
- Duke NetID Login
- 919.660.1100
- Duke Health Badge: 24-hour access
- Accounts & Access
- Databases, Journals & Books
- Request & Reserve
- Training & Consulting
- Request Articles & Books
- Renew Online
- Reserve Spaces
- Reserve a Locker
- Study & Meeting Rooms
- Course Reserves
- Pay Fines/Fees
- Recommend a Purchase
- Access From Off Campus
- Building Access
- Computers & Equipment
- Wifi Access
- My Accounts
- Mobile Apps
- Known Access Issues
- Report an Access Issue
- All Databases
- Article Databases
- Basic Sciences
- Clinical Sciences
- Dissertations & Theses
- Drugs, Chemicals & Toxicology
- Grants & Funding
- Interprofessional Education
- Non-Medical Databases
- Search for E-Journals
- Search for Print & E-Journals
- Search for E-Books
- Search for Print & E-Books
- E-Book Collections
- Biostatistics
- Global Health
- MBS Program
- Medical Students
- MMCi Program
- Occupational Therapy
- Path Asst Program
- Physical Therapy
- Researchers
- Community Partners

Conducting Research
- Archival & Historical Research
- Black History at Duke Health
- Data Analytics & Viz Software
- Data: Find and Share
- Evidence-Based Practice
- NIH Public Access Policy Compliance
- Publication Metrics
- Qualitative Research
- Searching Animal Alternatives
Systematic Reviews
- Test Instruments
Using Databases
- JCR Impact Factors
- Web of Science
Finding & Accessing
- COVID-19: Core Clinical Resources
- Health Literacy
- Health Statistics & Data
- Library Orientation
Writing & Citing
- Creating Links
- Getting Published
- Reference Mgmt
- Scientific Writing
Meet a Librarian
- Request a Consultation
- Find Your Liaisons
- Register for a Class
- Request a Class
- Self-Paced Learning
Search Services
- Literature Search
- Systematic Review
- Animal Alternatives (IACUC)
- Research Impact
Citation Mgmt
- Other Software
Scholarly Communications
- About Scholarly Communications
- Publish Your Work
- Measure Your Research Impact
- Engage in Open Science
- Libraries and Publishers
- Directions & Maps
- Floor Plans
Library Updates
- Annual Snapshot
- Conference Presentations
- Contact Information
- Gifts & Donations
- What is a Systematic Review?
- Types of Reviews
- Manuals and Reporting Guidelines
- Our Service
- 1. Assemble Your Team
2. Develop a Research Question
- 3. Write and Register a Protocol
- 4. Search the Evidence
- 5. Screen Results
- 6. Assess for Quality and Bias
- 7. Extract the Data
- 8. Write the Review
- Additional Resources
- Finding Full-Text Articles
A well-developed and answerable question is the foundation for any systematic review. This process involves:
- Systematic review questions typically follow a PICO-format (patient or population, intervention, comparison, and outcome)
- Using the PICO framework can help team members clarify and refine the scope of their question. For example, if the population is breast cancer patients, is it all breast cancer patients or just a segment of them?
- When formulating your research question, you should also consider how it could be answered. If it is not possible to answer your question (the research would be unethical, for example), you'll need to reconsider what you're asking
- Typically, systematic review protocols include a list of studies that will be included in the review. These studies, known as exemplars, guide the search development but also serve as proof of concept that your question is answerable. If you are unable to find studies to include, you may need to reconsider your question
Other Question Frameworks
PICO is a helpful framework for clinical research questions, but may not be the best for other types of research questions. Did you know there are at least 25 other question frameworks besides variations of PICO? Frameworks like PEO, SPIDER, SPICE, and ECLIPS can help you formulate a focused research question. The table and example below were created by the Medical University of South Carolina (MUSC) Libraries .
The PEO question framework is useful for qualitative research topics. PEO questions identify three concepts: population, exposure, and outcome. Research question : What are the daily living experiences of mothers with postnatal depression?
| opulation | Who is my question focused on? | mothers |
| xposure | What is the issue I am interested in? | postnatal depression |
| utcome | What, in relation to the issue, do I want to examine? | daily living experiences |
The SPIDER question framework is useful for qualitative or mixed methods research topics focused on "samples" rather than populations. SPIDER questions identify five concepts: sample, phenomenon of interest, design, evaluation, and research type.
Research question : What are the experiences of young parents in attendance at antenatal education classes?
| Element | Definition | Example |
|---|---|---|
| ample | Who is the group of people being studied? | young parents |
| henomenon of nterest | What are the reasons for behavior and decisions? | attendance at antenatal education classes |
| esign | How has the research been collected (e.g., interview, survey)? | interviews |
| valuation | What is the outcome being impacted? | experiences |
| esearch type | What type of research (qualitative or mixed methods)? | qualitative studies |
The SPICE question framework is useful for qualitative research topics evaluating the outcomes of a service, project, or intervention. SPICE questions identify five concepts: setting, perspective, intervention/exposure/interest, comparison, and evaluation.
Research question : For teenagers in South Carolina, what is the effect of provision of Quit Kits to support smoking cessation on number of successful attempts to give up smoking compared to no support ("cold turkey")?
| Element | Definition | Example |
|---|---|---|
| etting | Setting is the context for the question (where). | South Carolina |
| erspective | Perspective is the users, potential users, or stakeholders of the service (for whom). | teenagers |
| ntervention / Exposure | Intervention is the action taken for the users, potential users, or stakeholders (what). | provision of Quit Kits to support smoking cessation |
| omparison | Comparison is the alternative actions or outcomes (compared to what). | no support or "cold turkey" |
| valuation | Evaluation is the result or measurement that will determine the success of the intervention (what is the result, how well). | number of successful attempts to give up smoking with Quit Kits compared to number of successful attempts with no support |
The ECLIPSE framework is useful for qualitative research topics investigating the outcomes of a policy or service. ECLIPSE questions identify six concepts: expectation, client group, location, impact, professionals, and service.
Research question: How can I increase access to wireless internet for hospital patients?
| xpectation | What are you looking to improve or change? What is the information going to be used for? | to increase access to wireless internet in the hospital |
| lient group | Who is the service or policy aimed at? | patients and families |
| ocation | Where is the service or policy located? | hospitals |
| mpact | What is the change in service or policy that the researcher is investigating? | clients have easy access to free internet |
| rofessionals | Who is involved in providing or improving the service or policy? | IT, hospital administration |
| rvice | What kind of service or policy is this? | provision of free wireless internet to patients |
- << Previous: 1. Assemble Your Team
- Next: 3. Write and Register a Protocol >>
- Last Updated: Jun 18, 2024 9:41 AM
- URL: https://guides.mclibrary.duke.edu/sysreview
- Duke Health
- Duke University
- Duke Libraries
- Medical Center Archives
- Duke Directory
- Seeley G. Mudd Building
- 10 Searle Drive
- [email protected]
Health (Nursing, Medicine, Allied Health)
- Find Articles/Databases
- Reference Resources
- Evidence Summaries & Clinical Guidelines
- Drug Information
- Health Data & Statistics
- Patient/Consumer Facing Materials
- Images and Streaming Video
- Grey Literature
- Mobile Apps & "Point of Care" Tools
- Tests & Measures This link opens in a new window
- Citing Sources
- Selecting Databases
- Framing Research Questions
- Crafting a Search
- Narrowing / Filtering a Search
- Expanding a Search
- Cited Reference Searching
- Saving Searches
- Term Glossary
- Critical Appraisal Resources
- What are Literature Reviews?
- Conducting & Reporting Systematic Reviews
- Finding Systematic Reviews
- Tutorials & Tools for Literature Reviews
- Finding Full Text
Defining the Question: Foreground & Background Questions
In order to most appropriately choose an information resource and craft a search strategy, it is necessary to consider what kind of question you are asking: a specific, narrow "foreground" question, or a broader background question that will help give context to your research?
Foreground Questions
A "foreground" question in health research is one that is relatively specific, and is usually best addressed by locating primary research evidence.
Using a structured question framework can help you clearly define the concepts or variables that make up the specific research question.
Across most frameworks, you’ll often be considering:
- a who (who was studied - a population or sample)
- a what (what was done or examined - an intervention, an exposure, a policy, a program, a phenomenon)
- a how ([how] did the [what] affect the [who] - an outcome, an effect).
PICO is the most common framework for developing a clinical research question, but multiple question frameworks exist.
PICO (Problem/Population, Intervention, Comparison, Outcome)
Appropriate for : clinical questions, often addressing the effect of an intervention/therapy/treatment
Example : For adolescents with type II diabetes (P) does the use of telehealth consultations (I) compared to in-person consultations (C) improve blood sugar control (O)?
| Element | Description | Example |
|---|---|---|
| opulation / problem | Who is the group of people being studied? | adolescents with T2D |
| ntervention | What is the intervention being investigated? (independent variable) | telehealth consultations |
| omparison | To what is the intervention being compared? | in person consultations |
| utcome | What are the desired outcomes of the intervention? (dependent variable) | blood sugar control |
Framing Different Types of Clinical Questions with PICO
Different types of clinical questions are suited to different syntaxes and phrasings, but all will clearly define the PICO elements. The definitions and frames below may be helpful for organizing your question:
Intervention/Therapy
Questions addressing how a clinical issue, illness, or disability is treated.
"In__________________(P), how does__________________(I) compared to_________________(C) affect______________(O)?"
Questions that address the causes or origin of disease, the factors which produce or predispose toward a certain disease or disorder.
"Are_________________(P), who have_________________(I) compared with those without_________________(C) at_________________risk for/of_________________(O) over_________________(T)?"
Questions addressing the act or process of identifying or determining the nature and cause of a disease or injury through evaluation.
In_________________(P) are/is_________________(I) compared with_________________(C) more accurate in diagnosing_________________(O)?
Prognosis/Prediction:
Questions addressing the prediction of the course of a disease.
In_________________(P), how does_________________(I) compared to_________________ (C) influence_________________(O)?
Questions addressing how one experiences a phenomenon or why we need to approach practice differently.
"How do_________________(P) with_________________(I) perceive_________________(O)?"
Adapted from: Melnyk, B. M., & Fineout-Overholt, E. (2011). Evidence-based practice in nursing & healthcare: A guide to best practice. Philadelphia: Wolters Kluwer/Lippincott Williams & Wilkins.
Beyond PICO: Other Types of Question Frameworks
PICO is a useful framework for clinical research questions, but may not be appropriate for all kinds of reviews. Also consider:
PEO (Population, Exposure, Outcome)
Appropriate for : describing association between particular exposures/risk factors and outcomes
Example : How do preparation programs (E) influence the development of teaching competence (O) among novice nurse educators (P)?
| Element | Description | Example |
|---|---|---|
| opulation | Who is the group of people being studied? | novice nurse educators |
| xposure | What is the population being exposed to (independent variable)? | preparation programs |
| utcome | What is the outcome that may be affected by the exposure (dependent variable)? | teaching competence |
SPIDER (Sample, Phenomenon of Interest, Design, Evaluation, Research Type)
Appropriate for : questions of experience or perspectives (questions that may be addressed by qualitative or mixed methods research)
Example : What are the experiences and perspectives (E) of undergraduate nursing students (S) in clinical placements within prison healthcare settings (PI)?
| Element | Description | Example |
|---|---|---|
| ample | Who is the group of people being studied? | undergraduate nursing students |
| henomenon of nterest | What are the reasons for behavior and decisions? | clinical placements in prison healthcare settings |
| esign | How has the research been collected (e.g., interview, survey)? | interview and surveys |
| valuation | What is the outcome being impacted? | attitudes, experiences and reflections on learning |
| esearch type | What type of research? | qualitative, quantitative or mixed methods |
SPICE (Setting, Perspective, Intervention/phenomenon of Interest, Comparison, Evaluation)
Appropriate for : evaluating the outcomes of a service, project, or intervention
Example : What are the impacts and best practices for workplace (S) transition support programs (I) for the retention (E) of newly-hired, new graduate nurses (P)?
| Element | Description | Example |
|---|---|---|
| etting | What is the context for the question? (Where?) | nursing workplaces (healthcare settings) |
| erspective | For whom is this intervention/program/service designed (users, potential users, stakeholders)? | new graduate nurses |
| ntervention/Interest/Exposure | What action is taken for the users, potential users, or stakeholders? | long term transition support programs (residency/mentorship) |
| omparison | What are the alternative interventions? | no or limited transition support / orientation |
| valuation | What is the results of the intervention or service/how is success measured? | retention of newly hired nurses |
PCC (Problem/population, Concept, Context)
Appropriate for : broader (scoping) questions
Example : How do nursing schools (Context) teach, measure, and maintain nursing students ' (P) technological literacy (Concept))throughout their educational programs?
| Element | Description | Example |
|---|---|---|
| What are the important characteristics of the participants, or the problem of focus? | nursing students | |
| oncept | What is the core concept being examined by the review? | technological literacy |
| ontext | What is the context for the question? (Could include geographic location, or details about the setting of interest)? | nursing schools |
Background Questions
To craft a strong and reasonable foreground research question, it is important to have a firm understanding of the concepts of interest. As such, it is often necessary to ask background questions, which ask for more general, foundational knowledge about a disorder, disease, patient population, policy issue, etc.
For example, consider the PICO question outlined above:
"For adolescents with type II diabetes does the use of telehealth consultations compared to in-person consultations improve blood sugar control ?
To best make sense of the literature that might address this PICO question, you would also need a deep understanding of background questions like:
- What are the unique barriers or challenges related to blood sugar management in adolescents with TII diabetes?
- What are the measures of effective blood sugar control?
- What kinds of interventions would fall under the umbrella of 'telehealth'?
- What are the qualitative differences in patient experience in telehealth versus in-person interactions with healthcare providers?
- << Previous: Selecting Databases
- Next: Crafting a Search >>
- Last Updated: Aug 9, 2024 11:03 AM
- URL: https://guides.nyu.edu/health

Research Question Examples 🧑🏻🏫
25+ Practical Examples & Ideas To Help You Get Started
By: Derek Jansen (MBA) | October 2023
A well-crafted research question (or set of questions) sets the stage for a robust study and meaningful insights. But, if you’re new to research, it’s not always clear what exactly constitutes a good research question. In this post, we’ll provide you with clear examples of quality research questions across various disciplines, so that you can approach your research project with confidence!
Research Question Examples
- Psychology research questions
- Business research questions
- Education research questions
- Healthcare research questions
- Computer science research questions
Examples: Psychology
Let’s start by looking at some examples of research questions that you might encounter within the discipline of psychology.
How does sleep quality affect academic performance in university students?
This question is specific to a population (university students) and looks at a direct relationship between sleep and academic performance, both of which are quantifiable and measurable variables.
What factors contribute to the onset of anxiety disorders in adolescents?
The question narrows down the age group and focuses on identifying multiple contributing factors. There are various ways in which it could be approached from a methodological standpoint, including both qualitatively and quantitatively.
Do mindfulness techniques improve emotional well-being?
This is a focused research question aiming to evaluate the effectiveness of a specific intervention.
How does early childhood trauma impact adult relationships?
This research question targets a clear cause-and-effect relationship over a long timescale, making it focused but comprehensive.
Is there a correlation between screen time and depression in teenagers?
This research question focuses on an in-demand current issue and a specific demographic, allowing for a focused investigation. The key variables are clearly stated within the question and can be measured and analysed (i.e., high feasibility).

Examples: Business/Management
Next, let’s look at some examples of well-articulated research questions within the business and management realm.
How do leadership styles impact employee retention?
This is an example of a strong research question because it directly looks at the effect of one variable (leadership styles) on another (employee retention), allowing from a strongly aligned methodological approach.
What role does corporate social responsibility play in consumer choice?
Current and precise, this research question can reveal how social concerns are influencing buying behaviour by way of a qualitative exploration.
Does remote work increase or decrease productivity in tech companies?
Focused on a particular industry and a hot topic, this research question could yield timely, actionable insights that would have high practical value in the real world.
How do economic downturns affect small businesses in the homebuilding industry?
Vital for policy-making, this highly specific research question aims to uncover the challenges faced by small businesses within a certain industry.
Which employee benefits have the greatest impact on job satisfaction?
By being straightforward and specific, answering this research question could provide tangible insights to employers.
Examples: Education
Next, let’s look at some potential research questions within the education, training and development domain.
How does class size affect students’ academic performance in primary schools?
This example research question targets two clearly defined variables, which can be measured and analysed relatively easily.
Do online courses result in better retention of material than traditional courses?
Timely, specific and focused, answering this research question can help inform educational policy and personal choices about learning formats.
What impact do US public school lunches have on student health?
Targeting a specific, well-defined context, the research could lead to direct changes in public health policies.
To what degree does parental involvement improve academic outcomes in secondary education in the Midwest?
This research question focuses on a specific context (secondary education in the Midwest) and has clearly defined constructs.
What are the negative effects of standardised tests on student learning within Oklahoma primary schools?
This research question has a clear focus (negative outcomes) and is narrowed into a very specific context.
Need a helping hand?
Examples: Healthcare
Shifting to a different field, let’s look at some examples of research questions within the healthcare space.
What are the most effective treatments for chronic back pain amongst UK senior males?
Specific and solution-oriented, this research question focuses on clear variables and a well-defined context (senior males within the UK).
How do different healthcare policies affect patient satisfaction in public hospitals in South Africa?
This question is has clearly defined variables and is narrowly focused in terms of context.
Which factors contribute to obesity rates in urban areas within California?
This question is focused yet broad, aiming to reveal several contributing factors for targeted interventions.
Does telemedicine provide the same perceived quality of care as in-person visits for diabetes patients?
Ideal for a qualitative study, this research question explores a single construct (perceived quality of care) within a well-defined sample (diabetes patients).
Which lifestyle factors have the greatest affect on the risk of heart disease?
This research question aims to uncover modifiable factors, offering preventive health recommendations.
Examples: Computer Science
Last but certainly not least, let’s look at a few examples of research questions within the computer science world.
What are the perceived risks of cloud-based storage systems?
Highly relevant in our digital age, this research question would align well with a qualitative interview approach to better understand what users feel the key risks of cloud storage are.
Which factors affect the energy efficiency of data centres in Ohio?
With a clear focus, this research question lays a firm foundation for a quantitative study.
How do TikTok algorithms impact user behaviour amongst new graduates?
While this research question is more open-ended, it could form the basis for a qualitative investigation.
What are the perceived risk and benefits of open-source software software within the web design industry?
Practical and straightforward, the results could guide both developers and end-users in their choices.
Remember, these are just examples…
In this post, we’ve tried to provide a wide range of research question examples to help you get a feel for what research questions look like in practice. That said, it’s important to remember that these are just examples and don’t necessarily equate to good research topics . If you’re still trying to find a topic, check out our topic megalist for inspiration.

Psst... there’s more!
This post was based on one of our popular Research Bootcamps . If you're working on a research project, you'll definitely want to check this out ...
Research ideas on Political Science
Submit a Comment Cancel reply
Your email address will not be published. Required fields are marked *
Save my name, email, and website in this browser for the next time I comment.
- Print Friendly
- UR Research
- Citation Management
- NIH Public Access
More information on Research
- Apps and Mobile Resources
- Med Students Phase 1
- School of Nursing
- Faculty Resources
More information on Guides and Tutorials
- iPad Information and Support
More information on Computing and iPads
- Library Hours
- Make A Gift
- A-Z Site Index
More information on us
- Miner Library Classrooms
- Multimedia Tools and Support
- Meet with a Librarian
More information on Teaching and Learning
- History of Medicine
More information on Historical Services
- Order Articles and Books
- Find Articles
- Access Resources from Off Campus
- Get Info for my Patient
- Contact my Librarian
More information on our services
- Research and Publishing
- Guides and Tutorials
- Computing and iPads
- Teaching and Learning
- How Do I..?
Health Humanities: Primary and Secondary Sources: Research Question
- Research Question
- Primary Sources
- Secondary Sources
- Wheel of Sources
How to Develop your Research Question?
What is a research question? A research question is a question around which you center your research. It should be:
- clear : it provides enough specifics that one’s audience can easily understand its purpose without needing additional explanation.
- focused : it is narrow enough that it can be answered thoroughly in the space the writing task allows.
- concise : it is expressed in the fewest possible words.
- complex : it is not answerable with a simple “yes” or “no,” but rather requires synthesis and analysis of ideas and sources prior to the composition of an answer.
- arguable : its potential answers are open to debate rather than accepted facts.
You should ask a question about an issue that you are genuinely curious and/or passionate about.
The question you ask should be developed for the discipline you are studying. A question appropriate for Biology, for instance, is different from an appropriate one in Political Science or Sociology. If you are developing your question for a course other than the first-year composition, you may want to discuss your ideas for a research question with your professor.
Why is a Research Question Essential?
Why is a research question essential to the research process? Research questions help writers focus their research by providing a path through the research and writing process. The specificity of a well-developed research question helps writers avoid the “all-about” paper and work toward supporting a specific, arguable thesis.
Steps to developing a research question:
- Choose an interesting general topic. Most professional researchers focus on topics they are genuinely interested in studying. Writers should choose a broad topic about which they genuinely would like to know more. An example of a general topic might be “Slavery in the American South” or “Films of the 1930s.”
- Do some preliminary research on your general topic. Do a few quick searches in current periodicals and journals on your topic to see what’s already been done and to help you narrow your focus. What issues are scholars and researchers discussing, when it comes to your topic? What questions occur to you as you read these articles?
- Consider your audience. For most college papers, your audience will be academic, but always keep your audience in mind when narrowing your topic and developing your question. Would that particular audience be interested in the question you are developing?
- Start asking questions. Taking into consideration all of the above, start asking yourself open-ended “how” and “why” questions about your general topic. For example, “Why were slave narratives effective tools in working toward the abolishment of slavery?” or “How did the films of the 1930s reflect or respond to the conditions of the Great Depression?”
- Is your research question clear? With so much research available on any given topic, research questions must be as clear as possible in order to be effective in helping the writer direct his or her research.
- Is your research question focused? Research questions must be specific enough to be well covered in the space available.
- Is your research question complex? Research questions should not be answerable with a simple “yes” or “no” or by easily-found facts. They should, instead, require both research and analysis on the part of the writer. They often begin with “How” or “Why.”
- Begin your research . After you’ve come up with a question, think about the possible paths your research could take. What sources should you consult as you seek answers to your question? What research process will ensure that you find a variety of perspectives and responses to your question?
Sample Research Questions
Unclear: How should social networking sites address the harm they cause? Clear: What action should social networking sites like Instagram and Facebook take to protect users’ personal information and privacy? The unclear version of this question doesn’t specify which social networking sites or suggest what kind of harm the sites might be causing. It also assumes that this “harm” is proven and/or accepted. The clearer version specifies sites (Instagram and Facebook), the type of potential harm (privacy issues), and who may be experiencing that harm (users). A strong research question should never leave room for ambiguity or interpretation. Unfocused: What is the effect on the environment from global warming? Focused: What is the most significant effect of glacial melting on the lives of penguins in Antarctica?
The unfocused research question is so broad that it couldn’t be adequately answered in a book-length piece, let alone a standard college-level paper. The focused version narrows down to a specific effect of global warming (glacial melting), a specific place (Antarctica), and a specific animal that is affected (penguins). It also requires the writer to take a stance on which effect has the greatest impact on the affected animal. When in doubt, make a research question as narrow and focused as possible.
Too simple: How are doctors addressing diabetes in the U.S.? Appropriately Complex: What main environmental, behavioral, and genetic factors predict whether Americans will develop diabetes, and how can these commonalities be used to aid the medical community in the prevention of the disease?
The simple version of this question can be looked up online and answered in a few factual sentences; it leaves no room for analysis. The more complex version is written in two parts; it is thought-provoking and requires both significant investigation and evaluation from the writer. As a general rule of thumb, if a quick Google search can answer a research question, it’s likely not very effective.
- << Previous: Home
- Next: Primary Sources >>
- Last Updated: Apr 28, 2023 3:46 PM
- URL: https://libguides.urmc.rochester.edu/health-humanities
Memorial Sloan Kettering Library

The physical space of the MSK Library is permanently closed to visitors as of Friday, May 17, 2024. Please visit this guide for more information.
Evidence-Based Practice: Asking a Clinical Question (PICO)
- Asking a Clinical Question (PICO)
- Levels of Evidence
- Finding the Evidence
- Practice Guidelines
- Appraising the Evidence
- Integrating the Evidence
- Selected EBP Publications
What is PICO(T)
PICO(T) is a mnemonic that stands for:
PATIENT/PROBLEM/POPULATION
INTERVENTION
PICO Resources
- Asking Focused Questions Tips and strategies for asking focused clinical questions more... less... OCEBM
- Asking searchable, answerable clinical questions Fineout‐Overholt, Ellen, and Linda Johnston. "Teaching EBP: Asking searchable, answerable clinical questions." Worldviews on Evidence‐Based Nursing 2, no. 3 (2005): 157-160.
- Evidence-based practice, step by step: asking the clinical question: a key step in evidence-based practice. Stillwell, S. B., Fineout-Overholt, E., Melnyk, B. M., & Williamson, K. M. (2010). Evidence-based practice, step by step: asking the clinical question: a key step in evidence-based practice. AJN The American Journal of Nursing, 110(3), 58-61.
- The Well Built Clinical Question Clinical scenarios that walk you through how to turn them into clinical questions. more... less... WS Richardson, MC Wilson, J Nishikawa, RS Hayward. ACP Journal Club. Nov-Dec 1995;123;A12.
- Formulating Answerable Questions Practice answering clinical questions with guided scenarios and step by step instructions.
PICO for MSK Nursing Projects
Are you working on a evidence based project or N-CARE project? Use the PICO(T) format to frame your question.
- Population/ Patient Problem: Who is your patient? (Disease or Health status, age, race, sex) / What is the problem?
- Intervention: What do you plan to do for the patient? (Specific tests, therapies, medications)
- Comparison: What is the alternative to your plan? (ie. No treatment, different type of treatment, etc.)
- Outcome: What outcome do you seek? (Less symptoms, no symptoms, full health, etc.)
- Time: What is the time frame? (This element is not always included.)
Your PICO(T) question will fall under one of these types:
- Therapy/Prevention
Need Help? Get assistance from the library - Literature/PICO Search form !
This easy-to-follow tutorial from the Librarians at the Bodleian Library at Oxford University, in partnership with the Centre for Evidence Based Medicine (CEBM), walks you through an example of turning a clinical research question into PICO format and using that to create search terms.
- << Previous: N-CARE
- Next: Levels of Evidence >>
- Last Updated: Apr 2, 2024 2:01 PM
- URL: https://libguides.mskcc.org/ebp
77 interesting medical research topics for 2024
Last updated
25 November 2023
Reviewed by
Brittany Ferri, PhD, OTR/L
Short on time? Get an AI generated summary of this article instead
Medical research is the gateway to improved patient care and expanding our available treatment options. However, finding a relevant and compelling research topic can be challenging.
Use this article as a jumping-off point to select an interesting medical research topic for your next paper or clinical study.
- How to choose a medical research topic
When choosing a research topic , it’s essential to consider a couple of things. What topics interest you? What unanswered questions do you want to address?
During the decision-making and brainstorming process, here are a few helpful tips to help you pick the right medical research topic:
Focus on a particular field of study
The best medical research is specific to a particular area. Generalized studies are often too broad to produce meaningful results, so we advise picking a specific niche early in the process.
Maybe a certain topic interests you, or your industry knowledge reveals areas of need.
Look into commonly researched topics
Once you’ve chosen your research field, do some preliminary research. What have other academics done in their papers and projects?
From this list, you can focus on specific topics that interest you without accidentally creating a copycat project. This groundwork will also help you uncover any literature gaps—those may be beneficial areas for research.
Get curious and ask questions
Now you can get curious. Ask questions that start with why, how, or what. These questions are the starting point of your project design and will act as your guiding light throughout the process.
For example:
What impact does pollution have on children’s lung function in inner-city neighborhoods?
Why is pollution-based asthma on the rise?
How can we address pollution-induced asthma in young children?
- 77 medical research topics worth exploring in 2023
Need some research inspiration for your upcoming paper or clinical study? We’ve compiled a list of 77 topical and in-demand medical research ideas. Let’s take a look.
- Exciting new medical research topics
If you want to study cutting-edge topics, here are some exciting options:
COVID-19 and long COVID symptoms
Since 2020, COVID-19 has been a hot-button topic in medicine, along with the long-term symptoms in those with a history of COVID-19.
Examples of COVID-19-related research topics worth exploring include:
The long-term impact of COVID-19 on cardiac and respiratory health
COVID-19 vaccination rates
The evolution of COVID-19 symptoms over time
New variants and strains of the COVID-19 virus
Changes in social behavior and public health regulations amid COVID-19
Vaccinations
Finding ways to cure or reduce the disease burden of chronic infectious diseases is a crucial research area. Vaccination is a powerful option and a great topic to research.
Examples of vaccination-related research topics include:
mRNA vaccines for viral infections
Biomaterial vaccination capabilities
Vaccination rates based on location, ethnicity, or age
Public opinion about vaccination safety
Artificial tissues fabrication
With the need for donor organs increasing, finding ways to fabricate artificial bioactive tissues (and possibly organs) is a popular research area.
Examples of artificial tissue-related research topics you can study include:
The viability of artificially printed tissues
Tissue substrate and building block material studies
The ethics and efficacy of artificial tissue creation
- Medical research topics for medical students
For many medical students, research is a big driver for entering healthcare. If you’re a medical student looking for a research topic, here are some great ideas to work from:
Sleep disorders
Poor sleep quality is a growing problem, and it can significantly impact a person’s overall health.
Examples of sleep disorder-related research topics include:
How stress affects sleep quality
The prevalence and impact of insomnia on patients with mental health conditions
Possible triggers for sleep disorder development
The impact of poor sleep quality on psychological and physical health
How melatonin supplements impact sleep quality
Alzheimer’s and dementia
Cognitive conditions like dementia and Alzheimer’s disease are on the rise worldwide. They currently have no cure. As a result, research about these topics is in high demand.
Examples of dementia-related research topics you could explore include:
The prevalence of Alzheimer’s disease in a chosen population
Early onset symptoms of dementia
Possible triggers or causes of cognitive decline with age
Treatment options for dementia-like conditions
The mental and physical burden of caregiving for patients with dementia
- Lifestyle habits and public health
Modern lifestyles have profoundly impacted the average person’s daily habits, and plenty of interesting topics explore its effects.
Examples of lifestyle and public health-related research topics include:
The nutritional intake of college students
The impact of chronic work stress on overall health
The rise of upper back and neck pain from laptop use
Prevalence and cause of repetitive strain injuries (RSI)
- Controversial medical research paper topics
Medical research is a hotbed of controversial topics, content, and areas of study.
If you want to explore a more niche (and attention-grabbing) concept, here are some controversial medical research topics worth looking into:
The benefits and risks of medical cannabis
Depending on where you live, the legalization and use of cannabis for medical conditions is controversial for the general public and healthcare providers.
Examples of medical cannabis-related research topics that might grab your attention include:
The legalization process of medical cannabis
The impact of cannabis use on developmental milestones in youth users
Cannabis and mental health diagnoses
CBD’s impact on chronic pain
Prevalence of cannabis use in young people
The impact of maternal cannabis use on fetal development
Understanding how THC impacts cognitive function
Human genetics
The Human Genome Project identified, mapped, and sequenced all human DNA genes. Its completion in 2003 opened up a world of exciting and controversial studies in human genetics.
Examples of human genetics-related research topics worth delving into include:
Medical genetics and the incidence of genetic-based health disorders
Behavioral genetics differences between identical twins
Genetic risk factors for neurodegenerative disorders
Machine learning technologies for genetic research
Sexual health studies
Human sexuality and sexual health are important (yet often stigmatized) medical topics that need new research and analysis.
As a diverse field ranging from sexual orientation studies to sexual pathophysiology, examples of sexual health-related research topics include:
The incidence of sexually transmitted infections within a chosen population
Mental health conditions within the LGBTQIA+ community
The impact of untreated sexually transmitted infections
Access to safe sex resources (condoms, dental dams, etc.) in rural areas
- Health and wellness research topics
Human wellness and health are trendy topics in modern medicine as more people are interested in finding natural ways to live healthier lifestyles.
If this field of study interests you, here are some big topics in the wellness space:
Gluten sensitivity
Gluten allergies and intolerances have risen over the past few decades. If you’re interested in exploring this topic, your options range in severity from mild gastrointestinal symptoms to full-blown anaphylaxis.
Some examples of gluten sensitivity-related research topics include:
The pathophysiology and incidence of Celiac disease
Early onset symptoms of gluten intolerance
The prevalence of gluten allergies within a set population
Gluten allergies and the incidence of other gastrointestinal health conditions
Pollution and lung health
Living in large urban cities means regular exposure to high levels of pollutants.
As more people become interested in protecting their lung health, examples of impactful lung health and pollution-related research topics include:
The extent of pollution in densely packed urban areas
The prevalence of pollution-based asthma in a set population
Lung capacity and function in young people
The benefits and risks of steroid therapy for asthma
Pollution risks based on geographical location
Plant-based diets
Plant-based diets like vegan and paleo diets are emerging trends in healthcare due to their limited supporting research.
If you’re interested in learning more about the potential benefits or risks of holistic, diet-based medicine, examples of plant-based diet research topics to explore include:
Vegan and plant-based diets as part of disease management
Potential risks and benefits of specific plant-based diets
Plant-based diets and their impact on body mass index
The effect of diet and lifestyle on chronic disease management
Health supplements
Supplements are a multi-billion dollar industry. Many health-conscious people take supplements, including vitamins, minerals, herbal medicine, and more.
Examples of health supplement-related research topics worth investigating include:
Omega-3 fish oil safety and efficacy for cardiac patients
The benefits and risks of regular vitamin D supplementation
Health supplementation regulation and product quality
The impact of social influencer marketing on consumer supplement practices
Analyzing added ingredients in protein powders
- Healthcare research topics
Working within the healthcare industry means you have insider knowledge and opportunity. Maybe you’d like to research the overall system, administration, and inherent biases that disrupt access to quality care.
While these topics are essential to explore, it is important to note that these studies usually require approval and oversight from an Institutional Review Board (IRB). This ensures the study is ethical and does not harm any subjects.
For this reason, the IRB sets protocols that require additional planning, so consider this when mapping out your study’s timeline.
Here are some examples of trending healthcare research areas worth pursuing:
The pros and cons of electronic health records
The rise of electronic healthcare charting and records has forever changed how medical professionals and patients interact with their health data.
Examples of electronic health record-related research topics include:
The number of medication errors reported during a software switch
Nurse sentiment analysis of electronic charting practices
Ethical and legal studies into encrypting and storing personal health data
Inequities within healthcare access
Many barriers inhibit people from accessing the quality medical care they need. These issues result in health disparities and injustices.
Examples of research topics about health inequities include:
The impact of social determinants of health in a set population
Early and late-stage cancer stage diagnosis in urban vs. rural populations
Affordability of life-saving medications
Health insurance limitations and their impact on overall health
Diagnostic and treatment rates across ethnicities
People who belong to an ethnic minority are more likely to experience barriers and restrictions when trying to receive quality medical care. This is due to systemic healthcare racism and bias.
As a result, diagnostic and treatment rates in minority populations are a hot-button field of research. Examples of ethnicity-based research topics include:
Cancer biopsy rates in BIPOC women
The prevalence of diabetes in Indigenous communities
Access inequalities in women’s health preventative screenings
The prevalence of undiagnosed hypertension in Black populations
- Pharmaceutical research topics
Large pharmaceutical companies are incredibly interested in investing in research to learn more about potential cures and treatments for diseases.
If you’re interested in building a career in pharmaceutical research, here are a few examples of in-demand research topics:
Cancer treatment options
Clinical research is in high demand as pharmaceutical companies explore novel cancer treatment options outside of chemotherapy and radiation.
Examples of cancer treatment-related research topics include:
Stem cell therapy for cancer
Oncogenic gene dysregulation and its impact on disease
Cancer-causing viral agents and their risks
Treatment efficacy based on early vs. late-stage cancer diagnosis
Cancer vaccines and targeted therapies
Immunotherapy for cancer
Pain medication alternatives
Historically, opioid medications were the primary treatment for short- and long-term pain. But, with the opioid epidemic getting worse, the need for alternative pain medications has never been more urgent.
Examples of pain medication-related research topics include:
Opioid withdrawal symptoms and risks
Early signs of pain medication misuse
Anti-inflammatory medications for pain control
- Identify trends in your medical research with Dovetail
Are you interested in contributing life-changing research? Today’s medical research is part of the future of clinical patient care.
As your go-to resource for speedy and accurate data analysis , we are proud to partner with healthcare researchers to innovate and improve the future of healthcare.
Should you be using a customer insights hub?
Do you want to discover previous research faster?
Do you share your research findings with others?
Do you analyze research data?
Start for free today, add your research, and get to key insights faster
Editor’s picks
Last updated: 18 April 2023
Last updated: 27 February 2023
Last updated: 6 February 2023
Last updated: 5 February 2023
Last updated: 16 April 2023
Last updated: 9 March 2023
Last updated: 30 April 2024
Last updated: 12 December 2023
Last updated: 11 March 2024
Last updated: 4 July 2024
Last updated: 6 March 2024
Last updated: 5 March 2024
Last updated: 13 May 2024
Latest articles
Related topics, .css-je19u9{-webkit-align-items:flex-end;-webkit-box-align:flex-end;-ms-flex-align:flex-end;align-items:flex-end;display:-webkit-box;display:-webkit-flex;display:-ms-flexbox;display:flex;-webkit-flex-direction:row;-ms-flex-direction:row;flex-direction:row;-webkit-box-flex-wrap:wrap;-webkit-flex-wrap:wrap;-ms-flex-wrap:wrap;flex-wrap:wrap;-webkit-box-pack:center;-ms-flex-pack:center;-webkit-justify-content:center;justify-content:center;row-gap:0;text-align:center;max-width:671px;}@media (max-width: 1079px){.css-je19u9{max-width:400px;}.css-je19u9>span{white-space:pre;}}@media (max-width: 799px){.css-je19u9{max-width:400px;}.css-je19u9>span{white-space:pre;}} decide what to .css-1kiodld{max-height:56px;display:-webkit-box;display:-webkit-flex;display:-ms-flexbox;display:flex;-webkit-align-items:center;-webkit-box-align:center;-ms-flex-align:center;align-items:center;}@media (max-width: 1079px){.css-1kiodld{display:none;}} build next, decide what to build next, log in or sign up.
Get started for free
University of Tasmania, Australia
Systematic reviews for health: 1. formulate the research question.
- Handbooks / Guidelines for Systematic Reviews
- Standards for Reporting
- Registering a Protocol
- Tools for Systematic Review
- Online Tutorials & Courses
- Books and Articles about Systematic Reviews
- Finding Systematic Reviews
- Critical Appraisal
- Library Help
- Bibliographic Databases
- Grey Literature
- Handsearching
- Citation Searching
- 1. Formulate the Research Question
- 2. Identify the Key Concepts
- 3. Develop Search Terms - Free-Text
- 4. Develop Search Terms - Controlled Vocabulary
- 5. Search Fields
- 6. Phrase Searching, Wildcards and Proximity Operators
- 7. Boolean Operators
- 8. Search Limits
- 9. Pilot Search Strategy & Monitor Its Development
- 10. Final Search Strategy
- 11. Adapt Search Syntax
- Documenting Search Strategies
- Handling Results & Storing Papers

Step 1. Formulate the Research Question
A systematic review is based on a pre-defined specific research question ( Cochrane Handbook, 1.1 ). The first step in a systematic review is to determine its focus - you should clearly frame the question(s) the review seeks to answer ( Cochrane Handbook, 2.1 ). It may take you a while to develop a good review question - it is an important step in your review. Well-formulated questions will guide many aspects of the review process, including determining eligibility criteria, searching for studies, collecting data from included studies, and presenting findings ( Cochrane Handbook, 2.1 ).
The research question should be clear and focused - not too vague, too specific or too broad.
You may like to consider some of the techniques mentioned below to help you with this process. They can be useful but are not necessary for a good search strategy.
PICO - to search for quantitative review questions
| P | I | C | O |
|---|---|---|---|
| if appropriate | |||
| Most important characteristics of patient (e.g. age, disease/condition, gender) | Main intervention (e.g. drug treatment, diagnostic/screening test) | Main alternative (e.g. placebo, standard therapy, no treatment, gold standard) | What you are trying to accomplish, measure, improve, affect (e.g. reduced mortality or morbidity, improved memory) |
Richardson, WS, Wilson, MC, Nishikawa, J & Hayward, RS 1995, 'The well-built clinical question: A key to evidence-based decisions', ACP Journal Club , vol. 123, no. 3, pp. A12-A12 .
We do not have access to this article at UTAS.
A variant of PICO is PICOS . S stands for Study designs . It establishes which study designs are appropriate for answering the question, e.g. randomised controlled trial (RCT). There is also PICO C (C for context) and PICO T (T for timeframe).
You may find this document on PICO / PIO / PEO useful:
- Framing a PICO / PIO / PEO question Developed by Teesside University
SPIDER - to search for qualitative and mixed methods research studies
| S | PI | D | E | R |
|---|---|---|---|---|
| Sample | Phenomenon of Interest | Design | Evaluation | Research type |
Cooke, A, Smith, D & Booth, A 2012, 'Beyond pico the spider tool for qualitative evidence synthesis', Qualitative Health Research , vol. 22, no. 10, pp. 1435-1443.
This article is only accessible for UTAS staff and students.
SPICE - to search for qualitative evidence
| S | P | I | C | E |
|---|---|---|---|---|
| Setting (where?) | Perspecitve (for whom?) | Intervention (what?) | Comparison (compared with what?) | Evaluation (with what result?) |
Cleyle, S & Booth, A 2006, 'Clear and present questions: Formulating questions for evidence based practice', Library hi tech , vol. 24, no. 3, pp. 355-368.
ECLIPSE - to search for health policy/management information
| E | C | L | I | P | Se |
|---|---|---|---|---|---|
| Expectation (improvement or information or innovation) | Client group (at whom the service is aimed) | Location (where is the service located?) | Impact (outcomes) | Professionals (who is involved in providing/improving the service) | Service (for which service are you looking for information) |
Wildridge, V & Bell, L 2002, 'How clip became eclipse: A mnemonic to assist in searching for health policy/management information', Health Information & Libraries Journal , vol. 19, no. 2, pp. 113-115.
There are many more techniques available. See the below guide from the CQUniversity Library for an extensive list:
- Question frameworks overview from Framing your research question guide, developed by CQUniversity Library
This is the specific research question used in the example:
"Is animal-assisted therapy more effective than music therapy in managing aggressive behaviour in elderly people with dementia?"
Within this question are the four PICO concepts :
| P | elderly patients with dementia |
|---|---|
| I | animal-assisted therapy |
| C | music therapy |
| O | aggressive behaviour |
S - Study design
This is a therapy question. The best study design to answer a therapy question is a randomised controlled trial (RCT). You may decide to only include studies in the systematic review that were using a RCT, see Step 8 .
See source of example
Need More Help? Book a consultation with a Learning and Research Librarian or contact [email protected] .
- << Previous: Building Search Strategies
- Next: 2. Identify the Key Concepts >>
- Last Updated: Aug 6, 2024 10:44 AM
- URL: https://utas.libguides.com/SystematicReviews

The Graduate Health & Life Sciences Research Library at Georgetown University Medical Center
Evidence-based medicine resource guide.
- Defining EBM
Types of Clinical Questions
Formulating a well built clinical question, type of clinical question and study design.
- Point-of-Care Tools
- Databases for Clinical Research
- Drug Information
- Books & eBooks
- EBM Journals
- EBM Organizations & Centers
- Health Sciences: The Research Process
Resources and Types of Clinical Question
Background questions are best answered by medical textbooks, point-of-care tools such as DynaMed Plus and Essential Evidence Plus, and narrative reviews.
Foreground questions are best answered by consulting medical databases such as MEDLINE (via PubMed or Ovid), Embase, Cochrane Database of Systematic Reviews and ACP Journal Club.
DML's Clinical Quick Reference page is a great place to locate EBM resources. Each resource has been labeled background and/or foreground, for you!
Clinical questions may be categorized as either background or foreground. Why is this important?
Determining the type of question will help you to select the best resource to consult for your answer.
Background questions ask for general knowledge about an illness, disease, condition, process or thing. These types of questions typically ask who, what, where, when, how & why about things like a disorder, test, or treatment, etc.
For example
- How overweight is a woman to be considered slightly obese?
- What are the clinical manifestations of menopause?
- What causes migraines?
Foreground questions ask for specific knowledge to inform clinical decisions. These questions typically concern a specific patient or particular population. Foreground questions tend to be more specific and complex compared to background questions. Quite often, foreground questions investigate comparisons, such as two drugs, two treatments, two diagnostic tests, etc. Foreground questions may be further categorized into one of 4 major types: treatment/therapy, diagnosis, prognosis, or etiology/harm.
- Is Crixivan effective when compared with placebo in slowing the rate of functional impairment in a 45 year old male patient with Lou Gehrig's Disease?
- In pediatric patients with Allergic Rhinitis, are Intranasal steroids more effective than antihistamines in the management of Allergic Rhinitis symptoms?
According to the Centre for Evidence Based Medicine (CEBM) , "one of the fundamental skills required for practising EBM is the asking of well-built clinical questions. To benefit patients and clinicians, such questions need to be both directly relevant to patients' problems and phrased in ways that direct your search to relevant and precise answers."
A well-built clinical foreground question should have all four components. The PICO model is a helpful tool that assists you in organizing and focusing your foreground question into a searchable query. Dividing into the PICO elements helps identify search terms/concepts to use in your search of the literature.
P = Patient, Problem, Population (How would you describe a group of patients similar to you? What are the most important characteristics of the patient?)
I = Intervention, Prognostic Factor, Exposure (What main intervention are you considering? What do you want to do with this patient?)
C = Comparison (What are you hoping to compare with the intervention: another treatment, drug, placebo, a different diagnostic test, etc.? It's important to include this element and to be as specific as possible.)
O = Outcome (What are you trying to accomplish, measure, improve or affect? Outcomes may be disease-oriented or patient-oriented.)

Small collection. Updated monthly.
Two additional important elements of the well-built clinical question to consider are the type of foreground question and the type of study (methodology) . This information can be helpful in focusing the question and determining the most appropriate type of evidence.
Foreground questions can be further divided into questions that relate to therapy, diagnosis, prognosis, etiology/harm
- Therapy: Questions of treatment in order to achieve some outcome. May include drugs, surgical intervention, change in diet, counseling, etc.
- Diagnosis: Questions of identification of a disorder in a patient presenting with specific symptoms.
- Prognosis: Questions of progression of a disease or likelihood of a disease occurring.
- Etiology/Harm: Questions of negative impact from an intervention or other exposure.

Meta-analysis: A statistical technique that summarizes the results of several studies in a single weighted estimate, in which more weight is given to results of studies with more events and sometimes to studies of higher quality.
Systematic Review: a review in which specified and appropriate methods have been used to identify, appraise, and summarize studies addressing a defined question. (It can, but need not, involve meta-analysis). In Clinical Evidence, the term systematic review refers to a systematic review of RCTs unless specified otherwise.
Randomized Controlled Trial: a trial in which participants are randomly assigned to two or more groups: at least one (the experimental group) receiving an intervention that is being tested and another (the comparison or control group) receiving an alternative treatment or placebo. This design allows assessment of the relative effects of interventions.
Controlled Clinical Trial: a trial in which participants are assigned to two or more different treatment groups. In Clinical Evidence, we use the term to refer to controlled trials in which treatment is assigned by a method other than random allocation. When the method of allocation is by random selection, the study is referred to as a randomized controlled trial (RCT). Non-randomized controlled trials are more likely to suffer from bias than RCTs.
Cohort Study: a non-experimental study design that follows a group of people (a cohort), and then looks at how events differ among people within the group. A study that examines a cohort, which differs in respect to exposure to some suspected risk factor (e.g. smoking), is useful for trying to ascertain whether exposure is likely to cause specified events (e.g. lung cancer). Prospective cohort studies (which track participants forward in time) are more reliable than retrospective cohort studies.
Case control study: a study design that examines a group of people who have experienced an event (usually an adverse event) and a group of people who have not experienced the same event, and looks at how exposure to suspect (usually noxious) agents differed between the two groups. This type of study design is most useful for trying to ascertain the cause of rare events, such as rare cancers.
Case Series: analysis of series of people with the disease (there is no comparison group in case series).
- << Previous: Defining EBM
- Next: Point-of-Care Tools >>
- Last Updated: Aug 12, 2024 10:45 AM
- URL: https://guides.dml.georgetown.edu/ebm
The Responsible Use of Electronic Resources policy governs the use of resources provided on these guides. © Dahlgren Memorial Library, Georgetown University Medical Center. Unless otherwise stated, these guides may be used for educational or academic purposes as long as proper attribution is given. Please seek permission for any modifications, adaptations, or for commercial purposes. Email [email protected] to request permission. Proper attribution includes: Written by or adapted from, Dahlgren Memorial Library, URL.
- Dean's Message
- Mission Statement
- Diversity At VUSN
- Our History
- Faculty Fellows & Honors
- Accreditation
- Privacy Policy
- Academic Programs
- Master of Science in Nursing
- Master of Nursing
- Doctor of Nursing Practice
- PhD in Nursing Science
- Post-Master's Certificate
- Postdoctoral Program
- Special (Non-Degree) Students
- Admissions Information
- Admissions Requirements
- MSN Admissions
- MN Admissions
- DNP Admissions
- PhD Admissions
- Post-Master's Certificates
- Postdoctoral Admissions
- Center for Research Development and Scholarship (CRDS)
- Signature Areas
- CRDS Behavorial Labs
- Research Resources
- Faculty Scholarship Program
- Research Faculty
- Preparing For Practice
- Faculty Practice Network
- Credentialing Process
- Faculty Practice History
- Vanderbilt Nurse-Midwifery Faculty Practice
- What is Advanced Practice Nursing?
- Preceptor Resources
- The Vanderbilt Advantage
- Making A Difference
- Informatics
- Global Health
- Organizations
- Veterans/Military
Examples of Research Questions
Phd in nursing science program, examples of broad clinical research questions include:.
- Does the administration of pain medication at time of surgical incision reduce the need for pain medication twenty-four hours after surgery?
- What maternal factors are associated with obesity in toddlers?
- What elements of a peer support intervention prevent suicide in high school females?
- What is the most accurate and comprehensive way to determine men’s experience of physical assault?
- Is yoga as effective as traditional physical therapy in reducing lymphedema in patients who have had head and neck cancer treatment?
- In the third stage of labor, what is the effect of cord cutting within the first three minutes on placenta separation?
- Do teenagers with Type 1 diabetes who receive phone tweet reminders maintain lower blood sugars than those who do not?
- Do the elderly diagnosed with dementia experience pain?
- How can siblings’ risk of depression be predicted after the death of a child?
- How can cachexia be prevented in cancer patients receiving aggressive protocols involving radiation and chemotherapy?
Examples of some general health services research questions are:
- Does the organization of renal transplant nurse coordinators’ responsibilities influence live donor rates?
- What activities of nurse managers are associated with nurse turnover? 30 day readmission rates?
- What effect does the Nurse Faculty Loan program have on the nurse researcher workforce? What effect would a 20% decrease in funds have?
- How do psychiatric hospital unit designs influence the incidence of patients’ aggression?
- What are Native American patient preferences regarding the timing, location and costs for weight management counseling and how will meeting these preferences influence participation?
- What predicts registered nurse retention in the US Army?
- How, if at all, are the timing and location of suicide prevention appointments linked to veterans‘ suicide rates?
- What predicts the sustainability of quality improvement programs in operating rooms?
- Do integrated computerized nursing records across points of care improve patient outcomes?
- How many nurse practitioners will the US need in 2020?
PhD Resources
- REQUEST INFO
- CAREER SERVICES
- PRIVACY POLICY
- VANDERBILT UNIVERSITY
- VANDERBILT UNIVERSITY MEDICAL CENTER
DISTINCTIONS

- Off-Campus Access
- Find Resources
- Research Support
- Reserve Space
- --> X
- YouTube
- HSHSL Updates RSS Feed
- Connective Issues Newsletter

601 West Lombard Street Baltimore MD 21201-1512 Reference: 410-706-7996 Circulation: 410-706-7928
Evidence-Based Practice Tutorial: Asking Clinical Questions
- Asking Clinical Questions
- Acquiring the Evidence
- Appraising the Evidence
- Applying the Results
- Assessing the Outcome
- Practicing Evidence-Based Medicine
- Additional Resources
Using PICO to Create a Well-Built Clinical Question
It is important to be purposeful about creating a well-built clinical question so that you will be able to find the most relevant results possible. A well-built question will address four important items: P atient or Problem, I ntervention, C omparison, and O utcome. To help you remember this, you can use the mnemonic PICO. When you are designing your clinical question, here are some topics to take into consideration.
P= Patient or Problem:
How would you describe a group of patients similar to yours? What are the most important characteristics of the patient? This may include the primary problem, disease, or co-existing conditions. Sometimes the gender, age or race of a patient might be relevant to the diagnosis or treatment of a disease.
I= Intervention:
Which main intervention, prognostic factor, or exposure are you considering? What do you want to do for the patient? Prescribe a drug? Order a test? Order surgery? Or what factor may influence the prognosis of the patient - age, co-existing problems, or previous exposure?
C= Comparison:
What is the main alternative to compare with the intervention? Are you trying to decide between two drugs, a drug and no medication or placebo, or two diagnostic tests? Your clinical question may not always have a specific comparison.
O= Outcome:
What can you hope to accomplish, measure, improve or affect? What are you trying to do for the patient? Relieve or eliminate the symptoms? Reduce the number of adverse events? Improve function or test scores?
PICO Example
Using our clinical scenario, we will use PICO to develop a clinical question.
Question: In patients with type 2 diabetes and obesity, does bariatric surgery promote the management of diabetes and weight loss as compared to standard medical care?
Categories of Clinical Questions
Different types of clinical questions have certain kinds of studies that best answer them. The chart below lists the categories of clinical questions and the studies you should look for to answer them.

In our clinical scenario, we are want to determine whether or not bariatric surgery will benefit the patient, so this is a therapy question. As such, we will want to find randomized control trials to answer our question. If we found numerous RCTs on this topic, we might want to consider searching for a systematic review that synthesizes the results of these trials.
Hierarchy of Evidence
The strength of the evidence produced varies among the different types of studies. Filtered sources like systematic reviews and meta-analyses provide stronger evidence because they evaluate and compare a number of original studies. The image below demonstrates the relative strengths of the study types - generally, the higher up on the pyramid you go, the more rigorous the study design and the lesser likelihood of bias or systematic error.
Types of Studies
Types of studies we are going to cover all fall under one of two categories - primary sources or secondary sources. Primary sources are those that report original research and secondary sources are those that compile and evaluate original studies.
Primary Sources
Randomized Controlled Trials are studies in which subjects are randomly assigned to two or more groups; one group receives a particular treatment while the other receives an alternative treatment (or placebo). Patients and investigators are "blinded", that is, they do not know which patient has received which treatment. This is done in order to reduce bias.
Cohort Studies are cause-and-effect observational studies in which two or more populations are compared, often over time. These studies are not randomized.
Case Control Studies study a population of patients with a particular condition and compare it with a population that does not have the condition. It looks the exposures that those with the condition might have had that those in the other group did not.
Cross-Sectional Studies look at diseases and other factors at a particular point in time, instead of longitudinally. These are studies are descriptive only, not relational or causal. A particular type of cross-sectional study, called a Prospective, Blind Comparison to a Gold Standard, is a controlled trial that allows a research to compare a new test to the "gold standard" test to determine whether or not the new test will be useful.
Case Studies are usually single patient cases.
Secondary Sources
Systematic Reviews are studies in which the authors ask a specific clinical question, perform a comprehensive literature search, eliminate poorly done studies, and attempt to make practice recommendations based on the well-done studies.
Meta-Analyses are systematic reviews that combine the results of select studies into a single statistical analysis of the results.
Practice Guidelines are systematically developed statements used to assist practitioners and patients in making healthcare decisions.
- << Previous: Steps in the Evidence-Based Practice Process
- Next: Acquiring the Evidence >>
- Last Updated: Nov 22, 2022 3:19 PM
- URL: https://guides.hshsl.umaryland.edu/ebptutorial
Projects Maintained by the HSHSL
- Alumni & Associates
- Congenital Heart Disease
- Corporate Services
- Embracing mHealth
- Faith Community Nursing
- Project SHARE Curriculum
- Citizen Science: Gearing Up For Discovery (edX)
- UMB Digital Archive
- Weise Gallery
University of Maryland Schools
- School of Dentistry
- Graduate School
- School of Law
- School of Medicine
- School of Nursing
- School of Pharmacy
- School of Social Work
Connective Issues E-Newsletter
Read Our Latest Issue
Join our subscribers!
Contact Us | Hours | Directions | Privacy Policy | Disclaimers | Supporting the Library | Suggestion Box | HSHSL Building Work Order | Web Accessibility | Diversity Statement
601 W. Lombard Street | Baltimore, Maryland 21201-1512 | 410-706-7995
Explore the Best Medical and Health Research Topics Ideas
Table of contents
- 1 How to Choose Medical Research Paper Topics
- 2 New Medical Research Paper Topics
- 3 Medical Research Topics for College Students
- 4 Controversial Medical Topics for Research Paper
- 5 Health Research Topics
- 6 Medicine Research Topics
- 7 Healthcare Research Topics
- 8 Public Health Research Topics
- 9 Mental Health Research Paper Topics
- 10 Anatomy Research Topics
- 11 Biomedical Research Topics
- 12 Bioethics Research Topics
- 13 Cancer Research Topics
- 14 Clinical Research Topics
- 15 Critical Care Research Topics
- 16 Pediatric Research Topics
- 17 Dental Research Topics Ideas
- 18 Dermatology Research Topics
- 19 Primary Care Research Topics
- 20 Pharmaceutical Research Topics
- 21 Medical Anthropology Research Topics
- 22 Paramedic Research Paper Topics
- 23 Surgery Research Topics
- 24 Radiology Research Paper Topics
- 25 Anatomy and Physiology Research Paper Topics
- 26 Healthcare Management Research Paper Topics
- 27 Medical Ethics Research Paper Topics
- 28 Environmental Health and Pollution Research Paper Topics
- 29 Conclusion
Writing an original and compelling research paper is a daunting task in such a complex and broad field as medicine. Each student decides where his interests lie, from investigating public care concerns to cancer treatment studies. We aim to help students find new angles to study and focus on relevant topics. With our resources, you can write an engaging and rigorous paper.
How to Choose Medical Research Paper Topics
Choosing good research paper topics is often more challenging than the writing process itself. You need to select a captivating subject matter that will grab the reader’s attention, showcase your knowledge of a specific field, help you progress in your studies, and perhaps even inspire future research.
To accomplish that, you need to start with brainstorming, followed by thorough research. Here are some great tips to follow:
- Pick an interesting topic – The key is to pick something that you find interesting, and yet make sure it’s not too general or too narrow. It should allow you to delve deep into the subject matter and show that you’re a professional who is ready to take on a challenge when it comes to your chosen field of medicine.
- Narrow down your focus – Once you have a list of potential topics, sift through recent medical research papers to get up-to-date with the latest trends, developments, and issues in medicine and healthcare. Check out textbooks, news articles, and other relevant sources for more information related to your potential topics. If a particular condition or disease interests you (perhaps something that drew you to a career in medicine), there’s your cue for narrowing down your topic.
- Pinpoint the “why,” “how,” and “what” – Whether you are looking into nutrition research paper topics , controversial medical topics, nursing research topics, or anything in-between, ask yourself why each of them is important. How could they contribute to the available medical studies, if any? What new information could they bring to improve the future of medicine? Asking these questions will help you pick the right medical research paper topic that suits you and helps you move forward and reach your aspirations.
To help you on that quest, we’ve compiled a list of topics that you could use or that might inspire you to come up with something unique. Let’s dive in.
New Medical Research Paper Topics
Are you interested in the newest and most interesting developments in medicine? We put hours of effort into identifying the current trends in health research so we could provide you with these examples of topics. Whether you hire a research paper writing service for students or write a paper by yourself, you need an appealing topic to focus on.
- Epidemics versus pandemics
- Child health care
- Medical humanitarian missions in the developing world
- Effectiveness of mobile health clinics in rural Africa
- Homeopathic medicines – the placebo effect
- Comparative study of the efficacy of homeopathic treatments and conventional medicine in managing chronic pain
- Virus infections – causes and treatment
- Trends in COVID-19 vaccine uptake
- Advancements in the treatment of influenza
- Is medical research on animals ethical
- Vaccination – dangers versus benefits
- Artificial tissues and organs
- Rare genetic diseases
- Brain injuries
- Long-Term Effects of COVID-19
- Social behavior shifts due to COVID-19
Medical Research Topics for College Students
You don’t know where to start with your medical research paper? There are so many things you could write about that the greatest challenge is to narrow them down. This is why we decided to help.
- Antibiotics treatments
- Efficacy of mRNA vaccines against viral diseases
- Viability and function of 3D printed tissues
- Chronic diseases
- Palliative treatment
- Battling Alzheimer’s disease
- How modern lifestyle affects public health
- Professional diseases
- Sleep disorders
- Changes in physical and mental health due to aging
- Eating disorders
- Terminal diseases
Controversial Medical Topics for Research Paper
In healthcare, new discoveries can change people’s lives in the blink of an eye. This is also the reason why there are so many controversial topics in medicine, which involve anything from religion to ethics or social responsibility. Read on to discover our top controversial research topics.
- Ethical debates on artificial tissue engineering
- Public opinions on vaccination safety
- Implementing food standards
- Telehealth’s Role in Chronic Illness Management
- Gluten allergy
- Assisted suicide for terminal patients
- Testing vaccines on animals – ethical concerns
- Moral responsibilities regarding cloning
- Marijuana legalization for medical purposes
- Abortion – medical approaches
- Vegan diets – benefits and dangers
- Increased life expectancy: a burden on the healthcare system?
- Circumcision effects
Health Research Topics
Students conducting health research struggle with finding good ideas related to their medical interests. If you want to write interesting college papers, you can select a good topic for our list.
- Impact of location, ethnicity, or age on vaccination rates
- Uses of biomaterials in vaccination technology
- Deafness: communication disorders
- Household air pollution
- Diabetes – a public danger
- Coronaviruses
- Oral health assessment
- Tobacco and alcohol control
- Diseases caused by lack of physical exercise
- How urban pollution affects respiratory diseases
- Healthy diets

Medicine Research Topics
Regardless of the requirements in your research assignment, you can write about something that is both engaging and useful in your future career. Choose a topic from below.
- Causes for the increasing cancer cases
- Insulin resistance
- How terrorism affects mental health
- AIDS/HIV – latest developments
- Treating pregnant women versus non-pregnant women
- Latest innovations in medical instruments
- Genetic engineering
- Successful treatment of mental diseases
- Is autism a disease
- Natural coma versus artificial coma
- Treatments for sleep disorders and their effectiveness
- Role of melatonin supplements in sleep quality

Healthcare Research Topics
Healthcare research includes political and social aspects, besides medical. For college students who want to explore how medicine is affected by society’s values or principles, we provide examples of topics for papers. Select yours from the list below.
- Government investment in healthcare services in the EU versus the USA
- Inequalities in healthcare assistance and services
- Electronic health records systems – pros and cons
- Can asylums treat mental issues
- Health care for prison inmates
- Equipment for improving the treatment of AIDS
- Correlation between economic development and health care services across countries
- Impact of smoking on organs
- Heart attacks – causes and effects
- Breast cancer – recent developments
- Materials used in artificial tissue and their impacts
Public Health Research Topics
For current examples of public health topics, browse our list. We provide only original, researchable examples for which you can easily find supporting data and evidence.
- Public versus private hospitals
- Health Disparities in Diabetes Management Across Different Socioeconomic Groups
- Health care professionals – management principles
- Surgery failures – who is responsible
- What legal responsibilities has the hospital administration
- Patient service quality in public versus private hospitals
- What benefits do national health care systems have
- Estimated costs of cancer treatments
- Public health in developing countries
- Banning tobacco ads – importance for public health
- Government solutions to the anti-vaccine’s movement
- How the COVID-19 pandemic has changed public health regulations
Mental Health Research Paper Topics
Mental health is one of the most complex areas of medicine, where things are never as clear as with other medical issues. This increases the research potential of the field with plenty of topics left for debate.
- Mental Health Impact of Social Media on American Teenagers
- Causes of anxiety disorders
- Bulimia versus anorexia
- Childhood trauma
- Mental health public policies
- Impact of Lifestyle Factors on the Progression of Dementia in the Elderly Population
- Postpartum Depression
- Posttraumatic Stress Disorder
- Seasonal Affective Disorder
- Schizophrenia
- Stress and its effects on sleep quality
- Insomnia and its relation to mental health disorders
Anatomy Research Topics
Anatomy covers everything about the human body and how it works. If you find that intriguing and want to pay for medical research paper, start by selecting a topic.
- Causes and treatments of virus infections
- Chemotherapy: how it affects the body
- Thyroid glands – functions in the body
- Human endocrine system
- Preventative Measures and Treatments for Common Liver Diseases
- Heart diseases
- How does the human muscular system develop
- Lymphatic system – importance
- Investigating genetic diseases
- Digestive system
- Role of the Spleen in the Human Immune System and Related Disorders

Biomedical Research Topics
Biology and medicine often work together. For the newest changes in the biomedical field, check our topics.
- Comparative Efficacy of Alternative Medicine Practices in Chronic Pain Management
- Alzheimer’s disease – paths for treatment
- Vaccines and drug development in the treatment of Ebola
- Antibiotic resistance
- Biological effects caused by aging
- Air pollution effects on health
- Infectious disease past versus present
- Regenerative medicine
- Biomedical diagnostics
- Biomedical technology
- Advanced biomaterials for vaccine delivery
Bioethics Research Topics
A controversial area of medicine, bioethics is where you get the chance to add personal input to a research topic and come up with new insights. You could consider these subjects.
- Organ donation
- Alternative or complementary medicine
- Assisted suicide or the right to die
- Artificial insemination or surrogacy
- Chemical and biological warfare
- Contraception
- Environmental bioethics
- In Vitro Fertilization
- Ethical considerations in medical research on animals
Cancer Research Topics
Are you writing a paper related to cancer causes, diagnosis, treatment or effects? Look below for a hot topic that it’s easy to research and important for medical advance.
- The ability of immune system cells to fight cancer
- Computational oncology
- Metastasis affected by drug resistance
- Stem cells – applications for cancer treatment
- Tumor microenvironment
- Obesity and age in cancer occurrence
- Early cancer detection – benefits
- Artificial intelligence predicting cancer
- Hematologic malignancies
- Pathogen-related cancers
- Impact of COVID-19 on cancer treatment studies
Clinical Research Topics
Learn more about clinical medicine by conducting more in-depth research. We prepared for you a list of relevant issues to touch upon.
- Ethical concerns regarding research on human subjects
- Subject recruitment
- Budget preparation
- Human subject protection
- Clinical trials – financial support
- Clinical practices for health professionals
- Using vulnerable populations in clinical research
- Quality assurance in clinical research
- Academic clinical trials versus clinical trials units
- Data collection and management
- Evolution of clinical symptoms in COVID-19 patients
Critical Care Research Topics
Critical care is a key area in medical studies. Explore these topics in your research paper to gain more valuable knowledge in this field. You can also get in contact with nursing research paper writers .
- Obesity and asthma – clinical manifestations
- Chronic obstructive pulmonary disease
- Rhythm analysis for cardiac arrest
- Traumatic brain injury – fluid resuscitation
- Hydrocortisone for multiple trauma patients
- Care and nutrition for critically ill adults
- Diagnosis of hypersensitivity pneumonitis
- Coma and sedation scales
- Artificial airways suctioning
- Arterial puncture and arterial line
- Long-term cardiac and respiratory effects of COVID-19
Pediatric Research Topics
Any topic that refers to health care for children, pregnant women, mothers, and adolescents goes under pediatric care.
- Early Intervention Methods for Children Diagnosed with Autism Spectrum Disorder
- Preventive healthcare strategies for children
- Impact of early childhood nutrition on long-term health
- Attention deficit hyperactivity disorder (ADHD)
- Congenital heart disease in newborns
- Adolescent medicine
- Neonatal medicine
- Rare diseases in children and teenagers
- Obesity and weight fluctuations
- Behavioral sleep problems in children
- Children with anemia
- Child healthcare enhancements and innovations

Dental Research Topics Ideas
Choose a topic on oral health or dental care from this list of the most interesting topics in the field.
- How smoking affects oral health
- Children’s risk for dental caries
- Causes of Dental Anxiety and Effective Interventions for Reducing Fear in Patients
- Types of dental materials – new advances
- Bad breath bacteria
- How diabetes affects oral health
- Oral cancer
- Dental pain – types, causes
- Dental implants
- Oral health-related quality of life
- Advancements in treatments for virus infections
Dermatology Research Topics
Find the best research topic for your dermatology paper among our examples.
- Atopic dermatitis
- Contact dermatitis
- Epidemiology behind uncommon skin disorders
- Cutaneous aging
- Risk factors of melanoma skin cancer
- Acne versus rosacea
- Genetic testing for skin conditions
- Effects of cosmetic agents on skin health
- Improving skin barrier with pharmaceutical agents
- Skin manifestations of autoimmune disorders
- Study of virus effects on skin health
Primary Care Research Topics
Write a primary care paper that can demonstrate your research skills and interest in powerful scientific findings.
- Primary care for vulnerable/uninsured populations
- Interpersonal continuity in care treatment
- How primary care contributes to health systems
- Primary care delivery models
- Developments in family medicine
- Occupational/environmental health
- Pharmacotherapy approaches
- Formal allergy testing
- Oral contraception side effects
- Dietary or behavioral interventions for obesity management
Pharmaceutical Research Topics
Pharma students who need paper topics can use one from our list. We include all things related to pharmacy life.
- Drugs that can treat cancer
- Drug excretion
- Elimination rate constant
- Inflammatory stress drug treatment
- Aspirin poising
- Ibuprofen – dangers versus benefits
- Toxicodynamics
- Opioid use disorder
- Pharmacotherapy for schizophrenia
- Ketamine in depression treatment
Medical Anthropology Research Topics
Medical anthropology unites different areas of human knowledge. Find powerful ideas for a paper below.
- Cultural contexts regarding reproductive health
- Women sexuality
- Anthropological aspects of health care
- Contributions of social sciences to public health
- Euthanasia and medical ethics across cultures
- Health-related behavior in adults across cultures
- Transcultural nursing
- Forensic psychiatry
- Symptoms of Celiac Disease – a disease with no symptoms
- Nursing ethics
Paramedic Research Paper Topics
Topics for paramedic research must be based on evidence, data, statistics, or practical experience. Just like ours.
- Trends and statistics in EMS
- Disaster medicine
- Mass casualties
- Pandemics and epidemics
- Infection control
- Basic versus advanced life support
- Scene safety in EMS
- Shock management
- Motor vehicle accidents
- Challenges in medical humanitarian missions during pandemics
Surgery Research Topics
Discover all the intricacies of surgeries that save lives by writing about our topics.
- Medical malpractice and legal issues
- Methicillin-resistant Staphylococcus aureus
- Early Detection and Management Strategies for Sepsis in Hospital Settings
- Pain management
- Perioperative nursing
- Wound management
- Colorectal cancer surgery
- Breast cancer surgery
- Minimally invasive surgeries
- Vascular disease
- Changes in surgical practices during pandemics
Radiology Research Paper Topics
Find a radiology topic related to your academic interests to write a successful paper.
- Using MRI to diagnose hepatic focal lesions
- Multidetector computer tomography
- Ultrasound elastography in breast cancer
- Assessing traumatic spinal cord injuries with MRI diffusion tensor imaging
- Sonographic imaging to detect male infertility
- Role of tomography in diagnosing cancer
- Brain tumor surgery with magnetic resonance imaging
- Bacterial meningitis imaging
- Advanced imaging techniques for virus infection detection
Anatomy and Physiology Research Paper Topics
Any ideas for a medical research paper? We have included the most important topics for an anatomy and physiology paper.
- What role has the endocrine system
- Staphylococcus aureus
- Environmental factors that affect development of human muscular system
- What role has the lymphatic system
- An investigation of genetic diseases
- Explaining the aging process
- The digestive tract
- Effects of stress on cells and muscles
- Evolution of the human nervous system
- What role has the cardiovascular system
- Impact of viruses on respiratory health in urban settings
Healthcare Management Research Paper Topics
There are numerous topics you could write about when it comes to healthcare management. There’s a wide range of options to pick, from infrastructure, staff, and financial management to HR and patient management. Here are some of the top healthcare management research paper options.
Medical Ethics Research Paper Topics
Medical ethics is a field that opens the door to numerous compelling topics for research papers. Here are some of the most appealing ones you could tackle.
- Clinical research on humans
- Vaccines and immunization
- Religious beliefs in healthcare
- Euthanasia and physician-assisted suicide
- Ethical issues across cultures
- Amniocentesis or prenatal birth defect testing
- Medical malpractice and going back to work
- Racial and ethnic preferences and perceptions in organ donations
- Racial and ethnic disparities in healthcare
- Ethical concerns of AI in healthcare
- Debates on animal ethics in medical research
- Free unlimited checks
- All common file formats
- Accurate results
- Intuitive interface
Environmental Health and Pollution Research Paper Topics
- Environmental Pollutants and Respiratory Health in Urban Areas of the USA
- How environmental changes affect human health
- Long-Term Impact of PM2.5 Exposure on Lung, Heart, and Brain Function
- Health Risks of Air Pollution Across Different Life Stages
- Hospital Admissions and Air Quality in the USA
- Risk Reduction Strategies for Indoor Air Pollution from Gas Stoves
- Impact of Air Pollution on Cognitive Development and Socioeconomic Achievements
- Long-Term Health Effects of Early Childhood Exposure to Air Pollution
- Impact of Traffic Noise on Cardiovascular Health
If you need further assistance with your medical research paper, PapersOwl is here for you. Our expert writers can provide you with top-notch research and help you write an impressive paper. Contact us anytime, pick your writer, tell them more about your topic, and get a unique, plagiarism-free research paper with impeccable grammar and formatting.
Readers also enjoyed

WHY WAIT? PLACE AN ORDER RIGHT NOW!
Just fill out the form, press the button, and have no worries!
We use cookies to give you the best experience possible. By continuing we’ll assume you board with our cookie policy.
An official website of the United States government
The .gov means it’s official. Federal government websites often end in .gov or .mil. Before sharing sensitive information, make sure you’re on a federal government site.
The site is secure. The https:// ensures that you are connecting to the official website and that any information you provide is encrypted and transmitted securely.
- Publications
- Account settings
Preview improvements coming to the PMC website in October 2024. Learn More or Try it out now .
- Advanced Search
- Journal List
- v.14(9); 2022 Sep

Research Question, Objectives, and Endpoints in Clinical and Oncological Research: A Comprehensive Review
Addanki purna singh.
1 Physiology, Saint James School of Medicine, The Quarter, AIA
Praveen R Shahapur
2 Microbiology, Bijapur Lingayat District Educational Association (BLDE, Deemed to be University) Shri B.M. Patil Medical College, Vijayapur, IND
Sabitha Vadakedath
3 Biochemistry, Prathima Institute of Medical Sciences, Karimnagar, IND
Vallab Ganesh Bharadwaj
4 Microbiology, Trichy Sri Ramasamy Memorial (SRM) Medical College Hospital & Research Centre, Tiruchirapalli, IND
Dr Pranay Kumar
5 Anatomy, Prathima Institute of Medical Sciences, Karimnagar , IND
Venkata BharatKumar Pinnelli
6 Biochemistry, Vydehi Institute of Medical Science & Research Center, Bangalore, IND
Vikram Godishala
7 Biotechnology, Ganapathy Degree College, Parkal, IND
Venkataramana Kandi
8 Clinical Microbiology, Prathima Institute of Medical Sciences, Karimnagar, IND
Clinical research is a systematic process of conducting research work to find solutions for human health-related problems. It is applied to understand the disease process and assist in the diagnosis, treatment, and prevention. Currently, we are experiencing global unrest caused by the coronavirus disease (COVID-19) pandemic. The novel severe acute respiratory syndrome coronavirus (SARS-CoV-2) has been responsible for the deaths of more than 50 million people worldwide. Also, it has resulted in severe morbidity among the affected population. The cause of such a huge amount of influence on human health by the pandemic was the unavailability of drugs and therapeutic interventions to treat and manage the disease. Cancer is a disease condition wherein the normal cell function is deranged, and the cells multiply in an uncontrolled manner. Based on recent reports by the World Health Organization (WHO), cancer is the second leading cause of death globally. Moreover, the rates of cancers have shown an increasing trend in the past decade. Therefore, it is essential to improve the understanding concerning clinical research to address the health concerns of humans. In this review, we comprehensively discuss critical aspects of clinical research that include the research question, research objectives, patient-reported outcome measures (PROMs), intention-to-treat and per-protocol analysis, and endpoints in clinical and oncological research.
Introduction and background
Successful clinical research can be conducted by well-trained researchers. Other essential factors of clinical research include framing a research question and relevant objectives, documenting, and recording research outcomes, and outcome measures, sample size, and research methodology including the type of randomization, among others [ 1 , 2 ].
Clinicians/physicians and surgeons are increasingly dependent on the clinical research results for improved management of patients. Therefore, researchers need to work upon a relevant research question/hypothesis and specific objectives that may potentially deliver results that can be translated into practice in the form of evidence-based medicine [ 3 ].
Essential elements that facilitate a researcher to frame a research question are in-depth knowledge of the subject and identifying possible gaps. Moreover, the feasible, interesting, novel, ethical, relevant (FINER) approach and the population of interest/target group, intervention, comparison group, outcome of interest, and time of study (PICOT) approach were previously suggested for researchers to be able to frame appropriate research questions [ 4 ].
Moreover, the research objectives should be framed by the researcher before the initiation of the study: a specific, measurable, achievable, realistic, and time-defined (SMART) approach is utilized to devise the objectives based on the research question. It is preferable to have a single primary objective whereas the secondary objectives can be multiple and may be dependent on the amount of data collected. The objectives must be simple and specific and must reflect the research question [ 5 , 6 ].
Given the evolution and the increasing requirement for emergency care, clinical researchers are advised to adopt a population, exposure, comparator, outcome (PECO)/ population, exposure, comparator, outcome (PICO) approach to construct the study objectives and carry out quantitative research. In contrast, qualitative research which is carried out to understand, explore, and examine requires the researcher to understand what and why the research is undertaken along with the roles of the researcher, research process/steps, and participants [ 7 , 8 ].
Since clinical research is envisaged in finding a solution to a health problem, choosing the appropriate endpoint requires special focus. The endpoints are the specific measures of the outcomes of an intervention and therefore they must be chosen judiciously [ 9 ]. The endpoints in a clinical trial can be single or multiple in numbers. The primary endpoints assess the major research question, and the secondary endpoints may assess alternative research questions. Moreover, there are other endpoints like surrogate endpoints, intermediate clinical endpoints, clinical outcomes, clinical outcome assessments, clinician-reported outcomes, observer-reported outcomes, patient-reported outcomes, and performance outcomes [ 10 ] (Figure (Figure1 1 ).

This figure has been created by the authors
The clinical trial endpoints are essentially the indicators of the power of the interventions either to cure or control the disease progression. Due to the cost and the tedious nature of the clinical trials, it is suggested that multiple-arm trials that include more than one primary endpoint be used [ 11 ]. Integration of the primary endpoints with the patient prioritized endpoints was recently suggested especially among cardiovascular disease patients [ 12 ]. Cancer research is an increasingly evolving area because of the unavailability of therapeutic interventions for some malignancies like breast and lung cancer, among others [ 13 , 14 ]. Moreover, the drugs available for treating cancers are plagued by adverse reactions. However, since most cancer clinical trials apply overall survival as the preferred and gold standard clinical endpoint, it is difficult for the trial operators to sustain the costs associated with the long lengths of the study that follow-up patients for years to assess the clinical outcomes after interventions. In this study, we comprehensively review the essential elements of clinical research that include the research question, hypothesis, and clinical and oncological endpoints.
Research question
A research question can alternatively be called the aim of the researcher. It describes the problem that the researcher intends to solve vis-à-vis finding an answer to a question. A research question is the first step toward any kind of research process that includes both qualitative as well as quantitative research. Since the research question predicts the core of any project, it must be carefully framed. The essential elements to consider while determining a research question are feasibility, preciseness, and relevance to the real world.
A person interested in a broad subject area must first complete extensive reading of the available literature. This enables the researcher to find out the strengths, loopholes, deficiencies, and missing links that can form the basis for framing a research question. The problem to which a solution needs to be found and the potential causes/reasons for the problems help a researcher frame the research question.
The research questions should be framed in such a way that the researcher will find several possibilities as solutions to the research question rather than a simple yes or no. Among the various factors that determine the power of a research question, the most essential ones include the ability of research questions to find complex answers, focussed, and the specific nature of the question. The research questions must be answerable, debatable, and researchable [ 15 , 16 ]. Research questions differ from the type of research method selected by the researcher as shown in Figure Figure2 2 .

FINER: Feasible, interesting, novel, ethical, relevant; PICOT: Population of interest/target group, intervention, comparison group, outcome of interest, time of study; PECO: Population, exposure, comparator, outcome; PICO: Population, intervention, comparator, outcome; SMART: Specific, measurable, achievable, realistic, and time defined; COVID-19: Coronavirus disease-19
Hypothesis testing
A hypothesis is an assumption by the researcher that the answers drawn with reference to the research question are either true or false. The researcher performs hypothesis testing by using appropriate statistical methods on the data collected from the research.
The hypothesis is an assumption/observation of the researcher regarding the outcome of potential research that is being conducted. There are two types of hypotheses, the null hypothesis (H0), wherein, the researcher assumes that there is no relation/causality/effect. The alternate hypothesis (HA) is when the researcher believes/assumes that there is a relationship/effect [ 17 ]. Basically, there are two types of errors while testing a hypothesis. Type I error (α) (false positive) is when the researcher rejects the null hypothesis although it is true. Type II error (β) (false negative) is when the researcher accepts the null hypothesis although it is false.
The errors in hypothesis testing occur because of bias among many other reasons in the study. Many studies set the power of the studies to essentially rule out errors. Researchers consider 5% chance (α=0.05; range: 0.01-0.10) of error in case of a type I error and up to 20% chance (β =0.20; range: 0.05-0.20) in case of type II errors [ 18 ]. The characteristics of a good hypothesis are simple and specific. Moreover, it must be decided by the researcher prior to initiating the study and while writing the study proposal/protocol [ 18 ].
Hypothesis testing means sample testing, wherein the information gathered after sample testing is inferred after applying statistical methods. A hypothesis may be generated in several ways that include observations, anatomical features, and other physiological facts observed by physicians [ 19 ]. Hypothesis testing also can be performed by using appropriate statistical methods. The testing of the hypothesis is done to prove the null hypothesis or otherwise use the sample data.
As a researcher, one must assume a null hypothesis, or believe that the alternate hypothesis holds good in the sample selected. After the collection of data, analysis, and interpretation, the researcher either accepts or rejects the hypothesis. Therefore, it must be noted that while a study is initiated, there is only a 50% chance of either the null hypothesis or the alternative hypothesis coming true [ 20 ].
The step-by-step process of hypothesis testing starts with an assumption, criteria for interpretation of results, analysis, and conclusion. The level of significance (95% free of type I error and 80% free of type II error) also is decided initially to ensure that the study results are replicated by the other researchers [ 21 ].
Objectives in clinical research
The most significant objective in clinical research is to find out whether the intervention attempted was successful in curing the disease/medical condition. It is important to understand the fact that research planning greatly influences the research results, and no statistical method can improve the results but a well-designed and conducted clinical research [ 22 ].
The primary objective of clinical research studies includes improvement in patient management. Most clinical research studies are aimed at discovering a new/novel drug to treat a medical condition that presently has no specific treatment, or the available drugs are not particularly effective in curing the disease.
The objectives are formed to address the five 'W's, namely who (children, women, etc.); what (the medical condition/disease/infection); why (causes of the medical condition/disease/infection); when (conditions responsible for the medical condition/disease/infection); and where (geographical aspects of the medical condition/disease/infection) as shown in Figure Figure3 3 .

Clinical research can be of several types including primary research and secondary research. Also, the research can be observational (no intervention) and experimental/interventional. Clearly demarcated/framed research objectives are essential to improve the clarity, specificity, and focus of the clinical trial [ 23 ].
Patient-reported outcome measures (PROMs)
While conducting clinical research the investigators collect trial data through clinical observations, laboratory monitoring, and caregiver feedback. There are some aspects of the data like the patient-reported outcomes (PROs) that can be reported by the subject/patient him/herself either in the form of a questionnaire or through interviews. Such data collected from the patients in their words is termed PROMs. The PROMs include a global impression of the trial, the functional status and well-being, symptoms, health-related quality of life (HRQL), treatment compliance, and satisfaction.
The questionnaire used to collect the PROs is called a PRO instrument. The data collected through this instrument is used to establish the benefit-to-risk ratio of the clinical trial drug. The PRO instruments can be designed as generic (contains a wide variety of health-related aspects and therefore can be used among different patient types), disease-specific (rheumatoid arthritis, psoriasis, etc.), dimension-specific (physical activity, cognitive levels, etc.), region/site-specific, individualized, utility measures, and summary items [ 24 ]. The patient-reported experience measure (PREMs), and the patient and public involvement programs are used to collect the patient’s feedback that invariably helps in improving the quality of healthcare facilities [ 25 ].
It is important to develop PROM tools/instruments for various diseases, especially among children as noted by a recent research report. This study suggested that a suitable PROM instrument is required to measure the status of disease (wheezing/asthma/respiratory diseases) and its control among preschool children [ 26 ].
Intention to treat and per-protocol analysis
The intention to treat (ITT) analysis is used when all the study subject’s data is analyzed including all those participants who were enrolled in the study, and those who have deviated (not signed the informed consent, discontinued from the study, not taken the trial drug as suggested). The ITT studies minimize the bias and ensure both the study and the control groups are compared.
The per-protocol (PP) analysis usually includes the data from only those subjects who have remained till the study period ended, have taken the drugs as suggested by the protocol, and was available for regular follow-up. The disadvantages of PP are potential disturbances in the balance between the study groups (randomized/placebo/control), a lower number of study participants due to the exclusion of dropouts, and non-compliant subjects. Therefore, the results from the PPA studies could be biased [ 27 ].
The randomized clinical trial (RCT) studies of superiority type use ITT analyses as against the non-inferiority and equivalence studies wherein an ITT approach may favor the study hypothesis.
According to the Committee for Proprietary Medicinal Products (CPMP), and the Consolidated Standards for reporting trials (CONSORT), both the ITT and PP analyses must be assessed to effectively interpret the results of the clinical trial including the safety and efficacy [ 28 ].
Endpoints in clinical research
The endpoints in clinical research determine whether the clinical trial has been successful in finding out if an intervention/drug has proven beneficial in improving the survival and quality of life of the patient. The endpoints determine the validity of the clinical trial results. There are different types of endpoints like primary endpoints, secondary endpoints, tertiary endpoints, surrogate endpoints (laboratory measurements, physical signs), and others [ 9 ].
The clinical trial endpoints could be subjective and objective in nature. The objective endpoints are survival, disease progression/remission, and the development of disease/condition. The subjective endpoints include symptoms, quality of life, and other patient-reported outcomes [ 29 , 30 ]. The significance of endpoints in clinical trials and the importance of choosing appropriate endpoints were previously reported. This study suggested that the primary endpoints help in deriving the sample size and confirm the generalizability of the results. The secondary and surrogate endpoints could be used while conducting the clinical research without ignoring the aspects of the quality of life of the subjects [ 31 ].
Endpoints in oncology clinical trials
Cancer clinical trials assume increased significance because the drugs developed against the cancer are intended to increase the survival of the patients. Also, anti-cancer agents are associated with several side effects. Therefore, oncology trials include several endpoints, the primary being the overall survival, and the secondary endpoints include the assessment of other outcomes that indicate the quality of life (QoL), tumor-related endpoints, and others. The disadvantages of oncology clinical trials are the cost associated with the recruitment of a greater number of subjects and long-time follow-up of the patients [ 32 ].
Although primary endpoints are considered as most significant in oncological trials, a recent report stressed the importance of surrogate markers in assessing the efficacy of anti-cancer drugs [ 33 ].
The endpoints in oncology clinical trials, their applications, functions, and drawbacks are summarized in Table Table1 1 .
| Endpoint | Applications | Functions | Drawbacks |
| Overall survival | Gold standard primary oncological endpoint | Assesses time from randomization to death, clinical benefit, easily measurable, gives definite results, and eliminates researcher bias | Not effective in slowly progressing diseases, requires quite a high patient number, influenced by cross-over, subsequent therapies, and non-cancer deaths |
| Duration of clinical benefit | Primary endpoint | Assesses time from randomization to disease progression or death in patients who achieve a complete response, partial response, or stable disease for 24 weeks or longer | Needs disease-specific validation |
| Complete response | A primary endpoint that can also be used as a surrogate endpoint | Assesses time from randomization to survival advantage associated with improved overall survival and prolonged event-free survival in specific treatment studies | Needs disease-specific validation |
| Time to treatment failure | A primary endpoint when used in conjunction with secondary endpoints | Assesses the time from the initiation of chemotherapy treatment/intervention to its early discontinuation | The inclusion of older patients may affect the results |
| Health-related quality of life | Preferred as a secondary clinical endpoint | Assesses patient’s quality of life with respect to health status over time, can directly measure the patient’s benefit | Data may frequently be missing, and inadequate, the clinical relevance of exceedingly small changes is unknown and requires multiple analyses and validation |
| Progression-free survival | A surrogate marker for regular and accelerated approval | Assesses time from randomization until the first evidence of disease progression or death, requires a limited patient number, short follow-up period, objective and quantitative evaluation, cost-effective, and not influenced by crossovers or subsequent therapies | It cannot be statistically validated as a surrogate marker for survival, is not definitely measurable, is subject-dependent with a high risk of bias, definitions may differ between studies, and the time of evaluation needs to be balanced between treatment arms |
| Disease-free survival | A surrogate marker for regular and accelerated approval | Assesses time from randomization until evidence of disease recurrence, and requires limited patient number and short follow-up | It cannot be statistically validated as a surrogate marker for survival, not definitely measurable, and definitions may differ between studies |
| Objective response rate | A surrogate marker for regular and accelerated approval | Assesses how a specific treatment impacts tumor burden in a patient with a history of solid tumors, needs to be evaluated in single-arm studies, much quicker evaluation as compared with survival studies, and requires much more limited patient number | Benefits cannot be measured directly, and detailed measurement of drug activity is unavailable |
| Duration of response | Used as a surrogate marker | Assesses time from randomization to disease progression or death in patients who achieve complete or partial response | Needs disease-specific validation |
| Pathological complete response | Preferred as a surrogate marker | Assesses time from randomization to absence of residual invasive cancer upon evaluation of the resected breast tissue and regional lymph nodes | Disease-specific, especially in breast cancer |
| Disease control rate | Clinical benefit for survival | Assesses time from randomization to complete response, partial response, or stable disease | Can exaggerate the anticancer effect of the therapy |
| Clinical benefit rate | Clinical benefit for survival | Assesses time from randomization to complete response, partial response, or at least six months of stable disease | It does not necessarily measure clinical benefit |
| Milestone survival | Used as a surrogate endpoint/qualitative endpoint | Assesses time from randomization to survival probability at a given time point | Requires further validation |
| Time to progression | Preferred as a surrogate and not a primary endpoint | Assesses time from randomization to first evidence of disease progression and effectiveness of targeted therapy | Can be adversely affected by patients’ disease characteristics |
| Event free survival | Preferred as a surrogate and as an alternative to the primary endpoint | Assesses time from randomization to disease progression, discontinuation of the treatment, and/or death | It needs to be validated for each unique disease/tumor type, treatment, and stage of disease |
| Time to next treatment | Preferred as a surrogate endpoint for incurable diseases | Follows up the treatment response until the initiation of next-line therapy | Requires validations for specific disease |
Survival endpoints in oncology clinical trials
The survival endpoint considers the time from randomization to death. This type of follow-up (daily), although difficult to do, will remove bias associated with the investigator’s interpretation. The survival studies require large sample sizes and cross-over therapies act as confounding factors for survival. The survival studies consider patient benefit over drug toxicity.
Apart from the overall survival, oncology clinical trials use alternative ways to assess the efficacy of the drugs by using other endpoints like progression-free survival [ 34 ]. Other endpoints suggested are biomarkers, disease-free survival, objective response rate, time to progression, complete response, partial response, minor response, time to treatment failure, time to next treatment, duration of clinical benefit, objective response rate, complete response, pathological complete response, disease control rate, clinical benefit rate, milestone survival, event-free survival, and QoL [ 35 ].
Endpoints in immunological diseases and infections
Autoimmune diseases are usually chronic conditions that arise due to the immunologic responses against the self. They are usually associated with hyper-reactivity of immune cells towards the host's own cells/tissue and can cause significant morbidity and mortality among affected people.
Autoimmune diseases are generally genetic in origin, but many such diseases are attributed to other factors such as infection, food, drugs, and other substances. Frequently occurring autoimmune diseases are rheumatoid arthritis, psoriasis, systemic lupus erythematosus (SLE), ulcerative colitis, Crohn’s disease, and multiple sclerosis, among others [ 36 ].
Clinical trials with respect to the development of drugs/medicine to treat autoimmune diseases take into consideration the therapeutic efficacy of the trial drug, no risk, and only benefit to the patients. Therefore, the selection of endpoints for clinical trials in immune diseases must consider all these factors to effectively assess the pharmacological value of the trial drugs. The endpoints for autoimmune hepatitis include remission, incomplete response, treatment failure, and drug toxicity [ 37 ].
The endpoint related to the infections includes the direct measurement of the number of microorganisms. Other endpoints include the measurement of physiological aspects impaired by the infecting microbe and the measurement of immune responses against the infectious agent. The endpoints of human immunodeficiency virus (HIV) infection includes HIV-ribonucleic acid (RNA) viral load, maintenance, improvement, and decline of the cluster of differentiation 4 (CD4)+ T lymphocyte cell counts, and others.
Composite endpoints
Clinical trial for drugs is assessed based on several endpoints that establish efficacy and allow regulatory authorities to decide on approving the drugs for human use. In most instances, the clinical trials apply primary endpoints whereas in recent times the surge in candidate drugs and the necessity for life-saving drugs had ushered in the use of alternative endpoints like the composite endpoints. The composite endpoints are instrumental in reducing the trial costs, minimizing the long follow-ups, and lower subject recruitments. Since the composite endpoints combine more than one outcome during the drug trial, it enables the investigators to understand the efficacy of the drug in a short period of time [ 38 ].
While using multiple endpoints, it is important to understand that each one is as important as the other and the statistical methods must be used to confirm the overall efficacy of the trial drug. The drawbacks of applying composite endpoints in clinical trials are the complexity of the methods, low transparency, including invalid indicators, and the possibility of misleading results and conclusions [ 39 ].
Because there is no specific recommendation as to how the composite endpoints need to be selected, evaluated, and analyzed, there exists a possibility of bias. In a recent report, an index to evaluate the bias attributable to composite outcomes (BACO) was applied and suggested. The BACO index <0, 0 to <1, and >1 indicated that the composite endpoints were inverted, underestimated, and overestimated, respectively. A BACO index of 1 indicates that the composite endpoint usage resulted in unbiased results [ 40 ]. The composite endpoint in a clinical trial means the use of multiple endpoints. For the drug trials for migraine, the composite endpoints include no pain for two hours, nausea after two hours, and photosensitivity after two hours. The clinical trials with vaccines could have more than 20 endpoints. Other trials with multiple endpoints include rheumatoid arthritis (4), acne (4), sleep disorders (6), primary biliary cirrhosis (4), and glaucoma (9).
Conclusions
The clinical research design must be carefully accomplished keeping in mind the financial and time constraints. The trial must be initiated to address a specific research question, which essentially requires the research group to carry out an extensive literature search and identify the knowledge gaps. The research hypothesis needs to be carefully designed to avoid errors. The researchers should ensure the inclusion of appropriate objectives that guarantee quality outcomes and clinical benefits. It is essential to include the PROMs along with the clinician, researcher, and observer reported outcomes to assess the benefit-to-risk ratio of the investigational drug and improve the quality of healthcare facilities, among others. The safety and efficacy of a clinical trial drug should be carefully interpreted based on the results obtained from the ITT and PP analyses. Moreover, a clinical trial should incorporate specific and relevant endpoints that ensure the efficacy or otherwise of the intervention. Cancer clinical trials are even more complex because the interventions could potentially be life-saving and therefore, the selection of endpoints becomes critical as discussed briefly in this review.
The content published in Cureus is the result of clinical experience and/or research by independent individuals or organizations. Cureus is not responsible for the scientific accuracy or reliability of data or conclusions published herein. All content published within Cureus is intended only for educational, research and reference purposes. Additionally, articles published within Cureus should not be deemed a suitable substitute for the advice of a qualified health care professional. Do not disregard or avoid professional medical advice due to content published within Cureus.
The authors have declared that no competing interests exist.
Cookies on this website
We use cookies to ensure that we give you the best experience on our website. If you click 'Accept all cookies' we'll assume that you are happy to receive all cookies and you won't see this message again. If you click 'Reject all non-essential cookies' only necessary cookies providing core functionality such as security, network management, and accessibility will be enabled. Click 'Find out more' for information on how to change your cookie settings.

Asking focused questions
One of the fundamental skills required for practising EBM is the asking of well-built clinical questions. To benefit patients and clinicians, such questions need to be both directly relevant to patients’ problems and phrased in ways that direct your search to relevant and precise answers. In practice, well-built clinical questions usually contain four elements, summarised below.
| 1 | 2 | 3 | 4 | |
|---|---|---|---|---|
| Patient or Problem | Intervention (a cause, prognostic factor, treatment, etc.) | Comparison Intervention (if necessary) | Outcomes | |
| Tips for Building | Starting with your patient, ask “How would I describe a group of patients similar to mine?” Balance precision with brevity. | Ask “Which main intervention am I considering?”Be specific. | Ask “What is the main alternative to compare with the intervention?”Again, be specific. | Ask “What can I hope to accomplish?” or “What could this exposure really affect?”Again, be specific. |
| Example | “In patients with heart failure from dilated cardiomyopathy who are in sinus rhythm …” | “… would adding anticoagulation with warfarin to standard heart failure therapy …” | “… when compared with standard therapy alone …” | “… lead to lower mortality or morbidity from thromboembolism. Is this enough to be worth the increased risk of bleeding?” |
Educational Prescription (PDF)
Benefits of asking focused questions
One of the benefits of careful and thoughtful question-forming is that the search for evidence is easier. The well-formed question makes it relatively straightforward to elicit and combine the appropriate terms needed to represent your need for information in the query language of whichever searching service is available to you.
Once you have formed the question using the PICO structure, you can think about what type of question it is you are asking, and therefore what type of research would provide the best answer.
- Open access
- Published: 07 August 2024
Towards inclusive healthcare: evaluating knowledge, confidence and awareness of LGBTQ + health among Internal Medicine Trainees in London
- Andrew Crowe 1 ,
- Patrick Hogan 2 ,
- Christoper Morrison 3 ,
- Catherine Meads 1 &
- Daniel Bailey 4
BMC Medical Education volume 24 , Article number: 851 ( 2024 ) Cite this article
129 Accesses
Metrics details
Patients from the lesbian, gay, bisexual, transgender, queer plus (LGBTQ +) community face various health inequalities and report poor healthcare experiences. Little is known about how knowledgeable and confident UK doctors are around LGBTQ + health, and previous research demonstrates that UK medical schools rarely deliver teaching in this area. This research evaluated the level of knowledge, awareness and confidence of LGBTQ + health among Internal Medical Trainees (IMTs) in London.
London IMTs were invited to complete an online questionnaire evaluating knowledge, awareness and confidence in LGBTQ + health. Stratified analysis of results by demographics was performed.
Three hundred and fifteen surveys were analysed from 796 eligible trainees (40%). Confidence in caring for LGBTQ + patients was variable. Confidence in discussing gender identity was lower than for sexual orientation. Knowledge of health issues affecting LGBTQ + patients varied. Most participants had never received training on LGBTQ + health at undergraduate ( n = 201, 64%) or postgraduate level ( n = 252, 80%), but the majority of participants felt that training would be useful ( n = 233, 74%). Stratified analysis revealed that IMTs who received previous LGBTQ + teaching at undergraduate or postgraduate level were considerably more confident discussing sexual orientation with patients, compared to those who received no previous teaching.
Conclusions
There is a clear need for education on LGBTQ + health, given the varied levels of knowledge and confidence identified. A significant majority of IMTs in London have never received teaching on LGBTQ + health, although there exists a strong desire for this. LGBTQ + health topics should be integrated into undergraduate and postgraduate training and examinations for IMTs. This would support IMTs in delivering high quality and inclusive care for all patients, particularly those of sexual orientation and gender identity minorities. There are relatively few published studies exploring competency in LGBTQ + health among doctors, and this is the first among UK Internal Medicine Trainees.
Peer Review reports
In recent times, the spotlight on healthcare disparities faced by marginalised communities has grown stronger, and the voices of these communities have grown louder [ 1 ]. LGBTQ + communities are one such marginalised group, composed of people who are Lesbian, Gay, Bisexual, Transgender, or Queer. The “plus” denotes people who are part of the community, but for whom LGBTQ + neither accurately captures, nor reflects their identity. They frequently report negative encounters in the healthcare setting and experience unique health inequalities in areas such as physical health, sexual health, and mental health [ 2 ].
Cancer burden is greater in the LGBTQ + communities, with higher rates of anal cancer among men who have sex with men [ 3 ] and higher rates of cervical intraepithelial neoplasia among women who have sex exclusively with women [ 4 ]. In addition, lesbian and bisexual women in the UK have higher rates of asthma and obesity compared to heterosexual women [ 5 , 6 ]. Transgender individuals are significantly more likely to be living with chronic medical and psychiatric conditions (including dementia) and have suicide rates at least 5 times higher than their cisgender peers [ 7 , 8 ]. LGBTQ + patients of nearly all age groups are more likely to avoid seeing their GP, contributing to late diagnosis and poor outcomes [ 9 ].
One potential factor contributing to these health inequalities is the ‘Minority stress theory’, which suggests that LGBTQ + people experience chronic stress from both “distal” sources (e.g. discrimination, victimisation, bullying, stigmatisation, violence, and social injustice), and “proximal” sources (e.g. internalised homophobia and perceived prejudice) [ 10 ]. This chronic stress response may lead to increased risk of various physical health conditions, mental health conditions including suicidality, and increases the likelihood of engaging in high-risk and harmful behaviours [ 11 , 12 ]. Among transgender people, negative physical health outcomes were actually more common in those with past experiences of significant harassment or violence, compared to those without [ 13 ]. Through education, clinicians can become aware of the Minority stress theory, and actions that can potentially contribute to this either overtly (e.g. through expression of prejudicial opinions), or inadvertently (e.g. by using heteronormative and cisnormative language, by providing services that do not appear inclusive).
Another factor potentially contributing to health inequalities is engagement with healthcare. LGBTQ + people may have concerns about disclosing sexual orientation/gender identity to healthcare providers, based on previous experience of discrimination, the perception or fear of it, and concerns that services will neither understand, nor support their needs [ 14 ]. As a result, they may not engage with screening programmes or seek help for concerning symptoms, leading to missed opportunities for cancer detection, or primary/secondary prevention of disease [ 2 ]. For example, women who have sex exclusively with women are less likely to attend for cervical cancer screening [ 4 ], and as previously stated, they have higher rates of cervical intraepithelial neoplasia (a precursor to cervical cancer). Transgender people report higher rates of negative experiences in healthcare, and are more likely to avoid seeking care than their cisgender counterparts [ 15 ]. Studies demonstrate they have increased rates of chronic medical conditions, and poor mental health, particularly suicidal ideation [ 7 , 8 ].
LGBTQ + communities have contrasting experiences of health and healthcare compared to the general population. Current and future clinicians should be cognisant of these differences and their role in addressing them.
The area of LGBTQ + health remains understudied and under-researched; it is not widely covered in curricula of UK medical schools. For many medical schools, there is little or no exposure to LGBTQ + teaching during the undergraduate programmes [ 16 , 17 ]. Medical students feel unprepared for encounters with LGBTQ + patients, which could translate into poor quality of care [ 18 , 19 ]. Inclusion and cultural competence are increasingly recognised to be important in healthcare, and this knowledge gap may contribute to suboptimal care, and worsen health disparities experienced by LGBTQ + individuals. With increasing numbers of people identifying as LGBTQ + , doctors must be competent to provide care to patients from these communities [ 20 ].
There is a dearth of literature describing LGBTQ + health in medical education and little is known about the knowledge and confidence of UK clinicians around these issues. The vast majority of published literature in this area focuses on the undergraduate setting and explores how confident and knowledgeable medical students are, or evaluates the amount of LGBTQ + teaching in undergraduate curricula [ 16 , 17 ]. In relation to medical graduates (i.e. qualified doctors), there are very few published studies and only one other in the British setting which focuses solely on Oncologists [ 21 ], making this the first study of its kind among IMTs in the United Kingdom.
The core aim of this study was to evaluate the levels of knowledge, confidence, and awareness that Internal Medicine Trainees (IMTs) in London have around the health needs of patients from the LGBTQ + community. Our objectives included: assessing how confident IMTs feel when caring for patients from this community, examining how knowledgeable IMTs are in LGBTQ + health, determining how much prior teaching IMTs have received on LGBTQ + health and how useful they feel specialist teaching would be, and investigating the demographics of participants in a stratified analysis.
In this article, we use the terms MSM (men who have sex with men) and WSW (women who have sex with women). These terms describe sexual activities without assuming identities like gay, lesbian, or bisexual, recognising that not everyone who engages in same-sex activities identifies with these labels.
Through this research, we identify areas for improvement, and consequently, provide the evidence needed to design targeted interventions and implement curricular changes that could equip future doctors with the skills to confidently care for this marginalised and vulnerable population group.
Study design
We designed and conducted an observational cross-sectional study with mixed quantitative/qualitative methods. Our core research question was: What is the level of awareness, confidence and knowledge in LGBTQ + health among IMTs in London?
We included all 796 IMTs (years 1–3) currently training in a London Deanery. IMTs are qualified doctors who have completed Foundation Training and have chosen to train in Internal Medicine (they are at least 2 years after graduation). After completion of Internal Medicine training, the majority will enter specialist medical training (in Cardiology, Gastroenterology, Neurology, etc.).
We identified IMTs for inclusion as they form a large and accessible cohort of doctors, thus providing a suitable sample size. In addition, they interact with patients on a daily basis and are likely to encounter members of LGBTQ + community in a professional context. We focused on London as it has the largest proportion of LGBTQ + residents in the United Kingdom [ 22 ].
The online questionnaire was designed using Jisc software, a program for designing and distributing online surveys. The surveys were emailed to participants four times over a 2-month period via the London School of Medicine. These questionnaires were self-administered by participants, and participation was voluntary. Consent was compulsory in order to complete the questionnaire and participants were asked to read the Participation Information Leaflet and tick the consent box if in agreement. The participants were not asked for personally identifiable information such as name, date of birth or address, but were asked to provide some demographic details. There was a "prefer not to say" option for each demographic question.
There were 33 questions, in 5 sections. The majority were closed questions with true/false or yes/no answers. Other question formats included multiple choice questions, Likert scale questions and free text boxes for comments or feedback.
The first section assessed demographics, the second section explored levels of awareness and confidence in caring for LGBTQ + patients, the third section assessed prior teaching on LGBTQ + health received by participants, the fourth section examined knowledge of LGBTQ + health and the fifth section asked for comments and feedback. The correct answers to each question in the knowledge section, along with an explanation and reference to the literature, were provided upon completion of the survey to promote learning for all participants.
These questions were designed to focus on scenarios encountered by IMTs, thus making the survey directly relevant to their practice. A pilot questionnaire was completed by a small group of IMTs, and questions were refined based on their feedback. We concentrated on general internal medicine, an area often neglected in LGBTQ + health research, rather than other areas such as sexual health. We designed 3 separate question stems to individually test knowledge on gay male health, gay female health, and trans health. Due to limited survey space, we were unable to include as many identities as we wished (bisexual, non binary etc.).
Data analysis
Every survey answered was used in data analysis, which was done with SPSS software and descriptive analysis of the data. Data was presented in graphs and charts made using Microsoft Excel. In certain demographic questions and other parts of the results where fewer than five respondents answered, the results are reported in text and tables as < 5 in order to promote confidentiality and reduce risk of participant identification.
Ethical Approval was granted by the School Research Ethics Panel (SREP) of the Health, Education, Medicine and Social Care (HEMS) faculty of Anglia Ruskin University.
There were 315 responses (40% of the total eligible population). All surveys were fully completed. Most respondents were aged 26-30yrs. ( n = 198, 62.9%), and slightly more participants were female, with 160 female participants (50.8%), 140 male participants (44.4%), and the rest indicating 'prefer not to say' ( n = 15, 4.8%). 23.1% of participants identified as LGBTQ + , with 6.7% ticking "prefer not to say" for sexual orientation, and 5.7% for gender identity. For demographics—See Table 1 .
Confidence/awareness
When asked about confidence in discussing issues of sexual orientation and gender identity with patients (See Table 2 ), responses varied, but confidence levels around gender identity were lower than sexual orientation. Just over half of participants (54.3%) felt confident asking a patient about sexual orientation, while 27.6% did not feel confident, and 18.1% felt somewhat confident. Regarding gender identity, 45.1% of participants felt confident asking patients about gender identity, 33.3% did not feel confident, and 21.6% felt somewhat confident. Less than half (46.0%) felt confident using terms related to gender identity (pronouns, transgender, non-binary etc.), while 30.8% did not feel confident, and 23.2% felt somewhat confident. When asked if participants had ever treated patients who identified as LGBTQ + , 289 respondents (91.7%) replied Yes, 12 participants (3.8%) replied No, and 14 (4.4%) were not sure.
Most participants reported having no prior exposure to training on LGBTQ + health, (See Table 3 ), a slightly greater proportion of participants received LGBTQ + training during their undergraduate training than during postgraduate training (36.1% during undergraduate vs 20.0% during postgraduate). A large proportion felt that LGBTQ + teaching was useful: 233 participants (73.9%) felt it was "very useful", 79 participants (25.1%) felt it was "somewhat useful", and 3 participants (0.9%) felt it was "not useful". Participants were keen for teaching on various areas of LGBTQ + health but particularly on the topics of general medicine in LGBTQ + patients (85.4%) and transgender healthcare (66.7%).
Distribution of knowledge scores was varied (See Table 4 and Fig. 1 ). Below are some pertinent results from the knowledge section:
When asked about rates of asthma and average BMI in lesbian women, most answers were incorrect (90.8% incorrect and 72.1% incorrect respectively)
64.8% of respondents correctly identified that lesbian women in the UK do not have higher rates of cardiovascular disease compared to the general populations, and 60.6% correctly recognised that nulliparity is a risk factor for breast cancer in lesbian women (as for all nulliparous women)
72.1% correctly identified that men who have sex with men (MSM) are more likely to develop anal cancer than heterosexual men. However, over one third (34.0%) incorrectly believed they are more likely to develop colon cancer, compared to heterosexual men.
65.0% of respondents correctly answered than older gay men are twice as likely to be living alone compared to older heterosexual men.
67.9% correctly answered that older LGBTQ + individuals are less likely to attend their GP than non-LGBTQ + individuals.
A minority of respondents (41.0%) correctly answered that rates of Subjective Cognitive Decline (SCD) are higher among LGBTQ + individuals.
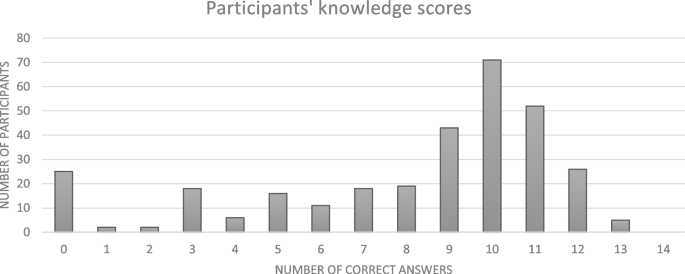
Distribution of knowledge scores
Stratified analysis
Stratified analysis (See Table 5 ) revealed that the participants who received previous LGBTQ + teaching at undergraduate or postgraduate level were considerably more confident discussing sexual orientation with patients, compared to those who received no previous teaching (statistically significant) These participants were also more confident in discussing gender identity with patients – this was statistically significant for participants who received teaching at undergraduate level, but not for those who received teaching at postgraduate level. Males felt slightly more confident discussing sexual orientation and gender identity with patients compared to females (not statistically significant). IMTs with prior teaching were more likely to feel that knowing a patient’s sexual orientation or gender identity is important when caring for them, compared to those who with no prior training (statistically significant).
Participants were invited to give feedback in two free text boxes (See Table 6 ). The first box asked if LGBTQ + teaching was worthwhile and how should it be done. The second box asked for any further comments or feedback. There were 113 responses in total. Some commonly occurring themes were;
Desire for teaching, particularly on trans healthcare and general internal medical issues in LGBTQ + patients:
“I have looked after patients who identify as LGBTQ+ and have felt ill-equipped to manage this well. Since these experiences I have tried to look up the correct terminology and language to use but it is still not an area of confidence for me, and I do not know much about the impact of this on general medicine for this patient cohort. Any teaching would be gratefully received”
Authenticity of teaching,
“Ideally teaching should be delivered in-person by people of the LGBTQ+ community so that they are not misrepresented and we can hear patient’s perspectives”
Negative experiences while working.
“Have seen some really transphobic and homophobic stuff working in the NHS and so we definitely need more education and open dialogues about LGBTQ health”
Summary of findings
Overall, this study reveals that knowledge levels around LGBTQ + health among IMTs in London are varied. They are moderately confident discussing sexual orientation with patients, but less confident discussing gender identity and its related terminology (transgender, non-binary, pronouns etc.). Most participants have never received any formal teaching on LGBTQ + health, which is consistent with the literature showing these topics are rarely covered at undergraduate or postgraduate level [ 16 , 17 ]. However, it is encouraging to see there is a strong demand for this, particularly teaching on general medicine for LGBTQ + patients and transgender healthcare.
Our results compare similarly to findings from two American studies [ 23 , 24 ]. In both studies, IMTs felt that LGBTQ + health was important, but they reported minimal prior teaching in this area and assessment of their knowledge revealed numerous deficits. Confidence levels were varied but increased after teaching.
A significant proportion of the surveyed IMTs felt under-confident discussing sexual orientation and gender identity with patients. Of note, participants were less confident discussing gender identity (and related terms such as transgender, non-binary and pronouns) than sexual orientation. One third of participants were not confident asking patients about gender identity. Stratified analysis revealed that participants who had received previous formal LGBTQ + training (at undergraduate or postgraduate level) reported higher levels of confidence in these areas compared to those who never received teaching, demonstrating the benefits of teaching, and reinforcing the need for formal education. Of note, participants who received training during university reported feeling more confident than those who did not. Although causation cannot be assumed, these findings suggest the effect of training in improving confidence may last for several years (at least 3 years in the case of this cohort of IMTs).
The proportion of surveyed participants identifying as gay (12.1%), bisexual (8.3%) or other (1.6%) was higher than the proportion in the general population. In the 2021 UK Census [ 17 ], 4.3% of London residents identified as lesbian, gay, bisexual, or other. Our figures could be explained by the younger age group of IMT participants (93.4% of participants were in the 26–35 age bracket) who are statistically more likely to identify as LGBTQ + than older age groups [ 22 ]. Additionally, these figures could reflect the potential responder bias associated with voluntary participation in surveys – for example, people identifying as gay, or bisexual are more likely to voluntarily take surveys about LGBTQ + issues. Regarding gender identity, just 0.3% of participants identified as transgender, and 1.6% as non-binary, which compares slightly differently to the general population of London residents where 0.78% identify as transgender/gender different from that assigned at birth, and 0.8% identify as non-binary [ 20 ].
Many feedback comments expressed a strong desire for LGBTQ + health teaching, with some calling for it to be mandatory during the IMT programme, and others calling for it to be integrated into the IMT curriculum. Some participants were enthusiastic for teaching to be partly delivered by members of the LGBTQ + community as they felt it was important to hear “first hand patient experiences”.
While most of the feedback was positive, it is important to acknowledge the criticisms. One participant felt that LGBTQ + training is important during IMT, but "should not be priority". Another participant called for LGBTQ + training to be "carefully balanced against other learning needs" and that it should be implemented and "governed according to clinical need only".
Strengths and limitations
Strengths of our study include the large sample size, and the fact that participants came from a diverse range of areas, both north and south London. Our research separately evaluated lesbian, gay, bisexual, and transgender health in certain questions, giving us a deeper insight into participants' understanding of these specific areas, something which is often omitted from studies in LGBTQ + health. The knowledge section presented three separate scenarios (lesbian woman, gay man, transgender man) while the confidence section examined sexual orientation and gender identity independently. Lastly, the knowledge section focused on areas of general medicine other than sexual health or mental health, which are often neglected in LGBTQ + medical education.
In terms of limitations, the generalisability of these results is restricted given the 40% response rate and the specific geographic location of this study. Participants were IMTs based in London, and consequently, one cannot draw accurate conclusions about levels of knowledge, confidence, and awareness among other groups of doctors, or doctors in other locations around the UK. Two potential explanations for the low response rate include the voluntary participation of the survey, and the fact that people may be reluctant to take surveys on “sensitive” topics (such as sexual orientation and gender identity). 23% of doctors in this survey identified as LGBTQ + , a higher proportion than expected in the general population, which could skew results. In the interests of time, and to avoid a lengthy survey, certain parameters were omitted, such as ethnicity (black, hispanic etc.), political affiliations (liberal, conservative, etc.), stage of Internal Medicine Training (IMT1, IMT2, IMT3), and attitudes towards LGBTQ + individuals.
Implications for practice
Educational programs.
Dedicated LGBTQ + educational programs are central in raising awareness among medical students and doctors about the healthcare disparities faced by LGBTQ + individuals and equipping them with the skills and knowledge to provide quality care. These programs should be designed by clinicians in conjunction with members of the LGTBQ + community. Constructivist educational activities should be prioritised, such as case-based discussions, patient interactions and role-play scenarios, as these promote active participation of learners which is key for cultural change [ 25 ]. Teaching should take place within a comfortable learning environment so that students feel safe to express opinions and critically examine various approaches to LGBTQ + healthcare, without feeling their views may be perceived as wrong or inappropriate. Educational programmes may be further enriched by embracing validated clinician self assessment tools; such as the Lesbian, Gay, Bisexual and Transgender Development of Clinical Skills assessment (LGBT-DOCSS); which allow trainees and clinicians to reflect upon their own knowledge and self-efficacy [ 26 ].
In designing education, we should avoid focusing solely on topics that are traditionally associated with LGBTQ + patients, such as sexual health. Links between the LGBTQ + community and general medical conditions such as cancer, cardiovascular disease, asthma and cognitive problems are less recognised, as evidenced by the results and feedback comments in our study. For example, the classic exam question of a gay male presenting with a new diagnosis of HIV or a sexually transmitted infection is useful to some degree, but it can lead to healthcare stereotyping [ 27 ] and fails to consider other associated medical conditions to which he is at risk. Our results show that doctors were particularly interested in teaching on transgender healthcare, especially terminology and relevant hormones. This is important to acknowledge, as we know that some clinicians feel under confident treating this group, and are not comfortable prescribing hormonal treatments [ 28 ].
Educators can be assisted in developing teaching materials by accessing support from partner organisations such as GLADD (The Association of LGBTQ + Doctors and Dentists) and the Fenway Health National LGBTQIA + Health Education Centre who can sign post to resources that providers might use, and support the building of networks that can share best practice in education [ 29 , 30 ].
Integration into examinations
Integration of LGBTQ + health topics into formal assessments, both at undergraduate and postgraduate level, is important to promote an inclusive healthcare environment. Integration can be achieved by weaving LGBTQ + health topics into examinations, for example multiple choice questions and essay questions. Integration can also be achieved by swapping heterosexual or cisgender patients for LGBTQ + patients in clinical scenarios. For example, a traditional examination of an elderly patient with Parkinson’s disease can be swapped for an elderly transgender man with Parkinson’s disease. Most of the marks are still awarded for taking an appropriate neurological history and eliciting the correct signs on physical examination, but a small number of marks go towards appropriate communication, using correct pronouns and inclusive language. This encourages normalisation of these encounters and helps build confidence for doctors caring for these communities. For IMTs, LGBTQ + health topics could be integrated into the MRCPUK (Membership of Royal College of Physicians of the United Kingdom) examinations, required for successful progression to higher medical training. These topics should feature in the written sections, as well as the clinical sections (PACES) as suggested by participants in the feedback.
Curricular change
One of the most practical ways to ensure a topic is covered effectively during training is through integration into a curriculum. Currently, LGBTQ + health is not mandatory in British medical undergraduate curricula and studies demonstrate that coverage of LGBTQ + health topics at university level is very limited and extremely dependent on the staff in each university [ 16 ]. Growing voices are calling for this to be mandated with regulation from the General Medical Council [ 31 ]. Looking to the postgraduate setting, the situation is relatively similar with no mandatory coverage of LGBTQ + health topics for Foundation level or IMT doctors. The curriculum of the UK Foundation Programme asks for doctors to develop an understanding of "equality and diversity in health" but it fails to elaborate and does not specifically mention the LGBTQ + community, or other marginalised groups [ 32 ]. Likewise, the curriculum of Internal Medicine Training in the UK vaguely asks that "training bodies comply with equality and diversity standards", but again, fails to mention anything specific to the LGBTQ + communities [ 33 ]. LGBTQ + health training needs to be integrated into curricula, both undergraduate and postgraduate, with direct reference to sexuality and gender identity minorities, and their health inequalities. Furthermore, framework resources for reforming undergraduate curricula have already been published [ 34 , 35 ], and these could be adapted for postgraduate curricula with relative ease.
Implications for research
Further studies are needed to evaluate levels of confidence and knowledge among other groups of clinicians. A comparative analysis could be done according to speciality (Psychiatrist, GP etc.), grade (registrar, consultant etc.) demographics, or geographic location, in an effort to identify factors associated with greater LGBTQ + health competency and disparities across various groups. Ideally, this would be carried out at a national level given that communities of LGBTQ + individuals are found throughout the country. Research should examine effective teaching methodologies to determine how best to integrate LGBTQ + topics into education and examinations. Longitudinal studies would help track changes in doctors' attitudes and behaviour over time, and examine competency before and after teaching interventions. In addition to targeting clinicians, future projects should explore the perspectives of LGBTQ + patients and their experiences in hospitals and clinics to determine the best ways of delivering high quality and healthcare.
The results show there is a clear need for education on LGBTQ + health, given the variable levels of knowledge and confidence identified among Internal Medicine Trainees in London. A significant majority of participants have never received teaching on LGBTQ + health, although there exists a strong desire for this, particularly teaching on general medical issues facing LGBTQ + patients and transgender healthcare. Recommendations from our research include the creation of LGBTQ + educational programs, curricular change to include LGBTQ + topics, and the integration of LGBTQ + cases in postgraduate training and examinations for IMTs. There are very few published studies exploring competency in LGBTQ + health among doctors, with only one other in the United Kingdom, but none among British Internal Medicine doctors, making this study the first of its kind.
Our research highlights the necessity to address the educational needs of Internal Medicine Trainees in London in relation to LGBTQ + health, to improve patient experiences and outcomes, and to promote an inclusive healthcare environment for all.
Availability of data and materials
The authors confirm that the data supporting the findings of this study are available within the article and its supplementary materials.
Lavizzo-Mourey, Besser, Williams, Understanding and Mitigating Health Inequities — Past, Current, and Future Directions. N Engl J Med. 2021; https://doi.org/10.1056/NEJMp2008628 .
Bailey D, Calasanti T, Crowe A, di Lorito C, Hogan P, de Vries B. Equal but different! Improving care for older LGBT+ adults. Age Ageing. 2022;51(6): https://doi.org/10.1093/ageing/afac142 . PMID: 35751872.
Quinn GP, Sanchez JA, Sutton SK, et al. Cancer and lesbian, gay, bisexual, transgender/transsexual, and queer/questioning (LGBTQ) populations. CA Cancer J Clin. 2015;65:384–400. https://doi.org/10.3322/caac.21288 .
Article Google Scholar
Saunders C, Massou E, Waller J, Meads C, Marlow LA, Usher-Smith J. Cervical screening attendance and cervical cancer risk among women who have sex with women. J Med Screen. 2021;28(3):349–56. https://doi.org/10.1177/0969141320987271 . Epub 2021 Jan 21. PMID: 33476213; PMCID: PMC8366122.
Meads C, Martin A, Grierson J, et al. Systematic review and meta-analysis of diabetes mellitus, cardiovascular and respiratory condition epidemiology in sexual minority women. BMJ Open. 2018;8(4):e020776. https://doi.org/10.1136/bmjopen-2017-020776 . PMID:29666136;PMCID:PMC5905763.
Semlyen J, Curtis TJ, Varney J. Sexual orientation identity in relation to unhealthy body mass index: individual participant data meta-analysis of 93 429 individuals from 12 UK health surveys. J Public Health (Oxf). 2020;42(1):98–106. https://doi.org/10.1093/pubmed/fdy224 .
Saunders, CL, Berner, A, Lund, J, et al. Demographic characteristics, long-term health conditions, and healthcare experiences of 6,333 trans and non-binary adults in England: nationally representative evidence from the 2021 GP Patient Survey. BMJ Open. 2023; https://doi.org/10.1136/bmjopen-2022-068099
Biggs M. Suicide by clinic-referred transgender adolescents in the United Kingdom. Arch Sex Behav. 2022;51:685–90. https://doi.org/10.1007/s10508-022-02287-7 .
National GP Patient survey 2022, NHS England, Available at: https://www.england.nhs.uk/statistics/statistical-work-areas/gp-patient-survey/
Kneale D, Thomas J, French R. Inequalities in Health and Care Among Lesbian, Gay, and Bisexual People Aged 50 and Older in the United Kingdom: a systematic review and meta-analysis of sources of individual participant data. J Gerontology B Psychol Sci Soc Sci. 2020;75(8):1758–71. https://doi.org/10.1093/geronb/gbaa071 .
Frost DM, Ilan H, et al. Minority stress theory: Application, critique, and continued relevance. Curr Opin Psychol. 2023;51:101579. https://doi.org/10.1016/j.copsyc.2023.101579 . ISSN 2352-250X.
Li H, Liu X, Zheng Q, et al. Minority stress, social support and mental health among lesbian, gay, and bisexual college students in China: a moderated mediation analysis. BMC Psychiatry. 2023;23:746. https://doi.org/10.1186/s12888-023-05202-z .
Veale JF. Transgender-related stigma and gender minority stress related health disparities in Aotearoa New Zealand: hypercholesterolemia, hypertension, myocardial infarction, stroke, diabetes, and general health. Lancet Reg Health West Pac. 2023;39:100816. https://doi.org/10.1016/j.lanwpc.2023.100816 .
Alencar Albuquerque G, de Lima GC, da Silva QG, et al. Access to health services by lesbian, gay, bisexual, and transgender persons: systematic literature review. BMC Int Health Hum Rights. 2016;14(16):2. https://doi.org/10.1186/s12914-015-0072-9 . PMID:26769484;PMCID:PMC4714514.
National LGBT Survey, United Kingdom, Published 2018. Available at: https://www.gov.uk/government/publications/national-lgbt-survey-summary-report .
Tollemache N, Shrewsbury D, Llewellyn C. Que(e) rying undergraduate medical curricula: a cross-sectional online survey of lesbian, gay, bisexual, transgender, and queer content inclusion in UK undergraduate medical education. BMC Med Educ. 2021;21:100. https://doi.org/10.1186/s12909-021-02532-y .
Obedin-Maliver J, Goldsmith ES, Stewart L, et al. Lesbian, gay, bisexual, and transgender-related content in undergraduate medical education. JAMA. 2011;306(9):971–7.
Greene MZ, France K, Kreider EF, et al. Comparing medical, dental, and nursing students’ preparedness to address lesbian, gay, bisexual, transgender, and queer health. PLoS One. 2018;13(9):e0204104. https://doi.org/10.1371/journal.pone.0204104 .
Barber A, Flach A, Bonnington J, et al. LGBTQ+ Healthcare Teaching in UK Medical Schools: An Investigation into Medical Students’ Understanding and Preparedness for Practice. J Med Educ Curric Dev. 2023;10. https://doi.org/10.1177/23821205231164893
Office of National Statistics, United Kingdom. Available at: https://www.ons.gov.uk/peoplepopulationandcommunity/culturalidentity/sexuality/bulletins/sexualidentityuk/2021and2022#:~:text=1.-,Main%20points,increase%20from%202.1%25%20in%202017 . Accessed 3 Jan 2024.
Berner AM, Hughes DJ, Tharmalingam H, et al. An evaluation of self-perceived knowledge, attitudes and behaviours of UK oncologists about LGBTQ+ patients with cancer. ESMO open. 2020;5(6): e000906. https://doi.org/10.1136/esmoopen-2020-000906 .
UK Census 2021, Office of National Statistics. Available at : https://census.gov.uk/census-2021-results
Carrie D. Johnston, Lee S. Shearer. Internal Medicine Resident Attitudes, Prior Education, Comfort, and Knowledge Regarding Delivering Comprehensive Primary Care to Transgender Patients., Transgend Health. 2017 91-95. https://doi.org/10.1089/trgh.2017.0007
Streed CG Jr, Hedian HF, Bertram A, et al. Assessment of Internal Medicine Resident Preparedness to Care for Lesbian, Gay, Bisexual, Transgender, and Queer/Questioning Patients. J Gen Intern Med. 2019;34(6):893–8. https://doi.org/10.1007/s11606-019-04855-5 . Epub 2019 Mar 7. PMID: 30847829; PMCID: PMC6544682.
Narayan R, Rodriguez C, Araujo J, et al. Constructivism—Constructivist learning theory. In: Irby BJ, Brown G, Lara-Alecio R, Jackson S, editors., et al., The handbook of educational theories. IAP Information Age Publishing; 2013. p. 169–83.
Google Scholar
Bidell MP. The Lesbian, Gay, Bisexual, and Transgender Development of Clinical Skills Scale (LGBT-DOCSS): Establishing a New Interdisciplinary Self-Assessment for Health Providers. J Homosex. 2017;64(10):1432–60. https://doi.org/10.1080/00918369.2017.1321389 .
Singleton M, Green D, Enguidanos S. Identifying Healthcare Stereotype Threat in Older Gay Men Living with HIV. J Appl Gerontol. 2023; 7334648231167944–7334648231167944.
Shires DA, Stroumsa D, Jaffee KD, Woodford MR. Primary care providers’ willingness to continue gender-affirming hormone therapy for transgender patients. Fam Pract. 2018;35(5):576–81. https://doi.org/10.1093/fampra/cmx119 .
GLAAD United Kingdom Education and Training. Available at: https://gladd.co.uk/#education-and-activism . Accessed 11 May 2024.
Education and Training at Fenway Health Institute. Available at : https://fenwayhealth.org/the-fenway-institute/education/ . Accessed 11 May 2024.
LGBT in Britain report - Health. Stonewall; 2018. Available from: https://www.stonewall.org.uk/lgbt-britain-health
UK Foundation Programmes Curriculum 2021, UK General Medical Council, UKFP Curriculum 2021 (gmc-uk.org), Available at: https://www.gmc-uk.org/-/media/documents/fp-curr-oct22-v7_pdf-101343583.pdf
JRCPTB IMT curriculum 2019, Internal Medicine stage 1 curriculum FINAL 111217.pdf (jrcptb.org.uk). Available at: https://www.jrcptb.org.uk/internal-medicine
Cooper MB, Chacko M, Christner J. Incorporating LGBT Health in an Undergraduate Medical Education Curriculum Through the Construct of Social Determinants of Health. MedEdPORTAL. 2018;7(14):10781. https://doi.org/10.15766/mep_2374-8265.10781 . PMID:30800981;PMCID:PMC6342423.
Burcheri A, Coutin A, Bigham, et al. Exploring a case for education about sexual and gender minorities in postgraduate emergency medicine training: forming recommendations for change. Postgrad Med. 2023;135(6):623–32. https://doi.org/10.1080/00325481.2023.2225329 .
Download references
Acknowledgements
Rachel McDonnell, Rahul Pathak, Gerald Edgbury, Michael Brady, Catherine Bryant, Ban Haider, Margot Turner, Muhammad Hyder Junejo
No funding was obtained for this research project.
Author information
Authors and affiliations.
Faculty of Health, Medicine and Social Care, Anglia Ruskin University, Cambridge, UK
Andrew Crowe & Catherine Meads
St Pancras Hospital, Central and North West London NHS Foundation Trust, London, UK
Patrick Hogan
Public Health Directorate, NHS Grampian, Aberdeen, UK
Christoper Morrison
Kings College Hospital NHS Foundation Trust, London, UK
Daniel Bailey
You can also search for this author in PubMed Google Scholar
Contributions
All authors were involved in the design of this study. AC was involved in acquisition, analysis, and interpretation of data, along with drafting the manuscript. All authors contributed equally to the critical review, editing and final approval of the manuscript and were responsible for the decision to submit the manuscript.
Corresponding author
Correspondence to Andrew Crowe .
Ethics declarations
Ethics approval and consent to participate.
Ethical Approval was granted by the School Research Ethics Panel (SREP) of the Health, Education, Medicine, and Social Care (HEMS) faculty of Anglia Ruskin University . All participants consented to participate before taking the online survey by reading the participant information leaflet and ticking a box. This was informed consent.
Consent for publication
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Crowe, A., Hogan, P., Morrison, C. et al. Towards inclusive healthcare: evaluating knowledge, confidence and awareness of LGBTQ + health among Internal Medicine Trainees in London. BMC Med Educ 24 , 851 (2024). https://doi.org/10.1186/s12909-024-05827-y
Download citation
Received : 16 February 2024
Accepted : 26 July 2024
Published : 07 August 2024
DOI : https://doi.org/10.1186/s12909-024-05827-y
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Medical education
- Postgraduate
- Sexual orientation
- Gender identity
- Internal medicine
BMC Medical Education
ISSN: 1472-6920
- Submission enquiries: [email protected]
- General enquiries: [email protected]
- Open access
- Published: 08 August 2024
HPV vaccine behaviors and intentions among a diverse sample of women aged 27-45 years: implications for shared clinical decision-making
- Jennifer D. Allen 1 ,
- Nadia N. Abuelezam 2 ,
- Raviv Rose 1 ,
- Katelin Isakoff 1 ,
- Gregory Zimet 3 &
- Holly B. Fontenot 4
BMC Public Health volume 24 , Article number: 2154 ( 2024 ) Cite this article
273 Accesses
Metrics details
The Advisory Committee on Immunization Practices issued a shared clinical decision-making (SCDM) recommendation for HPV vaccination in persons aged 27–45. Since expanded eligibility for the vaccine was issued, little information has been available about HPV vaccine behaviors and intentions among women in this age group.
We conducted a cross-sectional online survey among women aged 27–45 years recruited through a Qualtrics™ respondent panel ( N = 324) to answer the following questions (1) What is the prevalence of HPV vaccination among a diverse sample of adult women aged 27–45 years? (2) What are the characteristics of those who have or have not previously been vaccinated? and (3) What factors are associated with the intention to obtain the HPV vaccine among those who had never been vaccinated? Multivariable logistic regression analyses estimated adjusted odds ratios (AORs) and 95% confidence intervals (95% CIs).
Only 31.1% had at least one dose of the HPV vaccine. In multivariable analyses, those more likely to have been vaccinated were younger and were more likely to believe that the vaccine was effective. Of those unvaccinated or unsure, 54.8% indicated they were likely to get vaccinated in the future. Factors associated with future vaccine intention (compared to those not intending) included beliefs about vaccine testing, perceived likelihood of HPV infection, greater comfort in asking one’s provider for vaccination, and prior negative healthcare experiences.
Conclusions
Our findings suggest that many women in this age group are interested in HPV vaccination. While the recommendation is for SCDM rather than routine vaccination for all women in this age group, efforts to promote informed decision-making among mid-adult women may include educating women about the rigorous vaccine testing and approval process, their risk factors for HPV infection, and encouraging them to engage in SCDM with their medical providers. Targeted efforts to reach women who have had negative experiences with healthcare may also be needed.
Peer Review reports
Introduction
Despite the availability of primary prevention via vaccination, the human papillomavirus (HPV) remains the most common sexually transmitted infection in the United States (U.S.). Each year, approximately 14 million people in the U.S. are newly infected with HPV, and about 35,900 develop a new cervical or other HPV-related cancer [ 1 , 2 ]. Highly effective HPV vaccines have been available since 2006 and, until recently, were only approved for those aged 9 through 26 years by the U.S. Food and Drug Administration (FDA). In 2018, the FDA expanded its approval to include persons aged 27–45 years (“mid-adults”) [ 3 ]. Subsequently, in 2019, the Advisory Committee on Immunization Practices (ACIP) issued a shared clinical decision-making recommendation for HPV vaccination for mid-adults not previously vaccinated [ 4 ]. SCDM involves a discussion between patients and providers about the best available evidence for HPV vaccination, individual risks, and vaccine benefits versus potential harms to arrive at a mutually agreeable decision [ 5 ]. As this recommendation differs from routine vaccination that is recommended for younger women, there is a need to understand the extent to which the population of mid-adult women not previously vaccinated would prefer to be vaccinated with the knowledge that they are now potentially eligible [ 6 ].
While expansion of eligibility for the HPV vaccine among persons aged 27–45 years provides a significant step towards minimizing the potential burden of HPV-related cancers, uptake of the vaccine among populations for whom it had been previously recommended (routine recommendation for adolescents aged 11–13 years; catch up recommendation for those aged 18–26 years) has yet to meet the national goal of 80% series completion [ 7 ]. National data from 2018 show that only 65% of adolescents and 21.5% of women aged 18–26 had completed the recommended vaccine series [ 8 , 9 ]. Women not previously vaccinated but beyond the (previous) age limit of 26 years are now eligible for HPV vaccination. Despite the plethora of literature explicating factors associated with HPV vaccine uptake in general [ 10 , 11 , 12 , 13 ], there is limited research examining predictors among women ages 27–45 years, which may inform the SCDM process.
Our study was guided by tenets of the Theory of Reasoned Action, which postulates that behavioral intention is the strongest predictor of future behavior, barring environmental constraints. Intention, in turn, is shaped by attitudes, beliefs, subjective norms, perceived behavioral control, and environmental factors [ 14 ]. In this study, we examined attitudes and beliefs about vaccines (e.g., safety, efficacy), as well as the perceived risk of HPV infection and cervical cancer. For social influences, we examined perceptions of social pressure to be vaccinated, including the extent to which significant others (e.g., spouse, friends) support vaccination, and characteristics of the patient/provider relationship (e.g., trust, comfort). We assessed self-efficacy about requesting information or vaccination from one’s provider. We also evaluated environmental barriers, including vaccine cost and prior negative experiences with the healthcare system.
The objectives of this study were to answer three research questions (RQs): (1) What is the prevalence of HPV vaccination among a diverse sample of mid-adult women aged 27–45 years? (2) What are the characteristics of those who have or have not previously been vaccinated? and (3) What factors are associated with the intention to obtain the HPV vaccine among those who have never been vaccinated? Findings will guide healthcare provider vaccine communication strategies and efforts to promote SCDM about HPV vaccination among adult women.
Materials and methods
Participants.
We conducted an online survey of adult women between April 13, 2020, and June 8, 2020. Participants were drawn from the Qualtrics XM Panel, a database of potential online survey participants maintained by Qualtrics, XM and all respondents have already agreed to be contacted for research purposes. Criteria for inclusion were persons assigned female at birth, aged 27 to 45 years, resident of the U.S., and ability to read and understand English. Given documented differences in HPV vaccination across racial/ethnic groups [ 15 ], we oversampled women of minoritized racial/ethnic groups to produce a sample that was 25% Black, 25% Asian, and 25% Hispanic. A further sampling quota was set to recruit a sample that included 25% of respondents who identified as a sexual minority (e.g., bisexual, lesbian, queer, other). Study consent procedures were conducted electronically before the start of the survey (by clicking on a box indicating consent), participation was voluntary, and the standard Qualtrics XM remuneration ($6) was provided. This study was administered online and approved by Tufts University Institutional Review Board.
We utilized a number of existing measures on the survey (see supplementary file ). First, we assessed both prior receipt and future intentions to get the HPV vaccine, as well as key independent variables from our conceptual framework (i.e., attitudes/beliefs, social influences, self-efficacy, and barriers), as described below. All participants responded to questions related to demographics, vaccination status, and attitudes/beliefs toward HPV vaccination. Only those who reported never having had the HPV vaccine were asked about their future intentions to get vaccinated, and questions were asked to assess social influences, self-efficacy, and barriers toward future HPV vaccination.
Receipt of and intention to vaccinate . Using items from the Behavioral Risk Factor Surveillance System [ 16 ], we asked participants if they had ever received the HPV vaccine (yes, no or unsure). Those who had not been vaccinated or were unsure were asked, “How likely are you to get the HPV vaccine?” with responses on a 4-point Likert scale (“very likely” to “very unlikely”).
Attitudes and beliefs . To assess attitudes and beliefs about vaccines, we asked: [ 1 ] “Do you think vaccines are well tested before being made available to the public?” (yes, no, don’t know) [ 2 ], “In your opinion, how safe is the HPV vaccine?” (4-point Likert: “very safe” to “not at all safe”/ “don’t know” with higher scores indicating greater perceived safety) [ 3 ], “In your opinion, how effective is the HPV vaccine?” (4-point Likert, “very effective” to “not at all effective”/ “don’t know,” with higher scores indicating greater perceived effectiveness), and [ 4 ] “If you were making a decision about getting the HPV vaccine, how much would potential side effects influence your decision?” (4-point Likert, “a great deal” to “not at all,” with higher scores indicating greater concern about side effects). To assess perceived susceptibility to cervical cancer and HPV infection, we asked [ 1 ] “Compared to the average person your age, would you say you are” (“more likely to get cervical cancer,” “as likely to get cervical cancer,” “less likely to get cervical cancer”); and [ 2 ] “How likely are you to get HPV?” (4-point Likert, “very likely” to “very unlikely”/ “unsure”).
Social influences. To assess social factors that influence decision-making about getting the HPV vaccine, we asked, “How much would each of the following factors influence your decision?” Response options included: whether you have a spouse or long-term monogamous partner; how other people close to you might think about your decision; comfort and/or trust with [my] health care provider; and comfort and trust in seeking health care (4-point Likert, “a great deal” to “not at all” for each).
Self-efficacy. We assessed comfort with engaging in SCDM with health care providers by asking two items: [ 1 ] “How comfortable would you be asking your health care provider to give your information regarding the HPV vaccine?” and [ 2 ] “How comfortable would you be asking your health care provider to give you the HPV vaccine?” (4-point Likert, “very comfortable” to “very uncomfortable”/ “unsure/don’t know”).
Barriers. We assessed the extent to which cost and prior negative experiences with the healthcare system impacted vaccine decisions. Specifically, we asked: “If you were making a decision about getting the HPV vaccine, how much would each of the following factors influence your decision?” The factors listed were cost, insurance coverage, and previous negative health care experiences with response options on a 4-point Likert scale (“a great deal” to “not at all”).
Demographic Characteristics. Demographic characteristics were assessed using standard items from the Behavioral Risk Factors Surveillance Survey [ 16 ] and included: race/ethnicity [non-Hispanic (NH) White, NH Black, NH Asian, Hispanic and multi-racial]; age (27–29, 30–39, 40–49); income (<$34,000, $35–74,000, >$75,000, not sure); education (high school or less, college or some college, graduate degree); employment (employed, unemployed); and insurance status (public, private, none). We also assessed prior history of cervical cancer screening [ 1 ] “Have you ever had an abnormal Pap test?; [ 2 ] Have you ever had a positive HPV test?”; and 3) Have you ever been told by a healthcare provider that you had cervical cancer?” (“yes”, “no”, don’t know”).
Our analytic goals were to describe the prevalence of HPV vaccination among mid-adult women (RQ #1), identify factors associated with vaccination status (RQ #2), and examine factors associated with the intention to be vaccinated for HPV among those who had never been vaccinated (RQ #3). Descriptive statistics were tabulated for the total sample, previously vaccinated individuals (≥ 1 dose), and unvaccinated individuals (RQ #1). For RQ #2, we examined factors associated with already having received ≥ 1 dose of the HPV vaccine versus not having received any doses. For RQ #3, we focused only on those who had not received any doses of the HPV vaccine. For this analysis, we examined factors associated with falling into two categories: (1) those who report being very likely or likely to get the HPV vaccine and (2) those who report being very unlikely or unlikely to get the vaccine.
The data analysis was completed using SPSS Statistics Version 26 (IBM Corp., Armonk, NY). For RQs #2 and #3, Chi-squared tests were used to assess bivariate associations. For both RQ #2 and RQ #3, univariable logistic regression models were first run with each of the covariates. Then, stepwise selection was used in the multiple logistic regression analyses (slentry = 0.20 and slstay = 0.10); all covariates with association of p < 0.10 were included, and p ≤ 0.05 was considered statistically significant in the final models. For the multivariable analyses, models were run to assess (RQ #2) factors associated with vaccination status and (RQ #3) factors related to intention to be vaccinated for HPV among those who had never been vaccinated. For these models, those who identified as NH Asian and multi-racial were combined due to the small sample sizes.
Sample characteristics, HPV vaccination prevalence (RQ#1), and factors associations with HPV vaccination (RQ#2)
A total of 324 women completed the survey. The majority were between the ages of 30 and 39 years (51.2%), identified as heterosexual (73.8%), had some college or a college degree (61.1%), and had health insurance (33.3% public, 51.5% private). Approximately half of the sample was married (46.6%). Due to the use of quota sampling, race/ethnicity was distributed relatively evenly (NH White 25.6; NH Black 23.58%; NH Asian and another race 27.2%, and 23.8% Hispanic). Only 31.1% ( n = 101) of the sample reported that they had one or more doses of the HPV vaccine, and 68.9% ( n = 223) reported not being vaccinated or being unsure about vaccination status. See Table 1 .
In the bivariate analyses, age ( p < 0.001), health insurance status ( p = 0.047), report of ever having had an HPV test ( p < 0.01), and having been given a diagnosis of cervical cancer ( p < 0.01) were associated with having had at least one dose of the HPV vaccine. Attitudes and beliefs that were significantly associated with vaccination status included perceived safety of the HPV vaccine ( p < 0.001), perceived effectiveness of the HPV vaccine ( p < 0.001), and perceived likelihood of HPV infection ( p = 0.068) (Table 1 ).
In the multivariable analysis, those aged 30 years or older had lower odds of receiving the HPV vaccine compared to those aged 27–29 years (AOR = 0.50; 95% CI = 0.26–0.96). Women who expressed stronger belief in vaccine effectiveness had higher odds of being vaccinated than those who reported that they did not view the vaccine as effective or did not know about effectiveness (AOR = 2.75; 95% CI = 1.33–5.71) (Table 2 ).
RQ #3: Factors associated with the intention to obtain the HPV vaccine among those who had never been vaccinated or were unsure
Of the 223 participants who reported not being vaccinated, 6 did not complete the remainder of the survey items, so the final sample size for RQ #3 and RQ #4 was n = 217. In bivariate analyses, women who intended to be vaccinated were those who reported having had an abnormal Pap test ( p < 0.001) or positive HPV test ( p < 0.05), stronger beliefs that vaccines are well tested ( p < 0.001), believed that the HPV vaccine was safe ( p < 0.001) and effective ( p < 0.001), and perceived themselves to be more likely to get an HPV infection ( p < 0.05). Furthermore, social influences [i.e., other people’s opinions ( p = 0.08) and health care provider recommendation ( p < 0.05)] a self-efficacy [i.e., comfort asking a health care provider for information about HPV vaccine ( p < 0.01) and HPV vaccination ( p < 0.001)], were more likely to report the intention to get vaccinated. In contrast, barriers [i.e., reporting that previous negative healthcare experiences would influence vaccine decision-making ( p = 0.06)] were associated with lower future vaccination intentions (Table 3 ).
In the multivariable analysis, four factors were significantly associated with the intention to obtain an HPV vaccine. Compared with those who did not intend to be vaccinated, those reporting a belief that vaccines are well tested (AOR = 2.28; 95% CI = 1.12–4.63), that they were likely to get an HPV infection (AOR = 2.66; 95%CI = 1.16–6.05), and that they were comfortable asking a health care provider for the HPV vaccine (AOR = 4.53; 95%CI = 1.59–12.88) had greater odds of intending to be vaccinated. In contrast, those reporting that previous negative healthcare experiences would influence their HPV vaccination decision had lower odds of intending to be vaccinated (AOR = 0.40; 95% CI = 0.20–0.80) (Table 4 ).
The expansion of eligibility for the HPV vaccine to persons aged 27–45 years provides new opportunities to prevent HPV-related cancers, precancers, and genital warts. However, we found that less than a third of the women in this sample had received one or more doses of the HPV vaccine. Of those who were unvaccinated or unsure about their vaccination history, more than half reported that they were very likely or likely to accept HPV vaccination in the future.
Only a few national studies of mid-adult women (27–45 years) have been conducted since the change in ACIP guidelines [ 17 , 18 ], though several other studies have looked at both women and men [ 19 , 20 ]. Our finding that approximately half of those previously unvaccinated were willing to get the vaccine is similar to findings from other national surveys conducted since that time, which ranged from 33% among sexual minority women [ 21 ] and 43–54% among broader samples of women [ 13 , 19 ]. One study found that about 52% of individuals (both women and men) were willing to ask their providers for HPV vaccine information [ 13 ]. Studies have also found that perceived vaccine effectiveness, perceived safety [ 19 ], perceived vulnerability [ 19 ], and perceived likelihood of benefitting from the vaccine [ 22 ] were associated with willingness/intent to get the HPV vaccine in this age group. Our study provides new information about the relationship between prior negative healthcare experiences and HPV vaccination. This is notable since those who had negative experiences were 60% less likely to report they would be willing to be vaccinated.
Findings should be viewed in light of study limitations. First, this was a convenience sample from a Qualtrics™ panel, so the findings may not be generalizable to other populations. However, recent research has found that while participants recruited from online panels are not necessarily representative of the U.S. population, they are equivalently representative as traditional recruitment approaches [ 23 ]. Regardless, we recognize that those willing to complete research studies may be more likely to be receptive to health interventions than the general public. Additionally, we did not assess HPV vaccine intentions over a specific time frame, as is often done in “stages of change” models. Like most other studies in this field, vaccination status was assessed through self-report, which, while generally accurate among adults, is less accurate than status confirmed via medical records or vaccine registries [ 24 ]. We also acknowledge that some of the confidence intervals in our findings were wide. Therefore, findings should be interpreted with caution, as estimates may be unstable. Nevertheless, study strengths include the timeliness of findings vis-a-vis expansion of HPV vaccine eligibility, and the ability to compare across different racial/ethnic groups for whom there has been inadequate representation in prior research.
Implications for practice
Since SCDM, rather than routine HPV vaccination, is recommended for women in this age group, those not intending to be vaccinated may require no intervention so long as they are fully informed about the potential benefits of vaccination and their individual risk for infection and cervical cancer. However, prior studies indicate that most women do not know that there is now expanded vaccine eligibility [ 20 ]. Further, many women, especially those with lower levels of education and who are racially/ethnically minoritized, are not aware that HPV causes cervical cancer [ 25 ]. Although we did not assess knowledge of risk factors in this study, our finding that two-thirds of women were willing to be vaccinated and that being comfortable asking one’s healthcare provider for the vaccine suggests that efforts to build awareness and provide skills about engaging in SCDM with one’s provider may hold promise. These efforts will also likely require additional information about the rigor of HPV vaccine testing to promote confidence among some women. Targeted efforts may be needed to engage women who had prior negative experiences in the healthcare system, as they were 60% less likely to report that they would be vaccinated in the future. Existing evidence points to the role of medical mistrust in vaccine hesitancy in general [ 26 ], including HPV vaccination [ 27 ]. Building trust in the healthcare system will likely necessitate multilevel interventions, not only directed at individuals who have mistrust, but also to enhance trust with providers [ 28 , 29 ], as well as the trustworthiness of healthcare systems. Comprehensive efforts should also include provider-directed interventions, as the ACIP guidelines do not provide specific information about which patients may most benefit from vaccination. This is especially important since provider recommendation has been identified as the most influential factor in vaccine acceptance [ 30 ] and continues to be for this age group ( 21 ). Yet, in a recent study of primary care physicians, only 42% had recommended HPV vaccination to adults aged 27–45 years, and 57% were unsure about what to discuss during SCDM conversations [ 31 ].
The expanded eligibility of HPV vaccination for individuals aged 27–45 years has the potential to reduce cervical and other HPV-related cancers, as well as genital warts [ 32 ]. However, since a recommendation for SCDM for this age group differs from the routine HPV vaccination recommended for younger age groups, greater efforts to ensure awareness of expanded eligibility and potential benefits may be needed among patients and providers through a mutual discussion about vaccination. Our results suggest that many unvaccinated mid-adult women may be interested in receiving the HPV vaccine, and an even more significant number may be interested once efforts are made to increase their understanding of the safety, efficacy, and testing of the vaccine, as well as expanded eligibility.
Data availability
The data that support the findings of this study are available from the corresponding author, but restrictions apply to the availability of these data. The data are, however, available from the authors upon reasonable request.
Centers for Disease Control. HPV and Cancer. 2020 [cited 2021 Jul 19]. How Many Cancers Are Linked with HPV Each Year? | CDC. https://www.cdc.gov/cancer/hpv/statistics/cases.htm
National Foundation for Infectious Diseases. National Foundation for Infectious Diseases. 2019 [cited 2021 Jul 19]. HPV (Human Papillomavirus). https://www.nfid.org/infectious-diseases/hpv/
Commissioner O. of the. FDA. FDA; 2020 [cited 2024 Apr 3]. FDA approves expanded use of Gardasil 9 to include individuals 27 through 45 years old. https://www.fda.gov/news-events/press-announcements/fda-approves-expanded-use-gardasil-9-include-individuals-27-through-45-years-old
Meites E, Szilagyi PG, Chesson HW, Unger ER, Romero JR, Markowitz LE. Human papillomavirus vaccination for adults: updated recommendations of the Advisory Committee on Immunization practices. MMWR Morb Mortal Wkly Rep. 2019;68(32):698–702.
Article PubMed PubMed Central Google Scholar
Elwyn G, Frosch D, Thomson R, Joseph-Williams N, Lloyd A, Kinnersley P, et al. Shared decision making: a model for clinical practice. J GEN INTERN MED. 2012;27(10):1361–7.
Ellingson MK, Oliveira CR, Sheth SS, Sullivan EL, Torres A, Shapiro ED, et al. Patterns of recommended vaccine receipt among women ages 24–45 years: a cross-sectional analysis. BMC Public Health. 2021;21(1):1–8.
Article Google Scholar
Department of Health and Human Services. Increase the proportion of adolescents who get recommended doses of the HPV vaccine — IID–08 - Healthy People 2030 | health.gov [Internet]. Department of Health and Human Services; 2020 [cited 2021 Jul 24]. https://health.gov/healthypeople/objectives-and-data/browse-objectives/vaccination/increase-proportion-adolescents-who-get-recommended-doses-hpv-vaccine-iid-08
Boersma P. Hum Papillomavirus Vaccination among Adults. 2020;(354):8.
Walker TY, National. Regional, State, and Selected Local Area Vaccination Coverage Among Adolescents Aged 13–17 Years — United States, 2018. MMWR Morb Mortal Wkly Rep [Internet]. 2019 [cited 2021 Jul 24];68. https://www.cdc.gov/mmwr/volumes/68/wr/mm6833a2.htm
Brewer NT, Fazekas KI. Predictors of HPV vaccine acceptability: a theory-informed, systematic review. Prev Med. 2007;45(2):107–14.
Article PubMed Google Scholar
Kessels SJM, Marshall HS, Watson M, Braunack-Mayer AJ, Reuzel R, Tooher RL. Factors associated with HPV vaccine uptake in teenage girls: a systematic review. Vaccine. 2012;30(24):3546–56.
Newman PA, Logie CH, Lacombe-Duncan A, Baiden P, Tepjan S, Rubincam C, et al. Parents’ uptake of human papillomavirus vaccines for their children: a systematic review and meta-analysis of observational studies. BMJ Open. 2018;8(4):e019206.
Thompson EL, Garg A, Galvin AM, Moore JD, Kasting ML, Wheldon CW. Correlates of HPV Vaccination Intentions Among Adults Ages 27–45 Years Old in the U.S. J Community Health [Internet]. 2021 Feb 13 [cited 2021 Jul 24]; http://link.springer.com/ https://doi.org/10.1007/s10900-021-00968-3
Fishbein M, Ajzen I. Predicting and changing behavior: the reasoned Action Approach. New York: Psychology; 2009. p. 538.
Google Scholar
Agénor M, Pérez AE, Peitzmeier SM, Borrero S. Racial/ethnic disparities in human papillomavirus vaccination initiation and completion among U.S. women in the post-affordable Care Act era. Ethn Health. 2020;25(3):393–407.
Centers for Disease Control. CDC - BRFSS - Questionnaires [Internet]. 2021 [cited 2021 Jul 24]. https://www.cdc.gov/brfss/questionnaires/index.htm
Arevalo M, Brownstein NC, Whiting J, Vadaparampil ST, Head KJ, Meade CD, et al. Factors related to human papillomavirus vaccine uptake and intentions among adults aged 18–26 and 27–45 years in the United States: a cross-sectional study. Cancer. 2023;129(8):1237–52.
Article CAS PubMed Google Scholar
Polonijo AN, Mahapatra D, Brown B. I thought it was just for teenagers: knowledge, attitudes, and beliefs about HPV Vaccination among women aged 27 to 45. Womens Health Issues. 2022;32(3):301–8.
Galvin AM, Garg A, Griner SB, Moore JD, Thompson EL. Health Literacy Correlates to HPV Vaccination among US adults ages 27–45. J Cancer Educ. 2023;38(1):349–56.
Thompson EL, Wheldon CW, Rosen BL, Maness SB, Kasting ML, Massey PM. Awareness and knowledge of HPV and HPV vaccination among adults ages 27–45 years. Vaccine [Internet]. 2020 Feb 3 [cited 2020 Mar 6]; http://www.sciencedirect.com/science/article/pii/S0264410X20300797
Reiter PL, Bustamante G, McRee AL. HPV vaccine coverage and acceptability among a national sample of sexual minority women ages 18–45. Vaccine. 2020;38(32):4956–63.
Thompson EL, Garg A, Galvin AM, Moore JD, Kasting ML, Wheldon CW. Correlates of HPV vaccination intentions among adults ages 27–45 years old in the U.S. J Community Health. 2021;46(5):893–902.
Guo X, Vittinghoff E, Olgin JE, Marcus GM, Pletcher MJ. Volunteer Participation in the Health eHeart Study: a comparison with the US Population. Sci Rep. 2017;7(1):1956.
Rolnick SJ, Parker ED, Nordin JD, Hedblom BD, Wei F, Kerby T, et al. Self-report compared to electronic medical record across eight adult vaccines: do results vary by demographic factors? Vaccine. 2013;31(37):3928–35.
Article CAS PubMed PubMed Central Google Scholar
Stephens ES, Dema E, McGee-Avila JK, Shiels MS, Kreimer AR, Shing JZ. Human papillomavirus awareness by Educational Level and by race and ethnicity. JAMA Netw Open. 2023;6(11):e2343325.
Allen JD, Fu Q, Shrestha S, Nguyen KH, Stopka TJ, Cuevas A, et al. Medical mistrust, discrimination, and COVID-19 vaccine behaviors among a national sample U.S. adults. SSM Popul Health. 2022;20:101278.
Tsui J, Martinez B, Shin MB, Allee-Munoz A, Rodriguez I, Navarro J, et al. Understanding medical mistrust and HPV vaccine hesitancy among multiethnic parents in Los Angeles. J Behav Med. 2023;46(1–2):100–15.
Harrington N, Chen Y, O’Reilly AM, Fang CY. The role of trust in HPV vaccine uptake among racial and ethnic minorities in the United States: a narrative review. AIMS Public Health. 2021;8(2):352–68.
Greene J, Ramos C. A mixed methods examination of Health Care Provider behaviors that build patients’ Trust. Patient Educ Couns. 2021;104(5):1222–8.
Ojha RP, Tota JE, Offutt-Powell TN, Klosky JL, Ashokkumar R, Gurney JG. The accuracy of human papillomavirus vaccination status based on adult proxy recall or household immunization records for adolescent females in the United States: results from the National Immunization Survey-Teen. Ann Epidemiol. 2013;23(5):281–5.
Hurley LP, O’Leary ST, Markowitz LE, Crane LA, Cataldi JR, Brtnikova M, et al. US Primary Care Physicians’ viewpoints on HPV vaccination for adults 27 to 45 years. J Am Board Fam Med. 2021;34(1):162–70.
King LM, Lewnard JA, Niccolai LM. Clinical and Public Health Considerations for HPV Vaccination in Midadulthood: a narrative review. Open Forum Infect Dis. 2023;10(1):ofad004.
Download references
Acknowledgements
We wish to thank the study participants who shared their time and perspectives.
This work was supported by the Tufts University Office of the Vice Provost for Research (PI Allen). The funding agency had no role in the study.
Author information
Authors and affiliations.
Department of Community Health, Tufts University School of Arts and Sciences, 574 Boston Ave, Medford, MA, 02155, USA
Jennifer D. Allen, Raviv Rose & Katelin Isakoff
Connell School of Nursing, Boston College, 140 Commonwealth Ave., Chestnut Hill, MA, 02467, USA
Nadia N. Abuelezam
Department of Pediatrics, Division of Adolescent Medicine, School of Medicine, Indiana University, 410 W. 10th Street, HS 1001, Indianapolis, IN, 46202, USA
Gregory Zimet
Nancy Atmospera-Walch School of Nursing, University of Hawaii at Manoa, 2528 McCarthy Mall, Honolulu, HI, 96822, USA
Holly B. Fontenot
You can also search for this author in PubMed Google Scholar
Contributions
All authors made substantial contributions to the interpretation of data and have drafted the work or substantively revised it. All authors have approved the submitted version and agree both to be personally accountable for the author’s own contributions and to ensure that questions related to the accuracy or integrity of any part of the work, even ones in which the author was not personally involved, are appropriately investigated, resolved, and the resolution documented in the literature.
Corresponding author
Correspondence to Jennifer D. Allen .
Ethics declarations
Ethics approval and consent to participate.
All study protocols and procedures were approved by the Institutional Review Board at Tufts University (protocol number: 00001954). All study activities were carried out in accordance with the relevant guidelines and regulations of the Declaration of Helsinki. Informed consent was obtained from all participants.
Consent for study participation
Study consent procedures were conducted electronically before the start of the survey (by clicking on a box indicating consent box).
Consent for publication
Not applicable.
Competing interests
JDA, RR, KI, NA have no competing interests. Outside of the current work, Gregory Zimet has received consulting fees from Merck for work on HPV vaccination and has served on external advisory committees for Moderna (COVID-19 vaccination) and Pfizer (meningococcal vaccination), and through Indiana University, has also received investigator-initiated grant funding from Merck related to HPV vaccination. Dr. Fontenot has also received investigator-initiated grant funding from Merck regarding adolescent vaccination. Drs. Fontenot and Zimet confirm that their funding source(s) have had no involvement in the conduct of this work. No other authors have conflicts of interest to report, nor competing financial interests.
Additional information
Publisher’s note.
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Supplementary Material 1
Rights and permissions.
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/ . The Creative Commons Public Domain Dedication waiver ( http://creativecommons.org/publicdomain/zero/1.0/ ) applies to the data made available in this article, unless otherwise stated in a credit line to the data.
Reprints and permissions
About this article
Cite this article.
Allen, J.D., Abuelezam, N.N., Rose, R. et al. HPV vaccine behaviors and intentions among a diverse sample of women aged 27-45 years: implications for shared clinical decision-making. BMC Public Health 24 , 2154 (2024). https://doi.org/10.1186/s12889-024-18740-2
Download citation
Received : 04 January 2024
Accepted : 29 April 2024
Published : 08 August 2024
DOI : https://doi.org/10.1186/s12889-024-18740-2
Share this article
Anyone you share the following link with will be able to read this content:
Sorry, a shareable link is not currently available for this article.
Provided by the Springer Nature SharedIt content-sharing initiative
- Human papillomavirus
- Adult women
- HPV vaccine
- Attitudes and behaviors
BMC Public Health
ISSN: 1471-2458
- General enquiries: [email protected]

Sleep Expert Jamie Zeitzer, PhD, Seeks the Truth
By Sharon Brock, MEd, MS

Like a Renaissance explorer, the insatiable curiosity of Jamie Zeitzer, PhD, drives him to explore big questions as a senior researcher and co-director of the Stanford Center for Sleep and Circadian Sciences. After 25 years of conducting sleep research, including groundbreaking studies on circadian rhythms and how light affects the brain, Dr. Zeitzer still enjoys navigating the uncertain waters of research and helping us all get a better night’s sleep.
“What drives me in research is the fact that I don’t like uncertainty and I don’t like being wrong, and I picked a profession where I’m literally wrong all the time,” says Dr. Zeitzer, Professor (Research) of Psychiatry and Behavioral Sciences.
“Usually, the most obvious path in research is based on incomplete information, and it ends up not being true. So, we ask, what can we learn from this? How can we reformulate this question in a way to disprove this theory and try again? As a researcher, I’m consistently wrong but because I hate being wrong, I’m driven to get to the bottom of these big questions and seek the truth with a capital T,” says Dr. Zeitzer.
How Much Sleep Do We Need?
Since millions of Americans struggle with disordered sleep, Dr. Zeitzer is constantly fielding questions as a public health ambassador. However, it’s often difficult for him to give a straight answer because a) he doesn’t want to be wrong, and b) there’s a lot of grey area when it comes to sleep research.
For example, the number of hours of sleep we need for good health is different for everyone. For some, six hours of sleep is sufficient while others need more than nine. To assess how much sleep someone needs, Dr. Zeitzer recommends monitoring how sleepy they feel during the day without skewing the results with caffeine.
“If I get pressed, I say, ‘Most people need seven hours per night’,” says Dr. Zeitzer, “but, since many of us caffeinate away our sleepiness, we aren’t making the accurate self-assessment about how much sleep we really need.”
Other factors that affect sleep amount and quality are shift work, behavior choices, household or family obligations (i.e. having infants or teens in the house) or having anxiety about sleep.
“If you’re a bad sleeper, I say, ‘Don’t worry about it,’ because worrying about it just makes it worse. Now you have inadequate sleep and anxiety,” says Dr. Zeitzer. “With consistent bad sleep, yes, there is a slight increased risk of diabetes, Alzheimer’s, injury or poor cognition down the line, but the thing that’s really going to kill you is anxiety. So, if we first reduce anxiety about sleep, then we can work in a positive way about getting better sleep.”
Current Research
Even though millions of Americans struggle with sleep, others, such as many teens and 20-somethings, don’t prioritize sleep because they’d rather stay awake. Since there is delayed gratification related to the benefits of good sleep, many young people would prefer to stay out late with their friends or engage with technology into the night. Given the dopamine reward from receiving “likes” on social media or the stimulation of computer games, it’s more enjoyable for teens to stay awake on their phones rather than going to sleep.
“Growing up on Long Island in the 80s, I watched Star Trek every night, but by midnight, there was nothing on TV, so I went to sleep,” says Dr. Zeitzer. “But now, you can watch Netflix or play computer games all night because entertainment is optimized to never stop watching.”
Dr. Zeitzer is currently the father of three teenagers, so although he is working on several research projects, the one he is most excited about examines the sleep patterns of teens. He explains that two major factors are causing teens to go to sleep later—puberty causes the circadian system to shift to a later hour and entertainment is designed to make it more enjoyable to stay awake.
To address the circadian system so that teens feel sleepy at an earlier time, the research team uses lights that flash on a timer and shift the teenage brain into a different time zone. As a psychological intervention to promote behavior change, researchers also created videos with sleep information tailored for teens.
“We wanted the videos to be relatable and entertaining, so we came up with archetypes to represent three sleep patterns we find in teens,” says Dr. Zeitzer. “The night owl is named Otis, who doesn’t feel sleepy until 2:00 a.m. because of his circadian window. The mountain lion is Libby—she’s the alpha type who can’t turn her brain off. And the raccoon is named Rocky; he’s our gamer. Everyone loves Rocky—he plops on the couch after school, takes a nap, listens to EDM, then games all night.”
Educational Background
As an undergraduate, Dr. Zeitzer attended Vassar college in New York and received a diverse liberal arts education. Although he majored in biology, he took many English, philosophy, art, and medieval studies courses. He went on to attend Harvard University to earn a PhD in neurobiology and wrote his thesis on human circadian rhythms and how light impacts the circadian clock and regulates melatonin.
Dr. Zeitzer came to Stanford as a post-doc in 2001, achieved a faculty position in 2006, and has been running his own sleep lab for the past 18 years.
“I’ve been very fortunate that I’ve been able to explore a variety of scientific questions that I find intellectually compelling over the course of my career,” says Dr. Zeitzer. “I enjoy collaborating with other researchers who are experts in their field. My goal in science is not to have the highest-paid lab, but to get as close to the truth as possible—I would rather be close to the truth than anything else.”
You might also like

- Lifestyle Medicine Blog
- In the News
- Research Publications
- Movement & Exercise
- Healthful Nutrition
- Restorative Sleep
- Stress Management
- Social Engagement
- Cognitive Enhancement
- Gratitude & Reflection
Email (required) *
Example: Yes, I would like to receive emails from Lifestyle Medicine. (You can unsubscribe anytime)

New research identifies key protein complex in cellular quality control
- Download PDF Copy
Anyone who's tried to neatly gather a fitted sheet can tell you: folding is hard. Get it wrong with your laundry and the result can be a crumpled, wrinkled mess of fabric, but when folding fails among the approximately 7,000 proteins with an origami-like complexity that regulate essential cellular functions, the result can lead to one of a multitude of serious diseases ranging from emphysema and cystic fibrosis to Alzheimer's disease. Fortunately, our bodies have a quality-control system that identifies misfolded proteins and marks them either for additional folding work or destruction, but how, exactly, this quality-control process functions is not entirely known. Researchers at the University of Massachusetts Amherst have now made a major leap forward in our understanding of how this quality-control system works by discovering the "hot spot" where all the action takes place. The research was published recently in the Proceedings of the National Academy of Sciences .
DNA may be the master blueprint for life, but it is of proteins that we're built. While many of them are structurally simple, there are approximately 7,000 proteins that must be made in a cell's secretory pathway and will be either dispersed throughout the cell or secreted to the extracellular space in order to perform their essential functions.
The story begins in the endoplasmic reticulum-;the cellular protein factories responsible for correctly building thousands of different proteins-;and involves two main players: an enzyme known as UGGT and the partner protein Sep15. Senior authors Daniel Hebert, professor of biochemistry and molecular biology at UMass Amherst, and Lila Gierasch, distinguished professor of biochemistry and molecular biology and chemistry at UMass Amherst, along with co-author, Kevin Guay, a graduate student in the molecular cellular biology program at UMass Amherst, had shown in previous research that UGGT acts as a "gatekeeper" by reading carbohydrate tags, called N-glycans, embedded into the protein to determine whether or not the protein is correctly folded.
"But there's something else at work," says lead-author Rob Williams, a postdoctoral fellow with a joint appointment in both Hebert's and Gierasch's labs. "There's an exclusive club of proteins called 'selenoproteins,' which contain the rare element selenium. Out of approximately 20,000 different proteins in our bodies, only 25 of them are selenoproteins. The UGGT partner Sep15 is a selenoprotein. Sep15 is always associated with UGGT. But until now, no one knew what it was doing there."
Using an AI model called AlphaFold2, Williams and his co-authors predicted that the protein Sep15 forms a complex helical shape that looks something like a catcher's mitt, and that this mitt perfectly matches a complementary site on the UGGT enzyme. The specific site where SEP15 and UGGT bind is also where UGGT reads the N-glycan code that tells it whether or not a protein is correctly folded.
"Basically," says Hebert, "we've found the hotspot where all the action is taking place-;and Sep15 is the key."
To test their AlphaFold2-generated prediction, the research team designed an experiment using recombinant DNA re-engineering of UGGT to interrupt its binding to Sep15-;and, indeed, the modified UGGT failed to form a complex with Sep15.
Related Stories
- New treatment boosts pluripotent stem cell production in mice
- Researchers develop new method to study critical type of protein modification process
- Spear Bio secures $45 million in oversubscribed Series A financing to accelerate product launch for protein research and disease diagnostics
So what, exactly, is Sep15 doing? "There are two possibilities, both of which we're following up on," says Hebert. "Either Sep15 is giving the misfolded protein a chance at correcting its shape, or it is marking that protein for destruction."
"The complexity of the proteins we are studying allows higher forms of life to function," says Gierasch, "but the complexity of those proteins also means that they're more prone to misfolding errors, and misfolding errors can have catastrophic consequences if the quality control process fails."
Though there is still a great deal of basic research to be done, the team's research sets the stage for novel drug therapies that target the Sep15/UGGT interface. "This is an untapped pharmaceutical area," says Hebert, "and Williams's research has moved us in the right direction for eventual treatment."
This research was supported by the National Institutes of General Medical Sciences and facilitated by the availability of instrumentation in the UMass Amherst Institute for Applied Life Sciences.
University of Massachusetts Amherst
Williams, R. V., et al. (2024) Insights into the interaction between UGGT, the gatekeeper of folding in the ER, and its partner, the selenoprotein SEP15 . PNAS . doi.org/10.1073/pnas.2315009121 .
Posted in: Cell Biology | Medical Science News | Medical Research News
Tags: Alzheimer's Disease , Biochemistry , Carbohydrate , Cell , Cellular Biology , Cystic Fibrosis , DNA , Emphysema , Enzyme , Fibrosis , Glycan , Glycans , Molecular Biology , Protein , Quality Control , Recombinant DNA , Research , Selenium
Suggested Reading

Cancel reply to comment
- Trending Stories
- Latest Interviews
- Top Health Articles

Addressing Important Cardiac Biology Questions with Shotgun Top-Down Proteomics
In this interview conducted at Pittcon 2024, we spoke to Professor John Yates about capturing cardiomyocyte cell-to-cell heterogeneity via shotgun top-down proteomics.

A Discussion with Hologic’s Tim Simpson on the Future of Cervical Cancer Screening
Tim Simpson
Hologic’s Tim Simpson Discusses the Future of Cervical Cancer Screening.

From Waste to Taste: The Transformative Power of Fermented Foods
Maria Marco
In this interview conducted at Pittcon 2024 in San Diego, Maria Marco discusses her research on the health benefits, safety, and waste reduction potential of fermented foods, and the microbial processes involved in their production.

Latest News

Newsletters you may be interested in
Your AI Powered Scientific Assistant
Hi, I'm Azthena, you can trust me to find commercial scientific answers from News-Medical.net.
A few things you need to know before we start. Please read and accept to continue.
- Use of “Azthena” is subject to the terms and conditions of use as set out by OpenAI .
- Content provided on any AZoNetwork sites are subject to the site Terms & Conditions and Privacy Policy .
- Large Language Models can make mistakes. Consider checking important information.
Great. Ask your question.
Azthena may occasionally provide inaccurate responses. Read the full terms .
While we only use edited and approved content for Azthena answers, it may on occasions provide incorrect responses. Please confirm any data provided with the related suppliers or authors. We do not provide medical advice, if you search for medical information you must always consult a medical professional before acting on any information provided.
Your questions, but not your email details will be shared with OpenAI and retained for 30 days in accordance with their privacy principles.
Please do not ask questions that use sensitive or confidential information.
Read the full Terms & Conditions .
Provide Feedback

IMAGES
COMMENTS
The first question asks for a ready-made solution, and is not focused or researchable. The second question is a clearer comparative question, but note that it may not be practically feasible. For a smaller research project or thesis, it could be narrowed down further to focus on the effectiveness of drunk driving laws in just one or two countries.
First, because each question may require a different research design, and second, because the necessary statistical power of the study would demand unaffordable sample sizes. It is the duty of editors and reviewers to make sure that authors clearly identify the primary research question, and as a consequence, studies approaching more than one ...
Keywords: evidence-based medicine, outcomes, research, questions. Clinical Scenario. You are a surgeon and your patient is a 25-year-old man with a severe type III open tibial fracture as a result of a motorcycle accident. The options for treatment are reconstruction or amputation. ... For example, in the tibia fracture question, the best ...
Systematic Reviews. 2. Develop a Research Question. A well-developed and answerable question is the foundation for any systematic review. This process involves: Using the PICO framework can help team members clarify and refine the scope of their question. For example, if the population is breast cancer patients, is it all breast cancer patients ...
Here, we'll explore a variety of healthcare-related research ideas and topic thought-starters across a range of healthcare fields, including allopathic and alternative medicine, dentistry, physical therapy, optometry, pharmacology and public health. NB - This is just the start….
Abstract. Formulation of research question (RQ) is an essentiality before starting any research. It aims to explore an existing uncertainty in an area of concern and points to a need for deliberate investigation. It is, therefore, pertinent to formulate a good RQ. The present paper aims to discuss the process of formulation of RQ with stepwise ...
a who (who was studied - a population or sample) a what (what was done or examined - an intervention, an exposure, a policy, a program, a phenomenon) a how ([how] did the [what] affect the [who] - an outcome, an effect). PICO is the most common framework for developing a clinical research question, but multiple question frameworks exist.
A well-crafted research question (or set of questions) sets the stage for a robust study and meaningful insights. But, if you're new to research, it's not always clear what exactly constitutes a good research question. In this post, we'll provide you with clear examples of quality research questions across various disciplines, so that you can approach your research project with confidence!
Sample Research Questions. Unclear: ... and how can these commonalities be used to aid the medical community in the prevention of the disease? The simple version of this question can be looked up online and answered in a few factual sentences; it leaves no room for analysis. The more complex version is written in two parts; it is thought ...
Step 3: Explore Study Design Formats. The next step is selecting the study format you want to use to gather your data. "People often ask me what the best study design is to use for their work. But there is no one right answer," Robertson says. "We tend to think randomized clinical trials have the highest level of evidence.
This easy-to-follow tutorial from the Librarians at the Bodleian Library at Oxford University, in partnership with the Centre for Evidence Based Medicine (CEBM), walks you through an example of turning a clinical research question into PICO format and using that to create search terms. <<
These issues result in health disparities and injustices. Examples of research topics about health inequities include: The impact of social determinants of health in a set population. Early and late-stage cancer stage diagnosis in urban vs. rural populations. Affordability of life-saving medications.
Step 1. Formulate the Research Question. A systematic review is based on a pre-defined specific research question (Cochrane Handbook, 1.1).The first step in a systematic review is to determine its focus - you should clearly frame the question(s) the review seeks to answer (Cochrane Handbook, 2.1).It may take you a while to develop a good review question - it is an important step in your review.
According to the Centre for Evidence Based Medicine (CEBM), "one of the fundamental skills required for practising EBM is the asking of well-built clinical questions. To benefit patients and clinicians, such questions need to be both directly relevant to patients' problems and phrased in ways that direct your search to relevant and precise answers."
Examples of some general health services research questions are: Does the organization of renal transplant nurse coordinators' responsibilities influence live donor rates? What activities of nurse managers are associated with nurse turnover? 30 day readmission rates? What effect does the Nurse Faculty Loan program have on the nurse researcher ...
A multi-institutional research team explored these questions in a scoping review. ... Stephanie Betancur explored this PICO question in both an Honors Thesis and an article. Labor & Delivery. ... Among staff nurses on a medical-surgical unit, do implementing bedside shift reports lead to greater satisfaction with care when compared to ...
Different types of clinical questions have certain kinds of studies that best answer them. The chart below lists the categories of clinical questions and the studies you should look for to answer them. In our clinical scenario, we are want to determine whether or not bariatric surgery will benefit the patient, so this is a therapy question.
23 Surgery Research Topics. 24 Radiology Research Paper Topics. 25 Anatomy and Physiology Research Paper Topics. 26 Healthcare Management Research Paper Topics. 27 Medical Ethics Research Paper Topics. 28 Environmental Health and Pollution Research Paper Topics. 29 Conclusion. Writing an original and compelling research paper is a daunting task ...
This is a libguide for praticing evidence-based medicine. When well built, clinical questions usually have four components: P: The patient situation, population, or problem of interest. I: The main intervention, defined very broadly, including an exposure, a diagnostic test, a prognostic factor, a treatment, a patient perception and so forth. C: A comparison intervention or exposure (also ...
Introduction and background. Successful clinical research can be conducted by well-trained researchers. Other essential factors of clinical research include framing a research question and relevant objectives, documenting, and recording research outcomes, and outcome measures, sample size, and research methodology including the type of randomization, among others [1,2].
Since the beginning of 2017, Cancer Communications (former title: Chinese Journal of Cancer) has published a series of important questions regarding cancer research and clinical oncology, to provide an enhanced stimulus for cancer research, and to accelerate collaborations between institutions and investigators. In this edition, the following 8 valuable questions are presented.
These are a few of the publication types associated with evidence-based practice taken from the Glossary of EBM Terms from the Centre for Evidence-Based Medicine Toronto. A more detailed list can be found in the Study Designs document from the National Library of Medicine.. Meta-analysis: A systematic review that uses quantitative methods to synthesize and summarize results of studies.
Asking focused questions. One of the fundamental skills required for practising EBM is the asking of well-built clinical questions. To benefit patients and clinicians, such questions need to be both directly relevant to patients' problems and phrased in ways that direct your search to relevant and precise answers.
Research Proposal Example Here is a research proposal sample template (with examples) from the University of Rochester Medical Center. 4 The sections in all research proposals are essentially the same although different terminology and other specific sections may be used depending on the subject. Structure of a Research Proposal
Background Patients from the lesbian, gay, bisexual, transgender, queer plus (LGBTQ +) community face various health inequalities and report poor healthcare experiences. Little is known about how knowledgeable and confident UK doctors are around LGBTQ + health, and previous research demonstrates that UK medical schools rarely deliver teaching in this area. This research evaluated the level of ...
Participants. We conducted an online survey of adult women between April 13, 2020, and June 8, 2020. Participants were drawn from the Qualtrics XM Panel, a database of potential online survey participants maintained by Qualtrics, XM and all respondents have already agreed to be contacted for research purposes. Criteria for inclusion were persons assigned female at birth, aged 27 to 45 years ...
By Sharon Brock, MEd, MS Like a Renaissance explorer, the insatiable curiosity of Jamie Zeitzer, PhD, drives him to explore big questions as a senior researcher and co-director of the Stanford Center for Sleep and Circadian Sciences. After 25 years of conducting sleep research, including groundbreaking studies on circadian rhythms and how light affects the […]
ADHD is highly prevalent in the general population and is common in autistic youth. If we can find ways to increase resources in these neighborhoods, we have the potential to improve academic, social, mental and physical health outcomes, particularly for autistic youth, and also decrease long-term economic costs."
Terms. While we only use edited and approved content for Azthena answers, it may on occasions provide incorrect responses. Please confirm any data provided with the related suppliers or authors.
Researchers at Uppsala University Hospital and Uppsala University have demonstrated that a simple blood test that reflects brain health can predict which people are most at risk of suffering a stroke.