1 percent, given the BPCI Advanced model's experience initially having a higher percentage maximum adjustment and then reducing the percentage in later years of the model. We considered these alternative proposals because we believed that these guardrails would help us achieve a balance of providing predictability to participants and mitigating the risk that target prices would be disproportionately impacted by performance year shifts in spending patterns that could not have been foreseen.
We also requested comment on alternative ways to calculate the trend factor to both increase accuracy of prospective target prices and to mitigate the ratchet effect. We recognized that spending on some episodes, such as Lower Extremity Joint Replacement, has been decreasing over time and may reach a point where further decreases in spending could compromise quality and patient safety. While in the early years of CJR, our target prices failed to account for decreasing trends in spending for LEJR nationwide and thus were overinflated, that downward trend has since stabilized, suggesting that there may no longer be as much of an opportunity for participant savings as there was in the early years of CJR. In the case of an episode where spending has been decreasing but has since stabilized, trending the target price forward based on previous years' trends could result in target prices that are too low. In such a scenario, a retrospective trend adjustment might actually result in a higher target price than a fully prospective trend. We sought comment on ways to construct a trend factor that can result in a reasonable target price regardless of whether spending has been increasing, decreasing, or stabilizing.
For example, in the CY 2023 Physician Fee Schedule final rule, CMS finalized a policy to include a prospectively-determined component, the Accountable Care Prospective Trend (ACPT), in the factor used to update the benchmark to the performance year for accountable care organization (ACO) agreement periods starting on or after January 1, 2024 (see 87 FR 69881 to 69898) to help address the ratchet effect ( print page 69757) by insulating a portion of the update factor from the impact that ACO savings can have on retrospective national and regional spending trends. This type of trend is referred to as an administrative trend because it is not directly linked to ongoing observed FFS spending. However, we recognized that there may be some concerns using administrative trends for episode-based payment models, as opposed to population-based payment models like ACOs, because administrative trends may not capture episode-specific trends, which could lead to higher or lower preliminary target prices when compared to actual performance year spending. We requested comment on this type of trending approach, or other potential ways to increase the accuracy of prospective target prices and mitigate the ratchet effect when we update TEAM target prices.
We sought comment on our proposal at proposed § 512.540(b)(7) for calculating and applying a prospective trend factor.
The following is a summary of the comments received on the trending approach for TEAM, and how to include payment system updates and our responses to these comments:
Comment: A couple commenters requested that TEAM include Medicare payment system updates in the target prices. A commenter specifically expressed concerns that not applying update factors to update prices from the baseline year payment rates to the most current payment rates can lead to inaccurate target prices, specifically when there are major Medicare rate updates not reflected in the historical data.
Response: We acknowledge the concerns related with not applying payment system updates to the target prices. CMS will conduct additional analyses to understand the impact of update factors on the target price methodology and intends to include policy proposals in future notice and comment rulemaking, prior to the implementation of TEAM.
Comment: A couple of commenters suggested testing administrative trends in TEAM while another commenter supported the use of regional benchmark prices but urged to exercise caution while using Accountable Care Prospective Trend or ACPT (that is, administrative trends) because it could impact regions where the spending growth is faster than the national inflation.
A few commenters recommended using retrospective trends for TEAM similar to the peer group trend (PGT) Factor adjustment used in BPCI Advanced. One of the commenters suggested that applying a PGT Factor Adjustment would solve the ratcheting effect which specifically impacts the episodes for major joint replacement of the lower extremity. Another commenter suggested that applying retrospective trends would be critical for a mandatory model to ensure greater accuracy of the target prices and to reduce the financial stress on the participants.
Additionally, commenters made some suggestions on the capping for retrospective trends. A commenter suggested using an asymmetrical(−2/+5 percent) cap on the target prices so that the target prices do not get lowered substantially in the performance year due to improvements in care. Another commenter suggested that upper and lower bounds of 5 percent are too high and would subject the data to wide variations exacerbating the difficulty to trend the data and impede predictions.
Response: We thank the commenters for their suggestions about testing administrative trends for TEAM. CMS continues to believe that administrative trends may be more applicable for population-based models like ACOs especially because ACOs are likely to have a high market penetration at the regional level and thus, the regional trends can negatively impact their benchmark prices through strong historical performance. Clinical episode-specific trends are more appropriate for an episode-based model like TEAM which can capture changes including Medicare payment rate updates or behavioral changes specific to the episode categories. We agree with the comment that administrative trends can have a negative impact on the target prices leading to higher or lower preliminary target prices for some clinical episode categories which can put both CMS and providers at risk for high losses.
We acknowledge the comments on using PGT Factor Adjustment to solve the ratcheting effect and capping for target prices in the performance year. For TEAM, we proposed, and are finalizing, regional target prices where the participant hospitals' performance will be measured relative to their peers and not based on improvement relative to their own historical performance. This will mitigate concerns associated with the individual ratcheting effect. We acknowledge the concern for some clinical episode categories like LEJR which has been tested in both the CJR and BPCI Advanced models with a high participation rate. However, we disagree that the participating providers have a disadvantage in TEAM. CMS analyzed the post-discharge and post-acute care spending among providers participating and not participating in CJR and BPCI Advanced models and observed that both groups had similar spending trends, suggesting that there were opportunities for savings for LEJR in the post-discharge period for all providers.
We also acknowledge the suggestion that implementing retrospective trends (that is, adjusting the preliminary target price at reconciliation based on the observed trend during the performance year), will lead to target prices that more accurately reflect spending patterns during the performance period. We agree that retrospective trends will account for significant changes in the spending patterns in the performance year that are not accounted for in the baseline and make the target prices more comparable to the performance year spending. As discussed in the proposed rule, a retrospective trend adjustment might actually result in a higher target price than a fully prospective trend if episode spending shows a decrease in the baseline but has since stabilized, since trending the target price forward based on previous years' trends could result in target prices that are too low. An internal analysis simulating reconciliation as proposed in TEAM demonstrated that more TEAM participants would owe CMS a repayment amount than would earn a reconciliation payment amount due to prospective trends resulting in lower target prices than would have been calculated with a retrospective trend. The analysis also simulated different types of trend construction, including fully prospective, as stated in the proposed rule for TEAM, fully retrospective, and then capped at varying percentages, including 1 percent, 3 percent, and 5 percent. Results were least favorable for TEAM participants with a prospective trend and most favorable for TEAM participants with a retrospective trend, with the capped trends falling in between. We note that this simulation did not include behavioral effects, nor does it represent the same baseline period or performance years of TEAM. However, it does highlight the increased risk the TEAM participant may be exposed to when relying on a fully prospective trend that cannot capture unanticipated spending changes or may not be able to predict spending that is tapering off.
Given these findings, and commenters' concerns regarding too much financial risk in TEAM, we are finalizing our trend proposal with slight modifications to include a 3 percent ( print page 69758) capped retrospective trend factor adjustment applied during reconciliation to construct reconciliation target prices. While the prospective trend calculates average regional episode spending that occurred during the baseline period, the retrospective trend factor calculates realized average regional episode spending that occurred during the performance year. Thus, the retrospective trend factor adjustment will be calculated by taking the average regional capped performance year episode spending for each MS-DRG/HCPCS episode type divided by the average regional capped baseline period episode spending for each MS-DRG/HCPCS episode type. The retrospective trend factor adjustment will be capped at 3 percent, meaning that the maximum difference between the prospective trend and retrospective trend is 3 percent. We believe including a 3 percent capped retrospective trend adjustment will protect TEAM participants and CMS from excessive risk, while balancing predictability and stability for TEAM participants. Table X.A.-10 is an example of how the capped retrospective trend factor adjustment would be applied.

We note that caps on the performance year target prices protect both CMS and TEAM participants from significant changes in the spending patterns between the baseline year and performance year. We agree that lowering the cap would increase the predictability of target prices and drive improvements. However, applying an asymmetrical retrospective trend factor adjustment in the performance year would put the TEAM participants or CMS at a high risk for loss. Thus, we think applying a symmetrical capping for the retrospective trend factor adjustment in the performance year at ±3 percent provides both protection and predictability to TEAM participants.
After consideration of the public comments we received, we are slightly modifying our trending approach at § 512.545(f) to apply a retrospective trend factor adjustment to the reconciliation target prices. The retrospective trend factor adjustment will be calculated by taking the average regional capped performance year episode spending for each MS-DRG/HCPCS episode type divided by the average regional capped baseline period episode spending for each MS-DRG/HCPCS episode type. The retrospective trend factor adjustment will be capped at 3 percent, meaning that the maximum difference between the prospective trend and retrospective trend is 3 percent.
In addition to the prospective trend factor, at proposed § 512.540(c) we proposed to apply a discount factor to the benchmark price when calculating preliminary target prices. Specifically, we proposed in the proposed rule to apply a 3 percent discount factor to the benchmark price to serve as Medicare's portion of reduced expenditures from the episode. This discount would be similar to the 3 percent discount factor applied to target prices in the CJR model and to surgical episode target prices in BPCI Advanced.
However, we recognize that there may be different levels of opportunity for savings within different episode types. For instance, in BPCI Advanced, in recognition of the fact that participants were generally able to achieve greater savings in surgical, as opposed to medical, episodes, we incorporated a 3 percent discount into surgical episode target prices and a 2 percent discount into medical episode target prices. Given differential opportunities for ( print page 69759) savings across the different types of proposed episode categories, as well as our intention to incorporate additional episodes in future years of TEAM, we considered but did not propose varying the Medicare discount based on episode category. Specifically, we considered but did not propose lower discount factors including 2 percent, 1 percent, 0.5 percent, or no discount factor. We also considered but did not propose linking the discount to variability in episode spending during the baseline, such that an episode with minimal variability in baseline spending might have a lower discount percentage, given that lower variability in baseline spending might indicate fewer opportunities for savings in that episode, as opposed to episodes with greater spending variability. We also considered but did not propose lower discount factors, including 2 percent, 1 percent, 0.5 percent, or no discount factor, for specific types of TEAM participants. For example, we considered no discount factor for safety net hospitals given the proportion of underserved beneficiaries they care for and many of these safety net hospitals may be new to episode-based payment participation. Although we did not propose these alternatives in the proposed rule, we sought comment on whether we should include any of these alternatives in TEAM and also sought comment on different ways to adjust the Medicare discount based on differential savings opportunities for different episode types.
We sought comment on our proposal at proposed § 512.540(c) to apply a 3 percent discount factor to preliminary episode target prices for episodes.
The following is a summary of the public comments received on this proposal and our responses to those comments:
Comment: Numerous commenters expressed concern with the 3 percent discount rate for TEAM episodes and requested the discount rate be lower or eliminated. Many commenters recommended using different discount factors by episode category. Some commenters suggested applying discount factors only to post-acute care spending. Many commenters cited reasons for concern that were related to the shortened episode window of 30 days compared to the 90-day episode windows in the other episode-based payment models, differences between hospitals within certain regions such as high performers or hospitals with previous experience in value based care such as CJR or BPCI Advanced, larger portions of the episode being related to the surgical procedure rather than post-acute care, threats to quality of care, and that the discount rate would harm hospital operating margins and ability to invest in infrastructure.
Response: We thank commenters for sharing their concern regarding the 3 percent discount rate. We acknowledge that the shorter episode length compared to previous and current models that have used a 3 percent discount factor coupled with a 90-day post-discharge episode length (post-discharge period), means there is less spending captured in a 30-day post-discharge period, resulting in less potential for savings opportunities. However, target prices will also be calculated based on the shorter 30-post-discharge period and should taper target prices relative to 90-day post-discharge period.
We disagree with commenters that previous models have eliminated potential reductions in spending for TEAM participants. Participants in CJR and BPCI Advanced have continued to achieve reductions in spending without overall reductions in quality over the years. As noted in section X.A.3.d.(3)(b), we also do not agree that hospitals with past experience in episode-based payment models presently have a cost advantage or that hospitals in regions with high BPCI Advanced or CJR penetration have a disadvantage in TEAM. In designing TEAM, we examined distributions of spending for the proposed TEAM episodes “triggered” between January 2019 to June 2023 stratified by past participation in episode-based models (CJR participation in 2022 or 2023 for LEJR, or BPCI Advanced participation in 2022 or 2023 for all TEAM episode types). We found that episode spending distributions were similar for participants in episode-based models and for non-participants. For CABG, SHFFT, Spinal Fusion, and Major Bowel Procedure, we think this result may be driven by participants with higher BPCI Advanced target prices being more likely to remain in the model in the later years of BPCI Advanced. For LEJR, the explanation is less clear. Furthermore, TEAM will oversample mandatory core-based statistical areas (CBSAs) with safety net hospitals and low past exposure to bundled payment models, capturing more hospitals that may not have been influenced to spending reduction shifts in their markets, which may present greater opportunity to find efficiencies and savings. Therefore, we believe that TEAM participants will have potential for reductions to episode spending without reductions in quality of care.
We disagree that high performing, or more efficient, hospitals in certain regions will have less room to achieve further reductions in spending to meet their target price after the discount rate is factored in. This is because target prices are set regionally, high performing hospitals may be well-positioned to meet their target prices for episodes since less efficient hospitals in a region will tend to drag regional prices higher.
We disagree that the discount factor, if set at a reasonable percentage, will disincentivize quality of care or investment in the infrastructure needed to succeed in TEAM. The discount factor is intended to reflect Medicare's potential savings from TEAM, and once applied to benchmark prices to create a target price, may help to motivate providers to closely manage beneficiaries to improve their quality care and care transitions to avoid readmissions and unnecessary spending. However, we recognize that a discount factor that is set too high, may create an unreasonable target price, and limit the TEAM participant's ability to earn a reconciliation payment amount, even if they were able to reduce episode spending and provide high-quality care. Further, we acknowledge that a shorter episode length, that encompasses the anchor hospitalization or anchor procedure plus the 30-day post-discharge period results in larger portions of the episode spending being captured by the anchor hospitalization or anchor procedure and less episode spending captured during the 30-day post-discharge period. Current models like the CJR and BPCI Advanced model, have demonstrated that most participants were able to achieve savings primarily through reductions in post-acute care spending. Therefore, we recognize that a 3 percent discount may be too high for TEAM participants for the shorter episode lengths used in TEAM.
Given commenters concerns, we are persuaded that the discount factor in TEAM should be reduced and further took into consideration commenters' recommendations applying different discount factors for each episode category, as compared to a blanket discount factor where all the episode categories receive the same discount factor. We disagree with applying a discount factor only to post-acute care spending because there may be some episodes where there isn't any post-acute care spending. Additionally, we want to spur TEAM participants to find savings opportunities in other areas, such as reducing readmissions, and applying a discount factor to just one ( print page 69760) type of cost in the post-discharge period may not incentivize TEAM participants to identify improvements or efficiencies elsewhere. We do see value in setting different discount factors based on the episode category and their proportion of anchor hospitalization or anchor procedure spending, relative to their 30-day post-discharge period spending, given BPCI Advanced and CJR evaluation results demonstrating most participants achieve savings in post-acute care spending reductions.
We acknowledge certain episode categories may have a higher proportion of anchor hospitalization spending compared to other episode categories and that in itself may create challenges for TEAM participants to reduce spending. Our review of the proportion of anchor hospitalization and anchor procedure spending relative to the 30-day post-discharge spending identified the CABG and Major Bowel Procedure episodes categories had higher anchor hospitalization spending compared to LEJR, SHFFT, and Spinal Fusion episode categories. Given these differences, we believe a 1.5 percent discount factor for CABG and Major Bowel Procedure and a 2 percent discount factor for LEJR, SHFFT, and Spinal Fusion, are appropriate discount factors. We are finalizing a lower discount factor for CABG and Major Bowel Procedure because we recognize that there may be fewer opportunities for savings on these episodes as compared to LEJR, SHFFT, and Spinal Fusion. We believe these modified discount factors are more in-line with a 30-day post-discharge period and will allow TEAM participants to reduce spending and have opportunities to receive a reconciliation payment amount from CMS. Therefore, we will be finalizing these updated discount factors.
We note that we are cautious to assume that higher post-discharge spending necessarily means larger opportunities to reduce spending and achieve savings. For example, it may be clinically appropriate to have higher post-discharge period spending for a given episode category, because beneficiaries in that episode category require more institutional post-acute care spending and reducing that could compromise quality of care. We will monitor for unintended consequences and if warranted, will adjust policies in future notice and comment rulemaking.
Comment: Some commenters believe that the discount rate should be reduced or eliminated for safety net hospitals, rural hospitals, or hospitals with little to no experience in value-based care.
Response: We thank commenters for sharing their concern regarding safety net hospital and rural hospital participation in TEAM. We disagree that safety net hospitals and rural hospitals, as defined in section X.A.3.f of the preamble of this final rule, should have a different discount rate than other TEAM participants. However, safety net hospitals will be insulated from downside risk by being allowed to stay in Track 1, if they elect to do so, for the first three years of the model, as discussed in section X.A.3.a.(3) of the preamble of this final rule. Rural hospitals, and safety net hospitals, may elect to participate in Track 2, that has a stop loss/gain limit of ±5 percent to limit potential losses in revenue. Furthermore, TEAM participants in Track 2 will be subject to Composite Quality Score (CQS) adjustment up to 10 percent for positive reconciliation amounts and up to 15 percent for negative reconciliation amounts. This will also help to insulate rural hospitals and safety net hospitals from greater financial risk, while incentivizing cost reductions and improvements in quality of care.
Comment: A few commenters expressed concern that rebasing the target prices annually will lead to a ratcheting effect and make the discount factor infeasible even with a baseline period of the previous three years. A commenter said that heavier weighting on the most recent year will exacerbate this effect.
Response: We disagree with commenters that rebasing the target prices annually will make the discount factor unachievable and that the three-year baseline period for target prices is inadequate. In the 2021 CJR 3-Year Extension Final Rule, we finalized a policy to use a 1-year baseline period that would move forward every year (with the exception of skipping data from 2020 due to COVID-19 irregularities) ( 86 FR 23514 ). In combination with a retrospective market trend factor, using 1 year of baseline episode spending updated every year meant that the target prices would not be inflated as they had been under the initial CJR methodology. Moving the baseline period forward every year in TEAM like in the CJR extension while using multiple years in the baseline period like in the original CJR methodology will allow for both a baseline that is not subject to undue volatility and matches any changes to spending that may have happened for reasons other than TEAM.
Comment: A few commenters suggested that either the 30-day episode window be extended to 90 days, or the 3 percent discount factor be reduced, stating that the 30-day episode window means a greater proportion of the episodes are from the surgical procedure and there are fewer opportunities for cost reductions.
Response: We thank the commenters for their suggestions regarding the 30-day post-discharge episode length and the 3 percent discount factor. While we disagree that the 30-day post-discharge episode length should be extended to 90 days, as discussed in section X.A.3.b.(5)(d) of the preamble of this final rule, we agree that the shorter episode length creates fewer opportunities for spending reductions as a proportion of the episode spend in TEAM. As noted earlier, we are finalizing a modification to the discount factor that will reduce the percentage to 1.5 percent for CABG and Major Bowel Procedure episode categories and 2 percent for LEJR, SHFFT, and Spinal Fusion episode categories. We believe this updated discount factor paired with the shorter episode length will balance commenters concerns about savings opportunities and driving spending reductions.
Comment: A couple of commenters expressed concern that hospitals may be prohibited from employing physicians directly and thus do not control all aspects of spending related to TEAM episodes. This makes the ability of participants to reduce spending in an amount that is more than the discount factor, and thus achieving savings, onerous.
Response: We disagree that hospitals will be unable to achieve savings if they do not employ their own physicians. While some physicians may not be employed by a TEAM participant as proposed, CMS outlines physicians as proposed TEAM collaborators in section X.A.3.g.(3) of the preamble of the proposed rule, which includes skilled nursing facilities, home health agencies, long-term care hospitals, inpatient rehabilitation facilities, physicians, nonphysician practitioners, therapists in a private practice, comprehensive outpatient rehabilitation facilities, provider or suppliers of outpatient therapy services, physician group practices, hospitals, critical access hospitals, non-physician provider group practices, therapy group practices, and Medicare ACOs.
Comment: A couple of commenters recommended phasing in the discount factor. A commenter suggested a 1 percent discount, or a 2 percent discount for the first two years of the model followed by 1 percent, thereafter, would better reflect the risk and potential financial jeopardy TEAM ( print page 69761) could create from the annual rolling baseline.
Response: We thank commenters for sharing their concern regarding the 3 percent discount rate. We agree with the commenters' suggestion to reduce the discount rate and are finalizing the policy to reduce the CMS discount to 1.5 percent for CABG and Major Bowel Procedure episode categories and 2 percent for LEJR, SHFFT and Spinal Fusion episode categories. However, we disagree with the concept that the discount rate should be phased in or reduced further after the first two years of TEAM. We do not believe phasing in the discount factor is appropriate given the opportunity for all TEAM participants to participate in Track 1 with no downside risk in the first performance year, as discussed in section X.A.3.a.(3) of the preamble of this final rule. We also believe that the annual rolling baseline will not be a significant risk to TEAM participants. Additionally, we will continue to monitor and analyze TEAM Participant performance and target prices with respect to an appropriate discount rate and if warranted, would propose any policy changes in future notice and comment rulemaking.
Comment: A commenter described how the 3 percent discount factor may be harder to continually reach after inefficiencies are removed.
Response: We thank the commenter for their suggestion. We agree that a 3 percent discount factor, or any discount factor percentage, may not be a policy that should remain in perpetuity if inefficiencies are removed. However, we believe there are opportunities to reduce spending and eliminate inefficiencies given the variation in spending we have observed across the different episode categories when looking at a national set of hospital spending.
Comment: A commenter expressed concern that the discount factor is onerous for hospitals facing thin margins after changes in the labor market and absorption of new Medicaid obligations and that ideally TEAM will encourage a positive sum relationship between CMS and hospitals.
Response: We thank the commenter for sharing their concern regarding the 3 percent discount rate. We disagree with the commenter that TEAM is onerous for hospitals that have large obligations to Medicaid patients. While the model does not include Medicaid patients, we believe the care redesign processes that TEAM participants may implement can be applied to other populations of patients, encouraging greater care transformation and opportunities to improve patient care and reducing spending, within and outside of the model. We agree with the commenter's suggestion to reduce the discount rate and are making modifications to reduce the discount rate to be 1.5 percent for CABG and Major Bowel Procedure episode categories and 2 percent discount factor for LEJR, SHFFT, and Spinal Fusion episode categories to grant hospitals more flexibility to meet their target prices for TEAM.
Comment: A couple of commenters had concerns that the 3 percent discount factor can impact post-acute care discharge destinations. A commenter is concerned that the 30-day episode window and 3 percent discount rate will disincentivize discharge to IRF even when it is the optimal post-acute care setting for a patient. Another commenter cited concerns regarding patient freedom to select a post-acute care provider of their choice.
Response: We thank the commenter for sharing their concern regarding patient care and the discount rate. TEAM participants may not limit access to medically necessary items and services, nor limit the TEAM beneficiary's choice of Medicare providers and suppliers, including post-acute care providers such as long-term care hospitals and inpatient rehabilitation facilities. This means that TEAM beneficiaries are not precluded from seeking care from providers or suppliers who do not participate in TEAM and a TEAM participant is prohibited from limiting beneficiaries to a preferred or recommended providers list that is not compliant with restrictions existing under current statutes and regulations. We will monitor beneficiary care, as discussed in section X.A.3.i of the preamble of this final rule, to ensure beneficiary freedom of choice is not compromised. Monitoring CMS's efforts will aim to ensure steering or other efforts to limit beneficiary access or move beneficiaries out of the model are not occurring. We also note the breadth of monitoring activities, which includes audits, CMS monitoring of utilization and outcomes within the model, and the availability of Quality Improvement Organization (QIOs) and 1-800-MEDICARE for reporting beneficiary concerns, that can help us identify any beneficiary access or freedom of choice concerns in TEAM.
We also note that target prices are set in the aggregate by episode category, so individual patients can still be cared for as best meets their needs. Furthermore, target prices will be risk-adjusted, as discussed in section X.A.3.d.(4), to account for beneficiary-level risk adjusters that help to mitigate costs that are out of the control of the provider. Finally, TEAM participants' reconciliation amounts will be adjusted by their CQS to drive quality of care improvements. Therefore, we disagree that TEAM will disincentivize high quality care.
Comment: A commenter stated that the discount rate is difficult to achieve for hospitals with a history of managing population health spending through participating in CMS ACOs due to diminishing returns.
Response: We disagree that hospitals with previous experience in CMS ACOs will be unable to meet the target price after the discount factor due to past reductions in episode spending. Target prices are set regionally; therefore, TEAM participants with previous experience in bundled payment models and value-based care may be better situated to achieve reduce spending if they were successful in other value-based care initiatives.
Comment: A commenter suggested that discount factors should be set by provider spending variation. Providers with low variation could be given a low discount factor, while providers with more variation could be given a slightly higher discount factor.
Response: We thank the commenter for their suggestion of different discount factors by variation in spending. We disagree with varying discount factors according to episode variation. We believe that regionally set target prices, normalization and trend factor adjustments, risk-adjustment, and social risk-adjustment will more directly address for differences in spending variation.
After consideration of the public comments we received, we are finalizing the discount factor provision with slight modification at § 512.540(c) by incorporating a discount factor of 1.5 percent for CABG and Major Bowel episode categories and a discount factor of 2 percent for LEJR, SHFFT, and Spinal Fusion episode categories .
In both CJR and BPCI Advanced, we recognized that hospitals that perform a number of episodes below a certain volume threshold would have insufficient volume to receive a target price based on their own baseline data. In the 2015 CJR Final Rule ( 80 FR 73285 ), we acknowledged that such hospitals might not find it in their financial interests to make systemic care redesigns or engage in an active way with the CJR model. At 80 FR 73292 , we acknowledged commenter concerns about low volume providers, including ( print page 69762) but not limited to, observations that low volume providers could be less proficient in taking care of LEJR patients in an efficient and cost-effective manner, more financially vulnerable with fewer resources to respond to the financial incentives of the model, and disproportionately impacted by high-cost outlier cases. Despite these potential challenges, we stated that the inclusion of low volume hospitals in CJR was consistent with the goal of evaluating the impact of bundled payment and care redesign across a broad spectrum of hospitals with varying levels of infrastructure, care redesign experience, market position, and other considerations and circumstances ( 80 FR 73292 ).
In CJR, we set the low volume threshold as fewer than 20 CJR episodes across the 3-year baseline years of 2012-2014. Low volume hospitals received target prices based on 100 percent regional data, rather than a blended target price that incorporated their participant-specific data, because a target price based on limited data is less likely to be accurate and reliable. These hospitals were also subject to the lower stop-loss limits that we offered to rural hospitals, in recognition of the fact that they might be less prepared to take on downside risk than hospitals with higher episode volume. In the CJR 2017 Final Rule that reduced the number of mandatory metropolitan statistical areas (MSAs), low volume hospitals were among the types of hospitals that were required to opt in if they wanted to remain in the model ( 82 FR 57072 ). In the 2020 Final Rule, we removed the remaining low volume hospitals from the CJR extension when we limited the CJR participant hospital definition to those hospitals that had been mandatory participants throughout the model ( 86 FR 23497 ).
In BPCI Advanced, our low volume threshold policy was to not provide a target price for a given clinical episode category if performed at a hospital that did not meet the 41 clinical episode minimum volume threshold during the 4-year baseline period. This meant that no BPCI Advanced episodes would be triggered for that particular clinical episode category during the applicable performance period at that hospital. However, participants could continue to trigger other clinical episode categories for which they had enrolled and for which there was sufficient baseline volume. Additionally, clinical episodes that occurred at the hospital during the performance period, though not triggering a BPCI Advanced episode, would count toward the low volume threshold when that year became part of the baseline. Therefore, as the baseline shifted forward each year, bringing a more recent year into the baseline and dropping the oldest year, a hospital could potentially meet the volume threshold and receive a target price for the clinical episode category for a subsequent performance period.
In TEAM, we stated in the proposed rule that there will be a low volume threshold for purposes of reconciliation. This low volume threshold would apply to total episodes across all episode categories in the baseline period for a given PY. If a TEAM Participant did not meet the proposed low volume threshold of at least 31 total episodes in the baseline period for PY 1, CMS would still reconcile their episodes, but the TEAM participant would be subject to the Track 1 stop-loss and stop-gain limits for PY 1. If a TEAM Participant did not meet the proposed low volume threshold of at least 31 total episodes in the applicable baseline periods for PYs 2-5, the TEAM Participant would be subject to the Track 2 stop-loss and stop-gain limits for PYs 2-5, as described in section X.A.3.d.(5)(h) of the preamble of this final rule.
We considered, but did not propose, including alternative approaches to a minimum episode volume threshold in TEAM, including an approach similar to BPCI Advanced, where if a TEAM participant did not meet the 31 episode minimum volume threshold for a given episode category in the 3-year baseline period, the TEAM participant would not be held accountable for that episode category for the performance year that aligned with the 3-year baseline period. We also considered different minimum volume thresholds in the baseline period, including 51, 21, and 11. However, we are concerned that imposing a minimum volume threshold that removes TEAM participant accountability may restrict the number of hospitals eligible to participate in TEAM and limit beneficiary access to the benefits of value-based, coordinated care. We also considered, but did not propose, implementing minimum episode volume thresholds during the performance year. Specifically, we considered, but did not propose, not holding TEAM participants accountable for a given episode category if they initiated less than 11 or 6 episodes in a given episode category or less than 31 or 21 total episodes across episode categories in a performance year. However, we are concerned that including minimum episode volume thresholds during the performance year may introduce program integrity issues where TEAM participants steer TEAM beneficiaries to other providers to be below the threshold and not be accountable for episodes in TEAM. We sought comment on whether TEAM should consider implementing the alternatives to the minimum volume thresholds for either the 3-year baseline period or the performance year.
We sought comment on our proposal at proposed § 512.550(e)(3) for setting and applying the low volume threshold at reconciliation.
The following is a summary of the public comments received on the low volume hospital proposal and our responses to those comments:
Comment: Many commenters stated that practices that do not meet the low volume threshold should not be included in model reconciliation for the episode categories that do not meet the low volume threshold. Many commenters expressed concern that the low volume threshold placing TEAM participants in Track 2 is insufficient to protect low volume providers.
Response: We thank commenters for their concern regarding the inclusion of low volume TEAM participants in Track 2 model participation. We will not be finalizing this low volume threshold and will propose alternatives in future notice and comment rulemaking prior to the model start date.
Comment: Many commenters stated that the low volume threshold was set too low and noted that the threshold for BPCI Advanced and CJR were 41 episodes per episode category for BPCI Advanced and typically an average of 10 episodes per baseline year. TEAM, by contrast, is set at 10 episodes per year for all episode types. Practices could meet the requirement by having most episodes in one episode type and a few in others. As a result, providers could be subject to episode variation. Many commenters stated that using a low volume threshold per episode category would avoid this issue. Some other commenters stated the BPCI Advanced threshold of 41 episodes by episode category, 50 episodes by episode category, or 100 episodes total should be used.
Response: We thank commenters for their concern regarding the low volume threshold. We understand that this low volume threshold could create difficulties for TEAM participants. We will not be finalizing this low volume threshold and will propose alternatives in future notice and comment rulemaking prior to the model start date.
Comment: Some commenters stated that TEAM participants should be placed in Track 1 of the model, or have downside risk waived, if they are classified as a low volume hospital. ( print page 69763)
Response: We thank the commenters for their concern regarding downside risk in TEAM for low volume hospitals. We will not be finalizing this low volume threshold and will propose alternatives in future notice and comment rulemaking prior to the model start date.
Comment: A couple of commenters stated that low volume and rural hospitals are poor candidates for bundled payment models such as TEAM. Low volume and rural hospitals have difficulties recruiting, training, and maintaining the staff and clinicians required for TEAM goals. Additionally, these hospitals have less experience in these types of models, and lack the robust networks required by them.
Response: We thank these commenters for their concern regarding the feasibility of including low volume and rural hospitals in TEAM. We disagree that rural hospitals are unable to meet the requirements of TEAM and believe rural hospitals can succeed in implementation. To ameliorate some issues, TEAM will allow rural hospitals, as defined in section X.A.3.f of the preamble of this final rule, the ability to participate in Track 1 for the first performance year with no downside risk and Track 2 for performance years 2 through 5 with a 5 percent stop-gain and stop-loss limit. Also, as indicated in section X.A.3.a.(1) of the preamble of this final rule, we are providing TEAM participants with approximately 17 months to prepare before the model start date. We believe this time period will allow TEAM participants, including rural hospitals, the opportunity to develop care redesign interventions and review baseline period data, pursuant to a request and a TEAM data sharing agreement, as discussed in section X.A.3.k of the preamble of this final rule. However, we agree our proposed low volume hospital policy may not be sufficient to protect low volume hospitals, including rural hospitals from unnecessary financial risk. Therefore, we will not be finalizing this low volume threshold and will propose alternatives in future notice and comment rulemaking prior to the model start date.
Comment: A couple of commenters stated that the proposed low volume threshold could result in outlier episodes skewing the results without statistical significance. A low volume threshold should account for natural variation.
Response: We thank the commenters for their concern regarding outliers and variation in low volume hospitals. We will not be finalizing this low volume threshold and will propose alternatives in future notice and comment rulemaking prior to the model start date.
Comment: A commenter stated that CMS should seek more feedback on TEAM burdens on low volume hospitals before future implementation and finalizing a rule.
Response: We thank the commenter for their concern regarding TEAM burdens on low volume hospitals. We will not be finalizing this low volume threshold and will propose alternatives in future notice and comment rulemaking prior to the model start date.
After consideration of the public comments we received, we will not be finalizing a policy on low volume hospitals. Accordingly, we are modifying regulatory text in section § 512.550 (e)(3) to remove references to a low volume threshold.
We stated in the proposed rule that CMS would provide preliminary target prices to TEAM participants prior to the start of each performance year. For instance, since the earliest episodes for a given performance year would end on January 1, and most of these episodes would have been initiated by an anchor hospitalization or anchor procedure that occurred near the end of November or the beginning of December of the previous calendar year, we proposed in the proposed rule to provide preliminary target prices to the TEAM participant by the end of November prior to each performance year. We stated in the proposed rule that preliminary target prices would be based on regional episode spending during the baseline period. TEAM participants would receive the preliminary target prices for each MS-DRG/HCPCS episode type that corresponded to their region. In the proposed rule, we proposed that these preliminary target prices would incorporate a prospective trend factor (as described in section X.A.3.d.(3)(f) of the preamble of this final rule) and a discount factor (as described in section X.A.3.d.(3)(g) of the preamble of this final rule), as well as a prospective normalization factor (as described in section X.A.3.d.(4) of the preamble of this final rule) that would be subject to limited adjustment at reconciliation (as described in section X.A.3.d.(5)(h) of the preamble of this final rule).
In the original CJR methodology, we first proposed that risk adjustment be limited to providing separate target prices for episodes initiated by MS-DRG 469 versus MS-DRG 470, because MS-DRGs under the Inpatient Prospective Payment System (IPPS) are designed to account for some of the clinical and resource variations that exist and that impact hospitals' costs of providing care ( 80 FR 73338 ). In response to comments requesting further risk adjustment, in the 2015 CJR Final Rule we finalized a policy to risk adjust target prices based on the presence of hip fractures in order to capture a significant amount of patient-driven episode expenditure variation ( 80 FR 73339 ). As a result, we provided four separate target prices to participant hospitals based on MS-DRG 469 versus MS-DRG 470, and presence versus absence of a primary hip fracture. The impact of hip fractures on inpatient costs associated with a hip replacement was subsequently acknowledged by CMS' decision to create two new MS-DRGs (521 and 522) for hip replacements in the presence of a primary hip fracture ( 85 FR 58432 ). We incorporated these new MS-DRGs into the CJR model episode definition as of October 1, 2020, via the November 2020 Interim Final Rule with Comment (IFC) ( 85 FR 71170 ).
In the 2021 CJR 3-Year extension Final Rule, we acknowledged the need for further risk adjustment to account for beneficiary-level factors that tend to impact spending in a way that is beyond the control of the provider. We introduced age bracket (less than 65 years, 65 to 74 years, 75 to 84 years, and 85 years or more), CJR Hierarchical Condition Category (HCC) count (zero, one, two, three, and four or more), and dual eligibility (receiving both full Medicare and Medicaid benefits) as beneficiary-level risk adjustment factors that would be applied to each episode at reconciliation. The definition of these risk adjustment variables, and our reasoning for incorporating them into the risk adjustment methodology, is described in detail at 86 FR 23523 .
The coefficients for the risk adjustment variables in the CJR extension were calculated prospectively, prior to the beginning of each performance year, using a linear regression model. As we stated at 86 FR 23524 , this regression model approach would allow us to estimate the impact of each risk adjustment variable on the episode cost of an average beneficiary, based on typical spending patterns for a nationwide sample of beneficiaries with a given number of CMS-HCC conditions, within a given age bracket, and with dual eligibility or non-dual eligibility status. We used an exponential model to account for the fact that CJR episode costs are not normally distributed. A detailed description of the regression model begins at 86 FR 23524 . ( print page 69764)
At reconciliation, after applying the high-cost episode cap to remove outliers, the risk adjustment coefficients for the three risk adjustment variables were applied to the episode-level target price based on the applicable episode region and MS-DRG. However, since age, CJR HCC count, and dual eligibility status are inherently included in the regional target price, since regions with beneficiaries who are older, more medically complex, and socioeconomically disadvantaged tend to have higher average episode costs, we applied a normalization factor to remove the overall impact of adjusting for age, CJR HCC count, and dual eligibility on the national average target price, as described at 86 FR 23527 .
By contrast, BPCI Advanced has used a more complex risk adjustment model that includes many more risk adjustment coefficients, including both patient and provider characteristics. Categories of patient characteristics include (but are not limited to): HCCs (individual flags, interactions, and counts), recent resource use, and demographics. Provider characteristics, which are used to group hospitals into peer groups, include bed size, rural vs. urban, safety net vs. non-safety net, and whether or not the participant is a major teaching hospital. (We note that the term “provider characteristics” and the related term “provider-level risk adjusters” in BPCI Advanced referred to characteristics of the hospital where the patient was hospitalized or received the procedure, regardless of whether the BPCI Advanced participant was a PGP or a hospital. For increased clarity, since hospitals will be the participants, we will use the terms “hospital characteristics” and “hospital-level risk adjusters” for these same risk adjusters in TEAM.). The first stage of the BPCI Advanced risk adjustment methodology uses a compound log-normal model in order to account for the substantial right skew of the distribution of episode costs. This means that it combines two log-normal distributions in order to capture costs associated with both low-cost episodes (which are the majority of episodes) and very high-cost episodes (which are fewer in number but exert a strong influence on spending averages). However, participants have found the risk adjustment model difficult to interpret, particularly since is it is not widely used in other research or healthcare models.
In an effort to simplify the risk adjustment methodology for TEAM and allow participants to more easily calculate an episode level estimated target price, we proposed in the proposed rule to base our methodology on the CJR extension methodology, with a few key differences. Rather than calculate one national set of risk adjusters across all MS-DRGs for a given episode category, we stated in the proposed rule we would calculate risk adjustment coefficients at the MS-DRG/HCPCS episode type level. We considered, but did not propose, calculating risk adjustment at the MS-DRG/HCPCS episode type/region level. However, we believed that, when further subdivided into regions, the low volume of episodes for certain MS-DRG/HCPCS episode types would be insufficient to create accurate and reliable risk adjustment multipliers.
In the proposed rule, we proposed to use the same age bracket risk adjustment variable (less than 65 years, 65 to less than 75 years, 75 to less than 85 years, and 85 years or more) that we use in the CJR extension, based on the participant's age on the first day of the episode, as determined through Medicare enrollment data. We also proposed in the proposed rule to use an HCC count risk adjustment variable, but we to calculate it differently than the CJR HCC count risk adjustment variable. For this risk adjustment variable, which we would call the TEAM HCC count, we stated in the proposed rule we would conduct a 90-day lookback for each beneficiary, beginning with the day prior to the anchor hospitalization or anchor procedure. We would use the beneficiary's Medicare FFS claims from that 90-day lookback period to determine which HCC flags the beneficiary is assigned and create a count of those HCC flags. This methodology would be consistent with BPCI Advanced and would represent a more uniform way of measuring clinical complexity across beneficiaries, as opposed to using the annual HCC file that is used in CJR. It would also reduce the incentive for increased coding intensity at the time of the initiating procedure.
In the proposed rule, we proposed to use an expanded risk adjustment variable that accounts for multiple potential markers of beneficiary social risk. Although it would function as a single, binary (yes = 1 or no = 0) variable in our risk adjustment model, the variable would represent the union of three different potential markers of beneficiary social risk. The first would be full Medicare/Medicaid dual eligibility status, which is currently used in both CJR and BPCI Advanced. Additionally, we would incorporate two additional elements to the beneficiary social risk adjustment variable. We stated in the proposed rule that beneficiaries would also be assigned the value of yes = 1 for the social risk adjustment variable if they either fall into a state or national Area Deprivation Index (ADI) percentile beyond a certain threshold, or if they qualify for the Medicare Part D Low Income Subsidy. The beneficiary would be assigned a value of yes = 1 on this single, binary social risk variable if one or more of these three indicators of social risk applied to the beneficiary. In the proposed rule, we proposed to use a threshold of the 80th percentile for the national ADI and the 8th decile for the state ADI. Across other CMS Innovation Center models, as well as peer reviewed publications, and we did not find a consensus on a specific threshold that is universally used. For example, the Making Care Primary Model uses 75th percentile for the national ADI and in existing literature, some papers use a continuous measure, and some use a 75 percent, an 80 percent, or 85 percent cut-off. [ 929 930 931 932 933 ] Therefore, we feel that an 80 percent threshold is comparable to other risk adjustment methodologies. We sought comment on whether there are different thresholds for national and state ADI that we should consider. Lastly, we proposed in the proposed rule to enforce sign restrictions to avoid negative coefficients for beneficiary social risk adjustment. In other words, the adjustment to the preliminary or reconciliation target prices would only happen if the coefficient on the beneficiary social risk adjustment variable is positive. We believed enforcing sign restrictions would more accurately reflect episode spending for underserved beneficiaries who may ( print page 69765) experience access and underutilization issues. The beneficiary social risk variable proposed in the proposed rule and our reasons for choosing each component are described in detail in section X.A.3.f of the preamble of this final rule.
While we proposed a limited set of risk adjusters that is closer in number to the CJR methodology for simplicity in the proposed rule, we considered, but did not propose, using the same set of risk adjusters in the BPCI Advanced model because we recognize that there may be particular episode categories or MS-DRGs that would benefit from additional clinical risk adjusters. For instance, in BPCI Advanced, just over half (53 percent) of coronary artery bypass graft (CABG) procedures have been performed electively, with the remainder performed emergently. Some clinicians have stated their belief that CABG episodes should be priced differently based on whether they are performed electively (that is, scheduled in advance) or emergently, even when they are assigned to the same MS-DRG. They stated their belief that non-emergent procedures are generally performed on relatively healthier beneficiaries, and providers may have greater control over outcomes. Conversely, they stated that episodes following an emergency room visit on the same day or the day before an episode tend to involve sicker patients, leading to greater clinical variability and less predictable episode spending. We therefore requested comment on whether TEAM should use the BPCI Advanced episode-specific risk adjuster or if there are other potential episode-specific or MS-DRG-specific clinical risk adjusters, and how those clinical risk adjusters should be defined based on information available on the IPPS claim associated with the episode trigger.
We also considered, but did not propose, including peer group or hospital-specific risk adjusters in TEAM. Similar to the BPCI Advanced model, peer group adjusters would be based off of hospital characteristics, including hospital size (for example, number of hospital beds), safety net hospital status, location (for example, core-based statistical area (CBSA) urban and rural indicators and census division), and if the hospital was a major teaching hospital determined by looking at the intern to bed ratio in the provider specific files. [ 934 ] We recognized including this level of risk adjustment may improve pricing accuracy for hospitals, but it introduces an additional layer of complexity to the risk adjustment model that could be challenging for TEAM participants understand when factoring in the existing risk adjustment variable and other pricing components. Since TEAM is a mandatory model, and it may capture more hospitals that have not previously participated in an episode-based payment model, we wanted to create a pricing methodology that all TEAM participants, regardless of experience or resource, can understand. We sought comment on whether target prices in TEAM should include risk adjustment variables based on hospital characteristics.
Another key difference between our proposal and the current CJR risk adjustment methodology is that we proposed in the proposed rule to provide a prospective normalization factor with preliminary target prices. We stated in the proposed rule that the prospective normalization factor would be subject to a limited adjustment at reconciliation based on the observed case mix, up to ±5 percent. This would allow participants to better estimate their target prices, as it would incorporate the normalization factor prospectively, rather than only introducing the normalization factor at reconciliation. We believed that this approach strikes a balance between predictability and protecting TEAM participants and CMS from significant shifts in patient case mix between the final baseline year and the performance year.
A goal of TEAM's risk adjustment approach is to balance simplicity with accuracy to ensure our pricing methodology reflects episode spending those accounts for provider spending trends by region and MS-DRG as well as accounting for beneficiary acuity. The risk adjustment approach in our proposed rule relies on capturing data from Medicare claims or other sources of information that do not include patient functional assessment data. Evidence suggests that risk adjustment models may be improved when taking into account patient functional status. [ 935 ] We recognized there are existing data sets that capture patient functional status information. Specifically, the Improving Medicare Post-Acute Care Transformation Act of 2014 (the IMPACT Act) requires the reporting of standardized patient assessment data with regard to quality measures and standardized patient assessment data elements. The standardized patient assessment elements include functional status and are collected and reported by Long-Term Care Hospitals (LTCHs), Skilled Nursing Facilities (SNFs), Home Health Agencies (HHAs) and Inpatient Rehabilitation Facilities (IRFs). Since an episode encompasses post-acute care spend, the standardized patient assessment data could be incorporated into TEAM's risk adjustment methodology. However, we recognized inclusion of such data may increase the risk adjustment methodology complexity and make it challenging for TEAM participants to understand how it affects their preliminary or reconciliation target price. Therefore, we sought comment on the utility of including standardized patient assessment data in TEAM's risk adjustment methodology or whether there is other functional status data we should consider and whether standardized patient assessment data or other functional status data should be included in TEAM's risk adjustment methodology in future performance years.
To summarize, for TEAM we proposed in the proposed rule a risk adjustment methodology based on the CJR extension methodology, but with key differences that we believed would maximize target price predictability and transparency. As in CJR, this methodology would use baseline data to calculate risk adjustment multipliers and hold them constant at reconciliation. Participants would be provided with these risk adjustment multipliers prior to the start of the Performance Year and would be able to use them to estimate their episode-level target prices. Unlike in CJR, these risk adjustment multipliers would be calculated at the MS-DRG level, resulting in a separate set of risk adjustment multipliers for each MS-DRG episode type. We also proposed in the proposed rule to incorporate a prospective normalization factor into preliminary target prices, which would be subject to a limited adjustment at reconciliation. We sought comment on our proposals at proposed § 512.545(a-d) for risk adjusting episodes.
The following is a summary of the comments we received related to the proposed risk adjustment and normalization and our responses to these comments:
Comment: A commenter suggested to use regional risk adjustment for each of the episode types. For regions and episode types where the episode volume ( print page 69766) is not sufficient, the commenter suggested to use a national coefficient.
Response: We thank the commenter for their suggestion. As we pointed out in the proposed rule ( 89 FR 35934 ) and the preamble of this final rule, we are concerned that for most risk adjusters, we would not have sufficient episode volume to create accurate and reliable risk adjustment multipliers at the MS-DRG/HCPCS episode type/region level. Keeping the risk adjustment methodology as simple and consistent as possible is a primary focus in TEAM and using regional coefficients for some MS-DRG/HCPCS episode types/regions but a national coefficient for other MS-DRG/HCPCS episode types/regions goes against this goal.
Comment: A commenter suggested including elective episodes only, citing concerns that providers who have a higher proportion of urgent, emergent, and trauma admissions will be disadvantaged under the current proposal.
Response: We thank the commenter for their input on including elective episodes only. As stated in the proposed rule ( 89 FR 35934 ) and preamble of this final rule, 53 percent of CABG procedures in BPCI Advanced were elective procedures, with the remainder performed emergently. We are concerned that removing nearly half of CABG episodes would drop the episode volume substantially and therefore negatively impact the reach of the model. Exclusions are typically reserved for services or procedures that are expensive, but also rare.
Comment: A couple commenters recommended that CMS make considerations for the utilization of critical access hospital (CAH) swing beds. Commenters cited that CAH swing bed daily allowed claims can be ten-folder greater than traditional SNF daily allowed claims. If CAH swing bed claims are not adjusted for in some way, either in target prices or reconciliation calculations, target prices cannot be met.
Response: We thank the commenters for sharing their concerns regarding CAH swing bed claims. In response to the comments, we analyzed how frequently CAH swing bed claims are included in the 30-day post-discharge episode spending and what proportion of spending in the post-discharge period CAH swing bed claims make up. The episodes were constructed using Medicare FFS claims from 2022 and the first two quarters of 2023.
Our analysis showed that CAH swing bed stays are a low frequency event in episodes with 30-day post-discharge lengths. In 4 of the 5 episode categories proposed for TEAM, among episodes with at least one SNF claim (traditional or CAH swing bed), approximately 4 percent had at least one CAH swing bed claim. In the episode category CABG, approximately 7 percent of episodes had a CAH swing bed claim. Since CAHs swing beds are exempt from the SNF Prospective Payment System (PPS), they are reimbursed at a higher rate. However, these claims will not make up a large proportion of post-discharge spending, or episode spending as a whole in TEAM episodes. Our analysis showed that in 4 of the 5 episode categories, post-discharge spending in the SNF setting among episodes with at least one CAH swing bed claim only accounted for 11 percent to 12 percent of total post-discharge SNF spending among all episodes. In CABG episodes, the proportion was 19 percent of total post-discharge SNF spending.
We are concerned that applying adjustments for CAH swing bed claims will create unintended consequences for utilization of these CAH swing bed services. We believe that the high reimbursement rates for these CAH swing bed claims could encourage providers to seek out relationships with and to increase utilization of traditional SNFs. TEAM participants that have historically utilized CAH swing beds will be in a position to earn significant savings by establishing relationships with traditional SNFs and discharging patients they would otherwise move to CAH swing beds to traditional SNFs.
Comment: A few commenters expressed their support for the risk adjustment of target prices and appreciated the goal of maintaining simplicity of the methodology. A few commenters also supported the calculation of the risk adjustment coefficients at the MS-DRG/HCPCS episode type level. However, many commenters expressed support for the expansion of the TEAM risk adjustment methodology to better account for the clinical complexity of patients and resource use across hospitals. Commenters noted that HCC variables, in addition to the HCC count variable, should be included in order to accurately capture a hospital's patient complexity. Commenters expressed concern that a lack of proper risk adjustment would penalize hospitals which treat the sickest, most complex patients. A few commenters noted that additional risk adjusters are necessary to account for patients who previously lived in nursing homes since these beneficiaries may return to nursing homes and increase post-episode spending. Additional commenters asked CMS to consider including variables for the severity of illness and risk of mortality. A few commenters also asked CMS to include risk adjusters based on patient assessment data, such as the patient's functional status, since this can affect the type and amount of post-acute care that the patient receives. A couple commenters expressed concern that the risk adjustment methodology only incorporates HCC count variables, which assumes that the impact on costs for all HCCs are the same. A commenter noted that this would create an incentive for providers to favor the treatment of patients who have multiple mild chronic conditions and avoid patients with one or two conditions severe enough to increase the risk of poor surgical outcomes. A commenter also requested CMS to include disability as a risk adjustment variable. A commenter suggested that CMS should include demographic factors, clinical factors, and procedure-specific factors into the risk adjustment methodology to allow for appropriate clinical decisions for care teams and patients. A commenter expressed support for age variables to be included in the risk adjustment methodology. A commenter asked CMS to include a risk adjustment variable to capture the complexity of patients treated at academic medical centers. Some commenters requested the risk adjustment model to include additional risk adjusters for all HCCs and other beneficiary-level variables similar to the BPCI Advanced model.
Response: Given the numerous concerns from stakeholders regarding the proposed TEAM risk adjustment methodology, we recognized an updated methodology may be necessary to strengthen the risk adjustment model. As indicated in the proposed rule ( 89 FR 35934 ) and discussed in the preamble of this final rule, we considered using the BPCI Advanced model's risk adjusters, but we opted to not propose them and constructed TEAM's risk adjustment methodology similar to the CJR model to avoid adding complexity. We recognize that there may be a better balance in including more risk adjusters to increase target pricing accuracy while still limiting complexity. In order to expand the number of variables included in the risk adjustment model, we conducted a Lasso regression analysis using episodes built with Medicare FFS claims from CY 2019—2021 and received additional input from a Technical Expert Panel (TEP) of clinicians. The Lasso regression identified risk adjusters that minimize the residual sum of squares between the observed spending values and predicted spending values. The Lasso regression ( print page 69767) included an exhaustive list of beneficiary-level and hospital-level risk variables from the BPCI Advanced model, including HCC variables, interactions between multiple conditions or comorbidities, and hospital characteristics among others. The TEP conducted literature reviews, reviewed the results of the Lasso regression, and leveraged their own expertise to recommend a number of additional beneficiary-level risk variables per episode category. Based on the Lasso analysis and clinician input, we curated a list of additional variables for testing inclusive of the risk adjustment variables from the proposed rule. This updated list of risk adjustment variables, which contains no more than 25 risk adjustment variables per episode category, intends to maintain our goal of a simplified risk adjustment methodology, while including a more robust set of risk adjustment variables in order to capture spending accurately.
We compared the fit of the proposed TEAM regression model first including only the risk adjustment variables from the proposed rule (hereby referred to as Model 1) and then including the curated list of risk adjustment variables (hereby referred to as Model 2).
The accuracy of the predicted spending, relative to observed spending, of Model 2 was tested against Model 1. The analysis showed that Model 2 was more accurate in predicting spending compared to the observed spending at both the episode-level and hospital-level. In addition, goodness-of-fit statistics were produced to test model fit. The R-squared, adjusted R-squared, and coefficient of variation statistics showed that Model 2 has better model fit compared to Model 1.
Based on the Lasso regression, clinician input, spending analysis, and model fit analysis, and the commenters concerns about needing a more robust risk adjustment methodology, we are finalizing an updated list of risk adjustment variables to include in the TEAM risk adjustment methodology as follows:
For CABG episodes, the following 17 risk adjustment variables are now included: age bracket variable, HCC count variable, prior post-acute care use variable, beneficiary social risk variable, hospital bed size variable (which is based on four categories: 250 beds or fewer, 251—500 beds, 501—850 beds, and 850 beds or more), safety net hospital status variable, and the following 11 HCCs:
- HCC 18: Diabetes with Chronic Complications
- HCC 46: Severe Hematological Disorders
- HCC 58: Major Depressive, Bipolar, and Paranoid Disorders
- HCC 84: Cardio-Respiratory Failure and Shock
- HCC 85: Congestive Heart Failure
- HCC 86: Acute Myocardial Infarction
- HCC 96: Specified Heart Arrhythmias
- HCC 103: Hemiplegia/Hemiparesis
- HCC 111: Chronic Obstructive Pulmonary Disease
- HCC 112: Fibrosis of Lung and Other Chronic Lung Disorders
- HCC 134: Dialysis Status
For Surgical Hip/Femur Fracture Treatment (SHFFT) episodes, the following 21 risk adjustment variables are now included: age bracket variable, HCC count variable, beneficiary social risk variable, hospital bed size variable, safety net hospital status variable, and the following 16 HCCs:
- HCC 22: Morbid Obesity
- HCC 82: Respirator Dependence/Tracheostomy Status
- HCC 83: Respiratory Arrest
- HCC 157: Pressure Ulcer of Skin with Necrosis Through to Muscle, Tendon, or Bone
- HCC 158: Pressure Ulcer of Skin with Full Thickness Skin Loss
- HCC 161: Chronic Ulcer of Skin, Except Pressure
- HCC 170: Hip Fracture/Dislocation
For Major Bowel Procedure episodes, the following 18 risk adjustment variables are now included: age bracket variable, HCC count variable, beneficiary social risk variable, long-term institutional care use variable, hospital bed size variable, safety net hospital status variable, and the following 12 HCCs:
- HCC 11: Colorectal, Bladder, and Other Cancers
- HCC 21: Protein-Calorie Malnutrition
- HCC 33: Intestinal Obstruction/Perforation
- HCC 188: Artificial Openings for Feeding or Elimination
For LEJR episodes, the following 21 risk adjustment variables are now included: age bracket variable, HCC count variable, procedure-related variable (ankle procedure or reattachment, partial hip procedure, partial knee arthroplasty, total hip arthroplasty or hip resurfacing procedure, and total knee arthroplasty), variable for disability as the original reason for Medicare enrollment, dementia without complications variable, beneficiary social risk variable, prior post-acute care use variable, hospital bed size variable, safety net hospital status variable, and the following 12 HCCs:
- HCC 8: Metastatic Cancer and Acute Leukemia
- HCC 78: Parkinson's and Huntington's Diseases
For Spinal fusion episodes, the following 18 risk adjustment variables are now included in the updated TEAM risk adjustment methodology: age bracket variable, HCC count variable, prior post-acute care use variable, beneficiary social risk variable, hospital bed size variable, safety net hospital status variable, and the following 12 HCCs:
- HCC 40: Rheumatoid Arthritis and Inflammatory Connective Tissue Disease
HCC 111: Chronic Obstructive Pulmonary Disease ( print page 69768)
We thank the commenters for sharing their support and concerns regarding the TEAM risk adjustment methodology. We agree with the commenters that a more robust risk adjustment methodology is necessary for TEAM. We refer readers to the description above, which lists the additional beneficiary-level variables per episode category that will be included in the TEAM risk adjustment methodology to accurately capture the complexity of the patient case mix. Coefficient estimates for the risk adjustment variables will only be included in the regression if they are present in at least 21 episodes during the 3-year baseline period. This threshold will ensure that coefficient estimates are produced based on a reasonable sample size of episodes.
The updated risk adjustment methodology incorporates HCC variables, in addition to the HCC count variable, in order to create more accurate episode spending predictions that are based on the clinical complexity of the patient case mix and additional resource use. The updated risk adjustment methodology also includes a prior post-acute care variable to account for patients who have visited a post-acute care facility during the lookback period for Lower Extremity Joint Replacement (LEJR), CABG, and Spinal Fusion. These facilities include LTCH, SNF, HH, and IRF.
We acknowledge the comments regarding additional variables to account for severity of illness and risk of mortality. We also acknowledge the comments to include variables based on patient assessment data, including the functional status and disability of patients, into the risk adjustment methodology. The updated risk adjustment methodology incorporates disability as the original reason for Medicare enrollment for LEJR episodes. CMS will consider additional analyses to assess the appropriateness of the remaining variables and their impact on episode costs.
We thank the commenter for suggesting CMS incorporate variables for demographic factors, clinical factors, and procedure-specific factors. The updated risk adjustment methodology includes age brackets as a demographic variable. CMS is not considering any other demographic factors. The risk adjustment for LEJR episodes now includes five procedure-related variables to better account for spending specific to each type of procedure.
We acknowledge the comment regarding additional risk adjustment variables for patient treated at academic medical centers. As part of the Lasso regression analysis, CMS found that patients treated at major teaching hospitals did not have a statistically significant difference in episode spending for any of the episode categories.
We also acknowledge the comments requesting CMS consider an even larger risk adjustment model, similar to BPCI Advanced. While our updated risk adjustment model is predicated from the BPCI Advanced model and selects the variables that demonstrated the most promising findings, we still want to maintain the goal of a simplified risk adjustment methodology which still captures differences in episode spending based on patient complexity and resource use. The updated risk adjustment methodology achieves this goal without incorporating the full risk adjustment variables used in BPCI Advanced.
However, we will continue to review additional beneficiary-level risk adjustment variables based on commenters suggestions to better assess their impact on episode spending and if warranted, will propose additional risk adjustment variables in future notice and comment rulemaking.
Comment: Some commenters suggested that CMS include hospital-level characteristics in the TEAM risk adjustment methodology. Specifically, commenters asked CMS to consider safety net status, rural/urban location, size, and teaching status. A commenter noted that any additional risk adjusters should not result in lower target prices for rural or safety net hospitals. Particularly, safety-net hospitals may have higher episode payments than non-safety-net hospitals for certain conditions and may find difficulty reaching regional spending targets. A couple commenters noted that the hospital-level risk adjustment methodology should be more sophisticated, similar to the CJR and BPCI Advanced models.
Response: We agree that additional hospital-level variables are necessary for the TEAM risk adjustment model in order to accurately capture differences among hospitals. The updated hospital-level TEAM risk adjustment model will include variables for bed size and safety-net status for all episode categories. The hospital-level variables were identified from ones used in the BPCI Advanced model and were selected based on the Lasso regression analysis. The variable for bed size is based on four categories: 250 beds or fewer, 251—500 beds, 501—850 beds, and 850 beds or more. We believe that these modifications to the TEAM risk adjustment methodology will sufficiently capture the additional patient complexity and resource use across the various types of hospitals in TEAM. In addition, safety net hospitals, as defined in section X.A.3.f of the preamble of this final rule, will also have the option to remain in Track 1 for performance years 1 through 3, as discussed in section X.A.3.a.(3) of the preamble of this final rule, which is limited to only upside financial risk and includes a 10 percent stop-gain limit.
We acknowledge the comments regarding the inclusion of hospital-level risk adjustment variables for rural/urban status as well as teaching hospital status, as they were used in the BPCI Advanced model. As part of the Lasso regression analysis, which identified risk adjustment variables that minimize the residual sum of squares between the observed spending values and predicted spending values, we found that the variables for patients treated at rural hospitals and teaching hospitals were not selected by the Lasso model for any of the episode categories. Therefore, risk adjustment variables for rural status and teaching hospital status will not be included. However, rural hospitals, as defined in section X.A.3.f of the preamble of this final rule, will have additional flexibilities in TEAM, such as opting to participate in Track 2 of the model which has lower levels of risk and reward with a 5 percent stop-loss/stop-gain limit. We acknowledge the comments asking for a risk adjustment model which is more similar to CJR and BPCI Advanced models. The updated risk adjustment methodology we are finalizing, as described earlier, is a balance between the two models with the inclusion of the additional risk adjusters, while maintaining the goal of a simple risk adjustment model.
Comment: Some commenters urged CMS to ensure that the risk adjustment model accounts for the differences in episode costs between emergent and elective procedures. A couple commenters noted that considering whether an operation is scheduled/elective vs. non-scheduled/urgent can dramatically alter the expected cost. A couple commenters cited that there is a meaningful clinical difference that drives patient complexity and the need for more intensive care patterns. A couple commenters noted there is a high degree of variability and clinical complexity of cases, even within MS-DRGs, for emergent versus elective cases.
Response: We acknowledge that emergent procedures can be relatively more expensive than elective ( print page 69769) procedures, given that emergent patients are likely to be more clinically complex. In light of the comments, we will expand our proposed risk adjustment model by adding more clinically relevant risk adjustment variables. We believe this can account for the pricing differences between emergent and elective procedures, such as by adding HCCs for Cardio-Respiratory Failure and Shock, Congestive Heart Failure, Acute Myocardial Infarction, and Specified Heart Arrhythmias for the episode category CABG. We refer the readers our discussion earlier for the comprehensive list of additional risk adjustment variables, including individual HCCs, that will be included and finalized in TEAM. We appreciate the commenter's suggestion on extending the lookback period or including HCCs on the claims incurred during the anchoring hospital encounter. However, as stated in the proposed rule, we believe that using Medicare FFS claims from the lookback period, as opposed to the anchoring claim, is beneficial since it will reduce the incentive for increased coding intensity at the time of the initiating procedure.
Comment: Some commenters requested CMS create a separate target price for episodes initiated on an emergent basis. A commenter believed CMS's approach to calculate target prices does not adequately reflect many of the variables that go into the care of patients on a case-by-case basis. Another commenter encouraged CMS to refine the target price methodology to avoid performance disadvantage for centers where the most urgent, emergent care is provided. A commenter suggested segmenting episode types by the presence of a trauma diagnosis code, fracture diagnosis code, or an inpatient charge with an ER related revenue code.
Response: We appreciate the commenters' suggestions on the target price methodology for episodes initiated on an emergent basis. We believe that grouping emergent and elective procedures together, rather than stratifying them by an indicator or a separate target price, reduces the incentive for increasing coding intensity. We believe that the expansion of the proposed risk adjustment model, which will include additional clinical risk adjustment variables, should be sufficient in accounting for pricing differences and clinical complexities among emergent procedures. Thus, we are not finalizing any policy specific to stratifying emergent procedures. However, in light of comments received, we will consider additional adjustments for emergent procedures in future notice and comment rulemaking.
Comment: A commenter took issue with capturing HCCs documented within 90-days prior to the anchor hospitalization or procedure, citing this may not accurately capture the clinical complexity of patients, especially if a procedure is non-elective. The commenter cited that utilizing HCC count and not each HCC's unique weight will dilute the accuracy of risk adjustment, and suggested CMS utilize the standard Medicare HCC risk adjustment model. Another commenter expressed support for patient level clinical risk adjustment for pre-existing conditions, but requested the annual HCC file is used, similar to what the CJR model uses.
Response: We thank the commenters for their input on the risk adjustment model and suggestions to use the CMS-HCC risk adjustment model and annual HCC file. We acknowledge the commenter's concern on only capturing HCCs documented within 90-days prior to the anchor hospitalization or procedure.
We disagree that the standard Medicare HCC risk adjustment model should be utilized. The HCC model is not designed to predict costs within TEAM episodes, and thus may not accurately predict TEAM episode spending. The CMS-HCC risk adjustment model's intended use is to pay for Medicare Advantage (MA) plans appropriately. On the other hand, the TEAM risk adjustment model is tailored for specific episode categories in TEAM. For example, there will be adjusters for specific procedure groups in LEJR for total knee arthroplasty, partial knee arthroplasty, total hip arthroplasty/hip resurfacing procedure, partial hip procedure, and ankle procedures/reattachments). The CMS-HCC risk adjustment model is used to predict total Medicare expenditures in an upcoming year, which may not be appropriate for use when predicting expenditures in shorter time periods, such as 30-day episodes in TEAM. It is more accurate to develop specific lookback periods based on an episode's start date, as opposed to using the CMS-HCC risk adjustment model calculations, which are applicable to a calendar year.
However, we do agree that utilizing only HCC count may dilute the accuracy of risk adjustment. We believe we can improve upon the proposed approach by expanding the risk adjustment model to include curated HCCs for each episode category. The expanded risk adjustment model should account for cost differences between elective and non-elective procedures. We refer the readers to our earlier discussion in this section for the comprehensive list of risk adjustment variables, including individual HCCs per episode category, that will be included and finalized in TEAM.
We acknowledge the commenter's suggestion to use the annual HCC file, similar to CJR. As stated in the proposed rule, we proposed to use the beneficiary's Medicare FFS claims from the lookback period to determine which HCC flags the beneficiary is assigned. Using a lookback period represents a more uniform way of measuring clinical complexity across beneficiaries, as opposed to using the claims from the initiating procedure like the annual HCC file does. Using the lookback period reduces the incentive for increased coding intensity at the time of the initiating procedure. However, similar to CJR, we proposed to use baseline data to calculate risk adjustment multipliers and hold them constant at reconciliation. TEAM participants will be provided these risk adjustment multipliers prior to the start of the performance year and would be able to use them to estimate their episode-level target prices, similar to the annual HCC file provided to CJR participants.
Comment: Many commenters were concerned that the 90-day lookback period to determine the inclusion of beneficiary-level variables in risk adjustment was too short of a time period to accurately capture comorbidities and complications that are clinically relevant to the episode. A commenter noted that a majority of HCCs are documented during primary care provider visits. Given that Medicare beneficiaries are recommended to visit their primary care provider once a year, a 90-day lookback period may not capture HCCs which are clinically relevant to the episode if the primary care provider visit did not occur within the 90-day lookback period. Commenters suggested CMS to consider extending the lookback period to 180 days, 1 year, or 36 months. Commenters also suggested that the HCC variables captured from the anchor hospitalization should be included in the risk adjustment.
Response: We thank the commenters for sharing their concerns regarding the 90-day HCC lookback period. We did not consider a longer lookback period and therefore cannot finalize a longer period. However, we will review the data to determine whether a 90-day, 180-day, or 1-year HCC lookback period will accurately capture comorbidities and complications that are clinically relevant to the episode. We will not consider a 36-month lookback period as ( print page 69770) HCCs flagged as far back as 36 months from the start of an episode are unlikely to be clinically relevant and will additionally increase the level of administrative burden. In addition, we will not be including any risk adjustment variables that are captured during the anchor hospitalization because of the likelihood for increased coding intensity. We believe that variables captured prior to the anchor hospitalization provide the best predictive information regarding episode costs. The TEAM beneficiary-level risk adjustment methodology will continue to only include variables that are flagged prior to the anchor hospitalization or anchor procedure.
Comment: A commenter suggested that the complexity of referral patients living outside of a hospital's CBSA may not be accurately accounted for by HCC codes. They cited concerns related to factors outside of their control, including these patients seeking care from primary care providers outside of their health care system. They are concerned that the current benchmarking model is not accurately accounting for the increased complexity of these patients.
Response: We thank the commenter for sharing their concerns regarding the HCC codes of patients from outside a TEAM participant's CBSA area. We will consider analyzing whether patients residing outside of a TEAM participant's CBSA are more costly to treat for a given hospital and if it is appropriate to add new risk adjustment variables to improve pricing accuracy in such scenarios. If such is the case, then we would propose updates in future notice and comment rulemaking.
Comment: CMS received a comment that expressed concerns about coding intensity increasing over time and recommended limiting the settings in which HCCs are collected for the risk adjusters to hospital inpatient stays, hospital outpatient visits, and visits with clinicians. The commenter also urged that CMS remove codes generated from health risk assessments (including annual wellness visits) from the TEAM HCC count to ensure that diagnosis codes contribute to the risk score only if they are related to actual health care services received. This comment also touched on the possibility of increased coding intensity among model participants relative to an external population.
Response: We thank the commenter for sharing their thoughts. We agree that “coding creep” can be a concern in a model where payments are risk adjusted. Similarly, increased coding intensity among model participants relative to non-participants can be a concern. The performance year update to the normalization factor will reverse any coding creep that occurs nationally between the baseline and the performance year, though we have applied a 5 percent cap to the normalization update to provide model participants with more stability in their pricing estimates. Using a lookback period, rather than including diagnoses from the episode initiating admission/procedure will minimize the opportunities for participants to change coding intensity among their patients, relative to non-participants. We also note that when capturing HCCs during the lookback period, using the most updated version of HCC model may increase accuracy in terms of predicting resource needs and we will strive to incorporate the most recent version that can be used for our baseline period and performance years. We also want to clarify the language on page 36434 of the proposed rule regarding the settings from which the HCCs will be constructed, we intend to use only inpatient, outpatient, and carrier claims, as is currently done in the BPCI Advanced model. This is similar to the settings the commenter suggested with the exception that we do not intend to limit the carrier claims used to exclude non-clinician claims and claims from health risk assessments. We are concerned that removing diagnosis codes from non-clinicians and health risk assessments could result in important diagnoses being missed.
Comment: Many commenters expressed concern that outpatient procedures are included in the same episode categories as inpatient hospitalizations when setting target prices and recommended adjusting for inpatient and outpatient originating episodes in the risk adjustment models. The commenters pointed out that these cases can vary significantly in terms of complexity, resources required, care pathways, and recommended post-discharge treatment. Specifically, the commenter noted that safety-net hospitals that serve populations with health-related social needs will more likely have procedures performed on an inpatient basis and may be disadvantaged by the blended pricing structure. Commenters also noted CMS's proposal could result in financial incentives to shift care from the inpatient to the outpatient setting.
Response: We acknowledge the commenters concerns regarding the inclusion of outpatient procedures and inpatient hospitalizations in the same episode category. We continue to believe blended pricing methodology is more appropriate since it reduces any risks for beneficiaries to be inappropriately shifted from the inpatient to the outpatient setting. We agree that patient case-mix can vary between the inpatient and outpatient procedures and also recognize a blended pricing structure could create pressure for clinicians to recommend the lower cost outpatient setting to minimize total episode costs. However, we believe that our risk adjustment methodology will incentivize clinicians to continue performing LEJR and Spinal Fusion procedures in the appropriate clinical setting based on their assessment of each patients' complexity, particularly since performing these procedures on sicker patients in the outpatient setting could increase the risk of post-acute complications and lead to higher overall episode spending. We understand the concerns related with proposed risk adjustment methodology and as mentioned in earlier in this section of the final rule, we are finalizing the risk adjustment methodology to include additional beneficiary-level variables as well as some hospital-level variables. We believe these modifications will further address differences in patient characteristics as well as variation in spending between outpatient and inpatient cases with MCCs.
Comment: A few commenters suggested that CMS adjust for cases of fracture and non-fracture in the risk adjustment model due to the high degree of variability in the clinical complexity and recommended post-discharge treatment between these cases.
Response: We thank the commenters for their suggestions but disagree that a fracture flag is necessary in the LEJR and SHFFT episode types. LEJR comprises of MS-DRGs, 469, 470, 521, and 522 in the inpatient setting. MS-DRGs 521 and 522 specifically account for hip replacement with a principal diagnosis of hip fracture. Prior analyses in the BPCI Advanced model have shown that knee joint replacements with fractures account for a very small proportion of episodes within MS-DRGs 469 and 470. Ankle replacements, which make up a very small volume of LEJR episodes, are mostly performed in the outpatient setting. Similarly, the proposed SHFFT episode category, which contains MS-DRGs 480, 481, and 482, will primarily include beneficiaries who receive a hip fixation procedure in the presence of a hip fracture, other than hip arthroplasty. Since the MS-DRGs in the episode categories inherently account for fractures, and the risk-adjustment regression is run at the MS-DRG level, we do not think additional ( print page 69771) fracture risk adjusters are necessary in TEAM.
Comment: A commenter requested the following risk adjusters to be included for inpatient coronary artery bypass graft episodes: ejection fraction less than 30 percent, malnutrition, obesity, lung disease, chronic kidney disease, and congestive heart failure. The commenter also suggested CMS to include risk adjusters for intraoperative findings and exclude redo procedures.
Response: We thank the commenter for the additional suggestions regarding the risk adjustment methodology for inpatient coronary artery bypass graft episodes. As explained before, HCC 85 (congestive heart failure) and HCC 112 (fibrosis of lung and other chronic lung disorders) are now included as risk adjusters for inpatient coronary artery bypass graft episodes, in addition to nine other HCC flags. Although malnutrition, obesity, and chronic kidney disease have implications on clinical outcomes, CMS has concerns about the over-reporting for these conditions and do not plan to include these HCC flags in the risk adjustment methodology for inpatient coronary artery bypass graft episodes.
While an ejection fraction less than 30 percent certainly has an effect on clinical outcomes, there is no method to identify this scenario using claims data. We welcome suggestions for possible surrogate risk adjusters for an ejection fraction less than 30 percent which can be captured in claims data in future rulemaking.
The TEAM risk adjustment methodology is limited to beneficiary- and provider-level variables during the lookback period. Risk adjusters for intraoperative findings may be subject to variations among providers based on clinical expertise, and increased coding intensity if included.
Inpatient coronary artery bypass graft redo procedures have decreased over time and make up a small percentage of all inpatient coronary artery bypass graft episodes. [ 936 ] Redo procedures are also likely to indicate the quality of care provided during the initial procedure, which providers should be held accountable for as long as the redo procedure is conducted during the 30-day post-discharge period of the initial procedure.
Comment: A few commenters recommended that CMS make considerations for the utilization of IRF. A commenter noted that the costs for patients who typically received inpatient rehabilitation can be higher due to intensive therapy and care. Commenters cited concerns that TEAM target price would not account for the complexity of such patients and would impede the use of IRF care even when it is the best option for them. Commenters urged for updates to the risk adjustment to ensure that patient complexity is appropriately accounted for as well as retroactive adjustments to the target prices when IRF care is utilized such that providers do not incur negative financial impact when beneficiaries must use IRF.
Response: We thank the commenters for sharing their concerns regarding IRF utilization. We will consider assessing whether patients receiving IRF care have higher episode costs and if it is appropriate to add new risk adjusters to improve pricing accuracy for such cases. If such is the case, then we would make proposals in future notice and comment rulemaking. We also refer readers to our discussion earlier in this section which details the changes to the risk adjustment model including the addition of several beneficiary-level risk adjustment variables that will adjust the target prices to reflect the complexity of patients demonstrated in the lookback period.
Comment: A commenter expressed concerns regarding the proposed risk adjustment model that fails to account for differences in Medicare Advantage penetration in participating communities. Specifically, the commenter noted such communities can have very few beneficiaries enrolled in traditional Medicare leading to few episodes in TEAM, making it difficult to make investments for delivering care. Another concern they have noted is that healthier beneficiaries are more likely to be enrolled in Medicare Advantage Plan. Thus, less healthy beneficiaries enrolled in a traditional Medicare plan who are more likely to experience complications in the post discharge period would be left in TEAM and not adjusting for it in the model can incur penalties for such providers.
Response: We acknowledge the concerns shared regarding the differences in the beneficiary enrollment and characteristics between the Medicare Advantage and traditional Medicare plans in certain geographical areas and appreciate the recommendations made. We understand the concern that having fewer episodes in TEAM may not make a sufficient enough incentive for hospitals to make the investments needed to deliver care in different ways and hospitals may not find it in their financial interest to make systemic care redesigns or engage in the model in an active way. As noted in section X.A.3.d.(3)(h) of this rule, we are not finalizing the proposed low volume threshold policy and intend to propose a new policy in future notice and comment rulemaking. We also agree that healthier beneficiaries are more likely to enroll in Medicare Advantage plans than the traditional Medicare plan. We believe the changes we are finalizing for the risk adjustment, as discussed earlier in this section of the final rule—to add specific beneficiary level risk adjustment variables that are relevant to the episode types—will incentivize hospitals to perform these procedures on sicker patients, decrease the risk of post-acute complications, and lead to higher savings. Moreover, we also believe our regional target prices and additional hospital-level variables in the risk adjustment will help account for high/low FFS beneficiaries' penetration in specific regions.
Comment: A couple of commenters requested that CMS share all variables used in risk adjustment and target price creation to allow for participants to conduct their own assessments, predictions, and validations.
Response: We thank the commenters for their recommendation to share risk adjustment variables with TEAM participants. We recognize the importance of transparency and predictability in target pricing. We note that as discussed earlier in this section of the final rule, we stated in the proposed rule that participants would be provided with the risk adjustment multipliers prior to the start of the performance year and would be able to use them to estimate their episode-level target prices.
Comment: Some commenters expressed that the risk adjustment model should account for additional patient-level social risk indicators such as housing instability, food insecurity, financial needs, transportation problems, education, language, interpersonal safety, and homelessness. They also shared that the social risk adjustment model should go beyond a binary yes/no variable as the safety net status, specifically dual-eligibility status, of beneficiaries may not always be identified prior to the episode occurring.
A commenter also noted that the risk adjustment mechanism relies on prior episodic payment models that did not fully adjust for the additional costs of caring for safety net populations. Additionally, a commenter remarked that that dual eligibility is an imperfect proxy of social need and vulnerability and requested CMS to investigate ( print page 69772) potential new indicators for assessing individual-level health-related social needs (HRSN) correlated with negative health outcomes. Another commenter noted that hospitals in their state are concerned about potential disparities among providers, specifically those that primarily serve economically distressed counties where social determinants of health and socioeconomic barriers pose significant challenges to implementation.
A couple commenters did express support for the addition of adjustment for social risk by including dual eligibility status, LIS status, and state and national ADI as this would accurately capture the social risk faced by patients and the associated resource use for these patient populations.
Response: We thank the commenters who have expressed support for the social risk adjustment as well as those that have expressed concerns regarding it.
In the proposed rule, we noted that the social risk adjustment variable was chosen so that it can account for multiple potential markers of beneficiary social risk. Using Medicare/Medicaid dual eligibility status, LIS status, and living in areas in the top percentiles of either the national or state level ADI allows CMS to utilize existing indicators of social risk together and capture safety-net populations through multiple means. If dual-eligibility status has not been identified prior to the episode occurring, the ADI marker may still be able to identify the beneficiary at a higher social risk. Additionally, we proposed in the proposed rule to enforce sign restrictions to avoid negative coefficients for the beneficiary social risk adjuster, that is, the adjustment to the preliminary or reconciliation target prices would only happen if the coefficient on the beneficiary social risk adjustment variable is positive. We would not be able to enforce the sign restrictions if additional variables for social risk were separately added to the model.
As discussed earlier in this section of the final rule, we are also finalizing the addition of safety-net status of the hospital as a risk-adjustment variable to all episode types to address concerns about providers that primarily care for beneficiaries with dual-eligibility or LIS status. We believe the inclusion of the hospital's safety-net status will strengthen the risk adjustment model and appropriately set target prices for providers that serve economically distressed counties. While CMS is not including all of the risk adjustment variables tested in BPCI Advanced to maintain the simplicity required for hospitals without experience in value-based care to participate in TEAM, the updated risk adjustment model does take more patient-level and hospital level factors into account.
We acknowledge that dual eligibility may not be a perfect proxy of social need and vulnerability. As CMS mandates the collection of health-related social needs (HRSN) data from Medicare provider and suppliers more widely and strengthens the availability of HRSN data, we will consider if there is sufficient and high-quality data available in future baseline years for TEAM to utilize such alternative indicators for risk adjustment.
Comment: A couple of commenters expressed concerns that the application of the normalization factor to the target prices negates the application of risk adjustment to the model. In particular, a commenter expressed concern that the application of the normalization factor can be problematic for providers with low acuity patient mix compared to the nation and can disincentivize care improvement. Another commenter noted that CMS should consider removing the normalization factor entirely or consider applying a full prospective normalization factor in the subsequent year. A few commenters suggested limiting to only prospective normalization factor and not renormalizing during reconciliation. Similarly, another commenter expressed concern that risk adjustment process outlined in the proposed rule is not sufficient and the proposed renormalization policy will likely cancel out the risk adjustment. The commenter further noted that CMS should propose caps for normalization factor that would not offset the risk adjustment. Another commenter suggested CMS incorporate the normalization retrospectively. One other commenter suggested that CMS should considering capping the normalization factor for a given region.
Response: We appreciate the comments on the calculation of the normalization factor. We recognize the concerns expressed by commenters regarding the application of the normalization factor. However, we continue to believe that the application of normalization factor should not cancel or nullify the beneficiary level risk adjustment. At a high level, the purpose of the normalization factor is to prevent the double counting of the patient case-mix that would happen because of the application of the risk adjustment to regional benchmark prices. To elaborate further, the benchmark prices, which are calculated as average observed costs (after capping at the 99th percentile) for each region and MS-DRG combination, have the beneficiary level risk adjusters inherently included in the benchmark prices, as such DRG-regions with more complex patients like older beneficiaries or those with high HCC counts, will tend to have higher benchmark prices. If these high benchmark prices are further multiplied with the risk score multipliers without the application of the normalization factor, the effect of patient severity will be double counted leading to inaccurate target prices. We understand the concern related with capping the retrospective normalization factor. However, we believe the application of the normalization factor with limited adjustment at reconciliation will protect both CMS and TEAM participants from significant shifts in patient case-mix nationally between the final baseline year and performance year.
We appreciate the suggestions for capping the normalization factor at the regional level. However, since the normalization factor is calculated at the MS-DRG level, it is statistically more appropriate to cap it at the same level. We will continue to assess our target price methodology, including the application of the normalization factor, and may propose modifications if we believe they are necessary. We agree that the risk adjustment process outlined in the proposed rule ( 89 FR 35934 ) of using HCC counts, age bracket and social risk as risk adjustment variables are not sufficient. As we discussed earlier in this of the final rule, we are finalizing changes to risk adjustment process with additional beneficiary-level and hospital-level risk variables.
Comment: A few commenters suggested that the normalization factor should be capped so that the normalization factor does not have a greater impact than the risk adjustment itself and target prices remain stable and predictable.
Response: We refer readers to section X.A.3.d.(5)(h) of the preamble of the final rule where we proposed that a cap will be applied to the final normalization factor at Reconciliation, such that the final normalization factor would not exceed ±5 percent of the prospective normalization factor.
Comment: A commenter stated that CMS should broaden its scope to consider non-medical factors of care in risk adjustment, such as hospitals expanding insurance coverage or providing financial assistance programs for vulnerable patient populations.
Response: We thank the commenter for their concern regarding non-medical ( print page 69773) factors of care and barriers to care in the risk adjustment process. TEAM risk adjusts for several factors such as dual eligibility, age, HCC counts, and a social risk adjustment that includes patient population eligibility for Low-Income Subsidies in Medicare. We believe this social risk adjustment will adequately account for non-medical factors of risk-adjustment. We acknowledge that some hospitals (small, rural, those serving underserved beneficiaries, etc.) may find it harder to meet target prices and compete against other hospitals in their region. We expect the finalized risk adjustment methodology, which will adjust target prices to account for additional beneficiary-level and hospital-level variables, as discussed earlier in this section of the final rule, to mitigate some of these concerns. Additionally, we acknowledge the challenges faced by safety-net hospitals and will provide flexibilities to avoid downside risk and allow them to remain in Track 1 for performance years 1 through 3 and eligible to participate in Track 2 in performance years 4 through 5 with a lower stop-loss/stop-gain limit, as discussed in section X.A.3.a.(3) of the preamble of this final rule. Lastly, we will take into consideration the recommendations with regard to expanding coverage or providing financial assistance programs to underserved beneficiaries.
After consideration of the comments received, we are finalizing at § 512.545(a) our proposal to calculate risk adjustment coefficients at the MS-DRG/HCPCS episode type level. We are finalizing with modifications our proposed risk adjustment methodology to include two hospital level variables, hospital bed size and safety net hospital, to all episode categories at § 512.545(a). We are also finalizing with modification our proposed risk adjustment methodology to include additional beneficiary level variables at § 512.545(a)(6) that are episode category specific. Specifically, in addition to the five risk adjustment variables applicable to all episode categories, we are finalizing the addition of 12 beneficiary level risk adjustment variables for the CABG episode category, 16 beneficiary level risk adjustment variables for the LEJR episode category, 13 beneficiary level risk adjustment variables for the Major Bowel Procedure episode category, 16 beneficiary level risk adjustment variables for the SHFFT episode category, 13 beneficiary level risk adjustment variables for the Spinal Fusion episode category at § 512.545(a)(1). We are also finalizing with modification at § 512.545(d) that at the time of reconciliation, the preliminary target prices are risk adjusted using all the beneficiary level and provider level variables. We also are finalizing at § 512.540(b)(6) the proposal to calculate the normalization factor as the MS-DRG mean benchmark price for episodes during the baseline period divided by MS-DRG mean risk adjusted benchmark price for episodes during the baseline period and include this value prospectively when determining preliminary target prices. We are also finalizing at § 512.545(e)(1)(ii) the proposal that the prospective normalization factor would be subject to a limited adjustment at reconciliation based on the observed case mix, up to ±5 percent. We are finalizing at § 512.545(a)(1) our proposal to use a lookback period to determine which HCC flags the beneficiary is assigned. However, we are not yet finalizing the length of the lookback period due to concerns raised by commenters. We intend to propose and finalize a specific length for the lookback period through notice and comment rulemaking within the next year, so that participants will know the lookback period prior to the start of the model.
This section outlines our proposals on how we intend to reconcile performance year spending for a TEAM participant's beneficiaries in episodes against the reconciliation target price in order to determine if CMS owes the TEAM participant a reconciliation payment, or if the TEAM participant owes CMS a repayment (for all Track 3 participants and beginning in performance year 2 for Track 2 hospitals). In the proposed rule, we proposed to adjust the reconciliation amount for quality based on the TEAM participant's Composite Quality Score (CQS), which would be constructed from their quality measure performance, to calculate the quality-adjusted reconciliation amount. Stop-loss/stop-gain limits would be applied to the quality-adjusted reconciliation amount to determine the TEAM participant's Net Payment Reconciliation Amount (NPRA). Finally, we would adjust the NPRA for post-episode spending, when applicable, to determine the reconciliation payment or repayment amount.
We refer readers to section X.A.3.b.(5) of the preamble of this final rule for our definition of related services for our episodes, to section X.A.3.a.(1) of the preamble of this final rule for our definition of performance years, and to section X.A.3.d.(3) of the preamble of this final rule for our approach to establish preliminary target prices.
At proposed § 512.550 we stated in the proposed rule to conduct an annual reconciliation calculation that would compare performance year spending on episodes that ended during that PY with reconciliation target prices for those episodes to calculate a reconciliation amount for each TEAM participant. We would reconcile, on an annual basis, all episodes attributed to a TEAM participant that end in a given calendar year during the model performance period. This would be consistent with CJR and numerous other CMS value-based payment programs. We believed that one annual reconciliation accommodates the need for regular performance feedback while minimizing the administrative burden of more frequent reconciliations. Therefore, we proposed in the proposed rule to align the TEAM reconciliation approach with reconciliation in CJR, and to reconcile episodes based on performance years. We sought comment on this proposal to conduct one reconciliation for each performance year. We invited public comment regarding the proposal to conduct one reconciliation for each performance year. We received no comments on the proposal; therefore, we are finalizing these provisions without modification at § 512.550.
In the proposed rule, we proposed to conduct the annual reconciliation of each TEAM participant's actual episode payments against the target price(s) 6 months after the end of the performance year. This policy would be consistent with the 6 months of claims runout we allow for the CJR reconciliation for PYs 6-8. We believed that 6 months is sufficient time for claims runout given that an internal review of Medicare claims data found that 98.71 percent of inpatient (IP) claims had been received, and 89.96 percent were considered final, by 6 months after the date of service. [ 937 ] For hospital outpatient department (HOPD) claims, those rates were 98.10 percent and 95.78 percent, respectively. Similar rates were found for all other types of claims, including Carrier, skilled nursing facility (SNF), home health (HH), and durable medical equipment (DME), indicating that we would have a nearly complete picture of performance year spending by 6 months ( print page 69774) after the end of the performance year. For TEAM, we proposed in the proposed rule to capture claims submitted by July 1st following the end of the performance year and carry out the NPRA calculation as described previously to make a reconciliation payment or hold TEAM participants responsible for repayment, as applicable, in quarters 3 or 4 of that calendar year. We sought comment on our proposal at proposed § 512.550(b) to perform reconciliation 6 months after the end of the performance year.
The following is a summary of the public comments received on the timing of reconciliation proposal and our responses to those comments:
Comment: A few commenters supported CMS's proposal to offer a single reconciliation with at least six months of claims runout and encouraged CMS to release the reconciliation data on a consistent basis. A commenter also asked if CMS could consider other ways to shorten the data lag by providing provisional reconciliation results to some accountable care organization (ACO) participants.
Response: We thank the commenters for their support and plan to release beneficiary-identifiable claims data on a monthly basis, pursuant to a request and TEAM data sharing agreement, as described in section X.A.3.k of the preamble of this final rule. We would be unable to provide provisional reconciliation results since the claims data would not be available in time to produce those results, however, we will consider ways of providing more updated performance and trend data throughout the performance year to help TEAM participants gauge their performance in the model before reconciliation.
After consideration of the comments received, we are finalizing at § 512.550(b) the proposal to perform reconciliation 6 months after the end of the performance year.
We recognize that there may be TEAM participants that experience a reorganization event during a given performance year. At proposed § 512.505, we proposed in the proposed rule to define a reorganization event as a merger, consolidation, spin off or other restructuring that results in a new hospital entity under a given CMS Certification Number (CCN). As a result of such an event, the TEAM participant may begin billing under a different CCN, or an additional entity could be incorporated into the TEAM participant's existing CCN, resulting in a new hospital entity. For instance, TEAM participant A may merge with, or be purchased by, TEAM participant B and begin billing under TEAM participant B's CCN. In this case, we stated in the proposed rule we would perform separate reconciliation calculations for TEAM participant A and TEAM participant B for those episodes where the anchor hospitalization admission or the anchor procedure occurred before the effective date of the merger or purchase. In the proposed rule, we proposed to reconcile episodes where the anchor hospitalization admission or the anchor procedure occurred on or after the effective date of the merger or purchase under the new or surviving CCN that applies to the blended entity. We proposed this policy in recognition that the blended entity may have different spending patterns, or a different overall patient case mix, than the two separate entities prior to the merger. In a different instance, if a TEAM participant merges into or is purchased by a non-TEAM participant and begins billing under the CCN on the non-TEAM participant, we stated in the proposed rule we would reconcile episodes for the TEAM participant where the anchor hospitalization admission or the anchor procedure occurred before the effective date of the merger or purchase. This policy would allow for the TEAM participant to earn a reconciliation payment or owe a repayment for the episodes that occurred during the portion of the performance year that they were in the model. However, once the TEAM participant begins to bill under the non-TEAM participant's CCN, the blended entity would not be considered a TEAM participant, and we would not reconcile episodes where the anchor hospitalization admission or the anchor procedure occurred on or after the effective date of the merger or purchase under the new or surviving CCN that applies to the blended entity. We sought comment on our proposal at proposed § 512.550(b)(2) for conducting reconciliations for TEAM participants that experience a reorganization event during a given performance year.
We invited public comment regarding the proposal to conduct reconciliations for TEAM participants that experience a reorganization event during a given performance year. We received no comments on the proposal; therefore, we are finalizing these provisions without modification at § 512.550(b)(2).
As discussed in section X.A.3.d.(4) of the preamble of this final rule, we proposed in the proposed rule to apply beneficiary-level risk adjustment and a limited adjustment to the prospective normalization factor, as applicable, to increase the accuracy of our reconciliation calculations. At the time of reconciliation, we would apply these adjustments, if applicable, to the preliminary target prices we calculated and communicated to TEAM participants prior to the applicable performance year, as described in section X.A.3.d.(3)(i) of the preamble of this final rule. We noted that in some cases, the final target price applied to an episode in a given performance year at reconciliation would not change. In addition, in some cases the reconciliation target price would increase from the preliminary target price provided prior to the performance year, potentially benefitting TEAM participants. For instance, if the prospective normalization factor were calculated as 0.85, but the beneficiary case mix during the performance year differed from the case mix during the final year of the baseline such that the final normalization factor was calculated as 0.89, the reconciliation target price would incorporate the final normalization factor and therefore be higher than the preliminary target price.
Incorporating quality performance into the model payment structure is an essential component of TEAM, just as it is for the CJR model ( 80 FR 73370 ) and BPCI Advanced. Section X.A.3.c of the preamble of this final rule discusses the specific measures for which we proposed that TEAM participants would be held accountable. In addition to Quality Payment Program requirements to tie quality performance to payment for Advanced APMs, we believe it is important for TEAM to link the opportunity to earn a reconciliation payment with performance on quality measures to place greater emphasis on beneficiary quality of care and patient-centered care.
As discussed in section X.A.3.d.(5)(g) of the preamble of this final rule, which outlines the proposed process for incorporating quality into the reconciliation calculation, for each TEAM participant, we proposed in the proposed rule to calculate the difference between the TEAM participant's performance year spending and their reconciliation target price at ( print page 69775) reconciliation, identified as the reconciliation amount. In the proposed rule, we proposed that the reconciliation amount would then be adjusted based on the TEAM participant's quality performance. We proposed to use the quality measures discussed in section X.A.3.c of the preamble of this final rule to calculate a CQS in a similar manner to what we have implemented for many CMS models and initiatives, including CJR and BPCI Advanced. The CQS methodology would allow performance on each required TEAM quality measure to be meaningfully valued in the TEAM pay-for-performance methodology, incentivizing and rewarding cost savings in relation to the quality of episode care provided by the TEAM participant.
For TEAM, the actual level of quality performance achieved would be the most important factor in calculating the CQS to reward those TEAM participants furnishing high quality care to TEAM beneficiaries. Like the CJR model, TEAM would include a wide range of participants with varying levels of experience with value-based care and different current levels of quality performance. Other CMS programs also capture a wide range of participants and include quality performance methodologies that may directly affect the participant's financial performance. We noted that the Shared Savings Program utilizes similar features as the proposed TEAM CQS methodology, such as benchmarking quality performance, calculating scores for each measure and constructing an overall score (see 42 CFR 425.502 ). Additionally, the Hospital VBP Program and the HAC Reduction Program also utilize similar scoring methodologies, which apply weights to various measures and assign overall scores to hospitals ( 42 CFR 412.165 and 42 CFR 412.172 ). Despite the small number of quality measures proposed in the proposed rule for TEAM, the measures represent both clinical outcomes and patient experience, and each would carry substantial value in the TEAM CQS.
Although performance on each measure would be valued in the TEAM CQS methodology as proposed in the proposed rule, it is the TEAM participant's overall quality performance that would be considered in the pay-for-performance approach, rather than performance on each quality measure individually determining the financial opportunity under TEAM. The TEAM CQS methodology would also provide a framework for incorporating additional measures of meaningful outcomes for episodes in the future. The TEAM CQS methodology would provide the potential for financial reward for TEAM participants that reach an overall acceptable quality performance, thus incentivizing their continued efforts to improve the quality and efficiency of episodes. We sought comment on our proposal to use a CQS in the pay-for-performance methodologies of TEAM.
The CQS is one component of the reconciliation process and we proposed that it would be calculated based on the TEAM participant's performance on the quality measures proposed for the model. One of the primary purposes of the CQS is to create a comparative assessment for performance across episode categories and TEAM participants. Since not all quality measures apply to all episode categories, quality measures that apply to more episode categories will be volume-weighted more heavily in the CQS.
As indicated in section X.A.3.c.(3) of the preamble of this final rule, the TEAM quality measures proposed in the proposed rule would be collected from the CMS Hospital Inpatient Quality Reporting (IQR) Program and the Hospital-Acquired Condition (HAC) Reduction Program. The TEAM quality measures collected from the Hospital IQR Program and HAC Reduction Program would have raw quality measure scores, however, these raw quality measure scores may be in different measurement units making it difficult to make comparisons. Therefore, raw quality measure scores must be manipulated in order to produce a CQS. We proposed, similar to the BPCI Advanced model, for each performance year for each quality measure to convert the raw quality measure scores into scaled quality measure scores by comparing the raw quality measure scores to the distribution of raw quality measure score percentiles among the national cohort of hospitals, which would consist of TEAM participants and hospitals not participating in TEAM, in the CQS baseline period, so that each measure has a scaled quality measure score between 0 and 100 for each episode category. For example, if a TEAM participant's raw quality measure score of 71 percent in performance year (PY) 1 is equivalent to the 60th percentile during the CQS baseline period, their scaled quality measure score for that measure would be 60 in the performance year. We recognized there may be instances where the raw quality score may fall between percentiles or may be higher or lower than the raw quality scores in the CQS baseline period. Therefore, we proposed that if the raw quality measure score could belong to either of two percentiles in the CQS baseline period, then we would assign the higher percentile. Further we proposed to assign a scaled score of 100 if the TEAM participant has a raw quality measure score greater than the maximum of the raw quality measure scores in the CQS baseline period and assign a scaled quality measure score of zero if the TEAM participant has a raw quality score less than the minimum of the raw scores in the CQS baseline period. Lastly, we proposed not to assign a scaled quality measure score if the TEAM participant had no raw quality measure score.
In the proposed rule, we proposed the CQS baseline period to be CY 2025 for the duration of TEAM. We believed using CY 2025 as the CQS baseline period was similar to other CMS Innovation Center models, including the BPCI Advanced model, where the baseline period was established before the incentives of the model were in place in order to assess quality improvement. We considered, but did not propose, using a contemporaneous CQS baseline period, where the CQS baseline period would be the same as the performance year for each performance year, but we believed that may increase CQS calculation complexity and may create challenges for TEAM participants to implement meaningful quality improvement efforts. Lastly, we also considered, but did not propose, a rolling CQS baseline period, where the CQS baseline period would move forward by one year each performance year, but similarly to a contemporaneous CQS baseline period, we believed the simplicity of having a fixed CQS baseline period would be easier for TEAM participants to understand the CQS calculation methodology. However, as indicated in section X.A.3.b.(1) of the preamble of this final rule, we recognized the potential for additional episodes added to TEAM in future performance years, which may result in different quality measures being used in the CQS calculation. If new episodes categories or quality measures are introduced to TEAM, we would reassess the CQS baseline period and implement any changes in future notice and comment rulemaking.
Prior to calculating the CQS, we stated in the proposed rule that we would volume weight the quality measures based on the volume of episodes for a TEAM participant. ( print page 69776) Specifically, a normalized weight would be calculated by dividing the TEAM participant's volume of episodes for a given quality measure by the total volume of all the TEAM participant's episodes. This calculation would be applied to all quality measures for the TEAM participant (see Table X.A.-11). We believed it was important to volume weight the quality measures so that more weight is given to the quality measures that apply to more episode categories.

We would then take the quality measures' normalized weights and combine them with the scaled quality measure scores to determine the weighted scaled score. Specifically, we proposed in the proposed rule to calculate a weighted average by multiplying each quality measure's scaled quality measure score by its normalized weight to create weighted scaled scores for a TEAM participant. The weighted scaled scores would then be added together to construct the CQS for the TEAM participant (see Table X.A.-12).

As stated in the proposed rule, although the required set of quality measures proposed for TEAM are ones currently being reported through the Hospital IQR Program and HAC Reduction Program, we recognize that CMS may, in future regulations, remove current measures or require different measures for hospitals to report in the Hospital IQR Program and HAC Reduction Program. Therefore, CMS may propose changes to the TEAM measures and the methodology for constructing the composite quality score through future notice and comment rulemaking. We sought comment on our proposed methodology to calculate the TEAM composite quality score.
The following is a summary of the public comments received on the proposed CQS methodology and our responses to these comments.
Comment: A couple of commenters requested that CMS must provide monthly reporting in a form in which the participant can see the composite components and replicate the quality score due to the mandatory nature of the model. Specifically, a commenter requested CMS report each part of the CQS as part of an improved monthly reporting package.
Response: We thank commenters for sharing their suggestions on data sharing processes. We will take this suggestion into consideration as the model develops and will share more information on how data will be distributed in the future.
Comment: Some commenters expressed concern over the proposed CQS methodology. Specifically, commenters mentioned that CMS should reconsider setting a score of 100 to achieve full quality credit in reconciliation, ensuring the CQS payment adjustment work the same for hospitals receiving a positive or negative reconciliation payment. In other words, regardless of the costs of care, a higher quality score should be financially advantageous to hospitals. If hospitals would otherwise receive a positive payment adjustment, their adjustment should be increased by the amount of their quality score. A commenter suggested to balance quality and cost as part of value-based care, quality measures should be given an equal weighting to success as cost savings.
Response: We appreciate the concerns from commenters about the proposed CQS methodology and the need to achieve a perfect score to receive their full reconciliation payment amount. However, we believe the CQS methodology spurs TEAM participants to improve quality performance given the disincentive to perform poorly on quality. To that end, TEAM's CQS methodology also tries to protect TEAM participants from increased financial risk based on their CQS. Meaning, if a TEAM participant has a negative reconciliation amount, performing better on quality would reduce their repayment amount. For example, a TEAM participant that performs well in quality ( e.g., a CQS of 100) would have a lower repayment amount, as compared to a TEAM participant that did poorly on quality ( e.g., score of 0) and would owe their full repayment amount. We ( print page 69777) also note that TEAM's CQS methodology is similar to prior successful examples of episode-based payment models such as BPCI Advanced. Additionally, our methodology has similar features to how the Hospital VBP Program and the HAC Reduction Program apply their scoring methods, which applies weights to various measures and assigns an overall score to a hospital. However, we would like to clarify the calculation of specific quality measures that are considered inverse measures, where a lower raw quality measure score is considered favorable.
Lastly, CMS will take commenters' considerations into account and any updates to our CQS methodology will be proposed in future notice and comment rulemaking.
Comment: A few commenters expressed concern over the weighting of the CQS methodology due to the hospital wide measures holding more weight than the proposed total hip arthroplasty (THA)/total knee arthroplasty (TKA) Patient-Reported Outcome-Based Performance Measure (PRO-PM) given that this measure is specific to just the lower extremity joint replacement (LEJR) episode. Commenters mentioned that hospitals that do not have significant LEJR volume will in effect be held accountable to only the two hospital-wide measures. Commenters suggested that the weighting for the THA/TKA PRO-PM be increased.
Response: We would like to thank commenters for their suggestions. However, we decline to make any changes to the proposed CQS methodology. We believe that it is important to volume weight the quality measures so that more weight is given to the quality measures that apply to more episode categories. Specifically, we proposed to calculate a weighted average by multiplying each quality measure's scaled quality measure score by its normalized weight to create weighted scaled scores for a TEAM participant. Those weighted scaled scores would then be added together to construct the CQS for the TEAM participant.
Comment: A commenter mentioned being in full support of CMS' decision to use the quality adjustment methodology used in BPCI Advanced.
Response: We appreciate this commenter's support.
Comment: A couple of commenters mention being appreciative of CMS' proposal to calculate the quality scores based on measures that are already reported for other purposes. A commenter believes this is a great pathway to reduce burden related to quality reporting. However, commenters highlight some of the measures are in their first year of reporting and including a measure in a mandatory bundle with little data on how it will perform. Additionally, a commenter requested additional clarification on how other independent variables are being considered in terms of impacting the CQS baseline period or the performance relative to the baseline.
Response: We appreciate the concern and feedback from commenters regarding the CQS baseline period and the potential for unfamiliar measures being incorporated into the TEAM CQS in a later performance year. We will continue with the proposed CQS baseline period to be CY 2025 for the Hybrid Hospital-Wide All-Cause Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356) measure, CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135) measure, and the Hospital-Level Total Hip and/or Total Knee Arthroplasty (THA/TKA) Patient-Reported Outcome-Based Performance Measure (PRO-PM) (CMIT ID #1618) measure, as described in section X.A.3.c of the preamble of this final rule. We believe using a fixed CQS baseline period of CY 2025 is similar to other CMS Innovation Center models, including the BPCI Advanced model, where the baseline period was established before the incentives of the model were in place to assess the impact of the model.
We will assess TEAM participant performance on these measures and if warranted, will make policy updates in future notice and comment rulemaking.
Comment: Some commenters suggested that CMS adjust quality assessments to allow for high quality scores to reduce the discount factor and make other adjustments to reflect more meaningful quality evaluations in the model. A few commenters requested TEAM's CQS be designed like the CJR model where an excellent quality score reduces the discount factor down to zero. A commenter recommended including a quality improvement aspect into the CQS like the CJR model.
Response: We appreciate this commenter sharing their suggestion. However, CMS will continue to move forward with the proposed CQS methodology that was shared in this year's rule. As previously indicated, we do not think that reducing the discount factor based on quality performance is a sustainable, long-term policy. We recognize that eventually episode spending will level off and we cannot assume spending reductions will occur in perpetuity. Therefore, it may be reasonable to assume that a discount factor may not be needed when this scenario occurs, and CMS would want to incentivize TEAM participants to maintain spending rather reduce spending, especially to avoid compromising quality of care. We note that we do not believe TEAM participants, or Medicare Inpatient Prospective Payment System (IPPS) hospitals at large, have met that threshold yet, but given it may occur, we do not believe reducing the discount factor based on quality performance would be a policy to continue testing in TEAM. We also acknowledge the request for adjusting the CQS based on a TEAM participant's quality improvement on one or more of their quality measures. We will consider adding including quality improvement points to the CQS and we will continue to assess potential changes that may encourage greater quality of care improvement in TEAM. Any adjustments or updates to our proposed methodology will be shared in a future notice or comment rulemaking.
After consideration of the public comments we received, we are finalizing our proposals for the proposed composite quality score methodology with slight modification to the CQS baseline period. We acknowledge that by finalizing the Hospital Harm—Falls with Injury (CMIT ID #1518) measure, the Hospital Harm—Postoperative Respiratory Failure (CMIT ID #1788) measure, and the Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) measure for inclusion in TEAM starting in PY 2, a CQS baseline period of CY 2025 would be inappropriate given hospitals are not required to report on these measures for the Hospital IQR Program until 2026. A CQS baseline period of CY 2026 would be more appropriate for these measures. Therefore, we are finalizing our proposal with slight modification at § 512.547 to use a CQS baseline period of CY 2025 for the Hybrid Hospital-Wide All-Cause Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356) measure, CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135) measure, and the Hospital-Level Total Hip and/or Total Knee Arthroplasty (THA/TKA) Patient-Reported Outcome-Based Performance Measure (PRO-PM) (CMIT ID #1618) and a CQS baseline period of CY 2026 for the Hospital Harm—Falls with Injury (CMIT ID #1518) measure, the Hospital Harm— ( print page 69778) Postoperative Respiratory Failure (CMIT ID #1788) measure, and the Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) measure.
We are also modifying our scaled score methodology proposal for inverse quality measures, specifically the Hybrid Hospital-Wide All-Cause Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356) measure, the CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135) measure, the Hospital Harm—Falls with Injury (CMIT ID #1518) measure, the Hospital Harm—Postoperative Respiratory Failure (CMIT ID #1788) measure, and the Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134). We recognize our proposal in the proposed rule to assign a scaled score of 100 if the TEAM participant has a raw quality measure score greater than the maximum of the raw quality measure scores in the CQS baseline period and assign a scaled quality measure score of zero if the TEAM participant has a raw quality score less than the minimum of the raw scores in the CQS baseline period, would inadvertently penalize quality measure scoring for inverse quality measures. Therefore, we are modifying our proposal at § 512.547(b)(i)(C) slightly so that for the Hybrid Hospital-Wide All-Cause Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356) measure, the CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135) measure, the Hospital Harm—Falls with Injury (CMIT ID #1518) measure, the Hospital Harm—Postoperative Respiratory Failure (CMIT ID #1788) measure, and the Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) measure, we will assign a scaled score of 0 if the TEAM participant has a raw quality measure score greater than the maximum of the raw quality measure scores in the CQS baseline period and assign a scaled quality measure score of 100 if the TEAM participant has a raw quality score less than the minimum of the raw scores in the CQS baseline period. This slight modification acknowledges the difference between quality measures where a higher raw quality measure score is favorable and quality measures where a lower raw quality measure score is favorable.
After the completion of a performance year, we proposed in the proposed rule to retrospectively calculate a TEAM participant's actual episode performance based on the episode definition. We noted that episode spending would be subject to proration for services that extend beyond the episode (as described in section X.A.3.d.(3)(c) of the preamble of this final rule). In the proposed rule, we proposed to cap performance year spending at the high-cost outlier cap as described in section X.A.3.d.(3)(e) of the preamble of this final rule, and to apply the high-cost outlier cap to episodes in the performance year similarly to how we proposed to apply it to baseline episodes, using the 99th percentile for each MS-DRG/HCPCS episode type and region as the maximum. Any performance year episode spending amount above the high-cost outlier cap would be set to the amount of the high-cost outlier cap. We then proposed in the proposed rule to compare each TEAM participant's performance year spending to its reconciliation target prices. Specifically, we stated in the proposed rule we would define the reconciliation amount as the dollar amount representing the difference between the reconciliation target price and performance year spending, prior to adjustments for quality, stop-gain/stop-loss limits, and post-episode spending. We noted that, as discussed in section X.A.3.d.(3) of the preamble of this final rule, a TEAM participant would have multiple target prices for episodes ending in a given performance year, based on the MS-DRG/HCPCS episode type and the performance year when the episode was initiated. In the proposed rule, we proposed to determine the applicable reconciliation target price for each episode using the aforementioned criteria and calculate the difference between each TEAM participant's performance year spending and its aggregated reconciliation target price for all episodes in the performance year, resulting in the reconciliation amount. Specifically, we stated in the proposed rule we would define the reconciliation amount as the dollar amount representing the difference between the reconciliation target price and performance year spending, prior to adjustments for quality, stop-gain/stop-loss limits, and post-episode spending. Next, we would adjust the reconciliation amount for quality performance as discussed in section X.A.3.d.(5)(e) of the preamble of this final rule to determine the quality-adjusted reconciliation amount. Then we would apply the stop-loss and stop-gain limits to the quality-adjusted reconciliation amount, as discussed in section X.A.3.d.(5)(f) of the preamble of this final rule, creating the NPRA. Finally, we proposed in the proposed rule to combine the NPRA with the results of the post-episode payment calculation (as discussed in section X.A.3.d.(5)(g) of the preamble of this final rule), to create the reconciliation payment amount or repayment amount. We sought comment on our proposal at proposed § 512.550(c-g) for calculating the reconciliation payment amount or repayment amount.
We did not propose to include any TEAM reconciliation payments or repayments to Medicare under this model for a given performance year in the reconciliation amount for a subsequent performance year. We wanted to incentivize providers to provide high quality and efficient care in all years of the model. If reconciliation payments for a performance year are counted as performance year spending in a subsequent performance year, a hospital would experience higher performance year spending in the subsequent performance year as a consequence of providing high quality and efficient care in the prior performance year, negating some of the incentive to perform well in the prior year. Therefore, we proposed in the proposed rule to not have the reconciliation amount for a given performance year be impacted by TEAM Medicare repayments or reconciliation payments made in a prior performance year. We sought comment on our proposal not to include TEAM reconciliation payments or repayments in performance year spending.
Comment: A commenter recommended that TEAM participants should not be eligible for reconciliation payments for an episode category if the quality of care on episode-specific measures decreases during the performance period compared to the hospital's performance during the baseline period. At the same time, CMS should reward quality improvement independent of cost performance.
Response: We thank the commenter for their recommendation and refer them to sections X.A.3.d.(5)(h) and X.A.3.d.(5)(g) of the preamble of the proposed rule, that would allow TEAM participants to be rewarded for their work to improve quality and cost outcomes for their episodes, but not be held financially accountable if spending exceeds the reconciliation target price. We believe the 10 percent stop-gain ( print page 69779) limit and a CQS adjustment percentage of up to 10 percent for Track 1 are appropriate and would allow TEAM participants to be rewarded for spending and quality performance while easing into financial risk. Further Track 3 would have two-sided financial risk in the form of reconciliation payments or repayment amounts, subject to 20 percent stop-gain and stop-loss limits and a CQS adjustment percentage of up to 10 percent, as described in sections X.A.3.d.(5)(h) and X.A.3.d.(5)(g) of the preamble of this final rule, that would allow TEAM participants to have higher levels of reward and risk based on their quality and cost performance for their episodes. We are finalizing our policy to allow all TEAM participants to participate in Track 1 for the first performance year, and safety net hospitals, as defined in section X.A.3.f of the preamble of this final rule, to be eligible to participate in Track 1 for the first three performance years with no downside risk, as discussed in section X.A.3.a.(3) of the preamble of this final rule. With TEAM having a five-year model performance period we do not believe that making Track 1 available for more than one performance year would motivate TEAM participants to improve quality or spending performance since there would be no financial accountability when spending reductions are not achieved. Furthermore, we believe TEAMs pay-for-performance methodology does not need a CQS threshold since poor quality performance in TEAM would negatively affect any positive or negative reconciliation amount. After consideration of the public comments we received, we are finalizing our proposals without modification at regulation § 512.550(c-g) for calculating the reconciliation payment amount or repayment amount.
As indicated in section X.A.3.c of the preamble of this final rule, the TEAM quality measure assessment is a pay-for-performance methodology aimed to incentivize and reward cost savings in relation to the quality of episode care provided by the TEAM participant. Similar to the BPCI Advanced model, we proposed in the proposed rule that a TEAM participant's quality performance would be linked to payment by translating the CQS into a CQS adjustment percentage and applying the CQS adjustment percentage to any positive or negative reconciliation amount. Specifically, for Track 1 TEAM participants we stated in the proposed rule that the CQS adjustment percentage would adjust a positive reconciliation amount up to 10 percent, and because Track 1 does not have downside risk, there would be no CQS adjustment percentage for negative reconciliation amounts. In the event a TEAM participant in Track 1 would have earned a negative reconciliation amount, their CQS would still be reported in their reconciliation report so that they may use this information to improve their quality measure performance in the next performance year. For Track 2 we stated in the proposed rule that the CQS adjustment percentage would adjust a positive reconciliation amount up to 10 percent and a negative reconciliation amount up to 15 percent. In other words, the CQS adjustment percent would not adjust the positive reconciliation amount down by more than 10 percent, nor would it adjust the negative reconciliation amount up (meaning more towards a positive amount) by more than 15 percent. For Track 3 TEAM participants, we stated in the proposed rule that the CQS adjustment percentage would adjust a positive reconciliation amount up to 10 percent and a negative reconciliation amount up to 10 percent. We would determine the CQS adjustment percentage using the following proposed formulas in Table X.A.-13.

In the proposed rule, we stated the CQS adjustment percentage would be multiplied with the TEAM participant's positive or negative reconciliation amount to produce the CQS adjustment amount. The CQS adjustment amount would then be subtracted from the positive or negative reconciliation amount to create the quality-adjusted reconciliation amount. We proposed in the proposed rule to define the quality-adjusted reconciliation amount as the dollar amount representing the difference between the reconciliation target price and performance year spending, after adjustments for quality, but prior to application of stop-gain/stop-loss limits and the post-episode spending adjustment, as described in sections X.A.3.d.(5)(h). and X.A.3.d.(5)(i). of the preamble of this final rule. Since we indicated in the proposed rule that Track 2 participation is limited to TEAM participants who may care for a higher proportion of underserved TEAM beneficiaries, we believed an asymmetric application of the CQS adjustment percentage for Track 2 TEAM participants may help to mitigate some the negative financial burden that may be associated with caring for underserved beneficiaries who tend to be higher cost and have worse health outcomes. Table X.A.-14 illustrates TEAM's methodology in the proposed rule of incorporating CQS into payment using the different CQS adjustment percentage scenarios using rounded values.

We considered, but did not propose, an asymmetric application of the CQS adjustment percentage for TEAM participants in Track 3. We believed the symmetric application in the proposed rule was appropriate to balance the amount of financial risk associated with quality performance since Track 3 was meant to have higher risks and rewards. Further, we also considered different CQS adjustment percentages for TEAM participants in all tracks including 20 percent, 25 percent, 33 percent and 50 percent but felt that these percentages may be too high given TEAM participants will have varying levels of experience with value-based care and a pay-for-performance methodology. We also considered lower CQS adjustment percentages for TEAM participants in all tracks including 1 percent, 3 percent, and 5 percent, but we believe these percentages would be too low and minimize the importance of quality improvement and thus would not incentivize TEAM participants to strive for quality of care improvements.
We also considered other approaches to tying TEAM quality measure performance to payment, including how the CJR Model applied their CQS methodology to adjust the discount factor. However, we believe the TEAM's proposed approach creates a greater incentive to improve quality measure performance because a TEAM participant must achieve of a CQS of 100 to receive the maximum quality-adjusted reconciliation amount. While this may be perceived as setting a high standard, it is consistent with the approach we have taken in BPCI Advanced and emphasizes the importance of beneficiary quality of care. We also note that TEAM's CQS methodology also tries to protect TEAM participants from increased financial risk based on their CQS. Meaning, if a TEAM participant has a negative reconciliation amount, performing better on quality would reduce their repayment amount. For example, a TEAM participant that performs well in quality ( e.g., a CQS of 100) would have a lower repayment amount, as compared to a TEAM participant that did poorly on quality ( e.g., score of 0) and would owe their full repayment amount.
Lastly, we considered applying a CQS threshold to be eligible to receive a reconciliation payment in TEAM. A similar approach was used in the CJR model where a participant hospital had to achieve a minimum CQS to receive a reconciliation payment, however, a level of quality performance that was below acceptable would not affect participant hospitals' repayment responsibility. We believed TEAM's pay-for-performance methodology as outlined in the proposed rule does not need a CQS threshold since poor quality performance in TEAM would negatively affect any positive or negative reconciliation amount.
We sought comment on TEAM's proposed methodology at proposed § 512.550(d) to calculate and apply the CQS. We also sought comment on our proposed definition of quality-adjusted reconciliation amount at § 512.505.
We invited public comment regarding the proposal to calculate and apply the CQS and the definition of quality-adjusted reconciliation amount. We received no comments on the proposals on how to calculate and apply the CQS and are finalizing as proposed without modification at § 512.550(d). We are also finalizing our proposal at § 512.505 the definition of quality-adjusted reconciliation amount.
In the proposed rule, we stated that in CJR and BPCI Advanced, we included both stop-loss and stop-gain limits on the total amount that a participant could owe to CMS as a repayment or receive from CMS as a reconciliation payment. For CJR, this policy and its justification is described in the 2015 CJR Final Rule at 80 FR 73398 . For both CJR and BPCI Advanced, these limits were applied as a percentage of a participant's total aggregate target price at reconciliation. Stop-loss and stop-gain limits gradually increased over the first few years of the CJR model, reaching a maximum of 20 percent for most hospitals for performance years 4-8, while the BPCI Advanced model has maintained 20 percent limits every model year for all participants.
As with CJR, we proposed in the proposed rule to phase in risk in TEAM. We stated in the proposed rule that Track 1 TEAM participants would not be subject to downside risk in PY 1. We also stated in the proposed rule that a stop-gain limit of 10 percent would be used for Track 1 TEAM participants in PY 1, and that TEAM participants in Track 2 would be subject to downside and upside risk with a symmetric stop-gain and stop-loss limits of 10 percent for PY 2-5. We indicated in the proposed rule that we believe a 10 percent stop-gain and stop-loss limit of 10 percent is appropriate for Track 2 participants who can gain value-based care experience but have less financial risk. However, since Track 3 would be designed for TEAM participants with prior experience in value-based care or those who are prepared to accept greater financial risk in the first year of TEAM, we stated in the proposed rule that TEAM participants that opt into Track 3 of the model would be subject to both upside and downside risk, with symmetric stop-gain and stop-loss limits of 20 percent for all performance years. The greater level of downside risk in Track 3 would therefore be balanced by higher stop-gain limits for Track 3 compared to Track 1 or Track 2, which we indicated in the proposed rule we would continue for all performance years.
We considered, but we did not propose, higher and lower stop-gain and stop-loss limits for Track 3, including 25 percent, 15 percent, and 10 percent but we believed maintaining consistency with 20 percent stop-gain and stop-loss limits of previous episode-based payment models provides an appropriate balance of financial risk and reward to promote spending reductions with reasonable risk thresholds. We also considered, but did not propose, lower stop-gain and stop-loss limits for Track 2, including 5 percent, 3 percent and 1 percent limits, or asymmetric limits, ( print page 69781) such as 10 percent stop-gain and 5 percent stop-loss limits or 5 percent stop-gain and 3 percent or 1 percent stop-loss. We also considered, but did not propose, lower and asymmetric limits for certain TEAM participants. For example, we considered a 10 percent or 5 percent stop-gain paired with a 3 percent or 1 percent stop-loss for TEAM participants who meet the criteria of a safety net hospital. Since TEAM offers a one-year glide path where all TEAM participants could elect to participate in Track 1 with no downside risk for PY 1, we do not believe lower or asymmetric limits would be necessary for Track 2. By PY 2 when Track 2 is available for certain TEAM participants, they should have sufficient infrastructure in place to assume two-sided risk while having less financial risk compared to Track 3. We sought comment on these alternative proposals for stop-gain and stop-loss limits and whether there are other mechanisms we should consider to help limit a TEAM participant's financial risk in the model.
We also indicated in the proposed rule we would apply stop-loss and stop-gain limits after application of the CQS which would result in the NPRA. We would define NPRA as the dollar amount representing the difference between the reconciliation target price and performance year spending, after adjustments for quality and stop-gain/stop-loss limits, but prior to the post-episode spending adjustment, which is described in section X.A.3.d.(5)(g) of the preamble of this final rule. We believed applying the stop-loss and stop-gain limits after the CQS was appropriate because it limits the financial risk associated with episode spending and quality performance, which is similar to how the BPCI Advanced model and CJR model apply stop-loss and stop-gain limits.
We sought comment on our proposal at proposed § 512.550(c)(vi) for differential stop-gain and stop-loss limits for TEAM participants by Track and Performance Year. We also sought comment on our NPRA definition at proposed § 512.505.
The following is a summary of public comments received on our proposal for the application of stop-loss and stop-gain limits and our responses to these comments:
Comment: Numerous commenters requested lower financial risk for rural hospitals. Many commenters noted the strain on rural hospitals who are not as risk tolerant, especially when these hospitals have lower volumes and less financial resources compared to larger, urban hospitals. Some commenters recommend reducing the CMS discount or adjusting the stop-gain and stop-loss risk corridors in Track 2 to provide a higher incentive for rural hospitals and safety net hospitals to participate; specifically reducing the stop-loss limits from 10 percent to 5 percent. A commenter recommended CMS to reconsider Track 2's proposed risk arrangement by implementing parallel upside and downside risk of 10 percent for that track.
Response: We thank commenters for their recommendations surrounding financial risk for rural hospitals. We recognize rural hospitals, as defined and finalized in section X.A.3.f of the preamble of this final rule, and other types of hospitals, including Sole Community Hospitals as defined under 42 CFR 412.92 , Medicare Dependent Hospitals as defined under 42 CFR 412.108 , and essential access community hospitals as defined under 42 CFR 412.109 , often serve as the only access of care for beneficiaries living in rural areas, may have fewer resources to contain costs under this model, and may have more limited options on providers to coordinate care with. Similar to safety net hospitals, these hospitals have been underrepresented in previous episode-based models and can also experience financial challenges, in part due to their lower patient volumes, that can make it difficult to sustain their resources. While rural hospitals are not being oversampled in TEAM, the use of core-based statistical areas (CBSAs) may capture a greater number of rural providers compared to using metropolitan statistical areas (MSAs) like the CJR model, thus potentially increasing their presence in TEAM. CMS has considered the unique needs of rural hospitals and tested the Pennsylvania Rural Hospital Model to engage rural providers in value-based care. However, the Pennsylvania Rural Hospital Model is not an episode-based payment model, is limited in scope, and is not permitted to overlap with certain episode-based payment models like the BPCI Advanced model. Therefore, we acknowledge that rural hospitals and the other hospitals eligible for Track 2, as described in section X.A.3.a.(3) of the preamble of this final rule, may need reduced financial risk to provide a more equitable participation opportunity in the model. We are finalizing our Track 2 stop-loss and stop-gain limits with slight modification by reducing the limits, from 10 percent to 5 percent for all performance years of Track 2. We believe this reduction will help protect from significant financial losses and is an appropriate threshold for these hospitals' exposure to downside financial risk.
In addition, as discussed in section X.A.3.d.(3)(h) of the preamble of this final rule, we are not finalizing our policy for low volume hospitals and will propose an updated policy in future notice and comment rulemaking that will address the level of risk these TEAM participants may have. We believe that a future low volume hospital policy, paired with the stop-gain and stop-loss limit reductions for Track 2, will help facilitate TEAM participants' abilities to be successful under this model.
Lastly, we thank the commenter for the recommendation to apply symmetrical stop-gain and stop-loss limits and highlight that in the proposed rule ( 89 FR 35934 ), we proposed that Track 2 have parallel or symmetrical stop-gain and stop-loss limits. Further, the changes we are finalizing to the stop-gain and stop-loss limits for Track 2 are symmetrical, such that both limits are set at 5 percent.
Comment: A commenter suggested that the application of the stop-loss/stop-gain threshold should be at the individual beneficiary or Clinical Episode level instead of the hospital level for the relevant Reconciliation period. The commenter cited concerns about low volume hospitals randomly experiencing anomalous expensive Clinical Episodes and how the financial impact of a single outlier episode will be larger for a low volume provider than a higher volume provider.
Response: We thank the commenter for sharing their concerns; however, we disagree that the stop-loss/stop-gain threshold should be at the individual beneficiary or Clinical Episode level. The intention of the stop-loss/stop-gain thresholds are to protect both TEAM participants and CMS from losses. In order to ensure that TEAM participants have some protection from the downside risk associated with clinical outliers that incur high payment episodes, we will cap episode spending at the 99th percentile for each DRG and region combination in both the baseline and performance year, as discussed in section X.A.3.d.(3)(e) of the preamble of this final rule. We believe that applying the stop-loss/stop-gain threshold at the individual level would also blunt the incentives providers have to reduce the frequency of bad outcomes. As indicated in section X.A.3.d.(3)(h) of the preamble of this final rule, we intend to provide additional flexibility and protection to low volume hospitals and will propose a new policy in future notice and comment rulemaking prior to the model start date. ( print page 69782)
After consideration of the public comments we received, we are finalizing our proposal with some slight modification for differential stop-gain and stop-loss limits for TEAM participants by Track and Performance Year at regulation § 512.550(e)(1-3). Specifically, we are modifying our proposal for Track 2 to have 5 percent stop-gain at § 512.550(e)(2) and 5 percent stop-loss limits and § 512.550(e)(3). We are also finalizing our proposal to define NPRA at regulation § 512.505.
While the episodes as stated in the proposed rule would extend 30 days post-discharge from the anchor hospitalization or post-procedure (for outpatient episodes), some hospitals may have an incentive to withhold or delay medically necessary care until after an episode ends to reduce their actual episode payments. We did not believe this would be likely, but in order to identify and address such inappropriate shifting of care, we stated in the proposed rule we would calculate the total Medicare Parts A and B expenditures in the 30-day period following completion of each episode for all services covered under Medicare Parts A and B for each performance year, regardless of whether the services are included in the episode definition proposed in the proposed rule (section X.A.3.b.(5) of the preamble of this final rule). Because we based the episode definition on exclusions, identified by MS-DRGs for readmissions and ICD-10-CM diagnosis codes for Part B services as discussed in section X.A.3.b.(5)(a) of the preamble of this final rule, and Medicare beneficiaries may typically receive a wide variety of related (and unrelated) services during episodes, there is some potential for hospitals to inappropriately withhold or delay a variety of types of services until the episode concludes regardless of whether the service is included in the episode definition, especially for Part B services where diagnosis coding on claims may be less reliable. This inappropriate shifting could include both those services that are related to the episode (for which the hospital would bear financial responsibility as they would be included in the actual episode spending calculation) and those that are unrelated (which would not be included in the actual episode spending calculation), because a hospital engaged in shifting of medically necessary services outside the episode for potential financial benefit may be unlikely to clearly distinguish whether the services were related to the episode or not.
This calculation would include prorated payments for services that extend beyond the episode as discussed in section X.A.3.d.(3)(c) of this final rule. Specifically, at proposed § 512.550(f) we stated in the proposed rule we would identify whether the average 30-day post-episode spending for a TEAM participant in any given performance year is greater than three standard deviations above the regional average 30-day post-episode spending, based on the 30-day post-episode spending for episodes attributed to all TEAM regional hospitals in the same region as the TEAM participant. We stated in the proposed rule that beginning with PY 1 for Track 3 TEAM participants, and PY 2 for Track 2 TEAM participants, if the TEAM participant's average post-episode spending exceeds this threshold, the amount above the threshold would be subtracted from the reconciliation amount or added to the repayment amount for that performance year. The amount above the threshold would not be subject to the stop-loss limits proposed elsewhere in the proposed rule. We sought comment on this proposal at proposed § 512.550(f) to make TEAM participants responsible for making repayments to Medicare based on high spending in the 30 days after the end of the episode and for our proposed methodology to calculate the threshold for high post-episode spend.
The following is a summary of public comments received regarding the proposal to make TEAM participants responsible for making repayments to Medicare based on high post-episode spending and the proposed methodology to calculate the threshold for high post-episode spending, and our responses to these comments:
Comment: A commenter supported the use of post-episode spending calculations and NPRA adjustments to ensure care is not being postponed. Some commenters suggested that CMS should compare the post-episode spending for TEAM participants to their peers, citing concerns that region alone will not sufficiently capture differences in the hospital type and the patient populations (such as trauma and oncology). One of the commenters recommended using the same peer group characteristics as the BPCI Advanced model.
We thank the commenters for sharing their support and concerns with the proposed post-episode spending methodology; however, we continue to believe that participant post-episode spending should be compared to region-level distributions.
Since peer groups would include fewer hospitals than regions, the standard deviation of the distribution of post-episode spending could be larger. This would reduce CMS's ability to identify and address inappropriate withholding or delaying of medically necessary care for potential financial benefit. Furthermore, it is more straightforward to keep the level of comparison consistent with the in-episode spending calculations, which are at the region-level.
We acknowledge the commenters' specific concern for trauma and oncology patients. However, CMS believes that it is imperative to compare post-episode spending across hospitals regardless of the types of services provided, as a hospital engaged in shifting of medically necessary services outside the episode for potential financial benefit may be unlikely to clearly distinguish whether the services were related to the episode or not. We will continue to assess our target price methodology, including the construction of the post-episode spending calculation, and if we believe a modification is necessary, we will propose such modification in future notice and comment rulemaking.
After consideration of the public comments we received, we are finalizing our proposal at § 512.550(f) to compare post-episode spending for TEAM participants to the region-level distribution.
For the PY 1 reconciliation process for Track 1 TEAM participants, we indicated in the proposed rule we would combine a TEAM participant's NPRA and post-episode spending amount, as described previously in this section, and if positive, the TEAM participant would receive the amount as a one-time lump sum reconciliation payment from Medicare. If negative, the TEAM participant would not be responsible for repayment to Medicare, consistent with our proposal for a 1-year glide path to phase in greater financial responsibility in the model. For TEAM participants in Track 3 for PY 1, and Track 2 or Track 3 for PYs 2-5, if the amount is positive, the TEAM participant would receive the amount as a one-time lump sum reconciliation payment from Medicare. If the amount is negative, Medicare would hold the TEAM participant responsible for a one-time lump sum repayment. CMS would collect the one-time lump sum repayment in a manner that is ( print page 69783) consistent with all relevant federal debt collection laws and regulations.
We want participants to succeed in TEAM by providing high quality care to TEAM beneficiaries and reducing episode spending, but we understand there may be instances when a TEAM participant does not meet performance metrics and owes a repayment amount. We acknowledge paying back Medicare in a lump sum for a repayment amount may introduce financial hardship for some TEAM participants, especially those who may be new to value-based care with downside risk or those who have fewer financial resources. In some CMS Innovation Center models, certain participants are required to have financial guarantees, which act as a reinsurance policy for CMS if the participant is unable to pay back debts owed as a result of their performance in the model. For example, the BPCI Advanced model requires certain participants to have secondary repayment sources, generally in the form of a letter of credit or escrow agreement, that can be drawn upon if the participant is unable or fails to pay their repayment amount. Yet, financial guarantees require upfront capital and must be replenished in a timely manner for potential use in future debts. Further, financial guarantees generally need to be established before the model starts, thus before the TEAM participant would be eligible to use any TEAM payment amounts to fund the financial guarantee.
We do not believe financial guarantees would be appropriate for TEAM given the aforementioned concerns but recognize that providing some process to prolong recovery of a repayment amount may be needed to mitigate potential financial hardships. Existing Medicare policy allows the recovery of Medicare debt, defined as recoupment in 42 CFR 405.370 , and non-Medicare debt, defined as offset in 42 CFR 405.370 , by reducing present or future Medicare payments and applying the amount withheld to the indebtedness. To leverage the existing Medicare policy to recover debts in TEAM, we considered whether the reduction of present or future Medicare payments should be a dollar amount reduction, for example a $100 reduction of all Medicare payments, or a percentage reduction applied to all Medicare payments, for example a 2 percent reduction to Medicare payments. A dollar amount reduction may be simpler to calculate while translating a debt to a percentage reduction may be more complex to calculate. We also considered whether the reduction of present or future Medicare payments should only be associated with a TEAM participant's Medicare Part A payments for the corresponding episode categories tested in TEAM or for all of a TEAM participant's Medicare Part A payments. Limiting the Medicare payment reduction to only corresponding episode categories tested in TEAM may draw out the length of time for debt recovery, but it may ease TEAM participant bookkeeping when accounting for TEAM financial performance. Conversely, reduction of Medicare payments for all of a TEAM participant's Medicare Part A payments may reduce the length of time for debt recovery, but it may be more challenging to identify and track TEAM participant financial performance.
We did not propose to require financial guarantees or change existing Medicare recoupment or offsetting policies, but we sought comment on whether we should consider these options further or if there are other ways to reduce financial hardship for TEAM participants that owe a repayment amount. We also sought comment on whether we should consider a Medicare payment policy waiver to reduce financial hardship, what the waiver would waive, and if the waiver is necessary to avoid undue burden on TEAM participants.
We invited public comment on whether we should consider these options further or if there are other ways to reduce financial hardship for TEAM participants that owe a repayment amount. We also sought comment on whether we should consider a Medicare payment policy waiver to reduce financial hardship, what the waiver would waive, and if the waiver is necessary to avoid undue burden on TEAM participants.
We also considered an alternative approach to making reconciliation payments and collecting repayments from TEAM participants. Under this alternative approach, in lieu of making a lump sum payment to TEAM participants, or collecting a repayment amount from TEAM participants, we would instead make a percentage adjustment to future fee-for-service (FFS) claims for TEAM participants. The magnitude of the adjustments would be intended to approximate the same dollar amount that would be paid or recouped via a reconciliation process; adjustments would be made in the form of a multiplier on claims for the anchor procedures for the episodes included in TEAM. For example, we would make adjustments to IPPS claims containing the MS-DRGs included in the model, and the amounts of the adjustments for each TEAM participant over the course of a year would, in aggregate, be intended to approximately equal the dollar amount that would have otherwise been paid via a reconciliation payment (or recouped via a repayment amount). The alternative approach would look similar to the operational payment mechanisms used in other Medicare programs and initiatives such as the Hospital Value-Based Purchasing Program, the SNF Value-Based Purchasing Program, the Expanded Home Health Value-Based Purchasing Model, and the Hospital Readmissions Reduction Program. We considered a value-based purchasing payment approach because we believe it has the potential to be less operationally cumbersome than making separate reconciliation payments if TEAM is expanded nationally in the future. We also believed that a value-based purchasing payment approach that adjusts future FFS claims up or down would provide financial stability for TEAM participants, because they would receive notice of their adjustment amounts ahead of the year in which those adjustments would apply, and TEAM participants that would otherwise owe a repayment amount could effectively pay that debt over time automatically via claims adjustments, versus writing a check to CMS.
A value-based purchasing approach for TEAM would not be without challenges, however. First, preliminary modeling indicates that payment adjustment percentages for the proposed episodes may need to be relatively large in order to approximate the same dollar amount that would otherwise be paid out via a reconciliation payment or paid to CMS via a repayment amount. Although the adjustment percentages would be limited to a subset of FFS claims for a given TEAM participant, we believe we must be cautious that particularly for some providers, a negative adjustment to FFS claims could represent a financial hardship. Second, we considered whether claims adjustments should be made to only IPPS claims (for the MS-DRGs that trigger an anchor procedure/hospitalization for an episode), or also to Outpatient Prospective Payment System (OPPS) claims, given that we proposed to include episodes that initiate in the outpatient setting in TEAM for certain episode categories. Making adjustments to both IPPS and OPPS claims would add complexity, particularly since the IPPS payment updates are made on a fiscal year schedule, while the OPPS updates payments on a calendar year cycle. We ( print page 69784) sought comment on whether, for TEAM or other future initiatives that may consider a similar value-based purchasing approach, we should make adjustments to IPPS claims only or also OPPS claims that trigger model episodes.
We sought comment on our proposal making reconciliation payments to, and collecting repayment amounts from, TEAM participants as a one-time, lump sum payment, as well as the alternative considered to implement a value-based purchasing approach where we make payment adjustments to future FFS claims in lieu of lump sum payments or repayments.
Comment: A commenter suggested that the CMS Innovation Center could determine the likely reimbursement from the IRS, provide that money to the TEAM participant, and get reimbursed by the IRS at project completion. The commenter also suggested that we provide some way to link TEAM participants with other funding sources to overcome this same problem of time-delay of the reimbursements or provide some kind of loan program, available ONLY to TEAM participants.
Response: We thank the commenter for sharing their support and concerns regarding payment options for TEAM participants. This would require infrastructure and policy changes across multiple agencies that are outside of the scope of TEAM, but we will continue to consider improvements to how we collect repayment amounts from TEAM participants.
Comment: A commenter recommended CMS allow participants to pay money back after the reconciliation process rather than having CMS recoup future payments and encouraged CMS to finalize an annual reconciliation process. Also, the commenter recommended that CMS provide participants regular monthly reporting.
Response: We thank the commenter for their encouragement and recommendations. TEAM participants will have the opportunity to pay repayment amounts before they are recouped from future Medicare payments. TEAM participants will receive cumulative beneficiary-identifiable claims files on a monthly basis, pursuant to a request and TEAM data sharing agreement, for care coordination purposes and to help estimate their performance.
Comment: A commenter recommended that CMS should make reconciliation payments in its value-based models by adjusting future fee-for-service claims in lieu of lump sum reconciliation because it would eliminate the challenges with obtaining the demand letters. The commenter also recommended that CMS electronically deliver demand letters instead of mailing them as an alternative approach.
Response: We thank the commenter for their concern regarding their challenges associated with demand letters that are sent via regular mail. We disagree that the usage of the mail service is onerous for our participants but may consider their recommendations for future rulemaking.
After consideration of the comments received, we are finalizing without modification at § 512.550(g)(2-3) the proposed provisions for lump sum reconciliation payments and repayment amounts.
At proposed § 512.560, we stated in the proposed rule the following first level appeal process for TEAM participants to contest matters related to payment or reconciliation, of which the following is a non-exhaustive list: The calculation of the TEAM participant's reconciliation amount or repayment amount as reflected on a TEAM reconciliation report; the calculation of net payment reconciliation amount (NPRA); and the calculation of the Composite Quality Score (CQS). We stated in the proposed rule that TEAM participants would review their TEAM reconciliation report and be required to provide a notice of calculation error that must be submitted in a form and manner specified by CMS. Unless the participant provides such notice, we indicated in the proposed rule that the reconciliation report would be deemed final within 30 calendar days after it is issued, and CMS would proceed with payment or repayment. In the proposed rule, we proposed that if CMS receives a timely notice of an error in the calculation, CMS will respond in writing within 30 calendar days to either confirm or refute the calculation error, although CMS would reserve the right to an extension upon written notice to the TEAM participant. If a TEAM participant does not submit timely notice of calculation error in accordance with the timelines and processes specified by CMS, the TEAM participant would be precluded from later contesting any element of the TEAM reconciliation report for that performance year.
At proposed § 512.560(b) we proposed in the proposed rule an exception to the appeals process. If a TEAM participant contests a matter that does not involve an issue contained in, or a calculation that contributes to, a TEAM reconciliation report, a notice of calculation error is not required. A notice of calculation error form would not be an appropriate format for addressing issues other than calculation errors, given that it is tailored specifically to calculation errors. In these instances, we stated in the proposed rule that if CMS does not receive a request for reconsideration from the TEAM participant within 10 calendar days of the notice of the initial reconciliation, the initial determination is deemed final and CMS proceeds with the action indicated in the initial determination. We note that this proposed exception does not apply to the limitations on review in § 512.594.
We sought comment on our proposal for the first level appeals process.
We received no comments on these proposals and therefore are finalizing this provision without modification at § 512.560.
At proposed § 512.561, we proposed in the proposed rule a reconsideration process that is based on processes implemented under current models being tested by the CMS Innovation Center. The process would enable TEAM participants to contest determinations made by CMS. We proposed at § 512.594 to waive section 1869 of the Act, which governs determinations and appeals in Medicare and instead we proposed to codify a reconsideration process for TEAM participants to utilize. We proposed at § 512.561(a) that only TEAM participants may utilize the dispute resolution process. We believe establishing a reconsideration process is necessary to give TEAM participants a means to dispute certain determinations made by CMS.
This proposed reconsideration review process would be utilized in the case that a determination has been made and the TEAM participant disagrees with that determination. Part 512 subpart E would include specific details about when a determination is final and may be disputed through the reconsideration review processes.
At proposed § 512.561(b), we stated in the proposed rule that TEAM participants may request reconsideration of a determination made by CMS, only if such reconsideration is not precluded by section 1115A(d)(2) of the Act or this subpart. At proposed ( print page 69785) § 512.561(b)(1)(i) we stated in the proposed rule that a request for review of those final determinations made by CMS that are not precluded from administrative or judicial review would be submitted to a CMS reconsideration official. The CMS reconsideration official would be authorized to receive such requests and would not have been involved in the initial determination or, if applicable, the notice of calculation error process. At proposed § 512.561(b)(1)(ii) we stated in the proposed rule that the reconsideration review request would be required to include a copy of CMS's initial determination, contain a detailed written explanation of the basis for the dispute, and at proposed § 512.561(b)(1)(iii) we stated in the proposed rule that the request would have to be made within 30 days of the date of CMS's initial determination via email addressed to an address specified by CMS. At proposed § 512.561(b)(2), we indicated in the proposed rule that requests that do not meet the requirements of paragraphs (b)(1) are denied.
We indicated in the proposed rule that the reconsideration official would send a written acknowledgement to CMS and to the TEAM participant requesting reconsideration within 10 business days of receiving the reconsideration request. The acknowledgement would set forth the review procedures and a schedule that permits each party an opportunity to submit documentation in support of their position for consideration by the reconsideration official.
At proposed § 512.561(b)(1)(i)(B) we proposed, that, to access the reconsideration process for a determination concerning a TEAM payment, the TEAM participant would be required to satisfy the notice of calculation error requirements specified in section X.A.3.d.(6)(a) of the preamble of this final rule before submitting a reconsideration request under this process. In the event that the model participant fails to timely submit an error notice with respect to a TEAM payment, we stated in the proposed rule that the reconsideration review process would not be available to the TEAM participant with regard to that payment.
At proposed § 512.561(c), we stated in the proposed rule we would codify standards for the reconsideration. First, during the course of the reconsideration, both CMS and the party requesting the reconsideration must continue to fulfill all responsibilities and obligations during the course of any dispute arising under TEAM. Second, the reconsideration would consist of a review of documentation timely submitted to the reconsideration official and in accordance with the standards specified by the reconsideration official in the acknowledgement at proposed § 512.561(b)(3). Finally, we stated in the proposed rule that the burden of proof would be on the TEAM participant to prove to the reconsideration official, by a standard of clear and convincing evidence, that the determination made by CMS was inconsistent with the terms of TEAM.
At proposed § 512.561(d) we stated in the proposed rule that the reconsideration determination would be an on-the-record review. By this, we meant a review that would be conducted by a CMS reconsideration official who is a designee of CMS who is authorized to receive such requests under proposed § 512.561(b)(1)(i), of the position papers and supporting documentation that are timely submitted and meet the standards of submission under proposed § 512.561(b)(1) as well as any documents and data timely submitted to CMS by the TEAM participant in the required format before CMS made the initial determination. Under the proposed § 512.561(d)(2), the reconsideration official would issue to CMS and the TEAM participant a written reconsideration determination. Absent unusual circumstances in which the reconsideration official would reserve the right to an extension upon written notice to the TEAM participant, the reconsideration determination would be issued within 60 days of CMS's receipt of the timely filed position papers and supporting documentation. Under proposed § 512.561(d)(3), the determination made by the CMS reconsideration official would be final and binding 30 days after its issuance, unless the TEAM participant or CMS were to timely request review of the reconsideration determination by the CMS Administrator in accordance with proposed § 512.5610(e)(1) and (2).
We received no comments on these proposals and therefore are finalizing this provision without modification at § 512.561.
We stated in the proposed rule we would codify at proposed § 512.561(e) a process for the CMS Administrator to review reconsideration determinations made under proposed § 512.561(d). We indicated in the proposed rule that either the TEAM participant or CMS may request that the CMS Administrator review the reconsideration determination made by the reconsideration official. Under proposed § 512.561(e)(1), we stated in the proposed rule that the request to the CMS Administrator would have to be made via email, within 30 days of the reconsideration determination, to an email address specified by CMS. The request would have to include a copy of the reconsideration determination, as well as a detailed written explanation of why the model participant or CMS disagrees with the reconsideration determination. Under proposed § 512.561(e)(4), we stated in the proposed rule that promptly after receiving the request for review, the CMS Administrator would send the parties an acknowledgement of receipt that outlines whether the request for review was granted or denied and, should the request for review be granted, the review procedures and a schedule that would permit both CMS and the TEAM participant an opportunity to submit a brief in support of their positions for consideration by the CMS Administrator. Should the request for review be denied, under proposed § 512.561(e)(5), we indicated in the proposed rule that the reconsideration determination would be final and binding as of the date of denial of the request for review by the CMS Administrator. Under proposed § 512.561(e)(6), we indicated in the proposed rule that should the request for review by the CMS Administrator be granted, the record for review would consist solely of timely submitted briefs and evidence contained in the record before the reconsideration official and evidence as set forth in the documents and data described in proposed § 512.561(d)(1)(ii); the proposed rule indicated that the CMS Administrator would not consider evidence other than information set forth in the documents and data described in proposed § 512.561(d)(1)(ii). As stated in the proposed rule, the CMS Administrator would review the record and issue to CMS and the TEAM participant a written determination that would be final and binding as of the date the written determination was sent.
We solicited comments on the proposed reconsideration review process included in TEAM. The following is a summary of the public comments received on this proposal and our responses to those comments:
Comment: A commenter expressed support for the proposed processes and timelines for submission of calculation error notices (“CEN”) and reconsideration requests. Additionally, the commenter recommended we use an impartial third party in evaluating issues related to model implementation. ( print page 69786)
Response: We appreciate the commenter's support and recommendation. TEAM will use multiple levels of review to ensure a fair evaluation of all appeals submitted, including an Administrative Review by the CMS Office of the Administrator if the TEAM participant submits a timely request.
Comment: A commenter expressed support for the proposed process for requesting CMS Administrator Review.
After consideration of the comments received, we are finalizing at § 512.561 the proposed reconsideration review processes for TEAM.
In the proposed rule we stated that when determining the best strategy for addressing model overlap, we recognize we need to consider how to promote meaningful collaboration between providers and TEAM participants. In prior models, overlap policies were intended to be simple by avoiding duplicative incentive payments or giving precedence to a single accountable entity. However, what resulted were confusing methodologies or misaligned incentives which were difficult to navigate. Participants from prior models have also cited confusion with identifying to which model(s) a beneficiary may be aligned or attributed.
In earlier episode-based payment models, such as CJR (in certain circumstances) and BPCI, CMS addressed overlap by implementing a complex calculation and recouping a portion of the pricing discount for providers also participating in certain ACO initiatives. The recoupment was intended to prevent duplicate incentive payments for the same beneficiary's care; however, some participants perceived the resulting recoupment as a financial loss, discouraging providers from participating in both initiatives.
To avoid complexity, the CJR and BPCI Advanced models exclude beneficiaries aligned or assigned to certain ACOs, and these beneficiaries will not trigger a clinical episode. [ 938 ] While this exclusionary approach creates a clean demarcation of who is accountable for a beneficiary's care, it also limits the number of providers in accountable care relationships and becomes less tenable as we work towards the goal of increased accountability. Additionally, participants may be informed of beneficiary ACO alignment or assignment after the potential episode has been initiated and the expending of resources on unattributed beneficiaries. This concern highlights the opportunity to incentivize coordinated care, expand care redesign efforts to more patients, and strengthen APM participation.
Even passive avoidance of duplicated payments has its drawbacks such as lack of incentive to coordinate care. For example, the CJR and BPCI Advanced models allow overlap with the Medicare Shared Savings Program without a financial recoupment. [ 939 940 ] However, this policy does not encourage behavior change to ensure a smooth transition back to population-based providers.
In the proposed rule, we acknowledge that there may be circumstances where a Medicare beneficiary in an episode may also be assigned to an ACO, advanced primary care model, or other model or initiative being implemented through the CMS Innovation Center or otherwise through CMS. For the purposes of this final rule, “total cost of care” models or programs refer to models or programs in which episodes or performance periods include participant financial responsibility for all Part A and Part B spending, as well as some Part D spending in select cases. We use the term “shared savings” in the proposed rule to refer to models or programs in which the payment structure includes a calculation of savings (that is, the difference between FFS amounts and program or model benchmark) and CMS and the model or program participant each retain a particular percentage of that savings. We noted that there exists the possibility for overlap between episode-based payment model and shared savings models or programs such as Shared Savings Program, specialty care models such as the Enhancing Oncology Model (EOM), advanced primary care models such as Making Care Primary (MCP), state-based models such as the All-Payer Health Equity Approaches and Development model (AHEAD), or other CMS Innovation Center payment models that incorporate per-beneficiary-per-month (PBPM) fees or other payment structures.
We state in the proposed rule that in addition to the Shared Savings Program, there are other ACO and CMS Innovation Center models that make or will make, once implemented, providers accountable for total cost of care over a period of time (for example, 6 to 12 months). Some of these are shared savings models (or programs, in the case of the Shared Savings Program), while others are not shared savings but hold participating providers accountable for the total cost of care during a defined episode. Each of these payment models or programs holds providers accountable for the total cost of care over the course of an extended period or episode by applying various payment methodologies. We stated in the proposed rule that we believe it is important to simultaneously allow beneficiaries to participate in broader population-based and other total cost of care models, as well as episode payment models that target a specific episode with a shorter duration, such as TEAM. Allowing beneficiaries to receive care under both types of models may maximize the potential benefits to the Medicare Trust Funds and participating providers and suppliers, as well as beneficiaries. Research suggests that shared beneficiaries in episode-based payment models and ACOs can lead to lower post-acute care spending and reduced readmissions. [ 941 ] Beneficiaries stand to benefit from care redesign that may lead to improved quality for episodes even while also receiving care under these broader models, while entities that participate in other models and programs that assess total cost of care stand to benefit, at least in part, from the cost savings that accrue under TEAM. For example, a beneficiary receiving a procedure under TEAM may benefit from a hospital's care coordination efforts regarding care during the inpatient hospital stay. The same beneficiary may be attributed to a primary care physician affiliated with an ACO who is actively engaged in coordinating care for all the beneficiary's clinical conditions throughout the entire performance year, ( print page 69787) beyond the 30-day post-discharge period of the episode.
We proposed that a beneficiary could be in an episode in TEAM, as described in the Episodes section: X.A.3.b. of this final rule, by undergoing a procedure at a TEAM participant, and be attributed to a provider participating in a total cost of care or shared savings model or program. For example, a beneficiary may be attributed to a provider participating in the Shared Savings Program for an entire performance year, as well as have initiated an episode in TEAM during the ACO's performance year. Each model or program incorporates a reconciliation process, where total included spending during the performance period or episode are calculated, as well as any potential savings achieved by the model or program. We proposed to allow any savings generated on an episode in TEAM and any contribution to savings in the total cost of care model be retained by each respective participant. This would mean the episode spending in TEAM would be accounted for the in the total cost of care model's total expenditures, but TEAM's reconciliation payment amount or repayment amount would not be included in the total cost of care model's total expenditures. Likewise, the total cost of care model's savings payments or losses would not be included in the episode spending in TEAM.
In the proposed rule we noted that by allowing a beneficiary aligned to a total cost of care model participant to also be attributed to an episode in TEAM, we would be eliminating complexities experienced in prior models where it was difficult for participants to know when a beneficiary would trigger an episode and when the episode would be excluded. We stated that in prior models such as BPCI, we implemented a recoupment process after reconciliation to account for any duplicative savings generated on overlapping beneficiaries. This process involved disbursing reconciliation payments to BPCI participants and then submitting a recoupment demand for any savings generated on overlap. Overwhelming feedback from participants indicated that this recoupment process was perceived negatively and postured participants in BPCI and the total cost of care model into an adversarial relationship. Allowing overlap between beneficiaries aligned to a total cost of care model who also initiate an episode in TEAM and by allowing both participants to retain savings will have a positive impact on beneficiaries by fostering a cooperative relationship between accountable care and TEAM participants where all parties have interest in providing coordinated, longitudinal care.
We indicated in the proposed rule that overlap does mean that episode expenditures will be included in ACO expenditures and thus, have a potential impact on ACO performance. Whether or not this benefits an ACO's shared savings involves a variety of contributing factors that span beyond merely the results of episodes in TEAM. For example, an ACO's size and volume of aligned beneficiaries or the dynamics of certain markets in which an ACO operates could impact an ACO's expenditure calculations and shared savings. We stated in the proposed rule that CMS cannot isolate each variable that could influence an ACO's expenditures and shared savings, nor can CMS propose a singular policy that will ensure all ACOs benefit from interaction, or lack thereof, with TEAM. But because TEAM will be mandatory in specific markets, the model will be generally expected to similarly impact a Shared Savings Program ACO's episode spending and corresponding regional episode spending that contributes most of its retrospective benchmark update. This interaction is anticipated to largely mitigate potential overlapping incentive payments for the largest ACO program in traditional Medicare. We stated in the proposed rule that we believe allowing overlap and the retention of savings by ACOs and TEAM participants will encourage providers to collaboratively deliver coordinated care and yield improved outcomes to beneficiaries. This aligns with broader agency goals to foster increased beneficiary alignment to value-based care and allows us to learn from experience and avoid creating challenges managing shared beneficiaries between ACOs and episodes of care participants. In addition, there are other potential benefits to allowing overlap between a beneficiary aligned to a total cost of care model and initiate an episode in TEAM, such as strengthening the volume of episodes a TEAM participant is responsible for. We know from prior experience that low episode volume creates challenges for participants to generate meaningful savings and manage outlier cases with unusually high episode expenditures.
We also acknowledge in the proposed rule that certain ACOs may prefer for their aligned beneficiary population to not be included in TEAM. Since ACOs are accountable for total cost of care, they may prefer to manage their beneficiaries and have full control over all expenditures and beneficiary care instead of sharing that responsibility with a TEAM participant. Alternatively, we sought comment on prohibiting aligned beneficiaries from full-risk population-based care relationships (for example, Shared Savings Program Enhanced Track) from being in an episode in TEAM. We sought comment specifically on non-condition specific care relationships (that is, this would exclude condition-specific models such as the Enhancing Oncology Model (EOM)).
Additionally, we sought comment on the use of supplemental data (for example, shadow bundles [ 942 ] data) as providing a total cost of care or shared savings model participant with the ability to utilize episodes to improve care coordination and reduce cost.
The following is a summary of comments we received related to the proposed model overlap policy and our responses to those comments:
Comment: Overall, we received considerable support for our proposed policy to allow model overlap between beneficiaries aligned to a total cost of care model or shared savings model who also initiate an episode in TEAM. These comments included support for allowing both participants to retain savings.
Response: We appreciate the many comments of support received for this aspect of the model. In particular, we appreciate that commenters agree with our desire to simplify the attribution process, maintain higher TEAM participant volume, and incentivize engagement between total cost of care or shared savings model participants and TEAM participants to foster more integrated, patient-centered approach to care.
We believe by allowing model overlap, this encourages provides participating in TEAM and shared savings or total cost of care models or programs to engage collaboratively and strengthen care coordination for aligned beneficiaries. We also believe that by allowing TEAM participants and shared savings or total cost of care model or program participants to retain savings generated in their respective models, we are eliminating the risk of these participants forming an adversarial relationship.
Comment: We received some responses from commenters who ( print page 69788) opposed our proposed policy to allow model overlap between TEAM participants and total cost of care participants. Commenters mentioned that allowing model overlap could create confusion or duplication of effort, and place substantial administrative burdens on hospitals and the clinicians and staff. Other commenters mentioned that allowing overlap between TEAM and existing advanced alternative payment models, such as total cost of care models or shared savings models, is contrary to the philosophy of a total cost of care model and may disrupt ongoing initiatives, particularly if not all providers within an umbrella ACO must participate.
Response: We recognize that participants in total cost of care or shared savings models will have their own ongoing initiatives and strategies; however, we do not see TEAM as disrupting these efforts. By allowing TEAM episodes to overlap with a total cost of care or shared savings model, we are eliminating confusion over who is attributed, and therefore responsible, for the episode and encouraging coordination between TEAM participant and total cost of care or shared savings model participant by creating a shared goal of caring for the aligned beneficiary.
Comment: We sought comment on prohibiting aligned beneficiaries from full-risk population-based care relationships (for example, Shared Savings Program Enhanced Track or ACO REACH) from being in an episode in TEAM and we received responses from some commenters supporting this concept citing that these total cost of care or shared savings participants are already bearing full risk for any TEAM episodes initiated for their aligned population. Several commenters mention that requiring these participants to participate could avoid unnecessary complexity, duplication of efforts and confusion regarding how CMS will recognize these Medicare beneficiaries under these different types of accountable care models. A commenter suggested that allowing an opt-out would create an additional incentive for participation in two-sided risk total cost of care models and recognize that these entities are already accountable for cost and health outcomes for their population.
Response: We appreciate that these commenters want to acknowledge that full-risk model or program participants have taken on the greatest amount of risk for their aligned populations. CMS's goal is to create the ability for TEAM participants to engage and collaborate with other model or program participants through TEAM overlap policy, not to be a drain on resources or to elicit confusion. In fact, CMS learned from prior model experience that by excluding certain models or programs and by setting rules in which savings was not retained by both participants caused significant confusion and frustration. Specifically, when considering full risk models or programs, we expect these participants likely have experience in population-based care and are invested in the concept of value-based care. As such, we expect these participants to be able to manage their populations of aligned beneficiaries will coordinating effectively with TEAM participants on any overlap using their resources, when needed, to ensure smooth care coordination for their aligned beneficiaries.
Comment: A commenter suggested to allow total cost of care participants the ability to opt-out of mandatory participation for total cost of care participants who are actively managing episodes through another mechanism, such as a shadow bundle or different kind of focused care intervention.
Response: We acknowledge that total cost of care or shared savings model participants will have other initiatives they operate to support focused care intervention. However, for CMS to monitor total cost of care or shared savings participants to ensure they are actively utilizing another mechanism, such as a shadow bundle, CMS would have to create and operate an audit and election process which would be a significant drain on CMS resources to establish. Additionally, creating an audit or election process would be an administrative challenge for TEAM participants who moved in and out of model participation based on their varying interventions and strategies.
Comment: We received some comments that asked for clarity regarding how TEAM episodes would impact a total cost of care model's expenditures. Specifically, commenters asked if the TEAM target price or FFS expenditures would be used in total cost of care model expenditures used to calculate model shared savings and benchmarks. A commenter stated that using FFS expenditures instead of the TEAM target price would avoid being a source of friction across programs, allowing ACOs to clearly understand and track their performance throughout the performance period.
Response: CMS can confirm that FFS expenditures, not the TEAM target price, will be used in other model savings calculations. These expenditures represent the actual claims billed for services furnished to the aligned beneficiary.
Comment: Regarding our request on supplemental data to provide a total cost of care or shared savings model participant with the ability to utilize episodes to improve care coordination and reduce cost, a couple of commenters made requests for additional data elements. Additionally, commenters also requested that existing data initiatives provided by CMS, such as Shadow Bundles data, were not discontinued. A commenter requested that CMS include a data flag representing ACO assignment within the raw claims files that would be provided to a TEAM participant. Another commenter requested that CMS continue to provide shadow bundles data on all clinical episode categories currently offered (which includes the 34 BPCI Advanced clinical episode categories).
Response: CMS appreciates these comments and the desire to use data provided to TEAM participants to identify model overlap and use it to benefit engagement with shared savings or total cost of care model participants.
As CMS develops the structure of the raw claims files that will be provided on a regular basis to TEAM participants, we will explore the ability to add model overlap assignment information when feasible. This aligns with how model overlap is represented in raw claims files in prior and existing models such as CJR and BPCI Advanced.
Regarding shadow bundles data, CMS is currently committed to sharing this data on a regular basis. We are also exploring options for how to continue to make this data or similar available once the BPCI Advanced model ends.
After consideration of the public comments we received, we are finalizing our proposals for model overlap to allow overlap between shared savings or total cost of care model or program participants with TEAM episodes. This includes allowing both TEAM participant and total cost of care model or program participant to retain savings generated from their perspective performance in their model or program without recoupment.
In the proposed rule we stated that prior model experience has shown that it can be challenging for model participants to understand in real time whether a beneficiary's episode will be excluded, and we know that prior recoupment policies created friction between episode model participants and total cost of care model participants. We recognized the importance of coordination between a TEAM participant and total cost of care participant to ensure the beneficiary has continuous care moving beyond the structure of an episode. In order to accommodate a smooth transition for the aligned beneficiary, we considered, but did not propose there be a notification process required of the TEAM participant to ensure they are alerting the total cost of care participant of their aligned beneficiary's episode during the anchor hospitalization or anchor procedure. This notification process would allow the total cost of care participant the time to deploy their resources (for example, care coordination staff) and be prepared as the patient discharges from their anchor hospitalization or anchor procedure. However, we recognized that identifying beneficiaries aligned to a total cost of care participant may be challenging because it would require timely access to beneficiary alignment list for total cost of care participants and would increase burden to implement a notification process. We sought comment on ways to implement a notification process for shared savings or total cost of care participants that would be used to alert a shared savings or total cost of care participant that one of their aligned beneficiaries has initiated an episode in TEAM.
In the proposed rule we stated that total cost of care models (that is, ACOs) use their market's Health Information Exchange (HIE) to provide admission, discharge, and transfer (ADT) alerts. Others use less automated processes including fax or telephone to provide the alert. We recognized there is variation in the capabilities and sophistication of HIEs nationally and we recognized there is an increased administrative burden on participants when providing a telephonic or fax alert. Additionally, we recognized that there is variation in the timeframe in which these alerts can be issued based on the mechanism in which they are provided. We sought comment on what timeframe should be required to issue a notification and what process(es) should be used to provide a notification without causing undue burden on the TEAM participant, including both the processes cited previously or other processes not mentioned. We also sought comment on how broader use of ADT data exchange between TEAM participants and ACOs could improve care coordination, including any perceived barriers to better ADT exchange, and opportunities to improve ADT exchange, and how CMS could address these barriers and opportunities.
The following is a summary of comments and our responses to those comments received related to the notification process for shared savings or total cost of care model for which we sought comment.
Comment: A couple of commenters requested that TEAM participants be required to embed ACO beneficiary rosters into their electronic health records system to allow for better identification of aligned beneficiaries by the TEAM participant and to facilitate smoother communication between ACO and TEAM participant. One of these commenters recommended making this a requirement by the second year of TEAM, thus allowing time for the TEAM participant to fully integrate the ACO rosters into their electronic system after model launch.
Response: Although we acknowledge there would be benefits to a TEAM participant by having quick and immediate access to ACO alignment information in their EHR, making this a requirement could put undue financial and administrative burden on participants. In the final rule, we are not requiring TEAM participants to make any modifications or enhancements to their EHR; however, we encourage participants to consider how they can use their resources, including the data provided through TEAM participation, to enhance their ability to identify aligned beneficiaries.
Comment: We received a few comments supporting the concept of a beneficiary notification, but commenters asked that CMS hold the responsibility of contacting and notifying total cost of care or shared savings model participants rather than put the responsibility on the TEAM participant. A commenter mentioned that a required notification process is an administratively burdensome requirement that takes hospitals already limited resources away from more important patient care matters. A couple of other commenters mentioned that CMS has the greatest access to timely data and is already receives notifications from hospitals as part of eligibility checks and thus is best positioned to identify and provide the beneficiary notification.
Response: We appreciate receiving support for this policy and acknowledge the commenter's desire for CMS to own the responsibility of providing beneficiary notifications to total cost of care or shared savings model participants would alleviate administrative burden on TEAM participants. However, a key reason for requiring a beneficiary notification is to force connection and communication between TEAM participant and total cost of care or shared savings model. This forced connection means that information on the beneficiary is shared and both participants have an opportunity to collaborate on the beneficiary's immediate and long-term care. To have CMS stand as the intermediary and provide the notification takes away this required interaction, which could diminish the effectiveness of providing the notification and allowing for collaboration and coordination of the patient's care. As such, CMS will not take on the responsibility of providing the beneficiary notification to a total cost of care or shared savings model participant.
Comment: A commenter expressed concern over the time requirement of the notification and encouraged CMS to select a healthy time frame that ACOs and TEAM participants can comply with.
Response: The purpose in providing a beneficiary notification to a total cost of care or shared savings model participant is to ensure strong care coordination to ensure the best care for the beneficiary and allow the total cost of care or shared savings model participant the ability to deploy their own resources or be involved in the patient's care following discharge from the anchor hospitalization or anchor procedure. In order to effectively accomplish this, notification should occur by the time the patient discharges from their anchor hospitalization or anchor procedure to provide the patient with the warm hand off for ongoing care coordination.
Comment: A commenter who opposed the proposed requirement of beneficiary notification between a TEAM participant and total cost of care or shared savings model participant encouraging CMS to consider the extreme administrative burden it would place on participants.
Response: We acknowledge that providing a beneficiary notification to a total cost of care or shared savings participant adds a level of additional administrative effort on behalf of the ( print page 69790) TEAM participant, especially if the notification cannot be delivered electronically and requires a telephonic or fax notification. However, CMS believe the effort is outweighed by the benefit of alerting a total cost of care or shared savings model participant to which a beneficiary is aligned. The TEAM participant may benefit from leveraging their resources and support in ensuring the best outcomes for the patient.
Comment: We received some comments supporting the idea of a notification from a TEAM participant to a beneficiary's aligned total cost of care or shared savings model or program. Commenters mention that notifications such as ADT are relied upon to trigger a chain of action including timely post-discharge follow-up to reduce readmissions and decrease the number of patients going back to the emergency department unnecessarily.
Response: We appreciate the support received from commenters and agree that a beneficiary notification can serve to not only create opportunity for engagement between TEAM participant and shared savings or total cost of care participant, but also to create a smoother transition from episode back into longitudinal care and improve patient outcomes, such as avoiding unnecessary readmissions or emergency department visits.
Comment: We received a comment urging CMS to mandate that hospitals cannot participate in TEAM unless they provide admission, discharge, and transfer (ADT) notifications to community-based primary care physicians. This commenter states that the current lack of ADT notifications from hospitals is not a technological issue, but rather a behavioral issue that CMS could influence. They also state that ADT notifications are an essential part of ensuring appropriate transitions of care and are critical in helping ensure patients are able to have a longitudinal primary care relationship with their care team after an acute care episode, we urge CMS to mandate that hospitals cannot participate in TEAM unless they provide ADT notifications.
Response: We appreciate that this commenter considers the use of ADT notifications an essential part of care coordination and we agree that a beneficiary notification is an important step supporting and preparing for the patient's long-term care prior to leaving their anchor hospitalization or anchor procedure. We recognize there are many factors that influence the ability to transmit an ADT, including sophisticated HIE, market dynamics, hospital-specific resources, and technological capabilities, etc. However, the technology used to support a beneficiary notification is merely mean to assist in the action of providing the notification and should not deter a hospital from successfully making a notification to a beneficiary's aligned total cost of care or shared savings model or program.
Comment: A commenter in support of a beneficiary notification requested that CMS increase access to ADT alerting. This commenter specially mentioned that third-party vendors often offer notification services at a very high cost to ACOs and that although HIEs may be a more beneficial data source, they are not ever present or functional everywhere.
Response: CMS recognizes that there are many factors that impact how a hospital delivers a beneficiary notification to a total cost of care or shared savings model or program—whether the notification be electronic, by fax, telephonic, etc. We also acknowledge that there are costs associated by engaging a third-party vendor to support notification. CMS believes communication should be going on in value-based care and not be dependent on having a certain kind of technology. We view technology as merely an assist to support a beneficiary notification and do not consider technology or third-party vendors as a requirement to deliver a beneficiary notification to a total cost of care or shared savings model or program.
However, because we as an agency understand the need to improve the alerting process as it stands now, CMS has an outstanding Request for Information (RFI) where we are seeking feedback on the ADT process. We hope to use information gathered to identify where we can improve the alert process making it less burdensome and more useful for participants across models and programs.
We thank commenters for their input on beneficiary notifications and will address these comments, along with further proposals, in future notice and rulemaking.
We acknowledge there may be new models or programs that could have overlap with TEAM. This could occur because a beneficiary may trigger an episode in TEAM while being aligned to a new CMS model or program or because a TEAM participant also participates in another CMS model or program. We would plan to assess each new model to determine if the structure of payment and savings calculation are subject to the current proposed overlap policy or if there would be a need to bring forward any additional overlap requirements to account for the new model.
In the proposed rule we stated that consistent with President Biden's Executive Order 13985 on “Advancing Racial Equity and Support for Underserved Communities Through the Federal Government,” and Executive Order 14091 on “Further Advancing Racial Equity and Support for Underserved Communities Through the Federal Government,” CMS has made advancing health equity the first pillar in its Strategic Plan. [ 943 944 ] We define health equity as the attainment of the highest level of health for all people, where everyone has a fair and just opportunity to attain their optimal health regardless of race, ethnicity, disability, sexual orientation, gender identity, socioeconomic status, geography, preferred language, and other factors that affect access to care and health outcomes. We work to advance health equity by designing, implementing, and operationalizing policies and programs that support health for all the people served by our programs, eliminating avoidable differences in health outcomes experienced by people who are disadvantaged or underserved, and providing the care and support that our beneficiaries need to thrive. [ 945 ]
Disparities in access to surgical care by race/ethnicity, insurance status, income, and geography are well-documented, including disparities in the progression to surgery once surgical indication is determined and disparities in receipt of optimal surgical care. [ 946 ] Research has also highlighted disparities in readmissions rates following surgical intervention, indicating opportunities to tailor ( print page 69791) readmission-focused interventions to specific sites of care, such as safety net hospitals, to improve surgical outcomes. [ 947 948 ] For Medicare beneficiaries, higher health-related social need is also associated with a higher risk of complications, length of stay, 30-day readmission, and mortality following surgery. [ 949 ] Accordingly, there are opportunities to improve disparities in surgical outcomes by transforming infrastructure and care delivery processes, particularly for hospitals that serve higher proportions of historically underserved populations.
In this section, we discussed proposals for identifying safety net hospitals and rural hospitals within TEAM, and the associated flexibilities for TEAM participants meeting these definitions. We sought comment on the proposed safety net hospital and rural hospital definitions for TEAM, proposed model flexibilities for participants meeting each of these definitions, and the alternatives discussed.
In the proposed rule, we stated that a the goals of CMS's health equity pillar is to evaluate policies to determine how we can support safety net providers, partner with providers in underserved communities, and ensure care is accessible to those who need it. [ 950 ] There are also opportunities to engage more safety net providers in CMS Innovation Center models to increase the diversity of Medicare beneficiaries reached by models. [ 951 ] Although various approaches exist to identify “safety net providers,” this term is commonly used to refer to health care providers that furnish a substantial share of services to uninsured and low-income patients. [ 952 ] As such, safety net providers, including acute care hospitals, play a crucial role in the advancement of health equity by making essential services available to the uninsured, underinsured, and other populations that face barriers to accessing healthcare, including people from racial and ethnic minority groups, the LGBTQ+ community, rural communities, and members of other historically disadvantaged groups. Whether located in urban centers or geographically isolated rural areas, safety net hospitals are often the sole providers in their communities of specialized services such as burn and trauma units, neonatal care and inpatient psychiatric facilities. [ 953 ] They also frequently partner with local health departments and other institutions to sponsor programs that address homelessness, food insecurity and other social determinants of health, and offer culturally and linguistically appropriate care to their patients.
Because they serve many low-income and uninsured patients, safety net hospitals may experience greater financial challenges compared to other hospitals. Among the factors that negatively impact safety net hospital finances, MedPAC has pointed specifically to the greater share of patients insured by public programs, which MedPAC stated typically pay lower rates for the same services than commercial payers; the increased costs associated with treating low-income patients, whose conditions may be complicated by social determinants of health, such as homelessness and food insecurity, and the provision of higher levels of uncompensated care. [ 954 ]
In its June 2022 Report to Congress, MedPAC expressed concern over the financial position of safety net hospitals. [ 955 ] The Commission noted that the limited resources of many safety net hospitals may make it difficult for them to compete with other hospitals for labor and technology, and observed that “[t]his disadvantage, in turn, could lead to difficulty maintaining quality of care and even to hospital closure.” [ 956 ] Other research shows that the closure of a safety net hospital can have ripple effects within the community, making it more difficult for disadvantaged patients to access care and shifting uncompensated care costs onto neighboring facilities. [ 957 958 ]
Given the critical importance of safety net hospitals to the communities they serve, we considered different safety net hospital definitions to identify the best way to represent providers serving historically underserved populations in TEAM and/or provide flexibilities to those deemed as safety net providers. In the following section, we discuss multiple methodological options for identifying safety net providers in TEAM.
In the proposed rule, we stated that CMS Innovation Center's Strategy Refresh developed a definition of safety net providers to monitor the percent of safety net facilities participating in CMS Innovation Center models. The CMS Innovation Center's Strategy Refresh defined safety net hospitals as short-term hospitals and critical access hospitals (CAHs) that serve above a baseline threshold of beneficiaries with dual eligibility or Part D Low-Income Subsidy (LIS), as a proxy for low-income status. [ 959 ] Under the CMS Innovation Center's Strategy Refresh definition, hospitals are identified as safety net when their patient mix of beneficiaries with dual eligibility or Part D LIS exceeds the 75th percentile threshold for all congruent facilities who bill Medicare.
To calculate the hospital-level proportions of beneficiaries with dual eligibility and Part D LIS, a one-year or multiple-year retrospective baseline (for example, weighted three-year average) for each measure could be calculated for each TEAM participant. We would then determine the 75th percentile threshold for each measure separately based on the distribution of the two proportions (beneficiaries with dual eligibility or Part D LIS) for all PPS hospitals who bill ( print page 69792) Medicare. TEAM participants with proportions that meet or exceed the determined threshold for either dual eligibility or Part D LIS will be considered as a safety net hospital for the purposes of TEAM.
We considered that we could make safety net determinations based on the CMS Innovation Center's Strategy Refresh's definition using the described approach as of the model start date and hold the determinations constant for TEAM's duration. Alternatively, we considered calculating the hospital-level proportions of beneficiaries with dual eligibility and Part D LIS and the corresponding 75th percentile threshold for each measure annually, using a single year or rolling multiple-year weighted average of data from all PPS hospitals who bill Medicare. We could make redeterminations of safety net qualification under TEAM annually. This annual approach could mean that TEAM participants' safety net hospital qualifications could vary over the model's duration.
Another approach to identify safety net hospitals we considered was to use MedPAC's Safety Net Index (SNI), which is calculated as the sum of—(1) the share of the hospital's Medicare volume associated with low-income beneficiaries; (2) the share of its revenue spent on uncompensated care; and (3) an indicator of how dependent the hospital is on Medicare. MSNI is calculated at the hospital level using data from CMS cost reports for each hospital. [ 960 ]
For the share of the hospital's Medicare volume associated with low-income beneficiaries, MedPAC's definition of low-income beneficiaries includes all those who are dually eligible for full or partial Medicaid benefits, and those who do not qualify for Medicaid benefits in their states but who receive the Part D LIS because they have limited assets and an income below 150 percent of the Federal poverty level. Collectively, MedPAC refers to this population as “LIS beneficiaries” because those who receive full or partial Medicaid benefits are automatically eligible to receive the LIS. MedPAC states that its intent in defining low-income beneficiaries in this manner is to reduce the effect of variation in states' Medicaid policies on the share of beneficiaries whom MedPAC considers low-income, but to allow for appropriate variation across states based on the share of beneficiaries who are at or near the Federal poverty level. To calculate the LIS ratio for a hospital for a given fiscal year, we considered using the number of inpatient discharges of Medicare beneficiaries who are also LIS beneficiaries, using the most recent MedPAR claims for the discharge information, divided by the total number of inpatient discharges of Medicare beneficiaries.
For the share of a hospital's revenue spent on uncompensated care, we considered using the ratio of uncompensated care costs to total operating hospital revenue from the most recent available audited cost report data. [ 961 ] For further discussion on how this ratio could be calculated using audited cost report, please refer to 88 FR 26658 .
For the indicator of how dependent a hospital is on Medicare, MedPAC's recommendation is to use one-half of the Medicare share of total inpatient days. In calculating the Medicare share of total inpatient days for a hospital, we considered using the most recent available audited cost report data. For further information on how the numerator and denominator could be determined to calculate the indicator of how dependent a hospital is on Medicare from audited cost report data, please refer to 88 FR 26658 .
Using the sum of the three indicators as described, we considered that each TEAM participant could be assigned an SNI score, where a higher value means that a participant has either a high Medicare share of services, a high share of its Medicare patients with low incomes, and/or a high share of its revenue spent on uncompensated care.
To apply the Medicare Safety Net Index (MSNI) to identify safety net hospital participants in TEAM, we considered calculating the MSNI for TEAM participants using a one-year or multiple-year baseline period (for example, a three-year average). We considered setting a threshold to identify safety net providers with TEAM based on the distribution of scores for all PPS hospitals that bill Medicare (for example, providers with scores in the 75th percentile of SNI scores could be considered safety net providers). We considered making safety net determinations based on the described approach as of the model start date and hold the determinations constant for TEAM's duration. Alternatively, we considered calculating the SNI and corresponding threshold annually using a one-year or multiple-year moving average and make redeterminations of safety net designations annually. This annual approach could mean that TEAM participant safety net qualifications for TEAM could vary over the model's duration.
In the proposed rule, we stated that an approach to identifying safety net hospitals could be to use area-level indices. This approach could potentially better target policies to address the social determinants of health as well as address the lack of community resources that may increase risk of poor health outcomes and risk of disease in the population. In a recent environmental scan, the Office of the Assistant Secretary for Planning and Evaluation (ASPE) suggested that an area-level index could be used to prioritize communities for funding and other assistance to improve social determinants of health (SDOH)—such as affordable housing, availability of food stores, and transportation infrastructure. Although ASPE concluded that none of the existing area-level indices identified in the environmental scan were ideal, they concluded that the area deprivation index (ADI) was one of the best available choices when selecting an index for addressing health-related social needs or social determinants of health. [ 962 ]
The ADI was developed through research supported by the National Institutes of Health (NIH) with the goal of quantifying and comparing social disadvantage across geographic neighborhoods. It is a composite measure derived through a combination of 17 input variables—including measures of income, education, employment, and housing quality—from the American Community Survey (ACS) 5-year estimate datasets. [ 963 ] Each neighborhood is assigned an ADI value from 1 to 100 (corresponding to percentile), where a higher value means that a neighborhood is more deprived. The ADI measure is intended to capture local socioeconomic factors correlated with medical disparities and ( print page 69793) underservice. Several peer reviewed research studies demonstrate that neighborhood-level factors for those residing in disadvantaged neighborhoods also have a relationship to worse health outcomes for these residents. [ 964 965 966 ]
Medicare already uses ADI to assess underserved beneficiary populations in the Shared Savings Program, and ADI is also used in CMS Innovation Center models. In the CY 2023 PFS final rule, CMS adopted a policy to provide eligible Accountable Care Organizations (ACOs) with an option to receive advanced investment payments ( 87 FR 69778 ). Advance investment payments are intended to encourage low-revenue ACOs that are inexperienced with risk to participate in the Shared Savings Program and to provide additional resources to such ACOs in order to support care improvement for underserved beneficiaries ( 87 FR 69845 through 69849 ). The risk-factors based (using ADI) scores assigned to the beneficiaries assigned to the ACO form the basis for determining the quarterly advanced investment payment to the ACO. For additional detail, please see the quarterly payment amount calculation methodology at 42 CFR 425.630(f)(2) .
To use ADI to identify safety net hospitals for TEAM, we considered assigning episodes an ADI value based on the beneficiary's address found in the Common Medicare Environment (CME) file. Episodes meeting an established national ADI percentile threshold (for example, ADI 80) could be classified as high-ADI episodes, and a distribution of the proportion of high-ADI episodes could be constructed. We considered that those TEAM participants that fell above an established threshold of high-ADI episodes (for example, 75th percentile) could be classified as safety net hospitals. For PY 1, the proportion of high-ADI episodes and its corresponding distribution could be determined based on a single-year or multiple-year retrospective baseline (for example, three-year average). Those TEAM participants that met or exceeded the determined threshold would be designated as safety net. We could hold these designations constant for TEAM's duration or recalculate the proportion of high-ADI episodes annually (using a one-year or multiple-year moving average) and make safety net redeterminations based on an updated threshold on an annual basis. This annual approach could mean that TEAM participants' safety net qualifications for TEAM could vary over the model's duration.
We considered the previously mentioned methods for identifying safety net hospitals and we proposed to use the CMS Innovation Center's Strategy Refresh definition for identifying safety net hospitals within TEAM. Use of the CMS Innovation Center's Strategy Refresh's safety net definition allows for a consistent and streamlined approach to how the CMS Innovation Center plans to monitor safety net participation with CMS Innovation Center models. Further, the definition uses two recognized measures of social risk to identify hospitals serving a higher proportion of beneficiaries that may face barriers to receiving or accessing care.
Beneficiaries with dual eligibility are considered a vulnerable group for several reasons including the nature of dual eligibility requirements, a higher proclivity for experiencing chronic conditions, and an increased likelihood of mental health diagnosis. [ 967 968 ] In its 2016 “Report to Congress Social Risk Factors and Performance Under Medicare's Value-Based Purchasing Programs,” the Office of the Assistant Secretary for Planning and Evaluation (ASPE) found that dual eligibility status was the strongest predictor of poor outcomes of quality measures among multiple social risk factors examined. [ 969 ] TEAM's proposed approach to identify safety net hospitals is also similar to other approaches used in CMS Innovation Center models. For example, BPCI Advanced identifies safety net hospitals by tabulating the proportion of episodes with fully or partially dual eligible beneficiaries; if a hospital exceeded a 60 percent threshold of episodes based on the previous model year, then they would be considered a safety net hospital. [ 970 ]
While dual eligibility status does not fully capture all aspects of social risk, the incorporation of the proportion of patients with Part D LIS as a proxy for income into TEAM's proposed safety net definition broadens the range of possible beneficiary social risk factors used to make safety net hospital designations under the model. In its 2017 report on “Accounting for Social Risk Factors in Medicare Payment,” the National Academies found that accounting for dual eligibility alone may not be sufficient to capture all social risk factors, and the incorporation of multiple measures may help to better characterize overall social risk. [ 971 ] We sought comment on our proposal to identify safety net hospitals using the CMS Innovation Center's Strategy Refresh's definition in TEAM at § 512.505.
The following is a summary of the public comments received on the proposed definition of safety net hospitals in TEAM and our responses to these comments:
Comment: A commenter recommended CMS should use its authority to create a federal designation of essential health systems to target funding and other support across CMS programs, rather than creating a safety net hospital definition limited to TEAM.
Response: The proposed definition of safety net hospital, as defined and finalized in this section of the preamble of the final rule, is specific to the purposes of TEAM. Creating a standard federal designation for essential health system is not within the scope of this rule.
Comment: A commenter supported the use of the Hospital Value Based Purchasing Program's health equity adjustment (HEA) methodology to acknowledge socioeconomic inequities that differentially affect hospitals, especially safety net hospitals, by assigning additional points to hospitals that treat a greater proportion of patients who are dually eligible for Medicare and Medicaid and recommend TEAM to consider a similar approach. ( print page 69794)
Response: We thank the commenter for their suggestion and agree that the approach aims to rewards hospitals that serve higher proportions of dual-eligible patients for providing excellent care. TEAM recognizes safety net hospitals may need additional policies to protect them from significant financial risk given they typically have less resources and care for a higher proportion of underserved beneficiaries. TEAM includes provisions that allow safety net hospitals to participate in value-based care, with lower risks and rewards. In particular, TEAM will allow safety net hospitals to participate in Track 1 for the first three performance years of the model, as discussed in section X.A.3.a.(3) of the preamble of this final rule, which removes their exposure to downside risk. TEAM also recognizes differences in quality measure performance for safety net hospitals, and other hospitals that may elect to participate in Track 2, by adjusting their CQS adjustment percentage for negative reconciliation amounts by 15 percent, which further limits their financial risk, as discussed in section X.A.3.d.(5)(g) of the preamble of this final rule. We will take into consideration a health equity adjustment approach, and if warranted, would propose in future notice and comment rulemaking.
Comment: A few commenters supported the proposed definition of safety net hospitals using the CMS Innovation Center's Strategy Refresh's definition. A commenter supported the use of the CMS Innovation Center's definition because CMS conducted extensive stakeholder roundtables on the safety net definition, which led the CMS Innovation Center to use the Medicare Part D LIS indicator and dual-eligibility ratio. A few commenters noted their appreciation for TEAM's safety net definition because it recognizes the challenges of providing care to low-income Medicare beneficiaries. Several commenters noted that TEAM's proposed definition for safety net hospital would not fully capture the full range of hospitals within the safety net as it does not reflect data from all payers or the degree of patients without health insurance served by a hospital. A commenter suggested that a broader definition of safety net that more closely aligns with community need would be more appropriate to identify hospitals that care for the most vulnerable populations. A commenter suggested that the proposed TEAM definition would prioritize smaller hospitals and would miss several large essential hospitals that have long played a safety net role in their communities. A commenter noted that dual eligibility and qualification for the Part D LIS subsidy may be highly correlated, which may affect a hospital's qualification as a safety net participant under TEAM.
Response: We thank the commenters for sharing their support and concerns regarding TEAM's proposed safety net definition. We recognize that there are multiple approaches to identifying a safety net hospital for TEAM. The CMS Innovation Center definition reflects one measure of a hospital's patient mix through use of the percentage of beneficiaries that are dually eligible, and a proxy measure for the degree of low-income beneficiaries that are furnished services at a given facility. As discussed in this section of the preamble of the final rule, these measures have been shown to be associated with lower access to care and worse health outcomes. The use of the CMS Innovation Center safety net definition within TEAM would align with the broader use of the safety net definition in monitoring safety net participation across CMS Innovation Center models.
We acknowledge that measurement of community need could provide insights into the characteristics of a service area of a given hospital; however, the lack of readily available standardized data on community need across all possible TEAM participants beyond area-based indices could pose a challenge to incorporating the concept of community need into TEAM's safety net hospital definition.
To clarify a commenter's concern about the possible high degree of correlation between the two measures used in the proposed safety net definition under TEAM, the proposed safety net definition would allow a TEAM participant to qualify as a safety net hospital under TEAM should it exceed the 75th percentile of either measure based on the distribution of these measures from all hospitals billing Medicare.
Comment: A few commenters recommended against use of a safety net definition for TEAM that uses area-level indices, such as the ADI, due to shortcomings in measure design. A commenter advised against ADI as it does not capture patient-level social risk factors and only measures social risk factor data at the geographic level, specifically the characteristics of the hospital's geographic location. A commenter noted that an area-based index using a hospital's geographic location may not be a good proxy for determining whether a TEAM hospital should be considered a safety net hospital because patients may be transient. A commenter stated that while neighborhood factors are important determinants of health outcomes and spending, the lack of standardization in calculating the ADI score has made the measure overly dependent on median housing value and may disadvantage certain neighborhoods in large urban areas.
Response: We thank commenters for raising concerns of using ADI as a possible way to define a safety net hospital under TEAM. We recognize that ADI as an area-based index has several valid uses for identifying a geographic measure of neighborhood disadvantage at the census block group level. We are aware of potential concerns that have been raised how the lack of standardization of ADI variables may make the ADI primarily a function of a subset of variables included in calculation of the ADI. [ 972 ] Due to the concerns raised by commenters, we are not finalizing the use of ADI in determining safety net status for TEAM participants at this time as we feel use of the CMS Innovation Center's safety net provider definition is most appropriate for reasons discussed throughout this section. However, in response to commenters, we will assess the use of standardization in calculating ADI for target price risk adjustment purposes and may propose updates to our risk adjustment methodology in future rulemaking.
Comment: A couple commenters recommended against use of the MSNI as it favors hospitals with high Medicare volume and does not incorporate Medicaid volume into its formula. A commenter noted that MSNI favors hospitals with higher Medicare volume because of the weight the Medicare share of inpatient days has in the MSNI formula. A commenter noted that use of the MSNI could discourage hospitals from expanding access to care as hospitals that serve more Medicaid beneficiaries and patients without health insurance will have a lower share of Medicare inpatient days even if they continue to serve the same number of Medicare beneficiaries. A commenter also noted that neither the MSNI nor the ADI would accurately reflect the broad patient mix of safety net hospitals if used in TEAM's safety net definition.
Response: We thank commenters for raising concerns about the potential use of MSNI in determining safety net status under TEAM. Due to the concerns ( print page 69795) raised by commenters, we are not finalizing the use of MSNI in determining safety net status for TEAM participants at this time as we feel use of the CMS Innovation Center's safety net provider definition is most appropriate for reasons discussed throughout this section.
Comment: Many commenters found that the proposed TEAM definition does not account for the degree to which a hospital serves Medicaid beneficiaries and patients without health insurance. Several commenters suggested that the safety net definition under TEAM should account for the degree to which a hospital serves Medicaid beneficiaries. A commenter stated that the proposed TEAM safety net definition would adequately account for care provided to low-income Medicare beneficiaries but that low Medicare beneficiary volumes should not be a barrier to qualify as a safety net under TEAM for hospitals that otherwise serve large low-income populations. A few commenters noted that the proposed definition does not account for the financial difficulties of hospitals that treat a high number of Medicaid-eligible patients but may not have a relatively high volume of Medicare and Medicaid dually eligible patients. A commenter noted that a hospital's overall payer mix would be more useful in identifying facility-level characteristics that would influence the hospital's ability to fund infrastructure and investments for value-based care arrangements.
Response: We thank commenters for their recommendations on potentially incorporating measures of the degree to which a TEAM participant serves Medicaid beneficiaries or patients without health insurance in determining safety net status. We acknowledge that this type of data could provide a more comprehensive view of the payer mix of a given hospital. However, as we do not have access to this type of standardized data for all possible TEAM participants, it would be challenging to incorporate both measures as part of TEAM's safety net definition at this time. The CMS Innovation Center safety net definition reflects one measure of a hospital's patient mix through use of the percentage of beneficiaries that are dually eligible, and a proxy measure for the degree of low-income beneficiaries that are furnished services at a given facility.
Comment: Some commenters recommended that TEAM's safety net definition should consider the degree of uncompensated care provided by a hospital. These commenters suggested several existing measures related to uncompensated care that could be used in TEAM's safety net definition. A few commenters recommended that the disproportionate patient percentage (DPP), which captures a hospital's proportion of Medicaid and low-income Medicare patients, would be an appropriate measure of uncompensated care as it has long been used in Medicare's Disproportionate Share Hospital (DSH) program. A couple commenters suggested use of the Medicare uncompensated care payment factor (UCPF), which is a measure of a hospital's share of uncompensated care costs relative to all hospitals' uncompensated costs, as it can be used to identify the costs of care delivered to uninsured individuals. A few commenters recommended use of the deemed DSH hospital designation as it could identify hospitals that are statutorily required to receive Medicaid DSH payments because they serve a high share of Medicaid and low-income patients. A commenter expressed that the TEAM safety net definition could continue to use the Medicare-Medicaid dual eligibility and Part D LIS criteria but should add uncompensated care as a percentage of a hospital's total costs as a third eligibility criterion.
Response: We thank commenters for their suggestions on the range of measures related to DSH and uncompensated care payments that could be used in TEAM's safety net definition. In its June 2022 Report, MedPAC raised concerns about whether these payments appropriately target safety net hospitals. [ 973 ] We do not feel that it would be appropriate to use measures related to DSH or uncompensated care payments in TEAM's safety net definition as we feel that the CMS Innovation Center Strategy safety net definition use of dual eligibility and Part D LIS eligibility criteria is most appropriate in identifying TEAM participants as safety net participants. As discussed in this section, these two criteria have been shown to be associated with lower access to care and worse health outcomes.
Comment: A commenter recommended that participation in the 340B Program be considered a criterion for designating TEAM participants as safety net hospitals.
Response: Section 340B of the Public Health Service Act (340B) allows participating hospitals and other providers to purchase certain covered outpatient drugs or biologicals from manufacturers at discounted prices. We feel that using the HRSA-administrated 340B Drug Pricing Program participation alone would be insufficient to identify safety net hospitals under TEAM. Only certain types of hospitals are eligible to be covered entities in the 340B Drug Pricing Program. [ 974 ] Therefore, using 340B Drug Pricing Program eligibility for determining safety net hospital status under TEAM could restrict the types of eligible TEAM participants that could be considered safety net for the purposes of TEAM. The 340B Program also focuses on the purchasing of certain covered outpatient drugs or biologicals, which is not fully aligned with the inpatient focus of TEAM episodes at this time. Use of the CMS Innovation Center safety net definition would allow all TEAM participants to be considered for safety net hospital eligibility for the purposes of TEAM.
Comment: A few commenters requested that safety net determinations for TEAM be done at the beginning of the model and be held constant for the duration of the model. A couple commenters highlighted that participants require time for financial planning within the context of a model that creates financial uncertainty for participating providers, highlighting that a shift between tracks for eligible safety net TEAM participants could undermine the participant's success in the model.
Response: We thank the commenters for their perspectives on when safety net determinations should be made and whether they should be held for the duration of the model. We acknowledge the potential challenges that changing safety net determinations in each model year could create in a TEAM participant's ability to adequately plan for the model and that having a consistent designation for model's performance period may be beneficial. However, we do not believe holding the safety net determination for the duration of the model would create a sustainable, long-term policy, especially if TEAM could meet criteria to be expanded, as permitted under section 1115A(c) of the Act. Further, holding safety net determinations constant limits a TEAM participant's access to participating in different participation tracks in TEAM. As discussed in section X.A.3.a.(3) of ( print page 69796) the preamble of this final rule and finalized at § 512.520(b)(3) and (4), TEAM participants must satisfy the definition of safety net hospital at the time of participation track request for participation in Track 1 or Track 2.
After consideration of public comments, we received, we are finalizing as proposed our proposed definition of safety net hospital under TEAM at § 512.505.
Americans who live in rural areas of the nation make up about 20 percent of the United States (U.S.) population, and they often experience shorter life expectancy, higher all-cause mortality, higher rates of poverty, fewer local doctors, and greater distances to travel to see health care providers, compared to their urban and suburban counterparts. [ 975 ] The health care inequities that many rural Americans face raise serious concerns that the trend for poor health care access and worse outcomes overall in rural areas will continue unless the potential causes of such health care inequities are addressed. Barriers such as workforce shortages can impact health care access in rural communities and can lead to unmet health needs, delays in receiving appropriate care, inability to get preventive services, financial burdens, and preventable hospitalizations. [ 976 ]
Hospitals in rural areas often face other unique challenges. Rural hospitals may be the only source of healthcare services for beneficiaries living in rural areas, and beneficiaries have limited alternatives. Rural hospitals may also be in areas with fewer providers including fewer physicians and PAC facilities, rural hospitals may have more limited options in coordinating care and reducing spending while maintain quality of care under a value-based care arrangement. We believe that urban hospitals may not have similar concerns as they are often in areas with many other providers and have greater opportunity to develop efficiencies.
We did not propose to include any geographically rural areas for TEAM based on the proposed CBSAs as defined in 89 FR 36394 through 36412 . However, some hospitals in the proposed CBSAs for TEAM may be considered rural for other reasons, such as being reclassified as rural under the Medicare wage index regulations or being designated a rural referral center (RRC).
For the purposes of TEAM, we proposed a rural hospital to mean an IPPS hospital that is located in a rural area as defined under § 412.64 of this chapter; is located in a rural census tract defined under § 412.103(a)(1) of this chapter; has reclassified as a rural hospital under § 412.103 of this chapter, or is designated a rural referral center (RRC) under § 412.96 of this chapter. This definition would be an expanded version of the rural hospital definition used by the CJR model as defined in 42 CFR 510 .
For PY 1, we proposed that rural designations under TEAM would be based on the TEAM participant's rural classification as of the model start date. We recognized that rural designations and rural reclassification requests in accordance with § 412.103 may occur over on a rolling basis over the course of the model and can take several months to be reviewed and approved by CMS. We proposed that TEAM participants that receive an approved rural designation under the criteria defined in the preceding paragraph or an approved rural reclassification in accordance with § 412.103 must notify CMS at least 60 calendar days prior to the start of a model's performance year for CMS to consider classifying the TEAM participant as rural under the model for the following performance year. We proposed that model rural designations will occur only once at the beginning of each model performance year regardless of when a TEAM participant's rural classification may change within a given performance year.
We proposed that if a TEAM participant's classification is no longer rural pursuant to § 412.103 or any other criteria previously qualifying them as rural as defined earlier in this section, the TEAM participant must notify CMS in a manner chosen by CMS within 60 calendar days of receipt of this designation change. We proposed that TEAM participants would continue to receive the flexibilities for rural hospitals as described in 89 FR 36392 through 36394 through the remainder of the performance year in which the redesignation occurs, but the TEAM participant would no longer qualify for rural hospital flexibilities at the start of the next performance year.
We sought comment on our proposal to identify rural hospitals in this section. We did not propose to include a measure of hospital rurality within our risk adjustment model as described in 89 FR 36433 through 36435 but sought comments on whether inclusion of this risk adjustor would be warranted.
The following is a summary of public comments received on the proposed definition of a rural hospital under TEAM and our responses to these comments:
Comment: MedPAC noted that the proposed definition would encompass a large share of hospitals and recommended that rural hospitals should be defined as those located in geographically rural areas and not those that have been reclassified as rural or RRCs. MedPAC commented that nearly one-third of hospitals have gone through rural reclassifications and that TEAM should avoid a rural definition that could fuel reclassifications and should instead focus on a geographically based rural definition.
Response: We thank MedPAC for noting the large share of hospitals that would be included in the definition. Our proposed inclusion of hospitals that were reclassified as a rural hospital under § 412.103 of this chapter or is designated a rural referral center (RRC) under § 412.96 of this chapter in TEAM's rural definition was to consider a broader set of hospitals that are considered rural and to align with the rural definition used under the CJR model ( 42 CFR 510 ). Consistent use of a rural definition across CMS Innovation Center models and CMS programs can potentially provide continuity in a participant's rural classification across models and programs. However, in the context of a mandatory model, we understand that a narrower and rural definition based strictly on geographic area could prevent creating an incentive for a hospital to seek rural reclassification given the flexibilities offered to rural hospitals under TEAM.
Comment: A commenter recommended that CMS not reclassify hospitals as rural during the middle of TEAM's performance period as changes in rural classifications were viewed as a challenge in the BPCI Advanced model.
Response: We thank the commenters for their concern regarding the potential challenges of changing rural classifications over the model performance period. We acknowledge that determining a participant's rural status under TEAM at the beginning of the model performance period and maintaining it throughout the model performance period may provide a hospital with an understanding of their model track for the duration of the model, giving them the ability to plan accordingly. However, we do not ( print page 69797) believe holding the rural hospital classification for the duration of the model would create a sustainable, long-term policy, especially if TEAM could meet criteria to be expanded, as permitted under section 1115A(c) of the Act. Further, holding rural hospital classifications constant may limit a TEAM participant's access to participating in different participation tracks in TEAM, specifically Track 2. As discussed in section X.A.3.a.(3) of the preamble of this final rule and finalized at § 512.520(b)(3) and (4), TEAM participants must satisfy the definition of rural hospital at the time of participation track request for participation in Track 2.
Comment: A commenter suggested that some hospitals may be just outside an applicable rural zone and that CMS should considering a policy that would allow these hospitals to request a change in their designation.
Response: We understand that a hospital may wish to request a change in their rural designation under TEAM. Any definition chosen will have participants on the margin of the definition. The rural definition under TEAM must be applied consistently across all TEAM participants and allowing for such change requests could increase operational complexity of the model and would not allow for a consistent and standard definition of rurality to be applied across all TEAM participants.
After consideration of public comments, we received, we are finalizing our proposed definition of rural hospital under TEAM with slight modification to remove hospitals that have reclassified as a rural hospital under § 412.103 and hospitals that are a rural referral center (RRC) as given this term under § 412.96. For the purposes of TEAM, a rural hospital means an IPPS hospital that is located in a rural area as defined under § 412.64 of this chapter or is located in a rural census tract defined under § 412.103(a)(1) of this chapter as defined at § 512.505.
In recent years there has been a push for Medicare and other payers to include beneficiary social risk adjustment into financial methodologies that determine health care payments. [ 977 ] It is believed that the inclusion of beneficiary social risk adjustment may provide more resources to providers who care for underserved beneficiaries to offset the additional costs often attributed to SDOH. In other words, patients with limited resources or access to care may require more spending from providers to achieve equitable outcomes. Beneficiary social risk adjustment has been limited in previous episode-based payment models. The BPCI Advanced and CJR models included beneficiary social risk adjustment for beneficiary dual eligibility status, yet that single adjuster alone may not be sufficient in capturing spending differences for beneficiary social risk. Findings from the CJR model's 5th Annual Report found that, during the baseline period, historically underserved populations generally had higher episode payments, used more institutional post-acute care, had higher rates of emergency department use and readmissions, and received elective LEJRs at a lower rate than their reference populations. [ 978 ]
There is significant literature and research surrounding the inclusion of social risk adjustment in health care payments, especially given the varying social risk adjustment indicators available. [ 979 980 981 ] In a recent environmental scan, ASPE indicated that area-level deprivation indices tend to have the broadest coverage across the entire range of social risk factors. According to ASPE's report, area-level deprivation indices are, by definition, measured for geographic areas, which presents challenges in including them in payment models because a provider's patients are unlikely to be representative of the population of the geographic area in which the provider is located. [ 982 ]
Several CMS Innovation Center initiatives incorporate (or may incorporate) beneficiary social risk adjustment into their financial calculations or determining payment amounts, including the ACO REACH model, the Enhancing Oncology Model (EOM), the Making Care Primary (MCP) model, and the Guiding an Improved Dementia Experience (GUIDE) model. To avoid relying on a single indicator that may not be representative of the beneficiaries a provider cares for, these models incorporate multiple social risk indicators. Specifically, these models take into account one or more of the following indicators in their risk adjustment models: state and national ADI, Medicare Part D Low-Income Subsidy (LIS), and dually eligible beneficiaries enrolled in both Medicare and Medicaid. Factoring in multiple indices may avoid challenges when an underserved beneficiary lives in higher cost-of-care area or beneficiaries that have difficulty accessing care. For example, incorporating both state and national ADI allows the for the risk adjustment model to capture national and local socioeconomic factors correlated with medical disparities and underservice, while including the LIS measure will capture socioeconomic challenges that could affect a beneficiary's ability to access care. For these reasons, and to align with other CMS Innovation Center models, we proposed to incorporate and equally weight three social risk indicators in TEAM's target price methodology, see 89 FR 36433 through 36435 , specifically state and national ADI indicators, the Medicare Part D LIS indicator, and dual-eligibility status for Medicare and Medicaid. We believe that including these social risk indicators would ensure TEAM participants that serve disproportionately high numbers of underserved beneficiaries are not inadvertently penalized when setting TEAM target prices.
We sought comment on the proposed beneficiary social risk adjusters for TEAM and whether there were beneficiary social risk indicators we should consider in TEAM's target price methodology.
The following is a summary of public comments received on the beneficiary social risk adjustment indicators under TEAM and our responses to these comments. For further comments and responses related to the risk adjustment methodology, please see section ( print page 69798) X.A.3.d.(4) of the preamble of this final rule.
Comment: A few commenters supported the inclusion of social risk adjustment in the model's target pricing methodology. A commenter noted that inclusion of these adjustments can ensure that facilities that disproportionately serve underserved populations, such as communities of color and rural communities, are not penalized under the model given their higher investment needs due to higher levels of social risk. A commenter stated that failing to adjust for social risk variables could unfairly penalize hospitals and clinicians for serving more complex and underserved populations, and that adjusting for these factors could ensure a more accurate and fair assessment of quality. A commenter noted that the current proposal to use dual eligibility, Part D LIS status, or living in an area with a high ADI would not appropriately demonstrate a patient's social risk. A commenter recommended that TEAM take a cautious approach to social risk adjustment to ensure beneficiary needs are not excessively adjusted and potentially masked.
Response: We thank commenters for their support about the inclusion of social risk adjustors into TEAM's target price methodology and commenters for raising potential concerns about inclusions of such variables. In the proposed rule, we noted that the social risk adjustment variable was chosen so that it can account for multiple potential markers of beneficiary social risk. Using Medicare/Medicaid dual eligibility status, LIS status, and living in areas in the top percentiles of either the national or state level ADI allows CMS to utilize existing indicators of social risk together and capture safety-net populations through multiple means. If dual-eligibility status has not been identified prior to the episode occurring, the ADI marker may still be able to identify the beneficiary at a higher social risk. Additionally, we proposed to enforce sign restrictions to avoid negative coefficients for the beneficiary social risk adjuster, meaning that the adjustment to the preliminary or reconciliation target prices would only happen if the coefficient on the beneficiary social risk adjustment variable is positive. We would not be able to enforce the sign restrictions if additional variables for social risk were separately added to the model.
As indicated above in section X.A.3.d.(4) of this final rule, CMS is also finalizing the addition of safety net status of the hospital as a risk-adjuster to all episode types to address concerns about providers that primarily care for beneficiaries with dual-eligibility or LIS status. We believe the inclusion of the provider's safety net status will strengthen the risk adjustment model and appropriately set target prices for providers that serve economically distressed counties. While CMS is not including all the risk adjusters tested in BPCI Advanced to maintain the simplicity required for hospitals without experience in value-based care to participate in TEAM, the updated risk adjustment model does take more patient-level and hospital level factors into account.
Comment: A couple commenters suggested that rural status should be considered as a risk adjustor to support rural providers in taking on risk and to acknowledge the disparities that exist in rural settings.
Response: We acknowledge the comments regarding the inclusion of provider-level risk adjusters for rural/urban status. As part of the Lasso regression analysis to identify risk adjustors as described in section X.A.3.d.(4) of the preamble of this final rule, CMS found that the variables for patients treated at rural hospitals were not selected by the Lasso model for any of the episode categories. Therefore, risk adjusters for rural status will not be included. However, rural hospitals will have additional flexibilities in TEAM, such as opting for Track 2 of the model which has a lower level of risk sharing (5 percent stop-loss/stop-gain).
Comment: Each of the following sociodemographic variables were proposed by a commenter: age, disability status, educational level, preferred language, and socioeconomic status. A couple commenters recommended including patient-level health-related social need (HRNS) variables for risk adjustment such as housing instability, experiencing homelessness, food insecurity, financial needs, transportation problems, and interpersonal safety. A couple commenters suggested that Z codes related to social determinants of health (SDOH) and HRSNs could be used for risk adjustment purposes.
Response: We thank commenters for their suggestions on additional measures that could be considered to risk adjust for a beneficiary's levels of social risk, including the potential use of Z codes to identify SDOH-related variables.
As described in section X.A.3.d.(4) in the preamble of this final rule, the updated risk adjustment methodology for TEAM includes age brackets as a demographic variable for all TEAM episodes. We also note that the updated risk adjustment methodology will incorporate a measure of disability as the original reasons for Medicare enrollment for the LEJR episode as it was found to be statistically significant in the Lasso regression analysis. We recognize that there are additional measures of disability status that could be considered in exploration of future risk adjustment methodologies under TEAM should standardized and sufficient data be available across TEAM participants. Based on the results of the Lasso regression analysis to identify risk adjustors for TEAM episodes, we are not considering any other demographic factors—such as educational level, preferred language, and socioeconomic status—at this time but could consider them in future analyses that may lead to proposed adjustments in TEAM's risk adjustment methodology prior to the start of TEAM's performance period.
We appreciate commenters recommendations on additional variables related to HRSNs that could be considered in the risk adjustment methodology. Given variability in the use of Z codes to capture HRSN data, we would be concerned about the availability of standardized data across TEAM beneficiaries to meaningfully incorporate such measures into TEAM's risk adjustment methodology. The proposed beneficiary-level social risk variable included in TEAM's risk adjustment variable incorporates measures that have been shown to be correlated with a beneficiary's level of social risk as described in section X.A.3.d.(4) in the preamble of this final rule. In the absence of consistently available HRSN data at the beneficiary-level, we consider that the beneficiary-level social risk variable captures the degree of beneficiary's social risk and avoids potential overcontrolling of social risk variables within the risk adjustment models. We could consider further exploration of the recommended HRSN-related risk adjustors should TEAM adjust its risk adjustment methodology through notice and comment rulemaking.
Comment: A commenter recommended to continue using the BPCI Advanced and CJR risk adjustment methodology but to include LIS status and ADI to more accurately capture the social risk experienced by a beneficiary in TEAM's risk adjustment methodology.
Response: We appreciate the commenter's suggestion on expanding upon existing risk adjustment methodologies to incorporate measures of social risk. The updated risk adjustment methodology as described in section X.A.3.d.(4) in the preamble of ( print page 69799) this final rule more closely resembles the CJR and BPCI Advanced models with the additional risk adjusters that were selected based on statistical analyses and maintains the goal of a simple risk adjustment model for TEAM.
Comment: A commenter cautioned CMS in using the ADI as a measure of social risk as research has demonstrated that ADI is weakly correlated with self-reported social needs and with health care costs and may mask inequities in communities where there are high levels of wealth disparities. Another commenter recommended that CMS develop guardrails so that hospitals that receive higher reimbursement because of ADI factors should be required to make their services more accessible to Medicaid and dually eligible enrollees. A commenter suggested that beneficiary-level social risk factors should be used to better capture beneficiary-level disadvantage since beneficiaries living in high ADI area could have considerable social risk. A commenter suggested that use of ADI in risk adjustment could cause harm to providers in high-cost living areas by boosting the risk scores of healthy beneficiaries in low-cost living areas. A commenter supported the use of ADI as it creates a multi-dimensional picture of the social drivers of health within a community but may mask differences within a census block. A couple of commenters recommended CMS to consider using patient-level data in combination with similar indices to the ADI, like the Centers for Disease Control and Prevention's (CDC) Social Vulnerability Index (SVI), which includes data on race, ethnicity, and disability.
Response: We thank commenters for raising potential concerns around the use of ADI as one variable to determine the beneficiary social risk variable in risk adjustment. One benefit of ADI as a measure of social risk is that it measures several factors of socioeconomic position across the domains of education, income, home values, employment, and household information. The geographic level of an area-based index or indicator will inherently be a limitation in the use of any area-based measure, including potentially masking differences below the geographic unit of analysis. We believe ADI's use of census blocks groups provides an appropriate unit of geography by which to assess social risk for the purposes of risk adjustment under TEAM. We acknowledge that more granular beneficiary-level data on HRSNs could theoretically provide a more accurate assessment of an individual beneficiary's level of social risk compared to an area-based index; however, because HRSN data may be inconsistently available at the beneficiary level, we do not think that such adjustors would be appropriate to use for risk adjustment under TEAM at this time. We are aware of potential concerns that have been raised how the lack of standardization of ADI variables may make the ADI primarily a function of a subset of variables included in calculation of the ADI. [ 983 ] While we think that the use of ADI is appropriate as one way to capture a beneficiary's level of social, we will continue to explore whether standardization of the ADI variables would be appropriate for the purposes of TEAM's risk adjustment approach and would propose any such changes in future rulemaking.
As described in section X.A.3.d.(4) in the preamble of this final rule, we also note that the construction of the beneficiary social risk variable in TEAM's risk adjustment methodology represents the union of three different potential markers of beneficiary social risk: national-level ADI, state-level ADI, and dual eligibility status. While we acknowledge concerns that the ADI could mask differences in levels of deprivation lower than the census block, we believe that the use of national- and state-level ADIs would help mitigate potential concerns on the validity of the measure of social risk as it incorporates relative measures of deprivation at national scale and within a given state. Similarly, as dual eligibility status has been shown to be associated with a beneficiary's level of social risk, we feel that allowing three ways in which a beneficiary's level of social risk could be accounted for in risk adjustment is appropriate.
We disagree with the recommendation that TEAM participants that receive higher payment because of ADI factors alone should be encouraged to expand access to certain patient populations. The use of ADI in risk adjustment is done for target pricing, and performance against the risk-adjusted target price and quality measure benchmarks across all episodes in accordance with the participation track of the TEAM participant collectively determine the NPRA. Therefore, the influence of ADI performance alone on payment would not be an appropriate determination of whether a participant should be encouraged to expand access to services to Medicaid or dually eligible beneficiaries, nor would such an incentive structure be within the direct scope of TEAM.
We appreciate the suggestion for CMS to consider use of the SVI as a potential way to identify beneficiary-level social risk for the purposes of team. The SVI provides a ranking of social vulnerability, or the resilience of communities when confronted by external stresses on human health and incorporates 15 variables across 4 themes: socioeconomic status, household composition and disability, racial/ethnic minority status, and language. [ 984 ] The SVI is not as granular as ADI as ADI uses census block groups and SVI uses census tracts. SVI uses the American Community Survey (ACS) 5-year estimates, and the SVI accordingly inherits the limitations and timing of this source data. For these reasons, we did not propose SVI as potential risk adjustor because we did not feel it would have been appropriate to use SVI in place of the ADI for the purposes of risk adjustment under TEAM.
Comment: A commenter supported the use of dual eligibility status as a risk adjustor as research has shown that dual eligibility status is correlated with other measures of social drivers of health; however, the commenter also suggested that Medicaid eligibility may be a more comprehensive reflection of underserved populations for some hospitals given the variation in Medicaid eligibility across states.
Response: We thank the commenters for their support of using dual eligibility as a possible proxy for social risk under TEAM's risk adjustment methodology and for the suggestion to consider Medicaid eligibility. Given TEAM is a Medicare-based model, we feel that dual eligibility is an appropriate proxy for social risk that reflects both Medicare and Medicaid eligibility.
Comment: A commenter noted that CMS should continue to advance standardization of SDOH data in the context of social risk adjustment to explore ways to modify target prices to better account for historical underutilization.
Response: We appreciate the commenter's suggestion that standardization of SDOH data could help more easily incorporate measures of social risk into target price risk methodologies under team. As described in section X.A.3.d.(4) in the preamble of this final rule, we have identified statistically meaningful risk adjustors, including measures related to ( print page 69800) social risk, that can be reliably measured across TEAM participants and episodes to ensure that data is available to the extent possible for risk adjustment.
We refer readers to section X.A.3.d.(4) in the preamble of this final rule for the comprehensive list of risk adjustment variables, including those related to social risk, that will be included in TEAM's pricing methodology.
We believe it is important for TEAM participants to identify and monitor where disparities exist in their TEAM beneficiary population, and to use the data that they collect to implement evidence-based strategies aimed at addressing the identified health disparities and advancing health equity. To further align with other CMS Innovation Center models and promote health equity, we proposed that TEAM participants can voluntarily submit to CMS, in a form and manner and by the date(s) specified by CMS, a health equity plan for the first performance year. This proposal to make submission of a health equity plan voluntary in PY 1 recognized that constructing a health equity plan may require significant time and effort by the TEAM participant. Beginning in PY 2, we proposed that TEAM participants would be required to submit a health equity plan in a form and manner and by the date(s) specified by CMS. Beginning in PY 2 for those TEAM participants that voluntarily submitted a health equity plan in PY 1 and beginning in PY 3 for those TEAM participants that first reported a health equity plan in PY 2, we proposed that the TEAM participant would submit updates to their previously submitted health equity plans in a form and manner and by date(s) specified by CMS.
We proposed that the health equity plans submitted in all performance years would include the following elements:
- Identifies health disparities. We proposed to define “health disparities” as preventable differences in the burden of disease, injury, violence, or opportunities to achieve optimal health, health quality, or health outcomes that are experienced by one or more “underserved communities” [ 985 ] within the TEAM participant's population of TEAM beneficiaries that the participant will aim to reduce. We proposed to define “underserved communities” as populations sharing a particular characteristic, as well as geographic communities, that have been systematically denied a full opportunity to participate in aspects of economic, social, and civic life. [ 986 ] We proposed that the data sources used to inform the identification of health disparities should also be noted in the plan.
- Identifies health equity goals and describes how the TEAM participant will use the health equity goals to monitor and evaluate progress in reducing the identified health disparities. We proposed to define “health equity goals” as targeted outcomes relative to the health equity plan performance measures for the first PYs and all subsequent PYs.
- Describes the health equity plan intervention strategy. We proposed to define “health equity plan intervention strategy” as the initiative(s) the TEAM participant will create and implement to reduce the identified health disparities.
- Identifies health equity plan performance measure(s), the data sources used to construct the health equity plan performance measures, and an approach to monitor and evaluate the health equity plan performance measures. We proposed to define “health equity plan performance measure(s)” as one or more quantitative metrics that the TEAM participant will use to measure changes in health disparities arising from the health equity plan interventions.
We solicited comment on the proposed voluntary health equity plan submission in PY 1 and mandatory health equity plan submission in PY 2 and all following performance years as proposed in § 512.563. We also solicited comment on whether TEAM participants should be required to submit a health equity plan in PY 2 and for all subsequent performance years if a TEAM participant submits a health equity plan to CMS for another CMS Innovation Center model in the same performance year, or if the TEAM participant should be required to submit a health equity plan that is specific to TEAM and the TEAM participant's population of TEAM beneficiaries. We also solicited comment on the proposed elements of the health equity plan.
The following is a summary of comments we received on health equity plans under TEAM and our responses:
Comment: Some commenters expressed their support for TEAM's inclusion of a health equity plan. A commenter found the definition of health disparities to be broadly defined and requested that CMS narrow the scope of the health equity plan to focus on racial/ethnic, socioeconomic, or similar demographic disparities in key process or outcomes measures central to surgical care. A couple commenters requested that CMS allow hospitals to choose their own area of focus, population and/or process or outcome indicators in their action plan that are related to just one procedure. A commenter recommended that TEAM participants should be required or incentivized to reduce disparities in readmission rates, patient safety indicators, or PROMs. Another commenter raised a concern that requirements to close health equity gaps are well-intentioned, but CMS should consider protections and incentives to specifically encourage inclusion of vulnerable populations as incentives to close health equity gaps could inadvertently cause participants to adversely select beneficiaries.
Response: We thank commenters for their support of the proposed health equity plans under TEAM and for raising some concerns about the proposed components of the plan. The intent of the health equity plan under TEAM is to allow the TEAM participant flexibility to identify health equity goals and interventions that are most appropriate for their context and then work to improve identified health disparities. We agree that TEAM participants should be able to choose their own area of focus within the health equity plan as long as the participant's health equity plan meets all stated requirements. While we recognize that focusing on improving disparities in readmission rates, patient safety indicators, or PROMs may be appropriate for many TEAM participants, we believe that allowing for more contextually specific health equity goals is most appropriate for TEAM participants and will allow for alignment with other relevant health equity work in which the TEAM participant may be engaged. We acknowledge the concern that incentivizing improvements in health equity gaps could potentially lead to adverse selection in that a hospital could theoretically choose to furnish services to healthier patients to improve measures related to the TEAM's participant chosen health equity goals. However, given that payments under TEAM are not tied to performance on the health equity plan, we do not feel there is a significant likelihood that this adverse selection would occur solely as a result of a health equity plan under TEAM. ( print page 69801)
Comment: A couple commenters suggested that the health equity plan should be voluntary for the entirety of TEAM's performance period, while another commenter recommended that health equity plans should be mandatory for all performance years as CMS has already piloted these plans on a voluntary basis in other initiatives. A commenter had concerns that requiring the plan from the start may lead to a less thorough plan and recommended that the health equity plan should start in PY 2, or at a minimum delay the plan to PY 2 for participants in Tracks 1 and 2, to allow providers new to value-based care the opportunity to build infrastructure for addressing disparities in the hospital's service area.
Response: We thank reviewers for their comments on the timing of when health equity plans should be implemented and mandatory under TEAM. We acknowledge that developing and implementing a health equity plan under TEAM may require additional time for a TEAM participant to identify and quantify health disparities appropriate for TEAM's health equity plan and then develop a plan to address them. Accordingly, we believe that voluntary reporting for all model performance years would be appropriate to give sufficient time for TEAM participants interested in developing a TEAM-specific health equity plan, including those that are newer to value-based care, to develop a comprehensive TEAM health equity plan that meets all stated components.
Comment: A few commenters expressed concern over the increased burden of creating and implementing a standalone health equity plan for TEAM. A couple commenters recommended that CMS streamline requirements with other health equity plan requirements in other CMS quality reporting programs. A couple commenters raised concerns that the requirement of a TEAM health equity plan is significantly different from the Hospital Commitment to Health Equity (HCHE) requirement under the Hospital Inpatient Quality Reporting Program beginning in CY 2023, which requires a hospital to attest to five domains that demonstrate a hospital's commitment to health equity, as it will increase administrative burden and could shift the focus to meeting regulatory requirements at the expense of meaningful action to improve health disparities. A commenter expressed concern that CMS is requiring a standalone health equity plan for TEAM whereas the Joint Commission (TJC) requires hospitals to submit a health equity plan. A few commenters suggested CMS should allow overlap of health equity plans if a TEAM participant is also participating in a CMS Innovation Center model that requires a health equity plan to decrease burden. A commenter noted that health equity plans should be tailored to a specific model to align with the identified needs of the model's beneficiaries and that equity plans from other CMS Innovation Center models should not be used for the purposes of TEAM.
Response: We thank commenters for raising concerns around the potential duplication and differences in requirements across different health equity plan and reporting requirements under CMS programs. We disagree that having a health equity plan under TEAM would shift focus away from meaningful action to improve observed TEAM-related disparities as the plan would serve as an accountability mechanism for TEAM participants to identify disparities, work to improve them, and monitor progress against their stated health equity goals. We recognize that a TEAM participant may already report on health equity work under CMS' Hospital Inpatient Quality Reporting Program, such as through the required HCHE measure ( 87 FR 49191 through 49201 ), and other CMS Innovation Center models. However, we feel that TEAM's proposed health equity plan requirements are more appropriate to establishing and advancing specific health equity goals related to TEAM's proposed clinical focus areas than what is required under the HCHE measure in the Hospital Inpatient Quality Reporting Program. We agree with the commenter that health equity plans should be tailored to specific models to align with the identified needs of the model's target beneficiaries. We believe that voluntary reporting of health equity plans for all performance years would allow TEAM participants interested in developing and implementing a health equity plan the flexibility to appropriately focus their goals and interventions to areas of clinical focus most relevant to TEAM.
Comment: A commenter suggested that CMS should add an element to TEAM's health equity plan in future years on community engagement to better understand how TEAM participants are seeking input on model implementation and investing in structures and opportunities to partner with patients, caregivers, and communities with the greatest health inequities.
Response: We thank the commenter for highlighting that community engagement is an important aspect of advancing health equity goals. Given that TEAM's proposed health equity plan requirements allow TEAM participants that voluntarily submit a health equity plan to tailor health equity interventions to the specific needs of their identified beneficiaries, TEAM participants would be able to focus on community engagement strategies that support the improvement of health equity goals should the TEAM participant find it an appropriate intervention strategy for their context.
Comment: A commenter expressed concern that CAHs or safety net hospitals may have limited data available to sufficiently identify inequities and track performance. A commenter recommend that CMS consider providing technical assistance to safety net and rural providers, who may not have sufficient data analytics capacity to determine disparities experienced by the hospital's patient population.
Response: We thank the commenters for raising these concerns as we are aware of the different contexts in which rural and safety net hospitals operate. As part of the implementation of TEAM, we will consider if there are opportunities to provide technical assistance on data analytics for health equity for safety net and rural hospitals in TEAM.
Comment: A commenter recommended that CMS issue additional guidance on how accountability and enforcement of these plans will address health disparities. A commenter expressed concern that CMS has not defined clear guidelines and criteria for assessment of the health equity plans.
Response: We thank the commenters for raising concerns about how TEAM participants would be assessed and held accountable on health equity plans for TEAM. As part of the implementation of TEAM, CMS will provide further sub-regulatory guidance on how CMS will review and provide feedback on TEAM participants' health equity plans for those participants that voluntarily submit a health equity plan.
After consideration of the public comments we received, we are finalizing our proposal of voluntary health equity plan submission for all following performance years in a form and manner and by date(s) specified by CMS as defined in § 512.563(a). A health equity plan voluntarily submitted by a TEAM participant must include all proposed elements and must be specific to TEAM and the TEAM participant's population of TEAM beneficiaries as defined in § 512.563(a). ( print page 69802)
We recognize disparities exist for beneficiaries in the health care system, including those receiving episodic care. Health care disparities highlight the importance of data collection and analysis that includes race, ethnicity, language, disability, sexual orientation, gender identity, and sex characteristics or other demographics by health care facilities. Such data are necessary for integration of health equity in quality programs, because the data permits stratification by patient subpopulation. [ 987 988 ] Stratified data can produce meaningful measures that can be used to expose health disparities, develop focused interventions to reduce them, and monitor performance to ensure interventions to improve care do not have unintended consequences for certain patients. [ 989 ] Furthermore, quality programs are carried out with well-known and widely used standardized procedures including but not limited to root cause analysis, plan-do-study-act (PDSA) cycles, health care failure mode effects analysis, and fishbone diagrams. These approaches are common in the health care industry to uncover the causes of problems, show the potential causes of a specific event, test a change that is being implemented, prevent failure by correcting a process proactively, and identify possible causes of a problem and soft ideas into useful categories, respectively. [ 990 991 992 993 ] Adding a health equity prompt to these standardized procedures integrates a health equity lens within the quality structure and cues considerations of the patient subpopulations who receive care and services from a hospital. [ 994 ]
To align with other CMS efforts, we proposed that TEAM participants could voluntarily report to CMS demographic data of TEAM beneficiaries pursuant to 42 CFR 403.1110(b) in PY 1. Beginning in PY 2 and all subsequent performance years, we proposed that TEAM participants would be required to report demographic data of TEAM beneficiaries to CMS in a form and manner and by a date specified by CMS. We proposed that demographic data would also be required to conform to USCDI version 2 data standards, at a minimum. Collection of this data could provide synergies with goals articulated in the health equity plans of TEAM participants. Further, this demographic data reporting would allow CMS to gain more nuanced understanding of the expanded demographics of TEAM beneficiaries—including data on race, ethnicity, language, disability, sexual orientation, gender identity, sex characteristics, and other demographics—to monitor and evaluate the model.
We proposed that the TEAM participant would be required make a reasonable effort to collect demographic data from all TEAM beneficiaries beginning in PY 2; however, we recognized that this may require additional administrative effort to collect this data or identify TEAM beneficiaries that may elect to not provide this data. We recognized that CEHRT may help to reduce administrative burden once EHR platforms have been programmed to capture and exchange the types of demographic data elements of interest. We also recognized that this demographic data may already be reported to CMS for other CMS initiatives.
We sought comment on the proposed voluntary reporting of demographic data of TEAM beneficiaries in PY 1 and the proposed mandatory reporting of demographic data of TEAM beneficiaries beginning in PY 2 and all following performance years. We wished to minimize the reporting burden on TEAM participants to ensure sufficient time and effort is spent adjusting to the requirements of a mandatory model, and we sought comments on how reporting of this demographic data could minimize burden and if it could be collected from existing data sources.
The following is a summary of the public comments received on the proposed demographic data reporting requirements under TEAM and our responses:
Comment: Some commenters supported the proposal to collect and report demographic data under TEAM as it would provide a comprehensive understanding of health disparities, enabling targeted actions to promote health equity. A commenter supported the voluntary reporting in PY 1 followed by mandatory reporting in all other TEAM performance years.
Response: We thank commenters for their overall support of the proposed demographic data collection and reporting for TEAM beneficiaries as well as support for voluntary demographic data reporting in PY 1 followed by mandatory reporting beginning in PY 2 and for all following performance years. After a consideration of the full range of comments summarized in this section of the preamble of the final rule, we feel that allowing voluntary collection and reporting of demographic data for TEAM beneficiaries for all performance year would be most appropriate.
Comment: A few commenters supported use of USCDI standards for the demographic data reporting requirement under TEAM. A commenter recommended that CMS align coding and documentation requirements for the demographic data reporting under TEAM with national standards, like the USCDI version 3. A commenter appreciated CMS' interest in collecting more robust demographic data but was concerned that variation in data collection processes may result in data that does not confirm to USCDI version 2 standards, recommending that hospitals should have more flexibility in data collection standards. A commenter recommended that claims or QRDA 1 submissions would provide sufficient demographic information to meet the data reporting requirement and cautioned against requiring a separate data submission. A couple commenters recognized that demographic data reporting under TEAM would help to advance health equity goals but cautioned about requiring data collection when federal standards for collecting data are undergoing significant changes, and that attention should first be on data structuring instead of reporting.
Response: We thank commenters for suggestions related to how the proposed demographic data reporting requirements under TEAM could align ( print page 69803) with existing data standards. We believe that it is important that demographic data reported under TEAM follow adopted standards to allow for aggregation and comparability across the model. USCDI standards provide an appropriate national standard given their adoption by ONC. As raised by commenters, we acknowledge that TEAM participants may have different data collection and reporting capabilities that align with existing USCDI standards. We disagree with commenters concerns that demographic data collection and reporting should not occur due to changing national standards. ONC has adopted USCDI version 3 ( 45 CFR 170.213(b) ) which will become the baseline USCDI standard adopted in 45 CFR 170.213 on January 1, 2026, upon the expiration of USCDI v1. While we proposed that TEAM participants that report TEAM beneficiary demographic data would need to use USCDI version 2 at a minimum, we feel that a minimum of USCDI version 3 for the purposes of TEAM would be appropriate given that the USCDI version 3 would be the minimum standard adopted in 45 CFR 170.213 at the beginning of TEAM's performance period. We feel that it would be important to standardize the demographic data elements to the minimum of USCDI version 3 to ensure that we will have standardized data that can be aggregated from those TEAM participants that voluntarily report the data to better understand the demographics of TEAM beneficiaries to help advance the model's health equity goals. The current CMS Innovation Center Enhancing Oncology Model (EOM) has required use of USCDI version 3 standards in their collection and reporting of beneficiary sociodemographic data. [ 995 ]
We do not agree that QRDA-1 or claims-reported demographic data would align with the intended goals of advancing health equity under TEAM. Given that QRDA-1 is a framework for reporting patient-level data about quality measures, we feel that using this framework would not fully capture the required demographic data elements for all TEAM beneficiaries. Similarly, demographic data reported through standard Medicare claims forms does not provide the range of demographic information we hope to obtain on TEAM beneficiaries to help advance the health equity goals of TEAM and the Innovation Center.
Comment: A couple commenters also recommended that CMS should use existing tools for data collection, including HL7 and FHIR standards, or work with EHR vendors so this data could be requested via Application Programming Interfaces (API) from EHRs. A commenter recommended that the reporting of demographic data should be voluntary until it can be reported in an automatic fashion.
Response: We appreciate the comments recommended the use of tools, like HL7 and FHIR standards, to help facilitate the reporting of demographic data of TEAM beneficiaries. We recognize that ONC adopted the HL7 FHIR US Core Implementation Guide (IG) Standard for Trial Use version 6.1.0 at 45 CFR 170.215(b)(1)(ii) , which provides the latest consensus-based capabilities aligned with the USCDI version 3 data elements for FHIR APIs. However, TEAM participants may have varying abilities to access and use these tools to automate voluntary reporting of demographic data required under TEAM. CMS may explore automated solutions in the future that are leveraging certified technology used by providers to reduce the burden of the demographic data reporting required under TEAM. While we are finalizing the voluntary reporting of TEAM beneficiary demographic data, we disagree in principle that reporting should be voluntary until automation is feasible as waiting for automation could limit the period in which TEAM participants may voluntarily report the demographic data of TEAM beneficiaries.
Comment: A few commenters also cautioned that CMS should address patient privacy and data protection to ensure the protection of demographic data, including educating both providers and patients on how this data collection affects care and existing requirements of Health Insurance Portability and Accountability Act (HIPAA).
Response: We appreciate commenters' concerns about patient privacy and data protection as it relates to the proposed requirements for demographic data reporting under TEAM. We acknowledge that TEAM beneficiaries may not wish to disclose some or all of the demographic data elements reportable under TEAM with their TEAM participant or CMS. For those TEAM participants that choose to voluntarily report demographic data, we would expect that TEAM participants would attempt to ask every TEAM beneficiary for these demographic data elements, but TEAM participants would not be penalized should a TEAM beneficiary choose not to disclose some or all the requested information. For those TEAM participants that choose to voluntarily report demographic data, TEAM demographic data reporting requirements would not affect any existing obligations under privacy and security laws for patient information. We also appreciate the recommendation on how CMS could provide support to TEAM participants on demographic data collection efforts. We may consider developing resources on these topics as part of TEAM's implementation.
Comment: A couple commenters requested clarification on whether the demographic data would be reported at the beneficiary-level or in aggregate.
Response: For those TEAM participants that choose to voluntarily repot the demographic data, we clarify that demographic data would need to be reported at the TEAM beneficiary-level for all TEAM beneficiaries that are willing to share some or all of the requested information.
Comment: A couple commenters requested CMS to clarify the required demographics and demographic groups. A commenter requested clarification on the definitions of disability and sex characteristics, recommending that CMS align definitions with existing requirements.
Response: As discussed in the proposed rule at 89 FR 36451 , we would expect TEAM participants that voluntarily report TEAM beneficiary demographic data to consider reporting the following data elements: race, ethnicity, sex, gender identity, sexual orientation, preferred language, and disability status. We will provide further sub-regulatory guidance on the specifics of demographic data reporting and definitions for reportable data elements. We expect that definitions for race, ethnicity, sex, gender identity, sexual orientation, preferred language, disability status, and other possible data elements would align with the definitions under USCDI version 3, as USCDI version 3 will be the minimum USCDI standard adopted in 45 CFR 170.213 at the beginning of TEAM's performance period.
We clarify that sex characteristics as referenced in the proposed rule refers to sex as defined under USCDI version 3. [ 996 ]
There are multiple ways that disability status can be captured under USCDI. TEAM could use six well-tested questions endorsed by the Office of the Assistant Secretary for Planning and Evaluation and the CDC, among others, to support meeting the Affordable Care Act requirements under Section 4302 to collect standardized demographic data. [ 997 ] These questions align with the data elements defined under USCDI version 3 disability status assessment, which should be measured through assessment of a patient's physical, cognitive, intellectual, or psychiatric disabilities (for example, vision, hearing, memory, activities of daily living). [ 998 ] The disability status data element as defined under USCDI version 3 includes assessments related to hearing status; vision status; difficulty with concentration, memory, or decision-making due to a physical, mental or emotional condition; difficulty walking or climbing stairs; difficulty dressing or bathing, and difficulty doing errands alone due to a physical, mental, or emotional condition.
Comment: A commenter recommended that CMS not apply penalties for errors or incompleteness in data.
Response: We acknowledge that TEAM beneficiaries may not wish to disclose some, all, or none of the demographic data elements reportable under TEAM with their TEAM participant or CMS. For those TEAM participants that voluntarily collect and report demographic data, we would expect that TEAM participants ask every TEAM beneficiary for these demographic data elements, but TEAM participants would not receive a penalty should a beneficiary choose not to disclose some or all the requested information.
Comment: A commenter suggested that CMS should pay separately for data collection. A commenter suggested that CMS stratify all measures by patient-level factors, such as demographics, and that CMS consider adopting upside-only incentives to close measure gaps. A commenter recommended that CMS provide upfront payments to support data collection infrastructure.
Response: We thank commenters for their suggestions on how financial incentives could potentially improve the reporting of demographic data and improve the closing of health disparities in measures. We did seek comments on possible infrastructure payments for qualifying safety net TEAM participants that could support data infrastructure (see 89 FR 36452 through 36453 ) but are not finalizing any infrastructure payments in this final rule. Further, we did not propose to require the stratification of quality measure data by the demographic characteristics of TEAM beneficiaries, and we are therefore not able to consider upside-only incentives based on TEAM participants' performance on stratified quality measures.
After a review of public comments, we are finalizing our proposal for voluntary demographic data collection and reporting for all following performance years in a form and manner and by date(s) specified by CMS as described in § 512.563(c). We are finalizing that all demographic data collected for TEAM beneficiaries is to be reported at the beneficiary level as described in § 512.563(c). We will provide further sub-regulatory guidance on the demographic data elements and their definitions that can be voluntarily collected from TEAM beneficiaries and reported by TEAM participants.
The CMS Innovation Center is charged with testing innovations that improve quality and reduce the cost of health care. There is strong evidence that non-clinical drivers of health are the largest contributor to health outcomes and are associated with increased health care utilization and costs. [ 999 1000 ] These individual-level, adverse social conditions that negatively impact a person's health or healthcare are referred to as “health-related social needs” or HRSNs. CMS aims to expand the collection, reporting, and analysis of standardized HRSNs data in its efforts to drive quality improvement, reduce health disparities, and better understand and address the unmet social needs of patients. Standardizing HRSN screening and referral as a practice can inform larger, community-wide efforts to ensure the availability of and access to community services that are responsive to the needs of Medicare beneficiaries. While screening for HRSN is an important step to identify the unmet HRSNs of patients, it is also critical for providers to build referral relationships with community-based organizations and other social service organizations that can more directly support patients identified to have unmet HRSNs. Relationships with community-based organizations should include collaboration to identify available funding sources to support service provision to address unmet HRSNs, as needed.
While more common nationally, HRSN screening is not uniform across geography or health care setting. A literature review of national surveys measuring prevalence of HRSN screening found that 56-77 percent of health care payers and/or delivery organizations screened for HRSNs. [ 1001 ] The review also found that almost half of state Medicaid agencies have established managed care contracting requirements for HRSN screening in Medicaid. [ 1002 ] Despite screening proliferation and generally positive views toward screening among both patients and health care providers, implementation of screening and referral policies for beneficiaries of CMS programs with similar health—and even demographic—profiles may be inconsistent, potentially exacerbating disparities in the comprehensiveness and quality of care.
To help facilitate alignment of HRSN screening within inpatient settings, beginning in 2024, the Hospital Inpatient Quality Reporting (IQR) Program began mandatory reporting of a Screening for Social Drivers of Health (SDOH-1) measure (CMIT ID #1664), the proportion of admitted adults screened for five HRSNs, and a Screen Positive Rate for Social Drivers of Health (SDOH-2) measure (CMIT ID #1662), the percentage of screened admitted adults that screened positive ( print page 69805) for one or more HRSNs. The measures reflect screening for five HRSNs: housing instability, food insecurity, transportation needs, utility difficulties, and interpersonal safety. The CMS Innovation Center Strategy Refresh also established a goal to require all new models to collect and report demographic and social determinants of health (SDOH) data in support of broader system transformation that support goals of advancing health equity.
We proposed that beginning in PY 1, TEAM participants would be required to screen attributed TEAM beneficiaries for at least four HRSN domains—such as but not limited to food insecurity, housing instability, transportation needs, and utilities difficulty—because we believe these areas are most pertinent for the TEAM beneficiary population. We also considered requiring TEAM participants to screen on a standardized set of HRSN domains.
We also proposed that TEAM participants would need to report aggregated HRSN screening data and screened-positive data for each HRSN domain for TEAM beneficiaries that received screening to CMS in a form and manner and by date(s) specified by CMS beginning in PY 1 and for all following performance years. As part of this reporting to CMS, we also proposed that TEAM participants would report on policies and procedures for referring beneficiaries to community-based organizations, social service agencies, or similar organizations that may support patients in accessing services to address unmet social needs.
We recognize TEAM participants may already report some of this HRSN screening data through other CMS initiatives and requiring reporting of aggregated HRSN screening data in TEAM may be redundant. For example, the Hospital IQR Program will begin mandatory reporting beginning with the CY 2024 reporting period/FY 2026 payment determination of two evidence-based measures related to HRSN screening: the Screening for Social Drivers of Health measure and the Screen Positive Rate for Social Drivers of Health measure ( 87 FR 49201 through 49220 ). We therefore sought comment on reporting processes that would streamline reporting of aggregated HRSN screening data for attributed TEAM beneficiaries, including potential use of the Hospital IQR Program measures related to HRSN screening.
We also sought comment on how the reporting of aggregated HRSN screening data could incorporate data on referrals of beneficiaries screening positive for HRSNs to community-based organizations and other organizations helping to address beneficiaries' HRSNs.
The following is a summary of comments we received related to HRSN screening and data reporting requirements under TEAM and our responses:
Comment: Several commenters supported the HRSN screening requirement under TEAM as it could help advance health equity goals. However, many commenters recommended that we should align HRSN screening and data reporting requirements under TEAM with the existing HRSN requirements currently required under the Hospital IQR Program as this would reduce burden. A commenter specifically noted that differing standards with misaligned requirements could create an undue burden and confusion within the large body of work already underway at both the hospital and community levels to align and work toward existing HRSN goals. Another commenter stated that health equity data should be collected at the hospital level like how we proposed to evaluate the PSI 90 measure at the hospital level under TEAM (se 89 FR 36421 ). A commenter suggested that CMS should minimize provider burden on the collection of HRSN data by aligning with national data standards and only requiring reporting of aggregated HRSN screening and screened positive data. A commenter suggested that TEAM participants should be able to report HRSN screening data submitted through other CMS Innovation Center models to fulfill the TEAM requirements. A commenter recommended not requiring HRSN data collection under TEAM as some patients do not respond to these questions when screened.
Response: We thank commenters for their support of the proposed HRSN screening data reporting requirements under TEAM and for raising their suggestions on ways in which the collection and reporting of HRSN screening data could minimize burden on TEAM participants. We agree that standardization of the HRSN data requirements under TEAM with existing CMS programs that require HRSN screening and are applicable to TEAM participants could help to reduce burden under TEAM and minimize confusion with existing efforts at hospital and community levels to gain alignment around HRSN-related goals and interventions. We do not agree that TEAM should not consider requiring the collection of HRSN screening due to some patients choosing not to report this data when screened. As discussed in this section in the preamble of this final rule, HRSN screening is an important step to identify non-clinical drivers of a beneficiary's health and working to improve unmet social needs can support improvements in a beneficiary's overall health. We acknowledge that TEAM beneficiaries would have the right to refuse responding to questions related to HRSNs asked by TEAM participants without penalty to the TEAM participant.
We agree with the many commenters suggested that TEAM should align with the existing SDOH-1 and SDOH-2 measure reporting requirements under the Hospital IQR Program to minimize burden of reporting HRSN at the aggregated TEAM participant level. Specification of these two measures enable a consistent HRSN screening and screened-positive definition for five HRSN domains, as well as data that is aggregated and comparable at the hospital level. Given that we would permit voluntary reporting of aggregated HRSN screening data at the hospital level for TEAM participants using the Hospital IQR Program SDOH-1 and SDOH-2 measures, we do not feel that it would be necessary to allow participants to report HRSN data from other CMS Innovation Center models. Standardizing to the SDOH-1 and SDOH-2 measures in the Hospital IQR Program would ensure consistent reporting across all TEAM participants that voluntarily report this data.
While we agree that use of Hospital IQR Program would provide us with an understanding of HRSN screening and screened-positive data aggregated at the TEAM participant level, we also think that it would be important to gain more granular data on HRSN screening and screened-positive data for TEAM beneficiaries specifically. As TEAM participants would already collect and report the aggregated hospital-level data through the SDOH-1 and SDOH-2 measures in the Hospital IQR Program, we would want to consider ways in which TEAM participants could abstract data on HRSN screening and screened-positive data for TEAM beneficiaries to gain more granular information that could help advance TEAM's health equity goals. The proposal for beneficiary-level HRSN screening and screened-positive data for TEAM beneficiaries for a future performance year could undergo future notice and comment rulemaking.
Comment: A few commenters suggested that TEAM should require the collection of the same five HRSNs (housing instability, food insecurity, transportation needs, utility difficulties, and interpersonal safety) as required in ( print page 69806) the Hospital IQR Program to reduce burden. A couple commenters suggested that hospitals should select domains they wish to screen to tailor screening questions base on community needs. A commenter suggested including a measure of economic insecurity. A commenter suggested that CMS should identify a short list of the most essential HRSNs to standardize across models.
Response: We thank commenters for their perspectives on which HRSN domains should be screened and reported under TEAM. We agree that alignment with the HRSNs domains under the Hospital IQR Program SDOH-1 and SDOH-2 measures would be appropriate and would reduce administrative burden by aligning with existing requirements of other programs in which TEAM participants participate. As these measures have gone through notice and comment rulemaking, we feel that they would reflect an essential set of HRSNs for which TEAM participants should screen. Use of these measures also aligns with CMS Innovation Center priorities in incorporating HRSN screening into all new models. We acknowledge that HRSN can be context-specific and that hospitals may perceive benefits to screening for additional HRSNs beyond the five included in the SDOH-1 and SDOH-2 measures. However, we feel that it is important to obtain standardized HRSN data from all TEAM participants that voluntarily report this data.
Comment: A commenter recommended mandatory HRSN data reporting based on voluntary reporting of HRSN data from patients beginning in Model Year 2.
Response: We thank the commenter for their recommendation on the mandatory nature of reporting beginning in PY 1 of TEAM. While we recognize that TEAM participants will be prepared to report data through the Hospital IQR Program in PY 1 because the SDOH-1 and SDOH-2 measures are required to be reported by all IPPS hospitals as of CY 2024, we feel that voluntary reporting of this aggregated data to TEAM would be appropriate.
Comment: A commenter expressed concern about the clinical settings in which TEAM participants would be required to conduct screening given that some episodes may be initiated in settings like an ambulatory surgical center, and asked if HRSN screening should be conducted for all patients in the event they could trigger a TEAM episode at some point. A commenter noted that providers are at varying stages of HRSN data collection efforts and that the required reporting manner under TEAM could be incompatible with current hospital-level systems and processes.
Response: We thank commenters for raising concerns about the clinical settings in which TEAM participants would be excepted to screen for HRSNs. We expect that TEAM participants would align their screening procedures in applicable clinical settings, so they meet requirements under the SDOH-1 and SDOH-2 measure specifications ( 87 FR 49201 through 49220 ) under the Hospital IQR Program. As the SDOH-1 and SDOH-2 measures are required to reported by all IPPS hospitals as of CY24, we feel that TEAM participants would have sufficient capabilities to collect and voluntarily report the required HRSN screening and screened-positive data beginning in PY 1 of TEAM's performance period.
Comment: A couple commenters suggested that CMS should provide TEAM participants with educational resources that help clinicians explain to patients why HRSN data are being requested and how they will be used to help foster a foundation of trust between patient and providers. A commenter suggested that CMS should publish technical information to facilitate TEAM participants to configure their data systems and EMRs. Another commenter suggested that CMS provide resources to providers to help clinicians identify interventions as patients screen positive for different HRSNs.
Response: We appreciate the commenters suggestions on technical and educational resources that could support TEAM participants in HRSN screening and data reporting. We encourage TEAM participants to review resources available for reporting the SDOH-1 and SDOH-2 measures under the Hospital IQR Program given our alignment with those measure for the purposes of HSRN data reporting under TEAM. We will also consider the feasibility of developing other technical resources on the topics raised by commenters during TEAM's implementation.
Comment: A few commenters suggested that CMS should pay separately for HSRN screening and data collection, such as providing a monthly fee to deliver enhanced wraparound services or upside-only incentives to close observed gaps in social needs data.
Response: We thank commenters for their recommendations on potential of upside-only incentives for HRSN screening and closing HRSN gaps under TEAM. CMS could consider these recommendations in future notice and comment rulemaking for TEAM.
Comment: A commenter expressed concern over CMS setting specific thresholds for HRSN screening and referral rates since there is variability in staff capacity and resources to conduct HRSN screenings.
Response: We thank the commenter for raising this concern around screening thresholds. We currently do not envision that TEAM participants will be assessed against a specific threshold for HRSN screening, and HRSN screening will not be factored into TEAM payment adjustments.
Comment: A commenter supported the proposal for TEAM participants to report on policies and procedures for referring patients screening positive for HRSN to community-based organizations as it is essential to build referral relationships with community-based organizations and other social service organizations that have the history, expertise, and relationships to directly support patients identified to have unmet HRSNs. A commenter noted that TEAM should consider how primary care clinicians can facilitate access to culturally responsive care and support responding to HRSNs. A commenter noted their support for the HRSN data collection and reporting requirements but noted that many clinicians and staff experience data collection fatigue and anxiety when they lack resources to assist patients that screen positive for HRSNs. A couple commenters expressed concern that the proposed referral requirements may be excessively burdensome for TEAM participants to meet as participants may not have relationships with referral organizations or that organizations for referrals may not exist in some communities.
Response: We thank commenters for expressing support and raising concerns about the proposed requirements for TEAM participants to report on policies and procedures they have in place for referring beneficiaries screening positive for select HRSNs to community-based organizations. While we recognize that the availability of referral organizations may vary across TEAM participants, we think that allowing TEAM participants to voluntarily report either existing policies or a plan for developing policies and procedures for referring HRSN screened-positive patients, can help develop more specific strategies, relationships with external organizations, or approaches to be responsive to identified HRSNs of Medicare beneficiaries served by TEAM participants. TEAM participants would not be penalized based on the content of their HRSN referral policy and procedures voluntary reporting, but we ( print page 69807) feel that reporting in this area is an important accountability mechanism to help TEAM participants think beyond HRSN screening and identify the range of provider or organizational relationships, including those with primary care providers, that could help improve identified HRSNs of Medicare beneficiaries served by TEAM participants.
After a review of public comments, we are finalizing our proposal that all TEAM participants can voluntarily report HRSN screening and screened-positive using the SDOH-1 and SDOH-2 measures under the Hospital IQR Program beginning in PY 1 for all other performance years as described in § 512.563(b). We are also finalizing our proposal that TEAM participants that voluntarily report the HRSN data can voluntarily report on referral policies and procedures for screened-positive beneficiaries in a form and manner and by date(s) specified by CMS beginning in PY 1 and for all other performance years as described in § 512.563(b).
In addition to the preceding health equity proposals, we sought comment on possibly providing upfront infrastructure payments to qualified safety net hospital participants to further support safety net hospitals in the transformation of care delivery. Subject to certain limitations, these funds could be available to cover approved expenses aimed at supporting beneficiaries with unmet health and social needs. Payment could support Health Information Technology (health IT)/Electronic Health Records (EHR) enhancements, to the extent they involve population health analytics, support care coordination with other providers within and across care settings, and support referrals to address HRSNs (such as closed loop community-based organization referrals). Participants might also use the infrastructure payment to fund the upfront expenses involved in recruiting dedicated staff (for example, care managers). Participants could distribute or use infrastructure payments received under this model in accordance with existing law or the terms of applicable waivers. Such funds would ensure the infrastructure of safety net hospitals could support the transformational goals of the model and would not come out of the Medicare Parts A and B Trust Funds.
We believe that transformation of acute care delivery in underserved areas is fundamental to addressing persistent disparities and engaging safety net hospitals may broaden the landscape of clinicians focusing on value-based care. We would need to consider the amount of the infrastructure payment, which may include a standard fixed funding component and a variable component that depends on the size of the population served by the safety net hospital participant. We would also need to define a specific set of parameters and formula to calculate the infrastructure payment for each qualifying TEAM participant and sought feedback on the set of parameters we could consider using.
We sought feedback from hospitals and health IT vendors for estimates on the potential upfront start-up costs of health IT investments for safety net hospitals, such as new health information exchange capabilities, solutions to provide patients with access to their health data (for instance, patient portals), capabilities to capture patient-reported outcomes, event notification systems, and community referral capacity. Should we decide to provide such payments, we also expect the infrastructure improvement would require financial investment on the part of the participant, clinicians, and other payer partners, including those on the commercial side.
The goal of the infrastructure payment would be to assist safety net hospital participants, many of whom have less access to capital, participate in and be successful in this model. CMS recognizes that start-up and ongoing annual operating costs could vary greatly between participants for various reasons, including those related to the experience, size, and funding available to the participant.
Past CMS Innovation Center models have proven the utility of infrastructure payments in certain circumstances, which may or may not apply to TEAM. These models include the ACO Investment Model (AIM), a CMS Innovation Center model that tested the effects of making advanced payments to certain ACOs participating in the Shared Savings Program to assist them in transforming care by funding infrastructure investments or staffing. AIM ACOs overwhelmingly used these funds to invest in health IT systems and care management staff and to cover administrative and program compliance costs. At the start of the model, many AIM ACOs lacked the capacity and knowledge to implement population health initiatives, to manage claims-based analytics, and to coordinate practice management. The demonstrated Medicare savings by AIM ACOs suggest that financial accountability with upfront investments can succeed in allowing under-resourced clinicians serving underserved areas to deliver care more efficiently and afford them more flexibility in how they meet beneficiaries' needs without increasing Medicare spending.
To receive an infrastructure payment, we could consider the following requirements and sought comment on any changes: (1) require TEAM participants to be a safety net hospital, as defined by section X.A.3.f.(2)(c) of the preamble of this final rule. The TEAM participant would also submit a detailed plan that describes their intended use of the funds and how those funds would support the goals of the model and improve the care of underserved beneficiaries.
With respect to use of funds for technology investments that involve implementing, acquiring, or upgrading health IT, the hospital would also be required to ensure such technology is certified under the ONC Health IT Certification Program or utilizes nationally recognized, consensus-based standards adopted under section 3004 of the PHSA, 1003 where such criteria or standards are available for the health IT-related activity. Use of these standards and certification criteria ensure that technology investments would support interoperability across systems. Should we make an infrastructure payment to a safety net hospital, we would need to monitor the spending of infrastructure payments to prevent funds from being misdirected and ensure they are used for activities that constitute a permitted use of the funds (for example, health IT/EHR enhancements to the extent those involve population health analytics and support for referrals to address HRSNs, in addition to costs associated with recruiting and hiring dedicated staff). In addition to the initial plan of anticipated spending, should a safety net hospital participant receive upfront funds, they could also be required to submit annual reports (in a standardized format specified by CMS) that includes an itemization of how infrastructure payments were actually spent during the performance year, including expenditure categories, the dollar amounts spent on the various categories, any changes to the spend plan, and such other information as may be specified by CMS. This itemization could include expenditures not identified or anticipated in the submitted spend plan and any amounts remaining unspent. ( print page 69808) Any infrastructure payments that are spent for unauthorized purposes or are unspent at the end of a specified timeframe, that is, 3 years, must be repaid to CMS.
Should safety net hospital participants receive such payments, they would be required to retain adequate records to ensure that we have the information necessary to conduct appropriate monitoring and oversight of the use of infrastructure payments (for example, invoices, receipts, and other supporting documentation of disbursements). CMS would need to conduct audits on a percentage of funding recipients annually to monitor and assess a safety net hospital participant's use of infrastructure funds and participant compliance related to such payments. To encourage speedy resolution of noncompliance and provide an added safeguard against abuse, if CMS determines that a participant has spent infrastructure funds on an identified prohibited use, has unspent funds at the end of the designated eligible spending period, otherwise fails to comply with infrastructure requirements, and/or meets any of the grounds for termination, CMS may require repayment equal to the amount of any infrastructure funds spent on a prohibited use.
As mandatory model, one consideration in potentially implementing an infrastructure payment for qualifying safety net hospital TEAM participants is the long-term scalability of the model. With the goal of longer-term expansion of TEAM, inclusion of a one-time infrastructure payment for qualifying safety net hospitals as part of model design could present challenges to the financial sustainability of the model. Accordingly, the potential objectives and benefits of the infrastructure payment would need to be considered against the feasibility of implementing this model feature should the model be expanded.
We sought comment on the considerations surrounding provision of infrastructure payments and their utility in the acute care setting, including how to identify participants most likely to benefit. We also sought comment on how best to ensure the integrity of such payments in supporting the goal of addressing known health disparities among the episode categories we proposed to test via TEAM. We also sought comment on the proposed methodology and/or parameters that could be used in a formula to determine the infrastructure payment amounts for qualifying TEAM participants.
Comment: Commenters provided many recommendations related to possible infrastructure payments under TEAM.
Response: We greatly appreciate the wide range of suggestions on which TEAM participants would benefit the most from infrastructure payments, the scope and intended use of the payment, ensuring integrity and accountability for the payments, and how payments could be determined. We will continue to consider how infrastructure payments under TEAM could help support participants to strengthen health equity data reporting and improve identified health disparities related to TEAM.
We are expeditiously conducting an in-depth review of the comments we received. This review will help to inform and guide our future rulemaking and other actions in this area.
We believe it is necessary to provide TEAM participants with flexibilities that could support their performance in TEAM and allow for greater support for the needs of beneficiaries. These flexibilities are outlined in this section and include the ability to engage in financial arrangements to share a TEAM participant's reconciliation payment amounts and repayment amounts. Such flexibilities would allow TEAM participants to share all or some of the TEAM participant's reconciliation payment amount or repayment amount. Finally, we believe that TEAM participants caring for beneficiaries may want to offer beneficiary incentives to encourage adherence to recommended treatment and beneficiary engagement in recovery. These financial and beneficiary incentives may help a TEAM participant reach their quality and efficiency goals for the model. They may also benefit beneficiaries and the Medicare Trust Fund if the TEAM participant improves the quality and efficiency of care that results in reductions in hospital readmissions, complications, days in acute care, and mortality, while beneficiary recovery continues uninterrupted or accelerates.
We believe that TEAM participants may wish to enter into financial arrangements with certain providers and suppliers participating in TEAM activities to share their reconciliation payment amount or repayment amount resulting from participation in TEAM. Allowing these types of financial arrangements would allow the alignment of financial incentives of those providers and suppliers participating in TEAM activities to improve quality of care, drive equitable outcomes, and reduce Medicare spending through improved beneficiary care transitions and reduced fragmentation following select episodes of care. We expect that TEAM participants would identify key providers and suppliers caring for beneficiaries in the surrounding communities, and then could establish partnerships with these individuals and entities to promote accountability for the quality, cost, and overall care for beneficiaries, including managing and coordinating care; encouraging investment in infrastructure, enabling technologies, and redesigning care processes for high quality and efficient service delivery; and carrying out other obligations or duties under TEAM. These providers and suppliers may invest substantial time and other resources in these activities, yet they would not be the direct recipients of any reconciliation payment amounts or repayment amounts as they are not the risk bearing entity and do not directly participate in TEAM. Therefore, we believe it is possible that a TEAM participant that may receive a reconciliation payment amount or repayment amount may want to enter into financial arrangements with other providers or suppliers to share this reconciliation payment amount or repayment amount with the TEAM participant. It is a requirement that all financial relationships established between TEAM participants and providers or suppliers for purposes of TEAM comply with all applicable laws and regulations, including the applicable fraud and abuse laws and all applicable payment and coverage requirements in the finalized policy.
As discussed in section X.A.3.g.(9) of the preamble of this final rule, CMS has made the determination that the Anti-Kickback Statute (AKS) Safe Harbor for CMS-sponsored model arrangements ( 42 CFR 1001.952(ii) ) is available to protect certain remuneration finalized in this section when arrangements with eligible providers and suppliers are in compliance with the requirements established in the final rule and the conditions of the safe harbor for CMS-sponsored model arrangements established at 42 CFR 1001.952(ii) .
We recognize that there are numerous arrangements that TEAM participants may wish to enter other than the financial arrangements described in the proposed regulations for which safe harbor protection may be extended that could be beneficial to TEAM ( print page 69809) participants. For example, TEAM participants may choose to engage with organizations that are neither providers nor suppliers to assist with matters such as data analysis; local provider and supplier engagement; care redesign planning and implementation; beneficiary outreach; beneficiary care coordination and management; monitoring TEAM participants' compliance with the model's terms and conditions; or other model-related activities. Such organizations may play important roles in a TEAM participant's plans to implement the model based on the experience these organizations may bring, such as prior experience with episode-based payment models, care coordination expertise, familiarity with a particular locale, or knowledge of bundled data. We require all relationships established between TEAM participants and these organizations for purposes of the model would be those permitted only under existing law and regulation, including any relationships that would include the TEAM participant's sharing of the reconciliation payment amount or repayment amount, and must comply with all applicable laws and regulations. We require these relationships to be solely based on the level of engagement of the organization's resources to directly support the TEAM participants' model implementation.
As proposed, TEAM is a two-sided financial risk model, and the TEAM participant would bear sole financial risk for any repayment amount to CMS in the absence of financial arrangements. However, given the incentive to reduce episode spending to earn a reconciliation payment amount, as described in section X.A.3.d.(5)(j) of the preamble of this final rule, a TEAM participant may want to engage in financial arrangements with providers and suppliers or participants in Medicare ACO initiatives who are making contributions to the TEAM participant's performance in the model. Such arrangements would allow the TEAM participant to share reconciliation payment amounts or repayment amounts with individuals and entities that have a role in the TEAM participant's performance in the model. We proposed at ( 89 FR 36454 ) to use the term “TEAM collaborator” to refer to these individuals and entities.
Because TEAM participants would be accountable for spending and quality during the anchor hospitalization or anchor procedure and the 30-day post discharge period, as described in section X.A.3.b.(5) of the preamble of this final rule, providers and suppliers other than the TEAM participant may furnish services to the beneficiary during the model performance period. As such, for purposes of the Federal Anti-Kickback Statute Safe Harbor for CMS-sponsored model arrangements ( 42 CFR 1001.952(ii) ), we proposed at § 512.505 that the following types of providers and suppliers that are Medicare-enrolled and eligible to participate in Medicare or entities that are participating in a Medicare ACO initiative may be TEAM collaborators:
- Skilled Nursing Facility (SNF).
- Home Health Agency (HHA).
- Long-Term Care Hospital (LTCH).
- Inpatient Rehabilitation Facility (IRF).
- Nonphysician practitioner.
- Therapist in a private practice.
- Comprehensive Outpatient Rehabilitation Facility (CORF).
- Provider or supplier of outpatient therapy services.
- Physician Group Practice (PGP).
- Critical Access Hospital (CAH).
- Non-physician provider group practice (NPPGP).
- Therapy group practice (TGP).
- Medicare ACO.
We sought comment on the proposed definition of TEAM collaborators and any additional Medicare-enrolled providers or suppliers, such as Rural Emergency hospitals, Rural Health Clinics, and Federally Qualified Health Centers, that should be included in this definition.
The following is a summary of the public comments received on this proposal and our response to those comments.
Comment: Several commenters encouraged expanding the types of entities allowed as Team collaborators to include drug or device manufacturers to facilitate value-based contracts, which they stated are essential for addressing rising healthcare costs.
Commenters also recommended offering APM participants flexibility in post-acute care (PAC) payments, allowing them to negotiate rates and create partnerships with PAC providers. They stated this flexibility should extend to home health services, enabling providers to negotiate different rates for home care that better address patient needs.
In addition to flexibility for PAC payment arrangements, commenters also called for greater latitude for participants to create new payment and downstream risk arrangements with medical and community-based providers.
Response: Thank you for your valuable recommendations. We appreciate your insights on expanding the types of entities allowed as Team collaborators and offering flexibility in post-acute care payments. Your suggestions on allowing greater latitude for new payment and downstream risk arrangements are noted. We will take them into consideration in future rulemaking.
Comment: Many commenters had input and feedback on the proposed list of providers and suppliers eligible to be a TEAM collaborator.
A commenter noted that while Medicare hospice services are included in the TEAM episode of care, hospice providers are not listed as eligible TEAM collaborators. They request clarification on whether this exclusion is intentional and, if so, the reasons behind it. They acknowledge that post-discharge use of hospice services in the 30-day episode is rare but urge CMS to consider including hospice providers as eligible collaborators.
Additional recommendations included Registered Dietitian Nutritionists (RDNs) and Rural Emergency Hospitals (REHs) as TEAM collaborators. Finally, there is a strong encouragement for CMS to consider the role of medical technology and device manufacturers as collaborators, potentially offering incentives for innovative technologies that improve outcomes and generate savings.
Other commenters stated that, while LTCHs are listed as eligible to be TEAM collaborators, they have concerns that TEAM participants might limit collaboration with certain post-acute care providers, such as LTCHs, which could restrict patient freedom of choice in selecting their post-acute care provider. To address this, these commenters suggested that CMS require or incentivize hospitals to collaborate with all interested LTCHs and other post-acute care providers to ensure patient access and choice.
Response: Thank you for your comments regarding the inclusion of various providers and entities as TEAM collaborators in the proposed model. We appreciate your detailed feedback and suggestions.
While Medicare hospice services are included in the TEAM episode of care, hospice providers were not initially listed as eligible TEAM collaborators. We acknowledge your request for clarification on this exclusion. Given the rarity of post-discharge hospice use within the 30-day episode, this exclusion was based on the minimal impact these providers might have on the overall performance of TEAM. ( print page 69810) However, we recognize the importance of providing comprehensive care options and will consider your suggestion to include hospice providers as eligible TEAM collaborators to enhance patient access and choice in future iterations of rulemaking.
In addition, we appreciate the recommendations to include Registered Dietitian Nutritionists (RDNs) and Rural Emergency Hospitals (REHs) as TEAM collaborators. Both RDNs and REHs can play significant roles in improving patient outcomes and managing care transitions. We appreciate the commenters' suggestion to consider the role of medical technology and device manufacturers as collaborators. Innovative technologies have the potential to improve outcomes and generate savings. While these entities are not traditional care providers, their inclusion as collaborators could incentivize the adoption of advanced technologies that support the clinical aims of TEAM. We will explore the feasibility of including medical technology and device manufacturers as TEAM collaborators, potentially offering incentives for innovative technologies that enhance care quality and efficiency, in future rulemaking.
We understand the commenters' concerns regarding potential limitations on collaboration with certain post-acute care providers, such as Long-Term Care Hospitals (LTCHs). TEAM aims to ensure patient freedom of choice in selecting their post-acute care provider. We will consider strategies to require or incentivize hospitals to collaborate with all interested LTCHs and other post-acute care providers, ensuring that patient access and choice are maintained and enhanced in future rulemaking.
The feedback is invaluable as we refine TEAM to ensure it meets its goals of improving care quality, enhancing patient access, and reducing costs. We will carefully consider these recommendations in future rulemaking.
Comment: Another commenter emphasized the importance of managing total cost of care and coordinating care across the patient's care team, stating that the proposed model primarily addresses unit price without focusing on utilization, lacking clear incentives for physicians and others directly involved in patient care. This commenter also suggested that TEAM participation should include anesthesiologists and non-hospital entities in financial arrangements.
Response: We recognize the importance of managing total cost of care and ensuring comprehensive care coordination across the patient's care team. The commenters' insights on addressing both unit price and utilization are valuable, and we will consider these factors to enhance the model's incentives for physicians and other care providers involved in patient care. We also appreciate the detailed feedback regarding the inclusion of anesthesiologists and non-hospital entities in TEAM financial arrangements. We will take it into consideration in future rulemaking.
Comment: Another commenter stated appreciation for the opportunity to provide input on the definition of TEAM collaborators and noted the inclusion of nonphysician practitioners as potential collaborators. They requested CMS clarify and define which providers are considered nonphysician practitioners for this model, referencing page 43617 of the Federal Register for the Medicare Program's Alternative Payment Model Updates and the Increasing Organ Transplant Access (IOTA) Model Proposed Rule. Specifically, they asked for clarification on whether RDNs (Registered Dietitian Nutritionists) would be considered TEAM collaborators.
Response: We thank the commenter for highlighting the need for clarity regarding nonphysician practitioners in TEAM and appreciate the reference to page 43617 of the Federal Register for the Medicare Program's Alternative Payment Model Updates and the Increasing Organ Transplant Access (IOTA) Model Proposed Rule.
Similar to what was proposed under IOTA, at proposed § 512.505, we proposed that for purposes of TEAM that a nonphysician practitioner means one of the following: A physician assistant who satisfies the qualifications set forth at § 410.74(a)(2)(i) and (ii) of this chapter; A nurse practitioner who satisfies the qualifications set forth at § 410.75(b) of this chapter; A clinical nurse specialist who satisfies the qualifications set forth at § 410.76(b) of this chapter; A certified registered nurse anesthetist (as defined at § 410.69(b) of this chapter); A clinical social worker (as defined at § 410.73(a) of this chapter); A registered dietician or nutrition professional (as defined at § 410.134 of this chapter).
The commenters' feedback is invaluable as we work to refine these definitions and ensure comprehensive inclusion in future rulemaking cycles.
Comment: A commenter expressed concerns, stating the model's definition of a TEAM collaborator is narrow, and urged CMS to reconsider the current TEAM proposal design and seek more feedback before implementation.
Response: We acknowledge the concerns about the TEAM collaborator definition. This input is crucial in shaping a well-balanced and successful model and we will take it into consideration in future rulemaking.
After consideration of the public comments received, we are finalizing the proposal for the model definition of TEAM collaborators as proposed at § 512.505.
Similar to the CJR Model ( 42 CFR 510.500 ), we proposed at ( 89 FR 36454 ) that certain financial arrangements between a TEAM participant and a TEAM collaborator be termed “sharing arrangements.” For purposes of the Federal Anti-Kickback Statute Safe Harbor for CMS-sponsored model arrangements ( 42 CFR 1001.952(ii) ), we proposed that a sharing arrangement would be to share reconciliation payment amounts or repayment amounts. Where a payment from a TEAM participant to a TEAM collaborator is made pursuant to a sharing arrangement, we proposed to define that payment as a “gainsharing payment,” which is discussed in section X.A.3.g.(4)(c) of the preamble of this final rule. Where a payment from a TEAM collaborator to a TEAM participant is made pursuant to a sharing arrangement, we proposed to define that payment as an “alignment payment,” which is discussed in section X.A.3.g.(4)(c) of the preamble of this final rule. As proposed, a TEAM participant must not make a gainsharing payment or receive an alignment payment except in accordance with a sharing arrangement. As discussed in section X.A.3.g.(4)(b) of the preamble of this final rule, we proposed that a sharing arrangement must comply with all other applicable laws and regulations, including the applicable fraud and abuse laws and all applicable payment and coverage requirements. We proposed that the TEAM participant and TEAM collaborator must document this agreement in writing, and, per monitoring and compliance guidelines (§ 512.590), we proposed that it must be made available to CMS upon request.
We proposed that the TEAM participant must develop, maintain, and use a set of written policies for selecting individuals and entities to be TEAM collaborators. To safeguard against potentially fraudulent or abusive practices, we proposed that the selection criteria determined by the TEAM participant must include the quality of care delivered by the potential ( print page 69811) TEAM collaborator. Moreover, we proposed that the selection criteria could not be based directly or indirectly on the volume or value of referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, or any individual affiliated with a TEAM participant, TEAM collaborator, or collaboration agent. We proposed that, in addition to including quality of care in their selection criteria, TEAM participants must also consider selection of TEAM collaborators based on criteria that include the anticipated contribution to the performance of the TEAM participant in the model by the potential TEAM collaborator to ensure that the selection of TEAM collaborators takes into consideration the likelihood of their future performance.
Finally, we proposed that if a TEAM participant enters a sharing arrangement, its compliance program must include oversight of sharing arrangements and compliance with the applicable requirements of the model. We believe that requiring oversight of sharing arrangements to be included in the compliance program provides a program integrity safeguard.
We sought comment about all provisions described in the preceding discussion, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of the model are met.
We received no comments on these proposals and therefore are finalizing these proposals as proposed in our regulation at § 512.505.
At ( 89 FR 36455 ), we proposed several requirements for sharing arrangements to help ensure that their sole purpose is to create financial alignment between TEAM participants and TEAM collaborators toward the goals of the model while maintaining adequate program integrity safeguards. We proposed that the sharing arrangement must be in writing, signed by the parties, and entered into before care is furnished to TEAM beneficiaries under the sharing arrangement. In addition, we proposed that participation in a sharing arrangement must be voluntary and without penalty for nonparticipation. It is important that providers and suppliers rendering items and services to beneficiaries during the model performance period have the freedom to provide medically necessary items and services to beneficiaries without any requirement that they participate in a sharing arrangement to safeguard beneficiary freedom of choice, access to care, and quality of care. We proposed that a sharing arrangement must set out the mutually agreeable terms for the financial arrangement between the parties to guide and reward model care redesign for future performance toward model goals, rather than reflect the results of model performance years that have already occurred and where the financial outcome of the sharing arrangement terms would be known before signing.
We proposed that the sharing arrangement must require the TEAM collaborator and its employees, contractors, and subcontractors to comply with certain requirements that are important for program integrity under the arrangement. We note that, as proposed, the terms contractors and subcontractors include collaboration agents as defined later in this section. We proposed that a sharing arrangement must require all of the individuals and entities party to the arrangement to comply with the applicable provisions of this final rule, including proposed requirements regarding beneficiary notifications, at proposed § 512.582(b), access to records and record retention, at proposed § 512.586, and participation in any evaluation, monitoring, compliance, and enforcement activities performed by CMS or its designees, at proposed § 512.590 because these individuals and entities all would play a role in model care redesign and they would be part of financial arrangements under the model as proposed. We proposed that the sharing arrangement must also require all individuals and entities party to the arrangement who are providers or suppliers to comply with the applicable Medicare provider enrollment requirement at § 424.500, including having a valid and active TIN or NPI, during the term of the sharing arrangement. This proposed requirement helps ensure that these individuals and entities have the required enrollment relationship with CMS under the Medicare program, although we note that they are not responsible for complying with requirements that do not apply to them. Finally, the sharing arrangement as proposed must require individuals and entities to comply with all other applicable laws and regulations.
We proposed that the sharing arrangement must not pose a risk to beneficiary access, beneficiary freedom of choice, or quality of care so that financial relationships between TEAM participants and TEAM collaborators do not negatively impact beneficiary protections under the model. The sharing arrangement as proposed must require the TEAM collaborator to have a compliance program that includes oversight of the sharing arrangement and compliance with the requirements of the model, just as we proposed requiring TEAM participants to have a compliance program that covers oversight of the sharing arrangement for this purpose as a program integrity safeguard. We sought comment on the anticipated effect of the proposed compliance program requirement for TEAM collaborators, particularly with regard to individual physicians and nonphysician practitioners, small PGPs, NPPGPs, and TGPs and whether alternative compliance program requirements for all or a subset of TEAM collaborators should be adopted to mitigate any effect of the proposal that could make participation as a TEAM collaborator infeasible for any provider, supplier, or other entity on the proposed list of types of TEAM collaborators.
It is necessary that TEAM participants have adequate oversight over sharing arrangements to ensure that all arrangements meet the applicable requirements and to help provide program integrity safeguards. Therefore, we proposed that the board or other governing body of the TEAM participant have responsibility for overseeing the TEAM participant's participation in the model, its arrangements with TEAM collaborators, its payment of gainsharing payments, its receipt of alignment payments, and its use of beneficiary incentives in the model. Additionally, we proposed that the TEAM participant and TEAM collaborator must document this agreement in writing and, as part of the model's monitoring and compliance activities as proposed in (§ 512.590), we proposed that this agreement must be made available to CMS upon request.
For purposes of sharing arrangements under the model, we proposed at § 512.505 to define TEAM activities to be activities related to promoting accountability for the quality, cost, and overall care for TEAM beneficiaries and performance in the model, including managing and coordinating care; encouraging investment in infrastructure and redesigned care processes for high quality and efficient service delivery; or carrying out any other obligation or duty under the model. In addition to the quality of care provided during episodes, we believe the activities that would fall under this proposed definition encompass the totality of activities upon which it would be appropriate for sharing arrangements under the model to be based in order to value the contributions of providers, suppliers, and other entities toward meeting the performance ( print page 69812) goals of the model. We sought comment on the proposed definition of TEAM activities as an inclusive and comprehensive framework for capturing direct care and care redesign that contribute to performance toward model goals.
We proposed that the written agreement memorializing a sharing arrangement must specify the following parameters of the arrangement:
- The purpose and scope of the sharing arrangement.
- The identities and obligations of the parties, including specified TEAM activities and other services to be performed by the parties under the sharing arrangement.
- The date of the sharing arrangement.
- Management and staffing information, including type of personnel or contractors that will be primarily responsible for carrying out TEAM activities.
- The financial or economic terms for payment, including the following:
++ Eligibility criteria for a gainsharing payment.
++ Eligibility criteria for an alignment payment.
++ Frequency of gainsharing or alignment payment.
++ Methodology and accounting formula for determining the amount of a gainsharing payment that is solely based on quality of care and the provision of TEAM activities.
++ Methodology and accounting formula for determining the amount of an alignment payment.
Finally, we proposed to require that the terms of the sharing arrangement must not induce the TEAM participant, TEAM collaborator, or any employees, contractors, or subcontractors of the TEAM participant or TEAM collaborator to reduce or limit medically necessary services to any beneficiary or restrict the ability of a TEAM collaborator to make decisions in the best interests of its patients, including the selection of devices, supplies, and treatments. These requirements as proposed are to ensure that the quality of care for beneficiaries is not negatively affected by sharing arrangements under the model.
The proposals for the requirements for sharing arrangements under the model are included in proposed § 512.565. We sought comment on all of the requirements set out in the preceding discussion, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of the model are met.
We solicited comments on the above proposed policy. The following is a summary of the public comments received on this proposal and our response to those comments.
Comment: A few commenters highlighted the importance of requiring hospitals to pass on savings generated by the model to physicians leading patient care, ensuring fair financial distribution and better alignment with incentives. emphasized that savings should result from improved efficiencies, not just cost-cutting measures that could compromise patient care, and stated concerns about financial arrangements rewarding providers for using cheaper, but potentially inappropriate, products.
Commenters also suggested that clinically relevant specialties be integrated into TEAM leadership and governance to ensure appropriate care and savings based on genuine improvements. Additionally, commenters called for physicians to have adequate resources and flexibility to deliver quality outcomes without being at risk for uncontrollable costs or outcomes.
Commenters noted that hospitals will need to invest in preparing sharing agreements with TEAM collaborators, on top of other model requirements such as developing health equity plans. The commenters stated that success under TEAM should offset the costs of participation not covered by the model.
Finally, commenters recommended for mandatory shared savings agreements between acute care hospitals and surgeons, as previous models like CJR have seen limited voluntary participation in such agreements, despite their potential to enhance savings and care quality.
Response: We appreciate the commenters' thoughtful comments regarding the financial arrangements and sharing agreements within TEAM. We appreciate the emphasis on the importance of fair financial distribution and alignment of incentives to ensure high-quality patient care.
TEAM's proposed financial arrangements, similar to the CJR Model, are designed to promote accountability and encourage providers and suppliers to collaborate on improving care quality while reducing costs. These proposed arrangements, termed “sharing arrangements,” allow for the sharing of reconciliation payment amounts and repayment amounts, fostering financial alignment between TEAM participants and TEAM collaborators.
Under the proposed sharing arrangements, TEAM participants can enter into financial agreements with TEAM collaborators to share savings (gainsharing payments) or losses (alignment payments) resulting from their performance in the model. These arrangements must comply with all applicable laws and regulations, including fraud and abuse laws, and must be documented in writing. As proposed, this documentation must be made available to CMS upon request as part of our monitoring and compliance activities.
We proposed that the TEAM participant must develop, maintain, and use a set of written policies for selecting individuals and entities to be TEAM collaborators. To safeguard against potentially fraudulent or abusive practices as proposed, the selection criteria for TEAM collaborators must include the quality of care delivered and the anticipated contribution to the TEAM participant's performance in the model. Additionally, as proposed, the selection criteria cannot be based on the volume or value of referrals or business generated between the parties. We proposed that sharing arrangements must be voluntary and without penalty for nonparticipation, in order to allow providers and suppliers the freedom to provide medically necessary items and services to beneficiaries without any requirement that they participate in a sharing arrangement and therefore, to safeguard beneficiary freedom of choice, access to care, and quality of care. Additionally, as proposed, sharing arrangements must not induce the reduction or limitation of medically necessary services. As proposed, the terms of these arrangements must also ensure that TEAM collaborators have the ability to make decisions in the best interests of their patients. We recognize the need for adequate oversight of sharing arrangements to ensure compliance with the model's requirements. Therefore, TEAM participants must have a compliance program that includes oversight of sharing arrangements.
We appreciate the suggestion to integrate clinically relevant specialties into TEAM leadership and governance. Ensuring that clinicians have adequate resources and flexibility to deliver quality outcomes is crucial. The model is designed to incentivize genuine improvements in care efficiency and quality, rather than cost-cutting measures.
We acknowledge the recommendation for mandatory shared savings agreements between acute care hospitals and surgeons. TEAM aims to foster voluntary collaboration rather than mandatory agreements; however, we will consider this feedback in future comment and rulemaking. ( print page 69813)
We understand that hospitals may need to invest in preparing sharing agreements and developing health equity plans. We anticipate that the efficiencies and improvements achieved under TEAM will offset these participation costs.
Comment: Commenters requested flexibility in requirements for TEAM sharing arrangements and urged CMS to allow tailoring of compliance expectations based on the specific circumstances of each arrangement. Commenters stated that this flexibility is crucial to encourage participation from a diverse range of collaborators, including small physician group practices and non-physician providers, without imposing undue compliance burdens.
Response: We appreciate the commenters' comments on adding flexibility to the proposed requirements for TEAM sharing arrangements.
The proposed requirements for TEAM sharing arrangements are designed to protect beneficiary access, beneficiary freedom of choice, and quality of care so that financial relationships between TEAM participants and TEAM collaborators do not negatively impact beneficiary protections under the model. Additionally, TEAM sharing arrangement requirements safeguard against potentially fraudulent or abusive practices. Therefore, we believe these requirements to be necessary, and we finalize them as proposed.
We thank the commenters for their engagement and commitment to enhancing TEAM. We will continue to evaluate the TEAM sharing arrangement requirements, in order to ensure they offer the protections as intended, and whether modifications are necessary in future comment and rulemaking.
After consideration of the public comments received, we are finalizing our proposals for the model sharing arrangements requirements as proposed at § 512.565.
We proposed at ( 89 FR 36456 ) several conditions and limitations for gainsharing payments and alignment payments as program integrity protections for the payments to and from TEAM collaborators. We proposed to require that gainsharing payments be derived solely from a TEAM participant's reconciliation payment amounts, internal costs savings, or both; that they be distributed on an annual basis, not more than once per CY; that they not be a loan, advance payment, or payment for referrals or other business; and that they be clearly identified as a gainsharing payment at the time they are paid.
We believe that gainsharing payment eligibility for TEAM collaborators should be conditioned on two requirements—(1) quality of care criteria; and (2) the provision of TEAM activities. With respect to the first requirement, we proposed that to be eligible to receive a gainsharing payment, the TEAM collaborator must meet quality of care criteria during the performance year for which the TEAM participant earned a reconciliation payment amount that comprises the gainsharing payment. We proposed that this quality of care criteria be included in the sharing arrangement and be mutually agreed upon by the TEAM participant and TEAM collaborator. With regard to the second requirement, we proposed that, to be eligible to receive a gainsharing payment, or to be required to make an alignment payment, a TEAM collaborator other than a PGP, NPPGP, or TGP must have directly furnished a billable item or service to a TEAM beneficiary during the same performance year for which the TEAM participant earned a reconciliation payment amount or repayment amount. For purposes of this proposed requirement, we consider a hospital, CAH or post-acute care provider to have “directly furnished” a billable service if one of these entities billed for an item or service for a TEAM beneficiary in the performance year for which the TEAM participant earned a reconciliation payment amount or repayment amount. The phrase “episode,” as proposed, refers to all Part A and B items and services described in section X.A.3.b.(5) (excluding the items and services described in section X.A.3.b.(5)(a)) of the preamble of this final rule that are furnished to a beneficiary described in section X.A.3.b.(5)(b) of the preamble of this final rule, during the time period that begins with the beneficiary's admission to an anchor hospitalization or the date of the anchor procedure, as applicable, and ends on the 30th day of either the date of discharge from the anchor hospitalization or the date of service for the anchor procedure. These proposed requirements ensure that there is a required relationship between eligibility for a gainsharing payment and the direct care for TEAM beneficiaries during an episode for these TEAM collaborators. We believe the provision of direct care is essential to the implementation of effective care redesign, and the proposed requirement provides a safeguard against payments to TEAM collaborators other than a PGP, NPPGP, or TGP that are unrelated to direct care for TEAM beneficiaries during the model's performance year.
We proposed to establish similar requirements for PGPs, NPPGPs and TGPs; however, these proposed requirements take into account that these entities do not themselves directly furnish billable services. We proposed that to be eligible to receive a gainsharing payment or required to make an alignment payment for a given performance year, a PGP, NPPGP or TGP must have billed for an item or service that was rendered by one or more members of the PGP, NPPGP or TGP to a TEAM beneficiary during the episode that is attributed to the same performance year for which the TEAM participant earned a reconciliation payment amount or repayment amount. Like the proposal for TEAM collaborators that are not PGPs, these proposals also require a link between the TEAM collaborator that is the PGP, NPPGP or TGP and the provision of items and services to beneficiaries during the episode by PGP, NPPGP or TGP members.
Moreover, we further proposed that, because PGPs, NPPGPs and TGPs do not directly furnish items and services to beneficiaries, in order to be eligible to receive a gainsharing payment or be required to make an alignment payment, for a given performance year the PGP, NPPGP or TGP must have contributed to TEAM activities and been clinically involved in the care of beneficiaries during an episode that is attributed to the same performance year for which the TEAM participant earned a reconciliation payment amount or repayment amount that comprises the gainsharing payment.
We proposed that the amount of any gainsharing payments must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities. We considered whether this methodology could substantially, rather than solely, be based on quality of care and the provision of TEAM activities, but ultimately determined that basing the methodology solely on these two elements creates a model safeguard where gainsharing aligns directly with the model goal of quality of care and with TEAM activities. We proposed that the gainsharing methodology may take into account the amount of such TEAM activities provided by a TEAM collaborator relative to other TEAM collaborators. We emphasize that, as proposed, financial arrangements may not be conditioned directly or indirectly on the volume or value of referrals or business otherwise generated by, ( print page 69814) between or among TEAM participants, any TEAM collaborator, any collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, or collaboration agent so that the sole purpose of the arrangement is to align the financial incentives of the TEAM participant and TEAM collaborators toward the model. However, we believe that accounting for the relative amount of TEAM activities by TEAM collaborators in the determination of gainsharing payments does not undermine this objective. Rather, the requirement as proposed allowed flexibility in the determination of gainsharing payments where the amount of a TEAM collaborator's provision of TEAM activities (including direct care) to beneficiaries during a performance year may contribute to the TEAM participant's reconciliation payment amount that may be available for making a gainsharing payment.
Greater contributions of TEAM activities by one TEAM collaborator versus another TEAM collaborator that result in greater differences in the funds available for gainsharing payments may be appropriately valued in the methodology used to make gainsharing payments to those TEAM collaborators in order to reflect these differences in TEAM activities among TEAM collaborators.
However, we do not believe it would be appropriate to allow the selection of TEAM collaborators or the opportunity to make or receive a gainsharing payment or an alignment payment to take into account the amount of TEAM activities provided by a potential or actual TEAM collaborator relative to other potential or actual TEAM collaborators because these financial relationships are not to be based directly or indirectly on the volume or value of referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, or collaboration agent. Specifically, with respect to the selection of TEAM collaborators or the opportunity to make or receive a gainsharing payment or an alignment payment, we do not believe that the amount of model activities provided by a potential or actual TEAM collaborator relative to other potential or actual TEAM collaborators could be taken into consideration by the TEAM participant without a significant risk that the financial arrangement in those instances could be based directly or indirectly on the volume or value of referrals or business generated by, between or among the parties. Similarly, if the methodology for determining alignment payments was allowed to take into account the amount of TEAM activities provided by a TEAM collaborator relative to other TEAM collaborators, there would be a significant risk that the financial arrangement could directly account for the volume or value of referrals or business generated by, between or among the parties and, therefore, we proposed that the methodology for determining alignment payments may not directly take into account the volume or value of referrals or business generated by, between or among the parties.
We sought comment on this proposal, where any gainsharing payments must be determined in accordance with a methodology that is based on quality of care and the provision of TEAM activities. We also sought comment on whether the methodology must be based solely on these two elements, or if, alternately, the methodology must be based substantially on these two elements. We sought comment on this proposal for gainsharing payments, where the methodology could take into account the amount of TEAM activities provided by a TEAM collaborator relative to other TEAM collaborators. We were particularly interested in comments about whether this standard would provide sufficient additional flexibility in the gainsharing payment methodology to allow the financial reward of TEAM collaborators commensurate with their level of effort that achieves model goals. In addition, we were interested in comment on whether additional safeguards or a different standard is needed to allow for greater flexibility to provide certain performance-based payments consistent with the goals of program integrity, protecting against abuse and ensuring the goals of the model are met.
We proposed that for each performance year, the aggregate amount of all gainsharing payments that are derived from a reconciliation payment amount by the TEAM participant must not exceed the amount of the reconciliation payment amount. In accordance with the prior discussion, no entity or individual, whether a party to a sharing arrangement or not, may condition the opportunity to make or receive gainsharing payments or to make or receive alignment payments on the volume or value of referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, or collaboration agent. We proposed that a TEAM participant must not make a gainsharing payment to a TEAM collaborator that is subject to any action for noncompliance by CMS or any other federal or state entity or subject to noncompliance with any other federal or state laws or regulations, or for the provision of substandard care to beneficiaries or other integrity problems. Finally, we proposed that the sharing arrangement must require the TEAM participant to recover any gainsharing payment that contained funds derived from a CMS overpayment on a reconciliation payment amount or was based on the submission of false or fraudulent data. These requirements provide program integrity safeguards for gainsharing under sharing arrangements.
With respect to alignment payments, we proposed that alignment payments from a TEAM collaborator to a TEAM participant may be made at any interval that is agreed upon by both parties. We proposed that alignment payments must not be issued, distributed, or paid prior to the calculation by CMS of the repayment amount, and cannot be assessed in the absence of a repayment amount. We also proposed that TEAM participants must not receive any amounts under a sharing arrangement from a TEAM collaborator that are not alignment payments.
We also proposed certain limitations on alignment payments that are consistent with the CJR model. In the proposed policy, for a performance year, the aggregate amount of all alignment payments received by the TEAM participant from all of the TEAM participant's TEAM collaborators must not exceed 50 percent of the repayment amount. Given that the TEAM participant would be responsible for developing and coordinating care redesign strategies in response to its TEAM participation, we believe it is important that the TEAM participant retain a significant portion of its responsibility for repayment amounts. In addition, in the proposed policy, the aggregate amount of all alignment payments from a TEAM collaborator to the TEAM participant for a TEAM collaborator other than an ACO may not be greater than 25 percent of the TEAM participant's repayment amount. The aggregate amount of all alignment payments from a TEAM collaborator to the TEAM participant for a TEAM collaborator that is an ACO may not be greater than 50 percent of the TEAM participant's repayment amount, in the proposed policy. ( print page 69815)
We sought comment on our proposed aggregate and individual TEAM collaborator limitations on alignment payments.
We proposed that all gainsharing payments and any alignment payments must be administered by the TEAM participant in accordance with GAAP and Government Auditing Standards (The Yellow Book). Additionally, we proposed that all gainsharing payments and alignment payments must be made by check, electronic funds transfer, or another traceable cash transaction. We made this proposal to mitigate the administrative burden that the electronic fund transfer (EFT) requirement would place on the financial arrangements between certain TEAM participants and TEAM collaborators, especially individual physicians and nonphysician practitioners and small PGPs, NPPGPs or TGPs which could discourage participation of those suppliers as TEAM collaborators. We sought comment on the effect of this proposal.
The proposals for the conditions and restrictions on gainsharing payments, alignment payments, and internal cost savings under the model were included in proposed § 512.56. We sought comment about all of the conditions and restrictions set out in the preceding discussion, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of TEAM are met.
The following is a summary of the public comments received on these proposals and our response to those comments.
Comment: A few commenters emphasized that financial incentives could enhance patient care, throughput, and patient experiences. Commenters made recommendations including equitably distributing shared savings among physicians and clinical staff involved in surgical episodes, establishing performance parameters at the specialty level, and embracing clinical integration with an upside/downside approach to redistributing incentives.
Response: We thank the commenters for the valuable recommendations. We appreciate these insights and agree that financial incentives can impact patient care, throughput and care transitions, and patient experiences, and believe that as proposed, TEAM financial incentives will enhance these crucial elements.
We proposed two requirements for gainsharing payment eligibility for TEAM collaborators. The first proposed requirement is quality of care criteria, wherein to be eligible to receive a gainsharing payment, the TEAM collaborator must meet quality of care criteria, as included in the sharing arrangement and mutually agreed upon by the TEAM participant and TEAM collaborator, during the performance year where the TEAM participant earned a reconciliation payment amount that comprises the gainsharing payment. The second proposed requirement is that to be eligible to receive a gainsharing payment or make an alignment payment, a TEAM collaborator must have directly furnished a billable item or service to a TEAM beneficiary (or, in the case of a PGP, NPPGP or TGP, must have billed for an item or service that was rendered by one or more members of the PGP, NPPGP or TGP) during the same performance year where the TEAM participant earned a reconciliation payment amount or repayment amount. These proposed requirements entail that the amount of any gainsharing payments must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities. By ensuring there is a relationship between eligibility for a gainsharing payment and criteria for quality of care and the direct care for TEAM beneficiaries during an episode, we believe that crucial elements such as patient care, throughput and care transitions, and patient experiences for TEAM beneficiaries will be enhanced.
We also appreciate the commenters' suggestions related to the equitable distribution of shared savings, establishment of performance parameters at the specialty level, and ability to embrace clinical integration with an upside/downside approach. We believe that the proposed requirements that the amount of gainsharing payments be determined solely based on quality of care and provision of TEAM activities will create an equitable and fair approach to distributing gainsharing payments and establishing performance parameters through quality of care criteria. Additionally, we feel the proposed requirement that to be eligible for both gainsharing and alignment payments, the TEAM collaborator must have furnished a billable item or service allows the opportunity for appropriate and objective gainsharing payment and alignment payments for TEAM collaborators.
We are finalizing this proposal as proposed and without modification. We appreciate these comments and will continue to explore opportunities to further enhance the financial incentive requirements through future comment and rulemaking.
Comment: A commenter recommended that CMS allow PGPs to participate in TEAM through gainsharing agreements with the participating ACHs. The commenter suggested that this would allow for PGPs to contribute their knowledge and patient management skills, to better integrate the care that beneficiaries receive.
Response: We thank the commenter for the suggestion that PGPs should be able to participate in TEAM through gainsharing agreements with the participating ACHs. We acknowledged in the proposed rule that because TEAM participants would be accountable for quality during the anchor hospitalization or anchor procedure and the 30-day post discharge period, providers and suppliers other than the TEAM participant may furnish services to the Medicare beneficiary during the model performance period. As such, we proposed to allow TEAM participants to engage in financial arrangements with TEAM collaborators. PGPs are one such Medicare-enrolled provider type meeting the definition of a TEAM collaborator and, therefore, can engage in gainsharing with a TEAM participant. CMS believes that the TEAM collaborator term as proposed is broad enough to capture all provider/supplier types that may furnish services to the Medicare beneficiary during the model performance period, including PGPs.
Comment: A few commenters supported CMS' proposal to allow gainsharing in TEAM but expressed concern regarding the proposed 50 percent cap on shared losses. The commenters recommended that CMS remove the 50 percent cap on shared losses in order to reduce administrative burden for providers, strengthen integration between ACOs and specialists, and maintain consistency with prior bundled payment models like CJR and BPCI Advanced.
Response: We thank the commenters for their suggestions regarding the proposed 50 percent cap on shared losses. CMS believes, however, that given that the TEAM participant would be responsible for developing and coordinating care redesign strategies in response to its TEAM participation, it is important that the TEAM participant retain a significant portion of its responsibility for repayment amounts. With that said, we believe that the 50 percent cap on shared losses supports CMS' goal. However, we will consider this recommendation in future comment and rulemaking.
Comment: A commenter expressed concern that the proposed TEAM gainsharing policy does not tie ( print page 69816) gainsharing agreements to volume, and it would make it more difficult for TEAM participants to link the size of the gainsharing payment to the partnering organizations' level of involvement in TEAM.
Response: We thank the commenter for their concern regarding the proposed gainsharing policy. As proposed, the TEAM gainsharing methodology may take into account the amount of such TEAM activities provided by a TEAM collaborator relative to other TEAM collaborators, however it should not be based fully on volume of referrals. CMS believes this proposed requirement allows flexibility in the determination of gainsharing payments to TEAM collaborators, who have differing contributions to TEAM activities. We understand that this may result in greater differences in the funds available for gainsharing payments, and believe, as proposed, allows for gainsharing payments to be made appropriately, without tying them directly or indirectly to volume or value of referrals. We are finalizing the policy on gainsharing arrangements as proposed. However, we will continue to review this policy and will take your comments into consideration in future comment and rulemaking cycles.
Comment: A commenter proposed that CMS determine the financial arrangements between the initiating hospital, surgeon, primary care physician and other post-acute providers, and that CMS require the participating ACH to pass the shared savings generated in TEAM to the physicians.
Response: We thank the commenter for their recommendation regarding the distribution of TEAM reconciliation payment and repayment amounts. As proposed, CMS has a direct relationship with the TEAM participant and does not have a relationship with the other providers and suppliers that may be furnishing services to beneficiaries during a TEAM performance period. As such, under this proposal, CMS believes that the TEAM participant, as the risk bearing entity should determine the methodology used to identify key providers and suppliers providing care to an aligned beneficiary and establish financial arrangements as an incentive. In addition, CMS believes that the commenters recommendation that CMS identify a TEAM participant's financial arrangements creates a high level of operational burden. This policy, as proposed, provides TEAM participants the ability to identify financial arrangements in a manner that is most beneficial to the TEAM participants and the downstream providers and suppliers, rather than CMS determining those arrangements on behalf of the participant and downstream providers and suppliers. CMS believes that the gainsharing policy that is proposed aligns with the gainsharing policies of previous CMS Innovation Center bundled payment models, where the participant receives the payment or repayment amount, and can choose the methodology, within the requirements of the model, to distribute the payment and repayment amounts with other providers and suppliers, who are not the risk bearing entity. We are finalizing this policy as proposed. We will take this comment into consideration in future rulemaking cycles.
Comment: A commenter suggested that CMS should require hospitals participating in TEAM to gainshare incentives with downstream participants in order to provide required incentives to providers, who might otherwise not receive incentives depending on the financial arrangements of the TEAM participant.
Response: We thank the commenter for the suggestion and concern regarding proper incentives for TEAM collaborators. CMS does not believe that TEAM participants should be required to gainshare with TEAM collaborators, because TEAM collaborators are not the risk bearing entity. We believe that the goal of the TEAM gainsharing policy should be to offer TEAM participants the opportunity and enough flexibility to identify key providers and suppliers caring for aligned beneficiaries, and then establish partnerships with these individuals and entities to promote accountability for the quality, cost, and overall care for beneficiaries, including managing and coordinating care; encouraging investment in infrastructure, enabling technologies, and redesigning care processes for high quality and efficient service delivery; and carrying out other obligations or duties under TEAM. We are finalizing this policy as proposed. We will take this recommendation into consideration in future rulemaking cycles.
Comment: A commenter expressed concerns with CMS' proposal on gainsharing requirements. Specifically, the commenter suggested that CMS should permit gainsharing payments to be based substantially, rather than solely, on quality of care and the provisions of TEAM activities. The commenter believes that this would provide the appropriate flexibility for TEAM participants to construct their gainsharing agreements.
Response: We thank the commenter for their concern and suggestion to the proposed policy. CMS believes that there must be a model safeguard in place to ensure that gainsharing aligns directly with the TEAM goals of quality of care, and engagement in TEAM activities. We believe that in order for this model safeguard to be in place, gainsharing payments must be based solely on the quality of care and the provisions of TEAM activities. We are finalizing the policy as proposed and will take this commenters suggestion into account in future rulemaking.
Comment: A commenter expressed concerns over CMS' proposed policy of limiting gainsharing payments to TEAM collaborators so that the aggregate payment amount cannot exceed that year's reconciliation payment amount, where the Composite Quality Score is incorporated into the reconciliation payment amount. The commenter suggests that hospitals participating in TEAM need the flexibility to construct their gainsharing programs in their own ways. As such, the commenter stated that TEAM participants should limit aggregate gainsharing payments to the pre-quality adjusted reconciliation amount.
Response: We thank the commenter for their concern and their suggestion to allow TEAM participants to gainshare payments to TEAM collaborators, where the Composite Quality Score is not incorporated into the reconciliation payment amount. In the proposed policy, the reconciliation payment made to the TEAM participant would include the Composite Quality Score adjustment, and this payment amount would be the one that the TEAM participant could share with a TEAM collaborator. As such, CMS believes that the proposed policy that the aggregate gainsharing payments to TEAM collaborators cannot exceed that year's reconciliation payment amount (from CMS), where the composite quality score is incorporated into the reconciliation payment amount, is acceptable because this reconciliation amount paid to the TEAM participant would already include the Composite Quality Score adjustment.
Comment: A commenter recommended that the CMS Innovation Center must allow participants to develop and execute separate gainsharing arrangements with physicians (or physician practices) that are tied to the individual episodes for which the hospital is assuming performance-based risk, by performing reconciliation separately for each episode, not at the enterprise level.
Response: We thank the commenter for their recommendation. We considered whether the gainsharing methodology could substantially, rather ( print page 69817) than solely, be based on quality of care and the provision of TEAM activities, but ultimately determined that basing the methodology solely on these two elements creates a model safeguard where gainsharing aligns directly with the model goal of quality of care and with TEAM activities. The gainsharing methodology as proposed may take into account the amount of such TEAM activities provided by a TEAM collaborator relative to other TEAM collaborators. While we emphasize that financial arrangements may not be conditioned directly or indirectly on the volume or value of referrals or business otherwise generated by, between or among TEAM participants, any TEAM collaborator, any collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, or collaboration agent so that their sole purpose is to align the financial incentives of the TEAM participant and TEAM collaborators toward the model, we believe that accounting for the relative amount of TEAM activities by TEAM collaborators in the determination of gainsharing payments does not undermine this objective. Rather, the proposed requirement allows flexibility in the determination of gainsharing payments where the amount of a TEAM collaborator's provision of TEAM activities (including direct care) to beneficiaries during a performance year may contribute to the TEAM participant's reconciliation payment amount that may be available for making a gainsharing payment. Greater contributions of TEAM activities by one TEAM collaborator versus another TEAM collaborator that result in greater differences in the funds available for gainsharing payments may be appropriately valued in the methodology used to make gainsharing payments to those TEAM collaborators in order to reflect these differences in TEAM activities among TEAM collaborators.
However, we do not believe it would be appropriate to allow the selection of TEAM collaborators or the opportunity to make or receive a gainsharing payment or an alignment payment to take into account the amount of TEAM activities provided by a potential or actual TEAM collaborator relative to other potential or actual TEAM collaborators because these financial relationships are not to be based directly or indirectly on the volume or value of referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, or collaboration agent. Specifically, with respect to the selection of TEAM collaborators or the opportunity to make or receive a gainsharing payment or an alignment payment, we do not believe that the amount of model activities provided by a potential or actual TEAM collaborator relative to other potential or actual TEAM collaborators could be taken into consideration by the TEAM participant without a significant risk that the financial arrangement in those instances could be based directly or indirectly on the volume or value of referrals or business generated by, between or among the parties. Similarly, if the methodology for determining alignment payments was allowed to take into account the amount of TEAM activities provided by a TEAM collaborator relative to other TEAM collaborators, there would be a significant risk that the financial arrangement could directly account for the volume or value of referrals or business generated by, between or among the parties. Therefore, we proposed that the methodology for determining alignment payments may not directly take into account the volume or value of referrals or business generated by, between or among the parties. After consideration of the public comments we received, we are finalizing our proposals as proposed for the model gainsharing payment and alignment payment conditions and limitations without modification in our regulation at § 512.56.
To ensure the integrity of the sharing arrangements, we proposed at ( 89 FR 36458 ) that TEAM participants must meet a variety of documentation requirements for these arrangements. Specifically, we proposed that the TEAM participant must—
- Document the sharing arrangement contemporaneously with the establishment of the arrangement;
- Maintain accurate current and historical lists of all TEAM collaborators, including TEAM collaborator names and addresses; update such lists on at least a quarterly basis; and publicly report the current and historical lists of TEAM collaborators on a web page on the TEAM participant's website; and
- Maintain and require each TEAM collaborator to maintain contemporaneous documentation with respect to the payment or receipt of any gainsharing payment or alignment payment that includes at a minimum the—
++ Nature of the payment (gainsharing payment or alignment payment);
++ Identity of the parties making and receiving the payment;
++ Date of the payment;
++ Amount of the payment;
++ Date and amount of any recoupment of all or a portion of a TEAM collaborator's gainsharing payment; and
++ Explanation for each recoupment, such as whether the TEAM collaborator received a gainsharing payment that contained funds derived from a CMS overpayment of a reconciliation payment amount or was based on the submission of false or fraudulent data.
In addition, we proposed that the TEAM participant must keep records for all of the following:
- Its process for determining and verifying its potential and current TEAM collaborators' eligibility to participate in Medicare if the TEAM collaborator is a Medicare-enrolled provider or supplier.
- A description of current health information technology, including systems to track reconciliation payment amounts and repayment amounts.
- Its plan to track gainsharing payments and alignment payments.
Finally, we proposed that the TEAM participant must retain and provide access to and must require each TEAM collaborator to retain and provide access to, the required documentation as discussed in section X.A.3.j. of the preamble of this final rule and 42 CFR 1001.952(ii) .
The proposals for the requirements for documentation of sharing arrangements under the model are included in § 512.565. We sought comment about all of the requirements set out in the preceding discussion, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of the model are met.
We solicited public comment on our proposal regarding the requirements for documentation of sharing arrangements. We received no comments on these proposals and therefore are finalizing these proposals as proposed in our regulation at § 512.565.
Similar to the CJR model, we proposed at ( 89 FR 36458 ) that certain financial arrangements between TEAM collaborators and other individuals or entities called “collaboration agents” be termed “distribution arrangements.” A ( print page 69818) collaboration agent is an individual or entity that is not a TEAM collaborator and that is a PGP, NPPGP, or TGP member that has entered into a distribution arrangement with the same PGP, NPPGP, or TGP in which he or she is an owner or employee. For purposes of the Federal Anti-Kickback Statute Safe Harbor for CMS-sponsored model arrangements ( 42 CFR 1001.952(ii) ), we proposed that a distribution arrangement is a financial arrangement between a TEAM collaborator that is a PGP, NPPGP or TGP and a collaboration agent for the sole purpose of sharing a gainsharing payment received by the PGP, NPPGP or TGP. Where a payment from a TEAM collaborator to a collaboration agent is made pursuant to a TEAM distribution arrangement, we proposed to define that payment as a “distribution payment.” As proposed, a collaboration agent may only make a distribution payment in accordance with a distribution arrangement which complies with the provisions of this proposed model and all other applicable laws and regulations, including the fraud and abuse laws.
Like our proposal for gainsharing payments, we proposed that the amount of any distribution arrangements must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities. We considered whether this methodology could substantially, rather than solely, be based on quality of care and the provision of TEAM activities, but ultimately determined that basing the methodology solely on these two elements creates a model safeguard where gainsharing aligns directly with the model goal of quality of care and with TEAM activities.
The proposals for the general provisions for distribution arrangements under the model are included in proposed § 512.568. We sought comment about all of the provisions set out in the preceding discussion, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of the model are met.
We solicited public comment on our proposal regarding the requirements for general distribution arrangements. We received no comments on these proposals and therefore are finalizing these proposals as proposed in our regulation at § 512.568.
We proposed at ( 89 FR 36458 ) several specific requirements for distribution arrangements as a program integrity safeguard to help ensure that their sole purpose is to create financial alignment between TEAM collaborators and collaboration agents and performance toward TEAM goals. These proposed requirements largely parallel those proposed in section X.A.3.g.(4) of the preamble of this final rule for sharing arrangements and gainsharing payments based on similar reasoning for these two types of arrangements and payments. We proposed that all distribution arrangements must be in writing and signed by the parties, contain the effective date of the agreement, and be entered into before care is furnished to TEAM beneficiaries under the distribution arrangement. Furthermore, we proposed that participation must be voluntary and without penalty for nonparticipation, and the distribution arrangement must require the collaboration agent to comply with all applicable laws and regulations.
We sought comment on this proposal, where any distribution payments must be determined in accordance with a methodology that is based on quality of care and the provision of TEAM activities. We also sought comment on whether the methodology must be based solely on these two elements, or if the methodology must be based substantially on these two elements. Additionally, and also like our proposal for gainsharing payments, we proposed that the opportunity to make or receive a distribution payment must not be conditioned directly or indirectly on the volume or value of referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, or collaboration agent. We proposed more flexible standards for the determination of the amount of distribution payments from PGPs, NPPGPs and TGPs allowing TEAM collaborators and collaboration agents to create tailored distribution payments that supports the individual structure of their arrangement.
We note that for distribution payments made by a PGP to PGP members, by NPPGPs to NPPGP members, or TGPs to TGP members, the proposed requirement that the amount of any distribution payments must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities may be more limiting in how a PGP, NPPGP or TGP pays its members than is allowed under existing law. However, we believe quality of care is an important facet of episode-based payment models and making this a requirement for distribution payment supports greater emphasis on quality of care improvement in TEAM. Further this is consistent with the BPCI Advanced model that required their Net Payment Reconciliation Amount (NPRA) Shared Payments and Partner Distribution Payments to achieve quality performance targets to receive these payments.
We sought comment on this proposal and specifically whether there are additional safeguards or a different standard is needed to allow for greater flexibility in calculating the amount of distribution payments that would avoid program integrity risks and whether additional or different safeguards are reasonable, necessary, or appropriate for the amount of distribution payments from a PGP to its members, a NPPGP to its members or a TGP to its members.
Similar to our proposed requirements for sharing arrangements for those TEAM collaborators that furnish or bill for items and services, we proposed that a collaboration agent is eligible to receive a distribution payment only if the collaboration agent furnished or billed for an item or service rendered to a beneficiary during an episode that occurred during the same performance year for which the TEAM participant accrued the internal cost savings or earned a reconciliation payment amount that comprises the gainsharing payment being distributed. We note that, as proposed, all individuals and entities that fall within our proposed definition of collaboration agent may either directly furnish or bill for items and services rendered to beneficiaries. This proposal ensures that, there is the same required relationship between direct care for beneficiaries during a performance year and distribution payment eligibility that we require for gainsharing payment eligibility. We believe this requirement as proposed provides a safeguard against payments to collaboration agents that are unrelated to direct care for beneficiaries during the performance year.
We further proposed that with respect to the distribution of any gainsharing payment received by an ACO, PGP, NPPGP or TGP, the total amount of all distribution payments in a performance year must not exceed the amount of the gainsharing payment received by the TEAM collaborator from the TEAM participant for that performance year. Like gainsharing and alignment payments, we proposed that all distribution payments must be made by check, electronic funds transfer, or another traceable cash transaction. Under the proposal, the collaboration agent must retain the ability to make ( print page 69819) decisions in the best interests of the beneficiary, including the selection of devices, supplies, and treatments. Finally, under the proposal, the distribution arrangement must not induce the collaboration agent to reduce or limit medically necessary items and services to any Medicare beneficiary or reward the provision of items and services that are medically unnecessary.
We proposed that the TEAM collaborator must maintain contemporaneous documentation regarding distribution arrangements in accordance with proposed § 512.586, including—
- The relevant written agreements;
- The date and amount of any distribution payment(s);
- The identity of each collaboration agent that received a distribution payment; and
- A description of the methodology and accounting formula for determining the amount of any distribution payment.
We proposed that the TEAM collaborator may not enter into a distribution arrangement with any individual or entity that has a sharing arrangement with the same TEAM participant. This proposal ensures that the proposed separate limitations on the total amount of gainsharing payment and distribution payment to PGPs, NPPGPs, TGPs, physicians, and nonphysician practitioners that are solely based on quality of care and the provision of TEAM activities are not exceeded in absolute dollars by a PGP, NPPGP, TGP, physician, or nonphysician practitioner's participation in both a sharing arrangement and distribution arrangement for the care of the same TEAM beneficiaries during the performance year. Allowing both types of arrangements for the same individual or entity for care of the same beneficiary during the performance year could also allow for duplicate counting of the individual or entity's same contribution toward model goals and provision of TEAM activities in the methodologies for both gainsharing and distribution payments, leading to financial gain for the individual or entity that is disproportionate to the contribution toward model goals and provision of TEAM activities by that individual or entity. However, we recognize there could be instances where an individual or entity could have distribution arrangements with multiple TEAM collaborators. For example, a physician may practice with and have reassigned their Medicare billing rights to multiple PGPs, and those PGPs may each be TEAM collaborators. We sought comment on allowing an individual or entity to have distribution arrangements with multiple TEAM collaborators and whether there are additional program integrity safeguards that should be established in those scenarios. Finally, we proposed that the TEAM collaborator must retain and provide access to and must require collaboration agents to retain and provide access to, the required documentation in accordance with § 512.586.
The proposals for requirements for distribution arrangements under the model were included in proposed § 512.568. We sought comment about all of the proposed requirements set out in the preceding discussion, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of the model are met. In addition, we sought comment on how the regulation of the financial arrangements under this proposal may interact with how these or similar financial arrangements are regulated under the Medicare Shared Savings Program.
We solicited public comment on our proposal regarding the requirements for distribution arrangements. We received no comments on these proposals and are finalizing these proposals as proposed in our regulation at § 512.568.
We proposed at ( 89 FR 36460 ) that TEAM allow for certain financial arrangements within an ACO between a PGP and its members. Specifically, we proposed that certain financial arrangements between a collaboration agent that is both a PGP, NPPGP, or TGP and an ACO participant and other individuals termed “downstream collaboration agents” be termed a “downstream distribution arrangement.” A downstream distribution arrangement, as proposed, is a financial arrangement between a collaboration agent that is both a PGP, NPPGP, or TGP and an ACO participant and a downstream collaboration agent for the sole purpose of sharing a distribution payment received by the PGP, NPPGP, or TGP. A downstream collaboration agent, as proposed, is an individual who is not a TEAM collaborator or a collaboration agent and who is a PGP member, a NPPGP member, or a TGP member that has entered into a downstream distribution arrangement with the same PGP, NPPGP, or TGP in which he or she is an owner or employee, and where the PGP, NPPGP, or TGP is a collaboration agent. Where a payment from a collaboration agent to a downstream collaboration agent is made pursuant to a downstream distribution arrangement, we proposed to define that payment as a “downstream distribution payment.” As proposed, a collaboration agent may only make a downstream distribution payment in accordance with a downstream distribution arrangement which complies with the requirements of this section and all other applicable laws and regulations, including the fraud and abuse laws.
We sought comment about all of the provisions set out in the preceding discussion, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of TEAM are met.
We solicited public comment on our proposal regarding the downstream distribution arrangements. We received no comments on these proposals and are finalizing these proposals as proposed in our regulation at § 512.570.
We proposed at ( 89 FR 36460 ) several specific requirements for downstream distribution arrangements as a program integrity safeguard to help ensure that their sole purpose is to create financial alignment between collaboration agents that are PGPs, NPPGPs, or TGPs which are also ACO participants and downstream collaboration agents toward the goals of the TEAM to improve the quality and efficiency of episodes. These requirements as proposed largely parallel those proposed for sharing and distribution arrangements at proposed § 512.565 and § 512.568 and gainsharing and distribution payments at proposed § 512.565 and § 512.568 based on similar reasoning for these types of arrangements and payments. We proposed that all downstream distribution arrangements must be in writing and signed by the parties, contain the effective date of the agreement, and entered into before care is furnished to TEAM beneficiaries under the downstream distribution arrangement. Furthermore, we proposed that participation must be voluntary and without penalty for nonparticipation, and the downstream distribution arrangement must require the downstream collaboration agent to comply with all applicable laws and regulations.
Like our proposals for gainsharing and distribution payments, we proposed that the opportunity to make or receive a downstream distribution payment must not be conditioned directly or indirectly on the volume or value of ( print page 69820) referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, any downstream collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, collaboration agent, or downstream collaboration agent. We proposed the amount of any downstream distribution payments from an NPPGP to an NPPGP member or from a TGP to a TGP member must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities and that may take into account the amount of such TEAM activities provided by a downstream collaboration agent relative to other downstream collaboration agents. We believe that the amount of a downstream collaboration agent's provision of TEAM activities (including direct care) to TEAM beneficiaries during episodes may contribute to the TEAM participant's internal cost savings and reconciliation payment amount that may be available for making a gainsharing payment to the TEAM collaborator that is then shared through a distribution payment to the collaboration agent with which the downstream collaboration agent has a downstream distribution arrangement. Greater contributions of TEAM activities by one downstream collaboration agent versus another downstream collaboration agent that result in different contributions to the distribution payment made to the collaboration agent with which the downstream collaboration agents both have a downstream distribution arrangement may be appropriately valued in the methodology used to make downstream distribution payments to those downstream collaboration agents.
Similar to our proposed requirements for distribution arrangements for those TEAM collaborators that are PGPs, we proposed that a downstream collaboration agent is eligible to receive a downstream distribution payment only if the PGP billed for an item or service furnished by the downstream collaboration agent to a TEAM beneficiary during an episode that was attributed to the same performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment amount that comprise the gainsharing payment from which the ACO made the distribution payment to the PGP that is an ACO participant. This proposal ensures that there is the same required relationship between direct care for TEAM beneficiaries during episodes and downstream distribution payment eligibility that we require for gainsharing and distribution payment eligibility. We believe this proposed requirement provides a safeguard against payments to downstream collaboration agents that are unrelated to direct care for TEAM beneficiaries during episodes.
We further proposed that the total amount of all downstream distribution payments made to downstream collaboration agents must not exceed the amount of the distribution payment received by the collaboration agent (that is, the PGP, NPPGP, or TGP that is an ACO participant) from the ACO that is a TEAM collaborator. Like gainsharing, alignment, and distribution payments, we proposed that all downstream distribution payments must be made by check, electronic funds transfer, or another traceable cash transaction. As proposed, the downstream collaboration agent must retain the ability to make decisions in the best interests of the patient, including the selection of devices, supplies, and treatments. The distribution arrangement must not induce a downstream collaboration agent to reduce or limit medically necessary items and services to any Medicare beneficiary or reward the provision of items and services that are medically unnecessary.
We proposed that the PGP, NPPGP, or TGP must maintain contemporaneous documentation regarding downstream distribution arrangements in accordance with proposed § 512.586, including all of the following:
- The relevant written agreements.
- The date and amount of any downstream distribution payment(s).
- The identity of each downstream collaboration agent that received a downstream distribution payment.
- A description of the methodology and accounting formula for determining the amount of any downstream distribution payment.
We proposed that the PGP, NPPGP, or TGP may not enter into a downstream distribution arrangement with any PGP, NPPGP, or TGP member who has a sharing arrangement with a TEAM participant or distribution arrangement with the ACO the PGP, NPPGP, or TGP is a participant in. This proposal ensures that the proposed separate limitations on the total amount of gainsharing payment, distribution payment, and downstream distribution payment to PGP, NPPGP, or TGP members that are solely based on quality of care and the provision of TEAM activities are not exceeded in absolute dollars by a PGP, NPPGP, or TGP member's participation in more than one type of arrangement for the care of the same TEAM beneficiaries during episodes. Allowing more than one arrangement for the same PGP, NPPGP, or TGP member for the care of the same TEAM beneficiaries during episodes could also allow for duplicate counting of the PGP, NPPGP, or TGP member's effort in TEAM activities in the methodologies for the different payments. Finally, we proposed that the PGP, NPPGP, or TGP must retain and provide access to, and must require downstream collaboration agents to retain and provide access to, the required documentation in accordance with § 512.586.
We sought comment about all of the requirements, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of TEAM are met.
Comment: Commenters expressed concerns about TEAM's penalties based on Medicare spending, noting that hospitals may incur costs from unaffiliated providers. They expressed concerns that hospitals might consolidate services to control costs, potentially leading to higher prices. They suggested upfront infrastructure investments for safety net providers and financial support for preparation and ongoing costs. Additionally, they urged CMS to cover upfront costs for medication supplies to avoid barriers to participation and reduce health inequities.
Response: We appreciate the detailed feedback on TEAM. CMS recognizes the complexity and potential financial implications for hospitals, particularly concerning relationships with unaffiliated providers and the risk of increased consolidation. To address these concerns, CMS will explore options to provide upfront infrastructure investments, especially for safety net providers, and consider mechanisms to support both initial and ongoing operational costs. Furthermore, we acknowledge the importance of covering upfront costs for medication supplies to ensure equitable participation. These insights are invaluable in refining TEAM to balance cost control with high-quality care and accessibility. We will take this comment into consideration in future rulemaking.
Comment: A commenter expressed concerns that downstream participants are only subject to penalties without being eligible for savings, incentive ( print page 69821) payments or involvement in identifying appropriate post-acute discharge placements. They believe that downstream participants could be unfairly penalized for issues they are not involved in. The commenter suggests that hospitals should be required to share incentives with downstream participants and include them in TEAM strategy and pre-discharge placement decisions to address this imbalance.
Response: We appreciate the comments raised regarding the participation of downstream participants in the incentive and penalty structures in TEAM. We have carefully considered the suggestion to mandate that hospitals share incentives with downstream participants and involve them in pre-discharge placement decisions. However, we believe our current approach effectively addresses these issues while maintaining the integrity and goals of TEAM.
Our proposal includes several specific requirements for downstream distribution arrangements designed to ensure financial alignment and safeguard against potential abuses. Key elements of our proposed approach include:
1. Written and Signed Agreements: All downstream distribution arrangements must be documented in writing, signed by all parties, and established before care is furnished to TEAM beneficiaries. This ensures transparency and accountability.
2. Voluntary Participation: Participation in these arrangements is entirely voluntary, and there are no penalties for non-participation. This respects the autonomy of downstream participants while promoting collaboration.
3. Quality-Based Payments: The methodology for downstream distribution payments is based solely on the quality of care and the provision of TEAM activities, not on the volume or value of referrals. This focus on quality ensures that patient care remains the primary consideration.
4. Documentation and Compliance: We require rigorous documentation of all agreements, payments, and methodologies to ensure compliance with all applicable laws and regulations. This includes maintaining records of the relevant agreements, payment details, and the formulas used to determine payment amounts.
5. Safeguards Against Abuse: The arrangements include safeguards to prevent the reduction of medically necessary services or the provision of unnecessary services. Payments are made through traceable transactions, ensuring financial transparency.
6. Contribution-Based Payments: The amount of downstream distribution payments is tied to the downstream collaboration agent's contributions to TEAM activities, which may vary based on their involvement in direct care for TEAM beneficiaries. This ensures that payments reflect actual contributions to patient care and program goals.
7. Consistent Performance Year Attribution: To receive downstream distribution payments, the PGP must have billed for services furnished by the downstream collaboration agent during an episode attributed to the same performance year. This aligns payments with the specific period of care delivery, ensuring relevance and accuracy.
By adhering to these structured requirements, we aim to foster a collaborative environment that rewards quality care and ensures the appropriate distribution of incentives. Our proposed approach balances the need for financial alignment with the imperative to protect program integrity and avoid potential abuses. While we recognize the importance of involving downstream participants in incentive structures, our proposed safeguards and requirements provide a robust framework that supports the goals of TEAM without compromising on these critical principles.
After consideration of the public comments received, we are finalizing these proposals as proposed in our regulation at § 512.570.
We believe it is necessary and appropriate to provide additional flexibilities to TEAM participants for purposes of testing the model, to give TEAM participants additional access to the tools necessary to improve beneficiaries' quality of care, drive equitable outcomes, and reduce Medicare spending through improved beneficiary care transitions and reduced fragmentation during episodes of care. As proposed at ( 89 FR 36461 ), TEAM participants may choose to provide in-kind patient engagement incentives to beneficiaries in an episode, which may include but not be limited to items of technology, subject to the following conditions consistent with 42 CFR 510.515 .
As discussed in section X.A.3.g.(9) of the preamble of this final rule, we stated that if the proposed beneficiary incentives are finalized, we would expect to make a determination that the Anti-Kickback Statute Safe Harbor for CMS-sponsored model patient incentives ( 42 CFR 1001.952(ii) ) would be made available. This Safe Harbor will protect the beneficiary incentives proposed in this section when the incentives are offered in compliance with the requirements established in the final rule and the conditions for use of the Anti-Kickback Statute Safe Harbor set out at 42 CFR 1001.952(ii) .
As stated previously, TEAM participants may choose to provide in-kind engagement incentives, which may include but not be limited to items of technology, to TEAM beneficiaries in an episode, subject to the following proposed conditions. We proposed at ( 89 FR 36461 ) that the incentive must be provided directly by the TEAM participant or by an agent of the TEAM participant under their direction and control to the TEAM beneficiary during an episode. Additionally, we proposed that the item or service provided must be reasonably connected to the TEAM beneficiary's medical care and be a preventive care item or service or an item of service that advances a clinical goal, as described in section X.A.3.g.(7)(b) of the preamble of this final rule, by engaging the TEAM beneficiary in better managing their own health. We sought comment on the proposed conditions for TEAM beneficiary incentives, as outlined in 512.575. Specifically, we sought comment on whether these proposed conditions are reasonable, and whether additional conditions are appropriate to further engage TEAM beneficiaries in their own healthcare management while preventing fraud or abuse.
Comment: Many commenters supported the proposed beneficiary incentives aimed at encouraging adherence to recommended treatments and improving patient engagement in recovery. The commenters highlighted the benefits of such incentives in managing health, preventing disease development, and addressing health-related social needs, such as transportation to medical appointments. These incentives are seen as particularly beneficial for historically underserved populations by directly supporting chronic care management and promoting equitable outcomes.
Additionally, commenters suggested that beneficiary incentives in the model should align with its clinical aims. Commenters suggested that CMS consider adding flexibilities for TEAM participants to provide limited financial assistance, such as cost-sharing for non-opioid prescriptions, to further promote adherence to drug regimens, reduce ( print page 69822) opioid misuse, adhere to care plans and reduce complications. Commenters also recommend that TEAM participants seek direct input from beneficiaries on needed items or services to advance shared clinical goals.
Response: We appreciate the feedback and support regarding the proposed beneficiary incentives. We appreciate the recognition of these incentives' potential to enhance adherence to recommended treatments, manage health conditions, and address health-related social needs, particularly for historically underserved populations.
We value the suggestion to add flexibilities for TEAM participants to provide limited financial assistance, such as cost-sharing for non-opioid prescriptions. This aligns with our goal of promoting adherence to drug regimens and care plans while reducing the risk of opioid misuse and potential complications. We will consider this recommendation and its alignment with the clinical aims of TEAM.
Additionally, we acknowledge the importance of seeking direct input from beneficiaries on needed items or services that would advance shared clinical goals. We will explore mechanisms to incorporate beneficiary feedback to ensure that the incentives provided meet their specific needs and enhance their engagement in care.
The commenters' insights are valuable as we refine TEAM to achieve better health outcomes and equitable care for all beneficiaries. We are finalizing this proposal without modification; however, we will take these recommendations into consideration in future rulemaking.
Comment: A commenter supported the proposed beneficiary incentives designed to encourage adherence to recommended treatments and promote beneficiary engagement in recovery.
Response: We appreciate the commenter's support of the proposed beneficiary incentives aimed at encouraging adherence to recommended treatments and promoting beneficiary engagement in recovery. We appreciate the commenter's feedback and will continue to develop strategies that enhance patient participation and improve health outcomes.
After consideration of the public comments received, we are finalizing our proposals for the TEAM beneficiary incentives as proposed in our regulation at § 512.575. Therefore, as discussed in section X.A.3.g.(9) of the preamble of this final rule and since these proposed beneficiary incentives are finalized, we are making the determination that the Anti-Kickback Statute Safe Harbor for CMS-sponsored model patient incentives ( 42 CFR 1001.952(ii) ) is available. This Safe Harbor will protect the beneficiary incentives finalized in this section when the incentives are offered in compliance with the requirements established in the final rule and the conditions for use of the Anti-Kickback Statute Safe Harbor set out at 42 CFR 1001.952(ii) .
In some cases, items or services involving technology may be useful as beneficiary engagement incentives that can advance a clinical goal of TEAM by engaging a beneficiary in managing their health during the 30 days following discharge from the anchor hospitalization or anchor procedure. However, we believe specific enhanced safeguards are necessary for these items and services to prevent abuse, and our proposals are consistent with the CJR model policies ( 80 FR 73437 ). Specifically, we proposed at ( 89 FR 36461 ) that items or services involving technology provided to a beneficiary may not exceed $1,000 in retail value for any TEAM beneficiary in any episode (per episode), and that items or services involving technology provided to a TEAM beneficiary must be the minimum necessary to advance a clinical goal as discussed in this section for a TEAM beneficiary in an episode. We proposed additional enhanced requirements for items of technology exceeding $75 in retail value as an additional safeguard against misuse of these items as beneficiary engagement incentives. Specifically, we proposed that these items of technology that exceed $75 in retail value remain the property of the TEAM participant and be retrieved from the TEAM beneficiary at the end of the episode. As proposed, the TEAM participant must document all retrieval attempts, including the ultimate date of retrieval. We understand that TEAM participants may not always be able to retrieve these items after the episode ends, such as when a TEAM beneficiary dies or moves to another geographic area. Therefore, in cases when the item of technology is not able to be retrieved, we proposed that the TEAM participant must determine why the item was not retrievable and if it was determined that the item was used inappropriately (if it were sold, for example) preventing future beneficiary incentives for that TEAM beneficiary. Following this proposed process, the documentation of diligent, good faith attempts to retrieve items of technology will be deemed to meet the retrieval requirement.
Our proposals for enhanced requirements for technology provided to TEAM beneficiaries as beneficiary engagement incentives under TEAM are included in proposed § 512.575. We sought comment on our proposed requirements for beneficiary engagement incentives that involve technology. Additionally, we sought comment on the types of technology that may be useful to advance the goals of the model. We welcomed comment on additional or alternative program integrity safeguards for this type of beneficiary engagement incentive, including whether the financial thresholds proposed in this section are reasonable, necessary, and appropriate.
Comment: A commenter stated their belief that TEAM incentives for innovative and effective medical technology can significantly benefit patients with comorbidities or risk factors. These incentives would enable hospitals to enhance monitoring and management throughout surgical episodes, leading to more efficient care.
Response: We appreciate the commenter's insight regarding the impact of TEAM incentives for innovative medical technology, especially for patients with comorbidities or other risk factors. CMS acknowledges that such incentives can play a crucial role in improving monitoring and management throughout surgical episodes. We are committed to integrating these considerations into future models to support hospitals in providing efficient, high-quality care. This feedback helps us ensure that our programs effectively address the needs of diverse patient populations and enhance overall care outcomes.
After consideration of the public comments we received, we are finalizing our proposals for technology provided to a TEAM beneficiary as proposed in our regulation at § 512.575.
As discussed in section X.A.3.b. of the preamble of this final rule, the proposed episodes are broadly defined to include most Part A and Part B items and services furnished during episodes of care that extend 30 days following discharge from the anchor hospitalization or anchor procedure that begins the episode. Therefore, we believe that in-kind beneficiary engagement incentives may appropriately be provided for managing acute conditions arising from episodes, as well as chronic conditions if the ( print page 69823) condition is likely to have been affected by care during the episode or when substantial services are likely to be provided for the chronic condition during the episode. We proposed at ( 89 FR 36462 ) to allow TEAM participants to offer in-kind beneficiary engagement incentives, where such incentives must be closely related to the provision of high-quality care and advance a clinical goal for a TEAM beneficiary and should not serve as inducements for TEAM beneficiaries to seek care from the TEAM participants or other specific suppliers and providers. We proposed that beneficiary incentives must advance one of the following clinical goals of TEAM:
- Beneficiary adherence to drug regimens.
- Beneficiary adherence to a care plan.
- Reduction of readmissions and complications resulting from treatment during the episode.
- Management of chronic diseases and conditions that may be affected by treatment for the TEAM clinical condition.
Our proposals for beneficiary engagement incentives are included in § 512.575. We sought comment on our proposed clinical goals of TEAM, as well as whether the advancement of additional or different clinical goals through beneficiary engagement incentives may better advance the overarching goals of TEAM while maintaining appropriate program integrity safeguards.
We received no comments on these proposals and are finalizing these proposals as proposed without modification in our regulation at § 512.575.
As a program safeguard against misuse of beneficiary engagement incentives under TEAM, we proposed that TEAM participants must maintain documentation of items and services furnished as beneficiary engagement incentives that exceed $25 in retail value including items of technology. In addition, we proposed at ( 89 FR 36462 ) to require that the documentation established contemporaneously with the provision of the items and services must include at least the following:
- The date the incentive is provided.
- The incentive and estimated value of the item or service.
- The identity of the beneficiary to whom the item or service was provided.
We further proposed that the documentation regarding items of technology exceeding $75 in retail that are required to be retrieved from the beneficiary at the end of an episode must also include contemporaneous documentation of any attempt to retrieve technology. In instances where the item of technology is not able to be retrieved, we proposed that the TEAM participant must determine why it is not retrievable, and if the item were misappropriated (if it were sold, for example), then further steps must be taken to ensure that TEAM beneficiary does not receive further TEAM beneficiary incentives. Following this proposed process, documented, diligent, good faith attempts to retrieve items of technology will be deemed to meet the retrieval requirement.
Finally, we proposed that the TEAM participant must retain and provide access to the required documentation in accordance with § 512.586.
We sought comment on our proposed documentation requirements, including whether additional or different documentation requirements may provide better program integrity safeguards.
Comment: Several commenters express concerns that the enhanced documentation requirements for items of technology exceeding $75 in retail value will be burdensome for TEAM participants. They highlight that the requirement for TEAM participants to determine why an item was not retrievable may be impractical or impossible to meet. Commenters suggest CMS finalize the regulation without this requirement and consider raising the cost threshold above $75 for enhanced requirements. Additionally, some commenters are concerned that these documentation requirements could disincentivize the provision of beneficiary incentives and place an undue burden on both providers and patients. They recommend increasing the minimum retail value price threshold for items requiring documentation and retrieval to reduce administrative overhead and the burden on patients and caregivers.
Response: We thank the commenter for their feedback regarding the enhanced documentation requirements for beneficiary incentives, particularly for items of technology exceeding $75 in retail value. We understand the concerns about the potential burdens these requirements may place on TEAM participants, providers, and patients.
We have carefully considered the suggestion to finalize § 512.575 without the requirement to determine why an item was not retrievable and to raise the cost threshold above $75 for enhanced requirements. We also have considered the recommendation to increase the minimum retail value price threshold for items requiring documentation and retrieval to reduce administrative overhead.
However, these requirements are in alignment with other CMS policies, such as those found in CJR, on cost thresholds for beneficiary incentives. We understand it is important to reduce burden and remove barriers to using such incentives. Our goal is to ensure that beneficiary incentives effectively support patient care and engagement without imposing unnecessary burdens on providers or patients. We are finalizing this policy as proposed. However, we appreciate these insights and will take them into account in future rulemaking cycles, as we continue to refine TEAM's documentation requirements to strike the right balance between program integrity and practical implementation.
Comment: A commenter recommended that CMS add a non-scored PI measure to record participation in the data collection process of the Insights Measure reporting. This would incentivize hospitals and providers to contribute, helping ONC gain statistically useful and meaningful data. It is the commenter's opinion that including a Yes/No measure for provider participation in the Insights Measures program would likely encourage behavior that helps HHS achieve its goals.
Response: We thank the commenter for this valuable recommendation. We appreciate these insights on adding a non-scored PI measure to record participation in the data collection process of the Insights Measure reporting. The suggestion to include a Yes/No measure for provider participation to incentivize cooperation and help achieve HHS goals is noted. We will take it into consideration in future rulemaking.
Comment: A commenter suggested that beyond the proposed documentation requirements, CMS should establish data collection mandates to test the effectiveness of incentives on healthcare outcomes like hospital readmissions and patient experience, specifically beneficiary adherence to care plans. They recommended that this data be analyzed by demographic groups to design more effective incentive programs, especially for underserved populations.
Response: We appreciate the commenter's suggestion to enhance data ( print page 69824) collection to assess the effectiveness of TEAM incentives on healthcare outcomes and patient experience. We agree that such data, especially when analyzed by demographic groups, is crucial for informing and refining beneficiary incentive programs. CMS is committed to exploring ways to incorporate these recommendations to better serve all populations, particularly those that are underserved. We are finalizing this proposal as proposed. However, in future rulemaking cycles and as we continue to refine the model, we will consider the inclusion of additional data collection requirements to measure the impact of incentives on hospital readmissions, adherence to care plans, and other health outcomes. This feedback is valuable in helping us improve the design and implementation of TEAM to achieve equitable and high-quality care for all beneficiaries. We will take this comment into consideration in future rulemaking.
After consideration of the public comments we received, we are finalizing our proposals for the documentation of beneficiary engagement incentives as proposed in our regulation at § 512.575.
OIG authority is not limited or restricted by the provisions of the model, including the authority to audit, evaluate, investigate, or inspect the TEAM participant, TEAM collaborators, collaboration agents, downstream collaboration agents, or any other person or entity or their records, data, or information, without limitations. Additionally, as proposed, no model provisions limit or restrict the authority of any other Government Agency to do the same.
The proposals for enforcement authority under the model are included in § 512.150(e). We sought comment about all of the requirements set out in the preceding discussion, including whether additional or different safeguards would be needed to ensure program integrity, protect against abuse, and ensure that the goals of the model are met.
We received no comments on these proposals. These proposals are finalized at § 512.150(e).
Under section 1115A(d)(1) of the Act, the Secretary may waive such requirements of Titles XI and XVIII and of sections 1902(a)(1), 1902(a)(13), 1903(m)(2)(A)(iii) of the Act, and certain provisions of section 1934 of the Act as may be necessary solely for purposes of carrying out section 1115A of the Act with respect to testing models described in section 1115A(b) of the Act.
For this model and consistent with the authority under section 1115A(d)(1) of the Act, the Secretary may consider issuing waivers of certain fraud and abuse provisions in sections 1128A, 1128B, and 1877 of the Act. No fraud or abuse waivers are being issued in this document; fraud and abuse waivers, if any, would be set forth in separately issued documentation. Any such waiver would apply solely to TEAM and could differ in scope or design from waivers granted for other programs or models. Thus, not withstanding any provision of this final rule, TEAM participants, TEAM collaborators, collaboration agents, and downstream collaboration agents must comply with all applicable laws and regulations, except as explicitly provided in any such separately documented waiver issued pursuant to section 1115A(d)(1) of the Act specifically for TEAM.
At proposed § 512.576, we proposed to make the Federal Anti-Kickback Statute Safe Harbor for CMS-sponsored model arrangements available to protect remuneration furnished in TEAM in the form of the sharing arrangement's gainsharing payments and alignment payments, the distribution arrangement's distribution payments, and the downstream distribution arrangement's distribution payments provided that all of the financial arrangements associated with such payment meet all safe harbor requirements set forth in 42 CFR 1001.952(ii) , proposed § 512.565, proposed § 512.568, and proposed § 512.570. We considered, but did not propose, adopting an alternative approach in which the availability of the safe harbor for a specific type of financial arrangement would only be conditioned on compliance with the specific requirements for that type of financial arrangement and the compliance of the other financial arrangements associated with such payment would not implicate the availability of the safe harbor. For example, we considered, but did not propose, an alternative proposal making the availability of the safe harbor for the sharing arrangement's gainsharing payments only conditioned on compliance with the requirements associated with that type of financial arrangement and not also conditioned on the compliance of a downstream financial arrangement associated with such payment.
In the proposed rule at ( 89 FR 36463 ), we considered not allowing use of the safe harbor provisions. However, we decided that use of the safe harbor will encourage the goals of the model. We believe that a successful model requires integration and coordination among TEAM participants and other health care providers and suppliers. We believe the use of the safe harbor will encourage and improve beneficiary experience of care and coordination of care among providers and suppliers. We also believe this safe harbor offers flexibility for innovation and customization. The safe harbor allows for emerging arrangements that reflect up-to-date understandings in medicine, science, and technology.
We sought comment on this proposal, including that the Anti-Kickback Statue Safe Harbor for CMS-sponsored model arrangements ( 42 CFR 1001.952(ii)(1) ) and CMS-sponsored model patient incentives ( 42 CFR 1001.952(ii)(2) ) be available to TEAM participants and TEAM collaborators, collaboration agents, and downstream collaboration agents. After reviewing the public comments, we are finalizing that, in addition to or in lieu of a waiver of certain fraud and abuse provisions in sections 1128A and 1128B of the Act, the Anti-Kickback Statute (AKS) Safe Harbor for CMS-sponsored model arrangements and CMS-sponsored model patient incentives ( 42 CFR 1001.952(ii) (1) and 42 CFR 1001.952(ii) (2)) is available to protect remuneration exchanged pursuant to certain financial arrangements and patient incentives that may be permitted under the final rule. Specifically, in this final rule, we have determined that the CMS-sponsored models safe harbor is available to protect the following financial arrangements and incentives: the TEAM sharing arrangement's gainsharing payments and alignment payments, the distribution arrangement's distribution payments with TEAM collaborators and collaboration agents, the downstream distribution arrangements and downstream distribution payments with collaboration agents and downstream collaboration agents, and TEAM beneficiary incentives. We summarize and respond to public comments received on this proposal below.
Comment: Many commenters urge CMS to ensure that TEAM's policies align with the physician self-referral law and Anti-Kickback Statute (AKS) Safe Harbor related to arrangements that facilitate value-based health care delivery and payment finalized in 2020. There is a call for greater clarity and ( print page 69825) education on the types of arrangements and flexibilities allowed under these exceptions and safe harbor, as providers are uncertain about compliance, risking penalties or exclusion from federal programs.
Commenters emphasized the need for CMS and the Office of Inspector General (OIG) to provide fraud and abuse waivers and clear guidance to support TEAM participants and collaborators. Commenters noted the complexity of applying the fraud and abuse legal framework to new models like TEAM and recommended maximum security for participants to minimize compliance risks. A commenter stated that modernizing legal barriers, such as the Anti-Kickback Statute and physician self-referral law, is necessary to permit value-based contracts for drugs and devices structured as product/service guarantees or risk-sharing arrangements.
Response: We thank the commenter for this feedback regarding the alignment of TEAM's policies with the physician self-referral law exceptions and Anti-Kickback Statute (AKS) Safe Harbor related to the arrangements that facilitate value-based health care delivery and payment, as well as the concerns about compliance and the preparation required for TEAM. No fraud and abuse waivers are being issued for TEAM and we believe we have provided clarity in this final rule for participants to meet the requirements of an applicable exception under the physician self-referral law and an applicable AKS safe harbor. We appreciate these comments and suggestions for ensuring clear guidance and support for TEAM participants and collaborators.
After consideration of the public comments we received, we are finalizing our proposals as proposed in our regulation at § 521.576 for TEAM participants, TEAM collaborators, collaboration agents, and downstream collaboration agents to comply with all applicable laws and regulations, except as explicitly provided in any such separately documented waiver issued pursuant to section 1115A(d)(1) of the Act specifically for TEAM without modification. We again note that no fraud and abuse waivers are being finalized in this rule.
We believe it may be necessary and appropriate to provide flexibilities to hospitals participating in TEAM, as well as other providers and suppliers that furnish services to beneficiaries in episodes. The purpose of such flexibilities would be to increase episode quality, decrease episode spending or internal costs, or both of providers and suppliers, resulting in better, more coordinated care for beneficiaries and improved financial efficiencies for Medicare, providers, and beneficiaries. These possible additional flexibilities could include use of our waiver authority under section 1115A of the Act, which provides authority for the Secretary to waive such requirements of title XVIII of the Act as may be necessary solely for purposes of carrying out section 1115A of the Act with respect to testing models described in section 1115A(b) of the Act. This provision affords broad authority for the Secretary to waive statutory Medicare program requirements as necessary to carry out the provisions of section 1115A of the Act.
As we have stated elsewhere in section X.A.2.c. of the preamble of this final rule, our previous and current efforts in testing episode payment models have led us to believe that models where entities bear financial responsibility for total Medicare spending for episodes of care hold the potential to incentivize the most substantial improvements in episode quality and efficiency. As discussed in section X.A.3.a.(3) of the preamble of this final rule, we proposed that TEAM participants participating in Track 1 of this model be eligible for reconciliation payment amounts based on spending and quality performance in PY 1. TEAM participants in Track 2 would be eligible for repayment amounts and reconciliation payment amounts starting in PY 2, while TEAM participants in Track 3 are eligible for repayment amounts and reconciliation payment amounts starting in PY 1. We believe that where TEAM participants bear financial accountability for excess episode spending beyond the reconciliation target price while high quality care is valued, they will have an increased incentive to coordinate care furnished by the hospital and other providers and suppliers throughout the episode to improve the quality and efficiency of care. With these incentives present, there may be a reduced likelihood of over-utilization of services that could otherwise result from waivers of Medicare program rules. Given these circumstances, waivers of certain program rules for providers and suppliers furnishing services to TEAM beneficiaries may be appropriate to offer more flexibility than under existing Medicare rules for such providers and suppliers, so that they may provide appropriate, efficient care for beneficiaries. An example of such a program rule that could be waived to potentially allow more efficient inpatient episodes would be the 3-day inpatient hospital stay requirement prior to a covered skilled nursing facility (SNF) stay for beneficiaries who could appropriately be discharged to a SNF after less than a 3-day inpatient hospital stay. This type of waiver was implemented in a range of previous and existing CMS initiatives, including various episode-based payment models and accountable care initiatives.
We welcomed comments on possible waivers under section 1115A of the Act of certain Medicare program rules beyond those specifically discussed in the proposed rule that might be necessary to test this model. We will consider the comments that were received during the public comment period and may make future proposals regarding program rule waivers during the course of the model test if we determine that the additional flexibilities afforded by these waivers would be appropriate and beneficial to the model test. We were especially interested in comments explaining how such waivers could provide providers and suppliers with additional flexibilities that are not permitted under existing Medicare rules to increase quality of care and reduce unnecessary episode spending, but that could be appropriately used in the context of TEAM where TEAM participants bear full responsibility for total episode spending.
The following is a summary of the public comments we received on additional waivers of Medicare program rules beyond those specifically discussed in the proposed rule and our responses to those comments:
Comment: Many commenters suggested that we consider additional Medicare policy waivers beyond those which we proposed or considered for TEAM.
A few commenters suggested that we waive patient cost-sharing for certain services provided to TEAM beneficiaries. A couple of commenters cited the precedent for waiving patient-cost sharing in ACO REACH, indicating that this provision could improve access for patients with health-related social needs (HRSNs). A commenter suggested that we waive co-pays for telehealth visits.
A couple commenters recommended that we consider coverage of transportation costs for medical care.
A commenter suggested that we waive the requirement for physician certification of an outpatient physical ( print page 69826) therapy plan of care for TEAM beneficiaries. The commenter indicated that this requirement imposes an administrative burden on physical therapists and physicians and represents a barrier to initiating care. The commenter suggested that CMS accept the presence of an order or referral on file and the delivery of a physical therapy plan of care to the treating physician as satisfying the requirement for physician review of the plan of care, removing the requirement for a physician signature.
A commenter recommended that we include a waiver which would authorize nurse practitioners to order cardiac rehabilitation, noting the precedent for such a waiver in the ACO Realizing Equity, Access, and Community Health (REACH) and the planned States Advancing All-Payer Equity Approaches and Development (AHEAD) Models. The commenter cited CABG as a qualifying condition for cardiac rehabilitation and suggested that permitting nurse practitioners to order cardiac rehabilitation for TEAM beneficiaries would contribute to care coordination efforts under TEAM and improve access to cardiac rehabilitation.
A commenter recommended that TEAM episodes which do not qualify toward the compliance threshold for the IRF “60 Percent Rule”—that is, episodes that do not fall under the 13 conditions listed in § 412.29(b)(2) which must encompass at least 60 percent of an IRF's total inpatient population in order for the IRF to be classified for Medicare IRF payment—be excluded from an IRF's “60 Percent Rule” calculations altogether. The commenter indicated that the incentive for TEAM participants to reduce costs would eliminate the need for the “60 Percent Rule” to limit utilization and suggested that not waiving this rule for TEAM beneficiaries could limit the provision of IRF services as deemed appropriate by the TEAM participant.
A commenter recommended that we consider waiving the initiation of care regulations for home health, specifically requesting that therapy staff be permitted to initiate home health services in situations where both therapy and nursing are required under the home health plan of care. The commenter suggested that waiving this requirement under TEAM could promote access to care and care coordination.
A commenter requested that we include waivers in TEAM that match the waivers offered under the Medicare Advantage (MA) Value-Based Insurance Design (VBID) Model.
A commenter requested that we offer a waiver allowing TEAM participants the flexibility to negotiate alternative payment rates and structures with PAC providers.
A commenter requested that we consider flexible options for organizations to provide insurance coverage for home health services including increased custodial home health aides, home care, home nursing, and home therapy.
A commenter suggested that CMS cover up to two weeks of respite care for patients whose condition necessitates medication and pain management but does not allow for intensive therapies.
A commenter suggested that we consider increased coverage of daily wound care in situations where receiving such care would allow patients to return home rather than staying in a hospital or institutional PAC setting.
A commenter encouraged us to consider potential episode-specific waivers for TEAM. The commenter suggested that waivers should be focused on enabling participants to provide stable transitions of care and timely post-discharge follow-up.
Response: We thank the commenters for their suggestions for additional waivers and appreciate the information provided on the potential benefits of these waivers for TEAM participants and beneficiaries. As discussed in the proposed rule, we will consider the comments we received during the public comment period and our early model implementation experience, including experience in specific episodes, and may propose additional waivers for TEAM in future rulemaking.
Comment: A few commenters requested that CMS offer a waiver of beneficiary inducements to allow TEAM participants to conduct pre-surgical home visits. These commenters suggested that allowing such home visits before surgery would permit participants to proactively assess a patient's home environment and potentially make structural modifications, thereby improving the patient's chances of successful recovery at home. A couple of the commenters suggested that a beneficiary inducements waiver could reduce reliance on inpatient or SNF settings that present high costs and infection risk. A commenter offered the inclusion of a beneficiary inducement waiver in the Guiding an Improved Dementia Experience (GUIDE) Model as an example.
Response: We thank the commenters for their recommendation to include a beneficiary inducement waiver for pre-surgical home visits. We recognize the potential benefit to beneficiaries of environmental assessment and modification prior to surgery. However, we feel that this potential benefit must be balanced against the protection of beneficiary freedom of choice through the restriction of beneficiary inducements as defined in section 1128A(i)(6) of the Act. At this time and considering the clinical episodes proposed under TEAM and the precedents set in BPCI Advanced and CJR, we do not believe that the potential benefits of waiving beneficiary inducement rules outweigh the potential harms. We will monitor patient outcomes and PAC utilization throughout the model test and may consider additional waivers in future rulemaking.
Comment: Some commenters sought greater standardization of waivers and their requirements across models. A few commenters recommended that CMS establish a core standard set of waivers across all Medicare APMs. These commenters suggested that standardizing waiver offerings across models could ease administrative and compliance burdens and thereby motivate greater waiver utilization. A commenter recommended that CMS use the same guidelines for the SNF 3-day rule waiver under TEAM as are used in the corresponding waiver under MSSP, suggesting that consistent guidelines would benefit participants.
Response: We thank the commenters for their suggestions to establish a standard set of waivers across CMS APMs and to standardize waiver usage requirements across models. We recognize the administrative burden of billing for Medicare program waivers and the benefit of consistency for providers participating in multiple payment models either concurrently or successively. The proposed TEAM waivers of the SNF 3-day rule and the geographic site requirements and originating site requirements were included in the proposed rule with this consistency in mind. TEAM is designed to build upon progress made and lessons learned from BPCI Advanced and CJR in value-based care for Medicare beneficiaries undergoing surgery. We recognize that many TEAM participants will have experience coordinating and delivering care—including telehealth and SNF services—under these models. Thus, we proposed provisions for the applicable waivers in TEAM that we expect will be familiar to providers with experience in BPCI Advanced and/or CJR. Still, in recognition of the expected range of value-based care and APM experience ( print page 69827) among TEAM participants and consistent with learning system offerings in BPCI Advanced and CJR, we plan to provide informational support to TEAM participants, including support geared toward new or less experienced participants, regarding model provisions such as Medicare policy waivers.
We also note that internal efforts at CMS are underway to identify, analyze, and compare Medicare payment policy waiver utilization across models. In line with the intention of offering such flexibilities to APM participants under section 1115A of the Act, we share the goal of facilitating providers' use of the proposed waivers to deliver efficient and high-quality care to beneficiaries. As stated in the proposed rule at 89 FR 36463 , we will consider public comments on the proposed waivers and may make future proposals regarding program rule waivers during the course of the model test.
Comment: A few commenters requested that CMS further expand the waivers offered to TEAM participants in general.
Response: We thank the commenters for their suggestions. We plan to monitor utilization, spending, quality, and outcome trends throughout the model test and may consider additional waivers in future rulemaking.
Comment: A commenter suggested that we consider providing coverage for counseling services for patients and their families.
Response: We thank the commenter for the suggestion to cover counseling services for patients and their families. We direct the commenter's attention to the scope of included services under TEAM as defined in § 512.525(e) to include all Medicare Part A and B items and services except as excluded under § 512.525(f). Thus, Medicare Part B services included and covered in TEAM episodes would include certain counseling and mental health services, including family counseling if the main purpose is to help with the beneficiary's treatment.
We address the Medicare programmatic waivers we proposed in the proposed rule in the following sections. We decline at this time to waive any additional Medicare programmatic requirements. We will review the information provided by the commenters and our early model experience and may consider waiving additional requirements during the course of the model test.
Specific program rules for which we proposed waivers under TEAM to support provider and supplier efforts to increase quality and decrease episode spending and for which we invited comments are included in the sections that follow. We proposed that these waivers of program rules would apply to the care of beneficiaries who are in episodes at the time when the waiver is used to bill for a service that is furnished to the beneficiary, even if the episode is later cancelled as described in section X.A.3.b.(5)(e) of the preamble of this final rule. Finally, we proposed that if a service is found to have been billed and paid by Medicare under circumstances only allowed by a program rule waiver for a beneficiary not in TEAM at the time the service was furnished, CMS would recover payment for that service from the provider or supplier who was paid, and require that provider and supplier to repay the beneficiary for any coinsurance previously collected.
We expect that the broadly defined episodes with a duration of 30 days following an anchor hospitalization or anchor procedure discharge, as discussed in section X.A.3.b. of the preamble of this final rule, would result in TEAM participants redesigning care by increasing care coordination and management of beneficiaries following discharge from an anchor hospitalization or anchor procedure. This result would require TEAM participants to pay close attention to any underlying medical conditions that could be affected by the anchor hospitalization or anchor procedure and improving coordination of care across care settings and providers. Beneficiaries may have mobility limitations during certain episodes following discharge to their home or place of residence that may interfere with their ability to travel easily to physicians' offices or other health care settings. Increasing beneficiary adherence to and engagement with recommended treatment and follow-up care following discharge from the hospital or PAC setting would be important to high quality episode care. Evidence exists to support the use of home visits among Medicare beneficiaries in improving clinical outcomes and reducing readmissions following hospital discharge. [ 1004 1005 ] In addition, we believe the financial incentives in TEAM would encourage hospitals to closely examine the most appropriate PAC settings for beneficiaries, taking into consideration beneficiary choice and location of beneficiary home or place of residence, so that the clinically appropriate setting of the lowest acuity is recommended following discharge from the anchor hospitalization or anchor procedure. We expect that all these considerations would lead to greater interest on the part of hospitals and other providers and suppliers caring for TEAM beneficiaries in furnishing services to beneficiaries in their home or place of residence. Such services could include visits by licensed clinicians other than physicians and nonphysician practitioners.
In order for Medicare to pay for home health services, a beneficiary must be determined to be “home-bound” Specifically, sections 1835(a) and 1814(a) of the Act require that a physician certify (and recertify) that in the case of home health services under the Medicare home health benefit, such services are or were required because the individual is or was “confined to the home” and needs or needed skilled nursing care on an intermittent basis, or physical or speech therapy or has or had a continuing need for occupational therapy. A beneficiary is considered to be confined to the home if the beneficiary has a condition, due to an illness or injury, that restricts his or her ability to leave home except with the assistance of another individual or the aid of a supportive device (that is, crutches, a cane, a wheelchair or a walker) or if the beneficiary has a condition such that leaving his or her home is medically contraindicated. While a beneficiary does not have to be bedridden to be considered confined to the home, the condition of the beneficiary must be such that there exists a normal inability to leave home and leaving the home requires a considerable and taxing effort by the beneficiary. Absent this condition, it would be expected that the beneficiary could typically get the same services in an outpatient or other setting. Thus, the homebound requirement provides a way to help differentiate between patients that require medical care at home versus patients who could more appropriately receive care in a less costly outpatient setting. Additional information regarding the homebound requirement is available in the Medicare Benefit Manual (Pub 100-02); Chapter 7, “Home ( print page 69828) Health Services,” Section 30.1.1, “Patient Confined to the Home.”
We considered whether a waiver of the homebound requirement would be appropriate under TEAM. Waiving the homebound requirement would allow additional beneficiaries to receive home health care services in their home or place of residence. As previously discussed, physician certification that a beneficiary meets the homebound requirement is a prerequisite for Medicare coverage of home health services, and waiving the homebound requirement could result in lower episode spending in some instances. For example, if a beneficiary is allowed to have home health care visits, even if the beneficiary is not considered homebound, the beneficiary may avoid a hospital readmission. All other requirements for the Medicare home health benefit would remain unchanged. Thus, under such a waiver, only beneficiaries who otherwise meet all program requirements to receive home health services would be eligible for coverage of home health services without being homebound.
However, we did not propose to waive the homebound requirement under TEAM for several reasons. Based on the typical clinical course of beneficiaries after certain surgical procedures, we believe that many beneficiaries would meet the homebound requirement for home health services immediately following discharge from the anchor hospitalization or following discharge to their home or place of residence from a SNF that furnished PAC services immediately following the hospital discharge, so they could receive medically necessary home health services under existing program rules. Home health agencies (HHAs) are paid a national, standardized 30-day period payment rate if a period of care meets a certain threshold of home health visits. 30-day periods of care that do not meet the visit threshold are paid a per-visit payment rate for the discipline providing care. For those TEAM beneficiaries who could benefit from home visits by a licensed clinician for purposes of assessment and monitoring of their clinical condition, care coordination, and improving adherence with treatment but who are not homebound, we do not believe that paying for these visits as home health services under Medicare is necessary or appropriate, especially given that Medicare payments for home health services are set based on the clinical care furnished to beneficiaries who are truly homebound. Finally, in other CMS episode payment models, such as BPCI Advanced and CJR, we have not waived the homebound requirement for home health services.
In the BPCI Advanced and CJR models, we have provided a waiver of the “incident to” rule to allow a physician or nonphysician practitioner participating in care redesign under a participating provider to bill for services furnished to a beneficiary who does not qualify for Medicare coverage of home health services as set forth under § 409.42 where the services are furnished in the beneficiary's home during the episode after the beneficiary's discharge from an acute care hospital. The “incident to” rule is set forth in § 410.26(b)(5), which requires services and supplies furnished incident to the service of a physician or other practitioner must be provided under the direct supervision (as defined at § 410.32(b)(3)(ii)) of a physician or other practitioner.
In the BPCI Advanced and CJR models, the waiver is available only for services that are furnished by licensed clinical staff under the general supervision (as defined at § 410.32(b)(3)(i)) of a physician (or other practitioner), or other qualified health care professional, and who are allowed by law, regulation, and facility policy to perform or assist in the performance of a specific professional service, but do not individually report that professional service. While the services may be furnished by licensed clinical staff, they must be billed by the physician (or other practitioner) or participant to which the supervising physician has reassigned their billing rights in accordance with CMS instructions using a Healthcare Common Procedures Coding System (HCPCS) G-code created by CMS specifically for the BPCI Advanced or CJR model. In the case of the incident to waiver under BPCI Advanced, the waiver allows physician and nonphysician practitioners to furnish the services up to 13 home visits during each 90-day clinical episode. In the case of the incident to waiver under CJR, the waiver allows physician and nonphysician practitioners to furnish the services up to 9 home visits during each 90-day clinical episode. All other Medicare coverage and payment criteria must be met for both BPCI Advanced and CJR models.
We considered waiving the “incident to” rule set forth in § 410.26(b)(5) for TEAM, similar to the BPCI Advanced and CJR models, however, we reviewed this specific waiver utilization and found that there was very low uptake in these models. While waiving the “incident to” rule set forth in § 410.26(b)(5) could be beneficial in furnishing services to beneficiaries in their home or place of residence, we believe there has been a greater shift towards telemedicine as a modality for post-discharge follow-up, especially after the COVID-19 public health emergency which drove greater adoption and standard practice of telehealth services. Evidence suggests that telemedicine post-discharge visits were effective, safe, and did not negatively affect health care utilization as compared to in-person visits. [ 1006 1007 ] For these reasons, we did not propose to waive the “incident to” rule set forth in § 410.26(b)(5) for TEAM, but we sought comment if we should waive the “incident to” rule set forth in § 410.26(b)(5), if we should consider modifications or alternatives to this waiver, and how we could make this waiver beneficial to TEAM participants and beneficiaries.
The following is a summary of the public comments we received on the homebound requirement, the “incident to” rule, and other provisions related to the home health waivers that we considered, and our responses to those comments:
Comment: Some commenters requested that we waive the Medicare requirement that beneficiaries must be homebound in order for Medicare to cover home health services. Some of the commenters cited the experiences of ACOs which have been permitted to provide home health services covered by Medicare to beneficiaries who do not meet the homebound requirements, suggesting that these home visits can benefit patients beyond those that are homebound.
Response: We thank the commenters for their requests. We acknowledge the potential benefits of home health services for some patients who are not homebound. However, we believe that it is necessary to consider these potential benefits in light of clinical expectations and existing payment structures as described in the proposed rule at 89 FR 36464 . First, we expect that the typical clinical course of beneficiaries after certain surgical procedures will result in many TEAM beneficiaries meeting the homebound requirement immediately following hospital discharge. Second, ( print page 69829) we recognize that home health agencies (HHAs) are paid for their services based on standardized rates—either per 30-day period or per visit depending on volume—which are calculated based on care provided to patients who are homebound. Thus, we do not feel it is necessary or appropriate for Medicare to pay for home health services for TEAM beneficiaries who are not homebound. We will monitor utilization and payment trends throughout the model test and may consider additional home health waivers in future rulemaking.
Comment: Many commenters requested that we provide a post-discharge home visit waiver that would waive the direct supervision requirement for “incident to” services as defined in § 410.26(b)(5), thereby permitting the provision of home health services to TEAM beneficiaries by a wider range of health care professionals. Many commenters specifically requested that we provide these flexibilities through a Post-Discharge Home Visits waiver as offered in BPCI Advanced. Some commenters cited the Care Management Home Visit waiver offered in the Next Generation ACO Model. A commenter further requested that any potential home health waivers include coverage of home health services delivered electronically or in a community setting outside of a patient's home. A commenter cited a shift from institutional to home-based PAC services as a motivating factor in increasing home health care access.
Response: We thank the commenters for their recommendations and acknowledge the additional care management flexibility afforded to providers through a waiver of the direct supervision requirement for “incident to” services. However, we are not convinced that there is a need for such a waiver in TEAM. As discussed in the preamble of the proposed rule at 89 FR 36465 , a review of “incident to” waiver utilization in BPCI Advanced and CJR—the most direct precedents for TEAM—indicated very low uptake of this waiver in these models. Further, while we acknowledge a shift from institutional to home-based PAC, we also recognize a shift toward telemedicine and away from home health for post-discharge follow-up and acknowledge evidence that telemedicine post-discharge visits were effective, safe, and did not negatively affect health care utilization compared to in-person visits, as cited in the preamble of the proposed rule at 89 FR 36465 . We thus maintain that there is not sufficient evidence to suggest the necessity of a post-discharge home visit or “incident to” waiver in TEAM. However, we will monitor post-discharge care utilization and outcomes throughout the model test and may consider such a waiver in future rulemaking.
After consideration of the public comments we received, we are finalizing our proposal, without modification, to maintain the existing Medicare requirements for home health services, including the requirement that the beneficiary be homebound, when home health services are furnished to TEAM beneficiaries.
As discussed in the previous section, we expect that the proposed TEAM design features would lead to greater interest on the part of hospitals and other providers and suppliers caring for TEAM beneficiaries in furnishing services to beneficiaries in their home or place of residence, including physicians' professional services. TEAM would create new incentives for comprehensive episode care management for beneficiaries, including early identification and intervention regarding changes in health status following discharge from the anchor hospitalization or anchor procedures. Given that we are not waiving the “incident to” rule set forth in § 410.26(b)(5) for TEAM, we understand that TEAM participants may still want to engage physicians in furnishing timely visits to homebound or non-homebound TEAM beneficiaries in their homes or places of residence to address concerning symptoms or observations raised by beneficiaries themselves, clinicians furnishing home health services, or licensed clinicians furnishing post-discharge home visits, while physicians committed to TEAM care redesign may not be able to revise their practice patterns to meet this home visit need for TEAM beneficiaries.
Under section 1834(m) of the Act, Medicare pays for telehealth services furnished by a physician or practitioner under certain conditions even though the physician or practitioner is not in the same location as the beneficiary. The telehealth services must be furnished to a beneficiary located in one of the eight types of originating sites specified in section 1834(m)(4)(C)(ii) of the Act and the site must satisfy at least one of the requirements of section 1834(m)(4)(C)(i)(I) through (III) of the Act. Generally, for Medicare payment to be made for telehealth services under the Medicare Physician Fee Schedule several conditions must be met, as set forth under § 410.78(b). Specifically, the service must be on the Medicare list of telehealth services and meet all of the following other requirements for payment:
- The service must be furnished via an interactive telecommunications system.
- The service must be furnished to an eligible telehealth individual.
- The individual receiving the services must be in an eligible originating site.
When all of these conditions are met, Medicare pays a facility fee to the originating site and provides separate payment to the distant site practitioner for the service. Section 1834(m)(4)(F)(i) of the Act defines Medicare telehealth services to include professional consultations, office visits, office psychiatry services, and any additional service specified by the Secretary, when furnished via a telecommunications system. For the list of approved Medicare telehealth services, see the CMS website at https://www.cms.gov/medicare/coverage/telehealth/list-services . Under section 1834(m)(4)(F)(ii) of the Act, CMS has an annual process to consider additions to and deletions from the list of telehealth services. We do not include any services as telehealth services when Medicare does not otherwise make a separate payment for them.
Some literature suggests the benefits of telehealth technologies that enable health care providers to deliver care to patients in locations remote from providers are being increasingly used to complement face-to-face patient-provider encounters to increase access to care, especially in rural or underserved areas. [ 1008 ] In these cases, the use of remote access technologies may improve the accessibility and timeliness of needed care, increase communication between providers and patients, enhance care coordination, and improve the efficiency of care. We note that certain professional services that are commonly furnished remotely using telecommunications technology are paid under the same conditions as in-person physicians' services, and thus do not require a waiver to be considered as telehealth services. Such services that do not require the patient to be present in person with the practitioner when they are furnished are covered and paid in the same way as services delivered without the use of telecommunications technology when the practitioner is in person at the medical facility furnishing care to the patient.
In other CMS episode-based payment models, such as the BPCI Advanced and ( print page 69830) CJR models, participants were permitted to use telehealth waivers that applied to two provisions:
- CMS waived the geographic site requirements under 1834(m)(4)(C)(i)(I) through (III) of the Act which allowed telehealth services to be furnished to eligible telehealth individuals when they are located at one of the eight originating sites at the time the service is furnished via a telecommunications system but without regard to the site meeting one of the geographic site requirements.
- CMS waived the originating site requirements under section 1834(m)(4)(C)(ii)(I) through (VIII) of the Act which allowed the eligible telehealth individual to not be in an originating site when the otherwise eligible individual is receiving telehealth services in their home or place of residence.
These telehealth waivers allowed providers and suppliers furnishing services to model beneficiaries to utilize telemedicine for beneficiaries that are not classified as rural and allowed the greatest degree of efficiency and communication between providers and suppliers and beneficiaries by allowing beneficiaries to receive telehealth services at their home or place of residence. We believe similar telehealth waivers would be essential to maximize the opportunity to improve the quality of care and efficiency for episodes of care in TEAM.
Specifically, like the telehealth waivers in the BPCI Advanced and CJR models, we proposed to waive the geographic site requirements of section 1834(m)(4)(C)(i)(I) through (III) of the Act that limit telehealth payment to services furnished within specific types of geographic areas or in an entity participating in a federal telemedicine demonstration project approved as of December 31, 2000. Waiver of this requirement would allow beneficiaries located in any region to receive services related to the episode to be furnished via telehealth, as long as all other Medicare requirements for telehealth services are met. Any service on the list of Medicare approved telehealth services and reported on a claim that is not excluded from the proposed episode definition (see section X.A.3.b. of the preamble of this final rule) could be furnished to a TEAM beneficiary, regardless of the beneficiary's geographic location. Under TEAM, this waiver would support care coordination and increasing timely access to high quality care for all TEAM beneficiaries, regardless of geography. Additionally, we proposed for TEAM waiving the originating site requirements of section 1834(m)(4)(C)(ii)(I)-(VIII) of the Act that specify the particular sites at which the eligible telehealth individual must be located at the time the service is furnished via a telecommunications system. Specifically, we proposed to waive the requirement only when telehealth services are being furnished in the TEAM beneficiary's home or place of residence during the episode. Any service on the list of Medicare approved telehealth services that is not excluded from the proposed episode definition (see section X.A.3.b.(5)(a) of the preamble of this final rule) could be furnished to a TEAM beneficiary in their home or place of residence, unless the service's HCPCS code descriptor precludes delivering the service in the home or place of residence. For example, subsequent hospital care services could not be furnished to beneficiaries in their home since those beneficiaries would not be inpatients of the hospital.
The existing set of codes used to report evaluation and management (E/M) visits are extensively categorized and defined by the setting of the service, and the codes describe the services furnished when both the patient and the practitioner are located in that setting. Section 1834(m) of the Act provides for particular conditions under which Medicare can make payment for office visits when a patient is located in a health care setting (the originating sites authorized by statute) and the eligible practitioner is located elsewhere. However, we do not believe that the kinds of E/M services furnished to patients outside of health care settings via real-time, interactive communication technology are accurately described by any existing E/M codes. This would include circumstances when the patient is located in his or her home and the location of the practitioner is unspecified. In order to create a mechanism to report E/M services accurately, the BPCI Advanced and CJR models created specific sets of HCPCS G-codes to describe the E/M services furnished to the model beneficiaries in their homes via telehealth. Similarly for TEAM, we proposed to create a specific set of nine HCPCS G-codes to describe the E/M services furnished to TEAM beneficiaries in their homes via telehealth. If the proposed TEAM is finalized, we would specify the precise G-code created for TEAM and share them to TEAM participants prior to the first performance year.
Among the existing E/M visit services, we envision these services would be most similar to those described by the office and other outpatient E/M codes. Therefore, we proposed to structure the new codes similarly to the office/outpatient E/M codes but adjusted to reflect the location as the beneficiary's residence and the virtual presence of the practitioner. Specifically, we proposed to create a parallel structure and set of descriptors currently used to report office or other outpatient E/M services, see Table X.A.-15, for CPT codes 99201 through 99205 for new patient visits and CPT codes 99212 through 99215 for established patient visits. For example, the proposed G- code for a level 3 E/M visit for an established patient would be a telehealth visit for the evaluation and management of an established patient in the patient's home, which requires at least 2 of the following 3 key components:
- An expanded problem focused history;
- An expanded problem focused examination;
- Medical decision making of low complexity.
Counseling and coordination of care with other physicians, other qualified health care professionals or agencies are provided consistent with the nature of the problem(s) and the patient's or family's needs or both. Usually, the presenting problem(s) are of low to moderate severity. Typically, 15 minutes are spent with the patient or family or both via real-time, audio and video intercommunications technology.

We note that we did not propose a G-code to parallel the level 1 office/outpatient visit for an established patient, since that service does not require the presence of the physician or other qualified health professional.
We proposed to develop payment rates for these new telehealth G-codes for E/M services in the patient's home that are similar to the payment rates for the office/outpatient E/M services, since the codes will describe the work involved in furnishing similar services. Therefore, we proposed to include the resource costs typically incurred when services are furnished via telehealth. In terms of the relative resource costs involved in furnishing these services, we believe that the efficiencies of virtual presentation generally limit resource costs other than those related to the professional time, intensity, and malpractice risk to marginal levels. Therefore, we proposed to adopt work and malpractice (MP) RVUs associated with the corresponding level of office/outpatient codes as the typical service because the practitioner's time and intensity and malpractice liabilities when conducting a visit via telehealth are comparable to the office visit. We would include final RVUs under the CY 2026 Medicare Physician Fee Schedule for PY 1. Additionally, we proposed to update these values each performance year to correspond to final values established under the Medicare Physician Fee Schedule.
We considered whether each level of visit typically would warrant support by auxiliary licensed clinical staff within the context of TEAM. The cost of such staff and any associated supplies, for example, would be incorporated in the practice expense (PE) RVUs under the PFS. For the lower-level visits, levels 1 through 3 for new and 2 and 3 for established visits, we did not believe that the visit would necessarily require auxiliary medical staff to be available in the patient's home. We anticipated these lower-level visits would be the most commonly furnished and would serve as a mechanism for the patient to consult quickly with a practitioner for concerns that can be easily described and explained by the patient. We did not propose to include PE RVUs for these services, since we do not believe that virtual visits envisioned for this model typically incur the kinds of costs included in the PE RVUs under the Medicare Physician Fee Schedule. For higher level visits, we typically would anticipate some amount of support from auxiliary clinical staff. For example, wound examination and minor wound debridement would be considered included in an E/M visit and would require licensed clinical staff to be present in the beneficiary's home during the telehealth visit in order for the complete service to be furnished. We believed it would be rare for a practitioner to conduct as complex and detailed a service as a level 4 or 5 E/M home visit via telehealth for TEAM beneficiaries in episodes without licensed clinical staff support in the home.
We have considered support by auxiliary clinical staff to be typical for level 4 or 5 E/M visits furnished to TEAM beneficiaries in the home via telehealth, however, we did not propose to incorporate these costs through PE RVUs. Given the anticipated complexity of these visits, we would expect to observe level 4 and 5 E/M visits to be reported on the same claim with the same date of service as a home visit or during a period of authorized home health care. If neither of these occurs, we proposed to require the physician to document in the medical record that auxiliary licensed clinical staff were available on site in the patient's home during the visit and if they were not, to document the reason that such a high-level visit would not require such personnel.
We noted that because the services described by the proposed G-codes, by definition, are furnished remotely using telecommunications technology, they therefore are paid under the same conditions as in-person physicians' services, and they do not require a waiver to the requirements of section 1834(m) of the Act. We also noted that because these home telehealth services are E/M services, all other coverage and payment rules regarding E/M services would continue to apply.
Under TEAM, this proposal to waive the originating site requirements and create new home visit telehealth HCPCS codes would support the greatest efficiency and timely communication between providers and beneficiaries by allowing beneficiaries to receive telehealth services at their places of residence.
With respect to home health services paid under the home health prospective payment system (HH PPS), we emphasized that telehealth visits under this model cannot substitute for in-person home health visits per section 1895(e)(1)(A) of the Act. Furthermore, telehealth services by social workers cannot be furnished for TEAM beneficiaries who are in a home health episode because medical social services are included as home health services per section 1861(m) of the Act and paid for under the Medicare HH PPS. However, telehealth services permitted under section 1834 of the Act and furnished by physicians or other practitioners, specifically physician assistants, nurse practitioners, clinical nurse specialists, certified nurse midwives, nurse anesthetists, psychologists, and dieticians, can be furnished for TEAM beneficiaries who are in a home health episode. Finally, sections 1835(a) and 1814(a) of the Act require that the patient has a face-to-face encounter with the certifying physician or an allowed nonphysician practitioner (NPP) working in collaboration with or under the supervision of the certifying physician before the certifying physician certifies that the patient is eligible for home health services. Under ( print page 69832) § 424.22(a)(1)(v), the face-to-face encounter can be performed up to 90 days prior to the start of home health care or within 30 days after the start of home health care. Section 424.22(a)(1)(v)(A) also allows a physician, with privileges, who cared for the patient in an acute or PAC setting (from which the patient was directly admitted to home health) or an allowed NPP working in collaboration with or under the supervision of the acute or PAC physician to conduct the face-to-face encounter.
Although sections 1835(a) and 1814(a) of the Act allow the face-to-face encounter to be performed via telehealth, we did not propose that the waiver of the telehealth geographic site requirement for telehealth services and the originating site requirement for telehealth services furnished in the TEAM beneficiary's home or place of residence would apply to the face-to- face encounter required as part of the home health certification when that encounter is furnished via telehealth. In other words, when a face-to-face encounter furnished via telehealth is used to meet the requirement for home health certification, the usual Medicare telehealth rules apply with respect to geography and eligibility of the originating site. We expect that this policy would not limit TEAM beneficiaries' access to medically necessary home health services because beneficiaries receiving home health services during an episode will have had a face-to-face encounter with either the physician or an allowed NPP during their anchor hospitalization or a physician or allowed NPP during a post-acute facility stay prior to discharge directly to home health services.
Under the proposed waiver of the geographic site requirement and originating site requirement, all telehealth services would be required to be furnished in accordance with all Medicare coverage and payment criteria, and no additional payment would be made to cover set-up costs, technology purchases, training and education, or other related costs. The facility fee paid by Medicare to an originating site for a telehealth service would be waived if there is no facility as an originating site (that is, the service was originated in the beneficiary's home). Finally, providers and suppliers furnishing a telehealth service to a TEAM beneficiary in his or her home or place of residence during the episode would not be permitted to bill for telehealth services that were not fully furnished when an inability to provide the intended telehealth service is due to technical issues with telecommunications equipment required for that service. Beneficiaries would be able to receive services furnished pursuant to the telehealth waivers only during the episode.
We plan to monitor patterns of utilization of telehealth services under TEAM to monitor for overutilization or reductions in medically necessary care, and significant reductions in face-to-face visits with physicians and NPPs. We plan to specifically monitor the distribution of new telehealth home visits that we are proposing, as we anticipate greater use of lower-level visits. Given our concern that auxiliary licensed clinical staff be present for level 4 and 5 visits, we will monitor our proposed requirement that these visits be billed on the same claim with the same date of service as a home nursing visit, during a period authorized home health care, or that the physician document the presence of auxiliary licensed clinical staff in the home or an explanation as to the specific circumstances precluding the need for auxiliary staff for the specific visit. We sought comments on the proposed waivers with respect to telehealth services, and the proposed creation of the home visit telehealth codes.
The following is a summary of the public comments we received on the proposed telehealth waivers and creation of home visit telehealth codes and our responses to those comments:
Comment: Many commenters expressed support for the proposed telehealth waivers. Some of these commenters indicated that these waivers would facilitate care coordination, improve access to services, and promote equity. A couple commenters lauded the consistency of these waivers with those offered under BPCI Advanced. A few commenters expressed specific support for the proposed waiver of the originating site requirements. A commenter expressed specific support for the proposed waiver of the geographic site requirements.
Response: We thank the commenters for their support and concur that the proposed waivers can improve care access and equity, facilitate care coordination, and provide continuity for providers with experience utilizing waivers in BPCI Advanced.
Comment: A commenter requested that CMS make the proposed telehealth waivers available for all TEAM procedures, as in CJR and BPCI Advanced.
Response: We thank the commenter for this recommendation. We would like to note that the usage of the proposed telehealth waivers under TEAM, pursuant to the requirements outlined in § 512.580, is not restricted to certain TEAM clinical episodes.
Comment: A commenter suggested that we add a waiver permitting the delivery of physical therapist services via telehealth, noting the importance of initiating physical therapy soon after surgery. The commenter suggested that such a waiver could benefit individuals facing delays in care by allowing for the timely initiation of physical therapy following discharge.
Response: We thank the commenter for their suggestion and acknowledge the importance of physical therapy for many beneficiaries following a surgical procedure. We will take this recommendation into consideration and may propose such a waiver in future rulemaking if we believe it would improve beneficiary care and further the goals of the model.
Comment: A couple commenters provided input on the proposed creation of the home visit telehealth codes.
A commenter expressed support for the proposal to create new telehealth G-codes and HCPCS codes and corresponding payment rates for home health, indicating that telehealth can increase access to post-surgical care, particularly for patients facing barriers to access. This commenter further suggested that we permanently extend the new payment rates for these telehealth services beyond TEAM.
A commenter opposed the proposal to create new telehealth codes and lower payment rates for home health services in TEAM. This commenter indicated that the existing telehealth visit codes developed by the Current Procedural Terminology (CPT®) Editorial Panel should be available for use by all physicians.
Response: We thank the commenters for their responses regarding the proposed creation of TEAM-specific telehealth G-codes. As stated in the proposed rule at 89 FR 36466 , we recognize that the existing set of codes for evaluation and management (E/M) visits are categorized and defined by the setting of service and describe services provided when both the patient and the practitioner are located in that setting. As a result, and with consideration of the comments received, we continue to believe that the kinds of E/M services furnished to patients outside of health care settings via real-time, interactive communication technology are not accurately described by any existing E/M codes. Further, we note that the creation of model-specific telehealth G-codes for BPCI Advanced and CJR provide precedent for the establishment of such codes for TEAM. ( print page 69833)
Comment: A couple commenters suggested that we apply the proposed telehealth waivers to all Medicare Fee-for-Service (FFS) beneficiaries with billing codes that could trigger a TEAM episode at participating hospitals. One of the commenters indicated that waivers are often underutilized due to concerns that a patient may not qualify as a model beneficiary.
Response: We thank the commenters for their suggestion and recognize the potential challenges that TEAM participants may face in identifying beneficiaries who will trigger a TEAM episode. We believe that certain provisions already included in the proposed rule will mitigate these concerns for participants. Regarding the telehealth waivers, we note that covered telehealth services would be provided after discharge from the anchor stay or completion of the anchor procedure, and correspondingly expect that providers would have a working knowledge of their patients' TEAM beneficiary status as part of their care coordination. As a result, and in further acknowledgement of the administrative challenge that would be imposed on CMS to track TEAM beneficiary status in real time, we do not believe it is necessary or appropriate to automatically apply the telehealth waivers to all FFS beneficiaries that could trigger a TEAM episode at a participating hospital.
After consideration of the comments received, we are finalizing at § 512.580(a) the waivers pertaining to telehealth services as proposed. We are also finalizing at § 512.580(a)(3)(ii) the creation of telehealth home visit HCPCS codes as proposed.
Pursuant to section 1861(i) of the Act, a beneficiary must have a prior inpatient hospital stays of no fewer than 3 consecutive days to be eligible for Medicare coverage of inpatient SNF care. We refer to this as the SNF 3-day rule. We noted that the SNF 3-day rule has been waived for Medicare SNF coverage under other episode payment models, including the BPCI Advanced the CJR models. Model participants that elect to use the waiver can discharge model beneficiaries in fewer than 3 days from an anchor hospital stay or anchor procedure (in the case of the CJR model) to a SNF, where services are covered under Medicare Part A if all other coverage requirements for such services are satisfied.
Episode-based payment models like BPCI Advanced and CJR have the potential to mitigate the existing incentives under the Medicare program to overuse SNF benefits for beneficiaries, as well as to furnish many fragmented services that do not reflect significant coordinated attention to and management of complications following hospital discharge. These model participants considering the early discharge of a beneficiary pursuant to the waiver must evaluate whether early discharge to a SNF is clinically appropriate and SNF services are medically necessary. Next, they must balance that determination and the potential benefits to the hospital in the form of internal cost savings due to greater financial efficiency with the understanding that a subsequent hospital readmission, attributable to premature discharge or low quality SNF care, could substantially increase episode spending while also resulting in poorer quality of care for the beneficiary. Furthermore, early hospital discharge for a beneficiary who would otherwise not require a SNF stay (that is, the beneficiary has no identified skilled nursing or rehabilitation need that cannot be provided on an outpatient basis) following a hospital stay of typical length does not improve episode efficiency.
Because of the potential benefits we see for TEAM participants, their provider partners, and beneficiaries, we proposed to waive the SNF 3-day rule for coverage of a SNF stay following the anchor hospitalization or anchor procedure under TEAM. We proposed to use our authority under section 1115A of the Act with respect to certain SNFs that furnish Medicare Part A post-hospital extended care services to beneficiaries included in an episode in TEAM. We believe this waiver is necessary to the model test so that TEAM participants can redesign care throughout the episode continuum of care extending to 30 days post-discharge from the anchor hospital stay or anchor procedure to maximize quality and hospital financial efficiency, as well as reduce episode spending under Medicare. All other Medicare rules for coverage and payment of Part A-covered SNF services would continue to apply to TEAM beneficiaries in all performance years of the model. Further, to ensure protection to TEAM beneficiary safety and optimize health outcomes, we proposed to require that TEAM participants may only discharge a TEAM beneficiary under this proposed waiver of the SNF 3-day rule to a SNF rated an overall of three stars or better by CMS based on information publicly available at the time of hospital discharge from an anchor hospital stay or anchor procedure. Problem areas due to early hospital discharge may not be discovered through model monitoring and evaluation activities until well after the episode has concluded, and the potential for later negative findings alone may not afford sufficient beneficiary protections. CMS created a Five-Star Quality Rating System for SNFs to allow SNFs to be compared more easily and to help identify areas of concerning SNF performance. The Nursing Home Compare website gives each SNF an overall rating of between 1 and 5 stars. [ 1009 ] Those SNFs with 5 stars are considered to have much above average quality, and SNFs with 1 star are considered to have quality much below average. Published SNF ratings include distinct ratings of health inspection, staffing, and quality measures, with ratings for each of the three sources combined to calculate an overall rating. These areas of assessment are all relevant to the quality of SNF care following discharge from the anchor hospitalization or anchor procedure initiating an episode, especially if that discharge occurs after fewer than 3 days in the hospital. Because of the potential greater risks following early inpatient hospital discharge, we believe it is appropriate that all TEAM beneficiaries discharged from the TEAM participant to a SNF in fewer than 3 days be admitted to a SNF that has demonstrated that it can provide quality care to patients with significant unresolved post-surgical symptoms and problems. We believe such a SNF would need to provide care of at least average overall quality, which would be represented by an overall SNF 3-star or better rating.
Thus, the TEAM participant must discharge the beneficiary to a SNF that is qualified under the SNF 3-day rule waiver. We proposed that to be qualified under the SNF 3-day rule waiver a SNF must be included in the most recent calendar year quarter Five-Star Quality Rating System listing for SNFs on the Nursing Home Compare website for the date of the beneficiary's admission to the SNF. The qualified SNF must be rated an overall 3 stars or better for at least 7 of the 12 months based on a review of the most recent rolling 12 months of overall star ratings. We proposed to post on the CMS website the list of qualified SNFs in advance of the calendar quarter.
We recognized that there may be instances where a TEAM participant would like to use the SNF 3-day rule waiver, but the TEAM beneficiary receives inpatient PAC through swing ( print page 69834) bed arrangements in a hospital or Critical Access Hospital (CAH), as designated in § 485.606 of this chapter, which is not subject to the Five-Star Quality Rating System. For example, a TEAM beneficiary located in a rural area may wish to receive PAC care closer to their home but there are no qualified SNFs in their area. Allowing TEAM participants to use the SNF 3-day rule waiver for hospitals and CAHs operating under swing bed agreements may support beneficiary freedom of choice and provide greater flexibility to TEAM participants for their care coordination efforts. This approach is consistent with the Shared Savings Program, which offers a similar SNF 3-day rule waiver and allows their ACOs to partner with hospitals and CAHs to with swing bed arrangements to utilize the waiver. Therefore, we sought comment on whether we should allow TEAM participants to use hospitals and CAHs operating under swing bed agreements for the SNF 3-day rule waiver and what beneficiary protections we should include since the Five-Star Quality Rating System would not apply.
The following is a summary of the public comments we received on the possibility of including swing bed arrangements under the SNF 3-day rule waiver and our responses to these comments:
Comment: A commenter recommended that we allow TEAM participants to use the SNF 3-day rule waiver for Critical Access Hospitals (CAHs) providing post-acute care (PAC) under swing bed arrangements. The commenter indicated that including swing beds under the SNF 3-day rule waiver would increase access to PAC services for beneficiaries in rural areas or areas with health care shortages.
Response: We thank the commenter for the recommendation. We would like to clarify that the option of including swing beds under the SNF 3-day rule waiver for TEAM participants was considered but not proposed in the proposed rule. While we agree that including swing bed arrangements under the SNF 3-day rule waiver could promote freedom of choice and PAC access for beneficiaries in areas with limited SNF options, we remain concerned about the lack of a standard and comprehensive quality assessment metric for hospitals and CAHs operating under swing bed arrangements, which are not subject to the Five-Star Quality Rating System. As stated in the preamble of the proposed rule at 89 FR 36468-9 , we recognize that greater risks may be present for patients following early inpatient hospital discharge. The proposed SNF quality rating requirement for use of the SNF 3-day rule waiver offers an additional level of protection to beneficiaries following an early discharge by ensuring that all TEAM beneficiaries discharged to a SNF after a hospital stay of fewer than 3 days are admitted to a SNF that has demonstrated that it can provide quality care to patients with significant unresolved post-surgical symptoms and problems. At this time, we do not feel that an appropriate corresponding metric is in place for swing bed arrangements. However, we will continue to monitor SNF utilization throughout the model and may seek additional comment and consider alterations to the SNF 3-day rule waiver requirements in future rulemaking.
After consideration of the comments received, and as discussed further below, we are finalizing at § 512.580(b) the waiver of the SNF 3-day rule as proposed, including the provisions for determining qualified SNFs as proposed at § 512.580(b)(3), and without inclusion of swing bed arrangements.
We plan to monitor patterns of SNF utilization under the TEAM, particularly with respect to hospital discharge in fewer than 3 days to a SNF, to ensure that beneficiaries are not being discharged prematurely to SNFs and that they are able to exercise their freedom of choice without patient steering. We sought comment on our proposal to waive the SNF 3-day stay rule for stays in SNFs rated overall as 3 stars or better following discharge from the anchor hospitalization or anchor procedures for episodes in TEAM.
The following is a summary of the public comments we received on the proposal to waive the SNF 3-day rule for stays in SNFs rated overall as 3 stars or better and our responses to these comments:
Comment: Many commenters expressed support for the proposed SNF 3-day rule waiver. Some of these commenters indicated that this waiver would facilitate care coordination, improve access to services, and promote equity. A couple commenters lauded the consistency of this waiver with that offered under BPCI Advanced.
Response: We thank the commenters for their support and concur that the proposed waiver can improve care access and equity, facilitate care coordination, and provide continuity for providers with experience utilizing the SNF 3-day rule waiver in BPCI Advanced.
Comment: A couple commenters suggested that we apply the proposed SNF 3-day rule waiver to all Medicare Fee-for-Service (FFS) beneficiaries with billing codes that could trigger a TEAM episode at participating hospitals. A commenter indicated that waivers are often underutilized due to concerns that a patient may not qualify as a model beneficiary.
Response: We thank the commenters for their suggestion and recognize the potential challenges that TEAM participants may face in identifying beneficiaries who will trigger a TEAM episode. We believe that certain provisions already included in the proposed rule will mitigate these concerns for participants. Regarding the SNF 3-day rule waiver, we note in § 512.580(b)(1)-(2) that CMS determines eligibility for the SNF 3-day rule waiver based on TEAM beneficiary status on the date of discharge from the anchor hospitalization or the date of service of the anchor procedure, as applicable. As a result, and in further acknowledgement of the administrative challenge that would be imposed on CMS to track TEAM beneficiary status in real time, we do not believe it is necessary or appropriate to automatically apply the SNF 3-day rule waiver to all FFS beneficiaries that could trigger a TEAM episode at a participating hospital.
Comment: A commenter expressed support for the proposal to restrict usage of the SNF 3-day rule waiver to stays in SNFs that meet the three-star quality rating requirement.
Comment: A commenter requested that CMS make the proposed SNF 3-day rule waiver available for all TEAM procedures, as in CJR and BPCI Advanced.
Response: We thank the commenter for this recommendation. We would like to note that the usage of the SNF 3-day rule waiver under TEAM, pursuant to the requirements outlined in § 512.580, is not restricted to certain TEAM clinical episodes.
After consideration of the comments received, we are finalizing at § 512.580(b) the waiver of the SNF 3-day rule as proposed.
We believed that it will be necessary to propose beneficiary protections against financial liability in addition to the beneficiary protections discussed elsewhere in this proposed rule. Specifically, we believed it is important to discern whether a waiver applies to SNF services furnished to a particular beneficiary to ensure compliance with the conditions of the waiver and improve our ability to monitor waivers for misuse. ( print page 69835)
In considering additional beneficiary protections that may be necessary to ensure proper use of SNF 3-day rule waiver under the TEAM, we noted that there are existing, well-established payment and coverage policies for SNF services based on sections 1861(i), 1862(a)(1), and 1879 of the Act that include protections for beneficiaries from liability for certain non-covered SNF charges. These existing payment and coverage policies for SNF services continue to apply under TEAM, including SNF services furnished pursuant to the SNF 3-day rule waiver. (For example, see section 70 in the Medicare Claims Processing Manual, Chapter 30—Financial Liability Protections on the CMS website at https://www.cms.gov/regulations-and-guidance/guidance/manuals/downloads/clm104c30.pdf ; and Medicare Coverage of Skilled Nursing Facility Care https://www.medicare.gov/coverage/skilled-nursing-facility-snf-care ; Medicare Benefit Policy Manual, Chapter 8—Coverage of Extended Care (SNF) Services Under Hospital Insurance at https://www.cms.gov/regulations-and-guidance/guidance/manuals/downloads/bp102c08pdf.pdf ). In general, CMS requires that the SNF inform a beneficiary in writing about services and fees before the beneficiary is discharged to the SNF (§ 483.10(b)(6)); the beneficiary cannot be charged by the SNF for items or services that were not requested (§ 483.10(c)(8)(iii)(A)); a beneficiary cannot be required to request extra services as a condition of continued stay (§ 483.10(c)(8)(iii)(B)); and the SNF must inform a beneficiary that requests an item or service for which a charge will be made that there will be a charge for the item or service and what the charge will be (§ 483.10(c)(8)(iii)(C)). (See also section 6 of Medicare Coverage of Skilled Nursing Facility Care at https://www.cms.gov/regulations-and-guidance/guidance/manuals/downloads/bp102c06.pdf .)
As we discussed in the CJR final rule ( 80 FR 73454 through 73460 ), commenters expressed concern regarding the lag between a CJR beneficiary's Medicare coverage or eligibility status change and a TEAM participant's awareness of that change. There may be cases in which a SNF 3-day rule waiver is used by a TEAM participant because the TEAM participant believes that the beneficiary meets the inclusion criteria, based on the information available to the hospital and SNF at the time of the beneficiary's admission to the SNF, but in fact the beneficiary's Medicare coverage has changed and the hospital was unaware of it based on available information. We recognize that despite good faith efforts by TEAM participants and SNFs to determine a beneficiary's Medicare status for the model, it may occur that a beneficiary is not eligible to be included in the TEAM at the time the SNF 3-day rule waiver is used. In these cases, we will cover services furnished under the waiver when the information available to the provider at the time the services under the waiver were furnished indicated that the beneficiary was included in the model.
Based on our experience with the SNF 3-day rule waiver, including the CJR model, we believed there are situations where it would be appropriate to require additional beneficiary financial protections under the SNF 3-day rule waiver for the TEAM. Specifically, we were concerned about potential beneficiary financial liability for non-covered Part A SNF services that might be directly related to use of the SNF 3-day rule waiver under the TEAM. We were concerned that there could be scenarios where a beneficiary could be charged for non-covered SNF services that were a result of a TEAM participant's inappropriate use of the SNF 3-day rule waiver. Specifically, we were concerned that a beneficiary could be charged for non-covered SNF services if a TEAM participant discharges a beneficiary to a SNF that does not meet the quality requirement (3 stars or higher in 7 of the last 12 months), and payment for SNF services is denied for lack of a qualifying inpatient hospital stay. We recognized that requiring a discharge planning notice would help mitigate concerns about beneficiaries' potential financial liability for non-covered services. Nevertheless, we were concerned that in this scenario, once the claim is rejected, the beneficiary may not be protected from financial liability under existing Medicare rules because the waiver would not be available, and the beneficiary would not have had a qualifying inpatient hospital stay. Thus, the TEAM beneficiary could be charged by the SNF for non-covered SNF services that were a result of an inappropriate attempt to use the waiver. In this scenario, Medicare would deny payment of the SNF claim, and the beneficiary could potentially be charged by the SNF for these non-covered SNF services, potentially subjecting such beneficiaries to significant financial liability. In this circumstance, we assume the TEAM participant's intent was to rely upon the SNF 3-day rule waiver, but the waiver requirements were not met. We believe that in this scenario, the rejection of the claim could easily have been avoided if the hospital had confirmed that the requirements for use of the SNF 3-day rule waiver were satisfied or if the beneficiary had been provided the discharge planning notice and elected to go to a SNF that met the quality requirement.
The CJR model ( 82 FR 180 ) addressed beneficiary liability financial concerns for non-covered SNF services related to the waiver by generally placing the risk on the participant hospital and we believe it is appropriate to propose a similar policy for TEAM. CJR participant hospitals are generally held financially responsible for misusing the waiver in situations where waiver requirements are not met, because participant hospitals are required to be aware of the SNF 3-day rule waiver requirements. Participant hospitals are the entities financially responsible for episode spending under the model and will make the decision as to whether it is appropriate to discharge a beneficiary without a 3-day stay. In addition, the requirements for use of the SNF waiver are clearly laid out in the CJR final rule ( 80 FR 73273 ). CMS posts on the public website a list of qualifying SNFs (those with a 3-star or higher rating for 7 of the last 12 months). CJR participant hospitals are required to consult the published list of SNFs prior to utilizing the SNF 3-day rule waiver.
For participant hospitals that provide a beneficiary with the discharge planning notice, the hospital would not have financial liability for non-covered SNF services that result from inapplicability of the waiver. In other words, when the participant hospital has discharged a beneficiary to a SNF that does not qualify under the conditions of the waiver, and has not provided the required discharge planning notice so that the beneficiary is aware that he or she is accepting financial liability for non-covered SNF services as a result of not having a qualifying inpatient stay, the ultimate responsibility and financial liability for the non-covered SNF stay rests with the participant hospital. For this reason, we proposed to align with the CJR model policy and require TEAM participants to keep a record of discharge planning notice distribution to TEAM beneficiaries. We will monitor TEAM participants' use of discharge planning notices to assess the potential for their misuse.
To protect TEAM beneficiaries from being charged for non-covered SNF charges in instances when the waiver was used inappropriately, and similar to ( print page 69836) the CJR model ( 82 FR 180 ), we proposed to add certain beneficiary protection requirements that would apply for SNF services that would otherwise have been covered except for lack of a qualifying hospital stay. Specifically, we proposed that if a TEAM participant discharges a beneficiary without a qualifying 3-day inpatient stay to a SNF that is not on the published list of SNFs that meet the TEAM SNF 3-day rule waiver quality requirements as of the date of admission to the SNF, the TEAM participant will be financially liable for the SNF stay if no discharge planning notice is provided to the beneficiary, alerting them of potential financial liability. If the TEAM participant provides a discharge planning notice, then the TEAM participant will not be financially liable for the cost of the SNF stay and the normal Medicare FFS rules for coverage of SNF services will apply. In cases where the TEAM participant provides a discharge planning notice and the beneficiary chooses to obtain care from a non-qualified SNF without a qualifying inpatient stay, the beneficiary assumes financial liability for services furnished (except those that are covered by Medicare Part B during a non-covered inpatient SNF stay).
In the event a TEAM beneficiary is discharged to a SNF without a qualifying 3-day inpatient stay, but the SNF is not on the qualified list as of the date of admission to the SNF, and the TEAM participant has failed to provide a discharge planning notice, we proposed that CMS apply the following rules:
- CMS shall make no payment to the SNF for such services.
- The SNF shall not charge the beneficiary for the expenses incurred for such services; and the SNF shall return to the beneficiary any monies collected for such services.
- The hospital shall be responsible for the cost of the uncovered SNF stay.
We sought comment on these proposals. Specifically, we sought comment on whether it is reasonable to—(1) cover services furnished under the SNF waiver based on TEAM participant knowledge of beneficiary eligibility for the TEAM as determined by Medicare coverage status at the time the services under the waiver were furnished; and (2) to hold the TEAM participant financially responsible for rejected SNF claims if a TEAM beneficiary is discharged to a SNF without a qualifying 3-day inpatient stay, but the SNF is not on the qualified list as of the date of admission to the SNF, and the TEAM participant has failed to provide a discharge planning notice. Finally, we sought comment on any other related issues that we should consider in connection with these proposals to protect beneficiaries from significant financial liability for non-covered SNF services related to the waiver of the SNF 3-day rule under the proposed TEAM. We may address those issues through future notice and comment rulemaking.
The following is a summary of the public comments we received on beneficiary protection considerations in connection with the proposed waiver of the SNF 3-day rule and our responses to these comments:
Comment: A commenter expressed support for the proposal to waive responsibility for denied claims relying on the SNF 3-day rule waiver in instances where the participant would have no way of knowing that a patient would not ultimately be assigned as a TEAM beneficiary. The commenter indicated that this provision would encourage use of the waiver by ensuring coverage of SNF stays even if a patient is later excluded from the model.
Response: We thank the commenter for their support and concur that the provision to determine SNF 3-day rule waiver coverage based on TEAM beneficiary status at the time of discharge, as described in § 512.580(b), provides protection from unforeseen beneficiary ineligibility.
Comment: A commenter inquired whether it would be permissible to provide patients with a list of SNFs only including those with a quality rating of at least 3 stars, citing the 3-star requirement for coverage under the SNF 3-day rule waiver.
Response: We thank the commenter for their inquiry and suggestion. We appreciate the need to simultaneously maintain freedom of choice and protection for beneficiaries discharged to a SNF. We note that the TEAM waiver of the SNF 3-day rule represents an extension of existing PAC options covered by Medicare for TEAM beneficiaries deemed medically appropriate for discharge to a SNF after an inpatient hospital stay of less than 3 days. Medicare will continue to cover SNF stays for TEAM beneficiaries discharged to a SNF after an inpatient hospital stay of 3 days or more under existing Medicare rules, which do not include a star rating requirement.
As stated in the proposed rule, we recognize that the waiver of the SNF 3-day rule has the potential to introduce additional risk to beneficiaries through early hospital discharge. While model monitoring and evaluation activities may detect negative outcomes resulting from this risk, such results are likely to be known only after the end of the episode. The CMS Five-Star Quality Rating System for SNFs provides an additional means of beneficiary protection through the comparison and identification of SNFs based on quality of care. As stated in the proposed rule at 89 FR 36469 , we believe it is appropriate that all TEAM beneficiaries discharged from the TEAM participant to a SNF in fewer than 3 days be admitted to a SNF that has demonstrated that it can provide quality care to patients with significant unresolved post-surgical symptoms and problems. As stated in § 512.582(b).(2).(iv).(E), as part of discharge planning and referral, TEAM participants must provide a complete list of HHAs, SNFs, IRFs, or LTCHs that are participating in the Medicare program, and that serve the geographic area (as defined by the HHA) in which the patient resides, or in the case of a SNF, IRF, or LTCH, in the geographic area requested by the patient. We provide in § 512.582(a)(3)(iii) that TEAM participants may recommend preferred providers and suppliers, consistent with applicable statutes and regulations. We believe that this provision would permit TEAM participants to recommend SNF providers that meet the 3-star waiver use requirement, of which we propose to provide a list on a quarterly basis. We also note that participants are required to provide a discharge planning notice which would notify beneficiaries who are being discharged to SNF after less than 3 days in the hospital that they would not receive Medicare coverage for their stay at a SNF that does not meet the 3-star requirement for waiver usage. We expect that this would disincentivize beneficiaries in this situation from choosing non-covered SNFs. We believe that the combination of permitting participants to recommend preferred SNFs and notifying beneficiaries that stays at SNFs that do not meet the 3-star waiver use requirement would not be covered by Medicare provides sufficient means to meet the beneficiary protection goal of the 3-star provision while maintaining freedom of choice.
Comment: A commenter recommended additional provisions in response to the concern that beneficiaries could be charged for SNF services that do not meet the requirements for coverage under the SNF 3-day rule waiver. The commenter recommended that we institute a SNF affiliate agreement and that we include a beneficiary notice that TEAM participants would not be responsible for SNF stays that do not meet the ( print page 69837) quality rating requirement for use of the SNF 3-day rule waiver.
Response: We thank the commenter for their suggestions and concur that it is necessary to provide beneficiary protections surrounding coverage for SNF services under the SNF 3-day rule waiver. Regarding beneficiary notice, we note that a TEAM participant would be required in § 512.580(b)(3) to provide a discharge planning notice which, per § 512.580(b)(3)(ii), must include notification that the beneficiary would be responsible for payment for services rendered during a SNF stay that would not be covered by Medicare Part B due to not meeting the SNF 3-day rule waiver requirements outlined in § 512.580. Regarding agreements between TEAM participants and SNFs, we note that TEAM participants would be able to enter into financial arrangements with TEAM collaborators pursuant to the requirements defined in § 512.565. In light of these provisions and the expected administrative burden associated with a separate SNF affiliate agreement, we do not believe it is necessary to institute such an agreement at this time. We will continue to monitor SNF utilization during the model test and may consider alterations to the SNF 3-day rule waiver provisions in future rulemaking.
After consideration of the comments received, we are finalizing at § 512.580(b) all provisions regarding the SNF 3-day rule waiver as proposed, including the financial liability provisions as proposed at § 512.580(b)(5).
We proposed TEAM as we believe it is an opportunity to improve the quality of care and that the policies of the model support making care more easily accessible to consumers when and where they need it, increasing consumer engagement and thereby informing consumer choices. For example, under this model we proposed certain waivers which would offer TEAM participants additional flexibilities with respect to furnishing telehealth services and care in SNFs, as discussed in section X.A.3.h. of the preamble of this final rule. We believe that this model will improve beneficiary access and outcomes. Conversely, we do note that these same opportunities could be used to try to steer beneficiaries into lower cost services without an appropriate emphasis on maintaining or increasing quality. We direct readers to section X.A.3.d.(5) of the preamble of this final rule for discussion of the methodology for calculating the reconciliation payment amount or repayment amount under this model. We believe that existing Medicare provisions can be effective in protecting beneficiary freedom of choice and access to appropriate care under TEAM. However, because TEAM is designed to promote care delivery efficiencies for episodes, providers may seek greater control over the continuum of care and, in some cases, could attempt to direct beneficiaries into care pathways that save money at the expense of beneficiary choice or even beneficiary outcomes. As such, we acknowledge that some additional safeguards may be necessary under TEAM for program integrity purposes as providers are simultaneously seeking opportunities to decrease costs and utilization. We believe that it is important to consider any possibility of adverse consequences to patients and to ensure that sufficient controls are in place to protect Medicare beneficiaries in episodes under TEAM.
Because we have proposed that hospitals in selected geographic areas would be required to participate in the model, individual beneficiaries would not be able to opt out of TEAM when they receive care from a TEAM participant in the model. We do not believe that it is consistent with other Medicare programs to allow patients to opt out of a payment system that is unique to a particular geographic area. For example, the state of Maryland has a unique payment system under Medicare, but that payment system does not create an alternative care delivery system, and we do not expect it in any way to impact beneficiary decisions. Moreover, we do not believe that an ability to opt out of a payment system is a critical factor in upholding beneficiary choice if other safeguards are in place given that this model does not increase beneficiary cost-sharing. However, a beneficiary is not precluded from seeking care from providers or suppliers who do not participate in TEAM. We do believe that full notification and disclosure of the payment model and its possible implications is critical for beneficiary understanding and protection. It is important to create safeguards for beneficiaries to ensure that care recommendations are based on clinical needs and not inappropriate cost savings. It is also important for beneficiaries to know that they can raise any concerns with their clinicians, with 1-800-Medicare, or with their local Quality Improvement Organizations (QIOs).
In the proposed rule we stated that TEAM would not limit a beneficiary's ability to choose among Medicare providers or limit Medicare's coverage of items and services available to the beneficiary. Beneficiaries may continue to choose any Medicare participating provider, or any provider who has opted out of Medicare, with the same costs, copayments and responsibilities as they have with other Medicare services. The model would allow TEAM participants to enter into TEAM sharing arrangements, as discussed in section X.A.3.g.(4) of the preamble of this final rule, with certain providers and these preferred providers may be recommended to beneficiaries as long as those recommendations are made within the constraints of current law. However, TEAM participants may not limit beneficiaries to a preferred or recommended providers list that is not compliant with restrictions existing under current statutes and regulations.
Moreover, we indicated in the proposed rule that TEAM participants may not charge any TEAM collaborator, as discussed in section X.A.3.g.(3) of the preamble of this final rule, a fee to be included on any list of preferred providers or suppliers, nor may the hospital accept such payments, which would be considered to be outside the realm of risk-sharing agreements. Thus, TEAM does not create any restriction of beneficiary freedom to choose providers, including surgeons, hospitals, post-acute care or any other providers or suppliers. Moreover, as TEAM participants redesign care pathways, it may be difficult for providers to sort individuals based on health care insurance and to treat them differently. We anticipate that care pathway redesign occurring in response to the model will increase coordination of care, improve the quality of care, and decrease costs for all patients, not just for Medicare beneficiaries. We anticipate this broader care delivery impact to all patients may further promote consistent treatment of all beneficiaries.
In the proposed rule we stated we believe that beneficiary notification and engagement is essential because there will be a change in the way participating hospitals are paid. We believe that appropriate beneficiary notification should explain the model, advise patients of both their clinical needs and their care delivery choices, and should clearly specify any providers, suppliers, and ACOs holding a sharing arrangement with the TEAM participant should be identified to the beneficiary as a “financial partner of the hospital for the purposes of ( print page 69838) participation in TEAM.” These policies seek to enhance beneficiaries' understanding of their care, improve their ability to share in the decision-making, and ensure that they have the opportunity to consider competing benefits even as they are presented with cost-saving recommendations. We believe that appropriate beneficiary notification should do all of the following:
- Explain the model and how it will or will not impact the beneficiary's care.
- Inform patients that they retain freedom of choice to choose providers and services.
- Explain how patients can access care records and claims data through an available patient portal and through sharing access to caregivers to their Blue Button® electronic health information.
- Explain that TEAM participants may receive beneficiary-identifiable claims data.
- Advise patients that all standard Medicare beneficiary protections remain in place, including the ability to report concerns of substandard care to QIOs and 1-800-MEDICARE.
- Provide a list of the providers, suppliers, and ACOs with whom the TEAM participant has a sharing arrangement. We recognize an exhaustive list of providers, suppliers, and ACOs may lengthen the beneficiary notification unnecessarily, therefore this requirement may be fulfilled by the TEAM participant including in the beneficiary notification a web address where beneficiaries may access the list.
After carefully considering the appropriate timing and circumstances for the necessary beneficiary notification, we proposed at ( 89 FR 36472 ) that TEAM participants must require all ACOs, providers, and suppliers who execute a Sharing Arrangement with a TEAM participant to share beneficiary notification materials, to be developed or approved by CMS, that detail this proposed payment model with the beneficiary prior to discharge from the anchor hospitalization or prior to discharge from the anchor procedure for a Medicare FFS patient who would be included under the model. TEAM participants must require this notification as a condition of any Sharing Arrangement. Where a TEAM participant does not have Sharing Arrangements with providers or suppliers that furnish services to beneficiaries during an episode, or where the anchor hospitalization or anchor procedure for a Medicare FFS patient who would be included under the model was ordered by a physician who does not have a Sharing Arrangement, the beneficiary notification materials must be provided to the beneficiary by the TEAM participant. We indicated in the proposed rule that the purpose of this policy is to ensure that all TEAM beneficiaries receive the beneficiary notification materials, and that they receive such materials as early as possible but no later than discharge from the hospital or hospital outpatient department. We believe that this proposal targets beneficiaries for whom information is relevant and increases the likelihood that patients will become engaged and seek to understand the model and its potential impact on their care.
In addition, we proposed at § 512.582(b)(2) that TEAM participants must require every TEAM collaborator to provide written notice, to be developed by CMS, to applicable TEAM beneficiaries of the existence of its sharing arrangement with the TEAM participant and the basic quality and payment incentives under the model. We proposed that the notice must be provided no later than the time at which the beneficiary first receives an item or service from the TEAM collaborator during an episode. We recognize that due to the patient's condition, it may not be feasible to provide notification at such time, in which case the notification must be provided to the beneficiary or his or her representative as soon as is reasonably practicable. We note that beneficiaries are accustomed to receiving similar notices of rights and obligations from healthcare providers prior to the start of inpatient care. However, we also considered that this information might be best provided by hospitals at the point of admission for all beneficiaries, as hospitals provide other information concerning patient rights and responsibilities at that time. We invited comment on ways in which the timing and source of beneficiary notification could best serve the needs of beneficiaries without creating unnecessary administrative work for providers and suppliers. We believe that this notification is an important safeguard to help ensure that beneficiaries in the model receive all medically necessary services, but it is also an important clinical opportunity to better engage beneficiaries in defining their goals and preferences as they share in the planning of their own care.
The following is a summary of comments we received on the proposed beneficiary notification and beneficiary protections ( 89 FR 36471 ) in the proposed model:
Comment: We received a number of comments supporting our proposals to require notification of beneficiaries about their inclusion in a TEAM episode, and the sharing arrangements between TEAM participants and their collaborators, including our proposed timing for notification. Additionally, a commenter supported the idea that patients should be fully informed about TEAM and should be able to be active participants in their treatment decisions.
Response: We thank the commenters for their support and note that CMS will continue to strive for transparency in care delivery within TEAM.
Comment: Several commenters expressed concern with our proposed beneficiary notification policies. Some commenters stated CMS should take responsibility for the distribution of this letter. Other commenters felt the burden of this letter would be too high and should not be required. Commenters also made suggestions such as including the notification letter in other CMS materials such as the Medicare You handbook, Welcome to Medicare packet or the Medicare Beneficiary Manual. Commentors also expressed that timing of the beneficiary notification letter is important and should be distributed at the time services are provided and as early as possible.
Response: We acknowledge the need to streamline the beneficiary notification process to mitigate administrative burdens for participating hospitals. Additionally, we understand the importance of providing information to beneficiaries about their care in clear, plain language. We are committed to ensuring that notifications are concise and easily understandable, as well as detail the model's goals, structure, and potential impacts on care delivery and access. With regard to inclusion of the beneficiary notification information in other Medicare publications, we believe this would create confusion for beneficiaries, since not all beneficiaries who access or read those documents will be included in TEAM episodes. We also think it would not accomplish our goal of ensuring that TEAM participants directly provide notice to beneficiaries about their participation (and beneficiaries' inclusion) in the model. Finally, we also note that the proposed beneficiary notification letter would allow providers the ability to translate the notification letter into needed languages for their hospital population.
Comment: A commenter stated concern with the timing for beneficiary notifications, noting that current requirements that notifications be made prior to discharge from the anchor hospitalization, or prior to discharge from the anchor procedure, may inform ( print page 69839) beneficiaries too late for post-discharge planning. The commenter recommended notifications be made at the first encounter regardless of episode initiation.
Response: We appreciate the feedback regarding the timing of beneficiary notifications. Though the notification isn't required until discharge, we do expect discharge care to be coordinated prior to that time. CMS will review the suggestion to require notifications at the first encounter to better inform beneficiaries ahead of surgical decisions and we will take it into consideration in future rulemaking.
Comment: Several commenters provided general input on the proposed beneficiary protections under the model. A commenter recommended considering a longer lookback period than 90 days, such as 180 days or one year, in order to allow for an extended period to monitor changes in utilization and access to care. Additionally, a commenter suggested strengthening the model's emphasis on delivering high-quality care, and through monitoring, ensuring that cost-savings do not come at the expense of care delivery. Commenters urged CMS to design TEAM to reward primary care physicians for supporting optimal, long-term health outcomes. Furthermore, several commenters suggested that care under TEAM be person-centered, incorporating the beneficiary perspective, including recommendations such as patient-reported quality measures related to shared decision-making, developing a patient ombudsman role or contracting with patient navigators, and ensuring that community and beneficiary perspectives are included in the participant selection and evaluation processes.
Response: We thank the commenters for their suggestions and dedication to ensuring beneficiary protections for TEAM are in place. We will take this information under consideration when monitoring TEAM participants for potential compliance and access issues. We also refer readers to sections X.A.3.l and X.A.3.c of this final rule, where we finalize proposals to encourage TEAM participants to connect beneficiaries to primary care and include a patient-reported outcome measure for one of the episode categories in TEAM.
After consideration of the public comments we received, we are finalizing as proposed our proposals for beneficiary freedom of choice at § 512.582(a), TEAM participant beneficiary notification at § 512.582(b)(1) and TEAM collaborator notice at § 512.582(b)(2).
In the proposed rule we stated that since TEAM participants would receive a reconciliation payment when they are able to meet certain cost and quality performance thresholds, they could have an incentive to avoid complex, high-cost cases by referring them to nearby facilities or specialty referral centers. We intend to monitor the claims data from TEAM participants—for example, to compare a hospital's case mix relative to a pre-model historical baseline—to determine whether complex patients are potentially being systematically excluded. We indicated in the proposed rule that we will publish these data as part of the model evaluation to promote transparency and an understanding of the model's effects. We also proposed to continue to review and audit hospitals if we have reason to believe that they are compromising beneficiary access to care. For example, we may audit a hospital or conduct additional claims analyses where initial claims analysis indicates an unusual pattern of referral to regional hospitals located outside of the model catchment area or a clinically unexplained increase or decrease in surgical rates for procedures included in TEAM. We sought comment at ( 89 FR 36472 ) on our proposals to monitor TEAM participants.
The following is a summary of comments we received on the proposed monitoring for access to care:
Comment: A commenter supported the proposal to monitor inappropriate care patterns in TEAM.
Response: We thank the commenter for their support of our proposed monitoring and compliance of the model.
Comment: Several commenters referenced situations where monitoring will be especially important for TEAM. Some commenters are concerned that patients treated by TEAM participants should have access to the full range of treatment options necessary for their medical conditions, which are critical to the health and well-being of Medicare beneficiaries. Specifically, these commenters were concerned that TEAM participants might limit patient choice during the 30-day post-discharge period, potentially diverting patients away from essential post-acute services, and disregarding existing patient-physician relationships or avoiding specialist referrals that may be necessary. Additionally, commenters were concerned that participants might favor lower-cost, lower-utility devices over higher-cost technologies that are more appropriate for certain conditions, thereby compromising the quality of care for patients with more intensive or costly needs. A commenter also emphasized timeliness with regard to monitoring, to ensure CMS is able to quickly address any adverse effects discovered through monitoring activities.
Response: We appreciate the commenters' concerns and are committed to monitoring for, and promptly addressing, any potential negative impacts identified through ongoing monitoring. As proposed, we will compare hospitals' case mixes to historical baselines to detect potential exclusion of complex patients and publish these findings for transparency. We will also review and audit hospitals to uncover any signs of compromised beneficiary access, such as unusual referral patterns or unexplained changes in surgical rates.
After consideration of the public comments we received, we are finalizing as proposed our proposals for monitoring for access to care at § 512.584.
In the proposed rule we noted that in any payment system that promotes efficiencies of care delivery, there may be opportunities to direct patients away from more expensive services at the expense of outcomes and quality. We believe that professionalism, the quality measures in the model, and clinical standards can be effective in preventing beneficiaries from being denied medically necessary care in the inpatient setting, outpatient setting, and in post-acute care settings during the 30 days post-discharge. Accordingly, we believe that the potential for the denial of medically necessary care within TEAM will not be greater than that which currently exists under IPPS. However, we also believe that we have the authority and responsibility to audit the medical records and claims of participating hospitals and their TEAM collaborators in order to ensure that beneficiaries receive medically necessary services. Similarly, at § 512.590, we proposed to monitor arrangements between TEAM participants and their TEAM collaborators to ensure that such arrangements do not result in the denial of medically necessary care, or other program or patient abuse. We invited public comment on these proposals and on whether there are elements of TEAM that would require additional beneficiary protection for the appropriate delivery of inpatient care, and if so, what types of monitoring or safeguards would be most appropriate. ( print page 69840)
We stated in the proposed rule that we believe that these safeguards are all enhanced by beneficiary knowledge and engagement. Therefore, we proposed at § 512.582(a)(3) to require that TEAM participants must, as part of discharge planning, account for potential financial bias by providing TEAM beneficiaries with a complete list of all available post-acute care options in the Medicare program, including HHAs, SNFs, IRFs, or LTCHs, in the service area consistent with medical need, including beneficiary cost-sharing and quality information (where available and when applicable). This list should also indicate whether the TEAM participant has a sharing arrangement with the post-acute care provider. We expect that the treating surgeons or other treating practitioners, as applicable, will continue to identify and discuss all medically appropriate options with the beneficiary, and that hospitals will discuss the various facilities and providers who are available to meet the clinically identified needs. These proposed requirements for TEAM participants would supplement the existing discharge planning requirements under the hospital Conditions of Participation. We also specifically note that neither the Conditions of Participation nor this proposed transparency requirement preclude hospitals from recommending preferred providers within the constraints created by current law, as coordination of care and optimization of care are important factors for successful participation in this model. We invited comment on this proposal, including additional opportunities to ensure high quality care.
We received no comments on these proposals and are finalizing as proposed our proposals on monitoring for quality of care at § 512.582 and § 512.590.
We stated in the proposed rule that we believe TEAM would incentivize TEAM participants to create efficiencies in the delivery of care within a 30-day episode following an acute clinical event. Theoretically, TEAM also could create incentives for TEAM participants or their TEAM collaborators to delay services until after such 30-day window has closed. Consistent with the CJR model, we believe that existing Medicare safeguards are sufficient to protect beneficiaries in TEAM.
We indicated in the proposed rule that our experience with other episode-based payment models, such as the BPCI Advanced model, has shown that providers focus first on appropriate care and then on efficiencies only as obtainable in the setting of appropriate care. We believe that a 30-day post-discharge episode is sufficient to minimize the risk that TEAM participants and their TEAM collaborators would compromise services furnished in relation to a beneficiary's care. While we recognize that ongoing care for underlying conditions may be required after the 30-day episode, we believe that TEAM participants and other providers and suppliers would be unlikely to postpone key services beyond a 30-day period because the consequences of delaying care beyond such episode duration would be contrary to usual standards of care.
However, we also note in the proposed rule that additional monitoring would occur as a function of the proposed TEAM. As with the CJR model, we proposed as part of the reconciliation process (see section X.A.3.d.(5)(i) of the preamble of this final rule) that TEAM participants would be financially accountable for certain post-episode payments occurring in the 30 days after conclusion of the episode. We believe that including such a payment adjustment would create an additional deterrent to delaying care beyond the episode duration. In addition, we believe the data collection and calculations used to determine such adjustment would provide a mechanism to check whether providers are inappropriately delaying care. Finally, we noted in the proposed rule that the proposed quality measures create additional safeguards as such measures are used to monitor and influence clinical care at the institutional level.
We invited public comment on our proposed requirements for notification of beneficiaries and our proposed methods for monitoring participants' actions and ensuring compliance as well as on other methods to ensure that beneficiaries receive high quality, clinically appropriate care.
We received no comments on these proposals and are finalizing as proposed the proposals on monitoring for delayed care at § 512.582 and § 512.590.
In the proposed rule we stated that by virtue of their participation in an CMS Innovation Center model, TEAM participants and TEAM collaborators may receive model-specific payments, access to payment rule waivers, or some other model-specific flexibility ( 89 FR 36473 ). Therefore, we believe that CMS's ability to audit, inspect, investigate, and evaluate records and other materials related to participation in CMS Innovation Center models is necessary and appropriate. There is a need for CMS to be able to audit, inspect, investigate, and evaluate records and materials related to participation in CMS Innovation Center models to allow us to ensure that TEAM participants are not denying or limiting the coverage or provision of benefits for beneficiaries as part of their participation in the CMS Innovation Center model. We proposed at § 512.505 to define “model-specific payment” to mean a payment made by CMS only to TEAM participants, under the terms of the CMS Innovation Center model that is not applicable to any other providers or suppliers; the term “model-specific payment” would include, unless otherwise specified, the reconciliation payment, described in section X.A.3.d.(5)(j) of the preamble of this final rule.
We noted in the proposed rule that there are audit and record retention requirements under the Medicare Shared Savings Program ( 42 CFR 425.314 ) and in current models being tested under section 1115A (such as under 42 CFR 510.110 for the CMS Innovation Center's Comprehensive Care for Joint Replacement Model) ( 89 FR 36473 ). Building off those existing requirements, we proposed in § 512.135(a), that the Federal Government, including, but not limited to, CMS, HHS, and the Comptroller General, or their designees, would have a right to audit, inspect, investigate, and evaluate any documents and other evidence regarding implementation of a CMS Innovation Center model. Additionally, in order to align with the policy of current models being tested by the CMS Innovation Center, we proposed that the TEAM participant and its TEAM collaborators must maintain and give the Federal Government, including, but not limited to, CMS, HHS, and the Comptroller General, or their designees, access to all documents (including books, contracts, and records) and other evidence sufficient to enable the audit, evaluation, inspection, or investigation of the CMS Innovation Center model, including, without limitation, documents and other evidence regarding all of the following:
- Compliance by the TEAM participant and its TEAM collaborators with the terms of the CMS Innovation Center model, including proposed new subpart A of proposed part 512.
- The accuracy of model-specific payments made under the CMS Innovation Center model.
- The TEAM participant's payment of amounts owed to CMS, or payment ( print page 69841) adjustments, under the CMS Innovation Center model.
- Quality measure information and the quality of services performed under the terms of the CMS Innovation Center model, including proposed new subpart A of proposed part 512.
- Utilization of items and services furnished under the CMS Innovation Center model.
- The ability of the TEAM participant to bear the risk of potential losses and to repay any losses through claims adjustments to CMS, as applicable.
- Patient safety under TEAM.
- Any other program integrity issues.
We proposed that TEAM participants must maintain the documents and other evidence for a period of 6 years from the last payment determination for the TEAM participant under the CMS Innovation Center model or from the date of completion of any audit, evaluation, inspection, or investigation, whichever is later, unless—
- CMS determines there is a special need to retain a particular record or group of records for a longer period and notifies the TEAM participant at least 30 days before the normal disposition date; or
- There has been a termination, dispute, or allegation of fraud or similar fault against the TEAM participant in which case the records must be maintained for an additional 6 years from the date of any resulting final resolution of the termination, dispute, or allegation of fraud or similar fault.
We stated in the proposed rule that if CMS notifies the TEAM participant of a special need to retain a record or group of records at least 30 days before the normal disposition date, we proposed that the records must be maintained for such period of time determined by CMS ( 89 FR 36473 ). We also proposed that, if CMS notifies the TEAM participant of a special need to retain records or there has been a termination, dispute, or allegation of fraud or similar fault against the TEAM participant or its TEAM collaborators, the TEAM participant must notify its TEAM collaborators of the need to retain records for the additional period specified by CMS. This provision will ensure that that the government has access to the records. To avoid any confusion or disputes regarding the timelines outlined in this section of this final rule, we proposed to define the term “days” to mean calendar days.
We stated in the proposed rule that historically, the CMS Innovation Center has required participants in section 1115A models to retain records for at least 10 years, which is consistent with the outer limit of the statute of limitations for the Federal False Claims Act and is consistent with the Shared Savings Program's policy outlined at 42 CFR 425.314(b)(2) ( 89 FR 36474 ). For this reason, we solicited public comments on whether we should require hospital participants and TEAM collaborators to maintain records for less than 10 years.
We invited public comment on these proposed provisions described at § 512.586 regarding audits and record retention.
The following is a summary of comments we received on these proposed provisions regarding audits and record retention.
Comment: A commenter expressed support for the proposed requirements regarding Access and Record Retention for TEAM Participants and TEAM collaborators.
Response: CMS thanks the commentor for their support of the proposed requirement.
After consideration of the public comments we received, we are finalizing these proposed provisions described at § 512.586 regarding audits and record retention. Additionally, we recognize the overlap between model-specific payment and TEAM payment definitions, and therefore to minimize confusion, we are finalizing only the definition of TEAM payment at § 512.505 and not model-specific payment.
As discussed in the proposed rule at 89 FR 36384 and 89 FR 36419 , we aim to incentivize TEAM participants to engage in care redesign efforts to improve quality of care and reduce Medicare FFS spending for beneficiaries included in the model during the anchor hospitalization or anchor procedure and the 30 days post-discharge from the hospital or hospital outpatient department. These care redesign efforts would require TEAM participants to work with and coordinate care with other health care providers and suppliers to improve the quality and efficiency of care for Medicare beneficiaries.
We have experience with a range of efforts designed to improve care coordination for Medicare beneficiaries, including the BPCI Advanced and CJR models, both of which make certain Medicare data available to participants to better enable them to achieve their goals. For example, both the BPCI Advanced and CJR participants may request to receive beneficiary-identifiable claims data and financial performance data from the baseline period and throughout their tenure in the model to help them better understand the FFS beneficiaries that are receiving services from their providers and help them improve quality of care and conduct care coordination and other care redesign activities to improve patient outcomes or reduce health care for beneficiaries that could have initiated an episode in the model.
Based on our experience with these efforts, as discussed later in this section, we proposed to make certain beneficiary-identifiable claims data and regional aggregate data available to participants in TEAM regarding Medicare FFS beneficiaries who may initiate an episode and be attributed to them in the model. However, we also stated that we expect that TEAM participants are able to, or will work toward, independently identifying and producing their own data, through electronic health records, health information exchanges, or other means that they believe are necessary to best evaluate the health needs of their patients, improve health outcomes, and produce efficiencies in the provision and use of services.
We believe that TEAM participants may need access to certain Medicare beneficiary-identifiable data for the purposes of evaluating their performance, conducting quality assessment and improvement activities, conducting population-based activities relating to improving health or reducing health care costs, or conducting other health care operations listed in the first or second paragraph of the definition of “health care operations” under the HIPAA Privacy Rule, 45 CFR 164.501 . We recognize that there are issues and sensitivities surrounding the disclosure of beneficiary-identifiable health information, and that several laws place constraints on sharing individually identifiable health information. For example, section 1106 of the Act generally bars the disclosure of information collected under the Act without consent unless a law (statute or regulation) permits the disclosure. Here, the HIPAA Privacy Rule would allow for the proposed disclosure of individually identifiable health information by CMS. In the proposed rule, we proposed to make TEAM participants accountable for quality and cost outcomes for TEAM beneficiaries during an anchor hospitalization or ( print page 69842) anchor procedure and during the 30-day post-discharge period. We stated in the proposed rule that we believe that it is necessary for the purposes of this model to offer TEAM participants the ability to request summary or raw beneficiary-identifiable claims data for a 3-year baseline period as well as on a monthly basis during the performance year to help TEAM participants engage in care coordination and quality improvement activities for TEAM beneficiaries in an episode. For the 3-year baseline period, TEAM participants would only receive beneficiary-identifiable claims data for beneficiaries that initiated an episode in their hospital or hospital outpatient department in the 3-year baseline period, and the beneficiary-identifiable claims data shared with the TEAM participant would be limited to the items and services included in the episode. In other words, the TEAM participant would not receive beneficiary-identifiable claims data for beneficiaries that were admitted to their hospital or hospital outpatient department and did not initiate an episode in the baseline period. Nor would the TEAM participant receive beneficiary-identifiable claims data, for beneficiaries who did initiate an episode in their hospital or hospital outpatient department during the baseline period, for items and services that are not included in an episode, such as a primary care visit 5 days before the episode or a hospital readmission 1 day after the episode ends. We proposed to apply a similar approach for the beneficiary-identifiable claims data sharing during the performance year. We stated that we believe that these data would constitute the minimum information necessary to enable the TEAM participant to understand spending patterns during the episode, appropriately coordinate care, and target care strategies toward individual beneficiaries furnished care by the TEAM participant and other providers and suppliers.
Under the HIPAA Privacy Rule, covered entities (defined as health care plans, providers that conduct covered transactions, including hospitals, and health care clearinghouses) are barred from using or disclosing individually identifiable health information that is “protected health information” or PHI in a manner that is not explicitly permitted or required under the HIPAA Privacy Rule, without the individual's authorization. The Medicare FFS program, a “health plan” function of the Department, is subject to the HIPAA Privacy Rule limitations on the disclosure of PHI. Hospitals, which would be TEAM participants, and other Medicare providers and suppliers are also covered entities, provided they are health care providers as defined by 45 CFR 160.103 and they conduct (or someone on their behalf conducts) one or more HIPAA standard transactions electronically, such as for claims transactions. We noted that since TEAM participants are hospitals who are covered entities and are the only entities able to request the beneficiary-identifiable data and with whom CMS would share the beneficiary-identifiable data, we believe that the proposed disclosure of the beneficiary claims data for an anchor hospitalization or an anchor procedure plus 30-day post-discharge for episodes included under TEAM would be permitted by the HIPAA Privacy Rule under the provisions that permit disclosures of PHI for “health care operations” purposes. Under those provisions, a covered entity is permitted to disclose PHI to another covered entity for the recipient's health care operations purposes if both covered entities have or had a relationship with the subject of the PHI to be disclosed, the PHI pertains to that relationship, and the recipient will use the PHI for a “health care operations” function that falls within the first two paragraphs of the definition of “health care operations” in the HIPAA Privacy Rule ( 45 CFR 164.506(c)(4) ).
The first paragraph of the definition of health care operations includes “conducting quality assessment and improvement activities, including outcomes evaluation and development of clinical guidelines,” and “population-based activities relating to improving health or reducing health costs, protocol development, case management and care coordination” ( 45 CFR 164.501 ).
Under our proposal, TEAM participants would be using the data on their patients to evaluate the performance of the TEAM participant and other providers and suppliers that furnished services to the patient, conduct quality assessment and improvement activities, and conduct population-based activities relating to improved health for their patients. When done by or on behalf of a covered entity, these are covered functions and activities that would qualify as “health care operations” under the first and second paragraphs of the definition of health care operations at 45 CFR 164.501 . Hence, as previously discussed, we believe that this provision is extensive enough to cover the uses we would expect a TEAM participant to make of the beneficiary-identifiable data and would be permissible under the HIPAA Privacy Rule. Moreover, we noted that our proposed disclosures would be made only to HIPAA covered entities, specifically hospitals that are TEAM participants that have (or had) a relationship with the subject of the information, the information we would disclose would pertain to such relationship, and those disclosures would be for purposes listed in the first two paragraphs of the definition of “health care operations.”
When using or disclosing PHI, or when requesting this information from another covered entity, covered entities must make “reasonable efforts to limit” the information that is used, disclosed, or requested to a “minimum necessary” to accomplish the intended purpose of the use, disclosure, or request ( 45 CFR 164.502(b) ). We stated in the proposed rule that we believe that the provision of the proposed data elements, as described in section X.A.3.k.(2)(c) of the preamble of this final rule, would constitute the minimum data necessary to accomplish the TEAM's model goals of the TEAM participant.
The Privacy Act of 1974 also places limits on agency data disclosures. The Privacy Act applies when the federal government maintains a system of records by which information about individuals is retrieved by use of the individual's personal identifiers (names, Social Security numbers, or any other codes or identifiers that are assigned to the individual). The Privacy Act prohibits disclosure of information from a system of records to any third party without the prior written consent of the individual to whom the records apply ( 5 U.S.C. 552a(b) ).
“Routine uses” are an exception to this general principle. A routine use is a disclosure outside of the agency that is compatible with the purpose for which the data was collected. Routine uses are established by means of a publication in the Federal Register about the applicable system of records describing to whom the disclosure will be made and the purpose for the disclosure. For the proposed TEAM, the system of records would be covered in Master Demonstration, Evaluation, and Research Studies (DERS) for the Office of Research, Development and Information (ORDI) system of record ( 72 FR 19705 ). We stated that we believe that the proposed data disclosures are consistent with the purpose for which the data discussed in the proposed rule was collected and may be disclosed in accordance with the routine uses applicable to those records.
We noted that, as is the case with the CJR model, in the proposed rule, we ( print page 69843) proposed to disclose beneficiary-identifiable data to only the hospitals that are bearing risk for episodes and not with their collaborators. As stated in the final CJR rule ( 80 FR 73515 ), we believe that the hospitals that are specifically held financially responsible for an episode should make the determination as to which data are needed to manage care and care processes with their collaborators as well as which data they might want to re-disclose, if any, to their collaborators provided they are in compliance with the HIPAA Privacy Rule.
We stated that we believe our data sharing proposals are permitted by and are consistent with the authorities and protections available under the aforementioned statutes and regulations. We sought comments on our proposals regarding the authority to share beneficiary-identifiable data with TEAM participants.
We received no public comments on our proposals regarding the legal authority for CMS to share beneficiary-identifiable data with TEAM participants. Thus, we are finalizing at § 512.562(b)(1) the proposal to share beneficiary-identifiable data with TEAM participants as permitted under the referenced statutes and regulations.
Based on our experience with BPCI Advanced and CJR participants, we recognize that TEAM participants could vary with respect to the kinds of beneficiary-identifiable claims information that would best meet their needs. For example, while many TEAM participants might have the ability to analyze raw claims data, other TEAM participants could find it more useful to have a summary of these data. Given this, we proposed to make beneficiary-identifiable claims data for episodes in TEAM available through two formats, summary and raw, both for the baseline period and on an ongoing monthly basis during their participation in the model as we do for BPCI Advanced and CJR. As we explained in the proposed rule, summary beneficiary-identifiable claims data summarizes the claims data by combining and categorizing claims data to provide a broad view of the TEAM participant's health care expenditures and utilization. For example, a TEAM participant may use summary beneficiary-identifiable data to identify total episode spending for a given episode category across all of a TEAM participant's episodes in a given performance year. Raw beneficiary-identifiable claims data is unrefined and has not been grouped or combined and includes the specific claims fields, as described in the minimum necessary data discussion in section X.A.3.k.(2).(c). of the preamble of this final rule, at the episode level. For example, a TEAM participant may use raw beneficiary-identifiable data to look at a particular episode to identify the diagnosis code(s) that were associated with a hospital readmission for a TEAM beneficiary.
First, for TEAM participants who wish to receive summary Medicare Parts A and B claims data, we proposed to offer TEAM participants, that enter into a TEAM data sharing agreement with CMS, as specified in section X.A.3.k.(6). of the preamble of this final rule, the option to submit a formal data request for summary beneficiary-identifiable claims data that have been aggregated to provide summary-level spending and utilization data on TEAM beneficiaries who would be in an episode during the baseline period and performance years, in accordance with applicable privacy and security laws and established privacy and security protections. We explained that such summary beneficiary-identifiable claims data would provide tools to monitor, understand, and manage utilization and expenditure patterns as well as to develop, target, and implement quality improvement programs and initiatives. For example, if the data provided by CMS to a particular TEAM participant reflects that, relative to their peers, a certain provider is associated with significantly higher rates of inpatient readmissions than the rates experienced by other beneficiaries with similar care needs, that may be evidence that the TEAM participant could consider, among other things, the appropriateness of that provider, whether other alternatives might be more appropriate, and whether there exist certain care interventions that could be incorporated post-discharge to lower readmission rates.
Second, for TEAM participants who wish to receive raw Medicare Parts A and B claims data, we proposed to offer TEAM participants, that enter into a TEAM data sharing agreement with CMS, the opportunity to submit a formal data request for raw beneficiary-identifiable claims data for TEAM beneficiaries who would be in an episode during the baseline period and performance years, in accordance with applicable privacy and security laws and established privacy and security protections. We explained that these raw beneficiary-identifiable claims data would be much more detailed compared to the summary beneficiary-identifiable claims data and include all beneficiary-identifiable claims for all episodes in TEAM. In addition, they would include episode summaries, indicators for excluded episodes, diagnosis and procedure codes, and enrollment and dual eligibility information for beneficiaries that initiate episodes in TEAM. Through analysis, these raw beneficiary-identifiable claims data would provide TEAM participants with information to improve their ability to coordinate and target care strategies as well as to monitor, understand, and manage utilization and expenditure patterns. Such data would also aid them in developing, targeting, and implementing quality improvement programs and initiatives.
We explained that the summary and raw beneficiary-identifiable data would allow TEAM participants to assess summary and raw data on their relevant TEAM beneficiary population, giving them the flexibility to utilize the data based on their analytic capacity. Therefore, for both the baseline period and at a minimum on a monthly basis during an TEAM participant's performance year, we proposed to provide TEAM participants with an opportunity to request summary beneficiary-identifiable claims data and raw beneficiary-identifiable claims data that would meet HIPAA minimum necessary requirements in 45 CFR 164.502(b) and 164.514(d) and include Medicare Parts A and B beneficiary-identifiable claims data for TEAM beneficiaries in an episode during the 3-year baseline period and performance year. This means the summary and raw beneficiary-identifiable claims data would encompass the total expenditures and claims for the proposed episodes, including the anchor hospitalization or anchor procedure, and all non-excluded items and services in an episode covered under Medicare Parts A and B within the 30 days after discharge, including hospital care, post- acute care, and physician services for the TEAM participant's beneficiaries.
We proposed that if a TEAM participant wishes to receive beneficiary-identifiable claims data, they must submit a formal request for data on at least an annual basis in a manner form and by a date specified by CMS, indicating if they want summary beneficiary-identifiable data, raw beneficiary-identifiable data, or both, and sign a TEAM data sharing agreement. To comply with applicable laws and safeguards, we proposed the TEAM participant must attest that—
- The TEAM participant is requesting claims data of TEAM beneficiaries who would be in an episode during the ( print page 69844) baseline period or performance year as a HIPAA-covered entity;
- The TEAM participant's request reflects the minimum data necessary for the TEAM participant to conduct health care operations work that falls within the first or second paragraph of the definition of health care operations at 45 CFR 164.501 ;
- The TEAM participant's use of claims data will be limited to developing processes and engaging in appropriate activities related to coordinating care and improving the quality and efficiency of care and conducting population-based activities relating to improving health or reducing health care costs that are applied uniformly to all TEAM beneficiaries, in an episode during the baseline period or performance year, and that these data will not be used to reduce, limit or restrict care for specific Medicare beneficiaries.
We proposed that the summary and raw beneficiary-identifiable data would be packaged and sent to a data portal (to which the TEAM participants must request and be granted access) in a “flat” or binary format for the TEAM participant to retrieve. We also noted that, for both the summary and raw beneficiary-identifiable claims data, we would exclude information that is subject to the regulations governing the confidentiality of substance use disorder patient records ( 42 CFR part 2 ) from the data shared with a TEAM participant. We stated that we believe our proposal to make data available to TEAM participants, through the most appropriate means, may be useful to TEAM participants to determine appropriate ways to increase the coordination of care, improve quality, enhance efficiencies in the delivery system, and otherwise achieve the goals of the proposed model. TEAM beneficiaries would be informed of TEAM and the potential sharing of Medicare beneficiary-identifiable claims data through the beneficiary notification, as discussed in section X.A.3.i.(2) of the preamble of this final rule. Further, CMS would make beneficiary-identifiable claims data available to a TEAM participant for beneficiaries who may be included in episodes, in accordance with applicable privacy and security laws and only in response to the TEAM participant's request for such data, through the use of an executed TEAM data sharing agreement with CMS.
We requested comments on this proposal to share beneficiary-identifiable claims data with TEAM participants at § 512.562(b).
The following is a summary of the public comments we received on the proposal to share beneficiary-identifiable data with TEAM participants and our responses to those comments:
Comment: A couple of commenters expressed support for the proposal to share certain beneficiary-level data with TEAM participants. The commenters indicated that these data would enable participants to identify their patient populations, plan and improve care, and gauge the quality of post-acute care providers.
Response: We thank the commenters for their support for the proposal to share certain beneficiary-level data under this model, and concur with the stated benefits for TEAM participants in receiving such data.
Comment: Several commenters emphasized the burden for TEAM participants in processing and analyzing data provided by CMS. These commenters noted that hospitals may need to dedicate staff or hire outside consultants to translate performance data into usable insights, and one of the commenters expressed specific concerns about the burden of this analytical work for safety net and rural hospitals. A commenter requested that CMS work to provide participant hospitals with the type of analyzed data that hospitals would otherwise pay outside analysts to generate, and another commenter suggested that CMS develop one-page data reports tailored to various types of hospital staff.
Response: We thank the commenters for expressing their concerns about the burden placed on hospitals to analyze performance data and generate usable insights. We also appreciate the commenters' suggestions for the provision of data reports that are usable without the aid of external analysts and that are tailored to different hospital staff. We note that the proposed availability of both summary and raw beneficiary-level and aggregate data for the baseline and performance periods is intended to provide data that is useful for TEAM participants with a range of analytic capacities. We also note that TEAM participants may weigh their own analytic capacities when determining whether to request beneficiary-identifiable data from CMS. We also note that, as stated in the preamble of the proposed rule at 89 FR 36474 and in alignment with the goals of the model, we expect that TEAM participants are able to, or will work toward being able to, independently identify and produce their own data through electronic health records, health information exchanges, or other means as they deem necessary to evaluate patients, improve outcomes, and produce efficiencies. Still, we recognize that challenges may persist for TEAM participants in analyzing and improving patterns of care. Thus, we intend to provide additional supports to participants as we deem appropriate and feasible. For example, as with other models like BPCI Advanced and CJR, we plan to implement a learning system through which CMS can provide support to participants and participants can voluntarily share insights with each other. Additionally, similar to efforts undertaken to improve data transparency and utility during the course of BPCI Advanced, we will welcome input from TEAM participants and other interested parties on additional data sharing that could benefit participants and may consider such input in future rulemaking.
After consideration of the comments received, we are finalizing at § 512.562(b) the proposal to share certain beneficiary-identifiable claims data with TEAM participants.
We proposed TEAM participants must limit their beneficiary-identifiable data requests, for TEAM beneficiaries who are in an episode during the baseline period or performance year, to the minimum necessary to accomplish a permitted use of the data. We proposed the minimum necessary Parts A and B data elements may include but are not limited to the following data elements:
- Medicare beneficiary identifier (ID).
- Procedure code.
- Diagnosis code.
- The from and through dates of service.
- The provider or supplier ID.
- The claim payment type.
- Date of birth and death, if applicable.
- Tax identification number.
- National provider identifier.
We sought comment on the minimum necessary beneficiary-identifiable information for TEAM participants to request for purposes of conducting permissible health care operations purposes under this model at § 512.562(c).
The following is a summary of the public comments we received on the minimum necessary beneficiary-identifiable data for TEAM participants to request for health care operations purposes and our responses to those comments:
Comment: A couple of commenters further recommended that the ( print page 69845) beneficiary-level data include all claim types.
Response: We thank the commenters for their requests. We note that the proposed beneficiary-identifiable data sharing would encompass the total expenditures and claims for the proposed episodes in order to best support participants in health care operations, including performance monitoring, care coordination, quality improvement, and population-based activities. This would include all non-excluded items and services in an episode covered under Medicare Parts A and B, including hospital care, post-acute care settings, and physician services—in other words, all claim types that are included in TEAM episodes. We note that including any claim types in the beneficiary-identifiable data sharing that are not among the claim types included in TEAM episodes would be irrelevant to TEAM and thus fall beyond the minimum necessary data sharing.
We are finalizing at § 512.562(c) the proposal to make beneficiary-identifiable data available to TEAM participants, to include raw and summary Medicare Parts A and B beneficiary-identifiable claims data for TEAM beneficiaries in an episode during the 3-year baseline period and performance year.
As discussed in section X.A.3.d.(3) of the preamble of this final rule, we proposed to incorporate regional pricing data when establishing target prices for TEAM participants, similar to the CJR model's target prices that are constructed at the regional level. As indicated in the CJR final rule ( 80 FR 73510 ), we finalized our proposal to share regional pricing data with CJR participants because it was a factor affecting target prices. Given some of the similar features between the CJR model and TEAM proposed in the proposed rule, particularly our proposal to incorporate regional pricing data when establishing target prices under the model, we proposed to provide regional aggregate expenditure data available for all Parts A and B claims associated with episodes in TEAM for the U.S. Census Division in which the TEAM participant is located, as we similarly provide to hospitals participating in the CJR model. Specifically, we proposed to provide TEAM participants with regional aggregate data on the total expenditures during an anchor hospitalization or anchor procedure and the 30-day post-discharge period for all Medicare FFS beneficiaries who would have initiated an episode under our proposed episode definitions at 89 FR 36416 during the baseline period and performance years. This data would be provided at the regional level; that is, we proposed to share regional aggregate data with a TEAM participant for episodes initiated in the U.S. Census Division where the TEAM participant is located. These regional aggregate data would be in a format similar to the proposed summary beneficiary-identifiable claims data and would provide summary information on the average episode spending for episodes in TEAM in the U.S. Census Division in which the TEAM participant is located. However, the regional aggregate data would not be beneficiary-identifiable and would be de-identified in accordance with HIPAA Privacy Rule, 45 CFR 164.514(b) . Further, the regional aggregate data would also comply with CMS data sharing requirements, including the CMS cell suppression policy which stipulates that no cell (for example, admissions, discharges, patients, services, etc.) containing a value of 1 to 10 can be reported directly. Given the regional aggregate data is de-identified, we proposed TEAM participants would not have to submit a request to receive this data and the data would not be subject to the terms and conditions of the TEAM data sharing agreement.
We sought comments on our proposal at § 512.562(d) to provide these data to TEAM participants.
We received no public comments on the proposal to provide regional aggregate data to TEAM participants and thus are finalizing at § 512.562(d) the provision of regional aggregate data to TEAM participants as proposed, noting that only minor grammatical changes were made to the regulatory text.
We recognize that providing the ability for TEAM participants to request the summary and raw beneficiary-identifiable claims baseline data and receive regional aggregate baseline data would be important for TEAM participants to be able to detect unnecessary episode spending, coordinate care, and identify areas for practice transformation, and that early provision of this data, specifically before the model start date, as defined in § 512.505, could facilitate their efforts to do so. Also, as discussed in section X.A.3.d.(3)(a) of the preamble of this final rule, target prices would be calculated using a TEAM participant's historical episode spending during their baseline period. Further, we believe that TEAM participants would view the episode payment model effort as one involving continuous improvement. As a result, changes initially contemplated by a TEAM participant could be subsequently revised based on updated information and experiences.
Therefore, as with the BPCI Advanced model, we proposed to make 3-years of baseline period data available to TEAM participants, who enter into a TEAM data sharing agreement with CMS, for beneficiaries who would have been included in an episode had the model been implemented during the baseline period, and intend to make these data available upon request prior to the start of each performance year and in accordance with applicable privacy and security laws and established privacy and security protections. We would provide the 3 years of baseline period data for the summary and raw beneficiary-identifiable data and for the regional aggregate data. We believe that 3 years of baseline period data is sufficient to support a TEAM participant's ability to detect unnecessary episode spending, coordinate care, and identify areas for practice transformation. We believe that if a TEAM participant has access to baseline period data for the 3-year period for each performance year used to set target prices, then it would be better able to assess its practice patterns, identify cost drivers, and ultimately redesign its care practices to improve efficiency and quality. We considered proposing to make available 4 years of baseline period data, or offering 1 year of baseline period data, but we believe offering 4 years of baseline period data would not be necessary since target prices in TEAM are constructed from a 3-year baseline period and 1 year of data may not sufficiently help TEAM participants identify areas to improve beneficiary health and care coordination or reducing health costs.
Therefore, we proposed that the 3-year period utilized for the baseline period match the baseline data used to create TEAM participants target prices every performance year, and roll forward one year every performance year, as discussed in section X.A.3.d.(3)(a) of the preamble of this final rule. Specifically, we proposed that the baseline period data for the summary and raw beneficiary-identifiable data reports and regional aggregate data report would be shared annually at least 1 month prior to the start of a performance year and available for episodes for each of the following performance years:
Performance Year 1: Episodes that began January 1, 2022, through December 31, 2024 ( print page 69846)
- Performance Year 2: Episodes that began January 1, 2023, through December 31, 2025
- Performance Year 3: Episodes that began January 1, 2024, through December 31, 2026
- Performance Year 4: Episodes that began January 1, 2025, through December 31, 2027
- Performance Year 5: Episodes that began January 1, 2026, through December 31, 2028
We requested comments on these proposals at proposed § 512.562(b)(6)(i) and § 512.562(d)(1)(i) to share beneficiary-identifiable data and regional aggregate data for a 3-year baseline period at least 1 month prior to the start of a performance year. We note that the proposed periods of data sharing stated in the proposed rule at 89 FR 36477 for Performance Years 4 and 5 were erroneously listed as being only two years long, ending at the end of 2026 and 2027, respectively. The data sharing periods have been corrected in this final rule to reflect the 3-year baseline period consistently across performance years—namely, episodes beginning January 1, 2025, through December 31, 2027, for Performance Year 4, and episodes beginning January 1, 2026, through December 31, 2028, for Performance Year 5.
The following is a summary of the public comments we received on the proposal to share beneficiary-identifiable data and regional aggregate data for a 3-year baseline period with TEAM participants at least 1 month prior to the start of a performance year and our responses to these comments:
Comment: A couple commenters requested earlier data sharing from CMS prior to the start of the model performance period. A couple commenters requested that CMS provide TEAM participants with a full set of claims and quality data one year prior to model launch, indicating that this is needed for participants to conduct the care redesign necessary for successful performance in the model. Another commenter requested that CMS provide baseline claims data and target prices at least one year prior to the performance period.
Response: We thank the commenters for these requests. As stated in the proposed rule, we recognize that sharing baseline claims data with TEAM participants prior to the model start date could facilitate their efforts to detect unnecessary spending, coordinate care, and identify areas for practice transformation. The proposed commitment by CMS to provide baseline data at least one month prior to the start of the corresponding performance period is in recognition of the utility of these data for participants. However, we are not convinced that the provision of claims data to participants one year prior to model launch is necessary for participants' success in the model, nor that participants' success in the model will be determined by care redesign activities undertaken entirely prior to model launch. Instead, as stated in the proposed rule, we view TEAM participation as a process of continuous improvement and expect that participants will use baseline and performance period data to inform care redesign activities throughout their tenure in the model. In accordance with this expectation, we proposed to provide multiple sources of usable data to participants at regular intervals, including monthly beneficiary-identifiable claims data and yearly quality measure score data. For both claims and quality data, the proposed timing and cadence of data sharing was carefully considered and determined based on the utility of the data, availability of results, and administrative burden under this model specifically.
We also note that, as stated in the proposed rule ( 89 FR 36425 ), the proposed timing for CMS to share quality measure performance data with TEAM participants aligns with the established CMS Care Compare schedule found here: https://data.cms.gov/provider-data/topics/hospitals/measures-and-currentdata-collection-periods . Regarding target price data, we note that the timing of preliminary target price calculations is dependent on the availability of episode spending data for the baseline period. For example, as stated in the proposed rule ( 89 FR 36427 ), CMS would use baseline episode spending for episodes that started between January 1, 2022, and December 31, 2024, to determine baseline episode spending for Performance Year 1 (PY 1), which is proposed to begin on January 1, 2026. Since episodes starting on December 31, 2024, would be included in the PY 1 baseline spending calculations that form a primary component of PY 1 preliminary target prices, the timing of PY 1 target price calculations would need to allow the time not only for such episodes to end but also for claims run-out to capture any additional spending and for baseline spending and target price calculations to be performed. Thus, we conclude that it is not feasible for CMS to provide target price data, or indeed baseline spending data, a full year prior to the applicable performance period.
Comment: A commenter reflected on the use of beneficiary-identifiable claims data from the 3-year baseline period, suggesting that these baseline period data should be considered in aggregate and not used for individual patient care decisions.
Response: We thank the commenter for sharing this view and concur that there are limitations to the use of baseline data for clinical decision making. We proposed to make available to TEAM participants multiple forms of baseline and performance period data such that participants may gain insights into multiple aspects of their care delivery and model performance. As stated in the proposed rule ( 89 FR 36474 ), we also expect that participants will work toward developing and improving their own data identification and analysis capabilities as part of their care improvement efforts.
After consideration of the comments received, we are finalizing at § 512.562(b)(6)(i) and § 512.562(d)(1)(i) the proposal to share beneficiary-identifiable data and regional aggregate data for a 3-year baseline period at least 1 month prior to the start of a performance year.
As discussed in the proposed rule, the availability of periodically updated raw and summary beneficiary-identifiable claims data and regional aggregate data would assist TEAM participants to identify areas where they might wish to change their care practice patterns, as well as monitor the effects of any such changes. With respect to these purposes, we considered what would be the most appropriate period for making updated raw and summary beneficiary-identifiable claims data and regional aggregate data available to TEAM participants, while complying with the HIPAA Privacy Rule's “minimum necessary” provisions, described in 45 CFR 164.502(b) and 164.514(d) . We noted that we believe that monthly data updates would align with a 30-day post-discharge episode window given the episode's duration and the need to share data in a timely manner and identify areas for care improvement. Accordingly, we proposed to make updated raw and summary beneficiary-identifiable claims data and regional aggregate data available for a given performance year to TEAM participants upon receipt of a request for such information and execution of a TEAM data sharing agreement with CMS, that meets CMS's requirements to ensure the ( print page 69847) applicable HIPAA conditions for disclosure have been met, as frequently as on a monthly basis during the performance year and continue sharing the claims data for up to 6 months beyond the end of that performance year to capture claims run out. We stated that we believe 6 months of claims run out is sufficient given that an internal review of Medicare claims data found that the majority of Medicare claims had been received, and were considered final, by 6 months after the date of service and is also consistent with how we proposed claims run out for the reconciliation process, as described in section X.A.3.d.(5). of the preamble of this final rule. [ 1010 ]
To accomplish this for the first performance year of the TEAM (2026), we proposed to provide, upon request and execution of a TEAM data sharing agreement with CMS, and in accordance with the HIPAA Privacy Rule, beneficiary-identifiable claims data and aggregate regional data from January 1, 2026, to December 30, 2026 on as frequently as a running monthly basis, as claims are available. We would continue sharing beneficiary-identifiable claims data and regional aggregate data for episodes in PY 1 for an additional 6 months, so until June 30, 2027, to capture claims run out for items and services billed during this time period. These datasets would represent all potential episodes that were initiated in 2026 and capture a sufficient amount of time, up to 6 months, for relevant claims to have been processed. We would limit the content of this data set to the minimum data necessary for the TEAM participant to conduct quality assessment and improvement activities and effectively coordinate care of its patient population. This data sharing process would continue each performance year of TEAM. We considered proposing to extend this period to capture more than 30 days of data or updating on a quarterly frequency. However, as discussed in the proposed rule, we do not believe this would benefit the TEAM participant since it may create challenges to timely identify potential TEAM beneficiaries for care coordination efforts. We sought comment on whether we should consider extending the period to capture more than 30 days of data or updating the data on a frequency other than monthly.
We sought comments on this proposal at proposed § 512.562(b)(6)(ii) and § 512.562(d)(1)(ii) to make beneficiary-identifiable data and regional aggregate data available on a monthly basis and for up to 6 months after a performance year.
The following is a summary of the public comments we received on the proposal to make beneficiary-identifiable data and regional aggregate data available to TEAM participants on a monthly basis and for up to 6 months after a performance year and our responses to these comments:
Comment: A couple of commenters expressed concern about the potential for data lags limiting the ability of participants to make real-time improvements. The commenters requested that CMS work to provide timely data as soon as feasible.
Response: We thank the commenters for sharing their concerns about data lags. We recognize that delays in data sharing can limit the utility of these data for participants in assessing and improving care. In recognition of the benefits of timely data for continuous improvement, we proposed to share raw and summary beneficiary-identifiable claims data as frequently as on a monthly basis during the performance year and for up to 6 months beyond the end of the performance year to capture claims run-out. As stated in the preamble of the proposed rule ( 89 FR 36478 ), we believe that the proposed timing of performance period data sharing aligns appropriately with the proposed 30-day episode length and the need for timely data.
Comment: A commenter requested that CMS include updated preliminary target prices as part of the monthly data sharing, or alternatively provide more detailed information on target price model coefficients to allow participants to proactively calculate target prices.
Response: We thank the commenter for their suggestions for additional data sharing surrounding target prices. We note that, as described in section X.A.3.d.(3).(b) of this final rule, TEAM preliminary target prices will be constructed at a regional level and shared with participants prior to the performance year, then will be updated with realized trends and participant-specific risk adjustments during reconciliation. As we note in the responses to comments in section X.A.3.d.(3).(b) of this final rule, coefficients will be calculated and made available to participants prior to the start of the performance year, so participants would be able to use them to estimate their episode-level target prices.
After consideration of the comments, we are finalizing at § 512.562(b)(6)(ii) and § 512.562(d)(1)(ii) the proposal to make beneficiary-identifiable data and regional aggregate data available on a monthly basis and for up to 6 months after a performance year.
We proposed that if a TEAM participant wishes to retrieve the beneficiary-identifiable data, the TEAM participant would be required to first complete, sign, and submit—and thereby agree to the terms of—a data sharing agreement with CMS, which we would call the TEAM data sharing agreement. We proposed to define the TEAM data sharing agreement as an agreement between the TEAM participant and CMS that includes the terms and conditions for any beneficiary-identifiable data being shared with the TEAM participant under § 512.562. Further, we proposed to require TEAM participants to comply with all applicable laws and the terms of the TEAM data sharing agreement as a condition of retrieving the beneficiary-identifiable data. We also proposed that the TEAM data sharing agreement would include certain protections and limitations on the TEAM participant's use and further disclosure of the beneficiary-identifiable data and would be provided in a form and manner specified by CMS. Additionally, we proposed that a TEAM participant that wishes to retrieve the beneficiary-identifiable data would be required to complete, sign, and submit a signed TEAM data sharing agreement at least annually. We stated that we believe that it is important for the TEAM participant to complete and submit a signed TEAM data sharing agreement at least annually so that CMS has up-to-date information that the TEAM participant wishes to retrieve the beneficiary-identifiable data and information on the designated data custodian(s). As described in greater detail later in this section, we proposed that a designated data custodian would be the individual(s) that a TEAM participant would identify as responsible for ensuring compliance with all privacy and security requirements and for notifying CMS of any incidents relating to unauthorized disclosures of beneficiary-identifiable data.
CMS believes it is important for the TEAM participant to first complete and submit a signed TEAM data sharing agreement before it retrieves any beneficiary-identifiable data to help protect the privacy and security of any beneficiary-identifiable data shared by CMS with the TEAM participant. There ( print page 69848) are important sensitivities surrounding the sharing of this type of individually identifiable health information, and CMS must ensure to the best of its ability that any beneficiary-identifiable data that it shares with TEAM participants would be further protected in an appropriate fashion.
We considered an alternative proposal under which TEAM participants would not need to complete and submit a signed TEAM data sharing agreement, but we concluded that, if we proceeded with this option, we would not have adequate assurances that the TEAM participants would appropriately protect the privacy and security of the beneficiary-identifiable data that we proposed to share with them. We also considered an alternative proposal under which the TEAM participant would need to complete and submit a signed TEAM data sharing agreement only once for the duration of the TEAM. However, we concluded that this similarly would not give CMS adequate assurances that the TEAM participant would protect the privacy and security of the beneficiary-identifiable data from CMS. We concluded that it is critical that we have up-to-date information and designated data custodians, and that requiring the TEAM participant to submit a TEAM data sharing agreement at least annually would represent the best means of achieving this goal.
We solicited public comment on our proposal to define TEAM data sharing agreement at § 512.505. We also sought comment on our proposal to require, in § 512.562(e)(2), that the TEAM participant agree to comply with all applicable laws and the terms of the TEAM data sharing agreement as a condition of retrieving the beneficiary-identifiable data, and on our proposal in § 512.562(e)(1) that the TEAM participant would need to submit the signed TEAM data sharing agreement at least annually if the TEAM participant wishes to retrieve the beneficiary-identifiable data.
The following is a summary of the public comments we received on the proposals to define the TEAM data sharing agreement, to require compliance with the terms of the TEAM data sharing agreement as a condition of retrieving the beneficiary-identifiable data, and to require submission of the TEAM data sharing agreement at least annually, and our responses to these comments:
Comment: A commenter expressed support and appreciation for the proposed protections surrounding the sharing of beneficiary-identifiable data with TEAM participants. The commenter reiterated that any data sharing should be conducted in a manner that protects patient privacy and allows all points of care to maximize lessons learned and implement quality improvement activities.
Response: We thank the commenter for their support and agree that appropriate protections must be ensured in the sharing of beneficiary-identifiable data. As described in the proposed rule ( 89 FR 36478 ), we proposed that a TEAM participant requesting to receive such data from CMS would be required to submit a TEAM data sharing agreement at least annually and comply with the terms of said agreement. As proposed, the data sharing agreement would include certain protections and limitations on the TEAM participant's use and further disclosure of the beneficiary-identifiable data in compliance with the requirements imposed on covered entities by the HIPAA regulations. The TEAM participant would be required to designate a data custodian who would be responsible for ensuring compliance with all privacy and security requirements and for notifying CMS of any unauthorized disclosures of beneficiary-identifiable data. As detailed in the proposed rule ( 89 FR 36479 ), the data sharing agreement would also:
- Require the TEAM participant to comply with additional privacy, security, breach notification, and data retention requirements,
- Impose the same terms and conditions for any downstream recipient of the beneficiary-identifiable data that is a business associate of the TEAM participant or performs a similar function for the TEAM participant, and
- Revoke the eligibility of a TEAM participant to receive beneficiary-identifiable data in the event of any misuse or disclosure of such data in violation of any applicable statutory or regulatory requirements.
Comment: A commenter indicated that the need to request data on a monthly basis would impose an unnecessary burden on participants and requested that CMS automatically provide data on a monthly basis.
Response: We thank the commenter for their input and concur that requiring participants to request claims data on a monthly basis would constitute an unnecessary burden on participants. We wish to clarify that we do not intend to require participants to submit a request for claims data each month. As stated in the preamble of the proposed rule ( 89 FR 36478 ), TEAM participants would be required to complete, sign, and submit a TEAM data sharing agreement at least annually to receive beneficiary-identifiable claims data on as frequently as a monthly basis as described in the preamble of the proposed rule ( 89 FR 36478 ). We believe this cadence appropriately balances participant burden with the need for CMS to maintain up-to-date information on participants and their data custodians.
After consideration of the comments received, we are finalizing at § 512.505 the definition of TEAM data sharing agreement as an agreement entered into between the TEAM participant and CMS that includes the terms and conditions for any beneficiary-identifiable data shared with the TEAM participant under § 512.562, which includes a minor change for clarity (adding “entered into”) from the language proposed. In addition, we are finalizing at § 512.562(e)(1) the proposal that the TEAM participant would need to submit the signed TEAM data sharing agreement at least annually if the TEAM participant wishes to retrieve the beneficiary-identifiable data.
We are also finalizing at § 512.562(e)(2) the proposed requirement that the TEAM participant agree to comply with all applicable laws and the terms of the TEAM data sharing agreement as a condition of retrieving the beneficiary-identifiable data.
We proposed that, under the TEAM data sharing agreement, TEAM participants would agree to certain terms, namely: (1) To comply with the requirements for use and disclosure of this beneficiary-identifiable data that are imposed on covered entities by the HIPAA regulations and the requirements of TEAM; (2) to comply with additional privacy, security, and breach notification requirements to be specified by CMS in the TEAM data sharing agreement; (3) to contractually bind each downstream recipient of the beneficiary-identifiable data that is a business associate of the TEAM participant to the same terms and conditions to which the TEAM participant is itself bound in its data sharing agreement with CMS as a condition of the downstream recipient's receipt of the beneficiary-identifiable data retrieved by the TEAM participant under TEAM; and (4) that if the TEAM participant misuses or discloses the beneficiary-identifiable data in a manner that violates any applicable statutory or regulatory requirements or that is otherwise non-compliant with the provisions of the TEAM data sharing agreement, the TEAM participant would no longer be eligible to retrieve the ( print page 69849) beneficiary-identifiable data and may be subject to additional sanctions and penalties available under the law. As discussed in the proposed rule, CMS believes that these terms for sharing beneficiary-identifiable data with TEAM participants are appropriate and important, as CMS must ensure to the best of its ability that any beneficiary identifiable data that it shares with TEAM participants would be further protected by the TEAM participant, and any business associates of the TEAM participant, in an appropriate fashion. We stated that we believe that these proposals would allow CMS to accomplish that.
We sought public comment on the additional privacy, security, breach notification, and other requirements that we would include in the TEAM data sharing agreement. CMS has these types of agreements in place as part of the governing documents of other models tested under section 1115A of the Act and in the Medicare Shared Savings Program. In these agreements, CMS typically requires the identification of data custodian(s) and imposes certain requirements related to administrative, physical, and technical safeguards relating to data storage and transmission; limitations on further use and disclosure of the data; procedures for responding to data incidents and breaches; and data destruction and retention. We noted that these provisions would be imposed in addition to any restrictions required by law, such as those provided in the HIPAA privacy, security and breach notification regulations. These provisions would not prohibit the TEAM participant from making any disclosure of the data otherwise required by law.
We also sought public comment on what disclosures of the beneficiary-identifiable data might be appropriate to permit or prohibit under the TEAM data sharing agreement. For example, we considered prohibiting, in the TEAM data sharing agreement, any further disclosure, not otherwise required by law, of the beneficiary-identifiable data to anyone who is not a HIPAA covered entity or business associate, as defined in 45 CFR 160.103 , or to an individual practitioner in a treatment relationship with the TEAM beneficiary, or that practitioner's business associates. Such a prohibition would be similar to that imposed by CMS in other models tested under section 1115A of the Act in which CMS shares beneficiary identifiable data with model participants.
We considered these possibilities because there exist important legal and policy limitations on the sharing of the beneficiary-identifiable data and CMS must carefully consider the ways in which and reasons for which we would provide access to this data for purposes of the TEAM. CMS believes that some TEAM participants may require the assistance of business associates, such as contractors, to perform data analytics or other functions using this beneficiary-identifiable data to support the TEAM participant's review of their care management and coordination, quality improvement activities, or clinical treatment of TEAM beneficiaries. CMS also believes that this beneficiary-identifiable data may be helpful for any HIPAA covered entities who are in a treatment relationship with the TEAM beneficiary.
We sought public comment on how a TEAM participant might need to, and want to, disclose the beneficiary-identifiable data to other individuals and entities to accomplish the goals of the TEAM, in accordance with applicable law.
The following is a summary of the public comments we received on potential needs for TEAM participants to disclose beneficiary-identifiable data to other individuals or entities to accomplish the goals of TEAM, and our responses to these comments:
Comment: A couple of commenters requested that we include provisions for data sharing among additional health care providers involved in providing care to TEAM beneficiaries. A commenter requested that CMS guarantee data sharing among physicians, other clinicians, and relevant non-clinical staff. This commenter indicated that hospital-level data are important for quality, safety, and cost improvement among anesthesiologists. Another commenter requested that CMS provide an option for TEAM participants to share claims data with collaborators, noting that such data sharing could especially benefit safety net providers and providers new to value-based care.
Response: We thank the commenters for their requests regarding further data sharing beyond the TEAM participant. We agree that multiple health care providers working within and beyond the TEAM participant organization may play a critical role in providing care for TEAM beneficiaries. We further recognize the benefit of sharing beneficiary-identifiable data with such providers as well as the provisions of the HIPAA Privacy Rule which permit disclosures of PHI between covered entities for certain “health care operations” purposes ( 45 CFR 164.506(c)(4) ).
As noted in the proposed rule, in alignment with the CJR model, we proposed to enter into a data sharing agreement with, and disclose beneficiary-identifiable data to, only the hospitals that are bearing risk for episodes, and that CMS would not enter into a data sharing agreement with the business associates of TEAM participants. We reiterate that, as stated in the final CJR rule ( 80 FR 73515 ), we believe that the hospitals that are specifically held financially responsible for an episode should make the determination as to which data are needed to manage care and care processes with their collaborators as well as which data they might want to re-disclose, if any, to their collaborators provided they are in compliance with the HIPAA Privacy Rule. We proposed that under the TEAM data sharing agreement, the TEAM participant receiving beneficiary-identifiable data would agree to contractually bind each downstream recipient of the beneficiary-identifiable data that is a business associate of the TEAM participant to the same terms and conditions to which the TEAM participant is itself bound in its TEAM data sharing agreement with CMS as a condition of the business associate's receipt of the beneficiary-identifiable data retrieved by the TEAM participant under TEAM.
After considering the comments received, we are finalizing as proposed the content of the TEAM data sharing agreement as described at § 512.562(e), including the contractual obligations for downstream recipients of the beneficiary-identifiable data as proposed at § 512.562(e)(1)(iii). Specifically, we are finalizing the proposed requirements at § 512.562(e)(1)(i) through (iv) that the TEAM data sharing agreement would:
- Require the TEAM participant to comply with the requirements for use and disclosure of this beneficiary-identifiable data that are imposed on covered entities by the HIPAA regulations and the requirements of the TEAM;
- Require the TEAM participant to comply with additional privacy, security, breach notification, and data retention requirements specified by CMS—with one modification, removing “in the TEAM data sharing agreement” from the description of these requirements, to avoid redundancy;
- Contractually bind and impose the same terms and conditions for any downstream recipient of the beneficiary-identifiable data that is a business associate of the TEAM participant; and
- Revoke the eligibility of a TEAM participant to receive beneficiary- ( print page 69850) identifiable data in the event of any misuse or disclosure of such data in violation of any applicable statutory or regulatory requirements or non-compliance with the provisions of the TEAM data sharing agreement.
We are finalizing one modification to use the defined term for “TEAM data sharing agreement” in the revocation provision under § 512.562(e)(1)(iv).
Under our proposal, the TEAM data sharing agreement would include other provisions, including requirements regarding data security, retention, destruction, and breach notification. For example, we are considering including, in the TEAM data sharing agreement, a requirement that the TEAM participant designate one or more data custodians who would be responsible for ensuring compliance with the privacy, security and breach notification requirements for the data set forth in the TEAM data sharing agreement; various security requirements like those found in other models tested under section 1115A of the Act, but no less restrictive than those provided in the relevant Privacy Act system of records notices; how and when beneficiary-identifiable data could be retained by the TEAM participant or its downstream participants of the beneficiary identifiable data; procedures for notifying CMS of any breach or other incident relating to the unauthorized disclosure of beneficiary-identifiable data; and provisions relating to destruction of the data. These are only examples and are not the only terms CMS would potentially include in the TEAM data sharing agreement.
We solicited public comment on this proposal that CMS, by adding § 512.562(e)(1)(ii), would impose certain requirements in the TEAM data sharing agreement related to privacy, security, data retention, breach notification, and data destruction.
Finally, CMS proposed, at § 512.562(e)(1)(iv), that the TEAM data sharing agreement would include a term providing that if the TEAM participant misuses or discloses the beneficiary-identifiable data in a manner that violates any applicable statutory or regulatory requirements or that is otherwise non-compliant with the provisions of the TEAM data sharing agreement, the TEAM participant would no longer be eligible to retrieve beneficiary-identifiable data under proposed § 512.562(b) and may be subject to additional sanctions and penalties available under law. We also proposed that if CMS determines that one or more grounds for remedial action specified in § 512.592(a) has taken place, CMS may discontinue the provision of data sharing and reports to the model participant. We proposed that CMS may take remedial action if the model participant misuses or discloses the beneficiary-identifiable data in a manner that violates any applicable statutory or regulatory requirements or that is otherwise non-compliant with the provisions of the TEAM data sharing agreement.
We solicited public comment on this proposal, to prohibit the TEAM participant from obtaining beneficiary-identifiable data pertaining to the TEAM if the TEAM participant fails to comply with applicable laws and regulations, the terms of the TEAM, or the TEAM data sharing agreement.
We received no public comments regarding the proposed additional content of the TEAM data sharing agreement, namely the proposed addition of privacy, security, breach notification, and data retention requirements and the proposal to prohibit the TEAM participant from obtaining beneficiary-identifiable data pertaining to the TEAM if the TEAM participant fails to comply with applicable laws and regulations, the terms of the TEAM, or the TEAM data sharing agreement. Thus, we are finalizing at § 512.562(e)(1)(ii) the inclusion of additional requirements, as proposed, and at § 512.562(e)(1)(iv) the prohibition of a TEAM participant from receiving beneficiary-identifiable data in the event of non-compliance, as proposed.
We noted in this proposed rule, the CMS Innovation Center has placed accountable care at the center of our comprehensive strategy, with a goal of 100 percent of Medicare FFS beneficiaries (and most Medicaid beneficiaries as well) being in an accountable care relationship by 2030. Achieving the goal of increasing the number of beneficiaries in accountable care relationships and testing models and innovations supporting access to high-quality, integrated specialty care across the patient journey—both longitudinally and for procedural or acute services—will greatly depend on numerous factors, including the models and initiatives available for providers in value-based payment, but also our ability to create incentives for providers and suppliers to coordinate care across different aspects of care. With TEAM, we have an opportunity to further integrate care during the transition from an acute event- an episode- back to longitudinal care relationships, such as primary care.
We stated in the proposed rule that acute care hospitals commonly refer patients back to primary care providers in the community upon discharge from the hospital, given the connection between ongoing care follow-up and reduced readmissions, among other benefits. While the hospital Conditions of Participation for discharge planning at § 482.43(a) outline requirements for referring patients to post-acute providers as well as community-based providers and suppliers, there is no specific requirement for referral back to a supplier, as defined in in section 1861(d) of the Act and codified at § 400.202, of primary care services, as defined in section 1842(i)(4) of the Act, at hospital discharge for all patients. Under TEAM, we proposed that TEAM participants be required to include in hospital discharge planning a referral to a supplier of primary care services for a TEAM beneficiary, on or prior to discharge from an anchor hospitalization or anchor procedure. We also proposed that the TEAM participant must comply with beneficiary freedom of choice requirements, as described in the Beneficiary Choice and Notification section: X.A.3.i.(2) of this final rule and codified at § 512.582(a), and not limit a TEAM beneficiary's ability to choose among Medicare providers or suppliers. If a TEAM participant fails to comply with requiring a referral to a supplier of primary care services during hospital discharge planning, then we proposed the TEAM participant would be subject to remedial action, as described in the Remedial Action section: X.A.1.f. of this final rule.
Referring TEAM beneficiaries to a supplier of primary care services would require the TEAM participant to confirm the TEAM beneficiary's primary care provider status during the anchor hospitalization or anchor procedure and make the referral to primary care services by the point of the hospital discharge. By requiring a referral to primary care services, TEAM would be used to connect TEAM beneficiaries with ongoing care beyond the course of the episode. Further, TEAM participants would be required to ensure TEAM beneficiaries preference of suppliers are considered to ensure proper beneficiary protections.
In the proposed rule we stated that we recognize that TEAM is comprised of procedural episodes, which may mean TEAM beneficiaries have a greater need to stay connected to their surgeon or specialist involved in their episode, rather than make a connection to primary care for ongoing care. Additionally, we also recognize requiring a referral to primary care services for all TEAM beneficiaries may ( print page 69851) increase TEAM participant burden. However, we believe many hospitals already have this perform this process as a standard of care for discharge planning, therefore the burden on TEAM participants should be minimal.
We sought comment on our proposal at § 515.564 to require TEAM participants during hospital discharge planning to make a referral to a supplier of primary care services for a TEAM beneficiary on or prior to discharge from the anchor hospitalization or anchor procedure. We also sought comment on whether there are other mechanisms or ways to connect the TEAM beneficiary back to a supplier of primary care services that would support a patient's continuum of care.
The following is a summary of comments we received related to the proposed referral to primary care services policy and our responses to those comments:
Comment: We received some comments of support for our proposed policy to require TEAM participants to make a referral to a supplier of primary care services by the point of discharge from anchor hospitalization or anchor procedure. Several commenters acknowledged the importance of ensuring a handoff to primary care services as a critical component in good care coordination and improved patient outcomes.
Response: We appreciate the support received and the acknowledgement from commenters that a referral to primary care is an important aspect of ensuring strong care coordination and striving to improve patient outcomes. We agree with these commenters that requiring a referral to primary care services upon discharge will serve to give the patient the best opportunity for collaborative care.
Comment: We received some comments from commenters who express concern that physician access issues could make adherence to this policy difficult for some TEAM participants. These commenters mention that access could be a barrier in ensuring a referral occurred by the point of discharge from the anchor hospitalization or anchor procedure, stating that lack of available primary care resources could limited a TEAM participant's ability to adequately meet this requirement and asking that CMS consider an appropriate amount of time for a referral to occur.
A commenter recommended the use of other types of providers, such as telehealth or nurse-led programs, to better support broad access for a primary care referral by the point of discharge from the anchor procedure or anchor hospitalization.
Another commenter expressed concern over requiring a referral back to primary care specifically for spinal fusion episodes. They mentioned that there is a shortage of primary care physicians and a growing number of primary care physicians who are no longer accepting Medicare patients due to the downward trend of Medicare payments. Because of these reasons, the commenter stated that this requirement could unduly impact access for patients in this model (either pre- or post-surgery).
Additionally, a couple of other commenters recommended that TEAM participants located in Health Professional Shortage Areas (HPSA) be given exemption, opt-out option, or episode extension from this policy as they may experience the greatest shortage of primary care providers in their communities.
Response: We acknowledge that access to primary care services varies based on many factors, including geographic location, market dynamics—among others. Furthermore, we acknowledge that there are areas with shortages of healthcare providers and varying volumes of providers accepting Medicare patients. However, we also acknowledge that ensuring a beneficiary is connected to a supplier of primary care services serves as a critical component to ensuring the patient has continued care coordination upon discharge from their anchor hospitalization or anchor procedure across all areas, even those with areas with shortages of providers. We also do not anticipate that any one clinical episode category, such as spinal fusion, included in TEAM would experience greater issues with access.
Additionally, we recognize that a referral to primary care services is a common component to hospital discharge planning procedure and, as such, expects that many TEAM participants, regardless of access issues or HSPA status, will already have this policy integrated into their standardized discharge planning procedures.
Comment: A commenter suggested that rural hospitals generally—and Sole Community Hospitals (SCH) and Medicare Dependent Hospitals (MDH) in particular—also often do not have robust care networks that are needed to succeed as TEAM participants. This commenter mentions that to be effective in the model, hospitals would also likely need to form networks with post-acute care providers, among others. They state that many communities served by SCHs and MDHs are experiencing a shortage of primary care clinicians, which may make it difficult for participating hospitals to comply with the primary care referral requirement for reasons outside their control. Further, they state some rural communities simply do not have post-acute care providers. Those that do have limited options, and those post-acute providers may decline to collaborate with the hospitals in the ways that would be needed to succeed under TEAM.
Response: As mentioned in comment responses above, we recognize that access to primary care will vary depending on many factors. We also recognize that rural hospitals, including SCHs and MDHs, serve special patient populations and after often located in areas that have the potential to present unique challenges. However, these factors do not discount that these beneficiaries still need to be connected with a primary care provider to support ongoing care coordination beyond the scope of their TEAM episode.
Additionally, CMS disagrees that this policy will require providers to form networks—with primary care, post-acute care, or other types of healthcare providers—in order to be successful and adhere to this policy.
Comment: Some commenters expressed concern over the risk of disrupting a patient's existing primary care relationship through this policy. These commenters state the importance of ensuring patients with an existing primary care relationship be “tucked back into” their provider and not be referred to a different supplier of primary care services. A few commenters submitted comments requesting that TEAM participants be required to make referral decisions for primary care based on the patient's PCP status determined upon admission, thus ensuring TEAM participants would have the most up to date information on a patient's existing primary care relationship before making a referral and limit the risk the patient is referred away from their existing primary care provider. Additionally, a commenter encouraged CMS to create safeguards that can be used to monitor that a warm hand-off occurs back to the patient's existing primary care provider using CPT (Current Procedural Terminology) II codes, like the CPT II code used in Advanced Care Planning (ACP).
Response: We agree with these commenters on the importance of maintaining a patient's relationship with their existing primary care provider. Disrupting this relationship does not serve the patient's best interests and could cause delays or gaps ( print page 69852) in providing the best care due to lack of historical knowledge and relationship between provider and patient.
CMS also agrees that TEAM participants would benefit from identifying the patient's primary care provider upon admission to their anchor hospitalization or anchor procedure and we expect that many TEAM participants already have this step built into their standardized admission practices.
Although we recognize the value in monitoring this policy through a mechanism such as HCPCS code present in claims data, CMS does not agree that these kinds of safeguards should be implemented. Specifically for the request to use HCPCS coding to identify advance care planning conversations, we do not believe that requiring an advance care planning conversation to occur during a specific window of the beneficiary's episode to be the most effective. CMS's expectation is that many beneficiaries are having these conversations with their providers at various times, including prior to an episode initiating. Additionally, this kind of claims-based monitoring would be requiring increased resources and burden to both TEAM participant and CMS. As mentioned above, we proposed the TEAM participant would be subject to remedial action, as described in the Remedial Action section: X.A.1.f. of this final rule, which we believe is a sufficient guardrail to encourage adherence to this policy by TEAM participants.
Comment: A few commenters mention concern that the policy to require a referral to a supplier of primary care services could cause higher referrals to in-network or hospital-system affiliated providers, leading to an imbalance of referrals to hospital affiliated over community practices or reduced patient volume to independent practices or practices not included in any preferred network.
Response: As mentioned above, we recognize the important of ensuring a patient's existing primary care relationship not be disrupted due to this policy. Additionally, TEAM participants must ensure they comply with beneficiary freedom of choice requirements. CMS urges TEAM participants to make referral decisions based on timely connection to a primary care provider and not base referrals on their own profit or benefit from retaining a beneficiary within any specific network.
Comment: We received a few comments where commenters opposed the requirement to refer to only a primary care supplier and asked CMS to consider making the referral policy more flexible around which physician is engaged. These commenters state that not all patients would benefit from a primary care visit upon discharge from their TEAM episode, but rather may benefit from simply seeing their surgeon post-discharge.
Response: As TEAM is designed include multiple procedurally based clinical episode categories, we acknowledge that many beneficiaries that trigger a TEAM episode will likely engage in subsequent appointments with their specialist for follow-up and post-surgical care. We encourage these visits to take place and recognize the importance of the continued care from the patient's specialist after a procedure. However, this does not negate the importance of also ensuring the patient is engaged with a primary care provide to support their long-term care after the immediate care surrounding the patient's procedure has concluded. By requiring a referral to primary care, this creates opportunity for the patient's specialist and primary care providers to engage and support the patient with more collaborative care.
Comment: We received a comment that mentioned that TEAM covers the major surgeries that fall within the 90-day global period and asked for clarification on how a primary care provider would be compensated for the follow-up care provided to the patient during the 90-day global period.
Response: All providers furnishing services during a TEAM episode will be reimbursed per the applicable CMS fee schedule; CMS will not be issuing a global payment or any capitated payments as part of TEAM. We encourage this commenter and any other stakeholders with questions on how providers will be compensated to review sections on episode construction and reconciliation in the Items and Services Included in Episodes section: X.A.3(b)(5) and the Process for Reconciliation section: X.A.3(d)(5) of this final rule.
Comment: A commenter in support of this policy specifically asked CMS to consider how to use the referral process to ensure that primary care suppliers involved in care transitions are taking steps to prevent opioid misuse. This commenter states this could be done through requiring TEAM participants to counsel patients prior to discharge about their pain management options to ensure they are adequately informed about the benefits and risks of each potential treatment plan, document in the medical record a rationale for prescribing opioids, if applicable, together with a plan for (to the extent clinically appropriate) transitioning the beneficiary to a non-opioid alternative over the course of the patient's treatment journey, and, for beneficiaries prescribed opioids at discharge, establishing a care plan that involves joint monitoring by the TEAM participant and primary care supplier to monitor for any signs of opioid misuse or opioid use disorder.
Response: We appreciate this commenter's acknowledgement of the risk of opioid overuse and recommendation that suppliers of primary care are monitoring their patient's use of opioids and identify potential overuse. CMS agrees that primary care suppliers should be identifying potential overuse for their patients. However, at this time, CMS does not plan to incorporate any formal policy surrounding opioid management or misuse avoidance. We do expect that primary care providers are actively engaged in conversations with their beneficiaries about all medications prescribed and documenting medications used to ensure they are able to identify potential misuse and avoid misuse.
Comment: A commenter asked CMS to consider how to leverage digital tools, such as a digital platform presented in a dashboard interface, to aid referrals between specialists and primary care physicians. The commenter states that this kind of interface could display information about an existing relationship with a primary care provider or offer options for discharge referrals based on the patient's coverage and specific need, aiding in better care coordination.
Response: CMS agrees with this commenter that the ability to leverage digital tools like a dashboard interface can strongly support collaboration, data-sharing, and communication between providers, such as specialists and primary care providers. However, creating and maintaining these kinds of digital tools would cause a significant drain on time and resources for CMS and potentially, TEAM participants. CMS encourages participants to consider how they can leverage digital tools to aid their success in TEAM; however, CMS will not be incorporating the support for any digital tool into TEAM at this time.
Comment: We received a comment from a commenter who recommended that primary care referrals only be made for a beneficiary without an existing primary care relationship. For a beneficiary with an existing primary care relationship, that the TEAM participant shares medical record documentation with the primary care ( print page 69853) supplier to streamline care coordination.
Response: As mentioned in earlier comments, connection to a primary care provider upon discharge from a patient's anchor hospitalization or anchor procedure is critical to ensuring care coordination. CMS agrees that sharing of medical record documentation should be included in this process so primary care providers have the most up-to-date information so as to make this most informed decisions for their patient. However, CMS does not believe that the sharing of medical record documentation eliminates the need for patients to be referred for a visit to see their primary care providers. This connection between primary care and patient ensures all patients, even thoughts with existing primary care relationships, are involving the primary care providers in the ongoing care the patient needs.
Comment: Some commenters submitted comments asking to clarify how this policy would be monitored by CMS. A commenter mentioned there could be an increased risk for patients with high social determinants of health concerns and it would be helpful to know if these factors would be included in evaluation or monitoring of the policy.
Response: As mentioned above, if a TEAM participant fails to comply with requiring a referral to a supplier of primary care services during hospital discharge planning, the TEAM participant would be subject to remedial action, as described in the Remedial Action section: X.A.1.f. of this final rule. CMS does not plan to evaluate or monitor participants adherence to this policy for any specific populations of patients, such as those with social determinant of health concerns.
Comment: We received a few comments opposing the policy to require a referral to primary care services as it could add administrative burden on the TEAM participant. Commenters stated that requiring primary care referral at discharge does not further incentivize hospitals to coordinate care and states there are enough incentives in place now to coordinate care without additional documentation burden to the participant.
Response: CMS disagrees with these commenters and strongly believes that referring a patient to primary care upon discharge from anchor hospitalization or anchor procedure supports better care coordination and ongoing care for the patient beyond the care surrounding their procedure. For TEAM participants who do not currently refer to primary care at discharge, we understand this will be an additional administrative component to build in their discharge planning procedures. However, we also believe that most TEAM participants already have a referral to primary care built into their discharge planning, which would not add additional administrative burden for those hospitals.
After consideration of the public comments we received, we are finalizing our proposal as proposed that TEAM participants be required to include in hospital discharge planning a referral to a supplier of primary care services for a TEAM beneficiary, on or prior to discharge from an anchor hospitalization or anchor procedure at § 512.564. We also finalize our proposal that the TEAM participant must comply with beneficiary freedom of choice requirements, as described in Beneficiary and Notification section: X.A.3.i.(2) of this final rule and codified at § 512.582(a), and not limit a TEAM beneficiary's ability to choose among Medicare providers or suppliers.
We stated in the proposed rule ( 89 FR 36480 ) that in the Quality Payment Program ( 42 CFR 414.1415 ), an APM must meet three criteria to be considered an Advanced APM:
- Beginning with the CY 2025 Qualifying APM Participant (QP) performance period, an Advanced APM must require all eligible clinicians in each participating APM Entity, or for APMs in which hospitals are the participants, each hospital, to use Certified Electronic Health Record Technology (CEHRT).
- An Advanced APM must include quality measure performance as a factor when determining payment to participants for covered professional services under the terms of the APM.
- Meet the financial risk standard under 42 CFR 414.1415(c)(1) or (2) and the nominal amount standard under 42 CFR 414.1415(c)(3) or (4) .
We sought to align the design of TEAM with the Advanced APM criteria in the Quality Payment Program and enable CMS to have the necessary information on eligible clinicians to make the requisite QP determinations. Eligible clinicians, as defined in 42 CFR 414.1305 , that are captured on a CMS-maintained list for the APM entity, as defined in 42 CFR 414.1305 , may be eligible to receive benefits for participating in an Advanced APM, including burden reduction and financial incentives. We proposed that the TEAM participant would be considered the APM entity, but that the TEAM participant's eligible clinicians may be assessed for QP determinations depending on which track the TEAM participant is in and whether the CEHRT criteria are met ( 89 FR 36480 ). However, we also sought to ensure the design of TEAM meets the Merit-based Incentive Payment System (MIPS) APM criteria and that CMS has the necessary information on MIPS eligible clinicians, as defined in 42 CFR 414.1305 , so that they may be eligible for certain scoring benefits under MIPS. We proposed to adopt two different APM options for TEAM—an AAPM option in which TEAM participants would attest to meeting the CEHRT standards and in which the TEAM participant's eligible clinicians may be assessed for QP determinations (to the extent TEAM is determined to be an Advanced APM for Track 2 and Track 3), and a non-AAPM option in which TEAM participants would not meet CEHRT or financial risk standards and in which the TEAM participant's MIPS eligible clinicians may be assessed for reporting and scoring through the APM Performance Pathway (APP) (to the extent the TEAM is determined to be a MIPS APM for all tracks).
In the proposed rule we stated an Advanced APM must require participants to use CEHRT ( 42 CFR 414.1415(a) ), make payment based on quality measures ( 42 CFR 414.1415(b) ) and meet financial risk standards ( 42 CFR 414.1415(c) ) ( 89 FR 36481 ). We proposed to have two APM options in TEAM: a non-Advanced APM (non-AAPM) option and an Advanced APM (AAPM) option ( 89 FR 36481 ). The non-AAPM option would be for TEAM participants that do not meet the CEHRT or financial risk standards. These TEAM participants may still be considered APM entities in a MIPS APM. The AAPM option would be for TEAM participants in Tracks 2 and 3 that meet the CEHRT and financial risk standards. These TEAM participants would be considered APM entities in an Advanced APM., TEAM participants in Track 1 would automatically be assigned into the non-AAPM option since Track 1 would have no downside financial risk. The financial risk that we proposed in Tracks 2 and 3 would meet the generally applicable nominal amount standard, as defined in 42 CFR 414.1415(c)(3) , but there may be TEAM participants in Tracks 2 and 3 who do not meet the CEHRT standard. TEAM participants in Tracks 2 or 3 that do not meet and attest to the CEHRT use ( print page 69854) requirement would fall into the non-AAPM option of TEAM, but these TEAM participants may still be considered APM entities in a MIPS APM. TEAM participants that participate in Tracks 2 or 3 and meet and attest to the CEHRT use requirement would be in the AAPM option of TEAM.
We proposed to require TEAM participants who wish to participate in the AAPM option to attest to meeting the CEHRT use requirement that meets the CEHRT definition in our regulations at section 414.1305 on an annual basis prior to the start of each performance year in a form and manner and by a date specified by CMS ( 89 FR 36481 ). We proposed that the TEAM participant would be required to retain and provide CMS access to the attestation upon request. We further proposed that meeting and attesting to the CEHRT use criteria would be voluntary, and that CMS would assign TEAM participants who choose not to do so to the non-AAPM option. Lastly, we proposed to require TEAM participants who wish to participate in the AAPM option to provide their CMS Electronic Health Record (EHR) Certification IDs on an annual basis prior to the end of each performance year in a form and manner and by a date specified by CMS.
We stated in the proposed rule that we believe that a TEAM participant's decision to meet and attest to the CEHRT use criteria would not create significant additional administrative burden for the TEAM participant ( 89 FR 36481 ). Moreover, the choice of whether to meet and attest to the CEHRT use criteria would not otherwise affect the TEAM participant's requirements or opportunities under the model. However, a TEAM participant's decision to attest to CEHRT use may affect the ability of its clinicians to qualify as a QP. In other words, if a TEAM participant chose not to attest to CEHRT use, its clinicians would not be assessed for QPs status.
We sought comment on our proposals for the TEAM Advanced APM options and the associated requirements at § 512.522. We also sought comment on our proposed definitions for the AAPM option and non-AAPM option at § 512.505.
The following is a summary of comments we received on our proposals for the TEAM Advanced APM options, as well as comments on our proposed definitions for the AAPM option and non-AAPM option.
Comment: A commenter supports CMS' proposals related to Advanced APM options and QP status under TEAM and suggests that CMS can reduce burden by not requiring TEAM participants to report on the MIPS Promoting Interoperability requirement. The commenter is concerned that CMS will require APM Participants to report on the MIPS Promoting Interoperability requirement in 2025, which could present a non-financial disincentive to APM Participation.
Response: We thank the commenter for their support of CMS' proposed Advanced APM options under TEAM, and for their recommendation regarding the reduction of burden for APM Participants. The recommendation made by the commenter is related to policies set by the Quality Payment Program (QPP) and are beyond the scope of the proposed TEAM. CMS will consider this recommendation for future use; however, we are unable to adopt this recommendation regarding QPP policies under this proposed rule.
Summary: A commenter seeks clarification on TEAM requirements regarding the use of Certified Electronic Health Record Technology (CEHRT) for TEAM Participants and Collaborators.
Response: We thank the commenter for their interest in the TEAM CEHRT requirements. The use of CEHRT is not required for TEAM Participants or TEAM collaborators, however, CMS proposes two APM options in TEAM: an Advanced APM (AAPM) option and a non-Advanced APM (non-AAPM) option. The AAPM option would be for TEAM Participants in Track 2 and 3 that meet the financial risk standards and attest to meeting the CEHRT requirements. In the non-AAPM option, that do not meet the financial risk standards and/or do not attest to meeting the CEHRT requirements.
Comment: A commenter recommended that CMS should reduce QP determination thresholds around Medicare Part B payments and Medicare Beneficiaries for TEAM Participants and Collaborators to ensure that they are provided relief from reporting under MIPS if they do not meet QP thresholds.
Response: We thank the commenter for their recommendation regarding the QP determination thresholds, and the potential burden faced by TEAM Participants and Collaborators who do not meet the QP thresholds. The recommendation made by the commenter is related to policies set under the Quality Payment Program (QPP) and are beyond the scope of the proposed TEAM. CMS will consider this recommendation for future use; however, we are unable to adopt this recommendation regarding QPP policies under this proposed rule.
After consideration of the public comments we received, we are finalizing the TEAM Advanced APM options and the associated requirements at § 512.522, as well as the proposed definitions for the AAPM option and non-AAPM option at § 512.505.
We proposed that each TEAM participant would be required to submit information about the eligible clinicians or MIPS eligible clinicians who enter into financial arrangements with the TEAM participant for purposes of supporting the TEAM participants' cost or quality goals as discussed in section X.A.3.g. of the preamble of this final rule ( 89 FR 36481 ). This information would enable CMS to make determinations as to eligible clinicians who could be considered QPs based on the services furnished under TEAM (to the extent the model is determined to be an AAPM) and would be necessary for APP reporting and scoring for MIPS eligible clinicians (to the extent the model is determined to be a MIPS APM), We proposed that for purposes of TEAM, the eligible clinicians or MIPS eligible clinicians could be: (1) TEAM collaborators, as described in section X.A.3.g.(3). of the preamble of this final rule, engaged in sharing arrangements with a TEAM participant; (2) PGP, NPPGP, or TGP members who are collaboration agents engaged in distribution arrangements with a PGP, NPPGP, or TGP that is a TEAM collaborator, as described in section X.A.3.g.(5). of the preamble of this final rule; or (3) PGP, NPPGP, or TGP members who are downstream collaboration agents engaged in downstream distribution arrangements with a PGP, NPPGP, or TGP that is also an ACO participant in an ACO that is a TEAM collaborator, as described in section X.A.3.g.(6). of the preamble of this final rule. The list of physicians and nonphysician practitioners in these three groups that we proposed to require TEAM participants to submit to CMS would satisfy the criteria to be considered an Affiliated Practitioner List, as defined in § 414.1305. We proposed to use the list submitted by TEAM participants to make determinations regarding which physicians and nonphysician practitioners should receive QP determinations or be reported for the APP based on the services they furnish under TEAM ( 89 FR 36481 ).
We proposed for the reasons detailed above that each TEAM participant with eligible clinicians or MIPS eligible clinicians must submit to CMS a financial arrangements list in a form and manner and by the date specified by ( print page 69855) CMS on a quarterly basis during each performance year, or attest that there are no individuals to report on the financial arrangements list ( 89 FR 36481 ). We stated in the proposed rule that we believe submission of the financial arrangements list on a quarterly basis would align with the Quality Payment Program's QP determination dates, as described in § 414.1425. We proposed to define the financial arrangements list as the list of eligible clinicians or MIPS eligible clinicians that have a financial arrangement with the TEAM participant, TEAM collaborator, collaboration agent, or downstream collaboration agent. We proposed that the TEAM participant would be required to retain and provide CMS access to the financial arrangements list upon request. The proposed list must include the following information:
- For each TEAM collaborator who is a physician, nonphysician practitioner, or therapist during the performance year—
++ The name, tax identification number (TIN), and national provider identifier (NPI) of the TEAM collaborator; and
++ The start date and, if applicable, end date, for the sharing arrangement between the TEAM participant and the TEAM collaborator.
- For each collaboration agent who is a physician, nonphysician practitioner, or therapist during the performance year—
++ The name, TIN, and NPI of the collaboration agent and the name and TIN of the TEAM collaborator with which the collaboration agent has entered into a distribution arrangement; and
++ The start date and, if applicable, end date, for the distribution arrangement between the TEAM collaborator and the collaboration agent.
- For each downstream collaboration agent who is a physician or nonphysician practitioner, or therapist during the performance year—
++ The name, TIN, and NPI of the downstream collaboration agent and the name and TIN of the collaboration agent; and
++ The start date and, if applicable, end date, for the downstream distribution arrangement between the collaboration agent and the downstream collaboration agent.
- If there are no individuals that meet the reporting criteria above for TEAM collaborators, collaboration agents, or downstream collaboration agents, then the TEAM participant must attest on a quarterly basis in a form and manner and by a date specified by CMS that there are no individuals to report on the financial arrangements list.
We indicated in the proposed rule that while the submission of the financial arrangements list may create some additional administrative burdens for certain TEAM participants, we expect that TEAM Participants could modify their contractual relationships with their TEAM collaborators and, correspondingly, require those TEAM collaborators to include similar requirements in their contracts with collaboration agents and in the contracts of collaboration agents with downstream collaboration agents ( 89 FR 36482 ).
In the proposed rule we recognize there may be physicians and nonphysician practitioners who would not be listed on the financial arrangements list because they have not entered into a financial arrangement as a TEAM collaborator, collaboration agent, or downstream collaboration agent, but who may nevertheless participate in TEAM activities, as defined at § 512.505, and may be eligible for QP determinations or eligible for APP reporting because they are affiliated with and support the APM Entity. We proposed that, in order to capture these physicians and nonphysician practitioners who are not listed on the TEAM participant's financial arrangements list for QP determinations or APP reporting, TEAM participants must also submit to CMS a clinician engagement list in a form and manner and by a date specified by CMS on a quarterly basis every performance year. We proposed to use the clinician engagement list for assessing QP determinations and for APP reporting. The submission of the clinician engagement lists may create some additional administrative burdens for TEAM participants, but we expect the effort to be worthwhile since some of these QP determinations may result in eligible clinicians receiving burden reduction benefits and financial incentives, and some MIPS eligible clinicians may receive MIPS APM scoring benefits ( 89 FR 36482 ).
We proposed to define the clinician engagement list as the list of eligible clinicians or MIPS eligible clinicians that participate in TEAM activities and have a contractual relationship with the TEAM participant, and who are not listed on the financial arrangements list ( 89 FR 36482 ). We proposed that the TEAM participant must submit the list to CMS on a quarterly basis during each performance year in a form and manner and by a date specified by CMS or attest that there are no individuals to report on the clinician engagement list. We believe submission of the clinician engagement list on a quarterly basis would align with the Quality Payment Program's QP determination dates, as described in § 414.1425. We proposed that the TEAM participant would be required to retain and provide CMS access to the clinician engagement list upon request. We proposed that the clinician engagement list must include the following information:
- For each physician, nonphysician practitioner, or therapist who is not listed on the TEAM participant's financial arrangements list during the performance year but who does have a contractual relationship with the TEAM participant and participates in TEAM activities during the performance year—
++ The name, TIN, and NPI of the physician, nonphysician practitioner, or therapist; and
++ The start date and, if applicable, end date, for the contractual relationship between the physician, nonphysician practitioner, or therapist and the TEAM participant.
- We proposed that if there are no individuals that meet the requirements to be reported on the clinician engagement list, then the TEAM participant must attest on a quarterly basis in a form and manner and by a date specified by CMS that there are no individuals to report on the clinician engagement list ( 89 FR 36482 ).
We sought comments on the proposal to require TEAM participants to submit a financial arrangements list and clinician engagement list on a quarterly basis or attest that there are no individuals to report. We were especially interested in comments about approaches to information submission, including the content of the lists, and periodicity and method of submission to CMS that would minimize the reporting burden on TEAM participants while providing CMS with sufficient information about eligible clinicians to facilitate QP determinations and APP reporting to the extent that TEAM is considered to be an Advanced APM for Track 2 and Track 3 and a MIPS APM for all tracks, respectively. The following is a summary of the comments we received regarding the requirement for TEAM participants to submit a financial arrangements list and clinician engagement list to provide CMS with sufficient information about eligible clinicians to facilitate QP determinations and APP Reporting.
Comment: A commenter expressed support and approval of CMS' proposal to use the clinician engagement list for assessing QP determinations and for APP reporting. ( print page 69856)
Response: We thank the commenter for their support and approval of the TEAM proposal regarding the use of the clinician engagement list.
After consideration of the public comments we received, we are finalizing the requirement that TEAM participants submit a quarterly financial arrangements list and clinician engagement list, as applicable, in our regulation at § 512.522.
In the proposed rule we stated that improved interoperability of software systems and tools used to manage patients supports the goals of value-based care, enabling care coordination and data-driven decision making to improve outcomes and lower healthcare expenditures. Hospitals use electronic health record (EHR) systems to document patient medical history, which may include clinical data relevant to that person's care, including demographics, clinical notes, medications, vital signs, past medical and surgical history, immunizations, laboratory data and radiology reports. The EHR also has the ability to support other care-related and administrative activities directly or indirectly through various interfaces, including clinical decision support, quality improvement, and population-health outcomes reporting. While EHRs also include capabilities to coordinate care by sharing data in a structured system with other health care providers, health information exchanges (HIEs) and health information networks (HINs), as defined in 45 CFR 171.102 , have played an increasingly important role in assisting hospitals to connect with other health care providers and ensure that information supporting care coordination is consistently shared. [ 1011 ] A hospital may be connected to an HIE or HIN that focuses on exchange within a defined geographic area, or nationally across systems and regions. Evidence suggests that participation with an entity facilitating cross-system exchange may improve patient outcomes, including decreased hospital readmission rates, as well as decreased utilization, such as repeat laboratory or radiology studies. [ 1012 1013 ]
In the proposed rule, we stated that despite the growth of HIEs and HINs, important gaps remain for an infrastructure that supports the seamless exchange of clinical data across disparate healthcare organizations and software vendors. Barriers to interoperability create silos that limit care coordination between hospitals and other health care providers, especially during care transitions such as a patient being discharged from a hospital to a post-acute care facility. Existing HHS and CMS initiatives aim to support health care organizations engaging in interoperable exchange of health information. The Office of the National Coordinator for Health Information Technology (ONC) launched The Trusted Exchange Framework and Common Agreement (TEFCA), which establishes a universal governance, policy, and technical floor for nationwide interoperability; simplifies connectivity for organizations to securely exchange information to improve patient care, enhance the welfare of populations, and generate health care value; and enables individuals to gather their healthcare information. [ 1014 ]
CMS acknowledged the importance of TEFCA in the FY 2023 Inpatient Prospective Payment System (IPPS) final rule ( 87 FR 48780 ) by adding the Enabling Exchange under TEFCA ( 87 FR 49329 ) as a new measure under the Health Information Exchange Objective for the Medicare Promoting Interoperability Program. Participants in the Medicare Promoting Interoperability Program may also earn credit for the Health Information Exchange Objective by reporting on the previously finalized Health Information Exchange (HIE) Bidirectional Exchange measure ( 86 FR 45465 ).
In the CY 2023 Physician Fee Schedule final rule ( 87 FR 70067 through 70071 ), CMS also added a new optional measure, Enabling Exchange Under TEFCA, to the Health Information Exchange objective for the Merit-based Incentive Payment System (MIPS) Promoting Interoperability performance category beginning with the CY 2023 performance period/2025 MIPS payment year. Currently, for the CY 2024 performance period/2026 MIPS payment year, MIPS eligible clinicians may fulfill the Health Information Exchange objective via three avenues by reporting: (1) the two Support Electronic Referral Loops measures; (2) the Health Information Exchange Bidirectional Exchange measure; or (3) the Enabling Exchange under TEFCA measure ( 88 FR 79357 through 79362 ).
In the proposed rule ( 89 FR 36482 ) we stated that we would like to support TEAM participants' interoperability efforts that could lead to best practices across U.S. health care landscape. However, we recognized that given the existing federal interoperability initiatives, we do not want to create duplicate efforts or create unnecessary burden on TEAM participants. We sought comment on how CMS can promote interoperability in the proposed TEAM, in particular, to what extent TEAM participants are planning on participating in TEFCA in the next 1-2 years, as well as other means by which interoperability may support care coordination for an episode. Any further proposals related to interoperability included in TEAM would be done in future notice and comment rulemaking.
We thank commenters for their input and will address these comments, along with further proposals, in future notice and comment rulemaking.
TEAM is intended to enable CMS to better understand the effects of bundled payments models on a broader range of Medicare providers and capture a greater number of episodes of care than what is currently available under the CJR model and BPCI Advanced. Obtaining information that is representative of a wide and diverse group of providers and episodes of care will best inform us on how such a payment model might function were it to be more fully integrated within the Medicare program. All CMS Innovation Center models, which would include TEAM, are rigorously evaluated on their ability to improve quality and reduce costs. In addition, we routinely monitor CMS Innovation Center models for potential unintended consequences of the model that run counter to the stated objective of lowering costs without adversely affecting quality of care. Outlined later in this section are the design and evaluation methods, the data collection methods, key evaluation research questions, and the evaluation period and anticipated reports for TEAM.
In the proposed rule we stated that our evaluation methodology for TEAM would be consistent with the CMS Innovation Center evaluation ( print page 69857) approaches we have taken in other projects such as the BPCI initiative, BPCI Advanced and the CJR model, and o vcxther CMS Innovation Center models. Specifically, the evaluation design and methodology for the proposed TEAM would allow for a comparison of historic patterns of care among TEAM participants to any changes made in these patterns in response to the TEAM. In addition, the overall design would include a comparison of TEAM participants with hospitals not participating in TEAM to help us discern simultaneous and competing provider and market level forces that could influence our findings.
We indicated in the proposed rule that the evaluation methodology for this model builds upon the fact that we proposed CBSAs to be selected for participation in the model based on a stratified random assignment. In this approach, researchers evaluate the effects of the model on outcomes of interest by directly comparing CBSAs that are randomly selected to participate in the model to a comparison group of CBSs that were not randomly selected for the model (but could have been). Randomized evaluation designs of this kind are widely considered the “gold standard” for social science and medical research because they ensure that the systematic differences are reduced between units that do and do not experience an intervention, which ensures that (on average) differences in outcomes between participating and non-participating units reflect the effect of the intervention.
In the proposed rule we stated that we plan to use a range of analytic methods, including regression and other multivariate methods appropriate to the analysis of stratified randomized experiments to examine each of our measures of interest. Measures of interest could include, for example, quality of and access to care, utilization patterns, expenditures, and beneficiary experience. With these methodologies, we would be able to examine the experience of the TEAM participants over time relative to those in the comparison group controlling for as many of the relevant confounding factors as is possible. The evaluation would also include rigorous qualitative analyses to capture the evolving nature of care delivery transformation.
In the proposed rule we stated that in our design, we plan to evaluate the impact of the TEAM at the geographic unit level, the hospital level, and at the patient level. We also considered various statistical methods to address factors that could confound or bias our results. For example, we would use statistical techniques to account for clustering, that is how groups of patients may have commonalities in outcomes by receiving care in the same hospitals or in the same markets. Accounting for clustering ensures that we do not overstate our effective sample size by failing to recognize how performance of hospitals in each market may not be fully independent of one another. Alternatively, accounting for clustering may improve statistical precision or allow us to examine better how patterns of performance vary across hospitals. Thus, in our analysis, if a large hospital consistently has poor performance, clustering would allow us to still be able to detect improved performance in the other, smaller hospitals in a market rather than placing too much weight on the results of one hospital.
In the proposed rule we stated that we plan to use various statistical techniques to examine the effects of the TEAM while also taking into account the effects of other ongoing interventions such as Medicare Shared Savings Program. For example, we considered additional regression techniques to help identify and evaluate the incremental effects of adding the TEAM in areas where patients and market areas are already subject to other models as well as potential interactions among these efforts.
We considered multiple sources of data to evaluate the effects of TEAM. In the proposed rule we stated that we expect to base much of our analysis on secondary data sources such as the Medicare FFS claims. Beneficiary level claims data would be analyzed to estimate expenditures in total and by type of provider and service, as well as whether or not there was an inpatient hospital readmission. In conjunction with the secondary data sources mentioned previously, we considered collecting primary data through a CMS-administered survey, guided interviews and focus groups with beneficiaries who were cared for and resulting in a TEAM episode during the performance year. The survey would be administered to beneficiaries who were cared for as patients within a TEAM episode or similar beneficiaries selected as part of a control group. The primary focus of a survey would be to obtain information on the beneficiary's experience in TEAM episodes relative to usual care as represented in the control group. The administration of this beneficiary survey would be coordinated with administration of the HCAHPS (Hospital Consumer Assessment of Healthcare Providers and Systems) survey to not conflict with or compromise the HCAHPS efforts. Likewise, we considered a survey administered by CMS with providers including, but not limited to, TEAM participants, physicians, and PAC providers participating in the TEAM. These surveys would provide insight on providers' experience under the model and further information on the care redesign strategies undertaken by health care providers.
In addition, we considered CMS evaluation contractor administered site visits and focus groups with selected TEAM participants, physicians and PAC providers. In the proposed rule we stated that we believe that these qualitative methods would provide contextual information that would help us understand better the dynamics and interactions occurring among the providers in TEAM. For example, these data could help us understand hospitals' intervention plans as well as how they were implemented and what they achieved. In contrast to relying on quantitative methods alone, qualitative approaches would enable us to capture variations in implementation as well as identify factors that are associated with successful interventions and distinguish the effects of multiple interventions that may be occurring within participating providers, such as simultaneous ACO and bundled payment participation.
We considered primary data collection efforts with providers and beneficiaries within the control group. The systematic data collection from a control group would allow us to observe and separate changes in standard care from the TEAM impact. Additionally, primary data collection with beneficiaries who received care from hospitals and providers in the control group would provide critical information about the impact of the model on patient-reported health status, experience of care, and overall satisfaction.
Our evaluation would assess the impact of the TEAM on the aims of improved care quality and efficiency as well as reduced health care costs. This would include assessments of patient experience of care, utilization, outcomes, Medicare expenditures, provider costs, and beneficiary access. Our key evaluation questions would include, but are not limited to, the following:
- Payment. Is there a reduction in Medicare expenditures in absolute terms? By subcategories? Do the TEAM participants reduce or eliminate ( print page 69858) variations in expenditures that are not attributable to differences in health status? If so, how have they accomplished these changes? Did TEAM result in net savings to the Medicare program, after accounting for the financial incentives distributed under the model?
- Utilization. Are there changes in Medicare utilization patterns, overall and for specific types of services? How do these patterns compare to historic patterns, regional variations, and national patterns of care? How are these patterns of changing utilization associated with Medicare payments, patient experience and outcomes, and general clinical judgment of appropriate care?
- Referral Patterns and Market Impact. How has provider behavior in the selected mandatory CBSAs changed under the model? Is there evidence of broader changes to the market? Are provider relationships changing over the course of the model? Is the model facilitating continuity of care by connecting beneficiaries with new or existing primary care providers?
- Outcomes/Quality. Is there either a negative or positive impact on quality of care and/or better patient experiences of care? Did the incidence of relevant clinical outcomes such as complications remain constant or decrease? Were there changes in beneficiary outcomes under the model compared to appropriate comparison groups?
- Equity. Were there notable impacts of TEAM for subgroups based on beneficiary characteristics such as race/ethnicity, dual status, rurality, and other measures of socio-economic disadvantage? How did TEAM participants address health disparities in care? Did the financial performance differ for hospitals furnishing a substantial share of services to uninsured and low-income patients?
- Transformation. Is there evidence that the participants' changes in care delivery, that were made in the response to the model, will be sustained? Did TEAM enable positive spillover effects to other episodes of care, or other providers across the local market of the health system?
- Unintended Consequences. Did TEAM result in any unintended consequences, including adverse selection of patients, access problems, cost shifting beyond the agreed upon episode, evidence of stinting on appropriate care, anti-competitive effects on local health care markets, evidence of inappropriate referrals practices? If so, how, to what extent, and for which beneficiaries or providers?
- Potential for Extrapolation of Results. What was the typical patient case mix in the participating practices and how did this compare to regional and national patient populations? What were the characteristics of participating practices and to what extent were they representative of practices treating Medicare FFS beneficiaries? Was the model more successful in certain types of markets? To what extent would the results be able to be extrapolated to similar markets and/or nationally?
- Explanations for Variations in Impact. What factors are associated with the pattern of results (previously)? Specifically, are they related to:
- Characteristics of the model including variations by year and factors such as presence of downside risk or track assignment?
The TEAM participant's specific features and structure, including such factors as the number of relevant cases, provider mix, and health system affiliation?
- The TEAM participant's organizational culture and readiness
- The TEAM participant's care redesign interventions and their ability to carry out their proposed intervention?
- Characteristics and nature of interaction with partner providers including PAC provider community?
- Characteristics of market and CBSA such as resources, care infrastructure and supply of physicians and associated providers?
- Characteristics associated with the patient populations served?
As discussed in section X.A.3.a.(1) of the preamble of this final rule, TEAM would have a 5-year model performance period. The evaluation period would encompass this entire 5-year model performance period, a year baseline period and up to 2 years after. We plan to evaluate the TEAM on an annual basis. However, we recognize, that interim results are subject to changing policies as discussed in the preamble of this final rule, and issues such as sample size and random fluctuations in practice patterns. Hence, while CMS intends to conduct periodic summaries to offer useful insight during the model test, a final analysis after the end of the 5-year model performance period will be important for ultimately synthesizing and validating results.
We sought comments on our design, evaluation, data collection methods, and research questions.
The following is a summary of comments we received on the proposed evaluation approach for TEAM and our responses to these comments:
Comment: Commenters detailed evaluation topics and subgroups in which they were particularly interested. A couple commenters expressed support for evaluating the impact on equity, especially for historically disadvantaged groups. A commenter stressed the importance of assessing differential impacts for all key questions by sociodemographic factors. A couple of commenters noted model overlap and the ability to distinguish impacts from co-occurring model participation as a critical area to quantify. A commenter highlighted patients with fracture as a key population to assess separately as they are typically more elderly and medically complex.
Response: We thank the commenters for their list of topics and subgroups of interest. These topics and subgroups are in alignment with our stated areas of interest and will be considered in the development and implementation of the evaluation plan.
Comment: A couple of commenters emphasized the importance of the patients' experience and supported the inclusion of beneficiaries and caregivers in data collection to better access patient and family caregivers and/or support persons' experience with this model.
Response: We agree with the commenters' points of emphasis. The inclusion of the patient and caregiver perspective is an important aspect of the evaluation of the impact of the model. The survey of patients is a key component of the evaluation to address the very important issues of patient functional performance, pain reduction, experience, and access.
Comment: A couple of commenters expressed concerns about the anticipated burden associated with the evaluation. A commenter express concern about the costs associated with duplicative and overlapping evaluation efforts. The commenter suggested a concerted effort to limit site visits, data requests, and other reporting requirements to the minimum necessary to understand the impact of the program.
Response: We acknowledge the commenters' concern related to the burden associated with the evaluation and will minimize it to the extent possible while still ensuring a thorough assessment of the model and its impacts. We intend to use the site visit approach for data collection judiciously while being mindful of the impact on providers. Furthermore, while we expect TEAM participants and TEAM collaborators to abide by the terms of ( print page 69859) the model and to cooperate with the evaluation as discussed in section X.A.1.d of the preamble to this final rule, we point out that this final rule does not contain any provisions that the TEAM participants must incorporate the requirement that their TEAM collaborators host site visits in their agreements.
After consideration of the public comments received, we are finalizing the proposed approach to the evaluation without modification.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35934 ), we proposed a voluntary Decarbonization and Resilience Initiative within TEAM to assist hospitals in addressing the threats to the nation's health and its health care system presented by climate change and the effects of hospital carbon emissions on health outcomes, health care costs and quality of care. The voluntary initiative would have two elements: technical assistance for all interested TEAM participants and a proposed voluntary reporting option to capture information related to Scope 1 and Scope 2 emissions as defined by the Greenhouse Gas Protocol (GHGP) framework, [ 1015 ] with the potential to add Scope 3 in future years.
The threats presented by climate change to the health of beneficiaries and to health care operations are growing. These include acute climate-related events (for example, wildfires, high-powered storms, flooding) that can harm exposed populations and disrupt service delivery, exacerbations of chronic illness (for example, extreme heat impacts on cardiovascular and pulmonary health), and increases in water-borne and insect-borne illness. [ 1016 ] These risks often fall disproportionately on traditionally underserved populations, heightening existing health disparities. [ 1017 ] In view of these challenges, health care organizations must increase their resilience, and understand and address their patients' climate-related health risks.
Health systems have reduced their own significant emissions and ground-level air pollution, often through the introduction of energy efficiency solutions, renewable energy initiatives, and focused efficiency measures in clinical care delivery in areas including surgery (described throughout section X.A.3.p. of the preamble of this final rule). We believe these types of cumulative reductions have the potential to make significant contributions to nationwide emission reductions and produce savings. At an individual facility level, these reductions have the potential to save the facility money and enhance their operational resilience (as many sustainable energy solutions can create more energy independence for facilities), meaning decarbonization has bearing on quality of care and cost. More efficient utilization of resources in the surgical setting, specifically, can also reduce cost and improve sustainability; for example, although operating rooms represent a relatively small proportion of hospitals' physical footprint, they typically consume 3-6 times more energy per square foot as the hospital as a whole, [ 1018 ] account for 40-60 percent of the hospital's supply costs, and produce 30 percent of the hospital's waste. [ 1019 ]
Because hospital activities (such as surgical procedures) impact emissions and the work of hospitals requires uninterrupted service delivery, we believe TEAM presents an opportunity for CMS to learn more about key strategies for decarbonization (for example, clinical decarbonization approaches, approaches to reducing low-value services and physical waste) and improving resiliency in the health care system. It is hoped that this initiative would help bring savings to the health system and the Medicare Program, consistent with TEAM's goals.
Climate change driven by greenhouse gas (GHG) emissions threatens patients' health. The health care industry's contribution to those emissions is well-documented and accounts for between 4.4 and 4.6 percent of worldwide GHG emissions. [ 1020 ] In the U.S. in 2018, GHG emissions from the health care sector accounted for 8.5 percent of total U.S. GHG emissions. [ 1021 ] According to the National Climate Assessment, the US Government's official report on climate change impacts, children, older adults, and low-income communities are disproportionately affected by climate change and pollution, meaning the Medicare, Medicaid, and CHIP programs bear much of the medical expenses and caregiving services related to emissions. [ 1022 ] Medicare beneficiaries face several health conditions related to GHG emissions, including, but not limited to, heart disease, stroke, cancer, and respiratory diseases.” [ 1023 ] More discussion on the impact of climate to Medicare, Medicaid, and CHIP beneficiaries is presented in section X.A.3.p.(1).(c).(v). of the preamble of this final rule. The estimated disease burden from U.S. health care pollution is the same order of magnitude as years ( print page 69860) of life lost as a result of deaths from preventable medical errors. [ 1024 ]
In keeping with an increased focus on climate resilience and sustainability across HHS, the Biden Administration in 2021 called for the creation of a new Office of Climate Change and Health Equity (OCCHE) within HHS via executive order ( E.O. 14008 ), and for the first time HHS set an aim for addressing climate-related threats to health in its 2022-2026 strategic plan, requiring all Operating Divisions to contribute. In 2022, the Biden Administration launched the Health Sector Climate Pledge, a voluntary commitment to climate resilience and emissions reduction that invites health sector organizations to align with administration goals, cutting GHG emissions by 50 percent by 2030 and achieving net zero emissions by 2050. A group of 133 organizations representing 900 hospitals have signed the Pledge as of November 16, 2023. [ 1025 ] To support health sector efforts with climate resilience and emissions reduction, OCCHE developed a resource hub, [ 1026 ] featuring tools from across HHS such as a compendium of federal resources for the healthcare sector, information on how to leverage the IRA, an educational webinar series, and the Agency for Healthcare Research and Quality (AHRQ)'s Decarbonization Primer [ 1027 ] (referred to hereafter as the AHRQ primer). OCCHE also convenes federal health systems (for example, Indian Health Service, Veteran's Health Administration) to collaborate on meeting the administration's goals for emissions reduction, which can inform this initiative.
CMS has twice sought and received feedback on approaches to decarbonization and resilience through requests for information in proposed rules. The feedback to these requests was summarized in the final rules. The first request for information was published in the Patient Protection and Affordable Care Act; HHS Notice of Benefit and Payment Parameters for 2023 proposed rule ( 87 FR 693 through 694 ) and a summary presented in the final rule ( 87 FR 27354 ). The second was in the in the FY 2023 IPPS/LTCH PPS proposed rule ( 87 FR 28478 through 28479 ) and the summary was presented in the final rule ( 87 FR 49167 ). Overall, respondents showed a notable interest in reducing health sector emissions and increasing transparent GHG emissions reporting. CMS continues to update policies to promote energy efficiency and reduce GHG emissions. For example, in 2023, CMS issued the Categorical Waiver Health Care Microgrid System. CMS requires specified providers to have “emergency power for an essential electrical system (EES) to be supplied by a generator or battery system.” [ 1028 ] The waiver permits normal and emergency power to be supplied by sources other than a generator or battery system, including a health care microgrid systems that use sustainable sources of energy such as solar power.
When discussing GHG for this initiative, we refer to the Greenhouse Gas Protocol (GHGP) framework, which is a globally recognized standard for quantifying and reporting on emissions. The framework defines 3 scope levels. [ 1029 ] We have included examples that relate to health care. [ 1030 1031 ]
Scope 1: Direct emissions. Direct GHG emissions occur from sources that are owned or controlled by an organization or company. For health care, Scope 1 captures health care operations such as direct facilities emissions, anesthetic gases, and GHG emissions from leased or owned vehicles.
Scope 2: Indirect emissions from purchased energy. GHG emissions from the generation of purchased electricity consumed by the organization or company. For health care facilities, Scope 2 includes purchased or acquired electricity, and steam, heat, or cooling consumed by the reporting organization or company.
Scope 3: Other indirect GHG emissions. Scope 3 allows for the treatment of all other indirect emissions. Scope 3 incorporates upstream and downstream emissions in the supply chain. For health care, Scope 3 may include purchased pharmaceuticals and chemicals, medical devices and supplies, food, water, waste, employee and patient transportation, and additional emissions outside of Scopes 1 and 2. In Scope 3, all purchased and sold goods have an estimated emissions factor for their production, transportation, and life cycle. For example, in a health care setting, Scope 3 emissions may include prescribed medicine such as metered dose inhalers (MDI). Scope 3 uniquely incorporates intangible emissions through the organization's reported investments.
In a 2018 analysis, Scope 1 accounted for 7 percent of the U.S. National Health Care GHG emissions, Scope 2 accounted for 11 percent, and Scope 3 accounted for the remaining 82 percent. [ 1032 ] We ( print page 69861) believe that Scopes 1 and 2 emissions reduction measures represent areas where there are significant opportunities to increase hospital operating efficiency and reduce operating costs. Therefore, we proposed in section X.A.3.p.(4). of the FY 2025 IPPS/LTCH PPS proposed rule, ( 89 FR 35934 ), that TEAM participants could voluntarily respond to organizational questions and Scopes 1 and 2 metrics, as participants in TEAM would have direct oversight of these items. While we did not propose Scope 3 metrics in this rule, we recognize Scope 3 accounts for the largest portion of GHG emissions. Therefore, we sought comment in section X.A.3.p.(4).(a).(vii). of the FY 2025 IPPS/LTCH PPS proposed rule, ( 89 FR 35934 ) on how we might be able to standardize and collect this information in the future.
The CMS Innovation Center is granted discretion to collect data necessary for the purposes of evaluating and monitoring its models under section 1115A(b)(4)(B) of the Act. Overwhelming evidence points to GHG emission's deleterious effect on patient health and the disproportionate impact borne by Medicare, Medicaid, and CHIP beneficiaries. See section X.A.3.p.(1).(c).(v). of the preamble of this final rule, for GHG Emissions Impact on Medicare, Medicaid, and CHIP populations.
Given the established impact GHG emissions have on Medicare, Medicaid, and CHIP beneficiary health, in the FY 2025 IPPS/LTCH PPS proposed rule, ( 89 FR 35934 ), CMS proposed to collect data on GHG emissions, through voluntary reporting, as part of our monitoring and evaluation of the model, just as CMS monitors for other quality indicators that may impact beneficiary health.
Measuring GHG emissions is an important first step toward reducing GHG emissions, and such reductions could lead to outcome quality improvements among beneficiaries. By organizing a GHG emissions reporting system, CMS is supporting TEAM participants in establishing a baseline understanding of their GHG emissions, how much and how efficiently energy is used in their facilities, and the emissions generated by their facilities or activities. Establishing this baseline understanding is a necessary first step to lowering emissions. The proposed decarbonization initiative could directly lead to lower emissions through: (1) sharing benchmarkable data back to TEAM participants, which will support identification of opportunities to improve energy efficiency; (2) supporting their GHG emissions reporting activities, which will support TEAM participants in better understanding their current state energy consumption, GHG emissions, and opportunities to improve energy efficiency; and (3) providing technical assistance related to reporting, identifying, and accessing resources for and undertaking activities to reduce GHG emissions.
Given the association of emissions with chronic diseases, including respiratory and cardiovascular disease, the decarbonization and resilience initiative could improve health outcomes for the Medicare, Medicaid, and CHIP beneficiaries disproportionately affected by GHG emissions. In particular, the Environmental Protection Agency (EPA) released a report on the health impacts of GHG emissions, pollution, and climate change and health and pointed towards key health outcomes that are impacted—new asthma diagnoses in children age 0-17 due to particulate air pollution, premature deaths in adults ages 65 and older due to particulate air pollution, and deaths due to extreme temperatures. [ 1033 ] We would expect reductions in GHG emissions to improve these health outcomes for its patient populations.
In addition to these general health quality impacts, there are also resilience and continuity of care impacts associated with energy efficiency and a transition to sustainable energy sources for hospitals, which also impact quality outcomes. One study that examined 158 hospital evacuations between 2000 and 2017 found that nearly three-quarters were for climate-sensitive events such as wildfires or hurricanes. [ 1034 ] In addition to causing hospital evacuations, climate change can disrupt health care system operations by causing facility damage and closures, power outages, displacement of health care professionals, and disruptions in transportation. These climate impacts affect access to and quality of care.
By sharing back benchmarkable data with TEAM participants (as described in section X.A.3.p.(6).(a)., Individualized Feedback Reports to TEAM Participants, of the preamble of this final rule), providing technical assistance related to GHG emissions reporting, and providing technical assistance to improve energy efficiency and energy resilience, the Decarbonization and Resilience Initiative could directly support TEAM participants in building greater energy resilience to disasters and ensuring greater continuity of care. We expect the Decarbonization and Resilience Initiative to increase the energy efficiency of participating TEAM participants and the degree to which they have sustainable, more localized sources of energy that are resilient to disasters and other climate change related hazards. [ 1035 ] We expect this to lead to fewer hospital closures during disasters and therefore improve continuity of care and other health quality outcomes for effected beneficiaries. Greenwich Hospital offers an example of this. In 2008, the hospital invested in building a low-carbon, energy efficient energy infrastructure with the intention of it being able to withstand the impact of an extreme weather event. The investment proved to be valuable because when Hurricane Sandy hit the New Jersey coast in 2012, the hospital was still able to carry on with normal healthcare operations.
Reductions in operating costs and spending due to energy efficiency and more efficient provision of care (in the case of anesthetic gases) directly contribute to savings for CMS. GHG emissions reporting is a necessary first step for hospitals to begin to understand their emissions, how energy efficient their facilities and processes are, and to identify opportunities to increase efficiencies and lower operating costs and spending tied to GHG emissions and to overutilization of anesthetic gas. In turn, increased energy efficiency and reduced energy expenditures may reduce Medicare Program costs. Technical assistance provided under the initiative would also further help hospitals identify, resource, and implement energy efficiency improvements.
Medicare pays Critical Access Hospitals based on each hospital's reported costs outside of IPPS. Therefore, reductions in operating costs and some capital costs could lead to cost savings for the Medicare program. Medicare pays for capital and operating costs as part of IPPS payments, and efficiencies achieved through decarbonization could lead to savings to the Medicare program. In addition, reporting questions and metrics related to energy use could improve understanding of those costs and inform potential future policy development to secure further savings.
Medicare covers anesthesia services through both Part A and Part B. Research has shown that low-flow anesthesia techniques (≤1 L/min) are associated with lower costs, reduced emissions, and do not impact quality of care or health outcomes. [ 1036 ] The Patient Safety and Support of Positive Experiences with Anesthesia MIPS Value Pathway already includes an efficiency measure focused on encouraging the use of low flow inhalation general anesthesia during the maintenance phase of the anesthetic for patients aged 18 years or older who undergo an elective procedure lasting 30 minutes or longer (ABG44). Such improvements to the provision of care and anesthesia could simultaneously lower emissions and reduce costs/produce savings.
Medicare and Medicaid beneficiary health and program expenditures are directly impacted by GHG emissions. Older adults, or those 65 years old and older, experience poorer health outcomes because of rising temperatures, air pollution, and disaster events. Depending on global trajectories of global warming, particulate matter concentrations are estimated to result in approximately 2,000 to 6,000 premature U.S. deaths for those over 65 years old on an annual basis. Air pollution has other negative health consequences, including the exacerbation of chronic obstructive pulmonary disease and increased occurrence of heart attacks, especially for those living with diabetes or obesity. [ 1037 ] Other studies have documented the impact of weather- related events such as high temperatures, flood, storms, or hurricanes that may disproportionately affect older adults. [ 1038 1039 1040 1041 ]
Medicaid beneficiaries are typically lower-income populations, pregnant people, and children, all of whom experience many direct and indirect health challenges because of climate drivers and events, including greater exposure to air pollution, mortality and injury from extreme temperatures, and food insecurity. [ 1042 ]
Medicare and Medicaid beneficiaries are among the groups most vulnerable to the health effects of climate change and GHG emissions and bear the highest share of climate-sensitive health costs including those from GHG emissions which may account for billions in health-related costs to both programs. [ 1043 1044 ] The Office of Management and Budget's (OMB) 2022 Assessment of the Federal Government's Financial Risks to Climate Change estimates that “Federal climate-related healthcare spending in a few key areas could increase by between $824 million and $22 billion (2020$) by the end of the century.” [ 1045 ]
We proposed at § 512.505 that a Decarbonization and Resilience Initiative is an initiative for TEAM participants that includes technical assistance on decarbonization and a voluntary reporting program where TEAM participants may annually report questions and metrics related to emissions to CMS based on information that we describe in section X.A.3.p.(4). of the preamble of this final rule.
In section X.A.3.p.(4). of the FY 2025 IPPS/LTCH PPS proposed rule, ( 89 FR 35934 ), we proposed that CMS would make available to TEAM participants technical assistance related to decarbonization, emissions reduction, and energy efficiency as described of the preamble of this final rule. The voluntary reporting component of the initiative described in section X.A.3.p.(4). of the preamble of this final rule would allow TEAM participants to ( print page 69863) elect to report metrics including emissions data and assessment questions on four potential categories: organizational questions, building energy metrics, anesthetic gas metrics, and transportation metrics to CMS. As used in this initiative, the terms “metrics” and “measures” refer to data to be collected and reported by TEAM participants. We proposed the building energy metrics would be reported to CMS using the ENERGY STAR Portfolio Manager and all other metrics would be reported to CMS in a manner and form specified by CMS. TEAM participants that elect to report all the metrics after a performance year would receive individualized feedback reports and public recognition from CMS.
We summarize and respond to public comments received on the proposal to establish this initiative below.
Comment: Many commenters, including many providers, expressed strong support for the initiative. Commenters highlighted how the initiative will lessen the impact of climate change on health. Commenters also noted that the initiative would improve patient outcomes, provide cost savings, and help scale sustainable practices. A few commenters stated that the initiative provides helpful resources and incentives to reduce GHG emissions while minimizing the burden of participation. A commenter expressed their belief that this initiative will help signatories to the HHS Health Sector Climate Pledge strengthen their efforts.
Response: We thank commenters for their support. We agree that the initiative would have the potential to improve patient outcomes, may provide cost savings, and would help scale sustainable practices. The purpose of the initiative is to assist TEAM participants with addressing the threats to the nation's health and its health care system presented by climate change and addressing the effects of hospital carbon emissions on health outcomes, health care costs and quality of care. We believe the two elements of the initiative, technical assistance and voluntary reporting, will help achieve this purpose while minimizing burden on facilities.
Comment: Several commenters supported including this initiative in TEAM and recommended additional actions. A few commenters expressed their belief that the initiative is a starting point or first step for future decarbonization efforts, but that more action is necessary to reduce emissions in the health care system. A few commenters requested incentives or penalties be added, stating that these are critical to emission reduction at a systems level. A commenter recommended additional financial incentives to encourage hospital participation. Another commenter recommended tying emission reductions to Medicare payments.
Response: We thank commenters for their recommendations and support of this initiative. We will take commentators' recommendations into account as we continue to build the initiative. We believe that the initiative is an important step in addressing hospital carbon emission reduction. We refer readers to section X.A.3.p.(6).(d). of this final rule for a summary of comments we received regarding financial incentives.
Comment: A few commenters recommended that the initiative should be accelerated, beginning earlier than 2026. A commenter specifically called out the need to accelerate the transition from voluntary to mandatory carbon reporting. Another commenter requested the initiative to be put in place by January 1, 2025.
Response: We appreciate commenters recommendations. We note that we did not propose a transition from voluntary to mandatory reporting, and any changes would be proposed in future notice and comment rulemaking. In regard to timing, as the initiative is part of monitoring efforts for TEAM, this initiative will begin with TEAM's start date on January 1, 2026 ( 89 FR 35934 , at 35935) as stated in section X.A.3.a.(1). of this final rule. CMS will issue sub-regulatory guidance and technical assistance for the voluntary reporting ahead of TEAM's start date as feasible.
Comment: Several commenters noted their support for the initiative and expressed their opinion that it should be opened to all hospitals, not just TEAM participants. A few commenters noted that all hospitals need to participate due to the scale of the GHG emissions problem. Other commenters noted that the initiative, particularly technical assistance, would be of interest to a broader community and should be available for all hospitals and not restricted to TEAM. A commenter noted that this initiative and other CMS environmental sustainability initiatives should include hospital outpatient departments, ambulatory surgical centers, and office-based locations.
Response: We appreciate the commenters' support for this initiative and their feedback regarding this voluntary reporting initiative. Given these comments and other considerations such as the ability to scale reporting across health systems with TEAM participants, we will allow a TEAM participant hospital to voluntarily report its own metrics and respond to questions to CMS and additionally report on metrics and respond to questions to CMS on behalf of the TEAM participant's hospital corporate affiliates. While CMS will provide an individualized feedback report to the TEAM participant, CMS will not provide individualized feedback reports to a TEAM participant's hospital corporate affiliates. If a TEAM participant elects to report on all metrics and responds to questions to CMS, the participant is eligible to receive the badging provided by CMS as described in section X.A.3.p.(6).(b). If a TEAM participant voluntarily opts to report data regarding a corporate affiliate or affiliates, the badging, as described in section X.A.3.p.(6).(b) may include the corporate affiliate. This approach to reporting will allow acute care hospitals selected for TEAM to report on their own emissions as well as report on emissions across one or more of their hospital corporate affiliates within their larger health system, if they wish to do so, rather than needing to limit it to their acute care hospital alone. We agree with commenters that this voluntary reporting and feedback could be helpful to a TEAM participant if that additional voluntary expansion is warranted because the TEAM participant is just one part of a larger health system or other corporate affiliation structure. CMS emphasizes that this expanded reporting option is fully voluntary and at the discretion of the TEAM participant. In addition, in light of these strong supportive comments and given the alignment to CMS goals, CMS may consider expanding this voluntary reporting to additional providers, health care facilities and suppliers in future years.
Comment: Several commenters supported the initiative particularly because it is voluntary and optional. Commenters expressed concern that the potential cost, time, and resources to collect this data may be a barrier for some providers. A few commenters noted that this initiative will help hospitals reduce carbon emissions and improve emissions data reporting. A commenter noted that this initiative should be responsive to individual hospital needs because of the difference between hospitals.
Response: We thank the commenters for their support and understand their concerns that the time, cost, and resources needed to participate in this initiative may be prohibitive for some. As we gain experience with the technical assistance and voluntary reporting aspects of the initiative, we ( print page 69864) plan to share information and learnings to address these concerns. We appreciate commenters' belief that the initiative will help hospitals to reduce carbon emissions and improve data reporting about emissions, and we understand the commenter's concern that it should be responsive to individual hospital's needs. Again, as we gain experience through our technical assistance and voluntary reporting of data, we will additional ways to identify ways to address these needs.
Comment: A few commenters expressed support specifically for including this initiative within TEAM. These commenters noted the importance of climate change and noted that the initiative will benefit the health care sector by helping to improve efficiency and increase cost savings through climate action. Some commenters expressed their belief that the reporting and accompanying technical assistance will lay the groundwork and precedent for other programs.
Response: We appreciate the commenters' support for including this initiative in TEAM and their belief that this initiative may benefit the health sector by improving efficiency, increasing cost savings, and laying the groundwork for other programs.
After reviewing the public comments, for the reasons set forth in this rule, we are modifying § 512.598(a) to not only allow TEAM participants to voluntarily report on metrics and respond to questions to CMS for their acute care hospital but additionally allow TEAM participants to report on metrics and respond to questions that includes the TEAM participant's hospital corporate affiliates. With this modification, we are finalizing the proposal defining this initiative.
For the technical assistance portion of the Decarbonization and Resilience Initiative we proposed that CMS would provide three types of support to interested TEAM participants:
- Developing approaches to enhance organizational sustainability and resilience;
- Transitioning to care delivery methods that result in lower GHG emissions and are clinically equivalent to or better than previous care delivery methods (for example, switching from Desflurane to alternative inhaled anesthetics); and
- Identifying and using tools to measure emissions and associated measurement activities.
We summarize and respond to public comments received on the proposal to provide technical assistance to TEAM participants in this initiative below.
Comment: Several commenters stated that they support our providing technical assistance to TEAM participants. These commenters expressed the need for guidance with the initiative, transitioning care delivery to lower carbon emissions, and understanding the metrics in this area.
Response: We thank the commenters for their support and for identifying areas where our technical assistance may help guide TEAM participants.
Comment: A few commenters noted that geography and hospital characteristics need to be considered in technical assistance materials. A commenter believed that establishing technical assistance cohorts by region or selected core-based statistical area would help to develop regional approaches to address climate change. A commenter expressed concern about whether CMS is knowledgeable enough about the various emissions reduction opportunities that may exist for hospitals based on regional variations and individual hospital characteristics.
Response: We appreciate the comments asking us to take into consideration regional and geographic variations of hospitals as well as the unique characteristics of hospitals when developing our technical assistance for this initiative. As we develop the technical assistance aspect of this initiative, we will take these suggestions regarding geographic, regional, and hospital characteristics into consideration.
Comment: Several commenters believe that CMS should consider partnering with existing learning communities and leverage existing resources from groups such as OCCHE, NAM Climate Collaborative, and the American Climate Corps. A few commenters suggested that CMS should form technical assistance panels or advisory committees to promote learning between TEAM participants and experts. A commenter recommended that CMS align with TJC certification or help with compliance requirements for the TJC program.
Response: We thank the commenters for noting the importance of partnering with organizations that have been working with hospitals to successfully reduce their carbon emissions.
Comment: Several commenters requested that CMS go beyond technical assistance and provide funding for TEAM participants to assess their facility's emissions, set goals, reduce emissions, and successfully implement voluntary reporting.
Response: We thank the commenters for identifying these suggestions.
Comment: A few commenters noted that CMS need to better define what will be included in technical assistance and how that assistance will be distributed to physicians and clinicians within their facilities. They noted that there are numerous sustainability actions that a hospital can take but help would be needed to tailor activities to their particular situation. A few commenters noted that customized or targeted technical assistance is preferred and that CMS should consider providing such technical assistance by establishing technical assistance cohorts, possibly by hospital type or by focusing on some aspect of GHG emission measurements including the basics of GHG measurements.
Response: We thank the commenters for their comments that point out the types of technical assistance needed and how TEAM participants may have different needs depending on their facility's situation and we will take those considerations into account as we develop our technical assistance plan.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal to provide voluntary technical assistance in this initiative.
In the first support type, developing organizational approaches, CMS would offer interested TEAM participants guidance on best practices and methods for identifying opportunities to reduce GHG emissions while promoting sustainability and resilience. Particular attention will be placed on building efficiency and sustainable transportation. We would also help to identify potential non-Medicare financing strategies for this work, noting that TEAM participants have access to tax credits and grant programs that can support decarbonization and climate resilience investments through the Inflation Reduction Act, [ 1046 ] as well as other federal funding opportunities. [ 1047 ] ( print page 69865) OCCHE is leading a Catalytic Program to support safety-net health providers in taking advantage of these unprecedented opportunities; TEAM participants would be encouraged to take advantage of the recorded content and other materials from that program. [ 1048 ]
We summarize and respond to public comments received on the proposal to provide the first type of technical assistance support below.
Comment: Several commenters stressed that technical assistance in the absence of providing some form of direct, upfront funding may have little impact on reducing GHG emissions. A few commenters urged CMS to consider providing funding in the form of loans or grants. A commenter believed that upfront funding as compared to IRA tax credits that are received as reimbursement at a later point in time would be beneficial to TEAM participants. A commenter believed that IRA support for technical assistance regarding low carbon on site generation for the essential electrical system can be obtained only if the project succeeds but not if it fails.
Response: We thank these commenters for their suggestions regarding additional financial support options regarding this voluntary initiative.
Comment: A few commenters stated that some health systems need direct, hands-on technical assistance that will support their sustainability efforts such as tracking emissions, developing emissions reduction goals, navigating Treasury regulations for implementing the IRA, and linking TEAM participants to relevant funding sources. They noted that webinars and websites provide information that often is too general to be helpful.
Response: We thank the commenters who believe that direct, hands-on technical assistance would be most helpful to their health systems. TEAM participants will receive guidance on how to access the ENERGY STAR Portfolio Manager platform to report their facility's emissions and additional technical assistance for the other reporting areas. We will take these comments into consideration as we review which technical assistance strategies to implement.
Comment: A commenter believes that technical assistance that specifically addresses Scope 1 emissions and its potential overlap with other categories is needed.
Response: We thank the commenter for this suggestion. After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal to provide the first type of technical assistance support in this initiative.
With respect to the second type of support transitioning to lower-carbon clinical alternatives, we would offer guidance on strategies for reducing emissions associated with inhaled anesthetic gases in pursuit of improvements on the measures described later in this section (drawing in part on ongoing work by federal health systems in this area). Other types of care delivery transitions could benefit patients by reducing demand for hospital services through education, addressing health inequities, improving telehealth options, and improving upstream care management.
We summarize and respond to public comments received on the proposal to provide the second type of technical assistance support below.
Comment: A few commenters recommended that CMS encourage TEAM participants to transition to clinical practices, therapies, and technologies that emit less carbon. They urged CMS to find a way to build embodied carbon into the metrics of clinical quality. A commenter provided an example in which a clinician has two choices of treatment with similar outcomes on all scales with the exception of embodied carbon. The commenter believed that the lower carbon intervention should be preferred. A commenter noted that the clinical and therapeutic benefits of specific gases such as desflurane and sevoflurane must be weighed against the benefits of reduced emissions when treating patients who are older or who present with certain conditions and asked us to respect the clinician's assessment in determining the safest anesthetic agents for the patient.
Response: We thank these commenters for providing us with these helpful insights and recommendations. We will take these insights and recommendations into consideration as we work to develop our technical assistance strategies to support TEAM participants' efforts to transition to lower carbon clinical alternatives as well as the use of anesthetic gases in section X.A.3.p.(4).(a).(v). of this initiative.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal to provide the second type of technical assistance support in this initiative.
For the third type of support, developing emissions measurement strategies, we would identify relevant measures, existing tools (for example, the ENERGY STAR Portfolio Manager platform described in section X.A.3.p.(4). of the preamble of this final rule) and new tools as needed. We would also offer guidance on strategies for using emissions data to identify opportunities to save energy and reduce emissions (for example, ENERGY STAR Treasure Hunt to identify potential areas to reduce energy usage). [ 1049 ]
We summarize and respond to public comments received on the proposal to provide the third type of technical assistance support below.
Comment: A commenter urged CMS to provide interactive technical assistance in accounting methods, which include data sources and normalization methods, and believed that this interactive technical assistance should be provided directly from an expert oversight body at CMS or TJC, or both. The commenter urged CMS to not rely on third-party vendors to provide these accounting methods as third-party vendors may not have the expertise or may have conflicts of interest.
Response: We thank the commenter for their comment and will take these suggestions into consideration when developing technical assistance regarding emissions measurement strategies.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal to provide the third type of technical assistance support in this initiative.
As proposed in the FY 2025 IPPS/LTCH PPS proposed rule, ( 89 FR 35934 ), this technical assistance would be provided to interested TEAM participants.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal to provide technical assistance to TEAM participants who voluntarily participate in the Decarbonization and Resilience Initiative, including their corporate affiliates, as feasible. ( print page 69866)
For the voluntary reporting portion of the TEAM Decarbonization and Resiliency Initiative, we proposed at § 512.598(a) that TEAM participants may elect to report metrics and questions related to emissions to CMS on an annual basis following each performance year. TEAM participants that elect to report on all the initiative metrics and questions to CMS, in the form and manner required by CMS, would be eligible for benefits such as receiving individualized feedback reports and public recognition as well as potentially achieving operational savings (please note these savings would be incidental and not a result of model-related payments). In section X.A.3.p.(4). of the preamble of this final rule, we proposed the metrics and questions that would be included in the voluntary reporting initiative. In section X.A.3.p.(5). of the preamble of this final rule, we proposed how and when TEAM participants would report the metrics and respond to questions to CMS. Finally, in section X.A.3.p.(6). of the preamble of this final rule, we outline our proposals for benefits for TEAM participants that elect to engage in the voluntary reporting portion of the Decarbonization and Resiliency Initiative as well as document some potential indirect benefits, such as operational savings.
We summarize and respond to public comments received on the proposal for voluntary reporting by TEAM participants that elect to report below.
Comment: Many commenters supported voluntary reporting. Commenters expressed their belief that reporting is an important first step to taking action on climate change. Commenters noted the need to pilot the reporting prior to making it mandatory. Commenters also noted some voluntary reporting is needed because some TEAM participants have limited resources and limited capacity to participate; they expressed concern about the potential burden of collecting emissions data. These commenters further requested that CMS streamline reporting to minimize this burden. Commenters also noted that voluntary reporting will help guide TEAM participants to carbon emission reporting, which may help to reduce emissions, lead to better health outcomes, and set a precedent for other health care institutions.
Response: We appreciate the commenters' support. We intend to streamline the voluntary reporting as much as possible to minimize burden and increase participation. We also believe that technical assistance may provide TEAM participants with the support they may need to participate. We also believe that voluntary reporting is the first step to measure emission reductions and could lead to better health outcomes. In response to commenters' position that voluntary reporting may set a precedent for other health care entities, we agree and may consider other ways to integrate elements of the initiative in future models.
Comment: Several commenters stated their belief that this initiative should be mandatory rather than voluntary. Commenters argued the importance and urgency of curbing GHG emissions and the need to include as many health care facilities as possible. A commenter noted that they understood that the voluntary nature of this initiative is to encourage engagement, at least initially, but urged CMS to move toward some form of mandated participation in less than two years. Another commenter mentioned that starting in 2026 is too slow. A commenter recommended mandatory participation so that CMS could eventually modify the Composite Quality Score based on performance on this initiative.
Response: We appreciate the commenters' urgency on curbing GHG emissions; however, we believe that voluntary reporting, with technical assistance, is an important first step to understanding and curbing health care emissions. In regard to timing, as the initiative is part of monitoring efforts for TEAM, we cannot start this initiative until TEAM begins on January 1, 2026 ( 89 FR 35934 , at 35935) as stated in section X.A.3.a.(1). of the final rule. Similarly, we cannot at this time indicate if or and when we may propose to make this initiative to be mandatory.
Comment: A few commenters supported additional financial incentives to encourage TEAM participants to take part in the initiative. Some commenters noted this would be needed if the initiative ever became mandatory. A commenter noted the need for funding for TEAM participants to talk to patients about health and climate.
Response: We are not proposing to include financial incentives for the voluntary initiative. We refer readers to section X.A.3.p.(6).(d). of this final rule for more information on comments we received on integrating financial incentives in the future. We also agree that providers talking to patients about health and climate may be helpful and will consider whether that would be appropriate for technical assistance.
Comment: Several commenters believed that reporting on metrics was not sufficient. A few commenters believed the focus needs to move to setting goals and meeting outcomes rather than just reporting to meet national emission reduction goals. A commenter stated their belief that TEAM participants should also share raw data and measure performance with their sustainability teams, anesthesiologists, and other stakeholders first. Another commenter noted the need for health care clinicians, particularly those located in health centers in areas with high environmental injustice, to be able to talk to patients about the connection between climate and health. A commenter requested that CMS explore the feasibility of emission reduction benchmarks so that TEAM participants have a common target.
Response: We believe that this initiative's voluntary reporting, along with technical assistance, is a critical first step for TEAM participants to understand their GHG emissions to achieve the desired outcomes. Data is needed to understand current GHG emissions and to establish the desired outcomes of reduced GHG emissions. We believe this phased approach within this initiative will enable us to better understand opportunities for improvement, the impact on health, and potentially establish future benchmarks. We do encourage TEAM participants to work with and share data with their sustainability committees and clinicians in order to make progress on climate objectives, but we are not requiring that as part of this model. Similarly, we believe there may be value in health centers talking to their patients about the connection between climate and health but want to limit the first iteration of this initiative to selected metrics and assessment questions. We may consider whether examples of data sharing and discussion with patients would be appropriate for technical assistance.
Comment: A few commenters recommended that the metrics in this initiative be measured with verifiable data. Some commenters additionally asked that TEAM participants publicly disclose their GHG emissions measurements. A commenter recommended that CMS develop a process to ensure accuracy, either through auditing or third-party verification of data reports, especially if performance will become tied to bonus payments. Another commenter recommended CMS create a template for health care organizations to report their ( print page 69867) results so the reports are consistent across organizations.
Response: We agree it is important to verify the data that is reported through this initiative. As discussed in more detail in sections X.A.3.p.(4).(a).(iv). through X.A.3.p.(4).(a).(vi). of this final rule, we are anchoring our data collection on tools like ENERGY STAR Portfolio Manager and on verified documented records such as purchase records and administrative billing. We did not propose that TEAM participants must publicly disclose their GHG emission measurements, as we believe that could deter participation. We do intend to assess the metric data for validity, but because we are trying to minimize burden, we therefore did not propose to conduct audits or third-party validations. We may revisit whether audits or other validations are needed in the future. Finally, we do intend to create reporting templates for the information not reported through ENERGY STAR Portfolio Manager and will issue this through sub-regulatory guidance.
Comment: A few commenters expressed additional benefits from collecting GHG data. Some commenters expressed their belief that this initiative can establish benchmarks and help identify areas where emissions levels are undermining progress on health outcomes. Commenters also noted that data can help identify trends and provide opportunities for new analysis and strategies for positive changes. A few commenters noted that this initiative may create efficiency through standards that help reduce waste and facilitate more robust GHG emission assessments.
Response: We thank the commenters for their support. We agree there are many potential benefits from collecting GHG emissions. We anticipate developing benchmarks for the participant feedback reports. We will also assess if there are trends that are particularly relevant for TEAM participants. Finally, as discussed in section X.A.3.p.(6). of this final rule, we do agree there may be some operational benefits to TEAM participants from operational efficiencies and waste reduction.
Comment: A commenter recommended making the reporting option of this initiative available to the entire country and not limited to TEAM participants.
Response: We appreciate the commenter's request, but the voluntary reporting of the initiative is part of the monitoring of TEAM and not a separate model that has separate participants. In the future, we may explore adding similar initiatives to monitoring of other models.
Comment: A few commenters did not support disclosing carbon information, citing their belief that there is no clear evidence that public disclosure will result in the healthcare industry decarbonizing. A commenter noted that CMS does not reimburse differently regardless of a provider's carbon footprint. Another commenter believed that disclosure may prove regressive or counterproductive and may conflate improvements with reporting with actual GHG emissions reductions with improvements, and that published reports may be satisfying to investors. A commenter suggested that requiring a reduction or elimination of industry GHG emissions by regulation is far more effective, more durable and more than just this initiative.
Response: We recognize that reporting on GHG emissions and other assessment questions is not the same as reducing GHG emissions, but we think it is an important first step. As discussed in section X.A.3.p.(6).(d)., we included an RFI on potential ways we might integrate the initiative into future TEAM financial incentives. We emphasize that reporting in the initiative is voluntary and that we are not requesting or requiring TEAM participants to publicly disclose their information, and as discussed in section X.A.3.p.(6).(b). of this final rule, we intend to be clear on what the participation badge includes. As for the commenter who suggested we regulate the reduction or elimination of GHG emissions in the health care industry, we believe that would be beyond the scope of TEAM.
After consideration of public comments, we are finalizing the voluntary reporting proposal at § 512.598(a) and note that if TEAM participants elect to report metrics and questions related to emissions to CMS on an annual basis, they must report the information to CMS no later than 120 days in the year following each performance year, or a later date specified by CMS.
As discussed in section X.A.3.p.(1). of the preamble of this final rule, the GHGP establishes a framework for measuring Scope 1 and Scope 2 emissions. In identifying priority Scope 1 and Scope 2 categories and metrics for emissions reporting for TEAM participants, we considered guidance and research from several sources. In 2022, AHRQ convened an expert panel to develop a primer for identifying, prioritizing, monitoring, and reducing health care carbon emissions. In developing our proposals, we referred to this AHRQ primer to identify potential measures for Scopes 1 and 2. We also looked at guideline sources, such as: the new Sustainable Healthcare Certification requirements by TJC, for their elements on leadership, measurement, and performance improvement; and guidance from the National Academy of Medicine (NAM) for steps and key actions to reduce GHG emission within health care systems.
The AHRQ primer identified three categories that fit into Scopes 1 and 2: building energy, anesthetic gases, and transportation. NAM published key actions that facilities could take to address greenhouse gas emissions. [ 1050 ] These actions are broken into two steps. Step I focuses on actions to start a decarbonization journey and includes activities like assembling an executive sustainability team, performing a GHG inventory, and establishing specific decarbonization goals. Step II actions, which focus on specific interventions, include activities for reducing emissions from building energy, anesthetic gas, and transportation. TJC launched a Sustainable Healthcare Certification program that includes required standards for organizational performance and leadership, such as a sustainability plan, as well as requirements for collection of detailed emissions information for at least 3 different sources out of six—energy use (fuel combustion), purchased electricity (purchased grid electricity, district steam, chilled and hot water), anesthetic gas use (including volatile agents and nitrous oxide), pressurized metered-dose inhaler use), fleet vehicle carbon-based fuel use (from organization-owned vehicles), and waste disposal.
At this time, we proposed to limit metrics that TEAM participants may voluntarily report for the Decarbonization and Resilience Initiative to Scope 1 (direct emissions ( print page 69868) related to health care operations) and Scope 2 (emissions related to purchased electricity consumption). We believe that TEAM participants have more ability to track and report these metrics at this time and could use information from these metrics to assess ways to reduce their carbon emissions and improve their operating efficiency. TEAM participants would be encouraged to look at emissions across all three Scopes, but for this initial program, the proposed metrics would include Scopes 1 and 2. We sought comment on our proposal to limit the focus of Decarbonization and Resilience Initiative to Scopes 1 and 2 for the initial years of TEAM.
Based on programs and publications discussed in section X.A.3.p.(4).(a).(i). of the preamble of this final rule, we proposed four areas for reporting: (1) Organizational Questions; (2) Building Energy Metrics; (3) Anesthetic Gas Metrics; and (4) Transportation Metrics. We proposed at § 512.598(a) the metrics for the voluntary program. TEAM participants, if they so choose, would report on these four categories. In proposing these voluntary questions and areas for voluntary metric reporting, CMS is prioritizing alignment with existing initiatives such as those described in section X.A.3.p.(4).(a).(i). of the preamble of this final rule.
We summarize and respond to public comments received on the proposal regarding the background on scope and metrics sources below.
Comment: Several commenters supported using the GHGP and limiting the focus to Scopes 1 and 2 for the initial years of TEAM. A commenter noted the proposal had the right balance between administrative burden and highest potential for impact and has clear definitions. The commenter further noted that limiting to Scopes 1 and 2 would increase uptake of this initiative. A commenter recommended requiring all health facilities to measure and achieve reductions fully across Scopes 1 and 2 including energy use (Scope 1-Stationary Combustion, Mobile Fleet Combustion), purchased electricity (Scope 2 purchased grid electricity and district steam, chilled and hot water), fleet (based on fuel use) (Scope 1- Mobile Fuel Combustion), and waste anesthetic gas use (Scope 1-Fugitive Emissions). The commenter expressed the belief that these categories reflect those activities that many health facilities already track (for example, energy consumption) and/or are easy to quantify (for example., waste anesthetic gases from surgical procedures) and there are reasonable interventions for reducing these emissions in Scopes 1 and 2, and often, those interventions bring cost savings for health facilities.
Response: We thank the commenters for their support. We agree that starting with Scope 1 and 2 would have impact and be feasible for most TEAM participants to report.
Comment: Many commenters believed this initiative proposal did not go far enough because it did not include Scope 3 emissions which contribute significantly to the total emissions. These commenters recommended certain Scope 3 metrics they believed were attainable for TEAM participants to collect, including normalizing patient encounters when it came to reusables vs single use devices, collecting waste volumes, patient and employee travel by mileage, and food waste. A few commenters believed that Scope 3 data collection should also be mandatory.
Response: We understand Scope 3 accounts for a large portion of total healthcare emissions; however, we believe we should start with Scope 1 and Scope 2 for the initial years because we believe it is more feasible to collect and we want to encourage participation. We may add Scope 3 in the future. For additional comments related to Scope 3 metrics, please refer to section X.A.3.p.(4).(a).(vii)(A). of this final rule.
Comment: A commenter noted specifics related to the metrics under Scope 1 and Scope 2. A commenter suggested that CMS should not attempt to do all of Scope 1 and 2 emissions, and that there is some confusion in the industry around those boundaries. The commenter specifically noted that Scope 1 refers only to the Kyoto protocol gases, and not to all GHGs. Thus, some gases (such as desflurane) are not exactly Scope 1, even though they are directly emitted by the organization, and there are other sources of Scope 1 emissions (for example, refrigerants) that are not captured by ENERGY STAR. The commenter thus noted that the proposed measures are measuring some Scope 1 emissions and some other direct emissions. The commenter did support the measures that were proposed.
Response: We thank the commenter for the clarification. The gases listed in the GHGP include carbon dioxide (CO2), methane (CH4), nitrous oxide (N2O), hydrofluorocarbons (HFCs), perfluorocarbons (PFCs), sulphur hexafluoride (SF6) and nitrogen trifluoride (NF3). [ 1051 ] However, additional healthcare related documents related to emissions, including both the AHRQ Primer and the NAM, consider both desflurane and similar gases in their discussion. Therefore, we intend to measure these gases in this initiative. Also, we do not intend to capture all of Scope 1 and Scope 2, we are just collecting quantitative information on building energy, anesthetic gases, and transportation metrics (as discussed in section X.A.3.p.(4).(a).(vii).(a). of this final rule). We intend to update metrics as ENERGY STAR metrics evolve and are updated. We proposed at § 512.598(a)(2)(i) that these proposed building energy metrics would be based on the ENERGY STAR Portfolio Manager guidelines for the time of submission.
Comment: A commenter agreed with the need for normalization factors for comparing facilities. The commenter recommended CMS consider emission normalization factors related to patient outcomes, such as anesthetic hours (for volatile and nitrous oxide emissions), bed-days for inpatient care facility emissions, and potentially adjusted patient-days for total emissions.
Response: We thank the commenter for their suggestions. We agree on the importance of normalization factors and discuss that in the anesthetic gas metric and transportation metric portion of this final rule.
Comment: A commenter recommended that CMS align this initiative with other voluntary and mandatory initiatives, such as the Joint Commission Sustainable Healthcare Certification and the Department of Health and Human Services Health Sector Climate Pledge, mandatory state laws (for example, California) to avoid duplicative efforts.
Response: Our intention is to align with existing programs where possible when it comes to data collection and general questions. For the initiative, we considered questions from the Department of Health and Human Services Health Sector Climate Pledge and the Joint Commission Sustainable Healthcare Certification.
Comment: A commenter recommended CMS consider opportunities to improve reporting accuracy before focusing on opportunities to increase the number of TEAM participants voluntarily reporting information to CMS on Scope 1 and 2 metrics. The commenter noted they have seen significant variation in how this information is interpreted.
Response: We agree that reporting accuracy is important for the initiative. We are finalizing quantitative metrics which can be derived from purchase records, administrative billing, or other quantifiable data. We do intend to check submissions for data integrity and completeness. ( print page 69869)
Comment: A few commenters asked for clarity on the scope of the data collected. A commenter recommended reporting Scope 1 and 2 metrics at the facility level, ideally using a health care organization's HCO ID number (for example, “HCO ID #1” with two hospital facilities within the same campus constitutes the boundary). This would make the reporting consistent with TJC's approach in their Sustainable Healthcare Certification (SHC) program as well as with ENERGY STAR Portfolio Manager. Another commenter recommended that CMS broaden its approach to measures to include hospital outpatient departments, ambulatory surgical centers, and office-based locations. The commenter noted that an increasing number of procedures are conducted in nonoperating room anesthetizing locations as well as outpatient settings.
Response: For TEAM participants that elect to report, we would require the quantitative building energy, anesthesia, and transportation metrics at the inpatient facility level (using the identifier selected for TEAM) in a manner that is consistent with ENERGY STAR Portfolio Manager. We will provide an option, however, for TEAM participants to additionally submit information for their hospital corporate affiliates. We also intend to publish technical guidance which will provide more details on how the information may be submitted.
Comment: A few commenters recommended that ENERGY STAR Portfolio Manager should be the only benchmarking and reporting tool, data repository, calculator tool, feedback, and support center for all proposed metrics in this initiative. A commenter stated that centralization at ENERGY STAR will make it easier to integrate Scope 3 reporting more quickly and to identify with best practices. Another commenter stated their belief that ENERGY STAR Portfolio Manager will avert greenwashing of data.
Response: We appreciate the commenter feedback. However, ENERGY STAR Portfolio Manager does not currently collect information on anesthetic gas and transportation. We may revisit using ENERGY STAR Portfolio Manager in the future if CMS would like to collect, and TEAM participants would like to submit, a larger scope of information.
After reviewing public comments, for the reasons set forth in this rule, we are finalizing the proposal to base our metrics on Scope 1 and Scope 2 of the GHGP with the acknowledgement that some of the anesthetic gases we are discussing are technically not part of the GHGP but are important when reviewing emissions from anesthesia.
For the Decarbonization and Resilience Initiative, we proposed at § 512.598(a)(1) a set of organizational questions about the TEAM participants' sustainability team and sustainability activities. These questions are generally based on NAM's key action Step I shortlist. We proposed the organizational questions would include the following:
- Does your facility have a sustainability team? If so, does your facility's sustainability team include broad representation, seeking input across operational and clinical lines, and engaging staff, executive leaders, clinicians, board members, and patients?
- Does your facility perform a GHG inventory? If so, which of the following are included in your facility's GHG inventory:
++ Scope 1 emissions.
++ Scope 2 emissions.
++ Scope 3 emissions (business travel, employee commuting, waste)?
- Has your facility implemented a decarbonization goal that compares performance to a baseline year?
- What are your facility's decarbonization goals (for example, 10 percent GHG reduction annually across all operations, aiming to achieve 50 percent reduction by 2030)? What is the baseline year used to measure your facility's decarbonization success?
- Has your facility implemented a decarbonization plan?
- What is your facility's implementation plan? What milestones and deliverables to track progress are you documenting?
- Has your facility designated resources for decarbonization and resilience initiatives?
- Does your facility track operation room (OR) specific energy use or waste? If so, what, if any, OR energy efficiency or waste reduction initiatives have you implemented?
We anticipate these questions would be relatively straightforward to report on and provide a helpful baseline to inform technical assistance. We sought feedback on the potential burden of adding overall organizational questions to the Decarbonization and Resilience Initiative.
We summarize and respond to public comments received on the proposal to provide a set of organizational questions at § 512.598(a)(1) of the final rule later in this section.
Comment: A few commenters recommended adding additional questions to the organizational question section that address the TEAM participants' sustainability team and activities, including questions about the healthcare system's current pledges, commitments, and certifications, specific questions about how GHG emissions are calculated and reported, large medical equipment energy use, decarbonization challenges, linkages between compensation and achieving decarbonization goals, and adding open ended questions. A commenter noted that identifying current activities of the TEAM participants sustainability team members takes time and helps hospitals understand expectation for local implementation efforts.
Response: We thank commenters for their recommendations. CMS is committed to helping organizations accurately assess their current level of decarbonization efforts. We will finalize organizational questions but may revise the set of questions based on this feedback.
Comment: A few commenters recommended requiring a climate resilience plan from TEAM participants to align with the HHS Health Sector Climate Pledge and to ensure continued ability to provide health care services during and after extreme weather events.
Response: We thank commenters for their recommendations and may take this suggestion under consideration.
Comment: A few commenters supported asking organizational questions from TEAM participants and confirmed that these questions would not be burdensome.
Response: We thank commenters for their support and their feedback on the level of burden for these questions.
Comment: A commenter recommended that TEAM participants complete these organizational questions on an annual basis since they are general in nature.
Response: We thank the commenter for the recommendation and confirm that the organizational questions are submitted on an annual basis, as referenced in section X.A.3.p.(4).(a).(iii).
Comment: A commenter recommended identifying questions that contain a set of smaller attainable goals to receive buy-in across the hospital facility.
Response: We thank the commenter for their recommendation and believe the organizational questions are an appropriate start on a decarbonization journey as they focus on identification and assembly of a team, conducting an inventory of GHG emissions, and developing specific decarbonization goals and plans. ( print page 69870)
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing with modifications at § 512.598(a)(1) the set of organizational questions about the TEAM participants' sustainability team and sustainability activities to allow us to collect and analyze the responses in a more structured way and to allow us to make comparisons across TEAM participants.
For building energy usage, we proposed metrics that would assess both the raw GHG emissions (location-based and market-based methods of calculation) from energy use (direct and indirect), source information, and information to normalize these metrics. Specifically, we proposed at § 512.598(a)(2) a set of building energy metrics related to measuring and reporting GHG emissions related to energy use at TEAM participant facilities. We proposed at § 512.598(a)(2)(i) that these proposed building energy metrics would be based on the ENERGY STAR Portfolio Manager guidelines for the time of submission and that TEAM participants choosing to report these metrics must submit using ENERGY STAR Portfolio Manager according to the reporting and timing requirements proposed in section X.A.3.p.(5). of the preamble of this final rule. We proposed to adopt the ENERGY STAR Portfolio Manager guidelines at the time of submission to ensure that the metrics collected are consistent with current standards.
For the Decarbonization and Resilience initiative, we proposed at § 512.598(a)(2)(ii) the following metrics: ENERGY STAR Score for Hospitals, as well as the supporting data that goes into that calculation, and energy costs and basic energy consumption metrics such as total, direct, and indirect GHG emissions and emissions intensity as specified in the ENERGY STAR Portfolio Manager. [ 1052 ] As of the publication of the FY 2025 IPPS proposed rule, the most recent ENERGY STAR Score for Hospitals methodology was published in February 2021 [ 1053 ] and requires information such as energy use intensity, electricity, natural gas, and other source emissions usage and several normalizing factors such as building size, number of full-time equivalent workers, number of staffed beds, number of magnetic resonance imaging (MRI) machines, and zip code (to pull weather and climate related data such as the number of heating and cooling days). [ 1054 ] We proposed that this supporting data would be reported to CMS, as well. Having both the aggregate score and the underlying details would provide CMS additional detail to monitor the impact of emissions. As described in section X.A.3.p.(5). of the preamble of this final rule, TEAM participants who elect to report data would submit after the performance year. Should the ENERGY STAR Score for Hospitals method change, we would default to the methods that ENERGY STAR is using at the time of submission so that the data reported to CMS would be consistent with ENERGY STAR Score for Hospitals.
ENERGY STAR Portfolio Manager also allows users to track GHG emissions and energy costs, which captures total energy cost and can inform tracking of potential savings.
There are several reasons we proposed that TEAM participants use the ENERGY STAR Portfolio Manager for submitting building energy metrics. First, ENERGY STAR Portfolio Manager is a free, on-line benchmarking tool used by over 3,000 hospitals as of January 2024 (approximately half of the number of U.S. hospitals [ 1055 ] ) to benchmark energy, water, and waste. Approximately forty-seven state and local governments [ 1056 ] require its use to track and report energy usage and emissions on an annual basis. We believe that by using data and information collected in the ENERGY STAR Portfolio Manager tool, we would minimize the reporting burden for TEAM participants and maximize the benchmarking value of reporting, which should make comparisons and measuring progress easier. We also believe the information collected in the ENERGY STAR Score for Hospitals are similar to recommended measures in the AHRQ primer.
Finally, we also considered an alternative where we instead allowed private vendors with a relationship to the facility to submit equivalent information, aligned to the GHG Protocol, instead of ENERGY STAR Portfolio Manager. Ideally, we would like TEAM participants to have options to collect and capture their emissions data, but we also want to ensure that any benchmarks are consistent across TEAM participants.
We sought feedback on our proposed metrics reported through ENERGY STAR Portfolio Manager and on the alternative of allowing private vendors to submit equivalent information.
We summarize and respond to public comments received on the proposal to use building energy metrics related to measuring and reporting GHG emissions regarding energy use at TEAM participant facilities at § 512.598(a)(2) of the final rule below.
Comment: Several commenters supported the use of ENERGY STAR Portfolio Manager for the building energy metrics because it is the standard reporting structure health systems use to measure and track energy consumption and GHG emissions, was developed by the Environmental Protection Agency, limits reporting burden for providers, and is already used by the Department of Health and Human Services.
Response: We thank commenters for their support. We agree that the use of ENERGY STAR for the building energy metrics will limit reporting burden on providers.
Comment: A few commenters recommended that CMS use data tracking in ENERGY STAR Portfolio Manager and not require additional reporting of the building energy metrics to minimize reporting burden. A commenter recommended that CMS ensure easy integration of ENERGY STAR data for TEAM participants that participate in this initiative in order to minimize reporting burden.
Response: We thank commenters for their recommendations regarding the use of ENERGY STAR Portfolio Manager for our building energy metrics. The building energy metrics we finalize will be collected through ENERGY STAR Portfolio Manager and TEAM participants will be able to submit their information through that platform.
Comment: A commenter provided an alternative tool to ENERGY STAR Portfolio Manager. The commenter ( print page 69871) noted that Health Care Without Harm and Practice Greenhealth's Health Care Emissions Impact Calculator is a publicly available tool that allows facilities to track their GHG emissions across Scope 1 and 2.
Response: We thank commenters for their suggested alternative. At this time, based on public feedback, we are finalizing our proposal to report building energy metrics through ENERGY STAR Portfolio Manager.
Comment: A few commenters did not support the alternative of allowing private vendors to submit equivalent information to ENERGY STAR Portfolio Manager. In not supporting this alternative, commenters stated their belief that a single reporting structure is critical to the success of a program; that the ENERGY STAR Portfolio Manager is the standard tool used to benchmark energy, water, and waste; and by only having one reporting structure, it will reduce reporting burden; and that allowing private vendors to submit data may create adverse future effects.
Response: We thank commenters for sharing their concerns on the alternative of allowing private vendors to submit equivalent information to ENERGY STAR Portfolio Manager. We agree the ENERGY STAR Portfolio Manager is the standard, but we wanted to recognize that some TEAM participants may use third party vendors for emissions reporting. However, based on public feedback, at this time, we are not finalizing the alternative of allowing private vendors to submit equivalent information to ENERGY STAR Portfolio Manager.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing our proposed building energy metrics reported through ENERGY STAR Portfolio Manager. We are not finalizing our alternative proposal of allowing private vendors to submit equivalent information.
We believe anesthetic gas metrics are important to collect for the TEAM Decarbonization and Resilience Initiative because the TEAM's proposed initial performance focus is on surgical procedures which regularly utilize anesthetic gas, as discussed previously. We proposed at § 512.598(a)(3) a set of metrics related to measuring and managing emissions from anesthetic gas. These metrics include total GHG emissions from inhaled anesthetic gases (based on purchase records) along with the associated normalization factors, and additional assessment questions.
We evaluated methods to consistently capture anesthesia metrics. ENERGY STAR Portfolio Manager currently does not collect information or calculate benchmarks on anesthetic gases. Anesthetic gases may be collected through an optional field through ENERGY STAR Portfolio Manager, but we acknowledge that this is not a standardized field. We intend to update metrics as ENERGY STAR metrics evolve and are updated, as we proposed at § 512.598(a)(2)(i) that these proposed building energy metrics would be based on the ENERGY STAR Portfolio Manager guidelines for the time of submission. There are other calculators, such as Practice Greenhealth's® Health Care Emissions Impact Calculator that collect and calculate data related to anesthetic metrics, [ 1057 ] but we were concerned that using multiple tools to report metrics (considering we are already proposing to use ENERGY STAR Portfolio Manager for the building energy metrics) would increase reporting complexity and reporting burden. The AHRQ primer recommended total GHG emissions from inhaled anesthetics and mean gas flow rates, but we were concerned on the feasibility of capturing mean gas flow rates. Based on all these factors, we are therefore proposing at § 512.598(a)(3)(i) to include a metric for total GHG emissions from inhaled anesthetics using purchase records. The metric would include information such as volume of the bottle, the number of bottles, and/or the number of pounds, depending on the anesthetic gas. [ 1058 ] We believe purchase records provide a proxy for actual utilization and that purchase records may be easier for TEAM participants to report compared to actual usage which generally would have to be extracted from electronic health records. Also, we proposed at § 512.598(b)(3)(ii) normalization factors which we proposed to be anesthetic hours so we could more accurately compare the carbon impact across different facilities. We believe these metrics would provide information on anesthetic gases which would be most relevant to the episodes and provide a means for which to create anesthetic gas metric benchmarks.
At § 512.598(a)(3)(iii), we also proposed to include assessment questions broadly based on the key actions recommended by NAM Step II for reducing emissions from anesthetic gases that TEAM participants may choose to answer. Assessment questions include the following:
- Has your facility set an emissions reduction goal related to anesthetic gases? If so, what is that goal?
- Does your facility track and benchmark anesthetic gas emissions at the procedure and provider level?
- Has your facility removed the use of desflurane or removed vaporizers when using desflurane?
- Has your facility decommissioned piped nitrous oxide and substituted e-cylinders? If not, are these activities in process?
We believe answering these assessment questions would provide facilities with ideas and actions that could in turn reduce impact on emissions and would supplement the other anesthesia gases data.
We sought comment on our proposed anesthesia gas metrics which would include the total GHG emissions from inhaled anesthetics and anesthetic hours and assessment questions for anesthetic gases. We particularly sought feedback on the feasibility of reporting data based on purchase records or whether we should require actual records. We also sought comment on the feasibility of capturing anesthetic hours or if we should consider a different normalization factor such as number of operating rooms. We also sought feedback on whether we should consider other calculators, metrics and inputs to determine GHG emissions from anesthetic gases, or quality measures such as ABG44: Low Flow Inhalational General Anesthesia.
Finally, while we believe it is important to capture the data on total GHG emissions from inhaled anesthetics, anesthetic hours, and the assessment questions for anesthetic gases, we also considered whether we provide the TEAM participants an option of reporting either the total GHG emissions from inhaled anesthetics (with anesthetic hours) or reporting the assessment questions for the voluntary reporting program. We believe this flexibility for TEAM participants could reduce reporting burden and enhance participation, but we are concerned this alternative may not provide comparable data across the TEAM participants who voluntarily submit data. We sought feedback on this alternative for TEAM participants who choose to submit to report either anesthetic gases and anesthetic hours or to report the assessment questions.
We summarize and respond to public comments received on the proposal to ( print page 69872) use a set of metrics related to measuring and managing emissions from anesthetic gas at § 512.598(a)(3) below.
Comment: Several commenters supported collecting information on anesthetic gases using purchase records noting that purchase records have less administrative burden than actual records and it would provide needed data to build benchmarks.
Response: We thank commenters for their support. We agree that purchase records seem to be the most efficient way to collect anesthetic gas metrics and are finalizing that proposal. We do intend to evaluate potential benchmarks for the participant feedback reports.
Comment: A commenter recommended CMS pilot test collection metrics before implementing on a large scale to ensure that data elements can be captured uniformly.
Response: We agree on the importance of uniform data capture. We believe that rolling this initiative out in a voluntary manner will provide an opportunity to assess the comparability of the data captured.
Comment: A few commenters supported additional metrics for consideration. A commenter supports ABG44: Low Flow Inhalational General Anesthesia for the anesthesia Merit-based Incentive Payment Systems (MIPS) Value Pathway (MVP)—especially since many anesthesia groups do not have access to their hospital records or electronic health records. ABG44 is used to encourage anesthesia groups to track their gas flows. Another commenter recommended an outcome metric and performance metrics. The outcome metric is total anesthesia-related GHG emissions, normalized anesthesia-related GHG emissions (kgCO2e/hour). The performance metrics are (1) agent selection—percent of clinical use for each anesthetic agent (sevoflurane, isoflurane, desflurane) which could be assessed across an entire practice, or per individual clinician and (2) efficiency of use—fresh gas flow assessment (mean vs. median fresh gas flow (FGF) per group practice or individual clinician.
Response: We thank the commenters for their feedback. For the first years of this voluntary initiative, we are not finalizing additional metrics for collection. We note that ABG44: Low Flow Inhalational General Anesthesia is still reportable for clinicians via the MIPS. We do believe we could assess the percent of agent selection with the information we are collecting from TEAM participants, but we are not asking for this information at the clinician level. We will review areas where our reporting tool may already calculate normalized anesthesia-related GHG emissions. Finally, we are not proposing to capture fresh gas flow assessment because that information is not always available through purchase records or administrative claims data, although we may consider adding a question related to fresh gas flow to the assessment questions.
Comment: A few commenters strongly encouraged CMS to separate the assessment of volatile anesthetics (the “fluranes”: desflurane, sevoflurane, isoflurane) from nitrous oxide, noting the gases are clinically different, operationally different, and with different emission mitigation solutions. Commenters noted that anesthesia hours may be an appropriate normalizer for emissions are generated through clinical activity (measured in time). For nitrous oxide, much of the emissions come from central line systems. A few commenters recommended using operating rooms as a normalizing factor for nitrous oxide. A commenter requested CMS collect both the purchased information and utilization information for nitrous oxide noting that comparing these two numbers will accelerate the mitigation of fugitive nitrous oxide emissions by transitioning from central medical gas line distribution to decentralized distribution via localized E-tanks. A commenter noted that for areas where nitrous oxide is used outside of anesthesia care the normalizing factors would need to be numbers of patients receiving nitrous oxide analgesia because detailed quantitative data is not regularly captured in an EHR.
Response: We appreciate the feedback. We intend to look at nitrous oxide separately from the volatile anesthetics and as discussed below, we will be adding operating rooms onto our quantitative metrics to assess it as a potential normalizing factor for nitrous oxide. In section X.A.3.p.(4).(a).(vi). we are finalizing our proposal to capture patient encounters, so we will already have that data as a potential normalizing factor. We have heard other commenters that capturing actual utilization from an EHR may be cumbersome for some TEAM participants, so we are not requiring actual utilization for nitrous oxide at this point, but we could revisit that requirement in the future.
Comment: A few commenters supported the use of anesthetic hours to normalize the anesthetic metrics. Commenters noted while this information may not be exactly precise, it can be pulled from billing data and does not require a complicated extract from an electronic health record. A few commenters noted that anesthetic hours are appropriate for volatile gases and not for nitrous oxide. A commenter said anesthetic hours was better than operation rooms because many services occur outside of the operating room.
Response: We thank commenters for their support. We are finalizing anesthetic hours as one potential normalizing factor. As discussed elsewhere in this section, we are also considering other potential normalizing factors.
Comment: A few commenters did not support using anesthetic hours or suggested alternatives to anesthetic hours for normalizing anesthetic gases. A commenter noted logistical issues with using anesthetic hours such as the anesthesia start times varying based on the circumstances and that anesthetic hours would have to be converted from 15-minute increments into hours which could add to administrative burden. Another commenter recommended using procedures or patient encounters or asking each organization to select the normalization factor. The commenter noted TEAM participants could select operating rooms, anesthetic hours or procedures. By allowing organizations to select their normalization factor, CMS would get a sense for which ones would be most attractive. A commenter said the recommended normalization from the anesthesia community is “MAC-hour equivalents” compared with an ideal (sevoflurane) reference point. The commenter noted this is feasible for any organization that uses EHRs, and automated flow meters and this metric can account for the complex nuances that otherwise make normalizing anesthetics complicated.
Response: We understand there may be limitations using anesthetic hours as a normalizing factor, however, we believe this number can be relatively easily captured from administrative claims data and thus may be more feasible than information collected from an EHR. We are concerned that procedures may not be appropriate because the timing of procedures vary greatly and therefore, we are not requesting that information. As discussed in an earlier response, we will add operating rooms as a required element to evaluate it as a potential normalizing factor, especially for nitrous oxide. Finally, we are allowing, but not requiring, reporting of “MAC-hour equivalents.” A minimum alveolar concentration (MAC) hour equivalent allows comparison of the amount of administered anesthetic gas to an equivalent mass of carbon dioxide that would be emitted for driving an automobile a certain number of miles, for example. We are concerned that not all participants are able to retrieve this ( print page 69873) information easily from their EHR, but we understand the potential benefits of using MAC-hour equivalents as a normalizing factor, therefore, we will have an optional field for reporting this information.
Comment: A few commenters did not support reporting the assessment questions in lieu of reporting actual emissions from inhaled anesthetics. Commenters noted that CMS needed to start to collect this data in order to develop benchmarks and make comparisons. Commenters also noted this should be feasible as billing data and purchasing data should be universally available.
Response: We agree with commenters on the importance of capturing quantitative measures so TEAM participants can have benchmarks in comparison data. We also appreciate that data pulled from billing data and purchase records should be feasible. We do intend to finalize collection of anesthetic metrics with some modifications to the normalization factors.
Comment: A commenter supported the collection of responses to local structural questions as described in “Anesthetic Gas Metrics,” but asked for additional time before endorsing specific measures. The commenter noted that only a handful of anesthesia departments and their hospitals are collecting data. The commenter suggested that CMS develop a technical advisory panel to develop such measures or measure concepts and noted that they are planning to develop a suite of anesthesia-related environmental sustainability measures in the near future, with an eye toward gathering granular data from electronic health records, anesthesia machines, and other equipment. They requested CMS convene multistakeholder panels to develop other environmental sustainability measures and concepts that promote data exchange.
Response: In the future, CMS may consider establishing a technical advisory panel that is comprised of stakeholders which could provide feedback on measures, reporting and technical assistance.
Comment: A commenter recommended that the Decarbonization and Resilience Initiative include all areas within the hospital that use anesthetic gases, including nitrous oxide, and encourage responsibility from all service lines. The commenter noted an increase in the use of desflurane and isoflurane in non-operating room anesthetizing locations and that nitrous oxide is used services within the hospital such as obstetric, urological, and dental patients not provided by anesthesiologists. The commenter further stated their belief that hospitals provide anesthesia groups and departments with increased authority over administration of nitrous oxide and recommended that CMS be explicit in this proposal that all physicians and clinical staff have a responsibility to reduce their use of certain anesthetic agents.
Response: We intend to collect information for each facility but will allow health systems optionally to report for additional locations outside the inpatient facility. We do agree that facilities and clinicians should collectively work together to reduce their anesthetic emissions, but we do not believe we should inform TEAM participants who elect to participate in the Decarbonization and Resilience Initiative how they need to implement their efforts.
Comment: A few commenters had specific feedback on our proposed assessment question “Has your facility removed the use of desflurane or removed vaporizers when using desflurane?” Some commenters recommended modifying the question to assess the “mitigation of desflurane emissions” to allow for the clinically appropriate and medically necessary use of desflurane while encouraging mitigation of desflurane emissions, noting this wording more closely matches the language of the NAM Step II recommendation. Another commenter believed that the phrasing should be modified to “remove or eliminate desflurane from formulary.” Another commenter believed that changing the phrasing to “restrict access to desflurane vaporizers” would be more actionable.
Response: We thank the commenters for their feedback. We agree that desflurane at times may be clinically appropriate and medically necessary and intend to adjust the assessment questions to reflect that distinction.
Comment: A commenter had suggested edits to the other anesthetic assessment questions. The commenter recommended adding the question “Implement a low FGF practice notification in EMR.” and replace “Has your facility decommissioned piped nitrous oxide and substituted e-cylinders? If not, are these activities in process?” with two questions: “Deactivate central nitrous oxide supply systems” and “Transition to portable nitrous oxide E-cylinder supply.” The commenter noted the language and terms on this topic are important and carry specific meanings with facilities professionals and it is important for health and sustainability professionals to speak their language to gain their trust and partnership.
Response: We thank the commenters for their feedback. We will consider making these modifications to the assessment questions.
Comment: A commenter recommended the American Society of Anesthesiologists for reliable calculators, tools, and academic papers with up-to-date conversion factors.
Response: We thank the commenter for this suggestion. We will continue to evaluate tools as we assess the quantitative anesthetic data.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing at § 512.598(a)(3)(i) to include a metric for total GHG emissions from inhaled anesthetics using purchase records. We are finalizing with modification at § 512.598(a)(3)(ii) normalization factors that may include information on anesthetic hours, operating rooms, or MAC-hour equivalents. Finally, we are finalizing at § 512.598(b)(3)(iii) assessment questions based on key actions recommended for reducing emissions for anesthetic gases although we may modify those questions in the future.
The third category of information relevant to health care facilities is the GHG emissions related to leased or owned vehicles. We proposed at § 512.598(a)(4) a set of metrics that focus on greenhouse gases related to leased or owned vehicles. We proposed § 512.598(a)(4)(i) through (a)(4)(iii) metrics that include gallons for owned and leased vehicles consistent with GHGP Scope 1 requirements, patient encounter volume as a normalization factor, and assessment questions. For transportation emissions related to patient transportation and supply chain, please see the RFI on Scope 3 emissions which sought comment on the feasibility of reporting Scope 3 emissions such as those from Scope 3 transportation emissions (for example, patient transportation).
Including information on gallons for owned and leased vehicles aligns with the AHRQ primer core measure for transportation, and we anticipate that TEAM participants can capture this information. We also proposed that if TEAM participants choose to partake in the Decarbonization and Resilience Initiative Voluntary Reporting, we would require TEAM participants to capture patient encounter volume as a normalization factor and are considering a range of other factors consistent with ( print page 69874) GHG protocols such as full-time equivalents (FTEs).
We also proposed a series of assessment questions that align with the NAM recommended key actions to reduce transportation emissions. Assessment questions include the following:
- Has your facility set an emissions reduction goal related to transportation? If so, what is that goal?
- Has your facility executed plans to reduce fleet emissions (either from reducing miles or replacing with electric vehicles [EVs])?
- Has your facility identified measures to optimize product delivery?
- Has your facility provided (or in the process of providing) EV charging infrastructure?
We sought feedback on the proposed transportation metrics. Additionally, we sought feedback to the extent hospitals are tracking this information and the operational feasibility to track and report this information or if other alternative metrics may be more feasible (for example, mileage). Finally, while we believe it is important to capture both the data on the gallons of gas as well as the assessment questions, we also considered whether we provide the TEAM participants an option of reporting either the gallons data or reporting the assessment questions for the voluntary reporting program. We believe this flexibility for TEAM participants could reduce reporting burden and enhance participation, but we are concerned this alternative may not provide comparable data across the TEAM participants who voluntarily submit data. We sought feedback on this alternative for TEAM participants who choose to submit to report either gallons and patient encounter or to report the assessment questions.
We invited public comment on our proposal at § 512.598(a)(4) to use a set of metrics that focus on greenhouse gases related to leased or owned vehicles.
Comment: A commenter recommended that CMS expand the assessment questions outside of yes or no parameters. This commenter also recommended that we remove the criteria in the question “Has your facility executed plans to reduce fleet emissions (either from reducing miles or replacing with electric vehicles [EVs])?” which would reduce the question to “Has your facility executed plans to reduce fleet emissions?” The commenter believes that removing the criteria, additional emission reduction plans can be included. This commenter also believes that CMS should adopt questions relating to whether the facility has a policy or plan regarding reducing fleet emissions, the quantity of EV charging stations, and the distribution between owned and leased vehicles by the facility. Another commenter recommended that CMS eliminate the assessment question: “Has your facility identified measures to optimize product delivery?” because it is not related to Scope 1 emissions.
Response: We thank the commenters for their suggestions and may consider these suggestions for alterations to the assessment questions. CMS is considering modifications to the assessment questions to allow for more detailed structured responses and will publish the full and complete assessment questions in sub-regulatory guidance and/or technical reporting guidelines.
Comment: A commenter noted that CMS' “gallons for owned and leased vehicles” should include gas and diesel as these are the primary fuel types used. The commenter noted that additional fuel types include gas-electric hybrid, E-85, electricity, biodiesel, CNG, Hydrogen fuel cell, and could be collected as well.
Response: We thank commenter for their response. We note that “gallons for owned and leased vehicles” include gas and diesel fuel types but that other fuel types will be addressed in sub-regulatory guidance.
Comment: A commenter recommended allowing the reporting entity to select their normalization factor from a bounded set. The commenter did not support using patient encounter volume as a normalization factor. Another commenter noted that they collect a variety of patient volume data, total FTEs, and refined FTEs regarding telework for their normalization factors. The commenter recommended that should CMS collect only total outpatient visits and total FTEs, CMS should consider adjusting emission calculations per median telehealth and telework percent to totals.
Response: We thank commenters for their recommendations. We believe that patient encounter volume could be a normalization factor that helps to benchmark and compare performance both within and between institutions. [ 1059 1060 106 1062 ] We are collecting other normalization factors to compare TEAM participants to each other and are considering a range of other factors consistent with GHG protocols such as FTEs (which is already collected through ENERGY STAR Portfolio Manager. We may also consider the number of FTEs in the future when reviewing potential metrics regarding staff transportation if scope 3 is pursued.
Comment: A commenter provided feedback on our alternative proposal for TEAM participants who choose to submit to report either gallons and patient encounter or to report the assessment questions. The commenter recommended limiting reporting to the gallons data because it is congruent with TJC program and would align organizations around the same set of metrics.
Response: We thank the commenter for their suggestions on our alternative proposal for TEAM participants who choose to submit to report either gallons and patient encounter or to report the assessment questions. The assessment questions for facilities allow facilities to report whether they are taking key actions to reduce transportation emissions and provide a deeper understanding of a hospitals' commitment to reducing emissions. We believe that both the assessment questions and gallons reported are important in providing a whole-scope view on transportation at facilities.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal at § 512.598(a)(4) to use a set of metrics that focus on greenhouse gases related to leased or owned vehicles.
Both Scope 3 and MDI emissions account for a large percentage of medical carbon emissions and CMS is ( print page 69875) interested in potential ways in which to provide technical assistance to TEAM participants to assess available metrics to help reduce the enormity of this impact.
We believe Scope 3 emissions are relevant to a Decarbonization and Resilience Initiative connected to TEAM because Scope 3 emissions account for 82 percent of all U.S. health care emissions. Scope 3 includes all emissions upstream and downstream in the supply chain and other indirect emissions. We sought additional information regarding potential future voluntary reporting of Scope 3 emissions.
- What metrics or data collection elements would be appropriate for TEAM participants to accurately report Scope 3 emissions?
- Is there an industry standard tool that can be utilized for Scope 3 reporting?
- Which Scope 3 categories are most feasible and appropriate for hospitals participating in TEAM to report at this time?
- How can CMS and hospitals engage other parts of supply chain that contribute to Scope 3 emissions or incentivize their reduction of Scope 3 GHGs?
- Would hospital burden of Scope 3 reporting differ from Scope 1 and 2 reporting?
We summarize the feedback to our request for information as follows:
Comment: We received many comments related to Scope 3 reporting. Many commenters recommended certain Scope 3 metrics they believed were attainable for TEAM participants to collect, including normalizing patient encounters when it came to reusables vs single use devices, collecting waste volumes, patient and employee travel by mileage, and food waste. A few commenters recommended CMS to include Scope 3 metrics in the initiative, including making Scope 3 reporting mandatory and focusing on a limited but impactful set of metrics. A commenter suggested that Scope 3 is not burdensome to collect, and that there are a number of tools available for hospitals to use. This commenter recommended CMS to build its own tool in collaboration with EPA. A commenter recommended CMS provide leniency on Scope 3 metrics due to the complexity of these emissions in large hospital systems. A commenter recommended longer term technical assistance and for TEAM participants to use external regional vendors rather than CMS due to their familiarity with local supply information. A commenter gave an example of a health system who had undertaken and published a partial Scope 3 inventory for CMS to reference.
Response: We thank the commenters for their input, acknowledge their recommendations, and will take the feedback into consideration in future rulemaking. We also refer readers to section X.A.3.p.(4).(a).(ii). for additional comments and feedback on why we excluded Scope 3 from the first year of the initiative.
Comment: A commenter recommended CMS include metrics that encourage clinicians to choose treatments that have less carbon emissions.
Response: We agree with the commenter, and we believe that starting with Scope 1 and Scope 2 will help start the process. We will look into incorporating Scope 3 in future.
We thank commenters for their support and will consider this feedback for future iterations of the initiative.
Also, under Scope 3, we sought additional information regarding MDIs. We believe that further understanding of the MDI prescription and usage rates could assist in finding pathways of reduction and substitution to a less harmful environmental option. However, we understand that most MDI prescriptions and the management of related conditions occur in the outpatient setting and may not be directly relevant to TEAM participants. Hospital reductions in MDI prescriptions can still result in significant reductions of GHG emissions. For example, Providence Oregon hospitals identified clinically equivalent MDI formulations of albuterol with 3-fold differences in emissions. [ 1063 ] By prioritizing the lower emissions intensity inhalers, these emissions are projected to drop by 42 percent, or 298 tons of CO2e (the equivalent of 64 gasoline powered passenger vehicles driven) per year. We sought information on the feasibility of capturing information on MDI outpatient prescriptions as a percentage of all inhaler prescriptions relevant to TEAM participants.
What role do acute care hospitals, hospital-based pharmacies, or other providers in the inpatient setting play in prescribing MDIs and guiding patients toward environmentally preferable selections, such as dry powder inhaler, [ 1064 ] when clinically safe to do so?
We believe it would be important to record data such as the volume of each MDI cannister (micrograms) and number of MDI cannisters prescribed on an annual basis and this would be helpful to capture. We sought feedback on the feasibility of capturing information for the following questions:
- What is the utilization rate of MDIs and dry powder inhalers, for inpatients?
- What is the prescription rate of MDIs and dry powder inhalers?
- Is there a way to replace the MDI propellant from a hydrofluorocarbon to hydrofluoroalkane?
Comment: Many commenters provided feedback on MDIs. Commenters recommended ways of collecting MDIs, including number of prescriptions collected from EHRs, percentage of total possible MDI doses utilized, stratification by drug class, collecting formulation weights by grams rather than micrograms, requiring formularies to include environmental costs in their prioritization framework, frequency they are prescribing different albuterol formulations, including the dry powder form, and recommending devices that provide more actuations and less emissions. A commenter suggested that MDI emissions are complicated to assess and mitigate, requiring formulation-specific quantification within each drug class and action from multiple stakeholders. A commenter recommending using Medicare claims data to collect data rather than during the hospitalization when the patient is switched from an MDI to a Dry Powdered Inhaler (DPI). A commenter recommended using nebulized medications to reduce inpatient MDI use. A commenter supports collecting MDIs but believes that it does not seem to fit into the TEAM initiative since it focuses primarily on inpatients undergoing surgical procedures.
In response to the question of whether the MDI propellant from a ( print page 69876) hydrofluorocarbon can be replaced to hydrofluoroalkane, a commenter noted that both are in the same class of chemical, and it is up to the preference of the pharmaceutical company. This commenter noted that not all hydrofluoroalkanes are potent GHGs and that the pharmaceutical industry is working on bringing new low or no global warming potential propellants to the market that are also hydrofluoroalkanes. This same commenter also expressed concern that the development of new propellants would move this drug-device combination from generic back to patent protection, potential driving prices up for patients and payers, similar to what happened in the 2000s during the chlorofluorocarbon to hydrofluorocarbon propellant transition.
Response: We thank the commenters for their input and acknowledge their recommendations. We may take the feedback into consideration in future rulemaking.
Comment: A few commenters pointed out the difference between Scope 1 MDIs, which is directly released onsite, versus Scope 3 MDIs, which is used outside of the health system.
Response: We thank the commenters for their input, pointing to differences between Scope 1 MDIs and Scope 3 MDIs. While some MDI usage may be classified as Scope 1, we believe it would be important to review MDI usage in total and not try to separate between Scope 1 and Scope 3 for the initial years of the initiative.
We thank the commenters for their input and will take the feedback into consideration in future rulemaking.
For the Decarbonization and Resilience Initiative, we proposed at § 512.598(b) that, if TEAM participants so choose, they would report information annually to CMS after each performance period. The form and manner would be specified by CMS for each performance period including using ENERGY STAR Portfolio Manager for building energy metrics proposed in section X.A.3.p.(4).(a).(iv). of the preamble of this final rule. We anticipate reporting for the other metrics and assessment questions would be a survey and questionnaire in a form and manner specified by CMS. We also proposed at § 512.598(b) that the Decarbonization and Resilience Initiative information would need to be reported to CMS by no later than 120 days in the year following the performance period, or a later date as determined by CMS. We believe it is important to have flexibility to delay the reporting in case of an emergency or technical issue.
We also considered requiring reporting by June 1 after the performance period to align with the majority of the local decarbonization programs that report to ENERGY STAR. [ 1065 ] We sought comment on the proposed report timing and alternatives.
We summarize and respond to public comments received on the proposal at § 512.598(b) to require TEAM participants to report information below.
Comment: A few commenters supported annual reporting by TEAM participants to CMS after each performance period. On commenter recommended one year of technical assistance prior to requiring reporting to support education on the initiative requirements and how to structure their programs prior to initial reporting.
Response: We thank the commenters for their support. We will take into consideration the option of providing technical assistance prior to the launch of the Decarbonization and Resilience Initiative and will utilize TEAM participants feedback to inform future technical assistance.
Comment: A commenter encouraged CMS to shorten the reporting timeline, stating the health sector is behind other sectors.
Response: We thank the commenter for their feedback and may consider it in future rulemaking. Because the Decarbonization and Resilience Initiative is part of TEAM, TEAM participants who elect to voluntarily participate in the initiative will not need to begin reporting until TEAM starts. However, CMS may provide technical assistance to help TEAM participants to prepare for the initiative ahead of the start of this initiative as part of TEAM.
Comment: A commenter supported the reporting deadline of June 1st after the previous year's performance period.
Response: We thank the commenter for their support of the proposed reporting deadline.
After consideration of public comments received, we are finalizing our proposal at § 512.598(b) as proposed.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal to require TEAM participants to report information.
We proposed at § 512.598(c) that TEAM participants who elect to report all the metrics identified in section X.A.3.p.(4). of the preamble of this final rule in the manner described in section X.A.3.p.(5). of the preamble of this final rule would receive individualized feedback reports and be eligible to receive public recognition for their commitment to decarbonization. In addition to these proposed benefits, we believe TEAM participants may receive additional indirect benefits from engaging in the voluntary reporting portion of the Decarbonization and Resiliency Initiative.
We invited public comment on this proposal to offer benefits to TEAM participants who engage in voluntary reporting.
Comment: A few commenters supported proposed benefits to TEAM participants who elect to report in the decarbonization and resiliency initiative and recommended CMS reward TEAM participants by implementing a bonus to CQS or help offset the costs incurred by health care organizations with upfront funding or incentives for reporting.
Response: We thank comments for their support regarding the benefits to TEAM participants who elect to report in the decarbonization and resiliency initiative. At this time, we do not intend to modify the CQS, but we did seek feedback on how to incorporate financial incentives in the future and have a summary of comments in section X.A.3.p.(6).(d).
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal to offer benefits to TEAM participants who engage in voluntary reporting.
We proposed at § 512.598(c)(1) to provide individualized feedback reports to TEAM participants who voluntarily report to CMS the four emissions-related metrics in the Decarbonization and Resilience Initiative. We anticipate these reports would summarize facilities' emissions metrics and would include benchmarks, as feasible, for normalized metrics to compare facilities, in aggregate, to other TEAM participants in the Decarbonization and Resilience Initiative. While ENERGY STAR has many robust benchmarks related to building energy efficiency, we believe that TEAM participants would be able to learn additional information from peers about emissions from ( print page 69877) anesthetic gases and transportation emissions. See section X.A.3.p.(4).(a). of the preamble of this final rule for discussion of the proposed metrics and calculator tools to be used as part of the Decarbonization and Resilience Initiative. CMS does not intend to make these individualized feedback reports available to the public or other TEAM participants and intends them for the purpose of learning and improvement.
We invited public comment on this proposal to provide individualized feedback reports to TEAM participants.
Comment: Several commenters supported providing individualized feedback reports to TEAM participants who voluntarily report to CMS the four emissions-related metrics, noting the value of benchmark data to TEAM participants to help evaluate emissions and energy efficiencies, and with public recognition.
Comment: A few commenters recommended public disclosure of the general benchmarking data and verification at the facility level. A commenter requested CMS clarify with TEAM participants that reports can be shared with the public if a TEAM participant wants the data made public. A commenter recommended CMS provide individual feedback reports using relevant regional and hospital-type benchmarks.
Response: We appreciate the commenters' suggestion regarding public disclosure and verification of benchmarking data at the facility level. We will monitor data submitted by a TEAM participant for compliance with the requirements of this final rule and will ensure that the data is sufficiently complete. We will use processes similar to those used in other models in monitoring the data submitted by TEAM participants. At this time, due to limited resources, we will not be able to conduct audits of the data submitted. Finally, we want to caution that TEAM participant that receives data or information in an individualized feedback report from CMS as a participant in this initiative of TEAM must request in writing and receive written approval by CMS prior to publication or public disclosure of data or information contained in the individualized feedback report.
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal to provide individualized feedback reports to TEAM participants.
We proposed at § 512.598(c)(2) to establish a publicly reported hospital recognition badge for the commitment of the TEAM participant or of the TEAM participant's hospital corporate affiliate to decarbonization; CMS would post a hospital recognition badge on a CMS website. We would provide annual recognition to TEAM participants for reporting all the metrics detailed in section X.A.3.p.(4).(a). of the preamble of this final rule. The recognition badge would be reevaluated each year based on the reporting of performance year metrics to CMS. We believe adding this recognition to a consumer-facing CMS website would allow patients and families to choose hospitals that have participated in efforts to measure health care carbon emissions.
To encourage meaningful reductions in emissions, we sought comments on potentially expanding to a tiered recognition in future years. We believe a tiered approach could better acknowledge TEAM participants that have elected to voluntarily report their emissions data, actively engage in decarbonization activities that would result in reduced Scopes 1, 2, and 3 emissions, and meet absolute or relative standards of reported energy efficiency and lowered emissions. We sought comment on tiering such badging so as to recognize TEAM Participants that meet certain absolute or relative standards based on emissions reporting measures or other standards such as the Department of Energy's National Definition for a Zero Emission Building and may consider making select reported information public. [ 1066 ] Any modifications to the public recognition benefit would be addressed through future rulemaking.
We invited public comment on the proposed publicly reported hospital recognition of decarbonization commitment.
Comment: Several commenters supported a publicly reported hospital recognition badge for a TEAM participant's commitment to decarbonization. A few commenters recommended transparent and verifiable qualifications for a TEAM participant to receive a badge. A commenter recommended expanding the public recognition with a tiered approach to encourage continuous improvement in GHG reduction performance. A commenter recommended a sustainability badge on Care Compare to recruit and retain care staff and attract patients. A commenter recommended CMS recognition of TJC Sustainable Healthcare Certification to align sustainability programs.
Response: We thank the commenters for their support for a publicly reported hospital recognition badge for reporting all metrics detailed in section X.A.3.p.(4).(a). of the preamble of this final rule. As noted in section X.A.3.p.(3), we are allowing a TEAM participant to voluntarily report on metrics and respond to questions to CMS on behalf of the TEAM participant's hospital corporate affiliates. To support recognition of that additional voluntary reporting, a TEAM participant may receive a badge for the commitment of the TEAM participant or for the commitment of the TEAM participant's hospital corporate affiliates. We want to be clear that the publicly reported hospital recognition badge is for reporting and not for performance. We will monitor data submitted by a TEAM participant for compliance with the requirements of this final rule and will ensure that the data is sufficiently complete. At this time, due to limited resources, we will not be able to conduct audits of the data submitted. We will not be establishing a tiered approach as we want to gain experience with the data submitted by TEAM participants before further consideration of a tiered approach.
Comment: A commenter did not support a recognition badge for non-disclosed data and recommends verified data disclosure to ensure total transparency. Another commenter did not support a star-ranking system for environmental sustainability until future studies define what patients care about most and what the rankings mean in terms of patient safety and quality of care.
Response: We appreciate the commenters' concerns regarding recognition badges and the data used. We will monitor data submitted by a TEAM participant for compliance with the requirements of this final rule and will ensure that the data is sufficiently complete. At this time, we are not proposing public disclosure of data submitted by TEAM participants as doing so may act as a barrier to voluntary participation. Due to limited resources, we will not be able to conduct audits of the data submitted. ( print page 69878)
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposed publicly reported hospital recognition of decarbonization commitment.
We believe that in addition to the direct benefits of participating in the Decarbonization and Resilience Initiative there are several indirect benefits associated with the Initiative's efforts to assist interested TEAM participants in undertaking decarbonization and resilience activities. Decarbonization can help improve the financial well-being of health care facilities by reducing operational costs. Estimates indicate that up to 30 percent of the energy used in hospitals and other commercial buildings is consumed unnecessarily and investing in decarbonization has been shown to decrease operational costs through supply chain optimization and reduced energy consumption and expenditures. [ 1067 ]
Beyond the potential cost reduction benefit of decarbonization, investing in decarbonization may help to improve patient care and outcomes. For example, facilities that opt to reduce GHG emissions by switching to renewable energy sources increase their resilience and thus can bypass power outages in the electric grid during climate emergencies. Furthermore, by reducing GHG emissions, healthcare facilities are contributing to preventing or ameliorating adverse health outcomes that are linked to air pollution and climate change-related hazards like hurricanes (for example, respiratory illnesses, injury). [ 1068 ] Health systems could benefit patients by reduced demand for hospital services through encouraging health education, addressing health inequities perpetuated by social determinants of health, improving telehealth options, and improving upstream care management. A well-developed sustainability strategy could allow health systems to become more resilient to the consequences of extreme weather events, which exacerbate patients' chronic cardiac, respiratory, and other conditions. [ 1069 ]
We summarize and respond to public comments received regarding the indirect benefits for TEAM participants who elect to report on the metrics.
Comment: A few commenters agreed with the indirect benefits of participating in the Decarbonization and Resilience Initiative, including cost savings overall and specifically in anesthesia departments, and reductions in air pollution and improvements in air quality that impact healthcare savings and public health benefits.
Response: We thank commenters for their feedback.
Comment: A commenter suggested decarbonization may not reduce costs and may increase operational costs and cited possible ongoing depreciation expenses as an example.
Response: We thank the commenter for their input. We believe that decarbonization may provide opportunities to decrease operational costs. Reports have shown that the operational costs of electric vehicles are lower than that of a vehicle with an internal combustion engine. [ 1070 1071 ] Some efforts may not result in immediate cost savings but will be beneficial to the interests of hospitals and health systems. [ 1072 1073 ] It is important to note, that an exclusive focus on direct cost savings and immediate return on investment does not capture long-term costs associated with impacts of climate change (for example, infrastructure/physical damages, greater healthcare costs, etc.) and reputational costs. [ 1074 ]
After reviewing the public comments, for the reasons set forth in this rule, we are finalizing the proposal regarding the indirect benefits for TEAM participants who elect to report on the metrics.
At this time, we proposed not to include any bonuses, payments, or payment adjustments to TEAM participants for voluntary reporting in the Decarbonization and Resilience Initiative. We may add such a policy to the Decarbonization and Resilience Initiative in future years, subject to additional rulemaking. We sought feedback on the ways we could structure potential payments, bonuses, or payment adjustments. To offer some examples:
- A potential bonus added to the Composite Quality Score (CQS), which is discussed in section X.A.3.d.(5).(e). of the preamble of the proposed rule, for TEAM participants who report the information for the Decarbonization and Resilience Initiative. This would reward TEAM participants for collecting and reporting data, but not necessarily for better performance.
- We could elect to modify the CQS score by providing a bonus for those who perform well on the Decarbonization and Resilience Initiative. We welcomed thoughts on which metrics we should identify for measuring performance and how a bonus could be structured.
We invited public comment on the future bonuses, payments, or adjustments for participation in the Decarbonization and Resilience Initiative.
Comment: Many commenters recommended financial incentives to TEAM participants for voluntary reporting in the decarbonization and resilience initiative, including Medicare payment adjustments or bonus points; and linking reimbursement rates to emissions reductions or green medical supplies and pharmaceuticals. A few commenters recommended separate, targeted payments or upfront financial incentives, such as to hospitals serving low-income communities and focused on Scope 1 emissions; to small, independent, and rural hospitals; to hospital sustainability teams co-led by clinical and administrative leaders; and to hospitals to work with third parties. ( print page 69879)
A few commenters supported adding a potential bonus to the Composite Quality Score (CQS) for TEAM participants who report for the decarbonization and resilience initiative. A few commenters recommended revisions to the CQS score, to include establishing a climate resilience plan, and establishing an overall emissions reduction goal within the CQS.
A few commenters recommended additional administrative processes for the initiative including flexibility and a longer timeframe to address the uniqueness of healthcare operations as compared to other industries; audit or third-party verification of data reports to ensure accurate eligibility for bonus payments.
Response: We thank commenters for their input and acknowledge their recommendations and concerns. We may take commenters' feedback into consideration in future rulemaking related to financial incentives for voluntary reporting in the initiative.
Comment: A few commenters recommended non-financial incentives to TEAM participants for voluntary reporting in the decarbonization and resilience initiative including linking reporting to measurements of energy consumption, water usage, waste/disposal volume, and emission reduction; or the use of cloth gowns and scrubs, and reusable surgical equipment and tools.
Response: We thank the commenters for their suggestions and may consider other non-financial incentives for TEAM participants in future years.
Comment: A commenter did not support financial incentives to TEAM participants for voluntary reporting as bonuses or modifications related to GHG emissions would weaken the quality scoring system.
Response: We acknowledge the commenter's concern. At this point we are not finalizing financial incentives for the initiative. We will consider implications of a financial incentive on TEAM if we elect to do financial incentives in the future.
We appreciate the comments we received in response to the RFI and will consider them if we propose adding such a financial incentive policy to the initiative in future years, subject to additional rulemaking.
We note that we received comments and suggestions that were outside the scope of the proposed rule, which are not addressed in this final rule.
In the proposed rule we stated that the general provisions relating to termination of the model by CMS in 42 CFR 512.596 would apply to TEAM. Consistent with these provisions, in the event we terminate TEAM, we would provide written notice to TEAM participants specifying the grounds for termination and the effective date of such termination or ending. As provided by section 1115A(d)(2) of the Act and § 512.594, termination of the model under section 1115A(b)(3)(B) of the Act would not be subject to administrative or judicial review.
We received no comments on this proposal, and we are finalizing our proposal as proposed at § 512.596.
Section 1878 of the Act ( 42 U.S.C. 1395oo ) established by the Social Security Amendments of 1972, describes the role and function of the Provider Reimbursement Review Board (PRRB), a five-member administrative tribunal that adjudicates disputes over Medicare reimbursement for certain providers of services in the Medicare program. The statute requires the HHS Secretary to appoint individuals to the PRRB for a 3-year term of office; the law also established a shorter length of office for the first appointments for the newly created PRRB to permit staggered terms of office. To qualify for appointment to the PRRB, all members must be knowledgeable in the field of payment of providers of services; two members must be representative of a Medicare provider of services; and at least one member must be a certified public accountant. In 1974, the Social Security Administration (SSA), which administered the Medicare program prior to its transfer to the Health Care Financing Administration in the Department of Health and Human Services, promulgated the implementing regulations for the PRRB. The regulations governing the operation and administration of the PRRB reside at 42 CFR part 405 subpart R , with the provision governing the composition of the PRRB at 42 CFR 405.1845 . In addition to codifying the statutory requirements governing the composition of the PRRB, the regulations established that no Board Member is permitted to serve more than two consecutive 3-year terms of office and that the Secretary has the authority to terminate a Board Member's term of office for good cause.
When the PRRB was established more than 50 years ago, payment to providers participating in the Medicare program was on a cost reimbursement basis. Beginning October 1, 1983, Medicare transitioned to a prospective payment system for inpatient hospitals. These changes in reimbursement have led to changes in the types of cases adjudicated by the Board, the complexity of the matters that come before the Board, and often, the amount of time required to bring matters to resolution. While the limit on the number of consecutive terms served by a Board Member was established in the 1974 implementing regulations, CMS no longer believes that the current limitation on the number of consecutive terms a Board Member may serve makes good sense.
In the proposed rule, we sought public comment on our proposal to amend paragraphs (a) and (b) of 42 CFR 405.1845 , effective January 1, 2025.
- First, we sought to modify the requirement that Board Members shall be knowledgeable in the area of cost reimbursement, so that it instead requires them to be knowledgeable in the field of payment of providers under Medicare Part A.
- Second, we proposed to permit a Board Member to serve no more than three consecutive terms, instead of two consecutive terms allowed under current regulations.
- Third, we proposed to permit a Board Member who is designated as Chairperson in their second or third consecutive term to serve a fourth consecutive term to continue leading the Board as Chairperson.
The proposed change to paragraph (a) is intended to align the regulatory language with the statute, which, at section 1878(h) of the Act states, “All of the members of the Board shall be persons knowledgeable in the field of payment of providers of services . . .” As explained earlier in this preamble, Medicare payment to providers was on cost reimbursement basis when this provision became law; however, this change would clarify that a Board Member must have knowledge of Medicare Part A payment (which broadly covers the category of cases adjudicated by the PRRB, as opposed to the narrower subcategory of cost reimbursement matters). The proposed changes to paragraph (b) are intended to reduce the amount of turnover that occurs on the PRRB, enabling CMS to recruit and retain highly qualified individuals as they gain experience in adjudicating cases. We believe that these changes have the potential to expand the pool of applicants seeking to serve on the Board and who, because of the current two-term limitation, may not be willing to entertain a job change for what would be at most a 6-year period of service. Under current regulations, if a Board Member is serving in their first or second consecutive term and later ( print page 69880) designated as Chairperson, the total length of service on the PRRB remains 6 years, or two consecutive terms. In other words, a Board Member who is designated as Chairperson in year 4 or 5 of their second consecutive term is only permitted to serve 1 to 2 more years as Chairperson. Under the policy described in the proposed rule, the PRRB would continue to benefit from having an experienced Board Member serve for a total of 12 years, if they were designated as Chairperson in their second or third consecutive term.
We recognized in the proposed rule that the limit of two consecutive terms under current regulations creates more openings on the PRRB, which offers opportunities for newly appointed individuals to apply their unique skill sets, experience, and perspective to the work. However, we noted that there is an opportunity cost associated with the current level of turnover. Recruitment of Board Members occurs with regularity, generally every 1 to 3 years, and considerable time and effort have been expended by CMS and HHS in recruiting and vetting candidates as well as training newly appointed Board Members. Over time, it has been increasingly challenging to attract a large pool of qualified candidates who have relevant skills and experience in matters that come before the PRRB.
Even after a candidate is identified, they must be formally appointed to the PRRB by the Secretary. Upon accepting the appointment, a Board Member must devote significant time to learning the duties of the job. As a result, in our experience, a newer Board Member takes more time to complete tasks relative to their colleagues who have more experience in the role. While Board Members may have a strong legal, accounting, health care, or other professional background, this position often is the first time they are serving as an adjudicator. Conversely, when a Board Member departs, there is a loss of institutional knowledge and expertise that adversely impacts efficiency and productivity. Turnover also impacts the relationships among and between the Board Members, and it takes time for the newly constituted Board to learn how to work together. This proposal would decrease the frequency of turnover and permit lengthier periods of service for Board Members, which we believe would have the potential to increase the PRRB's efficiency and productivity.
The volume of cases filed with the PRRB has remained relatively steady over the past several decades with the average number of appeals filed and closed annually hovering around 2,000. The PRRB's docket has experienced years in which fewer appeals were filed in large part due to holds on issuing Notices of Program Reimbursement from which many providers file their appeals. A year or years with a lower appeals volume was then followed in subsequent years by spikes of new appeals once the holds were lifted. The PRRB's total docket has ranged from about 5,000 appeals to about 10,000 appeals over the last 30 years, with an average ending annual inventory of 8,700 cases. The PRRB's fiscal year 2023 docket ended with 8,698 open appeals.
Additionally, the nature of the PRRB's cases has evolved over time. For example, in the past decade, the PRRB has seen an increase in broad-based legal challenges to regulatory interpretations and fewer appeals of reimbursable expenses specific to individual providers, which were common in the early years of the PRRB's operation. For example, early on, disputes over a provider's allowable costs in its cost report involving such expenses as owners' compensation, malpractice insurance, and marketing expenses were the norm, and generally these issues are simpler matters to adjudicate. With the evolution of Part A reimbursement to a prospective payment system, appeals to the PRRB frequently involve nuanced issues that implicate highly specialized and complex areas of law. Cases that have been adjudicated by the PRRB often reach the federal courts, and on occasion, are decided by the U.S. Supreme Court. [ 1075 ] Permitting Board Members to serve more than two consecutive terms would allow them greater opportunity to follow the landscape of issues under judicial review, as it is not unusual for it to take years for cases to wind their way through the courts. Over their length of service, a Board Member develops an understanding of how certain issues are decided in the courts and applies that knowledge to the issues presented to the PRRB. The longer length of service would allow Board Members to obtain a deeper understanding of, and knowledge about, pertinent issues and caselaw.
In the proposed rule, we explained that we considered a policy of permitting a Board Member to serve four consecutive 3-year terms, which would have permitted an individual to serve as long as 12 years (with the potential of serving another 3 years, or 15 years total, if the Board Member would later be designated as Chairperson), as opposed to 9 years. Making a Board Member eligible to serve as many as four consecutive 3-year terms could have an advantage over three consecutive terms, as there will be less Member turnover and a greater ability to retain highly qualified Board Members. We sought public comment on this alternative option of four consecutive terms rather than three.
As explained in the proposed rule, we also considered permitting a Board Member who ascends to the position of Chairperson to serve an additional two or three consecutive terms, instead of the proposed one additional consecutive term. Such a policy would have permitted an individual to serve 15 or 18 years (three 3-year terms as a Board Member and another two or three 3-year terms as Chairperson). Allowing a Board Member who is later designated as Chairperson to serve two or three additional consecutive terms would have likely made all Board vacancies more attractive (given the prospect of career progression and a longer tenure) and provide a longer period for a Board Member to gain experience prior to assuming the role of Chairperson, as they developed the knowledge, skills, and abilities in serve in a leadership capacity on the Board. We solicited comment on these alternative options for the extended tenure of the Chairperson and whether our proposal or one of the alternative proposals best struck a balance between an appropriate level of turnover and CMS's desire to recruit and retain qualified Board Members.
Comment: Several commenters supporting our CMS's proposal to require Board Members to possess knowledge of Medicare Part A reimbursement, with the commenters noting that knowledge of payment to providers alone is insufficient. The commenters stated that it is critical that Board Members have specific knowledge and expertise in Medicare Part A reimbursement given the complex and unique types of Medicare cost report appeals that the PRRB adjudicates. These commenters also observed that this proposed amendment more appropriately reflects the statutory requirement of requiring Board Members to be knowledgeable in the field of “payment of providers of services.”
Response: We agree. As expressed earlier in this preamble, we seek to clarify that a Board Member must have knowledge of Medicare Part A payment, which covers the diversity of cases ( print page 69881) adjudicated by the PRRB, as opposed to cost reimbursement matters alone.
Comment: We received a comment expressing opposition to the proposed policy of allowing the term of Board Members be extended under certain circumstances up to 18 years. The commenter stated that the current system is working well and expressed concern about members with 9- to 18-year terms becoming entrenched. Other commenters expressed opposition to the proposed change and urged CMS instead to explore other options, such as higher pay which would serve as an incentive to attract highly qualified applicants to Board positions. Other commenters cautioned that the relaxation of term limits would deprive the Board of the regular infusion of fresh experience and perspectives that new Board Members bring.
Response: As explained earlier this preamble, turnover on the Board occurs with regularity, which has disruptive impacts on the Board's productivity and efficiency. Like anyone new to a position, it takes time to maximize a Board Member's contributions to the PRRB, which under regulations in effect prior to the effective date of this provision (January 1, 2025), would leave only one more 3-year term to apply the institutional knowledge and expertise they have acquired. Furthermore, a Board Member's departure at the end of their tenure creates a loss of such institutional knowledge and expertise; upon filling that vacancy, this cycle starts over again. However, we also recognize the commenters' concerns about permitting a Board Member to serve as long as 12 to 18 years and the risks of having such a long tenure. As such, we will not be finalizing the proposals to have Board Members serve more than three consecutive 3-year terms at this time.
Comment: Commenters questioned the effective date of January 1, 2025, as to when these PRRB composition-related changes were proposed to take effect. These commenters noted that by making them effective within months of issuance of the final rule, it creates an appearance that CMS seeks to reward current Board Members who might be sympathetic to the agency's position, which in turn undermines the legitimacy of the Board.
Response: We disagree with the commenters' characterization of the proposed effective date. As explained earlier in this preamble, it is time and resource intensive to recruit, screen, and appoint candidates to the PRRB, which occurs on a regular cadence. Furthermore, there is an upfront investment of time and effort on the part of the newly appointed Board Member to learn their role and responsibilities and grow into the position. This set of changes to the regulations preserves the Secretary's existing long-standing authority to decide whether to reappoint a Board Member to a consecutive term or terminate a Board Member's term of office for good cause.
After consideration of the public comments received, we are finalizing our proposal to require Board Members to be knowledgeable in the field of payment of providers under Medicare Part A. Additionally, we are finalizing our proposal to permit a Board Member to serve no more than three 3-year consecutive terms. At this time, we are not finalizing any policy that modifies the number of consecutive terms served by the Chairperson. These regulatory changes become effective January 1, 2025.
As described in the White House Blueprint for Addressing the Maternal Health Crisis and in the CMS Maternity Care Action Plan, we are committed to reducing maternal health disparities and improving maternal health outcomes during pregnancy, childbirth, and the postpartum period. [ 1076 1077 ] In alignment with our commitment to addressing the maternal health crisis, this RFI sought to gather information on differences between hospital resources required to provide inpatient pregnancy and childbirth services to Medicare patients as compared to non-Medicare patients. To the extent that the resources required differ between patient populations, we also requested information on the extent to which non-Medicare payers, or other commercial insurers, may be using the IPPS as a basis for determining their payment rates for inpatient pregnancy and childbirth services and the effect, if any, that the use of the IPPS as a basis for determining payment by those payers may have on maternal health outcomes.
As explained in section II.A. of the preamble of this final rule, section 1886(d)(4) of the Act requires the Secretary to establish a classification of inpatient hospital discharges by diagnosis-related groups and a methodology for classifying specific hospital discharges within these groups. We refer to these groups of diagnoses as the IPPS Medicare Severity Diagnosis Related Groups (MS-DRGs). For each MS-DRG, the Secretary is required to assign an appropriate weighting factor which reflects the relative hospital resources used with respect to discharges classified within that group compared to discharges classified within other groups. The Secretary is also required to adjust the MS-DRG classifications and weighting factors at least annually to reflect changes in treatment patterns, technology, and other factors which may change the relative use of hospital resources.
As discussed in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58652 ), our goal is always to use the best available data overall for ratesetting, including the calculation of the IPPS MS-DRG relative weights. We primarily utilize Medicare claims data and Medicare cost report data for IPPS ratesetting for inpatient hospital services. The claims data we utilize is specific to the Medicare beneficiaries population, which includes people 65 and older or people with disabilities, End-Stage Renal Disease, or amyotrophic lateral sclerosis (ALS) that qualifies them for Medicare earlier than the age of 65. [ 1078 ] Although most Medicare beneficiaries are 65 and older, in 2021 around 13% of the total share of Medicare beneficiaries were under the age of 65. [ 1079 ] Therefore, people of reproductive age may have Medicare as their primary health insurance. Notably, a study from the National Institutes of Health found that pregnant women with disabilities have higher risks for maternal mortality and severe complications during birth and pregnancy compared to other pregnant women. [ 1080 ] Thus, considering we utilize data that is specific to the Medicare beneficiary population in our ratesetting for inpatient hospital services we caution against using the IPPS rates and DRGs without first taking ( print page 69882) into account the characteristics of the Medicare beneficiary population.
This RFI generally sought to gather information on differences between the resources required to provide inpatient obstetrical services to Medicare patients, on which the IPPS MS-DRGs relative weights for those services are based, as compared to non-Medicare patients. To the extent that the resources required differ, we also sought information regarding the extent to which non-Medicare payers, such as state Medicaid programs, may be using the IPPS MS-DRG relative weights to determine payment for inpatient obstetrical services and the effect, if any, that use may have on maternal health outcomes. We asked some specific questions to help facilitate feedback on this issue and more broadly on maternal heath, including the questions that follow.
- What policy options could help drive improvements in maternal health outcomes?
- How can CMS support hospitals in improving maternal health outcomes?
- What payment models have impacted maternal health outcomes, and how?
- What payment models have been effective in improving maternal health outcomes, especially in rural areas?
- What factors influence the number of vaginal deliveries and cesarean deliveries?
- What types of modifications or assumptions, if any, are being made by payers when they are using the IPPS MS-DRG relative weights to account for the fact they are based on the Medicare beneficiary population?
- Does the use of the IPPS MS-DRG relative weights as the basis for setting rates for other payers, including state Medicaid programs, impact efforts to reduce low-risk cesarean deliveries?
- To what extent are Medicare claims and cost report data reflective of the differences in relative costs between vaginal births and cesarean section births for non-Medicare patients?
- Are there other data beyond claims and cost reports that Medicare should consider incorporating in development of relative weights for vaginal births and cesarean section births?
- What impact, if any, does the relatively lower numbers of births in Medicare have on the variability of the relative weights?
- What effect, if any, does potential variability in the relative weights on an annual basis have on maternal health outcomes?
We also noted our longstanding principle, reiterated each year in the IPPS rulemaking, that facilities should not consider differences in relative weights when making treatment decisions.
Comment: Generally, commenters expressed appreciation for CMS' and the Administration's interest and commitment to maternal health and improving maternal health outcomes. Commenters provided a wide range of feedback to the questions in the Maternity Care RFI, which is summarized in the following paragraphs.
Some commenters stated that payment rates to providers and health care professionals for pre/postnatal care, delivery, and related maternity care services were inadequate regardless of the payer. Some commenters said that there were structural issues with how payers pay for maternity care services and suggested payers restructure payments. Some commenters provided examples of payment restructuring that would include standby capacity payments, delivery fees, and federal add-on payments for labor and delivery.
Many commenters mentioned the importance of Disproportionate Share Hospital payments, Uncompensated Care Payments, and adequate payments from Medicare for all hospital services as keys to indirectly supporting hospitals that provide maternity care services.
Some commenters, including a national hospital association, stated that Medicare payment rates are generally not perceived to be a driver of practice patterns in maternity care.
Some commenters indicated that Medicare rates do impact other payers' payment rates in general, as well as specifically for maternity care services. For example, some comments discussed how Medicare payment rates are often used as a benchmark by state Medicaid programs for setting rates. With regard to commercial payers, various commenters pointed out that payment rates are set via contractual negotiations with providers, among other factors.
Various commenters indicated that Medicaid plays an important role in the delivery of maternity care services, and therefore encouraged CMS to work with state Medicaid agencies.
Most commenters acknowledged that resources to treat Medicare beneficiaries may differ from the resources required to treat a non-Medicare population. However, some commenters stated that they did not believe that the current MS-DRG structure and weights adequately reflect the resource consumption of maternity care services. Additionally, some of those commenters suggested that CMS use supplemental data to adjust the MS-DRGs and weights.
Other suggestions to support improvements in maternal health outcomes included use of value-based care arrangements, standardizing quality reporting across payers, establishing support programs for care for mental health and substance use disorder, and for addressing housing and food security challenges.
Response: We appreciate the many thoughtful comments we received from hospitals, hospital associations, health systems, beneficiary groups, and others. We will consider the comments received for future actions in our ongoing efforts to reduce maternal health disparities and improve maternal health outcomes during pregnancy, childbirth, and the postpartum period of maternal health.
The Payment Integrity Information Act of 2019 requires federal agencies to annually review programs susceptible to significant improper payments, estimate the amount of improper payments, report those estimates to Congress, and submit a report on actions the agency is taking to reduce the improper payments.
Medicaid and the Children's Health Insurance Program (CHIP) were identified as programs at risk for significant improper payments by the Office of Management and Budget (OMB). We measure Medicaid and CHIP improper payments through the Payment Error Rate Measurement (PERM) program. Under PERM, reviews are conducted in three component areas (FFS, managed care, and eligibility) for both the Medicaid program and CHIP. The results of these reviews are used to produce national program improper payment rates, as well as state-specific program improper payment rates. The PERM program uses a 17-state, 3-year rotation cycle for measuring improper payments, so every state is measured once every 3 years.
Section 202 of Division N of the Further Consolidated Appropriations Act, 2020 (FCAA, 2020) ( Pub. L. 116-94 ) amended Medicaid program integrity requirements in Puerto Rico. Puerto Rico was required to publish a plan, developed by Puerto Rico in coordination with CMS, and approved by the CMS Administrator, not later than 18 months after the FCAA's enactment, for how Puerto Rico would develop measures to comply with the PERM requirements of 42 CFR part 431, subpart Q . Puerto Rico published this ( print page 69883) plan on June 20, 2021, [ 1081 ] and it was approved by the CMS Administrator on June 22, 2021. In the proposed rule, we proposed to remove the exclusion of Puerto Rico from the PERM program found at 42 CFR 431.954(b)(3) . In compliance with section 202 of Division N of the FCAA, 2020, Puerto Rico has developed measures to comply with the PERM requirements of 42 CFR part 431, subpart Q . Including Puerto Rico in the PERM program will increase transparency in its Medicaid and CHIP operations and will improve program integrity efforts that protect taxpayer dollars from improper payments.
We proposed that Puerto Rico would be incorporated into the PERM program starting in RY27 (Cycle 3), which covers the payment period between July 1, 2025 through June 30, 2026.
We received no comments on this proposal and therefore are finalizing this provision with minor technical correction based on further review of current statute reference. Three references to the Improper Payments Information Act (IPIA) of 2002 ( Pub. L. 107-300 ) will be updated to the Payment Integrity Information Act (PIIA) of 2019 ( Pub. L. 116-117 ). Otherwise, the provision will be finalized without modification.
Under sections 1866 and 1902 of the Act, providers of services seeking to participate in the Medicare or Medicaid program, or both, must enter into an agreement with the Secretary or the state Medicaid agency, as appropriate. Hospitals (all hospitals to which the requirements of 42 CFR part 482 apply, including short-term acute care hospitals, LTC hospitals, rehabilitation hospitals, psychiatric hospitals, cancer hospitals, and children's hospitals) and CAHs seeking to be Medicare and Medicaid providers of services under 42 CFR part 485, subpart F , must be certified as meeting Federal participation requirements. Our conditions of participation (CoPs), conditions for coverage (CfCs), and requirements set out the patient health and safety protections established by the Secretary for various types of providers and suppliers. The specific statutory authority for hospital CoPs is set forth in section 1861(e) of the Act; section 1820(e) of the Act provides similar authority for CAHs. The hospital provision at section 1861(e)(9) of the Act authorizes the Secretary to issue any regulations he or she deems necessary to protect the health and safety of patients receiving services in those facilities; the CAH provision at section 1820(e)(3) of the Act authorizes the Secretary to issue such other criteria as he or she may require. The CoPs are codified at 42 CFR part 482 for hospitals, and at 42 CFR part 485, subpart F , f or CAHs.
Our CoPs at § 482.42 for hospitals and § 485.640 for CAHs require that hospitals and CAHs, respectively, have active facility-wide programs for the surveillance, prevention, and control of healthcare-associated infections (HAIs) and other infectious diseases and for the optimization of antibiotic use through stewardship. Additionally, the programs must demonstrate adherence to nationally recognized infection prevention and control guidelines, as well as to best practices for improving antibiotic use where applicable, and for reducing the development and transmission of HAIs and antibiotic-resistant organisms. Infection prevention and control problems and antibiotic use issues identified in the required hospital and CAH programs must also be addressed in coordination with facility-wide quality assessment and performance improvement (QAPI) programs.
Infection prevention and control is a primary goal and responsibility of hospitals and CAHs in their normal day-to-day operations, and these programs have been at the center of initiatives taking place in hospitals and CAHs since the beginning of the Public Health Emergency (PHE) for COVID-19. Our regulations for hospitals and CAHs at §§ 482.42(a)(3) and 485.640(a)(3), respectively, require infection prevention and control program policies to address any infection control issues identified by public health authorities.
On March 4, 2020, we issued guidance stating that hospitals should inform infection prevention and control services, local and state public health authorities, and other health care facility staff as appropriate about the presence of a person under investigation for COVID-19 (QSO-20-13-Hospitals). CMS followed this guidance with an interim final rule with comment period (IFC), “Medicare and Medicaid Programs, Clinical Laboratory Improvement Amendments (CLIA), and Patient Protection and Affordable Care Act; Additional Policy and Regulatory Revisions in Response to the COVID-19 Public Health Emergency,” published on September 2, 2020 ( 85 FR 54820 ), that required hospitals and CAHs to report important data critical to support the fight against COVID-19. The IFC provisions specifically required that hospitals and CAHs report specified information about COVID-19 in a format and frequency specified by the Secretary. Examples of data elements that could be required to be reported included things such as the number of staffed beds in a hospital and the number of those that are occupied, information about its supplies, and a count of patients currently hospitalized who have laboratory-confirmed COVID-19. These elements proved essential for developing and directing implementation of infection prevention and control guidance, as well as resource allocations and technical assistance during the PHE.
On August 10, 2022, we finalized revisions to the COVID-19 and Seasonal Influenza reporting standards for hospitals and CAHs (at §§ 482.42(e) and (f); and 485.640(d) and (e), respectively) in the FY 2023 IPPS final rule “Medicare Program; Hospital Inpatient Prospective Payment Systems for Acute Care Hospitals and the Long Term Care Hospital Prospective Payment System and Policy Changes and Fiscal Year 2023 Rates” ( 87 FR 48780 , 49409 ), to require that, beginning at the conclusion of the COVID-19 PHE declaration and continuing until April 30, 2024, hospitals and CAHs must electronically report information about COVID-19 and seasonal influenza virus, influenza-like illness, and severe acute respiratory infection in a standardized format specified by the Secretary. In establishing these requirements, we stressed that such reporting continued to be necessary for CMS to monitor whether individual hospitals and CAHs were appropriately tracking, planning for, responding to, and mitigating the spread and impact of COVID-19 and influenza on patients, the staff who care for them, and the general public ( 87 FR 49377 ). We also noted that the approach finalized in that rule would provide a path towards ending the overall reporting of COVID-19-related data between the end of the current PHE and April 2024, when those requirements would sunset ( 87 FR 49379 ).
The COVID-19 pandemic highlighted the importance of taking a broad view of patient safety—one that recognizes patient safety is determined not just by what is happening at the bedside, but also what is happening in the broader hospital, and in hospitals across the region, state, and country. At the same time, it also demonstrated the patient ( print page 69884) benefits of strong integration between public health and health care systems, particularly when data are available to direct collaborative actions that protect patient and public health and safety. Data from health care providers remain the key driver to identify and respond to public health threats, yet health care and public health data systems have long persisted on separate, often poorly compatible tracks.
Hospital and CAH-reported data on COVID-19, influenza, and RSV infections among patients, as well as hospital bed capacity and occupancy rates, continue to play a critical role in infection prevention and control efforts at every level of the health system. The value of these data extend beyond the COVID-19 PHE. For example, source control remains an important intervention during periods of higher respiratory virus transmission. [ 1082 ] Data on hospital admissions reported under the current CoPs continue to inform national, state, and county recommendations for community and health care mitigation measures. [ 1083 ] Notably, the CDC recommends that health care facilities consider levels of respiratory virus transmission in the whole community when making decisions about source control. Comprehensive and consistent surveillance across hospitals creates a shared resource that all health care facilities in a community could use to inform infection control policies. Hospital and CAH requirements to report this data ended in April 2024. Not maintaining this reporting would result in an absence of vital information on local, regional, and national transmission and impact of respiratory illness and overall healthcare system capacity, with significant implications for both patient care and public health mitigation.
In the proposed rule, we provided a detailed discussion regarding the data produced by the hospital and CAH respiratory virus reporting requirements and how the insight provided by the data collected positively impacted patient health and safety by guiding actions to reduce the prevalence of respiratory illnesses through enhanced planning, technical assistance, resource allocation, and coordination. We encourage readers to refer to the proposed rule for this detailed discussion ( 89 FR 36504-36505 ). [ 1084 ]
In response to the proposed rule, we received 1,377 total comments (709 unique) from patients, providers, medical professionals, national and state hospital associations, and pharmaceutical and biotech companies. In light of continued utility of respiratory illness data, the proposed policy aimed to continue national monitoring of COVID-19, influenza, and RSV cases to guide infection control interventions and hospital operations that directly relate to patient safety; monitor emerging and evolving respiratory illnesses; guide and motivate community-level disease control interventions; and enhance preparedness and resiliency to improve health system responses to future threats, including pandemics that pose catastrophic risks to patient safety and the health care system.
In this final rule, we provide a summary of the proposed provisions, a summary of the public comments received, and our responses to them, and an explanation for changes in the policies we are finalizing.
Comment: We received overwhelming support from patients and community members on our proposal to extend requirements for respiratory illness reporting in the hospital and CAH CoPs. Many commenters expressed that such data, when reported publicly, helps to inform both public and personal healthcare decisions. We received some anecdotes on personal experiences with long-COVID as well as stories of loved ones who died due to COVID. Although the PHE is over, commenters remind us that the threat to individuals remains, and many stated that accessible data is the number one factor in determining personal risk, especially for those who are immunocompromised. Many commenters mentioned how they rely on published public health data to inform their personal health decisions. For that reason, many recommend publishing the collected data to an easily accessible location, such as HealthData.gov . Likewise, a few state hospital organizations and epidemiology industry groups voiced support for the proposal, noting that data collection is vital for the informed preparation and coordination of hospital operations before, during, and after surges of respiratory illnesses.
Response: We thank commenters for their overwhelming support of the hospital data reporting and for sharing their personal stories regarding the impact of COVID. The compelling narratives shared by commenters demonstrate importance of data reporting to inform both public health and personal safety. We support the publication of data in a publicly accessible manner. Previously, the COVID-19 and influenza hospitalization data were displayed in multiple ways on the CDC website. CDC will use similar approaches to communicate these data in the future, including continuing to provide data visualizations on our website and posting updated weekly data aggregated by state on data.CDC.gov . Consistent data on COVID-19, influenza, and RSV hospitalizations will facilitate clear and comprehensive communication to health care organizations and to the public about major viral respiratory disease trends and burden. In addition to disseminating tabular data and descriptive statistics calculated from the data, respiratory virus hospital admission data has underpinned publicly available advanced analytics including short-term forecasts, longer-term scenario projections, and analyses that quantify the epidemic trajectory in near real-time. The data will continue to be used in these types of analyses, which help the public interpret the data and understand what is happening in their state.
Comment: A few hospital associations expressed concern about the appropriateness of data reporting as a CoP requirement. These comments emphasized that establishing CoPs may threaten access to Medicare participation, facility financial viability, access to care, and operational efficiency, and hinder true infection prevention efforts. They also noted that data reporting may not accurately reflect community prevalence. While these groups noted the value of data reporting, many reflected on the existing willingness of hospitals to participate in voluntary data sharing. A few commenters suggested alternative mechanisms to foster data collection rather than through the CoPs, such as supporting infrastructure for voluntary disclosure that could lead to long-term automated, efficient data sharing.
Response: We appreciate the feedback from these commenters; however, we ( print page 69885) disagree that the CoPs are an inappropriate means to assure essential data collection that protects the health and safety of patients. In our experience during the COVID-PHE, when similar data reporting requirements were first established, the data produced by hospital respiratory virus reporting requirements informed coordination of hospital operations and were especially important to anticipate and prepare for surge conditions. Collaborative, data driven approaches could help to manage patient transfers and alleviate strained hospitals, ultimately improving patient care by assuring that hospital resources are not overly strained to the point of patient harm. During the COVID PHE, state and local agencies, health care coalitions, and health systems used hospital capacity data to coordinate patient placement and reduce emergency department (ED) boarding and overcrowding, all of which improve the patient experience of care. Insight into hospital and CAH capacity helps ensure capabilities are available to meet patient needs with quality care through enhanced planning, technical assistance, resource allocation, and coordination.
Since the April 30, 2024, sunset of the COVID-19 data reporting requirement, data reporting has been voluntary for facilities. Since May 1, 2024, without the CoP requirement, reporting has dropped from near complete reporting by all US hospitals each week to only around 35 percent of hospitals reporting, representing nearly a 65 percentage point reduction. There is also significant variability by state, so that several states have zero reporting, leaving gaps in the visibility of the healthcare system and the burden of respiratory illness. The majority of hospitals that are continuing to report data to the National Healthcare Safety Network (NHSN) are doing so on a daily basis, and we appreciate their dedication and recognition of the importance of this reporting. Due to the dramatic decrease of data reporting during the voluntary period, we conclude that voluntary reporting is insufficient to capture this data on a consistently widespread and accurate basis. Information sharing across the health care ecosystem helps the health care community to prepare for, and effectively respond to, respiratory illness surges in ways that maintain the safety and availability of critical care services.
We recognize that neither the number of incident respiratory virus hospital admissions nor the number of prevalent respiratory virus hospitalizations are direct measures of community prevalence (that is, the proportion of people in a community who are infectious). Indeed, no respiratory virus surveillance system routinely used in the United States directly measures infection prevalence in the community. It is therefore necessary to use proxy measures. Rates of respiratory virus hospital admissions are strongly affected by transmission in the community and therefore these data serve as a valuable correlate of community transmission dynamics.
Overall, we note that the information reported would be shared with both CMS and CDC, retained, and publicly reported to support protecting the health and safety of patients as well as facility personnel and the general public. These requirements would support our efforts to proactively and transparently inform interested parties and ensure that the most complete information on viruses is available.
Comment: Some commenters suggest delaying the compliance date to ensure that hospitals would be prepared to comply. A commenter stated that an October 2024 start date for reporting influenza and RSV data is too soon since this data has not been previously required. The commenter explained that facilities would need time to operationalize this requirement.
Response: We understand the implementation timeline concerns expressed by commenters. However, due to the unpredictable nature of viruses, it is vital that this information be collected and recorded in a timely manner. We are working to avoid significant gaps in data and expect hospitals to be prepared to report, as the finalized policy is a reduction from the already familiar required reporting that ended in April 2024. Retaining the data reporting requirements is an important element of maintaining effective surveillance of novel viruses. In addition, there are still significant risks of morbidity and mortality for immunocompromised patients. Timely and actionable surveillance enables CMS to continue to respond to facilities in need of additional technical support and oversight to assure patient health and safety. As such, we are finalizing our proposal to extend a streamlined data set of required ongoing reporting and additional reporting in the event of a future PHE for an acute infectious illness, effective November 1, 2024. We note that there is a 90-day delay between the date of publication of this final rule and the effective date of these requirements, allowing hospitals sufficient time to prepare for implementing the streamlined data reporting requirements.
We proposed to revise the hospital and CAH infection prevention and control and antibiotic stewardship programs CoPs to extend a modified form of the current COVID-19 and influenza reporting requirements that would include data for RSV and reduce the frequency of reporting for hospitals and CAHs. Specifically, we proposed to replace the COVID-19 and Seasonal Influenza reporting standards for hospitals and CAHs at § 482.42(e) and (f) and § 485.640(d) and (e), respectively, with a new standard addressing respiratory illnesses to require that, beginning on October 1, 2024, hospitals and CAHs electronically report information about COVID-19, influenza, and RSV in a standardized format and frequency specified by the Secretary. To the extent determined by the Secretary, we proposed that the data elements for which reporting would be required at this time include—
- Confirmed infections of respiratory illnesses, including COVID-19, influenza, and RSV, among hospitalized patients;
- Hospital bed census and capacity (both overall and by hospital setting and population group [adult or pediatric]); and
- Limited patient demographic information, including age.
Therefore, outside of a declared PHE for an acute infectious illness, we proposed that hospitals and CAHs would have to report these data on a weekly basis (either in the form of weekly totals or snapshots of key indicators) through the NHSN or other CDC-owned or CDC-supported system as determined by the Secretary.
We noted that the proposed policy was scaled back and tailored from the post-COVID-19 PHE requirements that expired April 30, 2024, and that we intend to continue the collection of the minimally necessary data to maintain a level of situational awareness that would benefit patients and hospitals across the country while reducing reporting burden on hospitals and CAHs.
We welcomed public comments on our proposals and on ways that reporting burden could be minimized while still providing adequate and actionable data. We also requested feedback on any challenges of collecting and reporting these data; ways that CMS could reduce reporting burden for facilities; alternative reporting mechanisms or quality reporting programs through which CMS could ( print page 69886) instead effectively and sustainably incentivize reporting and the value of these data in protecting the health and safety of individuals receiving treatment and working in hospitals and CAHs.
In the proposed rule, we also discussed the impact of the COVID-19 pandemic on communities across the United States, and the disproportionate impact on socially vulnerable populations. We noted that from the beginning, reports indicated that people of color and people from economically disadvantaged communities were at an increased risk of becoming sick from COVID-19, being hospitalized due to COVID-19, and dying from COVID-19, compared to members of predominantly white and/or affluent communities. [ 1085 ] Unfortunately, the data necessary to detect and respond to these disparities were not consistently available from core data sources, including hospitalization data reported by hospitals and CAHs under existing requirements §§ 482.42(e) and (f); and 485.640(d) and (e), respectively.
We emphasized our commitment to protecting patients from all communities and preventing inequities caused or exacerbated by respiratory viruses like COVID-19, influenza, and RSV and how timely, complete data on racial and ethnic differences in hospitalizations are critical to meeting such a commitment in policy solutions. For that reason, we sought public comment on expanding the scope of demographic information collection to further support improvements in clinical outcomes while also protecting privacy and the safety of demographic groups.
Specifically, we invited comment as to whether race/ethnicity demographic information should be explicitly included as part of requirements for ongoing reporting beginning on the effective date of the final rule. We indicated our particular interest in comments that address the ways these additional data elements could be used to better protect patient and community health and safety both during and outside of a declared PHE and how to protect patient privacy within demographic groups, while being sure not to also stigmatize demographic groups.
Comment: Commenters requested further clarification and more detailed regulations to ensure that hospital and CAH data collections contribute to higher quality health care and do so at a lower cost to providers. Commenters requested clarity regarding the data elements required for reporting, including the respiratory pathogens, limited demographic information, hospital census and capacity data, as well as how CMS intends to use the data to ensure hospitals have enough resources for compliance. A commenter recommended revising the ongoing data collection requirements by collecting both adult and pediatric data, collecting the census of staffed beds, and reporting the level of care and equipment available. Some commenters recommended expanding reporting to all communicable diseases that might improve public health information available to the public and those working to ensure public health, while others limited their suggestions to expanding reporting to address H5N1 as well as “hospital acquired infections.” Some commenters also questioned the need for reporting hospital bed capacity data outside of a PHE. For instance, a commenter mentioned that hospital bed census and capacity is reported yearly in the annual NHSN survey. The commenter mentioned hospital bed capacity likely does not change drastically week to week, therefore this data element is excessive. Some commenters suggested that other surveillance indicators such as wastewater surveillance data or data from public health lab testing could be used instead. A commenter stated that respiratory virus hospitalizations “severely lag” community transmission.
Response: The proposed reporting requirements were written in a manner that would allow for maximum flexibility by covering a broad array of services and entities. In developing the proposed requirements, we considered the data elements that proved most actionable and informative over the course of the COVID-19 PHE with evidence of protecting health and safety, as well as more recent lessons that have emerged during the 2023-2024 respiratory virus response. We also considered ways to balance the burden of reporting on hospitals and CAHs with the need to maintain a level of situational awareness that benefits the patients and communities served by hospitals, as well benefitting hospitals directly. We believe that reporting all communicable disease and hospital acquired infections would be overly burdensome for providers. Likewise, reporting related to H5N1 on a routine basis would also be unduly burdensome at this time.
Respiratory virus surveillance is inherently multi-faceted, and other sources of data may be important adjuncts. However, they have their own significant limitations. For example, wastewater surveillance does not cover all jurisdictions. One of the key advantages of the proposed hospital-based surveillance is that it would provide data that are nationally comprehensive and can could be resolved at the sub-state level (for example, by health service area). A published CDC analysis found that COVID-19—associated hospital admissions lagged reported case incidence by one day and test positivity and ED visits by four days. When considered over the scale of epidemic waves that last for weeks or months, an indicator that lags by less than a week can be a valuable source of actionable information to inform infection control decisions within hospitals. [ 1086 ] In addition, per the bed capacity data elements, CDC is funding ($24.9 million from the Epidemiology and Laboratory Capacity Program) over 19 States to automate bed capacity reporting to NHSN via a State repository. [ 1087 ] The long-term goal of this project is to develop a fully automated approach that standardizes data elements.
Comment: Commenters asked for clarity on how hospitals should collect, format, and submit the requested data. Many commenters urged CMS to establish a modern automated, standardized reporting and collection system for providers and to partner with CDC, Administration for Strategic Preparedness and Response (ASPR), and the Office of the National Coordinator for Health IT (ONC). Such a platform would provide a single place where all the proposed data elements could be captured. Commenters mentioned Trusted Exchange Framework and Common Agreement (TEFCA), CDC's National Syndromic Surveillance Program (NSSP), and NHSN, and standards which could facilitate transfer with such a platform including Fast Healthcare Interoperability Resources (FHIR), United States Core Data for Interoperability (USCDI), USCDI Plus (USCDI+). In addition, commenters mentioned that CMS should establish a certification criterion for public health technologies used by Public Health Agencies (PHA).
Response: We appreciate the support of our goal of transitioning to, and using, more modern, flexible approaches and networks that support data exchange between and across ( print page 69887) public health and healthcare institutions to modernize the public health information infrastructure. We agree that easily adoptable universal standards are necessary for accurate data reporting and would reduce burden. We appreciate commenters support of TEFCA, FHIR, and USCDI/USCDI+. We agree that establishment of certification criteria for public health technologies used by PHAs would improve bi-directional exchange and help improve the quality, timeliness, and completeness of public health reporting. We refer readers to the Health Data, Technology, and Interoperability: Patient Engagement, Information Sharing, and Public Health Interoperability (HTI-2) Proposed Rule, in which ONC has proposed to establish certification criteria for health IT used by public health. [ 1088 ]
Through its data modernization efforts, CDC is working to strengthen public health digital infrastructure. These efforts include investments in core data sources and systems that provide actionable data for facilities, health systems, and response agencies. NHSN is actively engaged in developing approaches to data collection that can be automated and thus reduce the manual burden of reporting by healthcare facilities. NHSN is piloting automated versions of the respiratory virus data collection that, if successful, could be used during the 2025-2026 respiratory season. [ 1089 ] CDC has several pilot sites enrolling through the NHSN with the NHSNCoLab, a collaborative partnership that allows facilities to send automated data flows to NHSN via FHIR and other digital approaches. [ 1090 ] These pilot sites are ready to test out this automated activity that is designed to reduce burden. NSSP is also actively working to expand existing data flows collecting emergency department visits to include inpatient hospitalizations and direct admissions, capitalizing on the automated nature of admit, discharge, and transfer (ADT) messaging to improve public health understanding of hospitalizations. To further reduce burden, CMS will work with the CDC to ensure hospitals can continue to use existing, established systems to report data in the interim. The CDC will continue increasing the automation capabilities of the surveillance systems like NHSN and NSSP and their abilities to connect with other data submission techniques, vendors, and systems. The CDC, CMS, and ASPR are also working with ONC, jurisdictions, health information technology (health IT) vendors, hospitals and CAHs, and other public and private partners to establish national standards and interoperability requirements that reduce burden and promote standardization. We aim to build an infrastructure to create more automated, efficient, timely, and less burdensome processes for data reporting.
Comment: A significant number of commenters indicated that the reporting requirements were too burdensome, time consuming, and duplicative. Some commenters noted that the burden on hospitals and health systems may outweigh the benefits of mandatory reporting. Commenters were concerned that this type of reporting was resource intensive and would require hospitals to implement reporting processes and systems. These groups mentioned the increased paperwork burden that would be placed on staff to comply with reporting requirements noting that increased staff would be necessary to comply with all the steps of data collection, verification, and submission, while hospitals are currently facing staffing challenges.
Some commenters specifically highlighted the potential burden on rural facilities, citing the need for significant onboarding, capacity-building, technical assistance, and the lack of existing public health relationships. Commenters note that rural facilities may not have sufficient informatics support of their own and might not likely be able to find enough funding or workforce to support such reporting. Many commenters suggested financial support would be necessary to alleviate some burden.
In addition, some commenters mentioned that infection data may be already reported through other mandatory mechanisms, including manual case reporting, electronic case reporting (eCR), and NSSP. A commenter expressed concern that the same data reporting requirements were being required from multiple HHS agencies. Commenters suggested that CMS would have to allocate resources to hospitals to build capacity to report COVID cases should the CoP be finalized.
Response: We acknowledge the potential burden of our proposed reporting requirements, especially for rural and small facilities. We understand that some commenters felt these data were duplicative of other reporting measures or that the CoPs were an inappropriate method to implement data reporting requirements, and this could place unnecessary burdens on hospitals. Federal reporting requirements are used by State and local authorities to inform their operations and response for their particular populations. Due to the variation in mandates across States and localities, we will continue to require surveillance efforts at the Federal level and maintain the reporting requirements in more or less the form in use up through April 30, 2024.
CMS, CDC, and ASPR will work with hospitals, health systems, and State, territorial, local and Tribal agencies (STLTs) to streamline this Federal, State, and local reporting burden, utilizing the least burdensome technical exchange mechanism for reporting. CDC and ASPR, together with ONC, would also take steps to encourage State, local, jurisdictional partners to utilize HHS-adopted health IT standards such as USCDI that are already supported by existing systems for data exchange, which would further reduce burden on health care systems. We will also explore where guidance could leverage data sets being developed under the USCDI+ initiative, which focuses on develop and advancing use of standardized data elements for exchange for additional use cases that build on the USCDI.
Comment: Many commenters suggested ways to revise the proposed regulations in general. Commenters suggested that CMS ask each State, territory, or regional jurisdiction—but not individual hospitals—to voluntarily submit a retrospective file that covers the gap period between May 1 and September 30, 2024, when possible, to gain a better understanding of the viruses and have a complete data set. Many commenters also suggested adding COVID to the list of measures utilized by the Hospital Acquired Condition (HAC) Reduction Program and defining the measure “hospital-onset COVID” as infection within five days after admissions, opposed to the current fourteen days, because current variants only take two to three days from exposure to develop symptoms and the average hospital stay is 5.4 days. Other commenters provided recommendations for measures to improve hospital infection prevention, including mandatory staff masking, use of air purifiers, and use of UVC lights. Another comment suggested that all healthcare providers that see patients in primary care and medical practices settings should be required to report all ( print page 69888) infectious diseases within one week of a patient visit.
Response: We appreciate the suggestion to ask for voluntary retrospective data from each State, territory, or regional jurisdiction, however we note that many individual facilities have already been voluntarily submitting many of these data elements. We appreciate the suggestion to add COVID to the HAC Reduction program, defining hospital-onset COVID and specific infection control measures, however the suggestions are outside the scope of the CoPs. We appreciate the suggestion to require medical practices to report all infectious diseases, however this is also outside the scope of this rule.
Comment: A commenter expressed concerns that this requirement would be extremely burdensome for laboratories and strongly recommended delaying any reporting requirements for respiratory diseases during a non-PHE time until a clear process that leveraged existing EHR systems was in place to minimize the burden on laboratories and eliminate the need for laboratories to duplicate reporting to multiple agencies.
Response: We appreciate the commenter's feedback regarding the downstream impact on laboratories associated with requirements for hospital and CAHs to report data on respiratory illnesses. However, the CoPs set forth in this rule apply to hospitals and CAHs and do not apply to laboratories. While there are hospital (§ 483.27) and CAH (§ 485.635(b)(2)) CoPs that require hospitals and CAHs to have adequate laboratory services to meet the needs of their patients in accordance with the Clinical Laboratory Improvement Amendments (CLIA) program ( 42 CFR part 493 ), the requirements finalized in this rule are specific to reporting activities and do not address agreements that hospitals and CAHs must have with laboratories (either directly or through contracts) to address laboratory services and testing. Thus, this comment is outside the scope of this rule.
Comment: We received many comments advocating for ongoing data submissions to be more frequent than once a week, including daily. Those that fully support the proposed rule agree with continued ongoing weekly reporting. However, we also received many comments that suggested that facilities provide a snapshot of data from one day per week, which would reduce administrative burden compared to daily data for all days reported once per week and/or cumulative weekly totals. Many comments discussed the tradeoff between data granularity and burden on hospitals to comply.
Response: We appreciate the feedback that we received and the numerous suggestions of ways to revise reporting frequency. To clarify, weekly reporting would encapsulate daily data, but the facility would submit it once a week. While commenters had mixed response to weekly data reporting, the majority were in support of this frequency. Sustained data collection and reporting outside of emergencies would help ensure that hospitals and CAHs maintain a functional reporting capacity that could be mobilized quickly when a new threat emerges to inform and direct response efforts (for example, resource allocations or patient load balancing within and across facilities) that protect patients and their communities. It would also provide the baseline data necessary to forecast, detect, quantify and, ultimately, direct responses to signals of strain.
Some commenters also indicated that weekly reporting should be limited to a one-day-a-week snapshot, rather than aggregate totals, as means of further reducing reporting burden. We agree that following this approach makes sense in cases where the resulting data are still sufficiently useful to justify reporting. Taking into consideration the need to limit overall reporting burden and the specific uses of data elements, we identified as many data elements as possible for which a “one-day-a-week” snapshot would be acceptable. These included bed census and capacity data, as well as total confirmed existing infections of respiratory illnesses, including COVID-19, influenza, and RSV, among hospitalized patients.
However, a one-day-a-week snapshot for newly admitted patients with confirmed infections of respiratory illnesses, including COVID-19, influenza, and RSV, would significantly degrade the utility of reported data for patient and public health and safety applications. Accordingly, we are finalizing our proposal to require reporting of a limited set of respiratory disease and hospital capacity data on a weekly basis. We are further clarifying that new admissions of patients with confirmed respiratory illnesses, including COVID-19, influenza, and RSV by age group, would be reported as weekly totals. However, the other data elements, such as staffed bed capacity and occupancy, prevalence of hospitalizations and ICU patients by respiratory illnesses, required under this provision would be reported as one-day-a-week snapshots. We believe our approach of totals where most important/impactful, and snapshots where feasible, strikes an appropriate balance between value/burden, particularly since the overall impact of shifting to weekly totals and snapshots reporting already represents a significant reduction in burden relative to the proposed rule, which involved reporting daily totals on a weekly basis. These variables and reporting methodology will be part of the revised CDC information collection National Healthcare Safety Network (NHSN) Surveillance in Healthcare Facilities (OMB 0920-1317) or, as designated by the Secretary, other CDC systems and corresponding approved information collection packages. Changes may be made over time based on patient and population health needs and technology advances, but for FY 2025, the information collection will include:

Comment: Many commenters submitted suggestions related to the solicitation of public comments on the collection of demographic information. Most commenters agree that there are benefits to collecting race and ethnicity data to better address disparities in public health response. However, commenters note that some patients may be less willing to share additional demographic information, such as their socioeconomic or disability status, therefore there is a tradeoff between honoring patient choices and having a complete data set. A commenter noted ( print page 69889) that demographic factors such as socioeconomic or disability status are too burdensome and very subjective. Commenters also expressed concern about the consistency of the data set. Since there are not uniform requirements across States in terms of what questions are asked and how they are asked, the response options available, and how and when data is collected, comments stressed the importance of creating definitions and categories so that there is a standard classification for the entire nation. Some commenters had suggestions on appropriate timelines for future demographic reporting. For example, commenters suggested waiting to collect demographic data until finalization of the Office of Management and Budget Revised Statistical Policy Directive No. 15 (SPD-15), updated March 28, 2024, which would govern how federal agencies collect and use race and ethnicity data in their programs.
Some commenters weighed the pros and cons of aggregate versus patient-level data. For instance, age should be collected only in categories rather than each patient's exact age. A commenter noted the importance of protecting patient confidentiality in hospitals where there might be small numbers of a particular race or ethnicity. Lastly, some commenters highlighted how reporting on these additional data elements would increase burden. Specifically, many noted that all electronic health records are not prepared to collect and exchange data on disability and health related social needs in a standardized manner. While some larger facilities may already collect these data, smaller facilities that manually report data would have a large burden.
Response: We thank commenters for the information and perspectives that they provided on expanding the collection and submission of demographic data. While we are not expanding the collection of demographic data at this time due to the need to further refine this concept and the need to begin data collection by November 1, 2024, we acknowledge that not collecting this data would represent a gap in epidemiological information. We believe that demographic data plays an important role in informing healthcare decisions that ultimately impact the health and safety of patients. We intend to continue exploring ways to facilitate the collection of additional demographic data and close this gap in the future.
Final Rule Action: We are finalizing our proposal to require ongoing respiratory illness reporting in a modified form as proposed. Hospitals and CAHs, in a standardized format and frequency specified by the Secretary, must electronically report data related to COVID-19, influenza, and RSV including confirmed infections of respiratory illnesses among hospitalized patients, hospital bed census and capacity (both overall and by hospital setting and population group [adult or pediatric]), and limited patient demographic information, including age. Beginning November 1, 2024, hospitals and CAHs must electronically report this information to CDC's NHSN or other CDC-owned or CDC-supported system, as determined by the Secretary.
We proposed in the NPRM that during a declared federal, state, or local PHE for an acute infectious illness, or if an event was significantly likely to become a PHE for an acute infectious illness, the Secretary could require hospitals to report data up to a daily frequency without going through notice and comment rulemaking. Specifically, we proposed that the Secretary could require the reporting of additional or modified data elements relevant to an acute infectious illness PHE including but not limited to: confirmed infections of the infectious disease, facility structure and infrastructure operational status; hospital/ED diversion status; staffing and staffing shortages; supply inventory shortages (for example, equipment, blood products, gases); medical countermeasures and therapeutics; and additional, demographic factors.
We invited comments on whether, during a PHE, there should be any limits to the data the Secretary could require without notice and comment rulemaking, such as limits on the duration of additional reporting or the scope of the jurisdiction of reporting (that is, state or local PHEs). We also sought comments on whether and how the Secretary should still seek stakeholder feedback on additional elements during a PHE without notice and comment rulemaking and how HHS should notify hospitals of new required acute infectious illness data. We also invited comments on the evidence HHS should provide to demonstrate: (1) that an event is “significantly likely to become a PHE”; or (2) that the increased scope of required data would be used to protect patient and community health and safety. Finally, we invited comment on whether hospitals should be compensated for collecting and reporting these data if the burden reached a certain threshold of cost or time.
Comment: We received mixed responses regarding our proposal for reporting respiratory illness data during a PHE. Some stakeholders noted that the value of reporting respiratory illness data during a PHE outweighed the administrative burden and suggested that there should be incentives for hospitals to provide even more data during a PHE. However, commenters also noted that increased reporting during a PHE would significantly burden hospitals during the most vulnerable and resource-constrained period. Some mentioned that data collection during PHE was especially burdensome without any payoff for hospital facilities since staff would have to spend substantial time trying to oversee data collection while juggling patients and implementing infection control protocols.
In particular, hospital associations shared significant concern regarding the proposed flexibility provided to the Secretary to request increased reporting if there was a “likely threat” of a PHE, especially with the lack of notice and comment rulemaking to define what might rise to the level of a “significantly likely threat”. Commenters questioned CMS's authority, noting there is no legal standard or precedent to support such a requirement and requested CMS provide additional justification. Many commenters urged CMS to withdraw the proposal to adopt increased reporting for events that are “significantly likely” to become PHEs.
Commenters encouraged CMS to work with stakeholders to determine the necessary level of required reporting during a PHE and emphasized that public health organizations should play a role in the decision-making. Lastly, commenters emphasized that, on occasions when PHE reporting was required, the Secretary should be required to provide clear and detailed notification, such as the use of an automated system, noting that regional emergency coordinators could be an appropriate resource to disseminate information.
Response: We understand the need for clarity when PHE-related reporting is required; we acknowledge the efforts that would be required of providers and the strain that the COVID-19 PHE placed on the health care system. The experiences with the COVID-19 PHE and the new “normal” that we currently face with circulating respiratory illnesses that include COVID-19 is why we believe it is imperative to retain respiratory illness data reporting and ensure that facilities are informed and ( print page 69890) prepared in the event of another PHE. Routinely collected data from hospitals power forecasts that inform decision making during an emergency response.
In the face of future illness emergencies, we anticipate stakeholders—including health care systems—will continue to need data on how respiratory illnesses are affecting and burdening the health care system. Better understanding anticipated impacts empower hospitals and CAHs, health systems, and jurisdictions to take steps that protect patient safety and health care system capacity in the face of surges in respiratory virus cases, including low-probability, high-impact events such as pandemics that pose catastrophic risks to patient safety and the health care system. These include facility-initiated actions, such as delaying elective procedures or activating contracts for additional surge staffing support, as well as jurisdiction or federal-level actions to mobilize supplies, staffing, or other forms of support. Collaborations during the COVID-19 pandemic demonstrated the value of bringing together analysts, public health officials, and health care practitioners and leaders to use advanced analytics to guide emergency response, and data from hospitals were central to some of these efforts. The federal government has made significant investments to consolidate these gains and develop response-ready analytic tools that work at scale to meet the needs of the health care and public health systems. As part of CDC's recent reorganization, the Inform and Disseminate Division within the Office of Public Health Data, Surveillance, and Technology uses human-centered design to develop systems and products that effectively communicate public health data. CDC's Center for Forecasting and Outbreak Analytics uses hospital data to generate, evaluate, and continually refine analytic products such as real-time estimates of transmission rates, short-term forecasts, and scenario models that evaluate the impact of vaccines and other interventions. The Center also provides financial and technical support to state and local public health agencies, the healthcare sector, and academic collaborators to develop analytic tools that support decision making to combat infectious disease threats.
We acknowledge concerns about potential elasticity of proposed data categories, particularly those outlined for enhanced reporting during a PHE. In the event of a PHE, HHS will provide proper notification to hospitals and CAHs to activate increased PHE reporting and indicate the frequency and required additional elements that are necessary for reporting based on the specific circumstances at the time. We expect to use a communication mechanism, such a Quality Safety and Oversight Memo, that is readily available to the public, nationally accessible, and familiar to stakeholders, to ensure clarity and access to necessary information. Through the identified mechanism, the Secretary will provide clear, standardized definitions for any data to be reported, as well as instructions and the effective data for the PHE specific reporting. We also reiterate that the data elements proposed for PHE reporting will represent our best effort in identifying those categories that will be required for additional reporting. We note that these data elements, supply inventory shortages; staffing shortages; relevant medical countermeasures and therapeutic inventories, usage, or both; and facility structure and operating status, including hospital/ED diversion status, have all previously been reported on by hospitals and we expect that hospitals will be aware and prepared in the event they are required to again report this information during a PHE.
We recognize the concerns raised regarding the proposal to also require increased PHE reporting if a “likely threat” of a PHE exists. We also anticipate that the ongoing reporting requirements established in this rule will be sufficient to assure patient health and safety in times when the threat of a PHE is significantly likely. Therefore, in response to the concerns raised we are withdrawing the proposal that the Secretary may require increased reporting if the threat of a PHE is significantly likely. The Secretary may require increased reporting in the event of a future PHE declaration for a respiratory illness. We believe the benefits of data collection are necessary to protect patient safety and inform public health decisions and public policy and expect hospitals to consider the need to ramp up reporting during a future PHE. We encourage hospitals to utilize their required emergency preparedness plans and policies and procedures to promote readiness and actions that could reduce burden during a resource intense time (that is, during a PHE).
Final rule Action: We are finalizing as proposed our proposal to require additional reporting during a declared federal, state, or local PHE for an acute infectious illness. We have withdrawn our proposal to require additional reporting if the Secretary determines that an event is “significantly likely” to become a PHE for an infectious disease. During a declared federal, state, or local PHE for an acute infectious illness the Secretary may require reporting of data elements relevant to confirmed infections of the acute infectious illness, facility structure and infrastructure operational status, hospital/ED diversion status, staffing and staffing shortages, supply inventory shortages (for example, equipment, blood products, gases), medical countermeasures and therapeutics, and additional demographic factors.
In the proposed rule, we included a RFI on health care reporting to the CDC's National Syndromic Surveillance Program (NSSP), which is a collaboration among CDC, other federal agencies, local and state health departments, and academic and private sector partners who have formed a Community of Practice to collect, analyze, and share electronic patient encounter data received from emergency departments, urgent and ambulatory care centers, inpatient health care settings, and laboratories.
We emphasized that Syndromic surveillance is not a part of any condition of participation under this program, however the continued growth of national syndromic surveillance would benefit hospitals, health care, and public health. The goal of the RFI was to gather feedback to better understand what else could be done to ensure that this effort could continue to make progress and that this critical data source is available at all levels of public health to support health care preparedness, public health readiness, and responsiveness to existing and emerging health threats. We sought input on the following:
- How could CMS further advance hospital and CAH participation in CDC's NSSP?
- Should CMS require hospitals and CAHs to report data to CDC's NSSP, whether as a condition of participation or as a modification to current requirements under the Promoting Interoperability Program?
- Should CMS explore other incentive or existing quality and reporting programs to collect this information?
- What would be the potential burden for facilities in creating these connections in state and local public health jurisdictions that have not yet established syndromic programs and/or where state and local public health are not presently exchanging data with CDC's NSSP? Are there unique ( print page 69891) challenges in rural areas that CMS should take into consideration?
- Data reported as part of syndromic surveillance requirements could serve as an alternative source for the COVID-19, influenza, and RSV hospitalization reporting requirements proposed in this rule—and even support eventual evolution towards an all-hazards approach for monitoring inpatient hospitalizations for conditions of public health significance. Should CMS consider a future requirement or otherwise incentivize facilities to expand ADT-based reporting currently provided for emergency department visits to include data collected from inpatient settings as defined in the HHS COVID-19 reporting guidance, [ 1091 ] or a subset of these? If the latter, should a subset of inpatient locations be subject to such a requirement? What would be the potential value and burden trade-offs to facilities? And should any requirement specify that reporting also be to CDC's NSSP (in addition to more general reporting to state/local syndromic surveillance systems? (noting that often the reporting to CDC's NSSP happens through a given state/local system and that applicable law may apply).
- How could CMS leverage its authorities and programs to improve the quality of data reported to CDC's NSSP, especially for key elements that are sometimes incomplete, including discharge diagnoses, discharge disposition, and patient class? [ 1092 ]
- In addition to its value for public health, how could CDC's NSSP serve as a tool to directly improve clinical practice, patient safety, and overall situational awareness? What types of questions would you like the system to help answer?
Comment: We received varied responses to this comment solicitation. A commenter reminded us that NSSP is already a measure under the Public Health and Clinical Data Exchange objectives in the Promoting Interoperability Program, therefore introducing an unnecessary duplication in reporting by requiring hospitals to submit the same data to NSSP that is already being reported to State. Another commenter suggested that the CDC's NSSP could serve as, “a valuable tool to directly improve clinical practice, patient safety, and overall situational awareness by leveraging aggregated data for early warning signals without necessitating extensive line-level data sharing.” To further advance hospital and CAH participation in the NSSP a commenter recommended clearly defining reporting requirements specific to applicable care settings, such as emergency or inpatient settings. To address the potential burden for facilities in creating connections in state and local public health jurisdictions that have not yet established syndromic programs or where data exchange with CDC's NSSP is not currently happening, a commenter suggested (1) using standardized formats and vocabulary to accelerate adoption and simplify implementation and configuration efforts for both IT developers and healthcare providers (2) using a single submission point to submit a report, which could then be distributed to the appropriate jurisdictions in either identified or de-identified formats to improve efficiencies; and (3) Leveraging TEFCA to work under a single common agreement and trust framework.
Response: We appreciate the comments that we received on this subject and will use them to further inform our understanding of advancing hospital and CAH participation in the NSSP. We appreciate that hospital and CAH attestation to participate in syndromic surveillance reporting is a required measure under the Promoting Interoperability Program's Public Health and Clinical Data Exchange Objective. However, as noted in the proposed rule, inclusion in that program, while a valuable incentive, has not been sufficient to close the current participation gap. As also noted in the proposed rule, CMS is not proposing a new CoP specific to ED reporting at this time. However, we disagree that any such measure, if introduced, would result is significant duplicative reporting, as states could submit data on hospitals' behalf to CDC as many already do voluntary. CDC is in the early stages of a project, through NSSP, to expand existing data flows collecting ED visits from admit, discharge and transfer (ADT) messaging to include inpatient hospitalizations and direct admits. This effort will capitalize on the fully automated nature of this data exchange to improve CDC and STLT understanding of hospitalizations across all hazards while minimizing the additional burden of collection required for these data. Key principles of this effort include (1) improving upon processes initiated early in the COVID-19 pandemic by facilitating improvements in automation; (2) promoting efficiency by re-using, where possible, existing infrastructure and processes such as ADT messaging systems within healthcare, NSSP, and jurisdictional ED data pipelines; (3) and building upon fully automated processes that are maintained for coordinating patient care, such that once configured, minimal additional resources are needed for long term sustainment of the data exchange with public health.
Under section 1886(e)(4)(B) of the Act, the Secretary must consider MedPAC's recommendations regarding hospital inpatient payments. Under section 1886(e)(5) of the Act, the Secretary must publish in the annual proposed and final IPPS rules the Secretary's recommendations regarding MedPAC's recommendations. We have reviewed MedPAC's March 2024 “Report to the Congress: Medicare Payment Policy” and have given the recommendations in the report consideration in conjunction with the policies set forth in this final rule. MedPAC recommendations for the IPPS for FY 2025 are addressed in Appendix B to this final rule.
For further information relating specifically to the MedPAC reports or to obtain a copy of the reports, contact MedPAC at (202) 653-7226, or visit MedPAC's website at https://www.medpac.gov .
IPPS-related data are available on the internet for public use. The data can be found on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index . We listed the data files available in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36509 through 36510 ).
Commenters interested in discussing any data files used in construction of this final rule should contact Michael Treitel at (410) 786-4552.
Under the Paperwork Reduction Act (PRA) of 1995, we are required to provide 60-day notice in the Federal Register and solicit public comment before a collection of information requirement is submitted to the Office of Management and Budget (OMB) for review and approval. In order to fairly evaluate whether an information collection should be approved by OMB, ( print page 69892) section 3506(c)(2)(A) of the PRA of 1995 requires that we solicit comment on the following issues:
- The need for the information collection and its usefulness in carrying out the proper functions of our agency.
- The accuracy of our estimate of the information collection burden.
- The quality, utility, and clarity of the information to be collected.
- Recommendations to minimize the information collection burden on the affected public, including automated collection techniques.
In the proposed rule, we solicited public comment on each of these issues for the following sections of this document that contain information collection requirements (ICRs). The following ICRs are listed in the order of appearance within the preamble (see sections II. through X. of the preamble of this final rule).
As discussed in section V.F.2. of the preamble of this final rule, teaching hospitals would be able to submit electronic applications to CMS for resident slot increase requests under section 4122 of the Consolidated Appropriations Act (CAA), 2023. The burden associated with these requests will be captured under OMB control number 0938-1417 (expiration date March 31, 2025), currently approved for CMS to receive electronic applications for Medicare-funded GME Residency Positions submitted in accordance with Section 126 of the CAA, 2021. For that information collection, we estimated each eligible hospital (1,325 hospitals) would require 8 hours per eligible hospital annually to gather appropriate documentation, prepare and submit an application for a total burden of 10,600 hours (8 hours × 1,325 hospitals). The most recent data from the BLS reflects a mean salary for legal secretaries and administrative assistants of $26.05. [ 1093 ] With the fringe benefits included the salary is $52.10 ($26.05 × 2). The total cost related to this information collection is approximately $416.80 per eligible hospital per year ($52.10 × 8.0 hours per hospital). The total estimated burden is $552,260 ($52.10 × 10,600 hours). As a result of the final policies, for FY 2026, if an eligible hospital submits an electronic application to CMS for section 126 of the CAA, 2021 or for section 4122 of the CAA, 2023, the total annual burden remains the same. However, if an eligible hospital submits an electronic application to CMS for both section 126 of the CAA, 2021, and section 4122 of the CAA, 2023, we estimated that the new total annual burden to be 16 hours per eligible hospital. We estimated the adjustment in the number of hours from 8 hours to 16 hours, results in 21,200 hours (16 hours × 1,325 hospitals) at a cost of $1,104,520 ($52.10 × 21,200 hours) for FY 2026 only. We will submit the revised information collection request to OMB for approval under OMB control number 0938-1417 (expiration date March 31, 2025).
We received no comments on this proposal and therefore are finalizing this provision without modification.
In section V.J. of the preamble of this final rule, we finalized, for cost reporting periods beginning on or after October 1, 2024, a separate payment under IPPS to small, independent hospitals for establishing and maintaining access to buffer stocks of essential medicines to foster a more reliable, resilient supply of these medicines for these hospitals. The payment adjustments will be based on the reasonable cost incurred by the hospital for establishing and maintaining access to a 6-month buffer stock of one or more essential medicines during the cost reporting period. In order to calculate the essential medicines payment adjustment for each eligible cost reporting period, we will create a new supplemental cost reporting form that will collect the additional information from hospitals.
Specifically, the new cost reporting worksheet will collect the costs of a hospital that voluntarily requests separate payment under this policy for the costs associated with establishing and maintaining access to its buffer stock of one or more essential medicines. This new information will include the costs associated with contractual arrangements to establish and maintain access to buffer stock(s) of essential medicine(s) as well as the costs associated with directly establishing and maintaining buffer stock(s) of essential medicine(s) such as (but not limited to) utilities like cold chain storage and heating, ventilation, and air conditioning, warehouse space, refrigeration, management of stock including stock rotation, managing expiration dates, and managing recalls, administrative costs related to contracting and record-keeping, and dedicated staff for maintaining the buffer stock(s). This information will be used, along with other information already collected on the Hospitals and Health Care Complex Cost Report (Form CMS-2552-10) approved under OMB control number 0938-0050, to calculate the IPPS payment adjustment amount. This new cost report worksheet may be submitted by a provider of service as part of the annual filing of the cost report and the provider should make available to its contractor and CMS, documentation to substantiate the data included on this Medicare cost report worksheet. The documentation requirements are based on the recordkeeping requirements at current § 413.20, which require providers of services to maintain sufficient financial records and statistical data for proper determination of costs payable under Medicare.
The burden associated with filling out this new essential medicine cost report worksheet would be the time and effort necessary for the provider to locate and obtain the relevant supporting documentation to report the costs of a hospital to establish and maintain access to its buffer stock for the cost reporting period. We estimated the number of respondents to be approximately 500. This number is comprised of Medicare certified section 1886(d) hospitals that are small, independent hospitals that would be eligible for the payment adjustment. We estimated the average burden hours per facility to be 1.0 hour. This breaks down to approximately 0.4 hours per provider for recordkeeping, which includes a 0.10-hour burden associated with monitoring the quarterly communication from CMS regarding updates to the list of essential medicines that are considered to be in shortage for purposes of this policy, beginning when the hospital elects to establish a buffer stock of an essential medicine and when the hospital is not able to maintain a previously established 6 month buffer stock of an essential medicine. We estimated 0.6 hours per provider for obtaining and analyzing the data and reporting. We recognize this average varies depending on the provider size and complexity. In addition to general comment on this burden estimate, we specifically sought feedback on the burden estimate that is associated with monitoring the FDA shortage list as described. We stated that CMS would conduct provider education regarding ( print page 69893) additions and deletions to the publicly available FDA Drug Shortages Database to assist hospitals with the finalized policy.
We estimated the associated labor costs as follows. As explained earlier, the estimate of 0.4 hour is required for recordkeeping including time for bookkeeping activities. Based on the most recent data published by Bureau of Labor Statistics (BLS) in its 2022 Occupation Employment and Wage Statistics Program, the mean hourly wage for Bookkeeping, Accounting, and Auditing Clerks (Category 43-3031) is $22.81. We added 100 percent of the mean hourly wage to account for fringe and overhead benefits, which calculates to $45.62 ($22.81 + $22.81) and multiplied it by 0.4 hour, to determine the annual recordkeeping costs per hospital to be $18.25 ($45.62 per hour multiplied by 0.4 hour). The estimated 0.6 hours for reporting include time for accounting and audit professionals' activities. The mean hourly wage for Accountants and Auditors (Category 13-2011) is $41.70. We added 100 percent of the mean hourly wage to account for fringe and overhead benefits, which calculates to $83.40 ($41.70 plus $41.70) and multiplied it by 0.6 hour, to determine the annual reporting costs per hospital to be $50.04 ($83.40 per hour multiplied by 0.6 hour). We calculated the total average annual cost per hospital of $68.29 by adding the recordkeeping costs (which includes monitoring the quarterly communication from CMS regarding updates to the list of essential medicines) of $18.25 plus the reporting costs of $50.04. We estimated the total annual cost to be $34,145 ($68.29 cost per hospital multiplied by 500 hospitals). We sought comment on our estimates and cost of recordkeeping and oversight.
We did not receive any comments specific to our ICR for payment adjustments for establishing and maintaining access to essential medicines. We did, however, receive comments on the general administrative burden that eligible hospitals who voluntarily participate in this finalized policy are anticipated to experience. These comments are summarized in the V.J. pages of the preamble of this final rule.
We are not finalizing any changes to the Hospital Readmissions Reduction Program for FY 2025. All six of the current Hospital Readmissions Reduction Program's measures are claims-based measures. We believe that continuing to use these claims-based measures will not create or reduce any information collection burden for hospitals because they will continue to be collected using Medicare FFS claims that hospitals are already submitting to the Medicare program for payment purposes.
In section IX.B.2. of the preamble of this final rule, we discuss our updates to the Hospital VBP Program. Specifically, we are adopting an updated version of the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) Survey measure beginning with the FY 2030 program year to align with the adoption of the updated measure in the Hospital IQR Program beginning with the CY 2025 reporting period/FY 2027 payment determination. The updated HCAHPS Survey measure in the Hospital VBP Program adds three new survey dimensions, removes one existing survey dimension, and modifies one existing survey dimension. We are also modifying scoring of the HCAHPS Survey measure in the Hospital VBP Program for the FY 2027 to FY 2029 program years to only score on the six unchanged dimensions of the survey while the updates to the survey are adopted and publicly reported on in the Hospital IQR Program. In addition, we are modifying scoring on the HCAHPS Survey measure beginning with the FY 2030 program year to account for the updated measure.
Data collections for the Hospital VBP Program are associated with the Hospital IQR Program under OMB control number 0938-1022 (expiration date January 31, 2026), the National Healthcare Safety Network under OMB control number 0920-0666 (expiration date December 31, 2026), and the HCAHPS Survey under OMB control number 0938-0981 (expiration date January 31, 2025). The Hospital VBP Program will use data that are also used to calculate quality measures in these programs and Medicare FFS claims data that hospitals are already submitting to CMS for payment purposes, therefore, the program does not estimate any additional change in burden associated with these finalized updates outside of the burden that is associated with collecting that data under the Hospital IQR Program. There is also no estimated change in burden related to the finalized scoring methodology modification because the policy does not require hospitals to submit any additional information specific to the Hospital VBP Program but instead will change how hospitals are scored based on the information already being submitted under the Hospital IQR Program.
We discuss the burden associated with the adoption of the updated HCAHPS Survey measure under the Hospital IQR Program in section XII.B.6. of the preamble of this final rule. We note that respondents will only complete the HCAHPS Survey once for use in both programs, so there is no additional information collection burden for the Hospital VBP Program.
We summarized comments on the information collection burden estimates associated with the adoption of the updates to the HCAHPS Survey measure in section IX.B.2. of this final rule.
OMB has currently approved 28,800 hours of burden and approximately $1.2 million under OMB control number 0938-1352 (expiration date November 30, 2025), accounting for information collection burden experienced by the 400 subsection (d) hospitals selected for validation each year in the HAC Reduction Program.
As discussed in section V.M. of the preamble of this final rule above, we are not adding or removing any measures from the HAC Reduction Program.
Data collections for the Hospital IQR Program are associated with OMB control number 0938-1022. OMB has currently approved 2,286,977 hours of burden at a cost of approximately $80.3 million under OMB control number 0938-1022 (expiration date January 31, 2026), accounting for information collection burden experienced by approximately 3,150 IPPS hospitals and 1,350 non-IPPS hospitals for the FY 2026 payment determination. In this final rule, we describe the burden changes regarding collection of information, under OMB control number 0938-1022, for IPPS hospitals.
For more detailed information on what requirements we are changing for the Hospital IQR Program, we refer readers to sections IX.B.1., IX.B.2., and IX.C. of the preamble of this final rule. We are adopting seven new measures: (1) Age-Friendly Hospital measure beginning with the CY 2025 reporting period/FY 2027 payment determination; (2) Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 payment determination, with modifications; (3) Catheter- ( print page 69894) Associated Urinary Tract Infection (CAUTI) Standardized Infection Ratio Stratified for Oncology Locations measure beginning with the CY 2026 reporting period/FY 2028 payment determination; (4) Central Line-Associated Bloodstream Infection (CLABSI) Standardized Infection Ratio Stratified for Oncology Locations measure beginning with the CY 2026 reporting period/FY 2028 payment determination; (5) Hospital Harm—Falls with Injury electronic clinical quality measure (eCQM) beginning with the CY 2026 reporting period/FY 2028 payment determination; (6) Hospital Harm—Postoperative Respiratory Failure eCQM beginning with the CY 2026 reporting period/FY 2028 payment determination; and (7) Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) measure beginning with the July 1, 2023—June 30, 2025 reporting period/FY 2027 payment determination. We are refining two measures: (1) the Global Malnutrition Composite Score eCQM, beginning with the CY 2026 reporting period/FY 2028 payment determination; and (2) the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) Survey measure beginning with the CY 2025 reporting period/FY 2027 payment determination. We are also removing five measures: (1) Death Rate Among Surgical Inpatients with Serious Treatable Complications (CMS PSI-04) measure beginning with the July 1, 2023—June 30, 2025 reporting period/FY 2027 payment determination; (2) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Acute Myocardial Infarction (AMI) measure beginning with the July 1, 2021—June 30, 2024 reporting period/FY 2026 payment determination; (3) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Heart Failure (HF) measure beginning with the July 1, 2021—June 30, 2024 reporting period/FY 2026 payment determination; (4) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Pneumonia (PN) measure beginning with the July 1, 2021—June 30, 2024 reporting period/FY 2026 payment determination; and (5) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Elective Primary Total Hip Arthroplasty (THA) and/or Total Knee Arthroplasty (TKA) measure beginning with the April 1, 2021—March 31, 2024 reporting period/FY 2026 payment determination. We are finalizing a modified version of our proposal to increase the total number of eCQMs that must be reported each year. We are increasing the total number of eCQMs reported from six to eight for the CY 2026 reporting period/FY 2028 payment determination, from eight to nine for the CY 2027 reporting period/FY 2029 payment determination, and then from nine to eleven beginning with the CY 2028 reporting period/FY 2030 payment determination. Lastly, we are updating data validation policies, including updating the scoring methodology for eCQM validation, removing the requirement that hospitals must submit 100 percent of eCQM records to pass validation beginning with CY 2025 eCQM data affecting the FY 2028 payment determination, and no longer requiring hospitals to resubmit medical records as part of their request for reconsideration of validation beginning with CY 2025 discharges affecting the FY 2028 payment determination.
In the FY 2024 IPPS/LTCH PPS final rule, we utilized the median hourly wage rate for Medical Records Specialists, in accordance with the Bureau of Labor Statistics (BLS), to calculate our burden estimates for the Hospital IQR Program ( 88 FR 59312 ). Using the most recent May 2022 National Occupational Employment and Wage Estimates data from the BLS reflects a mean hourly wage of $24.56 per hour for all medical records specialists (SOC 29-2072), however, we are using the mean hourly wage for medical records specialists for the industry, “general medical and surgical hospitals,” which is $26.06. [ 1094 ] We believe the industry of “general medical and surgical hospitals” is more specific to our settings for use in our calculations than other industries that fall under medical records specialists, such as “office of physicians” or “nursing care facilities.” We calculated the cost of overhead, including fringe benefits, at 100 percent of the median hourly wage, consistent with previous years. This is necessarily a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, we believe that doubling the hourly wage rate ($26.06 × 2 = $52.12) to estimate total cost is a reasonably accurate estimation method. Accordingly, unless otherwise specified, we will calculate cost burden to hospitals using a wage plus benefits estimate of $52.12 per hour throughout the discussion in this section of this final rule for the Hospital IQR Program.
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59312 ), our burden estimates were based on an assumption of approximately 3,150 IPPS hospitals. For this final rule, based on data from the FY 2024 Hospital IQR Program payment determination, we are updating our assumption and estimate that approximately 3,050 IPPS hospitals will report data to the Hospital IQR Program for the CY 2025 reporting period and subsequent years, unless otherwise noted.
In section IX.C.5.a. of the preamble of this final rule, we discuss adoption of the Age Friendly Hospital measure beginning with the CY 2025 reporting period/FY 2027 payment determination. Hospitals will submit responses on an annual basis during the submission period through CMS' Hospital Quality Reporting (HQR) System. Specifically, for the Age Friendly Hospital measure, hospitals will be required to attest “yes” or “no” in response to questions across five domains annually for a given reporting period. Similar to the Hospital Commitment to Health Equity measure currently approved under OMB control number 0938-1022, which also requires a “yes” or “no” attestation to questions across five domains, we estimate the information collection burden associated with this measure to be, on average across all 3,050 IPPS hospitals, no more than 10 minutes per hospital per year ( 87 FR 49385 ). Using the estimate of 10 minutes (or 0.167 hour) per hospital per year, we estimate that adoption of this measure will result in a total annual burden increase of 509 hours across all participating IPPS hospitals (0.167 hour × 3,050 IPPS hospitals) at a cost of $26,529 (509 hours × $52.12).
We received no comments on our burden estimates.
In section IX.B.1. of the preamble of this final rule, we are finalizing with modification the Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 payment determination. Hospitals will submit responses on an annual basis ( print page 69895) during the submission period through the Centers for Disease Control and Prevention's (CDC) National Healthcare Safety Network (NHSN), which is a secure, internet-based surveillance system maintained and managed by the CDC that is provided free of charge to providers. To report to the NHSN, hospitals must first agree to the NHSN Agreement to Participate and Consent form, which specifies how NHSN data will be used, including fulfilling CMS's quality measurement reporting requirements for NHSN data. [ 1095 ] Specifically, hospitals will be required to provide responses and attest “yes” or “no” in response to a total of five domains for a given reporting period.
We note that burden estimates under OMB control number 0920-0666 (expiration date December 31, 2026) are calculated based on the CDC's Organization Identification Numbers (OrgIDs). The CDC's OrgID reflects physical locations, meaning that multiple OrgIDs may appear under a single IPPS-related CMS Certification Number, which results in a higher number of potential respondents for burden calculations. Accounting for relevant physical locations, we estimate that 3,900 OrgID locations will submit data to the NHSN for this measure. Similar to the Hospital Commitment to Health Equity measure currently approved under OMB control number 0938-1022, which also requires a “yes” or “no” response to each of five domains, we estimate the information collection burden associated with this measure to be, on average across all 3,900 OrgID locations, no more than 10 minutes per hospital per year. We are modifying the attestation statement in Domain 4 Statement B of the measure to remove the portion of the attestation related to voluntary reporting to the NPSD and focus instead on the beneficial activities possible through engagement with a PSO. Because this does not affect the number of responses each hospital must provide, we are not making a change to our burden assumptions. Using the estimate of 10 minutes (or 0.167 hour) per OrgID location per year, and the updated wage estimate as described previously, we estimate that the adoption of this measure will result in a total annual burden increase of 650 hours across all participating OrgID locations (0.167 hour × 3,900 OrgID locations) at a cost of $33,878 (650 hours × $52.12).,
We discuss the burden associated with the adoption of the Patient Safety Structural measure for the PCHQR Program in section XII.B.7.a. of this final rule. We will work with the CDC to submit the revised information collection estimates for NHSN data collection to OMB for approval under OMB control number 0920-0666. Therefore, we do not anticipate any changes in burden associated with OMB control number 0938-1022.
In section IX.C.5.b. of the preamble of this final rule, we discuss the adoption of two HAI measures beginning with the CY 2026 reporting period/FY 2028 payment determination: (1) the CAUTI Standardized Infection Ratio Stratified for Oncology Locations measure, and (2) the CLABSI Standardized Infection Ratio Stratified for Oncology Locations measure. We will collect data for both measures via the CDC's NHSN. Hospitals will provide data for both measures from their EHRs and report on a quarterly basis. The burden associated with submission of data via the NHSN continues to be accounted for under OMB control number 0920-0666. Therefore, we do not anticipate any changes in burden associated with OMB control number 0938-1022.
We received no comments on our assumptions regarding burden.
In sections IX.C.5.c. and IX.C.5.d of the preamble of this final rule, we are adopting two new eCQMs beginning with the CY 2026 reporting period/FY 2028 payment determination: (1) the Hospital Harm—Falls With Injury eCQM, and the (2) Hospital Harm—Postoperative Respiratory Failure eCQM, to add to the set of eCQMs from which hospitals may self-select to meet their eCQM reporting requirements. In section IX.C.7.a. of the preamble of this final rule, we discuss the modification of the Global Malnutrition Composite Score eCQM to add patients ages 18 to 64 to the current cohort of patients 65 years or older beginning with the CY 2026 reporting period/FY 2028 payment determination.
Under OMB control number 0938-1022, the currently approved burden estimate for reporting six eCQMs is 4 hours per IPPS hospital (0.167 hours/eCQM x 4 quarters x 6 eCQMs) for all six required eCQM measures. The addition of these two new Hospital Harm eCQMs and modification of the Global Malnutrition Composite Score eCQM will not affect the information collection burden associated with submitting eCQM data under the currently established Hospital IQR Program, which is that hospitals are not required to report more than a total of six eCQMs ( 87 FR 49299 through 49302 ). However, in the immediately following section of this Collection of Information section, we discuss the burden associated with increasing the total number of eCQMs that will be required to be reported.
In section IX.C.9.c. of the preamble of this final rule, we are finalizing with modification the eCQM reporting requirements whereby we will increase the total number of eCQMs to be reported from six to eight eCQMs for the CY 2026 reporting period/FY 2028 payment determination, from eight to nine eCQMs for the CY 2027 reporting period/FY 2029 payment determination, and then from nine to eleven eCQMs beginning with the CY 2028 reporting period/FY 2030 payment determination.
We previously finalized in the FY 2023 IPPS/LTCH PPS final rule that, for the CY 2024 reporting period/FY 2026 payment determination and subsequent years, hospitals are required to submit data for six eCQMs each year which must include the Safe Use of Opioids-Concurrent Prescribing, Cesarean Birth, and Severe Obstetric Complications eCQMs in addition to three self-selected eCQMs ( 87 FR 49387 ). In this final rule, we are finalizing our proposal with modification, such that for the CY 2026 reporting period/FY 2028 payment determination, hospitals will be required to submit data for eight total eCQMs: three self-selected eCQMs, and the Safe Use of Opioids, Severe Obstetric Complications, Cesarean Birth, Hospital Harm—Severe Hypoglycemia, and Hospital Harm—Severe Hyperglycemia eCQMs. We are also finalizing the proposal with modification, such that for the CY 2027 reporting period/FY 2029 payment determination, hospitals will be ( print page 69896) required to submit data for these eight eCQMs in addition to the Hospital Harm—Opioid-Related Adverse Events eCQM. Lastly, we are also finalizing this proposal with modification, such that beginning with the CY 2028 reporting period/FY 2030 payment determination, hospitals will be required to submit data for these nine eCQMs in addition to the Hospital Harm—Pressure Injury and Hospital Harm—Acute Kidney Injury eCQMs.
We continue to estimate the information collection burden associated with the eCQM reporting requirements to be 10 minutes per measure per quarter. For the increase in reporting from six to eight eCQMs for the CY 2026 reporting period/FY 2028 payment determination, we estimate a total of 20 minutes, or 0.33 hours (10 minutes × 2 eCQMs), per hospital per quarter. We estimate a total burden increase of 4,067 hours (0.33 hour × 3,050 IPPS hospitals × 4 quarters) at a cost of $211,972 (4,067 hours × $52.12). For the additional increase in reporting from eight to nine eCQMs beginning with the CY 2027 reporting period/FY 2029 payment determination, we estimate a total of 30 minutes, or 0.5 hours (10 minutes × 3 eCQMs), per hospital per quarter, accounting for both the increase of two eCQMs for the CY 2026 reporting period/FY 2028 payment determination and the increase of one eCQM for the CY 2027 reporting period/FY 2029 payment determination. We estimate a total burden increase of 6,100 hours annually (0.5 hour × 3,050 IPPS hospitals × 4 quarters) at a cost of $317,932 (6,100 hours × $52.12) compared to the currently approved burden estimate. Lastly, for the additional increase in reporting from nine to eleven eCQMs beginning with the CY 2028 reporting period/FY 2030 payment determination, we estimate a total of 50 minutes, or 0.83 hours (10 minutes × 5 eCQMs), per hospital per quarter, accounting for both the increase of two eCQMs for the CY 2026 reporting period/FY 2028 payment determination, the increase of one eCQM for the CY 2027 reporting period/FY 2029 payment determination, and the increase of two eCQMs for the CY 2028 reporting period/FY 2030 payment determination. We estimate a total burden increase of 10,126 hours annually (0.83 hour × 3,050 IPPS hospitals × 4 quarters) at a cost of $527,767 (10,126 hours × $52.12) compared to the currently approved burden estimate.
In section IX.C.5.e. of the preamble of this final rule, we are adopting the Thirty-day Risk-standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) measure beginning with the July 1, 2023-June 30, 2025 reporting period/FY 2027 payment determination. Because this measure is calculated using Medicare Advantage data and Medicare FFS claims that are already reported to the Medicare program for payment purposes, adopting this measure will not result in a change in burden associated with OMB control number 0938-1022.
In section IX.C.6.b. of the preamble of this final rule, we are removing four claims-based payment measures beginning with the FY 2026 payment determination: (1) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for AMI measure; (2) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for HF measure; (3) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Pneumonia measure; and (4) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Elective Primary THA and/or TKA measure. In section IX.C.6.a., we are also removing the Death Rate Among Surgical Inpatients with Serious Treatable Complications (CMS PSI-04) claims-based measure beginning with the FY 2027 payment determination.
Because these measures are calculated using Medicare FFS claims that are already reported to the Medicare program for payment purposes, removing these measures will not result in a change in burden associated with OMB control number 0938-1022.
In section IX.B.2.e. of the preamble of this final rule, we are modifying the HCAHPS Survey measure beginning with the CY 2025 reporting period/FY 2027 payment determination. Specifically, the updated measure includes adding three new sub-measures, removing one existing sub-measure, and revising one existing sub-measure. The new sub-measures will include: “Care Coordination,” “Restfulness of Hospital Environment,” and “Information about Symptoms.”
Under OMB control number 0938-0981 (expiration date January 31, 2025), we estimate the time to complete the HCAHPS Survey is approximately 7.25 minutes per respondent and approximately 2,309,985 respondents will complete and submit the HCAHPS Survey as part of the Hospital IQR Program. As stated in section IX.B.2.b. of this final rule, we estimate the combination of survey sub-measure removals and additions will result in an additional 0.75 minute (0.0125 hour) per respondent to complete the updated version of the HCAHPS Survey. Therefore, we estimate the updated time to complete the HCAHPS Survey will be 8 minutes per respondent (0.133 hour).
We believe that the cost for patients undertaking administrative and other tasks on their own time is a post-tax wage of $24.04/hr. The Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices identifies the approach for valuing time when individuals undertake activities on their own time. [ 1096 ] To derive the costs for patients, a measurement of the usual weekly earnings of wage and salary workers from BLS's Labor Force Statistics program, Current Population Survey (CPS) of $1,118 was divided by 40 hours to calculate an hourly pre-tax wage rate of $27.95/hr. [ 1097 ] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre- and post-tax income, [ 1098 ] resulting in the post-tax hourly wage rate of $24.04/hr. Unlike our state and private sector wage adjustments, we are not adjusting patients' wages for fringe benefits and other indirect costs since the individuals' activities, if any, will occur outside the scope of their employment. We therefore estimate a burden increase of 28,875 hours (2,309,985 respondents ( print page 69897) × 0.0125 hour) at a cost of $694,155 (28,875 hours × $24.04).
We will submit the revised information collection estimates to OMB for approval under OMB control number 0938-0981.
We summarized comments on the information collection burden associated with the adoption of the updates to the HCAHPS Survey measure in section IX.B.2. of this final rule.
In section IX.C.10. of the preamble of this final rule, we are updating the scoring methodology for eCQM validation, replacing the existing combined validation score for eCQMs and chart-abstracted measures with two separate validation scores for chart-abstracted measures and eCQMs, beginning with the FY 2028 payment determination, and removing the requirement that hospitals must submit 100 percent of eCQM records to pass validation beginning with CY 2025 eCQM data affecting the FY 2028 payment determination. We are also finalizing in section IX.C.13 of this final rule to no longer require hospitals to resubmit medical records as part of their request for reconsideration of validation, beginning with CY 2025 discharges affecting the FY 2028 payment determination.
Changes to the scoring methodology and validation score will not affect burden as neither the amount of data nor frequency of data submission is impacted. The removal of the requirement that hospitals must submit 100 percent of eCQM records to pass validation will not affect burden, as the implementation of eCQM validation scoring will still require hospitals to submit the same number of requested medical records to validate the accuracy of eCQM data (the extent to which data abstracted from the submitted medical record matches the data submitted in the QRDA I file). Lastly, as finalized in the FY 2011 IPPS/LTCH PPS final rule regarding information collection burden associated with the Hospital IQR Program's request for reconsideration process, information collection requirements imposed subsequent to an administrative action are not subject to the Paperwork Reduction Act (PRA) under 5 CFR 1320.4(a)(2) , therefore to no longer require hospitals to resubmit medical records as part of their request for reconsideration of validation will not affect burden ( 75 FR 50411 ).
In summary, under OMB control number 0938-1022, we estimate that the policies in this final rule will result in a total increase of 10,635 hours at a cost of $554,296 annually for 3,050 IPPS hospitals from the CY 2025 reporting period/FY 2027 payment determination through the CY 2028 reporting period/FY 2030 payment determination. Under OMB control number 0920-0666, we estimate that the policies in this final rule will result in a total increase of 650 hours at a cost of $33,878 annually for 3,900 hospitals beginning with the CY 2025 reporting period/FY 2027 payment determination. Under OMB control number 0938-0981, we estimate that the policies in this final rule will result in a total increase of 28,875 hours at a cost of $694,155 annually for patients responding to the HCAHPS Survey beginning with the CY 2025 reporting period/FY 2027 payment determination. The total increase in burden associated with the finalized information collections under OMB control numbers 0938-1022, 0920-0666, and 0938-0981 is approximately 40,160 hours (10,635 + 650 + 28,875) at a cost of $1,282,329 ($554,296 + $33,878 + $694,155). We will submit the revised information collection estimates to OMB for approval under OMB control numbers 0938-1022, 0920-0666, and 0938-0981.
With respect to any costs/burdens unrelated to data submission, we refer readers to the Regulatory Impact Analysis (section I.K. of Appendix A of this final rule).
Comment: A commenter stated that in general, the burden estimates for the program do not accurately reflect the total reporting process, with specific mention of information reporting to state and local health departments.
Response: We thank the commenter for its feedback on the burden associated with reporting quality measures. We understand that some hospitals may experience burden greater than our estimates, however, our estimates appropriately reflect the average across all IPPS and non-IPPS hospitals. CMS continues to make efforts to balance the burden of quality measure reporting with the value provided to both participating hospitals and the public by the measures included in the Hospital IQR Program measure set. We also note that the burden estimates in this final rule only reflect the time required to collect and submit required data to federal agencies under the Hospital IQR Program and not to state and local health departments due to any requirements external to the Hospital IQR Program.

OMB has currently approved 109 hours of burden at a cost of $2,452 under OMB control number 0938-1175 (expiration date January 31, 2027), accounting for the annual information collection requirements for 11 PCHs for the PCHQR Program. In this final rule, we are adopting the Patient Safety Structural measure with modification to one domain beginning with the CY 2025 reporting period/FY 2027 program year. This new measure will affect the information collection burden. In addition, we are modifying the HCAHPS Survey beginning with the CY 2025 reporting period/FY 2027 program year, which is currently approved under OMB control number 0938-0981 (expiration date January 31, 2025). We are also moving up the start date for public display of the Hospital Commitment to Health Equity (HCHE) measure. This policy will not affect the information collection burden associated with the PCHQR Program.
In the FY 2024 IPPS/LTCH PPS final rule, we utilized the median hourly wage rate for Medical Records Specialists, in accordance with the Bureau of Labor Statistics (BLS), to calculate our burden estimates for the PCHQR Program ( 88 FR 59317 ). While the most recent data from the BLS reflects a mean hourly wage of $24.56 per hour for all medical records specialists, $26.06 is the mean hourly wage for “general medical and surgical hospitals,” which is an industry within medical records specialists. [ 1099 ] We believe the industry of “general medical and surgical hospitals” is more specific to our settings for use in our calculations than other industries that fall under medical records specialists, such as “office of physicians” or “nursing care facilities.” We calculated the cost of overhead, including fringe benefits, at 100 percent of the mean hourly wage, consistent with previous years. This is necessarily a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, we believe that doubling the hourly wage rate ($26.06 × 2 = $52.12) to estimate total cost is a reasonably accurate estimation method. Accordingly, unless otherwise specified, we will calculate cost burden to hospitals using a wage plus benefits estimate of $52.12 per hour throughout the discussion in this section of this rule for the PCHQR Program.
In section IX.B.1. of the preamble of this final rule, we are adopting with modification the Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 program year. PCHs will submit responses on an annual basis during the submission period through the Center for Disease Control and Prevention's (CDC) National Healthcare Safety Network (NHSN). Specifically, PCHs will be required to provide responses and attest “yes” or “no” in response to a total of five domains for a given reporting period. Similar to the Hospital Commitment to Health Equity measure currently approved under OMB control number 0938-1022 (expiration date ( print page 69900) January 31, 2026), which also requires a “yes” or “no” response to each of five domains, we estimate the information collection burden associated with this measure to be, on average across all 11 PCHs, no more than 10 minutes per PCH per year. We are modifying the attestation statement in Domain 4 Statement B of the measure to remove the portion of the attestation related to voluntary reporting to the NPSD and focus instead on the beneficial activities possible through engagement with a PSO. Because this does not affect the number of responses each hospital must provide, we are not making a change to our burden assumptions. Using the estimate of 10 minutes (or 0.167 hours) per PCH per year, and the updated wage estimate as described previously, we estimate that the adoption of this measure will result in a total annual burden increase of 2 hours across all participating PCHs (0.167 hours × 11 PCHs) at a cost of $104 (2 hours × $52.12).
We discussed the burden associated with the adoption of the Patient Safety Structural measure for the Hospital IQR Program in section XII.B.6.c. of this final rule. We will work with CDC to submit the revised information collection estimates for NHSN data collection to OMB for approval under OMB control number 0920-0666.
We received no comments on this proposal and therefore are finalizing our burden estimates without modification.
In section IX.B.2.e of the preamble of this final rule, we are modifying the HCAHPS Survey measure beginning with the CY 2025 reporting period/FY 2027 program year. Specifically, we are refining the current HCAHPS Survey by adding three new sub-measures, removing one existing sub-measure, and revising one existing sub-measure. The new sub-measures will include: “Care Coordination,” “Restfulness of Hospital Environment,” and “Information about Symptoms.”
Under OMB control number 0938-0981 (expiration date January 31, 2025), we estimated the time to complete the HCAHPS Survey is approximately 7.25 minutes per respondent and approximately 13,105 respondents will complete and submit the HCAHPS Survey as part of the PCHQR Program. As stated in section IX.B.2.b of this final rule, we estimated the combination of sub-measure removals and additions will result in an additional 0.75 minutes (0.0125 hours) per respondent to complete the HCAHPS Survey. Therefore, we estimated the updated time to complete the HCAHPS Survey will be 8 minutes per respondent (0.133 hours).
We believe that the cost for beneficiaries undertaking administrative and other tasks on their own time is a post-tax wage of $24.04/hr. The Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices identifies the approach for valuing time when individuals undertake activities on their own time. [ 1100 ] To derive the costs for beneficiaries, a measurement of the usual weekly earnings of wage and salary workers of $1,118 was divided by 40 hours to calculate an hourly pre-tax wage rate of $27.95/hr. [ 1101 ] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre- and post-tax income, [ 1102 ] resulting in the post-tax hourly wage rate of $24.04/hr. Unlike our State and private sector wage adjustments, we are not adjusting beneficiary wages for fringe benefits and other indirect costs since the individuals' activities, if any, will occur outside the scope of their employment. We therefore estimate a burden increase of 164 hours (13,105 respondents × 0.0125 hours) at a cost of $3,943 (164 hours × $24.04).
We will submit the revised information collection request to OMB for approval under OMB control number 0938-0981.
We summarized comments on the proposed information collection burden associated with the adoption of the updates to the HCAHPS Survey measure in Section IX.B.2. of this final rule. We are finalizing our burden estimates without modification.
In section IX.D.5. of the preamble of this final rule, we are moving up the start date of PCH performance on the Hospital Commitment to Health Equity measure. Because we are not requiring PCHs to collect or submit any additional data, we do not estimate any change in information collection burden associated with this policy.
We received no comments on this proposal and are therefore finalizing our assumptions regarding burden without modification.
In summary, under OMB control number 0920-0666 (expiration date December 31, 2026), we estimate that the policies being finalized will result in a total increase of 2 hours at a cost of $104 annually for 11 PCHs beginning with the CY 2025 reporting period/FY 2027 program year. Under OMB control number 0938-0981 (expiration date January 31, 2025), we estimated that the policies being finalized will result in a total increase of 164 hours at a cost of $3,943 annually for 11 PCHs beginning with the CY 2025 reporting period/FY 2027 program year. The total increase in burden associated with this information collection will be approximately 166 hours at a cost of $4,047. We will submit the revised information collection request to OMB for approval under OMB control numbers 0920-0666 and 0938-0981.

An LTCH that does not meet the requirements of the LTCH QRP for a fiscal year will receive a 2-percentage point reduction to its otherwise applicable annual update for that fiscal year.
We believe that the burden associated with the LTCH QRP is the time and effort associated with complying with the requirements of the LTCH QRP. In sections IX.E.4.c. and IX.E.4.e. of the preamble of this final rule, we proposed to add four items to the LCDS and modify one item on the LCDS. The LCDS V5.1 has been approved under OMB control number 0938-1163 (Expiration date: 08/31/2025). The following is a discussion of this information collection.
In section IX.E.4.c. of this final rule, we proposed to adopt four new items as standardized patient assessment data elements under the SDOH category beginning with the FY 2028 LTCH QRP. The proposed items, Living Situation (one item), Food (two items), and Utilities (one item), will be collected at admission using the LCDS. These four new items will be added to the LCDS and will result in an increase of 0.02 hours (1.2 minutes/60) of clinical staff time at admission. In addition, as described in section IX.E.4.e. of this final rule, we also proposed to modify the current Transportation item on the LCDS, which is currently collected at admission and discharge. We proposed that the modified Transportation item will only be collected at admission beginning with the FY 2028 LTCH QRP as described in section . and IX.E.7.b. of this final rule. The burden associated with collecting this item at admission and discharge was accounted for in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42606 ) when the item was originally adopted. Additionally, LTCHs will no longer have to collect one item at discharge to meet LTCH QRP reporting requirements, which will result in a decrease of 0.005 hours (0.3 minutes/60) of clinical staff time at discharge. Using data collected for FY 2023, we estimate 130,050 total admissions and 96,890 planned discharges from 330 LTCHs annually. This equates to an increase of 2,117 hours for all LTCHs [(130,050 × 0.02 hour) minus (96,890 × 0.005 hour)] and 6.41 hours per LTCH.
We believe that the additional SDOH items will be completed equally by RNs and LPN/LVNs. Individual LTCHs determine the staffing resources necessary. We averaged BLS' National Occupational Employment and Wage Estimates (see Table XII.B-08) for these labor types and established a composite cost estimate using our adjusted wage estimates. The composite estimate of $65.31/hr was calculated by weighting each hourly wage equally [($78.10 + $52.52)/2]. We estimate the total cost would be increased by $418.88 per LTCH annually, or $138,231.88 for all LTCHs annually ([(130,050 admission assessments × 0.02 hour = 2,601 hours) × $65.31/hr] minus [(96,890 planned discharge assessments × 0.005 hour = 484.45 hours) × $65.31/hr] = $138,231.88); ($138,231.88/330 LTCHs = $418.88/LTCH).

As described in Table XII.B-09, under OMB control number 0938-1163, we estimate that the policies finalized in this final rule for the LTCH QRP would result in an overall increase of 2,117 hours annually for 330 LTCHs. The total cost increase related to this information collection is estimated at approximately $138,231.88. The increase in burden would be accounted for in a revised information collection request under OMB control number (0938-1163).

In section IX.E.7.c. of this final rule, we finalized the proposal to extend the LCDS Admission assessment window from three days to four days beginning with the FY 2028 LTCH QRP. However, this change will have no impact on burden since it is an administrative change and does not impact the number of items collected.
We invited public comments on these potential information collection requirements. We responded to these comments in section IX.E.4 and IX.E.7 of this final rule. After considering the public comments received, and for the reasons outlined in these sections of the final rule and our comment responses, we are finalizing the revisions as proposed.
In section IX.F. of the preamble of this final rule, we discussed several finalized policies for the Medicare Promoting Interoperability Program. As discussed in the most recent Paperwork Reduction Act (PRA) approval under OMB control number 0938-1278 (expiration date April 30, 2027), OMB has approved 29,625 hours of burden at a cost of approximately $1.3 million, accounting for information collection burden experienced by approximately 3,150 eligible hospitals and 1,350 CAHs for the EHR reporting period in CY 2024. In this final rule, we describe the burden changes regarding collection of information under OMB control numbers 0938-1278 and 0938-1022 for eligible hospitals and CAHs. The collection of information burden analysis is focused on all eligible hospitals and CAHs that could participate in the Medicare Promoting Interoperability Program and report the objectives and measures and electronic clinical quality measures (eCQMs), under the Medicare Promoting Interoperability Program for the EHR reporting periods in CY 2025 through CY 2028.
We are adopting two new eCQMs beginning with the CY 2026 reporting period: (1) the Hospital Harm—Falls with Injury eCQM, and (2) the Hospital Harm—Postoperative Respiratory Failure eCQM. In addition, we are separating the previously finalized Antimicrobial Use and Resistance (AUR) Surveillance measure into two separate measures, beginning with the EHR reporting period in CY 2025: (1) the Antimicrobial Use (AU) Surveillance measure, and (2) the Antimicrobial Resistance (AR) Surveillance measure. We are also modifying the Global Malnutrition Composite Score eCQM, beginning with the CY 2026 reporting period. In addition, we are increasing the total number of eCQMs that must be reported each year by eligible hospitals and CAHs from six to eight eCQMs for the CY 2026 reporting period, from eight to nine eCQMs for the CY 2027 reporting ( print page 69903) period, and then from nine to eleven eCQMs beginning with the CY 2028 reporting period. Lastly, we are increasing the minimum scoring threshold from 60 points to 70 points for the EHR reporting period in CY 2025 and then from 70 points to 80 points beginning with the EHR reporting period in CY 2026; this will not affect the information collection burden associated with the Medicare Promoting Interoperability Program.
In the FY 2024 IPPS/LTCH PPS final rule, we utilized the median hourly wage rate for Medical Records Specialists, in accordance with the BLS, to calculate our burden estimates for the Medicare Promoting Interoperability Program ( 88 FR 59325 ). While the most recent data, the May 2022 National Occupational Employment and Wage Estimates from the BLS, reflects a mean hourly wage of $24.56 per hour for all medical records specialists (SOC 29-2072); we use the mean hourly wage for medical records specialists for the industry, “general medical and surgical hospitals,” which is $26.06. [ 1103 ] We believe the industry of “general medical and surgical hospitals” is more specific to our settings for use in our calculations than other industries that fall under medical records specialists, such as “office of physicians” or “nursing care facilities.” We calculated the cost of overhead, including fringe benefits, at 100 percent of the median hourly wage, consistent with previous years. This is necessarily a rough adjustment, both because fringe benefits and overhead costs vary significantly by employer and methods of estimating these costs vary widely in the literature. Nonetheless, we believe that doubling the hourly wage rate ($26.06 × 2 = $52.12) to estimate total cost is a reasonably accurate estimation method. Accordingly, unless otherwise specified, we will calculate cost burden to eligible hospitals and CAHs using a wage plus benefits estimate of $52.12 per hour throughout the discussion in this section of this rule for the Medicare Promoting Interoperability Program.
In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59325 ), our burden estimates were based on an assumption of 4,500 eligible hospitals and CAHs. In the FY 2024 IPPS/LTCH PPS final rule, the Medicare Promoting Interoperability Program and Hospital IQR Program used the same estimate for the number of eligible hospitals and IPPS hospitals for both programs ( 88 FR 59325 ). In section XII.B.6.a. of the preamble of this final rule, we provide our updated estimate of 3,050 IPPS hospitals for the Hospital IQR Program for the CY 2025 reporting period. Upon further analysis, we believe it is no longer appropriate to use the same estimate for both programs as the approximately 100 eligible hospitals located in Maryland and Puerto Rico which were previously excluded from our estimate of IPPS hospitals and included in our estimate of non-IPPS hospitals should be included as eligible hospitals for the Medicare Promoting Interoperability Program. Therefore, based on data from the EHR reporting period in CY 2022, we estimated approximately 3,150 eligible hospitals and 1,400 CAHs will report data to the Medicare Promoting Interoperability Program for the EHR reporting period in CY 2025, for a total number of 4,550 respondents.
In section IX.F.6.a. of the preamble of this final rule, we are adopting two new eCQMs beginning with the CY 2026 reporting period: (1) the Hospital Harm—Falls With Injury eCQM and (2) the Hospital Harm—Postoperative Respiratory Failure eCQM, to add to the set of eCQMs from which hospitals may self-select to meet their eCQM reporting requirements. In section IX.F.6.a. of the preamble of this final rule, we are modifying the Global Malnutrition Composite Score eCQM to add patients ages 18 to 64 to the current cohort of patients 65 years or older beginning with the CY 2026 reporting period.
Under OMB control number 0938-1022 (expiration date January 31, 2026), the currently approved burden estimate for reporting of six eCQMs is four hours per CAH and the 100 eligible hospitals not included as IPPS hospitals for the Hospital IQR Program (0.167 hours/eCQM × 4 quarters × 6 eCQMs) for all six required eCQMs. The addition of these two new Hospital Harm eCQMs and modification of the Global Malnutrition Composite Score eCQM will not affect the information collection burden associated with submitting eCQM data under the Medicare Promoting Interoperability Program. As finalized in the FY 2023 IPPS/LTCH PPS final rule, current policy requires CAHs to select six eCQMs from the eCQM measure set on which to report ( 87 FR 49365 through 49367 ). In other words, although these new eCQMs are being added to the eCQM measure set, CAHs are not required to report more than a total of six eCQMs for the CY 2025 reporting period. We refer readers to section XII.B.7.f. of this final rule for discussion of the burden estimates associated with this policy impacting eligible hospitals (referred to as IPPS hospitals under the Hospital IQR Program).
In section XII.B.9.c. of this final rule (Collection of Information section), we account for the burden associated with increasing the total number of eCQMs reported from six to eight eCQMs for the CY 2026 reporting period, from eight to nine eCQMs for the CY 2027 reporting period, and then from nine to eleven eCQMs beginning with the CY 2028 reporting period. We refer readers to section XII.B.7.f. of this final rule for discussion of the burden estimates associated with these policies impacting eligible hospitals (referred to as IPPS hospitals under the Hospital IQR Program).
We received no comments on the proposals and are therefore finalizing our assumptions regarding burden without modification.
In section IX.F.6.b. of the preamble of this final rule, we are finalizing with modification our eCQM reporting requirements by increasing the total number of eCQMs to be reported from six to eight eCQMs for the CY 2026 reporting period, from eight to nine eCQMs for the CY 2027 reporting period, and from nine to eleven eCQMs beginning with the CY 2028 reporting period.
We previously finalized in the FY 2023 IPPS/LTCH PPS final rule that, for the CY 2024 reporting period, CAHs are required to annually submit data for six eCQMs each year, which must consist of the Safe Use of Opioids-Concurrent Prescribing, Cesarean Birth, and Severe Obstetric Complications eCQMs in addition to three self-selected eCQMs ( 87 FR 49394 through 49395 ). We are finalizing with modification that, for the CY 2026 reporting period, CAHs will be required to submit data for eight total eCQMs: three self-selected eCQMs, and the Safe Use of Opioids, Severe Obstetric Complications, Cesarean Birth Rate, Hospital Harm—Severe Hypoglycemia, and Hospital Harm—Severe Hyperglycemia eCQMs. We are also finalizing with modification that, for the CY 2027 reporting period, CAHs will be required to submit data for these eight eCQMs as well as the Hospital Harm—Opioid-Related Adverse Events eCQM, for nine total eCQMs. Lastly, we are finalizing with modification that, ( print page 69904) beginning with the CY 2028 reporting period, CAHs will be required to submit data for these nine eCQMs as well as the Hospital Harm—Pressure Injury and Hospital Harm—Acute Kidney Injury eCQMs for eleven total eCQMs.
To calculate the information collection burden associated, we estimate a total of 1,500 respondents, which includes the 100 eligible hospitals not included as IPPS hospitals for the Hospital IQR Program as well as the 1,400 CAHs required to report eCQM data for the Medicare Promoting Interoperability Program. We continue to estimate the information collection burden associated with the eCQM reporting and submission requirements to be 10 minutes per measure per quarter. For the increase in submission from six to eight eCQMs for the CY 2026 reporting period, we estimate a total of 20 minutes, or 0.33 hours (10 minutes × 2 eCQMs), per CAH per quarter. We estimate a total burden increase of 2,000 hours (0.33 hour × 1,400 CAHs and 100 eligible hospitals × 4 quarters) at a cost of $104,240 (2,000 hours × $52.12). For the additional increase in submission from eight to nine eCQMs beginning with the CY 2027 reporting period, we estimate a total of 30 minutes, or 0.5 hours (10 minutes × 3 eCQMs), per CAH per quarter. We estimate a total burden increase of 3,000 hours annually (0.5 hours x 1,500 CAHs × 4 quarters) at a cost of $156,360 (3,000 hours × $52.12). For the additional increase in submission from nine to eleven eCQMs beginning with the CY 2027 reporting period, we estimate a total of 50 minutes, or 0.83 hours (10 minutes × 5 eCQMs), per CAH per quarter. We estimate a total burden increase of 5,000 hours annually (0.83 hours × 1,500 CAHs × 4 quarters) at a cost of $260,600 (5,000 hours × $52.12). We refer readers to section XII.B.7.f. of this final rule for discussion of the burden estimates associated with this policy impacting eligible hospitals (referred to as IPPS hospitals under the Hospital IQR Program).
With respect to any costs/burdens related to eligible hospitals (referred to as IPPS hospitals under the Hospital IQR Program), we refer readers to section XII.B.7.f. of this final rule.
In section IX.F.2. of the preamble of this final rule, we are finalizing the modification of the AUR Surveillance measure by separating the single measure into two measures: (1) AU Surveillance, and (2) AR Surveillance, beginning with the EHR reporting period in CY 2025. In the CY 2023 IPPS/LTCH PPS final rule, we finalized a burden estimate of 0.5 minutes per eligible hospital and CAH to attest the AUR Surveillance measure ( 87 FR 49394 ). In association with this policy, we estimate an annual increase in burden for each eligible hospital and CAH to attest to both measures of 0.5 minutes (.0083 hours). Therefore, we estimate a total increase in burden of 38 hours across all eligible hospitals and CAHs (.0083 hours × 4,550 eligible hospitals and CAHs) annually at a cost of $1,981 (38 hours × $52.12).
In section IX.F.5. of the preamble of this final rule, we are finalizing an increase to the minimum scoring threshold from 60 points to 70 points for the EHR reporting period in CY 2025 and an increase to the minimum scoring threshold from 70 points to 80 points beginning with the EHR reporting period in CY 2026. Because we are not requiring eligible hospitals or CAHs to collect or submit any additional data, we do not estimate any change in information collection burden associated with the policy.
In summary, under OMB control number 0938-1278, we estimate that the policies in this final rule will result in an increase in burden of 38 hours at a cost of $1,981 across 4,550 hospitals. In addition, under OMB control number 0938-1022, we estimate that the policies in this final rule will result in an increase in burden of 5,000 hours at a cost of $260,600 across 4,550 hospitals. The total increase in burden associated with the finalized information collections under OMB control numbers 0938-1278 and 0938-1022 is approximately 5,038 hours (38 + 5,000) at a cost of $262,581 ($1,981 + $260,600). Based on these policies, the annual burden per eligible hospital and CAH will increase to 6 hours and 36 minutes (6.6 hours) as well as an additional 7.33 hours annually for CAHs and the 100 eligible hospitals that do not participate in the Hospital IQR Program to report eCQMs. We will submit the revised information collection estimates to OMB for approval under OMB control numbers 0938-1022 and 0938-1278. With respect to costs/burdens related to eligible hospitals (referred to as IPPS hospitals under the Hospital IQR Program), we refer readers to section XII.B.7.f. of this final rule.
With respect to any costs/burdens unrelated to data submission, we refer readers to the Regulatory Impact Analysis (section I.N. of Appendix A of this final rule).

In section X.A. of the preamble of this final rule, we discuss testing the Transforming Episode Accountability Model (TEAM) under the authority of the CMS Innovation Center. Section 1115A of the Act authorizes the CMS Innovation Center to test innovative payment and service delivery models that preserve or enhance the quality of care furnished to Medicare, Medicaid, and Children's Health Insurance Program beneficiaries while reducing program expenditures. As stated in section 1115A(d)(3) of the Act, Chapter 35 of title 44, United States Code , shall not apply to the testing and evaluation of models under section 1115A of the Act. As a result, the information collection requirements contained in this final rule for TEAM need not be reviewed by the Office of Management and Budget.
We received no comments on the information collection requirements and therefore are finalizing this provision without modification.
Section 431.970 defines state and provider submission responsibilities, including state submission of Medicaid and CHIP FFS claims and managed care payments on a quarterly basis; and provider submission of medical records. These claims and payments are rigorously reviewed by the Federal statistical contractor. Additionally, states are required to collect and submit (with an estimate of 4 submissions) state policies, including an initial submission ( print page 69906) and quarterly updates. The ongoing burden associated with the requirements under § 431.970 is the time and effort it will take each of the up to 36 state programs (17-18 Medicaid and 17-18 CHIP agencies for 17-18 states equates to maximum 36 total respondents each PERM year) to submit its claims universe, collect and submit state policies, and the time and effort it will take providers to furnish medical record documentation. We estimate that it will take 1,350 hours annually per state program to develop and submit its claims universe and state policies. The total estimated hours are broken down between the FFS, managed care, and eligibility components and is estimated at 900 hours for universe development and submission, and 450 hours for policy collection and submission. Per component it is estimated at 1,150 FFS hours, 100 managed care hours, and 100 eligibility hours for a total of 48,600 annual hours (1,350 hours × 36 respondents). The total estimated annual cost per respondent is $86,832 (1,350 hours × $64.32), and the total estimated annual cost across all respondents is $3,125,952 ($86,832 × 36 respondents). The preceding requirements and burden estimates will be submitted to OMB as reinstatements with changes of the information collection requests previously approved under control numbers 0938-0974, 0938-0994, and 0938-1012. Inclusion of Puerto Rico has added an additional burden of 2,700 hours and $173,664 for Information Submission and Systems Access Requirements.
Section 431.992 requires states to submit corrective action plans to address all improper payments and deficiencies found through the PERM review as defined at § 431.960(f)(1) and evaluate corrective actions from the previous PERM cycle as defined at § 431.992(b)(4). The ongoing burden associated with the requirements under § 431.992 is the time and effort it would take each of the up to 36 state programs (17-18 Medicaid and 17-18 CHIP agencies for 17-18 states equates to maximum 36 total respondents per PERM cycle) to submit its corrective action plan. We estimate that it will take 750 hours (250 hours for FFS, 250 hours for managed care and an additional 250 hours for eligibility), per PERM cycle per state program to submit its corrective action plan for a total estimated annual burden of 27,000 hours (750 hours × 36 respondents). We estimate the total cost per respondent to be $48,240 (750 hours × $64.32). The total estimated cost for all respondents is $1,736,640 ($48,240 × 36 respondents). The preceding requirements and burden estimates will be submitted to OMB as part of reinstatement of the information collection requests previously approved under control numbers 0938-0974, 0938-0994, and 0938-1012. total burden would amount to: 36 annual respondents, 36 annual responses, and 750 hours per corrective action plan Inclusion of Puerto Rico has added an additional burden of 1,500 hours and $96,480 for Corrective Action Plan requirements.
Section 431.998 allows states to dispute federal contractor findings. The ongoing burden associated with the requirements under § 431.998 is the time and effort it would take each of the up to 36 state programs (17-18 Medicaid and 17-18 CHIP agencies for 17-18 states equates to maximum 36 total respondents per PERM cycle) to review PERM findings and inform the Federal contractor(s) of any additional information and/or dispute requests. We estimated that it will take 1,625 hours (500 hours for FFS, 475 hours for managed care and an additional 650 hours for eligibility) per PERM cycle per state program to review PERM findings and inform federal contractor(s) of any additional information or dispute requests for FFS, managed care, and eligibility components for a total estimated annual burden of 58,500 hours (1,625 hours × 36 respondents). We estimate the total cost per respondent to be $104,520 (1,625 hours × $64.32). The total estimated cost for all respondents is $3,762,720 ($104,520 × 36 respondents). The preceding requirements and burden estimates will be submitted to OMB as reinstatements of the information collection requests previously approved under control numbers 0938-0974, 0938-0994, and 0938-1012. Total burden would amount to: 36 annual respondents, 36 annual responses, and 1,625 hours per PERM cycle.
Inclusion of Puerto Rico has added an additional burden of 3,250 hours and $209,040 for Difference Resolution and Appeal Process requirements.
The hospital must electronically report information on acute respiratory illnesses, including influenza, SARS-CoV-2/COVID-19, and RSV, in a standardized format and frequency specified by the Secretary. To the extent as required by the Secretary, this report must include the following data elements:
- Confirmed infections for a limited set of respiratory illnesses, including but not limited to influenza, SARS-CoV-2/COVID-19, and RSV, among newly admitted and hospitalized patients.
- Total bed census and capacity, including for critical hospital units and age groups.
- Limited patient demographic information, including but not limited to age.
For purposes of burden estimates, we do not differentiate among hospitals and CAHs as they all would collect data. For the estimated costs contained in the analysis that follows, we used data from the BLS to determine the mean hourly wage for the staff member responsible for reporting the required information for a hospital (or a CAH). [ 1104 ] Based on our experience with hospitals and CAHs and the previous COVID-19 and related reporting requirements, we believe that ( print page 69907) this would primarily be the responsibility of a registered nurse and we have used this position in this analysis at an average hourly salary of $39.05. For the total hourly cost, we doubled the mean hourly wage for a 100 percent increase to cover overhead and fringe benefits, according to standard HHS estimating procedures. If the total cost after doubling resulted in 0.50 or more, the cost was rounded up to the next dollar. If it was 0.49 or below, the total cost was rounded down to the next dollar. Therefore, we estimated the total hourly cost for a registered nurse to perform these duties would be $78.
We expect that facilities will need to review their existing policies and procedures to ensure they comply with the permanent reporting requirements finalized in this rule. We assume that a RN with responsibility for these activities will review and update the policies and procedures. In addition, prior to the actual submission of the data, we expect that compliance with ongoing reporting will require continuous efforts to collect and organize the information necessary to report the data through the NHSN or other CDC-owned or CDC supported system as determined by the Secretary. Based on the assumption of weekly reporting frequency, we estimate that total annual burden hours for all participating hospitals and CAHs to conduct these activities and comply with these requirements would be 248,976 hours based on weekly reporting of the required information by approximately 6,384 hospitals and CAHs × 52 weeks per year and at an average weekly response time of 0.75 hours for a registered nurse with an average hourly salary of $78. Therefore, the estimate for total annual costs for all hospitals and CAHs to comply with the required reporting provisions weekly would be $19,420,128 (248,976 hours × 6,384 facilities) or approximately $3,042 per facility annually ($19,420,128/6,384 facilities).
We will update the PRA packages for the hospital and CAH CoPs to include these preliminary estimates for these reporting activities (OMB control numbers 0938-0328 for hospitals and 0938-1043 for CAHs). We note that any additional ICR burden related to the specific instruments used for reporting and the time necessary to submit/report the data is the National Healthcare Safety Network (NHSN) Surveillance in Healthcare Facilities (OMB control number 0920-1317) package.
Furthermore, we note that this estimate likely overestimates the costs associated with reporting because it assumes that all hospitals and CAHs will report manually. Efforts are underway to automate hospital and CAH reporting that have the potential to significantly decrease reporting burden and improve reliability. Our preliminary estimates for these reporting activities (OMB control numbers 0938-0328 for hospitals and 0938-1043 for CAHs) can be found in the tables that follow.

In the event that the Secretary has declared a Public Health Emergency (PHE) for an acute infectious illness, the hospital must also electronically report the following data elements in a standardized format and frequency specified by the Secretary:
- Supply inventory shortages.
- Staffing shortages.
- Relevant medical countermeasures and therapeutic inventories, usage, or both.
- Facility structure and operating status, including hospital/ED diversion status.
Similar to the activities necessary to comply with ongoing reporting, hospitals and CAHs will need to ensure they have policies and procedures in place to activate PHE specific reporting and will require staff to gather the information necessary to support reporting at a frequency determined by the Secretary (OMB Control Nos. 0938-0328 and 0938-1043). Likewise, the specific information collection for the data reporting is the NHSN Surveillance in Healthcare Facilities (OMB Control Number 0920-1317). For purposes of burden estimates, we do not differentiate among hospitals and CAHs as they all would complete the same data collection. For the estimated costs ( print page 69908) contained in the analysis that follows, we used data from the U.S. Bureau of Labor Statistics (BLS) to determine the mean hourly wage for the staff member responsible for reporting the required information for a hospital (or a CAH). [ 1105 ] Based on our experience with hospitals and CAHs and the previous COVID-19 and related reporting requirements, we believe that this will primarily be the responsibility of a registered nurse and we have used this position in this analysis at an average hourly salary of $39.05. For the total hourly cost, we doubled the mean hourly wage for a 100 percent increase to cover overhead and fringe benefits, according to standard HHS estimating procedures. If the total cost after doubling resulted in 0.50 or more, the cost was rounded up to the next dollar. If it was 0.49 or below, the total cost was rounded down to the next dollar. Therefore, we estimated the total hourly cost for a registered nurse to perform these duties will be $78.
We acknowledge that the data elements and reporting frequency could increase or decrease due to the what the Secretary deems necessary for the given PHE; the changes would impact this burden estimate. For instance, data reporting requirements may be active for less than or more than a year. During the COVID-19 PHE, facilities reported daily. However, we cannot predict how often the Secretary would require data reporting for any future PHE. Therefore, we include two burden estimates to encapsule a range in frequency of reporting. The lower range is based on twice a week reporting. The higher range is based on daily reporting.
Based on the assumption of twice weekly reporting frequency, we estimate that total annual burden hours for all participating hospitals and CAHs to comply with these requirements will be 995,904 hours based on twice weekly reporting of the required information by approximately 6,384 hospitals and CAHs × 104 days a year and at an average twice weekly response time of 1.5 hours for a registered nurse with an average hourly salary of $78. Therefore, the estimate for total annual costs for all hospitals and CAHs to comply with the required reporting provisions weekly will be $77,680,512 (995,904 hours × $78) or approximately $12,168 ($77,680,512/6,384 facilities) per facility annually.
Based on the assumption of daily reporting frequency, we estimate that total annual burden hours for all participating hospitals and CAHs to comply with these requirements will be 3,495,240 hours based on daily reporting of the required information by approximately 6,384 hospitals and CAHs × 365 days a year and at an average daily response time of 1.5 hours for a registered nurse with an average hourly salary of $78. Therefore, the estimate for total annual costs for all hospitals and CAHs to comply with the required reporting provisions weekly will be $272,628,720 (3,495,240 hours × $78) or approximately $42,705 ($272,628,720/6,384 facilities) per facility annually.

Chiquita Brooks-LaSure, Administrator of the Centers for Medicare Medicaid Services, approved this document on July 26, 2024.
- Administrative practice and procedure
- Health facilities
- Health professions
- Medical devices
- Medicare Reporting and recordkeeping requirements
- Rural areas
- Puerto Rico
- Reporting and recordkeeping requirements
- Grant programs-health
- Health maintenance organizations (HMO)
- Health records
- Privacy, and Reporting and recordkeeping requirements
- Health care
- Health insurance
- Intergovernmental relations
For the reasons set forth in the preamble, the Centers for Medicare Medicaid Services amends 42 CFR chapter IV as follows:
1. The authority citation for part 405 continues to read as follows:
Authority: 42 U.S.C. 263a , 405(a) , 1302 , 1320b-12 , 1395x , 1395y(a) , 1395ff , 1395hh , 1395kk , 1395rr , and 1395ww(k) .
2. Effective January 1, 2025, amend § 405.1845 by revising paragraphs (a) and (b) and the paragraph (c) paragraph heading to read as follows:
(a) Composition of the Board. The Board consists of five members appointed by the Secretary.
(1) All members must be knowledgeable in the field of payment of providers under Medicare Part A.
(2) At least one member must be a certified public accountant.
(3) At least two Board members must be representative of providers of services.
(b) Terms of office. The term of office for Board members must be 3 years, except that initial appointments may be for such shorter terms as the Secretary may designate to permit staggered terms of office.
(1) No member may serve more than three consecutive terms of office.
(2) The Secretary has the authority to terminate a Board member's term of office for good cause.
(c) Role of the Chairperson. * * *
3. The authority citation for part 412 continues to read as follows:
Authority: 42 U.S.C. 1302 and 1395hh .
4. Section 412.1 is amended by revising paragraph (a)(1)(iv) to read as follows:
(iv) Additional payments are made for outlier cases, bad debts, indirect medical education costs, for serving a disproportionate share of low-income patients, for the additional resource costs of domestic National Institute for Occupational Safety and Health approved surgical N95 respirators, and for the additional resource costs for small, independent hospitals to establish and maintain access to buffer stocks of essential medicines.
5. Section 412.2 is amended by adding paragraph (f)(11) to read as follows:
(11) A payment adjustment for small, independent hospitals for the additional resource costs of establishing and maintaining access to buffer stocks of ( print page 69910) essential medicines as specified in § 412.113.
6. Section 412.23 is amended by revising paragraphs (e)(3)(i) and (iii), removing and reserving paragraph (e)(3)(iv) and revising and republishing paragraph (e)(4) to read as follows:
(i) Subject to the provisions of paragraphs (e)(3)(ii) through (vii) of this section and paragraphs (e)(4)(iv) and (v) of this section as applicable, the average Medicare inpatient length of stay specified under paragraph (e)(2)(i) of this section is calculated by dividing the total number of covered and noncovered days of stay of Medicare inpatients (less leave or pass days) by the number of total Medicare discharges for the hospital's most recent complete cost reporting period. Subject to the provisions of paragraphs (e)(3)(ii) through (vii) of this section, the average inpatient length of stay specified under paragraph (e)(2)(ii) of this section is calculated by dividing the total number of days for all patients, including both Medicare and non-Medicare inpatients (less leave or pass days) by the number of total discharges for the hospital's most recent complete cost reporting period.
(iii) If a change in a hospital's average length of stay specified under paragraph (e)(2)(i) or (e)(2)(ii) of this section would result in the hospital not maintaining an average Medicare inpatient length of stay of greater than 25 days, the calculation is made by the same method for the period of at least 5 consecutive months of the immediately preceding 6-month period.
(iv) [Reserved]
(4) For the purpose of calculating the average length of stay for hospitals seeking to become long-term care hospitals, with the exception of paragraphs (e)(3)(iii) and (v) of this section, the provisions of paragraph (e)(3) of this section apply.
(i) Definition. For the purpose of payment under the long-term care hospital prospective payment system under subpart O of this part, a new long-term care hospital is a provider of inpatient hospital services that meets the qualifying criteria in paragraphs (e)(1) and (e)(2) of this section; meets the applicable requirements of paragraphs (e)(4)(ii) through (v) of this section; and, under present or previous ownership (or both), its first cost reporting period as a LTCH begins on or after October 1, 2002.
(ii) Satellite facilities and remote locations of hospitals seeking to become new long-term care hospitals. Except as specified in paragraph (e)(4)(iii) of this section, a satellite facility (as defined in § 412.22(h)) or a remote location of a hospital (as defined in § 413.65(a)(2) of this chapter) that voluntarily reorganizes as a separate Medicare participating hospital, with or without a concurrent change in ownership, and that seeks to qualify as a new long-term care hospital for Medicare payment purposes must demonstrate through documentation that it meets the average length of stay requirement as specified under paragraphs (e)(2)(i) or (e)(2)(ii) of this section based on discharges that occur on or after the effective date of its participation under Medicare as a separate hospital.
(iii) Provider-based facility or organization identified as a satellite facility and remote location of a hospital prior to July 1, 2003. Satellite facilities and remote locations of hospitals that became subject to the provider-based status rules under § 413.65 as of July 1, 2003, that become separately participating hospitals, and that seek to qualify as long-term care hospitals for Medicare payment purposes may submit to the fiscal intermediary discharge data gathered during the period of at least 5 consecutive months of the immediate 6 months preceding the facility's separation from the main hospital for calculation of the average length of stay specified under paragraph (e)(2)(i) or paragraph (e)(2)(ii) of this section.
(iv) Qualifying period for hospitals seeking to become long-term care hospitals. A hospital may be classified as a long-term care hospital after a 6-month qualifying period, provided that the average length of stay during the period of at least 5 consecutive months of that 6-month qualifying period, calculated under paragraph (e)(2) of this section, is greater than 25 days. The 6-month qualifying period for a hospital is the 6 months immediately preceding the date of long-term care hospital classification.
(v) Special rule for hospitals seeking to become long-term care hospitals that experience a change in ownership. If a hospital seeks exclusion from the inpatient prospective payment system as a long-term care hospital and a change of ownership (as described in § 489.18 of this chapter) occurs within the period of at least 5 consecutive months of the 6-month period preceding its petition for long-term care hospital status, the hospital may be excluded from the inpatient prospective payment system as a long-term care hospital for the next cost reporting period if, for the period of at least 5 consecutive months of the 6 months immediately preceding the start of the cost reporting period for which the hospital is seeking exclusion from the inpatient prospective payment system as a long-term care hospital (including time before the change of ownership), the hospital has met the required average length of stay, has continuously operated as a hospital, and has continuously participated as a hospital in Medicare.
7. Section 412.88 is amended by adding paragraphs (a)(2)(ii)(C) and (b)(2)(iv) to read as follows:
(C) For a medical product that is a gene therapy that is indicated and used specifically for the treatment of sickle cell disease and approved for new technology add-on payments in the FY 2025 IPPS/LTCH PPS final rule, for discharges occurring on or after October 1, 2024, if the costs of the discharge (determined by applying the operating cost-to-charge ratios as described in § 412.84(h)) exceed the full DRG payment, an additional amount equal to the lesser of—
( 1 ) 75 percent of the costs of the new medical service or technology; or
( 2 ) 75 percent of the amount by which the costs of the case exceed the standard DRG payment.
(iv) For discharges occurring on or after October 1, 2024, for a medical product that is a gene therapy that is indicated and used specifically for the treatment of sickle cell disease and approved for new technology add-on payments in the FY 2025 IPPS/LTCH PPS final rule, 75 percent of the estimated costs of the new medical service or technology.
8. Section 412.90 is amended by revising paragraph (j) to read as follows:
(j) Medicare-dependent, small rural hospitals. For cost reporting periods beginning on or after April 1, 1990, and before October 1, 1994, and for ( print page 69911) discharges occurring on or after October 1, 1997 and before January 1, 2025, CMS adjusts the prospective payment rates for inpatient operating costs determined under subparts D and E of this part if a hospital is classified as a Medicare-dependent, small rural hospital.
9. Section 412.96 is amended by revising paragraph (c)(2)(ii) to read as follows:
(ii) For cost reporting periods beginning on or after January 1, 1986, an osteopathic hospital, recognized by the American Osteopathic Healthcare Association (or any successor organization), that is located in a rural area must have at least 3,000 discharges during its cost reporting period that began during the same fiscal year as the cost reporting periods used to compute the regional median discharges under paragraph (i) of this section to meet the number of discharges criterion. A hospital applying for rural referral center status under the number of discharges criterion in this paragraph must demonstrate its status as an osteopathic hospital.
10. Section 412.101 is amended by revising paragraphs (b)(2)(i) and (iii), (c)(1), and (c)(3) introductory text to read as follows:
(i) For FY 2005 through FY 2010, the portion of FY 2025 beginning on January 1, 2025 and subsequent fiscal years, a hospital must have fewer than 200 total discharges, which includes Medicare and non-Medicare discharges, during the fiscal year, based on the hospital's most recently submitted cost report, and be located more than 25 road miles (as defined in paragraph (a) of this section) from the nearest “subsection (d)” (section 1886(d) of the Act) hospital.
(iii) For FY 2019 through FY 2024 and the portion of FY 2025 beginning on October 1, 2024, and ending on December 31, 2024, a hospital must have fewer than 3,800 total discharges, which includes Medicare and non-Medicare discharges, during the fiscal year, based on the hospital's most recently submitted cost report, and be located more than 15 road miles (as defined in paragraph (a) of this section) from the nearest “subsection (d)” (section 1886(d) of the Act) hospital.
(1) For FY 2005 through FY 2010, the portion of FY 2025 beginning on January 1, 2025, and subsequent fiscal years, the adjustment is an additional 25 percent for each Medicare discharge.
(3) For FY 2019 through FY 2024 and the portion of FY 2025 beginning on October 1, 2024, and ending on December 31, 2024, the adjustment is as follows:
11. Section 412.103 is amended by revising paragraph (a)(1) to read as follows:
(1) The hospital is located in a rural census tract of a Metropolitan Statistical Area (MSA) as determined under the most recent version of the Goldsmith Modification, using the Rural-Urban Commuting Area codes and additional criteria, as determined by the Federal Office of Rural Health Policy (FORHP) of the Health Resources and Services Administration (HRSA), which is available at the web link provided in the most recent Federal Register notice issued by HRSA defining rural areas.
12. Section 412.104 is amended by revising paragraphs (b)(2) through (4) to read as follows:
(2)(i) Effective for cost reporting periods beginning before October 1, 2024, the estimated weekly cost of dialysis is the average number of dialysis sessions furnished per week during the 12-month period that ended June 30, 1983, multiplied by the average cost of dialysis for the same period.
(ii) Effective for cost reporting periods beginning on or after October 1, 2024, the estimated weekly cost of dialysis is calculated as 3 dialysis sessions per week multiplied by the applicable ESRD prospective payment system (PPS) base rate (as defined in 42 CFR 413.171 ) that corresponds with the fiscal year in which the cost reporting period begins.
(3) The average cost of dialysis used for purposes of determining the estimated weekly cost of dialysis for cost reporting periods beginning before October 1, 2024, includes only those costs determined to be directly related to the renal dialysis services. (These costs include salary, employee health and welfare, drugs, supplies, and laboratory services.)
(4) Effective for cost reporting periods beginning before October 1, 2024, the average cost of dialysis is reviewed and adjusted, if appropriate, at the time the composite rate reimbursement for outpatient dialysis is reviewed.
13. Section 412.105 is amended by adding paragraph (f)(1)(iv)(C)( 4 ) to read as follows:
( 4 ) Effective for portions of cost reporting periods beginning on or after July 1, 2026, a hospital may qualify to receive an increase in its otherwise applicable FTE resident cap if the criteria specified in § 413.79(q) of this subchapter are met.
14. Section 412.106 is amended by revising paragraph (i)(1) to read as follows:
(1) Interim payments are made during the payment year to each hospital that is estimated to be eligible for payments under this section at the time of the annual final rule for the hospital inpatient prospective payment system, subject to the final determination of eligibility at the time of cost report settlement for each hospital. For FY 2025, interim uncompensated care payments are calculated based on an average of the most recent 2 years of available historical discharge data. For FY 2026 and subsequent years, interim uncompensated care payments are calculated based on an average of the most recent 3 years of available historical discharge data.
15. Section 412.108 is amended by revising paragraphs (a)(1) introductory text and (c)(2)(iii) introductory text to read as follows:
(1) General considerations. For cost reporting periods beginning on or after April 1, 1990, and ending before October 1, 1994, or for discharges occurring on or after October 1, 1997, and before January 1, 2025, a hospital is classified as a Medicare-dependent, small rural hospital if it meets all of the following conditions:
(iii) For discharges occurring during cost reporting periods (or portions thereof) beginning on or after October 1, 2006, and before January 1, 2025, 75 percent of the amount that the Federal rate determined under paragraph (c)(1) of this section is exceeded by the highest of the following:
16. Section 412.113 is amended by adding paragraph (g) to read as follows:
(g) Additional resource costs of establishing and maintaining access to buffer stocks of essential medicines. (1) Essential medicines are the 86 medicines prioritized in the report Essential Medicines Supply Chain and Manufacturing Resilience Assessment developed by the U.S. Department of Health and Human Services Office of the Assistant Secretary for Preparedness and Response and published in May of 2022, and any subsequent revisions to that list of medicines. A buffer stock of essential medicines for a hospital is a supply, for no less than a 6-month period of one or more essential medicines.
(2) The additional resource costs of establishing and maintaining access to a buffer stock of essential medicines for a hospital are the additional resource costs incurred by the hospital to directly hold a buffer stock of essential medicines for its patients or arrange contractually for such a buffer stock to be held by another entity for use by the hospital for its patients. The additional resource costs of establishing and maintaining access to a buffer stock of essential medicines does not include the resource costs of the essential medicines themselves.
(3) For cost reporting periods beginning on or after October 1, 2024, a payment adjustment to a small, independent hospital for the additional resource costs of establishing and maintaining access to buffer stocks of essential medicines is made as described in paragraph (g)(4) of this section. For purposes of this section, a small, independent hospital is a hospital with 100 or fewer beds as defined in § 412.105(b) during the cost reporting period that is not part of a chain organization, defined as a group of two or more health care facilities which are owned, leased, or through any other device, controlled by one organization.
(4) The payment adjustment is based on the estimated reasonable cost incurred by the hospital for establishing and maintaining access to buffer stocks of essential medicines during the cost reporting period.
17. Section 412.140 is amended by revising paragraphs (d)(2)(ii) and (e)(2)(vii) introductory text to read as follows:
(ii)(A) Prior to the FY 2028 payment determination, a hospital meets the eCQM validation requirement with respect to a fiscal year if it submits 100 percent of sampled eCQM measure medical records in a timely and complete manner, as determined by CMS.
(B) For the FY 2028 payment determination and later years, a hospital meets the eCQM validation requirement with respect to a fiscal year if it achieves a 75-percent score, as determined by CMS.
(vii) If the hospital has requested reconsideration on the basis that CMS concluded it did not meet the validation requirement set forth in paragraph (d) of this section, the reconsideration request must contain a detailed explanation identifying which data the hospital believes was improperly validated by CMS and why the hospital believes that such data are correct.
18. In § 412.230 amend paragraph (a)(5)(i) by removing the phrase “in the rural area of the state” and adding in its place the phrase “either in its geographic area or in the rural area of the State”.
19. Amend § 412.273 by revising paragraphs (c)(1)(ii) and (c)(2) to read as follows:
(ii) After the MGCRB issues a decision, provided that the request for withdrawal is received by the MCGRB within 45 days of the date of filing for public inspection of the proposed rule at the website of the Office of the Federal Register, or within 7 calendar days of receiving a decision of the Administrator's in accordance with § 412.278, whichever is later concerning changes to the inpatient hospital prospective payment system and proposed payment rates for the fiscal year for which the application has been filed.
(2) A request for termination must be received by the MGCRB within 45 days of the date of filing for public inspection of the proposed rule at the website of the Office of the Federal Register, or within 7 calendar days of receiving a decision of the Administrator's in accordance with § 412.278, whichever is later concerning changes to the inpatient hospital prospective payment system and proposed payment rates for the fiscal year for which the termination is to apply.
20. The authority citation for part 413 continues to read as follows:
Authority: 42 U.S.C. 1302 , 1395d(d) , 1395f(b) , 1395g , 1395l(a) , (i), and (n), 1395x(v), 1395hh, 1395rr, 1395tt, and 1395ww.
21. Section 413.75 is amended in paragraph (b), in the introductory text of the definition of “Emergency Medicare GME Affiliated Group” by removing the reference “§ 413.79(f)(6)” and adding in its place the reference “§ 413.79(f)(7)”.
22. Section 413.78 is amended by—
a. In paragraph (e)(3)(iii), removing the reference “§ 413.79(f)(6)” and adding in its place the reference “§ 413.79(f)(7)”; and
b. In paragraph (f)(3)(iii) introductory text, removing the reference ( print page 69913) “§ 413.79(f)(6)” and adding in its place the reference “§ 413.79(f)(7)”.
23. Section 413.79 is amended by—
a. Revising paragraphs (d)(6), (f)(8) and (k)(2)(i); and
b. Adding paragraph (q).
The revisions and addition read as follows:
(6) Subject to the provisions of paragraph (h) of this section, FTE residents who are displaced by the closure of either another hospital or another hospital's program are added to the FTE count after applying the averaging rules in this paragraph (d), for the receiving hospital for the duration of the time that the displaced residents are training at the receiving hospital.
(8) FTE resident cap slots added under section 126 of Public Law 116-260 and section 4122 of Public Law 117-328 may be used in a Medicare GME affiliation agreement beginning in the fifth year after the effective date of those FTE resident cap slots.
(i)(A) For rural track programs started before October 1, 2012, for the first 3 years of the rural track's existence, the rural track FTE limitation for each urban hospital will be the actual number of FTE residents, subject to the rolling average specified in paragraph (d)(7) of this section, training in the rural track at the urban hospital and the rural nonprovider site(s).
(B) For rural track programs started on or after October 1, 2012, and before October 1, 2022, prior to the start of the urban hospital's cost reporting period that coincides with or follows the start of the sixth program year of the rural track's existence, the rural track FTE limitation for each urban hospital will be the actual number of FTE residents, subject to the rolling average specified in paragraph (d)(7) of this section, training in the rural track at the urban hospital and the rural nonprovider site(s).
(C) For cost reporting periods beginning on or after October 1, 2022, before the start of the urban or rural hospital's cost reporting period that coincides with or follows the start of the sixth program year of the Rural Track Program's existence, the rural track FTE limitation for each hospital will be the actual number of FTE residents training in the Rural Track Program at the urban or rural hospital and subject to the requirements under § 413.78(g), at the rural nonprovider site(s).
(q) Determination of an increase in the otherwise applicable resident cap under section 4122 of the Consolidated Appropriations Act ( Pub. L. 117-328 ). For portions of cost reporting periods beginning on or after July 1, 2026, a hospital may receive an increase in its otherwise applicable FTE resident cap (as determined by CMS) if the hospital meets the requirements and qualifying criteria under section 1886(h)(10) of the Act and if the hospital submits an application to CMS within the timeframe specified by CMS.
24. The authority citation for part 431 continues to read as follows:
Authority: 42 U.S.C. 1302 .
25. Section 431.954 is amended by:
a. In paragraph (a)(2), removing the phrase “Improper Payments Information Act of 2002 ( Pub. L. 107-300 )” and adding in its place the phrase “Payment Integrity Information Act (PIIA) of 2019 ( Pub. L. 116-117 )”.
b. In paragraph (b)(3) by removing the words “Puerto Rico”.
26. Section 431.960 is amended in paragraph (a) by removing the phrase “Improper Payments Information Act of 2002” and adding in its place the word “PIIA”.
27. Section 431.998 is amended in paragraph (f) by removing the word “IPIA” and adding in its place the word “PIIA”.
28. The authority citation for part 482 continues to read as follows:
Authority: 42 U.S.C. 1302 , 1395hh , and 1395rr , unless otherwise noted.
29. Effective November 1, 2024, amend § 482.42 by revising paragraph (e) and removing paragraph (f) to read as follows:
(e) Respiratory illness reporting —(1) Ongoing reporting. The hospital must electronically report information on acute respiratory illnesses, including influenza, SARS-CoV-2/COVID-19, and RSV.
(i) The report must be in a standardized format and frequency specified by the Secretary.
(ii) To the extent as required by the Secretary, this report must include all of the following data elements:
(A) Confirmed infections for a limited set of respiratory illnesses, including but not limited to influenza, SARS-CoV-2/COVID-19, and RSV, among newly admitted and hospitalized patients.
(B) Total bed census and capacity, including for critical hospital units and age groups.
(C) Limited patient demographic information, including but not limited to age.
(2) Public health emergency (PHE) reporting. In the event that the Secretary has declared a national, State, or local PHE for an acute infectious illness, the hospital must also electronically report the following data elements in a standardized format and frequency specified by the Secretary:
(i) Supply inventory shortages.
(ii) Staffing shortages.
(iii) Relevant medical countermeasures and therapeutic inventories, usage, or both.
(iv) Facility structure and operating status, including hospital/ED diversion status.
30. The authority citation for part 482 continues to read as follows:
31. Effective November 1, 2024, amend § 485.640 by revising paragraph (d) and removing paragraph (e) to read as follows:
(d) Respiratory illness reporting— (1) Ongoing reporting. The CAH must electronically report information on acute respiratory illnesses, including influenza, SARS-CoV-2/COVID-19, and RSV.
(ii) To the extent as required by the Secretary, the report must include the following data elements: ( print page 69914)
(2) Public health emergency (PHE) reporting. In the event that the Secretary has declared a national, State, or local PHE for an acute infectious illness, the CAH must also electronically report the following data elements in a standardized format and frequency specified by the Secretary:
(iv) Facility structure and operating status, including CAH/ED diversion status.
32. The authority citation for part 495 continues to read as follows:
33. Section 495.24 is amended by—
a. In paragraph (f)(1)(i)(B) removing the phrase “In 2023 and subsequent years” and adding in its place the phrase “In 2023 and 2024,”; and
b. Adding paragraphs (f)(1)(i)(C) and (D).
The addition reads as follows:
(C) In 2025 and subsequent years, earn a total score of at least 70 points.
(D) In 2026 and subsequent years, earn a total score of at least 80 points.
34. The authority citation for part 512 continues to read as follows:
Authority: 42 U.S.C. 1302 , 1315a , and 1395hh .
35. Revise the heading for part 512 to read as set forth above.
36. Add subparts D and E to read as follows:
(a) Basis. This subpart implements the test of the Transforming Episode Accountability Model (TEAM) under section 1115A(b) of the Act. Except as specifically noted in this part, the regulations under this subpart do not affect the applicability of other provisions affecting providers and suppliers under Medicare FFS, including the applicability of provisions regarding payment, coverage, and program integrity.
(b) Scope. This subpart sets forth the following:
(1) Participation in TEAM.
(2) Scope of episodes being tested.
(3) Pricing methodology.
(4) Quality measures and quality reporting requirements.
(5) Reconciliation and review processes.
(6) Data sharing and other requirements
(7) Financial arrangements and beneficiary incentives.
(8) Medicare program waivers
(9) Beneficiary protections.
(10) Cooperation in model evaluation and monitoring.
(11) Audits and record retention.
(12) Rights in data and intellectual property.
(13) Monitoring and compliance.
(14) Remedial action.
(15) Limitations on review.
(16) Miscellaneous provisions on bankruptcy and other notifications.
(17) Model termination by CMS.
(18) Decarbonization and resilience initiative.
For the purposes of this part, the following definitions are applicable unless otherwise stated:
AAPM stands for Advanced Alternative Payment Model.
AAPM option means the advanced alternative payment model option of TEAM for Track 2 and Track 3 TEAM participants that provide their CMS EHR Certification ID and attest to their use of CEHRT in accordance with § 512.522.
ACO means an accountable care organization, as defined at § 425.20 of this chapter.
ACO participant has the meaning set forth in § 425.20 of this chapter.
ACO provider/supplier has the meaning set forth in § 425.20 of this chapter. ( print page 69915)
Acute care hospital means a provider subject to the prospective payment system specified in § 412.1(a)(1) of this chapter.
Age bracket risk adjustment factor means the coefficient of risk associated with a patient's age bracket, calculated as described in § 512.545(a)(1).
Aggregated reconciliation target price refers to the sum of the reconciliation target prices for all episodes attributed to a given TEAM participant for a given performance year.
Alignment payment means a payment from a TEAM collaborator to a TEAM participant under a sharing arrangement, for the sole purpose of sharing the TEAM participant's responsibility for making repayments to Medicare.
AMI stands for acute myocardial infarction
Anchor hospitalization means the initial hospital stay upon admission for an episode category included in TEAM, as described in § 512.525(c), for which the institutional claim is billed through the inpatient prospective payment system (IPPS).
Anchor procedure means a procedure related to an episode category, as described in § 512.525(c), included in TEAM that is permitted and paid for by Medicare when performed in a hospital outpatient department (HOPD) and billed through the Hospital Outpatient Prospective Payment System (OPPS).
ADI stands for Area Deprivation Index.
APM stands for Alternative Payment Model.
APM Entity means an entity as defined in § 414.1305 of this chapter.
Baseline episode spending refers to total episode spending by all providers and suppliers associated with a given MS-DRG/HCPCS episode type for all hospitals in a given region during the baseline period.
Baseline period means the 3-year historical period used to construct the preliminary target price and reconciliation target price for a given performance year.
Baseline year means any one of the 3 years included in the baseline period.
Benchmark price means average standardized episode spending by all providers and suppliers associated with a given MS-DRG/HCPCS episode type for all hospitals in a given region during the applicable baseline period.
Beneficiary means an individual who is enrolled in Medicare FFS.
Beneficiary who is dually eligible means a beneficiary enrolled in both Medicare and full Medicaid benefits.
BPCI stands for Bundled Payments for Care Improvement, which was an episode_based payment initiative with four models tested by the CMS Innovation Center from April 2013 to September 2018.
BPCI Advanced stands for the Bundled Payments for Care Improvement Advanced Model, which is an episode-based payment model tested by the CMS Innovation Center from October 2018 to December 2025.
CABG (Coronary Artery Bypass Graft Surgery) means any coronary revascularization procedure paid through the IPPS under MS-DRGs 231-236, including both elective CABG and CABG procedures performed during initial acute myocardial infarction (AMI) treatment.
CCN stands for CMS certification number.
CEHRT means certified electronic health record technology that meets the requirements set forth in § 414.1305 of this chapter.
Change in control means any of the following:
(1) The acquisition by any “person” (as this term is used in sections 13(d) and 14(d) of the Securities Exchange Act of 1934) of beneficial ownership (within the meaning of Rule 13d-3 promulgated under the Securities Exchange Act of 1934), directly or indirectly, of voting securities of the TEAM participant representing more than 50 percent of the TEAM participant's outstanding voting securities or rights to acquire such securities.
(2) The acquisition of the TEAM participant by any individual or entity.
(3) The sale, lease, exchange, or other transfer (in one transaction or a series of transactions) of all or substantially all of the assets of the TEAM participant.
(4) The approval and completion of a plan of liquidation of the TEAM participant, or an agreement for the sale or liquidation of the TEAM participant.
CJR stands for the Comprehensive Care for Joint Replacement Model, which is an episode-based payment model tested by the CMS Innovation Center from April 2016 to December 2024.
Clinician engagement list means the list of eligible clinicians or MIPS eligible clinicians that participate in TEAM activities and have a contractual relationship with the TEAM participant, and who are not listed on the financial arrangements list, as described in § 512.522(c).
CMS Electronic Health Record (EHR) Certification ID means the identification number that represents the combination of Certified Health Information Technology that is owned and used by providers and hospitals to provide care to their patients and is generated by the Certified Health Information Technology Product List.
Collaboration agent means an individual or entity that is not a TEAM collaborator and that is either of the following:
(1) A member of a PGP, NPPGP, or TGP that has entered into a distribution arrangement with the same PGP, NPPGP, or TGP in which he or she is an owner or employee, and where the PGP, NPPGP, or TGP is a TEAM collaborator.
(2) An ACO participant or ACO provider/supplier that has entered into a distribution arrangement with the same ACO in which it is participating, and where the ACO is a TEAM collaborator.
Composite quality score (CQS) means a score computed for each TEAM participant to summarize the TEAM participant's level of quality performance and improvement on specified quality measures as described in § 512.547.
Core-based statistical area (CBSA) means a statistical geographic entity defined by the Office of Management and Budget (OMB) consisting of the county or counties associated with at least one core (urbanized area or urban cluster) of at least 10,000 population, plus adjacent counties having a high degree of social and economic integration with the core as measured through commuting ties with the counties containing the core.
CORF stands for comprehensive outpatient rehabilitation facility.
Covered services means the scope of health care benefits described in sections 1812 and 1832 of the Act for which payment is available under Part A or Part B of Title XVIII of the Act.
Critical access hospital (CAH) means a hospital designated under subpart F of part 485 of this chapter.
CQS adjustment amount means the amount subtracted from the positive or negative reconciliation amount to generate the reconciliation payment or repayment amount.
CQS adjustment percentage means the percentage CMS applies to the positive or negative reconciliation amount based on the TEAM participant's CQS performance.
CQS baseline period means the time period used to benchmark quality measure performance.
Days means calendar days.
Decarbonization and Resilience Initiative means an initiative for TEAM participants that includes technical assistance on decarbonization and a voluntary reporting program where TEAM participants may annually report ( print page 69916) metrics and questions related to emissions in accordance with § 512.598.
Descriptive TEAM materials and activities means general audience materials such as brochures, advertisements, outreach events, letters to beneficiaries, web pages, mailings, social media, or other materials or activities distributed or conducted by or on behalf of the TEAM participant or its downstream participants when used to educate, notify, or contact beneficiaries regarding TEAM. All of the following communications are not descriptive TEAM materials and activities:
(1) Communications that do not directly or indirectly reference TEAM (for example, information about care coordination generally).
(2) Information on specific medical conditions.
(3) Referrals for health care items and services, except as required by § 512.564.
(4) Any other materials that are excepted from the definition of “marketing” as that term is defined at 45 CFR 164.501 .
Discount factor means a set percentage included in the preliminary target price and reconciliation target price intended to reflect Medicare's potential savings from TEAM.
Distribution arrangement means a financial arrangement between a TEAM collaborator that is an ACO, PGP, NPPGP, or TGP and a collaboration agent for the sole purpose of distributing some or all of a gainsharing payment received by the ACO, PGP, NPPGP, or TGP.
Distribution payment means a payment from a TEAM collaborator that is an ACO, PGP, NPPGP, or TGP to a collaboration agent, under a distribution arrangement, composed only of gainsharing payments.
DME stands for durable medical equipment.
Downstream collaboration agent means an individual who is not a TEAM collaborator or a collaboration agent and who is a member of a PGP, NPPGP, or TGP that has entered into a downstream distribution arrangement with the same PGP, NPPGP, or TGP in which he or she is an owner or employee, and where the PGP, NPPGP, or TGP is a collaboration agent.
Downstream distribution arrangement means a financial arrangement between a collaboration agent that is both a PGP, NPPGP, or TGP and an ACO participant and a downstream collaboration agent for the sole purpose of sharing a distribution payment received by the PGP, NPPGP, or TGP.
Downstream participant means an individual or entity that has entered into a written arrangement with a TEAM participant, TEAM collaborator, collaboration agent, or downstream collaboration agent under which the downstream participant engages in one or more TEAM activities.
EHR stands for electronic health record.
Eligible clinician means a clinician as defined in § 414.1305 of this chapter.
Episode category means one of the five episodes tested in TEAM as described at § 512.525(d).
Episode means all Medicare Part A and B items and services described in § 512.525(e) (and excluding the items and services described in § 512.525(f)) that are furnished to a beneficiary described in § 512.535 during the time period that begins on the date of the beneficiary's admission to an anchor hospitalization or the date of the anchor procedure, as described at § 512.525(c), and ends on the 30th day following the date of discharge from the anchor hospitalization or anchor procedure, with the date of discharge or date of the anchor procedure itself being counted as the first day in the 30-day post-discharge period, as described at § 512.537. If an anchor hospitalization is initiated on the same day as or in the 3 days following an outpatient procedure that could initiate an anchor procedure for the same episode category, the outpatient procedure initiates an anchor hospitalization and the anchor hospitalization start date is that of the outpatient procedure.
Essential access community hospital means a hospital as defined under § 412.109 of this chapter.
Final normalization factor refers to the national mean of the benchmark price for each MS-DRG/HCPCS episode type divided by the national mean of the risk-adjusted benchmark price for the same MS-DRG/HCPCS episode type.
Financial arrangements list means the list of eligible clinicians or MIPS eligible clinicians that have a financial arrangement with the TEAM participant, TEAM collaborator, collaboration agent, and downstream collaboration agent, as described in § 512.522(b).
Gainsharing payment means a payment from a TEAM participant to a TEAM collaborator, under a sharing arrangement, composed of only reconciliation payments, internal cost savings, or both.
HCPCS stands for Healthcare Common Procedure Coding System, which is used to bill for items and services.
Health disparities mean preventable differences in the burden of disease, injury, violence, or opportunities to achieve optimal health, health quality, or health outcomes that are experienced by one or more underserved communities within the TEAM participant's population of TEAM beneficiaries that the TEAM participant will aim to reduce.
Health equity goal means a targeted outcome relative to health equity plan performance measures.
Health equity plan means a document that identifies health equity goals, intervention strategies, and performance measures to improve health disparities identified within the TEAM participant's population of TEAM beneficiaries that the TEAM participant will aim to reduce as described in § 512.563.
Health equity plan intervention strategy means the initiative the TEAM participant creates and implements to reduce the identified health disparities as part of the health equity plan.
Health equity plan performance measure means a quantitative metric that the TEAM participant uses to measure changes in health disparities arising from the health equity plan intervention strategies.
Health-related social need means an unmet, adverse social condition that can contribute to poor health outcomes and is a result of underlying social determinants of health, which refer to the conditions in the environments where people are born, live, learn, work, play, worship, and age that affect a wide range of health, functioning, and quality-of-life outcomes and risks.
HHA means a Medicare-enrolled home health agency.
High-cost outlier cap refers to the 99th percentile of regional spending for a given MS-DRG/HCPCS episode type in a given region, which is the amount at which episode spending would be capped for purposes of determining baseline and performance year episode spending.
Hospital means a hospital as defined in section 1886(d)(1)(B) of the Act.
Hospital discharge planning means the standards set forth in § 482.43 of this chapter.
ICD-CM stands for International Classification of Diseases, Clinical Modification.
Internal cost savings means the measurable, actual, and verifiable cost savings realized by the TEAM participant resulting from care redesign undertaken by the TEAM participant in connection with providing items and services to TEAM beneficiaries within an episode. Internal cost savings does not include savings realized by any individual or entity that is not the TEAM participant. ( print page 69917)
IPF stands for inpatient psychiatric facility.
IPPS stands for Inpatient Prospective Payment System, which is the payment system for subsection (d) hospitals as defined in section 1886(d)(1)(B) of the Act.
IRF stands for inpatient rehabilitation facility.
LIS stands for Medicare Part D Low-Income Subsidy.
Lower-Extremity Joint Replacement (LEJR) means any hip, knee, or ankle replacement that is paid under MS-DRG 469, 470, 521, or 522 through the IPPS or HCPCS code 27447, 27130, or 27702 through the OPPS.
LTCH stands for long-term care hospital.
Major Bowel Procedure means any small or large bowel procedure paid through the IPPS under MS-DRG 329-331.
Mandatory CBSA means a core-based statistical area selected by CMS in accordance with § 512.515 where all eligible hospitals are required to participate in TEAM.
MDC stands for Major Diagnostic Category.
Medically necessary means reasonable and necessary for the diagnosis or treatment of an illness or injury, or to improve the functioning of a malformed body member.
Medicare Severity Diagnosis-Related Group (MS-DRG) means, for the purposes of this model, the classification of inpatient hospital discharges updated in accordance with § 412.10 of this chapter.
Medicare-dependent, small rural hospital (MDH) means a specific type of hospital that meets the classification criteria specified under § 412.108 of this chapter.
Member of the NPPGP or NPPGP member means a nonphysician practitioner or therapist who is an owner or employee of an NPPGP and who has reassigned to the NPPGP his or her right to receive Medicare payment.
Member of the PGP or PGP member means a physician, nonphysician practitioner, or therapist who is an owner or employee of the PGP and who has reassigned to the PGP his or her right to receive Medicare payment.
Member of the TGP or TGP member means a therapist who is an owner or employee of a TGP and who has reassigned to the TGP his or her right to receive Medicare payment.
MIPS stands for Merit-based Incentive Payment System.
MIPS eligible clinician means a clinician as defined in § 414.1305 of this chapter.
Model performance period means the 60-month period from January 1, 2026, to December 31, 2030, during which TEAM is being tested and the TEAM participant is held accountable for spending and quality.
Model start date means January 1, 2026, the start of the model performance period.
MS-DRG/HCPCS episode type refers to the subset of episodes within an episode category that are associated with a given MS-DRG/HCPCS, as set forth at § 512.540(a)(1).
Non-AAPM option means the option of TEAM for TEAM participants in Track 1 or for TEAM participants in Track 2 or Track 3 that do not attest to use of CEHRT as described in § 512.522.
Nonphysician practitioner means one of the following:
(1) A physician assistant who satisfies the qualifications set forth at § 410.74(a)(2)(i) and (ii) of this chapter.
(2) A nurse practitioner who satisfies the qualifications set forth at § 410.75(b) of this chapter.
(3) A clinical nurse specialist who satisfies the qualifications set forth at § 410.76(b) of this chapter.
(4) A certified registered nurse anesthetist (as defined at § 410.69(b) of this chapter).
(5) A clinical social worker (as defined at § 410.73(a) of this chapter).
(6) A registered dietician or nutrition professional (as defined at § 410.134 of this chapter).
NPI stands for National Provider Identifier.
NPPGP stands for Non-Physician Provider Group Practice, which means an entity that is enrolled in Medicare as a group practice, includes at least one owner or employee who is a nonphysician practitioner, does not include a physician owner or employee, and has a valid and active TIN.
NPRA stands for Net Payment Reconciliation Amount, which means the dollar amount representing the difference between the reconciliation target price and performance year spending, after adjustments for quality and stop-gain/stop-loss limits, but prior to the post-episode spending adjustment.
OIG stands for the Department of Health and Human Services Office of the Inspector General.
OP means an outpatient procedure for which the institutional claim is billed by the hospital through the OPPS.
OPPS stands for the Outpatient Prospective Payment System.
PAC stands for post-acute care.
PBPM stands for per-beneficiary-per-month.
Performance year means a 12-month period beginning on January 1 and ending on December 31 of each year during the model performance period.
Performance year spending means the sum of standardized Medicare claims payments during the performance year for the items and services that are included in the episode in accordance with § 512.525(e), excluding the items and services described in § 512.525(f).
PGP stands for physician group practice.
Physician has the meaning set forth in section 1861(r) of the Act.
Post-episode spending amount means the sum of all Medicare Parts A and B payments for items and services furnished to a beneficiary within 30 days after the end of an episode and includes the prorated portion of services that began during the episode and extended into the 30-day post-episode period.
Preliminary target price refers to the target price provided to the TEAM participant prior to the start of the performance year, which is subject to adjustment at reconciliation, as set forth at § 512.540.
Primary care services has the meaning set forth in section 1842(i)(4) of the Act.
Prospective normalization factor refers to the multiplier incorporated into the preliminary target price to ensure that the average of the total risk-adjusted preliminary target price does not exceed the average of the total non-risk adjusted preliminary target price, calculated as set forth in § 512.540(b)(6).
Prospective trend factor refers to the multiplier incorporated into the preliminary target price to estimate changes in spending patterns between the baseline period and the performance year, calculated as set forth in § 512.540(b)(7).
Provider means a “provider of services” as defined under section 1861(u) of the Act and codified in the definition of “provider” at § 400.202 of this chapter.
Provider of outpatient therapy services means an entity that is enrolled in Medicare as a provider of therapy services and furnishes one or more of the following:
(1) Outpatient physical therapy services as defined in § 410.60 of this chapter.
(2) Outpatient occupational therapy services as defined in § 410.59 of this chapter.
(3) Outpatient speech-language pathology services as defined in § 410.62 of this chapter.
QP stands for Qualifying APM Participant as defined in § 414.1305 of this chapter.
Quality-adjusted reconciliation amount refers to the dollar amount representing the difference between the reconciliation target price and ( print page 69918) performance year spending, after adjustments for quality, but prior to application of stop-gain/stop-loss limits and the post-episode spending adjustment.
Raw quality measure score means the quality measure value as obtained from the Hospital Inpatient Quality Reporting Program and the Hospital-Acquired Condition Reduction Program.
Reconciliation amount means the dollar amount representing the difference between the reconciliation target price and performance year spending, prior to adjustments for quality, stop-gain/stop-loss limits, and post-episode spending.
Reconciliation payment amount means the amount that CMS may owe to a TEAM participant after reconciliation as determined in accordance with § 512.550(g).
Reconciliation target price means the target price applied to an episode at reconciliation, as determined in accordance with § 512.545.
Region means one of the nine U.S. census divisions, as defined by the U.S. Census Bureau.
Reorganization event refers to a merger, consolidation, spin off or other restructuring that results in a new hospital entity under a given CCN.
Repayment amount means the amount that the TEAM participant may owe to Medicare after reconciliation as determined in accordance with § 512.550(g).
Retrospective trend factor refers to the multiplier incorporated into the reconciliation target price to estimate realized changes in spending patterns during the performance year, calculated as set forth in § 512.545(f).
Rural hospital means an IPPS hospital that meets one of the following criteria:
(1) Is located in a rural area as defined under § 412.64 of this chapter.
(2) Is located in a rural census tract defined under § 412.103(a)(1) of this chapter.
Safety Net hospital means an IPPS hospital that meets at least one of the following criteria:
(1) Exceeds the 75th percentile of the proportion of Medicare beneficiaries considered dually eligible for Medicare and Medicaid across all PPS acute care hospitals in the baseline period.
(2) Exceeds the 75th percentile of the proportion of Medicare beneficiaries partially or fully eligible to receive Part D low-income subsidies across all PPS acute care hospitals in the baseline period.
Scaled quality measure score means the score equal to the percentile to which the TEAM participant's raw quality measure score would have belonged in the CQS baseline period.
Sharing arrangement means a financial arrangement between a TEAM participant and a TEAM collaborator for the sole purpose of making gainsharing payments or alignment payments under TEAM.
SNF stands for skilled nursing facility.
Sole community hospital (SCH) means a hospital that meets the classification criteria specified in § 412.92 of this chapter.
Spinal Fusion means any cervical, thoracic, or lumbar spinal fusion procedure paid through the IPPS under MS-DRG 402, 426, 427, 428, 429, 430, 447, 448, 450, 451, 471, 472, or 473, or through the OPPS under HCPCS codes 22551, 22554, 22612, 22630, or 22633.
Supplier means a supplier as defined in section 1861(d) of the Act and codified at § 400.202 of this chapter.
Surgical Hip and Femur Fracture Treatment (SHFFT) means a hip fixation procedure, with or without fracture reduction, but excluding joint replacement, that is paid through the IPPS under MS-DRGs 480-482.
TAA stands for total ankle arthroplasty.
TEAM activities mean any activity related to promoting accountability for the quality, cost, and overall care for TEAM beneficiaries and performance in the model, including managing and coordinating care; encouraging investment in infrastructure and redesigned care processes for high quality and efficient service delivery; or carrying out any other obligation or duty under the model.
TEAM beneficiary means a beneficiary who meets the beneficiary inclusion criteria in § 512.535 and who is in an episode.
TEAM collaborator means an ACO or one of the following Medicare-enrolled individuals or entities that enters into a sharing arrangement:
(5) Physician.
(6) Nonphysician practitioner.
(7) Therapist in private practice.
(9) Provider of outpatient therapy services.
(11) Hospital.
(13) NPPGP.
(14) Therapy Group Practice (TGP).
TEAM data sharing agreement means an agreement entered into between the TEAM participant and CMS that includes the terms and conditions for any beneficiary-identifiable data shared with the TEAM participant under § 512.562.
TEAM HCC count refers to the TEAM Hierarchical Condition Category count, which is a categorical risk adjustment variable designed to reflect a beneficiary's overall health status during a lookback period by grouping similar diagnoses into one related category and counting the total number of diagnostic categories that apply to the beneficiary.
TEAM participant means an acute care hospital that either—
(1) Initiates episodes and is paid under the IPPS with a CCN primary address located in one of the mandatory CBSAs selected for participation in TEAM in accordance with § 512.515; or
(2) Makes a voluntary opt-in participation election to participate in TEAM in accordance with § 512.510 and is accepted to participate in TEAM by CMS.
TEAM payment means a payment made by CMS only to TEAM participants, or a payment adjustment made only to payments made to TEAM participants, under the terms of TEAM that is not applicable to any other providers or suppliers.
TEAM reconciliation report means the report prepared after each reconciliation that CMS provides to the TEAM participant notifying the TEAM participant of the outcome of the reconciliation.
TGP or therapy group practice means an entity that is enrolled in Medicare as a therapy group in private practice, includes at least one owner or employee who is a therapist in private practice, does not include an owner or employee who is a physician or nonphysician practitioner, and has a valid and active TIN.
THA means total hip arthroplasty.
Therapist means one of the following individuals as defined at § 484.4 of this chapter:
(1) Physical therapist.
(2) Occupational therapist.
(3) Speech-language pathologist.
Therapist in private practice means a therapist that—
(1) Complies with the special provisions for physical therapists in private practice in § 410.60(c) of this chapter;
(2) Complies with the special provisions for occupational therapists in private practice in § 410.59(c) of this chapter; or
(3) Complies with the special provisions for speech-language pathologists in private practice in § 410.62(c) of this chapter. ( print page 69919)
TIN stands for taxpayer identification number.
TKA stands for total knee arthroplasty.
Track 1 means a participation track in TEAM in which any TEAM participant may participate for the first performance year and only TEAM participants who are a safety net hospital, as defined in § 512.505, may participate for performance years 1 through 3 of the model. TEAM participants in Track 1 are subject to all of the following:
(1) CQS adjustment percentage described in § 512.550(d)(1)(i).
(2) Limitations on gain described in § 512.550(e)(2).
(3) The calculation of the reconciliation payment described in § 512.550(g).
Track 2 means a participation track in TEAM in which certain TEAM participants, as described in § 512.520(b)(4), may request to participate in for performance years 2 through 5. TEAM participants in Track 2 are subject to all of the following:
(1) CQS adjustment percentage described in § 512.550(d)(1)(ii).
(2) Limitations on gain and loss described in § 512.550(e)(2) and § 512.550(e)(3).
(3) The calculation of the reconciliation payment or repayment amount described in § 512.550(g).
Track 3 means a participation track in TEAM in which a TEAM participant may participate in for performance years 1 through 5. TEAM participants in Track 3 are subject to all of the following:
(1) CQS adjustment percentage described in § 512.550(d)(1)(iii).
(2) Limitations on loss and gain described in § 512.550(e)(1) and in § 512.550(e)(2).
Underserved community means a population sharing a particular characteristic, including geography, that has been systematically denied a full opportunity to participate in aspects of economic, social, and civic life.
U.S. Territories means American Samoa, the Federated States of Micronesia, Guam, the Marshall Islands, and the Commonwealth of the Northern Mariana Islands, Palau, Puerto Rico, U.S. Minor Outlying Islands, and the U.S. Virgin Islands.
Weighted scaled score means the scaled quality measure score multiplied by its normalized weight.
(a) General. Hospitals that wish to voluntarily opt-in to TEAM for the full duration of the model performance period must submit a written participation election letter as described in paragraph (d) of this section during the voluntary participation election period specified in paragraph (c) of this section.
(b) Eligibility. A hospital must not be located in a mandatory CBSA selected for TEAM participation, in accordance with § 512.515, and must satisfy one of the following criteria to be eligible for voluntary opt-in participation election—
(1) Be a participant hospital in the CJR model that participates in CJR until the last day of the last performance year, December 31, 2024; or
(2) Be a hospital participating in the BPCI Advanced model, either as a participant or downstream episode initiator, that participates in BPCI Advanced until the last day of the last performance period, December 31, 2025.
(c) Voluntary participation election period. The voluntary participation election period begins on January 1, 2025 and ends on January 31, 2025.
(d) Voluntary participation election letter. The voluntary participation election letter serves as the model participation agreement. CMS may accept the voluntary participation election letter if the letter meets all of the following criteria:
(1) Includes all of the following:
(i) Hospital name.
(ii) Hospital address.
(iii) Hospital CCN.
(iv) Hospital contact name, telephone number, and email address.
(v) Model name (TEAM).
(2) Includes a certification that the hospital will—
(i) Comply with all applicable requirements of this part and all other laws and regulations applicable to its participation in TEAM; and
(ii) Submit data or information to CMS that is accurate, complete and truthful, including, but not limited to, the participation election letter and any other data or information that CMS uses for purposes of TEAM.
(3) Is signed by the hospital administrator, chief financial officer, or chief executive officer with authority to bind the hospital.
(4) Is submitted in the form and manner specified by CMS.
(e) CMS rejection of participation letter. CMS may reject a participation election letter for reasons including, but not limited to, program integrity concerns or ineligibility, and notifies the hospital of the rejection within 30 days of the determination.
(a) General. CMS uses stratified random sampling to select the mandatory CBSAs included in TEAM.
(b) Exclusions. CMS excludes from the selection of geographic areas CBSAs that meet any of the following criteria:
(1) Are located entirely in the State of Maryland.
(2) Are located partially in Maryland, and in which more than 50 percent of the five episode categories tested in TEAM were initiated at a Maryland hospital between January 1, 2022 and June 30, 2023.
(3) Did not have at least one episode for at least one of the five episode categories tested in TEAM between January 1, 2022 and June 30, 2023.
(c) Stratification. (1) Based on the median for each of the following four metrics, CMS designates the CBSAs that are not excluded in accordance with paragraph (b) of this section as “high” and “low”:
(i) Average episode spend for a broad set of episode categories tested in the BPCI Advanced Model, as described in § 512.505, between January 1, 2022 and June 30, 2023.
(ii) Number of acute care hospitals paid under the IPPS between January 1, 2022 and June 30, 2023.
(iii) Past exposure to CMS' bundled payment models, which are Bundled Payments for Care Improvement (BPCI) Models 2, 3, and 4, as described in § 512.505, Comprehensive Care for Joint Replacement (CJR) as described in § 512.505, or BPCI Advanced between October 1, 2013 and December 31, 2022.
(iv) Number of Safety Net hospitals in 2022 that have initiated at least one episode between January 1, 2022 and June 30, 2023 for at least one of the five episode categories tested in TEAM.
(2)(i) CMS stratifies the CBSAs into mutually exclusive groups corresponding to the 16 unique combinations of these “high” and “low” designations.
(ii) CMS assigns selection probabilities ranging from 20 percent to 33.3 percent to each of the 16 strata, with a higher selection probability for strata containing CBSAs with a high number of safety net hospitals or low past exposure to bundles and a lower selection probability for all other strata.
(3)(i) CMS recategorizes outlier CBSAs in these 16 strata with a very high number of safety net hospitals into a 17th stratum.
(ii) CMS assigns a selection probability of 50 percent to the 17th stratum. ( print page 69920)
(4)(i) CMS recategorizes CBSAs still remaining in the first 16 strata with at least one hospital participating in BPCI Advanced or CJR as of January 1, 2024 or those located in the states of Vermont, Connecticut, or Hawaii into an 18th stratum.
(ii) CMS assigns a selection probability of 20 percent to the 18th stratum.
(d) Random selection into TEAM. CMS randomly selects mandatory CBSAs into TEAM from each of the 18 strata according to selection probabilities described in paragraph (c) of this section.
(a) For performance year 1: (1) Any TEAM participant may choose to participate in Track 1 or Track 3.
(2) The TEAM participant must notify CMS of its track choice, prior to performance year 1, in a form and manner and by a date specified by CMS.
(3) CMS assigns the TEAM participant to Track 1 for performance year 1 if a TEAM participant does not choose a track in the form and manner and by the date specified by CMS.
(b) For performance years 2 through 5: (1) CMS assigns a TEAM participant to participate in Track 3 unless the TEAM participant requests to participate in Track 1 or Track 2 and receives approval from CMS to participate in Track 1 or Track 2, with the exception that a TEAM participant cannot request participation in Track 1 for performance years 4 and 5.
(2) The TEAM participant must notify CMS of its Track 1 or Track 2 request prior to performance year 2, and prior to every performance year thereafter, as applicable, in a form and manner and by a date specified by CMS.
(3) CMS does not approve a TEAM participant's request to participate in Track 1 submitted in accordance with paragraph (b)(2) of this section unless the TEAM participant is a safety net hospital, as defined in § 512.505, at the time of the request.
(4) CMS does not approve a TEAM participant's request to participate in Track 2 submitted in accordance with paragraph (b)(2) of this section unless the TEAM participant is one of the following hospital types at the time of the request:
(i) Medicare-dependent hospital (as defined in § 512.505).
(ii) Rural hospital (as defined in § 512.505).
(iii) Safety Net hospital (as defined in § 512.505).
(iv) Sole community hospital (as defined in § 512.505).
(v) Essential access community hospital (as defined in § 512.505).
(5) A TEAM participant who does not notify CMS of its Track 1 or Track 2 request prior to a given performance year in the form and manner and by the date specified by CMS or who is not a safety net hospital, as defined as defined in § 512.505, or one of the hospital types specified in paragraph (b)(4) of this section at the time of the request is assigned to Track 3 for the applicable performance year.
(a) TEAM APM options. For performance years 1 through 5, a TEAM participant may choose either of the following options based on their CEHRT use and track participation:
(1) AAPM option. A TEAM participant participating in Track 2 or Track 3 may select the AAPM option by attesting in a form and manner and by a date specified by CMS to their use of CEHRT, as defined in § 414.1305 of this chapter, on an annual basis prior to the start of each performance year.
(i) A TEAM participant that selects the AAPM option as provided for in paragraph (a)(1) must provide their CMS electronic health record certification ID in a form and manner and by a date specified by CMS on annual basis prior to the end of each performance year.
(ii) A TEAM participant that selects the AAPM option as provided for in paragraph (a)(1) must retain documentation of their attestation to CEHRT use and provide access to the documentation in accordance with § 512.586.
(2) Non-AAPM option. CMS assigns the TEAM participant to the non-AAPM option if the TEAM participant is in Track 1 or if the TEAM participant is in Track 2 or Track 3 and does not attest in a form and manner and by a date specified by CMS to their use of CEHRT as defined in § 414.1305 of this chapter.
(b) Financial arrangements list. A TEAM participant with TEAM collaborators, collaboration agents, or downstream collaboration agents during a performance year must submit to CMS a financial arrangements list in a form and manner and by a date specified by CMS on a quarterly basis for each performance year. The financial arrangements list must include the following:
(1) TEAM collaborators. For each physician, nonphysician practitioner, or therapist who is a TEAM collaborator during the performance year:
(i) The name, TIN, and NPI of the TEAM collaborator.
(ii) The start date and, if applicable, end date, for the sharing arrangement between the TEAM participant and the TEAM collaborator.
(2) Collaboration agents. For each physician, nonphysician practitioner, or therapist who is a collaboration agent during the performance year:
(i) The name, TIN, and NPI of the collaboration agent and the name and TIN of the TEAM collaborator with which the collaboration agent has entered into a distribution arrangement.
(ii) The start date and, if applicable, end date, for the distribution arrangement between the TEAM collaborator and the collaboration agent.
(3) Downstream collaboration agents. For each physician, nonphysician practitioner, or therapist who is a downstream collaboration agent during the performance year:
(i) The name, TIN, and NPI of the downstream collaboration agent and the name and TIN of the collaboration agent with which the downstream collaboration agent has entered into a downstream distribution arrangement.
(ii) The start date and, if applicable, end date, for the downstream distribution arrangement between the collaboration agent and the downstream collaboration agent.
(c) Clinician engagement list. A TEAM participant must submit to CMS a clinician engagement list in a form and manner and by a date specified by CMS on a quarterly basis during each performance year. The clinician engagement list must include the following:
(1) For each physician, nonphysician practitioner, or therapist who is not on a TEAM participant's financial arrangements list during the performance year but who does have a contractual relationship with the TEAM participant and participates in TEAM activities during the performance year:
(i) The name, TIN, and NPI of the physician, nonphysician practitioner, or therapist.
(ii) The start date and, if applicable, the end date for the contractual relationship between the physician, nonphysician practitioner, or therapist and the TEAM participant.
(d) Attestation to no individuals. A TEAM participant with no individuals that meet the criteria specified in paragraphs (b)(1) through (3) of this section for the financial arrangements list or paragraph (c) of this section for the clinician engagement list must attest in a form and manner and by a date specified by CMS that there are no financial arrangements or clinician engagements to report.
(e) Documentation requirements. A TEAM participant that submits a financial arrangements list specified in paragraph (b) of this section or a clinician engagement list specified in ( print page 69921) paragraph (c) of this section must retain and provide access to the documentation in accordance with § 512.586.
(a) Time periods. All episodes must begin on or after January 1, 2026 and end on or before December 31, 2030.
(b) Episode attribution. All items and services included in the episode are attributed to the TEAM participant at which the anchor hospitalization or anchor procedure, as applicable, occurs.
(c) Episode initiation. An episode is initiated by—
(1) A beneficiary's admission to a TEAM participant for an anchor hospitalization that is paid under a MS-DRG specified in paragraph (d) of this section; or
(2) A beneficiary's receipt of an anchor procedure billed under a HCPCS code specified in paragraph (d) of this section. If an anchor hospitalization is initiated on the same day as or in the 3 days following an outpatient procedure that could initiate an anchor procedure for the same episode category, the episode start date is that of the outpatient procedure rather than the admission date, and an anchor procedure is not initiated.
(d) Episode categories. The MS-DRGs and HCPCS codes included in the episodes are as follows:
(1) Lower Extremity Joint Replacement (LEJR): (i) IPPS discharge under MS-DRG 469, 470, 521, or 522; or
(ii) OPPS claim for HCPCS codes 27447, 27130, or 27702.
(2) Surgical Hip/Femur Fracture Treatment (SHFFT). IPPS discharge under MS-DRG 480 to 482.
(3) Coronary Artery Bypass Graft Surgery (CABG). IPPS discharge under MS-DRG 231 to 236.
(4) Spinal Fusion: (i) IPPS discharge under MS-DRG 402, 426, 427, 428, 429, 430, 447, 448, 450, 451, 471, 472, 473; or
(ii) OPPS claim for HCPCS codes 22551, 22554, 22612, 22630, or 22633.
(5) Major Bowel Procedure. IPPS discharge under MS-DRG 329 to 331.
(e) Included services. All Medicare Part A and B items and services are included in the episode, except as specified in paragraph (f) of this section. These services include, but are not limited to, the following:
(1) Physicians' services.
(2) Inpatient hospital services (including hospital readmissions).
(3) IPF services.
(4) LTCH services.
(5) IRF services.
(6) SNF services.
(7) HHA services.
(8) Hospital outpatient services.
(9) Outpatient therapy services.
(10) Clinical laboratory services.
(12) Part B drugs and biologicals, except for those excluded under paragraph (f) of this section.
(13) Hospice services.
(14) Part B professional claims dated in the 3 days prior to an anchor hospitalization if a claim for the surgical procedure for the same episode category is not detected as part of the hospitalization because the procedure was performed by the TEAM participant on an outpatient basis, but the patient was subsequently admitted as an inpatient.
(f) Excluded services. The following items, services, and payments are excluded from the episode:
(1) Select items and services considered unrelated to the anchor hospitalization or the anchor procedure for episodes in the baseline period and performance year, including, but not limited to, the following:
(i) Inpatient hospital admissions for MS-DRGs that group to the following categories of diagnoses:
(A) Oncology.
(B) Trauma medical.
(C) Organ transplant.
(D) Ventricular shunt.
(ii) Inpatient hospital admissions that fall into the following Major Diagnostic Categories (MDCs):
(A) MDC 02 (Diseases and Disorders of the Eye).
(B) MDC 14 (Pregnancy, Childbirth, and Puerperium).
(C) MDC 15 (Newborns).
(D) MDC 25 (Human Immunodeficiency Virus).
(2) New technology add-on payments, as defined in part 412, subpart F of this chapter for episodes in the baseline period and performance year.
(3) Transitional pass-through payments for medical devices as defined in § 419.66 of this chapter for episodes initiated in the baseline period and performance year.
(4) Hemophilia clotting factors provided in accordance with § 412.115 of this chapter for episodes in the baseline period and performance year.
(5) Part B payments for low-volume drugs, high-cost drugs and biologicals, and blood clotting factors for hemophilia for episodes in the baseline period and performance year, billed on outpatient, carrier, and DME claims, defined as—
(i) Drug/biological HCPCS codes that are billed in fewer than 31 episodes in total across all episodes in TEAM during the baseline period;
(ii) Drug/biological HCPCS codes that are billed in at least 31 episodes in the baseline period and have a mean cost of greater than $25,000 per episode in the baseline period; and
(iii) HCPCS codes corresponding to clotting factors for hemophilia patients, identified in the quarterly average sales price file for certain Medicare Part B drugs and biologicals as HCPCS codes with clotting factor equal to 1, HCPCS codes for new hemophilia clotting factors not included in the baseline period, and other HCPCS codes identified as hemophilia.
(6) Part B payments for low-volume drugs, high-cost drugs and biologicals, and blood clotting factors for hemophilia for episodes initiated in the performance year, billed on outpatient, carrier, and DME claims, defined as—
(i) Drug/biological HCPCS codes that were not captured in the baseline period and appear in 10 or fewer episodes in the performance year;
(ii) Drug/biological HCPCS codes that were not included in the baseline period, appear in more than 10 episodes in the performance year, and have a mean cost of greater than $25,000 per episode in the performance year; and
(iii) Drug/biological HCPCS codes that were not included in the baseline period, appear in more than 10 episodes in the performance year, have a mean cost of $25,000 or less per episode in the performance year, and correspond to a drug/biological that appears in the baseline period but was assigned a new HCPCS code between the baseline period and the performance year.
(iv) HCPCS codes for new hemophilia clotting factors not included in the baseline period.
(g) TEAM exclusions List. The list of excluded MS-DRGs, MDCs, and HCPCS codes is posted on the CMS website.
(h) Updating the TEAM exclusions list. The list of excluded services is updated through rulemaking to reflect all of the following:
(1) Changes to the MS-DRGs under the IPPS.
(2) Coding changes.
(3) Other issues brought to CMS' attention.
(a) Episodes tested in TEAM include only those in which care is furnished to beneficiaries who meet all of the following criteria upon admission for an anchor procedure or anchor hospitalization:
(1) Are enrolled in Medicare Parts A and B.
(2) Are not eligible for Medicare on the basis of having end stage renal disease, as described in § 406.13 of this chapter. ( print page 69922)
(3) Are not enrolled in any managed care plan (for example, Medicare Advantage, health care prepayment plans, or cost-based health maintenance organizations).
(4) Are not covered under a United Mine Workers of America health care plan.
(5) Have Medicare as their primary payer.
(b) The episode is canceled in accordance with § 512.537(b) if at any time during the episode a beneficiary no longer meets all criteria in this section.
(a) Episode conclusion. (1) An episode ends on the 30th day following the date of the anchor procedure or the date of discharge from the anchor hospitalization, as applicable, with the date of the anchor procedure or the date of discharge from the anchor hospitalization being counted as the first day in the 30-day post-discharge period.
(b) Cancellation of an episode. The episode is canceled and is not included in the reconciliation calculation as specified in § 512.545 if any of the following occur:
(1) The beneficiary ceases to meet any criterion listed in § 512.535.
(2) The beneficiary dies during the anchor hospitalization or the outpatient stay for the anchor procedure.
(3) The episode qualifies for cancellation due to extreme and uncontrollable circumstances. An extreme and uncontrollable circumstance occurs if both of the following criteria are met:
(i) The TEAM participant has a CCN primary address that—
(A) Is located in an emergency area, as those terms are defined in section 1135(g) of the Act, for which the Secretary has issued a waiver under section 1135 of the Act; and
(B) Is located in a county, parish, or tribal government designated in a major disaster declaration or emergency disaster declaration under the Stafford Act.
(ii) The date of admission to the anchor hospitalization or the date of the anchor procedure is during an emergency period (as defined in section 1135(g) of the Act) or in the 30 days before the date that the emergency period (as defined in section 1135(g) of the Act) begins.
(a) Preliminary target price application. CMS establishes preliminary target prices for TEAM participants for each performance year of the model as follows:
(1) MS-DRG/HCPCS episode type. CMS uses the MS-DRGs and, as applicable, HCPCS codes specified in § 512.525(d) when calculating the preliminary target prices for each MS-DRG/HCPCS episode type.
(i) CMS determines a separate preliminary target price for each of the 24 MS-DRGs specified in § 512.525(d).
(ii) Preliminary target prices for a subset of the MS-DRGs specified in § 512.525(d) include certain HCPCS codes as follows:
(A) HCPCS 27130 and 27447 are included in MS-DRG 470.
(B) HCPCS 27702 is included in MS-DRG 469.
(C) HCPCS 22551 and 22554 are included in MS-DRG 473.
(D) HCPCS 22612 and 22630 are included in MS-DRG 451.
(E) HCPCS 22633 is included in MS-DRG 402.
(2) Applicable time period for preliminary target prices. CMS calculates preliminary target prices for each MS-DRG/HCPCS episode type and region for each performance year and applies the preliminary target price to each episode based on the episode's date of discharge from the anchor hospitalization or the episode's date of the anchor procedure, as applicable.
(3) Episodes that begin in one performance year and end in the subsequent performance year. CMS applies the preliminary target price to the episode based on the date of discharge from the anchor hospitalization or the date of the anchor procedure, as applicable, but reconciles the episode based on the end date of the episode.
(b) Preliminary target price calculation. (1) CMS calculates preliminary target prices based on average baseline episode spending for the region where the TEAM participant is located.
(i) The region used for calculating the preliminary target price corresponds to the U.S. Census Division associated with the primary address of the CCN of the TEAM participant, and the regional episode spending amount is based on all hospitals in the region, except as specified in § 512.540(b)(1)(ii).
(ii) In cases where a TEAM participant is located in a mandatory CBSA selected for participation in TEAM which spans more than one region, the TEAM participant and all other hospitals in the mandatory CBSA are grouped into the region where the most populous city in the mandatory CBSA is located for pricing and payment calculations.
(2) CMS uses the following baseline periods to determine baseline episode spending:
(i) Performance Year 1: Episodes beginning on January 1, 2022 through December 31, 2024.
(ii) Performance Year 2: Episodes beginning on January 1, 2023 through December 31, 2025.
(iii) Performance Year 3: Episodes beginning on January 1, 2024 through December 31, 2026.
(iv) Performance Year 4: Episodes beginning on January 1, 2025 through December 31, 2027.
(v) Performance Year 5: Episodes beginning on January 1, 2026 through December 31, 2028.
(3) CMS calculates the benchmark price as the weighted average of baseline episode spending, applying the following weights:
(i) Baseline episode spending from baseline year 1 is weighted at 17 percent.
(ii) Baseline episode spending from baseline year 2 is weighted at 33 percent.
(iii) Baseline episode spending from baseline year 3 is weighted at 50 percent.
(4) Exception for high episode spending. CMS applies a high-cost outlier cap to baseline episode spending at the 99th percentile of regional spending for each of the MS-DRG/HCPCS episode types specified in § 512.540(a)(1)(ii).
(5) Exclusion of incentive programs and add-on payments under existing Medicare payment systems. Certain Medicare incentive programs and add-on payments are excluded from baseline episode spending by using, with certain modifications, the CMS Price (Payment) Standardization Detailed Methodology used for the Medicare spending per beneficiary measure in the Hospital Value-Based Purchasing Program.
(6) Prospective normalization factor. Based on the episodes in the most recent calendar year of the baseline period, CMS calculates a prospective normalization factor, which is a multiplier that ensures that the average risk adjusted target price does not exceed the average unadjusted target price, by doing the following:
(i) CMS applies risk adjustment multipliers, as specified in § 512.545(a)(1) through (3), to the most recent baseline year episodes to calculate the estimated risk-adjusted target price for all performance year episodes.
(ii) CMS divides the mean of the preliminary target price for each episode across all hospitals and regions by the mean of the estimated risk-adjusted ( print page 69923) target price calculated in § 512.540(b)(6)(i) for the same episode types across all hospitals and regions.
(7) Prospective trend factor. CMS calculates the following:
(i) The average regional episode spending for each MS-DRG/HCPCS episode type using the most recent calendar year of the applicable baseline period.
(ii) The difference between the average regional spending for each MS-DRG/HCPCS episode type during the most recent calendar year of the baseline period and the average regional spending for each MS-DRG/HCPCS episode type during the first years of the baseline period to determine the prospective trend factor.
(8) Communication of preliminary target prices. CMS communicates the preliminary target prices for each MS-DRG/HCPCS episode type for each region to the TEAM participant before the performance year in which they apply.
(c) Discount factor. CMS incorporates an episode category specific discount factor of 1.5 percent for CABG and Major Bowel episodes and 2 percent for LEJR, SHFFT, and Spinal Fusion episodes to the TEAM participant's preliminary episode target prices intended to reflect Medicare's potential savings from TEAM.
CMS calculates the reconciliation target price as follows:
(a) CMS risk adjusts the preliminary episode target prices computed under § 512.540 at the beneficiary level using a TEAM Hierarchical Condition Category (HCC) count risk adjustment factor, an age bracket risk adjustment factor, a social need risk adjustment factor, and at the hospital level using a hospital bed size risk adjustment factor and a safety net hospital risk adjustment factor, and at the episode category-specific beneficiary level using factors specified in paragraph (a)(6)(i) through (v) of this section.
(1) The TEAM HCC count risk adjustment factor uses five variables, representing beneficiaries with zero, one, two, three, or four or more CMS-HCC conditions based on a lookback period that ends on the day prior to the anchor hospitalization or anchor procedure.
(2) The age bracket risk adjustment factor uses four variables, representing beneficiaries in the following age groups as of the first day of the episode:
(i) Less than 65 years.
(ii) 65 to less than 75 years.
(iii) 75 years to less than 85 years.
(iv) 85 years or more.
(3) The social need risk adjustment factor uses two variables, representing beneficiaries that, as of the first day of the episode—
(i) Meet one or more of the following measures of social need:
(A) State ADI above the 8th decile.
(B) National ADI above the 80th percentile.
(C) Eligibility for the low-income subsidy.
(D) Eligibility for full Medicaid benefits.
(ii) Do not meet any of the three measures of social need in § 512.545(a)(1)(iii)(A).
(4) The hospital bed size risk adjustment factor uses four variables based on the TEAM participant's characteristics:
(i) 250 beds or fewer.
(ii) 251-500 beds.
(iii) 501-850 beds.
(iv) 850 beds or more.
(5) The safety net hospital risk adjustment factor is based on the TEAM participant meeting the definition of safety net hospital, as defined in § 512.505.
(6) Episode category-specific beneficiary level risk adjustment factors represent the presence or absence in beneficiaries, as of the first day of the episode, of each of the following conditions:
(i) CABG episode category.
(A) Prior post-acute care use.
(B) HCC 18: Diabetes with Chronic Complications.
(C) HCC 46: Severe Hematological Disorders.
(D) HCC 58: Major Depressive, Bipolar, and Paranoid Disorders.
(E) HCC 84: Cardio-Respiratory Failure and Shock.
(F) HCC 85: Congestive Heart Failure.
(G) HCC 86: Acute Myocardial Infarction.
(H) HCC 96: Specified Heart Arrhythmias.
(I) HCC 103: Hemiplegia/Hemiparesis.
(J) HCC 111: Chronic Obstructive Pulmonary Disease.
(K) HCC 112: Fibrosis of Lung and Other Chronic Lung Disorders.
(L) HCC 134: Dialysis Status.
(ii) LEJR episode category.
(A) Ankle procedure or reattachment, partial hip procedure, partial knee arthroplasty, total hip arthroplasty or hip resurfacing procedure, and total knee arthroplasty.
(B) Disability as the original reason for Medicare enrollment.
(C) Dementia without complications.
(D) Prior post-acute care use.
(E) HCC 8: Metastatic Cancer and Acute Leukemia.
(F) HCC 18: Diabetes with Chronic Complications.
(G) HCC 22: Morbid Obesity.
(H) HCC 58: Major Depressive, Bipolar, and Paranoid Disorders.
(I) HCC 78: Parkinson's and Huntington's Diseases.
(J) HCC 85: Congestive Heart Failure.
(K) HCC 86: Acute Myocardial Infarction.
(L) HCC 103: Hemiplegia/Hemiparesis.
(M) HCC 111: Chronic Obstructive Pulmonary Disease.
(N) HCC 112: Fibrosis of Lung and Other Chronic Lung Disorders.
(O) HCC 134: Dialysis Status.
(P) HCC 170: Hip Fracture/Dislocation.
(iii) Major Bowel Procedure episode category.
(A) Long-term institutional care use.
(B) HCC 11: Colorectal, Bladder, and Other Cancers.
(C) HCC 18: Diabetes with Chronic Complications.
(D) HCC 21: Protein-Calorie Malnutrition.
(E) HCC 33: Intestinal Obstruction/Perforation.
(F) HCC 82: Respirator Dependence/Tracheostomy Status.
(G) HCC 85: Congestive Heart Failure.
(H) HCC 86: Acute Myocardial Infarction.
(M) HCC 188: Artificial Openings for Feeding or Elimination.
(iv) SHFFT episode category.
(A) HCC 18: Diabetes with Chronic Complications.
(B) HCC 22: Morbid Obesity.
(C) HCC 82: Respirator Dependence/Tracheostomy Status.
(D) HCC 83: Respiratory Arrest.
(M) HCC 157: Pressure Ulcer of Skin with Necrosis Through to Muscle, Tendon, or Bone.
(N) HCC 158: Pressure Ulcer of Skin with Full Thickness Skin Loss.
(O) HCC 161: Chronic Ulcer of Skin, Except Pressure. ( print page 69924)
(v) Spinal Fusion episode category.
(B) HCC 8: Metastatic Cancer and Acute Leukemia.
(D) HCC 22: Morbid Obesity.
(E) HCC 40: Rheumatoid Arthritis and Inflammatory Connective Tissue Disease.
(F) HCC 58: Major Depressive, Bipolar, and Paranoid Disorders.
(I) HCC 96: Specified Heart Arrhythmias.
(J) HCC 103: Hemiplegia/Hemiparesis.
(K) HCC 111: Chronic Obstructive Pulmonary Disease.
(L) HCC 112: Fibrosis of Lung and Other Chronic Lung Disorders.
(M) HCC 134: Dialysis Status.
(b) All risk adjustment factors are computed prior to the start of the performance year via a linear regression analysis. The regression analysis is computed using 3 years of claims data as follows:
(1) For performance year 1, CMS uses claims data with dates of service dated January 1, 2022 to December 31, 2024.
(2) For performance year 2, CMS uses claims data with dates of service dated January 1, 2023 to December 31, 2025.
(3) For performance year 3, CMS uses claims data with dates of service dated January 1, 2024 to December 31, 2026.
(4) For performance year 4, CMS uses claims data with dates of service dated January 1, 2025 to December 31, 2027.
(5) For performance year 5, CMS uses claims data with dates of service dated January 1, 2026 to December 30, 2028.
(c) The annual linear regression analysis produces exponentiated coefficients to determine the anticipated marginal effect of each risk adjustment factor on episode costs. CMS transforms, or exponentiates, these coefficients, and the resulting coefficients are the beneficiary and hospital-level risk adjustment factors, specified in paragraphs (a)(1) through (6) of this section, that would be used during reconciliation for the subsequent performance year.
(d) At the time of reconciliation, the preliminary target prices computed under § 512.540 are risk adjusted by applying the applicable beneficiary level and hospital-level risk adjustment factors specific to the beneficiary in the episode, as set forth in paragraphs (a)(1) through (6) of this section.
(e) The risk-adjusted preliminary target prices are normalized at reconciliation to ensure that the average of the total risk-adjusted preliminary target price does not exceed the average of the total non-risk adjusted preliminary target price.
(1) The final normalization factor at reconciliation—
(i) Is the national mean of the benchmark price for each MS-DRG/HCPCS episode type divided by the national mean of the risk-adjusted benchmark price for the same MS-DRG/HCPCS episode type.
(ii) As applied, cannot exceed ±5 percent of the prospective normalization factor (as specified in § 512.540(b)(6)).
(2) CMS applies the final normalization factor to the previously calculated, beneficiary and provider level, risk-adjusted target prices specific to each region and MS-DRG/HCPCS episode type.
(f) Retrospective trend factor. CMS calculates the average regional capped performance year episode spending for each MS-DRG/HCPCS episode type divided by the average regional capped baseline period episode spending for each MS-DRG/HCPCS episode type.
(1) The retrospective trend factor is capped so that the maximum difference cannot exceed ±3 percent of the prospective trend factor (as specified in § 512.540(b)(7)).
(2) CMS applies the capped retrospective trend factor to the previously calculated normalized, risk adjusted target prices specific to each region and MS-DRG/HCPCS episode type, as specified in paragraph (e)(2) of this section, to calculate the reconciliation target prices, which are compared to performance year spending at reconciliation, as specified in § 512.550(c).
(a) Quality measures. CMS calculates the quality measures used to evaluate the TEAM participant's performance using Medicare claims data or patient-reported outcomes data that TEAM participants report under the Hospital Inpatient Quality Reporting Program and the Hospital-Acquired Condition Reduction Program. The following quality measures and CQS baseline periods are used for public reporting and for determining the TEAM participant's CQS as described in paragraph (b) of this section:
(1) For performance year 1:
(i) For all episode categories: Hybrid Hospital-Wide All-Cause Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356) with a CY 2025 CQS baseline period;
(ii) For all episode categories: CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135) with a CY 2025 CQS baseline period; and
(iii) For LEJR episodes: Hospital-Level Total Hip and/or Total Knee Arthroplasty (THA/TKA) Patient-Reported Outcome-Based Performance Measure (PRO-PM) (CMIT ID #1618) with a CY 2025 CQS baseline period.
(2) For performance years 2 through 5:
(ii) For all episode categories: Hospital Harm—Falls with Injury (CMIT ID #1518) with a CY 2026 CQS baseline period;
(iii) For all episode categories: Hospital Harm—Postoperative Respiratory Failure (CMIT ID #1788) with a CY 2026 CQS baseline period;
(iv) For all episode categories: Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) with a CY 2026 CQS baseline period; and
(v) For LEJR episodes: Hospital-Level Total Hip and/or Total Knee Arthroplasty (THA/TKA) Patient-Reported Outcome-Based Performance Measure (PRO-PM) (CMIT ID #1618) with a CY 2025 CQS baseline period.
(b) Calculation of the composite quality score (CQS). (1) CMS converts the TEAM participant's raw quality measure score for the performance year into a scaled quality measure score by comparing the raw quality measure score to the distribution of raw quality measure score percentiles among a national cohort of hospitals, consisting of TEAM participants and hospitals not participating in TEAM, in the CQS baseline period.
(i) CMS assigns a scaled quality measure score equal to the percentile to which the TEAM Participant's raw quality measure score would have belonged in the CQS baseline period.
(A) CMS assigns the higher scaled quality measure score if the TEAM participant's raw quality measure score straddles two percentiles in the CQS baseline period.
(B) For the Hospital-Level Total Hip and/or Total Knee Arthroplasty (THA/TKA) Patient-Reported Outcome-Based Performance Measure (PRO-PM) (CMIT ID #1618):
( 1 ) CMS assigns a scaled quality measure score of 100 if the TEAM participant's raw quality measure score ( print page 69925) is greater than the maximum of the raw quality measure scores in the CQS baseline period.
( 2 ) CMS assigns a scaled quality measure score of 0 if the raw quality measure score is less than the minimum of the raw quality measure scores in the baseline period.
(C) For the Hybrid Hospital-Wide All-Cause Readmission Measure with Claims and Electronic Health Record Data (CMIT ID #356) measure, the CMS Patient Safety and Adverse Events Composite (CMS PSI 90) (CMIT ID #135) measure, the Hospital Harm—Falls with Injury (CMIT ID #1518) measure, the Hospital Harm—Postoperative Respiratory Failure (CMIT ID #1788) measure, and the Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) (CMIT ID #134) measure:
( 1 ) CMS assigns a scaled quality measure score of 0 if the TEAM participant has a raw quality measure score greater than the maximum of the raw quality measure scores in the CQS baseline period.
( 2 ) CMS assigns a scaled quality measure score of 100 if the TEAM participant has a raw quality score less than the minimum of the raw scores in the CQS baseline period.
(D) CMS does not assign a scaled quality measure score if the TEAM participant has no raw quality measure score.
(2) CMS calculates a normalized weight for each quality measure by dividing the TEAM participant's volume of attributed episodes for a given quality measure by the total volume of all the TEAM participant's attributed episodes.
(3) CMS calculates a weighted scaled score for each quality measure by multiplying each quality measure's scaled quality measure score, computed under paragraph (b)(2) of this section, by its normalized weight, computed under paragraph (b)(3) of this section.
(4) CMS sums each quality measure's weighted scaled score, computed under paragraph (b)(4) of this section, to construct the CQS.
(c) Display of quality measures. CMS does all of the following:
(1) Displays quality measure results on the publicly available CMS website that is specific to TEAM, in a form and manner consistent with other publicly reported measures.
(2) Shares quality measures with the TEAM participant prior to display on the CMS website.
(3) Uses the following time periods to share quality measure performance:
(i) Quality measure performance in performance year 1 is reported in 2027.
(ii) Quality measure performance in performance year 2 is reported in 2028.
(iii) Quality measure performance in performance year 3 is reported in 2029.
(iv) Quality measure performance in performance year 4 is reported in 2030.
(v) Quality measure performance in performance year 5 is reported in 2031.
(a) General. Providers and suppliers furnishing items and services included in the episode bill for such items and services in accordance with existing Medicare rules.
(b) Reconciliation process. Six months after the end of each performance year, CMS does the following:
(1) Performs a reconciliation calculation to establish a reconciliation payment or repayment amount for each TEAM participant.
(2) For TEAM participants that experience a reorganization event in which one or more hospitals reorganize under the CCN of a TEAM participant, performs—
(i) Separate reconciliation calculations for each predecessor TEAM participant for episodes where the anchor hospitalization admission or the anchor procedure occurred before the effective date of the reorganization event; and
(ii) Reconciliation calculations for each new or surviving TEAM participant for episodes where the anchor hospitalization admission or anchor procedure occurred on or after the effective date of the reorganization event.
(c) Calculation of the reconciliation amount. CMS compares the reconciliation target prices described in § 512.545 and the TEAM participant's performance year spending to establish a reconciliation amount for the TEAM participant for each performance year as follows:
(1) CMS determines the performance year spending for each episode included in the performance year (other than episodes that have been canceled in accordance with § 512.537(b)) using claims data that is available 6 months after the end of the performance year.
(2) CMS calculates and applies the high-cost outlier cap for performance year episode spending by applying the calculation described in § 512.540(b)(4) to performance year episode spending.
(3) CMS applies the adjustments specified in § 512.545 to the preliminary target prices computed in accordance with § 512.540 to calculate the reconciliation target prices.
(4) CMS aggregates the reconciliation target prices computed in accordance with paragraph (c)(3) of this section for all episodes included in the performance year (other than episodes that have been canceled in accordance with § 512.537(b)).
(5) CMS subtracts the performance year spending amount determined under paragraph (c)(1-2) of this section from the aggregated reconciliation target price amount determined under paragraph (c)(4) of this section to determine the reconciliation amount.
(d) Calculation of the quality-adjusted reconciliation amount. CMS adjusts the reconciliation amount based on the Composite Quality Score as follows:
(1) CMS calculates a CQS adjustment percentage based on a TEAM participant's CQS, computed in accordance with § 512.547(b).
(i) CMS applies a CQS adjustment percentage up to 10 percent for positive reconciliation amounts for TEAM participants in Track 1.
(ii) CMS applies a CQS adjustment percentage up to 10 percent for positive reconciliation amounts and up to 15 percent for negative reconciliation amounts for TEAM participants in Track 2.
(iii) CMS applies a CQS adjustment percentage up to 10 percent for positive reconciliation amounts and up to 10 percent for negative reconciliation amounts for TEAM participants in Track 3.
(2) CMS multiplies the CQS adjustment percentage, computed under paragraph (d)(1) of this section, by the TEAM participant's positive or negative reconciliation amount calculated in paragraph (c) of this section to construct the CQS adjustment amount.
(3) CMS subtracts the CQS adjustment amount, computed from paragraph (d)(2) of this section, from the positive or negative reconciliation amount calculated in paragraph (c) of this section to construct the quality-adjusted reconciliation amount.
(e) Calculation of the net payment reconciliation amount (NPRA). CMS applies stop-loss and stop gain limits to the quality-adjusted reconciliation amount computed in paragraph (d) of this section to calculate the NPRA as follows:
(1) Limitation on loss. For TEAM participants in Track 3, except as provided in paragraph (e)(3) of this section, the repayment amount for a performance year cannot exceed 20 percent of the aggregated reconciliation target price amount calculated in paragraph (c)(3) of this section for the performance year. The post-episode spending calculation amount in ( print page 69926) paragraph (f) of this section is not subject to the limitation on loss.
(2) Limitation on gain. (i) For TEAM participants in Track 1, the reconciliation payment amount for a performance year cannot exceed 10 percent of the aggregated reconciliation target price amount calculated in accordance with paragraph (c)(3) of this section for the performance year.
(ii) For TEAM participants in Tracks 2, the reconciliation payment amount for a performance year cannot exceed 5 percent of the aggregated reconciliation target price amount calculated in accordance with paragraph (c)(3) of this section for the performance year.
(iii) For TEAM participants in Track 3, the reconciliation payment amount for a performance year cannot exceed 20 percent of the aggregated reconciliation target price amount calculated in accordance with paragraph (c)(3) of this section for the performance year.
(iv) The post-episode spending amount calculated in accordance with paragraph (f) of this section is not subject to the limitation on gain.
(3) Limitation on loss for certain providers. For performance years 2-5, the repayment amount for a TEAM participant in Track 2 defined at § 512.505, must not exceed 5 percent of the aggregated reconciliation target price amount calculated in accordance with paragraph (c)(3) of this section.
(f) Post-episode spending calculation. CMS calculates the post-episode spending amount as follows: If the average post-episode spending amount for a TEAM participant in the performance year being reconciled is greater than 3 standard deviations above the regional average post-episode spending amount for the performance year, then the post-episode spending amount that exceeds 3 standard deviations above the regional average post-episode spending amount for the performance year is subtracted from the NPRA for that performance year.
(g) Calculation of the reconciliation payment or repayment amount. (1) CMS applies the results of the post-episode spending calculation set forth in paragraph (f) of this section to the NPRA as follows:
(i) For TEAM participants whose post-episode spending amount does not exceed the limit calculated in paragraph (f) of this section, the reconciliation payment or repayment amount is equal to the NPRA.
(ii) If the TEAM participant's post-episode spending exceeds the limit calculated in paragraph (f) of this section, CMS subtracts the amount of post-episode spending exceeding the limit from the NPRA to calculate the reconciliation payment or repayment amount.
(2) If the amount calculated in paragraph (g)(1) of this section is positive, the TEAM participant is owed a reconciliation payment in that amount, to be paid by CMS in one lump sum payment.
(3) If the amount calculated in paragraph (g)(1) of this section is negative, CMS determines the repayment amount as follows:
(i) For TEAM participants in Track 1, the TEAM participant does not owe a repayment amount.
(ii) For TEAM participants in Track 2 or Track 3 for Performance Years 1-5, as applicable, the Team participant owes that amount as a repayment to CMS.
(h) TEAM reconciliation report. CMS issues each TEAM participant a TEAM reconciliation report for the performance year. Each TEAM reconciliation report contains the following:
(1) The total performance year spending for the TEAM participant.
(2) The TEAM participant's reconciliation target prices.
(3) The TEAM participant's reconciliation amount.
(4) The TEAM participant's composite quality score calculated in accordance with § 512.547(b).
(5) The TEAM participant's quality-adjusted reconciliation amount.
(6) The stop-loss and stop-gain limits that apply to the TEAM participant.
(7) The TEAM participant's NPRA.
(8) The TEAM participant's post-episode spending amount, if applicable.
(9) The amount of any reconciliation payment owed to the TEAM participant or repayment owed by the TEAM participant to CMS for the performance year, if applicable.
The TEAM does not replace any existing Medicare incentive programs or add-on payments. The TEAM payments are independent of, and do not affect, any incentive programs or add-on payments under existing Medicare payment systems.
(a) General. CMS prorates services included in the episode that extend beyond the episode so that only those portions of the services that were furnished during the episode are included in the calculation of the actual episode payments.
(b) Proration of services. CMS prorates payments for services that extend beyond the episode for the purposes of calculating both baseline episode spending and performance year spending using the following methodology:
(1) Non-IPPS inpatient services. Non-IPPS inpatient services that extend beyond the end of the episode are prorated according to the percentage of the actual length of stay (in days) that falls within the episode.
(2) Home health agency services. Home health agency services paid under the Medicare prospective payment system in accordance with part 484, subpart E of this chapter that extend beyond the episode are prorated according to the percentage of days, starting with the first billable service date and through and including the last billable service date, that occur during the episode.
(3) IPPS services. IPPS services that extend beyond the end of the episode are prorated according to the MS-DRG geometric mean length of stay, using the following methodology:
(i) The first day of the IPPS stay is counted as 2 days.
(ii) If the actual length of stay that occurred during the episode is equal to or greater than the MS-DRG geometric mean, the full MS-DRG payment is allocated to the episode.
(iii) If the actual length of stay that occurred during the episode is less than the MS-DRG geometric mean length of stay, the MS-DRG payment amount is allocated to the episode based on the number of inpatient days that fall within the episode.
(4) If the full amount of the payment is not allocated to the episode, any remainder amount is allocated to the post-episode spending calculation (defined in § 512.550(f)).
(a) Notice of calculation error (first level of appeal). Subject to the limitations on review in § 512.594, if a TEAM participant wishes to dispute calculations involving a matter related to payment, reconciliation amounts, repayment amounts, the use of quality measure results in determining the composite quality score, or the application of the composite quality score during reconciliation, the TEAM participant is required to provide written notice of the calculation error, in a form and manner and by a date specified by CMS.
(1) Unless the TEAM participant provides such written notice, CMS deems the TEAM reconciliation report to be final 30 calendar days after it is issued and proceeds with the payment or repayment processes as applicable. ( print page 69927)
(2) If CMS receives a notice of a calculation error within 30 calendar days of the issuance of the TEAM reconciliation report, CMS responds in writing within 30 calendar days to either confirm that there was an error in the calculation or verify that the calculation is correct. CMS reserves the right to extend the time for its response upon written notice to the TEAM participant.
(3) Only TEAM participants may use the calculation error process described in this part.
(b) Exception to the appeals process. If the TEAM participant contests a matter that does not involve an issue contained in, or a calculation that contributes to, a TEAM reconciliation report, a notice of calculation error is not required. In these instances, if CMS does not receive a request for reconsideration from the TEAM participant within 10 calendar days of the notice of the initial reconciliation, the initial determination is deemed final and CMS proceeds with the action indicated in the initial determination. This does not apply to the limitations on review in § 512.594.
(a) Applicability of this section. This section is applicable only where section 1869 of the Act has been waived or is not applicable for TEAM participants. This section is only applicable to TEAM participants.
(b) Right to reconsideration. The TEAM participant may request reconsideration of a determination made by CMS only if such reconsideration is not precluded by section 1115A(d)(2) of the Act or this subpart.
(1) A request for reconsideration by the TEAM participant must satisfy the following criteria:
(i) The request must be submitted to a designee of CMS (“Reconsideration Official”) who—
(A) Is authorized to receive such requests; and
(B) Did not participate in the determination that is the subject of the reconsideration request or, if applicable, the notice of calculation error process.
(ii) The request must include a copy of the initial determination issued by CMS and contain a detailed, written explanation of the basis for the dispute, including supporting documentation.
(iii) The request must be made within 30 days of the date of the initial determination for which reconsideration is being requested via email to an address as specified by CMS.
(2) Requests that do not meet the requirements of paragraph (b)(1) of this section are denied.
(3) Within 10 business days of receiving a request for reconsideration, the Reconsideration Official sends the parties a written acknowledgement of receipt of the reconsideration request. This acknowledgement sets forth the following:
(i) The review procedures.
(ii) A schedule that permits each party to submit position papers and supporting documentation in support of the party's position for consideration by the reconsideration official.
(4) The TEAM participant must satisfy the notice of calculation error requirements specified in this part before submitting a reconsideration request under paragraph (b) of this section.
(c) Standards for reconsideration. (1) The parties must continue to fulfill all responsibilities and obligations under TEAM during the course of any dispute arising under this part.
(2) The reconsideration consists of a review of documentation that is submitted timely and in accordance with the standards specified by the reconsideration official.
(3) The burden of proof is on the TEAM participant to demonstrate to the reconsideration official with clear and convincing evidence that the determination is inconsistent with the terms of this subpart.
(d) Reconsideration determination. (1) The reconsideration determination is based solely upon—
(i) Position papers and supporting documentation that are timely submitted to the reconsideration official per the schedule defined in paragraph (b)(3)(ii) and meet the standards for submission under paragraph (b)(1) of this section; and
(ii) Documents and data that were timely submitted to CMS in the required format before CMS made the determination that is the subject of the reconsideration request.
(2) The reconsideration official issues the reconsideration determination to CMS and to the TEAM participant in writing.
(3) Absent unusual circumstances, in which case the reconsideration official reserves the right to an extension upon written notice to the TEAM participant, the reconsideration determination is issued within 60 days of receipt of timely filed position papers and supporting documentation per the schedule defined in paragraph (b)(3)(ii) of this section.
(4) The reconsideration determination is final and binding 30 days after its issuance, unless the TEAM participant or CMS timely requests review of the reconsideration determination in accordance with paragraphs (e)(1) and (2) of this section.
(e) CMS Administrator review. The TEAM participant or CMS may request that the CMS Administrator review the reconsideration determination.
(1) The request must be made via email within 30 days of the date of the reconsideration determination to the address specified by CMS.
(2) The request must include a copy of the reconsideration determination and a detailed written explanation of why the TEAM participant or CMS disagrees with the reconsideration determination.
(3) The CMS Administrator promptly sends the parties a written acknowledgement of receipt of the request for review.
(4) The CMS Administrator sends the parties notice of the following:
(i) Whether the request for review is granted or denied.
(ii) If the request for review is granted, the review procedures and a schedule that permits each party to submit a brief in support of the party's position for consideration by the CMS Administrator.
(5) If the request for review is denied, the reconsideration determination is final and binding as of the date the request for review is denied.
(6) If the request for review is granted—
(i) The record for review consists solely of—
(A) Timely submitted briefs and the evidence contained in the record of the proceedings before the reconsideration official; and
(B) Evidence as set forth in the documents and data described in paragraph (d)(1)(ii) of this section;
(ii) The CMS Administrator reviews the record and issues to CMS and to the TEAM participant a written determination; and
(iii) The written determination of the CMS Administrator is final and binding as of the date the written determination is sent.
(a) General. CMS shares certain beneficiary-identifiable data as described in paragraphs (b), (c), and (e) of this section and certain regional aggregate data as described in paragraph (d) of this section with TEAM participants regarding TEAM beneficiaries and performance under the model. ( print page 69928)
(b) Beneficiary-identifiable claims data. CMS shares beneficiary-identifiable claims data with TEAM participants as follows:
(1) CMS makes available certain beneficiary-identifiable claims data described in paragraph (b)(5) of this section for TEAM participants to request for purposes of conducting health care operations work that falls within the first or second paragraph of the definition of health care operations at 45 CFR 164.501 regarding their TEAM beneficiaries.
(2) A TEAM participant that wishes to receive beneficiary-identifiable claims data for its TEAM beneficiaries must do all of the following:
(i) Submit a formal request for the data on at least an annual basis in a manner and form and by a date specified by CMS, indicating their selection of summary beneficiary-identifiable data, raw beneficiary-identifiable data, or both, and attest that—
(A) The TEAM participant is requesting claims data of TEAM beneficiaries who would be in an episode during the baseline period or performance year, as a HIPAA covered entity.
(B) The TEAM participant's request reflects the minimum data necessary, as set forth in paragraph (c) of this section, for the TEAM participant to conduct health care operations work that falls within the first or second paragraph of the definition of health care operations at 45 CFR 164.501 .
(C) The TEAM participant's use of claims data is limited to developing processes and engaging in appropriate activities related to coordinating care, improving the quality and efficiency of care, and conducting population-based activities relating to improving health or reducing health care costs that are applied uniformly to all TEAM beneficiaries, in an episode during the baseline period or performance year, and that these data are not to be used to reduce, limit or restrict care for specific Medicare beneficiaries.
(ii) Sign and submit a TEAM data sharing agreement, as defined in § 512.505, with CMS as set forth in paragraph (e) of this section.
(3) CMS shares this beneficiary-identifiable claims data with a TEAM participant in accordance with applicable privacy and security laws and established privacy and security protections.
(4) CMS omits from the beneficiary-identifiable claims data any information that is subject to the regulations in 42 CFR part 2 governing the confidentiality of substance use disorder patient records.
(5) The beneficiary-identifiable claims data includes, when available, the following:
(i) Unrefined (raw) Medicare Parts A and B beneficiary-identifiable claims data for TEAM beneficiaries in an episode during the 3-year baseline period and performance year.
(ii) Summarized (summary) Medicare Parts A and B beneficiary-identifiable claims data for TEAM beneficiaries in an episode during the 3-year baseline period and performance year.
(6) CMS makes available the beneficiary-identifiable claims data for retrieval by TEAM participants at the following frequency:
(i) Annually, at least 1 month prior to every performance year for baseline period data, based on the baseline periods described in § 512.540(b)(2).
(ii) Monthly during the performance year and for up to 6 months after the performance year for performance year data.
(c) Minimum necessary data. The TEAM participant must limit its request for beneficiary-identifiable data under paragraph (b) of this section to the minimum necessary Parts A and B data elements which may include, but are not limited to the following:
(1) Medicare beneficiary identifier (ID).
(2) Procedure code.
(3) Gender.
(4) Diagnosis code.
(5) Claim ID.
(6) The from and through dates of service.
(7) The provider or supplier ID.
(8) The claim payment type.
(9) Date of birth and death, if applicable.
(10) Tax identification number.
(11) National provider identifier.
(d) Regional aggregate data. (1) CMS shares regional aggregate data for the 3-year baseline period and performance years with TEAM participants as follows:
(i) Shares 3-year baseline period regional aggregate data annually at least 1 month before the performance year, based on the baseline periods described in § 512.540(b)(2).
(ii) Shares performance year regional aggregate data on a monthly basis during the performance year and for up to 6 months after the performance year.
(2) Regional aggregate data—
(i) Is aggregated based on all Parts A and B claims associated with episodes in TEAM for the U.S. Census Division in which the TEAM participant is located;
(ii) Summarizes average episode spending for episodes in TEAM in the U.S. Census Division in which the TEAM participant is located; and
(iii) Is de-identified in accordance with 45 CFR 164.514(b) .
(e) TEAM data sharing agreement. (1) A TEAM participant who wishes to retrieve the beneficiary-identifiable data specified in paragraph (b) of this section, must complete and submit, on at least an annual basis, a signed TEAM data sharing agreement, as defined in § 512.505, to be provided in a form and manner and by a date specified by CMS, under which the TEAM participant agrees:
(i) To comply with the requirements for use and disclosure of this beneficiary-identifiable data that are imposed on covered entities by the HIPAA regulations and the requirements of the TEAM set forth in this part.
(ii) To comply with additional privacy, security, breach notification, and data retention requirements specified by CMS.
(iii) To contractually bind each downstream recipient of the beneficiary-identifiable data that is a business associate of the TEAM participant to the same terms and conditions to which the TEAM participant is itself bound in its TEAM data sharing agreement with CMS as a condition of the business associate's receipt of the beneficiary-identifiable data retrieved by the TEAM participant under TEAM.
(iv) That if the TEAM participant misuses or discloses the beneficiary-identifiable data in a manner that violates any applicable statutory or regulatory requirements or that is otherwise non-compliant with the provisions of the TEAM data sharing agreement, CMS may deem the TEAM participant ineligible to retrieve beneficiary-identifiable data under paragraph (b) of this section for any amount of time, and the TEAM participant may be subject to additional sanctions and penalties available under the law.
(2) A TEAM participant must comply with all applicable laws and the terms of the TEAM data sharing agreement in order to retrieve the beneficiary-identifiable data.
(a) Health equity plans. (1) The TEAM participant may voluntarily submit a health equity plan to CMS for each performance year that includes the elements specified in paragraph (a)(2) of this section, in a form and manner and by the date specified by CMS.
(2) Health equity plans must include the following elements: ( print page 69929)
(i) Identifies health disparities in the TEAM participant's population of TEAM beneficiaries.
(ii) Identifies health equity goals and describes how the TEAM participant uses the health equity goals to monitor and evaluate progress in reducing the identified health disparities.
(iii) Describes the health equity plan intervention strategy.
(iv) Identifies health equity plan performance measure(s), the data sources used to construct the performance measures, and an approach to monitor and evaluate the measures.
(b) Health-related social needs screening and reporting. (1) For all performance years, the TEAM participant may voluntarily submit aggregated health-related social needs screening and screened-positive data in a form and manner and by the dates specified by CMS. The health-related social needs screening and reporting must include the elements specified in paragraph (a)(2) of this section.
(2) CMS uses the following measures from the Hospital Inpatient Quality Reporting Program for the TEAM participants who opt to voluntarily submit aggregated health-related social needs screening and screened-positive data.
(i) Screening for Social Drivers of Health (SDOH-1; CMIT ID #1664).
(ii) Screen Positive Rate for Social Drivers of Health (SDOH-2; CMIT ID #1662).
(3) For all performance years, TEAM participants that voluntarily submit data health-related social needs screening and screened-positive data as specified in paragraphs (b)(1) and (2) of this section may voluntarily submit information on referral policies and procedures for beneficiaries that screen positive for health-related social needs in a form and manner and by dates specified by CMS.
(c) Demographic data collection and reporting. For all performance years, the TEAM participant may voluntarily collect and submit to CMS, in a form and manner and by the dates specified by CMS, demographic data of TEAM beneficiaries that are willing to share demographic data elements with the TEAM participant and CMS.
(a) A TEAM participant must include in hospital discharge planning a referral to a supplier of primary care services for a TEAM beneficiary, on or prior to discharge from an anchor hospitalization or anchor procedure.
(b) In making the referral described in paragraph (a) of this section, the TEAM participant must comply with beneficiary freedom of choice, as described in § 512.582(a).
(c) A TEAM participant that does not comply with paragraph (a) of this section, may be subject to remedial action as described in § 512.592.
(a) General. (1) A TEAM participant may enter into a sharing arrangement with a TEAM collaborator to make a gainsharing payment, or to receive an alignment payment, or both. A TEAM participant must not make a gainsharing payment to a TEAM collaborator or receive an alignment payment from a TEAM collaborator except in accordance with a sharing arrangement.
(2) A sharing arrangement must comply with the provisions of this section and all other applicable laws and regulations, including the applicable fraud and abuse laws and all applicable payment and coverage requirements.
(3) TEAM participants must develop, maintain, and use a set of written policies for selecting individuals and entities to be TEAM collaborators.
(i) These policies must contain criteria related to, and inclusive of, the quality of care delivered by the potential TEAM collaborator and the provision of TEAM activities.
(ii) The selection criteria cannot be based directly or indirectly on the volume or value of past or anticipated referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, any downstream collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, collaboration agent, or downstream collaboration agent.
(iii) A selection criterion that considers whether a potential TEAM collaborator has performed a reasonable minimum number of services that would qualify as TEAM activities, as determined by the TEAM participant, will be deemed not to violate the volume or value standard if the purpose of the criterion is to ensure the quality of care furnished to TEAM beneficiaries.
(4) If a TEAM participant enters into a sharing arrangement, its compliance program must include oversight of sharing arrangements and compliance with the applicable requirements of TEAM.
(b) Requirements. (1) A sharing arrangement must be in writing and signed by the parties, and entered into before care is furnished to TEAM beneficiaries under the sharing arrangement.
(2) Participation in a sharing arrangement must be voluntary and without penalty for nonparticipation.
(3) The sharing arrangement must require the TEAM collaborator and its employees, contractors (including collaboration agents), and subcontractors (including downstream collaboration agents) to comply with all of the following:
(i) The applicable provisions of this part (including requirements regarding beneficiary notifications, access to records, record retention, and participation in any evaluation, monitoring, compliance, and enforcement activities performed by CMS or its designees).
(ii) All applicable Medicare provider enrollment requirements at § 424.500 of this chapter, including having a valid and active TIN or NPI, during the term of the sharing arrangement.
(iii) All other applicable laws and regulations.
(4) The sharing arrangement must require the TEAM collaborator to have or be covered by a compliance program that includes oversight of the sharing arrangement and compliance with the requirements of TEAM that apply to its role as a TEAM collaborator, including any distribution arrangements.
(5) The sharing arrangement must not pose a risk to beneficiary access, beneficiary freedom of choice, or quality of care.
(6) The board or other governing body of the TEAM participant must have responsibility for overseeing the TEAM participant's participation in TEAM, its arrangements with TEAM collaborators, its payment of gainsharing payments, its receipt of alignment payments, and its use of beneficiary incentives in TEAM.
(7) The specifics of the agreement must be documented in writing and must be made available to CMS upon request (as outlined in § 512.590).
(8) The sharing arrangement must specify the following:
(i) The purpose and scope of the sharing arrangement.
(ii) The obligations of the parties, including specified TEAM activities and other services to be performed by the parties under the sharing arrangement.
(iii) The date range for which the sharing arrangement is effective.
(iv) The financial or economic terms for payment, including the following:
(A) Eligibility criteria for a gainsharing payment.
(B) Eligibility criteria for an alignment payment.
(C) Frequency of gainsharing or alignment payments. ( print page 69930)
(D) Methodology and accounting formula for determining the amount of a gainsharing payment or alignment payment.
(9) The sharing arrangement must not—
(i) Induce the TEAM participant, TEAM collaborator, or any employees, contractors, or subcontractors of the TEAM participant or TEAM collaborator to reduce or limit medically necessary services to any Medicare beneficiary; or
(ii) Restrict the ability of a TEAM collaborator to make decisions in the best interests of its patients, including the selection of devices, supplies, and treatments.
(c) Gainsharing payment, alignment payment, and internal cost savings conditions and restrictions. (1) Gainsharing payments, if any, must—
(i) Be derived solely from reconciliation payment amounts, or internal cost savings, or both;
(ii) Be distributed on an annual basis (not more than once per calendar year);
(iii) Not be a loan, advance payment, or payment for referrals or other business; and
(iv) Be clearly identified as a gainsharing payment at the time it is paid.
(2)(i) To be eligible to receive a gainsharing payment, a TEAM collaborator must meet quality of care criteria for the performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment that comprises the gainsharing payment. The quality-of-care criteria must be established by the TEAM participant and directly relate to the episode.
(ii) To be eligible to receive a gainsharing payment, or to be required to make an alignment payment, a TEAM collaborator other than ACO, PGP, NPPGP, or TGP must have directly furnished a billable item or service to a TEAM beneficiary during an episode that was attributed to the same performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment amount or repayment amount that comprises the gainsharing payment or the alignment payment.
(iii) To be eligible to receive a gainsharing payment, or to be required to make an alignment payment, a TEAM collaborator that is a PGP, NPPGP, or TGP must meet the following criteria:
(A) The PGP, NPPGP, or TGP must have billed for an item or service that was rendered by one or more PGP member, NPPGP member, or TGP member respectively to a TEAM beneficiary during an episode that was attributed to the same performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment amount or repayment amount that comprises the gainsharing payment or the alignment payment.
(B) The PGP, NPPGP, or TGP must have contributed to TEAM activities and been clinically involved in the care of TEAM beneficiaries during the same performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment amount or repayment amount that comprises the gainsharing payment or the alignment payment. A non-exhaustive list of examples where, a PGP, NPPGP, or TGP might have been clinically involved in the care of TEAM beneficiaries includes—
( 1 ) Providing care coordination services to TEAM beneficiaries during or after inpatient admission;
( 2 ) Engaging with a TEAM participant in care redesign strategies, and performing a role in implementing such strategies, that are designed to improve the quality of care for episodes and reduce episode spending; or
( 3 ) In coordination with other providers and suppliers (such as PGP members, NPPGP members, or TGP members; the TEAM participant; and post-acute care providers), implementing strategies designed to address and manage the comorbidities of TEAM beneficiaries.
(iv) To be eligible to receive a gainsharing payment, or to be required to make an alignment payment, a TEAM collaborator that is an ACO must meet the following criteria:
(A) The ACO must have had an ACO provider/supplier that directly furnished, or an ACO participant that billed for, an item or service that was rendered to a TEAM beneficiary during an episode that was attributed to the same performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment amount or repayment amount that comprises the gainsharing payment or the alignment payment; and
(B) The ACO must have contributed to TEAM activities and been clinically involved in the care of TEAM beneficiaries during the performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment amount or repayment amount that comprises the gainsharing payment or the alignment payment. A non-exhaustive list of ways in which an ACO might have been clinically involved in the care of TEAM beneficiaries could include—
( 1 ) Providing care coordination services to TEAM beneficiaries during and/or after inpatient admission;
( 2 ) Engaging with a TEAM participant in care redesign strategies and performing a role in implementing such strategies that are designed to improve the quality of care and reduce spending for episodes; or
( 3 ) In coordination with providers and suppliers (such as ACO participants, ACO providers/suppliers, the TEAM participant, and post-acute care providers), implementing strategies designed to address and manage the comorbidities of TEAM beneficiaries.
(3) The methodology for accruing, calculating and verifying internal cost savings will be determined by the TEAM participant. The methodology—
(i) Must be transparent, measurable, and verifiable in accordance with generally accepted accounting principles (GAAP) and Government Auditing Standards (The Yellow Book).
(ii) Used to calculate internal cost savings must reflect the actual, internal cost savings achieved by the TEAM participant through the documented implementation of TEAM activities identified by the TEAM participant and must exclude—
(A) Any savings realized by any individual or entity that is not the TEAM participant; and
(B) “Paper” savings from accounting conventions or past investment in fixed costs.
(4) The amount of any gainsharing payments must be determined in accordance with a methodology that is based solely on quality of care and the provision of TEAM activities. The methodology may take into account the amount of TEAM activities provided by a TEAM collaborator relative to other TEAM collaborators.
(5) For a performance year, the aggregate amount of all gainsharing payments that are derived from reconciliation payment amounts must not exceed the amount of that year's reconciliation payment amount.
(6) No entity or individual, whether a party to a sharing arrangement or not, may condition the opportunity to make or receive gainsharing payments or to make or receive alignment payments directly or indirectly on the volume or value of past or anticipated referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, any downstream collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, collaboration agent, or downstream collaboration agent. ( print page 69931)
(7) A TEAM participant must not make a gainsharing payment to a TEAM collaborator if CMS has notified the TEAM participant that such TEAM collaborator is subject to any action by CMS, HHS or any other governmental entity, or its designees, for noncompliance with this part or the fraud and abuse laws, for the provision of substandard care to TEAM beneficiaries or other integrity problems, or for any other program integrity problems or noncompliance with any other laws or regulations.
(8) The sharing arrangement must require the TEAM participant to recoup any gainsharing payment that contained funds derived from a CMS overpayment on a reconciliation payment amount or was based on the submission of false or fraudulent data.
(9) Alignment payments from a TEAM collaborator to a TEAM participant may be made at any interval that is agreed upon by both parties, and must not be—
(i) Issued, distributed, or paid prior to the calculation by CMS of a repayment amount; payment;
(ii) Loans, advance payments, or payments for referrals or other business; or
(iii) Assessed by a TEAM participant in the absence of a repayment amount.
(10) The TEAM participant must not receive any amounts under a sharing arrangement from a TEAM collaborator that are not alignment payments.
(11) For a performance year, the aggregate amount of all alignment payments received by the TEAM participant must not exceed 50 percent of the TEAM participant's repayment amount.
(12) The aggregate amount of all alignment payments from a TEAM collaborator to the TEAM participant may not be greater than—
(i) With respect to a TEAM collaborator other than an ACO, 25 percent of the TEAM participant's repayment amount.
(ii) With respect to a TEAM collaborator that is an ACO, 50 percent of the TEAM participant's repayment amount.
(13) The amount of any alignment payments must be determined in accordance with a methodology that does not directly account for the volume or value of past or anticipated referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, any downstream collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, collaboration agent, or downstream collaboration agent.
(14) All gainsharing payments and any alignment payments must be administered by the TEAM participant in accordance with generally accepted accounting principles (GAAP) and Government Auditing Standards (The Yellow Book).
(15) All gainsharing payments and alignment payments must be made by check, electronic funds transfer, or another traceable cash transaction.
(d) Documentation requirements. (1) TEAM participants must—
(i) Document the sharing arrangement contemporaneously with the establishment of the arrangement;
(ii) Publicly post (and update on at least a quarterly basis) on a web page on the TEAM participant's website—
(A) Accurate lists of all current TEAM collaborators, including the TEAM collaborators' names and addresses as well as accurate historical lists of all TEAM collaborators.
(B) Written policies for selecting individuals and entities to be TEAM collaborators as required by § 512.565(a)(3).
(iii) Maintain, and require each TEAM collaborator to maintain, contemporaneous documentation with respect to the payment or receipt of any gainsharing payment or alignment payment that includes, at a minimum—
(A) Nature of the payment (gainsharing payment or alignment payment);
(B) Identity of the parties making and receiving the payment;
(C) Date of the payment;
(D) Amount of the payment; and
(E) Date and amount of any recoupment of all or a portion of a TEAM collaborator's gainsharing payment.
(F) Explanation for each recoupment, such as whether the TEAM collaborator received a gainsharing payment that contained funds derived from a CMS overpayment of a reconciliation payment or was based on the submission of false or fraudulent data.
(2) The TEAM participant must keep records of all of the following:
(i) Its process for determining and verifying its potential and current TEAM collaborators' eligibility to participate in Medicare.
(ii) Its plan to track internal cost savings.
(iii) Information on the accounting systems used to track internal cost savings.
(iv) A description of current health information technology, including systems to track reconciliation payment amounts, repayment amounts, and internal cost savings.
(v) Its plan to track gainsharing payments and alignment payments.
(3) The TEAM participant must retain and provide access to and must require each TEAM collaborator to retain and provide access to, the required documentation in accordance with § 512.586.
(a) General. (1) An ACO, PGP, NPPGP, or TGP that is a TEAM collaborator and has entered into a sharing arrangement with a TEAM participant may distribute all or a portion of any gainsharing payment it receives from the TEAM participant only in accordance with a distribution arrangement.
(2) All distribution arrangements must comply with the provisions of this section and all other applicable laws and regulations, including the fraud and abuse laws.
(b) Requirements. (1) All distribution arrangements must be in writing and signed by the parties, contain the effective date of the agreement, and be entered into before care is furnished to TEAM beneficiaries under the distribution arrangement.
(2) Participation in a distribution arrangement must be voluntary and without penalty for nonparticipation.
(3) The distribution arrangement must require the collaboration agent to comply with all applicable laws and regulations.
(4) The opportunity to make or receive a distribution payment must not be conditioned directly or indirectly on the volume or value of past or anticipated referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, any downstream collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, collaboration agent, or downstream collaboration agent.
(5) The amount of any distribution payments from an ACO, from an NPPGP to an NPPGP member, or from a TGP to a TGP member, must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities and that may take into account the amount of such TEAM activities provided by a collaboration agent relative to other collaboration agents.
(6) The amount of any distribution payments from a PGP must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities and that may take into account the amount of such TEAM activities provided by a collaboration agent relative to other collaboration agents. ( print page 69932)
(7) A collaboration agent is eligible to receive a distribution payment only if the collaboration agent furnished or billed for an item or service rendered to a TEAM beneficiary during an episode that was attributed to the same performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment amount that comprises the gainsharing payment being distributed.
(8) With respect to the distribution of any gainsharing payment received by an ACO, PGP, NPPGP, or TGP, the total amount of all distribution payments for a performance year must not exceed the amount of the gainsharing payment received by the TEAM collaborator from the TEAM participant for the same performance year.
(9) All distribution payments must be made by check, electronic funds transfer, or another traceable cash transaction.
(10) The collaboration agent must retain the ability to make decisions in the best interests of the patient, including the selection of devices, supplies, and treatments.
(11) The distribution arrangement must not—
(i) Induce the collaboration agent to reduce or limit medically necessary items and services to any Medicare beneficiary; or
(ii) Reward the provision of items and services that are medically unnecessary.
(12) The TEAM collaborator must maintain contemporaneous documentation regarding distribution arrangements in accordance with § 512.586, including all of the following:
(i) The relevant written agreements.
(ii) The date and amount of any distribution payment(s).
(iii) The identity of each collaboration agent that received a distribution payment.
(iv) A description of the methodology and accounting formula for determining the amount of any distribution payment.
(13) The TEAM collaborator may not enter into a distribution arrangement with any individual or entity that has a sharing arrangement with the same TEAM participant.
(14) The TEAM collaborator must retain and provide access to and must require collaboration agents to retain and provide access to, the required documentation in accordance with § 512.586.
(a) General. (1) An ACO participant that is a PGP, NPPGP, or TGP and that has entered into a distribution arrangement with a TEAM collaborator that is an ACO, may distribute all or a portion of any distribution payment it receives from the TEAM collaborator only in accordance with a downstream distribution arrangement.
(2) All downstream distribution arrangements must comply with the provisions of this section and all applicable laws and regulations, including the fraud and abuse laws.
(b) Requirements. (1) All downstream distribution arrangements must be in writing and signed by the parties, contain the effective date of the agreement, and be entered into before care is furnished to TEAM beneficiaries under the downstream distribution arrangement.
(2) Participation in a downstream distribution arrangement must be voluntary and without penalty for nonparticipation.
(3) The downstream distribution arrangement must require the downstream collaboration agent to comply with all applicable laws and regulations.
(4) The opportunity to make or receive a downstream distribution payment must not be conditioned directly or indirectly on the volume or value of past or anticipated referrals or business otherwise generated by, between or among the TEAM participant, any TEAM collaborator, any collaboration agent, any downstream collaboration agent, or any individual or entity affiliated with a TEAM participant, TEAM collaborator, collaboration agent, or downstream collaboration agent.
(5) The amount of any downstream distribution payments from an NPPGP to an NPPGP member or from a TGP to a TGP member must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities and that may take into account the amount of such TEAM activities provided by a downstream collaboration agent relative to other downstream collaboration agents.
(6) The amount of any downstream distribution payments from a PGP must be determined in accordance with a methodology that is solely based on quality of care and the provision of TEAM activities and that may take into account the amount of such TEAM activities provided by a downstream collaboration agent relative to other downstream collaboration agents.
(7) A downstream collaboration agent is eligible to receive a downstream distribution payment only if the downstream collaboration agent furnished an item or service to a TEAM beneficiary during an episode that is attributed to the same performance year for which the TEAM participant accrued the internal cost savings or earned the reconciliation payment amount that comprises the gainsharing payment from which the ACO made the distribution payment to the PGP, NPPGP, or TGP that is an ACO participant.
(8) The total amount of all downstream distribution payments made to downstream collaboration agents must not exceed the amount of the distribution payment received by the PGP, NPPGP, or TGP from the ACO.
(9) All downstream distribution payments must be made by check, electronic funds transfer, or another traceable cash transaction.
(10) The downstream collaboration agent must retain his or her ability to make decisions in the best interests of the beneficiary, including the selection of devices, supplies, and treatments.
(11) The downstream distribution arrangement must not—
(i) Induce the downstream collaboration agent to reduce or limit medically necessary services to any Medicare beneficiary; or
(12) The PGP, NPPGP, or TGP must maintain contemporaneous documentation regarding downstream distribution arrangements in accordance with § 512.586, including the following:
(ii) The date and amount of any downstream distribution payment.
(iii) The identity of each downstream collaboration agent that received a downstream distribution payment.
(iv) A description of the methodology and accounting formula for determining the amount of any downstream distribution payment.
(13) The PGP, NPPGP, or TGP may not enter into a downstream distribution arrangement with any PGP member, NPPGP member, or TGP member who has—
(i) A sharing arrangement with a TEAM participant.
(ii) A distribution arrangement with the ACO that the PGP, NPPGP, or TGP is a participant in.
(14) The PGP, NPPGP, or TGP must retain and provide access to, and must require downstream collaboration agents to retain and provide access to, the required documentation in accordance with § 512.586.
(a) General. TEAM participants may choose to provide in-kind patient engagement incentives including but not limited to items of technology to ( print page 69933) TEAM beneficiaries in an episode, subject to the following conditions:
(1) The incentive must be provided directly by the TEAM participant or by an agent of the TEAM participant under the TEAM participant's direction and control to the TEAM beneficiary during an episode.
(2) The item or service provided must be reasonably connected to medical care provided to a TEAM beneficiary during an episode.
(3) The item or service must be a preventive care item or service or an item or service that advances a clinical goal, as listed in paragraph (c) of this section, for a TEAM beneficiary in an episode by engaging the TEAM beneficiary in better managing his or her own health.
(4) The item or service must not be tied to the receipt of items or services outside the episode.
(5) The item or service must not be tied to the receipt of items or services from a particular provider or supplier.
(6) The availability of the items or services must not be advertised or promoted, except that a TEAM beneficiary may be made aware of the availability of the items or services at the time the TEAM beneficiary could reasonably benefit from them.
(7) The cost of the items or services must not be shifted to any Federal health care program, as defined at section 1128B(f) of the Act.
(b) Technology provided to a TEAM beneficiary. TEAM beneficiary engagement incentives involving technology are subject to the following additional conditions:
(1) Items or services involving technology provided to a TEAM beneficiary may not exceed $1,000 in retail value for any one TEAM beneficiary during any one episode.
(2) Items or services involving technology provided to a TEAM beneficiary must be the minimum necessary to advance a clinical goal, as listed in paragraph (c) of this section, for a beneficiary in an episode.
(3) Items of technology exceeding $75 in retail value must—
(i) Remain the property of the TEAM participant; and
(ii) Be retrieved from the TEAM beneficiary at the end of the episode, with documentation of the ultimate date of retrieval. The TEAM participant must document all retrieval attempts. In cases when the item of technology is not able to be retrieved, the TEAM participant must determine why the item was not retrievable. If it was determined that the item was misappropriated (if it were sold, for example), the TEAM participant must take steps to prevent future beneficiary incentives for that TEAM beneficiary. Following this process, documented, diligent, good faith attempts to retrieve items of technology will be deemed to meet the retrieval requirement.
(c) Clinical goals of TEAM. The following are the clinical goals of TEAM, which may be advanced through TEAM beneficiary incentives:
(1) Beneficiary adherence to drug regimens.
(2) Beneficiary adherence to a care plan.
(3) Reduction of readmissions and complications following an episode.
(4) Management of chronic diseases and conditions that may be affected by the TEAM procedure.
(d) Documentation of TEAM beneficiary incentives. (1) TEAM participants must maintain documentation of items and services furnished as beneficiary incentives that exceed $25 in retail value.
(2) The documentation must be established contemporaneously with the provision of the items and services with a record established and maintained to include at least the following:
(i) The date the incentive is provided.
(ii) The identity of the TEAM beneficiary to whom the item or service was provided.
(3) The documentation regarding items of technology exceeding $75 in retail value must also include contemporaneous documentation of any attempt to retrieve technology at the end of an episode, or why the items were not retrievable, as described in paragraph (b)(3) of this section.
(4) The TEAM participant must retain and provide access to the required documentation in accordance with § 512.586.
(a) Application of the CMS-sponsored model arrangements safe harbor. CMS has determined that the Federal Anti-Kickback Statute Safe Harbor for CMS-sponsored model arrangements ( 42 CFR 1001.952(ii)(1) ) is available to protect remuneration furnished in TEAM in the form of the sharing arrangement's gainsharing payments and alignment payments, the distribution arrangement's distribution payments, and the downstream distribution arrangement's distribution payments that meet all safe harbor requirements set forth in 42 CFR 1001.952(ii) , and §§ 512.565, 512.568, 512.570.
(b) Application of the CMS-sponsored model patient incentives safe harbor. CMS has determined that the Federal Anti-Kickback Statute Safe Harbor for CMS-sponsored model patient incentives ( 42 CFR 1001.952(ii)(2) ) is available to protect TEAM beneficiary incentives that meet all safe harbor requirements set forth in 42 CFR 1001.952(ii) and § 512.575.
(a) Waiver of certain telehealth requirements— (1) Waiver of the geographic site requirements. Except for the geographic site requirements for a face-to-face encounter for home health certification, CMS waives the geographic site requirements of section 1834(m)(4)(C)(i)(I) through (III) of the Act for episodes being tested in TEAM solely for services that—
(i) May be furnished via telehealth under existing Medicare program requirements; and
(ii) Are included in the episode in accordance with § 512.525(e).
(2) Waiver of the originating site requirements. Except for the originating site requirements for a face-to-face encounter for home health certification, CMS waives the originating site requirements under section 1834(m)(4)I(ii)(I) through (VIII) of the Act for episodes to permit a telehealth visit to originate in the beneficiary's home or place of residence solely for services that—
(3) Waiver of selected payment provisions. (i) CMS waives the payment requirements under section 1834(m)(2)(A) of the Act so that the facility fee normally paid by Medicare to an originating site for a telehealth service is not paid if the service is originated in the beneficiary's home or place of residence.
(ii) CMS waives the payment requirements under section 1834(m)(2)(B) of the Act to allow the distant site payment for telehealth home visit HCPCS codes unique to TEAM.
(4) Other requirements. All other requirements for Medicare coverage and payment of telehealth services continue to apply, including the list of specific services approved to be furnished by telehealth.
(b) Waiver of the SNF 3-day rule —(1) Episodes initiated by an anchor hospitalization. CMS waives the SNF 3-day rule for coverage of a SNF stay within 30 days of the date of discharge from the anchor hospitalization for a ( print page 69934) beneficiary who is a TEAM beneficiary on the date of discharge from the anchor hospitalization if the SNF is identified on the applicable calendar quarter list of qualified SNFs at the time of the TEAM beneficiary's admission to the SNF.
(2) Episodes initiated by an anchor procedure. CMS waives the SNF 3-day rule for coverage of a SNF stay within 30 days of the date of service of the anchor procedure for a beneficiary who is a TEAM beneficiary on the date of service of the anchor procedure if the SNF is identified on the applicable calendar quarter list of qualified SNFs at the time of the TEAM beneficiary's admission to the SNF.
(3) Determination of qualified SNFs. CMS determines the qualified SNFs for each calendar quarter based on a review of the most recent rolling 12 months of overall star ratings on the Five-Star Quality Rating System for SNFs on the Nursing Home Compare website. Qualified SNFs are rated an overall of 3 stars or better for at least 7 of the 12 months.
(4) Posting of qualified SNFs. CMS posts to the CMS website the list of qualified SNFs in advance of the calendar quarter.
(5) Financial liability for non-covered SNF services. If CMS determines that the waiver requirements specified in paragraph (b) of this section were not met, the following apply:
(i) CMS makes no payment to a SNF for SNF services if the SNF admits a TEAM beneficiary who has not had a qualifying anchor hospitalization or anchor procedure.
(ii) In the event that CMS makes no payment for SNF services furnished by a SNF as a result of paragraph (b)(5)(i) of this section, the beneficiary protections specified in paragraph (b)(5)(iii) of this section apply, unless the TEAM participant has provided the beneficiary with a discharge planning notice in accordance with § 512.582(b)(3).
(iii) If the TEAM participant does not provide the beneficiary with a discharge planning notice in accordance with § 512.582(b)(3)—
(A) The SNF must not charge the beneficiary for the expenses incurred for such services;
(B) The SNF must return to the beneficiary any monies collected for such services; and
(C) The TEAM participant is financially liable for the expenses incurred for such services.
(6) Coverage of SNF services and discharge planning notification. If the TEAM participant provided a discharge planning notice to the beneficiary in accordance with § 512.582(b)(3), then normal SNF coverage requirements apply, and the beneficiary may be financially liable for non-covered SNF services.
(c) Other requirements. All other Medicare rules for coverage and payment of Part A-covered services continue to apply except as otherwise waived in this part.
(a) Beneficiary freedom of choice. (1) A TEAM participant, TEAM collaborators, collaboration agents, downstream collaboration agent and downstream participants must not restrict Medicare beneficiaries' ability to choose to receive care from any provider or supplier.
(2) The TEAM participant and its downstream participants must not commit any act or omission, nor adopt any policy that inhibits beneficiaries from exercising their freedom to choose to receive care from any provider or supplier or from any health care provider who has opted out of Medicare. The TEAM participant and its downstream participants may communicate to TEAM beneficiaries the benefits of receiving care with the TEAM participant, if otherwise consistent with the requirements of this part and applicable law.
(3) As part of discharge planning and referral, TEAM participants must provide a complete list of HHAs, SNFs, IRFs, or LTCHs that are participating in the Medicare program, and that serve the geographic area (as defined by the HHA) in which the patient resides, or in the case of a SNF, IRF, or LTCH, in the geographic area requested by the patient.
(i) This list must be presented to TEAM beneficiaries for whom home health care, SNF, IRF, or LTCH services are medically necessary.
(ii) TEAM participants must specify on the list those post-acute care providers on the list with whom they have a sharing arrangement.
(iii) TEAM participants may recommend preferred providers and suppliers, consistent with applicable statutes and regulations.
(iv) TEAM participants may not limit beneficiary choice to any list of providers or suppliers in any manner other than as permitted under applicable statutes and regulations.
(v) TEAM participants must take into account patient and family preferences for choice of provider and supplier when they are expressed.
(4) TEAM participants may not charge any TEAM collaborator a fee to be included on any list of preferred providers or suppliers, nor may the TEAM participant accept such payments.
(b) Required beneficiary notification —(1) TEAM participant beneficiary notification —(i) Notification to beneficiaries. Each TEAM participant must provide written notification to any TEAM beneficiary that meets the criteria in § 512.535 of his or her inclusion in the TEAM model.
(ii) Timing of notification. Prior to discharge from the anchor hospitalization, or prior to discharge from the anchor procedure, as applicable, the TEAM participant must provide the TEAM beneficiary with a beneficiary notification as described in paragraph (b)(1)(iv) of this section.
(iii) List of beneficiaries who have received a notification. The TEAM participant must be able to generate a list of all beneficiaries who have received such notification, including the date on which the notification was provided to the beneficiary, to CMS or its designee upon request.
(iv) Content of notification. The beneficiary notification must contain all of the following:
(A) A detailed explanation of TEAM and how it might be expected to affect the beneficiary's care.
(B) Notification that the beneficiary retains freedom of choice to choose providers and services.
(C) Explanation of how patients can access care records and claims data through an available patient portal, if applicable, and how they can share access to their Blue Button® electronic health information with caregivers.
(D) Explanation of the type of beneficiary-identifiable claims data the TEAM participant may receive.
(E) A statement that all existing Medicare beneficiary protections continue to be available to the TEAM beneficiary. These include the ability to report concerns of substandard care to Quality Improvement Organizations or the 1-800-MEDICARE helpline.
(F) A list of the providers, suppliers, and ACOs with whom the TEAM participant has a sharing arrangement. This requirement may be fulfilled by the TEAM participant including in the detailed notification a Web address where beneficiaries may access the list.
(2) TEAM collaborator notice. A TEAM participant must require every TEAM collaborator to provide written notice to applicable TEAM beneficiaries of TEAM, including information on the quality and payment incentives under TEAM, and the existence of its sharing arrangement with the TEAM participant. ( print page 69935)
(i) With the exception of ACOs, PGPs, NPPGPs, and TGPs, a TEAM participant must require every TEAM collaborator that furnishes an item or service to a TEAM beneficiary during an episode to provide written notice to the beneficiary of TEAM, including basic information on the quality and payment incentives under TEAM, and the existence of the TEAM collaborator's sharing arrangement.
(A) The notice must be provided no later than the time at which the beneficiary first receives an item or service from the TEAM collaborator during an episode. In circumstances where, due to the patient's condition, it is not feasible to provide notification at such time, the notification must be provided to the beneficiary or his or her representative as soon as is reasonably practicable.
(B) The TEAM collaborator must be able to provide a list of all beneficiaries who received such a notice, including the date on which the notice was provided to the beneficiary, to CMS upon request.
(ii) A TEAM participant must require every PGP, NPPGP, or TGP that is a TEAM collaborator where a member of the PGP, member of the NPPGP, or member of the TGP furnishes an item or service to a TEAM beneficiary during an episode to provide written notice to the beneficiary of TEAM, including basic information on the quality and payment incentives under TEAM, and the existence of the entity's sharing arrangement.
(A)( 1 ) The notice must be provided no later than the time at which the beneficiary first receives an item or service from any member of the PGP, member of the NPPGP, or member of the TGP, and the required PGP, NPPGP, or TGP notice may be provided by that member respectively.
( 2 ) In circumstances where, due to the patient's condition, it is not feasible to provide notice at such times, the notice must be provided to the beneficiary or his or her representative as soon as is reasonably practicable.
(B) The PGP, NPPGP, or TGP must be able to provide a list of all beneficiaries who received such a notice, including the date on which the notice was provided to the beneficiary, to CMS upon request.
(iii) A TEAM participant must require every ACO that is a TEAM collaborator where an ACO participant or ACO provider/supplier furnishes an item or service to a TEAM beneficiary during an episode to provide written notice to the beneficiary of TEAM, including basic information on the quality and payment incentives under TEAM, and the existence of the entity's sharing arrangement.
(A)( 1 ) The notice must be provided no later than the time at which the beneficiary first receives an item or service from any ACO participant or ACO provider/supplier and the required ACO notice may be provided by that ACO participant or ACO provider/supplier respectively.
(B) The ACO must be able to provide a list of all beneficiaries who received such a notice, including the date on which the notice was provided to the beneficiary, to CMS upon request.
(3) Discharge planning notice. A TEAM participant must provide the beneficiary with a written notice of any potential financial liability associated with non-covered services recommended or presented as an option as part of discharge planning, no later than the time that the beneficiary discusses a particular post-acute care option or at the time the beneficiary is discharged from an anchor procedure or anchor hospitalization, whichever occurs earlier.
(i) If the TEAM participant knows or should have known that the beneficiary is considering or has decided to receive a non-covered post-acute care service or other non-covered associated service or supply, the TEAM participant must notify the beneficiary in writing that the service would not be covered by Medicare.
(ii) If the TEAM participant is discharging a beneficiary to a SNF after an inpatient hospital stay, and the beneficiary is being transferred to or is considering a SNF that would not qualify under the SNF 3-day waiver in § 512.580, the TEAM participant must notify the beneficiary in accordance with paragraph (b)(3)(i) of this section that the beneficiary will be responsible for payment for the services furnished by the SNF during that stay, except those services that would be covered by Medicare Part B during a non-covered inpatient SNF stay.
(4) Access to records and retention. Lists of beneficiaries that receive notifications or notices must be retained, and access provided to CMS, or its designees, in accordance with § 512.586.
(c) Availability of services. (1) The TEAM participant and its downstream participants must continue to make medically necessary covered services available to beneficiaries to the extent required by applicable law. TEAM beneficiaries and their assignees retain their rights to appeal claims in accordance with part 405, subpart I of this chapter.
(2) The TEAM participant and its downstream participants must not take any action to select or avoid treating certain Medicare beneficiaries based on their income levels or based on factors that would render the beneficiary an “at-risk beneficiary” as defined at § 425.20 of this chapter.
(3) The TEAM participant and its downstream participants must not take any action to selectively target or engage beneficiaries who are relatively healthy or otherwise expected to improve the TEAM participant's or downstream participant's financial or quality performance.
(d) Descriptive TEAM materials and activities. (1) The TEAM participant and its downstream participants must not use or distribute descriptive TEAM materials and activities that are materially inaccurate or misleading.
(2) The TEAM participant and its downstream participants must include the following statement on all descriptive TEAM materials and activities: “The statements contained in this document are solely those of the authors and do not necessarily reflect the views or policies of the Centers for Medicare Medicaid Services (CMS). The authors assume responsibility for the accuracy and completeness of the information contained in this document.”
(3) The TEAM participant and its downstream participants must retain copies of all written and electronic descriptive TEAM materials and activities and appropriate records for all other descriptive TEAM materials and activities in a manner consistent with § 512.135(c).
(4) CMS reserves the right to review, or have a designee review, descriptive TEAM materials and activities to determine whether or not the content is materially inaccurate or misleading. This review takes place at a time and in a manner specified by CMS once the descriptive TEAM materials and activities are in use by the TEAM participant.
The TEAM participant and its TEAM collaborators must comply with the requirements of § 403.1110(b) of this chapter and must otherwise cooperate with CMS' TEAM evaluation and monitoring activities as may be necessary to enable CMS to evaluate TEAM in accordance with section ( print page 69936) 1115A(b)(4) of the Act and to conduct monitoring activities under § 512.590, including producing such data as may be required by CMS to evaluate or monitor TEAM, which may include protected health information as defined in 45 CFR 160.103 and other individually-identifiable data.
(a) Right to audit. The Federal government, including CMS, HHS, and the Comptroller General, or their designees, has the right to audit, inspect, investigate, and evaluate any documents and other evidence regarding implementation of TEAM.
(b) Access to records. The TEAM participant and its TEAM collaborators must maintain and give the Federal government, including CMS, HHS, and the Comptroller General, or their designees, access to all such documents and other evidence sufficient to enable the audit, evaluation, inspection, or investigation of the implementation of TEAM, including without limitation, documents and other evidence regarding all of the following:
(1) The TEAM participant's and its downstream participants' compliance with the terms of TEAM.
(2) The accuracy of TEAM reconciliation payment amounts and repayment amounts.
(3) The TEAM participant's payment of amounts owed to CMS under TEAM.
(4) Quality measure information and the quality of services performed under the terms of TEAM.
(5) Utilization of items and services furnished under TEAM.
(6) The ability of the TEAM participant to bear the risk of potential losses and to repay any losses to CMS, as applicable.
(7) Patient safety.
(8) Other program integrity issues.
(c) Record retention. (1) The TEAM participant and its downstream participants must maintain the documents and other evidence described in paragraph (b) of this section and other evidence for a period of 6 years from the last payment determination for the TEAM participant under TEAM or from the date of completion of any audit, evaluation, inspection, or investigation, whichever is later, unless—
(i) CMS determines there is a special need to retain a particular record or group of records for a longer period and notifies the TEAM participant at least 30 days before the normal disposition date; or
(ii) There has been a termination, dispute, or allegation of fraud or similar fault against the TEAM participant or its downstream participants, in which case the records must be maintained for an additional 6 years from the date of any resulting final resolution of the termination, dispute, or allegation of fraud or similar fault.
(2) If CMS notifies the TEAM participant of the special need to retain records in accordance with paragraph (c)(1)(i) of this section or there has been a termination, dispute, or allegation of fraud or similar fault against the TEAM participant or its downstream participants described in paragraph (c)(1)(ii) of this section, the TEAM participant must notify its downstream participants of this need to retain records for the additional period specified by CMS.
(a) CMS may—
(1) Use any data obtained under §§ 512.584, 512.586, or 512.590 to evaluate and monitor TEAM; and
(2) Disseminate quantitative and qualitative results and successful care management techniques, including factors associated with performance, to other providers and suppliers and to the public. Data disseminated may include patient—
(i) De-identified results of patient experience of care and quality of life surveys, and patient; and
(ii) De-identified measure results calculated based upon claims, medical records, and other data sources.
(b) Notwithstanding any other provision of this part, for all data that CMS confirms to be proprietary trade secret information and technology of the TEAM participant or its downstream participants, CMS or its designee(s) will not release this data without the express written consent of the TEAM participant or its downstream participant, unless such release is required by law.
(c) If the TEAM participant or its downstream participant wishes to protect any proprietary or confidential information that it submits to CMS or its designee, the TEAM participant or its downstream participant must label or otherwise identify the information as proprietary or confidential. Such assertions are subject to review and confirmation by CMS prior to CMS' acting upon such assertions.
(a) Compliance with laws. The TEAM participant and each of its downstream participants must comply with all applicable laws and regulations.
(b) CMS monitoring and compliance activities. (1) CMS staff, or its approved designee, may conduct monitoring activities to ensure compliance by the TEAM participant and each of its downstream participants with the terms of TEAM under this subpart to—
(i) Understand TEAM participants' use of TEAM payments; and
(ii) Promote the safety of beneficiaries and the integrity of TEAM.
(2) Monitoring activities may include, without limitation, all of the following:
(i) Documentation requests sent to the TEAM participant and its downstream participants, including surveys and questionnaires.
(ii) Audits of claims data, quality measures, medical records, and other data from the TEAM participant and its downstream participants.
(iii) Interviews with members of the staff and leadership of the TEAM participant and its downstream participants.
(iv) Interviews with beneficiaries and their caregivers.
(v) Site visits to the TEAM participant and its downstream participants, performed in a manner consistent with paragraph (c) of this section.
(vi) Monitoring quality outcomes and clinical data, if applicable.
(vii) Tracking patient complaints and appeals.
(3) In conducting monitoring and oversight activities, CMS or its designees may use any relevant data or information including without limitation all Medicare claims submitted for items or services furnished to TEAM beneficiaries.
(c) Site visits. (1) In a manner consistent with § 512.584, the TEAM participant and its downstream participants must cooperate in periodic site visits performed by CMS or its designees in order to facilitate the evaluation of TEAM and the monitoring of the TEAM participant's compliance with the terms of TEAM.
(2) CMS or its designee provides, to the extent practicable, the TEAM participant or downstream participant with no less than 15 days advance notice of any site visit. CMS—
(i) Attempts, to the extent practicable, to accommodate a request for particular dates in scheduling site visits; and
(ii) Does not accept a date request from a TEAM participant or downstream participant that is more than 60 days after the date of the CMS initial site visit notice.
(3) The TEAM participant and its downstream participants must ensure that personnel with the appropriate responsibilities and knowledge associated with the purpose of the site visit are available during all site visits.
(4) CMS may perform unannounced site visits at the office of the TEAM ( print page 69937) participant and any of its downstream participants at any time to investigate concerns about the health or safety of beneficiaries or other patients or other program integrity issues.
(5) Nothing in this part shall be construed to limit or otherwise prevent CMS from performing site visits permitted or required by applicable law.
(d) Reopening of payment determinations. (1) CMS may reopen a TEAM payment determination on its own motion or at the request of a TEAM participant, within 4 years from the date of the determination, for good cause (as defined at § 405.986 of this chapter).
(2) CMS may reopen a TEAM payment determination at any time if there exists reliable evidence (as defined in § 405.902 of this chapter) that the determination was procured by fraud or similar fault (as defined in § 405.902 of this chapter).
(3) CMS's decision regarding whether to reopen a TEAM payment determination is binding and not subject to appeal.
(e) OIG authority. Nothing contained in the terms of TEAM limits or restricts the authority of the HHS Office of Inspector General or any other Federal government authority, including its authority to audit, evaluate, investigate, or inspect the TEAM participant or its downstream participants for violations of any Federal statutes, rules, or regulations.
(a) Grounds for remedial action. CMS may take one or more remedial actions described in paragraph (b) of this section if CMS determines that the TEAM participant or a downstream participant:
(1) Has failed to comply with any of the terms of TEAM, included in this subpart.
(2) Has failed to comply with any applicable Medicare program requirement, rule, or regulation.
(3) Has taken any action that threatens the health or safety of a beneficiary or other patient.
(4) Has submitted false data or made false representations, warranties, or certifications in connection with any aspect of TEAM.
(5) Has undergone a change in control that presents a program integrity risk.
(6) Is subject to any sanctions of an accrediting organization or a Federal, State, or local government agency.
(7) Is subject to investigation or action by HHS (including the HHS Office of Inspector General and CMS) or the Department of Justice due to an allegation of fraud or significant misconduct, including any of the following:
(i) Being subject to the filing of a complaint or filing of a criminal charge.
(ii) Being subject to an indictment.
(iii) Being named as a defendant in a False Claims Act qui tam matter in which the Federal government has intervened, or similar action.
(8) Has failed to demonstrate improved performance following any remedial action imposed under this section.
(9) Has misused or disclosed beneficiary-identifiable data in a manner that violates any applicable statutory or regulatory requirements or that is otherwise non-compliant with the provisions of the TEAM data sharing agreement.
(b) Remedial actions. If CMS determines that one or more grounds for remedial action described in paragraph (a) of this section has taken place, CMS may take one or more of the following remedial actions:
(1) Notify the TEAM participant and, if appropriate, require the TEAM participant to notify its downstream participants of the violation.
(2) Require the TEAM participant to provide additional information to CMS or its designees.
(3) Subject the TEAM participant to additional monitoring, auditing, or both.
(4) Prohibit the TEAM participant from distributing TEAM payments, as applicable.
(5) Require the TEAM participant to terminate, immediately or by a deadline specified by CMS, its agreement with a downstream participant with respect to TEAM.
(6) Require the TEAM participant to submit a corrective action plan in a form and manner and by a date specified by CMS.
(7) Discontinue the provision of data sharing and reports to the TEAM participant.
(8) Recoup TEAM payments.
(9) Reduce or eliminate a TEAM payment otherwise owed to the TEAM participant.
(10) Such other action as may be permitted under the terms of this part.
There is no administrative or judicial review under sections 1869 or 1878 of the Act or otherwise for all of the following:
(a) The selection of models for testing or expansion under section 1115A of the Act.
(b) The selection of organizations, sites, or participants to test TEAM, including a decision by CMS to remove a TEAM participant or to require a TEAM participant to remove a downstream participant from TEAM.
(c) The elements, parameters, scope, and duration of testing or dissemination, including without limitation the following:
(1) The selection of quality performance standards for TEAM by CMS.
(2) The methodology used by CMS to assess the quality of care furnished by the TEAM participant.
(3) The methodology used by CMS to attribute TEAM beneficiaries to the TEAM participant, if applicable.
(d) Determinations regarding budget neutrality under section 1115A(b)(3) of the Act.
(e) The termination or modification of the design and implementation of TEAM under section 1115A(b)(3)(B) of the Act.
(f) Determinations about expansion of the duration and scope of TEAM under section 1115A(c) of the Act, including the determination that TEAM is not expected to meet criteria described in paragraph (a) or (b) of this section.
(a) Notice of bankruptcy. If the TEAM participant has filed a bankruptcy petition, whether voluntary or involuntary, the TEAM participant must provide written notice of the bankruptcy to CMS and to the U.S. Attorney's Office in the district where the bankruptcy was filed, unless final payment has been made by either CMS or the TEAM participant under the terms of TEAM and all administrative or judicial review proceedings relating to any TEAM payments have been fully and finally resolved.
(1) The notice of bankruptcy must be sent by certified mail no later than 5 days after the petition has been filed and must contain a copy of the filed bankruptcy petition (including its docket number).
(2) The notice to CMS must be addressed to the CMS Office of Financial Management at 7500 Security Boulevard, Mailstop C3-01-24, Baltimore, MD 21244 or such other address as may be specified on the CMS website for purposes of receiving such notices.
(b) Notice of legal name change. A TEAM participant must furnish written notice to CMS within 30 days of any change in its legal name becomes effective. The notice of legal name change must meet all of the following:
(1) Be in a form and manner specified by CMS.
(2) Include a copy of the legal document effecting the name change, which must be authenticated by the appropriate State official. ( print page 69938)
(c) Notice of change in control. (1) A TEAM participant must furnish written notice to CMS in a form and manner specified by CMS at least 90 days before any change in control becomes effective.
(2) If CMS determines, in accordance with § 512.592(a)(5), that a TEAM participant's change in control would present a program integrity risk, CMS may—
(i) Take remedial action against the TEAM participant under § 512.160(b).
(ii) Require immediate reconciliation and payment of all monies owed to CMS by a TEAM participant that is subject to a change in control.
(a) Termination of TEAM. (1) CMS may terminate TEAM for reasons including, but not limited to, the following:
(i) CMS determines that it no longer has the funds to support TEAM.
(ii) CMS terminates TEAM in accordance with section 1115A(b)(3)(B) of the Act.
(2) If CMS terminates TEAM, CMS provides written notice to the TEAM participant specifying the grounds for termination and the effective date of such termination.
(b) Notice of a TEAM participant's termination from TEAM. If a TEAM participant receives notification that it has been terminated from TEAM and wishes to dispute the termination, it must provide a written notice to CMS requesting review of the termination within 10 calendar days of the notice.
(1) CMS has 30 days to respond to the TEAM participant's request for review.
(2) If the TEAM participant fails to notify CMS, the termination is deemed final.
(a) Voluntary reporting. A TEAM participant may elect to respond to questions and report metrics related to the TEAM participant's, or the TEAM participant's corporate affiliate's, emissions to CMS on an annual basis following each performance period. Voluntary reporting includes the following metrics:
(1) Organizational questions, which are a set of questions about the TEAM participants' sustainability team and sustainability activities.
(2) Building energy metrics, which are a set of metrics related to measuring and reporting GHG emissions related to energy use at TEAM participant facilities.
(i) Building energy metrics are based on the ENERGY STAR® Portfolio Manager® guidelines for the time of submission. TEAM participants reporting these metrics must submit using ENERGY STAR Portfolio Manager in the manner described in paragraph (b) of this section.
(ii) Metrics to be collected include all of the following:
(A) ENERGY STAR® Score for Hospitals as defined in the ENERGY STAR® Portfolio Manager® as well as supporting data which may include energy use intensity, electricity, natural gas, and other source emissions and normalizing factors such as building size, number of full-time equivalent workers, number of staffed beds, number of magnetic resonance imaging machines, zip codes, and heating and cooling days, as specified in the ENERGY STAR® Portfolio Manager®.
(B) Energy cost, to capture total energy costs, as specified in the ENERGY STAR® Portfolio Manager®.
(C) Total, direct, and indirect GHG emissions and emissions intensity as specified in the ENERGY STAR® Portfolio Manager®.
(3) Anesthetic gas metrics, which are a set of metrics related to measuring and managing emissions from anesthetic gas which include all of the following:
(i) Total greenhouse gas emissions from inhaled anesthetics based on purchase records.
(ii) Normalization factors that may include information on anesthetic hours, operating rooms, or MAC-hour equivalents.
(iii) Assessment questions based on key actions recommended for reducing emissions for anesthetic gases.
(4) Transportation metrics, which are a set metrics that focus on greenhouse gases related to leased or owned vehicles and may include any of the following:
(i) Gallons for owned and leased vehicles.
(ii) Normalization factors that may include patient encounter volume and the number of full-time equivalent (FTE) employees.
(iii) Assessment questions on key actions to reduce transportation emissions.
(b) Manner and timing of reporting. (1) If the TEAM participant elects to report the metrics in paragraph (b) of this section to CMS, such information must be reported to CMS in a form and manner specified by CMS for each performance year, including the use of ENERGY STAR® Portfolio Manager® for the building energy metrics at paragraph (a)(2) of this section and a survey and questionnaire for questions and metrics at paragraphs (a)(1), (3), and (4) of this section.
(2) If the TEAM participant chooses to participate, the TEAM participant must report the information to CMS—
(i) No later than 120 days in the year following the performance year; or
(ii) A later date as specified by CMS.
(c) Individualized feedback reports; recognition. If a TEAM participant elects to report all the metrics specified in paragraph (a) of this section to CMS, in the manner specified in paragraph (b) of this section, CMS annually provides the TEAM participant with the following:
(1) Individualized feedback reports, which may summarize facilities' emissions metrics and may include benchmarks, as feasible, for normalized metrics to compare facilities, in aggregate, to other TEAM participants in the Decarbonization and Resilience Initiative. A TEAM participant that receives individualized feedback reports from CMS must request approval from CMS in writing and receive written approval from CMS prior to publication or public disclosure of data or information contained in the individualized feedback reports.
(2) Publicly reported hospital recognition for the TEAM participant's commitment to decarbonization through a hospital recognition badge publicly reported on a CMS website, which may include recognition of the TEAM participant's corporate affiliates when such data has been submitted as specified in paragraph (a) of this section.
Xavier Becerra,
Secretary, Department of Health and Human Services.
In this Addendum, we are setting forth a description of the methods and data we used to determine the prospective payment rates for Medicare hospital inpatient operating costs and Medicare hospital inpatient capital-related costs for FY 2025 for acute care hospitals. We also are setting forth the rate-of-increase percentage for updating the target amounts for certain hospitals excluded from the IPPS for FY 2025. We note that, because certain hospitals excluded from the IPPS are paid on a reasonable cost basis subject to a rate-of-increase ceiling (and not by the IPPS), ( print page 69939) these hospitals are not affected by the figures for the standardized amounts, offsets, and budget neutrality factors. Therefore, in this final rule, we are setting forth the rate-of-increase percentage for updating the target amounts for certain hospitals excluded from the IPPS that would be effective for cost reporting periods beginning on or after October 1, 2024. In addition, we are setting forth a description of the methods and data we used to determine the LTCH PPS standard Federal payment rate that would be applicable to Medicare LTCHs for FY 2025.
In general, except for SCHs and MDHs, for FY 2025, each hospital's payment per discharge under the IPPS is based on 100 percent of the Federal national rate, also known as the national adjusted standardized amount. This amount reflects the national average hospital cost per case from a base year, updated for inflation.
SCHs are paid based on whichever of the following rates yields the greatest aggregate payment:
- The Federal national rate (including, as discussed in section IV.E. of the preamble of this final rule, uncompensated care payments under section 1886(r)(2) of the Act).
- The updated hospital-specific rate based on FY 1982 costs per discharge.
- The updated hospital-specific rate based on FY 1987 costs per discharge.
- The updated hospital-specific rate based on FY 1996 costs per discharge.
- The updated hospital-specific rate based on FY 2006 costs per discharge.
Under section 1886(d)(5)(G) of the Act, MDHs historically were paid based on the Federal national rate or, if higher, the Federal national rate plus 50 percent of the difference between the Federal national rate and the updated hospital-specific rate based on FY 1982 or FY 1987 costs per discharge, whichever was higher. However, section 5003(a)(1) of Public Law 109-171 extended and modified the MDH special payment provision that was previously set to expire on October 1, 2006, to include discharges occurring on or after October 1, 2006, but before October 1, 2011. Under section 5003(b) of Public Law 109-171 , if the change results in an increase to an MDH's target amount, we must rebase an MDH's hospital specific rates based on its FY 2002 cost report. Section 5003(c) of Public Law 109-171 further required that MDHs be paid based on the Federal national rate or, if higher, the Federal national rate plus 75 percent of the difference between the Federal national rate and the updated hospital specific rate. Further, based on the provisions of section 5003(d) of Public Law 109-171 , MDHs are no longer subject to the 12-percent cap on their DSH payment adjustment factor. Section 4102 of the Consolidated Appropriations Act, 2023 ( Pub. L. 117-328 ), enacted on December 29, 2022, extended the MDH program through FY 2024 (that is, for discharges occurring on or before September 30, 2024). Subsequently, section 307 of the Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, further extended the MDH program for FY 2025 discharges occurring before January 1, 2025. Prior to enactment of the CAA, 2024, the MDH program was only to be in effect through the end of FY 2024. Under current law, the MDH program will expire for discharges on or after January 1, 2025. We refer readers to section V.F. of the preamble of this final rule for further discussion of the MDH program.
As discussed in section V.B.2. of the preamble of this final rule, section 1886(n)(6)(B) of the Act was amended to specify that the adjustments to the applicable percentage increase under section 1886(b)(3)(B)(ix) of the Act apply to subsection (d) Puerto Rico hospitals that are not meaningful EHR users, effective beginning FY 2022. In general, Puerto Rico hospitals are paid 100 percent of the national standardized amount and are subject to the same national standardized amount as subsection (d) hospitals that receive the full update. Accordingly, our discussion later in this section does not include references to the Puerto Rico standardized amount or the Puerto Rico-specific wage index.
As discussed in section II. of this Addendum, we are making changes in the determination of the prospective payment rates for Medicare inpatient operating costs for acute care hospitals for FY 2025. In section III. of this Addendum, we discuss our policy changes for determining the prospective payment rates for Medicare inpatient capital-related costs for FY 2025. In section IV. of this Addendum, we are setting forth the rate-of-increase percentage for determining the rate-of-increase limits for certain hospitals excluded from the IPPS for FY 2025. In section V. of this Addendum, we discuss policy changes for determining the LTCH PPS standard Federal rate for LTCHs paid under the LTCH PPS for FY 2025. The tables to which we refer in the preamble of this final rule are listed in section VI. of this Addendum and are available via the internet on the CMS website.
The basic methodology for determining prospective payment rates for hospital inpatient operating costs for acute care hospitals for FY 2005 and subsequent fiscal years is set forth under § 412.64. The basic methodology for determining the prospective payment rates for hospital inpatient operating costs for hospitals located in Puerto Rico for FY 2005 and subsequent fiscal years is set forth under §§ 412.211 and 412.212. In this section, we discuss the factors we are using for determining the prospective payment rates for FY 2025.
In summary, the standardized amounts set forth in Tables 1A, 1B, and 1C that are listed and published in section VI. of this Addendum (and available via the internet on the CMS website) reflect—
- Equalization of the standardized amounts for urban and other areas at the level computed for large urban hospitals during FY 2004 and onward, as provided for under section 1886(d)(3)(A)(iv)(II) of the Act.
- The labor-related share that is applied to the standardized amounts to give the hospital the highest payment, as provided for under sections 1886(d)(3)(E) and 1886(d)(9)(C)(iv) of the Act. For FY 2025, depending on whether a hospital submits quality data under the rules established in accordance with section 1886(b)(3)(B)(viii) of the Act (hereafter referred to as a hospital that submits quality data) and is a meaningful EHR user under section 1886(b)(3)(B)(ix) of the Act (hereafter referred to as a hospital that is a meaningful EHR user), there are four possible applicable percentage increases that can be applied to the national standardized amount.
We refer readers to section V.B. of the preamble of this final rule for a complete discussion on the FY 2025 inpatient hospital update. The table that follows shows these four scenarios:

We note that section 1886(b)(3)(B)(viii) of the Act, which specifies the adjustment to the applicable percentage increase for “subsection (d)” hospitals that do not submit quality data under the rules established by the Secretary, is not applicable to hospitals located in Puerto Rico. In addition, section 602 of Public Law 114-113 amended section 1886(n)(6)(B) of the Act to specify that Puerto Rico hospitals are eligible for incentive payments for the meaningful use of certified EHR technology, effective beginning FY 2016, and also to apply the adjustments to the applicable percentage increase under section 1886(b)(3)(B)(ix) of the Act to subsection (d) Puerto Rico hospitals that are not meaningful EHR users, effective beginning FY 2022. Accordingly, the applicable percentage increase for subsection (d) Puerto Rico hospitals that are not meaningful EHR users for FY 2025 and subsequent fiscal years is adjusted by the adjustment for failure to be a meaningful EHR user under section 1886(b)(3)(B)(ix) of the Act. The regulations at 42 CFR 412.64(d)(3)(ii) reflect the current law for the update for subsection (d) Puerto Rico hospitals for FY 2022 and subsequent fiscal years.
- An adjustment to the standardized amount to ensure budget neutrality for DRG recalibration and reclassification, as provided for under section 1886(d)(4)(C)(iii) of the Act.
- An adjustment to the standardized amount to ensure budget neutrality for the permanent 10 percent cap on the reduction in a MS-DRG's relative weight in a given fiscal year, as discussed in section II.D.2.c. of the preamble of this final rule, consistent with our current methodology for implementing DRG recalibration and reclassification budget neutrality under section 1886(d)(4)(C)(iii) of the Act.
- An adjustment to ensure the wage index and labor-related share changes (depending on the fiscal year) are budget neutral, as provided for under section 1886(d)(3)(E)(i) of the Act (as discussed in the FY 2006 IPPS final rule ( 70 FR 47395 ) and the FY 2010 IPPS final rule ( 74 FR 44005 )). We note that section 1886(d)(3)(E)(i) of the Act requires that when we compute such budget neutrality, we assume that the provisions of section 1886(d)(3)(E)(ii) of the Act (requiring a 62-percent labor-related share in certain circumstances) had not been enacted.
- An adjustment to ensure the effects of geographic reclassification are budget neutral, as provided for under section 1886(d)(8)(D) of the Act, by removing the FY 2024 budget neutrality factor and applying a revised factor.
- An adjustment to the standardized amount to implement in a budget neutral manner the increase in the wage index values for hospitals with a wage index value below the 25th percentile wage index value across all hospitals (as described in section III.G.5 of the preamble of this final rule).
- An adjustment to the standardized amount to implement in a budget neutral manner the wage index cap policy (as described in section III.G.6. of the preamble of this final rule).
- An adjustment to ensure the effects of the Rural Community Hospital Demonstration program required under section 410A of Public Law 108-173 (as amended by sections 3123 and 10313 of Public Law 111-148 , which extended the demonstration program for an additional 5 years and section 15003 of Public Law 114-255 ), are budget neutral as required under section 410A(c)(2) of Public Law 108-173 .
- An adjustment to remove the FY 2024 outlier offset and apply an offset for FY 2025, as provided for in section 1886(d)(3)(B) of the Act.
For FY 2025, consistent with current law, we are applying the rural floor budget neutrality adjustment to hospital wage indexes. Also, consistent with section 3141 of the Affordable Care Act, instead of applying a State-level rural floor budget neutrality adjustment to the wage index, we are applying a uniform, national budget neutrality adjustment to the FY 2025 wage index for the rural floor.
For FY 2025, as we proposed, we are continuing to not remove the Stem Cell Acquisition Budget Neutrality Factor from the prior year's standardized amount and to not apply a new factor. If we removed the prior year's adjustment, we would not satisfy budget neutrality. We believe this approach ensures the effects of the reasonable cost-based payment for allogeneic hematopoietic stem cell acquisition costs under section 108 of the Further Consolidated Appropriations Act, 2020 ( Pub. L. 116-94 ) are budget neutral as required under section 108 of Public Law 116-94 . For a discussion of Stem Cell Acquisition Budget Neutrality Factor, we refer the reader to the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 59032 and 59033 ).
In general, the national standardized amount is based on per discharge averages of adjusted hospital costs from a base period (section 1886(d)(2)(A) of the Act), updated and otherwise adjusted in accordance with the provisions of section 1886(d) of the Act. The September 1, 1983 interim final rule ( 48 FR 39763 ) contained a detailed explanation of how base-year cost data ( print page 69941) (from cost reporting periods ending during FY 1981) were established for urban and rural hospitals in the initial development of standardized amounts for the IPPS.
Sections 1886(d)(2)(B) and 1886(d)(2)(C) of the Act require us to update base-year per discharge costs for FY 1984 and then standardize the cost data in order to remove the effects of certain sources of cost variations among hospitals. These effects include case-mix, differences in area wage levels, cost-of-living adjustments for Alaska and Hawaii, IME costs, and costs to hospitals serving a disproportionate share of low-income patients.
For FY 2025, as we proposed, we are continuing to use the national labor-related and nonlabor-related shares (which are based on the 2018-based hospital IPPS market basket) that were used in FY 2024. Specifically, under section 1886(d)(3)(E) of the Act, the Secretary estimates, from time to time, the proportion of payments that are labor-related and adjusts the proportion (as estimated by the Secretary from time to time) of hospitals' costs which are attributable to wages and wage-related costs of the DRG prospective payment rates. We refer to the proportion of hospitals' costs that are attributable to wages and wage-related costs as the “labor-related share.” For FY 2025, as discussed in section III.I. of the preamble of this final rule, as we proposed, we are using a labor-related share of 67.6 percent for the national standardized amounts for all IPPS hospitals (including hospitals in Puerto Rico) that have a wage index value that is greater than 1.0000. Consistent with section 1886(d)(3)(E) of the Act, as we proposed, we are applying the wage index to a labor-related share of 62 percent of the national standardized amount for all IPPS hospitals (including hospitals in Puerto Rico) whose wage index values are less than or equal to 1.0000.
The standardized amounts for operating costs appear in Tables 1A, 1B, and 1C that are listed and published in section VI. of the Addendum to this final rule and are available via the internet on the CMS website.
Section 1886(d)(3)(A)(iv)(II) of the Act requires that, beginning with FY 2004 and thereafter, an equal standardized amount be computed for all hospitals at the level computed for large urban hospitals during FY 2003, updated by the applicable percentage increase. Accordingly, as proposed, we are calculating the FY 2025 national average standardized amount irrespective of whether a hospital is located in an urban or rural location.
Section 1886(b)(3)(B) of the Act specifies the applicable percentage increase used to update the standardized amount for payment for inpatient hospital operating costs. We note that, in compliance with section 404 of the MMA, we are using the 2018-based IPPS operating and capital market baskets for FY 2025. As discussed in section V.B. of the preamble of this final rule, in accordance with section 1886(b)(3)(B) of the Act, as amended by section 3401(a) of the Affordable Care Act, we are reducing the FY 2025 applicable percentage increase (which for this final rule is based on IGI's second quarter 2024 forecast of the 2018-based IPPS market basket) by the productivity adjustment, as discussed elsewhere in this final rule.
Based on IGI's second quarter 2024 forecast of the hospital market basket percentage increase (as discussed in Appendix B of this final rule), the forecast of the hospital market basket percentage increase for FY 2025 for this final rule is 3.4 percent and the forecast of the productivity adjustment for FY 2025 for this final rule is 0.5 percent. As discussed earlier, for FY 2025, depending on whether a hospital submits quality data under the rules established in accordance with section 1886(b)(3)(B)(viii) of the Act and is a meaningful EHR user under section 1886(b)(3)(B)(ix) of the Act, there are four possible applicable percentage increases that can be applied to the standardized amount. We refer readers to section V.B. of the preamble of this final rule for a complete discussion on the FY 2025 inpatient hospital update to the standardized amount. We also refer readers to the previous table for the four possible applicable percentage increases that would be applied to update the national standardized amount. The standardized amounts shown in Tables 1A through 1C that are published in section VI. of this Addendum and that are available via the internet on the CMS website reflect these differential amounts.
Although the update factors for FY 2025 are set by law, we are required by section 1886(e)(4) of the Act to recommend, taking into account MedPAC's recommendations, appropriate update factors for FY 2025 for both IPPS hospitals and hospitals and hospital units excluded from the IPPS. Section 1886(e)(5)(A) of the Act requires that we publish our recommendations in the Federal Register for public comment. Our recommendation on the FY 2025 update factors is set forth in appendix B of this final rule.
The methodology we used to calculate the FY 2025 standardized amount is as follows:
- To ensure we are only including hospitals paid under the IPPS in the calculation of the standardized amount, we applied the following inclusion and exclusion criteria: include hospitals whose last four digits fall between 0001 and 0879 (section 2779A1 of Chapter 2 of the State Operations Manual on the CMS website at: https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/som107c02.pdf ); exclude CAHs at the time of this final rule; exclude hospitals in Maryland (because these hospitals are paid under an all payer model under section 1115A of the Act); and remove PPS excluded- cancer hospitals that have a “V” in the fifth position of their provider number or a “E” or “F” in the sixth position.
Section 125 of Division CC (section 125) of the CAA 2021 established a new rural Medicare provider type: Rural Emergency Hospitals (REHs). (We refer the reader to the CMS website at https://www.cms.gov/medicare/health-safety-standards/guidance-for-laws-regulations/hospitals/rural-emergency-hospitals for additional information on REHs.) In doing so, section 125 amended section 1861(e) of the Act, which provides the definition of a hospital and states that the term “hospital” does not include, unless the context otherwise requires, a critical access hospital (as defined in subsection (mm)(1)) or a rural emergency hospital (as defined in subsection (kkk)(2)). Section 125 also added section 1861(kkk) to the Act, which sets forth the requirements for REHs. Per section 1861(kkk)(2) of the Act, one of the requirements for an REH is that it does not provide any acute care inpatient services (other than post-hospital extended care services furnished in a distinct part unit licensed as a skilled nursing facility (SNF)). Therefore, we believe hospitals that have subsequently converted to REH status should be removed from the calculation of the standardized amount, because they are a separately certified Medicare provider type and are not comparable to other short-term, acute care hospitals as they do not provide inpatient hospital services. For FY 2025, we proposed to exclude REHs from the calculation of the standardized amount, including ( print page 69942) hospitals that subsequently became REHs after the period from which the data were taken. We did not receive any comments with regard to this proposal, and we are finalizing as proposed to exclude hospitals that have subsequently converted to REH from the calculation of the standardized amount, including hospitals that subsequently became REHs after the period from which the data were taken.
- As in the past, we are adjusting the FY 2025 standardized amount to remove the effects of the FY 2024 geographic reclassifications and outlier payments before applying the FY 2025 updates. We then applied budget neutrality offsets for outliers and geographic reclassifications to the standardized amount based on FY 2025 payment policies.
- We do not remove the prior year's budget neutrality adjustments for reclassification and recalibration of the DRG relative weights and for updated wage data because, in accordance with sections 1886(d)(4)(C)(iii) and 1886(d)(3)(E) of the Act, estimated aggregate payments after updates in the DRG relative weights and wage index should equal estimated aggregate payments prior to the changes. If we removed the prior year's adjustment, we would not satisfy these conditions.
Budget neutrality is determined by comparing aggregate IPPS payments before and after making changes that are required to be budget neutral (for example, changes to MS-DRG classifications, recalibration of the MS-DRG relative weights, updates to the wage index, and different geographic reclassifications). We include outlier payments in the simulations because they may be affected by changes in these parameters.
- Consistent with our methodology established in the FY 2011 IPPS/LTCH PPS final rule ( 75 FR 50422 through 50433 ), because IME Medicare Advantage payments are made to IPPS hospitals under section 1886(d) of the Act, we believe these payments must be part of these budget neutrality calculations. However, we note that it is not necessary to include Medicare Advantage IME payments in the outlier threshold calculation or the outlier offset to the standardized amount because the statute requires that outlier payments be not less than 5 percent nor more than 6 percent of total “operating DRG payments,” which does not include IME and DSH payments. We refer readers to the FY 2011 IPPS/LTCH PPS final rule for a complete discussion on our methodology of identifying and adding the total Medicare Advantage IME payment amount to the budget neutrality adjustments.
- Consistent with the methodology in the FY 2012 IPPS/LTCH PPS final rule, in order to ensure that we capture only fee-for-service claims, we are only including claims with a “Claim Type” of 60 (which is a field on the MedPAR file that indicates a claim is an FFS claim).
- Consistent with our methodology established in the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57277 ), in order to further ensure that we capture only FFS claims, we are excluding claims with a “GHOPAID” indicator of 1 (which is a field on the MedPAR file that indicates a claim is not an FFS claim and is paid by a Group Health Organization).
- Consistent with our methodology established in the FY 2011 IPPS/LTCH PPS final rule ( 75 FR 50422 through 50423 ), we examine the MedPAR file and remove pharmacy charges for anti-hemophilic blood factor (which are paid separately under the IPPS) with an indicator of “3” for blood clotting with a revenue code of “0636” from the covered charge field for the budget neutrality adjustments. We are removing organ acquisition charges, except for cases that group to MS-DRG 018, from the covered charge field for the budget neutrality adjustments because organ acquisition is a pass-through payment not paid under the IPPS. Revenue centers 081X-089X are typically excluded from ratesetting, however, we are not removing revenue center 891 charges from MS-DRG 018 claims during ratesetting because those revenue 891 charges were included in the relative weight calculation for MS-DRG 018, which is consistent with the policy finalized in the FY 2021 final rule ( 85 FR 58600 ). We note that a new MedPAR variable for revenue code 891 charges was introduced in April 2020.
- For FY 2025, we are continuing to remove allogeneic hematopoietic stem cell acquisition charges from the covered charge field for budget neutrality adjustments. As discussed in the FY 2021 IPPS/LTCH PPS final rule, payment for allogeneic hematopoietic stem cell acquisition costs is made on a reasonable cost basis for cost reporting periods beginning on or after October 1, 2020 ( 85 FR 58835 through 58842 ).
- The participation of hospitals under the BPCI (Bundled Payments for Care Improvement) Advanced model started on October 1, 2018. The BPCI Advanced model, tested under the authority of section 3021 of the Affordable Care Act (codified at section 1115A of the Act), is comprised of a single payment and risk track, which bundles payments for multiple services beneficiaries receive during a Clinical Episode. Acute care hospitals may participate in the BPCI Advanced model in one of two capacities: as a model Participant or as a downstream Episode Initiator. Regardless of the capacity in which they participate in the BPCI Advanced model, participating acute care hospitals would continue to receive IPPS payments under section 1886(d) of the Act. Acute care hospitals that are participants also assume financial and quality performance accountability for Clinical Episodes in the form of a reconciliation payment. For additional information on the BPCI Advanced model, we refer readers to the BPCI Advanced web page on the CMS Center for Medicare and Medicaid Innovation's website at: https://innovation.cms.gov/initiatives/bpci-advanced/ .
For FY 2025, consistent with how we treated hospitals that participated in the BPCI Advanced Model in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 59029 and 59030 ), as we proposed, we are including all applicable data from subsection (d) hospitals participating in the BPCI Advanced model in our IPPS payment modeling and ratesetting calculations. We believe it is appropriate to include all applicable data from the subsection (d) hospitals participating in the BPCI Advanced model in our IPPS payment modeling and ratesetting calculations because these hospitals are still receiving IPPS payments under section 1886(d) of the Act. For the same reasons, as we proposed, we included all applicable data from subsection (d) hospitals participating in the Comprehensive Care for Joint Replacement (CJR) Model in our IPPS payment modeling and ratesetting calculations.
- Consistent with our methodology established in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53687 through 53688 ), we believe that it is appropriate to include adjustments for the Hospital Readmissions Reduction Program and the Hospital VBP Program (established under the Affordable Care Act) within our budget neutrality calculations.
Both the hospital readmissions payment adjustment (reduction) and the hospital VBP payment adjustment (redistribution) are applied on a claim-by-claim basis by adjusting, as applicable, the base-operating DRG payment amount for individual subsection (d) hospitals, which affects the overall sum of aggregate payments on each side of the comparison within the budget neutrality calculations.
In order to properly determine aggregate payments on each side of the comparison, consistent with the ( print page 69943) approach we have taken in prior years, for FY 2025, we are applying a proxy based on the prior fiscal year hospital readmissions payment adjustment and a proxy based on the prior fiscal year hospital VBP payment adjustment on each side of the comparison, consistent with the methodology that we adopted in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53687 through 53688 ). Under this policy for FY 2025, we used the final FY 2024 readmissions adjustment factors from Table 15 of the FY 2024 IPPS/LTCH PPS final rule and the final FY 2024 hospital VBP adjustment factors from Table 16B of the FY 2024 IPPS/LTCH PPS final rule. These proxy factors are applied on both sides of our comparison of aggregate payments when determining all budget neutrality factors described in section II.A.4. of this Addendum. We refer the reader to section V.K. of the preamble of this final rule for a complete discussion on the Hospital Readmissions Reduction Program and section V.L. of the preamble of this final rule for a complete discussion on the Hospital VBP Program.
- The Affordable Care Act also established section 1886(r) of the Act, which modifies the methodology for computing the Medicare DSH payment adjustment beginning in FY 2014. Beginning in FY 2014, IPPS hospitals receiving Medicare DSH payment adjustments receive an empirically justified Medicare DSH payment equal to 25 percent of the amount that would previously have been received under the statutory formula set forth under section 1886(d)(5)(F) of the Act governing the Medicare DSH payment adjustment. In accordance with section 1886(r)(2) of the Act, the remaining amount, equal to an estimate of 75 percent of what otherwise would have been paid as Medicare DSH payments, reduced to reflect changes in the percentage of individuals who are uninsured and any additional statutory adjustment, is available to make additional payments to Medicare DSH hospitals based on their share of the total amount of uncompensated care reported by Medicare DSH hospitals for a given time period. In order to properly determine aggregate payments on each side of the comparison for budget neutrality, prior to FY 2014, we included estimated Medicare DSH payments on both sides of our comparison of aggregate payments when determining all budget neutrality factors described in section II.A.4. of this Addendum.
To do this for FY 2025 (as we did for the last 11 fiscal years), as we proposed, we are including estimated empirically justified Medicare DSH payments that would be paid in accordance with section 1886(r)(1) of the Act and estimates of the additional uncompensated care payments made to hospitals receiving Medicare DSH payment adjustments as described by section 1886(r)(2) of the Act. That is, we considered estimated empirically justified Medicare DSH payments at 25 percent of what would otherwise have been paid, and also the estimated additional uncompensated care payments for hospitals receiving Medicare DSH payment adjustments on both sides of our comparison of aggregate payments when determining all budget neutrality factors described in section II.A.4. of this Addendum.
We also are including the estimated supplemental payments for eligible IHS/Tribal hospitals and Puerto Rico hospitals on both sides of our comparison of aggregate payments when determining all budget neutrality factors described in section II.A.4. of this Addendum.
- When calculating total payments for budget neutrality, to determine total payments for SCHs, we model total hospital-specific rate payments and total Federal rate payments and then include whichever one of the total payments is greater. As discussed in section IV.G. of the preamble to this final rule and later in this section, we proposed to continue to use the FY 2014 finalized methodology under which we take into consideration uncompensated care payments in the comparison of payments under the Federal rate and the hospital-specific rate for SCHs. Therefore, we are including estimated uncompensated care payments in this comparison.
As discussed elsewhere in this final rule, section 307 of the Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, extended the MDH program for FY 2025 discharges occurring before January 1, 2025. Prior to enactment of the CAA, 2024, the MDH program was only to be in effect through the end of FY 2024. Therefore, under current law, the MDH program will expire for discharges on or after January 1, 2025. As a result, MDHs that currently receive the higher of payments made based on the Federal rate or the payments made based on the Federal rate plus 75 percent of the difference between payments based on the Federal rate and the hospital-specific rate will be paid based on the Federal rate starting January 1, 2025. In the proposed rule we stated that because of the timing of this legislation, the total payments for budget neutrality in the proposed rule did not reflect the extension of the MDH program for the first quarter of FY 2025. We further stated in the proposed rule that this extension will be reflected in the total payments for budget neutrality for the final rule. For this final rule, approximately 117 hospitals would receive payments under the MDH program for the first quarter of FY 2025. Upon further review and consideration, given the limited magnitude, for this final rule we did not include this extension in the total payments for budget neutrality. Accordingly, for this final rule, the budget neutrality factor calculations do not reflect the extension of the MDH program for the first quarter of FY 2025.
- As proposed, we included an adjustment to the standardized amount for those hospitals that are not meaningful EHR users in our modeling of aggregate payments for budget neutrality for FY 2025. Similar to FY 2024, we are including this adjustment based on data on the prior year's performance. Payments for hospitals would be estimated based on the applicable standardized amount in Tables 1A and 1B for discharges occurring in FY 2025.
- In our determination of all budget neutrality factors described in section II.A.4. of this Addendum, we used transfer-adjusted discharges.
We note, in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49414 through 49415 ), we finalized a change to the ordering of the budget neutrality factors in the calculation so that the RCH Demonstration budget neutrality factor is applied after all wage index and other budget neutrality factors. We refer the reader to the FY 2023 IPPS/LTCH PPS final rule for further discussion.
We note that the wage index value is calculated and assigned to a hospital based on the hospital's labor market area. Under section 1886(d)(3)(E) of the Act, beginning with FY 2005, we delineate hospital labor market areas based on the Core-Based Statistical Areas (CBSAs) established by the Office of Management and Budget (OMB). The current statistical areas used in FY 2024 are based on the OMB delineations that were adopted beginning with FY 2015 (based on the revised delineations issued in OMB Bulletin No. 13-01) to calculate the area wage indexes, with updates as reflected in OMB Bulletin Nos. 15-01, 17-01, and 18-04. For purposes of determining all of the FY 2024 budget neutrality factors, we determined aggregate payments on each side of the comparison for our budget neutrality calculations using wage indexes based on the current CBSAs.
On July 21, 2023, OMB released Bulletin No. 23-01. A copy of OMB ( print page 69944) Bulletin No. 23-01 may be obtained at https://www.whitehouse.gov/wp-content/uploads/2023/07/OMB-Bulletin-23-01.pdf . According to OMB, the delineations reflect the 2020 Standards for Delineating Core Based Statistical Areas (“the 2020 Standards”), which appeared in the Federal Register on July 16, 2021 ( 86 FR 37770 through 37778 ), and the application of those standards to Census Bureau population and journey-to-work data (for example, 2020 Decennial Census, American Community Survey, and Census Population Estimates Program data). In order to implement these revised standards for the IPPS, it was necessary to identify the new OMB labor market area delineation for each county and hospital in the country. As stated in section III.B. of the preamble of this final rule, we believe that using the revised delineations based on OMB Bulletin No. 23-01 will increase the integrity of the IPPS wage index system by more accurately representing current geographic variations in wage levels. As discussed in section III. of the preamble of this final rule, we are finalizing to adopt the new OMB labor market area delineations as described in the July 21, 2023 OMB Bulletin No. 23-01, effective for the FY 2025 IPPS wage index.
Consistent with our policy to adopt the new OMB delineations, in order to properly determine aggregate payments on each side of the comparison for our budget neutrality calculations, we are using wage indexes based on the new OMB delineations in the determination of all of the budget neutrality factors discussed later in this section. We also note that, consistent with past practice as finalized in the FY 2005 IPPS final rule ( 69 FR 49034 ), we are not adopting the new OMB delineations themselves in a budget neutral manner. We continue to believe that the revision to the labor market areas in and of itself does not constitute an “adjustment or update” to the adjustment for area wage differences, as provided under section 1886(d)(3)(E) of the Act.
Section 1886(d)(4)(C)(iii) of the Act specifies that, beginning in FY 1991, the annual DRG reclassification and recalibration of the relative weights must be made in a manner that ensures that aggregate payments to hospitals are not affected. As discussed in section II.D. of the preamble of this final rule, we normalized the recalibrated MS-DRG relative weights by an adjustment factor so that the average case relative weight after recalibration is equal to the average case relative weight prior to recalibration. However, equating the average case relative weight after recalibration to the average case relative weight before recalibration does not necessarily achieve budget neutrality with respect to aggregate payments to hospitals because payments to hospitals are affected by factors other than average case relative weight. Therefore, as we have done in past years, we are making a budget neutrality adjustment to ensure that the requirement of section 1886(d)(4)(C)(iii) of the Act is met.
For this FY 2025 final rule, as we proposed, to comply with the requirement that MS-DRG reclassification and recalibration of the relative weights be budget neutral for the standardized amount and the hospital-specific rates, we used FY 2023 discharge data to simulate payments and compared the following:
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2024 labor-related share percentages, the FY 2024 relative weights, and the FY 2024 pre-reclassified wage data, and applied the proxy hospital readmissions payment adjustments and proxy hospital VBP payment adjustments (as described previously); and
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2024 labor-related share percentages, the FY 2025 relative weights before applying the 10 percent cap, and the FY 2024 pre-reclassified wage data, and applied the same proxy hospital readmissions payment adjustments and proxy hospital VBP payment adjustments applied previously.
Because this payment simulation uses the FY 2025 relative weights (before applying the 10 percent cap), consistent with our policy in section V.I. of the preamble to this final rule, we applied the adjustor for certain cases that group to MS-DRG 018 in our simulation of these payments. We note that because the simulations of payments for all of the budget neutrality factors discussed in this section also use the FY 2025 relative weights, we are applying the adjustor for certain MS-DRG 018 (Chimeric Antigen Receptor (CAR) T-cell and other immunotherapies) cases in all simulations of payments for the budget neutrality factors discussed later in this section. We refer the reader to section V.I. of the preamble of this final rule for a complete discussion on the adjustor for certain cases that group to MS-DRG 018 and to section II.D.2.b. of the preamble of this final rule, for a complete discussion of the adjustment to the FY 2025 relative weights to account for certain cases that group to MS-DRG 018.
Based on this comparison, we computed a budget neutrality adjustment factor and applied this factor to the standardized amount. As discussed in section IV. of this Addendum, we are applying the MS-DRG reclassification and recalibration budget neutrality factor to the hospital-specific rates that are effective for cost reporting periods beginning on or after October 1, 2024. Please see the table later in this section setting forth each of the FY 2025 budget neutrality factors.
As discussed in section II.D.2.c of the preamble of this final rule, in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48897 through 48900 ), we finalized a permanent 10-percent cap on the reduction in an MS-DRG's relative weight in a given fiscal year, beginning in FY 2023. As also discussed in section II.D.2.c of the preamble of this final rule, and consistent with our current methodology for implementing budget neutrality for MS-DRG reclassification and recalibration of the relative weights under section 1886(d)(4)(C)(iii) of the Act, we apply a budget neutrality adjustment to the standardized amount for all hospitals so that this 10-percent cap on relative weight reductions does not increase estimated aggregate Medicare payments beyond the payments that would be made had we never applied this cap. We refer the reader to the FY 2023 IPPS/LTCH PPS final rule for further discussion.
To calculate this budget neutrality adjustment factor for FY 2025, we used FY 2023 discharge data to simulate payments and compared the following:
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2024 labor-related share percentages, the FY 2025 relative weights before applying the 10-percent cap, and the FY 2024 pre-reclassified wage data, and applied the proxy FY 2025 hospital readmissions payment adjustments and the proxy FY 2025 hospital VBP payment adjustments; and
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2024 labor-related share percentages, the FY 2025 relative weights after applying the 10-percent cap, and the FY 2024 pre-reclassified wage data, and applied the same proxy FY 2025 hospital readmissions payment adjustments and proxy FY 2025 hospital VBP payment adjustments applied previously. ( print page 69945)
Because this payment simulation uses the FY 2025 relative weights, consistent with our proposal in section V.I. of the preamble to this final rule and our historical policy, and as discussed in the preceding section, we applied the adjustor for certain cases that group to MS-DRG 018 in our simulation of these payments.
In addition, we applied the MS-DRG reclassification and recalibration budget neutrality adjustment factor before the cap (derived in the first step) to the payment rates that were used to simulate payments for this comparison of aggregate payments from FY 2024 to FY 2025. Based on this comparison, we computed a budget neutrality adjustment factor and applied this factor to the standardized amount. As discussed in section IV. of this Addendum, as we proposed, we are applying this budget neutrality factor to the hospital-specific rates that are effective for cost reporting periods beginning on or after October 1, 2024. Please see the table later in this section setting forth each of the FY 2025 budget neutrality factors.
Section 1886(d)(3)(E)(i) of the Act requires us to update the hospital wage index on an annual basis beginning October 1, 1993. This provision also requires us to make any updates or adjustments to the wage index in a manner that ensures that aggregate payments to hospitals are not affected by the change in the wage index. Section 1886(d)(3)(E)(i) of the Act requires that we implement the wage index adjustment in a budget neutral manner. However, section 1886(d)(3)(E)(ii) of the Act sets the labor-related share at 62 percent for hospitals with a wage index less than or equal to 1.0000, and section 1886(d)(3)(E)(i) of the Act provides that the Secretary shall calculate the budget neutrality adjustment for the adjustments or updates made under that provision as if section 1886(d)(3)(E)(ii) of the Act had not been enacted. In other words, this section of the statute requires that we implement the updates to the wage index in a budget neutral manner, but that our budget neutrality adjustment should not take into account the requirement that we set the labor-related share for hospitals with wage indexes less than or equal to 1.0000 at the more advantageous level of 62 percent. Therefore, for purposes of this budget neutrality adjustment, section 1886(d)(3)(E)(i) of the Act prohibits us from taking into account the fact that hospitals with a wage index less than or equal to 1.0000 are paid using a labor-related share of 62 percent. Consistent with current policy, for FY 2025, as we proposed, we are adjusting 100 percent of the wage index factor for occupational mix. We describe the occupational mix adjustment in section III.E. of the preamble of this final rule.
To compute a budget neutrality adjustment factor for wage index and labor-related share percentage changes, we used FY 2023 discharge data to simulate payments and compared the following:
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2025 relative weights and the FY 2023 pre-reclassified wage indexes, applied the FY 2024 labor-related share of 67.6 percent to all hospitals (regardless of whether the hospital's wage index was above or below 1.0000), and applied the proxy FY 2025 hospital readmissions payment adjustment and the proxy FY 2025 hospital VBP payment adjustment.
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2025 relative weights and the proposed FY 2025 pre-reclassified wage indexes, applied the labor-related share for FY 2025 of 67.6 percent to all hospitals (regardless of whether the hospital's wage index was above or below 1.0000), and applied the same proxy FY 2025 hospital readmissions payment adjustments and proxy FY 2025 hospital VBP payment adjustments applied previously.
In addition, we applied the MS-DRG reclassification and recalibration budget neutrality adjustment factor before the proposed cap (derived in the first step) and the 10 percent cap on relative weight reductions adjustment factor (derived from the second step) to the payment rates that were used to simulate payments for this comparison of aggregate payments from FY 2024 to FY 2025. Based on this comparison, we computed a budget neutrality adjustment factor and applied this factor to the standardized amount for changes to the wage index. Please see the table later in this section for a summary of the FY 2025 budget neutrality factors.
Section 1886(d)(8)(B) of the Act provides that certain rural hospitals are deemed urban. In addition, section 1886(d)(10) of the Act provides for the reclassification of hospitals based on determinations by the MGCRB. Under section 1886(d)(10) of the Act, a hospital may be reclassified for purposes of the wage index.
Under section 1886(d)(8)(D) of the Act, the Secretary is required to adjust the standardized amount to ensure that aggregate payments under the IPPS after implementation of the provisions of sections 1886(d)(8)(B) and (C) and 1886(d)(10) of the Act are equal to the aggregate prospective payments that would have been made absent these provisions. Additionally, as discussed, changes in the wage index are generally budget neutralized. We note, in the FY 2024 IPPS/LTCH final rule ( 88 FR 58971 through 58977 ), we finalized a policy beginning with FY 2024 to include hospitals with § 412.103 reclassification along with geographically rural hospitals in all rural wage index calculations, and only exclude “dual reclass” hospitals (hospitals with simultaneous § 412.103 and MGCRB reclassifications) in accordance with the hold harmless provision at section 1886(d)(8)(C)(ii) of the Act. Consistent with the previous policy, beginning with FY 2024, we include the data of all § 412.103 hospitals (including those that have an MGCRB reclassification) in the calculation of “the wage index for rural areas in the State in which the county is located” as referred to in section 1886(d)(8)(C)(iii) of the Act.
We refer the reader to the FY 2015 IPPS final rule ( 79 FR 50371 and 50372 ) for a discussion regarding the requirement of section 1886(d)(8)(C)(iii) of the Act. We further note that the wage index adjustments provided for under section 1886(d)(13) of the Act are not budget neutral. Section 1886(d)(13)(H) of the Act provides that any increase in a wage index under section 1886(d)(13) of the Act shall not be taken into account in applying any budget neutrality adjustment with respect to such index under section 1886(d)(8)(D) of the Act. To calculate the budget neutrality adjustment factor for FY 2025, we used FY 2022 discharge data to simulate payments and compared the following:
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2025 labor-related share percentage, the FY 2025 relative weights, and the FY 2025 wage data prior to any reclassifications, and applied the proxy FY 2025 hospital readmissions payment adjustments and the proxy FY 2025 hospital VBP payment adjustments.
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2025 labor-related share percentage, the FY 2025 relative weights, and the FY 2025 wage data after such reclassifications, and applied the same proxy FY 2025 hospital readmissions payment adjustments and ( print page 69946) the proxy FY 2025 hospital VBP payment adjustments applied previously.
We note that the reclassifications applied under the second simulation and comparison are those listed in Table 2 associated with this final rule, which is available via the internet on the CMS website. This table reflects reclassification crosswalks for FY 2025 and applies the policies explained in section III. of the preamble of this final rule. Based on this comparison, we computed a budget neutrality adjustment factor and applied this factor to the standardized amount to ensure that the effects of these provisions are budget neutral, consistent with the statute. Please see the table later in this section for a summary of the FY 2025 budget neutrality factors.
The FY 2025 budget neutrality adjustment factor was applied to the standardized amount after removing the effects of the FY 2024 budget neutrality adjustment factor. We note that the FY 2025 budget neutrality adjustment reflects FY 2025 wage index reclassifications approved by the MGCRB or the Administrator at the time of development of this final rule.
Comment: A commenter requested that CMS confirm that if an urban hospital has reclassified as rural under § 412.103, the “before” wage index value for the hospital in this simulation would be equal to the rural wage index for its state. The commenter further asked for confirmation if was this CMS's policy prior to FY 2024, or did it originate in FY 2024 when CMS decided to regard § 412.103 hospitals as rural for purposes of the rural wage index.
Response: The “before” wage index value uses a hospitals area wage data before any reclassifications or state rural wage index is applied. This is also referred to as the pre reclassified wage index. Therefore, if an urban hospital has reclassified as rural under section § 412.103, the “before” wage index value would be based on the hospitals urban area wage index prior to any reclassification or application of the state rural wage index. We also confirm that this has been the policy prior to FY 2024.
Comment: A commenter requested that CMS confirm that section 1886(d)(8)(C)(ii) has no effect on aggregate expenditures or the Reclassification Budget Neutrality Adjustment (RBNA). The commenter also referenced the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58976 ) with regard to the calculation of the rural wage index and requested that CMS confirm that when a state's rural wage index is determined under Calculations 2 or 3, the increase in aggregate expenditures is measured by reference to what the rural wage index for the state would have been under Calculation 1. Finally, the commenter also requested that CMS confirm that section 1886(d)(8)(C)(iii) has no effect on aggregate expenditures or the RBNA for geographically rural or § 412.103 hospitals that have a LUGAR or MGCRB reclassification.
Response: It appears that the commenter believes that calculation 1 should be used for the pre reclassified wage index. As previously mentioned, the “before” wage index value uses a hospitals area wage data before any reclassifications or state rural wage index is applied (the pre reclassified wage index). The “after” wage index for a rural area would be based on the greater of the three rural wage index calculations as discussed in the FY 2024 IPPS/LTCH final rule. Accordingly, there could be an impact on the budget neutrality factor due to sections 1886(d)(8)(C)(ii) and (iii) of the Act, and the “before” wage index uses the pre reclassified wage index and not Calculation 1.
Under § 412.64(e)(4), we make an adjustment to the wage index to ensure that aggregate payments after implementation of the rural floor under section 4410 of the BBA ( Pub. L. 105-33 ) are equal to the aggregate prospective payments that would have been made in the absence of this provision. Consistent with section 3141 of the Affordable Care Act and as discussed in section III.G. of the preamble of this final rule and codified at § 412.64(e)(4)(ii), the budget neutrality adjustment for the rural floor is a national adjustment to the wage index.
For FY 2025 there is one hospital in Puerto Rico with wage data. Therefore, for this final rule, we do not need to apply the calculation discussed in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50369 through 50370 ). In a future fiscal year, if there were no hospitals with wage data in rural Puerto Rico, we would then calculate a national rural Puerto Rico wage index based on the policy adopted in the FY 2008 IPPS final rule with comment period ( 72 FR 47323 ). That is, we would use the unweighted average of the wage indexes from all CBSAs (urban areas) that are contiguous to (share a border with) the rural counties to compute the rural floor ( 72 FR 47323 ; 76 FR 51594 ).
We note, in the FY 2024 IPPS/LTCH final rule ( 88 FR 58971-77 ), we finalized a policy beginning with FY 2024 to include hospitals with § 412.103 reclassification along with geographically rural hospitals in all rural wage index calculations and are only excluding “dual reclass” hospitals (hospitals with simultaneous § 412.103 and MGCRB reclassifications) in accordance with the hold harmless provision at section 1886(d)(8)(C)(ii) of the Act. Consistent with the previous policy, beginning with FY 2024, we include the data of all § 412.103 hospitals (including those that have an MGCRB reclassification) in the calculation of the rural floor.
To calculate the national rural floor budget neutrality adjustment factor, we used FY 2023 discharge data to simulate payments, the new OMB labor market area delineations adopted for FY 2025, and the post-reclassified national wage indexes and compared the following:
- National simulated payments without the rural floor.
- National simulated payments with the rural floor.
Based on this comparison, we determined a national rural floor budget neutrality adjustment factor. The national adjustment was applied to the national wage indexes to produce rural floor budget neutral wage indexes. Please see the table later in this section for a summary of the FY 2025 budget neutrality factors.
As further discussed in section III.G.2. of this final rule, we note that section 9831 of the American Rescue Plan Act of 2021 ( Pub. L. 117-2 ), enacted on March 11, 2021 amended section 1886(d)(3)(E)(i) of the Act ( 42 U.S.C. 1395ww(d)(3)(E)(i) ) and added section 1886(d)(3)(E)(iv) of the Act to establish a minimum area wage index (or imputed floor) for hospitals in all-urban States for discharges occurring on or after October 1, 2022. Unlike the imputed floor that was in effect from FY 2005 through FY 2018, section 1886(d)(3)(E)(iv)(III) of the Act provides that the imputed floor wage index shall not be applied in a budget neutral manner. Specifically, section 9831(b) of Public Law 117-2 amends section 1886(d)(3)(E)(i) of the Act to exclude the imputed floor from the budget neutrality requirement under section 1886(d)(3)(E)(i) of the Act. In the past, we budget neutralized the estimated increase in payments each year resulting from the imputed floor that was in effect from FY 2005 through FY 2018. For FY 2022 and subsequent years, in applying the imputed floor required under section 1886(d)(3)(E)(iv) of the Act, we are applying the imputed floor after the application of the rural floor and would apply no reductions to the standardized amount or to the wage index to fund the increase in payments to hospitals in all-urban States resulting from the application of the imputed floor. We ( print page 69947) refer the reader to section III.G.2. of the preamble of this final rule for a complete discussion regarding the imputed floor.
Comment: A commenter requested that CMS fully describe the interplay between the Reclassification Budget Neutrality Factor and the Rural Floor Budget Neutrality Factor and make available the calculations of both budget neutrality adjustments. The commenter stated that it is unclear how the Reclassification Budget Neutrality Factor is applied or potentially replaced with the Rural Floor Budget Neutrality Factor for a provider who receives both adjustments.
Response: With regard to the commenter requesting a description of the interplay between the Reclassification Budget Neutrality Factor and the Rural Floor Budget Neutrality Factor and a calculation of both adjustments, we refer the commenter to sections II.A.4.d. and II.A.4.e. of the Addendum of this final rule for a complete discussion of the budget neutrality impacts of reclassified hospitals and the rural floor. We also refer the commenter to the table in the Addendum summarizing the FY 2025 budget neutrality factors. Regarding the interplay of both adjustments and the impact on a hospital that receives both, we remind the commenter that the reclassification budget neutrality adjustment is applied to the standardized amount while the rural floor budget neutrality factor is applied to the wage index.
As discussed in section III.G.5. of the preamble of this final rule, we are continuing for FY 2025 the wage index policy finalized in the FY 2020 IPPS/LTCH PPS final rule to address wage index disparities by increasing the wage index values for hospitals with a wage index value below the 25th percentile wage index value across all hospitals (the low wage index hospital policy). As discussed in section III.G.3. of this final rule, consistent with our current methodology for implementing wage index budget neutrality under section 1886(d)(3)(E) of the Act, we are making a budget neutrality adjustment to the national standardized amount for all hospitals so that the increase in the wage index for hospitals with a wage index below the 25th percentile wage index, is implemented in a budget neutral manner.
We note that the FY 2020 low wage index hospital policy and the related budget neutrality adjustment are the subject of pending litigation in multiple courts. On July 23, 2024, the Court of Appeals for the D.C. Circuit held that the Secretary lacked authority under 1886(d)(3)(E) or 1886(d)(5)(I)(i) of the Act to adopt the low wage index hospital policy for FY 2020, and that the policy and related budget neutrality adjustment must be vacated. Bridgeport Hosp. v. Becerra, Nos. 22-5249, 22-5269, 2024 WL 3504407, at *7-*8 n.6 (D.C. Cir. July 23, 2024). As of the date of this Rule's publication, the time to seek further review of the D.C. Circuit's decision in Bridgeport Hospital has not expired. See Fed. R. App. P. 40(a)(1). The government is evaluating the decision and considering options for next steps.
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2025 labor-related share percentage, the FY 2025 relative weights, and the FY 2025 wage index for each hospital before adjusting the wage indexes under the low wage index hospital policy, and applied the proxy FY 2025 hospital readmissions payment adjustments and the proxy FY 2025 hospital VBP payment adjustments; and
- Aggregate payments using the new OMB labor market area delineations for FY 2025, the FY 2025 labor-related share percentage, the FY 2025 relative weights, and the FY 2025 wage index for each hospital after adjusting the wage indexes under the low wage index hospital policy, and applied the same proxy FY 2025 hospital readmissions payment adjustments and the proxy FY 2025 hospital VBP payment adjustments applied previously.
This final FY 2025 budget neutrality adjustment factor was applied to the standardized amount.
As noted previously, in section III.G.6. of the preamble to this final rule, in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49018 through 49021 ) we finalized a policy to apply a 5-percent cap on any decrease to a hospital's wage index from its wage index in the prior FY, regardless of the circumstances causing the decline. That is, a hospital's wage index would not be less than 95 percent of its final wage index for the prior FY. We also finalized the application of this permanent cap policy in a budget neutral manner through an adjustment to the standardized amount to ensure that estimated aggregate payments under our wage index cap policy for hospitals that will have a decrease in their wage indexes for the upcoming fiscal year of more than 5 percent will equal what estimated aggregate payments would have been without the permanent cap policy.
To calculate a wage index cap budget neutrality adjustment factor for FY 2025, we used FY 2023 discharge data to simulate payments and compared the following:
- Aggregate payments without the 5-percent cap using the FY 2025 labor-related share percentages, the new OMB labor market area delineations for FY 2025, the FY 2025 relative weights, the FY 2025 wage index for each hospital after adjusting the wage indexes under the low wage index hospital policy, and applied the proxy FY 2025 hospital readmissions payment adjustments and the proxy FY 2025 hospital VBP payment adjustments.
- Aggregate payments with the 5-percent cap using the FY 2025 labor-related share percentages, the new OMB labor market area delineations for FY 2025, the FY 2025 relative weights, the FY 2025 wage index for each hospital after adjusting the wage indexes under the low wage index hospital policy, and applied the same proxy FY 2025 hospital readmissions payment adjustments and the proxy FY 2025 hospital VBP payment adjustments applied previously.
We note, Table 2 associated with this final rule contains the wage index by provider before and after applying the low wage index hospital policy and the cap.
In section V.N. of the preamble of this final rule, we discuss the Rural Community Hospital (RCH) Demonstration program, which was originally authorized for a 5-year period by section 410A of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) ( Pub. L. 108-173 ), and extended for another 5-year period by sections 3123 and 10313 of the Affordable Care Act ( Pub. L. 111-148 ). Subsequently, section 15003 of the 21st Century Cures Act ( Pub. L. 114-255 ), enacted December 13, 2016, amended section 410A of Public Law 108-173 to require a 10-year extension period (in place of the 5-year extension required by the Affordable Care Act, as further discussed later in this section). Finally, Division CC, section 128(a) of the Consolidated Appropriations Act of 2021 ( Pub. L. 116-260 ) again amended section 410A to require a 15-year extension period in place of the 10-year period. We make an adjustment to the ( print page 69948) standardized amount to ensure the effects of the RCH Demonstration program are budget neutral as required under section 410A(c)(2) of Public Law 108-173 . We refer readers to section V.N. of the preamble of this final rule for complete details regarding the Rural Community Hospital Demonstration.
With regard to budget neutrality, as mentioned earlier, we make an adjustment to the standardized amount to ensure the effects of the Rural Community Hospital Demonstration are budget neutral, as required under section 410A(c)(2) of Public Law 108-173 . For FY 2025, based on the latest data for this final rule, the total amount that we are applying to make an adjustment to the standardized amounts to ensure the effects of the Rural Community Hospital Demonstration program are budget neutral is $19,414,819. Accordingly, using the most recent data available to account for the estimated costs of the demonstration program, for FY 2025, we computed a factor for the Rural Community Hospital Demonstration budget neutrality adjustment that would be applied to the standardized amount. Please see the table later in this section for a summary of the Proposed FY 2025 budget neutrality factors. We refer readers to section V.N. of the preamble of this final rule on complete details regarding the calculation of the amount we are applying to make an adjustment to the standardized amounts.
The following table is a summary of the FY 2025 budget neutrality factors, as discussed in the previous sections.

Section 1886(d)(5)(A) of the Act provides for payments in addition to the basic prospective payments for “outlier” cases involving extraordinarily high costs. To qualify for outlier payments, a case must have costs greater than the sum of the prospective payment rate for the MS-DRG, any IME and DSH payments, uncompensated care payments, supplemental payment for eligible IHS/Tribal hospitals and Puerto Rico hospitals, any new technology add-on payments, and the “outlier threshold” or “fixed-loss” amount (a dollar amount by which the costs of a case must exceed payments in order to qualify for an outlier payment). We refer to the sum of the prospective payment rate for the MS-DRG, any IME and DSH payments, uncompensated care payments, supplemental payment for eligible IHS/Tribal hospitals and Puerto Rico hospitals, any new technology add-on payments, and the outlier threshold as the outlier “fixed-loss cost threshold.” To determine whether the costs of a case exceed the fixed-loss cost threshold, a hospital's CCR is applied to the total covered charges for the case to convert the charges to estimated costs. Payments for eligible cases are then made based on a marginal cost factor, which is a percentage of the estimated costs above the fixed-loss cost threshold. The marginal cost factor for FY 2025 is 80 percent, or 90 percent for burn MS-DRGs 927, 928, 929, 933, 934 and 935. We have used a marginal cost factor of 90 percent since FY 1989 ( 54 FR 36479 through 36480 ) for designated burn DRGs as well as a marginal cost factor of 80 percent for all other DRGs since FY 1995 ( 59 FR 45367 ).
In accordance with section 1886(d)(5)(A)(iv) of the Act, outlier payments for any year are projected to be not less than 5 percent nor more than 6 percent of total operating DRG payments (which does not include IME and DSH payments) plus outlier payments. When setting the outlier threshold, we compute the projected percentage by dividing the total projected operating outlier payments by the total projected operating DRG payments plus projected operating outlier payments. As discussed in the next section, for FY 2025, we are incorporating an estimate of the impact of outlier reconciliation when setting the outlier threshold. We do not include any other payments such as IME and DSH within the outlier target amount. Therefore, it is not necessary to include Medicare Advantage IME payments in the outlier threshold calculation. Section 1886(d)(3)(B) of the Act requires the Secretary to reduce the average standardized amount by a factor to account for the estimated total of outlier payments as a proportion of total DRG payments. More information on outlier payments may be found on the CMS website at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/outlier.html .
The regulations in 42 CFR 412.84(i)(4) state that any outlier reconciliation at cost report settlement will be based on operating and capital cost-to-charge ratios (CCRs) calculated based on a ratio of costs to charges computed from the relevant cost report and charge data determined at the time the cost report coinciding with the discharge is settled. Instructions for outlier reconciliation are in section 20.1.2.5 of chapter 3 of the Claims Processing Manual (on line at https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/clm104c03.pdf ). The original instructions issued in July 2003 [ 1106 ] instruct MACs to identify for CMS any instances where: (1) a ( print page 69949) hospital's actual operating CCR for the cost reporting period fluctuates plus or minus 10 percentage points or more compared to the interim operating CCR used to calculate outlier payments when a bill is processed; and (2) the total operating and capital outlier payments for the hospital exceeded $500,000 for that cost reporting period. Cost reports that meet these criteria will have the hospital's outlier payments reconciled at the time of cost report final settlement if approved by the CMS Central Office. For the remainder of this discussion, we refer to these criteria as the original criteria for outlier reconciliation (or the original criteria).
On March 28, 2024, we issued Change Request (CR) 13566, which is available at https://www.cms.gov/medicare/regulations-guidance/transmittals/2024-transmittals/r12558cp . CR 13566 provides additional instructions to MACs that expand the criteria for identifying cost reports MACs are to refer to CMS for approval of outlier reconciliation. We anticipate that MACs will identify more cost reports to refer to CMS for outlier reconciliation approval. A report issued by the Office of the Inspector General (OIG) recommended that CMS require reconciliation of all hospital outlier payments during a cost-reporting period in its November 2019 report titled “Hospitals Received Millions in Excessive Outlier Payments Because CMS Limits the Reconciliation Process” (A-05-16-00060). [ 1107 ] CMS concurs with the OIG's recommendation and is exploring the administrative feasibility of reconciling the outlier payments for all hospitals.
Consistent with the OIG recommendation, CMS modified the original criteria for identifying cost reports to refer to CMS for outlier reconciliation approval in instructions to MACs in CR 13566. Specifically, CR 13566 states that for cost reports beginning on or after October 1, 2024, MACs shall identify for CMS any instances where: (1) the actual operating CCR is found to be plus or minus 20 percent or more from the operating CCR used during that time period to make outlier payments, and (2) the total operating and capital outlier payments for the hospital exceeded $500,000 for that cost reporting period. For the remainder of this discussion, we refer to these criteria as the new criteria for outlier reconciliation (or the new criteria). In the proposed rule we stated that we believe the new criteria balance current administrative feasibility with the goal of expanding the scope of cost reports identified for outlier reconciliation approval and conducting outlier reconciliation more frequently to increase the accuracy of outlier payments. These new criteria for identifying hospital cost reports that MACs should identify for outlier reconciliation approval are in addition to the original criteria for reconciliation described previously. That is, under the new criteria, MACs identify hospitals for outlier reconciliation that would not have met the original criteria. For example, in an instance where a hospital was paid with an operating CCR of 0.09 and its actual operating CCR was 0.07, then the hospital would not have met the 10-percentage point criterion under the original criteria (the hospital's operating CCR would have to be a negative number, which is not possible). Under the new criteria, a hospital that had a change in their actual operating CCR that was greater than 20 percent from the CCR used for payment during the cost reporting period would be referred to CMS. Using the same example, while the operating CCR changed by a difference of −0.02 percentage point (0.07−0.09), the percentage change operating CCR is −22.2 percent ((0.07/0.09)−1), which meets the new 20 percent criterion. In addition, CR 13566 instructs that for cost reporting periods that begin on or after October 1, 2024, a hospital in its first cost reporting period will be referred for reconciliation of outlier payments at the time of cost report final settlement. As such, new hospitals will be referred for outlier reconciliation regardless of the change to the operating CCR and no matter the amount of outlier payments during the cost reporting period.
If we determine that a hospital's outlier payments should be reconciled, we reconcile both operating and capital outlier payments. We refer readers to section 20.1.2.5 of Chapter 3 of the Medicare Claims Processing Manual for complete instructions regarding outlier reconciliation, including the update to the outlier reconciliation criteria provided in CR 13566.
Comment: Commenters were concerned that CMS has added new criteria for determining which hospitals will have their outlier payments reconciled in CR 13566. The commenters stated CMS has not explained the grounds for the new criteria or its retention of the old criteria, and the new criteria were adopted without notice and comment rulemaking. The commenters stated their belief that new reconciliation criteria constitute a substantive change to CMS' payment policy that cannot be adopted without notice and comment rulemaking. The commenters urged CMS to withdraw the CR.
MedPAC supported changes in CR 13566 and agreed with CMS that expanding the criteria for identifying hospitals for outlier reconciliation approval and increasing the frequency reconciliation would increase the accuracy of outlier payments while maintaining relatively low administrative burden. MedPAC also encouraged CMS to continue to monitor outlier payments and administrative burden to inform if additional changes to referral criteria are warranted in future years.
Response: CMS established the outlier reconciliation regulation under § 412.84(i)(4) effective for discharges on or after August 8, 2003 which makes all hospital outlier payments subject to reconciliation. CMS has not modified the outlier regulation. The instructions CMS has issued via CR 13566 have set forth an enforcement policy that determines when MACs will identify additional hospitals for reconciliation referral. They do not change the legal standards that govern the hospitals.
We appreciate MedPAC's supporting comment. As we explained in the proposed rule, we believe the new criteria balance current administrative feasibility with the goal of expanding the scope of cost reports identified for outlier reconciliation approval to increase the accuracy of outlier payments. These new criteria for identifying hospital cost reports that MACs should be referred for outlier reconciliation approval are in addition to the original criteria for reconciliation described previously.
The regulations at § 412.84(m) further state that at the time of any outlier reconciliation under § 412.84(i)(4), outlier payments may be adjusted to account for the time value of any underpayments or overpayments. Section 20.1.2.6 of Chapter 3 of the Medicare Claims Processing Manual contains instructions on how to assess the time value of money for reconciled outlier amounts.
If the operating CCR of a hospital approved for outlier reconciliation is lower at cost report settlement compared to the operating CCR used for payment, the hospital would owe CMS money. Conversely, if the operating CCR increases at cost report settlement compared to the operating CCR used for payment, CMS would owe the hospital money.
In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42623 through 42635 ), we ( print page 69950) finalized a methodology to incorporate outlier reconciliation in the FY 2020 outlier fixed loss cost threshold. As discussed in the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19592 ), we stated that rather than trying to predict which claims and/or hospitals may be subject to outlier reconciliation, we believe a methodology that incorporates an estimate of outlier reconciliation dollars based on actual outlier reconciliation amounts reported in historical cost reports would be a more feasible approach and provide a better estimate and predictor of outlier reconciliation for the upcoming fiscal year. We also stated that we believe the methodology addresses stakeholders' concerns about the impact of outlier reconciliation on the modeling of the outlier threshold. For a detailed discussion of additional background regarding the incorporation of outlier reconciliation into the outlier fixed loss cost threshold, we refer the reader to the FY 2020 IPPS/LTCH PPS final rule. Consistent with the instructions to MACs that added new criteria that identify additional cost reports for reconciliation referral beginning with FY 2025 cost reports, we proposed changes to our methodology to reflect the estimated reconciled outlier payments of the additional hospital cost reports identified under the new criteria. Specifically, we proposed to make modifications to the steps of our methodology in section II.A.4.i.1.a. of this Addendum to reflect the estimated reconciled outlier payments under the new criteria in the projection of outlier reconciliations for the FY 2025 outlier fixed loss cost threshold.
Based on the methodology finalized in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42623 through 42625 ), for FY 2025, we proposed to continue to incorporate outlier reconciliation in the FY 2025 outlier fixed loss cost threshold, with modifications to reflect the expansion of outlier reconciliations under the new criteria in CR 13566 (described previously).
As discussed in the FY 2020 IPPS/LTCH PPS final rule, for FY 2020, we used the historical outlier reconciliation amounts from the FY 2014 cost reports (cost reports with a begin date on or after October 1, 2013, and on or before September 30, 2014), which we believed would provide the most recent and complete available data to project the estimate of outlier reconciliation. We refer the reader to the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42623 through 42625 ) for a discussion on the use of the FY 2014 cost report data for purposes of projecting outlier reconciliations for the FY 2020 outlier threshold calculation. For FY 2024, we applied the same methodology finalized in FY 2020, using the historical outlier reconciliation amounts from the FY 2018 cost reports (cost reports with a begin date on or after October 1, 2017, and on or before September 30, 2018).
Similar to the FY 2024 methodology, we proposed to determine a projection of outlier reconciliations for the FY 2025 outlier threshold calculation by advancing the historical data used by 1 year. Specifically, we proposed to use FY 2019 cost reports (cost reports with a begin date on or after October 1, 2018, and on or before September 30, 2019). For FY 2025, we proposed to use the methodology from FY 2020 to incorporate a projection of operating outlier reconciliations for the FY 2025 outlier threshold calculation, modified to reflect additional cost reports that would be identified for reconciliation under the new criteria in CR 13566. Because the new criteria are not effective until FY 2025 cost reports, to estimate outlier reconciliation dollars under the new criteria, we proposed to apply the new criteria to FY 2019 cost reports as if they had been in place at the time of final cost report settlement (as described in more detail later in this section).
As described previously, under the expanded outlier reconciliation criteria in CR 13566, for cost reporting periods beginning on or after October 1, 2024, new hospitals will have their outlier payments referred for outlier reconciliation by the MAC to CMS in their first cost reporting period regardless of the change to the operating CCR or the amount of outlier payments during the cost reporting period. For purposes of the methodology for incorporating a projection of operating outlier reconciliations for the FY 2025 outlier threshold calculation to reflect additional cost reports that would be identified for reconciliation under the criteria added by CR 13566, we did not propose to include the first cost reporting periods of new hospitals because the lack of predictability of new hospitals' data may impact the reliability of our projection. We noted in the proposed rule that we expect the proposed modifications to our methodology for incorporating a projection of operating outlier reconciliations into the outlier threshold calculation would be necessary for 6 years, at which point the additional FY 2025 cost reports with outlier payments reconciled under the new criteria will be reflected in the HCRIS data available to be used to set the threshold.
For FY 2019 hospital cost reports that were reconciled using the original criteria for referral for outlier reconciliation, in the FY 2025 proposed rule, we used the December 2023 HCRIS extract of the cost report data to calculate the proposed percentage adjustment for outlier reconciliation. For the FY 2025 final rule, we proposed to use the latest quarterly HCRIS extract that is publicly available at the time of the development of that rule which, for FY 2025, would be the March 2024 extract. As discussed in the FY 2024 IPPS/LTCH final rule ( 88 FR 59346 ), we stated that we generally expect historical cost reports for the applicable fiscal year to be available by March, and we have worked with our MACs so that historical cost reports for the applicable fiscal year can be made available with the March HCRIS update for the final rule.
To account for the additional hospital cost reports that would be reconciled as a result of the new criteria, we proposed to use data from the Provider Specific File (PSF) and the cost report to identify the FY 2019 cost reports that would have met the new criteria if those criteria had been in effect. This is because the FY 2019 cost reports in HCRIS would not have been identified as meeting the new criteria for outlier reconciliation since those new criteria are not being used until cost reports beginning with FY 2025. As such, these FY 2019 cost reports do not have an amount reported for operating or capital outlier reconciliation dollars. Therefore, we proposed to modify our methodology to estimate the outlier reconciliation dollars based on the operating and capital outlier amounts reported on the FY 2019 cost reports and supplemental data collected from the MACs, as described further in this section.
The following proposed steps are similar to those finalized in the FY 2020 final rule, with updated data for FY 2025 and additional steps to reflect the cost reports that would be identified with new criteria under the updated instructions:
Step 1. —Identify hospital cost reports that meet the original criteria or the new criteria.
Step 1a. —Identify hospitals that report on their cost report the operating outlier reconciliation dollars on Worksheet E, Part A, Line 2.01. We note, these were hospitals that were identified by the MACs that met the original criteria for outlier reconciliation and were approved by CMS for outlier reconciliation. We use the Federal FY ( print page 69951) 2019 cost reports for hospitals paid under the IPPS from the most recent publicly available quarterly HCRIS extract available at the time of development of the proposed and final rules and exclude sole community hospitals (SCHs) that were paid under their hospital-specific rate (that is, if Worksheet E, Part A, Line 48 is greater than Line 47). We note that when there are multiple columns available for the lines of the cost report described in the following steps and the provider was paid under the IPPS for that period(s) of the cost report, then we believe it is appropriate to use multiple columns to fully represent the relevant IPPS payment amounts, consistent with our methodology for the FY 2020 final rule.
Step 1b. —For hospitals that were not included in Step 1a, to identify hospitals that would be referred for outlier reconciliation under the new criteria, we proposed to use data from the latest PSF and cost report data from the most recent publicly available quarterly HCRIS extract. We identified hospitals with cost reports where the actual operating CCR for the cost reporting period fluctuates plus or minus 20 percent or more compared to the interim operating CCR used to calculate outlier payments when a bill is processed. To do this, we compared the operating CCR calculated from the FY 2019 cost report in the most recent publicly available quarterly HCRIS extract (the December 2023 HCRIS for the proposed rule) to the weighted operating CCR used for claim payment during the FY 2019 cost reporting period from the latest quarterly PSF update (December 2023 for the proposed rule). We then determined whether the hospital had total operating and capital outlier payments greater than $500,000 during the FY 2019 cost reporting period based on the most recent publicly available quarterly HCRIS (the December 2023 HCRIS for the proposed rule). If the hospital met both of these criteria, we included the operating outlier payments from the MAC using CCRs from the FY 2019 cost report (as described in Step 2b-2). For the final rule, to identify hospitals that would be referred for reconciliation, we proposed to use the most recent HCRIS and PSF data available, which would be the March 2024 update. We note that for this purpose we assumed that all hospitals that would be referred for outlier reconciliation under the new criteria would have their outlier payments reconciled.
Step 2. —Determine the aggregate amount of operating outlier reconciliation dollars (under both the original criteria and the new criteria).
Step 2a. —Calculate the aggregate amount of historical total of operating outlier reconciliation dollars (Worksheet E, Part A, Line 2.01) using the Federal FY 2019 cost reports from Step 1a.
Step 2b. —For the hospitals that would have met the new criteria as identified in Step 1b, to determine the aggregate amount of operating outlier reconciliation dollars, we proposed to use the following process:
We collected supplemental estimated outlier payment data from the MACs for claims with discharges occurring during the hospital's FY 2019 cost reporting period to estimate the change in the hospital's outlier payments. Specifically, for each hospital identified in Step 1b, the MACs used the actual operating CCR calculated from the FY 2019 cost report and the utility in the claims system along with that CCR to determine total outlier payments for claims with discharges occurring during the hospital's FY 2019 cost report (this is the same process MACs would have used if the cost report had been identified for reconciliation had the new criteria been in place for FY 2019 cost reports). For those same claims with discharges occurring during the hospital's 2019 cost report, the MAC provided to CMS the outlier payment as reported on the claim (which was based on the hospital's CCR in the PSF at the time of claim payment).
Using this supplemental estimated outlier payment data, we computed a ratio of the outlier payments based on the actual operating CCR for the FY 2019 cost reporting period and the CCR used at the time of claim payment. This ratio is then applied to the operating outlier payment reported on the FY 2019 cost report to impute an operating outlier payment for the FY 2019 cost report. In the proposed rule we stated that we believe it is appropriate to impute the operating outlier payment for the cost report using the supplemental data from the MACs described previously rather than use the actual amount reported on the cost report because the claims data in the claims processing system may slightly differ from the cost report data in the HCRIS due to timing. This approach would also allow CMS to use more recent data (from the most recent publicly available quarterly HCRIS extract, which was from December 2023 for the proposed rule) to estimate outlier reconciliation dollars as compared to estimating outlier reconciliation dollars using the supplemental outlier payment data from the MACs, which was submitted by the MACs to CMS beginning in November 2022 (as described in this section). This is also the same data used to determine the aggregate amount of operating outlier reconciliation dollars for hospitals from the FY 2019 cost report data using the December 2023 HCRIS extract in Step 2a.
As presented in the table that follows, to calculate the imputed operating outlier payment for the FY 2019 cost report, we multiplied the operating outlier payment reported on the FY 2019 cost report by the following ratio (determined from the supplemental data collected from the MACs described previously): Operating Outlier Payments from MAC using the CCR from FY 2019 Cost Report divided by Operating Outlier Payments from MAC Based on Claim Payment. The general formula is the following: Operating Outlier Payments Reported on the Cost Report* (Operating Outlier Payments from MAC Using CCRs from FY 2019 Cost Report/Operating Outlier Payments from MAC Based on Claim Payment).
To calculate the Estimated Operating Outlier Reconciliation Dollars, we then subtracted the Imputed Operating Outlier Amount for the FY 2019 Cost Report (Step 2b-5) from the Operating Outlier Payment Reported on the FY 2019 Cost Report (Step 2b-1).
The following is an example to illustrate our proposed calculation to determine the estimated amount of operating outlier reconciliation dollars for the hospitals that would have met the new criteria:

We noted the following, with regard to the data used in the calculation:
- Due to system limitations the MACs needed 13 months to process all providers' claims through the claims utility (for Steps 2b-2 and 2b-3). The MACs used the operating and capital CCR from the FY 2019 cost reports based on the September 2022 HCRIS extract and began processing the supplemental data for FY 2019 outlier payments in November 2022. We proposed to move this forward each year, using the September HCRIS for future fiscal years for the CCRs (for example, for FY 2026, MACs would use CCRs from the FY 2020 cost reports based on the September 2023 HCRIS).
- For FY 2025, for the “Operating Outlier Payment Reported on the FY 2019 Cost Report” (Step 2b-1) we used operating outlier payments reported on Worksheet E, Part A, Lines 2.02, 2.03, and 2.04 from the FY 2019 cost report using the most recent publicly available quarterly HCRIS extract for the proposed rule (that is, the December 2023 HCRIS extract). We proposed to move this forward each year and use the most recent publicly available quarterly HCRIS extract (for example, for FY 2026, we would use operating outlier payments reported on Worksheet E, Part A, Lines 2.02, 2.03, and 2.04 from the FY 2020 cost reports using the most recent publicly available quarterly HCRIS extract).
- For the hospitals identified in Step 1b, for the proposed rule we posted a public use file that included the operating CCR calculated from the FY 2019 cost report in the most recent publicly available quarterly HCRIS extract (the December 2023 HCRIS for the proposed rule), the weighted operating CCR used for claim payment during the FY 2019 cost reporting period from the latest quarterly PSF update (December 2023 for the proposed rule), supplemental data from the MACs and operating outlier payment reported on the FY 2019 cost report.
Step 3. —Calculate the aggregate amount of total Federal operating payments across all applicable hospitals using the Federal FY 2019 cost reports. The total Federal operating payments consist of the Federal payments (Worksheet E, Part A, Line 1.01 and Line 1.02, plus Line 1.03 and Line 1.04), outlier payments (Worksheet E, Part A, Lines 2.02, 2.03, and 2.04), and the outlier reconciliation amounts from Steps 2a and 2b. We noted that a negative amount on Worksheet E, Part A, Line 2.01 from Step 2a for outlier reconciliation indicates an amount that was owed by the hospital, and a positive amount indicates this amount was paid to the hospital. Similarly, a negative amount from Step 2b for outlier reconciliation indicates an amount that would have been owed by the hospital, and a positive amount indicates an amount that would have been paid to the hospital.
Step 4. —Divide the aggregate amount from Step 2 (that is, the sum of the amounts from Steps 2a and 2b) by the amount from Step 3 and multiply the resulting amount by 100 to produce the percentage of total operating outlier reconciliation dollars to total Federal operating payments for FY 2019. For FY 2025, the proposed ratio was a negative 0.03979 percent ((−$34,513,755/$86,740,955,496) × 100), which, when rounded to the second digit, is −0.04 percent. We stated that this percentage amount would be used to adjust the outlier target for FY 2025 as described in Step 5.
Step 5. —Because the outlier reconciliation dollars are only available on the cost reports, and not in the Medicare claims data in the MedPAR file used to model the outlier threshold, we proposed to target 5.1 percent minus the percentage determined in Step 4 in determining the outlier threshold. Using the FY 2019 cost reports, because the aggregate outlier reconciliation dollars from Step 2 are negative, we are targeted an amount higher than 5.1 percent for outlier payments for FY 2025 under our proposed methodology. Therefore, for FY 2025, we proposed to incorporate a projection of outlier reconciliation dollars by targeting an outlier threshold at 5.14 percent [5.1 percent−(−0.04 percent)].
In the proposed rule we stated that when the percentage of operating outlier reconciliation dollars to total Federal operating payments rounds to a negative value (that is, when the aggregate amount of outlier reconciliation as a percent of total operating payments rounds to a negative percent), the effect is a decrease to the outlier threshold compared to an outlier threshold that is calculated without including this estimate of operating outlier reconciliation dollars.
As explained in the FY 2020 IPPS/LTCH PPS proposed rule ( 84 FR 19593 ), we would continue to use a 5.1 percent target (or an outlier offset factor of 0.949) in calculating the outlier offset to the standardized amount. Therefore, the proposed operating outlier offset to the standardized amount was 0.949 (1−0.051). In section II.A.4.i.(2). of this Addendum, we provided the FY 2025 outlier threshold as calculated for the proposed rule both with and without including this proposed percentage estimate of operating outlier reconciliation.
We invited public comment on our proposed methodology for projecting an estimate of outlier reconciliation and incorporating that estimate into the modeling for the fixed-loss cost outlier threshold for FY 2025.
We did not receive any comments with regard to the steps described previously and we are finalizing as proposed without modification the methodology to project an estimate of outlier reconciliation and incorporating that estimate into the modeling for the fixed-loss cost outlier threshold for FY 2025. As we proposed and where stated in the previous steps, we are finalizing to use the most recent HCRIS and PSF data available for this final rule, which is the March 2024 update. Also, for the hospitals identified in Step 1b, similar to the proposed rule, for this final rule, we have posted a public use file that includes the operating CCR calculated from the FY 2019 cost report in the most recent publicly available quarterly HCRIS extract (the March 2023 HCRIS for this final rule), the weighted operating CCR used for claim payment during the FY 2019 cost reporting period from the latest quarterly PSF update (March 2023 for this final rule), supplemental data from the MACs and operating outlier payment reported on the FY 2019 cost report. ( print page 69953)
With regard to step 4, the final ratio is a negative 0.041994 percent ((−$36,439,127/$86,772,005,692) × 100), which, when rounded to the second digit, is −0.04 percent. This percentage amount is used to adjust the outlier target for FY 2025 as described in Step 5. Based on step 5, for FY 2025, we are incorporating a projection of outlier reconciliation dollars by targeting an outlier threshold at 5.14 percent [5.1 percent−(−0.04 percent)]. In section II.A.4.i.(2). of this Addendum, we provide the FY 2025 outlier threshold as calculated for this final rule both with and without including this percentage estimate of operating outlier reconciliation.
We establish an outlier threshold that is applicable to both hospital inpatient operating costs and hospital inpatient capital related costs ( 58 FR 46348 ). Similar to the calculation of the adjustment to the standardized amount to account for the projected proportion of operating payments paid as outlier payments, as discussed in greater detail in section III.A.2. of this Addendum, we proposed to reduce the FY 2025 capital standard Federal rate by an adjustment factor to account for the projected proportion of capital IPPS payments paid as outliers. The regulations in 42 CFR 412.84(i)(4) state that any outlier reconciliation at cost report settlement would be based on operating and capital CCRs calculated based on a ratio of costs to charges computed from the relevant cost report and charge data determined at the time the cost report coinciding with the discharge is settled. As such, any reconciliation also applies to capital outlier payments.
For FY 2025, we proposed to continue to use the methodology from FY 2020 to adjust the FY 2025 capital standard Federal rate by an adjustment factor to account for the projected proportion of capital IPPS payments paid as outliers, with modifications to reflect the expansion of outlier reconciliations under the new criteria in CR 13566 (described previously).
For purposes of the methodology for incorporating a projection of capital outlier reconciliations for the FY 2025 outlier adjustment to the capital standard Federal rate to reflect additional cost reports that would be identified for reconciliation under the criteria added by CR 13566, as we discussed in section II.A.4.i.1.a. of the Addendum of this final rule regarding the projection of the operating outlier reconciliation, we did not propose to include the first cost reporting periods of new hospitals because the lack of predictability of new hospitals' data may impact the reliability of our projection. As noted, we expect the proposed modifications to our methodology for incorporating a projection of capital outlier reconciliations into the outlier adjustment to the capital standard federal rate would be necessary for 6 years, at which point the additional FY 2025 cost reports with outlier payments reconciled under the new criteria will be reflected in the HCRIS data available to be used to determine this adjustment.
For FY 2019 hospital cost reports that were reconciled using the original criteria for referral for outlier reconciliation, for the FY 2025 proposed rule, we used the December 2023 HCRIS extract of the cost report data to calculate the proposed percentage adjustment for outlier reconciliation. For the FY 2025 final rule, we proposed to use the latest quarterly HCRIS extract that is publicly available at the time of the development of that rule which, for FY 2025, would be the March 2024 extract. As discussed in the FY 2024 IPPS/LTCH final rule ( 88 FR 59347 ), we generally expect historical cost reports for the applicable fiscal year to be available by March, and we have worked with our MACs so that historical cost reports for the applicable fiscal year can be made available with the March HCRIS update for the final rule.
To account for the additional hospital cost reports that would be reconciled as a result of the new criteria, we proposed to use data from the PSF and the cost report to identify the FY 2019 cost reports that would have met the new criteria if those criteria had been in effect. This is because the FY 2019 cost reports in HCRIS would not have been identified as meeting the new criteria for outlier reconciliation since those new criteria are not being used until cost reports beginning with FY 2025. As such, these FY 2019 cost reports do not have an amount reported for operating or capital outlier reconciliation dollars. Therefore, we proposed to modify our methodology to estimate the outlier reconciliation dollars based on the operating and capital outlier amounts reported on the FY 2019 cost reports and supplemental data collected from the MACs as described further in this section.
Similar to FY 2020, as part of our proposal for FY 2025 to incorporate into the outlier model the total outlier reconciliation dollars from the most recent and most complete fiscal year cost report data, we also proposed to adjust our estimate of FY 2025 capital outlier payments to incorporate a projection of capital outlier reconciliation payments when determining the adjustment factor to be applied to the capital standard Federal rate to account for the projected proportion of capital IPPS payments paid as outliers (that is, the capital outlier payment adjustment factor). To do so, we proposed to use the following methodology, which generally parallels the proposed methodology to incorporate a projection of operating outlier reconciliation payments for the FY 2025 outlier threshold calculation, including updated data for FY 2025 and additional steps to reflect the cost reports that would be identified with new criteria under the updated instructions.
Step 1a. —Identify hospitals that report on their cost report the capital outlier reconciliation dollars on Worksheet E, Part A, Line 93, Column 1. We note, these were hospitals that were identified by the MACs that met the original criteria for outlier reconciliation and were approved by CMS for outlier reconciliation. We used the Federal FY 2019 cost reports for hospitals paid under the IPPS from the most recent publicly available quarterly HCRIS extract available at the time of development of the proposed and final rules and exclude SCHs that were paid under their hospital-specific rate (that is, if Worksheet E, Part A, Line 48 is greater than Line 47). We note that when there are multiple columns available for the lines of the cost report described in the following steps and the provider was paid under the IPPS for that period(s) of the cost report, then we believe it is appropriate to use multiple columns to fully represent the relevant IPPS payment amounts, consistent with our methodology for the FY 2020 final rule.
Step 1b. —For hospitals that were not included in Step 1a, to identify hospitals that would be referred for outlier reconciliation under the new criteria, we used the same hospitals that were identified in Step 1b of the operating methodology. We note, as discussed previously, the new criteria from CR 13566 is based on the change to the operating CCR (not the capital CCR) where the actual operating CCR for the cost reporting period fluctuates plus or minus 20 percent or more compared to the interim operating CCR used to calculate outlier payments when a bill ( print page 69954) is processed and the hospital had total operating and capital outlier payments greater than $500,000 during the cost reporting period.
Step 2. —Determine the aggregate amount of capital outlier reconciliation dollars (under both the original criteria and the new criteria).
Step 2a. —Calculate the aggregate amount of the historical total of capital outlier reconciliation dollars (Worksheet E, Part A, Line 93, Column 1) using the Federal FY 2019 cost reports from Step 1.
Step 2b. —For the hospitals that would have met the new criteria as identified in Step 1b, to determine the aggregate amount of capital outlier reconciliation dollars, we proposed to use the following process (we note this process is the same as Step 2b of the operating methodology):
We collected supplemental estimated outlier payment data from the MACs for claims with discharges occurring during the hospital's FY 2019 cost reporting period to estimate the change in the hospital's outlier payments. Specifically, for each hospital identified in Step 1b, the MACs used the actual capital CCR calculated from the FY 2019 cost report and the utility in the claims system along with that CCR to determine total outlier payments for claims with discharges occurring during the hospital's FY 2019 cost report (this is the same process MACs would have used if the cost report had been identified for reconciliation had the new criteria been in place for FY 2019 cost reports). For those same claims with discharges occurring during the hospital's 2019 cost report, the MAC provided to CMS the outlier payment as reported on the claim (which was based on the hospital's CCR in the PSF at the time of claim payment).
Using this supplemental estimated outlier payment data, we computed a ratio of the outlier payments based on the actual capital CCR for the FY 2019 cost reporting period and the capital CCR used at the time of claim payment. This ratio is then applied to the capital outlier payment reported on the FY 2019 cost report to impute a capital outlier payment for the FY 2019 cost report. We stated that we believe it is appropriate to impute the capital outlier payment for the cost report using the supplemental data from the MACs described previously rather than use the actual amount reported on the cost report because the claims data in the claims processing system may slightly differ from the cost report data in the HCRIS due to timing. This approach would also allow CMS to use more recent data (from the most recent publicly available quarterly HCRIS extract, which was December 2023 for the proposed rule) to estimate outlier reconciliation dollars as compared to estimating outlier reconciliation dollars using the supplemental data from the MACs which was submitted by the MACs to CMS beginning in November 2022 (as described in this section). This is also the same data used to determine the aggregate amount of capital outlier reconciliation dollars for hospitals from the FY 2019 cost report data using the December 2023 HCRIS extract in Step 2a.
As presented in the table that follows, to calculate the imputed capital outlier payment for the FY 2019 cost report, we multiplied the capital outlier payment reported on the FY 2019 cost report by the following ratio (determined from the supplemental data collected from the MACs described previously): Capital Outlier Payments from MAC using the CCR from FY 2019 Cost Report divided by Capital Outlier Payments from MAC Based on Claim Payment. The general formula is the following: Capital Outlier Payments Reported on the Cost Report * (Capital Outlier Payments from MAC Using CCRs from FY 2019 Cost Report/Capital Outlier Payments from MAC Based on Claim Payment).
To calculate the Estimated Capital Outlier Reconciliation Dollars, we then subtracted the Imputed Capital Outlier Amount for the FY 2019 Cost Report (Step 2b-5) from the Capital Outlier Payment Reported on the FY 2019 Cost Report (Step 2b-1).
The following is an example to illustrate our proposed calculation to determine the estimated amount of capital outlier reconciliation dollars for the hospitals that would have met the new criteria:
We noted the following in the proposed rule, with regard to the data used in the calculation:
- Due to system limitations the MACs needed 13 months to process all providers' claims through the claims utility (for Steps 2b-2 and 2b-3). The MACs used the operating and capital CCR from the FY 2019 cost reports based on the September 2022 HCRIS extract and began processing the supplemental data for FY 2019 outlier payments in November 2022. We proposed to move this forward each year, using the September HCRIS for future fiscal years for the CCRs (for example, for FY 2026, MACs would use CCRs from the 2020 cost reports based on the September 2023 HCRIS).
- For FY 2025, for the “Capital Outlier Payment Reported on the FY 2019 Cost Report” (Step 2b-1) we used capital outlier payments reported on Worksheet L, Part I, Line 2 and Line 2.01 from the FY 2019 cost report using the most recent publicly available quarterly HCRIS extract for the proposed rule (that is, the December 2023 HCRIS extract). We proposed to move this forward each year and use the most recent publicly available quarterly HCRIS extract (for example, for FY 2026, we would use operating capital payments reported on Worksheet L, Part I, Line 2 and Line 2.01 from the FY 2020 cost reports using the most recent publicly available quarterly HCRIS extract).
- For the hospitals identified in Step 1b, we posted a public use file that includes the operating CCR calculated from the FY 2019 cost report in the most recent publicly available quarterly HCRIS extract (the December 2023 HCRIS for the proposed rule), the weighted operating CCR used for claim payment during the FY 2019 cost reporting period from the latest quarterly PSF update (December 2023 for the proposed rule), supplemental data from the MACs and capital outlier ( print page 69955) payments reported on the FY 2019 cost report.
Step 3. —Calculate the aggregate amount of total capital Federal payments across all applicable hospitals using the Federal FY 2019 cost reports. The total capital Federal payments consist of the capital DRG payments, including capital outlier payments, capital indirect medical education (IME) and capital disproportionate share hospital (DSH) payments (Worksheet E, Part A, Line 50, Column 1) and the capital outlier reconciliation amounts from Steps 2a and 2b. We note that a negative amount on Worksheet E, Part A, Line 93 from Step 2a for capital outlier reconciliation indicates an amount that was owed by the hospital, and a positive amount indicates this amount was paid to the hospital. Similarly, a negative amount from Step 2b for capital outlier reconciliation indicates an amount that would have been owed by the hospital, and a positive amount indicates an amount that would have been paid to the hospital.
Step 4. —Divide the aggregate amount from Step 2 (that is, the sum of the amounts from Steps 2a and 2b) by the amount from Step 3 and multiply the resulting amount by 100 to produce the percentage of total capital outlier reconciliation dollars to total capital Federal payments for FY 2019. This percentage amount would be used to adjust the estimate of capital outlier payments for FY 2025 as described in Step 5.
Step 5. —Because the outlier reconciliation dollars are only available on the cost reports, and not in the specific Medicare claims data in the MedPAR file used to estimate outlier payments, we proposed that the estimate of capital outlier payments for FY 2025 would be determined by adding the percentage in Step 5 to the estimated percentage of capital outlier payments otherwise determined using the shared outlier threshold that is applicable to both hospital inpatient operating costs and hospital inpatient capital-related costs. (We noted that this percentage is added for capital outlier payments but subtracted in the analogous step for operating outlier payments. We have a unified outlier payment methodology that uses a shared threshold to identify outlier cases for both operating and capital payments. The difference stems from the fact that operating outlier payments are determined by first setting a “target” percentage of operating outlier payments relative to aggregate operating payments which produces the outlier threshold. Once the shared threshold is set, it is used to estimate the percentage of capital outlier payments to total capital payments based on that threshold. Because the threshold is already set based on the operating target, rather than adjusting the threshold (or operating target), we adjust the percentage of capital outlier to total capital payments to account for the estimated effect of capital outlier reconciliation payments. This percentage is adjusted by adding the capital outlier reconciliation percentage from Step 5 to the estimate of the percentage of capital outlier payments to total capital payments based on the shared threshold.) We noted, when the aggregate capital outlier reconciliation dollars from Steps 2a and 2b are negative, the estimate of capital outlier payments for FY 2025 under our proposed methodology would be lower than the percentage of capital outlier payments otherwise determined using the shared outlier threshold.
For the FY 2025 proposed rule, the estimated percentage of FY 2025 capital outlier payments otherwise determined using the shared outlier threshold was 4.26 percent (estimated capital outlier payments of $290,612,698 divided by (estimated capital outlier payments of $290,612,698 plus the estimated total capital Federal payment of $6,532,600,813)). The proposed ratio in Step 5 was a negative −0.026446 percent ((−$2,056,344/$7,775,606,401) × 100), which, when rounded to the second digit, is −0.03 percent. Therefore, for the FY 2025 proposed rule, taking into account projected capital outlier reconciliation under our proposed methodology decreased the estimated percentage of FY 2025 aggregate capital outlier payments by 0.03 percent.
As discussed in section III.A.2. of this Addendum, we proposed to incorporate the capital outlier reconciliation dollars from Step 5 when applying the outlier adjustment factor in determining the capital Federal rate based on the estimated percentage of capital outlier payments to total capital Federal rate payments for FY 2025.
We invited public comment on our proposed methodology for projecting an estimate of capital outlier reconciliation and incorporating that estimate into the modeling of the estimate of FY 2025 capital outlier payments for purposes of determining the capital outlier adjustment factor.
We did not receive any comments with regard to the steps described previously and we are finalizing as proposed without modification the methodology for projecting an estimate of capital outlier reconciliation and incorporating that estimate into the modeling of the estimate of FY 2025 capital outlier payments for purposes of determining the capital outlier adjustment factor. As we proposed and where stated in the previous steps, for this final rule, we used the most recent HCRIS and PSF data available for this final rule, which is the March 2024 update. Also, for the hospitals identified in Step 1b, similar to the proposed rule, for this final rule, we have posted a public use file that includes the operating CCR calculated from the FY 2019 cost report in the most recent publicly available quarterly HCRIS extract (the March 2024 HCRIS for this final rule), the weighted operating CCR used for claim payment during the FY 2019 cost reporting period from the latest quarterly PSF update (March 2024 for this final rule), supplemental data from the MACs and operating outlier payment reported on the FY 2019 cost report.
With regard to step 5, for this FY 2025 final rule, the estimated percentage of FY 2025 capital outlier payments otherwise determined using the shared outlier threshold is 4.26 percent (estimated capital outlier payments of $292,195,135 divided by (estimated capital outlier payments of $292,195,135 plus the estimated total capital Federal payment of $6,564,012,091)). The ratio in Step 5 is a negative −0. 028042 percent ((−$2,181,440/$7,779,306,800) × 100), which, when rounded to the second digit, is −0.03 percent. Therefore, for this FY 2025 final rule, taking into account projected capital outlier reconciliation under our methodology will decrease the estimated percentage of FY 2025 aggregate capital outlier payments by 0.03 percent.
As discussed in section III.A.2. of this Addendum, we are incorporating the capital outlier reconciliation dollars from Step 5 when applying the outlier adjustment factor in determining the capital Federal rate based on the estimated percentage of capital outlier payments to total capital Federal rate payments for FY 2025.
In the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50977 through 50983 ), in response to public comments on the FY 2013 IPPS/LTCH PPS proposed rule, we made changes to our methodology for projecting the outlier fixed-loss cost threshold for FY 2014. We refer readers to the FY 2014 IPPS/LTCH PPS final rule for a detailed discussion of the changes. ( print page 69956)
As we have done in the past, to calculate the proposed FY 2025 outlier threshold, we simulated payments by applying FY 2025 payment rates and policies using cases from the FY 2023 MedPAR file. As noted in section II.C. of this Addendum, we specify the formula used for actual claim payment which is also used by CMS to project the outlier threshold for the upcoming fiscal year. The difference is the source of some of the variables in the formula. For example, operating and capital CCRs for actual claim payment are from the Provider-Specific File (PSF) while CMS uses an adjusted CCR (as described later in this section) to project the threshold for the upcoming fiscal year. In addition, charges for a claim payment are from the bill while charges to project the threshold are from the MedPAR data with an inflation factor applied to the charges (as described earlier).
To determine the FY 2025 outlier threshold, we inflated the charges on the MedPAR claims by 2 years, from FY 2023 to FY 2025. Consistent with the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42626 and 42627 ), we proposed to use the following methodology to calculate the charge inflation factor for FY 2025:
- Include hospitals whose last four digits fall between 0001 and 0899 (section 2779A1 of Chapter 2 of the State Operations Manual on the CMS website at https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/som107c02.pdf ); include CAHs and REHs that were IPPS hospitals for the time period of the MedPAR data being used to calculate the charge inflation factor; include hospitals in Maryland; and remove PPS-excluded cancer hospitals that have a “V” in the fifth position of their provider number or a “E” or “F” in the sixth position.
- Include providers that are in both periods of charge data that are used to calculate the 1-year average annual rate of-change in charges per case. We note this is consistent with the methodology used since FY 2014.
- We excluded Medicare Advantage IME claims for the reasons described in section I.A.4. of this Addendum. We refer readers to the FY 2011 IPPS/LTCH PPS final rule for a complete discussion on our methodology of identifying and adding the total Medicare Advantage IME payment amount to the budget neutrality adjustments.
- In order to ensure that we capture only FFS claims, we included claims with a “Claim Type” of 60 (which is a field on the MedPAR file that indicates a claim is an FFS claim).
- In order to further ensure that we capture only FFS claims, we excluded claims with a “GHOPAID” indicator of 1 (which is a field on the MedPAR file that indicates a claim is not an FFS claim and is paid by a Group Health Organization).
- We examined the MedPAR file and removed pharmacy charges for anti-hemophilic blood factor (which are paid separately under the IPPS) with an indicator of “3” for blood clotting with a revenue code of “0636” from the covered charge field. We also removed organ acquisition charges from the covered charge field because organ acquisition is a pass-through payment not paid under the IPPS. As noted previously, we are removing allogeneic hematopoietic stem cell acquisition charges from the covered charge field for budget neutrality adjustments. As discussed in the FY 2021 IPPS/LTCH PPS final rule, payment for allogeneic hematopoietic stem cell acquisition costs is made on a reasonable cost basis for cost reporting periods beginning on or after October 1, 2020 ( 85 FR 58835 through 58842 ).
- Because this payment simulation uses the FY 2025 relative weights, consistent with our policy discussed in section IV.I. of the preamble to this final rule, we applied the adjustor for certain cases that group to MS-DRG 018 in our simulation of these payments.
Our general methodology to inflate the charges computes the 1-year average annual rate-of-change in charges per case which is then applied twice to inflate the charges on the MedPAR claims by 2 years since we typically use claims data for the fiscal year that is 2 years prior to the upcoming fiscal year.
In the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42627 ), we modified our charge inflation methodology. We stated that we believe balancing our preference to use the latest available data from the MedPAR files and stakeholders' concerns about being able to use publicly available MedPAR files to review the charge inflation factor can be achieved by modifying our methodology to use the publicly available Federal fiscal year period (that is, for FY 2020, we used the charge data from Federal fiscal years 2017 and 2018), rather than the most recent data available to CMS which, under our prior methodology, was based on calendar year data. We refer the reader to the FY 2020 IPPS/LTCH PPS final rule for a complete discussion regarding this change.
For the same reasons discussed in that rulemaking, for FY 2025, we proposed to use the same methodology as FY 2020 to determine the charge inflation factor. That is, for FY 2025, we proposed to use the MedPAR files for the two most recent available Federal fiscal year time periods to calculate the charge inflation factor, as we did for FY 2020. Specifically, for the proposed rule we used the December 2022 MedPAR file of FY 2022 (October 1, 2021 to September 30, 2022) charge data (released for the FY 2024 IPPS/LTCH PPS proposed rule) and the December 2023 MedPAR file of FY 2023 (October 1, 2022 to September 30, 2023) charge data (released for the FY 2025 IPPS/LTCH PPS proposed rule) to compute the proposed charge inflation factor. We proposed that for the FY 2025 final rule, we would use more recently updated data, that is the MedPAR files from March 2023 for the FY 2022 time period and March 2024 for the FY 2023 time period.
For FY 2025, under this proposed methodology, to compute the 1-year average annual rate-of-change in charges per case, we compared the average covered charge per case of $82,570.13 ($574,544,024,043/6,958,255) from October 1, 2021 through September 30, 2022, to the average covered charge per case of $85,990.03 ($593,444,028,889/6,901,312) from October 1, 2022 through September 30, 2023. This rate-of-change was 4.142 percent (1.04142) or 8.4555 percent (1.084555) over 2 years. The billed charges are obtained from the claims from the MedPAR file and inflated by the inflation factor specified previously.
As we have done in the past, in this FY 2025 IPPS/LTCH PPS proposed rule, we proposed to establish the FY 2025 outlier threshold using hospital CCRs from the December 2023 update to the Provider-Specific File (PSF), the most recent available data at the time of the development of the proposed rule. We proposed to apply the following edits to providers' CCRs in the PSF. We believe these edits are appropriate to accurately model the outlier threshold. We first search for Indian Health Service providers and those providers assigned the statewide average CCR from the current fiscal year. We then replace these CCRs with the statewide average CCR for the upcoming fiscal year. We also assign the statewide average CCR (for the upcoming fiscal year) to those providers that have no value in the CCR field in the PSF or whose CCRs exceed the ceilings described later in this section (3.0 standard deviations from the mean of the log distribution of CCRs for all hospitals). We do not apply the adjustment factors described later in this section to hospitals assigned the statewide average CCR. For FY 2025, we proposed to continue to apply an adjustment factor to the CCRs to account for cost and charge inflation (as explained later in this section). We also proposed that, if more recent data ( print page 69957) become available, we would use that data to calculate the final FY 2025 outlier threshold.
In the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50979 ), we adopted a new methodology to adjust the CCRs. Specifically, we finalized a policy to compare the national average case-weighted operating and capital CCR from the most recent update of the PSF to the national average case-weighted operating and capital CCR from the same period of the prior year.
Therefore, as we have done in the past, we proposed to adjust the CCRs from the December 2023 update of the PSF by comparing the percentage change in the national average case weighted operating CCR and capital CCR from the December 2022 update of the PSF to the national average case weighted operating CCR and capital CCR from the December 2023 update of the PSF. We note that, in the proposed rule, we used total transfer-adjusted cases from FY 2023 to determine the national average case weighted CCRs for both sides of the comparison. As stated in the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50979 ), we believe that it is appropriate to use the same case count on both sides of the comparison because this will produce the true percentage change in the average case-weighted operating and capital CCR from one year to the next without any effect from a change in case count on different sides of the comparison.
Using the proposed methodology, for the proposed rule, we calculated a December 2022 operating national average case-weighted CCR of 0.246416 and a December 2023 operating national average case-weighted CCR of 0.254624.We then calculated the percentage change between the two national operating case-weighted CCRs by subtracting the December 2022 operating national average case-weighted CCR from the December 2023 operating national average case-weighted CCR and then dividing the result by the December 2022 national operating average case-weighted CCR. This resulted in a proposed one-year national operating CCR adjustment factor of 1.03331.
We used this same proposed methodology to adjust the capital CCRs. Specifically, we calculated a December 2022 capital national average case-weighted CCR of 0.018005 and a December 2023 capital national average case-weighted CCR of 0.017765. We then calculated the percentage change between the two national capital case-weighted CCRs by subtracting the December 2022 capital national average case-weighted CCR from the December 2023 capital national average case-weighted CCR and then dividing the result by the December 2022 capital national average case-weighted CCR. This resulted in a proposed one-year national capital CCR adjustment factor of 0.98667.
For purposes of estimating the proposed outlier threshold for FY 2025, we used a wage index that reflects the policies discussed in the proposed rule. This includes the following:
- Application of the proposed rural and imputed floor adjustment.
- The proposed frontier State floor adjustments in accordance with section 10324(a) of the Affordable Care Act.
- The proposed out-migration adjustment as added by section 505 of Public Law 108-173 .
- Incorporating the proposed FY 2025 low wage index hospital policy (described in section III.G.5 of the preamble of this final rule) for hospitals with a wage index value below the 25th percentile, where the increase in the wage index value for these hospitals would be equal to half the difference between the otherwise applicable final wage index value for a year for that hospital and the 25th percentile wage index value for that year across all hospitals.
- Incorporating our policy (described in section III.6. of the preamble of this final rule) to apply a 5-percent cap on any decrease to a hospital's wage index from its wage index in the prior FY, regardless of the circumstances causing the decline.
As stated earlier, if we did not take the aforementioned into account, our estimate of total FY 2025 payments would be too low, and, as a result, our outlier threshold would be too high, such that estimated outlier payments would be less than our projected 5.1 percent of total payments (which includes outlier reconciliation).
As described in sections V.K. and V.L., respectively, of the preamble of this final rule, sections 1886(q) and 1886(o) of the Act establish the Hospital Readmissions Reduction Program and the Hospital VBP Program, respectively. We do not believe that it is appropriate to include the proposed hospital VBP payment adjustments and the hospital readmissions payment adjustments in the proposed outlier threshold calculation or the proposed outlier offset to the standardized amount. Specifically, consistent with our definition of the base operating DRG payment amount for the Hospital Readmissions Reduction Program under § 412.152 and the Hospital VBP Program under § 412.160, outlier payments under section 1886(d)(5)(A) of the Act are not affected by these payment adjustments. Therefore, outlier payments would continue to be calculated based on the unadjusted base DRG payment amount (as opposed to using the base-operating DRG payment amount adjusted by the hospital readmissions payment adjustment and the hospital VBP payment adjustment). Consequently, we proposed to exclude the estimated hospital VBP payment adjustments and the estimated hospital readmissions payment adjustments from the calculation of the proposed outlier fixed-loss cost threshold.
We note that, to the extent section 1886(r) of the Act modifies the DSH payment methodology under section 1886(d)(5)(F) of the Act, the uncompensated care payment under section 1886(r)(2) of the Act, like the empirically justified Medicare DSH payment under section 1886(r)(1) of the Act, may be considered an amount payable under section 1886(d)(5)(F) of the Act such that it would be reasonable to include the payment in the outlier determination under section 1886(d)(5)(A) of the Act. As we have done since the implementation of uncompensated care payments in FY 2014, for FY 2025, we proposed to allocate an estimated per-discharge uncompensated care payment amount to all cases for the hospitals eligible to receive the uncompensated care payment amount in the calculation of the outlier fixed-loss cost threshold methodology. We continue to believe that allocating an eligible hospital's estimated uncompensated care payment to all cases equally in the calculation of the outlier fixed-loss cost threshold would best approximate the amount we would pay in uncompensated care payments during the year because, when we make claim payments to a hospital eligible for such payments, we would be making estimated per-discharge uncompensated care payments to all cases equally.
Furthermore, we continue to believe that using the estimated per-claim uncompensated care payment amount to determine outlier estimates provides predictability as to the amount of uncompensated care payments included in the calculation of outlier payments. Therefore, consistent with the methodology used since FY 2014 to calculate the outlier fixed-loss cost threshold, for FY 2025, we proposed to include estimated FY 2025 uncompensated care payments in the computation of the proposed outlier fixed-loss cost threshold. Specifically, we proposed to use the estimated per-discharge uncompensated care payments to hospitals eligible for the ( print page 69958) uncompensated care payment for all cases in the calculation of the proposed outlier fixed-loss cost threshold methodology.
In addition, consistent with the methodology finalized in the FY 2023 final rule, we proposed to include the estimated supplemental payments for eligible IHS/Tribal hospitals and Puerto Rico hospitals in the computation of the FY 2025 proposed outlier fixed-loss cost threshold. Specifically, we proposed to use the estimated per-discharge supplemental payments to hospitals eligible for the supplemental payment for all cases in the calculation of the proposed outlier fixed-loss cost threshold methodology.
Using this methodology, we used the formula described in section I.C.1. of this Addendum to simulate and calculate the Federal payment rate and outlier payments for all claims. In addition, as described in the earlier section to this Addendum, we proposed to incorporate an estimate of FY 2025 outlier reconciliation in the methodology for determining the outlier threshold. As noted previously, for the FY 2025 proposed rule, the ratio of outlier reconciliation dollars to total Federal Payments (Step 4) is a negative 0.039789 percent, which, when rounded to the second digit, is −0.04 percent. Therefore, for FY 2025, we proposed to incorporate a projection of outlier reconciliation dollars by targeting an outlier threshold at 5.14 percent [5.1 percent − (−.04 percent)]. Under this proposed approach, we determined a proposed threshold of $49,237 and calculated total outlier payments of $4,330,371,122 and total operating Federal payments of $79,917,085,666. We then divided total outlier payments by total operating Federal payments plus total outlier payments and determined that this threshold matched with the 5.14 percent target, which reflected our proposal to incorporate an estimate of outlier reconciliation in the determination of the outlier threshold (as discussed in more detail in the previous section of this Addendum). We noted that, if calculated without applying our proposed methodology for incorporating an estimate of outlier reconciliation in the determination of the outlier threshold, the proposed threshold would be $49,601. We proposed an outlier fixed-loss cost threshold for FY 2025 equal to the prospective payment rate for the MS-DRG, plus any IME, empirically justified Medicare DSH payments, estimated uncompensated care payment, estimated supplemental payment for eligible IHS/Tribal hospitals and Puerto Rico hospitals, and any add-on payments for new technology, plus $49,237.
Comment: A commenter requested that CMS apply trims when calculating charge inflation as it does under the LTCH PPS to “remove all claims from providers whose growth in average charges was a statistical outlier”.
Response: We responded to a similar comment in the FY 2024 IPPS/LTCH final rule ( 88 FR 59351 ). As we explained in that final rule, there are many more providers and claims under the IPPS compared to the LTCH PPS. When we analyzed the LTCH PPS claims data, a single LTCH provider had substantial increases in its charges with average charges per case of approximately $10 million which significantly influenced the charge inflation factor. Since there are fewer hospitals and claims under the LTCH PPS, the potential for a single provider to influence the charge inflation factor is much more significant. We are not aware of a similar situation with a hospital having such high average charges under the IPPS. Therefore, we believe it is not necessary to apply the same trim to hospitals included in the IPPS charge inflation factor. We refer the reader to the FY 2024 IPPS/LTCH final rule for our complete response.
Comment: Commenters were concerned about the proposed increase in the high-cost outlier threshold, a 15 percent increase from the FY 2024 threshold, which they stated would significantly decrease the number of cases that qualify for an outlier payment. The commenters stated that the proposed increase in the threshold compared to FY 2024 is substantial and comes after a decade of increases which amount to a 126 percent increase from FY 2013 through FY 2025.
Commenters stated that they believe much of the increase in FY 2025 is being driven by the fact that CMS has estimated and proposed to use a one-year national operating CCR adjustment factor of 1.03331. These commenters stated that the CCR adjustment factor is much higher than it has been in the past and is largely driven by CCRs that are reflecting the high-cost inflation, namely labor costs, that have been experienced by hospitals during 2022 and 2023.
A commenter stated that CMS' proposed operating CCR adjustment factor for FY 2025 is anomalous and would be the first use of a projected increase in the CCRs since FY 2013. The commenter asserted that the anomalous first-time year-over-year increase in CCRs, used for the proposed FY 2025 adjustment factor, is driven by CCRs skewed by costs—largely labor costs—incurred during the peak inflationary period of the COVID-19 PHE in 2022 and early 2023. The commenter explained that CMS' projection of a one-year 1.033 change in CCRs suggest that average costs per case increased by over 7 percent, given that CMS estimated average charge inflation of about 4.4 percent from FY 2022 to FY 2023. The commenter stated that CMS' current data and projections reflect a Q4 2022 peak in the four-quarter moving average percent change to the market basket index level followed by a slowing in cost inflation.
The commenter further stated that a comparison of the March 2023 and March 2024 updates of the PSF (in lieu of the December 2022 and December 2023 updates of the PSF used in the proposed rule) shows that CCRs are again declining such that a CCR adjustment factor greater than 1.0 is unreasonable. Despite this decline, the commenter expressed concern that a CCR adjustment factor calculated from this data will continue to be skewed by data from an anomalous period of rapid inflation. The commenter asserted that a preliminary review of HCRIS data indicates that CCRs are in fact declining in ways that are not yet reflected in the PSF. Therefore, the commenter urged CMS to not only use more recent data from the PSF when finalizing a CCR adjustment factor, but also to address the skewing impact of older CCR data (namely that from 2022 and earlier) in the PSF so as to develop a CCR adjustment factor that reasonably projects CCRs for FY 2025.
In addition, the commenter stated that in the past, CMS has deviated from its general methodology for calculating the CCR adjustment factor when appropriate to ensure that the CCR adjustment factor provides a reasonable approximation of anticipated CCR trends. The commenter cited the FY 2023 IPPS/LTCH final rule ( 87 FR 48780 , 48797 ) where CMS did not use its usual methodology because it would have produced an “abnormally high” CCR adjustment factor of approximately 1.03 and instead applied a CCR adjustment factor from the last 1-year period prior to the COVID-19 PHE. The commenter concluded that it is likewise unreasonable to assume that CCRs will continue to increase at the abnormally high rates seen during a period of rapid and significant cost increases and urged CMS to modify its method for FY 2025 to develop a CCR adjustment factor that is consistent with historical pre-PHE period CCR changes and that reflects the most recent CCR data, which demonstrate a consistent trend of decreasing CCRs. ( print page 69959)
Other commenters recommended that CMS examine its methodology more closely and consider making additional, temporary changes to help mitigate the substantial increases that are occurring in the outlier threshold. The commenters suggested that CMS could instead apply the FY 2024 CCR adjustment factor in calculating the FY 2025 outlier threshold, which they stated would mitigate the anomalous increase. Another commenter suggested that CMS should keep the outlier threshold flat due to the increase in the threshold finalized in the FY 2024 IPPS/LTCH PPS final rule. The commenter stated that since MS-DRGs were introduced back in 2007, the fixed cost outlier threshold has increased at a steady, gradual pace. A commenter suggested that CMS review methodological changes to improve base MS-DRG payment rates that would facilitate a decrease in the number of cases for which outlier payments are made on a routine basis. Another commenter stated that CMS be more consistent in determining the outlier threshold so there are not significant variations year over year. The commenter suggested that one alternative would be to consider establishing a new outlier baseline and then increasing the outlier threshold each year by the approved market basket percentage or CMS could implement a three-year rolling average for calculating the outlier threshold as a stabilizing factor. A commenter encouraged CMS to limit the final FY 2025 and future year over year increases in the outlier threshold to ensure more accurate and appropriate reimbursement rates across high cost cases.
Response: We appreciate the commenters' feedback. While the proposed CCR adjustment factor was based on a comparison of average CCRs from the December 2022 and December 2023 updates of the PSF, consistent with our usual practice we are using more recent CCR data for the final rule. Specifically, for this final rule, as discussed in greater detail below, based on the latest data available, we calculated a national operating CCR adjustment factor of 1.015123, which is slightly over 1.0 and less than half the increase of the proposed factor of 1.03331 in the proposed rule.
We agree that prior to recent years the CCRs have decreased year-over-year, and as such the CCR adjustment factors were slightly less than 1.0 compared to the slightly over 1.0 value calculated for this final rule. In FY 2023 we did not use the factor based on the most recent data available at that time, stating that we believed the abnormally high CCR adjustment factor as compared to historical levels was partially due to the high number of COVID-19 cases with higher charges that were treated in IPPS hospitals in FY 2021. As noted by the commenters, there are other factors aside from COVID-19 cases, such as higher labor costs and inflation, that may be contributing to the change in average CCRs above 1.0. We acknowledge there can be variation in the annual changes in CCRs and charges, as noted by the commenter's examples. At this time, it is challenging to precisely predict the relative relationship between hospitals' costs and charging practices for the upcoming FY. It is reasonable to assume balancing older historical data ( i.e., pre COVID-19 PHE) used to determine the CCR adjustment factor) with more recent data ( i.e., during and post COVID-19 PHE) to calculate the CCR adjustment factor that the resulting CCR adjustment factor may be slightly over 1.0 or slightly under 1.0. In other words, and as explained earlier, the current conditions such as higher labor costs and inflation were not as prevalent with the historical older data as the CCR adjustment factor was consistently under 1.0, so it is unclear at this time whether change in average CCRs would return to being under 1.0 or remain above 1.0. Given that the use of the most recent data available, after updating it for this final rule, results in a CCR adjustment factor that is within that reasonable range and that it is challenging to precisely predict the relative relationship between hospitals' costs and charging practices for the upcoming FY, we do not believe it is necessary to deviate from our usual practice of using the most recent data available to determine the CCR adjustment factor for FY 2025. We believe a national operating CCR adjustment factor of 1.015123, which is slightly over 1.0, used in conjunction with the charge inflation factor discussed below results in a reasonable prediction of average costs in FY 2025 for purposes of the outlier threshold calculation.
With regard to the other suggestions from the commenters, as noted previously, section 1886(d)(5)(A)(iv) of the Act states that outlier payments may not be less than 5 percent nor more than 6 percent of the total payments projected or estimated to be made based on DRG prospective payment rates for discharges in that year. We believe that the commenters' suggestion to maintain the FY 2024 outlier fixed-loss cost threshold for FY 2025 or using a rolling average or limiting the increase in the outlier threshold would be inconsistent with the statute as such a threshold would not result in a projection outlier payments that are not less than 5 percent nor more than 6 percent of projected total payments for FY 2025.
With regard to the commenter that suggested CMS review methodological changes to improve base MS-DRG payment rates that would facilitate a decrease in the number of cases for which outlier payments are made on a routine basis, the commenter did not provide any specific suggestions and we welcome suggestions from the commenter that we can consider for future rulemaking.
Comment: A commenter requested that CMS consider whether it is appropriate to include extreme cases when calculating the threshold. This commenter explained that high charge cases have a significant impact on the threshold. The commenter stated that it examined the data to understand the factors that drove an increase of over 80 percent between FY 2017 and FY 2024, and to propose to increase the threshold almost an additional 15 percent for FY 2025, and stated that it observed that the inclusion of extreme cases in the calculation of the threshold, the rate of which are increasing over time, significantly impacts CMS' determination of the fixed-loss threshold. If this trend continues (that is, if the number (and proportion) of extreme cases continues to increase each year), the commenter stated that the impact of this population of cases on the threshold will likewise increase. Thus, the commenter recommended that CMS carefully consider what is causing this trend, whether the inclusion of these cases in the calculation of the threshold is appropriate, or whether a separate outlier mechanism should apply to these cases that more closely hews outlier payments to marginal costs.
Response: We responded to a similar comment in prior rulemaking, most recently in the FY 2024 IPPS/LTCH final rule ( 88 FR 59352 ). Specifically, in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38526 ) and other prior rulemaking, we explained the methodology used to calculate the outlier threshold includes all claims to account for all different types of cases, including high charge cases, to ensure that CMS meets the 5.1 percent target. As the commenter pointed out, the volume of these cases continues to rise, making their impact on the threshold significant. We believe excluding these cases would artificially lower the threshold. We believe it is important to include all cases in the calculation of the threshold no matter how high or low ( print page 69960) the charges. Including these cases with high charges lends more accuracy to the threshold, as these cases have an impact on the threshold and continue to rise in volume. Therefore, we believe the inclusion of the high-cost outlier cases in the calculation of the outlier threshold is appropriate.
Also, as we explained in the FY 2024 IPPS/LTCH final rule ( 88 FR 59352 ). in response to commenter's recommendation that CMS consider whether a separate outlier mechanism should apply to these cases that more closely hews outlier payments to marginal costs, we believe the current calculation of outlier payment meets these goals. If a case has high charges that once reduced to cost significantly exceed the payment plus the threshold, then the case will receive a larger outlier payment reflective of the higher costs. Therefore, we believe the current payment system provides such a mechanism.
Comment: A commenter noted the final fixed-loss threshold established by CMS has consistently been lower than the threshold set forth in the proposed rule, and the variance between the proposed and final thresholds has generally exceeded 4 percent. The commenter emphasized that this demonstrates that CMS must ordinarily use the most recent data to appropriately calculate the outlier threshold.
Response: We responded to similar comments in the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50378 through 50379 ) and refer readers to that rule for our response. We reiterate that CMS' historical policy is to use the best available data when setting the payment rates and factors in both the proposed and final rules. Sometimes there are variables that change between the proposed and final rule as result of the availability of more recent data, such as the charge inflation factor and the CCR adjustment factors that can cause fluctuations in the threshold amount. Other factors such as changes to the wage indexes and market basket increase can also cause the outlier fixed loss cost threshold to fluctuate between the proposed rule and the final rule each year. We use the latest data that is available at the time of the development of the proposed and final rules, such as the most recent update of MedPAR claims data and CCRs from the most recent update of the PSF.
After consideration of the public comments we received and for the reasons discussed, we are finalizing to use the same methodology we proposed, without modifications, to calculate the final outlier threshold for FY 2025.
For the FY 2025 final outlier threshold, we used the March 2023 MedPAR file of FY 2022 (October 1, 2021 through September 30, 2022) charge data (released in conjunction with the FY 2024 IPPS/LTCH PPS final rule) and the March 2024 MedPAR file of FY 2023 (October 1, 2022 through September 30, 2023) charge data (released in conjunction with this FY 2025 IPPS/LTCH PPS final rule) to determine the charge inflation factor. To compute the 1-year average annual rate-of-change in charges per case, we compared the average covered charge per case of $ 82,677.79 ($577,981,065,082/6,990,766 cases) from October 1, 2021 through September 31, 2022, to the average covered charge per case of $ 86,082.41 ($596,812,542,644/6,933,037 cases) from October 1, 2021 through September 31, 2023. This rate-of-change was 4.1 percent (1.04118) or 8.4 percent (1.08406) over 2 years. The billed charges are obtained from the claims from the MedPAR file and inflated by the inflation factor specified previously.
As we have done in the past, we are establishing the FY 2025 outlier threshold using hospital CCRs from the March 2024 update to the Provider-Specific File (PSF)—the most recent available data at the time of the development of the final rule. We applied the following edits to providers' CCRs in the PSF. We believe these edits are appropriate to accurately model the outlier threshold. We first search for Indian Health Service providers and those providers assigned the statewide average CCR from the current fiscal year. We then replaced these CCRs with the statewide average CCR for the upcoming fiscal year. We also assigned the statewide average CCR (for the upcoming fiscal year) to those providers that have no value in the CCR field in the PSF or whose CCRs exceed the ceilings described later in this section (3.0 standard deviations from the mean of the log distribution of CCRs for all hospitals). We did not apply the adjustment factors described later in this section to hospitals assigned the statewide average CCR. For FY 2025, we also are continuing to apply an adjustment factor to the CCRs to account for cost and charge inflation (as explained later in this section).
For this final rule, as we have done since FY 2014 (with the exception of FYs 2022 and 2023, as discussed in the FY 2022 and FY 2023 IPPS/LTCH PPS proposed and final rules), we are adjusting the CCRs from the March 2024 update of the PSF by comparing the percentage change in the national average case-weighted operating CCR and capital CCR from the March 2023 update of the PSF to the national average case-weighted operating CCR and capital CCR from the March 2024 update of the PSF. We note that we used total transfer-adjusted cases from FY 2023 to determine the national average case weighted CCRs for both sides of the comparison. As stated in the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50979 ), we believe that it is appropriate to use the same case count on both sides of the comparison because this will produce the true percentage change in the average case-weighted operating and capital CCR from one year to the next without any effect from a change in case count on different sides of the comparison.
Using the methodology noted earlier, for this final rule, we calculated a March 2023 operating national average case-weighted CCR of 0.24849 and a March 2024 operating national average case-weighted CCR of 0.252248. We then calculated the percentage change between the two national operating case-weighted CCRs by subtracting the March 2023 operating national average case weighted CCR from the March 2024 operating national average case-weighted CCR and then dividing the result by the March 2023 national operating average case-weighted CCR. This resulted in a national operating CCR adjustment factor of 1.015123.
We used the same methodology earlier to adjust the capital CCRs. Specifically, for this final rule, we calculated a March 2023 capital national average case-weighted CCR of 0.017716 and a March 2024 capital national average case-weighted CCR of 0.017666. We then calculated the percentage change between the two national capital case weighted CCRs by subtracting the March 2023 capital national average case-weighted CCR from the March 2024 capital national average case-weighted CCR and then dividing the result by the March 2023 capital national average case-weighted CCR. This resulted in a national capital CCR adjustment factor of 0.997178.
As discussed previously, for purposes of estimating the final outlier threshold for FY 2025, we used a wage index that reflects the policies discussed in this final rule. This includes the following:
- Application of the rural and imputed floor adjustment.
- The frontier State floor adjustments in accordance with section 10324(a) of the Affordable Care Act.
- The out-migration adjustment as added by section 505 of Public Law 108-173 .
- Incorporating the FY 2025 low wage index hospital policy (described in ( print page 69961) section III.G.5 of the preamble of this final rule) for hospitals with a wage index value below the 25th percentile, where the increase in the wage index value for these hospitals would be equal to half the difference between the otherwise applicable final wage index value for a year for that hospital and the 25th percentile wage index value for that year across all hospitals.
As stated previously, if we did not take the above into account, into our estimate of total FY 2025 payments would be too low, and, as a result, our outlier threshold would be too high, such that estimated outlier payments would be less than our projected 5.14 percent of total payments (which reflects the estimate of outlier reconciliation calculated for this final rule).
- We excluded the hospital VBP payment adjustments and the hospital readmissions payment adjustments from the calculation of the outlier fixed-loss cost threshold.
- We used the estimated per-discharge uncompensated care payments to hospitals eligible for the uncompensated care payment for all cases in the calculation of the outlier fixed-loss cost threshold methodology.
- Based on the policy finalized, as previously described, we used the estimated per-discharge supplemental payments to hospitals eligible for the supplemental payment for all cases in the calculation of the proposed outlier fixed-loss cost threshold methodology.
Using this methodology, we used the formula described in section I.C.1. of this Addendum to simulate and calculate the Federal payment rate and outlier payments for all claims. In addition, as described in the earlier section to this Addendum, we are finalizing to incorporate an estimate of FY 2025 outlier reconciliation in the methodology for determining the outlier threshold. As noted previously, for this final rule, the ratio of outlier reconciliation dollars to total Federal Payments (Step 4) is a negative 4.1994 percent, which, when rounded to the second digit, is -0.04 percent. Therefore, for FY 2025, we incorporated a projection of outlier reconciliation dollars by targeting an outlier threshold at 5.14 percent [5.1 percent-(-.04 percent)]. Under this approach, we determined a threshold of $ 46,152 and calculated total outlier payments of $ 4,349,520,041and total operating Federal payments of $ 80,269,760,637. We then divided total outlier payments by total operating Federal payments plus total outlier payments and determined that this threshold matched with the 5.14 percent target, which incorporated an estimate of outlier reconciliation in the determination of the outlier threshold (as discussed in more detail in the previous section of this Addendum). We note that, if calculated without applying our methodology for incorporating an estimate of outlier reconciliation in the determination of the outlier threshold, the threshold would be $46,502. We are finalizing an outlier fixed-loss cost threshold for FY 2025 equal to the prospective payment rate for the MS-DRG, plus any IME, empirically justified Medicare DSH payments, estimated uncompensated care payment, estimated supplemental payment for eligible IHS/Tribal hospitals and Puerto Rico hospitals, and any add on payments for new technology, plus $46,152.
As stated in the FY 1994 IPPS final rule ( 58 FR 46348 ), we establish an outlier threshold that is applicable to both hospital inpatient operating costs and hospital inpatient capital-related costs. When we modeled the combined operating and capital outlier payments, we found that using a common threshold resulted in a higher percentage of outlier payments for capital-related costs than for operating costs. We project that the threshold for FY 2025 (which reflects our methodology to incorporate an estimate of operating outlier reconciliation) would result in outlier payments that would equal 5.1 percent of operating DRG payments and we estimate that capital outlier payments would equal 4.23 percent of capital payments based on the Federal rate (which reflects our methodology discussed previously to incorporate an estimate of capital outlier reconciliation).
In accordance with section 1886(d)(3)(B) of the Act and as discussed previously, we reduce the FY 2025 standardized amount by 5.1 percent to account for the projected proportion of payments paid as outliers.
The outlier adjustment factors that would be applied to the operating standardized amount and capital Federal rate based on the FY 2025 outlier threshold are as follows:

We are applying the outlier adjustment factors to the FY 2025 payment rates after removing the effects of the FY 2024 outlier adjustment factors on the standardized amount.
To determine whether a case qualifies for outlier payments, we currently apply hospital-specific CCRs to the total covered charges for the case. Estimated operating and capital costs for the case are calculated separately by applying separate operating and capital CCRs. These costs are then combined and compared with the outlier fixed-loss cost threshold.
Under our current policy at § 412.84, we calculate operating and capital CCR ceilings and assign a statewide average CCR for hospitals whose CCRs exceed 3.0 standard deviations from the mean of the log distribution of CCRs for all hospitals. Based on this calculation, for hospitals for which the MAC computes operating CCRs greater than 1.283 or capital CCRs greater than 0.132 or hospitals for which the MAC is unable to calculate a CCR (as described under § 412.84(i)(3) of our regulations), statewide average CCRs are used to determine whether a hospital qualifies for outlier payments. Table 8A listed in section VI. of this Addendum (and available via the internet on the CMS website) contains the statewide average operating CCRs for urban hospitals and for rural hospitals for which the MAC is unable to compute a hospital-specific CCR within the range previously specified. These statewide average ratios would be effective for discharges occurring on or after October 1, 2024 ( print page 69962) and would replace the statewide average ratios from the prior fiscal year. Table 8B listed in section VI. of this Addendum (and available via the internet on the CMS website) contains the comparable statewide average capital CCRs. As previously stated, the CCRs in Tables 8A and 8B would be used during FY 2025 when hospital-specific CCRs based on the latest settled cost report either are not available or are outside the range noted previously. Table 8C listed in section VI. of this Addendum (and available via the internet on the CMS website) contains the statewide average total CCRs used under the LTCH PPS as discussed in section V. of this Addendum.
We finally note that section 20.1.2 of chapter three of the Medicare Claims Processing Manual (on the internet at https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/clm104c03.pdf ) covers an array of topics, including CCRs, reconciliation, and the time value of money. Per the regulations at 42 CFR 412.84(i)(1) , a hospital may request that its MAC use a different (higher or lower) cost-to-charge ratio based on substantial evidence presented by the hospital. We encourage hospitals that are assigned the statewide average operating and/or capital CCRs to work with their MAC on a possible alternative operating and/or capital CCR as explained in the manual. Use of an alternative CCR developed by the hospital in conjunction with the MAC can avoid possible overpayments or underpayments at cost report settlement, thereby ensuring better accuracy when making outlier payments and negating the need for outlier reconciliation. We also note that a hospital may request an alternative operating or capital CCR at any time as long as the guidelines of the manual are followed. In addition, the manual outlines the outlier reconciliation process for hospitals and Medicare contractors. We refer hospitals to the manual instructions for complete details on outlier reconciliation.
Our current estimate, using available FY 2023 claims data, is that actual outlier payments for FY 2023 were approximately 5.27 percent of actual total MS-DRG payments. Therefore, the data indicate that, for FY 2023, the percentage of actual outlier payments relative to actual total payments is higher than we projected for FY 2023. Consistent with the policy and statutory interpretation we have maintained since the inception of the IPPS, we do not make retroactive adjustments to outlier payments to ensure that total outlier payments for FY 2023 are equal to 5.1 percent of total MS-DRG payments. As explained in the FY 2003 Outlier final rule ( 68 FR 34502 ), if we were to make retroactive adjustments to all outlier payments to ensure total payments are 5.1 percent of MS-DRG payments (by retroactively adjusting outlier payments), we would be removing the important aspect of the prospective nature of the IPPS. Because such an across-the-board adjustment would either lead to more or less outlier payments for all hospitals, hospitals would no longer be able to reliably approximate their payment for a patient while the patient is still hospitalized. We believe it would be neither necessary nor appropriate to make such an aggregate retroactive adjustment. Furthermore, we believe it is consistent with the statutory language at section 1886(d)(5)(A)(iv) of the Act not to make retroactive adjustments to outlier payments. This section states that outlier payments be equal to or greater than 5 percent and less than or equal to 6 percent of projected or estimated (not actual) MS-DRG payments. We believe that an important goal of a PPS is predictability. Therefore, we believe that the fixed-loss outlier threshold should be projected based on the best available historical data and should not be adjusted retroactively. A retroactive change to the fixed-loss outlier threshold would affect all hospitals subject to the IPPS, thereby undercutting the predictability of the system as a whole.
We note that, because the MedPAR claims data for the entire FY 2024 period would not be available until after September 30, 2024, we are unable to provide an estimate of actual outlier payments for FY 2024 based on FY 2024 claims data in this final rule. We will provide an estimate of actual FY 2024 outlier payments in the FY 2026 IPPS/LTCH PPS proposed rule.
The adjusted standardized amount is divided into labor-related and nonlabor-related portions. Tables 1A and 1B listed and published in section VI. of this Addendum (and available via the internet on the CMS website) contain the national standardized amounts that we are applying to all hospitals, except hospitals located in Puerto Rico, for FY 2025. The standardized amount for hospitals in Puerto Rico is shown in Table 1C listed and published in section VI. of this Addendum (and available via the internet on the CMS website). The amounts shown in Tables 1A and 1B differ only in that the labor-related share applied to the standardized amounts in Table 1A is 67.6 percent, and the labor-related share applied to the standardized amounts in Table 1B is 62 percent. In accordance with sections 1886(d)(3)(E) and 1886(d)(9)(C)(iv) of the Act, we are applying a labor-related share of 62 percent, unless application of that percentage would result in lower payments to a hospital than would otherwise be made. In effect, the statutory provision means that we would apply a labor-related share of 62 percent for all hospitals whose wage indexes are less than or equal to 1.0000.
In addition, Tables 1A and 1B include the standardized amounts reflecting the applicable percentage increases for FY 2025.
The labor-related and nonlabor-related portions of the national average standardized amounts for Puerto Rico hospitals for FY 2025 are set forth in Table 1C listed and published in section VI. of this Addendum (and available via the internet on the CMS website). Similarly, section 1886(d)(9)(C)(iv) of the Act, as amended by section 403(b) of Public Law 108-173 , provides that the labor-related share for hospitals located in Puerto Rico be 62 percent, unless the application of that percentage would result in lower payments to the hospital.
The following table illustrates the changes from the FY 2024 national standardized amounts to the FY 2025 national standardized amounts. The second through fifth columns display the changes from the FY 2024 standardized amounts for each applicable FY 2025 standardized amount. The first row of the table shows the updated (through FY 2024) average standardized amount after restoring the FY 2024 offsets for outlier payments, geographic reclassification, rural demonstration, lowest quartile, and wage index cap policy budget neutrality. The MS-DRG reclassification and recalibration wage index, and stem cell acquisition budget neutrality factors are cumulative (that is, we have not restored the offsets). Accordingly, those FY 2024 adjustment factors have not been removed from the base rate in the following table. Additionally, for FY 2025 we have applied the budget neutrality factors for the lowest quartile hospital policy, described previously.

Tables 1A through 1C, as published in section VI. of this Addendum (and available via the internet on the CMS website), contain the labor-related and nonlabor-related shares that we are using to calculate the prospective payment rates for hospitals located in the 50 States, the District of Columbia, and Puerto Rico for FY 2025. This section addresses two types of adjustments to the standardized amounts that are made in determining the prospective payment rates as described in this Addendum.
Sections 1886(d)(3)(E) and 1886(d)(9)(C)(iv) of the Act require that we make an adjustment to the labor-related portion of the national prospective payment rate to account for area differences in hospital wage levels. This adjustment is made by multiplying the labor-related portion of the adjusted standardized amounts by the appropriate wage index for the area in which the hospital is located. For FY 2025, as discussed in section IV.B.3. of the preamble of this final rule, we are applying a labor-related share of 67.6 percent for the national standardized amounts for all IPPS hospitals (including hospitals in Puerto Rico) that have a wage index value that is greater than 1.0000. Consistent with section 1886(d)(3)(E) of the Act, we are applying the wage index to a labor-related share of 62 percent of the national standardized amount for all IPPS hospitals (including hospitals in Puerto Rico) whose wage index values are less than or equal to 1.0000. In section III. of the preamble of this final rule, we discuss the data and methodology for the FY 2025 wage index. ( print page 69964)
Section 1886(d)(5)(H) of the Act provides discretionary authority to the Secretary to make adjustments as the Secretary deems appropriate to take into account the unique circumstances of hospitals located in Alaska and Hawaii. Higher labor-related costs for these two States are taken into account in the adjustment for area wages described previously. To account for higher non-labor-related costs for these two States, we multiply the nonlabor-related portion of the standardized amount for hospitals in Alaska and Hawaii by an adjustment factor.
In the FY 2013 IPPS/LTCH PPS final rule, we established a methodology to update the COLA factors for Alaska and Hawaii that were published by the U.S. Office of Personnel Management (OPM) every 4 years (coinciding with the update to the labor-related share of the IPPS market basket), beginning in FY 2014. We refer readers to the FY 2013 IPPS/LTCH PPS proposed and final rules for additional background and a detailed description of this methodology ( 77 FR 28145 through 28146 and 77 FR 53700 through 53701 , respectively). For FY 2022, in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45546 through 45547 ), we updated the COLA factors published by OPM for 2009 (as these are the last COLA factors OPM published prior to transitioning from COLAs to locality pay) using the methodology that we finalized in the FY 2013 IPPS/LTCH PPS final rule. Based on the policy finalized in the FY 2013 IPPS/LTCH PPS final rule, we are continuing to use the same COLA factors in FY 2025 that were used in FY 2024 to adjust the nonlabor-related portion of the standardized amount for hospitals located in Alaska and Hawaii. The following table lists the COLA factors for FY 2025.

Lastly, as we finalized in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53700 and 53701 ), we intend to update the COLA factors at the same time as the update to the labor-related share of the IPPS market basket.
In general, the operating prospective payment rate for all hospitals (including hospitals in Puerto Rico) paid under the IPPS, except SCHs and MDHs, for FY 2025 equals the Federal rate (which includes uncompensated care payments). As previously discussed, section 4102 of the Consolidated Appropriations Act, 2023 ( Pub. L. 117-328 ), enacted on December 29, 2022, extended the MDH program through FY 2024 (that is, for discharges occurring on or before September 30, 2024). Subsequently, section 307 of the Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, further extended the MDH program for discharges occurring before January 1, 2025. Prior to enactment of the CAA, 2024, the MDH program was only to be in effect through the end of FY 2024. Under current law, the MDH program will expire for discharges on or after January 1, 2025.
- The Federal national rate (which, as discussed in section IVE. of the preamble of this final rule, includes uncompensated care payments).
- The updated hospital-specific rate based on FY 2006 costs per discharge to determine the rate that yields the greatest aggregate payment.
The prospective payment rate for SCHs for FY 2025 equals the higher of the applicable Federal rate, or the hospital-specific rate as described later in this section. The prospective payment rate for MDHs for FY 2025 discharges occurring before January 1, 2025 equals the higher of the Federal rate, or the Federal rate plus 75 percent of the difference between the Federal rate and the hospital-specific rate as described in this section. For MDHs, the updated hospital-specific rate is based on FY 1982, FY 1987, or FY 2002 costs per discharge, whichever yields the greatest aggregate payment.
The formula specified in this section is used for actual claim payment and is also used by CMS to project the outlier threshold for the upcoming fiscal year. The difference is the source of some of the variables in the formula. For example, operating and capital CCRs for actual claim payment are from the PSF while CMS uses an adjusted CCR (as described previously) to project the threshold for the upcoming fiscal year. In addition, ( print page 69965) charges for a claim payment are from the bill while charges to project the threshold are from the MedPAR data with an inflation factor applied to the charges (as described earlier).
Step 1—Determine the MS-DRG and MS-DRG relative weight (from Table 5) for each claim primarily based on the ICD-10-CM diagnosis and ICD-10-PCS procedure codes on the claim.
Step 2—Select the applicable average standardized amount depending on whether the hospital submitted qualifying quality data and is a meaningful EHR user, as described previously.
Step 3—Compute the operating and capital Federal payment rate:
—Federal Payment Rate for Operating Costs = MS-DRG Relative Weight × [(Labor-Related Applicable Standardized Amount × Applicable CBSA Wage Index) + (Nonlabor-Related Applicable Standardized Amount × Cost-of-Living Adjustment)] × (1 + IME + (DSH * 0.25))
—Federal Payment for Capital Costs = MS-DRG Relative Weight × Federal Capital Rate × Geographic Adjustment Fact × (l + IME + DSH)
Step 4—Determine operating and capital costs:
—Operating Costs = (Billed Charges × Operating CCR)
—Capital Costs = (Billed Charges × Capital CCR)
Step 5—Compute operating and capital outlier threshold (CMS applies a geographic adjustment to the operating and capital outlier threshold to account for local cost variation):
—Operating CCR to Total CCR = (Operating CCR)/(Operating CCR + Capital CCR)
—Operating Outlier Threshold = [Fixed Loss Threshold × ((Labor-Related Portion × CBSA Wage Index) + Nonlabor-Related portion)] × Operating CCR to Total CCR + Federal Payment with IME, DSH + Uncompensated Care Payment + supplemental payment for eligible IHS/Tribal hospitals and Puerto Rico hospitals + New Technology Add-On Payment Amount
—Capital CCR to Total CCR = (Capital CCR)/(Operating CCR + Capital CCR)
—Capital Outlier Threshold = (Fixed Loss Threshold × Geographic Adjustment Factor × Capital CCR to Total CCR) + Federal Payment with IME and DSH
Step 6—Compute operating and capital outlier payments:
—Marginal Cost Factor = 0.80 or 0.90 (depending on the MS-DRG)
—Operating Outlier Payment = (Operating Costs—Operating Outlier Threshold) × Marginal Cost Factor
—Capital Outlier Payment = (Capital Costs—Capital Outlier Threshold) × Marginal Cost Factor
The payment rate may then be further adjusted for hospitals that qualify for a low-volume payment adjustment under section 1886(d)(12) of the Act and 42 CFR 412.101(b) . The base-operating DRG payment amount may be further adjusted by the hospital readmissions payment adjustment and the hospital VBP payment adjustment as described under sections 1886(q) and 1886(o) of the Act, respectively. Payments also may be reduced by the 1-percent adjustment under the HAC Reduction Program as described in section 1886(p) of the Act. We also make new technology add-on payments in accordance with section 1886(d)(5)(K) and (L) of the Act. Finally, we add the uncompensated care payment and supplemental payment for eligible IHS/Tribal hospitals and Puerto Rico hospitals to the total claim payment amount. As noted in the previous formula, we take uncompensated care payments, supplemental payments for eligible IHS/Tribal hospitals and Puerto Rico hospitals, and new technology add-on payments into consideration when calculating outlier payments.
Section 1886(b)(3)(C) of the Act provides that SCHs are paid based on whichever of the following rates yields the greatest aggregate payment: the Federal rate; the updated hospital-specific rate based on FY 1982 costs per discharge; the updated hospital-specific rate based on FY 1987 costs per discharge; the updated hospital-specific rate based on FY 1996 costs per discharge; or the updated hospital-specific rate based on FY 2006 costs per discharge to determine the rate that yields the greatest aggregate payment. As discussed previously, currently MDHs are paid based on the Federal national rate or, if higher, the Federal national rate plus 75 percent of the difference between the Federal national rate and the greater of the updated hospital-specific rates based on either FY 1982, FY 1987, or FY 2002 costs per discharge. As noted, under current law, the MDH program is effective for FY 2025 discharges on or before December 31, 2024.
For a more detailed discussion of the calculation of the hospital-specific rates, we refer readers to the FY 1984 IPPS interim final rule ( 48 FR 39772 ); the April 20, 1990 final rule with comment period ( 55 FR 15150 ); the FY 1991 IPPS final rule ( 55 FR 35994 ); and the FY 2001 IPPS final rule ( 65 FR 47082 ).
Section 1886(b)(3)(B)(iv) of the Act provides that the applicable percentage increase applicable to the hospital-specific rates for SCHs and MDHs equals the applicable percentage increase set forth in section 1886(b)(3)(B)(i) of the Act (that is, the same update factor as for all other hospitals subject to the IPPS). Because the Act sets the update factor for SCHs and MDHs equal to the update factor for all other IPPS hospitals, the update to the hospital-specific rates for SCHs and MDHs is subject to the amendments to section 1886(b)(3)(B) of the Act made by sections 3401(a) and 10319(a) of the Affordable Care Act. Accordingly, the applicable percentage increases to the hospital-specific rates applicable to SCHs and MDHs are the following:

For a complete discussion of the applicable percentage increase applied to the hospital-specific rates for SCHs and MDHs, we refer readers to section V.F. of the preamble of this final rule.
In addition, because SCHs and MDHs use the same MS-DRGs as other hospitals when they are paid based in whole or in part on the hospital-specific rate, the hospital-specific rate is adjusted by a budget neutrality factor to ensure that changes to the MS-DRG classifications and the recalibration of the MS-DRG relative weights are made in a manner so that aggregate IPPS payments are unaffected. Therefore, the hospital specific-rate for an SCH or MDH is adjusted by the MS-DRG reclassification and recalibration budget neutrality factor, as discussed in section III. of this Addendum and listed in the table in section II. of this Addendum. In addition, as discussed in section II.E.2.d. of the preamble this final rule and previously, we are applying a permanent 10-percent cap on the reduction in a MS-DRG's relative weight in a given fiscal year, as finalized in the FY 2023 IPPS/LTCH PPS final rule. Because SCHs and MDHs use the same MS-DRGs as other hospitals when they are paid based in whole or in part on the hospital-specific rate, consistent with the policy adopted in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 48897 through 48900 and 49432 through 49433 ), the hospital specific-rate for an SCH or MDH would be adjusted by the MS-DRG 10-percent cap budget neutrality factor. The resulting rate is used in determining the payment rate that an SCH or MDH would receive for its discharges beginning on or after October 1, 2024.
The PPS for acute care hospital inpatient capital-related costs was implemented for cost reporting periods beginning on or after October 1, 1991. The basic methodology for determining Federal capital prospective rates is set forth in the regulations at 42 CFR 412.308 through 412.352 . In this section of this Addendum, we discuss the factors that we used to determine the capital Federal rate for FY 2025, which would be effective for discharges occurring on or after October 1, 2024.
All hospitals (except “new” hospitals under § 412.304(c)(2)) are paid based on the capital Federal rate. We annually update the capital standard Federal rate, as provided in § 412.308(c)(1), to account for capital input price increases and other factors. The regulations at § 412.308(c)(2) also provide that the capital Federal rate be adjusted annually by a factor equal to the estimated proportion of outlier payments under the capital Federal rate to total capital payments under the capital Federal rate. In addition, § 412.308(c)(3) requires that the capital Federal rate be reduced by an adjustment factor equal to the estimated proportion of payments for exceptions under § 412.348. (We note that, as discussed in the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53705 ), there is generally no longer a need for an exceptions payment adjustment factor.) However, in limited circumstances, an additional payment exception for extraordinary circumstances is provided for under § 412.348(f) for qualifying hospitals. Therefore, in accordance with § 412.308(c)(3), an exceptions payment adjustment factor may need to be applied if such payments are made. Section 412.308(c)(4)(ii) requires that the capital standard Federal rate be adjusted so that the effects of the annual DRG reclassification and the recalibration of DRG weights and changes in the geographic adjustment factor (GAF) are budget neutral.
Section 412.374 provides for payments to hospitals located in Puerto Rico under the IPPS for acute care hospital inpatient capital-related costs, which currently specifies capital IPPS payments to hospitals located in Puerto Rico are based on 100 percent of the Federal rate.
In the discussion that follows, we explain the factors that we used to determine the capital Federal rate for FY 2025. In particular, we explain why the FY 2025 capital Federal rate will increase approximately 1.33 percent, compared to the FY 2024 capital Federal rate. As discussed in the impact analysis in Appendix A to this final rule, we estimate that capital payments per discharge will increase approximately 2.8 percent during that same period. Because capital payments constitute approximately 10 percent of hospital payments, a 1-percent change in the capital Federal rate yields only approximately a 0.1 percent change in actual payments to hospitals.
Under § 412.308(c)(1), the capital standard Federal rate is updated on the basis of an analytical framework that takes into account changes in a capital input price index (CIPI) and several other policy adjustment factors. Specifically, we adjust the projected CIPI rate of change, as appropriate, each year for case-mix index-related changes, for intensity, and for errors in previous CIPI forecasts. The update factor for FY 2025 under that framework is 3.1 percent based on a projected 2.6 percent increase in the 2018-based CIPI, a 0.0 percentage point adjustment for intensity, a 0.0 percentage point adjustment for case-mix, a 0.0 percentage point adjustment for the DRG reclassification and recalibration, and a forecast error correction of 0.5 percentage point. As discussed in section III.C. of this Addendum, we continue to believe that the CIPI is the most appropriate input price index for capital costs to measure capital price ( print page 69967) changes in a given year. We also explain the basis for the FY 2025 CIPI projection in that same section of this Addendum. In this final rule, we describe the policy adjustments that we applied in the update framework for FY 2025.
The case-mix index is the measure of the average DRG weight for cases paid under the IPPS. Because the DRG weight determines the prospective payment for each case, any percentage increase in the case-mix index corresponds to an equal percentage increase in hospital payments.
The case-mix index can change for any of several reasons—
- The average resource use of Medicare patient changes (“real” case-mix change);
- Changes in hospital documentation and coding of patient records result in higher-weighted DRG assignments (“coding effects”); or
- The annual DRG reclassification and recalibration changes may not be budget neutral (“reclassification effect”).
We define real case-mix change as actual changes in the mix (and resource requirements) of Medicare patients, as opposed to changes in documentation and coding behavior that result in assignment of cases to higher-weighted DRGs, but do not reflect higher resource requirements. The capital update framework includes the same case-mix index adjustment used in the former operating IPPS update framework (as discussed in the May 18, 2004 IPPS proposed rule for FY 2005 ( 69 FR 28816 )). (We no longer use an update framework to make a recommendation for updating the operating IPPS standardized amounts, as discussed in section II. of appendix B to the FY 2006 IPPS final rule ( 70 FR 47707 ).)
For FY 2025, we are projecting a 0.5 percent total increase in the case-mix index. We estimated that the real case-mix increase would equal 0.5 percent for FY 2025. The net adjustment for change in case-mix is the difference between the projected real increases in case mix and the projected total increase in case mix. Therefore, as proposed, the net adjustment for case-mix change in FY 2025 is 0.0 percentage point.
The capital update framework also contains an adjustment for the effects of DRG reclassification and recalibration. This adjustment is intended to remove the effect on total payments of prior year's changes to the DRG classifications and relative weights, to retain budget neutrality for all case-mix index-related changes other than those due to patient severity of illness. Due to the lag time in the availability of data, there is a 2-year lag in data used to determine the adjustment for the effects of DRG reclassification and recalibration. For example, for this final rule, we have the FY 2023 MedPAR claims data available to evaluate the effects of the FY 2023 DRG reclassification and recalibration as part of our update for FY 2025. We assume for purposes of this adjustment, that the estimate of FY 2023 DRG reclassification and recalibration would result in no change in the case-mix when compared with the case mix index that would have resulted if we had not made the reclassification and recalibration changes to the DRGs. Therefore, as proposed, we are making a 0.0 percentage point adjustment for reclassification and recalibration in the update framework for FY 2025.
The capital update framework also contains an adjustment for forecast error. The input price index forecast is based on historical trends and relationships ascertainable at the time the update factor is established for the upcoming year. In any given year, there may be unanticipated price fluctuations that may result in differences between the actual increase in prices and the forecast used in calculating the update factors. In setting a prospective payment rate under the framework, we make an adjustment for forecast error only if our estimate of the change in the capital input price index for any year is greater than 0.25 percentage point in absolute terms. There is a 2-year lag between the forecast and the availability of data to develop a measurement of the forecast error. Historically, when a forecast error of the CIPI is greater than 0.25 percentage point in absolute terms, it is reflected in the update recommended under this framework. A forecast error of 0.5 percentage point was calculated for the FY 2023 update, for which there are historical data. That is, current historical data indicate that the forecasted FY 2023 CIPI increase (2.5 percent) used in calculating the FY 2023 update factor is 0.5 percentage point lower than actual realized price increases (3.0 percent). As this exceeds the 0.25 percentage point threshold, we are making an adjustment of 0.5 percentage point for the FY 2023 forecast error in the update for FY 2025.
Under the capital IPPS update framework, we also make an adjustment for changes in intensity. Historically, we calculate this adjustment using the same methodology and data that were used in the past under the framework for operating IPPS. The intensity factor for the operating update framework reflects how hospital services are utilized to produce the final product, that is, the discharge. This component accounts for changes in the use of quality-enhancing services, for changes within DRG severity, and for expected modification of practice patterns to remove noncost-effective services. Our intensity measure is based on a 5-year average.
We calculate case-mix constant intensity as the change in total cost per discharge, adjusted for price level changes (the CPI for hospital and related services) and changes in real case-mix. Without reliable estimates of the proportions of the overall annual intensity changes that are due, respectively, to ineffective practice patterns and the combination of quality-enhancing new technologies and complexity within the DRG system, we assume that one-half of the annual change is due to each of these factors. Thus, the capital update framework provides an add-on to the input price index rate of increase of one-half of the estimated annual increase in intensity, to allow for increases within DRG severity and the adoption of quality-enhancing technology.
In this final rule, as proposed, we are continuing to use a Medicare-specific intensity measure that is based on a 5-year adjusted average of cost per discharge for FY 2025 (we refer readers to the FY 2011 IPPS/LTCH PPS final rule ( 75 FR 0436 ) for a full description of our Medicare-specific intensity measure). Specifically, for FY 2025, we are using an intensity measure that is based on an average of cost-per-discharge data from the 5-year period beginning with FY 2018 and extending through FY 2022. Based on these data, we estimated that case-mix constant intensity declined during FYs 2018 through 2022. In the past, when we found intensity to be declining, we believed a zero (rather than a negative) intensity adjustment was appropriate. Consistent with this approach, because we estimated that intensity declined during that 5-year period, we believe it is appropriate to continue to apply a zero-intensity adjustment for FY 2025. Therefore, as proposed, we are making a 0.0 percentage point adjustment for intensity in the update for FY 2025.
Earlier, we described the basis of the components we used to develop the 3.1 percent capital update factor under the capital update framework for FY 2025, as shown in the following table.

Section 412.312(c) establishes a unified outlier payment methodology for inpatient operating and inpatient capital-related costs. A shared threshold is used to identify outlier cases for both inpatient operating and inpatient capital-related payments. Section 412.308(c)(2) provides that the standard Federal rate for inpatient capital-related costs be reduced by an adjustment factor equal to the estimated proportion of capital-related outlier payments to total inpatient capital-related PPS payments. The outlier threshold is set so that operating outlier payments are projected to be 5.1 percent of total operating IPPS DRG payments. For FY 2025, as proposed, we incorporated the impact of estimated operating outlier reconciliation payment amounts into the outlier threshold model. (For more details on our incorporation of the estimated operating outlier reconciliation payment amounts into the outlier threshold model, including modifications to our methodology to reflect the estimate of operating outlier reconciliation payment amounts under the new criteria which expands the scope of cost reports identified for outlier reconciliation approval in FY 2025, see section II.A.4.i. of this Addendum to this final rule.)
For FY 2024, we estimated that outlier payments for capital-related PPS payments will equal 4.02 percent of inpatient capital-related payments based on the capital Federal rate. Based on the threshold discussed in section II.A. of this Addendum, we estimate that prior to taking into account projected capital outlier reconciliation payments, outlier payments for capital-related costs will equal 4.26 percent of inpatient capital-related payments based on the capital Federal rate in FY 2025. Using the methodology outlined in section II.A.4.i. of this Addendum, we estimate that taking into account projected capital outlier reconciliation payments will decrease the estimated percentage of FY 2025 capital outlier payments by 0.03 percent. Therefore, accounting for estimated capital outlier reconciliation, the estimated outlier payments for capital-related PPS payments will equal 4.23 percent (4.26 percent−0.03 percent) of inpatient capital-related payments based on the capital Federal rate in FY 2025. Accordingly, we applied an outlier adjustment factor of 0.9577 in determining the capital Federal rate for FY 2025. Thus, we estimate that the percentage of capital outlier payments to total capital Federal rate payments for FY 2025 will be higher than the percentage we estimated for FY 2024. (For more details on our methodology for incorporating the impact of estimated capital outlier reconciliation payment amounts into the calculation of the capital outlier adjustment factor for FY 2025, including modifications made to our methodology to reflect the estimate of capital outlier reconciliation payment amounts under the new criteria which expands the scope of cost reports identified for outlier reconciliation approval in FY 2025, see section II.A.4.i. of this Addendum to this final rule.)
The outlier reduction factors are not built permanently into the capital rates; that is, they are not applied cumulatively in determining the capital Federal rate. The FY 2025 outlier adjustment of 0.9577 is a −0.22 percent change from the FY 2024 outlier adjustment of 0.9598. Therefore, the net change in the outlier adjustment to the capital Federal rate for FY 2024 is 0.9978 (0.9577/0.9598) so that the outlier adjustment will decrease the FY 2025 capital Federal rate by approximately −0.22 percent compared to the FY 2024 outlier adjustment.
Section 412.308(c)(4)(ii) requires that the capital Federal rate be adjusted so that aggregate payments for the fiscal year based on the capital Federal rate, after any changes resulting from the annual DRG reclassification and recalibration and changes in the GAF, are projected to equal aggregate payments that would have been made on the basis of the capital Federal rate without such changes.
As discussed in section III.G.5. of the preamble of this final rule, in the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42325 through 42339 ), we finalized a policy to help reduce wage index disparities between high and low wage index hospitals by increasing the wage index values for hospitals with a wage index value below the 25th percentile wage index. We stated that this policy would be effective for at least 4 years, beginning in FY 2020. This policy was applied in FYs 2020 through 2024, and we are finalizing our proposal to continue to apply this policy for at least 3 more years, beginning in FY 2025. We note that the FY 2020 low wage index hospital policy and the related budget neutrality adjustment are the subject of pending litigation in multiple courts. On July 23, 2024, the Court of Appeals for the D.C. Circuit held that the Secretary lacked authority under 1886(d)(3)(E) or 1886(d)(5)(I)(i) of the Act to adopt the low wage index hospital policy for FY 2020, and that the policy and related budget neutrality adjustment must be vacated. Bridgeport Hosp. v. Becerra, Nos. 22-5249, 22-5269, 2024 WL 3504407, at *7-*8 n.6 (D.C. Cir. July 23, 2024). As of the date of this Rule's publication, the time to seek further review of the D.C. Circuit's decision in Bridgeport Hospital has not expired. See Fed. R. App. P. 40(a)(1). The government is evaluating the ( print page 69969) decision and considering options for next steps.
In addition, beginning in FY 2023, we finalized a permanent 5-percent cap on any decrease to a hospital's wage index from its wage index in the prior FY regardless of the circumstances causing the decline. That is, under this policy, a hospital's wage index value would not be less than 95 percent of its prior year value ( 87 FR 49018 through 49021 ).
We have established a 2-step methodology for computing the budget neutrality factor for changes in the GAFs in light of the effect of those wage index changes on the GAFs. In the first step, we first calculate a factor to ensure budget neutrality for changes to the GAFs due to the update to the wage data, wage index reclassifications and redesignations, and application of the rural floor policy, consistent with our historical GAF budget neutrality factor methodology. In the second step, we calculate a factor to ensure budget neutrality for changes to the GAFs due to our policy to increase the wage index for hospitals with a wage index value below the 25th percentile wage index, which will continue in FY 2025, and our policy to place a 5-percent cap on any decrease in a hospital's wage index from the hospital's final wage index in the prior fiscal year. In this section, we refer to the policy that we applied in FYs 2020 through FY 2024 and continue to apply in FY 2025, of increasing the wage index for hospitals with a wage index value below the 25th percentile wage index, as the lowest quartile hospital wage index adjustment (also known as low wage index hospital policy). We refer to our policy to place a 5-percent cap on any decrease in a hospital's wage index from the hospital's final wage index in the prior fiscal year as the 5-percent cap on wage index decreases policy.
The budget neutrality factors applied for changes to the GAFs due to the update to the wage data, wage index reclassifications and redesignations, and application of the rural floor policy are built permanently into the capital Federal rate; that is, they are applied cumulatively in determining the capital Federal rate. However, the budget neutrality factor for the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy is not permanently built into the capital Federal rate. This is because the GAFs with the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy applied from the previous year are not used in the budget neutrality factor calculations for the current year. Accordingly, and consistent with this approach, prior to calculating the GAF budget neutrality factors for FY 2025, we removed from the capital Federal rate the budget neutrality factor applied in FY 2024 for the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy. Specifically, we divided the capital Federal rate by the FY 2024 budget neutrality factor of 0.9964 ( 88 FR 59362 ). We refer the reader to the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45552 ) for additional discussion on our policy of removing the prior year budget neutrality factor for the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases from the capital Federal rate.
In light of the changes to the wage index and other wage index policies for FY 2025 discussed previously, which directly affect the GAF, we continue to compute a budget neutrality adjustment for changes in the GAFs in two steps. We discuss our 2-step calculation of the GAF budget neutrality factors for FY 2025 as follows.
To determine the GAF budget neutrality factors for FY 2025, we first compared estimated aggregate capital Federal rate payments based on the FY 2024 MS-DRG classifications and relative weights and the FY 2024 GAFs to estimated aggregate capital Federal rate payments based on the FY 2024 MS-DRG classifications and relative weights and the FY 2025 GAFs without incorporating the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy. To achieve budget neutrality for these changes in the GAFs, we calculated an incremental GAF budget neutrality adjustment factor of 0.9887 for FY 2025. Next, we compared estimated aggregate capital Federal rate payments based on the FY 2025 GAFs with and without the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy. For this calculation, estimated aggregate capital Federal rate payments were calculated using the FY 2025 MS-DRG classifications and relative weights (after application of the 10-percent cap discussed later in this section) and the FY 2025 GAFs (both with and without the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy). (We note, for this calculation the GAFs included the imputed floor, out-migration, and Frontier state adjustments.) To achieve budget neutrality for the effects of the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy on the FY 2025 GAFs, we calculated an incremental GAF budget neutrality adjustment factor of 0.9958. As discussed earlier in this section, the budget neutrality factor for the lowest quartile hospital wage index adjustment factor and the 5-percent cap on wage index decreases policy is not permanently built into the capital Federal rate. Consistent with this, we present the budget neutrality factor for the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy calculated under the second step of this 2-step methodology separately from the other budget neutrality factors in the discussion that follows, and this factor is not included in the calculation of the combined GAF/DRG adjustment factor described later in this section.
In the FY 2023 IPPS/LTCH PPS final rule, we finalized a permanent 10-percent cap on the reduction in an MS-DRG's relative weight in a given fiscal year, beginning in FY 2023. Consistent with our historical methodology for adjusting the capital standard Federal rate to ensure that the effects of the annual DRG reclassification and the recalibration of DRG weights are budget neutral under § 412.308(c)(4)(ii), we finalized to apply an additional budget neutrality factor to the capital standard Federal rate so that the 10-percent cap on decreases in an MS-DRG's relative weight is implemented in a budget neutral manner ( 87 FR 49436 ). Specifically, we augmented our historical methodology for computing the budget neutrality factor for the annual DRG reclassification and recalibration by computing a budget neutrality adjustment for the annual DRG reclassification and recalibration in two steps. We first calculate a budget neutrality factor to account for the annual DRG reclassification and recalibration prior to the application of the 10-percent cap on MS-DRG relative weight decreases. Then we calculate an additional budget neutrality factor to account for the application of the 10-percent cap on MS-DRG relative weight decreases.
To determine the DRG budget neutrality factors for FY 2025, we first compared estimated aggregate capital Federal rate payments based on the FY 2024 MS-DRG classifications and relative weights to estimated aggregate capital Federal rate payments based on the FY 2025 MS-DRG classifications and relative weights prior to the application of the 10-percent cap. For these calculations, estimated aggregate capital Federal rate payments were calculated using the FY 2025 GAFs without the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases ( print page 69970) policy. The incremental adjustment factor for DRG classifications and changes in relative weights prior to the application of the 10-percent cap is 0.9970. Next, we compared estimated aggregate capital Federal rate payments based on the FY 2025 MS-DRG classifications and relative weights prior to the application of the 10-percent cap to estimated aggregate capital Federal rate payments based on the FY 2025 MS-DRG classifications and relative weights after the application of the 10-percent cap. For these calculations, estimated aggregate capital Federal rate payments were also calculated using the FY 2025 GAFs without the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy. The incremental adjustment factor for the application of the 10-percent cap on relative weight decreases is 0.9999. Therefore, to achieve budget neutrality for the FY 2025 MS-DRG reclassification and recalibration (including the 10-percent cap), based on the calculations described previously, we are applying an incremental budget neutrality adjustment factor of 0.9969 (0.9970 × 0.9999) for FY 2025 to the capital Federal rate. We note that all the values are calculated with unrounded numbers.
The incremental adjustment factor for the FY 2025 MS-DRG reclassification and recalibration (0.9969) and for changes in the FY 2025 GAFs due to the update to the wage data, wage index reclassifications and redesignations, and application of the rural floor policy (0.9887) is 0.9856 (0.9969 x 0.9887). This incremental adjustment factor is built permanently into the capital Federal rates. To achieve budget neutrality for the effects of the continuation of the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy on the FY 2025 GAFs, as described previously, we calculated a budget neutrality adjustment factor of 0.9958 for FY 2025. We refer to this budget neutrality factor for the remainder of this section as the lowest quartile/cap adjustment factor.
We applied the budget neutrality adjustment factors described previously to the capital Federal rate. This follows the requirement under § 412.308(c)(4)(ii) that estimated aggregate payments each year be no more or less than they would have been in the absence of the annual DRG reclassification and recalibration and changes in the GAFs.
The methodology used to determine the recalibration and geographic adjustment factor (GAF/DRG) budget neutrality adjustment is similar to the methodology used in establishing budget neutrality adjustments under the IPPS for operating costs. One difference is that, under the operating IPPS, the budget neutrality adjustments for the effect of updates to the wage data, wage index reclassifications and redesignations, and application of the rural floor policy are determined separately. Under the capital IPPS, there is a single budget neutrality adjustment factor for changes in the GAF that result from updates to the wage data, wage index reclassifications and redesignations, and application of the rural floor policy. In addition, there is no adjustment for the effects that geographic reclassification, the lowest quartile hospital wage index adjustment, or the 5-percent cap on wage index decreases policy described previously have on the other payment parameters, such as the payments for DSH or IME.
The incremental GAF/DRG adjustment factor of 0.9856 accounts for the MS-DRG reclassifications and recalibration (including application of the 10-percent cap on relative weight decreases) and for changes in the GAFs that result from updates to the wage data, the effects on the GAFs of FY 2025 geographic reclassification decisions made by the MGCRB compared to FY 2024 decisions, and the application of the rural floor policy. The lowest quartile/cap adjustment factor of 0.9958 accounts for changes in the GAFs that result from our continuation of the policy to increase the wage index values for hospitals with a wage index value below the 25th percentile wage index and the 5-percent cap on wage index decreases policy. However, these factors do not account for changes in payments due to changes in the DSH and IME adjustment factors.
For FY 2024, we established a capital Federal rate of $503.83 ( 88 FR 59363 ). We are establishing an update of 3.1 percent in determining the FY 2025 capital Federal rate for all hospitals. As a result of this update and the budget neutrality factors discussed earlier, we are establishing a national capital Federal rate of $510.51 for FY 2025. The national capital Federal rate for FY 2025 was calculated as follows:
- The FY 2025 update factor is 1.031; that is, the update is 3.1 percent.
- The FY 2025 GAF/DRG budget neutrality adjustment factor that is applied to the capital Federal rate for changes in the MS-DRG classifications and relative weights (including application of the 10-percent cap on relative weight decreases) and changes in the GAFs that result from updates to the wage data, wage index reclassifications and redesignations, and application of the rural floor policy is 0.9856.
- The FY 2025 lowest quartile/cap budget neutrality adjustment factor that is applied to the capital Federal rate for changes in the GAFs that result from our policy to increase the wage index values for hospitals with a wage index value below the 25th percentile wage index and the 5-percent cap on wage index decreases policy is 0.9958.
- The FY 2025 outlier adjustment factor is 0.9577.
We are providing the following chart that shows how each of the factors and adjustments for FY 2025 affects the computation of the FY 2025 national capital Federal rate in comparison to the FY 2024 national capital Federal rate. The FY 2025 update factor has the effect of increasing the capital Federal rate by 3.1 percent compared to the FY 2024 capital Federal rate. The GAF/DRG budget neutrality adjustment factor has the effect of decreasing the capital Federal rate by 1.44 percent. The FY 2025 lowest quartile/cap budget neutrality adjustment factor has the effect of decreasing the capital Federal rate by 0.07 percent compared to the FY 2024 capital Federal rate. The FY 2025 outlier adjustment factor has the effect of decreasing the capital Federal rate by 0.22 percent compared to the FY 2024 capital Federal rate. The combined effect of all the changes would increase the national capital Federal rate by approximately 1.33 percent, compared to the FY 2024 national capital Federal rate.

For purposes of calculating payments for each discharge during FY 2025, the capital Federal rate is adjusted as follows: (Standard Federal Rate) × (DRG weight) × (GAF) × (COLA for hospitals located in Alaska and Hawaii) × (1 + DSH Adjustment Factor + IME Adjustment Factor, if applicable). The result is the adjusted capital Federal rate.
Hospitals also may receive outlier payments for those cases that qualify under the threshold established for each fiscal year. Section 412.312(c) provides for a shared threshold to identify outlier cases for both inpatient operating and inpatient capital-related payments. The outlier threshold for FY 2025 is in section II.A. of this Addendum. For FY 2025, a case will qualify as a cost outlier if the cost for the case is greater than the prospective payment rates for the MS-DRG plus IME and DSH payments (including the empirically justified Medicare DSH payment and the estimated uncompensated care payment), estimated supplemental payment for eligible IHS/Tribal hospitals and Puerto Rico hospitals, and any add-on payments for new technology, plus the fixed-loss amount of $46,152.
Currently, as provided under § 412.304(c)(2), we pay a new hospital 85 percent of its reasonable costs during the first 2 years of operation, unless it elects to receive payment based on 100 percent of the capital Federal rate. Effective with the third year of operation, we pay the hospital based on 100 percent of the capital Federal rate (that is, the same methodology used to pay all other hospitals subject to the capital PPS).
Like the operating input price index, the capital input price index (CIPI) is a fixed-weight price index that measures the price changes associated with capital costs during a given year. The CIPI differs from the operating input price index in one important aspect—the CIPI reflects the vintage nature of capital, which is the acquisition and use of capital over time. Capital expenses in any given year are determined by the stock of capital in that year (that is, capital that remains on hand from all current and prior capital acquisitions). An index measuring capital price changes needs to reflect this vintage nature of capital. Therefore, the CIPI was developed to capture the vintage nature of capital by using a weighted-average of past capital purchase prices up to and including the current year.
For this final rule, we are using the IPPS operating and capital market baskets that reflect a 2018 base year. For a complete discussion of the 2018-based market baskets, we refer readers to section IV. of the preamble of the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45194 through 45213 ).
Based on IHS Global Inc.'s second quarter 2024 forecast, for this final rule, we are forecasting the 2018-based CIPI to increase 2.6 percent in FY 2025. This reflects a projected 3.1 percent increase in vintage-weighted depreciation prices (building and fixed equipment, and movable equipment), and a projected 4.2 percent increase in other capital expense prices in FY 2025, partially offset by a projected 1.5 percent decline in vintage-weighted interest expense prices in FY 2025. The weighted average of these three factors produces the forecasted 2.6 percent increase for the 2018-based CIPI in FY 2025. As proposed, we are using the more recent data available for this final rule to determine the FY 2025 increase in the 2018-based CIPI for this final rule.
Payments for services furnished in children's hospitals, 11 cancer hospitals, and hospitals located outside the 50 States, the District of Columbia and Puerto Rico (that is, short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa) that are excluded from the IPPS are paid on the basis of reasonable costs based on the hospital's own historical cost experience, subject to a rate-of-increase ceiling. A per discharge limit (the target amount, as defined in § 413.40(a) of the regulations) is set for each hospital, based on the hospital's own cost experience in its base year, and updated annually by a rate-of-increase percentage specified in § 413.40(c)(3). In addition, as specified in the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38536 ), effective for cost reporting periods beginning during FY 2018, the annual update to the target amount for extended neoplastic disease care hospitals (hospitals described in § 412.22(i) of the regulations) also is the rate-of-increase percentage specified in § 413.40(c)(3). (We note that, in accordance with § 403.752(a), religious nonmedical health care institutions (RNHCIs) are also subject to the rate-of- ( print page 69972) increase limits established under § 413.40 of the regulations.)
For the FY 2025 IPPS/LTCH PPS proposed rule, based on IGI's 2023 fourth quarter forecast, we estimated that the 2018-based IPPS operating market basket percentage increase for FY 2025 would be 3.0 percent (that is, the estimate of the market basket rate-of-increase). Based on this estimate, the FY 2025 rate-of-increase percentage that we proposed to apply to the FY 2024 target amounts to calculate the FY 2025 target amounts for children's hospitals, the 11 cancer hospitals, RNCHIs, and short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa was 3.0 percent, in accordance with the applicable regulations at 42 CFR 413.40 . However, we proposed that if more recent data became available for the FY 2025 IPPS/LTCH PPS final rule, we would use such data, if appropriate, to calculate the final IPPS operating market basket update for FY 2025. Based on more recent data available (IGI's second quarter 2024 forecast), we estimate that the 2018-based IPPS operating market basket percentage increase for FY 2025 is 3.4 percent (that is, the estimate of the market basket rate-of-increase). Based on this estimate, the FY 2025 rate-of-increase percentage that we will apply to the FY 2024 target amounts to calculate the FY 2025 target amounts for children's hospitals, the 11 cancer hospitals, RNCHIs, and short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa is 3.4 percent, in accordance with the applicable regulations at 42 CFR 413.40 .
IRFs and rehabilitation distinct part units, IPFs and psychiatric units, and LTCHs are excluded from the IPPS and paid under their respective PPSs. The IRF PPS, the IPF PPS, and the LTCH PPS are updated annually. We refer readers to section VIII. of the preamble and section V. of the Addendum of this final rule for the changes to the Federal payment rates for LTCHs under the LTCH PPS for FY 2025. The annual updates for the IRF PPS and the IPF PPS are issued by the agency in separate Federal Register documents.
We received no comments on this proposal and therefore are finalizing this provision without modification. Incorporating more recent data available for this final rule, as we proposed, we are adopting a 3.4 percent update for FY 2025.
In section VIII. of the preamble of this final rule, we discuss our annual updates to the payment rates, factors, and specific policies under the LTCH PPS for FY 2025.
Under § 412.523(c)(3) of the regulations, for FY 2012 and subsequent years, we updated the standard Federal payment rate by the most recent estimate of the LTCH PPS market basket at that time, including additional statutory adjustments required by sections 1886(m)(3) (citing sections 1886(b)(3)(B)(xi)(II) and 1886(m)(4) of the Act as set forth in the regulations at § 412.523(c)(3)(viii) through (xvii)). (For a summary of the payment rate development prior to FY 2012, we refer readers to the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38310 through 38312 ) and references therein.)
Section 1886(m)(3)(A) of the Act specifies that, for rate year 2012 and each subsequent rate year, any annual update to the standard Federal payment rate shall be reduced by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act as discussed in section VIII.C.2. of the preamble of this final rule. This section of the Act further provides that the application of section 1886(m)(3)(B) of the Act may result in the annual update being less than zero for a rate year, and may result in payment rates for a rate year being less than such payment rates for the preceding rate year. (As noted in section VIII.C.2. of the preamble of this final rule, the annual update to the LTCH PPS occurs on October 1 and we have adopted the term “fiscal year” (FY) rather than “rate year” (RY) under the LTCH PPS beginning October 1, 2010. Therefore, for purposes of clarity, when discussing the annual update for the LTCH PPS, including the provisions of the Affordable Care Act, we use the term “fiscal year” rather than “rate year” for 2011 and subsequent years.)
For LTCHs that fail to submit the required quality reporting data in accordance with the LTCH QRP, the annual update is reduced by 2.0 percentage points as required by section 1886(m)(5) of the Act.
Consistent with our historical practice and § 412.523(c)(3)(xvii), for FY 2025, as we proposed, we are applying the annual update to the LTCH PPS standard Federal payment rate from the previous year. Furthermore, in determining the LTCH PPS standard Federal payment rate for FY 2025, we also are making certain regulatory adjustments, consistent with past practices. Specifically, in determining the FY 2025 LTCH PPS standard Federal payment rate, as we proposed, we are applying a budget neutrality adjustment factor for the changes related to the area wage level adjustment (that is, changes to the wage data and labor-related share) as discussed in section V.B.6. of this Addendum.
In this final rule, we are establishing an annual update to the LTCH PPS standard Federal payment rate of 3.0 percent (that is, the most recent estimate of the 2022-based LTCH market basket increase of 3.5 percent less the productivity adjustment of 0.5 percentage point). Therefore, in accordance with § 412.523(c)(3)(xvii), we are applying an update factor of 1.030 to the FY 2024 LTCH PPS standard Federal payment rate of $48,116.62 to determine the FY 2025 LTCH PPS standard Federal payment rate. Also, in accordance with § 412.523(c)(3)(xvii) and (c)(4), we are required to reduce the annual update to the LTCH PPS standard Federal payment rate by 2.0 percentage points for LTCHs that fail to submit the required quality reporting data for FY 2025 as required under the LTCH QRP. Therefore, for LTCHs that fail to submit quality reporting data under the LTCH QRP, we are establishing an annual update to the LTCH PPS standard Federal payment rate of 1.0 percent (or an update factor of 1.010). This update reflects the annual market basket update of 3.5 percent reduced by the 0.5 percentage point productivity adjustment, as required by section 1886(m)(3)(A)(i) of the Act, minus 2.0 percentage points for LTCHs failing to submit quality data under the LTCH QRP, as required by section 1886(m)(5) of the Act. Consistent with § 412.523(d)(4), we are applying an area wage level budget neutrality factor to the FY 2025 LTCH PPS standard Federal payment rate of 0.9964315, based on the best available data at this time, to ensure that any changes to the area wage level adjustment (that is, the annual update of the wage index (including the update to the CBSA labor market areas and the application of the 5-percent cap on wage index decreases, discussed later in this section), and labor-related share) will not result in any change (increase or decrease) in estimated aggregate LTCH PPS standard Federal payment rate payments. Accordingly, we are ( print page 69973) establishing an LTCH PPS standard Federal payment rate of $49,383.26 (calculated as $48,116.62 × 1.030 × 0.9964315) for FY 2025. For LTCHs that fail to submit quality reporting data for FY 2025, in accordance with the requirements of the LTCH QRP under section 1866(m)(5) of the Act, we are establishing an LTCH PPS standard Federal payment rate of $48,424.36 (calculated as $48,116.62 × 1.010 × 0.9964315) for FY 2025.
Under the authority of section 123 of the BBRA, as amended by section 307(b) of the BIPA, we established an adjustment to the LTCH PPS standard Federal payment rate to account for differences in LTCH area wage levels under § 412.525(c). The labor-related share of the LTCH PPS standard Federal payment rate is adjusted to account for geographic differences in area wage levels by applying the applicable LTCH PPS wage index. The applicable LTCH PPS wage index is computed using wage data from inpatient acute care hospitals without regard to reclassification under section 1886(d)(8) or section 1886(d)(10) of the Act.
The FY 2025 LTCH PPS standard Federal payment rate wage index values that will be applicable for LTCH PPS standard Federal payment rate discharges occurring on or after October 1, 2024, through September 30, 2025, are presented in Table 12A (for urban areas) and Table 12B (for rural areas), which are listed in section VI. of this Addendum and available via the internet on the CMS website.
In adjusting for the differences in area wage levels under the LTCH PPS, the labor-related portion of an LTCH's Federal prospective payment is adjusted by using an appropriate area wage index based on the geographic classification (labor market area) in which the LTCH is located. Specifically, the application of the LTCH PPS area wage level adjustment under existing § 412.525(c) is made based on the location of the LTCH—either in an “urban area,” or a “rural area,” as defined in § 412.503. Under § 412.503, an “urban area” is defined as a Metropolitan Statistical Area (MSA) (which includes a Metropolitan division, where applicable), as defined by the Executive OMB, and a “rural area” is defined as any area outside of an urban area ( 75 FR 37246 ).
The geographic classifications (labor market area definitions) currently used under the LTCH PPS, effective for discharges occurring on or after October 1, 2014, are based on the Core Based Statistical Areas (CBSAs) established by OMB, which are based on the 2010 decennial census data. In general, the current statistical areas (which were implemented beginning with FY 2015) are based on revised OMB delineations issued on February 28, 2013, in OMB Bulletin No. 13-01. (We note we have adopted minor revisions and updates in the years between the decennial censuses.) We adopted these labor market area delineations because they were at that time based on the best available data that reflect the local economies and area wage levels of the hospitals that are currently located in these geographic areas. We also believed that these OMB delineations would ensure that the LTCH PPS area wage level adjustment most appropriately accounted for and reflected the relative hospital wage levels in the geographic area of the hospital as compared to the national average hospital wage level. We noted that this policy was consistent with the IPPS policy adopted in FY 2015 under § 412.64(b)(1)(ii)(D) ( 79 FR 49951 through 49963 ). (For additional information on the CBSA-based labor market area (geographic classification) delineations currently used under the LTCH PPS and the history of the labor market area definitions used under the LTCH PPS, we refer readers to the FY 2015 IPPS/LTCH PPS final rule ( 79 FR 50180 through 50185 ).)
In general, it is our historical practice to update the CBSA-based labor market area delineations annually based on the most recent updates issued by OMB. Generally, OMB issues major revisions to statistical areas every 10 years, based on the results of the decennial census. However, OMB occasionally issues minor updates and revisions to statistical areas in the years between the decennial censuses. OMB Bulletin No. 17-01, issued August 15, 2017, established the delineations for the Nation's statistical areas, and the corresponding changes to the CBSA-based labor market areas were adopted in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41731 ). A copy of this bulletin may be obtained on the website at: https://www.whitehouse.gov/wp-content/uploads/legacy_drupal_files/omb/bulletins/2017/b-17-01.pdf .
On April 10, 2018, OMB issued OMB Bulletin No. 18-03, which superseded OMB Bulletin No. 17-01 (August 15, 2017). On September 14, 2018, OMB issued OMB Bulletin No. 18-04, which superseded OMB Bulletin No. 18-03 (April 10, 2018). Historically OMB bulletins issued between decennial censuses have only contained minor modifications to CBSA delineations based on changes in population counts. However, OMB's 2010 Standards for Delineating Metropolitan and Micropolitan Standards created a larger mid-decade redelineation that takes into account commuting data from the American Commuting Survey. As a result, OMB Bulletin No. 18-04 (September 14, 2018) included more modifications to the CBSAs than are typical for OMB bulletins issued between decennial censuses. We adopted the updates set forth in OMB Bulletin No. 18-04 in the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 59050 through 59051 ). A copy of OMB Bulletin No. 18-04 (September 14, 2018) may be obtained at https://www.whitehouse.gov/wp-content/uploads/2018/09/Bulletin-18-04.pdf .
On March 6, 2020, OMB issued Bulletin No. 20-01, which provided updates to and superseded OMB Bulletin No. 18-04, which was issued on September 14, 2018. The attachments to OMB Bulletin No. 20-01 provided detailed information on the update to statistical areas since September 14, 2018. (For a copy of this bulletin, we refer readers to the following website: https://www.whitehouse.gov/wp-content/uploads/2020/03/Bulletin-20-01.pdf .) In OMB Bulletin No. 20-01, OMB announced one new Micropolitan Statistical Area and one new component of an existing Combined Statistical Area. After reviewing OMB Bulletin No. 20-01, we determined that the changes in OMB Bulletin 20-01 encompassed delineation changes that would not affect the CBSA-based labor market area delineations used under the LTCH PPS. Therefore, we adopted the updates set forth in OMB Bulletin No. 20-01 in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45556 through 45557 ) consistent with our general policy of adopting OMB delineation updates; however, the LTCH PPS area wage level adjustment was not altered as a result of adopting the updates because the CBSA-based labor market area delineations were the same as the CBSA-based labor market area delineations adopted in the FY 2021 IPPS/LTCH PPS final rule based on OMB Bulletin No. 18-04 ( 85 FR 59050 through 59051 ). Thus, most recently in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59366 ), we continued to use the CBSA-based labor market area delineations as established in OMB Bulletin 18-04 and OMB Bulletin 20-01.
In the July 16, 2021 Federal Register ( 86 FR 37777 ), OMB finalized a ( print page 69974) schedule for future updates based on results of the decennial Census updates to commuting patterns from the American Community Survey. In accordance with that schedule, on July 21, 2023, OMB released Bulletin No. 23-01, which superseded OMB Bulletin No. 20-01. A copy of OMB Bulletin No. 23-01 may be obtained at https://www.whitehouse.gov/wp-content/uploads/2023/07/OMB-Bulletin-23-01.pdf . According to OMB, the delineations reflect the 2020 Standards for Delineating Core Based Statistical Areas (“the 2020 Standards”), which appeared in the Federal Register on July 16, 2021 ( 86 FR 37770 through 37778 ), and the application of those standards to Census Bureau population and journey-to-work data (that is, 2020 Decennial Census, American Community Survey, and Census Population Estimates Program data). In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36584 through 36586 ), we proposed to adopt the revised delineations announced in OMB Bulletin No. 23-01 effective for FY 2025 under the LTCH PPS. We did not receive any public comments on this proposal. Therefore, in this final rule, under the authority of section 123 of the BBRA, as amended by section 307(b) of the BIPA, we are adopting the revised delineations announced in OMB Bulletin No. 23-01 effective for FY 2025 under the LTCH PPS, as we proposed, without modification. We believe that adopting the CBSA-based labor market area delineations established in OMB Bulletin 23-01 will ensure that the LTCH PPS area wage level adjustment most appropriately accounts for and reflects the relative hospital wage levels in the geographic area of the hospital as compared to the national average hospital wage level based on the best available data that reflect the local economies and area wage levels of the hospitals that are currently located in these geographic areas ( 81 FR 57298 ). Our adoption of the revised delineations announced in OMB Bulletin No. 23-01 is consistent with the changes under the IPPS for FY 2025 as discussed in section III.B. of the preamble of this final rule. A summary of these changes is presented in the discussion that follows in this section. For complete details on the changes, we refer readers to section III.B. of the preamble of this final rule.
CBSAs are made up of one or more constituent counties. Analysis of the revised labor market area delineations (based upon OMB Bulletin No. 23-01) that we are adopting, beginning in FY 2025, shows that a total of 53 counties (and county equivalents) that were located in an urban CBSA pursuant to OMB Bulletin No. 20-01 will be located in a rural area under the revised OMB delineations. The chart in section III.B.4. of the preamble of this final rule lists the 53 urban counties that will be rural under these revised OMB delineations.
Analysis of the revised labor market area delineations (based upon OMB Bulletin No. 23-01) that we are adopting, beginning in FY 2025, shows that a total of 54 counties (and county equivalents) that were located in a rural area pursuant to OMB Bulletin No. 20-01 will be located in an urban CBSA under the revised OMB delineations. The chart in section III.B.5. of the preamble of this final rule lists the 54 rural counties that will be urban under these revised OMB delineations.
In addition to rural counties becoming urban and urban counties becoming rural, some urban counties will shift from one urban CBSA to another urban CBSA under our adoption of the revised delineations announced in OMB Bulletin No. 23-01. In other cases, the adoption of the revised delineations announced in OMB Bulletin No. 23-01 will involve a change only in CBSA name and/or number, while the CBSA continues to encompass the same constituent counties. For example, CBSA 23844 (Gary, IN) will experience both a change to its number and its name and become CBSA 29414 (Lake County-Porter County-Jasper County, IN), while all of its four constituent counties will remain the same. In other cases, only the name of the CBSA will be modified, and none of the currently assigned counties will be reassigned to a different urban CBSA. The chart in section III.B.6. of the preamble of this final rule lists the CBSAs where only the name and/or CBSA number changed.
There are also counties that will shift between existing and new CBSAs, changing the constituent makeup of the CBSAs, under our adoption of the revisions to the OMB delineations based on OMB Bulletin No. 23-01. For example, some CBSAs will be split into multiple new CBSAs, or a CBSA will lose one or more counties to other urban CBSAs. The chart in section III.B.6 of the preamble of this final rule lists the urban counties that will move from one urban CBSA to a new or modified CBSA under our adoption of these revisions to the OMB delineations.
For FY 2025, we are continuing to use the Federal Information Processing Standard (FIPS) county codes, maintained by the U.S. Census Bureau, for purposes of cross walking counties to CBSAs. In a June 6, 2022 Federal Register notice ( 87 FR 34235 through 34240 ), the Census Bureau announced that it was implementing the State of Connecticut's request to replace the 8 counties in the State with 9 new “Planning Regions.” Planning regions now serve as county-equivalents within the CBSA system. OMB Bulletin No. 23-01 is the first set of revised delineations that referenced the new county-equivalents for Connecticut. For the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36585 ), we evaluated the change in hospital assignments for Connecticut LTCHs and proposed to adopt the planning regions as county equivalents for wage index purposes. As all forthcoming county-based delineation data will utilize these new county-equivalent definitions for the Connecticut, we believe it is necessary to adopt this migration from counties to planning region county-equivalents in order to maintain consistency with OMB Bulletin No. 23-01 and future OMB updates. We did not receive any public comments on this proposal. Therefore, in this final rule, we are adopting our proposal to adopt the planning regions as county equivalents for wage index purposes, without modification. Our adoption of the planning regions as county equivalents for wage index purposes is consistent with the changes under the IPPS for FY 2025 as discussed in section III.B.3. of the preamble of this final rule. We are providing the following crosswalk for each LTCH in Connecticut with the current (FY 2024) and new (FY 2025) FIPS county and county-equivalent codes and CBSA assignments.

As previously discussed, we are adopting the revisions announced in OMB Bulletin No. 23-01 to the CBSA-based labor market area delineations under the LTCH PPS, effective October 1, 2024. Accordingly, the FY 2025 LTCH PPS wage index values in Tables 12A and 12B listed in section VI. of the Addendum to this final rule (which are available via the internet on the CMS website) reflect the revisions to the CBSA-based labor market area delineations previously described. We also are including in a supplemental data file an updated county-to-CBSA crosswalk that reflects the revisions to the CBSA-based labor market area delineations. This supplemental data file for public use will be posted on the CMS website for this final rule at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html .
Under the payment adjustment for the differences in area wage levels under § 412.525(c), the labor-related share of an LTCH's standard Federal payment rate is adjusted by the applicable wage index for the labor market area in which the LTCH is located. The LTCH PPS labor-related share currently represents the sum of the labor-related portion of operating costs and a labor-related portion of capital costs using the applicable LTCH market basket. Additional background information on the historical development of the labor-related share under the LTCH PPS can be found in the RY 2007 LTCH PPS final rule ( 71 FR 27810 through 27817 and 27829 through 27830 ) and the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51766 through 51769 and 51808 ).
For FY 2013, we rebased and revised the market basket used under the LTCH PPS by adopting a 2009-based LTCH market basket. In addition, for FY 2013 through FY 2016, we determined the labor-related share annually as the sum of the relative importance of each labor-related cost category of the 2009-based LTCH market basket for the respective fiscal year based on the best available data. (For more details, we refer readers to the FY 2013 IPPS/LTCH PPS final rule ( 77 FR 53477 through 53479 ).) For FY 2017, we rebased and revised the 2009-based LTCH market basket to reflect a 2013 base year. In addition, for FY 2017 through FY 2020, we determined the labor-related share annually as the sum of the relative importance of each labor-related cost category of the 2013-based LTCH market basket for the respective fiscal year based on the best available data. (For more details, we refer readers to the FY 2017 IPPS/LTCH PPS final rule ( 81 FR 57085 through 57096 ).) Then, effective for FY 2021, we rebased and revised the 2013-based LTCH market basket to reflect a 2017 base year and determined the labor-related share annually as the sum of the relative importance of each labor-related cost category in the 2017-based LTCH market basket using the most recent available data. (For more details, we refer readers to the FY 2021 IPPS/LTCH PPS final rule ( 85 FR 58909 through 58926 ).)
As discussed in section VIII.D of the preamble to this final rule, effective for FY 2025, as we proposed, we are rebasing and revising the 2017-based LTCH market basket to reflect a 2022 base year. In addition, as discussed in section VIII.D. of the preamble of this final rule, as we proposed, we are establishing that the LTCH PPS labor-related share for FY 2025 is the sum of the FY 2025 relative importance of each labor-related cost category in the 2022-based LTCH market basket using the most recent available data. For more information on comments related to our proposed labor-related share based on the labor-related cost categories in the 2022-based LTCH market basket as well as our responses to those comments, we refer readers to section VIII.D of the preamble of this final rule. Also, as we proposed, consistent with our historical practice, we are using the most recent data available to determine the final FY 2025 labor-related share in this final rule.
Table EEEE9 in section VIII.D. of the preamble of this final rule shows the FY 2025 labor-related share using the 2022-based LTCH market basket and the FY 2024 labor-related share using the 2017-based LTCH market basket. The labor-related share for FY 2025 is the sum of the labor-related portion of operating costs from the 2022-based LTCH market basket (that is, the sum of the FY 2025 relative importance shares of Wages and Salaries; Employee Benefits; Professional Fees: Labor-Related; Administrative and Facilities Support Services; Installation, Maintenance, and Repair Services; All Other: Labor-Related Services) and a portion of the relative importance of Capital-Related cost weight from the 2022-based LTCH market basket. The relative importance reflects the different rates of price change for these cost categories between the base year (2022) and FY 2025. Based on IHS Global Inc.'s second quarter 2024 forecast of the 2022-based LTCH market basket, the sum of the FY 2025 relative importance for Wages and Salaries; Employee Benefits; Professional Fees: Labor-Related; Administrative and Facilities Support Services; Installation, Maintenance, and Repair Services; and All Other: Labor-Related Services is 68.9 percent. The portion of capital-related costs that is influenced by the local labor market is estimated to be 46 percent (that is, the same percentage applied to the 2009-based, 2013-based, and 2017-based LTCH market basket capital-related costs relative importance). Since the FY 2025 relative importance for capital-related costs is 8.4 percent based on IHS Global Inc.'s second quarter 2024 forecast of the 2022-based LTCH market basket, we took 46 percent of 8.4 percent to determine the labor-related share of capital-related costs for FY 2025 of 3.9 percent. Therefore, we are finalizing a total labor-related share for FY 2025 of 72.8 percent (the sum of 68.9 percent for the labor-related share of operating costs and 3.9 percent for the labor-related share of capital-related costs). The total difference between the FY 2025 labor-related share using the 2022 based LTCH market basket (72.8 percent) and the FY 2024 labor-related share using the 2017 based LTCH market basket (68.5 percent) is 4.3 percentage points. As discussed in greater detail in section VIII.D. of the preamble of this final rule, this difference is primarily attributable to the revision to the base year cost weights for those categories included in the labor-related share.
Historically, we have established LTCH PPS area wage index values calculated from acute care IPPS hospital wage data without taking into account geographic reclassification under sections 1886(d)(8) and 1886(d)(10) of the Act ( 67 FR 56019 ). The area wage level adjustment established under the ( print page 69976) LTCH PPS is based on an LTCH's actual location without regard to the “urban” or “rural” designation of any related or affiliated provider. As with the IPPS wage index, wage data for multicampus hospitals with campuses located in different labor market areas (CBSAs) are apportioned to each CBSA where the campus (or campuses) are located. We also employ a policy for determining area wage index values for areas where there are no IPPS wage data.
Consistent with our historical methodology, to determine the applicable area wage index values for the FY 2025 LTCH PPS standard Federal payment rate, under the broad authority of section 123 of the BBRA, as amended by section 307(b) of the BIPA, as we proposed, we are continuing to employ our historical practice of using the same data we used to compute the FY 2025 acute care hospital inpatient wage index, as discussed in section III. of the preamble of this final rule (that is, wage data collected from cost reports submitted by IPPS hospitals for cost reporting periods beginning during FY 2021) because these data are the most recent complete data available.
In addition, as we proposed, we computed the FY 2025 LTCH PPS standard Federal payment rate area wage index values consistent with the “urban” and “rural” geographic classifications (that is, the labor market area delineations as previously discussed in section V.B. of this Addendum) and our historical policy of not taking into account IPPS geographic reclassifications under sections 1886(d)(8) and 1886(d)(10) of the Act in determining payments under the LTCH PPS. As we proposed, we also continued to apportion the wage data for multicampus hospitals with campuses located in different labor market areas to each CBSA where the campus or campuses are located, consistent with the IPPS policy. Lastly, consistent with our existing methodology for determining the LTCH PPS wage index values, for FY 2025, as we proposed, we continued to use our existing policy for determining area wage index values for areas where there are no IPPS wage data. Under our existing methodology, the LTCH PPS wage index value for urban CBSAs with no IPPS wage data is determined by using an average of all of the urban areas within the State, and the LTCH PPS wage index value for rural areas with no IPPS wage data is determined by using the unweighted average of the wage indices from all of the CBSAs that are contiguous to the rural counties of the State.
Based on the FY 2021 IPPS wage data that we used to determine the FY 2025 LTCH PPS area wage index values in this final rule, there are no IPPS wage data for the urban area of Hinesville, GA (CBSA 25980). Consistent with our existing methodology, we calculated the FY 2025 wage index value for CBSA 25980 as the average of the wage index values for all of the other urban areas within the State of Georgia (that is, CBSAs 10500, 12020, 12054, 12260, 15260, 16860, 17980, 19140, 23580, 31420, 31924, 40660, 42340, 46660, and 47580), as shown in Table 12A, which is listed in section VI. of this Addendum.
Based on the FY 2021 IPPS wage data that we used to determine the FY 2025 LTCH PPS area wage index values in this final rule, there are no IPPS wage data for rural North Dakota (CBSA 35). Consistent with our existing methodology, we calculated the FY 2025 wage index value for CBSA 35 as the average of the wage index values for all CBSAs that are contiguous to the rural counties of the State (that is, CBSAs 13900, 22020, 24220, and 33500), as shown in Table 12B, which is listed in section VI. of this Addendum. We note that, as IPPS wage data are dynamic, it is possible that the number of urban and rural areas without IPPS wage data will vary in the future.
Comment: A commenter stated that CMS should account for geographic reclassification of IPPS hospitals when determining the LTCH PPS wage index. The commenter believes that not accounting for geographic reclassification when determining the LTCH PPS wage index disadvantages LTCHs when competing with IPPS hospitals for clinical staff.
Response: We did not propose to account for geographic reclassification of IPPS hospitals when determining the LTCH PPS wage index as suggested by the commenter and do not believe such a policy is necessary at this time, but we will take this comment into consideration to potentially inform future rulemaking.
Comment: A commenter stated that there are discrepancies between the IPPS and LTCH PPS wage indexes for the same CBSAs. This commenter requested that CMS provide additional information on these differences and provide the data and information necessary to replicate the LTCH PPS wage index calculations. The commenter did not provide examples of specific CBSAs that were of particular concern to them.
Response: We thank the commenter for the feedback. As we described previously, the LTCH PPS wage index values are calculated from acute care IPPS hospital wage data without taking into account geographic reclassification. There are also several other adjustments made in determining the IPPS wage index that are not applicable to the LTCH PPS wage index, such as the occupational mix adjustment. For these reasons, differences between the LTCH PPS wage index and the IPPS wage index are to be expected. We refer the reader to section III.C. of the preamble of this final rule for details on the methodology for computing the IPPS wage index. We note that the wage and hours data from the acute care IPPS hospital used in calculating the LTCH PPS wage index values are available in the Public Use Files released with each proposed and final rule each fiscal year. The Public Use Files for this final rule will be posted on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html .
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49440 through 49442 ), we finalized a policy that applies a permanent 5-percent cap on any decrease to an LTCH's wage index from its wage index in the prior year. Consistent with the requirement at § 412.525(c)(2) that changes to area wage level adjustments are made in a budget neutral manner, we include the application of this policy in the determination of the area wage level budget neutrality factor that is applied to the standard Federal payment rate, as is discussed later in section V.B.6. of this Addendum.
Under this policy, an LTCH's wage index will not be less than 95 percent of its wage index for the prior fiscal year. An LTCH's wage index cap adjustment is determined based on the wage index value applicable to the LTCH on the last day of the prior Federal fiscal year. However, for newly opened LTCHs that become operational on or after the first day of the fiscal year, these LTCHs will not be subject to the LTCH PPS wage index cap since they were not paid under the LTCH PPS in the prior year. For example, newly opened LTCHs that become operational during FY 2025 would not be eligible for the LTCH PPS wage index cap in FY 2025. These LTCHs would receive the calculated wage index for the area in which they are geographically located, even if other LTCHs in the same geographic area are receiving a wage ( print page 69977) index cap. The cap on wage index decreases policy is reflected at § 412.525(c)(1).
For each LTCH we identify in our rulemaking data, we are including in a supplemental data file the wage index values from both fiscal years used in determining its capped wage index. This includes the LTCH's final prior year wage index value, the LTCH's uncapped current year wage index value, and the LTCH's capped current year wage index value. Due to the lag in rulemaking data, a new LTCH may not be listed in this supplemental file for a few years. For this reason, a newly opened LTCH could contact their MAC to ensure that its wage index value is not less than 95 percent of the value paid to it for the prior Federal fiscal year. This supplemental data file for public use will be posted on the CMS website for this final rule at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html .
Comment: A commenter expressed their appreciation of the permanent cap on LTCH PPS wage index decreases policy.
Response: We thank the commenter for their support of this policy.
Determining LTCH PPS payments for short-stay-outlier cases (reflected in § 412.529) and site neutral payment rate cases (reflected in § 412.522(c)) requires calculating an “IPPS comparable amount.” For information on this “IPPS comparable amount” calculation, we refer the reader to the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49608 through 49610 ). Determining LTCH PPS payments for LTCHs that do not meet the applicable discharge payment percentage (reflected in § 412.522(d)) requires calculating an “IPPS equivalent amount.” For information on this “IPPS equivalent amount” calculation, we refer the reader to the FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42439 through 42445 ).
Calculating both the “IPPS comparable amount” and the “IPPS equivalent amount” requires adjusting the IPPS operating and capital standardized amounts by the applicable IPPS wage index for nonreclassified IPPS hospitals. That is, the standardized amounts are adjusted by the IPPS wage index for nonreclassified IPPS hospitals located in the same geographic area as the LTCH. In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49442 through 49443 ), we finalized a policy that applies a permanent 5-percent cap on decreases in an LTCH's applicable IPPS comparable wage index from its applicable IPPS comparable wage index in the prior year. Historically, we have not budget neutralized changes to LTCH PPS payments that result from the annual update of the IPPS wage index for nonreclassified IPPS hospitals. Consistent with this approach, the cap on decreases in an LTCH's applicable IPPS comparable wage index is not applied in a budget neutral manner.
Under this policy, an LTCH's applicable IPPS comparable wage index will not be less than 95 percent of its applicable IPPS comparable wage index for the prior fiscal year. An LTCH's applicable IPPS comparable wage index cap adjustment is determined based on the wage index value applicable to the LTCH on the last day of the prior Federal fiscal year. However, for newly opened LTCHs that become operational on or after the first day of the fiscal year, these LTCHs will not be subject to the applicable IPPS comparable wage index cap since they were not paid under the LTCH PPS in the prior year. For example, newly opened LTCHs that become operational during FY 2025 would not be eligible for the applicable IPPS comparable wage index cap in FY 2025. This means that these LTCHs would receive the calculated applicable IPPS comparable wage index for the area in which they are geographically located, even if other LTCHs in the same geographic area are receiving a wage cap. The cap on IPPS comparable wage index decreases policy is reflected at § 412.529(d)(4)(ii)(B) and (d)(4)(iii)(B).
Similar to the information we are making available for the cap on the LTCH PPS wage index values (described previously), for each LTCH we identify in our rulemaking data, we are including in a supplemental data file the wage index values from both fiscal years used in determining its capped applicable IPPS comparable wage index. Due to the lag in rulemaking data, a new LTCH may not be listed in this supplemental file for a few years. For this reason, a newly opened LTCH could contact its MAC to ensure that its applicable IPPS comparable wage index value is not less than 95 percent of the value paid to them for the prior Federal fiscal year. This supplemental data file for public use will be posted on the CMS website for this final rule at: https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html .
Historically, the LTCH PPS wage index and labor-related share are updated annually based on the latest available data. Under § 412.525(c)(2), any changes to the area wage index values or labor-related share are to be made in a budget neutral manner such that estimated aggregate LTCH PPS payments are unaffected; that is, will be neither greater than nor less than estimated aggregate LTCH PPS payments without such changes to the area wage level adjustment. Under this policy, we determine an area wage level adjustment budget neutrality factor that is applied to the standard Federal payment rate to ensure that any changes to the area wage level adjustments are budget neutral such that any changes to the area wage index values or labor-related share would not result in any change (increase or decrease) in estimated aggregate LTCH PPS payments. Accordingly, under § 412.523(d)(4), we have applied an area wage level adjustment budget neutrality factor in determining the standard Federal payment rate, and we also established a methodology for calculating an area wage level adjustment budget neutrality factor. (For additional information on the establishment of our budget neutrality policy for changes to the area wage level adjustment, we refer readers to the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51771 through 51773 and 51809 ).)
For FY 2025, in accordance with § 412.523(d)(4), we are applying an area wage level budget neutrality factor to adjust the LTCH PPS standard Federal payment rate to account for the estimated effect of the adjustments or updates to the area wage level adjustment under § 412.525(c)(1) on estimated aggregate LTCH PPS payments, consistent with the methodology we established in the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51773 ). As discussed in section V.B.6. of this Addendum, consistent with, § 412.525(c)(2), we include the application of the 5-percent cap on wage index decreases in the determination of the area wage level budget neutrality factor. Specifically, as we proposed, we determined an area wage level adjustment budget neutrality factor that is applied to the LTCH PPS standard Federal payment rate under § 412.523(d)(4) for FY 2025 using the following methodology:
Step 1 —Simulate estimated aggregate LTCH PPS standard Federal payment ( print page 69978) rate payments using the FY 2024 wage index values and the FY 2024 labor-related share of 68.5 percent. We note that the FY 2024 wage index values are based on the existing CBSA labor market areas used in the FY 2024 IPPS/LTCH PPS final rule.
Step 2 —Simulate estimated aggregate LTCH PPS standard Federal payment rate payments using the FY 2025 wage index values (including the update to the CBSA labor market areas and the application of the 5 percent cap on wage index decreases) and the FY 2025 labor-related share of 72.8 percent. (As noted previously, the changes to the wage index values based on updated hospital wage data are discussed in section V.B.4. of this Addendum and the labor-related share is discussed in section V.B.3. of this Addendum.)
Step 3 —Calculate the ratio of these estimated total LTCH PPS standard Federal payment rate payments by dividing the estimated total LTCH PPS standard Federal payment rate payments using the FY 2024 area wage level adjustments (calculated in Step 1) by the estimated total LTCH PPS standard Federal payment rate payments using the FY 2025 updates to the area wage level adjustment (calculated in Step 2) to determine the budget neutrality factor for updates to the area wage level adjustment for FY 2025 LTCH PPS standard Federal payment rate payments.
Step 4 —Apply the FY 2025 updates to the area wage level adjustment budget neutrality factor from Step 3 to determine the FY 2025 LTCH PPS standard Federal payment rate after the application of the FY 2025 annual update.
As we proposed, we used the most recent data available, including claims from the FY 2023 MedPAR file, in calculating the FY 2025 LTCH PPS standard Federal payment rate area wage level adjustment budget neutrality factor. We note that, because the area wage level adjustment under § 412.525(c) is an adjustment to the LTCH PPS standard Federal payment rate, consistent with historical practice, we only used data from claims that qualified for payment at the LTCH PPS standard Federal payment rate under the dual rate LTCH PPS to calculate the FY 2025 LTCH PPS standard Federal payment rate area wage level adjustment budget neutrality factor.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49448 ), we discussed the abnormal charging practices of an LTCH (CCN 312024) in FY 2021 that led to the LTCH receiving an excessive amount of high-cost outlier payments. In that rule, we stated our understanding that, based on information we received from the provider, these abnormal charging practices would not persist into FY 2023. Therefore, we did not include their cases in our model for determining the FY 2023 outlier fixed-loss amount. In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59376 ), we stated that the FY 2022 MedPAR claims also reflect the abnormal charging practices of this LTCH. Therefore, we removed claims from CCN 312024 when determining the fixed-loss amount for LTCH PPS standard Federal payment rate cases for FY 2024 and all other FY 2024 ratesetting calculations, including the MS-LTC-DRG relative weights and the calculation of the area wage level adjustment budget neutrality factor. Given recent actions by the Department of Justice regarding CCN 312024 (see https://www.justice.gov/opa/pr/new-jersey-hospital-and-investors-pay-united-states-306-million-alleged-false-claims-related ), as we proposed, we again removed claims from CCN 312024 when determining the area wage level adjustment budget neutrality factor for FY 2025 and all other FY 2025 ratesetting calculations, including the MS-LTC-DRG relative weights and the fixed-loss amount for LTCH PPS standard Federal payment rate cases.
For this final rule, using the steps in the methodology previously described, we determined a FY 2025 LTCH PPS standard Federal payment rate area wage level adjustment budget neutrality factor of 0.9964315. Accordingly, in section V.A. of this Addendum, we applied the area wage level adjustment budget neutrality factor of 0.9964315 to determine the FY 2025 LTCH PPS standard Federal payment rate, in accordance with § 412.523(d)(4).
Under § 412.525(b), a cost-of-living adjustment (COLA) is provided for LTCHs located in Alaska and Hawaii to account for the higher costs incurred in those States. Specifically, we apply a COLA to payments to LTCHs located in Alaska and Hawaii by multiplying the nonlabor-related portion of the standard Federal payment rate by the applicable COLA factors established annually by CMS. Higher labor-related costs for LTCHs located in Alaska and Hawaii are taken into account in the adjustment for area wage levels previously described. The methodology used to determine the COLA factors for Alaska and Hawaii is based on a comparison of the growth in the Consumer Price Indexes (CPIs) for Anchorage, Alaska, and Honolulu, Hawaii, relative to the growth in the CPI for the average U.S. city as published by the Bureau of Labor Statistics (BLS). It also includes a 25-percent cap on the CPI-updated COLA factors. Under our current policy, we have updated the COLA factors using the methodology as previously described every 4 years (at the same time as the update to the labor-related share of the IPPS market basket) and we last updated the COLA factors for Alaska and Hawaii published by OPM for 2009 in FY 2022 ( 86 FR 45559 through 45560 ).
We continue to believe that determining updated COLA factors using this methodology would appropriately adjust the nonlabor-related portion of the LTCH PPS standard Federal payment rate for LTCHs located in Alaska and Hawaii. Therefore, in this final rule, for FY 2025, under the broad authority conferred upon the Secretary by section 123 of the BBRA, as amended by section 307(b) of the BIPA, to determine appropriate payment adjustments under the LTCH PPS, as we proposed, we are continuing to use the COLA factors based on the 2009 OPM COLA factors updated through 2020 by the comparison of the growth in the CPIs for Anchorage, Alaska, and Honolulu, Hawaii, relative to the growth in the CPI for the average U.S. city as established in the FY 2022 IPPS/LTCH PPS final rule. (For additional details on our current methodology for updating the COLA factors for Alaska and Hawaii and for a discussion on the FY 2022 COLA factors, we refer readers to the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45559 through 45560 ).)

From the beginning of the LTCH PPS, we have included an adjustment to account for cases in which there are extraordinarily high costs relative to the costs of most discharges. Under this policy, additional payments are made based on the degree to which the estimated cost of a case (which is calculated by multiplying the Medicare allowable covered charge by the hospital's overall hospital CCR) exceeds a fixed-loss amount. This policy results in greater payment accuracy under the LTCH PPS and the Medicare program, and the LTCH sharing the financial risk for the treatment of extraordinarily high-cost cases.
We retained the basic tenets of our HCO policy in FY 2016 when we implemented the dual rate LTCH PPS payment structure under section 1206 of Public Law 113-67 . LTCH discharges that meet the criteria for exclusion from the site neutral payment rate (that is, LTCH PPS standard Federal payment rate cases) are paid at the LTCH PPS standard Federal payment rate, which includes, as applicable, HCO payments under § 412.523(e). LTCH discharges that do not meet the criteria for exclusion are paid at the site neutral payment rate, which includes, as applicable, HCO payments under § 412.522(c)(2)(i). In the FY 2016 IPPS/LTCH PPS final rule, we established separate fixed-loss amounts and targets for the two different LTCH PPS payment rates. Under this bifurcated policy, the historic 8-percent HCO target was retained for LTCH PPS standard Federal payment rate cases, with the fixed-loss amount calculated using only data from LTCH cases that would have been paid at the LTCH PPS standard Federal payment rate if that rate had been in effect at the time of those discharges. For site neutral payment rate cases, we adopted the operating IPPS HCO target (currently 5.1 percent) and set the fixed-loss amount for site neutral payment rate cases at the value of the IPPS fixed-loss amount. Under the HCO policy for both payment rates, an LTCH receives 80 percent of the difference between the estimated cost of the case and the applicable HCO threshold, which is the sum of the LTCH PPS payment for the case and the applicable fixed-loss amount for such case.
To maintain budget neutrality, consistent with the budget neutrality requirement at § 412.523(d)(1) for HCO payments to LTCH PPS standard Federal rate payment cases, we also adopted a budget neutrality requirement for HCO payments to site neutral payment rate cases by applying a budget neutrality factor to the LTCH PPS payment for those site neutral payment rate cases. (For additional details on the HCO policy adopted for site neutral payment rate cases under the dual rate LTCH PPS payment structure, including the budget neutrality adjustment for HCO payments to site neutral payment rate cases, we refer readers to § 412.522(c)(2)(i) of the regulations and to the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49617 through 49623 ).)
As noted previously, CCRs are used to determine payments for HCO adjustments for both payment rates under the LTCH PPS and are also used to determine payments for site neutral payment rate cases. As noted earlier, in determining HCO and the site neutral payment rate payments (regardless of whether the case is also an HCO), we generally calculate the estimated cost of the case by multiplying the LTCH's overall CCR by the Medicare allowable charges for the case. An overall CCR is used because the LTCH PPS uses a single prospective payment per discharge that covers both inpatient operating and capital-related costs. The LTCH's overall CCR is generally computed based on the sum of LTCH operating and capital costs (as described in section 150.24, Chapter 3, of the Medicare Claims Processing Manual (Pub. 100-4)) as compared to total Medicare charges (that is, the sum of its operating and capital inpatient routine and ancillary charges), with those values determined from either the most recently settled cost report or the most recent tentatively settled cost report, whichever is from the latest cost reporting period. However, in certain instances, we use an alternative CCR, such as the statewide average CCR, a CCR that is specified by CMS, or one that is requested by the hospital. (We refer readers to § 412.525(a)(4)(iv) of the regulations for further details regarding CCRs and HCO adjustments for either LTCH PPS payment rate and § 412.522(c)(1)(ii) for the site neutral payment rate.)
The LTCH's calculated CCR is then compared to the LTCH total CCR ceiling. Under our established policy, an LTCH with a calculated CCR in excess of the applicable maximum CCR threshold (that is, the LTCH total CCR ceiling, which is calculated as 3 standard deviations from the national geometric average CCR) is generally assigned the applicable statewide CCR. ( print page 69980) This policy is premised on a belief that calculated CCRs in excess of the LTCH total CCR ceiling are most likely due to faulty data reporting or entry, and CCRs based on erroneous data should not be used to identify and make payments for outlier cases.
Consistent with our historical practice, as we proposed, we used the best available data to determine the LTCH total CCR ceiling for FY 2025 in this final rule. Specifically, in this final rule, we used our established methodology for determining the LTCH total CCR ceiling based on IPPS total CCR data from the March 2024 update of the Provider Specific File (PSF), which is the most recent data available. Accordingly, we are establishing an LTCH total CCR ceiling of 1.368 under the LTCH PPS for FY 2025 in accordance with § 412.525(a)(4)(iv)(C)( 2 ) for HCO cases under either payment rate and § 412.522(c)(1)(ii) for the site neutral payment rate. (For additional information on our methodology for determining the LTCH total CCR ceiling, we refer readers to the FY 2007 IPPS final rule ( 71 FR 48117 through 48119 ).)
We did not receive any public comments on our proposals and are finalizing our proposals as described previously.
Our general methodology for determining the statewide average CCRs used under the LTCH PPS is similar to our established methodology for determining the LTCH total CCR ceiling because it is based on “total” IPPS CCR data. (For additional information on our methodology for determining statewide average CCRs under the LTCH PPS, we refer readers to the FY 2007 IPPS final rule ( 71 FR 48119 through 48120 ).) Under the LTCH PPS HCO policy at § 412.525(a)(4)(iv)(C), the SSO policy at § 412.529(f)(4)(iii), and the site neutral payment rate at § 412.522(c)(1)(ii), the MAC may use a statewide average CCR, which is established annually by CMS, if it is unable to determine an accurate CCR for an LTCH in one of the following circumstances: (1) New LTCHs that have not yet submitted their first Medicare cost report (a new LTCH is defined as an entity that has not accepted assignment of an existing hospital's provider agreement in accordance with § 489.18); (2) LTCHs whose calculated CCR is in excess of the LTCH total CCR ceiling; and (3) other LTCHs for whom data with which to calculate a CCR are not available (for example, missing or faulty data). (Other sources of data that the MAC may consider in determining an LTCH's CCR include data from a different cost reporting period for the LTCH, data from the cost reporting period preceding the period in which the hospital began to be paid as an LTCH (that is, the period of at least 6 months that it was paid as a short-term, acute care hospital), or data from other comparable LTCHs, such as LTCHs in the same chain or in the same region.)
Consistent with our historical practice of using the best available data, in this final rule, as we proposed, we are using our established methodology for determining the LTCH PPS statewide average CCRs, based on the most recent complete IPPS “total CCR” data from the March 2024 update of the PSF. As we proposed, we are establishing LTCH PPS statewide average total CCRs for urban and rural hospitals that will be effective for discharges occurring on or after October 1, 2024, through September 30, 2025, in Table 8C listed in section VI. of this Addendum (and available via the internet on the CMS website).
Under the LTCH PPS labor market areas for FY 2025, all areas in the District of Columbia, New Jersey, and Rhode Island are classified as urban. Therefore, there are no rural statewide average total CCRs listed for those jurisdictions in Table 8C. This policy is consistent with the policy that we established when we revised our methodology for determining the applicable LTCH statewide average CCRs in the FY 2007 IPPS final rule ( 71 FR 48119 through 48121 ) and is the same as the policy applied under the IPPS. In addition, consistent with our existing methodology, in determining the urban and rural statewide average total CCRs for Maryland LTCHs paid under the LTCH PPS, as we proposed, we are continuing to use, as a proxy, the national average total CCR for urban IPPS hospitals and the national average total CCR for rural IPPS hospitals, respectively. We are using this proxy because we believe that the CCR data in the PSF for Maryland hospitals may not be entirely accurate (as discussed in greater detail in the FY 2007 IPPS final rule ( 71 FR 48120 )).
Furthermore, although Connecticut, Massachusetts, and North Dakota have areas that are designated as rural under the LTCH PPS labor market areas for FY 2025, in our calculation of the LTCH statewide average CCRs, there were no trimmed CCR data available from IPPS hospitals located in these rural areas as of March 2024. We refer the reader to section II.A.4.i.(2). of this Addendum for details on the trims applied to the IPPS CCR data from the March 2024 update of the PSF, which are the same data used to calculate the LTCH statewide average total CCRs. Therefore, consistent with our existing methodology, we used the national average total CCR for rural IPPS hospitals for rural Connecticut, Massachusetts, and North Dakota in Table 8C. We note that there were no LTCHs located in these rural areas as of March 2024.
We did not receive any public comments on our proposals. We are finalizing our proposals as described previously.
Under the HCO policy at § 412.525(a)(4)(iv)(D), the payments for HCO cases are subject to reconciliation (regardless of whether payment is based on the LTCH standard Federal payment rate or the site neutral payment rate). Specifically, any such payments are reconciled at settlement based on the CCR that was calculated based on the cost report coinciding with the discharge. For additional information on the reconciliation policy, we refer readers to sections 150.26 through 150.28 of the Medicare Claims Processing Manual (Pub. 100-4), as added by Change Request 7192 (Transmittal 2111; December 3, 2010) and the RY 2009 LTCH PPS final rule ( 73 FR 26820 through 26821 ), and most recently modified by Change Request 13566 (Transmittal 12558; March 28, 2024) with an update to the outlier reconciliation criteria.
Comment: Commenters were concerned that CMS has added new criteria for determining which LTCHs will have their outlier payments reconciled in CR 13566. The commenters stated their belief that new reconciliation criteria constitute a substantive change to CMS' payment policy that cannot be adopted without notice and comment rulemaking. The commenters urged CMS to withdraw the CR.
Response: CMS established the outlier reconciliation regulation under § 412.525(a)(4)(iv)(D) effective for discharges on or after October 1, 2006, which makes all LTCH outlier payments subject to reconciliation. CMS has not modified the outlier regulation. The instructions CMS has issued via CR 13566 have set forth an enforcement policy that determines when MACs will identify additional LTCHs for reconciliation referral. They do not change the legal standards that govern the LTCHs. ( print page 69981)
Under the regulations at § 412.525(a)(2)(ii) and as required by section 1886(m)(7) of the Act, the fixed-loss amount for HCO payments is set each year so that the estimated aggregate HCO payments for LTCH PPS standard Federal payment rate cases are 99.6875 percent of 8 percent (that is, 7.975 percent) of estimated aggregate LTCH PPS payments for LTCH PPS standard Federal payment rate cases. (For more details on the requirements for high-cost outlier payments in FY 2018 and subsequent years under section 1886(m)(7) of the Act and additional information regarding high-cost outlier payments prior to FY 2018, we refer readers to the FY 2018 IPPS/LTCH PPS final rule ( 82 FR 38542 through 38544 ).)
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36590 through 36592 ), we presented our proposed methodology for determining the outlier fixed-loss amount for LTCH PPS standard Federal payment rate cases and proposed an outlier fixed-loss amount of $90,921. In the proposed rule, we acknowledged that the proposed increase to the fixed-loss amount from the FY 2024 fixed-loss amount ($59,873) was substantial. We also acknowledged that the FY 2024 fixed-loss amount was substantially higher than the FY 2023 fixed-loss amount ($38,518). Recognizing that such substantial increases to the fixed-loss amount in consecutive years could impact LTCH operations, in the proposed rule, we considered an alternative approach for determining the proposed fixed-loss threshold for FY 2025. As discussed in full in section I.O.4. of Appendix A of the proposed rule ( 89 FR 36664 ), the alternative approach we considered would have established the FY 2025 fixed-loss amount as an average of the FY 2024 fixed-loss amount and our modelled FY 2025 fixed-loss amount. Under this approach, the proposed fixed-loss amount would have been $75,397 (($59,873 + $90,921)/2). In the proposed rule, we solicited comments on our proposed fixed-loss amount for FY 2025 as well as on the alternative approach that we considered for determining the fixed-loss amount for FY 2025. In this section, we first summarize and respond to the comments received in response to those solicitations. Later in this section, we present the detailed application of our finalized methodology after consideration of the comments received.
Comment: Several commenters objected to the charge inflation factor we proposed to apply under our proposed methodology for determining the FY 2025 fixed-loss amount. Many commenters urged CMS to return to the methodology employed prior to FY 2022 in which the charge inflation factor was set equal to the market basket update. These commenters stated that returning to this methodology would provide greater stability and predictability to the outlier fixed-loss amount. A commenter asserted that the proposed charge inflation methodology has led to inaccurate fixed-loss amounts in previous years and therefore CMS should return to our previous methodology. This commenter stated that CMS significantly overpaid outliers in FY 2023 relative to our statutory 7.975 percent target and would have significantly underpaid outliers in FY 2024 but for the modifications CMS made to the methodology in the FY 2024 final rule, in which CMS applied a charge inflation factor and CCR adjustment factor based on data prior to the COVID-19 PHE rather than based on the most recently available data. Another commenter stated that CMS should return to its previous methodology because of the uncertain impacts of inflation on charges and lagged availability of changes in CCRs. A commenter requested that CMS calculate the charge inflation factor based on data from prior to the COVID-19 PHE. Another commenter stated that CMS should modify the statistical outlier trim used in our methodology for determining the charge inflation factor by removing claims for providers with a calculated charge growth factor that exceeds 1 standard deviation from the mean provider charge growth factor. A commenter requested that when determining the two-year charge inflation factor, CMS double the one-year charge inflation factor rather than squaring the one-year charge inflation factor. The same commenter stated that the CCR adjustment factor is a double adjustment of the charge inflation factor and requested that CMS remove the CCR adjustment factor from the calculation of the outlier fixed-loss amount.
Response: We appreciate the feedback and suggestions that commenters provided on the proposed charge inflation factor. We appreciate the importance of more stability and predictability in the annual fixed-loss amount. We agree that it is reasonable to evaluate the effectiveness and accuracy of our fixed-loss amount methodology by determining how close actual outlier payments were to our statutory target of 7.975 percent of total payments for LTCH PPS standard Federal payment rate cases. We also acknowledge that in recent years and in FY 2025, the calculated fixed-loss amount would have been lower if we had estimated charge inflation based on the market basket update. However, we do not agree with commenters that the fixed-loss amounts calculated under the previous market basket methodology would have yielded outlier payments closer to the statutory target in either FY 2022 or FY 2023. We estimate that high cost outlier payments significantly exceeded the statutory 7.975 percent target in both FY 2022 and FY 2023. Using the previous market basket methodology for FY 2022 and FY 2023 would have resulted in lower estimates of costs per discharge and lower fixed-loss amounts. These lowered fixed-loss amounts would have resulted in high cost outlier payments exceeding the statutory target by even more than we estimate actually occurred. While commenters also implied that this market basket approach would have yielded a more accurate fixed-loss amount in FY 2024, we note that commenters are referring to our projection of FY 2024 outlier payments included in the proposed rule. The FY 2024 payment projections in both the proposed rule and this final rule are not based on actual FY 2024 claims but rather based on a payment model that uses FY 2023 claims. We refer the reader to section J.3.C. of the Appendix to this final rule for a full description of our methodology for modelling FY 2024 payments using FY 2023 claims. We believe it is more appropriate to evaluate the effectiveness and accuracy of our fixed-loss amount methodology based on an analysis of actual FY 2024 payments rather than a projection of FY 2024 payments. For these reasons we continue to believe using a charge inflation factor based on actual growth rates in charges from historical claims data rather than one based on quarterly market basket update values leads to better accuracy in calculating the fixed-loss amount that would result in actual outlier payments meeting the statutory target.
We also disagree with the other modifications commenters suggested we make to the charge inflation factor. We believe using the most recent data available is appropriate for projecting charge inflation for FY 2025. In FYs 2022 through 2024, we used a charge ( print page 69982) inflation factor based on data prior to the COVID-19 PHE. However, after analyzing actual LTCH PPS claims from FY 2022 and FY 2023, we believe actual outlier payments during these years would have been closer to the statutory target if we had used the most recent available data to determine the charge inflation factor when establishing the fixed-loss amounts for these fiscal years. We note that the commenter that requested CMS remove claims for providers with a calculated charge growth factor that exceeds 1 standard deviation from the mean provider charge growth factor did not provide any justification for making this methodology change. We continue to believe that removing providers from the charge inflation factor calculation with a calculated charge growth factor that exceeds 3 standard deviations from the mean provider charge growth factor is effective at removing actual aberrations in the data that would distort the measure of average charge growth. We also note that using 3 standard deviations from the mean as a threshold for removing aberrations in ratesetting data is a standard method that CMS uses in other IPPS and LTCH PPS ratesetting calculations, such as the calculation of the relative weights. We also disagree with the comment that the CCR adjustment factor is duplicative of the charge inflation factor or that it should be applied in an additive rather than a multiplicative manner. The charge inflation factor accounts for the historical growth in charges for LTCHs while the CCR adjustment factor accounts for historical changes in the relationship between costs and charges for LTCHs. We believe both factors are necessary for estimating costs of LTCH cases in FY 2025 from historical LTCH data. To account for annual growth in LTCHs' charges, we also continue to believe that squaring the one-year charge inflation factor is the appropriate calculation for projecting year-over-year charge inflation for a two-year period That is, to increase the charges from the FY 2023 MedPAR claims to projected FY 2025 charge levels, it is necessary to multiply the FY 2023 charges by the one-year charge inflation factor two times (FY 2023 charges × 1-year charge inflation factor × 1-year charge inflation factor). To simplify this equation, the FY 2023 charges can instead be multiplied by the 1-year charge inflation factor squared (FY 2023 charges × (1-year charge inflation factor ^2)) and achieve the same resulting estimate of projected FY 2025 charges.
Comment: Some commenters asserted that the data CMS proposed to use were significantly impacted by the COVID-19 pandemic. These commenters stated that the claims and cost report data CMS proposed to use reflect patient acuity and cost trends that are unlikely to be repeated in FY 2025. Examples provided by commenters included differences in patient acuity during the COVID-19 pandemic, levels of COVID-19 hospitalizations, and changes in vaccination and immunity rates. These commenters also believe that CMS should adjust our ratesetting methodologies for FY 2025, including our methodology for determining the fixed-loss amount, to account for these pandemic-era impacts. We note that commenters did not provide recommendations for specific technical adjustments CMS could make to the ratesetting data to account for the impact of COVID-19.
Similar to last year, some commenters urged CMS to exclude dialysis patients from the FY 2023 claims data when determining the outlier fixed-loss amount. Commenters again stated that since the start of the COVID-19 pandemic, the cost of providing in-hospital dialysis to LTCH patients has increased significantly. These commenters stated that many LTCHs continue to face significant increases in the rates charged by third-party dialysis vendors or have begun providing dialysis services “in-house” at higher costs. Some commenters stated that they expect this trend to continue as the staffing and wage pressures faced by third-party vendors continue. Commenters also stated that LTCHs continue to face challenges discharging dialysis patients due to limited space in outpatient dialysis clinics, which has led to longer lengths of stay and costs for these cases. Commenters believe that dialysis cases are skewing the fixed-loss amount calculation and believe removing dialysis cases would allow for a more accurate forecast of what costs and charges will look like when these issues subside.
A few commenters encouraged CMS to use more recent data to determine the fixed-loss amount in the final rule. Some commenters requested that CMS incorporate claims data from FY 2024 into the calculation of the fixed-loss amount for FY 2025. Another commenter stated that CMS should use data from more recent cost reports in the calculation. This commenter stated that their analysis has found that using data from cost reports with a July 31st cutoff historically has resulted in a more accurate fixed-loss amount.
Response: We thank the commenters for their suggestions to modify the data used in calculating the fixed-loss threshold to account for COVID-19 impacts. In the FY 2023 claims data used for this final rule, we found that approximately 4.0 percent of LTCH standard payment rate claims had a COVID-19 diagnosis code. We do not have reason to assume that the percentage of claims with a COVID-19 diagnosis code in FY 2025 will be meaningfully different than FY 2023. Furthermore, using the March 2024 update of the FY 2023 MedPAR file, we estimate that actual high-cost outlier payments as a percentage of total LTCH PPS standard Federal payment rate payments in FY 2023 would only decrease by 0.1 percentage point if we were to remove all claims with a COVID-19 diagnosis. Therefore, while we do not believe a modification is necessary, we also believe that making such modification would not have had a significant influence on the fixed-loss amount for FY 2025. For these reasons, we are not adopting commenters' suggestion to use different data from the data we proposed to use in calculating the fixed-loss threshold to account for the impact of the COVID-19 pandemic.
We thank the commenters for the suggestion to exclude dialysis claims when calculating the fixed-loss threshold. Although commenters described why dialysis cases were costly in FY 2023, similar to our response to such comments last year ( 88 FR 59374-59375 ), we still do not find that commenters provided sufficient evidence to support why costs for these types of patients would differ significantly from FY 2023 to FY 2025, such that it would be appropriate to exclude them from our calculations. Some commenters explicitly stated in their comments that they expect LTCHs will continue to incur higher costs of providing dialysis services to patients. For these reasons, we are not adopting commenters' suggestion to exclude dialysis claims when calculating the fixed-loss threshold for FY 2025.
We thank the commenters for the suggestion to use more recent data for calculating the fixed-loss threshold in this final rule. As discussed later in this section, we are using more recent data than we used in the proposed rule. Specifically, we are using the March 2024 update of the FY 2023 MedPAR file and the March 2024 update of the Provider Specific File (PSF) to calculate the fixed-loss threshold in this final rule. At the time of developing this final rule, the March 2024 update of the FY 2023 MedPAR file was the most recent full year of publicly available claims data. Similarly, at the time of developing this final rule, the March 2024 update of the PSF was the most ( print page 69983) recent publicly available version of the PSF. With regards to the comment requesting we use the most recently available cost report data, we note that the PSF generally contains CCR data from an LTCH's most recently settled or tentatively settled cost report, whichever is from the latest cost reporting period.
We continue to believe it is most appropriate to use one full year of publicly available claims data in our ratesetting calculations. The use of one full year of publicly available claims data is consistent with our historical practice and is not susceptible to the seasonality issues affiliated with using partial year data. Therefore, we are not adopting commenters' suggestion to incorporate claims from the first part of FY 2024 in our calculation of the fixed-loss threshold for FY 2025.
Comment: Several commenters believe that CMS needs to update its high-cost outlier policy to better account for the effects of the dual rate LTCH PPS payment structure on outlier payments. Several commenters stated that the under the dual rate payment structure, the majority of LTCH standard Federal payment rate cases have become concentrated to only a few MS-LTC-DRGs. The commenters stated that there is great variation in patient severity and costs among the cases grouped to these MS-LTC-DRGs which they believe leads to many of them qualifying for outlier payments, and that this pattern is contributing to the proposed increase in the fixed-loss amount. Commenters highlighted standard Federal payment rate cases grouped to base MS-LTC-DRGs 189 and 207 in particular. These two base MS-DRGs, which accounted for over 40 percent of standard Federal payment rate cases in FY 2023, are not subdivided based on the presence or absence of a complication or comorbidity (CC) or a major complication or comorbidity (MCC). Commenters requested that CMS refine certain MS-LTC-DRGs, such as by creating subgroups within these base MS-DRGs based on the presence or absence of CCs and MCCs, which they believe would increase LTCH PPS payment accuracy thereby reducing the outlier payments made to cases grouped to such MS-LTC-DRGs.
Response: We appreciate commenters' suggestions on possible refinements to certain MS-LTC-DRGs, in particular the concerns regarding the absence of CC or MCC subgroups within certain high-volume MS-LTC-DRGs, and commenters' thoughts on the impact this may have on LTCH PPS outlier payments. We note that we did not propose to make any adjustments or create CC or MCC subgroups within the MS-LTC-DRGs as requested by commenters. We also recognize that such adjustments would have differential impacts on individual LTCHs based on each LTCH's case mix. As such, we would like to have the opportunity to explore and analyze such adjustments more before making this type of change. Therefore, we are not adopting any of the changes to the MS-LTC-DRGs suggested by commenters in this final rule. However, we may consider these comments for future rulemaking.
Comment: Commenters expressed concern with the impact of the LTCH PPS dual rate payment system on the claims data CMS uses for calculating the fixed-loss amount. Commenters asserted that because CMS only uses cases that would have been paid the standard Federal rate, the claims dataset used in the calculation is smaller and on average has a higher acuity than the claims datasets CMS used prior to the start of the dual rate payment structure. The commenters believe this change has led to fluctuations in the fixed-loss amount. A commenter stated that CMS should reconsider whether the statutory outlier payment target of 7.975 percent is still an appropriate target for LTCH PPS standard Federal rate cases under the dual rate payment system.
Response: We thank the commenters for this feedback. We note that commenters did not provide specific recommendations on how CMS could address the decreasing number of cases available for LTCH PPS ratesetting. We agree with commenters and believe it is reasonable to expect that, given the statutory patient criteria for payment at the LTCH PPS standard Federal rate, the average acuity of the LTCH claims data used for determining the FY 2025 outlier fixed-loss amount is higher than the average acuity of the claims data used prior to the start of the dual rate payment structure. However, section 1886(m)(7) of the Act directs the Secretary to establish a fixed-loss amount for LTCH PPS standard Federal payment rate cases that would result in total estimated outlier payments being equal to 7.975 percent of projected total LTCH PPS payments for LTCH PPS standard Federal payment rate cases.
Comment: In general, commenters expressed concern with the proposed increase to the outlier fixed-loss amount and believe it would have negative financial impacts on LTCHs. A commenter stated that most LTCHs would not be able to absorb the level of financial losses that they believe would result from the proposed fixed-loss amount. Another commenter stated that the proposed increase to the outlier fixed-loss amount conflicts with CMS's principle for stability and predictability in reimbursement rates. Commenters stated that the proposed outlier fixed-loss amount would reduce access to LTCHs, such as restricting the number of patients they admit with pressure injuries. Many commenters stated that decreased access to LTCH services would lead to significant increases in their length of stays and costs in the intensive care units of IPPS hospitals. Several commenters stated that the proposed outlier fixed-loss amount would lead to LTCH closures. Many commenters expressed that even the outlier fixed-loss amount determined using the alternative approach we considered would require LTCHs to experience significant financial losses. A commenter stated that the alternative approach we considered for determining the outlier fixed-loss amount would only delay the implementation of a steep financial cliff for LTCHs. Commenters provided a variety of recommendations for CMS to consider when determining the fixed-loss amount in this final rule.
We received several comments requesting that we adopt a modified version of the alternative approach we considered in the proposed rule for determining the FY 2025 fixed-loss amount. Many of these commenters requested that instead of phasing in the increase in the fixed-loss amount over two-years, we phase in the increase over a longer period, such as a four-year period. Commenters believe this modified approach would create a more stable transition.
A commenter requested that CMS set the FY 2025 fixed-loss amount equal to the FY 2023 fixed-loss amount. Other commenters similarly requested that CMS set the FY 2025 fixed-loss amount equal to the FY 2024 fixed-loss amount. Several commenters requested that CMS adopt a non-budget neutral cap on annual increases to the fixed-loss amount. Commenters stated that this cap would be similar to the cap policies CMS already applies to the LTCH PPS wage index and MS-LTC-DRG relative weights. Some commenters suggested that such a cap be temporary while others suggested it become a permanent part of the methodology. Such a limit on annual increases to the fixed-loss amount suggested by commenters included a cap of no more than 5 percent, of no more than 10 percent, and set equal to the annual market basket percent increase. In general, commenters believe a cap on annual increases would provide stability and predictability to the LTCH PPS. ( print page 69984)
Response: We thank the commenters for the feedback, including the variety of suggestions on alternative methods for determining the FY 2025 fixed-loss amount. In the proposed rule we acknowledged that the proposed increase to the fixed-loss amount was substantial and sought comments on our proposed fixed-loss amount as well as an alternative approach we considered. Specifically, in the FY 2025 IPPS/LTCH PPS proposed rule, we proposed a fixed-loss amount for FY 2025 of $90,921 that would result in estimated outlier payments projected to be equal to 7.975 percent of estimated FY 2025 payments for such cases. In that same proposed rule, we also discussed an alternative approach we considered which would have established the FY 2025 fixed-loss amount as an average of the FY 2024 fixed amount and our modelled FY 2025 fixed-loss amount. Under this approach, the proposed fixed-loss amount would have been $75,397. This fixed-loss amount would have resulted in estimated outlier payments projected to exceed the 7.975 percent statutory target, and we would have used the broad authority conferred upon the Secretary under section 307(b)(1) of the BIPA to make this “adjustment” to “outliers” under the LTCH PPS.
As discussed in greater detail later in this section, with the use of more recent data available for this final rule, our proposed methodology for determining the fixed-loss amount results in a fixed-loss amount of $77,048, which is significantly lower than the fixed-loss amount of $90,921 that we proposed. Given this significant reduction, at this time we do not believe it is necessary or appropriate to use our adjustments authority to adjust outlier payments by using an alternative methodology to set the fixed-loss amount that would not result in total estimated outlier payments being projected to be equal to the statutory target of 7.975 percent in section 1886(m)(7) of the Act. As discussed in the proposed rule ( 88 FR 36592 ), we currently estimate that high-cost outlier payments in both FYs 2023 and 2024 will account for a percentage of total LTCH PPS standard Federal payment rate payments that is much higher than the budget neutral statutory target of 7.975 percent. For example, as discussed in Appendix A to this final rule, based on the most recent available data, we currently model that high-cost outlier payments in FY 2024 will account for 8.8 percent of total LTCH PPS standard Federal payment rate payments. At this time, we believe that using our proposed historical methodology which results in a fixed-loss amount of $77,048 for FY 2025 strikes an appropriate balance between accurately estimating high cost outlier payments and considering the financial effect on LTCHs caused by increases in the fixed-loss amount . We understand commenters' concerns regarding payment stability and access to care under the LTCH PPS, and will continue to consider those issues for future rulemaking.
After consideration of comments received, we are finalizing our proposed methodology for determining the fixed-loss amount for LTCH PPS standard Federal payment rate cases for FY 2025 without modification. In this section of this Addendum, we present the detailed application of our finalized methodology.
When we implemented the LTCH PPS, we established a fixed-loss amount so that total estimated outlier payments are projected to equal 8 percent of total estimated payments (that is, the target percentage) under the LTCH PPS ( 67 FR 56022 through 56026 ). When we implemented the dual rate LTCH PPS payment structure beginning in FY 2016, we established that, in general, the historical LTCH PPS HCO policy would continue to apply to LTCH PPS standard Federal payment rate cases. That is, the fixed-loss amount for LTCH PPS standard Federal payment rate cases would be determined using the LTCH PPS HCO policy adopted when the LTCH PPS was first implemented, but we limited the data used under that policy to LTCH cases that would have been LTCH PPS standard Federal payment rate cases if the statutory changes had been in effect at the time of those discharges.
To determine the applicable fixed-loss amount for LTCH PPS standard Federal payment rate cases, we estimate outlier payments and total LTCH PPS payments for each LTCH PPS standard Federal payment rate case (or for each case that would have been an LTCH PPS standard Federal payment rate case if the statutory changes had been in effect at the time of the discharge) using claims data from the MedPAR files. In accordance with § 412.525(a)(2)(ii), the applicable fixed-loss amount for LTCH PPS standard Federal payment rate cases results in estimated total outlier payments being projected to be equal to 7.975 percent of projected total LTCH PPS payments for LTCH PPS standard Federal payment rate cases.
In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49448 ), we discussed the abnormal charging practices of an LTCH (CCN 312024) in FY 2021 that led to the LTCH receiving an excessive amount of high-cost outlier payments. In that rule, we stated our belief, based on information we received from the provider, that these abnormal charging practices would not persist into FY 2023. Therefore, we did not include their cases in our model for determining the FY 2023 outlier fixed-loss amount. In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59376 ), we stated that the FY 2022 MedPAR claims also reflect the abnormal charging practices of this LTCH. Therefore, we removed claims from CCN 312024 when determining the fixed-loss amount for LTCH PPS standard Federal payment rate cases for FY 2024 and all other FY 2024 ratesetting calculations, including the MS-LTC-DRG relative weights and the calculation of the area wage level adjustment budget neutrality factor. Given recent actions by the Department of Justice regarding CCN 312024 (see https://www.justice.gov/opa/pr/new-jersey-hospital-and-investors-pay-united-states-306-million-alleged-false-claims-related ), as we proposed, we again removed claims from CCN 312024 when determining the fixed-loss amount for LTCH PPS standard Federal payment rate cases for FY 2025 and all other FY 2025 ratesetting calculations, including the MS-LTC-DRG relative weights and the calculation of the area wage level adjustment budget neutrality factor.
Under the LTCH PPS, the cost of each claim is estimated by multiplying the charges on the claim by the provider's CCR. Due to the lag time in the availability of claims data, when estimating costs for the upcoming payment year we typically inflate the charges from the claims data by a uniform factor.
For greater accuracy in calculating the fixed-loss amount, in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45562 through 45566 ), we finalized a technical change to our methodology for determining the charge inflation factor. Similar to the method used under the IPPS hospital payment methodology (as discussed in section II.A.4.i.(2). of this Addendum), our methodology determines the LTCH charge inflation factor based on the historical growth in charges for LTCH PPS standard Federal payment rate cases, calculated using historical MedPAR claims data. In this section of this Addendum, we describe our charge inflation factor methodology.
Step 1 —Identify LTCH PPS Standard Federal Payment Rate Cases
The first step in our methodology is to identify LTCH PPS standard Federal ( print page 69985) payment rate cases from the MedPAR claim files for the two most recently available Federal fiscal year time periods. For both fiscal years, consistent with our historical methodology for determining payment rates for the LTCH PPS, we remove any claims submitted by LTCHs that were all-inclusive rate providers as well as any Medicare Advantage claims. For both fiscal years, we also remove claims from providers that only had claims in one of the fiscal years.
Step 2 —Remove Statistical Outliers
The next step in our methodology is to remove all claims from providers whose growth in average charges was a statistical outlier. We remove these statistical outliers prior to calculating the charge inflation factor because we believe they may represent aberrations in the data that would distort the measure of average charge growth. To perform this statistical trim, we first calculate each provider's average charge in both fiscal years. Then, we calculate a charge growth factor for each provider by dividing its average charge in the most recent fiscal year by its average charge in the prior fiscal year. Then we remove all claims for providers whose calculated charge growth factor was outside 3 standard deviations from the mean provider charge growth factor.
Step 3 —Calculate the Charge Inflation Factor.
The final step in our methodology is to use the remaining claims to calculate a national charge inflation factor. We first calculate the average charge for those remaining claims in both fiscal years. Then we calculate the national charge inflation factor by dividing the average charge in the more recent fiscal year by the average charge in the prior fiscal year.
Following the methodology described previously, as we proposed, we computed a charge inflation factor based on the most recently available data. Specifically, we used the March 2024 update of the FY 2023 MedPAR file and the March 2023 update of the FY 2022 MedPAR as the basis of the LTCH PPS standard Federal payment rate cases for the two most recently available Federal fiscal year time periods, as described previously in our methodology. Therefore, we trimmed the March 2024 update of the FY 2023 MedPAR file and the March 2023 update of the FY 2022 MedPAR file as described in steps 1 and 2 of our methodology. To compute the 1-year average annual rate-of-change in charges per case, we compared the average covered charge per case of $281,402 ($11,630,925,449/41,332 cases) from FY 2022 to the average covered charge per case of $301,946 ($12,740,324,507/42,194 cases) from FY 2023. This rate-of-change was 7.3005 percent, which results in a 1-year charge inflation factor of 1.073005, and a 2-year charge inflation factor of 1.15134 (calculated by squaring the 1-year factor). We inflated the billed charges obtained from the FY 2023 MedPAR file by this 2-year charge inflation factor of 1.15134 when determining the fixed-loss amount for LTCH PPS standard Federal payment rate cases for FY 2025.
For greater accuracy in calculating the fixed-loss amount, in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45562 through 45566 ), we finalized a technical change to our methodology for determining the CCRs used to calculate the fixed-loss amount. Similar to the methodology used for IPPS hospitals (as discussed in section II.A.4.i.(2). of this Addendum), our methodology adjusts CCRs obtained from the best available PSF data by an adjustment factor that is calculated based on historical changes in the average case-weighted CCR for LTCHs. We believe these adjusted CCRs more accurately reflect CCR levels in the upcoming payment year because they account for historical changes in the relationship between costs and charges for LTCHs. In this section of this Addendum, we describe our CCR adjustment factor methodology.
Step 1 —Assign Providers Their Historical CCRs
The first step in our methodology is to identify providers with LTCH PPS standard Federal payment rate cases in the most recent MedPAR claims file (excluding all-inclusive rate providers and providers with only Medicare Advantage claims). For each of these providers, we then identify the CCR from the most recently available PSF. For each of these providers we also identify the CCR from the PSF that was made available one year prior to the most recently available PSF.
Step 2 —Trim Providers with Insufficient CCR Data
The next step in our methodology is to remove from the CCR adjustment factor calculation any providers for which we cannot accurately measure changes to their CCR using the PSF data. We first remove any provider whose CCR was missing in the most recent PSF or prior year PSF. We next remove any provider assigned the statewide average CCR for their State in either the most recent PSF or prior year PSF. We lastly remove any provider whose CCR was not updated between the most recent PSF and prior year PSF (determined by comparing the effective date of the records).
Step 3 —Remove Statistical Outliers
The next step in our methodology is to remove providers whose change in their CCR is a statistical outlier. To perform this statistical trim, for those providers remaining after application of Step 2, we calculate a provider-level CCR growth factor by dividing the provider's CCR from the most recent PSF by its CCR in the prior year's PSF. We then remove any provider whose CCR growth factor was outside 3 standard deviations from the mean provider CCR growth factor. These statistical outliers are removed prior to calculating the CCR adjustment factor because we believe that they may represent aberrations in the data that would distort the measure of average annual CCR change.
Step 4 —Calculate a CCR Adjustment Factor
The final step in our methodology is to calculate, across all remaining providers after application of Step 3, an average case-weighted CCR from both the most recent PSF and prior year PSF. The provider case counts that we use to calculate the case-weighted average are determined from claims for LTCH standard Federal rate cases from the most recent MedPAR claims file. We note when determining these case counts, consistent with our historical methodology for determining the MS-LTC-DRG relative weights, we do not count short stay outlier claims as full cases but instead as a fraction of a case based on the ratio of covered days to the geometric mean length of stay for the MS-LTC-DRG grouped to the case. We calculate the national CCR adjustment factor by dividing the case-weighted CCR from the most recent PSF by the case-weighted CCR from the prior year PSF.
Following the methodology described previously, as we proposed, we computed a CCR adjustment factor based on the most recently available data. Specifically, we used the March 2024 PSF as the most recently available PSF and the March 2023 PSF as the PSF that was made available one year prior to the most recently available PSF, as described in our methodology. In addition, we used claims from the March 2024 update of the FY 2023 MedPAR file in our calculation of average case-weighted CCRs described in Step 4 of our methodology. Specifically, following the methodology described previously and, for providers with LTCH PPS standard Federal payment rate cases in the March 2024 update of the FY 2023 MedPAR file, we ( print page 69986) identified their CCRs from both the March 2023 PSF and March 2024 PSF. After performing the trims outlined in our methodology, we used the LTCH PPS standard Federal payment rate case counts from the FY 2023 MedPAR file (classified using finalized Version 42 of the GROUPER) to calculate case-weighted average CCRs. Based on this data, we calculated a March 2023 national average case-weighted CCR of 0.236968 and a March 2024 national average case-weighted CCR of 0.234910. We then calculated the proposed national CCR adjustment factor by dividing the March 2024 national average case-weighted CCR by the March 2023 national average case-weighted CCR. This results in a proposed 1-year national CCR adjustment factor of 0.991315. When calculating the fixed-loss amount for FY 2025, we assigned the statewide average CCR for the upcoming fiscal year to all providers who were assigned the statewide average in the March 2024 PSF or whose CCR was missing in the March 2024 PSF. For all other providers, we multiplied their CCR from the March 2024 PSF by the 1-year national CCR adjustment factor of 0.991315. We note that the March 2024 PSF national average case-weighted CCR was 1.4 percent lower than the December 2023 PSF national average case-weighted CCR. We also note that the 1-year national adjustment CCR adjustment factor calculated in this final rule is 3.1 percent lower than the 1-year national adjustment CCR factor that we proposed. The incorporation of more recent cost-to-charge ratio data into our payment model was the primary driver of the reduction in the fixed-loss amount calculated in this final rule compared to the fixed-loss amount calculated in the proposed rule.
In this final rule, for FY 2025, using the best available data and the steps described previously, we calculated a fixed-loss amount that would maintain estimated HCO payments at the projected 7.975 percent of total estimated LTCH PPS payments for LTCH PPS standard Federal payment rate cases as required by section 1886(m)(7) of the Act and in accordance with § 412.525(a)(2)(ii) (based on the payment rates and policies for these cases presented in this final rule). Consistent with our historical practice, we use the best available LTCH claims data and CCR data, when determining the fixed-loss amount for LTCH PPS standard Federal payment rate cases for FY 2025 in the final rule. Therefore, based on LTCH claims data from the March 2024 update of the FY 2023 MedPAR file adjusted for charge inflation and adjusted CCRs from the March 2024 update of the PSF, under the broad authority of section 123(a)(1) of the BBRA and section 307(b)(1) of the BIPA, we are establishing a fixed-loss amount for LTCH PPS standard Federal payment rate cases for FY 2025 of $77,048 that will result in estimated outlier payments projected to be equal to 7.975 percent of estimated FY 2025 payments for such cases. As such, we will make an additional HCO payment for the cost of an LTCH PPS standard Federal payment rate case that exceeds the HCO threshold amount that is equal to 80 percent of the difference between the estimated cost of the case and the outlier threshold (the sum of the adjusted LTCH PPS standard Federal payment rate payment and the fixed-loss amount for LTCH PPS standard Federal payment rate cases of $77,048).
When we implemented the application of the site neutral payment rate in FY 2016, in examining the appropriate fixed-loss amount for site neutral payment rate cases issue, we considered how LTCH discharges based on historical claims data would have been classified under the dual rate LTCH PPS payment structure and the CMS' Office of the Actuary projections regarding how LTCHs will likely respond to our implementation of policies resulting from the statutory payment changes. We again relied on these considerations and actuarial projections in FY 2017 and FY 2018 because the historical claims data available in each of these years were not all subject to the LTCH PPS dual rate payment system. Similarly, for FYs 2019 through 2024, we continued to rely on these considerations and actuarial projections because, due to the transitional blended payment policy for site neutral payment rate cases and the provisions of section 3711(b)(2) of the CARES Act, the historical claims data available in each of these years were not subject to the full effect of the site neutral payment rate.
For FYs 2016 through 2024, our actuaries projected that the proportion of cases that would qualify as LTCH PPS standard Federal payment rate cases versus site neutral payment rate cases under the statutory provisions would remain consistent with what is reflected in the historical LTCH PPS claims data. Although our actuaries did not project an immediate change in the proportions found in the historical data, they did project cost and resource changes to account for the lower payment rates. Our actuaries also projected that the costs and resource use for cases paid at the site neutral payment rate would likely be lower, on average, than the costs and resource use for cases paid at the LTCH PPS standard Federal payment rate and would likely mirror the costs and resource use for IPPS cases assigned to the same MS-DRG, regardless of whether the proportion of site neutral payment rate cases in the future remains similar to what is found based on the historical data. As discussed in the FY 2016 IPPS/LTCH PPS final rule ( 80 FR 49619 ), this actuarial assumption is based on our expectation that site neutral payment rate cases would generally be paid based on an IPPS comparable per diem amount under the statutory LTCH PPS payment changes that began in FY 2016, which, in the majority of cases, is much lower than the payment that would have been paid if these statutory changes were not enacted. In light of these projections and expectations, we discussed that we believed that the use of a single fixed-loss amount and HCO target for all LTCH PPS cases would be problematic. In addition, we discussed that we did not believe that it would be appropriate for comparable LTCH PPS site neutral payment rate cases to receive dramatically different HCO payments from those cases that would be paid under the IPPS ( 80 FR 49617 through 49619 and 81 FR 57305 through 57307 ). For those reasons, we stated that we believed that the most appropriate fixed-loss amount for site neutral payment rate cases for FYs 2016 through 2024 would be equal to the IPPS fixed-loss amount for that particular fiscal year. Therefore, we established the fixed-loss amount for site neutral payment rate cases as the corresponding IPPS fixed-loss amounts for FYs 2016 through 2024. In particular, in FY 2024, we established the fixed-loss amount for site neutral payment rate cases as the FY 2024 IPPS fixed-loss amount of $42,750 ( 88 FR 59378 ).
For this final rule, we used FY 2023 data in the FY 2025 LTCH PPS ratesetting. We note that section 3711(b)(2) of the CARES Act provided a waiver of the application of the site neutral payment rate for LTCH cases admitted during the COVID-19 PHE period. The COVID-19 PHE expired on May 11, 2023. Therefore, all LTCH PPS cases in FY 2023 with admission dates on or before the PHE expiration date were paid the LTCH PPS standard Federal rate regardless of whether the ( print page 69987) discharge met the statutory patient criteria. Because not all FY 2023 claims in the data used for this final rule were subject to the site neutral payment rate, we continue to rely on the same considerations and actuarial projections used in FYs 2016 through 2024 when developing a fixed-loss amount for site neutral payment rate cases for FY 2025. Our actuaries continue to project that the costs and resource use for FY 2025 cases paid at the site neutral payment rate would likely be lower, on average, than the costs and resource use for cases paid at the LTCH PPS standard Federal payment rate and will likely mirror the costs and resource use for IPPS cases assigned to the same MS-DRG, regardless of whether the proportion of site neutral payment rate cases in the future remains similar to what was found based on the historical data. (Based on the FY 2023 LTCH claims data used in the development of this final rule, if the provisions of the CARES Act had not been in effect, approximately 71 percent of LTCH cases would have been paid the LTCH PPS standard Federal payment rate and approximately 29 percent of LTCH cases would have been paid the site neutral payment rate for discharges occurring in FY 2023.)
For these reasons, we continue to believe that the most appropriate fixed-loss amount for site neutral payment rate cases for FY 2025 is the IPPS fixed-loss amount for FY 2025. Therefore, for FY 2025, as we proposed, we are establishing that the applicable HCO threshold for site neutral payment rate cases is the sum of the site neutral payment rate for the case and the IPPS fixed-loss amount. That is, we are establishing a fixed-loss amount for site neutral payment rate cases of $46,152, which is the same FY 2025 IPPS fixed-loss amount discussed in section II.A.4.i.(2). of this Addendum. Accordingly, under this policy, for FY 2025, we will calculate an HCO payment for site neutral payment rate cases with costs that exceed the HCO threshold amount that is equal to 80 percent of the difference between the estimated cost of the case and the outlier threshold (the sum of the site neutral payment rate payment and the fixed-loss amount for site neutral payment rate cases of $46,152).
In establishing an HCO policy for site neutral payment rate cases, we established a budget neutrality adjustment under § 412.522(c)(2)(i). We established this requirement because we believed, and continue to believe, that the HCO policy for site neutral payment rate cases should be budget neutral, just as the HCO policy for LTCH PPS standard Federal payment rate cases is budget neutral, meaning that estimated site neutral payment rate HCO payments should not result in any change in estimated aggregate LTCH PPS payments.
To ensure that estimated HCO payments payable to site neutral payment rate cases in FY 2025 would not result in any increase in estimated aggregate FY 2025 LTCH PPS payments, under the budget neutrality requirement at § 412.522(c)(2)(i), it is necessary to reduce site neutral payment rate payments by 5.1 percent to account for the estimated additional HCO payments payable to those cases in FY 2025. Consistent with our historical practice, as we proposed, we are continuing this policy.
As discussed earlier, consistent with the IPPS HCO payment threshold, we estimate the fixed-loss threshold would result in FY 2025 HCO payments for site neutral payment rate cases to equal 5.1 percent of the site neutral payment rate payments that are based on the IPPS comparable per diem amount. As such, to ensure estimated HCO payments payable for site neutral payment rate cases in FY 2025 would not result in any increase in estimated aggregate FY 2025 LTCH PPS payments, under the budget neutrality requirement at § 412.522(c)(2)(i), it is necessary to reduce the site neutral payment rate amount paid under § 412.522(c)(1)(i) by 5.1 percent to account for the estimated additional HCO payments payable for site neutral payment rate cases in FY 2025. To achieve this, for FY 2025, as we proposed, we are applying a budget neutrality factor of 0.949 (that is, the decimal equivalent of a 5.1 percent reduction, determined as 1.0−5.1/100 = 0.949) to the site neutral payment rate for those site neutral payment rate cases paid under § 412.522(c)(1)(i). We note that, consistent with our current policy, this HCO budget neutrality adjustment will not be applied to the HCO portion of the site neutral payment rate amount ( 81 FR 57309 ).
Comment: A few commenters stated that they were concerned with the proposed increase to the fixed-loss amount for site-neutral rate cases.
Response: We acknowledge the commenters' concern. We note that the commenters did not elaborate on the basis for their concerns with the proposed fixed-loss amount for site-neutral rate cases. The commenters also did not suggest any modifications for CMS to make in establishing the fixed-loss amount for site-neutral rate cases in this final rule. Therefore, after consideration of comments received, we are finalizing our proposals as described previously, without modification.
In the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50766 ), we established a policy to reflect the changes to the Medicare IPPS DSH payment adjustment methodology made by section 3133 of the Affordable Care Act in the calculation of the “IPPS comparable amount” under the SSO policy at § 412.529 and the “IPPS equivalent amount” under the site neutral payment rate at § 412.522. Historically, the determination of both the “IPPS comparable amount” and the “IPPS equivalent amount” includes an amount for inpatient operating costs “for the costs of serving a disproportionate share of low-income patients.” Under the statutory changes to the Medicare DSH payment adjustment methodology that began in FY 2014, in general, eligible IPPS hospitals receive an empirically justified Medicare DSH payment equal to 25 percent of the amount they otherwise would have received under the statutory formula for Medicare DSH payments prior to the amendments made by the Affordable Care Act. The remaining amount, equal to an estimate of 75 percent of the amount that otherwise would have been paid as Medicare DSH payments, reduced to reflect changes in the percentage of individuals under the age of 65 who are uninsured, is made available to make additional payments to each hospital that qualifies for Medicare DSH payments and that has uncompensated care. The additional uncompensated care payments are based on the hospital's amount of uncompensated care for a given time period relative to the total amount of uncompensated care for that same time period reported by all hospitals that receive Medicare DSH payments.
To reflect the Medicare DSH payment adjustment methodology statutory changes in section 3133 of the Affordable Care Act in the calculation of the “IPPS comparable amount” and the “IPPS equivalent amount” under the LTCH PPS, we stated in the FY 2014 IPPS/LTCH PPS final rule ( 78 FR 50766 ) that we will include a reduced Medicare DSH payment amount that reflects the projected percentage of the payment amount calculated based on the statutory Medicare DSH payment formula prior to the amendments made by the Affordable Care Act that will be paid to eligible IPPS hospitals as ( print page 69988) empirically justified Medicare DSH payments and uncompensated care payments in that year (that is, a percentage of the operating Medicare DSH payment amount that has historically been reflected in the LTCH PPS payments that are based on IPPS rates). We also stated, in the FY 2014 IPPS/LTC PPS final rule ( 78 FR 50766 ), that the projected percentage will be updated annually, consistent with the annual determination of the amount of uncompensated care payments that will be made to eligible IPPS hospitals. We believe that this approach results in appropriate payments under the LTCH PPS and is consistent with our intention that the “IPPS comparable amount” and the “IPPS equivalent amount” under the LTCH PPS closely resemble what an IPPS payment would have been for the same episode of care, while recognizing that some features of the IPPS cannot be translated directly into the LTCH PPS ( 79 FR 50766 through 50767 ).
As discussed in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36593 through 36594 ), for FY 2025, based on the most recent data available at that time, we proposed to establish that the calculation of the “IPPS comparable amount” under § 412.529 would include an applicable operating Medicare DSH payment amount that is equal to 71.61 percent of the operating Medicare DSH payment amount that would have been paid based on the statutory Medicare DSH payment formula absent the amendments made by the Affordable Care Act. Furthermore, consistent with our historical practice, we proposed that, if more recent data became available, we would use that data to determine the applicable operating Medicare DSH payment amount used to calculate the “IPPS comparable amount” in the final rule.
We did not receive any public comments in response to our proposal, and as such are finalizing this proposal. However, as we proposed, we are determining the applicable operating Medicare DSH payment amount used to calculate the “IPPS comparable amount” in this final rule using more recent data.
For FY 2025, as discussed in greater detail in section IV.E.2.b. of the preamble of this final rule, based on the most recent data available, our estimate of 75 percent of the amount that would otherwise have been paid as Medicare DSH payments (under the methodology outlined in section 1886(r)(2) of the Act) is adjusted to 54.29 percent of that amount to reflect the change in the percentage of individuals who are uninsured. The resulting amount is then used to determine the amount available to make uncompensated care payments to eligible IPPS hospitals in FY 2025. In other words, the amount of the Medicare DSH payments that would have been made prior to the amendments made by the Affordable Care Act is adjusted to 40.72 percent (the product of 75 percent and 54.29 percent) and the resulting amount is used to calculate the uncompensated care payments to eligible hospitals. As a result, for FY 2025, we project that the reduction in the amount of Medicare DSH payments pursuant to section 1886(r)(1) of the Act, along with the payments for uncompensated care under section 1886(r)(2) of the Act, will result in overall Medicare DSH payments of 65.72 percent of the amount of Medicare DSH payments that would otherwise have been made in the absence of the amendments made by the Affordable Care Act (that is, 25 percent + 40.72 percent = 65.72 percent).
Therefore, for FY 2025, consistent with our proposal, we are establishing that the calculation of the “IPPS comparable amount” under § 412.529 will include an applicable operating Medicare DSH payment amount that is equal to 65.72 percent of the operating Medicare DSH payment amount that would have been paid based on the statutory Medicare DSH payment formula absent the amendments made by the Affordable Care Act.
Under the dual rate LTCH PPS payment structure, only LTCH PPS cases that meet the statutory criteria to be excluded from the site neutral payment rate are paid based on the LTCH PPS standard Federal payment rate. Under § 412.525(c), the LTCH PPS standard Federal payment rate is adjusted to account for differences in area wages; we make this adjustment by multiplying the labor-related share of the LTCH PPS standard Federal payment rate for a case by the applicable LTCH PPS wage index (the final FY 2025 values are shown in Tables 12A through 12B listed in section VI. of this Addendum and are available via the internet on the CMS website). The LTCH PPS standard Federal payment rate is also adjusted to account for the higher costs of LTCHs located in Alaska and Hawaii by the applicable COLA factors (the final FY 2025 factors are shown in the chart in section V.C. of this Addendum) in accordance with § 412.525(b). In this final rule, we are establishing an LTCH PPS standard Federal payment rate for FY 2025 of $49,383.26, as discussed in section V.A. of this Addendum. We illustrate the methodology to adjust the LTCH PPS standard Federal payment rate for FY 2025, applying our finalized LTCH PPS amounts for the standard Federal payment rate, MS-LTC-DRG relative weights, and wage index in the following example:
During FY 2025, a Medicare discharge that meets the criteria to be excluded from the site neutral payment rate, that is, an LTCH PPS standard Federal payment rate case, is from an LTCH that is located in CBSA 16984, which has a FY 2025 LTCH PPS wage index value of 1.0207 (as shown in Table 12A listed in section VI. of this Addendum). The Medicare patient case is classified into MS-LTC-DRG 189 (Pulmonary Edema Respiratory Failure), which has a relative weight for FY 2025 of 0.9787 (as shown in Table 11 listed in section VI. of this Addendum). The LTCH submitted quality reporting data for FY 2025 in accordance with the LTCH QRP under section 1886(m)(5) of the Act.
To calculate the LTCH's total adjusted Federal prospective payment for this Medicare patient case in FY 2025, we computed the wage-adjusted Federal prospective payment amount by multiplying the unadjusted FY 2025 LTCH PPS standard Federal payment rate ($49,383.26) by the labor-related share (72.8 percent) and the wage index value (1.0207). This wage-adjusted amount was then added to the nonlabor-related portion of the unadjusted LTCH PPS standard Federal payment rate (27.2 percent; adjusted for cost of living, if applicable) to determine the adjusted LTCH PPS standard Federal payment rate, which is then multiplied by the MS-LTC-DRG relative weight (0.9787) to calculate the total adjusted LTCH PPS standard Federal prospective payment for FY 2025 ($49,059.74). The table illustrates the components of the calculations in this example.
| Unadjusted LTCH PPS Standard Federal Prospective Payment Rate | $49,383.26 |
| Labor-Related Share | × 0.728 |
| Labor-Related Portion of the LTCH PPS Standard Federal Payment Rate | = $35,951.01 |
| Wage Index (CBSA 16984) | × 1.0207 |
| Wage-Adjusted Labor Share of the LTCH PPS Standard Federal Payment Rate | = $36,695.20 |
| ( print page 69989) | |
| Nonlabor-Related Portion of the LTCH PPS Standard Federal Payment Rate ($49,383.26 × 0.272) | + $13,432.25 |
| Adjusted LTCH PPS Standard Federal Payment Amount | = $50,127.45 |
| MS-LTC-DRG 189 Relative Weight | × 0.9787 |
| Total Adjusted LTCH PPS Standard Federal Prospective Payment | = $49,059.74 |
This section lists the tables referred to throughout the preamble of this final rule and in the Addendum. In the past, a majority of these tables were published in the Federal Register as part of the annual proposed and final rules. However, similar to FYs 2012 through 2024, for the FY 2025 rulemaking cycle, the IPPS and LTCH PPS tables will not be published in the Federal Register in the annual IPPS/LTCH PPS proposed and final rules and will be on the CMS website. Specifically, all IPPS tables listed in the final rule, with the exception of IPPS Tables 1A, 1B, 1C, and 1D, and LTCH PPS Table 1E, will generally be available on the CMS website. IPPS Tables 1A, 1B, 1C, and 1D, and LTCH PPS Table 1E are displayed at the end of this section and will continue to be published in the Federal Register as part of the annual proposed and final rules.
Tables 7A and 7B historically contained the Medicare prospective payment system selected percentile lengths of stay for the MS-DRGs for the prior year and upcoming fiscal year. We note, in the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49452 ), we finalized beginning with FY 2023, to provide the percentile length of stay information previously included in Tables 7A and 7B in the supplemental AOR/BOR data file. The AOR/BOR files can be found on the FY 2025 IPPS final rule home page on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html .
After hospitals have been given an opportunity to review and correct their calculations for FY 2025, we will post Table 15 (which will be available via the CMS website) to display the final FY 2025 readmissions payment adjustment factors that will be applicable to discharges occurring on or after October 1, 2024. We expect Table 15 will be posted on the CMS website in the Fall 2024.
Readers who experience any problems accessing any of the tables that are posted on the CMS websites identified in this final rule should contact Michael Treitel at (410) 786-4552.
The following IPPS tables for this final rule are generally available on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/index.html . Click on the link on the left side of the screen titled “FY 2025 IPPS Final Rule Home Page” or “Acute Inpatient—Files—for Download.”
Table 2.—Final Case-Mix Index and Wage Index Table by CCN—FY 2025 Final Rule
Table 3.—Final Wage Index Table by CBSA—FY 2025 Final Rule
Table 4A.—Final List of Counties Eligible for the Out-Migration Adjustment Under Section 1886(d)(13) of the Act—FY 2025 Final Rule
Table 4B.—Final Counties Redesignated Under Section 1886(d)(8)(B) of the Act (LUGAR Counties)—FY 2025 Final Rule
Table 5.—Final List of Medicare Severity Diagnosis-Related Groups (MS-DRGs), Relative Weighting Factors, and Geometric and Arithmetic Mean Length of Stay—FY 2025 Final Rule
Table 6A.—New Diagnosis Codes—FY 2025
Table 6B.—New Procedure Codes—FY 2025
Table 6C.—Invalid Diagnosis Codes—FY 2025
Table 6D.—Invalid Procedure Codes—FY 2025
Table 6E.—Revised Diagnosis Code Titles—FY 2025
Table 6F.—Revised Procedure Code Titles—FY 2025
Table 6G.1.—Secondary Diagnosis Order Additions to the CC Exclusions List—FY 2025
Table 6G.2.—Principal Diagnosis Order Additions to the CC Exclusions List—FY 2025
Table 6H.1.—Secondary Diagnosis Order Deletions to the CC Exclusions List—FY 2025
Table 6H.2.—Principal Diagnosis Order Deletions to the CC Exclusions List—FY 2025
Table 6I.—Complete MCC List—FY 2025
Table 6I.1.—Additions to the MCC List—FY 2025
Table 6J.—Complete CC List—FY 2025
Table 6J.1.—Additions to the CC List—FY 2025
Table 6J.2.—Deletions to the CC List—FY 2025
Table 6K.—Complete CC Exclusions List—FY 2025
Table 6P.—ICD-10-CM and ICD-10-PCS Codes for Final MS-DRG Changes and Analysis With Application of the NonCC Subgroup Criteria—FY 2025 (Table 6P contains multiple tables, 6P.1a. through 6P.4d that include the ICD-10-CM and ICD-10-PCS code lists relating to specific final MS-DRG changes or other analyses). These tables are referred to throughout section II.C. of the preamble of this final rule.
Table 8A.—Final FY 2025 Statewide Average Operating Cost-to-Charge Ratios (CCRs) for Acute Care Hospitals (Urban and Rural)
Table 8B.—Final FY 2025 Statewide Average Capital Cost-to-Charge Ratios (CCRs) for Acute Care Hospitals
Table 16A.—Updated Proxy Hospital Value-Based Purchasing (VBP) Program Adjustment Factors for FY 2025
Table 18.—Final FY 2025 Medicare DSH Uncompensated Care Payment Factor 3
The following LTCH PPS tables for this FY 2025 final rule are available through the internet on the CMS website at https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/LongTermCareHospitalPPS/index.html under the list item for Regulation Number CMS-1808-F:
Table 8C.—Final FY 2025 Statewide Average Total Cost-to-Charge Ratios (CCRs) for LTCHs (Urban and Rural)
Table 11.—Final MS-LTC-DRGs, Relative Weights, Geometric Average Length of Stay, and Short-Stay Outlier (SSO) Threshold for LTCH PPS Discharges Occurring From October 1, 2024, Through September 30, 2025
Table 12A.—Final LTCH PPS Wage Index for Urban Areas for Discharges Occurring From October 1, 2024, Through September 30, 2025
Table 12B.—Final LTCH PPS Wage Index for Rural Areas for Discharges Occurring From October 1, 2024, Through September 30, 2025

This final rule is necessary to make payment and policy changes under the IPPS for Medicare acute care hospital inpatient services for operating and capital-related costs as well as for certain hospitals and hospital units excluded from the IPPS. This final rule also is necessary to make payment and policy changes for Medicare hospitals under the LTCH PPS. Also, as we note later in this Appendix, the primary objective of the IPPS and the LTCH PPS is to create incentives for hospitals to operate efficiently and minimize unnecessary costs, while at the same time ensuring that payments are sufficient to adequately compensate hospitals for their legitimate costs in delivering necessary care to Medicare beneficiaries. In addition, we share national goals of preserving the Medicare Hospital Insurance Trust Fund.
We believe that the changes in this final rule, such as the updates to the IPPS and LTCH PPS rates, and the final policies and discussions relating to applications for new technology add-on payments, are needed to further each of these goals while maintaining the financial viability of the hospital industry and ensuring access to high quality health care for Medicare beneficiaries.
We expect that these changes would ensure that the outcomes of the prospective payment systems are reasonable and provide equitable payments, while avoiding or minimizing unintended adverse consequences.
In accordance with section 1886(b)(3)(B) of the Act and as described in section V.B. of the preamble to this final rule, we are updating the national standardized amount for inpatient hospital operating costs by the applicable percentage increase of 2.9 percent (that is, a 3.4 percent market basket update with a reduction of 0.5 percentage point for the productivity adjustment). We are also applying the applicable percentage increase (including the market basket update and the productivity adjustment) to the hospital-specific rates.
Subsection (d) hospitals that do not submit quality information under rules established by the Secretary and that are meaningful EHR users under section 1886(b)(3)(B)(ix) of the Act would receive an applicable percentage increase of 2.05 percent which reflects a one-quarter percent reduction of the market basket update for failure to submit quality data. Hospitals that are identified as not meaningful EHR users and do submit quality information under section 1886(b)(3)(B)(viii) of the Act would receive an applicable percentage increase of 0.35 percent which reflects a three-quarter percent reduction of the market basket update for being identified as not a meaningful EHR user.
Hospitals that are identified as not meaningful EHR users under section 1886(b)(3)(B)(ix) of the Act and also do not submit quality data under section 1886(b)(3)(B)(viii) of the Act would receive an applicable percentage increase of −0.5 percent, which reflects a one-quarter percent reduction of the market basket update for failure to submit quality data and a three-quarter percent reduction of the market basket update for being identified as not a meaningful EHR user.
Consistent with sections 1886(d)(5)(K) and (L) of the Act, we review applications for new technology add-on payments based on the eligibility criteria at 42 CFR 412.87 . As set forth in 42 CFR 412.87(f)(1) , we consider whether a technology meets the criteria for the new technology add-on payment and announce the results as part of the annual updates and changes to the IPPS. New technology add-on payments are not budget neutral.
As discussed in section II.E.8. of the preamble of this final rule, we are finalizing our proposal that, beginning with new technology add-on payments for FY 2026, in assessing whether to continue the new technology add-on payments for those technologies that are first approved for new technology add-on payments in FY 2025 or a subsequent year, we will extend new technology add-on payments for an additional fiscal year when the 3-year anniversary date of the product's entry onto the U.S. market occurs on or after October 1 of the upcoming fiscal year. For technologies that were first approved for new technology add-on payments prior to FY 2025, including for technologies we determine to be substantially similar to those technologies, we will continue to use the midpoint of the upcoming fiscal year (April 1) when determining whether a technology would still be considered “new” for purposes of new technology add-on payments. Similarly, we are also finalizing that beginning with applications for new technology add-on payments for FY 2026, we will use the start of the fiscal year (October 1) instead of April 1 to determine whether to approve new technology add-on payment for that fiscal year. We note that this change will be effective beginning with new technology add-on payments for FY 2026, and there would be no impact of this change in FY 2025. For purposes of estimating the impact of our finalized changes to the calculation of the inpatient new technology add-on payment—under the assumption that all of the FY 2025 new technology add-on payment applications that have been FDA-approved or -cleared or have a documented delay in market availability between October 1, 2023, and March 30, 2024 (as discussed in section II.E.5. and section II.E.6. of the preamble of this final rule), and that are first approved for new technology add-on payments in FY 2025, would continue to meet the specified criteria for new technology add-on payments for FY 2026 and FY 2027—this policy would increase IPPS spending by approximately $459 million in FY 2027. Because it is difficult to predict the actual new technology add-on payment for each case, the estimated impact in this final rule is based on the applicants' estimated cost and volume projections at the time they submitted their application (or based on updated figures provided during the public comment period) and as if every claim that would qualify for a new technology add-on payment would receive the maximum add-on payment.
As discussed in section II.E.9. of the preamble of this final rule, we are finalizing our proposal that beginning with new technology add-on payment applications for FY 2026, we will no longer consider a hold status to be an inactive status for the purposes of eligibility for the new technology add-on payment under our existing policy for technologies that are not already FDA market authorized for the indication that is the subject of the new technology add-on payment application. Under existing policy, applicants must have a complete and active FDA market authorization request at the time of new technology add-on payment application submission and must provide documentation of FDA acceptance (for a 510k application or De Novo Classification request) or filing (for a PMA, NDA, or BLA) to CMS at the time of application submission, consistent with the type of FDA marketing authorization application the applicant has submitted to FDA. We note that the cost impact of this proposal is not estimable. We expect that some applicants who were ineligible in FY 2025 may apply for new technology add-on payments for FY 2026.
As discussed in section II.E.10. of the preamble of this final rule, we are finalizing our proposal that, subject to our review of the new technology add-on payment eligibility criteria, for a gene therapy approved for new technology add-on payments in the FY 2025 IPPS/LTCH PPS final rule that is indicated and used specifically for the treatment of sickle cell disease (SCD), effective with discharges on or after October 1, 2024 and concluding at the end of the 2- to 3-year newness period for such therapy, if the costs of a discharge (determined by applying CCRs as described in § 412.84(h)) involving the use of such therapy for the treatment of SCD exceed the full DRG payment (including payments for IME and DSH, but excluding outlier payments), Medicare will make an add-on payment equal to the lesser of: (1) 75 percent of the costs of the new medical service or technology; or (2) 75 percent of the amount by which the costs of the case exceed the standard DRG payment. We estimate that for the two gene therapy technologies that are approved for new technology add-on payments in this final rule that are indicated for and used in the treatment of SCD (as discussed in section II.E.5. of the preamble of this final rule), these changes to the calculation of the inpatient new technology add-on payment will increase IPPS spending by approximately $38 million in FY 2025. Because it is difficult to predict the actual new technology add-on payment for each case, the estimated impact in this final rule is based on the applicants' estimated cost and volume projections at the time they submitted their application and as if every claim that would qualify for a new technology add-on payment would receive the maximum add-on payment. ( print page 69992)
To help mitigate wage index disparities between high wage and low wage hospitals, in the FY 2020 IPPS/LTCH PPS rule ( 84 FR 42326 through 42332 ), we adopted a policy to increase the wage index values for certain hospitals with low wage index values (the low wage index hospital policy). This policy was adopted in a budget neutral manner through an adjustment applied to the standardized amounts for all hospitals. We indicated our intention that this policy would be effective for at least 4 years, beginning in FY 2020, to allow employee compensation increases implemented by these hospitals sufficient time to be reflected in the wage index calculation. We also stated we intended to revisit the issue of the duration of this policy in future rulemaking as we gained experience under the policy. As discussed in section III.G.5. of the preamble of this final rule, while we are using the FY 2021 cost report data for the FY 2025 wage index, we are unable to comprehensively evaluate the effect, if any, the low wage index hospital policy had on hospitals' wage increases during the years the COVID-19 PHE was in effect. We believe it is necessary to wait until we have useable data from fiscal years after the PHE before reaching any conclusions about the efficacy of the policy. Therefore, for FY 2025, we are finalizing that the low wage index hospital policy and the related budget neutrality adjustment would be effective for at least 3 more years, beginning in FY 2025.
As discussed in section V.G.2. of the preamble of this final rule, we are finalizing our proposal to implement section 4122 of the Consolidated Appropriations Act (CAA) of 2023. Section 4122(a) of the CAA, 2023, amended section 1886(h) of the Act by adding a new section 1886(h)(10) of the Act requiring the distribution of additional residency positions (also referred to as slots) to hospitals. Section 4122 of the CAA of 2023 makes available 200 residency positions, to be distributed beginning in FY 2026, with priority given to hospitals in 4 statutorily specified categories. At least 100 of the 200 residency positions made available under section 4122 of the CAA of 2023 shall be distributed for psychiatry or psychiatry subspecialty residency training programs. We expect these changes will make appropriate Medicare GME payments to hospitals for Medicare's share of the direct costs to operate the hospital's approved medical residency program, and for IPPS hospitals the indirect costs associated with residency programs that may result in higher patient care costs, consistent with the law. We expect that these changes will ensure that the outcomes of these Medicare payment policies are reasonable and provide equitable payments, while avoiding or minimizing unintended adverse consequences.
In this final rule, as required by section 1886(r)(2) of the Act, we are updating our estimates of the 3 factors used to determine uncompensated care payments for FY 2025. Beginning with FY 2023, we adopted a multiyear averaging methodology to determine Factor 3 of the uncompensated care payment methodology, which would help to mitigate against large fluctuations in uncompensated care payments from year to year. Under this methodology, for FY 2025 and subsequent fiscal years, we would determine Factor 3 for all eligible hospitals using a 3-year average of the data on uncompensated care costs from Worksheet S-10 for the 3 most recent fiscal years for which audited data are available. Specifically, we would use a 3-year average of audited data on uncompensated care costs from Worksheet S-10 from the FY 2019, FY 2020, and FY 2021 cost reports to calculate Factor 3 for FY 2025 for all eligible hospitals.
Beginning with FY 2023 ( 87 FR 49047 through 49051 ), we also established a supplemental payment for IHS and Tribal hospitals and hospitals located in Puerto Rico. In section IV.D. of the preamble of this final rule, we summarize the ongoing methodology for supplemental payments.
The Rural Community Hospital Demonstration (RCHD) was authorized originally for a 5-year period by section 410A of the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA) ( Pub. L. 108-173 ), and it was extended for another 5-year period by section 3123 and 10313 of the Affordable Care Act ( Pub. L. 111-148 ). Section 15003 of the 21st Century Cures Act (Cures Act) ( Pub. L. 114-255 ) extended the demonstration for an additional 5-year period, and section 128 of the Consolidated Appropriations Act of 2021 ( Pub. L. 116-159 ) included an additional 5-year re-authorization. CMS has conducted the demonstration since 2004, which allows enhanced, cost-based payment for Medicare inpatient services for up to 30 small rural hospitals.
The authorizing legislation imposes a strict budget neutrality requirement. In this final rule, we summarize the status of the demonstration program, and the ongoing methodologies for implementation and budget neutrality.
The Frontier Community Health Integration Project (FCHIP) demonstration was authorized under section 123 of the Medicare Improvements for Patients and Providers Act of 2008 ( Pub. L. 110-275 ), as amended by section 3126 of the Affordable Care Act of 2010 ( Pub. L. 114-158 ), and most recently re-authorized and extended by the Consolidated Appropriations Act of 2021 ( Pub. L. 116-260 ). The legislation authorized a demonstration project to allow eligible entities to develop and test new models for the delivery of health care in order to improve access to and better integrate the delivery of acute care, extended care and other health care services to Medicare beneficiaries in certain rural areas. The FCHIP demonstration initial period was conducted in 10 critical access hospitals (CAHs) from August 1, 2016, to July 31, 2019, and the demonstration “extension period” began on January 1, 2022, to run through June 30, 2027.
The authorizing legislation requires the FCHIP demonstration to be budget neutral. In this final rule, we proposed to continue with the budget neutrality approach used in the demonstration initial period for the demonstration extension period—to offset payments across CAHs nationally—should the demonstration incur costs to Medicare.
As discussed in section VIII.D. of the preamble of this final rule, we are rebasing and revising the 2017-based LTCH market basket to reflect a 2022 base year. The update to the LTCH PPS standard Federal payment rate for FY 2025 is discussed in section VIII.C.2. of the preamble of this final rule. For FY 2025, we are updating the LTCH PPS standard Federal payment rate by 3.0 percent (that is, a 3.5 percent market basket update with a reduction of 0.5 percentage point for the productivity adjustment, as required by section 1886(m)(3)(A)(i) of the Act). LTCHs that failed to submit quality data, as required by 1886(m)(5)(A)(i) of the Act would receive an update of 1.0 percent for FY 2025, which reflects a 2.0 percentage point reduction for failure to submit quality data.
Section 1886(b)(3)(B)(viii) of the Act requires subsection (d) hospitals to report data in accordance with the requirements of the Hospital IQR Program for purposes of measuring and making publicly available information on health care quality and links the quality data submission to the annual applicable percentage increase. Sections 1886(b)(3)(B)(ix), 1886(n), and 1814(l) of the Act require eligible hospitals and CAHs to demonstrate they are meaningful users of certified EHR technology for purposes of electronic exchange of health information to improve the quality of health care and links the submission of information demonstrating meaningful use to the annual applicable percentage increase for eligible hospitals and the applicable percent for CAHs. Section 1886(m)(5) of the Act requires each LTCH to submit quality measure data in accordance with the requirements of the LTCH QRP for purposes of measuring and making publicly available information on health care quality, and in order to avoid a 2-percentage point reduction. Section 1886(o) of the Act requires the Secretary to establish a value-based purchasing program under which value-based incentive payments are made in a fiscal year to hospitals that meet the performance standards established on an announced set of quality and efficiency measures for the fiscal year. The purposes of the Hospital VBP Program include measuring the quality of hospital inpatient care, linking hospital measure performance to payment, and making publicly available information on hospital quality of care. Section 1886(p) of the Act requires a reduction in payment ( print page 69993) for subsection (d) hospitals that rank in the worst-performing 25 percent with respect to measures of hospital-acquired conditions under the HAC Reduction Program for the purpose of measuring HACs, linking measure performance to payment, and making publicly available information on health care quality. Section 1886(q) of the Act requires a reduction in payment for subsection (d) hospitals for excess readmissions based on measures for applicable conditions under the Hospital Readmissions Reduction Program for the purpose of measuring readmissions, linking measure performance to payment, and making publicly available information on health care quality. Section 1866(k) of the Act applies to hospitals described in section 1886(d)(1)(B)(v) of the Act (referred to as “PPS-exempt cancer hospitals” or “PCHs”) and requires PCHs to report data in accordance with the requirements of the PCHQR Program for purposes of measuring and making publicly available information on the quality of care furnished by PCHs. However, there is no reduction in payment to a PCH that does not report data.
In section X.A. of the preamble of this final rule, we are testing a new alternative payment model called the Transforming Episode Accountability Model (TEAM). Section 1115A of the Act authorizes the testing of innovative payment and service delivery models that preserve or enhance the quality of care furnished to Medicare, Medicaid, and CHIP beneficiaries while reducing program expenditures. The underlying issue addressed by the model is that under FFS, Medicare makes separate payments to providers and suppliers for items and services furnished to a beneficiary over the course of an episode. Because providers and suppliers are paid for each individual item or service delivered, this may lead to care that is fragmented, unnecessary or duplicative, while making it challenging to invest in quality improvement or care coordination that would maximize patient benefit. We anticipate the model may reduce costs while maintaining or improving quality of care by bundling payment for items and services for a given episode and holding TEAM participants accountable for spending and quality performance, as well as by providing incentives to promote high quality and efficient care.
This final rule will create and test an episode-based payment model under the authority at section 1115A of the Act in which selected acute care hospitals, located within the mandatory Core Based Statistical Areas (CBSAs) that CMS selected for model implementation, will be required to participate. CMS will allow a one-time opportunity for hospitals that participate until the last day of the last performance period in the BPCI Advanced model or the last day of the last performance year of the CJR model, that are not located in a mandatory CBSA selected for TEAM participation to voluntarily opt into TEAM. [ 1108 ] The model builds on and incorporates certain model features from other CMS Innovation Center episode-based payment models such as the BPCI Advanced Model and the CJR Model. Testing this new model allows us to learn more about the patterns of potentially inefficient utilization of health care services, as well as how to improve the beneficiary care experience during care transitions and incentivize quality improvements for common surgical episodes. This information may inform future Medicare payment policy and potentially establish the framework for managing clinical episodes as a standard practice in Traditional Medicare.
Under the model, acute care hospitals will be accountable for five episode categories: coronary artery bypass graft, lower extremity joint replacement, major bowel procedure, surgical hip/femur fracture treatment excluding lower extremity joint replacement, and spinal fusion. We believe the model may benefit Medicare beneficiaries through improving the coordination of items and services paid for through Medicare FFS payments, encouraging provider investment in health care infrastructure and redesigned care processes, and incentivizing higher value care across the inpatient and post-acute care settings for the episode. The model will also provide an opportunity to evaluate the nature and extent of reductions in the cost of treatment by providing financial incentives for providers to coordinate their efforts to meet patient needs and prevent future costs. The model may benefit beneficiaries by holding hospitals accountable for the quality and cost of care for 30 day episodes after a beneficiary is discharged from the inpatient stay or hospital outpatient procedure, which could encourage investment in infrastructure and redesigned care processes the promote high quality and efficient service delivery that focuses on patient-centered care.
We received no comments on the statement of need and therefore are finalizing this provision without modification.
Section 1878 of the Act ( 42 U.S.C. 1395oo ) established by the Social Security Amendments of 1972, requires the Secretary to appoint individuals to the PRRB for a 3-year term of office. In regulations promulgated after the enactment of this provision, 42 CFR 405.1845 stipulated that no member shall serve more than two consecutive 3-year terms of office. In section X.B. of the preamble of this final rule, we finalize our proposal to increase from two to three the number of consecutive terms that a PRRB Member is eligible to serve. We believe that extending the length of service of Board Members could have an increased effect on the PRRB's productivity and efficiency as well as increase the number of individuals who seek a position on the PRRB.
Section 202 of the Further Consolidated Appropriations Act of 2020 (CAA; Pub. L. 116-94 ) amended Medicaid program integrity requirements in Puerto Rico. Puerto Rico was required to publish a plan, developed by Puerto Rico in coordination with CMS, and approved by the CMS Administrator, not later than 18 months after the CAA's enactment, for how Puerto Rico would develop measures to comply with the PERM requirements of 42 CFR part 431, subpart Q . Puerto Rico published this plan on June 20, 2021, that was approved by the CMS Administrator on June 22, 2021.
In section X.E. of the preamble of this final rule, we discuss the proposal to remove the exclusion of Puerto Rico from the PERM program found at 42 CFR 431.954(b)(3) . In compliance with section 202 of the CAA, Puerto Rico has developed measures to comply with the PERM requirements of 42 CFR part 431, subpart Q . Therefore, we proposed that the PERM program become applicable to Puerto Rico. We are finalizing our proposal and believe that including Puerto Rico in the PERM program will increase visibility into its Medicaid and CHIP operations and improve its program integrity efforts, that protect taxpayer dollars from improper payments.
Under sections 1861(e)(9) and 1820(e)(3) of the Act, hospitals and CAHs, respectively, under the Medicare and Medicaid programs must meet standards for the health and safety of patients receiving services in those facilities. Rules issued under that statutory authority require such facilities to engage in the surveillance, prevention, and control of health care-associated acute respiratory illnesses. In 2020, we published detailed reporting standards related specifically to COVID-19 for hospitals and CAHs. Those standards sunset on April 30, 2024. In section X.F. of the preamble of this final rule, we will establish streamlined standards that apply to a range of acute respiratory illnesses, not just to COVID-19, and will contribute to the ability to combat potential future threats from either existing or potential future sources of such infections.
We have examined the impacts of this final rule as required by Executive Order 12866 on Regulatory Planning and Review (September 30, 1993), Executive Order 13563 on Improving Regulation and Regulatory Review (January 18, 2011), Executive Order 14094 on Modernizing Regulatory Review (April 6, 2023), the Regulatory Flexibility Act (RFA) (September 19, 1980, Pub. L. 96-354), section 1102(b) of the Act, section 202 of the Unfunded Mandates Reform Act of 1995 (March 22, 1995; Pub. L. 104-4 ), Executive Order 13132 on Federalism (August 4, 1999), and the Congressional Review Act (CRA) ( 5 U.S.C. 804(2) ).
Executive Orders 12866 and 13563 direct agencies to assess all costs and benefits of available regulatory alternatives and, if regulation is necessary, to select regulatory approaches that maximize net benefits (including potential economic, environmental, public health and safety effects, distributive impacts, and equity). Executive Order 14094 amends section 3(f) of Executive Order 12866 to define a ( print page 69994) “significant regulatory action” as any regulatory action that is likely to result in a rule that may: (1) have an annual effect on the economy of $200 million or more in any 1 year, or adversely affect in a material way the economy, productivity, competition, jobs, the environment, public health or safety, or state, local, territorial, or tribal governments or communities; (2) create a serious inconsistency or otherwise interfere with an action taken or planned by another agency; (3) materially alter the budgetary impacts of entitlement grants, user fees, or loan programs or the rights and obligations of recipients thereof; or (4) raise legal or policy issues for which centralized review would meaningfully further the President's priorities or the principles set forth in this Executive Order.
A regulatory impact analysis (RIA) must be prepared for a regulatory action that is significant under section 3(f)(1). Based on our estimates, OMB'S Office of Information and Regulatory Affairs (OIRA) has determined this rulemaking is significant under section 3(f)(1) of E.O. 12866 . Accordingly, we have prepared a regulatory impact analysis that to the best of our ability presents the costs and benefits of the rulemaking. Pursuant to Subtitle E of the Small Business Regulatory Enforcement Fairness Act of 1996 (also known as the Congressional Review Act), OIRA has also determined that this rule meets the criteria set forth in 5 U.S.C. OMB has reviewed these regulations, and the Departments have provided the following assessment of their impact.
We estimate that the changes for FY 2025 acute care hospital operating and capital payments would redistribute amounts in excess of $200 million to acute care hospitals. The applicable percentage increase to the IPPS rates required by the statute, in conjunction with other payment changes in this final rule, would result in an estimated $2.9 billion increase in FY 2025 payments, primarily driven by the changes in FY 2025 operating payments, including uncompensated care payments, FY 2025 capital payments, the expiration of the temporary changes in the low-volume hospital program and the expiration of the MDH program. These changes are relative to payments made in FY 2024. The impact analysis of the capital payments can be found in section I.I. of the Appendix in this final rule. In addition, as described in section I.J. of this Appendix, LTCHs are expected to experience an increase in payments by approximately $58 million in FY 2025 relative to FY 2024.
Our operating payment impact estimate includes the 2.9 percent hospital update to the standardized amount (reflecting the 3.4 percent market basket update reduced by the 0.5 percentage point productivity adjustment). The estimates of IPPS operating payments to acute care hospitals do not reflect any changes in hospital admissions or real case-mix intensity, which would also affect overall payment changes.
The analysis in this Appendix, in conjunction with the remainder of this document, demonstrates that this final rule is consistent with the regulatory philosophy and principles identified in Executive Orders 12866 and 13563, the RFA, and section 1102(b) of the Act. This final rule would affect payments to a substantial number of small rural hospitals, as well as other classes of hospitals, and the effects on some hospitals may be significant. Finally, in accordance with the provisions of Executive Order 12866 , the Office of Management and Budget has reviewed this final rule.
The primary objective of the IPPS and the LTCH PPS is to create incentives for hospitals to operate efficiently and minimize unnecessary costs, while at the same time ensuring that payments are sufficient to adequately compensate hospitals for their costs in delivering necessary care to Medicare beneficiaries. In addition, we share national goals of preserving the Medicare Hospital Insurance Trust Fund.
We believe that the changes in this final rule would further each of these goals while maintaining the financial viability of the hospital industry and ensuring access to high quality health care for Medicare beneficiaries. We expect that these changes would ensure that the outcomes of the prospective payment systems are reasonable and equitable, while avoiding or minimizing unintended adverse consequences.
Because this final rule contains a range of policies, we refer readers to the section of the final rule where each policy is discussed. These sections include the rationale for our decisions, including the need for the policy.
The following quantitative analysis presents the projected effects of our policy changes, as well as statutory changes effective for FY 2025, on various hospital groups. We estimate the effects of individual policy changes by estimating payments per case, while holding all other payment policies constant. We use the best data available, but, generally unless specifically indicated, we do not attempt to make adjustments for future changes in such variables as admissions, lengths of stay, case mix, changes to the Medicare population, or incentives. In addition, we discuss limitations of our analysis for specific policies in the discussion of those policies as needed.
The prospective payment systems for hospital inpatient operating and capital related- costs of acute care hospitals encompass most general short-term, acute care hospitals that participate in the Medicare program. There were 25 Indian Health Service hospitals in our database, which we excluded from the analysis due to the special characteristics of the prospective payment methodology for these hospitals. Among other short term, acute care hospitals, hospitals in Maryland are paid in accordance with the Maryland Total Cost of Care Model, and hospitals located outside the 50 States, the District of Columbia, and Puerto Rico (that is, 6 short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa) receive payment for inpatient hospital services they furnish on the basis of reasonable costs, subject to a rate-of-increase ceiling.
As of March 2024, there were 3,082 IPPS acute care hospitals included in our analysis. This represents approximately 53 percent of all Medicare-participating hospitals. The majority of this impact analysis focuses on this set of hospitals. There also are approximately 1,381 CAHs. These small, limited service hospitals are paid on the basis of reasonable costs, rather than under the IPPS. IPPS-excluded hospitals and units, which are paid under separate payment systems, include IPFs, IRFs, LTCHs, RNHCIs, children's hospitals, cancer hospitals, extended neoplastic disease care hospital, and short-term acute care hospitals located in the Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa. Changes in the prospective payment systems for IPFs and IRFs are made through separate rulemaking. Payment impacts of changes to the prospective payment systems for these IPPS-excluded hospitals and units are not included in this final rule. The impact of the update and policy changes to the LTCH PPS for FY 2025 is discussed in section I.J. of this Appendix.
In this final rule, we are announcing policy changes and payment rate updates for the IPPS for FY 2025 for operating costs of acute care hospitals. The FY 2025 updates to the capital payments to acute care hospitals are discussed in section I.I. of the Appendix in this final rule.
Based on the overall percentage change in payments per case estimated using our payment simulation model, we estimate that total FY 2025 operating payments would increase by 2.8 percent, compared to FY 2024. The impacts do not reflect changes in the number of hospital admissions or real case-mix intensity, which would also affect overall payment changes.
We have prepared separate impact analyses of the changes to each system. This section deals with the changes to the operating inpatient prospective payment system for acute care hospitals. Our payment simulation model relies on the best available claims data to enable us to estimate the impacts on payments per case of certain changes in this final rule. However, there are other changes for which we do not have data available that would allow us to estimate the payment impacts using this model. For those changes, we have attempted to predict the payment impacts based upon our experience and other more limited data.
The data used in developing the quantitative analyses of changes in payments per case presented in this section are taken from the FY 2023 MedPAR file and the most current Provider-Specific File (PSF) that is used for payment purposes. Although the analyses of the changes to the operating PPS do not incorporate cost data, data from the best available hospital cost reports were used to categorize hospitals. Our analysis has several qualifications. First, in this analysis, we do not adjust for future changes in such ( print page 69995) variables as admissions, lengths of stay, or underlying growth in real case-mix. Second, due to the interdependent nature of the IPPS payment components, it is very difficult to precisely quantify the impact associated with each change. Third, we use various data sources to categorize hospitals in the tables. In some cases, particularly the number of beds, there is a fair degree of variation in the data from the different sources. We have attempted to construct these variables with the best available source overall. However, for individual hospitals, some miscategorizations are possible.
Using cases from the FY 2023 MedPAR file, we simulate payments under the operating IPPS given various combinations of payment parameters. As described previously, Indian Health Service hospitals and hospitals in Maryland were excluded from the simulations. The impact of payments under the capital IPPS, and the impact of payments for costs other than inpatient operating costs, are not analyzed in this section. Estimated payment impacts of the capital IPPS for FY 2025 are discussed in section I.I. of this Appendix. We note, as discussed in section III. of the preamble of this final rule, we are finalizing our proposal to adopt the new OMB labor market area delineations as described in the July 21, 2023 OMB Bulletin No. 23-01, effective for the FY 2025 IPPS wage index. We also note, as discussed in section II.A.4. of the Addendum of this final rule, we used wage indexes based on the new OMB delineations in determining aggregate payments on each side of the comparison for the changes discussed below, except where otherwise noted (for example, the FY 2024 baseline simulation model). This is consistent with our discussion in section II.A.4. of the Appendix of this final rule, to use wage indexes based on the new OMB delineations in the determination of all of the budget neutrality factors in order to properly determine aggregate payments on each side of the comparison for our budget neutrality calculations. We further note that as discussed in that same section, consistent with past practice as finalized in the FY 2005 IPPS final rule ( 69 FR 49034 ), we are not adopting the new OMB delineations themselves in a budget neutral manner. We continue to believe that the revision to the labor market areas in and of itself does not constitute an “adjustment or update” to the adjustment for area wage differences, as provided under section 1886(d)(3)(E) of the Act.
We discuss the following changes:
- The effects of the application of the applicable percentage increase of 2.9 percent (that is, a 3.4 percent market basket update with a reduction of 0.5 percentage point for the productivity adjustment), and the applicable percentage increase (including the market basket update and the productivity adjustment) to the hospital-specific rates.
- The effects of the changes to the relative weights and MS-DRG GROUPER.
- The effects of the changes in hospitals' wage index values reflecting updated wage data from hospitals' cost reporting periods beginning during FY 2021, compared to the FY 2020 wage data, to calculate the FY 2025 wage index.
- The effects of the geographic reclassifications by the MGCRB (as of publication of this final rule) that would be effective for FY 2025.
- The effects of the rural floor with the application of the national budget neutrality factor to the wage index.
- The effects of the imputed floor wage index adjustment. This provision is not budget neutral.
- The effects of the frontier State wage index adjustment under the statutory provision that requires hospitals located in States that qualify as frontier States to not have a wage index less than 1.0. This provision is not budget neutral.
- The effects of the implementation of section 1886(d)(13) of the Act, which provides for an increase in a hospital's wage index if a threshold percentage of residents of the county where the hospital is located commute to work at hospitals in counties with higher wage indexes for FY 2025. This provision is not budget neutral.
- The effects of the expiration of the special payment status for MDHs beginning January 1, 2025 under current law. As discussed elsewhere in this final rule, section 307 of the Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, extended the MDH program for FY 2025 discharges occurring before January 1, 2025. Prior to enactment of the CAA, 2024, the MDH program was only to be in effect through the end of FY 2024. Therefore, under current law, the MDH program will expire for discharges on or after January 1, 2025. As a result, MDHs that currently receive the higher of payments made based on the Federal rate or the payments made based on the Federal rate plus 75 percent of the difference between payments based on the Federal rate and the hospital-specific rate will be paid based on the Federal rate starting January 1, 2025.
- The total estimated change in payments based on the FY 2025 policies relative to payments based on FY 2024 policies.
In accordance with section 1886(b)(3)(B)(i) of the Act, each year we update the national standardized amount for inpatient hospital operating costs by a factor called the “applicable percentage increase.” For FY 2025, depending on whether a hospital submits quality data under the rules established in accordance with section 1886(b)(3)(B)(viii) of the Act (hereafter referred to as a hospital that submits quality data) and is a meaningful EHR user under section 1886(b)(3)(B)(ix) of the Act (hereafter referred to as a hospital that is a meaningful EHR user), there are four possible applicable percentage increases that can be applied to the national standardized amount.

To illustrate the impact of the FY 2025 changes, our analysis begins with a FY 2024 baseline simulation model using: the FY 2024 applicable percentage increase of 2.6 percent; the FY 2024 MS-DRG GROUPER (Version 41); the FY 2024 CBSA designations for hospitals based on the OMB definitions from the 2010 Census; the FY 2024 wage index; and no MGCRB reclassifications. Outlier payments are set at 5.1 percent of total operating MS-DRG and outlier payments for modeling purposes.
We note the following at the time this impact analysis was prepared:
- 90 hospitals are estimated to not receive the full market basket rate-of-increase for FY 2025 because they failed the quality data submission process or did not choose to participate, but are meaningful EHR users. For purposes of the simulations shown later in this section, we modeled the payment changes for FY 2025 using a reduced update for these hospitals.
- 82 hospitals are estimated to not receive the full market basket rate-of-increase for FY 2025 because they are identified as not meaningful EHR users that do submit quality information under section 1886(b)(3)(B)(viii) of the Act. For purposes of the simulations shown in this section, we modeled the payment changes for FY 2025 using a reduced update for these hospitals.
- 27 hospitals are estimated to not receive the full market basket rate-of-increase for FY 2025 because they are identified as not meaningful EHR users that do not submit quality data under section 1886(b)(3)(B)(viii) of the Act.
Each policy change, statutory or otherwise, is then added incrementally to this baseline, finally arriving at an FY 2025 model incorporating all of the changes. This simulation allows us to isolate the effects of each change.
Our comparison illustrates the percent change in payments per case from FY 2024 to FY 2025. Two factors not discussed separately have significant impacts here. The first factor is the update to the standardized amount (see the table earlier in this section that shows the four applicable percentage increases that can be applied to the national standardized amount for FY 2025). We note, section 1886(b)(3)(B)(iv) of the Act provides that the applicable percentage increase applicable to the hospital-specific rates for SCHs and MDHs equals the applicable percentage increase set forth in section 1886(b)(3)(B)(i) of the Act (that is, the same update factor as for all other hospitals subject to the IPPS). Because the Act sets the update factor for SCHs and MDHs equal to the update factor for all other IPPS hospitals, the update to the hospital-specific rates for SCHs and MDHs is subject to the amendments to section 1886(b)(3)(B) of the Act made by sections 3401(a) and 10319(a) of the Affordable Care Act. Accordingly, the applicable percentage increases to the hospital-specific rates applicable to SCHs and MDHs for FY 2025 are the same as the four applicable percentage increases in the table earlier in this section.
A second significant factor that affects the changes in hospitals' payments per case from FY 2024 to FY 2025 is the change in hospitals' geographic reclassification status from one year to the next. That is, payments may be reduced for hospitals reclassified in FY 2024 that are no longer reclassified in FY 2025. Conversely, payments may increase for hospitals not reclassified in FY 2024 that are reclassified in FY 2025.
Table I displays the results of our analysis of the changes for FY 2025. The table categorizes hospitals by various geographic and special payment consideration groups to illustrate the varying impacts on different types of hospitals. The top row of the table shows the overall impact on the 3,082 acute care hospitals included in the analysis.
The next two rows of Table I contain hospitals categorized according to their geographic location: urban and rural. There are 2,392 hospitals located in urban areas and 690 hospitals in rural areas included in our analysis. The next two groupings are by bed-size categories, shown separately for urban and rural hospitals. The last groupings by geographic location are by census divisions, also shown separately for urban and rural hospitals.
The second part of Table I shows hospital groups based on hospitals' FY 2025 payment classifications, including any reclassifications under section 1886(d)(10) of the Act. For example, the rows labeled urban and rural show that the numbers of hospitals paid based on these categorizations after consideration of geographic reclassifications (including reclassifications under sections 1886(d)(8)(B) and 1886(d)(8)(E) of the Act) are 1,714, and 1,368, respectively.
The next three groupings examine the impacts of the changes on hospitals grouped by whether or not they have GME residency ( print page 69997) programs (teaching hospitals that receive an IME adjustment) or receive Medicare DSH payments, or some combination of these two adjustments. There are 1,832 nonteaching hospitals in our analysis, 958 teaching hospitals with fewer than 100 residents, and 292 teaching hospitals with 100 or more residents.
In the DSH categories, hospitals are grouped according to their DSH payment status, and whether they are considered urban or rural for DSH purposes. The next category groups together hospitals considered urban or rural, in terms of whether they receive the IME adjustment, the DSH adjustment, both, or neither.
The next six rows examine the impacts of the changes on rural hospitals by special payment groups (SCHs and RRCs) and reclassification status from urban to rural in accordance with section 1886(d)(8)(E) of the Act. Of the hospitals that are not reclassified from urban to rural, there are 155 RRCs, 244 SCHs, and 119 hospitals that are both SCHs and RRCs. Of the hospitals that are reclassified from urban to rural, there are 579 RRCs, 34 SCHs, and 46 hospitals that are both SCHs and RRCs.
The next series of groupings are based on the type of ownership and the hospital's Medicare and Medicaid utilization expressed as a percent of total inpatient days. These data were taken from the most recent available Medicare cost reports.
The next grouping concerns the geographic reclassification status of hospitals. The first subgrouping is based on whether a hospital is reclassified or not. The second and third subgroupings are based on whether urban and rural hospitals were reclassified by the MGCRB for FY 2025 or not, respectively. The fourth subgrouping displays hospitals that reclassified from urban to rural in accordance with section 1886(d)(8)(E) of the Act. The fifth subgrouping displays hospitals deemed urban in accordance with section 1886(d)(8)(B) of the Act.

As discussed in section V.B. of the preamble of this final rule, this column includes the hospital update, including the 3.4 percent market basket rate-of-increase reduced by the 0.5 percentage point for the productivity adjustment. As a result, we are making a 2.9 percent update to the national standardized amount. This column also includes the update to the hospital-specific rates which includes the 3.4 percent market basket rate-of-increase reduced by 0.5 percentage point for the productivity adjustment. As a result, we are making a 2.9 percent update to the hospital-specific rates.
Overall, hospitals would experience a 2.9 percent increase in payments primarily due to the combined effects of the hospital update to the national standardized amount and the hospital update to the hospital-specific rate.
Column 2 shows the effects of the changes to the MS-DRGs and relative weights with the application of the recalibration budget neutrality factor to the standardized amounts. Section 1886(d)(4)(C)(i) of the Act requires us annually to make appropriate classification changes to reflect changes in treatment patterns, technology, and any other factors that may change the relative use of hospital resources. Consistent with section 1886(d)(4)(C)(iii) of the Act, we calculated a recalibration budget neutrality factor to account for the changes in MS-DRGs and relative weights to ensure that the overall payment impact is budget neutral. We also applied the permanent 10-percent cap on the reduction in a MS-DRG's relative weight in a given year and an associated recalibration cap budget neutrality factor to account for the 10-percent cap on relative weight reductions to ensure that the overall payment impact is budget neutral.
As discussed in section II.D. of the preamble of this final rule, for FY 2025, we calculated the MS-DRG relative weights using the FY 2023 MedPAR data grouped to the Version 42 (FY 2025) MS-DRGs. The reclassification changes to the GROUPER are described in more detail in section II.C. of the preamble of this final rule.
The “All Hospitals” line in Column 2 indicates that changes due to the MS-DRGs and relative weights would result in a 0.0 percent change in payments with the application of the recalibration budget neutrality factor of 0.99719 and the recalibration cap budget neutrality factor of 0.999874 to the standardized amount.
Column 3 shows the impact of the updated wage data, with the application of the wage budget neutrality factor. The wage index is calculated and assigned to hospitals on the basis of the labor market area in which the hospital is located. Under section 1886(d)(3)(E) of the Act, beginning with FY 2005, we delineate hospital labor market areas based on the Core Based Statistical Areas (CBSAs) established by OMB. The current statistical standards (based on OMB standards) used in FY 2025 are discussed in section III.A.2. of the preamble of this final rule. Specifically, we are implementing the new OMB delineations as described in the July 21, 2023 OMB Bulletin No. 23-01, effective beginning with the FY 2025 IPPS wage index.
Section 1886(d)(3)(E) of the Act requires that, beginning October 1, 1993, we annually update the wage data used to calculate the wage index. In accordance with this requirement, the wage index for acute care hospitals for FY 2025 is based on data submitted for hospital cost reporting periods, beginning on or after October 1, 2020 and before October 1, 2021. The estimated impact of the updated wage data on hospital payments is isolated in Column 3 by holding the other payment parameters constant in this simulation. That is, Column 3 shows the percentage change in payments when going from a model using the FY 2024 wage index, the labor-related share of 67.6 percent, and having a 100-percent occupational mix adjustment applied, to a model using the FY 2025 pre-reclassification wage index with the labor-related share of 67.6 percent, also having a 100-percent occupational mix adjustment applied, while holding other payment parameters, such as use of the Version 42 MS-DRG GROUPER constant. As noted earlier and as discussed in section II.A.4. of the Addendum of this final rule, we used wage indexes based on the new OMB delineations in determining aggregate payments on each side of the comparison/model. The FY 2025 occupational mix adjustment is based on the CY 2022 occupational mix survey.
In addition, the column shows the impact of the application of the wage budget neutrality to the national standardized amount. In FY 2010, we began calculating separate wage budget neutrality and recalibration budget neutrality factors, in accordance with section 1886(d)(3)(E) of the Act, which specifies that budget neutrality to account for wage index changes or updates made under that subparagraph must be made without regard to the 62 percent labor-related share guaranteed under section 1886(d)(3)(E)(ii) of the Act. Therefore, for FY 2025, we are calculating the wage budget neutrality factor to ensure that payments under the updated wage data and the labor-related share of 67.6 percent are budget neutral, without regard to the lower labor-related share of 62 percent applied to hospitals with a wage index less than or equal to 1.0. In other words, the wage budget neutrality factor is calculated under the assumption that all hospitals receive the higher labor-related share of the standardized amount. The FY 2025 wage budget neutrality factor is 1.000114 and the overall payment change is 0 percent.
Column 3 shows the impacts of updating the wage data. Overall, the new wage data and the labor-related share, combined with the wage budget neutrality adjustment, would lead to no change for all hospitals, as shown in Column 3.
In looking at the wage data itself, the national average hourly wage would increase 9.20 percent compared to FY 2024. Therefore, the only manner in which to ( print page 70001) maintain or exceed the previous year's wage index was to match or exceed the 9.20 percent increase in the national average hourly wage.
The following chart compares the shifts in wage index values for hospitals due to changes in the average hourly wage data for FY 2025 relative to FY 2024. These figures reflect changes in the “pre-reclassified, occupational mix-adjusted wage index,” that is, the wage index before the application of geographic reclassification, the rural floor, the out-migration adjustment, and other wage index exceptions and adjustments. We note that the “post-reclassified wage index” or “payment wage index,” which is the wage index that includes all such exceptions and adjustments (as reflected in Tables 2 and 3 associated with this final rule) is used to adjust the labor-related share of a hospital's standardized amount, either 67.6 percent or 62 percent, depending upon whether a hospital's wage index is greater than 1.0 or less than or equal to 1.0. Therefore, the pre-reclassified wage index figures in the following chart may illustrate a somewhat larger or smaller change than would occur in a hospital's payment wage index and total payment.
The following chart shows the projected impact of changes in the area wage index values for urban and rural hospitals based on the wage data used for this final rule.

Our impact analysis to this point has assumed acute care hospitals are paid on the basis of their actual geographic location (with the exception of ongoing policies that provide that certain hospitals receive payments on bases other than where they are geographically located, such as hospitals with a § 412.103 reclassification or “LUGAR” status). The changes in Column 4 reflect the per case payment impact of moving from this baseline to a simulation incorporating the MGCRB decisions for FY 2025.
By spring of each year, the MGCRB makes reclassification determinations that would be effective for the next fiscal year, which begins on October 1. The MGCRB may approve a hospital's reclassification request for the purpose of using another area's wage index value. Hospitals may appeal denials by the MGCRB of reclassification requests to the CMS Administrator. Further, hospitals have 45 days from the date the IPPS final rule is issued in the Federal Register to decide whether to withdraw or terminate an approved geographic reclassification for the following year.
The overall effect of geographic reclassification is required by section 1886(d)(8)(D) of the Act to be budget neutral. Therefore, for purposes of this impact analysis, we are applying an adjustment of 0.962791 to ensure that the effects of the reclassifications under sections 1886(d)(8)(B) and (C) and 1886(d)(10) of the Act are budget neutral (section II.A. of the Addendum to this final rule).
Geographic reclassification generally benefits hospitals in rural areas. We estimate that the geographic reclassification would increase payments to rural hospitals by an average of 2.4 percent. By region, most rural hospital categories would experience increases in payments due to MGCRB reclassifications.
Table 2 listed in section VI. of the Addendum to this final rule and available via the internet on the CMS website reflects the reclassifications for FY 2025.
As discussed in section III.G.1. of the preamble of this final rule, section 4410 of Public Law 105-33 established the rural floor by requiring that the wage index for a hospital in any urban area cannot be less than the wage index applicable to hospitals located in rural areas in the same state. We apply a uniform budget neutrality adjustment to the wage index. Column 5 shows the effects of the rural floor.
The Affordable Care Act requires that we apply one rural floor budget neutrality factor to the wage index nationally. We have calculated a FY 2025 rural floor budget neutrality factor to be applied to the wage index of 0.977499, which would reduce wage indexes by 2.3 percent compared to the rural floor provision not being in effect.
Column 5 shows the projected impact of the rural floor with the national rural floor budget neutrality factor applied to the wage index. The column compares the post-reclassification FY 2025 wage index of providers before the rural floor adjustment to the post-reclassification FY 2025 wage index of providers with the rural floor adjustment.
We estimate that 771 hospitals would receive the rural floor in FY 2025. All IPPS hospitals in our model would have their wage indexes reduced by the rural floor budget neutrality adjustment of 0.977499. We project that, in aggregate, rural hospitals would experience a 0.7 percent decrease in payments as a result of the application of the rural floor budget neutrality adjustment because the rural hospitals do not benefit from the rural floor, but have their wage indexes downwardly adjusted to ensure that the application of the rural floor is budget neutral overall. We project that, in the aggregate, hospitals located in urban areas would experience a 0.1 percent increase in payments, because increases in payments to hospitals benefitting from the rural floor offset decreases in payments to non-rural floor urban hospitals whose wage index is downwardly adjusted by the rural floor budget neutrality factor. Urban hospitals in the Pacific region would experience a 2.3 percent increase in payments primarily due to the application of the rural floor in California.
This column shows the combined effects of the application of the following: (1) the imputed floor under section 1886(d)(3)(E)(iv)(I) and (II) of the Act, which provides that for discharges occurring on or after October 1, 2021, the area wage index applicable to any hospital in an all-urban State may not be less than the minimum area wage index for the fiscal year for hospitals in that State established using the methodology described in § 412.64(h)(4)(vi) as in effect for FY 2018; (2) section 10324(a) of the Affordable Care Act, which requires that we establish a minimum post-reclassified wage index of 1.00 for all hospitals located in “frontier States;” and (3) the effects of section 1886(d)(13) of the Act, which provides for an increase in the wage index for hospitals located in certain counties that have a relatively high percentage of hospital employees who reside in the county, but work in a different area with a higher wage index.
These three wage index provisions are not budget neutral and would increase payments overall by 0.3 percent compared to the provisions not being in effect.
Section 1886(d)(3)(E)(iv)(III) of the Act provides that the imputed floor wage index for all-urban States shall not be applied in a budget neutral manner. Therefore, the imputed floor adjustment is estimated to increase IPPS operating payments by approximately $203 million. There are an estimated 76 providers in Washington DC, New Jersey, Puerto Rico, and Rhode Island ( print page 70002) that would receive the imputed floor wage index.
The term “frontier States” is defined in the statute as States in which at least 50 percent of counties have a population density less than 6 persons per square mile. Based on these criteria, 5 States (Montana, Nevada, North Dakota, South Dakota, and Wyoming) are considered frontier States, and an estimated 41 hospitals located in Montana, North Dakota, South Dakota, and Wyoming would receive a frontier wage index of 1.0000. We note, the rural floor for Nevada exceeds the frontier state wage index of 1.000, and therefore no hospitals in Nevada receive the frontier state wage index. Overall, this provision is not budget neutral and is estimated to increase IPPS operating payments by approximately $55 million.
In addition, section 1886(d)(13) of the Act provides for an increase in the wage index for hospitals located in certain counties that have a relatively high percentage of hospital employees who reside in the county but work in a different area with a higher wage index. Hospitals located in counties that qualify for the payment adjustment would receive an increase in the wage index that is equal to a weighted average of the difference between the wage index of the resident county, post-reclassification and the higher wage index work area(s), weighted by the overall percentage of workers who are employed in an area with a higher wage index. There are an estimated 203 providers that would receive the out-migration wage adjustment in FY 2025. This out-migration wage adjustment is not budget neutral, and we estimate the impact of these providers receiving the out-migration increase would be approximately $65 million.
Column 7 shows our estimate of the changes in payments due to the expiration of MDH status, a nonbudget neutral payment provision. Section 102 of the Continuing Appropriations and Ukraine Supplemental Appropriations Act, 2023 ( Pub. L. 117-180 ), extended the MDH program (which, under previous law, was to be in effect for discharges before October 1, 2022 only) through December 16, 2022. Section 102 of the Further Continuing Appropriations and Extensions Act, 2023 ( Pub. L. 117-229 ) extended the MDH program through December 23, 2022. Section 4102 of the Consolidated Appropriations Act, 2023 ( Pub. L. 117-328 ), extended the MDH program through FY 2024 (that is for discharges occurring before October 1, 2024). As previously noted, section 307 of the CAA, 2024 ( Pub. L. 118-42 ), enacted on March 9, 2024, further extended the MDH program for FY 2025 discharges occurring before January 1, 2025. Prior to enactment of the CAA, 2024, the MDH program was only to be in effect through the end of FY 2024. Therefore, under current law, the MDH program will expire for discharges on or after January 1, 2025. Hospitals that qualify to be MDHs receive the higher of payments made based on the Federal rate or the payments made based on the Federal rate amount plus 75 percent of the difference between payments based on the Federal rate and payments based on the hospital-specific rate (a hospital-specific cost-based rate). Because this provision is not budget neutral, the expiration of this payment provision is estimated to result in a 0.1 percent decrease in payments overall. There are currently 173 MDHs, of which we estimate 117 would be paid under the blended payment of the Federal rate and hospital-specific rate if the MDH program were not set to expire. Because those 117 MDHs will no longer receive the blended payment and will be paid only under the Federal rate for FY 2025 discharges beginning on or after January 1, 2025, it is estimated that those hospitals would experience an overall decrease in payments of approximately $152 million. The $152 million overall decrease reflects the 3-month extension of the MDH program through December 31, 2024 under section 307 of the CAA, 2024.
Column 8 shows our estimate of the changes in payments per discharge from FY 2024 and FY 2025, resulting from all changes reflected in this final rule for FY 2025. It includes combined effects of the year-to-year change of the factors described in previous columns in the table.
The average increase in payments under the IPPS for all hospitals is approximately 2.8 percent for FY 2025 relative to FY 2024 and for this row is primarily driven by the changes reflected in Column 1. Column 8 includes the annual hospital update of 2.9 percent to the national standardized amount. This annual hospital update includes the 3.4 percent market basket rate-of-increase reduced by the 0.5 percentage point productivity adjustment. Hospitals paid under the hospital-specific rate would receive a 2.9 percent hospital update. As described in Column 1, the annual hospital update for hospitals paid under the national standardized amount, combined with the annual hospital update for hospitals paid under the hospital-specific rates, combined with the other adjustments described previously and shown in Table I, would result in a 2.48 percent increase in payments in FY 2025 relative to FY 2024.
This column also reflects the estimated effect of outlier payments returning to their targeted levels in FY 2025 as compared to the estimated outlier payments for FY 2024 produced from our payment simulation model. As discussed in section II.A.4.i. of the Addendum to this final rule, the statute requires that outlier payments for any year are projected to be not less than 5 percent nor more than 6 percent of total operating DRG payments plus outlier payments, and also requires that the average standardized amount be reduced by a factor to account for the estimated proportion of total DRG payments made to outlier cases. We continue to use a 5.1 percent target (or an outlier offset factor of 0.949) in calculating the outlier offset to the standardized amount, just as we did for FY 2024. Therefore, our estimate of payments per discharge for FY 2025 from our payment simulation model reflects this 5.1 percent outlier payment target. Our payment simulation model shows that estimated outlier payments for FY 2024 were less than that target by approximately 0.05 percent. Therefore, our estimate of the changes in payments per discharge from FY 2024 to FY 2025 in Column 8 reflects the estimated 0.05 percent change in outlier payments produced by our payment simulation model when returning to the 5.1 percent outlier target for FY 2025. There are also interactive effects among the various factors comprising the payment system that we are not able to isolate, which may contribute to our estimate of the changes in payments per discharge from FY 2024 and FY 2025 in Column 8.
Overall payments to hospitals paid under the IPPS due to the applicable percentage increase and changes to policies related to MS-DRGs, geographic adjustments, and outliers are estimated to increase by 2.8 percent for FY 2025. Hospitals in urban areas would experience a 2.8 percent increase in payments per discharge in FY 2025 compared to FY 2024. Hospital payments per discharge in rural areas are estimated to increase by 2.6 percent in FY 2025.
Table II presents the projected impact of the changes for FY 2025 for urban and rural hospitals and for the different categories of hospitals shown in Table I. It compares the estimated average payments per discharge for FY 2024 with the estimated average payments per discharge for FY 2025, as calculated under our models. Therefore, this table presents, in terms of the average dollar amounts paid per discharge, the combined effects of the changes presented in Table I. The estimated percentage changes shown in the last column of Table II equal the estimated percentage changes in average payments per discharge from Column 8 of Table I.

Advancing health equity is the first pillar of CMS's 2022 Strategic Framework. [ 1109 ] To gain insight into how the IPPS policies could affect health equity, we have added Table III, Provider Deciles by Beneficiary Characteristics, for informational purposes. Table III details providers in terms of the beneficiaries they serve, and shows differences in estimated average payments per case and changes in estimated average payments per case relative to other providers.
As noted in section I.C. of this Appendix, this final rule contains a range of policies, and there is a section of the final rule where each policy is discussed. Each section includes the rationale for our decisions, including the need for the final policy. The information contained in Table III is provided solely to demonstrate the quantitative effects of our policies across a number of health equity dimensions and does not form the basis or rationale for the policies.
Patient populations that have been disadvantaged or underserved by the healthcare system may include patients with the following characteristics, among others: members of racial and ethnic minorities; members of federally recognized Tribes, people with disabilities; members of the lesbian, gay, bisexual, transgender, and queer (LGBTQ+) community; individuals with limited English proficiency, members of rural communities, and persons otherwise adversely affected by persistent poverty or inequality. The CMS Framework for Health Equity was developed with particular attention to disparities in chronic and infectious diseases; as an example of a chronic disease associated with significant disparities, we therefore also detail providers in terms of the percentage of their claims for beneficiaries receiving ESRD Medicare coverage.
Because we do not have data for all characteristics that may identify disadvantaged or underserved patient populations, we use several proxies to capture these characteristics, based on claims data from the FY 2023 MedPAR file and Medicare enrollment data from Medicare's Enrollment Database (EDB), including: race/ethnicity, dual eligibility for Medicaid and Medicare, Medicare low income subsidy (LIS) enrollment, a joint indicator for dual or LIS enrollment, presence of an ICD-10-CM Z code indicating a “social determinant of health” (SDOH), presence of a behavioral health diagnosis code, receiving ESRD Medicare coverage, qualifying for Medicare due to disability, living in a rural area, and living in an area with an area deprivation index (ADI) greater than or equal to 85. We refer to each of these proxies as characteristics in Table III and the discussion that follows.
The first health equity-relevant grouping presented in Table III is race/ethnicity. To assign the race/ethnicity variables used in Table III, we utilized the Medicare Bayesian Improved Surname Geocoding (MBISG) data in conjunction with the MedPAR data. The method used to develop the MBISG data involves estimating a set of six racial and ethnic probabilities (White, Black, Hispanic, American Indian or Alaskan Native, Asian or Pacific Islander, and multiracial) from the surname and address of beneficiaries by using previous self-reported data from a national survey of Medicare beneficiaries, post-stratified to CMS enrollment files. The MBISG method is used by the CMS Office of Minority Health in its reports analyzing Medicare Advantage plan performance on Healthcare Effectiveness Data and Information Set (HEDIS) measures, and is being considered by CMS for use in other CMS programs. To estimate the percentage of discharges for each specified racial/ethnic category for each hospital, the sum of the probabilities for that category for that hospital was divided by the hospital's total number of discharges.
The two main proxies for income available in the Medicare claims and enrollment data are dual eligibility for Medicare and Medicaid and Medicare LIS status. Dual-enrollment status is a powerful predictor of poor outcomes on some quality and resource use measures even after accounting for additional social and functional risk factors. [ 1110 ] Medicare LIS enrollment refers to a beneficiary's enrollment in the low-income subsidy program for the Part D prescription drug benefit. This program covers all or part of the Part D premium for qualifying Medicare beneficiaries and gives them access to reduced copays for Part D drugs. (We note that beginning on January 1, 2024, eligibility for the full low-income subsidy was expanded to include individuals currently eligible for the partial low-income subsidy.) Because Medicaid eligibility rules and benefits vary by state/territory, Medicare LIS enrollment identifies beneficiaries who are likely to have low income but may not be eligible for Medicaid. Not all beneficiaries who qualify for the duals or LIS programs actually enroll. Due to differences in the dual eligibility and LIS qualification criteria and less than complete participation in these programs, sometimes beneficiaries were flagged as dual but not LIS or vice versa. Hence this analysis also used a “dual or LIS” flag as a third proxy for low income. The dual and LIS flags were constructed based on enrollment/eligibility status in the EDB during the month of the hospital discharge.
Social determinants of health (SDOH) are the conditions in the environments where people are born, live, learn, work, play, worship, and age that affect a wide range of health, functioning, and quality-of-life outcomes and risks. [ 1111 ] These circumstances or determinants influence an individual's health status and can contribute to wide health disparities and inequities. ICD-10-CM contains Z-codes that describe a range of issues related—but not limited—to education and literacy, employment, housing, ability to obtain adequate amounts of food or safe drinking water, and occupational exposure to toxic agents, dust, or radiation. The presence of ICD-10-CM Z-codes in the range Z55-Z65 identifies beneficiaries with these SDOH characteristics. The SDOH flag used for this analysis was turned on if one of these Z-codes was recorded on the claim for the hospital stay itself (that is, the beneficiary's prior claims were not examined for additional Z-codes). Since these codes are not required for Medicare FFS patients and did not impact payment under the IPPS in FY 2023, we believe they may be underreported in the claims data from the FY 2023 MedPAR file used for this analysis and not reflect the actual rates of SDOH. In 2019, 0.11 percent of all Medicare FFS claims were Z code claims and 1.59 percent of continuously enrolled Medicare FFS beneficiaries had claims with Z codes. [ 1112 ] However, we expect the reporting of Z codes on claims may increase over time, because of newer quality measures in the Hospital Inpatient Quality Reporting (IQR) Program that capture screening and identification of patient-level, health-related social needs (MUC21-134 and MUC21-136) ( 87 FR 49201 through 49220 ). In the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58755 through 58759 ), we also finalized a change to the severity designation of the following three ICD-10-CM diagnosis codes from non-CC to CC: Z59.00 (Homelessness, unspecified), Z59.01 (Sheltered homelessness) and Z59.02 (Unsheltered homelessness). We also refer the reader to section II.C.12.c.1. of the preamble of this final rule, where we discuss our final policy to change the severity level designation of the following seven ICD-10-CM diagnosis codes from non-CC to CC for FY 2025: Z59.10 (Inadequate housing, unspecified), Z59.11 (Inadequate housing environmental temperature), Z59.12 (Inadequate housing utilities), Z59.19 (Other inadequate housing), Z59.811 (Housing instability, housed, with risk of homelessness), Z59.812 (Housing instability, housed, homelessness in past 12 months), and Z59.819 (Housing instability, housed unspecified).
Beneficiaries with behavioral health diagnoses often face co-occurring physical illnesses, but often experience difficulty accessing care. [ 1113 ] The combination of physical and behavioral health conditions can exacerbate both conditions and result in poorer outcomes than one condition alone. [ 1114 ] Additionally, the intersection of behavioral health and health inequities is a core aspect of CMS' Behavioral Health Strategy. [ 1115 ] We used the presence of one or more ICD-10-CM codes in the range of F01-F99 to identify beneficiaries with a behavioral health diagnosis.
Beneficiaries with disabilities are categorized as being disabled because of a medically determinable physical or mental impairment(s) that has lasted or is expected to last for a continuous period of at least 12 months or is expected to result in death. [ 1116 ] Beneficiaries with disabilities often have complex healthcare needs and difficulty accessing care. Beneficiaries with disabilities were classified as such persons for the purposes of this analysis if their original reason for qualifying for Medicare was disability; this information was obtained from Medicare's EDB. We note that this is likely an underestimation of disability because it does not account for beneficiaries who became disabled after becoming entitled to Medicare. This metric also does not capture all individuals who would be considered to have a disability under 29 U.S.C. 705(9)(B) .
Beneficiaries with ESRD have high healthcare needs and high medical spending, and often experience comorbid conditions and poor mental health. Beneficiaries with ESRD also experience significant disparities, such as a limited life expectancy. [ 1117 ] Beneficiaries were classified as ESRD for the purposes of this analysis if they were receiving Medicare ESRD coverage during the month of the discharge; this information was obtained from Medicare's EDB.
Beneficiaries in some geographic areas—particularly rural areas or areas with concentrated poverty—often have difficulty accessing care. [ 1118 1119 ] For this impact analysis, beneficiaries were classified on two dimensions: from a rural area and from an area with an area deprivation index (ADI) greater than or equal to 85.
Rural status is defined for purposes of this analysis using the primary Rural-Urban Commuting Area (RUCA) codes 4-10 (including micropolitan, small town, and rural areas) corresponding to each beneficiary's zip code. RUCA codes are defined at the census tract level based on measures of population density, urbanization, and daily commuting. The ADI is obtained from a publicly available dataset designed to capture socioeconomic disadvantage at the neighborhood level. [ 1120 ] It utilizes data on income, education, employment, housing quality, and 13 other factors from the American Community Survey and combines them into a single raw score, which is then used to rank neighborhoods (defined at various levels), with higher scores reflecting greater deprivation. The version of the ADI used for this analysis is at the Census Block Group level and the ADI corresponds to the Census ( print page 70006) Block Group's percentile nationally. Living in an area with an ADI score of 85 or above, a validated measure of neighborhood disadvantage, is shown to be a predictor of 30-day readmission rates, lower rates of cancer survival, poor end of life care for patients with heart failure, and longer lengths of stay and fewer home discharges post-knee surgery even after accounting for individual social and economic risk factors. [ 1121 1122 1123 1124 1125 ] The MedPAR discharge data was linked to the RUCA using beneficiaries' five-digit zip code and to the ADI data using beneficiaries' 9-digit zip codes, both of which were derived from Common Medicare Enrollment (CME) files. Beneficiaries with no recorded zip code were treated as being from an urban area and as having an ADI less than 85.
For each of these characteristics, the hospitals were classified into groups as follows. First, all discharges at IPPS hospitals (excluding Maryland and IHS hospitals) in the FY 2023 MedPAR file were flagged for the presence of the characteristic, with the exception of race/ethnicity, for which probabilities were assigned instead of binary flags, as described further in this section. Second, the percentage of discharges at each hospital for the characteristic was calculated. Finally, the hospitals were divided into four groups based on the percentage of discharges for each characteristic: decile group 1 contains the 10% of hospitals with the lowest rate of discharges for that characteristic; decile group 2 to 5 contains the hospitals with less than or equal to the median rate of discharges for that characteristic, excluding those in decile group 1; decile group 6 to 9 contains the hospitals with greater than the median rate of discharges for that characteristic, excluding those in decile group 10; and decile group 10 contains the 10% of hospitals with the highest rate of discharges for that characteristic. These decile groups provide an overview of the ways in which the average estimated payments per discharge vary between the providers with the lowest and highest percentages of discharges for each characteristic, as well as those above and below the median.
We note that a supplementary provider-level dataset containing the percentage of discharges at each hospital for each of the characteristics in Table III is available on our website.
- Column 1 of Table III specifies the beneficiary characteristic.
- Column 2 specifies the decile group.
- Column 3 specifies the percentiles covered by the decile group.
- Column 4 specifies the percentage range of discharges for each decile group specified in the first column.
- Columns 5 and 6 present the average estimated payments per discharge for FY 2024 and average estimated payments per discharge for FY 2025, respectively.
- Column 7 shows the percentage difference between these averages.
The average payment per discharge, as well as the percentage difference between the average payment per discharge in FY 2024 and FY 2025, can be compared across decile groups. For example, providers with the lowest decile of discharges for Dual (All) or LIS Enrolled beneficiaries have an average FY 2024 payment per discharge of $13,660.95, while providers with the highest decile of discharges for Dual (All) or LIS Enrolled beneficiaries have an average FY 2024 payment per discharge of $21,150.86. This pattern is also seen in the average FY 2025 payment per discharge.

As discussed in section II.E.4. of the preamble of this final rule, we are continuing to make new technology add-on payments in FY 2025 for the 24 technologies that would still be considered “new” for purposes of new technology add-on payments for FY 2025. Under § 412.88(a)(2), the new technology add-on payment for each case would be limited to the lesser of: (1) 65 percent of the costs of the new technology (or 75 percent of the costs for technologies designated as Qualified Infectious Disease Products (QIDPs) or approved under the Limited Population Pathway for Antibacterial and Antifungal Drugs (LPAD) pathway); or (2) 65 percent of the amount by which the costs of the case exceed the standard MS-DRG payment for the case (or 75 percent of the amount for technologies designated as QIDPs or approved under the LPAD pathway). Because it is difficult to predict the actual new technology add-on payment for each case, the estimated total payments in this final rule are based on the applicant's estimated cost and volume projections at the time they submitted their application (or based on updated figures provided during the public comment period) and the increase in new technology add-on payments for FY 2025 as if every claim that would qualify for a new technology add-on payment would receive the maximum add-on payment.
In the following table, we present estimated payment for the 24 technologies for which we are continuing to make new technology add-on payments in FY 2025:

In sections II.E.5. and 6. of the preamble to this final rule are 21 discussions of technologies for which we received applications for add-on payments for new medical services and technologies for FY 2025 (including Casgevy TM (exagamglogene autotemcel) for which the applicant submitted a single application for two separate indications, each of which is discussed separately; and ELREXFIO TM (elranatamab-bcmm) and TALVEY TM (talquetamab-tgvs), which are substantially ( print page 70010) similar to each other and evaluated as one application for new technology add-on payments under the IPPS). We note that of the 39 applications (23 alternative and 16 traditional) we received, 8 applications were not eligible for consideration for new technology add-on payment (7 alternative and 1 traditional), and 10 applicants withdrew their application (5 alternative and 5 traditional) prior to the issuance of this final rule (including the withdrawal of the application for DefenCath® (taurolidine/heparin), which received conditional approval for new technology add-on payments for FY 2024, subsequently was eligible to receive new technology add-on payments beginning with discharges on or after January 1, 2024, and for which we proposed and are finalizing to continue making new technology add-on payments for FY 2025). In the 21 discussions of technologies in the preamble of this final rule, there are a total of 15 new approvals for 14 technologies (3 traditional and 11 alternative) for new technology add-on payments for FY 2025. As explained in the preamble to this final rule, add-on payments for new medical services and technologies under section 1886(d)(5)(K) of the Act are not required to be budget neutral.
As discussed in section II.E.6. of the preamble of this final rule, under the alternative pathway for new technology add-on payments, new technologies that are medical products with a QIDP designation, approved through the FDA LPAD pathway, or are designated under the Breakthrough Device program will be considered not substantially similar to an existing technology for purposes of the new technology add-on payment under the IPPS, and will not need to demonstrate that the technology represents a substantial clinical improvement. These technologies must still be within the 2- to 3-year newness period, as discussed in section II.E.1.a.(1). of the preamble this final rule, and must also still meet the cost criterion.
As fully discussed in section II.E.6. of the preamble of this final rule, we are approving 12 new technology add-on payments for 11 technologies that applied under the alternative pathway for new technology add-on payments for FY 2025 (including ZEVTERA TM (ceftobiprole medocaril) for which the applicant submitted a single application for multiple indications, and for which we are approving two separate new technology add-on payments). The approvals include 10 technologies that received a Breakthrough Device designation from FDA and 1 that was designated as a QIDP by FDA. We did not receive any LPAD applications for add-on payments for new technologies for FY 2025.
Based on information from the applicants at the time of this final rule, we estimate that total payments for the technologies approved under the alternative pathway will be approximately $171.5 million for FY 2025. Total estimated FY 2025 payments for new technologies that are designated as a QIDP are approximately $5.6 million, and the total estimated FY 2025 payments for new technologies that are part of the Breakthrough Device program are approximately $165.9 million.
In the following table, we present detailed estimates for the 11 technologies for which we are approving 12 new technology add-on payments under the alternative pathway in FY 2025:

As fully discussed in section II.E.6. of the preamble of this final rule, we are approving new technology add-on payments for 3 technologies that applied under the traditional pathway for new technology add-on payments for FY 2025. We are also providing new technology add-on payments for 2 technologies that were evaluated as one application due to substantial similarity, and which are also considered substantially similar to a technology that was approved for new technology add-on payments for FY 2024 and is still considered “new” for purposes of new technology add-on payments for FY 2025. Based on information from the applicants at the time of rulemaking, we estimate that total payments for the technologies for which we are making new technology add-on payments is approximately $335.6 million for FY 2025. ( print page 70011)
In the following table, we present detailed estimates for the 6 technologies for which we are providing 5 new technology add-on payments under the traditional pathway in FY 2025:

In the following table, we present summary estimates for all technologies approved for new technology add-on payments for FY 2025:

As discussed in section IV.E. of the preamble of this final rule, under section 3133 of the Affordable Care Act, hospitals that are eligible to receive Medicare DSH payments will receive 25 percent of the amount they previously would have received under the statutory formula for Medicare DSH payments under section 1886(d)(5)(F) of the Act. The remainder, equal to an estimate of 75 percent of what formerly would have been paid as Medicare DSH payments (Factor 1), reduced to reflect changes in the percentage of uninsured individuals (Factor 2), is available to make additional payments to each hospital that qualifies for Medicare DSH payments and that has reported uncompensated care. Each hospital that is eligible for Medicare DSH payments will receive an additional payment based on its estimated share of the total amount of uncompensated care for all hospitals eligible for Medicare DSH payments. The uncompensated care payment methodology has redistributive effects based on the proportion of a hospital's amount of uncompensated care relative to the aggregate amount of uncompensated care of all hospitals eligible for Medicare DSH payments (Factor 3). The change to Medicare DSH payments under section 3133 of the Affordable Care Act is not budget neutral.
In this final rule, we are establishing the amount to be distributed as uncompensated care payments (UCP) to DSH-eligible hospitals for FY 2025, which is $5,705,743,275.00. This figure represents 75 percent of the amount that otherwise would have been paid for Medicare DSH payment adjustments adjusted by a Factor 2 of 54.29 percent. For FY 2024, the amount available to be distributed for uncompensated care was $5,938,006,756.87 or 75 percent of the amount that otherwise would have been paid for Medicare DSH payment adjustments adjusted by a Factor 2 of 59.29 percent. In addition, eligible IHS/Tribal hospitals and hospitals located in Puerto Rico are estimated to receive approximately $79,884,597 million in supplemental payments in FY 2025, as determined based on the difference between each hospital's FY 2022 UCP (decreased by 20.67 percent, which is the projected change between the FY 2025 total uncompensated care payment amount and the total uncompensated care payment amount for FY 2022) and its FY 2025 UCP as calculated using the methodology for FY 2025. If this difference is less than or equal to zero, the hospital will not receive a supplemental payment. For this final rule, the total UCP and supplemental payments equal approximately $5.786 billion. For FY 2025, we are using 3 years of data on ( print page 70012) uncompensated care costs from Worksheet S-10 of the FYs 2019, 2020, and 2021 cost reports to calculate Factor 3 for all DSH-eligible hospitals, including IHS/Tribal hospitals and Puerto Rico hospitals. For a complete discussion regarding the methodology for calculating Factor 3 for FY 2025, we refer readers to section IV.E. of the preamble of this final rule. For a discussion regarding the methodology for calculating the supplemental payments, we refer readers to section IV.D. of the preamble of this final rule.
To estimate the impact of the combined effect of the changes in Factors 1 and 2, as well as the changes to the data used in determining Factor 3, on the calculation of Medicare UCP along with changes to supplemental payments for IHS/Tribal hospitals and hospitals located in Puerto Rico, we compared total UCP and supplemental payments estimated in the FY 2024 IPPS/LTCH PPS final rule correction notice ( 88 FR 68484 ) to the combined total of the proposed UCP and the supplemental payments estimated in this FY 2025 IPPS/LTCH PPS final rule. For FY 2025, we calculated 75 percent of the estimated amount that would be paid as Medicare DSH payments absent section 3133 of the Affordable Care Act, adjusted by a Factor 2 of 59.29 percent and multiplied by a Factor 3 calculated using the methodology described in the FY 2024 IPPS/LTCH PPS final rule. For FY 2025, we calculated 75 percent of the estimated amount that would be paid as Medicare DSH payments during FY 2025 absent section 3133 of the Affordable Care Act, adjusted by a Factor 2 of 54.29 percent and multiplied by a Factor 3 calculated using the methodology described previously. For this final rule, the supplemental payments for IHS/Tribal hospitals and Puerto Rico hospitals are calculated as the difference between the hospital's adjusted base year amount (as determined based on the hospital's FY 2022 uncompensated care payment) and the hospital's FY 2025 uncompensated care payment.
Our analysis included 2,399 hospitals that are projected to be DSH-eligible in FY 2025. Our analysis did not include hospitals that had terminated their participation in the Medicare program as of February 2, 2024, Maryland hospitals, new hospitals, and SCHs that are expected to be paid based on their hospital-specific rates. The 23 hospitals that are anticipated to be participating in the Rural Community Hospital Demonstration Program were also excluded from this analysis, as participating hospitals are not eligible to receive empirically justified Medicare DSH payments and uncompensated care payments. In addition, the data from merged or acquired hospitals were combined under the surviving hospital's CMS certification number (CCN), and the non-surviving CCN was excluded from the analysis. The estimated impact of the changes in Factors 1, 2, and 3 on UCP and supplemental payments for eligible IHS/Tribal hospitals and Puerto Rico hospitals across all hospitals projected to be DSH-eligible in FY 2025, by hospital characteristic, is presented in the following table:

The changes in projected FY 2025 UCP and supplemental payments compared to the total of UCP and supplemental payments in FY 2024 are driven by changes in Factor 1 and Factor 2. Factor 1 has increased from the FY 2024 final rule's Factor 1 of $10.015 billion to this final rule's Factor 1 of $10.457 billion. Factor 2 has decreased from the FY 2024 final rule's Factor 2 of 59.29 percent to this final rule's Factor 2 of 54.29 percent. In addition, we note that there is a slight increase in the number of projected DSH-eligible hospitals to 2,399 at the time of the development of this final rule compared to the 2,384 DSH-eligible hospitals in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 58640 ). Based on the changes, the impact analysis found that, across all projected DSH-eligible hospitals, FY 2025 UCP and supplemental payments are estimated at approximately $5.786 billion, or a decrease of approximately 3.91 percent from FY 2024 UCP and supplemental payments (approximately $6.021 billion). While the changes result in a net decrease in the total amount available to be distributed in UCP and supplemental payments, the projected payment amounts vary by hospital type. This redistribution of payments is caused by changes in Factor 3 and the amount of the supplemental payment for DSH-eligible IHS/Tribal hospitals and Puerto Rico hospitals. As seen in the previous table, a percent change of less than negative 3.91 percent indicates that hospitals within the specified category are projected to experience a larger decrease in payments, on average, compared to the universe of projected FY 2025 DSH-eligible hospitals. Conversely, a percentage change greater than negative 3.91 percent indicates that a hospital type is projected to have a smaller decrease compared to the overall average. The variation in the distribution of overall payments by hospital characteristic is largely dependent on a given hospital's uncompensated care costs as reported on the Worksheet S-10 and used in the Factor 3 computation and whether the hospital is eligible to receive the supplemental payment.
Rural hospitals, in general, are projected to experience a smaller decrease in UCP compared to the decrease their urban counterparts are projected to experience. Overall, rural hospitals are projected to receive a 1.16 percent decrease in payments, while urban hospitals are projected to receive a 4.07 percent decrease in payments, which is slightly larger than the overall hospital average.
By bed size, rural hospitals with 0 to 99 beds are projected to receive a smaller than average decrease of 2.97 percent in payments, while those with 100 to 249 beds are projected to receive an increase of 1.14. Additionally, rural hospitals with 250+ beds are projected to receive a 0.59 percent increase in payments. Among urban hospitals, the smallest urban hospitals, those with 0 to 99 beds, are projected to receive a 3.43 percent increase in payments. In contrast, larger urban hospitals with 100-249 beds and urban hospitals with 250+ beds are projected to receive decreases in payments that are larger than the overall hospital average, by 4.81 and 4.26 percent, respectively.
By region, rural hospitals are projected to receive a varied range of payment changes. Rural hospitals in the New England, West North Central, and Middle Atlantic regions are projected to receive larger than average decreases in payments. Rural hospitals in all other regions are projected to receive either increases in payments or smaller than average decreases in payments. Urban hospitals in the West South Central, Mountain, and Pacific regions are projected to receive either increases in payments or smaller than average decreases in payments, while urban hospitals in all other regions are projected to receive larger than average decreases in payments.
By payment classification, hospitals in urban payment areas overall are expected to receive a 3.71 percent decrease in UCP and supplemental payments. Hospitals in large urban payment areas are projected to receive a smaller than average decrease in payments of 2.36 percent. In contrast, hospitals in other urban payment areas and hospitals in rural ( print page 70015) payment areas are projected to receive larger than average decreases in payments of 5.69 and 4.13 percent, respectively.
Nonteaching hospitals and teaching hospitals with 100+ residents are projected to receive smaller than average payment decreases of 3.27 percent and 3.47 percent, respectively. Teaching hospitals with fewer than 100 residents are projected to receive larger than average payment decreases of 4.87 percent. Voluntary hospitals are projected to receive larger than average decreases of 4.61 percent, while government-owned hospitals and proprietary hospitals are expected to receive smaller than average payment increases of 2.63 percent and 3.59 percent, respectively. Hospitals with less than 25 percent Medicare utilization are projected to receive smaller than average decreases of 3.22 percent. Hospitals with Medicare utilization between 25-50 percent, 50-65 percent, and greater than 65 percent are projected to receive larger than average decreases of 5.58 percent, 8.28 percent, and 7.06 percent, respectively. Hospitals with 50-65 percent Medicaid utilization are projected to receive a smaller than average decrease in payments of 1.93 percent, while those with greater than 65 percent Medicaid utilization are projected to receive a 5.79 percent increase in payments. Meanwhile, hospitals with less than 25 percent Medicaid utilization and those with Medicaid utilization between 25-50 percent are projected to receive larger than average decreases of 4.44 percent and 4.31 percent, respectively.
The impact table reflects the modeled FY 2025 UCP and supplemental payments for IHS/Tribal and Puerto Rico hospitals. We note that the supplemental payments to IHS/Tribal hospitals and Puerto Rico hospitals are estimated to be approximately $79.9 million in FY 2025.
In section V.D. of the preamble of this final rule, we discuss the legislative extension of the temporary changes to the low-volume hospital payment policy originally provided for by the Affordable Care Act and extended by subsequent legislation. Specifically, section 306 of the CAA, 2024 further extended the modified definition of low-volume hospital and the methodology for calculating the payment adjustment for low-volume hospitals under section 1886(d)(12) through December 31, 2024. Beginning January 1, 2025, the low-volume hospital qualifying criteria and payment adjustment will revert to the statutory requirements that were in effect prior to FY 2011, and the preexisting low-volume hospital payment adjustment methodology and qualifying criteria, as implemented in FY 2005, will resume. Effective for FY 2025, discharges occurring on or after January 1, 2025 and subsequent years, in order to qualify as a low-volume hospital, a subsection (d) hospital must be more than 25 road miles from another subsection (d) hospital and have less than 200 discharges (that is, less than 200 discharges total, including both Medicare and non-Medicare discharges) during the fiscal year. We recognize the importance of this extension with respect to the goal of advancing health equity by addressing the health disparities that underlie the health system, which is one of CMS' strategic pillars and a Biden-Harris Administration priority, as described in section I.A.2. of the preamble of this final rule. The provisions of section 306 of the CAA, 2024 are projected to increase payments to IPPS hospitals by approximately $89 million in FY 2025 relative to what the payments would have been in the absence of section 306.
Based upon the best available data at this time, we estimate the expiration of the temporary changes to the low-volume hospital payment policy for FY 2025 discharges occurring on or after January 1, 2025 will decrease aggregate low-volume hospital payments by $267 million in FY 2025 as compared to FY 2024. These payment estimates were determined based on the estimated payments for the approximately 600 providers that are expected to no longer qualify under the criteria that will apply beginning on January 1, 2025. These impacts were calculated using the same methodology used in developing the quantitative analyses of changes in payments per case discussed previously in section I.G. of this Appendix A of this final rule.
In section V.F.2. of this final rule, we are finalizing our proposal to implement section 4122 of the CAA, 2023, which requires that the Secretary initiate an application round to distribute 200 residency positions (also referred to as slots) with at least 100 of the positions being distributed for psychiatry or psychiatry subspecialty residency programs. The residency positions distributed under section 4122 are effective July 1, 2026.
Under our final policy, we'll first distribute slots by prorating the available 200 positions among all qualifying hospitals that apply for such slots, such that each qualifying applicant hospital will receive up to 1.00 FTE − that is, 1.00 FTE or a fraction of 1.00 FTE. According to our final policy, a qualifying hospital is a Category One, Category Two, Category Three, or Category Four hospital, or one that meets the definitions of more than one of these categories, as defined at section 1886(h)(10)(F)(iii) of the Act. [ 1126 ] We also finalized that if any residency slots remain after distributing up to 1.00 FTE to each such qualifying hospital, we will prioritize the distribution of the remaining slots based on the HPSA score associated with the program for which each qualifying hospital is applying using the methodology we finalized for purposes of implementing section 126 of the CAA, 2021 ( 86 FR 73434 through 73440 ). Using this HPSA prioritization method, a qualifying hospital's total award under section 4122 of the CAA, 2023, will be limited to 10.00 additional FTEs consistent with section 1886(h)(10)(C)(i) of the Act. We believe including such a prioritization will further support the training of residents in underserved and rural areas thereby helping to address physician shortages and the larger issue of health inequities in these areas.
The CMS Office of the Actuary (OACT) estimates an increase of $10 million in Medicare payments to teaching hospitals for FY 2026, and an increase in Medicare payments to teaching hospitals of $280 million for FYs 2026 through 2030 (over 5 years). In total, for FYs 2026 through 2036, Medicare payments to teaching hospitals are estimated to increase by $740 million.
In addition, we are finalizing a modification to our methodology for distributing slots under section 126 of the CAA, 2021. Section 1886(h)(9)(B)(ii) of the Act requires the Secretary to distribute at least 10 percent of the aggregate number of total residency positions available to the same four categories of hospitals. Section 126 of the CAA, 2021, makes available 1,000 residency positions and therefore, at least 100 residency positions must be distributed to hospitals qualifying in each of the four categories. In the final rule implementing section 126 of the CAA, 2021, we stated we would track progress in meeting all statutory requirements and evaluate the need to modify the distribution methodology in future rulemaking ( 86 FR 73441 ). To date, we have the completed the distribution of residency slots under rounds 1 and 2 of the section 126 distributions and have determined that only 12.76 DGME slots and 18.06 IME slots were distributed to hospitals qualifying under Category Four. Under our final policy, in rounds 4 and 5 we will prioritize the distribution of slots to hospitals that qualify under Category Four, regardless of HPSA score, to ensure that at least 100 residency slots are distributed to these hospitals. The remaining slots awarded under rounds 4 and 5 will be distributed using the existing methodology based on HPSA score ( 86 FR 73434 through 73440 ). That is, the remaining slots will be distributed to hospitals qualifying under Category One, Category Two, or Category ( print page 70016) Three, or hospitals that meet the definition of more than one of these categories, based on the HPSA score associated with the program for which each hospital is applying. We believe there is a minimal impact on Medicare payments associated with this change in methodology as the number of total slots distributed will remain the same.
As discussed in section V.I. of the preamble of this final rule, we are finalizing our proposal to update our payment methodology for determining the ESRD add-on payment for hospitals with a high percentage of ESRD beneficiary discharges. Currently under § 412.104(b), the ESRD add-on is based on the average length of stay (in days) for ESRD beneficiaries in the hospital, expressed as a ratio to 1 week (7 days), multiplied by the estimated weekly cost of dialysis, then multiplied by the number of ESRD beneficiary discharges (Worksheet E Part A Column 1 line 41.01). After consideration of public comments, we are finalizing our proposal that, effective for cost reporting periods beginning on or after October 1, 2024, the estimated weekly cost of dialysis will be calculated as the ESRD PPS base rate (as defined in 42 CFR 413.171 ) multiplied by three. As proposed, under this policy, the CY 2025 ESRD PPS base rate will be used for all cost reports beginning during Federal FY 2025 (that is, for cost reporting periods starting on or after October 1, 2024, through September 30, 2025).
Our impact analysis includes 91 hospitals that were eligible for the ESRD add-on payment based on the historical composite rate in the FY 2017 cost report data, which is a historical year that has a high percentage of final settled cost report data regarding ESRD add-on payments. As we did in the proposed rule ( 89 FR 36620 ), we estimated the impact of the payment methodology by comparing total ESRD add-on payments from the December 2023 update of the FY 2017 cost report data to the estimated FY 2025 ESRD add-on payments using, for illustrative purposes, the CY 2024 ESRD PPS base rate published in the CY 2024 ESRD PPS final rule ( 88 FR 76345 ), which is $271.02. (As previously noted, the CY 2025 ESRD PPS base rate will be used for all cost reports beginning during Federal FY 2025 (that is, for cost reporting periods starting on or after October 1, 2024, through September 30, 2025).) The total ESRD add-on payments based on the FY 2017 cost report data are approximately $22 million. The total estimated FY 2025 ESRD add-on payments, as estimated using the CY 2024 ESRD PPS base rate, will be approximately $31.4 million. Therefore, we estimated the ESRD add-on payments will increase by approximately $10 million.
As discussed in section V.K.1. of the preamble of this final rule, we are finalizing IPPS payment adjustments for the Medicare inpatient share of additional resource costs that small, independent hospitals incur in establishing and maintaining access to a 6-month buffer stock of one or more essential medicine(s), effective for cost reporting periods beginning on or after October 1, 2024.
We are finalizing this payment adjustment under the IPPS for the additional Medicare inpatient share of resource costs of establishing and maintaining access to a buffer stock of essential medicines under section 1886(d)(5)(I) of the Act.
The data currently available to calculate a spending estimate for FY 2025 under the IPPS is limited. However, we believe the methodology described in this section to calculate this spending estimate under the IPPS for FY 2025 is reasonable based on the information available.
To estimate total spending associated with this finalized policy under the IPPS, we used the following information for all eligible hospitals with completed 12-month or greater cost reporting periods concluding in CY 2021 (the most recent cost reporting period for which data was available):
- Estimated spend per eligible hospital on its applicable essential medicines, expressed as a percentage of the total Drugs Charged to Patients cost center, as found on Worksheet B, Part 1, line 73, column 26 on Form CMS-2552-2010. For purposes of this estimate, we believe it is reasonable to assume that the cost of a given hospital's essential medicines will be 1 percent of its total Drugs Charged to Patients costs.
- Multiplicative factor of 50 percent to estimate the total cost of the essential medicines that are in the 6-month buffer stock.
- Assumed cost of carrying essential medicines, expressed as a percentage of the total cost of the essential medicines that are in the buffer stock. Based on commenter feedback on the CY 2024 OPPS/ASC proposed rule, [ 1127 ] we believe it is reasonable to assume for purposes of this spending estimate a cost of carrying essential medicines of 20 percent of the cost of the essential medicines themselves. This assumption of a 20 percent cost of carrying would apply to any size of buffer stock of essential medicine.
- The provider-specific inpatient Medicare share percentage, expressed as the percentage of inpatient Medicare costs to total hospital costs.
To calculate the estimated aggregate IPPS payments under this finalized policy, we multiplied together the four factors listed for each eligible hospital and summed across all eligible hospitals. Based on the latest hospital cost report data available, we identified approximately 500 IPPS hospitals that would potentially be eligible for this finalized payment. Eligible IPPS hospitals are those providers that: (1) had 100 or fewer beds as defined in § 412.105(b); and (2) answered “N” to line 140, column 1 and did not fill out any part of lines 141 through 143 on Worksheet S2 Part I on Form CMS-2552-10. We estimate that the aggregate FY 2025 IPPS payments under this finalized policy, given the assumptions detailed previously, would be approximately $0.3 million, and the mean IPPS payment per eligible hospital would be approximately $620 over the course of a year. This policy would not be budget neutral under the IPPS.
We also estimated the total costs for eligible hospitals to establish and maintain buffer stocks of essential medicines in order to inform the public what portion of the total costs would be separately paid under the finalized policy. To calculate this, we multiplied together the first three factors listed previously for each eligible hospital, but not the fourth factor ( i.e. we did not multiply by the provider specific inpatient Medicare share percentage) and summed across all eligible hospitals. We estimate that the total annual costs for eligible hospitals to establish and maintain buffer stocks of essential medicines would be approximately $2.8 million, and the mean cost per eligible hospital would be approximately $5,610. The IPPS payments under this finalized policy represent approximately 11 percent of that amount, or $0.3 million.
As discussed earlier, our estimate was calculated at the hospital level and then summed. However, for illustrative purposes the calculation can be described alternatively as starting with the aggregated total Drugs Charged to Patients across eligible hospitals of approximately $2.8 billion, assuming the annual cost of essential medicines to be 1 percent of that amount or $28 million (=$2.8 billion * .01), calculating the cost of 6 months of essential medicines as half that amount or $14 million (=$28 million * .50), assuming that the cost of carrying essential medicines is 20 percent of that amount or $2.8 million (=$14 million * .20), and then calculating the Medicare inpatient share of that amount at 11 percent or $0.3 million (= $2.8 million * .11).
We sought comment on these assumptions and estimates.
Comment: Although many commenters raised concerns that the estimated average payment is inadequate to cover the costs of buffer stock acquisition, storage, maintenance, and other related costs, we did not receive comments regarding our assumptions and estimates for purposes of estimating the effects of the IPPS payment adjustment for establishing and maintaining access to essential medicines.
Response: We thank the commenters for their feedback regarding the payments. We agree with commenters that the IPPS payment adjustment under this policy does not equal to the total reasonable costs to establish and maintain a buffer stock of essential medicines. This is by definition as the policy is limited to the Medicare inpatient share of that amount. We also note that the average IPPS payment does not reflect the range of potential payments under this policy as these payments will be hospital specific depending on each hospital's reasonable costs of maintaining and establishing its buffer stocks and its Medicare inpatient share of those costs. In response to comments and to illustrate this point, we have calculated selected percentiles of estimated total reasonable costs and the estimated IPPS payments under this policy in Table K-CDD-1.

After consideration of the comments received, we continue to believe the methodology described in this section to calculate this spending estimate under the IPPS for FY 2025 is reasonable based on the information currently available.
In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36238 ), we did not propose to add, modify, or remove any measures or policies for the FY 2025 Hospital Readmissions Reduction Program; the policies finalized in FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49081 through 49094 ) continue to apply. This program requires a reduction to a hospital's base operating diagnosis-related group (DRG) payments to account for excess readmissions of selected applicable conditions and procedures. Table I.G.7.-01 and the analysis in this final rule illustrate the estimated financial impact of the Hospital Readmissions Reduction Program payment adjustment methodology by hospital characteristic. Hospitals are sorted into quintiles based on the proportion of dual-eligible stays among Medicare fee-for-service (FFS) and managed care stays between July 1, 2020 and June 30, 2023 (that is, the FY 2025 Hospital Readmissions Reduction Program's applicable period, which is the most recently available data at the time of publication of this final rule). Hospitals' excess readmission ratios (ERRs) are assessed relative to their peer group median and a neutrality modifier is applied in the payment adjustment factor calculation to maintain budget neutrality. In this FY 2025 IPPS/LTCH PPS final rule, we are providing an updated estimate of the financial impact using the proportion of dually-eligible beneficiaries, ERRs, and aggregate payments for each condition/procedure and all discharges for applicable hospitals from the FY 2025 Hospital Readmissions Reduction Program applicable period (that is, July 1, 2020, through June 30, 2023).
The results in Table I.G.7.-01 include 2,828 non-Maryland hospitals estimated as eligible to receive a penalty during the performance period. Hospitals are eligible to receive a penalty if they have 25 or more eligible discharges for at least one measure between July 1, 2020, and June 30, 2023. The second column in Table I.G.7.-01 indicates the total number of non-Maryland hospitals with available data for each characteristic that have an estimated payment adjustment factor less than 1 (that is, penalized hospitals).
The third column in Table I.G.7.-01 indicates the estimated percentage of penalized hospitals among those eligible to receive a penalty by hospital characteristic. For example, 78.34 percent of eligible hospitals characterized as non-teaching hospitals are expected to be penalized. Among teaching hospitals, 88.57 percent of eligible hospitals with fewer than 100 residents and 90.14 percent of eligible hospitals with 100 or more residents are expected to be penalized. The fourth column in Table I.G.7.-01 estimates the financial impact on hospitals by hospital characteristic. Table I.G.7.-01 also shows the share of penalties as a percentage of all base operating DRG payments for hospitals with each characteristic. This is calculated as the sum of penalties for all hospitals with that characteristic over the sum of all base operating DRG payments for those hospitals between October 1, 2022, through September 30, 2023 (FY 2023). For example, the penalty as a share of payments for non-teaching hospitals is 0.45 percent. This means that total penalties for all non-teaching hospitals are 0.45 percent of total payments for non-teaching hospitals. Measuring the financial impact on hospitals as a percentage of total base operating DRG payments accounts for differences in the amount of base operating DRG payments for hospitals with the characteristic when comparing the financial impact of the program on different groups of hospitals.

The Secretary makes value-based incentive payments to hospitals under the Hospital Value-Based Purchasing Program based on their performance on measures during the performance period with respect to a fiscal year. These incentive payments will be funded for FY 2025 through a reduction to the FY 2025 base operating DRG payment amount for hospital discharges for such fiscal year, as required by section 1886(o)(7)(B) of the Act. The applicable percentage for FY 2025 and subsequent years is two percent. The total amount available for value-based incentive payments must be equal to the total amount of reduced payments for all hospitals for the fiscal year, as estimated by the Secretary. In section V.L.1.b. of the preamble of this final rule, we estimate the available pool of funds for value-based incentive payments in the FY 2025 program year, which, in accordance with section 1886(o)(7)(C)(v) of the Act, will be 2.00 percent of base operating DRG payments, or a total of approximately $1.67 billion. This estimated available pool for FY 2025 is based on the historical pool of hospitals that were eligible to participate in the FY 2024 program year and the payment information from the March 2024 update to the FY 2023 MedPAR file.
The estimated impacts of the FY 2025 program year by hospital characteristic, found in Table I.8.-01., are based on historical TPSs. We used the FY 2024 program year's TPSs to calculate the proxy adjustment factors used for this impact analysis. These are the most recently available scores that hospitals were given an opportunity to review and correct. The proxy adjustment factors use estimated annual base operating DRG payment amounts derived from the March 2024 update to the FY 2023 MedPAR file. The proxy adjustment factors can be found in Table 16A associated with this final rule (available via the internet on the CMS website).
The impact analysis shows that, for the FY 2025 program year, the number of hospitals with a positive percent change in base operating DRG (49.7 percent) is lower than the number of hospitals with a negative percent change (50.3 percent). Approximately half of all hospitals experience a percent change in base operating DRG between -2.1 percent and 0.0 percent. On average, urban and rural hospitals in the West North Central and Pacific regions have the highest positive percent change in base operating DRG. Urban hospitals in the Middle Atlantic, East South Central, and West South Central regions experience a negative average percent change in base operating DRG. All other regions (both urban and rural) experience a positive average % change in base operating DRG. With respect to hospitals' Medicare utilization as a percent of inpatient days (MCR), as the MCR percent increases, the average percent change in base operating DRG increases. As DSH percent increases, the average percent change in base operating DRG generally decreases. On average, non-teaching hospitals have a higher percent change in base operating DRG compared to teaching hospitals.

We are presenting the estimated impact of the FY 2025 Hospital-Acquired Condition (HAC) Reduction Program on hospitals by hospital characteristic based on previously adopted policies for the program. In the FY 2025 IPPS/LTCH PPS proposed rule, we did not propose to add or remove any measures from the HAC Reduction Program, nor did we propose any changes to reporting or submission requirements which would have any significant economic impact for the FY 2025 program year or future years. The table in this section presents the estimated proportion of hospitals in the worst-performing quartile of Total HAC Scores by hospital characteristic. Hospitals' CMS Patient Safety and Adverse Events Composite (CMS PSI 90) measure results are based on Medicare fee-for-service (FFS) discharges from July 1, 2021 through June 30, 2023 and version 14.0 of the PSI software. Hospitals' measure results for Centers for Disease Control and Prevention (CDC) Central Line-Associated Bloodstream Infection (CLABSI), Catheter-Associated Urinary Tract Infection (CAUTI), Colon and Abdominal Hysterectomy Surgical Site Infection (SSI), Methicillin-resistant Staphylococcus aureus (MRSA) bacteremia, and Clostridium difficile Infection (CDI) are derived from standardized infection ratios (SIRs) calculated with hospital surveillance data reported to the CDC's National Healthcare Safety Network (NHSN) for infections occurring between January 1, 2022 and December 31, 2023. Hospital characteristics are based on the FY 2025 IPPS Proposed Rule Impact File.
This table includes 2,933 non-Maryland hospitals with an estimated FY 2025 Total HAC Score based on the most recently available data at the time of publication of this final rule. Maryland hospitals and hospitals without a Total HAC Score are excluded from the table. Actual results for FY 2025 will be determined in the fall of 2024 after a 30-day review and corrections period for hospitals to review their program results. The first column presents a breakdown of each characteristic, and the second column indicates the number of hospitals for the respective characteristic.
The third column in the table indicates the estimated number of hospitals for each characteristic that would be in the worst-performing quartile of Total HAC Scores. For example, with regard to teaching status, 426 hospitals out of 1,700 hospitals characterized as non-teaching hospitals would be subject to a payment reduction. Among teaching hospitals, 196 out of 935 hospitals with fewer than 100 residents and 102 out of 285 hospitals with 100 or more residents would be subject to a payment reduction.
The fourth column in the table indicates the estimated proportion of hospitals for each characteristic that would be in the worst-performing quartile of Total HAC Scores and thus receive a payment reduction under the FY 2025 HAC Reduction Program. For example, 25.1 percent of the 1,700 hospitals characterized as non-teaching hospitals, 21.0 percent of the 935 teaching hospitals with fewer than 100 residents, and 35.8 percent of the 285 teaching hospitals with 100 or more ( print page 70023) residents would be subject to a payment reduction.

In section II.A.4.h. of the Addendum of this final rule for FY 2025, we discussed our budget neutrality methodology for section 410A of Public Law 108-173 , as amended by sections 3123 and 10313 of Pub. L 111-148 , by section 15003 of Public Law 114-255 , and most recently, by section 128 of Public Law 116-260 , which requires the Secretary to conduct a demonstration that would modify payments for inpatient services for up to 30 rural hospitals.
Section 128 of Public Law 116-260 requires the Secretary to conduct the Rural Community Hospital Demonstration for a 15-year extension period (that is, for an additional 5 years beyond the previous extension period). In addition, the statute provides for continued participation for all hospitals participating in the demonstration program as of December 30, 2019.
Section 410A(c)(2) of Public Law 108-173 requires that in conducting the demonstration program under this section, the Secretary shall ensure that the aggregate payments made by the Secretary do not exceed the amount which the Secretary would have paid if the demonstration program under this section was not implemented (budget neutrality). We propose to adopt the general methodology used in previous years, whereby we estimated the additional payments made by the program for each of the participating hospitals as a result of the demonstration, and then adjusted the national IPPS rates by an amount sufficient to account for the added costs of this demonstration. In other words, we have applied budget neutrality across the payment system as a whole rather than across the participants of this demonstration. The language of the statutory budget neutrality requirement permits the agency to implement the budget neutrality provision in this manner. The statutory language requires that aggregate payments made by the Secretary do not exceed the amount which the Secretary would have paid if the demonstration was not implemented, but does not identify the range across which aggregate payments must be held equal.
For this final rule, the resulting amount applicable to FY 2025 for 22 participating hospitals is $49,914,526, which we are incorporating into the budget neutrality offset adjustment for FY 2025. This estimated amount is based on the specific assumptions regarding the data sources used, that is, recently available “as submitted” cost reports and historical and currently finalized update factors for cost and payment.
In previous years, we have incorporated a second component into the budget neutrality offset amounts identified in the final IPPS rules. As finalized cost reports became available, we determined the amount by which the actual costs of the demonstration for an earlier, given year differed from the estimated costs for the demonstration set forth in the final IPPS rule for the corresponding fiscal year, and we incorporated that amount into the budget neutrality offset amount for the upcoming fiscal year. We have calculated this difference for FYs 2005 through 2018 between the actual costs of the demonstration as determined from finalized cost reports once available, and estimated costs of the demonstration as identified in the applicable IPPS final rules for these years.
With the extension of the demonstration for another 5-year period, as authorized by section 128 of Public Law 116-260 , we will continue this general procedure. Thus, we are including in the budget neutrality offset amount in the FY 2025 final rule the amount by which the actual costs of the demonstration, as determined from finalized cost reports and revisions by the MACs for ( print page 70025) the 27 hospitals that completed cost reporting periods beginning in FY 2019, differed from the estimated costs identified in the FY 2019 final rule. Accordingly, the actual costs of the demonstration for FY 2019 fell short of the estimated amount in the FY 2019 by $30,499,707. This amount is subtracted from the estimated amount for FY 2025, resulting in $19,414,819, which represents the budget neutrality offset amount to be applied to the national IPPS rates for FY 2025.
Response: We appreciate the comments. We have conducted the demonstration program in accordance with Congressional mandates. Title XVIII does not extend authority to make the demonstration a permanent program. With regard to any further actions, we intend to work with the commenter and other rural stakeholders to examine the issues involved.
As described in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59119 through 59122 ), CMS waived certain Medicare rules for CAHs participating in the demonstration extension period to allow for alternative reasonable cost-based payment methods in the three distinct intervention service areas: telehealth services, ambulance services, and skilled nursing facility/nursing facility services. These waivers were implemented with the goal of increasing access to care with no net increase in costs. As we explained in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59119 through 59122 ), section 129 of Public Law 116-159 , stipulates that only the 10 CAHs that participated in the initial period of the FCHIP Demonstration are eligible to participate during the extension period. Among the eligible CAHs, five elected to participate in the extension period. The selected CAHs are located in two states—Montana and North Dakota—and are implementing the three intervention services.
As explained in the FY 2024 IPPS/LTCH PPS final rule, we based our selection of CAHs for participation in the demonstration with the goal of maintaining the budget neutrality of the demonstration on its own terms meaning that the demonstration would produce savings from reduced transfers and admissions to other health care providers, offsetting any increase in Medicare payments as a result of the demonstration. However, because of the small size of the demonstration and uncertainty associated with the projected Medicare utilization and costs, the policy we finalized for the demonstration extension period of performance in the FY 2024 IPPS/LTCH PPS final rule provides a contingency plan to ensure that the budget neutrality requirement in section 123 of Public Law 110-275 is met.
In the FY 2024 IPPS/LTCH PPS final rule, we adopted the same budget neutrality policy contingency plan used during the demonstration initial period to ensure that the budget neutrality requirement in section 123 of Public Law 110 275 is met during the demonstration extension period. If analysis of claims data for Medicare beneficiaries receiving services at each of the participating CAHs, as well as from other data sources, including cost reports for the participating CAHs, shows that increases in Medicare payments under the demonstration during the 5-year extension period is not sufficiently offset by reductions elsewhere, we will recoup the additional expenditures attributable to the demonstration through a reduction in payments to all CAHs nationwide.
As explained in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59119 through 59122 ), because of the small scale of the demonstration, we indicated that we did not believe it would be feasible to implement budget neutrality for the demonstration extension period by reducing payments to only the participating CAHs. Therefore, in the event that this demonstration extension period is found to result in aggregate payments in excess of the amount that would have been paid if this demonstration extension period were not implemented, CMS policy is to comply with the budget neutrality requirement finalized in the FY 2024 IPPS/LTCH PPS final rule, by reducing payments to all CAHs, not just those participating in the demonstration extension period.
In the FY 2024 IPPS/LTCH PPS final rule, we stated that we believe it is appropriate to make any payment reductions across all CAHs because the FCHIP Demonstration was specifically designed to test innovations that affect delivery of services by the CAH provider category. As we explained in the FY 2024 IPPS/LTCH PPS final rule, we believe that the language of the statutory budget neutrality requirement at section 123(g)(1)(B) of Public Law 110-275 permits the agency to implement the budget neutrality provision in this manner. The statutory language merely refers to ensuring that aggregate payments made by the Secretary do not exceed the amount which the Secretary estimates would have been paid if the demonstration project was not implemented and does not identify the range across which aggregate payments must be held equal.
In the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45323 through 45328 ), CMS concluded that the initial period of the FCHIP Demonstration had satisfied the budget neutrality requirement described in section 123(g)(1)(B) of Pub L. 110-275 . Therefore, CMS did not apply a budget neutrality payment offset policy for the initial period of the demonstration. As explained in the FY 2022 IPPS/LTCH PPS final rule, we finalized a policy to address the demonstration budget neutrality methodology and analytical approach for the initial period of the demonstration. In the FY 2024 IPPS/LTCH PPS final rule, we finalized a policy to adopt the same budget neutrality methodology and analytical approach used during the demonstration initial period to be used for the demonstration extension period. As stated in the FY 2024 IPPS/LTCH PPS final rule ( 88 FR 59119 through 59122 ), our policy for implementing the 5-year extension period for section 129 of Public Law 116-260 follows same budget neutrality methodology and analytical approach as the demonstration initial period methodology. While we expect to use the same methodology that was used to assess the budget neutrality of the FCHIP Demonstration during initial period of the demonstration to assess the financial impact of the demonstration during this extension period, upon receiving data for the extension period, we may update and/or modify the FCHIP budget neutrality methodology and analytical approach to ensure that the full impact of the demonstration is appropriately captured. Therefore, we did not propose to apply a budget neutrality payment offset to payments to CAHs in FY 2025. This policy will have no impact for any national payment system for FY 2025. We received no comments on this provision and therefore are finalizing this provision without modification.
In section X.A. of the preamble of this final rule, we are finalizing the test of a new mandatory episode-based payment model titled the Transforming Episode Accountability Model (TEAM) under the authority of the CMS Center for Medicare and Medicaid Innovation (CMS Innovation Center). Section 1115A of the Act authorizes the CMS Innovation Center to test innovative payment and service delivery models that preserve or enhance the quality of care furnished to Medicare, Medicaid, and Children's Health Insurance Program beneficiaries while reducing program expenditures. The intent of TEAM is to improve beneficiary care through financial accountability for episode categories that begin with one of the following procedures: coronary artery bypass graft, lower extremity joint replacement, major bowel procedure, surgical hip/femur fracture treatment, and spinal fusion. TEAM will test whether financial accountability for these episode categories reduces Medicare expenditures while preserving or enhancing the quality of care for Medicare beneficiaries. We anticipate that TEAM may benefit Medicare beneficiaries through improving the coordination of items and services paid for through Medicare fee-for-service (FFS) payments, encouraging provider investment in health care infrastructure and redesigned care processes, and incentivizing higher value care across the inpatient and post-acute care settings for the episode.
TEAM will require acute care hospitals located within selected mandatory CBSAs to participate in the model. CMS will allow a one-time opportunity for hospitals that participate until the last day of the last ( print page 70026) performance period in the BPCI Advanced model or the last day of the last performance year of the CJR model, that are not located in a mandatory CBSA selected for TEAM participation to voluntarily opt into TEAM. [ 1128 ] This episode-based payment model will begin on January 1, 2026, and end on December 31, 2030. Payment approaches that hold providers accountable for episode cost and performance can potentially create incentives for the implementation and coordination of care redesign between participants and other providers and suppliers such as physicians and post-acute care providers. TEAM could enable hospitals to consider the most appropriate strategies for care redesign, including (1) increasing post-hospitalization follow-up and medical management for patients; (2) coordinating care across the inpatient and post-acute care spectrum; (3) conducting appropriate discharge planning; (4) improving adherence to treatment or drug regimens; (5) reducing readmissions and complications during the post-discharge period; (6) managing chronic diseases and conditions that may be related to the proposed episodes; (7) choosing the most appropriate post-acute care setting; and (8) coordinating between providers and suppliers such as hospitals, physicians, and post-acute care providers.
Under this model, TEAM participants will continue to bill Medicare under the traditional FFS system for items and services furnished to Medicare FFS beneficiaries. The TEAM participant may receive a reconciliation payment from CMS if Medicare FFS expenditures for a performance year are less than the reconciliation target price, subject to a quality adjustment. TEAM will not have downside risk for Track 1 and TEAM participants will only be accountable for performance year spending below their reconciliation target price, subject to a quality adjustment, that will result in a reconciliation payment amount. For Track 2 and Track 3, TEAM will be a two-sided risk model that requires TEAM participants to be accountable for performance year spending above or below their reconciliation target price, subject to a quality adjustment, that will result in a reconciliation payment amount or a repayment amount.
TEAM is a mandatory episode-based payment model which will have a direct effect on the Medicare program because TEAM participants will be incentivized to reduce Medicare spending. Additionally, TEAM participants may receive a reconciliation payment amount from CMS or have to pay CMS a repayment amount based on their spending and quality performance. Table I.G.12-01 shows the projected financial impacts of TEAM over the course of the five-year model test. The first performance year (2026) of TEAM is expected to cost the Medicare program $38 million because we assume most TEAM participants will elect participation in Track 1, which is not subject to downside risk. In performance year 2 (2027), TEAM participants in Track 1 will have no downside risk while TEAM participants in Track 2 and Track 3 will be subject to both upside and downside risk, and we estimate TEAM participants on net (that is, repayment amounts less reconciliation payments) will pay $37 million to CMS, and that TEAM will save the Medicare program $96 million. To protect TEAM participants from significant financial risk, we have finalized a 5 percent stop-loss and stop-gain limit for TEAM participants in Track 2 and a 20 percent stop-loss and stop-gain limit for TEAM participants in Track 3. These limits will cap the total amount of repayments paid by TEAM participants to CMS or cap the total amount of reconciliation payment amounts paid by CMS to TEAM participants. In performance year 3 (2028), we estimate TEAM participants on net will pay $68 million to CMS, and that TEAM will save the Medicare program $129 million. We estimate that TEAM participants on net will pay CMS $93 million in performance year 4 and $77 million in performance year 5, and that TEAM will save the Medicare program $154 million and $140 million for these performance years, respectively. We estimate that, CMS will pay TEAM participants $442 million and TEAM participants will pay CMS $622 million, and that TEAM will save the Medicare program approximately $481 million over the 5 performance years (2026 through 2030).

We assumed TEAM episode volume is estimated to grow at the same rate as projected Medicare FFS enrollment as indicated in the 2023 Medicare Trustees Report. [ 1129 ] Further, an internal sample set of hospitals was used to estimate financial impacts and simulate TEAM participation. The amount of national episode spending captured by the sample set of hospitals was 29 percent in 2023.
We note that TEAM participants are estimated to reduce episode spending by 1 percent as a result of participating in TEAM. The fifth annual evaluation report of the Comprehensive Care for Joint Replacement (CJR) model indicated that CJR resulted in roughly a 4 percent reduction in lower extremity joint replacement (LEJR) spending (not including reconciliation payments) for participants over the course of the model. [ 1130 ] Since participation in CJR is mandatory in 34 metropolitan statistical areas, and LEJR episodes make up a significant portion of the episodes included in TEAM, the CJR evaluation results appear to be a reasonable proxy for what to expect in TEAM. However, the episode length in CJR is 90 days, whereas in TEAM the proposed length is 30 days. Internal analysis indicated that the 30-day episode is approximately 75 percent as costly as a 90-day episode for LEJR procedures. In addition, post-acute care spending has been declining in recent years for episodes that we are proposing to include in TEAM, which could limit the potential for TEAM participants to achieve significant improvements in efficiency. Thus, we believe that the intervention effect of TEAM on episode spending will be a reduction of 0 to 3 percent (see Table I.G.12-02 for a sensitivity analysis for how the financial impact is affected by changes in this assumption).
We also note that starting from actual episode spending that occurred in the first half 2023, average baseline spending per episode is estimated to increase by 1.5 ( print page 70027) percent every year. The national average per episode spending growth for all TEAM episode types in years 2018, 2019, 2022, and 2023 was approximately 1.3 percent. Annual growth rates for each episode type were weighted by spending, and historical experience during 2020 and 2021 were excluded due to possible impacts from the peak of the COVID-19 pandemic. Since some of the historical experience in these years includes Medicare policy changes for LEJR episodes that resulted in surgeries occurring in more efficient care settings, translating to spending decreases that may not be duplicated in future years, the assumed annual trend is slightly greater than the observed average trend from the historical experience.
Additionally, our estimates do not include the impact of TEAM beneficiary overlap with total cost of care models, such as when a TEAM beneficiary is also assigned to a Medicare Shared Savings Program ACO. However, given the precision in the Shared Savings Program projections, we do not anticipate a practical difference in the ACO's shared savings estimates. Nor do we anticipate TEAM beneficiary overlap with total cost of care models having a meaningful effect to TEAM's projected financial impacts, described in Table I.G.12-01.
TEAM will allow hospitals in the CJR and BPCI Advanced models that have remained in their respective models until the conclusion of those initiatives the option to voluntarily participate in TEAM. Impacts from these potential participants have not been included in our estimates due to the high degree of uncertainty regarding the level of interest that these potential participants will have in TEAM. We would expect that the majority of voluntary opt-in TEAM participants would come from the CJR model due to the large amount of attrition that has occurred in the BPCI Advanced model. We also expect that hospitals who would choose to opt into TEAM would include CJR participant hospitals that have consistently received positive reconciliation payments in recent years. Given the magnitude of reconciliation payments for CJR participant hospitals in recent years, we assume that the maximum potential costs of the voluntary opt-in policy will not jeopardize the overall direction of the net savings estimate.
Because the financial impact is based on projections of spending, the estimates implicitly assume that there will be no significant difference between the projected episode spending used to calculate the prospective target prices and actual episode spending. This assumption has a large degree of uncertainty, and the actual TEAM financial impacts will be sensitive to this difference. However, some the of the financial risk of the projection error is mitigated by the retrospective trend factor. Target prices will still be susceptible to some error risk if the projection error exceeds the retrospective trend factor cap. The direction, magnitude and timing of projection inaccuracies would all affect the overall financial impact estimate.
We also performed a sensitivity analysis to assess various intervention effects on TEAM. Overall financial impacts are sensitive to the intervention effect TEAM would have on TEAM participants' episode spending. Table I.G.12-02 includes financial impacts at various intervention effect assumptions (note that negative values indicate savings):

The sensitivity is due to the lack of the requirement that participants participate in downside risk during performance year 1 and the effect that reductions in episode spending during performance years would have on target prices for future performance years.
The following is a summary of comments we received on the effects to Medicare and our responses to these comments:
Comment: A commenter indicated that the impact analysis in the proposed rule estimated that TEAM will generate $705 million in net savings for the Medicare program and on net $403 million is projected to result from hospitals paying CMS because actual episode spending exceeded the target price but CMS did not estimate how much it will pay hospitals that generated a reconciliation payment amount.
Response: We thank the commenter for their question. For this final rule we have updated the Table I.G.12.-01, to reflect updated estimates as a result of final policy modifications. This updated table now separates reconciliation payment amounts and repayment amounts on discrete rows, rather than netting them on a single row, to allow the public to view the payments to TEAM participants (reconciliation payment amounts) and payments to CMS (repayment amounts) over the course of the five-year model performance period. The updated estimates indicate that we anticipate CMS will pay TEAM participants $442 million and TEAM participants will pay CMS $622 million as a result of participation in the model.
Comment: A couple of commenters had concerns that the proposed rule's impact analysis did not consider what it will cost hospitals to participate in TEAM. A commenter indicated that based on the volume projections for each episode and assuming 25% of the targeted CBSAs are required to participate, it is estimated that hospital costs to participate will range between $530 million and $744 million, which is between 75% to 106% of CMS' net projected savings and represent an unfunded mandate by the agency. Further, the costs to participate will either be cross-subsidized by the private sector, require hospitals to redeploy funding and resources from other outcome improvement efforts that are targeted to communities' needs, and/or result in further loss of access to services for Medicare beneficiaries and the broader community. Another commenter requested CMS estimate the potential costs of participation, and then draw on potentially relevant experience in the similar BPCI Advanced or CJR models to put forward a good faith estimate of what fraction of participating hospitals can expect to gain or lose money through participation.
Response: We appreciate the commenters' concerns or the potential economic impact on TEAM participants. We disagree with the commenter's estimate for how much TEAM will cost to implement and that it represents and unfunded mandate. We believe TEAM participants will not incur significant costs to implement TEAM because the administrative, monitoring, and compliance requirements for TEAM will not substantially diverge from existing requirements for Medicare providers. TEAM will not be adding to quality measure reporting or health equity reporting burden because we are using quality measures that TEAM participants will already be reporting for other CMS quality reporting programs and health equity reporting is voluntary. Nor does TEAM require TEAM participants to alter the way items and serviced are billed to Medicare, invest in technology or analytics, or increase human capital. A TEAM participant may wish to not change their behaviors or care practices, or devote resources to implementing the model, and they will still have financial protections that would prevent hospital costs equating to the commenter's estimates. Specifically, TEAM includes provisions such as the high-cost outlier cap that limits high-cost episode payments, and the stop-loss limit that restricts how much a TEAM participant may be required to pay CMS, as discussed in sections X.A.3.f.(3)(e) ( print page 70028) and X.A.3.f.(5)(h) of the preamble of this final rule. Further, all TEAM participants are eligible to participate in Track 1 for the first performance year, as discussed in section X.A.3.a.(3) of the preamble of this final rule, with no downside financial risk or up to two additional years of no downside risk for TEAM participants that are safety net hospitals, as defined in section X.A.3.f.(2) of the preamble of this final rule.
We also do not agree that TEAM will be cross-subsidized by the private sector, but in contrast we anticipate that private or commercial patients and payers may benefit from the care redesign inventions implemented by the TEAM participant. We believe many hospitals already have established standard care pathways and care teams that have experience managing beneficiaries who receive these procedures, so we do not expect TEAM to require a significant overhaul to care practices or significantly increase operating costs, but to rather encourage TEAM participants to introduce refinements to existing process that will create the efficiencies to improve quality and reduce spending. These efficiencies may result in spillover effects to other patients in the hospital that yield broader quality and spending improvements beyond TEAM, generating a positive financial impact for the TEAM participant and potentially other payers.
We also disagree that TEAM will result in Medicare beneficiaries losing access to services. TEAM participants may not limit access to medically necessary items and services, nor limit the TEAM beneficiary's choice of Medicare providers and suppliers. We will monitor beneficiary care, as discussed in section X.A.3.i of the preamble of this final rule, to ensure beneficiary access to care and freedom of choice is not compromised.
Lastly, we acknowledge the commenter's recommendation on drawing from experience in the BPCI Advanced and CJR models to estimate the fiscal impact to a hospital. However, we do not believe that this is something that can be accurately modeled given the high amount of uncertainty regarding individual hospital resources, capabilities, and care redesign interventions that might potentially be spurred by TEAM.
We believe that episode-based payment models may have the potential to benefit beneficiaries because the intent of the models is to test whether providers are able to improve the coordination and transition of care, invest in infrastructure and redesigned care processes for high quality and efficient service delivery and incentivize higher value care across the inpatient and post-acute care spectrum. We believe that episode-based payment models have a patient-centered focus such that they incentivize improved healthcare delivery and communication based on the needs of the beneficiary, thus potentially benefitting beneficiaries. We anticipate the model will not affect beneficiary cost sharing for items and services that beneficiaries receive from TEAM participants or premiums paid by beneficiaries. If there is a shift in the utilization of items and services within each episode, then beneficiary cost sharing could be higher or lower than would otherwise be experienced.
We are including a patient reported outcome measure, specific to LEJR episode categories, in the TEAM quality measures that will be tied to payment with the belief that doing so would encourage TEAM participants to focus on and deliver improved quality of care for Medicare beneficiaries. Additionally, TEAM participants must perform well on quality measure performance to achieve their maximum reconciliation payment. The accountability of TEAM participants for both quality and the cost of care that is furnished to TEAM beneficiaries within an episode provides TEAM participants with new incentives to improve the health and well-being of the Medicare beneficiaries they treat.
Additionally, the model does not affect the beneficiary's freedom of choice to obtain health services from any individual or organization qualified to participate in the Medicare program as guaranteed under section 1802 of the Act. Eligible beneficiaries who receive one of the five proposed surgical episode categories from a TEAM participant will not have the option to opt their episodes out of the model. TEAM participants may not prevent or restrict beneficiaries to any list of preferred or recommended providers.
Many controls exist under Medicare to ensure beneficiary access and quality, and we will use our existing authority, if necessary, to audit TEAM participants if claims analysis indicates an inappropriate change in delivered services. Given that TEAM participants may receive a reconciliation payment, subject to a quality adjustment, when they are able to reduce spending below the reconciliation target price, they could have an incentive to avoid complex, high-cost cases by referring them to nearby facilities or specialty referral centers. We intend to monitor the claims data from TEAM participants—for example, to compare a hospital's case mix relative to a pre-model historical baseline to determine whether complex patients are being systematically excluded. Furthermore, we are requiring TEAM participants to supply beneficiaries with written information regarding the hospital's participation in TEAM as well as their rights under Medicare, including their right to use their provider of choice.
We will implement safeguards to ensure that Medicare beneficiaries do not experience a delay in services. Specifically, to avoid perverse incentives to withhold or delay medically necessary care until after an episode ends, TEAM participants will remain responsible for episode spending in the 30-day period following completion of each episode for all services covered under Medicare Parts A and B, regardless of whether the services are included in the episode definition.
Importantly, approaches to savings will include taking steps that facilitate patient recovery, shorten recovery duration, and minimize post-operative problems that might lead to readmissions. Thus, the model itself rewards better patient care.
Lastly, we note that TEAM will not change Medicare FFS payments, beneficiary copayments, deductibles, or coinsurance. Beneficiaries may benefit if TEAM participants are able to systematically improve the quality of care while reducing costs. We welcomed but did not receive public comments on our estimates of the impact of TEAM on Medicare beneficiaries.
There may be spillover effects in the non-Medicare market, or even in the Medicare market in other areas as a result of this model. Testing changes in Medicare payment policy may have implications for non-Medicare payers. As an example, non-Medicare patients may benefit if participating hospitals introduce system-wide changes that improve the coordination and quality of health care. Other payers may also be developing payment models and may align their payment structures with CMS or may be waiting to utilize results from CMS' evaluations of payment models. Because it is unclear whether and how this evidence applies to a test of these new payment models, our analyses assume that spillover effects on non-Medicare payers will not occur, although this assumption is subject to considerable uncertainty. We welcomed comments on this assumption and evidence on how this rulemaking, would impact non-Medicare payers and patients but did not receive any comments.
In section X.B. of the preamble of this final rule, we finalized proposed changes to 42 CFR 405.1845 to permit individuals to serve one additional consecutive term as PRRB Members, relative to the current regulations, which allow two consecutive 3-year terms (6 consecutive years). Based on historical experience, PRRB Members generally serve 6 consecutive years as permitted by the current regulations; under the final rule, a PRRB Member would be eligible to serve for 9 years. We anticipate achieving productivity gains and greater efficiencies from retaining experienced Board Members over a longer period, particularly since Board Members spend a portion of their initial term acclimating to the adjudicatory responsibilities and deepening their expertise in the wide scope of specialized matters that come before the Board. Accordingly, under the policy we are adopting in the final rule, we anticipate that a Board Member would be in a better position to efficiently address increasingly complex and technical issues and a higher volume of cases as they gain additional seniority. Furthermore, the possibility of having a 9-year tenure on the PRRB might make the position more attractive to prospective applicants, thereby increasing the size and quality of the candidate pool. We believe for example that otherwise qualified individuals might refrain from applying, knowing that the position is limited to no more than 6 years. Therefore, this policy will result in a no cost impact relative to the requirements of Executive Orders 12866, 13563, and 14094. There may be negligible government savings attributable to reducing human resource-related costs such as recruitment and hiring activity. We ( print page 70029) received no comments on this aspect of our proposal and are finalizing this provision without modification.
In section X.D. of the preamble of this final rule, we discuss in detail the changes to the administration of the existing PERM program. The Further Consolidated Appropriations Act of 2020 ( Pub. L. 116-94 ) required Puerto Rico to publish a plan, developed in coordination with CMS, and approved by the CMS Administrator, not later than 18 months after the FCAA's enactment, for how Puerto Rico would develop measures to comply with the PERM requirements of 42 CFR part 431, subpart Q . Currently, Puerto Rico is excluded from PERM via regulation at 42 CFR 431.954(b)(3) . Puerto Rico will be incorporated into the PERM program starting in reporting year 2027 (Cycle 3), which covers the payment period between July 1, 2025 through June 30, 2026.
In the proposed rule, we noted that including Puerto Rico in the PERM program will increase transparency into its Medicaid and CHIP operations and should improve program integrity efforts that protect taxpayer dollars from improper payments. A state [ 1131 ] in the PERM program will be reviewed only once every 3 years and it is not likely that a provider would be selected more than once per program cycle to provide supporting documentation, minimizing the annual burden on both the state and its providers. Therefore, we estimate the cost to Puerto Rico for participating in the PERM program will be approximately $3.5 million annually. More detail about the cost and burden hours associated with response to requests for information (approximately 6,000 hours annually) can be found in the program PRA package (CMS—10166, CMS—10178, CMS—10184). Therefore, we did not anticipate this to be a significant administrative cost.
We did not prepare an analysis for this policy under the Regulatory Flexibility Act (RFA) because we determined that the policy will not have a significant impact on a substantial number of small entities.
We did not prepare an analysis for section 1102(b) of the Act because we determined that this policy will not have a significant impact on the operations of a substantial number of small rural hospitals.
Section 202 of the Unfunded Mandates Reform Act of 1995 also requires that agencies assess anticipated costs and benefits before issuing any rule that may result in expenditure in any one year of $100 million in 1995 dollars, updated annually for inflation. That threshold level is currently approximately $183 million. This policy will not result in an impact of $183 million or more on State, local or tribal governments, in the aggregate, or on the private sector.
Executive Order 13132 establishes certain requirements that an agency must meet when it promulgates a proposed rule (and subsequent final rule) that imposes substantial direct requirement costs on State and local governments, preempts State law, or otherwise has Federalism implications. Because this policy does not impose substantial costs on State or local governments, the requirements of Executive Order 13132 are not applicable.
In section X.F. of the preamble of this final rule, we discuss our finalized requirements related to the reporting of acute respiratory illnesses that will have potentially major public health benefits. Routine reporting of these illnesses absent any new emergency makes it possible to use the data to determine which hospitals faced unusually high or low reported levels of such illnesses. Such comparisons will allow individual hospitals, individual cities or states, or the federal government, to analyze outlier hospitals (either high or low rates of acute respiratory infections) to determine if there were any local factors that might suggest some form of intervention will be beneficial to redress problems or to export successes among the universe of hospitals and CAHs. For example, if hospitals in a particular geographic area were finding an unusually high rate of these illnesses among admitted patients from a particular geographic area, investigation of potential causes might lead to improvements in that area's immunization outreach efforts. It will not take many such interventions to have potentially substantial life-saving effects. In the hopefully unlikely case where an outbreak of acute respiratory illness was so substantial as to require the declaration of a public health emergency, the life-saving benefits could be high. For example, an “early warning” signal could speed the development of a vaccine, effective use of particular medicines for treatments, or other interventions to prevent or ameliorate adverse outcomes ranging from a single instance of illness to a national epidemic.
We received no public comments on our estimates.
As of July 2024, there were 92 children's hospitals, 11 cancer hospitals, 6 short term acute care hospitals located in the Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa, 1 extended neoplastic disease care hospital, and 11 RNHCIs being paid on a reasonable cost basis subject to the rate-of-increase ceiling under § 413.40. (In accordance with § 403.752(a) of the regulation, RNHCIs are paid under § 413.40.) Among the remaining providers, the rehabilitation hospitals and units, and the LTCHs, are paid the Federal prospective per discharge rate under the IRF PPS and the LTCH PPS, respectively, and the psychiatric hospitals and units are paid the Federal per diem amount under the IPF PPS. As stated previously, IRFs and IPFs are not affected by the rate updates discussed in this final rule. The impacts of the changes on LTCHs are discussed in section I.J. of this appendix.
For the children's hospitals, cancer hospitals, short-term acute care hospitals located in the Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa, the extended neoplastic disease care hospital, and RNHCIs, the update of the rate-of-increase limit (or target amount) is the estimated FY 2025 percentage increase in the 2018-based IPPS operating market basket, consistent with section 1886(b)(3)(B)(ii) of the Act, and §§ 403.752(a) and 413.40 of the regulations. Consistent with current law, based on IGI's second quarter 2024 forecast of the 2018-based IPPS market basket increase, we are estimating the FY 2025 update to be 3.4 percent (that is, the estimate of the market basket rate-of-increase), as discussed in section V.A. of the preamble of this final rule. We proposed that if more recent data became available for the final rule, we would use such data, if appropriate, to calculate the final IPPS operating market basket update for FY 2025. The Affordable Care Act requires a productivity adjustment (0.5 percentage point reduction for FY 2025), resulting in a 2.9 percent applicable percentage increase for IPPS hospitals that submit quality data and are meaningful EHR users, as discussed in section V.B. of the preamble of this final rule. Children's hospitals, cancer hospitals, short term acute care hospitals located in the Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa, the extended neoplastic disease care hospital, and RNHCIs that continue to be paid based on reasonable costs subject to rate-of-increase limits under § 413.40 of the regulations are not subject to the reductions in the applicable percentage increase required under the Affordable Care Act. Therefore, for those hospitals paid under § 413.40 of the regulations, the update is the percentage increase in the 2018-based IPPS operating market basket for FY 2025, currently estimated at 3.4 percent.
The impact of the update in the rate-of-increase limit on those excluded hospitals depends on the cumulative cost increases experienced by each excluded hospital since its applicable base period. For excluded hospitals that have maintained their cost increases at a level below the rate-of-increase limits since their base period, the major effect is on the level of incentive payments these excluded hospitals receive. Conversely, for excluded hospitals with cost increases above the cumulative update in their rate-of-increase limits, the major effect is the amount of excess costs that would not be paid.
We note that, under § 413.40(d)(3), an excluded hospital that continues to be paid under the TEFRA system and whose costs exceed 110 percent of its rate-of-increase limit receives its rate-of-increase limit plus the lesser of: (1) 50 percent of its reasonable costs in excess of 110 percent of the limit; or (2) 10 percent of its limit. In addition, under the various provisions set forth in § 413.40, ( print page 70030) hospitals can obtain payment adjustments for justifiable increases in operating costs that exceed the limit.
For the impact analysis presented in this section of this final rule, we used data from the March 2024 update of the FY 2023 MedPAR file and the March 2024 update of the Provider-Specific File (PSF) that was used for payment purposes. Although the analyses of the changes to the capital prospective payment system do not incorporate cost data, we used the March 2024 update of the most recently available hospital cost report data to categorize hospitals. Our analysis has several qualifications and uses the best data available, as described later in this section of this final rule.
Due to the interdependent nature of the IPPS, it is very difficult to precisely quantify the impact associated with each change. In addition, we draw upon various sources for the data used to categorize hospitals in the tables. In some cases (for instance, the number of beds), there is a fair degree of variation in the data from different sources. We have attempted to construct these variables with the best available sources overall. However, it is possible that some individual hospitals are placed in the wrong category.
Using cases from the March 2024 update of the FY 2023 MedPAR file, we simulated payments under the capital IPPS for FY 2024 and the payments for FY 2025 for a comparison of total payments per case. Short-term, acute care hospitals not paid under the general IPPS (for example, hospitals in Maryland) are excluded from the simulations.
The methodology for determining a capital IPPS payment is set forth at § 412.312. The basic methodology for calculating the capital IPPS payments in FY 2025 is as follows:
(Standard Federal rate) × (DRG weight) × (GAF) × (COLA for hospitals located in Alaska and Hawaii) × (1 + DSH adjustment factor + IME adjustment factor, if applicable).
In addition to the other adjustments, hospitals may receive outlier payments for those cases that qualify under the threshold established for each fiscal year. We modeled payments for each hospital by multiplying the capital Federal rate by the geographic adjustment factor (GAF) and the hospital's case-mix. Then we added estimated payments for indirect medical education, disproportionate share, and outliers, if applicable. For purposes of this impact analysis, the model includes the following assumptions:
- The capital Federal rate was updated, beginning in FY 1996, by an analytical framework that considers changes in the prices associated with capital-related costs and adjustments to account for forecast error, changes in the case-mix index, allowable changes in intensity, and other factors. As discussed in section III.A.1. of the Addendum to this final rule, the update to the capital Federal rate is 3.1 percent for FY 2025.
- In addition to the FY 2025 update factor, the FY 2025 capital Federal rate was calculated based on a GAF/DRG budget neutrality adjustment factor of 0.9856, a budget neutrality factor for the lowest quartile hospital wage index adjustment and the 5-percent cap on wage index decreases policy of 0.9958, and an outlier adjustment factor of 0.9577.
We used the payment simulation model previously described in section I.I. of Appendix A of this final rule to estimate the potential impact of the changes for FY 2025 on total capital payments per case, using a universe of 3,082 hospitals. As previously described, the individual hospital payment parameters are taken from the best available data, including the March2024 update of the FY 2023 MedPAR file, the March 2024 update to the PSF, and the most recent available cost report data from the March 2024 update of HCRIS. In Table III, we present a comparison of estimated total payments per case for FY 2024 and estimated total payments per case for FY 2025 based on the FY 2025 payment policies. Column 2 shows estimates of payments per case under our model for FY 2024. Column 3 shows estimates of payments per case under our model for FY 2025. Column 4 shows the total percentage change in payments from FY 2024 to FY 2025. The change represented in Column 4 includes the 3.1 percent update to the capital Federal rate and other changes in the adjustments to the capital Federal rate. The comparisons are provided by: (1) geographic location; (2) region; and (3) payment classification.
The simulation results show that, on average, capital payments per case in FY 2025 are expected to increase 2.8 percent compared to capital payments per case in FY 2024. This expected increase is primarily due to the 3.1 percent update to the capital Federal rate being partially offset by an expected decrease in capital outlier payments. In general, regional variations in estimated capital payments per case in FY 2025 as compared to capital payments per case in FY 2024 are primarily due to the changes in GAFs, and are generally consistent with the projected changes in payments due to the changes in the wage index (and policies affecting the wage index), as shown in Table I in section I.F. of this appendix.
The net impact of these changes is an estimated 2.8 percent increase in capital payments per case from FY 2024 to FY 2025 for all hospitals (as shown in Table III). The geographic comparison shows that, on average, hospitals in both urban and rural classifications would experience an increase in capital IPPS payments per case in FY 2025 as compared to FY 2024. Capital IPPS payments per case would increase by an estimated 2.7 percent for hospitals in urban areas while payments to hospitals in rural areas would increase by 3.8 percent from FY 2024 to FY 2025. The primary factor contributing to the difference in the projected increase in capital IPPS payments per case for rural hospitals as compared to urban hospitals is the estimated increase in capital payments to rural hospitals due to the effect of changes in the GAFs.
The comparisons by region show that the change in capital payments per case from FY 2024 to FY 2025 for urban areas range from a 0.1 percent decrease for the Pacific urban region to a 5.0 percent increase for the East South Central and East North Central urban regions. Meanwhile, the change in capital payments per case from FY 2024 to FY 2025 for rural areas range from a 0.3 percent decrease for the Pacific rural region to a 6.0 percent increase for the East North Central rural region. Capital IPPS payments per case for hospitals located in Puerto Rico are projected to increase by an estimated 2.2 percent. These regional differences are primarily due to the changes in the GAFs.
The comparison by hospital type of ownership (Voluntary, Proprietary, and Government) shows that proprietary hospitals are expected to experience an increase in capital payments per case from FY 2024 to FY 2025 of 3.2 percent and government hospitals are expected to experience an increase per case from FY2024 to FY 2025 of 2.3 percent. Meanwhile, voluntary hospitals are expected to experience an increase in capital payments per case from FY 2024 to FY 2025 of 2.8 percent.
Section 1886(d)(10) of the Act established the MGCRB. Hospitals may apply for reclassification for purposes of the wage index for FY 2025. Reclassification for wage index purposes also affects the GAFs because that factor is constructed from the hospital wage index. To present the effects of the hospitals being reclassified as of the publication of this final rule for FY 2025, we show the average capital payments per case for reclassified hospitals for FY 2025. Urban reclassified hospitals are expected to experience an increase in capital payments of 3.0 percent; urban nonreclassified hospitals are expected to experience an increase in capital payments of 2.3 percent. Rural reclassified hospitals are expected to experience an increase in capital payments of 4.1 percent; rural nonreclassified hospitals are expected to experience an increase in capital payments of 3.3 percent. The higher expected increase in payments for rural reclassified hospitals compared to rural nonreclassified hospitals is primarily due to the changes in the GAFs.

In section VIII. of the preamble of this final rule and section V. of the Addendum to this final rule, we set forth the annual update to the payment rates for the LTCH PPS for FY 2025. In the preamble of this final rule, we specify the statutory authority for the provisions that are presented, identify the policies for FY 2025, and present rationales for our provisions as well as alternatives that were considered. In this section, we discuss the impact of the changes to the payment rate, factors, and other payment rate policies related to the LTCH PPS that are presented in the preamble of this final rule in terms of their estimated fiscal impact on the Medicare budget and on LTCHs.
There are 331 LTCHs included in this impact analysis. We note that, although there are currently approximately 339 LTCHs, for purposes of this impact analysis, we excluded the data of all-inclusive rate providers consistent with the development of the FY 2025 MS-LTC-DRG relative weights (discussed in section VIII.B.3. of the preamble of this final rule). We have also excluded data for CCN 312024 from this impact analysis due to their abnormal charging practices. We note this is consistent with our removal of this LTCH from the calculation of the FY 2025 MS-LTC-DRG relative weights, the area wage level adjustment budget neutrality factor, and the fixed-loss amount for LTCH PPS standard Federal payment rate cases (discussed in section VIII.B.3. of the preamble of this final rule). Moreover, another LTCH, only had one claim in the claims data used for this final rule. Because the number of covered days of care that are chargeable to Medicare utilization for the stay was reported as 0 on this claim, we excluded this claim and LTCH from our impact analysis. Lastly, in the claims data used for this final rule, one of the 331 LTCHs included in our impact analysis only had claims for site neutral payment rate cases and, therefore, does not affect our impact analysis for LTCH PPS standard Federal payment rate cases presented in Table IV (that is, the impact analysis presented in Table IV is based on the data for 330 LTCHs).
In the impact analysis, we used the payment rate, factors, and policies presented in this final rule, the 3.0 percent annual update to the LTCH PPS standard Federal payment rate, the update to the MS-LTC-DRG classifications and relative weights, the update to the wage index values (including the update to the CBSA labor market areas) and labor-related share, and the best available claims and CCR data to estimate the change in payments for FY 2025.
Under the dual rate LTCH PPS payment structure, payment for LTCH discharges that meet the criteria for exclusion from the site neutral payment rate (that is, LTCH PPS standard Federal payment rate cases) is based on the LTCH PPS standard Federal payment rate. Consistent with the statute, the site neutral payment rate is the lower of the IPPS comparable per diem amount as determined under § 412.529(d)(4), including any applicable outlier payments as specified in § 412.525(a), reduced by 4.6 percent for FYs 2018 through 2026; or 100 percent of the estimated cost of the case as determined under § 412.529(d)(2). In addition, there are two separate high cost outlier targets—one for LTCH PPS standard Federal payment rate cases and one for site neutral payment rate cases.
Based on the best available data for the 331 LTCHs in our database that were considered in the analyses used for this final rule, we estimate that overall LTCH PPS payments in FY 2025 will increase by approximately 2.2 percent (or approximately $58 million) based on the rates and factors presented in section VIII. of the preamble and section V. of the Addendum to this final rule.
Based on the FY 2023 LTCH cases that were used for the analysis in this final rule, approximately 29 percent of those cases were classified as site neutral payment rate cases (that is, 29 percent of LTCH cases would not meet the statutory patient-level criteria for exclusion from the site neutral payment rate). We note that section 3711(b)(2) of the CARES Act provided a waiver of the application of the site neutral payment rate for LTCH cases admitted during the COVID-19 PHE period. The COVID-19 PHE expired on May 11, 2023. Therefore, all LTCH PPS cases in FY ( print page 70033) 2023 with admission dates on or before the PHE expiration date were paid the LTCH PPS standard Federal rate regardless of whether the discharge met the statutory patient criteria. Because not all FY 2023 cases were subject to the site neutral payment rate, for purposes of this impact analysis, we continue to rely on the same considerations and actuarial projections used in FYs 2016 through 2024. Our Office of the Actuary currently estimates that the percent of LTCH PPS cases that will be classified as site neutral payment rate cases in FY 2025 will not change significantly from the most recent historical data. To estimate FY 2025 LTCH PPS payments for site neutral payment rate cases, we calculated the IPPS comparable per diem amounts using the FY 2025 IPPS rates and factors along with other changes that will apply to the site neutral payment rate cases in FY 2025. We estimate that aggregate LTCH PPS payments for these site neutral payment rate cases will increase by approximately 4.2 percent (or approximately $13 million). This projected increase in payments to LTCH PPS site neutral payment rate cases is primarily due to the finalized updates to the IPPS rates and factors reflected in our estimate of the IPPS comparable per diem amount, as well as an increase in estimated costs for these cases determined using the charge and CCR adjustment factors described in section V.D.3.b. of the Addendum to this final rule. We note that we estimate payments to site neutral payment rate cases in FY 2025 will represent approximately 12 percent of estimated aggregate FY 2025 LTCH PPS payments.
Based on the FY 2023 LTCH cases that were used for the analysis in this final rule, approximately 71 percent of LTCH cases will meet the patient-level criteria for exclusion from the site neutral payment rate in FY 2025, and will be paid based on the LTCH PPS standard Federal payment rate. We estimate that total LTCH PPS payments for these LTCH PPS standard Federal payment rate cases in FY 2025 will increase approximately 2.0 percent (or approximately $45 million). This estimated increase in LTCH PPS payments for LTCH PPS standard Federal payment rate cases in FY 2025 is primarily due to the 3.0 percent annual update to the LTCH PPS standard Federal payment rate being partially offset by a projected 0.8 percentage point decrease in high cost outlier payments as a percentage of total LTCH PPS standard Federal payment rate payments, which is discussed later in this section.
Based on the 331 LTCHs that were represented in the FY 2023 LTCH cases that were used for the analyses in this final rule presented in this appendix, we estimate that aggregate FY 2024 LTCH PPS payments will be approximately $2.581 billion, as compared to estimated aggregate FY 2025 LTCH PPS payments of approximately $2.638 billion, resulting in an estimated overall increase in LTCH PPS payments of approximately $58 million. We note that the estimated $58 million increase in LTCH PPS payments in FY 2025 does not reflect changes in LTCH admissions or case-mix intensity, which will also affect the overall payment effects of the policies in this final rule.
The LTCH PPS standard Federal payment rate for FY 2024 is $48,116.62. For FY 2025, we are establishing an LTCH PPS standard Federal payment rate of $49,383.26 which reflects the 3.0 percent annual update to the LTCH PPS standard Federal payment rate and the budget neutrality factor for updates to the area wage level adjustment of 0.9964315 (discussed in section V.B.6. of the Addendum to this final rule). For LTCHs that fail to submit data for the LTCH QRP, in accordance with section 1886(m)(5)(C) of the Act, we are establishing an LTCH PPS standard Federal payment rate of $48,424.36. This LTCH PPS standard Federal payment rate reflects the updates and factors previously described, as well as the required 2.0 percentage point reduction to the annual update for failure to submit data under the LTCH QRP.
Table IV shows the estimated impact for LTCH PPS standard Federal payment rate cases. The estimated change attributable solely to the annual update of 3.0 percent to the LTCH PPS standard Federal payment rate is projected to result in an increase of 2.9 percent in payments per discharge for LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025, on average, for all LTCHs (Column 6). The estimated increase of 2.9 percent shown in Column 6 of Table IV also includes estimated payments for short-stay outlier (SSO) cases, a portion of which are not affected by the annual update to the LTCH PPS standard Federal payment rate, as well as the reduction that is applied to the annual update for LTCHs that do not submit the required LTCH QRP data. For most hospital categories, the projected increase in payments based on the LTCH PPS standard Federal payment rate to LTCH PPS standard Federal payment rate cases also rounds to approximately 2.9 percent.
For FY 2025, we are updating the wage index values based on the most recent available data (data from cost reporting periods beginning during FY 2021 which is the same data used for the FY 2025 IPPS wage index) and the revised CBSA labor market areas delineations that we are adopting (as discussed in section V.B.2. of the Addendum to this final rule). In addition, we are establishing a labor-related share of 72.8 percent for FY 2025, based on the most recent available data (IGI's second quarter 2024 forecast) of the relative importance of the labor-related share of operating and capital costs of the 2022-based LTCH market basket. We are also applying an area wage level budget neutrality factor of 0.9964315 to ensure that the changes to the area wage level adjustment will not result in any change in estimated aggregate LTCH PPS payments to LTCH PPS standard Federal payment rate cases.
For LTCH PPS standard Federal payment rate cases, we currently estimate high-cost outlier payments as a percentage of total LTCH PPS standard Federal payment rate payments will decrease from FY 2024 to FY 2025. Based on the FY 2023 LTCH cases that were used for the analyses in this final rule, we estimate that the FY 2024 high-cost outlier threshold of $59,873 (as established in the FY 2024 IPPS/LTCH PPS final rule) will result in estimated high cost outlier payments for LTCH PPS standard Federal payment rate cases in FY 2024 that are projected to exceed the 7.975 percent target. Specifically, we currently estimate that high-cost outlier payments for LTCH PPS standard Federal payment rate cases will be approximately 8.8 percent of the estimated total LTCH PPS standard Federal payment rate payments in FY 2024. Combined with our estimate that FY 2025 high-cost outlier payments for LTCH PPS standard Federal payment rate cases will be 7.975 percent of estimated total LTCH PPS standard Federal payment rate payments in FY 2025, this will result in an estimated decrease in high cost outlier payments as a percentage of total LTCH PPS standard Federal payment rate payments of approximately 0.8 percentage point between FY 2024 and FY 2025. We note that, in calculating these estimated high cost outlier payments, we inflated charges reported on the FY 2023 claims by the charge inflation factor described in section V.D.3.b. of the Addendum to this final rule. We also note that, in calculating these estimated high-cost outlier payments, we estimated the cost of each case by multiplying the inflated charges by the adjusted CCRs that we determined using our finalized methodology described in section V.D.3.b. of the Addendum to this final rule.
Table IV shows the estimated impact of the payment rate and policy changes on LTCH PPS payments for LTCH PPS standard Federal payment rate cases for FY 2025 by comparing estimated FY 2024 LTCH PPS payments to estimated FY 2025 LTCH PPS payments. (As noted earlier, our analysis does not reflect changes in LTCH admissions or case-mix intensity.) We note that these impacts do not include LTCH PPS site neutral payment rate cases as discussed in section I.J.3. of this appendix.
Comment: We received comments expressing concerns about the adequacy of the 1.2 percent increase in payments to LTCH PPS standard Federal payment rate cases that we projected in the proposed rule. These comments primarily focused on the impact that the proposed annual update to the LTCH PPS standard Federal payment rate and the proposed fixed-loss amount for high-cost outlier cases would have on payments to LTCH PPS standard Federal payment rate cases in FY 2025.
Response: We appreciate commenters' concerns about the proposed 1.2 percent increase in payment to LTCH PPS standard Federal payment rate cases. As explained in the proposed rule ( 89 FR 36635 ), that estimated increase of approximately 1.2 percent was primarily due to the proposed 2.8 percent annual update to the LTCH PPS standard Federal payment rate being partially offset by a projected 1.3 percent decrease in high-cost outlier payments as a percentage of total LTCH PPS standard Federal payment rate payment. We received several comments on the proposed annual update to the LTCH PPS standard Federal payment rate which we have summarized and responded to in sections VIII.C. and VIII.D. of the preamble to this final rule. We also received several comments on the proposed fixed-loss amount for high-cost outlier cases which we have summarized and responded to in section V.D.3. of the Addendum to this final rule. ( print page 70034)
Based on the finalized payment rates and factors in this final rule, we now project a 2.0 percent increase in payments to LTCH PPS standard Federal payment rate cases for FY 2025 (as compared to our projection of 1.2 percent in the proposed rule). This increase in our projected percentage change in payments is partially being driven by the upward revision in this final rule to the annual update for FY 2025 in the proposed rule. The final annual update factor of 3.0 percent is 0.2 percentage point higher than the proposed annual update factor. As discussed in section VIII.C.2. of the preamble to this final rule, we believe this LTCH market basket increase appropriately reflects the input price growth that LTCHs will incur providing medical services in FY 2025. The increase in our projected percentage change in payments is also partially being driven by a downward revision in this final rule to our estimate of FY 2024 high cost outlier payments to LTCH PPS standard Federal payment rate cases. In this final rule, after incorporating into our payment model more recent data, as discussed in section V.D.3. of the Addendum to this final rule, we now estimate that high cost outlier payments for LTCH PPS standard Federal payment rate cases will be approximately 8.8 percent of the estimated total LTCH PPS standard Federal payment rate payments in FY 2024 (as compared to our estimate of 9.3 percent in the proposed rule). The reduction in our estimate of the FY 2024 outlier payment percentage has the effect of increasing our projected percent change in payments from FY 2024 to FY 2025.
As we discuss in detail throughout this final rule, based on the best available data, we believe that the provisions of this final rule relating to the LTCH PPS, which are projected to result in an overall increase in estimated aggregate LTCH PPS payments (for both LTCH PPS standard Federal payment rate cases and site neutral payment rate cases), and the resulting LTCH PPS payment amounts will result in appropriate Medicare payments that are consistent with the statute.
For purposes of section 1102(b) of the Act, we define a small rural hospital as a hospital that is located outside of an urban area and has fewer than 100 beds. As shown in Table IV, we are projecting a 2.8 percent increase in estimated payments for LTCH PPS standard Federal payment rate cases for LTCHs located in a rural area. This increase is primarily due to the combination of the 3.0 percent annual update to the LTCH PPS standard Federal payment rate for FY 2025, the changes to the area wage level adjustment, and estimated changes in outlier payments. This estimated impact is based on the FY 2023 data for the 19 rural LTCHs (out of 330 LTCHs) that were used for the impact analyses shown in Table IV.
Section 123(a)(1) of the BBRA requires that the PPS developed for LTCHs “maintain budget neutrality.” We believe that the statute's mandate for budget neutrality applies only to the first year of the implementation of the LTCH PPS (that is, FY 2003). Therefore, in calculating the FY 2003 standard Federal payment rate under § 412.523(d)(2), we set total estimated payments for FY 2003 under the LTCH PPS so that estimated aggregate payments under the LTCH PPS were estimated to equal the amount that would have been paid if the LTCH PPS had not been implemented.
Section 1886(m)(6)(A) of the Act establishes a dual rate LTCH PPS payment structure with two distinct payment rates for LTCH discharges beginning in FY 2016. Under this statutory change, LTCH discharges that meet the patient-level criteria for exclusion from the site neutral payment rate (that is, LTCH PPS standard Federal payment rate cases) are paid based on the LTCH PPS standard Federal payment rate. LTCH discharges paid at the site neutral payment rate are generally paid the lower of the IPPS comparable per diem amount, reduced by 4.6 percent for FYs 2018 through 2026, including any applicable high cost outlier (HCO) payments, or 100 percent of the estimated cost of the case, reduced by 4.6 percent.
As discussed in section I.J.1. of this appendix, we project an increase in aggregate LTCH PPS payments in FY 2025 of approximately $58 million. This estimated increase in payments reflects the projected increase in payments to LTCH PPS standard Federal payment rate cases of approximately $45 million and the projected increase in payments to site neutral payment rate cases of approximately $13 million under the dual rate LTCH PPS payment rate structure required by the statute beginning in FY 2016.
As discussed in section V.D. of the Addendum to this final rule, our actuaries project cost and resource changes for site neutral payment rate cases due to the site neutral payment rates required under the statute. Specifically, our actuaries project that the costs and resource use for cases paid at the site neutral payment rate will likely be lower, on average, than the costs and resource use for cases paid at the LTCH PPS standard Federal payment rate, and will likely mirror the costs and resource use for IPPS cases assigned to the same MS-DRG. While we are able to incorporate this projection at an aggregate level into our payment modeling, because the historical claims data that we are using in this final rule to project estimated FY 2025 LTCH PPS payments (that is, FY 2023 LTCH claims data) do not reflect this actuarial projection, we are unable to model the impact of the change in LTCH PPS payments for site neutral payment rate cases at the same level of detail with which we are able to model the impacts of the changes to LTCH PPS payments for LTCH PPS standard Federal payment rate cases. Therefore, Table IV only reflects changes in LTCH PPS payments for LTCH PPS standard Federal payment rate cases and, unless otherwise noted, the remaining discussion in section I.J.3. of this appendix refers only to the impact on LTCH PPS payments for LTCH PPS standard Federal payment rate cases. In the following section, we present our provider impact analysis for the changes that affect LTCH PPS payments for LTCH PPS standard Federal payment rate cases.
The basic methodology for determining a per discharge payment for LTCH PPS standard Federal payment rate cases is currently set forth under §§ 412.515 through 412.533 and 412.535. In addition to adjusting the LTCH PPS standard Federal payment rate by the MS-LTC-DRG relative weight, we make adjustments to account for area wage levels and SSOs. LTCHs located in Alaska and Hawaii also have their payments adjusted by a COLA. Under our application of the dual rate LTCH PPS payment structure, the LTCH PPS standard Federal payment rate is generally only used to determine payments for LTCH PPS standard Federal payment rate cases (that is, those LTCH PPS cases that meet the statutory criteria to be excluded from the site neutral payment rate). LTCH discharges that do not meet the patient-level criteria for exclusion are paid the site neutral payment rate, which we are calculating as the lower of the IPPS comparable per diem amount as determined under § 412.529(d)(4), reduced by 4.6 percent for FYs 2018 through 2026, including any applicable outlier payments, or 100 percent of the estimated cost of the case as determined under existing § 412.529(d)(2). In addition, when certain thresholds are met, LTCHs also receive HCO payments for both LTCH PPS standard Federal payment rate cases and site neutral payment rate cases that are paid at the IPPS comparable per diem amount.
To understand the impact of the changes to the LTCH PPS payments for LTCH PPS standard Federal payment rate cases presented in this final rule on different categories of LTCHs for FY 2025, it is necessary to estimate payments per discharge for FY 2024 using the rates, factors, and the policies established in the FY 2024 IPPS/LTCH PPS final rule and estimate payments per discharge for FY 2025 using the rates, factors, and the policies in this final rule (as discussed in section VIII. of the preamble of this final rule and section V. of the Addendum to this final rule). As discussed elsewhere in this final rule, these estimates are based on the best available LTCH claims data and other factors, such as the application of inflation factors to estimate costs for HCO cases in each year. The resulting analyses can then be used to compare how our policies applicable to LTCH PPS standard Federal payment rate cases affect different groups of LTCHs.
For the following analysis, we group hospitals based on characteristics provided in the OSCAR data, cost report data in HCRIS, and PSF data. Hospital groups included the following:
- Location: large urban/other urban/rural.
- Participation date.
- Ownership control.
- Census region.
For purposes of this impact analysis, to estimate the per discharge payment effects of our policies on payments for LTCH PPS ( print page 70035) standard Federal payment rate cases, we simulated FY 2024 and FY 2025 payments on a case-by-case basis using historical LTCH claims from the FY 2023 MedPAR files that met or would have met the criteria to be paid at the LTCH PPS standard Federal payment rate if the statutory patient-level criteria had been in effect at the time of discharge for all cases in the FY 2023 MedPAR files. For modeling FY 2024 LTCH PPS payments, we used the FY 2024 standard Federal payment rate of $48,116.62 (or $47,185.03 for LTCHs that failed to submit quality data as required under the requirements of the LTCH QRP). Similarly, for modeling payments based on the FY 2025 LTCH PPS standard Federal payment rate, we used the FY 2025 standard Federal payment rate of $49,383.26 (or $48,424.36 for LTCHs that failed to submit quality data as required under the requirements of the LTCH QRP). In each case, we applied the applicable adjustments for area wage levels and the COLA for LTCHs located in Alaska and Hawaii. Specifically, for modeling FY 2024 LTCH PPS payments, we used the current FY 2024 labor-related share (68.5 percent), the wage index values established in the Tables 12A and 12B listed in the Addendum to the FY 2024 IPPS/LTCH PPS final rule (which are available via the internet on the CMS website), the FY 2024 HCO fixed-loss amount for LTCH PPS standard Federal payment rate cases of $59,873 (as reflected in the FY 2024 IPPS/LTCH PPS final rule), and the FY 2024 COLA factors (shown in the table in section V.C. of the Addendum to that final rule) to adjust the FY 2024 nonlabor-related share (31.5 percent) for LTCHs located in Alaska and Hawaii. Similarly, for modeling FY 2025 LTCH PPS payments, we used the FY 2025 LTCH PPS labor-related share (72.8 percent), the FY 2025 wage index values from Tables 12A and 12B listed in section VI. of the Addendum to this final rule (which are available via the internet on the CMS website), the FY 2025 HCO fixed-loss amount for LTCH PPS standard Federal payment rate cases of $77,048 (as discussed in section V.D.3. of the Addendum to this final rule), and the FY 2025 COLA factors (shown in the table in section V.C. of the Addendum to this final rule) to adjust the FY 2025 nonlabor-related share (27.2 percent) for LTCHs located in Alaska and Hawaii. We note that in modeling payments for HCO cases for LTCH PPS standard Federal payment rate cases, we inflated charges reported on the FY 2023 claims by the charge inflation factors in section V.D.3.b. of the Addendum to this final rule. We also note that in modeling payments for HCO cases for LTCH PPS standard Federal payment rate cases, we estimated the cost of each case by multiplying the inflated charges by the adjusted CCRs that we determined using our finalized methodology described in section V.D.3.b. of the Addendum to this final rule.
The impacts that follow reflect the estimated “losses” or “gains” among the various classifications of LTCHs from FY 2024 to FY 2025 based on the payment rates and policy changes applicable to LTCH PPS standard Federal payment rate cases presented in this final rule. Table IV illustrates the estimated aggregate impact of the change in LTCH PPS payments for LTCH PPS standard Federal payment rate cases among various classifications of LTCHs. (As discussed previously, these impacts do not include LTCH PPS site neutral payment rate cases.)
- The first column, LTCH Classification, identifies the type of LTCH.
- The second column lists the number of LTCHs of each classification type.
- The third column identifies the number of LTCH cases expected to meet the LTCH PPS standard Federal payment rate criteria.
- The fourth column shows the estimated FY 2024 payment per discharge for LTCH cases expected to meet the LTCH PPS standard Federal payment rate criteria (as described previously).
- The fifth column shows the estimated FY 2025 payment per discharge for LTCH cases expected to meet the LTCH PPS standard Federal payment rate criteria (as described previously).
- The sixth column shows the percentage change in estimated payments per discharge for LTCH cases expected to meet the LTCH PPS standard Federal payment rate criteria from FY 2024 to FY 2025 due to the annual update to the standard Federal rate (as discussed in section V.A.2. of the Addendum to this final rule).
- The seventh column shows the percentage change in estimated payments per discharge for LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025 due to the changes to the area wage level adjustment (that is, the updated hospital wage data, labor market areas, and labor-related share) and the application of the corresponding budget neutrality factor (as discussed in section V.B.6. of the Addendum to this final rule).
- The eighth column shows the percentage change in estimated payments per discharge for LTCH PPS standard Federal payment rate cases from FY 2024 (Column 4) to FY 2025 (Column 5) due to all changes.

Based on the FY 2023 LTCH cases (from 330 LTCHs) that were used for the analyses in this final rule, we have prepared the following summary of the impact (as shown in Table IV) of the LTCH PPS payment rate and policy changes for LTCH PPS standard Federal payment rate cases presented in this final rule. The impact analysis in Table IV shows that estimated payments per discharge for LTCH PPS standard Federal payment rate cases are projected to increase 2.0 percent, on average, for all LTCHs from FY 2024 to FY 2025 as a result of the payment rate and policy changes applicable to LTCH PPS standard Federal payment rate cases presented in this final rule. This estimated 2.0 percent increase in LTCH PPS payments per discharge was determined by comparing estimated FY 2025 LTCH PPS payments (using the finalized payment rates and factors discussed in this final rule) to estimated FY 2024 LTCH PPS payments for LTCH discharges which will be LTCH PPS standard Federal payment rate cases if the dual rate LTCH PPS payment structure was or had been in effect at the time of the discharge (as described in section I.J.3. of this appendix).
As stated previously, we are finalizing an annual update to the LTCH PPS standard Federal payment rate for FY 2025 of 3.0 percent. For LTCHs that fail to submit quality data under the requirements of the LTCH QRP, as required by section 1886(m)(5)(C) of the Act, a 2.0 percentage point reduction is applied to the annual update to the LTCH PPS standard Federal payment rate. Consistent with § 412.523(d)(4), we also are applying a budget neutrality factor for changes to the area wage level adjustment of 0.9964315 (discussed in section V.B.6. of the Addendum to this final rule), based on the best available data at this time, to ensure that any changes to the area wage level adjustment will not result in any change (increase or decrease) in estimated aggregate LTCH PPS standard Federal payment rate payments. As we also explained earlier in this section of the final rule, for most categories of LTCHs (as shown in Table IV, Column 6), the estimated payment increase due to the 3.0 percent annual update to the LTCH PPS standard Federal payment rate is projected to result in approximately a 2.9 percent increase in estimated payments per discharge for LTCH PPS standard Federal payment rate cases for all LTCHs from FY 2024 to FY 2025. We note our estimate of the changes in payments due to the update to the LTCH PPS standard Federal payment rate also includes estimated payments for short-stay outlier (SSO) cases, a portion of which are not affected by the annual update to the LTCH PPS standard Federal payment rate, as well as the reduction that is applied to the annual update for LTCHs that do not submit data under the requirements of the LTCH QRP.
Based on the most recent available data, the vast majority of LTCHs are located in urban areas. Only approximately 6 percent of the LTCHs are identified as being located in a rural area, and approximately 4 percent of all LTCH PPS standard Federal payment rate cases are expected to be treated in these rural hospitals. The impact analysis presented in Table IV shows that the overall average percent increase in estimated payments per discharge for LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025 for all hospitals is 2.0 percent. Urban LTCHs are projected to experience an increase of 1.9 percent. Meanwhile, rural LTCHs are projected to experience an increase of 2.8 percent.
LTCHs are grouped by participation date into four categories: (1) before October 1983; (2) between October 1983 and September 1993; (3) between October 1993 and September 2002; and (4) October 2002 and after. Based on the best available data, the categories of LTCHs with the largest expected percentage of LTCH PPS standard Federal payment rate cases (approximately 41 percent and 45 percent, respectively) are in LTCHs that began participating in the Medicare program between October 1993 and September 2002 and after October 2002. These LTCHs are expected to experience an increase in estimated payments per discharge for LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025 of 2.2 percent and 1.9 percent, respectively. LTCHs that began participating in the Medicare program between October 1983 and September 1993 are projected to experience an increase in estimated payments per discharge for LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025 of 2.0 percent, as shown in Table IV. Approximately 3 percent of LTCHs began participating in the Medicare program before October 1983, and these LTCHs are projected to experience a decrease in estimated payments per discharge for LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025 of 0.3 percent, partially due to the changes to the area wage level adjustment.
LTCHs are grouped into three categories based on ownership control type: voluntary, proprietary, and government. Based on the best available data, approximately 16 percent of LTCHs are identified as voluntary (Table IV). The majority (approximately 81 percent) of LTCHs are identified as proprietary, while government owned and operated LTCHs represent approximately 3 percent of LTCHs. Based on ownership type, proprietary LTCHs are expected to experience an increase in payments to LTCH PPS standard Federal payment rate cases of 2.1 percent. Voluntary LTCHs are expected to experience an increase in payments to LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025 of 1.3 percent. Government owned and operated LTCHs are expected to experience an increase in payments to LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025 of 1.2 percent.
The comparisons by region show that the changes in estimated payments per discharge for LTCH PPS standard Federal payment rate cases from FY 2024 to FY 2025 are projected to range from an increase of 0.4 percent in the New England region to increases of 2.7 percent in both the East South Central region and the Mountain region. These regional variations are primarily due to the changes to the area wage adjustment and estimated changes in outlier payments. ( print page 70038)
LTCHs are grouped into six categories based on bed size: 0-24 beds; 25-49 beds; 50-74 beds; 75-124 beds; 125-199 beds; and greater than 200 beds. We project that LTCHs with 125-199 beds will experience an increase in payments for LTCH PPS standard Federal payment rate cases of 1.0 percent. LTCHs with 25-49 beds are projected to experience the largest increase in payments, 2.5 percent. The remaining bed size categories are projected to experience an increase in payments in the range of 1.4 to 1.9 percent.
As stated previously, we project that the provisions of this final rule will result in an increase in estimated aggregate LTCH PPS payments to LTCH PPS standard Federal payment rate cases in FY 2025 relative to FY 2024 of approximately $45 million (or approximately 2.0 percent) for the 331 LTCHs in our database. Although, as stated previously, the hospital-level impacts do not include LTCH PPS site neutral payment rate cases, we estimate that the provisions of this final rule will result in an increase in estimated aggregate LTCH PPS payments to site neutral payment rate cases in FY 2025 relative to FY 2024 of approximately $13 million (or approximately 4.2 percent) for the 331 LTCHs in our database. (As noted previously, we estimate payments to site neutral payment rate cases in FY 2025 will represent approximately 12 percent of total estimated FY 2025 LTCH PPS payments.) Therefore, we project that the provisions of this final rule will result in an increase in estimated aggregate LTCH PPS payments for all LTCH cases in FY 2025 relative to FY 2024 of approximately $58 million (or approximately 2.2 percent) for the 331 LTCHs in our database.
Under the LTCH PPS, hospitals receive payment based on the average resources consumed by patients for each diagnosis. We do not expect any changes in the quality of care or access to services for Medicare beneficiaries as a result of this final rule, but we continue to expect that paying prospectively for LTCH services will enhance the efficiency of the Medicare program. As discussed previously, we do not expect the continued implementation of the site neutral payment system to have a negative impact on access to or quality of care, as demonstrated in areas where there is little or no LTCH presence, general short-term acute care hospitals are effectively providing treatment for the same types of patients that are treated in LTCHs.
In sections IX.B.1., IX.B.2., and IX.C. of the preamble of this final rule, we discuss the finalized requirements for hospitals reporting quality data under the Hospital IQR Program to receive the full annual percentage increase for the FY 2027 payment determination and subsequent years.
In the preamble of this final rule, we are adopting seven new measures: (1) Age-Friendly Hospital measure beginning with the CY 2025 reporting period/FY 2027 payment determination; (2) Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 payment determination, with modifications; (3) Catheter-Associated Urinary Tract Infection (CAUTI) Standardized Infection Ratio Stratified for Oncology Locations measure beginning with the CY 2026 reporting period/FY 2028 payment determination; (4) Central Line-Associated Bloodstream Infection (CLABSI) Standardized Infection Ratio Stratified for Oncology Locations measure beginning with the CY 2026 reporting period/FY 2028 payment determination; (5) Hospital Harm—Falls with Injury electronic clinical quality measure (eCQM) beginning with the CY 2026 reporting period/FY 2028 payment determination; (6) Hospital Harm—Postoperative Respiratory Failure eCQM beginning with the CY 2026 reporting period/FY 2028 payment determination; and (7) Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue) measure beginning with the July 1, 2023-June 30, 2025 reporting period/FY 2027 payment determination. We are modifying two measures: (1) the Global Malnutrition Composite Score eCQM, beginning with the CY 2026 reporting period/FY 2028 payment determination; and (2) the Hospital Consumer Assessment of Healthcare Providers and Systems (HCAHPS) Survey measure beginning with the CY 2025 reporting period/FY 2027 payment determination. We are also removing five measures: (1) Death Rate Among Surgical Inpatients with Serious Treatable Complications (CMS PSI-04) measure beginning with the July 1, 2023-June 30, 2025 reporting period/FY 2027 payment determination; (2) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Acute Myocardial Infarction (AMI) measure beginning with the July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination; (3) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Heart Failure (HF) measure beginning with the July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination; (4) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Pneumonia (PN) measure beginning with the July 1, 2021-June 30, 2024 reporting period/FY 2026 payment determination; and (5) Hospital-level, Risk-Standardized Payment Associated with a 30-Day Episode-of-Care for Elective Primary Total Hip Arthroplasty (THA) and/or Total Knee Arthroplasty (TKA) measure beginning with the April 1, 2021-March 31, 2024 reporting period/FY 2026 payment determination. We are finalizing a modified version of our proposal to increase the total number of eCQMs that must be reported each year. We are increasing the total number of eCQMs reported from six to eight for the CY 2026 reporting period/FY 2028 payment determination, from eight to nine for the CY 2027 reporting period/FY 2029 payment determination, and then from nine to eleven beginning with the CY 2028 reporting period/FY 2030 payment determination. Lastly, we are updating data validation policies, including updating the scoring methodology for eCQM validation, removing the requirement that hospitals must submit 100 percent of eCQM records to pass validation beginning with CY 2025 eCQM data affecting the FY 2028 payment determination, and no longer requiring hospitals to resubmit medical records as part of their request for reconsideration of validation beginning with CY 2025 discharges affecting the FY 2028 payment determination.
As shown in the summary tables in section XII.B.6.k. of the preamble of this final rule, we estimate a total information collection burden increase of 40,160 hours at a cost of $1,282,329 annually associated with the finalized policies across a 3-year period from the CY 2025 reporting period/FY 2027 payment determination through the CY 2028 reporting period/FY 2030 payment determination, compared to our currently approved information collection burden estimates.
In sections IX.C.5.a. and IX.B.1 of the preamble of this final rule, we are adopting the Age Friendly Hospital and Patient Safety Structural measures. In order for hospitals to receive a point for each of the domains in the measures, affirmative attestations are required for each of the statements within a domain. As noted in the FY 2023 IPPS/LTCH PPS final rule when we finalized the Hospital Commitment to Health Equity measure, hospitals that are unable to attest affirmatively for a statement and desire to improve their measure performance by earning additional points under the measure, will likely have additional costs associated with activities such as updating hospital policies, protocols, or processes; engaging senior leadership; conducting required analyses, surveys, and screenings; performing data analysis and collection; and training staff ( 87 FR 49492 ). The extent of these costs will vary from hospital to hospital depending on what policies the hospital already has in place, what activities the hospital is already performing, hospital size, and the individual choices each hospital makes to meet the criteria necessary to attest affirmatively. There may also be some non-recurring costs associated with changes in workflow and information systems to collect patient screening data if a hospital is not already doing so, however, the extent of these costs is difficult to quantify as different hospitals may utilize different modes of data collection (for example paper-based, electronically patient-directed, clinician-facilitated, etc.).
For the Age Friendly Hospital measure, there may be additional impacts incurred by patients admitted to hospitals that do not currently conduct patient screenings but decide to do so. Hospitals will be able to conduct these screenings via multiple methods, however, we believe most hospitals will likely collect data through a screening tool incorporated into their electronic health record (EHR) or other patient intake process. For the Frailty Screening and Intervention domain, we assume patients will be screened using a combination of validated tools such as the Katz Index of Independence in Activities of Daily Living, the Lawton and Brody Instrumental Activities of Daily-Living ( print page 70039) Scale, the Mini-Cog screening for early dementia, and the Patient Health Questionnaire-2 depression module. [ 1132 1133 1134 1135 1136 ] For the Social Vulnerability domain, we assume patients will be screened using a tool such as the Emergency Department Senior Abuse Identification (ED Senior AID) tool, [ 1137 ] which is currently undergoing validation. We estimate each patient will require no more than 20 minutes (0.33 hours) to complete the screenings for both domains.
We believe that the cost for beneficiaries undertaking administrative and other tasks on their own time is a post-tax wage of $24.04/hr. The Valuing Time in U.S. Department of Health and Human Services Regulatory Impact Analyses: Conceptual Framework and Best Practices identifies the approach for valuing time when individuals undertake activities on their own time. [ 1138 ] To derive the costs for beneficiaries, a measurement of the usual weekly earnings of wage and salary workers of $1,118 was divided by 40 hours to calculate an hourly pre-tax wage rate of $27.95/hr. [ 1139 ] This rate is adjusted downwards by an estimate of the effective tax rate for median income households of about 14 percent calculated by comparing pre- and post-tax income, [ 1140 ] resulting in the post-tax hourly wage rate of $24.04/hr. Unlike our state and private sector wage adjustments, we are not adjusting beneficiary wages for fringe benefits and other indirect costs since the individuals' activities, if any, will occur outside the scope of their employment.
Based on information collected by the Agency for Healthcare Research and Quality for CY 2010 through CY 2019, [ 1141 ] we estimate approximately 7,600,000 patients may be screened annually across all participating IPPS hospitals (12,850,233 average annual admissions of patients aged 65 and over × (3,050 IPPS hospitals ÷ 5,157 total U.S. community hospitals [ 1142 ] )) or an average of 2,492 patients per IPPS hospital. For the CY 2025 reporting period and subsequent years, for each IPPS hospital that elects to perform these screenings, we estimate it will require patients an average of 831 hours (2,492 respondents × 0.33 hours) at a cost of $19,969 (831 hours × $24.04) to complete the screenings.
In sections IX.C.5.c. and IX.C.5.d. of the preamble of this final rule, we are adopting two new eCQMs. As noted in the FY 2022 IPPS/LTCH PPS final rule regarding adoption of eCQMs, while there is no change in information collection burden related to the finalized policies with regard to reporting of measure data, we believe that costs associated with adopting two new eCQMs are multifaceted and include not only the burden associated with reporting, but also the costs associated with implementing and maintaining all of the eCQMs available for use in the Hospital IQR Program in hospitals' EHR systems ( 86 FR 45607 ). We do not believe the remaining policies will result in any additional economic impact beyond the additional collection of information burden discussed in section XII.B.6 of this final rule.
Historically, 100 hospitals, on average, that participate in the Hospital IQR Program do not receive the full annual percentage increase in any fiscal year due to the failure to meet all requirements of the Hospital IQR Program. We anticipate that the number of hospitals not receiving the full annual percentage increase will be approximately the same as in past years based on review of previous performance.
We received no comments on our assumptions regarding effects of requirements discussed in this final rule.
In section IX.D. of the preamble of this final rule, we discuss finalized requirements for PPS-exempt cancer hospitals (PCHs) reporting quality data under the PCH Quality Reporting (PCHQR) Program. The PCHQR Program is authorized under section 1866(k) of the Act. There is no financial impact to PCH Medicare reimbursement if a PCH does not submit data.
In the preamble of this final rule, we are: (1) adopting the Patient Safety Structural measure beginning with the CY 2025 reporting period/FY 2027 program year with modification to one domain; (2) modifying the HCAHPS Survey beginning with the CY 2025 reporting period/FY 2027 program year; and (3) moving up the start date for public display of PCH performance on the Hospital Commitment to Health Equity measure.
As shown in the summary table in section XII.B.7.d. of this final rule, we estimate a total information collection burden increase for 11 PCHs of 166 hours at a cost of $4,047 annually associated with our finalized policies and updated burden estimates beginning with the CY 2025 reporting period/FY 2027 program year compared with our currently approved information collection burden estimates. We refer readers to section XII.B.7. of this final rule (Collection of Information) for a detailed discussion of the calculations estimating the changes to the information collection burden for submitting data to the PCHQR Program.
In section IX.B.1. of the preamble of this final rule, we are adopting the Patient Safety Structural measure. We finalized that in order for a PCH to receive a point for a domain in the measure, the PCH will be required to affirmatively attest to each of the statements within that domain. We estimate that if a PCH is unable to attest affirmatively to all of the statements in a domain and, in a future program year, desires to earn a point for that domain, the PCH will likely incur costs associated with activities needed to be able to affirmatively attest, which can include updating policies, protocols, or processes; engaging senior leadership; conducting required analyses; or training staff ( 87 FR 49492 ). The extent of these costs will vary from PCH to PCH depending on what policies the PCH already has in place, what activities the PCH is already performing, facility size, and the individual choices each PCH makes in order to meet the criteria necessary to attest affirmatively.
We do not believe the remaining policies to modify the HCAHPS Survey beginning with the CY 2025 reporting period/FY 2027 program year and to move up the start date for public display of PCH performance on the Hospital Commitment to Health Equity measure will result in any additional economic impact beyond the additional collection of information burden discussed in section XII.B.7. of this final rule.
We received no comments and are therefore finalizing our assumptions regarding effects of requirements discussed in this final rule without modification.
In section IX.E. of this final rule, we are finalizing four new items as standardized patient assessment data elements under the SDOH category and to modify the current Transportation item on the LCDS beginning with the FY 2028 LTCH QRP. We are finalizing that LTCHs will collect the four new items at admission using the LCDS for: Living Situation (one item), Food (two items), and Utilities (one item). We are finalizing modifications for the Transportation item, which is currently collected via the LCDS at admission and discharge. We are finalizing that the modified Transportation item will only be collected at admission beginning with the FY 2028 LTCH QRP. We also are finalizing our proposal to extend the admission assessment window for the LCDS from three to four days beginning with the FY 2028 LTCH QRP. Finally, we sought and received information on two topics: future measure concepts for the LTCH QRP and a future LTCH Star Rating system.
The effect of these finalized proposals for the LTCH QRP will be an overall increase in burden for LTCHs participating in the LTCH QRP. As shown in summary table XII.B-09 in section XII.B.8. of this final rule, we estimate a total information collection burden increase for 330 eligible LTCHs of 2,116.55 hours for a cost increase of $138,231.88 annually associated with our proposed policies and updated burden ( print page 70040) estimates for the FY 2028 LTCH QRP program year compared to our currently approved information collection burden estimates. We refer readers to section XII.B.8. of this final rule, where we have provided an estimate of the burden and cost to LTCHs, and note that it will be included in a revised information collection request under OMB control number 0938-1163.
In section IX.F. of the preamble of this final rule, we discuss finalized requirements for eligible hospitals and critical access hospitals (CAHs) to report objectives and measures and electronic clinical quality measures (eCQMs) under the Medicare Promoting Interoperability Program.
In this final rule, we are: (1) adopting the Hospital Harm—Falls with Injury eCQM beginning with the CY 2026 reporting period; (2) adopting the Hospital Harm—Postoperative Respiratory Failure eCQM beginning with the CY 2026 reporting period; (3) modifying the Antimicrobial Use and Resistance (AUR) Surveillance measure by splitting it into an Antimicrobial Use Surveillance measure and an Antimicrobial Resistance Surveillance measure beginning with the electronic health record (EHR) reporting period in CY 2025; (4) modifying the Global Malnutrition Composite Score eCQM, beginning with the CY 2026 reporting period; (5) increasing the total number of eCQMs that must be reported from six to eight eCQMs for the CY 2026 reporting period, from eight to nine eCQMs for the CY 2027 reporting period, and then from nine to eleven eCQMs beginning with the CY 2028 reporting period; and (6) increasing the minimum scoring threshold from 60 points to 70 points for the EHR reporting period in CY 2025 and then from 70 points to 80 points beginning with the EHR reporting period in CY 2026.
As shown in the summary table in section XII.B.9. of this final rule, we estimate a total information collection burden increase of 5,038 hours at a cost of $262,581 annually associated with our finalized policies and updated burden estimates over the four-year period from the EHR reporting period in CY 2025 through the EHR reporting period in CY 2028 compared to our currently approved information collection burden estimates. We refer readers to section XII.B.9.f. of this final rule (Collection of Information) for a detailed discussion of the calculations estimating the changes to the information collection burden for submitting data to the Medicare Promoting Interoperability Program.
In section IX.F.6.a. of the preamble of this final rule, we are adopting two new eCQMs and modifying one eCQM. Similar to our previous discussion in the FY 2022 IPPS/LTCH PPS final rule regarding adoption of eCQM measures for the Hospital IQR Program ( 86 FR 45607 ), costs associated with adopting new or modified eCQMs can be multifaceted and variable, and include not only the burden associated with reporting data to CMS, but also the costs associated with implementing and maintaining all of the eCQMs available for use in the Medicare Promoting Interoperability Program in eligible hospitals' and CAHs' EHR systems.
In section IX.F.5. of the preamble of this final rule, we are finalizing increases to the performance-based scoring threshold for eligible hospitals and CAHs reporting under the Medicare Promoting Interoperability Program from 60 points to 70 points for the EHR reporting period in CY 2025 and from 70 points to 80 points beginning with the EHR reporting period in CY 2026. Our review of the CY 2022 Medicare Promoting Interoperability Program's performance results indicates 98.5 percent of eligible hospitals and CAHs currently successfully meet the threshold of 60 points and 92.8 percent of eligible hospitals and CAHs currently meet the threshold of 70 points, while 81.5 percent of eligible hospitals and CAHs currently exceed a score of 80 points. Therefore, the 11.3 percent and 17 percent of eligible hospitals and CAHs that meet the current threshold of 60 points but not the finalized threshold of 70 points for the EHR reporting period in CY 2025 and 80 points beginning with the EHR reporting period in CY 2026, respectively, will be required to better align their health information systems with evolving industry standards, increase data exchange, or both, to raise their performance score or be subject to a potential downward payment adjustment. We do not believe the remaining policies will result in any additional economic impact beyond the additional collection of information burden discussed in section XII.B.9. of this final rule.
We received no comments on our assumptions regarding these effects.
This final rule contains a range of policies. It also provides descriptions of the statutory provisions that are addressed, identifies the finalized policies, and presents rationales for our decisions and, where relevant, alternatives that were considered.
Section 4122(a) of the CAA, 2023 amended section 1886(h) of the Act by adding a new section 1886(h)(10) of the Act requiring the distribution of an additional 200 residency positions (also referred to as slots) to qualifying hospitals. Section 1886(h)(10)(B)(iii) of the Act further requires that each qualifying hospital that submits a timely application receive at least 1 (or a fraction of 1) of the slots made available under section 1886(h)(10) of the Act before any qualifying hospital receives more than 1 residency position.
As discussed in section V.F.2. of this final rule, after consideration of public comments, we are finalizing our proposal, with minor modifications, to implement section 4122 of the CAA, 2023. In section V.F.2. of the proposed rule, we discussed our proposal to first distribute slots by prorating the available 200 positions among all qualifying hospitals such that each qualifying hospital receives up to 1.00 FTE—that is, 1.00 FTE or a fraction of 1.00 FTE. We proposed that a qualifying hospital is a Category One, Category Two, Category Three, or Category Four hospital, or one that meets the definitions of more than one of these categories, as defined at section 1886(h)(10)(F)(iii) of the Act. [ 1143 ] We proposed that if any residency slots remain after distributing up to 1.00 FTE to each qualifying hospital, we will prioritize the distribution of the remaining slots based on the HPSA score associated with the program for which each qualifying hospital is applying using the methodology we finalized for purposes of implementing section 126 of the CAA, 2021 ( 86 FR 73434 through 73440 ). Using this HPSA prioritization method, we proposed to limit a qualifying hospital's total award under section 4122 of the CAA, 2023, to 10.00 additional FTEs, consistent with section 1886(h)(10)(C)(i) of the Act.
We considered alternative approaches for distribution of additional residency positions under the provisions of section 4122 of the CAA. An alternative we considered placed greater emphasis on the distribution of additional residency positions to hospitals that are training residents in geographic and population HPSAs. Under this approach, the statutory requirement that each qualifying hospital receive 1 slot or a fraction of 1 slot would be met by awarding each qualifying hospital 0.01 FTE. The remaining residency slots would be prioritized for distribution based on the HPSA score associated with the program for which each hospital is applying using the HPSA prioritization methodology we finalized for purposes of implementing section 126 of the CAA, 2021 ( 86 FR 73434 through 73440 ). After consideration of the public comments, as discussed in section V.F.2. of the preamble of this final rule, we did not adopt this alternative.
As discussed in section V.J. of the preamble of this final rule, we are establishing a separate payment under the IPPS to small, independent hospitals of 100 beds or fewer that are not part of a chain organization for the additional resource costs involved in voluntarily establishing and ( print page 70041) maintaining access to 6-month buffer stocks of essential medicines. Although at the current time we do not believe it would be appropriate to expand the pool of hospitals eligible for this initial implementation of the policy due primarily to the existing purchasing power of larger hospitals and chain hospitals and concerns regarding inducing or exacerbating shortages, as we and stakeholders gain experience with the policy it may become appropriate to consider expansion of eligibility in future rulemaking, potentially in conjunction with domestic manufacturing requirements as may be feasible based on increases in the domestic manufacturing capacity of essential medicines.
We are finalizing our proposal to add four new assessment items to the LCDS and modify one assessment item on the LCDS in sections IX.E.4. and IX.E.7.b. of this final rule. We believe adopting these four new assessment items as standardized patient assessment data elements and modifying the current Transportation item will advance the CMS National Quality Strategy Goals of equity and engagement. We considered the alternative of delaying the collection of these four new assessment items and modifying the current Transportation item. However, given the fact they will encourage meaningful collaboration between healthcare providers, caregivers, and community-based organizations to address HRSNs prior to discharge from the LTCH, we believe further delay is unwarranted.
We are also finalizing our proposal to extend the LCDS Admission assessment window in section IX.E.7.c. of this final rule. We considered the option of maintaining the current 3-day assessment period versus extending it to 4 days. However, this policy is responsive to LTCHs' feedback that we received regarding the difficulty of collecting the required LCDS data elements within the 3-day assessment window when medically complex patients are admitted prior to and on weekends. Additionally, extending the assessment period will have no impact on the calculation of LTCH QRP measures, and will only require minimal revisions to the LCDS guidance manuals.
In section X.A. of the preamble of this final rule, we are finalizing the test of a new mandatory episode-based payment model called the Transforming Episode Accountability Model (TEAM). TEAM is designed to improve beneficiary care through financial accountability for episodes categories that begin with one of the following procedures: coronary artery bypass graft, lower extremity joint replacement, major bowel procedure, surgical hip/femur fracture treatment, and spinal fusion. TEAM will test whether financial accountability for these episode categories reduces Medicare expenditures while preserving or enhancing the quality of care for Medicare beneficiaries. We anticipate that TEAM will benefit Medicare beneficiaries through improving the coordination of items and services paid for through Medicare FFS payments, encouraging provider investment in health care infrastructure and redesigned care processes, and incentivizing higher value care across the inpatient and post-acute care settings for the episode.
Throughout this final rule, we have identified our policies and alternatives that we have considered and provided information as to the effects of these alternatives and the rationale for each of the policies. For example, we considered allowing physician group practices (PGPs) to be TEAM participants, however, we are concerned that PGPs are generally smaller entities and care for a lower volume of Medicare beneficiaries which could make it challenging to take on the level of financial risk to participate in the model. In the proposed rule we solicited comments on our proposals, on the alternatives we have identified, and on other alternatives that we should consider. We note that our estimates are limited to acute care hospitals that are selected to participate in this model and do not include the acute care hospitals that voluntarily opt-in to TEAM due to the high degree of uncertainty regarding the level of interest that these potential participants will have in TEAM. This model will not directly affect hospitals that are not participating in the model. However, the model may encourage innovations in health care delivery in other areas or in care reimbursed through other payers. For example, a TEAM participant may choose to extend their arrangements to arrangements outside of the model for all surgical procedures they provide, as permitted by all applicable laws, not just those reimbursed by Medicare and tested in TEAM. We welcomed comments on our proposals and the alternatives we have identified in the preamble of this final rule. In each section of the final rule that we received comments, we have addressed them accordingly.
Acute care hospitals are estimated to experience an increase of approximately $2.9 billion in FY 2025, including operating, capital, and the combined effects of (1) the changes to add-on payments for certain ESRD discharges, (2) the payment adjustment for establishing and maintaining access to a buffer stock of essential medicines, (3) new technology add-on payment changes, and (4) the statutory expiration of the MDH program and the temporary changes to the low-volume hospital payment adjustment on January 1, 2025. The estimated change in operating payments is approximately $2.7 billion (discussed in sections I.F. of this Appendix). The estimated change in capital payments is approximately $0.209 billion (discussed in section I.I. of this Appendix). The estimated change in the combined effects of (1) the changes to add-on payments for certain ESRD discharges; (2) the payment adjustment for establishing and maintaining access to a buffer stock of essential medicines; (3) new technology add-on payment changes; and (4) the statutory expiration of the temporary changes to the low-volume hospital payment adjustment on January 1, 2025 is approximately $0.021 billion as discussed in sections I.F and I.G. of this Appendix. Totals may differ from the sum of the components due to rounding.
Table I. of section I.F. of this Appendix also demonstrates the estimated redistributional impacts of the IPPS budget neutrality requirements for the MS-DRG and wage index changes, and for the wage index reclassifications under the MGCRB.
We estimate that hospitals will experience a 2.8 percent increase in capital payments per case, as shown in Table III. of section I.I. of this Appendix. We project that there will be a $209 million increase in capital payments in FY 2025 compared to FY 2024.
The discussions presented in the previous pages, in combination with the remainder of this final rule, constitute a regulatory impact analysis.
Overall, LTCHs are projected to experience an increase in estimated payments in FY 2025. In the impact analysis, we are using the rates, factors, and policies presented in this final rule based on the best available claims and CCR data to estimate the change in payments under the LTCH PPS for FY 2025. Accordingly, based on the best available data for the 331 LTCHs included in our analysis, we estimate that overall FY 2025 LTCH PPS payments would increase approximately $58 million relative to FY 2024, primarily due to the annual update to the LTCH PPS standard Federal rate partially offset by an estimated decrease in high-cost outlier payments.
If regulations impose administrative costs on private entities, such as the time needed to read and interpret a rule, we should estimate the cost associated with regulatory review. Due to the uncertainty involved with accurately quantifying the number of entities that would review the final rule, we assumed that the total number of timely pieces of correspondence on this year's proposed rule would be the number of reviewers of the final rule. We acknowledge that this assumption may understate or overstate the costs of reviewing the rule. It is possible that not all commenters reviewed this year's rule in detail, and it is also possible that some reviewers chose not to comment on the proposed rule. For these reasons, we believe that the number of past commenters would be a fair estimate of the number of reviewers of the final rule.
We recognize that different types of entities are in many cases affected by mutually exclusive sections of the rule. Thus, for the purposes of our estimate we assume that each reviewer read approximately 50 percent of the proposed rule. Finally, in our estimates, we have used the 6,180 number of timely pieces of correspondence on the FY 2025 IPPS/LTCH PPS proposed rule as our estimate for the number of reviewers of the final rule. We continue to acknowledge the uncertainty involved with using this number, but we believe it is a fair estimate due to the variety of entities affected and the likelihood that some of them choose to rely (in full or in part) on press releases, newsletters, fact sheets, or other sources rather than the ( print page 70042) comprehensive review of preamble and regulatory text.
Using the wage information from the BLS for medical and health service managers (Code 11-9111), we estimate that the cost of reviewing the final rule is $129.28 per hour, including overhead and fringe benefits ( https://www.bls.gov/oes/current/oes_nat.htm ). Assuming an average reading speed, we estimate that it would take approximately 32.94 hours for the staff to review half of this final rule. For each IPPS hospital or LTCH that reviews this final rule, the estimated cost is $4,258.48 (32.94 hours × $129.28). Therefore, we estimate that the total cost of reviewing this final rule is $26,317,406 ($4,258.48 × 6,180 reviewers).
As required by OMB Circular A-4 (available at https://www.whitehouse.gov/wp-content/uploads/legacy_drupal_files/omb/circulars/A4/a-4.pdf ), in Table V. of this Appendix, we have prepared an accounting statement showing the classification of the expenditures associated with the provisions of this final rule as they relate to acute care hospitals. This table provides our best estimate of the change in Medicare payments to providers as a result of the changes to the IPPS presented in this final rule. All expenditures are classified as transfers to Medicare providers.
As shown in Table V. of this Appendix, the net costs to the Federal Government associated with the policies in this final rule are estimated at $2.9 billion.

As discussed in section I.J. of this Appendix, the impact analysis of the payment rates and factors presented in this final rule under the LTCH PPS is projected to result in an increase in estimated aggregate LTCH PPS payments in FY 2025 relative to FY 2024 of approximately $58 million based on the data for 331 LTCHs in our database that are subject to payment under the LTCH PPS. Therefore, as required by OMB Circular A-4 (available at https://www.whitehouse.gov/wp-content/uploads/legacy_drupal_files/omb/circulars/A4/a-4.pdf ), in Table VI. of this Appendix, we have prepared an accounting statement showing the classification of the expenditures associated with the provisions of this final rule as they relate LTCHs. Table VI. of this Appendix provides our best estimate of the estimated change in Medicare payments under the LTCH PPS as a result of the payment rates and factors and other provisions presented in this final rule based on the data for the 331 LTCHs in our database. All expenditures are classified as transfers to Medicare providers (that is, LTCHs).
As shown in Table VI. of this Appendix, the net cost to the Federal Government associated with the policies for LTCHs in this final rule are estimated at $58 million.

The RFA requires agencies to analyze options for regulatory relief of small entities. For purposes of the RFA, small entities include small businesses, nonprofit organizations, and small government jurisdictions. We estimate that most hospitals and most other providers and suppliers are small entities as that term is used in the RFA. The great majority of hospitals and most other health care providers and suppliers are small entities, either by being nonprofit organizations or by meeting the SBA definition of a small business (having revenues of less than $8.0 million to $41.5 million in any 1 year). (For details on the latest standards for health care providers, we refer readers to page 38 of the Table of Small Business Size Standards for NAIC 622 found on the SBA website at https://www.sba.gov/sites/default/files/files/Size_Standards_Table.pdf .)
For purposes of the RFA, all hospitals and other providers and suppliers are considered to be small entities. Because all hospitals are considered to be small entities for purposes of the RFA, the hospital impacts described in this final rule are impacts on small entities. Individuals and States are not included in the definition of a small entity. MACs are not considered to be small entities because they do not meet the SBA definition of a small business.
HHS's practice in interpreting the RFA is to consider effects “economically significant” if greater than 5 percent of providers reach a threshold of 3 to 5 percent or more of total revenue or total costs. We believe that the provisions of this final rule relating to IPPS hospitals would have an economically significant impact on small entities as explained in this Appendix. Therefore, the Secretary has certified that this final rule would have a significant economic impact on a substantial number of small entities. For example, the majority of the 3,082 IPPS hospitals included in the impact analysis shown in “Table I.—Impact Analysis of Changes to the IPPS for Operating Costs for FY 2025,” on average are expected to see increases in the range of 2.8 percent, primarily due to the hospital rate update, as discussed in section I.F. of this Appendix. On average, the rate update for these hospitals is estimated to be 2.9 percent.
The 330 LTCH PPS hospitals included in the impact analysis shown in “Table IV: Impact of Payment Rate and Policy Changes to LTCH PPS Payments for LTCH PPS Standard Federal Payment Rate Cases for FY 2025 (Estimated FY 2024 Payments Compared to Estimated FY 2025 Payments)” on average are expected to see an increase of approximately 2.0 percent, primarily due to the annual standard Federal rate update for FY 2025 (3.0 percent) being partially offset by a projected 0.8 percentage point decrease in high cost outlier payments as a percentage of total LTCH PPS standard Federal payment rate payments, as discussed in section I.J. of this Appendix.
This final rule contains a range of final policies. It provides descriptions of the statutory provisions that are addressed, identifies the finalized policies, and presents rationales for our decisions and, where ( print page 70043) relevant, alternatives that were considered. The analyses discussed in this Appendix and throughout the preamble of this final rule constitutes our regulatory flexibility analysis. We solicited public comments on our estimates and analysis of the impact of our proposals on small entities.
Section 1102(b) of the Act requires us to prepare a regulatory impact analysis for any proposed or final rule that may have a significant impact on the operations of a substantial number of small rural hospitals. This analysis must conform to the provisions of section 603 of the RFA. With the exception of hospitals located in certain New England counties, for purposes of section 1102(b) of the Act, we define a small rural hospital as a hospital that is located outside of an urban area and has fewer than 100 beds. Section 601(g) of the Social Security Amendments of 1983 (Pub. L. 98-21) designated hospitals in certain New England counties as belonging to the adjacent urban area. Thus, for purposes of the IPPS and the LTCH PPS, we continue to classify these hospitals as urban hospitals.
As shown in Table I. in section I.F. of this Appendix, rural IPPS hospitals with 0-49 beds (341 hospitals) are expected to experience an increase in payments from FY 2024 to FY 2025 of 1.6 percent and rural IPPS hospitals with 50-99 beds (182 hospitals) are expected to experience an increase in payments from FY 2024 to FY 2025 of 1.4 percent. These changes are primarily driven by the hospital rate update offset by the statutory expiration of the MDH program. We refer readers to Table I. in section I.F. of this Appendix for additional information on the quantitative effects of the policy changes under the IPPS for operating costs.
All rural LTCHs (19 hospitals) shown in Table IV. in section I.J. of this Appendix have less than 100 beds. These hospitals are expected to experience an increase in payments from FY 2024 to FY 2025 of 2.8 percent. This increase is primarily due to the combination of the 3.0 percent annual update to the LTCH PPS standard Federal payment rate for FY 2025, the changes to the area wage level adjustment, and estimated changes in outlier payments, as discussed in section I.J. of this Appendix.
Section 202 of the Unfunded Mandates Reform Act of 1995 ( Pub. L. 104-4 ) also requires that agencies assess anticipated costs and benefits before issuing any rule whose mandates require spending in any 1 year of $100 million in 1995 dollars, updated annually for inflation. In 2024, that threshold level is approximately $183 million. This final rule would not mandate any requirements that meet the threshold for State, local, or tribal governments, nor would it affect private sector costs.
Executive Order 13132 establishes certain requirements that an agency must meet when it promulgates a proposed rule (and subsequent final rule) that imposes substantial direct requirement costs on state and local governments, preempts state law, or otherwise has federalism implications. This final rule would not have a substantial direct effect on state or local governments, preempt states, or otherwise have a federalism implication.
Executive Order 13175 directs agencies to consult with Tribal officials prior to the formal promulgation of regulations having tribal implications. Section 1880(a) of the Act states that a hospital of the Indian Health Service, whether operated by such Service or by an Indian tribe or tribal organization, is eligible for Medicare payments so long as it meets all of the conditions and requirements for such payments which are applicable generally to hospitals. Consistent with section 1880(a) of the Act, this final rule contains general provisions also applicable to hospitals and facilities operated by the Indian Health Service or Tribes or Tribal organizations under the Indian Self-Determination and Education Assistance Act. We continue to engage in consultations with Tribal officials on IPPS issues of interest. We use input received from these consultations, as well as the comments on the proposed rule, to inform our rulemaking.
In accordance with the provisions of Executive Order 12866 , the Office of Management and Budget reviewed this final rule.
Section 1886(e)(4)(A) of the Act requires that the Secretary, taking into consideration the recommendations of MedPAC, recommend update factors for inpatient hospital services for each fiscal year that take into account the amounts necessary for the efficient and effective delivery of medically appropriate and necessary care of high quality. Under section 1886(e)(5) of the Act, we are required to publish update factors recommended by the Secretary in the proposed and final IPPS rules. Accordingly, this Appendix provides the recommendations for the update factors for the IPPS national standardized amount, the hospital-specific rate for SCHs and MDHs, and the rate-of-increase limits for certain hospitals excluded from the IPPS, as well as LTCHs. In prior years, we made a recommendation in the IPPS proposed rule and final rule for the update factors for the payment rates for IRFs and IPFs. However, for FY 2025, consistent with our approach for FY 2024, we are including the Secretary's recommendation for the update factors for IRFs and IPFs in separate Federal Register documents at the time that we announce the annual updates for IRFs and IPFs. We also discuss our response to MedPAC's recommended update factors for inpatient hospital services.
As discussed in section V.B. of the preamble to this final rule, for FY 2025, consistent with section 1886(b)(3)(B) of the Act, as amended by sections 3401(a) and 10319(a) of the Affordable Care Act, we are setting the applicable percentage increase by applying the following adjustments in the following sequence. Specifically, the applicable percentage increase under the IPPS is equal to the rate-of-increase in the hospital market basket for IPPS hospitals in all areas, subject to a reduction of one-quarter of the applicable percentage increase (prior to the application of other statutory adjustments; also referred to as the market basket update or rate-of-increase (with no adjustments)) for hospitals that fail to submit quality information under rules established by the Secretary in accordance with section 1886(b)(3)(B)(viii) of the Act and a reduction of three-quarters of the applicable percentage increase (prior to the application of other statutory adjustments; also referred to as the market basket update or rate-of-increase (with no adjustments)) for hospitals not considered to be meaningful electronic health record (EHR) users in accordance with section 1886(b)(3)(B)(ix) of the Act, and then subject to an adjustment based on changes in economy-wide productivity (the productivity adjustment). Section 1886(b)(3)(B)(xi) of the Act, as added by section 3401(a) of the Affordable Care Act, states that application of the productivity adjustment may result in the applicable percentage increase being less than zero.
We note that, in compliance with section 404 of the MMA, in the FY 2022 IPPS/LTCH PPS final rule ( 86 FR 45194 through 45204 ), we replaced the 2014-based IPPS operating and capital market baskets with the rebased and revised 2018-based IPPS operating and capital market baskets beginning in FY 2022.
In the FY 2025 IPPS/LTCH PPS proposed rule, in accordance with section 1886(b)(3)(B) of the Act, we proposed to base the proposed FY 2025 market basket update used to determine the applicable percentage increase for the IPPS on IGI's fourth quarter 2023 forecast of the 2018-based IPPS market basket rate-of-increase with historical data through third quarter 2023, which was estimated to be 3.0 percent. In accordance with section 1886(b)(3)(B) of the Act, as amended by section 3401(a) of the Affordable Care Act, in section IV.B. of the preamble of the FY 2025 IPPS/LTCH PPS proposed rule, based on IGI's fourth quarter 2023 forecast, we proposed a productivity adjustment of 0.4 percentage point for FY 2025. We also proposed that if more recent data subsequently became available, we would use such data, if appropriate, to determine the FY 2025 market basket update and productivity adjustment for the FY 2025 IPPS/LTCH PPS final rule.
In the FY 2025 IPPS/LTCH PPS proposed rule, based on IGI's fourth quarter 2023 forecast of the 2018-based IPPS market basket update and the productivity adjustment, depending on whether a hospital submits quality data under the rules established in accordance with section 1886(b)(3)(B)(viii) of the Act (hereafter referred to as a hospital that submits quality data) and is a meaningful EHR user under section 1886(b)(3)(B)(ix) of the Act (hereafter referred to as a hospital that is a meaningful EHR ( print page 70044) user), we presented four possible applicable percentage increases that could be applied to the standardized amount.
In accordance with section 1886(b)(3)(B) of the Act, as amended by section 3401(a) of the Affordable Care Act, we are establishing the applicable percentages increase for the FY 2025 updates based on IGI's second quarter 2024 forecast of the 2018-based IPPS market basket of 3.4 percent and the productivity adjustment of 0.5 percentage point, as discussed in section V.A of the preamble of this final rule, depending on whether a hospital submits quality data under the rules established in accordance with section 1886(b)(3)(B)(viii) of the Act and is a meaningful EHR user under section 1886(b)(3)(B)(ix) of the Act, as shown in the table that follows.

Section 1886(b)(3)(B)(iv) of the Act provides that the FY 2025 applicable percentage increase in the hospital-specific rate for SCHs and MDHs equals the applicable percentage increase set forth in section 1886(b)(3)(B)(i) of the Act (that is, the same update factor as for all other hospitals subject to the IPPS).
Section 307 of the Consolidated Appropriations Act, 2024 (CAA, 2024) ( Pub. L. 118-42 ), enacted on March 9, 2024, extended the MDH program for FY 2025 discharges occurring before January 1, 2025. Prior to enactment of the CAA, 2024, the MDH program was only to be in effect through the end of FY 2024. Therefore, under current law, the MDH program will expire for discharges on or after January 1, 2025. We refer readers to section V.E. of the preamble of this final rule for further discussion of the MDH program.
As previously stated, the update to the hospital specific rate for SCHs and MDHs is subject to section 1886(b)(3)(B)(i) of the Act, as amended by sections 3401(a) and 10319(a) of the Affordable Care Act. Accordingly, depending on whether a hospital submits quality data and is a meaningful EHR user, we are establishing the same four possible applicable percentage increases in the previous table for the hospital-specific rate applicable to SCHs and MDHs.
Because Puerto Rico hospitals are no longer paid with a Puerto Rico-specific standardized amount under the amendments to section 1886(d)(9)(E) of the Act, there is no longer a need for us to make an update to the Puerto Rico standardized amount. Hospitals in Puerto Rico are now paid 100 percent of the national standardized amount and, therefore, are subject to the same update to the national standardized amount discussed under section V.B.1. of the preamble of this final rule.
In addition, as discussed in section V.B.2. of the preamble of this final rule, section 602 of Public Law 114-113 amended section 1886(n)(6)(B) of the Act to specify that subsection (d) Puerto Rico hospitals are eligible for incentive payments for the meaningful use of certified EHR technology, effective beginning FY 2016. In addition, section 1886(n)(6)(B) of the Act was amended to specify that the adjustments to the applicable percentage increase under section 1886(b)(3)(B)(ix) of the Act apply to subsection (d) Puerto Rico hospitals that are not meaningful EHR users, effective beginning FY 2022.
Section 1886(b)(3)(B)(ix) of the Act in conjunction with section 602(d) of Public Law 114-113 requires that for FY 2024 and subsequent fiscal years, any subsection (d) Puerto Rico hospital that is not a meaningful EHR user as defined in section 1886(n)(3) of the Act and not subject to an exception under section 1886(b)(3)(B)(ix) of the Act will have a reduction of three-quarters of the applicable percentage increase (prior to the application of other statutory adjustments).
Based on IGI's fourth quarter 2023 forecast of the 2018-based IPPS market basket update with historical data through third quarter 2023, in the FY 2025 IPPS/LTCH PPS proposed rule, in accordance with section 1886(b)(3)(B) of the Act, as previously discussed, for Puerto Rico hospitals, we proposed a market basket update of 3.0 percent and a productivity adjustment of 0.4 percentage point. Therefore, for FY 2025, depending on whether a Puerto Rico hospital is a meaningful EHR user, we stated that there are two possible applicable percentage increases that can be applied to the standardized amount. Based on these data, we proposed the following applicable percentage increases to the standardized amount for FY 2025 for Puerto Rico hospitals:
- For a Puerto Rico hospital that is a meaningful EHR user, we proposed an applicable percentage increase to the operating standardized amount of 2.6 percent (that is, the FY 2025 estimate of the proposed market basket rate-of-increase of 3.0 percent less an adjustment of 0.4 percentage point for the proposed productivity adjustment).
- For a Puerto Rico hospital that is not a meaningful EHR user, we proposed an applicable percentage increase to the operating standardized amount of 0.35 percent (that is, the FY 2025 estimate of the proposed market basket rate-of-increase of 3.0 percent, less an adjustment of 2.25 percentage point (the proposed market basket rate-of-increase of 3.0 percent × 0.75 for failure to be a meaningful EHR user), and less an adjustment of 0.4 percentage point for the proposed productivity adjustment).
As noted previously, we proposed that if more recent data subsequently became available, we would use such data, if appropriate, to determine the FY 2025 market basket update and the productivity adjustment for the FY 2025 IPPS/LTCH PPS final rule.
As discussed in section V.A.1. of the preamble of this final rule, based on more recent data available for this FY 2025 IPPS/LTCH PPS final rule, we estimate that the FY 2025 market basket update used to determine the applicable percentage increase for the IPPS is 3.4 percent less a productivity adjustment of 0.5 percentage point. Therefore, in accordance with section 1886(b)(3)(B) of the Act, for this final rule, for Puerto Rico hospitals the more recent update of the market basket update is 3.4 percent less a productivity adjustment of 0.5 percentage point. For FY 2025, depending on whether a Puerto Rico hospital is a meaningful EHR user, there are two possible applicable percentage increases that can be applied to the standardized amount. Based on these data, we determined the following applicable percentage increases to the standardized amount for FY 2025 for Puerto Rico hospitals:
- For a Puerto Rico hospital that is a meaningful EHR user, an applicable ( print page 70045) percentage increase to the FY 2025 operating standardized amount of 2.9 percent (that is, the FY 2025 estimate of the market basket rate-of-increase of 3.4 percent less 0.5 percentage point for the productivity adjustment).
- For a Puerto Rico hospital that is not a meaningful EHR user, an applicable percentage increase to the operating standardized amount of 0.35 percent (that is, the FY 2025 estimate of the market basket rate-of-increase of 3.4 percent, less an adjustment of 2.55 percentage point (the market basket rate-of-increase of 3.4 percent × 0.75 for failure to be a meaningful EHR user), and less 0.5 percentage point for the productivity adjustment).
Section 1886(b)(3)(B)(ii) of the Act is used for purposes of determining the percentage increase in the rate-of-increase limits for children's hospitals, cancer hospitals, and hospitals located outside the 50 States, the District of Columbia, and Puerto Rico (that is, short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and America Samoa). Section 1886(b)(3)(B)(ii) of the Act sets the rate-of-increase limits equal to the market basket percentage increase. In accordance with § 403.752(a) of the regulations, religious nonmedical health care institutions (RNHCIs) are paid under the provisions of § 413.40, which also use section 1886(b)(3)(B)(ii) of the Act to update the percentage increase in the rate-of-increase limits.
Currently, children's hospitals, PPS-excluded cancer hospitals, RNHCIs, and short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa are among the remaining types of hospitals still paid under the reasonable cost methodology, subject to the rate-of-increase limits. In addition, in accordance with § 412.526(c)(3) of the regulations, extended neoplastic disease care hospitals (described in § 412.22(i) of the regulations) also are subject to the rate-of-increase limits. As discussed in section VI. of the preamble of this final rule, we proposed to use the percentage increase in the 2018-based IPPS operating market basket to update the target amounts for children's hospitals, PPS-excluded cancer hospitals, RNHCIs, short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa, and extended neoplastic disease care hospitals for FY 2025 and subsequent fiscal years. Accordingly, for FY 2025, the rate-of-increase percentage to be applied to the target amount for these children's hospitals, cancer hospitals, RNHCIs, extended neoplastic disease care hospitals, and short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa is the FY 2025 percentage increase in the 2018-based IPPS operating market basket. For this final rule, the current estimate of the IPPS operating market basket percentage increase for FY 2025 is 3.4 percent.
Section 123 of Public Law 106-113 , as amended by section 307(b) of Public Law 106-554 (and codified at section 1886(m)(1) of the Act), provides the statutory authority for updating payment rates under the LTCH PPS.
As discussed in section V.A. of the Addendum to this final rule, we are updating the LTCH PPS standard Federal payment rate for FY 2025 by 3.0 percent, consistent with section 1886(m)(3) of the Act which provides that any annual update be reduced by the productivity adjustment described in section 1886(b)(3)(B)(xi)(II) of the Act (that is, the productivity adjustment). Furthermore, in accordance with the LTCH QR Program under section 1886(m)(5) of the Act, we are reducing the annual update to the LTCH PPS standard Federal rate by 2.0 percentage points for failure of a LTCH to submit the required quality data. Accordingly, we are establishing an update factor of 1.030 in determining the LTCH PPS standard Federal rate for FY 2025. For LTCHs that fail to submit quality data for FY 2025, we are establishing an annual update to the LTCH PPS standard Federal rate of 1.0 percent (that is, the annual update for FY 2025 of 3.0 percent less 2.0 percentage points for failure to submit the required quality data in accordance with section 1886(m)(5)(C) of the Act and our rules) by applying a update factor of 1.010 in determining the LTCH PPS standard Federal rate for FY 2025. (We note that, as discussed in section VII.D. of the preamble of this final rule, the update to the LTCH PPS standard Federal payment rate of 3.0 percent for FY 2025 does not reflect any budget neutrality factors.)
MedPAC is recommending inpatient hospital rates be updated by the amount specified in current law plus 1.5 percent. MedPAC's rationale for this update recommendation is described in more detail in this section. As previously stated, section 1886(e)(4)(A) of the Act requires that the Secretary, taking into consideration the recommendations of MedPAC, recommend update factors for inpatient hospital services for each fiscal year that take into account the amounts necessary for the efficient and effective delivery of medically appropriate and necessary care of high quality. Consistent with current law, depending on whether a hospital submits quality data and is a meaningful EHR user, we are recommending the four applicable percentage increases to the standardized amount listed in the table under section II. of this Appendix B. We are recommending that the same applicable percentage increases apply to SCHs and MDHs.
In addition to making a recommendation for IPPS hospitals, in accordance with section 1886(e)(4)(A) of the Act, we are recommending update factors for certain other types of hospitals excluded from the IPPS. Consistent with our policies for these facilities, we are recommending an update to the target amounts for children's hospitals, cancer hospitals, RNHCIs, short-term acute care hospitals located in the U.S. Virgin Islands, Guam, the Northern Mariana Islands, and American Samoa and extended neoplastic disease care hospitals of 3.4 percent.
For FY 2025, consistent with policy set forth in section VII. of the preamble of this final rule, for LTCHs that submit quality data, we are recommending an update of 3.0 percent to the LTCH PPS standard Federal rate. For LTCHs that fail to submit quality data for FY 2025, we are recommending an annual update to the LTCH PPS standard Federal rate of 1.0 percent.
In its March 2024 Report to Congress, MedPAC assessed the adequacy of current payments and costs, and the relationship between payments and an appropriate cost base. MedPAC recommended an update to the hospital inpatient rates by the amount specified in current law plus 1.5 percent. MedPAC anticipates that their recommendation to update the IPPS payment rate by the amount specified under current law plus 1.5 percent in 2025 would generally be adequate to maintain beneficiaries' access to hospital inpatient and outpatient care and keep IPPS payment rates close to, if somewhat below, the cost of delivering high-quality care efficiently.
MedPAC stated that their recommended update to IPPS and OPPS payment rates of current law plus 1.5 percent may not be sufficient to ensure the financial viability of some Medicare safety-net hospitals with a poor payer mix. MedPAC recommends redistributing the current Medicare safety-net payments (disproportionate share hospital and uncompensated care payments) using the MedPAC-developed Medicare Safety-Net Index (MSNI) for hospitals. In addition, MedPAC recommends adding $4 billion to this MSNI pool of funds to help maintain the financial viability of Medicare safety-net hospitals and recommended to Congress transitional approaches for a MSNI policy.
We refer readers to the March 2024 MedPAC report, which is available for download at www.medpac.gov , for a complete discussion on these recommendations.
In light of these recommendations, and in particular those concerning safety net hospitals, we look forward to working with Congress on these matters. In the FY 2024 IPPS/LTCH PPS proposed rule, we sought comments on the challenges faced by safety-net hospitals and potential approaches to help safety-net hospitals meet those challenges. These comments will inform and guide our future rulemaking and other actions in this area.
We are establishing an applicable percentage increase for FY 2025 of 2.9 percent as described in section 1886(b)(3)(B) of the Act, provided the hospital submits quality data and is a meaningful EHR user consistent with these statutory requirements. We note that, because the operating and capital payments in the IPPS remain separate, we are continuing to use separate updates for operating and capital payments in the IPPS. The update to the capital rate is discussed in section III. of the Addendum to this final rule. ( print page 70046)
We note that section 1886(d)(5)(F) of the Act provides for additional Medicare payment adjustments, called Medicare disproportionate share hospital (DSH) payments, for subsection (d) hospitals that serve a significantly disproportionate number of low-income patients. Section 1886(r) of the Act provides that, for FY 2014 and each subsequent fiscal year, the Secretary shall pay each such subsection (d) hospital that is eligible for Medicare DSH payments an empirically justified DSH payment equal to 25 percent of the Medicare DSH adjustment they would have received under section 1886(d)(5)(F) of the Act if subsection (r) did not apply. The remaining amount, equal to an estimate of 75 percent of what otherwise would have been paid as Medicare DSH payments if subsection (r) of the Act did not apply, reduced to reflect changes in the percentage of individuals who are uninsured, is available to make additional payments to each hospital that qualifies for Medicare DSH payments and has uncompensated care. These additional payments are called uncompensated care payments. We refer readers to section IV. of preamble of this final rule for a further discussion of Medicare DSH and uncompensated care payments.
1. https://www.cms.gov/about-cms/what-we-do/cms-strategic-plan .
2. https://www.whitehouse.gov/priorities/ .
3. For the BPCI Advanced model, the last day of the last performance period is December 31, 2025. For the CJR model, the last day of the last performance year is December 31, 2024.
[1]. Available at 86 FR 7009 (January 25, 2021) ( https://www.federalregister.gov/documents/2021/01/25/2021-01753/advancing-racial-equity-and-support-for-underserved-communities-through-the-federal-government ).
4. Zhang Y, et al. Association of total pre-existing comorbidities with stroke risk: a large-scale community-based cohort study from China. BMC Public Health. 2021; 21(1):1910.
5. As noted earlier in the discussion, the code titles were updated but the code numbers themselves did not change.
6. Smith, et al. Global Spine J. 2023 Nov 21.
7. Sadrameli S, et al. ISASS 2024.
8. Available at: https://www.federalregister.gov/documents/2021/01/25/2021-01753/advancing-racial-equity-and-support-for-underserved-communities-through-the-federal-government .
9. Available at: https://health.gov/healthypeople/priority-areas/social-determinants-health .
10. US Bureau of the Census. American Housing Survey (AHS). Washington, DC: US Bureau of the Census; 2010. Available at http://www.census.gov/hhes/www/housing/ahs/ahs.html .
11. US Bureau of the Census. Codebook for the American Housing Survey, public use file: 1997 and later. Washington, DC: US Bureau of the Census; 2009. Available at http://www.huduser.org/portal/datasets/ahs/AHS_Codebook.pdf .
12. Hernández, D. (2016). Affording housing at the expense of health: Exploring the housing and neighborhood strategies of poor families. Journal of Family Issues, 37 (7), 921-946. doi: 10.1177/0192513X14530970.
13. Joint Center for Housing Studies. (2020). The state of the nation's housing 2020. Harvard University. https://www.jchs.harvard.edu/sites/default/files/reports/files/Harvard_JCHS_The_State_of_the_Nations_Housing_2020_Report_Revised_120720.pdf .
14. Krieger J., Higgins D.L., Housing and health: time again for public health action. Am. J. Public Health. 2002 May;92(5):758-68. doi: 10.2105/ajph.92.5.758. PMID: 11988443; PMCID: PMC1447157.
15. Office of Disease Prevention and Health Promotion. Retrieved on December 27, 2023 from https://health.gov/healthypeople/priority-areas/social-determinants-health/literature-summaries/housing-instability .
16. Gu, K.D., Faulkner, K.C. & Thorndike, A.N. Housing instability and cardiometabolic health in the United States: a narrative review of the literature. BMC Public Health 23, 931 (2023). https://doi.org/10.1186/s12889-023-15875-6 .
17. Gertz A.H., Pollack C.C., Schultheiss M.D., Brownstein J.S., Delayed medical care and underlying health in the United States during the COVID-19 pandemic: A cross-sectional study. Prev Med Rep. 2022 Aug;28:101882. doi: 10.1016/j.pmedr.2022.101882. Epub 2022 Jul 5. PMID: 35813398; PMCID: PMC9254505.
18. Rollings KA, Kunnath N, Ryus CR, Janke AT, Ibrahim AM. Association of Coded Housing Instability and Hospitalization in the US. JAMA Netw Open. 2022;5(11):e2241951. doi:10.1001/jamanetworkopen.2022.41951.
19. Berkowitz S.A., Seligman H.K., Meigs J.B., Basu S., Food insecurity, healthcare utilization, and high cost: a longitudinal cohort study. Am. J. Manag. Care. 2018 Sep;24(9):399-404. PMID: 30222918; PMCID: PMC6426124.
20. https://www.cms.gov/files/document/r10571cp.pdf .
21. https://www.cms.gov/files/document/r11727cp.pdf .
22. FDA—Drug Product Distribution After a Complete Response Action to a Changes Being Effected Supplement https://www.fda.gov/media/107451/download .
23. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
24. Background articles are not included in the following table but can be accessed via the online posting for the technology.
25. Frangoul H, et al. Presented at the 65th Annual American Society of Hematology. 11 Dec 2023.
26. Locatelli F, et al. Presented at the 28th Annual European Hematology Association; 11 June 2023.
27. Frangoul H., et al. Exagamglogene Autotemcel for Severe Sickle Cell Disease, New England Journal of Medicine, 390, 18, (1649-1662), (2024). doi/full/10.1056/NEJMoa2309676.
28. Frangoul H., et al. Exagamglogene Autotemcel for Severe Sickle Cell Disease, New England Journal of Medicine, 390, 18, (1649-1662), (2024). doi/full/10.1056/NEJMoa2309676.
29. Centers for Medicare & Medicaid Services. CMS Sickle Cell Disease Action Plan. https://www.cms.gov/files/document/sickle-cell-disease-action-plan.pdf (September 2023).
30. Kanter J, et al. 2023.
31. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
32. Background articles are not included in the following table but can be accessed via the online posting for the technology.
33. Locatelli F, et al. Presented at the 28th Annual European Hematology Association; 11 June 2023.
34. Frangoul H., et al. Exagamglogene Autotemcel for Severe Sickle Cell Disease. N Eng J Med. 2024;390:1649-62. doi/full/10.1056/NEJMoa2309676.
35. NASDAQ. Marizyme, Inc. Completes Acquisition of Somahlution, Inc. and Raises $7.0 Million in Private Placement | Nasdaq (accessed 1/23/2023).
36. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the outline posting for the technology.
37. Background articles are not included in the following table but can be accessed via the online posting for the technology.
38. Szalkiewicz, P, Emmert, MY, and Heinisch, PP, et al (2022). Graft Preservation confers myocardial protection during coronary artery bypass grafting. Frontiers in Cardiovascular Medicine, July 2022, pp 1-10. DOI 10.3389/fcvm.2022.922357.
39. Perrault, LP, Carrier, M, and Voisine, P, et al (2021). Sequential multidetector computed tomography assessments after venous graft treatment solution in coronary artery bypass grafting. Journal of Thoracis and Cardiovascular Surgery. Jan. 2021, Vol. 161, Number 1, 96-106. https://doi.org/10.1016/j.jtcvs.2019.10.115 .
40. Lopez-Menendez J, Castro-Pinto M, and Fajardo E, Miguelena J, et al. Vein graft preservation with an endothelial damage inhibitor in isolated coronary artery bypass surgery: an observational propensity score-matched analysis. J Thorac Dis 2023;15(10):5549-5558.
41. Haime, M, McLean RR, and Kurgansky KE, et al (2018). Relationship between intra-operative vein graft treatment with DuraGraft® or saline and clinical outcomes after coronary artery bypass grafting, Expert Review of Cardiovascular Therapy, 16:12, 963-970. DOI: 10.1080/14779072.2018.1532289.
42. Lopez-Menendez J, Castro-Pinto M, and Fajardo E, Miguelena J, et al. Vein graft preservation with an endothelial damage inhibitor in isolated coronary artery bypass surgery: an observational propensity score-matched analysis. J Thorac Dis 2023;15(10):5549-5558.
43. Perrault et al.(2021) “Sequential multidetector computed tomography assessments after venous graft treatment solution in coronary artery bypass grafting” Journal of Cardiothoracic and Cardiovascular Surgery, 2021, Vol 161, Number 1, 96-106.
44. Internal Study Report comparing mortality over three years in patients from the DuraGraft Registry undergoing isolated CABG to three-year mortality in standard of care patients from the Society of Thoracic Surgeons (STS) Registry. The data is reported in an Internal Study Report and is expected to be published by August 2024.
45. Shahian DM, O'Brian SM, Sheng S, et al. Predictors of long-term survival after coronary artery bypass grafting surgery: Results from the Society of Thoracic Surgeons Adult Cardiac Surgery Database (The ASCERT Study). Circulation 2012; 125:1491-1500.
46. Gaudino M et al., Operative Outcomes of Women Undergoing Coronary Artery Bypass Surgery in the US, 2011 to 2020. JAMA Surg. 2023 May 1;158(5):494-502. doi: 10.1001/jamasurg.2022.8156. PMID: 36857059; PMCID: PMC9979009.
47. Goldstein DJ et al., External Support for Saphenous Vein Grafts in Coronary Artery Bypass Surgery: A Randomized Clinical Trial. JAMA C ardiol. 2022 Aug 1;7(8):808-816. doi: 10.1001/jamacardio.2022.1437. PMID: 35675092; PMCID: PMC9178499.
48. KFF. Distribution of Medicare beneficiaries by race/ethnicity ( https://www.kff.org/medicare/state-indicator/medicare-beneficiaries-by-raceethnicity/?currentTimeframe=0&sortModel%7B%22colId%22:%22Location%22,%22sort%22:%22asc%22%7D , accessed 06/23/2024).
49. Gupta S, Lui B, Ma X, et al. Sex differences in outcomes after coronary artery bypass grafting. Journal of Cardiothoracic and Vascular Anesthesia 34(2020) 3259-3266.
50. Robinson NB, Naik A, Rahouma M, et al. Sex differences in outcomes following coronary artery bypass grafting: a meta-analysis. Interactive CardioVascular and Thoracic Surgery (2021) 841-847.
51. Dassanayake MT, Norton EL, Ward AF, et al. Sex-specific disparities in patients undergoing isolated CABG. American Heart Journal Plus: Cardiology Research and Practice 35(2023) 100334.
52. Zea-Vera R, Asokan S, Shah RM, et al. Racial/ethnic differences persist in treatment choice and outcomes in isolated intervention for coronary artery disease. The Journal of Thoracic and Cardiovascular Surgery 2022. 166(4). https://doi.org/10.1016/j.jtcvs.2022.01.034 .
53. Dokollari A, Sicouri S, Ramlawi B, et al. Risk predictors of race disparity in patients undergoing coronary artery bypass grafting: a propensity-matched analysis. Interdisciplinary CardioVascular and Thoracic Surgery 2024. 38(1), ivae002.
54. Lala A, Louis C, Vervoort D, et al. Clinical trial diversity, equity, and inclusion: Roadmap of the Cardiothoracic Surgical Trials Network. The Annals of Thoracic Surgery 2024 https://doi.org/10.1016/j.athoracsur.2024.03.016
55. Long C, Williams AO, McGovern AM, et al. Diversity in randomized clinical trials for peripheral artery disease: a systematic review. International Journal for Equity in Health 2024) Volume 23, article number 29.
56. Fleiss JL, Levin B, and Paik MC. Statistical Methods for Rates and Proportions. Third edition. 2003. John Wiley & Sons, Inc.
57. Button KS, Ioannidis JP, Mokrysz C, et al. Power failure: why small sample size undermines the reliability of neuroscience. Nature Reviews Neuroscience. 14, 365-376. 2013.
58. Toto F, Torre T, Turchetto L, Lo Cicero V, Soncin S, Klersy C, Demertzis S, Ferrari E. Efficacy of Intraoperative Vein Graft Storage Solutions in Preserving Endothelial Cell Integrity during Coronary Artery Bypass Surgery. J Clin Med. 2022 Feb 18;11(4):1093. doi: 10.3390/jcm11041093. PMID: 35207364; PMCID: PMC8877698.
59. Bernhard Winkler, David Reineke, Paul Philip Heinisch, Florian Schönhoff, Christoph Huber, Alexander Kadner, Lars Englberger, Thierry Carrel, Graft preservation solutions in cardiovascular surgery, Interactive CardioVascular and Thoracic Surgery, Volume 23, Issue 2, August 2016, Pages 300-309, https://doi.org/10.1093/icvts/ivw056 .
60. Firestone R, Socci N, Shekarkhand T, et al. Antigen escape as a shared mechanism of resistance to BCMA-directed therapies in multiple myeloma. Blood 2024 May 10:blood.2023023557. doi: 10.1182/blood.2023023557.
61. K223843, May 3, 2023; K222242, December 9, 2022; and K200337, March 24, 2020.
62. K191388, June 21, 2019.
63. K223843, May 3, 2023; K222242, December 9, 2022; and K200337, March 24, 2020.
64. K191388, June 21, 2019.
65. Drugs Approved for Melanoma, National Cancer Institute, http://www.cancer.gov/about-cancer/treatment/drugs/melanoma (last updated April 1, 2024).
66. Aronson JK. Meyler's side effects of drugs: The international encyclopedia of adverse drug reactions and interactions. Elsevier Science & Technology; 2015:822.
67. Zager JS, Orloff M, Ferrucci PF, et al. Efficacy and Safety of the Melphalan/Hepatic Delivery System in Patients with Unresectable Metastatic Uveal Melanoma: Results from an Open-Label, Single-Arm, Multicenter Phase 3 Study. Ann Surg Oncol. Published online May 4, 2024. doi:10.1245/s10434-024-15293-x.
68. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
69. Background articles are not included in the following table but can be accessed via the online posting for the technology.
70. Bruning R, Tiede M, Schneider M, et al. Unresectable Hepatic Metastasis of Uveal Melanoma: Hepatic Chemosaturation with High-Dose Melphalan-Long-Term Overall Survival Negatively Correlates with Tumor Burden. Radiol Res Pract. 2020.
71. Vogl TJ, Koch SA, Lotz G, et al. Percutaneous Isolated Hepatic Perfusion as a Treatment for Isolated Hepatic Metastases of Uveal Melanoma: Patient Outcome and Safety in a Multi-centre Study. Cardiovasc Intervent Radiol. Jun 2017;40(6):864-872.
72. Dewald CLA, Hinrichs JB, Becker LS, et al. Chemosaturation with Percutaneous Hepatic Perfusion: Outcome and Safety in Patients with Metastasized Uveal Melanoma. Rofo. Aug 2021;193(8):928-936.
73. Meijer TS, Burgmans MC, de Leede EM, et al. Percutaneous Hepatic Perfusion with Melphalan in Patients with Unresectable Ocular Melanoma Metastases Confined to the Liver: A Prospective Phase II Study. Ann Surg Oncol. Feb 2021;28(2):1130-1141.
74. Karydis I, Gangi A, Wheater MJ, et al. Percutaneous hepatic perfusion with melphalan in uveal melanoma: A safe and effective treatment modality in an orphan disease. J Surg Oncol. May 2018;117(6):1170-1178.
75. Artzner C, Mossakowski O, Hefferman G, et al. Chemosaturation with percutaneous hepatic perfusion of melphalan for liver-dominant metastatic uveal melanoma: a single center experience. Cancer Imaging. Mayphip 30 2019;19(1):31.
76. Tong TML, Samim M, Kapiteijn E, et al. Predictive parameters in patients undergoing percutaneous hepatic perfusion with melphalan for unresectable liver metastases from uveal melanoma: a retrospective pooled analysis. Cardiovasc Intervent Radiol. 2022;45(9):1304-1313.
77. Delcath ASCO 2022 FOCUS Trial Poster; FOCUS Trial Ongoing (See online posting for HepzatoTM. Kit).
78. Hughes MS, Zager J, Faries M, et al. Results of a Randomized Controlled Multicenter Phase III Trial of Percutaneous Hepatic Perfusion Compared with Best Available Care for Patients with Melanoma Liver Metastases. Ann Surg Oncol. Apr 2016;23(4):1309-19.
79. Delcath ASCO 2022 FOCUS Trial Poster; FOCUS Trial Ongoing (See online posting for HepzatoTM. Kit).
80. Carvajal 2023, supra note 2, p. 107.
81. Zager JS, Orloff M, Ferrucci PF, et al. Efficacy and Safety of the Melphalan/Hepatic Delivery System in Patients with Unresectable Metastatic Uveal Melanoma: Results from an Open-Label, Single-Arm, Multicenter Phase 3 Study. Ann Surg Oncol. Published online May 4, 2024. doi:10.1245/s10434-024-15293-x.
82. Dewald CLA, Hinrichs JB, et al. Chemosaturation with Percutaneous Hepatic Perfusion: Outcome and Safety in Patients with Metastasized Uveal Melanoma. Rofo. 2021 Aug;193(8):928-936. English, German. doi: 10.1055/a1348-1932. Epub 2021 Feb 3.
83. Dewald CLA, Warnke MM, et al. Percutaneous Hepatic Perfusion (PHP) with Melphalan in Liver-Dominant Metastatic Uveal Melanoma: The German Experience. Cancers (Basel). 2022 Jan; 14(1): 118. doi:10.3390/cancers14010118.
84. Zager JS, Orloff M, Ferrucci PF, et al. Efficacy and Safety of the Melphalan/Hepatic Delivery System in Patients with Unresectable Metastatic Uveal Melanoma: Results from an Open-Label, Single-Arm, Multicenter Phase 3 Study. Ann Surg Oncol. Published online May 4, 2024. doi:10.1245/s10434-024-15293-x.
85. Tong TML, Fiocco M, van Duijn-de Vreugd JJ, et al. Quality of life analysis of patients treated with percutaneous hepatic perfusion for uveal melanoma liver metastases. Cardiovasc Intervent Radiol. Published online April 8, 2024. doi:10.1007/s00270-024-03713-0.
86. Zager JS, Orloff M, Ferrucci PF, et al. Efficacy and Safety of the Melphalan/Hepatic Delivery System in Patients with Unresectable Metastatic Uveal Melanoma: Results from an Open-Label, Single-Arm, Multicenter Phase 3 Study. Ann Surg Oncol. Published online May 4, 2024. doi:10.1245/s10434-024-15293-x.
87. Nathan et al, New England Journal of Medicine, 2021;385:1196-1206.
88. CellTrans Inc., Cellular, Tissue, and Gene Therapies Advisory Committee Briefing Document Lantidra TM (donislecel) for the Treatment of Brittle Type 1 Diabetes Mellitus. https://www.fda.gov/media/147529/download April 15, 2021. Pages 22 and 105.
89. Background articles are not included in the following table but can be accessed via the online posting for the technology.
90. CellTrans, Inc. 2021, Table 20, p. 60.
91. Ibid.
92. Anderson, S.M., et al., Hybrid Closed-Loop Control Is Safe and Effective for People with Type 1 Diabetes Who Are at Moderate to High Risk for Hypoglycemia. Diabetes Technol Ther, 2019. 21(6): p. 356-363.
93. Nishihama, K., et al., Sudden Death Associated with Severe Hypoglycemia in a Diabetic Patient During Sensor Augmented Pump Therapy with the Predictive Low Glucose Management System. Am J Case Rep, 2021. 22: p. e928090.
94. Kovatchev, B., et al., Randomized Controlled Trial of Mobile Closed-Loop Control. Diabetes Care, 2020. 43(3): p. 607-615.
95. Levy, C.J., et al., 100-LB: Closed-Loop Control Reduces Hypoglycemia without Increased Hyperglycemia in Subjects with Increased Prestudy Hypoglycemia: Results from the iDCL DCLP3 Randomized Trial. Diabetes, 2020. 69(Supplement 1): p. 100-LB.
96. Häggström, E., M. Rehnman, and L. Gunningberg, Quality of life and social life situation in islet transplanted patients: time for a change in outcome measures? Int J Organ Transplant Med, 2011. 2(3): p. 117-25.
97. Radosevich, D.M., et al., Comprehensive health assessment and five-yr follow-up of allogeneic islet transplant recipients. Clin Transplant, 2013. 27(6): p. E715-24.
98. Per the applicant, the data cutoff date was May 20, 2020, which is the date of BLA submission.
99. Vantyghem, Marie-Christine et al. “Ten-Year Outcome of Islet Alone or Islet After Kidney Transplantation in Type 1 Diabetes: A Prospective Parallel-Arm Cohort Study.” Diabetes Care vol. 42,11 (2019): 2042-2049. doi:10.2337/dc19-0401.
100. Czarnecka, Zofia et al. “The Current Status of Allogenic Islet Cell Transplantation.” Cells vol. 12,20 2423. 10 Oct. 2023, doi:10.3390/cells12202423.
101. Marfil-Garza BA, Imes S, Verhoeff K, et al. Pancreatic islet transplantation in type 1 diabetes: 20-year experience from a single-centre cohort in Canada. Lancet Diabetes Endocrinol. 2022;10(7):519-532. doi:10.1016/S2213-8587(22)00114-0.
102. Hering, Bernhard J et al. “Factors associated with favourable 5 year outcomes in islet transplant alone recipients with type 1 diabetes complicated by severe hypoglycaemia in the Collaborative Islet Transplant Registry.” Diabetologia vol. 66,1 (2023): 163-173. doi:10.1007/s00125-022-05804-4.
103. FDA Clears New Insulin Pump and Algorithm-Based Software to Support Enhanced Automatic Insulin Delivery https://www.fda.gov/news-events/press-announcements/fda-clears-new-insulin-pump-and-algorithm-based-software-support-enhanced-automatic-insulin-delivery .
104. FDA approves first automated insulin delivery device for type 1 diabetes https://www.fda.gov/news-events/press-announcements/fda-approves-first-automated-insulin-delivery-device-type-1-diabetes .
105. FDA Roundup: April 21, 2023 https://www.fda.gov/news-events/press-announcements/fda-roundup-april-21-2023 .
106. FDA authorizes first interoperable, automated insulin dosing controller designed to allow more choices for patients looking to customize their individual diabetes management device system https://www.fda.gov/news-events/press-announcements/fda-authorizes-first-interoperable-automated-insulin-dosing-controller-designed-allow-more-choices .
107. Anderson, S.M., et al., Hybrid Closed-Loop Control Is Safe and Effective for People with Type 1 Diabetes Who Are at Moderate to High Risk for Hypoglycemia. Diabetes Technol Ther, 2019. 21(6): p. 356-363.
108. Nishihama, K., et al., Sudden Death Associated with Severe Hypoglycemia in a Diabetic Patient During Sensor Augmented Pump Therapy with the Predictive Low Glucose Management System. Am J Case Rep, 2021. 22: p. e928090.
109. Hering, Bernhard J et al. “ Factors associated with favourable 5 year outcomes in islet transplant alone recipients with type 1 diabetes complicated by severe hypoglycaemia in the Collaborative Islet Transplant Registry. ” Diabetologia vol. 66,1 (2023): 163-173. doi:10.1007/s00125-022-05804-4.
110. BLA Clinical Review Memorandum https://www.fda.gov/media/170827/download .
111. Marfil-Garza BA, Imes S, Verhoeff K, et al. Pancreatic islet transplantation in type 1 diabetes: 20-year experience from a single-centre cohort in Canada. Lancet Diabetes Endocrinol. 2022;10(7):519-532. doi:10.1016/S2213-8587(22)00114-0.
112. Olson D, et al. Immune checkpoint inhibitors (ICI) treatment after progression on anti-PD-1 therapy in advanced melanoma: a systematic literature review. National Comprehensive Care Network (NCCN) Annual Conference, Poster. March-April 2023.
113. Schumacher TN, Schreiber RD: Neoantigens in cancer immunotherapy. Science 348:69-74, 2015.
114. Simpson-Abelson MR, Hilton F, Fardis M, et al: Iovance generation-2 tumor-infiltrating lymphocyte (TIL) product is reinvigorated during the manufacturing process. Ann Oncol 31:S645-S671, 2020 (suppl 4).
115. Raskov H, et al. British Journal of Cancer (2021) 124:359-367; https://doi.org/10.1038/s41416-020-01048-4 .
116. Fardis M, et al. Current and future directions for tumor infiltrating lymphocyte therapy for the treatment of solid tumors. Cell and Gene Therapy Insights, 2020; 6(6), 855-863.
117. FDA. https://www.fda.gov/media/176417/download?attachment —AMTAGVI prescribing information.
118. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
119. Background articles are not included in the following table but can be accessed via the online posting for the technology.
120. Chesney J, et al. J Immunother Cancer 2022;10:3005755. Doi:10.1136/jitc-2022-005755.
121. https://www.cms.gov/Research-Statistics-Data-and-Systems/Statistics-Trends-and-Reports/Chronic-Conditions/Medicare_Beneficiary_Characteristics .
122. Centers for Medicare and Medicaid Services. Chronic Conditions among Medicare Beneficiaries, Chartbook, 2012 Edition. Baltimore, MD. 2012. https://www.cms.gov/research-statistics-data-and-systems/statistics-trends-and-reports/chronic-conditions/downloads/2012chartbook.pdf .
123. Cher, B., Ryan, A.M., Hoffman, G.J., & Sheetz, K.H. (2020). Association of Medicaid Eligibility With Surgical Readmission Among Medicare Beneficiaries. JAMA network open, 3(6), e207426. https://doi.org/10.1001/jamanetworkopen.2020.7426 .
124. Sarnaik A, et al. leucel, a tumor-infiltrating lymphocyte therapy, in metastatic melanoma. J Clin Oncol. 2021;39(24):2656-66. doi:10.1200/JCO.21.00612 (Published online first: 2021/05/13).
125. Chesney J, et al. J Immunother Cancer 2022;10:3005755. Doi:10.1136/jitc-2022-005755.
126. C-144-01 Sources: Chesney J, et al. JITC 2022; Sarnaik A, et al, J Clin Oncol 2021; Sarnaik A, et al. SITC 2022. Chemotherapy Sources: Ribas A, et al. Lancet Oncol 2015; Larkin J, et al. J Clin Oncol 2018; Weichenthal M, et al. J Clin Oncol 2018.
127. Chesney J, et al. J Immunother Cancer 2022; 10 e005755. Doi:101136/jitc-2022-005755.
128. Sarnaik A, et al. Oral presentation. 37th Annual Meeting and Pre-Conference Programs, Society for Immunotherapy of Cancer (SITC). November 10, 2022.
129. Sarnaik A, et al. SITC 2022; Buysse M, Piedbois P. On the relationship between response to treatment and survival. Stat Med. 1996;15:2797-2812.
130. FDA. Clinical trial endpoints for the approval of cancer drugs and biologics. December 2018. https://www.fda.gov/media/71195/download .
131. Haanen JBAG, et al. ESMO Congress 2022. September 2022.
132. Rohaan MW, et al. N Engl J Med 2022;387:2113-25.
133. Seitter SJ, et al. Clin Cancer Res. 2021;27(19):5289-5298.
134. Hassel JC, et al. European Society for Medical Oncology (ESMO) Immunology Annual Congress. Oral presentation, December 2022.
135. Larkin J, et al. European Society for Blood and Marrow Transplantation (EBMT) 49th Annual Meeting. Poster P223 supplement, April 2023.
136. https://www.accessdata.fda.gov/drugsatfda_docs/label/2023/125514s128lbl.pdf .
137. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/125377s115lbl.pdf .
138. https://www.accessdata.fda.gov/drugsatfda_docs/label/2020/125554s078lbl.pdf .
139. https://www.fda.gov/drugs/resources-information-approved-drugs/fda-approves-opdualag-unresectable-or-metastatic-melanoma .
140. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
141. Background articles are not included in the following table but can be accessed via the online posting for the technology.
142. Kanter, J., Walters, M.C., Krishnamurti, L., Mapara, M.Y., Kwiatkowski, J.L, Rifkin-Zenenberg, S., Aygun, B., Kasow, K.A., Pierciey, Jr., F.J., Bonner, M., Miller, A., Zhang, X., Lynch, J., Kim, D., Ribeil, J.A., Asmal, M., Goyal, S., Thompson, A.A., & Tisdale, J.F. (2022). Biologic and Clinical Efficacy of LentiGlobin for Sickle Cell Disease. The New England Journal of Medicine, 386, 617-628. https://doi.org/10.1056/nejmoa2117175 .
143. Kanter J, et al. 65th ASH Annual Meeting and Exposition. December 9-12, 2023. Abstract 1051. Oral presentation (December 11th).
144. Wilson-Frederick SM, et al. Prevalence of sickle cell disease among Medicare Fee-for-Service beneficiaries, age 18-75 years, in 2016. CMS Data Highlight, No 15, June 2019.
145. Food and Drug Administration (FDA). 510(k) Premarket notification for Medtronic Navigation, Inc.'s StealthViz Advanced Planning Application with StealthDTI Package. K081512. May 16, 2008.
146. FDA. K203518. 2021.
147. Hendricks B, Scherschinkski L, Jubran J, et al. Supratentorial Cavernous Malformation Surgery: The Seven Hotspots of Novel Cerebral Risk. Unpublished manuscript.
148. Morell AA, Eichberg DG, Shah AH, et al. Using machine learning to evaluate large-scale brain networks in patients with brain tumors: Traditional and non-traditional eloquent areas. Neurooncol Adv. 2022 Sep 19;4(1):vdac142. Doi: 10.1093/noajnl/vdac142. PMID: 36299797; PMCID: PMC9586213.
149. Shimony J, Zhang D, Johnston JM, et al. Resting-state spontaneous fluctuations in brain activity: A new paradigm for presurgical planning using fMRI. Academic Radiology 16:578-583.
150. Hacker CD, Roland JL, Kim AH, et al. Resting-state network mapping in neurosurgical practice: a review. Neurosurgical Focus December 2019. Volume 47.
151. Lee MH, Smyser CD, and Shimony JS. Resting-state fMRI: A review of methods and clinical applications. American Journal of Neuroradiology Oct 2013. 34:1866-72
152. Shimony J, Zhang D, Johnston JM, et al. Resting-state spontaneous fluctuations in brain activity: A new paradigm for presurgical planning using fMRI. Academic Radiology 16:578-583.
153. Hacker CD, Roland JL, Kim AH, et al. Resting-state network mapping in neurosurgical practice: a review. Neurosurgical Focus December 2019. Volume 47.
154. Lee MH, Smyser CD, and Shimony JS. Resting-state fMRI: A review of methods and clinical applications. American Journal of Neuroradiology Oct 2013. 34:1866-72.
155. 21 CFR 3.2(k) .
156. The Medicare Inpatient Hospitals by Provider and Service dataset provides information on inpatient discharges for Original Medicare Part A beneficiaries by IPPS hospitals. It includes information on the use, payment, and hospital charges for more than 3,000 U.S. hospitals that received IPPS payments. The data are organized by hospital and Medicare Severity Diagnosis Related Group (DRG): https://data.cms.gov/provider-summary-by-type-of-service/medicare-inpatient-hospitals/medicare-inpatient-hospitals-by-provider-and-service .
157. Background articles are not included in the following table but can be accessed via the online posting for the technology.
158. Dadario NB, Sughrue ME. Should Neurosurgeons Try to Preserve Non-Traditional Brain Networks? A Systematic Review of the Neuroscientific Evidence. Journal of Personalized Medicine. 2022; 12(4):587. https://doi.org/10.3390/jpm12040587 .
159. Shah HA, Ablyazova F, Alrez A, et al. Intraoperative awake language mapping correlates to preoperative connectomics imaging: An instructive case. Clin Neurol Neurosurg. 2023 Jun;229:107751. Doi: 10.1016/j.clineuro.2023.107751. Epub 2023 Apr 29. PMID: 3714997. 2.
160. Wu Z, Hu G, Cao B, Liu X, et al. Non-traditional cognitive brain network involvement in insulo-Sylvian gliomas: a case series study and clinical experience using Quicktome. Chin Neurosurg J. 2023 May 26;9(1):16. Doi: 10.1186/s41016-023-00325-4 PMID: 37231522; PMCID: PMC10214670.
161. Morell AA, Eichberg DG, Shah AH, et al. Using machine learning to evaluate large-scale brain networks in patients with brain tumors: Traditional and non-traditional eloquent areas. Neurooncol Adv. 2022 Sep 19;4(1):vdac142. Doi: 10.1093/noajnl/vdac142 PMID: 36299797; PMCID: PMC9586213.
162. Hendricks B, Scherschinkski L, Jubran J, et al. Supratentorial Cavernous Malformation Surgery: The Seven Hotspots of Novel Cerebral Risk (SUBMITTED MANUSCRIPT).
163. Elsamadicy, AA, Sergesketter, A, Adogwa, O, et al. Complications and 30-Day readmission rates after craniotomy/craniectomy: A single Institutional study of 243 consecutive patients, Journal of Clinical Neuroscience, Volume 47, 2018, Pages 178-182, ISSN 0967-5868, https://doi.org/10.1016/ .
164. Phillips KR, Enriquez-Marulanda A, Mackel C, et al. Predictors of extended length of stay related to craniotomy for tumor resection. World Neurosurg X. 2023 Mar 31;19:100176. doi:10.1016/j.wnsx.2023.100176 PMID: 37123627; PMCID: PMC10139985.
165. Vysotski S, Madura C, Swan B, et al. Preoperative FMRI Associated with Decreased Mortality and Morbidity in Brain Tumor Patients. Interdiscip Neurosurg. 2018 Sep;13:40-45. doi: 10.1016/j.inat.2018.02.001 Epub 2018 Feb 14. PMID: 31341789; PMCID: PMC6653633.
166. Dadario NB, Sughrue ME. Should Neurosurgeons Try to Preserve Non-Traditional Brain Networks? A Systematic Review of the Neuroscientific Evidence. Journal of Personalized Medicine. 2022; 12(4):587. https://doi.org/10.3390/jpm12040587 .
167. Department of Health and Human Services (December 13, 2023). HHS Finalizes Rule to Advance Health IT Interoperability and Algorithm Transparency | HHS.gov , accessed 2/20/2024.
168. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
169. Selfi, A, Jafari, S, and Mirmoeeni, S et al. (June 16, 2022) Trends in inpatient utilization of head computerized tomography scans in the United States: A brief cross-sectional study. Cureus 14(6): e26018. DOI 10.7759/cureus.26018.
170. Codes referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
171. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
172. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
173. List of Breakthrough Devices with Marketing Authorization: https://www.fda.gov/medical-devices/how-study-and-market-your-device/breakthrough-devices-program .
174. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
175. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
176. https://www.accessdata.fda.gov/cdrh_docs/pdf23/P230017B.pdf .
177. https://www.aha.org/websites/2017-12-17-aha-central-office .
178. List of Breakthrough Devices with Marketing Authorization: https://www.fda.gov/medical-devices/how-study-and-market-your-device/breakthrough-devices-program .
179. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
180. K222789, January 9, 2023; K200596, October 13, 2020; K193423, May 22, 2020; and K190545, June 20, 2019.
181. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included in the online posting for the technology.
182. Lists referenced here may be found in the cost criterion codes and MS-DRGs attachment included on the online posting for the technology.
183. Section 5 U.S.C. 553(b) .
184. Biden-Harris Administration Announces Action to Increase Access to Sickle Cell Disease Treatments https://www.hhs.gov/about/news/2024/01/30/biden-harris-administration-announces-action-increase-access-sickle-cell-disease-treatments.html .
185. Biden-Harris Administration Announces Action to Increase Access to Sickle Cell Disease Treatments https://www.hhs.gov/about/news/2024/01/30/biden-harris-administration-announces-action-increase-access-sickle-cell-disease-treatments.html .
186. Biden-Harris Administration Announces Action to Increase Access to Sickle Cell Disease Treatments https://www.hhs.gov/about/news/2024/01/30/biden-harris-administration-announces-action-increase-access-sickle-cell-disease-treatments.html .
187. Biden-Harris Administration Announces Action to Increase Access to Sickle Cell Disease Treatments https://www.hhs.gov/about/news/2024/01/30/biden-harris-administration-announces-action-increase-access-sickle-cell-disease-treatments.html .
188. American Hospital Association. AHA FY 2020 IPPS Proposed Rule Comment Letter; Analysis of data from FY 2013-FY 2018. June 24, 2019. Online: https://www.aha.org/system/files/media/file/2019/06/aha-comments-cms-inpatient-pps-fy-2020-proposed-rule-6-24-2019.pdf .
189. We note that while OMB Bulletin 20-01 superseded Bulletin No. 18-04, it included no changes that required CMS to formally adopt the revisions.
190. Known as the “Goldsmith Modification” for its principal developer, Harold F. Goldsmith, this method is described in detail in the paper “Improving the Operational Definition of `Rural Areas' for Federal Programs” available at https://www.ruralhealthinfo.org/pdf/improving-the-operational-definition-of-rural-areas.pdf .
191. UAs are defined by the Census Bureau as densely settled areas with a total population of at least 50,000 people ( 86 FR 2418 ).
192. In accordance with section 1886(d)(8)(C)(i) of the Act, the wage index for hospitals located in a geographic area cannot be reduced by the inclusion of reclassified hospitals. Therefore, if the inclusion of reclassified hospitals reduces the combined wage index by more than 1 percentage point, hospitals reclassified into the area would receive a wage index that includes their data, whereas hospitals geographically located there would receive a wage index that does not.
193. According to the Census Bureau, the effects of the PHE on ACS activities in 2020 resulted in a lower number of addresses (~2.9 million) in the sample, as well as fewer interviews than a typical year.
194. In the FY 2020 IPPS/LTCH proposed rule, we agreed with respondents to a previous request for information who indicated that some current wage index policies create barriers to hospitals with low wage index values from being able to increase employee compensation due to the lag between when hospitals increase the compensation and when those increases are reflected in the calculation of the wage index. We noted that this lag results from the fact that the wage index calculations rely on historical data. We also agreed that addressing this systemic issue did not need to wait for comprehensive wage index reform given the growing disparities between low and high wage index hospitals, including rural hospitals that may be in financial distress and facing potential closure ( 84 FR 19394 and 19395 ).
195. As discussed in the FY 2020 IPPS final rule, the low wage index hospital policy was implemented in a budget neutral manner. In order to ensure that the overall effect of the application of the low wage index hospital policy was budget neutral, we applied a budget neutrality factor of 0.997987 to the FY 2020 standardized amount ( 84 FR 42667 ). The IPPS spending associated with the accounting statement in the FY 2020 IPPS final rule was approximately $113 billion. Applying the budget neutrality adjustment to the IPPS spending associated with the accounting statement results in roughly a $230 million impact of the low wage index hospital policy.
196. MedPAC. (2024). March 2024 Report to the Congress: Medicare Payment Policy. Chapter 3—Hospital inpatient and outpatient services. https://www.medpac.gov/wp-content/uploads/2024/03/Mar24_Ch3_MedPAC_Report_To_Congress_SEC.pdf .
197. https://oig.hhs.gov/oas/reports/region1/12000502.asp .
198. “What are the recent trends in health sector employment?” Peterson-KFF Health System Tracker, March 27, 2024. https://www.healthsystemtracker.org/chart-collection/what-are-the-recent-trends-health-sectoremployment/#Cumulative%20%%20change%20in%20average%20weekly%20earnings,%20since%20February%202020%20-%20January%202024 .
199. https://www.medpac.gov/document/march-2007-report-to-the-congress-medicare-payment-policy/ .
200. https://www.cms.gov/Medicare/Medicare-Fee-for-Service-Payment/AcuteInpatientPPS/dsh .
201. The Rural Community Hospital Demonstration Program was extended for a subsequent 5-year period by sections 3123 and 10313 of the Affordable Care Act ( Pub. L. 111-148 ). The period of performance for this 5-year extension period ended on December 31, 2016. Section 15003 of the 21st Century Cures Act (Pub. L. 114 255), enacted on December 13, 2016, again amended section 410A of Public Law 108-173 to require a 10-year extension period (in place of the 5-year extension required by the Affordable Care Act), therefore requiring an additional 5-year participation period for the demonstration program. Section 15003 of Public Law 114-255 also required a solicitation for applications for additional hospitals to participate in the demonstration program. The period of performance for this 5-year extension period ended December 31, 2021. The Consolidated Appropriations Act, 2021 ( Pub. L. 116-260 ) amended section 410A of Public Law 108-173 to extend the demonstration program for an additional 5-year period.
202. As we have in the past, for additional information on the development of the President's Budget, we refer readers to the Office of Management and Budget website at https://www.whitehouse.gov/omb/budget .
203. We note that the annual reports of the Medicare Boards of Trustees to Congress represent the Federal Government's official evaluation of the financial status of the Medicare Program.
204. https://www.cms.gov/research-statistics-data-and-systems/statistics-trends-and-reports/reportstrustfunds/downloads/technicalpanelreport2010-2011.pdf .
205. For a discussion of general issues regarding Medicaid projections, we refer readers to the 2018 Actuarial Report on the Financial Outlook for Medicaid, which is available at https://www.cms.gov/files/document/2018-report.pdf .
206. https://www.cms.gov/files/document/certification-rates-uninsured-2025-proposed-rule.pdf .
208. The projected decline in Medicaid enrollment from its monthly peak (or the month in which enrollment is at its highest level) is larger than when it is calculated on an average monthly enrollment basis, which conceptually reflects the summation of the monthly enrollment estimates for a given year and divided by 12. As a result, comparisons of Medicaid enrollment across months, or for FY versus CY, can differ notably. This partly explains the Medicaid enrollment estimate differences in the assumptions regarding Medicaid enrollment used in the proposed rule's Factor 1 and 2.
209. OACT Memorandum on Certification of Rates of Uninsured. Available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/disproportionate-share-hospital-dsh .
210. For example, in determining Factor 3 for FY 2023, we did not use the same cost report to determine a hospital's uncompensated care costs for both FY 2018 and FY 2019. Rather, we used the cost report that spanned the entirety of FY 2019 to determine uncompensated care costs for FY 2019 and used the hospital's most recent prior cost report to determine its uncompensated care costs for FY 2018, provided that cost report spanned some portion of FY 2018.
211. In the FY 2023 IPPS/LTCH PPS final rule ( 87 FR 49042 ), we explained our belief that applying the scaling factor is appropriate for purposes of calculating Factor 3 for all hospitals, including new hospitals and hospitals that are treated as new hospitals, to improve consistency and predictability across all hospitals.
212. For example, if a hospital's FY 2018 cost report is determined to include potentially aberrant data, data from its FY 2019 cost report would be used for the ratio calculation.
213. For example, if a hospital does not have a FY 2020 cost report because the hospital's FY 2019 cost report spanned the FY 2020 time period, we will use the FY 2019 cost report that spanned the FY 2020 time period for this step. Using the same example, where the hospital's FY 2019 report is used for the FY 2020 time period, we will use the hospital's FY 2018 report if it spans some of the FY 2019 time period. We will not use the same cost report for both the FY 2020 and the FY 2019 time periods.
214. https://www.cms.gov/regulations-and-guidance/guidance/transmittals/2017downloads/r11p240.pdf .
215. For example, if the report does not reflect audit results due to MAC mishandling, or the most recent report differs from a previously accepted, amended report due to MAC mishandling.
216. https://www.cms.gov/oact/tr/2024 .
221. The M+C program in Part C of Medicare was renamed the Medicare Advantage (MA) Program under the Medicare Prescription Drug, Improvement, and Modernization Act of 2003 (MMA), which was enacted in December 2003.
222. Formerly Medicare+Choice.
223. https://www.fda.gov/news-events/expanded-access/expanded-access-keywords-definitions-and-resources .
224. 42 CFR 413.215(a) and 413.220 .
225. § 413.230.
226. Department of Health and Human Services, Review of Pharmaceuticals and Active Pharmaceutical Ingredients (pp. 207-250), June 2021: https://www.whitehouse.gov/wp-content/uploads/2021/06/100-day-supply-chain-review-report.pdf .
227. Department of Health and Human Services, National Strategy for a Resilient Public Health Supply Chain, July 2021: https://www.phe.gov/Preparedness/legal/Documents/National-Strategy-for-Resilient-Public-Health-Supply-Chain.pdf .
228. Senate Committee on Homeland Security & Governmental Affairs, Short Supply: The Health and National Security Risks of Drug Shortages, March 2023: https://www.hsgac.senate.gov/wp-content/uploads/2023-06-06-HSGAC-Majority-Draft-Drug-Shortages-Report.-FINAL-CORRECTED.pdf .
229. Vizient, Drug Shortages and Labor Costs: Measuring the Hidden Costs of Drug Shortages on U.S. Hospitals, June 2019: https://wieck-vizient-production.s3.us-west-1.amazonaws.com/page-Brum/attachment/c9dba646f40b9b5def8032480ea51e1e85194129 .
230. Department of Health and Human Services, National Strategy for a Resilient Public Health Supply Chain, July 2021: https://www.phe.gov/Preparedness/legal/Documents/National-Strategy-for-Resilient-Public-Health-Supply-Chain.pdf .
231. Vizient, Drug Shortages and Labor Costs: Measuring the Hidden Costs of Drug Shortages on U.S. Hospitals, June 2019: https://wieck-vizient-production.s3.us-west-1.amazonaws.com/page-Brum/attachment/c9dba646f40b9b5def8032480ea51e1e85194129 .
232. American Journal of Health System Pharmacology, National Survey on the Effect of Oncology Drug Shortages on Cancer Care, 2013: https://pubmed.ncbi.nlm.nih.gov/23515514/ .
233. JCO Oncology Practice, National Survey on the Effect of Oncology Drug Shortages in Clinical Practice, 2022: https://pubmed.ncbi.nlm.nih.gov/35544740/ .
234. Journal of the American Medical Association, Association between U.S. Norepinephrine Shortage and Mortality Among Patients with Septic Shock, 2017: https://pubmed.ncbi.nlm.nih.gov/28322415/ .
235. Clinical Infectious Diseases, The Effect of a Piperacillin/Tazobactam Shortage on Antimicrobial Prescribing and Clostridium difficile Risk in 88 US Medical Centers, 2017: https://pubmed.ncbi.nlm.nih.gov/28444166/ .
236. New England Journal of Medicine, The Impact of Drug Shortages on Children with Cancer: The Example of Mechlorethamine, 2012: https://pubmed.ncbi.nlm.nih.gov/23268661/ .
237. Senate Committee on Homeland Security & Governmental Affairs, Short Supply: The Health and National Security Risks of Drug Shortages, March 2023: https://www.hsgac.senate.gov/wp-content/uploads/2023-06-06-HSGAC-Majority-Draft-Drug-Shortages-Report.-FINAL-CORRECTED.pdf .
238. Department of Health and Human Services, ASPE Report to Congress: Impact of Drug Shortages on Consumer Costs, May 2023: https://aspe.hhs.gov/reports/drug-shortages-impacts-consumer-costs .
239. Department of Health and Human Services, Review of Pharmaceuticals and Active Pharmaceutical Ingredients (pp. 207-250), June 2021: https://www.whitehouse.gov/wp-content/uploads/2021/06/100-day-supply-chain-review-report.pdf .
240. Department of Health and Human Services, National Strategy for a Resilient Public Health Supply Chain, July 2021: https://www.phe.gov/Preparedness/legal/Documents/National-Strategy-for-Resilient-Public-Health-Supply-Chain.pdf .
241. https://www.accessdata.fda.gov/scripts/drugshortages/default.cfm
242. https://www.fda.gov/about-fda/reports/executive-order-13944-list-essential-medicines-medical-countermeasures-and-critical-inputs
243. U.S. Congress, U.S. House of Representatives Committee on Ways and Means, Subcommittee on Health, Health Care Consolidation: The Changing Landscape of the U.S. Health Care System, May 2023: https://www.rand.org/content/dam/rand/pubs/testimonies/CTA2700/CTA2770-1/RAND_CTA2770-1.pdf .
244. American Hospital Association, Rural Hospital Closures Threaten Access: Solutions to Preserve Care in Local Communities, September 2022: https://www.aha.org/system/files/media/file/2022/09/rural-hospital-closures-threaten-access-report.pdf .
245. The White House, The Biden-Harris Administration is taking actions to improve the health of rural communities and help rural health care providers stay open, November 2023: https://www.hhs.gov/about/news/2023/11/03/department-health-human-services-actions-support-rural-america-rural-health-care-providers.html .
246. The White House, Fact Sheet: Biden Administration Takes Steps to Address Covid-19 in Rural America and Build Rural Health Back Better, August 2021: https://www.whitehouse.gov/briefing-room/statements-releases/2021/08/13/fact-sheet-biden-administration-takes-steps-to-address-covid-19-in-rural-america-and-build-rural-health-back-better/ .
247. https://www.fda.gov/drugs/drug-safety-and-availability/drug-shortages
248. https://www.hhs.gov/about/news/2023/11/27/biden-harris-administration-announces-actions-bolster-medical-supply-chain.html
249. We remind hospitals that the costs of establishing and maintaining a buffer stock of an essential medicine do not include the cost of the essential medicine itself, meaning that the cost of compounding would not be included in the cost for establishing and maintaining a buffer stock of an essential medicine.
250. https://www.ashp.org/drug-shortages/current-shortages/fda-and-ashp-shortage-parameters?loginreturnUrl=SSOCheckOnly .
251. https://newsroom.vizientinc.com/en-US/releases/blogs-the-source-of-truth-a-pharmacy-buyers-drug-shortage-list .
252. https://aspe.hhs.gov/reports/global-drug-shortages .
253. https://www.fda.gov/drugs/drug-shortages/frequently-asked-questions-about-drug-shortages .
254. https://www.fda.gov/drugs/drug-shortages/frequently-asked-questions-about-drug-shortages .
255. https://www.fda.gov/drugs/drug-safety-and-availability/drug-shortages .
256. http://www.bea.gov/papers/pdf/IOmanual_092906.pdf .
257. The 65 percent is based on a survey conducted by CMS in 2008 as detailed in the FY 2010 IPPS/LTCH PPS final rule ( 74 FR 43850 through 43856 ). This was also used to determine the Professional Fees: Labor-related cost weight in the 2017-based LTCH market basket.
258. Institute of Medicine (US) Committee on Quality of Health Care in America, Kohn, L.T., Corrigan, J.M., & Donaldson, M.S. (Eds.). (2000). To Err is Human: Building a Safer Health System. National Academies Press (US).
259. Institute of Medicine (US) Committee on Quality of Health Care in America. (2001). Crossing the Quality Chasm: A New Health System for the 21st Century. National Academies Press (US).
260. Agency for Healthcare Research and Quality. (February 2021). National Healthcare Quality and Disparities Report chartbook on patient safety. Rockville, MD. Available at: https://www.ahrq.gov/sites/default/files/wysiwyg/research/findings/nhqrdr/chartbooks/patientsafety/2019qdr-patient-safety-chartbook.pdf .
261. Rodwin BA, Bilan VP, Merchant NB, Steffens CG, Grimshaw AA, Bastian LA, Gunderson CG. Rate of Preventable Mortality in Hospitalized Patients: a Systematic Review and Meta-analysis. J Gen Intern Med. 2020 Jul;35(7):2099-2106. doi: 10.1007/s11606-019-05592-5. Epub 2020 Jan 21. PMID: 31965525; PMCID: PMC7351940.
262. Bates DW, Levine DM, Salmasian H, et al. The Safety of Inpatient Health Care. New England Journal of Medicine. 2023;388(2):142-153. https://doi.org/10.1056/nejmsa2206117 .
263. Lastinger LM, Alvarez CR, Kofman A, Konnor RY, Kuhar DT, Nkwata A, Patel PR, Pattabiraman V, Xu SY, Dudeck MA. Continued increases in the incidence of healthcare-associated infection (HAI) during the second year of the coronavirus disease 2019 (COVID-19) pandemic. Infect Control Hosp Epidemiol. 2023 Jun;44(6):997-1001. doi: 10.1017/ice.2022.116. Epub 2022 May 20. PMID: 35591782; PMCID: PMC9237489.
264. Patel, PR, Weiner-Lastinger, LM, Dudeck, MA, et al. Impact of COVID-19 pandemic on central-line-associated bloodstream infections during the early months of 2020, National Healthcare Safety Network. Infect Control Hosp Epidemiol 2021. doi: 10.1017/ice.2021.108.
265. Baker MA, Sands KE, Huang SS, Kleinman K, Septimus EJ, Varma N, Blanchard J, Poland RE, Coady MH, Yokoe DS, Fraker S, Froman A, Moody J, Goldin L, Isaacs A, Kleja K, Korwek KM, Stelling J, Clark A, Platt R, Perlin JB; CDC Prevention Epicenters Program. The Impact of Coronavirus Disease 2019 (COVID-19) on Healthcare-Associated Infections. Clin Infect Dis. 2022 May 30;74(10):1748-1754. doi: 10.1093/cid/ciab688. PMID: 34370014; PMCID: PMC8385925.
266. Centers for Disease Control and Prevention. (2021). 2021 National and State Healthcare-Associated Infections Progress Report. Available at: https://www.cdc.gov/hai/data/archive/2021-HAI-progress-report.html#2018 .
267. Centers for Disease Control and Prevention. (2022). 2022 National and State Healthcare-Associated Infections Progress Report. Available at: https://www.cdc.gov/healthcare-associated-infections/php/data/progress-report.html?CDC_AAref_Val=https://www.cdc.gov/hai/data/portal/progress-report.html#cdc_report_pub_study_section_2-2022-hai-progress-report .
268. Agency for Healthcare Research and Quality. (2021). AHRQ PSNet Annual Perspective: Impact of the COVID-19 Pandemic on Patient Safety. https://psnet.ahrq.gov/perspective/ahrq-psnet-annual-perspective-impact-covid-19-pandemic-patient-safety .
269. Fleisher, L.A., Schreiber, M.D., Cardo, D., and Srinivasan, M.D. (2022). Health care safety during the pandemic and beyond—building a system that ensures resilience. N Engl J Med, 386: 609-611. https://www.nejm.org/doi/full/10.1056/NEJMp2118285 .
270. Implications of the COVID-19 pandemic for patient safety: a rapid review. Geneva: World Health Organization; 2022. Licence: CC BY-NC-SA 3.0 IGO.
271. Li, Z., Lin, F., Thalib, L., & Chaboyer, W. (2020). Global prevalence and incidence of pressure injuries in hospitalised adult patients: A systematic review and meta-analysis. International Journal of Nursing Studies, Vol. 105. https://doi.org/10.1016/j.ijnurstu.2020.103546 .
272. Dykes, P. C., Curtin-Bowen, M., Lipsitz, S., Franz, C., Adelman, J., Adkison, L., Bogaisky, M., Carroll, D., Carter, E., Herlihy, L., Lindros, M. E., Ryan, V., Scanlan, M., Walsh, M. A., Wien, M., & Bates, D. W. (2023). Cost of Inpatient Falls and Cost-Benefit Analysis of Implementation of an Evidence-Based Fall Prevention Program. JAMA Health Forum, 4 (1), e225125. https://doi.org/10.1001/jamahealthforum.2022.5125 .
273. Sabate S., Mazo V., Canet J. (2014). Predicting Postoperative Pulmonary Complications: Implications for Outcomes and Costs. Case Reports in Anesthesiology. 27(2), 201-209.
274. Rosen, A. K., Loveland, S., Shin, M., Shwartz, M., Hanchate, A., Chen, Q., Kaafarani, H. M., & Borzecki, A. (2013). Examining the impact of the AHRQ Patient Safety Indicators (PSIs) on the Veterans Health Administration: the case of readmissions. Medical Care, 51(1), 37-44.
275. Lawson E.H., Hall B.L., Louie R., et al. (2013). Association Between Occurrence of a Postoperative Complication and Readmission: Implications for Quality Improvement and Cost Savings. Annals of Surgery, 258(1),10-18.
276. Thongprayoon, C., Hansrivijit, P., Kovvuru, K., Kanduri, S. R., Torres-Ortiz, A., Acharya, P., Gonzalez-Suarez, M. L., Kaewput, W., Bathini, T., & Cheungpasitporn, W. (2020). Diagnostics, Risk Factors, Treatment and Outcomes of Acute Kidney Injury in a New Paradigm. Journal of clinical medicine, 9(4), 1104.
277. Hoste, E. A., & Schurgers, M. (2008). Epidemiology of acute kidney injury: how big is the problem? Critical care medicine, 36(4 Suppl), S146-S151.
278. AHRQ. (2023). National Action Alliance for Patient and Workforce Safety. https://www.ahrq.gov/cpi/about/otherwebsites/action-alliance.html .
279. AHRQ. (2023). National Action Alliance for Patient and Workforce Safety. https://www.ahrq.gov/cpi/about/otherwebsites/action-alliance.html .
280. President's Council of Advisors on Science and Technology. (2023). Report to the President: A Transformational Effort on Patient Safety. https://www.whitehouse.gov/wp-content/uploads/2023/09/PCAST_Patient-Safety-Report_Sept2023.pdf .
281. President's Council of Advisors on Science and Technology. (2023). Report to the President: A Transformational Effort on Patient Safety. https://www.whitehouse.gov/wp-content/uploads/2023/09/PCAST_Patient-Safety-Report_Sept2023.pdf .
282. Patient Safety Network. Systems Approach. Agency for Healthcare Research and Quality. Published September 7, 2019. https://psnet.ahrq.gov/primer/systems-approach .
283. National Patient Safety Foundation. Free from Harm: Accelerating Patient Safety Improvement Fifteen Years after To Err Is Human. Boston, MA: National Patient Safety Foundation; 2015.
284. Gandhi, T. K., Feeley, D., & Schummers, D. (2020b). Zero Harm in Health Care. NEJM Catalyst, 1(2). https://doi.org/10.1056/cat.19.1137 .
285. Pronovost, P. Transforming patient safety: A sector-wide systems approach. Published January 8, 2015.
286. Frankel A, Haraden C, Federico F, Lenoci-Edwards J. A Framework for Safe, Reliable, and Effective Care. White Paper. Cambridge, MA: Institute for Healthcare Improvement and Safe & Reliable Healthcare; 2017. (Available on https://www.ihi.org/resources/white-papers/framework-safe-reliable-and-effective-care ).
287. American College of Healthcare Executives and IHI/NPSF Lucian Leape Institute. Leading a Culture of Safety: A Blueprint for Success. Boston, MA: American College of Healthcare Executives and Institute for Healthcare Improvement; 2017.
288. National Steering Committee for Patient Safety. Safer Together: A National Action Plan to Advance Patient Safety. Boston, Massachusetts: Institute for Healthcare Improvement; 2020. (Available at www.ihi.org/SafetyActionPlan ).
289. National Steering Committee for Patient Safety. Safer Together: A National Action Plan to Advance Patient Safety. Boston, Massachusetts: Institute for Healthcare Improvement; 2020.
290. Centers for Medicare & Medicaid Services. (2023). CMS National Quality Strategy Handout. Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
291. Centers for Medicare & Medicaid Services. (2023). CMS National Quality Strategy Handout. Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
292. Centers for Medicare & Medicaid Services. Meaningful Measures Framework. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/meaningful-measures-20 .
293. Centers for Medicare & Medicaid Services. (2021). Meaningful Measures 2.0: Moving from Measure Reduction to Modernization. Available at: https://www.cms.gov/meaningful-measures-20-moving-measure-reduction-modernization . We note that Meaningful Measures 2.0 is still under development.
294. Centers for Medicare & Medicaid Services. (December 1, 2023). 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
295. Centers for Medicare & Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
296. Centers for Medicare & Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
297. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures . The draft Attestation Guide, version 1.0, was available at both: https://qualitynet.gov/inpatient/iqr/proposedmeasures and https://qualitynet.cms.gov/pch/pchqr/proposedmeasures at the time of the proposed rule. We note that examples provided in this guide are for illustrative purposes.
298. DiCuccio MH. The Relationship Between Patient Safety Culture and Patient Outcomes: A Systematic Review. J Patient Saf. 2015;11(3):135-42. doi:10.1097/PTS.0000000000000058.
299. Yale New Haven Health Services Corporation—Center for Outcomes Research and Evaluation. Summary of Technical Expert Panel (TEP) Meetings Patient Safety Structural Measure (PSSM). Available at: https://mmshub.cms.gov/sites/default/files/PSSM-TEP-Summary-Report-202306.pdf .
300. Ibid.
301. Ibid.
302. Battelle—Partnership for Quality Measurement. Compiled MUC List Public Comment Posting. Available at: https://p4qm.org/sites/default/files/2024-01/Compiled-MUC-List-Public-Comment-Posting.xlsx .
303. Battelle—Partnership for Quality Measurement. 2023 Measures Under Consideration Public Comment Summary Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-01/PRMR-Hospital-Public-Comments-Final-Summary.pdf .
304. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures . The draft Attestation Guide, version 1.0, was available at both: https://qualitynet.gov/inpatient/iqr/proposedmeasures and https://qualitynet.cms.gov/pch/pchqr/proposedmeasures at the time of the proposed rule. We note that examples provided in this guide are for illustrative purposes.
305. A “just culture” is defined by the Agency for Healthcare Research and Quality as a system that holds itself accountable, holds staff members accountable, and has staff members that hold themselves accountable. (The CUSP Method. https://www.ahrq.gov/hai/cusp/index.html .)
306. Agency for Healthcare Research and Quality. (2019, September 7). Root Cause Analysis. https://psnet.ahrq.gov/primer/root-cause-analysis .
307. Agency for Healthcare Research and Quality. Federally-Listed Patient Safety Organizations (PSOs). Retrieved January 5, 2024, from https://pso.ahrq.gov/pso/listed?f%5B0%5D=resources_provided%3A2 .
308. Agency for Healthcare Research and Quality. (2022). Communication and Optimal Resolution (CANDOR). https://www.ahrq.gov/patient-safety/settings/hospital/candor/index.html .
309. Yale New Haven Health Services Corporation—Center for Outcomes Research and Evaluation. Summary of Technical Expert Panel (TEP) Meetings Patient Safety Structural Measure (PSSM). Available at: https://mmshub.cms.gov/sites/default/files/PSSM-TEP-Summary-Report-202306.pdf .
310. Office of the National Coordinator for Health Information Technology (ONC). SAFER Guides. Available at https://www.healthit.gov/topic/safety/safer-guides .
311. AHRQ. Safer Together: A National Action Plan to Advance Patient Safety. Available at: https://www.ahrq.gov/patient-safety/reports/safer-together.html .
312. CMS. The CMS National Quality Strategy. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy .
313. Yale New Haven Health Services Corporation—Center for Outcomes Research and Evaluation. Summary of Technical Expert Panel (TEP) Meetings Patient Safety Structural Measure (PSSM). Available at: https://mmshub.cms.gov/sites/default/files/PSSM-TEP-Summary-Report-202306.pdf .
314. Yale New Haven Health Services Corporation—Center for Outcomes Research and Evaluation. Summary of Technical Expert Panel (TEP) Meetings Patient Safety Structural Measure (PSSM). Available at: https://mmshub.cms.gov/sites/default/files/PSSM-TEP-Summary-Report-202306.pdf .
315. Ponto J. Understanding and Evaluating Survey Research. J Adv Pract Oncol. 2015 Mar-Apr;6(2):168-71. Epub 2015 Mar 1. PMID: 26649250; PMCID: PMC4601897.
316. Ponto J. Understanding and Evaluating Survey Research. J Adv Pract Oncol. 2015 Mar-Apr;6(2):168-71. Epub 2015 Mar 1. PMID: 26649250; PMCID: PMC4601897.
317. DiCuccio MH. The Relationship Between Patient Safety Culture and Patient Outcomes: A Systematic Review. J Patient Saf. 2015;11(3):135- 42. doi:10.1097/PTS.0000000000000058.
318. Forbes J, Arrieta A Comparing hospital leadership and front-line workers' perceptions of patient safety culture: an unbalanced panel study BMJ Leader Published Online First: 03 April 2024. doi: 10.1136/leader-2023-000922.
319. Were hospitals already scoring highly on the measure there would be no benefit to adopting it. See 42 CFR 412.140(g)(3)(i) (providing for the removal of measures on which hospitals are performing “so high and unvarying that meaningful distinction and improvements in performance can no longer be made.”).
320. Lewis B. Success of Patient and Family Advisory Councils: The Importance of Metrics. J Patient Exp. 2023 Apr 10;10:23743735231167972. doi: 10.1177/23743735231167972. PMID: 37064819; PMCID: PMC10103250.
321. US Department of Health and Human Services; Office of the Inspector General, September 2019. OIG Report No. OEI-01-17-00420. https://oig.hhs.gov/oei/reports/oei-01-17-00420.asp .
322. For more information about the QIO Program we refer readers to: https://www.cms.gov/medicare/quality/quality-improvement-organizations#:~:text=QIO%20Program%20priorities%3A&text=Improve%20care%20coordination%20and%20the,Increase%20patient%20safety .
323. For AHRQ's patient safety resources, we refer readers to: https://www.ahrq.gov/patient-safety/resources/index.html .
324. Battelle—Partnership for Quality Measurement. Compiled MUC List Public Comment Posting. Available at: https://p4qm.org/sites/ default/files/2024-01/Compiled-MUC-List-Public-Comment-Posting.xlsx .
325. Battelle—Partnership for Quality Measurement. 2023 Measures Under Consideration Public Comment Summary Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-01/PRMR-Hospital-Public-Comments-Final-Summary.pdf .
326. DiCuccio MH. The Relationship Between Patient Safety Culture and Patient Outcomes: A Systematic Review. J Patient Saf. 2015;11(3):135- 42. doi:10.1097/PTS.0000000000000058.
327. Yale New Haven Health Services Corporation—Center for Outcomes Research and Evaluation. Summary of Technical Expert Panel (TEP) Meetings Patient Safety Structural Measure (PSSM). Available at: https://mmshub.cms.gov/sites/default/files/PSSM-TEP-Summary-Report-202306.pdf .
328. National Steering Committee for Patient Safety. Self-Assessment Tool: A National Action Plan to Advance Patient Safety. Boston, Massachusetts: Institute for Healthcare Improvement; 2020.
329. AHA. Sample Policy for Open Board Meetings. Available at: https://trustees.aha.org/sites/default/files/trustees/sample-policy-for-open-board-meetings-for-public-or-government-hospitals.pdf .
330. National Steering Committee for Patient Safety. Self-Assessment Tool: A National Action Plan to Advance Patient Safety. Boston, Massachusetts: Institute for Healthcare Improvement; 2020.
331. Center for Clinical Standards and Quality, Quality, Safety & Oversight Group. Revision to State Operations Manual (SOM), Hospital Appendix A—Interpretive Guidelines for 42 CFR 482.21 , Quality Assessment & Performance Improvement (QAPI) Program.
332. Centers for Medicare and Medicaid Services. CMS National Quality Strategy. 2024. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy .
333. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures .
334. AHRQ. Quality Indicators. Available at: https://qualityindicators.ahrq.gov/Downloads/Modules/V2023/AHRQ_QI_Indicators_List.pdf .
335. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures .
336. Gallo, A. What Is Psychological Safety. Harvard Business Review. Available at: https://hbr.org/2023/02/what-is-psychological-safety .
337. AHRQ. The CUSP Method. Available at: https://www.ahrq.gov/hai/cusp/index.html .
338. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures .
339. Agency for Healthcare Research and Quality. The Comprehensive Unitbased Safety Program. 2019. Accessed on 10/24/2023. https://www.ahrq.gov/hai/cusp/index.html
340. Agency for Healthcare Research and Quality. Communication and Optimal Resolution Toolkit. 2022 Accessed on 10/24/2023. https://www.ahrq.gov/patient-safety/settings/hospital/candor/modules.html .
341. Centers for Disease Control and Prevention. Infection Control Assessment and Response Tool for General Infection Prevention and Control. Accessed on 10/24/2023. https://www.cdc.gov/healthcare-associated-infections/?CDC_AAref_Val=https://www.cdc.gov/hai/prevent/infection%25C2%25ADcontrol%25C2%25ADassessment%25C2%25ADtools.html .
342. Agency for Healthcare Research and Quality. Team Strategies & Tools to Enhance Performance and Patient Safety. 2023.Accessed on 10/24/2023. https://www.ahrq.gov/teamstepps-program/index.html .
343. Institute for Healthcare Improvement. Root Cause Analyses and Actions to Prevent Harm. 2019. Accessed on 10/24/2023. https://www.ihi.org/resources/tools/rca2-improving-root-cause-analyses-and-actions-prevent-harm .
344. Agency for Healthcare Research and Quality. The SHARE Approach. 2014. Accessed on 10/24/2023. https://www.ahrq.gov/health-literacy/professional-training/shared-decision/index.html .
345. Agency for Healthcare Research and Quality. Tools for Reducing Central Line-Associated Blood Stream Infections. 2023. Accessed on 10/24/2023. https://www.ahrq.gov/hai/clabsi-tools/index.html .
346. Institute for Healthcare Improvement. PlanDoStudyAct Worksheet. 2023. Accessed on 10/24/2023. https://www.ihi.org/resources/tools/plan-do-study-act-pdsa-worksheet .
347. AHRQ. Safer Together: A National Action Plan to Advance Patient Safety. Available at: https://www.ahrq.gov/patient-safety/reports/safer-together.html .
348. Bethune RM, Ball S, Doran N, Harris M, Medina-Lara A, Fornasiero M, Hill M, Lang I, McGregor-Harper J, Sheaff R. How Safety Culture Surveys Influence the Quality and Safety of Healthcare Organisations. Cureus. 2023 Sep 3;15(9):e44603. doi: 10.7759/cureus.44603. PMID: 37795070; PMCID: PMC10546949.
349. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures .
350. AHRQ. Hospital Survey on Patient Safety Culture. Topics Covered by the SOPS Hospital Survey 2.0. Available at: https://www.ahrq.gov/sops/surveys/hospital/index.html .
351. Safety Attitudes: Frontline Perspectives from this Patient Care Area. Available at: https://www.uth.edu/chqs/assets/docs/saq-short-form.pdf .
352. AHRQ. PSNet. Glossary: Patient Safety. Available at: https://psnet.ahrq.gov/glossary-0?f%5B0%5D=glossary_az_content_title%3AP .
353. AHRQ. PSNet. Glossary: Patient Safety. Available at: https://psnet.ahrq.gov/glossary-0?f%5B0%5D=glossary_az_content_title%3AP .
354. AHRQ. PSNet. Glossary: High Reliability Organization. Available at: https://psnet.ahrq.gov/glossary-0?f%5B0%5D=glossary_az_content_title%3AP .
355. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures . https://qualitynet.cms.gov/pch/measures .
356. Agency for Healthcare Research and Quality. Federally-Listed Patient Safety Organizations (PSOs). Retrieved January 5, 2024, from https://pso.ahrq.gov/pso/listed?f%5B0%5D=resources_provided%3A2 .
357. Gallo, A. What Is Psychological Safety. Harvard Business Review. Available at: https://hbr.org/2023/02/what-is-psychological-safety .
358. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures .
359. Agency for Healthcare Research and Quality. Federally-Listed Patient Safety Organizations (PSOs). Retrieved January 5, 2024, from https://pso.ahrq.gov/pso/listed?f%5B0%5D=resources_provided%3A2 .
360. AHRQ's published list of PSOs is available at: https://pso.ahrq.gov/pso/listed .
361. For additional information on the PSO Privacy Protection Center, we refer readers to https://www.psoppc.org/psoppc_web/ .
362. https://www.ntsb.gov/safety/Pages/default.aspx .
363. Strategies to Improve Patient Safety: Final Report to Congress Required by the Patient Safety and Quality Improvement Act of 2005. Rockville, MD: Agency for Healthcare Research and Quality; December 2021. AHRQ Publication No. 22-0009. https://pso.ahrq.gov/sites/default/files/wysiwyg/strategies-improve-patient-safety-final.pdf .
364. Agency for Healthcare Research and Quality. Federally-Listed Patient Safety Organizations (PSOs). Retrieved January 5, 2024, from https://pso.ahrq.gov/pso/listed?f%5B0%5D=resources_provided%3A2 .
365. Office of the Inspector General. Patient Safety Organizations: Hospital Participation, Value, and Challenges. September 2019. Available at: https://oig.hhs.gov/oei/reports/oei-01-17-00420.pdf .
366. Yale New Haven Health Services Corporation—Center for Outcomes Research and Evaluation. Summary of Technical Expert Panel (TEP) Meetings Patient Safety Structural Measure (PSSM). Available at: https://mmshub.cms.gov/sites/default/files/PSSMTEP-Summary-Report-202306.pdf .
367. Patient Safety Network. Systems Approach. Agency for Healthcare Research and Quality. Published September 7, 2019. https://psnet.ahrq.gov/primer/systems-approach .
368. National Patient Safety Foundation. Free from Harm: Accelerating Patient Safety Improvement Fifteen Years after To Err Is Human. Boston, MA: National Patient Safety Foundation; 2015.
369. Gandhi, T.K., Feeley, D., & Schummers, D. (2020b). Zero Harm in Health Care. NEJM Catalyst, 1(2). https://doi.org/10.1056/cat.19.1137 .
370. Pronovost, P. Transforming patient safety: A sector-wide systems approach. Published January 8, 2015.
371. Frankel A, Haraden C, Federico F, Lenoci-Edwards J. A Framework for Safe, Reliable, and Effective Care. White Paper. Cambridge, MA: Institute for Healthcare Improvement and Safe & Reliable Healthcare; 2017. (Available on https://www.ihi.org/resources/white-papers/framework-safe-reliable-and-effective-care ).
372. American College of Healthcare Executives and IHI/NPSF Lucian Leape Institute. Leading a Culture of Safety: A Blueprint for Success. Boston, MA: American College of Healthcare Executives and Institute for Healthcare Improvement; 2017.
373. National Steering Committee for Patient Safety. Safer Together: A National Action Plan to Advance Patient Safety. Boston, Massachusetts: Institute for Healthcare Improvement; 2020. (Available at www.ihi.org/SafetyActionPlan ).
374. Office of the Inspector General. Patient Safety Organizations: Hospital Participation, Value, and Challenges. September 2019. Available at: https://oig.hhs.gov/oei/reports/oei-01-17-00420.pdf .
375. Office of the Inspector General. Patient Safety Organizations: Hospital Participation, Value, and Challenges. September 2019. Available at: https://oig.hhs.gov/oei/reports/oei-01-17-00420.pdf .
376. Strategies to Improve Patient Safety: Final Report to Congress Required by the Patient Safety and Quality Improvement Act of 2005. Rockville, MD: Agency for Healthcare Research and Quality; December 2021. AHRQ Publication No. 22-0009. https://pso.ahrq.gov/sites/default/files/wysiwyg/strategies-improve-patient-safety-final.pdf .
377. For additional information on the PSO Privacy Protection Center, we refer readers to https://www.psoppc.org/psoppc_web/ .
378. 42 U.S.C. 299b-21(5)(B) , 299b-24(a)(1)(A) and (2)(A) .
379. Strategies to Improve Patient Safety: Final Report to Congress Required by the Patient Safety and Quality Improvement Act of 2005. Rockville, MD: Agency for Healthcare Research and Quality; December 2021. AHRQ Publication No. 22-0009. https://pso.ahrq.gov/sites/default/files/wysiwyg/strategies-improve-patient-safety-final.pdf .
380. AHRQ. AHRQ Common Formats for Event Reporting—Hospital Version 2.0a. Available at: https://www.psoppc.org/psoppc_web/DLMS/downloadDocument?groupId=1410&pageName=common%20formats%20Hospital%20V2.0a . Accessed on June 5, 2024.
381. The CMS Proposed Patient Safety Structural Measure (PSSM). A Patients for Patient Safety US Advocacy Event. December 6, 2023. Slides Available at: https://irp.cdn-website.com/812f414d/files/uploaded/PSSM%20PFPS%20US%20webinar%20120623.pdf
382. CMS National Quality Strategy. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy . For specific information about our goals regarding patient safety, see the Safety Goal, “Achieve Zero Preventable Harm” Under the “Ensure Safe and Resilient Health Care Systems” domain.
383. CMS. Quality in Motion: Acting on the CMS National Quality Strategy: April 2024. Available at https://www.cms.gov/files/document/quality-motion-cms-national-quality-strategy.pdf .
384. Office of the Inspector General. Patient Safety Organizations: Hospital Participation, Value, and Challenges. September 2019. Available at: https://oig.hhs.gov/oei/reports/oei-01-17-00420.pdf .
385. https://pso.ahrq.gov/common-formats .
386. https://pso.ahrq.gov/sites/default/files/wysiwyg/pso-brochure.pdf .
387. Guide to Patient and Family Engagement in Hospital Quality and Safety. Content last reviewed March 2023. Agency for Healthcare Research and Quality, Rockville, MD. https://www.ahrq.gov/patient-safety/patients-families/engagingfamilies/index.html .
388. National Patient Safety Foundation's Lucian Leape Institute. Safety Is Personal: Partnering with Patients and Families for the Safest Care. Boston: National Patient Safety Foundation; 2014.
389. Johnson B, Abraham M, Conway J, et al. Bethesda, MD: Institute for Family-Centered Care; April 2008.
390. National Steering Committee for Patient Safety. Self-Assessment Tool: A National Action Plan to Advance Patient Safety. Boston, Massachusetts: Institute for Healthcare Improvement; 2020.
391. Guide to Patient and Family Engagement in Hospital Quality and Safety. Content last reviewed March 2023. Agency for Healthcare Research and Quality, Rockville, MD. https://www.ahrq.gov/patient-safety/patients-families/engagingfamilies/index.html .
392. National Patient Safety Foundation's Lucian Leape Institute. Safety Is Personal: Partnering with Patients and Families for the Safest Care. Boston: National Patient Safety Foundation; 2014.
393. Johnson B, Abraham M, Conway J, et al. Bethesda, MD: Institute for Family-Centered Care; April 2008.
394. Centers for Medicare & Medicaid Services, Patient Safety Structural Measure Attestation Guide, available at: https://qualitynet.cms.gov/inpatient/iqr/measures and https://qualitynet.cms.gov/pch/measures . The draft Attestation Guide, version 1.0, was available at both: https://qualitynet.gov/inpatient/iqr/proposedmeasures and https://qualitynet.cms.gov/pch/pchqr/proposedmeasures at the time of the proposed rule. We note that examples provided in this guide are for illustrative purposes.
395. A “just culture” is defined by the Agency for Healthcare Research and Quality as a system that holds itself accountable, holds staff members accountable, and has staff members that hold themselves accountable. (The CUSP Method. https://www.ahrq.gov/hai/cusp/index.html .)
396. Agency for Healthcare Research and Quality. (2019, September 7). Root Cause Analysis. https://psnet.ahrq.gov/primer/root-cause-analysis .
397. Agency for Healthcare Research and Quality. Federally-Listed Patient Safety Organizations (PSOs). Retrieved January 5, 2024, from https://pso.ahrq.gov/pso/listed?f%5B0%5D=resources_provided%3A2 .
398. Agency for Healthcare Research and Quality. (2022). Communication and Optimal Resolution (CANDOR). https://www.ahrq.gov/patient-safety/settings/hospital/candor/index.html .
399. Agency for Healthcare Research and Quality. (2023) 2023 National Healthcare Quality and Disparities Report. Available at: https://www.ahrq.gov/research/findings/nhqrdr/nhqdr23/index.html .
400. Centers for Medicare & Medicaid Services (2023) Aligning Quality Measures Across CMS—the Universal Foundation. Available at: https://www.cms.gov/aligning-quality-measures-across-cms-universal-foundation .
401. Centers for Medicare and Medicaid Services. (2024) CMS National Quality Strategy. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy .
402. Battelle—Partnership for Quality Measurement. (2024). Pre-Rulemaking Measure Review Measures Under Consideration 2023 Recommendations Report. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-2023-MUC-Recommendations-Report-Final.pdf .
403. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
404. Battelle—Partnership for Quality Measurement. HCAHPS (Hospital Consumer Assessment of Healthcare Providers and Systems) Survey. Available at: https://p4qm.org/measures/0166 .
405. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
406. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
407. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
408. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
409. Elliott, M.N., Zaslavsky, A.M., Goldstein, E. et al. (2009) Effects of Survey Mode, Patient Mix, and Nonresponse on CAHPS Hospital Survey Scores. Health Services Research. 44: 501-518. https://doi.org/10.1111/j.1475-6773.2008.00914.x .
410. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
411. HCAHPS Online. (2021) HCAHPS Mode Experiment Survey Instrument. Available at: https://hcahpsonline.org/globalassets/hcahps/whats-new/mode-experiment/hcahps-mode-experiment-survey-instrument.pdf .
412. https://hcahpsonline.org/en/hcahps-star-ratings/#TechNotes .
413. Crofton C, Darby C, Farquhar M, Clancy CM. (2005) The CAHPS Hospital Survey: development, testing, and use. Jt Comm J Qual Patient Saf. 31(11):655-9, 601. doi: 10.1016/s1553-7250(05)31084-1. PMID: 16335067.
414. Giordano LA, Elliott MN, Goldstein E, Lehrman WG, and Spencer PA. (2010) Development, Implementation, and Public Reporting of the HCAHPS Survey. Medical Care Research and Review, 67 (1): 27-37. https:// journals.sagepub.com/doi/10.1177/1077558709341065.
415. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
416. Burkhart Q, Orr N, Brown JA, et al. (2021) Associations of Mail Survey Length and Layout with Response Rates Medical Care Research and Review 78(4): 441-448. DOI: https://doi.org/10.1177/1077558719888407 .
417. Beckett MK, Elliott MN, Gaillot S, et al. (2016) Establishing limits for supplemental items on a standardized national survey. Public Opinion Quarterly 80(4): 964-976 DOI: https://doi.org/10.1093/poq/nfw028 .
418. Elliott MN, Beckett MK, Cohea CW, et al. (2023) Changes in Patient Experiences of Hospital Care During the COVID-19 Pandemic. JAMA Health Forum 2023;4(8):e232766. DOI: https://doi.org/10.1001/jamahealthforum.2023.2766 .
419. Anhang-Price R, Elliott MN, et al. (2014) Examining the Role of Patient Experience Surveys in Measuring Health Care Quality. Medical Care Research and Review 71(5):522-54.-DOI: https://doi.org/10.1177/1077558714541480 .
420. Anhang Price R, Elliott MN, Cleary PD, Zaslavsky AM, Hays RD. (2015) Should Health Care Providers be Accountable for Patients' Care Experiences?. Journal of General Internal Medicine 30(2): 253-6. DOI: https://doi.org/10.1007/s11606-014-3111-7 .
421. Crofton C, Darby C, Farquhar M, Clancy CM. (2005) The CAHPS Hospital Survey: development, testing, and use. Jt Comm J Qual Patient Saf. 31(11):655-9, 601. doi: 10.1016/s1553-7250(05)31084-1. PMID: 16335067.
422. Giordano LA, Elliott MN, Goldstein E, Lehrman WG, and Spencer PA. (2010) Development, Implementation, and Public Reporting of the HCAHPS Survey. Medical Care Research and Review, 67 (1): 27-37. https://journals.sagepub.com/doi/10.1177/1077558709341065 .
423. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
424. HCAHPS Quality Assurance Guidelines, V18.0. Available at: https://hcahpsonline.org/en/quality-assurance/ .
425. Adams C, Walpola R, Schembri AM, Harrison R. (2022) The ultimate question? Evaluating the use of Net Promoter Score in healthcare: A systematic review. Health Expect. (5):2328-2339. doi: 10.1111/hex.13577.
426. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
427. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
428. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
429. Elliott MN, Brown JA, Hambarsoomian K, et al. (2024) Survey Protocols, Response Rates, and Representation of Underserved Patients: A Randomized Clinical Trial. JAMA Health Forum. 5(1):e234929. doi: 10.1001/jamahealthforum.2023.4929.
430. HCAHPS Online. (2022) “Improving Representativeness of the HCAHPS Survey” podcast. Available at: https://hcahpsonline.org/en/podcasts/#ImprovingRepresentativeness .
431. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
432. Ibid.
433. Stewart NH, Arora VM. (2022) Sleep in Hospitalized Older Adults. Sleep Med Clin. 17(2):223-232. doi: 10.1016/j.jsmc.2022.02.002. Epub 2022 Apr 22. PMID: 35659075.
434. Hedges C, Hunt C, Ball P. (2019) Quiet Time Improves the Patient Experience. J Nurs Care Qual. 34(3):197-202. doi: 10.1097/NCQ.0000000000000363. PMID: 30198951.
435. Battelle—Partnership for Quality Measurement. (2023). 2023 Pre-Rulemaking Measure Review (PRMR) Preliminary Assessment Report: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2023-12/PRMR-Hospital-Committee-PA-Final-Report.pdf .
436. HCAHPS Online. (2023) Patient Mix Adjustment. Available at: https://hcahpsonline.org/en/mode--patient-mix-adj/#jan2023publiclyreported .
437. Medicare Hospital Quality Chartbook. National Rates over Time. Available at: https://www.cmshospitalchartbook.com/visualization/national-rates-over-time . Accessed March 12, 2024.
438. Nuckols TK, Fingar KR, Barrett ML, et al. Returns to Emergency Department, Observation, or Inpatient Care Within 30 Days After Hospitalization in 4 States, 2009 and 2010 Versus 2013 and 2014. J Hosp Med. 2018;13(5):296-303.
439. Shammas NW, Kelly R, Lemke J, et al. Assessment of Time to Hospital Encounter after an Initial Hospitalization for Heart Failure: Results from a Tertiary Medical Center. Cardiol Res Pract. 2018; 2018:6087367.
440. Sabbatini AK, Joynt-Maddox KE, Liao JM, et al. Accounting for the growth of observation stays in the assessment of Medicare's hospital readmissions reduction program. JAMA Netw Open. 2022;5(11):e2242587.
441. Sabbatini AK, Wright B. Excluding observation stays from readmission rates—what quality measures are missing. New Engl J Med. 2018;378(22):2062-2065.
442. Wadhera RK, Joynt Maddox KE, Kazi DS, Shen C, Yeh RW. Hospital revisits within 30 days after discharge for medical conditions targeted by the Hospital Readmissions Reduction Program in the United States: national retrospective analysis. BMJ. 2019;366: l4563.
443. Observation care is a well-defined set of specific, clinically appropriate services, which include ongoing short-term treatment, assessment, and reassessment before a decision can be made regarding whether patients will require further treatment as hospital inpatients or if they are able to be discharged from the hospital. Observation services are commonly ordered for patients who present to the emergency department and who then require a significant period of treatment or monitoring in order to make a decision concerning their admission or discharge. See additional explanation here: https://www.cms.gov/Regulations-and-Guidance/Guidance/Manuals/Downloads/bp102c06.pdf .
444. Goldstein, J.N., Schwartz, J.S., McGraw, P. et al. “Implications of cost-sharing for observation care among Medicare beneficiaries: a pilot survey”. BMC Health Serv Res 19, 149 (2019). https://doi.org/10.1186/s12913-019-3982-8 .
445. Kripalani S, Theobald CN, Anctil B, Vasilevskis EE. Reducing hospital readmission rates: current strategies and future directions. Annu Rev Med. 2014;65:471-85. doi: 10.1146/annurev-med-022613-090415. Epub 2013 Oct 21.
446. Hoyer EH, Brotman DJ, Apfel A, Leung C, Boonyasai RT, Richardson M, Lepley D, Deutschendorf A. Improving Outcomes After Hospitalization: A Prospective Observational Multicenter Evaluation of Care Coordination Strategies for Reducing 30-Day Readmissions to Maryland Hospitals. J Gen Intern Med. 2018 May; 33(5): 621-627. Published online 2017 Nov 27. doi: 10.1007/s11606-017-4218-4.
447. Kripalani S, Chen G, Ciampa P, Theobald C, Cao A, McBride M, Dittus RS, Speroff T. A Transition Care Coordinator Model Reduces Hospital Readmissions and Costs. Contemp Clin Trials. 2019 Jun; 81: 55-61.
Published online 2019 Apr 25. doi: 10.1016/j.cct.2019.04.014.
448. Centers for Medicare Medicaid Services. 2023 MUC List. Available at: https://mmshub.cms.gov/measure-lifecycle/measure-implementation/pre-rulemaking/lists-and-reports .
449. Z. Austrian, SE Alexander, M.C. Piazza, C. Clouse. Mission, vision, and capacity of place-based safety net hospitals: Leveraging the power of anchors to strengthen local economies and communities. Journal of Community Practice, 23 (3-4) (2015), pp. 348-366. 10.1080/10705422.2015.1091416.
450. Franz B, Skinner D, Kerr AM, Penfold R, Kelleher K. Hospital-Community Partnerships: Facilitating Communication for Population Health on Columbus' South Side. Health Commun. 2018 Dec;33(12):1462-1474. doi: 10.1080/10410236.2017.1359033. Epub 2017 Aug 29. PMID: 28850263.
451. H.K. Koh, A. Bantham, A.C. Geller, M.A. Rukavina, K.M. Emmons, P. Yatsko, et al. Anchor institutions: Best practices to address social needs and social determinants of health. American Journal of Public Health, 110 (3) (2020), pp. 309-316, 10.2105/AJPH.2019.305472.
452. Douglas et al., Aligning Quality Measures across CMS—The Universal Foundation, N Engl J Med 2023;388:776-779 DOI: 10.1056/NEJMp2215539, VOL. 388 NO. 9.
453. Centers for Medicare Medicaid Services. (2022). What is the National Quality Strategy? Available at: https://www.cms.gov/Medicare/Quality-Initiatives-Patient-Assessment-Instruments/Value-Based-Programs/CMS-Quality-Strategy .
454. See section 1890A(a)(2) of the Social Security Act ( 42 U.S.C. 1395aaa-1(a)(2) ).
455. Centers for Disease Control and Prevention. (September 2022). Promoting Health for Older Adults. Retrieved from: https://www.cdc.gov/chronic-disease/?CDC_AAref_Val=https://www.cdc.gov/chronicdisease/resources/publications/factsheets/promoting-health-for-older-adults.htm .
456. Vespa, J., Armstrong, D.M., Medina, L. (Rev Feb 2020). Demographic turning points for the United States: Population projections for 2020 to 2060. Washington, DC: U.S. Department of Commerce, Economics and Statistics Administration, U.S. Census Bureau.
457. Quiñones, A.R., Markwardt, S., Botoseneanu, A. (2016). Multimorbidity combinations and disability in older adults. Journals of Gerontology Series A: Biomedical Sciences and Medical Sciences, 71(6), 823-830.
458. Centers for Disease Control and Prevention. (September 2022). Promoting Health for Older Adults. Retrieved from: https://www.cdc.gov/chronicdisease/resources/publications/factsheets/promoting-health-for-older-adults.htm .
459. Centers for Disease Control and Prevention. (September 2022). Promoting Health for Older Adults. Retrieved from: https://www.cdc.gov/chronic-disease/?CDC_AAref_Val=https://www.cdc.gov/chronicdisease/resources/publications/factsheets/promoting-health-for-older-adults.htm .
460. Boyd, C., Smith, C. D., Masoudi, F. A., Blaum, C. S., Dodson, J. A., Green, A. R., ... Tinetti, M. E. (2019). Decision making for older adults with multiple chronic conditions: executive summary for the American Geriatrics Society guiding principles on the care of older adults with multimorbidity. Journal of the American Geriatrics Society, 67(4), 665-673.
461. Lochner KA, Cox CS. Prevalence of Multiple Chronic Conditions Among Medicare Beneficiaries, United States, 2010. Prev Chronic Dis 2013;10:120137. DOI: http://dx.doi.org/10.5888/pcd10.120137 .
462. Salive, M. E. (2013). Multimorbidity in older adults. Epidemiologic reviews, 35(1), 75-83.
463. American Geriatrics Society Expert Panel on the Care of Older Adults with Multimorbidity. (2012). Guiding principles for the care of older adults with multimorbidity: an approach for clinicians. Journal of the American Geriatrics Society, 60 (10), E1-E25.
464. Boyd, C., Smith, C. D., Masoudi, F. A., Blaum, C. S., Dodson, J. A., Green, A. R., ... Tinetti, M. E. (2019). Decision making for older adults with multiple chronic conditions: executive summary for the American Geriatrics Society guiding principles on the care of older adults with multimorbidity. Journal of the American Geriatrics Society, 67(4), 665-673.
465. Tinetti, M. (January 2019). [Blog] How focusing on What Matters simplifies complex care for older adults. Institute for Healthcare Improvement. Available at: https://www.ihi.org/insights/how-focusing-what-matters-simplifies-complex-care-older-adults .
466. Institute for Healthcare Improvement. (2022). Age-friendly health systems: Guide to using the 4Ms in the care of older adults in hospitals and ambulatory practices. Available at: https://forms.ihi.org/hubfs/IHIAgeFriendlyHealthSystems_GuidetoUsing4MsCare.pdf .
467. Ibid.
468. Ibid.
469. Institute for Healthcare Improvement. (2022). Age-friendly health systems: Guide to using the 4Ms in the care of older adults in hospitals and ambulatory practices. Available at: https://forms.ihi.org/hubfs/IHIAgeFriendlyHealthSystems_GuidetoUsing4MsCare.pdf .
470. Institute for Healthcare Improvement. (2022). Age-friendly health systems: Guide to using the 4Ms in the care of older adults in hospitals and ambulatory practices. Available at: https://forms.ihi.org/hubfs/IHIAgeFriendlyHealthSystems_GuidetoUsing4MsCare.pdf .
471. Centers for Medicare Medicaid Services. 2022 MUC List. Available at: https://mmshub.cms.gov/measure-lifecycle/measure-implementation/pre-rulemaking/lists-and-reports .
472. Centers for Medicare Medicaid Services. MAP 2022-2023 Final Recommendations. Available at: https://mmshub.cms.gov/measure-lifecycle/measure-implementation/pre-rulemaking/lists-and-reports .
473. Centers for Medicare Medicaid Services. (2023). CMS National Quality Strategy. Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
474. Ibid.
475. Centers for Medicare Medicaid Services. (December 1, 2023). 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
476. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
477. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Final MUC Recommendation Report. Available at: https://p4qm.org/PRMR .
478. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Final MUC Recommendation Report. Available at: https://p4qm.org/PRMR .
479. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Final MUC Recommendation Report. Available at: https://p4qm.org/PRMR .
480. Siddiqi N, Harrison JK, Clegg A, et al. Interventions for preventing delirium in hospitalised non-ICU patients. Cochrane Database Syst Rev. Mar 11 2016;3:CD005563. doi:10.1002/14651858.CD005563.pub3.
481. Inouye SK, Bogardus ST, Jr., Charpentier PA, et al. A multicomponent intervention to prevent delirium in hospitalized older patients. N Engl J Med. Mar 4 1999;340(9):669-76. doi:10.1056/NEJM199903043400901.
482. Park, C., et al. (2022). Association Between Implementation of a Geriatric Trauma Clinical Pathway and Changes in Rates of Delirium in Older Adults With Traumatic Injury. JAMA Surg 157(8): 676-683.
483. Inouye SK, Bogardus ST, Jr., Charpentier PA, et al. A multicomponent intervention to prevent delirium in hospitalized older patients. N Engl J Med. Mar 4 1999;340(9):669-76. doi:10.1056/NEJM199903043400901.
484. Barnes DE, Palmer RM, Kresevic DM, et al. Acute care for elders units produced shorter hospital stays at lower cost while maintaining patients' functional status. Health Aff (Millwood). Jun 2012;31(6):1227-36. doi:10.1377/hlthaff.2012.0142.
485. Landefeld CS, Palmer RM, Kresevic DM, Fortinsky RH, Kowal J. A randomized trial of care in a hospital medical unit especially designed to improve the functional outcomes of acutely ill older patients. N Engl J Med. May 18 1995;332(20):1338-44. doi:10.1056/NEJM199505183322006.
486. Inouye SK, Bogardus ST, Jr., Baker DI, Leo-Summers L, Cooney LM, Jr. The Hospital Elder Life Program: a model of care to prevent cognitive and functional decline in older hospitalized patients. Hospital Elder Life Program. J Am Geriatr Soc. Dec 2000;48(12):1697-706. doi:10.1111/j.1532-5415.2000.tb03885.x.
487. Capezuti E, Boltz M, Cline D, et al. Nurses Improving Care for Healthsystem Elders—a model for optimising the geriatric nursing practice environment. J Clin Nurs. Nov 2012;21(21-22):3117-25. doi:10.1111/j.1365-2702.2012.04259.x.
488. Ingraham AM, Richards KE, Hall BL, Ko CY. Quality improvement in surgery: the American College of Surgeons National Surgical Quality Improvement Program approach. Adv Surg. 2010;44:251-67. doi:10.1016/j.yasu.2010.05.003.
489. American Geriatrics Society Expert Panel on the Care of Older Adults with Multimorbidity. (2012). Guiding principles for the care of older adults with multimorbidity: an approach for clinicians. Journal of the American Geriatrics Society, 60(10), E1-E25.
490. Boyd, C., Smith, C.D., Masoudi, F.A., Blaum, C.S., Dodson, J.A., Green, A.R., . . . Tinetti, M.E. (2019). Decision making for older adults with multiple chronic conditions: executive summary for the American Geriatrics Society guiding principles on the care of older adults with multimorbidity. Journal of the American Geriatrics Society, 67(4), 665-673.
491. Institute for Healthcare Improvement. (2022). Age-friendly health systems: Guide to using the 4Ms in the care of older adults in hospitals and ambulatory practices. Available at: https://forms.ihi.org/hubfs/IHIAgeFriendlyHealthSystems_GuidetoUsing4MsCare.pdf .
492. Centers for Medicare Medicaid Services. (2023). CMS National Quality Strategy. Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
493. Centers for Medicare Medicaid Services Quality Net. Public Reporting Overview. Available at: https://qualitynet.cms.gov/inpatient/public-reporting .
494. CDC. (2024). HAI Data Portal. Available at: https://www.cdc.gov/healthcare-associated-infections/php/data/index.html .
495. Ibid.
496. CDC. (2021). Health Topics—Healthcare-associated Infections (HAI). Available at: https://www.cdc.gov/policy/polaris/healthtopics/hai/index.html#:~:text=HAIs%20in%20U.S.%20hospitals%20have,least%20%2428.4%20billion%20each%20year .
497. Bearman, G., Doll, M., Cooper, K. et al. Hospital Infection Prevention: How Much Can We Prevent and How Hard Should We Try? Curr Infect Dis Rep 21, 2. (2019). https://doi.org/10.1007/s11908-019-0660-2 .
498. da Silva R, Casella T. (2022). Healthcare-associated infections in patients who are immunosuppressed due to chemotherapy treatment: a narrative review. J Infect Dev Ctries 16:1784-1795. doi: 10.3855/jidc.16495.
499. Biscione A, Corrado G, Quagliozzi L, Federico A, Franco R, Franza L, Tamburrini E, Spanu T, Scambia G, Fagotti A. Healthcare associated infections in gynecologic oncology: clinical and economic impact. Int J Gingerol Cancer. 2023 Feb 6;33(2):278-284. doi: 10.1136/ijgc-2022-003847. PMID: 36581487.
500. Sammon, J., Trinh, V.Q., Ravi, P., Sukumar, S., Gervais, M.-K., Shariat, S.F., Larouche, A., Tian, Z., Kim, S.P., Kowalczyk, K.J., Hu, J.C., Menon, M., Karakiewicz, P.I., Trinh, Q.-D. and Sun, M. (2013), Health care-associated infections after major cancer surgery. Cancer, 119: 2317-2324. https://doi.org/10.1002/cncr.28027 .
501. Ibid.
502. Sankaran SP, Villa A, Sonis S. Healthcare-associated infections among patients hospitalized for cancers of the lip, oral cavity and pharynx. Infect Prev Pract. 2021 Jan 13;3(1):100115. doi: 10.1016/j.infpip.2021.100115. PMID: 34368735; PMCID: PMC8336044.
503. CDC. (2024). Basic Infection Control and Prevention Plan for Outpatient Oncology Settings. Available at: https://www.cdc.gov/healthcare-associated-infections/hcp/prevention-healthcare/outpatient-oncology.html .
504. The White House. Cancer Moonshot. https://www.whitehouse.gov/cancermoonshot/ .
505. CDC. (2024). Urinary Tract Infection (Catheter-Associated Urinary Tract Infection [CAUTI] and Non-Catheter-Associated Urinary Tract Infection [UTI]) Events. Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/7psccauticurrent.pdf .
506. Ibid.
507. CDC. (2022). Antibiotic Resistance Patient Safety Portal: Catheter-Associated Urinary Tract Infections. Available at: https://arpsp.cdc.gov/profile/nhsn/cauti .
508. CDC. (2024). Urinary Tract Infection (Catheter-Associated Urinary Tract Infection [CAUTI] and Non-Catheter-Associated Urinary Tract Infection [UTI]) Events. Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/7psccauticurrent.pdf .
509. Ibid.
510. Sampathkumar, P., Barth, J.W., Johnson, M., Marosek, N., Johnson, M., Worden, W., Lembke, J., Twing, H., Buechler, T., Dhanorker, S., Keigley, D., Thompson, R. (2016). Mayo Clinic Reduces Catheter-Associated Urinary Tract Infections Through a Bundled 6-C Approach. Joint Commission journal on quality and patient safety, 42(6), 254-261. https://doi.org/10.1016/s1553-7250(16)42033-7 .
511. Baker, Susan BSN, RN; Shiner, Darcy BSN, RN; Stupak, Judy MSN, RN, CNRN; Cohen, Vicki MSN, RN, CNRN; Stoner, Alexis BSN, RN. Reduction of Catheter-Associated Urinary Tract Infections: A Multidisciplinary Approach to Driving Change. Critical Care Nursing Quarterly 45(4):p 290-299, October/December 2022. | DOI: 10.1097/CNQ.0000000000000429.
512. CDC. (2024). Guideline for Prevention of Catheter-Associated Urinary Tract Infections. Available at: https://www.cdc.gov/infection-control/hcp/cauti/index.html .
513. da Silva R, Casella T. (2022). Healthcare-associated infections in patients who are immunosuppressed due to chemotherapy treatment: a narrative review. J Infect Dev Ctries 16:1784-1795. doi: 10.3855/jidc.16495.
514. Chan JY, Semenov YR, Gourin CG. Postoperative urinary tract infection and short-term outcomes and costs in head and neck cancer surgery. Otolaryngol Head Neck Surg. 2013 Apr;148(4):602-10. doi: 10.1177/0194599812474595. Epub 2013 Jan 24. PMID: 23348871.
515. Mercadel, A.J., Holloway, S.B., Saripella, M., Lea, J.S. (2023). Risk factors for catheter-associated urinary tract infections following radical hysterectomy for cervical cancer. American journal of obstetrics and gynecology, 228(6), 718.e1-718.e7. https://doi.org/10.1016/j.ajog.2023.02.019 .
516. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
517. CMS National Quality Strategy. (2023). Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
518. Ibid.
519. Centers for Medicare Medicaid Services. (December 1, 2023). 2023 Measures Under Consideration (MUC) List. Available at: https:// mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
520. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
521. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf .
522. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Final MUC Recommendation Report. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-2023-MUC-Recommendations-Report-Final-.pdf .
523. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf .
524. Battelle—Partnership for Quality Measurement. NHSN Catheter-Associated Urinary Tract Infection (CAUTI) Outcome Measure. Available at: https://p4qm.org/measures/0138 .
525. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
526. CDC. (2024). CDC Locations and Descriptions and Instructions for Mapping Patient Care Locations. Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/15locationsdescriptions_current.pdf .
527. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
528. CDC. (2024). NHSN SIR Guide. Available at: https://www.cdc.gov/nhsn/pdfs/ps-analysis-resources/nhsn-sir-guide.pdf .
529. Battelle—Partnership for Quality Measurement. NHSN Catheter-Associated Urinary Tract Infection (CAUTI) Outcome Measure. Available at: https://p4qm.org/measures/0138 .
530. CDC. (2023). FAQs About NHSN Agreement to Participate and Consent. Available at: https://www.cdc.gov/nhsn/about-nhsn/faq-agreement-to-participate.html .
531. CDC. (2024). National and State Healthcare-associated Infections Progress Report. Available at: https://www.cdc.gov/healthcare-associated-infections/php/data/progress-report.html .
532. CDC. (2024). Urinary Tract Infection (Catheter-Associated Urinary Tract Infection [CAUTI] and Non-Catheter-Associated Urinary Tract Infection [UTI]) Events. Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/7psccauticurrent.pdf .
533. Medical News Today. (2023). What are central venous catheters? Available at: https://www.medicalnewstoday.com/articles/central-venous-catheters .
534. CDC. (2024). Central Line-associated Bloodstream Infections: Resources for Patients and Healthcare Providers. Available at: https://www.cdc.gov/clabsi/about/index.html .
535. Novosad, S.A., Fike, L., Dudeck, M.A., Allen-Bridson, K., Edwards, J.R., Edens, C., Sinkowitz-Cochran, R., Powell, K., Kuhar, D. (2020). Pathogens causing central-line-associated bloodstream infections in acute-care hospitals-United States, 2011-2017. Infection control and hospital epidemiology, 41 (3), 313-319. https://doi.org/10.1017/ice.2019.303 .
536. Brunelli, S.M., Turenne, W., Sibbel, S., Hunt, A., Pfaffle, A. (2016). Clinical and economic burden of bloodstream infections in critical care patients with central venous catheters. Journal of Critical Care, 35, 69-74. https://doi.org/10.1016/j.jcrc.2016.04.035 .
537. Agency for Healthcare Research and Quality. (2017). Estimating the Additional Hospital Inpatient Cost and Mortality Associated With Selected Hospital-Acquired Conditions. Available at: https://www.ahrq.gov/hai/pfp/haccost2017-results.html .
538. CDC. (2022). Antibiotic Resistance Patient Safety Portal: Central Line-Associated Bloodstream Infections. Available at: https://arpsp.cdc.gov/profile/nhsn/clabsi .
539. Ibid.
540. DiBiase, L., Summerlin-Long, S., Stancill, L., Vavalle, E., Teal, L., Weber, D. (2023). Examining CLABSI rates by central-line type. Antimicrobial Stewardship Healthcare Epidemiology, 3 (S2), S48-S49. doi:10.1017/ash.2023.288.
541. Chovanec, K., Arsene, C., Gomez, C., Brixey, M., Tolles, D., Galliers, J.W., Kopaniasz, R., Bobash, T., Goodwin, L. (2021). Association of CLABSI With Hospital Length of Stay, Readmission Rates, and Mortality: A Retrospective Review. Worldviews on evidence-based nursing, 18 (6), 332-338. https://doi.org/10.1111/wvn.12548 .
542. Bell, T., O'Grady, N.P. (2017). Prevention of Central Line-Associated Bloodstream Infections. Infectious disease clinics of North America, 31(3), 551-559. https://doi.org/10.1016/j.idc.2017.05.007 .
543. CDC. (2011). Guidelines for the Prevention of Intravascular Catheter-Related Infections. Available at: https://www.cdc.gov/infection-control/media/pdfs/Guideline-BSI-H.pdf .
544. Grigonis, A.M., Dawson, A.M., Burkett, M., Dylag, A., Sears, M., Helber, B., Snyder, L.K. (2016). Use of a Central Catheter Maintenance Bundle in Long-Term Acute Care Hospitals. American journal of critical care: an official publication, American Association of Critical-Care Nurses, 25 (2), 165-172. https://doi.org/10.4037/ajcc2016894 .
545. Wei, A.E., Markert, R.J., Connelly, C., Polenakovik, H. (2021). Reduction of central line-associated bloodstream infections in a large acute care hospital in Midwest United States following implementation of a comprehensive central line insertion and maintenance bundle. Journal of infection prevention, 22 (5), 186-193. https://doi.org/10.1177/17571774211012471 .
546. Burke, C., Jakub, K., Kellar, I. (2021). Adherence to the central line bundle in intensive care: An integrative review. American journal of infection control, 49 (7), 937-956. https://doi.org/10.1016/j.ajic.2020.11.014 .
547. CDC. (2024). Guidelines for the Prevention of Intravascular Catheter-Related Infections. Available at: https://www.cdc.gov/infection-control/hcp/intravascular-catheter-related-infection/index.html .
548. Page, J., Tremblay, M., Nicholas, C., James, T.A. (2016). Reducing Oncology Unit Central Line-Associated Bloodstream Infections: Initial Results of a Simulation-Based Educational Intervention. Journal of oncology practice, 12 (1), e83-e87. https://doi.org/10.1200/JOP.2015.005751 .
549. Raad, I., Chaftari, A.M. (2014). Advances in prevention and management of central line-associated bloodstream infections in patients with cancer. Clinical infectious diseases: an official publication of the Infectious Diseases Society of America, 59 Suppl 5, S340-S343. https://doi.org/10.1093/cid/ciu670 .
550. Page, J., Tremblay, M., Nicholas, C., James, T.A. (2016). Reducing Oncology Unit Central Line-Associated Bloodstream Infections: Initial Results of a Simulation-Based Educational Intervention. Journal of oncology practice, 12 (1), e83-e87. https://doi.org/10.1200/JOP.2015.005751 .
551. Raad, I., Chaftari, A.M. (2014). Advances in prevention and management of central line-associated bloodstream infections in patients with cancer. Clinical infectious diseases:an official publication of the Infectious Diseases Society of America, 59 Suppl 5, S340-S343. https://doi.org/10.1093/cid/ciu670 .
552. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
553. CMS National Quality Strategy. (2023). Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
554. Centers for Medicare Medicaid Services. (December 1, 2023). 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
555. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
556. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf .
557. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Final MUC Recommendation Report. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-2023-MUC-Recommendations-Report-Final-.pdf .
558. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf .
559. Battelle—Partnership for Quality Measurement. NHSN Central Line-Associated Bloodstream Infection (CLABSI) Outcome Measure. Available at: https://p4qm.org/measures/0139 .
560. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
561. CDC. (2024). CDC Locations and Descriptions and Instructions for Mapping Patient Care Locations. Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/15locationsdescriptions_current.pdf .
562. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
563. CDC. (2024). NHSN SIR Guide. Available at: https://www.cdc.gov/nhsn/pdfs/ps-analysis-resources/nhsn-sir-guide.pdf .
564. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
565. CDC. (2023). FAQs About NHSN Agreement to Participate and Consent. Available at: https://www.cdc.gov/nhsn/about-nhsn/faq-agreement-to-participate.html .
566. CDC. (2024). National and State Healthcare-associated Infections Progress Report. Available at: https://www.cdc.gov/healthcare-associated-infections/php/data/progress-report.html .
567. CDC. (2024). Bloodstream Infection Event (Central Line-Associated Bloodstream Infection and Non-central Line Associated Bloodstream Infection). Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/4psc_clabscurrent.pdf .
568. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Final MUC Recommendation Report. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-2023-MUC-Recommendations-Report-Final-.pdf .
569. CDC. (2022). Antibiotic Resistance Patient Safety Portal: Catheter-Associated Urinary Tract Infections. Available at: https://arpsp.cdc.gov/profile/nhsn/cauti .
570. CDC. (2022). Antibiotic Resistance Patient Safety Portal: Central Line-Associated Bloodstream Infections. Available at: https://arpsp.cdc.gov/profile/nhsn/clabsi .
571. CDC. (2024). National and State Healthcare-associated Infections Progress Report. Available at: https://www.cdc.gov/healthcare-associated-infections/php/data/progress-report.html .
572. CDC. (2024). CDC Locations and Descriptions and Instructions for Mapping Patient Care Locations. Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/15locationsdescriptions_current.pdf .
573. CDC. (2024). CDC Locations and Descriptions and Instructions for Mapping Patient Care Locations. Available at: https://www.cdc.gov/nhsn/pdfs/pscmanual/15locationsdescriptions_current.pdf .
574. Bysshe, T., Gao, Y., Heaney-Huls, K., et al. (2017). Final Report Estimating the Additional Hospital Inpatient Cost and Mortality Associated with Selected Hospital Acquired Conditions.
575. Morello, R.T., Barker, A.L., Watts, J.J., Haines, T., Zavarsek, S.S., Hill, K.D., Brand, C., Sherrington, C., Wolfe, R., Bohensky, M.A., Stoelwinder, J.U. (2015). The Extra Resource Burden of In-hospital Falls: a Cost of Falls Study. The Medical Journal of Australia, 203 (9), 367. https://doi.org/10.5694/mja15.00296 .
576. Dykes, P.C., Curtin-Bowen, M., Lipsitz, S., Franz, C., Adelman, J., Adkison, L., Bogaisky, M., Carroll, D., Carter, E., Herlihy, L., Lindros, M.E., Ryan, V., Scanlan, M., Walsh, M.A., Wien, M., Bates, D.W. (2023). Cost of Inpatient Falls and Cost-Benefit Analysis of Implementation of an Evidence-Based Fall Prevention Program. JAMA Health Forum, 4 (1), e225125. https://doi.org/10.1001/jamahealthforum.2022.5125 .
577. AHRQ. (2019). Patient Safety Primer: Falls. Retrieved July 24, 2019, from AHRQ PSNet website: https://psnet.ahrq.gov/primers/primer/40/ Falls.
578. Currie, L. (2008). Fall and Injury Prevention. In E. Hughes RG (Ed.), Patient Safety and Quality: An Evidence-Based Handbook for Nurses (pp. 195-250). Rockville: Agency for Healthcare Research and Quality.
579. Montero-Odasso, M., Van der Velde, N., Martin, F.C., et al. (2022). World Guidelines for Falls Prevention and Management for Older Adults: A Global Initiative. Age and Ageing, 51(9), 1-36.
580. Staggs, V.S., Mion, L.C., Shorr, R.I. (2015). Consistent Differences in Medical Unit Fall Rates: Implications for Research and Practice. Journal of the American Geriatrics Society, 63(5), 983-987. https://doi.org/10.1111/jgs.13387 .
581. Registered Nurses' Association of Ontario. (2017). Preventing Falls and Reducing Injury from Falls (4th ed.). Toronto, ON: Registered Nurses' Association of Ontario.
582. National Institute of Health and Care Excellence. (2013). Falls in Older People: Assessing Risk and Prevention.
583. ACS National Surgical Quality Improvement Program (NSQIP)/American Geriatrics Society (AGS). (2016). Optimal Perioperative Management of the Geriatric Patient: Best Practices Guideline from ACS NSQIP/AGS. https://www.facs.org/media/y5efmgox/acs-nsqip-geriatric-2016-guidelines.pdf .
584. PSI 90 Technical Specification can be found here: https://qualitynet.cms.gov/inpatient/measures/psi/resources .
585. Battelle—Partnership for Quality Measurement. Hospital Harm—Falls with Injury. Available at: https://p4qm.org/measures/4120e .
586. Ibid.
587. CMS National Quality Strategy. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy .
588. President's Council of Advisors on Science and Technology. (2023). Report to the President: A Transformational Effort on Patient Safety. https://www.whitehouse.gov/wp-content/uploads/2023/09/PCAST_Patient-Safety-Report_Sept2023.pdf .
589. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
590. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
591. Hshieh, T.T., Yue, J., Oh, E., Puelle, M., Dowal, S., Travison, T., Inouye, S. K. (2015). Effectiveness of multicomponent nonpharmacological delirium interventions: a meta-analysis. JAMA internal medicine, 175(4), 512-520. https://doi.org/10.1001/jamainternmed.2014.7779 .
592. Montero-Odasso, M., van der Velde, N., Martin, F.C., Petrovic, M., Tan, M.P., Ryg, J., Aguilar-Navarro, S., Alexander, N.B., Becker, C., Blain, H., Bourke, R., Cameron, I.D., Camicioli, R., Clemson, L., Close, J., Delbaere, K., Duan, L., Duque, G., Dyer, S.M., . . . Rixt Zijlstra, G.A. (2022). World guidelines for falls prevention and management for older adults: a global initiative. Age and Ageing, 51(9), 1-36.
593. Battelle—Partnership for Quality Measurement. Hospital Harm—Fall Injury Measure Specifications. Available at: https://p4qm.org/measures/4120e .
594. Battelle—Partnership for Quality Measurement. 2023 Management of Acute and Chronic Events Meeting Summary. https://p4qm.org/sites/default/files/Management%20of%20Acute%20Events%2C%20Chronic%20Disease%2C%20Surgery%2C%20and%20Behavioral%20Health/material/EM-Acute-Chronic-Events-Fall2023-Endorsement-Meeting-Summary.pdf
595. To access the value sets for the measure, please visit the Value Set Authority Center (VSAC), sponsored by the National Library of Medicine, at https://vsac.nlm.nih.gov/ .
596. Battelle—Partnership for Quality Measurement. Hospital Harm—Falls with Injury. Available at: https://p4qm.org/measures/4120e .
597. Ibid.
598. Hshieh, T.T., Yue, J., Oh, E., Puelle, M., Dowal, S., Travison, T., Inouye, S.K. (2015). Effectiveness of multicomponent nonpharmacological delirium interventions: a meta-analysis. JAMA internal medicine, 175(4), 512-520. https://doi.org/10.1001/jamainternmed.2014.7779 .
599. Battelle—Partnership for Quality Measurement. (2024). Hospital Harm—Falls with Injury. Available at: https://p4qm.org/measures/4120e .
600. Battelle—Partnership for Quality Measurement. (2024). Hospital Harm—Falls with Injury. Available at: https://p4qm.org/measures/4120e .
601. QRDA—Quality Reporting Document Architecture, https://ecqi.healthit.gov/qrda?qt-tabs_qrda=about .
602. Gangopadhyaya, A., Pugazhendhi, A., Austin, M., Campione, A., Danforth, M. (2023) Racial, ethnic, and payer disparities in adverse safety events: Are there differences across Leapfrog Hospital Safety Grades? The Leapfrog Group. https://www.leapfroggroup.org/racial-ethnic-and-payer-disparities-adverse-safety-events-are-there-differences-across-leapfrog .
603. Battelle—Partnership for Quality Measurement. (2024). Hospital Harm—Falls with Injury. Available at: https://p4qm.org/measures/4120e .
604. Battelle—Partnership for Quality Measurement. (2024). Hospital Harm—Falls with Injury. Available at: https://p4qm.org/measures/4120e .
605. Battelle—Partnership for Quality Measurement. (2024). Hospital Harm—Falls with Injury. Available at: https://p4qm.org/measures/4120e .
606. eCQI Resource Center, ONC Project Tracking System (Jira), https://ecqi.healthit.gov/tool/onc-project-tracking-system-jira .
607. CMS, Reference Brief: Digital Quality Measurement eCQMs, https://ecqi.healthit.gov/sites/default/files/Digital%20Quality%20Measurement%20eCQMs%20reference%20brief_508ed.pdf .
608. CMS, dQMs—Digital Quality Measures, available at https://ecqi.healthit.gov/dqm?qt-tabs_dqm=about-dqms .
609. CMS, dQMs—Digital Quality Measures, https://ecqi.healthit.gov/dqm?qt-tabs_dqm=dqm-strategic-roadmap .
610. Stocking, J.C., Utter, G.H., Drake, C., Aldrich, J.M., Ong, M.K., Amin, A., Marmor, R.A., Godat, L., Cannesson, M., Gropper, M.A., Romano, P.S. (2020). Postoperative Respiratory Failure: An Update on the Validity of the Agency for Healthcare Research and Quality Patient Safety Indicator 11 in an Era of Clinical Documentation Improvement Programs. American Journal of Surgery, 220 (1), 222-228. https://doi.org/10.1016/j.amjsurg.2019.11.019
611. Sabate S., Mazo V., Canet J. (2014). Predicting Postoperative Pulmonary Complications: Implications for Outcomes and Costs. Case Reports in Anesthesiology. 27(2), 201-209.
612. Rosen, A.K., Loveland, S., Shin, M., Shwartz, M., Hanchate, A., Chen, Q., Kaafarani, H.M., Borzecki, A. (2013). Examining the impact of the AHRQ Patient Safety Indicators (PSIs) on the Veterans Health Administration: the case of readmissions. Medical Care, 51(1), 37-44.
613. Lawson E.H., Hall B.L., Louie R., et al. (2013). Association Between Occurrence of a Postoperative Complication and Readmission: Implications for Quality Improvement and Cost Savings. Annals of Surgery, 258(1),10-18.
614. Stocking, J.C., Drake, C., Aldrich, J.M., Ong, M.K., Amin, A., Marmor, R.A., Godat, L., Cannesson, M., Gropper, M.A., Romano, P.S., Sandrock, C., Bime, C., Abraham, I., Utter, G.H. (2022). Outcomes and Risk Factors for Delayed-onset Postoperative Respiratory Failure: A Multi-center Case-control Study by the University of California Critical Care Research Collaborative (UC3. RC). BMC Anesthesiology, 22(1), 146.
615. Encinosa, W.E., Hellinger, F.J. (2008). The Impact of Medical Errors on Ninety-day Costs and Outcomes: An Examination of Surgical Patients. Health Services Research, 43(6), 2067-2085.
616. Zrelak, P.A., Utter, G.H., Sadeghi, B., Cuny, J., Baron, R., Romano, P.S. (2012). Using the Agency for Healthcare Research and Quality patient safety indicators for targeting nursing quality improvement. Journal of Nursing Care Quality, 27(2), 99-108.
617. Rahman, M., Neal, D., Fargen, K.M., Hoh, B.L. (2013). Establishing Standard Performance Measures for Adult Brain Tumor Patients: A Nationwide Inpatient Sample Database Study. Neuro-oncology, 15(11), 1580-1588.
618. PSI 90 Technical Specification can be found here: https://qualitynet.cms.gov/inpatient/measures/psi/resources .
619. Battelle—Partnership for Quality Measurement. Hospital Harm—Postoperative Respiratory Failure. Available at: https://p4qm.org/measures/4130e .
620. Ibid.
621. CMS National Quality Strategy. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy .
622. President's Council of Advisors on Science and Technology. (2023). Report to the President: A Transformational Effort on Patient Safety. https://www.whitehouse.gov/wp-content/uploads/2023/09/PCAST_Patient-Safety-Report_Sept2023.pdf .
623. CMS National Quality Strategy. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy .
624. Centers for Medicare Medicaid Services. (December 1, 2023). 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
625. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
626. Battelle—Partnership for Quality Measurement. Hospital Harm—Postoperative Respiratory Failure. Available at: https://p4qm.org/measures/4130e .
627. Battelle—Partnership for Quality Measurement. Fall 2023 Management of Acute and Chronic Events Meeting Summary. Available at: https://p4qm.org/sites/default/files/Management%20of%20Acute%20Events%2C%20Chronic%20Disease%2C%20Surgery%2C%20and%20Behavioral%20Health/material/EM-Acute-Chronic-Events-Fall2023-Endorsement-Meeting-Summary.pdf .
628. To access the value sets for the measure, please visit the Value Set Authority Center (VSAC), sponsored by the National Library of Medicine, at https://vsac.nlm.nih.gov/ .
629. Battelle—Partnership for Quality Measurement. Hospital Harm—Postoperative Respiratory Failure. Available at: https://p4qm.org/measures/4130e .
630. Ibid.
631. Ruscic KJ, Grabitz SD, Rudolph MI, Eikermann M. Prevention of respiratory complications of the surgical patient: actionable plan for continued process improvement. Curr Opin Anaesthesiol. 2017 Jun;30(3):399-408.
632. One of the 13 hospitals did not always use their structured fields to capture mechanical ventilation. For this reason, the site opted to not proceed with reliability and validity phases of testing.
633. For more details, see Partnership for Quality Measurement. Endorsement and Maintenance (EM) Guidebook. (2023). Available at: https://p4qm.org/sites/default/files/2023-12/Del-3-6-Endorsement-and-Maintenance-Guidebook-Final_0_0.pdf .
634. Battelle—Partnership for Quality Measurement. (2024). Hospital Harm—Postoperative Respiratory Failure. Available at: https://p4qm.org/measures/4130e .
635. Centers for Medicare Medicaid. Measures Management System Hub. Available at: https://mmshub.cms.gov .
636. Partnership for Quality Measurement. Endorsement and Maintenance (EM) Guidebook. (2023). Available at: https://p4qm.org/sites/default/files/2023-12/Del-3-6-Endorsement-and-Maintenance-Guidebook-Final_0_0.pdf .
637. One of the 13 hospitals did not always use their structured fields to capture mechanical ventilation. For this reason, the site opted to not proceed with reliability and validity phases of testing.
638. Becker's Hospital Review. Epic vs. Cerner: EHR Market Share. Available at: https://www.beckershospitalreview.com/ehrs/epic-vs-cerner-ehr-market-share.html?oly_enc_id=3703D1456278B3W .
639. https://oncprojectracking.healthit.gov/olp/ .
640. Silber JH, Williams SV, Krakauer H, Schwartz JS. Hospital and patient characteristics associated with death after surgery. A study of adverse occurrence and failure to rescue. Med Care. 1992 Jul;30(7):615-29. doi: 10.1097/00005650-199207000-00004.
641. Needleman J, Buerhaus P, Mattke S, Stewart M, Zelevinsky K. Nurse-staffing levels and the quality of care in hospitals. N Engl J Med. 2002 May 30;346(22):1715-22. doi: 10.1056/NEJMsa012247.
642. Portuondo JI, Shah SR, Singh H, Massarweh NN. Failure to Rescue as a Surgical Quality Indicator: Current Concepts and Future Directions for Improving Surgical Outcomes. Anesthesiology. 2019 Aug;131(2):426-437. doi: 10.1097/ALN.0000000000002602.
643. Silber, J. H., Rosenbaum, P. R., Ross, R. (1995). Comparing the Contributions of Groups of Predictors: Which Outcomes Vary with Hospital Rather than Patient Characteristics? Journal of the American Statistical Association, 90 (429), 7-18. https://doi.org/10.2307/2291124 .
644. Liao, L. M., Sun, X. Y., Yu, H., Li, J. W. (2016). The Association of Nurse Educational Preparation and Patient Outcomes: Systematic Review and Meta-Analysis. Nurse Education Today, 42, 9-16. https://doi.org/10.1016/j.nedt.2016.03.029 .
645. Burke, J. R., Downey, C., Almoudaris, A. M. (2022). Failure to Rescue Deteriorating Patients: A Systematic Review of Root Causes and Improvement Strategies. Journal of Patient Safety, 18 (1), e140-e155. https://doi.org/10.1097/PTS.0000000000000720 .
646. Hall K.K., Lim A., Gale B. (2020). Failure To Rescue. In: Hall, K. K., Shoemaker-Hunt, S., Hoffman, et al. Making Healthcare Safer III: A Critical Analysis of Existing and Emerging Patient Safety Practices. Agency for Healthcare Research and Quality (US).
647. Hall K.K., Lim A., Gale B. (2020). The Use of Rapid Response Teams to Reduce Failure to Rescue Events: A Systematic Review. Journal of Patient Safety. 16(3S Suppl 1):S3-S7.
648. Johnston, M. J., Arora, S., King, D., Bouras, G., Almoudaris, A. M., Davis, R., Darzi, A. (2015). A Systematic Review to Identify the Factors that Affect Failure to Rescue and Escalation of Care in Surgery. Surgery, 157 (4), 752-763. https://doi.org/10.1016/j.surg.2014.10.017 .
649. Kaestner, R., Silber, J. H. (2010). Evidence on the Efficacy of Inpatient Spending on Medicare Patients. The Milbank Quarterly, 88 (4), 560-594.
650. Silber, J. H., Kaestner, R., Even-Shoshan, O., Wang, Y., Bressler, L. J. (2010). Aggressive Treatment Style and Surgical Outcomes. Health Services Research, 45 (6 Pt 2), 1872-1892.
651. Centers for Medicare Medicaid Services. (2023). CMS National Quality Strategy. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy .
652. Centers for Medicare Medicaid Services. (December 1, 2023). 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
653. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
654. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
655. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
656. Battelle—Partnership for Quality Measurement. (February 2024). 2023 Pre-Rulemaking Measure Review (PRMR) Meeting Summary: Hospital Committee. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-Hospital-Recommendation-Group-Meeting-Summary-Final.pdf
657. Battelle—Partnership for Quality Measurement. (February 2024). Fall 2023 Management of Acute and Chronic Events Meeting Summary. Available at: https://p4qm.org/sites/default/files/Management%20of%20Acute%20Events,%20Chronic%20Disease,%20Surgery,%20and%20Behavioral%20Health/material/EM-Acute-Chronic-Events-Fall2023-Endorsement-Meeting-Summary.pdf .
658. Battelle—Partnership for Quality Measurement. (February 2024). Fall 2023 Management of Acute and Chronic Events Meeting Summary. Available at: https://p4qm.org/sites/default/files/Management%20of%20Acute%20Events,%20Chronic%20Disease,%20Surgery,%20and%20Behavioral%20Health/material/EM-Acute-Chronic-Events-Fall2023-Endorsement-Meeting-Summary.pdf .
659. Battelle—Partnership for Quality Measurement. Thirty-day Risk-Standardized Death Rate among Surgical Inpatients with Complications (Failure-to-Rescue). Available at: https://p4qm.org/measures/4125 .
660. Ibid.
661. Ibid.
662. https://doi.org/10.1016/j.jcjq.2017.08.003 .
663. https://p4qm.org/measures/4125 .
664. Ibid.
665. https://p4qm.org/measures/4125 .
666. Battelle—Partnership for Quality Measurement. (February 2024). Fall 2023 Management of Acute and Chronic Events Meeting Summary. Available at: https://p4qm.org/sites/default/files/Management%20of%20Acute%20Events,%20Chronic%20Disease,%20Surgery,%20and%20Behavioral%20Health/material/EM-Acute-Chronic-Events-Fall2023-Endorsement-Meeting-Summary.pdf .
667. https://p4qm.org/measures/4125 .
668. Centers for Medicare Medicaid Services. (November 2022). Encounter Data Submission and Processing Guide Version 5.0. Available at: https://csscoperations.com/internet/csscw3_files.nsf/F2/2022ED_Submission_Processing_Guide_20221130.pdf/$FILE/2022ED_Submission_Processing_Guide_20221130.pdf .
669. Centers for Medicare Medicaid Services. (November 2022). Encounter Data Submission and Processing Guide Version 5.0. Available at: https://csscoperations.com/internet/csscw3_files.nsf/F2/2022ED_Submission_Processing_Guide_20221130.pdf/$FILE/2022ED_Submission_Processing_Guide_20221130.pdf .
670. Centers for Medicare Medicaid Services. (November 2022). Encounter Data Submission and Processing Guide Version 5.0. Available at: https://csscoperations.com/internet/csscw3_files.nsf/F2/2022ED_Submission_Processing_Guide_20221130.pdf/$FILE/2022ED_Submission_Processing_Guide_20221130.pdf .
671. For more information on the CBE measure review process we refer readers to https://p4qm.org/MSR .
672. We refer readers to the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41540 through 41544 ) for a summary of the Hospital IQR Program's removal Factors. Removal Factors were codified at 42 CFR 412.140(g)(2) and (3) . ( 88 FR 59144 ).
673. Agency for Healthcare Research and Quality. (2023). AHRQ Quality Indicators TM (AHRQ QI TM ) ICD-9-CM Specification Version 6.0. Available at: https://qualityindicators.ahrq.gov/Downloads/Modules/PSI/V2023/TechSpecs/PSI_04_Death_Rate_among_Surgical_Inpatients_with_Serious_Treatable_Complications.pdf .
674. Partnership for Quality Measurement. (2023). Death Rate among Surgical Inpatients with Serious Treatable Complications (PSI 04). Available at: https://p4qm.org/measures/0351 .
675. Nilsson, U., Gruen, R., Myles, P.S. (2020). Postoperative recovery: The importance of the team. Anesthesia, 75(S1). https://doi.org/10.1111/anae.14869 .
676. Azad, T.D., Rodriguez, E., Raj, D., Xia, Y., Materi, J., Rincon-Torroella, J., Gonzalez, L.F., Suarez, J.I., Tamargo, R.J., Brem, H., Haut, E.R., Bettegowda, C. (2023). Patient Safety Indicator 04 Does Not Consistently Identify Failure to Rescue in the Neurosurgical Population. Neurosurgery, 92(2), 338-343. https://doi.org/10.1227/neu.0000000000002204 .
677. Ibid.
678. Ibid.
679. Azad, T.D., Rodriguez, E., Raj, D., Xia, Y., Materi, J., Rincon-Torroella, J., Gonzalez, L.F., Suarez, J.I., Tamargo, R.J., Brem, H., Haut, E.R., Bettegowda, C. (2023). Patient Safety Indicator 04 Does Not Consistently Identify Failure to Rescue in the Neurosurgical Population. Neurosurgery, 92(2), 338-343. https://doi.org/10.1227/neu.0000000000002204 .
680. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
681. Rosero, E.B., Romito, B.T., Joshi, G.P. (2021). Failure to rescue: A quality indicator for postoperative care. Best Practice amp; Research Clinical Anesthesiology, 35(4), 575-589. https://doi.org/10.1016/j.bpa.2020.09.003 .
682. Hall KK, Lim A, Gale B. Failure To Rescue. In: Hall KK, Shoemaker-Hunt S, Hoffman L, et al. Making Healthcare Safer III: A Critical Analysis of Existing and Emerging Patient Safety Practices [internet]. Rockville (MD): Agency for Healthcare Research and Quality (US); 2020 Mar. 2. Available at: https://www.ncbi.nlm.nih.gov/books/NBK555513/ .
683. Rodziewicz TL, Houseman B, Hipskind JE. Medical Error Reduction and Prevention. [Updated 2023 May 2]. In: StatPearls [internet]. Treasure Island (FL): StatPearls Publishing; 2023 Jan-. Available at: https://www.ncbi.nlm.nih.gov/books/NBK499956/ .
684. Ward, S.T., Dimick, J.B., Zhang, W., Campbell, D.A., Ghaferi, A.A. (2019). Association Between Hospital Staffing Models and Failure to Rescue. Annals of surgery, 270(1), 91-94. https://doi.org/10.1097/SLA.0000000000002744 .
685. Nilsson, U., Gruen, R., Myles, P.S. (2020). Postoperative recovery: The importance of the team. Anesthesia, 75(S1). https://doi.org/10.1111/anae.14869 .
686. We refer readers to the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41540 through 41544 ) for a summary of the Hospital IQR Program's removal Factors. Removal Factors were codified at 42 CFR 412.140(g)(3) .
687. When substantive updates to measure specifications are needed, we have had to readopt the measure and updates into the Hospital IQR Program first. The measure was initially adopted into the Hospital IQR Program in the FY 2012 IPPS/LTCH PPS final rule ( 76 FR 51618 ) and then was finalized for removal in the FY 2019 IPPS/LTCH PPS final rule ( 83 FR 41559 through 41560 ) to deduplicate the measure sets across programs and reduce burden for hospitals.
688. These refinements are available in a summary of the measure re-evaluation on the CMS QualityNet website, Medicare Spending Per Beneficiary (MSPB) Measure Methodology. Available at: https://qualitynet.cms.gov/inpatient/measures/hvbp-mspb .
689. Centers for Medicare Medicaid Services. (2024). Medicare Spending Per Beneficiary—National https://data.cms.gov/provider-data/dataset/3n5g-6b7f .
690. Partnership for Quality Measurement. (2023). Global Malnutrition Composite Score. Available at: https://p4qm.org/measures/3592e .
691. Centers for Medicare Medicaid Services Measures Inventory Tool. (2023). Global Malnutrition Composite Score. Available at: https://cmit.cms.gov/cmit/#/MeasureView?variantId=5120sectionNumber=1 .
692. United States Agency for Healthcare Research and Quality. (2016). Non-maternal and non-neonatal inpatient stays in the United States involving malnutrition 2016. Available at: https://hcup-us.ahrq.gov/reports/ataglance/HCUPMalnutritionHospReport_083018.pdf .
693. Kabashneh, S., Alkassis, S., Shanah, L., Ali, H. (2020). A Complete Guide to Identify and Manage Malnutrition in Hospitalized Patients. Cureus, 12 (6), e8486. https://doi.org/10.7759/cureus.8486 .
694. Anghel, S., Kerr, K.W., Valladares, A.F., Kilgore, K.M., Sulo, S. (2021). Identifying patients with malnutrition and improving use of nutrition interventions: A quality study in four US hospitals. Nutrition, 91-92, 111360. https://doi.org/10.1016/j.nut.2021.111360 .
695. Sauer, A.C., Goates, S., Malone, A., Mogensen, K.M., Gewirtz, G., Sulz, I., Moick, S., Laviano, A., Hiesmayr, M. (2019). Prevalence of malnutrition risk and the impact of nutrition risk on hospital outcomes: Results from nutrition day in the U.S. Journal of Parenteral and Enteral Nutrition, 43(7), 918-926. https://doi.org/10.1002/jpen.1499 .
696. Suela Sulo, Leah Gramlich, Jyoti Benjamin, Sharon McCauley, Jan Powers, Krishnan Sriram Kristi Mitchell (2020) Nutrition Interventions Deliver Value in Healthcare: Real-World Evidence, Nutrition and Dietary Supplements, 12:, 139-146, DOI: 10.2147/NDS.S262364.
697. Centers for Medicare Medicaid Services. (2023). CMS Framework for Health Equity. Available at: https://www.cms.gov/priorities/health-equity/minority-health/equity-programs/framework .
698. Blankenship, J., Blancato, R. B. (2022). Nutrition Security at the Intersection of Health Equity and Quality Care. Journal of the Academy of Nutrition and Dietetics, 122(10S), S12-S19. https://doi.org/10.1016/j.jand.2022.06.017 .
699. Centers for Medicare Medicaid Services. (2023). CMS National Quality Strategy. Available at: https://www.cms.gov/medicare/quality/meaningful-measures-initiative/cms-quality-strategy .
700. Centers for Medicare Medicaid Services. (December 1, 2023). 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
701. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
702. Centers for Medicare Medicaid Services. 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
703. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
704. Battelle-Partnership for Quality Measurement. (February 2024). Pre-Rulemaking Measure Review Measures Under Consideration 2023 RECOMMENDATIONS REPORT. Available at: https://p4qm.org/sites/default/files/2024-02/PRMR-2023-MUC-Recommendations-Report-Final-.pdf .
705. Battelle-Partnership for Quality Measurement. Global Malnutrition Composite Score eCQM. Available at: https://p4qm.org/measures/3592e
706. Valladares AF, McCauley SM, Khan M, D'Andrea C, Kilgore K, Mitchell K. Development and Evaluation of a Global Malnutrition Composite Score. J Acad Nutr Diet. 2022 Feb;122(2):251-258. doi: 10.1016/j.jand.2021.02.002. Epub 2021 Mar 10. PMID: 33714687.
707. Centers for Medicare Medicaid Services Measures Inventory Tool. (2023). Global Malnutrition Composite Score. Available at: https://cmit.cms.gov/cmit/#/MeasureView?variantId=5120sectionNumber=1 .
708. Centers for Medicare Medicaid Services Measures Inventory Tool. (2023). Global Malnutrition Composite Score. Available at: https://cmit.cms.gov/cmit/#/MeasureView?variantId=5120sectionNumber=1 .
709. Suela Sulo, Leah Gramlich, Jyoti Benjamin, Sharon McCauley, Jan Powers, Krishnan Sriram Kristi Mitchell (2020) Nutrition Interventions Deliver Value in Healthcare: Real-World Evidence, Nutrition and Dietary Supplements, 12:, 139-146, DOI: 10.2147/NDS.S262364.
710. Suela Sulo, Leah Gramlich, Jyoti Benjamin, Sharon McCauley, Jan Powers, Krishnan Sriram Kristi Mitchell (2020) Nutrition Interventions Deliver Value in Healthcare: Real-World Evidence, Nutrition and Dietary Supplements, 12:, 139-146, DOI: 10.2147/NDS.S262364.
711. AHRQ. (2023). National Action Alliance To Advance Patient and Workforce Safety. Available at: https://www.ahrq.gov/cpi/about/otherwebsites/action-alliance.html .
712. President's Council of Advisors on Science and Technology. (2023). Report to the President: A Transformational Effort on Patient Safety. Available at: https://www.whitehouse.gov/wp-content/uploads/2023/09/PCAST_Patient-Safety-Report_Sept2023.pdf .
713. Centers for Medicare Medicaid Services. (2023). CMS National Quality Strategy. Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
714. Centers for Medicare Medicaid Services. (2023). CMS National Quality Strategy. Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
715. Centers for Medicare Medicaid Services. (April 2024). Quality in Motion: Acting on the CMS National Quality Strategy. Available at: https://www.cms.gov/files/document/quality-motion-cms-national-quality-strategy.pdf
716. Definition of dQM available on the eCQI Resource Center at: https://ecqi.healthit.gov/dqm?qt-tabs_dqm=about-dqms .
717. Centers for Medicare Medicaid Services. (2023). CMS National Quality Strategy. Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
718. Centers for Medicare Medicaid Services. (March 2022). Digital Quality Measurement Strategic Roadmap. Available at: https://ecqi.healthit.gov/sites/default/files/CMSdQMStrategicRoadmap_032822.pdf .
719. Centers for Medicare Medicaid Services. (April 2024). Quality in Motion: Acting on the CMS National Quality Strategy. Available at: https://www.cms.gov/files/document/quality-motion-cms-national-quality-strategy.pdf .
720. Centers for Medicare Medicaid Services. (2023). CMS National Quality Strategy. Available at: https://www.cms.gov/files/document/cms-national-quality-strategy-handout.pdf .
721. To provide clarity and to better align with the Hospital IQR Program, we have changed the name of the Facility Commitment to Health Equity measure in the PCHQR Program to the Hospital Commitment to Health Equity measure. This is a non-substantive change and does not impact the measure's specifications or reporting requirements.
722. Items may also be referred to as “data elements.”
723. As noted in section IX.E of the proposed rule and section IX.E. of this final rule, hospitals are required to report whether they have screened patients for five standardized SDOH categories: housing instability, food insecurity, utility difficulties, transportation needs, and interpersonal safety.
724. Office of the Assistant Secretary for Planning and Evaluation (ASPE). Second Report to Congress on Social Risk and Medicare's Value-Based Purchasing Programs. June 28, 2020. Available at: https://aspe.hhs.gov/reports/second-report-congress-social-risk-medicares-value-based-purchasing-programs .
725. World Health Organization. Social determinants of health. Available at: https://www.who.int/health-topics/social-determinants-of-health#tab=tab_1 .
726. Using Z Codes: The Social Determinants of Health (SDOH). Data Journey to Better Outcomes.
727. Improving the Collection of Social Determinants of Health (SDOH) Data with ICD-10-CM Z Codes. https://www.cms.gov/files/document/cms-2023-omh-z-code-resource.pdf .
728. CMS.gov. Measures Management System (MMS). CMS Focus on Health Equity. Health Equity Terminology and Quality Measures. https://mmshub.cms.gov/about-quality/quality-at-CMS/goals/cms-focus-on-health-equity/health-equity-terminology .
729. Centers for Disease Control and Prevention. Social Determinants of Health (SDOH) and PLACES Data.
730. “U.S. Playbook To Address Social Determinants Of Health” from the White House Office Of Science And Technology Policy (November 2023).
731. These SDOH data are also collected for purposes outlined in section 2(d)(2)(B) of the Improving Medicare Post-Acute Care Transitions Act (IMPACT Act). For a detailed discussion on SDOH data collection under section 2(d)(2)(B) of the IMPACT Act, see the FY 2020 LTCH PPS final rule ( 84 FR 42577 through 42579 ).
732. Social Determinants of Health. Healthy People 2020. https://www.healthypeople.gov/2020/topics-objectives/topic/social-determinants-of-health . (February 2019).
733. National Academies of Sciences, Engineering, and Medicine. 2020. Leading Health Indicators 2030: Advancing Health, Equity, and Well-Being. Washington, DC: The National Academies Press. https://doi.org/10.17226/25682 .
734. Centers for Medicare Medicaid Services. “A Guide to Using the Accountable Health Communities Health-Related Social Needs Screening Tool: Promising Practices and Key Insights.” August 2022. Available at https://www.cms.gov/priorities/innovation/media/document/ahcm-screeningtool-companion .
735. Hugh Alderwick and Laura M. Gottlieb, “Meanings and Misunderstandings: A Social Determinants of Health Lexicon for Health Care Systems: Milbank Quarterly,” Milbank Memorial Fund, November 18, 2019, https://www.milbank.org/quarterly/articles/meanings-and-misunderstandings-a-social-determinants-of-health-lexicon-for-health-care-systems/ .
736. Hugh Alderwick and Laura M. Gottlieb, “Meanings and Misunderstandings: A Social Determinants of Health Lexicon for Health Care Systems: Milbank Quarterly,” Milbank Memorial Fund, November 18, 2019, https://www.milbank.org/quarterly/articles/meanings-and-misunderstandings-a-social-determinants-of-health-lexicon-for-health-care-systems/ .
737. American Hospital Association. (2020). Health Equity, Diversity Inclusion Measures for Hospitals and Health System Dashboards. December 2020. Accessed: January 18, 2022. Available at: https://ifdhe.aha.org/system/files/media/file/2020/12/ifdhe_inclusion_dashboard.pdf .
738. In October 2023, we released two new annual Health Equity Confidential Feedback Reports to LTCHs: The Discharge to Community (DTC) Health Equity Confidential Feedback Report and the Medicare Spending Per Beneficiary (MSPB) Health Equity Confidential Feedback Report. The PAC Health Equity Confidential Feedback Reports stratified the DTC and MSPB measures by dual-enrollment status and race/ethnicity. For more information on the Health Equity Confidential Feedback Reports, please refer to the Education and Outreach materials available on the LTCH QRP Training web page at https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-quality-reporting-training .
739. Brooks-LaSure, C. (2021). My First 100 Days and Where We Go from Here: A Strategic Vision for CMS. Centers for Medicare Medicaid. Available at: https://www.cms.gov/blog/my-first-100-days-and-where-we-go-here-strategic-vision-cms .
740. The White House. The Biden-Harris Administration Immediate Priorities [website]. https://www.whitehouse.gov/priorities/ .
741. The AHC Model was a five year demonstration project run by the Centers for Medicare Medicaid Innovation between May 1, 2017 and April 30, 2022. For more information go to https://www.cms.gov/priorities/innovation/innovation-models/ahcm .
742. More information about the AHC HRSN Screening Tool is available on the website at https://innovation.cms.gov/Files/worksheets/ahcm-screeningtool.pdf .
743. Centers for Medicare Medicaid Services, FY2023 IPPS/LTCH PPS final rule ( 87 FR 49191 through 49194).
744. Centers for Medicare Medicaid Services, FY2024 Inpatient Psychiatric Prospective Payment System—Rate Update ( 88 FR 51107 through 51121 ).
745. https://health.gov/healthypeople/priority-areas/social-determinants-health .
746. Healthy People 2030 is a long-term, evidence-based effort led by the U.S. Department of Health and Human Services (HHS) that aims to identify nationwide health improvement priorities and improve the health of all Americans.
747. Kushel, M.B., Gupta, R., Gee, L., Haas, J.S. (2006). Housing instability and food insecurity as barriers to health care among low-income Americans. Journal of General Internal Medicine, 21 (1), 71-77. doi: 10.1111/j.1525-1497.2005.00278.x.
748. Homelessness is defined as “lacking a regular nighttime residence or having a primary nighttime residence that is a temporary shelter or other place not designed for sleeping.” Crowley, S. (2003). The affordable housing crisis: Residential mobility of poor families and school mobility of poor children. Journal of Negro Education, 72 (1), 22-38. doi: 10.2307/3211288.
749. The 2023 Annual Homeless Assessment Report (AHAR) to Congress. The U.S. Department of Housing and Urban Development 2023. https://www.huduser.gov/portal/sites/default/files/pdf/2023-AHAR-Part-1.pdf .
750. Baggett, T.P., Hwang, S.W., O'Connell, J.J., Porneala, B.C., Stringfellow, E.J., Orav, E.J., Singer, D.E., Rigotti, N.A. (2013). Mortality among homeless adults in Boston: Shifts in causes of death over a 15-year period. JAMA Internal Medicine, 173(3), 189-195. doi: 10.1001/jamainternmed.2013.1604. Schanzer, B., Dominguez, B., Shrout, P.E., Caton, C.L. (2007). Homelessness, health status, and health care use. American Journal of Public Health, 97(3), 464-469. doi: 10.2105/AJPH.2005.076190.
751. U.S. Department of Health Human Services (HHS), Call to Action, “Addressing Health Related Social Needs in Communities Across the Nation.” November 2023. https://aspe.hhs.gov/sites/default/files/documents/3e2f6140d0087435cc6832bf8cf32618/hhs-call-to-action-health-related-social-needs.pdf .
752. Henderson, K.A., Manian, N., Rog, D.J., Robison, E., Jorge, E., AlAbdulmunem, M. “Addressing Homelessness Among Older Adults” (Final Report). Washington, DC: Office of the Assistant Secretary for Planning and Evaluation, U.S. Department of Health and Human Services. October 26, 2023.
753. More information about the AHC HRSN Screening Tool is available on the website at https://innovation.cms.gov/Files/worksheets/ahcm-screeningtool.pdf .
754. The AHC HRSN Screening Tool Living Situation item includes two questions. In an effort to limit LTCH burden, we only proposed the first question.
755. National Association of Community Health Centers and Partners, National Association of Community Health Centers, Association of Asian Pacific Community Health Organizations, Association OPC, Institute for Alternative Futures. “PRAPARE.” 2017. https://prapare.org/the-prapare-screening-tool/ .
756. U.S. Department of Agriculture, Economic Research Service. (n.d.). Definitions of food security. Retrieved March 10, 2022, from https://www.ers.usda.gov/topics/food-nutrition-assistance/food-security-in-the-u-s/definitions-of-food-security/ .
757. Hernandez, D.C., Reesor, L.M., Murillo, R. (2017). Food insecurity and adult overweight/obesity: Gender and race/ethnic disparities. Appetite, 117, 373-378.
758. Banerjee, S., Radak, T., Khubchandani, J., Dunn, P. (2021). Food Insecurity and Mortality in American Adults: Results From the NHANES-Linked Mortality Study. Health promotion practice, 22(2), 204-214. https://doi.org/10.1177/1524839920945927 .
759. National Center for Health Statistics. (2022, September 6). Exercise or Physical Activity. Retrieved from Centers for Disease Control and Prevention: https://www.cdc.gov/nchs/fastats/exercise.htm .
760. Food and Nutrition Security. (n.d.). USDA. https://www.usda.gov/nutrition-security .
761. Food and Nutrition Service. (March 2022). USDA. https://www.usda.gov/sites/default/files/documents/usda-actions-nutrition-security.pdf .
762. Ziliak, J.P., Gundersen, C. (2019). The State of Senior Hunger in America 2017: An Annual Report. Prepared for Feeding America. Available at: https://www.feedingamerica.org/research/senior-hunger-research/senior .
763. The Malnutrition Quality Collaborative. (2020). National Blueprint: Achieving Quality Malnutrition Care for Older Adults, 2020 Update. Washington, DC: Avalere Health and Defeat Malnutrition Today. Available at: https://defeatmalnutrition.today/advocacy/blueprint/ .
764. Food Research Action Center (FRAC). “Hunger is a Health Issue for Older Adults: Food Security, Health, and the Federal Nutrition Programs.” December 2019. https://frac.org/wp-content/uploads/hunger-is-a-health-issue-for-older-adults-1.pdf .
765. The White House Challenge to End Hunger and Build Health Communities (Challenge) was a nationwide call-to-action released on March 24, 2023 to stakeholders across all of society to make commitments to advance President Biden's goal to end hunger and reduce diet-related diseases by 2030—all while reducing disparities. More information on the White House Challenge to End Hunger and Build Health Communities can be found: https://www.whitehouse.gov/briefing-room/statements-releases/2023/03/24/fact-sheet-biden-harris-administration-launches-the-white-house-challenge-to-end-hunger-and-build-healthy-communities-announces-new-public-private-sector-actions-to-continue-momentum-from-hist/ .
766. Schroeder K, Smaldone A. Food Insecurity: A Concept Analysis. Nurse Forum. 2015 Oct-Dec;50(4):274-84. doi: 10.1111/nuf.12118. Epub 2015 Jan 21. PMID: 25612146; PMCID: PMC4510041.
767. Tsega M, Lewis C, McCarthy D, Shah T, Coutts K. Review of Evidence for Health-Related Social Needs Interventions. July 2019. The Commonwealth Fund. https://www.commwealthfund.org/sites/default/files/2019-07/ROI-EVIDENCE-REVIEW-FINAL-VERSION.pdf .
768. More information about the HFSS tool can be found at https://www.ers.usda.gov/topics/food-nutrition-assistance/food-security-in-the-u-s/survey-tools/ .
769. The AHC HRSN Screening Tool Food item includes two questions. In an effort to limit LTCH burden, we only proposed the first question.
770. Hernández D. Understanding `energy insecurity' and why it matters to health. Soc Sci Med. 2016 Oct; 167:1-10. doi: 10.1016/j.socscimed.2016.08.029. Epub 2016 Aug 21. PMID: 27592003; PMCID: PMC5114037.
771. US Energy Information Administration. “One in Three U.S. Households Faced Challenges in Paying Energy Bills in 2015.” 2017 Oct 13. https://www.eia.gov/consumption/residential/reports/2015/energybills/ .
772. Hernández D. “Understanding `energy insecurity' and why it matters to health.” Soc Sci Med. 2016; 167:1-10.
773. Hernández D. Understanding `energy insecurity' and why it matters to health. Soc Sci Med. 2016 Oct;167:1-10. doi: 10.1016/j.socscimed.2016.08.029. Epub 2016 Aug 21. PMID: 27592003; PMCID: PMC5114037.
774. Hernández D. “What `Merle' Taught Me About Energy Insecurity and Health.” Health Affairs, VOL.37, NO.3: Advancing Health Equity Narrative Matters. March 2018. https://doi.org/10.1377/hlthaff.2017.1413 .
775. US Energy Information Administration. “One in Three U.S. Households Faced Challenges in Paying Energy Bills in 2015.” 2017 Oct 13. https://www.eia.gov/consumption/residential/reports/2015/energybills/ .
776. Hernández D. Understanding `energy insecurity' and why it matters to health. Soc Sci Med. 2016 Oct;167:1-10. doi: 10.1016/j.socscimed.2016.08.029. Epub 2016 Aug 21. PMID: 27592003; PMCID: PMC5114037.
777. Institute of Medicine. (2004). Damp Indoor Spaces and Health. Washington, DC: National Academies Press. http://www.nap.edu/openbook.php?record_id=11011page=R2 .
778. Siegal et al., “Energy Insecurity Indicators Associated with Increased Odds of Respiratory, Mental Health, And Cardiovascular Conditions.” Health Affairs 43, NO. 2 (2024): 260-268. https://doi.org/10.1377/hlthaff.2023.01052 .
779. National Council on Aging (NCOA). “How to Make It Easier for Older Adults to Get Energy and Utility Assistance.” Promising Practices Clearinghouse for Professionals. Jan 13, 2022. https://www.ncoa.org/article/how-to-make-it-easier-for-older-adults-to-get-energy-and-utility-assistance .
780. This validated survey was developed as a clinical indicator of household energy security among pediatric caregivers. Cook, J.T., D.A. Frank., P.H. Casey, R. Rose-Jacobs, M.M. Black, M. Chilton, S. Ettinger de Cuba, et al. “A Brief Indicator of Household Energy Security: Associations with Food Security, Child Health, and Child Development in US Infants and Toddlers.” Pediatrics, vol. 122, no. 4, 2008, pp. e874-e875. https://doi.org/10.1542/peds.2008-0286
781. National Academies of Sciences, Engineering, and Medicine. 2016. Accounting for Social Risk Factors in Medicare Payment: Identifying Social Risk Factors. Washington, DC: The National Academies Press. https://doi.org/10.17226/21858 .
782. National Academies of Sciences, Engineering, and Medicine. 2020. Leading Health Indicators 2030: Advancing Health, Equity, and Well-Being. Washington, DC: The National Academies Press. https://doi.org/10.17226/25682 .
783. Healthy People 2030 Framework. Healthy People 2030. https://health.gov/healthypeople/about/healthy-people-2030-framework .
784. Green K, Zook M. When Talking About Social Determinants, Precision Matters. HealthAffairs. Published October 29, 2019. https://www.healthaffairs.org/do/10.1377/hblog20191025.776011/full/ .
785. https://www.cms.gov/priorities/innovation/data-and-reports/2023/ahc-second-eval-rpt-fg .
786. Billioux, A., Verlander, K., Anthony, S., Alley, D. (2017). Standardized Screening for Health-Related Social Needs in Clinical Settings: The Accountable Health Communities Screening Tool. NAM Perspectives, 7(5). Available at: https://doi.org/10.31478/201705b . Accessed on June 9, 2024.
787. Billioux, A., Verlander, K., Anthony, S., Alley, D. (2017). Standardized Screening for Health-Related Social Needs in Clinical Settings: The Accountable Health Communities Screening Tool. NAM Perspectives, 7(5). Available at: https://doi.org/10.31478/201705b . Accessed on June 9, 2024.
788. Centers for Medicare Medicaid Services. (2021). Accountable Health Communities Model. Accountable Health Communities Model | CMS Innovation Center. Available at: https://innovation.cms.gov/innovation-models/ahcm . Accessed on February 20, 2023.
789. https://www.cms.gov/priorities/innovation/data-and-reports/2023/ahc-second-eval-rpt-fg .
790. Accountable Health Communities (AHC) Model Evaluation, Second Evaluation Report. May 2023. This project was funded by the Centers for Medicare Medicaid Services under contract no. HHSM-500-2014-000371, Task Order75FCMC18F0002. https://www.cms.gov/priorities/innovation/data-and-reports/2023/ahc-second-eval-rpt .
791. In October 2023, we released two new annual Health Equity Confidential Feedback Reports to LTCHs: The Discharge to Community (DTC) Health Equity Confidential Feedback Report and the Medicare Spending Per Beneficiary (MSPB) Health Equity Confidential Feedback Report. The PAC Health Equity Confidential Feedback Reports stratified the DTC and MSPB measures by dual-enrollment status and race/ethnicity. For more information on the Health Equity Confidential Feedback Reports, please refer to the Education and Outreach materials available on the LTCH QRP Training web page at https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-quality-reporting-training .
792. Brooks-LaSure, C. (2021). My First 100 Days and Where We Go from Here: A Strategic Vision for CMS. Centers for Medicare Medicaid. Available at https://www.cms.gov/blog/my-first-100-days-and-where-we-go-here-strategic-vision-cms .
793. The Biden-Harris Administration's strategic approach to addressing health related social needs can be found in The U.S. Playbook to Address Social Determinants of Health (SDOH) (2023): https://www.whitehouse.gov/wp-content/uploads/2023/11/SDOH-Playbook-3.pdf .
794. Social Determinants of Health. Healthy People 2020. https://www.healthypeople.gov/2020/topics-objectives/topic/social-determinants-of-health . (February 2019).
795. National Academies of Sciences, Engineering, and Medicine. 2020. Leading Health Indicators 2030: Advancing Health, Equity, and Well-Being. Washington, DC: The National Academies Press. https://doi.org/10.17226/25682 .
796. The CMS Data Element Library (DEL) is the centralized resource for CMS assessment instrument data elements ( e.g., questions and responses) and their associated health information technology (IT) standards. https://del.cms.gov/DELWeb/pubHome .
797. These cue cards are currently available on the LTCH QRP Training web page at https://www.cms.gov/medicare/quality/inpatient-rehabilitation-facility/LTCH-quality-reporting-training .
798. Haff, N, Choudhry, N.K., Bhatkhande, G., Li, Y., Antol, D., Renda, A., Laufffenburger, J. Frequency of Quarterly Self-reported Health-Related Social Needs Among Older Adults, 2020. JAMA Network Open. 2022;5(6):e2219645. Doi:101001/jamanetworkopen.2022.19645. Accessed June 9, 2024.
799. Haff, N, Choudhry, N.K., Bhatkhande, G., Li, Y., Antol, D., Renda, A., Laufffenburger, J. Frequency of Quarterly Self-reported Health-Related Social Needs Among Older Adults, 2020. JAMA Network Open. 2022;5(6):e2219645. Doi:101001/jamanetworkopen.2022.19645. Accessed June 9, 2024.
800. The seven SDOH items are ethnicity, race, preferred language, interpreter services, health literacy, transportation, and social isolation ( 84 FR 42577 through 42579 ).
801. Centers for Medicare Medicaid Services, FY2024 Inpatient Psychiatric Prospective Payment System—Rate Update ( 88 FR 51107 through 51121 ).
802. Centers for Medicare Medicaid Services, FY2023 IPPS/LTCH PPS final rule ( 87 FR 49202 through 49215 ).
803. Centers for Medicare Medicaid Services, FY2024 Inpatient Psychiatric Prospective Payment System—Rate Update ( 88 FR 51107 through 51121 ).
804. In the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 36350 ), we inadvertently stated here that a draft of the proposed Living Situation item could be found at the LCDS and LTCH Manual web page. We have corrected this typographical error here in this final rule to refer to the proposed modified Transportation item.
805. Haff, N, Choudhry, N.K., Bhatkhande, G., Li, Y., Antol, D., Renda, A., Laufffenburger, J. Frequency of Quarterly Self-reported Health-Related Social Needs Among Older Adults, 2020. JAMA Network Open. 2022;5(6):e2219645. Doi:101001/jamanetworkopen.2022.19645. Accessed June 9, 2024.
806. The Post-Acute Care (PAC) and Hospice Quality Reporting Program Cross-Setting TEP summary report will be published in early summer or as soon as technically feasible. LTCHs can monitor the Partnership for Quality Measurement website at https://mmshub.cms.gov/get-involved/technical-expert-panel/updates for updates.
807. Centers for Medicare Medicaid Services. Aligning Quality Measures Across CMS—the Universal Foundation. November 17, 2023. https://www.cms.gov/aligning-quality-measures-across-cms-universal-foundation
808. A composite measure can summarize multiple measures through the use of one value or one piece of information. More information can be found at https://www.cms.gov/medicare/quality-initiatives-patient-assessment-instruments/mms/downloads/composite-measures.pdf .
809. CMS Measures Inventory Tool. Adult immunization status measure found at https://cmit.cms.gov/cmit/#/FamilyView?familyId=26 .
810. CMS Measure Inventory Tool. Clinical Depression Screening and Follow-Up measure found at https://cmit.cms.gov/cmit/#/FamilyView?familyId=672 .
811. Centers for Medicare Medicaid Services. Aligning Quality Measures Across CMS—the Universal Foundation. November 17, 2023. https://www.cms.gov/aligning-quality-measures-across-cms-universal-foundation .
812. The Patient Health Questionnaire (PHQ-2 to-9) is a validated interview for symptoms of depression. It provides a standardized severity score and a rating for evidence of a depression disorder. Chapter 3, Section D, LCDS Manual.
813. Principal Illness Navigation (PIN) services describe services that auxiliary personnel, including care navigators or peer support specialists, may perform incidental to the professional services of a physician or other billing practitioner, under general supervision. Two codes describe PIN services, and two codes describe Principal Illness Navigation-Peer Support (PIN-PS) services, which are intended more for patients with high-risk behavioral health conditions and have slightly different service elements that better describe the scope of practice of peer support specialists. In general, where we describe aspects of PIN, it also applies to PIN-PS unless otherwise specified. MLN9201074 January 2024. https://www.cms.gov/files/document/mln9201074-health-equity-services-2024-physician-fee-schedule-final-rule.pdf-0
814. The Patient Safety Structural Measure (PSSM) is proposed for the Hospital IQR Program and PPS-Exempt Cancer Hospital Quality Reporting (PCHQR) Program in the FY 2025 IPPS/LTCH PPS proposed rule ( 89 FR 35937 through 35938 ). It is an attestation-based measure that assesses whether hospitals have a structure and culture that prioritizes safety as demonstrated by the following five domains: (1) leadership commitment to eliminating preventable harm; (2) strategic planning and organizational policy; (3) culture of safety and learning health system; (4) accountability and transparency; and (5) patient and family engagement.
815. The Universal Foundation PAC Add-On Set can be found at: https://www.cms.gov/medicare/quality/cms-national-quality-strategy/aligning-quality-measures-across-cms-universal-foundation .
816. Improving Medicare Post-Acute Care Transformation Act of 2014. Public Law 113-185 —OCT. 6, 2014 https://www.govinfo.gov/content/pkg/PLAW-113publ185/pdf/PLAW-113publ185.pdf
817. FY 2020 IPPS/LTCH PPS final rule ( 84 FR 42588 through 42590 ).
818. Due to data availability of LTCH SDOH standardized patient assessment data elements, this is based on three quarters of Transportation data.
819. The analysis is limited to patients who responded to the Transportation item at both admission and discharge.
820. Office of the Federal Register of the National Archives and Records Administration and the U.S. Government Publishing Office. Medicare Program; Hospital Inpatient Prospective Payment Systems for Acute Care Hospitals and the Long-Term Care Hospital Prospective Payment System and FY 2012 Rates; Hospitals' FTE Resident Caps for Graduate Medical Education Payment. 2011. https://www.federalregister.gov/d/2011-19719/p-3517 .
821. Centers for Medicare Medicaid Services (CMS). Long-Term Care Hospital (LTCH) Continuity Assessment Record and Evaluation (CARE) Data Set (LCDS) LCDS Manual. 2023. https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-care-data-set-ltch-qrp-manual .
822. A summary of the LTCH Listening Session can be found on the LTCH QRP Measures Information web page at: https://www.cms.gov/medicare/quality/long-term-care-hospital/ltch-quality-reporting-measures-information .
823. https://www.cdc.gov/nhsn/psc/aur/index.html .
824. https://www.cdc.gov/nhsn/pdfs/pscmanual/11pscaurcurrent.pdf .
825. For more information, see: https://www.healthit.gov/topic/laws-regulation-and-policy/health-data-technology-and-interoperability-certification-program .
826. https://www.healthit.gov/topic/safety/safer-guides .
827. https://www.healthit.gov/isa/united-states-core-data-interoperability-uscdi#uscdi-v3 .
828. https://csrc.nist.gov/pubs/sp/800/66/r2/final .
829. https://aspr.hhs.gov/cyber/Documents/Health-Care-Sector-Cybersecurity-Dec2023-508.pdf .
830. https://hphcyber.hhs.gov/performance-goals.html .
831. Innovation Center Strategy Refresh: https://www.cms.gov/priorities/innovation/strategic-direction-whitepaper .
832. The CMS Innovation Center's Strategy to Support Person-centered, Value-based Specialty Care: https://www.cms.gov/blog/cms-innovation-centers-strategy-support-person-centered-value-based-specialty-care .
833. https://www.cms.gov/blog/cms-innovation-centers-strategy-support-person-centered-value-based-specialty-care .
834. https://www.federalregister.gov/documents/2023/07/18/2023-15169/request-for-information-episode-based-payment-model .
835. Papanicolas, I., Woskie, L., Jha, A.K. (2018). Health care spending in the United States and other High-Income countries. JAMA, 319(10), 1024. https://doi.org/10.1001/jama.2018.1150 .
836. Timmins, L., Urato, C., Kern, L.M., Ghosh, A., Rich, E.C. (2022). Primary care redesign and care fragmentation among Medicare beneficiaries. The American Journal of Managed Care, 28(3), e103-e112. https://doi.org/10.37765/ajmc.2022.88843 .
837. The Center for Healthcare Research Transformation. (2013). Payment Strategies: A Comparison of Episodic and Population-based Payment Reform. Retrieved November 14, 2023, from https://www.chrt.org/wp-content/uploads/2013/11/CHRT_Payment-Strategies-A-Comparison-of-Episodic-and-Population-based-Payment-Reform-.pdf .
838. https://www.cms.gov/priorities/innovation/data-and-reports/2022/wp-eval-synthesis-21models .
839. Fry, D.E., Pine, M., Jones, B., Meimban, R.J. (2011). The impact of ineffective and inefficient care on the excess costs of elective surgical procedures. Journal of the American College of Surgeons, 212(5), 779-786. https://doi.org/10.1016/j.jamcollsurg.2010.12.046 .
840. Justiniano, C.F., Xu, Z., Becerra, A.Z., Aquina, C.T., Boodry, C.I., Swanger, A.A., Temple, L.K., Fleming, F.J. (2017). Long-term deleterious impact of surgeon care fragmentation after colorectal surgery on survival: Continuity of care continues to count. Diseases of the Colon Rectum, 60(11), 1147-1154. https://doi.org/10.1097/dcr.0000000000000919 .
841. Tsai, T.C., Orav, E.J., Jha, A.K. (2015). Care fragmentation in the postdischarge period. JAMA Surgery, 150(1), 59. https://doi.org/10.1001/jamasurg.2014.2071 .
842. Tsai, T.C., Greaves, F., Zheng, J., Orav, E.J., Zinner, M.J., Jha, A.K. (2016). Better patient care at High-Quality hospitals may save Medicare money and bolster Episode-Based payment models. Health Affairs, 35(9), 1681-1689. https://doi.org/10.1377/hlthaff.2016.0361 .
843. Scally, C.P., Thumma, J.R., Birkmeyer, J.D., Dimick, J.B. (2015). Impact of surgical quality improvement on payments in Medicare patients. Annals of Surgery, 262(2), 249-252. https://doi.org/10.1097/sla.0000000000001069 .
844. Ljungqvist, O., Scott, M.J., Fearon, K.C.H. (2017). Enhanced recovery after surgery. JAMA Surgery, 152(3), 292. https://doi.org/10.1001/jamasurg.2016.4952 .
845. Cline, K.M., Clement, V., Rock-Klotz, J., Kash, B.A., Steel, C.A., Miller, T.R. (2020). Improving the cost, quality, and safety of perioperative care: A systematic review of the literature on implementation of the perioperative surgical home. Journal of Clinical Anesthesia, 63, 109760. https://doi.org/10.1016/j.jclinane.2020.109760 .
846. Agarwal, R., Liao, J.M., Gupta, A., Navathe, A.S. (2020). The Impact of bundled payment on health care spending, utilization, and quality: A Systematic review. Health Affairs, 39(1), 50-57. https://doi.org/10.1377/hlthaff.2019.00784 .
847. Steenhuis, S., Struijs, J.N., Koolman, X., Ket, J.C.F., Van Der Hijden, E. (2020). Unraveling the complexity in the design and implementation of bundled payments: A scoping review of key elements from a payer's perspective. The Milbank Quarterly, 98(1), 197-222. https://doi.org/10.1111/1468-0009.12438 .
848. Sutherland, A., Boudreau, E., Bowe, A., Huang, Q., Liao, J.M., Flagg, M., Cousins, D., Antol, D.D., Shrank, W.H., Powers, B., Navathe, A.S. (2023). Association between a bundled payment program for lower extremity joint replacement and patient outcomes among Medicare Advantage beneficiaries. JAMA Health Forum, 4(6), e231495. https://doi.org/10.1001/jamahealthforum.2023.1495 .
849. Medicare Acute Care Episode Demonstration ( https://innovation.cms.gov/innovation-models/ace ), BPCI Initiative ( https://www.cms.gov/priorities/innovation/innovation-models/bundled-payments ), BPCI Advanced Model ( https://www.cms.gov/priorities/innovation/innovation-models/bpci-advanced ), CJR Model ( https://www.cms.gov/priorities/innovation/innovation-models/CJR ).
850. Evaluation of the Medicare Acute Care Episode (ACE) Demonstration. (2013). Centers for Medicare Medicaid Services. Retrieved December 1, 2023, from http://downloads.cms.gov/files/cmmi/ACE-EvaluationReport-Final-5-2-14.pdf .
851. Evaluation and Monitoring of the Bundled Payments for Care Improvement Model 1 Initiative. (2016). Centers for Medicare Medicaid Services. Retrieved December 1, 2023, from https://www.cms.gov/priorities/innovation/files/reports/bpci-mdl1yr2annrpt.pdf .
852. CMS Bundled Payments for Care Improvement Initiative Models 2-4: Year 7 Evaluation Monitoring Annual Report. (2021). Centers for Medicare Medicaid Services. Retrieved December 1, 2023, from https://www.cms.gov/priorities/innovation/data-and-reports/2021/bpci-models2-4-yr7evalrpt .
853. CMS Bundled Payments for Care Improvement Initiative Models 2-4: Year 7 Evaluation Monitoring Annual Report. (2021). Centers for Medicare Medicaid Services. Retrieved December 1, 2023, from https://www.cms.gov/priorities/innovation/data-and-reports/2021/bpci-models2-4-yr7evalrpt .
854. CMS Bundled Payments for Care Improvement Advanced Model: Fourth Evaluation Report. (2023). Centers for Medicare Medicaid Services. Retrieved December 1, 2023, from https://www.cms.gov/priorities/innovation/data-and-reports/2023/bpci-adv-ar4 .
855. CMS Comprehensive Care for Joint Replacement Model: Performance Year 5 Evaluation Report. (2023). Centers for Medicare Medicaid Services. Retrieved December 1, 2023, from https://www.cms.gov/priorities/innovation/data-and-reports/2023/cjr-py5-annual-report .
856. CMS Innovation Center's Specialty Care Strategy Listening Session ( https://www.cms.gov/priorities/innovation/media/document/spec-care-listening-session-slides ).
857. Centers for Medicare Medicaid Services. (2021). Innovation Center Strategy Refresh. https://www.cms.gov/priorities/innovation/strategic-direction-whitepaper .
858. The CMS Innovation Center's strategy to support person-centered, value-based specialty care | CMS. (2022). https://www.cms.gov/blog/cms-innovation-centers-strategy-support-person-centered-value-based-specialty-care#_ftn1 .
859. https://www.federalregister.gov/public-inspection/2024-14828/medicare-and-medicaid-programs-calendar-year-2025-payment-policies-under-the-physician-fee-schedule .
860. https://www.cms.gov/priorities/innovation/data-and-reports/2022/wp-eval-synthesis-21models .
861. https://www.cms.gov/priorities/innovation/data-and-reports/2023/cjr-py5-annual-report .
862. https://www.cms.gov/priorities/innovation/data-and-reports/2024/bpci-adv-ar5 .
863. CMS Bundled Payments for Care Improvement Advanced Model: Third Evaluation Report. (2022). Centers for Medicare Medicaid Services. Retrieved November 28, 2023, from https://www.cms.gov/priorities/innovation/data-and-reports/2022/bpci-adv-ar3 .
864. CMS Bundled Payments for Care Improvement Advanced Model: Year 2 Evaluation Report. (2021). Centers for Medicare Medicaid Services. Retrieved November 28, 2023, from https://www.cms.gov/priorities/innovation/data-and-reports/2021/bpci-yr2-annual-report .
865. https://www.cms.gov/files/document/mln006400-information-critical-access-hospitals.pdf .
866. https://www.federalregister.gov/public-inspection/2024-14828/medicare-and-medicaid-programs-calendar-year-2025-payment-policies-under-the-physician-fee-schedule .
867. Erikson, C., Pittman, P., LaFrance, A., Chapman, S. (2017). Alternative payment models lead to strategic care coordination workforce investments. Nursing Outlook, 65(6), 737-745. https://doi.org/10.1016/j.outlook.2017.04.001 .
868. Current BPCI Advanced hospitals would need to participate in BPCI Advanced until December 31, 2025 and current CJR participant hospitals would need to participate in the CJR model until December 31, 2024.
869. https://www.cbo.gov/system/files/2023-09/59274-CMMI.pdf .
870. https://www.medpac.gov/wp-content/uploads/2022/06/Jun22_MedPAC_Report_to_Congress_v4_SEC.pdf .
871. https://www.cms.gov/medicare/quality/value-based-programs/hospital-purchasing .
872. https://www.cms.gov/priorities/innovation/data-and-reports/2024/bpci-adv-ar5 .
873. https://www.cbo.gov/publication/59612 .
874. https://www.cms.gov/priorities/innovation/data-and-reports/2024/bpci-adv-ar5 .
875. https://www.cms.gov/priorities/innovation/data-and-reports/2020/bpciadvanced-firstannevalrpt .
876. https://www.healthaffairs.org/content/forefront/cms-innovation-center-s-strategy-support-person-centered-value-based-specialty-care .
877. For the BPCI Advanced model, the last day of the last performance period, performance period 14, is December 31, 2025. For the CJR model, the last day of the last performance year, performance year 8, is December 31, 2024.
878. Termination from BPCI Advanced includes the BPCI Advanced participant voluntarily terminating their BPCI Advanced participation agreement or CMS terminating the BPCI Advanced participation agreement with the BPCI Advanced participant.
879. CMS Bundled Payments for Care Improvement Advanced Model: Year 2 Evaluation Report. (2021). Centers for Medicare Medicaid Services. Retrieved November 28, 2023, from https://www.cms.gov/priorities/innovation/data-and-reports/2021/bpci-yr2-annual-report .
880. Medicare Information on the Transition to Alternative Payment Models by Providers in Rural, Health Professional Shortage, or Underserved Areas: Report to Congressional Committees. (2021). United States Government Accountability Office. Retrieved December 1, 2023, from https://www.gao.gov/assets/gao-22-104618.pdf .
881. This list was generated using the criteria and methods that are being proposed, and is subject to change if different criteria and methods end up being finalized.
882. See the technical resources section of the following web page on how these episode categories were constructed: https://www.cms.gov/priorities/innovation/innovation-models/bpci-advanced/participant-resources .
883. For the BPCI Advanced model, the last day of the last performance period, performance period 14, is December 31, 2025. For the CJR model, the last day of the last performance year, performance year 8, is December 31, 2024.
884. FY 2025 ICD-10 MS-DRG Definitions Manual Version 42. Available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
885. MedPAC March 2021 Report to the Congress. https://www.medpac.gov/ .
886. FY 2025 ICD-10 MS-DRG Definitions Manual Version 42. Available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
887. FY 2025 ICD-10 MS-DRG Definitions Manual Version 42. Available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
888. FY 2025 ICD-10 MS-DRG Definitions Manual Version 42. Available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
889. FY 2025 ICD-10 MS-DRG Definitions Manual Version 42. Available at: https://www.cms.gov/medicare/payment/prospective-payment-systems/acute-inpatient-pps/ms-drg-classifications-and-software .
890. MedPAC March 2021 Report to the Congress. https://www.medpac.gov/ .
891. A complete list of excluded items, services, and readmission MS-DRGs can be found in the “BPCI Advanced Exclusions List—MY7 (XLS)” available under Participant Resources at the CMS BPCI Advanced website.
892. MDCs are formed by dividing all possible principal diagnoses (from ICD-10-CM) into 25 mutually exclusive diagnosis areas. The diagnoses in each MDC correspond to a single organ system or etiology and in general are associated with a particular medical specialty.
893. This exclusion is applied during the payment standardization process.
894. To determine if a drug HCPCS code meets the cost or volume thresholds for exclusion, the episodes are pooled across all episode categories.
895. https://www.cms.gov/medicare/payment/all-fee-service-providers/medicare-part-b-drug-average-sales-price/asp-pricing-files .
896. MCC: major complications or comorbidities.
897. CC: complication or comorbidity.
898. Damberg CL et al., Research Report: Measuring Success in Health Care Value-Based Purchasing Programs, Summary and Recommendations. RAND (2014). Available from http://www.rand.org/content/dam/rand/pubs/research_reports/RR300/RR306z1/RAND_RR306z1.pdf . http://www.rand.org/content/dam/rand/pubs/research_reports/RR300/RR306z1/RAND_RR306z1.pdf .
899. https://www.cms.gov/medicare-coverage-database/view/ncacal-decision-memo.aspx?proposed=NNCAId=288 .
900. Centers for Medicare Medicaid Services. (December 1, 2023). 2023 Measures Under Consideration (MUC) List. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsxhttps://mmshub.cms.gov/sites/default/files/2023-MUC-List.xlsx .
901. Centers for Medicare Medicaid Services. (December 2023). Overview of the List of Measures Under Consideration. Available at: https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf https://mmshub.cms.gov/sites/default/files/2023-MUC-List-Overview.pdf .
902. Frankl SE, Breeling JL, Goldman L. Preventability of emergent hospital readmission. American Journal of Medicine. Jun 1991;90(6):667-674.
903. https://qualitynet.cms.gov/inpatient/measures/hybrid/methodology .
904. Keenan PS, Normand SL, Lin Z, Drye EE, Bhat KR, Ross JS, et al. An administrative claims measure suitable for profiling hospital performance on the basis of 30-day all-cause readmission rates among patients with heart failure. Circ Cardiovasc Qual Outcomes. 2008 Sep;1(1):29-37. PubMed PMID: 20031785. Epub 2008/09/01. eng.
905. Krumholz HM, Lin Z, Drye EE, Desai MM, Han LF, Rapp MT, et al. An administrative claims measure suitable for profiling hospital performance based on 30-day all-cause readmission rates among patients with acute myocardial infarction. Circ Cardiovasc Qual Outcomes. 2011 Mar;4(2):243-52. PubMed PMID: 21406673. PMCID: PMC3350811. Epub 2011/03/17. eng.
906. Adverse Effects of the Medicare PSI 90 Hospital Penalty System on Revenue-Neutral Hospital-Acquired Conditions (Jun 2020).
907. Zhang, Y. and J.M. Jordan, Epidemiology of osteoarthritis. Clin Geriatr Med, 2010. 26(3): p. 355-69.
908. Centers for Disease Control and Prevention. Osteoarthritis (OA). 2020; Available from: https://www.cdc.gov/arthritis/basics/osteoarthritis.htm .
909. Guccione, A.A., et al., The effects of specific medical conditions on the functional limitations of elders in the Framingham Study. Am J Public Health, 1994. 84(3): p. 351-8.
910. Michaud, C.M., et al., The burden of disease and injury in the United States 1996. Popul Health Metr, 2006. 4: p. 11.
911. Levit, K., et al. HCUP Facts and Figures, 2006: Statistics on Hospital-based Care in the United States. 2008; Available from: https://www.hcupus.ahrq.gov/reports/factsandfigures/facts_figures_2006.jsp .
912. Healthcare Cost and Utilization Project. HCUP Fast Stats—Most Common Diagnoses for Inpatient Stays 2021; Available from: https://www.hcupus.ahrq.gov/faststats/NationalDiagnosesServlet?year1=2018characteristic1=0included1=1year2=2017characteristic2=0included2=1expansionInfoState=hidedataTablesState=hidedefinitionsState=hideexportState=hide .
913. Liang, L., B. Moore, and A. Soni, National Inpatient Hospital Costs: The Most Expensive Conditions by Payer, 2017. HCUP Statistical Brief #261. 2020.
914. Lopez, C.D., et al., Hospital and Surgeon Medicare Reimbursement Trends for Total Joint Arthroplasty. Arthroplast Today, 2020. 6(3): p. 437-444.
915. Rissanen, P., et al., Health and quality of life before and after hip or knee arthroplasty. The Journal of Arthroplasty, 1995. 10(2): p. 169-175.
916. Lopez, C.D., et al., Hospital and Surgeon Medicare Reimbursement Trends for Total Joint Arthroplasty. Arthroplast Today, 2020. 6(3): p. 437-444.
917. Mintz, J., Duprey, M.S., Zullo, A.R., Lee, Y., Kiel, D.P., Daiello, L.A., Rodriguez, K.E., Venkatesh, A.K., Berry, S.D. (2022). Identification of Fall-Related Injuries in Nursing Home Residents Using Administrative Claims Data. The journals of gerontology. Series A, Biological sciences and medical sciences, 77(7), 1421-1429. https://doi.org/10.1093/gerona/glab274 .
918. Network of Patient Safety Databases Chartbook, 2022. Rockville, MD: Agency for Healthcare Research and Quality; September 2022. AHRQ Pub. No. 22-0051.
919. National Quality Forum. Serious Reportable Events. http://www.qualityforum.org/topics/sres/serious_reportable_events.aspx . Accessed July 24, 2019.
920. WHO. (2009). Conceptual Framework for the International Classification for Patient Safety, Version 1.1. https://apps.who.int/iris/bitstream/handle/10665/70882/WHO_IER_PSP_2010.2_eng.pdf .
921. Arozullah AM, Daley J, Henderson WG, Khuri SF. (2000). Multifactorial risk index for predicting postoperative respiratory failure in men after major noncardiac surgery. The National Veterans Administration Surgical Quality Improvement Program. Annals of surgery. 232(2):242-253.
922. Arozullah AM, Daley J, Henderson WG, Khuri SF. (2000). Multifactorial risk index for predicting postoperative respiratory failure in men after major noncardiac surgery. The National Veterans Administration Surgical Quality Improvement Program. Annals of surgery. 232(2):242-253.
923. Miller MR, Elixhauser A, Zhan C, Meyer GS. (2001). Patient Safety Indicators: using administrative data to identify potential patient safety concerns. Health services research. 36(6 Pt 2):110-132.
924. Thompson SL, Lisco SJ. Postoperative Respiratory Failure. Int Anesthesiol Clin. 2018;56(1):147-164.
925. Rahman M, Neal D, Fargen KM, Hoh BL. Establishing standard performance measures for adult brain tumor patients: a Nationwide Inpatient Sample database study. Neuro Oncol. 2013;15(11):1580-1588.
926. https://www.cms.gov/priorities/innovation/innovation-models/bpci-advanced/participant-resources
927. Carey, K., Lin, M-Y. (2022). Safety-net hospital performance under Comprehensive Care for Joint Replacement. Health Services Research, 2022(1-6). https://doi:10.1111/1475-6773.14042 .
928. Shashikumar, S.A., Ryan, A.M., Joynt Maddox, K.E. (2022). Equity implications of hospital penalties during 4 years of the Comprehensive Care for Joint Replacement Model, 2016 to 1019. JAMA Health Forum, 3(12). https://doi.org/10.1001/jamahealthforum.2022.4455 .
929. Kind, A., Jencks, S., Brock, J. E., Yu, M., Bartels, C. M., Ehlenbach, W. J., Greenberg, C., Smith, M. (2014). Neighborhood socioeconomic disadvantage and 30-Day rehospitalization. Annals of Internal Medicine, 161(11), 765. https://doi.org/10.7326/m13-2946 .
930. Díaz, A., Lindau, S. T., Obeng‐Gyasi, S., Dimick, J. B., Scott, J. W., Ibrahim, A. M. (2023). Association of hospital quality and neighborhood deprivation with mortality after inpatient surgery among Medicare beneficiaries. JAMA Network Open, 6(1), e2253620. https://doi.org/10.1001/jamanetworkopen.2022.53620 .
931. Bose, S., Dun, C., Zhang, G. Q., Walsh, C., Makary, M. A., Hicks, C. W. (2022). Medicare beneficiaries in disadvantaged neighborhoods increased telemedicine use during the COVID-19 pandemic. Health Affairs, 41(5), 635-642. https://doi.org/10.1377/hlthaff.2021.01706 .
932. Tung, E. L., Peek, M. E., Rivas, M., Yang, J. P., Volerman, A. (2021). Association of neighborhood disadvantage with racial disparities in COVID-19 positivity in Chicago. Health Affairs, 40(11), 1784-1791. https://doi.org/10.1377/hlthaff.2021.00695 .
933. Durfey, S. N. M., Kind, A., Gutman, R., Monteiro, K., Buckingham, W. R., DuGoff, E. H., Trivedi, A. N. (2018). Impact of risk adjustment for socioeconomic status on Medicare Advantage Plan quality Rankings. Health Affairs, 37(7), 1065-1072. https://doi.org/10.1377/hlthaff.2017.1509 .
934. https://www.cms.gov/medicare/payment/prospective-payment-systems/provider-specific-data-public-use-text-format .
935. Benefits and Challenges of Payment Adjustments Based on Beneficiaries' Ability to Perform Daily Tasks (GAO-18-588). (2018). United States Government Accountability Office. https://www.gao.gov/assets/gao-18-588.pdf .
936. Bakaeen, F. G., Akras, Z., Svensson, L. G. (2018). Redo coronary artery bypass grafting. Indian journal of thoracic and cardiovascular surgery, 34 (Suppl 3), 272-278. https://doi.org/10.1007/s12055-018-0651-1 .
937. Medicare Claims Maturity: CCW White Paper accessed at https://www2.ccwdata.org/web/guest/white-papers?p_l_back_url=%2Fweb%2Fguest%2Fsearch%3Fq%3Dmedicare%2Bclaims%2Bmaturity . on Jan, 26, 2024 https://www2.ccwdata.org/web/guest/white-papers?p_l_back_url=%2Fweb%2Fguest%2Fsearch%3Fq%3Dmedicare%2Bclaims%2Bmaturity .
938. Currently, the BPCI Advanced model does not allow overlap with the ACO Realizing Equity, Access, and Community Health (ACO REACH) model, the Vermont Medicare ACO Initiative, and the Comprehensive Kidney Care Contracting (CKCC) Options of the Kidney Care Choices (KCC) Model. The CJR model does not allow overlap with the ENHANCED Track of the Medicare Shared Savings Program.
939. The Medicare Shared Savings Program benchmark updates include retrospective county-level trends that implicitly reflect BPCI Advanced and CJR spending changes; such methodology helps mitigate potential overlap of federal outlays.
940. The CJR model only allows overlap with the BASIC track of the Medicare Shared Savings Program.
941. Navathe, A.S., Liao, J.M., Wang, E., Isidro, U., Zhu, J., Cousins, D., Werner, R.M. (2021). Association of patient outcomes with bundled payments among hospitalized patients attributed to accountable care organizations. JAMA Health Forum, 2(8), e212131. https://doi.org/10.1001/jamahealthforum.2021.2131 .
942. Shadow bundles are claims data for services, supplies, and their associated payments grouped into discrete procedural- and/or condition-specific episodes of care. Episodes are constructed based on a consistent set of rules for ACO-attributed beneficiaries who meet the criteria to trigger an episode. Target prices are incorporated to measure performance and provide opportunity for sharing savings with providers.
943. https://www.federalregister.gov/documents/2021/01/25/2021-01753/advancing-racial-equity-and-support-for-underserved-communities-through-the-federal-government .
944. 88 FR 10825 (February 22, 2023) ( https://www.federalregister.gov/documents/2023/02/22/2023-03779/further-advancing-racial-equity-and-support-for-underserved-communities-through-the-federal ).
945. https://www.cms.gov/sites/default/files/2022-04/Health%20Equity%20Pillar%20Fact%20Sheet_1.pdf .
946. de Jager E, Levine AA, Udyavar NR, et al. Disparities in Surgical Access: A Systematic Literature Review, Conceptual Model, and Evidence Map. J Am Coll Surg. 2019;228(3):276-298. doi:10.1016/j.jamcollsurg.2018.12.028 https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6391739/https://www.ncbi.nlm.nih.gov/pmc/articles/PMC6391739/ .
947. Tsai TC, Orav EJ, Joynt KE. Disparities in surgical 30-day readmission rates for Medicare beneficiaries by race and site of care. Ann Surg. 2014;259(6):1086-1090. doi:10.1097/SLA.0000000000000326. https://pubmed.ncbi.nlm.nih.gov/24441810/ https://pubmed.ncbi.nlm.nih.gov/24441810/ .
948. Paredes AZ, Hyer JM, Diaz A, Tsilimigras DI, Pawlik TM. Examining healthcare inequities relative to United States safety net hospitals. Am J Surg. 2020;220(3):525-531. doi:10.1016/j.amjsurg.2020.01.044 https://pubmed.ncbi.nlm.nih.gov/32014296/ .
949. Paro A, Hyer JM, Diaz A, Tsilimigras DI, Pawlik TM. Profiles in social vulnerability: The association of social determinants of health with postoperative surgical outcomes. Surgery. 2021;170(6):1777-1784. doi:10.1016/j.surg.2021.06.001 https://pubmed.ncbi.nlm.nih.gov/34183179/https://pubmed.ncbi.nlm.nih.gov/34183179/ .
950. https://www.cms.gov/sites/default/files/2022-04/Health%20Equity%20Pillar%20Fact%20Sheet_1.pdf .
951. https://www.healthaffairs.org/content/forefront/advancing-health-equity-through-cms-innovation-center-first-year-progress-and-s-come .
952. https://www.ncbi.nlm.nih.gov/books/NBK224519/ .
953. https://www.ncbi.nlm.nih.gov/books/NBK224521/ .
954. https://www.medpac.gov/wp-content/uploads/2022/06/Jun22_MedPAC_Report_to_Congress_v2_SEC.pdf .
955. The June 2022 Report sets forth a conceptual framework for identifying safety-net hospitals and a rationale for better-targeted Medicare funding for such hospitals through a new Medicare Safety-Net Index (MSNI), as discussed in more detail later in this request for information. In its March 2023 Report to Congress, MedPAC discusses its recommendation to Congress to redistribute disproportionate share hospital and uncompensated care payments through the MSNI: https://www.medpac.gov/wp-content/uploads/2023/03/Mar23_MedPAC_Report_To_Congress_SEC.pdf .
956. https://www.medpac.gov/wp-content/uploads/2022/06/Jun22_MedPAC_Report_to_Congress_v2_SEC.pdf .
957. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC3272769/ .
958. https://www.healthaffairs.org/do/10.1377/forefront.20180503.138516/full/ .
959. https://www.cms.gov/priorities/innovation/data-and-reports/2022/cmmi-strategy-refresh-imp-tech-report .
960. MedPAC. “March 2023 Report to Congress: Medicare Payment Policy, Chapter 3”. https://www.medpac.gov/document/chapter-3-hospital-inpatient-and-outpatient-services-march-2023-report/https://www.medpac.gov/document/chapter-3-hospital-inpatient-and-outpatient-services-march-2023-report/ .
961. The most recent available cost report data for this purpose generally lags 4 years behind the rulemaking year (for example, FY 2020 cost report data are available for this FY 2024 proposed rule.)
962. Report: “Landscape of Area-Level Deprivation Measures and Other Approaches to Account for Social Risk and Social Determinants of Health in Health Care Payments.” Accessed at https://aspe.hhs.gov/reports/area-level-measures-account-sdoh on September 27, 2022.
963. https://www.neighborhoodatlas.medicine.wisc.edu/ .
964. Kind AJ, et al., “Neighborhood socioeconomic disadvantage and 30-day rehospitalization: a retrospective cohort study.” Annals of Internal Medicine. No. 161(11), pp 765-74, doi: 10.7326/M13-2946 (December 2, 2014), available at https://www.acpjournals.org/doi/epdf/10.7326/M13-2946 .
965. Jencks SF, et al., “Safety-Net Hospitals, Neighborhood Disadvantage, and Readmissions Under Maryland's All-Payer Program.” Annals of Internal Medicine. No. 171, pp 91-98, doi:10.7326/M16-2671 (July 16, 2019), available at https://www.acpjournals.org/doi/epdf/10.7326/M16-2671 .
966. Khlopas A, et al., “Neighborhood Socioeconomic Disadvantages Associated With Prolonged Lengths of Stay, Nonhome Discharges, and 90-Day Readmissions After Total Knee Arthroplasty.” The Journal of Arthroplasty. No. 37(6), pp S37-S43, doi: 10.1016/j.arth.2022.01.032 (June 2022), available at https://www.sciencedirect.com/science/article/pii/S0883540322000493 .
967. https://www.cms.gov/Medicare-Medicaid-Coordination/Medicare-and-Medicaid-Coordination/Medicare-Medicaid-Coordination-Office/Downloads/MMCO_Factsheet.pdf .
968. https://www.cms.gov/Medicare-Medicaid-Coordination/Medicare-and-Medicaid-Coordination/Medicare-Medicaid-Coordination-Office/Downloads/NationalProfile_2012.pdf .
969. https://aspe.hhs.gov/reports/report-congress-social-risk-factors-performance-under-medicares-value-based-purchasing-programs .
970. https://www.cms.gov/files/document/bpcia-model-trg-price-specs-my7.pdf .
971. National Academies of Sciences, Engineering, and Medicine. 2017. Accounting for social risk factors in Medicare payment. Washington, DC: The National Academies Press. doi: 10.17226/23635.
972. Petterson S. Deciphering the Neighborhood Atlas Area Deprivation Index: the consequences of not standardizing. Health Aff Sch. 2023;1(5):qxad063. Published 2023 Nov 3. doi:10.1093/haschl/qxad063.
973. https://www.medpac.gov/wp-content/uploads/2022/06/Jun22_MedPAC_Report_to_Congress_v2_SEC.pdf .
974. Section 340B(a)(4) of the Public Health Service Act considers the following types of hospitals as covered entities that can participate in the 340B Drug Pricing Program: children's hospitals, critical access hospitals, disproportionate, share hospitals, free standing cancer hospitals, rural referral centers, and sole community hospitals. For further information, refer to https://www.hrsa.gov/opa/eligibility-and-registration .
975. Rural Health Research Gateway. (2018). Rural Communities: Age, Income, and Health Status. https://www.ruralhealthresearch.org/assets/2200-8536/rural-communities-age-income-health-status-recap.pdf .
976. Healthy People 2020 (n.d.) Access to Health Services. https://www.healthypeople.gov/2020/topics-objectives/topic/Access-to-Health-Services .
977. Adjusting Medicare payments for social risk to better support social needs. (2021). [Dataset]. In Forefront Group. https://doi.org/10.1377/forefront.20210526.933567 .
978. CMS Comprehensive Care for Joint Replacement Model: Performance Year 5 Evaluation Report. (2023). Centers for Medicare Medicaid Services. Retrieved December 1, 2023, from https://www.cms.gov/priorities/innovation/data-and-reports/2023/cjr-py5-annual-report .
979. Powers, B., Figueroa, J.F., Canterberry, M., Gondi, S., Franklin, S.M., Shrank, W.H., Maddox, K.E.J. (2023). Association between Community-Level Social Risk and spending among Medicare beneficiaries. JAMA Health Forum, 4(3), e230266. https://doi.org/10.1001/jamahealthforum.2023.0266 .
980. Irvin, J., Kondrich, A., Ko, M., Rajpurkar, P., Haghgoo, B., Landon, B.E., Phillips, R.L., Petterson, S., Ng, A.Y., Basu, S. (2020). Incorporating machine learning and social determinants of health indicators into prospective risk adjustment for health plan payments. BMC Public Health, 20(1). https://doi.org/10.1186/s12889-020-08735-0 .
981. Addressing social risk factors in Value-Based Payment: Adjusting payment not performance to optimize outcomes and fairness. (2021). [Dataset]. In Forefront Group. https://doi.org/10.1377/forefront.20210414.379479 .
982. Landscape of Area-Level Deprivation Measures and Other Approaches to Account for Social Risk and Social Determinants of Health in Health Care Payments. (2022). Office of the Assistant Secretary for Planning and Evaluation. Retrieved December 1, 2023, from https://aspe.hhs.gov/sites/default/files/documents/ce8cdc5da7d1b92314eab263a06efd03/Area-Level-SDOH-Indices-Report.pdf .
983. Petterson S., Deciphering the Neighborhood Atlas Area Deprivation Index: the consequences of not standardizing. Health Aff Sch. 2023;1(5):qxad063. Published 2023 Nov 3. doi:10.1093/haschl/qxad063.
984. https://www.atsdr.cdc.gov/placeandhealth/svi/at-a-glance_svi.html .
985. https://www.cms.gov/priorities/innovation/key-concepts/health-equity#:~:text=(Source3ACMS),underservedpopulations(AdaptedfromCDC) .
986. https://www.federalregister.gov/d/2021-01753/p-6 .
987. IOM (Institute of Medicine). 2009. Race, Ethnicity, and Language Data: Standardization for Health Care Quality Improvement (p.287). The National Academies Press https://www.ahrq.gov/sites/default/files/publications/files/iomracereport.pdf , https://www.ahrq.gov/sites/default/files/publications/files/iomracereport.pdf .
988. Sivashanker, K., Gandhi, T.K., (2020). Advancing Safety and Equity Together. New England Journal of Medicine, 382 (4), 301-303. https://doi.org/10.1056/nejmp1911700 , https://doi.org/10.1056/nejmp1911700 .
989. Weinick, R.M., Hasnain-Wynia, R. (2011). Quality Improvement Efforts Under Health Reform: How To Ensure That They Help Reduce Disparities—Not Increase Them. Health Affairs, 30 (10), 1837-1843. https://doi.org/10.1377/hlthaff.2011.0617 , https://doi.org/10.1377/hlthaff.2011.0617 .
990. American Society for Quality. (2019). What is root cause analysis (RCA)? Asq.org. https://asq.org/quality-resources/root-cause-analysis .
991. Agency for Healthcare Research and Quality. (2020). Plan-Do-Study-Act (PDSA) directions and examples. www.ahrq.gov . https://www.ahrq.gov/health-literacy/improve/precautions/tool2b.html .
992. Failure Modes and Effects Analysis (FMEA) Tool | IHI—Institute for Healthcare Improvement. (2017). www.ihi.org . https://www.ihi.org/resources/Pages/Tools/FailureModesandEffectsAnalysisTool.aspx .
993. Kane, R. (2014). How to Use the Fishbone Tool for Root Cause Analysis. https://www.cms.gov/medicare/provider-enrollment-and-certification/qapi/downloads/fishbonerevised.pdf .
994. Sivashanker, K., Gandhi, T.K. (2020). Advancing Safety and Equity Together. New England Journal of Medicine, 382 (4), 301-303. https://doi.org/10.1056/nejmp1911700 .
995. https://www.cms.gov/priorities/innovation/innovation-models/enhancing-oncology-model#part .
996. https://www.healthit.gov/isp/taxonomy/term/731/uscdi-v3 .
997. Office of the Assistant Secretary for Planning and Evaluation (ASPE). (2011). HHS Implementation Guidance on Data Collection Standards for Race, Ethnicity, Sex, Primary Language, and Disability Status. Retrieved from: https://aspe.hhs.gov/reports/hhs-implementation-guidance-data-collection-standards-race-ethnicity-sexprimary-language-disability-0 .
998. https://www.healthit.gov/isp/taxonomy/term/3276/uscdi-v3 .
999. Booske, B.C., Athens, J.K., Kindig, D.A., Park, H., Remington, P.L. (2010). County Health Rankings (Working Paper). https://www.countyhealthrankings.org/sites/default/files/differentPerspectivesForAssigningWeightsToDeterminantsOfHealth.pdf .
1000. ROI Calculator for Partnerships to Address the Social Determinants of Health Review of Evidence for Health-Related Social Needs Interventions. (2019). https://www.commonwealthfund.org/sites/default/files/2019-07/COMBINED-ROI-EVIDENCE-REVIEW-7-1-19.pdf .
1001. De Marchis EH, Brown E, Aceves B, et al. State of the Science of Screening in Healthcare Settings. Social Interventions Research Evaluation Network, 2022. https://sirenetwork.ucsf.edu/tools-resources/resources/state-science-social-screening-healthcare-settings . https://sirenetwork.ucsf.edu/tools-resources/resources/state-science-social-screening-healthcare-settings .
1002. De Marchis EH, Brown E, Aceves B, et al. State of the Science of Screening in Healthcare Settings. Social Interventions Research Evaluation Network, 2022. https://sirenetwork.ucsf.edu/tools-resources/resources/state-science-social-screening-healthcare-settings . https://sirenetwork.ucsf.edu/tools-resources/resources/state-science-social-screening-healthcare-settings .
1003. For more information ONC Health IT Certification Criteria, see https://www.healthit.gov/topic/certification-ehrs/certification-criteria.https://www.healthit.gov/topic/certification-ehrs/certification-criteria . For standards and implementation specifications adopted under PHSA section 3004, see 45 CFR part 170, subpart B .
1004. Nabagiez, J.P., Shariff, M.A., Khan, M.A., Molloy, W.J., McGinn, J.T. (2013). Physician assistant home visit program to reduce hospital readmissions. The Journal of Thoracic and Cardiovascular Surgery, 145(1), 225-233. https://doi.org/10.1016/j.jtcvs.2012.09.047 .
1005. Hall, M.L., Esposito, G., Pekmezaris, R., Lesser, M., Moravick, D., Jahn, L., Blenderman, R., Akerman, M., Nouryan, C., Hartman, A.R. (2014). Cardiac surgery nurse practitioner home visits prevent coronary artery bypass graft readmissions. The Annals of Thoracic Surgery, 97(5), 1488-1495. https://doi.org/10.1016/j.athoracsur.2013.12.049 .
1006. Harkey, K., Kaiser, N., Zhao, J., Gutnik, B., Kelz, R.R., Matthews, B.D., Reinke, C.E. (2023). Utilization of telemedicine to provide post-discharge care: A comparison of pre-pandemic vs. pandemic care. The American Journal of Surgery, 226(2), 163-169. https://doi.org/10.1016/j.amjsurg.2023.03.007 .
1007. Grauer, A., Cornelius, T., Abdalla, M., Moise, N., Kronish, I.M., Ye, S. (2023). Impact of early telemedicine follow-up on 30-Day hospital readmissions. PLOS ONE, 18(5), e0282081. https://doi.org/10.1371/journal.pone.0282081 .
1008. Gajarawala, S.N., Pelkowski, J.N. (2021). Telehealth benefits and barriers. The Journal for Nurse Practitioners, 17(2), 218-221. https://doi.org/10.1016/j.nurpra.2020.09.013 .
1009. https://www.medicare.gov/care-compare/?redirect=trueproviderType=NursingHome .
1010. Medicare Claims Maturity: CCW White Paper accessed at https://www2.ccwdata.org/web/guest/white-papers?p_l_back_url=%2Fweb%2Fguest%2Fsearch%3Fq%3Dmedicare%2Bclaims%2Bmaturity on Jan, 26, 2024.
1011. https://www.healthit.gov/topic/health-it-and-health-information-exchange-basics/health-information-exchange .
1012. Chen, M., Guo, S., Tan, X. (2019). Does health information exchange improve patient outcomes? Empirical evidence from Florida hospitals. Health Affairs, 38(2), 197-204. https://doi.org/10.1377/hlthaff.2018.05447 .
1013. Menachemi, N., Rahurkar, S., Harle, C.A., Vest, J.R. (2018). The benefits of health information exchange: an updated systematic review. Journal of the American Medical Informatics Association, 25(9), 1259-1265. https://doi.org/10.1093/jamia/ocy035 .
1014. https://www.healthit.gov/topic/interoperability/policy/trusted-exchange-framework-and-common-agreement-tefca .
1015. Janet Ranganathan, Laurent Corbier, Pankaj Bhatia, Simon Schultz, Peter Gage, Kjeli Oren. The Greenhouse Gas Protocol: A Corporate Accounting and Reporting Standard (Revised Edition). World Business Council for Sustainable Development and World Resources Institute. 2004. https://ghgprotocol.org/sites/default/files/standards/ghg-protocol-revised.pdf https://ghgprotocol.org/sites/default/files/standards/ghg-protocol-revised.pdf .
1016. Allison R. Crimmins Alexa K. Jay (eds.). U.S. Global Change Research Program. Fifth National Climate Assessment. U.S. Global Change Research Program. 2023. https://nca2023.globalchange.gov/https://nca2023.globalchange.gov/ .
1017. EPA's Office of Atmospheric Programs. Climate Change and Social Vulnerability in the United States: A Focus on Six Impacts. U.S. Environmental Protection Agency. U.S. Environmental Protection Agency. EPA 430-R-21-003. September 2021. https://www.epa.gov/system/files/documents/2021-09/climate-vulnerability_september-2021_508.pdf https://www.epa.gov/system/files/documents/2021-09/climate-vulnerability_september-2021_508.pdf .
1018. Andrea J. MacNeill, Robert Lillywhite, Carl J. Brown. The Impact of Surgery on Global Climate: A Carbon Footprinting Study Of Operating Theatres in Three Health Systems. Lancet Planetary Health, vol. 1, no. 9, pp. E381-E388. December 2017. https://www.thelancet.com/journals/lanplh/article/PIIS2542-5196(17)30162-6/fulltexthttps://www.thelancet.com/journals/lanplh/article/PIIS2542-5196(17)30162-6/fulltext .
1019. Maya A. Babu, Angela K. Dalenberg, Glen Goodsell, Amanda B. Holloway, Marcia M. Belau, Michael J. Link. Greening the Operating Room: Results of a Scalable Initiative to Reduce Waste and Recover Supply Costs. Neurosurgery, vol. 85, no. 3, pp. 432-437. September 1, 2019. https://pubmed.ncbi.nlm.nih.gov/30060055/ . https://pubmed.ncbi.nlm.nih.gov/30060055/ .
1020. Matthew J. Eckelman, Kaixin Huang, Robert Lagasse, Emily Senay, Robert Dubrow, Jodi D. Sherman. Health Care Pollution and Public Health Damage in The United States: An Update. Health Affairs, vol. 39, no. 12, pp. 2071-2079. December 2020. https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01247https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01247 .
1021. Matthew J. Eckelman, Kaixin Huang, Robert Lagasse, Emily Senay, Robert Dubrow, Jodi D. Sherman. Health Care Pollution and Public Health Damage in The United States: An Update. Health Affairs, vol. 39, no. 12, pp. 2071-2079. December 2020. https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01247https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01247 .
1022. USGCRP, 2018: Impacts, Risks, and Adaptation in the United States: Fourth National Climate Assessment, Volume II [Reidmiller, D.R., C.W. Avery, D.R. Easterling, K.E. Kunkel, K.L.M. Lewis, T.K. Maycock, and B.C. Stewart (eds.)]. U.S. Global Change Research Program, Washington, DC, USA, 1515 pp. doi: 10.7930/NCA4.2018.
1023. Joel D. Kaufman, Sara D. Adar, R. Graham Barr, et al. Association Between Air Pollution and Coronary Artery Calcification Within Six Metropolitan Areas in the USA (The Multi-Ethnic Study of Atherosclerosis and Air Pollution): A Longitudinal Cohort Study. Lancet, vol. 388, no. 10045, pp. 696-704. August 13, 2017. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5019949/https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5019949/ .
1024. Joel D. Kaufman, Sara D. Adar, R. Graham Barr, et al. Association Between Air Pollution and Coronary Artery Calcification Within Six Metropolitan Areas in the USA (The Multi-Ethnic Study of Atherosclerosis and Air Pollution): A Longitudinal Cohort Study. Lancet, vol. 388, no. 10045, pp. 696-704. August 13, 2017. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5019949/https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5019949/ .
1025. HHS Office of Climate Change Health Equity. Health Sector Commitments to Emissions Reduction and Resilience. HHS Office of the Assistant Secretary for Health—Health Sector Pledge. January 3, 2024. https://www.hhs.gov/climate-change-health-equity-environmental-justice/climate-change-health-equity/actions/health-sector-pledge/index.htmlhttps://www.hhs.gov/climate-change-health-equity-environmental-justice/climate-change-health-equity/actions/health-sector-pledge/index.html .
1026. HHS Office of Climate Change Health Equity. Compendium of Federal Resources for Health Sector Emissions Reduction and Resilience. HHS Office of the Assistant Secretary for Health—Health Sector Pledge. December 7, 2023. https://www.hhs.gov/climate-change-health-equity-environmental-justice/climate-change-health-equity/actions/health-care-sector-pledge/federal-resources/index.htmlhttps://www.hhs.gov/climate-change-health-equity-environmental-justice/climate-change-health-equity/actions/health-care-sector-pledge/federal-resources/index.html .
1027. Bhargavi Sampath, Matthew Jensen, Jennifer Lenoci-Edwards, Kevin Little, Hardeep Singh, Jodi D. Sherman. Reducing Health care Carbon Emissions: A Primer on Measures and Actions for Health Care Organizations to Mitigate Climate Change. U.S. Agency for Healthcare Research Quality. AHRQ pub. No. 22-M011. September 2023. Reducing Healthcare Carbon Emissions: A Primer on Measures and Actions to Mitigate Climate Change ( ahrq.gov )Reducing Healthcare Carbon Emissions: A Primer on Measures and Actions to Mitigate Climate Change ( ahrq.gov ).
1028. CMS Quality, Safety, Oversight Group (QSOG) Director and CMS Survey Operations Group (SOG) Director. Categorical Waiver—Health Care Microgrid Systems (HCMSs). CMS Center for Clinical Standards and Quality reference no. QSO-23-11-LSC. March 31, 2023. https://www.cms.gov/medicare/provider-enrollment-and-certification/surveycertificationgeninfo/policy-and-memos-states/categorical-waiver-health-care-microgrid-systems-hcmsshttps://www.cms.gov/medicare/provider-enrollment-and-certification/surveycertificationgeninfo/policy-and-memos-states/categorical-waiver-health-care-microgrid-systems-hcmss .
1029. Janet Ranganathan, Laurent Corbier, Pankaj Bhatia, Simon Schultz, Peter Gage, Kjeli Oren. The Greenhouse Gas Protocol: A Corporate Accounting and Reporting Standard (Revised Edition). World Business Council for Sustainable Development and World Resources Institute. 2004. https://ghgprotocol.org/sites/default/files/standards/ghg-protocol-revised.pdfhttps://ghgprotocol.org/sites/default/files/standards/ghg-protocol-revised.pdf .
1030. Nick Watts (ed.). Delivering a `Net Zero' National Health Service. NHS England NHS Improvement publication no. PAR133. July 2022. B1728-delivering-a-net-zero-nhs-july-2022.pdf ( england.nhs.uk )B1728-delivering-a-net-zero-nhs-july-2022.pdf ( england.nhs.uk ).
1031. Matthew J. Eckelman, Kaixin Huang, Robert Lagasse, Emily Senay, Robert Dubrow, Jodi D. Sherman. Health Care Pollution and Public Health Damage in The United States: An Update. Health Affairs, vol. 39, no. 12, pp. 2071-2079. December 2020. https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01247https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01247 .
1032. Matthew J. Eckelman, Kaixin Huang, Robert Lagasse, Emily Senay, Robert Dubrow, Jodi D. Sherman. Health Care Pollution and Public Health Damage in The United States: An Update. Health Affairs, vol. 39, no. 12, pp. 2071-2079. December 2020. https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01247https://www.healthaffairs.org/doi/10.1377/hlthaff.2020.01247 .
1033. EPA's Office of Atmospheric Programs. Climate Change and Social Vulnerability in the United States: A Focus on Six Impacts. U.S. Environmental Protection Agency. U.S. Environmental Protection Agency. EPA 430-R-21-003. September 2021. https://www.epa.gov/system/files/documents/2021-09/climate-vulnerability_september-2021_508.pdfhttps://www.epa.gov/system/files/documents/2021-09/climate-vulnerability_september-2021_508.pdf .
1034. Sharon E. Mace Aishwarya Sharma. Hospital Evacuations Due to Disasters in the United States in the Twenty-First Century. American Journal of Disaster Medicine, vol. 15, no. 1, pp. 7-22. January 2020, Hospital evacuations due to disasters in the United States in the twenty-first century—PubMed ( nih.gov ).
1035. NOAA Climate Program Office. Hospital Plans Ahead for Power, Serves the Community Through Hurricane Sandy. U.S. National Oceanic Atmospheric Administration Climate Resilience Toolkit. February 15, 2018. https://toolkit.climate.gov/case-studies/hospital-plans-ahead-power-serves-community-through-hurricane-sandyhttps://toolkit.climate.gov/case-studies/hospital-plans-ahead-power-serves-community-through-hurricane-sandy .
1036. Alicia Edmonds, Hilary Stambaugh, Scot Pettey, Kenn B. Daratha. Evidence-Based Project: Cost Savings and Reduction in Environmental Release With Low-Flow Anesthesia. AANA J, vol. 89, no. 1, pp. 27-33. February 2021. Evidence-Based Project: Cost Savings and Reduction in Environmental Release With Low-Flow Anesthesia—PubMed ( nih.gov )Evidence-Based Project: Cost Savings and Reduction in Environmental Release With Low-Flow Anesthesia—PubMed ( nih.gov ).
1037. Lulin Wang, Junqing Xie, Yonghua Hu, Yaohua Tian. Air Pollution and Risk of Chronic Obstructed Pulmonary Disease: The Modifying Effect of Genetic Susceptibility and Lifestyle. Lancet eBioMedicine, vol. 79, pp. 103994. May 2022. Air pollution and risk of chronic obstructed pulmonary disease: The modifying effect of genetic susceptibility and lifestyle—PMC ( nih.gov )Air pollution and risk of chronic obstructed pulmonary disease: The modifying effect of genetic susceptibility and lifestyle—PMC ( nih.gov ).
1038. Marina Romanello, Alice McGushin, Claudia Di Napoli, et al. The 2021 Report of the Lancet Countdown on Health and Climate Change: Code Red for a Healthy Future. Lancet, vol. 398, no. 10311, pp. 1619-1662. October 20, 2021. The 2021 report of the Lancet Countdown on health and climate change: code red for a healthy future—The LancetThe 2021 report of the Lancet Countdown on health and climate change: code red for a healthy future—The Lancet.
1039. Janet L. Gamble John Balbus. Chapter. 9: Populations of Concern. In: U.S. Global Change Research Program. The Impacts of Climate Change on Human Health in the United States: A Scientific Assessment. 2016. The Impacts of Climate Change on Human Health in the United States: A Scientific Assessment ( globalchange.gov )The Impacts of Climate Change on Human Health in the United States: A Scientific Assessment ( globalchange.gov ).
1040. Diarmid Campbell-Lendrum Nicola Wheeler. COP24 Special Report: Health Climate Change. World Health Organization Special Report. 2018. 9789241514972-eng.pdf ( who.int )9789241514972-eng.pdf ( who.int ).
1041. Laura P. Sands, Quyen Do, Pang Du, Yunnan Xu, Rachel Pruchno. Long Term Impact of Hurricane Sandy on Hospital Admissions of Older Adults. Social Science Medicine, vol. 293, pp. 114659. January 1, 2023. Long term impact of Hurricane Sandy on hospital admissions of older adults—PMC ( nih.gov )Long term impact of Hurricane Sandy on hospital admissions of older adults—PMC ( nih.gov ).
1042. Wim Thiery, Stefan Lange, Joeri Rogel, et al. Intergenerational Inequities in Exposure to Climate Extremes. Science, vol. 374, no. 6564, pp. 158-160. September 26, 2021. Intergenerational inequities in exposure to climate extremes—PubMed ( nih.gov ).
1043. Vijay S. Limaye, Wendy Max, Juanita Constible, Kim Knowlton. Estimating the Health-Related Costs of 10 Climate-Sensitive U.S. Events During 2012. GeoHealth, vol. 3, no. 9, pp. 245-265. September 17, 2019. Estimating the Health‐Related Costs of 10 Climate-Sensitive U.S. Events During 2012—PMC ( nih.gov )Estimating the Health-Related Costs of 10 Climate-Sensitive U.S. Events During 2012—PMC ( nih.gov ).
1044. Ibid.
1045. U.S. Office of Management Budget. Climate Risk Exposure: An Assessment of the Federal Government's Financial Risks to Climate Change. OMB White Paper. April 2022. https://www.whitehouse.gov/wp-content/uploads/2022/04/OMB_Climate_Risk_Exposure_2022.pdfhttps://www.whitehouse.gov/wp-content/uploads/2022/04/OMB_Climate_Risk_Exposure_2022.pdf .
1046. HHS Office of Climate Change Health Equity. (OCCHE) Quickfinder for Leveraging the Inflation Reduction Act for the Health Sector. HHS Office of the Assistant Secretary for Health. February 27, 2024. The Office of Climate Change and Health Equity (OCCHE) Quickfinder for Leveraging the Inflation Reduction Act for the Health Sector | HHS.gov The Office of Climate Change and Health Equity (OCCHE) Quickfinder for Leveraging the Inflation Reduction Act for the Health Sector | HHS.gov.
1047. HHS Office of Climate Change Health Equity. Compendium of Federal Resources for Health Sector Emissions Reduction and Resilience. HHS Office of the Assistant Secretary for Health. December 7, 2023. Compendium of Federal Resources for Health Sector Emissions Climate Change Technical Assistance for Territories Reduction and Resilience | HHS.govCompendium of Federal Resources for Health Sector Emissions Climate Change Technical Assistance for Territories Reduction and Resilience | HHS.gov.
1048. HHS Office of Climate Change Health Equity. Catalytic Program on Utilizing the IRA. HHS Office of the Assistant Secretary for Health Resource Hub. March 1, 2024. https://www.hhs.gov/climate-change-health-equity-environmental-justice/climate-change-health-equity/health-sector-resource-hub/new-catalytic-program-utilizing-ira/index.html . https://www.hhs.gov/climate-change-health-equity-environmental-justice/climate-change-health-equity/health-sector-resource-hub/new-catalytic-program-utilizing-ira/index.html
1049. Energy Star Treasure Hunts, https://www.energystar.gov/industrial_plants/treasure_hunt . https://www.energystar.gov/industrial_plants/treasure_hunt .
1050. Kathy Gerwig, Hardeep Singh, Jodi Sherman, Walt Vernon, Beth Schenk. Action Collaborative on Decarbonizing the Health Sector. Key Actions to Reduce Greenhouse Gas Emissions by U.S. Hospitals and Health Systems. National Academy of Medicine Climate Collaborative. 2022. https://nam.edu/programs/climate-change-and-human-health/action-collaborative-on-decarbonizing-the-u-s-health-sector/key-actions-to-reduce-greenhouse-gas-emissions-by-u-s-hospitals-and-health-systems/https://nam.edu/programs/climate-change-and-human-health/action-collaborative-on-decarbonizing-the-u-s-health-sector/key-actions-to-reduce-greenhouse-gas-emissions-by-u-s-hospitals-and-health-systems/ .
1051. https://ghgprotocol.org/corporate-standard .
1052. EPA Office of Air Programs. ENERGY STAR Portfolio Manager Glossary. U.S. Environmental Protection Agency U.S. Department of Energy. Undated. https://portfoliomanager.energystar.gov/pm/glossaryhttps://portfoliomanager.energystar.gov/pm/glossary .
1053. EPA Office of Air Programs. ENERGY STAR Score for Hospitals (General Medical and Surgical). U.S. Environmental Protection Agency U.S. Department of Energy. February 19, 2021. https://www.energystar.gov/buildings/tools-and-resources/energy-star-score-hospitals-general-medical-and-surgicalhttps://www.energystar.gov/buildings/tools-and-resources/energy-star-score-hospitals-general-medical-and-surgical .
1054. EPA Office of Air Programs. Technical Reference: ENERGY STAR Score for Hospitals in the United States. U.S. Environmental Protection Agency U.S. Department of Energy. February 2021. https://www.energystar.gov/sites/default/files/tools/Hospital_TechnicalReference_Feb2021_508.pdfhttps://www.energystar.gov/sites/default/files/tools/Hospital_TechnicalReference_Feb2021_508.pdf .
1055. AHA Health Forum. Fast Facts on Hospitals. American Hospital Association. 2024. https://www.aha.org/statistics/fast-facts-us-hospitalshttps://www.aha.org/statistics/fast-facts-us-hospitals .
1056. EPA Office of Air Programs. State/Local Compliance Ordinances. U.S. Environmental Protection Agency U.S. Department of Energy. February 20, 2024. State/local compliance ordinances ( site.com )State/local compliance ordinances ( site.com ).
1057. Practice Greenhealth. Health Care Emissions Impact Calculator. 2023. https://practicegreenhealth.org/tools-and-resources/health-care-emissions-impact-calculator . https://practicegreenhealth.org/tools-and-resources/health-care-emissions-impact-calculator .
1058. We recognize that certain gases and compounds are most easily measured by volume and others in weight as they are not purchased by bottle (for example, Nitrous Oxide).
1059. Bhargavi Sampath, Matthew Jensen, Jennifer Lenoci-Edwards, Kevin Little, Hardeep Singh, Jodi D. Sherman. Reducing Health care Carbon Emissions: A Primer on Measures and Actions for Health Care Organizations to Mitigate Climate Change. U.S. Agency for Healthcare Research Quality. AHRQ pub. No. 22-M011. September 2023. Available at: https://www.ahrq.gov/sites/default/files/wysiwyg/healthsystemsresearch/decarbonization/decarbonization.pdf
1060. Tennison I. Roschnik S. Ashby B et al. (2021). Health care's response to climate change: a carbon footprint assessment of the NHS in England. Lancet Planet Health. 5(2):e84-e92. www.doi.org/10.1016/S2542-5196(20)30271-0 .
1061. Singh H. Eckelman M. Berwick DM Sherman JD. (2022). Mandatory Reporting of Emissions to Achieve Net-Zero Health Care. N Engl J Med 387(26): 2469-27476. www.doi.org/10.1056/NEJMsb2210022 .
1062. Bhargavi Sampath, Matthew Jensen, Jennifer Lenoci-Edwards, Kevin Little, Hardeep Singh, Jodi D. Sherman. Reducing Health care Carbon Emissions: A Primer on Measures and Actions for Health Care Organizations to Mitigate Climate Change. U.S. Agency for Healthcare Research Quality. AHRQ pub. No. 22-M011. September 2023. Reducing Healthcare Carbon Emissions: A Primer on Measures and Actions to Mitigate Climate Change ( ahrq.gov )Reducing Healthcare Carbon Emissions: A Primer on Measures and Actions to Mitigate Climate Change ( ahrq.gov ).
1063. Bhargavi Sampath, Matthew Jensen, Jennifer Lenoci-Edwards, Kevin Little, Hardeep Singh, Jodi D. Sherman. Reducing Health care Carbon Emissions: A Primer on Measures and Actions for Health Care Organizations to Mitigate Climate Change. U.S. Agency for Healthcare Research Quality. AHRQ pub. No. 22-M011. September 2023. Reducing Healthcare Carbon Emissions: A Primer on Measures and Actions to Mitigate Climate Change ( ahrq.gov ). Reducing Healthcare Carbon Emissions: A Primer on Measures and Actions to Mitigate Climate Change ( ahrq.gov ).
1064. Kimberly Wintemute Fiona Miller. Dry Powder Inhalers Are Environmentally Preferable to Metered-Dose Inhalers. CMAJ, vol. 192, no. 29, pp. E846. July 20, 2020. https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7828988/https://www.ncbi.nlm.nih.gov/pmc/articles/PMC7828988/ .
1065. EPA Office of Air Programs. State/Local Compliance Ordinances. U.S. Environmental Protection Agency U.S. Department of Energy. February 20, 2024. State/local compliance ordinances ( site.com ).
1066. Kent Peterson, Paul Torcellini, Roger Grant. A Common Definition for Zero Energy Buildings. National Institute of Building Sciences. September 2015. DOE/EE-1247. https://www.energy.gov/sites/default/files/2015/09/f26/bto_common_definition_zero_energy_buildings_093015.pdf https://www.energy.gov/sites/default/files/2015/09/f26/bto_common_definition_zero_energy_buildings_093015.pdf .
1067. Hardeep Singh, Walt Vernon, Terri Scannell, Kathy Gerwig. (2023). Crossing the Decarbonization Chasm: A Call to Action for Hospital and Health System Leaders to Reduce Their Greenhouse Gas Emissions. National Academy of Medicine Discussion Paper. November 29, 2023. https://nam.edu/crossing-the-decarbonization-chasm-a-call-to-action-for-hospital-and-health-system-leaders-to-reduce-their-greenhouse-gas-emissions/https://nam.edu/crossing-the-decarbonization-chasm-a-call-to-action-for-hospital-and-health-system-leaders-to-reduce-their-greenhouse-gas-emissions/ .
1068. Vijay S. Limaye, Wendy Max, Juanita Constible, Knowlton. Estimating the Health‐Related Costs of 10 Climate‐Sensitive U.S. Events During 2012. GeoHealth, vol. 3, no. 9, pp. 245-265. September 17, 2019. Estimating the Health‐Related Costs of 10 Climate‐Sensitive U.S. Events During 2012—PMC ( nih.gov ). Estimating the Health‐Related Costs of 10 Climate‐Sensitive U.S. Events During 2012—PMC ( nih.gov ).
1069. The Joint Commission. Sustainable Healthcare Certification. 2024. Sustainable Healthcare Certification | The Joint Commission. Sustainable Healthcare Certification | The Joint Commission.
1070. Sivak M and Schoettle B. (2018) Relative Costs of Driving Electric and Gasoline Vehicles in the Individual U.S. States. Transportation Research Board. Available at: https://trid.trb.org/view/1508116 .
1071. Baldwin R, Richie S, Vanderwerp D. (2022). EV vs. Gas: Which Cars Are Cheaper to Own? Car and Driver. Available at: https://www.caranddriver.com/shopping-advice/a32494027/ev-vs-gas-cheaper-to-own/ .
1072. Dzau VJ, Levine R, Barrett G, Witty A. (2021). Decarbonizing the U.S. Health Sector—A Call to Action. N Engl J Med 385;23, 2117-2119. www.doi.org/10.1056/NEJMp2115675 www.doi.org/10.1056/NEJMp2115675 .
1073. Singh H, Vernon W, Scannell T, Gerwig K. (2023). Crossing the Decarbonization Chasm: A Call to Action for Hospital and Health System Leaders to Reduce Their Greenhouse Gas Emissions. NAM Perspectives. Discussion Paper, National Academy of Medicine, Washington, DC. https://doi.org/10.31478/202311g .
1074. Ibid.
1075. See e.g.,: Becerra v. Empire Health Found. , for Valley Hosp. Med. Ctr., 142 S. Ct. 2354 (2022); Sebelius v. Auburn Reg'l Med. Ctr., 568 U.S. 145 (2013); Your Home Visiting Nurse Servs., Inc. v. Shalala , 525 U.S. 449 (1999); and Bethesda Hosp. Ass'n v. Bowen , 485 U.S. 399 (1988).
1076. White House. White House Blueprint for Addressing the Maternal Health Crisis. 2022. Accessed January 2, 2024. https://www.whitehouse.gov/wp-content/uploads/2022/06/Maternal-Health-Blueprint.pdf .
1077. CMS. CMS Cross Cutting Initiative: Maternity Care Action Plan. 2022. Accessed January 2, 2023. https://www.cms.gov/files/document/cms-maternity-care-action-plan.pdf .
1078. Who's eligible for Medicare? U.S. Department of Health and Human Services. Accessed January 2, 2024. https://www.hhs.gov/answers/medicare-and-medicaid/who-is-eligible-for-medicare/index.html .
1079. Medicare Beneficiaries at a Glance 2023 Edition. Centers for Medicare and Medicaid Services. https://data.cms.gov/infographic/medicare-beneficiaries-at-a-glance .
1080. Gleason JL, Grewal J, Chen Z, Cernich AN, Grantz KL. Risk of Adverse Maternal Outcomes in Pregnant Women With Disabilities. JAMA Netw Open. 2021;4(12):e2138414. Published 2021 Dec 1. doi:10.1001/jamanetworkopen.2021.38414.
1081. https://www.medicaid.pr.gov/pdf/Congress/PRDOH_Congressional%20Report%202%20PERM%20Compliance%20Plan_FINAL[2][1].pdf .
1082. https://www.cdc.gov/infectioncontrol/guidelines/core-practices/index.html?CDC_AA_refVal=https%3A%2F%2Fwww.cdc.govhicpacrecommendationscore-practices.html .
1083. Infection Control: Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) | CDC; 2023.12.14—IDPH Recommends Healthcare Facilities Adopt Mitigation Measures as Respiratory Viruses Increase ( illinois.gov ) 2024-doh-masking-advisory.pdf ( ny.gov ); Health Alert Network (HAN)—00503 | Urgent Need to Increase Immunization Coverage for Influenza, COVID-19, and RSV and Use of Authorized/Approved Therapeutics in the Setting of Increased Respiratory Disease Activity During the 2023-2024 Winter Season ( cdc.gov ).
1084. FY2025 IPPS Proposed Rule. https://www.govinfo.gov/content/pkg/FR-2024-05-02/pdf/2024-07567.pdf .
1085. https://oig.hhs.gov/oei/reports/OEI-05-20-00540.asp ; https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9533809/#:~:text=In%20this%20study%20cohort%2C%2062,%2C%20and%205%25%20were%20Hispanic .
1086. https://www.cdc.gov/mmwr/volumes/72/wr/mm7219e2.htm
1087. https://www.cdc.gov/nhsn/pdfs/training/D1_Connectivity-Initiative_-Hospital-Bed-Capacity-Project_508c.pdf
1088. The HTI-2 proposed rule is available on ONC's website at https://www.healthit.gov/sites/default/files/page/2024-07/ONC_HTI-2_Proposed_Rule.pdf .
1089. https://www.cdc.gov/nhsn/pdfs/training/D1_Introducing-NHSNs-New-Digital-Quality-Measures_508c.pdf .
1090. https://www.cdc.gov/nhsn/nhsncolab/index.html .
1091. https://www.hhs.gov/sites/default/files/covid-19-faqs-hospitals-hospital-laboratory-acute-care-facility-data-reporting.pdf .
1092. https://www.cdc.gov/nssp/technical-pubs-and-standards.html#Dictionaries .
1093. U.S. Bureau of Labor Statistics. Occupational Outlook Handbook, Legal Secretaries and Administrative Assistants. Accessed on February 6, 2024. Available at: https://www.bls.gov/oes/current/oes436012.htm .
1094. U.S. Bureau of Labor Statistics. Occupational Outlook Handbook, Medical Records Specialists. Accessed January 3, 2024. Available at: https://www.bls.gov/oes/current/oes292072.htm .
1095. CDC. (2023). FAQs About NHSN Agreement to Participate and Consent. Available at: https://www.cdc.gov/nhsn/about-nhsn/faq-agreement-to-participate.html .
1096. https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework .
1097. https://www.bls.gov/news.release/pdf/wkyeng.pdf . Accessed January 1, 2024.
1098. https://www.census.gov/library/stories/2023/09/median-household-income.html . Accessed January 2, 2024.
1099. U.S. Bureau of Labor Statistics. Occupational Outlook Handbook, Medical Records Specialists. Accessed on January 2, 2024. Available at: https://www.bls.gov/oes/current/oes292072.htm .
1100. https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework .
1101. https://www.bls.gov/news.release/pdf/wkyeng.pdf . Accessed January 1, 2024.
1102. https://www.census.gov/library/stories/2023/09/median-household-income.html . Accessed January 2, 2024.
1103. U.S. Bureau of Labor Statistics. Occupational Outlook Handbook, Medical Records Specialists. Accessed on January 3, 2024. Available at: https://www.bls.gov/oes/current/oes292072.htm .
1104. https://www.bls.gov/oes/2022/may/oes291141.htm .
1105. https://www.bls.gov/oes/2022/may/oes291141.htm
1106. Change Request 2785 (Transmittal A-03-058; July 3, 2003) found at https://www.cms.gov/regulations-and-guidance/guidance/transmittals/downloads/a03058.pdf .
1107. This report is available on the OIG website at: https://oig.hhs.gov/oas/reports/region5/51600060.pdf .
1108. For the BPCI Advanced model, the last day of the last performance period is December 31, 2025. For the CJR model, the last day of the last performance year is December 31, 2024.
1109. Available at: https://www.cms.gov/files/document/2022-cms-strategic-framework.pdf .
1110. https://aspe.hhs.gov/sites/default/files/migrated_legacy_files//195046/Social-Risk-in-Medicare%E2%80%99s-VBP-2nd-Report-Executive-Summary.pdf .
1111. Available at: https://health.gov/healthypeople/priority-areas/social-determinants-health .
1112. See “Utilization of Z Codes for Social Determinants of Health among Medicare Fee-for-Service Beneficiaries, 2019,” available at https://www.cms.gov/files/document/z-codes-data-highlight.pdf .
1113. Viron M, Zioto K, Schweitzer J, Levine G. Behavioral Health Homes: an opportunity to address healthcare inequities in people with serious mental illness. Asian J Psychiatr. 2014 Aug; 10:10-6. doi: 10.1016/j.ajp.2014.03.009.
1114. Cully, J.A., Breland, J.Y., Robertson, S. et al. Behavioral health coaching for rural veterans with diabetes and depression: a patient randomized effectiveness implementation trial. BMC Health Serv Res 14, 191 (2014). https://doi.org/10.1186/1472-6963-14-191 .
1115. https://www.cms.gov/cms-behavioral-health-strategy .
1116. https://www.ssa.gov/disability/professionals/bluebook/general-info.htm .
1117. Smart NA, Titus TT. Outcomes of early versus late nephrology referral in chronic kidney disease: a systematic review. Am J Med. 2011 Nov;124(11):1073-80.e2. doi: 10.1016/j.amjmed.2011.04.026. PMID: 22017785.
1118. National Healthcare Quality and Disparities Report chartbook on rural health care. Rockville, MD: Agency for Healthcare Research and Quality; October 2017. AHRQ Pub. No. 17(18)-0001-2-EF available at https://www.ahrq.gov/sites/default/files/wysiwyg/research/findings/nhqrdr/chartbooks/qdr-ruralhealthchartbook-update.pdf .
1119. Muluk, S, Sabik, L, Chen, Q, Jacobs, B, Sun, Z, Drake, C. Disparities in geographic access to medical oncologists. Health Serv Res. 2022; 57(5): 1035-1044. doi:10.1111/1475-6773.13991.
1120. https://www.neighborhoodatlas.medicine.wisc.edu/ .
1121. 7 U.S. Department of Health Human Services, “Executive Summary: Report to Congress: Social Risk Factors and Performance in Medicare's Value-Based Purchasing Program,” Office of the Assistant Secretary for Planning and Evaluation, March 2020. Available at https://aspe.hhs.gov/sites/default/files/migrated_legacy_files//195046/Social-Risk-inMedicare%E2%80%99s-VBP-2nd-Report-Executive-Summary.pdf .
1122. Kind AJ, et al., “Neighborhood socioeconomic disadvantage and 30-day rehospitalization: a retrospective cohort study.” Annals of Internal Medicine. No. 161(11), pp 765-74, doi: 10.7326/M13-2946 (December 2, 2014), available at https://www.acpjournals.org/doi/epdf/10.7326/M13-2946 .
1123. Jencks SF, et al., “Safety-Net Hospitals, Neighborhood Disadvantage, and Readmissions Under Maryland's All-Payer Program.” Annals of Internal Medicine. No. 171, pp 91-98, doi:10.7326/M16-2671 (July 16, 2019), available at https://www.acpjournals.org/doi/epdf/10.7326/M16-2671 .
1124. Cheng E, et al., “Neighborhood and Individual Socioeconomic Disadvantage and Survival Among Patients With Nonmetastatic Common Cancers.” JAMA Network Open Oncology. No. 4(12), pp 1-17, doi: 10.1001/jamanetworkopen.2021.39593 (December 17, 2021), available at https://onlinelibrary.wiley.com/doi/epdf/10.1111/jrh.12597 .
1125. Khlopas A, et al., “Neighborhood Socioeconomic Disadvantages Associated With Prolonged Lengths of Stay, Nonhome Discharges, and 90-Day Readmissions After Total Knee Arthroplasty.” The Journal of Arthroplasty. No. 37(6), pp S37-S43, doi: 10.1016/j.arth.2022.01.032 (June 2022), available at https://www.sciencedirect.com/science/article/pii/S0883540322000493 .
1126. Category One consists of hospitals that are located in a rural area (as defined in section 1886(d)(2)(D) of the Act) or have been reclassified being located in a rural area (pursuant to section 1886(d)(8)(E) of the Act). Category Two consists of hospitals in which the reference resident level of the hospital (as specified in section 1886(h)(10)(F)(iv) of the Act) is greater than the otherwise applicable resident limit. Category Three consists of hospitals located in States with new medical schools that received `Candidate School' status from the Liaison Committee on Medical Education (LCME) or that received `Pre-Accreditation' status from the American Osteopathic Association (AOA) Commission on Osteopathic College Accreditation (the COCA) on or after January 1, 2000, and that have achieved or continue to progress toward `Full Accreditation' status (as such term is defined by the LCME) or toward `Accreditation' status (as such term is defined by the COCA); or additional locations and branch campuses established on or after January 1, 2000, by medical schools with `Full Accreditation' status (as such term is defined by LCME) or `Accreditation' status (as such term is defined by the COCA). Category Four consists of hospitals that serve areas designated as HPSAs under section 332(a)(1)(A) of the Public Health Service Act (PHSA), as determined by the Secretary.
1127. https://www.regulations.gov/comment/CMS-2023-0120-3326 .
1128. For the BPCI Advanced model, the last day of the last performance period is December 31, 2025. For the CJR model, the last day of the last performance year is December 31, 2024.
1129. https://www.cms.gov/oact/tr/2023 .
1130. https://www.cms.gov/priorities/innovation/data-and-reports/2023/cjr-py5-annual-report .
1131. For PERM, a “state” represents an entity receiving Medicaid and CHIP funding that is measured for improper payments, which includes the 50 states, the District of Columbia, and now Puerto Rico.
1132. Park, C., et al. (2022). “Association Between Implementation of a Geriatric Trauma Clinical Pathway and Changes in Rates of Delirium in Older Adults With Traumatic Injury.” JAMA Surg 157(8): 676-683.
1133. https://mini-cog.com/#:~:text=The%20Mini%2DCog%C2%A9%20is,cognitive%20impairment%20in%20older%20patients .
1134. https://www.physio-pedia.com/Katz_ADL#:~:text=The%20Katz%20ADL%2C%20is%20an,to%20perform%20and%20requires%20training .
1135. https://cde.nida.nih.gov/sites/nida_cde/files/PatientHealthQuestionnaire-2_v1.0_2014Jul2.pdf .
1136. https://geriatrictoolkit.missouri.edu/funct/Lawton_IADL.pdf .
1137. Platts-Mills TF, Dayaa JA, Reeve BB, et al. Development of the Emergency Department Senior Abuse Identification (ED Senior AID) tool. J Elder Abuse Negl. Aug-Oct 2018;30(4):247-270. doi:10.1080/08946566.2018.1460285.
1138. https://aspe.hhs.gov/reports/valuing-time-us-department-health-human-services-regulatory-impact-analyses-conceptual-framework .
1139. https://www.bls.gov/news.release/pdf/wkyeng.pdf . Accessed January 2, 2024.
1140. https://www.census.gov/library/stories/2023/09/median-household-income.html . Accessed January 2, 2024.
1141. https://datatools.ahrq.gov/hcupnet/ . Accessed January 3, 2024.
1142. https://www.aha.org/statistics/fast-facts-us-hospitals . Accessed January 3, 2024.
1143. Category One consists of hospitals that are located in a rural area (as defined in section 1886(d)(2)(D) of the Act) or have been reclassified being located in a rural area (pursuant to section 1886(d)(8)(E) of the Act). Category Two consists of hospitals in which the reference resident level of the hospital (as specified in section 1886(h)(10)(F)(iv) of the Act) is greater than the otherwise applicable resident limit. Category Three consists of hospitals located in States with new medical schools that received `Candidate School' status from the Liaison Committee on Medical Education (LCME) or that received `Pre-Accreditation' status from the American Osteopathic Association (AOA) Commission on Osteopathic College Accreditation (the COCA) on or after January 1, 2000, and that have achieved or continue to progress toward `Full Accreditation' status (as such term is defined by the LCME) or toward `Accreditation' status (as such term is defined by the COCA); or additional locations and branch campuses established on or after January 1, 2000, by medical schools with `Full Accreditation' status (as such term is defined by LCME) or `Accreditation' status (as such term is defined by the COCA). Category Four consists of hospitals that serve areas designated as HPSAs under section 332(a)(1)(A) of the Public Health Service Act (PHSA), as determined by the Secretary.
BILLING CODE 4120-01-P
BILLING CODE 4120-01-C
[ FR Doc. 2024-17021 Filed 8-1-24; 4:15 pm]
- Executive Orders
Reader Aids
Information.
- About This Site
- Legal Status
- Accessibility
- No Fear Act
- Continuity Information
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- HHS Author Manuscripts
- PMC10524827

Late-onset hereditary ataxias with dementia
Anthony j. linares.
1 Department of Neurology, David Geffen School of Medicine, University of California, Los Angeles, Los Angeles, CA, 90095 USA
Brent L. Fogel
2 Department of Human Genetics, David Geffen School of Medicine, University of California Los Angeles, 90095 USA
Purpose of review
Late-onset genetic cerebellar ataxias are clinically heterogenous with variable phenotypes. Several of these conditions are commonly associated with dementia. Recognition of the relationship between ataxia and dementia can guide clinical genetic evaluation.
Recent findings
Spinocerebellar ataxias often present with variable phenotypes that may include dementia. Genomic studies have begun to identify links between incomplete penetrance and such variable phenotypes in certain hereditary ataxias. Recent studies evaluating the interaction of TBP repeat expansions and STUB1 sequence variants provide a framework to understand how genetic interactions influence disease penetrance and dementia risk in spinocerebellar ataxia types 17 and 48. Further advances in next generation sequencing methods will continue to improve diagnosis and create new insights into the expressivity of existing disorders.
The late-onset hereditary ataxias are a clinically heterogenous group of disorders with complex presentations that can include cognitive impairment and/or dementia. Genetic evaluation of late-onset ataxia patients with dementia follows a systemic testing approach that often utilizes repeat expansion testing followed by next-generation sequencing. Advances in bioinformatics and genomics is improving both diagnostic evaluation and establishing a basis for phenotypic variability. Whole genome sequencing will likely replace exome sequencing as a more comprehensive means of routine testing.
INTRODUCTION
The hereditary ataxias are neurodegenerative conditions that affect the cerebellum and associated pathways, including the brainstem and corticospinal tracts.( 1 – 3 ) Core clinical features include cerebellar ataxia of gait, trunk, and/or limbs. Additional clinical findings can include dysarthria, ocular motor disturbances, peripheral neuropathy, spasticity, parkinsonism, seizures, and dementia. This makes the hereditary ataxias a clinically heterogenous group of conditions with significant phenotypic overlap posing a notable diagnostic challenge. Fortunately, advances in next-generation sequencing have enabled the development of molecular testing strategies to identify causative mutations.( 4 – 7 )
Many late-onset ataxia patients have complex presentations characterized by slowly progressive ataxia with variable extracerebellar features.( 1 ) In this review, we will focus on the emerging relationship between the late-onset hereditary ataxias and a specific associated clinical feature, dementia. A number of hereditary ataxias have known association with dementia phenotypes.( 8 , 9 ) In atypical cases, dementia may even be the main presenting symptom, which may further confound diagnostic evaluation.( 10 ) Below we will 1) briefly review the common genetic ataxias, 2) highlight late-onset hereditary ataxias associated with dementia, and 3) provide guidance regarding the genetic evaluation for hereditary ataxic disorders in patients presenting with dementia.
THE HEREDITARY ATAXIAS
The hereditary ataxias are typically classified by their mode of inheritance as autosomal dominant, autosomal recessive, X-linked, and mitochondrial. Repeat expansion disorders represent the most common molecular etiology, the majority of which show dominant inheritance.( 11 ) A brief review of the common classes of hereditary ataxias is provided, with an emphasis on the autosomal dominant disorders.
Autosomal dominant ataxias
The autosomal dominant ataxias are commonly referred to as the spinocerebellar ataxias (SCAs), although virtually all genetic ataxias involve degeneration of the cerebellum and spinocerebellar pathways. The SCAs make up the the most common causes of late-onset hereditary ataxia, with nearly 50 known associated disorders designated as SCA types.( 12 ) While nucleotide repeat expansions are responsible for the most frequently seen disorders, others arise from different causative molecular etiologies including point mutations and copy number variation ( Table 1 ).
Representative Genetic Ataxias.
| Disease | Gene | Class | Mutation | Selected Clinical Features |
|---|---|---|---|---|
| SCA1 | Dominant | CAG repeats | Pyramidal signs Amyotrophy Extrapyramidal signs Ophthalmoparesis | |
| SCA2 | Dominant | CAG repeats | Slow saccades Extrapyramidal signs Dementia (rarely) Ophthalmoplegia Peripheral neuropathy Pyramidal signs | |
| SCA3 | Dominant | CAG repeats | Pyramidal signs Amyotrophy Exophthalmos Extrapyramidal signs Ophthalmoparesis | |
| SCA5 SCAR14 | Dominant Recessive | Conventional mutations | Pure cerebellar ataxia (late onset) Cognitive impairment (early onset) Pyramidal signs (early onset) | |
| SCA6 | Dominant | CAG repeats | Pure cerebellar ataxia Late onset, usually >50 years | |
| SCA7 | Dominant | CAG repeats | Pigmentary macular degeneration Ophthalmoplegia Pyramidal signs | |
| SCA8 | Dominant | CAG/CTG repeats | Pyramidal signs Diminished vibratory sense Spastic and ataxic dysarthria | |
| SCA10 | Dominant | ATTCT repeats | Seizures Cognitive impairment Polyneuropathy Pyramidal signs | |
| SCA12 | Dominant | CAG repeats | Upper extremity tremor Mild or absent gait ataxia Hyperreflexia | |
| SCA14 | Dominant | Conventional mutations | Pure cerebellar ataxia Rarely chorea and cognitive deficits | |
| SCA15/SCA16/ SCA29 | Dominant | Conventional mutations | Pure cerebellar ataxia Rare tremor or cognitive impairment Congenital nonprogressive ataxia (SCA29) | |
| SCA17 | Dominant | CAG repeats | Chorea Dementia Extrapyramidal features Hyperreflexia Psychiatric symptoms | |
| SCA27A SCA27B | Dominant | Conventional mutations (SCA27A) GAA repeats (SCA27B) | Cognitive impairment (SCA27A) Orofacial dyskinesias (SCA27A) Tremor (SCA27A) Episodic (SCA27B) Vertigo (SCA27B) | |
| SCA48 | Dominant | Conventional mutations | Chorea Cognitive-Affective syndrome Dystonia Parkinsonism | |
| DRPLA Dentatorubral- Pallidoluysian Atrophy | Dominant | CAG repeats | Myoclonic epilepsy Choreoathetosis Dementia | |
| Friedreich Ataxia | Recessive | GAA repeats | Hyporeflexia Sensory polyneuropathy | |
| Ataxia telangiectasia | Recessive | Conventional mutations | Malignancy Oculomotor apraxia Telangiectasias | |
| Mediated Ataxia | Recessive | AAGGG repeats | Sensory polyneuropathy Vestibular dysfunction Chronic cough | |
| Autosomal recessive ataxia of Charlevoix-Saguenay | Recessive | Conventional mutations | Polyneuropathy Pyramidal signs | |
| Cerebrotendinous xanthomatosis | Recessive | Conventional mutations | Cataracts Tendon xanthomas | |
| Abetalipoproteinemia | Recessive | Conventional mutations | Lipid malabsorption Pigmentary retinopathy | |
| Ataxia with vitamin E deficiency | Recessive | Conventional mutations | Pigmentary retinopathy Polyneuropathy | |
| Ataxia with oculomotor apraxia, type 1 | Recessive | Conventional mutations | Oculomotor apraxia Polyneuropathy | |
| Ataxia with oculomotor apraxia, type 2 | Recessive | Conventional mutations | Oculomotor apraxia Polyneuropathy | |
| DNA polymerase gamma disorders | Recessive | Conventional mutations | Epilepsy Polyneuropathy Progressive external ophthalmoplegia | |
| Spinocerebellar ataxia autosomal recessive, type 8 | Recessive | Conventional mutations | Pure cerebellar | |
| FXTAS | X-linked | CGG repeats | Dementia Parkinsonism Polyneuropathy Pyramidal signs | |
| MELAS | Mitochondrial | Conventional mutations | Encephalopathy Myopathy Polyneuropathy Seizures | |
| MERRF | Mitochondrial | Conventional mutations | Epilepsy Myoclonus Myopathy Polyneuropathy |
Polyglutamine (CAG) repeat expansions are the most prevalent repeat expansion and are responsible for several types of SCAs including SCA1, SCA2, SCA3, SCA6, SCA7, SCA12, SCA17, and dentatorubral-pallidoluysian atrophy (DRPLA).( 13 – 15 ) Of all the repeat expansion disorders, SCA3 is the most frequently observed worldwide.( 13 ) Despite their commonality, the molecular mechanisms for how CAG repeat expansions result in ataxia are not completely understood. Potential mechanisms include toxic gain of function of RNA and protein biomolecules.( 13 )
Several repeat expansions occur in non-coding regions of their corresponding genes and the resulting disease including SCA8, SCA10, SCA12, SCA27B, SCA31, and SCA36.( 12 , 13 ) These expansions most frequently present in intronic regions but may also involve other mechanisms such as anti-sense transcripts (e.g., SCA8).( 16 ) Potential pathogenic causes include disrupted transcription and sequestration of mRNA processing complexes.( 12 , 13 ) Whole genome sequencing and improvements in bioinformatic analytic tools has recently enabled the discovery of novel non-coding repeat expansions, specifically SCA27B, which may be quite common worldwide.( 17 – 19 )
Hereditary ataxias caused by conventional mutations include point mutations, deletions, or duplications.( 20 ) Many of the resulting disorders can be grouped into general categories of function. For example, several genes are associated with ion-channel dysfunction (causing SCA5, SCA13, SCA15, SCA19, SCA27, SCA41, and the episodic ataxias) and others impair signal transduction (causing SCA11, SCA14, and SCA23).( 21 ) Copy number variations are typically causative in SCA15, SCA20, and SCA39.
Autosomal recessive ataxias
The autosomal recessive cerebellar ataxias (ARCAs) or SCARs (spinocerebellar ataxias, recessive) represent a heterogenous group of disorders associated with nearly 200 genes ( Table 1 ).( 22 ) Most ARCAs present in childhood or early adulthood, alongside prominent extracerebellar and non-neurological manifestations. Friedrich’s ataxia (FA), an intronic repeat expansion disorder, and ataxia-telangiectasia (AT) are the most common ARCAs worldwide, although the recently discovered non-coding repeat expansion disorder, RFC1- mediated ataxia, may be similarly common.( 23 – 25 ) While many ARCAs are early-onset disorders, there is phenotypic variation within conditions like FA, where both early-onset and late-onset presentations occur.( 26 , 27 ) The molecular pathways associated with the ARCAs are diverse and include defective DNA repair (e.g., AT), mitochondrial dysfunction (e.g., FA), impaired lipoprotein assembly (e.g., vitamin E deficiency), and numerous inborn errors of metabolism, among others.( 27 , 28 )
X-linked ataxias
The X-linked ataxias range from pure cerebellar phenotypes to more complex presentations involving developmental delay, parkinsonism, and dementia.( 29 ) The most common X-linked ataxia is Fragile X-associated tremor/ataxia syndrome (FXTAS), caused by repeat expansion in the FMR1 gene that is also associated with Fragile-X syndrome ( Table 1 ).( 30 ) While X-linked disease primarily affects hemizygous males, variable X-inactivation can occasionally result in female heterozygous presentations.( 29 )
Mitochondrial ataxias
Although several hereditary ataxias involve mitochondrial dysfunction (e.g., FA), mitochondrial ataxias are typically thought of as the maternally inherited disorders resulting from mutations in the mitochondrial genome.( 31 , 32 ) Notable mitochondrial ataxias include mitochondrial encephalomyopathy, lactic acidosis and stroke-like episodes (MELAS), myoclonic epilepsy with ragged red fibers (MERRF), and Kearns-Sayre syndrome ( Table 1 ). Given the key role of mitochondria in energy homeostasis, these conditions often have systemic and extracerebellar features including seizures, myopathy, myoclonus, and peripheral neuropathy.( 31 , 32 )
LATE-ONSET HEREDITARY ATAXIAS WITH DEMENTIA
Patients with late-onset hereditary ataxia, generally defined as having onset in adulthood to distinguish from childhood onset conditions, may exhibit features of cognitive impairment and dementia.( 8 , 9 ) Hereditary ataxias commonly associated with dementia include SCA2, SCA17, SCA48, DRPLA, and FXTAS ( Table 2 ). Others SCAs less frequently associated with cognitive impairment include SCA1, SCA3, SCA7, SCA8, and SCA12 ( Table 2 ). Extracerebellar features and radiographic findings may assist with distinguishing these conditions. However, variable progression and expression makes clinical diagnosis challenging. Here, we review the genetic conditions to consider when encountering a late-onset ataxia patient with a dementia phenotype.
Dominant late-onset hereditary ataxias associated with neurocognitive impairment.
| Disorder | Gene | Mutation | Neurocognitive Phenotype | Selected Neuroimaging Findings |
|---|---|---|---|---|
| SCA1 | CAG repeats | Cognitive impairment | Olivopontocerebellar atrophy. | |
| SCA2 | CAG repeats | Dementia | Olivopontocerebellar atrophy Cortical atrophy. | |
| SCA3 | CAG repeats | Cognitive impairment | Pontine, cortical, and spinal cord atrophy. | |
| SCA7 | CAG repeats | Cognitive impairment | Pontine and spinal cord atrophy. | |
| SCA8 | CAG repeats | Cognitive impairment | Pontine atrophy. | |
| SCA12 | CAG repeats | Cognitive impairment | Cortical atrophy. | |
| SCA17 | CAG repeats | Dementia | Pontocerebellar, cortical, and subcortical atrophy. | |
| SCA48 | Conventional mutations | Dementia | Posterior vermis atrophy. | |
| DRPLA | CAG repeats | Dementia | Brainstem and superior cerebellar peduncle atrophy. Hyperintensities in brainstem, thalamus, and cerebellum. | |
| FXTAS | CGG repeats | Dementia | Cortical atrophy. White matter hyperintensities in cerebrum and middle cerebellar peduncles. |
Repeat expansion disorders associated with dementia
SCA2 is characterized by slowly progressive ataxia and associated oculomotor deficits.( 33 ) Relevant extracerebellar findings include peripheral neuropathy, parkinsonism, and dementia. Mean age of onset is in the fourth decade of life. Affected individuals have 33 or more CAG repeats in the ATXN2 gene. Radiographic imaging demonstrates cortical, cerebellar, and brainstem atrophy ( Figure 1 ). Observational cohort studies have identified dementia as a relevant clinical feature, including examples of familial segregation.( 34 , 35 )

Sagittal and coronal views are shown. Regions of neurodegeneration typically seen on magnetic resonance imaging are indicated by darker shading.
DRPLA is characterized by ataxia, myoclonus, and epilepsy.( 36 ) DRPLA is most common in Asian populations (primarily Japanese). Affected individuals have 48–93 CAG repeats in the ATN1 gene. While mean age of onset is in the third decade of life, the age of clinical presentation is variable and can occur in the sixth and seventh decades of life. In older adults, core characteristic features include ataxia, choreoathetosis, and dementia. Movement disorders may mask the presentation of ataxia and mimic Huntington disease phenotypes.( 37 ) However, radiographic imaging demonstrates atrophy of the cerebellum and brainstem ( Figure 1 ).( 38 )
FXTAS is an X-linked ataxia, characterized by late-onset ataxia, tremor, and dementia.( 39 , 40 ) Additional extracerebellar findings including mild parkinsonism, peripheral neuropathy, and psychiatric symptoms. The average age of onset is in the sixth decade of life, with hemizygous males more commonly affected than heterozygous females. Clinical presentation is heterogenous with varied dominant signs. Affected individuals have a premutation-sized repeat of 55–200 CGG repeats in the 5’ UTR of the FMR1 gene. Radiographic imaging demonstrates generalized brain atrophy, and white matter hyperintensities in the cerebrum and middle cerebellar peduncles ( Figure 1 ).( 39 , 40 )
Linking ataxia and dementia in SCA17 and SCA48 through the digenic interaction of TBP and STUB1
SCA17 and SCA48 are dominantly inherited SCAs with significant phenotypic overlap resembling Huntington disease.( 37 , 41 , 42 ) Recent studies highlight the role of digenic interactions in SCA17 and SCA48, with implications for disease penetrance and dementia risk.( 43 – 45 ) These studies have identified genetic interactions between the TBP and STUB1 genes, which are responsible for SCA17 and SCA48 respectively.
SCA17 is characterized by ataxia, dementia, chorea, and dystonia.( 42 ) There is consequently a significant phenotypic overlap with Huntington disease.( 37 ) Radiographic imaging demonstrates variable cortical, brainstem, and cerebellar atrophy ( Figure 1 ).( 46 ) Affected individuals have >50 CAG repeats in the TBP gene, while intermediate-size expansions (41–49 repeats) show incomplete penetrance. These recent studies observed that 49–97% individuals with intermediate-sized TBP expansions also carried a heterozygous sequence variant in the STUB1 gene.( 43 , 44 ) Segregation analyses found that the presence of either the intermediate TBP expansion or the STUB1 variant alone was insufficient to cause disease. These findings suggest that STUB1 modifies SCA17 penetrance in intermediate sized TBP expansions ( Figure 2 ).

Intermediate sized TBP repeat expansions (41–49 repeats) are shown in blue. Such smaller repeat sizes are considered to be incompletely penetrance and, in this model, by themselves they do not cause disease (arrow). In the presence of a heterozygous STUB1 variant (yellow), however, these repeats become penetrant and cause SCA17. These STUB1 variants are insufficient to cause disease in absence of the TBP allele (arrow). However, in patients with a pathogenic STUB1 variant associated with SCA48 (red), the presence of an intermediate TBP repeat expansion modifies the phenotype of SCA48, leading to an increased risk of severe dementia.
SCA48 is characterized by ataxia and dementia.( 41 ) Additional clinical features include neuropsychiatric symptoms, parkinsonism, chorea, and dystonia so it is therefore also considered a phenotypic mimic of Huntington disease.( 41 ) Radiographic imaging demonstrates atrophy of the posterior cerebellar vermis ( Figure 1 ).( 38 ) Affected individuals carry a pathogenic variant in the STUB1 gene. A recent study observed that 40% of SCA48 patients carry an intermediate-sized TBP expansion.( 43 ) The study also observed that the longer the TBP repeat length, the more likely the occurrence of cognitive impairment and the rate of disease progression.( 43 ) This implicates TBP repeat expansions as a disease modifier of SCA48, leading to an increased risk of dementia ( Figure 2 ). Together these studies illustrate how complex interactions among genes can affect disease development, penetrance, progression, and prognosis.( 47 )
SHARED MECHANISMS OF CEREBELLAR ATAXIA AND DEMENTIA
The mechanisms mediating dementia in the late-onset genetic cerebellar ataxia are likely multifactorial. This includes the role of the cerebellum in cognition, extracerebellar neurodegeneration, and the presence of co-occurring neurodegenerative pathologies.
The cerebellum and cognition
The cerebellum is recognized to regulate cognitive processing and emotional control.( 48 ) Functional studies have implicated cerebellar posterior lobe dysfunction in cerebellar cognitive affective syndrome (CCAS).( 49 ) CCAS is characterized by impairments in executive function, visual spatial processing, language, and affect. To quantify cognitive impairment secondary to cerebellar dysfunction, the CCAS scale was developed.( 50 ) Application of the CCAS scale in SCA3 patients detected neuropsychological deficits that correlated with disease severity.( 51 ) Further research is needed, however, to broadly characterize the cerebellar-cognitive affective phenotypes across all the late-onset hereditary ataxias.
Extracerebellar degeneration and cognition
Extracerebellar degeneration, such as cortical atrophy, is observed in late-onset hereditary ataxias such as SCA2, SCA17, and FXTAS ( Figure 1 ).( 38 ) In these cases, atrophy of key association areas may contribute to cognitive impairment. In SCA17, extracerebellar atrophy involves the basal ganglia in addition to the frontotemporal lobes. The resulting disruption of fronto-subcortical circuits may result in motor, cognitive, and behavioral dysfunction, similar to a Huntington’s disease pathology.( 52 ) In-depth neurocognitive testing may aid in differentiating the pathways mediating cognitive impairment specifically associated with cerebellar versus extracerebellar degeneration.
Late-onset hereditary ataxias and other co-morbidities
Like the general population, patients with hereditary ataxias are at risk for developing age-related neurodegenerative disorders such as Alzheimer’s disease (AD). While intronic variants in ATXN1 have been associated with AD risk, variations in CAG repeat number in AD patients have not been associated.( 53 ) However, neuropathological studies often demonstrate neuronal intranuclear inclusions in CAG disorders and extra-cerebellar degeneration.( 54 , 55 ) Whether or not there are shared genetic risk modifiers between late-onset ataxia and dementia, and whether patients with genetic cerebellar ataxias may have an altered risk for the development of AD, is not well understood.
GENETIC EVALUTION OF LATE-ONSET ATAXIA AND DEMENTIA
Here we provide an approach for the genetic evaluation of the hereditary ataxias in patients with dementia. A detailed clinical phenotype is important for guiding a differential diagnosis and subsequent genetic testing. In some clinical cases, patients may have a primary dementia presentation with evidence of cerebellar dysfunction on examination and/or neuroimaging.
Phenotypic evaluation
The history and physical should focus on identifying signs of cerebellar and extracerebellar dysfunction.( 56 ) Features of cerebellar dysfunction include dysarthria, dysphagia, ocular dysmetria, direction-changing nystagmus, limb dysmetria, and gait imbalance. Relevant extracerebellar findings, in addition to cognitive impairment, include tremor, parkinsonism, chorea, dysautonomia, and neuropsychiatric features. If dementia is noted in the presence of ataxia, evaluation for the presence of the common cerebellar cognitive affective syndrome should be considered.( 50 )
A positive family history increases the likelihood of a genetic etiology. Relating the family history to inheritance patterns may further inform the differential. For example, late-onset ataxia with dementia in a male proband with a grandson with intellectual disability and/or autism should raise suspicion for FXTAS.( 39 ) A negative family history should not exclude a genetic basis because family histories may be limited, certain inheritances may not be apparent due to small family trees, or mutations may be de novo .( 4 , 57 )
Magnetic resonance imaging (MRI) of the brain will assess the degree of cerebellar atrophy and associated cortical and brainstem atrophy. Evidence of significant cerebellar atrophy in a primary dementia patient should raise suspicion for a late-onset genetic cerebellar ataxia. Radiographic findings such as the “middle cerebellar peduncle” sign is an indicator of a potential FXTAS diagnosis.( 38 ) Imaging findings that include cerebellar, fronto-temporal, and basal ganglia atrophy should raise suspicion for SCA17.( 42 ) While radiographic findings are not entirely specific, they can guide an initial differential ( Figure 1 ).
Neurocognitive screening should be performed in symptomatic patients. Clinical screening tests include the Mini-Mental Status Examination (MMSE), the Montreal Cognitive Assessment (MoCA), and CCAS scale.( 50 ) In-depth neuropsychological testing may assist with differentiating cerebellar, extracerebellar, and other neurodegenerative processes. Patients who are identified to have hereditary ataxias associated with dementia and cognitive impairment should have baseline and interval testing performed to aid dementia management and assess progression.
Genetic testing
The initial evaluation of patients with late-onset cerebellar ataxia and dementia involves the investigation of reversible etiologies. Evaluating treatable causes is essential, especially in sporadic cases with rapid progression. While such acquired evaluations are reviewed in more-depth elsewhere, they broadly evaluate for metabolic, autoimmune, nutritional, paraneoplastic, and infectious etiologies.( 21 ) Once acquired causes are excluded, a systematic examination for genetic etiologies is appropriate for both familial and sporadic cases.( 4 , 5 ) We recommend a two-staged genetic testing approach for the hereditary ataxias in patients with dementia.( 57 )
Initial genetic testing should comprehensively evaluate for the known repeat expansion disorders associated with the late-onset SCAs. SCA1, SCA2, SCA3, SCA6, and SCA7 represent 50–60% of all dominantly inherited ataxia disorders.( 12 , 21 ) Ataxia repeat expansion panel testing is often the most cost-effective and high yield initial testing methodology and should include the genes associated with dementia (SCA2, SCA17, DRPLA, and FXTAS) and cognitive impairment (SCA1, SCA3, SCA7, SCA8, and SCA12).( 2 , 57 ) It is important to note that, at present, repeat expansion testing requires specialized testing that quantifies repeat size and such mutations will not be recognized on the short-read next-generation sequencing platforms typically used for exome or genome sequencing.( 58 )
Once common repeat expansion disorders have been excluded, a comprehensive evaluation for non-repeat expansion disorders is recommended. Whole exome sequencing (WES) is often the preferred modality because it has increases the diagnostic rate of the genetic ataxias in a cost-effective manner.( 58 , 59 ) WES will comprehensively cover virtually all other dominant, recessive, and X-linked disorders, particularly if bioinformatic assessment of copy number variation is included. In an adult-onset cohort of cerebellar ataxia and/or spastic paraplegia patients, WES can typically identify 25–50% of clinically relevant genetic variants.( 4 , 5 )
Additional clinical genetic testing not adequately covered by repeat expansion panels and WES can be considered on a case-by-case basis and the patient’s phenotype. For example, ataxia repeat expansions tests are panel based and typically do not cover other disease-causing expansions such as C9orf72 , which has rarely been reported to present with ataxia and cerebellar degeneration.( 60 ) Likewise, Huntington disease shares phenotypic overlap with DRPLA, SCA48, and SCA17 and HTT expansion may need to be considered. Technical limitations of WES should be considered, including the detection of chromosomal variants, repeat expansions, non-coding variants, and mitochondrial genomic variants.( 58 )
In the future, whole genome sequencing (WGS) may prove a more efficient means to overcome the disease-specific limitations of directed panel testing and the technical limitations of WES.( 58 ) WGS can already evaluate and discover causative genetic variants in coding and non-coding regions, including structural changes such as copy number variations (CNVs).( 58 , 61 ) A recent study demonstrated, both retrospectively and prospectively, how advances in bioinformatic technologies have the potential to enable WGS to detect most individuals with certain repeat expansions.( 62 )
Conclusions
The hereditary ataxias are a clinically heterogeneous group of disorders with significant phenotypic overlap. Dementia is an important extracerebellar feature of several late-onset hereditary ataxias and is most frequently associated with SCA2, SCA17, SCA48, DRPLA, and FXTAS. Cognitive dysfunction and subclinical cognitive impairment are likely unrecognized features in the SCAs. An evolving challenge is understanding the interplay between incomplete penetrance, variable phenotypes such as dementia, and disease prognosis in the late-onset hereditary ataxias. Recent advances in genomics are beginning to uncover such relationships. As we leverage comprehensive genomic information from exome and genome sequencing, a future goal will be to identify multigenic risk factors that may better inform clinicians and patients about disease development and progression.
Key points:
- Genetic cerebellar ataxias are clinically heterogeneous with variable phenotypes, inheritance patterns, and molecular etiologies.
- Several forms of late-onset genetic ataxia are frequently associated with dementia or cognitive impairment.
- Advances in genomics and bioinformatics are improving diagnostic capability, aiding the discovery of new ataxia genes, and identifying disease-modifying genetic interactions among variants in known genes.
Acknowledgements:
Funding sources for the study:.
AJL was supported by the National Institutes of Health R25 NS065723.
Financial disclosure/Conflict of Interest:
All authors declare that there is no conflict of interest.

IMAGES
VIDEO
COMMENTS
Fragile X syndrome (FXS), also known as Martin-Bell syndrome in the past, is a non-Mendelian trinucleotide repeat disorder. FXS is the most prevalent inherited cause of mild-to-severe intellectual disability and the most common monogenic cause of autism spectrum disorder.[1][2] It accounts for about one-half of cases of X-linked intellectual disability and is the most common cause of mental ...
Fragile X syndrome is the monogenetic condition that produces more cases of autism and intellectual disability. The repetition of CGG triplets (> 200) and their methylation entail the silencing of the FMR1 gene. ... Fragile X syndrome: clinical presentation, pathology and treatment Gac Med Mex. 2020;156(1):60-66. doi: 10.24875/GMM.19005275.
This review aims to assemble many years of research and clinical experience in the fields of neurodevelopment and neuroscience to present an up-to-date understanding of the clinical presentation, molecular and brain pathology associated with Fragile X syndrome, a neurodevelopmental condition that develops with the full mutation of the FMR1 gene, located in the q27.3 loci of the X chromosome ...
Patients may have recurrent sinusitis, otitis media, and decreased visual acuity. During the history taking, ask about apnea. [ 16] Fragile X syndrome, also termed Martin-Bell syndrome or marker X syndrome, is the most common cause of inherited mental retardation, intellectual disability, and autism and is the second most common cause of ...
Fragile X syndrome (FXS) is an X-linked disorder and the most common inherited cause of intellectual disability [ 1 ]. Both males and females can be affected. The clinical features and diagnosis of FXS in children and adolescents are discussed in this topic review. Prenatal screening and the management of FXS in children and adolescents are ...
Fragile X syndrome (FXS) is a genetic disorder and one of the most common causes of inherited intellectual disability. FXS affects both males and females. However, females often have milder symptoms than males. A diagnosis of FXS can be helpful to the family because it can provide a reason for a child's intellectual disabilities and behavior ...
Abstract. Fragile X syndrome is the monogenetic condition that produces more cases of autism and intellectual disability. The repetition. of CGG triplets (> 200) and their methylation entail the ...
Genetics of fragile X. Fragile X syndrome (FXS), an X-linked condition first described by Martin and Bell (), is the leading cause of inherited intellectual disability (ID).Estimates report that FXS affects approximately 1 in 2,500 to 5,000 men and 1 in 4,000 to 6,000 women (2, 3).FXS is caused by mutations in the FMR1 gene, which is located on the X chromosome and whose locus at Xq27.3 ...
This website requires cookies, and the limited processing of your personal data in order to function. By using the site you are agreeing to this as outlined in our privacy notice and cookie policy.
Screening for fragile X syndrome is performed by polymerase chain reaction and current recommendation of the American Academy of Pediatrics is to test individuals with intellectual disability, global developmental retardation or with a family history of presence of the mutation or premutation. Fragile X syndrome is the monogenetic condition that produces more cases of autism and intellectual ...
Fragile X syndrome (FXS) is a neurodevelopmental disorder caused by the full mutation as well as highly localized methylation of the fragile X mental retardation 1 (FMR1) gene on the long arm of the X chromosome.Children with FXS are commonly co-diagnosed with Autism Spectrum Disorder, attention and learning problems, anxiety, aggressive behavior and sleep disorder, and early interventions ...
The first clinical clue in children often is delayed attainment of one or more developmental milestones. 2, 3 On average, boys with fragile X syndrome sit without support at 10 months of age and ...
This review aims to assemble many years of research and clinical experience in the fields of neurodevelopment and neuroscience to present an up-to-date understanding of the clinical presentation, molecular and brain pathology associated with Fragile X syndrome, a neurodevelopmental condition that develops with the full mutation of the FMR1 gene, located in the q27.3 loci of the X chromosome ...
The following sections describe pathophysiology and clinical presentation of FXS, as well as a variety of therapeutic approaches. 2. The Pathophysiology of Fragile X Syndrome. FXS is the most common form of inherited intellectual disability (ID) and monogenic cause of Autism Spectrum Disorder (ASD) [ 22 ].
The clinical spectrum of FXS is wide, presenting not only as an isolated intellectual disability but as a multi-systemic condition, involving predominantly the central nervous system but potentially affecting any apparatus. ... Background: Fragile X Syndrome (FXS) is the second cause of intellectual disability after Down syndrome and the most ...
Fragile X syndrome is the monogenetic condition that produces more cases of autism and intellectual disability. The repetition of CGG triplets (> 200) and their methylation entail the silencing of the FMR1 gene. The FMRP protein (product of the FMR1 gene) interacts with ribosomes by controlling the translation of specific messengers, and its ...
Fragile X syndrome: clinical presentation, pathology and treatment ... Fragile X syndrome is the monogenetic condition that produces more cases of autism and intellectual disability. The repetition
Summary. The fragile X mental retardation 1 gene, which codes for the fragile X mental retardation 1 protein, usually has 5 to 40 CGG repeats in the 5′ untranslated promoter. The full mutation is the almost always the cause of fragile X syndrome (FXS). The prevalence of FXS is about 1 in 4,000 to 1 in 7,000 in the general population although ...
Presented by Elizabeth Berry-Kravis, MD, PhD, Craig A. Erickson, MD, and Randi J. Hagerman, MD. This session will be a family-friendly joint presentation of results from recently completed, and a description of all currently active clinical trials and development programs for new medications in fragile X syndrome (FXS).
Keynote: Clinical Trials in Fragile X Syndrome — Presentation Dany Petraska 2023-12-20T16:57:25-05:00 Jul 15, 2022 | This keynote session explore completed and active clinical trials relating to Fragile X syndrome.
11560 Ciudad de México (México) Mallorca, 310. 08037 Barcelona (España) Legal Notice (Aviso Legal) | Permissions & Reprints | Data Protection Policy | Ethics Code | Conflicts of Interest. Fragile X syndrome is the monogenetic condition that produces more cases of autism and intellectual disability. The repetition of CGG triplets (> 200...
Fragile X syndrome (FXS) is the most common monogenic form of inherited intellectual disability and autism spectrum disorder (ASD). More than 99% of individuals with FXS are caused by the unstable expansion of CGG repeats located within the 5'-untranslated region of the FMR1 gene. The clinical features of FXS include various degrees of ...
Background Dermatofibromas, also known as benign fibrous histiocytomas, are among the most common cutaneous soft-tissue lesions. Association of multiple dermatofibromas with some diseases was described and it has not been reported with Ehlers-Danlos syndrome before. We present a case with Ehlers-Danlos syndrome and multiple dermatofibromas. Case presentation An 18-year-old Iranian woman ...
The UC Davis MIND Institute is an internationally recognized research, education and clinical care center. It brings together researchers and providers in many different specialties, all dedicated to neurodevelopmental conditions such as autism, fragile X syndrome and Down syndrome.
AGENCY: Centers for Medicare & Medicaid Services (CMS), Department of Health and Human Services (HHS). ACTION: Final rule. SUMMARY: This final rule revises the Medicare hospital inpatient prospective payment systems (IPPS) for operating and capital-related costs of acute care hospitals; makes changes relating to Medicare graduate medical education (GME) for teaching hospitals; updates the ...
The X-linked ataxias range from pure cerebellar phenotypes to more complex presentations involving developmental delay, parkinsonism, and dementia. The most common X-linked ataxia is Fragile X-associated tremor/ataxia syndrome (FXTAS), caused by repeat expansion in the FMR1 gene that is also associated with Fragile-X syndrome (Table 1). While X ...